Abstract
Objective:
We aimed to examine associations between neuropsychiatric symptoms (NPS) and white matter hyperintensities (WMH) status in older adults without dementia under the hypothesis that WMH increased the odds of having NPS.
Design:
Longitudinal analysis of data acquired from the National Alzheimer’s Coordinating Center Uniform Data Set.
Settings:
Data were derived from 46 National Institute on Aging - funded Alzheimer’s Disease Research Centers.
Participants:
NACC participants aged ≥50 years with available data on WMH severity with a diagnosis of mild cognitive impairment (MCI) or who were cognitively unimpaired (CU) were studied. Among 4617 CU participants, 376 had moderate and 54 extensive WMH. Among 3170 participants with MCI, 471 had moderate and 88 had extensive WMH.
Measurements:
Using Cardiovascular Health Study (CHS) scores, WMH were coded as no to mild (CHS score: 0–4), moderate (score: 5–6) or extensive (score: 7–8). NPS were quantified on the Neuropsychiatric Inventory Questionnaire. Binary logistic regression models estimated the odds of reporting each of 12 NPS by WMH status separately for individuals with MCI or who were CU.
Results:
Compared to CU individuals with no to mild WMH, the odds of having elation [9.87,(2.63–37.10)], disinhibition [4.42,(1.28–15.32)], agitation [3.51,(1.29–9.54)] or anxiety [2.74,(1.28–5.88)] were higher for the extensive WMH group, whereas the odds of having disinhibition were higher for the moderate WMH group [1.94,(1.05–3.61)]. In the MCI group, he odds of NPS did not vary by WMH status.
Conclusions:
Extensive WMH were associated with higher odds of NPS in CU older adults but not in those with MCI.
Keywords: elation, disinhibition, agitation, anxiety
Introduction
White matter hyperintensities (WMH) are a very common MRI (magnetic resonance imaging) finding in older individuals, appearing with increased signal on T2-weighted and fluid-attenuated inversion recovery sequences. While they are most often regarded as a feature of small vessel disease, multiple pathophysiological mechanisms have been incriminated such as blood-brain barrier leakage, neuroinflammation and neurodegeneration (Fernando et al., 2006; Shim et al., 2015). Demyelination, axonal loss and gliosis with variable pathological severity are also among the non-specific neuropathological substrates of WMH (Gouw et al., 2011). WMH prevalence increases with age and vascular risk burden (Ryu et al., 2014). WMH have been related to cognitive decline, incident stroke and dementia, as well as fatigue, physical (e.g., imbalance, gait abnormalities) and neuropsychiatric symptoms (NPS) (Clancy et al., 2021; Debette and Markus, 2010).
NPS (such as depression, anxiety, delusions, and apathy) are non-cognitive disturbances that are quite prevalent in individuals with MCI and almost universal in those with dementia (Lyketsos et al., 2011, 2002). Among cognitively unimpaired (CU) older adults, NPS have been linked to worse cognitive test performance (Liampas et al., 2022b), more precipitous cognitive decline (Krell-Roesch et al., 2021) and an elevated hazard of Alzheimer’s (AD) or non-AD dementia (Liew, 2020). In individuals with MCI, NPS have been linked to steeper cognitive trajectories (Roberto et al., 2021) and inflated risk of future dementia (Liew, 2019), while in those with dementia, NPS are a harbinger of more abrupt cognitive decline (Defrancesco et al., 2020), among other unfavourable outcomes (Bränsvik et al., 2021). Therefore, the presence of NPS in older adults should be regarded as a precursor of cognitive decline throughout the normal cognitive aging to dementia continuum.
Of note, in the continuum of healthy aging - dementia, Taragano and colleagues introduced the construct of mild behavioural impairment (MBI) – the neuropsychiatric equivalent of MCI – , as a transitional stage between normal aging and dementia which confers greater risk of incident dementia than MCI (Taragano et al., 2009). Although an affinity towards non-AD dementias is apparent (frontotemporal dementia -FTD- and Lewy body dementia -LBD-), many individuals with MBI may convert to AD as well, owing to its considerably larger prevalence (Taragano et al., 2018). The predominant hypothesis suggests that the association between NPS and cognitive decline probably reflects the relationship of NPS with undergoing neuropathological alterations (Peters and Lyketsos, 2015). Different NPS have been related to different neurodegenerative processes and by extension to heterogeneous cognitive trajectories and progression to different neurocognitive entities. For instance, psychosis has been linked to neuritic plaques, neurofibrillary tangles and Lewy body disease (an in turn, incident AD and Lewy body dementia -LBD), agitation and aggression have been associated with TDP-43 pathology (a common substrate of FTD), whereas results on the involvement of vascular lesions in MBI have been inconsistent (Devanand et al., 2022; Matsuoka et al., 2023).
To date, few have investigated the relationship between WMH and NPS in individuals without dementia (MCI or normal cognition). These studies had several limitations such as small samples, not accounting for the confounding of neurocognitive status with vascular risk, non-distinctions between CU and MCI individuals, use of composite NPS scores over individual NPS (and then focus on depression and anxiety). It is not surprising that they have reported contradictory results (Chan et al., 2022; Clancy et al., 2021; Miao et al., 2021; Misquitta et al., 2020; Staekenborg et al., 2008; Tumati et al., 2023; Yang et al., 2022). The aim in this was to estimate the associations between WMH and individual NPS in older adults without dementia. Specifically, we hypothesized that the odds of having NPS would be to the presence of WMH in both CU and MCI.
Methods and Materials
This cross-sectional analysis capitalised on data from the ongoing Uniform Data Set (UDS). UDS is a central repository of multidisciplinary, longitudinally collected data by National Institute on Aging / National Institute of Health - funded Alzheimer’s Disease Research Centers (ADRCs) across the United States (Beekly et al., 2007; Morris et al., 2006; Siokas et al., 2022; Weintraub et al., 2009). UDS was initiated in 2005 and since has been stewarded by the National Alzheimer’s Coordinating Center (NACC). Clinician-, self- and family-referred volunteers, or actively recruited individuals with a cognitive status ranging from normal cognition to dementia are enrolled according to each ADRC’s discrete protocol. Standardized evaluations take place on an approximately annual basis. Participants or surrogates provide informed consent before participation. All procedures are overseen by local Institutional Review Board(s) and performed in accordance with the ethical standards of the declaration of Helsinki and its later amendments. For further information on access to the NACC database, please contact NACC at https://naccdata.org/.
Eligibility Criteria and Diagnostic Procedures
The current analysis was based on UDS data from the December 2022 data freeze, collected from a total of 46 ADRCs. Older (≥50 years) participants with available data on WMH (only the 1st visit with available data was considered for eligibility) and a concurrent diagnosis of MCI or CU, were eligible (those with dementia or cognitive impairment not MCI were excluded). Cognitive diagnoses were established by either expert consensus panels (in the majority of cases) or single physicians (i.e., those who conducted the examination), according to each ADRC’s discrete protocol. CU was defined by the absence of a diagnosis of dementia, MCI or cognitive impairment not MCI. MCI [subjective and objective (based on the typical threshold of 1.5 standard deviations) cognitive disorder in the absence of repercussions on daily life] and dementia were diagnosed using standard clinical criteria (McKeith et al., 2017; McKhann et al., 1984; Neary et al., 1998; Petersen et al., 1999; Roman et al., 1993; Winblad et al., 2004). Participants with cognitive impairment who did not clearly fit into these categories were diagnosed with cognitive impairment – not MCI.
