Abstract
Aims: To determine any associations between the Helicobacter pylori genes babA2, oipA, cagA and the s and m alleles of vacA. In addition, to verify whether these genes work synergistically or independently in causing gastritis, peptic ulcer, and intestinal metaplasia.
Methods: One hundred and sixty seven H pylori positive patients were studied (52 antral gastritis, 41 diffuse gastritis, 41 peptic ulcer, and 33 duodenitis). Helicobacter pylori virulence genes were amplified by means of the polymerase chain reaction.
Results: Significant associations were found between babA2 and the other H pylori genes studied. When considered singly, all the genes were associated with disease diagnosis, inflammation, and intestinal metaplasia. Four H pylori groups were defined. Group A: cagA−, s2m2, babA2−; group B: cagA+, s1m1, babA2+; group C: cagA+, s1m2, babA2+; group D: cagA+, s1m2, babA2−. Group A infecting strains were associated with less severe endoscopic and inflammatory conditions, whereas group B strains were associated with the worst endoscopic and inflammatory findings. Intestinal metaplasia was a rare finding in group A infected patients (< 10%), whereas it was frequent in those infected with group B strains (48%).
Conclusions: The H pylori genes cagA, oipA “on”, s1 and m1 vacA, and babA2 are associated with each other, possibly as a result of shared selective pressure. When coexpressed by the same H pylori strain, cagA, s1 and m1 vacA, and babA2 work synergistically in worsening inflammation. Infections caused by strains coexpressing cagA, s1m1 vacA, and babA2 are those at higher risk for intestinal metaplasia.
Keywords: babA2, intestinal metaplasia, cagA, vacA
The H pylori genome consists of a conserved core of more than 1000 genes that encode functional proteins and strain specific genes, which may encode adaptation to genetically diverse hosts.1 The data indicate that: (1) quasi-species coexist within the H pylori population infecting one individual; (2) H pylori can lose and acquire exogenous DNA, thus achieving a model of continuous microevolution, which is characterised by a very high recombination rate during mixed colonisation by unrelated strains; (3) there is a very high mutational frequency.1–6 Therefore, an effort must now be made to clarify the clinical relevance of the main H pylori virulence factors, to understand more fully the exact role of each in determining the outcome of an infection known to be responsible for a spectrum of diseases ranging from gastritis, to peptic ulcer, to gastric adenocarcinoma and mucosal associated lymphoid tissue lymphoma.7–13
Two steps can be identified in the pathogenesis of H pylori gastric mucosa colonisation: (1) adhesion to the epithelial cell layer and (2) the induction of proinflammatory cytokine release.14–16
Adhesion to gastric epithelial cells is mediated by H pylori adhesins, among which BabA binds the Lewis b epitope of the host, and this molecule has been suggested to play a crucial role in the development of gastric adenocarcinoma, peptic ulcer, and severe gastritis.17–23 BabA is encoded by a polymorphic gene named babA2.21 The release of proinflammatory cytokines from the gastric mucosa—including interleukin 1β (IL-1β), IL-6, and IL-8—is mainly induced by genes, like cagA, located in the pathogenicity island (PAI).14,16,24–26 It was recently demonstrated in vitro that some H pylori genes located outside the PAI—oipA in particular—can also induce the mucosal release of IL-8.27
Helicobacter pylori strains that posses the PAI in their genome usually also contain the s1 and m1 alleles of the vacA gene and can produce an active vacuolating cytotoxin.7,8,12,16,28–31
“The release of proinflammatory cytokines from the gastric mucosa is mainly induced by genes, like cagA, located in the pathogenicity island”
The aims of our present study were: (1) to define a polymerase chain reaction (PCR) method for the identification of babA2 possessing strains, comparing it with methods previously described in the literature; (2) to determine any associations between the H pylori virulence genes babA2, oipA, and cagA and the s and m alleles of vacA; and (3) to verify whether the different H pylori virulence genes cooperate or act singly in causing gastritis, peptic ulcer, and intestinal metaplasia.
