Abstract
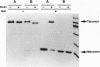
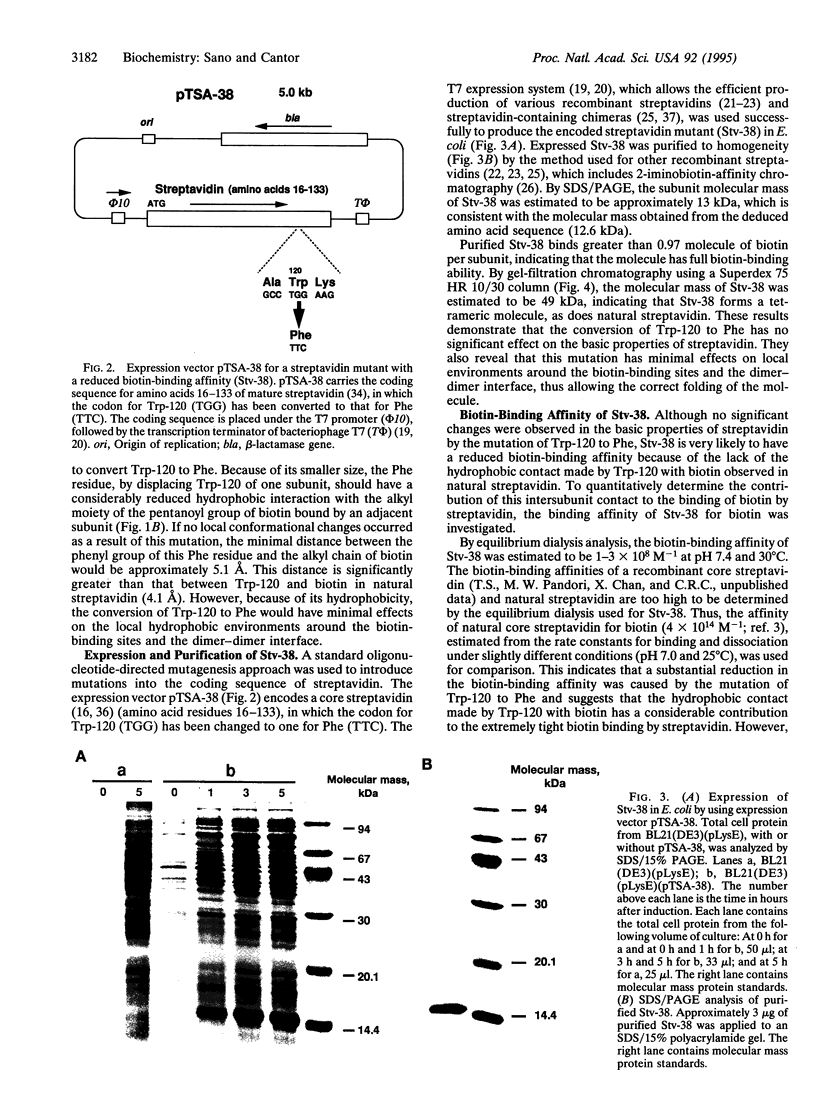
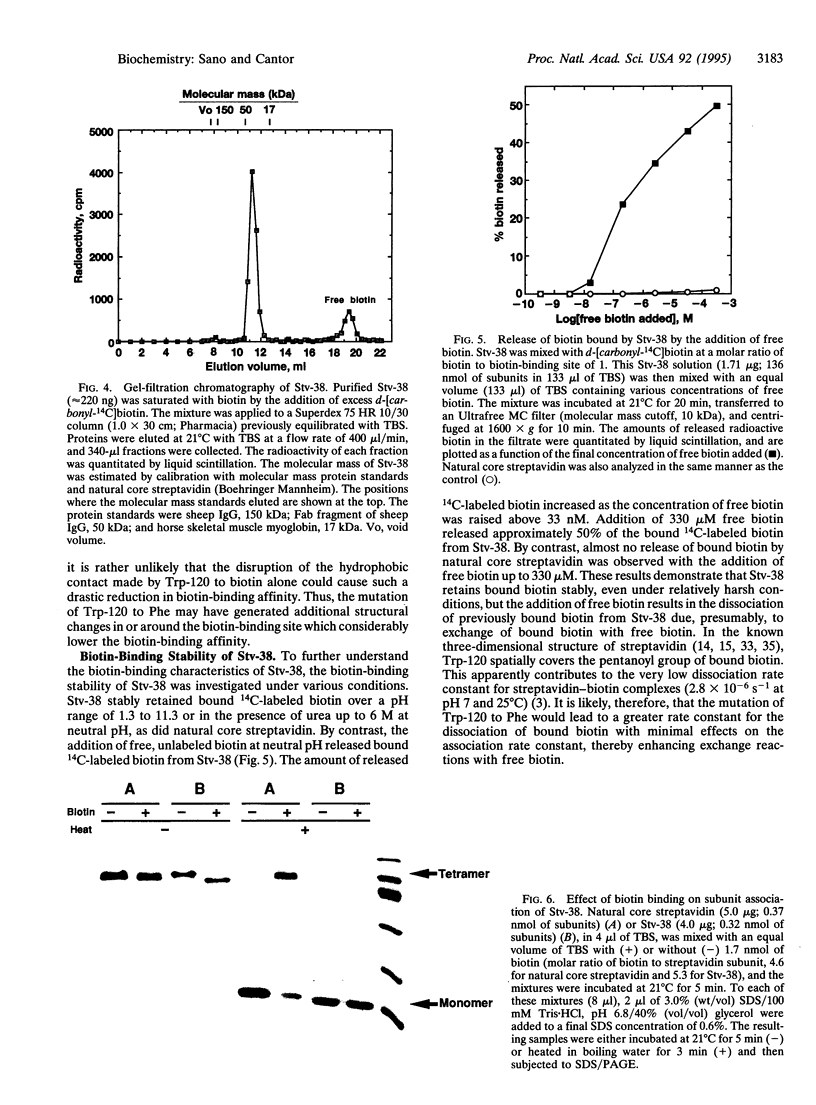
In natural streptavidin, tryptophan 120 of each subunit makes contacts with the biotin bound by an adjacent subunit through the dimer-dimer interface. To understand quantitatively the role of tryptophan 120 and its intersubunit communication in the properties of streptavidin, a streptavidin mutant in which tryptophan 120 is converted to phenylalanine was produced and characterized. The streptavidin mutant forms a tetrameric molecule and binds one biotin per subunit, as does natural streptavidin, indicating that the mutation of tryptophan 120 to phenylalanine has no significant effect on the basic properties of streptavidin. However, its biotin-binding affinity was reduced substantially, to approximately 10(8) M-1, indicating that the contact made by tryptophan 120 to biotin has a considerable contribution to the extremely tight biotin binding by streptavidin. The mutant retained bound biotin over a wide pH range or with the addition of urea up to 6 M at neutral pH. However, bound biotin was efficiently released by the addition of excess free biotin due, presumably, to exchange reactions. Electrophoretic analysis revealed that the intersubunit contact made by tryptophan 120 to biotin through the dimer-dimer interface is the major interaction responsible for the biotin-induced, tighter subunit association of streptavidin. In addition, the mutant has weaker subunit association than natural streptavidin even in the absence of biotin, indicating that tryptophan 120 also contributes to the subunit association of tetramers in the absence of biotin.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Argaraña C. E., Kuntz I. D., Birken S., Axel R., Cantor C. R. Molecular cloning and nucleotide sequence of the streptavidin gene. Nucleic Acids Res. 1986 Feb 25;14(4):1871–1882. doi: 10.1093/nar/14.4.1871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bayer E. A., Ben-Hur H., Hiller Y., Wilchek M. Postsecretory modifications of streptavidin. Biochem J. 1989 Apr 15;259(2):369–376. doi: 10.1042/bj2590369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blankenburg R., Meller P., Ringsdorf H., Salesse C. Interaction between biotin lipids and streptavidin in monolayers: formation of oriented two-dimensional protein domains induced by surface recognition. Biochemistry. 1989 Oct 3;28(20):8214–8221. doi: 10.1021/bi00446a037. [DOI] [PubMed] [Google Scholar]