Measurement of NPS
The Neuropsychiatric Inventory Questionnaire (NPI-Q) is an informant administered, widely used tool for the evaluation of NPS in dementia research (Kaufer et al., 2000; Liampas et al., 2024). NPI-Q evaluates 12 domains: delusions, hallucinations, agitation/aggression, depression/dysphoria, anxiety, elation/euphoria, apathy/indifference, disinhibition, irritability/lability, aberrant motor behaviour, night-time behaviours, and eating behaviours. Informants initially report the presence or absence of cardinal symptomatology for each domain in the month preceding the examination and subsequently rate the severity of any symptoms according to a 3-point severity scale: mild (noticeable, but not a significant change); moderate (significant, but not a dramatic change); or severe (very marked or prominent; a dramatic change) (Liampas et al., 2022c). For the purposes of the current analysis, participants were dichotomised for presence of each NPS (0: absent; 1: present). Delusions and hallucinations were grouped together (psychotic symptoms) owing to their very low prevalence. Two additional composite NPS indices were analysed: total number of NPS (0–11) and total NPS severity score (0–22). For the latter, absence of NPS was scored with 0, mild symptomatology conferred 1 point and moderate to severe symptomatology conferred 2 points.
Measurement of WMH
The Cardiovascular Health Study (CHS) score is an ordinal quantification scale that uses visual inspection of a MRI to WMH burden ranging from 0 to 8 (Manolio et al., 1994). Periventricular and subcortical volumes of WMH are assessed on spin density-weighted axial images and scored between 0 (no WMH) and 8 (extensive, confluent WMH). Areas of large vessel infarction or small vessel lacunar strokes are excluded from the scoring. In the NACC database, based on WMH status, participants are coded as follows: with no to mild WMH (CHS score: 0–4), with moderate WMH (CHS score: 5–6) and with extensive WMH (CHS score: 7–8).
Covariates Considered
Chronological age upon entry to the study and years of formal education were treated as scale variables. Sex, race (Caucasian, African American, other) and the following comorbidities were treated as categorical (yes/no) variables: cerebrovascular disease (CEVD), atrial fibrillation (AF), hypertension, diabetes mellitus (DM) and dyslipidaemia. CEVD was defined as a positive history of stroke and/or transient ischemic attack. Comorbidities were primarily evaluated based on participant or co-participant reporting. However, to limit the amount of missing data, clinician reporting was utilized when necessary.
Regarding cognition, the five MCI subtypes in the NACC database were treated as dichotomous variables, i.e., MCI memory: yes/no; MCI language: yes/no; MCI executive function: yes/no; MCI visuospatial: yes/no; MCI attention: yes/no. On the other hand, Mini-Mental State Examination (MMSE) scores were treated as scale variables. Instead of MMSE, Montreal Cognitive Assessment (MoCA) was utilised in the last (3rd) version of UDS. To limit the amount of missing data, MoCA values were converted to MMSE scores according to conversion tables provided by a NACC crosswalk study (Liampas et al., 2023; Monsell et al., 2016).
Statistical Analysis
Individuals with unimpaired cognition or with MCI were analysed separately using the same approach. Demographic and other characteristics of the three groups defined by WMH status were compared using analysis of variance (ANOVA; scale variables) and Pearson’s chi-squared test (categorical variables). The frequencies of different NPS subtypes were also compared between the three WMH groups using Pearson’s chi-squared test.
The unadjusted and adjusted odds, and 95% confidence intervals, of having each NPS by WMH severity were estimated. Binary logistic regression models were performed to estimate the adjusted odds of reporting each NPS by WMH status. Models were adjusted for age, years of education, sex, race, history of CeVD, AF, hypertension, DM and dyslipidaemia. To account for cognitive status, analyses involving CU participants were additionally adjusted for MMSE scores, whereas analyses involving MCI participants were additionally adjusted for MCI subtypes. Finally, composite NPS measures (scale variables: total number of NPS or total NPS severity) were sequentially inserted into univariate generalized linear models (GLMs) as dependent variables (robust to violations of normality). Again, both measures were separately analysed in CU and MCI individuals. GLMs were adjusted for the same covariates as described before.
Statistical analyses were performed using the IBM SPSS Statistics Software Version 26 (Chicago, IL, USA). Despite performing multiple comparisons, the conventional threshold of α= .05 was implemented for the revelation of statistical significance in order to retain a fair statistical power for our analyses, considering the low frequency of certain NPS, and the low prevalence of extensive WMH.
Results
Participant Characteristics – CU Group
In total, 4617 CU participants with available WMH assessments were eligible for the analysis. Of these 376 had moderate and 54 had extensive WMH. Baseline differences by WMH status are in Table 1. The sample comprised predominantly older, well-educated, Caucasian individuals. CU individuals with greater WMH burden were older, more often of female sex and African American, and performed worse on MMSE. Vascular comorbidities were more common in the presence of more severe WMH. Participants with normal cognition and extensive WMH had greater average severity of NPS. Anxiety and elation were the only NPS that were more prevalent in the high severity WMH group.
Table 1.
Baseline comparison of older, cognitively unimpaired individuals by white matter hyperintensities (WMH) status.
| Variable | CHS score <5 (n= 4187) |
CHS score: 5–6 (n= 376) |
CHS score: 7–8 (n= 54) |
P-value |
|---|---|---|---|---|
| Age in years | 69.6 ±8.7 | 74.8 ±8.5 | 77.4 ±7.9 | <.001 |
| Formal education in years | 16.3 ±2.8 | 16.1 ±2.8 | 16.7 ±2.7 | .112 |
| Mini-mental state-examination score | 29.3 ±1.2 | 29.0 ±1.5 | 28.6 ±2.3 | <.001 |
| Sex (male/female %) | 1502/2685 (93/90%) | 102/274 (6/9%) | 19/35 (1/1%) | .003 |
| Race (Caucasian / African American / other %) | 3422/500/265 (92/85/92%) | 274/83/19 (7/14/7%) | 45/6/3 (1/1/1%) | <.001 |
| Cerebrovascular disease(No/Yes %) | 4090/97 (91/79%) | 357/19 (8/15%) | 47/7 (1/6%) | <.001 |
| Atrial fibrillation (No/Yes %) | 3975/198 (91/86%) | 344/29 (8/12%) | 47/4 (1/2%) | .024 |
| Diabetes mellitus (No/Yes %) | 3686/239 (91/87%) | 310/33 (8/12%) | 43/4 (1/1%) | .031 |
| Hypertension (No/Yes %) | 2473/1704 (94/86%) | 146/228 (5/12%) | 16/38 (1/2%) | <.001 |
| Dyslipidaemia (No/Yes %) | 2051/2083 (93/89%) | 141/228 (6/10%) | 24/29 (1/1%) | <.001 |
| Average number of NPS per individual | 0.74 ±1.36 | 0.79 ±1.44 | 1.35 ±1.86 | .017 |
| Average NPS severity per individual | 0.97 ±2.00 | 1.00 ±1.97 | 1.85 ±2.75 | .021 |
| Psychotic symptoms (No/Yes %) | 3814/31 (91/91%) | 343/3 (8/9%) | 41/0 (1/0%) | .839 |
| Depression (No/Yes %) | 3242/592 (91/91%) | 297/48 (8/7%) | 31/10 (1/2%) | .209 |
| Anxiety (No/Yes %) | 3306/527 (91/90%) | 295/49 (8/8%) | 29/11 (1/2%) | .043 |
| Agitation (No/Yes %) | 3643/199 (91/90%) | 329/17 (8/8%) | 36/5 (1/2%) | .128 |
| Disinhibition (No/Yes %) | 3738/100 (91/85%) | 331/14 (8/12%) | 38/3 (1/3%) | .059 |
| Irritability (No/Yes %) | 3359/482 (91/91%) | 303/42 (8/8%) | 33/8 (1/1%) | .397 |
| Elation (No/Yes %) | 3808/34 (91/90%) | 345/1 (8/3%) | 38/3 (1/8%) | <.001 |
| Apathy (No/Yes %) | 3631/201 (91/90%) | 326/20 (8/9%) | 38/3 (1/1%) | .774 |
| Motor symptoms (No/Yes %) | 3766/64 (91/92%) | 340/5 (8/7%) | 40/1 (1/1%) | .883 |
| Appetite disorders (No/Yes %) | 3633/194 (91/88%) | 320/23 (8/10%) | 36/5 (1/2%) | .059 |
| Night-time behaviours (No/Yes %) | 3265/466 (91/89%) | 289/46 (8/9%) | 32/9 (1/2%) | .162 |
Bold denotes statistically significant differences between the two groups; CHS: cardiovascular health study.
