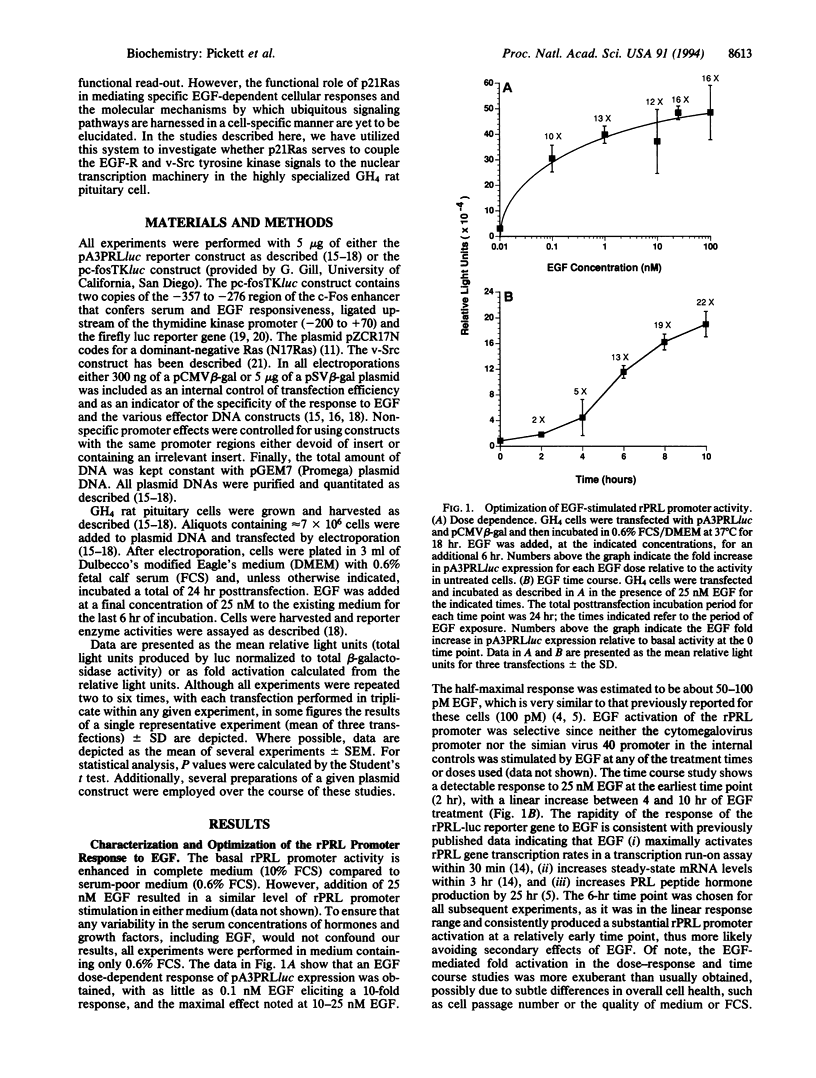
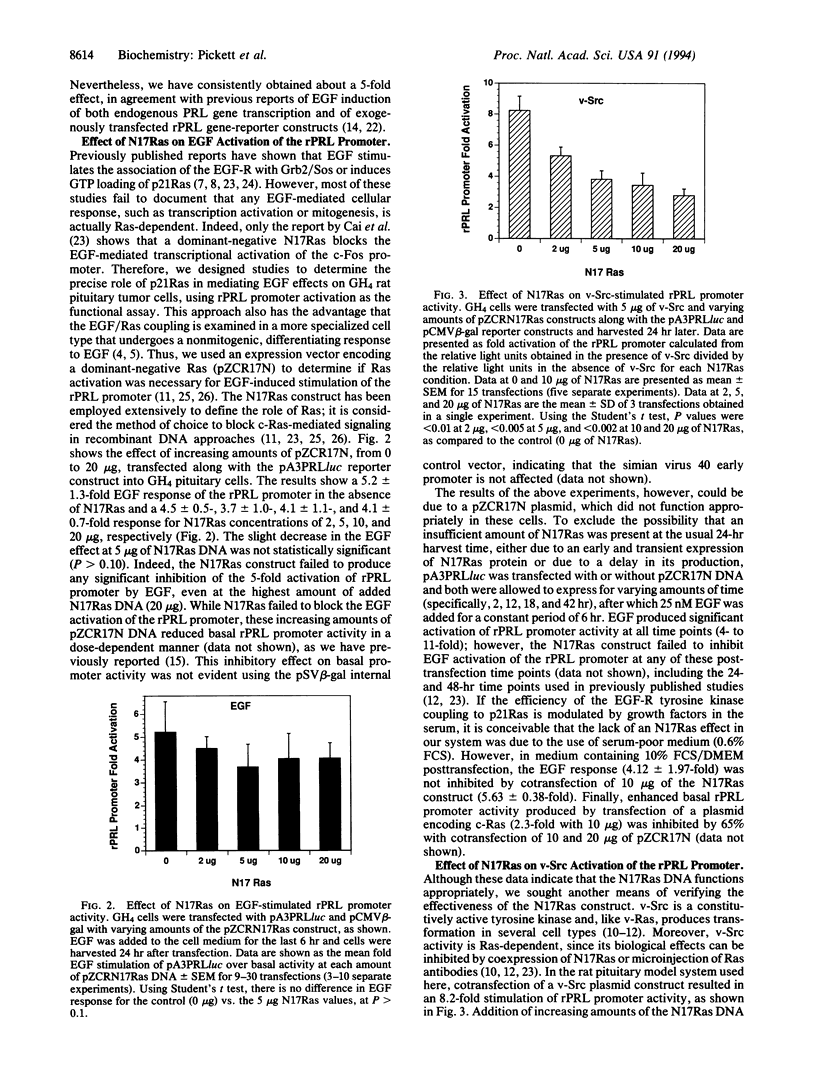
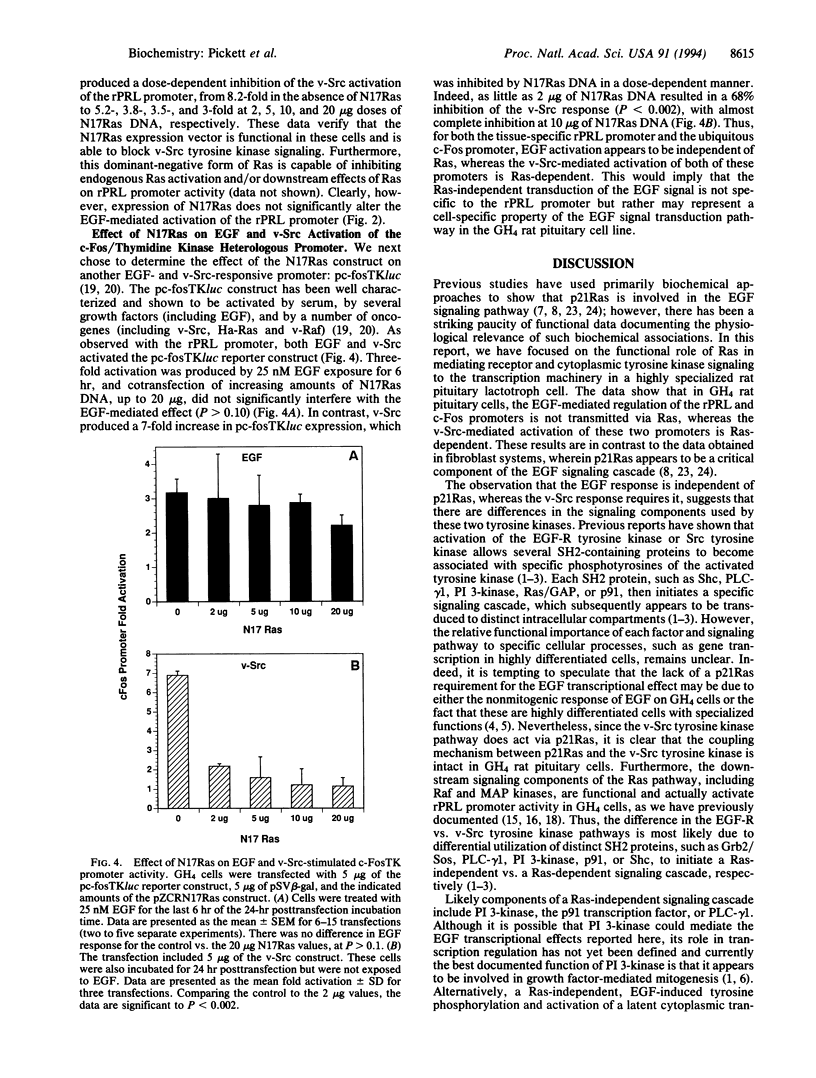
Abstract
p21Ras has been implicated as a critical signaling component in mediating the effects of many growth factor receptor/tyrosine kinases on cell growth and differentiation. However, the precise functional role of Ras in establishing a cell-specific transcriptional response to a ubiquitous growth factor remains unclear. We have utilized a transient cotransfection model system in epidermal growth factor (EGF)-responsive cultured GH4 rat pituitary neuroendocrine cells to investigate the role of Ras in coupling EGF receptor (EGF-R) and v-Src tyrosine kinase signals to the activation of a cell-specific promoter for the rat (r) prolactin (PRL) gene. A significant dose- and time-dependent EGF stimulation of the transfected rPRL promoter was obtained. A similar degree of activation of the rPRL promoter was obtained by cotransfection of a plasmid encoding v-Src. Cotransfection of a construct encoding the dominant-negative Ras, N17Ras, produced almost complete inhibition of v-Src-induced rPRL promoter activity, while EGF-stimulated rPRL promoter activity was unaffected. Similarly, EGF activation of a c-Fos promoter was unaffected by N17Ras, while v-Src activation was blocked. Hence, using transcription regulation as a functional assay, we show that Ras is not required for the EGF-mediated control of the rPRL and c-Fos promoters, whereas Ras is critical in mediating the v-Src effects to these two promoters. These observations emphasize that, despite current biochemical data linking the EGF-R and Ras pathways, the functional significance of such an interaction should be analyzed in a biologically relevant manner and may differ as a function of cell type.