Abstract
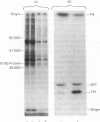
1. Effects of corticotropin-(1--24)-tetracosapeptide on the endogenous phosphorylation of proteins and lipids were studied in a membrane/cytosol fraction prepared from a lysed crude mitochondrial/synaptosomal fraction. 2. The labelling of proteins and lipids was monitored by incubation of the subcellular fraction for 10s with [gamma-32P]ATP. 3. The phosphorylation of proteins was dose-dependently inhibited by the peptide (40% of control incubations at 100 microM-corticotropin). 4. Of the membrane phospholipids only phosphatidylinositol phosphate, phosphatidylinositol bisphosphate and phosphatidic acid became labelled. Corticotropin dose-dependently increased the formation of phosphatidylinositol bisphosphate and inhibited the production of phosphatidic acid (470% and 50% respectively of control incubations, at 100 microM of the peptide) and had no effect on phosphatidylinositol phosphate. 5. Phosphatase activity was observed to act on phosphatidylinositol bisphosphate, phosphatidylinositol phosphate and phosphoprotein but not on phosphatidic acid. 6. Corticotropin interacted with the kinases rather than with the phosphatases. 7. The formation of phosphatidylinositol bisphosphate and phosphatidic acid was maximal at 1--10mM-Mg2+ in the absence of Ca2+, and the production of phosphatidylinositol phosphate was maximal at 30mM-Mg2+. 8. The basal value of lipid phosphorylation decreased with increasing Ca2+ concentration. 9. Ca2+ abolished the effect of corticotropin on phosphatidylinositol bisphosphate formation (470%, 190% and 100% of control incubations at respectively 0, 0.1 and 1 mM-Ca2+). 10. The data provide evidence that the effects of corticotropin on protein phosphorylation and on polyphosphoinositide metabolism in brain membranes are related.
Full text
PDF








Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abdel-Latif A. A., Akhtar R. A., Hawthorne J. N. Acetylcholine increases the breakdown of triphosphoinositide of rabbit iris muscle prelabelled with [32P] phosphate. Biochem J. 1977 Jan 15;162(1):61–73. doi: 10.1042/bj1620061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Abdel-Latif A. A., Green K., Smith J. P. Sympathetic denervation and the triphosphoinositide effect in the iris smooth muscle: a biochemical method for the determination of alpha-adrenergic receptor denervation supersensitivity. J Neurochem. 1979 Jan;32(1):225–228. doi: 10.1111/j.1471-4159.1979.tb04532.x. [DOI] [PubMed] [Google Scholar]
- Akhtar R. A., Abdel-Latif A. A. Studies on the properties of triphosphoinositide phosphomonoesterase and phosphodiesterase of rabbit iris smooth muscle. Biochim Biophys Acta. 1978 Nov 10;527(1):159–170. doi: 10.1016/0005-2744(78)90265-6. [DOI] [PubMed] [Google Scholar]
- Booth R. F., Clark J. B. A rapid method for the preparation of relatively pure metabolically competent synaptosomes from rat brain. Biochem J. 1978 Nov 15;176(2):365–370. doi: 10.1042/bj1760365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dawson R. M. 'Phosphatido-peptide'-like complexes formed by the interaction of calcium triphosphoinositide with protein. Biochem J. 1965 Oct;97(1):134–138. doi: 10.1042/bj0970134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dawson R. M., Eichberg J. Diphosphoinositide and triphosphoinositide in animal tissues. Extraction, estimation and changes post mortem. Biochem J. 1965 Sep;96(3):634–643. doi: 10.1042/bj0960634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farese R. V., Sabir A. M., Vandor S. L. Adrenocorticotropin acutely increases adrenal polyphosphoinositides. J Biol Chem. 1979 Aug 10;254(15):6842–6844. [PubMed] [Google Scholar]
