Abstract
Tandem repeat sequences are frequently associated with gene silencing phenomena. The Arabidopsis thaliana FWA gene contains two tandem repeats and is an efficient target for RNA-directed de novo DNA methylation when it is transformed into plants. We showed that the FWA tandem repeats are necessary and sufficient for de novo DNA methylation and that repeated character rather than intrinsic sequence is likely important. Endogenous FWA can adopt either of two stable epigenetic states: methylated and silenced or unmethylated and active. Surprisingly, we found small interfering RNAs (siRNAs) associated with FWA in both states. Despite this, only the methylated form of endogenous FWA could recruit further RNA-directed DNA methylation or cause efficient de novo methylation of transgenic FWA. This suggests that RNA-directed DNA methylation occurs in two steps: first, the initial recruitment of the siRNA-producing machinery, and second, siRNA-directed DNA methylation either in cis or in trans. The efficiency of this second step varies depending on the nature of the siRNA-producing locus, and at some loci, it may require pre-existing chromatin modifications such as DNA methylation itself. Enhancement of RNA-directed DNA methylation by pre-existing DNA methylation could create a self-reinforcing system to enhance the stability of silencing. Tandem repeats throughout the Arabidopsis genome produce siRNAs, suggesting that repeat acquisition may be a general mechanism for the evolution of gene silencing.
InArabidopsis, methylation and silencing mediated by theFWA transgene are found to require the tandem repeat sequences upstream ofFWA and to be independent of siRNA association.
Introduction
Cytosine methylation of DNA protects eukaryote genomes from transposons, and also regulates developmental gene expression [1,2]. Tandem repeat sequences are common in methylated regions of the genome. The tandem repeats in retrotransposons are essential for their mobility and are frequently transcriptionally silenced by DNA methylation [3,4]. Tandem repeats upstream of the maize b1 gene are required for paramutation, a process in which a heritable silent state is transferred from one allele to another [5]. In addition, tandem repeats control imprinting of the RasGRF1 gene in mammals and are associated with other imprinted genes in mammals and in plants [6–9]. One such example is the FWA gene of Arabidopsis thaliana. In adult tissues, FWA is silenced by DNA methylation on two pairs of tandem repeats at the 5' end of its transcribed region [10]. The maternal copy of FWA is specifically demethylated and expressed in the extra-embryonic endosperm tissue, while paternal FWA remains methylated and silent [6]. Because FWA is only demethylated in the terminally differentiating endosperm, it remains heritably methylated throughout the plant life cycle. Rare unmethylated epigenetic alleles of FWA (termed fwa) ectopically overexpress the FWA transcription factor in adult tissues, causing a dominant late-flowering phenotype [10]. These alleles are very stable, and remethylation that re-establishes an early-flowering phenotype has never been observed.
In both plants and animals, de novo DNA methylation of previously unmethylated sequences is mediated by DNA methyltransferase enzymes of the Dnmt3 family (DOMAINS REARRANGED METHYLTRANSFERASE2 [DRM2] in Arabidopsis) [11]. DRM2 is responsible for de novo DNA methylation in all known sequence contexts: CG, CNG (where N is any base), and asymmetric or CHH (where H = A, T, or C) [11]. However, the maintenance of pre-existing DNA methylation is sequence context–dependent. CG DNA methylation is maintained by MET1 (a homolog of mammalian Dnmt1), whereas CNG and asymmetric methylation are maintained by the overlapping functions of CHROMOMETHYLASE3 (CMT3) and DRM2 [2,12].
De novo DNA methylation can be studied in Arabidopsis by transforming plants with an additional copy of FWA [11]. Despite the extreme stability of an unmethylated fwa endogenous allele, a transgenic copy of FWA introduced into wild-type plants by Agrobacterium-mediated transformation is always de novo methylated and silenced. Because both endogenous and transgenic FWA are silenced, transformed plants do not display a late-flowering phenotype. However, when drm2 mutant plants are transformed with FWA, the introduced transgenes remain unmethylated, and FWA overexpression causes a late-flowering phenotype. In drm2 transformants, endogenous FWA retains its methylation and silencing, because DRM2 only controls de novo methylation but not maintenance methylation of CG sites [11]. Intriguingly, when unmethylated FWA transgenes from drm2 transformants are outcrossed to reintroduce wild-type DRM2, the transgenes remain stably unmethylated [11]. This is consistent with the stability of unmethylated fwa endogenous alleles and suggests that FWA de novo DNA methylation only occurs during the process of plant transformation.
RNA interference (RNAi) is a versatile silencing pathway in which double-stranded RNA is processed by Dicer nucleases to generate 21–24-nucleotide small interfering RNAs (siRNAs) [13]. siRNAs can down-regulate gene expression through mRNA cleavage, translational inhibition, or transcriptional silencing. Previously, we used the FWA transformation assay to demonstrate that de novo DNA methylation by DRM2 is guided by an RNAi pathway including the NUCLEAR RNA POLYMERASE IV A (NRPD1a) subunit of DNA-dependent RNA polymerase IV (RNA Pol IV), RNA-DEPENDENT RNA POLYMERASE2 (RDR2), DICER-LIKE3 (DCL3), and ARGONAUTE4 (AGO4) [14]. The recent discovery that a maize ortholog of RDR2 is required for paramutation demonstrates that RNA-directed DNA methylation at tandem repeats is conserved in both monocots and eudicots [15]. In addition to their defects in establishment of DNA methylation, Arabidopsis RNAi mutants reduce or eliminate CNG and asymmetric DNA methylation at endogenous tandem repeats and other sequences in a manner analogous to that of drm2 mutants. However, at most loci, the maintenance of CG methylation in drm2 or the RNAi mutants is generally unaffected. These results show that RNAi-related processes are required to propagate non-CG DNA methylation; meaning that, even after establishment, siRNA production likely persists at particular genomic loci.
Although it seems clear that de novo DNA methylation derives its sequence specificity from siRNAs, it is not known why transformed FWA is targeted for DNA methylation, unlike the majority of single-copy Arabidopsis genes transformed into plants. Here we show that the tandem repeats of FWA are necessary and sufficient for triggering de novo DNA methylation, and that repeated character (rather than the FWA sequence itself) is likely critical. Transformed FWA can silence the unmethylated endogenous fwa gene in trans, and this property also requires tandem repeats. Surprisingly, the methylation state of endogenous FWA can affect silencing of an incoming transgene, because FWA transformed into fwa-1 is inefficiently methylated compared to wild type. The fact that DNA methylation enhances the ability of endogenous FWA to communicate with transformed FWA is particularly striking, because both methylated and unmethylated forms of endogenous FWA produce equivalent levels of siRNAs. Furthermore, we show that FWA siRNA synthesis is dependent on wild-type NRPD1a, RDR2, and DCL3, but not on the putatively downstream components NRPD1b, AGO4, DEFECTIVE IN RNA-DIRECTED DNA METHYLATION1 (DRD1), and DRM2. Our results suggest a two-step model for DNA methylation, in which tandem repeats are sufficient to recruit NRPD1a, RDR2, and DCL3 to produce siRNAs, but a second triggering event such as plant transformation is required to allow siRNAs to target downstream DNA methylation. Finally, a genomic analysis suggests that unique tandem repeats, such as those at FWA, are common targets for the siRNA machinery, indicating the potential generality of this gene silencing mechanism on an evolutionary time scale.
Results
FWA De Novo DNA Methylation during Plant Transformation Can Occur after Fertilization of the Female Gametophyte
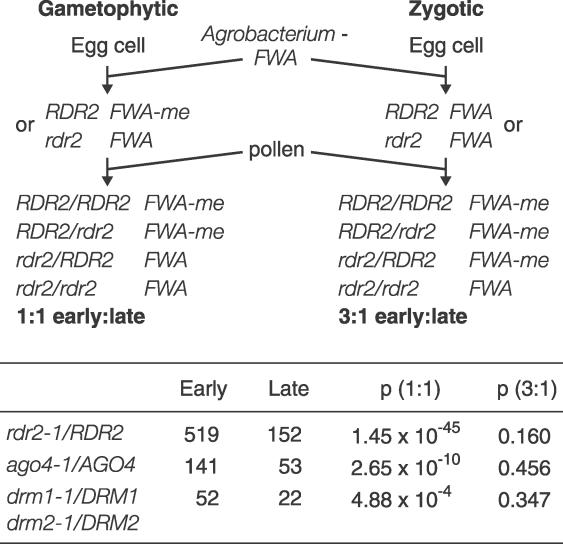
Although an unmethylated fwa endogenous allele never becomes spontaneously methylated, de novo DNA methylation of transformed FWA occurs with 100% efficiency [11,14]. Furthermore, unmethylated FWA transgenes in drm2 transformants are not de novo methylated when crossed to plants containing functional DRM2. This suggests that de novo methylation of FWA occurs either during the transformation process, or shortly thereafter, and we sought to determine more precisely when this event occurs. During the floral dip procedure, Agrobacterium tumefaciens transforms the female gametophytic lineage of Arabidopsis (probably the egg cell itself), integrating its transferred DNA (T-DNA) only into the maternal genome [16,17]. We transformed FWA into heterozygous rdr2–1/RDR2, ago4–1/AGO4, and drm1–1/DRM1 drm2–1/DRM2 plants (Figure 1). If DNA methylation and gene silencing must be established in the female gametophyte, then the haploid genotype of the gametophyte should control de novo DNA methylation and one-half of the transformed T1 progeny would be late flowering. However, if a wild-type RDR2, AGO4, or DRM2 gene from the pollen could rescue de novo DNA methylation in the fertilized zygote, only one in four plants would fail to silence FWA and flower late (Figure 1; this ratio could also be explained if de novo DNA methylation requires a factor that moves from the fertilized extra-embryonic endosperm into the zygote). A third possibility is that maternally supplied protein or mRNA could rescue de novo DNA methylation, causing all progeny of a heterozygous mutant to be early flowering.
