Abstract
The inhibition of apoptosis by Toxoplasma gondii is governed by its modulation of several signaling cascades including the NFκappaB and JNK pathways. This is evident in the dysregulation of JNK activation following treatment with UV and TNFα, both apoptogenic stimuli. Infection-mediated interference with the JNK cascade was found to be highly reproducible in HeLa cells. In light of emerging evidence regarding cross talk between the JNK and NFκB cascades, we examined the impact of infection in wild type and RelA/p65−/− mouse embryonic fibroblasts (MEF). Remarkably, parasite infection failed to significantly impact both UV and TNFα-mediated JNK phosphorylation in both cell lines suggesting a cell type specific effect. Furthermore siRNA-mediated knockdown of RelA/p65 failed to impact the parasite mediated effects on stimulus dependent activation of JNK in HeLa cells. Finally, the infection mediated suppression of JNK phosphorylation in HeLa cells did not result in decreased JNK kinase activity. Rather, the reduced levels of phospho-JNK in infected cells correlated with increased phosphatase activity noted by the partial rescue of the phenotype following treatment with okadaic acid. Taken together the results indicate that manipulation of the JNK-pathway does not involve NFκB and is furthermore not a central component of the parasite enforced block of apoptosis. It further highlights the complexity of these systems and the danger of extrapolating results both within and across pathogen-host cell systems based on limited studies.
Keywords: Toxoplasma gondii, apoptosis, JNK, NFκB
Introduction
Toxoplasma gondii is an obligate intracellular, protozoan parasite capable of infecting all warm blooded animals [1]. Acute toxoplasmosis is generally asymptomatic but is life threatening in immunocompromised individuals and can cause severe neurological sequelae in children if infection occurs during pregnancy [1]. Upon infection, T. gondii induces the host cell to become refractory to all apoptotic stimuli investigated to date [2–7]. While several infection-dependent changes, such as the activation of host nuclear factor kappa B (NFκB) [3] and phosphoinositide 3-kinase [8], are known to play important roles, the contributions of the MAP-Kinase family are poorly understood (reviewed in [9]). The stress/mitogen activated protein kinase (MAPK) JNK is involved in multiple cellular processes including the regulation of apoptosis, cellular proliferation, and the inflammatory response [10].
In an earlier study we found that exposure of infected HeLa cells to ultraviolet light (UV) resulted in a marked inhibition of JNK activation as determined by levels of phospho-JNK [2]. The observations by Tournier et al. that JNK activity is required for the release of mitochondrial cytochrome c and the subsequent execution of UV induced apoptosis [11] suggested the apparent inhibition in parasite infected HeLa cells [2] was linked to this step.
Due to its role in multiple homeostatic processes, the activation of JNK remains under tight control. This regulation occurs both upstream of JNK activation (the prevention of JNK phosphorylation) [12, 13] and downstream of JNK activation (the dephosphorylation of active JNK) [14, 15]. One of the most important pro-survival, anti-apoptotic pathways regulating JNK is the NFκB pathway [12, 13]. While its pro-survival, anti-apoptotic activity can be attributed to the transcription of multiple genes [16], a subset of these have been found to specifically prevent apoptosis by inhibiting the activation of JNK [12, 13, 17–22]. The dephosphorylation of JNK ensures that it remains inactive in the absence of the appropriate signals and that the duration of its activation is controlled in a manner consistent with the stimulus [15].
The profound inhibition of JNK activation in HeLa cells [2] led us to address its molecular basis. In the course of these studies, we found that the parasite-directed effect in HeLa cells is context specific as it is not observed mouse embryonic fibroblasts (MEF). We further find that T. gondii-directed interference with JNK signaling is not directly dependent on NFκB, a transcriptional pathway central to the parasite-directed inhibition of apoptosis [3]. Our data indicate that while the disruption of JNK signaling may be a feature of Toxoplasma infection it does not play a generalized role in the inhibition of apoptosis. These results illustrate the complexity of signaling pathways and the need to examine their manipulation by pathogens in a context-specific manner while avoiding overreaching extrapolations that attempt to broadly oversimplify these complex events.
Materials and Methods
Cell and parasite maintenance
HeLa (ATCC CCL-2) were maintained in Dulbecco’s Modified Eagle Medium (Gibco-Invitrogen, Carlsbad, CA) supplemented with 7% heat-inactivated fetal bovine serum (Gemini Bio-Products, Woodland CA), 100 units ml−1 penicillin, 100 ug ml−1 streptomycin, and 2 mM L-glutamine (Gibco-Invitrogen) (DMEM). Immortalized wild type MEF and RelA-2 (p65/RelA−/−) knock out cells [23], provided by Dr. Alexander Hoffmann (UCSD), and Vero cells (ATCC CCL-81) were maintained in alpha minimum essential medium (αMEM) (Gibco-Invitrogen) supplemented with 7% heat-inactivated fetal bovine serum (Gemini Bio-Products), 100 U ml−1 penicillin, 100 μg ml−1 streptomycin, and 2 mM L-glutamine (αMEM) (Gibco-Invitrogen). The RH strain of T. gondii strain lacking the HXGPRT gene expression (RHΔHX) [24] (NIH Aids Research and Reference Program) was maintained and passaged in Vero cells as described previously [3].
Induction of JNK phosphorylation
JNK phosphorylation was induced using ultraviolet light (UV, 254 nM) or tumor necrosis factor-alpha (TNFα). UV irradiation was delivered as described previously [2]. To investigate the ability of T. gondii to inhibit TNFα-dependent JNK phosphorylation, HeLa cells were exposed to 10 ng ml−1 recombinant human TNFα (R&D Systems, Minneapolis, MN) and mouse embryonic fibroblasts (both wild type and p65−/−) were exposed to 10 ng ml−1 recombinant murine TNFα (R&D Systems, Minneapolis, MN). TNFα was reconstituted as recommended by the manufacturer and diluted to the appropriate concentration in the growth medium. Stimulation was performed by the addition of 2 ml TNFα containing medium to cell monolayers plated in 6-well Falcon tissue culture plate (BD Biosciences, San Jose, CA). Following UV or TNFα exposure cells were harvested at the time points indicated in the figures and the level of phospho-JNK was analyzed via immunoblotting. The cell samples labeled 0 min were harvested immediately prior to exposure to UV or TNFα.
Immunoblot analysis
Following the induction of JNK phosphorylation, the cells were harvested at the times described in the figures. Cells were processed as previously described [2] for immunoblot analysis with 15 to 25 μg total protein loaded per well. Detection of phospho-JNK was achieved using a rabbit polyclonal antibody (1:1000, cat#9251, Cell Signaling, Danvers, MA), while total JNK was detected using another rabbit polyclonal antibody (1:1000, sc-571, Santa Cruz Biotechnology, Santa Cruz, CA). Anti-calnexin (1:3000) [25] was used as a loading control. The filters were probed sequentially following stripping of prior bound antibody.
Densitometric analysis
Densitometric analysis of the immunoblots was performed using ImageJ software (National Institutes of Health, USA) to calculate the integrated density of the bands in question. Multiple exposures were obtained for each immunoblot permitting the selection of the exposure condition that provided the best linearity for the calnexin loading control and the total JNK signals. Scanned images of the immunoblot films were opened using ImageJ software. The background was subtracted from the image using the subtract background function and the image was inverted. The membranes had been exposed to film for multiple lengths of time and the exposure judged to be in the linear range was quantified. A box was made around the largest band and this size box was used to measure each band for a given exposure. In addition to measuring the integrated density of the bands representing the proteins of interest, the corresponding calnexin band was measured and used to normalize for gel loading. In the case of phospho-JNK, the integrated density of a phospho-JNK band was normalized by dividing that value with the value representing total JNK levels (i.e. P-JNK/(JNK). Finally, the resulting values for multiple experiments were averaged and subjected to statistical analyses (see below). In the case of undetectable proteins, the ratio was generated using the integrated density of the site in the lane matching the size of the protein. The integrated density in this area was measured using a box the same size as that used to measure the bands in the other lanes.
For the JNK activity assay, the bands representing 32P-cJUN were quantified using the ImageQuant software (Molecular Dynamics, GE Healthcare, Piscataway, NJ), designed for the analysis of gels scanned with the Molecular Dynamics phosphorimager described above. A uniform box was used to measure the band intensity. A box was placed in a section of the image outside of the lanes containing protein. The value of the region encompassed by this box was subtracted from the volume values measured for the bands of interest.
Statistical Analyses
Densitometric analyses, while typically consistent within a given immunoblot, exhibited considerable variation from blot to blot. Thus, while comparing multiple experiments for the level of phospho-JNK relative to total JNK signal, significant variation in absolute levels was noted. For this reason, raw data (relative intensity of P-JNK/JNK), in the form of dot plots generated from each replicate, is presented as it accurately reflects the range of results. In addition, for the sake of simplicity we only present densitometric data for the p54 JNK isoform as the phosphorylation patterns for p46 JNK are not different (data not shown).
