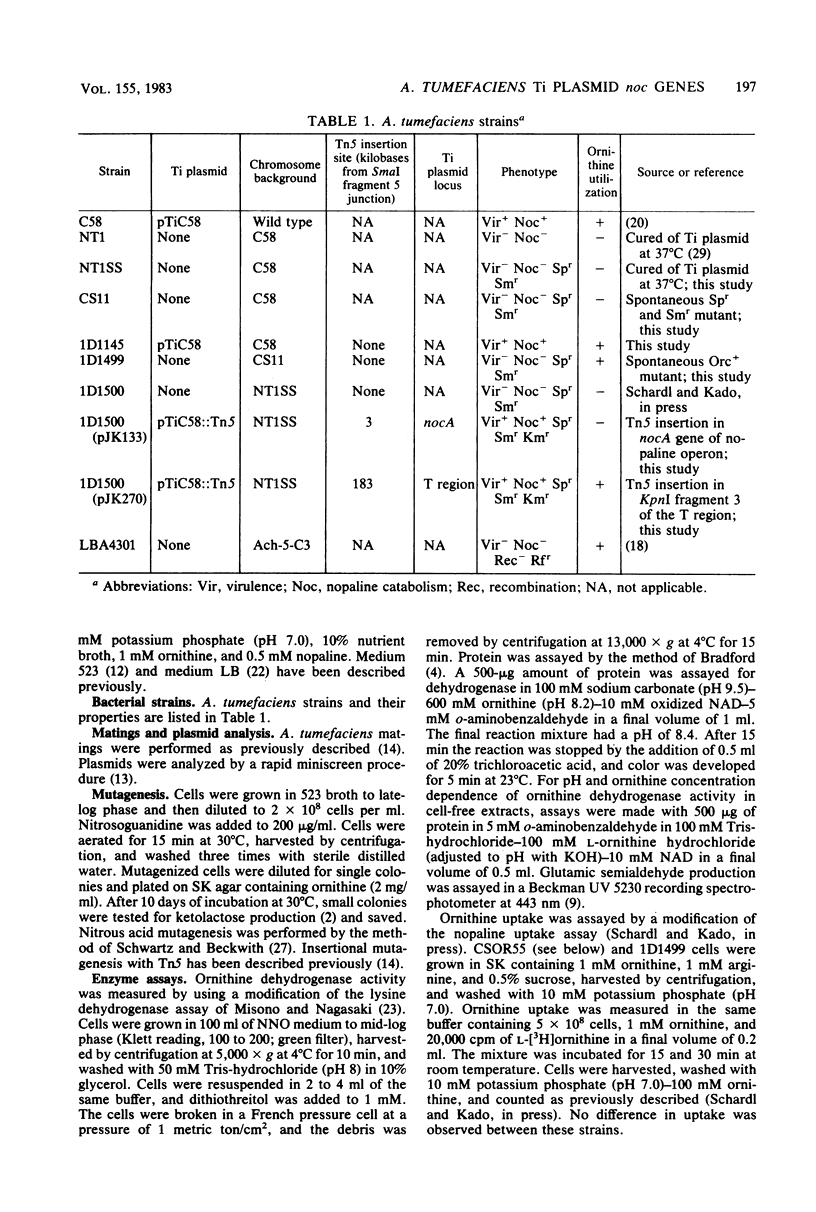
Abstract
The pTiC58 plasmid noc genes of Agrobacterium tumefaciens C58 code for nopaline oxidase (nocC), nopaline permease (nocP), the inducible periplasmic protein n1 (nocB), and a function(s) required for ornithine catabolism (nocA). In addition, strains C58 and Ach-5 of A. tumefaciens have chromosomal ornithine catabolism genes. The chromosomal orc gene codes for ornithine dehydrogenase. Strain C58 is normally orc, but orc+ mutants can be selected. We have characterized both chromosomal orc and pTiC58 nocA plasmid genes. Complementation of most chromosomal orc mutants by pTiC58 restored growth on both nopaline and L-ornithine but did not restore ornithine dehydrogenase activity. We conclude that ornithine is an intermediate of nopaline degradation and that the Ti plasmid and chromosome both code for ornithine-degradative enzymes. A model for nopaline catabolism is presented.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Applebaum D., Sabo D. L., Fischer E. H., Morris D. R. Biodegradative ornithine decarboxylase of Escherichia coli. Purification, properties, and pyridoxal 5'-phosphate binding site. Biochemistry. 1975 Aug 12;14(16):3675–3681. doi: 10.1021/bi00687a025. [DOI] [PubMed] [Google Scholar]
- Bomhoff G., Klapwijk P. M., Kester H. C., Schilperoort R. A., Hernalsteens J. P., Schell J. Octopine and nopaline synthesis and breakdown genetically controlled by a plasmid of Agrobacterium tumefaciens. Mol Gen Genet. 1976 May 7;145(2):177–181. doi: 10.1007/BF00269591. [DOI] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1006/abio.1976.9999. [DOI] [PubMed] [Google Scholar]
- Ellis J. G., Kerr A., Tempé J., Petit A. Arginine catabolism: a new function of both octopine and nopaline Ti-plasmids of Agrobacterium. Mol Gen Genet. 1979 Jun 20;173(3):263–269. doi: 10.1007/BF00268636. [DOI] [PubMed] [Google Scholar]
- Guirard B. M., Snell E. E. Purification and properties of ornithine decarboxylase from Lactobacillus sp. 30a. J Biol Chem. 1980 Jun 25;255(12):5960–5964. [PubMed] [Google Scholar]
- Holsters M., Silva B., Van Vliet F., Genetello C., De Block M., Dhaese P., Depicker A., Inzé D., Engler G., Villarroel R. The functional organization of the nopaline A. tumefaciens plasmid pTiC58. Plasmid. 1980 Mar;3(2):212–230. doi: 10.1016/0147-619x(80)90110-9. [DOI] [PubMed] [Google Scholar]
- Jensen R. E., Zdybak W. T., Yasuda K., Chilton W. S. A useful synthesis of nopaline, a crown gall tumor metabolite. Biochem Biophys Res Commun. 1977 Apr 25;75(4):1066–1070. doi: 10.1016/0006-291x(77)91490-5. [DOI] [PubMed] [Google Scholar]
- Jubier M. -F. Degradation of lysopine by an inducible membrane-bound oxidase in Agrobacterium tumefaciens. FEBS Lett. 1972 Dec 1;28(2):129–132. doi: 10.1016/0014-5793(72)80693-8. [DOI] [PubMed] [Google Scholar]
- Kado C. I., Liu S. T. Rapid procedure for detection and isolation of large and small plasmids. J Bacteriol. 1981 Mar;145(3):1365–1373. doi: 10.1128/jb.145.3.1365-1373.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kao J. C., Perry K. L., Kado C. I. Indoleacetic acid complementation and its relation to host range specifying genes on the Ti plasmid of Agrobacterium tumefaciens. Mol Gen Genet. 1982;188(3):425–432. doi: 10.1007/BF00330044. [DOI] [PubMed] [Google Scholar]
