Abstract
Biology uses many signaling mechanisms. Among them, calcium and membrane potential are two prominent mediators for cellular signaling. TRPM4 and TRPM5, two calcium-activated monovalent cation-selective ion channels, offer a direct linkage between these two signals. Their activities convert a rise in the intracellular calcium level—a chemical signal—into depolarization of membrane potential—an electrical signal. Interestingly, membrane depolarization can in turn alter the electrical driving force or membrane permeability for calcium entry, hence offers feedback mechanisms for regulating calcium signaling. By converging two powerful cellular signals, TRPM4 and TRPM5 can contribute to many fundamental biological processes including cardiovascular biology, immunology, insulin release, chemo-sensation, and others. Numerous mutations in TRPM4 are linked to human hereditary cardiac and skin diseases, whereas knocking out TRPM5 in mice abolishes perception of sweet, umami and bitter tastes. This review summarizes what are currently known about the signaling roles of these unique TRP channels, and what remain mysterious.
Keywords: calcium signaling, membrane potential, activation gating, PIP2, cardiac physiology, insulin release, immune response, skin disease
1. Introduction
Life is a balance between homeostasis (maintaining a stable biological state) and short- or long-term changes (1). To orchestrate this dynamic balance, communication between organs, tissues, cells, and molecules is essential. Among numerous means of biological communication, calcium ions stand out as a universal mediator for signaling (2, 3). Virtually all cell types utilize calcium ions to switch on and off gene translation, trigger protein conformational arrangements, drive cell contraction and migration, induce or terminate inter-molecular interactions, administrate trafficking and secretion, and so on. Specialized organelles and proteins have emerged over the course of evolution to detect, capture, store, and release calcium ions. Calcium signaling works at all scales, from the systematic level to the subcellular and molecular levels. Membrane potential, as a product of asymmetrical ion distribution across biological membrane that is permeable to them, exists also in all living cells. It contributes to dynamic functions of excitable cells; for example, changes in membrane potential constitute the basic language for the nervous system, the master control of muscle contraction, and the switch for fertilization and immune response. In non-excitable cells and intracellular organelles, membrane potential plays a crucial role in influencing a wide range of cellular physiology such as cross-membrane transport and ATP synthesis. Even single-cell organisms have evolved to utilize these two prominent signaling systems.
It should therefore come as no surprise that these two signaling systems in biology do not operate independently. Connecting them presents obvious and powerful advantages. Communication between calcium ions and membrane potential occurs in both directions. Negative membrane potential serves as a strong driving force for influx of divalent cations like calcium (e.g., through Orai/STIM channels), and a barrier for its outward transport across the plasma membrane (e.g., by sodium-calcium exchanger NCX). A similar situation applies to intracellular organelles (4). Membrane depolarization reduces the driving force for calcium influx; however, when reaching a sufficiently high level, it can open voltage-gated Cav channels to allow rapid calcium influx. Calcium ions in turn regulate membrane potential in several ways. Transmembrane calcium flux by itself can alter membrane potential if it is at a sufficiently high rate (such as that seen during a cardiac action potential). Calcium ions also switch on or inhibit the transmembrane flux of cations and anions to shift membrane potential, often rapidly and powerfully. Calcium can shift membrane potential in either direction, depolarization or hyperpolarization. Calcium ions hyperpolarize the membrane when they activate, either directly by binding or indirectly through calcium-binding proteins, calcium-activated chloride channels and several potassium channels including large-conductance potassium (BK) and small-conductance potassium (SK) channels. Calcium can also depolarize the cell membrane. This is achieved by activating the TRPM4 and TRPM5 channels*.
2. The CAN channels and their molecular cloning
The existence of calcium-activated channels with an elevated reversal potential has been known for a long time. Calcium-activated, depolarizing currents were observed in cardiomyocytes (5) (Figure 1A&B), Purkinje tissue (6, 7), neuroblastomas (8), and others. These currents are not carried by calcium per se, whose equilibrium potential is the most positive among ions relevant to electrophysiology and far from the reversal potential of the observed calcium-induced current, at near zero millivolt (9, 10). Based on the same rationale, the depolarizing currents also cannot be mediated by sodium alone but jointly by sodium and potassium under physiological conditions (Figure 1B). Therefore, the channels responsible for mediating these currents are nonselective among monovalent cations, and hence were referred to as CAN (calcium-activated nonselective) channels before their identities have been revealed. Depolarizing currents exhibiting features associated with the CAN channels have been observed broadly in many cell types, though they may appear to vary in certain properties; for example, some may be long-lasting whereas others transient (11). The calcium concentrations required to activate this type of currents were generally found to be in the low micromolar range.
Figure 1.
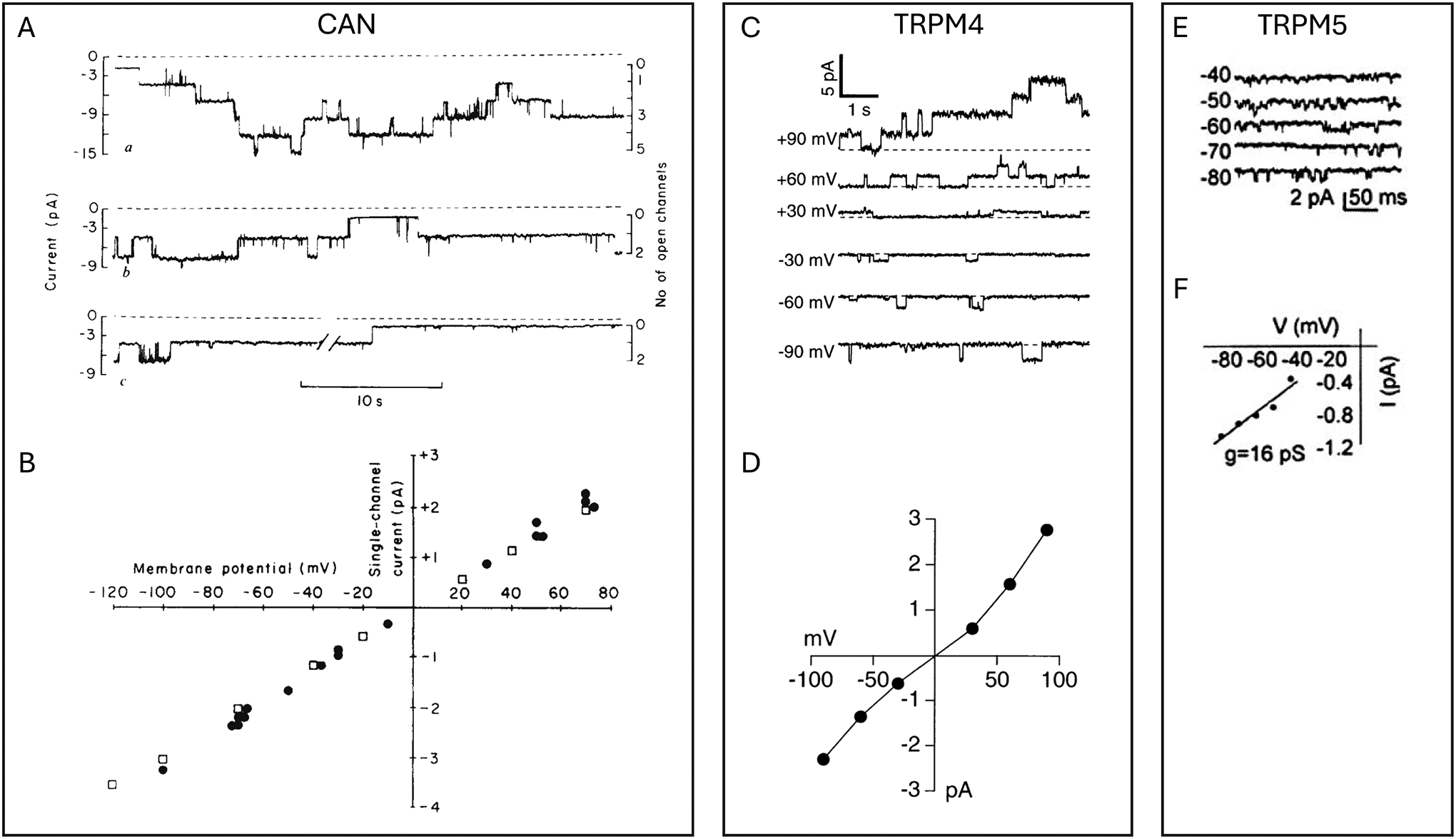
Representative current traces and I-V relationships of the CAN channels and cloned TRPM4 and TRPM5 channels. A&B, Example single-channel recordings of calcium-activated non-selective cation channels from cultured cardiac cells (A) and their I-V relationship (B). Recording voltage was −70 mV; intracellular calcium concentration was 6 μM (top), 1.5 μM (middle), and zero μM (bottom). Filled circles and open squares in B are data points with symmetrical sodium saline solutions and with intracellular potassium/extracellular sodium saline solutions, respectively, with reversal potentials around zero mV. Reproduced from Colquhoun et al. (5); used with permission under CC-BY license. C&D, TRPM4 single-channel currents activated by 0.3 μM calcium (C) and their I-V relationship with intracellular potassium/extracellular sodium saline solutions (D), with a reversal potential around zero mV. Reproduced from Launay et al. (13) with permission. E&F, TRPM5 currents activated by 12 μM calcium (E) and the I-V relationship (F). Reproduced from Liu and Liman (20) with permission.
One of the first ion channels with characteristics of the CAN channels was cloned from human cDNA libraries at the turn of the century (12). Shortly after, a longer form was identified (13) (Figure 1C&D). Based on sequence similarities, this new channel fits in the TRPM subfamily of the transient receptor potential (TRP) channel family, which is a large group of cation channels that exhibit the common feature as cellular sensors for diverse environmental stimuli as well as internal signaling (14). The short and long variants were named TRPM4a and TRPM4b, respectively. A variant shorter than TRPM4a was later found, and named TRPM4c (15). These variants differ in the length of their intracellular N-terminal segment. Rat TRPM4a and TRPM4b isoforms have been identified (16); only one mouse TRPM4 gene, corresponding to human TRPM4b, has been identified (15). TRPM4b (often referred to simply as TRPM4) is the predominantly studied form and the one discussed in this review. A close homolog of TRPM4 was cloned a little earlier (17), which was originally named MTR1 but later renamed TRPM5 due to its close resemblance in sequence and functional properties to TRPM4 (18) (Figure 1E&F). There are now eight members of the TRPM subfamily; among them only TRPM4 and TRPM5 are calcium-activated non-selective monovalent cation channels (19). TRPM4 exhibits a permeability sequence of Na+ ~ K+ > Cs+ > Li+, with no permeability to divalent cations (13). TRPM5 is equally permeable to Na+, K+, and Cs+ but not to Ca2+ (20). At very high concentrations, intracellular calcium ions actually inhibit TRPM4 current (21).
3. Structural features of TRPM4 and TRPM5
TRPM4 and TRPM5 were predicted by sequence analysis to be tetrameric channels like most other TRP channels. They have intracellularly located N and C termini, six transmembrane segments (S1 to S6), and a re-entry loop between the fifth (S5) and sixth (S6) transmembrane segments that contributes to the formation of the ion permeation pore and the ion selectivity filter. This topological prediction was confirmed when high-resolution structures of TRPM4 (22–26) and TRPM5 (27, 28) became available (Figure 2A). Homotetrameric channels are formed in a four-fold symmetric, domain-swapped architecture, meaning that the S1-S4 “voltage sensor like domain” of one subunit directly interacts with the S5-S6 pore-forming domain of a neighboring subunit. Most of the protein mass is located intracellularly, where potential interaction domains with regulatory proteins and small molecules have been proposed.
Figure 2.
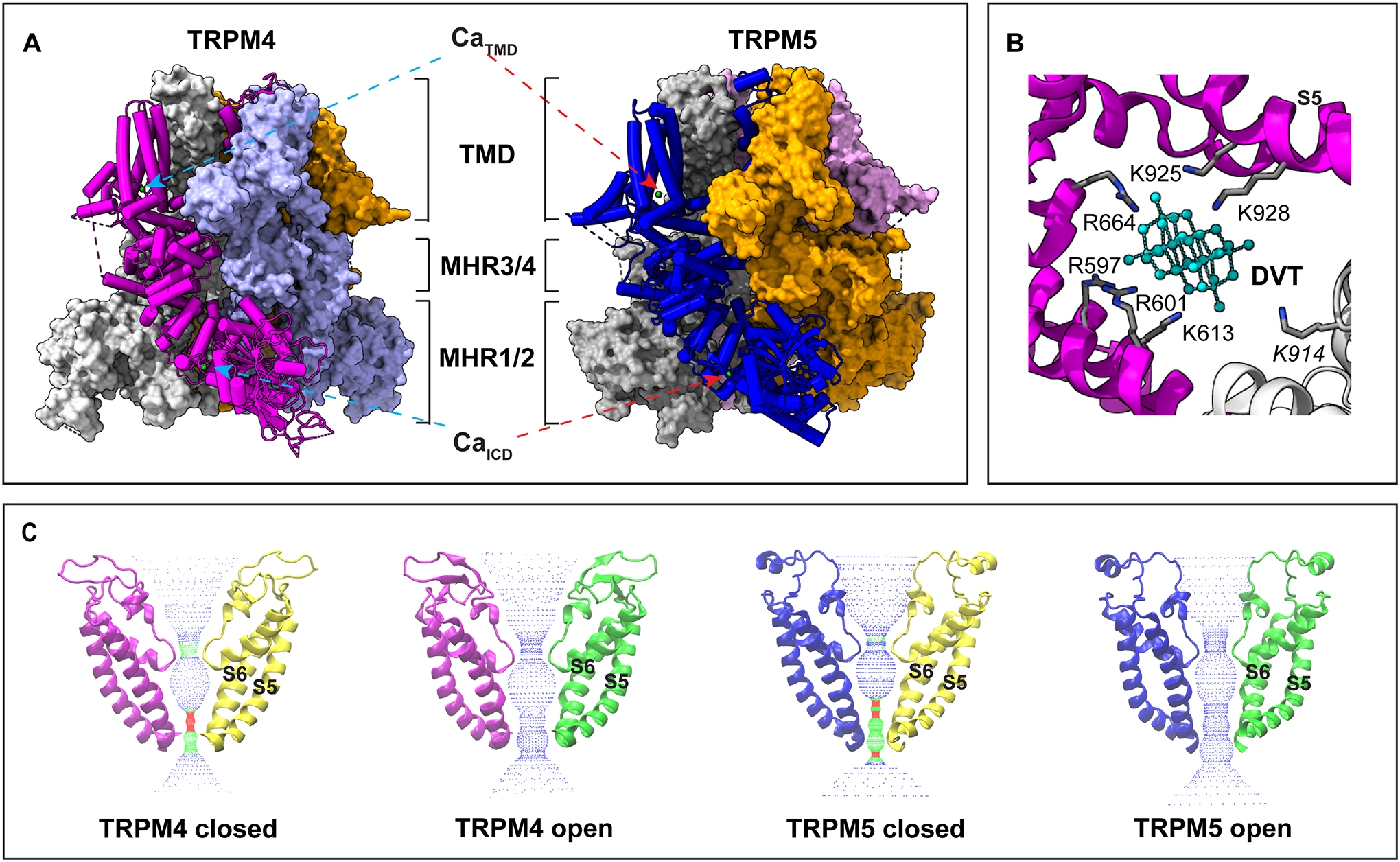
Structural features of TRPM4 and TRPM5 revealed by cryo-EM. A. Overall structure of TRPM4 (PDB ID 9B8Y, left panel) and TRPM5 (7MBQ, right panel). The light green spheres represent bound Ca2+ ions. TMD, transmembrane domain; ICD, intracellular domain; MHR, melastatin homology region. B. The binding pocket of decavanadate (DVT, cyan) in TRPM4 (9B8Y). Two neighboring subunits are colored in magenta and gray, respectively. Key positively charged residues in direct contact with DVT are highlighted. C. The closed states and open states of TRPM4 and TRPM5 (PDB ID from left to right: 6BQR, 9B8Y, 7MBR, and 7MBQ), highlighting the ion permeation pore. S5 and S6 represent the fifth and sixth transmembrane helices, respectively.
Multiple functional domains within the channel proteins have been identified. The intracellular N-terminal segment of TRPM4 contains an end-binding (EB) protein binding motif (SWIP) that mediate TRPM4-EB interaction and trafficking (29). There are also four melastatin homology regions (MHR1–4) in the N-terminal segment, with MHR1 and MHR2 forming an intact domain structure and MHR3 connecting MHR1/2 and MHR4 (23) (Figure 2A). MHR1/2 forms extensive interactions with MHR3 of the same subunit and MHR3 of an adjacent subunit. Within this domain, densities corresponding to ATP and decavanadate (one of three sites) have been observed (22, 23). The C-terminal region of TRPM4 consists of a coiled-coil helix with a segment extending nearly parallel to the membrane which then bends about 120° into vertical helices that are parallel to the vertical helices of the other subunits. The C-terminus has been previously proposed to contain modulation sites for PIP2, calmodulin binding, phosphorylation, and is important for tetrameric formation (30–32). In addition, functional studies suggested decavanadate regulates TRPM4 activation by binding to its C-terminal domain (33). Indeed, structural studies observed a second decavanadate binding site at the positively charged hinge of the C-terminal coiled-coil domain (23). Previous functional studies observed that the presence of decavanadate increased sensitivity of TRPM4 to ATP inhibition (33). Thus, the binding of decavanadate to the C-terminal domain is consistent with this site being involved in the positive modulation effects as it should not competitively antagonize ATP binding to the N-terminal site. A third decavanadate binding site, at the interface between the intracellular domain and transmembrane domain, was found in a new TRPM4 structure when the protein samples were prepared at the physiological temperature (37°C) (26)(Figure 2B). Only in the presence of this decavanadate and calcium ions a large fraction of the TRPM4 proteins kept at 37°C resided in the open state.
Directly relevant to the calcium-sensing function of TRPM4 and TRPM5 channels are the proposed calcium-binding sites (Figure 3). One set of calcium-binding sites were first identified when the cryo-EM structures of TRPM4 were solved (24). Located within the membrane-spanning domain formed by the S1-S4 segments and close to the intracellular side of the membrane, these calcium-binding sites stood out as they were empty in the absence of divalent cations but were occupied by a density resembling a calcium ion when 5 mM calcium were present during sample preparation (24) (Figure 3A&B). Each of the transmembrane calcium-binding site is surrounded by four acidic or polar residues; in human TRPM4, these residues are E828, Q831, N865, and D868. When a higher resolution structure of TRPM5 (at 2.3 Å) became available, a conserved binding site was also observed in each subunit, formed by E768, Q771, D797, and N794 of the zebrafish TRPM5 (27). Two water molecules could be identified in the binding site to join the four residues as calcium-coordinating ligands (27), hence satisfying the octahedral coordination chemistry for a calcium ion (34). Interestingly, equivalent cation binding sites are also found in other TRPM channels and even TRPC channels that are not particularly activated (but are positively regulated) by calcium ions (Figure 3A&B) (35, 36). A pathway connecting the transmembrane calcium binding site to the intracellular milieu for the entrance and exit of calcium ions can be envisaged in the TRPM4 structure, indicating that occupation of the binding site by calcium (and likely other cations under experimental conditions) can be a dynamic process (24). This calcium entrance pathway is seen in TRPM5 as well (27).
Figure 3.
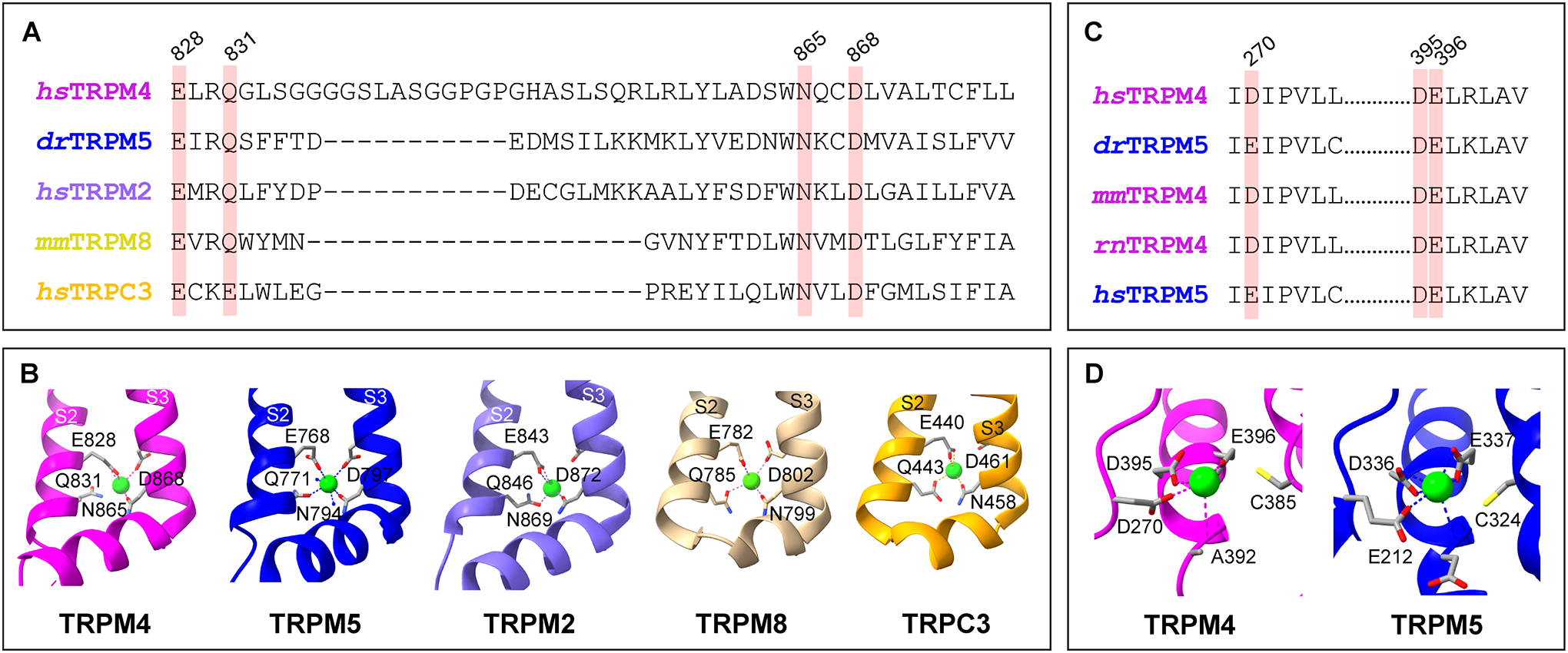
Calcium binding sites of representative TRP channels. A. Amino acid sequence alignment of the S2-S3 regions of human (hs) TRPM4, zebrafish (dr) TRPM5, human TRPM2, mouse (mm) TRPM8, and human TRPC3, with the key conserved residues for calcium binding highlighted. B. Transmembrane calcium binding pockets of hsTRPM4 (magenta, 9B8Y), drTRPM5 (deep blue, 7MBQ), hsTRPM2 (purple, 6PUS), mmTRPM8 (light yellow, 7WRB), and hsTRPC3 (golden, 7DXB). S2 and S3 represent the second and third transmembrane helices, respectively. C. Amino acid sequence alignment of the intracellular N-terminal calcium binding sites of TRPM4 and TRPM5 of several species, with the key conserved residues highlighted. D. Intracellular calcium binding pockets of TRPM4 (magenta, 9B8Y) and TRPM5 (deep blue, 7MBQ). The light green spheres represent bound Ca2+ ions. Amino acid numbers are for human TRPM4.
The TRPM5 structures offer exciting new insights into the Ca2+ activation mechanism (27). Unlike all the previously available TRPM4 structures, the Ca2+-bound TRPM5 structure is thought to represent an open state, with the hydrophobic gate at lower S6 widened substantially compared to the apo (closed) state. An allosterically coupled regulatory site for Ca2+ is also identified in each subunit. This second calcium binding site is located intracellularly at the interface between the MHR1/2 and MHR3/4 domains (Figure 3C&D); binding of Ca2+ to this site causes local conformational rearrangements of the MHR domains. Apparently, the effect of Ca2+ binding to the intracellular site can be felt at the transmembrane Ca2+-binding site some 70 Å away, yielding a substantially increased apparent binding affinity and positive allosteric regulation for channel activation.
It turns out that TRPM4 also persists an intracellular Ca2+-binding site equivalent to that of TRPM5, at the interface between MHR1/2 and MHR3/4 (26) (Figure 3C&D). This site could be observed when the protein samples were first warmed to physiological temperatures before rapid freezing. To form this site, there are substantial rearrangements in MHR1/2 and MHR3/4 from the closed state structure. The observations of two Ca2+-binding sites and substantial structural changes in the intracellular domain suggest that activation gating of TRPM4 and TRPM5 may share similar structural mechanisms involving large conformational rearrangements. Interestingly, TRPC channels contain one or two intracellular calcium binding sites; calcium binding to these sites allosterically regulates channel activities (36).
Like many cation channels (37), TRPM4 and TRPM5 have an ion permeation pore whose architecture resembles two hourglasses fused together—the hourglass on the top is formed by the re-entry loops from the four subunits surrounding the pore in a symmetric arrangement; the hourglass on the bottom is formed by the S6 segments also in a symmetric arrangement (Figure 2C). A hydrophobic residue in S6 (I1040 in TRPM4; I966 in TRPM5) is located at the neck of the bottom hourglass. Activation gating involves widening of this restriction, as can be visualized by comparing the apo and calcium-bound TRPM5 structures (27). A hereditary skin disease mutation at this point (I1040T), to be discussed later, makes the TRPM4 channel easier to open. At peripheral positions the pore is surrounded by the S5 segments, which are in turn pinched by the S4-S5 linkers that extend to the transmembrane calcium-binding pocket in each subunit. The post-S6 segment forms a helical structure underneath the S4-S5 linker. Named TRP helix, this segment has a highly conserved sequence among TRP channels (38). The TRP helix of TRPM4 and TRPM5 (and many other TRP channels) is highly sensitive to structural perturbations such as those produced by mutations, suggesting a steady role in supporting the conformation stability of the pore and even a dynamic role in transducing stimuli to the activation gate (39).
The top hourglass structure is where ion selection mainly occurs. A short segment from each subunit forms the narrowing (the hourglass neck) that is the ion selectivity filter (Figure 2C). Residues of the TRPM4 and TRPM5 ion selectivity filters point their sidechain inwards to directly interact with permeant ions. For highly potassium-selective channels, the ion selectivity filter residues bury their sidechain inside and form interactions with surrounding residues to stabilize the pore architecture while exposing their backbone oxygen atom for interaction with permeant potassium ions (40). Above the selectivity filter, a gradually widening outer pore entrance helps funnel permeant ions into the ion selectivity filter. Acidic residues lining the surface of this entrance likely attract cations into this space via electrostatic forces. TRPM4 and TRPM5 of various species normally exhibit a single-channel slope or chord conductance in the 16-to-25 pS range (13, 20, 41, 42).
4. Activation and desensitization
Activation of TRPM4 and TRPM5 exhibits clear calcium-concentration dependence (Figure 4 A&B). The Hill slope of their calcium concentration dependence curve is steeper than one, suggesting the likelihood that calcium ions bind to all four subunits cooperatively to promote channel activation (20, 43, 44). In excised inside-out patches, TRPM4 channels have been generally found to exhibit an increase in activity when the intracellular calcium concentration reaches 100 μM or higher (43, 44). Whole-cell recordings showed a much higher calcium sensitivity, but the obtainable current amplitudes did not seem to be proportional to what were recorded from cell-free membrane patches (43, 44). The high concentrations of calcium ions needed for observing channel activation from cell-free patches is concerning. A speculative view is that perhaps TRPM4 channels are juxtaposed next to calcium entry or release sites, where the local calcium concentration might reach a level sufficiently high to induce meaningful TRPM4 activities under physiological conditions. Alternatively, the channels been recorded in expression systems might lack certain cofactor(s), or perhaps they have quickly gone through functional changes such as desensitization, which has been demonstrated to reduce the channel’s calcium sensitivity (30, 44) (Figure 4A–C).
Figure 4.