METHODS
Patients
The series consisted of 167 H pylori positive patients (78 men, 89 women; age range, 17 to 94 years) who consecutively underwent upper gastrointestinal endoscopy. Diagnoses, made on the basis of endoscopic findings, were: antral gastritis only (52), diffuse gastritis (41), duodenal ulcer (33), gastric ulcer (eight), and duodenitis (33). All patients with duodenal ulcer, gastric ulcer, or duodenitis also had antral gastritis.
At endoscopy, three antral and two body biopsies were obtained from each patient for the histological evaluation. Two antral biopsies were also obtained for H pylori culture.
Histological evaluation
The following histological parameters were semiquantitatively scored, as described previously32,33: (1) bacterial peak density grade (0, absent; 1 to 3, from few and isolated bacteria to colonies); (2) polymorphonuclear cell infiltrate (activity) (0, absent; 1 to 3, from mild to severe); (3) mononuclear cell infiltrate (chronic inflammation) (0, absent; 1 to 6, from mild superficial to severe glandular); (4) intestinal metaplasia (0, absent; 1 to 3, type I to type III).
Helicobacter pylori culture and babA2, ureA, cagA, and vacA amplification
Helicobacter pylori culture and DNA extraction from positive colonies were carried out as described previously.33
The babA2 gene was PCR amplified under the conditions described by Gerhard et al.17 The sense (BABA2F) and antisense (BABA2R) primers were: AAT CCA AAA AGG AGA AAA AGT ATG AAA and TGT TAG TGA TTT CGG TGT AGG ACA, respectively. A second protocol for babA2 amplification (designed by us) used the forward primer described by Gerhard et al (BABA2F) and the reverse primer BABA2R607 (CTT TGA GCG CGG GTA AGC). A 3 μl aliquot of extracted DNA was used for babA2 amplification in a 25 μl reaction volume containing: 1× Taq polymerase buffer and 1.5 U Taq polymerase (Promega, Madison, Wisconsin, USA), 1.5mM MgCl2, 200μM each dNTP, and 500nM each primer.
The cycling conditions were: (1) 95°C for two minutes; (2) five cycles at 95°C for 30 seconds, 61°C for 30 seconds, 72°C for one minute; (3) 40 cycles at 95°C for 30 seconds, 55°C for 30 seconds, and 72°C for one minute; and (4) 72°C for seven minutes.
PCR products (832 bp after BABA2R and BABA2F; 607 bp after BABA2F and BABA2R607 amplification) were separated by electrophoresis on a 1.3% agarose gel and stained with ethidium bromide. Four 607 bp positive samples were subjected to sequence analysis under the following conditions: the purified PCR products (Millipore, Billerida, Massachusetts, USA) were subjected to fluorescence dye terminator cycle sequencing (Applied Biosystems, Foster City, California, USA).
The ureA, vacA, and cagA genes were PCR amplified as described previously.7,8,12,33
oipA gene sequence analysis
Helicobacter pylori oipA (HP0638) signal sequences were PCR amplified and sequenced in 60 clinical isolates.27,33
Statistical analysis
The data were analysed by means of the χ2 test and the Mann Whitney U test.
RESULTS
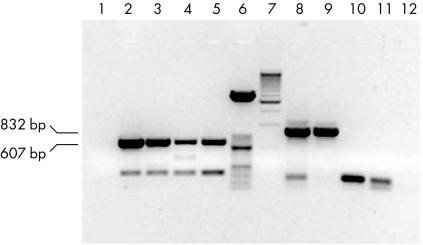
Figure 1 shows agarose gel electrophoresis of babA2 amplification products obtained using the primer pairs BABA2F/BABA2R and BABA2F/BABA2R607. Using these two primer pairs, 832 bp and 607 bp products were obtained, respectively. The figure shows negative controls and isolates from four patients. Two isolates, negative after amplification with the primer pair BABA2F/BABA2R, were positive after amplification with the primer pair BABA2F/BABA2R607. All the four positive isolates were sequenced and were found to correspond exactly to the babA2 gene sequence (Gene Bank accession number, AF033654).
Figure 1.
