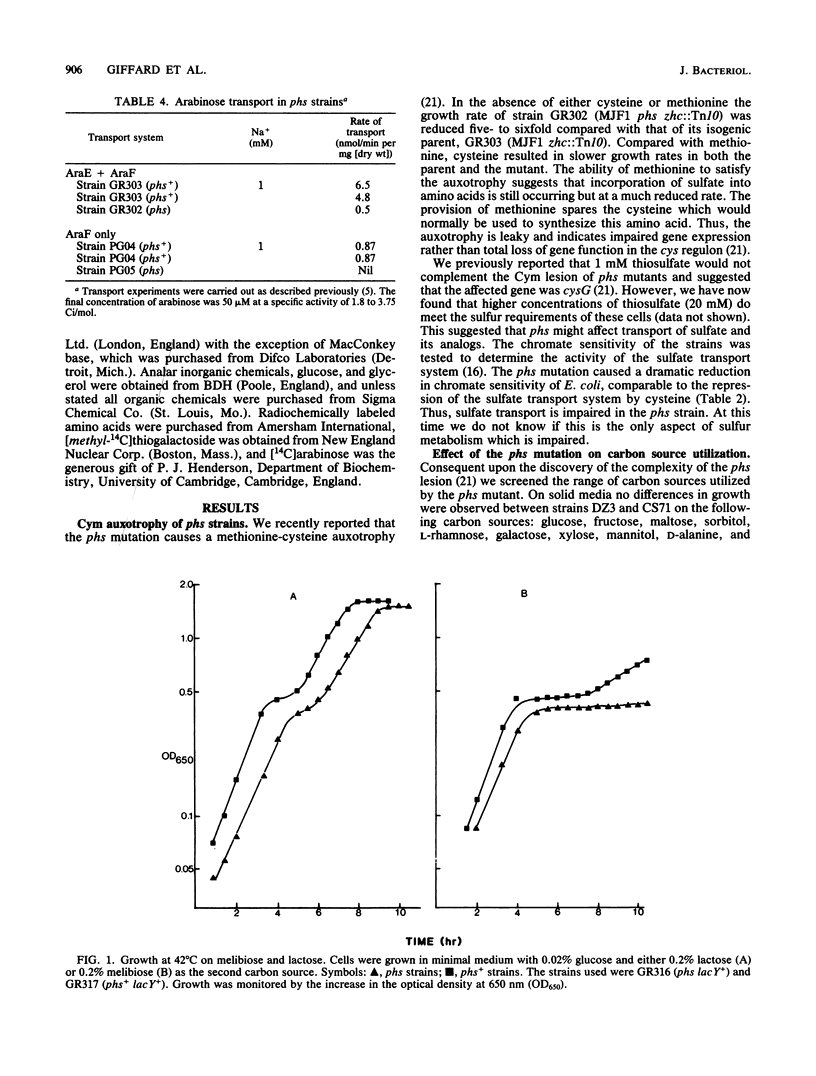
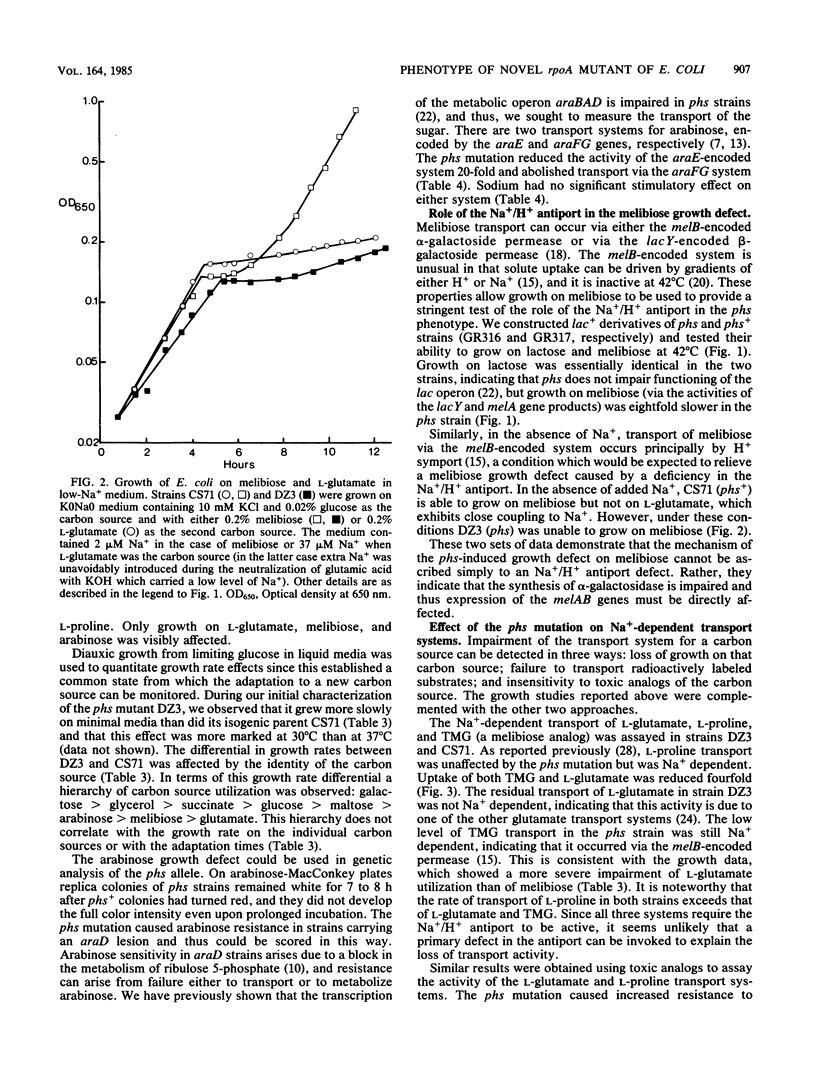
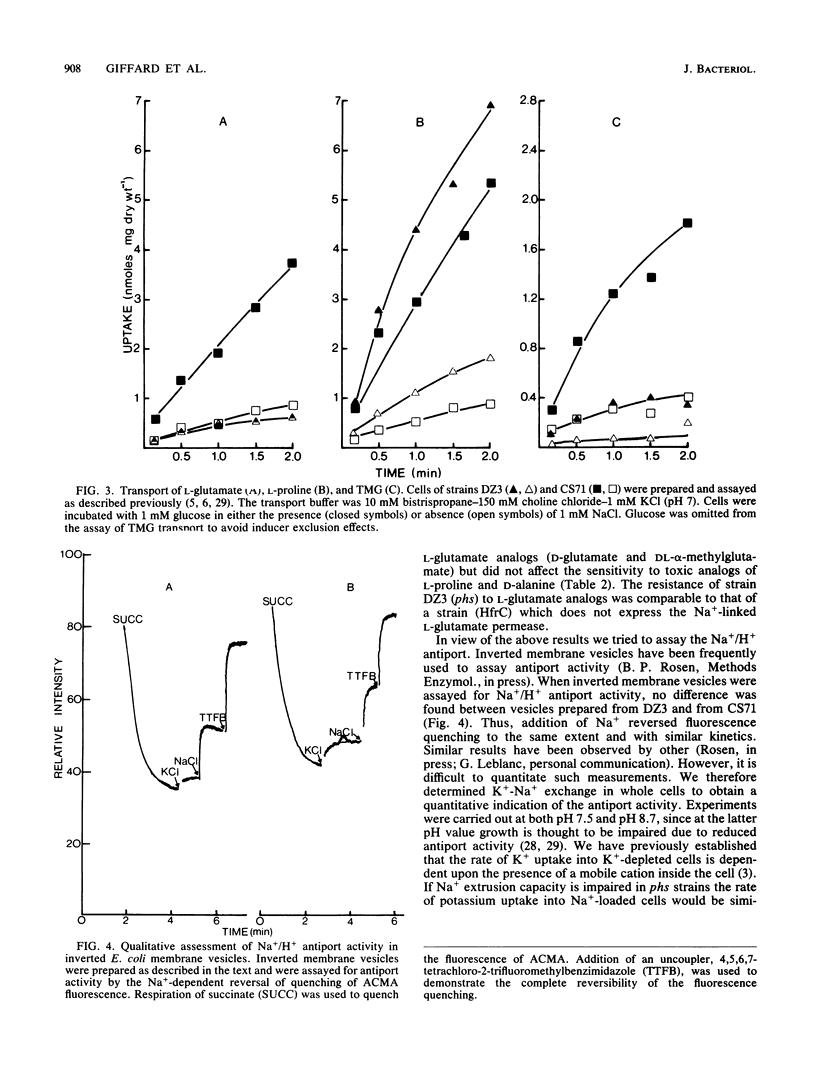
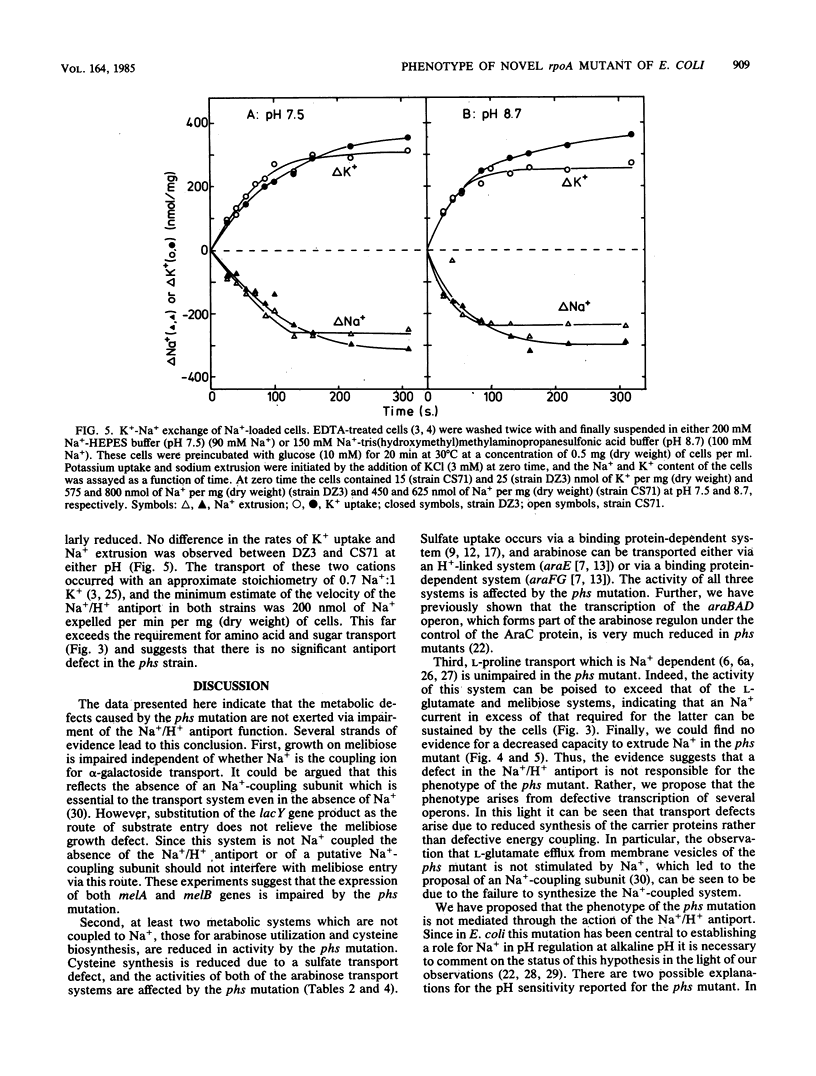
Abstract
The phs mutation of Escherichia coli has been suggested to affect the Na+/H+ antiport (D. Zilberstein, E. Padan, and S. Schuldiner, FEBS Lett. 168:327-330, 1980). We have recently shown that the mutation affects the rpoA gene and thus affects transcription. The extent of the pleiotropy of the phs mutation was investigated. In addition to the previously reported growth defect on L-glutamate and melibiose, the mutation also affects at least two other metabolic systems. The transport and metabolism of arabinose is impaired and the transport of sulfate is reduced. The extent to which the effects of the phs mutation on metabolism are due to a defect in the Na+/H+ antiport was investigated, and no causal role for this transport system in the metabolic defects was found.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alper M. D., Ames B. N. Transport of antibiotics and metabolite analogs by systems under cyclic AMP control: positive selection of Salmonella typhimurium cya and crp mutants. J Bacteriol. 1978 Jan;133(1):149–157. doi: 10.1128/jb.133.1.149-157.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bachmann B. J. Linkage map of Escherichia coli K-12, edition 7. Microbiol Rev. 1983 Jun;47(2):180–230. doi: 10.1128/mr.47.2.180-230.