Abstract
Cataracts (opacities of the lens) are frequent in the elderly, but rare in paediatric practice. Congenital cataracts (in industrialized countries) are mainly caused by mutations affecting lens development. Much of our knowledge about the underlying mechanisms of cataractogenesis has come from the genetic analysis of affected families: there are contributions from genes coding for transcription factors (such as FoxE3, Maf, Pitx3) and structural proteins such as crystallins or connexins. In addition, there are contributions from enzymes affecting sugar pathways (particularly the galactose pathway) and from a quite unexpected area: axon guidance molecules like ephrins and their receptors. Cataractous mouse lenses can be identified easily by visual inspection, and a remarkable number of mutant lines have now been characterized. Generally, most of the mouse mutants show a similar phenotype to their human counterparts; however, there are some remarkable differences. It should be noted that many mutations affect genes that are expressed not only in the lens, but also in tissues and organs outside the eye. There is increasing evidence for pleiotropic effects of these genes, and increasing consideration that cataracts may act as early and readily detectable biomarkers for a number of systemic syndromes.
Keywords: paediatric, cataracts, human, mouse, genetics
1. Introduction
The definition of a cataract is ‘opacification of the normally transparent crystalline lens’. To be classified as congenital, it should be present at birth, whereas those appearing after birth but before 16 years of age can be termed juvenile or paediatric. Given the chronology, congenital cataracts must be caused by changes that occur during embryonic development, whereas juvenile cataracts may result from alterations in gene expression after birth. For more details on lens development, lens fibre differentiation and all other aspects of lens morphology and structure, please refer to earlier articles in this issue. In this article, we shall focus on advances in the clinical and experimental aspects of congenital and paediatric cataracts, including a detailed discussion of mutations in genes causing cataracts that are expressed both during and after embryonic development. A glossary of clinical terms can be found at the end of this article.
2. Clinical aspects of Congenital or Paediatric Cataract
(a). Consequences for vision
Bilateral congenital cataract is the most common cause of treatable childhood blindness. In the developing world, it accounts for between 12 (India) and 39 per cent (Jamaica) of the causes of blindness [1]. In the UK, the incidence of congenital cataract has been calculated at 2.49–3.46 per 10 000 [2], and accounted for 3 per cent of all new blindness or partial sight certifications among children in England and Wales in 1990 [3]. Screening is carried out in the maternity wards or by the general practitioner at the six to eight week check by looking for a red retinal reflex. If this is absent, then prompt referral to an ophthalmologist is essential as ocular pathology, such as a cataract, may be present. Opacification of the lens is not necessarily sight threatening especially if it occurs in the peripheral cortex. However, disruption to the passage of light through the central visual axis of the lens will certainly impact on vision, resulting in varying degrees of stimulus deprivation amblyopia. If nystagmus has become established, then good vision is rarely recoverable despite surgery [4]. The age of onset, position and size of the central defect are relevant. For instance, dense nuclear cataracts, which are usually present at birth and non-progressive, have a greater negative impact on vision than anterior polar cataracts. Posterior polar cataracts have a variable effect dependent on the size and density of the opacity, but have a better visual prognosis as they often appear later. Lamellar cataracts are invariably bilateral and slowly progressive, frequently requiring removal at a later age, such as prior to starting school. Timing of surgery is dictated not only by promptness of referral, visual responsiveness and clinical examination, but also by the risks and benefits of surgery. A latent period for visual development has been described in the first six weeks of life when vision is subcortically mediated. During this time, the risk of developing stimulus deprivation amblyopia is low [5–8]. However, operating on very young eyes has disadvantages too. It has been shown that eyes operated upon before one month of age carry the highest risk of secondary glaucoma. One study found that the 5 year risk of glaucoma in at least one eye was 50 per cent (95% CI 27.8–77.1) in the group operated on early compared with 14.9 per cent (95% CI 6.5–32.1) when surgery was performed later (log-rank test, p = 0.012) [9]. Given this evidence, most surgeons would agree that the optimal time for removing dense, bilateral nuclear cataracts is around six to eight weeks of age. Good visual results can be obtained only by lensectomy, prompt optical correction of aphakia and occlusion therapy.
In unilateral cases, the brain favours the contralateral eye and the amblyopia is harder to correct even after removal of the cataract and optical correction. The poor vision may result from more than one insult: initial visual deprivation, aniseikonia and anisometropic and strabismic amblyopia [10]. A high percentage of unilateral cataract eyes develop strabismus. Aggressive patching regimes have been described whereby the phakic eye is occluded for 1 h per day for each month of life until six months of age and then between 50 and 100 per cent of the waking day depending on the age and interocular difference in visual acuity [11]. More recently, however, objective measuring techniques have demonstrated that 3–6 h of occlusion in children over 4 years of age (and less for children under 4 years) is sufficient and achieves a similar visual outcome to 6–12 h of occlusion [12]. There is still controversy as to when to abandon patching, with some clinicians preferring to prescribe ‘part-time’ occlusion to maintain vision until a child reaches 7 years of age (by which time visual development is felt to be established). However, one study has shown that vision can still be maintained or even improve when patching is stopped prior to 6 years of age [10], and others adopt the view that a trial of occlusion is worth considering even if the child presents at 6 or 7 years of age. Several factors need to be taken into account, including the severity and complexity of the amblyopia: is it because of sensory deprivation alone or is it more complex containing elements of anisometropic and strabismic amblyopia [13]?
There are a few longer term studies looking at visual outcomes in infants following cataract surgery: the outcome of the treatment of unilateral cataracts is not as good as that for bilateral cataracts. Gouws et al. [14] reported on 18 infants undergoing surgery at less than 1 year of age with a mean follow-up of 95 months: best outcomes were in the bilateral group where 50 per cent of eyes achieved 6/18 or better, with a best acuity of 6/9. Acuities were worse in the unilateral group where only 38 per cent achieved 6/60 or better, with a best acuity of 6/24. Slightly better results were reported by Barry et al. [15] where almost 50 per cent of infants under 1 year of age achieved 6/12 or better best-corrected visual acuities.
(b). Clinical management
Congenital cataracts affect not only children but also their immediate family. It is unnecessary to do a large number of tests and investigations on all children with cataract. It is better to take a careful history (including illnesses or drugs used in pregnancy) and a family history, and determine whether the child is developing normally in all other areas. All blind children will experience some developmental delay, and this is usually reversed if vision is restored. However, speech and hearing development should be normal. It may be helpful to involve a paediatrician, who will examine for other congenital anomalies, and determine whether the child is fit for general anaesthesia [16].
Most clinicians agree that combining the findings of an ocular examination and best-corrected distance and near acuities is paramount in determining the need and timing of surgical intervention. For instance, a zonular cataract in a child may obscure the red reflex but the vision may still be remarkably good. Furthermore, as cataract surgery largely removes the ability of the eye to accommodate (change focus from looking at distance to near objects), one may wish to consider other factors: a very active child may function better retaining a partial cataract giving a visual acuity of 6/18 and the full ability to adjust accommodation rapidly rather than undergoing surgery that may give a better visual acuity of 6/9 but a very reduced depth of visual field; a young infant or child with mobility problems, however, may be happier with a small range of pseudo-accommodation following surgery (because of papillary miosis) but clearer vision.
Surgical aphakia can be corrected in one of three ways: intraocular lens (IOL) implantation, contact lens or spectacles, with the choice being individualized in each case. IOL implantation is generally the preferred option even in babies aged eight weeks, providing there are no other ocular contraindications, such as microphthalmia (pathologically small eye), short axial length, anterior or posterior segment anomalies. A recent UK survey regarding children undergoing cataract surgery less than or equal to 2 years of age found that 100 per cent of paediatric ophthalmologists used a hydrophobic acrylic IOL (90% used an Acrysof model) and 75 per cent used the SRK/T formula for calculating IOL power. The majority performed limbal incisions (sutured), manual anterior capsulorrhexis, lens aspiration, manual posterior capsulotomy and an anterior vitrectomy [17]. A Scandanavian study showed that posterior capsulotomy and anterior vitrectomy were not necessary in children older than 6 years [18].
Determining the power of the IOL can be complex. At birth, the human lens is more spherical than in adults, with a power of about 30 D. This decreases to about 20–22 D by the age of 5 years, with an accompanying reduction in the power of the cornea and axial lengthening of the globe. This means that an IOL that gives normal vision to an infant will lead to significant myopia when he or she is older. Most surgeons choose an IOL that leaves the infant hypermetropic (approx. +8 D at six weeks of age, +3 D at 12 months, +2 D at 2 years) to allow the eye to ‘grow into the IOL’, correcting the resulting ammetropia with spectacles [11,19]. The requirement of accommodation for near tasks should be considered for each age group. It is reasonable to prescribe glasses post-operatively to clarify the near vision initially in an infant, whereas an older child will require clarity for both distance and near vision with bifocals that incorporate a large reading segment. When it has not been possible to insert an IOL during the initial surgery then high-water-content aphakic contact lenses or aphakic spectacles (bifocals in school-age children) can be prescribed. In some children, a secondary IOL may be implanted when they are older or if they do not tolerate contact lens wear.
Regardless of the means of optical correction, amblyopia therapy should be started at the first follow-up and refraction should be checked every four months up to 2 years of age and annually thereafter. The use of post-operative topical antibiotics, once daily mydriatics and 1–2 hourly topical steroids (decreasing over two to three months) are essential to minimize the risk of infection and inflammation. There is a longer term risk of retinal detachment in eyes that have had surgery for congenital cataract, often occurring more than 30 years after the original surgery [20].
(c). Clinical advances and future speculations
It is interesting to speculate on the future, and the following ideas are some of the advances that may move this field forward into the next decade. Many have been tried in humans but some are still in the animal research stage of progression.
The last 10 years have seen surgeons operating and implanting IOLs in infants as young as a few weeks of age. This has been assisted by improvements in surgical technique and instrumentation. In particular, new-generation viscoelastics provide increased protection for the corneal endothelium and allow easier intra-operative manipulation of ocular tissues, most notably during capsulorhexis. Sharma et al. [21] demonstrated that Trypan blue staining of the posterior capsule can help in posterior capsulorhexis and optic capture. With increasing numbers of surgeons preserving a rim of the posterior lens capsule to support either a primary or secondary IOL, there is a corresponding need to reduce the speed and severity with which the posterior lens capsule opacifies. Post-operative posterior capsule opacification (PCO; owing to migration and proliferation of lens epithelial cells behind the IOL) is common, reduces vision and requires either subsequent surgical or laser disruption of the lens capsule, usually under anaesthesia some months or years later. There have been some advances in the ongoing challenge to tackle this. A recent Cochrane review [22] of 53 studies looking at interventions for preventing PCO found that sharp-edged IOLs gave a significantly lower PCO score and YAG (yttrium aluminium garnet) laser rate than round-edged IOLs. It is conceivable that IOLs will be designed that can deliver slow release molecules to reduce inflammation (dexamethasone-coated IOL reduced post-operative inflammation in rabbit eyes) [23] or PCO rate, or devices such as the Perfect Capsule [24], irrigated with antimitotic agents such as 5-fluorouracil, will be used to prevent PCO (shown to be successful in young rabbit eyes) [25].
Another major complication in these young eyes is the development of glaucoma. Advances in our ability to measure central corneal thickness (CCT) and intraocular pressure in young children will improve our diagnosis of true glaucoma rather than having to rely on optic nerve appearances, axial length measurements or increasing myopia. Simsek et al. [26] have shown that CCT increases in children who have undergone cataract surgery and this may result in an overestimation of true glaucoma and therefore an overdiagnosis.
Prevention of cataracts would be the ultimate clinical advance and while unlikely in the foreseeable future, there are measures that can reduce incidence in developing countries, such as immunization programmes for rubella and measles and improved nutrition. More effective screening and earlier referral to an ophthalmologist through focused training of the relevant healthcare workers will improve visual prognosis across the world. Also, finally, a more thorough understanding of the genetic causes of cataracts will enable more accurate genetic counselling to families.