Associations between WMH Status and NPS – CU Group
After adjusting for age, education, MMSE scores, sex, race and vascular risk factors, the average number of NPS was 1.28 (95% CI: 1.08 – 1.48) for those with no to mild WMH, 1.38 (1.13 – 1.63) for those with moderate and 1.99 (1.50 – 2.49) for those with severe. Post hoc comparisons (pooled p= 0.007) found that the total number of NPS was greater in CU individuals with extensive WMH compared to those with no to mild WMH [Mean difference (MD)= 0.71 (0.24 – 1.18), p= 0.003] or compared to those with moderate WMH [MD= 0.61 (0.12 – 1.10), p= 0.014]. Average NPS severity by WMH status was 1.71 (1.41 – 2.00) for those with no to mild WMH, 1.83 (1.46 – 2.19) for those with moderate and 2.76 (2.03 – 3.49) for those with severe. Post hoc comparisons (pooled p= 0.008) revealed that total NPS severity was greater in the group of CU individuals with extensive WMH compared to those with no to mild WMH [MD= 1.05 (0.37 – 1. 74), p= 0.003], and compared to those with moderate WMH [MD= 0.93 (0.21 – 1.65), p= 0.011].
The odds of having each NPS by WMH status in CU individuals are in Table 2. The odds of elation (~9.9 times), disinhibition (~4.4 times), agitation (~3.5 times) and anxiety (~2.7 times) were significantly elevated in CU older adults with extensive WMH compared to those with no or mild WMH(Figure 1). On the other hand, compared to those with no or mild WMH,moderate WMH increased only the odds of having disinhibition by about 1.9 times. Of note, the older adults with extensive WMH had higher odds (between 1.7 and 2.1 times) of having every remaining NPS, apart from psychotic symptoms, but findings were not significant.
Table 2.
Odds of having neuropsychiatric symptoms by white matter hyperintensities (WMH) status among older, cognitively unimpaired adults. The group with the lowest cardiovascular health study (CHS) score (<5) was used as reference.
| Variable | Pooled P-value | CHS score: 5–6 OR (95%CI) |
P-value | CHS score: 7–8 OR (95%CI) |
P-value |
|---|---|---|---|---|---|
| Adjusted analyses | |||||
| Psychotic symptoms | p= 1.000 | 0.95 (0.27 – 3.34) | .936 | NA | .998 |
| Depression | p= .144 | 0.93 (0.65 – 1.33) | .694 | 2.14 (0.98 – 4.70) | .057 |
| Anxiety | p= .034 | 1.07 (0.75 – 1.53) | .707 | 2.74 (1.28 – 5.88) | .010 |
| Agitation | p= .049 | 1.06 (0.60 – 1.86) | .843 | 3.51 (1.29 – 9.54) | .014 |
| Disinhibition | p= .011 | 1.94 (1.05 – 3.61) | .035 | 4.42 (1.28 – 15.32) | .019 |
| Irritability | p= .540 | 1.01 (0.69 – 1.49) | .963 | 1.68 (0.67 – 4.18) | .267 |
| Elation | p= .002 | 0.34 (0.05 – 2.93) | .360 | 9.87 (2.63 – 37.10) | <.001 |
| Apathy | p= .608 | 0.96 (0.54 – 1.68) | .873 | 1.85 (0.54 – 6.38) | .330 |
| Motor symptoms | p= .814 | 0.89 (0.31 – 2.56) | .830 | 1.86 (0.24 – 14.53) | .554 |
| Appetite disorders | p= .164 | 1.32 (0.81 – 2.16) | .266 | 2.44 (0.83 – 7.15) | .105 |
| Night-time behaviours | p= .162 | 1.27 (0.88 – 1.81) | .198 | 1.91 (0.81 – 4.49) | .137 |
| Unadjusted analyses | |||||
| Psychotic symptoms | p= .839 | 1.08 (0.33 – 3.54) | p= .904 | NA | .564 |
| Depression | p= .209 | 0.89 (0.64 – 1.22) | p= .450 | 1.77 (0.86 – 3.62) | .116 |
| Anxiety | p= .043 | 1.04 (0.76 – 1.43) | p= .799 | 2.38 (1.18 – 4.79) | .012 |
| Agitation | p= .128 | 0.95 (0.57 – 1.57) | p= .830 | 2.54 (0.99 – 6.55) | .061 |
| Disinhibition | p= .059 | 1.58 (0.89 – 2.80) | p= .112 | 2.95 (0.90 – 9.72) | .062 |
| Irritability | p= .397 | 0.97 (0.69 – 1.35) | p= .840 | 1.69 (0.78 – 3.68) | .182 |
| Elation | p< .001 | 0.33 (0.04 – 2.38) | p= .243 | 8.84 (2.60 – 30.04) | < .001 |
| Apathy | p= .774 | 1.11 (0.69 – 1.78) | p= .670 | 1.43 (0.44 – 4.66) | .555 |
| Motor symptoms | p= .883 | 0.87 (0.35 – 2.17) | p= .757 | 1.47 (0.20 – 10.87) | .703 |
| Appetite disorders | p= .059 | 1.35 (0.86 – 2.11) | p= .191 | 2.60 (1.01 – 6.70) | .040 |
| Night-time behaviours | p= .162 | 1.12 (0.81 – 1.55) | p= .512 | 1.97 (0.94 – 4.15) | .069 |
Bold denotes statistically significant differences between the two groups; between group differences were considered significant only if among group differences were significant as well – p-value for among group differences in provided by each NPS; analyses were adjusted for age, education, MMSE scores, sex, race and vascular risk factors; OR: odds ratio; CI: confidence interval; NA: non-applicable – no participant with extensive WMH had psychotic symptoms.
Figure 1.
