Abstract
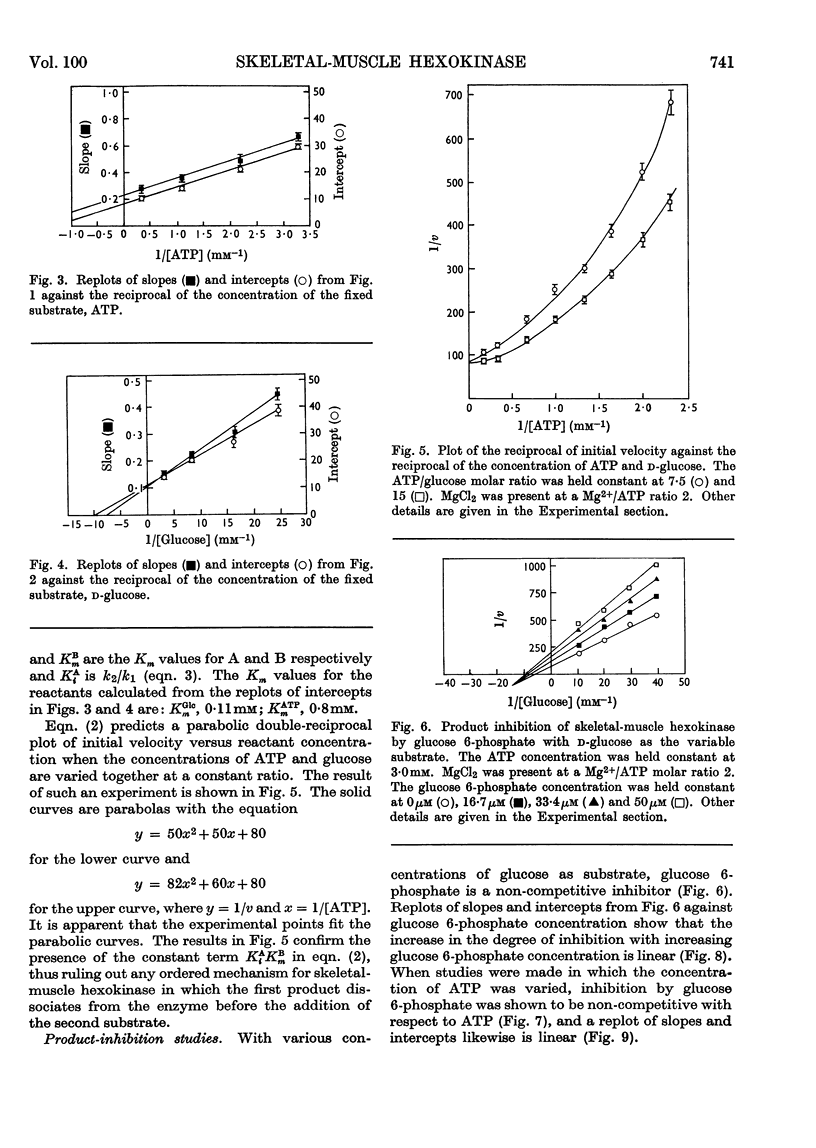
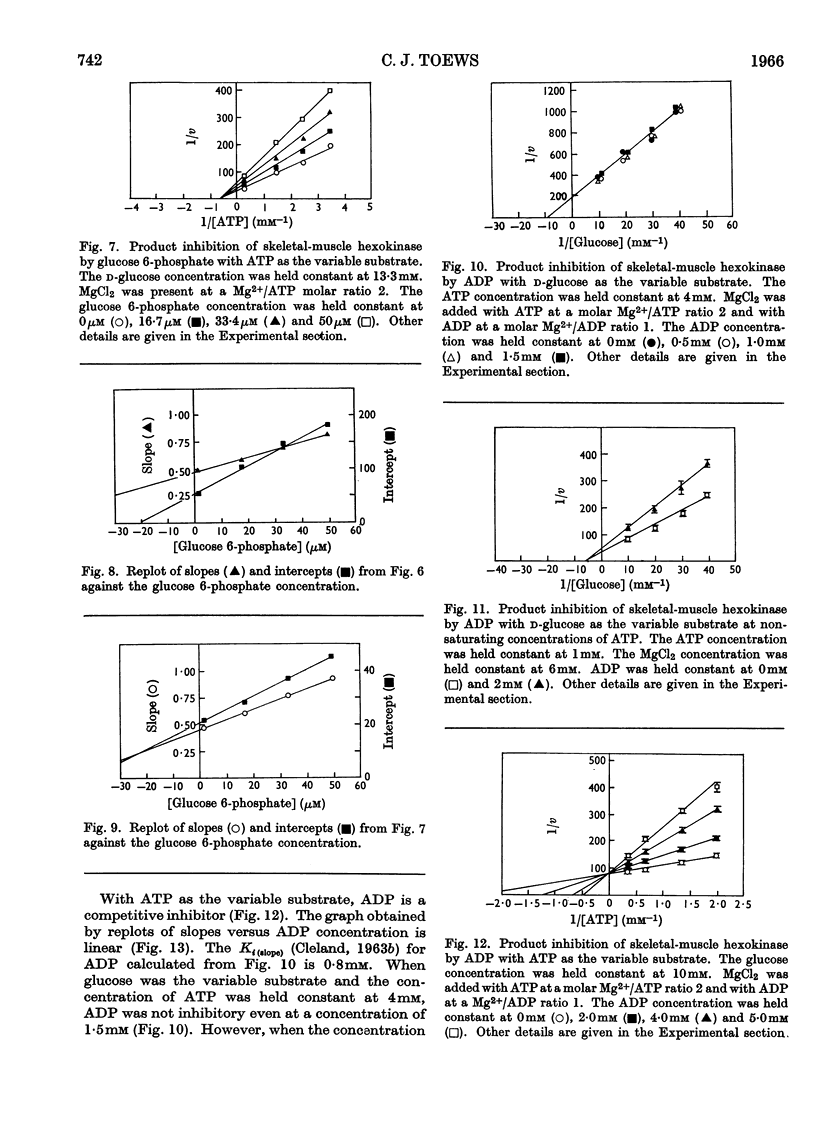
Rat skeletal-muscle hexokinase was partially purified by ammonium sulphate fractionation and gel filtration. The mechanism of the skeletal-muscle hexokinase was studied kinetically by initial-velocity analysis and product inhibition. Glucose 6-phosphate was a non-competitive inhibitor of glucose and ATP. ADP was a non-competitive inhibitor of glucose and a competitive inhibitor of ATP. The data on product inhibition and initial-velocity analysis of skeletal-muscle hexokinase support an ordered sequential mechanism (ordered Bi Bi) where the addition of substrates and release of products is in the order: ATP, glucose, glucose 6-phosphate and ADP.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- CLELAND W. W. The kinetics of enzyme-catalyzed reactions with two or more substrates or products. I. Nomenclature and rate equations. Biochim Biophys Acta. 1963 Jan 8;67:104–137. doi: 10.1016/0006-3002(63)91800-6. [DOI] [PubMed] [Google Scholar]
- CLELAND W. W. The kinetics of enzyme-catalyzed reactions with two or more substrates or products. II. Inhibition: nomenclature and theory. Biochim Biophys Acta. 1963 Feb 12;67:173–187. doi: 10.1016/0006-3002(63)91815-8. [DOI] [PubMed] [Google Scholar]
- CRANE R. K., SOLS A. The non-competitive inhibition of brain hexokinase by glucose-6-phosphate and related compounds. J Biol Chem. 1954 Oct;210(2):597–606. [PubMed] [Google Scholar]
- FRIEDEN C. Glutamic dehydrogenase. III. The order of substrate addition in the enzymatic reaction. J Biol Chem. 1959 Nov;234:2891–2896. [PubMed] [Google Scholar]
- FROMM H. J., SILVERSTEIN E., BOYER P. D. EQUILIBRIUM AND NET REACTION RATES IN RELATION TO THE MECHANISM OF YEAST HEXOKINASE. J Biol Chem. 1964 Nov;239:3645–3652. [PubMed] [Google Scholar]