- Klapwijk P. M., Scheulderman T., Schilperoort R. A. Coordinated regulation of octopine degradation and conjugative transfer of Ti plasmids in Agrobacterium tumefaciens: evidence for a common regulatory gene and separate operons. J Bacteriol. 1978 Nov;136(2):775–785. doi: 10.1128/jb.136.2.775-785.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klapwijk P. M., Schilperoort R. A. Negative control of octopine degradation and transfer genes of octopine Ti plasmids in Agrobacterium tumefaciens. J Bacteriol. 1979 Aug;139(2):424–431. doi: 10.1128/jb.139.2.424-431.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klapwijk P. M., van Beelen P., Schilperoort R. A. Isolation of a recombination deficient Agrobacterium tumefaciens mutant. Mol Gen Genet. 1979 Jun 7;173(2):171–175. doi: 10.1007/BF00330307. [DOI] [PubMed] [Google Scholar]
- Lemmers M., De Beuckeleer M., Holsters M., Zambryski P., Depicker A., Hernalsteens J. P., Van Montagu M., Schell J. Internal organization, boundaries and integration of Ti-plasmid DNA in nopaline grown gall tumours. J Mol Biol. 1980 Dec 15;144(3):353–376. doi: 10.1016/0022-2836(80)90095-9. [DOI] [PubMed] [Google Scholar]
- Lin B. C., Kado C. I. Studies on Agrobacterium tumefaciens. VIII. Avirulence induced by temperature and ethidium bromide. Can J Microbiol. 1977 Nov;23(11):1554–1561. doi: 10.1139/m77-229. [DOI] [PubMed] [Google Scholar]
- Liu S. T., Perry K. L., Schardl C. L., Kado C. I. Agrobacterium Ti plasmid indoleacetic acid gene is required for crown gall oncogenesis. Proc Natl Acad Sci U S A. 1982 May;79(9):2812–2816. doi: 10.1073/pnas.79.9.2812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Misono H., Nagasaki S. Occurrence of L-lysine epsilon-dehydrogenase in Agrobacterium tumefaciens. J Bacteriol. 1982 Apr;150(1):398–401. doi: 10.1128/jb.150.1.398-401.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montoya A. L., Chilton M. D., Gordon M. P., Sciaky D., Nester E. W. Octopine and nopaline metabolism in Agrobacterium tumefaciens and crown gall tumor cells: role of plasmid genes. J Bacteriol. 1977 Jan;129(1):101–107. doi: 10.1128/jb.129.1.101-107.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwartz D. O., Beckwith J. R. Mutagens which cause deletions in Escherichia coli. Genetics. 1969 Feb;61(2):371–376. doi: 10.1093/genetics/61.2.371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Larebeke N., Engler G., Holsters M., Van den Elsacker S., Zaenen I., Schilperoort R. A., Schell J. Large plasmid in Agrobacterium tumefaciens essential for crown gall-inducing ability. Nature. 1974 Nov 8;252(5479):169–170. doi: 10.1038/252169a0. [DOI] [PubMed] [Google Scholar]
- Watson B., Currier T. C., Gordon M. P., Chilton M. D., Nester E. W. Plasmid required for virulence of Agrobacterium tumefaciens. J Bacteriol. 1975 Jul;123(1):255–264. doi: 10.1128/jb.123.1.255-264.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiss E., Mamay H. K., Dasch G. A. Ornithine metabolism in the genus Rochalimaea. J Bacteriol. 1982 Apr;150(1):245–250. doi: 10.1128/jb.150.1.245-250.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zaenen I., Van Larebeke N., Van Montagu M., Schell J. Supercoiled circular DNA in crown-gall inducing Agrobacterium strains. J Mol Biol. 1974 Jun 15;86(1):109–127. doi: 10.1016/s0022-2836(74)80011-2. [DOI] [PubMed] [Google Scholar]