All statistical analyses were performed using the Graph Pad Prism 4.0 software package. The data sets for the relative intensity of phospho-JNK were analyzed using 2-factor (time and infection) ANOVA analysis. The degree of interaction between these main factors was also determined. In addition a Bonferroni’s post-hoc test was applied in a pairwise manner to identify the points of variance using both time and infection as anchors (data not shown). We determined p-values under 0.01 as highly significant (***), 0.05–0.01 as moderately significant (**). Statistics with p-value scores >0.05 were considered as not significant (ns). In experiments where data is presented as a bar graph the statistic of the standard error of the mean is presented to account for the data distribution. ANOVA results for these data are presented in the figure legends.
siRNA-mediated depletion of p65/Rel
The depletion of p65/RelA in HeLa cells was performed using ON-TARGET plus Smart pool siRNA specific for human relA, prepared as recommended by the manufacturer (gene ID 5970) (003533, Dharmacon, Lafayette, CO). HeLa cells were seeded into 24- or 6-well dishes and incubated overnight. Transfection was performed when the cells reached 70–80% confluency. For immunofluorescence assays, HeLa cells were seeded onto 12 mm glass gelatin coated cover slips (12–545-80, Fisher Scientific,). The siRNA was transfected into the HeLa cells using Lipofectamine™ RNAiMAX reagent (Invitrogen, cat# 13778-075) essentially as recommended by the manufacturer. The siRNA-lipid transfection reagent mixture (800 nM in 150μl Optimem with 2 μl RNAiMAX reagent) was incubated at room temperature for 20 min. A dilution series of lipid-siRNA complexes was added to the cell monolayers as indicated in the figure legend for 7 hours following which the medium was replaced with DMEM and the cells permitted to recover.
The cells were incubated for 24, 48, and/or 72 hr following transfection depending on the experiment, harvested and prepared for immunoblot analysis using anti-p65/RelA rabbit polyclonal antibody (sc-372, Santa Cruz Biotechnology, Santa Cruz, CA). The extent of p65/RelA depletion was also analyzed via immunofluorescence as described below. In experiments designed to determine the role of NFκB signaling in the decreased levels of phospho-JNK observed in infected cells, mock (lipid transfection reagent alone) or siRNA (100 nM) transfected cells were infected with T. gondii (m.o.i= 8) 48 hr after transfection and incubated for approximately 20 hr. After this incubation, cells were exposed to UV, harvested, and the levels of phospho-JNK analyzed as described above.
Immunofluorescence analysis
Immunofluorescence analysis was performed as previously described [3] using an anti-p65/RelA antibody (sc-372, Santa Cruz Biotechnology) and Oregon green 488 conjugated-anti-rabbit secondary antibodies (Molecular Probes/Invitrogen,) in 20% goat serum in PBS. Cells were fixed with 3% paraformaldehyde at 24, 48, and 72 hr post transfection. The nuclei were visualized using Hoechst dye (1:25,000 dilution of 1 mg ml−1 stock, Molecular Probes).
JNK immunoprecipitation and in vitro activity assay
The level of JNK activity in uninfected and infected cells following exposure to UV was determined using JNK immunoprecipitates. HeLa and MEF were seeded into 10-cm dishes and incubated for 24 h at 37°C. Prior to infection, the cells from two plates were trypsinized and counted to determine the total number of parasites needed to infect at a multiplicity of infection of 8. Cells from at least two dishes were harvested for each condition (untreated and UV with or without infection) and centrifuged at 2000 rpm for 10 min (Allegra™ CK centrifuge, Beckman Coulter, Fullerton, CA). The supernatant was removed, and the pellet was resuspended in 1 ml kinase binding buffer (KBB) (20 mM HEPES, 50 mM KCl, 2.5 mM MgCl2, 0.1 mM EDTA, 0.2% Triton X-100, 2mM β-mercaptoethanol, 20 mM β-glycerol-phosphate, and 50 μM sodium orthovanadate) supplemented with 1.0 mg ml−1 pNpp (SIGMA FAST™ p-nitrophenyl phosphate tablets, Sigma, St. Louis, MO) and a protease inhibitor cocktail (1 tablet per 10 ml KBB, cOmplete Mini, EDTA-free, Roche, Indianapolis, IN). This suspension was incubated on ice for 15 min and the insoluble material was removed by a 5 min centrifugation at 13,200 rpm at 4°C. The resulting supernatant was incubated with 20 μl of 10% protein A sepharose beads (GE Healthcare, Little Chalfont, England) in PBS at 4°C for 1 hr with constant end over end agitation to remove any proteins that bound non-specifically to the beads. The suspension was centrifuged at 4,000 rpm for 2 min and the supernatant was removed and incubated with 1.6 μg (6 μl) of anti-JNK antibody (sc-571, Santa Cruz Biotechnology) for 4 hr at 4°C with constant end-over-end mixing. The antibody-antigen complexes were removed from the supernatant by adding 25 ul of 10% protein A sepharose beads followed by an additional incubation period of 1 hr at 4°C with constant end-over-end mixing. The beads were centrifuged and washed four times with KBB. Kinase assays were performed as described by Molestina et al. [26] using GST-cJUN (Santa Cruz Biotechnology, Inc., Santa Cruz, CA, sc-4113) as a substrate. Samples were removed prior to immunoprecipitation for immunoblot analysis to confirm the inhibition of JNK phosphorylation by T. gondii-infection by immunoblot analysis.
Phosphatase inhibition
The effect of phosphatase inhibition on the T. gondii-infection dependent decrease in phospho-JNK observed following UV irradiation was determined using the cell permeable phosphatase inhibitor okadaic acid. HeLa cells were seeded into four 6-well dishes and incubated at 37°C for 24 hr. The HeLa cells were infected with T. gondii RH tachyzoites at an MOI of 6 for 18 hr prior to the introduction of okadaic acid. Okadaic acid (Calbiochem) is a cell permeable compound that inhibits protein phosphatases 1, 2A, and 2B depending on the concentration. Uninfected and infected cells were pre-treated with either 20 nM (data not shown) or 100 nM okadaic acid diluted in DMEM for 2 hr prior to exposure to 100 mJ cm−2 UV as described previously. Following irradiation, media containing okadaic acid was returned to the wells and the cells were harvested at the time intervals indicated.
RESULTS
T. gondii-infected cells exhibit decreased JNK activation following UV
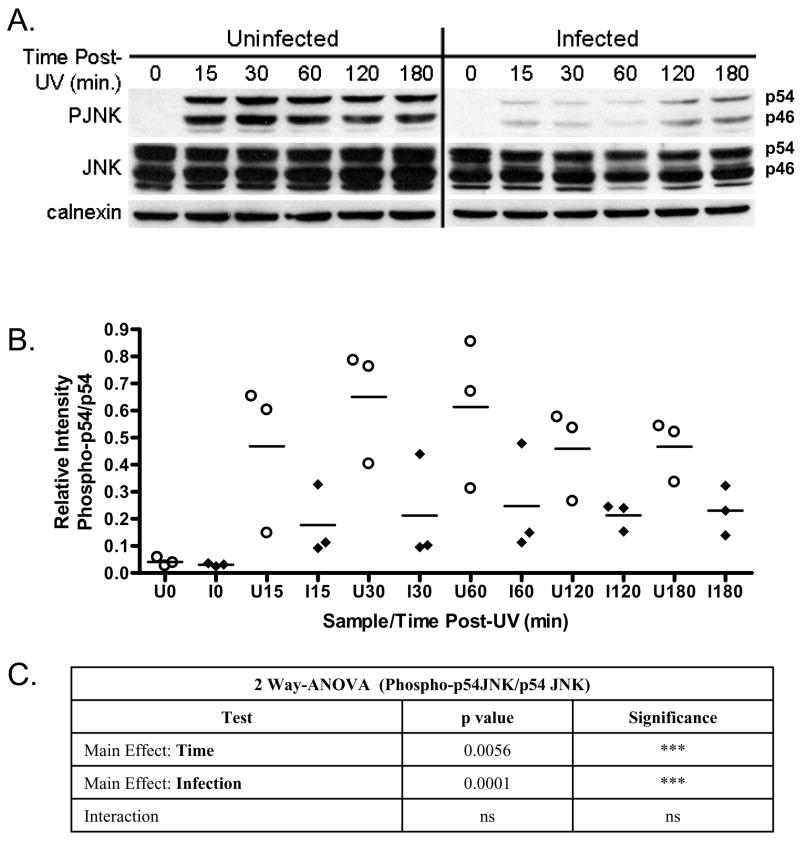
In the case of UV-induced apoptosis, one of the key signaling components required for triggering cytochrome c release from mitochondria is c-Jun N-terminal kinase (JNK) [11]. Exposure to UV results in the rapid and transient phosphorylation of JNK [27, 28]. Previously we found that T. gondii infection results in a marked decrease in phospho- JNK levels following UV irradiation of HeLa-cytochrome C-GFP expressing cells [2]. These experiments were replicated in unmodified HeLa cells to determine whether or not the overexpression of cytochrome c-GFP played a role in the previous observations (Fig. 1A). A marked difference in the levels of phospho-JNK between uninfected and infected UV-irradiated cells was observed across multiple experiments following densitometric analysis for p54 JNK (Fig. 1B) and p46-JNK respectively (data not shown). Variation was observed between experiments as noted by the distribution of relative p54 JNK phosphorylation (Fig. 1B). Statistical analysis confirms that both time post-UV and infection serve as highly significant though independent sources of variance in 2-way ANOVA analysis (Figure 1C). Notably, densitometric analyses of total JNK levels relative to the loading control calnexin showed no significant impact of infection or stimulation with UV (data not shown). Thus the effect on JNK phosphorylation is not due to changes in JNK levels due to infection.
Fig. 1. JNK activation is inhibited in T. gondii-infected HeLa cells following exposure to UV.
(A) A representative immunoblot of uninfected and T. gondii infected HeLa cells following exposure to UV irradiation and harvested at the time points indicated. JNK-phosphorylation (PJNK) occurs within 15 minutes post-UV and is sustained for the duration of the experiment in uninfected HeLa cells. This response is markedly inhibited both with regard to the extent of JNK phosphorylation and its kinetics in cells infected with T. gondii. Total JNK (JNK) and calnexin serve as loading controls. (B) Densitometric analysis representing the level of phospho-p54 JNK relative to total JNK in three independent experiments depicts the range of values inherent in such experiments. Relative phospho-p54 levels in uninfected cells (circle) versus infected cells (diamond) confirm a marked inhibition of mean phospho-p54 JNK accumulation (horizontal line) in parasite infected cells relative to uninfected cells. Densitometric analysis was performed as described in the materials and methods. (C) Statistical analysis using two way ANOVA indicates highly significant effects for both time post UV and infection. Time and infection as variables however do not exhibit significant interaction. Significance: p values <0.01 are considered highly significant (***), while p values between 0.01 and 0.05 are considered significant (**) and p-values > 0.05 are deemed not significant (ns).