Figure 1. De Novo DNA Methylation of Transformed FWA Can Be Rescued by Wild-Type Genes from Pollen.
Heterozygous rdr2–1, ago4–1, and drm1–1 drm2–1 plants were transformed with FWA. If the gametophytic genotype controls de novo DNA methylation and silencing, a 1:1 ratio of early- to late-flowering plants is predicted. If a wild-type gene from pollen can rescue de novo DNA methylation in the fertilized zygote, a 3:1 early- to late-flowering ratio results. The ratio of early- to late-flowering plants was determined in the T1 generation (see Figure S1 for determination of early versus late flowering in each ecotype). The probability of the observed results occurring by chance assuming a 1:1 or 3:1 ratio was calculated with the χ2 test.
We found that all three experiments showed an early:late flowering ratio close to 3:1 amongst FWA T1 transformants (Figure 1). This early-to-late ratio of flowering plants is consistent with zygotic control, but it could also arise if de novo DNA methylation occurred in the gamete but half the mutant gametes were rescued by maternally supplied protein or mRNA. To eliminate this possibility, we used PCR-based molecular markers to genotype 23 late-flowering T1 progeny of rdr2–1/RDR2 plants and 18 late-flowering T1 progeny of ago4–1/AGO4 plants. All were homozygous mutant for rdr2–1 or ago4–1, respectively (unpublished data), indicating that the genotype of the fertilized zygote or endosperm determines whether an FWA transgene is silenced. Thus, although FWA de novo DNA methylation requires plant transformation, the particular conditions that facilitate de novo DNA methylation must persist until sometime after fertilization.
FWA Tandem Repeats Are Necessary and Sufficient to Trigger De Novo Methylation
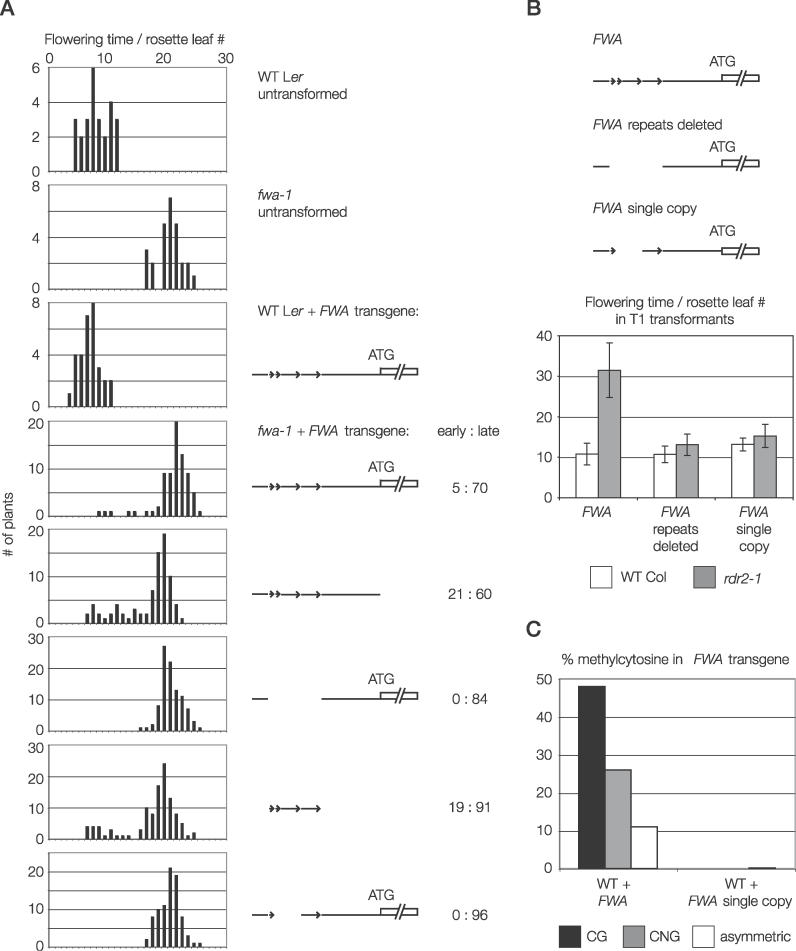
DNA methylation of FWA is restricted to the two pairs of tandem repeats at the farthest 5' end of its transcribed region, suggesting that they play a key role in controlling gene silencing [10]. The FWA tandem repeats are 2 × 38 base pairs (bp) and 2 × 198 bp in length. To test the role of tandem repeats in FWA de novo DNA methylation, we cloned several deletion variants of FWA. Because some of these variants did not contain the FWA open reading frame, they would not be expected to cause late flowering when transformed into wild-type Arabidopsis, meaning that we could not use transgene-induced late flowering as an assay. Therefore, we took advantage of the observation that transforming the late-flowering fwa-1 mutant with a cosmid containing FWA causes the occasional appearance of early-flowering plants among first generation T1 transformants [10]. Despite the fact that fwa-1 plants never regain DNA methylation during normal plant growth, both transgenic and endogenous FWA genes contain DNA methylation in early-flowering fwa-1 + FWA plants [10]. This shows that transformed FWA can target de novo DNA methylation to the unmethylated endogenous fwa gene, albeit inefficiently.
We transformed fwa-1 plants with a 6.1 kilobase FWA transgene, and found that 5%–25% of transformants had an early-flowering phenotype (Figure 2A), an effect similar to that caused by FWA cosmid transformation. Deleting the FWA open reading frame did not abolish the ability of the transgene to silence endogenous FWA, but a version lacking the tandem repeats altogether yielded no early-flowering transformants. The tandem repeats alone cloned into an Agrobacterium vector were able to cause trans-silencing, showing that this region of FWA is responsible for the activity. Most importantly, an FWA transgene containing a single copy of each tandem repeat sequence (“single-copy FWA”) was unable to silence endogenous fwa. The “repeats deleted” and “single-copy” transgenes did not cause late flowering when transformed into the rdr2–1 mutant (Figure 2B), indicating that they are not transcribed even when de novo DNA methylation is compromised. Therefore, the flowering time of fwa-1 plants that were transformed with these transgenes is not influenced by transgene expression, and transgenes lacking tandem repeat character are incapable of silencing endogenous FWA.
Figure 2. Recruitment of RNA-Directed DNA Methylation to Transformed Direct Repeats.
(A) Silencing of endogenous FWA by transformed FWA requires tandem repeats in the incoming transgene. Wild-type (WT) Ler or fwa-1 plants were transformed with a complete FWA transgene or the deletion variants shown at right. FWA contains two tandem repeats, which are depicted as two sets of arrows. Parental fwa-1 plants are uniformly late flowering. Early flowering in fwa-1 transformants occurs when endogenous fwa is silenced by de novo DNA methylation. Plants were scored as early- (17 leaves or fewer) or late-flowering (18 leaves or more) by comparison to untransformed Ler and fwa-1.
(B) FWA transgenes lacking tandem repeats do not cause late flowering when transformed into rdr2–1. The full FWA gene, “FWA repeats deleted” transgene, and “FWA single copy” transgenes are diagrammed. Wild-type Col and rdr2–1 plants were transformed with each transgene, and flowering time was assayed in the T1 generation.
(C) De novo DNA methylation of transformed FWA requires tandem repeat character. Wild-type Col plants were transformed with full-length FWA or with single-copy FWA. DNA methylation of the FWA transgene was assayed by bisulfite sequencing.