(d). Causes of congenital and paediatric cataract
Causes of congenital or juvenile cataracts can be divided into:
—intrauterine: infection (toxoplasmosis, rubella, cytomegalovirus, herpes, varicella, syphilis), drug exposure (corticosteroids, vitamin A), ionizing radiation (X-rays), metabolic disorder (maternal diabetes);
—hereditary: (i) isolated—without associated eye or systemic disorder (autosomal dominant inheritance); (ii) complex—associated ocular or multi-system syndrome (anterior segment dysgenesis, aniridia, Stickler syndrome, myotonic dystrophy, Norrie disease, Lowe syndrome, Alport syndrome, Nance–Horan, incontinentia pigmenti, congenital icthyosis);
—chromosomal (Down syndrome: trisomy 21, Turner syndrome);
—extrinsic: malnutrition, acute dehydrating diseases;
—metabolic disease (galactosaemia, hypocalcaemia);
—trauma.
Unilateral congenital cataract, is generally not associated with systemic disease, is rarely inherited and in the majority of cases is idiopathic. Anterior polar cataract may arise from abnormal separation of the lens vesicle in early pregnancy and be associated with remnants of the tunica vasculosa lentis. Posterior cataracts may be associated with remnants of the primitive hyaloid vascular system (Mittendorf's dot) or with lenticonus/lentiglobus. It is important in unilateral cases to examine the other eye very carefully to ensure that this is not a bilateral disease with asymmetric lens involvement. Examining both parents is an essential part of any work-up given that, in developed countries, about one-third of bilateral cataracts are hereditary (without a systemic disease) and the majority are dominantly inherited [27]. An Australian study looked at the causes of paediatric cataract over a 25 year period. They identified 39 families and found the mode of inheritance was autosomal dominant in 30 families, X-linked in four, autosomal recessive in two and uncertain in three [28]. Much of our understanding of inherited cataracts in man comes from animal studies. The mouse provides an excellent model for studying genetic disease and the remainder of this review will be devoted to the knowledge we have gained and have yet to gain about the causes of congenital and juvenile-onset human cataracts from experimental studies conducted in the mouse.
3. Animal models for juvenile cataracts
Mice are one of the best animal model systems because their genetics is so well understood. Initially, cataract mutants were identified rather randomly as those mice with visible white spots in their eyes; later, systematic mutagenesis studies were undertaken (for a detailed review, see [29]). We shall focus mainly on mutants produced in this way and knockout mice models (loss of function of a particular gene) to facilitate comparisons with human cataracts. The knockout mutants represent classic null mutations and have the great advantage that the affected gene is known by default. We shall not be discussing transgenic mice models where a particular gene is over-expressed either in the lens or ectopically.
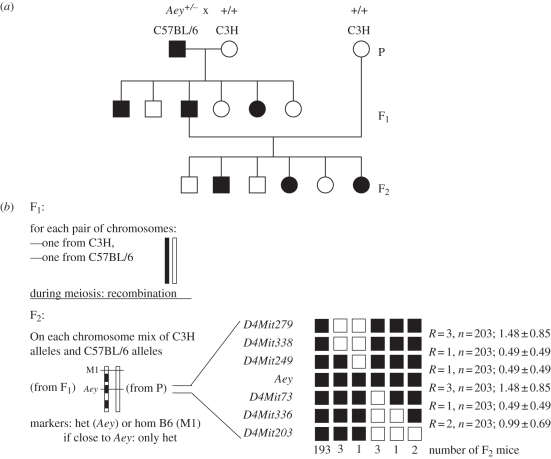
In all spontaneously or randomly induced mouse (cataract) mutants, one must initially perform linkage analysis (as in human families; figure 1). The outstanding advantage of the mouse system (compared with the rat) is the high density and diversity of already available molecular markers, which is due to the large number of inbred strains (being homozygous at all loci). This high density and diversity of elaborated and well-characterized markers is an absolute prerequisite for linkage analysis—using microsatellite markers and single nucleotide polymorphisms (SNPs). The power of resolution depends on the number of F2 offspring observed: using approximately 50 carriers, one can obtain linkage to a specific chromosome and approximately the chromosomal region of interest. However, with approximately 400 F2 offspring one can reduce the genetic interval of interest to just a few megabases. It is then a matter of identifying and sequencing the candidate genes within this region (this technique is also referred to as a ‘positional candidate approach’). In the mouse, mRNA can be extracted from the lens, allowing sequence analysis at the cDNA level and facilitating the detection of splice variants. Genomic mutations are confirmed by DNA analysis, proof of segregation within the breeding colony and only after exclusion of polymorphisms between the various inbred strains.
Figure 1.
(a) Linkage analysis: general schedule for dominant mutations. A mutant mouse with a dominant phenotype (abnormality of the eye, Aey) was identified in a C57BL/6 colony after treatment with a mutagen (e.g. ethylnitroso urea, ENU). It is crossed with a wild-type mouse of a different inbred strain (e.g. C3H); in the F1 generation, 50% of the offspring show the mutant phenotype. A mutant F1 mouse with the clinical phenotype is backcrossed to the wild-type parental strain (C3H) leading again in the F2 generation to 50% mice with the clinical phenotype. (b) The F1 mice have one chromosome from each parent. During meiosis, in a few cases, recombination processes occur between the parental chromosomes, which can be visualized in the F2 generation using different genetic markers (M1, Aey). The example shows 10 recombinations among 203 F2 mice. Recombinations are (in a first approximation) randomly distributed among the chromosome; their relative frequency increases with the distance from a given point (here: Aey). Analysis of haplotypes (carrying the same combination of marker alleles) indicates the individual recombination events in each F2 mouse and allows the precise localization of the mutation. The distance of the markers is given in genetic units (here: 2 cM), but by comparison with the already published sequence of the mouse genome, the physical distances can be calculated. The critical interval in the example given here is 1.1 MB (genomic position of the marker D4Mit249 is 125.4 MB and of the marker D4Mit73 is 126.5 MB). It is obvious that the acuracy of the position increases with the number of F2 mice analysed. This positional cloning approach allowed the identification of a point mutation in the Col8a2 gene, which is responsible for a thinner cornea in this mutant. Filled symbols represent heterozygotes, clinical phenotype present; open symbols represent homozygous wild-type; (b) modified according to Puk et al. [30]; with permission from ARVO.
4. Genes causing juvenile cataract
This section focuses on the genetic causes of congenital or paediatric cataracts in humans while providing comparable information on the corresponding mouse models. The molecular mechanisms, expression studies and histology have been much better established in the murine models. We have selected a representative sample of studies to discuss and have not provided an exhaustive list of all known mutations (refer also to the OMIM database; http://www.ncbi.nlm.nih.gov/omim/ for human mutations, to the MGI database (www.informatics.jax.org) and previous reviews by the authors [29,31,32]).
We will discuss mutations (in order) in genes coding for enzymes, structural proteins (soluble in the cytosol), cytoskeletal or membrane-associated proteins, transcription factors and signalling molecules. Human genes are denoted in italic capitals, whereas only the initial letter is capitalized for mouse genes. For an initial overview, see table 1.
Table 1.
Genes involved in congenital and paediatric cataracts. References are given in the body of the text; databases used are: OMIM (http://www.ncbi.nlm.nih.gov/omim) for human mutations; MGI (http://www.informatics.jax.org/) for mouse mutations. ASOD, anterior segment ocular dysgenesis (also referred to as ASMD, anterior segment mesenchymal dysgenesis); n.d., no ocular phenotype detected (genes are listed only if at least in humans or in mice cataracts have been reported); rec, recessive; dom, dominant; cat, cataract; chr, chromosome.
| genea | human |
mouse |
||||
|---|---|---|---|---|---|---|
| chr | dom/rec | phenotypeb | chr | dom/rec | phenotypeb | |
| enzymes | ||||||
| GALE | 1p36 | rec | (risk for) cataract | 4 | no mutation | |
| GALK1 | 17q24 | rec | cataract | 11 | n.d. | |
| NSDHL | Xq28 | n.d. | X | dom | cataract | |
| PCBD1 | 10q22 | n.d. | 10 | rec | cataract | |
| SORD | 15q15 | rec | cataract | 2 | rec | cataract |
| structural proteins (soluble in cytosol) | ||||||
| CRYAA | 21q22 | dom/rec | cataract | 17 | dom/rec | cataract |
| CRYAB | 11q22 | dom/rec | cataract | 9 | n.d. | |
| CRYBA1 | 17q11 | dom | cataract | 11 | dom | cataract |
| CRYBA3 | 22q11 | dom | cataract | 5 | n.d. | |
| CRYBB1 | 22q11 | dom | cataract | 5 | n.d. | |
| CRYBB2 | 22q11 | dom | cataract | 5 | dom | cataract |
| CRYBB3 | 22q11 | rec | cataract | 5 | n.d. | |
| CRYGA | 2q33 | n.d. | 1 | dom | cataract | |
| CRYGB | 2q33 | n.d. | 1 | dom | cataract | |
| CRYGC | 2q33 | dom | cataract | 1 | dom | cataract |
| CRYGD | 2q33 | dom | cataract | 1 | dom | cataract |
| CRYGE | 2q33 | pseudogene | 1 | dom | cataract | |
| CRYGF | 2q33 | pseudsogene | 1 | dom | cataract | |
| CRYGS | 3q27 | dom | cataract | 16 | dom/rec | cataract |
| structural proteins (membrane or cytoskeleton) | ||||||
| BFSP1 | 20p12 | rec | cataract | 2 | semi-dom | cataract |
| BFSP2 | 3q21 | dom | cataract | 9 | slight opacity | |
| COL4A1 | 13q34 | n.d. | 8 | dom | ASOD, cataract | |
| GJA1 | 6q21 | dom/rec | oculodentodigital Dysplasia | 10 | dom | abnormal iris, cornea opacity |
| GJA3 | 13q11 | dom | cataract | 14 | rec | cataract |
| GJA8 | 1q21 | dom | cataract | 3 | dom | cataract |
| GJF1 | n.d. | 10 | dom | (Aey12); cataract | ||
| LIM2 | 19q13 | rec | presenile cataract | 7 | dom | (To3); cataract |
| MIP | 12q13 | dom | cataract | 10 | dom | cataract |
| NHS | Xp22 | dom | Nance–Horan syndrome (cataract) | X | semi-dom | cataract |
| NRCAM | 7q31 | n.d. | 12 | rec | cataract | |
| SPARC | 5q31 | n.d. | 11 | rec | cataract–age onset? | |
| VIM | 10p13 | dom | cataract | 2 | n.d. | |
| transcription factors | ||||||
| FOXE3 | 1p32 | dom; rec | ASOD, cataract; aphakia | 4 | rec | cataract (dyl) |
| HSF4 | 16q21 | dom | lamellar cataract | 8 | rec | cataract (lop11) |
| v-MAF | 16q22 | dom | pulverulent cataract | 8 | dom | cataract (Ofl) |
| PAX6 | 11p13 | dom | aniridia, Peters' anomaly, cataract | 2 | dom | small eye, cataract |
| PITX3 | 10q25 | dom | posterior polar cataract | 19 | rec | no lens (ak, eyl) |
| SIX5 | 19q13 | ?? | (adult) iridescent cataract | 7 | dom | cataract |
| SOX1 | 13q34 | n.d. | 8 | ?? | microphthalmia, cataract | |
| SOX2 | 3q26 | dom | anophthalmia, microphthalmia | 3 | dom? | anophthalmia |
| signalling molecules | ||||||
| EPHA2 | 1p36 | dom | cataract | 4 | rec | (age-related) cataract |
| EFNA5 | 5q21 | n.d. | 17 | rec | cataract | |
aGene symbols are given for humans; the corresponding mouse symbol has mostly the same letters, but only the first letter is capitalized.
bOnly ocular features are given. In many cases, the mutations in the affected genes have additional, pleiotropic effects.
