Abstract
The stabilization of suspended planar lipid membranes, or black lipid membranes (BLMs), through polymerization of mono- and bis-functionalized dienoyl lipids was investigated. Electrical properties, including capacitance, conductance, and dielectric breakdown voltage, were determined for BLMs composed of mono-DenPC, bis-DenPC, mono-SorbPC, and bis-SorbPC both prior to and following photopolymerization, with diphytanoyl phosphocholine (DPhPC) serving as a control. Poly(lipid) BLMs exhibited significantly longer lifetimes and increased the stability to air-water transfers. BLM stability followed the order: bis-DenPC > mono-DenPC ≈ mono-SorbPC > bis-SorbPC. The conductance of bis-SorbPC BLMs was significantly higher than that of the other lipids, which is attributed to a high density of hydrophilic pores, resulting in relatively unstable membranes. The use of poly(lipid) BLMs as matrices for supporting the activity of an ion channel protein (IC) was explored using α – hemolysin (α-HL), a model IC. Characteristic i-V plots of α-HL were maintained following photopolymerization of bis-DenPC, mono-DenPC, and mono-SorbPC, demonstrating the utility of these materials for preparing more durable BLMs for single channel recordings of reconstituted ICs.
INTRODUCTION
Biological membranes are a complex mixture of lipids, proteins, and other molecules that serve as a semi-permeable barrier between two aqueous compartments.1, 2 Artificial membranes are frequently used as simplified models to systematically investigate the interplay between the phospholipid bilayer and other membrane components.3, 4 The suspended lipid bilayer, also known as a black lipid membrane (BLM), is one type of artificial membrane that has has been utilized for decades to characterize the activity of reconstituted ion channel proteins (ICs).5–8 The BLM geometry allows introduction of activators and inhibitors to both sides of the membrane and can be characterized using both electrochemical and spectroscopic methods.
A disadvantage of conventional BLMs composed of fluid phase lipids is that they are associated via relatively weak intermolecular interactions and thus are easily ruptured by exposure to mechanical, thermal, and/or chemical disruptions.9 Methods for stabilizing BLMs include aperture miniaturization,10, 11 gel support/encapsulation,12–15 droplet interface bilayers,16–19 and polymerization.20–24 Glass nanopore electrodes with apertures ranging from 100–400 nm have been used to reconstitute Staph. aurerus α-hemolysin (α –HL) with enhanced lifetimes due to the small membrane surface area.10 Steinem and co-workers used nanoporous alumina substrates as a support for suspended lipid bilayers containing reconstituted ICs.11 Gels have been used as BLM supports while still allowing for the access of solution to the membrane and measurement of electrical properties through the porous material.12–14 Development of agarose-encapsulated bilayers with single protein pores for stochastic sensing lengthened BLM lifetimes to upwards of 3 weeks.15 Schmidt and colleagues conjugated BLMs to substrate-anchored hydrogels to maintain GΩ resistance for up to 12 days.25–28 The droplet interface bilayer is a novel alternative to conventional BLMs.16–19, 29 It is formed when two aqueous droplets are suspended in a hydrocarbon in which either liposomes are present in the droplets or lipids are dissolved in the organic phase. A lipid bilayer forms at the interface between two droplets, and the activity of ICs reconstituted into the bilayer can be maintained for several days.
Polymerizable lipids are used to generate covalent networks within the bilayer to increase its stability, and have been used to form BLMs while maintaining the function of incorporated ICs.20–24 Benz and coworkers used dienoyl-containing lipids to study ionic transport in photopolymerized membranes and were successful in lengthening the lifetime from 1 hour before polymerization to between 4 and 5 hours post-polymerization.22 α-HL and gramicidin have been incorporated into partially polymerized membranes BLMs composed of diacteylenefunctionalized lipids.20, 21 Schmidt et al. and Meier and coworkers used triblock copolymers to form free standing membranes that support the function of ICs, including OmpF, maltoporin, α-HL, and alamethicin.30–32
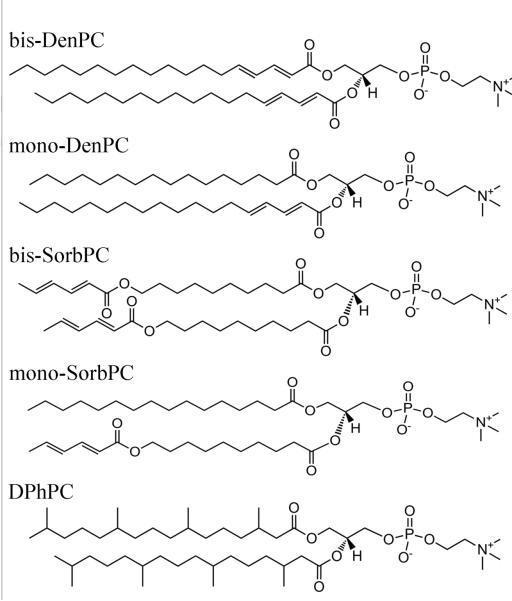
Recently we prepared photopolymerized BLMs using a bis-dienoyl lipid (bis-DenPC) that remained intact, as indicated by maintenance of a high resistance, in excess of 3 weeks.24 α-HL inserted into the membrane before polymerization exhibited functional IC activity for up to 7 days. In this paper, we have expanded that study to three other polymerizable lipids, mono-DenPC, mono-SorbPC, and bis-SorbPC (Fig. 1) to assess the influence of the location and number of dienoyl groups on BLM formation and properties. The electrical properties of BLMs, including capacitance, conductance, and dielectric breakdown, are characterized, as well as BLM stability and maintenance of α-HL activity. The results show that lipid cross-linking near the headgroup provides the best combination of BLM resistance, stability, and maintenance of α-HL activity.
Figure 1.
Structures of bis-DenPC, mono-DenPC, bis-SorbPC and mono-SorbPC, and DPhPC.
