Abstract
Introduction
Management of muscular dystrophies (MD) relies on conservative non-pharmacological treatments, but evidence of their effectiveness is limited and inconclusive.
Objective
To investigate the effectiveness of conservative non-pharmacological interventions for MD physical management.
Methods
This systematic review and meta-analysis followed Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines and searched Medline, CINHAL, Embase, AMED and Cochrane Central Register of Controlled Trial (inception to August 2022). Effect size (ES) and 95% Confidence Interval (CI) quantified treatment effect.
Results
Of 31,285 identified articles, 39 studies (957 participants), mostly at high risk of bias, were included. For children with Duchenne muscular dystrophy (DMD), trunk-oriented strength exercises and usual care were more effective than usual care alone in improving distal upper-limb function, sitting and dynamic reaching balance (ES range: 0.87 to 2.29). For adults with Facioscapulohumeral dystrophy (FSHD), vibratory proprioceptive assistance and neuromuscular electrical stimulation respectively improved maximum voluntary isometric contraction and reduced pain intensity (ES range: 1.58 to 2.33). For adults with FSHD, Limb-girdle muscular dystrophy (LGMD) and Becker muscular dystrophy (BMD), strength-training improved dynamic balance (sit-to-stand ability) and self-perceived physical condition (ES range: 0.83 to 1.00). A multicomponent programme improved perceived exertion rate and gait in adults with Myotonic dystrophy type 1 (DM1) (ES range: 0.92 to 3.83).
Conclusions
Low-quality evidence suggests that strength training, with or without other exercise interventions, may improve perceived exertion, distal upper limb function, static and dynamic balance, gait and well-being in MD. Although more robust and larger studies are needed, current evidence supports the inclusion of strength training in MD treatment, as it was found to be safe.
WHAT IS ALREADY KNOWN ON THIS TOPIC.
The management of muscular dystrophies (MD) primarily rely on conservative non-pharmacological interventions, including strength and aerobic training, orthotic devices, aids and assistive technologies, as there is currently no cure to halt MD progression. However, evidence supporting the effectiveness of these conservative non-pharmacological interventions is limited and inconclusive. This systematic review and meta-analysis was undertaken to better understand the effectiveness of these interventions on the physical manifestations of MD, including non-randomised and uncontrolled studies, which constitute the predominant body of literature in this field but have been omitted from prior evidence syntheses.
WHAT THIS STUDY ADDS
This study revealed that strength training, alone or combined with other exercise interventions, may improve perceived exertion, distal upper limb function, static and dynamic balance, gait and well-being in individuals with MD despite no significant structural changes.
HOW THIS STUDY MIGHT AFFECT RESEARCH, PRACTICE OR POLICY
These positive findings, although based on low-quality evidence, have possible clinical implications as they contradict historical apprehensions about the potential harm associated with exercise interventions in MD. Therefore, our findings suggest that strength training, whether as a standalone intervention or within a multicomponent programme, should be considered a fundamental component in the treatment of MD.
Introduction
Muscular dystrophies (MD) have an estimated prevalence ranging between 19.8 and 25.1/100,000.1 MD are a clinically and genetically heterogenous group of muscle disorders, sharing common dystrophic pathological features, such as variable muscle fibre size and necrosis, resulting in fatty or fibrotic tissue replacement and progressive muscle wasting and weakness.2 3 MD are commonly progressive and disabling over time, despite differences in their rate and pattern of progression with variation in how the limb, axial, facial, ocular and pharyngeal muscles are affected.4 Specific MD forms can also affect respiratory and cardiac muscles.5 These result in people with MD experiencing reduced motor functioning, social participation and quality of life.6
There is no cure to halt MD progression despite therapeutic advances in the most common forms of MD in the last two decades.7 Individuals with MD rely on surgical and non-pharmacological treatments, including strength and aerobic training, orthotic devices, aids and assistive technologies.8 Although conservative non-pharmacological interventions are essential part of the MD management, evidence supporting their effectiveness is limited and inconclusive.9 10 Despite some common impairments between MD, there may be pathobiological mechanisms unique to each MD subtype.2 Therefore, the effectiveness of an intervention may not be transferrable between MD subtypes as the therapeutic mechanism may not be optimal. Conservative interventions require appropriately tailoring to the symptoms and abilities of individuals with MD in order to be beneficial and not harmful. Inappropriate intervention type and dose can lead to detrimental effects, such as overwork weakness, fatigue, discomfort and pain.11
A wider understanding of the effects of conservative non-pharmacological interventions in people with MD is needed. This systematic review and meta-analysis aimed to determine the effectiveness of existing interventions, focusing only on those targeting physical impairments, excluding respiratory management.
Methods
This systematic review with meta-analysis was conducted using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines.12 The review protocol was not registered.
Literature search strategy
Medline, CINHAL, Embase, AMED and the Cochrane Central Register of Controlled Trial were systematically searched from inception to 22 August 2022. The search was supplemented by searching the reference lists of relevant articles and reviews. Search strategies available in online supplemental file 1.
jnnp-2023-331988supp001.pdf (225.8KB, pdf)
Study selection process
Records were exported into a reference management tool (Mendeley, Elsevier B.V.) and duplicates were removed. Any remaining duplicates were removed manually. One reviewer (EL) screened the records, with verification by two other researchers (FP, AP). Records were initially screened by title and abstract, followed by full-text analysis of relevant studies.
Inclusion and exclusion criteria
Studies were included if (1) involved humans with MD or neuromuscular diseases (NMD), data were separate for MD, (2) investigated conservative non-pharmacological interventions, alone or combined with other conservative non-pharmacological therapies, (3) had ≥5 participants per group, (4) had full texts available and (5) published in peer-reviewed journals.
Studies were excluded if (1) included pharmacological or surgical treatments, (2) unavailable data, (3) assessed conservative non-pharmacological interventions for respiratory and cognitive manifestations and (4) solely focused on non-pertinent outcomes (e.g. respiratory and cognitive outcomes).
Authors were contacted for missing data. If no response or unextractable data, studies were excluded.
Data extraction
One reviewer (EL) extracted data (online supplemental file 2). Non-English articles were translated by the research team where appropriate.
jnnp-2023-331988supp002.pdf (363.1KB, pdf)
Quality assessment
Two reviewers (EL, FP) independently assessed the methodological quality of the included Randomised Controlled Trials (RCTs) using the Cochrane ‘Risk of Bias’ Tool.13 Studies with research designs other than RCT are by nature at high risk of bias, and so no formal quality appraisal was undertaken. Uncertainties and disagreements between reviewers were resolved in team discussions.
Statistical analysis
ES was calculated using Cohen’s d formula.14 As most available evidence comes from non-randomised, uncontrolled studies, ES calculation was performed in all studies, including quasi-controlled studies, although they were a priori considered at high risk of bias. Further information about ES calculation is provided in online supplemental file 3. The absolute magnitude of ES was classified as small (0.20–0.49), medium (0.50–0.79), large (0.80–1.29) and very large (≥1.3).14 If the 95% Confidence Interval (CI) range of the ES did not include zero, the result was considered statistically significant. Included studies were primarily clustered based on the type of intervention tested (orthotic devices, manual therapy, assistive technologies and exercise interventions), secondly by intervention subtype.
jnnp-2023-331988supp003.pdf (328.3KB, pdf)
ESs were reported for the intervention types and subtypes, and outcomes were mapped to the International Classification of Functioning, Disability and Health (ICF).15 Formal heterogeneity assessment was not conducted as statistical heterogeneity was expected due to variability in sample sizes, MD type, conservative non-pharmacological treatment types and outcome measures. Following data extraction, ES data were normalised. Regardless of the direction of change in the outcome, positive signs were used to represent positive changes from baseline, while negative signs were used to indicate negative changes with scores worsening compared with baseline.
