ABSTRACT
Members of the genus Xanthomonas are among the most important phytopathogens. A key feature of Xanthomonas pathogenesis is the translocation of type III secretion system (T3SS) effector proteins (T3SEs) into the plant target cells via a T3SS. Several T3SEs and a murein lytic transglycosylase gene (mlt, required for citrus canker symptoms) are found associated with three transposition-related genes in Xanthomonas citri plasmid pXAC64. These are flanked by short inverted repeats (IRs). The region was identified as a transposon, TnXax1, with typical Tn3 family features, including a transposase and two recombination genes. Two 14-bp palindromic sequences within a 193-bp potential resolution site occur between the recombination genes. Additional derivatives carrying different T3SEs and other passenger genes occur in different Xanthomonas species. The T3SEs include transcription activator-like effectors (TALEs). Certain TALEs are flanked by the same IRs as found in TnXax1 to form mobile insertion cassettes (MICs), suggesting that they may be transmitted horizontally. A significant number of MICs carrying other passenger genes (including a number of TALE genes) were also identified, flanked by the same TnXax1 IRs and delimited by 5-bp target site duplications. We conclude that a large fraction of T3SEs, including individual TALEs and potential pathogenicity determinants, have spread by transposition and that TnXax1, which exhibits all of the essential characteristics of a functional transposon, may be involved in driving MIC transposition. We also propose that TALE genes may diversify by fork slippage during the replicative Tn3 family transposition. These mechanisms may play a crucial role in the emergence of Xanthomonas pathogenicity.
IMPORTANCE
Xanthomonas genomes carry many insertion sequences (IS) and transposons, which play an important role in their evolution and architecture. This study reveals a key relationship between transposons and pathogenicity determinants in Xanthomonas. We propose that several transposition events mediated by a Tn3-like element carrying different sets of passenger genes, such as different type III secretion system effectors (including transcription activation-like effectors [TALEs]), were determinant in the evolution and emergence of Xanthomonas pathogenicity. TALE genes are DNA-binding effectors that modulate plant transcription. We also present a model for generating TALE gene diversity based on fork slippage associated with the replicative transposition mechanism of Tn3-like transposons. This may provide a mechanism for niche adaptation, specialization, host-switching, and other lifestyle changes. These results will also certainly lead to novel insights into the evolution and emergence of the various diseases caused by different Xanthomonas species and pathovars.
INTRODUCTION
The genus Xanthomonas represents an important group of bacterial plant pathogens with global economic agricultural impacts. Xanthomonas species are clearly distinctive in their pathology and plant host ranges (1). Among the more economically important are Xanthomonas citri, causing citrus canker, Xanthomonas oryzae pathovars, causing bacterial blight and leaf streak in rice (Oryza sativa) (2, 3), Xanthomonas campestris pv. vesicatoria and Xanthomonas arboricola pv. pruni, responsible for bacterial spot on a wide range of commercial plants, including pepper, tomato, and Prunus species (1, 4), and Xanthomonas fuscans subsp. fuscans and Xanthomonas axonopodis pv. manihotis, responsible for common bean blight and bacterial cassava blight, respectively (1, 5).
Citrus canker is an important disease affecting many citrus species. It is endemic in many parts of the world and has a severe economic impact on the citrus-producing industry (2, 6). Asiatic type A citrus canker caused by X. citri subsp. citri pathotype A infection is the most widespread and destructive citrus canker type (2). Types B and C result from infection by different strains of X. fuscans subsp. aurantifolii, and these are restricted to South America (2, 7). Citrus canker can be easily transmitted by wind-blown rain. Infection causes hyperplasia and necrosis, leading to cell lysis (8). As is the case for other diseases caused by the Xanthomonas genus, citrus canker involves a complex network of interactions implicating bacterial type II (T2SS) and type III secretion systems (T3SS) and biofilm formation (8). As observed in other pathosystems (9, 10), T3SS proteins are an important part of the Xanthomonas pathogenesis repertoire (11, 12), creating a bridge between the bacterium and the plant host cell and permitting the injection of bacterial T3SS effector proteins (T3SEs) into the plant cell cytoplasm (6). Over 60 type III effector proteins (http://www.xanthomonas.org/t3e.html), including transcription activator-like effectors (TALEs; see below) (13, 14), have been identified.
Xanthomonas genome sequences have proved a rich source of information concerning the evolution, lifestyle, and pathogenicity of members of the genus. The genome of X. citri subsp. citri pathotype A was deciphered over a decade ago (6), and the genomes of four other citrus canker-related species (X. fuscans subsp. aurantifolii strains ICPB 11122 and ICPB 10535, X. citri subsp. citri strain Aw 12879, and X. axonopodis pv. citrumelo strain F1) (2, 7) were partially or completely sequenced recently. Those of several additional species and pathovars are currently available or in process (http://www.xanthomonas.org).
An intriguing feature of the xanthomonads is their extraordinary genome plasticity and highly mosaic architecture (8), largely due to genomic rearrangements involving genomic islands and insertion sequences (IS) (15, 16). Indeed, IS are important landmarks of some Xanthomonas genomes. X. oryzae pv. oryzae and X. oryzae pv. oryzicola are rich in IS transposases, which can represent up to 10% of their genomes, whereas in X. citri subsp. citri strain 306 and X. campestris pv. vesicatoria, IS transposases represent less than 2% of the genome (17).
It has been noted that several Xanthomonas T3SEs are flanked by short (<100 bp) inverted repeat (IR) sequences (18) that form genetic structures typical of mobile insertion cassettes (MIC) (19). These are composed of passenger genes flanked by IS-related inverted repeats. MICs do not encode their own transposases but can be activated by a cognate transposase from a related IS (19). It has been suggested that these effector modules may have been transmitted by transposition and lateral gene transfer (18), although no specific IS family repeats have been reported to be predominantly associated with T3SEs (3).
The T3SEs found in the MIC structures include TALE genes. The products of these are transcription factors which modulate plant host gene expression to the benefit of the pathogen by interacting with various host cell factors and interfering with cell growth and the defense response (20, 21). Members of this class of DNA binding protein are found in a number of xanthomonads and are a key pathogenic feature (8, 13, 18, 22). Host DNA sequence specificity is determined by conserved near-identical tandem repeats, usually 34 amino acids long, that include a repeat-variable diresidue (RVD) at positions 12 and 13 (23). Each RVD recognizes a given nucleotide, and the string of tandem repeats therefore recognize a specific DNA binding sequence (20, 23, 24). TALEs are becoming powerful DNA-targeting tools in biotechnology (23, 25).
One of the largest and most diverse transposon groups is the Tn3 family. Members range in size and structure from 3,000 bp to more than 50,000 bp. All include a transposase (TnpA) of the DDE motif (RNase H fold) class (26, 27), and most carry a site-specific recombinase system, as well as passenger genes for functions such as resistance to antibiotics and heavy metals like mercury (28, 29) or copper (30), which have been sprayed for many years on vegetable and fruit crops to limit the spread of plant-pathogenic bacteria and fungi (31). They transpose by replicative replicon fusion, generating a cointegrate with a copy of the transposon in direct orientation at each junction between the fused donor and target replicons (26). Transposition terminates by recombination (resolution) between the two directly repeated Tn copies at a particular site, res or rst, in a reaction catalyzed by a transposon-encoded site-specific recombinase. In many Tn3 family members, this is a serine recombinase, the resolvase (TnpR) (26). In others, it may be a single tyrosine recombinase related to phage integrases (TnpI) (32) or a pair of genes encoding TnpT and TnpS (33). TnpS is related to the bacteriophage P1 Cre tyrosine recombinase, whereas TnpT shows no similarity to any known protein. Shorter derivatives have also been identified that encode only the transposase and are devoid of the resolvase (e.g., ISVsa19, ISShfr9, ISBusp1, and IS1071D) (34). These therefore resemble simple bacterial ISs.
In the studies reported here, high-quality IS annotation coupled with comparative genomics revealed an important group of xanthomonad Tn3 family transposons and derivatives with associated T3SEs in general and TALE genes in particular. This includes a complete Tn3 family transposon in the X. citri subsp. citri pathotype A genome and a large number of derivatives in plasmids and chromosomes of other Xanthomonas species.
The X. citri subsp. citri pathotype A Tn3-like transposons carry T3SE passenger genes, together with a highly conserved murein lytic transglycosylase gene (mlt). The analysis also underlined the importance of related transposon-like structures in the transmission of other pathogenicity factors between different xanthomonads. Not only were the four previously known X. citri subsp. citri pathotype A plasmid (pXAC33 and pXAC64) TALE genes located in a MIC structure flanked by the same Tn3-like repeats as the TnXax1 transposon, but many other TALE and pathogenicity genes from other xanthomonads were observed to be associated with similar structures. These results provide strong evidence that, at least in the case of X. citri subsp. citri pathotype A and other closely related species, key pathogenicity and virulence genes are spread by a Tn3-like transposition mechanism and use (or, in the case of X. oryzae, have used) conjugative plasmids as intercellular vectors. We propose that the transposition process can inherently generate TALE gene diversity and is a key agent in modulating bacterial virulence and host specificity in this group of plant pathogens.
RESULTS
Tn3 family elements in the pXAC64 plasmid and the distribution of their specific IRs among the xanthomonads.