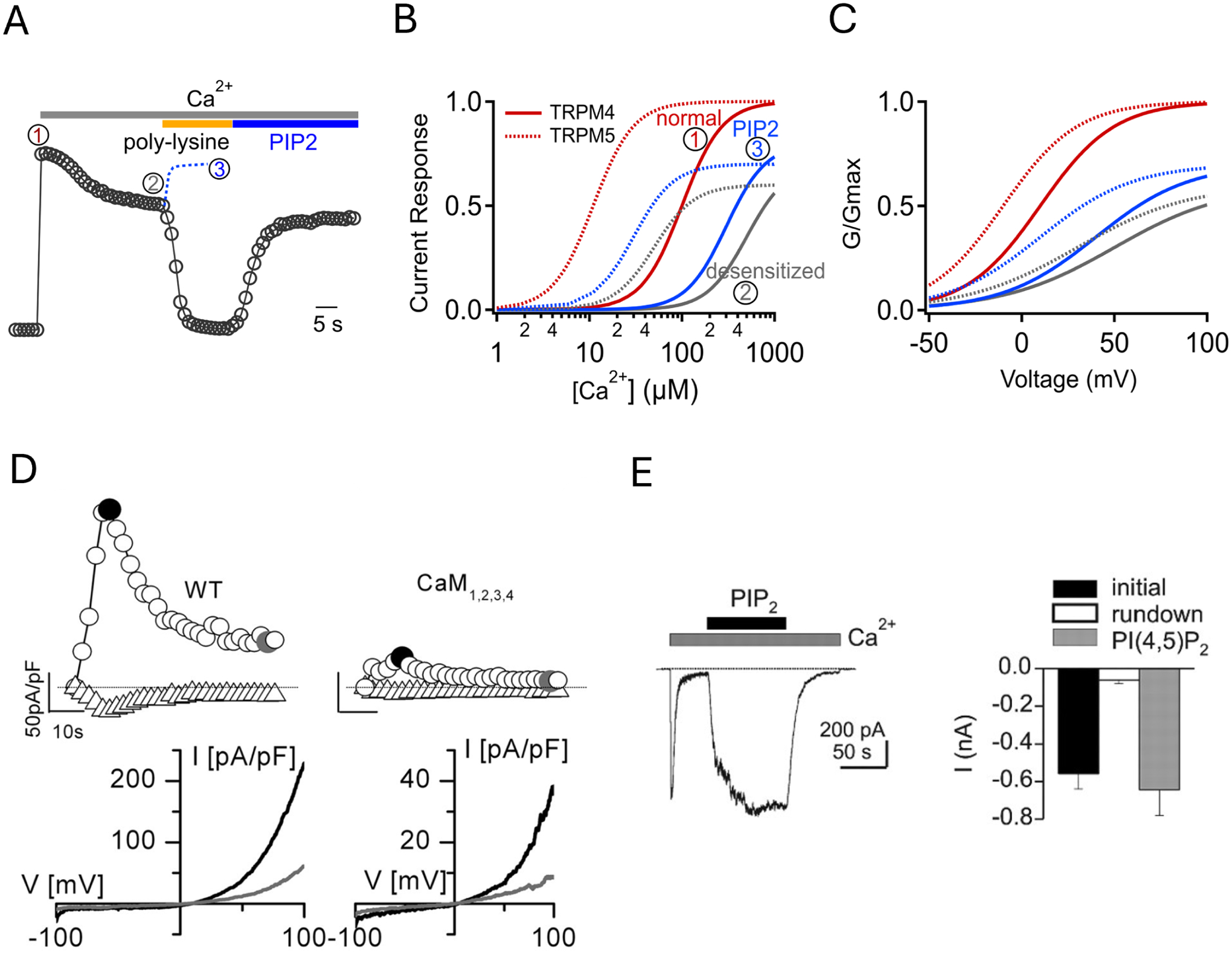
Activation and regulation of TRPM4 and TRPM5. A. A representative current trace of human TRPM4 recorded at 80 mV from an inside-out patch in response to 3 mM Ca2+, 10 μg/mL poly-lysine, and 50 μM diC8-PIP2. Dotted blue trace is from another recording when diC8-PIP2 was applied before complete desensitization due to application of poly-lysine. Labels 1, 2, and 3 represent initial peak current, spontaneously desensitized current, and current recovered by PIP2, respectively. B. Simulated calcium-dependent activation of TRPM4 (solid curves) and TRPM5 (dash curves) at time point 1 (red), 2 (gray), and 3 (blue) in panel A. Naïve TRPM4 and TRPM5 channels have an apparent EC50 value in 100 μM and 10 μM, respectively, in inside-out patch recordings. C. Voltage-dependent activation of TRPM4 and TRPM5, using the same color scheme as B. D. Example mouse TRPM4 current traces in response to 100 μM Ca2+ over time at −100 mV and 100 mV (top left); co-expressing a calcium-insensitive calmodulin mutant CAM1,2,3,4 reduces the current amplitude (top right). The bottom panels show the corresponding I-V relationships. Reproduced with permission from Nilius et al. (2005) JBC. E. An example trace at −80 mV (left) and summary (right) of mouse TRPM4 current activated by 100 μM Ca2+ and recovery by 10 μM diC8-PIP2. Reproduced from Zhang et al. (44) with permission under CC-BY license.
What might be the source of calcium increase that activates TRPM4 and TRPM5 under physiological conditions? While in most cases the question remains to be answered, sceneries such as release from intracellular calcium store and influx from extracellular milieu have been considered. In taste cells, an increase in calcium concentration is likely due to release from the intracellular store as a downstream event of the phospholipase C (PLC) signaling pathway (45). A major task for future research is to identify the calcium source in various tissues and cell types where TRPM4 and TRPM5 activities have been observed. The functional roles of the two sets of calcium binding sites remain to be determined, as well as how calcium binding leads to channel activation.
At non-physiologically high calcium concentrations, TRPM4 currents exhibit a transient peak upon both calcium application and removal (46). The presence of these transient current peaks indicates the possible existence of a calcium-dependent inhibition process and the likelihood that recovery from this inhibition upon removal of calcium occurs faster than channel deactivation upon calcium removal. How much this inhibition process helps shape the calcium response of TRPM4 at physiological concentrations remains to be determined. At even higher calcium concentrations (>1 mM), which would reduce the amplitude of the steady state current through apparent pore block (21), an additional mysterious third peak emerges after the second peak (47). This peak diminishes as the channel desensitizes, indicating a likely shared mechanism.
As mentioned above, desensitization of TRPM4 and TRPM5 can substantially alter their calcium responses. Both the current amplitude and the kinetic properties change upon desensitization. Desensitized channels respond to a step increase in intracellular calcium concentration much slower than naïve channels but deactivate much faster upon calcium removal (44). These changes in kinetic properties may contribute or even underlie the observed decline in stable current amplitude during channel desensitization. The third transient peak mentioned above also drops in amplitude, and even disappears, upon extensive desensitization (47). TRPM5 channels of mammalian species desensitize much faster than zebrafish TRPM5 (28). While desensitization of TRPM4 and TRPM5 appears to be a complex process, one well-established cause for desensitization is depletion of PIP2. This will be discussed in detail later. Treatment with hydrogen peroxide can also remove TRPM4 desensitization (48).
So far, intracellular calcium is the only known biological activator for TRPM4 and TRPM5. It remains an open question whether other biologically relevant molecules can activate these channels. TRPM5 is not activated by 1-oleoyl-2-acetyl-sn-gylcerol (OAG), an analog for diacylglycerol (DAG), at 100 μM, or IP3 at 10 μM (20, 44). While PIP2 plays an important role in maintaining calcium-induced activation, by itself PIP2 does not activate TRPM4 or TRPM5.
5. Channel regulation mechanisms
Like most ion channels, TRPM4 and TRPM5 are dynamically regulated under physiological and pathological conditions. Rich information on regulation of TRPM4 and TRPM5 activities has been accumulated through extensive research. These channels are known to be sensitive to regulatory lipids such as PIP2, calmodulin, temperature changes, and even the transmembrane voltage their activity regulates. Many agonists and antagonists have been identified and studied over the years. For example, extracellular protons have two types of inhibitory effects on TRPM5: they reduce current by an apparent blockade mechanism and an inactivation mechanism (49). These two effects are related but distinct; mutations to an acidic residue (E830) in the S3-S4 linker and a histidine residue in the pre-S6 loop (H934) of the mouse TRPM5 both simultaneously reduce proton-dependent block and inactivation, whereas a mutation to another histidine residue in the after-S5 loop spared acid block but caused significant recovery from acid-enhanced inactivation. Here we summarize what are known about channel regulation mediated by the membrane potential, calmodulin, PIP2, ATP, protein kinase C (PKC), polyamine and temperature.
a. Membrane potential
While TRPM4 and TRPM5 serve as calcium-activated effectors through altering the electrical potential of the plasma membrane, their calcium sensitivity is in turn regulated by membrane potential. At a constant intracellular calcium concentration, the amplitude of TRPM4 and TRPM5 currents recorded from a cell or macro-patch increases super-linearly upon membrane depolarization (Figure 4C). This change can be observed within the physiologically relevant voltage range. For TRPM4, the half-activation voltage, V1/2, defined as the voltage where activity reaches 50% of its peak level, is measured (by fitting the normalized current-voltage relationship to a Boltzmann function) at room temperature to be 25 mV when 100 μM calcium was present (15). The voltage-dependent activation curve shifts leftward at higher calcium concentrations or temperatures. For TRPM5, the V1/2 value appears to be even more negative, which reflects its higher calcium sensitivity (20, 43). Hence, a physiological role can be predicted for the dynamic regulatory process by voltage. The single-channel currents of cloned TRPM4 and TRPM5 channels exhibit a near ohmic dependence on voltage, at a conductance level of about 20 pS (13, 15, 20, 41, 42). The super-linear increase in macroscopic current amplitude in response to depolarization is therefore due to an increase in channel open probability. This voltage dependence produces strong outward rectification of the macroscopic current (15). Voltage sensitivity of TRPM4 and TRPM5 channels are rather weak when compared to the Kv and Nav channels involved in action potential firing. The effective gating charge estimated from published data is less than one elementary charge (e0), whereas for Kv channels the value is 13 to 16 e0 (50).
The voltage-sensing mechanism of TRPM4 and TRPM5 is still unknown. Given the weak voltage sensitivity, the voltage sensor can reside in many possible channel structures (51). Many other TRP channels exhibit comparable voltage sensitivity. For TRPV1, the voltage sensing mechanism involves extracellular structures (52); for TRPM8, a charged residue near the bottom of the fourth transmembrane segment S4 serves as the voltage sensor (53). Voltage regulation serves as a positive feedback mechanism for TRPM4 and TRPM5, as the channel activity increases upon depolarization.
b. Calmodulin
Given the precedence that calcium activation of the SK channels is mediated by the calcium-sensor calmodulin instead of direct binding of calcium to the channel protein (54), it was attempting to envision that TRPM4 and TRPM5 could be activated in a similar manner. This however turned out to be unlikely (30, 41). Nonetheless, calmodulin was found to play a role in dynamically regulating TRPM4 activity (30, 55) (Figure 4D). Introducing a mutant version of calmodulin that cannot bind calcium (CaM1,2,3,4) substantially reduced, but however did not eliminate, TRPM4 current. Supplying recombinant calmodulin to cell-free patches could slow down channel desensitization and reduce its extent (30). Nonetheless, divalent cations Mn2+, Ni2+, and Co2+ were found to comparably potentiate calcium induced TRPM4 currents, even though the latter two have no effect on calmodulin (55). Sequence analysis suggested several candidate calmodulin binding sites in the N- and C-terminal of TRPM4; deletions of these sequences led to reduction of channel current (30). There is currently no TRPM4-calmodulin complex structure.
Multiple lines of evidence suggest that TRPM5 is not activated or modulated by calmodulin (41): (1) Ba2+ ions alone at concentrations as high as 10 mM, which is expected to stimulate calmodulin, could not activate TRPM5; (2) the calmodulin inhibitor calmidazolium did not alter TRPM5 activity; (3) adding a calmodulin-binding peptide, the myosin light chain kinase (MLCK) peptide, also failed to exert an effect.
c. PIP2
Many proteins in the plasma membrane exhibit strong sensitivity to PIP2. Multiple lines of evidence support the idea that PIP2 plays a regulatory role for TRPM4. After desensitization, direct application of PIP2 to the membrane patch could partially recover TRPM4 channel current (Figure 4E); application of Mg2ATP in millimolar concentrations to desensitized channels could also partially revive the current (22, 30, 44). Application of PIP2 to mouse sinoatrial node cells promoted native TRPM4 channel activities (56). It is known that phosphatidylinositol phosphate kinases are stimulated by Mg2ATP. Consistent with this idea, the potentiation effect of ATP could be obtained only when Mg2+ was co-applied with ATP. Wortmannin, a lipid kinase inhibitor, at concentrations that block phosphatidylinositol 4-kinase (PI4K) and phosphatidylinositol 3-kinase (PI3K), inhibited the Mg2ATP-induced partial current recovery, whereas a PI3K-specific inhibitor LY294002 did not show any inhibitory effect (44). PLC inhibitor U73122 was found to slow down desensitization (31). Furthermore, application of the PIP2 scavenger poly-lysine could speed up desensitization and extend its impact on current amplitude (31, 44). Application of diC8-PIP2, a short-tail, more water-soluble derivative of PIP2, to inside-out patches could at least partially recover the TRPM4 current from desensitization (31, 44). PIP2 shifts voltage-dependent activation towards more negative voltages and dramatically increases the channel’s calcium sensitivity (Figure 4C). These experimental observations establish a link between PIP2 depletion and TRPM4 desensitization, indicating that association of PIP2 with TRPM4 stabilizes its open conformation.
TRPM5 also desensitizes in a PIP2-related manner. Extended application of calcium caused the calcium concentration-dependent activation curve to shift downward and rightward (20). Adding diC8-PIP2 to desensitized TRPM5 channels could enhance current response to calcium. As a result, the calcium concentration dependent activation curve exhibited a large uplift as well as a clear left shift (Figure 4B&C). However, TRPM5 desensitization appears to be a complex process. Desensitization of mammalian TRPM5 channels also exhibits calcium concentration dependence (28).
Locations of the PIP2-binding sites in TRPM4 and TRPM5 have not been found. Mutations of basic residues in the TRP domain of TRPM4 (and of TRPM8 and TRPV5) reduced PIP2 sensitivity and enhanced inhibition by PIP2 depletion (57). The Nilius group found TRP domain mutations did not have a dramatic effect on PIP2-dependent current recovery after desensitization, although the apparent sensitivity to PIP2 was decreased (31). Instead, a cluster of positive charges more distally located in the C-terminus of TRPM4 seemed to exhibit a larger influence on PIP2 binding; mutations of basic residues in this region decreased the apparent affinity for PIP2 and the activity of the channels.
Since many signaling processes affect PIP2 in the plasma membrane, the activity of TRPM4 and TRPM5 may be dynamically regulated as a result. Indeed, desensitization (or sensitization) to repetitive or sustained stimulation is a general feature of many TRP channels, for which PIP2 has been found to play an important role (58). It is therefore important to better understand how PIP2 interacts with these channels.
d. ATP, PKC and polyamine
Perfusing Mg2ATP to inside-out patches could increase TRPM4’s calcium sensitivity to partially reverse channel desensitization (30, 44), as mentioned earlier. The potentiating effect of Mg2ATP is thought to be indirect, through activation of a kinase that restores the level of membrane-bound PIP2 (44). In addition, phosphorylation is thought to potentiate TRPM4. It is proposed phosphorylation is mediated by PKC, as treating TRPM4-expression cells with phorbol 12-myristate 13-acetate (PMA) increased the current density (30). PKC was found to also stimulate TRPM4 trafficking and insertion into the plasma membrane, resulting in a boost of channel current (59).
Interestingly, ATP was found to reversibly inhibit TRPM4 current when applied at micromolar concentrations to the intracellular side without Mg2+; so did ADP, AMP and AMP-PNP but not GTP, UTP or CTP (21). When the cryo-EM structure of TRPM4 in complex with ATP was solved, ATP was observed to be bound to the lower tier of the intracellular N-terminal domain, between adjacent subunits; a tetrameric channel complex can accommodate four ATP molecules (22). ATP inhibition is therefore likely achieved through an allosteric mechanism. The channel protein interacts with the adenine moiety of ATP via three aromatic residues (H160, W214, and Y228 in the mouse TRPM4) (22), explaining the poor selectivity for channel inhibition among ATP, ADP, and AMP. These residues are not conserved in TRPM5, explaining the observation that ATP does not inhibit TRPM5 (43). The triphosphate group of ATP interacts with arginine residues. Binding of ATP causes a local separation between subunits that can be discerned at the nucleotide binding domain. The intracellular channel structure containing the ATP-binding site moves over a large distance in the open state of TRPM4; it is thought that the bound ATP prevents the activation transition in the pore by disrupting the coupling process (26).
Intracellular polyamine also inhibits TRPM4 and TRPM5, with an IC50 value in the tens of micromolar range (21, 43). Polyamine inhibits inward rectifier potassium channels by binding inside the pore to block ion permeation (60). It is likely that the same mechanism underlies polyamine inhibition of TRPM4 and TRPM5 channels; this however remains to be experimentally tested.
e. Temperature
Several TRP channels are known for their outstanding heat sensitivity in activation gating, which allows them to play a role in detecting changes in temperature (38, 61, 62). Sensitivity of TRPM4 and TRPM5 activation to temperature has been noticed. Their Q10 values, measured from current amplitude changes between 15°C and 35°C, are around 10 (63). It is thought that the sensitivity of TRPM5 activation gating to temperature plays a role in sensory physiology, allowing a better appreciation of the sweet taste (63). The structural underpinning for TRPM4’s temperature sensitivity has been recently revealed (26). It was found that the intracellular domain of TRPM4 resides in drastically different conformations at below room temperature and at 37°C. In the cryo-EM structure obtained from samples first heated to 37°C, the lower part of the channel pore, a.k.a. the neck of the second hourglass, was seen to be wider, likely representing an open state.
6. TRPM4 and TRPM5 pharmacology
The available pharmacological tools for TRPM4 and TRPM5 have been lacking for a long time, which limited biophysical and physiological investigations. Most of the reported modulators lack specificity or high efficacy. The situation however is improving, as the obvious pharmaceutical opportunities these ion channels present attract research activities in both academia and industry looking for effective natural or synthesized molecules. One can anticipate that these activities will yield highly efficacious and selective agonists and antagonists in the very near future. Here we review briefly what pharmacological tools are currently available (Table 1).
Table 1.
TRPM4 modulators
| Molecule | EC50/IC50 (μM) | References |
|---|---|---|
| Activator / Potentiator | ||
| Intracellular Ca2+ | 0.3–1000 | Colquhoun, 1981; Yellen, 1982; Launay, 2002; Nilius, 2003; Nilius, 2004; Nilius, 2005; Ullrich, 2005; Yarishkin, 2008; Sala-Rabanal, 2012; Amarouch, 2013; Yamaguchi, 2014; Winkler, 2017; Autzen, 2018; Hu, 2024 |
| Intracellular Co2+ with Ca2+ | 107 | Yamaguchi, 2014 |
| Intracellular Mn2+ with Ca2+ | ND | Yamaguchi, 2014 |
| Intracellular Ni2+ with Ca2+ | ND | Yamaguchi, 2014 |
| PIP2 with Ca2+ | 4–42.2 | Zhang, 2005; Nilius, 2006; Yamaguchi, 2014; Guo, 2017 |
| BTP2 | 0.008 | Takezawa, 2006 |
| Decavanadate with Ca2+ | 1.9 | Nilius, 2004; Winkler, 2017; Hu, 2024 |
| Mg2ATP | ND | Zhang, 2005; Nilius, 2006; Guo, 2017 |
| Inhibitor | ||
| Adenosine | 630 | Sturgess, 1986; Nilius, 2004 |
| AMP | 19 | Sturgess, 1986; Nilius, 2004 |
| ADP | 2.2 | Sturgess, 1986; Nilius, 2004 |
| ATP | 0.8–2.3 | Sturgess, 1986; Nilius, 2004; Guo, 2017; Arullampalam 2021; Hu, 2024 |
| AMP-PNP | 19 | Thorn, 1992; Nilius, 2004 |
| Spermine | 61 | Nilius, 2004 |
| Flufenamic acid | 2.8–5.5 | Korbmacher, 1995; Guinamard, 2006; Demion, 2007; Morita, 2007 |
| Clotrimazole | ND | Ullrich, 2005 |
| Glibenclamide | ND | Guinamard, 2006; Demion, 2007; Sala-Rabanal, 2012 |
| 9-phenanthrol | 10–30 | Grand, 2008; Gonzales, 2010 |
| CBA (Compound 5) | 0.7–1.8 | Ozhathil, 2018 |
| NBA (Compound 6) | 0.1–0.2 | Ozhathil, 2018; Arullampalam, 2021 |
ND, not determined.
Intracellular calcium remains to be the only known physiological activator for TRPM4 and TRPM5. Its known pharmacological properties and binding sites have been discussed in Section 4. Several molecules are found to potentiate TRPM4 and/or TRPM5 activities. Both PIP2 and Mg2ATP are discussed previously. A highly negatively charged vanadate oligomer, decavanadate, also mentioned earlier, was found to potentiate TRPM4 activities. BTP2, a 3,5-Bis(trifluoromethyl)pyrazole derivative and an inhibitor of CRAC channels, was found to potentiate TRPM4 (64). While many divalent cations were found to be either ineffective or inhibitory of the TRPM4 current, Co2+, Mn2+ and Ni2+ applied intracellularly exhibited a potentiation effect but only in the presence of intracellular Ca2+ (55). Arachidonic acid, a polyunsaturated omega-6 fatty acid precursor for leukotrienes, prostaglandins, and thromboxanes, was reported to potentiate TRPM5 currents when applied from the extracellular side (65).
Adenosine and it phosphate derivatives such as ATP, ADP, AMP, AMP-PNP are all found to inhibit TRPM4, so did polyamines such as spermine (21), as discussed earlier. Flufenamic acid and chlotrimazole are non-specific inhibitors for TRPM4 and TRPM5, apparently by modifying their gating activities (43, 56). Flufenamic acid, which inhibits TRPM4 with an IC50 of 3-to-6 micromole, also affects many other channels including GABAA and CLC-1 (66). A hydroxyl derivative of the tricyclic compound phenanthrene, 9-phenanthrol, was identified as a TRPM4 inhibitor; it selects TRPM4 over TRPM5 and other TRP channels such as TRPC3, TRPC6, TRPM7 (67). The 9-phenanthrol compound is structurally similar to MPB-104 (5-butyl-7-chloro-6-hydroxybenzo[c]-quinolizinium chloride) that inhibits TRPM4 with an IC50 of 20 micromole but also activates CFTR (67). Still, 9-phenanthrol was found to activate the intermediate conductance calcium-activated potassium channel at the low micromolar range (68) and inhibit several potassium channels (69). Several more favorable TRPM4 inhibitory compounds were developed based on 9-phenanthrol, among them CBA and NBA have gained popularity (70). They exhibited better efficacy than 9-phenanthrol; CBA appeared to exhibit some species preferences, inhibiting only human TRPM4 but not mouse TRPM4, whereas NBA inhibited both human and mouse TRPM4 channels (71). Extracellular protons inhibit TRPM5 through two mechanisms: they promote channel inactivation and block current through the open channel (49). Extracellular zinc ions also inhibit TRPM5, by binding to the pore loop (72). N’-(3,4-dimethoxybenzylidene)-2-(naphthalen-1-yl)acetohydrazide (NDNA) inhibits TRPM5 by binding between the S1-S4 domain and the pore domain (27).
Following an observation that the intracellular ATP regulated swelling of rat brain astrocytes (73), the Simard group proposed that the effect was carried out by ATP inhibition of the sulfonylurea receptor type 1 (SUR1) that associates with TRPM4 (74). This would allow sulfonylurea drugs like glibenclamide and tolbutamide to regulate TRPM4 activity through competitive binding to SUR1 (73). Glibenclamide, an antidiabetic medication, binds to ATP binding cassette (ABC) proteins such as CFTR and SUR1. It was found to strongly inhibit TRPM4 currents at the 10-to-100 micromolar range; however, as expected, it also affects ABC proteins such as CFTR, and KATP channels due to its association with SUR1 (56). Direct association of SUR1 and TRPM4 in COS and HEK cells was reported based on evidence from co-immunoprecipitation and fluorescence resonance energy transfer (FRET) (75). The Simard group further reported that aquaporin AQP4 forms a heteromultimeric complex with TRPM4 and SUR1, hence potentially linking calcium signaling to water transport and osmotic regulation (76). However, the Nichols group reported that, at least in COS cells, there was no detectable evidence that over-expressed SUR1 and TRPM4 associated or directly interacted (77). They also failed to observe an inhibitory effect of glibenclamide or tolbutamide after the exogenously expressed TRPM4 channels went through spontaneous desensitization. It is unclear what might have caused the discrepancy. No TRPM4/SUR1 complex structure has been reported.
7. Tissue and organ distribution
The distribution of calcium-sensing TRPM4 and TRPM5 channels among tissues and organs carries important information on their potential functional contributions to physiology. Indeed, conversion of intracellular calcium signaling to changes in membrane potential may powerfully impact cellular function. It is therefore anticipated that functional expression of TRPM4 and TRPM5 would have a substantial contribution to the physiological roles of its host cells. The human disease-causing gain-of-function and loss-of-function TRPM4 mutations, to be discussed later in Section 9, offer supportive cases for this view. A general observation from the published studies so far is that TRPM4 has a broad distribution among tissues and cell types; in comparison, TRPM5 has a more restricted expression pattern. The wide distribution of TRPM4 and TRPM5 channels is consistent with observations on the CAN channels from the pre-cloning age.
The distribution of TRPM4 and TRPM5 has been investigated at the mRNA and protein levels, using PCR, Northern blot, Western blot (WB), immunocytochemistry (ICC), and immunofluorescence (IF) staining of tissue samples. Distribution of GFP-tagged proteins among tissues in transgenic animals was also documented. These methods have different specificities and sensitivities. Results from different studies are sometimes not consistent, which presents challenges to come up with an integrated picture. Functional confirmation of native TRPM4 and TRPM5 channel expression using patch-clamp recordings would offer evidence from a different angle and indeed has been used in many studies. However, the lack of highly selective and effective activators and inhibitors presents limitations. It is therefore likely that the issue of native expression will be revisited in future studies of TRPM4 and TRPM5 in certain tissues or organs. In this section, we give a brief overview of the native expression of TRPM4 and TRPM5 in noticeable organs and tissues. Specific observations will be discussed in the next section when individual physiological functions are examined.
TRPM4: When the short (TRPM4a) and long (TRPM4b) versions of the channel were first discovered, it was noticed that mRNAs encoding them could be detected in a variety of organs, including heart, kidney, liver, pancreas, placenta, skeletal muscle, thymus, spleen, prostate, colon but was absent in leukocytes (12, 13). Subsequently, many studies investigated the expression pattern of TRPM4. For systematic surveys, the McNulty group at GlaxoSmithKline conducted a study of TRPM4 (and other TRPM channels) using PCR (78), and the Oh group at Seoul National University conducted a comparative study of the mRNA levels of TRPM4 (and all other TRP channels) in different tissues (79). Results from these studies confirmed the widespread expression pattern, which is substantiated by a large body of studies in specific tissues or organs (see Table 2 and Figure 5). It should be noted that, when various reports are examined, there could be differences in details such as the relative signal intensities among tissue and organ types.
Table 2.
TRPM4 expression in mammalian species
| Organ/Tissue | Species | References | Methods |
|---|---|---|---|
| Cardiovascular system | |||
| heart | human | Launay, 2002, Fonfria, 2006; Kruse, 2009; Dragun, 2019; Frede, 2020; Feng, 2021 | RT-PCR, qPCR, Northern blot, WB, ICC, IHC, IF, patch clamp |
| rat | Guinamard, 2006; Teruyama, 2011; Piao, 2015; Son, 2016; Frede, 2020 | ||
| mouse | Demion, 2007; Jang, 2012; Simard, 2012; Kecskés, 2015; Gueffier, 2017; Medert, 2020; Frede, 2020; Guo, 2021; Hedon, 2021; Medert, 2021; Arullampalam, 2023 | ||
| dog | Dienes, 2021 | ||
| rabbit | Hof, 2016 | ||
| blood vessel | human | Thilo, 2011; Echeverría, 2015 | |
| rat | Earley, 2004; Yang, 2006; Gerzanich, 2009; Crnich, 2010; Li, 2014; Ding, 2017; Gong, 2019; Csipo, 2022 | ||
| mouse | Ali, 2021; Yamasaki, 2023 | ||
| Immune system | |||
| bone marrow | human | Fonfria, 2006 | qPCR, Northern blot, IHC, IP, WB, patch clamp |
| spleen | Launay, 2002; Fonfria, 2006 | ||
| thymus | Launay, 2002; Launay, 2004 | ||
| blood mononuclear cell | Fonfria, 2006; Serafini, 2012; Malhotra, 2013; Morita, 2020; Wang, 2020; Çakir, 2021; Ozcan, 2021 | ||
| macrophage | Fonfria, 2006 | ||
| T cell | mouse | Launay, 2004; Vennekens, 2007; Weber, 2010 | |
| mast cell | Vennekens, 2007 | ||
| macrophage | Serafini, 2012 | ||
| dendritic cell | Barbet, 2008 | ||
| Nervous system | |||
| cerebrum | human | Fonfria, 2006; Schattling, 2012; Tosun, 2013; Mehta, 2015; Hristov, 2016 | RT-PCR, qPCR, WB, ICC, IHC, IF, IP, in situ hybridization, FRET, patch clamp |
| rat | Li, 2010; Teruyama, 2011; Tosun, 2013; Huang, 2015; Huang, 2016; Chen, 2019; Ma, 2023 | ||
| mouse | Crowder, 2007; Jang, 2012; Schattling, 2012; Riquelme, 2018; Yan, 2020; Riquelme, 2021 | ||
| cerebellum | mouse | Kim, 2013 | |
| brainstem | rat | Koizumi, 2018 | |
| mouse | Koizumi, 2018; Li, 2021 | ||
| pituitary | human | Fonfria, 2006 | |
| spinal cord | human | Schattling, 2012 | |
| rat | Woo, 2013; Turtle, 2019 | ||
| mouse | Schattling, 2012; Vandewauw, 2013; Bianchi, 2018; Yao, 2018; Tsymbalyuk, 2021 | ||
| Urogenital system | |||
| bladder | human | Hristov, 2016 | RT-PCR, qPCR, Northern blot, WB, ICC, IHC, IF, patch clamp |
| rat | Smith, 2013a | ||
| mouse | Yu, 2011; Kullmann, 2018 | ||
| guinea pig | Smith, 2013b; Maxwell, 2021 | ||
| kidney | human | Launay, 2002; Fonfria, 2006 | |
| rat | Teruyama, 2011; Dusmez, 2014; Piao, 2015 | ||
| mouse | Jang, 2012 | ||
| placenta | human | Launay, 2002; Fonfria, 2006 | |
| uterus | human | Fonfria, 2006; De Clercq, 2015; Persoons, 2018 | |
| mouse | De Clercq, 2017 | ||
| prostate | human | Fonfria, 2006; Hristov, 2016 | |
| rat | Wang, 2007 | ||
| testis | rat | Li, 2010; Jang, 2012 | |
| Respiratory system | |||
| lung | human | Fonfria, 2006 | RT-PCR, qPCR, IHC |
| rat | Piao, 2015 | ||
| mouse | Jang, 2012 | ||
| Digestive system | |||
| liver | human | Launay, 2002; Fonfria, 2006 | RT-PCR, qPCR, Northern blot |
| rat | Piao, 2015 | ||
| stomach | human | Fonfria, 2006 | |
| intestine | human | Fonfria, 2006 | |
| Sensory system | |||
| inner ear | mouse | Takumida, 2009; Sukuraba, 2014 | RT-PCR, qPCR, WB, IHC, IF, patch clamp |
| eye | human | Yarishkin, 2022 (trabecular meshwork cells) | |
| mouse | Choi, 2015 (optic nerve head) | ||
| tongue | rat | Teruyama, 2011 | |
| Others | |||
| pancreas | human | Launay, 2002; Fonfria, 2006; Marigo, 2009 | RT-PCR, qPCR, Northern blot, WB, IP, ICC, IHC, patch clamp |
| rat | Cheng, 2007; Marigo, 2009; Piao, 2015, Ma, 2017 | ||
| mouse | Marigo, 2009; Nelson, 2011; Diszházi, 2021 | ||
| hamster | Marigo, 2009 | ||
| skin | human | Fonfria, 2006; Vennekens, 2007; Wang 2019; Otsuka Saito, 2023 | RT-PCR, qPCR, Northern blot, WB, patch clamp |
| skeletal muscle | human | Launay, 2002 | |
| mouse | Krüger, 2008 | ||
| breast | human | Wong, 2020 | |
| stem cell | human | Tran, 2014 | |
| rat | Nelson, 2013 | ||
| adipose | human | Fonfria, 2006; Tran, 2014 | |
Figure 5.