MATERIALS AND METHODS
Pipet Fabrication and Modification
Pipet fabrication and modification were described previously.23, 24 Briefly, borosilicate capillaries were pulled to a sharp tip using a micropipet puller, then broken to an opening of ca. 200 μm and fire polished using a microforge to obtain an aperture with an internal diameter at the tip of ca. 20 μm. Pipets were rinsed with 0.1 M HNO3 for 30 minutes followed by H2O and acetonitrile. Pipets were then filled with 2% (v/v) 3-cyanopropyldimethylchlorosilane in acetonitrile and stored for 12 hours, then rinsed with acetonitrile, ethanol, and water to remove excess silane reagent, then air dried for storage until use.10
BLM Formation and Polymerization
1,2-Diphytanoyl-sn-glycero-3-phosphocholine (DPhPC) in chloroform was purchased from Avanti Polar Lipids. Bis-dienoylphosphatidylcholine18,18 (bis-DenPC), bissorbylphosphatidylcholine17,17 (bis-SorbPC), mono-dienoylphophatidylcholine18,16 (mono-DenPC), and mono-sorbylphosphatidylcholine17,16 (mono-SorbPC)33 were synthesized as previously described by Dorn et al.,34 Lamparski et al.,35, 36 Liu,37 and Bondurant.38 Lipids were dried under Ar and residual solvents were removed under vacuum for a minimum 4 hours. Dried lipids were dispersed in n-decane to a final concentration of ~10 mg/ml. Prior to polymerization, all work with dienoyl lipids was performed under yellow light to minimize autopolymerization.
Formation and analysis of BLMs were performed at 23 ± 1 °C. A BLM was formed by applying a small volume (ca. 3 μL) of lipid solution to the tip of the pipet. After allowing the solvent to evaporate, the pipet was back-filled with recording buffer, the tip was recoated with lipid solution, and placed in a bath containing recording buffer (1 M KCl, 5 mM HEPES, pH 7.5). A disposable pipet tip was dragged across the glass pipet tip while monitoring the resistance using patch clamp amplifier, until a resistance of 10–50 GΩ was observed (only 0.1 GΩ for bis-SorbPC), signifying BLM formation. If no increase in resistance was observed, the tip dragging process was repeated until an increase was observed. BLM formation was further verified by applying a 1 V potential across the pipet and observing rupture of the BLM as indicated by a return (decrease) to open pipet resistance. The BLM was then easily reformed by dragging a disposable pipet across the glass pipet. Additional criteria that were used to verify BLM formation are given in Supporting Information (SI). Pipettes were used for multiple experiments by cleaning the pipet to remove residual lipid from the previous trial.
Polymerization of BLMs was performed by irradiation with a UV pen lamp (UVP, Upland, CA, Model 90-0012-01) placed 5 centimeters from the pipet tip for 15 minutes. This duration was determined to be sufficient to convert >95% of monomer to polymer (see below and SI).23, 24
Electrical Properties of BLMs
Electrical properties of BLMs were measured using a HEKA Electronik EPC-8 patch clamp amplifier configured with an ITC-16 A/D converter controlled with HEKA Pulse (v.8.53) data acquisition software. Data recordings were processed using the HEKA PulseFit (v. 8.53) analysis software.
For the characterization of BLM electrical properties, a single pipet with a ca. 20 μm aperture was used to minimize variations in the area of the bilayer and the adjacent annulus, providing for more reproducible measurements. Conductance values of BLMs suspended on this pipet were determined by applying sequential pulses, 50 ms in length, from −100 to +100 mV at 10 mV intervals. For 10 ms before and after each pulse, the potential was returned to a holding potential of 0 mV. The current output was averaged from 30 to 50 ms following the start of pulse and plotted versus applied potential.
The slope of the i-V plot is the conductance (GBLM) of the BLM which is inversely related to the BLM resistance, RBLM, by Equation 1.
| (1) |
All conductance measurements were corrected for the specific area of the pipet39 and are reported in units of S/cm2. A minimum of 20 BLMs was analyzed for each specific conductance value reported below.
Capacitance measurements were performed by applying a 50 ms square pulse of 100 mV to the BLM while recording the current response. The capacitive decay upon charging and release of the applied potential was then analyzed via one of two methods:8, 40, 41
1. The area under the capacitive peak was integrated and corrected for voltage applied to obtain capacitance, using Equation 2
| (2) |
where CBLM is the capacitance of the BLM, V is applied potential, and Q is the integrated charge from application of the potential (at time t = 0) until reaching a steady state current (t = x).
2. The RC time constant, τ, of the exponential decay was used to calculate capacitance using the measured resistance of BLM.
| (3) |
Both methods utilized computer programs written in-house to analyze the data. A minimum of 20 BLMs was analyzed for each specific capacitance value reported below.
Capacitance data were used to estimate the effective hydrocarbon thickness of membranes from Equation 4
| (4) |
where CBLM is the capacitance of the membrane, ε0 is the permittivity of free space (8.854 × 10−12 F m−1), ε is the dielectric constant of the material, A is the area of the membrane,39 and d is the thickness.
Dielectric breakdown voltages were measured in a similar manner as conductance. Voltage steps were applied to the BLM at 10 mV increments while monitoring current until rupture of the membrane occurred as indicated by a large increase in current. The maximum voltage accessible with the instrumentation used in these experiments is ±1 V.
Long-Term and Air-Water Stability of BLMs
Long-term stability of unpolymerized and polymerized BLMs was investigated by monitoring conductance and capacitance changes as a function of time with the pipet immersed in recording buffer at room temperature. Measurements were made on a daily basis until BLM rupture was observed. For BLMs having relatively short lifetimes (less than 5 hrs), the electrical properties were monitored more frequently. Air-water stability of BLMs was tested by repeatedly passing the pipet across the air-water interface. Each transfer corresponds to two crossings of the air-water interface (emersion from and immersion into recording buffer). Conductance and capacitance was measured after each transfer until BLM rupture was observed. Long-term and air-water stability studies were performed using several pipettes having an aperture diameter of ca. 20 μm.
α-Hemolysin Experiments
α-HL experiments were performed as previously described.24 Briefly, aliquots of α-HL (Sigma Aldrich) were added to the recording buffer in contact with the exterior of the BLM to a final concentration of 0.50 μg/mL. After formation of an α-HL pore in the BLM, which was measured as a step change in current, the buffer was rapidly exchanged to remove excess α-HL monomer, thus preventing insertion of another channel. In experiments involving polymerized membranes, the BLM was formed, protein added and allowed to insert, followed by buffer exchange and UV irradiation to polymerize the lipids. i-V curves were acquired before and after polymerization to compare the electrical properties of α-HL in unpolymerized and polymerized membranes. Recordings of IC activity were acquired at −40 mV with recording buffer on both sides of the BLM. Background currents were not subtracted because they were very low (ca. 1–3 pA). The conductance of BLMs functionalized with α-HL was determined from the slope of i-V plots measured over the range of −100 mV to 0 mV. Recordings were acquired using multiple pipettes with aperture diameters of ca. 20 μm.
