Abstract
Dendritic cells (DCs) play a central role in coordinating immune responses by linking innate and adaptive immunity through their exceptional antigen-presenting capabilities. Recent studies reveal that metabolic reprogramming—especially pathways involving acetyl-coenzyme A (acetyl-CoA)—critically influences DC function in both physiological and pathological contexts. This review consolidates current knowledge on how environmental factors, tumor-derived signals, and intrinsic metabolic pathways collectively regulate DC development, subset differentiation, and functional adaptability. Acetyl-CoA emerges as a dual-function metabolite, serving not only as an energy carrier but also as an epigenetic regulator that controls DC fate via lipid biosynthesis, mitochondrial metabolism, and chromatin modification. In the tumor microenvironment (TME), DCs may experience immune suppression polarization and insufficient T cell activation due to disrupted acetyl-CoA related metabolic pathways. While existing DC-based therapies remain constrained by TME-induced metabolic limitations, emerging approaches that restore acetyl-CoA related metabolic pathways balance show enhanced antitumor efficacy. The review further examines distinct metabolic adaptations among DC subsets and their relevance to autoimmune diseases, infectious immunity, and transplant outcomes. By integrating current research on targeting DC metabolic targets, we outline strategies for developing immunotherapies that target DC metabolic flexibility. Remaining hurdles include tailoring interventions to specific subsets, refining metabolic manipulation techniques, and addressing TME heterogeneity through combination therapies. These findings position acetyl-CoA as a key therapeutic target for recalibrating immunometabolism circuits, with significant implications for DC-focused cancer treatment.
Keywords: Dendritic cells, Acetyl-CoA, Metabolic reprogramming, Tumor microenvironment, Immunotherapy, Cancer therapy
Introduction
Dendritic cells (DCs), first identified by Ralph Steinman in 1973 as the "professional sentinels" of the immune system, serve as the critical bridge between innate and adaptive immunity by orchestrating immune surveillance, antigen presentation, and T cell priming. Over decades, their developmental ontogeny, subset specialization (e.g., cDC1, cDC2, pDC), and functional plasticity have been extensively characterized, revealing their indispensable roles in pathogen clearance, autoimmune regulation, and antitumor immunity [1, 2]. However, the tumor microenvironment (TME) imposes profound metabolic and functional constraints on DCs, leading to impaired antigen presentation, disrupted migration, and tolerogenic polarization—key barriers to effective cancer immunotherapy [3]. Emerging evidence highlights metabolic reprogramming as a central axis governing DCs functionality, with acetyl-coenzyme A (acetyl-CoA) emerging as a pivotal metabolite that integrates lipid synthesis, epigenetic regulation, and mitochondrial energetics to dictate DCs fate in tumors.
Recent advances in single-cell multi-omics and spatial metabolomics have unveiled unprecedented heterogeneity in DCs subsets and their metabolic adaptations. For instance, tumor-infiltrating cDC1s exhibit glycolytic defects under melanoma-associated hypoxia, while cDC2s adopt lipid-laden phenotypes due to SREBP-driven lipogenesis, impairing their cross-presentation capacity [4–6]. Recent studies indicate acetyl-CoA may influences immune cells maturation, migration, and cytokine production by regulating lipid metabolism, oxidative phosphorylation (OXPHOS), and epigenetic modifications [7–10]. This review integrates DCs biology from a metabolic perspective, aiming to offer a mechanistic framework for understanding how metabolic interventions, especially targeting acetyl-CoA, could restore DCs function in cancer therapy. We summarize key research advances based on the following issues: First, while DCs subsets are well—classified, systematic integration of their metabolic remodeling in tumors is lacking. Second, acetyl-CoA's dual roles as an energy source and a chromatin modifier position it as a nexus of DCs functional regulation, yet mechanistic insights remain fragmented. Third, although preclinical metabolic targeting has succeeded, clinical translation lags due to incomplete understanding of TME—specific metabolic crosstalk.
This review begins by delineating the ontogeny and heterogeneity of DCs subtypes to establish the basis for their functional diversity. The next section explores the metabolic regulation of DCs, with a focus on glycolysis, mammalian Target of Rapamycin (mTOR)/AMP-activated protein kinase (AMPK) signaling and lipid metabolism. This is followed by an in-depth discussion of acetyl-CoA's biological roles in DCs. We then proceed to examine DCs dysfunction in various non-tumor diseases, focusing on DCs functional impairment in tumors and the underlying mechanisms. The review emphasizes how acetyl-CoA influences anti-tumor immunity through the regulation of DCs metabolism and function, culminating in therapeutic strategies that exploit DCs metabolic plasticity. This framework links molecular mechanisms to clinical innovation, with a particular emphasis on acetyl-CoA as a key component for developing next-generation DCs therapies.
Ontogeny and subsets of DCs
DCs are crucial for initiating adaptive immune responses and maintaining immune homeostasis. They originate from pluripotent progenitor cells and differentiate into distinct subpopulations, each with unique functional roles and molecular markers. DCs development is strictly regulated by multiple cytokines. Under the influence of different cytokines, DCs precursors are induced to differentiate into distinct subpopulations. This may be due to the activation of different signaling pathways and metabolic changes. Environmental stimuli, such as those in the tumor microenvironment, can also regulate DCs differentiation, leading to functional heterogeneity [11]. Therefore, understanding the individual development and subpopulation characteristics of DCs is crucial for developing effective immunotherapies [12].
Developmental pathways
DCs are typically categorized into four functionally distinct subsets: cDC1, cDC2, pDC, and moDC. While their direct precursor cells differ, all DCs subsets originate from multipotent progenitors [13]. The cytokine FMS-like tyrosine kinase 3 (FLT3) plays a pivotal role in determining DCs fate during development. Studies in humans and mice confirm that FLT3 drives hematopoietic progenitor cell differentiation into DCs and sustains DCs homeostasis both in vitro and in vivo [14, 15]. Granulocyte–macrophage colony-stimulating factor (GM-CSF), another critical regulator, strongly promotes moDCs induction in vitro [16, 17]. Unlike GM-CSF, FLT3-mediated DCs differentiation does not favor specific subsets but simultaneously generates multiple cDCs and pDCs populations. Combined stimulation with GM-CSF and FLT3L in vitro preferentially directs cell differentiation toward cDCs subsets [18] (Table 1 [19–25]).
Table 1.
Characteristics and functions of different DCs subsets
| DCs subsets | Location | Makers (Human) | Function | Production | Chemokines | |
|---|---|---|---|---|---|---|
| cDC1 | Lymph, peripheral tissues, blood | CD11c, MHC-II, BDCA3, XCR1, CADM1, Clec9A, BTLA, TLR8, TLR10, IDO2, IDO1, FLT3, CLNK, CD26, CD205 |
Cross-expression of exogenous antigens in MHCI Immunity of Th1 and CD8+ T cells to intracellular pathogens and tumors |
INF-λ, IL-27, IL-12, INF-γ, CXCL9, IL-6, IL-10, TNF-α, IL-1β, CXCL10 | CCL5, XCL1, CCL7 | |
| cDC2 | DC2 | Lymph, peripheral tissues, blood | CD11c, HLA-DR, CD1c, SIRPA, CD11b, TLR1, TLR2, TLR3, TLR7, CLEC10A, FceR1A |
Direct expression of exogenous antigens on MHCII Activation of immunogenic CD4+ Th and regulatory T cells to clear extracellular pathogens |
IL-12, INF-γ, IL-18, TNF-α, IL-6 | CCL21, CCR7 |
| DC3 | Lymph, peripheral tissues, blood | CD36, CD163 | Significant activity in polarizing CD8+ T cells to CD8+CD103+ tissue-resident T cells | S100A8, S100A9, INF-γ, IL-12 | CCL21, CCR7 | |
| CD16+DC | Blood | CD16, CD85d, CD85h, CD115, CD31, SLC7A7 | Uptake processing, antigen presentation, immune tolerance | Siglic-10, TNF, IL-10 | No reported | |
| pDC | Lymphoid Residue, blood, tonsils | BDCA2, BDCA4, CLEC4C, BTLA, ILT7, CCR9, SCA1, PTPRS | Type I interferon secretion, antiviral response | IFN-I, IL-3, IL-6, TNF-α | MIP-1α, CXCL9, MIP-β | |
| LC | Epidermis, lymph nodes | Langerin, CD1a, E-Cadherin, EPCAM, CD205, HLA-DR, | Involved in non-specific immune, acquire exogenous antigens | TNF, IL-12, CCR6 | CCL20 | |
| moDC | Derive from monocytes | CD11c, HLA-DR, CD14, MMR, CD11b, CD64, CD1c, CD1a | Environment-dependent activation of CD8+ T cells, Th1, Th2 and Th17-type immunity | TNF-α, IL-23, NO, MCP-1, CXCL10 | CCL2, CCL3 | |
Specific subsets can be distinguished by their unique transcription factor profiles. cDCs subsets originate from pre-cDCs differentiation downstream of common DCs progenitors, a process governed by transcription factors such as interferon regulatory factor (IRF) 8, BATF3, and ID2 [26]. In the cDC2s subset, IRF4 and ZEB2 emerge as critical regulators of differentiation [27]. pDCs develop from pre-pDCs through mechanisms involving E proteins, RUNX1, and IRF8, while also expressing genes typically found in B cells [13, 28]. moDCs represent an inflammatory DCs subset sharing markers with cDC2s and deriving from monocytes downstream of MDP [29]. Single-cell sequencing has revealed DCs markers and developmental pathways across different contexts, including the discovery of mregDCs—a TME-enriched subset expressing immune regulatory molecules—and the delineation of DC2s and DC3s subsets that clarify DCs functional diversity [30, 31].
Environmental stimuli drive DCs differentiation into distinct phenotypes, generating functional heterogeneity among subsets. Tumor-derived prostaglandin E2 (PGE2) suppresses DCs progenitor development, while (transforming growth factor) TGF-β and IL-10 promote tolerogenic cDCs phenotypes [32–34]. By impairing DCs function, tumors disrupt immune response initiation and facilitate immune escape. These findings underscore how environmental modulation during DCs development can alter differentiation trajectories and functional outcomes—a key consideration for improving tumor-associated DCs (TADCs) efficacy.
Subset characteristics
Distinct subsets of DCs express specific surface molecules that enable their identification and reflect their specialized functions. Human cDC1s are typically identified by the markers CD11c (low), HLA-DR+, Clec9A+, XCR1+, and CD141+ [35, 36]. Despite their relatively low abundance among DCs subsets, cDC1s serve as critical antigen-presenting cells in antitumor immunity by capturing damage-associated molecular patterns (DAMPs) from primary tumors. These cells process tumor antigens through HLA-I molecules, migrate to lymph nodes, and present antigens to CD8+ T cells to initiate tumor-specific cellular immunity [11, 36]. cDC1s also secrete tumor necrosis factor (TNF)-α, IL-6, IL-12, and type I interferons (IFN-I) to amplify immune cell activation [37–39], while their production of CXCL9 and CXCL10 recruits T cells to tumor sites [40]. The STING signaling pathway is functionally linked to cDC1s activity, with the DCs-STING-IFN axis contributing significantly to various therapeutic outcomes [41–43]. Unlike other DCs subgroups, cDC1s uniquely express Toll-like receptor (TLR)1/3/6/8/10, conferring distinct molecular pattern recognition capabilities for detecting viral and intracellular pathogens [35, 44, 45]. cDC2s primarily activate CD4+ T cells via HLA-II, subsequently enhancing CD8+ T cell responses and cytotoxic consolidation [46, 47], with human cDC2s identified by CD11c+, CD11b+, HLA-DR+, and CD1c+ markers [26, 35]. Upon activation, these cells release IL-12 and IL-1β to stimulate Th17, Th1, and Th2 responses, modulating diverse immune functions [48, 49]. Evidence suggests cDC2s contribute to tumor vaccine efficacy, ICI responses, and immune memory maintenance [47, 50, 51], with TLR2 expression governing their pathogen recognition in anti-infection immunity [35].
Human pDCs are characterized by HLA-DR+, CD123+, CD303+, CD304+, and CCR2+ markers and specialize in antiviral immunity through IFN-I production [26, 36]. Surface TLR7 and TLR9 activation triggers mTOR and NF-κB pathways upon nucleic acid recognition, inducing IFN-I secretion [25, 52, 53]. While IFN-I can promote antitumor immunity, pDCs correlate with poor prognosis in melanoma and breast cancer [54–56]. Tumor-associated pDCs stimulated by TGF-β express indoleamine 2,3-dioxygenase (IDO) to induce tolerance and employ ICOSL-mediated Treg differentiation to suppress immunity [57, 58]. However, TLR-activated pDCs can regain antigen-presenting capacity and stimulate antitumor responses [59].
moDCs and cDC2s share common surface markers such as CD11c, CD11b, and CD1c, with moDCs differentiating from monocytes under inflammatory conditions [29]. moDCs promote inflammation by inducing IL-17 secretion from CD4+ T cells, which drives Th17 cell differentiation [60]. These cells also initiate Th2-mediated immune responses that contribute to allergen-induced inflammation [61]. Within tumors, moDCs infiltration enhances cytotoxic lymphocyte (CTL) activity through nitric oxide synthase (iNOS)-mediated nitric oxide production, facilitating tumor cell killing [62, 63]. moDCs further demonstrate the capacity to activate T cells and improve ICI responses [64–66]. In the TME, moDCs enable antigen cross-presentation and subsequent T-cell activation, a mechanism exploited by DCs-based vaccines for cancer therapy [67, 68]. Immune checkpoint blockade in melanoma models revealed that tumor-infiltrating lymphocyte efficacy strongly correlated with moDCs populations expressing elevated CD80, CD86, and MHC-II. These moDCs supported tumor-infiltrating lymphocyte expansion via iNOS expression, suggesting monocyte-to-moDCs differentiation could potentiate antitumor immunity [68–70] (Fig. 1).
Fig. 1.
Differentiation and development of DCs. MMP differentiates into CMP and CLP in the bone marrow. CMP differentiates into GMP and MDP, with GMP differentiating into monocytes and becoming moDC in inflammation. MDP differentiates into CDP, which differentiates into pre-pDC and pre-cDC. Pre-cDC is the precursor of both cDC1 and cDC2. It differentiates towards cDC1 under the control of IRF8 and towards cDC2 under the control of IRF4. The differentiation of pre-pDC into pDC depends on transcription factors like E2-2. DC subsets express markers and perform functions. CDP, common DC progenitors; CLP, common lymphoid precursor; CMP, common-myeloid progenitors; MDP, Monocyte-dendritic cell progenitors; MPP, multipotent blood progenitors
Metabolic regulation of DCs
The metabolic regulation of DCs has been extensively studied, revealing inherent variations among DCs subsets. cDC1s and moDCs demonstrate stronger mitochondrial function and OXPHOS capacity compared to cDC2s and pDCs, potentially explaining their divergent responses to identical stimuli in both physiological and experimental settings [71–73]. Metabolic profiles further differ across DCs developmental stages: early activation involves concurrent glycolysis and fatty acid oxidation (FAO)-dependent OXPHOS elevation, while prolonged activation shifts metabolism toward exclusive glycolytic dependence as OXPHOS capacity diminishes [74–76]. This metabolic transition coincides with phenotypic maturation, characterized by enhanced cytokine secretion (including IL-6 and IL-12) and upregulated CD80/CD86 expression [77]. Current evidence suggests iNOS-derived NO suppresses OXPHOS by inhibiting the electron transport chain [78, 79], while GM-CSF and FLT3L stimulation activates the mTOR-hypoxia inducible factor (HIF)−1α axis to promote glycolytic enzyme activity [80–82]. Understanding how DCs metabolism shapes functional outcomes across biological contexts remains essential for identifying novel therapeutic targets.
Metabolic pathways
Glycolysis
Glycolysis plays a critical role in DCs maturation by supplying both ATP and essential metabolic intermediates that maintain DCs functionality. The glycolysis inhibitor 2-deoxyglucose reduces CD80/CD86 co-stimulatory molecule expression, suppresses TNF-α and IL-12 secretion, and impairs DCs migratory capacity in both ex vivo and in vivo models through downregulation of CCL7 [77, 83], ultimately compromising T cell activation. Glycolytic metabolites further contribute to DCs maturation, with mitochondrial pyruvate import representing a particularly crucial step in this process [75, 77]. Citrate derived from pyruvate via the tricarboxylic acid (TCA) cycle generates cytoplasmic acetyl-CoA for lipid biosynthesis [75], which supports DCs physiological functions by maintaining ER and Golgi apparatus activity for proper cytokine secretion and antigen processing [75, 77, 84]. Under LPS stimulation or IL-4 activation, DCs primarily utilize glycogen reserves to fuel glycolysis before upregulating glucose transporter protein1 for external glucose uptake [85, 86]. This imported glucose undergoes glycogen synthesis followed by glycolytic breakdown, completing the metabolic reprogramming characteristic of DCs activation [83, 85]. Early LPS-induced glycolytic activation depends on TBK1/IKKε/AKT-mediated hexokinase II stimulation [77], while sustained enzyme expression (including phosphofructokinase and glucose transporter protein1) is regulated by PI3K/AKT/mTOR pathway activation and AMPK pathway inhibition [74, 83].
mTOR and AMPK
The influence of mTOR on DCs depends on their developmental stage. mTOR pathway activation during DCs differentiation and maturation supports their proper development. Rapamycin-mediated mTOR inhibition in vitro decreases the generation of cDC1s, cDC2s, and pDCs subsets [80, 81], yet paradoxically enhances the functional capacity and T-cell stimulatory potential of mature bone marrow-derived DCs in mice. Genetic ablation of TSC1 in DCs disrupts MHC-I and IL-7 expression through mTOR hyperactivation, compromising T-cell homeostasis [87–89]. These divergent outcomes likely reflect distinct metabolic requirements at different DCs life stages. The AMPK pathway counteracts mTOR signaling [90], with AMPK activation in tumor-associated DCs typically inducing tolerogenic properties characterized by improved mitochondrial function, increased FAO and OXPHOS, and diminished CD80/CD86 expression [3, 91, 92]. Exogenous AMP additionally stimulates DCs A2b receptors, triggering TGF-β and IL-10 production – immunosuppressive cytokines that may exert their inhibitory effects through AMPK pathway engagement [3, 93–97]. Conversely, extracellular ATP drives DCs inflammatory polarization by promoting IL-1β secretion for T-cell priming while limiting adenosine accumulation and ameliorating hypoxic stress [98–100].
Lipid metabolism
LPS-activated lipid accumulation in DCs correlates with enhanced antigen cross-presentation, a pivotal mechanism for initiating tumor-specific immune responses [101]. While lipid accumulation in DCs generally supports T cell activation, excessive lipid buildup in TADCs disrupts their normal functions [102]. The origin of these lipids remains unclear, though potential sources include external lipid uptake via macrophage scavenger receptors (Msr) or endogenous synthesis driven by tumor-induced metabolic reprogramming. Melanoma-derived Wnt5a, for instance, activates β-catenin signaling in TADCs, where subsequent PPAR-γ activation enhances OXPHOS coupled with FAO to drive lipid accumulation. Etomoxir-mediated FAO inhibition in DCs has been shown to improve immunotherapy outcomes [8, 103, 104]. These accumulated lipids undergo oxidation upon reactive oxygen species (ROS) exposure, forming oxidized derivatives that impair antigen presentation in murine cDC1s by binding heat shock protein 70 and blocking MHC-peptide complex formation [105]. ROS additionally activates the IRE1α-XBP1 pathway, inducing endoplasmic reticulum (ER) stress that further compromises TADCs-mediated T cell activation while stimulating additional lipid synthesis [104, 106], creating a self-reinforcing cycle of lipid accumulation and oxidative damage.
Amino acid metabolism
Tryptophan and arginine metabolism critically regulates DCs functional homeostasis. IDO mediates tryptophan conversion to kynurenine [107], and its expression by TADCs activates Tregs while suppressing immune responses. Kynurenine sustains IDO expression through activation of the DC's aromatic hydrocarbon receptor (AhR) [108, 109]. Treg-DCs interactions via CTLA-4 binding to B7 receptors further enhance IDO expression [110, 111], establishing two positive feedback loops: IDO-kynurenine-AhR-IDO and IDO-Treg-CTLA-4-B7-IDO, both reinforcing immunosuppressive TADCs function. iNOS metabolizes arginine to generate citrulline and NO [112], which modulates cellular behavior within the TME [113, 114]. Polyamines derived from arginase 1-mediated catabolism regulate IDO phosphorylation and signaling in DCs, with TGF-β-stimulated IDO production requiring Arg1 expression [115].
Vitamin metabolism
Vitamins regulate essential enzymatic processes and maintain metabolic homeostasis [116], influencing intracellular metabolism and DCs phenotypes through signaling pathway modulation. Vitamin D alters DCs glucose metabolism by increasing PFKFB4 activity, which enhances glucose oxidation [117]. These metabolic shifts promote a tolerogenic DCs phenotype characterized by Treg differentiation while suppressing DCs maturation and inflammatory responses [118]. Vitamin A governs DCs function via retinoic acid metabolism, a pathway critical for maintaining intestinal DCs tolerance and immune homeostasis; dysregulation of this process can intensify intestinal inflammation [119, 120]. As a potent antioxidant, vitamin E protects membrane lipids and modulates ROS/RNS production. Emerging evidence indicates that vitamin E enhances antigen presentation in TADCs through SHP1 targeting [121], potentially amplifying therapeutic responses to chemotherapy and radiotherapy. In obese mouse models, this effect appears mediated through FAO regulation during oxidative stress [122]. Tocopherol-based nanoemulsions facilitate DCs vaccine delivery into the TME by mitigating lipid accumulation and oxidative damage while enhancing T cell recruitment from tumors and lymph nodes [123], demonstrating vitamin E's dual capacity for metabolic and immune modulation.
In summary, metabolic reprogramming drives the functional plasticity of DCs. Glycolysis fuels DCs maturation and migration by generating ATP and metabolic intermediates like pyruvate, with TBK1/IKKε/AKT and PI3K/mTOR signaling cascades regulating key enzymatic steps. mTOR and AMPK pathways display reciprocal regulation during DCs development: mTOR activation stimulates DCs differentiation and pro-inflammatory responses, while AMPK promotes tolerogenic DCs through enhanced FAO and OXPHOS. Lipid metabolism exerts dual effects on DCs function—moderate lipid accumulation improves antigen cross-presentation, whereas TME-induced lipid overload, mediated by β-catenin/PPAR-γ activation, impairs DCs activity via oxidative damage and ER stress. The tryptophan-IDO-aryl hydrocarbon receptor axis and arginine-iNOS/Arg1-polyamine pathway mediate DCs immunosuppression, with IDO-driven regulatory T cell expansion reinforcing immune evasion in tumors. Vitamins influence DCs metabolism and immune responses by modulating enzymatic activity, exemplified by vitamin D's upregulation of PFKFB4, and redox balance through mechanisms like vitamin E's targeting of SHP1. Therapeutic interventions targeting these pathways may counteract DCs dysfunction and inform new approaches to cancer immunotherapy.
Biological functions of Acetyl-CoA
Acetyl-CoA serves as a central metabolite that fuels the TCA cycle and OXPHOS, enabling complete oxidation of sugars, lipids, and proteins for ATP production [124]. This molecule also acts as a biosynthetic precursor for cholesterol, ketone bodies, and various physiologically active compounds [125]. Within the nucleus, compartmentalized acetyl-CoA levels modulate histone acetylation through lysine acetyltransferase (KAT) activity, a process further influenced by lipid synthesis pathways [126–128]. Recent studies demonstrate that acetyl-CoA exerts broad functional impacts on TME cells through metabolic regulation [7, 129].
Source and transport of Acetyl-CoA
Cells synthesize acetyl-CoA through multiple metabolic pathways, such as pyruvate decarboxylation by the pyruvate dehydrogenase complex (PDC), fatty acid β-oxidation, acetate-CoA ligation via acyl-CoA short-chain synthetases (ACSS), branched-chain amino acid degradation, and citrate cleavage by ATP citrate lyase (ACLY) [7, 130–133]. The PDC-mediated conversion of pyruvate remains the primary acetyl-CoA source in most cells, feeding the TCA cycle to sustain OXPHOS and cellular energy production. Ketone body-derived acetyl-CoA not only fuels extrahepatic tissues but also acts as a signaling molecule with potential implications in cancer therapy [7]. Branched-chain amino acid catabolism substantially contributes to acetyl-CoA pools through sequential actions of branched-chain amino acid aminotransferase and branched-chain keto acid dehydrogenase kinase, with these pathways implicated in pancreatic disease pathogenesis [134–136].
Cytosolic acetyl-CoA pools interconnect through derivative intermediates. The citrate-pyruvate cycle mediates acetyl-CoA exchange between mitochondria and cytosol via SLC25A1-mediated citrate transport [137, 138]. Oxaloacetate derived from this cycle undergoes malate conversion before mitochondrial reimport through SLC25A10 and SLC25A11 [139, 140]. Malate-derived pyruvate re-enters mitochondria via the mitochondrial pyruvate carrier [141]. These redundant transport systems maintain compartment-specific acetyl-CoA levels for diverse functions, including nuclear gene regulation under metabolic stress, ensuring cellular homeostasis [7, 142–144].
Metabolic pathways and biological roles of Acetyl-CoA
Acetyl-CoA metabolism varies across cell types and plays a critical role in cellular maturation and development. Macrophages and DCs exhibit impaired TCA cycle activity, releasing acetyl-CoA after citrate synthesis for lipid biosynthesis and gene expression regulation—a prerequisite for early cellular activation [76]. In contrast, most cells primarily rely on the intact TCA cycle for energy production. The high-energy thioester bond in acetyl-CoA facilitates acetyl group transfer [145], establishing its role as a key acetyl donor for protein acetylation mediated by enzymes like KAT. Both histone acetylation, which modulates gene expression, and non-histone protein acetylation influence fundamental processes including cell division and autophagy [146–148].
As the initiating substrate for fatty acid synthase (FAS) [149], acetyl-CoA serves essential functions in lipid biosynthesis. Carnitine bridges acetyl-CoA and fatty acid metabolism, with SLC25A20 transporting cytosolic fatty acids as acylcarnitine alongside converted carnitine derivatives, maintaining metabolic equilibrium [128, 150, 151]. These lipids constitute cellular membranes and support organelle expansion in the ER and Golgi apparatus [85, 87], enabling normal physiological functions. Statins, which inhibit cholesterol synthesis, have been shown to impair DCs invasiveness in immune disorders [152–154]. Lipids also function as crucial intracellular signaling molecules [155], with multiple pathways requiring stable lipid levels for proper signal transduction.
Beyond being a glycolytic byproduct, acetyl-CoA orchestrates cellular functionality. Glycolysis-driven epigenetic regulation through acetyl-CoA production allows immune cells to maintain histone acetylation patterns [156–158]. Tumor cells with PTEN-inducible kinase 1 overexpression exhibit suppressed glycolysis and reduced acetyl-CoA levels, leading to apoptotic cell death. Acetyl-CoA further integrates glycolysis with fatty acid metabolism while regulating glycolytic flux [159]. During inflammation, FAO-derived acetyl-CoA enhances glycolysis [160], with acetyl-CoA-mediated acetylation states modulating signaling pathways that drive metabolic reprogramming [161].
Branched-chain amino acid catabolism contributes substantially to acetyl-CoA pools, while TCA cycle intermediates derived from acetyl-CoA serve as precursors for glutamate, arginine, and other amino acids [162]. Fluctuations in acetyl-CoA and amino acid levels reflect cellular nutrient status, enabling metabolic regulation through diverse signaling pathways [162, 163].
Acetyl-CoA both depends on and regulates enzymatic activity. Declining acetyl-CoA concentrations reduce the activity of acetyl-CoA carboxylase (ACC) isoforms ACACA and ACACB, along with HMG-CoA reductase [164, 165]. The acetyl-CoA/CoA ratio additionally modulates KAT enzymes, including KAT2A (GCN5), KAT2B (PCAF), and p300/CBP—key regulators of both metabolism and immune function [166, 167]. These findings establish acetyl-CoA as a central integrator of carbohydrate, lipid, and amino acid metabolism, coordinating metabolic states through acetylation processes and mTOR pathway modulation to maintain cellular functional homeostasis (Fig. 2).
Fig. 2.
