Summary
Background
Li-Fraumeni Syndrome (LFS) is a predisposition associated with early onset malignant tumors caused by germline pathogenic variants in the TP53 gene. Although rare worldwide, LFS is prevalent in Southern Brazil due to the founder pathogenic variant R337H. Here, we assessed tumor patterns and temporal trends, cancer risk, and sex differences of adult R337H carriers and carriers of other LFS-associated variants.
Methods
We retrospectively analyzed 708 adults, combining data from two sources: the Brazilian Li-Fraumeni Syndrome Study cohort and the NCI TP53 database. We assessed the clinical characteristics of 303 adults with R337H and compared them with those associated with 405 carriers of other TP53 variants.
Findings
R337H carriers, compared to adult carriers of other TP53 variants typical of LFS, had a lower cumulative risk of developing cancer (54% vs 78%). Female R337H carriers were at a higher risk than males (65% vs 30%) and had a higher risk of developing a second primary cancer, underscoring a strong sex bias not observed in carriers of other variants. The most common cancers were breast cancer and soft tissue sarcoma in females, and soft tissue sarcoma and prostate cancer in males. Common second malignancies were breast cancer in females and lung cancer in males.
Interpretation
This study shows that R337H is associated with a lifetime risk of multiple LFS-spectrum cancers but with incomplete penetrance, particularly in males. Our findings suggest that R337H carriers would benefit from tailored surveillance and risk reduction strategies.
Funding
São Paulo Research Foundation, Conselho Nacional de Pesquisa, and Hospital Sírio-Libanês.
Keywords: Li-Fraumeni syndrome, R337H variant, TP53, Germline
Research in context.
Evidence before this study
Since 2001, the TP53 variant R337H (c.1010G>A; p.Arg337His) has frequently been reported in the germline of children with adrenocortical carcinoma and adults in Southern Brazil, with an estimated prevalence of 0.3% in the local population. We conducted a systematic search of PubMed and Google Scholar databases from January 2001 to December 2023, using the search terms “p.Arg337His”, “R337H”, “TP53 R337H”, and “R337H adult”. Studies were included if they reported on TP53 R337H carriers and cancer outcomes, with no language restrictions. Our literature review identified approximately 140 published studies (PubMed). Most studies focused on pediatric adrenocortical carcinoma or small case series. Assessment of the existing evidence revealed a significant knowledge gap regarding comprehensive tumor spectrum and cancer risk assessment in adult R337H carriers, as previous studies were limited by small cohorts, pediatric focus, or lack of long-term follow-up data in adult populations.
Added value of this study
This study presents the Brazilian Li-Fraumeni Syndrome Study (BLiSS), which comprises the largest cohort of adult TP53 R337H carriers ever assembled, including 303 carriers from 160 families. While previous studies have debated whether R337H carriers exhibit full or attenuated Li-Fraumeni Syndrome (LFS) features, our comprehensive analysis provides a complete systematic assessment of cancer manifestations in this population. Through detailed investigation of the BLiSS cohort, we establish several key findings: (1) a comprehensive tumor spectrum and lifetime cancer risk assessment specific to adult R337H carriers; (2) temporal patterns of tumor development that distinguish R337H from other pathogenic TP53 variants; (3) significant sex-specific differences in cancer risk, with female carriers showing notably higher cancer incidence, particularly for early-onset breast cancer; and (4) distinct age-of-onset patterns that differentiate R337H from other LFS-associated variants. These findings demonstrate that while R337H carriers face substantial cancer risks within the LFS spectrum, their risk pattern represents a distinct subset of the classic LFS phenotype.
Implications of all the available evidence
The unique tumor spectrum and risk patterns identified in adult R337H carriers demonstrate the need for tailored cancer surveillance strategies distinct from standard LFS protocols. Given the high prevalence of R337H in the Brazilian population (0.3%), our findings may have clinical and public health implications. For healthcare professionals and genetic counselors, these data may help the development of R337H-specific screening guidelines, particularly emphasizing earlier and more intensive breast cancer surveillance in female carriers. The sex-specific differences in cancer risk and age of onset should inform personalized risk assessment and prevention strategies. From a public health perspective, our findings may help to justify population-specific screening policies in regions with high R337H prevalence. Future research should focus on identifying the molecular and environmental factors that contribute to the variant's variable penetrance and distinct tumor spectrum. Additionally, prospective studies are needed to evaluate the effectiveness of targeted screening protocols in reducing cancer-related mortality in this population.
Introduction
TP53 is a multi-functional gene commonly mutated in human tumors, with more than 50% of cancers carrying loss-of-function somatic mutations in TP53.1,2 De novo germline pathogenic (P) or likely pathogenic (LP) TP53 variants are thought to occur at a frequency of about 1:5000 births3 and carriers are associated with a lifetime predisposition of developing multiple primary tumors known as Li-Fraumeni Syndrome (LFS).4,5 The LFS cancer spectrum is characterized by early-life tumors including adrenocortical carcinoma, premenopausal breast cancer, bone and soft tissue sarcomas, central nervous system (CNS) cancers, and hematopoietic malignancies, among others.6
In 2001, Ribeiro and colleagues reported that an atypical TP53 variant, c.1010G>A; p.Arg337His (R337H), was frequently found in the germline of children with adrenocortical carcinoma in Southern Brazil.7 In subsequent years, a high number of children and adult carriers of this variant were detected in the same area, presenting complete or partial LFS traits.8 R337H differs from most TP53 P/LP variants by its location in exon 10, encoding the oligomerization domain of the p53 protein, whereas the majority of variants causing LFS occurs in exons 4–9 (encoding the DNA-binding domain). R337H variant is carried by a founder haplotype of Lusitanian/Hispanic origin, estimated to be present in about 0.3% of the population of Southern Brazil.8, 9, 10, 11, 12 Over the past 15 years, the magnitude of cancer risk caused by R337H has been a matter of debate, some carriers presenting with full LFS traits and many others with attenuated LFS features or remaining apparently cancer-free over lifetime. Given its high prevalence, R337H confers a significant cancer risk in the Brazilian population. Therefore, detailed information on lifetime cancer incidence, tumor spectrum and incidence of multiple primary tumors is crucial for developing awareness and appropriate screening and risk reduction strategies.13 Of note, with the development of genome sequencing as a basis for molecular diagnosis, R337H represents a frequent finding in adults, but current knowledge of tumor profiles in R337H adult carriers is scarce14,15 and limited to studies with few samples or individuals.12,16,17
Here, we report on the Brazilian Li-Fraumeni Syndrome Study (BLiSS), the largest cohort of Brazilian LFS adults available to date. We provide a detailed assessment of tumor spectrum, lifetime cancer risk, temporal tumor trends and sex differences associated with R337H and we compare them with those associated with other P/LP variants found in Brazilians or in carriers from other origins. We show that R337H entails a significant lifetime risk of multiple cancers, which recapitulates many, but not all traits of LFS, with striking differences between males and females. Overall, these results suggest that tailored screening and risk reduction strategies could be developed and adapted to the needs of R337H carriers.