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bakker E. P., Mangerich W. E. Interconversion of components of the bacterial proton motive force by electrogenic potassium transport. J Bacteriol. 1981 Sep;147(3):820–826. doi: 10.1128/jb.147.3.820-826.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Booth I. R., Hamilton W. A. Quantitative analysis of proton-linked transport system. beta-Galactoside exit in Escherichia coli. Biochem J. 1980 May 15;188(2):467–473. doi: 10.1042/bj1880467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cairney J., Higgins C. F., Booth I. R. Proline uptake through the major transport system of Salmonella typhimurium is coupled to sodium ions. J Bacteriol. 1984 Oct;160(1):22–27. doi: 10.1128/jb.160.1.22-27.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen C. C., Tsuchiya T., Yamane Y., Wood J. M., Wilson T. H. Na+ (Li+)-proline cotransport in Escherichia coli. J Membr Biol. 1985;84(2):157–164. doi: 10.1007/BF01872213. [DOI] [PubMed] [Google Scholar]
- DREYFUSS J. CHARACTERIZATION OF A SULFATE- AND THIOSULFATE-TRANSPORTING SYSTEM IN SALMONELLA TYPHIMURIUM. J Biol Chem. 1964 Jul;239:2292–2297. [PubMed] [Google Scholar]
- Daruwalla K. R., Paxton A. T., Henderson P. J. Energization of the transport systems for arabinose and comparison with galactose transport in Escherichia coli. Biochem J. 1981 Dec 15;200(3):611–627. doi: 