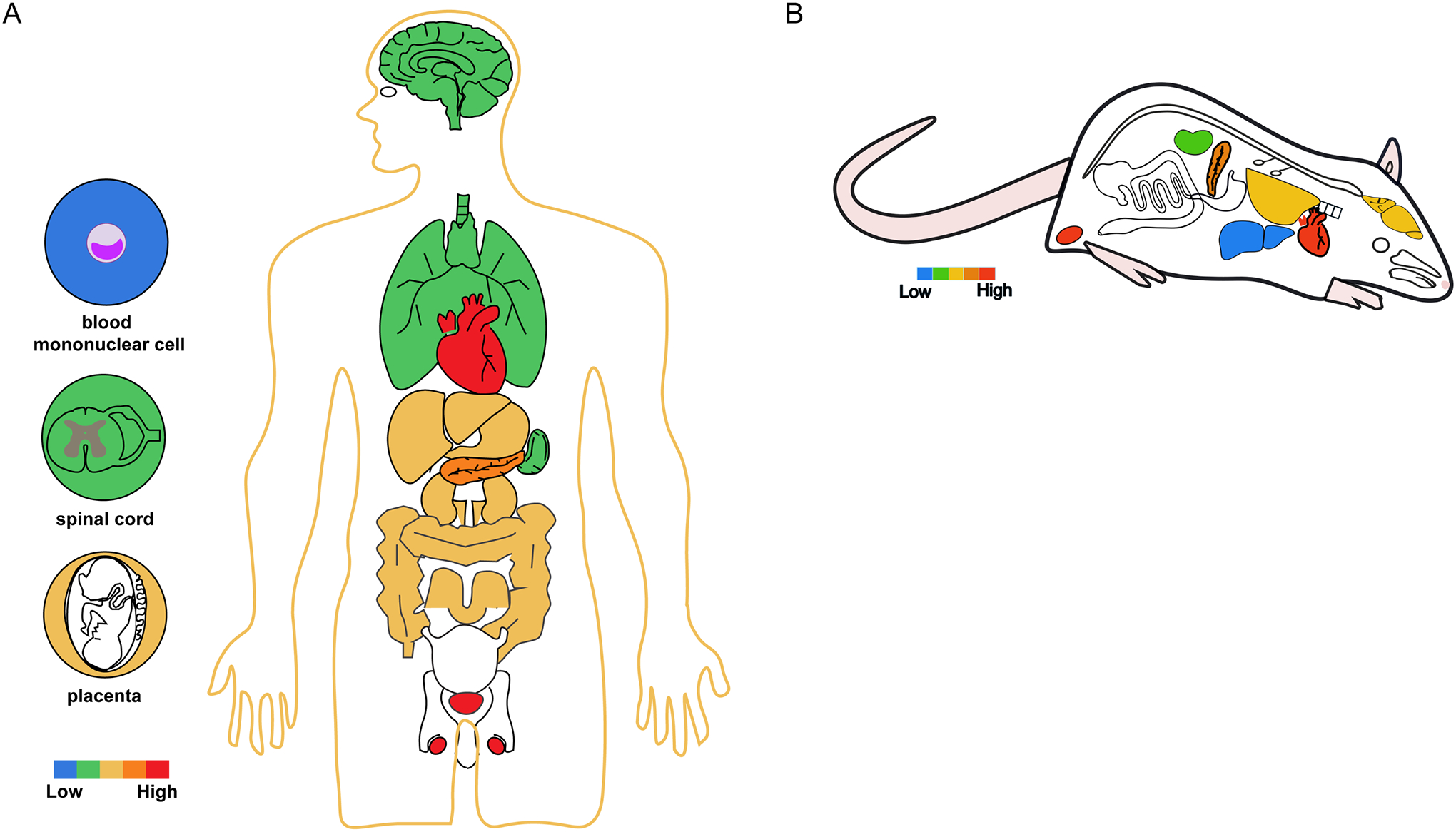
Summary of reported TRPM4 distribution in human (A) and rodents (B). The color scale indicates relative levels of expression based on consensus of reports in the literature. The colored circles depict TRPM4-expressing specific cells, tissues or organs in humans.
TRPM5: In contrast to the broad expression pattern of TRPM4, expression of TRPM5 appears to be restricted to specific cell types, as summarized in Table 3 and Figure 6. A systematic survey of TRPM5 expression of juvenile and adult mice using a reporter mouse model is consistent with this view (80). Substantial attention has been focused on the effector role of TRPM5 in chemical sensing. Accordingly, there is a much-detailed understanding of TRPM5 distribution in sensory cells. Nonetheless, expression of TRPM5 in other cell types has been widely reported.
Table 3.
TRPM5 expression in mammal species
| Organ/Tissue | Species | References | Methods |
|---|---|---|---|
| Taste tissue | |||
| tongue | monkey | Gonda, 2013 | RT-PCR, qPCR, Northern blot, in situ hybridization, IF, WB, TRPM5-GFP mice, patch clamp |
| rat | Teruyama, 2011; Padalhin, 2022 | ||
| mouse | Pérez, 2002; Pérez, 2003; Zhang, 2003; Clapp, 2006; Kaske, 2007; Zhang, 2007; Liu, 2011; Herrera Moro Chao, 2016; Cui, 2019; Wu, 2020; Nguyen, 2021 | ||
| chicken | Yoshida, 2017 | ||
| palate | chicken | Yoshida, 2017 | |
| Olfactory system | |||
| main olfactory epithelium | mouse | Kaske, 2007; Lin, 2007; Lin, 2008b; Nakashimo, 2010; Oshimoto, 2013; López, 2014; Yamaguchi, 2014; Fu, 2018; Genovese, 2018; Lemons, 2020; Dnate Baxter, 2021 | in situ hybridization, IHC, IF, TRPM5-GFP mice, patch clamp |
| GI tract | |||
| oesophagus | human | Young, 2008 | RT-PCR, qPCR, IHC, IF, Northern blot, in situ hybridization, WB, TRPM5-eGFP mice |
| stomach | human | Young, 2008; Widmayer, 2012 | |
| monkey | Gonda, 2013 | ||
| mouse | Pérez, 2002, Pérez, 2003; Kaske, 2007; Cui, 2019 | ||
| intestine | human | Fonfria, 2006; Young, 2008 | |
| monkey | Gonda, 2013 | ||
| rat | Feng, 2017; Kang, 2020 | ||
| mouse | Pérez, 2002; Pérez, 2003; Kaske, 2007; Bezençon, 2008; Young, 2008; Kokrashvili, 2009; Herrera Moro Chao, 2016; Howitt, 2016; Cui, 2019; Zheng, 2023 | ||
| Respiratory system | |||
| lung | mouse | Herrera Moro Chao, 2016 | RT-PCR, qPCR, IHC, IF, TRPM5-GFP mice, patch clamp |
| trachea | rat | Kaske, 2007 | |
| mouse | Kaske, 2007; Herrera Moro Chao, 2016; Genovese, 2018 | ||
| bronchus | mouse | Kaske, 2007 | |
| nose | human | Barham, 2013 | |
| mouse | Kaske, 2007; Lin, 2008a | ||
| Brain | |||
| third ventricle | mouse | Yu, 2023 | RT-PCR, qPCR, ICC, IHC, IF, TRPM5-GFP mice |
| hippocampus | mouse | Herrera Moro Chao, 2016 | |
| cortex | mouse | Herrera Moro Chao, 2016 | |
| hypothalamus | rat | Teruyama, 2011 | |
| mouse | Herrera Moro Chao, 2016 | ||
| amygdala | mouse | Thompson, 2012 | |
| cerebellum | mouse | Kim, 2013 | |
| brainstem | mouse | Crowder, 2007; Herrera Moro Chao, 2016 | |
| pituitary | human | Fonfria, 2006 | |
| Others | |||
| pancreas | human | Fonfria, 2006 | RT-PCR, qPCR, WB, IF, patch clamp |
| rat | Prawitt, 2003 | ||
| mouse | Colsoul, 2010; Colsoul, 2014 | ||
| heart | rat | Teruyama, 2011 | |
| kidney | human | Fonfria, 2006 | |
| rat | Liu, 2011; Teruyama, 2011 | ||
| mouse | Crowder, 2007; Cui, 2019 | ||
| prostate | human | Fonfria, 2006 | |
| skin | human | Mardaryev, 2021 | |
| odontoblast | mouse | Khatibi Shahidi, 2015 | |
Figure 6.
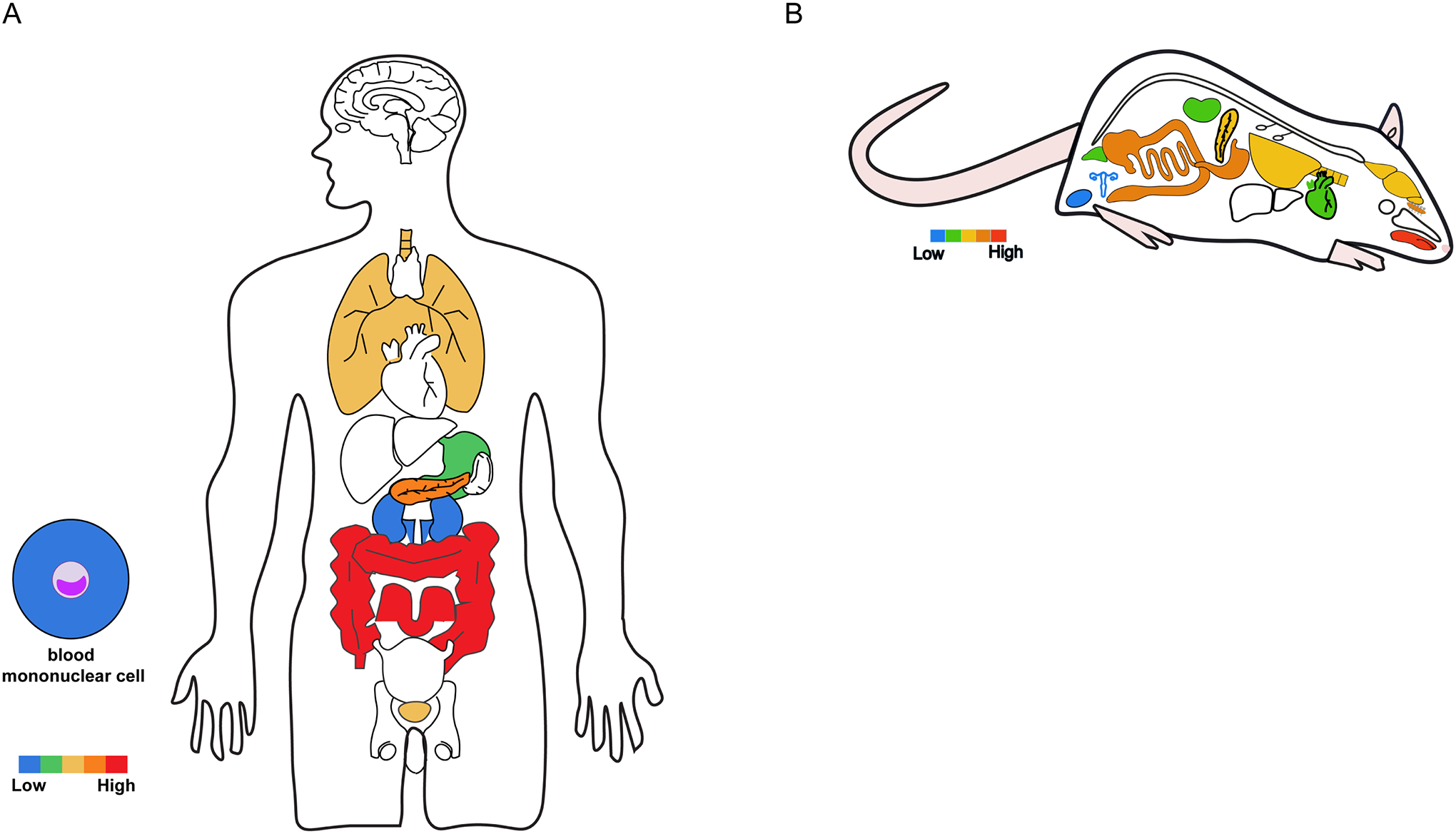
Summary of reported TRPM5 distribution in human (A) and rodents (B). The color scale indicates relative levels of expression based on consensus of reports in the literature. The colored circles depict TRPM4-expressing specific cells, tissues or organs in humans.
8. Potential functional roles in physiology
TRPM4 and TRPM5 have been implicated in a variety of physiological processes. In general terms, activities of TRPM4 and TRPM5 influence physiology in two ways—by depolarizing the plasma membrane and by its feedback control on calcium signaling. Membrane depolarization is the direct effect. This contribution is better appreciated in excitable cells such as cardiomyocytes, immune cells, pancreatic beta cells, and neuronal cells such as taste cells. Given their comparable permeability to Na+ and K+, the reversal potential of currents mediated by TRPM4 and TRPM5 is close to zero mV. For an excitable cell that typically has a deeply negative resting membrane potential, TRPM4 or TRPM5 activities would have a strong depolarizing effect. It may help elevate membrane potential to increase excitability and, upon reaching the threshold, trigger action potential firing; they may also contribute to extending the duration of action potentials. However, most excitable cells express a collection of excitatory ion channels, many of which can be sensitive to calcium. The challenge in determining whether and to what extent TRPM4 or TRPM5 participates in the cellular physiology of a particular cell type lies in the ability (or the lack of) in quantifying their relative contribution to membrane depolarization. The lack of highly selective pharmacological tools for these channels contributes to this challenge.
The second way TRPM4 and TRPM5 contribute to cellular physiology is by feeding back on the intracellular calcium signaling that activates them (Figure 7). For cells expressing voltage-gated Cav channels, membrane depolarization due to TRPM4 or TRPM5 activation may activate these highly calcium permeable channels, leading to a dramatic increase in calcium influx. This is a positive feedback process and has the potential to quickly bring the intracellular calcium concentration to a high level to promote calcium-dependent events. (Note that activities of TRPM4 and TRPM5 also exhibit a weak voltage sensitivity—see Section 5a—which provides an intrinsic positive feedback mechanism.) For cells expressing voltage-insensitive calcium-permeable ion channels such as CRAC channels, for example, many non-excitable cell types, the effect of TRPM4 or TRPM5 activation would be the opposite. Membrane depolarization would reduce the electrical driving force for calcium influx; the effect is expected to be more prominent in cells having a weakly negative resting membrane potential. This is a negative feedback process leading to a drop of the intracellular calcium concentration. An additional challenge in determining the feedback role of TRPM4 and TRPM5 is the fact that most calcium-permeable ion channels are themselves sensitive to the intracellular calcium level. The ability to distinguish contributions by these channels from that by TRPM4 or TRPM5 would be crucial in assessing the potential role of the latter to cellular physiology.
Figure 7.
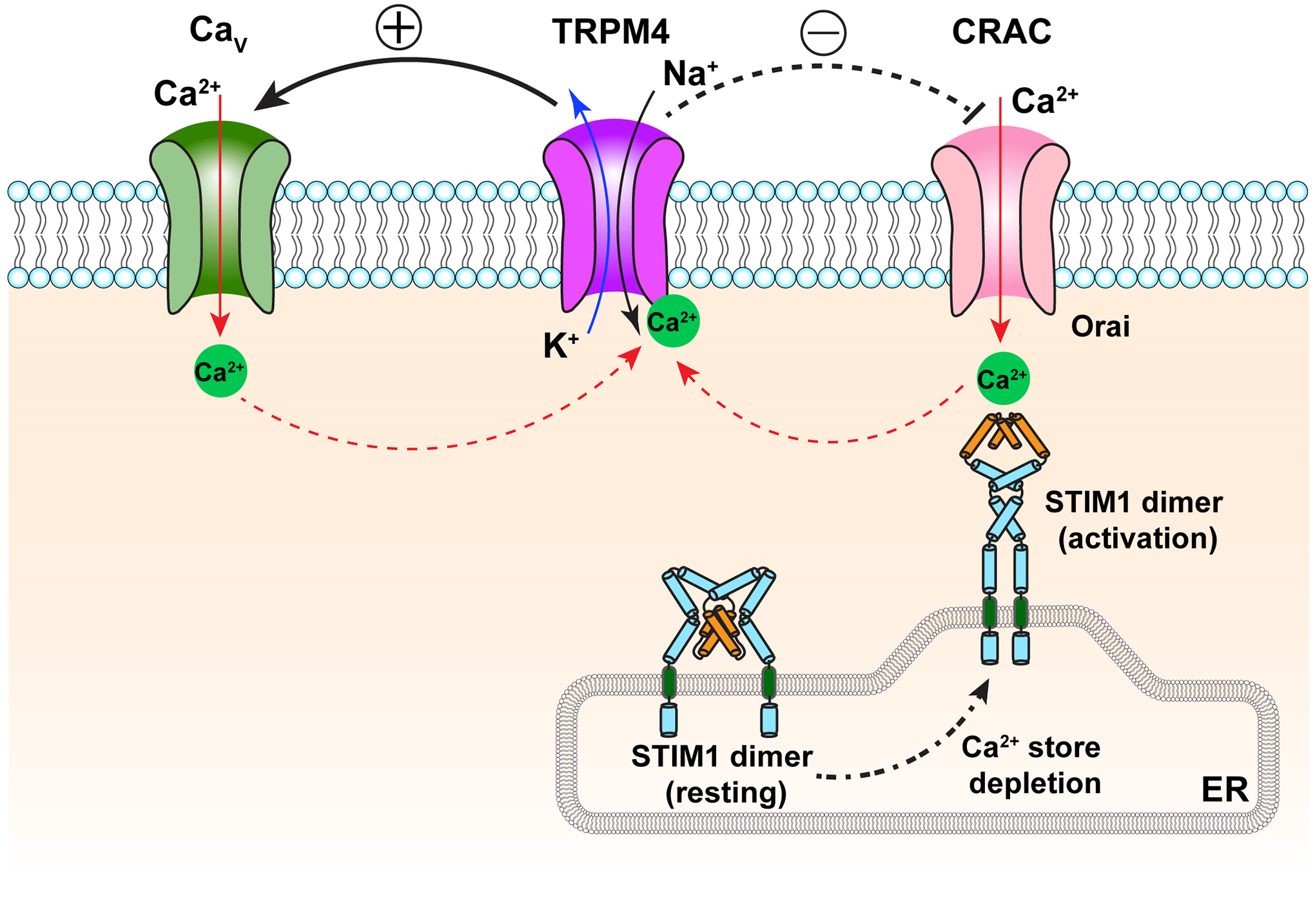
Positive (left side) and negative (right side) feedback regulation of calcium signaling by TRPM4. Solid red arrows indicate calcium flow; dash red arrows indicate calcium regulation targets. Solid black and deep blue arrows indicate sodium and potassium flow, respectively. CRAC, calcium release-activated calcium channel; STIM, stromal-interaction molecule; ER, endoplasmic reticulum.
As mentioned above, one limitation in deciphering the physiological functions of TRPM4 and TRPM5 is the lack of selective pharmacological tools to activate or inhibit them. The following cases illustrate this point well. The TRPM4 inhibitory compound 9-phenanthrol (81) has been widely used in published studies. In one study focusing on the mast cells, treatment with 9-phenanthrol was found to indeed induced mast cell degranulation (82). However, the effect did not seem to depend on TRPM4 but instead might be due to an off-target effect of 9-phenanthrol activating KCa3.1. The reagent, while sparing TRPM5 (67), also inhibits TMEM16A channels (83). It was further observed that 9-phenanthrol and other known TRPM4 inhibitors exhibit species and application dependent effects (71). These examples highlight the pressing need for better pharmacological tools for the study of TRPM4 and TRPM5 (70), and underscore the need to use caution in interpreting functional data. As new pharmacological tools with better efficacy and specificity are emerging (84), one can be optimistic that our ability to assess the physiological roles of TRPM4 and TRPM5 will be much improved in the near future.
In the following sections, we overview major working hypotheses on the physiological functions of TRPM4 and TRPM5.
a. TRPM4 in the cardiovascular system
The heart is a calcium-driven pump whose rhythm is controlled by electrical signals (Figure 8). The vasculature tone is also dynamically regulated by calcium and membrane potential. TRPM4, with its ability to link calcium and electrical signaling, is well-equipped to exert an influence on the cardiovascular system. Several lines of evidence strongly argue for a role of TRPM4 in the cardiovascular system. (i) TRPM4 expression in various parts of the heart and in vasculature has been detected in many labs (Figure 5). (ii) The observed expression pattern is consistent with earlier observations of CAN channels in these tissues (see Section 2). (iii) A long list of gain-of-function and loss-of-function TRPM4 mutations are linked to human hereditary heart diseases such as the long QT syndrome and the Brugada syndrome. (iv) The TRPM4 inhibitor 9-phenanthol exhibited protective effects for ischemia heart (85, 86) and reduced the action potential duration in Purkinje fibers (87). (v) TRPM4 knock-out mice exhibited elevated or reduced cardiac hypertrophy under stress conditions (88–90).
Figure 8.
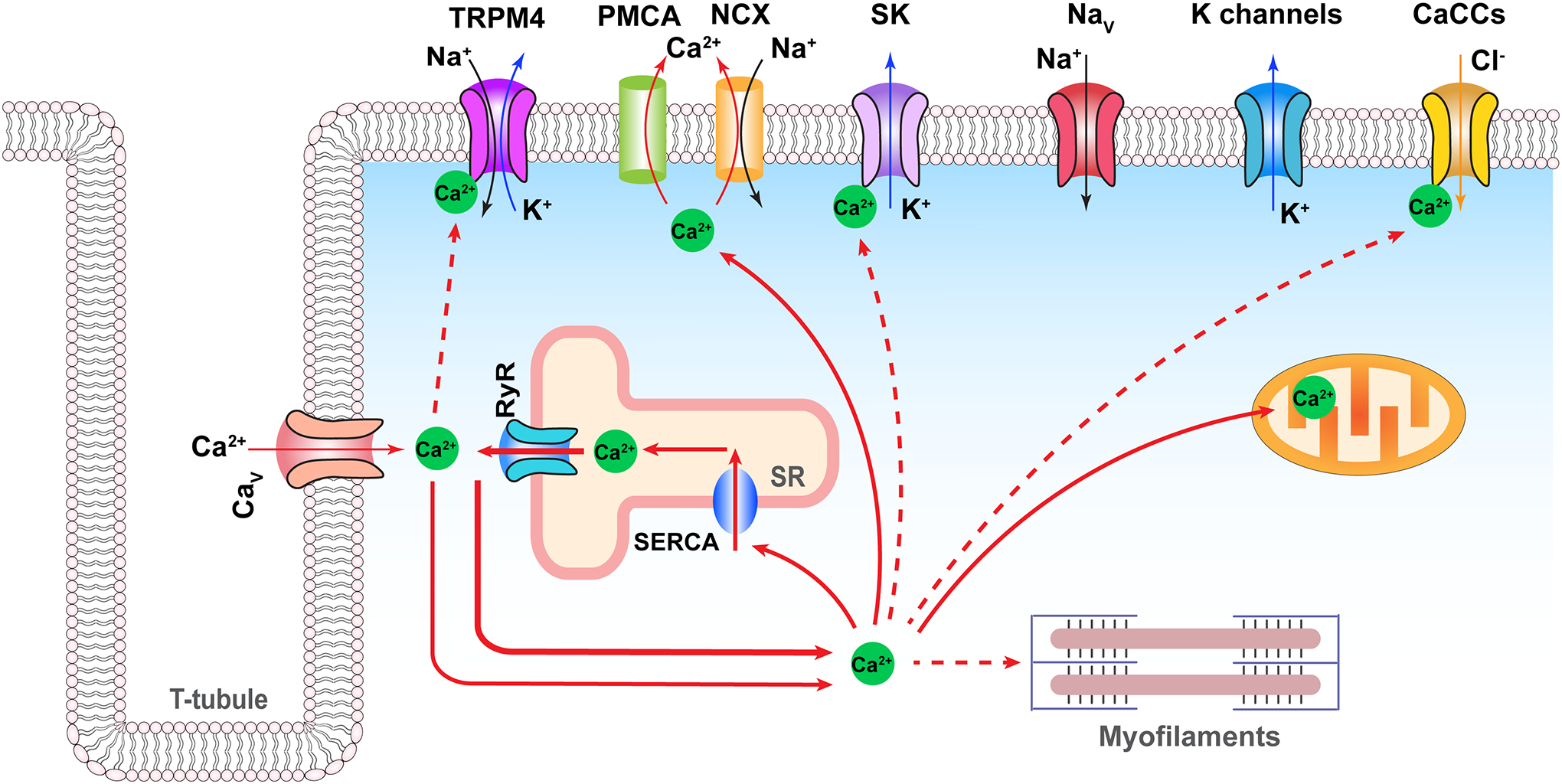
Physiological function of TRPM4 in cardiomyocytes. Solid red arrows indicate calcium flow; dash red arrows indicate calcium regulation targets. Solid black, deep blue, and orange arrows indicate sodium, potassium, and chloride flow, respectively. Only selected ion channels are highlighted in this illustration. PMCA, plasma membrane Ca2+ ATPase; NCX, sodium-calcium exchanger; CaCCs, calcium-activated chloride channels; SERCA, sarcoendoplasmic reticulum calcium ATPase; SR, sarcoplasmic reticulum; Nav, voltage-gated sodium channel subtype 1.5; Cav, voltage-gated calcium channel subtype 1.2; K channels represent a collection of potassium-selective channels.
How would TRPM4 contribute to heart physiology? With each heartbeat cycle, there is a dynamic wave of intracellular free calcium increase and decrease. The opportunity to convert a transient increase in calcium concentration into membrane depolarization has clear and attractive utilities in the heart and the vasculature (as well as other tissues such as the nervous system). One possible role of TRPM4 is to shape the pacemaker potential, a spontaneous diastolic depolarization of the sino-atrial cells. The depolarizing pacemaker current is mediated by a number of ion channels and transporters including the hyperpolarization-activated cyclic nucleotide-gated channel HCN4 (of the voltage clock) and Na+/Ca2+ exchanger NCX (of the calcium clock) (91). A component of the pacemaker current is mediated by calcium influx. While NCX activity is electrogenic and sensitive to calcium, replacing sodium with the NCX-impermanent ions K+ or Li+ did not eliminate the calcium-sensitive component (92, 93). As further evidence, 9-phenathrol reduced the beating rate with a half-maximal inhibition concentration comparable to that of TRPM4 and only in wildtype mice but not TRPM4 knockout mice (94). Another possibility is that TRPM4 activity contributes to the plateau phase of a cardiac action potential (though the TRPM4 current is expected to be small when the membrane potential is close to its reversal potential at near zero mV). If so, one can imagine that enhancing it could lead to a longer QT interval and disruption to other aspects of the cardiac electrical activity as seen in patients carrying one of the TRPM4 gain-of-function mutations; on the flip side, reducing TRPM4 activity over time may lead to hypertrophy of the heart as seen in the animal models—indeed, 9-phenathrol application reduced atrial action potential duration in the wildtype but not TRPM4 knockout mice (95). The heart electrical activity is so delicately controlled, it does not require a robust current to alter it to an extent that exhibits physiological and pathological consequences.