RESULTS AND DISCUSSION
Electrical Characterization of Unpolymerized BLMs
DPhPC was used as a reference lipid to verify the validity of the characterization methods employed here. When DPhPC BLMs were analyzed, the following electrical properties were obtained: specific conductance of 8.5 × 10−2 pS/μm2, specific capacitance of 0.54 μF/cm2, dielectric thickness of 4.9 nm, and dielectric breakdown voltage of 1.6 × 106 V/cm. These values agree well with previously reported values.30, 42–44
The polymerizable lipids investigated in this study are bis-SorbPC, bis-DenPC, mono-SorbPC, and mono-DenPC (Figure 1). A considerable body of work has described use of these lipids to form highly stable lipid vesicles and planar supported lipid bilayers (PSLBs). e.g. 45–54 In contrast, bis-DenPC is the only one of this group that has been used to form BLMs.24 The main phase transition temperature, Tm, has been measured for vesicles composed of these lipids (data listed in Table 1). Based on these data, at the measurement temperature used throughout this work (23 ± 1°C), bis-SorbPC, mono-SorbPC, and mono-DenPC should be in the gel phase while bis-DenPC should be in the fluid phase. However, the BLMs studied here contain decane and some fraction of the lipids are adsorbed to the walls of the pipet aperture. The Tm value of a BLM may therefore differ from that of a vesicle composed of the same lipid.
Table 1.
Specific conductance values before (Gunpoly) and after UV irradiation (Gpoly) of dienoyl lipids and DPhPC.
Here BLMs were formed successfully on silanized glass pipets with all four polymerizable lipids, according to the criteria described above and in SI. i-V curves measured on unpolymerized BLMs are presented in SI. The specific conductance values, listed in Table 1, were in most cases comparable to that of DPhPC. The exception is bis-SorbPC; its conductance, 1300 pS/μm2, is at least 100-fold greater than any other lipid tested. Previous studies have shown that unpolymerized bis-SorbPC vesicles are highly permeable to ions and small molecules.51, 55 The passive transport of cations across a protein-free lipid bilayer is thought to occur through transient, water-filled pores that rapidly form and re-seal.56 Due to the sorbyl groups at the distal ends of the lipid tails, the interior of a bis-SorbPC bilayer is more hydrophilic than a bilayer in which the interior is composed of pure hydrocarbon chains. This difference likely lowers the energetic barrier for formation of transient pores through the bilayer, producing a more permeable membrane. In mono-SorbPC, a palmitoyl chain replaces one of the sorbyl-functionalized tails in bis-SorbPC and this greatly reduces the specific conductance of mono-SorbPC BLMs (to 7.6 pS/μm2, comparable to that of DPhPC). Thus the presence of only one distal sorbyl group does not significantly affect membrane permeability. The specific conductance values of BLMs composed of bis-DenPC and mono-DenPC were also comparable to that of DPhPC (Table 1). Like bis-SorbPC, bis-DenPC contains two polymerizable moieties, but they are adjacent to the glycerol backbone rather than in the center of the bilayer. The difference in location clearly has a significant effect on the ionic permeability of the membrane.
Capacitance values were determined using both methods described above and produced statistically similar results (only those obtained using the RC time constant method will be reported here). The specific capacitances of the polymerizable lipids are listed in Table 2 and ranged from 0.5 to 0.8 μF/cm2, comparable to that measured for DPhPC. The exception is bis-SorbPC which forms extremely conductive BLMs that do not allow for significant accumulation of charge at the interface, which prevented measurement of capacitance values.
Table 2.
Capacitance, thickness, and dielectric breakdown voltages of non-irradiated and UV irradiated BLMs composed of dienoyl lipids and DPhPC.
| Non-Irradiated | UV Irradiateda,b | ||||
|---|---|---|---|---|---|
| Lipid Name | CBLM (μF cm−2)c | dBLM (nm)c | VBR (× 105 V/cm)c | CBLM (μF cm −2)c | dBLM, (nm)c |
| DPhPC | 0.54 ± 0.01 | 3.4 ± 0.1 | 23 ± 1 | 0.52 ± 0.03 | 3.5 ± 0.3 |
| bis-DenPC | 0.71 ± 0.04 | 2.6 ± 0.2 | 31 ± 3 | 0.66 ± 0.03 | 2.8 ± 0.2 |
| bis-SorbPC | N/A | N/A | N/A | N/A | N/A |
| mono-DenPC | 0.69 ± 0.03 | 2.7 ± 0.2 | 26 ± 2 | 0.60 ± 0.02 | 3.1 ± 0.2 |
| mono-SorbPC | 0.59 ± 0.03 | 3.2 ± 0.3 | 23 ± 2 | 0.52 ± 0.05 | 3.6 ± 0.4 |
Dielectric breakdown voltages were not determined for polymerized BLMs composed of dienoyl lipids because the maximum voltage range of the instrument is ±1 V.
Dielectric breakdown voltages for UV irradiated DPhPC BLMs were comparable to non-irradiated BLMs (22 ± 2 × 105 V/cm).
Errors are ± one standard deviation.
Dielectric breakdown voltages in the range of 500 – 800 mV were measured for all BLM compositions tested except for bis-SorbPC. Using Equation 4 and an estimate of 2.1 for the dielectric constant of lipid,8 the thickness of the hydrophobic region of each type BLM was calculated, from which dielectric breakdown voltages expressed in V/cm were calculated (Table 2). The values for mono-SorbPC, mono-DenPC, and bis-DenPC ranged from 23 × 105 V/cm to 31 × 105 V/cm, comparable to that of DPhPC and consistent with published values.10 Rupture of bis-SorbPC BLMs occurred at lower applied voltages, typically 300 – 500 mV, than all other lipids tested, but this result cannot be expressed in V/cm due to the absence of capacitance and thickness data. Applying a strong electric field (or mechanical stress) to a lipid membrane is thought to increase the frequency that meta-stable, hydrophilic pores are formed, and promote expansion of these pores, leading to membrane rupture.56–59 When the radius of a pore is smaller than a critical value, the pore can re-seal, whereas when the pore size is larger than the critical radius, irreversible expansion occurs, eventually causing rupture. As discussed above, the unusually high conductance of bis-SorbPC BLMs suggests that these membranes contain larger and/or more numerous pores, and these likely provide a greater density of initiation sites for irreversible pore expansion.