Acetyl-CoA related metabolism. Glucose produces pyruvate, which is converted into acetyl-CoA and enters the TCA cycle, providing the conditions for OXPHOS to occur. In mitochondria, acetic acid and BCAA serve as additional sources of acetyl-CoA. The intermediate metabolites generated by acetyl-CoA in the TCA cycle exert a significant influence on cellular metabolism and function. α-KG can be interconverted with Glu, and citrate can be used as an acetyl-CoA carrier to enter the cytoplasm and participate in lipid metabolism. Citric acid is broken down into acetyl-CoA, which is used to produce fatty acids and cholesterol. Fatty acids can be converted to acetyl -CoA through FAO. Additionally, acetyl-CoA have various source within the nucleus. Acetyl-CoA provides acetyl groups for histone acetylation and regulates gene expression. Furthermore, the expression and activity of acetyl-CoA related enzymes are regulated by transcription factors and signaling pathways. ACTP, acetate transporter protein; α-KG, α-ketoglutaric acid; BACA, branched chain amino acid; BCAT, branched-chain amino acid aminotransferase; CPT, Carnitine acyl transferase; HMGCL, HMG-CoA lyase; HMGCS, HMG-CoA synthase; MVA, Mevalonate; OAA, oxaloacetic acid
Role of Acetyl-CoA in DCs development and differentiation
The regulatory role of metabolic pathways in DCs biology has been well documented, with acetyl-CoA emerging as a key metabolic regulator due to its broad influence on cellular metabolism. During in vitro DCs differentiation induced by GM-CSF and FLT3L, metabolic reprogramming occurs through modulation of the mTOR and AMPK signaling pathways, which respectively promote anabolic and catabolic states [91, 168, 169]. Acetyl-CoA deprivation triggers AMPK activation while suppressing mTOR signaling [170], suggesting this direct pathway mediates acetyl-CoA's effects on DCs function. Genetic studies demonstrate that TSC1 deficiency activates mTOR/PPAR-γ signaling, enhancing neuropilin-1 expression and naïve T-cell proliferation while impairing antigen presentation and specific T-cell activation. TSC1 additionally regulates MHC-II expression through IRF4 and CIITA [171–173]. The TSC1/mTOR axis not only controls acetyl-CoA levels by modulating metabolic enzyme activity but also determines its functional utilization, positioning acetyl-CoA as a critical downstream effector in this pathway [170, 174].
In co-cultures of GM-CSF-derived macrophages and DCs, macrophage-derived NO inhibits OXPHOS while promoting DCs glycolysis. While moderate NO production sustains glycolytic metabolism during DCs activation, excessive NO impairs acetyl-CoA-dependent histone acetylation [175–177], indicating NO may mediate acetyl-CoA's regulatory effects. Activated DCs accumulate lactate, which is exported via monocarboxylate transporters [3, 75], creating an acidic microenvironment that attenuates DCs-mediated T-cell activation [178]. Intracellular acidification similarly enhances histone deacetylase activity [179], potentially reducing acetyl-CoA-dependent acetylation of immune genes and compromising DCs function.
DCs respond to acetyl-CoA-derived metabolites including citrate, fatty acids, and succinate by altering their functional properties. Succinate receptor signaling enhances DCs migration and TNF-α secretion [76, 180–182], while lauric acid upregulates CD80/CD86 expression [183]. In contrast, n-3 polyunsaturated fatty acids and palmitic acid inhibit DCs maturation and induce an IL-23-driven transcriptome that promotes Treg-mediated immunosuppression [183–185].
These findings collectively demonstrate that acetyl-CoA influences DCs biology through both direct signaling pathway activation and indirect metabolite-mediated mechanisms. The diverse functional outcomes depending on specific metabolites underscore the complexity of acetyl-CoA's role in DCs maturation and differentiation, highlighting the need for precise targeting in therapeutic applications (Fig. 3).
Fig. 3.
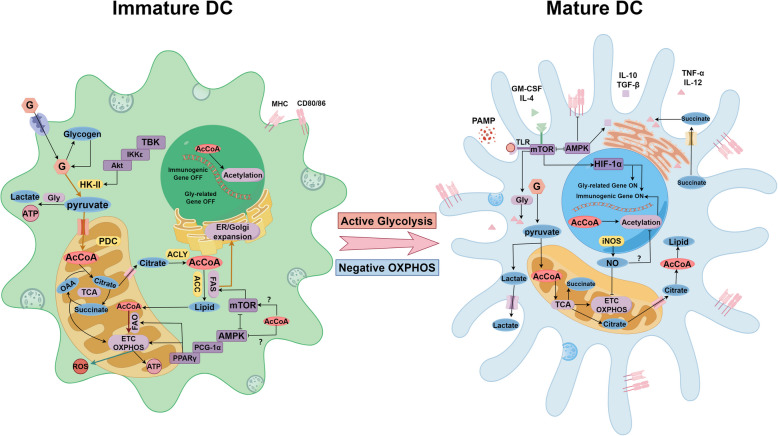
Metabolic differences between different DC states and the regulatory role of acetyl-CoA. As DCs mature, their metabolic profile shifts. Immature DCs initially rely on glycolysis, driven by hexokinase II activated via the TBK-IKKε pathway. Pyruvate, generated from glucose, is converted to acetyl-CoA by PDC and enters the TCA cycle. The electron transport chain and OXPHOS generate ATP to support early DC differentiation, particularly into cDC1. During activation, enhanced OXPHOS and ROS production are crucial for cDC1 and cDC2 differentiation, respectively. Citrate from the TCA cycle is transported to the cytoplasm and converted to acetyl-CoA by ACLY, then to fatty acids by ACC, aiding ER and Golgi expansion. Upon sufficient antigen activation and cytokine stimulation, mature DCs maintain high glycolysis levels, supporting cytokine secretion and migration to lymph nodes. This is regulated by mTOR pathway activation, driving gene expression changes via HIF-1α. HIF-1α and iNOS-derived NO inhibit OXPHOS in mature DCs, while AMPK counteracts the effects of mTOR. The effect of acetyl-CoA on overall metabolic status may regulate mTOR and AMPK. Mature DCs exhibit elevated cytokine secretion and T cell activation but reduced antigen uptake and phagocytosis. Acetyl-CoA derivatives also regulate DC surface molecule expression and cytokine secretion
DCs in non-oncological diseases
It is evident that DCs fulfil a pivotal function in preserving immune system homeostasis and modulating immune responses. They play a pivotal regulatory role in immune activation and tolerance through antigen presentation and cytokine secretion. In recent years, numerous studies have revealed the diversity and complexity of DCs in autoimmune diseases, allergic diseases, transplant immunology, infectious diseases, and chronic inflammatory diseases. This section provides a comprehensive overview of the functional characteristics, immune regulatory mechanisms, and associations with disease progression of DCs in different disease contexts. In addition, it explores their potential applications in disease treatment.
Autoimmune disease
DCs serve as pivotal regulators of both immune activation and tolerance, maintaining systemic homeostasis. In the thymus, DCs promote central tolerance by presenting self-antigens via MHC-II, enabling the clearance of autoreactive CD4+ T cells [186]. Peripheral DCs further enforce tolerance by eliminating self-reactive CD8+ T cells through clonal deletion, incompetence, or Treg induction [187]. Breakdown of these mechanisms underlies the immune dysregulation characteristic of autoimmune diseases. Rheumatoid arthritis (RA) synovium harbors inflammatory DCs that drive pathogenic Th1/Th17 differentiation, a hallmark of disease progression [188]. Both RA and systemic lupus erythematosus (SLE) exhibit DCs accumulation at inflammatory sites, mediated by CCL20-dependent recruitment [189–192]. SLE patients demonstrate particularly pronounced pDCs infiltration in affected skin and kidneys, implicating these cells in tissue-specific pathology [193, 194]. Dermatomyositis lesions show CCR7-deficient DCs that fail to migrate to lymph nodes, promoting their pathological retention and disease exacerbation [195]. Within RA synovium, DCs-derived IL-23 and IL-12 sustain Th1/Th17 dominance over regulatory subsets, perpetuating tissue damage [196]. IL-17 emerges as a central mediator of cartilage destruction, both by amplifying inflammation and directly inducing matrix-degrading enzymes [197–199]. Hypoxia in synovial tissue disrupts OXPHOS and FAO while augmenting glycolysis in cDCs, leading to increased immunoreactivity and enhanced T-cell activation. Conversely, the unique metabolic characteristics of pDCs, likely explain their minimal role in driving inflammation in RA [200]. Metabolite-driven DCs polarization occurs in RA, where succinate sensing triggers pro-inflammatory pathways [201]. In SLE, pDCs initiate autoimmunity via IFN-I-induced B-cell activation and autoantibody production [202]. Metabolic disturbances involving mTOR, ROS, and mitochondrial dysfunction further impair pDCs regulation, with oxidized mtDNA promoting aberrant IFN responses that amplify autoreactive lymphocyte activity [203].
Allergic diseases
DCs are pivotal in allergic responses, orchestrating allergen interactions and T-cell activation. PPAR-γ serves as a critical regulator of DCs-mediated allergic inflammation, with pharmacological activation attenuating DCs responsiveness to respiratory allergens and mitigating Th2-driven airway inflammation [204]. This mechanism involves PPAR-γ-mediated suppression of NF-κB signaling, which diminishes IL-12 production and curbs Th2 differentiation [205]. Elevated FAO is activated by PPAR-γ, fostering tolerogenic DCs development [206]. Pulmonary conventional DCs primarily engulf and process allergens during hypersensitivity reactions before migrating to lymph nodes to prime antigen-specific T-cell responses. Conversely, plasmacytoid DCs promote fungal spore clearance and restrain allergic responses via Treg activation and IL-10 secretion [207, 208]. Langerhans cells, the epidermal DCs population, are fundamentally implicated in atopic dermatitis pathogenesis. Alongside dermal DCs, they drive disease progression by recruiting Th cells through CCL17 and CCL22 chemokine production [209]. DCs-initiated Th2 immunity constitutes a central pathway in atopic dermatitis development, propelled by inflammatory cytokines (IL-31, IL-33, IL-22, IL-4) that promote T-cell recruitment and activation via DCs crosstalk [210]. DCs additionally amplify Th2 and Th22 expansion through non-integrin molecule uptake and DCs-SIGN-mediated IL-6 and TNF-α release [211]. Th subset distribution evolves dynamically during atopic dermatitis progression: Th2 cells dominate acute-phase inflammation, while chronic lesions exhibit mixed Th1, Th17, and Th22 infiltration [212].
Transplantation immunity
In organ transplantation, DCs exhibit dual regulatory roles in allograft rejection through both immunosuppressive and immunostimulatory mechanisms. Hepatic DCs typically demonstrate a tolerogenic phenotype that suppresses immune responses [213], yet during liver transplantation, they shift to a pro-inflammatory state upon exposure to mediators such as TNF-α, IL-6, and activated pattern-recognition receptors. These inflammatory signals correlate strongly with ischemia–reperfusion injury common in transplantation procedures [214, 215]. Mature DCs elevate expression of co-stimulatory molecules, chemokines, and adhesion molecules, enhancing alloantigen presentation and antigen-specific T-cell cytotoxicity. Hepatic cDCs transfer into alloantigen-specific recipients under steady-state conditions induces IL-10-mediated immune tolerance [216]. In kidney transplant rejection, donor and recipient DCs serve distinct functions: recipient DCs present alloantigens while donor DCs facilitate antigen transport. MHC molecule transfer between donor and recipient DCs enables recipient T-cell activation and subsequent graft injury [217]. Ischemia–reperfusion injury activates donor DCs to drive acute rejection, whereas recipient DCs predominantly mediate chronic rejection [218, 219]. Renal cDC2s promotes chronic injury through antibody-dependent pathways involving B-cell activation and antibody production in transplant contexts [220].
Infectious disease
Infections caused by bacteria
Helicobacter pylori secrete ADP-heptose, a metabolic product that attenuates IFN-I signaling during bacterial infection, disrupting DCs activation feedback and impairing CD8+ T cell responses to facilitate chronic infection through TLR2-dependent mechanisms [221]. This DCs dysfunction compromises antitumor immunity, as evidenced by reduced tumor-specific T cell priming in infected mice and shorter progression-free survival among H. pylori-positive lung cancer patients receiving PD-1 blockade therapy [222]. The pathogen simultaneously drives gastric inflammation via its virulence factor CagA, which triggers NF-κB activation in DCs, elevating IL-6 production and promoting IL-17A+ T cell differentiation [223]. These findings reveal how H. pylori balances immune evasion with inflammatory responses through DCs modulation. Therapeutic strategies targeting bacterial metabolites to restore DCs function or modifying microbiota composition may overcome immunosuppression and improve treatment outcomes.
In tuberculosis, DCs play a pivotal role in Mycobacterium tuberculosis (MTB) antigen presentation. Following granuloma formation, DCs transfer antigens to peripheral counterparts, maintaining T-cell activation in draining lymph nodes [224, 225]. TB patients exhibit defective DCs-mediated T-cell priming, resulting in diminished pulmonary T-cell recruitment and markedly lower circulating DCs counts than healthy individuals [226–228]. MTB further subverts host defenses by altering DCs differentiation, increasing moDCs while decreasing IFN-γ-producing CD4+ T cells critical for bacterial clearance [229]. These moDCs display an immunosuppressive profile characterized by reduced IL-12 and elevated IL-10 secretion [230]. Paradoxically, pDCs in TB patients produce abundant IFN-I while adopting anti-inflammatory lung phenotypes, potentially suppressing protective immunity through IL-12 downregulation—a hypothesis supported by post-treatment normalization of pDCs numbers and IL-12+ DCs frequencies [231–233]. Active TB additionally features DCs with upregulated BTLA expression, which correlates with diminished CD80, CD83, and IL-12 production, impairing antigen presentation. These BTLA+ DCs further promote immune tolerance via TGF-β-dependent regulatory T cell induction [234].
Infections caused by viruses
HIV infection causes AIDS by severely compromising the immune system, with CD4+ T cells serving as the primary viral target. DCs play a pivotal role in HIV pathogenesis, frequently encountering the virus during early infection [235]. These cells recognize HIV through both CD4 and DCs-SIGN receptors [236–238]. Following viral uptake, DCs present HIV antigens to CD4+ T cells via MHC-II, primarily through DCs-SIGN interactions with the gp120 glycoprotein [235, 237]. The virological synapse conversely facilitates HIV transfer from DCs to CD4+ T cells, mediated by CD4 and CCR5 binding to gp160 [239, 240]. The antigen presentation capacity of DCs may relate to HIV's N-glycan composition, with increased mannose content potentially enhancing this function [241, 242]. Gp120-exposed plasmacytoid DCs exhibit impaired TLR9 signaling. While viral stimuli normally promote DCs maturation through inflammatory pathways, HIV suppresses TLR-mediated activation in DCs, as demonstrated in multiple studies. TLR agonists could therefore mitigate HIV's suppressive effects by improving viral internalization and immune response initiation in DCs [243, 244].
In HCV infection, DCs provide critical immune defense by detecting viral RNA through TLR7, stimulating IFN and interleukin production to modulate immunity [245]. pDCs recognize RNA from infected hepatocytes, inducing IFN-I production to amplify immune responses [238]. HCV infection diminishes peripheral plasmacytoid DCs counts and disrupts DCs functionality. The virus modifies co-stimulatory molecule and MHC-II expression, modulates IDO via the JAK-STAT pathway, and elevates inhibitory molecules (ICOS, PD-L1), collectively suppressing T-cell activation [246–248]. Studies demonstrate that HCV Core and NS proteins impair DCs function through TLR2 activation, reducing co-stimulatory molecule expression, compromising antigen presentation, and diminishing IL-12 secretion, thereby weakening DCs-mediated immune activation [245, 249].
Infections caused by fungal pathogens
Fungal infections persistently colonize immunocompromised hosts, contributing to substantial mortality and representing a major global health burden [250]. In atopic dermatitis, Malassezia promotes DCs maturation through macrophage mannose receptor-mediated uptake, eliciting a Th2-biased immune response characterized by IL-1β and TNF-α secretion with limited IL-12p70 production. This pathogen further alters DCs-NK cell interactions by suppressing NK cell cytotoxicity while inhibiting DCs apoptosis, thereby perpetuating chronic inflammation [251]. Emerging evidence demonstrates that Malassezia engages DCs through C-type lectin receptors, triggering SYK/CARD9-dependent potassium efflux that activates the NLRP3 inflammasome and IL-1β release, ultimately polarizing Th17/Th22 responses [252]. Lipid antigens derived from both follicular CD1a+ Langerhans cells and Malassezia's Mal f 1 allergen stimulate DCs activation via C-type lectin receptors-1, enhancing expression of TNF-α, IL-1β, and co-stimulatory markers including CD80, which may sustain Th22/Th17-driven inflammation [253, 254].
The opportunistic pathogen Candida induces divergent DCs responses depending on its morphological state: yeast forms stimulate Th1-polarizing IL-12 production, whereas hyphae suppress IL-12 while inducing Th2-associated IL-4, demonstrating morphology-dependent immunomodulation [251]. Candida albicans additionally subverts DCs metabolic pathways by modulating farnesol secretion, which impairs mitochondrial respiration during moDCs differentiation, accelerates lipid droplet accumulation, and compromises protective T cell priming [255]. Future investigations should delineate fungus-specific signaling mechanisms and develop targeted strategies to disrupt these pathogenic pathways.
Infections caused by parasites
DCs serve as critical orchestrators of adaptive immunity in parasitic infections, demonstrating distinct functional adaptations depending on the protozoan or helminth context. We examine DCs-parasite dynamics using leishmaniasis and helminthiasis as model systems. During Leishmania infection, DCs detect pathogen-associated molecular patterns such as lipophosphoglycan through TLR2/4 receptors, triggering p38/JNK MAPK pathways that promote Th1 polarization via IL-12 production [256–258]. Certain species like L. amazonensis disrupt this process by preferentially activating ERK1/2 signaling over p38/JNK, thereby suppressing IL-12 secretion and sustaining pathogenic Th2 responses [259]. The parasite further manipulates host immunity through TLR2-mediated induction of SOCS1/3, which inhibits JAK/STAT signaling and subsequent proinflammatory cytokine release [260]. Interestingly, Leishmania-secreted extracellular vesicles demonstrate both immunostimulatory and suppressive effects, simultaneously enhancing moDCs activation and CD8+ T cell recruitment while facilitating immunosuppressive DCs-NK cell communication [261, 262].
In helminth infections, Schistosoma egg antigens stimulate DCs-SIGN/TLR4 to preferentially activate ERK1/2 rather than p38 MAPK, shifting cytokine production toward IL-10 at the expense of IL-12 [263, 264]. Helminth-derived factors modulate DCs surface markers, upregulating OX40L and PD-L2 to enhance Th2 priming through PD-1 engagement. Ascaris cystatin exemplifies this immunomodulation by simultaneously promoting SREBP-dependent cholesterol biosynthesis and inhibiting CD80 expression [265], and while chronic hookworm infection expands plasmacytoid DCs populations to sustain systemic immune tolerance [266]. Understanding the precise spatiotemporal regulation of DCs-helminth interactions may reveal novel approaches to restore immune homeostasis, potentially leveraging parasite-derived molecules for therapeutic immunomodulation without compromising protective immunity.
Chronic inflammatory diseases
DCs critically influence chronic inflammatory diseases, particularly in establishing intestinal immune tolerance. Their dysfunction strongly correlates with inflammatory bowel disease (IBD) pathogenesis, encompassing both ulcerative colitis (UC) and Crohn's disease (CD). pDCs accumulate at lesion sites and contribute to IBD initiation. CD patients exhibit increased mucosal pDCs infiltration and elevated IL-1β concentrations. Peripheral blood pDCs from IBD patients demonstrate higher CD40/CD86 ratios along with enhanced secretion of IL-6, IL-8, and TNF-α [267, 268]. TNF-α not only drives IBD progression but also suppresses IL-22 production in DCs, impairing mucosal repair. The reduction in DCs populations during acute IBD phases implies their depletion occurs before mucosal damage becomes evident [269–271]. CD103+ DCs in both CD and UC patients show upregulated PD-L1 expression, increased pro-inflammatory cytokine release, and enhanced T-cell activation, likely resulting from Smad7 deficiency and impaired TGF-β signaling [272]. NAFLD represents a major pathway to liver fibrosis and cirrhosis, with its progression involving dysregulated innate immunity through T cells, neutrophils, macrophages, and DCs [273]. Heightened cDC1s activity in non-alcoholic steatohepatitis parallels disease severity and promotes progression by triggering metabolic reprogramming in inflammatory T cells within hepatic lymph nodes [274]. The contribution of DCs to NAFLD pathogenesis remains debated across experimental models. Methionine-choline deficient diet models implicate DCs in pro-inflammatory responses, while contrasting evidence suggests they may exert protective effects [275–277]. Emerging data indicate DCs can suppress fibroproliferation and inflammation during NAFLD resolution [278]. Resolving current discrepancies requires additional investigation to define the precise mechanisms through which DCs influence NAFLD progression (Table 2).
Table 2.
Partial functional and metabolic changes of DC in non-oncological diseases
| Diseases | Main DC subsets | Metabolic change | Functional change | Changed signal pathway | |
|---|---|---|---|---|---|
| Autoimmune Disease | RA [201, 279] | cDC, moDC | Enhanced glycolysis; Reduced OXPHOS; Reduced FAO; Succinate increases | Increased secretion of inflammatory factors; Inducing an increase in Th1/17 differentiation | HIF-1α; JAK-STAT; PPARγ |
| SLE [203, 279] | cDC, pDC | Disruptive mitochondrial metabolism; ROS overload; mtDNA damage | Inducing an increase in Th17 differentiation; pDC secrete more IFN | JAK-STAT; mTOR; cGAS-SING | |
| Allergic Diseases [206, 210] | cDC, pDC | Reduced OXPHOS; Reduced FAO; May depend on the types of lipids and proteins in intestine; Sphingolipids homeostasis is disorder | Increased production of chemokines and inflammatory factors; Inducing B cell secretion of specific antibodies and T cell differentiation | PPARγ; mTOR; ERK-TIM-4; NF-κB | |
| Transplantation reaction [214, 217] | Rejection | cDC | Metabolic changes initiated by ischemia–reperfusion injury; Enhanced sensitivity to ATP and adenosine | Towards Mature state; Enhanced migration to lymph nodes and antigen presentation ability; Release inflammatory factors; Enhanced T cell activation ability | HIF-1α; mTOR |
| Tolerance | cDC | IDO expression upregulated, active tryptophan metabolism; Increased secretion of NO; Enhanced arginine metabolism | Towards semi-mature state; Inducing Treg differentiation; Inhibit T cell killing; High expression of immune checkpoint and anti-inflammatory factors | STAT3; NF-κB | |
| Infectious Disease | Bacteria [10, 223] | cDC, moDC | Enhanced glycolysis driven by TLR; Reduced OXPHOS; Enhanced FAS; iNOS activation; Lipid accumulation and synthesis; | Enhanced antigen phagocytosis in the early stage and enhanced presentation ability in the later stage; Secreting inflammatory factors; Enhanced migration capability | AMPK; PI3K/Akt; mTOR; NF-κB |
| Viruses [238, 248, 280] | pDC, cDC | Inflammation drives normal DC activation related metabolism; Damaged glycolysis and OXPHOS of pDC lead to antiviral immunosuppression; | Damaged metabolism leads to impaired secretion of IFN-I by pDC; Forming virus synapses, leading to the spread of the virus; Inducing immune checkpoint expression leads to immune tolerance | Ras/MAPK; JAK/STAT; PI3K/Akt; mTOR; β-catenin | |
| Fungus [251, 255, 281] | cDC, moDC | Enhanced glycolysis; Damaged OXPHOS of cDC lead to immune activation; Enhanced arginine metabolism; Enhanced FAO and FAS; Lipid accumulation; Damaged OXPHOS of moDC lead to immune inhibition; | Secreting inflammatory factors and inducing Th1/2/17 polarization to activate immune response; | PI3K/Akt; PPAR-γ; mTOR/SREBP; HIF-1α; NF-κB | |
| Parasites [256, 282] | cDC, moDC | Inducing mitochondrial synthesis to enhance OXPHOS and enhancing FAS to support DC activation; Enhancement of OXPHOS and inhibition of protein synthesis leads to immune tolerance; Increase ROS and NO production to kill pathogens | Promote the expression of immune checkpoints and inhibit the expression of co- stimulatory molecules; Inducing Treg differentiation; Hindering the maturity and migration of DC; Inducing Th1/2 differentiation and increasing secretion of pro-inflammatory cytokines | SOCS1/3; MAPK; PI3K; JAK-STAT; PPAR-γ; PGC-1α | |
| Chronic Inflammatory Diseases [120, 272, 273] | cDC, pDC | Enhanced tryptophan metabolism to induce immune tolerance; During the progression of the disease, metabolic characteristics similar to activated DC; Retinoic acid synthesis is beneficial for the formation of tolerant DC | pDC intestinal infiltration increases; pDC co- stimulatory molecule expression and massive release of inflammatory factors; cDC show impaired ability to induce Treg differentiation, but it promotes the activation of Th1/2/17 and produces massive inflammatory factors | PLCβ-PKC; mTOR; AMPK; NF-κB; HIF-1α | |
DCs and tumor immunity
Within the TME, DCs undergo profound functional dysregulation driven by immunosuppressive factors, hypoxic stress, and stromal interactions. These perturbations converge on metabolic reprogramming centered on acetyl-CoA flux, which orchestrates lipid metabolism, OXPHOS, and epigenetic modifications. Consequently, acetyl-CoA availability dictates DCs antigen presentation, migration, and cytokine secretion—pivotal processes for antitumor immunity. Elucidating acetyl-CoA-mediated metabolic-immune crosstalk unveils novel therapeutic strategies to reinvigorate DCs function and overcome tumor immune evasion.
DCs dysfunction in the TME
Tumor derived factors interfere with DCs function
The TME consists of tumor cells, immune cells, stromal cells, cytokines, and microvasculature. Tumor-derived factors facilitate immune evasion by disrupting DCs function and inducing a tolerogenic phenotype, thereby enabling tumors to evade immune detection. TGF-β, produced by Tregs, fibroblasts, and macrophages, establishes an immunosuppressive TME that drives angiogenesis, metastasis, therapy resistance, and epithelial-mesenchymal transition [283]. This cytokine, along with PGE2, upregulates PD-L1 on DCs while stimulating IDO and CCL22 secretion, which promotes Treg differentiation and infiltration [284–286]. Pancreatic cancer models demonstrate that TGF-β/PD-L1 blockade enhances effector DCs activity [287], whereas TGF-β receptor knockdown in DCs boosts IFN-γ production and tumor cell clearance in cholangiocarcinoma models [288]. TGF-β inhibition also potentiates radiotherapy by improving DCs cytoskeletal reorganization, lymph node homing, and CTL activation in ovarian cancer [289]. Emerging evidence indicates that TGF-β disrupts DCs metabolism, compromising antigen processing and migration, with these effects amplified by the acidic TME [290]. Smad4, a critical TGF-β signaling component, restores tumor immunogenicity in Smad4-deficient cells, driving cDC1s activation and antigen uptake [291].
Under physiological conditions, insulin-like growth factor (IGF) stimulates protein synthesis and cell proliferation through ERK and Akt signaling [292]. However, in tumors, IGF impedes TADCs maturation, triggering IL-10 and TNF-α release, which disrupts DCs function and facilitates immune evasion [293]. IL-10, associated with poor clinical outcomes, suppresses DCs activity via JAK-STAT3 signaling, impairing antigen presentation, MHC-I expression, and DCs differentiation [294–298]. It also amplifies Treg-mediated suppression of T-cell activation [299, 300]. By downregulating pro-inflammatory cytokines (e.g., IFN-γ, TNF-α, IL-12) and CCL7, IL-10 further obstructs DCs migration [301, 302]. Neutralizing IL-10 in the TME may improve immunotherapy outcomes.
VEGF, induced by hypoxia via HIF-1, stimulates angiogenesis while inhibiting DCs function through VEGFR binding, which attenuates T-cell responses [303, 304]. VEGFR1 and VEGFR2 distinctly suppress DCs maturation and migration, respectively [305, 306]. Murine studies show that VEGF blockade synergizes with peptide-pulsed DCs to amplify antitumor immunity [307, 308], whereas in vitro, VEGF impedes monocyte differentiation into DCs [305, 309]. In contrast, DCs-derived VEGF-C, upregulated by IFN-γ, drives lymphangiogenesis and DCs maturation, aggravating tissue injury in transplantation and autoimmune disorders [310–312]. VEGF-C also augments DCs trafficking via CCL21, enhancing radiotherapy efficacy in meningioma [313]. These findings highlight the context-dependent duality of VEGF in DCs regulation, shaped by isoform and receptor specificity.
Tumor hypoxic environment and stromal cells disrupt DCs function
The TME demonstrates heightened lipid metabolism that sustains tumor proliferation and migration. Beyond fueling tumor growth, lipids actively suppress immune cell function, thereby accelerating tumor progression [314]. Polyunsaturated fatty acids (PUFAs) play a pivotal role, as their peroxidation yields lipid peroxides [315]. In DCs, PUFA phospholipids initiate COX- and LOX-mediated pathways that generate immunosuppressive mediators such as PGE2 [316]. Hypoxia reduces glutathione peroxidase 4 expression, compromising redox balance and potentially inducing ferroptosis [317, 318]. Ferroptosis-derived peroxidized lipids further suppress T-cell activity and hinder DCs antigen cross-presentation, exacerbating immune evasion [318, 319].
