Abstract
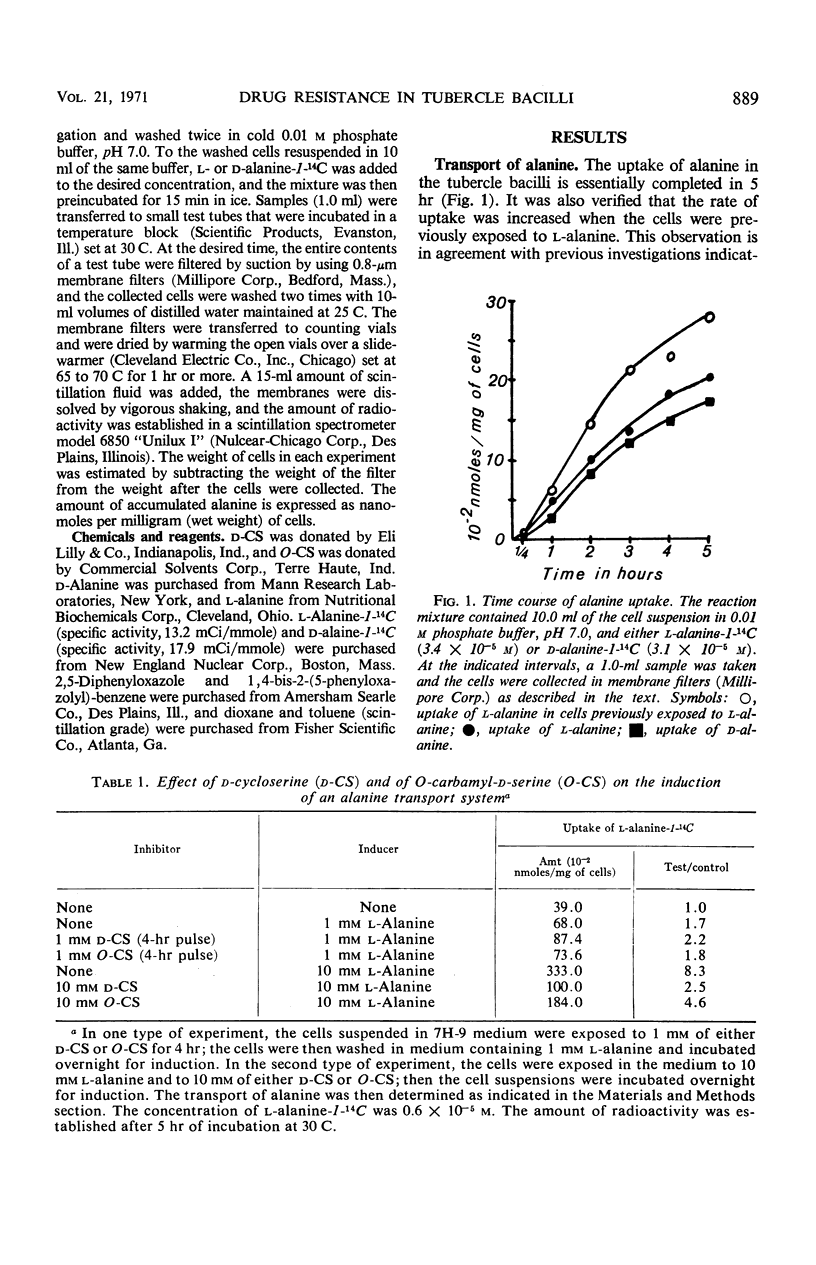
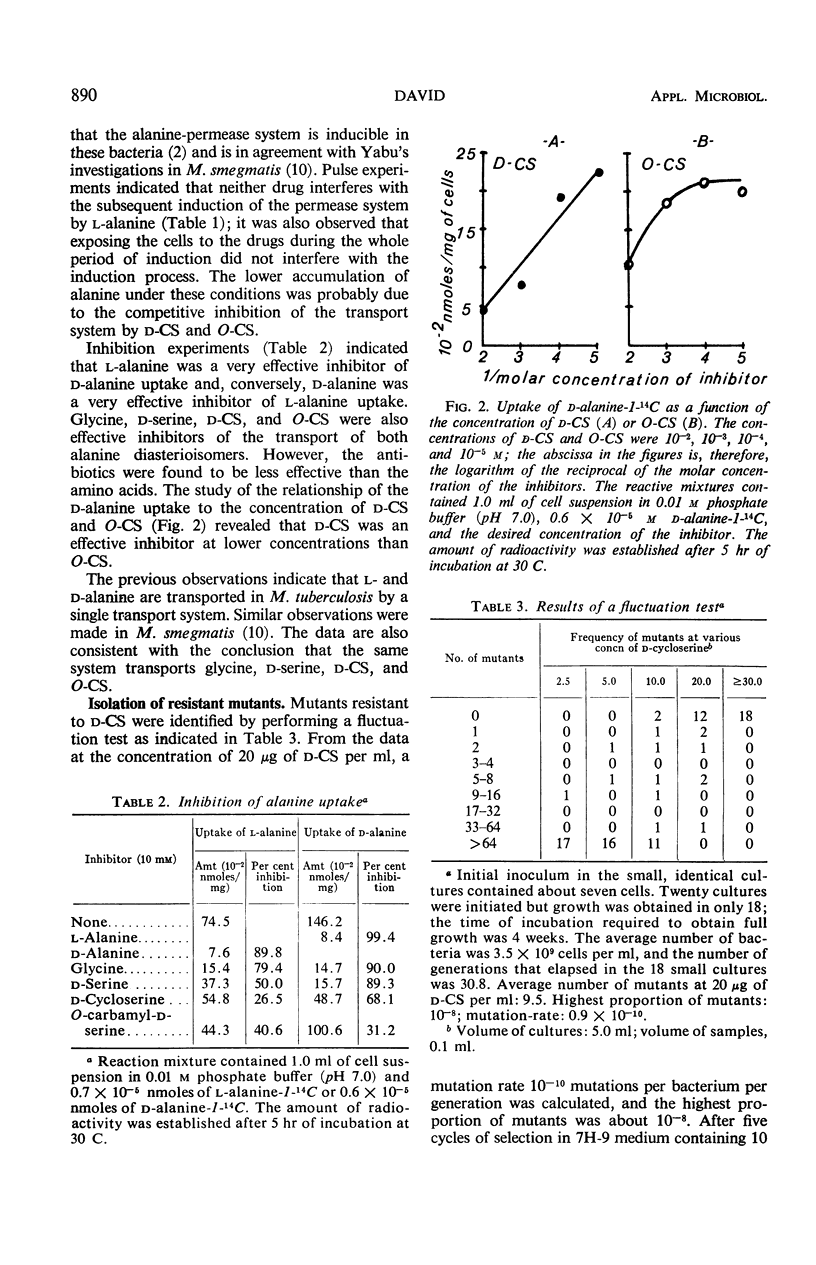
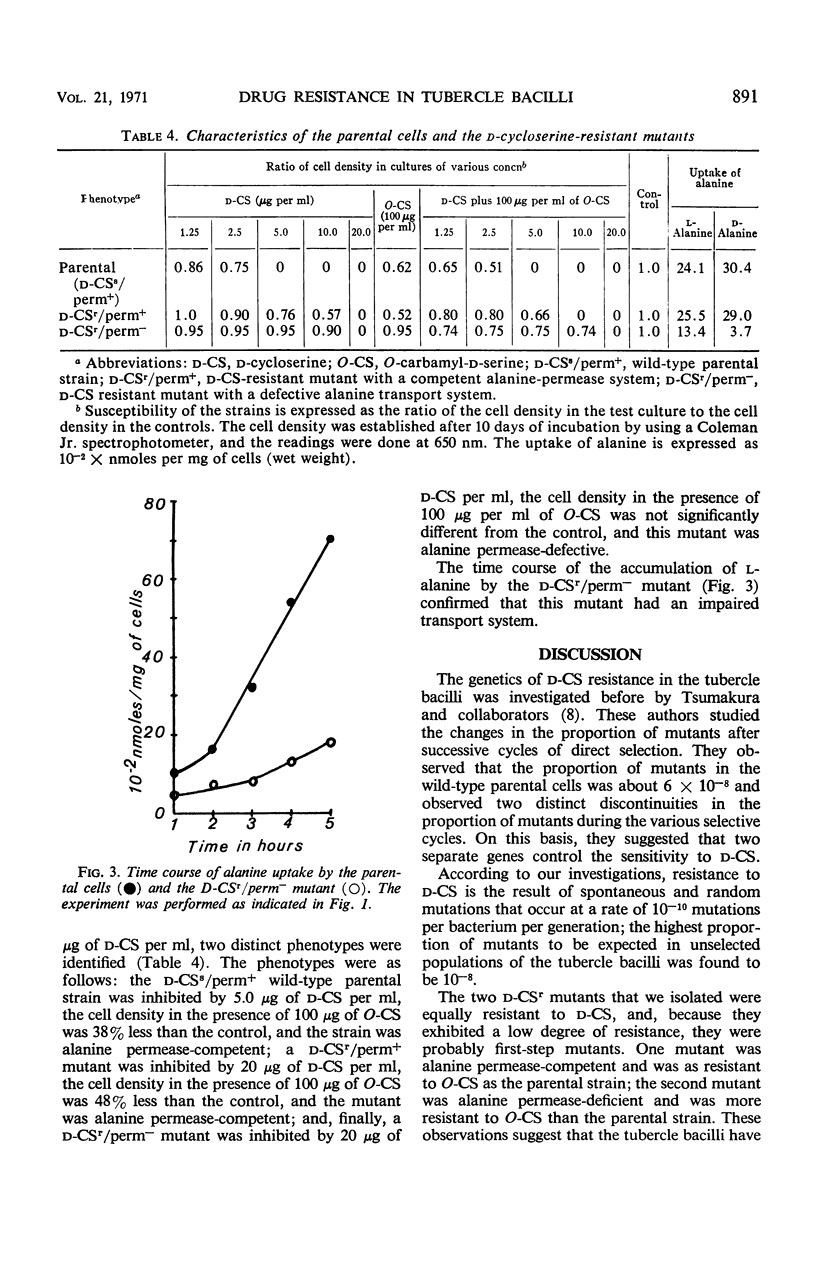
A single transport system was found to accumulate l- and d-alanine, glycine and d-serine in Mycobacterium tuberculosis. The results of inhibition experiments suggested that the antibiotics d-cycloserine and O-carbamyl-d-serine were also transported by the alanine-glycine-d-serine system. A d-cycloserine-resistant permease-competent (d-CSr/perm+) mutant and a d-cycloserine-resistant permease-defective (d-CSr/perm−) mutant were isolated. The d-CSr/perm− mutant was not found to be more resistant to the drug than was the d-CSr/perm+ mutant. The data were consistent with the conclusion that resistance to d-cycloserine in the tubercle bacilli is primarily due to mutations in the gene(s) controlling the enzyme d-alanyl-d-alanine synthetase. The mutation rate was calculated to be about 10−10 mutations per bacterium per generation.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Curtiss R., 3rd, Charamella L. J., Berg C. M., Harris P. E. Kinetic and genetic analyses of D-cycloserine inhibition and resistance in Escherichia coli. J Bacteriol. 