(a). Mutations in genes encoding enzymes
(i). Galactosaemia
Inborn errors in the galactose pathway have been recognized as a cause of paediatric cataract for more than 90 years (OMIM 230400). In classical galactosaemia, cataracts can be observed within a few days after birth. Mutations in the gene coding for galactokinase (GALK1) are the major cause of galactosaemia; galactokinase catalyses the first step in the galactose pathway (galactose → galactose-1-phosphate). In the disease state, galactose accumulates in the lens and is converted to the corresponding sugar alcohol (galactitol) by aldose reductase. Accumulation of galactitol leads to an influx of water with the subsequent development of cataracts. One of the most prominent pathological alleles is the A198V polymorphism in GALK, which has a prevalence of 3–4% among Asians. It has been shown to be significantly over-represented in Japanese individuals with bilateral cataracts [33]. Another ‘common pathological allele’ is P28T, which has a prevalence of approximately 5 per cent among the Romani population (Gypsies). It is calculated that approximately one in 2000 newborns suffers from galactosaemia and cataracts, suggesting that this allele may be an important cause of early childhood blindness in this population [34].
For a long time, appropriate mouse models were missing. One hypothesis was that aldose reductase has a very low activity in the mouse when compared with humans. Therefore, it was not surprising that the galactokinase (Glk1) knockout in the mouse did not have cataracts. However, the introduction of a human aldose reductase transgene into a Glk1-deficient background resulted in cataract formation by the first postnatal day [35], thus highlighting the importance of aldose reductase in sugar-dependent cataract formation.
(b). Mutations in genes encoding structural proteins
The crystallins were discovered more than 100 years ago by Mörner [36] as the main structural proteins of the vertebrate eye lens. Since that time, the major mammalian crystallins, referred to as α-, β- and γ-crystallins, were characterized with respect to their genetic organization, regulation of their expression pattern and participation in cataracts, but also in several other diseases. Since the focus of this article is on the genetics of cataracts, we cannot cover all details of the cellular and biochemical functions of the different crystallins (for a recent review of crystallins, see [31] and references therein).
(i). β-/γ-crystallin genes
The largest subgroup of mouse cataracts are caused by mutations in the γ-crystallins (gene symbol for humans, CRYG; mouse, Cryg). The CRYG/Cryg genes belong to the superfamily of β- and γ-crystallin-encoding genes and comprise six closely related genes (CRYGA/Cryga → CRYGF/Crygf) on human chromosome 2q33 and mouse chromosome 1 as well as CRYGN/Crygn and CRYGS/Crygs on two further chromosomes (human chromosomes 7q36 and 3q25 and mouse chromosomes 5 and 16). The CRYG/Cryg genes are expressed in the lens at a very high level and are thought to encode structural proteins; the γ-crystallin proteins are characterized by four so-called Greek-key motifs. The first CRYG/Cryg mutation was characterized in 1992 by Cartier et al. [37] in the mouse (Elo, eye lens obsolescence). To date, 23 mouse mutants affecting the Cryg gene cluster have been published, affecting all six genes of the Cryg gene cluster (for a review, see [31] and references therein).
In man, an increasing number of families suffering from hereditary congenital dominant cataracts have been identified as having mutations in the CRYG genes. It might be of evolutionary interest that two of the six CRYG genes are pseudogenes (ψCRYGE and ψCRYGF), found not to be necessary for human lens function [38]. The first mutation in CRYGC was identified by Héon et al. [39] as producing the Coppock-like cataract. It is worth mentioning that all mutations that have been characterized so far in the CRYG gene cluster have been found in CRYGC and CRYGD only, and none in CRYGA or CRYGB.
Current knowledge suggests that mutations in the CRYG/Cryg genes only affect the lens; however, there is a striking diversity in the cataract phenotypes. Across the spectrum of mice with a mutated Cryg gene, the consequences for the lens varies and might be related to distinct functions of the individual γ-crystallin or to the affected domains within a particular γ-crystallin or to the time point when gene expression starts and/or to the particular level of expression. Loss of solubility of the mutated crystallin protein has been frequently suggested as the cause for cataract formation [40]. However, for at least a subset of three mutations, a key feature of cataractogenesis is the formation of intra-nuclear inclusions containing the altered γ-crystallins in the primary lens fibre cells. It precedes not only the first gross morphological changes in the lens, but also the first signs of cataract. The inclusions contain filamentous material that can be stained with the amyloid-detecting dye, Congo red. Furthermore, in vitro studies confirm that this type of cataract is caused by a mechanism involving nuclear targeting and disrupting of nuclear functions via deposition of amyloid-like inclusions [41].
Evolutionary intermediate members of the β-/γ-crystallin superfamily are genes encoding the γS- and γN-crystallins on chromosomes 3q27 and 7q26, respectively. In Crygn, the first two Greek key motifs are encoded by individual exons (this is the Cryb type), but the remaining two by one single exon (it is the Cryg type). In contrast, the Crygs gene belongs from the genetic point of view clearly to the Cryg gene family; however, the biochemical feature (blocked N-terminus) caused its past designation as βs-crystallin [42]. In humans, two CRYGS mutations have been reported resulting in dominant juvenile cataracts, either cortical [43] or progressive [44]. In the mouse, two cataract mutations have been identified in Crygs, Opj (opacity due to poor junctions) as a semi-dominant progressive cataract and a recessive nuclear cataract (allele symbol: rncat). Histology of the homozygous Opj-cataract shows that both cortical fibre cell morphology and the loss of maturing fibre cell nuclei are severely disrupted from early stages. The underlying molecular alteration is a highly non-conservative exchange, F9S [45]. In contrast, the rncat mutation leads to a truncation of the γS-crystallin (W163X). Histological analysis of the homozygous mutants shows a severe degeneration of the epithelial cells underneath the anterior lens capsule; the cells in the equatorial region display an excessive proliferation and migration. Within the cortical area underneath the posterior lens capsule, vacuoles and Morgagnian-like bodies are seen. In the embryonic nucleus, blue-stained spherical bodies are observed forming a Y-like pattern [46]. No mutations have been reported in Crygn. This might be due to the relatively low expression level in the mouse. The role of CRYGN in humans is less clear, as illustrated by the observation that CRYGN undergoes major changes leading to different splice forms and expression in the retinal pigmented epithelium, hippocampus and testes, but not in the lens [47].
The family of β-crystallins can be divided into the more acidic (βA-) and the more basic (βB-) crystallins. Each subgroup is encoded by three genes (Cryba1, -2, -4; Crybb1, -2, -3). Cryba1 codes for two proteins (βA1- and βA3-crystallin). This feature is conserved among all mammals, birds and frogs. In humans and mice, the six CRYB/Cryb genes are mapped to three different chromosomes. Beta-crystallins are expressed early during lens development, with expression continuing to rise after birth so that the highest concentrations are usually found in the lens cortex. The expression pattern varies among the individual β-crystallins, but there is increasing evidence that Crybb2 is expressed not only in the lens, but also in the testes, retina and the brain [48–50].
Several human cataract mutations have been attributed to the CRYB genes, most of which are dominant, but two are recessive (one allele each in CRYBB1 and CRYBB3). The first mutation in a human CRYB gene was identified in CRYBB2 by Litt et al. [51]; the Q155X mutation leads to a cerulean cataract. After detection of several independent human mutations with the same genotype (but variable clinical manifestations), Vanita et al. [52] postulated that all these mutations have a common mechanism: gene conversion to a pseudogene, which is closely linked to the functional CRYB gene cluster. Cataract-causing mutations have been found also in other CRYB genes, such as CRYBA1, CRYBA4, CRYBB1 and CRYBB3.
In the mouse, three mutations in the Crybb2 gene have been reported, and one of these, found in the Philly mouse, was the first cataract mutant to be fully characterized by sequencing [53]. All have progressive cataracts with slightly different morphologies while the mutations affect the same region of the βB2-crystallin: the beginning of the fourth Greek key motif. Recently, it was demonstrated that the Philly mice suffer from reduced fertility since the Crybb2 mutation also affects testicular function [49]. As wild-type and mutant βB2-crystallins are expressed in other tissues, such as the cerebellum, olfactory bulb, cerebral cortex and hippocampus, additional behavioural or systemic effects might be expected [50].
(ii). α-crystallin genes
The α-crystallins form large complexes with a molecular mass of approximately 800–1000 kDa. They are mainly composed of two related proteins, αA- and αB-crystallin, which are encoded by two genes (CRYAA and CRYAB) on human chromosomes 21q22 and 11q22, respectively (and on mouse chromosomes 17 and 9; Cryaa and Cryab). The two CRYA/Crya genes are expressed at very high levels in the lens.
The αA-crystallin protein is expressed in the mouse lens cup at E10.0 to E10.5, later in the posterior half of the lens vesicle, and after birth, αA-crystallin becomes very abundant in lens fibre cells [54,55]; αA-crystallins are found also in the retina [56]. Mutations in the mouse Cryaa gene show features that are interesting because of their phenotypic diversity (figure 2). There are two mouse models leading to dominant cataracts (V124E; Y118D). In contrast, the report of the Cryaa knockout mice by Brady et al. [57] was surprising, since only the homozygous null mutants developed cataracts. Similarly, one point mutation in the Cryaa gene produced a recessive phenotype in the mouse; the missense mutation 161G → A converts at codon 54 Arg → His (R54H). Surprisingly, a point mutation at position 160 (C → T) leading to the same R54H exchange was characterized as a dominant cataract mutation in the mouse. In humans, the same mutation (160C → T) but leading to an Arg → Cys exchange at pos 54 (R54C) was reported to cause dominant [44] and semi-dominant cataracts [60]. A ‘true’ recessive cataract is caused by a nonsense mutation affecting the N-terminal region of the αA-crystallin (W9X [61]). It is of great interest from a genetic point of view that mutations in the CRYAA/Cryaa gene lead to both recessive and dominant phenotype; (an overview of the known mutations in the human CRYAA and the corresponding mouse Cryaa gene is shown in figure 3). The underlying mechanisms explaining this remain unknown but are unlikely to be due solely to differences in the so-called ‘genetic background’. The influence of particular polymorphic alleles (SNPs) within the CRYAA/Cryaa genes themselves might play a role, as well as a stronger or weaker expression of other genes that overlap at least part of the function of the mutated gene(s) (functional redundancy). The elaboration of the underlying molecular mechanisms remains a challenge for active and future researchers in this field.
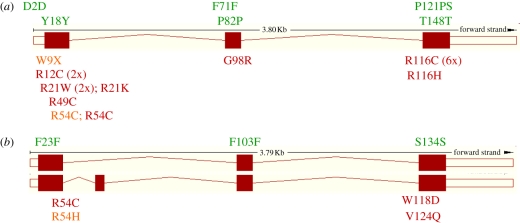
Figure 2.
Cataractous lenses from mouse Cryaa mutations. The panels show different mouse mutations affecting Cryaa leading to (a) recessive or (b) dominant cataracts. In the recessive mode of inheritance, only the homozygous carriers suffer from severe nuclear cataracts associated with a small lens size. In dominant mutations the heterozygotes show cataracts, but in the cases reported here, the homozygous mutants are more severely affected. Since the pictures are from different sources and represent different ages, the lens size cannot be compared directly. (According to [57], with permission from PNAS; [58] and [59], with permissions from ARVO.)
Figure 3.
Mutations in CRYAA/Cryaa gene of humans and mice. Sequence alterations in (a) the human CRYAA and (b) mouse Cryaa gene are shown as amino-acid exchanges (single-letter code) at the corresponding position. The mouse Cryaa has two alternative transcripts, the insertion between exons 1 and 2 is present in approximately 10% of the transcripts and is not observed in humans. It is obvious that most of the cataract-causing mutations affect the first or third exons. Mutations in the second exon are rare. SNPs (above the exons) are synonymous; only the human polymorphism P121PS shows an insertion of 3 bp leading to an additional serine residue. Mutations in the CRYAA/Cryaa gene lead mainly to dominant cataracts (dark grey), but in some cases also to a recessive mode of inheritance (light grey). (Data from www.ensembl.org; www.informatics.jax.org; www.ncbi.nlm.nih.gov/snp.)