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Cai H., Szeberényi J., Cooper G. M. Effect of a dominant inhibitory Ha-ras mutation on mitogenic signal transduction in NIH 3T3 cells. Mol Cell Biol. 1990 Oct;10(10):5314–5323. doi: 10.1128/mcb.10.10.5314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen W. S., Lazar C. S., Poenie M., Tsien R. Y., Gill G. N., Rosenfeld M. G. Requirement for intrinsic protein tyrosine kinase in the immediate and late actions of the EGF receptor. 1987 Aug 27-Sep 2Nature. 328(6133):820–823. doi: 10.1038/328820a0. [DOI] [PubMed] [Google Scholar]
- Conrad K. E., Gutierrez-Hartmann A. The ras and protein kinase A pathways are mutually antagonistic in regulating rat prolactin promoter activity. Oncogene. 1992 Jul;7(7):1279–1286. [PubMed] [Google Scholar]
- Conrad K. E., Oberwetter J. M., Vaillancourt R., Johnson G. L., Gutierrez-Hartmann A. Identification of the functional components of the Ras signaling pathway regulating pituitary cell-specific gene expression. Mol Cell Biol. 1994 Mar;14(3):1553–1565. doi: 10.1128/mcb.14.3.1553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elsholtz H. P., Mangalam H. J., Potter E., Albert V. R., Supowit S., Evans R. M., Rosenfeld M. G. Two different cis-active elements transfer the transcriptional effects of both EGF and phorbol esters. Science. 1986 Dec 19;234(4783):1552–1557. doi: 10.1126/science.3491428. [DOI] [PubMed] [Google Scholar]
- Farnsworth C. L., Feig L. A. Dominant inhibitory mutations in the Mg(2+)-binding site of RasH prevent its activation by GTP. Mol Cell Biol. 1991 Oct;11(10):4822–4829. doi: 10.1128/mcb.11.10.4822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feig L. A., Cooper G. M. Inhibition of NIH 3T3 cell proliferation by a mutant ras protein with preferential affinity for GDP. Mol Cell Biol. 1988 Aug;8(8):3235–3243. doi: 10.1128/mcb.8.8.3235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feig L. A., Cooper G. M. Relationship among guanine nucleotide exchange, GTP