Plasmid pXAC64 and closely related plasmids are widely distributed in xanthomonads (4, 7), including X. oryzae strains isolated from North America, although they appear to be absent in the sequenced X. oryzae and X. oryzae pv. oryzicola genomes (3, 35). This distribution presumably occurred by conjugal transfer, since the plasmids carry a transfer operon (a type IV secretion system [T4SS]) with a relaxase gene related to trwC of plasmid R388 and traI of the classical conjugative F plasmid (36, 37).
Noël et al. (38) identified this region in the X. citri subsp. citri pathotype A plasmid pXAC64 (6) and noted that the xopE2 effector and mlt genes were associated with three transposon-related genes and that the entire structure was flanked by IR sequences. However, the nature of these transposon-related genes was not addressed.
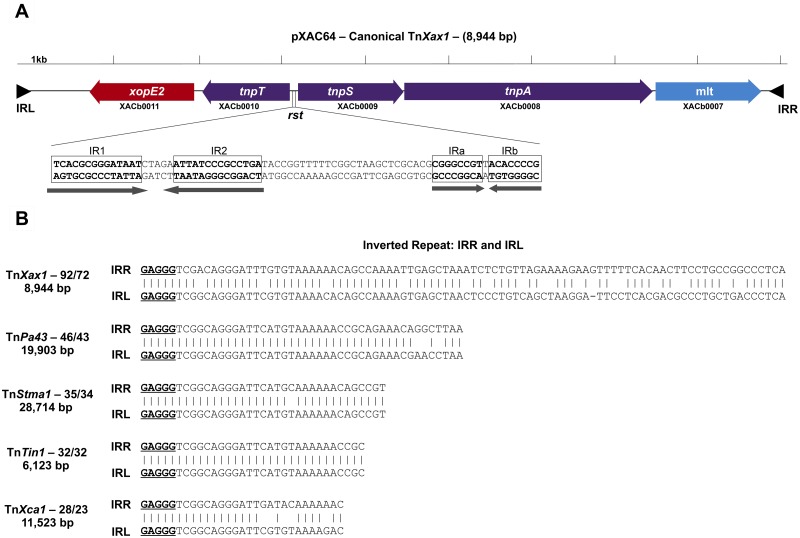
We have revisited this question during our ongoing studies of X. citri subsp. citri pathotype A pathogenesis. We found that not only do the flanking IR sequences correspond to the highly conserved terminal IR of members of the large Tn3 transposon family but the associated transposon-related genes are also typical of Tn3 family members. This potentially transposable structure, which we have called TnXax1, has a length of 8,944 bp (Fig. 1A).
FIG 1 .
(A) Genetic organization of the canonical TnXax1 located in X. citri subsp. citri strain 306 plasmid pXAC64. Genes are indicated by colored boxes, with the direction of transcription shown by the arrowheads. Transposition-related genes are shown in purple, and passenger genes in red and blue. The presumed resolution site is located between the tnpT and tnpS genes, as observed in the transposable element Tn4651. This includes two palindromes: IR1 and IR2 are probably part of the core site at which recombination occurs, recognized by the TnpS recombinase, whereas IRa and IRb are potential binding sites for TnpT. The terminal inverted repeats (IRL and IRR) are shown as black triangles. The convention used for orientation of the transposon is that the transposase (TnpA) is transcribed from left to right. TnpA is closely related to other Tn3-like elements from Shewanella (ISSod9; 47% identity), Acinetobacter (ISAcsp1; 46%), Arthrobacter (ISArsp6; 43%), Azospirillum (ISAzs17; 41%), Salmonella (ISSwi1; 42%), and Nostoc (ISNpu13; 43%) species and to the mercury resistance transposon Tn5044 from X. campestris (84%). (B) IRs identified from other Tn3-like transposable elements. All include the GAGGG tips sharing sequence similarities to TnXax1 IRs. These are TnPa43 from Pseudomonas aeruginosa, TnStma1 from Stenotrophomonas maltophilia strain D457, TnTin1 from Thiomonas intermedia strain K12, and TnXca1 from X. campestris pv. vesicatoria plasmid pXCV183.
The TnXax1 IR sequences are shown in Fig. 1B. They are 92 bp long with 72 bp of identity. Although this is rather long for Tn3 IRs, several other family members are also known to carry long IRs (ISfinder database). The TnXax1 IRs begin with GAGGG instead of the more common GGGGG. These full-length TnXax1-like IRs are generally restricted to the Gammaproteobacteria, particularly to Xanthomonas species. There are at least 264 different occurrences of these in the Xanthomonadales (see Table S1 in the supplemental material), often forming part of TnXax1 derivatives and putative MIC elements, although there are several examples of solo IRs. Intriguingly, 6 full-length TnXax1-like IRs are also found in a unique sequenced betaproteobacterial genome, that of the cucurbit pathogen Acidovorax avenae subsp. citrulli (Table S1), flanking a XopJ2 effector (see below). Tn3 family IRs with GAGGG tips occur in several other Tn3-like transposons (Fig. 1B; Table S2). However, the IRs are all significantly shorter than those of TnXax1.
TnXax1 transposition functions.
The putative transposition functions of TnXax1 (Fig. 1A) are closely related to those of TnPa43. The longest open reading frame (ORF), XACb0008, resembles a Tn3 transposase (62% identity at the protein level to that of TnPa43). The second, XACb0009, shows high similarity to the TnPa43 tyrosine recombinase, TnpS (49% identity), and the third, XACb0010, is related to the Tn4651 auxiliary cointegrate resolution protein T (TnpT) (43% identity) (33). TnpS and TnpT are oriented divergently in both Tn4651 and TnXax1.
For Tn4651, the intergenic region between the divergent tnpS and tnpT genes is 188 bp and includes two short palindromes within a 136-bp functional resolution recombination site (33), the Tn3 family rst site (26). One pair are putative binding sites for TnpT, whereas the other pair are part of the core site at which recombination occurs (33).
The TnXax1 tnpT-tnpS intergenic region is 193 bp and also includes two pairs of palindromic sequences; one pair, IR1 and IR2, are 14 bp with a potential core recombination site of 5 bp (Fig. 1A), and the other pair are two short IRs, IRa and IRb, which could represent a putative TnpT binding site (33).
Other TnXax1-related derivatives.
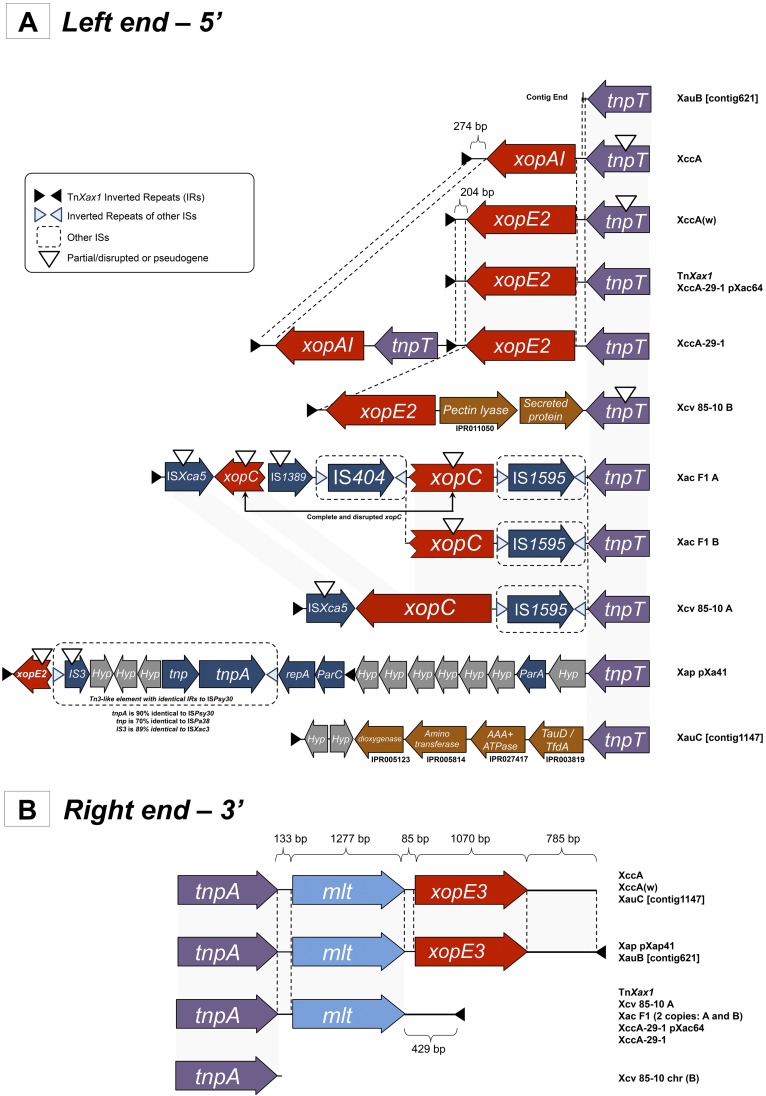
A TnXax1-related structure was previously identified in plasmid pXap41, which is present in a variety of X. arboricola pv. pruni species (4). We have identified a number of additional TnXax1-related structures in a variety of xanthomonads (Fig. 2), both in plasmids and chromosomes. One striking feature of these is that, while the right ends are quite similar (Fig. 2B), the left ends exhibit a large degree of variability (Fig. 2A). Clearly the left end can act as a platform for accommodating various passenger genes, as well as different IS and other Tn3 family transposons. Additional IR copies are also observed in some cases, permitting the transposition of alternative structures using alternative combinations of ends. Many contain mutations which would render them unable to transpose (e.g., an in-frame stop codon in tnpT and the absence of a right IR [IRR] in X. campestris pv. vesicatoria strain 85-10 chromosomal copy B) (Fig. 2A). In addition, it seems probable that intertransposon recombination has also played a role in generating these structures. For example, the X. citri subsp. citri strain 29-1 chromosomal TnXax1 derivative (called XccA-29-1 herein) includes xopE2 and xopAI effector genes and two left IRs (IRLs). It resembles a chimera derived by recombination between the TnXax1 from the pXAC64 plasmid and the X. citri subsp. citri pathotype A chromosomal TnXax1 copy (Fig. 2A).
