Abstract
Regulated growth and cell shape control are fundamentally important to the function of plant cells, tissues, and organs. The signal transduction cascades that control localized growth and cell shape, however, are not known. To better understand the relationship between cytoskeletal organization, organelle positioning, and regulated vesicle transport, we conducted a forward genetic screen to identify genes that regulate cytoskeletal organization in plants. Because of the distinct requirements for microtubules and actin filaments during leaf trichome development, a trichome-based morphology screen is an efficient approach to identify genes that affect cytoplasmic organization. The seedling lethal spike1 mutant was identified based on trichome, cotyledon, and leaf-shape defects. The predicted SPIKE1 protein shares amino acid identity with a large family of adapter proteins present in humans, flies, and worms that integrate extracellular signals with cytoskeletal reorganization. Both the trichome phenotype and immunolocalization data suggest that SPIKE1 also is involved in cytoskeletal reorganization. The assembly of laterally clustered foci of microtubules and polarized growth are early events in cotyledon development, and both processes are misregulated in spike1 epidermal cells.
INTRODUCTION
In multicellular organisms, specialized cytoplasmic organization and cell shape underlie the unique functions of cells, tissues, and organs. It is reasonable to propose that the dynamic properties of the microtubule and actin cytoskeletons and the proteins that bind them underlie much of the observed asymmetry in plant cells. During leaf and root trichoblast morphogenesis, organized actin filaments and microtubules are required (Bibikova et al., 1999; Mathur et al., 1999; Szymanski et al., 1999; Baluska et al., 2000). We are using the diagnostic shape defects of cytoskeleton-disrupted leaf trichomes to guide mutant screens for essential genes that participate in cytoskeletal reorganization and morphogenesis.
Decades of cytological and biochemical research have demonstrated the importance of the actin filament and microtubule cytoskeletons during plant morphogenesis (for reviews of the interphase microtubule and actin cytoskeletons in plant cells, see Giddings and Staehelin, 1991; Cyr, 1994; Staiger, 2000). In most plant cell types, the interphase microtubule array is cortical. The most commonly cited function of the cortical microtubule array is the regulation of the alignment of newly synthesized cellulose microfibrils. However, there are cases in which microtubules and cellu-lose microfibrils are not coaligned (Baskin et al., 1999; Wasteneys, 2000). The interphase actin filament cytoskeleton is composed of bundled transvacuolar filaments, nucleus-associated filaments, and cortical actin filaments (Traas et al., 1987). Actin filaments provide both a scaffolding for the relatively immobile network of the endoplasmic reticulum and tracks for the rapid intracellular transport of Golgi stacks (Satiat-Jeunemaitre and Hawes, 1996; Boevink et al., 1998; Nebenfuhr et al., 1999). In growing cells, the actin cytoskeleton is not a static structure. Fine and dynamic cortical actin filaments may define regions of high rates of exocytosis and growth. Several studies have correlated the presence of fine cortical actin filaments with subcellular regions of localized growth (Thimann et al., 1992; Waller and Nick, 1997; Gibbon et al., 1999; Szymanski et al., 1999; Fu et al., 2001).
In the context of polarized growth, the roles of the actin and microtubule cytoskeletons vary depending on the species and cell type. An unperturbed F-actin cytoskeleton is required for the establishment of polarity in Fucus and Pelvetia embryos (Quatrano, 1973; Alessa and Kropf, 1999). Actin filaments also are the primary cytoskeletal determinant of polarized tip growth in pollen tubes (Mascarenhas and LaFountain, 1972; Heslop-Harrison et al., 1986; Gibbon et al., 1999; Fu et al., 2001). Much of leaf and cotton trichome growth is caused by polarized diffuse growth, and the resulting pharmacological sensitivities are quite different from those of pollen tubes. In general, microtubule-disrupting drugs block the initiation of polarized growth, and actin filament inhibitors mainly affect the maintenance of polarized growth (Tiwari and Wilkins, 1995; Mathur et al., 1999; Szymanski, 2000). The requirements for both microtubules and actin filaments also have been examined during the polarized growth of other leaf cell types. Lateral microtubule association precedes localized secondary wall formation in developing Zinnia tracheary elements and may be sufficient to localize secondary wall formation (Falconer and Seagull, 1985b). Drugs that disrupt microtubules block localized secondary wall formation, whereas those that destabilize actin filaments do not (Kobayashi et al., 1988). Wheat mesophyll cells also are highly polarized, lobed cells. Constricted regions of the cell correspond to locations of increased wall thickness (Jung and Wernicke, 1991). Pharmacological and localization experiments suggest that lateral microtubule clustering is an early essential event during wheat mesophyll cell lobe initiation (Wernicke and Jung, 1992). Lateral microtubule clustering seems to be a general mechanism for polarized growth in leaf cells with a lobed morphology (Apostolakos et al., 1991; Panteris et al., 1993a, 1994). Although a class of candidate microtubule-associated proteins that form lateral cross-links has been identified in plants, their in vivo function is not known (Chan et al., 1999; Smertenko et al., 2000).
A forward genetic analysis of cytoskeleton-based functions in Arabidopsis is a feasible approach to understand how plants have solved the problem of regulated cytoskeletal organization during polarized morphogenesis. For example, the reduced trichome branch phenotypes of zwichel (zwi) and fragile fiber2 (fra2) phenocopy the effects of microtubule-disrupting, but not actin filament–disrupting, agents (Oppenheimer et al., 1997; Mathur et al., 1999; Szymanski et al., 1999; Burk et al., 2001). The ZWI and FRA2 gene products share amino acid sequence identity with the known microtubule binding proteins kinesin and katanin, respectively. In the case of ZWI (also known as KCBP), minus end–directed motor activity has been determined experimentally (Song et al., 1997). The “distorted” class of trichome mutants appears to be affected in actin-dependent processes (Szymanski, 2000). Clearly, a molecular genetic approach will provide important functional information about the plant cytoskeleton.
Our objective is to use genetic screens based on trichome shape to identify additional genes that affect cell signaling, cytoskeletal reorganization, and polarized growth. Unlike most of the reduced trichome branch mutants reported to date, the spike1 (spk1) mutant displays defects in polarized growth in all cotyledon and leaf epidermal cell types as well as seedling viability. The SPK1 gene encodes a 207-kD modular protein that shares amino acid identity with the CDM family of adapter proteins present in humans, flies, and worms (Hasegawa et al., 1996; Nolan et al., 1998; Wu and Horvitz, 1998). CDM proteins are thought to integrate extracellular signals and cytoskeletal reorganization. Consistent with the notion that a cytoskeletal defect is associated with the spk1 phenotype, localized lateral microtubule clustering, which usually precedes lobe formation in developing epidermal pavement cells, is not observed in the mutant.
RESULTS
Characterization of the spk1 Phenotype
To improve our chances of finding genes that affect cytoskeletal organization during seedling development, we used the diagnostic cell shape defects of trichomes to suggest whether microtubule- or actin filament–dependent activities may be affected (Szymanski, 2000). After family screens of pooled lines of T-DNA–mutagenized plants, the seedling lethal spk1-1 allele was recovered in a heterozygous plant. In segregating F2 populations, spk1-1 behaved as a simple recessive mutation (∼3 wild-type:1 mutant). spk1-1 displayed a reduction in trichome branching and stalk elongation: its phenotype was similar to that of microtubule-disrupted cells and the trichomes on zwi and fra2 leaves. At 30 to 50% humidity, the spk1-1 allele caused seedling lethality of soil-grown plants, usually within 2 to 3 days after germination. When grown at 100% humidity, soil-grown spk1-1 plants survived for longer periods of time but were dwarfed compared with those of the wild type, and the cotyledons and leaves of spk1-1 were very narrow (Figure 1A). When cultured on agar-based media, spk1-1 fresh weight was similar to that of the wild type after 12 days of growth, but the organ shape and trichome defects persisted (Figures 1B and 1C). Although an inflorescence formed occasionally, the mutant plants were completely sterile. The sterility defect was sporophytic, because in reciprocal crosses the mutant allele was transmitted efficiently through both the male and female gametophytes of heterozygous plants.
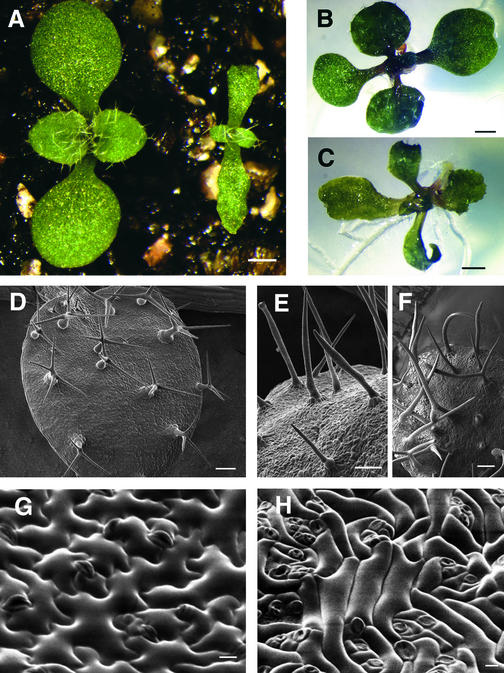
Figure 1.
Arabidopsis Wild-Type and spk1-1 Seedling Phenotypes Grown under Different Conditions.
(A) One-week-old seedlings grown on soil at 100% humidity. The wild type is at left, spk1-1 mutant at right.
(B) Wild-type seedling at 9 DAG on agar-based medium.
(C) spk1-1 seedling at 9 DAG on agar-based medium.