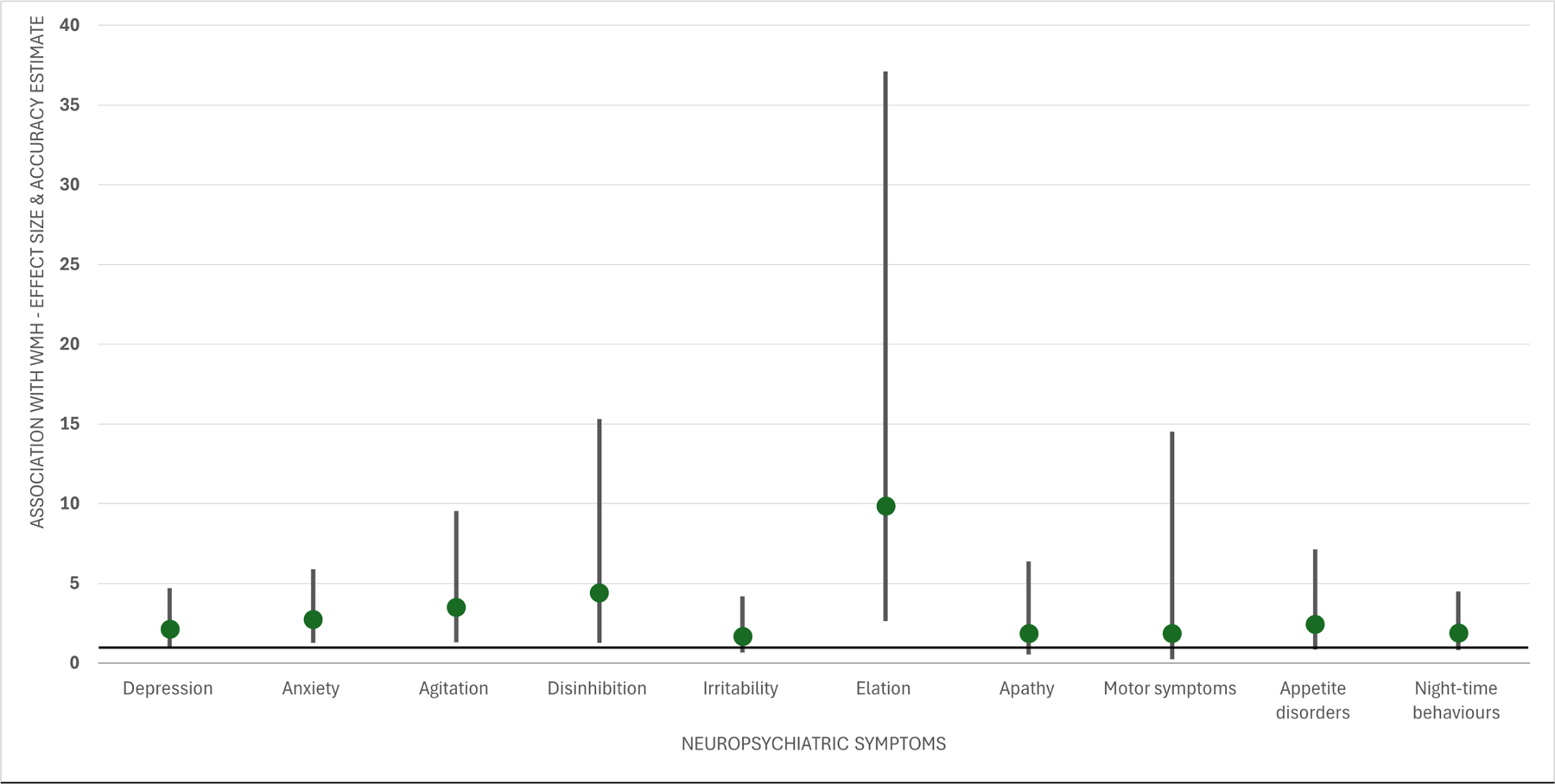
Associations between extensive white matter hyperintensities (WMH) and neuropsychiatric symptoms in cognitively unimpaired older adults. Odds ratios (green dots) and 95% confidence intervals (95%Cis – upper and lower limits) are provided.
Participant Characteristics – MCI Group
In total, 3170 participants with MCI had available WMH assessments and were eligible for the analysis. Among them, 471 had moderate and 88 had extensive WMH. Baseline differences by WMH status are in Table 3. The MCI group primarily consisted of older, well-educated, Caucasian individuals. Older adults with MCI and greater WMH burden were older, more often African American and performed worse on MMSE. Vascular comorbidities were more prevalent in the presence of more severe WMH. The number of total NPS, total NPS severity, as well as the presence of individual NPS did not differ by extent of WMH changes.
Table 3.
Baseline comparison of older individuals with mild cognitive impairment (MCI) by white matter hyperintensities (WMH) status.
| Variable | CHS score <5 (n= 2611) |
CHS score: 5–6 (n= 471) |
CHS score: 7–8 (n= 88) |
P-value |
|---|---|---|---|---|
| Age in years | 71.8 ±8.8 | 76.9 ±7.2 | 78.8 ±7.3 | <.001 |
| Formal education in years | 16.1 ±3.1 | 16.0 ±3.0 | 15.5 ±3.4 | .283 |
| Mini-mental state-examination score | 27.4 ±2.6 | 27.1 ±2.5 | 26.8 ±2.5 | .012 |
| Sex (male/female %) | 1355/1256 (83/81%) | 226/245 (14/16%) | 48/40 (3/3%) | .246 |
| Race (Caucasian / African American / other %) | 2240/240/131 (83/73/82%) | 374/74/23 (14/22/15%) | 68/15/5 (3/5/3%) | <.001 |
| Cerebrovascular disease(No/Yes %) | 2486/125 (84/62%) | 415/56 (14/27%) | 66/22 (2/11%) | <.001 |
| Atrial fibrillation (No/Yes %) | 2423/177 (83/73%) | 416/54 (14/22%) | 75/11 (3/5%) | <.001 |
| Diabetes mellitus (No/Yes %) | 2250/234 (83/76%) | 380/62 (14/20%) | 67/11 (3/4%) | .007 |
| Hypertension (No/Yes %) | 1362/1238 (88/77%) | 161/310 (10/19%) | 23/65 (2/4%) | <.001 |
| Dyslipidaemia (No/Yes %) | 1156/1431 (86/80%) | 155/309 (12/17%) | 26/62 (2/3%) | <.001 |
| Average number of NPS per individual | 2.10 ±2.14 | 2.07 ±2.09 | 1.61 ±1.99 | .136 |
| Average NPS severity per individual | 2.84 ±3.31 | 2.80 ±3.21 | 2.15 ±3.18 | .203 |
| Psychotic symptoms (No/Yes %) | 2337/129 (82/83%) | 421/23 (15/15%) | 78/3 (3/2%) | .830 |
| Depression (No/Yes %) | 1602/856 (82/84%) | 300/141 (15/14%) | 60/21 (3/2%) | .145 |
| Anxiety (No/Yes %) | 1654/806 (82/83%) | 294/149 (15/15%) | 62/19 (3/2%) | .190 |
| Agitation (No/Yes %) | 2041/423 (82/84%) | 369/74 (15/14%) | 73/8 (3/2%) | .225 |
| Disinhibition (No/Yes %) | 2172/291 (83/81%) | 382/61 (14/17%) | 72/9 (3/2%) | .490 |
| Irritability (No/Yes %) | 1668/797 (83/82%) | 293/150 (14/15%) | 57/24 (3/3%) | .701 |
| Elation (No/Yes %) | 2400/66 (82/84%) | 430/13 (15/16%) | 81/0 (3/0%) | .308 |
| Apathy (No/Yes %) | 1912/554 (82/84%) | 352/91 (15/14%) | 67/14 (3/2%) | .386 |
| Motor symptoms (No/Yes %) | 2273/194 (82/83%) | 408/34 (15/15%) | 76/5 (3/2%) | .852 |
| Appetite disorders (No/Yes %) | 2045/414 (82/84%) | 374/69 (15/14%) | 70/11 (3/2%) | .617 |
| Night-time behaviours (No/Yes %) | 1762/658 (82/84%) | 323/112 (15/14%) | 62/14 (3/2%) | .207 |
Bold denotes statistically significant differences between the two groups; CHS: cardiovascular health study.
Associations between WMH Status and NPS – MCI Group
After adjusting for age, education, MCI subtypes, sex, race and vascular risk factors, the average total number of NPS was 2.32 (2.03 – 2.61) for those with no to mild, 2.50 (2.17– 2.83) for those with moderate and 1.89 (1.34 – 2.45) for those with severe WMH. Post hoc comparisons showed that the total number of NPS did not differ between groups. Average NPS severity was 3.14 (2.69 – 3.59) for those with no to mild, 3.41 (2.90 – 3.92) for those with moderate and 2.56 (1.71 – 3.41) for those with severe WMH. Post hoc comparisons found no NPS differences by WMH group. The odds of having each NPS by WMH status in older adults with MCI are in Table 4. Those with moderate WMH had approximately 34% more odds of having anxiety. However, considering the small effect size and inconsistency (no other significant associations were found between WMH and NPS in the MCI group – an opposite trend was revealed) this finding is likely the result of chance.