It is possible that transcriptional activity of an FWA transgene contributes to the silencing of the endogenous copy of FWA in trans. Therefore, we sought to test de novo DNA methylation of the transgene directly by transforming single-copy FWA into wild-type plants and measuring DNA methylation of the transgene by bisulfite genomic sequencing [10]. The complete FWA gene transformed into wild type was robustly methylated in the T1 generation, but a single-copy FWA transgene was completely unmethylated (Figure 2C). We showed previously that de novo DNA methylation of FWA requires the siRNA-metabolizing factors PolIVa, RDR2, DCL3, and AGO4. Together with the data presented here, these results suggest that RNA-directed DNA methylation is recruited to the FWA tandem repeat sequences during plant transformation. Although tandem repeats are associated with many gene-silencing phenomena, in this instance it is clear that a single copy of the same sequence lacks the behavior of the tandem repeat. We interpret these results to mean that the repeated character of transformed FWA, rather than its intrinsic sequence, is required to target DNA methylation to the unmethylated endogenous gene. An alternate explanation is that a sequence-specific factor recognizes transformed FWA and binds cooperatively in a manner that requires two copies of the repeats.
Efficient Silencing of Transformed FWA Requires DNA Methylation of Endogenous FWA
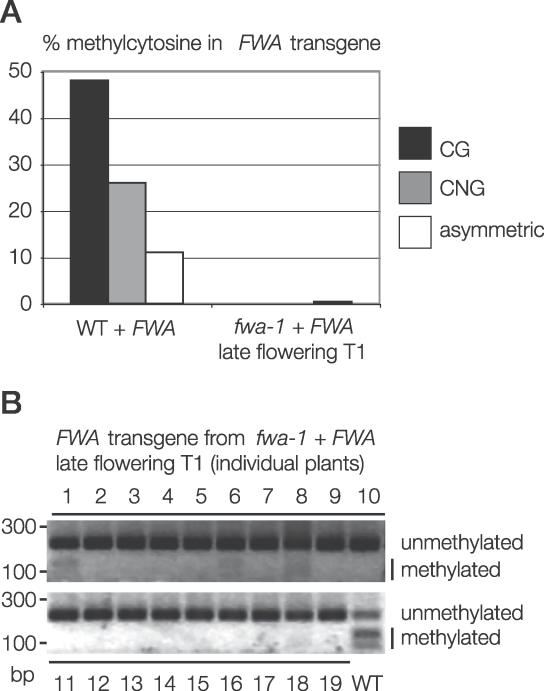
Silencing of unmethylated, endogenous fwa by transformed FWA shows that an incoming transgene can communicate with an identical endogenous sequence. When unmethylated fwa-1 mutants are transformed with FWA, early-flowering transformants must have silenced both the transgenic and endogenous FWA genes, because overexpression of any copy of FWA causes a dominant late-flowering phenotype. We sought to determine the fate of the FWA transgene in the majority of late-flowering fwa-1 + FWA transformants, where endogenous FWA is still overexpressed. We expected that the FWA transgene would be efficiently methylated in all transformants, reasoning that cis methylation of the transgene would be more efficient than trans methylation of the endogenous gene induced by the transgene (overexpression of the unmethylated endogenous gene would still cause late flowering, masking silencing of the transgenic copy). To our surprise, however, we found that transgenic FWA was completely unmethylated in late-flowering fwa-1 + FWA T1 transformants, when assayed either by bisulfite genomic sequencing (Figure 3A), or by testing 19 individual transformants using a bisulfite PCR/restriction enzyme assay (Figure 3B). FWA transformed into wild-type plants is methylated with 100% efficiency, yet our experiment shows that FWA transformed into fwa-1 plants is seldom methylated. Thus, efficient de novo DNA methylation of transformed FWA requires DNA methylation of endogenous FWA. This demonstrates a second type of communication, in which the endogenous methylated FWA gene assists DNA methylation of an incoming transgenic copy.
Figure 3. Communication between Methylated Endogenous FWA and Transformed FWA .
(A) Efficient de novo DNA methylation of transgenic FWA requires DNA methylation at endogenous FWA. DNA methylation of an FWA transgene introduced into wild-type or fwa-1 plants was assayed by bisulfite sequencing. For fwa-1 + FWA, three clones were sequenced from each of eight late-flowering individuals (each transformant had >20 rosette leaves at bolting). Early-flowering fwa-1 transformants were discarded, because they have silenced the FWA transgene by de novo DNA methylation.
(B) Transgenic FWA is unmethylated in many late-flowering fwa-1 + FWA T1 individuals. Before bisulfite treatment, genomic DNA from late-flowering fwa + FWA T1 plants was digested with BglII to destroy the endogenous FWA gene. DNA methylation of transgenic FWA was assayed by PCR from bisulfite-treated DNA followed by ClaI digestion. CG DNA methylation protects the ClaI site from bisulfite conversion, allowing restriction digestion after bisulfite treatment. Wild-type DNA (not digested with BglII) was assayed as a control.
siRNAs Are Produced from Both Methylated and Unmethylated Forms of FWA
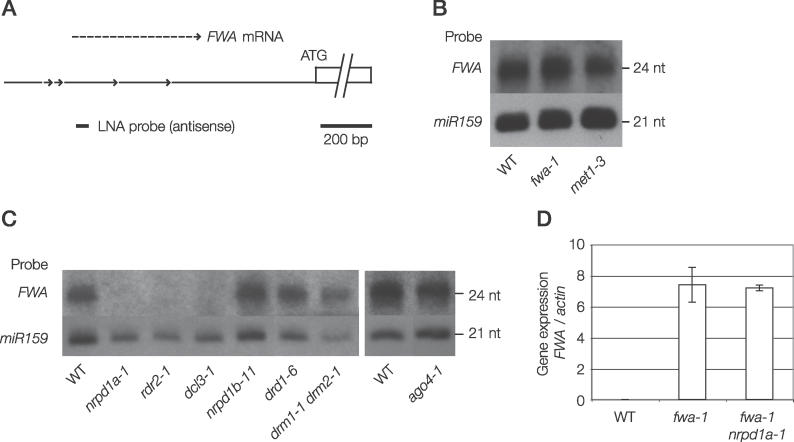
We wished to correlate these gene silencing phenomena with the production of FWA siRNAs. Massively parallel signature sequencing of Arabidopsis small RNA has shown that FWA siRNAs have very low abundance [18]. Only four siRNAs matching the FWA tandem repeats were cloned. Their abundance measured in transcripts per quarter million (TPQ) is 18 in libraries made from floral tissue, in contrast to microRNA 167, which has a TPQ of 59951 in the same libraries [18]. Two previous studies have used Northern blot analysis to detect FWA siRNAs using conventional RNA probes [4,19]. However, the FWA siRNA signal in these previous experiments was very close to background levels, making it difficult to accurately assess the abundance of FWA siRNAs in different genetic backgrounds. To circumvent these problems, we synthesized a locked nucleic acid (LNA) oligonucleotide probe to FWA [20], with an LNA backbone substitution at every third position (Figure 4A). Using this strategy, we were able to detect a robust signal for FWA 24-nucleotide siRNAs (Figure 4B).
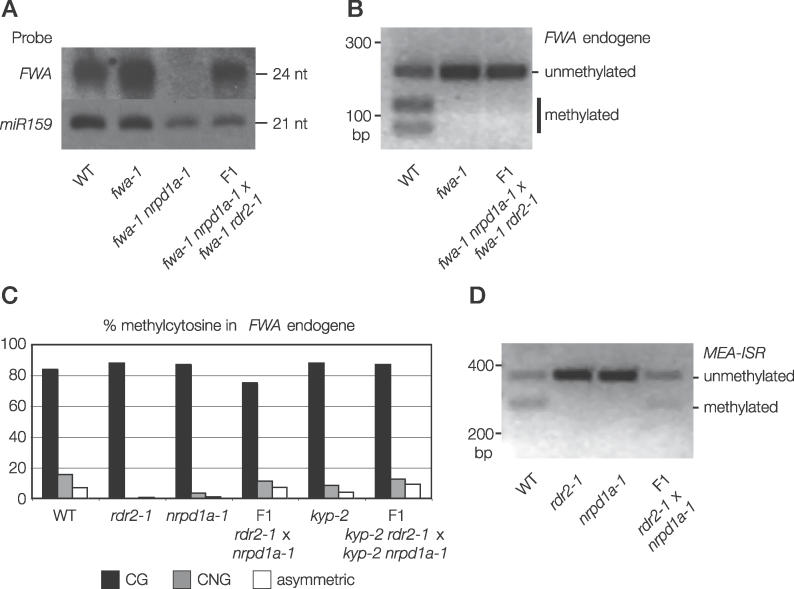
Figure 4. Characterization of FWA siRNAs in Different Genetic Backgrounds.