(D) Scanning electron microscopy of wild-type leaf trichomes.
(E) Scanning electron microscopy of spk1-1 leaf trichomes with aborted branches.
(F) Scanning electron microscopy of spk1-1 leaf trichomes with two branches.
(G) Leaf epidermal pavement cells of the wild type.
(H) Leaf epidermal pavement cells of spk1-1.
Bar in (A) = 0.5 mm; bars in (B) and (C) = 1 mm; bars in (D) to (F) = 100 μm; bars in (G) and (H) = 10 μm.
Scanning electron micrographs of the normal and mutant leaf adaxial epidermal phenotypes are shown in Figures 1D to 1H. Compared with the normal trichomes of the wild type with three branches (Figure 1D), those of spk1-1 leaves displayed two types of aberrant morphologies. Type 1 trichomes were highly elongated, unbranched, and often abnormally swollen or blebbed at various positions along the stalk (Figure 1E). Type 2 spk1-1 trichomes had a short stalk and two branches roughly parallel to the leaf surface (Figure 1F). The wild-type leaf epidermis is composed of a two-dimensional array of interdigitated lobed pavement cells (Figure 1G). The pavement cells of spk1-1 leaves were simple in shape; lobes were either absent or severely reduced in size, and most cell expansion occurred along a single axis (Figure 1H).
Given the interesting cell shape defects of the spk1-1 mutant, an experimental system was developed to examine the cell and tissue level functions of SPK1. We focused on coty-ledon epidermal development for several reasons. First, postgermination cotyledon growth is the first recognizable spk1-1 phenotype in the shoot, and therefore it is the stage at which we are most likely to identify the direct effects of the mutation. Second, cotyledon epidermal cell growth is both highly polarized and synchronous, and it involves the coordinated growth of a population of cells. Third, we have sensitive methods to analyze the organization of microtubules and actin filaments in epidermal cells at several developmental stages. Our current inability to visualize the cytoskeletons of intact spk1-1 trichomes has hindered our studies of SPK1 function in this cell type.
The shape of the cotyledon was affected severely in the spk1-1 mutant. At 2 days after germination (DAG), wild-type cotyledons were roughly circular (Figure 2A). Their epidermal pavement cell shapes were variable; some cells initiated several lobes, whereas others had a simple shape with either no clear indication of polarized growth or a rudimentary bulge within the plane of the epidermis (Figure 2B). In this article, the extended regions of the lobe are referred to as protrusions and the lateral flanks of the lobe are referred to as indentations. At 1 DAG, spk1-1 cotyledon shape was indistinguishable from that of the wild type, but at 2 DAG, cotyledons were slightly more narrow (Figures 2E and 3, Table 1). Some of the spk1-1 epidermal cells were slightly irregular in shape, and there was no clear indication of lobe initiation along the cell perimeter (Figure 2F). At 5 DAG, wild-type cotyledons retained a circular shape, and almost all cells contained multiple, well-elaborated protrusions (Figures 2C and 2D). At the same time, the spk1-1 cotyledons were narrow, and the epidermal cells lacked protrusions and were quite small compared with those of the wild type (Figures 2G and 2H, Table 1). Gaps were evident between mutant epidermal cells (Figure 2H). At 8 to 10 DAG, the cotyledon continued to expand, and the defects in cell-to-cell adhesion became quite severe. These large gaps in the epidermis explain the developmental stage–specific seedling lethality of the mutant at low humidity. A thorough analysis of the carbohydrate composition and ultrastructure of cell walls of mutant and wild-type plants failed to reveal any significant differences (D. Szymanski and N. Carpita, unpublished results).
Figure 2.
Cell and Organ Shape Defects during spk1-1 Cotyledon Development.
(A) to (D) Scanning electron microscopy of the adaxial surface of wild-type cotyledons.
(A) Wild-type cotyledon at 2 DAG.
(B) Higher magnification of wild-type cotyledon epidermal cells at 2 DAG.
(C) Wild-type cotyledon at 5 DAG.
(D) Higher magnification of wild-type cotyledon epidermal cells at 5 DAG.
(E) to (H) Scanning electron microscopy of the adaxial surface of spk1-1 cotyledons.
(E) spk1-1 cotyledon at 2 DAG.
(F) Higher magnification of spk1-1 cotyledon epidermal cells at 2 DAG.
(G) spk1-1 cotyledon at 5 DAG.
(H) Higher magnification of spk1-1 cotyledon epidermal cells at 5 DAG.
Bars in (A), (C), (E), and (G) = 100 μm; bars in (B), (D), (F), and (H) = 10 μm.
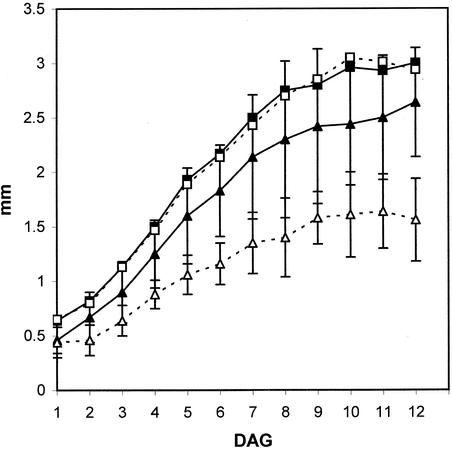
Figure 3.
Growth Rates of Wild-Type and spk1-1 Cotyledons.
Maximum cotyledon lengths and widths were measured from 1 to 12 DAG. Closed squares, wild-type cotyledon length; open squares, spk1-1 cotyledon length; closed triangles, wild-type cotyledon width; open triangles, spk1-1 cotyledon width. Error bars indicate ±sd × 1.96 to reflect a 95% confidence interval for significance.
Table 1.
Cotyledon Size and Cell Number of spk1 and Wild-Type Cotyledons at Different Times
| 2 DAG
|
5 DAG
|
|||
|---|---|---|---|---|
| Size and Number | spk1a | Wild Type | spk1 | Wild Type |
| Length (mm)b | 0.80 ± 0.26 | 0.82 ± 0.29 | 1.89 ± 0.57 | 1.93 ± 0.57 |
| Width (mm)b | 0.47 ± 0.14 | 0.67 ± 0.24 | 1.07 ± 0.18 | 1.60 ± 0.45 |
| Area (mm2)c | 0.25 ± 0.08 | 0.35 ± 0.08 | 1.37 ± 0.55 | 2.20 ± 0.73 |
| Cell Numberd | 456 ± 151 | 382 ± 161 | 715 ± 174 | 409 ± 82 |
a All measurements are expressed as the mean ±1.96 × sd, which defines the 95% confidence level for significance.
Maximum length and width values were measured for each cotyledon.
Adaxial surface area.
Measurements were taken from scanning electron microscopy images of the adaxial epidermis. Cell number is total cotyledon area/mean cell area (see Methods).
To determine if spk1-1 differentially affected cotyledon length and width, we measured the cotyledon shape of wild-type and spk1-1 cotyledons. At the earliest time point (1 DAG), the mean length and width measurements of wild-type and mutant cotyledons were nearly identical (Figure 3). In addition, the duration and rate of cotyledon elongation of the mutant were not appreciably different from those in the wild type (Figure 3, Table 1). However, the lateral growth rate of spk1-1 cotyledons was reduced. Qualitative differences in cotyledon diameter were apparent at 2 DAG and were statistically significant at 12 DAG (Figure 3, Table 1). The mean cell surface area was reduced 29 and 38% in 2- and 5-DAG mutant cotyledons, respectively (Table 1).
The contribution of cell number and cell size to the spk1-1 cotyledon shape defects also was determined. Consistent with the observation that very little cell division occurs during Arabidopsis cotyledon development (Tsukaya et al., 1994), the mean cotyledon cell number in the wild type increased from 382 cells at 2 DAG to 409 cells at 5 DAG (Table 1). Although this is not a substantial difference, using scanning electron microscopy, we routinely observed recently divided cells in wild-type cotyledons from 5 to 8 DAG. At 2 DAG, the spk1-1 mean cotyledon cell number was not significantly different from that of the wild type, although some individual spk1-1 cotyledons had increased cell numbers that were not observed in the wild type. At 5 DAG, the spk1-1 epidermis contained 75% more cells than the wild type (Table 1). Within the midblade clusters of dividing cells, neither the cell division planes nor the direction of cell expansion was oriented with respect to the long axis of the leaf (Figures 2G and 2H). Therefore, the additional cells in spk1-1 cotyledons do not contribute preferentially to cotyledon elongation. In most spk1-1 cotyledons, a subset of epidermal cells near the tip and along the perimeter expanded preferentially along the long axis of the cotyledon (Figures 2E and 2G).
Cloning the SPK1 Gene
To initiate a functional analysis of the control of cell shape in the epidermis, we cloned the SPK1 gene. In a segregating population, we failed to detect any recombination between the kanamycin resistance gene (Knr) of the T-DNA element and the spk1-1 mutation (n = 88). Therefore, the spk1-1 allele is within 1.2 centimorgans of the T-DNA insertion. Sequence data from the left border of the T-DNA located the insertion to the 18th intron of a large open reading frame on chromosome 4 (Figure 4A). DNA gel blots of genomic DNA from wild-type and mutant plants confirmed that the flanking genomic sequences are linked to the T-DNA and defined an 81-bp interval within which the effects of the insertion were limited (data not shown). We used a polymerase chain reaction (PCR)–based reverse genetic approach to identify an independent heterozygous line (spk1-2) that segregated ∼3:1 for both Knr and the spk1 phenotype (McKinney et al., 1995; Krysan et al., 1996). The mutation was linked to the insertion; of 102 spk1-2 plants examined, all harbored the selectable marker associated with the T-DNA. Sequence information from the plant DNA that flanked the left border of the T-DNA located the insertion to the seventh intron of the SPK1 gene (Figure 4A). The gross cotyledon, leaf, and trichome phenotypes of the spk1-1 and spk1-2 alleles were indistinguishable (data not shown).