hydrolysis, and transforming potential of mutated ras proteins. Mol Cell Biol. 1988 Jun;8(6):2472–2478. doi: 10.1128/mcb.8.6.2472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gale N. W., Kaplan S., Lowenstein E. J., Schlessinger J., Bar-Sagi D. Grb2 mediates the EGF-dependent activation of guanine nucleotide exchange on Ras. Nature. 1993 May 6;363(6424):88–92. doi: 10.1038/363088a0. [DOI] [PubMed] [Google Scholar]
- Gulbins E., Coggeshall K. M., Baier G., Katzav S., Burn P., Altman A. Tyrosine kinase-stimulated guanine nucleotide exchange activity of Vav in T cell activation. Science. 1993 May 7;260(5109):822–825. doi: 10.1126/science.8484124. [DOI] [PubMed] [Google Scholar]
- Gupta S. K., Gallego C., Johnson G. L., Heasley L. E. MAP kinase is constitutively activated in gip2 and src transformed rat 1a fibroblasts. J Biol Chem. 1992 Apr 25;267(12):7987–7990. [PubMed] [Google Scholar]
- Gutman A., Wasylyk B. Nuclear targets for transcription regulation by oncogenes. Trends Genet. 1991 Feb;7(2):49–54. doi: 10.1016/0168-9525(91)90231-E. [DOI] [PubMed] [Google Scholar]
- Hernández-Sotomayor S. M., Carpenter G. Epidermal growth factor receptor: elements of intracellular communication. J Membr Biol. 1992 Jun;128(2):81–89. doi: 10.1007/BF00231881. [DOI] [PubMed] [Google Scholar]
- Jackson S. M., Keech C. A., Williamson D. J., Gutierrez-Hartmann A. Interaction of basal positive and negative transcription elements controls repression of the proximal rat prolactin promoter in nonpituitary cells. Mol Cell Biol. 1992 Jun;12(6):2708–2719. doi: 10.1128/mcb.12.6.2708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson L. K., Baxter J. D., Vlodavsky I., Gospodarowicz D. Epidermal growth factor and expression of specific genes: effects on cultured rat pituitary cells are dissociable from the mitogenic response. Proc Natl Acad Sci U S A. 1980 Jan;77(1):394–398. doi: 10.1073/pnas.77.1.394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koch C. A., Anderson D., Moran M. F., Ellis C., Pawson T. SH2 and SH3 domains: elements that control interactions of cytoplasmic signaling proteins. Science. 