Table 4.
Odds of having neuropsychiatric symptoms by white matter hyperintensities (WMH) status among older adults with mild cognitive impairment (MCI). The group with the lowest cardiovascular health study (CHS) score (<5) was used as reference.
| Variable | Pooled P-value | CHS score: 5–6 OR (95%CI) |
P-value | CHS score: 7–8 OR (95%CI) |
P-value |
|---|---|---|---|---|---|
| Adjusted analyses | |||||
| Psychotic symptoms | p= .552 | 1.15 (0.70 – 1.89) | .573 | 0.52 (0.12 – 2.21) | .379 |
| Depression | p= .643 | 1.05 (0.83 – 1.34) | .668 | 0.80 (0.46 – 1.39) | .430 |
| Anxiety | p= .032 | 1.34 (1.05 – 1.69) | .017 | 0.78 (0.44 – 1.40) | .405 |
| Agitation | p= .147 | 1.03 (0.76 – 1.38) | .870 | 0.43 (0.18 – 1.01) | .054 |
| Disinhibition | p= .068 | 1.47 (1.05 – 2.04) | .024 | 1.35 (0.65 – 2.81) | .425 |
| Irritability | p= .164 | 1.25 (0.98 – 1.58) | .068 | 0.91 (0.53 – 1.56) | .728 |
| Elation | p= .486 | 1.49 (0.78 – 2.84) | .230 | NA | .997 |
| Apathy | p= .721 | 1.00 (0.76 – 1.32) | .990 | 0.77 (0.40 – 1.47) | .420 |
| Motor symptoms | p= .574 | 1.12 (0.73 – 1.72) | .602 | 0.59 (0.18 – 1.94) | .386 |
| Appetite disorders | p= .794 | 0.98 (0.72 – 1.34) | .894 | 0.78 (0.38 – 1.61) | .499 |
| Night-time behaviours | p= .140 | 1.01 (0.78 – 1.30) | .951 | 0.51 (0.26 – 1.00) | .049 |
| Unadjusted analyses | |||||
| Psychotic symptoms | p= .830 | 0.99 (0.63 – 1.56) | .965 | 0.70 (0.22 – 2.24) | .542 |
| Depression | p= .145 | 0.88 (0.71 – 1.09) | .246 | 0.66 (0.40 – 1.08) | .097 |
| Anxiety | p= .190 | 1.04 (0.84 – 1.29) | .720 | 0.63 (0.37 – 1.06) | .078 |
| Agitation | p= .225 | 0.97 (0.74 – 1.27) | .812 | 0.53 (0.25 – 1.11) | .085 |
| Disinhibition | p= .490 | 1.19 (0.89 – 1.60) | .246 | 0.93 (0.46 – 1.87) | .847 |
| Irritability | p= .701 | 1.07 (0.87 – 1.33) | .528 | 0.88 (0.54 – 1.43) | .609 |
| Elation | p= .308 | 1.10 (0.60 – 2.01) | .758 | NA | .136 |
| Apathy | p= .386 | 0.89 (0.70 – 1.15) | .369 | 0.72 (0.40 – 1.29) | .270 |
| Motor symptoms | p= .852 | 0.98 (0.67 – 1.43) | .902 | 0.77 (0.31 – 1.93) | .577 |
| Appetite disorders | p= .617 | 0.91 (0.69 – 1.20) | .512 | 0.78 (0.41 – 1.48) | .440 |
| Night-time behaviours | p= .207 | 0.93 (0.74 – 1.17) | .532 | 0.61 (0.34 – 1.09) | .090 |
Bold denotes statistically significant differences between the two groups; between group differences were considered significant only if among group differences were significant as well – p-value for among group differences in provided by each NPS; analyses were adjusted for age, education, MCI subtypes, sex, race and vascular risk factors; OR: odds ratio; CI: confidence interval; NA: non-applicable – no participant with extensive WMH had elation.
Discussion
We report that CU older adults with extensive WMH in MRI studies had higher odds of having NPS compared to individuals with no to mild WMH, in particular, elation, disinhibition, agitation, and anxiety. In the CU group with moderate WMH only disinhibition was associated with WMH in comparison with the reference group (no to mild WMH). Our estimates accounted for important demographic and vascular-related factors, as well as cognitive testing. These findings align with the hypothesis that severe WMH contribute to the overall neuropsychiatric burden of CU individuals, independent of cognitive impairment or vascular risk burden. Moderate WMH do not appear to appreciably increase NPS risk overall in this group.
Older individuals with MCI and extensive or moderate WMH did not have greater odds of NPS compared to those with no to mild WMH. This lack of association may stem from the fact that co-existing neuropathologic alterations in older adults with MCI are almost universal: AD pathology predominates with vascular and Lewy body (LB) pathologies not infrequent (Dugger et al., 2015; Schneider et al., 2009). Therefore, those with no to mild (as well as moderate) WMH may have more co-existing neurodegenerative alterations that account for cognitive impairment. Considering the well-established, strong relationships between amyloid or LB pathology and several NPS it is likely that any association of WMH with NPS was overwhelmed by that of the other pathologies (Gibson et al., 2023; Goukasian et al., 2019; Krell-Roesch et al., 2019). These findings may provide a potential explanation for the incongruous results of previous publications that did not consistently reveal a relationship between WMH and NPS in individuals without dementia: analysing individuals without dementia, especially small groups, with and without MCI, with MCI owing to heterogeneous neuropathologies or with different levels of MCI and NPS severity may modulate true associations between WMH and NPS.
WMH have been associated with disrupted brain network dynamics (Tuladhar et al., 2015). Impaired transferring of information between interconnected cerebral areas is theorized to underlie the relationship between WMH and cognitive impairment (Yang et al., 2020). The same mechanism could be crucial in the occurrence of neuropsychiatric symptoms in individuals with WMH (Desmarais et al., 2021). The functional connectivity of the brain is compromised across multiple psychiatric conditions with younger ages of onset (e.g., autism, attention deficit hyperactivity disorder, bipolar disorder, schizophrenia, and so on) and may be similarly undermined in older adults with late-onset NPS. Therefore, the correlation of greater WMH load with reduced functional connectivity may provide a potential explanation for the prominent associations between extensive WMH and NPS (Crockett et al., 2021; Quandt et al., 2020). Future research conducting mediation analyses could explore whether disrupted brain connectivity assumes a pivotal role in NPS among older individuals, whether different mechanisms are implicated or whether WMH are just epiphenomena of neurodegeneration with variable severity across the spectrum of heterogeneous neuropathologies.
Of note, WMH may not only contribute directly to the epidemiology of NPS but also via their interference with cognition. WMH burden has been correlated with global but also domain-specific cognitive impairment (Prins and Scheltens, 2015). More specifically, frontal operations such as executive function and attention appear to be correlated to the volume of periventricular and subcortical WMH (Puzo et al., 2019; Sudo et al., 2012). Again, disrupted brain (mainly fronto-parietal) networks seem to mediate these cognitive associations (Li et al., 2015). Of interest, specific cognitive deficits have been more strongly linked to particular NPS: impairments in principally frontally mediated functions have been specifically associated with anxiety and lability symptoms such as disinhibition, agitation, irritability and elation (Liampas et al., 2022b; Rosenberg et al., 2011). Of course, some spatial specificity is to be expected; WMH in strategic brain regions may be more or less related to different cognitive and NPS manifestations (Brugulat-Serrat et al., 2020; Lampe et al., 2019). Based on the above, our findings may at least partially be driven by the cognitive associations of WMH.