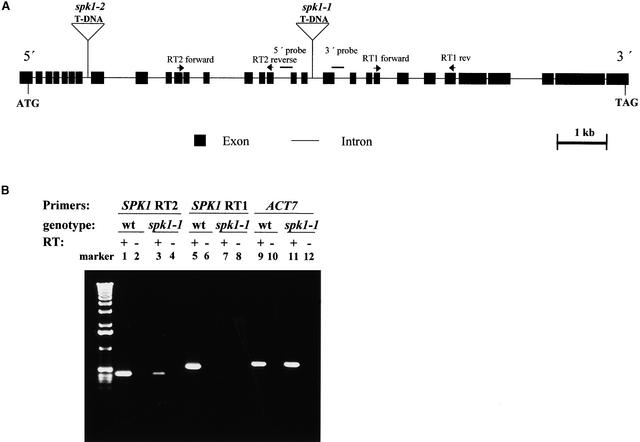
Figure 4.
Structure of the SPK1 Gene and Nature of spk1 Mutant Alleles.
(A) Exon and intron organization of the SPK1 gene. The positions and lengths of exons and introns are indicated by closed rectangles and lines, respectively. The positions of T-DNA inserts associated with spk1-1 and spk1-2 are shown above the gene diagram.
(B) Altered expression of the SPK1 gene in spk1-1 plants. RT-PCR analysis shows SPK1 mRNA accumulation in spk1-1 and the wild type. The locations of the upstream (SPK1 RT2) and downstream (SPK1 RT1) primer pairs are labeled. Gene-specific primers for the ACT7 gene were used as a control. (+), with reverse transcriptase (RT); (−), without reverse transcriptase.
If the insertions into the SPK1 gene cause the mutant phenotype, then the expression of the gene may be altered in mutant plants. To address this question, we assayed SPK1 gene expression in mutant and wild-type plants using reverse transcription (RT)–PCR. SPK1-specific primers upstream (Figure 4B, lanes 1 to 4) and downstream (Figure 4B, lanes 5 to 8) from the spk1-1 T-DNA insertion detected SPK1 message in wild-type plants (lanes 1 and 5). In all cases, the signal was derived from mRNA because no amplified product was obtained in the absence of RT (lanes 2, 4, 6, 8, 10, and 12). In RNA samples derived from spk1-1 plants, accumulation of SPK1 mRNA was either reduced greatly (lane 3) or undetectable (lane 7). The failure to detect SPK1 mRNA was not the result of a problem with the RNA sample because the ACTIN7 (ACT7) gene was detected easily (lanes 11 and 12). SPK1 message was not detectable in the spk1-2 background using PCR primers that flanked the T-DNA insertion (data not shown).
To confirm that disruption of the SPK1 gene caused the spk1 phenotype, we transformed wild-type plants with a transgene that transcribes double-stranded RNA corresponding to the SPK1 gene. Control plants transformed with either the empty vector (Figure 5A) or a double-stranded RNA construct corresponding to an intron of SPK1 (Figure 5B) had no detectable trichome, pavement cell, or cotyledon shape phenotype. However, plants transformed with double-stranded RNA–expressing constructs corresponding to 1089 bp (DS-1; Figure 5C) and 677 bp (DS-2; Figure 5D) of the SPK1 cDNA sequence displayed all aspects of the spk1 phenotype: 88.5% of DS-1 and 93.2% of DS-2 transformed plants displayed the narrow cotyledon phenotype. For both DS-1 and DS-2 transformed lines, trichome branching was reduced noticeably in <5% of the viable T1 plants. Many of the severely affected transformed plants that displayed a trichome phenotype were seedling lethal. Together, these results prove that we identified the SPK1 gene correctly.
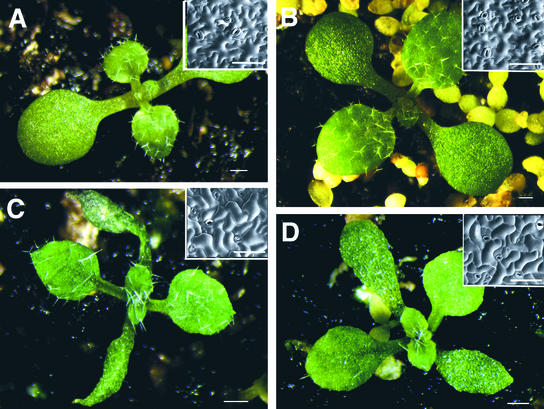
Figure 5.
Expression of Double-Stranded SPK1 RNA Causes a spk1 Phenotype.
(A) Wild-type plants transformed with the empty binary vector.
(B) Wild-type plants transformed with DSCK. DSCK expresses double-stranded RNA corresponding to the 27th intron of SPK1.
(C) Wild-type plants transformed with DS1. DS1 corresponds to 1089 bp of the SPK1 coding sequence.
(D) Wild-type plants transformed with DS2. DS2 corresponds to 677 bp of the SPK1 coding sequence.
Insets in each panel show scanning electron micrographs of the adaxial cotyledon epidermal cells of similarly staged plants. Bars = 1 mm; inset bars = 100 μm.
Full-Length SPK1 cDNA Sequence and Expression
The spk1-1 insertion event occurred within a predicted long open reading frame (ORF 4200c) on the south arm of chromosome 4 (Bevan et al., 1998). Because of an extra T in the genome sequence (see below), a deduced reading frame error occurred and the annotation of the genomic sequence did not identify a candidate start codon. RNA gel blot experiments conducted using poly(A) mRNA indicated that the SPK1 message is nearly 6 kb, twice as long as the 3.3-kb predicted coding region of ORF 4200c (data not shown). Using a 3-kb partial-length cDNA obtained from a seedling library (Kieber et al., 1993) as a starting point, one 3′ rapid amplification of cDNA ends (RACE) step and three 5′ RACE steps were required to identify both the poly(A) site and an initiation codon that contained an upstream stop codon in the SPK1 reading frame. The complete 5.8-kb cDNA sequence and the deduced amino acid sequence are available in GenBank. A scheme of the SPK1 protein and its conserved domains is shown in Figure 6. There were 30 exons and 29 introns in the SPK1 gene. All splice donor and acceptor sites conformed to observed Arabidopsis splice junction border sequences. We identified 6 bp in the full-length SPK1 cDNA that differed from the published genome sequence. We confirmed that the cDNA sequence was correct by sequencing both strands of the cosmid clone g5845 at each ambiguous base.
Figure 6.
Scheme of the SPK1 Coding Sequence.
The MOD1, MOD2, and CDMS domains are labeled to scale according to their positions in the predicted protein.
The SPK1 cDNA encodes a predicted protein of 1830 amino acids and 207 kD whose mRNA is present in all major organs (data not shown). The predicted protein contained several 21–amino acid hydrophobic regions that are candidate membrane-spanning segments (Kyte and Doolittle, 1982). However, the protein lacked an N-terminal signal sequence, and the TMHMM algorithm, which is the most accurate method for identifying membrane-spanning segments, predicted SPK1 to be a soluble protein (Krogh et al., 2001). The solubility of SPK1 in cells remains to be determined. The predicted protein contained at least three conserved regions. The most C-terminal is ∼370 amino acids long and shares at least 20% pairwise identity with the animal proteins Caenorhabditis elegans CED-5, human DOCK180, and Drosophila MYOBLAST CITY, which constitute the CDM family of proteins (Hasegawa et al., 1996; Nolan et al., 1998; Wu and Horvitz, 1998). Although the pairwise identity is not immediately impressive, nearly 25% of the conserved residues that show pairwise identity with animal proteins are identical across several phylogenetic groups (Figure 7). This region of the SPK1 protein is defined as the CED-5, DOCK180, MBC, SPK1 (CDMS) domain. N-terminal to the CDMS domain, the SPK1 gene product contained two ad-ditional conserved regions of ∼130 and 510 amino acids (Figure 6). Because these domains are found only in the genomes of multicellular organisms, we refer to the 130– and 510–amino acid domains as multicellular organism domain 1 (MOD1) and MOD2, respectively. The CDM family proteins do not encode conserved MOD1 and MOD2 domains.
Figure 7.
Alignment of the CDMS Domain of SPK1 with Homo sapiens DOCK180, Caenorhabditis elegans CED-5, Drosophila melanogaster MYOBLAST CITY (MBC), Saccharomyces cerevisiae Ylr422wp, and Dictyostelium discoideum DocA.
Amino acid residues in black indicate identity, and those in gray indicate conserved substitutions. Residues that are identical or similar in four of seven sequences are highlighted. The numerical amino acid scale for each sequence is labeled at right.