T. gondii-infected cells fail to phosphorylate JNK following TNFα exposure
Tumor necrosis factor alpha (TNFα), another well studied apoptogenic trigger that induces the phosphorylation of JNK [29, 30], was used to stimulate uninfected and infected HeLa cells to examine the ability of T. gondii to block JNK phosphorylation due to a stimulus different from UV. Uninfected and infected (20 hr) cells were treated with 10 ng ml−1 TNFα and harvested at 7.5, 15, 30, 60, and 120 min reflecting the kinetics of TNFα mediated JNK phosphorylation (Fig. 2A). The cells harvested at 0 min were harvested prior to the addition of TNFα. Uninfected cells treated with TNFα exhibit a rapid and short lived period of JNK activation (Fig. 2A). Phospho-JNK levels increased dramatically in the uninfected cells after 15 min, peaked 30 min after the addition of TNFα and rapidly decreased, disappearing altogether after 2 hr (Fig. 2A). Consistent with the observation using UV irradiation, the response in T. gondii-infected cells was markedly attenuated, though only at the peak timepoint for phospho-JNK at 30 minutes (Fig. 2A). This pattern was confirmed across multiple experiments following densitometric analysis (Fig. 2B) and was not due to changes in the levels of JNK itself (2A, data not shown). As observed for the UV trigger, ANOVA analysis confirms both time post treatment and infection as highly significant effects contributing to variance (Fig. 2C). As noted with UV treatment, no significant interaction was noted between time and infection as co-variant effects (Fig. 2C).
Fig. 2. JNK activation is inhibited in T. gondii-infected HeLa cells following exposure to TNFα.
(A) A representative immunoblot of uninfected and T. gondii infected HeLa cells following exposure to TNFα and harvested at the time points indicated. JNK-phosphorylation (PJNK) occurs transiently, peaking at 30 minutes post exposure. This response is markedly inhibited in cells infected with T. gondii although the kinetics of the response do not appear to be impacted. Total JNK (JNK) and calnexin serve as loading controls. (B) Densitometric analysis representing the level of phospho-p54 JNK relative to total p54 JNK in four independent experiments depicts the range of values inherent in such experiments. Relative phospho-p54 levels in uninfected cells (open circle) versus infected cells (diamond) confirm a marked inhibition of mean phospho-p54 JNK accumulation (horizontal line) in parasite infected cells relative to uninfected cells. Densitometric analysis was performed as described in the materials and methods. (C) Statistical analysis using 2 way ANOVA indicate highly significant effects for both time post-TNFα exposure and infection. Time and infection do not exhibit significant interaction. Significance: p values of 0.01 and under are considered highly significant (***), p values between 0.01 and 0.05 are considered significant (**) while those above 0.05 are not significant (ns).
T. gondii-infection does not prevent JNK phosphorylation in mouse embryonic fibroblasts following exposure to UV
A substantial body of evidence indicates a critical role for NFκB signaling in the manipulation of the apoptotic cascade by T. gondii (reviewed in: [9]). The emerging evidence for crosstalk between the JNK and NFκB pathways [31, 32] led us to examine the effect of infection in both wild type mouse embryonic fibroblasts (MEF) and MEF derived from RelA/p65−/− transgenic embryos. This, we reasoned, would provide an efficient means to assess whether crosstalk between JNK and NFκB was manipulated by parasite infection.
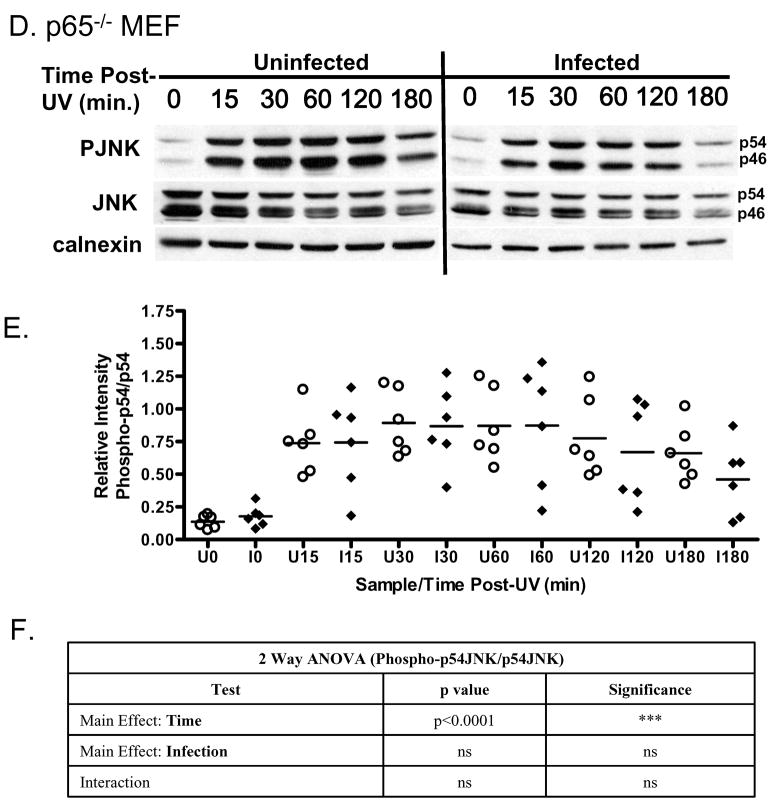
Uninfected and infected wild type (WT) MEF were UV irradiated at 100 mJ cm−2 in a fashion identical to that of the HeLa cells (Fig. 3A). Contrary to the observation in HeLa cells, JNK phosphorylation was consistently observed in both uninfected and infected MEF (Fig. 3A). This observation was confirmed densitiometrically (Fig. 3B). Both the mean relative phospho-JNK levels and the distribution patterns were indistinguishable between uninfected and infected cells (Fig. 3AB). The absence of inhibition of JNK phosphorylation was confirmed with the statistical analyses. ANOVA analysis indicates a highly significant effect for time post UV but not for infection (Fig. 3C). Thus in contrast to UV irradiated HeLa cells, no significant difference was noted at any time point between uninfected and infected wild type MEF (Fig. 3C).
Fig. 3. Phospho-JNK levels are not affected by T. gondii infection in wild type mouse embryonic fibroblasts and p65−/− cells following UV exposure.
(A) A representative immunoblot of uninfected and T. gondii-infected wild type mouse embryonic fibroblasts following exposure to UV irradiation and harvested at the time points indicated. JNK-phosphorylation (PJNK) occurs within 15 minutes post UV and is sustained for the duration of the experiment in uninfected MEF cells similar to the pattern observed in HeLa cells. This response is fundamentally identical with regard to the extent of JNK phosphorylation and its kinetics in cells infected with T. gondii. Total JNK (JNK) and calnexin serve as loading controls. (B) Densitometric of phospho-p54 JNK levels relative to total JNK in six independent experiments depicts the range of values inherent in such experiments. Relative phospho-p54 levels in uninfected cells (circle) versus infected cells (diamond) confirm an absence of inhibition of mean phospho-p54JNK accumulation (horizontal line) in parasite-infected cells relative (diamond) to uninfected cells (circle). Densitometric analysis was performed as described in the materials and methods (C) Statistical analysis using two-way ANOVA indicate a highly significant effect for time post-UV but not for infection. Time and infection do not exhibit significant interaction. The identical experiment to that performed with wild type MEF was replicated in p65−/− fibroblasts. Results indicate the absence of any significant inhibition in relative phospho-p54 JNK levels. (E) Densitometric analysis of six independent experiments confirms the broad range of responses and the lack of apparent differences in the mean levels of phospho-p54 JNK comparing uninfected (circle) and infected (diamond) p65−/− cells. (F) Two way ANOVA analysis reveals a highly significant effect for time post-UV but not infection. Significance: p values of 0.01 and lower are considered highly significant (***), p values between 0.01 and 0.05 are considered significant (**) while those of above 0.05 are not significant (ns).
The experiment was replicated in RelA/p65−/− cells which are deficient in NFκB signaling. The outcome was essentially identical to that observed in wild type MEF where T. gondii infection failed to prevent the phosphorylation of JNK following UV irradiation (Fig. 3D). As observed with wild type MEF (Fig. 3B), p65−/− cells exhibited a broad range of responses following UV in both uninfected and infected cells (Fig 3E). The statistical analysis using ANOVA confirms time post-UV as a highly significant effect (Fig 3F) but no net effect for infection (Fig. 3E). This suggests that the inhibition of JNK phosphorylation seen in HeLa cells is influenced by the cell background. The failure to observe any effect of infection on JNK phosphorylation following UV in both wild type and p65−/− cells indicate the host cell background is a key contributor to the response but does not rule out an effect with a different stimulus like TNFα.