1965 Nov;90(5):1238–1250. doi: 10.1128/jb.90.5.1238-1250.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- David H. L. Effect of O-carbamyl-D-serine on the growth of mycobacterium tuberculosis. Am Rev Respir Dis. 1970 Jul;102(1):68–74. doi: 10.1164/arrd.1970.102.1.68. [DOI] [PubMed] [Google Scholar]
- David H. L. Probability distribution of drug-resistant mutants in unselected populations of Mycobacterium tuberculosis. Appl Microbiol. 1970 Nov;20(5):810–814. doi: 10.1128/am.20.5.810-814.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- David H. L., Takayama K., Goldman D. S. Susceptibility of mycobacterial D-alanyl-D-alanine synthetase to D-cycloserine. Am Rev Respir Dis. 1969 Oct;100(4):579–581. doi: 10.1164/arrd.1969.100.4.579. [DOI] [PubMed] [Google Scholar]
- Luria S. E., Delbrück M. Mutations of Bacteria from Virus Sensitivity to Virus Resistance. Genetics. 1943 Nov;28(6):491–511. doi: 10.1093/genetics/28.6.491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reitz R. H., Slade H. D., Neuhaus F. C. The biochemical mechanisms of resistance by streptococci to the antibiotics D-cycloserine and O-carbamyl-D-serine. Biochemistry. 1967 Aug;6(8):2561–2570. doi: 10.1021/bi00860a038. [DOI] [PubMed] [Google Scholar]
- TANAKA N., UMEZAWA H. SYNERGISM OF D-4-AMINO-3-ISOXAZOLIDONE AND O-CARBAMYL-D-SERINE. J Antibiot (Tokyo) 1964 Jan;17:8–10. [PubMed] [Google Scholar]
- TSUKAMURA M., NODA Y., HAYASHI M., YAMAMOTO M. A genetic study on the cycloserine-resistance of Mycobacterium tuberculosis var. hominis. Jpn J Microbiol. 1959 Jan;3:1–8. doi: 10.1111/j.1348-0421.1959.tb00095.x. [DOI] [PubMed] [Google Scholar]
- Wargel R. J., Shadur C. A., Neuhaus F. C. Mechanism of D-cycloserine action: transport systems for D-alanine, D-cycloserine, L-alanine, and glycine. J Bacteriol. 1970 Sep;103(3):778–788. doi: 10.1128/jb.103.3.778-788.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yabu K. Amino acid transport in Mycobacterium smegmatis. J Bacteriol. 1970 Apr;102(1):6–13. doi: 10.1128/jb.102.1.6-13.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]