Cytoskeletal Localization in Developing Cotyledon Epidermal Cells
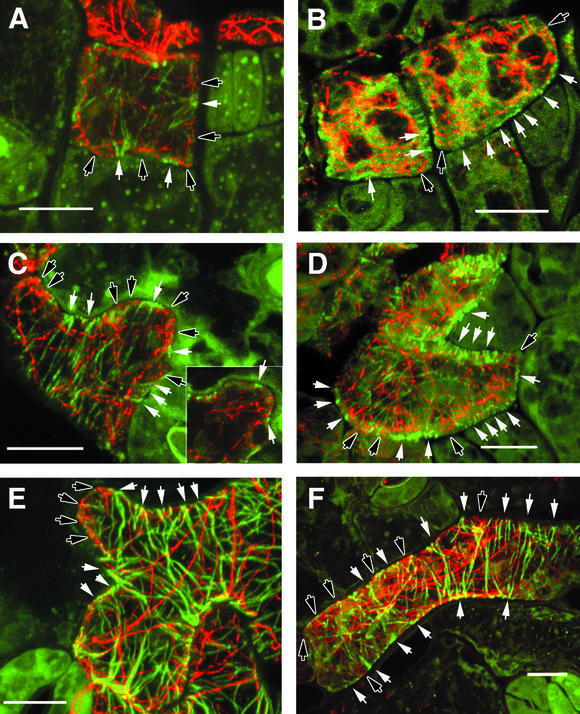
In each of the experimental systems in which the function of CDM proteins has been examined, a role in integrating extracellular information with cytoskeletal reorganization has been hypothesized. To begin to test for a role for SPK1 in cytoskeletal organization, we compared the mutant and wild-type actin and microtubule cytoskeletons in developing cotyledon epidermal cells. We double labeled actin and tubulin with antibodies at three stages of development: (1) symmetrical, cuboidal cells common in 1-DAG cotyledons; (2) cells with newly formed lobes common in 2-DAG cotyledons; and (3) cells with well-defined lobes common in 5-DAG cotyledons. Confocal images of double-labeled wild-type and mutant cells are displayed in Figure 8. Each image is a maximum projection of the tubulin (green channel) and actin (red channel) signals from the outer halves of epidermal cells. In wild-type epidermal cells that either had divided recently or had yet to initiate clear lobe-like structures, foci of laterally associated cortical microtubules were seen frequently on anticlinal walls (walls perpendicular to the leaf surface). Near the junction of the periclinal walls (walls parallel to the leaf surface) and anticlinal walls, the cortical microtubules splayed out along the cell cortex. A representative cell displaying this sort of microtubule organization is shown in Figure 8A.
Figure 8.
Immunolocalization and Confocal Micrographs of the Cytoskeleton in Developing Wild-Type and spk1-1 Cotyledon Epidermal Cells.
Epidermal cells were double labeled with antibodies directed against α-tubulin (green channel) or actin (red channel). Each image is a maximum projection of the upper half of the cells.
(A), (C), and (E) Wild-type epidermal cells.
(A) Cuboidal wild-type epidermal cell at 1 DAG.
(B) Cuboidal spk1-1 epidermal cell at 1 DAG.
(C) A wild-type epidermal cell at 2 DAG that has initiated lobe formation. The inset shows a single optical section through a plane of the transvacuolar cytoplasm.
(B), (D), and (F) spk1-1 epidermal cells.
(D) spk1-1 epidermal cell at 2 DAG.
(E) A rapidly growing wild-type epidermal cell at 5 DAG.
(F) An elongated spk1-1 epidermal cell at 5 DAG.
White arrows, regions containing cortical microtubules that are in close proximity to the plasma membrane; black arrows, regions of the cell containing actin filaments that are in close proximity to the plasma membrane. Bars = 10 μm.
At these early stages of development, the actin cytoskeleton often was distributed between association with putative provacuoles and the cell cortex. Actin filaments were excluded largely from the plasma membrane in regions of the cell in which cortical microtubules were present. In microtubule-free zones of the cell, some actin filaments were associated closely with the plasma membrane (Figure 8A). Throughout epidermal development, this predominantly nonoverlapping pattern of actin and tubulin signal is observed consistently (Figure 8). Almost all of the yellow color seen in Figure 8 is the result of compressing three-dimensional localization signal into two dimensions. However, isolated puncta of colocalized actin and tubulin within a single image plane occasionally was detected at indentations near the flanks of laterally associated microtubules. At 2 DAG, laterally associated microtubules were observed consistently in close association with the plasma membrane along cell indentations (Figure 8C). Fine actin filaments or bundles within actively growing protrusions were both cortical and transvacuolar; many terminated in very close proximity to the plasma membrane (Figure 8C). In protrusions, much of the actin signal was either diffuse or punctate, and we do not know if this is a fixation artifact or the real actin organization in vivo.
Cortical microtubules were not excluded from protrusions, and the pattern of nonoverlapping cortical microtubules and actin filaments often was observed within a growing protrusion. For example, in Figure 8C, three repeating units of alternating microtubule- and actin filament–populated plasma membrane zones are clearly defined within a single protrusion. The inset shows a single optical section that includes a plane of transvacuolar cytoplasm that is enriched in actin filaments; the reticulate network of actin bundles clearly terminates near the plasma membrane between two sets of laterally associated microtubules. At 5 DAG, laterally associated cortical microtubules populated the anticlinal wall along the indentations, but many of the tips of protrusions were dominated by actin signal (Figure 8E). Most of the actin signal in Figure 8E was derived from the cortical cytoskeleton, with very little contribution from transvacuolar strands.
The cytoskeletal organization of spk1 epidermal cells was altered at the onset of the mutant cell shape phenotype. At 1 DAG, there were no significant differences in overall organ shape (Figure 3), but at the cellular level, spk1 epidermal cells were more rounded and lacked the small protrusions that were observed along wild-type anticlinal walls (Figure 8B). Populations of actin filaments and microtubules were present in mutant cells. However, evenly distributed cortical microtubules were detected only faintly in mutant cells, and no pattern of alternating foci of laterally associated cortical microtubules and cortical actin filaments along the perimeter of the anticlinal walls was observed. Instead, putative membrane-associated actin filaments were detected most often at cell corners or at the ends of elongated cells (Figure 8B). At 2 DAG, cortical microtubules were aligned in an extended radial pattern and often formed evenly spaced circumferential hoops around elongated cells (Figure 8D). The absence of regions of highly clustered microtubules at 2 DAG was not caused by a complete lack of lateral cross-linking activity, because pairs of radial microtubules could be found in electron micrographs taken from ultrathin paradermal sections (D.B. Szymanski, unpublished results). However, distinct foci of laterally associated microtubules were not detected along the anticlinal walls of mutant cells during early developmental stages. In expanding mutant cells at 2 and 5 DAG, cortical actin filaments in close proximity to the plasma membrane often were detected at seemingly random positions along the perimeter of the anticlinal cell wall, but their cortical localization was not correlated with any particular cell shape (Figures 8D and 8F).
DISCUSSION
Forward genetic approaches using Arabidopsis will be extremely useful for understanding the signaling pathways that affect cytoskeletal reorganization during plant development (Oppenheimer et al., 1997; Burk et al., 2001; Whittington et al., 2001). Our long-term goal is to understand how the organization of the cytoskeleton is regulated and its function during epidermal cell and tissue morphogenesis. To achieve this goal, we have initiated a functional analysis of SPK1. The ubiquitously expressed SPK1 gene described in this article is required for normal cytoskeletal organization, polarized cell expansion, and tissue development in the leaf and cotyledon.
A Complex Cotyledon spk1 Phenotype
At 1 DAG, the organ shape and cell number of the mutant were not appreciably different from those of the wild type. However, at 2 DAG, the trend toward decreased cell size, decreased cotyledon width, and increased cell number was apparent (Table 1). It is possible that SPK1 has multiple functions in the cell, but we suspect that some of these pleiotropic cotyledon phenotypes are secondary effects of the failure to organize the cytoplasm in spk1 cells properly at a previous stage of development. For example, the adhesion defects between mutant epidermal cells commonly seen between 2 and 5 DAG also are observed in regions of high mechanical strain, such as leaf margins and the apical hook of dark-grown hypocotyls (data not shown). It is possible that both the reduction in the interdigitation of the unlobed cells and subtle wall defects contribute to the widespread failure of spk1 epidermal cells to withstand the forces of organ expansion. The increased cell number phenotype also may be a secondary response to defective cell expansion or adhesion. The ability of adjacent cells to “sense” and compensate for the reduced growth of slow-growing sectors has been observed in maize leaves (Freeling, 1992). In any case, increased cell division is not sufficient to restore wild-type width to spk1 cotyledons. It is not clear why cotyledon width is affected more severely than length. Similar defects in cotyledon shape and trichome branching have been reported for the angustifolia (an) mutant (Rédei, 1962; Hülskamp et al., 1994). It has been proposed that genetically distinct pathways control cotyledon elongation and lateral expansion (Tsuge et al., 1996). In the spk1 background, the elongation of a subpopulation of cells along the cotyledon margin and near the tip may play a major role in determining organ length.
SPK1 Gene Product
A single copy of the SPK1 gene is present in the Arabidopsis genome. It is a large gene consisting of 30 exons that span 12.7 kb of genomic DNA. The reported 5.8-kb cDNA sequence agrees well with the size of the SPK1 mRNA detected in RNA gel blots analysis. The predicted protein contains a conserved CDMS domain at the C terminus that provides some clues to its function. The known CDMS-encoding gene family members in animal cells are hypothesized adapter proteins that mediate cytoskeletal reorganization in response to diverse extracellular signals (Hasegawa et al., 1996; Erickson et al., 1997). For each of the CDM family members, there is genetic and biochemical evidence for the involvement of RAC GTPases during cell signaling (Kiyokawa et al., 1998; Nolan et al., 1998; Reddien and Horvitz, 2000). RAC GTPases are nucleotide-gated switches that affect the cytoskeleton indirectly by modulating the activity of effector proteins. In the case of human DOCK180, the founding member of the family, the CDMS domain is required to coimmunoprecipitate DOCK180 and RAC in vivo, but it is not known if the interaction is direct (Kobayashi et al., 2001). Mutations within the CDMS domains of MBC and CED5 cause severe loss-of-function phenotypes (Erickson et al., 1997; Wu and Horvitz, 1998). The details of how the proposed RAC-CDM protein complex regulates cytoskeletal organization are not known. In Arabidopsis, there are 11 AtROP genes whose encoded proteins share a high level of amino acid sequence identity with animal RAC gene products (Li and Yang, 2000). Perhaps AtROPs are involved in SPK1-dependent signaling processes.