Agarose gel electrophoresis of babA2 PCR products obtained from the clinical isolates of four patients using the primer pairs BABA2F/BABA2R607 (lanes 2–5, 607 bp amplicons) and BABA2F/BABA2R (lanes 8–11, 832 bp amplicons). Lanes 1 and 12, negative controls of BABA2F/BABA2R607 and BABA2F/BABA2R PCR, respectively. Lane 6, 50 bp molecular weight marker; lane 7, 500 bp molecular weight marker.
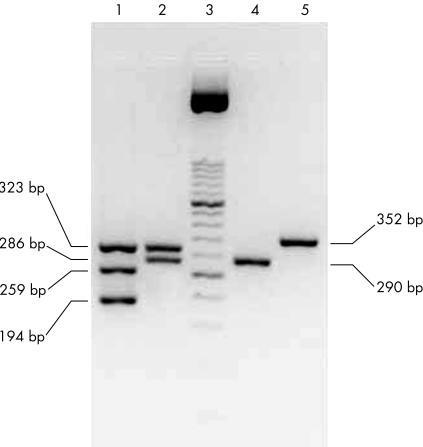
Figure 2 shows cagA positive and negative strains, in addition to s and m vacA alleles obtained from different clinical isolates after agarose gel electrophoresis.
Figure 2.
UreaseA (323 bp), cagA (194 bp), and the s (s1, 259 bp; s2, 286 bp) and m (m1, 290 bp; m2, 352 bp) vacA alleles after agarose gel electrophoresis. UreaseA, cagA, and the s1 and s2 vacA alleles were coamplified in the first PCR round (lines 1 and 2); m1 and m2 vacA alleles were coamplified in the second PCR round (lines 4 and 5). Two clinical isolates are shown: lines 1 and 4, Helicobacter pylori first clinical isolate (cagA+, s1m1 vacA); lines 2 and 5, H pylori second clinical isolate (cagA−, s2m2 vacA); line 3, 50 bp molecular weight marker. Primers and amplification conditions were those previously described by us.34
Table 1 shows the number (and relative percentages) of babA2 positive H pylori isolates obtained after PCR amplification using the primer pairs BABA2F/BABA2R and BABA2F/BABA2R607. The subsequent analysis of data was carried out taking into account the results obtained with the primer pair BABA2F/BABA2R607.
Table 1.
Number and percentage of positive babA2 Helicobacter pylori clinical isolates after polymerase chain reaction (PCR) amplification
| BABA2F/BABA2R | ||
| Positive (%) | Negative (%) | |
| BABA2F/BABA2R607 | ||
| Positive | 40 (24%) | 20 (12%) |
| Negative | 0 | 107 (64%) |
PCR used the primer pair BABA2F/BABA2R6 or the primer pair BABA2F/BABA2R607 (designed by us). In total, 167 isolates were studied.
The presence of the babA2 gene was significantly associated with all the other H pylori virulence genes studied (cagA, vacA, and oipA) (table 2).
Table 2.
Associations between babA2 and oipA, cagA, and the s and m alleles of vacA
| babA2 | ||
| Positive | Negative | |
| oipA | ||
| On | 17 | 19 |
| Off | 3 | 21 |
| χ2=7.81; p<0.01 | ||
| cagA | ||
| Positive | 57 | 48 |
| Negative | 3 | 59 |
| χ2=42.39; p<0.001 | ||
| s vacA | ||
| s1 | 57 | 48 |
| s2 | 3 | 59 |
| χ2=42.39; p<0.001 | ||
| m vacA | ||
| m1 | 30 | 34 |
| m2 | 29 | 74 |
| χ2=5.99; p<0.05 | ||
The number of Helicobacter pylori isolates belonging to each category is reported. The data were analysed by means of the χ2 test.
The presence of cagA was significantly associated not only with babA2, but also with oipA (χ2 = 38.57; p < 0.001), and the s1 (χ2 = 139.4; p < 0.001) and m1 alleles of vacA (χ2 = 57.2; p < 0.001). Helicobacter pylori cagA negative isolates were also babA2 negative (95.2%), oipA “off” (100%), and had the s2 (100%) and m2 (98.4%) vacA alleles, whereas H pylori cagA positive isolates were babA2 positive (52.8%), oipA “on” (85.7%), and had the s1 (100%) and m1 (61.5%) vacA alleles.
babA2, cagA, and s1 vacA were significantly more frequent in patients with diffuse gastritis, peptic ulcer, or duodenitis than in patients with antral gastritis only (table 3).