TNFα-mediated activation of JNK is not repressed in T. gondii infected MEF
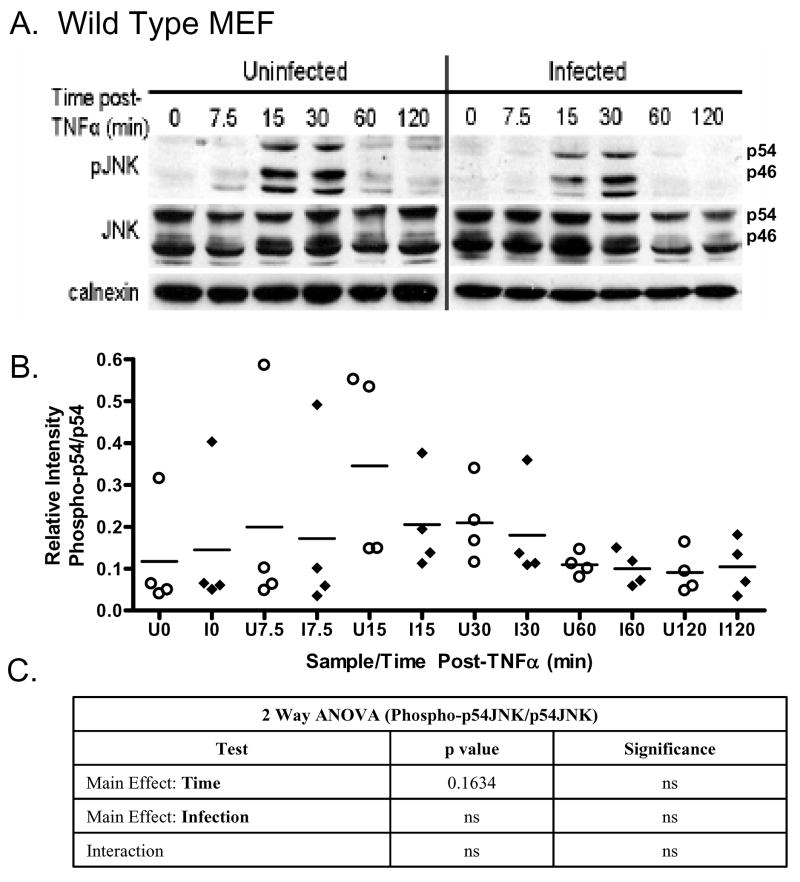
In light of the absence of inhibition of JNK phosphorylation in infected wild type MEF following UV irradiation, we examined the effect of infection on TNFα–dependent JNK phosphorylation. TNFα induced a rapid increase in the level of phospho-JNK in both uninfected and infected wild type MEF (Fig. 4A). The absence of an infection mediated difference in the extent and temporal profile of JNK phosphorylation following TNFα is evident across experiments, as is the marked variability in the responses (Fig. 4B). The ANOVA analysis confirms the lack of significance for both time and infection (Fig. 4C).. Notably, cell type specific effects on sustained JNK in response to TNFα have been previously noted [33].
Fig. 4. Phospho-JNK levels are not affected by T. gondii infection in wild type mouse embryonic fibroblasts and p65−/− cells following TNFα exposure.
(A) A representative immunoblot of uninfected and T. gondii infected wild type mouse embryonic fibroblasts following exposure to TNFα and harvested at the time points indicated. JNK-phosphorylation (PJNK) occurs within 15 minutes post-UV and is transient with a pattern similar to that seen in HeLa cells. This response is fundamentally identical with regard to the extent of JNK phosphorylation and its kinetics in cells infected with T. gondii. Total JNK (JNK) and calnexin serve as loading controls. (B) Densitometric analysis representing the level of phospho-p54 JNK relative to total JNK in four independent experiments depicts the range of values inherent in such experiments. Relative phospho-p54 levels in uninfected cells (circle) versus infected cells (diamond) confirm an absence of inhibition of mean phospho-p54 JNK accumulation (horizontal line) in parasite infected cells relative (diamond) to uninfected cells (circle). Densitometric analysis was performed as described in the Materials and methods (C) Statistical analysis using two way ANOVA indicate a modest effect for time post-treatment but not for infection. Time and infection do not exhibit significant interaction.. The identical experiment to that performed with wild type MEF was replicated in p65−/− fibroblasts. Results indicate the absence of any significant inhibition in relative phospho-p54JNK levels. (E) Densitometric analysis of six independent experiments confirms the broad range of responses and the lack of apparent differences in the mean levels of phospho-p54 JNK comparing uninfected (circle) and infected (diamond) p65−/− cells. (F) Two-way ANOVA analysis reveals a highly significant effect for time post-UV but not infection. Significance: p values of 0.01 and lower are considered highly significant (***), p values between 0.01 and 0.05 are considered significant (**) while those above 0.05 are not significant (ns).
An examination of the TNFα mediated effects in RelA/p65−/− cells effectively mirrored the results in wild type MEF (Fig. 4D). The apparent shift in the timing of JNK-phosphorylation (Fig 4D) was not borne out by statistical analysis (Fig. 4E,F). Consistent with the outcome in wild type MEF, ANOVA analysis indicated a significant effect for time but not infection. The absence, in MEF, of an infection mediated effect on JNK phosphorylation in response to both UV and TNFα treatment precludes any assessment of the role of NFκB in the manipulation of JNK signaling in this cell background. This does not however preclude a difference in the infection-responsive HeLa cells.
JNK phosphorylation in p65 knock-down HeLa cells
In order to assess the contribution of NFκB to the infection mediated suppression of JNK phosphorylation in HeLa cells, we sought to exploit siRNA knockdown technology targeting RelA/p65. Optimal conditions to effect a knockdown of p65 (p65-KD) were empirically determined. Cells transfected with 100 nM anti-p65 siRNA exhibited decreased levels of p65 as early as 24 hr post-transfection decreasing further at 48 hr post transfection (data not shown). Cells transfected with a non-specific siRNA (fluorescent siGLO transfection efficiency reporter (Dharmacon)) largely exhibited no change in p65 levels. However, at 72 hr post-transfection cells transfected with the highest level of siGLO exhibited a slight decrease in p65 (data not shown).
Immunofluorescence analysis was performed to determine the degree of p65 depletion at a single cell level as described in the materials and methods. Microscopic examination of these stained cells revealed a dose dependent decrease in the level of p65 at 48 hr (data not shown) and 72 hr post-transfection (Fig. 5A) using 25 (data not shown) and 100 nM specific siRNA. Transfection of 100 nM p65-siRNA resulted in profoundly reduced levels of staining by immunofluorescence using this qualitative measure (Fig. 5A).
Fig. 5. Depletion of RelA/p65 in HeLa cells does not affect the parasite mediated inhibition of JNK-phosphorylation in response to UV.
(A) Immunofluorescence analysis of untransfected HeLa cells reveals high levels of p65 in the cytoplasm relative to cells transfected with p65-targeting siRNA. Images were acquired under identical exposure conditions. Nuclei in the cells are labeled with Hoescht dye (blue). (B) Depletion of p65 recorded by immunoblot analysis comparing mock transfected cells (siGLO reagent) to target siRNA (p65) transfected cells. Aliquots of cells used in the experiments to examine the effect on JNK phosphorylation post-UV account for the different time points in the immunoblot. Note that UV exposure (comparison of t=0 relative to other time points) results in the apparent degradation of p65 selectively in infected siRNA transfected cells. (C) Immunoblot analysis confirms the effect of UV on JNK-phosphorylation as well as the inhibition of this effect in infected mock-transfected cells. ANOVA analysis indicated highly significant effects for time (p=0.0057) and infection (p=0.0138) with no interaction between these factors for mock treated cells. (D) siRNA mediated depletion of p65 (p65KD) does not have any effect on either the kinetics or temporal profile of JNK-phosphorylation in uninfected HeLa cells. ANOVA analysis confirms highly significant effects for time (p=0.0054) and infection (p=0.0013) with no interaction between these factors following p65 depletion. In addition, the capacity of T. gondii to affect JNK phosphorylation in response to UV is not affected by the depletion of p65. (E,F) Side by side comparison of mock transfected (M) vs p65 knock down (p65KD) HeLa cells reveals no significant effect on JNK phosphorylation in either uninfected (E) or infected (F) cells. Error bars represent the standard error of the mean from four independent experiments at each time point examined.
Following these pilot experiments, HeLa cells were transfected with 100 nM siRNA and incubated for 48 hr to deplete p65 prior to infection. After 48 hr, the cells were infected with T. gondii at an MOI of 8 for 20 hr. After 20 hr, mock (Lipofectamine reagent alone) and siRNA transfected cells with or without infection were exposed to UV (100 mJ cm−2) and harvested at 15, 30, 60, and 120 min post-UV. Again 0 min cells were harvested prior to UV exposure. Immunoblot analysis was performed to determine the degree of p65 depletion confirming marked reduction in steady state levels of p65 in both uninfected and infected cells relative to the calnexin control (Fig. 5B). Surprisingly, siRNA transfected, T. gondii-infected cells consistently exhibited a UV-dependent decrease in the level of p65 (Fig. 5B). Although the mechanism responsible for this consistent decrease is unknown, based on this observation we hypothesize that the concomitance of UV exposure and T. gondii exposure results in an increased rate of NFκB degradation (Fig. 5B). This may rely on an independent parasite derived activity.
The impact of UV irradiation was examined for mock and p65-targeted siRNA transfected HeLa cells. Uninfected mock-treated cells exhibited a rapid increase in levels of phospho-JNK which was sustained (Fig. 5C). Notably, depletion of p65 levels using siRNA (p65KD) had no effect on the phosphorylation of JNK. These data were borne out by the statistics in three independent experiments (Fig. 5E).
As predicted, the extent of JNK phosphorylation in mock-treated parasite- infected cells was lower relative to uninfected cells (Fig. 5C). The inhibition of JNK phosphorylation was not significantly altered following depletion of p65 using siRNA (Fig. 5D, F). These data suggest that even in the HeLa cell background where parasite infection inhibits UV mediated JNK phosphorylation NFκB does not play a significant role in the phenotype.
