Abstract
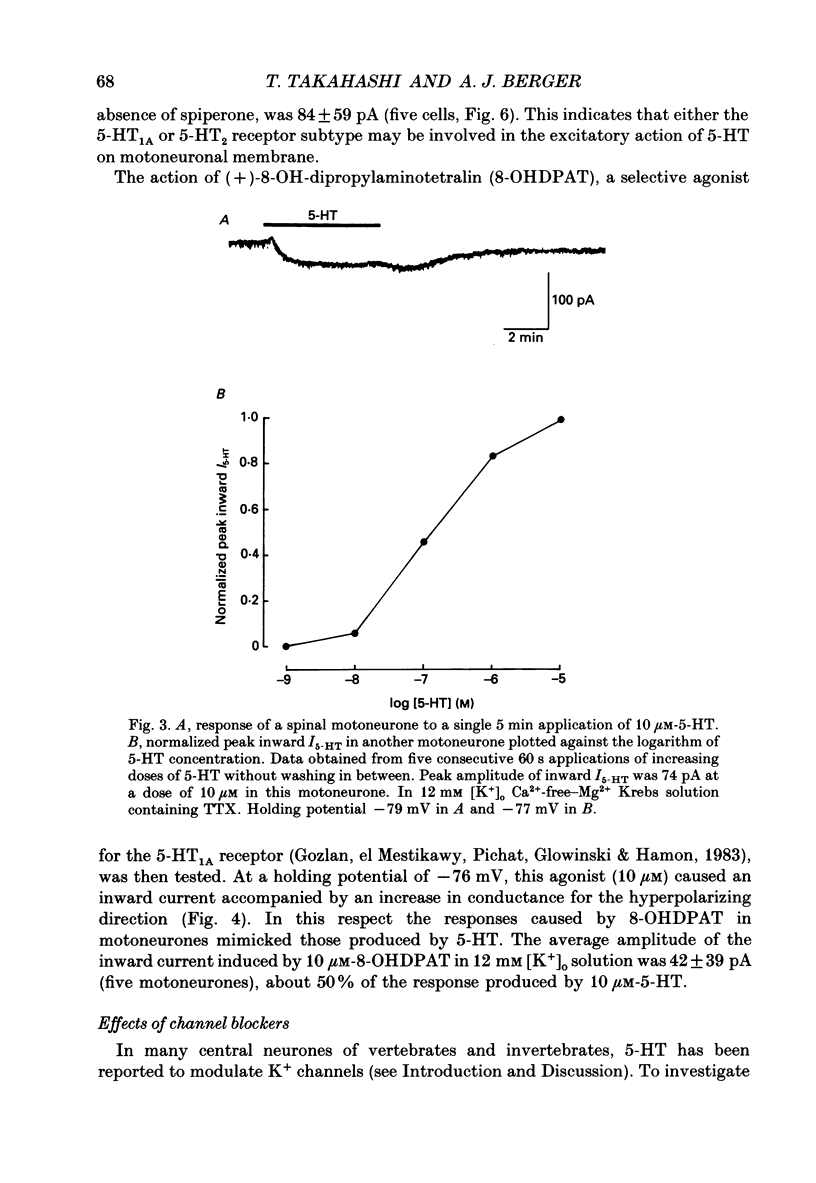
1. The effects of serotonin (5-HT) on visually identified motoneurones were investigated using the whole-cell recording technique in a neonatal rat spinal cord slice preparation. 2. In current-clamp recordings, bath application of 5-HT depolarized motoneurones. This effect was observed after synaptic inputs were abolished by replacing external Ca2+ with Mg2+. 3. In voltage-clamp recordings at holding potentials of -70 to -90 mV, 5-HT induced an inward current (I5-HT) in motoneurones in a Ca2(+)-free-Mg2+ solution containing tetrodotoxin. This inward current was accompanied by an increase in membrane conductance, which was prominent at voltages negative to the holding potential. 4. The inward I5-HT response declined with repeated short applications of 5-HT. I5-HT produced by a single prolonged application (5 min) was only slightly diminished during the application period. 5. The minimum effective dose of 5-HT for initiating the inward I5-HT was less than 10 nM. At 10 microM, I5-HT approached maximal levels. The averaged dissociation constant (Kd) for 5-HT was approximately 120 nM. 6. Application of spiperone, the mixed 5-HT1A, 5-HT2 receptor antagonist, blocked the inward I5-HT. Application of (+)-8-OH-dipropylaminotetralin (8-OHDPAT), a 5-HT1A agonist, mimicked the action of 5-HT. 7. Various K+ channel blockers including tetraethylammonium chloride (30 mM), 4-aminopyridine (4 mM) and apamin (100 nM) did not abolish I5-HT. Application of extracellular Cs+ (10 mM) blocked I5-HT. 8. Peak inward I5-HT became larger with increasing extracellular K+. With low Cl- pipette solution (less than 1 mM), or in low extracellular Na+ solution (26 mM), the inward I5-HT was not abolished. 9. The current-voltage relation of I5-HT displayed inward rectification. In high external K+ concentration (20 mM), the reversal potential was about -29 mV, which is close to that of the inward rectifier evoked in motoneurones by membrane hyperpolarization. 10. The current generated by 5-HT displayed similar characteristics to the inward rectifying current induced in motoneurones by membrane hyperpolarization. It is thus suggested that the 5-HT-induced current is possibly mediated by the intrinsic inward rectifier conductance.