In contrast to CRYAA/Cryaa, CRYAB/Cryab is expressed ubiquitously. In mice lenses, αB-crystallin is present at E9.5 and later on is found preferentially in the epithelial cells [55]. Several recessive and dominant cataract mutations are described in human CRYAB. Because of the ubiquitous expression pattern of the human CRYAB, at least some of these patients suffer from other diseases such as myopathies/cardiomyopathies. It is also well known that αB-crystallin is associated with neurodegenerative disorders (including Alexander's disease, Alzheimer's disease, Parkinson's disease), where it is found in the characteristic pathological structures (Rosenthal fibres, Lewy bodies; for a recent review, see [31]). Surprisingly, the homozygous mouse knockout of the Cryab gene revealed a phenotype that could not be distinguished from the wild-type. The mutant lenses developed like the wild-type, and all other crystallins were present. The authors concluded from these results that mouse Cryab is not essential for normal lens development [62]. In a similar fashion to human patients, Cryab−/− mice after an ischaemia–reperfusion-induced damage of the myocardium show a twofold reduction in contractile recovery as well as increased necrosis and apoptosis when compared with the wild-type controls [63].
(c). Mutations in genes encoding cytoskeletal proteins
There are three major cytoskeletal proteins in the lens, filensin (CP94 or beaded filament structural protein 1; gene symbol BFSP1/Bfsp1), phakinin (also referred to as CP49 or beaded filament structural protein 2; gene symbol BFSP2/Bfsp2) and vimentin (gene symbol VIM/Vim). In a human dominant congenital pulverulent cataract, Müller et al. [64] identified a mutation (596G → A; E151K) in the VIM gene; however, vimentin knockout mice mutants do not show an ocular phenotype [65].
In humans, a mutation in the filensin-encoding gene BFSP1 has been reported to cause recessive cataract [66], but mutations in the CP49-encoding gene BFSP2 were found to be responsible for dominant cataracts [67,68]; their phenotypes, however, seem to be variable ranging from congenital nuclear, sutural or stellate cataracts to juvenile-onset cataracts.
Disruption of the Bfsp1 gene in the mouse reduces levels of filensin's assembly partner CP49 and prevents the assembly of beaded filaments. These knockouts show evidence of light scattering by two months, which worsens with age. Heterozygous animals exhibit an intermediate phenotype with a moderate light scattering at five months. However, the Bfsp2 knockout (or deletion of a splice-acceptor site) has only a subtle loss of optical clarity in the lens. This splice-site mutation is present in several mouse strains (129, 101, CBA and FVB) and might interfere with other mutations or targeted deletions and, therefore, have important implications for future lens studies using these strains [69,70].
Another protein that is associated with the lens cytoskeleton and epithelial cell junctions is the Nhs1 protein, which, when mutated, causes the X-linked dominant Nance–Horan syndrome (NHS), also known as cataract–dental syndrome. Affected males have dense nuclear cataracts and frequently have microcornea, whereas heterozygous females show suture cataracts with small corneas and slightly reduced vision (OMIM 302350). Mutations in the NHS gene have been identified in several families [71,72]. A large insertion between exon 1 and exon 2 of the mouse Nhs1 gene induced by paternal radiation is responsible for the X-linked dominant cataract Xcat. Histological analysis during embryonic development revealed that in the affected embryos the primary lens fibre cells are irregularly arranged and show small foci of cellular disintegration; the fibres progressively degenerate. At the molecular level, the insertion inhibits the expression of the Nhs1 isoform containing exon 1 and results in exclusive expression of the alternative isoform containing exon 1A. The presence of Nhs1 exon 1 is critical for localization of the protein to the cytoplasm, and proteins lacking Nhs1 exon 1 are predominantly in the cell nucleus [73].
(d). Mutations in genes encoding membrane-associated proteins
(i). Connexins
Connexins are well known as a family of proteins, and have been shown to be structural and functional building blocks of gap junctions (intercellular channels). They span two plasma membranes and provide a direct pathway for the movement of signalling molecules and ionic currents between adjacent cells [74]. In the lens, three connexins play major roles during development and adulthood: connexin43 (Cx43), connexin46 (Cx46) and connexin50 (Cx50). The gene symbols are GJA1/Gja1, GJA3/Gja3 and GJA8/Gja8 (gap junction subunit α1, α3 or α8), and they map to human chromosomes 6q21, 13q11 and 1q21, respectively. In the mouse, they are located on chromosomes 3 (Gja3 and Gja8) and 10 (Gja1). The designation as connexin43, -46 or -50 is based upon their apparent molecular weight.
Cx43 (encoded by GJA1/Gja1) can be found in the mouse lens placode (E10) and later on in the optic vesicle, predominantly in the posterior portion that will become the pigmented layer of the retina. When the lens vesicle has been formed, Cx43 is present primarily in the cells destined to become the anterior epithelium. Cx43 gradually disappears at the margin of the epithelial layer, and only low concentrations can be identified between the lens fibre cells [75]. Mutations in human GJA1 lead to dominant and recessive forms of oculodentodigital dysplasia; the ocular phenotype includes microphthalmia and microcornea (OMIM).
In Gja1 knockout mice, the lens epithelial cells are connected more loosely compared with the wild-type. However, organization of adjacent membranes among lens fibre cells and between fibre and epithelial cells differs dramatically in the Gja1−/− lens: fibre cells in Gja1−/− lenses are largely separated from apical surfaces of epithelial cells, and large vacuolar spaces are apparent between fibre cells, most notably in the deeper cortical regions. These changes suggest that the osmotic balance within these cells is markedly altered.
Gja3 coding for Cx46 is expressed mainly in the lens fibres, but not in lens epithelium [76]. In humans, several cataract-causing mutations in GJA3 have been reported (OMIM 121015). In mice, a Gja3 gene knockout mutant has been described resulting in nuclear cataract in homozygous mutants and associated with the proteolysis of γ-crystallins [77]. Interestingly, the genetic background of the loss of Gja3 gene activity influences the severity of cataract. While Gja3 null mutations on two 129 mouse strains suffered from severe cataracts with γ-crystallin cleavage, Gja3−/− mice on the C57Bl6 background had far milder cataracts with no detectable γ-crystallin cleavage [78]. Moreover, cataract formation can be prevented in Gja3−/− mice by the general cysteine protease inhibitor E-64 (its primary targets being the calcium-dependent proteases m-calpain and Lp82) [79]. More recent studies suggest that calpain 3 is responsible for the γ-crystallin cleavage and for the formation of nuclear cataracts in Gja3−/− mice [80].
Cx50 (encoded by the gene Gja8) is present mainly in the lens fibre cells, but also in the lens epithelial cells. In the mouse, White et al. [81] showed that Cx50 influences epithelial cell proliferation; the contribution of Cx50 is highest during early postnatal proliferation but progressively declines with age thereafter. In humans, the importance of Cx50 for lens development and function is demonstrated by several dominant cataracts caused by mutations in the GJA8 gene (R23T, E48K, P88S, OMIM 600897). Of particular interest is the I247M allele, which was shown to co-segregate perfectly with the cataract in a three-generation Russian family [82]. However, Graw et al. [83] found that, while present, this allele did not co-segregate with cataracts in a German family. Furthermore, since the I247M allele is capable of inducing gap-junctional currents in pairs of Xenopus oocytes, it has to be considered as a rare polymorphism, rather than a cataract-causing allele.
In the mouse, four point mutations have been reported affecting the Gja8 gene with bilateral, congenital nuclear opacities; in some lines, however, the severity is greater in the homozygous mutants. In three cases, the first extracellular loop is affected (D47A, S50P, V64A) and in the fourth mutant (G22R) the N-terminal part of the protein is affected. Additionally, knockout mutations of the Gja8 gene have been created (MGI database). Among the point mutations, the S50P mutation is of particular interest: in situ dye-transfer experiments showed that this allele greatly inhibited epithelial cell gap-junctional communication by diminishing electrical coupling in vitro. The authors [84] suggest that dominant inhibition of Cx43-mediated epithelial cell coupling may play a role in the lens pathophysiology caused by the Gja8S50P mutation.
(ii). Aquaporin0 and other membrane-associated proteins
Aquaporin0, also referred to as MIP (major intrinsic protein of lens fibres), forms specialized junctions between the fibre cells. Mip transcripts and Mip protein were first detected at E11 in the differentiating primary fibre cells; its synthesis continued through the adult stage in the secondary fibre cells [85]. The human MIP gene is located on chromosome 12q13, and Berry et al. [86] identified two dominant mutations (G134E and T138R) leading to polymorphic and lamellar cataract.
One of the first mouse cataract strains that was identified is known as Cataract Fraser (CatFr [87]). In this mutant, the cell nuclei in the deep cortex become abnormally pycnotic (beginning at E14); sequence analysis revealed that the CatFr mutation is due to a transposon-induced splicing error leading to a truncated form of Mip transcripts. There are two further mouse mutants characterized by mutations in the Mip gene; a single amino acid substitution in MipLop inhibits targeting of Mip to the cell membrane, and MipTohm is characterized by a 12 bp deletion affecting the second transmembrane region.
LIM2/Lim2 is located on human chromosome 19q13 (mouse chromosome 7) and codes for a lens-specific integral membrane protein, also referred to as MP19. In humans, two mutations in the LIM2 gene are associated with recessive cataracts [88,89].
In mice, Lim2 mRNA were found in the head region of embryos from embryonic day 12 by Zhou et al. [90]. Mice heterozygous or homozygous for a Gly15Val mutation in Lim2 exhibit dominant, dense cataracts. Moreover, homozygotes have microphthalmia and posterior lens rupture.
(e). Mutations in genes coding cell signalling proteins
One of the central genes in both mouse and human eye development is the paired-box gene PAX6/Pax6, mutations in which are known to cause aniridia (in man) with and without cataracts and Peters anomaly [91]; it is localized on chromosome 11p13. However, there is mounting evidence that PAX6 mutations may also cause other behavioural and neurodevelopmental phenotypes [92,93] as well as disorders of the pancreas [94]. The PAX6 database contains more than 300 entries of human mutations (http://lsdb.hgu.mrc.ac.uk/home.php?select_db=PAX6).
In mice and rats, mutations in Pax6 are responsible for the dominant Small eye (Sey) mutants. Pax6 maps to mouse chromosome 2; the Jackson Laboratory list contains 40 alleles in the mouse, 10 of which are targeted or gene-trapped mutations (http://www.informatics.jax.org; January 2011). Usually, the expressivity of heterozygous Pax6 mutations is variable, with individuals exhibiting a range of phenotypes from small anterior polar cataracts to the more visually significant anterior polar lens opacities, corneal adhesions, iris abnormalities and microphthalmia. Furthermore, the degree of phenotype expressed between the two eyes of the same person is variable ([95] and references therein).
Another interesting gene to consider is Pitx3. Heterozygous mutations in the human PITX3 gene (chromosome 10q25) can give rise to dominant congenital cataracts and, in some cases, the phenotype includes other features of anterior segment dysgenesis [96]. There is one report of a homozygous PITX3 mutation (ΔG650) in a consanguineous family; the corresponding patients suffer from microphthalmia and significant neurological impairment; the heterozygous carriers suffer from posterior polar cataract [97].
The recessive mouse mutant aphakia (ak) carries two deletions in the promoter of the Pitx3 gene, leading to a small lens vesicle attached directly to the cornea via a ‘lens stalk’. In later stages, the lens vesicle is degraded and a lens-less eye is formed giving this mutant its name. It should be noted that the ocular phenotype in the mouse is quite different from that in humans with PITX3 mutations. Another Pitx3 allele, eyeless, is characterized by a single-base pair insertion (416insG). The ocular phenotype is similar to aphakia; the heterozygotes have normal eyes. However, the homozygous mutants also suffer from Parkinsonism and from enhanced nociception [98]. This correlates well with the fact that Pitx3 is also expressed in the dopaminergic neurons of the substantia nigra in the brain and when mutated leads to selective neural loss and malformation of the mesencephalic dopamine system [99]. Epidemiological studies in humans have shown associations between polymorphic sites in the PITX3 gene and Parkinson's disease [100].