Hypoxic niches accumulate Tregs, M2 macrophages, and tolerogenic DCs [320]. Hypoxia regulates DCs function through ROS: moderate ROS levels stimulate DCs activation via p38-MAPK and ERK1/2 pathways, whereas excessive ROS promotes lipid peroxidation and ER stress, disrupting antigen presentation [107, 321]. TLR-activated DCs generate superoxide anions through NOX2, elevating endosomal pH and impairing antigen processing [322, 323]. While these observations underscore the need for deeper investigation of ROS mechanisms in TADCs, impaired antigen presentation appears more consequential than altered co-stimulatory molecule expression [324, 325]. Hypoxia-induced HIF activation also attenuates DCs migration by suppressing CCR7 and modulates cytokine secretion (e.g., VEGF, IL-12, IFN-γ), thereby inhibiting T-cell priming [326, 327]. Paradoxically, HIF may enhance DCs maturation during inflammation by upregulating glycolytic pathways [328]. Emerging evidence implicates hypoxia-driven DCs dysfunction in hepatocellular carcinoma via the SPP1-CD44 axis [329], warranting further mechanistic studies.
Cancer-associated fibroblasts (CAFs), a dominant stromal component, exhibit strong prognostic associations with disease outcomes [330]. Both CAFs and endothelial cells (ECs) critically modulate immune cell behavior. In liver cancer, activated hepatic stellate cells—the principal CAF precursors—orchestrate key immunosuppressive TME remodeling. CAF-derived IL-6 activates the STAT3 pathway in monocytes, inducing an inhibitory DCs phenotype characterized by downregulated CD86, CD40, and MHC-II [331, 332]. CAFs additionally secrete TGF-β and VEGF to further remodel the TME and support tumor advancement [332]. In lung cancer models, fibroblast-derived COX-2/PGE2 disrupts DCs function, fostering metastatic spread [333]. Col13a1+ CAFs preferentially recruit tumor-associated macrophages and Tregs while excluding DCs, thereby attenuating immune responses [334]. ECs form selective vascular barriers that facilitate Treg infiltration while restricting DCs, effector T cells, and NK cells [335, 336]. Through Stk11-Scf signaling, ECs govern DCs precursor differentiation and drive VEGF-mediated MAPK/ERK activation to induce endothelial-like DCs transformation [337, 338]. Notably, EC/DCs hybrid cells demonstrate superior T-cell stimulatory capacity and antitumor activity in preclinical models, suggesting therapeutic potential for targeting EC-DCs crosstalk [339, 340].
Acetyl-CoA and DCs metabolism
Metabolic reprogramming, a process where cells dynamically adjust their metabolic activities in response to diverse stimuli, engages multiple interconnected metabolic pathways and plays a pivotal role in shaping disease progression [156, 341]. Within the TME, metabolic reprogramming serves as a cornerstone mechanism that profoundly influences the behavior of both tumor cells and immune cells. Among the various metabolic regulators, acetyl-CoA has emerged as a critical factor. Delving into the mechanisms of how acetyl-CoA modulates the metabolism of TADCs could unlock novel therapeutic targets, potentially enhancing the efficacy of TADCs in cancer treatment.
Lipid metabolism reprogramming
Lipid accumulation is a hallmark of TADCs, most notably through elevated triglyceride levels. While this lipid buildup substantially impairs DCs antigen processing capacity, its effect on antigen uptake remains limited. The influence on DCs co-stimulatory molecules and pro-inflammatory cytokines also appears modest [8, 342, 343]. TLR-mediated DCs activation often coincides with a metabolic shift from FAO to FAS, which enhances ER and Golgi function to support cytokine secretion and co-stimulatory molecule expression [3, 77, 201]. Two principal mechanisms drive lipid accumulation in TADCs: the endogenous pathway, potentially activated by SREBP transcription factors that upregulate genes encoding ACLY and FASN [344–346]. and the exogenous pathway, primarily mediated by increased fatty acid uptake through Msr [8], the exogenous pathway likely plays a more dominant role.
TADCs lipid accumulation produces distinct effects compared to normal DCs, potentially through synthesized fatty acids fueling OXPHOS to induce tolerogenic DCs phenotypes, while extracellular lipid uptake from tumor vesicles alters DCs signaling and organelle function [3, 8, 77, 182]. Combining tumor vaccines (Ad-survivin-transduced DCs or tyrosinase peptide-loaded DCs) with the ACC inhibitor TOFA improves therapeutic outcomes [8], possibly by increasing the proportion of DCs-NL (normal lipid DCs) within tumors. Although excessive fatty acid accumulation generally impairs DCs-mediated T cell activation, this effect varies with the TME, potentially explaining observed differences in DCs dysfunction across tumor types [347].
The balance between FAO and FAS also influences DCs differentiation, where etomoxir promotes cDC2s while suppressing cDC1s, contributing to immune dysfunction [13, 348]. In TADCs, nutrient competition may reduce acetyl-CoA availability, activating AMPK to enhance FAO and alter differentiation phenotypes [71, 349, 350], suggesting a mechanism by which acetyl-CoA modulates DCs anti-tumor function. Acetyl-CoA and lipid metabolism further regulate mTOR pathway activity: acetyl-CoA can activate mTORC1 via Raptor acetylation [351], whereas ACC-derived malonyl-CoA binds mTORC1 to inhibit Raptor [352]. This intricate crosstalk between acetyl-CoA, lipid metabolism, and mTOR may underlie the variable efficacy of targeting these pathways individually in cancer therapy.
Oxidative metabolism
Acetyl-CoA participates in the TCA cycle to produce NADH and FADH2 within mitochondria, subsequently fueling the electron transport chain for OXPHOS — the dominant ATP generation pathway in most cells [353]. Impaired OXPHOS in DCs correlates with diminished therapeutic efficacy of DCs vaccines in melanoma patients, establishing a direct link between DCs metabolic state and clinical outcomes [354, 355]. This metabolic impairment primarily stems from restricted acetyl-CoA influx, with OXPHOS efficiency being governed by mitochondrial integrity through mammalian sterile 20-like kinase (Mst) 1/2 regulation [72]. Tumor-mediated Mst signaling disrupts DCs mitochondrial function, suppressing OXPHOS and consequently impairing T-cell activation via the iNOS pathway [79]. Paradoxically, Mst deficiency enhances DCs development in vitro, likely through mitochondrial destabilization that reduces OXPHOS during DCs differentiation [356]. The Mst1/2 pathway appears crucial for acetyl-CoA-mediated OXPHOS regulation, potentially by modulating endogenous acetyl-CoA synthesis in DCs.
Tumors create nutrient competition by aggressively consuming oxygen and resources from their microenvironment [79]. Hypoxia triggers DCs to upregulate HIF-1α, which shifts metabolism toward glycolysis while suppressing OXPHOS [78, 79] and stimulating pro-inflammatory cytokine secretion (TNF-α, IL-6, IL-1β) [357, 358]. However, sustained HIF-1α activation in TADCs induces immune tolerance [359]. This transcription factor inhibits pyruvate dehydrogenase via pyruvate dehydrogenase kinase 1, while its own expression is conversely regulated by acetyl-CoA-dependent histone acetylation at HIF-1α target gene promoters [360, 361]. This reciprocal regulation between HIF-1α and acetyl-CoA suggests a metabolic checkpoint for DCs function that could inform therapeutic strategies.
OXPHOS restoration in DCs enhances immunotherapy responses [355], positioning acetyl-CoA modulation as a promising intervention. However, excessive OXPHOS or FAO risks ROS accumulation that triggers lipid peroxidation, ER stress, and antigen presentation defects [106, 184]. IRE1α-XBP1 activation further disrupts DCs metabolism by driving lipogenesis [106, 362] and inducing DNA damage-mediated apoptosis [104]. TADCs accumulating oxidized lipids exhibit compromised antigen presentation capacity, highlighting the need for precise OXPHOS modulation when using acetyl-CoA to enhance DCs function. Furthermore, the dependence of different DCs subpopulations on OXPHOS exhibited variation. And AMPK activation-driven enhancement of OXPHOS frequently results in the formation of a DCs tolerance phenotype. This further underscore the necessity for further exploration of the effects of acetyl-CoA-driven OXPHOS enhancement on DCs phenotypes under different spatiotemporal localizations.
Acetyl-CoA modulates DCs-mediated antitumor immunity response
The anti-tumor immune function of DCs depends primarily on their ability to present antigens efficiently, migrate to lymph nodes and regulate the immune system through cytokines. These critical functions are profoundly influenced by metabolic reprogramming in the TME, particularly by fluctuations in acetyl-CoA levels. As a key metabolic intermediate and substrate for epigenetic modifications (e.g. histone acetylation), acetyl-CoA regulates the expression of MHC and co-stimulatory molecules, chemokine receptor-mediated migration and the secretion of various key cytokines. Therefore, disruptions to acetyl-CoA metabolism within the TME that interfere with these mechanisms constitute a fundamental basis for DCs dysfunction and tumor immune escape.
Antigen presentation and activation
DCs mediate tumor immunity by presenting antigens via MHC molecules, enabling CD8+ T cells to recognize and eliminate tumor cells [363]. Growing evidence highlights the importance of CD4+ T cell responses triggered by MHC-II in antitumor immunity [364]. Acetyl-CoA modulates MHC gene expression through its involvement in regulatory acetylation events. MHC-II gene expression depends on acetylation dynamics governed by histone acetyltransferases (HATs) and histone deacetylases (HDACs). The master regulator CIITA drives MHC-II transcription through histone acetylation, binding the MHC-II promoter with other transcription factors to acetylate H3 and H4 histones [9]. Acetyl-CoA may thus enhance MHC-II expression by facilitating acetylation of transcriptional activators. Histone H4 acetylation also activates the enhancer A region of MHC-I genes [365]. The transcriptional co-activator p300/CBP maintains MHC-I expression, while its associated factor PCAF counteracts HDAC activity [366]. Metabolic reprogramming in TADCs alters acetyl-CoA availability, potentially impairing MHC expression and antigen presentation to promote immunosuppression. Although cellular acetyl-CoA exhibits compartmentalized distribution [7], elevated FAS and glucose deprivation in TADCs may deplete nuclear acetyl-CoA pools, disrupting expression of MHC-like genes.
CD80/86 serve as essential DCs co-stimulatory molecules, with mitochondrial p32 regulating acetyl-CoA flux into the TCA cycle to sustain citrate levels required for their expression [367]. Lipid mediators like PGE2 modulate both co-stimulatory molecule expression and DCs maturation by suppressing NK cell activity. Tumor-derived PGE2 inhibits DCs maturation and DAMP uptake, consequently impairing T cell activation [368, 369]. COX inhibition can restore DCs immune function [368]. Since prostaglandin synthesis from phospholipids requires acetyl-CoA, recent studies have investigated the interplay between acetyl-CoA and prostaglandin metabolism [370].
DCs migration
DCs must present antigen in lymph nodes to fully activate T cells before these effector cells can migrate to and attack tumors [371, 372]. CCR7 and its ligand CCL21 mediate DCs migration through lymphatic vessels, with inflammatory cytokines further facilitating this process [373–375]. Melanoma patients exhibiting low CCR7 expression show poorer clinical outcomes, underscoring this chemokine's prognostic significance [376]. Cellular metabolism directly regulates CCR7 expression in DCs, as demonstrated by glycolysis-dependent promotion of CCR7 oligomerization and subsequent effects on migratory capacity [83]. Cholesterol metabolism also influences DCs function in immune disorders, where balanced levels stabilize MHC/antigen complexes while excess cholesterol impairs lymph node homing [377–379]. Lipid composition changes during DCs maturation further modulate migration dynamics [380]. While acetyl-CoA's metabolic control over these pathways is established, its specific relationship with DCs migratory behavior in tumors remains an active research focus. Epigenetic regulation through methylation and acetylation additionally governs chemokine expression, though the exact connection to acetyl-CoA requires clarification [376, 381].
Tumor cells suppress DCs recruitment by inhibiting CCL4 and CCL5 secretion via β-catenin pathway activation [103, 382]. In glioblastoma, HMGCL knockdown decreases acetyl-CoA levels and consequently suppresses β-catenin signaling [383]. Aberrant β-catenin activation in TADCs appears to promote immune tolerance, suggesting a mechanistic link between acetyl-CoA metabolism and impaired DCs function [103, 384]. These findings collectively advance our understanding of how acetyl-CoA influences DCs migration and antitumor activity.
Cytokines
Primary T cells depend on persistent MHC molecule stimulation and intermittent IL-7 signaling to sustain long-term survival while preserving homeostasis and functionality [385–387]. TSC1-deficient DCs exhibit reduced T cell activation due to compromised IL-7 secretion [87, 344]. Stromal cells and DCs produce IL-7, a cytokine increasingly employed in T-cell therapies [388–390]. Through STAT5 and mTOR pathway activation, IL-7 induces histone acetylation at the II9 promoter, governing Th9 cell differentiation and antitumor responses [391], with acetyl-CoA serving as a crucial mediator of mTOR-dependent acetylation regulation [87]. These findings suggest acetyl-CoA may influence DCs function both directly and through DCs-mediated modulation of other immune cells.
The PI3K/Akt/mTOR pathway becomes activated in TME cells following ACSS2 upregulation, the primary enzyme for acetyl-CoA production [168, 392–394]. Since both ACSS2 and mTOR regulate DCs cytokine secretion, with acetyl-CoA mediating this effect, an ACSS2-mTOR axis likely governs TADCs functionality. Additionally, DCs suppress CD8+ T cell activation through mTOR-dependent β-catenin signaling that drives IL-10 secretion [3, 87].
pDCs and certain cDC2s subsets produce IFN-I to upregulate DCs MHC expression, enhancing antigen presentation while promoting cDC1s migration and activation [395]. Yet TME inhibit pDCs IFN-I secretion, potentially through TGF-β and other suppressive factors. As tumor-derived TGF-β levels correlate with acetyl-CoA concentrations [56, 396], and given the established link between cellular metabolism and IFN production [157, 397], acetyl-CoA-mediated epigenetic regulation of IFN loci likely contributes to functional differences among tumor-associated DCs subsets (Fig. 4).
Fig. 4.
The effect of acetyl-CoA on DC. a Acetyl CoA and DC metabolism. Pyruvate produced by DC glycolysis is converted to acetyl-CoA to enter TCA cycle, catalyzed by PDC and transported by MPC. Acetyl-CoA regulates the expression of the HIF gene, which inhibits PDC activity via PDK, leading to impaired glycolysis. Acetyl-CoA prepares for oxidative phosphorylation through the TCA cycle, which generates ROS. The accumulation of ROS leads to lipid peroxidation and ER stress, impairing antigen processing and presentation. The lipid synthesis of DCs is also initiated by acetyl-CoA, which is catalyzed by the ACC to produce Mal-CoA. Acetyl-CoA can regulate the activity of mTOR to further influence the overall metabolic phenotype of the cell. Mal-CoA inhibits the mTOR pathway by suppressing Raptor. Mal-CoA also synthesizes lipids, which may contribute to the accumulation of oxidized lipids that interfere with the immune response. b Acetyl CoA and DC function. In the nucleus, acetyl-CoA affects histone acetylation, which controls the expression of genes involved in immune function. Outside the nucleus, glycolysis occurs with CCR7 oligomerization. Citric acid and cholesterol affect CD80/86 expression and MHC stability. The above processes may be regulated by mTOR, affecting DC functions such as antigen presentation, cytokine secretion and migration. This suggests a link between acetyl-CoA and DC function. PDK, pyruvate dehydrogenase kinase
Therapeutic potential and clinical applications
Given the critical role of metabolism in TADCs function, modulating TADCs metabolism has emerged as a promising strategy to enhance tumor immunotherapy efficacy. DCs vaccines represent the most widely used DCs-based treatment, offering relatively mild side effects compared to other approaches. Nevertheless, functional limitations persist, particularly when DCs encounter tumor-associated signals in vivo, often leading to impaired activity. This dysfunction stems from intricate cellular interactions within the TME. Acetyl-CoA not only regulates TADCs function but also influences T-cell activity and tumor cell behavior. Consequently, therapeutic targeting of acetyl-CoA metabolism may provide an innovative approach to improve DCs immunotherapy outcomes.
Acetyl-CoA-targeting strategies
The efficacy of DCs vaccines depends critically on successful tumor antigen loading, and tumor cell lysates effectively enhance DCs-mediated T cell activation [398–400]. Acetyl-CoA regulates autophagy through acetylation, modulating both autophagy gene transcription and protein modifications [401]. In colon cancer, overexpression of PTEN-inducible kinase 1 depletes acetyl-CoA and induces tumor cell autophagy [159]. The mTOR and AMPK pathways respond to acetyl-CoA fluctuations, thereby regulating autophagy initiation [170]. This acetyl-CoA-mediated autophagic mechanism may improve DCs therapy outcomes.
Acetyl-CoA directly enhances DCs antitumor activity by modulating IL-12 production, a pivotal cytokine for DCs vaccine efficacy [402]. While ACC inhibitors suppress IL-12 secretion [403], TOFA synergizes with DCs vaccines to improve therapeutic outcomes[8], suggesting DCs maturation state influences these metabolic interventions. TLR3 agonists like ARNAX induce antitumor immunity in DCs through cytokine-independent mechanisms [404]. Although poly I:C-activated TLR3 triggers acetyl-CoA-dependent acetylation in endothelial cells [405], its role in DCs remains underexplored and warrants further investigation.
Metabolic heterogeneity among DCs subsets suggests that acetyl-CoA manipulation could steer in vitro differentiation toward more effective vaccine phenotypes. CD8α+ DCs rely heavily on oxidative metabolism and dominate IL-12 production, enabling robust CD8+ T cell activation [72, 406]. As discussed earlier, acetyl-CoA promotes this oxidative phenotype, implying its utility in engineering DCs for enhanced antitumor immunity. Acetyl-CoA further intersects with PD-1 signaling—maintaining c-Myc acetylation to upregulate PD-L1 in NSCLC while PD-1 suppresses T cell proliferation via ACLY inhibition [407, 408]. In PD-1-deficient models, elevated mitochondrial metabolites like cholesterol drive DCs inflammatory polarization and antigen presentation [400, 409], supporting combined acetyl-CoA modulation with DCs vaccines to potentiate immune checkpoint inhibition. Immune checkpoint silencing in DCs augments their T cell stimulatory capacity [410, 411]. Further combination of such modified DCs may be able to increase the sensitivity of tumor cells to ICI treatment while avoiding checkpoint inhibition of DCs vaccines.
Acetyl-CoA-metabolizing enzymes also contribute directly to tumorigenesis [412, 413], prompting clinical development of inhibitors targeting ACC (PF-05221304), ACSS2 (MTB-9655), and ACLY (SB-204990179) [414–418] (Table 3).
Table 3.
Application of Acetyl-CoA related targets in cancer therapy
| Target | Molecule | Cancer | Status |
|---|---|---|---|
| ACC | TOFA [419–421] | ESCC, Breast cancer, OV | Preclinical |
| ND-646 [422, 423] | CRC, PAAD, Lung cancer | Preclinical | |
| ND-654 [424] | HCC | Preclinical | |
| phenoxy-phenyl isoxazoles [425] | Breast cancer | Preclinical | |
| ND-630 [426] | PRAD | Preclinical | |
| Bezafibrate [427, 428] | NSCLC | Phase I trial (UMIN000017854) | |
| Soraphen A [429] | Breast cancer | Preclinical | |
| Bezafibrate [428] | Breast cancer | Preclinical | |
| ACLY | BMS-303141 [418] | HCC | Preclinical |
| Hydroxycitrate [430] | PRAD, SKCM, Lung cancer | Preclinical | |
| SB-204990 [431] | Thyroid cancer | Preclinical | |
| Bempedoic acid/ETC-1002 [432] | HCC | Preclinical | |
| Cucurbitacin B [433] | PRAD | Preclinical | |
| Emodin derivatives [434] | Lung cancer | Preclinical | |
| HCA[435] | PAAD | Preclinical | |
| NDI-091143[431] | Thyroid cancer | Preclinical | |
| PDC | CPI-163 [436] | Leukemia | Phase I trial |
| DCA [437, 438] | Brain tumor | Phase I trial (NCT01111097) | |
| Soid tumor | Phase I trial | ||
| HMGCR | Statin [439, 440] | Head and neck cancer | Approved |
| PRAD | Preclinical | ||
| ACSS2 | VY-3–249 [441] | Breast cancer | Preclinical |
| AD-8007 [442] | Brain metastases | Preclinical | |
| KAT | WM-1119 [443] | Lymphoma | Preclinical |
| PF-07248144 [444] | Breast cancer | Phase I trial (NCT04606446) | |
| CCS1477 [445] | PRAD | Phase I/II trial (NCT03568656) | |
| FT-6876 [446] | Breast cancer | Preclinical | |
| FT-7051 [446] | PRAD | Phase I trial (NCT04575766) | |
| Curcumin [447] | CRC | Preclinical | |
| WM-3855 [448] | PRAD | Preclinical | |
| MG149 [449] | Gallbladder cancer | Preclinical | |
| ICG-001 [450] | Endometrial cancer | Preclinical | |
| HDAC | Vorinostat [451] | Cervical cancer | Approved |
| Resminostat [452] | PAAD | Approved | |
| Romidepsin [453] | Lymphoma | Phase I/II trial (NCT02783625) | |
| Panobinostat [454] | Myeloma | Phase III trial | |
| Belinostat [455] | PTCL | Approved | |
| LMK-235 [456] | PAAD | Preclinical | |
| Ricolinostat [457] | Breast cancer | Phase I trial (NCT02632071) | |
| Chidamide [458] | CRC | Phase II trial (NCT04724239) | |
| Tucidinostat [459] | PTCL | Phase II trial (NCT02953652) | |
| Entinostat [460] | NSCLC | Phase I trial (NCT04631029) | |
| FASN | Orlistat [461] | CRC | Preclinical |
| TVB-2640 [462] | HCC | Preclinical | |
| Cerulenin [463] | Cervical cancer | Preclinical | |
| C75 [464] | Breast cancer | Preclinical | |
| Omeprazole [465] | PRAD | Phase II trial (NCT04337580) | |
| CPT | Etomoxir [466] | Breast cancer, Leukemia | Preclinical |
| St1326 [467] | Lymphoma | Preclinical | |
| Perhexiline [468] | Glioblastoma | Preclinical | |
| CRAT | Mildronate [469] | HCC | Preclinical |
| SIRT1 | Resveratrol [470] | Cervical cancer | Preclinical |
| GLS | CB-839 [471] | Breast cancer | Preclinical |
| DRP-104 [472] | PAAD | Preclinical | |
| IACS-6274 [473, 474] | NSCLC | Preclinical |
ACC Acetyl-CoA carboxylase, ACLY ATP citrate lyase, ACSS2 Acetyl-CoA synthetase 2, BRCA CPT Carnitine palmitoyltransferase, CRC Colorectal cance, CRAT Carnitine acetyltransferase, ESCC Esophageal squamous cell carcinoma, FASN fatty acid synthase, GLS Glutaminase, HCC Hepatocellular carcinoma, HDAC Histone deacetylases, HMGCR 3-hydroxy-3- methylglutaryl-CoA reductase, KAT Lysine acetyltransferase, NSCLC Non-small cell lung cancer, OV Ovarian carcinoma, PAAD Pancreatic adenocarcinoma, PDC pyruvate dehydrogenase complex, PRAD Prostate adenocarcinoma, PTCL Peripheral T cell lymphomas, SIRT1 Sirtuin 1, SKCM skin cutaneous melanoma
In conclusion, acetyl-CoA may regulate DCs and DCs-associated cells through multi-omics mechanisms at metabolic and genetic levels, potentially enhancing the therapeutic efficacy of DCs-based treatments. However, further studies are required to validate its clinical applicability and broader potential (Fig. 5).
Fig. 5.
The therapeutic potential of acetyl-CoA. DC vaccines are the most widely used DC-based immunotherapy. By collecting blood from patients and sorting the cells, we can induce differentiation and maturation using activating factors. Acetyl-CoA may target the metabolism to control differentiation and maturation, and modulating this process with acetyl-CoA may further promote the generation of more effector phenotypes of DC. Co-culturing tumor antigens with activated DCs to load them with antigens and using acetyl-CoA to modulate tumor autophagy and DC phagocytosis may help tum our vaccines work better. The efficacy of antigen-loaded DCs re-infused into the patient depends on T cells being activated by molecules like IL-12 and CD80/86. The effect of acetyl-CoA on these molecules shows promise in improving DC efficacy. Acetyl-CoA affects other cells in the TME, making it a fully integrated strategy for therapeutic development
DCs metabolic engineering
Targeting lipid metabolism with TOFA and FASN inhibitors like cerulenin restored TADCs immunological activity across multiple tumor types, while simultaneously reducing aberrant lipid accumulation [8, 475]. The XBP1 pathway not only drives endogenous FAS in TADCs but also mediates ER stress, with polyethyleneimine-siRNA nanocomposites targeting IRE1α-XBP1 effectively suppressing lipid accumulation and boosting TADCs immunostimulatory function [104]. The in situ nanovaccine TPOP demonstrated dual efficacy in modulating lipid metabolism and activating innate immunity, thereby improving TADCs cross-presentation [476]. Another nanoparticle, TS-PP@FU, binds TADCs Msr1 to deliver triple metabolic inhibition—blocking exogenous lipid uptake, endogenous synthesis, and lipid-related gene transcription—which restored TADCs antitumor capacity in cytokine secretion and CTL activation [102]. Notably, combining these lipid-targeting approaches with ICI therapy substantially enhanced treatment efficacy [8, 102, 476].
PGE2-mediated TADCs suppression was reversed by COX-2 inhibition with NS-398, while aspirin and celecoxib improved anti-PD-1 responses in melanoma [477, 478]. EP4 receptor antagonists AAT-008 and E7046 increased cDC1s frequency and CXCL10-dependent CTL recruitment respectively, with E7046 synergizing with anti-CTLA-4 in colon cancer models [479].
Tryptophan metabolism similarly impairs tumor-infiltrating immune cells, where tumor-secreted IDO inhibits TADCs function. Clinical-stage IDO inhibitors like epacadostat enhanced DCs cytokine production while suppressing Tregs [480], whereas indoximod potentiated DCs vaccine responses by downregulating IDO via AhR signaling [481, 482].
Combination therapies
Combination therapies demonstrate significant efficacy in cancer treatment, particularly immune checkpoint blockade and its integration with conventional radiotherapy or chemotherapy. Integrating DCs therapy with chemotherapy, adjuvants, or ICI improves outcomes while reducing adverse effects [483]. DCs metabolism directly influences immune checkpoint expression and activation, with PD-L1, CD40, and TIM-3 levels modulated by the interplay between mitochondrial metabolism and glycolysis in DCs subsets. TME-derived lactate suppresses both OXPHOS and glycolysis in DCs via monocarboxylate transporters, impairing DCs maturation and subsequent T cell activation. Concurrent metabolic inhibition with DCs vaccines and ICI therapy could restore antitumor responses and potentiate immune checkpoint blockade [4]. Pretreating DCs vaccines with metabolic inhibitors or administering these agents in vivo may heighten patient responsiveness to ICI and chemotherapy. Emerging evidence suggests mTOR inhibitors amplify tumor vaccine efficacy through both systemic delivery and ex vivo DCs treatment [88, 484]. While chemotherapy triggers immunogenic cell death and promotes DCs maturation, the scarcity of Ly6c+CD103+ DCs—critical for antitumor immunity—limits its effectiveness. BTK-IDO axis inhibition attenuates tryptophan metabolism while stimulating differentiation of these DCs subsets, potentially augmenting chemotherapy responses [485]. Paradoxically, chemotherapy elevates IDO expression in DCs; 1-MT-mediated IDO inhibition reduces Treg infiltration, enhances antitumor immunity, and improves survival [486]. Metabolic checkpoint targeting in DCs could optimize vaccine resistance to the TME, serving as a pivotal adjuvant strategy when combined with radiotherapy, chemotherapy, ICI, or CAR-T therapies. Restructuring DCs and global tumor metabolism may prove essential for overcoming immunosuppression. In particular, modifications that target the immunosuppressive crosstalk between dendritic cells and T cells may hold the key to achieving a significant improvement in patient survival [487]. Preclinical data indicate metabolic checkpoint modulation generates an immune-permissive TME, potentially improving CAR-T/CAR-NK cell therapy efficacy and ICI sensitivity.
Conclusion
DCs are crucial for antitumor immunity, but their dysfunction in the TME limits immunotherapy efficacy. This review examines DCs ontogeny, subset diversity, and metabolic flexibility, with a focus on acetyl-CoA as a key metabolic regulator of immunometabolism reprogramming. We analyze how acetyl-CoA regulates lipid synthesis, mitochondrial dynamics, and epigenetic changes in DCs, linking these processes to antigen presentation, T cell activation, and migration deficits in tumors. Additionally, we explore different principles of DCs dysfunction in infections, autoimmune diseases, and chronic inflammation to achieve comprehensive recognition of DCs functional state changes.