Overall, our findings enhance current knowledge on the associations between neurodegenerative alterations and NPS in older adults without dementia. Previous positron emission tomography (PET) imaging studies of individuals without dementia has indicated that higher Aβ42 deposition is related to higher NPI-Q scores (Ng et al., 2021). Apathy, anxiety, depression and psychotic symptoms were most consistently associated with amyloid pathology – while agitation, disinhibition or elation present weaker to marginal associations (Gibson et al., 2023; Goukasian et al., 2019; Krell-Roesch et al., 2019; Ng et al., 2021). On the other hand, elation and disinhibition appear to be linked to the presence of frontotemporal lobar degeneration, whereas psychotic symptoms and impulse control disorders are very prevalent with LB and PD neuropathology, respectively (Cajanus et al., 2019; Cotta Ramusino et al., 2021; Sokołowski et al., 2023). Although significant overlap is to be expected, based on the neuropsychiatric manifestations of an individual -especially as part of a comprehensive examination- clinicians and researcher can make some inferences about potential ongoing neurodegenerative alterations and select more sophisticated laboratory means to establish more accurate diagnoses.
Strengths and Limitations
Our study has several strengths including the large sample of individuals with available WMH assessments with an adequate number of individuals with extensive WMH. The NPI-Q was uniformly used to assess the presence of NPS. The neurocognitive status of the participants (along with important demographic and vascular-related confounders) was accounted for in the analytical part of the article.
This analysis has several weaknesses, as well. First, the number of certain NPS (especially psychotic and motor symptoms and secondarily apathy) was very small, underpowering several aspects of our analysis. Therefore, it is not surprising that we failed to reproduce some previously established associations, such as between WMH and apathy (Manca et al., 2022). This is reflected in the broad confidence intervals and may have obscured several non-trivial associations. Second, although several crucial factors and covariates were taken into account, our findings may have been driven by residual confounding (it is not be possible to capture the effect of every potential confounder) or the non-trivial proportion of missing data (Liampas et al., 2022a; Samara et al., 2022). Third, the presence or absence of WMH was not uniformly assessed by a central, blinded evaluator (or group of evaluators). Some variability is expected among different assessors in the quantification of WMH. In addition, we did not correct our findings for multiple comparisons to retain a fair statistical power despite the low frequency of certain NPS and the low prevalence of extensive WMH. Nevertheless, in view of the sizeable and consistent associations in the CU group we are confident that our results reflect true associations. Moreover, we did not include additional imaging (or not) biomarkers, such as global or parietal atrophy, hippocampal volumes, and so on. Finally, another limitation is the observational nature of our study. Hence, it is not possible to make etiologic inferences about NPS and WMH.
Conclusions
Extensive WMH were associated with the overall neuropsychiatric burden of CU individuals, independent of cognitive impairment or vascular risk burden. The odds of elation, disinhibition, agitation and anxiety were particularly elevated. On the other hand, WMH were not related to the neuropsychiatric burden of individuals with MCI. Considering that alternative neuropathologic alterations in older adults with MCI (especially AD, vascular and LB pathology) may account for cognitive impairment (instead of extensive WMH), the very strong established relationships between these pathologies and NPS is likely overwhelming the association of WMH with NPS. Therefore, it would be interesting if future research looked into the same associations using larger samples of individuals with different MCI subtypes and by extension different underlying pathologies.
Acknowledgements
The NACC database is funded by NIA/NIH Grant U24 AG072122. NACC data are contributed by the NIA-funded ADRCs: P30 AG062429 (PI James Brewer, MD, PhD), P30 AG066468 (PI Oscar Lopez, MD), P30 AG062421 (PI Bradley Hyman, MD, PhD), P30 AG066509 (PI Thomas Grabowski, MD), P30 AG066514 (PI Mary Sano, PhD), P30 AG066530 (PI Helena Chui, MD), P30 AG066507 (PI Marilyn Albert, PhD), P30 AG066444 (PI John Morris, MD), P30 AG066518 (PI Jeffrey Kaye, MD), P30 AG066512 (PI Thomas Wisniewski, MD), P30 AG066462 (PI Scott Small, MD), P30 AG072979 (PI David Wolk, MD), P30 AG072972 (PI Charles DeCarli, MD), P30 AG072976 (PI Andrew Saykin, PsyD), P30 AG072975 (PI David Bennett, MD), P30 AG072978 (PI Neil Kowall, MD), P30 AG072977 (PI Robert Vassar, PhD), P30 AG066519 (PI Frank LaFerla, PhD), P30 AG062677 (PI Ronald Petersen, MD, PhD), P30 AG079280 (PI Eric Reiman, MD), P30 AG062422 (PI Gil Rabinovici, MD), P30 AG066511 (PI Allan Levey, MD, PhD), P30 AG072946 (PI Linda Van Eldik, PhD), P30 AG062715 (PI Sanjay Asthana, MD, FRCP), P30 AG072973 (PI Russell Swerdlow, MD), P30 AG066506 (PI Todd Golde, MD, PhD), P30 AG066508 (PI Stephen Strittmatter, MD, PhD), P30 AG066515 (PI Victor Henderson, MD, MS), P30 AG072947 (PI Suzanne Craft, PhD), P30 AG072931 (PI Henry Paulson, MD, PhD), P30 AG066546 (PI Sudha Seshadri, MD), P20 AG068024 (PI Erik Roberson, MD, PhD), P20 AG068053 (PI Justin Miller, PhD), P20 AG068077 (PI Gary Rosenberg, MD), P20 AG068082 (PI Angela Jefferson, PhD), P30 AG072958 (PI Heather Whitson, MD), P30 AG072959 (PI James Leverenz, MD).
Funding
This research received no specific grant from any funding agency, commercial or not-for-profit sectors.
Footnotes
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Standards
Participants or surrogates provide informed consent before participation. All procedures are overseen by Institutional Review Boards at each ADRC. The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008.
Availability of Data and Materials
For further information on access to the NACC database, please contact NACC (contact details can be found at https://naccdata.org/).