FIG 2 .
Genetic organization of the left (A) and right (B) ends of the TnXax1-related structures found in other Xanthomonas species. Abbreviations for the TnXax1-related structures: Xap pXap41, X. arboricola pv. pruni, plasmid pXap41; XccA, X. citri subsp. citri pathotype A, chromosomal copy; TnXax1, X. citri subsp. citri pathotype A, plasmid copy and canonical element; Xac F1 A and Xac F1 B, X. axonopodis pv. citrumelo strain F1, 2 chromosomal copies; Xcv A and Xcv B, X. campestris pv. vesicatoria strain 85-10, chromosomal copies A and B; XccA(w), X. citri subsp. citri strain Aw 12879, chromosomal copy; XccA-29-1, X. citri subsp. citri strain 29-1, chromosomal copy; XccA-29-1 pXac64, X. citri subsp. citri strain 29-1, plasmid copy; XauB ctg 621_0, X. fuscans subsp. aurantifolii strain ICPB 11122; XauC ctg 1147_0, X. fuscans subsp. aurantifolii strain ICPB 10535.
Passenger genes: effector proteins and Mlt.
These transposon-like structures were initially identified in regions containing a number of T3SE pathogenicity genes. The annotated regions shown in Fig. 2 show that these occur as passenger genes within many of the TnXax1 derivative structures. These are principally the effector genes xopE2, xopE3, xopAI, xopC, and mlt, which have received much attention over the past few years. They are central for pathogenicity (38–40).
The mlt gene is not considered to be a T3SE family member (4), but except for a single case from X. campestris pv. vesicatoria 85-10 chromosomal copy B, it is consistently found in these structures (Fig. 2B). Laia and coauthors previously suggested that Mlt may be important for T3SS apparatus assembly and activity (41). This conservation suggests that mlt is maintained by a significant selective pressure. It is noteworthy that mlt homologues are also present in a variety of xanthomonads (see Tables S3 and S4 in the supplemental material) and in other genera, such as Pseudomonas and Ralstonia. Mlt shares 63% sequence identity with HopAJ1 from Pseudomonas syringae pv. tomato DC3000 (4, 41). Although HopAJ1 is not a T3SS substrate, it probably enables the T3SS to penetrate the peptidoglycan layer in the bacterial periplasm and deliver virulence proteins into host cells (42). In X. citri subsp. citri pathotype A, it appears to be expressed specifically during in vivo multiplication, and its deletion reduces the severity of citrus canker (41, 43).
TnXax1 from plasmid pXAC64 and from the X. citri subsp. citri pathotype A chromosome carry two different T3SE genes. That from pXAC64 includes xopE2, a putative transglutaminase that acts on the plant cell plasma membrane, suggesting a role in virulence and in the suppression of the hypersensitive response of the host (40). The X. citri subsp. citri pathotype A chromosome copy includes xopAI, homologous to the P. syringae pv. tomato virulence factors hopU1, hopO1-1, and hopO1-2. These probably have a posttranslational modification function (44) (Fig. 2A). Interestingly, the two effectors share about 200 bp at their 5′ ends. The C-terminal end of XopAI shows some similarity to the arginine ADP-ribosyltransferase family (PF01129). The chromosomal X. citri subsp. citri pathotype A TnXax1 derivative also includes an additional 274 bp of noncoding DNA between xopAI and the IRL, whereas the derivative X. citri subsp. citri strain Aw 12879 has 204 bp of noncoding DNA between xopE2 and IRL. Other differences between the TnXax1 structures are much more complex, suggesting that a high level of recombination may be involved in their formation (Fig. 2A).
Both X. axonopodis pv. citrumelo strain F1 TnXax1 relatives (A and B) carry a disrupted xopC gene whose product is secreted normally and translocates into the plant cell (Fig. 2A) (38). The xopC copies found in the X. axonopodis pv. citrumelo F1 A TnXax1 derivative are disrupted by IS404 and IS1389 insertions (Fig. 2A) that are not present in X. axonopodis pv. citrumelo F1 B.
As shown in Fig. 2 and in Table S2 in the supplemental material, many other passenger genes carried by these transposon-like structures are also related to virulence.
TALE gene distribution among the Xanthomonadales: a story of transposition.
Although several of the >264 different IR occurrences (see Table S1 in the supplemental material) were found to form part of TnXax1 derivatives, many occurred as inverted repeats flanking passenger genes without associated transposition genes and form putative MIC elements.
Analysis of the passenger genes neighboring the 92 bp IR showed that many were TALE gene family members. TALE genes have attracted attention by their simple and predictable specific DNA sequence recognition readout, which uses a number of repeating peptide blocks, some of which include a dipeptide with specific recognition properties for a given base (20, 24). In their original xanthomonad hosts, TALE proteins are transmitted to the host plant cell nucleus, where they activate or inactivate important genes, thus contributing directly to pathogenicity. Indeed, a single amino acid substitution in a TALE sequence can affect citrus canker development (22). However, the detailed mechanism of transcriptional regulation by TALEs is still not fully understood (20).
Previous studies had revealed that a number of these in different Xanthomonas strains are flanked by 62 bp IRs, and it was suggested that they may have been transmitted as mobile cassettes (18). Two were identified on each of the two X. citri subsp. citri strain 306 plasmids, pXAC33 (carrying pthA1 and pthA2) and pXAC64 (carrying pthA3 and pthA4) (18). The four genes are slightly different, with the principal variations located in the repeating peptide blocks. The analysis performed here showed that they are flanked by the same 92 bp (rather than 62 bp) IR sequence as the Xanthomonas-specific TnXax1 IR. This suggests that the Tn3-like element may provide transposition functions for these MIC elements.
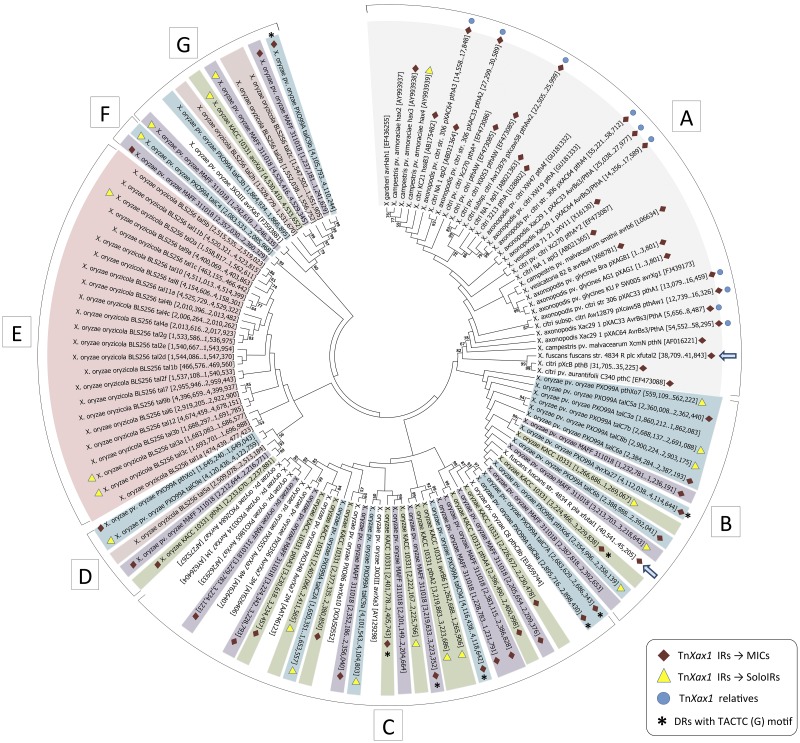
Boch and Bonas described 113 known TALEs in different Xanthomonas species (18) (see also Bogdanove et al. [14]). Here, we analyzed these genes together with newly identified TALE genes deposited in the public databases, focusing principally on those from complete and fully sequenced genomes. A maximum-likelihood tree constructed from the full-length DNA sequences of 122 different TALEs is shown in Fig. 3. The topology shows well-defined groups, including two major groups (Fig. 3B and C) and three minor groups (Fig. 3D, F, and G) composed of TALEs from various X. oryzae strains (Fig. 3, purple, green, blue, and white), a group composed of TALEs from X. citri, X. axonopodis, X. campestris pv. vesicatoria, and X. campestris (Fig. 3A, gray), and a group representing most of the X. oryzae pv. oryzicola BLS256 TALEs (pink) (Fig. 3E). A similar topology was obtained using the set of full-length proteins (data not shown). The general topology is consistent with that obtained in other studies, which used nucleotide sequences without the RVD repeats (14), concatenated N- and C-terminal portions of the proteins (45), or C-terminal regions alone (46).
FIG 3 .