Methods
Brazilian Li-Fraumeni syndrome study (BLiSS) cohort
We retrospectively investigated tumor spectrum, clinical and demographic status in a cohort of 708 adults with germline TP53 P/LP variants. P/LP variant status was based on ClinGen TP53 Expert Panel Specifications to the ACMG/AMP Variant Interpretation Guidelines for TP53 (Version 1.3.0, https://cspec.genome.network/cspec/ui/svi/doc/GN009). The cohort was constituted from two datasets: (1) BLiSS, comprising 345 Brazilian adult heterozygous carriers (age range 18–82 years, median 42 years), including 303 carriers (88%, 160 families) of TP53 c.1010G>A (R337H) and 42 carriers (12%, 29 families) of other TP53 P/LP variants and (2) 363 adult carriers of other TP53 P/LP variants (age range 18–90, median 38 years) from diverse geographic areas, extracted from the NCI TP53 database (version R20; https://tp53.isb-cgc.org/), a resource that compiles annotations on TP53 carriers from the literature (Supplementary Table S1). For the purposes of this study, carriers of P/LP TP53 variants other than R337H will be referred to as ‘non-R337H carriers’. In detail, the BLISS cohort comprises 345 adult individuals with germline TP53 pathogenic/likely pathogenic (P/LP) variants, of whom 303 carry the R337H variant and 42 have non-R337H variants. Among the R337H carriers, 155 were index cases, with 110 of these tested due to a cancer diagnosis and 45 identified through cascade testing of family members, being presymptomatic at the time of testing. Of the 42 non-R337H carriers in the BLISS cohort, 26 were index cases, with 24 tested due to a cancer diagnosis and 2 identified pre-symptomatically. Regarding the 363 non-R337H carriers from the TP53 database cohort, index case information was available for 215 individuals, of whom 21 were confirmed as index cases. However, information about the timing of testing was not available for this group. This breakdown helps account for potential testing bias by distinguishing between individuals tested due to cancer diagnosis and those identified pre-symptomatically (Supplementary Table S2).
To differentiate between second primary breast tumors and relapsed breast cancers, we applied the following criteria: (1) Any new breast cancer in the opposite breast was classified as a second primary tumor and (2) For cancers occurring in the same breast as the initial tumor, we considered it a second primary tumor if one or more of the following conditions were met: (a) The tumor showed a different invasiveness profile (e.g., ductal carcinoma in situ followed by invasive ductal carcinoma, or vice versa); (b) The new tumor was located in a different quadrant of the breast; and (c) The immunohistochemistry (IHC) subtype differed from the initial tumor, specifically considering changes in estrogen receptor (ER) and HER2 status. Any ipsilateral breast cancer that did not meet these criteria was classified as a relapsed or recurrent cancer and was not included in our analysis of second primary tumors. Additionally, detailed histological subtypes of CNS cancers, soft tissue sarcomas, and bone tumors are provided in Supplementary Figure S1. This classification is based on the histopathological diagnoses recorded in the BLISS cohort and the TP53 database.
Individuals recruited into BLISS were detected between 2018 and 2022 in a high familial cancer risk clinic at Hospital Sírio-Libanês (Sao Paulo, Brazil). All subjects tested positive for a germline P/LP TP53 variant either by conventional targeted gene sequencing or by multigene next generation sequencing panels. All clinical and personal data were de-identified before data sharing and analyses. Participants in the BLISS cohort were not recruited through population screening for R337H carriers. Patients were identified through cancer screening guidelines, by meeting clinical criteria for LFS, or through cascade testing of family members. This ascertainment method was consistent for both R337H and non-R337H carriers from our cohort. Importantly, no population-based screening of unaffected individuals was conducted to include additional patients in our cohort. This study was approved by the institutional research ethics committees in all centers (approval at the coordinating center, Hospital Sírio-Libanês #3.830.276). All patients signed a written informed consent. Data from the NCI TP53 dataset are fully anonymous and open-source. Finally, it is important to note that our study specifically focused on characterizing the tumor profile of Li-Fraumeni Syndrome (LFS) individuals in adulthood. Consequently, our inclusion criteria deliberately excluded individuals with reported pediatric-onset tumors.
Statistical analyses
Data processing, statistical analyses, and figures were produced in the R software (version 4.0.3), using the following R packages: ggplot2 (v3.3.6), ggpubr (v0.4.0), survival (v3.2.13), survminer (v0.4.9), tidyverse (v1.3.1), trackViewer (v1.24.2), and webr (v0.1.5). Figure colors and esthetic edition were performed using the Inkscape software (https://inkscape.org/). Sankey diagrams were created using the SankeyMATIC web tool (https://sankeymatic.com/). Statistical comparisons between patient groups were performed using Wilcoxon rank sum test and significance of Kaplan Meier (K-M) curves were assessed by log-rank tests. All statistical tests were considered significant at p-value <0.05, and, if applicable, adjusted to multiple test comparisons using false discovery rate.
Role of funding source
The funder of the study had no role in study design, data collection, data analysis, data interpretation, or writing of the article.
Results
Study cohort
The goal of this study was to rigorously compare cancer phenotypes and lifetime risk in Brazilian adult R337H carriers with those of adult carriers of other P/LP TP53 variants (non-R337H), either from Brazil or from other regions of the world. R337H carriers were identified within the BLiSS study and the comparison group was constructed by lumping non-R337H carriers from BLiSS with a subset of carriers with similar age range and median age as BLISS carriers, taken from the public NCI TP53 dataset (see Methods). Overall, we compiled data on a total of 708 adults with P/LP variants, including 345 subjects from BLiSS registry and 363 from the NCI TP53 database (https://tp53.isb-cgc.org/), Fig. 1A. Of these 708 carriers, 303 (160 families) carried the founder R337H variant and 405 carried non-R337H P/LP variants (172 families), including 42 Brazilian carriers (29 families) (Fig. 1A). Fig. 1B shows the occurrence and position of these variants with respect to p53 domains. Aside from R337H (303 occurrences), which is located in the oligomerization domain, a total of 46 different other variants were identified, mostly located within the DNA-binding domain. These variants included the missense variants M133T (c.398T>C, n = 22 carriers), R175H (c.524G>A, n = 21), R248W (c.742C>T, n = 19), G245S (c.733G>A, n = 18), R213Q (c.638G>A, n = 16), and R273H (c.818G>A, n = 15), as well as the silent variant T125 = (c.375G>A, n = 19), which has been shown to inactivate p53 by disrupting gene splicing and preventing p53 mRNA synthesis.18 This dataset represents the largest cohort to date of adult carriers of P/LP TP53 variants, including R337H carriers.