Table 3.
Association between Helicobacter pylori genes and endoscopic findings
| Antral gastritis | Diffuse gastritis | Peptic ulcer | Duodenitis | |
| babA2 | ||||
| Positive | 9 | 17 | 20 | 14 |
| Negative | 43 | 24 | 21 | 19 |
| χ2=11.8; p<0.05 | ||||
| oipA | ||||
| Positive | 5 | 10 | 14 | 7 |
| Negative | 7 | 9 | 5 | 3 |
| χ2=4.0; NS | ||||
| cagA | ||||
| Positive | 20 | 26 | 32 | 27 |
| Negative | 32 | 15 | 9 | 6 |
| χ2=22.4; p<0.001 | ||||
| s vacA | ||||
| s1 | 20 | 26 | 32 | 27 |
| s2 | 32 | 15 | 9 | 6 |
| χ2=22.4; p<0.01 | ||||
| m vacA | ||||
| m1 | 12 | 20 | 17 | 14 |
| m2 | 40 | 21 | 24 | 19 |
| χ2=6.6; NS | ||||
The number of patients belonging to each category is reported. The data were analysed by means of the χ2 test, comparing each virulence gene in the 8 reported groups.
The antral H pylori colonisation grade was significantly associated with cagA (χ2 = 9.83; p < 0.05) and the m1 vacA allele (χ2 = 13.55; p < 0.01), but not with the s1 vacA allele (χ2 =6.34, p = not significant (NS)), babA2 (χ2 = 2.17; p = NS), or oipA “on” (χ2 = 4.30; p = NS).
Antral inflammation and activity were significantly associated with cagA (χ2 = 17.84; p < 0.01 and χ2 = 10.10; p < 0.05, respectively) and the s1 (χ2 = 14.38; p < 0.05 and χ2 = 9.06; p < 0.05, respectively) vacA allele, but not with the other H pylori virulence genes.
Intestinal metaplasia was significantly associated with babA2 (χ2 = 4.45; p < 0.05), oipA “on” (χ2 = 6.47; p < 0.05), cagA (χ2 = 25.21; p < 0.001), and the s1 (χ2 = 21.11; p < 0.001) and m1 (χ2 = 15.63; p < 0.001) vacA alleles.
We subdivided patients into four groups on the basis of their genotype for the virulence factors babA2, cagA, and vacA alleles:
Group A (negative for all virulence genes): cagA−, s2m2, babA2−.
Group B (positive for all virulence genes): cagA+, s1m1, babA +.
Group C (positive for three virulence genes): cagA+, s1m2, babA2+.
Group D (positive for two virulence genes): cagA+, s1m2, babA2−.
oipA was excluded from the analysis because it was strictly linked to cagA and it was determined in a subgroup of 60 H pylori strains. The remaining possible combinations between babA2, cagA, and the vacA alleles were not considered because of their very low prevalence.
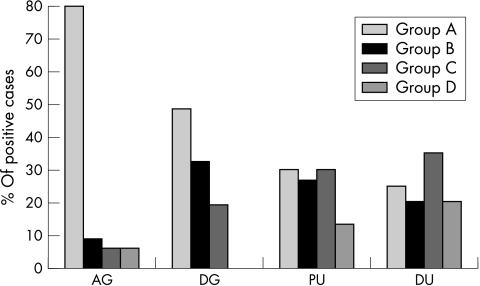
Figure 3 shows the proportion of H pylori infected patients with antral or diffuse gastritis, with peptic ulcer, or with duodenitis belonging to groups A to D (χ2 = 30.0; p < 0.001).
Figure 3.
Percentage of patients with antral gastritis (AG; n = 52), diffuse gastritis (DG; n = 41), peptic ulcer (PU; n = 41), or duodenitis (DU; n = 33), after dividing the infecting Helicobacter pylori strains into group A (cagA−, s2, m2, babA2−), group B (cagA+, s1m1, babA2+), group C (cagA+, s1m2, babA2+), and group D (cagA+, s1m2, babA2−).