- GRAY E. G., WHITTAKER V. P. The isolation of nerve endings from brain: an electron-microscopic study of cell fragments derived by homogenization and centrifugation. J Anat. 1962 Jan;96:79–88. [PMC free article] [PubMed] [Google Scholar]
- Gispen W. H., Schotman P., de Kloet E. R. Brain RNA and hypophysectomy; a topographical study. Neuroendocrinology. 1972;9(5):285–296. doi: 10.1159/000122060. [DOI] [PubMed] [Google Scholar]
- Gispen W. H., van Ree J. M., de Wied D. Lipotropin and the central nervous system. Int Rev Neurobiol. 1977;20:209–250. doi: 10.1016/s0074-7742(08)60654-6. [DOI] [PubMed] [Google Scholar]
- Greengard P. Phosphorylated proteins as physiological effectors. Science. 1978 Jan 13;199(4325):146–152. doi: 10.1126/science.22932. [DOI] [PubMed] [Google Scholar]
- Griffin H. D., Hawthorne J. N. Calcium-activated hydrolysis of phosphatidyl-myo-inositol 4-phosphate and phosphatidyl-myo-inositol 4,5-bisphosphate in guinea-pig synaptosomes. Biochem J. 1978 Nov 15;176(2):541–552. doi: 10.1042/bj1760541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hawthorne J. N., Pickard M. R. Phospholipids in synaptic function. J Neurochem. 1979 Jan;32(1):5–14. doi: 10.1111/j.1471-4159.1979.tb04503.x. [DOI] [PubMed] [Google Scholar]
- Hess H. H., Derr J. E. Assay of inorganic and organic phosphorus in the 0.1-5 nanomole range. Anal Biochem. 1975 Feb;63(2):607–613. doi: 10.1016/0003-2697(75)90388-7. [DOI] [PubMed] [Google Scholar]
- Irvine R. F., Hemington N., Dawson R. M. The calcium-dependent phosphatidylinositol-phosphodiesterase of rat brain. Mechanisms of suppression and stimulation. Eur J Biochem. 1979 Sep;99(3):525–530. doi: 10.1111/j.1432-1033.1979.tb13284.x. [DOI] [PubMed] [Google Scholar]
- Jolles J., Wirtz K. W., Schotman P., Gispen W. H. Pituitary hormones influence polyphosphoinositide metabolism in rat brain. FEBS Lett. 1979 Sep 1;105(1):110–114. doi: 10.1016/0014-5793(79)80897-2. [DOI] [PubMed] [Google Scholar]
- Jolles J., Zwiers H., van Dongen C. J., Schotman P., Wirtz K. W., Gispen W. H. Modulation of brain polyphosphoinositide metabolism by ACTH-sensitive protein phosphorylation. Nature. 1980 Aug 7;286(5773):623–625. doi: 10.1038/286623a0. [DOI] [PubMed] [Google Scholar]
- Kai M., Salway J. G., Hawthorne J. N. The diphosphoinositide kinase of rat brain. Biochem J. 1968 Feb;106(4):791–801. doi: 10.1042/bj1060791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kai M., White G. L., Hawthorne J. N. The phosphatidylinositol kinase of rat brain. Biochem J. 1966 Nov;101(2):328–337. doi: 10.1042/bj1010328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanoh H., Akesson B. Properties of microsomal and soluble diacylglycerol kinase in rat liver. Eur J Biochem. 1978 Apr;85(1):225–232. doi: 10.1111/j.1432-1033.1978.tb12230.x. [DOI] [PubMed] [Google Scholar]
- Krebs E. G., Beavo J. A. Phosphorylation-dephosphorylation of enzymes. Annu Rev Biochem. 1979;48:923–959. doi: 10.1146/annurev.bi.48.070179.004423. [DOI] [PubMed] [Google Scholar]
- Lugtenberg B., Meijers J., Peters R., van der Hoek P., van Alphen L. Electrophoretic resolution of the "major outer membrane protein" of Escherichia coli K12 into four bands. FEBS Lett. 1975 Oct 15;58(1):254–258. doi: 10.1016/0014-5793(75)80272-9. [DOI] [PubMed] [Google Scholar]