Electrical Characterization of Polymerized BLMs
Polymerization of dienoyl lipids can be performed using a variety of methods including UV irradiation, redox radical initiation, and thermal free-radical initiation.60, 61 Bis-substituted lipids form cross-linked polymers whereas the mono-substituted lipids form linear polymers. UV polymerization produces a lower degree of polymerization than redox- and thermally-initiated polymerization.37, 45, 46, 52
The present study is focused exclusively on UV-initiated polymerization, which was performed by irradiation of BLMs for 15 min. During this time period, >95% of the lipid monomers were reacted based on measuring changes in the absorbance of vesicles illuminated under conditions equivalent to those used for irradiation of BLMs (see SI). Vesicles were used for these experiments because there is insufficient material in a BLM to measure the extent of monomer to polymer conversion by absorbance spectroscopy. However, it cannot be assumed that the polymerization kinetics of the two membrane geometries are equivalent, especially when considering that the BLMs contain decane whereas vesicles do not. To address this uncertainty, the capacitance of BLMs was monitored during irradiation. Figure S3 in SI shows that the measured changes were complete within 10 minutes, which provides additional evidence that the 15 min irradiation period was sufficient to effect near-quantitative polymerization.
i-V curves measured on UV irradiated BLMs are presented in SI. Specific conductance data are listed in Table 1 along with the percent change in conductance relative to that measured before irradiation. There was no change in the conductance of DPhPC BLMs, which served as a control. Polymerization of mono-SorbPC, mono-DenPC, and bis-DenPC produced decreases in specific conductance ranging from minor (−9% for bis-DenPC) to significant (−60% and −71% for mono-SorbPC and mono-DenPC, respectively). These decreases are not unexpected: Lipid polymerization is expected to reduce the surface area per molecule62 which should produce a more tightly packed, less permeable membrane; e.g., the permeability of poly(bis-DenPC) vesicles to glucose was ~50% relative to unpolymerized vesicles.34
The behavior of bis-SorbPC was considerably different - the specific conductance increased nearly 200% after polymerization. The cause of this increase is not clear but may be related to an attribute of bis-SorbPC that distinguishes it from the other three dienoyl lipids examined here: Cross-linking polymerization can occur both within and between leaflets at the center of the bilayer.47, 61 Although this creates a bilayer that is highly resistant to delamination (see below), it is may also stabilize the hydrophilic pores that are already quite numerous before polymerization. Furthermore, polymerization-induced phase segregation63–65 may promote aggregation of pores, creating larger defects. This is a subject that merits further investigation.
Small decreases in specific capacitance were observed upon polymerization of mono-SorbPC, mono-DenPC, and bis-DenPC BLMs (−8% to −12%; listed in Table 2), and these were statistically indistinguishable from the −4% decline observed for DPhPC. These decreases can be interpreted as small increases in bilayer thickness, also listed in Table 2. An increase can be rationalized because polymerization should restrict rotational motion and formation of gauche defects, producing a more rigid, elongated structure. In contrast, polymerization of diacetylenefunctionalized lipid bilayers causes the lipid chains to tilt, which decreases the bilayer thickness and significantly increases the capacitance (up to 40%, dependent on extent of irradiation).20, 21 To maintain the activity of reconstituted ICs, a minimal (or no) change in membrane thickness upon polymerization is needed to minimize hydrophobic mismatch between the protein and the hydrophobic core of the bilayer.
Dielectric breakdown voltages of BLMs were not obtainable after polymerization because the instrumentation used to make the measurements is limited to a maximum applied voltage of ±1 V. Within this range, rupture of polymerized BLMs was not observed. Membrane polymerization clearly provides enhanced stability because prior to UV irradiation, breakdown was always observed within the range of ±1V, regardless of lipid composition.
Stability of Poly(Lipid) BLMs
In addition to measuring dielectric breakdown voltages, the stability of unpolymerized and photopolymerized BLMs was evaluated as the amount of time that a pipet with an intact BLM could be stored in buffer before rupture occurred. Rupture was detected by measuring step changes in specific conductance and capacitance, as previously described.23, 24 The results are listed in Table 3, and a representative set of specific conductance and capacitance data for each lipid is presented in SI. Unpolymerized BLMs composed of dienoyl lipids had lifetimes of a few hundred minutes, similar to DPhPC, except bis-SorbPC for which very short lifetimes were measured. Upon UV irradiation, DPhPC BLMs showed no change in stability whereas all dienoyl lipids had increased lifetimes. Moderate lifetimes, up to 3 days, were observed for polymerized bis-SorbPC and the mono-substituted lipids, whereas for poly(bis-DenPC), the mean lifetime was 24 days.
Table 3.
Lifetimes of BLMs when stored in buffera.
| Lipid | Lifetime (min) (Non-Irradiated) | Lifetime (Days) (UV Irradiated) | Ratio of Irradiated to Non-Irradiated Lifetime |
|---|---|---|---|
| DPhPC (n = 5) | 300 (±70)b | 0.17 (±0.02)b | 0 8 |
| bis-DenPC (n = 5) | 240 (±29)b | 24 (±4)b | 144 |
| bis-SorbPC (n = 5) | 18 (±35) | 2 (±0) | 160 |
| mono-DenPC (n = 5) | 220 (±25) | 2.8 (±0.4) | 18 |
| mono-SorbPC (n = 5) | 224 (±26) | 2.8 (±0.4) | 18 |
Stability was also assessed by repetitively transferring a BLM/pipet across the air-water interface and measuring conductance and capacitance after each transfer, as previously described.23, 24 BLM rupture was observed as return of the conductance and capacitance to open pipet values. For unpolymerized BLMs, typically only three transfers were performed before rupture occurred for all lipids tested (see Table 4). UV irradiation had no effect on DPhPC and resulted in varying degrees of enhanced stability for dienoyl lipids. Manually transferring a BLM/pipet repeatedly across the air-water interface is an inherently less reproducible process than simply storing it in buffer. Regardless, the results of the two stability tests are consistent, which validates air-water transfer as a method of assessing stability.
Table 4.
Number of air-water transfers prior to rupture of BLMs.a
Both tests show that poly(bis-DenPC) generates the most stable BLMs, which is attributable to a combination of cross-linking in both lipid leaflets without introducing additional membrane porosity (i.e., conductance did not change). Poly(mono-DenPC) and poly(mono-SorbPC) BLMs are less stable than poly(bis-DenPC) BLMs, which is consistent with studies showing that linearly polymerized lipid bilayers are generally less stable than cross-linked lipid bilayers.37, 47, 48 Cross-linking polymerization of bis-SorbPC also provides a considerable stability enhancement, extending the mean lifetime to 2 days from 18 min, a ratio of 160 which is comparable to the ratio of 144 measured for bis-DenPC (see Table 3, column 3). However, with respect to absolute longevity, poly(bis-DenPC) is clearly superior. This is not surprising given that poly(bis-SorbPC) is the most porous of the membranes tested and, as discussed above, porosity in a BLM leads to rupture.