- CHAIET L., MILLER T. W., TAUSIG F., WOLF F. J. ANTIBIOTIC MSD-235. II. SEPARATION AND PURIFICATION OF SYNERGISTIC COMPONENTS. Antimicrob Agents Chemother (Bethesda) 1963;161:28–32. [PubMed] [Google Scholar]
- CHAIET L., WOLF F. J. THE PROPERTIES OF STREPTAVIDIN, A BIOTIN-BINDING PROTEIN PRODUCED BY STREPTOMYCETES. Arch Biochem Biophys. 1964 Jul 20;106:1–5. doi: 10.1016/0003-9861(64)90150-x. [DOI] [PubMed] [Google Scholar]
- Florin E. L., Moy V. T., Gaub H. E. Adhesion forces between individual ligand-receptor pairs. Science. 1994 Apr 15;264(5157):415–417. doi: 10.1126/science.8153628. [DOI] [PubMed] [Google Scholar]
- Gitlin G., Bayer E. A., Wilchek M. Studies on the biotin-binding site of streptavidin. Tryptophan residues involved in the active site. Biochem J. 1988 Nov 15;256(1):279–282. doi: 10.1042/bj2560279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Green N. M. Avidin and streptavidin. Methods Enzymol. 1990;184:51–67. doi: 10.1016/0076-6879(90)84259-j. [DOI] [PubMed] [Google Scholar]
- Helm C. A., Knoll W., Israelachvili J. N. Measurement of ligand-receptor interactions. Proc Natl Acad Sci U S A. 1991 Sep 15;88(18):8169–8173. doi: 10.1073/pnas.88.18.8169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hendrickson W. A., Pähler A., Smith J. L., Satow Y., Merritt E. A., Phizackerley R. P. Crystal structure of core streptavidin determined from multiwavelength anomalous diffraction of synchrotron radiation. Proc Natl Acad Sci U S A. 1989 Apr;86(7):2190–2194. doi: 10.1073/pnas.86.7.2190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hofmann K., Wood S. W., Brinton C. C., Montibeller J. A., Finn F. M. Iminobiotin affinity columns and their application to retrieval of streptavidin. Proc Natl Acad Sci U S A. 1980 Aug;77(8):4666–4668. doi: 10.1073/pnas.77.8.4666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuriyan J., Weis W. I. Rigid protein