Of various human organs, the heart is one of the most noticeable for TRPM4 expression. Besides those mentioned earlier, the Pongs group observed that, among heart tissues, the highest level of TRPM4 mRNA was found in Purkinje fibers, followed by septum and right ventricle (96). In other studies of the human heart, TRPM4 mRNA was detected in the left ventricle (97); TRPM4 protein was detected in right ventricle (98) and left ventricle (99). Patch-clamp recordings identified currents with TRPM4 characteristics from the left ventricular fibroblasts (99). In addition, TRPM4 mRNA was detected in the cerebral vascular tissues (100); TRPM4 protein was detected in umbilical vein endothelial cells (101).
Findings from animal studies lend support for the conclusion that TRPM4 expresses broadly in the cardiovascular system. A survey of mRNA presence found messengers for TRPM4, among several TRPM and TRPV channels, in rat pulmonary arteries and aorta (102). Detection of TRPM4 protein in rat ventricular myocytes is consistent with functional data indicating that the TRPM4 inhibitor 9-phenanthrol preserved cardiac contractile function and protected the heart from ischemia reperfusion damage (85, 86). Both mRNA and protein were detected from mouse sino-atrial node cells; patch-clamp recordings confirmed the presence of a current with TRPM4 channel characteristics (56). A comparative study in mice suggested the highest levels of TRPM4 mRNA in lung and testis though it was positively detected the heart (79). Interestingly, 9-phenanthrol was found to be ineffective in reducing action potential duration in rabbit ventricle, though the same treatment was effective in Purkinje fibers (87). Selective deletion of TRPM4 from the mouse heart was linked to cardiac hypertrophy after chronic angiotensin treatment (103); similarly, TRPM4 knock-out (KO) mice developed cardiac hypertrophy during endurance training (104). Curiously, the TRPM4 expression level in the ventricle was found to be reduced in a monocrotaline-induced pressure load rat model (98) but increased in a spontaneously hypertensive rat model (105). It was also observed that selective deletion of TRPM4 in mouse cardiomyocytes led to a reduction in the left ventricular hypertrophy (89); in vitro experiments suggested that activation of TRPM4 in atrial myocytes can be regulated by the mechanosensitive Piezo1 channels (106). Results from additional knock-out studies further support a dynamic functional role of TRPM4 in the heart (90, 107–109).
Beyond the heart, TRPM4 is found to also contribute to the regulation of vascular tone (Figure 9). Myogenic constriction of arteries and arterioles is regulated by intracellular calcium. While potassium and chloride channels are known to contribute to the regulation of membrane potential that controls the Cav channel activity (110), TRPM4 is well-posed to amplify vascular responses. Indeed, a stretch-activated cation channel resembling TRPM4 was observed (111). TRPM4 in smooth muscles indeed can have a major role in myogenic constriction of the cerebral arteries, consistent with the presence of TRPM4 (but not TRPM5) mRNA and single-channel currents with properties resembling those of TRPM4 (112, 113). Interestingly, it appears that activation of TRPM4 channels in cerebral artery myocytes is not regulated by calcium entry from Cav channels but likely from calcium released from the intracellular store sarcoplasmic reticulum (114). An additional, more global regulatory mechanism than that shown in Figure 8 was identified, involving dynamic membrane trafficking of TRPM4 channels (59, 115). It was found that reduced TRPM4 activity is linked to the Gould syndrome (116), and inhibiting TRPM4 activity by nitric oxide causes blood vessel dilation (117). Simulations suggest that overexpression of TRPM4 (as seen in conduction block patients with gain-of-function TRPM4 mutants) may lead to propagation failure (118).
Figure 9.
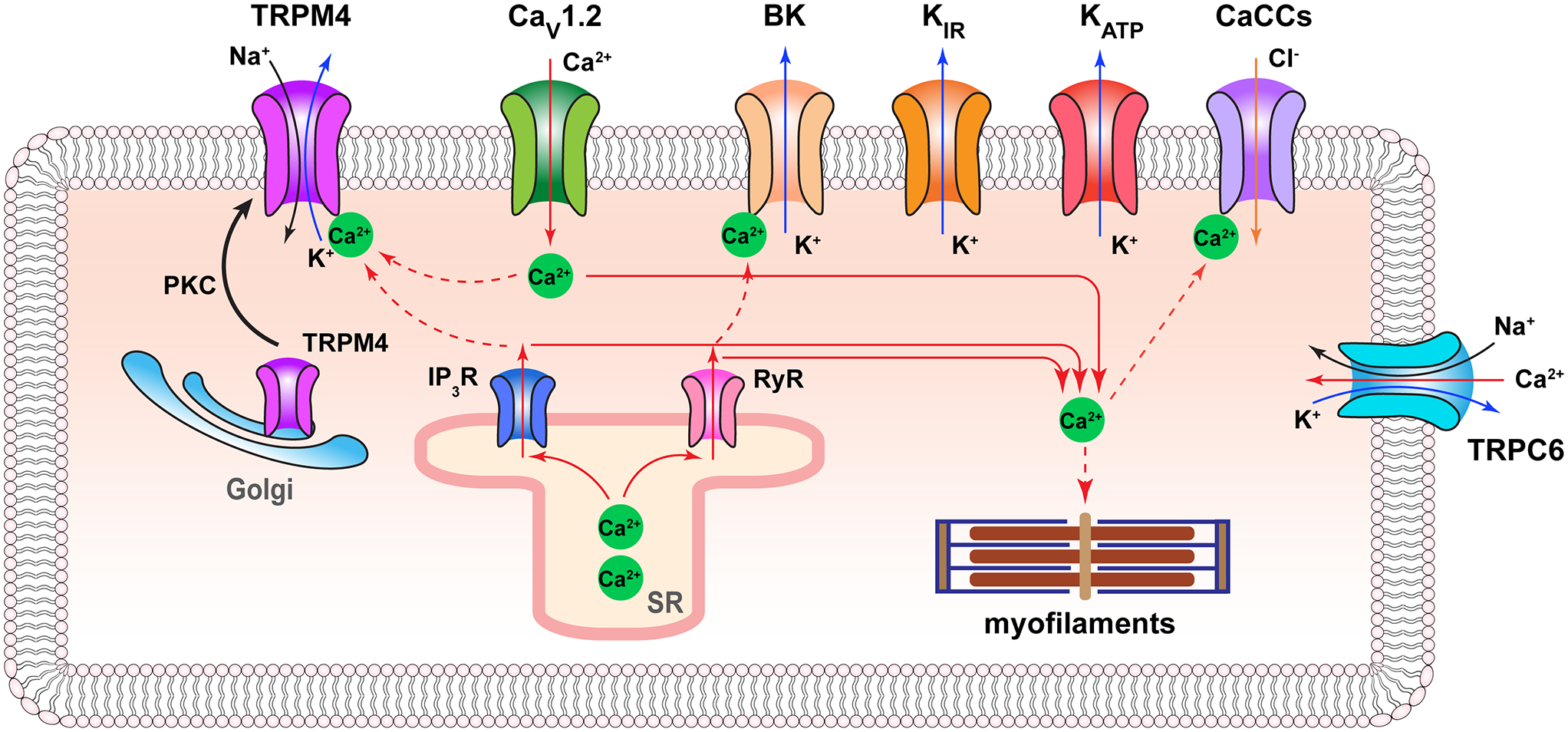
Physiological function of TRPM4 in vascular smooth muscle cell. Solid red arrows indicate calcium flow; dash red arrows indicate calcium regulation targets. Solid black, deep blue, and orange arrows indicate sodium, potassium, and chloride flow, respectively. Only selected ion channels are shown. CaCCs, calcium-activated chloride channels. PKC, protein kinase C; SR, sarcoplasmic reticulum.
In support of a role TRPM4 plays in vasculature, it was shown that suppression of TRPM4 expression or function reduced myogenic tone and induced vasoconstriction of rat cerebral parenchymal arterioles (119). Channels with TRPM4 characteristics were detected in freshly isolated rat vascular smooth muscle cells, lending functional supports to findings of TRPM4 (and TRPM5) mRNA in these cells (112). Additional studies found TRPM4 expression in rat mesenteric artery endothelial cells (120, 121), rat cerebral artery myocytes (117, 122), and mouse cerebral arteries (116). Suppressing TRPM4 expression with antisense oligodeoxynucleotides eliminated pressure-induced membrane depolarization and stimulated arterial constriction (112). In another study, the basal level of TRPM4 expression in rat capillaries was found to be low but was abundantly upregulated following spinal cord injury, which was linked to the severity of the induced secondary hemorrhage (123).
Nonetheless, there are arguments against a substantial role of TRPM4 in the cardiovascular system under physiological conditions. Indeed, the heart harbors a long list of ion channels whose symphonious activities drive rhythmic heart beating during the life span of the heart (Figure 8). While most of these channels are well-characterized (91), evidence for TRPM4 activity in the heart is relatively weak, leading to concerns on whether it has a substantial contribution to normal cardiac electrophysiology. One can point to Cav channels, SK channels, calcium-activated chloride channels, and NCX in the heart when looking for a linkage between intracellular calcium signaling and membrane depolarization (though they exhibit clear differences in functional properties from TRPM4). Even the strong linkage between gain-of-function TRPM4 mutations and hereditary heart diseases could be interpreted as a gained phenotype produced by an abnormally amplified TRPM4 current. Observations using 9-phenanthol might arise from its off-target effect, so do systemic Trpm4 knock-out. Most of the cardiac phenotypes of TRPM4 knock-out mice were not obvious unless the mice were put through various stresses. On top of these concerns is the high calcium concentrations required to activate TRPM4 in the majority of existing reports. However, none of these concerns appear to be sufficiently strong to confidently rule out a role TRPM4 plays in the cardiovascular system. For example, the experimentally observed high calcium activation concentrations could be the result of channel desensitization or the lack of co-factor(s) in expression systems (124).
b. TRPM4 in the immune system
During an immune response, long-lasting oscillation of the intracellular calcium concentration is associated with T cell activation (125, 126). Membrane potential serves a critical regulatory role for calcium entry across the plasma membrane, by providing the electrical driving force for ion influx (e.g., through CRAC channels) instead of directly controlling voltage-gated calcium channels (127, 128) (Figure 10). Both voltage-gated potassium channels (Kv1.3) and calcium-activated potassium channels (KCa3.1 and KCa2.2) are known to contribute to membrane potential regulation in T cells (128). However, being potassium-permeating ion channels, their activities would hyperpolarize the membrane potential. Also present in T cells are chloride-permeable channels (LRRC8), which are likely the most abundant among T cell ion channels. LRRC8 is activated by a decrease in ionic strength and the subsequent cell swelling. Like the currents mediated by potassium channels, the LRRC8 current is hyperpolarizing. Activation of TRPM4 by calcium, in contrast, would elevate membrane potential, reducing the electrical driving force for calcium entry through the Orai1/STIM1 CRAC channels. In this way, it may provide a negative feedback mechanism to limit calcium entry, acting as a rapid break to keep calcium entry in check. T cell activation is driven by the tyrosine kinase/phospholipase C pathway, which breaks down PIP2 to produce the signaling molecule IP3 to trigger calcium release from IP3 receptors. Since PIP2 binding to TRPM4 is required for channel activation, removal of PIP2 from the plasma membrane has the parallel effect of downregulating TRPM4 activity to allow Kv and KCa channels to hyperpolarize the cell. In support of the working hypothesis that TRPM4 contributes to an immune response, inhibiting TRPM4 activity led to a sustained elevation of T cell intracellular calcium concentration and enhanced interleukin-2 production (32). Similar observations were made in T helper cells, in which small interference RNA suppression of TRPM4 expression increased calcium influx and the oscillatory level in Th2 cells, and decreased influx and oscillations in Th1 cells (129). This cell-type dependence may be related to the relatively higher expression of TRPM4 in Th2 cells compared to Th1 cells.
Figure 10.
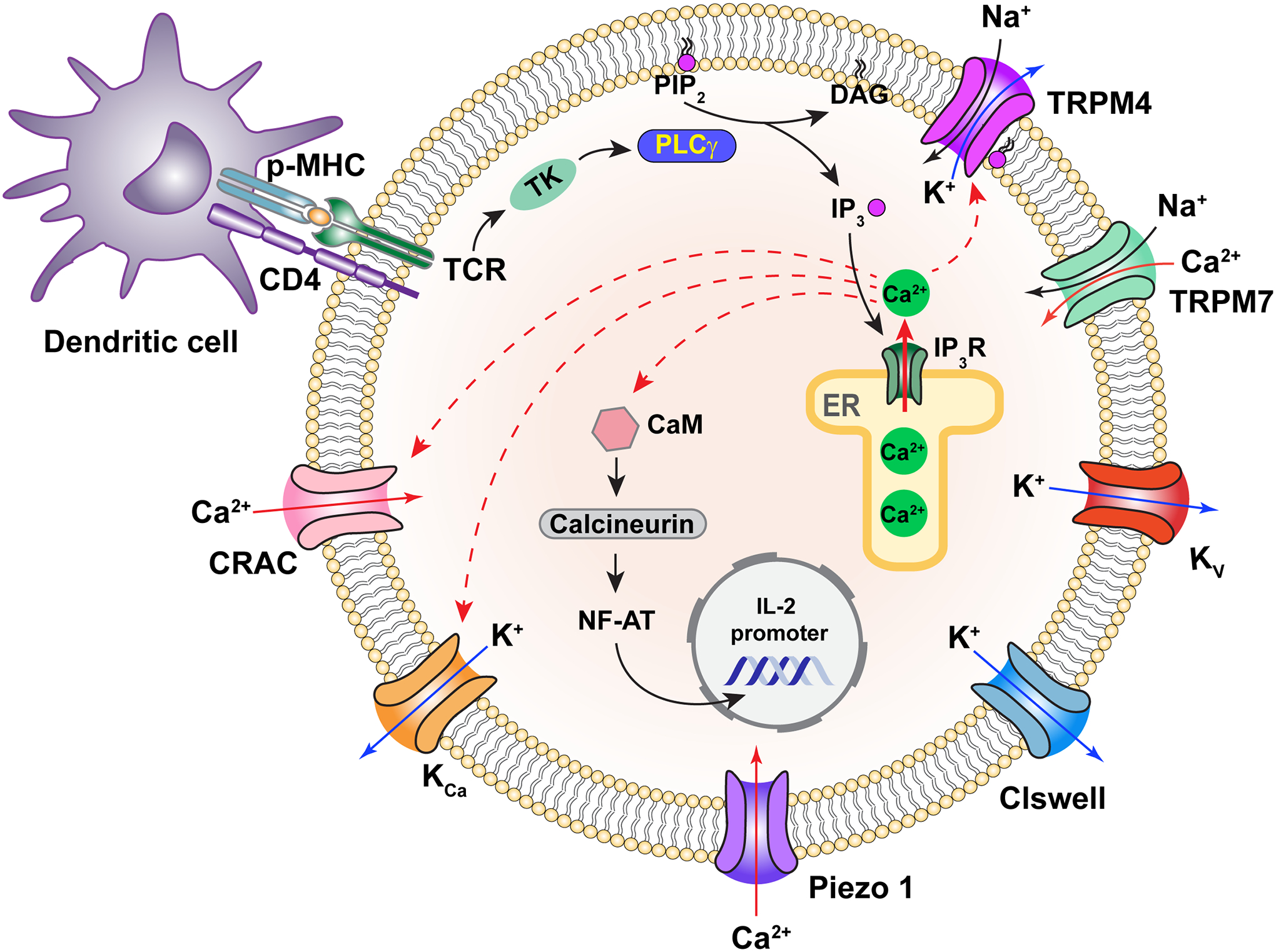
Physiological function of TRPM4 in T cells. Solid red arrows indicate calcium flow; dash arrows indicate calcium regulation targets. Solid black and deep blue arrows indicate sodium and potassium flow, respectively. Only selected ion channels are shown. CRAC, calcium release-activated calcium channel; CaM, calmodulin; p-MHC, peptide presented by major histocompatibility complex; CD4, cluster of differentiation 4; TCR, T-cell receptor; TK, tyrosine kinase; PLCγ, phosphoinositide phospholipase C γ subunit; IL-2, interleukin-2; ER, endoplasmic reticulum.
In support of the role of TRPM4 in the immune system, TRPM4 mRNA was detected in spleen, thymus, and cell-line cells for monocytes (U937), B lymphocytes (Ramos), T lymphocytes (Jurkat) (13, 78) but not in leukocytes when they were examined as a whole (12). A survey for TRP channels in peripheral blood mononuclear cells from psoriasis patients identified a decrease in the TRPM4 mRNA level (130). TRPM4 protein expression was detected by antibody in thymocytes, D10.G4, Molt-4 and Jurkat cells (32), and in mouse dendritic cells (131). Both TRPM4 mRNA and protein were identified from bone marrow-derived mast cells and CD3+CD4+ T cells but not CD3+CD8+ T cells or CD19+ B cells; no expression of TRPM5 in these cells was detected (132). Two studies provided strong evidence for functional TRPM4 expression in human and murine T cells using PCR and WB/immunoprecipitation (IP) combined with calcium imaging or patch-clamp techniques; both studies found that inhibition of TRPM4 expression increases Ca2+ influx in T cells (32, 129). The Weber et al. study further found that TRPM4 mRNA has a higher expression level in Th2 cells than Th1 cells; inhibition of TRPM4 expression in Th2 and Th1 cells exhibited opposite effects on the basal Ca2+ level and oscillation, indicating that different expression levels of TRPM4 in the two types of T helper cells play a distinct role in T cell function by differentially regulating Ca2+ signaling (129). Besides T cells, high levels of TRPM4 mRNA and protein have been reported in mast cells derived from bone marrow and skin. For example, based on the differences in current amplitude in Trpm4+/+ and Trpm4−/− mice bone marrow-derived mast cells (BMMCs), it has been suggested that TRPM4 expression in mast cells contributes at least partially to the endogenous CAN current; indeed, Trpm4−/− BMMCs had more Ca2+ entry compared to Trpm4+/+ BMMCs after FcεRI stimulation (132). Collectively, these findings indicate a critical role TRPM4 plays in immune system. Recently, several studies were conducted to test TRPM4 mRNA expression in peripheral blood mononuclear cells of patients with various diseases including multiple sclerosis, psoriasis, as well as inflammatory bowel disease; most of them however did not observe any significant change of TRPM4 expression compared to the control group except for one study of psoriasis patients (133–135). Given the cell-type dependence mentioned above (129), information from cell-type specific analyses is needed.
c. TRPM4 in Skin Health
It was reported that a hereditary TRPM4 mutation (I1040T) and a sporadic mutation (I1033M), both gain-of-function in nature, are linked to a severe form of skin disease named progressive symmetric erythrokeratodermia (PSEK) (136). Both mutant channels exhibited higher basal activities and increased sensitivity to calcium; HEK cells overexpressing them had an elevated resting membrane potential. The finding of skin disease-liked mutations highlights a potential role of TRPM4 in skin health. This is consistent with reports of TRPM4 expression in various skin cells (78). A recent study offers supportive evidence for the TRPM4 presence in keratinocytes: besides detecting TRPM4 protein in human keratinocytes and HaCat cells, it was found that BTP2 and aluminum potassium sulfate—both are TRPM4 agonists—could reduce cytokine production induced by tumor necrosis factor alpha in these cells (but not in Trpm4 deficient HaCaT cells) (137). TRPM4 expression was also detected in connective tissue mast cells of the skin (138).
Several lines of evidence in the Wang et al. report point to a potential role of TRPM4 in regulating keratinocyte proliferation and migration (136). HaCaT cell stably overexpressing either of the two mutant TRPM4 channels exhibited enhanced cell viability; EdU-based proliferation assay confirmed a significantly increased percentage of proliferating cells; higher rates of S and G2/M phase cells were identified in the population. Up-regulation of markers for proliferation and differentiation of keratinocytes such as Ki-67 (a nuclear marker for cell proliferation) and keratin 14 (a marker for undifferentiated keratinocytes) was observed in the affected skin tissues. However, the skin is not simply a physical protective barrier; it also serves as an immune organ to fend off pathogens from the ambient environment. It is just as likely that the skin disease PSEK is due to a disturbance to the immune function caused by elevated TRPM4 activities. Indeed, knock-in mice carrying one of the PSEK mutant are predisposed to more severe psoriasiform dermatitis compared to control mice (139). In a recently reported clinical case, the humanized anti-interleukin 36 receptor monoclonal antibody spesolimab was successfully used in treating a generalized pustular psoriasis patient carrying a TRPM4 mutation (140). Psoriasis is an immune disorder caused by immune system overactivity that renders skin cells to multiply too quickly. It remains to be determined whether overactive TRPM4 channels in the keratinocytes or dendritic cells exhibit a dominant role in psoriasis.
d. TRPM4 and TRPM5 in the Nervous system
TRPM4 expression in the nervous system has been widely noticed. Both TRPM4 mRNA and protein were detected in human cortex as well as spinal cord using quantitative real-time PCR (qPCR), in situ hybridization, and immunostaining; importantly, TRPM4 expression was upregulated in active demyelinating white matter brain lesions of individuals with multiple sclerosis in comparison to control samples but without any changes in the neuronal somata of the same samples (141). In rodents, TRPM4 expression was detected in several brain regions, such as cortex, hippocampus, hypothalamus, and spinal cord. TRPM4 was found to couple with NMDA receptors in mouse cortex and hippocampus (142). Furthermore, two recent neuroanatomy studies have characterized the postnatal developmental TRPM4 expression in mouse medial prefrontal cortex and hippocampus using immunostaining and patch-clamp recording (143, 144).
The functional significance of TRPM4 activities in the nervous system is not well understood. In hippocampal neurons, TRPM4 may contribute to postsynaptic depolarization and long-term potentiation (LTP) (144–146). TRPM4’s ability to link membrane potential changes and calcium signaling appears to be important for the control of hormone release from hypothalamic neurosecretory cells (147). TRPM4 channels are found to exhibit important roles in neuronal cell death (148), CA1 pyramidal cell excitability (149), loss of mossy cells from hippocampal dentate gyrus in temporal lobe epilepsy (150). TRPM4 mRNA or protein expression was detected in human trabecular meshwork (TM) cells (151) and murine optic nerve head (152). Functional expression of TRPM4 in the human eye was detected in TM cells using whole-cell and single-channel patch-clamp recordings; it is suggested that interactions between TRPM4 and another TRP channel, TRPV4, play an essential role in emergent temporal signaling in TM cells (151). TRPM4 mRNA and protein levels were found to be upregulated in brain or/and spinal cord in an experimental auto-immune encephalomyelitis (EAE) mouse model (141, 153). The usage of glibenclamide or ablation of Trpm4 gene could reduce the EAE-induced neuronal injury, indicating that TRPM4 is a potential target for EAE treatment (141). Upregulated TRPM4 protein expression was also detected in surrounding rat brain tissue 24 hours after traumatic brain injury (TBI), whereas 9-phenanthrol significantly diminished this upregulation (154). In a rat subarachnoid hemorrhage (SAH) model, it was shown that TRPM4 expression was three-times higher in cortex adjacent to SAH compared to uninjured control (155). In addition, TRPM4 mRNA and protein were upregulated in rat cortex and hippocampus after cardiac arrest, whereas glibencalmide could downregulate the elevated protein level when combined with target temperature management (156, 157).
Like TRPM4, TRPM5 has been suggested by a large body of literature to play a role in the nervous system. Both TRPM4 and TRPM5 were found to be expressed in rat hypothalamus but with different distribution patterns: TRPM4 mainly expressed in cell bodies of vasopressin and oxytocin neurons, whereas TRPM5 was predominantly found in dendrites of vasopressin neurons (158). TRPM5 mRNA expression was observed in many rodent brain regions such as cortex, hippocampus, hypothalamus and brainstem, and was suggested to be involved in energy homeostasis (159). In particular, the brainstem, where TRPM5 mRNA was found to be abundant, exhibited significant downregulation in both obese-hyperglycemic (ob/ob) mice and diet-induced obese mice compared to control group. Expression of GFP-tagged TRPM5 was detected in tanycytes in the third ventricle of mice brain; it is suggested that these tanycytes not only mediate bidirectional communication between the cerebrospinal fluid and blood but also play critical roles in making responses to metabolic hormones like leptin and in activating insulin secretion (160). Additionally, both TRPM4 and TRPM5 mRNA were detected in murine pre-Bötzinger complex of brainstem that regulates inspiratory burst activity, indicating that TRPM4 and TRPM5 may participate in inspiratory burst generation (161). In addition to the above brain regions, TRPM5 mRNA or protein expression has been reported in the mouse amygdala (162) and human pituitary (78). TRPM5 has been found to contribute to electrical activities of the spinal motoneurons (163).
TRPM4 and TRPM5 are also found to be expressed in Purkinje cells from mouse cerebellum, but they were not functionally required for depolarization-induced slow current (164). TRPM4 mRNA and protein were found in rodent respiratory neurons; it is proposed that TRPM4 expressed in pre-Bötzinger complex is important for respiratory pattern formation but is not required for respiratory rhythm generation (165). TRPM4 expression in retrotrapezoid nucleus (RTN) neurons may contribute to pacemaker firing and breathing (166). In addition, several studies have observed TRPM4 protein expression in rodent spinal cord, which was upregulated after spinal cord or peripheral nerve injury (167–170).
e. TRPM5 in the sensory system
Since TRPM5 expression has been detected in relatively limited cell types in comparison to TRPM4, research activities had mostly focused on its potential role in sensory physiology, insulin and endogenous opioids release. Shortly after the cloning of the Trpm5 gene (17, 171) and the detection of high levels of TRPM5 expression in taste cells (172), a gene knockout study revealed its critical role in sensing sweet, bitter and umami tastes (45) (Figure 11). Unlike the sour and salty taste modalities, these three taste modalities are mediated by G-protein coupled receptors T1Rs and T2Rs; downstream signaling involves PLC-catalyzed hydrolysis of PIP2 to produce IP3, which in turn triggers calcium release from intracellular store. Knocking out TRPM5 eliminated sweet and bitter reception (45) and at least dramatically reduced taste reception for umami and nicotine (45, 173, 174). TRPM5 channels (unlike TRPM4 channels) are blocked by extracellular protons at the pH levels of 6.0 or lower (49); this property would make TRPM5 channels unfit for sensing sour taste. TRPM5 knock-out mice would have to develop a preference for sucrose by an alternative pathway (175). TRPM5 appears to be the downstream effector of PLCβ2, which co-expresses with TRPM5 in taste bud sensory neurons and its removal by knock-out exhibited a similar phenotype to knocking out TRPM5 (45); in vitro experiments showed activation of TRPM5 by IP3-induced depletion of intracellular calcium store (172). In TRPM5-expressing taste cells, both IP3 and calcium elicited excitatory currents (176) and ATP release (177). Therefore, TRPM5 current activated by calcium release from intracellular store serves as the effector of taste signal transduction (178). The TRPM5-mediated excitation of taste cells also transduces sensory signaling for fat and carbohydrate (179–181), regulates glucose homeostasis (160), and may contribute to warmth enhancement and bitter suppression of sweet sensation (63, 182). Taken together, these findings indicate that TRPM5 is indispensable in the taste transduction pathway.
Figure 11.
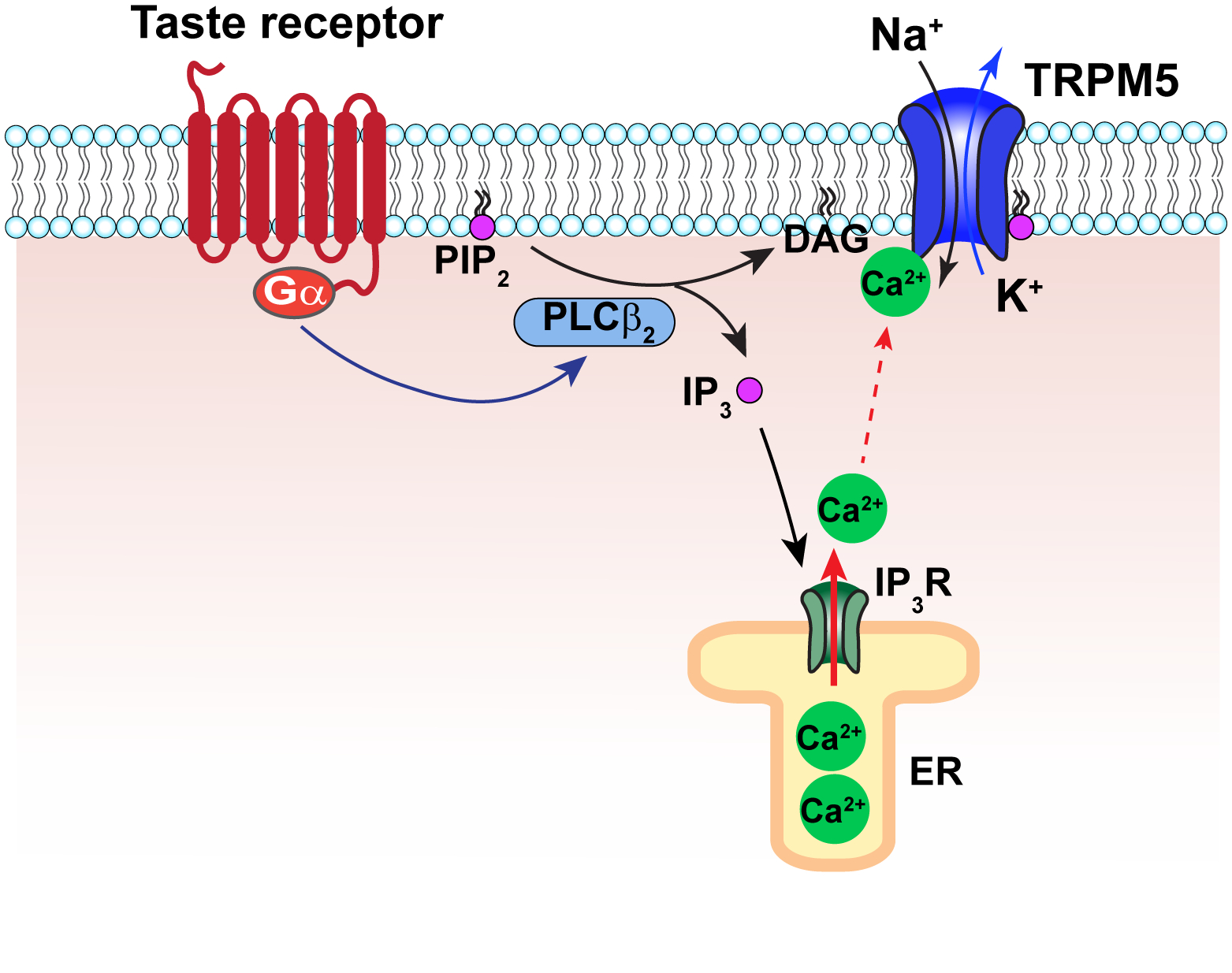
Physiological function of TRPM5 in taste cells. Solid red arrow indicates calcium flow; dash arrow indicates calcium regulation targets. Solid black and deep blue arrows indicate sodium and potassium flow, respectively. Gα, G protein α subunit; ER, endoplasmic reticulum.