Antral and body inflammation and H pylori density grade were more pronounced in patients infected with H pylori strains possessing all the virulence genes than in those with none of the virulence genes (table 4).
Table 4.
Results of antral Helicobacter pylori density grade, body H pylori density grade, antral inflammation, body inflammation, antral activity, and body activity
| H pylori infecting groups | AHPDG* Median (range) | AI* Median (range) | AA** Median (range) | BHPDG* Median (range) | BI* Median (range) | BA*, ** Median (range) |
| Group A | 2.00 (0–3) | 2.00 (1–6) | 2.00 (0–3) | 2.00 (0–3) | 2.00 (0–5) | 1.00 (0–3) |
| Group B | 2.0 (2–3) | 2.0 (1–5) | 2.00 (1–3) | 2.00 (2–3) | 2.00 (1–7) | 0 (0–1) |
| Group C | 2.00 (1–3) | 2.00 (1–5) | 2.00 (1–3) | 2.00 (1–5) | 2.00 (0–5) | 2.00 (0–2) |
| Group D | 2.00 (0–2) | 3.5 (2–5) | 2.00 (0–3) | 2.00 (0–2) | 1.5 (0–5) | 0.50 (0–2) |
Patients had been subdivided on the basis of H pylori infecting groups A to D. Group A: cagA−, s2, m2, babA2−; group B: cagA+, s1, m1, babA2+; group C: cagA+, s1, m2, babA2+; group D: cagA+, s1, m2, babA2−.
*p<0.05, group A v group B;
**p<0.05, group A v group C; Mann-Whitney test.
AA, antral activity, AHPDG, antral H pylori density grade; AI, antral inflammation; BA, body activity; BHPDG, body H pylori density grade; BI, body inflammation.
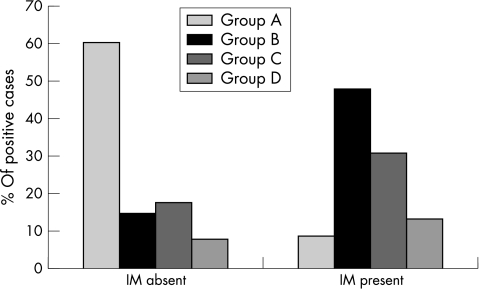
The risk estimates for intestinal metaplasia increased as the number of virulence genes in the H pylori infecting strain increased (fig 4; table 5).
Figure 4.
Percentage of patients infected with Helicobacter pylori strains belonging to groups A to D after they had been subdivided on the basis of the presence (n = 34) or absence (n = 133) of intestinal metaplasia (IM). Group A (cagA−, s2, m2, babA2−), group B (cagA+, s1m1, babA2+), group C (cagA+, s1m2, babA2+), and group D (cagA+, s1m2, babA2−).
Table 5.
Risk estimates (odds ratios) for intestinal metaplasia when comparing Helicobacter pylori infected patients belonging to groups B to D with those belonging to group A
| H pylori infecting strain | Odds ratio | 95% confidence interval |
| Group B | 23.3 | 4.6 to 117.9 |
| Group C | 12.0 | 2.3 to 63.7 |
| Group D | 11.8 | 1.7 to 83.2 |
Patients were subdivided on the basis of H pylori infecting groups A to D. Group A: cagA−, s2, m2, babA2−; group B: cagA+, s1, m1, babA2+; group C: cagA+, s1, m2, babA2+; group D: cagA+, s1, m2, babA2−.
DISCUSSION
Using the method described by Gerhard et al,17 babA2 was positive in 24% of the H pylori strains, which is slightly lower than that reported in the literature.17,20,22,23,35 This might relate to the fact that the reverse primer recognises a target DNA sequence that lies in a highly variable babA2 region (nucleotides 612 to 1046).21 Therefore, to reduce false negative results caused by target sequence variability, we adopted another protocol for babA2 amplification: the same forward primer paired with a reverse one targeting a sequence flanking the 5′ end of the variable region (BABA2R607). The sequence of the amplicons corresponded exactly to babA2, thus indicating that our protocol was specific for the babA2 gene. Our method, without reducing specificity, increased the sensitivity for babA2 from 24% to 36%.