Decreased phospho-JNK in infected cells does not correlate with a decrease in the level of JNK activity
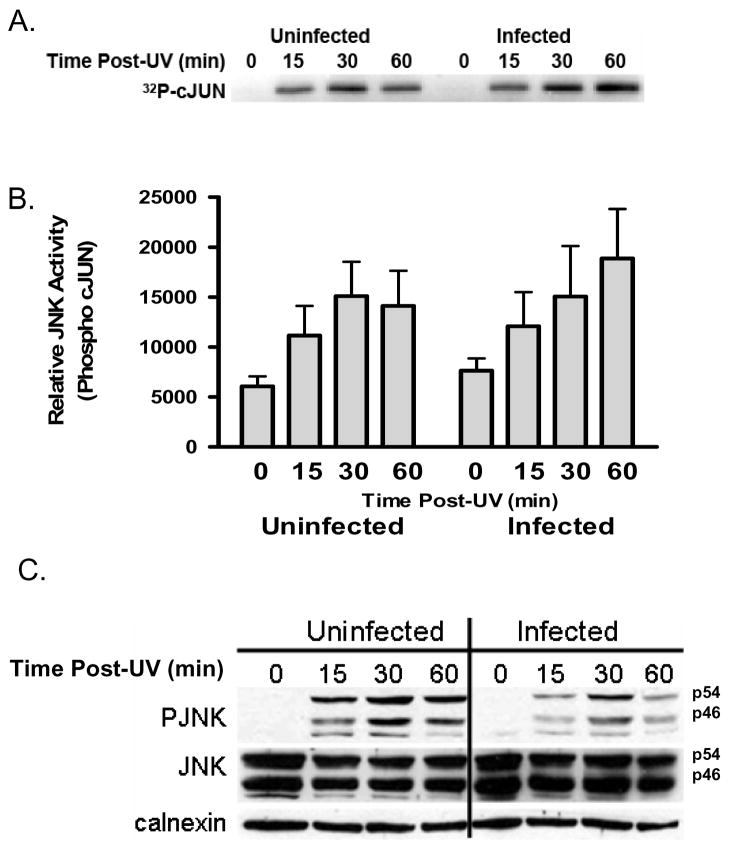
The decreased levels of phospho-JNK observed in infected HeLa cells following both UV and TNFα treatment via immunoblot analysis suggest that the parasite may curtail JNK activation. In vitro JNK assays were performed with immunoprecipitated JNK from uninfected and infected cells exposed to 100 mJ cm−2 to directly test this hypothesis. We specifically measured the direct phosphorylation of the recombinant JNK substrate GST-cJUN using densitometric analysis of signal on the phosphor imager (Fig. 6A). The capacity of immunoprecipitated activated JNK to phosphorylate cJUN was not impaired in parasite infected cells in three independent experiments (Fig 6A, B). A caveat, that immunoprecipitation can activate a dormant kinase however has not been ruled out [34]. An aliquot from each experiment was subjected to western blot analysis to confirm the inhibition of JNK-phosphorylation (Fig. 6C). This is a reproducible pattern which may reflect the differential phosphorylation of cJUN by various stimuli that does not reflect the entire spectrum of JNK substrates [29, 35–38]. It further suggests that a factor downstream of JNK itself, that is not co-immunoprecipitated, is responsible for the reduced phospho-JNK levels in infected cells. We therefore explored the possibility of an infection mediated phosphatase activity in the reduced steady state levels of phospho-JNK.
Fig. 6. T. gondii-dependent inhibition of JNK phosphorylation in HeLa cells does not correlate with a decrease in the level of JNK activity.
(A) The phosphorylation of cJUN by immunoprecipitated JNK following activation with UV was used as an indicator of JNK activity. Following the incubation of the immunoprecipitate with 32P-labeled ATP and recombinant cJUN, the phosphorylated cJUN was resolved via SDS-PAGE and visualized using a phosphorimager. A representative autoradiogram indicates UV-dependent activation JNK activity in both uninfected and infected cells. (B) Densitometric analysis was performed of three independent experiments. The analysis of 32P-cJUN levels confirmed that infection failed to decrease JNK activity but rather prolonged it. ANOVA analysis revealed no significant effect for time post UV or infection (data not shown). Error bars represent standard error of the mean from three independent experiments. (C) The absence of inhibition of JNK kinase activity as measured by cJUN phosphorylation following the immunoprecipitation of JNK does not correlate with the inhibition of phospho-JNK levels seen by immunoblot in an aliquot of the sample used for the immunoprecipitation.
T. gondii-mediated modulation of JNK activation in cells treated with okadaic acid
In addition to the inhibition of its activation, JNK is negatively regulated by specific protein phosphatases [15, 39]. The lack of inhibition of JNK activity following immunoprecipitation (Fig. 6) suggests that the reduced levels of phospho-JNK in infected cells may be due to a phosphatase activity that does not co-immunoprecipitate with JNK. The role of phosphatases in the inhibition of JNK phosphorylation by infection was tested by pre-treating HeLa cells with okadaic acid (OA). Okadaic acid is a cell permeable compound that inhibits protein phosphatases 1, 2A, and 2B in a dose dependent manner. While specific JNK-phosphatase inhibitors exist they are non-membrane permeant precluding their use in whole cell systems [40, 41].
We reasoned that if an OA-sensitive phosphatase was responsible for the lack of accumulation of phospho-JNK following UV treatment, drug treatment would reverse the phenotype. Following a 2 hr pre-treatment with 100 nM okadaic acid, cells were exposed to 100 mJ cm−2 UV and processed as in prior experiments. Treatment of uninfected cells with okadaic acid had no effect on either the extent or temporal profile of JNK phosphorylation in response to UV relative to carrier control treated cells (Fig. 7A,B,C). This is consistent with okadaic acid not targeting the dual specificity phosphatases that account for classical JNK-phosphatase activities [40, 41]. We next examined the effect of okadaic acid on JNK-phosphorylation in parasite infected cells following UV irradiation. Control treated infected cells exhibited the inhibition of JNK phosphorylation relative to uninfected cells as predicted (Fig. 7A,C,D). Interestingly, okadaic acid was found to have an effect on the phosphorylation of JNK following UV treatment in infected cells with a small but reproducible increase in levels of phospho-JNK (Fig. 7B,D) relative to the control treated cells. This was not observed in the uninfected okadaic acid treated cells (Fig. 7C). This would suggest a phosphatase activity other than the conventional JNK-phosphatases contributes to the turnover of phospho-JNK. It is tempting to suggest that this activity may be parasite derived in light of recent evidence for parasite proteins being secreted into the host cell during invasion. The direct confirmation of the role of JNK-phosphatases will have to await the availability of membrane permeant inhibitors of dual specificity phosphatases.
Fig. 7. Effect of inhibition of phosphatase activity using okadaic acid on phospho-JNK levels.
HeLa cells were pre-treated with okadaic acid (100 nM) or the carrier control for 2 hr prior to UV irradiation (100 mJ cm−2). Cells were returned to either prewarmed treatment media or carrier control containing media immediately following irradiation and the kinetics of PJNK accumulation determined. (A,B) Treatment of uninfected cells with okadaic acid failed to increase the levels of phospho-JNK relative to the carrier control. Infected cells treated with both carrier control and okadaic acid exhibit lower levels of phospho-JNK compared to treatment and time matched uninfected cells. However, comparison of phospho-JNK levels between control and okadaic acid treated cells suggest a potential increase in relative phospho-JNK levels. (C,D) Densitometric analysis compiling the results from three independent experiments confirm the absence of any effect from okadaic acid in uninfected cells except possible a marginally increased loss of signal in okadiac acid treated cells. In contrast, treatment of infected cells with okadaic acid results in a significant increase in the levels of phospho-JNK relative to total JNK which are comparable to those seen in uninfected cells. This suggests an okadaic acid sensitive activity is partially responsible for the reduced steady state levels of phospho-JNK following UV stimulation. 2-way ANOVA analysis revealed highly significant effect for time post UV (p<0.0001). Okadaic acid treatment exhibited no significant effect in uninfected cells but was moderately significant in infected cells (p=0.0265). Error bars represent standard error of the mean from three independent experiments.
DISCUSSION
Apoptotic pathways are under considerable regulatory control [42, 43]. The MAPK, JNK, [44] has been shown to be required for the transmission of the apoptogenic stimulus of UV irradiation and is required for the release of cytochrome c from mitochondria [11]. UV irradiation results in a transient increase in the level of JNK phosphorylation and activity [27, 45, 46]. The role of JNK, as either a pro-apoptotic or pro-survival mediator, is dependent upon the magnitude and duration of JNK activation and appears to be cell type specific [46, 47]. The regulation of JNK activity is governed both by upstream signaling cascades and downstream effectors, most notably phosphatases.
In an earlier study, we found that Toxoplasma gondii infection decreases the level of phospho-JNK in HeLa cells expressing a cytochrome c-GFP reporter exposed to UV irradiation [2]. The decreased phospho-JNK levels correlated with a marked inhibition of cytochrome c-GFP release from host cell mitochondria [2]. The inhibition by infection of UV-mediated JNK phosphorylation was observed in unmodified HeLa cells (Fig. 1A) and confirmed using densitometry across multiple experiments (Fig. 1B).
To determine the extent of the T. gondii-mediated modulation of the JNK pathway, JNK phosphorylation in response to TNFα treatment was also determined [29, 48]. TNFα-mediated JNK activation provides an alternative stimulus with which to examine the impact of parasite infection, which is well known to trigger inflammatory responses [49]. The observation that in HeLa cells T. gondii infection effectively inhibits JNK-phosphorylation following TNFα stimulation (Figure 2A,B) suggests a common mechanism may be involved for these stimuli despite the unique pathways each of them activate.