Molecular phylogenetic analysis by maximum-likelihood method of the TALEs genes found in completely sequenced Xanthomonas genomes. Each TALE gene is shown with its respective genomic coordinates (for TALEs extracted from complete genomes) or GenBank accession number inside the brackets. Symbols: red diamonds, TALE genes associated with MIC structures; yellow triangles, TALE genes associated with a solo IR flanking one of their extremities; blue circles, genomes which carry TnXax1 or related structures; asterisks, MICs with the direct TACTC(G) target repeat; blue arrow, the TALE gene from X. fuscans subsp. fuscans strain 4834-R plasmids pla and plc. TALEs from different X. oryzae pathovars are highlighted as follows: blue, X. oryzae pv. oryzae strain PXO99A; green, X. oryzae pv. oryzae strain KACC 10331; purple, X. oryzae pv. oryzae strain MAFF311018; pink, X. oryzae pv. oryzicola strain BLS256. TALEs from X. citri, X. axonopodis, X. campestris pv. vesicatoria, and X. campestris are highlighted in gray.
Association of TALE genes with TnXax1 IR.
A substantial number of TALE genes from this collection (55 or 45%) (Fig. 3) proved to be associated with the TnXax1 IRs and to form MIC structures. These include both MICs with full-length IRs and MICs with IRs ranging from 33 bp to 97 bp but always maintaining the GAGGG tip (see Table S3 in the supplemental material). MIC-associated TALEs are found in a majority of the Asian X. oryzae and the X. citri/axonopodis/campestris/campestris pv. vesicatoria group, suggesting that transposition by a Tn3 mechanism may be an important driver of TALE dispersion and diversification in these groups. However, they are much less frequent in the X. oryzae pv. oryzicola strains.
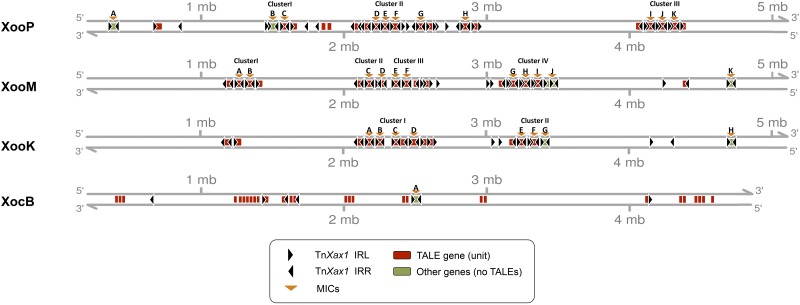
In addition to the MIC structures, 20 solo IRs were identified next to TALEs from the X. oryzae pv. oryzae and X. oryzae pv. oryzicola groups, and a potential solo IR was identified in the X. citri/axonopodis/campestris/campestris pv. vesicatoria group (Fig. 3; see Table S1 in the supplemental material). Solo IRs may reflect previous transposition or recombination events involving the adjacent TALE gene. The chromosomal TALE-containing MICs are particularly notable in the Asian X. oryzae group (Fig. 4), where, assuming that there have been no major errors in genome assembly, there are small clusters of tandemly repeated MIC units. Figure 4 and Fig. S1 show the distribution of all MIC structures and their association with the TALE genes and solo IRs. Table S3 lists these elements in more detail. In all three X. oryzae strains, the tandemly repeated MIC units are distributed in a complex cluster organization that is distinct for each strain (Fig. 4).
FIG 4 .
Linear genomic representation of the distribution of the IRs and tandemly repeated MIC clusters in X. oryzae genomes. Abbreviations: XooP, X. oryzae pv. oryzae strain PXO99A: XooM, X. oryzae pv. oryzae strain MAFF311018; XooK, X. oryzae pv. oryzae strain KACC 10331; XocB, X. oryzae pv. oryzicola strain BLS256.
Moreover, the DNA located between the MICs of each cluster shows a higher-than-average percentage of genes encoding transposases from other transposable element (TE) families, integrases, and phage-related functions and could represent a plasticity region in which insertions and rearrangements do not adversely affect cell viability. Since more than one MIC structure and IR are associated with each locus, transposition of alternative structures using different combinations of neighboring ends may be possible. In addition, these X. oryzae genomes also carry other isolated TALE genes and solo IRs which may also act as substrates for recombination and transposition of alternative structures.
For some MICs, it was possible to identify 5 bp target duplications that are commonly generated by Tn3 family elements, indicating that these insertions represent true transposition events (Fig. 3; see Table S3 in the supplemental material). These DRs are diverse in sequence (Table S3), but a particular sequence, TACTC(G), is shared by a number of MIC elements of all three X. oryzae strains, although the associated TALEs are different and grouped in different X. oryzae subgroups (Fig. 3B, C, and G, asterisks). This may indicate either that these insertions exhibit a preferred target sequence or that the TALEs have evolved from a single MIC which has been displaced and amplified and has diverged as part of a larger structure (Table S3).
Relationship between TALEs from different xanthomonads.
Not all TALE genes are included in well-defined MICs. The X. oryzae pv. oryzicola strain BSL256 carries many such copies (Fig. 3, pink). The X. oryzae pv. oryzicola tal5a without IRs is located on the phylogenetic tree within a small group of X. oryzae MIC-associated TALEs (Fig. 3D, blue, purple, and green) in a branch separate from the main X. oryzae groups (Fig. 3B and C) and more closely related to X. oryzae pv. oryzicola strain BSL256 TALEs (Fig. 3E). Another subset of X. oryzae pv. oryzicola BSL256 TALEs without IRs, tal2c, tal2b, and tal2h, forms part of the second X. oryzae TALE clade (Fig. 3G), which also includes MIC-associated TALEs (Fig. 3G, blue, purple and green). It is possible that X. oryzae pv. oryzicola BSL256 first acquired these TALEs by transposition and has subsequently lost the IRs.
Although the majority of X. oryzae pv. oryzicola BSL256 TALEs are not associated with flanking IRs (Fig. 3C), three appear to have a single IR copy. This is also consistent with the notion of IR decay.
There are a number of unexpected incongruences in the phylogenetic tree. In particular, X. fuscans subsp. fuscans strain 4834-R carries both X. oryzae and X. citri/axonopodis/campestris/campestris pv. vesicatoria-related TALEs. These are plasmid located (5). The TALE from one of the three resident plasmids, X. fuscans subsp. fuscans plasmid A, is grouped with those of the X. oryzae strains (Fig. 3B, blue arrow), whereas that of X. fuscans subsp. fuscans plasmid C is grouped with those of the X. citri/axonopodis/campestris/campestris pv. vesicatoria strains (Fig. 3A, blue arrow). The presence of both types of TALE in different clades is therefore most simply explained by plasmid-mediated horizontal transfer.
MICs that include other pathogenicity functions.
In addition to TALE genes, MIC structures contain other T3SEs. The unique MIC of X. oryzae pv. oryzicola BLS256 includes several complete and partial TEs of different families and the effector gene xopU (see Table S3 in the supplemental material). This effector is exclusive to X. oryzae strains and is found in different MIC structures. Other potential effector proteins and pathogenicity functions are shown in Fig. S1 and Table S3. While MICs carrying TALE genes tend to be rather simple, those carrying other potential effector and pathogenicity factors appear more complex and can include more than two IRs. They vary in length from 3 to 19 kb. Closely related elements are present in different related strains. This is the case for the two 16.2 kb chromosomal Mic A from X. citri subsp. citri ATCC 33913 and X. citri subsp. citri 8004 (which are inverted with respect to each other in the genome) and the 18.2 kb X. oryzae MIC J (strain MAFF311018) and MIC G (strain KACC 10331). It should also be noted that MIC B (X. oryzae strain PXO99A) is nearly identical to the MAFF311018 and KACC 10331 copies but contains additional DNA, including an extra IR and two different T3SEs (xopU and xopX).
In general, all these MICs carry known T3SS effector genes, such as xopJ2, xopJ5, xopT, xopU and xopX, as well as mlt and genes for pectin lyase, protein tyrosine phosphatase, a resistance-nodulation-cell division (RND) efflux pump, and members of a two-component system. Type IA DNA topoisomerase, potential tyrosine recombinase, toxin/antitoxin, and acriflavin resistance genes are also represented, as well as many unrelated hypothetical protein genes (see Table S3 in the supplemental material). Moreover, MIC H from X. oryzae KACC 10331, carrying a polymerase/histidinol phosphatase-like protein, generated 5 bp target site duplication, clearly indicating that it had inserted into the genome by transposition.
Transposase sources for MIC mobility.
Clearly, the presence of flanking target DR observed for a number of MIC structures in these Xanthomonas strains indicates that they have undergone true transposition events (e.g., those marked with asterisks in Fig. 3B, C, and G and in Table S3 in the supplemental material). This requires an external source of the cognate transposase. Many TALEs of the X. citri/axonopodis/campestris/campestris pv. vesicatoria group are located on plasmids. In these cases, transposition functions could be supplied either from the plasmid TnXax1 derivatives or, when present, from a chromosomal TnXax1 derivative. On the other hand, Asian X. oryzae and X. oryzae pv. oryzicola group strains, whose TALEs are uniquely chromosomal, do not carry plasmid or chromosomal TnXax1 derivatives or indeed isolated transposition functions. Thus, further MIC transposition is unlikely. To explain the appearance of TALEs in Asian X. oryzae and X. oryzae pv. oryzicola group strains, it seems probable that they were acquired by transmission from an ancestral plasmid which has subsequently been lost. The loss of this type of plasmid has been observed in some North American X. oryzae strains (which are more closely related to X. oryzae pv. oryzicola [35]). Most contain a plasmid similar to that of X. campestris pv. vesicatoria pXCV38, with a Tn3-like element (ISXc4) whose transposase is 40% identical to TnpA of TnXax1, and others appear to have lost the plasmid during laboratory culture (35).