- Matthews E. K., Saffran M. Ionic dependence of adrenal steroidogenesis and ACTH-induced changes in the membrane potential of adrenocortical cells. J Physiol. 1973 Oct;234(1):43–64. doi: 10.1113/jphysiol.1973.sp010333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Michell R. H. Inositol phospholipids and cell surface receptor function. Biochim Biophys Acta. 1975 Mar 25;415(1):81–47. doi: 10.1016/0304-4157(75)90017-9. [DOI] [PubMed] [Google Scholar]
- Palmer F. B., Dawson R. M. Complex-formation between triphosphoinositide and experimental allergic encephalitogenic protein. Biochem J. 1969 Mar;111(5):637–646. [PMC free article] [PubMed] [Google Scholar]
- Podesta E. J., Milani A., Steffen H., Neher R. Adrenocorticotropin (ACTH) induces phosphorylation of a cytoplasmic protein in intact isolated adrenocortical cells. Proc Natl Acad Sci U S A. 1979 Oct;76(10):5187–5191. doi: 10.1073/pnas.76.10.5187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Routtenberg A., Ehrlich Y. H. Endogenous phosphorylation of four cerebral cortical membrane proteins: role of cyclic nucleotides, ATP and divalent cations. Brain Res. 1975 Jul 18;92(3):415–430. doi: 10.1016/0006-8993(75)90326-1. [DOI] [PubMed] [Google Scholar]
- Shaikh N. A., Palmer F. B. Phosphoinositide kinases in chick brain and sciatic nerve, a developmental study. J Neurochem. 1977 Feb;28(2):395–402. doi: 10.1111/j.1471-4159.1977.tb07760.x. [DOI] [PubMed] [Google Scholar]
- Takai Y., Kishimoto A., Iwasa Y., Kawahara Y., Mori T., Nishizuka Y. Calcium-dependent activation of a multifunctional protein kinase by membrane phospholipids. J Biol Chem. 1979 May 25;254(10):3692–3695. [PubMed] [Google Scholar]
- Takai Y., Kishimoto A., Kikkawa U., Mori T., Nishizuka Y. Unsaturated diacylglycerol as a possible messenger for the activation of calcium-activated, phospholipid-dependent protein kinase system. Biochem Biophys Res Commun. 1979 Dec 28;91(4):1218–1224. doi: 10.1016/0006-291x(79)91197-5. [DOI] [PubMed] [Google Scholar]
- Torda C. Model of molecular mechanism able to generate a depolarization-hyperpolarization cycle. Int Rev Neurobiol. 1974;16(0):1–66. doi: 10.1016/s0074-7742(08)60194-4. [DOI] [PubMed] [Google Scholar]
- Walter U., Uno I., Liu A. Y., Greengard P. Study of autophosphorylation of isoenzymes of cyclic AMP-dependent protein kinases. J Biol Chem. 1977 Oct 10;252(19):6588–6590. [PubMed] [Google Scholar]
- Wiegant V. M., Zwiers H., Schotman P., Gispen W. H. Endogenous phosphorylation of rat brain synaptosomal plasma membranes in vitro: some methodological aspects. Neurochem Res. 1978 Aug;3(4):443–453. doi: 10.1007/BF00966326. [DOI] [PubMed] [Google Scholar]
- Wouters W., Van Den Bercken J. Effects of ACTH4-10 on synaptic transmission in frog sympathetic ganglion. Eur J Pharmacol. 1979 Aug 15;57(4):353–363. doi: 10.1016/0014-2999(79)90498-9. [DOI] [PubMed] [Google Scholar]
- Zwiers H., Schotman P., Gispen W. H. Purification and some characteristics of an ACTH-sensitive protein kinase and its substrate protein in rat brain membranes. J Neurochem. 1980 Jun;34(6):1689–1699. doi: 10.1111/j.1471-4159.1980.tb11262.x. [DOI] [PubMed] [Google Scholar]
- Zwiers H., Tonnaer J., Wiegant V. M., Schotman P., Gispen W. H. ACTH-sensitive protein kinase from rat brain membranes. J Neurochem. 1979 Jul;33(1):247–256. doi: 10.1111/j.1471-4159.1979.tb11726.x. [DOI] [PubMed] [Google Scholar]
- Zwiers H., Wiegant V. M., Schotman P., Gispen W. H. ACTH-induced inhibition of endogenous rat brain protein phosphorylation in vitro: structure activity. Neurochem Res. 1978 Aug;3(4):455–463. doi: 10.1007/BF00966327. [DOI] [PubMed] [Google Scholar]