1991 May 3;252(5006):668–674. doi: 10.1126/science.1708916. [DOI] [PubMed] [Google Scholar]
- Li N., Batzer A., Daly R., Yajnik V., Skolnik E., Chardin P., Bar-Sagi D., Margolis B., Schlessinger J. Guanine-nucleotide-releasing factor hSos1 binds to Grb2 and links receptor tyrosine kinases to Ras signalling. Nature. 1993 May 6;363(6424):85–88. doi: 10.1038/363085a0. [DOI] [PubMed] [Google Scholar]
- Murdoch G. H., Potter E., Nicolaisen A. K., Evans R. M., Rosenfeld M. G. Epidermal growth factor rapidly stimulates prolactin gene transcription. Nature. 1982 Nov 11;300(5888):192–194. doi: 10.1038/300192a0. [DOI] [PubMed] [Google Scholar]
- Oberwetter J. M., Conrad K. E., Gutierrez-Hartmann A. The Ras and protein kinase C signaling pathways are functionally antagonistic in GH4 neuroendocrine cells. Mol Endocrinol. 1993 Jul;7(7):915–923. doi: 10.1210/mend.7.7.8413316. [DOI] [PubMed] [Google Scholar]
- Ohmichi M., Sawada T., Kanda Y., Koike K., Hirota K., Miyake A., Saltiel A. R. Thyrotropin-releasing hormone stimulates MAP kinase activity in GH3 cells by divergent pathways. Evidence of a role for early tyrosine phosphorylation. J Biol Chem. 1994 Feb 4;269(5):3783–3788. [PubMed] [Google Scholar]
- Osborne R., Tashjian A. H., Jr Tumor-promoting phorbol esters affect production of prolactin and growth hormone by rat pituitary cells. Endocrinology. 1981 Apr;108(4):1164–1170. doi: 10.1210/endo-108-4-1164. [DOI] [PubMed] [Google Scholar]
- Pellegrini S., Schindler C. Early events in signalling by interferons. Trends Biochem Sci. 1993 Sep;18(9):338–342. doi: 10.1016/0968-0004(93)90070-4. [DOI] [PubMed] [Google Scholar]