References
- Beekly DL, et al. 2007. The National Alzheimer’s Coordinating Center (NACC) Database: The Uniform Data Set. Alzheimer Disease & Associated Disorders 21, 249–258. 10.1097/WAD.0b013e318142774e [DOI] [PubMed] [Google Scholar]
- Bränsvik V, Granvik E, Minthon L, Nordström P, Nägga K, 2021. Mortality in patients with behavioural and psychological symptoms of dementia: a registry-based study. Aging Ment Health 25, 1101–1109. 10.1080/13607863.2020.1727848 [DOI] [PubMed] [Google Scholar]
- Brugulat-Serrat A, et al. 2020. Patterns of white matter hyperintensities associated with cognition in middle-aged cognitively healthy individuals. Brain Imaging Behav 14, 2012–2023. 10.1007/s11682-019-00151-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cajanus A, et al. 2019. The Association Between Distinct Frontal Brain Volumes and Behavioral Symptoms in Mild Cognitive Impairment, Alzheimer’s Disease, and Frontotemporal Dementia. Frontiers in Neurology 10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chan CK, et al. 2022. Association Between Late-Life Neuropsychiatric Symptoms and Cognitive Decline in Relation to White Matter Hyperintensities and Amyloid Burden. J Alzheimers Dis 86, 1415–1426. 10.3233/JAD-215267 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clancy U, Gilmartin D, Jochems ACC, Knox L, Doubal FN, Wardlaw JM, 2021. Neuropsychiatric symptoms associated with cerebral small vessel disease: a systematic review and meta-analysis. Lancet Psychiatry 8, 225–236. 10.1016/S2215-0366(20)30431-4 [DOI] [PubMed] [Google Scholar]
- Cotta Ramusino M, et al. 2021. Correlation of Frontal Atrophy and CSF Tau Levels With Neuropsychiatric Symptoms in Patients With Cognitive Impairment: A Memory Clinic Experience. Front Aging Neurosci 13, 595758. 10.3389/fnagi.2021.595758 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crockett RA, et al. 2021. Painting by lesions: White matter hyperintensities disrupt functional networks and global cognition. NeuroImage 236, 118089. 10.1016/j.neuroimage.2021.118089 [DOI] [PubMed] [Google Scholar]
- Debette S, Markus HS, 2010. The clinical importance of white matter hyperintensities on brain magnetic resonance imaging: systematic review and meta-analysis. BMJ 341, c3666. 10.1136/bmj.c3666 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Defrancesco M, et al. 2020. Specific Neuropsychiatric Symptoms Are Associated with Faster Progression in Alzheimer’s Disease: Results of the Prospective Dementia Registry (PRODEM-Austria). J Alzheimers Dis 73, 125–133. 10.3233/JAD-190662 [DOI] [PubMed] [Google Scholar]
- Desmarais P, et al. 2021. White matter hyperintensities in autopsy-confirmed frontotemporal lobar degeneration and Alzheimer’s disease. Alzheimer’s Research & Therapy 13, 129. 10.1186/s13195-021-00869-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Devanand DP, Lee S, Huey ED, Goldberg TE, 2022. Associations Between Neuropsychiatric Symptoms and Neuropathological Diagnoses of Alzheimer Disease and Related Dementias. JAMA Psychiatry 79, 359–367. 10.1001/jamapsychiatry.2021.4363 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dugger BN, et al. 2015. Neuropathological comparisons of amnestic and nonamnestic mild cognitive impairment. BMC Neurol 15, 146. 10.1186/s12883-015-0403-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fernando MS, et al. for the MRC Cognitive Function and Ageing Neuropathology Study Group, 2006. White matter lesions in an unselected cohort of the elderly: molecular pathology suggests origin from chronic hypoperfusion injury. Stroke 37, 1391–1398. 10.1161/01.STR.0000221308.94473.14 [DOI] [PubMed] [Google Scholar]
- Gibson LL, et al. 2023. Neuropathological correlates of neuropsychiatric symptoms in dementia. Alzheimer’s & Dementia 19, 1372–1382. 10.1002/alz.12765 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goukasian N, et al. 2019. Association of brain amyloidosis with the incidence and frequency of neuropsychiatric symptoms in ADNI: a multisite observational cohort study. BMJ Open 9, e031947. 10.1136/bmjopen-2019-031947 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gouw AA, et al. 2011. Heterogeneity of small vessel disease: a systematic review of MRI and histopathology correlations. J Neurol Neurosurg Psychiatry 82, 126–135. 10.1136/jnnp.2009.204685 [DOI] [PubMed] [Google Scholar]
- Kaufer DI, et al. 2000. Validation of the NPI-Q, a brief clinical form of the Neuropsychiatric Inventory. J Neuropsychiatry Clin Neurosci 12, 233–239. 10.1176/jnp.12.2.233 [DOI] [PubMed] [Google Scholar]
- Krell-Roesch J, et al. 2021. Neuropsychiatric symptoms and the outcome of cognitive trajectories in older adults free of dementia: The Mayo Clinic Study of Aging. Int J Geriatr Psychiatry 36, 1362–1369. 10.1002/gps.5528 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krell-Roesch J, et al. 2019. Cortical β-amyloid burden, neuropsychiatric symptoms, and cognitive status: the Mayo Clinic Study of Aging. Transl Psychiatry 9, 1–8. 10.1038/s41398-019-0456-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lampe L, et al. 2019. Lesion location matters: The relationships between white matter hyperintensities on cognition in the healthy elderly. J Cereb Blood Flow Metab 39, 36–43. 10.1177/0271678X17740501 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li X, et al. 2015. Disrupted Frontoparietal Network Mediates White Matter Structure Dysfunction Associated with Cognitive Decline in Hypertension Patients. J Neurosci 35, 10015–10024. 10.1523/JNEUROSCI.5113-14.2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liampas I, et al. 2022a. Antihypertensive Medication Class and the Risk of Dementia and Cognitive Decline in Older Adults: A Secondary Analysis of the Prospective HELIAD Cohort. J Alzheimers Dis 89, 709–719. 10.3233/JAD-220439 [DOI] [PubMed] [Google Scholar]
- Liampas I, Siokas V, Lyketsos CG, Dardiotis E, 2023. Cognitive Performance and Incident Alzheimer’s Dementia in Men Versus Women. J Prev Alzheimers Dis. 10.14283/jpad.2023.90 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liampas I, Siokas V, Lyketsos CG, Dardiotis E, 2022b. The Relationship between Neuropsychiatric Symptoms and Cognitive Performance in Older Adults with Normal Cognition. Medicina (Kaunas) 58, 1586. 10.3390/medicina58111586 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liampas I, Siokas V, Lyketsos CG, Dardiotis E, 2022c. Associations between neuropsychiatric symptoms and incident Alzheimer’s dementia in men versus women. J Neurol. 10.1007/s00415-022-11541-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liampas I, Siokas V, Zoupa E, Lyketsos CG, Dardiotis E, 2024. Neuropsychiatric symptoms and incident Lewy body dementia in male versus female older adults with mild cognitive impairment. Psychiatry Clin Neurosci 78, 144–146. 10.1111/pcn.13621 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liew TM, 2020. Neuropsychiatric symptoms in cognitively normal older persons, and the association with Alzheimer’s and non-Alzheimer’s dementia. Alzheimers Res Ther 12, 35. 10.1186/s13195-020-00604-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liew TM, 2019. Symptom Clusters of Neuropsychiatric Symptoms in Mild Cognitive Impairment and Their Comparative Risks of Dementia: A Cohort Study of 8530 Older Persons. J Am Med Dir Assoc 20, 1054.e1–1054.e9. 10.1016/j.jamda.2019.02.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lyketsos CG, et al. 2011. Neuropsychiatric symptoms in Alzheimer’s disease. Alzheimers Dement 7, 532–539. 10.1016/j.jalz.2011.05.2410 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lyketsos CG, Lopez O, Jones B, Fitzpatrick AL, Breitner J, DeKosky S, 2002. Prevalence of neuropsychiatric symptoms in dementia and mild cognitive impairment: results from the cardiovascular health study. JAMA 288, 1475–1483. 10.1001/jama.288.12.1475 [DOI] [PubMed] [Google Scholar]
- Manca R, Jones SA, Venneri A, 2022. Macrostructural and Microstructural White Matter Alterations Are Associated with Apathy across the Clinical Alzheimer’s Disease Spectrum. Brain Sci. 12(10):1383. doi: 10.3390/brainsci12101383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manolio TA, et al. 1994. Magnetic resonance abnormalities and cardiovascular disease in older adults. The Cardiovascular Health Study. Stroke 25, 318–327. 10.1161/01.str.25.2.318 [DOI] [PubMed] [Google Scholar]