In support of a role of TRPM5 in taste sensation, TRPM5 expression in the tongue has been widely noticed. It was reported that TRPM5 expression exhibits a restricted manner with particularly high levels in taste cells, together with the other signal transduction components such as Gγ13, PLCβ2 and inositol 1,4,5-trisphosphate receptor type III (IP3R) (172, 178). This study also showed a noticeable but lower level of TRPM5 expression in the GI system, to be discussed later. TRPM5 was found to co-express with T1R and T2R taste receptors in murine tongue, and deficiency of TRPM5 or PLC- β2 could eliminate sweet, umami and bitter taste perception without affecting sour or salty tastes (45). In Trpm5-GFP mice tongue, voltage-dependent Na+ and K+ currents could be recorded but not Ca2+ currents, consistent with TRPM5 being a CAN channel (183). Moreover, the Liman group also detected the expression of TRPM5 in murine tongue using the Trpm5-GFP mice, and demonstrated that both IP3 and calcium could trigger TRPM5-dependent currents in intact taste cells, but only intracellular calcium could activate TRPM5 in excised patches (176). TRPM5 expression in the tongue was confirmed in several additional studies (159, 184, 185). High TRPM5 expression in chicken palate rather than the tongue was reported (186). TRPM5 was found to abundantly express in mouse tongue epithelium, and ablation of Trpm5 or application of TRPM5 inhibitor caused impaired aversion to high-salt diet and developed salt-induced hypertension (187). In a follow-up study, long-term intake of bitter melon extracts was found to reinforce the aversion to high-salt diet by upregulating TRPM5 expression and function in mice, thereby preventing high-salt induced cardiovascular dysfunction and angiotensin II-induced hypertension (188). TRPM5 channels in murine taste buds may play a role in the linoleic acid transduction pathway (189).
Olfactory transduction, like taste, uses GPCR-mediated signaling but cyclic nucleotide-gated (CNG) channels and calcium-activated chloride channels as sequential effectors. Nonetheless, the PLC-TRPM5 pathway is found to operate in olfactory sensory neurons (OSNs) to transmit reception of semiochemicals, perhaps operating in parallel with the CNG/chloride channels pathway (190). Comparative functional tests with Trpm5-GFP mice, Trpm5 KO mice and SKn-1a KO mice revealed that TRPM5-expressing microvillous cells (MVCs) play important roles not only in maintaining the olfactory function, but also in regulating main olfactory epithelium (MOE) multicellular network activity, region-specific cell proliferation, and apoptosis during chemical exposure MOE (191–193). Knocking out TRPM5 in mice also eliminated the trigeminal response to airborne xenobiotics (194). Microarray data suggest TRPM5-expressing MVCs are likely involved in viral infection of olfactory epithelium (195). It is interesting to notice that calcium entry through CNG channels triggers negative feedback mechanisms to induce desensitization to sustained odor presence; TRPM5 channels on the other hand are activated by calcium. For certain odorants, it might be advantageous to retain long-lasting sensitive detection.
In support of the view that TRPM5 plays critical roles not only in the taste system but also in the olfactory system, there is growing evidence demonstrating strong TRPM5 expression in OSNs and MVCs of murine main olfactory epithelium. It was found that TRPM5 colocalizes with cyclic nucleotide-gated channel CNGA2 subunit and components of the PLC pathway in OSNs; TRPM5-expressing OSNs project axons to glomeruli in the ventral area of the MOE and participate in signal transduction of putative pheromones (190). TRPM5 immunoreactivity was detected in newborn or adult mice OSNs (196–198). However, the expression of TRPM5 mRNA in adult OSNs is controversial. In contrast to the previous studies, it was also reported that TRPM5 mRNA expression could not be detected in adult OSNs but in adult MVCs and embryonic OSNs (199).
Many studies offered supportive evidence for expression of TRPM5 in MVCs using Trpm5-GFP mice. For instance, morphological and immunocytochemical properties of the TRPM5-expressing MVCs in the MOE were described, as well as evidence for voltage-gated Na+ currents and Ca2+-activated cation currents using patch-clamp recordings (196). Using two-color in situ hybridization method, it was found that transcription factor Skn-1a co-expressed with TRPM5 in MOE of embryonic and adult mice, and deficiency of Skn-1a abolished the expression of TRPM5 in MOE (200). It was concluded that Skn-1a/Pou2f3 is indispensable for generation of TRPM5-expressing MVCs in the murine MOE. TRPM5-exressing MVCs share similar components of transduction signaling cascade with solitary chemosensory cells (SCCs) but, unlike SCCs, they possess only sparse trigeminal innervation (201). Two additional studies also showed the expression of TRPM5 in the olfactory epithelium (202, 203).
Interestingly, TRPM5-mediated chemosensation also exists in intestinal tissues and the pulmonary system. Duodenal enteroendocrine cells appear to use it to regulate endogenous opioids release (204). This sensory function of TRPM5 was found to influence insulin release, which will be discussed in the next section. TRPM5 in tracheal brush cells is important for acetylcholine release in response to bitter substances released by bacteria (205). TRPM4 has been found to also contribute to taste sensation in peripheral taste receptor cells, where PLCβ3 instead of PLCβ2 mediates taste signal transduction (206, 207).
Many studies revealed abundant TRPM5 expression in the primates or rodents GI tract from esophagus to large intestine. A broad expression profile of TRPM5 mRNA in human GI tract biopsy samples with or without type II diabetes was observed (208). High-level TRPM5 mRNA expression was also detected in human and monkey stomach and intestine tissue (78, 184). Interestingly, TRPM5 mRNA and protein have been shown to be upregulated in morbidly obese patients compared to normal weight people (209). However, a similar inverse correlations with blood sugar concentration in both normal and diabetic groups was also reported (208). In rodents, many studies detected TRPM5 mRNA or protein expression in GI tract, mostly in the intestine and a few in the stomach (172, 178, 187, 202, 204, 208, 210–214). A systematic TRPM5 immunoreactive profiling in rodent detected solitary TRPM5-positive cells with brush cell markers throughout the GI tract and respiratory system (202). Another study also described that TRPM5-expressing intestinal cells are mostly brush cells (210). To explore the biological role of TRPM5 in these tuft cells, one study noticed that SCCs co-expressing TRPM5 and endogenous opioids exist in duodenum, suggesting that TRPM5 is required for secretion of these opioid peptides (204). It has also been suggested, based on evidence from Trpm5-eGFP mice and Trpm5-null mice, that TRPM5-expressing tuft cell could mediate tuft cell response to intestinal parasites (212).
TRPM5 channels are found to be critical for small intestinal remodeling in responding to helminths and protists, which requires TRPM5-expressing type 2 innate lymphoid cells (ILC2) (215, 216). It also regulates mucin secretion from colon goblet cells (217) in cooperation with the sodium/calcium exchanger NCX (218). Knocking out Trpm5 abrogates accumulation of the taste-chemosensory tuft cells in intestine, the primary source of the parasite-induced cytokine interleukin-25 (212). In B lymphocytes, TRPM5 channels were found to regulate calcium responses to influence proliferation and inflammatory responses (219). TRPM5 channels are also suggested to contribute to thymic function (220).
In addition to the GI tract section, Kaske et al. described a detailed distribution pattern of TRPM5 protein in the rodent respiratory system using immunohistochemistry (IHC). They found TRPM5-expressing tuft cells in nose, trachea and bronchus (202). TRPM5 mRNA in murine trachea and lung was also detected and found to be upregulated in diet-induced obesity mice only in trachea compared to control group (159). In addition, based on observations from Trpm5-GFP mice, IHC, and event-related potential (ERP) recording as well as calcium imaging, TRPM5-expressing SCCs in murine nasal cavity are suggested to respond to odorous irritants and play an essential role in monitoring inhaled air quality (221). A comparative study of TRPM5 expression in human nasal tissues from patients with or without rhinitis found that both groups showed similar TRPM5 mRNA expression levels (222).
f. TRPM4 and TRPM5 in pancreas endocrine release
Insulin release by beta-cells is accompanied by both an increase in intracellular calcium concentration and membrane depolarization. It is well-established that L-type voltage-gated Cav channels and KATP channels participate in these signaling events (128), though CAN channels have been recorded in pancreatic acini as early as the 1980s (223). Now it is well-established that TRPM4 and TRPM5 are expressed in pancreas beta-cells and alpha-cells. Since their activities can contribute to both calcium signaling and electrical signaling, it is attractive to hypothesize that these channels play a role in regulating insulin release (Figure 12). Indeed, it was observed that suppressing TRPM4 expression led to significantly decreased insulin secretion from rat pancreatic beta-cell line INS-1 cells upon glucose or arginine vasopressin stimulus (224). Increases in TRPM4 activity can be the result of intracellular calcium release (representing a rapid phase upon calcium application) and triggered TRPM4 trafficking and insertion into plasma membrane (representing a second phase that took minutes) (224). In pancreatic acinar cells, evidence suggests that TRPM4 serves as a linkage between calcium signaling and membrane potential (225). It has been shown that the natural plant products steviol glycosides enhance pancreatic beta-cell function (and taste sensation) by potentiation of TRPM5 activity (226).
Figure 12.
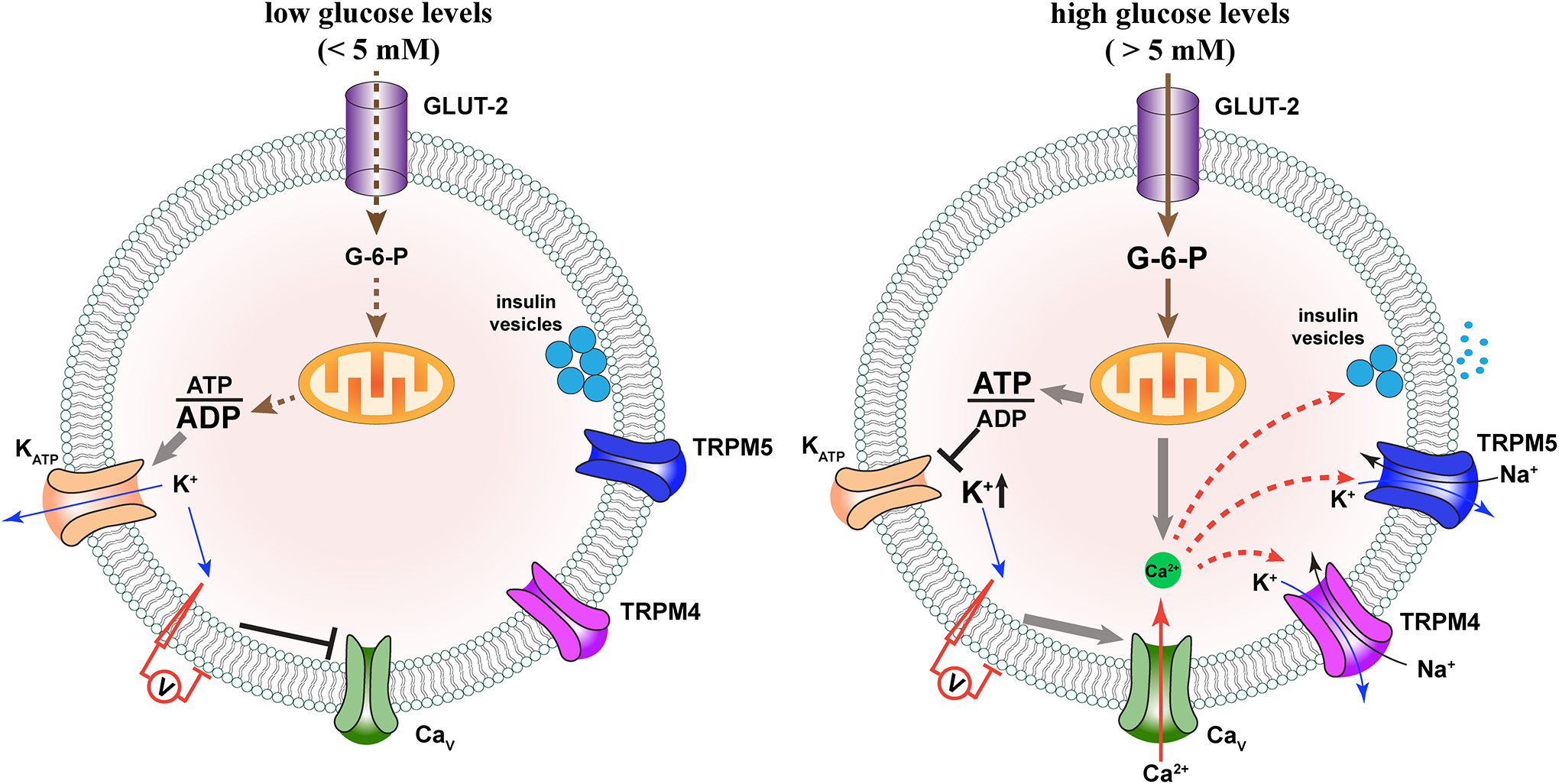
Physiological function of TRPM4 and TRPM5 in pancreas beta-cells at low (A) and high (B) blood glucose levels. Solid red arrows indicate calcium flow; dash arrows indicate calcium regulation targets. Solid black, deep blue arrows indicate sodium and potassium flow, respectively. Endoplasmic reticulum serves as an important intracellular calcium store for beta-cells but is omitted due to space limitation. Only selected ion channels are shown. GLUT-2, glucose transporter 2; G-6-P, glucose 6-phosphate.
Additional evidence supports a role TRPM4 plays in insulin secretion. TRPM4 mRNA was identified in the pancreas (13). The presence of TRPM4 at mRNA and protein levels in both alpha-cells (227, 228) and beta-cells (224, 228) has been reported. Currents with TRPM4 characteristics were recorded from the mouse pancreatic alpha-cell line αTC1–6 (227). The increase in PIP2 level in beta-cells upon glucose-induced activation may potentiate TRPM4 activity. In addition, the glibenclamide is known to inhibit TRPM4 (56). Indeed, application of 9-phenanthrol inhibited insulin secretion from rat islets of Langerhans (229). Glucagon-like peptide 1 (GLP-1) is an incretin hormone working through the cAMP/PKA (protein kinase A) and PLC/PKC signaling pathways (230). It was observed that, when applied to mouse islets, GLP-1 caused membrane depolarization, increased action potential firing, and enhanced insulin secretion even in the presence of KATP channel blocker tolbutamide and the L-type calcium channel blocker isradipine; the effects could be reduced by lowering extracellular concentration of TRPM4-permeating Na+ ions (231). The GLP-1 effects are thought to be due to the PKC and ATP sensitivity of TRPM4. In the αTC1–6 cell line, suppressing TRPM4 expression reduced intracellular calcium signaling and glucagon secretion (227).
Like TRPM4, TRPM5 may also participate in regulating insulin secretion. It was found that knocking out TRPM5 suppresses glucose-induced calcium oscillation in pancreatic islets which may lead to glucose tolerance (232); insulin in turn downregulates pancreatic islet TRPM5 expression (233). Several studies reported the detection of endogenous TRPM5 expression in rodent beta-cell lines as well as human pancreatic islets. In support of TRPM5 mRNA expression in beta-cells from various species, whole-cell patch-clamp recordings of rat beta-cells revealed a transient current activated by intracellular calcium that exhibited outward rectification and calcium concentration dependence similar to those seen in TRPM5-expressing HEK293 cells (17). Expression of TRPM5 in pancreatic beta-cells at mRNA and protein level was also detected using qPCR and immunostaining techniques; a calcium-activated cation current in single pancreatic islet cells was likely mediated by TRPM5 based on the differences in current pattern in Trpm5 KO and wild-type mice (232). The detected level of TRPM5 mRNA reduced in isolated pancreatic islets from diabetic-obese (db/db) and ob/ob mice, and insulin downregulated TRPM5 mRNA expression in pancreatic islets from leptin-deficient type II diabetes murine models (233). In summary, these findings indicate that, in addition to TRPM4, TRPM5 in beta cells is important in regulating insulin release and glucose levels in mammals.
As mentioned earlier, the chemo-sensing function of TRPM5 in intestine allows it to participate in regulating pancreas function and insulin release, and its malfunction may contribute to metabolic diseases. In a rat Type II diabetes model, high expression of TRPM5 mRNA in ileum rather than duodenum was observed and, interestingly, TRPM5 expression was downregulated in ileum of diabetic rat (211). However, quinoa was found to reduce upregulated TRPM5 protein in colon from a mouse type II diabetes model in another study (214). This discrepancy might be due to the difference of species and sampled tissues. In addition, a study of obesity observed considerable enrichment of TRPM5 mRNA in murine duodenum, which was downregulated in diet-induced obese mice group (159). Interestingly, one group observed that monosodium glutamate (MSG) could increase cholecystokinin (CCK), a hormone known for reducing food intake, accompanied by elevated expressions of taste receptor and TRPM5 mRNA in rat duodenum (213). Consistent with findings of TRPM5 functions in taste cells, it is reported that the sweet taste receptor in beta cells also relies on PLC and TRPM5 for signaling (234).
g. Additional potential functions
TRPM4 expression was detected in the human and rodent urogenital system including bladder, uterus, kidney, prostate, testis, and placenta. Using rodent bladder tissues, Yu et al. conducted a systematic study of TRP channel expression at the mRNA and protein levels, and identified that TRPM4 has a relatively high expression level in urothelium (235). Consistent with the evidence of TRPM4 mRNA and protein expression in rat detrusor smooth muscle (DSM) by means of RT-PCR, WB and immunostaining, patch-clamp recordings demonstrated that 9-phenanthrol could significantly reduce the spontaneous inward current activity in freshly isolated rat DSM cells (236). Similar results were obtained with guinea pig or human DSM and support that TRPM4 plays a critical role in human and rodent DSM excitability and contractility (237, 238). Indeed, age-dependent decrease in TRPM4 expression could attenuate urinary bladder smooth muscle contractility (239). Additional functional evidence supports a potential role of TRPM4 in detrusor overactivity after spinal cord transection in mice (240). Potential linage between TRPM4/TRPM5 channels and cancer has been discussed in a recent review (241).
Both TRPM4 and TRPM5 may help control breath patten (161, 166, 242). TRPM5 in particular mediates tracheal brush cell activation (243). In mice, TRPM4 contributes to uterine contraction (244). Multiple lines of evidence from RT-PCR, WB, electrophysiology, calcium imaging, as well as TRPM4 shRNA indicate that TRPM4 in human adipose-derived stem cells (HASCs) mediates histamine-induced calcium signaling (245). TRPM4 expression in uterus was revealed by mRNA expression in human and murine endometrium, suggesting a role in endometrium function (78, 246–248).
Although TRPM4 expression in kidney (13, 78, 79, 85, 158, 249), prostate (78, 237, 250), testis (79, 251), and placenta (13, 78) has been reported, the functional role of TRPM4 in these organs is still unknown. In addition, TRPM4 mRNA or protein was detected in human and rodent lung (78, 79, 85), liver (13, 78, 85), stomach and intestine (78), tongue (158), skeletal muscle (13, 158, 252), inner ear (253, 254), breast (255), and adipose (78, 245). TRPM5 expression in human and rodent heart (158), prostate (78), skin (256), odontoblast (257), and kidney (78, 158, 161, 187, 189) was also detected. Function of TRPM4 and TRPM5 in these tissues or cells remains to be examined.
9. Human diseases and transgenic animal models
When various splice variants of TRPM4 were cloned and examined functionally (12, 13), their resemblance to the CAN channels was immediately recognized. However, CAN currents had been recorded from many cell types. What might be the physiological role of the cloned TRPM4 channels in these cells? An exciting early finding came from a gene linkage study of progressive familial heart block type I (PFHBI) patients (96). PFHBI is an autosomal-dominantly inherited heart disease of the His-Purkinje system that progressively disrupts cardiac electrical conduction. The study of one large pedigree revealed an E7K mutation in TRPM4 (96). Located at the N-terminal end of the channel, this mutation does not appear to affect TRPM4’s function; nonetheless, the mutant channels were found to accumulate abnormally at the cell surface. This was caused by attenuation of deSUMOylation of the TRPM4 protein that impaired endocytosis. Subsequently, gain-of-function TRPM4 mutations due to disrupted endocytosis and resultant elevated surface expression have been described in numerous reports of patients suffering from the Brugada syndrome and the long QT syndrome (Figure 13, left). This mounting evidence, together with earlier reports of CAN channels in the cardiovascular system, strongly suggests a role of TRPM4 in regulating normal cardiac electrical conduction. However, even if the heart diseases are caused by a gain of TRPM4 function, logically it remains possible that TRPM4 current does not contribute significantly to the electrical activity of a normal heart. It is further noticed that another calcium-activated cation channel, SK2, exists in the heart; being a potassium-selective channel, the functional role of SK2 would be to convert a calcium signal into membrane hyperpolarization (91). Interestingly, reports of cardiac diseases linked to loss-of-function TRPM4 mutations start to emerge (Figure 13, right).
Figure 13.
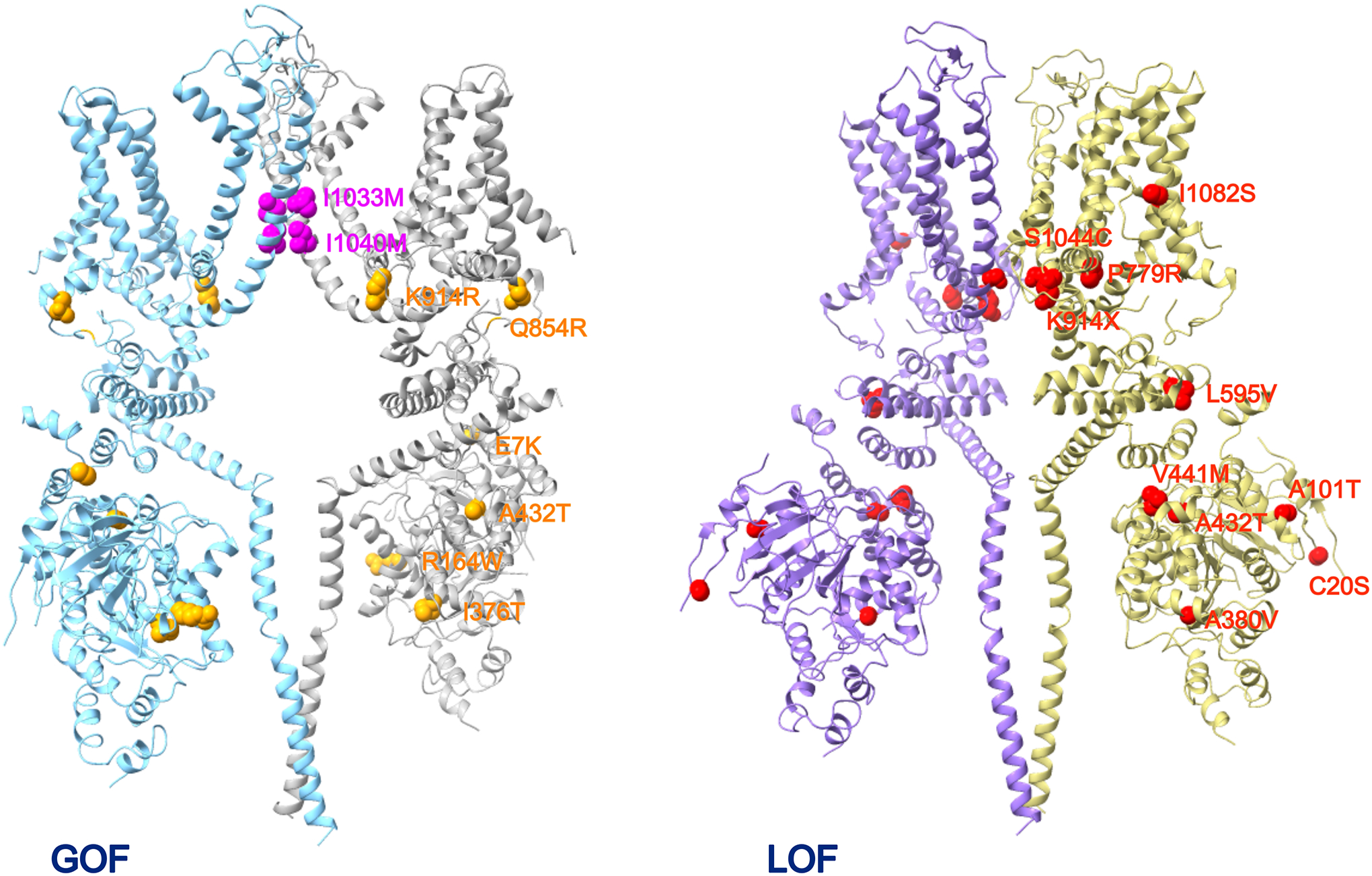
Location of human TRPM4 mutations linked to cardiac and skin diseases, shown on two subunits. (A) The gain-of-function (GOF) mutations associated with skin (magenta) and cardiac diseases (orange). (B) The loss-of-function (LOF) mutations associated with cardiac diseases (red).
In addition to naturally occurring channel mutations, it is found that the transcription factor p53 can also affect the expression level of TRPM4 (258). The process may play a role in cancer cells in which expression of TRPM4 has been found to be upregulated; it is suggested that overexpression of TRPM4 may contribute to increased adhesion, migration, proliferation, and invasion (255, 259–265). Increased TRPM4 activity can also be achieved by activation of Ca2+/calmodulin-dependent protein kinase II (CaMKII), resulting in arrhythmogenicity (266). Protein tyrosine phosphatase non-receptor type 6 (PTPN6) can affect the surface level of TRPM4 through direct binding (267).
The identification of two TRPM4 gain-of-function mutants in PSEK patients called for attention of the channel’s potential physiological role in the skin (136) (Figure 14A). PSEK is a severe skin disorder characterized by early-onset, slowly progressive, symmetrical, erythematous skin plaques predominantly on the extensor aspect of the extremities (Figure 14B&C). Both identified mutations are in the S6 segment that lines the ion permeation pore (Figure 13, left). They are both gain of function in nature, yielding higher calcium sensitivity in the mutant channels (Figure 14D&E); increased channel activity leads to elevation of the resting membrane potential. Surface expression level was pointedly checked but no change was detected. Enhanced cell proliferation was observed in patient skin tissue and mutant channel-expressing cell-line cells. Interesting, while the severe skin phenotype started shortly after birth and progressed during development, this progression stopped at puberty and gradually reversed itself. It is yet unknown what developmental change underlines this peculiar course. Even more intriguingly, none of the 21 PSEK patients was found to exhibit any detectable heart defect, and a skin problem has not been mentioned for the heart disease patients associated with TRPM4 gain-of-function mutations affecting trafficking (summarized earlier) or gating (268).
Figure 14.