motion as a model for crystallographic temperature factors. Proc Natl Acad Sci U S A. 1991 Apr 1;88(7):2773–2777. doi: 10.1073/pnas.88.7.2773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurzban G. P., Gitlin G., Bayer E. A., Wilchek M., Horowitz P. M. Biotin binding changes the conformation and decreases tryptophan accessibility of streptavidin. J Protein Chem. 1990 Dec;9(6):673–682. doi: 10.1007/BF01024762. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Leckband D. E., Schmitt F. J., Israelachvili J. N., Knoll W. Direct force measurements of specific and nonspecific protein interactions. Biochemistry. 1994 Apr 19;33(15):4611–4624. doi: 10.1021/bi00181a023. [DOI] [PubMed] [Google Scholar]
- Livnah O., Bayer E. A., Wilchek M., Sussman J. L. The structure of the complex between avidin and the dye, 2-(4'-hydroxyazobenzene) benzoic acid (HABA). FEBS Lett. 1993 Aug 9;328(1-2):165–168. doi: 10.1016/0014-5793(93)80986-5. [DOI] [PubMed] [Google Scholar]
- Livnah O., Bayer E. A., Wilchek M., Sussman J. L. Three-dimensional structures of avidin and the avidin-biotin complex. Proc Natl Acad Sci U S A. 1993 Jun 1;90(11):5076–5080. doi: 10.1073/pnas.90.11.5076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyamoto S., Kollman P. A. Absolute and relative binding free energy calculations of the interaction of biotin and its analogs with streptavidin using molecular dynamics/free energy perturbation approaches. Proteins. 1993 Jul;16(3):226–245. doi: 10.1002/prot.340160303. [DOI] [PubMed] [Google Scholar]
- Moy V. T., Florin E. L., Gaub H. E. Intermolecular forces and energies between ligands and receptors. Science. 1994 Oct 14;266(5183):257–259. doi: 10.1126/science.7939660. [DOI] [PubMed] [Google Scholar]
- Pähler A., Hendrickson W. A., Kolks M. A., Argaraña C. E., Cantor C. R. Characterization and crystallization of core streptavidin. J Biol Chem. 1987 Oct 15;262(29):13933–13937. [PubMed] [Google Scholar]
- Sano T., Cantor C. R. Expression of a cloned streptavidin gene in Escherichia coli. Proc Natl Acad Sci U S A. 1990 Jan;87(1):142–146. doi: 10.1073/pnas.87.1.142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sano T., Cantor C. R. Expression vectors for streptavidin-containing chimeric proteins. Biochem Biophys Res Commun. 1991 Apr 30;176(2):571–577. doi: 10.1016/s0006-291x(05)80222-0. [DOI] [PubMed] [Google Scholar]