Acetyl-CoA directs DCs functional fate through three interconnected pathways. In lipid metabolism, TADCs exhibit pathological lipid accumulation from disordered FAS and Msr-mediated uptake. This overload impairs antigen cross-presentation via ER stress and oxidized lipid-induced MHC-peptide destabilization. ACC or FAO inhibition restores DCs immune activity and enhances ICI-induced T cell infiltration. Acetyl-CoA-driven FAS normally supports DCs activation but becomes pathological in the TME, possibly due to altered lipid species and localization. DCs subset-specific transcriptional regulation of metabolic enzymes may further dictate lipid responses. Epigenetically, acetyl-CoA regulates histone acetylation at immunogenic loci via KATs. TADCs hypoacetylation, caused by acetyl-CoA depletion or NO-mediated KAT inhibition, may silence immune activation genes. HDAC inhibitors or acetyl-CoA supplementation could be potential treatments, though outcome may be limited by incomplete understanding of acetylation dynamics and DCs subset heterogeneity. In mitochondrial dynamics, acetyl-CoA supports OXPHOS for DCs migration and cDC1s-mediated IL-12 secretion/CD8+ T cell activation. The TME may disrupt this via hypoxia/NO-induced ETC inhibition and HIF-1α-driven tolerance. However, OXPHOS effects are condition-dependent, with excessive FAO-driven OXPHOS potentially causing ROS-mediated lipid peroxidation. Precise modulation based on DCs state and microenvironmental cues is essential.
Translational insights underscore the potential of acetyl-CoA-centric therapies. Metabolic checkpoint targeting via ACLY or ACC inhibition reduces tumor lipids and enhances DCs antigen presentation. Advanced nanoformulations enable co-delivery of metabolic inhibitors with immune agonists, reprogramming TADCs and reversing checkpoint-driven exhaustion. In the future, it may become feasible to achieve local remodeling of DCs metabolism by further developing pH/ROS-responsive vectors for precise release of metabolic checkpoint inhibitors or acetyl-CoA derivatives. This approach could facilitate precise disease management and address the necessity for adaptation to the internal environment. Combination strategies integrating acetyl-CoA pathway modulation with DCs vaccines may be helpful to combat functional impairment in TME. Thus, metabolic checkpoint-modified DCs vaccines are expected to be a novel adjuvant therapy. Strategic integration with radiotherapy, chemotherapy, or ICI may enhance antitumor efficacy through broad immune activation.
Metabolic regulation, particularly through metabolic pathways integrated by acetyl-CoA, is a key signal that shapes DCs function. Exploring spatiotemporal dynamics of acetyl-CoA metabolism in DCs can link metabolism to immunity, enhancing immunotherapy approaches. The acetyl-CoA-DCs axis might offer new routes for immunometabolism therapies, helping remodel the TME and turn “cold” tumors “hot”.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grant numbers: 82071796) and the National Key R&D Program of China (2022YF A1305700). The figures were assembled in Figdraw (https://www.figdraw.com/#).
Authors’ contributions
YW provided guidance on the research direction and ideas for this paper and participated in the supervision and editing of the manuscript. GY provided research ideas and made manuscript revisions, and participated in immunological discussions. WL is responsible for the initial drafting and subsequent revisions of the manuscript, conducted an exhaustive search of the relevant literature, and created the illustrations. CY helped write the manuscript and drew up the tables. XZ and YG searched relevant literature and participated in content discussions. XH, RX and JX provided effective assistance and participated in the improvement of the content, offering numerous practical suggestions that enhanced the article. All authors have read and approved the final manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (Grant numbers: 82071796) and the National Key R&D Program of China (2022YF A1305700).
Data availability
No applicable.
Declarations
Ethics approval and consent to participate
No applicable.
Consent for publication
No applicable.
Competing interests
The author asserts that no competitive or conflicting interests are present in this article.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Ganjun Yu, Email: yugj@immunol.org.
Yanfeng Wu, Email: wuyf@immunol.org.
References
- 1.Gardner A, Ruffell B. DCs and Cancer Immunity. Trends Immunol. 2016;37(12):855–65. 10.1016/j.it.2016.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Murphy TL, Grajales-Reyes GE, Wu X, Tussiwand R, Briseño CG, Iwata A, et al. Transcriptional Control of DC Development. Annu Rev Immunol. 2016;34:93–119. 10.1146/annurev-immunol-032713-120204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Giovanelli P, Sandoval TA, Cubillos-Ruiz JR. DC Metabolism and Function in Tumors. Trends Immunol. 2019;40(8):699–718. 10.1016/j.it.2019.06.004. [DOI] [PubMed] [Google Scholar]
- 4.Niveau C, Cettour-Cave M, Mouret S, Sosa Cuevas E, Pezet M, Roubinet B, et al. MCT1 lactate transporter blockade re-invigorates anti-tumor immunity through metabolic rewiring of dendritic cells in melanoma. Nat Commun. 2025;16(1):1083. 10.1038/s41467-025-56392-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wang C, Ma J, Zhang N, Yang Q, Jin Y, Wang Y. The acetyl-CoA carboxylase enzyme: a target for cancer therapy? Expert Rev Anticancer Ther. 2015;15(6):667–76. 10.1586/14737140.2015.1038246. [DOI] [PubMed] [Google Scholar]
- 6.Ghosh-Choudhary S, Liu J, Finkel T. Metabolic regulation of cell fate and function. Trends Cell Biol. 2020;30(3):201–12. 10.1016/j.tcb.2019.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Guertin DA, Wellen KE. Acetyl-CoA metabolism in cancer. Nat Rev Cancer. 2023;23(3):156–72. 10.1038/s41568-022-00543-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Herber DL, Cao W, Nefedova Y, Novitskiy SV, Nagaraj S, Tyurin VA, et al. Lipid accumulation and DC dysfunction in cancer. Nat Med. 2010;16(8):880–6. 10.1038/nm.2172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Beresford GW, Boss JM. CIITA coordinates multiple histone acetylation modifications at the HLA-DRA promoter. Nat Immunol. 2001;2(7):652–7. 10.1038/89810. [DOI] [PubMed] [Google Scholar]
- 10.Du X, Chapman NM, Chi H. Emerging roles of cellular metabolism in regulating DC subsets and function. Front Cell Dev Biol. 2018;6:152. 10.3389/fcell.2018.00152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Jhunjhunwala S, Hammer C, Delamarre L. Antigen presentation in cancer: insights into tumour immunogenicity and immune evasion. Nat Rev Cancer. 2021;21(5):298–312. 10.1038/s41568-021-00339-z. [DOI] [PubMed] [Google Scholar]
- 12.Heras-Murillo I, Adán-Barrientos I, Galán M, Wculek SK, Sancho D. Dendritic cells as orchestrators of anticancer immunity and immunotherapy. Nat Rev Clin Oncol. 2024;21(4):257–77. 10.1038/s41571-024-00859-1. [DOI] [PubMed] [Google Scholar]
- 13.Anderson DA 3rd, Dutertre CA, Ginhoux F, Murphy KM. Genetic models of human and mouse dendritic cell development and function. Nat Rev Immunol. 2021;21(2):101–15. 10.1038/s41577-020-00413-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Onai N, Obata-Onai A, Schmid MA, Ohteki T, Jarrossay D, Manz MG. Identification of clonogenic common Flt3+M-CSFR+ plasmacytoid and conventional dendritic cell progenitors in mouse bone marrow. Nat Immunol. 2007;8(11):1207–16. 10.1038/ni1518. [DOI] [PubMed] [Google Scholar]
- 15.Musumeci A, Lutz K, Winheim E, Krug AB. What makes a pDC: recent advances in understanding plasmacytoid DC development and heterogeneity. Front Immunol. 2019;10:1222. 10.3389/fimmu.2019.01222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zeng Q, Zhou Y, Schwarz H. CD137L-DCs, potent immune-stimulators-history, characteristics, and perspectives. Front Immunol. 2019;10:2216. 10.3389/fimmu.2019.02216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Greter M, Helft J, Chow A, Hashimoto D, Mortha A, Agudo-Cantero J, et al. GM-CSF controls nonlymphoid tissue dendritic cell homeostasis but is dispensable for the differentiation of inflammatory dendritic cells. Immunity. 2012;36(6):1031–46. 10.1016/j.immuni.2012.03.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Esashi E, Wang YH, Perng O, Qin XF, Liu YJ, Watowich SS. The signal transducer STAT5 inhibits plasmacytoid dendritic cell development by suppressing transcription factor IRF8. Immunity. 2008;28(4):509–20. 10.1016/j.immuni.2008.02.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Edwards AD, Chaussabel D, Tomlinson S, Schulz O, Sher A, Reis e Sousa C. Relationships among murine CD11c(high) DC subsets as revealed by baseline gene expression patterns. J Immunol. 2003;171(1):47–60. 10.4049/jimmunol.171.1.47 [DOI] [PubMed]
- 20.Comi M, Avancini D, Santoni de Sio F, Villa M, Uyeda MJ, Floris M, et al. Coexpression of CD163 and CD141 identifies human circulating IL-10-producing DCs (DC-10). Cell Mol Immunol. 2020;17(1):95–107. 10.1038/s41423-019-0218-0 [DOI] [PMC free article] [PubMed]
- 21.Loughland JR, Woodberry T, Boyle MJ, Tipping PE, Piera KA, Amante FH, et al. Plasmodium falciparum Activates CD16+ DCs to Produce Tumor Necrosis Factor and Interleukin-10 in Subpatent Malaria. J Infect Dis. 2019;219(4):660–71. 10.1093/infdis/jiy555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Satoh T, Toledo MAS, Boehnke J, Olschok K, Flosdorf N, Götz K, et al. Human DC3 Antigen Presenting DCs From Induced Pluripotent Stem Cells. Front Cell Dev Biol. 2021;9: 667304. 10.3389/fcell.2021.667304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Fromm PD, Silveira PA, Hsu JL, Papadimitrious MS, Lo TH, Ju X, et al. Distinguishing human peripheral blood CD16+ myeloid cells based on phenotypic characteristics. J Leukoc Biol. 2020;107(2):323–39. 10.1002/JLB.5A1119-362RRR. [DOI] [PubMed] [Google Scholar]
- 24.Worah K, Mathan TSM, Vu Manh TP, Keerthikumar S, Schreibelt G, Tel J, et al. Proteomics of Human DC Subsets Reveals Subset-Specific Surface Markers and Differential Inflammasome Function. Cell Rep. 2016;16(11):2953–66. 10.1016/j.celrep.2016.08.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Reizis B. Plasmacytoid DCs: development, regulation, and function. Immunity. 2019;50(1):37–50. 10.1016/j.immuni.2018.12.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Balan S, Saxena M, Bhardwaj N. Dendritic cell subsets and locations. Int Rev Cell Mol Biol. 2019;348:1–68. 10.1016/bs.ircmb.2019.07.004. [DOI] [PubMed] [Google Scholar]
- 27.Guilliams M, Dutertre CA, Scott CL, McGovern N, Sichien D, Chakarov S, et al. Unsupervised high-dimensional analysis aligns dendritic cells across tissues and species. Immunity. 2016;45(3):669–84. 10.1016/j.immuni.2016.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Yoshida H, Lareau CA, Ramirez RN, Rose SA, Maier B, Wroblewska A, et al. The cis-Regulatory Atlas of the Mouse Immune System. Cell. 2019;176(4):897–912. 10.1016/j.cell.2018.12.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Nutt SL, Chopin M. Transcriptional networks driving dendritic cell differentiation and function. Immunity. 2020;52(6):942–56. 10.1016/j.immuni.2020.05.005. [DOI] [PubMed] [Google Scholar]
- 30.Maier B, Leader AM, Chen ST, Tung N, Chang C, LeBerichel J, et al. A conserved dendritic-cell regulatory program limits antitumour immunity. Nature. 2020;580(7802):257–62. 10.1038/s41586-020-2134-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Brown CC, Gudjonson H, Pritykin Y, Deep D, Lavallée VP, Mendoza A, et al. Transcriptional Basis of Mouse and Human Dendritic Cell Heterogeneity. Cell. 2019;179(4):846–63. 10.1016/j.cell.2019.09.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Pittet MJ, Di Pilato M, Garris C, Mempel TR. Dendritic cells as shepherds of T cell immunity in cancer. Immunity. 2023;56(10):2218–30. 10.1016/j.immuni.2023.08.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Di Blasio S, van Wigcheren GF, Becker A, van Duffelen A, Gorris M, Verrijp K, et al. The tumour microenvironment shapes dendritic cell plasticity in a human organotypic melanoma culture. Nat Commun. 2020;11(1):2749. 10.1038/s41467-020-16583-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hargadon KM, Bishop JD, Brandt JP, Hand ZC, Ararso YT, Forrest OA. Melanoma-derived factors alter the maturation and activation of differentiated tissue-resident dendritic cells. Immunol Cell Biol. 2016;94(1):24–38. 10.1038/icb.2015.58. [DOI] [PubMed] [Google Scholar]
- 35.Wculek SK, Cueto FJ, Mujal AM, Melero I, Krummel MF, Sancho D. Dendritic cells in cancer immunology and immunotherapy. Nat Rev Immunol. 2020;20(1):7–24. 10.1038/s41577-019-0210-z. [DOI] [PubMed] [Google Scholar]
- 36.Macri C, Pang ES, Patton T, O’Keeffe M. Dendritic cell subsets. Semin Cell Dev Biol. 2018;84:11–21. 10.1016/j.semcdb.2017.12.009. [DOI] [PubMed] [Google Scholar]
- 37.Del Prete A, Salvi V, Soriani A, Laffranchi M, Sozio F, Bosisio D, et al. Dendritic cell subsets in cancer immunity and tumor antigen sensing. Cell Mol Immunol. 2023;20(5):432–47. 10.1038/s41423-023-00990-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Trinchieri G. Interleukin-12 and the regulation of innate resistance and adaptive immunity. Nat Rev Immunol. 2003;3(2):133–46. 10.1038/nri1001. [DOI] [PubMed] [Google Scholar]
- 39.Xu YD, Cheng M, Shang PP, Yang YQ. Role of IL-6 in dendritic cell functions. J Leukoc Biol. 2022;111(3):695–709. 10.1002/JLB.3MR0621-616RR. [DOI] [PubMed] [Google Scholar]
- 40.Spranger S, Dai D, Horton B, Gajewski TF. Tumor-residing Batf3 dendritic cells are required for effector T cell trafficking and adoptive T cell therapy. Cancer Cell. 2017;31(5):711–23. 10.1016/j.ccell.2017.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Cao LL, Kagan JC. Targeting innate immune pathways for cancer immunotherapy. Immunity. 2023;56(10):2206–17. 10.1016/j.immuni.2023.07.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Deng L, Liang H, Xu M, Yang X, Burnette B, Arina A, et al. STING-dependent cytosolic DNA sensing promotes radiation-induced type I interferon-dependent antitumor immunity in immunogenic tumors. Immunity. 2014;41(5):843–52. 10.1016/j.immuni.2014.10.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Lam KC, Araya RE, Huang A, Chen Q, Di Modica M, Rodrigues RR, et al. Microbiota triggers STING-type I IFN-dependent monocyte reprogramming of the tumor microenvironment. Cell. 2021;184(21):5338–56. 10.1016/j.cell.2021.09.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.O’Keeffe M, Mok WH, Radford KJ. Human dendritic cell subsets and function in health and disease. Cell Mol Life Sci. 2015;72(22):4309–25. 10.1007/s00018-015-2005-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Seya T, Shime H, Takeda Y, Tatematsu M, Takashima K, Matsumoto M. Adjuvant for vaccine immunotherapy of cancer–focusing on Toll-like receptor 2 and 3 agonists for safely enhancing antitumor immunity. Cancer Sci. 2015;106(12):1659–68. 10.1111/cas.12824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Saito Y, Komori S, Kotani T, Murata Y, Matozaki T. The role of type-2 conventional dendritic cells in the regulation of tumor immunity. Cancers. 2022;14(8):1976. 10.3390/cancers14081976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ahrends T, Spanjaard A, Pilzecker B, Bąbała N, Bovens A, Xiao Y, et al. CD4+ T Cell Help Confers a Cytotoxic T Cell Effector Program Including Coinhibitory Receptor Downregulation and Increased Tissue Invasiveness. Immunity. 2017;47(5):848–61. 10.1016/j.immuni.2017.10.009. [DOI] [PubMed] [Google Scholar]
- 48.Yin X, Yu H, Jin X, Li J, Guo H, Shi Q, et al. Human blood CD1c+ dendritic cells encompass CD5high and CD5low subsets that differ significantly in phenotype, gene expression, and functions. J Immunol. 2017;198(4):1553–64. 10.4049/jimmunol.1600193. [DOI] [PubMed] [Google Scholar]
- 49.Mashayekhi M, Sandau MM, Dunay IR, Frickel EM, Khan A, Goldszmid RS, et al. CD8α(+) dendritic cells are the critical source of interleukin-12 that controls acute infection by Toxoplasma gondii tachyzoites. Immunity. 2011;35(2):249–59. 10.1016/j.immuni.2011.08.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Shedlock DJ, Shen H. Requirement for CD4 T cell help in generating functional CD8 T cell memory. Science. 2003;300(5617):337–9. 10.1126/science.1082305. [DOI] [PubMed] [Google Scholar]
- 51.Binnewies M, Mujal AM, Pollack JL, Combes AJ, Hardison EA, Barry KC, et al. Unleashing Type-2 Dendritic Cells to Drive Protective Antitumor CD4+ T Cell Immunity. Cell. 2019;177(3):556-71.e16. 10.1016/j.cell.2019.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Dasgupta S, Erturk-Hasdemir D, Ochoa-Reparaz J, Reinecker HC, Kasper DL. Plasmacytoid dendritic cells mediate anti-inflammatory responses to a gut commensal molecule via both innate and adaptive mechanisms. Cell Host Microbe. 2014;15(4):413–23. 10.1016/j.chom.2014.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Cao W, Manicassamy S, Tang H, Kasturi SP, Pirani A, Murthy N, et al. Toll-like receptor-mediated induction of type I interferon in plasmacytoid dendritic cells requires the rapamycin-sensitive PI(3)K-mTOR-p70S6K pathway. Nat Immunol. 2008;9(10):1157–64. 10.1038/ni.1645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Zitvogel L, Galluzzi L, Kepp O, Smyth MJ, Kroemer G. Type I interferons in anticancer immunity. Nat Rev Immunol. 2015;15(7):405–14. 10.1038/nri3845. [DOI] [PubMed] [Google Scholar]
- 55.Vermi W, Bonecchi R, Facchetti F, Bianchi D, Sozzani S, Festa S, et al. Recruitment of immature plasmacytoid dendritic cells (plasmacytoid monocytes) and myeloid dendritic cells in primary cutaneous melanomas. J Pathol. 2003;200(2):255–68. 10.1002/path.1344. [DOI] [PubMed] [Google Scholar]
- 56.Sisirak V, Vey N, Goutagny N, Renaudineau S, Malfroy M, Thys S, et al. Breast cancer-derived transforming growth factor-β and tumor necrosis factor-α compromise interferon-α production by tumor-associated plasmacytoid dendritic cells. Int J Cancer. 2013;133(3):771–8. 10.1002/ijc.28072. [DOI] [PubMed] [Google Scholar]
- 57.Munn DH, Sharma MD, Hou D, Baban B, Lee JR, Antonia SJ, et al. Expression of indoleamine 2,3-dioxygenase by plasmacytoid dendritic cells in tumor-draining lymph nodes. J Clin Invest. 2004;114(2):280–90. 10.1172/JCI21583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Conrad C, Gregorio J, Wang YH, Ito T, Meller S, Hanabuchi S, et al. Plasmacytoid dendritic cells promote immunosuppression in ovarian cancer via ICOS costimulation of Foxp3(+) T-regulatory cells. Cancer Res. 2012;72(20):5240–9. 10.1158/0008-5472.CAN-12-2271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Le Mercier I, Poujol D, Sanlaville A, Sisirak V, Gobert M, Durand I, et al. Tumor promotion by intratumoral plasmacytoid dendritic cells is reversed by TLR7 ligand treatment. Cancer Res. 2013;73(15):4629–40. 10.1158/0008-5472.CAN-12-3058. [DOI] [PubMed] [Google Scholar]
- 60.Segura E, Touzot M, Bohineust A, Cappuccio A, Chiocchia G, Hosmalin A, et al. Human inflammatory dendritic cells induce Th17 cell differentiation. Immunity. 2013;38(2):336–48. 10.1016/j.immuni.2012.10.018. [DOI] [PubMed] [Google Scholar]
- 61.Hammad H, Plantinga M, Deswarte K, Pouliot P, Willart MA, Kool M, et al. Inflammatory dendritic cells–not basophils–are necessary and sufficient for induction of Th2 immunity to inhaled house dust mite allergen. J Exp Med. 2010;207(10):2097–111. 10.1084/jem.20101563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Verneau J, Sautés-Fridman C, Sun CM. Dendritic cells in the tumor microenvironment: prognostic and theranostic impact. Semin Immunol. 2020;48: 101410. 10.1016/j.smim.2020.101410. [DOI] [PubMed] [Google Scholar]
- 63.Marigo I, Zilio S, Desantis G, Mlecnik B, Agnellini AHR, Ugel S, et al. T cell cancer therapy requires CD40-CD40L activation of tumor necrosis factor and inducible nitric-oxide-synthase-producing dendritic cells. Cancer Cell. 2016;30(3):377–90. 10.1016/j.ccell.2016.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Xia L, Tian E, Yu M, Liu C, Shen L, Huang Y, et al. Rorγt agonist enhances anti-PD-1 therapy by promoting monocyte-derived dendritic cells through CXCL10 in cancers. J Exp Clin Cancer Res. 2022;41(1):155. 10.1186/s13046-022-02289-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Ma Y, Adjemian S, Mattarollo SR, Yamazaki T, Aymeric L, Yang H, et al. Anticancer chemotherapy-induced intratumoral recruitment and differentiation of antigen-presenting cells. Immunity. 2013;38(4):729–41. 10.1016/j.immuni.2013.03.003. [DOI] [PubMed] [Google Scholar]
- 66.Carreno BM, Magrini V, Becker-Hapak M, Kaabinejadian S, Hundal J, Petti AA, et al. Cancer immunotherapy. A dendritic cell vaccine increases the breadth and diversity of melanoma neoantigen-specific T cells. Sci. 2015;348(6236):803–8. 10.1126/science.aaa3828 [DOI] [PMC free article] [PubMed]
- 67.Mazzoccoli L, Liu B. Dendritic cells in shaping anti-tumor T cell response. Cancers (Basel). 2024;16(12):2211. 10.3390/cancers16122211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Guo Z, Yuan Y, Chen C, Lin J, Ma Q, Liu G, et al. Durable complete response to neoantigen-loaded dendritic-cell vaccine following anti-PD-1 therapy in metastatic gastric cancer. NPJ Precis Oncol. 2022;6(1):34. 10.1038/s41698-022-00279-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Briseño CG, Haldar M, Kretzer NM, Wu X, Theisen DJ, Kc W, et al. Distinct transcriptional programs control cross-priming in classical and monocyte-derived dendritic cells. Cell Rep. 2016;15(11):2462–74. 10.1016/j.celrep.2016.05.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Schetters STT, Rodriguez E, Kruijssen LJW, Crommentuijn MHW, Boon L, Van den Bossche J, et al. Monocyte-derived APCs are central to the response of PD1 checkpoint blockade and provide a therapeutic target for combination therapy. J Immunother Cancer. 2020;8(2): e000588. 10.1136/jitc-2020-000588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Kratchmarov R, Viragova S, Kim MJ, Rothman NJ, Liu K, Reizis B, et al. Metabolic control of cell fate bifurcations in a hematopoietic progenitor population. Immunol Cell Biol. 2018;96(8):863–71. 10.1111/imcb.12040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Du X, Wen J, Wang Y, Karmaus PWF, Khatamian A, Tan H, et al. Hippo/Mst signalling couples metabolic state and immune function of CD8α+ DCs. Nature. 2018;558(7708):141–5. 10.1038/s41586-018-0177-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Carroll KC, Viollet B, Suttles J. AMPKα1 deficiency amplifies proinflammatory myeloid APC activity and CD40 signaling. J Leukoc Biol. 2013;94(6):1113–21. 10.1189/jlb.0313157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Krawczyk CM, Holowka T, Sun J, Blagih J, Amiel E, DeBerardinis RJ, et al. Toll-like receptor-induced changes in glycolytic metabolism regulate dendritic cell activation. Blood. 2010;115(23):4742–9. 10.1182/blood-2009-10-249540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Pearce EJ, Everts B. Dendritic cell metabolism. Nat Rev Immunol. 2015;15(1):18–29. 10.1038/nri3771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Williams NC, O’Neill LAJ. A role for the Krebs cycle intermediate citrate in metabolic reprogramming in innate immunity and inflammation. Front Immunol. 2018;9: 141. 10.3389/fimmu.2018.00141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Everts B, Amiel E, Huang SC, Smith AM, Chang CH, Lam WY, et al. TLR-driven early glycolytic reprogramming via the kinases TBK1-IKKɛ supports the anabolic demands of dendritic cell activation. Nat Immunol. 2014;15(4):323–32. 10.1038/ni.2833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Everts B, Amiel E, van der Windt GJ, Freitas TC, Chott R, Yarasheski KE, et al. Commitment to glycolysis sustains survival of NO-producing inflammatory dendritic cells. Blood. 2012;120(7):1422–31. 10.1182/blood-2012-03-419747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Lawless SJ, Kedia-Mehta N, Walls JF, McGarrigle R, Convery O, Sinclair LV, et al. Glucose represses dendritic cell-induced T cell responses. Nat Commun. 2017;8:15620. 10.1038/ncomms15620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Sathaliyawala T, O’Gorman WE, Greter M, Bogunovic M, Konjufca V, Hou ZE, et al. Mammalian target of rapamycin controls dendritic cell development downstream of Flt3 ligand signaling. Immunity. 2010;33(4):597–606. 10.1016/j.immuni.2010.09.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Haidinger M, Poglitsch M, Geyeregger R, Kasturi S, Zeyda M, Zlabinger GJ, et al. A versatile role of mammalian target of rapamycin in human dendritic cell function and differentiation. J Immunol. 2010;185(7):3919–31. 10.4049/jimmunol.1000296. [DOI] [PubMed] [Google Scholar]
- 82.Snyder JP, Amiel E. Regulation of dendritic cell immune function and metabolism by cellular nutrient sensor mammalian target of rapamycin (mTOR). Front Immunol. 2019;9:3145. 10.3389/fimmu.2018.03145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Guak H, Al Habyan S, Ma EH, Aldossary H, Al-Masri M, Won SY, et al. Glycolytic metabolism is essential for CCR7 oligomerization and dendritic cell migration. Nat Commun. 2018;9(1):2463. 10.1038/s41467-018-04804-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Neefjes J, Jongsma ML, Paul P, Bakke O. Towards a systems understanding of MHC class I and MHC class II antigen presentation. Nat Rev Immunol. 2011;11(12):823–36. 10.1038/nri308. [DOI] [PubMed] [Google Scholar]