The Fox transcription factors are characterized by a 110-amino acid motif originally defined as a DNA-binding domain in the Drosophila transcription factor forkhead (Fox: forkhead box). In humans, heterozygous mutations in FOXE3 lead to anterior segment dysgenesis [101]. One consanguineous family has been described with a homozygous nonsense mutation in FOXE3 causing primary congenital aphakia, microphthalmia and complete agenesis of the anterior segment [102]. In mice, a mutation in FoxE3 causes the phenotype observed in another mouse mutant, dysgenic lens (dyl). In this mutant, the lens vesicle fails to separate from the ectoderm, causing the lens and the cornea to fuse. Mutations in the human FOXC1 and corresponding mouse mutant lead to similar phenotypes, including iris hypolplasia, anterior segment dysgenesis, glaucoma but no cataracts [103,104].
Recently, the molecular features underlying the vacuolated lens (vl) mutation have been characterized. This mutation arose spontaneously on the C3H/HeSnJ background [105], and homozygous mutants suffer from congenital cataracts and neural tube defects. Positional cloning strategies mapped the mutation to the distal part of mouse chromosome 1, and identified finally an 8 bp deletion in the gene Gpr161 encoding an orphan G protein-coupled receptor. Together with this causative mutation three modifier loci have been identified, one of them being FoxE3 [106]).
The Maf family of basic region leucine zipper (bZIP) transcription factors was first identified through the v-maf oncogene, an avian retrovirus transforming gene [107]. Maf binds as a homo- or heterodimer to two known Maf responsive elements (MAREs), with varying affinities and transactivation potentials. Two families with mutations in MAF have been identified with ocular developmental abnormalities (in one case, cataract is associated with anterior segment dysgenesis and microphthalmia; in the other case, cataract is associated with microcornea and iris coloboma) [108]. In mice, targeted deletion of c-Maf leads to arrested elongation of primary lens fibres at the lens vesicle stage and a point mutation in the basic region of the DNA-binding domain of c-Maf causes a mild pulverulent cataract mutant in mouse (opaque flecks in the lens, Ofl).
Heat-shock transcription factor 4 (gene symbol HSF4/Hsf4) is important in lens development and differentiation. In humans, mutations in HSF4 are associated with various recessive and dominant forms of congenital and age-related cataracts [109–115]. Similarly, the Hsf4 knockout mice (Hsf4−/−) develop cataracts. Detailed analyses of their lenses demonstrated downregulation of several genes implicated in cataract formation, including the genes encoding γS-crystallin (Crygs) and beaded filament proteins 1 and 2 (Bfsp1 and Bfsp2). Further studies have confirmed these genes as direct downstream targets of Hsf4 [116].
Ephrin-A5 (gene symbol: EFNA5/Efna5) is well known as a ligand of the ephrin receptor tyrosine kinase and to be involved in axonal guidance and cell differentiation. It is also vital for regular lens development and differentiation, since knockout mutants of Efna5 develop cataract in approximately 87 per cent of the mutants. The lens fibre cells appear rounded and irregular in cross section [117]. Furthermore, ephrin-A5 is known to interact with the ephrin-A2 receptor (gene symbol EPHA2/Epha2) to regulate the adherens junction complex by recruitment of β-catenin to N-cadherin. It should be noted that mutations in the human EPHA2 gene lead to childhood cataracts [118,119], but also to age-related cataracts [120]. Moreover, similar to the transcription factor Pitx3 (which is crucial for early lens development), ephrin-A5 is also involved in the formation of dopaminergic neurons in the substantia nigra [121]. It might be speculated that Pitx3 and Efna5 are acting in the same signalling cascade both in the eye and brain.
5. Conclusions and outlook
This review summarizes the genetic and clinical diversity of congenital hereditary cataracts. It is obvious that there is no single cause for cataracts as outlined in table 1, showing 42 genes that are known to lead to cataract in humans or mice if mutated. Since there are still families suffering from cataracts with mutations in unknown genes and cataractous mouse mutant lines that have not yet been characterized at the molecular level, we can expect even more genes to be involved in cataract formation. However, it is also obvious that the same clinical phenotype may have distinct genetic origins. This is exemplified in figure 4 where the same phenotype of pulverulent cataracts is caused by mutations in three different genes: CRYBA1, GJA8 or MAF. This difference in the genetic origin of a given cataract is not only of academic interest, but may also have clinical consequences as these genes are expressed in other tissues (e.g. in mice, Cryba1 in the thymus, Gja8 in the brain and Maf in bone marrow, cerebellum, hypothalamus, kidney, pancreas and thymus). In some individuals, non-ocular complications might be anticipated and may require further investigation. In the future, therefore, clinicians might come to consider cataracts not only as a diagnosis in their own right but also as a biomarker for other more complex disorders.
Figure 4.
Genetic heterogeneity of pulverulent cataracts. Phenotype–genotype correlations in hereditary, congenital cataracts are generally not possible. For example, several pulverulent cataracts are shown, caused by mutations in different genes as indicated above by corresponding slit-lamp or Scheimpflug photographs. Similar examples could be given for any other type of cataract. (According to [58], with permission from the BJM group; [122] and [123], with permission from Mol. Vis.; [124] with permission from the BMJ group.)
Footnotes
One contribution of 10 to a Theme Issue ‘The ocular lens: a classic model for development, physiology and disease’.
A glossary of clinical terms
- Amblyopia or ‘lazy eye’
Partial or complete loss of vision. This can be caused by anything that interfers with the clarity of vision and prevents normal binocular visual development such as a ‘turn’ in one eye (strabismic amblyopia) or a difference in the refractive power of each eye (anisometropic amblyopia)
- Aniseikonia
A condition in which the shape and size of the ocular image differs in each eye
- Aphakia
Absence of a lens
- Capsulorhexis
A continuous circular tear in the anterior lens capsule made during cataract surgery to allow removal of the lens
- Capsulotomy
A hole made in either the anterior or posterior lens capsule to allow access to either the lens (anterior capsulotomy) or structures behind the lens (posterior capsulotomy) e.g. vitreous gel
- IOL (intraocular lens)
Artificial lens placed inside the eye to improve vision
- Nystagmus
Rapid, repetitive, rhythmic involuntary eye movements which can be horizontal, vertical or rotary
- Occlusion therapy
Treatment whereby the better sighted eye is covered for a period of hours each day to improve visual development in the other eye
- Posterior capsule opacification (PCO)
Clouding of the normally transparent posterior capsule due to migration and proliferation of lens epithelial cells following cataract surgery
- PCO Score
A measure of PCO which may be subjective (visual deterioration or capsule appearance) or objective (light scattering from the capsule measured by area densitometry)
- Refractive power
Ability of the eye to focus an image clearly on the retina. Determined by the shape of the cornea, lens and axial length of the eyeball
- Strabismus
A condition in which the visual axes of the eyes are not parallel and the eyes appear to be looking in different directions
- Viscoelastics
Gel substances injected into the eye during surgery to maintain form and also used to protect the corneal endothelium from mild abrasive trauma during eye surgery
- Vitrectomy
Surgical removal of the vitreous gel in the eye
- YAG rate
Measure of the frequency of YAG laser capsulotomies that are performed to improve vision that has deteriorated due to PCO.
References
- 1.Wilson M. E., Pandey S. K., Thakur J. 2003. Paediatric cataract blindness in the developing world: surgical techniques and intraocular lenses in the new millennium. Br. J. Ophthalmol. 87, 14–19 10.1136/bjo.87.1.14 (doi:10.1136/bjo.87.1.14) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Rahi J. S., Dezateux C. 2001. Measuring and interpreting the incidence of congenital ocular anomalies: lessons from a national study of congenital cataract in the UK. Invest. Ophthalmol. Vis. Sci. 42, 1444–1448 [PubMed] [Google Scholar]
- 3.Evans J. 1995. Causes of blindness and partial sight in England and Wales 1990–91. Studies on medical and population subjects, vol. 57 London, UK: HMSO [Google Scholar]
- 4.Kim K.-H., Ahn K., Chung E.-S., Chung T.-Y. 2008. Clinical outcomes of surgical techniques in congenital cataracts. Korean J. Ophthalmol. 22, 87–91 10.3341/kjo.2008.22.2.87 (doi:10.3341/kjo.2008.22.2.87) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Birch E. E., Stager D. R. 1996. The critical period for surgical treatment of dense congenital unilateral cataract. Invest Ophthalmol. Vis. Sci. 37, 1532–1538 [PubMed] [Google Scholar]
- 6.Elston J. S., Timms C. 1992. Clinical evidence for the onset of the sensitive period in infancy. Br. J. Ophthalmol. 76, 327–328 10.1136/bjo.76.6.327 (doi:10.1136/bjo.76.6.327) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Awaya S., Miyake S. 1988. Form vision deprivation amblyopia: further observations. Graefes Arch. Clin. Exp. Ophthalmol. 226, 132–136 10.1007/BF02173300 (doi:10.1007/BF02173300) [DOI] [PubMed] [Google Scholar]
- 8.Dubowitz L. M., Mushin J., De Vries L., Arden G. B. 1986. Visual function in the newborn infant: is it cortically mediated? Lancet 327, 1139–1141 10.1016/S0140-6736(86)91847-7 (doi:10.1016/S0140-6736(86)91847-7) [DOI] [PubMed] [Google Scholar]
- 9.Vishwanath M., Cheong-Leen R., Taylor D., Russell-Eggitt I., Rahi J. 2004. Is early surgery for congenital cataract a risk factor for glaucoma? Br. J. Ophthalmol. 88, 905–910 10.1136/bjo.2003.040378 (doi:10.1136/bjo.2003.040378) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lambert S. R. 2007. Treating amblyopia in aphakia and pseudophakic children. Am. Orthopt. J. 57, 35–40 10.3368/aoj.57.1.35 (doi:10.3368/aoj.57.1.35) [DOI] [Google Scholar]