(A) Schematic diagram of the FWA locus and position of LNA probe used to detect siRNAs.
(B) FWA siRNAs are still produced when the gene is unmethylated. siRNAs from wild type, fwa-1, and met1–3 were analyzed by Northern blotting. miR159 was probed as a loading control.
(C) FWA siRNAs from plants with the indicated genotype were analyzed by Northern blotting.
(D) siRNAs in fwa-1 do not cause mRNA destruction. The ratio of FWA to ACTIN7 mRNA levels in rosette leaf tissue was measured by RT-qPCR. Reverse transcription was performed with a poly-T oligonucleotide. There was no PCR amplification when reverse transcriptase was omitted (unpublished data).
We initially hypothesized that differences in siRNA production might explain why methylated FWA in wild-type cells can enhance de novo methylation of transformed FWA, whereas unmethylated endogenous fwa does not. However, instead we found that fwa-1 plants contain FWA 24-nucleotide siRNAs at levels comparable to wild type (Figure 4B). We confirmed the existence of FWA siRNAs that depend on RDR2 and are present in fwa-1 by using two additional LNA probes from nonoverlapping regions of the tandem repeats (Figure S2). As further confirmation that siRNAs are produced from the unmethylated FWA gene, we also measured FWA siRNAs in a met1–3 DNA methyltransferase mutant that had lost all FWA DNA methylation and displayed a late-flowering phenotype [10,21]. Although it was previously reported using conventional probes that FWA siRNAs were reduced in met1 [4], we found using LNA probes that similar levels of FWA siRNAs were detected in met1 and wild-type strains (Figure 4B). Our observation that siRNAs are produced from both unmethylated fwa and methylated FWA is interesting, because only plants containing a methylated FWA endogene can efficiently de novo methylate an incoming transgene. One possibility is that siRNAs produced at methylated FWA are qualitatively different from those at unmethylated fwa. For example, they might be associated with different downstream effector complexes.
FWA siRNA Production Relies on only a Subset of Factors Required for FWA De Novo Methylation
To gain further insight into siRNA production at tandem repeats, we tested for the presence of siRNAs in a battery of mutants that are defective for FWA de novo methylation. We previously showed that mutations in four RNA silencing genes—NRPD1a, RDR2, DCL3, and AGO4—phenocopy the FWA de novo methylation defect of the DNA methyltransferase mutant drm2 [14]. We have more recently found that mutations in two other genes share this phenotype. The first is NRPD1b. DNA-dependent RNA polymerase IV exists in two forms in Arabidopsis [19,22–24]. Both share the small subunit encoded by NRPD2a but have different large subunits encoded by either NRPD1a or by NRPD1b. We found that, similar to the other RNA silencing mutants, an nrpd1b-11 T-DNA mutation blocks de novo methylation and silencing of transformed FWA [25]. The second new gene involved in FWA de novo DNA methylation is DRD1, which was isolated in a screen for mutants that lack RNA-directed DNA methylation triggered by a transgenic inverted repeat, and encodes a SNF2-like putative chromatin remodeling protein [26]. The drd1–6 allele [26] again blocked FWA de novo methylation and silencing [27].
We observed that FWA siRNA hybridization signal was absent in nrpd1a-1, rdr2–1, and dcl3–1 (Figure 4C). However, we observed 24-nucleotide FWA siRNA signals similar to wild-type levels in the nrpd1b-11, ago4–1, drd1–6, and drm1–1 drm2–1 mutants, all of which are defective for FWA de novo DNA methylation (Figure 4C). These data suggest that NRPD1a, RDR2 and DCL3 act in the initial formation of siRNAs, whereas NRPD1b, AGO4, DRD1, and DRM2 may act at more downstream steps of RNA-directed DNA methylation. Our finding that FWA siRNAs are stable in the absence of AGO4 is particularly interesting, because ARGONAUTE proteins are the siRNA binding moieties in RNAi pathways [13]. It is possible that other Arabidopsis ARGONAUTE proteins bind to FWA siRNAs in ago4–1 mutant plants, preserving their stability (the Arabidopsis genome encodes ten ARGONAUTE proteins). If this is the case, however, these alternative ARGONAUTEs are not capable of replacing the function of AGO4 in RNA-directed DNA methylation. The observation that NRPD1a is required for FWA siRNA production, yet NRPD1b is not, supports the conclusion of Kanno et al. and Pontier et al. that the two forms of RNA Pol IV play distinct roles in the process of RNA-directed DNA methylation [19,23]. The behavior of FWA siRNAs contrasts with that of siRNAs from the small euchromatic SINE transposon AtSN1, the levels of which are greatly reduced in ago4, nrpd1b, and drm1 drm2 mutants [19,28,29]; AtSN1 may be a locus where feedback from downstream DNA methylation is required to maintain siRNA production.
It is curious that NRPD1a/RDR2/DCL3–dependent FWA siRNAs are produced from unmethylated fwa alleles and yet do not cause DNA methylation. This prompted us to ask if these siRNAs instead might be causing post-transcriptional mRNA destruction. To test this, we used reverse transcription coupled to real-time quantitative PCR (RT-qPCR) to compare the levels of mRNA produced in either fwa-1 plants that produce FWA siRNAs or in fwa-1 nrpd1a-1 plants, where FWA siRNAs are undetectable (Figures 4D and 5A). FWA mRNA was present at equivalent levels in fwa-1 and fwa-1 nrpd1a-1, showing that the siRNAs present in fwa-1 are not diverted into a post-transcriptional gene-silencing pathway if DNA methylation is absent. Thus the function of FWA siRNAs in the unmethylated fwa-1 mutant remains obscure. Other endogenous Arabidopsis siRNAs match regions of the genome not known to contain DNA methylation [30], and the function of these are also unknown.
Figure 5. Separable Recruitment of siRNA Production and Non-CG DNA Methylation at Chromosomal Tandem Repeats.
(A) Unmethylated chromosomal tandem repeats can recruit siRNA production. fwa-1 nrpd1a-1 and fwa-1 rdr2–1 lack FWA siRNAs, but when these mutants are crossed together, Northern blotting shows that FWA siRNA production is restored in F1 plants.
(B) Unmethylated chromosomal tandem repeats cannot recruit de novo DNA methylation. When fwa-1 nrpd1a-1 and fwa-1 rdr2–1 mutants are crossed together, FWA remains unmethylated despite resumption of siRNA production. DNA methylation of FWA was assayed by PCR from bisulfite-treated DNA followed by ClaI digestion.
(C) CG DNA methylation recruits siRNA-directed non-CG DNA methylation to FWA. When the recessive rdr2–1 and nrpd1a-1 mutants are crossed together, non-CG DNA methylation returns to CG-methylated FWA in the F1 plants. DNA methylation was assayed by bisulfite sequencing.
(D) CG DNA methylation recruits siRNA-directed non-CG DNA methylation to MEA-ISR. Non-CG DNA methylation returns to CG-methylated MEA-ISR in rdr2–1 × nrpd1a-1 F1 plants. Asymmetric DNA methylation was assayed by PCR from bisulfite-treated DNA followed by BamHI digestion. DNA methylation protects the BamHI site from bisulfite conversion, allowing restriction digestion after bisulfite treatment.
Unmethylated Tandem Repeats Can Recruit siRNA Production without Downstream De Novo DNA Methylation
During Agrobacterium-mediated transformation, tandem repeats are recognized by siRNA-producing factors that guide DNA methylation, yet our fwa-1 results showed that an unmethylated gene can also produce siRNAs. To test whether unmethylated tandem repeats can recruit siRNA production independent of plant transformation, we first constructed two double mutants, fwa-1 nrpd1a-1 and fwa-1 rdr2–1, which lack DNA methylation at FWA and are also defective for siRNA production. We then crossed these plants together to reintroduce wild-type NRPD1a and RDR2. In the F1 of this cross, we observed that siRNA production at FWA immediately resumed (Figure 5A). This result shows directly that the FWA tandem repeats in their native chromosomal context can recruit Pol IVa, RDR2 and DCL3 activity to produce siRNAs independently of the DNA transport and integration steps of plant transformation. Interestingly, the fwa gene remained unmethylated in the F1 of the fwa-1 nrpd1a-1 × fwa-1 rdr2–1 cross (Figure 5B). This result shows that FWA siRNAs cannot cause de novo DNA methylation of unmethylated fwa in its native genomic context, even when their production is initiated afresh as it must be at a transformed copy of FWA.
