Abstract
The capabilities of the human ear are remarkable. We can normally detect acoustic stimuli down to a threshold sound-pressure level of 0 dB (decibels) at the entrance to the external ear, which elicits eardrum vibrations in the picometer range. From this threshold up to the onset of pain, 120 dB, our ears can encompass sounds that differ in power by a trillionfold. The comprehension of speech and enjoyment of music result from our ability to distinguish between tones that differ in frequency by only 0.2%. All these capabilities vanish upon damage to the ear's receptors, the mechanoreceptive sensory hair cells. Each cochlea, the auditory organ of the inner ear, contains some 16,000 such cells that are frequency-tuned between ∼20 Hz (cycles per second) and 20,000 Hz. Remarkably enough, hair cells do not simply capture sound energy: they can also exhibit an active process whereby sound signals are amplified, tuned, and scaled. This article describes the active process in detail and offers evidence that its striking features emerge from the operation of hair cells on the brink of an oscillatory instability—one example of the critical phenomena that are widespread in physics.
Keywords: auditory system, cochlea, gating spring, hair bundle, transduction, vestibular system
Introduction
The mills of the gods grind slowly, it is said, but they grind exceedingly fine. Although the evidence for that proposition is anecdotal, much the same can be said—with far better scientific documentation—for the process of evolution through natural selection. Over unfathomable amounts of time, the stark choice between life and death has yielded the adaptations that delight children and astonish biologists, older individuals who have not altogether lost their sense of wonder.
A case in point is hearing. A predator that can hear its prey before its prey hears it enjoys an improved opportunity for a good meal and for advancing its species. Although a herbivore gains little by listening to plants, a prey animal—a niche often assigned to a herbivore—enhances its probability of survival if it can hear a potential predator before the predator hears it. The perpetual arms race between predators and prey has led to the auricular specializations of canids versus lagomorphs, of felids versus rodents, and of numerous other paired adversaries. Even more remarkably, natural selection has taught the apparatus of hearing a profoundly important lesson in fundamental physics. We intend in this article to convey how the ear succeeds at its tasks by poising its sensory receptor cells on the brink of an oscillatory instability—a critical point. We shall relate the principle of criticality in hearing to other critical behaviors in biology and physics.
The Ear's Apparatus
The characteristics of an auditory system are shaped by the nature of the relevant stimuli. Sounds are pressure disturbances, normally including several sinusoidal components, that propagate through fluids—air or water—and sometimes solids—rocks, earth, plants, and the skull. In each instance, longitudinal waves of alternate compression and rarefaction move at a speed set by the density and compressibility of the medium. Propagation is modulated by refraction, reflection, and absorbance, phenomena that further restrict the range of sounds suitable for use by animals.
An auditory receptor organ must capture the physical stimulus—a sound wave—and convert it into the electrical currency of the nervous system. Although ears differ widely in their details, one fundamental feature is conserved. In every organ of every chordate that can hear, sound is detected by a remarkable biological strain gauge, the hair cell (Fig. 1). Dating from the Cambrian Period or even earlier, the hair cell evidently performs so well that it has never been supplanted. Furthermore, the hair cell serves in other contexts for which mechanical sensitivity is needed, including the vestibular organs that detect head accelerations, such as that owing to gravity, and the lateral-line organs that render fishes and some amphibians responsive to water motion.
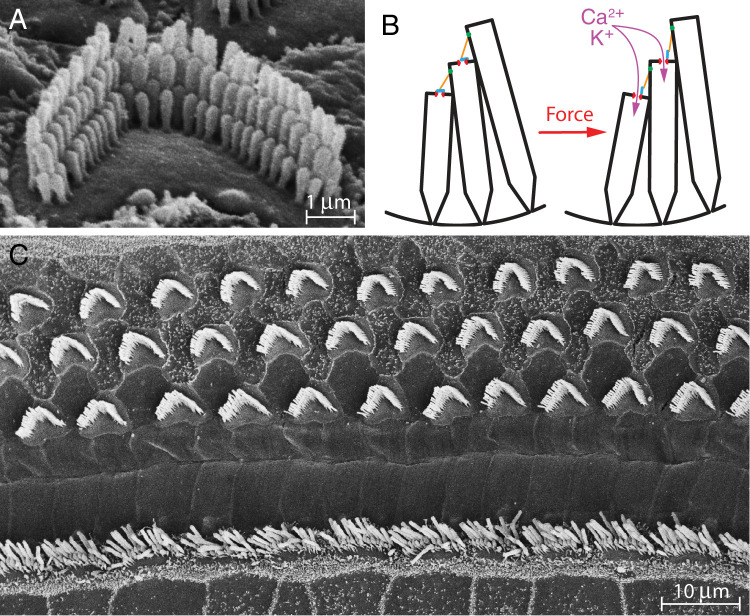
Figure 1.
Hair cells: the sensory receptors of the inner ear and potential substrate for critical behavior. A, The hair bundle that protrudes from the apical surface of a cochlear outer hair cell is a palisade of ∼85 stereocilia, each of which is an enlarged microvillus with a core of cross-linked actin filaments. B, A schematic figure depicts the mechanoelectrical transduction process. In the undisturbed hair bundle (left), most ion channels (red) at the stereociliary tips are closed, as represented here by gates (blue). When mechanical stimulation from a sound moves the bundle in the positive direction, toward its tall edge (right), the shear between adjacent stereocilia increases the tension in tip links (orange) that interconnect the stereocilia. As a result, the channels are opened, cations enter the cell and depolarize it, and the cell communicates this excitation to an afferent nerve fiber. C, This scanning electron micrograph of a short cochlear segment from a young mouse shows hair bundles in the organ of Corti. The 10 straight bundles near the bottom belong to inner hair cells, which send electrical signals into the brain. Threefold as many bundles near the top extend from outer hair cells, the sites of the cochlear active process. In addition to protruding from a common sheet of hair cells, the bundles are ordinarily surmounted by a continuous tectorial membrane that has been removed during preparation. These two interconnections among the bundles constitute a potential means of eliciting cooperative and critical behaviors.
The mechanically sensitive component of any hair cell is its hair bundle, a cluster of tens to a few hundreds of stereocilia, which are cylindrical processes—essentially giant microvilli—that protrude from the cell's apical surface (Fig. 1A). The stereocilia are graded in length such that one edge of a hair bundle is short and the opposite edge is tall. Along that axis, the tip of each stereocilium is joined to the side of a longer adjacent stereocilium by a cadherin-containing tip link ∼150 nm in length. At the lower insertion of each tip link, mechanical force is sensed by a transduction channel—a protein complex that includes the pore-forming protein TMC (Pan et al., 2018; Qiu and Müller, 2022). When the top of a hair bundle is deflected in the direction of its tall edge, defined as the positive direction, tension in the tip link increases, which opens transduction channels (Fig. 1B) and leads to a cellular depolarization (Hudspeth and Corey, 1977). Conversely, negative stimulation reduces the tension, allows channels to close, and cause a hyperpolarization.
The arrangement of hair cells within a receptor organ determines the nature of the relevant stimulus. We shall consider the example of most immediate interest: the human auditory apparatus. When we hear a sound, each elastic eardrum vibrates in response to the pressure changes, and its movement is communicated by the three tiny bones of the middle ear to the inner ear. In mammals, this auditory receptor organ is the cochlea, whose name stems from the Greek “κοκλοσ,” a gastropod’s shell. Like a snail's shell, the cochlea is a tapered helical spiral, which in humans encompasses almost three turns. The interior of the structure includes three liquid-filled chambers, or scalae, which coil around a common axis and are separated from one another by a pair of elastic partitions. The thicker of these, the basilar membrane, supports the epithelial organ of Corti, which includes some 16,000 hair cells (Retzius, 1884) generally arrayed in four parallel rows (Fig. 1C).