Results
Study identification
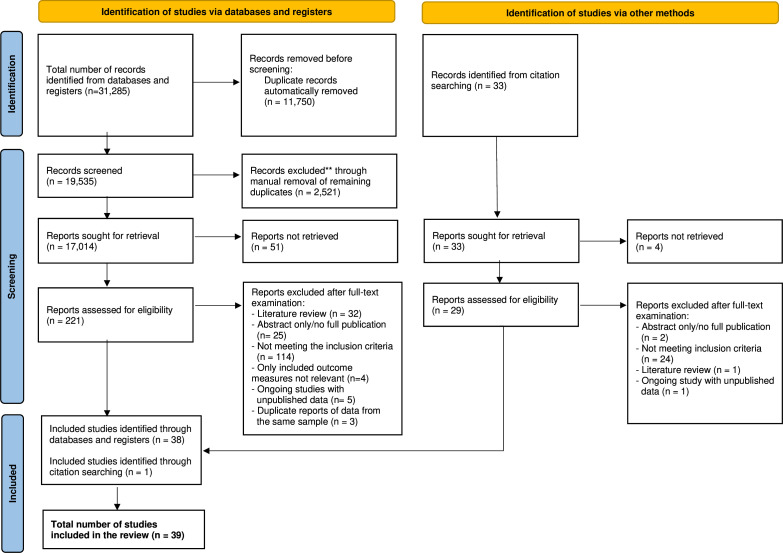
The search identified 31,285 articles, reduced to 17,014 after removing duplicates. After screening the titles and abstracts, 221 were deemed suitable for full-text review; 38 of them fulfilled the eligibility criteria and were included. A further manual search identified 33 additional articles; of them, 29 progressed to full-text review and once reviewed, one was considered eligible for this study. Finally, a total of 39 articles were included. Figure 1 describes the study selection process.
Figure 1.
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow chart of the study selection process. CBT, Cognitive Behavioural Therapy.
Study characteristics
Included studies comprised 15 RCTs and 21 quasi-controlled studies, predominantly before–after designs without a control group (18/21). Most studies were published in the last decade (74%, 29/39), mainly from Europe (69%, 27/39). Included studies involved a total of 957 participants (range: 6–255). DMD (n=13 studies), DM1 (n=11 studies) and FSHD (n=8 studies) were the most explored MD forms (online supplemental file 4). Most cohorts consisted of adults only (67%, 26/39). All except one16 of the paediatric studies involved children with DMD. Most studies had a male predominance (male:female ratio=2:1). Most studies included ambulatory participants, but mobility levels were variable, ranging from independently mobile,17–22 mobile with or without assistive aids23–28 to wheelchair dependent.29 30 Similarly, upper-limb abilities varied from grade one, where individuals could lift their arms above their head, to grade four on the Brooke Upper-Extremity Scale,31 where they could raise hands to mouth, but they could not bring a glass of water to their mouth. Further study characteristics supplied in online supplemental file 5.
jnnp-2023-331988supp004.pdf (401.3KB, pdf)
jnnp-2023-331988supp005.pdf (228.2KB, pdf)
Risk of bias
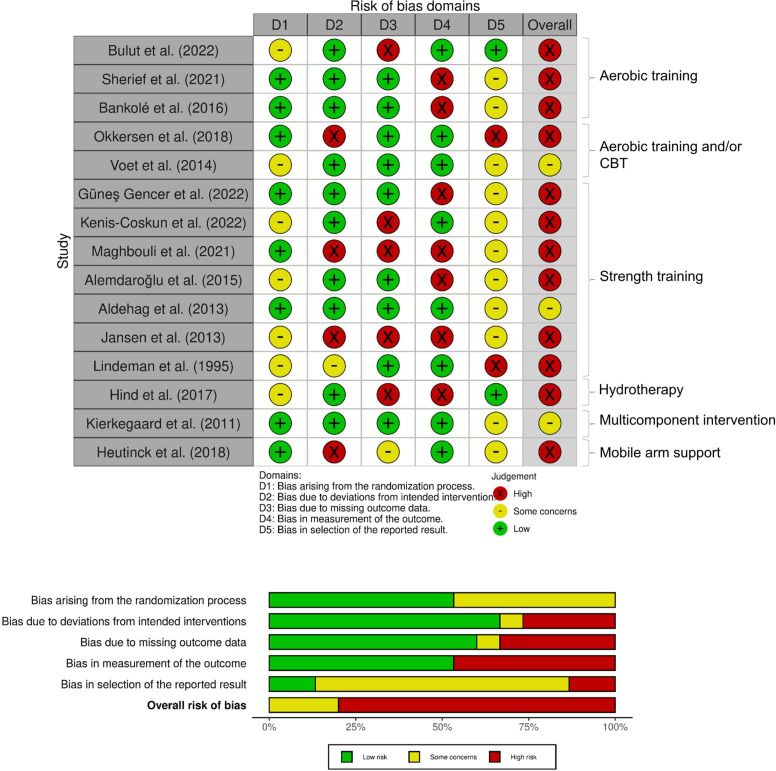
Figure 2 shows the risk of bias for the included RCTs. Twelve of the 15 RCTs17 23 28–30 32–38 were considered at high risk of bias and ‘some concerns’ arose for three RCTs.20 24 39 All studies confirmed randomisation, but four studies29 36–38 did not report concealed allocation information. Baseline differences between study groups were observed in three studies.17 24 35 Patient participants were not blinded in any of the RCTs, but six studies20 24 30 35 38 39 reported blinding of the assessors. Nevertheless, almost 50% of studies (7/15)17 28 29 32–34 36 were considered at high risk of bias in outcome measurement as they adopted assessor dependent outcome measures. Eight studies3 17 23 28–30 35 38 had attrition bias due to missing outcome data or study participants’ drop out17 23 28–30 35 38 39; however, most studies adopted an appropriate analysis to explore the effect of assignment to intervention. Only one study documented protocol deviations.23 Thirteen studies did not provide a statistical analysis plan, causing selection bias risk.
Figure 2.
Risk of bias summary for randomised controlled trials only.
Conservative non-pharmacological interventions
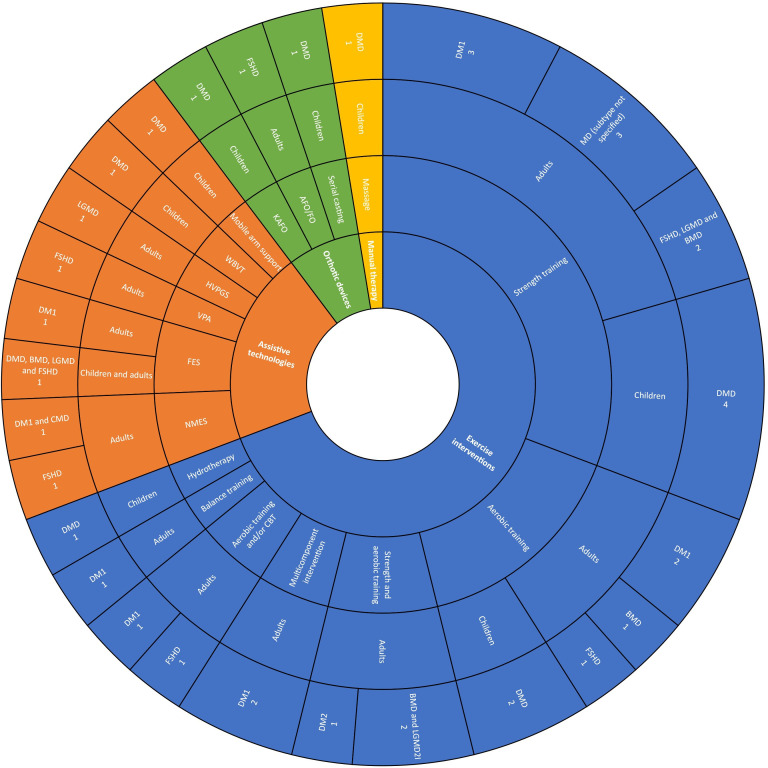
Conservative non-pharmacological interventions were classified into four types: orthotic devices, manual therapy, assistive technologies and exercise interventions. An overview of the intervention types, subtypes and MD subgroups studied is presented in figure 3. Further details are available in online supplemental files 6 and 7.