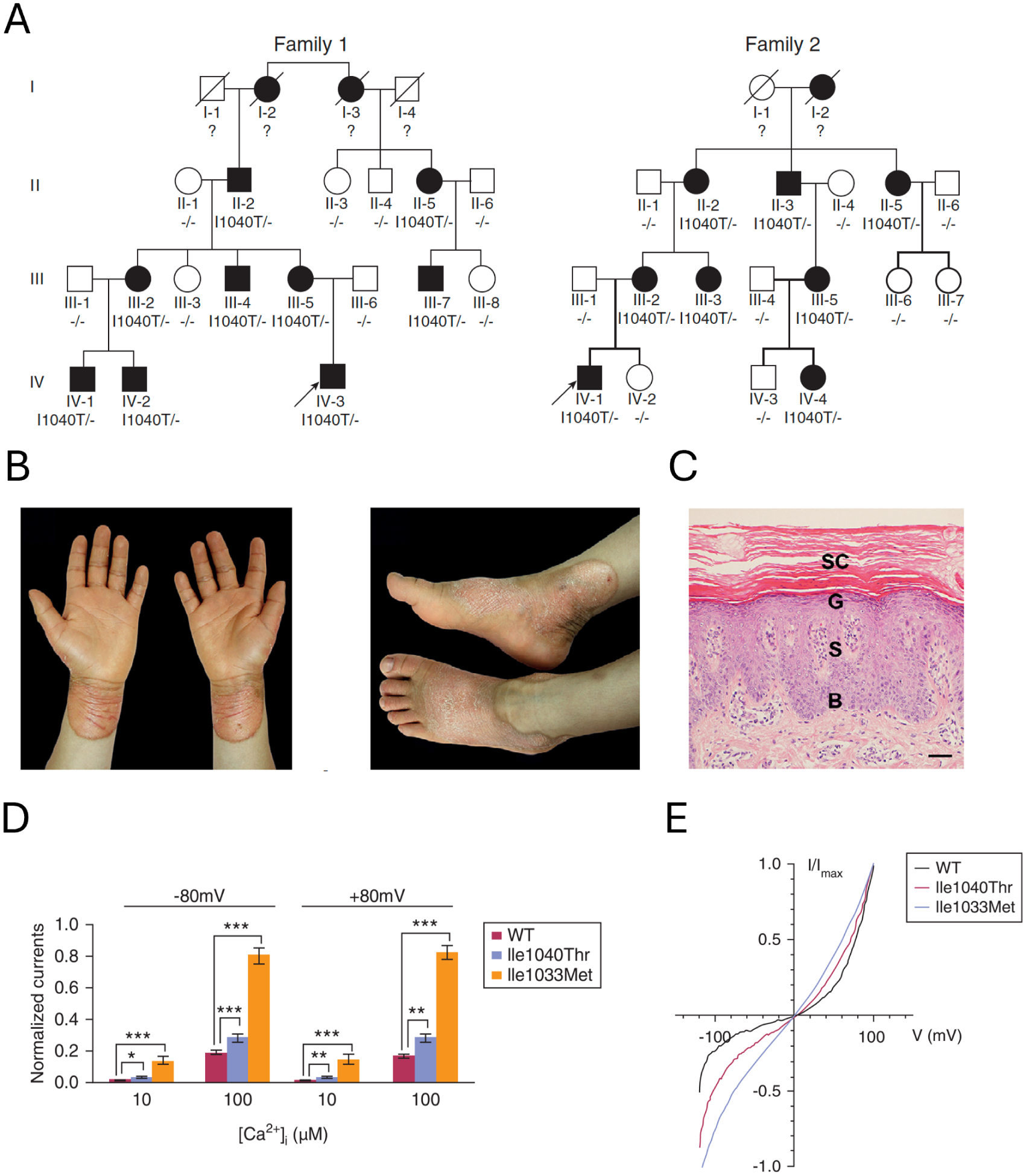
TRPM4 gain-of-function mutations cause progress symmetric erythrokeratodermia (PSEK). A. Two example pedigrees containing the I1040T mutation. B. Erythematous hyperkeratotic plaques on the patient’s hands and feet. C. Affected skin shows psoriasiform hyperplasia with focal parakeratosis and mild perivascular lymphocytic infiltration in the superficial dermis. D. Increased current responses of TRPM4 mutants. E. Comparison of the I-V relationship between TRPM4 wildtype and mutants. Modified from Wang et al. (136) with permission.
There is so far no report of human immune system disease that is directly linked to genetic defects in TRPM4. Nonetheless, changes in the level of TRPM4 expression have been observed in patients. A survey among TRP channels of peripheral blood mononuclear cells of inflammatory bowel disease patients suggested that TRPM4 expression might be negatively correlated with severeness of ulcerative colitis (135). Evidence for TRPM4 over-expression in acute myeloid leukemia patients was reported, and small interfering RNA inhibition of TRPM4 expression led to cell cycle arrest in lysine [K]-specific methyltransferase 2A (KMT2A)-rearranged leukemia cells (269). However, a survey of 64 multiple sclerosis patients found no change in the TRPM4 mRNA level compared to healthy controls (134). This finding was later confirmed in a survey of 30 relapsing-remitting multiple sclerosis patients (133). This group nonetheless observed a decrease in TRPM4 mRNA level in psoriasis patients (130). Again, it is unknown why no abnormality in heart health was found in these patients, just like no immune system disease was observed in heart or skin diseases patients carrying a TRPM4 gain-of-function or loss-of-function mutation.
Many studies examined mouse models with disruption of the Trpm4 gene; some have been mentioned earlier. In support of a role TRPM4 plays in the cardiovascular system, cardiac muscle of Trpm4−/− mice exhibited abnormalities in ß-adrenergic inotropic response and duration of cardiac action potential (270). The Trpm4−/− mice exhibit cardiac hypertrophy and multilevel conduction blocks (88). TRPM4 also contributes to smooth muscle functions (113). Bone marrow-derived mast cells from TRPM4 knock-out mice exhibited increased calcium entry and degranulation upon stimulation, released more histamine, leukotrienes and tumor necrosis factor (132). These Trpm4−/− mice exhibited exaggerated IgE-mediated acute passive cutaneous anaphylactic response. In another study, dendritic cells from Trpm4−/− mice exhibited impaired chemokine-induced migration, with a concomitant downregulation of the expression of phospholipase C that is thought to be due to calcium overload (131). Interestingly, maturation of dendritic cells was not hindered by the elimination of TRPM4 current, only migration was affected. This might be expected if the role of TRPM4 activity is to limit calcium entry through plasma membrane. In supporting this idea, migration of bone marrow-derived mast cells was found to be hindered by knocking out Trpm4, which appeared to be due to reduction in F-actin formation (271). It is found that, in migrating T cells, TRPM4 channels relocalize to the leading-edge (272). Suppression of TRPM4 function due to methyltransferase-induced production of non-functional TRPM4 splicing variant proteins leads to reduction in interleukin-2 production in mouse T cell (273). In addition, TRPM4 deletion increased mouse mortality in a model of sepsis (274). It is important to note that the gene knock-out approach (and knock-in approach discussed below) has a global effect on all tissues and cells that express the channel. Observations obtained from these mice need to be confirmed with experimental methods with better specificity.
The only TRPM4 gain-of-function mouse model reported so far carries the human I1033M mutation that increases the channel’s calcium sensitivity and is linked to a severe form of skin disease, as mentioned earlier (136, 139). The mice exhibited no abnormality in the skin under specific pathogen-free conditions; they bred well and showed no noticeable health issue. However, upon imiquimod challenges, they presented more severe psoriasiform dermatitis than wildtype mice (139). Greater accumulation of CCR6-expressing γδ T cells and higher mRNA levels of Il17a were observed; dendritic cells showed enhanced migration. Increased keratinocyte proliferation was also observed.
No human disease has been linked to changes in genetic coding or expression level of TRPM5. Nonetheless, knocking out of Trpm5 in mice revealed the channel’s physiological role as the effector of taste transduction (45). Just like knocking out PLCβ2, a key mediator of the taste transduction pathway, knocking out Trpm5 abolished sweet, bitter and umami taste reception (while leaving sour and salty tastes unaffected). This landmark study highlighted the functional role of TRPM5 in taste perception and, despite a report of detectable umami reception in another TRPM5 knock-out mouse model (173), attracted intensive studies of the role of TRPM5 in chemosensory physiology. Knocking out TRPM5 reduced bitter aversion (275) and metallic taste (276). It was quickly found that TRPM5 expressed in olfactory neurons participates in sensing semiochemicals (such as pheromones) (190). It was found that the TRPM5-expressing olfactory sensory neurons (OSNs) mediate pheromone transduction via a novel pathway that distinct from a canonical CNG channel-ANO2 channel coupling pathway (197). In another study, based on previous electroolfactogram (EOG) observations that Trpm5 knock-out mice showed similar responses to pheromones (and odors) compared to wild-type mice, EOG recordings were conducted with Trpm5 KO mice under unilateral naris occlusion condition. It was observed that Trpm5-null mice were less sensitive to putative pheromones than wild-type mice, and the number of OSNs expressing TRPM5 increased in wild-type mice with naris occlusion (198).
In addition to the roles in sweet, umami and bitter perception, TRPM5 was found to also mediate fat taste perception (180, 189). In support of this view, the long-chain unsaturated free fatty acid linoleic acid (LA) activates TRPM5 channels expressed in murine taste buds, thereby depolarizing taste cells and inducing a robust intracellular calcium rise. In single taste cells isolated from Trpm5-null mice tongue, LA-induced inward current and intracellular calcium rises were substantially decreased; consistent with the results from in vitro study, Trpm5-null mice could not distinguish LA compared to wild-type and were less sensitive to LA, indicating that TRPM5 plays an essential role in the LA-transduction pathway.
Even though TRPM4 has been proposed to regulate insulin release, no hyperglycemia, insulin deficiency, or diabetes mellitus was reported for the Trpm4 knock-out mice. Nonetheless, effects of the incretin hormone glucagon-like peptide 1 (GLP-1) could not be observed in Trpm4 or Trpm5 knock-out mice islets (231). Trpm5 knock-out mice however exhibited noticeable glucose intolerance—the blood glucose level was seen elevated for more than an hour compared to the wildtype, and hyperglycemia and arginine-induced insulin secretion was diminished in pancreatic islets (277).
10. Conclusion
The unique combination of being monovalent cation-selective in permeation and calcium sensitive in activation bestow TRPM4 and TRPM5 unique powers to influence cellular physiology. While their contributions as the unidentified CAN channels had been well documented, molecular cloning at the turn of century and recent high-resolution structural revelation set the stage for detailed investigation of their physiological functions. Their critical roles in a number of cellular signaling processes start to emerge. Despite these exciting developments, the biophysical properties of TRPM4 and TRPM5 are not fully characterized. Their calcium sensitivity under physiological conditions and factors influencing the sensitivity remain to be elucidated. Pharmacological tools with high selectivity are still lacking. These limitations add to the challenge in deciphering their physiological roles. The recent comparative study of TRPM4 structures at near body core temperatures and at the room temperature or lower (26) highlights the need to investigate heat-sensitive TRPM4 and TRPM5 channels (63) at conditions more closely resembling real physiology. Nonetheless, the intensive investigation being attracted to these channels will likely yield a much better understanding of the physiology, pharmacology, and pathology related to TRPM4 and TRPM5 in the near future, which will open the possibilities to address pathophysiological conditions related to their dysregulation or genetic variations.
Clinical Highlights.
Calcium and membrane potential are two prominent cellular signals. They are involved in virtually every aspect of physiology and are implicated in numerous pathological conditions. TRPM4 and TRPM5 ion channels offer a direct linkage between these two signals.
TRPM4 and TRPM5 are found to contribute to fundamental physiological processes such as cardiovascular function, immune response, pancreas endocrine release, taste reception, and others.
Many hereditary gain-of-function or loss-of-function TRPM4 mutations have been linked to cardiovascular diseases such as Brugada syndrome, long-QT syndrome, conduction block, and a severe skin disease progress symmetric erythrokeratodermia (PSEK). Knocking out Trpm5 in mice abolished sweet, bitter and umami taste reception.
Acknowledgements
We thank Don Bers, Mike Cahalan, Byung Chang Suh, and many other colleagues for insightful discussions. We regret the space limitation prevents citation and discussion of most of the over 1000 original reports and review articles in this active research field. Research on TRPM4 and TRPM5 in the Zheng lab is funded by NIH grant R01NS128180.
Footnotes
Activities of many other TRP channels are also sensitive to calcium, hence may contribute to chemical-to-electrical signal conversion. While noticeable similarities have been noticed—for example, the calcium-binding site in the transmembrane domain shared by multiple TRPM and TRPC channels—this review focuses on TRPM4 and TRPM5 and mentions other ion channels only when directly relevant.
Reference
- 1.Boron WF, and Boulpaep EL. Human Physiology. Elsevier, 2016. [Google Scholar]
- 2.Clapham DE. Calcium signaling. Cell 131: 1047–1058, 2007. [DOI] [PubMed] [Google Scholar]
- 3.Berridge MJ, Lipp P, and Bootman MD. The versatility and universality of calcium signalling. Nat Rev Mol Cell Biol 1: 11–21, 2000. [DOI] [PubMed] [Google Scholar]
- 4.Hu M, Feng X, Liu Q, Liu S, Huang F, and Xu H. The Ion Channels of Endomembranes. Physiol Rev 2024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Colquhoun D, Neher E, Reuter H, and Stevens CF. Inward current channels activated by intracellular Ca in cultured cardiac cells. Nature 294: 752–754, 1981. [DOI] [PubMed] [Google Scholar]
- 6.Ferrier GR, and Moe GK. Effect of calcium on acetylstrophanthidin-induced transient depolarizations in canine Purkinje tissue. Circ Res 33: 508–515, 1973. [DOI] [PubMed] [Google Scholar]
- 7.Lederer WJ, and Tsien RW. Transient inward current underlying arrhythmogenic effects of cardiotonic steroids in Purkinje fibres. J Physiol 263: 73–100, 1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Yellen G Single Ca2+-activated nonselective cation channels in neuroblastoma. Nature 296: 357–359, 1982. [DOI] [PubMed] [Google Scholar]
- 9.Kass RS, Lederer WJ, Tsien RW, and Weingart R. Role of calcium ions in transient inward currents and aftercontractions induced by strophanthidin in cardiac Purkinje fibres. J Physiol 281: 187–208, 1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kass RS, Tsien RW, and Weingart R. Ionic basis of transient inward current induced by strophanthidin in cardiac Purkinje fibres. J Physiol 281: 209–226, 1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Vu S, and Zheng J. TRPM4 and TRPM5 Channels as Chemical-to-Electrical Signal Converter. In: TRP-Mediated Signaling, edited by Zhu MX. Boca Raton, FL: Taylor and Francis, 2024. [Google Scholar]
- 12.Xu XZ, Moebius F, Gill DL, and Montell C. Regulation of melastatin, a TRP-related protein, through interaction with a cytoplasmic isoform. Proc Natl Acad Sci U S A 98: 10692–10697, 2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Launay P, Fleig A, Perraud AL, Scharenberg AM, Penner R, and Kinet JP. TRPM4 is a Ca2+-activated nonselective cation channel mediating cell membrane depolarization. Cell 109: 397–407, 2002. [DOI] [PubMed] [Google Scholar]
- 14.Clapham DE. TRP channels as cellular sensors. Nature 426: 517–524, 2003. [DOI] [PubMed] [Google Scholar]
- 15.Nilius B, Prenen J, Droogmans G, Voets T, Vennekens R, Freichel M, Wissenbach U, and Flockerzi V. Voltage dependence of the Ca2+-activated cation channel TRPM4. J Biol Chem 278: 30813–30820, 2003. [DOI] [PubMed] [Google Scholar]
- 16.Yoo JC, Yarishkin OV, Hwang EM, Kim E, Kim DG, Park N, Hong SG, and Park JY. Cloning and characterization of rat transient receptor potential-melastatin 4 (TRPM4). Biochem Biophys Res Commun 391: 806–811, 2010. [DOI] [PubMed] [Google Scholar]
- 17.Prawitt D, Enklaar T, Klemm G, Gartner B, Spangenberg C, Winterpacht A, Higgins M, Pelletier J, and Zabel B. Identification and characterization of MTR1, a novel gene with homology to melastatin (MLSN1) and the trp gene family located in the BWS-WT2 critical region on chromosome 11p15.5 and showing allele-specific expression. Hum Mol Genet 9: 203–216, 2000. [DOI] [PubMed] [Google Scholar]
- 18.Montell C, Birnbaumer L, Flockerzi V, Bindels RJ, Bruford EA, Caterina MJ, Clapham DE, Harteneck C, Heller S, Julius D, Kojima I, Mori Y, Penner R, Prawitt D, Scharenberg AM, Schultz G, Shimizu N, and Zhu MX. A unified nomenclature for the superfamily of TRP cation channels. Mol Cell 9: 229–231, 2002. [DOI] [PubMed] [Google Scholar]
- 19.Owsianik G, Talavera K, Voets T, and Nilius B. Permeation and selectivity of TRP channels. Annu Rev Physiol 68: 685–717, 2006. [DOI] [PubMed] [Google Scholar]
- 20.Liu D, and Liman ER. Intracellular Ca2+ and the phospholipid PIP2 regulate the taste transduction ion channel TRPM5. Proc Natl Acad Sci U S A 100: 15160–15165, 2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Nilius B, Prenen J, Voets T, and Droogmans G. Intracellular nucleotides and polyamines inhibit the Ca2+-activated cation channel TRPM4b. Pflugers Arch 448: 70–75, 2004. [DOI] [PubMed] [Google Scholar]
- 22.Guo J, She J, Zeng W, Chen Q, Bai XC, and Jiang Y. Structures of the calcium-activated, non-selective cation channel TRPM4. Nature 552: 205–209, 2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Winkler PA, Huang Y, Sun W, Du J, and Lu W. Electron cryo-microscopy structure of a human TRPM4 channel. Nature 552: 200–204, 2017. [DOI] [PubMed] [Google Scholar]
- 24.Autzen HE, Myasnikov AG, Campbell MG, Asarnow D, Julius D, and Cheng Y. Structure of the human TRPM4 ion channel in a lipid nanodisc. Science 359: 228–232, 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Duan J, Li Z, Li J, Santa-Cruz A, Sanchez-Martinez S, Zhang J, and Clapham DE. Structure of full-length human TRPM4. Proc Natl Acad Sci U S A 115: 2377–2382, 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hu J, Park SJ, Walter T, Orozco IJ, O’Dea G, Ye X, Du J, and Lu W. Physiological temperature drives TRPM4 ligand recognition and gating. Nature 630: 509–515, 2024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ruan Z, Haley E, Orozco IJ, Sabat M, Myers R, Roth R, Du J, and Lu W. Structures of the TRPM5 channel elucidate mechanisms of activation and inhibition. Nat Struct Mol Biol 28: 604–613, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Karuppan S, Schrag LG, Pastrano CM, Jara-Oseguera A, and Zubcevic L. Structural dynamics at cytosolic interprotomer interfaces control gating of a mammalian TRPM5 channel. Proc Natl Acad Sci U S A 121: e2403333121, 2024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Blanco C, Morales D, Mogollones I, Vergara-Jaque A, Vargas C, Alvarez A, Riquelme D, Leiva-Salcedo E, Gonzalez W, Morales D, Maureira D, Aldunate I, Caceres M, Varela D, and Cerda O. EB1- and EB2-dependent anterograde trafficking of TRPM4 regulates focal adhesion turnover and cell invasion. FASEB J 33: 9434–9452, 2019. [DOI] [PubMed] [Google Scholar]
- 30.Nilius B, Prenen J, Tang J, Wang C, Owsianik G, Janssens A, Voets T, and Zhu MX. Regulation of the Ca2+ sensitivity of the nonselective cation channel TRPM4. J Biol Chem 280: 6423–6433, 2005. [DOI] [PubMed] [Google Scholar]
- 31.Nilius B, Mahieu F, Prenen J, Janssens A, Owsianik G, Vennekens R, and Voets T. The Ca2+-activated cation channel TRPM4 is regulated by phosphatidylinositol 4,5-biphosphate. EMBO J 25: 467–478, 2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Launay P, Cheng H, Srivatsan S, Penner R, Fleig A, and Kinet JP. TRPM4 regulates calcium oscillations after T cell activation. Science 306: 1374–1377, 2004. [DOI] [PubMed] [Google Scholar]
- 33.Nilius B, Prenen J, Janssens A, Voets T, and Droogmans G. Decavanadate modulates gating of TRPM4 cation channels. J Physiol 560: 753–765, 2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zheng J, Zeng X, and Wang S. Calcium ion as cellular messenger. Sci China Life Sci 58: 1–5, 2015. [DOI] [PubMed] [Google Scholar]
- 35.Huang Y, Fliegert R, Guse AH, Lu W, and Du J. A structural overview of the ion channels of the TRPM family. Cell Calcium 85: 102111, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Baron J, Groschner K, and Tiapko O. Calcium transport and sensing in TRPC channels - New insights into a complex feedback regulation. Cell Calcium 116: 102816, 2023. [DOI] [PubMed] [Google Scholar]
- 37.Zheng J, and Ma L. Structure and function of the thermoTRP channel pore. Current topics in membranes 74: 233–257, 2014. [DOI] [PubMed] [Google Scholar]
- 38.Zheng J Molecular mechanism of TRP channels. Compr Physiol 3: 221–242, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Montell C. Physiology, phylogeny, and functions of the TRP superfamily of cation channels. Sci STKE 2001: re1, 2001. [DOI] [PubMed] [Google Scholar]
- 40.Doyle DA, Morais Cabral J, Pfuetzner RA, Kuo A, Gulbis JM, Cohen SL, Chait BT, and MacKinnon R. The structure of the potassium channel: molecular basis of K+ conduction and selectivity. Science 280: 69–77, 1998. [DOI] [PubMed] [Google Scholar]
- 41.Hofmann T, Chubanov V, Gudermann T, and Montell C. TRPM5 is a voltage-modulated and Ca(2+)-activated monovalent selective cation channel. Curr Biol 13: 1153–1158, 2003. [DOI] [PubMed] [Google Scholar]
- 42.Prawitt D, Monteilh-Zoller MK, Brixel L, Spangenberg C, Zabel B, Fleig A, and Penner R. TRPM5 is a transient Ca2+-activated cation channel responding to rapid changes in [Ca2+]i. Proc Natl Acad Sci U S A 100: 15166–15171, 2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ullrich ND, Voets T, Prenen J, Vennekens R, Talavera K, Droogmans G, and Nilius B. Comparison of functional properties of the Ca2+-activated cation channels TRPM4 and TRPM5 from mice. Cell Calcium 37: 267–278, 2005. [DOI] [PubMed] [Google Scholar]
- 44.Zhang Z, Okawa H, Wang Y, and Liman ER. Phosphatidylinositol 4,5-bisphosphate rescues TRPM4 channels from desensitization. J Biol Chem 280: 39185–39192, 2005. [DOI] [PubMed] [Google Scholar]
- 45.Zhang Y, Hoon MA, Chandrashekar J, Mueller KL, Cook B, Wu D, Zuker CS, and Ryba NJ. Coding of sweet, bitter, and umami tastes: different receptor cells sharing similar signaling pathways. Cell 112: 293–301, 2003. [DOI] [PubMed] [Google Scholar]
- 46.Al Tekreeti A, and Zheng J. Calcium inhibition of calcium activated TRPM4 current. Biophysical Society Annual Meeting abstract 2022. [Google Scholar]
- 47.Tian Y, Al Tekreeti A, and Zheng J. Complex waveform of the calcium-activated TRPM4 ion channel current. Biophysical Journal 123: 390A, 2024. [Google Scholar]
- 48.Simon F, Leiva-Salcedo E, Armisen R, Riveros A, Cerda O, Varela D, Eguiguren AL, Olivero P, and Stutzin A. Hydrogen peroxide removes TRPM4 current desensitization conferring increased vulnerability to necrotic cell death. J Biol Chem 285: 37150–37158, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Liu D, Zhang Z, and Liman ER. Extracellular acid block and acid-enhanced inactivation of the Ca2+-activated cation channel TRPM5 involve residues in the S3-S4 and S5-S6 extracellular domains. J Biol Chem 280: 20691–20699, 2005. [DOI] [PubMed] [Google Scholar]
- 50.Schoppa NE, McCormack K, Tanouye MA, and Sigworth FJ. The size of gating charge in wild-type and mutant Shaker potassium channels. Science 255: 1712–1715, 1992. [DOI] [PubMed] [Google Scholar]
- 51.Sigworth FJ. Voltage gating of ion channels. Q Rev Biophys 27: 1–40, 1994. [DOI] [PubMed] [Google Scholar]
- 52.Yang F, Xu L, Lee BH, Xiao X, Yarov-Yarovoy V, and Zheng J. An Unorthodox Mechanism Underlying Voltage Sensitivity of TRPV1 Ion Channel. Adv Sci (Weinh) 7: 2000575, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Voets T, Owsianik G, Janssens A, Talavera K, and Nilius B. TRPM8 voltage sensor mutants reveal a mechanism for integrating thermal and chemical stimuli. Nat Chem Biol 3: 174–182, 2007. [DOI] [PubMed] [Google Scholar]
- 54.Adelman JP. SK channels and calmodulin. Channels (Austin) 10: 1–6, 2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Yamaguchi S, Tanimoto A, Otsuguro K, Hibino H, and Ito S. Negatively charged amino acids near and in transient receptor potential (TRP) domain of TRPM4 channel are one determinant of its Ca2+ sensitivity. J Biol Chem 289: 35265–35282, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Demion M, Bois P, Launay P, and Guinamard R. TRPM4, a Ca2+-activated nonselective cation channel in mouse sino-atrial node cells. Cardiovasc Res 73: 531–538, 2007. [DOI] [PubMed] [Google Scholar]
- 57.Rohacs T, Lopes CM, Michailidis I, and Logothetis DE. PI(4,5)P2 regulates the activation and desensitization of TRPM8 channels through the TRP domain. Nat Neurosci 8: 626–634, 2005. [DOI] [PubMed] [Google Scholar]
- 58.Rohacs T, and Nilius B. Regulation of transient receptor potential (TRP) channels by phosphoinositides. Pflugers Arch 455: 157–168, 2007. [DOI] [PubMed] [Google Scholar]
- 59.Crnich R, Amberg GC, Leo MD, Gonzales AL, Tamkun MM, Jaggar JH, and Earley S. Vasoconstriction resulting from dynamic membrane trafficking of TRPM4 in vascular smooth muscle cells. Am J Physiol Cell Physiol 299: C682–694, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Nichols CG, and Lee SJ. Polyamines and potassium channels: A 25-year romance. J Biol Chem 293: 18779–18788, 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Bandell M, Macpherson LJ, and Patapoutian A. From chills to chilis: mechanisms for thermosensation and chemesthesis via thermoTRPs. Curr Opin Neurobiol 17: 490–497, 2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Julius D TRP channels and pain. Annu Rev Cell Dev Biol 29: 355–384, 2013. [DOI] [PubMed] [Google Scholar]
- 63.Talavera K, Yasumatsu K, Voets T, Droogmans G, Shigemura N, Ninomiya Y, Margolskee RF, and Nilius B. Heat activation of TRPM5 underlies thermal sensitivity of sweet taste. Nature 438: 1022–1025, 2005. [DOI] [PubMed] [Google Scholar]
- 64.Takezawa R, Cheng H, Beck A, Ishikawa J, Launay P, Kubota H, Kinet JP, Fleig A, Yamada T, and Penner R. A pyrazole derivative potently inhibits lymphocyte Ca2+ influx and cytokine production by facilitating transient receptor potential melastatin 4 channel activity. Mol Pharmacol 69: 1413–1420, 2006. [DOI] [PubMed] [Google Scholar]
- 65.Oike H, Wakamori M, Mori Y, Nakanishi H, Taguchi R, Misaka T, Matsumoto I, and Abe K. Arachidonic acid can function as a signaling modulator by activating the TRPM5 cation channel in taste receptor cells. Biochim Biophys Acta 1761: 1078–1084, 2006. [DOI] [PubMed] [Google Scholar]
- 66.Guinamard R, Simard C, and Del Negro C. Flufenamic acid as an ion channel modulator. Pharmacol Ther 138: 272–284, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Grand T, Demion M, Norez C, Mettey Y, Launay P, Becq F, Bois P, and Guinamard R. 9-phenanthrol inhibits human TRPM4 but not TRPM5 cationic channels. Br J Pharmacol 153: 1697–1705, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Garland CJ, Smirnov SV, Bagher P, Lim CS, Huang CY, Mitchell R, Stanley C, Pinkney A, and Dora KA. TRPM4 inhibitor 9-phenanthrol activates endothelial cell intermediate conductance calcium-activated potassium channels in rat isolated mesenteric artery. Br J Pharmacol 172: 1114–1123, 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Veress R, Baranyai D, Hegyi B, Kistamas K, Dienes C, Magyar J, Banyasz T, Nanasi PP, Szentandrassy N, and Horvath B. Transient receptor potential melastatin 4 channel inhibitor 9-phenanthrol inhibits K(+) but not Ca(2+) currents in canine ventricular myocytes. Can J Physiol Pharmacol 96: 1022–1029, 2018. [DOI] [PubMed] [Google Scholar]
- 70.Ozhathil LC, Delalande C, Bianchi B, Nemeth G, Kappel S, Thomet U, Ross-Kaschitza D, Simonin C, Rubin M, Gertsch J, Lochner M, Peinelt C, Reymond JL, and Abriel H. Identification of potent and selective small molecule inhibitors of the cation channel TRPM4. Br J Pharmacol 175: 2504–2519, 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Arullampalam P, Preti B, Ross-Kaschitza D, Lochner M, Rougier JS, and Abriel H. Species-Specific Effects of Cation Channel TRPM4 Small-Molecule Inhibitors. Front Pharmacol 12: 712354, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Uchida K, and Tominaga M. Extracellular zinc ion regulates transient receptor potential melastatin 5 (TRPM5) channel activation through its interaction with a pore loop domain. J Biol Chem 288: 25950–25955, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Chen M, and Simard JM. Cell swelling and a nonselective cation channel regulated by internal Ca2+ and ATP in native reactive astrocytes from adult rat brain. J Neurosci 21: 6512–6521, 2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Chen M, Dong Y, and Simard JM. Functional coupling between sulfonylurea receptor type 1 and a nonselective cation channel in reactive astrocytes from adult rat brain. J Neurosci 23: 8568–8577, 2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Woo SK, Kwon MS, Ivanov A, Gerzanich V, and Simard JM. The sulfonylurea receptor 1 (Sur1)-transient receptor potential melastatin 4 (Trpm4) channel. J Biol Chem 288: 3655–3667, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Stokum JA, Kwon MS, Woo SK, Tsymbalyuk O, Vennekens R, Gerzanich V, and Simard JM. SUR1-TRPM4 and AQP4 form a heteromultimeric complex that amplifies ion/water osmotic coupling and drives astrocyte swelling. Glia 66: 108–125, 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Sala-Rabanal M, Wang S, and Nichols CG. On potential interactions between non-selective cation channel TRPM4 and sulfonylurea receptor SUR1. J Biol Chem 287: 8746–8756, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Fonfria E, Murdock PR, Cusdin FS, Benham CD, Kelsell RE, and McNulty S. Tissue distribution profiles of the human TRPM cation channel family. J Recept Signal Transduct Res 26: 159–178, 2006. [DOI] [PubMed] [Google Scholar]