Fig. 1.
Overview of R337H and non-R337H study cohorts. A) Study flowchart showing the selected R337H (n = 303) and non-R337H (n = 405) individuals. B) Frequency of TP53 variants included in our study and affected TP53 domains. Variations found in at least 1% of individuals (corresponding to 286 of the 405) are shown. Number of non-R337H carriers only from the BLiSS cohort is shown in parenthesis when appropriate.
Cancer occurrence and patterns in R337H carriers
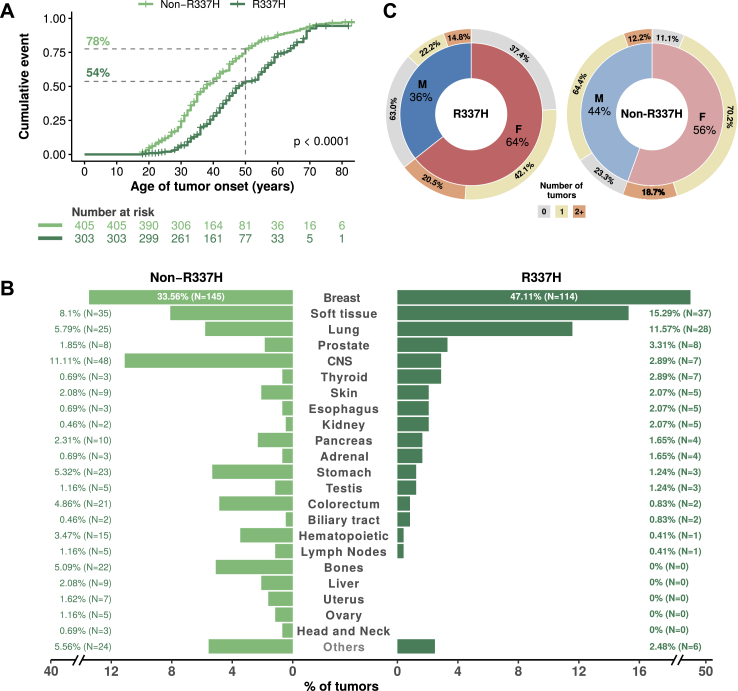
R337H carriers received fewer primary tumor diagnoses compared to non-R337H Li-Fraumeni carriers (log-rank test p-value < 0.0001, Fig. 2A; Supplementary Figure S2). At age 50 years, 54% (164/303) of R337H carriers had at least one cancer diagnosis, compared to 78% (316/405) of non-R337H carriers. Noteworthy, by the age of 65, 80% (242/303) R337H and 91% (369/405) non-R337H carriers had at least one cancer diagnosis, respectively, suggesting that R337H-affected individuals tended to develop cancers at later ages than non-R337H carriers. Among R337H carriers, 242 tumors were diagnosed in 162 carriers, while 141 carriers (46.5%) did not present with a cancer diagnosis. The most common diagnoses were breast cancer (47.1%; 114/242), soft-tissue sarcoma (15.3%; 37/242), and lung cancer (11.6%; 28/242), comprising about 75% of all diagnoses in R337H carriers (Fig. 2B). Among non-R337H carriers, the most frequent diagnoses were breast cancer (33.6%; 145/432), CNS cancer (11.1%; 48/432), and soft tissue sarcoma (8.1%; 35/432), comprising 52.8% of all tumors diagnosed in this group, whereas 67 carriers (16.5%; 67/405) were cancer-free. Overall, 4 cancer types were proportionally more represented in R337H than non-R337H carriers: breast cancer (47.1% (114/242) vs 33.6% (145/432), Fisher's test p-value < 0.001), soft tissue sarcoma (15.3% (37/242) vs 8.1% (35/432), Fisher's test p-value = 0.006), lung cancer (11.6% (28/242) vs 5.8% (25/432), Fisher's test p-value = 0.01), and thyroid cancer (2.9% (7/242) vs 0.7% (3/432), Fisher's test p-value = 0.04). In contrast, CNS cancer (for details of CNS cancers, see Supplementary Figure S1) was more represented in non-R337H carriers (Fisher's test p-value < 0.001), as well as bone sarcoma (Fisher's test p-value < 0.0001), a cancer that represented 5.1% (22/432) of diagnoses in non-R337H carriers but was not observed in R337H carriers (Fig. 2B). Additionally, we performed Kaplan–Meier analyses for the fourth most common cancer types (breast, soft tissue sarcoma, prostate, and lung). We found that non-R337H carriers have a higher cancer risk than R337H carriers for these specific cancer types (Supplementary Figure S3, which includes numbers at risk, censored observations, and log-rank test results), which is consistent with the overall cancer risk difference shown in Fig. 2A.
Fig. 2.
Comparison of cancer onset, tumor types, and patient demographics between R337H and non-R337H carriers. A) Kaplan–Meier curves showing the cumulative risks of R337H and non-R337H carriers of developing a (first) primary tumor; B) Cancer patterns in R337H and non-R337H carriers; C) Proportions of male (M) and female (F) carriers without reported tumors, diagnosed with 1 primary tumor or with 2 or more primary tumors in R337H and non-R337H cohorts.
Overall, these results show that R337H carriers retain a high risk of cancer during adult life, albeit lower than that of non-R337H Li-Fraumeni carriers. At age 50, for example, R337H carriers show a significantly lower cumulative cancer risk compared to non-R337H carriers (see Fig. 2A). However, it's important to note that R337H carriers still exhibit a high frequency of breast cancer and soft tissue sarcoma relative to the general population,19 even though these frequencies are lower than those observed in non-R337H Li-Fraumeni carriers in our cohort.
Differences between R337H male and female carriers
Striking differences between R337H and non-R337H carriers were even evident after stratifying by sex. Among female R337H carriers (median age: 44 years; Fig. 3A), 122/195 (62.6%; Fig. 2C, outer ring in yellow represents 42.1% and orange represents 20.5%) were diagnosed with at least one cancer, while 200/225 (88.9%; Fig. 2C, outer ring in yellow represents 70.2% and orange represents 18.7%) of female non-R337H carriers (median age: 38 years; Fig. 3A) had one or more primary tumors (Fisher's exact test; p-value < 0.0001, Fig. 2C). For males, only 40/108 (37.0%; Fig. 2C, outer ring in yellow represents 22.2% and orange represents 14.8%) of R337H carriers presented a cancer diagnosis while in male non-R337H carriers, 138/180 (76.6%; Fig. 2C, outer ring in yellow represents 64.4% and orange represents 12.2%) had at least one tumor diagnosis (Fisher's exact test; p-value < 0.0001). This difference is remarkable given the median age at follow-up, which is higher in R337H (44 years) than in non-R337H (38 years) (Wilcoxon test p-value < 0.05; Fig. 2C and 3A).