- 85.Thwe PM, Pelgrom LR, Cooper R, Beauchamp S, Reisz JA, D’Alessandro A, et al. Cell-intrinsic glycogen metabolism supports early glycolytic reprogramming required for DC immune responses. Cell Metab. 2017;26(3):558-67.e5. 10.1016/j.cmet.2017.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Maroof A, English NR, Bedford PA, Gabrilovich DI, Knight SC. Developing dendritic cells become “lacy” cells packed with fat and glycogen. Immunology. 2005;115(4):473–83. 10.1111/j.1365-2567.2005.02181.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Shi L, Chen X, Zang A, Li T, Hu Y, Ma S, et al. TSC1/mTOR-controlled metabolic-epigenetic cross talk underpins DC control of CD8+ T-cell homeostasis. PLoS Biol. 2019;17(8): e3000420. 10.1371/journal.pbio.3000420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Amiel E, Everts B, Freitas TC, King IL, Curtis JD, Pearce EL, et al. Inhibition of mechanistic target of rapamycin promotes dendritic cell activation and enhances therapeutic autologous vaccination in mice. J Immunol. 2012;189(5):2151–8. 10.4049/jimmunol.1103741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Amiel E, Everts B, Fritz D, Beauchamp S, Ge B, Pearce EL, et al. Mechanistic target of rapamycin inhibition extends cellular lifespan in dendritic cells by preserving mitochondrial function. J Immunol. 2014;193(6):2821–30. 10.4049/jimmunol.1302498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Saravia J, Raynor JL, Chapman NM, Lim SA, Chi H. Signaling networks in immunometabolism. Cell Res. 2020;30(4):328–42. 10.1038/s41422-020-0301-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Garcia D, Shaw RJ. AMPK: Mechanisms of cellular energy sensing and restoration of metabolic balance. Mol Cell. 2017;66(6):789–800. 10.1016/j.molcel.2017.05.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Malinarich F, Duan K, Hamid RA, Bijin A, Lin WX, Poidinger M, et al. High mitochondrial respiration and glycolytic capacity represent a metabolic phenotype of human tolerogenic dendritic cells. J Immunol. 2015;194(11):5174–86. 10.4049/jimmunol.1303316. [DOI] [PubMed] [Google Scholar]
- 93.Novitskiy SV, Ryzhov S, Zaynagetdinov R, Goldstein AE, Huang Y, Tikhomirov OY, et al. Adenosine receptors in regulation of dendritic cell differentiation and function. Blood. 2008;112(5):1822–31. 10.1182/blood-2008-02-136325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Yang M, Ma C, Liu S, Shao Q, Gao W, Song B, et al. HIF-dependent induction of adenosine receptor A2b skews human dendritic cells to a Th2-stimulating phenotype under hypoxia. Immunol Cell Biol. 2010;88(2):165–71. 10.1038/icb.2009.77. [DOI] [PubMed] [Google Scholar]
- 95.Ben Addi A, Lefort A, Hua X, Libert F, Communi D, Ledent C, et al. Modulation of murine dendritic cell function by adenine nucleotides and adenosine: involvement of the A(2B) receptor. Eur J Immunol. 2008;38(6):1610–20. 10.1002/eji.200737781. [DOI] [PubMed] [Google Scholar]
- 96.Challier J, Bruniquel D, Sewell AK, Laugel B. Adenosine and cAMP signalling skew human dendritic cell differentiation towards a tolerogenic phenotype with defective CD8(+) T-cell priming capacity. Immunology. 2013;138(4):402–10. 10.1111/imm.12053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Panther E, Corinti S, Idzko M, Herouy Y, Napp M, la Sala A, et al. Adenosine affects expression of membrane molecules, cytokine and chemokine release, and the T-cell stimulatory capacity of human dendritic cells. Blood. 2003;101(10):3985–90. 10.1182/blood-2002-07-2113. [DOI] [PubMed] [Google Scholar]
- 98.Aymeric L, Apetoh L, Ghiringhelli F, Tesniere A, Martins I, Kroemer G, et al. Tumor cell death and ATP release prime dendritic cells and efficient anticancer immunity. Cancer Res. 2010;70(3):855–8. 10.1158/0008-5472.CAN-09-3566. [DOI] [PubMed] [Google Scholar]
- 99.Silva-Vilches C, Ring S, Mahnke K. ATP and its metabolite adenosine as regulators of dendritic cell activity. Front Immunol. 2018;9:2581. 10.3389/fimmu.2018.02581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Wu L, Xie W, Li Y, Ni Q, Timashev P, Lyu M, et al. Biomimetic nanocarriers guide extracellular ATP homeostasis to remodel energy metabolism for activating innate and adaptive immunity system. Adv Sci. 2022;9(17): 2105376. 10.1002/advs.202105376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Bougnères L, Helft J, Tiwari S, Vargas P, Chang BH, Chan L, et al. A role for lipid bodies in the cross-presentation of phagocytosed antigens by MHC class I in dendritic cells. Immunity. 2009;31(2):232–44. 10.1016/j.immuni.2009.06.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Xu C, Ji X, Zhou Y, Cheng Y, Guo D, Li Q, et al. Slimming and Reinvigorating Tumor-Associated Dendritic Cells with Hierarchical Lipid Rewiring Nanoparticles. Adv Mater. 2023;35(30): e2211415. 10.1002/adma.202211415. [DOI] [PubMed] [Google Scholar]
- 103.Zhao F, Xiao C, Evans KS, Theivanthiran T, DeVito N, Holtzhausen A, et al. Paracrine Wnt5a-β-Catenin Signaling Triggers a Metabolic Program that Drives Dendritic Cell Tolerization. Immunity. 2018;48(1):147-60.e7. 10.1016/j.immuni.2017.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Cubillos-Ruiz JR, Silberman PC, Rutkowski MR, Chopra S, Perales-Puchalt A, Song M, et al. ER stress sensor XBP1 controls anti-tumor immunity by disrupting dendritic cell homeostasis. Cell. 2015;161(7):1527–38. 10.1016/j.cell.2015.05.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.V eglia F, Tyurin VA, Mohammadyani D, Blasi M, Duperret EK, Donthireddy L, et al. Lipid bodies containing oxidatively truncated lipids block antigen cross-presentation by dendritic cells in cancer. Nat Commun. 2017;8(1): 2122. 10.1038/s41467-017-02186-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Lee AH, Scapa EF, Cohen DE, Glimcher LH. Regulation of hepatic lipogenesis by the transcription factor XBP1. Science. 2008;320(5882):1492–6. 10.1126/science.1158042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Mellor AL, Munn DH. IDO expression by dendritic cells: tolerance and tryptophan catabolism. Nat Rev Immunol. 2004;4(10):762–74. 10.1038/nri1457. [DOI] [PubMed] [Google Scholar]
- 108.Wang F, Liu M, Ma D, Cai Z, Liu L, Wang J, et al. Dendritic cell-expressed IDO alleviates atherosclerosis by expanding CD4+CD25+Foxp3+Tregs through IDO-Kyn-AHR axis. Int Immunopharmacol. 2023;116: 109758. 10.1016/j.intimp.2023.109758. [DOI] [PubMed] [Google Scholar]
- 109.Li Q, Harden JL, Anderson CD, Egilmez NK. Tolerogenic phenotype of IFN-γ-induced IDO+ dendritic cells is maintained via an autocrine IDO-kynurenine/AhR-IDO loop. J Immunol. 2016;197(3):962–70. 10.4049/jimmunol.1502615. [DOI] [PubMed] [Google Scholar]
- 110.Belladonna ML, Orabona C, Grohmann U, Puccetti P. TGF-beta and kynurenines as the key to infectious tolerance. Trends Mol Med. 2009;15(2):41–9. 10.1016/j.molmed.2008.11.006. [DOI] [PubMed] [Google Scholar]
- 111.Grohmann U, Orabona C, Fallarino F, Vacca C, Calcinaro F, Falorni A, et al. CTLA-4-Ig regulates tryptophan catabolism in vivo. Nat Immunol. 2002;3(11):1097–101. 10.1038/ni846. [DOI] [PubMed] [Google Scholar]
- 112.Mori M, Gotoh T. Arginine metabolic enzymes, nitric oxide and infection. J Nutr. 2004;134(10 Suppl):2820S-S2853. 10.1093/jn/134.10.2820S. [DOI] [PubMed] [Google Scholar]
- 113.Cartwright ANR, Suo S, Badrinath S, Kumar S, Melms J, Luoma A, et al. Immunosuppressive Myeloid Cells Induce Nitric Oxide-Dependent DNA Damage and p53 Pathway Activation in CD8+ T Cells. Cancer Immunol Res. 2021;9(4):470–85. 10.1158/2326-6066.CIR-20-0085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Nicolas A, Cathelin D, Larmonier N, Fraszczak J, Puig PE, Bouchot A, et al. Dendritic cells trigger tumor cell death by a nitric oxide-dependent mechanism. J Immunol. 2007;179(2):812–8. 10.4049/jimmunol.179.2.812. [DOI] [PubMed] [Google Scholar]
- 115.Mondanelli G, Bianchi R, Pallotta MT, Orabona C, Albini E, Iacono A, et al. A relay pathway between arginine and tryptophan metabolism confers immunosuppressive properties on dendritic cells. Immunity. 2017;46(2):233–44. 10.1016/j.immuni.2017.01.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Ghahramanipour Z, Alipour S, Masoumi J, Rostamlou A, Hatami-Sadr A, Heris JA, et al. Regulation of Dendritic Cell Functions by Vitamins as Promising Therapeutic Strategy for Immune System Disorders. Adv Biol. 2023;7(12): e2300142. 10.1002/adbi.202300142. [DOI] [PubMed] [Google Scholar]
- 117.Vanherwegen AS, Eelen G, Ferreira GB, Ghesquière B, Cook DP, Nikolic T, et al. Vitamin D controls the capacity of human dendritic cells to induce functional regulatory T cells by regulation of glucose metabolism. J Steroid Biochem Mol Biol. 2019;187:134–45. 10.1016/j.jsbmb.2018.11.011. [DOI] [PubMed] [Google Scholar]
- 118.Saul L, Mair I, Ivens A, Brown P, Samuel K, Campbell JDM, et al. 1,25-dihydroxyvitamin D3 restrains CD4+ T cell priming ability of CD11c+ dendritic cells by upregulating expression of CD31. Front Immunol. 2019;10:600. 10.3389/fimmu.2019.00600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Rivera CA, Randrian V, Richer W, Gerber-Ferder Y, Delgado MG, Chikina AS, et al. Epithelial colonization by gut dendritic cells promotes their functional diversification. Immunity. 2022;55(1):129-44.e8. 10.1016/j.immuni.2021.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Deka A, Kumar N, Basu S, Chawla M, Bhattacharya N, Ali SA, et al. Non-canonical NF-κb signaling limits the tolerogenic β-catenin-Raldh2 axis in gut dendritic cells to exacerbate intestinal pathologies. EMBO J. 2024;43(18):3895–915. 10.1038/s44318-024-00182-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Yuan X, Duan Y, Xiao Y, Sun K, Qi Y, Zhang Y, et al. Vitamin E enhances cancer immunotherapy by reinvigorating dendritic cells via targeting checkpoint SHP1. Cancer Discov. 2022;12(7):1742–59. 10.1158/2159-8290.CD-21-0900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Chen IC, Awasthi D, Hsu CL, Song M, Chae CS, Dannenberg AJ, et al. High-fat diet-induced obesity alters dendritic cell homeostasis by enhancing mitochondrial fatty acid oxidation. J Immunol. 2022;209(1):69–76. 10.4049/jimmunol.2100567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Lu Y, Shi Y, Liu Y, Luo Z, Zhang J, Jiang M, et al. A therapeutic DC vaccine with maintained immunological activity exhibits robust anti-tumor efficacy. J Control Release. 2022;349:254–68. 10.1016/j.jconrel.2022.06.059. [DOI] [PubMed] [Google Scholar]
- 124.Chakrabarty RP, Chandel NS. Mitochondria as signaling organelles control mammalian stem cell fate. Cell Stem Cell. 2021;28(3):394–408. 10.1016/j.stem.2021.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Tan SK, Hougen HY, Merchan JR, Gonzalgo ML, Welford SM. Fatty acid metabolism reprogramming in ccRCC: mechanisms and potential targets. Nat Rev Urol. 2023;20(1):48–60. 10.1038/s41585-022-00654-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Menzies KJ, Zhang H, Katsyuba E, Auwerx J. Protein acetylation in metabolism - metabolites and cofactors. Nat Rev Endocrinol. 2016;12(1):43–60. 10.1038/nrendo.2015.181. [DOI] [PubMed] [Google Scholar]
- 127.Shen Y, Wei W, Zhou DX. Histone acetylation enzymes coordinate metabolism and gene expression. Trends Plant Sci. 2015;20(10):614–21. 10.1016/j.tplants.2015.07.005. [DOI] [PubMed] [Google Scholar]
- 128.Izzo LT, Trefely S, Demetriadou C, Drummond JM, Mizukami T, Kuprasertkul N, et al. Acetylcarnitine shuttling links mitochondrial metabolism to histone acetylation and lipogenesis. Sci Adv. 2023;9(18): eadf0115. 10.1126/sciadv.adf0115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.St Paul M, Saibil SD, Han S, Israni-Winger K, Lien SC, Laister RC, et al. Coenzyme A fuels T cell anti-tumor immunity. Cell Metab. 2021;33(12):2415-27.e6. 10.1016/j.cmet.2021.11.010. [DOI] [PubMed] [Google Scholar]
- 130.Greenspan MD, Germershausen JI, Mackow R. Effect of halofenate and clofibrate on lipid synthesis in rat adipocytes. Biochim Biophys Acta. 1975;380(2):190–8. 10.1016/0005-2760(75)90005-3. [DOI] [PubMed] [Google Scholar]
- 131.Liu X, Cooper DE, Cluntun AA, Warmoes MO, Zhao S, Reid MA, et al. Acetate Production from Glucose and Coupling to Mitochondrial Metabolism in Mammals. Cell. 2018;175(2):502-13.e13. 10.1016/j.cell.2018.08.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Vysochan A, Sengupta A, Weljie AM, Alwine JC, Yu Y. ACSS2-mediated acetyl-CoA synthesis from acetate is necessary for human cytomegalovirus infection. Proc Natl Acad Sci U S A. 2017;114(8):E1528–35. 10.1073/pnas.1614268114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Sivanand S, Viney I, Wellen KE. Spatiotemporal control of Acetyl-CoA metabolism in chromatin regulation. Trends Biochem Sci. 2018;43(1):61–74. 10.1016/j.tibs.2017.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Adeva-Andany MM, López-Maside L, Donapetry-García C, Fernández-Fernández C, Sixto-Leal C. Enzymes involved in branched-chain amino acid metabolism in humans. Amino Acids. 2017;49(6):1005–28. 10.1007/s00726-017-2412-7. [DOI] [PubMed] [Google Scholar]
- 135.Carrer A, Trefely S, Zhao S, Campbell SL, Norgard RJ, Schultz KC, et al. Acetyl-CoA metabolism supports multistep pancreatic tumorigenesis. Cancer Discov. 2019;9(3):416–35. 10.1158/2159-8290.CD-18-0567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Li JT, Yin M, Wang D, Wang J, Lei MZ, Zhang Y, et al. BCAT2-mediated BCAA catabolism is critical for development of pancreatic ductal adenocarcinoma. Nat Cell Biol. 2020;22(2):167–74. 10.1038/s41556-019-0455-6. [DOI] [PubMed] [Google Scholar]
- 137.Mosaoa R, Kasprzyk-Pawelec A, Fernandez HR, Avantaggiati ML. The mitochondrial citrate carrier SLC25A1/CIC and the fundamental role of citrate in cancer, inflammation and beyond. Biomolecules. 2021;11(2):141. 10.3390/biom11020141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Roosterman D, Cottrell GS. Rethinking the citric acid cycle: connecting pyruvate carboxylase and citrate synthase to the flow of energy and material. Int J Mol Sci. 2021;22(2): 604. 10.3390/ijms22020604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Zhao Q, Zhou X, Curbo S, Karlsson A. Metformin downregulates the mitochondrial carrier SLC25A10 in a glucose dependent manner. Biochem Pharmacol. 2018;156:444–50. 10.1016/j.bcp.2018.09.015. [DOI] [PubMed] [Google Scholar]
- 140.Granchi C. ATP citrate lyase (ACLY) inhibitors: an anti-cancer strategy at the crossroads of glucose and lipid metabolism. Eur J Med Chem. 2018;157:1276–91. 10.1016/j.ejmech.2018.09.001. [DOI] [PubMed] [Google Scholar]
- 141.Yiew NKH, Finck BN. The mitochondrial pyruvate carrier at the crossroads of intermediary metabolism. Am J Physiol Endocrinol Metab. 2022;323(1):E33-52. 10.1152/ajpendo.00074.2022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Mews P, Donahue G, Drake AM, Luczak V, Abel T, Berger SL. Acetyl-CoA synthetase regulates histone acetylation and hippocampal memory. Nature. 2017;546(7658):381–6. 10.1038/nature22405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Bulusu V, Tumanov S, Michalopoulou E, van den Broek NJ, MacKay G, Nixon C, et al. Acetate Recapturing by Nuclear Acetyl-CoA Synthetase 2 Prevents Loss of Histone Acetylation during Oxygen and Serum Limitation. Cell Rep. 2017;18(3):647–58. 10.1016/j.celrep.2016.12.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Huang Z, Zhang M, Plec AA, Estill SJ, Cai L, Repa JJ, et al. ACSS2 promotes systemic fat storage and utilization through selective regulation of genes involved in lipid metabolism. Proc Natl Acad Sci U S A. 2018;115(40):E9499-506. 10.1073/pnas.1806635115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Shi L, Tu BP. Acetyl-CoA and the regulation of metabolism: mechanisms and consequences. Curr Opin Cell Biol. 2015;33:125–31. 10.1016/j.ceb.2015.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Campbell SL, Wellen KE. Metabolic signaling to the nucleus in cancer. Mol Cell. 2018;71(3):398–408. 10.1016/j.molcel.2018.07.015. [DOI] [PubMed] [Google Scholar]
- 147.Cai L, Sutter BM, Li B, Tu BP. Acetyl-CoA induces cell growth and proliferation by promoting the acetylation of histones at growth genes. Mol Cell. 2011;42(4):426–37. 10.1016/j.molcel.2011.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Bradshaw PC. Acetyl-CoA metabolism and histone acetylation in the regulation of aging and lifespan. Antioxidants. 2021;10(4): 572. 10.3390/antiox10040572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Koundouros N, Poulogiannis G. Reprogramming of fatty acid metabolism in cancer. Br J Cancer. 2020;122(1):4–22. 10.1038/s41416-019-0650-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Pasquadibisceglie A, Quadrotta V, Polticelli F. In silico analysis of the structural dynamics and substrate recognition determinants of the human mitochondrial carnitine/acylcarnitine SLC25A20 transporter. Int J Mol Sci. 2023;24(4): 3946. 10.3390/ijms24043946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Kienesberger K, Pordes AG, Völk TG, Hofbauer R. L-carnitine and PPARα-agonist fenofibrate are involved in the regulation of carnitine acetyltransferase (CrAT) mRNA levels in murine liver cells. BMC Genomics. 2014;15(1):514. 10.1186/1471-2164-15-514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Lingwood D, Simons K. Lipid rafts as a membrane-organizing principle. Science. 2010;327(5961):46–50. 10.1126/science.1174621. [DOI] [PubMed] [Google Scholar]
- 153.Leuenberger T, Pfueller CF, Luessi F, Bendix I, Paterka M, Prozorovski T, et al. Modulation of DC immunobiology via inhibition of 3-hydroxy-3-methylglutaryl-CoA (HMG-CoA) reductase. PLoS ONE. 2014;9(7): e100871. 10.1371/journal.pone.0100871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Kofler S, Schlichting C, Jankl S, Nickel T, Weis M. Dual mode of HMG-CoA reductase inhibition on DC invasion. Atherosclerosis. 2008;197(1):105–10. 10.1016/j.atherosclerosis.2007.08.005. [DOI] [PubMed] [Google Scholar]
- 155.Martin-Perez M, Urdiroz-Urricelqui U, Bigas C, Benitah SA. The role of lipids in cancer progression and metastasis. Cell Metab. 2022;34(11):1675–99. 10.1016/j.cmet.2022.09.023. [DOI] [PubMed] [Google Scholar]
- 156.Hochrein SM, Wu H, Eckstein M, Arrigoni L, Herman JS, Schumacher F, et al. The glucose transporter GLUT3 controls T helper 17 cell responses through glycolytic-epigenetic reprogramming. Cell Metab. 2022;34(4):516-32.e11. 10.1016/j.cmet.2022.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Peng M, Yin N, Chhangawala S, Xu K, Leslie CS, Li MO. Aerobic glycolysis promotes T helper 1 cell differentiation through an epigenetic mechanism. Science. 2016;354(6311):481–4. 10.1126/science.aaf6284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Lauterbach MA, Hanke JE, Serefidou M, Mangan MSJ, Kolbe CC, Hess T, et al. Toll-like receptor signaling rewires macrophage metabolism and promotes histone acetylation via ATP-citrate lyase. Immunity. 2019;51(6):997-1011.e7. 10.1016/j.immuni.2019.11.009. [DOI] [PubMed] [Google Scholar]
- 159.Yin K, Lee J, Liu Z, Kim H, Martin DR, Wu D, et al. Mitophagy protein PINK1 suppresses colon tumor growth by metabolic reprogramming via p53 activation and reducing acetyl-CoA production. Cell Death Differ. 2021;28(8):2421–35. 10.1038/s41418-021-00760-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Wang T, Yao W, Li J, He Q, Shao Y, Huang F. Acetyl-CoA from inflammation-induced fatty acids oxidation promotes hepatic malate-aspartate shuttle activity and glycolysis. Am J Physiol Endocrinol Metab. 2018;315(4):E496-510. 10.1152/ajpendo.00061.2018. [DOI] [PubMed] [Google Scholar]
- 161.Liu TM, Shyh-Chang N. SIRT2 and glycolytic enzyme acetylation in pluripotent stem cells. Nat Cell Biol. 2017;19(5):412–4. 10.1038/ncb3522. [DOI] [PubMed] [Google Scholar]
- 162.Martínez-Reyes I, Chandel NS. Mitochondrial TCA cycle metabolites control physiology and disease. Nat Commun. 2020;11(1):102. 10.1038/s41467-019-13668-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Jouandin P, Marelja Z, Shih YH, Parkhitko AA, Dambowsky M, Asara JM, et al. Lysosomal cystine mobilization shapes the response of TORC1 and tissue growth to fasting. Science. 2022;375(6582): eabc4203. 10.1126/science.abc4203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Chen L, Duan Y, Wei H, Ning H, Bi C, Zhao Y, et al. Acetyl-CoA carboxylase (ACC) as a therapeutic target for metabolic syndrome and recent developments in ACC1/2 inhibitors. Expert Opin Investig Drugs. 2019;28(10):917–30. 10.1080/13543784.2019.1657825. [DOI] [PubMed] [Google Scholar]
- 165.Arnedo M, Latorre-Pellicer A, Lucia-Campos C, Gil-Salvador M, Antoñanzas-Peréz R, Gómez-Puertas P, et al. More than one HMG-CoA lyase: the classical mitochondrial enzyme plus the peroxisomal and the cytosolic ones. Int J Mol Sci. 2019;20(24): 6124. 10.3390/ijms20246124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Lerin C, Rodgers JT, Kalume DE, Kim SH, Pandey A, Puigserver P. GCN5 acetyltransferase complex controls glucose metabolism through transcriptional repression of PGC-1alpha. Cell Metab. 2006;3(6):429–38. 10.1016/j.cmet.2006.04.013. [DOI] [PubMed] [Google Scholar]
- 167.Oksenych V, Su D, Daniel JA. Acetyltransferases GCN5 and PCAF are required for B lymphocyte maturation in mice. Biomolecules. 2021;12(1): 61. 10.3390/biom12010061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Sukhbaatar N, Hengstschläger M, Weichhart T. mTOR-mediated regulation of DC differentiation and function. Trends Immunol. 2016;37(11):778–89. 10.1016/j.it.2016.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Saxton RA, Sabatini DM. mTOR signaling in growth, metabolism, and disease. Cell. 2017;169(2):361–71. 10.1016/j.cell.2017.03.035. [DOI] [PubMed] [Google Scholar]
- 170.Galluzzi L, Pietrocola F, Bravo-San Pedro JM, Amaravadi RK, Baehrecke EH, Cecconi F, et al. Autophagy in malignant transformation and cancer progression. EMBO J. 2015;34(7):856–80. 10.15252/embj.201490784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Wang Y, Huang G, Zeng H, Yang K, Lamb RF, Chi H. Tuberous sclerosis 1 (Tsc1)-dependent metabolic checkpoint controls development of DCs. Proc Natl Acad Sci U S A. 2013;110(50):E4894–903. 10.1073/pnas.1308905110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Luo Y, Liu J, Sun X, Feng T, Fang L, Chen S, et al. Tsc1-dependent transcriptional programming of DC homeostasis and function. Exp Cell Res. 2018;363(1):73–83. 10.1016/j.yexcr.2017.12.028. [DOI] [PubMed] [Google Scholar]
- 173.Luo Y, Li W, Yu G, Yu J, Han L, Xue T, et al. Tsc1 expression by DCs is required to preserve T-cell homeostasis and response. Cell Death Dis. 2017;8(1): e2553. 10.1038/cddis.2016.487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.He A, Chen X, Tan M, Chen Y, Lu D, Zhang X, et al. Acetyl-CoA Derived from Hepatic Peroxisomal β-Oxidation Inhibits Autophagy and Promotes Steatosis via mTORC1 Activation. Mol Cell. 2020;79(1):30-42.e4. 10.1016/j.molcel.2020.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Minarrieta L, Godoy GJ, Velazquez LN, Ghorbani P, Sparwasser T, Berod L. Regulation of DC metabolism by nitric oxide in murine GM-CSF cultures. Eur J Immunol. 2023;53(2): e2149691. 10.1002/eji.202149691. [DOI] [PubMed] [Google Scholar]
- 176.Russo M, Gualdrini F, Vallelonga V, Prosperini E, Noberini R, Pedretti S, et al. Acetyl-CoA production by Mediator-bound 2-ketoacid dehydrogenases boosts de novo histone acetylation and is regulated by nitric oxide. Mol Cell. 2024;84(5):967-80.e10. 10.1016/j.molcel.2023.12.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Thwe PM, Amiel E. The role of nitric oxide in metabolic regulation of DC immune function. Cancer Lett. 2018;412:236–42. 10.1016/j.canlet.2017.10.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Leone RD, Powell JD. Metabolism of immune cells in cancer. Nat Rev Cancer. 2020;20(9):516–31. 10.1038/s41568-020-0273-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.McBrian MA, Behbahan IS, Ferrari R, Su T, Huang TW, Li K, et al. Histone acetylation regulates intracellular pH. Mol Cell. 2013;49(2):310–21. 10.1016/j.molcel.2012.10.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Mills E, O’Neill LA. Succinate: a metabolic signal in inflammation. Trends Cell Biol. 2014;24(5):313–20. 10.1016/j.tcb.2013.11.008. [DOI] [PubMed] [Google Scholar]
- 181.Rubic T, Lametschwandtner G, Jost S, Hinteregger S, Kund J, Carballido-Perrig N, et al. Triggering the succinate receptor GPR91 on DCs enhances immunity. Nat Immunol. 2008;9(11):1261–9. 10.1038/ni.1657. [DOI] [PubMed] [Google Scholar]
- 182.O’Neill LA, Pearce EJ. Immunometabolism governs DC and macrophage function. J Exp Med. 2016;213(1):15–23. 10.1084/jem.20151570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Weatherill AR, Lee JY, Zhao L, Lemay DG, Youn HS, Hwang DH. Saturated and polyunsaturated fatty acids reciprocally modulate DC functions mediated through TLR4. J Immunol. 2005;174(9):5390–7. 10.4049/jimmunol.174.9.5390. [DOI] [PubMed] [Google Scholar]
- 184.Mogilenko DA, Haas JT, L'homme L, Fleury S, Quemener S, Levavasseur M, et al. Metabolic and Innate Immune Cues Merge into a Specific Inflammatory Response via the UPR. Cell. 2019;177(5):1201–16.e19. 10.1016/j.cell.2019.03.018 [DOI] [PubMed]
- 185.Wertheimer T, Zwicky P, Rindlisbacher L, Sparano C, Vermeer M, de Melo BMS, et al. IL-23 stabilizes an effector Treg cell program in the tumor microenvironment. Nat Immunol. 2024;25(3):512–24. 10.1038/s41590-024-01755-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Ohnmacht C, Pullner A, King SB, Drexler I, Meier S, Brocker T, et al. Constitutive ablation of dendritic cells breaks self-tolerance of CD4 T cells and results in spontaneous fatal autoimmunity. J Exp Med. 2009;206(3):549–59. 10.1084/jem.20082394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Joffre OP, Segura E, Savina A, Amigorena S. Cross-presentation by dendritic cells. Nat Rev Immunol. 2012;12(8):557–69. 10.1038/nri3254. [DOI] [PubMed] [Google Scholar]
- 188.Coutant F, Miossec P. Altered dendritic cell functions in autoimmune diseases: distinct and overlapping profiles. Nat Rev Rheumatol. 2016;12(12):703–15. 10.1038/nrrheum.2016.147. [DOI] [PubMed] [Google Scholar]
- 189.Chan VS, Nie YJ, Shen N, Yan S, Mok MY, Lau CS. Distinct roles of myeloid and plasmacytoid dendritic cells in systemic lupus erythematosus. Autoimmun Rev. 2012;11(12):890–7. 10.1016/j.autrev.2012.03.004. [DOI] [PubMed] [Google Scholar]
- 190.Jongbloed SL, Lebre MC, Fraser AR, Gracie JA, Sturrock RD, Tak PP, et al. Enumeration and phenotypical analysis of distinct dendritic cell subsets in psoriatic arthritis and rheumatoid arthritis. Arthritis Res Ther. 2006;8(1):R15. 10.1186/ar1864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Meitei HT, Jadhav N, Lal G. CCR6-CCL20 axis as a therapeutic target for autoimmune diseases. Autoimmun Rev. 2021;20(7): 102846. 10.1016/j.autrev.2021.102846. [DOI] [PubMed] [Google Scholar]
- 192.Page G, Miossec P. Paired synovium and lymph nodes from rheumatoid arthritis patients differ in dendritic cell and chemokine expression. J Pathol. 2004;204(1):28–38. 10.1002/path.1607. [DOI] [PubMed] [Google Scholar]