Figure 3.
Sunburst chart of the characteristics of the included studies. AFO, Ankle Foot Orthosis; BMD, Becker Muscular Dystrophy; CBT, Cognitive Behavioural Therapy; CMD, Congenital Myotonic Dystrophy; DM1, Myotonic Dystrophy type 1; DM2, Myotonic Dystrophy type 2; DMD, Duchenne Muscular Dystrophy; FES, Functional Electrical Stimulation; FO; Foot Orthosis; FSHD, Facioscapulohumeral Dystrophy; HVPGS, High Voltage Pulsed Galvanic Stimulation; KAFO, Knee Ankle Foot Orthosis; LGMD, Limb Girdle Muscular Dystrophy; LGMD2l, Limb Girdle Muscular Dystrophy type 2l; MD, Muscular Dystrophy; NMES, Neuromuscular Electrical Stimulation; VPA, Vibratory Proprioceptive Assistance; WBVT, Whole-Body Vibration Training.
jnnp-2023-331988supp006.pdf (433.9KB, pdf)
jnnp-2023-331988supp007.pdf (256.5KB, pdf)
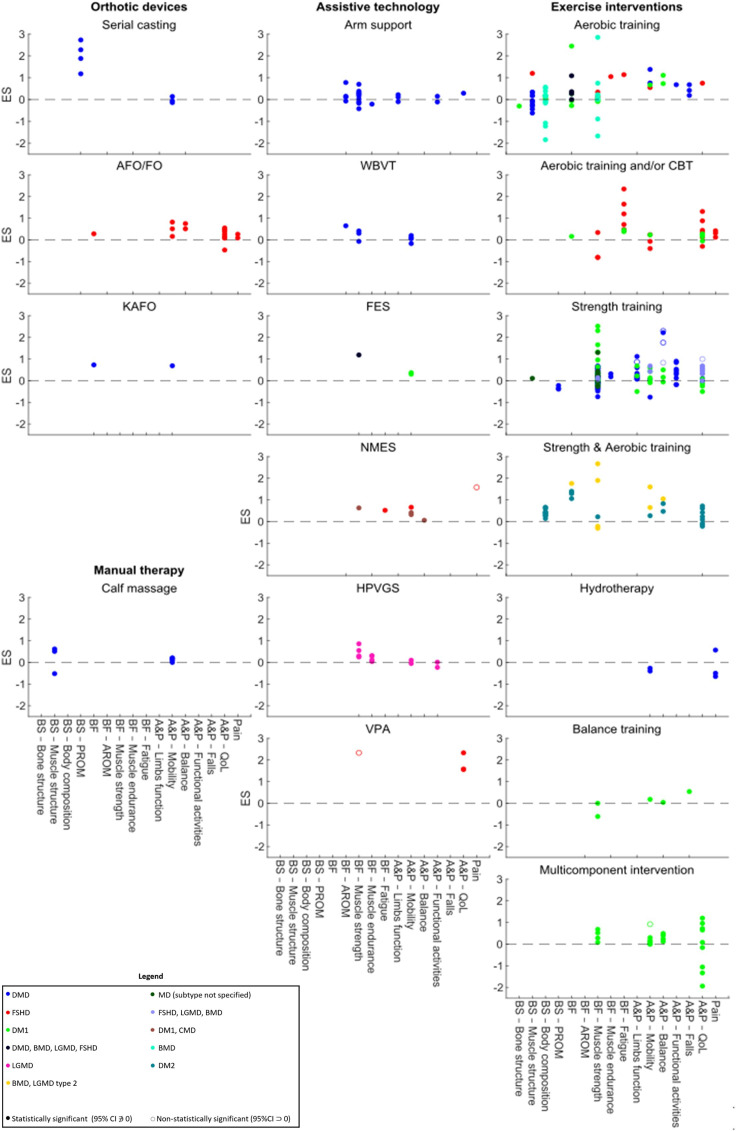
Of the 39 included studies, seven 19–21 25 34 36 40 (three RCTs20 34 36 and four non-RCTs19 21 25 40) observed a statistically significant treatment effect in one or more of their outcomes, with a total of nine outcome measures being improved. Significant improvements were mainly seen in functional outcomes (5/9),21 25 32 34 mostly pertaining balance abilities (3/9).25 34 Most outcomes (7/9) improved following supervised exercise interventions.21 25 34 36 40 Seven of the nine outcomes, which included perceived exertion, distal upper-limb function, gait, static and dynamic balance and self-perceived physical condition, improved following training programmes including strength exercises alone25 40 or combined with other exercise modalities (e.g. aerobic and balance training).20 21 34 Non-exercise interventions significantly improved only a small number of outcome measures (2/9).19 41 The largest significant ES was observed in Borg Rating of Perceived Exertion (RPE) (ES: 3.83 (95% CI: 2.86 to 4.79)),20 and the smallest significant ES was observed in dynamic balance (sit-to-stand ability) following strength training (ES: 0.83 (95% CI: 0.08 to 1.58)).25 There were a large number of outcomes, and many studies reported non-significant treatment effects. Therefore, the following paragraphs will provide a narrative overview only of those intervention subtypes with fewer studies. For intervention subtypes with a large number of studies, only the largest and significant ESs will be reported. Results for all interventions are displayed in figure 4 with further details presented in online supplemental file 8. A graphical representation of statistically significant ESs across all included studies is presented in figure 5.
Figure 4.
Scatter plots of the effect sizes of the included conservative non-pharmacological interventions. A&P, Activity and Participation; AFO/FO, Ankle Foot Orthosis/Foot Orthosis; AROM, Active Range of Motion; BF, Body Function; BMD, Becker Muscular Dystrophy; BS, Body Structure; CBT, Cognitive Behavioural Therapy; CMD, Congenital Muscular Dystrophy; DM1, Myotonic Dystrophy type 1; DM2, Myotonic Dystrophy type 2; DMD, Duchenne Muscular Dystrophy; ES, Effect Size; FES, Functional Electrical Stimulation; FSHD, Facioscapulohumeral Muscular Dystrophy; HPVGS, High Volt Pulsed Galvanic Stimulator; KAFO, Knee Ankle Foot Orthosis; LGMD, Limb-Girdle Muscular Dystrophy; MD, Muscular Dystrophy; NMES, Neuromuscular Electrical Stimulation; PROM, Passive Range Of Motion; QoL, Quality of Life; VPA, Vibratory Proprioceptive Assistance; WBVT, Whole-Body Vibration Training.
Figure 5.
Forest plot of the significant effect sizes in the included studies. ES, Effect Size; MVIC, Maximum Voluntary Isometric Contraction; NMES, Neuromuscular Electrical Stimulation; PSPP-R, Physical Self-Perception Profile Revised; PUL, Performance of Upper Limb; RPE, Rating of Perceived Exertion; STS, Sit To Stand; TCMS, Trunk Control Measurement Scale; VAS, Visual Analogue Scale; VPA, Vibratory Proprioceptive Assistance.
jnnp-2023-331988supp008.pdf (298.3KB, pdf)
Orthotic devices
Three studies42–44 (two43 44 before–after studies without control group and one42 case series study) involving a total of 33 participants investigated orthotic devices (serial casting42 and AFO/FO43 in children with DMD, and KAFO in adults with FSHD44). None of the improvements recorded were statistically significant; positive ESs ranged from 0.09 (VAS pain after AFO/FO43) to 2.73 (ankle dorsiflexion passive range of motion after serial casting42).