These results appear to contradict previous studies showing that polymerization of bis-SorbPC produces highly stable and uniform planar supported lipid bilayers (PSLBs).47, 61 The major stability test used in those studies was withdrawal from water into air, which frequently causes delamination of the two leaflets in a PSLB (i.e., the lower leaflet remains adsorbed to the substrate). Interleaflet cross-linking of bis-SorbPC produces a PSLB that is highly stable against delamination. However, as discussed above, the failure mode of a BLM is not delamination, it is rupture caused by irreversible pore expansion.
α-HL in Poly(Lipid) BLMs
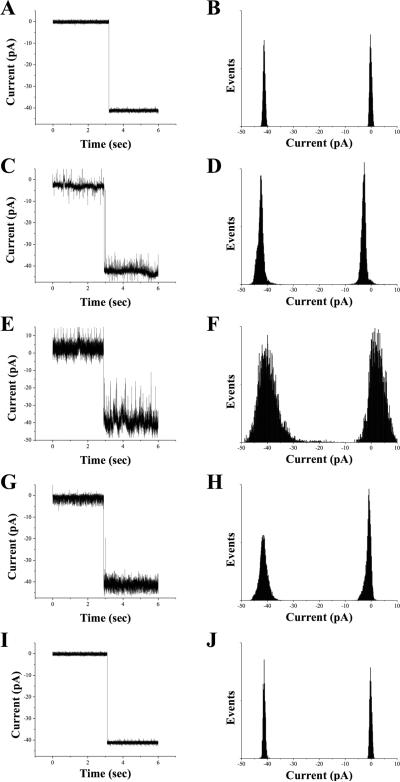
BLMs composed of dienoyl lipids were evaluated for their capability to support reconstitution of functional α-HL channels and maintenance of channel conductance after photopolymerization. α-HL insertion and IC activity was observed in all four dienoyl lipids as demonstrated by the representative recordings and all-points histograms shown in Figure 2. Table 5 lists the mean change in current measured upon insertion of single α-HL channels into BLMs. There was no statistical difference among the five lipids tested, showing that the structural differences among these lipids had no effect on ion flux through reconstituted α-HL channels.
Figure 2.
Representative current recordings showing insertion of single α-HL channels into (A) bis-DenPC, (C) bis-SorbPC, (E) mono-DenPC, (G) mono-SorbPC and (I) DPhPC BLMs, with respective all-points histograms in (B), (D), (F), (H), and (J).
Table 5.
| Lipid | Mean Current Before Insertion (pA) | Mean Open Current After Insertion (pA) | Difference Current (pA) |
|---|---|---|---|
| DPhPC (n = 5) | −0.2 ± 0.4 | −41.2 ± 0.4 | −41.0 ± 0.6 |
| bis-DenPC (n = 5) | −0.2 ± 0.4 | −41.2 ± 0.4 | −41.0 ± 0.6 |
| bis-SorbPC (n = 5) | −2± 1 | −42 ± 1 | −40 ± 1 |
| mono-DenPC (n = 5) | 3 ± 3 | −39 ± 4 | −42 ± 5 |
| mono-SorbPC (n = 5) | −2 ± 1 | −41 ± 2 | −39 ± 2 |
Five trials were conducted for each lipid composition. In each trial, the mean current was measured for one minute before insertion and for one minute after insertion.
BLMs were not irradiated. Note that α-HL channels do not insert into BLMs composed of polymerizable lipids after they have been irradiated.
In contrast, Figure 2 shows that in α-HL channel recordings, the background fluctuations in current varied significantly among the five lipids. From “noisiest” to least “noisy” the trend was: mono-DenPC > mono-SorbPC ≈ bisSorbPC > bis-DenPC ≈ DPhPC. Although the underlying cause(s) is not clear, this series appears to be correlated with the lipid phase, with the gel-phase lipids producing the highest noise levels (although as noted above, the Tm values listed in Table 1 may be different in BLMs). Additional experiments are needed to assess the effects of lipid structure and phase on the noise level in single channel recordings.
Although the α-HL concentration in the recording buffer was a constant in these experiments, the time required to observe formation of a functional α-HL channel varied significantly among the lipids tested. Insertion into a DPhPC BLM typically occurred between 5 and 10 minutes after addition of α-HL. For bis-DenPC, bis-SorbPC, mono-DenPC, and mono-SorbPC, the respective times were 20–30 minutes, ca. 30 minutes, 30–60 minutes, and 1–2 hours. Thus the insertion times increased with the Tm of the lipid, which is not unexpected. Adsorption, partitioning, and lateral diffusion of α-HL monomers in a lipid bilayer is thought to precede formation of a functional heptameric channel,66 and this process should be slower when the bilayer is in the gel phase. α-HL activity in bis-SorbPC BLMs was particularly difficult to achieve and maintain. Only about 10% of the insertions were long-lived enough to allow a thorough characterization of the BLM electrical properties before and after lipid polymerization. This low success rate is a consequence of the inherent instability of bis-SorbPC BLMs: Insertion typically required about 30 minutes whereas the mean BLM lifetime was only 18 minutes (Table 3). When insertion was successful, BLM rupture usually occurred within minutes thereafter (see SI for an example recording of insertion followed by failure).