- 193.Farkas L, Beiske K, Lund-Johansen F, Brandtzaeg P, Jahnsen FL. Plasmacytoid dendritic cells (natural interferon- alpha/beta-producing cells) accumulate in cutaneous lupus erythematosus lesions. Am J Pathol. 2001;159(1):237–43. 10.1016/s0002-9440(10)61689-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Fiore N, Castellano G, Blasi A, Capobianco C, Loverre A, Montinaro V, et al. Immature myeloid and plasmacytoid dendritic cells infiltrate renal tubulointerstitium in patients with lupus nephritis. Mol Immunol. 2008;45(1):259–65. 10.1016/j.molimm.2007.04.029. [DOI] [PubMed] [Google Scholar]
- 195.Katou F, Ohtani H, Nakayama T, Nagura H, Yoshie O, Motegi K. Differential expression of CCL19 by DC-Lamp+ mature dendritic cells in human lymph node versus chronically inflamed skin. J Pathol. 2003;199(1):98–106. 10.1002/path.1255. [DOI] [PubMed] [Google Scholar]
- 196.Brandum EP, Jørgensen AS, Rosenkilde MM, Hjortø GM. Dendritic cells and CCR7 expression: an important factor for autoimmune diseases, chronic inflammation, and cancer. Int J Mol Sci. 2021;22(15): 8340. 10.3390/ijms22158340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Hot A, Miossec P. Effects of interleukin (IL)-17A and IL-17F in human rheumatoid arthritis synoviocytes. Ann Rheum Dis. 2011;70(5):727–32. 10.1136/ard.2010.143768. [DOI] [PubMed] [Google Scholar]
- 198.Benderdour M, Tardif G, Pelletier JP, Di Battista JA, Reboul P, Ranger P, et al. Interleukin 17 (IL-17) induces collagenase-3 production in human osteoarthritic chondrocytes via AP-1 dependent activation: differential activation of AP-1 members by IL-17 and IL-1beta. J Rheumatol. 2002;29(6):1262–72. 10.3899/jrheum.130032. [PubMed] [Google Scholar]
- 199.Pacquelet S, Presle N, Boileau C, Dumond H, Netter P, Martel-Pelletier J, et al. Interleukin 17, a nitric oxide-producing cytokine with a peroxynitrite-independent inhibitory effect on proteoglycan synthesis. J Rheumatol. 2002;29(12):2602–10. 10.3899/jrheum.130033. [PubMed] [Google Scholar]
- 200.Canavan M, Marzaioli V, McGarry T, Bhargava V, Nagpal S, Veale DJ, et al. Rheumatoid arthritis synovial microenvironment induces metabolic and functional adaptations in dendritic cells. Clin Exp Immunol. 2020;202(2):226–38. 10.1111/cei.13479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Suwa Y, Nagafuchi Y, Yamada S, Fujio K. The role of dendritic cells and their immunometabolism in rheumatoid arthritis. Front Immunol. 2023;14:1161148. 10.3389/fimmu.2023.1161148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Litinskiy MB, Nardelli B, Hilbert DM, He B, Schaffer A, Casali P, et al. DCs induce CD40-independent immunoglobulin class switching through BLyS and APRIL. Nat Immunol. 2002;3(9):822–9. 10.1038/ni829. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 203.Quintero-González DC, Muñoz-Urbano M, Vásquez G. Mitochondria as a key player in systemic lupus erythematosus. Autoimmunity. 2022;55(8):497–505. 10.1080/08916934.2022.2112181. [DOI] [PubMed] [Google Scholar]
- 204.Hammad H, de Heer HJ, Soullié T, Angeli V, Trottein F, Hoogsteden HC, et al. Activation of peroxisome proliferator-activated receptor-gamma in dendritic cells inhibits the development of eosinophilic airway inflammation in a mouse model of asthma. Am J Pathol. 2004;164(1):263–71. 10.1016/s0002-9440(10)63116-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Gilles S, Mariani V, Bryce M, Mueller MJ, Ring J, Jakob T, et al. Pollen-derived E1-phytoprostanes signal via PPAR-gamma and NF-kappaB-dependent mechanisms. J Immunol. 2009;182(11):6653–8. 10.4049/jimmunol.0802613. [DOI] [PubMed] [Google Scholar]
- 206.Rodriguez-Coira J, Villaseñor A, Izquierdo E, Huang M, Barker-Tejeda TC, Radzikowska U, et al. The importance of metabolism for immune homeostasis in allergic diseases. Front Immunol. 2021;12: 692004. 10.3389/fimmu.2021.692004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Vroman H, Hendriks RW, Kool M. Dendritic cell subsets in asthma: impaired tolerance or exaggerated inflammation? Front Immunol. 2017;8:941. 10.3389/fimmu.2017.00941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Furlong-Silva J, Cook PC. Fungal-mediated lung allergic airway disease: the critical role of macrophages and dendritic cells. PLoS Pathog. 2022;18(7): e1010608. 10.1371/journal.ppat.1010608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Miake S, Tsuji G, Takemura M, Hashimoto-Hachiya A, Vu YH, Furue M, et al. IL-4 augments IL-31/IL-31 receptor alpha interaction leading to enhanced Ccl 17 and Ccl 22 production in dendritic cells: implications for atopic dermatitis. Int J Mol Sci. 2019;20(16): 4053. 10.3390/ijms20164053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Liu P, Kang C, Zhang J, Liu Y, Liu J, Hu T, et al. The role of dendritic cells in allergic diseases. Int Immunopharmacol. 2022;113(Pt B): 109449. 10.1016/j.intimp.2022.109449. [DOI] [PubMed] [Google Scholar]
- 211.Zhang Y, Luo Y, Li W, Liu J, Chen M, Gu H, et al. DC-SIGN promotes allergen uptake and activation of dendritic cells in patients with atopic dermatitis. J Dermatol Sci. 2016;84(2):128–36. 10.1016/j.jdermsci.2016.08.008. [DOI] [PubMed] [Google Scholar]
- 212.Tokura Y, Hayano S. Subtypes of atopic dermatitis: from phenotype to endotype. Allergol Int. 2022;71(1):14–24. 10.1016/j.alit.2021.07.003. [DOI] [PubMed] [Google Scholar]
- 213.Nakano R, Tran LM, Geller DA, Macedo C, Metes DM, Thomson AW. Dendritic cell-mediated regulation of liver ischemia-reperfusion injury and liver transplant rejection. Front Immunol. 2021;12: 705465. 10.3389/fimmu.2021.705465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Du X, Li M, Huan C, Lv G. Dendritic cells in liver transplantation immune response. Front Cell Dev Biol. 2023;11:1277743. 10.3389/fcell.2023.1277743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.LaRosa DF, Rahman AH, Turka LA. The innate immune system in allograft rejection and tolerance. J Immunol. 2007;178(12):7503–9. 10.4049/jimmunol.178.12.7503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Khanna A, Morelli AE, Zhong C, Takayama T, Lu L, Thomson AW. Effects of liver-derived dendritic cell progenitors on Th1- and Th2-like cytokine responses in vitro and in vivo. J Immunol. 2000;164(3):1346–54. 10.4049/jimmunol.164.3.1346. [DOI] [PubMed] [Google Scholar]
- 217.Lin J, Wang H, Liu C, Cheng A, Deng Q, Zhu H, et al. Dendritic cells: versatile players in renal transplantation. Front Immunol. 2021;12: 654540. 10.3389/fimmu.2021.654540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Dai H, Thomson AW, Rogers NM. Dendritic cells as sensors, mediators, and regulators of ischemic injury. Front Immunol. 2019;10:2418. 10.3389/fimmu.2019.02418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Zhuang Q, Lakkis FG. Dendritic cells and innate immunity in kidney transplantation. Kidney Int. 2015;87(4):712–8. 10.1038/ki.2014.430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Kurts C, Robinson BW, Knolle PA. Cross-priming in health and disease. Nat Rev Immunol. 2010;10(6):403–14. 10.1038/nri2780. [DOI] [PubMed] [Google Scholar]
- 221.Neuper T, Frauenlob T, Dang HH, Krenn PW, Posselt G, Regl C, et al. ADP-heptose attenuates Helicobacter pylori-induced dendritic cell activation. Gut Microbes. 2024;16(1): 2402543. 10.1080/19490976.2024.2402543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Oster P, Vaillant L, Riva E, McMillan B, Begka C, Truntzer C, et al. Helicobacter pylori infection has a detrimental impact on the efficacy of cancer immunotherapies. Gut. 2022;71(3):457–66. 10.1136/gutjnl-2020-323392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Guo Y, Chen J, Huang Y, Ke S, Xie F, Li D, et al. Increased infiltration of CD4+ IL-17A+ FOXP3+ T cells in Helicobacter pylori-induced gastritis. Eur J Immunol. 2024;54(3): e2350662. 10.1002/eji.202350662. [DOI] [PubMed] [Google Scholar]
- 224.Hedlund S, Persson A, Vujic A, Che KF, Stendahl O, Larsson M. Dendritic cell activation by sensing Mycobacterium tuberculosis-induced apoptotic neutrophils via DC-SIGN. Hum Immunol. 2010;71(6):535–40. 10.1016/j.humimm.2010.02.022. [DOI] [PubMed] [Google Scholar]
- 225.Giacomini E, Iona E, Ferroni L, Miettinen M, Fattorini L, Orefici G, et al. Infection of human macrophages and dendritic cells with Mycobacterium tuberculosis induces a differential cytokine gene expression that modulates T cell response. J Immunol. 2001;166(12):7033–41. 10.4049/jimmunol.166.12.7033. [DOI] [PubMed] [Google Scholar]
- 226.Reiley WW, Calayag MD, Wittmer ST, Huntington JL, Pearl JE, Fountain JJ, et al. ESAT-6-specific CD4 T cell responses to aerosol Mycobacterium tuberculosis infection are initiated in the mediastinal lymph nodes. Proc Natl Acad Sci U S A. 2008;105(31):10961–6. 10.1073/pnas.0801496105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Uehira K, Amakawa R, Ito T, Tajima K, Naitoh S, Ozaki Y, et al. Dendritic cells are decreased in blood and accumulated in granuloma in tuberculosis. Clin Immunol. 2002;105(3):296–303. 10.1006/clim.2002.5287. [DOI] [PubMed] [Google Scholar]
- 228.Wolf AJ, Linas B, Trevejo-Nuñez GJ, Kincaid E, Tamura T, Takatsu K, et al. Mycobacterium tuberculosis infects dendritic cells with high frequency and impairs their function in vivo. J Immunol. 2007;179(4):2509–19. 10.4049/jimmunol.179.4.2509. [DOI] [PubMed] [Google Scholar]
- 229.Søndergaard JN, Laursen JM, Rosholm LB, Brix S. Mycobacterium tuberculosis promotes Th17 expansion via regulation of human dendritic cells toward a high CD14 and low IL-12p70 phenotype that reprograms upon exogenous IFN-γ. Int Immunol. 2014;26(12):705–16. 10.1093/intimm/dxu085. [DOI] [PubMed] [Google Scholar]
- 230.Gagliardi MC, Lemassu A, Teloni R, Mariotti S, Sargentini V, Pardini M, et al. Cell wall-associated alpha-glucan is instrumental for Mycobacterium tuberculosis to block CD1 molecule expression and disable the function of dendritic cell derived from infected monocyte. Cell Microbiol. 2007;9(8):2081–92. 10.1111/j.1462-5822.2007.00940.x. [DOI] [PubMed] [Google Scholar]
- 231.Gupta V, Jaiswal A, Behera D, Prasad HK. Disparity in circulating peripheral blood dendritic cell subsets and cytokine profile of pulmonary tuberculosis patients compared with healthy family contacts. Hum Immunol. 2010;71(7):682–91. 10.1016/j.humimm.2010.03.010. [DOI] [PubMed] [Google Scholar]
- 232.Remoli ME, Giacomini E, Lutfalla G, Dondi E, Orefici G, Battistini A, et al. Selective expression of type I IFN genes in human dendritic cells infected with Mycobacterium tuberculosis. J Immunol. 2002;169(1):366–74. 10.4049/jimmunol.169.1.366. [DOI] [PubMed] [Google Scholar]
- 233.Kool M, van Nimwegen M, Willart MA, Muskens F, Boon L, Smit JJ, et al. An anti-inflammatory role for plasmacytoid dendritic cells in allergic airway inflammation. J Immunol. 2009;183(2):1074–82. 10.4049/jimmunol.0900471. [DOI] [PubMed] [Google Scholar]
- 234.Zhang JA, Lu YB, Wang WD, Liu GB, Chen C, Shen L, et al. BTLA-expressing dendritic cells in patients with tuberculosis exhibit reduced production of IL-12/IFN-α and increased production of IL-4 and TGF-β, favoring Th2 and Foxp3+ treg polarization. Front Immunol. 2020;11:518. 10.3389/fimmu.2020.00518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Manches O, Frleta D, Bhardwaj N. Dendritic cells in progression and pathology of HIV infection. Trends Immunol. 2014;35(3):114–22. 10.1016/j.it.2013.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Wilen CB, Tilton JC, Doms RW. HIV: cell binding and entry. Cold Spring Harb Perspect Med. 2012;2(8): a006866. 10.1101/cshperspect.a006866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Martín-Moreno A, Muñoz-Fernández MA. Dendritic cells, the double agent in the war against HIV-1. Front Immunol. 2019;10:2485. 10.3389/fimmu.2019.02485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Mulherkar TH, Gómez DJ, Sandel G, Jain P. Co-infection and cancer: host-pathogen interaction between dendritic cells and HIV-1, HTLV-1, and other oncogenic viruses. Viruses. 2022;14(9):2037. 10.3390/v14092037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Kim JT, Chang E, Sigal A, Baltimore D. Dendritic cells efficiently transmit HIV to T cells in a tenofovir and raltegravir insensitive manner. PLoS One. 2018;13(1): e0189945. 10.1371/journal.pone.0189945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Botting RA, Rana H, Bertram KM, Rhodes JW, Baharlou H, Nasr N, et al. Langerhans cells and sexual transmission of HIV and HSV. Rev Med Virol. 2017;27(2): e1923. 10.1002/rmv.1923. [DOI] [PubMed] [Google Scholar]
- 241.van Montfort T, Eggink D, Boot M, Tuen M, Hioe CE, Berkhout B, et al. HIV-1 N-glycan composition governs a balance between dendritic cell-mediated viral transmission and antigen presentation. J Immunol. 2011;187(9):4676–85. 10.4049/jimmunol.1101876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 242.Jan M, Upadhyay C, Hioe CE. HIV-1 envelope glycan composition as a key determinant of efficient virus transmission via DC-SIGN and resistance to inhibitory lectins. iScience. 2019;21:413–27. 10.1016/j.isci.2019.10.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Martinelli E, Cicala C, Van Ryk D, Goode DJ, Macleod K, Arthos J, et al. HIV-1 gp120 inhibits TLR9-mediated activation and IFN-α secretion in plasmacytoid dendritic cells. Proc Natl Acad Sci U S A. 2007;104(9):3396–401. 10.1073/pnas.0611353104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Hertoghs N, van der Aar AM, Setiawan LC, Kootstra NA, Gringhuis SI, Geijtenbeek TB. SAMHD1 degradation enhances active suppression of dendritic cell maturation by HIV-1. J Immunol. 2015;194(9):4431–7. 10.4049/jimmunol.1403016. [DOI] [PubMed] [Google Scholar]
- 245.Sepulveda-Crespo D, Resino S, Martinez I. Innate immune response against hepatitis C virus: targets for vaccine adjuvants. Vaccines. 2020;8(2): 313. 10.3390/vaccines8020313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Rana D, Chawla YK, Duseja A, Dhiman R, Arora SK. Functional reconstitution of defective myeloid dendritic cells in chronic hepatitis C infection on successful antiviral treatment. Liver Int. 2012;32(7):1128–37. 10.1111/j.1478-3231.2011.02754.x. [DOI] [PubMed] [Google Scholar]
- 247.Della Bella S, Crosignani A, Riva A, Presicce P, Benetti A, Longhi R, et al. Decrease and dysfunction of dendritic cells correlate with impaired hepatitis C virus-specific CD4+ T-cell proliferation in patients with hepatitis C virus infection. Immunology. 2007;121(2):283–92. 10.1111/j.1365-2567.2007.02577.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 248.Tomer S, Chawla YK, Duseja A, Arora SK. Dominating expression of negative regulatory factors downmodulates major histocompatibility complex class-II expression on dendritic cells in chronic hepatitis C infection. World J Gastroenterol. 2016;22(22):5173–82. 10.3748/wjg.v22.i22.5173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 249.Krishnadas DK, Ahn JS, Han J, Kumar R, Agrawal B. Immunomodulation by hepatitis C virus-derived proteins: targeting human dendritic cells by multiple mechanisms. Int Immunol. 2010;22(6):491–502. 10.1093/intimm/dxq033. [DOI] [PubMed] [Google Scholar]
- 250.Jafarlou M. Unveiling the menace: a thorough review of potential pandemic fungal disease. Front Fungal Biol. 2024;5:1338726. 10.3389/ffunb.2024.1338726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Buentke E, Scheynius A. Dendritic cells and fungi. APMIS. 2003;111(7–8):789–96. 10.1034/j.1600-0463.2003.11107810.x. [DOI] [PubMed] [Google Scholar]
- 252.Tuor M, Stappers MHT, Desgardin A, Ruchti F, Sparber F, Orr SJ, et al. Card9 and MyD88 differentially regulate Th17 immunity to the commensal yeast Malassezia in the murine skin. Mucosal Immunol. 2025;18(1):205–19. 10.1016/j.mucimm.2024.11.004. [DOI] [PubMed] [Google Scholar]
- 253.Ito A, Sugita K, Goto H, Yamamoto O. Implication of perifollicular clusters and folliculotropic distribution of dendritic cells in the pathogenesis of seborrhoeic dermatitis. Acta Derm Venereol. 2020;100(13): adv00183. 10.2340/00015555-3519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254.Li L, Wang S, Zou Z, Tao A, Ai Y. Activation profile of THP-1 derived dendritic cells stimulated by allergen Mal f 1 beyond its IgE-binding ability. Int Immunopharmacol. 2018;62:139–46. 10.1016/j.intimp.2018.05.013. [DOI] [PubMed] [Google Scholar]
- 255.Batliner M, Schumacher F, Wigger D, Vivas W, Prell A, Fohmann I, et al. The Candida albicans quorum-sensing molecule farnesol alters sphingolipid metabolism in human monocyte-derived dendritic cells. mBio. 2024;15(8):e0073224. 10.1128/mbio.00732-24 [DOI] [PMC free article] [PubMed]
- 256.Motran CC, Ambrosio LF, Volpini X, Celias DP, Cervi L. Dendritic cells and parasites: from recognition and activation to immune response instruction. Semin Immunopathol. 2017;39(2):199–213. 10.1007/s00281-016-0588-7. [DOI] [PubMed] [Google Scholar]
- 257.de Veer MJ, Curtis JM, Baldwin TM, DiDonato JA, Sexton A, McConville MJ, et al. MyD88 is essential for clearance of Leishmania major: possible role for lipophosphoglycan and Toll-like receptor 2 signaling. Eur J Immunol. 2003;33(10):2822–31. 10.1002/eji.200324128. [DOI] [PubMed] [Google Scholar]
- 258.Whitaker SM, Colmenares M, Pestana KG, McMahon-Pratt D. Leishmania pifanoi proteoglycolipid complex P8 induces macrophage cytokine production through Toll-like receptor 4. Infect Immun. 2008;76(5):2149–56. 10.1128/IAI.01528-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259.Boggiatto PM, Jie F, Ghosh M, Gibson-Corley KN, Ramer-Tait AE, Jones DE, et al. Altered dendritic cell phenotype in response to Leishmania amazonensis amastigote infection is mediated by MAP kinase, ERK. Am J Pathol. 2009;174(5):1818–26. 10.2353/ajpath.2009.080905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 260.Carow B, Rottenberg ME. SOCS3, a major regulator of infection and inflammation. Front Immunol. 2014;5: 58. 10.3389/fimmu.2014.00058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 261.Valério-Bolas A, Meunier M, Palma-Marques J, Rodrigues A, Santos AM, Nunes T, et al. Exploiting Leishmania-primed dendritic cells as potential immunomodulators of canine immune response. Cells. 2024;13(5):445. 10.3390/cells13050445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 262.Valério-Bolas A, Meunier M, Rodrigues A, Palma-Marques J, Ferreira R, Cardoso I, et al. Unveiling the Interplay Between Dendritic Cells and Natural Killer Cells as Key Players in Leishmania Infection. J Immunol Res. 2025;2025:3176927. 10.1155/jimr/3176927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 263.Thomas PG, Carter MR, Atochina O, Da’Dara AA, Piskorska D, McGuire E, et al. Maturation of dendritic cell 2 phenotype by a helminth glycan uses a toll-like receptor 4-dependent mechanism. J Immunol. 2003;171(11):5837–41. 10.4049/jimmunol.171.11.5837. [DOI] [PubMed] [Google Scholar]
- 264.Everts B, Smits HH, Hokke CH, Yazdanbakhsh M. Helminths and dendritic cells: sensing and regulating via pattern recognition receptors, Th2 and Treg responses. Eur J Immunol. 2010;40(6):1525–37. 10.1002/eji.200940109. [DOI] [PubMed] [Google Scholar]
- 265.Acevedo N, Lozano A, Zakzuk J, Llinás-Caballero K, Brodin D, Nejsum P, et al. Cystatin from the helminth Ascaris lumbricoides upregulates mevalonate and cholesterol biosynthesis pathways and immunomodulatory genes in human monocyte-derived dendritic cells. Front Immunol. 2024;15:1328401. 10.3389/fimmu.2024.1328401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 266.Manurung MD, Sonnet F, Hoogerwerf MA, Janse JJ, Kruize Y, Bes-Roeleveld L, et al. Controlled human hookworm infection remodels plasmacytoid dendritic cells and regulatory T cells towards profiles seen in natural infections in endemic areas. Nat Commun. 2024;15(1): 5960. 10.1038/s41467-024-50313-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 267.Mitsialis V, Wall S, Liu P, Ordovas-Montanes J, Parmet T, Vukovic M, et al. Single-cell analyses of colon and blood reveal distinct immune cell signatures of ulcerative colitis and Crohn’s disease. Gastroenterology. 2020;159(2):591-608.e10. 10.1053/j.gastro.2020.04.074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 268.Baumgart DC, Metzke D, Guckelberger O, Pascher A, Grötzinger C, Przesdzing I, et al. Aberrant plasmacytoid dendritic cell distribution and function in patients with Crohn’s disease and ulcerative colitis. Clin Exp Immunol. 2011;166(1):46–54. 10.1111/j.1365-2249.2011.04439.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 269.Vulliemoz M, Brand S, Juillerat P, Mottet C, Ben-Horin S, Michetti P. TNF-alpha blockers in inflammatory bowel diseases: practical recommendations and a user’s guide: an update. Digestion. 2020;101(Suppl 1):16–26. 10.1159/000506898. [DOI] [PubMed] [Google Scholar]
- 270.Ninnemann J, Winsauer C, Bondareva M, Kühl AA, Lozza L, Durek P, et al. TNF hampers intestinal tissue repair in colitis by restricting IL-22 bioavailability. Mucosal Immunol. 2022;15(4):698–716. 10.1038/s41385-022-00506-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 271.Faleiro R, Liu J, Karunarathne D, Edmundson A, Winterford C, Nguyen TH, et al. Crohn’s disease is facilitated by a disturbance of programmed death-1 ligand 2 on blood dendritic cells. Clin Transl Immunol. 2019;8(7): e01071. 10.1002/cti2.1071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 272.Jiménez-Cortegana C, Palomares F, Alba G, Santa-María C, de la Cruz-Merino L, Sánchez-Margalet V, et al. Dendritic cells: the yin and yang in disease progression. Front Immunol. 2024;14:1321051. 10.3389/fimmu.2023.1321051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 273.Ma DW, Ha J, Yoon KS, Kang I, Choi TG, Kim SS. Innate immune system in the pathogenesis of non-alcoholic fatty liver disease. Nutrients. 2023;15(9):2068. 10.3390/nu15092068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 274.Deczkowska A, David E, Ramadori P, Pfister D, Safran M, Li B, et al. XCR1+ type 1 conventional dendritic cells drive liver pathology in non-alcoholic steatohepatitis. Nat Med. 2021;27(6):1043–54. 10.1038/s41591-021-01344-3. [DOI] [PubMed] [Google Scholar]
- 275.Schuster S, Cabrera D, Arrese M, Feldstein AE. Triggering and resolution of inflammation in NASH. Nat Rev Gastroenterol Hepatol. 2018;15(6):349–64. 10.1038/s41575-018-0009-6. [DOI] [PubMed] [Google Scholar]
- 276.Nati M, Haddad D, Birkenfeld AL, Koch CA, Chavakis T, Chatzigeorgiou A. The role of immune cells in metabolism-related liver inflammation and development of non-alcoholic steatohepatitis (NASH). Rev Endocr Metab Disord. 2016;17(1):29–39. 10.1007/s11154-016-9339-2. [DOI] [PubMed] [Google Scholar]
- 277.Henning JR, Graffeo CS, Rehman A, Fallon NC, Zambirinis CP, Ochi A, et al. Dendritic cells limit fibroinflammatory injury in nonalcoholic steatohepatitis in mice. Hepatology. 2013;58(2):589–602. 10.1002/hep.26267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 278.Bernsmeier C, Albano E. Liver dendritic cells and NAFLD evolution: a remaining open issue. J Hepatol. 2017;66(6):1120–2. 10.1016/j.jhep.2017.02.018. [DOI] [PubMed] [Google Scholar]
- 279.Cheng H, Chen W, Lin Y, Zhang J, Song X, Zhang D. Signaling pathways involved in the biological functions of dendritic cells and their implications for disease treatment. Mol Biomed. 2023;4(1):15. 10.1186/s43556-023-00125-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280.Greene TT, Jo Y, Chiale C, Macal M, Fang Z, Khatri FS, et al. Metabolic deficiencies underlie reduced plasmacytoid dendritic cell IFN-I production following viral infection. Nat Commun. 2025;16(1):1460. 10.1038/s41467-025-56603-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 281.Srivastava M, Bencurova E, Gupta SK, Weiss E, Löffler J, Dandekar T. Aspergillus fumigatus challenged by human dendritic cells: metabolic and regulatory pathway responses testify a tight battle. Front Cell Infect Microbiol. 2019;9:168. 10.3389/fcimb.2019.00168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 282.Chen JY, Zhou JK, Pan W. Immunometabolism: towards a better understanding the mechanism of parasitic infection and immunity. Front Immunol. 2021;12: 661241. 10.3389/fimmu.2021.661241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 283.Li L, Yang L, Wang L, Wang F, Zhang Z, Li J, et al. Impaired T cell function in malignant pleural effusion is caused by TGF-β derived predominantly from macrophages. Int J Cancer. 2016;139(10):2261–9. 10.1002/ijc.30289. [DOI] [PubMed] [Google Scholar]
- 284.Scarlett UK, Rutkowski MR, Rauwerdink AM, Fields J, Escovar-Fadul X, Baird J, et al. Ovarian cancer progression is controlled by phenotypic changes in dendritic cells. J Exp Med. 2012;209(3):495–506. 10.1084/jem.20111413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 285.Zhang X, He T, Li Y, Chen L, Liu H, Wu Y, et al. Dendritic cell vaccines in ovarian cancer. Front Immunol. 2021;11: 613773. 10.3389/fimmu.2020.613773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 286.Munn DH, Mellor AL. Ido in the tumor microenvironment: inflammation, counter-regulation, and tolerance. Trends Immunol. 2016;37(3):193–207. 10.1016/j.it.2016.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 287.Zhang H, Chen J, Bai J, Zhang J, Huang S, Zeng L, et al. Single dual-specific anti-PD-L1/TGF-β antibody synergizes with chemotherapy as neoadjuvant treatment for pancreatic ductal adenocarcinoma: a preclinical experimental study. Int J Surg. 2024;110(5):2679–91. 10.1097/JS9.0000000000001226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 288.Thepmalee C, Panya A, Sujjitjoon J, Sawasdee N, Poungvarin N, Junking M, et al. Suppression of TGF-β and IL-10 receptors on self-differentiated dendritic cells by short-hairpin RNAs enhanced activation of effector T-cells against cholangiocarcinoma cells. Hum Vaccin Immunother. 2020;16(10):2318–27. 10.1080/21645515.2019.1701913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 289.Zhou Z, Zhou Q, Zhao J, Hou X, Yan J, Sun X, et al. Rebalancing TGF-β/PGE2 breaks RT-induced immunosuppressive barriers by enhancing tumor-infiltrated dendritic cell homing. Int J Biol Sci. 2024;20(1):367–86. 10.7150/ijbs.87867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 290.Trempolec N, Degavre C, Doix B, Brusa D, Corbet C, Feron O. Acidosis-induced TGF-β2 production promotes lipid droplet formation in dendritic cells and alters their potential to support anti-mesothelioma T cell response. Cancers (Basel). 2020;12(5):1284. 10.3390/cancers12051284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 291.Xiong W, He W, Wang T, He S, Xu F, Wang Z, et al. Smad4 Deficiency Promotes Pancreatic Cancer Immunogenicity by Activating the Cancer-Autonomous DNA-Sensing Signaling Axis. Adv Sci. 2022;9(7): e2103029. 10.1002/advs.202103029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 292.Liefers-Visser JAL, Meijering RAM, Reyners AKL, van der Zee AGJ, de Jong S. IGF system targeted therapy: Therapeutic opportunities for ovarian cancer. Cancer Treat Rev. 2017;60:90–9. 10.1016/j.ctrv.2017.08.012. [DOI] [PubMed] [Google Scholar]