Manual therapy
One crossover study18 investigated calf massage in children with DMD. Small to moderate ESs observed in lower-limb muscles length and gait were not statistically significant. ESs ranged from −0.52 in knee extension range of motion after calf massage to 0.62 in ankle dorsiflexion range of motion with knee extended.
Assistive technologies
Nine studies,16 19 22 30 41 45–47 of which all except one30 had before–after study design, explored assistive technologies interventions. Sample sizes ranged from six to 24 participants, totaling 97 participants, with study durations of two to 20 weeks. Overall evidence quality was low, as eight studies were at high risk of bias due to research designs. The only RCT30 had methodological limitations due to deviations from the intended intervention.
Gravity-compensated training with a suspension mobile arm support,30 whole-body vibration training,45 high volt pulsed galvanic stimulator22 and functional electrical stimulation16 47 did not produce statistically significant treatment effects. In contrast, NMES significantly reduced pain in nine adults with FSHD (ES: 1.58 (95% CI: 0.61 to 2.55)).19 Furthermore, vibratory proprioceptive assistance (VPA) (4-week training, 8 total sessions at 80 Hz) significantly improved shoulder abduction muscle strength in nine adults with FSHD (ES: 2.33, 95% CI: 1.32 to 3.34).41 ESs across studies ranged from −0.42 in shoulder abduction strength after arm support30 to 2.33 in both shoulder abduction muscle strength and self-rated health state after VPA.41
Exercise interventions
A total of 27 studies,17 20 21 23–29 32–40 48–55 including 14 RCTs,17 20 23 24 28 29 32–39 12 before–after studies25–27 29 40 48–50 52–55 (three with a control group49 54 55) and one retrospective study,21 explored exercise interventions lasting from four weeks to 10 months (807 total participants, range: 6–255). Interventions included strength training or aerobic training, alone or in combination with each other or other conservative treatments like cognitive–behavioural therapy (CBT), balance training and hydrotherapy. Overall evidence quality was low, with 13 studies at high risk of bias due to study design. Fourteen RCTs were at either moderate or high risk of bias, predominantly due to assessor-dependent outcome measures, attrition and inappropriate analysis of intervention assignment.
ESs ranged from −1.94 in the social functioning item of the 36-Item Short Form Survey (SF-36) after a multicomponent intervention20 to 4.00, observed in lunges performance after an intervention combining aerobic and strength training.51
Aerobic training
Six studies32 33 38 53–55 used aerobic training as an intervention, with five studies32 38 53–55 using a cycle ergometer, either solely for lower-limb training32 38 53 54 or compared with a treadmill.32 One study37 used a stationary ergocycle for lower-limb training only. Half of the studies were RCTs32 33 38 and half were before–after studies53–55 (two had a control group54 55). A total of 115 participants underwent aerobic training (range: 11–30 participants), with follow-up periods ranging from 12 weeks to 12 months. Quality of evidence was low due to a high risk of bias resulting from missing outcomes and outcome measurement.
None of the aerobic exercise interventions yielded statistically significant effects in the measured outcomes.
Aerobic training and/or CBT
Two RCTs23 24 explored aerobic training and CBT, given alone, in combination with each other or in addition to usual care, in 312 adults with FSHD24 and DM1.23 Okkersen et al 23 found no superiority of CBT and aerobic training combined with usual care over usual care alone (ES range: −0.05 to 0.45). Despite improvements in fatigue (ES range: 0.41 to 2.35) and quality of life (ES range: 0.12 to 1.31), Voet et al. 24 showed no statistically significant changes in any outcomes.
Strength training
Twelve studies,25–29 34–37 39 40 49 predominantly using RCT design (7/12),28 29 34–37 39 explored strength interventions in 276 participants (range: 9–40 participants). Four29 34–36 studies involved children with DMD, while the remaining studies25–28 37 39 40 49 examined adults with DM1, DM2, FSHD, LGMD, BMD and unspecified MD subtypes. The quality of evidence for RCTs ranged from ‘some concerns’ to ‘high’ due to moderate risk of selection bias and high risk of bias from missing data and outcome measurement.
Strength training interventions did not yield statistically significant changes in terms of muscle structure,49 passive range of motion,29 muscle strength25–29 35–37 39 and mobility.25–27 29 35 36 Nevertheless, Güneş-Gencer et al 34 found that eight weeks of trunk-oriented training combined with conventional care was significantly more effective than conventional care alone for improving distal wrist and hand function (PUL distal subscore) (ES: 0.87 (95% CI: 0.04 to 1.69)) in children with DMD.
Balance abilities significantly improved following strength training in two studies.25 34 Güneş Gencer and Yilmaz34 observed that eight-week trunk-oriented exercises combined with usual care were more effective than usual care alone in improving static sitting (ES: 1.76 (95% CI: 0.82 to 2.71)) and dynamic reaching balance (ES: 2.29 (95% CI: 0.37 to 4.22)) in children with DMD. Additionally, Bostock et al 25 observed that the timed sit to stand test of adults with FSHD, LGMD and BMD significantly improved after a 12-week strength training (ES: 0.83 (95% CI: 0.08 to 1.58)).
In adults with FSHD, LGMD and BMD, the same research group25 observed a significant improvement in self-perception of physical condition (ES: 1.00 (95% CI: 0.17 to 1.82)).40 Strength training did not improve any other quality-of-life domains (ES range: −0.50 to 0.67).29 35 39 40
Strength and aerobic training
Three before–after studies without control group50–52 adopted intervention programmes involving strength and aerobic training in a total of 26 adults (range: 8–10 participants) with DM2, BMD and LGMD2I. One study52 used a four-week programme (two times/week) on a stationary bicycle, while the other two studies,50 51 from the same research group, used a 10-week programme (three times/week, 40 min/session) on an antigravity treadmill. No statistically significant differences were noted in the outcome measures assessed.
Hydrotherapy
One RCT17 involving 12 children with DMD found that six-month hydrotherapy combined with land-based exercises had non-significant effects on all outcome measures (ES range: −0.65 to −0.27).
Balance training
One uncontrolled before–after study48 with 11 adults with DM1 explored a group balance programme (one to three times/week, 60 min/session, for 10 weeks), reporting non-significant treatment effects.
Multicomponent interventions
Two studies,20 21 one RCT20 and one retrospective study,21 used multicomponent rehabilitation programmes, comprising flexibility, aerobic, strength and balance training, for a total of 55 adults with DM1. One study21 showed significantly greater fast walking speed (ES: 0.92 (95% CI: 0.26 to 1.57)), while the other31 observed statistically significant changes in the Borg RPE score after training (ES: 3.83 (95% CI: 2.86 to 4.79)).
Adverse events
Over 35% (14/39) of studies reported adverse events, with low frequency among participants. The most reported adverse event was pain (n=33 participants)24 27 29 30 37 42 45 49 which mostly affected the lower back24 27 37 and lower limbs.29 49 Other adverse events were muscle soreness,17 cramp,45 fatigue,53 falls,17 23 foot fracture,45 physical discomfort,49 skin redness19 42 and skin irritation.45 Most participants successfully completed programmes with only transient symptoms consistent with the interventions. Okkersen et al 23 reported that that 50% of the participants (n=255) experienced adverse events, which were serious in 13% of the cases. However, not all reported adverse events were directly attributed to the exercise interventions (e.g. upper respiratory tract infection and neoplasm). Another three studies17 29 30 reported adverse events unrelated to the intervention tested. Only one study27 reported significant long-term adverse events (low back pain) from exercise protocol. Only two patients dropped out due to adverse events (back pain) related to the intervention.37 49
Discussion
This systematic review and meta-analysis explored the effectiveness of conservative non-pharmacological interventions for the MD physical management. Although various improvements were observed, most intervention effects were non-significant. Low-quality evidence suggests that exercise interventions incorporating strength training may enhance functional abilities. Specifically, a multicomponent rehabilitation programme, consisting of strength, flexibility and balance training, improved gait speed in adults with DM1.20 Additionally, adults with FSHD, LGMD and BMD25 showed enhanced dynamic balance (sit-to-stand ability) after strength exercises. For children with DMD, trunk-oriented strength exercises combined with usual care were more effective than usual care alone to improve distal upper-limb function, static sitting and dynamic reaching balance.34 Interestingly, these functional improvements occurred without substantial muscle strength changes. Only one study implementing VPA in FSHD41 found statistically significant muscle strength changes. This may suggest that conservative non-pharmacological interventions, including strength training, may help maintain, rather than improve, muscle strength in MD. However, the strength interventions durations did not exceed six months, making it unclear whether continuous interventions can maintain muscle strength in the long term despite the MD progressive nature. Furthermore, it is important to acknowledge the divergence in muscle strength findings; while some studies reported non-significant improvements, others observed non-significant declines after training. This decline could be attributable to the MD degenerative nature, but the exact cause remains uncertain, as the variability in intervention characteristics (e.g. intensity, duration) and study populations (e.g., disease type and severity) where this was observed makes it difficult to determine the responsible factors.