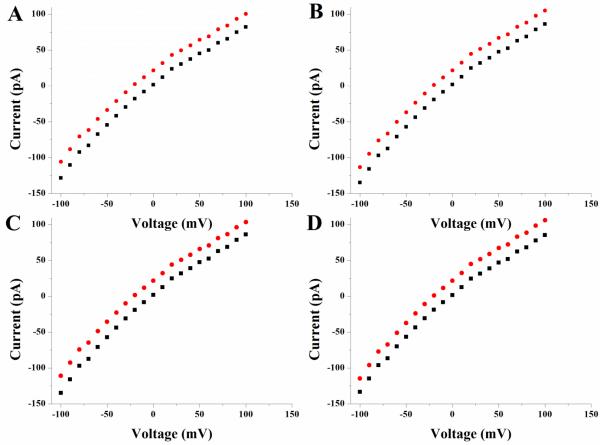
For each lipid, i-V curves were measured on BLMs that contained a single α-HL channel. BLMs were then irradiated for 15 minutes with UV light, then i-V curves were measured again (excepting bis-SorbPC, for which measurements were not feasible based on the reasons stated above). Figure 3 shows representative i-V curves obtained before and after UV irradiation of BLMs composed of DPhPC (A), bis-DenPC (B), mono-DenPC (C), and mono-SorbPC (D). The slight rectification characteristic of α-HL67 was observed both before and after UV irradiation for all four lipids tested. Conductance data obtained from 12 trials on each lipid are listed in Table 6. Insertions of α-HL resulted in reproducible conductance states of ~1 nS and this was not altered by lipid polymerization. Noise levels of recordings following polymerization also were not changed. i-V curves were measured daily on polymerized BLMs containing single α-HL channels. The characteristic channel conductance (~1 nS) was maintained in mono-DenPC and mono-SorbPC BLMs for about two days, and in bis-DenPC BLMs for about seven days (Table 6). Overall, the data listed in Tables 5 and 6 show that functional α-HL channels can be reconstituted into BLMs composed of dienoyl lipids and maintain IC activity after UV photopolymerization for up to one week.
Figure 3.
i-V plots of BLMs containing a single α-HL channel prior to (■) and following ( ) UV irradiation: (A) DPhPC, (B) bis-DenPC, (C) mono-DenPC, and (D) mono-SorbPC. UV irradiated i-V plots are offset by +20 pA to enable visual comparison.
) UV irradiation: (A) DPhPC, (B) bis-DenPC, (C) mono-DenPC, and (D) mono-SorbPC. UV irradiated i-V plots are offset by +20 pA to enable visual comparison.
Table 6.
Conductance values of BLMs containing single α-HL channels prior to and following UV irradiation.a
| Lipid | Gα-HL (nS)c | Gα-HL, +UV (nS)d | Gα-HL, +UV (nS) - Day 2b,e | Gα-HL, +UV (nS)- Day 7b,e |
|---|---|---|---|---|
| DPhPC | 1.04 ± 0.03 | 1.07 ± 0.05 | N/A | N/A |
| bis-DenPC | 1.05 ± 0.02 | 1.08 ± 0.03 | 1.1 ± 0.1 | 1.2 ± 0.2 |
| bis-SorbPC | 1.03 ± 0.08 | N/A | N/A | N/A |
| mono-DenPC | 1.08 ± 0.09 | 1.07 ± 0.06 | 1.0 ± 0.1 | N/A |
| mono-SorbPC | 1.07 ± 0.07 | 1.05 ± 0.09 | 1.1 ± 0.1 | N/A |
Gα-HL and Gα-HL, +UV are conductances values measured before and after UV irradiation, respectively. Values were background corrected for the respective conductances of the pure lipid BLMs. Errors are ± one standard deviation. N/A means that BLM rupture occurred by this stage of the experiment.
Conductance values measured 2 days and 7 days after BLM formation.
For each composition, n = 12, except bis-SorbPC for which n = 3.
For each composition, n = 12.
For each composition, n = 7.
For mono-DenPC and mono-SorbPC BLMs, the channel conductance was maintained until BLM rupture occurred. In other words, channel closing events were rarely observed under the analysis conditions employed here, consistent with previous studies.68, 69 The exception was bis-DenPC BLMs - the channel permanently closed after about one week. At this point, the conductance decreased to that of a pure poly(bis-DenPC) BLM (i.e., a BLM lacking a α-HL channel) for the subsequent two-week period before rupture occurred (compare Tables 3 and 6). The cause for the loss of α-HL conductance after one week is not known and will be the subject of further investigations.
SUMMARY AND CONCLUSIONS
The electrical properties of BLMs composed of several polymerizable dienoyl lipids were characterized, including capacitance, conductance, and dielectric breakdown voltages, prior to and following photopolymerization. BLM stability to long-term storage in buffer and repeated transfer across the air-water interface was also examined. Unpolymerized bis-SorbPC BLMs are highly conductive and rupture rapidly after formation, which is attributed to porosity caused by the presence of hydrophilic dienoyl moieties in the center of the bilayer. Cross-linking of bis-SorbPC increases porosity even further but also improves BLM stability, comparable to that observed for BLMs composed of the linearly polymerized lipids (mono-SorbPC, and mono-DenPC). BLMs composed of poly(bis-DenPC), which is cross-linked adjacent to the glycerol backbone, provide the best combination of low conductance and long-term stability. Insertion of single α-HL channels into unpolymerized BLMs was successful for all four dienoyl lipids. The conductance of single α-HL channels in BLMs was measured before and after lipid polymerization, and no significant perturbations were observed for three of the four lipids tested: bis-DenPC, mono-SorbPC, and mono-DenPC. Detailed characterization of α-HL channels in bis-SorbPC BLMs was not feasible because of their instability. Overall, this study shows that lipid cross-linking near the headgroup in a BLM maintains its high electrical resistance (comparable to that of DPhPC), provides greatly enhanced long-term stability to rupture (typically for three weeks), and maintains α-HL activity (typically for one week). These polymerized BLMs offer the potential for long-term electrophysiological studies of reconstituted ICs and development of IC-based biosensors, including arrays for high throughput screening of drug libraries.
Supplementary Material
ACKNOLWDGEMENTS
This material is based upon work supported by the National Institutes of Health under Grant No. EB007047 and the National Science Foundation under Grant No. CHE-0548167. Any opinions, findings, and conclusions or recommendations expressed in this material are those of the authors and do not necessarily reflect the views of the National Science Foundation or the National Institutes of Health.
Footnotes
SUPPORTING INFORMATION Additional criteria to assess BLM formation, absorbance and capacitance data measured for vesicles and BLMs, respectively, as a function of UV irradiation time, i-V plots of BLMs before and after UV irradiation, conductance and capacitance data obtained on polymerized BLMs as a function of storage time, and an example of insertion of an α-HL channel into a bis-SorbPC BLM followed by rupture. This information is available free of charge via the Internet at http://pubs.acs.org.