Our earlier studies implicate the NFκB regulatory cascade as a central component in the T. gondii-mediated blockade of host apoptosis [3, 9, 50]. This centrally important transcription factor has recently been shown to negatively regulate the activation of JNK [12, 13, 19, 51, 52]. These effects have been noted both with regard to UV, TNFα and other stressors in human epithelial cells [53]. In light of this potential crosstalk, we sought to examine the role of NFκB in the inhibition of JNK phosphorylation in T. gondii-infected cells. To facilitate these studies, we shifted from the HeLa cell background to mouse embryonic fibroblasts (MEF) as this cell system affords the use of cell lines lacking the NFκB component RelA/p65 [23]. These cells were instrumental in defining the role of NFκB in the parasite mediated blockade of apoptosis [3]. Despite the robust phenotype observed in both unmodified (Figure 2,3) and cytochrome c-GFP expressing HeLa cells [2], T. gondii-infection did not reduce the level of phospho-JNK in MEF following exposure to either UV (Fig. 3A–C) or TNFα (Fig. 4A–C). In addition, the effect of both treatments on JNK phosphorylation was found to be variable with regard to the extent and timing of JNK-phosphorylation in both uninfected and parasite infected cells (Fig. 3B,4B). Despite the absence of an infection mediated effect (ANOVA analysis Fig. 3C,4C) in wild type cells, we examined whether infection of p65−/− fibroblasts altered the response profile. The outcome in the NFκB deficient cells (p65−/−) (Fig. 3D–F. 4.D–F) was essentially identical to that observed in wild type MEF. This suggests that the modulation of host cell signaling pathways is dependent on both the parasite and the cell it infects. Differential cell type specific responses to activating stimuli like TNFα have been previously reported [33]. The reason for this profound difference is unclear but may lie in the fact that HeLa cells are transformed while the MEF represent immortalized primary cells. Such differences have been observed in the susceptibility of tumor cells to apoptosis following expression of the protein Par-4 to which non-transformed cells are refractory [54]. In addition, a similar differential phenotype directly connected to JNK activation is observed with normal cells and epithelial cell tumors in co-culture systems with macrophages as a source of TNFα [55].
The failure to detect an effect of NFκB in the parasite-mediated suppression of phospho-JNK levels following stimulation in MEF does not rule out an effect in a responsive cell line such as a HeLa cell. We therefore employed siRNA knockdown technology to achieve a targeted knockdown of RelA/p65−/− in the responsive HeLa background. Efficient knock down of p65 was achieved as confirmed by both immunofluorescence analysis (Fig. 5A) and immunoblot analysis (Fig. 5B). In the specific p65 targeting siRNA treated cells, comparably low steady state levels of RelA/p65 were present un both uninfected and infected cells (t=0) although UV treatment markedly destabilized the protein selectively in the infected cell population (Fig. 5B). As expected, mock transfected cells exhibited robust phosphorylation of JNK in response to UV which was markedly attenuated in parasite infected cells (Fig. 5C). This effect was not significantly impacted in cells knocked down for p65 (p65KD) either with regard to the extent of JNK phosphorylation or the parasite mediated inhibition of that response (Fig. 5. D–F). Acknowledging the caveat that a gene knockdown is not a knockout and that there may be enough p65 to reflect wild type conditions, we conclude that NFκB does not contribute to the parasite mediated suppression of JNK phosphorylation. Given the critical importance of NFkB in the blockade of apoptosis [3] we propose that the manipulation of JNK phosphorylation does not play a generalized role in the parasite-mediated blockade of apoptosis. While reduced phospho-JNK levels correlate well with the blockade of cytochrome c release from mitochondria and apoptosis seen in HeLa cells [2], its effect is clearly cell type and context specific.
The phosphorylation of JNK serves as a convenient predictor of JNK activity following stimulation. The reduced levels of phospho-JNK in parasite infected HeLa cells suggest reduced JNK activity and suppression of JNK-activated functions. We sought to directly measure the levels of JNK activity following the UV stimulation of uninfected and parasite infected cells using the capacity of immunoprecipitated JNK to phosphorylate a recombinant JNK substrate (GST-cJUN) as a measure (Fig. 6A). As predicted, non-stimulated uninfected cells lacked active JNK with markedly increased activity in subsequent time points (Fig. 6A). This activity was found to plateau between the 30 and 60 minute time points (Fig 6B). Surprisingly, despite an efficient suppression of JNK phosphorylation using immunoblot analysis in matched infected cells (Fig. 6C), actual JNK activity was found to be on par with uninfected cells and increasing at a time when that in the uninfected population plateaus (Fig. 6A,B). This surprising observation further reinforces the contention that while the manipulation of JNK may occur during parasite infection its purpose is not in the manipulation of the apoptotic cascade. A caveat, that the act of immunoprecipitation alters the activity of JNK by either directly activating the kinase [34] or displacing an inhibitory phosphatase must be presented. Arguing against the kinase being activated by antibody binding is the complete lack of activity in the t=0 time point for both uninfected and infected cells (Fig. 6A). Toward examining the latter caveat, we explored whether a phosphatase, potentially displaced in the immunoprecipitation, regulated JNK activity in parasite infected cells.
In defining the properties of the potential target responsible for the inhibition of phospho-JNK accumulation in infected HeLa cells, we focused on the potential role of phosphatases. The distinct upstream signaling pathways used by UV and TNFα to activate JNK [44] make the downstream JNK-phosphatase activities the most likely effectors of the absence of phospho-JNK in infected cells. Known JNK-phosphatases are classified as dual specificity phosphatases, targeting both phospho-tyrosine and phospho-threonine residues [14, 41]. These dual specificity phosphatases have been shown to be important in regulating JNK activity in response to stimuli including UV [56–58] and cytokines, including TNFα, triggered during inflammation [59–62]. Furthermore, a role for reactive oxygen species [48, 63], similar to those induced by UV and TNFα are reported to govern JNK-phosphatase activities. Unfortunately, effective cell permeable inhibitors targeting dual specificity phosphatases in intact cells are not available [14, 41]. We therefore used okadaic acid, a well characterized cell permeable broad spectrum phosphatase inhibitor [64], to assess whether phosphatases are responsible for the decreased phospho-JNK levels observed in infected HeLa cells following exposure to UV or TNFα. Okadiac acid is not an efficient inhibitor of dual specificity phosphatases [64], a factor highlighted by the absence of any meaningful suppression (which should manifest as an increased level of JNK-phosphorylation) in uninfected cells following UV (Fig. 7C). In contrast, treatment of infected cells with okadaic acid resulted in an increased relative level of phospho-JNK relative to the carrier control. This effect is significant in that the levels of relative phosphorylation between the uninfected and infected okadaic acid treated cells is comparable (Fig. 7C,D). Furthermore, this observation suggests that an okadaic acid sensitive activity, distinct from the conventional JNK-phosphatases is involved. This is intriguing in light of the recent identification of a parasite protein phosphatase 2C activity secreted by the parasite into the host cell [65]. While the differential response dependent on the host cell infected (HeLa (Figure 1,2) vs. MEF (Fig. 3,4)) suggests that a parasite activity such as a secreted phosphatase is not responsible, it cannot be ruled out as the cellular milieus may differ in the presence of an activity that inhibits a parasite phosphatase.
The differences in how a critical regulatory pathway like JNK is impacted by infection and is affected by the host cell type raises several important issues. Primary among these is the need for caution in our interpretation of the impact of any infectious agent on the of the host cell as the outcome may be highly context specific [9]. This study reveals the peril in extrapolating data generated in a single host cell system to define the cellular response to infection with a pathogenic agent. While central themes may be apparent, exceptions to the “rule” should be accepted rather than dismissed as artifact. Such differences in host cell types, transformation status and experimental models likely reflect the spectrum of reported observations that contribute to what are perceived as “controversial interpretations” where in reality no controversy may exist. An acceptance of the diversity of responses triggered in a host cell, itself a highly dynamic and responsive entity, by an equally dynamic pathogen provides a more complex but likely more accurate picture of pathogen-host interactions.
Acknowledgments
The authors wish to acknowledge members of the Sinai laboratory for their insights in the execution of this work. This work was supported by NIH grant AI49367 to APS and T32 AI49795 to JCC. JCC was also supported by a Dissertation Year fellowship from the University of Kentucky.