Fig. 3.
Comparison of cancer characteristics between R337H and non-R337H carriers by sex. A) Ages at last follow-up of male and female individuals carrying TP53 R337H or non-R337H variants. B) Ages of male and female individuals at first tumor diagnosis (Wilcoxon tests); C) Kaplan–Meier curves showing the cumulative risks of male and female individuals (R337H and non-R337H carriers) of developing a first tumor (pairwise log-rank tests; FDR adjusted p-values); D) Tumor profile in male and female individuals carrying TP53 R337H or non-R337H variants; E) Temporal patterns of tumor occurrence in male and female individuals (Wilcoxon tests). P-value representation: “∗”: p < 0.05; “∗∗”: p < 0.01; “∗∗∗∗”: p < 0.0001; ns: not significant.
To better characterize these differences, we estimated the cumulative risks of developing a first primary cancer in R337H and non-R337H carriers stratified by sex. Male carriers were significantly older at first diagnosis than females in both R337H (median ages: 46 and 41 years, respectively) and non-R337H groups (median ages: 38 and 35 years, respectively) (Fig. 3B). Among R337H carriers, both males and females showed significantly lower lifetime risks of developing cancer than non-R337H counterparts (Fig. 3C). While among non-R337H carriers, the cumulative cancer risk at 50 years was 82% for females and 72% for males, female R337H carriers had a 2 times greater cumulative risk of developing cancer than males at age 50 years (65% vs 30%, pairwise log-rank tests; FDR adjusted p-values: <0.0001) (Fig. 3C).
Sex-specific differences were also noted in the tumor spectrum (Fig. 3D). Among females, beyond the two most common tumor types—breast cancer (49.2%, 96/195) and soft tissue sarcomas (8.7%, 17/195)—lung cancer (8.2%, 16/195), thyroid (2.6%, 5/195), and skin tumors (2.0%, 4/195) were the most common diagnoses in R337H carriers, but were proportionally rarer in non-R337H (4% (9/225), 1.3% (3/225), and 0.9% (2/225), respectively). Conversely, CNS cancers (9.3%, 21/225) and colorectal cancers (4%, 9/225) were more frequent in non-R337H than in R337H female carriers (1.5% (3/195) and 0.5% (1/195), respectively) (Fisher test p-values 0.0005 and 0.02, respectively). Of note, two cases of male breast cancers were detected among R337H carriers, compared to none in non-R337H carriers.
Next, we analyzed the temporal patterns of specific cancer in both sexes (Fig. 3E). Both breast and lung cancers were diagnosed at later ages in female R337H carriers (median ages: 41 and 56.5 years) in comparison to non-R337H (median ages: 35 and 52 years) (Wilcoxon test p-values <0.001 and <0.05, respectively). When considering male carriers, soft-tissue sarcomas and stomach cancers also showed delayed occurrence in R337H carriers (median ages: 61 and 56 years) compared to non-R337H carriers (median ages: 40 and 35 years) (Wilcoxon test p-values <0.001 and <0.05, respectively). In contrast, prostate tumors tended to occur at similar median ages (>60 years) in R337H and non-R337H male carriers.
These observations show that cancer risk patterns differ between R337H and non-R337H adult carriers. For both sexes, R337H carriers had a lower lifetime risk of cancer, developing at later ages than non-R337H carriers. Furthermore, tumor patterns in R337H, while dominated by diagnoses typical of the LFS spectrum (breast cancer, soft tissue sarcoma) showed significant differences with non-R337H carriers, with a lower proportion of CNS cancers and bone sarcoma and a higher proportion of lung and prostate cancers.
Lifetime risk and trajectories of multiple cancers
We investigated the occurrence of multiple malignancies in R337H carriers among 122 females and 40 males who were affected by a first primary cancer during adulthood. Overall, 40 females (32.8%) and 16 males (40.0%) had more than one tumor diagnosed, respectively (Supplementary Table S1). Both males and females with the R337H variant developed second tumors at later ages than non-R337H carriers (Wilcoxon test p-values: <0.05 and <0.01, respectively), Fig. 4A. Among R337H carriers, females appeared to be twice as likely than males to develop a second tumor by 50 years of age (32% vs 16%; log-rank test, p < 0.01), Fig. 4B. No such statistical differences could be observed among non-R337H carriers. Among R337H carriers, males had second primary malignancies at later ages than females (median ages: 61 and 47 years, respectively; Wilcoxon test p-value < 0.05), Fig. 4A. These second malignancies were diagnosed on average 4 and 6 years after the first tumor in female and male carriers, respectively (Supplementary Figure S4).
Fig. 4.
Lifetime risk of developing a second tumor in R337H and non-R337H males and females. A) Ages of male and female individuals at second tumor diagnosis (Wilcoxon tests); B) Kaplan–Meier curves showing the cumulative risks of male and female individuals (R337H and non-R337H carriers) of developing a second tumor (pairwise log-rank tests; FDR adjusted p-values); C) and D) Proportions of additional tumors diagnosed in R337H and non-R337H C) female and D) male carriers (from second to last diagnosis). P-value representation: “∗”: p < 0.05; “∗∗”: p < 0.01; ns: not significant.
Breast cancer was the most frequent second diagnosis among female carriers of any variant, affecting 50.0% (20/40), and 64.3% (27/42) of R337H and non-R337H carriers, respectively, Fig. 4C. Other frequent tumors included soft-tissue sarcoma (R337H: 22.5% (9/40), non-R337H: 11.9% (5/42)) and lung cancer (R337H: 20.0% (8/40), non-R337H: 11.9% (5/42)). Among male R337H carriers, the spectrum of second malignancies comprised predominantly lung cancer (31.2%, 5/16), soft-tissue sarcoma (25.0%, 4/16), prostate (18.8%, 3/16), and kidney cancer (18.8%, 3/16). In non-R337H male carriers, the most frequent second tumors were CNS cancers (27.3%, 6/22), soft-tissue sarcomas (22.7%, 5/22), and stomach cancers (18.2%, 4/22), Fig. 4D. Notably, among the R337H patients with CNS cancer, we have four patients with glioblastoma (GBM), three of whom have a survival rate of more than 5 years (Supplementary Table S3). This is uncommon, given that the average survival for this tumor is 1.5 years after diagnosis. Taken together, these results indicate that R337H female carriers are at a noticeably higher lifetime risk of developing multiple primary tumors and with earlier cancer occurrence patterns.