A remarkable feature of a cochlea is tonotopy, the systematic representation of stimulus frequencies (pitches). Each position along the human cochlea's 35 mm spiral (von Békésy, 1960) responds best to a specific frequency of stimulation, which is called the characteristic frequency for that place. Moreover, the characteristic frequencies are arranged in a monotonic progression, with the lowest frequencies, ∼20 Hz, at the organ's apex and the highest frequencies—roughly 20,000 Hz—at the cochlear base. The mapping of distance onto frequency is approximately logarithmic: a specific increment of cochlear length corresponds to a particular ratio of the frequencies at the two ends of that segment. In the human, for example, the characteristic frequency doubles every 5 mm or so (Greenwood, 1990).
The Requirements of Hearing
What requirements must an auditory system address? The first is speed. Frequencies in the auditory range—up to 100 kHz in some mammals—correspond to pressure disturbances that are too fast for a transduction mechanism based on chemical reactions. Instead, transduction by the sensory hair cells of the inner ear relies on direct mechanical gating of ion channels to produce an electrical response (Corey and Hudspeth, 1979). However, frictional forces on the inner ears’ moving parts, including the sensory hair bundles (Bormuth et al., 2014), threaten to dissipate the energy derived from sound inputs.
A second requirement is sensitivity. Almost any animal is likely to benefit from hearing with the greatest practical sensitivity. Humans are no exception: our hearing extends nearly to the physical limits set by thermal noise at the eardrum, in the liquids of the inner ear, and at the mechanosensory hair bundles of individual hair cells. At the hearing threshold for a 1 kHz tone, the air pressure disturbance and the acoustic energy flux that reach the auricle are ∼20 µPa and 1 pW·m−2, respectively (Sivian and White, 1933). The external ear captures and funnels the acoustic stimulus to the eardrum, where the sound-pressure level at 1 kHz is ∼2.6-fold as large as that in the free field (Killion, 1978). Assuming a middle-ear stiffness of 1.5 kN·m−1 (Aibara et al., 2001), a 30 mm2 eardrum vibrates at threshold with an estimated amplitude of 0.6 pm, which is comparable with or even smaller than the root-mean-squared magnitude of its own thermal fluctuations. Within the inner ear, on the basilar membrane that supports the organ of Corti, mechanical vibrations are larger, with an amplitude in the nanometer range (Robles and Ruggero, 2001), but comparable with thermal motion of the mechanosensory hair bundles (Denk et al., 1989).
High sensitivity might in principle stem from mechanical resonance, which is an effective means of capturing periodic signals—such as most important sounds—that involve protracted sinusoidal disturbances. A resonant system oscillates in response to stimulus energy, and its response grows as it accumulates energy over multiple stimulus cycles. The ratio of the energy stored in the system in the steady state to the average energy dissipated in one cycle of oscillation provides a measure of the quality of the resonance. Friction from viscous drag by the inner ear's fluids (Gold and Gray, 1948), and also from internal degrees of freedom within the organ of Corti and hair cells (Bormuth et al., 2014; Roldán et al., 2021), challenges the inner ear's ability to benefit from passive resonance.
A third requirement for hearing is a fine-grained ability to discriminate among the frequencies within the auditory range. Sonograms of human speech or birdsong illustrate the complexity of sounds that must be analyzed to recognize particular speech phrases or conspecific songs (Fig. 2). For human speech dominated by frequencies of 200–3,200 Hz (Fant, 2004), a signal only 100 ms in length—a duration typical of phonemes—encompasses 20–320 cycles, enough that resonance is useful to enhance sensitivity and increase the signal-to-noise ratio. Moreover, Fourier analysis assures us that even the complex waveforms of natural sounds can be decomposed into their sinusoidal components. The cochlea disperses the energies derived from these components to appropriate positions along its tonotopic axis, with higher frequencies closer to the base and lower frequencies closer the apex. The signals are then detected by different hair cells that are tuned to the corresponding frequencies.
Figure 2.
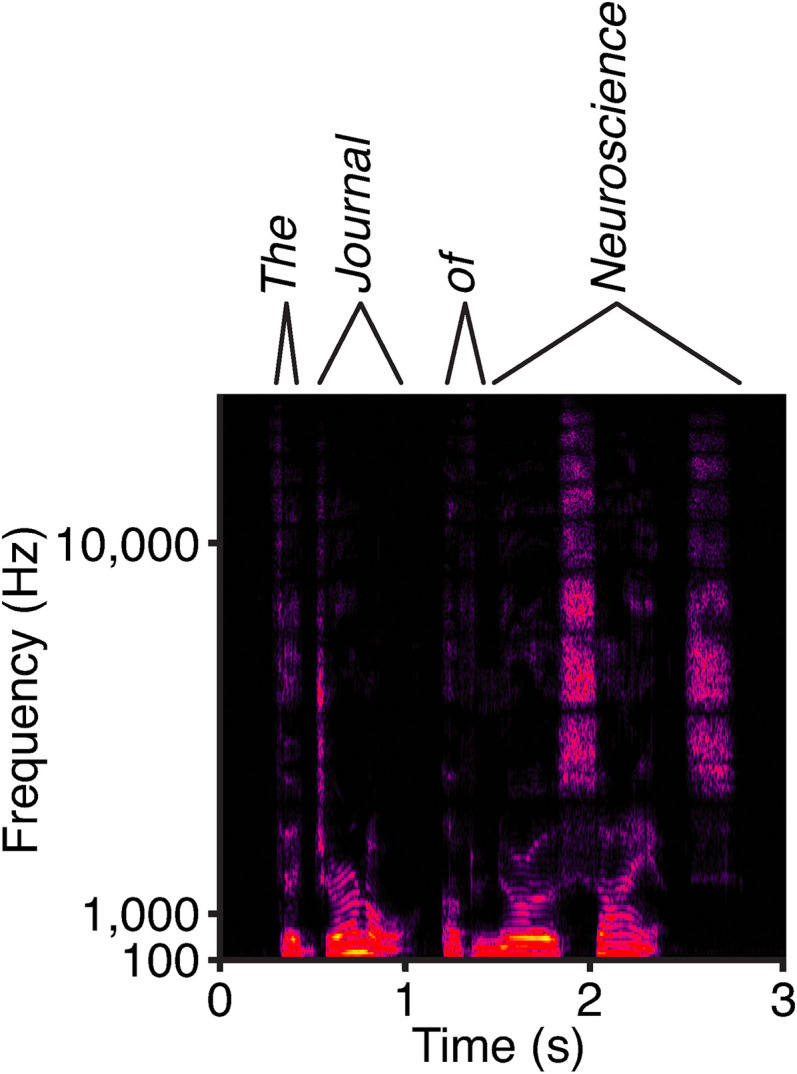
The ear's daily task. A sonogram, which depicts the frequency content of a 3 s sample of human speech as a function of time, demonstrates the complexity of the auditory signals. Sound intensity is represented on a scale from blue, for the weakest signals, to red and finally yellow for the most intense. The auditory system uses frequency, intensity, and timing information to decode the utterance as “The Journal of Neuroscience.” Some words, such as “the” and “of,” garner only rudimentary representation. Note the precise timing of the hard “j” at the onset of “journal.” Equally striking are the high-frequency sibilants at the beginning and conclusion of “science.” The protracted vowel sounds in “journal,” “neuro,” and “science” involve distinctive stacks of low frequencies, the formants.
The fourth important requirement of an auditory system is a broad dynamic range. Natural sounds vary enormously in intensity. From the limit set by thermal noise in an ear to the deafening peal of thunder, the sound-pressure level increases from 0 dB (decibels) to 120 dB, which corresponds to a millionfold variation in the magnitude of pressure disturbances or 12 orders of magnitude in acoustic intensity. To encompass that range, an auditory system must achieve high sensitivity without being saturated or possibly damaged by the loudest likely sounds. These requirements come at a price: a compressive nonlinearity in the relation between the input and the output. The auditory nonlinearity introduces distortions in our perception of natural sounds, but also suppresses noise and thereby enhances our perception of speech in noisy environments (Goldstein, 1967; Avan et al., 2013).