References
- 1.Tenter AM, Heckeroth AR, Weiss LM. Toxoplasma gondii: from animals to humans. Int J Parasitol. 2000;30:1217–58. doi: 10.1016/s0020-7519(00)00124-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Carmen JC, Hardi L, Sinai AP. Toxoplasma gondii inhibits ultraviolet light-induced apoptosis through multiple interactions with the mitochondrion-dependent programmed cell death pathway. Cell Microbiol. 2006;8:301–15. doi: 10.1111/j.1462-5822.2005.00622.x. [DOI] [PubMed] [Google Scholar]
- 3.Payne TM, Molestina RE, Sinai AP. Inhibition of caspase activation and a requirement for NF-kappaB function in the Toxoplasma gondii-mediated blockade of host apoptosis. J Cell Sci. 2003;116:4345–58. doi: 10.1242/jcs.00756. [DOI] [PubMed] [Google Scholar]
- 4.Nash PB, Purner MB, Leon RP, Clarke P, Duke RC, Curiel TJ. Toxoplasma gondii-infected cells are resistant to multiple inducers of apoptosis. J Immunol. 1998;160:1824–30. [PubMed] [Google Scholar]
- 5.Goebel S, Gross U, Luder CG. Inhibition of host cell apoptosis by Toxoplasma gondii is accompanied by reduced activation of the caspase cascade and alterations of poly(ADP-ribose) polymerase expression. J Cell Sci. 2001;114:3495–505. doi: 10.1242/jcs.114.19.3495. [DOI] [PubMed] [Google Scholar]
- 6.Vutova P, Wirth M, Hippe D, Gross U, Schulze-Osthoff K, Schmitz I, Luder CG. Toxoplasma gondii inhibits Fas/CD95-triggered cell death by inducing aberrant processing and degradation of caspase 8. Cell Microbiol. 2007;9:1556–70. doi: 10.1111/j.1462-5822.2007.00893.x. [DOI] [PubMed] [Google Scholar]
- 7.Hippe D, Lytovchenko O, Schmitz I, Luder CG. Fas/CD95-mediated apoptosis of type II cells is blocked by Toxoplasma gondii primarily via interference with the mitochondrial amplification loop. Infect Immun. 2008;76:2905–12. doi: 10.1128/IAI.01546-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kim L, Denkers EY. Toxoplasma gondii triggers Gi-dependent PI 3-kinase signaling required for inhibition of host cell apoptosis. J Cell Sci. 2006;119:2119–26. doi: 10.1242/jcs.02934. [DOI] [PubMed] [Google Scholar]
- 9.Carmen JC, Sinai AP. Suicide prevention: disruption of apoptotic pathways by protozoan parasites. Mol Microbiol. 2007;64:904–16. doi: 10.1111/j.1365-2958.2007.05714.x. [DOI] [PubMed] [Google Scholar]
- 10.Weston CR, Davis RJ. The JNK signal transduction pathway. Curr Opin Cell Biol. 2007;19:142–9. doi: 10.1016/j.ceb.2007.02.001. [DOI] [PubMed] [Google Scholar]
- 11.Tournier C, Hess P, Yang DD, Xu J, Turner TK, Nimnual A, Bar-Sagi D, Jones SN, Flavell RA, Davis RJ. Requirement of JNK for stress-induced activation of the cytochrome c-mediated death pathway. Science. 2000;288:870–4. doi: 10.1126/science.288.5467.870. [DOI] [PubMed] [Google Scholar]
- 12.Papa S, Bubici C, Zazzeroni F, Pham CG, Kuntzen C, Knabb JR, Dean K, Franzoso G. The NF-kappaB-mediated control of the JNK cascade in the antagonism of programmed cell death in health and disease. Cell Death Differ. 2006;13:712–29. doi: 10.1038/sj.cdd.4401865. [DOI] [PubMed] [Google Scholar]
- 13.Bubici C, Papa S, Pham CG, Zazzeroni F, Franzoso G. The NF-kappaB-mediated control of ROS and JNK signaling. Histol Histopathol. 2006;21:69–80. doi: 10.14670/HH-21.69. [DOI] [PubMed] [Google Scholar]
- 14.Abraham SM, Clark AR. Dual-specificity phosphatase 1: a critical regulator of innate immune responses. Biochem Soc Trans. 2006;34:1018–23. doi: 10.1042/BST0341018. [DOI] [PubMed] [Google Scholar]
- 15.Theodosiou A, Ashworth A. MAP kinase phosphatases. Genome Biol. 2002;3:REVIEWS3009. doi: 10.1186/gb-2002-3-7-reviews3009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Karin M, Lin A. NF-kappaB at the crossroads of life and death. Nat Immunol. 2002;3:221–7. doi: 10.1038/ni0302-221. [DOI] [PubMed] [Google Scholar]
- 17.De Smaele E, Zazzeroni F, Papa S, Nguyen DU, Jin R, Jones J, Cong R, Franzoso G. Induction of gadd45beta by NF-kappaB downregulates pro-apoptotic JNK signalling. Nature. 2001;414:308–13. doi: 10.1038/35104560. [DOI] [PubMed] [Google Scholar]
- 18.Reuther-Madrid JY, Kashatus D, Chen S, Li X, Westwick J, Davis RJ, Earp HS, Wang CY, Baldwin AS., Jr The p65/RelA subunit of NF-kappaB suppresses the sustained, antiapoptotic activity of Jun kinase induced by tumor necrosis factor. Mol Cell Biol. 2002;22:8175–83. doi: 10.1128/MCB.22.23.8175-8183.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tang G, Minemoto Y, Dibling B, Purcell NH, Li Z, Karin M, Lin A. Inhibition of JNK activation through NF-kappaB target genes. Nature. 2001;414:313–7. doi: 10.1038/35104568. [DOI] [PubMed] [Google Scholar]
- 20.Papa S, Zazzeroni F, Bubici C, Jayawardena S, Alvarez K, Matsuda S, Nguyen DU, Pham CG, Nelsbach AH, Melis T, De Smaele E, Tang WJ, D’Adamio L, Franzoso G. Gadd45 beta mediates the NF-kappa B suppression of JNK signalling by targeting MKK7/JNKK2. Nat Cell Biol. 2004;6:146–53. doi: 10.1038/ncb1093. [DOI] [PubMed] [Google Scholar]
- 21.Pham CG, Bubici C, Zazzeroni F, Papa S, Jones J, Alvarez K, Jayawardena S, De Smaele E, Cong R, Beaumont C, Torti FM, Torti SV, Franzoso G. Ferritin heavy chain upregulation by NF-kappaB inhibits TNFalpha-induced apoptosis by suppressing reactive oxygen species. Cell. 2004;119:529–42. doi: 10.1016/j.cell.2004.10.017. [DOI] [PubMed] [Google Scholar]
- 22.Lee EG, Boone DL, Chai S, Libby SL, Chien M, Lodolce JP, Ma A. Failure to regulate TNF-induced NF-kappaB and cell death responses in A20-deficient mice. Science. 2000;289:2350–4. doi: 10.1126/science.289.5488.2350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gapuzan ME, Schmah O, Pollock AD, Hoffmann A, Gilmore TD. Immortalized fibroblasts from NF-kappaB RelA knockout mice show phenotypic heterogeneity and maintain increased sensitivity to tumor necrosis factor alpha after transformation by v-Ras. Oncogene. 2005;24:6574–83. doi: 10.1038/sj.onc.1208809. [DOI] [PubMed] [Google Scholar]
- 24.Donald RG, Roos DS. Gene knock-outs and allelic replacements in Toxoplasma gondii: HXGPRT as a selectable marker for hit-and-run mutagenesis. Mol Biochem Parasitol. 1998;91:295–305. doi: 10.1016/s0166-6851(97)00210-7. [DOI] [PubMed] [Google Scholar]
- 25.Hebert DN, Foellmer B, Helenius A. Glucose trimming and reglucosylation determine glycoprotein association with calnexin in the endoplasmic reticulum. Cell. 1995;81:425–33. doi: 10.1016/0092-8674(95)90395-x. [DOI] [PubMed] [Google Scholar]
- 26.Molestina RE, Sinai AP. Detection of a novel parasite kinase activity at the Toxoplasma gondii parasitophorous vacuole membrane capable of phosphorylating host IkappaBalpha. Cell Microbiol. 2005;7:351–62. doi: 10.1111/j.1462-5822.2004.00463.x. [DOI] [PubMed] [Google Scholar]
- 27.Rosette C, Karin M. Ultraviolet light and osmotic stress: activation of the JNK cascade through multiple growth factor and cytokine receptors. Science. 1996;274:1194–7. doi: 10.1126/science.274.5290.1194. [DOI] [PubMed] [Google Scholar]
- 28.Sudo T, Karin M. Assays for JNK and p38 mitogen-activated protein kinases. Methods Enzymol. 2000;322:388–92. doi: 10.1016/s0076-6879(00)22037-5. [DOI] [PubMed] [Google Scholar]
- 29.Deng Y, Ren X, Yang L, Lin Y, Wu X. A JNK-dependent pathway is required for TNFalpha-induced apoptosis. Cell. 2003;115:61–70. doi: 10.1016/s0092-8674(03)00757-8. [DOI] [PubMed] [Google Scholar]
- 30.Westwick JK, Weitzel C, Minden A, Karin M, Brenner DA. Tumor necrosis factor alpha stimulates AP-1 activity through prolonged activation of the c-Jun kinase. J Biol Chem. 1994;269:26396–401. [PubMed] [Google Scholar]
- 31.Bubici C, Papa S, Dean K, Franzoso G. Mutual cross-talk between reactive oxygen species and nuclear factor-kappa B: molecular basis and biological significance. Oncogene. 2006;25:6731–48. doi: 10.1038/sj.onc.1209936. [DOI] [PubMed] [Google Scholar]
- 32.Bubici C, Papa S, Pham CG, Zazzeroni F, Franzoso G. NF-kappaB and JNK: an intricate affair. Cell Cycle. 2004;3:1524–9. doi: 10.4161/cc.3.12.1321. [DOI] [PubMed] [Google Scholar]