Moreover, a second related plasmid, pXCV183 from X. campestris pv. vesicatoria, carries a potential transposon, TnXca1 (see Table S2 in the supplemental material), which shares the same IR tips and carries TnpA, TnpS, and TnpT with 59, 98, and 91% identity, respectively, to those from TnXax1 (Table S4). Although the Asian X. oryzae group strains do not carry TnXax1 relatives, X. oryzae PXO carries structures similar to TnXca1, which carries only 29 instead of 92 bp TnXax1 IRs. The X. oryzae PXO TnXca1 derivative carries tnpA and tnpS genes with 59 and 80% identity, respectively, to those in TnXax1 (Table S4). An additional and partial copy without IRs is also found in X. citri subsp. citri strain B100 (Table S4). Since the TnXca1 IRs and transposition-related genes share considerable similarity to those of TnXax1, TnXca1 may be able to provide a functional transposase for the MICs found in X. oryzae PXO. It is noteworthy that a partial TnXca1-related structure is also found in the genome of Pseudoxanthomonas spadix strain BD-a59, thus indicating that this class of transposon is not exclusive to the Xanthomonas genus (Table S4).
DISCUSSION
We have identified a novel class of Tn3 family-derived transposable elements in Xanthomonas species which could potentially mediate combinatorial spread and diversification of plant virulence factors. The group includes TnXax1, an apparently intact transposon from Xanthomonas citri, and we have also identified a number of related structures in a variety of xanthomonads. These exhibit several different genetic organizations and carry different types of passenger genes, mostly T3SEs. Although the right sides of these structures fall into relatively homogenous groups, the left sides are highly variable: each derivative appears to have a unique left side, indicating the occurrence of different recombination and duplication events. The IRs of these structures are commonly associated with passenger genes, mostly TALE genes, forming MIC elements. Target site duplications of 5 bp, commonly generated by elements from the Tn3 family, were also identified in some of the TnXax1-related structures and MICs, strongly supporting the idea that these are products of true transposition events.
Studies with Bacillus cereus strains showed that their MICs (which carry genes encoding products such as endopeptidase, antibiotic resistance proteins, or regulatory factors) can be mobilized in trans by a cognate transposase (47). Different MIC structures carrying TALE genes are often found in Xanthomonas strains, and their role in the emergence of pathogenicity may be considerable.
Relationship of MIC clusters to the emergence of Xanthomonas pathogenicity.
The transposition of TALE-carrying MICs in Xanthomonas strains presents an intriguing puzzle, since they appear to be grouped in clusters. This may indicate that the MICs in each cluster were amplified and diverged from a single insertion event. Moreover, in several citrus pathogens and related species, the MIC-associated TALE genes are often found in plasmids, whereas in the X. oryzae and X. oryzae pv. oryzicola species, these occur in the chromosome. In addition, the presence of plasmids may be essential to the development of disease caused by X. fuscans subsp. aurantifolii strains. For instance, the plasmid pXcB from X. fuscans subsp. aurantifolii can be fully transferred in planta and is central to the development of citrus canker (37). Since some X. oryzae group members from North America carry plasmids similar to pXCV38, it is possible that a plasmid-borne TnXax1 relative and TALE-carrying MICs transiently invaded members of the X. oryzae group and subsequently spread by transposition driven by TnXax1 or by a cognate transposase from a closely related element (such as ISXc4 or TnXca1). This mechanism may have been responsible for delivering the pthA4- and pthAw-carrying MICs to X. citri subsp. citri pathotype A and X. citri subsp. citri strain Aw 12879, respectively. These effectors are considered central for the development of the citrus canker (43). The X. oryzae pv. oryzicola strain BLS256 does not have TALE-carrying MICs, and the majority of its TALEs group in a monophyletic clade. Moreover, the lines of evidence presented and the tree topology, shown in Fig. 3, strongly suggest that, after acquisition by transposition, the X. oryzae pv. oryzicola BSL256 TALEs evolved principally by duplication rather than by transposition. Although gene duplication is considered one of the major mechanisms through which new genetic material is generated, transposition can promote bacterial evolution in quantum leaps. Since X. oryzae pv. oryzicola BSL256 is less widespread and causes less severe disease than members of the X. oryzae group (3), the absence of MIC-carrying TALEs observed in this strain may have affected its pathogenicity.
Generating T3SE and TALE diversity by transposition.
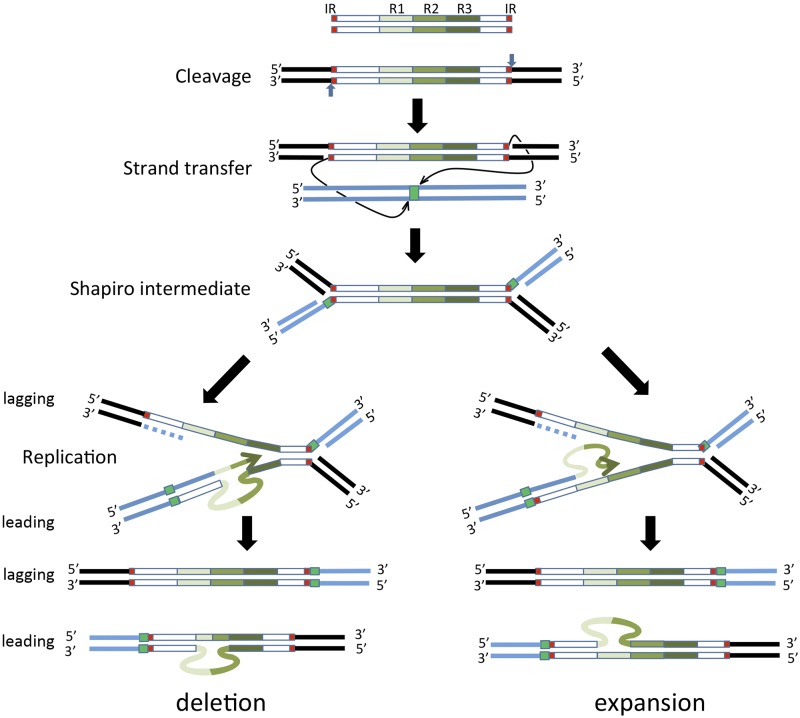
In addition to insertion and genome expansion, the Tn3 family replicative transposition mechanism could itself generate sequence variation in several ways. Reassortment of passenger genes leading to changes in the T3SE repertoire could occur by “resolution” (site-specific recombination) between alternative rst sites from different elements, as shown for Tn4651-related transposons (33). Replicative Tn3 transposition would itself provide an exquisite mechanism for generating TALE diversity. TALE genes include a number of conserved nearly identical tandem repeat sequences that encode the 34-amino-acid repeats, including the RVD site. Changes in the repeat numbers, RVDs, and sequences in the TALE genes might occur by replication slippage between direct repeats and errors during the replicative stage of transposition (Fig. 5) or by unequal crossing-over, as has been proposed for amplification of other repeated DNA segments (48, 49). The example presented in Fig. 5 shows that replication slippage on the template strand (bottom left) would give rise to a reduction in the number of repeat sequences, while slippage on the nascent strand (bottom right) would result in repeat expansion following a second round of replication. Similar activities might occur on the lagging strand and lagging strand template. This mechanism may remodel the bacterial T3SE arsenal, producing novel effectors capable of binding to new DNA motifs to regulate new sets of host genes. Alternatively, such rearrangements may be generated similarly during replication of the host replicons or during conjugative transfer of the plasmid vectors. Experiments are under way to test these hypotheses.
FIG 5 .
A model for generating variability in the number of TALE gene repeats. The proposed steps of Tn3 family replicative transposition are shown. Top to bottom: double-strand transposon showing the terminal inverted repeats (IRs, red boxes) and three repeat sequences (green scale boxes); the transposon in situ in the transposon donor molecule (flanking black bars), indicating the polarity of the two DNA strands and the polarity of cleavage (vertical blue arrows); transfer of the 3′ OH strand at each end of the transposon (thin arrows) at staggered positions (green square) into the target molecule (blue bars); formation of the branched Shapiro intermediate with both donor (black bars) and target (blue bars) DNA attached; replication of the intermediate structure showing leading strand synthesis (bottom branch of the fork) and lagging strand synthesis (top branch of the fork); replication fork slippage on the leading strand template (left) resulting in removal of repeat sequences or slippage on the nascent strand (right) resulting in expansion of repeat sequences, which are each resolved in a second round of replication.
Tn3 family elements from closely related genera.