The Active Process of Auditory Organs
Although natural selection might have addressed the requirements of hearing independently, it found a single strategy that provides a solution to all four! This solution involves the so-called active process, that is, a metabolically powered apparatus that enhances the ear's performance as a receptor of sound. Although such a mechanism might in principle occur anywhere in the ear, evidence suggests that it is situated in hair cells, the cochlea's sensory receptors.
The strongest hint that the human ear possesses an active process is the presence of spontaneous otoacoustic emissions, pure tones that emerge from the ears of ∼70% of people with normal hearing (Kemp, 1978; Talmadge et al., 1993; Penner and Zhang, 1997). Because these signals represent the broadcasting of sound energy, they must inevitably stem from an energy-consuming active process. From individual to individual, emissions vary in number from one to >30 (Braun, 2013) and are most common and strongest in newborns—especially premature infants—but decline in number and strength with age (Abdala et al., 2017). Because spontaneous emissions also occur in amphibians, and in reptiles including birds, they betoken an active process that is ubiquitous and perhaps universal in tetrapod vertebrates (Manley, 2001).
The second indication that the human ear possesses an active process is that, even though the basilar membrane and organ of Corti move through viscous liquid, the ear acts as if its resonance is underdamped (Gold and Gray, 1948). In other words, energy derived from a protracted sound stimulus can accumulate progressively until the sound is detected. The most likely explanation for this effect is that fluid damping is countered by the mechanical activity of some cochlear component. The combination of measurements and theoretical descriptions of basilar-membrane vibrations confirms that the organ of Corti pumps energy into sound-evoked vibrations. The result is net energy gain at positions just basal to the characteristic place along the basilar membrane at which vibrations are maximal for a given frequency of sound (Zweig, 1991; Shera, 2007; Fisher et al., 2012; Dong and Olson, 2013).
Cochlear hair cells can make use of a unique external energy source to power the active process. As a constituent of a continuous epithelium, each hair cell separates dissimilar ionic solutions. Its basolateral surface is bathed in perilymph, an ordinary extracellular medium with ∼120 mM Na+, 2 mM K+, and 2 mM Ca2+. The cell's apical surface, the site of its mechanosensitive hair bundle, is exposed instead to a unique solution termed endolymph with a composition of 2 mM Na+, 120 mM K+, and only 20 µM Ca2+. Because the interior of the hair cell is at a potential near the usual value of −70 mV with respect to that in perilymph, the opening of transduction channels in the hair bundle allows K+ in endolymph to carry a positive current into the cell. The stria vascularis, an extensive epithelial surface lining the cochlea's scala media, expends energy to maintain the K+ gradient—but does much more. Through an elaborate combination of pumps, transporters, and channels, the stria additionally generates a standing transepithelial potential of 100 mV, with the hair cell's apical surface positive (Adachi et al., 2013). This arrangement means that the electromotive force driving K+ into a hair cell totals 170 mV, which enhances the magnitude of the hair cell's transduction current and receptor potential. Moreover, the system provides a remarkable energetic advantage: although the hair cell produces large electrical signals, K+ enters its apical surface and exits its basolateral surface passively, so the cell needs to perform no metabolic work to support signaling.
What does a hair cell do with its special energy source? In the mammalian cochlea, receptor potentials lead to somatic length changes of outer hair cells (Ashmore, 2008). This phenomenon—termed electromotility—is widely thought to be involved in cycle-by-cycle amplification of sound-evoked vibrations within the organ of Corti (Dallos et al., 2008; Dewey et al., 2021), although this point is still debated (van der Heijden and Vavakou, 2021). In addition, as discussed below, the hair bundle not only operates as the hair cell's mechanosensory antenna but also harnesses Ca2+ entry through its transduction channels to produce active movements (Hudspeth, 2008; Martin, 2008). Unlike electromotility, which appears to operate only in mammals, active hair-bundle motility has been reported in other tetrapods, including frogs, turtles, and birds, and might be at work in mammals as well.
Evidence of Mechanical Activity in Hair Bundles
For the extensively investigated hair cells of the bullfrog, there is definitive evidence that hair bundles can mobilize internal resources of energy to counter liquid viscosity, power spontaneous oscillations, and perform work to amplify their responsiveness. A first manifestation of mechanical activity is apparent from simple inspection of spontaneous hair-bundle oscillations. Any process at thermal equilibrium must be time reversible: by the principle of microscopic reversibility, dynamical trajectories in one direction must be indistinguishable from those in the opposite direction. Some hair-bundle movements are strikingly asymmetrical in time, however, and thus reveal the arrow of time. Each half-cycle of these relaxation oscillations has two components, a fast initial stroke followed by a slower, smooth relaxation in the same direction (Martin et al., 2003). Oscillations of this sort inevitably involve dissipation and must therefore stem from a process that consumes energy. Quantifying time irreversibility affords a means of demonstrating activity and estimating a lower bound for the power that must be dissipated—or the rate at which entropy must be produced—by the hair cell to sustain spontaneous oscillations (Roldán et al., 2021). For the oscillatory hair bundles of the bullfrog's sacculus, however, this approach provides estimates of only ∼1 zJ per cycle, which is small indeed. By way of comparison, the viscous drag in water on a sinusoidally oscillating sphere with the size of a hair bundle would dissipate 4 zJ per cycle at a frequency and an amplitude of motion comparable with those evinced by hair bundles, 10 Hz and 20 nm, respectively. Underestimating the power dissipated comes as no surprise when only the hair bundle's position is measured, for at least one additional, but hidden, degree of freedom is necessary to account for active oscillations. Hair-bundle oscillations are thought to result from a dynamic interplay between gating of the mechanotransduction channels, tip-link tensioning by myosin motors, and feedback by Ca2+ ions that flow through open transduction channels (Martin et al., 2003; Martin and Hudspeth, 2021).
Another signature of the active process involves the regularity of spontaneous oscillations. An accurate timepiece is often an expensive one, and the precise control of oscillation frequency likewise imposes a cost in terms of free energy (Barato and Seifert, 2015; Cao et al., 2015; Oberreiter et al., 2022). At one extreme, imagine a passive hair bundle buffeted by water molecules. Although the thermal fluctuations of this bundle could show some regularity if its passive mechanical properties allowed for resonance at a specific frequency and thus filtered the noise, individual hair bundles are overdamped by a viscous medium. For a bundle from the bullfrog's sacculus, typical values for the bundle's properties—the stiffness , the mass , and the friction coefficient —yield a resonator's quality factor, , well below unity: viscous drag is too large to permit cycle-by-cycle energy accumulation into the hair bundle. The power spectrum of position fluctuations of a passive bundle correspondingly shows low-pass characteristics (Denk et al., 1989). Actively oscillating bundles instead exhibit peaks in their power spectra that correspond to well-defined periods of oscillation (Martin et al., 2003; Barral et al., 2018). Noise nevertheless matters, for the phase coherence of hair-bundle position is lost after only a few cycles of oscillation (Martin et al., 2001). Models that account for the internal motor responsible for the oscillation can provide a tight bound on the rate of entropy production (Roldán et al., 2021; Tucci et al., 2022). For a hair bundle of the bullfrog's sacculus, this free-energy expenditure is at least 400 zJ per cycle, which amounts to a hundredfold the thermal energy—100·kBT—or the consumption of 10 ATP molecules per cycle. Oscillation at 10 Hz thus requires a power dissipation of 4 aW, an energy flux of 1,000·kBT per second, or the hydrolysis of 100 ATP molecules per second.