- 11.Taylor D., Wright K. W., Amaya L., Cassidy L., Nischal K., Russell-Eggitt I. 2001. Should we aggressively treat unilateral congenital cataracts? Br. J. Ophthalmol. 85, 1120–1126 10.1136/bjo.85.9.1120 (doi:10.1136/bjo.85.9.1120) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Stewart C. E., Stephens D. A., Fielder A. R., Moseley M. J. & the ROTAS Cooperative 2007. Objectively monitored patching regimens for treatment of amblyopia: randomised trial. Br. J. Med. 335, 707. 10.1136/bmj.39301.460150.55 (doi:10.1136/bmj.39301.460150.55) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tacagni D. J., Stewart C. E., Mosely M. J., Fielder A. R. 2007. Factors affecting the stability of visual function following cessation of occlusion therapy for amblyopia. Graefe's Arch. Clin. Ophthalmol. 245, 811–816 10.1007/s00417-006-0395-2 (doi:10.1007/s00417-006-0395-2) [DOI] [PubMed] [Google Scholar]
- 14.Gouws P., Hussin H. M., Markham R. H. C. 2006. Long term results of primary posterior chamber intraocular lens implantation for congenital cataract in the first year of life. Br. J. Ophthalmol. 90, 975–978 10.1136/bjo.2006.094656 (doi:10.1136/bjo.2006.094656) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Barry J.-S., Ewings P., Gibbon C., Quinn A. G. 2006. Refractive outcomes after cataract surgery with primary lens implantation in infants. Br. J. Ophthalmol. 90, 1386–1389 10.1136/bjo.2006.097469 (doi:10.1136/bjo.2006.097469) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Yorston D. 2004. Surgery for congenital cataract. Community Eye Health 17, 24–25 [PMC free article] [PubMed] [Google Scholar]
- 17.Solebo A. L., Russell-Eggitt I., Nischal K. K., Moore A. T., Cumberland P., Rahi J. S. & on behalf of the British Isles Congenital Cataract Interest Group 2009. Cataract surgery and primary intraocular lens implantation in children ≤2 years old in the UK and Ireland: finding of national surveys. Br. J. Ophthalmol. 93, 1495–1498 10.1136/bjo.2009.160069 (doi:10.1136/bjo.2009.160069) [DOI] [PubMed] [Google Scholar]
- 18.Zetterström C., Lundvall A., Kugelberg M. 2005. Cataracts in children. J. Cataract Refract. Surg. 31, 824–840 10.1016/j.jcrs.2005.01.012 (doi:10.1016/j.jcrs.2005.01.012) [DOI] [PubMed] [Google Scholar]
- 19.Flitcroft D. I., Knight-Nanan D., Bowell R., Lanigan B., O'Keefe M. 1999. Intraocular lenses in children: changes in axial length, corneal curvature, and refraction. Br. J. Ophthalmol. 83, 265–269 10.1136/bjo.83.3.265 (doi:10.1136/bjo.83.3.265) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Yorston D., Yang Y. F., Sullivan P. M. 2005. Retinal detachment following surgery for congenital cataract: presentation and outcomes. Eye 19, 317–321 10.1038/sj.eye.6701463 (doi:10.1038/sj.eye.6701463) [DOI] [PubMed] [Google Scholar]
- 21.Sharma N., Balasubramanya R., Dada V. K., Vajpayee R. B. 2006. Efficacy of trypan blue in posterior capsulorhexis with optic capture in pediatric cataracts (ISRCTN48221688). BMC Ophthalmol. 6, 12. 10.1186/1471-2415-6-12 (doi:10.1186/1471-2415-6-12) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Findl O., Buehl W., Bauer P., Sycha T. 2010. Interventions for preventing posterior capsule opacification. Cochrane Database Syst. Rev. 2, CD003738. 10.1002/14651858.CD003738.pub3 (doi:10.1002/14651858.CD003738.pub3) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kugelberg M., Shafiei K., van der Ploeg I., Zetterström C. 2010. Intraocular lens as a drug delivery system for dexamethasone. Acta Ophthalmol. 88, 241–244 10.1111/j.1755-3768.2008.01419.x (doi:10.1111/j.1755-3768.2008.01419.x) [DOI] [PubMed] [Google Scholar]
- 24.Maloof A., Neilson G., Milverton E. J., Pandey S. K. 2003. Selective and specific targeting of lens epithelial cells during cataract surgery using sealed-capsule irrigation. J. Cataract Refract. Surg. 29, 1566–1568 10.1016/S0886-3350(03)00058-0 (doi:10.1016/S0886-3350(03)00058-0) [DOI] [PubMed] [Google Scholar]
- 25.Abdelwahab M. T., Kugelberg M., Seregard S., Zetterstrom C. 2007. Safety of irrigation with 5-fluorouracil in a sealed-capsule irrigation device in the rabbit eye. J. Cataract Refract. Surg. 33, 1619–1623 10.1016/j.jcrs.2007.05.012 (doi:10.1016/j.jcrs.2007.05.012) [DOI] [PubMed] [Google Scholar]
- 26.Simsek T., Mutluay A. H., Elgin U., Gursel R., Batman A. 2006. Glaucoma and increased central corneal thickness in aphakic and pseudophakic patients after congenital cataract surgery. Br. J. Ophthalmol. 90, 1103–1106 10.1136/bjo.2006.096370 (doi:10.1136/bjo.2006.096370) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wright K., Kolin T., Matsumoto E. 1995. Lens abnormalities. In Pediatric ophthalmology and strabismus (ed. Wright K. W.), pp. 367–387 St Louis: Mosby-Year Book, Inc [Google Scholar]
- 28.Wirth M. G., Russell-Eggitt I. M., Craig J. E., Elder J. E., Mackey D. A. 2002. Aetiology of congenital and paediatric cataract in an Australian population. Br. J. Ophthalmol. 86, 782–786 10.1136/bjo.86.7.782 (doi:10.1136/bjo.86.7.782) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Graw J. 2009. Mouse models for cataracts. J. Genet. 88, 469–486 10.1007/s12041-009-0066-2 (doi:10.1007/s12041-009-0066-2) [DOI] [PubMed] [Google Scholar]
- 30.Puk O., Dalke C., Calzada-Wack J., Ahmad N., Klaften M., Wagner S., Hrabé de Angelis M., Graw J. 2009. Reduced corneal thickness and enlarged anterior chamber in a novel ColVIIIa2G257D mutant mouse. Invest. Ophthalmol. Vis. Sci. 50, 5653–5661 10.1167/iovs.09-3550 (doi:10.1167/iovs.09-3550) [DOI] [PubMed] [Google Scholar]
- 31.Graw J. 2009. Genetics of crystallins: cataract and beyond. Exp. Eye Res. 88, 173–189 10.1016/j.exer.2008.10.011 (doi:10.1016/j.exer.2008.10.011) [DOI] [PubMed] [Google Scholar]
- 32.Graw J. 2003. The genetic and molecular basis of congenital eye defects. Nat. Rev. Genet. 4, 877–888 10.1038/nrg1202 (doi:10.1038/nrg1202) [DOI] [PubMed] [Google Scholar]
- 33.Okano Y., et al. 2001. A genetic factor for age-related cataract: identification and characterization of a novel galactokinase variant, ‘Osaka’, in Asians. Am. J. Hum. Genet. 68, 1036–1042 10.1086/319512 (doi:10.1086/319512) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kalaydjieva L., et al. 1999. A founder mutation in the GK1 gene is responsible for galactokinase deficiency in Roma (Gypsies). Am. J. Hum. Genet. 65, 1299–1307 10.1086/302611 (doi:10.1086/302611) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ai Y., et al. 2000. A mouse model of galactose-induced cataracts. Hum. Mol. Genet. 12, 1821–1827 10.1093/hmg/9.12.1821 (doi:10.1093/hmg/9.12.1821) [DOI] [PubMed] [Google Scholar]
- 36.Mörner C. T. 1893. Untersuchungen der Proteinsubstanzen in den lichtbrechenden Medien des Auges. Z. Physiol. Chem. 18, 61–106 [Google Scholar]
- 37.Cartier M., Breitman M. L., Tsui L. C. 1992. A frameshift mutation in the γ E-crystallin gene of the Elo mouse. Nat. Genet. 2, 42–45 10.1038/ng0992-42 (doi:10.1038/ng0992-42) [DOI] [PubMed] [Google Scholar]
- 38.Brakenhoff R. H., Aarts H. J. M., Reek F. H., Lubsen N. H., Schoenmakers J. G. G. 1990. Human γ-crystallin gene—a gene family on its way to extinction. J. Mol. Biol. 216, 519–532 10.1016/0022-2836(90)90380-5 (doi:10.1016/0022-2836(90)90380-5) [DOI] [PubMed] [Google Scholar]
- 39.Héon E., Priston M., Schorderet D. F., Billingsley G. D., Girard P. O., Lubsen N., Munier F. L. 1999. The γ-crystallins and human cataracts: a puzzle made clearer. Am. J. Hum. Genet. 65, 1261–1267 10.1086/302619 (doi:10.1086/302619) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Bloemendal H., de Jong W., Jaenicke R., Lubsen N. H., Slingsby C., Tardieu A. 2004. Ageing and vision: structure, stability and function of lens crystallins. Progr. Biophys. Mol. Biol. 86, 407–485 10.1016/j.pbiomolbio.2003.11.012 (doi:10.1016/j.pbiomolbio.2003.11.012) [DOI] [PubMed] [Google Scholar]
- 41.Sandilands A., et al. 2002. Altered aggregation properties of mutant γ-crystallins cause inherited cataract. EMBO J. 21, 6005–6014 10.1093/emboj/cdf609 (doi:10.1093/emboj/cdf609) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.van Rens G. L. M., Raats J. M. H., Driessen H. P. C., Oldenburg M., Wijnen J. T., Khan P. M., de Jong W. W., Bloemendal H. 1989. Structure of the bovine eye lens γs-crystallin gene (formerly βs). Gene 78, 225–233 10.1016/0378-1119(89)90225-4 (doi:10.1016/0378-1119(89)90225-4) [DOI] [PubMed] [Google Scholar]
- 43.Sun H., et al. 2005. Gamma-S crystallin gene (CRYGS) mutation causes dominant progressive cortical cataract in humans. J. Med. Genet. 42, 706–710 10.1136/jmg.2004.028274 (doi:10.1136/jmg.2004.028274) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Devi R. R., Yao W., Vijayalakshmi P., Sergeev Y. V., Sundaresan P., Hejtmancik J. F. 2008. Crystallin gene mutations in Indian families with inherited pediatric cataract. Mol. Vis. 14, 1157–1170 [PMC free article] [PubMed] [Google Scholar]
- 45.Sinha D., et al. 2001. A temperature-sensitive mutation of Crygs in the murine Opj cataract. J. Biol. Chem. 276, 9308–9315 10.1074/jbc.M010583200 (doi:10.1074/jbc.M010583200) [DOI] [PubMed] [Google Scholar]
- 46.Bu L., et al. 2002. The γS-crystallin gene is mutated in autosomal recessive cataract in mouse. Genomics 80, 38–44 10.1006/geno.2002.6803 (doi:10.1006/geno.2002.6803) [DOI] [PubMed] [Google Scholar]
- 47.Wistow G., et al. 2005. γN-crystallin and the evolution of the βγ-crystallin superfamily in vertebrates. FEBS J. 272, 2276–2291 10.1111/j.1742-4658.2005.04655.x (doi:10.1111/j.1742-4658.2005.04655.x) [DOI] [PubMed] [Google Scholar]
- 48.Magabo K. S., Horwitz J., Piatigorsky J., Kantorow M. 2000. Expression of βB2-crystallin mRNA and protein in retina, brain, and testis. Invest. Ophthalmol. Vis. Sci. 41, 3056–3060 [PMC free article] [PubMed] [Google Scholar]
- 49.DuPrey K. M., Robinson K. M., Wang Y., Taube J. R., Duncan M. K. 2007. Subfertility in mice harboring a mutation in βB2-crystallin. Mol. Vis. 13, 366–373 [PMC free article] [PubMed] [Google Scholar]
- 50.Ganguly K., et al. 2008. Novel allele of Crybb2 in the mouse and its expression in the brain. Invest. Ophthalmol. Vis. Sci. 49, 1533–1541 10.1167/iovs.07-0788 (doi:10.1167/iovs.07-0788) [DOI] [PubMed] [Google Scholar]