Tandem Repeats with Pre-Existing CG DNA Methylation Recruit Both siRNA Production and RNA-Directed Non-CG Methylation
The inability of FWA siRNAs to de novo DNA methylate the fwa endogene can be contrasted with methylated FWA, where siRNAs persistently target non-CG DNA methylation. One feature of methylated FWA that may facilitate siRNA-directed DNA methylation is CG DNA methylation, which does not require siRNAs for its inheritance. We tested whether recruitment of siRNA production to CG-methylated FWA could target non-CG DNA methylation by crossing together homozygous nrpd1a-1 and rdr2–1 mutants, thereby reintroducing wild-type NRPD1A and RDR2. In nrpd1a and rdr2 (like in drm2), tandem repeats at FWA and MEDEA INTERGENIC SUBTELOMERIC REPEAT (MEA-ISR) lose their non-CG DNA methylation but fully retain CG methylation [14]. FWA remains silent in these plants (which have an early-flowering phenotype), because CG methylation is sufficient to suppress FWA expression. In nrpd1a-1 × rdr2–1 F1 plants, non-CG DNA methylation was immediately restored at both FWA and MEA-ISR (Figure 5C and 5D). In comparison, unmethylated fwa never spontaneously regains DNA methylation. These data suggest that CG methylation, or another chromatin modification that is associated with CG methylation, can recruit RNA silencing effector complexes and DRM2 to methylate non-CG sites. The fact that MEA-ISR behaves like FWA is important because it illustrates that recruitment of RNA-directed DNA methylation to tandem repeats is not a specialized property of a single gene.
Histone H3 lysine 9 dimethylation (H3K9me2) is a post-translational modification associated with gene silencing and has been shown to control non-CG DNA methylation in Arabidopsis [31,32]. Furthermore, H3K9me2 acts downstream of CG DNA methylation, because the majority of H3K9me2 is lost in a met1 mutant [33]. We therefore sought to test whether H3K9me2 might be involved in the recruitment of siRNA-directed non-CG methylation factors to CG methylated tandem repeats. The majority of H3K9me2 in Arabidopsis is maintained by the SET domain protein KRYPTONITE (KYP)/SUVH4, and the kyp-2 mutation strongly reduces H3K9me2 at the FWA tandem repeats [34]. We constructed double mutants of kyp-2 nrpd1a-1 and kyp-2 rdr2–1, then crossed these plants together to reintroduce wild-type NRPD1a and RDR2 in a background that is still homozygous for kyp-2. In the F1 of the kyp-2 nrpd1a-1 × kyp-2 rdr2–1 cross, asymmetric DNA methylation at FWA returned to the same levels as kyp-2 (Figure 5C). Thus, tandem repeats at FWA that have CG DNA methylation but lack KYP-dependent H3K9me2 are still capable of recruiting RNA-directed non-CG DNA methylation.
Tandem Repeat Sequences throughout the Arabidopsis Genome Are Enriched for siRNAs
Our results using FWA as a model provide evidence that tandem repeats recruit RNA silencing proteins to produce siRNAs. To assess the generality of these findings, we correlated tandem repeat sequences throughout the Arabidopsis genome with a large database of siRNAs cloned by massively parallel signature sequencing [18]. We used the program Tandem Repeats Finder [35] to define each tandem repeat in the genome with a repeat unit length greater than 20 bp (to avoid microsatellites). Tandem repeats that are found in multiple genomic locations may resemble transposons in that they could be sensed by a cellular mechanism devoted to high-copy dispersed repeats. We therefore filtered our tandem repeat library to remove any type of dispersed or inverted repeat character. We used BLAST (set to detect short, nearly perfect matches) to filter tandem repeats present at more than one genomic locus, RepBase to remove additional annotated transposons and repeats, and Inverted Repeats Finder [36] to exclude inverted repeats. Lastly, we restricted our analysis to tandem repeats outside of pericentromeric heterochromatin, because these should be less susceptible to silencing by adjacent transposons and high-copy sequences.
This preliminary analysis identified a set of 1494 “unique” tandem repeats from euchromatic regions of the Arabidopsis genome (Figure 6A), similar to the repeats found in FWA. We compared these to previously defined dense or moderate siRNA clusters, representing regions containing >11 cloned siRNAs without an intervening 500-bp gap [18] (single isolated siRNAs or sparse siRNA clusters were not considered in this study, because they might represent random matches). Thirty unique tandem repeats spread throughout the genome overlapped with siRNA clusters, representing 2.0% of unique tandem repeats. To compare unique tandem repeats with a relevant control sample of the Arabidopsis genome, we constructed a library of random 141-bp windows (the mean length of unique tandem repeats) from the euchromatic left arm of chromosome 1 using the same filtering procedure and threshold to remove repeated sequences or transposons. From a library of 5945 random windows, we found 14 that overlapped with dense or moderate siRNA clusters (0.24%). Thus, tandem repeats are nearly an order of magnitude more likely to produce siRNAs than random, nonrepeated sequences (Figure 6B). This enrichment is statistically significant, because the probability of selecting 30 siRNA-producing sequences at random was calculated at 1.0951 × 10−12 using a one-sided binomial distribution.
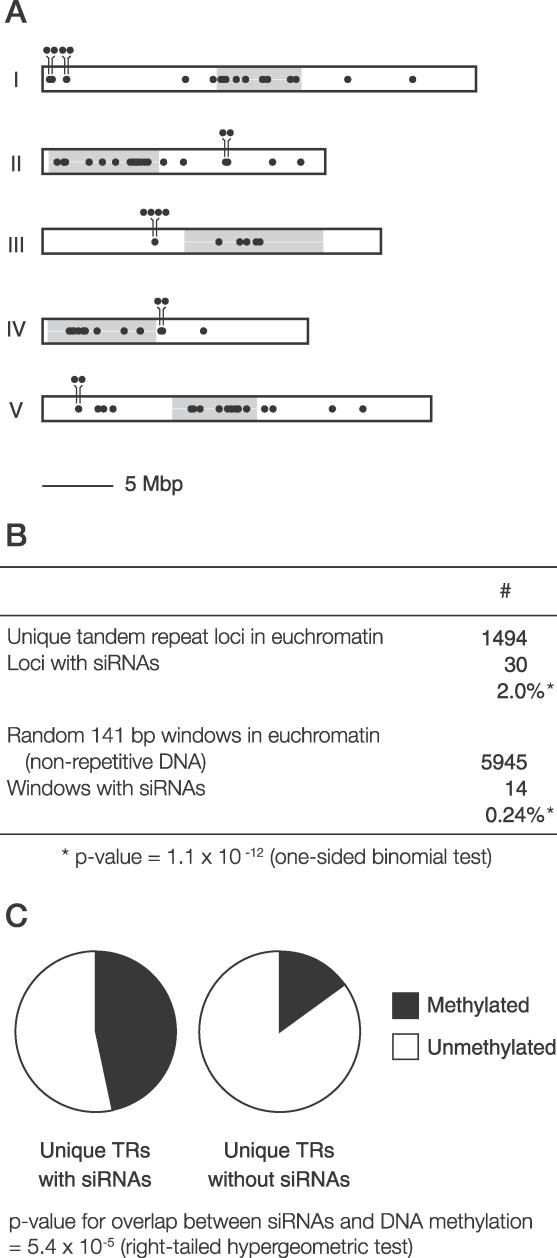
Figure 6. Association of siRNAs with Tandem Repeats throughout the Arabidopsis Genome.
(A) Chromosomal distribution of unique tandem repeats that produce siRNAs. Single-copy tandem repeats were identified (see text for details) and compared against a large database of cloned siRNAs [18]. The five Arabidopsis chromosomes are shown as rectangular boxes. Tandem repeats that overlap with dense or moderate siRNA clusters are shown as black dots (dots above the box represent euchromatic tandem repeats that were relatively close to each other). Pericentromeric heterochromatin is shown as gray shading.
(B) Unique tandem repeats in euchromatin are enriched for siRNA production relative to randomly chosen sequences. Unique tandem repeats and a set of random sequences of similar size (141 bp is the median size of unique tandem repeats) were assessed for overlap with dense or moderate siRNA clusters. The probability of selecting 30/1494 siRNA-producing loci by chance (assuming the same siRNA-producing frequency as randomly chosen windows) was calculated with a one-sided binomial test.
(C) DNA methylation of unique tandem repeats in euchromatin. Unique tandem repeats with and without siRNAs were compared to genome-wide DNA methylation data from an immunoprecipitation/tiling microarray experiment [37]. The fraction 221/1464 unique tandem repeats without siRNAs were methylated, compared with 14/30 unique tandem repeats that had siRNAs. The statistical significance of overlap between siRNA production and DNA methylation was calculated with a right-tailed hypergeometric test.