Sinusoidal stimulation of a hair bundle with a flexible glass fiber provides a more direct means both of assessing activity and of emphasizing its benefits for auditory responsiveness. When an oscillatory bundle is stimulated at a frequency less than its characteristic or natural frequency, it exhibits a phase lead with respect to the stimulus (Martin and Hudspeth, 1999). In other words, although the bundle is subjected to an external force by the fiber and moves in a one-to-one relationship with it, the bundle's motion actually anticipates that of the fiber. Such a pattern, which cannot occur in a passive system, implies that the bundle possesses an internal source of power that allows it to outpace the fiber. Plotting the force produced by the bundle against its motion results in an oval trajectory whose area—the integral of force with respect to displacement—represents the work done by the bundle (Martin and Hudspeth, 1999). The experiment demonstrates that the bundle pumps mechanical energy into the fiber by trying to hurry it along at the bundle's chosen frequency. Dissipation is thus negative: the bundle's internal activity more than counters the resistance imposed by the liquid and other sources of dissipation. The energy consumption per cycle is again at least several tens of zeptojoules per cycle.
The final demonstration of hair-bundle activity rests on a general result of statistical physics for systems at thermal equilibrium: the fluctuation–dissipation theorem (Kubo, 1966; Chandler, 1987). As an object is moved through a viscous liquid such as water by application of an external force, its motion is resisted by drag that stems from molecular collisions. When no force is applied, the object nonetheless undergoes random displacements—Brownian motion—as a result of such collisions. If the object operates at thermal equilibrium, the fluctuation–dissipation theorem rigorously relates the drag on the object to the magnitude of thermal noise. Because measurements indicate that a hair bundle violates this relationship near the characteristic frequency of spontaneous oscillation, the bundle cannot be passive at those frequencies (Martin et al., 2001). In conjunction with the previously described phenomena, this result demonstrates that an individual hair bundle is capable of using internal sources of energy to boost its response to external forces. The sensitivity to weak stimuli is maximal for stimulation near the bundle's characteristic frequency but decreases at greater or lesser frequencies (Martin and Hudspeth, 2001). Sensitivity is thus actively enhanced and tuned near the characteristic frequency of spontaneous oscillation.
A Hopf Bifurcation
In addition to spontaneous otoacoustic emissions, and in keeping with the desired features of auditory systems, three phenomena are generally associated with the active process in the ears of all tetrapod classes (Manley and Köppl, 1998; Manley, 2000, 2001). First, acoustic stimuli are mechanically amplified by healthy ears. In the mammalian cochlea, amplification lowers the sound-pressure level that elicits a threshold response by a hundred- to a thousandfold, which corresponds to a gain in sensitivity of 40–60 dB (Robles and Ruggero, 2001). When our biological hearing aids fail, acoustic sensitivity falls to <1% of the control value and we become hard of hearing. Second, frequency tuning is sharpened within tetrapod hearing organs. Humans can distinguish pure tones that differ in frequency by as little as 0.2% (Moore, 1973). This resolution is key to our ability to parse the subtle nuances of speech. And third, as mentioned above, hearing displays a compressive nonlinearity. As the amplitude of a stimulus increases, the response of the basilar membrane grows at a considerably lower rate—roughly as the cube root of the stimulus (Ruggero et al., 1997): as many as six orders of magnitude of stimulation are squeezed into only two or three orders of magnitude of response (Fig. 3).
Figure 3.
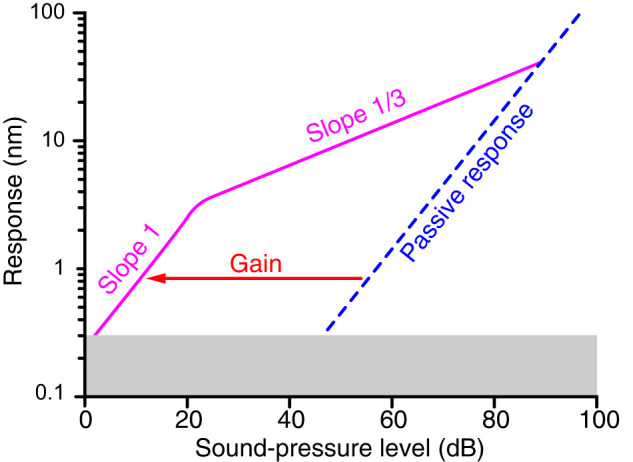
Signature of criticality in the cochlea. An idealized “level function” portrays the logarithm of a cochlear response to the sound-pressure level—also a logarithmic measure—that elicits it. The response represents the oscillatory movement of the basilar membrane or of some component of the organ of Corti for a frequency of stimulation—the characteristic frequency—that accords with the tonotopic map. In the present instance, the response above the noise floor (gray) covers three orders of magnitude, a much narrower range than the six orders of magnitude in sound-pressure level that are characteristic of human hearing. Over most of the range, the slope of the level function is 1/3 (pink), indicating a power law relationship with this exponent. This form of compressive nonlinearity is characteristic of a system operating near a Hopf bifurcation and provides strong evidence for critical oscillators in the cochlea. The response is instead linear (slope 1) for signals so weak that Brownian motion is comparable with the movements elicited by the stimulus, which limits the sensitivity of hearing. When the active process fails, and one becomes “hard of hearing,” the passive response of the cochlea is nearly linear at all sound-pressure levels (blue dashed line) and hearing is confined to a narrow range of quite strong stimuli. At each sound intensity, the magnitude ratio between the active response (solid pink line) and the passive one (dashed blue line) quantifies amplification by the active process. Amplification is highest when we need it most, for faint stimuli, and decreases progressively at increasing sound-pressure levels. For very strong stimuli that overwhelm active force production by the hair cells, the response of the organ of Corti is dictated by its passive mechanics. Nonlinear amplification of cochlear vibrations lowers the threshold of hearing by 40–50 dB (red arrow).
Individual hair bundles, the sensory organelles of the inner ear, exhibit all three useful characteristics of the active process—amplification, tuning, and compression—as well as unprovoked oscillations that might underlie spontaneous otoacoustic emissions. A hair bundle can amplify its mechanical inputs (Martin and Hudspeth, 1999), respond to a narrow band of frequencies (Martin and Hudspeth, 2001; Barral et al., 2010; Salvi et al., 2015), and compress a range of stimuli in proportion to the cube root of their magnitudes (Martin and Hudspeth, 2001). Because hair bundles are conserved throughout the chordates, and certainly among the tetrapods, they are plausible candidates to mediate the active process in general (Hudspeth, 2008, 2014; Hudspeth et al., 2010; Martin and Hudspeth, 2021).
Aside from their occurrence in hair bundles, what could link the four characteristics of the active process? Remarkably enough, they would all emerge from operation at a Hopf bifurcation (Camalet et al., 2000; Eguíluz et al., 2000; Hudspeth, 2008, 2014; Hudspeth et al., 2010; Reichenbach and Hudspeth, 2014; Martin and Hudspeth, 2021). By definition, a bifurcation occurs in a system when continuous variation of some parameter's value causes an abrupt qualitative change in the system's response. A structural column, for example, may bear a range of axial loads while exhibiting a proportional compression. At some point, however, the column can fail abruptly by buckling, a qualitatively different behavior. A Hopf bifurcation, which can arise in an active dynamical system with two or more degrees of freedom, involves an abrupt transition between a quiescent state and a dynamic state of spontaneous oscillation at a characteristic frequency (Fig. 4A). Such a bifurcation occurs, for example, when raising the amplification of a public-address system ceases to produce successively louder signals, but instead suddenly elicits an ear-splitting howl. With hair bundles, the extracellular Ca2+ concentration in the surrounding endolymph (Martin et al., 2003; Tinevez et al., 2007), the transmembrane potential of the hair cell (Meenderink et al., 2015), an external load (Salvi et al., 2015), and the synaptic activity of efferent nerve fibers (Lin and Bozovic, 2020) have been identified as control parameters for the dynamic state.
Figure 4.