- Sano T., Glazer A. N., Cantor C. R. A streptavidin-metallothionein chimera that allows specific labeling of biological materials with many different heavy metal ions. Proc Natl Acad Sci U S A. 1992 Mar 1;89(5):1534–1538. doi: 10.1073/pnas.89.5.1534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sano T., Smith C. L., Cantor C. R. A streptavidin mutant containing a cysteine stretch that facilitates production of a variety of specific streptavidin conjugates. Biotechnology (N Y) 1993 Feb;11(2):201–206. doi: 10.1038/nbt0293-201. [DOI] [PubMed] [Google Scholar]
- Sayers J. R., Krekel C., Eckstein F. Rapid high-efficiency site-directed mutagenesis by the phosphorothioate approach. Biotechniques. 1992 Oct;13(4):592–596. [PubMed] [Google Scholar]
- Studier F. W., Moffatt B. A. Use of bacteriophage T7 RNA polymerase to direct selective high-level expression of cloned genes. J Mol Biol. 1986 May 5;189(1):113–130. doi: 10.1016/0022-2836(86)90385-2. [DOI] [PubMed] [Google Scholar]
- Studier F. W., Rosenberg A. H., Dunn J. J., Dubendorff J. W. Use of T7 RNA polymerase to direct expression of cloned genes. Methods Enzymol. 1990;185:60–89. doi: 10.1016/0076-6879(90)85008-c. [DOI] [PubMed] [Google Scholar]
- Vajda S., Weng Z., Rosenfeld R., DeLisi C. Effect of conformational flexibility and solvation on receptor-ligand binding free energies. Biochemistry. 1994 Nov 29;33(47):13977–13988. doi: 10.1021/bi00251a004. [DOI] [PubMed] [Google Scholar]
- Weber P. C., Ohlendorf D. H., Wendoloski J. J., Salemme F. R. Structural origins of high-affinity biotin binding to streptavidin. Science. 1989 Jan 6;243(4887):85–88. doi: 10.1126/science.2911722. [DOI] [PubMed] [Google Scholar]
- Wilchek M., Bayer E. A. Applications of avidin-biotin technology: literature survey. Methods Enzymol. 1990;184:14–45. doi: 10.1016/0076-6879(90)84257-h. [DOI] [PubMed] [Google Scholar]
- Wilchek M., Bayer E. A. Introduction to avidin-biotin technology. Methods Enzymol. 1990;184:5–13. doi: 10.1016/0076-6879(90)84256-g. [DOI] [PubMed] [Google Scholar]