REFERENCES
- (1).Singer SJ, Nicolson GL. Science. 1972;175:720–731. doi: 10.1126/science.175.4023.720. [DOI] [PubMed] [Google Scholar]
- (2).Nagle JF, Tristram-Nagle S. Biochim. Biophys. Acta. 2000;1469:159–195. doi: 10.1016/s0304-4157(00)00016-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (3).Kunitake T. Angew. Chem., Int. Ed. 1992;31:709–726. [Google Scholar]
- (4).Tien HT, Ottova ALJ. Membr. Sci. 2001;189:83–117. [Google Scholar]
- (5).Mey I, Stephan M, Schmitt EK, Muller MM, Ben Amar M, Steinem C, Janshoff AJ. Am. Chem. Soc. 2009;131:7031–7039. doi: 10.1021/ja809165h. [DOI] [PubMed] [Google Scholar]
- (6).Mueller P, Rudin DO, Tien HT, Wescott WC. Nature. 1962;194:979–980. doi: 10.1038/194979a0. [DOI] [PubMed] [Google Scholar]
- (7).Mueller P, Wescott WC, Rudin DO, Tien HT. J. Phys. Chem. 1963;67:534–535. [Google Scholar]
- (8).Montal M, Mueller P. Proc. Natl. Acad. Sci. U.S.A. 1972;69:3561–3566. doi: 10.1073/pnas.69.12.3561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (9).Deamer DW, Akeson M. Trends Biotechnol. 2000;18:147–151. doi: 10.1016/s0167-7799(00)01426-8. [DOI] [PubMed] [Google Scholar]
- (10).White RJ, Ervin EN, Yang T, Chen X, Daniel S, Cremer PS, White HS. J. Am. Chem. Soc. 2007;129:11766–11775. doi: 10.1021/ja073174q. [DOI] [PubMed] [Google Scholar]
- (11).Hennesthal C, Drexler J, Steinem C. ChemPhysChem. 2002;3:885–889. doi: 10.1002/1439-7641(20021018)3:10<885::AID-CPHC885>3.0.CO;2-9. [DOI] [PubMed] [Google Scholar]
- (12).Maurer JA, White VE, Dougherty DA, Nadeau JL. Biosens. Bioelectron. 2007;22:2511–2584. doi: 10.1016/j.bios.2006.10.017. [DOI] [PubMed] [Google Scholar]
- (13).Ziegler W, Gaburjakova J, Gaburjakova M, Sivak B, Rehacek V, Tvarozek V, Hianik T. Colloids Surf., A. 1998;140:357–367. [Google Scholar]
- (14).Lu XD, Ottova AL, Tien HT. Bioelectrochem. Bioenerg. 1996;39:285–289. [Google Scholar]
- (15).Kang XF, Cheley S, Rice-Ficht AC, Bayley H. J. Am. Chem. Soc. 2007;129:4701–4705. doi: 10.1021/ja068654g. [DOI] [PubMed] [Google Scholar]
- (16).Bayley H, Cronin B, Heron A, Holden MA, Hwang WL, Syeda R, Thompson J, Wallace M. Mol. BioSyst. 2008;4:1191–1208. doi: 10.1039/b808893d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (17).Holden MA, Needham D, Bayley H. J. Am. Chem. Soc. 2007;129:8650–8655. doi: 10.1021/ja072292a. [DOI] [PubMed] [Google Scholar]
- (18).Hwang WL, Chen M, Cronin B, Holden MA, Bayley H. J. Am. Chem. Soc. 2008;130:5878–5879. doi: 10.1021/ja802089s. [DOI] [PubMed] [Google Scholar]
- (19).Hwang WL, Holden MA, White S, Bayley H. J. Am. Chem. Soc. 2007;129:11854–11864. doi: 10.1021/ja074071a. [DOI] [PubMed] [Google Scholar]
- (20).Daly SM, Heffernan LA, Barger WR, Shenoy DK. Langmuir. 2006;22:1215–1222. doi: 10.1021/la052327p. [DOI] [PubMed] [Google Scholar]
- (21).Shenoy DK, Barger WR, Singh A, Panchal RG, Misakian M, Stanford VM, Kasianowicz J. J. Nano Lett. 2005;5:1181–1185. doi: 10.1021/nl050481q. [DOI] [PubMed] [Google Scholar]
- (22).Benz R, Elbert R, Prass W, Ringsdorf H. Eur. Biophys. J. 1986;14:83–92. doi: 10.1007/BF00263066. [DOI] [PubMed] [Google Scholar]
- (23).Heitz BA, Jones IW, Hall HK, Aspinwall CA, Saavedra SS. J. Am. Chem. Soc. 2010;132:7086–7093. doi: 10.1021/ja100245d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (24).Heitz BA, Xu JH, Hall HK, Aspinwall CA, Saavedra SS. J. Am. Chem. Soc. 2009;131:6662–6663. doi: 10.1021/ja901442t. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (25).Jeon TJ, Malmstadt N, Schmidt JJ. J. Am. Chem. Soc. 2006;128:42–43. doi: 10.1021/ja056901v. [DOI] [PubMed] [Google Scholar]
- (26).Jeon TJ, Malmstadt N, Poulos J, Schmidt J. J. Biointerphases. 2007;2:652A–652A. doi: 10.1116/1.2948314. [DOI] [PubMed] [Google Scholar]
- (27).Jeon TJ, Malmstadt N, Poulos JL, Schmidt J. J. Biointerphases. 2008;3:FA96–FA100. doi: 10.1116/1.2948314. [DOI] [PubMed] [Google Scholar]
- (28).Malmstadt N, Jeon LJ, Schmidt J. J. Adv. Mater. 2008;20:84–89. [Google Scholar]
- (29).Syeda R, Holden MA, Hwang WL, Bayley H. J. Am. Chem. Soc. 2008;130:15543–15548. doi: 10.1021/ja804968g. [DOI] [PubMed] [Google Scholar]
- (30).Wong D, Jeon TJ, Schmidt J. Nanotechnol. 2006;17:3710–3717. [Google Scholar]
- (31).Nardin C, Winterhalter M, Meier W. Langmuir. 2000;16:7708–7712. [Google Scholar]
- (32).Meier W, Nardin C, Winterhalter M. Angew. Chem., Int. Ed. 2000;39:4599–4602. [PubMed] [Google Scholar]
- (33).Subscripts following lipid names denote length of acyl chains in the 1 and 2 positions, respectively.
- (34).Dorn K, Klingbiel RT, Specht DP, Tyminski PN, Ringsdorf H, O'Brien DF. J. Am. Chem. Soc. 1984;106:1627–1633. [Google Scholar]
- (35).Lamparski H. Ph.D. Dissertation. University of Arizona; Tucson, AZ: 1993. [Google Scholar]
- (36).Barry JA, Lamparski H, Shyamsunder E, Osterberg F, Cerne J, Brown MF, O'Brien DF. Biochemistry. 1992;31:10114–10120. doi: 10.1021/bi00156a035. [DOI] [PubMed] [Google Scholar]
- (37).Liu SC, Sisson TM, O'Brien DF. Macromolecules. 2001;34:465–473. [Google Scholar]
- (38).Bondurant B. Ph.D. Dissertation. University of Arizona; Tucson, AZ: 2000. [Google Scholar]
- (39).Pipet internal diameter at the tip was used as an estimate of membrane area.