The involvement of Tn3 family elements with virulence and pathogenicity is not an exclusive feature of the Xanthomonas genus. Additional Tn3 family transposons identified in other Gammaproteobacteria, such as Pseudomonas aeruginosa (TnPa43) (50, 51) and Stenotrophomonas maltophilia (TnStma1) (52, 53), share TnXax1 IR sequences (GAGGG tips). These are also involved in assisting the bacterial host-related lifestyle, since they also carry genes directly or indirectly involved in pathogenicity and virulence or tolerance of high levels of various toxic metals. Xanthomonas, Pseudomonas, and Stenotrophomonas species are gammaproteobacteria and share a close common ancestor. Lateral gene transfer mediated by these Tn3-like transposable elements may have had an important role in speciation, shaping the life styles of their bacterial hosts. The Xanthomonas and Pseudomonas genera are composed of several species capable of infecting different plant hosts. Even though more than 200 plant species are hosts for Xanthomonadales and Pseudomonadales species (54), most Xanthomonas and Pseudomonas species exhibit a strict host range, resulting in specific combinations of plant-pathogen interactions that result in pathogen infection (or disease). The mobilization of TnXax1 and its relatives and the potential recombination and shuffling of the T3SEs and TALEs in MIC structures are probably major sources of pathogenicity variation in these plant pathogens. However, we would like to point out that, in this study, we have only addressed transposons of a subclass of the Tn3 family, those with IRs whose sequences end in GAGGG. It is possible that some of the many other transposable elements present in some of these xanthomonad genomes are also involved in transmitting pathogenic determinants. Work is under way to determine this.
MATERIALS AND METHODS
Bioinformatics analysis.
Insertion sequences were annotated using the ISfinder database (55) and ISsaga tool (56). Global comparisons using sequenced Xanthomonadales genomes deposited in the public GenBank repository were made using BLAST (57) with standard parameters, except that the low-complexity-region filter was deactivated and a word size of 7 was used.
The molecular phylogenetic analysis of the TALE genes was performed using the maximum-likelihood method based on the general time-reversible model (58). The full-length nucleotide sequences of the TALES genes were aligned and edited to maintain the order of the conserved tandem repeats, with no gap inclusion in order to respect the targeted nucleotide sequence in the plant and, therefore, the TALE specificity. The bootstrap consensus tree was inferred with 500 replicates (59). Branches corresponding to partitions reproduced in less than 50% of bootstrap replicates were collapsed. Initial trees for the heuristic search were obtained automatically by applying the Neighbor Joining and BioNJ algorithms to a matrix of pairwise distances estimated using the maximum composite likelihood (MCL) approach and then selecting the topology with superior log likelihood value. A discrete gamma distribution was used to model evolutionary rate differences among sites (3 categories [+G, parameter = 1.0586]). The analysis involved 122 TALE sequences, including a total of 8,174 positions in the final data set. Evolutionary analyses were conducted in MEGA6 (60).
SUPPLEMENTAL MATERIAL
Linear genomic representation of the distribution of the IRs and potential MIC structures, TALEs, and TnXax1 relatives. (A) Genomic representation of all plasmids. (B) Genomic representation in the main chromosomes. Abbreviations: Xcv pXCV183, X. campestris pv. vesicatoria strain 85-10 plasmid pXCV183; XccA(w) pXcaw58, X. citri subsp. citri strain Aw 12879; Xff, X. fuscans subsp. fuscans strain 4834-R; Xff pla, X. fuscans subsp. fuscans strain 4834-R plasmid pla; Xff plc, X. fuscans subsp. fuscans strain 4834-R plasmid plc; Xap pXap41, X. arboricola pv. pruni plasmid pXap41; Xc pXcb, X. citri plasmid pXcB; Xag 8ra pXAG81, X. axonopodis pv. glycines strain 8ra plasmid pXAG81; Xa GPE PC73, X. albilineans strain GPE PC73 plasmid; Xag AG1 pAG1, X. axonopodis pv. glycines strain AG1 plasmid pAG1; A. citrulli, Acidovorax avenae subsp. citrulli strain AAC00-1; Xcc ATCC 33913, X. campestris pv. campestris strain ATCC 33913; Xcc B100, X. campestris pv. campestris strain B100; Xcc 8004, X. campestris pv. campestris strain 8004; Xa GPE PC73, X. albilineans strain GPE PC73. Download
List of occurrences of the TnXax1 full-length IRs in Xanthomonadales (Gammaproteobacteria) sequences deposited in GenBank (excluding draft or incomplete genomes). The genome coordinates represent the orientation of each IR. The numbers of positions in the identified sequences that are conserved with respect to the 92 bp right IR of TnXax1 are shown in the third column. This table also shows degenerate IRs that maintain the GAGGG tips and vary from 33 bp to 92 bp. These TnXax1 IRs are also found forming a potential MIC element in a unique Betaproteobacteria genome, that of Acidovorax avenae subsp. citrulli strain AAC00-1, a watermelon pathogen.
Tn3-like transposable elements with IRs (GAGGG tips) similar to those of TnXax1, including the transposition and passenger genes identified in TnPa43 from Pseudomonas aeruginosa (A), TnStma1 from Stenotrophomonas maltophilia strain D457 (B), and TnXca1 from X. campestris pv. vesicatoria plasmid pXCV183 (C). The grey-shaded rows indicate the transposition-related genes. The coordinates represent the position of each open reading frame in the transposon itself.
Potential MIC structures and their genomic features and gene content.
Distribution, genomic coordinates, and genome context of the mlt, TnpA, TnpS, resolution site (res site), and TnpT homologues (>40% DNA sequence identity) over those of other Xanthomonas species that do not carry the TnXax1 canonical element or relatives. The identity values were calculated based on the canonical sequence of TnXax1 from X. citri subsp. citri strain 306 pXAC64. The genome context is shown inside the brackets and represents the open reading frame located up- and downstream.
ACKNOWLEDGMENTS
R.M.F. was supported by a Ph.D. fellowship from CAPES (Coordenação de Aperfeiçoamento do Pessoal de Ensino Superior), A.C.P.D.O. was recipient of an Undergraduate fellowship from UNESP—Universidade Estadual Paulista, and M.I.T.F. and J.A.F. are recipients of Productivity Grants from CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico).
We thank Dave Lane (LMGM/CNRS) and all the members of the Laboratory of Molecular Biology of the Department of Technology (UNESP-Universidade Estadual Paulista).
We would also like to thank Mathieu Arlat, Bernard Hallet and Marshal Stark for comments which greatly improved the manuscript.
Footnotes
Citation Marini Ferreira R, de Oliveira ACP, Moreira LM, Belasque J, Jr, Gourbeyre E, Siguier P, Ferro MIT, Ferro JA, Chandler M, Varani AM. 2015. A TALE of transposition: Tn3-like transposons play a major role in the spread of pathogenicity determinants of Xanthomonas citri and other xanthomonads. mBio 6(1):e02505-14. doi:10.1128/mBio.02505-14.
REFERENCES
- 1.Mansfield J, Genin S, Magori S, Citovsky V, Sriariyanum M, Ronald P, Dow M, Ronald P, Dow M, Verdier V, Beer SV, Machado MA, Toth I, Salmond G, Foster GD. 2012. Top 10 plant pathogenic bacteria in molecular plant pathology. Mol Plant Pathol 13:614–629. doi: 10.1111/j.1364-3703.2012.00804.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Jalan N, Yan Q, Kogenaru S, Guo Y, Jones JB, Graham JH, Wang N. 2014. Genomics of Xanthomonas citri and related species, p 151–176. In Gross DC, Lichens-Park A, Kole C (ed), Genomics of plant-associated bacteria. Springer, Berlin, Germany. [Google Scholar]
- 3.Triplett L, Koebnik R, Verdier V, Leach JE. 2014. The genomics of Xanthomonas oryzae, p 127–150. In Gross DC, Lichens-Park A, Kole C (ed), Genomics of plant-associated bacteria. Springer, Berlin, Germany. [Google Scholar]
- 4.Pothier JF, Vorhölter F-J, Blom J, Goesmann A, Pühler A, Smits THM, Duffy B. 2011. The ubiquitous plasmid pXap41 in the invasive phytopathogen Xanthomonas arboricola pv. pruni: complete sequence and comparative genomic analysis. FEMS Microbiol Lett 323:52–60. doi: 10.1111/j.1574-6968.2011.02352.x. [DOI] [PubMed] [Google Scholar]