The positive findings from strength training interventions have possible clinical implications given historical concerns about exercise interventions potentially causing harm in MD.56 They support the use of strength training, either independently or with complementary exercise interventions, as this approach not only has proven to be safe when carried out using existing parameters and could be cost-effective, but also seem to ensure an efficient training stimulus.9 Using strength training in multimodal interventions may address multiple impairments simultaneously. While intervention characteristics were variable, the strength training component within interventions that yielded statistically significant differences typically included supervised active or active-assisted strength exercises, using free weights or exercise machines. Exercise sessions generally took place twice a week (ranging from twice a week to daily) for eight weeks, and each session lasted approximately 40–45 min (range: 40 min to two hours); exercises typically consisted of five to 10 repetitions for one to three sets. While available studies suggest the beneficial effects of this dosage, there is not yet sufficient evidence to make recommendations. Notably, supervision may have influenced health behaviour change and the effectiveness of these interventions, resulting from education, opportunities and capability which facilitate adherence levels.57 It is important to note that strength interventions improved balance abilities, while balance-specific programmes failed to yield any significant balance changes.47 This may indicate that (1) strength training, alone or combined with other exercise forms, may be more beneficial than balance-specific exercises alone; (2) strength loss significantly impacts other body structures and activities and (3) some balance assessments (e.g. timed up and go test) may show improvements reflective of muscle strength changes. These observations also suggest that intervention effectiveness assessment through functional outcomes may be more appropriate than relying solely on strength measures, considering that muscle strength improvements may not always occur due to progressive muscle weakness in these conditions.
Most non-exercise interventions (manual therapy, orthotic devices and assistive technologies interventions) did not produce statistically significant treatment effects. This was likely due to study limitations, including small sample sizes, lack of blinding, missing data and dropouts. These limitations were more pronounced in non-exercise interventions due to their smaller scale, potentially explaining their lower frequency of significant results compared with exercise interventions. Non-exercise intervention studies typically involved the smallest sample sizes (range: 9–24 participants), further exacerbated by dropouts observed in a third of the non-exercise intervention studies. Furthermore, most non-exercise interventions were tested within single MD clinical groups, leaving treatment effects variations across MD conditions unexplored. Unlike exercise interventions that often used more structured and supervised regimens, non-exercise interventions were primarily unsupervised, leaving adherence rates uncertain and potentially influencing their effectiveness. Additionally, the assessment of non-exercise interventions focused on a limited number of outcome measures, which may have not captured the full spectrum of potential effects, possibly leading to an underestimation of their impact. Despite most non-exercise interventions lacking statistical significance, it is important to acknowledge that many of them are commonly integrated into the MD clinical management or recommended on an individual basis. In the absence of a robust evidence base, healthcare professionals and clinical services use these interventions as they are deemed to be acceptable, feasible, and beneficial by healthcare providers and people with MD. With no cure available, offering these conservative non-pharmacological interventions addresses symptoms, meets patient expectations, and empowers healthcare professionals to make a positive impact on their patients' lives. For individuals living with MD, receiving these interventions goes beyond merely alleviating physical symptoms; it potentially cultivates hope and fosters resilience, improving their overall experiences of living with MD. In clinical practice, these interventions target impairments affecting fatigue levels, psychological well-being, independence, engagement in social activities and quality of life. However, the perceived benefits of these interventions haven not been substantiated by this systematic review and meta-analysis due to inherent study limitations, rather than the ineffectiveness of the interventions themselves, which may have confounded their true effects, making them appear less impactful than they are in routine clinical practice.
Existing guidelines for conservative non-pharmacological management of MD, as well as other NMD, are limited56 or neglect conservative non-pharmacological approaches58 due to the absence of robust evidence supporting their clinical benefits, negatively affecting clinical decision-making.59 While existing guidelines often overlook these interventions, their role within an evidence-based practice framework, their potential efficacy, their safety and the absence of alternative treatments may justify their clinical use, especially strength training alone or combined with other exercise forms. However, to employ conservative non-pharmacological interventions of which evidence is yet to fully become available, these interventions should align with first principles, be acceptable to patients and healthcare professionals, pose no harm or financial burden, and be continually reviewed for adherence to evidence-based practice. Where the evidence suggests treatment is beneficial (e.g. strength training), we should continue to work towards monitoring risk of harm while also exploring how to optimise the prescriptions to maximise benefits. Furthermore, while this review and meta-analysis did not include studies combining conservative non-pharmacological treatments with pharmacological interventions, preventing a direct comparison of their effectiveness, previous research in MD and NMD suggests that conservative non-pharmacological interventions have comparable, if not superior, effectiveness than pharmacological treatments.60 61 For instance, very large, although non-significant, ESs (range: 1.46–8.93) strongly favoured conservative interventions over pharmacological treatments in specific outcomes (e.g. Berg balance, grip strength, 2-min walk test, physical functioning, role limitations due to physical health and emotional problems) in the study by Connor et al.60
Numerous questions regarding the effectiveness of these interventions remain unanswered, specifically about certain outcomes, populations and intervention types. Pain, a common and distressing symptom in MD,62 63 offers anecdotal indications of potential relief through non-pharmacological interventions such as hydrotherapy. However, the impact of conservative interventions on pain is underexplored, with most studies failing to demonstrate statistically significant improvements. Only a small-scale NMES study in adults with FSHD reported a significant reduction in pain.19 Furthermore, most studies involved adults only, and all except one16 of the studies involving children were conducted in DMD. No studies have explored the effectiveness of conservative non-pharmacological interventions in infantile and juvenile cases of MD forms other than DMD. Additionally, there is limited exploration of interventions in the early stages of these conditions. Most studies focused on exercise interventions, leaving the effectiveness of non-exercise interventions unknown. Studies on conservative non-pharmacological interventions in adults with DMD are scarce, and the effectiveness of upper-limb orthoses remain unclear.
Future studies are required to advance current understanding of the effectiveness of conservative non-pharmacological interventions for MD. To overcome existing challenges and conduct studies that genuinely capture the effectiveness of these interventions, several actions are imperative.
Alternative study designs and methods for determining effectiveness of therapy interventions in MD are needed. While RCTs remain the gold standard for assessing intervention effectiveness, the diseases rarity, MD phenotypic heterogeneity, logistical complexities and patients’ geographical dispersion often make RCTs challenging. Where existing treatment options are limited or non-existent, people with MD usually prefer access to an active intervention rather than a placebo. Within a research landscape increasingly focused on developing disease-modifying treatments, individuals may find participation in drug trials more attractive than enrolling in conservative non-pharmacological intervention studies. This preference can hinder the recruitment of participants for future non-pharmacological clinical research, as this may also make them ineligible. Additionally, RCTs can be cost-prohibitive, and the substantial costs may not align with the priorities of funding bodies, which may prefer to allocate resources to research in more common diseases. While government research councils have offered funding schemes, these opportunities are relatively limited, and industry or charitable organisations often serve as primary research funding sources in this field. Relying on non-governmental sources can complicate building sustainable research infrastructure, requiring innovative funding and collaborative efforts to advance MD research effectively.