It is unlikely that the CDMS domain of SPK1 is the only region of functional significance. There are at least two additional regions in the protein that are highly conserved in a large family of animal genes with no known function. Many of the MOD1- and MOD2-encoding genes also encode a C-terminal CDMS domain, but the overall domain organization of this class of genes is diverse. For example, in the current database of full-length gene sequences, four of the seven possible combinations of MOD1, MOD2, and CDMS are present. One Drosophila gene and one C. elegans gene encode ordered MOD1, MOD2, and CDMS domains. Perhaps these conserved regions are additional domains within SPK1 that either recruit additional proteins and/or autoregulate the activity of other domains.
Cytoskeletal Basis of the Epidermal Shape Defects
Based on the reduced trichome branching phenotype and the observed differences in lateral microtubule association at the early stages of pavement cell development, it is possible that SPK1 regulates the organization of the interphase microtubule array. In differentiating cells, the postmitotic cortical microtubule array is organized randomly at first; however, depending on the cell type, it eventually adopts a more ordered symmetrical pattern (Flanders et al., 1990; Granger and Cyr, 2000; Bichet et al., 2001). In Arabidopsis, the transition to an ordered interphase microtubule array is a crucially important step in regulating cell and tissue organization (Bichet et al., 2001; Burk et al., 2001). In the leaf, microtubule clustering and localized wall deposition are evolutionarily conserved processes in many leaf cell types that display a lobed cell morphology (Panteris et al., 1994; Wasteneys et al., 1997). In many cases, microtubule bundling is an early differentiation event that leads to localized cell wall synthesis (Falconer and Seagull, 1985a; Kobayashi et al., 1988; Wernicke and Jung, 1992; Panteris et al., 1993b). In Vigna sinensis, the deposition of newly synthesized wall material in the leaf epidermal cells is aligned with clustered microtubules and is concentrated near the junction of the anticlinal and periclinal walls (Panteris et al., 1993b). This region of the cell, in which the transition from laterally associated to isolated microtubules at the outer periclinal membrane occurs, may define specialized domains for the motor-dependent transport of vesicles and cellulose synthase complexes.
At all stages of pavement cell morphogenesis examined, actin filaments are largely excluded from the plasma membrane in regions of the cell where clustered microtubules are present. During polarized tracheary element and wheat mesophyll cell morphogenesis, inhibitor and localization studies suggest that microtubule clustering precedes cortical actin filament deposition (Kobayashi et al., 1988; Jung and Wernicke, 1991; Wernicke and Jung, 1992). Although there certainly are other possibilities, the simplest explanation is that cortical microtubule clustering occurs first and locally excludes actin filament–associated structures from the plasma membrane. By default, cortical actin filaments and bundles would be capable of populating plasma membrane domains in which clustered microtubules are absent. Although nonoverlapping in space, the organization of the microtubule and actin filament cytoskeletons may be interdependent. For example, the extremely fine actin filaments and punctate signal detected in epidermal lobes may have unique functional properties and may be required to maintain localized regions of laterally associated microtubules during development. Therefore, at this time, we cannot exclude the possibility that SPK1 affects the organization of the actin cytoskeleton and that the microtubule phenotype is a secondary effect.
If microtubule clustering is the first event in organizing the cytoplasm during pavement cell morphogenesis, the means by which this activity is regulated is not known. Certainly, in the well-defined lobes of Arabidopsis, microtubule clustering corresponds to cell indentations (Figure 8E) (Wasteneys et al., 1997). At early stages of pavement cell development, microtubule clustering often is correlated with the presence of a small indentation (Figure 8A). The protrusions of neighboring cells may initiate the morphogenesis process. Either a slow-acting force or a specific cell wall component could contain the spatial information for microtubule clustering. A mechanism for repetitive lobe initiation events must be considered: as the pavement cells expand, new lobes are formed, presumably through an iterative process. Although the lobes of wild-type cells are enriched for actin-containing structures, microtubules also are present (Figures 8C and 8E). Perhaps as the protrusion expands, several cortical microtubules invade the lobe and become stabilized laterally along the anticlinal wall. Alternatively, an actin-containing membrane-associated cortical complex may recruit microtubules or promote microtubule polymerization.
Lobe formation does not occur in spk1 epidermal cells, and foci of laterally associated microtubules along the anticlinal walls are not observed at early developmental stages. Cortical microtubules are present, but most often they adopt an extended radial arrangement perpendicular to the long axis of the cell. Pairs of laterally associated cortical microtubules are observed in ultrathin sections of both wild-type and spk1 pavement cells (D.B. Szymanski, unpublished results). Therefore, it is more likely that the position, timing, or extent of lateral association is affected in the mutant. Actin filaments, many of them cortical, also are widespread in developing spk1 epidermal cells at 1 and 2 DAG. Although the organization of the actin cytoskeleton is similar in mutant and wild-type cells, the location of cortical actin filaments along spk1-1 anticlinal walls does not reflect cell shape (Figures 8D and 8E). It appears that actin filament access to the plasma membrane is not sufficient for localized cell expansion in spk1 epidermal cells.
Several pieces of circumstantial evidence lead us to suspect that SPK1 is involved in cytoskeletal reorganization. However, at this time, we do not understand how SPK1 participates in this process. Current experiments are focused on understanding how far SPK1 function is removed from cytoskeletal reorganization. If AtROP binding and microtubule organization are primary effects of SPK1 signaling, this differs from the commonly observed effects of RAC on the actin cytoskeleton. Almost all publications in the animal and plant literature cite the actin cytoskeleton as the downstream target of RAC-like GTPase signaling events (reviewed by Hall, 1998; Li and Yang, 2000). However, dominant negative forms of RAC affect both the microtubule and actin filament cytoskeletons during Drosophila wing development (Eaton et al., 1996; Turner and Adler, 1998). Furthermore, RAC-dependent signaling affects the microtubule-destabilizing activity of stathmin (Daub et al., 2001). Considering the unique roles of actin and microtubules in plant cells and the presence of only a single family of RAC-like GTPases, AtROP-dependent signaling in Arabidopsis may not conform strictly to commonly observed functional boundaries.
Conclusion
The SPK1 gene is a regulator of cell shape and tissue organization in plant cells. Both the trichome phenotype and the defects of lateral microtubule association during the early stages of pavement cell development suggest that SPK1-dependent processes alter the microtubule cytoskeleton. SPK1 encodes the conserved CDMS domain, an essential region of the animal CDM family of adapter proteins. SPK1 contains additional regions of conserved amino acid sequence that may regulate the localization or activity of SPK1-containing complexes. Our working hypothesis is that SPK1 integrates developmental cues with cytoskeletal rearrangement during cell and tissue development. If the primary consequence of SPK1-dependent signaling is cytoskeletal reorganization, much work remains to elucidate the control mechanisms of this important signal transduction pathway.
METHODS
Plant Materials and Growth Conditions
For all phenotypic characterizations, Arabidopsis thaliana ecotype Columbia seed were grown as described by Szymanski et al. (1999). For preparation of genomic DNA and RNA from spk1 plants, seed were germinated and grown in liquid medium containing Murashige and Skoog (1962) salts and vitamin mixture (Gibco BRL, Gaithersburg, MD) with 1% sucrose with shaking (150 to ∼200 rpm) at room temperature under continuous illumination at 23°C.
Microscopy
Low-magnification light micrographs of plants were obtained using a Leica MZ12.5 stereomicroscope (Leica Microsystems, Heerbrugg, Switzerland) with a SPOT RT camera (Diagnostic Instruments, Sterling Heights, MI). Confocal images were collected using an MRC Bio-Rad 1024 confocal microscope mounted on an inverted Optiphot-2 microscope and a ×60 plan apo 1.4 numerical aperture objective (Nikon, Tokyo, Japan). Fluorescein isothiocyanate fluorescence was detected with excitation at 488 nm and a 522 DF 32 bandpass filter. Rhodamine fluorescence was detected with excitation at 568 nm and a 585 longpass filter. In most cases, both channels were imaged simultaneously, and 14% of the fluorescein isothiocyanate channel was subtracted from the rhodamine channel to remove bleed- through signal. For scanning electron microscopy, samples were frozen by plunging them into liquid N2. They were transferred to the cryo prechamber (GATAN, Inc., Pleasanton, CA) and sputter coated with gold for 4 sec at approximately −160°C. Samples were transferred to the main cryo stage and imaged at −140°C in a JEOL JSM-840 scanning electron microscope using 4- or 5-kV accelerating voltage.