Full text
PDF












Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Andrade R., Nicoll R. A. Pharmacologically distinct actions of serotonin on single pyramidal neurones of the rat hippocampus recorded in vitro. J Physiol. 1987 Dec;394:99–124. doi: 10.1113/jphysiol.1987.sp016862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Atsumi S., Sakamoto H., Yokota S., Fujiwara T. Substance P and 5-hydroxytryptamine immunoreactive presynaptic boutons on presumed alpha-motoneurons in the chicken ventral horn. Arch Histol Jpn. 1985 Apr;48(2):159–172. doi: 10.1679/aohc.48.159. [DOI] [PubMed] [Google Scholar]
- Barasi S., Roberts M. H. The modification of lumbar motoneurone excitability by stimulation of a putative 5-hydroxytryptamine pathway. Br J Pharmacol. 1974 Nov;52(3):339–348. doi: 10.1111/j.1476-5381.1974.tb08601.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benson J. A., Levitan I. B. Serotonin increases an anomalously rectifying K+ current in the Aplysia neuron R15. Proc Natl Acad Sci U S A. 1983 Jun;80(11):3522–3525. doi: 10.1073/pnas.80.11.3522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bobker D. H., Williams J. T. Serotonin augments the cationic current Ih in central neurons. Neuron. 1989 Jun;2(6):1535–1540. doi: 10.1016/0896-6273(89)90041-x. [DOI] [PubMed] [Google Scholar]
- Cardona A., Rudomin P. Activation of brainstem serotoninergic pathways decreases homosynaptic depression of monosynaptic responses of frog spinal motoneurons. Brain Res. 1983 Dec 5;280(2):373–378. doi: 10.1016/0006-8993(83)90070-7. [DOI] [PubMed] [Google Scholar]
- Colino A., Halliwell J. V. Differential modulation of three separate K-conductances in hippocampal CA1 neurons by serotonin. Nature. 1987 Jul 2;328(6125):73–77. doi: 10.1038/328073a0. [DOI] [PubMed] [Google Scholar]
- Conn P. J., Sanders-Bush E., Hoffman B. J., Hartig P. R. A unique serotonin receptor in choroid plexus is linked to phosphatidylinositol turnover. Proc Natl Acad Sci U S A. 1986 Jun;83(11):4086–4088. doi: 10.1073/pnas.83.11.4086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Constanti A., Galvan M. Fast inward-rectifying current accounts for anomalous rectification in olfactory cortex neurones. J Physiol. 1983 Feb;335:153–178. doi: 10.1113/jphysiol.1983.sp014526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DiFrancesco D., Ojeda C. Properties of the current if in the sino-atrial node of the rabbit compared with those of the current iK, in Purkinje fibres. J Physiol. 1980 Nov;308:353–367. doi: 10.1113/jphysiol.1980.sp013475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fargin A., Raymond J. R., Lohse M. J., Kobilka B. K., Caron M. G., Lefkowitz R. J. The genomic clone G-21 which resembles a beta-adrenergic receptor sequence encodes the 5-HT1A receptor. Nature. 1988 Sep 22;335(6188):358–360. doi: 10.1038/335358a0. [DOI] [PubMed] [Google Scholar]
- Fulton B. P., Walton K. Electrophysiological properties of neonatal rat motoneurones studied in vitro. J Physiol. 1986 Jan;370:651–678. doi: 10.1113/jphysiol.1986.sp015956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gerschenfeld H. M., Paupardin-Tritsch D. Ionic mechanisms and receptor properties underlying the responses of molluscan neurones to 5-hydroxytryptamine. J Physiol. 1974 Dec;243(2):427–456. doi: 10.1113/jphysiol.1974.sp010761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gozlan H., El Mestikawy S., Pichat L., Glowinski J., Hamon M. Identification of presynaptic serotonin autoreceptors using a new ligand: 3H-PAT. Nature. 1983 Sep 8;305(5930):140–142. doi: 10.1038/305140a0. [DOI] [PubMed] [Google Scholar]