We finally examine the sequence in which multiple cancer may develop over adult lifetime in R337H and non-R337H carriers (Fig. 5). In females, breast cancer was the most frequently first (R337H: n = 29, 72.5%; non-R337H: n = 22, 52.4%), second (R337H: n = 17, 42.5%; non-R337H: n = 23, 54.8%), and third (R337H: n = 4, 33.3%; non-R337H: n = 5, 50.0%) diagnosis, not only in patients who presented with first diagnosis of breast cancer but also in patients with any other first malignancy (Fig. 5A). Notably, of the 17 R337H patients presenting with a second breast cancer, 12 (70.6%) exhibited contralateral breast cancers (Supplementary Table S4), underscoring the importance of continued bilateral breast screening even after an initial breast cancer diagnosis in these patients. We lack this specific information for the full non-R337H cohort. Besides breast cancer, female R337H carriers (40 in total) tended to develop lung (15.0%, n = 6), soft-tissue sarcoma (10.0%, n = 4), and thyroid cancer (7.5%, n = 3) as their most common second diagnoses. Among males (16 in total), the most frequent second diagnoses in R337H carriers were lung cancer (31%, n = 5), kidney cancer (19%, n = 3), and soft-tissue sarcoma (12%, n = 2), whereas prostate cancer was the most common third diagnosis (50%, 2/4) (Fig. 5B). In contrast, in non-R337H male carriers (22 in total), the most frequent second diagnoses were soft-tissue sarcoma (23%, n = 5), CNS cancer (23%, n = 5), and gastric cancer (14%, n = 3).
Fig. 5.
Spectrum of second and third primary tumors in adult R337H carriers and non-R337H carriers. Sankey diagrams show the patterns of occurrence of the first, second, and third tumor types diagnosed in A) female and B) male individuals carrying the TP53 R337H and non-R337H variants.
Overall, these observations support that R337H carriers have a significant lifetime risk of multiple cancers, with patterns of occurrence that differ from those of non-R337H carriers. Lung cancer, in particular, appeared to be a common second diagnosis in both male (31%, 5/16) and female (15%, 6/40) R337H carriers (compared to 9% (2/22) and 7% (3/42), respectively, in non-R337H carriers).
Finally, we produced a comprehensive summary of the tumor spectrum and cumulative cancer risks in adult R337H carriers, stratified by sex and age (Fig. 6). For male R337H carriers, soft tissue sarcomas were the most common first tumor (30.0%, 12/40), followed by prostate cancer (12%, 5/40) and lung cancer (10.0%, 4/40). For female R337H carriers, breast cancer was overwhelmingly the most frequent first tumor (75.4%, 92/122), followed by soft tissue sarcomas (9.0%, 11/122) and lung cancer (8.2%, 10/122). Regarding second tumors (Fig. 6A, outer pie charts), women with primary breast cancer often developed a second breast cancer (15.2% (14/92); Fig. 6A, outer pie chart). For patients with primary soft tissue or lung tumors, the second most common tumors were also soft tissue (18.2%, 2/11) and lung (20%, 2/10), respectively. For men with a primary soft tissue tumor, the most common second cancers were kidney (16.7%, 2/12), biliary tract (8.3%, 1/12), and prostate (8.3%, 1/12). Notably, a substantial proportion of carriers (shown in gray) did not develop a second malignancy within the follow-up period (<5 years or >5 years), while others died after the first tumor (labeled as “D”). In terms of cumulative cancer risks, by age 60, male R337H carriers exhibited a 47% risk of developing a first tumor, with soft tissue sarcomas (30%), prostate cancer (12%), and lung cancer (10%) being most prevalent (Fig. 6B). In contrast, female carriers showed a markedly higher cumulative risk of 87% by age 60, dominated by breast cancer (75%), followed by soft tissue sarcomas (9%) and lung cancer (8%). The risk of developing a second tumor also differed between sexes. By age 65, males showed a 45% cumulative risk, primarily composed of lung (31%), kidney (19%), and soft tissue (12%) cancers. Females demonstrated a higher second tumor risk of 88%, with breast (42%), lung (15%), and soft tissue (10%) cancers being most common. These findings highlight the sex-specific differences in cancer risks and patterns among R337H carriers, emphasizing the need for tailored screening and management strategies.
Fig. 6.
A summary of the tumor spectrum and cumulative cancer risks in adult R337H carriers by sex and age. A) Pie/donut charts showing percentages of first (central pie charts) and second (outer pie charts) tumor types diagnosed in R337H carriers. Carriers with no second malignancies (in gray) were further stratified into: dead (D), alive for up to 5 years of follow-up (≤5) or more than 5 years (>5); B) Schematic representation of a decision tree to support clinical decisions on screening of R337H adult carriers based on our findings.
Discussion
This study is the first systematic and comparative analysis of the risk and patterns of cancer in adult carriers of the TP53 R337H Brazilian founder allele versus carriers of other P/LP TP53 variants. It compiles and employs the largest database of adult carriers assembled to date, including 303 Brazilian R337H carriers from 160 families, compared with 42 carriers of non-R337H P/LP variants from 29 Brazilian families and 363 carriers of P/LP variants from 172 non-Brazilian families of mostly Caucasian background (see Methods). Results show that adult carriers of R337H allele have a lifetime pattern of risk that matches the Li-Fraumeni spectrum, characterized by excess risk of breast cancer in females and soft tissue sarcoma in both sexes. However, when compared to other P/LP variants, risk patterns in R337H carriers only partially recapitulates the LFS spectrum. Previous studies have provided valuable insights into the tumor spectrum of R337H carriers8,16; however, these investigations were predominantly focused on pediatric populations, specific tumor types, or were constrained by small sample sizes and limited cohorts.14,15,17 Our study addresses critical gaps by examining the largest Brazilian cohort to date, focusing on adulthood, and providing comprehensive analyses of lifetime cancer risk, temporal tumor trends, sex differences, and the risk of multiple primary tumors in R337H carriers.
The main traits that characterize adult R337 carriers versus other variants carriers (summarized in Fig. 6A) are: (1) low prevalence, if not absence, of typical LFS tumors such as CNS cancers and osteosarcomas; (2) high prevalence of some cancers not considered as typical of the LFS spectrum (renal cancer, thyroid cancers); (3) frequent occurrence of lung and prostate cancers, in particular as second diagnoses in males; and (4) strong sex-related difference in lifetime risk, females having a risk almost twice higher than males. Thus, our findings collectively demonstrate that the cancer risk associated with the R337H variant is complex and multifaceted. Curiously, another variant, P152L, also exhibits a non-typical LFS cancer profile,20 with lower age-related risks for all non-adrenal tumors compared to other LFS patients (particularly those with mutations in codons 245 and 248). Interestingly, while R337H retains a high risk of breast cancer, P152L is notable for its absence of breast cancer cases. These variations in cancer patterns among different TP53 variants underscore the complexity of genotype–phenotype correlations in LFS and highlight the importance of variant-specific risk assessments. Given the high population prevalence of R337H, our results will raise awareness of cancer risk in this adult population and will help in designing appropriate strategies for carrier surveillance and early diagnosis (Fig. 6B). However, it is important to acknowledge that our results may include a bias towards cancer cases. This ascertainment bias could potentially lead to an overestimation of cancer risk, particularly when compared to individuals identified through population-based R337H screening programs. Thus, clinicians should interpret our risk estimates cautiously when counseling individuals identified as R337H carriers through population screening, keeping in mind that these individuals may have a lower cancer risk than suggested by our cohort data.