- 293.Huang CT, Chang MC, Chen YL, Chen TC, Chen CA, Cheng WF. Insulin-like growth factors inhibit dendritic cell-mediated anti-tumor immunity through regulating ERK1/2 phosphorylation and p38 dephosphorylation. Cancer Lett. 2015;359(1):117–26. 10.1016/j.canlet.2015.01.007. [DOI] [PubMed] [Google Scholar]
- 294.Zhang L, Liu W, Wang X, Wang X, Sun H. Prognostic value of serum IL-8 and IL-10 in patients with ovarian cancer undergoing chemotherapy. Oncol Lett. 2019;17(2):2365–9. 10.3892/ol.2018.9842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 295.Li L, Yu R, Cai T, Chen Z, Lan M, Zou T, et al. Effects of immune cells and cytokines on inflammation and immunosuppression in the tumor microenvironment. Int Immunopharmacol. 2020;88: 106939. 10.1016/j.intimp.2020.106939. [DOI] [PubMed] [Google Scholar]
- 296.Liu CZ, Zhang L, Chang XH, Cheng YX, Cheng HY, Ye X, et al. Overexpression and immunosuppressive functions of transforming growth factor 1, vascular endothelial growth factor and interleukin-10 in epithelial ovarian cancer. Chin J Cancer Res. 2012;24(2):130–7. 10.1007/s11670-012-0130-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 297.Llopiz D, Ruiz M, Silva L, Sarobe P. Enhancement of antitumor vaccination by targeting dendritic cell-related IL-10. Front Immunol. 2018;9:1923. 10.3389/fimmu.2018.01923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 298.Thepmalee C, Panya A, Junking M, Chieochansin T, Yenchitsomanus PT. Inhibition of IL-10 and TGF-β receptors on dendritic cells enhances activation of effector T-cells to kill cholangiocarcinoma cells. Hum Vaccin Immunother. 2018;14(6):1423–31. 10.1080/21645515.2018.1431598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 299.Tan Z, Liu L, Chiu MS, Cheung KW, Yan CW, Yu Z, et al. Virotherapy-recruited PMN-MDSC infiltration of mesothelioma blocks antitumor CTL by IL-10-mediated dendritic cell suppression. Oncoimmunology. 2018;8(1): e1518672. 10.1080/2162402X.2018.1518672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 300.Boks MA, Kager-Groenland JR, Haasjes MS, Zwaginga JJ, van Ham SM, ten Brinke A. IL-10-generated tolerogenic dendritic cells are optimal for functional regulatory T cell induction–a comparative study of human clinical-applicable DC. Clin Immunol. 2012;142(3):332–42. 10.1016/j.clim.2011.11.011. [DOI] [PubMed] [Google Scholar]
- 301.Moore KW, de Waal Malefyt R, Coffman RL, O’Garra A. Interleukin-10 and the interleukin-10 receptor. Annu Rev Immunol. 2001;19:683–765. 10.1146/annurev.immunol.19.1.683. [DOI] [PubMed] [Google Scholar]
- 302.Batchu RB, Gruzdyn OV, Kolli BK, Dachepalli R, Umar PS, Rai SK, et al. IL-10 signaling in the tumor microenvironment of ovarian cancer. Adv Exp Med Biol. 2021;1290:51–65. 10.1007/978-3-030-55617-4_3. [DOI] [PubMed] [Google Scholar]
- 303.Lee WS, Yang H, Chon HJ, Kim C. Combination of anti-angiogenic therapy and immune checkpoint blockade normalizes vascular-immune crosstalk to potentiate cancer immunity. Exp Mol Med. 2020;52(9):1475–85. 10.1038/s12276-020-00500-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 304.Gabrilovich DI, Chen HL, Girgis KR, Cunningham HT, Meny GM, Nadaf S, et al. Production of vascular endothelial growth factor by human tumors inhibits the functional maturation of dendritic cells. Nat Med. 1996;2(10):1096–103. 10.1038/nm1096-1096. [DOI] [PubMed] [Google Scholar]
- 305.Mimura K, Kono K, Takahashi A, Kawaguchi Y, Fujii H. Vascular endothelial growth factor inhibits the function of human mature dendritic cells mediated by VEGF receptor-2. Cancer Immunol Immunother. 2007;56(6):761–70. 10.1007/s00262-006-0234-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 306.Dikov MM, Ohm JE, Ray N, Tchekneva EE, Burlison J, Moghanaki D, et al. Differential roles of vascular endothelial growth factor receptors 1 and 2 in dendritic cell differentiation. J Immunol. 2005;174(1):215–22. 10.4049/jimmunol.174.1.215. [DOI] [PubMed] [Google Scholar]
- 307.Oyama T, Ran S, Ishida T, Nadaf S, Kerr L, Carbone DP, et al. Vascular endothelial growth factor affects dendritic cell maturation through the inhibition of nuclear factor-kappa B activation in hemopoietic progenitor cells. J Immunol. 1998;160(3):1224–32. 10.4049/jimmunol.160.3.1224. [PubMed] [Google Scholar]
- 308.Gabrilovich DI, Ishida T, Nadaf S, Ohm JE, Carbone DP. Antibodies to vascular endothelial growth factor enhance the efficacy of cancer immunotherapy by improving endogenous dendritic cell function. Clin Cancer Res. 1999;5(10):2963–70. 10.1158/1078-0432.CCR-05-2963. [PubMed] [Google Scholar]
- 309.Osada T, Chong G, Tansik R, Hong T, Spector N, Kumar R, et al. The effect of anti-VEGF therapy on immature myeloid cell and dendritic cells in cancer patients. Cancer Immunol Immunother. 2008;57(8):1115–24. 10.1007/s00262-007-0441-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 310.Gagliostro V, Seeger P, Garrafa E, Salvi V, Bresciani R, Bosisio D, et al. Pro-lymphangiogenic properties of IFN-γ-activated human dendritic cells. Immunol Lett. 2016;173:26–35. 10.1016/j.imlet.2016.03.008. [DOI] [PubMed] [Google Scholar]
- 311.Olariu R, Tsai C, Abd El Hafez M, Milusev A, Banz Y, Lese I, et al. Presence of Donor Lymph Nodes Within Vascularized Composite Allotransplantation Ameliorates VEGF-C-mediated Lymphangiogenesis and Delays the Onset of Acute Rejection. Transplant. 2021;105(8):1747–59. 10.1097/TP.0000000000003601 [DOI] [PubMed]
- 312.Li J, Li XL, Li CQ. Immunoregulation mechanism of VEGF signaling pathway inhibitors and its efficacy on the kidney. Am J Med Sci. 2023;366(6):404–12. 10.1016/j.amjms.2023.09.005. [DOI] [PubMed] [Google Scholar]
- 313.Zhou C, Ma L, Xu H, Huo Y, Luo J. Meningeal lymphatics regulate radiotherapy efficacy through modulating anti-tumor immunity. Cell Res. 2022;32(6):543–54. 10.1038/s41422-022-00639-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 314.Mohamed E, Al-Khami AA, Rodriguez PC. The cellular metabolic landscape in the tumor milieu regulates the activity of myeloid infiltrates. Cell Mol Immunol. 2018;15(5):421–7. 10.1038/s41423-018-0001-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 315.van Vlerken-Ysla L, Tyurina YY, Kagan VE, Gabrilovich DI. Functional states of myeloid cells in cancer. Cancer Cell. 2023;41(3):490–504. 10.1016/j.ccell.2023.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 316.Korbecki J, Bobiński R, Dutka M. Self-regulation of the inflammatory response by peroxisome proliferator-activated receptors. Inflamm Res. 2019;68(6):443–58. 10.1007/s00011-019-01231-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 317.Kim R, Hashimoto A, Markosyan N, Tyurin VA, Tyurina YY, Kar G, et al. Ferroptosis of tumour neutrophils causes immune suppression in cancer. Nature. 2022;612(7939):338–46. 10.1038/s41586-022-05443-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 318.Kagan VE, Mao G, Qu F, Angeli JP, Doll S, Croix CS, et al. Oxidized arachidonic and adrenic PEs navigate cells to ferroptosis. Nat Chem Biol. 2017;13(1):81–90. 10.1038/nchembio.2238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 319.Ugolini A, Tyurin VA, Tyurina YY, Tcyganov EN, Donthireddy L, Kagan VE, et al. Polymorphonuclear myeloid-derived suppressor cells limit antigen cross-presentation by dendritic cells in cancer. JCI Insight. 2020;5(15): e138581. 10.1172/jci.insight.138581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 320.Suthen S, Lim CJ, Nguyen PHD, Dutertre CA, Lai HLH, Wasser M, et al. Hypoxia-driven immunosuppression by Treg and type-2 conventional dendritic cells in HCC. Hepatology. 2022;76(5):1329–44. 10.1002/hep.3241. [DOI] [PubMed] [Google Scholar]
- 321.Paardekooper LM, Vos W, van den Bogaart G. Oxygen in the tumor microenvironment: effects on dendritic cell function. Oncotarget. 2019;10(8):883–96. 10.18632/oncotarget.26608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 322.Kotsias F, Hoffmann E, Amigorena S, Savina A. Reactive oxygen species production in the phagosome: impact on antigen presentation in dendritic cells. Antioxid Redox Signal. 2013;18(6):714–29. 10.1089/ars.2012.4557. [DOI] [PubMed] [Google Scholar]
- 323.Savina A, Jancic C, Hugues S, Guermonprez P, Vargas P, Moura IC, et al. NOX2 controls phagosomal pH to regulate antigen processing during crosspresentation by dendritic cells. Cell. 2006;126(1):205–18. 10.1016/j.cell.2006.05.035. [DOI] [PubMed] [Google Scholar]
- 324.Elia AR, Cappello P, Puppo M, Fraone T, Vanni C, Eva A, et al. Human dendritic cells differentiated in hypoxia down-modulate antigen uptake and change their chemokine expression profile. J Leukoc Biol. 2008;84(6):1472–82. 10.1189/jlb.0208082. [DOI] [PubMed] [Google Scholar]
- 325.Zhao W, Darmanin S, Fu Q, Chen J, Cui H, Wang J, et al. Hypoxia suppresses the production of matrix metalloproteinases and the migration of human monocyte-derived dendritic cells. Eur J Immunol. 2005;35(12):3468–77. 10.1002/eji.200526262. [DOI] [PubMed] [Google Scholar]
- 326.Kheshtchin N, Arab S, Ajami M, Mirzaei R, Ashourpour M, Mousavi N, et al. Inhibition of HIF-1α enhances anti-tumor effects of dendritic cell-based vaccination in a mouse model of breast cancer. Cancer Immunol Immunother. 2016;65(10):1159–67. 10.1007/s00262-016-1879-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 327.Hammami A, Abidin BM, Heinonen KM, Stäger S. HIF-1α hampers dendritic cell function and Th1 generation during chronic visceral leishmaniasis. Sci Rep. 2018;8(1):3500. 10.1038/s41598-018-21891-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 328.Jantsch J, Chakravortty D, Turza N, Prechtel AT, Buchholz B, Gerlach RG, et al. Hypoxia and hypoxia-inducible factor-1 alpha modulate lipopolysaccharide-induced dendritic cell activation and function. J Immunol. 2008;180(7):4697–705. 10.4049/jimmunol.180.7.4697. [DOI] [PubMed] [Google Scholar]
- 329.Chen S, Liao C, Hu H, Liao J, Chen Z, Li S, et al. Hypoxia-driven tumor stromal remodeling and immunosuppressive microenvironment in scirrhous HCC. Hepatology. 2024;79(4):780–97. 10.1097/HEP.0000000000000599. [DOI] [PubMed] [Google Scholar]
- 330.Guo T, Xu J. Cancer-associated fibroblasts: a versatile mediator in tumor progression, metastasis, and targeted therapy. Cancer Metastasis Rev. 2024;43(3):1095–116. 10.1007/s10555-024-10186-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 331.Hsieh CC, Hung CH, Chiang M, Tsai YC, He JT. Hepatic stellate cells enhance liver cancer progression by inducing myeloid-derived suppressor cells through interleukin-6 signaling. Int J Mol Sci. 2019;20(20): 5079. 10.3390/ijms20205079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 332.Ezhilarasan D. Hepatic stellate cells in the injured liver: perspectives beyond hepatic fibrosis. J Cell Physiol. 2022;237(1):436–49. 10.1002/jcp.30582. [DOI] [PubMed] [Google Scholar]
- 333.Gong Z, Li Q, Shi J, Wei J, Li P, Chang CH, et al. Lung fibroblasts facilitate pre-metastatic niche formation by remodeling the local immune microenvironment. Immunity. 2022;55(8):1483-500.e9. 10.1016/j.immuni.2022.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 334.Herzog BH, Baer JM, Borcherding N, Kingston NL, Belle JI, Knolhoff BL, et al. Tumor-associated fibrosis impairs immune surveillance and response to immune checkpoint blockade in non-small cell lung cancer. Sci Transl Med. 2023;15(699): eadh8005. 10.1126/scitranslmed.adh8005. [DOI] [PubMed] [Google Scholar]
- 335.De Sanctis F, Ugel S, Facciponte J, Facciabene A. The dark side of tumor-associated endothelial cells. Semin Immunol. 2018;35:35–47. 10.1016/j.smim.2018.02.002. [DOI] [PubMed] [Google Scholar]
- 336.Nagl L, Horvath L, Pircher A, Wolf D. Tumor endothelial cells (TECs) as potential immune directors of the tumor microenvironment - new findings and future perspectives. Front Cell Dev Biol. 2020;8:766. 10.3389/fcell.2020.00766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 337.Zhao Q, Han YM, Song P, Liu Z, Yuan Z, Zou MH. Endothelial cell-specific expression of serine/threonine kinase 11 modulates dendritic cell differentiation. Nat Commun. 2022;13(1):648. 10.1038/s41467-022-28316-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 338.Lu J, Zhao J, Liu K, Zhao J, Yang H, Huang Y, et al. MAPK/ERK1/2 signaling mediates endothelial-like differentiation of immature DCs in the microenvironment of esophageal squamous cell carcinoma. Cell Mol Life Sci. 2010;67(12):2091–106. 10.1007/s00018-010-0316-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 339.Leone P, Di Lernia G, Solimando AG, Cicco S, Saltarella I, Lamanuzzi A, et al. Bone marrow endothelial cells sustain a tumor-specific CD8+ T cell subset with suppressive function in myeloma patients. Oncoimmunology. 2018;8(1): e1486949. 10.1080/2162402X.2018.1486949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 340.Huang Y, Mao Q, He J, Su J, Peng Y, Liang W, et al. Fusions of tumor-derived endothelial cells with dendritic cells induces antitumor immunity. Sci Rep. 2017;7: 46544. 10.1038/srep46544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 341.Hou Y, Wei D, Zhang Z, Guo H, Li S, Zhang J, et al. FABP5 controls macrophage alternative activation and allergic asthma by selectively programming long-chain unsaturated fatty acid metabolism. Cell Rep. 2022;41(7): 111668. 10.1016/j.celrep.2022.111668. [DOI] [PubMed] [Google Scholar]
- 342.Gao F, Liu C, Guo J, Sun W, Xian L, Bai D, et al. Radiation-driven lipid accumulation and DC dysfunction in cancer. Sci Rep. 2015;5:9613. 10.1038/srep09613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 343.Ramakrishnan R, Tyurin VA, Veglia F, Condamine T, Amoscato A, Mohammadyani D, et al. Oxidized lipids block antigen cross-presentation by DCs in cancer. J Immunol. 2014;192(6):2920–31. 10.4049/jimmunol.1302801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 344.Düvel K, Yecies JL, Menon S, Raman P, Lipovsky AI, Souza AL, et al. Activation of a metabolic gene regulatory network downstream of mTOR complex 1. Mol Cell. 2010;39(2):171–83. 10.1016/j.molcel.2010.06.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 345.Laplante M, Sabatini DM. mTOR signaling in growth control and disease. Cell. 2012;149(2):274–93. 10.1016/j.cell.2012.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 346.Glaviano A, Foo ASC, Lam HY, Yap KCH, Jacot W, Jones RH, et al. PI3K/AKT/mTOR signaling transduction pathway and targeted therapies in cancer. Mol Cancer. 2023;22(1):138. 10.1186/s12943-023-01827-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 347.Ibrahim J, Nguyen AH, Rehman A, Ochi A, Jamal M, Graffeo CS, et al. DC populations with different concentrations of lipid regulate tolerance and immunity in mouse and human liver. Gastroenterology. 2012;143(4):1061–72. 10.1053/j.gastro.2012.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 348.Naik SH, Proietto AI, Wilson NS, Dakic A, Schnorrer P, Fuchsberger M, et al. Cutting edge: generation of splenic CD8+ and CD8- DC equivalents in Fms-like tyrosine kinase 3 ligand bone marrow cultures. J Immunol. 2005;174(11):6592–7. 10.4049/jimmunol.174.11.6592. [DOI] [PubMed] [Google Scholar]
- 349.Mariño G, Pietrocola F, Eisenberg T, Kong Y, Malik SA, Andryushkova A, et al. Regulation of autophagy by cytosolic acetyl-coenzyme A. Mol Cell. 2014;53(5):710–25. 10.1016/j.molcel.2014.01.016. [DOI] [PubMed] [Google Scholar]
- 350.Pietrocola F, Galluzzi L, Bravo-San Pedro JM, Madeo F, Kroemer G. Acetyl-CoA: a central metabolite and second messenger. Cell Metab. 2015;21(6):805–21. 10.1016/j.cmet.2015.05.014. [DOI] [PubMed] [Google Scholar]
- 351.Sivanand S, Vander Heiden MG. Emerging roles for branched-chain amino acid metabolism in cancer. Cancer Cell. 2020;37(2):147–56. 10.1016/j.ccell.2019.12.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 352.Nicastro R, Brohée L, Alba J, Nüchel J, Figlia G, Kipschull S, et al. Malonyl-CoA is a conserved endogenous ATP-competitive mTORC1 inhibitor. Nat Cell Biol. 2023;25(9):1303–18. 10.1038/s41556-023-01198-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 353.Murphy MP. How mitochondria produce reactive oxygen species. Biochem J. 2009;417(1):1–13. 10.1042/BJ20081386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 354.Mao Y, Zhang J, Zhou Q, He X, Zheng Z, Wei Y, et al. Hypoxia induces mitochondrial protein lactylation to limit oxidative phosphorylation. Cell Res. 2024;34(1):13–30. 10.1038/s41422-023-00864-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 355.Adamik J, Munson PV, Maurer DM, Hartmann FJ, Bendall SC, Argüello RJ, et al. Immuno-metabolic DC vaccine signatures associate with overall survival in vaccinated melanoma patients. Nat Commun. 2023;14(1):7211. 10.1038/s41467-023-42881-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 356.Cho KM, Kim MS, Jung HJ, Choi EJ, Kim TS. Mst1-deficiency induces hyperactivation of monocyte-derived DCs via Akt1/c-myc pathway. Front Immunol. 2019;10:2142. 10.3389/fimmu.2019.02142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 357.McGettrick AF, O’Neill LAJ. The role of HIF in immunity and inflammation. Cell Metab. 2020;32(4):524–36. 10.1016/j.cmet.2020.08.002. [DOI] [PubMed] [Google Scholar]
- 358.Köhler T, Reizis B, Johnson RS, Weighardt H, Förster I. Influence of hypoxia-inducible factor 1α on dendritic cell differentiation and migration. Eur J Immunol. 2012;42(5):1226–36. 10.1002/eji.201142053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 359.Tran Janco JM, Lamichhane P, Karyampudi L, Knutson KL. Tumor-infiltrating dendritic cells in cancer pathogenesis. J Immunol. 2015;194(7):2985–91. 10.4049/jimmunol.1403134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 360.Karagiota A, Kanoura A, Paraskeva E, Simos G, Chachami G. Pyruvate dehydrogenase phosphatase 1 (PDP1) stimulates HIF activity by supporting histone acetylation under hypoxia. FEBS J. 2023;290(8):2165–79. 10.1111/febs.16694. [DOI] [PubMed] [Google Scholar]
- 361.Kim JW, Tchernyshyov I, Semenza GL, Dang CV. HIF-1-mediated expression of pyruvate dehydrogenase kinase: a metabolic switch required for cellular adaptation to hypoxia. Cell Metab. 2006;3(3):177–85. 10.1016/j.cmet.2006.02.002. [DOI] [PubMed] [Google Scholar]
- 362.Cubillos-Ruiz JR, Bettigole SE, Glimcher LH. Molecular Pathways: Immunosuppressive Roles of IRE1α-XBP1 Signaling in DCs of the Tumor Microenvironment. Clin Cancer Res. 2016;22(9):2121–6. 10.1158/1078-0432.CCR-15-1570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 363.Lu YC, Yao X, Crystal JS, Li YF, El-Gamil M, Gross C, et al. Efficient identification of mutated cancer antigens recognized by T cells associated with durable tumor regressions. Clin Cancer Res. 2014;20(13):3401–10. 10.1158/1078-0432.CCR-14-0433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 364.Axelrod ML, Cook RS, Johnson DB, Balko JM. Biological consequences of MHC-II expression by tumor cells in cancer. Clin Cancer Res. 2019;25(8):2392–402. 10.1158/1078-0432.CCR-18-3200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 365.Garrido F, Cabrera T, Lopez-Nevot MA, Ruiz-Cabello F. HLA class I antigens in human tumors. Adv Cancer Res. 1995;67:155–95. 10.1016/s0065-230x(08)60713-7. [DOI] [PubMed] [Google Scholar]
- 366.Brockmann D, Lehmkühler O, Schmücker U, Esche H. The histone acetyltransferase activity of PCAF cooperates with the brahma/SWI2-related protein BRG-1 in the activation of the enhancer A of the MHC class I promoter. Gene. 2001;277(1–2):111–20. 10.1016/s0378-1119(01)00703-x. [DOI] [PubMed] [Google Scholar]
- 367.Gotoh K, Morisaki T, Setoyama D, Sasaki K, Yagi M, Igami K, et al. Mitochondrial p32/C1qbp is a critical regulator of DC metabolism and maturation. Cell Rep. 2018;25(7):1800–15. 10.1016/j.celrep.2018.10.057. [DOI] [PubMed] [Google Scholar]
- 368.Barry KC, Hsu J, Broz ML, Cueto FJ, Binnewies M, Combes AJ, et al. A natural killer-DC axis defines checkpoint therapy-responsive tumor microenvironments. Nat Med. 2018;24(8):1178–91. 10.1038/s41591-018-0085-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 369.Hayashi K, Nikolos F, Lee YC, Jain A, Tsouko E, Gao H, et al. Tipping the immunostimulatory and inhibitory DAMP balance to harness immunogenic cell death. Nat Commun. 2020;11(1):6299. 10.1038/s41467-020-19970-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 370.O’Neill LA. A critical role for citrate metabolism in LPS signalling. Biochem J. 2011;438(3):e5-6. 10.1042/BJ20111386. [DOI] [PubMed] [Google Scholar]
- 371.Guermonprez P, Valladeau J, Zitvogel L, Théry C, Amigorena S. Antigen presentation and T cell stimulation by DCs. Annu Rev Immunol. 2002;20:621–67. 10.1146/annurev.immunol.20.100301.064828. [DOI] [PubMed] [Google Scholar]
- 372.Liu J, Zhang X, Cheng Y, Cao X. DC migration in inflammation and immunity. Cell Mol Immunol. 2021;18(11):2461–71. 10.1038/s41423-021-00726-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 373.Sozzani S. DC trafficking: more than just chemokines. Cytokine Growth Factor Rev. 2005;16(6):581–92. 10.1016/j.cytogfr.2005.04.008. [DOI] [PubMed] [Google Scholar]
- 374.Braun A, Worbs T, Moschovakis GL, Halle S, Hoffmann K, Bölter J, et al. Afferent lymph-derived T cells and DCs use different chemokine receptor CCR7-dependent routes for entry into the lymph node and intranodal migration. Nat Immunol. 2011;12(9):879–87. 10.1038/ni.2085. [DOI] [PubMed] [Google Scholar]
- 375.Martin-Fontecha A, Sebastiani S, Höpken UE, Uguccioni M, Lipp M, Lanzavecchia A, et al. Regulation of DC migration to the draining lymph node: impact on T lymphocyte traffic and priming. J Exp Med. 2003;198(4):615–21. 10.1084/jem.20030448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 376.Roberts EW, Broz ML, Binnewies M, Headley MB, Nelson AE, Wolf DM, et al. Critical role for CD103(+)/CD141(+) DCs bearing CCR7 for tumor antigen trafficking and priming of T cell immunity in melanoma. Cancer Cell. 2016;30(2):324–36. 10.1016/j.ccell.2016.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 377.Tiniakou I, Drakos E, Sinatkas V, Van Eck M, Zannis VI, Boumpas D, et al. High-density lipoprotein attenuates Th1 and th17 autoimmune responses by modulating DC maturation and function. J Immunol. 2015;194(10):4676–87. 10.4049/jimmunol.1402870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 378.Machy P, Serre K, Baillet M, Leserman L. Induction of MHC class I presentation of exogenous antigen by dendritic cells is controlled by CD4+ T cells engaging class II molecules in cholesterol-rich domains. J Immunol. 2002;168(3):1172–80. 10.4049/jimmunol.168.3.1172. [DOI] [PubMed] [Google Scholar]
- 379.Angeli V, Llodrá J, Rong JX, Satoh K, Ishii S, Shimizu T, et al. Dyslipidemia associated with atherosclerotic disease systemically alters dendritic cell mobilization. Immunity. 2004;21(4):561–74. 10.1016/j.immuni.2004.09.003. [DOI] [PubMed] [Google Scholar]
- 380.Lühr JJ, Alex N, Amon L, Kräter M, Kubánková M, Sezgin E, et al. Maturation of monocyte-derived DCs leads to increased cellular stiffness, higher membrane fluidity, and changed lipid composition. Front Immunol. 2020;11: 590121. 10.3389/fimmu.2020.590121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 381.Ferrara G, Benzi A, Sturla L, Marubbi D, Frumento D, Spinelli S, et al. Sirt6 inhibition delays the onset of experimental autoimmune encephalomyelitis by reducing DC migration. J Neuroinflammation. 2020;17(1):228. 10.1186/s12974-020-01906-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 382.Böttcher JP, Bonavita E, Chakravarty P, Blees H, Cabeza-Cabrerizo M, Sammicheli S, et al. NK cells stimulate recruitment of cDC1 into the tumor microenvironment promoting cancer immune control. Cell. 2018;172(5):1022–37. 10.1016/j.cell.2018.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 383.Sun Y, Mu G, Zhang X, Wu Y, Wang S, Wang X, et al. Metabolic modulation of histone acetylation mediated by HMGCL activates the FOXM1/β-catenin pathway in glioblastoma. Neuro Oncol. 2024;26(4):653–69. 10.1093/neuonc/noad232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 384.Spranger S, Bao R, Gajewski TF. Melanoma-intrinsic β-catenin signalling prevents anti-tumour immunity. Nature. 2015;523(7559):231–5. 10.1038/nature14404. [DOI] [PubMed] [Google Scholar]
- 385.Almeida AR, Rocha B, Freitas AA, Tanchot C. Homeostasis of T cell numbers: from thymus production to peripheral compartmentalization and the indexation of regulatory T cells. Semin Immunol. 2005;17(3):239–49. 10.1016/j.smim.2005.02.002. [DOI] [PubMed] [Google Scholar]
- 386.Sprent J, Surh CD. Normal T cell homeostasis: the conversion of naive cells into memory-phenotype cells. Nat Immunol. 2011;12(6):478–84. 10.1038/ni.2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 387.Kimura MY, Pobezinsky LA, Guinter TI, Thomas J, Adams A, Park JH, et al. IL-7 signaling must be intermittent, not continuous, during CD8⁺ T cell homeostasis to promote cell survival instead of cell death. Nat Immunol. 2013;14(2):143–51. 10.1038/ni.2494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 388.Kim GY, Hong C, Park JH. Seeing is believing: illuminating the source of in vivo interleukin-7. Immune Netw. 2011;11(1):1–10. 10.4110/in.2011.11.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 389.Wang C, Kong L, Kim S, Lee S, Oh S, Jo S, et al. The role of IL-7 and IL-7R in cancer pathophysiology and immunotherapy. Int J Mol Sci. 2022;23(18): 10412. 10.3390/ijms231810412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 390.Adachi K, Kano Y, Nagai T, Okuyama N, Sakoda Y, Tamada K. IL-7 and CCL19 expression in CAR-T cells improves immune cell infiltration and CAR-T cell survival in the tumor. Nat Biotechnol. 2018;36(4):346–51. 10.1038/nbt.4086. [DOI] [PubMed] [Google Scholar]
- 391.Bi E, Ma X, Lu Y, Yang M, Wang Q, Xue G, et al. Foxo1 and Foxp1 play opposing roles in regulating the differentiation and antitumor activity of TH9 cells programmed by IL-7. Sci Signal. 2017;10(500): eaak9741. 10.1126/scisignal.aak9741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 392.He W, Li Q, Li X. Acetyl-CoA regulates lipid metabolism and histone acetylation modification in cancer. Biochimica et Biophysica Acta (BBA). 2023;1878(1): 188837. 10.1016/j.bbcan.2022.188837. [DOI] [PubMed] [Google Scholar]