- Gundersen C. B., Miledi R., Parker I. Serotonin receptors induced by exogenous messenger RNA in Xenopus oocytes. Proc R Soc Lond B Biol Sci. 1983 Aug 22;219(1214):103–109. doi: 10.1098/rspb.1983.0062. [DOI] [PubMed] [Google Scholar]
- Hagiwara S., Miyazaki S., Moody W., Patlak J. Blocking effects of barium and hydrogen ions on the potassium current during anomalous rectification in the starfish egg. J Physiol. 1978 Jun;279:167–185. doi: 10.1113/jphysiol.1978.sp012338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halliwell J. V., Adams P. R. Voltage-clamp analysis of muscarinic excitation in hippocampal neurons. Brain Res. 1982 Oct 28;250(1):71–92. doi: 10.1016/0006-8993(82)90954-4. [DOI] [PubMed] [Google Scholar]
- Holtman J. R., Jr, Dick T. E., Berger A. J. Involvement of serotonin in the excitation of phrenic motoneurons evoked by stimulation of the raphe obscurus. J Neurosci. 1986 Apr;6(4):1185–1193. doi: 10.1523/JNEUROSCI.06-04-01185.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hounsgaard J., Hultborn H., Jespersen B., Kiehn O. Bistability of alpha-motoneurones in the decerebrate cat and in the acute spinal cat after intravenous 5-hydroxytryptophan. J Physiol. 1988 Nov;405:345–367. doi: 10.1113/jphysiol.1988.sp017336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hounsgaard J., Kiehn O. Ca++ dependent bistability induced by serotonin in spinal motoneurons. Exp Brain Res. 1985;57(2):422–425. doi: 10.1007/BF00236551. [DOI] [PubMed] [Google Scholar]
- Huang J. C., Peroutka S. J. Identification of 5-hydroxytryptamine binding site subtypes in rat spinal cord. Brain Res. 1987 Dec 8;436(1):173–176. doi: 10.1016/0006-8993(87)91572-1. [DOI] [PubMed] [Google Scholar]
- Hugues M., Romey G., Duval D., Vincent J. P., Lazdunski M. Apamin as a selective blocker of the calcium-dependent potassium channel in neuroblastoma cells: voltage-clamp and biochemical characterization of the toxin receptor. Proc Natl Acad Sci U S A. 1982 Feb;79(4):1308–1312. doi: 10.1073/pnas.79.4.1308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Julius D., MacDermott A. B., Axel R., Jessell T. M. Molecular characterization of a functional cDNA encoding the serotonin 1c receptor. Science. 1988 Jul 29;241(4865):558–564. doi: 10.1126/science.3399891. [DOI] [PubMed] [Google Scholar]
- Klein M., Camardo J., Kandel E. R. Serotonin modulates a specific potassium current in the sensory neurons that show presynaptic facilitation in Aplysia. Proc Natl Acad Sci U S A. 1982 Sep;79(18):5713–5717. doi: 10.1073/pnas.79.18.5713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lotshaw D. P., Levitan I. B. Reciprocal modulation of calcium current by serotonin and dopamine in the identified Aplysia neuron R15. Brain Res. 1988 Jan 26;439(1-2):64–76. doi: 10.1016/0006-8993(88)91462-x. [DOI] [PubMed] [Google Scholar]
- McCall R. B., Aghajanian G. K. Serotonergic facilitation of facial motoneuron excitation. Brain Res. 1979 Jun 15;169(1):11–27. doi: 10.1016/0006-8993(79)90370-6. [DOI] [PubMed] [Google Scholar]