- 5.Darrasse A, Carrère S, Barbe V, Boureau T, Arrieta-Ortiz ML, Bonneau S, Briand M, Brin C, Cociancich S, Durand K, Fouteau S, Gagnevin L, Guérin F, Guy E, Indiana A, Koebnik R, Lauber E, Munoz A, Noël LD, Pieretti I, Poussier S, Pruvost O, Robène-Soustrade I, Rott P, Royer M, Serres-Giardi L, Szurek B, van Sluys M-A, Verdier V, Vernière V, Arlat M, Manceau C, Jacques MA. 2013. Genome sequence of Xanthomonas fuscans subsp. fuscans strain 4834-R reveals that flagellar motility is not a general feature of xanthomonads. BMC Genomics 14:761. doi: 10.1186/1471-2164-14-761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Da Silva ACR, Ferro JA, Reinach FC, Farah CS, Furlan LR, Quaggio RB, Monteiro-Vitorello CB, Van Sluys MA, Almeida NF, Alves LMC, Do Amaral AM, Bertolini MC, Camargo LEA, Camarotte G, Cannavan F, Cardozo J, Chambergo F, Ciapina LP, Cicarelli RMB, Coutinho LL, Cursino-Santos JR, El-Dorry H, Faria JB, Ferreira AJS, Ferreira RCC, Ferro MIT, Formighieri EF, Franco MC, Greggio CC, Gruber A, Katsuyama AM, Kishi LT, Leite RP, Lemos EGM, Lemos MVF, Locali EC, Machado MA, Madeira AMBN, Martinez-Rossi NM, Martins EC, Meidanis J, Menck CFM, Miyaki CY, Moon DH, Moreira LM, Novo MTM, Okura VK, Oliveira MC, Oliveira VR, Pereira HA, et al.. 2002. Comparison of the genomes of two Xanthomonas pathogens with differing host specificities. Nature 417:459–463. doi: 10.1038/417459a. [DOI] [PubMed] [Google Scholar]
- 7.Moreira LM, Almeida NF Jr, Potnis N, Digiampietri LA, Adi SS, Bortolossi JC, da Silva AC, da Silva AM, de Moraes FE, de Oliveira JC, de Souza RF, Facincani AP, Ferraz AL, Ferro MI, Furlan LR, Gimenez DF, Jones JB, Kitajima EW, Laia ML, Leite RP Jr, Nishiyama MY, Rodrigues Neto J, Nociti LA, Norman DJ, Ostroski EH, Pereira HA Jr, Staskawicz BJ, Tezza RI, Ferro JA, Vinatzer BA, Setubal JC. 2010. Novel insights into the genomic basis of citrus canker based on the genome sequences of two strains of Xanthomonas fuscans subsp. aurantifolii. BMC Genomics 11:238. doi: 10.1186/1471-2164-11-238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ryan RP, Vorhölter F-J, Potnis N, Jones JB, Van Sluys M-A, Bogdanove AJ, DOW JM. 2011. Pathogenomics of Xanthomonas: understanding bacterium-plant interactions. Nat Rev Microbiol 9:344–355. doi: 10.1038/nrmicro2558. [DOI] [PubMed] [Google Scholar]
- 9.Galán JE, Collmer A. 1999. Type III secretion machines: bacterial devices for protein delivery into host cells. Science 284:1322–1328. doi: 10.1126/science.284.5418.1322. [DOI] [PubMed] [Google Scholar]
- 10.Cornelis GR, Van Gijsegem F. 2000. Assembly and function of type III secretory systems. Annu Rev Microbiol 54:735–774. doi: 10.1146/annurev.micro.54.1.735. [DOI] [PubMed] [Google Scholar]
- 11.Rossier O, Wengelnik K, Hahn K, Bonas U. 1999. The Xanthomonas Hrp type III system secretes proteins from plant and mammalian bacterial pathogens. Proc Natl Acad Sci U S A 96:9368–9373. doi: 10.1073/pnas.96.16.9368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Büttner D, He SY. 2009. Type III protein secretion in plant pathogenic Bacteria. Plant Physiol 150:1656–1664. doi: 10.1104/pp.109.139089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kay S, Bonas U. 2009. How Xanthomonas type III effectors manipulate the host plant. Curr Opin Microbiol 12:37–43. doi: 10.1016/j.mib.2008.12.006. [DOI] [PubMed] [Google Scholar]
- 14.Bogdanove AJ, Schornack S, Lahaye T. 2010. TAL effectors: finding plant genes for disease and defense. Curr Opin Plant Biol 13:394–401. doi: 10.1016/j.pbi.2010.04.010. [DOI] [PubMed] [Google Scholar]
- 15.Lima WC, Paquola ACM, Varani AM, Van Sluys M-A, Menck CFM. 2008. Laterally transferred genomic islands in Xanthomonadales related to pathogenicity and primary metabolism. FEMS Microbiol Lett 281:87–97. doi: 10.1111/j.1574-6968.2008.01083.x. [DOI] [PubMed] [Google Scholar]
- 16.Monteiro-Vitorello CB, de Oliveira MC, Zerillo MM, Varani AM, Civerolo E, Van Sluys MA. 2005. Xylella and Xanthomonas Mobil’omics. Omics 9:146–159. doi: 10.1089/omi.2005.9.146. [DOI] [PubMed] [Google Scholar]
- 17.Varani AM, Monteiro-Vitorello CB, Nakaya HI, Van Sluys M-A. 2013. The role of prophage in plant pathogenic bacteria. Annu Rev Phytopathol 51:429–451. doi: 10.1146/annurev-phyto-081211-173010. [DOI] [PubMed] [Google Scholar]
- 18.Boch J, Bonas U. 2010. Xanthomonas AvrBs3 family-type III effectors: discovery and function. Annu Rev Phytopathol 48:419–436. doi: 10.1146/annurev-phyto-080508-081936. [DOI] [PubMed] [Google Scholar]
- 19.Siguier P, Varani A, Perochon J, Chandler M. 2012. Exploring bacterial insertion sequences with ISfinder: objectives, uses, and future developments. Methods Mol Biol 859:91–103. doi: 10.1007/978-1-61779-603-6_5. [DOI] [PubMed] [Google Scholar]
- 20.Grau J, Wolf A, Reschke M, Bonas U, Posch S, Boch J. 2013. Computational predictions provide insights into the biology of TAL effector target sites. PLoS Comput Biol 9:e1002962. doi: 10.1371/journal.pcbi.1002962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Moscou MJ, Bogdanove AJ. 2009. A simple cipher governs DNA recognition by TAL effectors. Science 326:1501. doi: 10.1126/science.1178817. [DOI] [PubMed] [Google Scholar]
- 22.Shiotani H, Fujikawa T, Ishihara H, Tsuyumu S, Ozaki K. 2007. A pthA homolog from Xanthomonas axonopodis pv. citri responsible for host-specific suppression of virulence. J Bacteriol 189:3271–3279. doi: 10.1128/JB.01790-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Streubel J, Blücher C, Landgraf A, Boch J. 2012. TAL effector RVD specificities and efficiencies. Nat Biotechnol 30:593–595. doi: 10.1038/nbt.2304. [DOI] [PubMed] [Google Scholar]
- 24.Boch J, Scholze H, Schornack S, Landgraf A, Hahn S, Kay S, Lahaye T, Nickstadt A, Bonas U. 2009. Breaking the code of DNA binding specificity of TAL-type III effectors. Science 326:1509–1512. doi: 10.1126/science.1178811. [DOI] [PubMed] [Google Scholar]
- 25.Moore R, Chandrahas A, Bleris L. 2014. Transcription activator-like effectors: a toolkit for synthetic biology. ACS Synth Biol 3:708–716. doi: 10.1021/sb400137b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Grindley NDF. 2002. The movement of Tn3-like elements: transposition and cointegrate resolution, p 272–302. In Craig N, Craigie R, Gellert M, Lambowitz A (ed), Mobile DNA II. ASM Press, Washington, DC. [Google Scholar]
- 27.Ohta S, Tsuchida K, Choi S, Sekine Y, Shiga Y, Ohtsubo E. 2002. Presence of a characteristic D-D-E motif in IS1 transposase. J Bacteriol 184:6146–6154. doi: 10.1128/JB.184.22.6146-6154.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Partridge SR, Hall RM. 2004. Complex multiple antibiotic and mercury resistance region derived from the r-det of NR1 (R100). Antimicrob Agents Chemother 48:4250–4255. doi: 10.1128/AAC.48.11.4250-4255.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kholodii G, Yurieva O, Mindlin S, Gorlenko Z, Rybochkin V, Nikiforov V. 2000. Tn5044, a novel Tn3 family transposon coding for temperature-sensitive mercury resistance. Res Microbiol 151:291–302. doi: 10.1016/S0923-2508(00)00149-2. [DOI] [PubMed] [Google Scholar]
- 30.Chiou CS, Jones AL. 1993. Nucleotide sequence analysis of a transposon (Tn5393) carrying streptomycin resistance genes in Erwinia amylovora and other gram-negative bacteria. J Bacteriol 175:732–740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Voloudakis AE, Reignier TM, Cooksey DA. 2005. Regulation of resistance to copper in Xanthomonas axonopodis pv. vesicatoria. Appl Environ Microbiol 71:782–789. doi: 10.1128/AEM.71.2.782-789.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Vanhooff V, Galloy C, Agaisse H, Lereclus D, Révet B, Hallet B. 2006. Self-control in DNA site-specific recombination mediated by the tyrosine recombinase TnpI. Mol Microbiol 60:617–629. doi: 10.1111/j.1365-2958.2006.05127.x. [DOI] [PubMed] [Google Scholar]
- 33.Yano H, Genka H, Ohtsubo Y, Nagata Y, Top EM, Tsuda M. 2013. Cointegrate-resolution of toluene-catabolic transposon Tn4651: determination of crossover site and the segment required for full resolution activity. Plasmid 69:24–35. doi: 10.1016/j.plasmid.2012.07.004. [DOI] [PubMed] [Google Scholar]
- 34.Di Gioia D, Peel M, Fava F, Wyndham RC. 1998. Structures of homologous composite transposons carrying cbaABC genes from Europe and North America. Appl Environ Microbiol 64:1940–1946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Triplett LR, Hamilton JP, Buell CR, Tisserat NA, Verdier V, Zink F, Leach JE. 2011. Genomic analysis of Xanthomonas oryzae isolates from rice grown in the United States reveals substantial divergence from known X. oryzae pathovars. Appl Environ Microbiol 77:3930–3937. doi: 10.1128/AEM.00028-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Garcillán-Barcia MP, Francia MV, de la Cruz F. 2009. The diversity of conjugative relaxases and its application in plasmid classification. FEMS Microbiol Rev 33:657–687. doi: 10.1111/j.1574-6976.2009.00168.x. [DOI] [PubMed] [Google Scholar]