A universally agreed core outcome set for evaluating therapy interventions in MD is still lacking. Validated outcome measures specific to MD are often scarce, necessitating the use of measures validated for other conditions. However, differences in disease mechanisms, progression, manifestations and the heterogeneity of MD diseases can compromise the validity of these extrapolated measures. Additionally, outcome measures designed for different clinical populations may lack face validity for some MD subgroups, potentially leading to underestimation of the interventions’ significance and inaccurate conclusions regarding effectiveness. The absence of standardised core outcome sets also complicates the intervention effectiveness assessment and hinders meaningful comparisons between studies. Future efforts should focus on developing patient-centred core outcome sets, which should reflect what is important to individuals living with different MD, account for the unique MD progressions, establish clinically meaningful differences and ensure measures are valid and reliable.
Registries and clinical databases serve as valuable tools for gathering data on individuals with NMD.64 In cases where evidence is scarce, they offer a potentially sustainable approach to determining effectiveness.65 66 However, their effectiveness depends on the engagement of patient and centres, integration of relevant electronic health records, consideration of measurement burden, appropriate outcome measures inclusion, high data completion rates and regular updates in response to emerging evidence. While existing registries have been used for exploratory research, their utility in informing clinical decision-making has not been fully developed, possibly owing to a failure to address the requirements above.
The scarcity of comprehensive natural history data in MD poses a significant challenge for assessing intervention effectiveness. Without this baseline knowledge, distinguishing intervention effects from natural disease progression is difficult, as exemplified by the challenging interpretation of strength decline following strength training. Additionally, the lack of natural history data affects decisions on intervention timing and duration. Robust natural history studies are necessary to provide insights into disease progression, guide treatment strategies and establish a benchmark for intervention impact evaluation.
Taken together, these actions will enhance research quality, inform guidelines, support healthcare professionals, standardise care across clinical sites and ultimately improve care for individuals with MD.
Strengths
The analysis of the effectiveness of conservative non-pharmacological interventions in MD was comprehensive. This was achieved by search strategies without restrictions on date, language, study designs and outcomes.
Limitations
Most data came from non-RCT studies, which may have limitations such as overestimated treatment effects, uncontrolled designs and small sample sizes. This study focused on the conservative non-pharmacological interventions for the MD physical manifestations, excluding respiratory interventions that however are integral to the non-pharmacological management of these conditions. Included articles predominantly involved adults, and paediatric studies consisted largely of children with DMD, which could limit the applicability of the findings to children with MD forms other than DMD. Limitations also included heterogeneity in the study populations and outcome measures, use of non-validated outcome measures and, in some studies, inclusion of multiple patient populations whose data could not be separated by individual conditions. Future studies should present separate MD group data to identify specific group intervention interactions.
Conclusion
This systematic review and meta-analysis indicated that most conservative non-pharmacological interventions produced non-statistically significant treatment effects. Nevertheless, identified interventions did not appear to be harmful. Furthermore, low-quality evidence suggested that supervised strength training, alone or combined with other exercise interventions, may improve perceived exertion, distal arm function, balance, gait and well-being despite no significant structural changes. Therefore, strength training, alone or as part of a multicomponent programme, should be considered as the mainstay in the treatment of MD. Evidence on the effectiveness of assistive technologies, orthotic devices and manual therapy interventions remains limited and inconclusive. Future large-scale, well-designed and well-reported studies are needed to clarify the role of non-pharmacological interventions in MD and to support their clinical use.
Footnotes
@leone_enza
Contributors: EL: contribution to the design and conceptualisation of the study, acquisition, analysis and interpretation of the data, drafting and revising the manuscript, and final approval of the version to be published. AP: supervision of the study, contribution to design and conceptualisation of the study, analysis and interpretation of the data, revising the manuscript and final approval of the version to be published. AR, RK and JH: contribution to the design and conceptualisation of the study, revising the manuscript and final approval of the version to be published. FP: supervision of the study, contribution to the design and conceptualisation of the study, analysis and interpretation of the data, drafting and revising the manuscript, and final approval of the version to be published.
EL, as the study guarantor, accepts full responsibility for the finished work and/or the conduct of the study, had access to the data, and controlled the decision to publish.
Funding: This work was conducted as part of a PhD program funded by the Faculty of Medicine and Health Sciences Doctoral Fellowship Scheme at Keele University. The funder was not involved in the review.
Competing interests: None declared.
Provenance and peer review: Not commissioned; internally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
All data relevant to the study are included in the article or uploaded as supplementary information.
Ethics statements
Patient consent for publication
Not applicable.
Ethics approval
Not applicable.
References
- 1. Theadom A, Rodrigues M, Roxburgh R, et al. Prevalence of muscular dystrophies: a systematic literature review. Neuroepidemiology 2014;43:259–68. 10.1159/000369343 [DOI] [PubMed] [Google Scholar]
- 2. Mercuri E, Bönnemann CG, Muntoni F. Muscular dystrophies. Lancet 2019;394:2025–38. 10.1016/S0140-6736(19)32910-1 [DOI] [PubMed] [Google Scholar]
- 3. Lovering RM, Porter NC, Bloch RJ. The muscular Dystrophies: from genes to therapies. Phys Ther 2005;85:1372–88. 10.1093/ptj/85.12.1372 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Mercuri E, Muntoni F. Muscular dystrophies. Lancet 2013;381:845–60. 10.1016/S0140-6736(12)61897-2 [DOI] [PubMed] [Google Scholar]
- 5. Verhaert D, Richards K, Rafael-Fortney JA, et al. Cardiac involvement in patients with muscular dystrophies: magnetic resonance imaging phenotype and genotypic considerations. Circ Cardiovasc Imaging 2011;4:67–76. 10.1161/CIRCIMAGING.110.960740 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Grootenhuis MA, de Boone J, van der Kooi AJ. Living with muscular dystrophy: health related quality of life consequences for children and adults. Health Qual Life Outcomes 2007;5:31. 10.1186/1477-7525-5-31 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Leung DG, Wagner KR. Therapeutic advances in muscular dystrophy. Ann Neurol 2013;74:404–11. 10.1002/ana.23989 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Cup EH, Pieterse AJ, Ten Broek-Pastoor JM, et al. Exercise therapy and other types of physical therapy for patients with neuromuscular diseases: a systematic review. Arch Phys Med Rehabil 2007;88:1452–64. 10.1016/j.apmr.2007.07.024 [DOI] [PubMed] [Google Scholar]