- 51.Litt M., Valenzuela C. R., Lamorticella D. M., Schultz D. W., Mitchell T. N., Kramer P., Maumenee I. H. 1997. Autosomal dominant cerulean cataract is associated with a chain termination mutation in the human β-crystallin gene CRYBB2. Hum. Mol. Genet. 6, 665–668 10.1093/hmg/6.5.665 (doi:10.1093/hmg/6.5.665) [DOI] [PubMed] [Google Scholar]
- 52.Vanita S. V., Reis A., Jung M., Singh D., Sperling K., Singh J. R., Bürger J. 2001. A unique form of autosomal dominant cataract explained by gene conversion between β-crystallin B2 and its pseudogene. J. Med. Genet. 38, 392–396 10.1136/jmg.38.6.392 (doi:10.1136/jmg.38.6.392) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Chambers C., Russell P. 1991. Deletion mutation in an eye lens β-crystallin. J. Biol. Chem. 266, 6742–6746 [PubMed] [Google Scholar]
- 54.Tréton J. A., Jacquemin E., Courtois Y., Jeanny J. C. 1991. Differential localization by in-situ hybridization of specific crystallin transcripts during mouse lens development. Differentiation 47, 143–147 10.1111/j.1432-0436.1991.tb00232.x (doi:10.1111/j.1432-0436.1991.tb00232.x) [DOI] [PubMed] [Google Scholar]
- 55.Robinson M. L., Overbeek P. A. 1996. Differential expression of αA- and αB-crystallin during murine ocular development. Invest. Ophthalmol. Vis. Sci. 37, 2276–2284 [PubMed] [Google Scholar]
- 56.Xi J., Farjo R., Yoshida S., Kern T. S., Swaroop A., Andley O. P. 2003. A comprehensive analysis of the expression of crystallins in mouse retina. Mol. Vis. 9, 410–419 [PubMed] [Google Scholar]
- 57.Brady J. P., Garland D., Duglas-Tabor Y., Robison W. G., Jr, Groome A., Wawrousek E. F. 1997. Targeted disruption of the mouse αA-crystallin gene induces cataract and cytoplasmic inclusion bodies containing the small heat shock protein αB-crystallin. Proc. Natl Acad. Sci. USA 94, 884–889 10.1073/pnas.94.3.884 (doi:10.1073/pnas.94.3.884) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Xia C. H., Liu H., Chang B., Cheng C., Cheung D., Wang M., Huang Q., Horwitz J., Gong X. 2006. Arginine 54 and Tyrosine 118 residues of αA-crystallin are crucial for lens formation and transparency. Invest. Ophthalmol. Vis. Sci. 47, 3004–3010 10.1167/iovs.06-0178 (doi:10.1167/iovs.06-0178) [DOI] [PubMed] [Google Scholar]
- 59.Graw J., Löster J., Soewarto D., Fuchs H., Meyer B., Reis A., Wolf E., Balling R., Hrabé de Angelis M. 2001. Characterization of a new, dominant V124E mutation in the mouse αA-crystallin. Invest. Ophthalmol. Vis. Sci. 42, 2909–2915 [PubMed] [Google Scholar]
- 60.Khan A. O., Aldahmesh M. A., Meyer B. 2007. Recessive congenital total cataract with microcornea and heterozygote carrier signs caused by a novel missense CRYAA mutation (R54C). Am. J. Ophthalmol. 144, 949–952 10.1016/j.ajo.2007.08.005 (doi:10.1016/j.ajo.2007.08.005) [DOI] [PubMed] [Google Scholar]
- 61.Pras E., Frydman M., Levy-Nissenbaum E., Bakhan T., Raz J., Assia E. I., Goldman B., Pras E. 2000. A nonsense mutation (W9X) in CRYAA causes autosomal recessive cataract in an inbred Jewish Persian family. Invest. Ophthalmol. Vis. Sci. 41, 3511–3515 [PubMed] [Google Scholar]
- 62.Brady J. P., Garland D. L., Green D. E., Tamm E. R., Giblin F. J., Wawrousek E. F. 2001. αB-crystallin in lens development and muscle integrity: a gene knockout approach. Invest. Ophthalmol. Vis. Sci. 42, 2924–2934 [PubMed] [Google Scholar]
- 63.Morrison L. E., Whittaker R. J., Klepper R. E., Wawrousek E. F., Glembotski C. C. 2004. Roles for αB-crystallin and HSPB2 in protecting the myocardium from ischemia-reperfusion-induced damage in a KO mouse model. Am. J. Physiol. Heart Circ. Physiol. 286, H847–H855 10.1152/ajpheart.00715.2003 (doi:10.1152/ajpheart.00715.2003) [DOI] [PubMed] [Google Scholar]
- 64.Müller M., Bhattacharya S. S., Moore T., Prescott Q., Weding T., Herrmann H., Magin T. M. 2009. Dominant cataract formation in association with vimentin assembly disrupting mutation. Hum. Mol. Genet. 18, 1052–1057 10.1093/hmg/ddn440 (doi:10.1093/hmg/ddn440) [DOI] [PubMed] [Google Scholar]
- 65.Colucci-Guyon E., Portier M.-M., Dunia I., Paulin D., Pournin S., Babinet C. 1994. Mice lacking vimentin develop and reproduce without an obvious phenotype. Cell 79, 679–694 10.1016/0092-8674(94)90553-3 (doi:10.1016/0092-8674(94)90553-3) [DOI] [PubMed] [Google Scholar]
- 66.Ramachandran R. D., Perumalsamy V., Hejtmancik J. F. 2007. Autosomal recessive juvenile onset cataract associated with mutation in BFSP1. Hum. Genet. 121, 475–482 10.1007/s00439-006-0319-6 (doi:10.1007/s00439-006-0319-6) [DOI] [PubMed] [Google Scholar]
- 67.Conley Y. P., et al. 2000. A juvenile-onset, progressive cataract locus on chromosome 3q21-q22 is associated with a missense mutation in the beaded filament structural protein-2. Am. J. Hum. Genet. 66, 1426–1431 10.1086/302871 (doi:10.1086/302871) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Jakobs P. M., Hess J. F., FitzGerald P. G., Kramer P., Weleber R. G., Litt M. 2000. Autosomal-dominant congenital cataract associated with a deletion mutation in the human beaded filament protein BFSP2. Am. J. Hum. Genet. 66, 1432–1436 10.1086/302872 (doi:10.1086/302872) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Sandilands A., Wang X., Hutcheson A. M., James J., Prescott A. R., Wegener A., Pekny M., Gong X., Quinlan R. A. 2004. Bfsp2 mutation found in mouse 129 strains causes the loss of CP49 and induces vimentin-dependent changes in the lens fibre cell cytoskeleton. Exp. Eye Res. 78, 875–889 10.1016/j.exer.2003.09.028 (doi:10.1016/j.exer.2003.09.028) [DOI] [PubMed] [Google Scholar]
- 70.Simirskii V. N., Lee R. S., Wawrousek E. F., Duncan M. K. 2006. Inbred FVB/N mice are mutant at the cp49/Bfsp2 locus and lack beaded filament proteins in the lens. Invest. Ophthalmol. Vis. Sci. 47, 4931–4934 10.1167/iovs.06-0423 (doi:10.1167/iovs.06-0423) [DOI] [PubMed] [Google Scholar]
- 71.Ramprasad V. L., Thool A., Murugan S., Nancarrow D., Vyas P., Rao S. K., Vidhya A., Ravishankar K., Kumaramanickavel G. 2005. Truncating mutation in the NHS gene: phenotypic heterogeneity of Nance–Horan Syndrome in an Asian Indian family. Invest. Ophthalmol. Vis. Sci. 46, 17–23 10.1167/iovs.04-0477 (doi:10.1167/iovs.04-0477) [DOI] [PubMed] [Google Scholar]
- 72.Florijn R. J., Loves W., de Buy Wenniger-Prick L. J. J. M. M., Mannens M. M. A. M., Tijmes N., Brooks S. P., Hardcastle A. J., Bergen A. A. 2006. New mutations in the NHS gene in Nance–Horan Syndrome families from the Netherlands. Eur. J. Hum. Genet. 14, 986–990 10.1038/sj.ejhg.5201671 (doi:10.1038/sj.ejhg.5201671) [DOI] [PubMed] [Google Scholar]
- 73.Huang K. M., Wu J., Duncan M. K., Moy C., Dutra A., Favor J., Da T., Stambolian D. 2006. Xcat, a novel mouse model for Nance-Horan syndrome inhibits expression of the cytoplasmic-targeted Nhs1 isoform. Hum. Mol. Genet. 15, 319–327 10.1093/hmg/ddi449 (doi:10.1093/hmg/ddi449) [DOI] [PubMed] [Google Scholar]
- 74.Stout C., Goddenough D. A., Paul D. L. 2004. Connexins: functions without junctions. Curr. Opin. Cell Biol. 16, 507–512 10.1016/j.ceb.2004.07.014 (doi:10.1016/j.ceb.2004.07.014) [DOI] [PubMed] [Google Scholar]
- 75.Yancey S. B., Biswal S., Revel P. 1992. Spatial and temporal patterns of distribution of the gap junction protein connexin43 during mouse gastrulation and organogenesis. Development 114, 203–212 [DOI] [PubMed] [Google Scholar]
- 76.Paul D. L., Ebihara L., Takemoto L. J., Swenson K. I., Goodenough D. A. 1991. Connexin46, a novel lens gap junction protein, induces voltage-gated currents in nonjunctional plasma membrane of Xenopus oocytes. J. Cell Biol. 115, 1077–1089 10.1083/jcb.115.4.1077 (doi:10.1083/jcb.115.4.1077) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Gong X., Li E., Klier G., Huang Q., Wu Y., Lei H., Kumar N. M., Horwitz J., Gilula N. B. 1997. Disruption of α3 connexin gene leads to proteolysis and cataractogenesis in mice. Cell 91, 833–843 10.1016/S0092-8674(00)80471-7 (doi:10.1016/S0092-8674(00)80471-7) [DOI] [PubMed] [Google Scholar]
- 78.Gong X., Agopian K., Kumar N. M., Gilula N. B. 1999. Genetic factors influence cataract formation in α3 connexin knockout mice. Dev. Genet. 24, 27–32 (doi:10.1002/(SICI)1520-6408(1999)24:1/2<27::AID-DVG4>3.0.CO;2-7) [DOI] [PubMed] [Google Scholar]
- 79.Baruch A., Greenbaum D., Levy E. T., Nielsen P. A., Gilula N. B., Kumar N. M., Bogyo M. 2001. Defining a link between jap junction communication, proteolysis, and cataract formation. J. Biol. Chem. 276, 28 999–29 006 10.1074/jbc.M103628200 (doi:10.1074/jbc.M103628200) [DOI] [PubMed] [Google Scholar]
- 80.Tang Y., Liu X., Zoltoski R. K., Novak L. A., Herrera R. A., Richard I., Kuszak J. R., Kumar N. M. 2007. Age-related cataracts in α3Cx46-knockout mice are dependent on a calpain 3 isoform. Invest. Ophthalmol. Vis. Sci. 48, 2685–2694 10.1167/iovs.06-0926 (doi:10.1167/iovs.06-0926) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.White T. W., Gao Y., Li L., Sellitto C., Srinivas M. 2007. Optimal lens epithelial cell proliferation is dependent on the connexin isoform providing gap junctional coupling. Invest. Ophthalmol. Vis. Sci. 48, 5630–5637 10.1167/iovs.06-1540 (doi:10.1167/iovs.06-1540) [DOI] [PubMed] [Google Scholar]
- 82.Polyakov A. V., Shagina I. A., Khlebnikova O. V., Evgrafov O. V. 2001. Mutation in the connexin 50 gene (GJA8) in a Russian family with zonular pulverulent cataract. Clin. Genet. 60, 476–478 10.1034/j.1399-0004.2001.600614.x (doi:10.1034/j.1399-0004.2001.600614.x) [DOI] [PubMed] [Google Scholar]
- 83.Graw J., et al. 2009. The GJA8 allele encoding CX50I247M is a rare polymorphism, not a cataract-causing mutation. Mol. Vis. 14, 1881–1885 [PMC free article] [PubMed] [Google Scholar]
- 84.DeRosa A. M., Meşe G., Li L., Sellitto C., Brink P. R., Gong X., White T. W. 2009. The cataract causing Cx50-S50P mutant inhibits Cx43 and intercellular communication in the lens epithelium. Exp. Cell Res. 315, 1063–1075 10.1016/j.yexcr.2009.01.017 (doi:10.1016/j.yexcr.2009.01.017) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Varadaraj K., Kumari S. S., Mathias R. T. 2007. Functional expression of aquaporins in embryonic, postnatal, and adult mouse lenses. Dev. Dyn. 236, 1319–1328 10.1002/dvdy.21125 (doi:10.1002/dvdy.21125) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Berry V., Francis P., Kaushal S., Moore A., Bhattacharya S. 2000. Missense mutations in MIP underlie autosomal dominant ‘polymorphic’ and lamellar cataracts linked to 12q. Nat. Genet. 25, 15–17 10.1038/75538 (doi:10.1038/75538) [DOI] [PubMed] [Google Scholar]