Cytoskeleton Localization
The fixation and localization protocols used here closely followed the procedures described by Szymanski et al. (1999). Briefly, cotyledons were dissected and immersed rapidly in microtubule stabilization buffer (100 mM Pipes-KOH, pH 6.9, 5 mM EGTA, 2 mM MgCl2, and 0.05% Triton X-100) supplemented with 2% formaldehyde and 0.5% glutaraldehyde for 60 min. The cell walls of fixed tissue were cracked using the freeze shattering procedure (Wasteneys et al., 1997). Samples then were extracted in PBS containing 1% Triton X-100 and 25 mM glycine. For immunodetection, monoclonal clone DM1A (Sigma, St. Louis, MO) IgG1 directed against chicken brain tubulin was used at a 1:100 dilution. Monoclonal clone JLA20 (Oncogene Research Products, Boston, MA) IgM directed against chicken gizzard actin was used at a 1:100 dilution. Conjugated secondary antibodies and F(ab′)2 fragments were obtained from Jackson Immunochemicals (West Grove, PA).
Cotyledon Measurements
Cotyledons were dissected from seedlings to remove the petiole. Cotyledons were placed in a drop of water and mounted on a glass slide. Calibrated digital images were imported into NIH Image 1.61 (National Institutes of Health, Bethesda, MD; ftp://codon.nih.gov/pub/nih-image/). The cotyledon length, width, and area were measured according to the magnification. Adaxial epidermal cell numbers were counted using scanning electron microscopy images. Cell number was estimated by dividing the total cotyledon area by the mean cell area. Total cotyledon area was measured at low magnification. Single-cell area was estimated by measuring the number of cells within three or four different subregions of known area within the same cotyledon. Four different cotyledons of each sample were counted. The accuracy of measurement was confirmed by hand counting all of the cells in three different cotyledons at 2 days after germination (DAG).
DNA and RNA Analyses
DNA and RNA gel blot analyses were performed according to standard protocols (Ausubel et al., 1994) except for RNA gel blotting, for which ULTRAhyb (Ambion, Austin, TX) was used for hybridization. For DNA gel blotting, DNA was hybridized with digoxigenin-labeled probe (DIG Prime DNA Labeling and Detection Starter Kit II; Roche). RNA was isolated using GTC buffer (Newman et al., 1993). Poly(A)+ RNA was purified with Oligotex (Qiagen, Valencia, CA). For reverse transcriptase–mediated polymerase chain reaction (RT-PCR), 100 ng of mRNA was reverse transcribed with 500 ng of random hexanucleotides (Random Primer6; New England Biolabs, Beverly, MA) and SuperScript II RNase H Reverse Transcriptase (Gibco BRL). Synthesized cDNA was purified with the Qiagen PCR Purification Kit. The PCR primers for amplifying SPK1 were RT1 (5′-CGGTACCAG-AAAGCTGAAGATAA-3′; 5′-CCCAAAGTTCCAGTTTTTGC-3′) and RT2 (5′-CTCCAGACAAAGCTTGAAGCT-3′; 5′-TATCCACCATGG-CTCTGAATG-3′). PCR conditions were as follows: preincubation at 94°C for 2.5 min, then 30 cycles at 94°C for 30 sec, 52°C for 30 sec, and 72°C for 1 min. The primers for ACT7 were 5′-TGCTCTTCC-TCATGCTATCCTTC-3′ and 5′-CCACGAACCAGATAAGACAAGAC-3′. For RT-PCR analysis of SPK1 expression in different organs, primers RT2 were used. The primers used to amplify control glyceraldehyde-3-phosphate dehydrogenase were 5′-CACTTGAAGGGTGGTGCC-AAG-3′ and 5′-CCTGTTGTCGCCAACGAAGTC-3′ (Ohad et al., 1999).
Rapid Amplification of cDNA Ends and Construction of the Full-Length cDNA
Partial SPK1 cDNA clones was isolated from the Arabidopsis ecotype Columbia cDNA library (Kieber et al., 1993). The 5′ and 3′ ends of SPK1 cDNA were obtained by 5′ and 3′ rapid amplification of cDNA ends (RACE) according to the manufacturer's directions (Gibco BRL) with some modifications. For 5′ RACE, first-strand synthesis was primed using a gene-specific primer (GSP) and poly(A)+ RNA as a template. PCR was performed first using the upstream anchor primer AAP and gene-specific RACE primer; subsequently, nested amplification using AUAP and gene-specific nested primer was conducted when necessary. Four rounds of 5′ RACE were required to obtain the 5′ end of SPK1. The first-round GSP is 5′-GCTTCATCAGCCATTTCG-3′, RACE primer 5′-CCGTGCTTCCCACTC-TCTTCT-3′, and nested primer 5′-GGTGATCCAGCTCCTTCACTA-ACA-3′. The second-round GSP is 5′-GCATGATACATCGGA-AAACA-3′, RACE primer 5′-GTGCTTGCTCCAATGCCATTGAT-3′, and nested primer 5′-GCAAGGCAGTAGAATCAACGTGCT-3′. The third-round GSP is 5′-ATGTGGCAGGAAGAGAGA-3′, RACE primer 5′-CCCTAGCACCAACAGCGACCTGTGT-3′, and nested primer 5′-TCGGATATCTGTGTCATCCTTTCTCA-3′. The fourth-round GSP is 5′-CCAGTGTTTGTGGTGAGAT-3′, RACE primer 5′-CTCTCAGTT-AAGTGCACAGGCTCTT-3′, and nested primer 5′-AAGCAGGCA-AACTGATGCTGATG-3′. For 3′ RACE, oligo(dT) primer (5′-GGC-CACGCGTCGACTAGTACTTTTTTTTTTTTTTTTT[ACG][ACGT]-3′) was used for first-strand synthesis. PCR was performed using AUAP and gene-specific RACE primer 5′-AGGAATTCCATACTCAGCTT-3′.
Based on the 5′ RACE sequences, we constructed the full-length cDNA of SPK1 in two steps. First, for RT-PCR amplification of an ∼2.2-kb fragment with primers 5′-GCGTCGACCATCACATCATCTGTTATT-CACT-3′ and 5′-CCGTGCTTCTCCACTCTCTTC-3′, the fragment was cloned to SalI and NdeI sites of a partial SPK1 cDNA clone in pBluescript SK−. Second, for RT-PCR amplification of an ∼2.3-kb fragment with primers 5′-GCGTCGACTTACAGCTCCAGCTCGACTTTG-3′ and 5′-CCACCATGGCTCTGAATGCTGCAAC-3′, the fragment was cloned to SalI and NcoI sites of the recombinant partial SPK1 cDNA in the first step. PCR was performed with Pfu DNA polymerase (Promega, Madison, WI), and the conditions were as follows: preincubation at 95°C for 2 min, then 35 cycles of 95°C for 30 sec, 58°C for 30 sec, and 72°C for 5-min. The PCR products were cloned into pCRII-TOPO (Invitrogen, Carlsbad, CA) and sequenced before subcloning.
Reverse Genetic Screen
Pooled genomic DNA from 6500 T-DNA insertion lines in a Wassilewskija background (Arabidopsis Biological Resource Center [Columbus, OH] stock CS6502 [Feldmann, 1991]) were screened as described by Krysan (1996). Pools of DNA were screened by PCR using T-DNA left and right border-specific primers and primers specific for SPK1 every 2 kb in both directions. PCR was performed with increasingly deconvoluted DNA template pools until individual plants containing the insertion were identified. Cosegregation of the spk1 phenotype and the kanamycin resistance gene T-DNA marker was tested by plating seed from selfed heterozygous plants. The location of the T-DNA insertion was confirmed by amplifying genomic DNA by PCR with SPK1 and left border primers from the heterozygous plant and sequencing the PCR product.
Double-Stranded RNA Constructs and Agrobacterium tumefaciens–Mediated Transformation
Two constructs, DS1 corresponding to 1089 bp and DS2 corresponding to 677 bp of the SPK1 cDNA, were made, along with control construct DSCK with sequence (419 bp) against the 27th intron of the SPK1 gene. All of the fragments were amplified by PCR and inserted into XhoI and EcoRI sites in the sense direction and BamHI and XbaI sites in the antisense direction of pRNA69; then they were digested with NotI and inserted into the binary vector pJL10. For DS1, sense direction primers 5′-GCCTCGAGCGCCAAGAAGTCA-GGCTAGA-3′ and 5′-GCGAATTCCTTGCTCCACACACCATCAT-3′ and antisense direction primers 5′-GCGGATCCCTTGCTCCACACA-CCATCAT-3′ and 5′-GCTCTAGACGCCAAGAAGTCAGGCTAGA-3′ were used. For DS2, sense direction primers 5′-GCCTCGAGG-CAGCTGAAGTCGAGGGTTA-3′ and 5′-GCGAATTCCCTCAGTCTGCAGCACTGTT-3′ and antisense direction primers 5′-GCGGAT-CCCCTCAGTCTGCAGCACTGTT-3′ and 5′-GCTCTAGAGCAGCTG-AAGTCGAGGGTTA-3′ were used. For DSCK, sense direction primers 5′-TTGCTGGTGTCAGTGACGGAAAA-3′ and 5′-GCGAATTCC-GTGACAGGAAATGGTGCAAACA-3′ and antisense direction primers 5′-GCGGATCCCGTGACAGGAAATGGTGCAAACA-3′ and 5′-GCT-CTAGATTCCTCCACGTCGACACCTATT-3′ were used.
A. tumefaciens strain C58/pGV90 carrying double-stranded RNA constructs in pJL10 was used to transform Arabidopsis ecotype Columbia by floral dip (Clough and Bent, 1998). Transformed Arabidopsis lines were selected on soil by spraying a 1:100 dilution of Finale (AgrEvo Environmental Health, Montvale, NJ).
Sequence Analysis
Sequence alignments and presentation were made using ClustalW (Higgins et al., 1996) (http://www.ch.embnet.org/software/ClustalW.html) and BOXSHADE (http://www.ch.embnet.org/software/BOX_form.html), respectively.