- 37.El Yacoubi B, Brunings AM, Yuan Q, Shankar S, Gabriel DW. 2007. In planta horizontal transfer of a major pathogenicity effector gene. Appl Environ Microbiol 73:1612–1621. doi: 10.1128/AEM.00261-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Noël L, Thieme F, Gäbler J, Büttner D, Bonas U. 2003. XopC and XopJ, two novel type III effector proteins from Xanthomonas campestris pv. vesicatoria. J Bacteriol 185:7092–7102. doi: 10.1128/JB.185.24.7092-7102.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Jalan N, Kumar D, Andrade MO, Yu F, Jones JB, Graham JH, White FF, Setubal JC, Wang N. 2013. Comparative genomic and transcriptome analyses of pathotypes of Xanthomonas citri subsp. citri provide insights into mechanisms of bacterial virulence and host range. BMC Genomics 14:551. doi: 10.1186/1471-2164-14-551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Thieme F, Szczesny R, Urban A, Kirchner O, Hause G, Bonas U. 2007. New type III effectors from Xanthomonas campestris pv. vesicatoria trigger plant reactions dependent on a conserved N-myristoylation motif. Mol Plant Microbe Interact 20:1250–1261. doi: 10.1094/MPMI-20-10-1250. [DOI] [PubMed] [Google Scholar]
- 41.Laia ML, Moreira LM, Dezajacomo J, Brigati JB, Ferreira CB, Ferro MI, Silva AC, Ferro JA, Oliveira JC. 2009. New genes of Xanthomonas citri subsp. citri involved in pathogenesis and adaptation revealed by a transposon-based mutant library. BMC Microbiol 9:12. doi: 10.1186/1471-2180-9-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Oh H-S, Kvitko BH, Morello JE, Collmer A. 2007. Pseudomonas syringae lytic transglycosylases coregulated with the type III secretion system contribute to the translocation of effector proteins into plant cells. J Bacteriol 189:8277–8289. doi: 10.1128/JB.00998-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hu Y, Zhang J, Jia H, Sosso D, Li T, Frommer WB, Yang B, White FF, Wang N, Jones JB. 2014. Lateral organ boundaries 1 is a disease susceptibility gene for citrus bacterial canker disease. Proc Natl Acad Sci U. S. A. 111:201313271. doi: 10.1073/pnas.1313271111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Nicaise V, Joe A, Jeong BR, Korneli C, Boutrot F, Westedt I, Staiger D, Alfano JR, Zipfel C. 2013. Pseudomonas HopU1 modulates plant immune receptor levels by blocking the interaction of their mRNAs with GRP7. EMBO J 32:701–712. doi: 10.1038/emboj.2013.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Yu Y, Streubel J, Balzergue S, Champion A, Boch J, Koebnik R, Feng J, Verdier V, Szurek B. 2011. Colonization of rice leaf blades by an African strain of Xanthomonas oryzae pv. oryzae depends on a new TAL effector that induces the rice nodulin-3 Os11N3 gene. Mol Plant Microbe Interact 24:1102–1113. doi: 10.1094/MPMI-11-10-0254. [DOI] [PubMed] [Google Scholar]
- 46.Pereira AL, Carazzolle MF, Abe VY, de Oliveira ML, Domingues MN, Silva JC, Cernadas RA, Benedetti CE. 2014. Identification of putative TAL effector targets of the citrus canker pathogens shows functional convergence underlying disease development and defense response. BMC Genomics 15:157. doi: 10.1186/1471-2164-15-157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Chen Y, Braathen P, Léonard C, Mahillon J. 1999. MIC231, a naturally occurring mobile insertion cassette from Bacillus cereus. Mol Microbiol 32:657–668. doi: 10.1046/j.1365-2958.1999.01388.x. [DOI] [PubMed] [Google Scholar]
- 48.Meyer J, Iida S. 1979. Amplification of chloramphenicol resistance transposons carried by phage P1Cm in Escherichia coli. Mol Gen Genet 176:209–219. [DOI] [PubMed] [Google Scholar]
- 49.Chandler M, de la Tour EB, Willems D, Caro L. 1979. Some properties of the chloramphenicol resistance transposon Tn9. Mol Gen Genet 176:221–231. [DOI] [PubMed] [Google Scholar]
- 50.Zaborin A, Gerdes S, Holbrook C, Liu DC, Zaborina OY, Alverdy JC. 2012. Pseudomonas aeruginosa overrides the virulence inducing effect of opioids when it senses an abundance of phosphate. PLoS One 7:e34883. doi: 10.1371/journal.pone.0034883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Bains M, Fernández L, Hancock REW. 2012. Phosphate starvation promotes swarming motility and cytotoxicity of Pseudomonas aeruginosa. Appl Environ Microbiol 78:6762–6768. doi: 10.1128/AEM.01015-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Pages D, Rose J, Conrod S, Cuine S, Carrier P, Heulin T, Achouak W. 2008. Heavy metal tolerance in Stenotrophomonas maltophilia. PLoS One 3:e1539. doi: 10.1371/journal.pone.0001539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Ryan RP, Monchy S, Cardinale M, Taghavi S, Crossman L, Avison MB, Berg G, van der Lelie D, DOW JM. 2009. The versatility and adaptation of bacteria from the genus Stenotrophomonas. Nat Rev Microbiol 7:514–525. doi: 10.1038/nrmicro2163. [DOI] [PubMed] [Google Scholar]
- 54.Kado CI. 2010. Plant bacteriology. American Phytopathological Society, St. Paul, MN. [Google Scholar]
- 55.Siguier P, Perochon J, Lestrade L, Mahillon J, Chandler M. 2006. ISfinder: the reference centre for bacterial insertion sequences. Nucleic Acids Res 34:D32–D36. doi: 10.1093/nar/gkj014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Varani AM, Siguier P, Gourbeyre E, Charneau V, Chandler M. 2011. ISsaga is an ensemble of web-based methods for high throughput identification and semi-automatic annotation of insertion sequences in prokaryotic genomes. Genome Biol 12:R30. doi: 10.1186/gb-2011-12-3-r30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Altschul SF, Madden TL, Schäffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ. 1997. Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402. doi: 10.1093/nar/25.17.3389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Kumar S, Tamura K, Nei M. 2004. MEGA3: integrated software for Molecular Evolutionary Genetics Analysis and sequence alignment. Brief Bioinform 5:150–163. doi: 10.1093/bib/5.2.150. [DOI] [PubMed] [Google Scholar]
- 59.Felsenstein J. 1997. An alternating least squares approach to inferring phylogenies from pairwise distances. Syst Biol 46:101–111. doi: 10.1093/sysbio/46.1.101. [DOI] [PubMed] [Google Scholar]
- 60.Tamura K, Stecher G, Peterson D, Filipski A, Kumar S. 2013. MEGA6: Molecular Evolutionary Genetics Analysis version 6.0. Mol Biol Evol 30:2725–2729. doi: 10.1093/molbev/mst197. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Linear genomic representation of the distribution of the IRs and potential MIC structures, TALEs, and TnXax1 relatives. (A) Genomic representation of all plasmids. (B) Genomic representation in the main chromosomes. Abbreviations: Xcv pXCV183, X. campestris pv. vesicatoria strain 85-10 plasmid pXCV183; XccA(w) pXcaw58, X. citri subsp. citri strain Aw 12879; Xff, X. fuscans subsp. fuscans strain 4834-R; Xff pla, X. fuscans subsp. fuscans strain 4834-R plasmid pla; Xff plc, X. fuscans subsp. fuscans strain 4834-R plasmid plc; Xap pXap41, X. arboricola pv. pruni plasmid pXap41; Xc pXcb, X. citri plasmid pXcB; Xag 8ra pXAG81, X. axonopodis pv. glycines strain 8ra plasmid pXAG81; Xa GPE PC73, X. albilineans strain GPE PC73 plasmid; Xag AG1 pAG1, X. axonopodis pv. glycines strain AG1 plasmid pAG1; A. citrulli, Acidovorax avenae subsp. citrulli strain AAC00-1; Xcc ATCC 33913, X. campestris pv. campestris strain ATCC 33913; Xcc B100, X. campestris pv. campestris strain B100; Xcc 8004, X. campestris pv. campestris strain 8004; Xa GPE PC73, X. albilineans strain GPE PC73. Download
List of occurrences of the TnXax1 full-length IRs in Xanthomonadales (Gammaproteobacteria) sequences deposited in GenBank (excluding draft or incomplete genomes). The genome coordinates represent the orientation of each IR. The numbers of positions in the identified sequences that are conserved with respect to the 92 bp right IR of TnXax1 are shown in the third column. This table also shows degenerate IRs that maintain the GAGGG tips and vary from 33 bp to 92 bp. These TnXax1 IRs are also found forming a potential MIC element in a unique Betaproteobacteria genome, that of Acidovorax avenae subsp. citrulli strain AAC00-1, a watermelon pathogen.
Tn3-like transposable elements with IRs (GAGGG tips) similar to those of TnXax1, including the transposition and passenger genes identified in TnPa43 from Pseudomonas aeruginosa (A), TnStma1 from Stenotrophomonas maltophilia strain D457 (B), and TnXca1 from X. campestris pv. vesicatoria plasmid pXCV183 (C). The grey-shaded rows indicate the transposition-related genes. The coordinates represent the position of each open reading frame in the transposon itself.
Potential MIC structures and their genomic features and gene content.
Distribution, genomic coordinates, and genome context of the mlt, TnpA, TnpS, resolution site (res site), and TnpT homologues (>40% DNA sequence identity) over those of other Xanthomonas species that do not carry the TnXax1 canonical element or relatives. The identity values were calculated based on the canonical sequence of TnXax1 from X. citri subsp. citri strain 306 pXAC64. The genome context is shown inside the brackets and represents the open reading frame located up- and downstream.