- 9. Voet NB, van der Kooi EL, van Engelen BG, et al. Strength training and aerobic exercise training for muscle disease. Cochrane Database Syst Rev 2019;12:CD003907. 10.1002/14651858.CD003907.pub5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Pedlow K, McDonough S, Lennon S, et al. Assisted standing for duchenne muscular dystrophy. Cochrane Database Syst Rev 2019;10:CD011550. 10.1002/14651858.CD011550.pub2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Narayanaswami P, Weiss M, Selcen D, et al. Evidence-based guideline summary: diagnosis and treatment of limb-girdle and distal dystrophies: report of the guideline development subcommittee of the American academy of neurology and the practice issues review panel of the American association of neuromuscular & electrodiagnostic medicine. Neurology 2014;83:1453–63. 10.1212/WNL.0000000000000892 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ 2009;339:b2535. 10.1136/bmj.b2535 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Higgins JPT, Savović J, Page M, et al. Assessing risk of bias in a randomized trial. In: Higgins JPT, Thomas J, Chandler J, eds. Cochrane handbook for systematic reviews of interventions. 2nd ed. Wiley-Blackwell, 2019: 205–28. 10.1002/9781119536604 [DOI] [Google Scholar]
- 14. Lakens D. Calculating and reporting effect sizes to facilitate cumulative science: a practical primer for t-tests and ANOVAs. Front Psychol 2013;4:863. 10.3389/fpsyg.2013.00863 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. World health organization . International classification of functioning, disability and health (ICF). 2001. [Google Scholar]
- 16. Zupan A, Gregorič M, Valenčic V. Long-lasting effects of electrical stimulation upon muscles of patients suffering from progressive muscular dystrophy. Clin Rehabil 1995;9:102–9. 10.1177/026921559500900203 [DOI] [Google Scholar]
- 17. Hind D, Parkin J, Whitworth V, et al. Aquatic therapy for children with duchenne muscular dystrophy: a pilot feasibility randomised controlled trial and mixed-methods process evaluation. Health Technol Assess 2017;21:1–120. 10.3310/hta21270 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Carroll K, Yiu EM, Ryan MM, et al. The effects of calf massage in boys with duchenne muscular dystrophy: a prospective interventional study. Disabil Rehabil 2021;43:3803–9. 10.1080/09638288.2020.1753829 [DOI] [PubMed] [Google Scholar]
- 19. Colson SS, Benchortane M, Tanant V, et al. Neuromuscular electrical stimulation training: a safe and effective treatment for facioscapulohumeral muscular dystrophy patients. Arch Phys Med Rehabil 2010;91:697–702. 10.1016/j.apmr.2010.01.019 [DOI] [PubMed] [Google Scholar]
- 20. Kierkegaard M, Harms-Ringdahl K, Edström L, et al. Feasibility and effects of a physical exercise programme in adults with myotonic dystrophy type 1: a randomized controlled pilot study. J Rehabil Med 2011;43:695–702. 10.2340/16501977-0833 [DOI] [PubMed] [Google Scholar]
- 21. Missaoui B, Rakotovao E, Bendaya S, et al. Posture and gait abilities in patients with myotonic dystrophy (Steinert disease). Evaluation on the short-term of a rehabilitation program. Ann Phys Rehabil Med 2010;53:387–98. 10.1016/j.rehab.2010.06.004 [DOI] [PubMed] [Google Scholar]
- 22. Kilinç M, Yildirim SA, Tan E. The effects of electrical stimulation and exercise therapy in patients with limb girdle muscular dystrophy: a controlled clinical trial. Neurosciences (Riyadh) 2015;20:259–66. 10.17712/nsj.2015.3.201501097 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Okkersen K, Jimenez-Moreno C, Wenninger S, et al. Cognitive behavioural therapy with optional graded exercise therapy in patients with severe fatigue with myotonic dystrophy type 1: a multicentre, single-blind, randomised trial. Lancet Neurol 2018;17:671–80. 10.1016/S1474-4422(18)30203-5 [DOI] [PubMed] [Google Scholar]
- 24. Voet N, Bleijenberg G, Hendriks J, et al. Both aerobic exercise and cognitive-behavioral therapy reduce chronic fatigue in FSHD. Neurology 2014;83:1914–22. 10.1212/WNL.0000000000001008 [DOI] [PubMed] [Google Scholar]
- 25. Bostock EL, O’Dowd DN, Payton CJ, et al. The effects of resistance exercise training on strength and functional tasks in adults with limb-girdle, becker, and facioscapulohumeral dystrophies. Front Neurol 2019;10:1216. 10.3389/fneur.2019.01216 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Roussel M-P, Hébert LJ, Duchesne E. Strength-training effectively alleviates skeletal muscle impairments in myotonic dystrophy type 1. Neuromuscul Disord 2020;30:283–93. 10.1016/j.nmd.2020.02.015 [DOI] [PubMed] [Google Scholar]
- 27. Lessard I, Gaboury S, Gagnon C, et al. Effects and acceptability of an individualized home-based 10-week training program in adults with myotonic dystrophy type 1. J Neuromuscul Dis 2021;8:137–49. 10.3233/JND-200570 [DOI] [PubMed] [Google Scholar]
- 28. Maghbouli N, Shirzad N, Fateh HR, et al. Efficacy of a 6-week supervised strengthening exercise program with emg-biofeedback in patients with muscular dystrophy: a randomized controlled trial. Muscle Ligaments Tendons J 2021;11:728. 10.32098/mltj.04.2021.16 [DOI] [Google Scholar]
- 29. Jansen M, van Alfen N, Geurts ACH, et al. Assisted bicycle training delays functional deterioration in boys with duchenne muscular dystrophy: the randomized controlled trial “no use Is disuse” Neurorehabil Neural Repair 2013;27:816–27. 10.1177/1545968313496326 [DOI] [PubMed] [Google Scholar]
- 30. Heutinck L, Jansen M, van den Elzen Y, et al. Virtual reality computer gaming with dynamic arm support in boys with duchenne muscular dystrophy. J Neuromuscul Dis 2018;5:359–72. 10.3233/JND-180307 [DOI] [PubMed] [Google Scholar]
- 31. Brooke MH, Griggs RC, Mendell JR, et al. Clinical trial in duchenne dystrophy. I. The design of the protocol. Muscle Nerve 1981;4:186–97. 10.1002/mus.880040304 [DOI] [PubMed] [Google Scholar]
- 32. Sherief A, Abd ElAziz HG, Ali MS. Efficacy of two intervention approaches on functional walking capacity and balance in children with duchene muscular dystrophy. J Musculoskelet Neuronal Interact 2021;21:343–50. [PMC free article] [PubMed] [Google Scholar]
- 33. Bankolé L-C, Millet GY, Temesi J, et al. Safety and efficacy of a 6-month home-based exercise program in patients with facioscapulohumeral muscular dystrophy: a randomized controlled trial. Medicine (Baltimore) 2016;95:e4497. 10.1097/MD.0000000000004497 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Güneş Gencer GY, Yilmaz Ö. The effect of trunk training on trunk control, upper extremity, and pulmonary function in children with duchenne muscular dystrophy: a randomized clinical trial. Clin Rehabil 2022;36:369–78. 10.1177/02692155211043265 [DOI] [PubMed] [Google Scholar]
- 35. Kenis-Coskun O, Imamoglu S, Karamancioglu B, et al. Comparison of telerehabilitation versus home-based video exercise in patients with duchenne muscular dystrophy: a single-blind randomized study. Acta Neurol Belg 2022;122:1269–80. 10.1007/s13760-022-01975-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Alemdaroğlu I, Karaduman A, Yilmaz ÖT, et al. Different types of upper extremity exercise training in duchenne muscular dystrophy: effects on functional performance, strength, endurance, and ambulation. Muscle Nerve 2015;51:697–705. 10.1002/mus.24451 [DOI] [PubMed] [Google Scholar]
- 37. Lindeman E, Leffers P, Spaans F, et al. Strength training in patients with myotonic dystrophy and hereditary motor and sensory neuropathy: a randomized clinical trial. Arch Phys Med Rehabil 1995;76:612–20. 10.1016/s0003-9993(95)80629-6 [DOI] [PubMed] [Google Scholar]