We next analyzed whether the presence of siRNAs was positively correlated with the presence of DNA methylation at unique tandem repeats, by comparing these loci to a genome-wide DNA methylation survey in which methylated DNA was immunoprecipitated and hybridized to Arabidopsis whole genome tiling microarrays (Figure 6C) [37]. Tandem repeats with siRNAs were methylated at a frequency of 46.7%, whereas only 15.1% of tandem repeats without siRNAs were methylated (similar to the genome average of 19%). This difference was highly statistically significant (Figure 6C). As tandem repeats with siRNAs are more frequently methylated than those without siRNAs, we infer that siRNA production may contribute to the maintenance of DNA methylation at these loci.
Because only a subset of unique tandem repeats produces siRNA, some other factors must determine whether stable siRNA production can exist at a given locus. We compared unique tandem repeats with and without siRNAs for several properties, including the overall length of the tandem repeat, repeat unit length, repeat copy number, average identity of repeat units, percentage of insertions and deletions (indels), and nucleotide sequence content. We found that tandem repeats with siRNAs were not significantly different in any of these characteristics (Figure S3). Thus, it may be that chromosomal context, some unknown feature of the DNA repeat sequence, or a particular set of chromatin modifications may influence which tandem repeats maintain siRNA production. We also analyzed the expression level of genes with unique tandem repeats in their promoters, and found that their expression was not significantly lower than those without tandem repeats (Figure S4). However, the importance of this result is unclear, because these tandem repeats may not affect promoter regions required for gene expression. Furthermore, tandem duplications in the genome may have been selected for those that do not repress important genes.
The euchromatic unique tandem repeats we analyzed represent only a small fraction of all tandem repeats that produce siRNAs. On a genome-wide scale, there is a strong correlation between tandem repeat character, siRNA production, and DNA methylation [4,18,37]; the vast majority of siRNA-producing tandem repeats are in pericentromeric heterochromatin, and are usually found at multiple genomic loci. Our results suggest that local repetitive nature is likely important in the initial recruitment of siRNA production to both unique and dispersed tandem repeats. Furthermore, continued siRNA production is correlated with the maintenance of DNA methylation. Thus, recognition of tandem repeats by RNA silencing proteins may be a general process for targeting gene silencing.
Discussion
Our results suggest a two-step model for de novo DNA methylation at tandem repeats (Figure 7). Most tandem repeats probably arise from single-copy sequences by internal duplication. The FWA tandem repeats were proposed to arise from a SINE transposon insertion [4]. However, plant SINE elements do not contain tandem repeats, and a secondary duplication event would therefore be necessary to create the current FWA 5′ untranslated region [38]. Additionally, the low level of similarity between the FWA tandem repeats and other Arabidopsis sequences (or any sequences in GenBank) suggests that other possibilities for their origin may be plausible. In particular, we propose a hypothetical yet parsimonious model in which the FWA tandem repeats could have arisen from an ancestral unrepeated sequence by a single internal duplication event (Figure 7). After their creation, tandem repeats appear to be preferential targets for siRNA-producing machinery, both at FWA and throughout the genome.
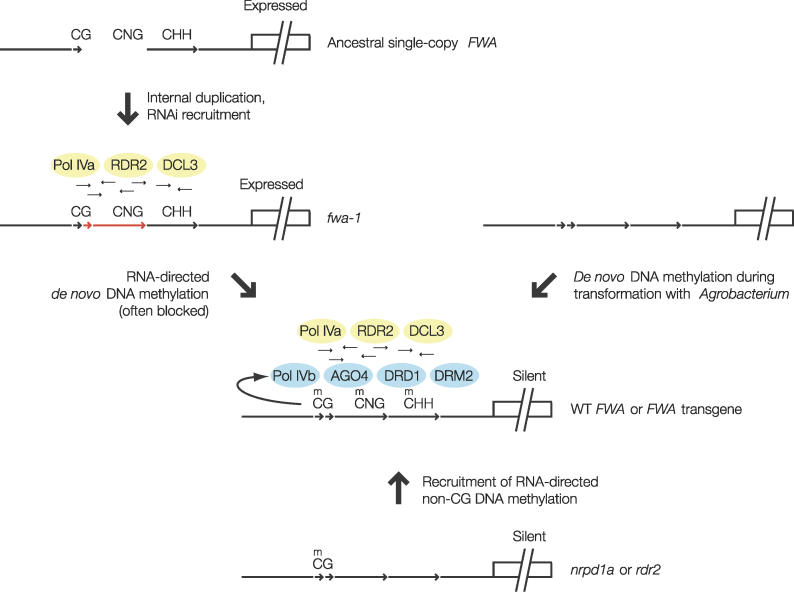
Figure 7. Model for RNA-Directed DNA Methylation at Tandem Repeats.
An ancestral single-copy gene may undergo internal duplication, creating tandem repeats that recruit siRNA-producing factors. After initial siRNA production, downstream RNA-directed DNA methylation may or may not occur efficiently at a particular locus, resulting in stable methylated or unmethylated genes, both of which produce siRNAs (small arrows). Transformed FWA efficiently recruits both siRNA production and downstream de novo DNA methylation. CG DNA methylation allows recruitment of siRNA-directed DNA methylation, providing a self-reinforcing feedback loop at silent loci.
During the first step in which siRNAs are produced from tandem repeats, it seems likely that NRPD1a-containing Pol IV produces a single-stranded RNA that is a substrate for RDR2, which then produces a double-stranded RNA that is diced by DCL3. How NRPD1a-Pol IV might initially recognize a tandem repeat however is not known. Robust siRNA production can occur at this stage without the presence of DNA methylation, showing that RNAi proteins recognize the intrinsic properties of tandem repeats without features of silent chromatin. In the second step, downstream DNA methylation factors use siRNAs to cause DNA methylation. Our discovery that recruitment of siRNA production is separable from downstream RNA-directed DNA methylation is consistent with previously published data showing the existence of unmethylated Arabidopsis loci that produce siRNAs [30].
NRPD1b-containing Pol IV is not required to produce siRNAs, and we feel its role may be to generate a single-stranded transcript that is bound by an AGO4/siRNA complex, which in turn recruits the DRM2 DNA methyltransferase to a particular genomic locus [19,23]. DRD1 may remodel chromatin, assisting the activity of DRM2. This model for siRNA-directed DNA methylation is attractive, because it does not require the AGO4/siRNA complex to unwind the chromosomal double helix and interact with homologous single-stranded DNA. It is also consistent with evidence from Schizosaccharomyces pombe showing that transcription by RNA Pol II plays a role in RNAi-mediated transcriptional gene silencing [39,40].
A key insight from these studies is that the ability of siRNAs to cause DNA methylation depends on the nature of the siRNA-producing locus. siRNAs produced from methylated and unmethylated forms of FWA appear to be made by the same upstream RNAi components. However, siRNAs from unmethylated fwa do not target de novo DNA methylation in cis, and lack the ability to enhance silencing of transformed FWA in trans. One possible explanation is that these siRNAs may not be loaded into AGO4 after they are released from the DCL3 nuclease, or that factors downstream of AGO4 are not recruited to unmethylated loci.
In contrast to unmethylated loci, tandem repeats with pre-existing CG DNA methylation are susceptible to RNA-directed DNA methylation when siRNA production is recruited. This is shown by the fact that non-CG DNA methylation by NRPD1b/AGO4/DRD1/DRM2 returns to FWA and MEA-ISR when RNAi proteins are restored in a nrpd1a × rdr2 cross (Figure 7). Exactly how CG DNA methylation facilitates RNA-directed DNA methylation is unclear. One possibility is that methyl-binding domain proteins interpret CG DNA methylation directly to recruit AGO4 and associated factors. Alternatively, another chromatin modification associated with DNA methylation could be key. The Arabidopsis genome contains many sites of CG-only DNA methylation, particularly in the coding region of genes [41]. One hypothesis that may explain why these sites do not recruit the non-CG methylation machinery is that CG-only sites lack tandem repeats and hence do not produce siRNAs. Although the maintenance methyltransferase enzyme MET1 is capable of maintaining CG-only regions of DNA methylation though a passive maintenance mechanism, tandem repeats that have both CG maintenance DNA methylation and persistent siRNA-directed non-CG DNA methylation may be more likely to retain their silent status over evolutionary time scales.