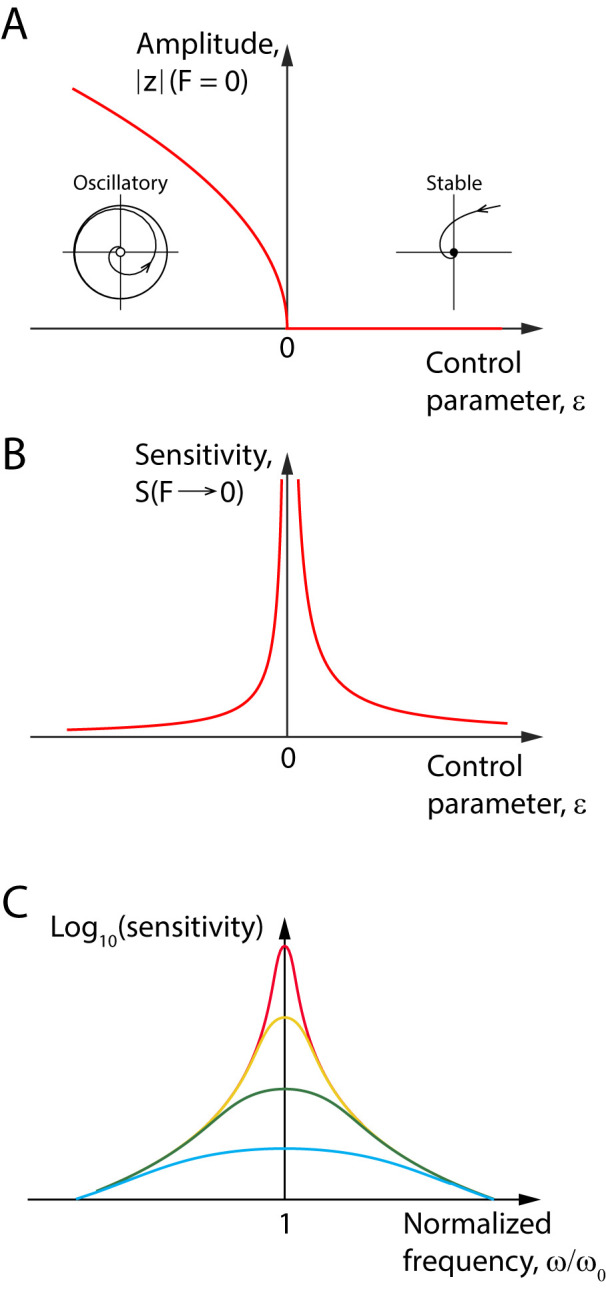
Critical behavior at a Hopf bifurcation. A, A bifurcation diagram shows the amplitude of spontaneous oscillations as a function of a control parameter, , in an active dynamical system that undergoes an oscillatory instability—a Hopf bifurcation—at the critical value . At positive values of the control parameter, the system is endowed with a stable fixed point: it relaxes to this point in response to an external perturbation (right inset). When the control parameter crosses zero and becomes negative, the fixed point becomes unstable and the system reaches a limit cycle oscillation (left inset). Near the bifurcation point, the magnitude of the limit cycle grows as the square root of the distance, , to the critical point. This behavior is analogous to that of the spontaneous magnetization that appears in ferromagnetic materials below the Curie temperature, an example of a second-order phase transition (Kardar, 2007). B, When the system approaches criticality, , the sensitivity to vanishing amplitudes of an external periodic stimulus at the characteristic frequency of spontaneous oscillation becomes arbitrarily large. This critical behavior mirrors that of the magnetic susceptibility of a material operating at the paramagnetic-to-ferromagnetic transition. In both cases, criticality expands the dynamical range of responsiveness (Kardar, 2007). C, The decimal logarithm of the sensitivity of a critical oscillator is depicted as a function of the ratio of the frequency, , of a sinusoidal stimulus and the characteristic frequency, , of the oscillator. The sensitivity is tuned at . The peak sensitivity decreases and the bandwidth increases with the stimulus level (by a factor of 64 from red to blue) according to generic power laws (Eq. 4). Away from the characteristic frequency, the sensitivity is low and all the curves superimpose: the output is proportional to the input and the system thus evinces a linear behavior.
Any dynamical system that operates near a Hopf bifurcation exhibits generic properties that do not depend on the detailed mechanism that brings the system to the brink of spontaneous oscillation (Fig. 4B,C). More specifically, and irrespectively of complexity, the system's behavior at steady state can be described by only two degrees of freedom. In the case of a hair cell, for instance, two variables—the hair bundle's displacement from its resting position, x, and the state of the system's adaptation, y—can account for operation near a Hopf bifurcation. Proximity to the bifurcation additionally dictates that the equation of motion for the complex variable obeys the following normal form:
In the normal form, is a characteristic frequency, represents an external force, is a parameter with units of a friction coefficient, is a phase parameter, and is a complex number that sets the strength of the cubic nonlinear term. The condition ensures stability. In addition, the parameter represents a small deviation of a control parameter, , from its critical value . Beyond the critical value and with no external force , the system becomes unstable and oscillates spontaneously at the characteristic frequency, , and with an amplitude, , set by the stabilizing nonlinearity (Fig. 4A). An active dynamical system operating precisely at the bifurcation point, which corresponds to or , is said to be “critical.”
In response to a sinusoidal force at angular frequency and of Fourier amplitude , the normal form imposes that the amplitude of the phase-locked response be given by the following:
with coefficients for the linear term and for the nonlinear term. At criticality , the system acquires a remarkable behavior that cannot occur in a passive system: when the system is stimulated at its natural frequency, the linear term of the equation vanishes. Although inertia may cancel elasticity in a passive system driven at an appropriate frequency, canceling the frictional component of the linear response requires an active process. The cubic term in the equation of motion then assumes control:
The system thus evinces level functions that are described by power laws, with diverging sensitivity at low forces: there is no stimulus level low enough to elicit a regime of linear responsiveness. Away from the characteristic frequency, however, the linear response reasserts itself, resulting in a drop in sensitivity. As a result, the response is tuned, with a bandwidth, , that increases as the stimulus grows (Fig. 4C). Faint stimuli are therefore detected with both higher sensitivity and sharper frequency selectivity.
The generic properties of a critical oscillator capture salient behaviors both of individual hair bundles and of local vibrations by the entire basilar membrane (Fig. 3; Hudspeth et al., 2010). In particular, the normal form implies that a critical oscillator evinces negative friction—net energy pumping into the environment—when driven below its characteristic frequency by a weak stimulus but instead dissipates energy when driven above, as demonstrated experimentally in oscillatory hair bundles (Martin et al., 2001; Martin and Hudspeth, 2021). This generic property ensures that a traveling wave riding on a tonotopic organization of critical oscillators with descending characteristic frequencies benefits from spatial accumulation of energy gain, an experimental property of active cochlear mechanics (Shera, 2007; Fisher et al., 2012). The frequency-dependent 1/3-power law for the response additionally accounts for the level functions of distortion products that are perceived by the human ear in response to two-tone stimulation at nearby frequencies f1 and f2 (Goldstein, 1967; Jülicher et al., 2001; Barral and Martin, 2012).
Ith is remarkable that the hallmarks of a Hopf bifurcation have also been reported in the auditory organs of arthropods such as fruit flies, mosquitoes, and even the ancient bush crickets (Nadrowski et al., 2008; Mhatre and Robert, 2013; Albert and Kozlov, 2016). The mechanosensitivity of these organisms is based on very different organ structures and protein ensembles. The common mathematical description of active mechanosensitivity for sound detection in evolutionarily distant species provides compelling evidence for criticality as a general principle for hearing.