10.1042/bj2000611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ENGLESBERG E., ANDERSON R. L., WEINBERG R., LEE N., HOFFEE P., HUTTENHAUER G., BOYER H. L-Arabinose-sensitive, L-ribulose 5-phosphate 4-epimerase-deficient mutants of Escherichia coli. J Bacteriol. 1962 Jul;84:137–146. doi: 10.1128/jb.84.1.137-146.1962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kacser H., Burns J. A. The molecular basis of dominance. Genetics. 1981 Mar-Apr;97(3-4):639–666. doi: 10.1093/genetics/97.3-4.639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karbonowska H., Wiater A., Hulanicka D. Sulphate permease of Escherichia coli K12. Acta Biochim Pol. 1977;24(4):329–334. [PubMed] [Google Scholar]
- Kolodrubetz D., Schleif R. L-arabinose transport systems in Escherichia coli K-12. J Bacteriol. 1981 Nov;148(2):472–479. doi: 10.1128/jb.148.2.472-479.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lopilato J., Tsuchiya T., Wilson T. H. Role of Na+ and Li+ in thiomethylgalactoside transport by the melibiose transport system of Escherichia coli. J Bacteriol. 1978 Apr;134(1):147–156. doi: 10.1128/jb.134.1.147-156.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mizobuchi K, Demerec M, Gillespie D H. Cysteine Mutants of Salmonella Typhimurium. Genetics. 1962 Nov;47(11):1617–1627. doi: 10.1093/genetics/47.11.1617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ota N., Galsworthy P. R., Pardee A. B. Genetics of sulfate transport by Salmonella typhimurium. J Bacteriol. 