We found significant associations between babA2 and the other H pylori virulence genes. These findings are in agreement with those reported by other authors, as are the significant associations between cagA and the s1 and m1 alleles of vacA and oipA “on”.7,8,12,17,20,22,23,27–29,35,36 Interestingly, H pylori cagA negative strains were almost all babA2 negative, oipA “off”, and had the s2 and m2 vacA alleles, suggesting that the absence of the pathogenicity island correlates with the absence of a series of other virulence genes located outside the PAI. It is possible that there is a shared selective pressure for H pylori strains possessing different virulence genes and PAI genes. In line with this hypothesis, cagA positive strains were carriers of the other virulence genes studied, although in different percentages: almost all had the s1 allele of vacA and were oipA “on”, but only 61.5% and 52.8% had the m1 allele of vacA or the babA2 gene. Negative babA2 or positive m2 vacA findings correlated with an endoscopic diagnosis of antral gastritis (82.7% and 76.9% of cases, respectively). In agreement with other authors,11,16,27–30,35,37,38 cagA, oipA “on”, and s1 vacA were found more frequently in patients with peptic ulcer or duodenitis than in patients with antral gastritis only, confirming that these virulence genes play a role in modifying gastroduodenal physiology, leading to mucosal erosions and ulcer.
cagA and s1 vacA played a leading role in determining antral inflammation because higher grades of inflammation and activity were associated with the above virulence genes, but not with m1 vacA or babA2. Unlike inflammation, intestinal metaplasia was significantly associated with all the H pylori virulence genes studied, suggesting that each had a potential pathogenic role in the onset of this precancerous lesion.
“Infection with a more virulent H pylori strain was associated with a higher degree of antral and body colonisation grade, inflammation, and activity”
We divided H pylori into four groups on the basis of positive or negative findings for the virulence genes babA2, cagA, and vacA. Most patients (80%) with antral gastritis only were infected with group A (cagA−, s2m2 vacA, babA2−) H pylori strains. The spread of gastritis to the gastric body was associated with a reduced frequency of group A infecting strains (48.4%) and an increased proportion of H pylori expressing two or more virulence genes. However, we found no specific virulence gene pattern associated with the diagnosis of diseases. With respect to the histology results, gastric mucosal damage was more severe in group B (cagA+, s1m1 vacA, babA2+) than in group A H pylori infected subjects. This means that infection with a more virulent H pylori strain was associated with a higher degree of antral and body colonisation grade, inflammation, and activity. The presence of the two virulence genes, cagA and the s1 allele of vacA, was associated with a higher grade of inflammation, but not with H pylori colonisation grade or activity. babA2 associated with cagA and s1 vacA was related to a higher degree of activity, both in the antrum and the body. We suggest that the H pylori induced gastric inflammatory reaction does not depend on a single factor, but probably results from the synergistic effect of multiple virulence factors, which work together in a complex way, causing damage to the host. This was more clearly demonstrated when intestinal metaplasia was analysed. This precancerous lesion was almost completely absent (96.5%) in the H pylori infected group A patients, whereas it was often seen (36.8%) in patients infected with strains possessing at least one of the virulence genes studied. The risk of developing intestinal metaplasia was progressively higher as the number of virulence genes possessed by the infecting strain increased, suggesting separate roles for each of them in causing this histological abnormality. In conclusion, the worst degrees of inflammation and intestinal metaplasia were seen in those patients infected by H pylori strains coexpressing cagA, s1 vacA, babA2, and m1 vacA.
Take home messages .