Critical Behaviors
Operation at a Hopf bifurcation is but one example of a phenomenon termed criticality. Physics abounds with systems that operate in two or more phases with distinct properties; magnetism provides a prototypical example. Iron can maintain a magnetic field indefinitely at room temperature. This behavior emerges because quantum interactions between the magnetic moments of individual iron atoms at the microscopic level result in global alignment of the moments at the macroscopic level. When the temperature exceeds a specific value called the Curie temperature, however, thermal agitation is strong enough to randomize the orientation of the magnetic moments: permanent magnetization vanishes at this critical point in favor of ordinary paramagnetism. The continuous transition between the ordered and disordered phases provides an example of a second-order phase transition. Continuous transitions and criticality can also occur between different dynamical states, which extend the concept of phase to dynamical systems (Fig. 4A).
The remarkable feature of criticality is that, at the boundary between two phases, a system exhibits emergent properties that are not observed in the conventional phases: power-law dependencies for correlations of observables as a function of distance, diverging susceptibility to small external stimuli that maximizes the dynamical range of responsiveness (Fig. 4B), and long-term memory of small perturbations. These behaviors are generic: numerous systems, although entirely distinct in their particulars, exhibit similar mathematical characteristics. The Earth's transitions into and out of ice ages, for example, can be analyzed in the same terms as the synchronized flashing of fireflies (Korobeinikov and Mcnabb, 2001; Ditlevsen, 2009; Saikia and Bora, 2020).
Power laws, a fundamental signature of criticality, have been observed in numerous biological systems (Mora and Bialek, 2011). At least in theory, gene networks can operate in this manner (Verdugo, 2018; Ma, 2021), and the excitability of neuronal networks exhibits similar patterns (Huang et al., 2007; Hesse and Gross, 2014; Yuan and Wang, 2016; Popov and Fedorov, 2023; Chandra and Magnasco, 2024). Finally, the interactions of predators and prey can display bifurcations both in theory and in practice (Fussmann et al., 2000; Toth, 2008; Stollenwerk et al., 2017).
Criticality is an idealized concept whereby the interactions of many elements at the microscopic scale collectively affect the dynamics of a system as a whole. Any real system, however, is subject to fluctuations. As a result, a critical behavior does not occur at a precise value of a control parameter but is instead smoothened. Hair cells are no exception: the transition between irregular fluctuations and regular oscillations of a hair bundle occurs over a relatively broad range of control-parameter values (Nadrowski et al., 2004; Salvi et al., 2016). A hair bundle is composed of a few tens of stereocilia, each of which is endowed with only a few, perhaps just two, transduction channels. Although these elements are mechanically coupled to promote concerted channel gating (Kozlov et al., 2007), their number is small enough that channel clatter, fluctuations of the ion channels between their open and closed states, produces a significant amount of noise (Nadrowski et al., 2004). This noise limits the maximal sensitivity that a single hair cell can achieve in response to sinusoidal stimulation to ∼10-fold the value afforded by its passive mechanical properties. Nevertheless, as detailed above, noise is low enough that the signatures of criticality remain even at the scale of a single hair cell. Mechanical coupling between hair cells of similar characteristics provides a means of increasing the size of the active system by effectively reducing the noise intensity that each cell experiences (Dierkes et al., 2008; Barral et al., 2010). The power-law scaling of level functions is accordingly observed over a broader range of input magnitude and the maximal sensitivity of each hair cell in the coupled system is enhanced.
In the cochlea, different frequencies are detected by different cells that are tuned to different characteristic frequencies and spatially distributed along the organ according to a tonotopic map. A sound input near the threshold of hearing is expected to elicit significant vibrations over a segment of the organ of Corti that spans a few hundred micrometers (Ren, 2002) and thus to recruit on the order of 100 coupled hair cells, depending on the organ's tonotopic map and the sharpness of frequency tuning. The functional unit of auditory detection thus lies at a broader scale than that of an individual hair cell. Hair-cell cooperation may explain how the cochlea raises its sensitivity by a hundred- to a thousandfold, whereas amplification by a single hair cell is much weaker. Endowed with only 16,000 hair cells, the cochlea must evidently cope with a tradeoff between devoting a sufficiently large number of hair cells to the detection of one frequency and the necessity of responding over a broad range of frequencies. Moreover, remaining at the bifurcation is straightforward: through the process of self-tuned criticality, a sensory system can automatically, promptly, and robustly take advantage of the bifurcation's many benefits even as the environment varies (Camalet et al., 2000; Hesse and Gross, 2014; Graf and Machta, 2024).
In conclusion, natural selection has endowed ears with an amazingly useful set of features seemingly custom-built for the needs of hearing. Yet the emergence of a Hopf bifurcation does not require any special intervention: as the examples above illustrate, an extensive range of dynamical systems can readily achieve criticality. Although natural selection had evidently endowed chordates with an active process by the Cambrian Period, chordates have required an additional 500 million years to understand how that process works.
References
- Abdala C, Luo P, Shera CA (2017) Characterizing spontaneous otoacoustic emissions across the human lifespan. J Acoust Soc Am 141:1874. 10.1121/1.4977192 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Adachi N, Yoshida T, Nin F, Ogata G, Yamaguchi S, Suzuki T, Komune S, Hisa Y, Hibino H, Kurachi Y (2013) The mechanism underlying maintenance of the endocochlear potential by the K+ transport system in fibrocytes of the inner ear. J Physiol 591:4459–4472. 10.1113/jphysiol.2013.258046 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aibara R, Welsh JT, Puria S, Goode RL (2001) Human middle-ear sound transfer function and cochlear input impedance. Hear Res 152:100–109. 10.1016/S0378-5955(00)00240-9 [DOI] [PubMed] [Google Scholar]
- Albert JT, Kozlov AS (2016) Comparative aspects of hearing in vertebrates and insects with antennal ears. Curr Biol 26:R1050–R1061. 10.1016/j.cub.2016.09.017 [DOI] [PubMed] [Google Scholar]
- Ashmore J (2008) Cochlear outer hair cell motility. Physiol Rev 88:38. 10.1152/physrev.00044.2006 [DOI] [PubMed] [Google Scholar]
- Avan P, Buki B, Petit C (2013) Auditory distortions: origins and functions. Physiol Rev 93:1563–1619. 10.1152/physrev.00029.2012 [DOI] [PubMed] [Google Scholar]
- Barato AC, Seifert U (2015) Thermodynamic uncertainty relation for biomolecular processes. Phys Rev Lett 114:158101. 10.1103/PhysRevLett.114.158101 [DOI] [PubMed] [Google Scholar]
- Barral J, Dierkes K, Lindner B, Jülicher F, Martin P (2010) Coupling a sensory hair-cell bundle to cyber clones enhances nonlinear amplification. Proc Natl Acad Sci U S A 107:8079–8084. 10.1073/pnas.0913657107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barral J, Jülicher F, Martin P (2018) Friction from transduction channels’ gating affects spontaneous hair-bundle oscillations. Biophys J 114:425–436. 10.1016/j.bpj.2017.11.019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barral J, Martin P (2012) Phantom tones and suppressive masking by active nonlinear oscillation of the hair-cell bundle. Proc Natl Acad Sci U S A 109:E1344–E1351. 10.1073/pnas.1202426109 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bormuth V, Barral J, Joanny J-F, Jülicher F, Martin P (2014) Transduction channels’ gating can control friction on vibrating hair-cell bundles in the ear. Proc Natl Acad Sci U S A 111:7185–7190. 10.1073/pnas.1402556111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braun M (2013) High-multiple spontaneous otoacoustic emissions confirm theory of local tuned oscillators. Springerplus 2:135. 10.1186/2193-1801-2-135 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Camalet S, Duke T, Jülicher F, Prost J (2000) Auditory sensitivity provided by self-tuned critical oscillations of hair cells. Proc Natl Acad Sci U S A 97:3183–3188. 10.1073/pnas.97.7.3183 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao Y, Wang H, Ouyang Q, Tu Y (2015) The free energy cost of accurate biochemical oscillations. Nat Phys 11:772–778. 10.1038/nphys3412 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chandler D (1987) Introduction to modern statistical mechanics. New York: Oxford University Press. [Google Scholar]
- Chandra A, Magnasco MO (2024) On the dynamics of convolutional recurrent neural networks near their critical point. Available at: https://arxiv.org/abs/2405.13854. Accessed August 13, 2024.