1971 Mar;105(3):1053–1062. doi: 10.1128/jb.105.3.1053-1062.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- PRESTIDGE L. S., PARDEE A. B. A SECOND PERMEASE FOR METHYL-THIO-BETA-D-GALACTOSIDE IN ESCHERICHIA COLI. Biochim Biophys Acta. 1965 May 4;100:591–593. doi: 10.1016/0304-4165(65)90029-2. [DOI] [PubMed] [Google Scholar]
- Qureshi M. A., Smith D. A., Kingsman A. J. Mutants of Salmonella typhimurium responding to cysteine or methionine: their nature and possible role in the regulation of cysteine biosynthesis. J Gen Microbiol. 1975 Aug;89(2):353–370. doi: 10.1099/00221287-89-2-353. [DOI] [PubMed] [Google Scholar]
- Rotman B., Ganesan A. K., Guzman R. Transport systems for galactose and galactosides in Escherichia coli. II. Substrate and inducer specificities. J Mol Biol. 1968 Sep 14;36(2):247–260. doi: 10.1016/0022-2836(68)90379-3. [DOI] [PubMed] [Google Scholar]
- Rowland G. C., Giffard P. M., Booth I. R. Genetic studies of the phs locus of Escherichia coli, a mutation causing pleiotropic lesions in metabolism and pH homeostasis. FEBS Lett. 1984 Aug 6;173(2):295–300. doi: 10.1016/0014-5793(84)80794-2. [DOI] [PubMed] [Google Scholar]
- Rowland G. C., Giffard P. M., Booth I. R. phs Locus of Escherichia coli, a mutation causing pleiotropic lesions in metabolism, is an rpoA allele. J Bacteriol. 1985 Nov;164(2):972–975. doi: 10.1128/jb.164.2.972-975.