- FROMM H. J., ZEWE V. Kinetic studies of the brain hexokinase reaction. J Biol Chem. 1962 May;237:1661–1667. [PubMed] [Google Scholar]
- FROMM H. J., ZEWE V. Kinetic studies of yeast hexokinase. J Biol Chem. 1962 Oct;237:3027–3032. [PubMed] [Google Scholar]
- Hanson T. L., Fromm H. J. Rat skeletal muscle hexokinase. I. Kinetics and reaction mechanism. J Biol Chem. 1965 Nov;240(11):4133–4139. [PubMed] [Google Scholar]
- KERLY M., LEABACK D. H. The characteristics of hexokinase from Locusta migratoria muscle. Biochem J. 1957 Oct;67(2):245–250. doi: 10.1042/bj0670245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LIEBECQ C. The hexokinase activity of rat muscle extract and the lability of the Mg-ATP complex. Biochem J. 1953 Jun;54(3):xxii–xxiii. [PubMed] [Google Scholar]
- NOVOA W. B., WINER A. D., GLAID A. J., SCHWERT G. W. Lactic dehydrogenase. V. Inhibition by oxamate and by oxalate. J Biol Chem. 1959 May;234(5):1143–1148. [PubMed] [Google Scholar]
- SALAS J., SALAS M., VINUELA E., SOLS A. GLUCOKINASE OF RABBIT LIVER. J Biol Chem. 1965 Mar;240:1014–1018. [PubMed] [Google Scholar]
- SLEIN M. W., CORI G. T., CORI C. F. A comparative study of hexokinase from yeast and animal tissues. J Biol Chem. 1950 Oct;186(2):763–780. [PubMed] [Google Scholar]
- WEIL-MALHERBE H., BONE A. D. Studies on hexokinase. 1. The hexokinase activity of rat-brain extracts. Biochem J. 1951 Aug;49(3):339–347. doi: 10.1042/bj0490339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WRATTEN C. C., CLELAND W. W. PRODUCT INHIBITION STUDIES ON YEAST AND LIVER ALCOHOL DEHYDROGENASES. Biochemistry. 1963 Sep-Oct;2:935–941. doi: 10.1021/bi00905a007. [DOI] [PubMed] [Google Scholar]