- Corey DP, Hudspeth AJ (1979) Response latency of vertebrate hair cells. Biophys J 26:499–506. 10.1016/S0006-3495(79)85267-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dallos P, et al. (2008) Prestin-based outer hair cell motility is necessary for mammalian cochlear amplification. Neuron 58:333–339. 10.1016/j.neuron.2008.02.028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Denk W, Webb WW, Hudspeth AJ (1989) Mechanical properties of sensory hair bundles are reflected in their Brownian motion measured with a laser differential interferometer. Proc Natl Acad Sci U S A 86:5371–5375. 10.1073/pnas.86.14.5371 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dewey JB, Altoè A, Shera CA, Applegate BE, Oghalai JS (2021) Cochlear outer hair cell electromotility enhances organ of Corti motion on a cycle-by-cycle basis at high frequencies in vivo. Proc Natl Acad Sci U S A 118:e2025206118. 10.1073/pnas.2025206118 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dierkes K, Lindner B, Julicher F (2008) Enhancement of sensitivity gain and frequency tuning by coupling of active hair bundles. Proc Natl Acad Sci U S A 105:18669–18674. 10.1073/pnas.0805752105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ditlevsen PD (2009) Bifurcation structure and noise-assisted transitions in the Pleistocene glacial cycles. Paleoceanography 24:2008PA001673. 10.1029/2008PA001673 [DOI] [Google Scholar]
- Dong W, Olson ES (2013) Detection of cochlear amplification and its activation. Biophys J 105:1067–1078. 10.1016/j.bpj.2013.06.049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eguíluz VM, Ospeck M, Choe Y, Hudspeth AJ, Magnasco MO (2000) Essential nonlinearities in hearing. Phys Rev Lett 84:5232–5235. 10.1103/PhysRevLett.84.5232 [DOI] [PubMed] [Google Scholar]
- Fant G (2004) Speech acoustics and phonetics: selected writing, graph. Dordrecht: Kluwer Academic; Darst. [Google Scholar]
- Fisher JA, Nin F, Reichenbach T, Uthaiah RC, Hudspeth AJ (2012) The spatial pattern of cochlear amplification. Neuron 76:989–997. 10.1016/j.neuron.2012.09.031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fussmann GF, Ellner SP, Shertzer KW, Hairston NG (2000) Crossing the hopf bifurcation in a live predator-prey system. Science 290:1358–1360. 10.1126/science.290.5495.1358 [DOI] [PubMed] [Google Scholar]
- Gold T, Gray J (1948) Hearing. II. The physical basis of the action of the cochlea. Proc R Soc Lond B Biol Sci 135:492–498. 10.1098/rspb.1948.0025 [DOI] [Google Scholar]
- Goldstein JL (1967) Auditory nonlinearity. J Acoust Soc Am 41:676–689. 10.1121/1.1910396 [DOI] [PubMed] [Google Scholar]
- Graf IR, Machta BB (2024) A bifurcation integrates information from many noisy ion channels and allows for milli-kelvin thermal sensitivity in the snake pit organ. Proc Natl Acad Sci U S A 121:e2308215121. 10.1073/pnas.2308215121 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greenwood DD (1990) A cochlear frequency-position function for several species—29 years later. J Acoust Soc Am 87:2592–2605. 10.1121/1.399052 [DOI] [PubMed] [Google Scholar]
- Hesse J, Gross T (2014) Self-organized criticality as a fundamental property of neural systems. Front Syst Neurosci 8:1–14. 10.3389/fnsys.2014.00166 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang C, He Y, Huang L, Zhaohui Y (2007) Hopf bifurcation analysis of two neurons with three delays. Nonlinear Anal Real World Appl 8:903–921. 10.1016/j.nonrwa.2006.03.014 [DOI] [Google Scholar]
- Hudspeth AJ (2008) Making an effort to listen: mechanical amplification in the ear. Neuron 59:530–545. 10.1016/j.neuron.2008.07.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hudspeth AJ (2014) Integrating the active process of hair cells with cochlear function. Nat Rev Neurosci 15:600–614. 10.1038/nrn3786 [DOI] [PubMed] [Google Scholar]
- Hudspeth AJ, Corey DP (1977) Sensitivity, polarity, and conductance change in the response of vertebrate hair cells to controlled mechanical stimuli. Proc Natl Acad Sci U S A 74:2407–2411. 10.1073/pnas.74.6.2407 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hudspeth AJ, Jülicher F, Martin P (2010) A critique of the critical cochlea: Hopf–a bifurcation–is better than none. J Neurophysiol 104:1219–1229. 10.1152/jn.00437.2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jülicher F, Andor D, Duke T (2001) Physical basis of two-tone interference in hearing. Proc Natl Acad Sci U S A 98:9080–9085. 10.1073/pnas.151257898 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kardar M (2007) Statistical physics of fields. Cambridge, New York: Cambridge University Press. [Google Scholar]
- Kemp DT (1978) Stimulated acoustic emissions from within the human auditory system. J Acoust Soc Am 64:1386–1391. 10.1121/1.382104 [DOI] [PubMed] [Google Scholar]
- Killion MC (1978) Revised estimate of minimum audible pressure: where is the “missing 6 dB”? J Acoust Soc Am 63:1501–1508. 10.1121/1.381844 [DOI] [PubMed] [Google Scholar]
- Korobeinikov A, Mcnabb A (2001) Long-term global climate dynamics: a Hopf bifurcation causingrecurrent ice ages. J Appl Math Decis Sci 5:201–214. 10.1207/S15327612JAMD0504_01 [DOI] [Google Scholar]
- Kozlov AS, Risler T, Hudspeth AJ (2007) Coherent motion of stereocilia assures the concerted gating of hair-cell transduction channels. Nat Neurosci 10:87–92. 10.1038/nn1818 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kubo R (1966) The fluctuation-dissipation theorem. Rep Prog Phys 29:255–284. 10.1088/0034-4885/29/1/306 [DOI] [Google Scholar]
- Lin C-HJ, Bozovic D (2020) Effects of efferent activity on hair bundle mechanics. J Neurosci 40:2390–2402. 10.1523/JNEUROSCI.1312-19.2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma SM (2021) Hopf bifurcation of a gene-protein network module with reaction diffusion and delay effects. Int J Mod Nonlinear Theory Appl 10:91–105. 10.4236/ijmnta.2021.103007 [DOI] [Google Scholar]
- Manley GA (2000) Cochlear mechanisms from a phylogenetic viewpoint. Proc Natl Acad Sci U S A 97:11736–11743. 10.1073/pnas.97.22.11736 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manley GA (2001) Evidence for an active process and a cochlear amplifier in nonmammals. J Neurophysiol 86:541–549. 10.1152/jn.2001.86.2.541 [DOI] [PubMed] [Google Scholar]
- Manley GA, Köppl C (1998) Phylogenetic development of the cochlea and its innervation. Curr Opin Neurobiol 8:468–474. 10.1016/S0959-4388(98)80033-0 [DOI] [PubMed] [Google Scholar]
- Martin P (2008) Active hair-bundle motility of the hair cells of vestibular and auditory organs. In: Active processes and otoacoustic emissions in hearing. Springer handbook of auditory research (Manley GA, Popper AN, Fay RR, Popper AN, eds), pp 93–143. New York: Springer. [Google Scholar]