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schairer H. U., Friedl P., Schmid B. I., Vogel G. The use of several energy-coupling reactions in characterizing mutants of Escherichia coli K12 defective in oxidative phosphorylation. Eur J Biochem. 1976 Jul 1;66(2):257–268. doi: 10.1111/j.1432-1033.1976.tb10515.x. [DOI] [PubMed] [Google Scholar]
- Schellenberg G. D., Furlong C. E. Resolution of the multiplicity of the glutamate and aspartate transport systems of Escherichia coli. J Biol Chem. 1977 Dec 25;252(24):9055–9064. [PubMed] [Google Scholar]
- Tsuchiya T., Yamane Y., Shiota S., Kawasaki T. Cotransport of proline and Li+ in Escherichia coli. FEBS Lett. 1984 Mar 26;168(2):327–330. doi: 10.1016/0014-5793(84)80272-0. [DOI] [PubMed] [Google Scholar]
- Zilberstein D., Agmon V., Schuldiner S., Padan E. Escherichia coli intracellular pH, membrane potential, and cell growth. J Bacteriol. 1984 Apr;158(1):246–252. doi: 10.1128/jb.158.1.246-252.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zilberstein D., Agmon V., Schuldiner S., Padan E. The sodium/proton antiporter is part of the pH homeostasis mechanism in Escherichia coli. J Biol Chem. 1982 Apr 10;257(7):3687–3691. [PubMed] [Google Scholar]
- Zilberstein D., Ophir I. J., Padan E., Schuldiner S. Na+ gradient-coupled porters of EScherichia coli share a common subunit. J Biol Chem. 1982 Apr 10;257(7):3692–3696. [PubMed] [Google Scholar]