The process of plant transformation can also facilitate RNA-directed DNA methylation, because an FWA gene introduced by Agrobacterium is efficiently de novo methylated upon integration. Transformation also allows incoming FWA to cause de novo DNA methylation of the endogenous FWA gene (at a low frequency), bypassing the requirement for preexisting CG DNA methylation in recruiting RNA-directed DNA methylation. Agrobacterium introduces its T-DNA into plant cells as a single-stranded DNA molecule, which is copied into double-stranded DNA before its integration into the plant genome [42]. Multiple steps in this pathway could be responsible for potentiating siRNA-directed de novo DNA methylation. For example, increased siRNA levels could be a key factor associated with plant transformation. The effect of very high siRNA levels on de novo DNA methylation is shown by highly transcribed inverted repeat transgenes that target DNA methylation to homologous sequences independent of plant transformation [26]. Alternatively, a newly integrated transgene may possess a chromatin state that is permissive for RNA-directed DNA methylation, perhaps because of a particular set of histone modifications or lower nucleosome density. Such a permissive state might be maintained for several cell divisions after transformation (consistent with our observation that de novo DNA methylation occurs after fertilization of the female egg cell). A similarly susceptible state might occur for newly mobilized transposons.
Tandem repeats within a chromosome can increase their copy number by unequal crossing over or by gene conversion. Because these processes involve DNA breakage and rejoining reactions, they may act in the same way as Agrobacterium transformation to recruit molecular components that use siRNAs to cause de novo DNA methylation. In this way, the presence of siRNAs at unmethylated tandem repeats may potentiate DNA methylation during such expansions, or during periods of genome stress when chromatin is perturbed. Most tandem repeats are found at more than one genomic location and may be associated with transposons. In these cases, siRNA production may act as a kind of “immune memory” of dispersed repeats, allowing de novo DNA methylation to respond when selfish DNAs increase their copy number. The example of maize paramutation shows that tandem repeats in their natural context can indeed silence other genes in this manner [5].
Our findings also highlight the molecular mystery of how tandem repeats are initially recognized by RNAi proteins. Double-stranded RNAs that provide a Dicer substrate can be produced from inverted repeats by monodirectional transcription and fold-back, or from high-copy sequences by transcription of dispersed repeats in opposite directions. However, the mechanism that distinguishes unique tandem repeats from single copies of the same sequence remains unclear. One possibility is that NRPD1a-containing Pol IV and/or RDR2 may recognize unusual DNA or RNA structures that can only be made by tandem repeats. Regardless of the exact recognition mechanisms involved, the cellular RNAi machinery may be poised to produce siRNAs from newly created tandem repeats. In addition to providing a defense against potentially harmful sequences, this molecular mechanism may facilitate the evolution of genes whose normal expression is controlled by siRNAs and DNA methylation.
Materials and Methods
Plant materials.
Plants were grown under continuous light. All mutants have been previously described [10,11,22,24,26,29–31,33].
FWA transformation and flowering time analysis.
FWA transformation was performed as described [11]. For heterozygous mutants experiments, early- and late-flowering time was determined by comparison to isogenic ecotypes (Figure S1).
FWA deletion analysis.
Unless noted, FWA deletions were within the context of the XbaI-HindIII fragment from −2124 to 3876 (where the start codon is at +1) and were cloned into pCAMBIA1300. An FWA transgene where a BglII site is changed to EcoRI has been described [27]. The “repeats deleted” transgene lacks −1064 to −564. The “single-copy” transgene lacks −1026 to −779. The FWA promoter lacking the open reading frame contains nucleotides −2124 to 0, placed before a GFP-GUS open reading frame and cloned into pCAMBIA1300 from XbaI to HindIII (GFP-GUS was derived from pBGWFS7) [43]. The “repeats only” construct includes nucleotides −1072 to −559, placed before GFP-GUS and cloned into pCAMBIA1300.
Bisulfite sequencing and bisulfite PCR/restriction enzyme digestion assays.
Bisulfite sequencing was performed as described [14,28]. For “single-copy” FWA, the region analyzed had the same boundaries as full-length FWA (nucleotides −1055 to −608). Bisulfite sequencing data are summarized in Table S1. PCR primer sequences for bisulfite sequencing and bisulfite PCR/restriction digest assays are listed in Table S2.
Northern blot analysis of small RNAs.
Northern blots were performed as described [30]. The sequences of the FWA LNA probes used in this study are listed in Table S2.
RT-qPCR analysis of gene expression.
RNA was extracted from rosette leaves with TRIzol reagent (Invitrogen, Carlsbad, California, United States), DNAse treated with the DNA-free kit (Ambion, Austin, Texas, United States), and converted to cDNA with SuperScript II reverse transcriptase (Invitrogen) using a poly-T primer. FWA and ACT7 PCR primer sequences are listed in Table S2.
Supplementary Information
(126 KB PDF)
(558 KB PDF)
(233 KB PDF)
(86 KB PDF)
(79 KB PDF)
(26 KB PDF)
Acknowledgments
We thank Ian Henderson, Lianna Johnson, Jason Chien, Lauren Ivans, Govind Shah, and Ana Marie Palanca for technical assistance; Pam Green, Blake Meyers, and members of the Jacobsen lab for helpful discussions; Jim Carrington for information on LNA probes; and Matteo Pellegrini and Shawn Cokus for statistical assistance.
Abbreviations
- LNA
locked nucleic acid
- RNAi
RNA interference
- siRNA
small interfering RNA
Footnotes
¤ Current address: Section of Plant Biology, University of California Davis, Davis, California, United States of America
Competing interests. The authors have declared that no competing interests exist.
Author contributions. SWLC and SEJ conceived and designed the experiments. SWLC, XZ, and YVB performed the experiments. SWLC and SEJ analyzed the data. SWLC and XZ contributed reagents/materials/analysis tools. SWLC and SEJ wrote the paper.
Funding. This research was funded by NIH grant R01 GM060398. SEJ is an investigator of the Howard Hughes Medical Institute. SW-LC was a U.S. Department of Energy fellow of the Life Sciences Research Foundation.
References
- Bird A. DNA methylation patterns and epigenetic memory. Genes Dev. 2002;16:6–21. doi: 10.1101/gad.947102. [DOI] [PubMed] [Google Scholar]
- Chan SW, Henderson IR, Jacobsen SE. Gardening the genome: DNA methylation in Arabidopsis thaliana . Nat Rev Genet. 2005;6:351–360. doi: 10.1038/nrg1601. [DOI] [PubMed] [Google Scholar]
- Craig NL, Craigie RC, Gellert M, Lambowitz AM. Mobile DNA II. Washington (DC): ASM Press; 2002. 1182 [Google Scholar]
- Lippman Z, Gendrel AV, Black M, Vaughn MW, Dedhia N, et al. Role of transposable elements in heterochromatin and epigenetic control. Nature. 2004;430:471–476. doi: 10.1038/nature02651. [DOI] [PubMed] [Google Scholar]
- Stam M, Belele C, Dorweiler JE, Chandler VL. Differential chromatin structure within a tandem array 100 kb upstream of the maize b1 locus is associated with paramutation. Genes Dev. 2002;16:1906–1918. doi: 10.1101/gad.1006702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kinoshita T, Miura A, Choi Y, Kinoshita Y, Cao X, et al. One-way control of FWA imprinting in Arabidopsis endosperm by DNA methylation. Science. 2004;303:521–523. doi: 10.1126/science.1089835. [DOI] [PubMed] [Google Scholar]
- Reinhart B, Eljanne M, Chaillet JR. Shared role for differentially methylated domains of imprinted genes. Mol Cell Biol. 2002;22:2089–2098. doi: 10.1128/MCB.22.7.2089-2098.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoon BJ, Herman H, Sikora A, Smith LT, Plass C, et al. Regulation of DNA methylation of Rasgrf1 . Nat Genet. 2002;30:92–96. doi: 10.1038/ng795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hutter B, Helms V, Paulsen M. Tandem repeats in the CpG islands of imprinted genes. Genomics. 2006;88:323–332. doi: 10.1016/j.ygeno.2006.03.019. [DOI] [PubMed] [Google Scholar]