- Qureshi S. A., Alexandropoulos K., Rim M., Joseph C. K., Bruder J. T., Rapp U. R., Foster D. A. Evidence that Ha-Ras mediates two distinguishable intracellular signals activated by v-Src. J Biol Chem. 1992 Sep 5;267(25):17635–17639. [PubMed] [Google Scholar]
- Raffioni S., Bradshaw R. A. Activation of phosphatidylinositol 3-kinase by epidermal growth factor, basic fibroblast growth factor, and nerve growth factor in PC12 pheochromocytoma cells. Proc Natl Acad Sci U S A. 1992 Oct 1;89(19):9121–9125. doi: 10.1073/pnas.89.19.9121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sadowski H. B., Shuai K., Darnell J. E., Jr, Gilman M. Z. A common nuclear signal transduction pathway activated by growth factor and cytokine receptors. Science. 1993 Sep 24;261(5129):1739–1744. doi: 10.1126/science.8397445. [DOI] [PubMed] [Google Scholar]
- Satoh T., Endo M., Nakafuku M., Akiyama T., Yamamoto T., Kaziro Y. Accumulation of p21ras.GTP in response to stimulation with epidermal growth factor and oncogene products with tyrosine kinase activity. Proc Natl Acad Sci U S A. 1990 Oct;87(20):7926–7929. doi: 10.1073/pnas.87.20.7926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlessinger J., Ullrich A. Growth factor signaling by receptor tyrosine kinases. Neuron. 1992 Sep;9(3):383–391. doi: 10.1016/0896-6273(92)90177-f. [DOI] [PubMed] [Google Scholar]
- Schonbrunn A., Krasnoff M., Westendorf J. M., Tashjian A. H., Jr Epidermal growth factor and thyrotropin-releasing hormone act similarly on a clonal pituitary cell strain. Modulation of hormone production and inhbition of cell proliferation. J Cell Biol. 1980 Jun;85(3):786–797. doi: 10.1083/jcb.85.3.786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith M. R., DeGudicibus S. J., Stacey D. W. Requirement for c-ras proteins during viral oncogene transformation. Nature. 1986 Apr 10;320(6062):540–543. doi: 10.1038/320540a0. [DOI] [PMC free article] [PubMed] [Google Scholar]