Any study of the variables that influence a genetic predisposition to develop cancer must take into account and explore those variables that impact the phenotypes of that cancer. Clearly, different TP53 variant alleles can influence cancer phenotypes as a consequence of their structural and functional impact on the p53 protein. In particular, TP53 variants occurring in different domains of the p53 protein may exert tissue specific effects. For example, somatic mutations in the p53 proline domain most often occur in skin cancers,21 suggesting enhanced effects of such variants in skin cells. The R337H variant is atypical among LFS predisposing variants in its location in the p53 protein structure (tetramerization domain, whereas 80% of missense LFS variants fall in the DNA-binding domain) and its biochemical characteristics (uniquely sensitive to pH, enabling normal p53 oligomer assembly at neutral cell pH but preventing it at slightly elevated pH).22 In vitro studies and mouse models have shown that the p.R337H protein can retain at least a degree of wild-type p53 transcriptional activities.23 Based on these observations, the R337H variant has been qualified as hypomorphic, potentially causing an attenuated phenotype compared to common P/LP variants. However, the present study suggests that this suspected hypomorphic effect does not equally affect LFS features at all ages and in all tissues of R337H carriers. Additionally, it is important to note that our non-R337H comparison group includes both Brazilian carriers and carriers from diverse geographic areas as reported in the NCI TP53 database. This composition provides a broad representation of TP53 variants but also introduces potential variability in genetic backgrounds, environmental exposures, and healthcare practices. While this diversity strengthens the generalizability of our findings regarding the distinct characteristics of R337H carriers, it also necessitates caution in interpreting direct comparisons between the R337H and non-R337H groups. Future studies focusing on region-specific comparisons may provide additional insights into the unique features of the R337H variant in the Brazilian population.
An analysis of the cumulative risk of developing cancer at age 50 years (R337H = 54% vs non-R337H = 78%, p= < 0.0001) demonstrates that over the first 50 years of life, R337H carries a lower risk than other P/LP, but by age 65 years, these percentages are of 78% vs 81%, respectively, indicating that the rates of cancer occurrence strongly increases from 50 to 65 years of age in R337H carriers. Cancer patterns in this age group include postmenopausal breast cancer, sarcomas (in particular leiomyosarcoma) as well as lung or prostate cancers. Thus age appears to be an important variable, with different TP53 alleles having different rates of cancer development as a function of age. Such selective effect of age on cancer risk has also been observed in children and adolescent carriers of R337H, who frequently develop adrenal cortical carcinoma but show much fewer osteogenic sarcomas, choroid plexus papilloma/carcinomas, rhabdomyosarcomas or medulloblastomas than carriers of other P/LP variants.8,24 These observations clearly stress that the effect of the R337H variant is far from uniform across tissues and age and suggest that the risk of developing a particular cancer with different mutant TP53 alleles is largely tissue specific. Indeed, the levels of p53 activity required to suppress cancer may differ from one tissue (or cell type) to another. Thus, a relatively modest attenuation of p53 activity caused by the R337H variant may be sufficient to a high risk of cancer in some tissues (breast, connective tissues) but not in others (bone, for example). The reasons underlying these cell or tissue differences in the sensitivity to p53 activity dosage are far from being understood.
Among other factors that may influence phenotypic variations in cancer predisposition, the genetic ancestry of the patient and the chromosomal context surrounding the specific variant allele, play an essential role. In the case of TP53, this notion has been experimentally addressed by comparing tumor patterns in mice from seven different inbred strains, each having the same R172H trp53 mutant allele (mouse counterpart of human R175H) in the heterozygous form. This study uncovered significant differences in penetrance, age of onset and tissue type of tumors across different inbred strains, suggesting that the genetic ancestry or background had a clear influence on these phenotypes.25 In the case of the Brazilian R337H allele, the genetic founder effect began several hundred years ago and expanded over several centuries of population growth in Southern Brazil, resulting in a population in which about one in three hundred individuals nowadays carry a heterozygous R337H variant on a Lusitanian/Hispanic haplotype of European background.9,26,27 There is indeed evidence that the majority of Brazilian R337H carriers are of Lusitanian origin.26 Nevertheless, since the population of Southern Brazil is composed of successive waves of migration from Europe, Asia and Africa over a background of native Amerindian populations, the genomes of these carriers are likely to reflect these influences, which is an uncontrolled variable in the population under study. This genetic diversity may represent an important variable to explain why a proportion of R337H carriers may develop cancer only as adults or even remain cancer-free over their entire lifespan.
Sex is part of the genetic makeup of an individual and this study clearly shows that the sex of the patient is in itself a risk factor, with R337H females having a significantly higher risk of developing cancers (65%) than their male counterparts (30%) early in life. Unlike spontaneous cancers where males have a higher incidence of cancers, in LFS TP53 mutant carriers, females have a higher incidence of cancers at earlier ages than males. This is largely due to the fact that breast tumors are the most common tumor in LFS, independent of the mutant allele. On the other hand males with R337H alleles develop more soft tissue sarcomas, at all ages, than their females counterparts.
In conjunction to the factors discussed above, multiple other factors may modulate the phenotypes caused by a genetic predisposition. The environment, through its genetic and epigenetic effects, may impact individual risk trajectories in relation with age and cumulative exposure to exogenous risk factors. Immunological response to the tumor and to the variant itself may also be an important factor in determining the lifetime risk.28 There are also certainly stochastic events occurring over a lifetime that may contribute to the outcomes of a genetic predisposition to cancer.25 Finally, studies have shown that certain XAF1 polymorphisms may influence the age of cancer onset and overall cancer risk in TP53 R337H carriers.29,30 This gene–gene interaction suggests that genomic variations in XAF1 may act as ancestry-specific modifiers, potentially explaining some of the variability in cancer manifestation observed among R337H carriers. However, the functional relationship between XAF1 variants and the R337H mutation is not yet fully understood. Further research into this relationship could provide valuable insights into the molecular mechanisms underlying cancer development in R337H carriers. Understanding the relative importance of these factors, and their interactions, in determining individual cancer risk, is the focus of molecular epidemiology and is a daunting task in human populations where all these variables can show tremendous variations. A unique genetic predisposition context associated with a founder allele such as R337H may provide an unprecedented setup to further dissect these influences and to better understand the genetic program that leads to the formation of a tumor, its tissue specificity, age of onset, sex, and projected outcomes. Such studies will considerably contribute to a better understanding of the properties that enable stem cells to evolve towards early cancer in a p53-haploid functional or haplo-insufficient environment, and to the mechanisms that underlie the continuous occurrence of such cancers over a lifetime.