- (40).Benz R, Janko K. Biochim. Biophys. Acta. 1976;455:721–738. doi: 10.1016/0005-2736(76)90043-2. [DOI] [PubMed] [Google Scholar]
- (41).Kramar P, Miklavcic D, Lebar AM. Bioelectrochemistry. 2007;70:23–27. doi: 10.1016/j.bioelechem.2006.03.022. [DOI] [PubMed] [Google Scholar]
- (42).Weiskopf D, Schmitt EK, Kluhr MH, Dertinger SK, Steinem C. Langmuir. 2007;23:9134–9139. doi: 10.1021/la701080u. [DOI] [PubMed] [Google Scholar]
- (43).Baba T, Toshima Y, Minamikawa H, Hato M, Suzuki K, Kamo N. Biochim. Biophys. Acta, Biomembr. 1999;1421:91–102. doi: 10.1016/s0005-2736(99)00114-5. [DOI] [PubMed] [Google Scholar]
- (44).Funakoshi K, Suzuki H, Takeuchi S. Anal. Chem. 2006;78:8169–8174. doi: 10.1021/ac0613479. [DOI] [PubMed] [Google Scholar]
- (45).Lamparski H, O'Brien DF. Macromolecules. 1995;28:1786–1794. [Google Scholar]
- (46).Liu SC, O'Brien DF. Macromolecules. 1999;32:5519–5524. [Google Scholar]
- (47).Ross EE, Rozanski LJ, Spratt T, Liu SC, O'Brien DF, Saavedra SS. Langmuir. 2003;19:1752–1765. [Google Scholar]
- (48).Sisson TM, Lamparski HG, Kolchens S, Elayadi A, O'Brien DF. Macromolecules. 1996;29:8321–8329. [Google Scholar]
- (49).Conboy JC, Liu SC, O'Brien DF, Saavedra SS. Biomacromolecules. 2003;4:841–849. doi: 10.1021/bm0256193. [DOI] [PubMed] [Google Scholar]
- (50).Ross EE, Joubert JR, Wysocki RJ, Jr., Nebesny K, Spratt T, O'Brien DF, Saavedra SS. Biomacromolecules. 2006;7:1393–1398. doi: 10.1021/bm050727l. [DOI] [PubMed] [Google Scholar]
- (51).Cheng ZL, D'Ambruoso GD, Aspinwall CA. Langmuir. 2006;22:9507–9511. doi: 10.1021/la061542i. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (52).Lei JT, Sisson TM, Lamparski HG, O'Brien DF. Macromolecules. 1999;32:73–78. [Google Scholar]
- (53).Ohno H, Ogata Y, Tsuchida E. Macromolecules. 1987;20:929–933. [Google Scholar]
- (54).Ohno H, Takeoka S, Iwai H, Tsuchida E. J. Polym. Sci., Part A: Polym. Chem. 1987;25:2737–2746. [Google Scholar]
- (55).Ratnayaka SN. Ph.D. Dissertation. University of Arizona; Tucson, AZ: 2008. [Google Scholar]
- (56).Leontiadou H, Mark AE, Marrink SJ. Biophys. J. 2004;86:2156–2164. doi: 10.1016/S0006-3495(04)74275-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (57).Evans E, Heinrich V, Ludwig F, Rawicz W. Biophys. J. 2003;85:2342–2350. doi: 10.1016/s0006-3495(03)74658-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (58).Melikov KC, Frolov VA, Shcherbakov A, Samsonov AV, Chizmadzhev YA, Chernomordik LV. Biophys. J. 2001;80:1829–1836. doi: 10.1016/S0006-3495(01)76153-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (59).Tieleman DP, Leontiadou H, Mark AE, Marrink SJ. J. Am. Chem. Soc. 2003;125:6382–6383. doi: 10.1021/ja029504i. [DOI] [PubMed] [Google Scholar]
- (60).O'Brien DF, Armitage B, Benedicto A, Bennett DEL,HG, Lee YS, Srisiri W, Sisson TM. Acc. Chem. Res. 1998;31:861–868. [Google Scholar]
- (61).Zhang H, Joubert JR, Saavedra SS. Adv. Polym. Sci. 2010;224:1–42. [Google Scholar]
- (62).Grainger DW, Reichert A, Ringsdorf H, Salesse C, Davies DE, Lloyd JB. Biochim. Biophys. Acta. 1990;1022:146–154. doi: 10.1016/0005-2736(90)90108-z. [DOI] [PubMed] [Google Scholar]
- (63).Ahl PL, Price R, Smuda J, Gaber BP, Singh A. Biochim. Biophys. Acta. 1990;1028:141–153. doi: 10.1016/0005-2736(90)90148-h. [DOI] [PubMed] [Google Scholar]
- (64).Gaub H, Buschl R, Ringsdorf H, Sackmann E. Chem. Phys. Lipids. 1985;37:19–43. [Google Scholar]
- (65).Mueller A, O'Brien DF. Chem. Rev. 2002;102:727–757. doi: 10.1021/cr000071g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (66).Kawate T, Gouaux E. Protein Sci. 2003;12:997–1006. doi: 10.1110/ps.0231203 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- (67).Gu LQ, Cheley S, Bayley H. J. Gen. Physiol. 2001;118:481–493. doi: 10.1085/jgp.118.5.481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (68).Braha O, Walker B, Cheley S, Kasianowicz JJ, Song LZ, Gouaux JE, Bayley H. Chem. Biol. 1997;4:497–505. doi: 10.1016/s1074-5521(97)90321-5. [DOI] [PubMed] [Google Scholar]
- (69).Kasianowicz JJ, Brandin E, Branton D, Deamer DW. Proc. Natl. Acad. Sci. U.S.A. 1996;93:13770–13773. doi: 10.1073/pnas.93.24.13770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (70).Lamparski H, Lee YS, Sells TD, O'Brien DF. J. Am. Chem. Soc. 1993;115:8096–8102. [Google Scholar]
- (71).Lindsey H, Petersen NO, Chan SI. Biochim. Biophys. Acta. 1979;555:147–167. doi: 10.1016/0005-2736(79)90079-8. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.