- Martin P, Bozovic D, Choe Y, Hudspeth AJ (2003) Spontaneous oscillation by hair bundles of the bullfrog’s sacculus. J Neurosci 23:4533–4548. 10.1523/JNEUROSCI.23-11-04533.2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin P, Hudspeth AJ (1999) Active hair-bundle movements can amplify a hair cell’s response to oscillatory mechanical stimuli. Proc Natl Acad Sci U S A 96:14306–14311. 10.1073/pnas.96.25.14306 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin P, Hudspeth AJ (2001) Compressive nonlinearity in the hair bundle’s active response to mechanical stimulation. Proc Natl Acad Sci U S A 98:14386–14391. 10.1073/pnas.251530498 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin P, Hudspeth AJ (2021) Mechanical frequency tuning by sensory hair cells, the receptors and amplifiers of the inner ear. Annu Rev Condens Matter Phys 12:29–49. 10.1146/annurev-conmatphys-061020-053041 [DOI] [Google Scholar]
- Martin P, Hudspeth AJ, Jülicher F (2001) Comparison of a hair bundle’s spontaneous oscillations with its response to mechanical stimulation reveals the underlying active process. Proc Natl Acad Sci U S A 98:14380–14385. 10.1073/pnas.251530598 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meenderink SW, Quinones PM, Bozovic D (2015) Voltage-mediated control of spontaneous bundle oscillations in saccular hair cells. J Neurosci 35:14457–14466. 10.1523/JNEUROSCI.1451-15.2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mhatre N, Robert D (2013) A tympanal insect ear exploits a critical oscillator for active amplification and tuning. Curr Biol 23:1952–1957. 10.1016/j.cub.2013.08.028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moore BC (1973) Frequency difference limens for short-duration tones. J Acoust Soc Am 54:610–619. 10.1121/1.1913640 [DOI] [PubMed] [Google Scholar]
- Mora T, Bialek W (2011) Are biological systems poised at criticality? J Stat Phys 144:268–302. 10.1007/s10955-011-0229-4 [DOI] [Google Scholar]
- Nadrowski B, Albert JT, Gopfert MC (2008) Transducer-based force generation explains active process in Drosophila hearing. Curr Biol 18:1365–1372. 10.1016/j.cub.2008.07.095 [DOI] [PubMed] [Google Scholar]
- Nadrowski B, Martin P, Jülicher F (2004) Active hair-bundle motility harnesses noise to operate near an optimum of mechanosensitivity. Proc Natl Acad Sci U S A 101:12195–12200. 10.1073/pnas.0403020101 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oberreiter L, Seifert U, Barato AC (2022) Universal minimal cost of coherent biochemical oscillations. Phys Rev E 106:014106. 10.1103/PhysRevE.106.014106 [DOI] [PubMed] [Google Scholar]
- Pan B, et al. (2018) TMC1 forms the pore of mechanosensory transduction channels in vertebrate inner ear hair cells. Neuron 99:736–753.e6. 10.1016/j.neuron.2018.07.033 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Penner MJ, Zhang T (1997) Prevalence of spontaneous otoacoustic emissions in adults revisited. Hear Res 103:28–34. 10.1016/S0378-5955(96)00162-1 [DOI] [PubMed] [Google Scholar]
- Popov IY, Fedorov EG (2023) Hopf bifurcations in a network of FitzHugh–Nagumo biological neurons. Int J Nonlinear Sci Numer Simul 24:847–866. 10.1515/ijnsns-2021-0188 [DOI] [Google Scholar]
- Qiu X, Müller U (2022) Sensing sound: cellular specializations and molecular force sensors. Neuron 110:3667–3687. 10.1016/j.neuron.2022.09.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reichenbach T, Hudspeth AJ (2014) The physics of hearing: fluid mechanics and the active process of the inner ear. Rep Prog Phys 77:076601. 10.1088/0034-4885/77/7/076601 [DOI] [PubMed] [Google Scholar]
- Ren T (2002) Longitudinal pattern of basilar membrane vibration in the sensitive cochlea. Proc Natl Acad Sci U S A 99:17101–17106. 10.1073/pnas.262663699 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Retzius G (1884) Das Gehörorgan der Wirbelthiere. Stockholm: Samson & Wallin. [Google Scholar]
- Robles L, Ruggero MA (2001) Mechanics of the mammalian cochlea. Physiol Rev 81:1305–1352. 10.1152/physrev.2001.81.3.1305 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roldán É, Barral J, Martin P, Parrondo JMR, Jülicher F (2021) Quantifying entropy production in active fluctuations of the hair-cell bundle from time irreversibility and uncertainty relations. New J Phys 23:083013. 10.1088/1367-2630/ac0f18 [DOI] [Google Scholar]
- Ruggero MA, Rich NC, Recio A, Narayan SS, Robles L (1997) Basilar-membrane responses to tones at the base of the chinchilla cochlea. J Acoust Soc Am 101:2151–2163. 10.1121/1.418265 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saikia D, Bora MP (2020) Nonlinear model of the firefly flash. Nonlinear Dyn 101:1301–1315. 10.1007/s11071-020-05830-0 [DOI] [Google Scholar]
- Salvi JD, Ó Maoiléidigh D, Fabella BA, Tobin M, Hudspeth AJ (2015) Control of a hair bundle’s mechanosensory function by its mechanical load. Proc Natl Acad Sci U S A 112:E1000–E1009. 10.1073/pnas.1501453112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salvi JD, Ó Maoiléidigh D, Hudspeth AJ (2016) Identification of bifurcations from observations of noisy biological oscillators. Biophys J 111:798–812. 10.1016/j.bpj.2016.07.027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shera CA (2007) Laser amplification with a twist: traveling-wave propagation and gain functions from throughout the cochlea. J Acoust Soc Am 122:22. 10.1121/1.2783205 [DOI] [PubMed] [Google Scholar]
- Sivian LJ, White SD (1933) On minimum audible sound fields. J Acoust Soc Am 4:288–321. 10.1121/1.1915608 [DOI] [Google Scholar]
- Stollenwerk N, Sommer PF, Kooi B, Mateus L, Ghaffari P, Aguiar M (2017) Hopf and torus bifurcations, torus destruction and chaos in population biology. Ecol Complex 30:91–99. 10.1016/j.ecocom.2016.12.009 [DOI] [Google Scholar]
- Talmadge CL, Long GR, Murphy WJ, Tubis A (1993) New off-line method for detecting spontaneous otoacoustic emissions in human subjects. Hear Res 71:170–182. 10.1016/0378-5955(93)90032-V [DOI] [PubMed] [Google Scholar]
- Tinevez JY, Julicher F, Martin P (2007) Unifying the various incarnations of active hair-bundle motility by the vertebrate hair cell. Biophys J 93:4053–4067. 10.1529/biophysj.107.108498 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toth DJA (2008) Bifurcation structure of a chemostat model for an age-structured predator and its prey. J Biol Dyn 2:428–448. 10.1080/17513750802360853 [DOI] [PubMed] [Google Scholar]
- Tucci G, Roldán É, Gambassi A, Belousov R, Berger F, Alonso RG, Hudspeth AJ (2022) Modeling active non-Markovian oscillations. Phys Rev Lett 129:030603. 10.1103/PhysRevLett.129.030603 [DOI] [PubMed] [Google Scholar]
- van der Heijden M, Vavakou A (2021) Rectifying and sluggish: outer hair cells as regulators rather than amplifiers. Hear Res 423:108367. 10.1016/j.heares.2021.108367 [DOI] [PubMed] [Google Scholar]
- Verdugo A (2018) Hopf bifurcation analysis of the repressilator model. Am J Comput Math 8:137–152. 10.4236/ajcm.2018.82011 [DOI] [Google Scholar]
- von Békésy G (1960) Experiments in hearing. New York: McGraw-Hill Book Company. [Google Scholar]
- Yuan C, Wang J (2016) Hopf bifurcation analysis and control of three-dimensional Prescott neuron model. J Vibroeng 18:4105–4115. 10.21595/jve.2016.16933 [DOI] [Google Scholar]
- Zweig G (1991) Finding the impedance of the organ of Corti. J Acoust Soc Am 89:1229–1254. 10.1121/1.400653 [DOI] [PubMed] [Google Scholar]