The Helicobacter pylori genes cagA, oipA “on”, s1 and m1 vacA, and babA2 are associated with each other, possibly as a result of shared selective pressure
When coexpressed by the same H pylori strain, cagA, s1 and m1 vacA, and babA2 work synergistically in worsening inflammation
The worst degrees of inflammation and intestinal metaplasia were seen in those patients infected by H pylori strains coexpressing cagA, s1 vacA, babA2, and m1 vacA
Abbreviations
IL, interleukin
NS, not significant
PAI, pathogenicity island
PCR, polymerase chain reaction
REFERENCES
- 1.Salama N, Guillemin K, McDaniel TK, et al. A whole-genome microarray reveals genetic diversity among Helicobacter pylori strains. Proc Natl Acad Sci U S A 2000;97:14668–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Blaser MJ, Kirschner D. Dynamics of Helicobacter pylori colonization in relation to the host response. Proc Natl Acad Sci U S A 1999;96:8359–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Falush D, Kraft C, Taylor NS, et al. Recombination and mutation during long-term gastric colonization by Helicobacter pylori: estimates of clock rates, recombination size, and minimal age. Proc Natl Acad Sci U S A 2001;98:15056–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Israel DA, Salama N, Krishna U, et al. Helicobacter pylori genetic diversity within the gastric niche of a single human host. Proc Natl Acad Sci U S A 2001;98:14625–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kuipers EJ, Israel DA, Kusters JG, et al. Quasispecies development of Helicobacter pylori observed in paired isolates obtained years apart from the same host. J Infect Dis 2000;181:273–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Webb GF, Blaser MJ. Dynamics of bacterial phenotype selection in a colonized host. Proc Natl Acad Sci U S A 2002;99:3135–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Basso D, Navaglia F, Brigato L, et al. Analysis of Helicobacter pylori vacA and cagA genotypes and serum antibody profile in benign and malignant gastroduodenal diseases. Gut 1998;43:182–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Basso D, Navaglia F, Brigato L, et al. Helicobacter pylori non-cytotoxic genotype enhances mucosal gastrin and mast cell tryptase. J Clin Pathol 1999;52:210–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bayerdörffer E, Neubauer A, Rudolph B, et al. Regression of primary gastric lymphoma of mucosa-associated lymphoid tissue type after cure of Helicobacter pylori infection. Lancet 1995;345:1591–4. [DOI] [PubMed] [Google Scholar]
- 10.Blaser MJ. Linking Helicobacter pylori to gastric cancer. Nat Med 2000;6:376–7. [DOI] [PubMed] [Google Scholar]
- 11.Figura N. Helicobacter pylori factors involved in the development of gastroduodenal mucosal damage and ulceration. J Clin Gastroenterol 1997;25:S149–63. [DOI] [PubMed] [Google Scholar]
- 12.Navaglia F, Basso D, Piva MG, et al. Helicobacter pylori cytotoxic genotype is associated with peptic ulcer and influences serology. Am J Gastroenterol 1998;93:227–30. [DOI] [PubMed] [Google Scholar]
- 13.Uemura N, Okamoto S, Yamamoto S, et al. Helicobacter pylori infection and the development of gastric cancer. N Engl J Med 2001;345:784–9. [DOI] [PubMed] [Google Scholar]
- 14.Crabtree JE. Role of cytokines in pathogenesis of Helicobacter pylori-induced mucosal damage. Dig Dis Sci 1998;43:46S–55S. [PubMed] [Google Scholar]
- 15.Evans DJ, Evans DG. Helicobacter pylori adhesins: review and perspectives. Helicobacter 2000;5:183–95. [DOI] [PubMed] [Google Scholar]
- 16.Montecucco C, Rappuoli R. Living dangerously: how Helicobacter pylori survives in the human stomach. Nat Rev Mol Cell Biol 2001;2:457–66. [DOI] [PubMed] [Google Scholar]
- 17.Gerhard M, Lehn N, Neumayer N, et al. Clinical relevance of the Helicobacter pylori gene for blood-group antigen-binding adhesin. Proc Natl Acad Sci U S A 1999;96:12778–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Guruge JL, Falk PG, Lorenz RG, et al. Epithelial attachment alters the outcome of Helicobacter pylori infection. Proc Natl Acad Sci U S A 1998;95:3925–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ilver D, Arnqvist A, Ögren J, et al. Helicobacter pylori adhesin binding fucosylated histo-blood group antigens revealed by retagging. Science 1998;279:373–7. [DOI] [PubMed] [Google Scholar]