- Matsuoka T, Imai A, Narumoto J, 2023. Neuroimaging of mild behavioral impairment: A systematic review. Psychiatry and Clinical Neurosciences Reports 2, e81. 10.1002/pcn5.81 [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKeith IG, et al. 2017. Diagnosis and management of dementia with Lewy bodies: Fourth consensus report of the DLB Consortium. Neurology 89, 88–100. 10.1212/WNL.0000000000004058 [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKhann G, Drachman D, Folstein M, Katzman R, Price D, Stadlan EM, 1984. Clinical diagnosis of Alzheimer’s disease: Report of the NINCDS-ADRDA Work Group* under the auspices of Department of Health and Human Services Task Force on Alzheimer’s Disease. Neurology 34, 939–939. 10.1212/WNL.34.7.939 [DOI] [PubMed] [Google Scholar]
- Miao R, Chen H-Y, Robert P, Smith EE, Ismail Z, MEMENTO Study Group, 2021. White matter hyperintensities and mild behavioral impairment: Findings from the MEMENTO cohort study. Cereb Circ Cogn Behav 2, 100028. 10.1016/j.cccb.2021.100028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Misquitta K, Dadar M, Louis Collins D, Tartaglia MC, 2020. White matter hyperintensities and neuropsychiatric symptoms in mild cognitive impairment and Alzheimer’s disease. Neuroimage Clin 28, 102367. 10.1016/j.nicl.2020.102367 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Monsell SE, et al. 2016. Results from the NACC Uniform Data Set neuropsychological battery Crosswalk Study Running head: Neuropsychological Battery Crosswalk Study Results. Alzheimer Dis Assoc Disord 30, 134–139. 10.1097/WAD.0000000000000111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morris JC, et al. 2006. The Uniform Data Set (UDS): Clinical and Cognitive Variables and Descriptive Data From Alzheimer Disease Centers. Alzheimer Disease & Associated Disorders 20, 210–216. 10.1097/01.wad.0000213865.09806.92 [DOI] [PubMed] [Google Scholar]
- Neary D, et al. 1998. Frontotemporal lobar degeneration: A consensus on clinical diagnostic criteria. Neurology 51, 1546–1554. 10.1212/WNL.51.6.1546 [DOI] [PubMed] [Google Scholar]
- Ng KP, Chiew H, Rosa-Neto P, Kandiah N, Ismail Z, Gauthier S, 2021. Associations of AT(N) biomarkers with neuropsychiatric symptoms in preclinical Alzheimer’s disease and cognitively unimpaired individuals. Translational Neurodegeneration 10, 11. 10.1186/s40035-021-00236-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peters ME, Lyketsos CG, 2015. Beyond Memory: A Focus on the Other Neuropsychiatric Symptoms of Dementia. Am J Geriatr Psychiatry 23, 115–118. 10.1016/j.jagp.2014.11.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petersen RC, Smith GE, Waring SC, Ivnik RJ, Tangalos EG, Kokmen E, 1999. Mild Cognitive Impairment: Clinical Characterization and Outcome. Arch Neurol 56, 303. 10.1001/archneur.56.3.303 [DOI] [PubMed] [Google Scholar]
- Prins ND, Scheltens P, 2015. White matter hyperintensities, cognitive impairment and dementia: an update. Nat Rev Neurol 11, 157–165. 10.1038/nrneurol.2015.10 [DOI] [PubMed] [Google Scholar]
- Puzo C, et al. 2019. Independent effects of white matter hyperintensities on cognitive, neuropsychiatric, and functional decline: a longitudinal investigation using the National Alzheimer’s Coordinating Center Uniform Data Set. Alzheimer’s Research & Therapy 11, 64. 10.1186/s13195-019-0521-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quandt F, et al. 2020. Higher white matter hyperintensity lesion load is associated with reduced long-range functional connectivity. Brain Commun 2, fcaa111. 10.1093/braincomms/fcaa111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberto N, et al. 2021. Neuropsychiatric Profile as a Predictor of Cognitive Decline in Mild Cognitive Impairment. Frontiers in Aging Neuroscience 13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roman GC, et al. 1993. Vascular dementia: Diagnostic criteria for research studies: Report of the NINDS-AIREN International Workshop. Neurology 43, 250–250. 10.1212/WNL.43.2.250 [DOI] [PubMed] [Google Scholar]
- Rosenberg PB, Mielke MM, Appleby B, Oh E, Leoutsakos J-M, Lyketsos CG, 2011. Neuropsychiatric symptoms in MCI subtypes: the importance of executive dysfunction. Int J Geriatr Psychiatry 26, 364–372. 10.1002/gps.2535 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ryu W-S, et al. 2014. Grading and interpretation of white matter hyperintensities using statistical maps. Stroke 45, 3567–3575. 10.1161/STROKEAHA.114.006662 [DOI] [PubMed] [Google Scholar]
- Samara AA, et al. 2022. Preeclampsia, gestational hypertension and incident dementia: A systematic review and meta-analysis of published evidence. Pregnancy Hypertens 30, 192–197. 10.1016/j.preghy.2022.10.008 [DOI] [PubMed] [Google Scholar]
- Schneider JA, Arvanitakis Z, Leurgans SE, Bennett DA, 2009. The Neuropathology of Probable Alzheimer’s Disease and Mild Cognitive Impairment. Ann Neurol 66, 200–208. 10.1002/ana.21706 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shim YS, et al. 2015. Pathological Correlates of White Matter Hyperintensities on MRI. Dement Geriatr Cogn Disord 39, 92–104. 10.1159/000366411 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siokas V, Liampas I, Lyketsos CG, Dardiotis E, 2022. Association between Motor Signs and Cognitive Performance in Cognitively Unimpaired Older Adults: A Cross-Sectional Study Using the NACC Database. Brain Sci 12, 1365. 10.3390/brainsci12101365 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sokołowski A, et al. 2023. Neuropsychiatric symptoms and imbalance of atrophy in behavioral variant frontotemporal dementia. Hum Brain Mapp 44, 5013–5029. 10.1002/hbm.26428 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Staekenborg SS, Gillissen F, Romkes R, Pijnenburg YAL, Barkhof F, Scheltens P, van der Flier WM, 2008. Behavioural and psychological symptoms are not related to white matter hyperintensities and medial temporal lobe atrophy in Alzheimer’s disease. Int J Geriatr Psychiatry 23, 387–392. 10.1002/gps.1891 [DOI] [PubMed] [Google Scholar]
- Sudo FK, et al. 2012. Dysexecutive syndrome and cerebrovascular disease in non-amnestic mild cognitive impairment: A systematic review of the literature. Dement Neuropsychol 6, 145–151. 10.1590/S1980-57642012DN06030006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taragano FE, et al. 2018. Risk of Conversion to Dementia in a Mild Behavioral Impairment Group Compared to a Psychiatric Group and to a Mild Cognitive Impairment Group. J Alzheimers Dis 62, 227–238. 10.3233/JAD-170632 [DOI] [PubMed] [Google Scholar]
- Taragano FE, et al. 2009. Mild behavioral impairment and risk of dementia: a prospective cohort study of 358 patients. J Clin Psychiatry 70, 584–592. 10.4088/jcp.08m04181 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tuladhar AM, et al. 2015. Relationship Between White Matter Hyperintensities, Cortical Thickness, and Cognition. Stroke 46, 425–432. 10.1161/STROKEAHA.114.007146 [DOI] [PubMed] [Google Scholar]
- Tumati S, et al. 2023. Neuropsychiatric symptoms and their relationship with white matter hyperintensities in mild vascular cognitive impairment, mild cognitive impairment, Alzheimer’s disease and mixed dementia – a COMPASS-ND study. The American Journal of Geriatric Psychiatry 31, S45–S46. 10.1016/j.jagp.2022.12.203 [DOI] [Google Scholar]
- Weintraub S, et al. 2009. The Alzheimer’s Disease Centers’ Uniform Data Set (UDS): The Neuropsychologic Test Battery. Alzheimer Disease & Associated Disorders 23, 91–101. 10.1097/WAD.0b013e318191c7dd [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winblad B, et al. 2004. Mild cognitive impairment - beyond controversies, towards a consensus: report of the International Working Group on Mild Cognitive Impairment. J Intern Med 256, 240–246. 10.1111/j.1365-2796.2004.01380.x [DOI] [PubMed] [Google Scholar]
- Yang D, et al. 2020. Impaired Structural Network Properties Caused by White Matter Hyperintensity Related to Cognitive Decline. Frontiers in Neurology 11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang L, Shu J, Yan A, Yang F, Xu Z, Wei W, 2022. White matter hyperintensities-related cortical changes and correlation with mild behavioral impairment. Advances in Medical Sciences 67, 241–249. 10.1016/j.advms.2022.06.002 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
For further information on access to the NACC database, please contact NACC (contact details can be found at https://naccdata.org/).