- Monroe P. J., Smith D. J. Characterization of multiple [3H]5-hydroxytryptamine binding sites in rat spinal cord tissue. J Neurochem. 1983 Aug;41(2):349–355. doi: 10.1111/j.1471-4159.1983.tb04749.x. [DOI] [PubMed] [Google Scholar]
- Pape H. C., McCormick D. A. Noradrenaline and serotonin selectively modulate thalamic burst firing by enhancing a hyperpolarization-activated cation current. Nature. 1989 Aug 31;340(6236):715–718. doi: 10.1038/340715a0. [DOI] [PubMed] [Google Scholar]
- Paupardin-Tritsch D., Deterre P., Gerschenfeld H. M. Relationship between two voltage-dependent serotonin responses of molluscan neurones. Brain Res. 1981 Jul 27;217(1):201–206. doi: 10.1016/0006-8993(81)90201-8. [DOI] [PubMed] [Google Scholar]
- Peroutka S. J., Snyder S. H. Multiple serotonin receptors: differential binding of [3H]5-hydroxytryptamine, [3H]lysergic acid diethylamide and [3H]spiroperidol. Mol Pharmacol. 1979 Nov;16(3):687–699. [PubMed] [Google Scholar]
- Steinbusch H. W. Distribution of serotonin-immunoreactivity in the central nervous system of the rat-cell bodies and terminals. Neuroscience. 1981;6(4):557–618. doi: 10.1016/0306-4522(81)90146-9. [DOI] [PubMed] [Google Scholar]
- Takahashi T. Inhibitory miniature synaptic potentials in rat motoneurons. Proc R Soc Lond B Biol Sci. 1984 Mar 22;221(1222):103–109. doi: 10.1098/rspb.1984.0025. [DOI] [PubMed] [Google Scholar]
- Takahashi T. Inward rectification in neonatal rat spinal motoneurones. J Physiol. 1990 Apr;423:47–62. doi: 10.1113/jphysiol.1990.sp018010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahashi T. Membrane currents in visually identified motoneurones of neonatal rat spinal cord. J Physiol. 1990 Apr;423:27–46. doi: 10.1113/jphysiol.1990.sp018009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahashi T., Neher E., Sakmann B. Rat brain serotonin receptors in Xenopus oocytes are coupled by intracellular calcium to endogenous channels. Proc Natl Acad Sci U S A. 1987 Jul;84(14):5063–5067. doi: 10.1073/pnas.84.14.5063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- VanderMaelen C. P., Aghajanian G. K. Intracellular studies showing modulation of facial motoneurone excitability by serotonin. Nature. 1980 Sep 25;287(5780):346–347. doi: 10.1038/287346a0. [DOI] [PubMed] [Google Scholar]
- White S. R., Neuman R. S. Facilitation of spinal motoneurone excitability by 5-hydroxytryptamine and noradrenaline. Brain Res. 1980 Apr 21;188(1):119–127. doi: 10.1016/0006-8993(80)90561-2. [DOI] [PubMed] [Google Scholar]
- Williams J. T., Colmers W. F., Pan Z. Z. Voltage- and ligand-activated inwardly rectifying currents in dorsal raphe neurons in vitro. J Neurosci. 1988 Sep;8(9):3499–3506. doi: 10.1523/JNEUROSCI.08-09-03499.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yanagihara K., Irisawa H. Inward current activated during hyperpolarization in the rabbit sinoatrial node cell. Pflugers Arch. 1980 May;385(1):11–19. doi: 10.1007/BF00583909. [DOI] [PubMed] [Google Scholar]
- Zhang L., Krnjević K. Apamin depresses selectively the after-hyperpolarization of cat spinal motoneurons. Neurosci Lett. 1987 Feb 10;74(1):58–62. doi: 10.1016/0304-3940(87)90051-6. [DOI] [PubMed] [Google Scholar]