- 87.Fraser F. C., Schabtach G. 1962. ‘Shrivelled’: a hereditary degeneration of the lens in the house mouse. Genet. Res. 3, 383–387 10.1017/S0016672300003220 (doi:10.1017/S0016672300003220) [DOI] [Google Scholar]
- 88.Pras E., Levy-Nissenbaum E., Bakhan T., Lahat H., Assia E., Geffen-Carmi N., Frydman M., Goldman B., Pras E. 2002. A missense mutation in the LIM2 gene is associated with autosomal recessive presenile cataract in an inbred Iraqi Jewish family. Am. J. Hum. Genet. 70, 1363–1367 10.1086/340318 (doi:10.1086/340318) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Ponnam S. P. G., Ramesha K., Tejwani S., Matalia J., Kannabiran C. 2008. A missense mutation in LIM2 causes autosomal recessive congenital cataract. Mol. Vis. 14, 1204–1208 [PMC free article] [PubMed] [Google Scholar]
- 90.Zhou L., Chen T., Church R. L. 2002. Temporal expression of three mouse lens fiber cell membrane protein genes during early development. Mol. Vis. 8, 143–148 [PubMed] [Google Scholar]
- 91.Hever A. M., Williamson K. A., van Heyningen V. 2006. Developmental malformations of the eye: the role of PAX6, SOX2 and OTX2. Clin. Genet. 69, 459–470 10.1111/j.1399-0004.2006.00619.x (doi:10.1111/j.1399-0004.2006.00619.x) [DOI] [PubMed] [Google Scholar]
- 92.Davis L. K., Meyer K. J., Rudd D. S., Librant A. L., Epping E. A., Sheffield V. C., Wassink T. H. 2008. Pax6 3′ deletion results in aniridia, autism and mental retardation. Hum. Genet. 123, 371–378 10.1007/s00439-008-0484-x (doi:10.1007/s00439-008-0484-x) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Tsonis P. A., Fuentes E. J. 2006. Focus on molecules: Pax-6, the eye master. Exp. Eye Res. 83, 233–234 10.1016/j.exer.2005.11.019 (doi:10.1016/j.exer.2005.11.019) [DOI] [PubMed] [Google Scholar]
- 94.Sander M., Neubüser A., Kalamaras J., Ee H. C., Martin G. R., German M. S. 1997. Genetic analysis reveals that PAX6 is required for normal transcription of pancreatic hormone genes and islet development. Genes Dev. 11, 1662–1673 10.1101/gad.11.13.1662 (doi:10.1101/gad.11.13.1662) [DOI] [PubMed] [Google Scholar]
- 95.Favor J., Gloeckner C. J., Neuhäuser-Klaus A., Pretsch W., Sandulache R., Saule S., Zaus I. 2008. Relationship of Pax6 activity levels to the extent of eye development in the mouse, Mus musculus. Genetics 179, 1345–1355 10.1534/genetics.108.088591 (doi:10.1534/genetics.108.088591) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Semina E. V., Ferrell R. E., Mintz-Hittner H. A., Bitoun P., Alward W. L. M., Reiter R. S., Funkhauser C., Daack-Hirsch S., Murray J. C. 1998. A novel homeobox gene PITX3 is mutated in families with autosomal-dominant cataracts and ASMD. Nat. Genet. 19, 167–170 10.1038/527 (doi:10.1038/527) [DOI] [PubMed] [Google Scholar]
- 97.Bidinost C., et al. 2006. Heterozygous and homozygous mutations in PITX3 in a large Lebanese family with posterior polar cataracts and neurodevelopmental abnormalities. Invest. Ophthalmol. Vis. Sci. 47, 1274–1280 10.1167/iovs.05-1095 (doi:10.1167/iovs.05-1095) [DOI] [PubMed] [Google Scholar]
- 98.Rosemann M., et al. 2010. Microphthalmia, Parkinsonism and enhanced nociception in Pitx3416insG mice. Mamm. Genome 21, 13–27 10.1007/s00335-009-9235-0 (doi:10.1007/s00335-009-9235-0) [DOI] [PubMed] [Google Scholar]
- 99.Smidt M. P., Smits S. M., Bouwmeester H., Hamers F. P. T., van der Linden A. J. A., Hellemons A. J. C. G. M., Graw J., Burbach J. P. 2004. Early developmental failure of substantia nigra dopamine neurons in mice lacking the homeodomain gene Pitx3. Development 131, 1145–1155 10.1242/dev.01022 (doi:10.1242/dev.01022) [DOI] [PubMed] [Google Scholar]
- 100.Fuchs J., et al. 2009. The transcription factor PITX3 is associated with sporadic Parkinson's disease. Neurobiol. Aging 30, 731–738 10.1016/j.neurobiolaging.2007.08.014 (doi:10.1016/j.neurobiolaging.2007.08.014) [DOI] [PubMed] [Google Scholar]
- 101.Semina E. V., Brownell I., Mintz-Hittner H. A., Murray J. C., Jamrich M. 2001. Mutations in the human forkhead transcription factor FOXE3 associated with anterior segment ocular dysgenesis and cataracts. Hum. Mol. Genet. 10, 231–236 10.1093/hmg/10.3.231 (doi:10.1093/hmg/10.3.231) [DOI] [PubMed] [Google Scholar]
- 102.Valleix S., Niel F., Nedelec B., Algros M. P., Schwartz C., Delbosc B., Delpech M., Kantelip B. 2006. Homozygous nonsense mutation in the FOXE3 gene as a cause of congenital primary aphakia in humans. Am. J. Hum. Genet. 79, 358–364 10.1086/505654 (doi:10.1086/505654) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Nishimura D. Y., et al. 2001. A spectrum of FOXC1 mutations suggests gene dosage as a mechanism for developmental defects of the anterior chamber of the eye. Am. J. Hum. Genet. 68, 364–372 10.1086/318183 (doi:10.1086/318183) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Hong H. K., Lass J. H., Chakravarti A. 1999. Pleiotropic skeletal and ocular phenotypes of the mouse mutation congenital hydrocephalus (ch/Mf1) arise from a winged helix/forkhead transcription factor gene. Hum. Mol. Genet. 8, 625–637 10.1093/hmg/8.4.625 (doi:10.1093/hmg/8.4.625) [DOI] [PubMed] [Google Scholar]
- 105.Dickie M. M. 1967. Vacuolated lens. Mouse News Lett. 36, 39–40 [Google Scholar]
- 106.Matteson P. G., et al. 2008. The orphan G protein-coupled receptor, Gpr161, encodes the vacuolated lens locus and controls neurulation and lens development. Proc. Natl Acad. Sci. USA 105, 2088–2093 10.1073/pnas.0705657105 (doi:10.1073/pnas.0705657105) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Nishizawa M., Kataoka K., Goto N., Fujiwara K. T., Kawai S. 1989. v-maf, a viral oncogene that encodes a ‘leucine zipper’ motif. Proc. Natl Acad. Sci. USA 86, 7711–7715 10.1073/pnas.86.20.7711 (doi:10.1073/pnas.86.20.7711) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Jamieson R. V., et al. 2002. Domain disruption and mutation of the bZIP transcription factor, MAF, associated with cataract, ocular anterior segment dysgenesis and coloboma. Hum. Mol. Genet. 11, 33–42 10.1093/hmg/11.1.33 (doi:10.1093/hmg/11.1.33) [DOI] [PubMed] [Google Scholar]
- 109.Bu L., et al. 2002. Mutant DNA-binding domain of HSF4 is associated with autosomal dominant lamellar and Marner cataract. Nat. Genet. 31, 276–278 10.1038/ng921 (doi:10.1038/ng921) [DOI] [PubMed] [Google Scholar]
- 110.Smaoui N., Beltaief O., BenHamed S., M'Rad R., Maazoul F., Ouertani A., Chaabouni H., Hejtmancik J. F. 2004. A homozygous splice mutation in the HSF4 gene is associated with an autosomal recessive congenital cataract. Invest. Ophthalmol. Vis. Sci. 45, 2716–2721 10.1167/iovs.03-1370 (doi:10.1167/iovs.03-1370) [DOI] [PubMed] [Google Scholar]
- 111.Forshew T., et al. 2005. Locus heterogeneity in autosomal recessive congenital cataracts: linkage to 9q and germline HSF4 mutations. Hum. Genet. 117, 452–459 10.1007/s00439-005-1309-9 (doi:10.1007/s00439-005-1309-9) [DOI] [PubMed] [Google Scholar]
- 112.Ke T., Wang Q. K., Ji B., Wang X., Liu P., Zhang X., Tang Z., Ren X., Liu M. 2006. Novel HSF4 mutation causes congenital total white cataract in a Chinese family. Am. J. Ophthalmol. 142, 298–303 10.1016/j.ajo.2006.03.056 (doi:10.1016/j.ajo.2006.03.056) [DOI] [PubMed] [Google Scholar]
- 113.Shi Y., et al. 2008. Mutation screening of HSF4 in 150 age-related cataract patients. Mol. Vis. 14, 1850–1855 [PMC free article] [PubMed] [Google Scholar]
- 114.Sajjad N., Goebel I., Kakar N., Cheema A. M., Kubisch C., Ahmad J. 2008. A novel HSF4 gene mutation (p. R405X) causing autosomal recessive congenital cataracts in a large consanguineous family from Pakistan. BMC Med. Genet. 9, 99. 10.1186/147I-2350-9-99 (doi:10.1186/147I-2350-9-99) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Hansen L., Mikkelsen A., Nürnberg P., Nürnberg G., Anjum I., Eiberg H., Rosenberg T. 2009. Comprehensive mutational screening in a cohort of Danish families with hereditary congenital cataract. Invest. Ophthalmol. Vis. Sci. 50, 3291–3303 10.1167/iovs.08-3149 (doi:10.1167/iovs.08-3149) [DOI] [PubMed] [Google Scholar]
- 116.Shi X., Cui B., Wang Z., Weng L., Xu Z., Ma J., Xu G., Kong X., Hu L. 2009. Removal of Hsf4 leads to cataract development in mice through down-regulation of γS-crystallin and Bfsp expression. BMC Mol. Biol. 10, 10. 10.1186/1471-2199-10-10 (doi:10.1186/1471-2199-10-10) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Cooper M. A., Son A. I., Komlos D., Sun Y., Kleiman N. J., Zhou R. 2008. Loss of ephrin-A5 function disrupts lens fiber cell packing and leads to catarat. Proc. Natl Acad. Sci. USA 105, 16 620–16 625 10.1073/pnas.0808987105 (doi:10.1073/pnas.0808987105) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Shiels A., Bennett T. M., Knopf H. L. S., Maraini G., Li A., Jiao X., Hejtmancik J. F. 2008. The EPHA2 gene is associated with cataracts linked to chromosome 1p. Mol. Vis. 14, 2042–2055 [PMC free article] [PubMed] [Google Scholar]
- 119.Zhang T., et al. 2009. Mutations of the EPHA2 receptor tyrosine kinase gene cause autosomal dominant congenital cataract. Hum. Mutat. (Mutation in brief no. 1059) 30, E603–E611 [DOI] [PubMed] [Google Scholar]
- 120.Jun G., et al. 2009. EPHA2 is associated with age-related cortical cataract in mice and humans. PLoS Genet. 5, e1000584. 10.1371/journal.pgen.1000584 (doi:10.1371/journal.pgen.1000584) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Cooper M. A., Kobayashi K., Zhou R. 2009. Ephrin-A5 regulates the formation of the ascending midbrain dopaminergic pathways. Develop. Neurobiol. 69, 36–46 10.1002/dneu.20685 (doi:10.1002/dneu.20685) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Jiang H., Jin Y., Bu L., Zhang W., Liu J., Cui B., Kong X., Hu L. 2003. A novel mutation in GJA3 (connexin46) for autosomal dominant congenital nuclear pulverulent cataract. Mol. Vis. 9, 579–583 [PubMed] [Google Scholar]
- 123.Lu S., Zhao C., Jiao H., Kere J., Tang X., Zhao F., Zhang X., Zhao K., Larsson C. 2007. Two Chinese families with pulverulent congenital cataracts and ΔG91 CRYBA1 mutations. Mol. Vis. 13, 1154–1160 [PubMed] [Google Scholar]
- 124.Jamieson R. V., Munier F., Balmer A., Farrar N., Perveen R., Black G. C. 2003. Pulverulent cataract with variably associated microcornea and iris coloboma in a MAF mutation family. Br. J. Ophthalmol. 87, 411–412 10.1136/bjo.87.4.411 (doi:10.1136/bjo.87.4.411) [DOI] [PMC free article] [PubMed] [Google Scholar]