- 79.Jang Y, Lee Y, Kim SM, Yang YD, Jung J, and Oh U. Quantitative analysis of TRP channel genes in mouse organs. Arch Pharm Res 35: 1823–1830, 2012. [DOI] [PubMed] [Google Scholar]
- 80.Wyatt A, Wartenberg P, Candlish M, Krasteva-Christ G, Flockerzi V, and Boehm U. Genetic strategies to analyze primary TRP channel-expressing cells in mice. Cell Calcium 67: 91–104, 2017. [DOI] [PubMed] [Google Scholar]
- 81.Guinamard R, Hof T, and Del Negro CA. The TRPM4 channel inhibitor 9-phenanthrol. Br J Pharmacol 171: 1600–1613, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Hartmann AK, Aranda Lopez P, Zajac M, Freichel M, Schild H, Radsak MP, and Stassen M. 9-Phenanthrol enhances the generation of an CD8(+) T cell response following transcutaneous immunization with imiquimod in mice. J Dermatol Sci 87: 260–267, 2017. [DOI] [PubMed] [Google Scholar]
- 83.Burris SK, Wang Q, Bulley S, Neeb ZP, and Jaggar JH. 9-Phenanthrol inhibits recombinant and arterial myocyte TMEM16A channels. Br J Pharmacol 172: 2459–2468, 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Binkle-Ladisch L, Pironet A, Zaliani A, Alcouffe C, Mensching D, Haferkamp U, Willing A, Woo MS, Erdmann A, Jessen T, Hess SD, Gribbon P, Pless O, Vennekens R, and Friese MA. Identification and development of TRPM4 antagonists to counteract neuronal excitotoxicity. iScience 27: 2024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Piao H, Takahashi K, Yamaguchi Y, Wang C, Liu K, and Naruse K. Transient receptor potential melastatin-4 is involved in hypoxia-reoxygenation injury in the cardiomyocytes. PLoS One 10: e0121703, 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Son MJ, Kim JC, Kim SW, Chidipi B, Muniyandi J, Singh TD, So I, Subedi KP, and Woo SH. Shear stress activates monovalent cation channel transient receptor potential melastatin subfamily 4 in rat atrial myocytes via type 2 inositol 1,4,5-trisphosphate receptors and Ca(2+) release. J Physiol 594: 2985–3004, 2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Hof T, Salle L, Coulbault L, Richer R, Alexandre J, Rouet R, Manrique A, and Guinamard R. TRPM4 non-selective cation channels influence action potentials in rabbit Purkinje fibres. J Physiol 594: 295–306, 2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Demion M, Thireau J, Gueffier M, Finan A, Khoueiry Z, Cassan C, Serafini N, Aimond F, Granier M, Pasquie JL, Launay P, and Richard S. Trpm4 gene invalidation leads to cardiac hypertrophy and electrophysiological alterations. PLoS One 9: e115256, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Guo Y, Yu ZY, Wu J, Gong H, Kesteven S, Iismaa SE, Chan AY, Holman S, Pinto S, Pironet A, Cox CD, Graham RM, Vennekens R, Feneley MP, and Martinac B. The Ca(2+)-activated cation channel TRPM4 is a positive regulator of pressure overload-induced cardiac hypertrophy. Elife 10: 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Hedon C, Lambert K, Chakouri N, Thireau J, Aimond F, Cassan C, Bideaux P, Richard S, Faucherre A, Le Guennec JY, and Demion M. New role of TRPM4 channel in the cardiac excitation-contraction coupling in response to physiological and pathological hypertrophy in mouse. Prog Biophys Mol Biol 159: 105–117, 2021. [DOI] [PubMed] [Google Scholar]
- 91.Bers DM, and Grandi E. Ion Channels of the heart. In: Textbook of Ion Channels, edited by Zheng J, and Trudeau MC. Boca Raton, FL: CRC Press, 2023. [Google Scholar]
- 92.Guo J, Ono K, and Noma A. Monovalent cation conductance of the sustained inward current in rabbit sinoatrial node cells. Pflugers Arch 433: 209–211, 1996. [DOI] [PubMed] [Google Scholar]
- 93.Sasse P, Zhang J, Cleemann L, Morad M, Hescheler J, and Fleischmann BK. Intracellular Ca2+ oscillations, a potential pacemaking mechanism in early embryonic heart cells. J Gen Physiol 130: 133–144, 2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Hof T, Simard C, Rouet R, Salle L, and Guinamard R. Implication of the TRPM4 nonselective cation channel in mammalian sinus rhythm. Heart Rhythm 10: 1683–1689, 2013. [DOI] [PubMed] [Google Scholar]
- 95.Simard C, Hof T, Keddache Z, Launay P, and Guinamard R. The TRPM4 non-selective cation channel contributes to the mammalian atrial action potential. J Mol Cell Cardiol 59: 11–19, 2013. [DOI] [PubMed] [Google Scholar]
- 96.Kruse M, Schulze-Bahr E, Corfield V, Beckmann A, Stallmeyer B, Kurtbay G, Ohmert I, Schulze-Bahr E, Brink P, and Pongs O. Impaired endocytosis of the ion channel TRPM4 is associated with human progressive familial heart block type I. J Clin Invest 119: 2737–2744, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Dragun M, Gazova A, Kyselovic J, Hulman M, and Matus M. TRP Channels Expression Profile in Human End-Stage Heart Failure. Medicina (Kaunas) 55: 2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Frede W, Medert R, Poth T, Gorenflo M, Vennekens R, Freichel M, and Uhl S. TRPM4 Modulates Right Ventricular Remodeling Under Pressure Load Accompanied With Decreased Expression Level. J Card Fail 26: 599–609, 2020. [DOI] [PubMed] [Google Scholar]
- 99.Feng J, Zong P, Yan J, Yue Z, Li X, Smith C, Ai X, and Yue L. Upregulation of transient receptor potential melastatin 4 (TRPM4) in ventricular fibroblasts from heart failure patients. Pflugers Arch 473: 521–531, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Thilo F, Suess O, Liu Y, and Tepel M. Decreased expression of transient receptor potential channels in cerebral vascular tissue from patients after hypertensive intracerebral hemorrhage. Clin Exp Hypertens 33: 533–537, 2011. [DOI] [PubMed] [Google Scholar]
- 101.Echeverria C, Montorfano I, Cabello-Verrugio C, Armisen R, Varela D, and Simon F. Suppression of transient receptor potential melastatin 4 expression promotes conversion of endothelial cells into fibroblasts via transforming growth factor/activin receptor-like kinase 5 pathway. J Hypertens 33: 981–992, 2015. [DOI] [PubMed] [Google Scholar]
- 102.Yang XR, Lin MJ, McIntosh LS, and Sham JS. Functional expression of transient receptor potential melastatin- and vanilloid-related channels in pulmonary arterial and aortic smooth muscle. Am J Physiol Lung Cell Mol Physiol 290: L1267–1276, 2006. [DOI] [PubMed] [Google Scholar]
- 103.Kecskes M, Jacobs G, Kerselaers S, Syam N, Menigoz A, Vangheluwe P, Freichel M, Flockerzi V, Voets T, and Vennekens R. The Ca(2+)-activated cation channel TRPM4 is a negative regulator of angiotensin II-induced cardiac hypertrophy. Basic Res Cardiol 110: 43, 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Gueffier M, Zintz J, Lambert K, Finan A, Aimond F, Chakouri N, Hedon C, Granier M, Launay P, Thireau J, Richard S, and Demion M. The TRPM4 channel is functionally important for the beneficial cardiac remodeling induced by endurance training. J Muscle Res Cell Motil 38: 3–16, 2017. [DOI] [PubMed] [Google Scholar]
- 105.Guinamard R, Demion M, Magaud C, Potreau D, and Bois P. Functional expression of the TRPM4 cationic current in ventricular cardiomyocytes from spontaneously hypertensive rats. Hypertension 48: 587–594, 2006. [DOI] [PubMed] [Google Scholar]
- 106.Guo Y, Cheng D, Yu ZY, Schiatti T, Chan AY, Hill AP, Peyronnet R, Feneley MP, Cox CD, and Martinac B. Functional coupling between Piezo1 and TRPM4 influences the electrical activity of HL-1 atrial myocytes. J Physiol 2023. [DOI] [PubMed] [Google Scholar]
- 107.Medert R, Jungmann A, Hildebrand S, Busch M, Grimm D, Flockerzi V, Muller OJ, Most P, Schumacher D, and Freichel M. Development of an AAV9-RNAi-mediated silencing strategy to abrogate TRPM4 expression in the adult heart. Pflugers Arch 473: 533–546, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Medert R, Pironet A, Bacmeister L, Segin S, Londono JEC, Vennekens R, and Freichel M. Genetic background influences expression and function of the cation channel TRPM4 in the mouse heart. Basic Res Cardiol 115: 70, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Arullampalam P, Essers MC, Boukenna M, Guichard S, Rougier JS, and Abriel H. Knockdown of the TRPM4 channel alters cardiac electrophysiology and hemodynamics in a sex- and age-dependent manner in mice. Physiol Rep 11: e15783, 2023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Jackson WF. Ion channels and the regulation of myogenic tone in peripheral arterioles. Curr Top Membr 85: 19–58, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Morita H, Honda A, Inoue R, Ito Y, Abe K, Nelson MT, and Brayden JE. Membrane stretch-induced activation of a TRPM4-like nonselective cation channel in cerebral artery myocytes. J Pharmacol Sci 103: 417–426, 2007. [DOI] [PubMed] [Google Scholar]
- 112.Earley S, Waldron BJ, and Brayden JE. Critical role for transient receptor potential channel TRPM4 in myogenic constriction of cerebral arteries. Circ Res 95: 922–929, 2004. [DOI] [PubMed] [Google Scholar]
- 113.Earley S TRPM4 channels in smooth muscle function. Pflugers Arch 465: 1223–1231, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Gonzales AL, Amberg GC, and Earley S. Ca2+ release from the sarcoplasmic reticulum is required for sustained TRPM4 activity in cerebral artery smooth muscle cells. Am J Physiol Cell Physiol 299: C279–288, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Garcia ZI, Bruhl A, Gonzales AL, and Earley S. Basal protein kinase Cdelta activity is required for membrane localization and activity of TRPM4 channels in cerebral artery smooth muscle cells. Channels (Austin) 5: 210–214, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Yamasaki E, Ali S, Sanchez Solano A, Thakore P, Smith M, Wang X, Labelle-Dumais C, Gould DB, and Earley S. Faulty TRPM4 channels underlie age-dependent cerebral vascular dysfunction in Gould syndrome. Proc Natl Acad Sci U S A 120: e2217327120, 2023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Ali S, Solano AS, Gonzales AL, Thakore P, Krishnan V, Yamasaki E, and Earley S. Nitric Oxide Signals Through IRAG to Inhibit TRPM4 Channels and Dilate Cerebral Arteries. Function (Oxf) 2: zqab051, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Gaur N, Hof T, Haissaguerre M, and Vigmond EJ. Propagation Failure by TRPM4 Overexpression. Biophys J 116: 469–476, 2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Li Y, Baylie RL, Tavares MJ, and Brayden JE. TRPM4 channels couple purinergic receptor mechanoactivation and myogenic tone development in cerebral parenchymal arterioles. J Cereb Blood Flow Metab 34: 1706–1714, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Ding XQ, Ban T, Liu ZY, Lou J, Tang LL, Wang JX, Chu WF, Zhao D, Song BL, and Zhang ZR. Transient Receptor Potential Melastatin 4 (TRPM4) Contributes to High Salt Diet-Mediated Early-Stage Endothelial Injury. Cell Physiol Biochem 41: 835–848, 2017. [DOI] [PubMed] [Google Scholar]
- 121.Csipo T, Czikora A, Fulop GA, Gulyas H, Rutkai I, Toth EP, Porszasz R, Szalai A, Bolcskei K, Helyes Z, Pinter E, Papp Z, Ungvari Z, and Toth A. A Central Role for TRPM4 in Ca(2+)-Signal Amplification and Vasoconstriction. Int J Mol Sci 23: 2022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Gong Y, Du MY, Yu HL, Yang ZY, Li YJ, Zhou L, Mei R, Yang L, and Wang F. Increased TRPM4 Activity in Cerebral Artery Myocytes Contributes to Cerebral Blood Flow Reduction After Subarachnoid Hemorrhage in Rats. Neurotherapeutics 16: 901–911, 2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Gerzanich V, Woo SK, Vennekens R, Tsymbalyuk O, Ivanova S, Ivanov A, Geng Z, Chen Z, Nilius B, Flockerzi V, Freichel M, and Simard JM. De novo expression of Trpm4 initiates secondary hemorrhage in spinal cord injury. Nat Med 15: 185–191, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Gonzales AL, and Earley S. Endogenous cytosolic Ca(2+) buffering is necessary for TRPM4 activity in cerebral artery smooth muscle cells. Cell Calcium 51: 82–93, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Lewis RS. Calcium signaling mechanisms in T lymphocytes. Annu Rev Immunol 19: 497–521, 2001. [DOI] [PubMed] [Google Scholar]
- 126.Feske S, Wulff H, and Skolnik EY. Ion channels in innate and adaptive immunity. Annu Rev Immunol 33: 291–353, 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Cahalan MD. Voltage-gated Ca(2+) channel proteins do not function as ion channels in T cells. Sci Signal 15: eabq5594, 2022. [DOI] [PubMed] [Google Scholar]
- 128.Cahalan MD, and DeCoursey TE. Ion channels in immune cells. In: Textbook of Ion Channels 2023. [Google Scholar]
- 129.Weber KS, Hildner K, Murphy KM, and Allen PM. Trpm4 differentially regulates Th1 and Th2 function by altering calcium signaling and NFAT localization. J Immunol 185: 2836–2846, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Ozcan SS, Gurel G, and Cakir M. Gene expression profiles of transient receptor potential (TRP) channels in the peripheral blood mononuclear cells of psoriasis patients. Hum Exp Toxicol 40: 1234–1240, 2021. [DOI] [PubMed] [Google Scholar]
- 131.Barbet G, Demion M, Moura IC, Serafini N, Leger T, Vrtovsnik F, Monteiro RC, Guinamard R, Kinet JP, and Launay P. The calcium-activated nonselective cation channel TRPM4 is essential for the migration but not the maturation of dendritic cells. Nat Immunol 9: 1148–1156, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Vennekens R, Olausson J, Meissner M, Bloch W, Mathar I, Philipp SE, Schmitz F, Weissgerber P, Nilius B, Flockerzi V, and Freichel M. Increased IgE-dependent mast cell activation and anaphylactic responses in mice lacking the calcium-activated nonselective cation channel TRPM4. Nat Immunol 8: 312–320, 2007. [DOI] [PubMed] [Google Scholar]
- 133.Cakir M, Sacmaci H, and Sabah-Ozcan S. Selected transient receptor potential channel genes’ expression in peripheral blood mononuclear cells of multiple sclerosis. Hum Exp Toxicol 40: S406–S413, 2021. [DOI] [PubMed] [Google Scholar]
- 134.Malhotra S, Castillo J, Negrotto L, Merino-Zamorano C, Montaner J, Vidal-Jordana A, Montalban X, and Comabella M. TRPM4 mRNA expression levels in peripheral blood mononuclear cells from multiple sclerosis patients. J Neuroimmunol 261: 146–148, 2013. [DOI] [PubMed] [Google Scholar]
- 135.Morita T, Mitsuyama K, Yamasaki H, Mori A, Yoshimura T, Araki T, Morita M, Tsuruta K, Yamasaki S, Kuwaki K, Yoshioka S, Takedatsu H, and Torimura T. Gene Expression of Transient Receptor Potential Channels in Peripheral Blood Mononuclear Cells of Inflammatory Bowel Disease Patients. J Clin Med 9: 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Wang H, Xu Z, Lee BH, Vu S, Hu L, Lee M, Bu D, Cao X, Hwang S, Yang Y, Zheng J, and Lin Z. Gain-of-Function Mutations in TRPM4 Activation Gate Cause Progressive Symmetric Erythrokeratodermia. J Invest Dermatol 139: 1089–1097, 2019. [DOI] [PubMed] [Google Scholar]
- 137.Otsuka Saito K, Fujita F, Toriyama M, Utami RA, Guo Z, Murakami M, Kato H, Suzuki Y, Okada F, Tominaga M, and Ishii KJ. Roles of TRPM4 in immune responses in keratinocytes and identification of a novel TRPM4-activating agent. Biochem Biophys Res Commun 654: 1–9, 2023. [DOI] [PubMed] [Google Scholar]
- 138.Vennekens R, and Nilius B. Insights into TRPM4 function, regulation and physiological role. Handb Exp Pharmacol 269–285, 2007. [DOI] [PubMed] [Google Scholar]
- 139.Yamada D, Vu S, Wu X, Shi Z, Morris D, Bloomstein JD, Huynh M, Zheng J, and Hwang ST. Gain-of-function of TRPM4 predisposes mice to psoriasiform dermatitis. Front Immunol 13: 1025499, 2022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Hsieh DM, Shang P, Gao Y, and Zhao J. Successful treatment with spesolimab for generalized pustular psoriasis in an elderly patient with TRPM4 mutation. J Dermatol 2024. [DOI] [PubMed] [Google Scholar]
- 141.Schattling B, Steinbach K, Thies E, Kruse M, Menigoz A, Ufer F, Flockerzi V, Bruck W, Pongs O, Vennekens R, Kneussel M, Freichel M, Merkler D, and Friese MA. TRPM4 cation channel mediates axonal and neuronal degeneration in experimental autoimmune encephalomyelitis and multiple sclerosis. Nat Med 18: 1805–1811, 2012. [DOI] [PubMed] [Google Scholar]
- 142.Yan J, Bengtson CP, Buchthal B, Hagenston AM, and Bading H. Coupling of NMDA receptors and TRPM4 guides discovery of unconventional neuroprotectants. Science 370: 2020. [DOI] [PubMed] [Google Scholar]
- 143.Riquelme D, Silva I, Philp AM, Huidobro-Toro JP, Cerda O, Trimmer JS, and Leiva-Salcedo E. Subcellular Localization and Activity of TRPM4 in Medial Prefrontal Cortex Layer 2/3. Front Cell Neurosci 12: 12, 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Riquelme D, Cerda O, and Leiva-Salcedo E. TRPM4 Expression During Postnatal Developmental of Mouse CA1 Pyramidal Neurons. Front Neuroanat 15: 643287, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Menigoz A, Ahmed T, Sabanov V, Philippaert K, Pinto S, Kerselaers S, Segal A, Freichel M, Voets T, Nilius B, Vennekens R, and Balschun D. TRPM4-dependent post-synaptic depolarization is essential for the induction of NMDA receptor-dependent LTP in CA1 hippocampal neurons. Pflugers Arch 468: 593–607, 2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Bovet-Carmona M, Menigoz A, Pinto S, Tambuyzer T, Krautwald K, Voets T, Aerts JM, Angenstein F, Vennekens R, and Balschun D. Disentangling the role of TRPM4 in hippocampus-dependent plasticity and learning: an electrophysiological, behavioral and FMRI approach. Brain Struct Funct 223: 3557–3576, 2018. [DOI] [PubMed] [Google Scholar]
- 147.Armstrong WE, Wang L, Li C, and Teruyama R. Performance, properties and plasticity of identified oxytocin and vasopressin neurones in vitro. J Neuroendocrinol 22: 330–342, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Cho CH, Kim E, Lee YS, Yarishkin O, Yoo JC, Park JY, Hong SG, and Hwang EM. Depletion of 14–3-3gamma reduces the surface expression of Transient Receptor Potential Melastatin 4b (TRPM4b) channels and attenuates TRPM4b-mediated glutamate-induced neuronal cell death. Mol Brain 7: 52, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Combe CL, Upchurch CM, Canavier CC, and Gasparini S. Cholinergic modulation shifts the response of CA1 pyramidal cells to depolarizing ramps via TRPM4 channels with potential implications for place field firing. Elife 12: 2023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Mundrucz L, Kecskes A, Henn-Mike N, Kobor P, Buzas P, Vennekens R, and Kecskes M. TRPM4 regulates hilar mossy cell loss in temporal lobe epilepsy. BMC Biol 21: 96, 2023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Yarishkin O, Phuong TTT, Vazquez-Chona F, Bertrand J, van Battenburg-Sherwood J, Redmon SN, Rudzitis CN, Lakk M, Baumann JM, Freichel M, Hwang EM, Overby D, and Krizaj D. Emergent Temporal Signaling in Human Trabecular Meshwork Cells: Role of TRPV4-TRPM4 Interactions. Front Immunol 13: 805076, 2022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Choi HJ, Sun D, and Jakobs TC. Astrocytes in the optic nerve head express putative mechanosensitive channels. Mol Vis 21: 749–766, 2015. [PMC free article] [PubMed] [Google Scholar]
- 153.Bianchi B, Smith PA, and Abriel H. The ion channel TRPM4 in murine experimental autoimmune encephalomyelitis and in a model of glutamate-induced neuronal degeneration. Mol Brain 11: 41, 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Ma P, Huang N, Tang J, Zhou Z, Xu J, Chen Y, Zhang M, Huang Q, and Cheng Y. The TRPM4 channel inhibitor 9-phenanthrol alleviates cerebral edema after traumatic brain injury in rats. Front Pharmacol 14: 1098228, 2023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Tosun C, Kurland DB, Mehta R, Castellani RJ, deJong JL, Kwon MS, Woo SK, Gerzanich V, and Simard JM. Inhibition of the Sur1-Trpm4 channel reduces neuroinflammation and cognitive impairment in subarachnoid hemorrhage. Stroke 44: 3522–3528, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Huang K, Gu Y, Hu Y, Ji Z, Wang S, Lin Z, Li X, Xie Z, and Pan S. Glibenclamide Improves Survival and Neurologic Outcome After Cardiac Arrest in Rats. Crit Care Med 43: e341–349, 2015. [DOI] [PubMed] [Google Scholar]
- 157.Huang K, Wang Z, Gu Y, Hu Y, Ji Z, Wang S, Lin Z, Li X, Xie Z, and Pan S. Glibenclamide Is Comparable to Target Temperature Management in Improving Survival and Neurological Outcome After Asphyxial Cardiac Arrest in Rats. J Am Heart Assoc 5: 2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Teruyama R, Sakuraba M, Kurotaki H, and Armstrong WE. Transient receptor potential channel m4 and m5 in magnocellular cells in rat supraoptic and paraventricular nuclei. J Neuroendocrinol 23: 1204–1213, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Herrera Moro Chao D, Argmann C, Van Eijk M, Boot RG, Ottenhoff R, Van Roomen C, Foppen E, Siljee JE, Unmehopa UA, Kalsbeek A, and Aerts JM. Impact of obesity on taste receptor expression in extra-oral tissues: emphasis on hypothalamus and brainstem. Sci Rep 6: 29094, 2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Yu Q, Gamayun I, Wartenberg P, Zhang Q, Qiao S, Kusumakshi S, Candlish S, Gotz V, Wen S, Das D, Wyatt A, Wahl V, Ectors F, Kattler K, Yildiz D, Prevot V, Schwaninger M, Ternier G, Giacobini P, Ciofi P, Muller TD, and Boehm U. Bitter taste cells in the ventricular walls of the murine brain regulate glucose homeostasis. Nat Commun 14: 1588, 2023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Crowder EA, Saha MS, Pace RW, Zhang H, Prestwich GD, and Del Negro CA. Phosphatidylinositol 4,5-bisphosphate regulates inspiratory burst activity in the neonatal mouse preBotzinger complex. J Physiol 582: 1047–1058, 2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Thompson JA, Salcedo E, Restrepo D, and Finger TE. Second-order input to the medial amygdala from olfactory sensory neurons expressing the transduction channel TRPM5. J Comp Neurol 520: 1819–1830, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Bos R, Drouillas B, Bouhadfane M, Pecchi E, Trouplin V, Korogod SM, and Brocard F. Trpm5 channels encode bistability of spinal motoneurons and ensure motor control of hindlimbs in mice. Nat Commun 12: 6815, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Kim YS, Kang E, Makino Y, Park S, Shin JH, Song H, Launay P, and Linden DJ. Characterizing the conductance underlying depolarization-induced slow current in cerebellar Purkinje cells. J Neurophysiol 109: 1174–1181, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Koizumi H, John TT, Chia JX, Tariq MF, Phillips RS, Mosher B, Chen Y, Thompson R, Zhang R, Koshiya N, and Smith JC. Transient Receptor Potential Channels TRPM4 and TRPC3 Critically Contribute to Respiratory Motor Pattern Formation but not Rhythmogenesis in Rodent Brainstem Circuits. eNeuro 5: 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Li K, Abbott SBG, Shi Y, Eggan P, Gonye EC, and Bayliss DA. TRPM4 mediates a subthreshold membrane potential oscillation in respiratory chemoreceptor neurons that drives pacemaker firing and breathing. Cell Rep 34: 108714, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Tsymbalyuk O, Gerzanich V, Mumtaz A, Andhavarapu S, Ivanova S, Makar TK, Sansur CA, Keller A, Nakamura Y, Bryan J, and Simard JM. SUR1, newly expressed in astrocytes, mediates neuropathic pain in a mouse model of peripheral nerve injury. Mol Pain 17: 17448069211006603, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Turtle JD, Henwood MK, Strain MM, Huang YJ, Miranda RC, and Grau JW. Engaging pain fibers after a spinal cord injury fosters hemorrhage and expands the area of secondary injury. Exp Neurol 311: 115–124, 2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Vandewauw I, Owsianik G, and Voets T. Systematic and quantitative mRNA expression analysis of TRP channel genes at the single trigeminal and dorsal root ganglion level in mouse. BMC Neurosci 14: 21, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Yao Y, Xu J, Yu T, Chen Z, Xiao Z, Wang J, Hu Y, Wu Y, and Zhu D. Flufenamic acid inhibits secondary hemorrhage and BSCB disruption after spinal cord injury. Theranostics 8: 4181–4198, 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Enklaar T, Esswein M, Oswald M, Hilbert K, Winterpacht A, Higgins M, Zabel B, and Prawitt D. Mtr1, a novel biallelically expressed gene in the center of the mouse distal chromosome 7 imprinting cluster, is a member of the Trp gene family. Genomics 67: 179–187, 2000. [DOI] [PubMed] [Google Scholar]
- 172.Perez CA, Huang L, Rong M, Kozak JA, Preuss AK, Zhang H, Max M, and Margolskee RF. A transient receptor potential channel expressed in taste receptor cells. Nat Neurosci 5: 1169–1176, 2002. [DOI] [PubMed] [Google Scholar]
- 173.Rong M, He W, Yasumatsu K, Kokrashvili Z, Perez CA, Mosinger B, Ninomiya Y, Margolskee RF, and Damak S. Signal transduction of umami taste: insights from knockout mice. Chem Senses 30 Suppl 1: i33–34, 2005. [DOI] [PubMed] [Google Scholar]
- 174.Oliveira-Maia AJ, Stapleton-Kotloski JR, Lyall V, Phan TH, Mummalaneni S, Melone P, Desimone JA, Nicolelis MA, and Simon SA. Nicotine activates TRPM5-dependent and independent taste pathways. Proc Natl Acad Sci U S A 106: 1596–1601, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.de Araujo IE, Oliveira-Maia AJ, Sotnikova TD, Gainetdinov RR, Caron MG, Nicolelis MA, and Simon SA. Food reward in the absence of taste receptor signaling. Neuron 57: 930–941, 2008. [DOI] [PubMed] [Google Scholar]
- 176.Zhang Z, Zhao Z, Margolskee R, and Liman E. The transduction channel TRPM5 is gated by intracellular calcium in taste cells. J Neurosci 27: 5777–5786, 2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Huang YA, and Roper SD. Intracellular Ca(2+) and TRPM5-mediated membrane depolarization produce ATP secretion from taste receptor cells. J Physiol 588: 2343–2350, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Perez CA, Margolskee RF, Kinnamon SC, and Ogura T. Making sense with TRP channels: store-operated calcium entry and the ion channel Trpm5 in taste receptor cells. Cell Calcium 33: 541–549, 2003. [DOI] [PubMed] [Google Scholar]