- 20.Mizushima T, Sugiyama T, Komatsu Y, et al. Clinical relevance of babA2 genotype of Helicobacter pylori in Japanese clinical isolates. J Clin Microbiol 2001;39:2463–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pride TD, Meinersmann RJ, Blaser MJ. Allelic variation within Helicobacter pylori babA and babB. Infect Immun 2001;69:1160–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Prinz C, Schoniger M, Rad R, et al. Key importance of the Helicobacter pylori adherence factor blood group antigen binding adhesin during chronic gastric inflammation. Cancer Res 2001;61:1903–9. [PubMed] [Google Scholar]
- 23.Rad R, Gerhard M, Lang R, et al. The Helicobacter pylori blood group antigen-binding adhesin facilitates bacterial colonization and augments a nonspecific immune response. J Immunol 2002;168:3033–41. [DOI] [PubMed] [Google Scholar]
- 24.Crabtree JE, Farmery SM, Lindley IJD, et al. CagA/ cytotoxic strains of Helicobacter pylori and interleukin-8 in gastric epithelial cell lines. J Clin Pathol 1994;47:945–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Israel DA, Salama N, Arnold CN, et al. Helicobacter pylori strain-specific differences in genetic content, identified by microarray, influence host inflammatory responses. J Clin Invest 2001;107:611–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Yamaoka Y, Kita M, Kodama T, et al. Helicobacter pylori cagA gene and expression of cytokine messenger RNA in gastric mucosa. Gastroenterology 1996;110:1744–52. [DOI] [PubMed] [Google Scholar]
- 27.Yamaoka Y, Kwon DH, Graham DY. A Mr 34,000 proinflammatory outer membrane protein (oipA) of Helicobacter pylori. Proc Natl Acad Sci U S A 2000;97:7533–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Atherton JC, Cao P, Peek RM, et al. Mosaicism in vacuolating cytotoxin alleles of Helicobacter pylori. Association of specific vacA types with cytotoxin production and peptic ulceration. J Biol Chem 1995;270:17771–7. [DOI] [PubMed] [Google Scholar]
- 29.Atherton JC, Peek RM, Tham KT, et al. Clinical and pathological importance of heterogeneity in vacA, the vacuolating cytotoxin gene of Helicobacter pylori. Gastroenterology 1997;112:92–9. [DOI] [PubMed] [Google Scholar]
- 30.Cover T. The vacuolating cytotoxin of Helicobacter pylori. Mol Microbiol 1996;20:241–6. [DOI] [PubMed] [Google Scholar]
- 31.Reyrat JM, Pelicic V, Papini E, et al. Towards deciphering the Helicobacter pylori cytotoxin. Mol Microbiol 1999;34:197–204. [DOI] [PubMed] [Google Scholar]
- 32.Dixon MF, Genta RM, Yardley JH, et al. Classification and grading of gastritis. The updated Sydney system. Am J Surg Pathol 1996;20:1161–81. [DOI] [PubMed] [Google Scholar]
- 33.Zambon CF, Basso D, Navaglia F, et al. H. pylori virulence genes and host IL1-RN and IL-1β genes interplay in favoring the development of peptic ulcer and intestinal metaplasia. Cytokine 2002;18:242–51. [DOI] [PubMed] [Google Scholar]
- 34.Navaglia F, Basso D, Plebani M. Touchdown PCR: a rapid method to genotype Helicobacter pylori infection. Clin Chim Acta 1997;262:157–60. [DOI] [PubMed] [Google Scholar]
- 35.Kim SY, Woo CW, Lee YM, et al. Genotyping CagA, VacA subtype, IceA1, and BabA of Helicobacter pylori isolates from Korean patients, and their association with gastroduodenal diseases. J Korean Med Sci 2001;16:579–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ando T, Peek RM, Pride D, et al. Polymorphism of Helicobacter pylori HP0938 reflect geographic origin and correlate with cagA status. J Clin Microbiol 2002;40:239–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Cesini S, Lange C, Xiang Z, et al. cag, a pathogenicity island of Helicobacter pylori, encodes type I-specific and disease-associated virulence factors. Proc Natl Acad Sci U S A 1996;93:14648–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Graham DY, Yamaoka Y. Disease-specific Helicobacter pylori virulence factors: unfulfilled promise. Helicobacter 2000;5:S3–9. [DOI] [PubMed] [Google Scholar]