- 33.Wicovsky A, Muller N, Daryab N, Marienfeld R, Kneitz C, Kavuri S, Leverkus M, Baumann B, Wajant H. Sustained JNK activation in response to tumor necrosis factor is mediated by caspases in a cell type-specific manner. J Biol Chem. 2007;282:2174–83. doi: 10.1074/jbc.M606167200. [DOI] [PubMed] [Google Scholar]
- 34.Cui J, Holgado-Madruga M, Su W, Tsuiki H, Wedegaertner P, Wong AJ. Identification of a specific domain responsible for JNK2alpha2 autophosphorylation. J Biol Chem. 2005;280:9913–20. doi: 10.1074/jbc.M412165200. [DOI] [PubMed] [Google Scholar]
- 35.Papadakis ES, Finegan KG, Wang X, Robinson AC, Guo C, Kayahara M, Tournier C. The regulation of Bax by c-Jun N-terminal protein kinase (JNK) is a prerequisite to the mitochondrial-induced apoptotic pathway. FEBS Lett. 2006;580:1320–6. doi: 10.1016/j.febslet.2006.01.053. [DOI] [PubMed] [Google Scholar]
- 36.Lei K, Davis RJ. JNK phosphorylation of Bim-related members of the Bcl2 family induces Bax-dependent apoptosis. Proc Natl Acad Sci U S A. 2003;100:2432–7. doi: 10.1073/pnas.0438011100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Tsuruta F, Sunayama J, Mori Y, Hattori S, Shimizu S, Tsujimoto Y, Yoshioka K, Masuyama N, Gotoh Y. JNK promotes Bax translocation to mitochondria through phosphorylation of 14-3-3 proteins. Embo J. 2004;23:1889–99. doi: 10.1038/sj.emboj.7600194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Oleinik NV, Krupenko NI, Krupenko SA. Cooperation between JNK1 and JNK2 in activation of p53 apoptotic pathway. Oncogene. 2007;26:7222–30. doi: 10.1038/sj.onc.1210526. [DOI] [PubMed] [Google Scholar]
- 39.Kondoh K, Nishida E. Regulation of MAP kinases by MAP kinase phosphatases. Biochim Biophys Acta. 2007;1773:1227–37. doi: 10.1016/j.bbamcr.2006.12.002. [DOI] [PubMed] [Google Scholar]
- 40.Lazo JS, Nunes R, Skoko JJ, Queiroz de Oliveira PE, Vogt A, Wipf P. Novel benzofuran inhibitors of human mitogen-activated protein kinase phosphatase-1. Bioorg Med Chem. 2006;14:5643–50. doi: 10.1016/j.bmc.2006.04.036. [DOI] [PubMed] [Google Scholar]
- 41.Vogt A, Lazo JS. Discovery of protein kinase phosphatase inhibitors. Methods Mol Biol. 2007;356:389–400. doi: 10.1385/1-59745-217-3:389. [DOI] [PubMed] [Google Scholar]
- 42.Danial NN, Korsmeyer SJ. Cell death: critical control points. Cell. 2004;116:205–19. doi: 10.1016/s0092-8674(04)00046-7. [DOI] [PubMed] [Google Scholar]
- 43.Ott M, Gogvadze V, Orrenius S, Zhivotovsky B. Mitochondria, oxidative stress and cell death. Apoptosis. 2007;12:913–22. doi: 10.1007/s10495-007-0756-2. [DOI] [PubMed] [Google Scholar]
- 44.Davis RJ. Signal transduction by the JNK group of MAP kinases. Cell. 2000;103:239–52. doi: 10.1016/s0092-8674(00)00116-1. [DOI] [PubMed] [Google Scholar]
- 45.Chen YR, Wang X, Templeton D, Davis RJ, Tan TH. The role of c-Jun N-terminal kinase (JNK) in apoptosis induced by ultraviolet C and gamma radiation. Duration of JNK activation may determine cell death and proliferation. J Biol Chem. 1996;271:31929–36. doi: 10.1074/jbc.271.50.31929. [DOI] [PubMed] [Google Scholar]
- 46.Liu J, Lin A. Role of JNK activation in apoptosis: a double-edged sword. Cell Res. 2005;15:36–42. doi: 10.1038/sj.cr.7290262. [DOI] [PubMed] [Google Scholar]
- 47.Johnson GL, Nakamura K. The c-jun kinase/stress-activated pathway: regulation, function and role in human disease. Biochim Biophys Acta. 2007;1773:1341–8. doi: 10.1016/j.bbamcr.2006.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kamata H, Honda S, Maeda S, Chang L, Hirata H, Karin M. Reactive oxygen species promote TNFalpha-induced death and sustained JNK activation by inhibiting MAP kinase phosphatases. Cell. 2005;120:649–61. doi: 10.1016/j.cell.2004.12.041. [DOI] [PubMed] [Google Scholar]
- 49.Konen-Waisman S, Howard JC. Cell-autonomous immunity to Toxoplasma gondii in mouse and man. Microbes Infect. 2007;9:1652–61. doi: 10.1016/j.micinf.2007.09.005. [DOI] [PubMed] [Google Scholar]
- 50.Molestina RE, Payne TM, Coppens I, Sinai AP. Activation of NF-kappaB by Toxoplasma gondii correlates with increased expression of antiapoptotic genes and localization of phosphorylated IkappaB to the parasitophorous vacuole membrane. J Cell Sci. 2003;116:4359–71. doi: 10.1242/jcs.00683. [DOI] [PubMed] [Google Scholar]
- 51.Lin A. Activation of the JNK signaling pathway: breaking the brake on apoptosis. Bioessays. 2003;25:17–24. doi: 10.1002/bies.10204. [DOI] [PubMed] [Google Scholar]
- 52.Liu J, Yang D, Minemoto Y, Leitges M, Rosner MR, Lin A. NF-kappaB is required for UV-induced JNK activation via induction of PKCdelta. Mol Cell. 2006;21:467–80. doi: 10.1016/j.molcel.2005.12.020. [DOI] [PubMed] [Google Scholar]
- 53.Yuan ZQ, Feldman RI, Sun M, Olashaw NE, Coppola D, Sussman GE, Shelley SA, Nicosia SV, Cheng JQ. Inhibition of JNK by cellular stress- and tumor necrosis factor alpha-induced AKT2 through activation of the NF kappa B pathway in human epithelial Cells. J Biol Chem. 2002;277:29973–82. doi: 10.1074/jbc.M203636200. [DOI] [PubMed] [Google Scholar]
- 54.Ranganathan P, Rangnekar VM. Regulation of cancer cell survival by Par-4. Ann N Y Acad Sci. 2005;1059:76–85. doi: 10.1196/annals.1339.046. [DOI] [PubMed] [Google Scholar]
- 55.Hagemann T, Wilson J, Kulbe H, Li NF, Leinster DA, Charles K, Klemm F, Pukrop T, Binder C, Balkwill FR. Macrophages induce invasiveness of epithelial cancer cells via NF-kappa B and JNK. J Immunol. 2005;175:1197–205. doi: 10.4049/jimmunol.175.2.1197. [DOI] [PubMed] [Google Scholar]
- 56.Cadalbert L, Sloss CM, Cameron P, Plevin R. Conditional expression of MAP kinase phosphatase-2 protects against genotoxic stress-induced apoptosis by binding and selective dephosphorylation of nuclear activated c-jun N-terminal kinase. Cell Signal. 2005;17:1254–64. doi: 10.1016/j.cellsig.2005.01.003. [DOI] [PubMed] [Google Scholar]
- 57.Hamdi M, Kool J, Cornelissen-Steijger P, Carlotti F, Popeijus HE, van der Burgt C, Janssen JM, Yasui A, Hoeben RC, Terleth C, Mullenders LH, van Dam H. DNA damage in transcribed genes induces apoptosis via the JNK pathway and the JNK-phosphatase MKP-1. Oncogene. 2005;24:7135–44. doi: 10.1038/sj.onc.1208875. [DOI] [PubMed] [Google Scholar]
- 58.Hirsch DD, Stork PJ. Mitogen-activated protein kinase phosphatases inactivate stress-activated protein kinase pathways in vivo. J Biol Chem. 1997;272:4568–75. doi: 10.1074/jbc.272.7.4568. [DOI] [PubMed] [Google Scholar]
- 59.Chen P, Li J, Barnes J, Kokkonen GC, Lee JC, Liu Y. Restraint of proinflammatory cytokine biosynthesis by mitogen-activated protein kinase phosphatase-1 in lipopolysaccharide-stimulated macrophages. J Immunol. 2002;169:6408–16. doi: 10.4049/jimmunol.169.11.6408. [DOI] [PubMed] [Google Scholar]
- 60.Chi H, Barry SP, Roth RJ, Wu JJ, Jones EA, Bennett AM, Flavell RA. Dynamic regulation of pro- and anti-inflammatory cytokines by MAPK phosphatase 1 (MKP-1) in innate immune responses. Proc Natl Acad Sci U S A. 2006;103:2274–9. doi: 10.1073/pnas.0510965103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.De Cesaris P, Starace D, Starace G, Filippini A, Stefanini M, Ziparo E. Activation of Jun N-terminal kinase/stress-activated protein kinase pathway by tumor necrosis factor alpha leads to intercellular adhesion molecule-1 expression. J Biol Chem. 1999;274:28978–82. doi: 10.1074/jbc.274.41.28978. [DOI] [PubMed] [Google Scholar]
- 62.Shepherd EG, Zhao Q, Welty SE, Hansen TN, Smith CV, Liu Y. The function of mitogen-activated protein kinase phosphatase-1 in peptidoglycan-stimulated macrophages. J Biol Chem. 2004;279:54023–31. doi: 10.1074/jbc.M408444200. [DOI] [PubMed] [Google Scholar]
- 63.Pham CG, Papa S, Bubici C, Zazzeroni F, Franzoso G. Oxygen JNKies: phosphatases overdose on ROS. Dev Cell. 2005;8:452–4. doi: 10.1016/j.devcel.2005.03.005. [DOI] [PubMed] [Google Scholar]
- 64.Dounay AB, Forsyth CJ. Okadaic acid: the archetypal serine/threonine protein phosphatase inhibitor. Curr Med Chem. 2002;9:1939–80. doi: 10.2174/0929867023368791. [DOI] [PubMed] [Google Scholar]
- 65.Gilbert LA, Ravindran S, Turetzky JM, Boothroyd JC, Bradley PJ. Toxoplasma gondii targets a protein phosphatase 2C to the nuclei of infected host cells. Eukaryot Cell. 2007;6:73–83. doi: 10.1128/EC.00309-06. [DOI] [PMC free article] [PubMed] [Google Scholar]