Collectively, our findings suggest that the cancer risk profile of R337H carriers differs from that of individuals with other pathogenic/likely pathogenic TP53 variants, potentially warranting modifications to existing surveillance protocols. While the core elements of LFS surveillance remain crucial, such as breast cancer screening for women and whole-body MRI for soft tissue sarcoma detection, some adjustments may be considered. The high prevalence of lung cancer in both sexes (10% in males, 8% in females) suggests that incorporating lung-specific screening, might be beneficial, particularly for individuals over 45. For male R337H carriers, enhanced prostate cancer screening may be warranted given its prevalence (12% of first tumors). The significant increase in cancer risk between ages 45 and 60 indicates that surveillance should be particularly intensive during this period. Moreover, the high cumulative risk of second tumors (45% in males and 88% in females by age 65) underscores the need for continued vigilant screening after a first cancer diagnosis. Importantly, the higher overall cancer risk in female R337H carriers (87% vs 47% at age 60 for first tumor) may justify a more intensive surveillance approach for women. Although many R337H families meet attenuated LFS criteria, there are some R337H families that meet classic LFS criteria. Other environmental and genetic modifiers may play a role in these cases. For this reason, Brazilian surveillance protocol modifications should take into account not only the R337H status but also family phenotype. We believe that a combination of genotype and phenotype should guide surveillance. Finally, while these proposed modifications aim to tailor surveillance to the specific risk profile of R337H carriers, it is crucial to emphasize that any changes to established protocols should be carefully evaluated through prospective clinical studies before widespread implementation.
In terms of limitations and future directions, while our study provides valuable insights into the cancer risk profiles of R337H carriers, we acknowledge several limitations in our current data that warrant further investigation. Firstly, our study does not address the potential impact of the R337H variant on treatment outcomes. The response to various cancer therapies, including chemotherapy and targeted treatments, may differ between R337H carriers and those with other TP53 variants. This is a critical area for future research, as it could inform personalized treatment strategies for these patients. Secondly, we did not have data on the use of radiation therapy in our cohort. Given the known sensitivity of TP53 mutation carriers to ionizing radiation, understanding how radiation therapy might alter the clinical course in R337H carriers compared to other TP53 variant carriers is crucial. Future studies should aim to collect detailed treatment data and long-term follow-up to assess the impact of radiation therapy on both tumor control and the risk of second malignancies. Thirdly, our study was not designed to detect potential differences in treatment response or prognosis between different TP53 variants. Such differences, if they exist, could have significant implications for clinical management. Larger, prospective studies with standardized treatment protocols and long-term follow-up will be necessary to elucidate these potential variant-specific effects. Lastly, while we applied specific criteria to distinguish between second primary breast cancers and relapses, the accuracy of this classification may be limited by the available clinical and pathological data. More advanced molecular techniques, such as comprehensive genomic profiling, could provide more definitive classification in future studies.
Finally, we believe that our comprehensive analysis of cancer patterns in carriers of the R337H TP53 founder variant will help doctors, genetic counselors, patients and families to elaborate appropriate strategies for increasing awareness, informing and providing adequate early detection measures to the thousands of adult carriers who are at risk in the Brazilian population.
Contributors
PAFG and GDAG performed statistical data analyses, preparation of figures and writing. MIA, RLS, and JP conducted patient data collection and verification, and participated in the writing and figure preparation. PAFG, GDAG, JP, and MIA verified the data; PAFG, GDAG, JP, and MIA had access to raw data. PAFG and MIA had final responsibility for the decision to submit for publication. All authors read, reviewed and approved the final manuscript.
Data sharing statement
Data from the R337H cohort described in the manuscript is available in the Supplementary Materials. Data from the TP53 mutation database is publicly accessible at https://tp53.cancer.gov.
Declaration of interests
The authors have declared no conflicts of interest.
Acknowledgements
Authors express their gratitude to the patients and their families for their invaluable participation in this study. This work was supported by grant #2018/15579-8, São Paulo Research Foundation (FAPESP) to PAFG, grant: #2017/19541-2 to GDAG and grant: #308785/2020-7 to MIA, Conselho Nacional de Pesquisa (CNPq), and Hospital Sírio-Libanês to PAFG and GDAG. PAP was supported by Hospital de Clínicas de Porto Alegre and Rio Grande do Sul Research Foundation (FAPERGS).
Funders had no participation in the study design, in the collection, analysis, and interpretation of data, in the writing of the manuscript, nor in the decision to submit the paper for publication.
Footnotes
Supplementary data related to this article can be found at https://doi.org/10.1016/j.lana.2024.100982.
Contributor Information
Pedro A.F. Galante, Email: pgalante@mochsl.org.br.
Maria Isabel Achatz, Email: miachatz@mochsl.org.br.