Accession Numbers
The GenBank accession numbers for the sequences mentioned in this article are AF465831 (SPK1), A71430 (Arabidopsis ORF 4200c), AAF51561 (Drosophila unknown MOD1, MOD2, CDMS-domain gene), T34274 (C. elegans unknown MOD1, MOD2, CDMS-domain gene), BAA09454 (Homo sapiens DOCK180), AAC38973 (C. elegans CED-5), AAB69648 (Drosophila MBC), AAB70856 (Dictyostelium discoideum DocA), AAB67508 (Saccharomyces cerevisiae Ylr422wp).
Acknowledgments
We thank Eileen Mallery for excellent technical assistance and the Purdue Cytoskeleton Group for useful discussions. The Arabidopsis Biological Resource Center provided several strains and clones. Vector pRNA69 was a generous gift from John Bowman and John Emery. We also thank Stan Gelvin and colleagues for help in identifying the spk1-2 allele. This research was supported by Purdue University and Cooperative Research, Education, and Extension Service United States Department of Agriculture Grant No. 99-35304-8525 to D.B.S and National Science Foundation Integrative Biology and Neuroscience Grant No. 9506192 to M.D.M. The original spk1-1 allele was generated by Neil Olszewski, who is supported by National Science Foundation Molecular and Cellular Biosciences Grant No. 9604126.
Article, publication date, and citation information can be found at www.plantcell.org/cgi/doi/10.1105/tpc.010346.
References
- Alessa, L., and Kropf, D.L. (1999). F-actin marks the rhizoid pole in living Pelvetia compressa zygotes. Development 126, 201–209. [DOI] [PubMed] [Google Scholar]
- Apostolakos, P., Galatis, B., and Panteris, E. (1991). Microtubules in cell morphogenesis and intercellular space formation in Zea mays leaf mesophyll and Pilea cadierei Epithem. J. Plant Physiol. 137, 591–601. [Google Scholar]
- Ausubel, F., Brent, R., Kingston, R., Moore, D., Seidman, J., Smith, J., and Struhl, K. (1994). Current Protocols in Molecular Biology. (New York: John Wiley and Sons).
- Baluska, F., Salaj, J., Mathur, J., Braun, M., Jasper, F., Samaj, J., Chua, N.H., Barlow, P.W., and Volkmann, D. (2000). Root hair formation: F-actin-dependent tip growth is initiated by local assembly of profilin-supported F-actin meshworks accumulated within expansin-enriched bulges. Dev. Biol. 227, 618–632. [DOI] [PubMed] [Google Scholar]
- Baskin, T.I., Meekes, H.T.H.M., Liang, B.M., and Sharp, R.E. (1999). Regulation of growth anisotropy in well-watered and water-stressed maize roots. II. Role of cortical microtubules and cellulose microfibrils. Plant Physiol. 119, 681–692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bevan, M., et al. (1998). Analysis of 1.9 Mb of contiguous sequence from chromosome 4 of Arabidopsis thaliana. Nature 391, 485–488. [DOI] [PubMed] [Google Scholar]
- Bibikova, T.N., Blancaflor, E.B., and Gilroy, S. (1999). Microtubules regulate tip growth and orientation in root hairs of Arabidopsis thaliana. Plant J. 17, 657–665. [DOI] [PubMed] [Google Scholar]
- Bichet, A., Desnos, T., Turner, S., Grandjean, O., and Hofte, H. (2001). BOTERO1 is required for normal orientation of cortical microtubules and anisotropic cell expansion in Arabidopsis. Plant J. 25, 137–148. [DOI] [PubMed] [Google Scholar]
- Boevink, P., Oparka, K., Santa Cruz, S., Martin, B., Betteridge, A., and Hawes, C. (1998). Stacks on tracks: The plant Golgi apparatus traffics on an actin/ER network. Plant J. 15, 441–447. [DOI] [PubMed] [Google Scholar]
- Burk, D.H., Liu, B., Zhong, R., Morrison, W.H., and Ye, Z.H. (2001). A katanin-like protein regulates normal cell wall biosynthesis and cell elongation. Plant Cell 13, 807–827. [PMC free article] [PubMed] [Google Scholar]
- Chan, J., Jensen, C.G., Jensen, L.C.W., Bush, M., and Lloyd, C.W. (1999). The 65-kDa carrot microtubule-associated protein forms regularly arranged filamentous cross-bridges between microtubules. Proc. Natl. Acad. Sci. USA 96, 14931–14936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clough, S., and Bent, A. (1998). Floral dip: A simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 16, 735–743. [DOI] [PubMed] [Google Scholar]
- Cyr, R.J. (1994). Microtubules in plant morphogenesis: Role of the cortical array. Annu. Rev. Cell Biol. 10, 153–180. [DOI] [PubMed] [Google Scholar]
- Daub, H., Gevaert, K., Vandekerckhove, J., Sobel, A., and Hall, A. (2001). Rac/Cdc42 and p65PAK regulate the microtubule-destabilizing protein stathmin through phosphorylation at serine 16. J. Biol. Chem. 276, 1677–1680. [DOI] [PubMed] [Google Scholar]
- Eaton, S., Wepf, R., and Simons, K. (1996). Roles for Rac1 and Cdc42 in planar polarization and hair outgrowth in the wing of Drosophila. J. Cell Biol. 135, 1277–1289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Erickson, M.R.S., Galletta, B.S., and Abmayr, S.M. (1997). Drosophila myoblast city encodes a conserved protein that is essential for myoblast fusion, dorsal closure, and cytoskeletal organization. J. Cell Biol. 138, 589–603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Falconer, M.M., and Seagull, R.W. (1985. a). Immunofluorescent and calcofluor white staining of developing tracheary elements in Zinnia elegans L. suspension cultures. Protoplasma 125, 190–198. [Google Scholar]
- Falconer, M.M., and Seagull, R.W. (1985. b). Xylogenesis in tissue culture: Taxol effects on microtubule reorientation and lateral association in differentiating cells. Protoplasma 128, 157–166. [Google Scholar]
- Feldman, K. (1991). T-DNA insertion mutagenesis in Arabidopsis: Mutational spectrum. Plant J. 1, 71–83. [Google Scholar]
- Flanders, D.J., Rawlins, D.J., Shaw, P.J., and Lloyd, C.W. (1990). Re-establishment of the interphase microtubule array in vacuolated plant cells, studied by confocal microscopy and 3-D imaging. Development 110, 897–904. [Google Scholar]
- Freeling, M. (1992). A conceptual framework for maize leaf development. Dev. Biol. 153, 44–58. [DOI] [PubMed] [Google Scholar]
- Fu, Y., Wu, G., and Yang, Z. (2001). Rop GTPase-dependent dynamics of tip-localized F-actin controls tip growth in pollen tubes. J. Cell Biol. 152, 1019–1032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibbon, B.C., Kovar, D.R., and Staiger, C.J. (1999). Latrunculin B has different effects on pollen germination and tube growth. Plant Cell 11, 2349–2363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giddings, T.H., and Staehelin, L.A. (1991). Microtubule-mediated control of microfibril deposition: A re-examination of the hypothesis. In The Cytoskeletal Basis of Plant Growth and Form, C.W. Lloyd, ed (San Diego, CA: Academic Press), pp. 85–99.
- Granger, C.L., and Cyr, R.J. (2000). Microtubule reorganization in tobacco BY-2 cells stably expressing GFP-MBD. Planta 210, 502–509. [DOI] [PubMed] [Google Scholar]
- Hall, A. (1998). Rho GTPases and the actin cytoskeleton. Science 279, 509–514. [DOI] [PubMed] [Google Scholar]
- Hasegawa, H., Kiyokawa, E., Tanaka, S., Nagashima, K., Gotoh, N., Shibuya, M., Kurata, T., and Matsuda, M. (1996). DOCK180, a major CRK-binding protein, alters cell morphology upon translocation to the cell membrane. Mol. Cell. Biol. 16, 1770–1776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heslop-Harrison, J., Heslop-Harrison, Y., Cresti, M., Tiezzi, A., and Ciampolini, F. (1986). Actin during pollen tube germination. J. Cell Sci. 86, 1–8. [Google Scholar]
- Higgins, D.G., Thompson, J.D., and Gibson, T.J. (1996). Using CLUSTAL for multiple sequence alignments. Methods Enzymol. 266, 383–402. [DOI] [PubMed] [Google Scholar]
- Hülskamp, M., Misra, S., and Jürgens, G. (1994). Genetic dissection of trichome cell development in Arabidopsis. Cell 76, 555–566. [DOI] [PubMed] [Google Scholar]
- Jung, G., and Wernicke, W. (1991). Patterns of actin filaments during cell shaping in developing mesophyll of wheat (Triticum aestivum L.). Eur. J. Cell Biol. 56, 139–146. [PubMed] [Google Scholar]
- Kieber, J., Rothenberg, M., Roman, G., Feldmann, K., and Ecker, J. (1993). CTRl, a negative regulator of the ethylene response pathway in Arabidopsis, encodes a member of the Raf family of protein kinases. Cell 72, 427–441. [DOI] [PubMed] [Google Scholar]
- Kiyokawa, E., Hashimoto, Y., Kobayashi, S., Sugimura, H., Kurata, T., and Matsuda, M. (1998). Activation of Rac1 by a Crk SH3-binding protein, DOCK180. Genes Dev. 12, 3331–3336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kobayashi, H., Fukuda, H., and Shibaoka, H. (1988). Interrelation between the spatial disposition of actin filaments and microtubules during the differentiation of tracheary elements in cultured Zinnia cells. Protoplasma 143, 29–37. [Google Scholar]
- Kobayashi, S., Shirai, T., Kiyokawa, E., Mochizuki, N., Matsuda, M., and Fukui, Y. (2001). Membrane recruitment of DOCK180 by binding Ptdlns(3,4,5)P3. Biochem. J. 354, 73–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krogh, A., Larsson, B., von Heijne, G., and Sonnhammer, E.L.L. (2001). Predicting transmembrane protein topology with a hidden Markov model: Application to complete genomes. J. Mol. Biol. 305, 567–580. [DOI] [PubMed] [Google Scholar]
- Krysan, P.J., Young, J.C., Tax, F., and Sussman, M.R. (1996). Identification of transferred DNA insertions within Arabidopsis genes involved in signal transduction and ion transport. Proc. Natl. Acad. Sci. USA 93, 8145–8150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kyte, J., and Doolittle, R.F. (1982). A simple method for displaying the hydropathic character of a protein. J. Mol. Biol. 157, 105–132. [DOI] [PubMed] [Google Scholar]
- Li, H., and Yang, Z. (2000). Rho GTPases and the actin cytoskeleton. In Actin: A Dynamic Framework for Multiple Plant Cell Functions, C.J. Staiger, F. Baluska, D. Volkmann, and P.W. Barlow, eds (Dordrecht, The Netherlands: Kluwer Academic Publishers), pp. 301–322.