- 179.Sclafani A, and Ackroff K. Fat preference deficits and experience-induced recovery in global taste-deficient Trpm5 and Calhm1 knockout mice. Physiol Behav 246: 113695, 2022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Sclafani A, Zukerman S, Glendinning JI, and Margolskee RF. Fat and carbohydrate preferences in mice: the contribution of alpha-gustducin and Trpm5 taste-signaling proteins. Am J Physiol Regul Integr Comp Physiol 293: R1504–1513, 2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Glendinning JI, Gillman J, Zamer H, Margolskee RF, and Sclafani A. The role of T1r3 and Trpm5 in carbohydrate-induced obesity in mice. Physiol Behav 107: 50–58, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Talavera K, Yasumatsu K, Yoshida R, Margolskee RF, Voets T, Ninomiya Y, and Nilius B. The taste transduction channel TRPM5 is a locus for bitter-sweet taste interactions. FASEB J 22: 1343–1355, 2008. [DOI] [PubMed] [Google Scholar]
- 183.Clapp TR, Medler KF, Damak S, Margolskee RF, and Kinnamon SC. Mouse taste cells with G protein-coupled taste receptors lack voltage-gated calcium channels and SNAP-25. BMC Biol 4: 7, 2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Gonda S, Matsumura S, Saito S, Go Y, and Imai H. Expression of taste signal transduction molecules in the caecum of common marmosets. Biol Lett 9: 20130409, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Nguyen QT, Beck Coburn GE, Valentino A, Karabucak B, and Tizzano M. Mouse Mandibular Retromolar Taste Buds Associated With a Mucus Salivary Gland. Chem Senses 46: 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Yoshida Y, Kawabata F, Kawabata Y, Nishimura S, and Tabata S. Expression levels of taste-related genes in palate and tongue tip, and involvement of transient receptor potential subfamily M member 5 (TRPM5) in taste sense in chickens. Anim Sci J 89: 441–447, 2018. [DOI] [PubMed] [Google Scholar]
- 187.Cui Y, Wu H, Li Q, Liao J, Gao P, Sun F, Zhang H, Lu Z, Wei X, He C, Ma T, Wei X, Chen X, Zheng H, Yang G, Liu D, and Zhu Z. Impairment of Bitter Taste Sensor Transient Receptor Potential Channel M5-Mediated Aversion Aggravates High-Salt Intake and Hypertension. Hypertension 74: 1021–1032, 2019. [DOI] [PubMed] [Google Scholar]
- 188.Wu H, Cui Y, He C, Gao P, Li Q, Zhang H, Jiang Y, Hu Y, Wei X, Lu Z, Ma T, Liu D, and Zhu Z. Activation of the bitter taste sensor TRPM5 prevents high salt-induced cardiovascular dysfunction. Sci China Life Sci 63: 1665–1677, 2020. [DOI] [PubMed] [Google Scholar]
- 189.Liu P, Shah BP, Croasdell S, and Gilbertson TA. Transient receptor potential channel type M5 is essential for fat taste. J Neurosci 31: 8634–8642, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Lin W, Margolskee R, Donnert G, Hell SW, and Restrepo D. Olfactory neurons expressing transient receptor potential channel M5 (TRPM5) are involved in sensing semiochemicals. Proc Natl Acad Sci U S A 104: 2471–2476, 2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Fu Z, Ogura T, Luo W, and Lin W. ATP and Odor Mixture Activate TRPM5-Expressing Microvillous Cells and Potentially Induce Acetylcholine Release to Enhance Supporting Cell Endocytosis in Mouse Main Olfactory Epithelium. Front Cell Neurosci 12: 71, 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Lemons K, Fu Z, Aoude I, Ogura T, Sun J, Chang J, Mbonu K, Matsumoto I, Arakawa H, and Lin W. Lack of TRPM5-Expressing Microvillous Cells in Mouse Main Olfactory Epithelium Leads to Impaired Odor-Evoked Responses and Olfactory-Guided Behavior in a Challenging Chemical Environment. eNeuro 4: 2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Lemons K, Fu Z, Ogura T, and Lin W. TRPM5-expressing Microvillous Cells Regulate Region-specific Cell Proliferation and Apoptosis During Chemical Exposure. Neuroscience 434: 171–190, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Tizzano M, Gulbransen BD, Vandenbeuch A, Clapp TR, Herman JP, Sibhatu HM, Churchill ME, Silver WL, Kinnamon SC, and Finger TE. Nasal chemosensory cells use bitter taste signaling to detect irritants and bacterial signals. Proc Natl Acad Sci U S A 107: 3210–3215, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Baxter BD, Larson ED, Merle L, Feinstein P, Polese AG, Bubak AN, Niemeyer CS, Hassell J, Jr., Shepherd D, Ramakrishnan VR, Nagel MA, and Restrepo D. Transcriptional profiling reveals potential involvement of microvillous TRPM5-expressing cells in viral infection of the olfactory epithelium. BMC Genomics 22: 224, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Lin W, Ezekwe EA Jr., Zhao Z, Liman ER, and Restrepo D. TRPM5-expressing microvillous cells in the main olfactory epithelium. BMC Neurosci 9: 114, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Lopez F, Delgado R, Lopez R, Bacigalupo J, and Restrepo D. Transduction for pheromones in the main olfactory epithelium is mediated by the Ca2+ -activated channel TRPM5. J Neurosci 34: 3268–3278, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Oshimoto A, Wakabayashi Y, Garske A, Lopez R, Rolen S, Flowers M, Arevalo N, and Restrepo D. Potential role of transient receptor potential channel M5 in sensing putative pheromones in mouse olfactory sensory neurons. PLoS One 8: e61990, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Pyrski M, Eckstein E, Schmid A, Bufe B, Weiss J, Chubanov V, Boehm U, and Zufall F. Trpm5 expression in the olfactory epithelium. Mol Cell Neurosci 80: 75–88, 2017. [DOI] [PubMed] [Google Scholar]
- 200.Yamaguchi T, Yamashita J, Ohmoto M, Aoude I, Ogura T, Luo W, Bachmanov AA, Lin W, Matsumoto I, and Hirota J. Skn-1a/Pou2f3 is required for the generation of Trpm5-expressing microvillous cells in the mouse main olfactory epithelium. BMC Neurosci 15: 13, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Genovese F, and Tizzano M. Microvillous cells in the olfactory epithelium express elements of the solitary chemosensory cell transduction signaling cascade. PLoS One 13: e0202754, 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Kaske S, Krasteva G, Konig P, Kummer W, Hofmann T, Gudermann T, and Chubanov V. TRPM5, a taste-signaling transient receptor potential ion-channel, is a ubiquitous signaling component in chemosensory cells. BMC Neurosci 8: 49, 2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Nakashimo Y, Takumida M, Fukuiri T, Anniko M, and Hirakawa K. Expression of transient receptor potential channel vanilloid (TRPV) 1–4, melastin (TRPM) 5 and 8, and ankyrin (TRPA1) in the normal and methimazole-treated mouse olfactory epithelium. Acta Otolaryngol 130: 1278–1286, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Kokrashvili Z, Rodriguez D, Yevshayeva V, Zhou H, Margolskee RF, and Mosinger B. Release of endogenous opioids from duodenal enteroendocrine cells requires Trpm5. Gastroenterology 137: 598–606, 606 e591–592, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Hollenhorst MI, Jurastow I, Nandigama R, Appenzeller S, Li L, Vogel J, Wiederhold S, Althaus M, Empting M, Altmuller J, Hirsch AKH, Flockerzi V, Canning BJ, Saliba AE, and Krasteva-Christ G. Tracheal brush cells release acetylcholine in response to bitter tastants for paracrine and autocrine signaling. FASEB J 34: 316–332, 2020. [DOI] [PubMed] [Google Scholar]
- 206.Dutta Banik D, and Medler KF. Defining the role of TRPM4 in broadly responsive taste receptor cells. Front Cell Neurosci 17: 1148995, 2023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Dutta Banik D, Martin LE, Freichel M, Torregrossa AM, and Medler KF. TRPM4 and TRPM5 are both required for normal signaling in taste receptor cells. Proc Natl Acad Sci U S A 115: E772–E781, 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Young RL, Sutherland K, Pezos N, Brierley SM, Horowitz M, Rayner CK, and Blackshaw LA. Expression of taste molecules in the upper gastrointestinal tract in humans with and without type 2 diabetes. Gut 58: 337–346, 2009. [DOI] [PubMed] [Google Scholar]
- 209.Widmayer P, Kuper M, Kramer M, Konigsrainer A, and Breer H. Altered expression of gustatory-signaling elements in gastric tissue of morbidly obese patients. Int J Obes (Lond) 36: 1353–1359, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Bezencon C, Furholz A, Raymond F, Mansourian R, Metairon S, Le Coutre J, and Damak S. Murine intestinal cells expressing Trpm5 are mostly brush cells and express markers of neuronal and inflammatory cells. J Comp Neurol 509: 514–525, 2008. [DOI] [PubMed] [Google Scholar]
- 211.Feng R, Qian C, Liu Q, Jin Y, Liu L, Li S, Liao Y, Zhou H, Liu W, Rayner CK, and Ma J. Expression of sweet taste receptor and gut hormone secretion in modelled type 2 diabetes. Gen Comp Endocrinol 252: 142–149, 2017. [DOI] [PubMed] [Google Scholar]
- 212.Howitt MR, Lavoie S, Michaud M, Blum AM, Tran SV, Weinstock JV, Gallini CA, Redding K, Margolskee RF, Osborne LC, Artis D, and Garrett WS. Tuft cells, taste-chemosensory cells, orchestrate parasite type 2 immunity in the gut. Science 351: 1329–1333, 2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Kang C, Wang L, Feng J, Zhu W, and Hang S. l-Glutamate stimulates cholecystokinin secretion via the T1R1/T1R3 mediated PLC/TRPM5 transduction pathway. J Sci Food Agric 100: 4818–4825, 2020. [DOI] [PubMed] [Google Scholar]
- 214.Zheng CY, An T, Liang ZT, Lv BH, Liu YT, Hu XH, Zhang YL, Liu NN, Tao SY, Deng RX, Liu JX, and Jiang GJ. Revealing the mechanism of quinoa on type 2 diabetes based on intestinal flora and taste pathways. Food Sci Nutr 11: 7930–7945, 2023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Schneider C, O’Leary CE, von Moltke J, Liang HE, Ang QY, Turnbaugh PJ, Radhakrishnan S, Pellizzon M, Ma A, and Locksley RM. A Metabolite-Triggered Tuft Cell-ILC2 Circuit Drives Small Intestinal Remodeling. Cell 174: 271–284 e214, 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Luo XC, Chen ZH, Xue JB, Zhao DX, Lu C, Li YH, Li SM, Du YW, Liu Q, Wang P, Liu M, and Huang L. Infection by the parasitic helminth Trichinella spiralis activates a Tas2r-mediated signaling pathway in intestinal tuft cells. Proc Natl Acad Sci U S A 116: 5564–5569, 2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Mitrovic S, Nogueira C, Cantero-Recasens G, Kiefer K, Fernandez-Fernandez JM, Popoff JF, Casano L, Bard FA, Gomez R, Valverde MA, and Malhotra V. TRPM5-mediated calcium uptake regulates mucin secretion from human colon goblet cells. Elife 2: e00658, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Cantero-Recasens G, Butnaru CM, Brouwers N, Mitrovic S, Valverde MA, and Malhotra V. Sodium channel TRPM4 and sodium/calcium exchangers (NCX) cooperate in the control of Ca(2+)-induced mucin secretion from goblet cells. J Biol Chem 294: 816–826, 2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Sakaguchi T, Okumura R, Ono C, Okuzaki D, Kawai T, Okochi Y, Tanimura N, Murakami M, Kayama H, Umemoto E, Kioka H, Ohtani T, Sakata Y, Miyake K, Okamura Y, Baba Y, and Takeda K. TRPM5 Negatively Regulates Calcium-Dependent Responses in Lipopolysaccharide-Stimulated B Lymphocytes. Cell Rep 31: 107755, 2020. [DOI] [PubMed] [Google Scholar]
- 220.Miller CN, Proekt I, von Moltke J, Wells KL, Rajpurkar AR, Wang H, Rattay K, Khan IS, Metzger TC, Pollack JL, Fries AC, Lwin WW, Wigton EJ, Parent AV, Kyewski B, Erle DJ, Hogquist KA, Steinmetz LM, Locksley RM, and Anderson MS. Thymic tuft cells promote an IL-4-enriched medulla and shape thymocyte development. Nature 559: 627–631, 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Lin W, Ogura T, Margolskee RF, Finger TE, and Restrepo D. TRPM5-expressing solitary chemosensory cells respond to odorous irritants. J Neurophysiol 99: 1451–1460, 2008. [DOI] [PubMed] [Google Scholar]
- 222.Barham HP, Cooper SE, Anderson CB, Tizzano M, Kingdom TT, Finger TE, Kinnamon SC, and Ramakrishnan VR. Solitary chemosensory cells and bitter taste receptor signaling in human sinonasal mucosa. Int Forum Allergy Rhinol 3: 450–457, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Maruyama Y, and Peterson OH. Single-channel currents in isolated patches of plasma membrane from basal surface of pancreatic acini. Nature 299: 159–161, 1982. [DOI] [PubMed] [Google Scholar]
- 224.Cheng H, Beck A, Launay P, Gross SA, Stokes AJ, Kinet JP, Fleig A, and Penner R. TRPM4 controls insulin secretion in pancreatic beta-cells. Cell Calcium 41: 51–61, 2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Diszhazi G, Magyar ZE, Lisztes E, Toth-Molnar E, Nanasi PP, Vennekens R, Toth BI, and Almassy J. TRPM4 links calcium signaling to membrane potential in pancreatic acinar cells. J Biol Chem 297: 101015, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Philippaert K, Pironet A, Mesuere M, Sones W, Vermeiren L, Kerselaers S, Pinto S, Segal A, Antoine N, Gysemans C, Laureys J, Lemaire K, Gilon P, Cuypers E, Tytgat J, Mathieu C, Schuit F, Rorsman P, Talavera K, Voets T, and Vennekens R. Steviol glycosides enhance pancreatic beta-cell function and taste sensation by potentiation of TRPM5 channel activity. Nat Commun 8: 14733, 2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Nelson PL, Zolochevska O, Figueiredo ML, Soliman A, Hsu WH, Feng JM, Zhang H, and Cheng H. Regulation of Ca(2+)-entry in pancreatic alpha-cell line by transient receptor potential melastatin 4 plays a vital role in glucagon release. Mol Cell Endocrinol 335: 126–134, 2011. [DOI] [PubMed] [Google Scholar]
- 228.Marigo V, Courville K, Hsu WH, Feng JM, and Cheng H. TRPM4 impacts on Ca2+ signals during agonist-induced insulin secretion in pancreatic beta-cells. Mol Cell Endocrinol 299: 194–203, 2009. [DOI] [PubMed] [Google Scholar]
- 229.Ma Z, Bjorklund A, and Islam MS. A TRPM4 Inhibitor 9-Phenanthrol Inhibits Glucose- and Glucagon-Like Peptide 1-Induced Insulin Secretion from Rat Islets of Langerhans. J Diabetes Res 2017: 5131785, 2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Shigeto M, Cha CY, Rorsman P, and Kaku K. A role of PLC/PKC-dependent pathway in GLP-1-stimulated insulin secretion. J Mol Med (Berl) 95: 361–368, 2017. [DOI] [PubMed] [Google Scholar]
- 231.Shigeto M, Ramracheya R, Tarasov AI, Cha CY, Chibalina MV, Hastoy B, Philippaert K, Reinbothe T, Rorsman N, Salehi A, Sones WR, Vergari E, Weston C, Gorelik J, Katsura M, Nikolaev VO, Vennekens R, Zaccolo M, Galione A, Johnson PR, Kaku K, Ladds G, and Rorsman P. GLP-1 stimulates insulin secretion by PKC-dependent TRPM4 and TRPM5 activation. J Clin Invest 125: 4714–4728, 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Colsoul B, Schraenen A, Lemaire K, Quintens R, Van Lommel L, Segal A, Owsianik G, Talavera K, Voets T, Margolskee RF, Kokrashvili Z, Gilon P, Nilius B, Schuit FC, and Vennekens R. Loss of high-frequency glucose-induced Ca2+ oscillations in pancreatic islets correlates with impaired glucose tolerance in Trpm5−/− mice. Proc Natl Acad Sci U S A 107: 5208–5213, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Colsoul B, Jacobs G, Philippaert K, Owsianik G, Segal A, Nilius B, Voets T, Schuit F, and Vennekens R. Insulin downregulates the expression of the Ca2+-activated nonselective cation channel TRPM5 in pancreatic islets from leptin-deficient mouse models. Pflugers Arch 466: 611–621, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Kyriazis GA, Soundarapandian MM, and Tyrberg B. Sweet taste receptor signaling in beta cells mediates fructose-induced potentiation of glucose-stimulated insulin secretion. Proc Natl Acad Sci U S A 109: E524–532, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Yu W, Hill WG, Apodaca G, and Zeidel ML. Expression and distribution of transient receptor potential (TRP) channels in bladder epithelium. Am J Physiol Renal Physiol 300: F49–59, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Smith AC, Parajuli SP, Hristov KL, Cheng Q, Soder RP, Afeli SA, Earley S, Xin W, Malysz J, and Petkov GV. TRPM4 channel: a new player in urinary bladder smooth muscle function in rats. Am J Physiol Renal Physiol 304: F918–929, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Hristov KL, Smith AC, Parajuli SP, Malysz J, Rovner ES, and Petkov GV. Novel regulatory mechanism in human urinary bladder: central role of transient receptor potential melastatin 4 channels in detrusor smooth muscle function. Am J Physiol Cell Physiol 310: C600–611, 2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Smith AC, Hristov KL, Cheng Q, Xin W, Parajuli SP, Earley S, Malysz J, and Petkov GV. Novel role for the transient potential receptor melastatin 4 channel in guinea pig detrusor smooth muscle physiology. Am J Physiol Cell Physiol 304: C467–477, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Maxwell SE, Leo MD, Malysz J, and Petkov GV. Age-dependent decrease in TRPM4 channel expression but not trafficking alters urinary bladder smooth muscle contractility. Physiol Rep 9: e14754, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Kullmann FA, Beckel JM, McDonnell B, Gauthier C, Lynn AM, Wolf-Johnston A, Kanai A, Zabbarova IV, Ikeda Y, de Groat WC, and Birder LA. Involvement of TRPM4 in detrusor overactivity following spinal cord transection in mice. Naunyn Schmiedebergs Arch Pharmacol 391: 1191–1202, 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Borgstrom A, Peinelt C, and Stoklosa P. TRPM4 in Cancer-A New Potential Drug Target. Biomolecules 11: 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 242.Picardo MCD, Sugimura YK, Dorst KE, Kallurkar PS, Akins VT, Ma X, Teruyama R, Guinamard R, Kam K, Saha MS, and Del Negro CA. Trpm4 ion channels in pre-Botzinger complex interneurons are essential for breathing motor pattern but not rhythm. PLoS Biol 17: e2006094, 2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Perniss A, Boonen B, Tonack S, Thiel M, Poharkar K, Alnouri MW, Keshavarz M, Papadakis T, Wiegand S, Pfeil U, Richter K, Althaus M, Oberwinkler J, Schutz B, Boehm U, Offermanns S, Leinders-Zufall T, Zufall F, and Kummer W. A succinate/SUCNR1-brush cell defense program in the tracheal epithelium. Sci Adv 9: eadg8842, 2023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Fouchet A, Bangando HM, Aize M, Simard C, and Guinamard R. TRPM4 contribution in mouse uterine contractions. Reproduction 166: 77–87, 2023. [DOI] [PubMed] [Google Scholar]
- 245.Tran TD, Zolochevska O, Figueiredo ML, Wang H, Yang LJ, Gimble JM, Yao S, and Cheng H. Histamine-induced Ca(2)(+) signalling is mediated by TRPM4 channels in human adipose-derived stem cells. Biochem J 463: 123–134, 2014. [DOI] [PubMed] [Google Scholar]
- 246.De Clercq K, Held K, Van Bree R, Meuleman C, Peeraer K, Tomassetti C, Voets T, D’Hooghe T, and Vriens J. Functional expression of transient receptor potential channels in human endometrial stromal cells during the luteal phase of the menstrual cycle. Hum Reprod 30: 1421–1436, 2015. [DOI] [PubMed] [Google Scholar]
- 247.De Clercq K, Van den Eynde C, Hennes A, Van Bree R, Voets T, and Vriens J. The functional expression of transient receptor potential channels in the mouse endometrium. Hum Reprod 32: 615–630, 2017. [DOI] [PubMed] [Google Scholar]
- 248.Persoons E, Hennes A, De Clercq K, Van Bree R, Vriens G, O DF, Peterse D, Vanhie A, Meuleman C, Voets T, Tomassetti C, and Vriens J. Functional Expression of TRP Ion Channels in Endometrial Stromal Cells of Endometriosis Patients. Int J Mol Sci 19: 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 249.Dusmez D, Cengiz B, Yumrutas O, Demir T, Oztuzcu S, Demiryurek S, Tutar E, Bayraktar R, Bulut A, Simsek H, Dagli SN, Kilic T, and Bagci C. Effect of verapamil and lidocaine on TRPM and NaV1.9 gene expressions in renal ischemia-reperfusion. Transplant Proc 46: 33–39, 2014. [DOI] [PubMed] [Google Scholar]
- 250.Wang HP, Pu XY, and Wang XH. Distribution profiles of transient receptor potential melastatin-related and vanilloid-related channels in prostatic tissue in rat. Asian J Androl 9: 634–640, 2007. [DOI] [PubMed] [Google Scholar]
- 251.Li S, Wang X, Ye H, Gao W, Pu X, and Yang Z. Distribution profiles of transient receptor potential melastatin- and vanilloid-related channels in rat spermatogenic cells and sperm. Mol Biol Rep 37: 1287–1293, 2010. [DOI] [PubMed] [Google Scholar]
- 252.Kruger J, Kunert-Keil C, Bisping F, and Brinkmeier H. Transient receptor potential cation channels in normal and dystrophic mdx muscle. Neuromuscul Disord 18: 501–513, 2008. [DOI] [PubMed] [Google Scholar]
- 253.Sakuraba M, Murata J, Teruyama R, Kamiya K, Yamaguchi J, Okano H, Uchiyama Y, and Ikeda K. Spatiotemporal expression of TRPM4 in the mouse cochlea. J Neurosci Res 92: 1409–1418, 2014. [DOI] [PubMed] [Google Scholar]
- 254.Takumida M, Ishibashi T, Hamamoto T, Hirakawa K, and Anniko M. Expression of transient receptor potential channel melastin (TRPM) 1–8 and TRPA1 (ankyrin) in mouse inner ear. Acta Otolaryngol 129: 1050–1060, 2009. [DOI] [PubMed] [Google Scholar]
- 255.Wong KK, and Hussain FA. TRPM4 is overexpressed in breast cancer associated with estrogen response and epithelial-mesenchymal transition gene sets. PLoS One 15: e0233884, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 256.Mardaryev A, van Lessen M, Alam M, Jimenez Acosta F, Biro T, and Paus R. Hair Follicle Chemosensation: TRPM5 Signaling Is Required for Anagen Maintenance. J Invest Dermatol 141: 2300–2303, 2021. [DOI] [PubMed] [Google Scholar]
- 257.Khatibi Shahidi M, Krivanek J, Kaukua N, Ernfors P, Hladik L, Kostal V, Masich S, Hampl A, Chubanov V, Gudermann T, Romanov RA, Harkany T, Adameyko I, and Fried K. Three-dimensional Imaging Reveals New Compartments and Structural Adaptations in Odontoblasts. J Dent Res 94: 945–954, 2015. [DOI] [PubMed] [Google Scholar]
- 258.Kappel S, Ross-Kaschitza D, Hauert B, Rother K, and Peinelt C. p53 alters intracellular Ca(2+) signaling through regulation of TRPM4. Cell Calcium 104: 102591, 2022. [DOI] [PubMed] [Google Scholar]
- 259.Borgstrom A, Hauert B, Kappel S, Zoni E, Kiener M, Stoklosa P, Baur R, Spahn M, Kruithof-de Julio M, and Peinelt C. Small Molecular Inhibitors Block TRPM4 Currents in Prostate Cancer Cells, with Limited Impact on Cancer Hallmark Functions. J Mol Biol 433: 166665, 2021. [DOI] [PubMed] [Google Scholar]
- 260.Sagredo AI, Sagredo EA, Cappelli C, Baez P, Andaur RE, Blanco C, Tapia JC, Echeverria C, Cerda O, Stutzin A, Simon F, Marcelain K, and Armisen R. TRPM4 regulates Akt/GSK3-beta activity and enhances beta-catenin signaling and cell proliferation in prostate cancer cells. Mol Oncol 12: 151–165, 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 261.Hong X, and Yu JJ. MicroRNA-150 suppresses epithelial-mesenchymal transition, invasion, and metastasis in prostate cancer through the TRPM4-mediated beta-catenin signaling pathway. Am J Physiol Cell Physiol 316: C463–C480, 2019. [DOI] [PubMed] [Google Scholar]
- 262.Armisen R, Marcelain K, Simon F, Tapia JC, Toro J, Quest AF, and Stutzin A. TRPM4 enhances cell proliferation through up-regulation of the beta-catenin signaling pathway. J Cell Physiol 226: 103–109, 2011. [DOI] [PubMed] [Google Scholar]
- 263.Rivas J, Diaz N, Silva I, Morales D, Lavanderos B, Alvarez A, Saldias MP, Pulgar E, Cruz P, Maureira D, Flores G, Colombo A, Blanco C, Contreras HR, Jana F, Gallegos I, Concha ML, Vergara-Jaque A, Poblete H, Gonzalez W, Varela D, Trimmer JS, Caceres M, and Cerda O. KCTD5, a novel TRPM4-regulatory protein required for cell migration as a new predictor for breast cancer prognosis. FASEB J 34: 7847–7865, 2020. [DOI] [PubMed] [Google Scholar]
- 264.Verigos J, Kordias D, Papadaki S, and Magklara A. Transcriptional Profiling of Tumorspheres Reveals TRPM4 as a Novel Stemness Regulator in Breast Cancer. Biomedicines 9: 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 265.Kappel S, Stoklosa P, Hauert B, Ross-Kaschitza D, Borgstrom A, Baur R, Galvan JA, Zlobec I, and Peinelt C. TRPM4 is highly expressed in human colorectal tumor buds and contributes to proliferation, cell cycle, and invasion of colorectal cancer cells. Mol Oncol 13: 2393–2405, 2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 266.Hu Y, Kaschitza DR, Essers M, Arullampalam P, Fujita T, Abriel H, and Inoue R. Pathological activation of CaMKII induces arrhythmogenicity through TRPM4 overactivation. Pflugers Arch 473: 507–519, 2021. [DOI] [PubMed] [Google Scholar]
- 267.Lee DK, Park JY, Yoo JC, Byun EH, Bae YJ, Lee YS, Park N, Kang D, Han J, Park JY, Hwang E, and Hong SG. PTPN6 regulates the cell-surface expression of TRPM4 channels in HEK293 cells. Pflugers Arch 470: 1449–1458, 2018. [DOI] [PubMed] [Google Scholar]
- 268.Xian W, Hui X, Tian Q, Wang H, Moretti A, Laugwitz KL, Flockerzi V, Ruppenthal S, and Lipp P. Aberrant Deactivation-Induced Gain of Function in TRPM4 Mutant Is Associated with Human Cardiac Conduction Block. Cell Rep 24: 724–731, 2018. [DOI] [PubMed] [Google Scholar]
- 269.Wang F, Wu P, Gong S, Chen Y, Gao J, Wang S, Shen Q, Tao H, Hua F, Zhou Z, Zou Z, Ma T, and Jia Y. Aberrant TRPM4 expression in MLL-rearranged acute myeloid leukemia and its blockade induces cell cycle arrest via AKT/GLI1/Cyclin D1 pathway. Cell Signal 72: 109643, 2020. [DOI] [PubMed] [Google Scholar]
- 270.Mathar I, Kecskes M, Van der Mieren G, Jacobs G, Camacho Londono JE, Uhl S, Flockerzi V, Voets T, Freichel M, Nilius B, Herijgers P, and Vennekens R. Increased beta-adrenergic inotropy in ventricular myocardium from Trpm4−/− mice. Circ Res 114: 283–294, 2014. [DOI] [PubMed] [Google Scholar]
- 271.Shimizu T, Owsianik G, Freichel M, Flockerzi V, Nilius B, and Vennekens R. TRPM4 regulates migration of mast cells in mice. Cell Calcium 45: 226–232, 2009. [DOI] [PubMed] [Google Scholar]
- 272.Kuras Z, Yun YH, Chimote AA, Neumeier L, and Conforti L. KCa3.1 and TRPM7 channels at the uropod regulate migration of activated human T cells. PLoS One 7: e43859, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 273.Sengupta S, West KO, Sanghvi S, Laliotis G, Agosto LM, Lynch KW, Tsichlis PN, Singh H, Patrick KL, and Guerau-de-Arellano M. PRMT5 Promotes Symmetric Dimethylation of RNA Processing Proteins and Modulates Activated T Cell Alternative Splicing and Ca(2+)/NFAT Signaling. Immunohorizons 5: 884–897, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 274.Serafini N, Dahdah A, Barbet G, Demion M, Attout T, Gautier G, Arcos-Fajardo M, Souchet H, Jouvin MH, Vrtovsnik F, Kinet JP, Benhamou M, Monteiro RC, and Launay P. The TRPM4 channel controls monocyte and macrophage, but not neutrophil, function for survival in sepsis. J Immunol 189: 3689–3699, 2012. [DOI] [PubMed] [Google Scholar]
- 275.Devantier HR, Long DJ, Brennan FX, Carlucci SA, Hendrix C, Bryant RW, Salemme FR, and Palmer RK. Quantitative assessment of TRPM5-dependent oral aversiveness of pharmaceuticals using a mouse brief-access taste aversion assay. Behav Pharmacol 19: 673–682, 2008. [DOI] [PubMed] [Google Scholar]
- 276.Riera CE, Vogel H, Simon SA, Damak S, and le Coutre J. Sensory attributes of complex tasting divalent salts are mediated by TRPM5 and TRPV1 channels. J Neurosci 29: 2654–2662, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 277.Brixel LR, Monteilh-Zoller MK, Ingenbrandt CS, Fleig A, Penner R, Enklaar T, Zabel BU, and Prawitt D. TRPM5 regulates glucose-stimulated insulin secretion. Pflugers Arch 460: 69–76, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]