- 393.Gu D, Ye M, Zhu G, Bai J, Chen J, Yan L, et al. Hypoxia upregulating ACSS2 enhances lipid metabolism reprogramming through HMGCS1 mediated PI3K/AKT/mTOR pathway to promote the progression of pancreatic neuroendocrine neoplasms. J Transl Med. 2024;22(1):93. 10.1186/s12967-024-04870-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 394.Márquez S, Fernández JJ, Mancebo C, Herrero-Sánchez C, Alonso S, Sandoval TA, et al. Tricarboxylic Acid Cycle Activity and Remodeling of Glycerophosphocholine Lipids Support Cytokine Induction in Response to Fungal Patterns. Cell Rep. 2019;27(2):525–36. 10.1016/j.celrep.2019.03.033. [DOI] [PubMed] [Google Scholar]
- 395.Diamond MS, Kinder M, Matsushita H, Mashayekhi M, Dunn GP, Archambault JM, et al. Type I interferon is selectively required by DCs for immune rejection of tumors. J Exp Med. 2011;208(10):1989–2003. 10.1084/jem.20101158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 396.Sisirak V, Faget J, Gobert M, Goutagny N, Vey N, Treilleux I, et al. Impaired IFN-α production by plasmacytoid DCs favors regulatory T-cell expansion that may contribute to breast cancer progression. Cancer Res. 2012;72(20):5188–97. 10.1158/0008-5472.CAN-11-3468. [DOI] [PubMed] [Google Scholar]
- 397.Siska PJ, Rathmell JC. Metabolic signaling drives IFN-γ. Cell Metab. 2016;24(5):651–2. 10.1016/j.cmet.2016.10.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 398.Bol KF, Schreibelt G, Gerritsen WR, de Vries IJ, Figdor CG. Dendritic Cell-Based Immunotherapy: State of the Art and Beyond. Clin Cancer Res. 2016;22(8):1897–906. 10.1158/1078-0432.CCR-15-1399. [DOI] [PubMed] [Google Scholar]
- 399.El-Ashmawy NE, El-Zamarany EA, Khedr EG, El-Bahrawy HA, El-Feky OA. Immunotherapeutic strategies for treatment of hepatocellular carcinoma with antigen-loaded dendritic cells: in vivo study. Clin Exp Med. 2018;18(4):535–46. 10.1007/s10238-018-0521-6. [DOI] [PubMed] [Google Scholar]
- 400.Peng X, He Y, Huang J, Tao Y, Liu S. Metabolism of dendritic cells in tumor microenvironment: for immunotherapy. Front Immunol. 2021;12: 613492. 10.3389/fimmu.2021.613492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 401.Xu Y, Wan W. Acetylation in the regulation of autophagy. Autophagy. 2023;19(2):379–87. 10.1080/15548627.2022.2062112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 402.Carreno BM, Becker-Hapak M, Huang A, Chan M, Alyasiry A, Lie WR, et al. IL-12p70-producing patient DC vaccine elicits Tc1-polarized immunity. J Clin Invest. 2013;123(8):3383–94. 10.1172/JCI68395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 403.Nguyen-Phuong T, Chung H, Jang J, Kim JS, Park CG. Acetyl-CoA carboxylase-1/2 blockade locks dendritic cells in the semimature state associated with FA deprivation by favoring FAO. J Leukoc Biol. 2022;111(3):539–51. 10.1002/JLB.1A0920-561RR. [DOI] [PubMed] [Google Scholar]
- 404.Matsumoto M, Takeda Y, Seya T. Targeting toll-like receptor 3 in dendritic cells for cancer immunotherapy. Expert Opin Biol Ther. 2020;20(8):937–46. 10.1080/14712598.2020.1749260. [DOI] [PubMed] [Google Scholar]
- 405.Lai L, Reineke E, Hamilton DJ, Cooke JP. Glycolytic switch is required for transdifferentiation to endothelial lineage. Circulation. 2019;139(1):119–33. 10.1161/CIRCULATIONAHA.118.035741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 406.Shortman K, Heath WR. The CD8+ dendritic cell subset. Immunol Rev. 2010;234(1):18–31. 10.1111/j.0105-2896.2009.00870.x. [DOI] [PubMed] [Google Scholar]
- 407.Wang J, Yang Y, Shao F, Meng Y, Guo D, He J, et al. Acetate reprogrammes tumour metabolism and promotes PD-L1 expression and immune evasion by upregulating c-Myc. Nat Metab. 2024;6(5):914–32. 10.1038/s42255-024-01037-4. [DOI] [PubMed] [Google Scholar]
- 408.Wartewig T, Daniels J, Schulz M, Hameister E, Joshi A, Park J, et al. PD-1 instructs a tumor-suppressive metabolic program that restricts glycolysis and restrains AP-1 activity in T cell lymphoma. Nat Cancer. 2023;4(10):1508–25. 10.1038/s43018-023-00635-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 409.Strauss L, Mahmoud MAA, Weaver JD, Tijaro-Ovalle NM, Christofides A, Wang Q, et al. Targeted deletion of PD-1 in myeloid cells induces antitumor immunity. Sci Immunol. 2020;5(43): eaay1863. 10.1126/sciimmunol.aay1863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 410.Ghorbaninezhad F, Masoumi J, Bakhshivand M, Baghbanzadeh A, Mokhtarzadeh A, Kazemi T, et al. CTLA-4 silencing in dendritic cells loaded with colorectal cancer cell lysate improves autologous T cell responses in vitro. Front Immunol. 2022;13: 931316. 10.3389/fimmu.2022.931316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 411.Choi SY, Kim Y, Lim B, Wee CB, Chang IH, Kim CS. Prostate cancer therapy using immune checkpoint molecules to target recombinant dendritic cells. Investig Clin Urol. 2024;65(3):300–10. 10.4111/icu.20230348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 412.Porstmann T, Santos CR, Griffiths B, Cully M, Wu M, Leevers S, et al. SREBP activity is regulated by mTORC1 and contributes to Akt-dependent cell growth. Cell Metab. 2008;8(3):224–36. 10.1016/j.cmet.2008.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 413.Ning Z, Guo X, Liu X, Lu C, Wang A, Wang X, et al. USP22 regulates lipidome accumulation by stabilizing PPARγ in hepatocellular carcinoma. Nat Commun. 2022;13(1):2187. 10.1038/s41467-022-29846-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 414.Svensson RU, Parker SJ, Eichner LJ, Kolar MJ, Wallace M, Brun SN, et al. Inhibition of acetyl-CoA carboxylase suppresses fatty acid synthesis and tumor growth of non-small-cell lung cancer in preclinical models. Nat Med. 2016;22(10):1108–19. 10.1038/nm.4181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 415.Liefwalker DF, Ryan M, Wang Z, Pathak KV, Plaisier S, Shah V, et al. Metabolic convergence on lipogenesis in RAS, BCR-ABL, and MYC-driven lymphoid malignancies. Cancer Metab. 2021;9(1):31. 10.1186/s40170-021-00263-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 416.Pearce NJ, Yates JW, Berkhout TA, Jackson B, Tew D, Boyd H, et al. The role of ATP citrate-lyase in the metabolic regulation of plasma lipids. Hypolipidaemic effects of SB-204990, a lactone prodrug of the potent ATP citrate-lyase inhibitor SB-201076. Biochem J. 1998;334(Pt 1):113–9. 10.1042/bj3340113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 417.Hatzivassiliou G, Zhao F, Bauer DE, Andreadis C, Shaw AN, Dhanak D, et al. ATP citrate lyase inhibition can suppress tumor cell growth. Cancer Cell. 2005;8(4):311–21. 10.1016/j.ccr.2005.09.008. [DOI] [PubMed] [Google Scholar]
- 418.Zheng Y, Zhou Q, Zhao C, Li J, Yu Z, Zhu Q. ATP citrate lyase inhibitor triggers endoplasmic reticulum stress to induce hepatocellular carcinoma cell apoptosis via p-eIF2α/ATF4/CHOP axis. J Cell Mol Med. 2021;25(3):1468–79. 10.1111/jcmm.16235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 419.Gu CW, Qian H, Liu YZ, Zhao BS. Zhongguo ying yong sheng li xue za zhi = Zhongguo yingyong shenglixue zazhi = Chinese journal of applied physiology. 2022;38(4):317–21. 10.12047/j.cjap.6236.2022.060 [DOI] [PubMed]
- 420.Chajès V, Cambot M, Moreau K, Lenoir GM, Joulin V. Acetyl-CoA carboxylase alpha is essential to breast cancer cell survival. Cancer Res. 2006;66(10):5287–94. 10.1158/0008-5472.CAN-05-1489. [DOI] [PubMed] [Google Scholar]
- 421.Zhou W, Han WF, Landree LE, Thupari JN, Pinn ML, Bililign T, et al. Fatty acid synthase inhibition activates AMP-activated protein kinase in SKOV3 human ovarian cancer cells. Cancer Res. 2007;67(7):2964–71. 10.1158/0008-5472.CAN-06-3439. [DOI] [PubMed] [Google Scholar]
- 422.Chu Q, An J, Liu P, Song Y, Zhai X, Yang R, et al. Repurposing a tricyclic antidepressant in tumor and metabolism disease treatment through fatty acid uptake inhibition. J Exp Med. 2023;220(3): e20221316. 10.1084/jem.20221316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 423.Li EQ, Zhao W, Zhang C, Qin LZ, Liu SJ, Feng ZQ, et al. Synthesis and anti-cancer activity of ND-646 and its derivatives as acetyl-CoA carboxylase 1 inhibitors. Eur J Pharm Sci. 2019;137: 105010. 10.1016/j.ejps.2019.105010. [DOI] [PubMed] [Google Scholar]
- 424.Lally JSV, Ghoshal S, DePeralta DK, Moaven O, Wei L, Masia R, et al. Inhibition of Acetyl-CoA Carboxylase by Phosphorylation or the Inhibitor ND-654 Suppresses Lipogenesis and Hepatocellular Carcinoma. Cell Metab. 2019;29(1):174-82.e5. 10.1016/j.cmet.2018.08.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 425.Wu X, Yu Y, Huang T. Synthesis and biological evaluation of 4-phenoxy-phenyl isoxazoles as novel acetyl-CoA carboxylase inhibitors. J Enzyme Inhib Med Chem. 2021;36(1):1236–47. 10.1080/14756366.2021.1936514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 426.Wu YP, Zheng WC, Huang Q, Huang XY, Lin F, Ke ZB, et al. ND630 controls ACACA and lipid reprogramming in prostate cancer by regulating the expression of circKIF18B_003. J Transl Med. 2023;21(1):877. 10.1186/s12967-023-04760-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 427.Tanaka K, Chamoto K, Saeki S, Hatae R, Ikematsu Y, Sakai K, et al. Combination bezafibrate and nivolumab treatment of patients with advanced non-small cell lung cancer. Sci Transl Med. 2022;14(675): eabq0021. 10.1126/scitranslmed.abq0021. [DOI] [PubMed] [Google Scholar]
- 428.Zhang L, Zhou X, Liu B, Shi X, Li X, Xu F, et al. HBXIP blocks myosin-IIA assembly by phosphorylating and interacting with NMHC-IIA in breast cancer metastasis. Acta Pharm Sin B. 2023;13(3):1053–70. 10.1016/j.apsb.2022.11.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 429.Stoiber K, Nagło O, Pernpeintner C, Zhang S, Koeberle A, Ulrich M, et al. Targeting de novo lipogenesis as a novel approach in anti-cancer therapy. Br J Cancer. 2018;118(1):43–51. 10.1038/bjc.2017.374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 430.Schwartz L, Abolhassani M, Guais A, Sanders E, Steyaert JM, Campion F, et al. A combination of alpha lipoic acid and calcium hydroxycitrate is efficient against mouse cancer models: preliminary results. Oncol Rep. 2010;23(5):1407–16. 10.3892/or_00000778. [DOI] [PubMed] [Google Scholar]
- 431.Huang SS, Tsai CH, Kuo CY, Li YS, Cheng SP. ACLY inhibitors induce apoptosis and potentiate cytotoxic effects of sorafenib in thyroid cancer cells. Endocrine. 2022;78(1):85–94. 10.1007/s12020-022-03124-6. [DOI] [PubMed] [Google Scholar]
- 432.Velez BC, Petrella CP, DiSalvo KH, Cheng K, Kravtsov R, Krasniqi D, et al. Combined inhibition of ACLY and CDK4/6 reduces cancer cell growth and invasion. Oncol Rep. 2023;49(2):32. 10.3892/or.2022.8469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 433.Alafnan A, Alamri A, Hussain T, Rizvi SMD. Cucurbitacin-B exerts anticancer effects through instigation of apoptosis and cell cycle arrest within human prostate cancer PC3 cells via downregulating JAK/STAT signaling cascade. Pharmaceuticals (Basel). 2022;15(10): 1229. 10.3390/ph15101229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 434.Shen F, Ge C, Yuan P. Aloe-emodin induces autophagy and apoptotic cell death in non-small cell lung cancer cells via Akt/mTOR and MAPK signaling. Eur J Pharmacol. 2020;886: 173550. 10.1016/j.ejphar.2020.173550. [DOI] [PubMed] [Google Scholar]
- 435.Beteta-Göbel R, Miralles M, Fernández-Díaz J, Rodríguez-Lorca R, Torres M, Fernández-García P, et al. HCA (2-Hydroxy-Docosahexaenoic acid) induces apoptosis and endoplasmic reticulum stress in pancreatic cancer cells. Int J Mol Sci. 2022;23(17): 9902. 10.3390/ijms23179902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 436.Pardee TS, Lee K, Luddy J, Maturo C, Rodriguez R, Isom S, et al. A phase I study of the first-in-class antimitochondrial metabolism agent, CPI-613, in patients with advanced hematologic malignancies. Clin Cancer Res. 2014;20(20):5255–64. 10.1158/1078-0432.CCR-14-1019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 437.Dunbar EM, Coats BS, Shroads AL, Langaee T, Lew A, Forder JR, et al. Phase 1 trial of dichloroacetate (DCA) in adults with recurrent malignant brain tumors. Invest New Drugs. 2014;32(3):452–64. 10.1007/s10637-013-0047-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 438.Chu QS, Sangha R, Spratlin J, Vos LJ, Mackey JR, McEwan AJ, et al. A phase I open-labeled, single-arm, dose-escalation, study of dichloroacetate (DCA) in patients with advanced solid tumors. Invest New Drugs. 2015;33(3):603–10. 10.1007/s10637-015-0221-y. [DOI] [PubMed] [Google Scholar]
- 439.Ricco N, Kron SJ. Statins in cancer prevention and therapy. Cancers (Basel). 2023;15(15): 3948. 10.3390/cancers15153948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 440.Craig EL, Stopsack KH, Evergren E, Penn LZ, Freedland SJ, Hamilton RJ, et al. Statins and prostate cancer-hype or hope? The epidemiological perspective. Prostate Cancer Prostatic Dis. 2022;25(4):641–9. 10.1038/s41391-022-00554-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 441.Miller KD, Pniewski K, Perry CE, Papp SB, Shaffer JD, Velasco-Silva JN, et al. Targeting ACSS2 with a transition-state mimetic inhibits triple-negative breast cancer growth. Cancer Res. 2021;81(5):1252–64. 10.1158/0008-5472.CAN-20-1847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 442.Esquea EM, Ciraku L, Young RG, Merzy J, Talarico AN, Ahmed NN, et al. Selective and brain-penetrant ACSS2 inhibitors target breast cancer brain metastatic cells. Front Pharmacol. 2024;15:1394685. 10.3389/fphar.2024.1394685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 443.Baell JB, Leaver DJ, Hermans SJ, Kelly GL, Brennan MS, Downer NL, et al. Inhibitors of histone acetyltransferases KAT6A/B induce senescence and arrest tumour growth. Nature. 2018;560(7717):253–7. 10.1038/s41586-018-0387-5. [DOI] [PubMed] [Google Scholar]
- 444.Mukohara T, Park YH, Sommerhalder D, Yonemori K, Hamilton E, Kim SB, et al. Inhibition of lysine acetyltransferase KAT6 in ER+HER2- metastatic breast cancer: a phase 1 trial. Nat Med. 2024;30(8):2242–50. 10.1038/s41591-024-03060-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 445.Welti J, Sharp A, Brooks N, Yuan W, McNair C, Chand SN, et al. Targeting the p300/CBP axis in lethal prostate cancer. Cancer Discov. 2021;11(5):1118–37. 10.1158/2159-8290.CD-20-0751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 446.Caligiuri M, Williams GL, Castro J, Battalagine L, Wilker E, Yao L, et al. FT-6876, a potent and selective inhibitor of CBP/p300, is active in preclinical models of androgen receptor-positive breast cancer. Target Oncol. 2023;18(2):269–85. 10.1007/s11523-023-00949-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 447.Pricci M, Girardi B, Giorgio F, Losurdo G, Ierardi E, Di Leo A. Curcumin and colorectal cancer: from basic to clinical evidences. Int J Mol Sci. 2020;21(7): 2364. 10.3390/ijms21072364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 448.Mi YY, Ji Y, Zhang L, Sun CY, Wei BB, Yang DJ, et al. A first-in-class HBO1 inhibitor WM-3835 inhibits castration-resistant prostate cancer cell growth in vitro and in vivo. Cell Death Dis. 2023;14(1):67. 10.1038/s41419-023-05606-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 449.Xie M, Zhou L, Li T, Lin Y, Zhang R, Zheng X, et al. Targeting the KAT8/YEATS4 Axis Represses Tumor Growth and Increases Cisplatin Sensitivity in Bladder Cancer. Adv Sci (Weinh). 2024;11(22): e2310146. 10.1002/advs.202310146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 450.Hsin IL, Wu PJ, Tang SC, Ou CC, Chang HY, Shen HP, et al. Β-catenin inhibitor ICG-001 suppress cell cycle progression and induce autophagy in endometrial cancer cells. J Cell Physiol. 2023;238(10):2440–50. 10.1002/jcp.31103. [DOI] [PubMed] [Google Scholar]
- 451.Parveen R, Harihar D, Chatterji BP. Recent histone deacetylase inhibitors in cancer therapy. Cancer. 2023;129(21):3372–80. 10.1002/cncr.34974. [DOI] [PubMed] [Google Scholar]
- 452.Ikeda M, Ohno I, Ueno H, Mitsunaga S, Hashimoto Y, Okusaka T, et al. Phase I study of resminostat, an HDAC inhibitor, combined with S-1 in patients with pre-treated biliary tract or pancreatic cancer. Invest New Drugs. 2019;37(1):109–17. 10.1007/s10637-018-0634-5. [DOI] [PubMed] [Google Scholar]
- 453.Horwitz SM, Nirmal AJ, Rahman J, Xu R, Drill E, Galasso N, et al. Duvelisib plus romidepsin in relapsed/refractory T cell lymphomas: a phase 1b/2a trial. Nat Med. 2024;30(9):2517–27. 10.1038/s41591-024-03076-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 454.San-Miguel JF, Hungria VT, Yoon SS, Beksac M, Dimopoulos MA, Elghandour A, et al. Panobinostat plus bortezomib and dexamethasone versus placebo plus bortezomib and dexamethasone in patients with relapsed or relapsed and refractory multiple myeloma: a multicentre, randomised, double-blind phase 3 trial. Lancet Oncol. 2014;15(11):1195–206. 10.1016/S1470-2045(14)70440-1. [DOI] [PubMed] [Google Scholar]
- 455.O’Connor OA, Horwitz S, Masszi T, Van Hoof A, Brown P, Doorduijn J, et al. Belinostat in patients with relapsed or refractory peripheral T-cell lymphoma: results of the pivotal phase II belief (CLN-19) study. J Clin Oncol. 2015;33(23):2492–9. 10.1200/JCO.2014.59.2782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 456.Wanek J, Gaisberger M, Beyreis M, Mayr C, Helm K, Primavesi F, et al. Pharmacological inhibition of class IIA HDACs by LMK-235 in pancreatic neuroendocrine tumor cells. Int J Mol Sci. 2018;19(10): 3128. 10.3390/ijms19103128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 457.Zeleke TZ, Pan Q, Chiuzan C, Onishi M, Li Y, Tan H, et al. Network-based assessment of HDAC6 activity predicts preclinical and clinical responses to the HDAC6 inhibitor ricolinostat in breast cancer. Nat Cancer. 2023;4(2):257–75. 10.1038/s43018-022-00489-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 458.Wang F, Jin Y, Wang M, Luo HY, Fang WJ, Wang YN, et al. Combined anti-PD-1, HDAC inhibitor and anti-VEGF for MSS/pMMR colorectal cancer: a randomized phase 2 trial. Nat Med. 2024;30(4):1035–43. 10.1038/s41591-024-02813-1. [DOI] [PubMed] [Google Scholar]
- 459.Rai S, Kim WS, Ando K, Choi I, Izutsu K, Tsukamoto N, et al. Oral HDAC inhibitor tucidinostat in patients with relapsed or refractory peripheral T-cell lymphoma: phase IIb results. Haematologica. 2023;108(3):811–21. 10.3324/haematol.2022.280996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 460.Gentzler RD, Villaruz LC, Rhee JC, Horton B, Mock J, Hanley M, et al. Phase I study of entinostat, atezolizumab, carboplatin, and etoposide in previously untreated extensive-stage small cell lung cancer, ETCTN 10399. Oncologist. 2023;28(11):1007-e1107. 10.1093/oncolo/oyad221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 461.Ye M, Hu C, Chen T, Yu P, Chen J, Lu F, et al. FABP5 suppresses colorectal cancer progression via mTOR-mediated autophagy by decreasing FASN expression. Int J Biol Sci. 2023;19(10):3115–27. 10.7150/ijbs.85285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 462.Huang J, Tsang WY, Fang XN, Zhang Y, Luo J, Gong LQ, et al. Fasn inhibition decreases MHC-I degradation and synergizes with PD-L1 checkpoint blockade in hepatocellular carcinoma. Cancer Res. 2024;84(6):855–71. 10.1158/0008-5472.CAN-23-0966. [DOI] [PubMed] [Google Scholar]
- 463.Du Q, Liu P, Zhang C, Liu T, Wang W, Shang C, et al. Fasn promotes lymph node metastasis in cervical cancer via cholesterol reprogramming and lymphangiogenesis. Cell Death Dis. 2022;13(5):488. 10.1038/s41419-022-04926-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 464.Flavin R, Peluso S, Nguyen PL, Loda M. Fatty acid synthase as a potential therapeutic target in cancer. Future Oncol. 2010;6(4):551–62. 10.2217/fon.10.11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 465.Scheinberg T, Mak B, Butler L, Selth L, Horvath LG. Targeting lipid metabolism in metastatic prostate cancer. Ther Adv Med Oncol. 2023;15:17588359231152840. 10.1177/17588359231152839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 466.Wang J, Xiang H, Lu Y, Wu T, Ji G. The role and therapeutic implication of CPTs in fatty acid oxidation and cancers progression. Am J Cancer Res. 2021;11(6):2477–94. [PMC free article] [PubMed] [Google Scholar]
- 467.Ricciardi MR, Mirabilii S, Allegretti M, Licchetta R, Calarco A, Torrisi MR, et al. Targeting the leukemia cell metabolism by the CPT1a inhibition: functional preclinical effects in leukemias. Blood. 2015;126(16):1925–9. 10.1182/blood-2014-12-617498. [DOI] [PubMed] [Google Scholar]
- 468.Kant S, Kesarwani P, Guastella AR, Kumar P, Graham SF, Buelow KL, et al. Perhexiline demonstrates FYN-mediated antitumor activity in glioblastoma. Mol Cancer Ther. 2020;19(7):1415–22. 10.1158/1535-7163.MCT-19-1047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 469.Wang J, Zhou Y, Zhang D, Zhao W, Lu Y, Liu C, et al. CRIP1 suppresses BBOX1-mediated carnitine metabolism to promote stemness in hepatocellular carcinoma. EMBO J. 2022;41(15): e110218. 10.15252/embj.2021110218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 470.Shafabakhsh R, Reiter RJ, Aschner M, Mirzaei H, Asemi Z. Resveratrol and cervical cancer: a new therapeutic option. Mini Rev Med Chem. 2023;23(2):159–69. 10.2174/1389557522666220128155525. [DOI] [PubMed] [Google Scholar]
- 471.Gross MI, Demo SD, Dennison JB, Chen L, Chernov-Rogan T, Goyal B, et al. Antitumor activity of the glutaminase inhibitor CB-839 in triple-negative breast cancer. Mol Cancer Ther. 2014;13(4):890–901. 10.1158/1535-7163.MCT-13-0870. [DOI] [PubMed] [Google Scholar]
- 472.Encarnación-Rosado J, Sohn ASW, Biancur DE, Lin EY, Osorio-Vasquez V, Rodrick T, et al. Targeting pancreatic cancer metabolic dependencies through glutamine antagonism. Nat Cancer. 2024;5(1):85–99. 10.1038/s43018-023-00647-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 473.Soth MJ, Le K, Di Francesco ME, Hamilton MM, Liu G, Burke JP, et al. Discovery of IPN60090, a clinical stage selective glutaminase-1 (GLS-1) inhibitor with excellent pharmacokinetic and physicochemical properties. J Med Chem. 2020;63(21):12957–77. 10.1021/acs.jmedchem.0c01398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 474.Cyriac R, Lee K. Glutaminase inhibition as potential cancer therapeutics: current status and future applications. J Enzyme Inhib Med Chem. 2024;39(1): 2290911. 10.1080/14756366.2023.2290911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 475.Jiang L, Fang X, Wang H, Li D, Wang X. Ovarian cancer-intrinsic fatty acid synthase prevents anti-tumor immunity by disrupting tumor-infiltrating dendritic cells. Front Immunol. 2018;9:2927. 10.3389/fimmu.2018.02927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 476.Qin YT, Liu XH, An JX, Liang JL, Li CX, Jin XK, et al. Dendritic cell-based in situ nanovaccine for reprogramming lipid metabolism to boost tumor immunotherapy. ACS Nano. 2023;17(24):24947–60. 10.1021/acsnano.3c06784. [DOI] [PubMed] [Google Scholar]
- 477.Pandey VK, Amin PJ, Shankar BS. COX-2 inhibitor prevents tumor induced down regulation of classical DC lineage specific transcription factor Zbtb46 resulting in immunocompetent DC and decreased tumor burden. Immunol Lett. 2017;184:23–33. 10.1016/j.imlet.2017.01.019. [DOI] [PubMed] [Google Scholar]
- 478.Zelenay S, van der Veen AG, Böttcher JP, Snelgrove KJ, Rogers N, Acton SE, et al. Cyclooxygenase-dependent tumor growth through evasion of immunity. Cell. 2015;162(6):1257–70. 10.1016/j.cell.2015.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 479.Take Y, Koizumi S, Nagahisa A. Prostaglandin E receptor 4 antagonist in cancer immunotherapy: mechanisms of action. Front Immunol. 2020;11:324. 10.3389/fimmu.2020.00324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 480.Jochems C, Fantini M, Fernando RI, Kwilas AR, Donahue RN, Lepone LM, et al. The IDO1 selective inhibitor epacadostat enhances dendritic cell immunogenicity and lytic ability of tumor antigen-specific T cells. Oncotarget. 2016;7(25):37762–72. 10.18632/oncotarget.9326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 481.Soliman H, Khambati F, Han HS, Ismail-Khan R, Bui MM, Sullivan DM, et al. A phase-1/2 study of adenovirus-p53 transduced dendritic cell vaccine in combination with indoximod in metastatic solid tumors and invasive breast cancer. Oncotarget. 2018;9(11):10110–7. 10.18632/oncotarget.24118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 482.Brincks EL, Adams J, Wang L, Turner B, Marcinowicz A, Ke J, et al. Indoximod opposes the immunosuppressive effects mediated by IDO and TDO via modulation of AhR function and activation of mTORC1. Oncotarget. 2020;11(25):2438–61. 10.18632/oncotarget.27646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 483.Ghorbaninezhad F, Nour MA, Farzam OR, Saeedi H, Vanan AG, Bakhshivand M, et al. The tumor microenvironment and dendritic cells: developers of pioneering strategies in colorectal cancer immunotherapy? Biochimica et Biophysica Acta (BBA). 2025;1880(2): 189281. 10.1016/j.bbcan.2025.189281. [DOI] [PubMed] [Google Scholar]
- 484.Li Q, Rao R, Vazzana J, Goedegebuure P, Odunsi K, Gillanders W, et al. Regulating mammalian target of rapamycin to tune vaccination-induced CD8(+) T cell responses for tumor immunity. J Immunol. 2012;188(7):3080–7. 10.4049/jimmunol.1103365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 485.Sharma MD, Pacholczyk R, Shi H, Berrong ZJ, Zakharia Y, Greco A, et al. Inhibition of the BTK-IDO-mTOR axis promotes differentiation of monocyte-lineage dendritic cells and enhances anti-tumor T cell immunity. Immunity. 2021;54(10):2354-71.e8. 10.1016/j.immuni.2021.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 486.Li Q, Liu J, Fan H, Shi L, Deng Y, Zhao L, et al. Ido-inhibitor potentiated immunogenic chemotherapy abolishes primary tumor growth and eradicates metastatic lesions by targeting distinct compartments within tumor microenvironment. Biomaterials. 2021;269: 120388. 10.1016/j.biomaterials.2020.120388. [DOI] [PubMed] [Google Scholar]
- 487.Shahverdi M, Masoumi J, Ghorbaninezhad F, Shajari N, Hajizadeh F, Hassanian H, et al. The modulatory role of dendritic cell-T cell cross-talk in breast cancer: challenges and prospects. Adv Med Sci. 2022;67(2):353–63. 10.1016/j.advms.2022.09.001. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No applicable.