- Mascarenhas, J.P., and LaFountain, J. (1972). Protoplasmic streaming, cytochalasin B, and growth of the pollen tube. Tissue Cell 4, 11–14. [DOI] [PubMed] [Google Scholar]
- Mathur, J., Spielhofer, P., Kost, B., and Chua, N. (1999). The actin cytoskeleton is required to elaborate and maintain spatial patterning during trichome cell morphogenesis in Arabidopsis thaliana. Development 126, 5559–5568. [DOI] [PubMed] [Google Scholar]
- McKinney, E.C., Ali, N., Traut, A., Feldmann, K.A., Belostotsky, D.A., McDowell, J.M., and Meagher, R.B. (1995). Sequence-based identification of T-DNA insertion mutations in Arabidopsis: Actin mutants act2-1 and act4-1. Plant J. 8, 613–622. [DOI] [PubMed] [Google Scholar]
- Murashige, T., and Skoog, F. (1962). A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol. Plant. 15, 473–497. [Google Scholar]
- Nebenfuhr, A., Gallagher, L.A., Dunahay, T.G., Frohlick, J.A., Mazurkiewicz, A.M., Meehl, J.B., and Staehelin, L.A. (1999). Stop-and-go movements of plant Golgi stacks are mediated by the acto-myosin system. Plant Physiol. 121, 1127–1141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newman, T., Ohme-Takagi, M., Taylor, C., and Green, P. (1993). DST sequences, highly conserved among plant SAUR genes, target reporter transcripts for rapid decay in tobacco. Plant Cell 5, 701–714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nolan, K.M., Barrett, K., Lu, Y., Hu, K.Q., Vincent, S., and Settleman, J. (1998). Myoblast city, the Drosophila homolog of DOCK180/CED-5, is required in a rac signaling pathway utilized for multiple developmental processes. Genes Dev. 12, 3337–3342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohad, N., Yadegari, R., Margossian, L., Hannon, M., Michaeli, D., Harade, J., Goldberg, R., and Fischer, R. (1999). Mutations in FIE, a WD polycomb group gene, allow endosperm development without fertilization. Plant Cell 11, 407–415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oppenheimer, D.G., Pollock, M.A., Vacik, J., Szymanski, D.B., Ericson, B., Feldmann, K., and Marks, M.D. (1997). Essential role of a kinesin-like protein in Arabidopsis trichome morphogenesis. Proc. Natl. Acad. Sci. USA 94, 6261–6266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Panteris, E., Apostolakos, P., and Galatis, B. (1993. a). Microtubule organization, mesophyll cell morphogenesis, and intercellular space formation in Adiantum capillus veneris leaflets. Protoplasma 172, 97–110. [DOI] [PubMed] [Google Scholar]
- Panteris, E., Apostolakos, P., and Galatis, B. (1993. b). Microtubules and morphogenesis in ordinary epidermal cells of Vigna sinensis leaves. Protoplasma 174, 91–100. [Google Scholar]
- Panteris, E., Apostolakos, P., and Galatis, B. (1994). Sinuous ordinary epidermal cells: Behind several patterns of waviness, a common morphogenetic mechanism. New Phytol. 127, 771–780. [DOI] [PubMed] [Google Scholar]
- Quatrano, R.S. (1973). Separation of processes associated with differentiation of two-celled Fucus embryos. Dev. Biol. 30, 209–213. [DOI] [PubMed] [Google Scholar]
- Reddien, P.W., and Horvitz, H.R. (2000). CED-2/crkll and CED-10/rac control phagocytosis and cell migration in Caenorhabditis elegans. Nat. Cell Biol. 2, 131–136. [DOI] [PubMed] [Google Scholar]
- Rédei, G.P. (1962). Single locus heterosis. Z. Vererbungsl. 93, 164–170. [Google Scholar]
- Satiat-Jeunemaitre, B., and Hawes, C. (1996). Golgi-membrane dynamics are cytoskeleton dependent: A study on Golgi stack movement induced by brefeldin A. Protoplasma 191, 21–33. [Google Scholar]
- Smertenko, A., Saleh, N., Igarashi, H., Mori, H., Hauser-Hahn, I., Jiang, C.J., Sonobe, S., Lloyd, C.W., and Hussey, P.J. (2000). A new class of microtubule-associated proteins in plants. Nat. Cell Biol. 2, 750–753. [DOI] [PubMed] [Google Scholar]
- Song, H., Golovkin, M., Reddy, A.S.N., and Endow, S.A. (1997). In vitro motility of AtKCBP, a calmodulin-binding kinesin protein of Arabidopsis. Proc. Natl. Acad. Sci. USA 94, 322–327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Staiger, C.J. (2000). Signaling to the actin cytoskeleton in plants. Annu. Rev. Plant Physiol. Plant Mol. Biol. 51, 257–288. [DOI] [PubMed] [Google Scholar]
- Szymanski, D.B. (2000). The role of actin during Arabidopsis trichome morphogenesis. In Actin: A Dynamic Framework for Multiple Plant Cell Functions, C.J. Staiger, F. Baluska, D. Volkmann, and P. Barlow, eds (Dordrecht, The Netherlands: Kluwer Academic Publishers), pp. 391–410.
- Szymanski, D.B., Marks, M.D., and Wick, S.M. (1999). Organized F-actin is essential for normal trichome morphogenesis in Arabidopsis. Plant Cell 11, 2331–2347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thimann, K.V., Reese, K., and Nachmias, V.T. (1992). Actin and the elongation of plant cells. Protoplasma 171, 153–166. [Google Scholar]
- Tiwari, S.C., and Wilkins, T.A. (1995). Cotton (Gossypium hirsutum) seed trichomes expand via diffuse growing mechanisms. Can. J. Bot. 73, 746–757. [Google Scholar]
- Traas, J.A., Doonan, J.H., Rawlins, D.J., Shaw, P.J., Watts, J., and Lloyd, C.W. (1987). An actin network is present in the cytoplasm throughout the cell cycle of carrot cells and associates with the dividing nucleus. J. Cell Biol. 105, 387–395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsuge, T., Tsukaya, H., and Uchimiya, H. (1996). Two independent and polarized processes of cell elongation regulate leaf blade expansion in Arabidopsis thaliana (L.) Heynh. Development 122, 1589–1600. [DOI] [PubMed] [Google Scholar]
- Tsukaya, H., Tsuge, T., and Uchimiya, H. (1994). The cotyledon: A superior system for studies of leaf development. Planta 195, 309–312. [Google Scholar]
- Turner, C.M., and Adler, P.N. (1998). Distinct roles for the actin and microtubule cytoskeletons in the morphogenesis of epidermal hairs during wing development in Drosophila. Mech. Dev. 70, 181–192. [DOI] [PubMed] [Google Scholar]
- Waller, F., and Nick, P. (1997). Response of actin microfilaments during phytochrome-controlled growth of maize seedlings. Protoplasma 200, 154–162. [Google Scholar]
- Wasteneys, G.O. (2000). The cytoskeleton and growth polarity. Curr. Opin. Plant Biol. 3, 503–511. [DOI] [PubMed] [Google Scholar]
- Wasteneys, G.O., Willingale-Theune, J., and Menzel, D. (1997). Freeze shattering: A simple and effective method for permeabilizing higher plant cell walls. J. Microsc. 188, 51–61. [DOI] [PubMed] [Google Scholar]
- Wernicke, W., and Jung, G. (1992). Role of cytoskeleton in cell shaping of developing mesophyll of wheat (Triticum aestivum L.). Eur. J Cell. Biol. 57, 88–94. [PubMed] [Google Scholar]
- Whittington, A.G., Vugrek, O., Wei, K.J., Hasenbein, N.G., Sugimoto, K., Rashbrooke, M.C., and Wasteneys, G.O. (2001). MOR1 is essential for organizing cortical microtubules in plants. Nature 411, 610–614. [DOI] [PubMed] [Google Scholar]
- Wu, Y.C., and Horvitz, H.R. (1998). C. elegans phagocytosis and cell-migration protein CED-5 is similar to human DOCK180. Nature 392, 501–504. [DOI] [PubMed] [Google Scholar]