- 38. Bulut N, Karaduman A, Alemdaroğlu-Gürbüz İ, et al. The effect of aerobic training on motor function and muscle architecture in children with duchenne muscular dystrophy: a randomized controlled study Clin Rehabil 2022;36:1062–71. 10.1177/02692155221095491 [DOI] [PubMed] [Google Scholar]
- 39. Aldehag A, Jonsson H, Lindblad J, et al. Effects of hand-training in persons with myotonic dystrophy type 1-a randomised controlled cross-over pilot study. Disabil Rehabil 2013;35:1798–807. 10.3109/09638288.2012.754952 [DOI] [PubMed] [Google Scholar]
- 40. O’Dowd DN, Bostock EL, Smith D, et al. The effects of 12 weeks’ resistance training on psychological parameters and quality of life in adults with facioscapulohumeral, becker, and limb–girdle dystrophies. Disabil Rehabil 2022;44:5950–6. 10.1080/09638288.2021.1955306 [DOI] [PubMed] [Google Scholar]
- 41. Ribot-Ciscar E, Milhe-De Bovis V, Aimonetti J-M, et al. Functional impact of vibratory proprioceptive assistance in patients with facioscapulohumeral muscular dystrophy. Muscle Nerve 2015;52:780–7. 10.1002/mus.24605 [DOI] [PubMed] [Google Scholar]
- 42. Glanzman AM, Flickinger JM, Dholakia KH, et al. Serial casting for the management of ankle contracture in duchenne muscular dystrophy. Pediatr Phys Ther 2011;23:275–9. 10.1097/PEP.0b013e318227c4e3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Aprile I, Bordieri C, Gilardi A, et al. Balance and walking involvement in facioscapulohumeral dystrophy: a pilot study on the effects of custom lower limb orthoses. Eur J Phys Rehabil Med 2013;49:169–78. [PubMed] [Google Scholar]
- 44. Taktak DM, Bowker P. Lightweight, modular knee-ankle-foot orthosis for duchenne muscular dystrophy: design, development, and evaluation. Arch Phys Med Rehabil 1995;76:1156–62. 10.1016/s0003-9993(95)80126-x [DOI] [PubMed] [Google Scholar]
- 45. Vry J, Schubert IJ, Semler O, et al. Whole-body vibration training in children with duchenne muscular dystrophy and spinal muscular atrophy. Eur J Paediatr Neurol 2014;18:140–9. 10.1016/j.ejpn.2013.09.005 [DOI] [PubMed] [Google Scholar]
- 46. Chisari C, Bertolucci F, Dalise S, et al. Chronic muscle stimulation improves muscle function and reverts the abnormal surface EMG pattern in myotonic dystrophy: a pilot study. J Neuroeng Rehabil 2013;10:94. 10.1186/1743-0003-10-94 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Pegoraro V, Cudia P, Baba A, et al. Myomirnas and myostatin as physical rehabilitation biomarkers for myotonic dystrophy. Neurol Sci 2020;41:2953–60. 10.1007/s10072-020-04409-2 [DOI] [PubMed] [Google Scholar]
- 48. Hammarén E, Lindberg C, Kjellby-Wendt G. Effects of a balance exercise programme in myotonic dystrophy type 1: a pilot study. Eur J Physiother 2015;17:123–31. 10.3109/21679169.2015.1049204 [DOI] [Google Scholar]
- 49. Tollbäck A, Eriksson S, Wredenberg A, et al. Effects of high resistance training in patients with myotonic dystrophy. Scand J Rehabil Med 1999;31:9–16. 10.1080/003655099444678 [DOI] [PubMed] [Google Scholar]
- 50. Berthelsen MP, Husu E, Christensen SB, et al. Anti-gravity training improves walking capacity and postural balance in patients with muscular dystrophy. Neuromuscul Disord 2014;24:492–8. 10.1016/j.nmd.2014.03.001 [DOI] [PubMed] [Google Scholar]
- 51. Jensen BR, Berthelsen MP, Husu E, et al. Body weight-supported training in becker and limb girdle 2I muscular dystrophy. Muscle Nerve 2016;54:239–43. 10.1002/mus.25039 [DOI] [PubMed] [Google Scholar]
- 52. Kontou E, Papadopoulos C, Papadimas G, et al. Effect of exercise training on functional capacity and body composition in myotonic dystrophy type 2 patients. Muscle Nerve 2021;63:477–83. 10.1002/mus.27156 [DOI] [PubMed] [Google Scholar]
- 53. Orngreen MC, Olsen DB, Vissing J. Aerobic training in patients with myotonic dystrophy type 1. Ann Neurol 2005;57:754–7. 10.1002/ana.20460 [DOI] [PubMed] [Google Scholar]
- 54. Sveen ML, Jeppesen TD, Hauerslev S, et al. Endurance training improves fitness and strength in patients with becker muscular dystrophy. Brain 2008;131(Pt 11):2824–31. 10.1093/brain/awn189 [DOI] [PubMed] [Google Scholar]
- 55. Mikhail AI, Nagy PL, Manta K, et al. Aerobic exercise elicits clinical adaptations in myotonic dystrophy type 1 patients independently of pathophysiological changes. J Clin Invest 2022;132:e156125. 10.1172/JCI156125 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Voet NBM. Exercise in neuromuscular disorders: a promising intervention. Acta Myol 2019;38:207–14. [PMC free article] [PubMed] [Google Scholar]
- 57. Michie S, van Stralen MM, West R. The behaviour change wheel: a new method for characterising and designing behaviour change interventions. Implement Sci 2011;6:42. 10.1186/1748-5908-6-42 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Van den Bergh PYK, van Doorn PA, Hadden RDM, et al. European academy of neurology/peripheral nerve society guideline on diagnosis and treatment of chronic inflammatory demyelinating polyradiculoneuropathy: report of a joint task force-second revision. J Peripher Nerv Syst 2021;26:242–68. 10.1111/jns.12455 [DOI] [PubMed] [Google Scholar]
- 59. Nierse CJ, Abma TA, Horemans AMC, et al. Research priorities of patients with neuromuscular disease. Disabil Rehabil 2013;35:405–12. 10.3109/09638288.2012.694964 [DOI] [PubMed] [Google Scholar]
- 60. Connor SG, Fairchild TJ, Learmonth YC, et al. Testosterone treatment combined with exercise to improve muscle strength, physical function and quality of life in men affected by inclusion body myositis: a randomised, double-blind, placebo-controlled, crossover trial. PLoS One 2023;18:e0283394. 10.1371/journal.pone.0283394 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. van der Kooi EL, Vogels OJM, van Asseldonk R, et al. Strength training and albuterol in facioscapulohumeral muscular dystrophy. Neurology 2004;63:702–8. 10.1212/01.wnl.0000134660.30793.1f [DOI] [PubMed] [Google Scholar]
- 62. Jacques MF, Stockley RC, Bostock EI, et al. Frequency of reported pain in adult males with muscular dystrophy. PLoS One 2019;14:e0212437. 10.1371/journal.pone.0212437 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Jensen MP, Hoffman AJ, Stoelb BL, et al. Chronic pain in persons with myotonic dystrophy and facioscapulohumeral dystrophy. Arch Phys Med Rehabil 2008;89:320–8. 10.1016/j.apmr.2007.08.153 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Lofra RM, Walker H, Turner C, et al. P200 The UK myotonic dystrophy patient registry - empowering clinical research and patient voice with an effective translational research tool. Neuromuscul Disord 2023;33:S144. 10.1016/j.nmd.2023.07.309 [DOI] [Google Scholar]
- 65. Harvey S, Rowan K, Harrison D, et al. Using clinical databases to evaluate healthcare interventions. Int J Technol Assess Health Care 2010;26:86–94. 10.1017/S0266462309990572 [DOI] [PubMed] [Google Scholar]
- 66. Schwartz MH, Ries AJ. Rectus femoris transfer in children with cerebral palsy: comparing a propensity score-matched observational study to a randomized controlled trial. Dev Med Child Neurol 2021;63:196–203. 10.1111/dmcn.14709 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
jnnp-2023-331988supp001.pdf (225.8KB, pdf)
jnnp-2023-331988supp002.pdf (363.1KB, pdf)
jnnp-2023-331988supp003.pdf (328.3KB, pdf)
jnnp-2023-331988supp004.pdf (401.3KB, pdf)
jnnp-2023-331988supp005.pdf (228.2KB, pdf)
jnnp-2023-331988supp006.pdf (433.9KB, pdf)
jnnp-2023-331988supp007.pdf (256.5KB, pdf)
jnnp-2023-331988supp008.pdf (298.3KB, pdf)
Data Availability Statement
All data relevant to the study are included in the article or uploaded as supplementary information.