Appendix ASupplementary data
References
- 1.Olivier M., Hollstein M., Hainaut P. TP53 mutations in human cancers: origins, consequences, and clinical use. Cold Spring Harb Perspect Biol. 2010;2 doi: 10.1101/cshperspect.a001008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Levine A.J., Oren M. The first 30 years of p53: growing ever more complex. Nat Rev Cancer. 2009;9:749–758. doi: 10.1038/nrc2723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.de Andrade K.C., Frone M.N., Wegman-Ostrosky T., et al. Variable population prevalence estimates of germline TP53 variants: a gnomAD-based analysis. Hum Mutat. 2019;40:97–105. doi: 10.1002/humu.23673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Li F.P., Fraumeni J.F., Jr., Mulvihill J.J., et al. A cancer family syndrome in twenty-four kindreds. Cancer Res. 1988;48:5358–5362. [PubMed] [Google Scholar]
- 5.Olivier M., Goldgar D.E., Sodha N., et al. Li-Fraumeni and related syndromes: correlation between tumor type, family structure, and TP53 genotype. Cancer Res. 2003;63:6643–6650. [PubMed] [Google Scholar]
- 6.Kratz C.P., Freycon C., Maxwell K.N., et al. Analysis of the Li-Fraumeni spectrum based on an international germline TP53 variant data set: an international agency for research on cancer TP53 database analysis. JAMA Oncol. 2021;7:1800–1805. doi: 10.1001/jamaoncol.2021.4398. https://jamanetwork.com/journals/jamaoncology/article-abstract/2785678 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ribeiro R.C., Sandrini F., Figueiredo B., et al. An inherited p53 mutation that contributes in a tissue-specific manner to pediatric adrenal cortical carcinoma. Proc Natl Acad Sci U S A. 2001;98:9330–9335. doi: 10.1073/pnas.161479898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Achatz M.I.W., Olivier M., Le Calvez F., et al. The TP53 mutation, R337H, is associated with Li-Fraumeni and Li-Fraumeni-like syndromes in Brazilian families. Cancer Lett. 2007;245:96–102. doi: 10.1016/j.canlet.2005.12.039. [DOI] [PubMed] [Google Scholar]
- 9.Garritano S., Gemignani F., Palmero E.I., et al. Detailed haplotype analysis at the TP53 locus in p.R337H mutation carriers in the population of Southern Brazil: evidence for a founder effect. Hum Mutat. 2010;31:143–150. doi: 10.1002/humu.21151. [DOI] [PubMed] [Google Scholar]
- 10.Palmero E.I., Schüler-Faccini L., Caleffi M., et al. Detection of R337H, a germline TP53 mutation predisposing to multiple cancers, in asymptomatic women participating in a breast cancer screening program in Southern Brazil. Cancer Lett. 2008;261:21–25. doi: 10.1016/j.canlet.2007.10.044. [DOI] [PubMed] [Google Scholar]
- 11.Custodio G., Taques G.R., Figueiredo B.C., et al. Increased incidence of choroid plexus carcinoma due to the germline TP53 R337H mutation in Southern Brazil. PLoS One. 2011;6 doi: 10.1371/journal.pone.0018015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Pinto E.M., Zambetti G.P. What 20 years of research has taught us about the TP53 p.R337H mutation. Cancer. 2020;126:4678–4686. doi: 10.1002/cncr.33143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kratz C.P., Achatz M.I., Brugières L., et al. Cancer screening recommendations for individuals with Li-Fraumeni syndrome. Clin Cancer Res. 2017;23:e38–e45. doi: 10.1158/1078-0432.CCR-17-0408. [DOI] [PubMed] [Google Scholar]
- 14.Volc S.M., Ramos C.R.N., Galvão H.C.R., et al. The Brazilian TP53 mutation (R337H) and sarcomas. PLoS One. 2020;15 doi: 10.1371/journal.pone.0227260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Seidinger A.L., Mastellaro M.J., Paschoal Fortes F., et al. Association of the highly prevalent TP53 R337H mutation with pediatric choroid plexus carcinoma and osteosarcoma in Southeast Brazil. Cancer. 2011;117:2228–2235. doi: 10.1002/cncr.25826. [DOI] [PubMed] [Google Scholar]
- 16.Ferreira A.M., Brondani V.B., Helena V.P., et al. Clinical spectrum of Li-Fraumeni syndrome/Li-Fraumeni-like syndrome in Brazilian individuals with the TP53 p.R337H mutation. J Steroid Biochem Mol Biol. 2019;190:250–255. doi: 10.1016/j.jsbmb.2019.04.011. [DOI] [PubMed] [Google Scholar]
- 17.Formiga M.N.D.C., de Andrade K.C., Kowalski L.P., Achatz M.I. Frequency of thyroid carcinoma in Brazilian TP53 p.R337H carriers with Li Fraumeni syndrome. JAMA Oncol. 2017;3:1400–1402. doi: 10.1001/jamaoncol.2016.6389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Pinto E.M., Maxwell K.N., Halalsheh H., et al. Clinical and functional significance of TP53 exon 4-intron 4 splice junction variants. Mol Cancer Res. 2022;20:207–216. doi: 10.1158/1541-7786.MCR-21-0583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Siegel R.L., Giaquinto A.N., Jemal A. Cancer statistics, 2024. CA Cancer J Clin. 2024;74:12–49. doi: 10.3322/caac.21820. [DOI] [PubMed] [Google Scholar]
- 20.Evans D.G., Harkness E.F., Woodward E.R. TP53 c.455C>T p.(Pro152Leu) pathogenic variant is a lower risk allele with attenuated risks of breast cancer and sarcoma. J Med Genet. 2023;60:1057–1060. doi: 10.1136/jmg-2022-109133. [DOI] [PubMed] [Google Scholar]
- 21.Hoyos D., Greenbaum B., Levine A.J. The genotypes and phenotypes of missense mutations in the proline domain of the p53 protein. Cell Death Differ. 2022;29:938–945. doi: 10.1038/s41418-022-00980-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.DiGiammarino E.L., Lee A.S., Cadwell C., et al. A novel mechanism of tumorigenesis involving pH-dependent destabilization of a mutant p53 tetramer. Nat Struct Biol. 2002;9:12–16. doi: 10.1038/nsb730. [DOI] [PubMed] [Google Scholar]
- 23.Jeffers J.R., Pinto E.M., Rehg J.E., et al. The common germline mutation is hypomorphic and confers incomplete penetrance and late tumor onset in a mouse model. Cancer Res. 2021;81:2442–2456. doi: 10.1158/0008-5472.CAN-20-1750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Achatz M.I., Zambetti G.P. The inherited p53 mutation in the Brazilian population. Cold Spring Harb Perspect Med. 2016;6 doi: 10.1101/cshperspect.a026195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Chan C.S., Sun Y., Ke H., et al. Genetic and stochastic influences upon tumor formation and tumor types in Li-Fraumeni mouse models. Life Sci Alliance. 2021;4 doi: 10.26508/lsa.202000952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Paskulin D.D., Giacomazzi J., Achatz M.I., et al. Ancestry of the Brazilian TP53 c.1010G>A (p.Arg337His, R337H) founder mutation: clues from haplotyping of short tandem repeats on chromosome 17p. PLoS One. 2015;10 doi: 10.1371/journal.pone.0143262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Giacomazzi J., Graudenz M.S., Osorio C.A., et al. Prevalence of the TP53 p.R337H mutation in breast cancer patients in Brazil. PLoS One. 2014;9 doi: 10.1371/journal.pone.0099893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hoyos D., Zappasodi R., Schulze I., et al. Fundamental immune-oncogenicity trade-offs define driver mutation fitness. Nature. 2022;606:172–179. doi: 10.1038/s41586-022-04696-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pinto E.M., Figueiredo B.C., Chen W., et al. XAF1 as a modifier of p53 function and cancer susceptibility. Sci Adv. 2020;6 doi: 10.1126/sciadv.aba3231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Pinto E.M., Fridman C., Figueiredo B.C., et al. Multiple TP53 p.R337H haplotypes and implications for tumor susceptibility. HGG Adv. 2024;5 doi: 10.1016/j.xhgg.2023.100244. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.