- Soppe WJ, Jacobsen SE, Alonso-Blanco C, Jackson JP, Kakutani T, et al. The late flowering phenotype of fwa mutants is caused by gain-of-function epigenetic alleles of a homeodomain gene. Mol Cell. 2000;6:791–802. doi: 10.1016/s1097-2765(05)00090-0. [DOI] [PubMed] [Google Scholar]
- Cao X, Jacobsen SE. Role of the arabidopsis DRM methyltransferases in de novo DNA methylation and gene silencing. Curr Biol. 2002;12:1138–1144. doi: 10.1016/s0960-9822(02)00925-9. [DOI] [PubMed] [Google Scholar]
- Cao X, Jacobsen SE. Locus-specific control of asymmetric and CpNpG methylation by the DRM and CMT3 methyltransferase genes. Proc Natl Acad Sci U S A. 2002;99:16491–16498. doi: 10.1073/pnas.162371599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meister G, Tuschl T. Mechanisms of gene silencing by double-stranded RNA. Nature. 2004;431:343–349. doi: 10.1038/nature02873. [DOI] [PubMed] [Google Scholar]
- Chan SW, Zilberman D, Xie Z, Johansen LK, Carrington JC, et al. RNA silencing genes control de novo DNA methylation. Science. 2004;303:1336. doi: 10.1126/science.1095989. [DOI] [PubMed] [Google Scholar]
- Alleman M, Sidorenko L, McGinnis K, Seshadri V, Dorweiler JE, et al. An RNA-dependent RNA polymerase is required for paramutation in maize. Nature. 2006;442:295–298. doi: 10.1038/nature04884. [DOI] [PubMed] [Google Scholar]
- Desfeux C, Clough SJ, Bent AF. Female reproductive tissues are the primary target of Agrobacterium-mediated transformation by the Arabidopsis floral-dip method. Plant Physiol. 2000;123:895–904. doi: 10.1104/pp.123.3.895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ye GN, Stone D, Pang SZ, Creely W, Gonzalez K, et al. Arabidopsis ovule is the target for Agrobacterium in planta vacuum infiltration transformation. Plant J. 1999;19:249–257. doi: 10.1046/j.1365-313x.1999.00520.x. [DOI] [PubMed] [Google Scholar]
- Lu C, Tej SS, Luo S, Haudenschild CD, Meyers BC, et al. Elucidation of the small RNA component of the transcriptome. Science. 2005;309:1567–1569. doi: 10.1126/science.1114112. [DOI] [PubMed] [Google Scholar]
- Pontier D, Yahubyan G, Vega D, Bulski A, Saez-Vasquez J, et al. Reinforcement of silencing at transposons and highly repeated sequences requires the concerted action of two distinct RNA polymerases IV in Arabidopsis . Genes Dev. 2005;19:2030–2040. doi: 10.1101/gad.348405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valoczi A, Hornyik C, Varga N, Burgyan J, Kauppinen S, et al. Sensitive and specific detection of microRNAs by northern blot analysis using LNA-modified oligonucleotide probes. Nucleic Acids Res. 2004;32:e175. doi: 10.1093/nar/gnh171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saze H, Scheid OM, Paszkowski J. Maintenance of CpG methylation is essential for epigenetic inheritance during plant gametogenesis. Nat Genet. 2003;34:65–69. doi: 10.1038/ng1138. [DOI] [PubMed] [Google Scholar]
- Herr AJ, Jensen MB, Dalmay T, Baulcombe DC. RNA polymerase IV directs silencing of endogenous DNA. Science. 2005;308:118–120. doi: 10.1126/science.1106910. [DOI] [PubMed] [Google Scholar]
- Kanno T, Huettel B, Mette MF, Aufsatz W, Jaligot E, et al. Atypical RNA polymerase subunits required for RNA-directed DNA methylation. Nat Genet. 2005;37:761–765. doi: 10.1038/ng1580. [DOI] [PubMed] [Google Scholar]
- Onodera Y, Haag JR, Ream T, Nunes PC, Pontes O, et al. Plant nuclear RNA polymerase IV mediates siRNA and DNA methylation-dependent heterochromatin formation. Cell. 2005;120:613–622. doi: 10.1016/j.cell.2005.02.007. [DOI] [PubMed] [Google Scholar]
- Li CF, Pontes O, El-Shami M, Henderson IR, Chan SW, et al. An ARGONAUTE4-containing nuclear processing center co-localized with Cajal bodies in Arabidopsis thaliana . Cell. 2006;126:93–106. doi: 10.1016/j.cell.2006.05.032. [DOI] [PubMed] [Google Scholar]
- Kanno T, Mette MF, Kreil DP, Aufsatz W, Matzke M, et al. Involvement of putative SNF2 chromatin remodeling protein DRD1 in RNA-directed DNA methylation. Curr Biol. 2004;14:801–805. doi: 10.1016/j.cub.2004.04.037. [DOI] [PubMed] [Google Scholar]
- Chan SW, Henderson IR, Zhang X, Shah G, Chien JS, et al. RNAi, DRD1, and Histone methylation actively target developmentally important non-CG DNA methylation in Arabidopsis . PLoS Genet. 2006; 2((6)): e83. doi: 10.1371/journal.pgen.0020083. DOI: 10.1371/journal.pgen.0020083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zilberman D, Cao X, Jacobsen SE. ARGONAUTE4 control of locus-specific siRNA accumulation and DNA and histone methylation. Science. 2003;299:716–719. doi: 10.1126/science.1079695. [DOI] [PubMed] [Google Scholar]
- Zilberman D, Cao X, Johansen LK, Xie Z, Carrington JC, et al. Role of Arabidopsis ARGONAUTE4 in RNA-directed DNA methylation triggered by inverted repeats. Curr Biol. 2004;14:1214–1220. doi: 10.1016/j.cub.2004.06.055. [DOI] [PubMed] [Google Scholar]
- Xie Z, Johansen LK, Gustafson AM, Kasschau KD, Lellis AD, et al. Genetic and functional diversification of small RNA pathways in plants. PLoS Biol . 2004;2((5)): E104. doi: 10.1371/journal.pbio.0020104. DOI: 10.1371/journal.pbio.0020104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jackson JP, Lindroth AM, Cao X, Jacobsen SE. Control of CpNpG DNA methylation by the KRYPTONITE histone H3 methyltransferase. Nature. 2002;416:556–560. doi: 10.1038/nature731. [DOI] [PubMed] [Google Scholar]
- Malagnac F, Bartee L, Bender J. An Arabidopsis SET domain protein required for maintenance but not establishment of DNA methylation. EMBO J. 2002;21:6842–6852. doi: 10.1093/emboj/cdf687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tariq M, Saze H, Probst AV, Lichota J, Habu Y, et al. Erasure of CpG methylation in Arabidopsis alters patterns of histone H3 methylation in heterochromatin. Proc Natl Acad Sci U S A. 2003;100:8823–8827. doi: 10.1073/pnas.1432939100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson L, Mollah S, Garcia BA, Muratore TL, Shabanowitz J, et al. Mass spectrometry analysis of Arabidopsis histone H3 reveals distinct combinations of post-translational modifications. Nucleic Acids Res. 2004;32:6511–6518. doi: 10.1093/nar/gkh992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benson G. Tandem repeats finder: A program to analyze DNA sequences. Nucleic Acids Res. 1999;27:573–580. doi: 10.1093/nar/27.2.573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warburton PE, Giordano J, Cheung F, Gelfand Y, Benson G. Inverted repeat structure of the human genome: The X-chromosome contains a preponderance of large, highly homologous inverted repeats that contain testes genes. Genome Res. 2004;14:1861–1869. doi: 10.1101/gr.2542904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang X, Yazaki J, Sundaresan A, Cokus S, Chan SW, et al. Genome-wide high-resolution mapping and functional analysis of DNA methylation in Arabidopsis . Cell. 2006. In press. [DOI] [PubMed]
- Deragon JM, Zhang X. Short interspersed elements (SINEs) in plants: Origin, classification and use as phylogenetic markers. Syst Biol. 2006. In press. [DOI] [PubMed]
- Kato H, Goto DB, Martienssen RA, Urano T, Furukawa K, et al. RNA polymerase II is required for RNAi-dependent heterochromatin assembly. Science. 2005;309:467–469. doi: 10.1126/science.1114955. [DOI] [PubMed] [Google Scholar]
- Djupedal I, Portoso M, Spahr H, Bonilla C, Gustafsson CM, et al. RNA Pol II subunit Rpb7 promotes centromeric transcription and RNAi-directed chromatin silencing. Genes Dev. 2005;19:2301–2306. doi: 10.1101/gad.344205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tran RK, Henikoff JG, Zilberman D, Ditt RF, Jacobsen SE, et al. DNA methylation profiling identifies CG methylation clusters in Arabidopsis genes. Curr Biol. 2005;15:154–159. doi: 10.1016/j.cub.2005.01.008. [DOI] [PubMed] [Google Scholar]
- Gelvin SB. Agrobacterium and plant genes involved in T-DNA transfer and integration. Annu Rev Plant Physiol Plant Mol Biol. 2000;51:223–256. doi: 10.1146/annurev.arplant.51.1.223. [DOI] [PubMed] [Google Scholar]
- Karimi M, De Meyer B, Hilson P. Modular cloning in plant cells. Trends Plant Sci. 2005;10:103–105. doi: 10.1016/j.tplants.2005.01.008. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(126 KB PDF)
(558 KB PDF)
(233 KB PDF)
(86 KB PDF)
(79 KB PDF)
(26 KB PDF)