Abstract
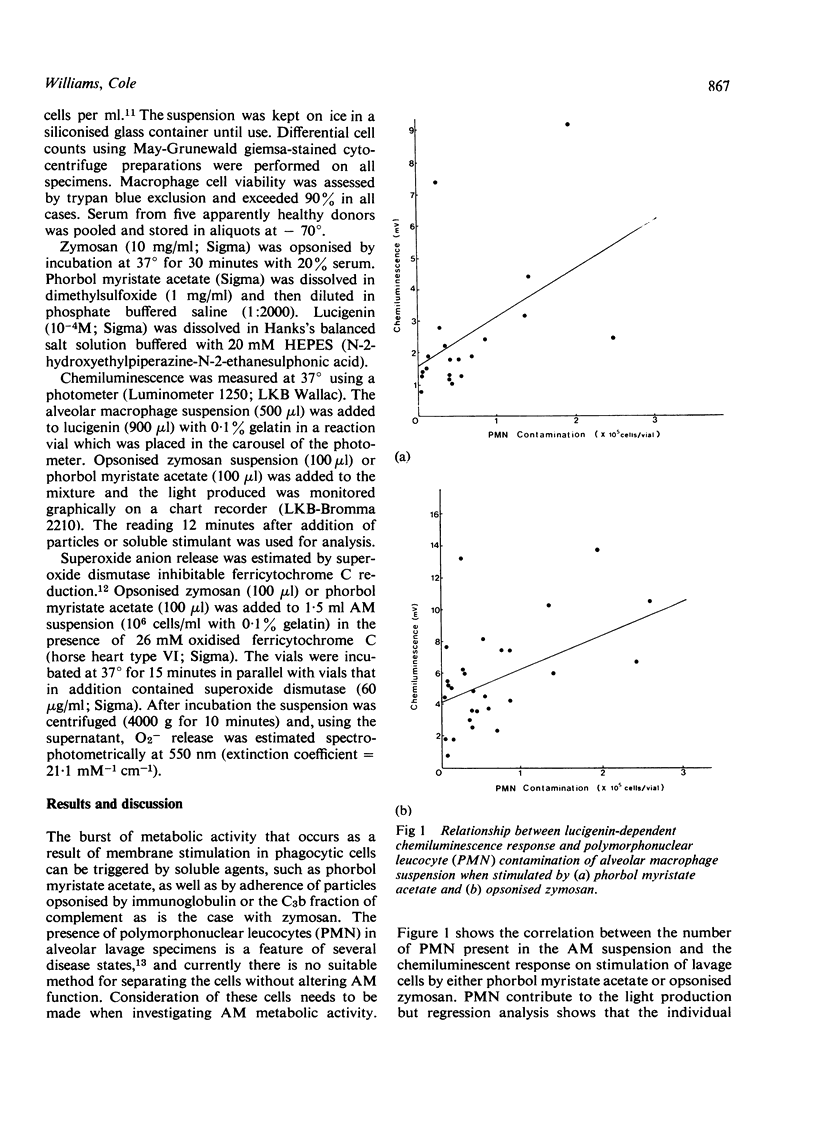
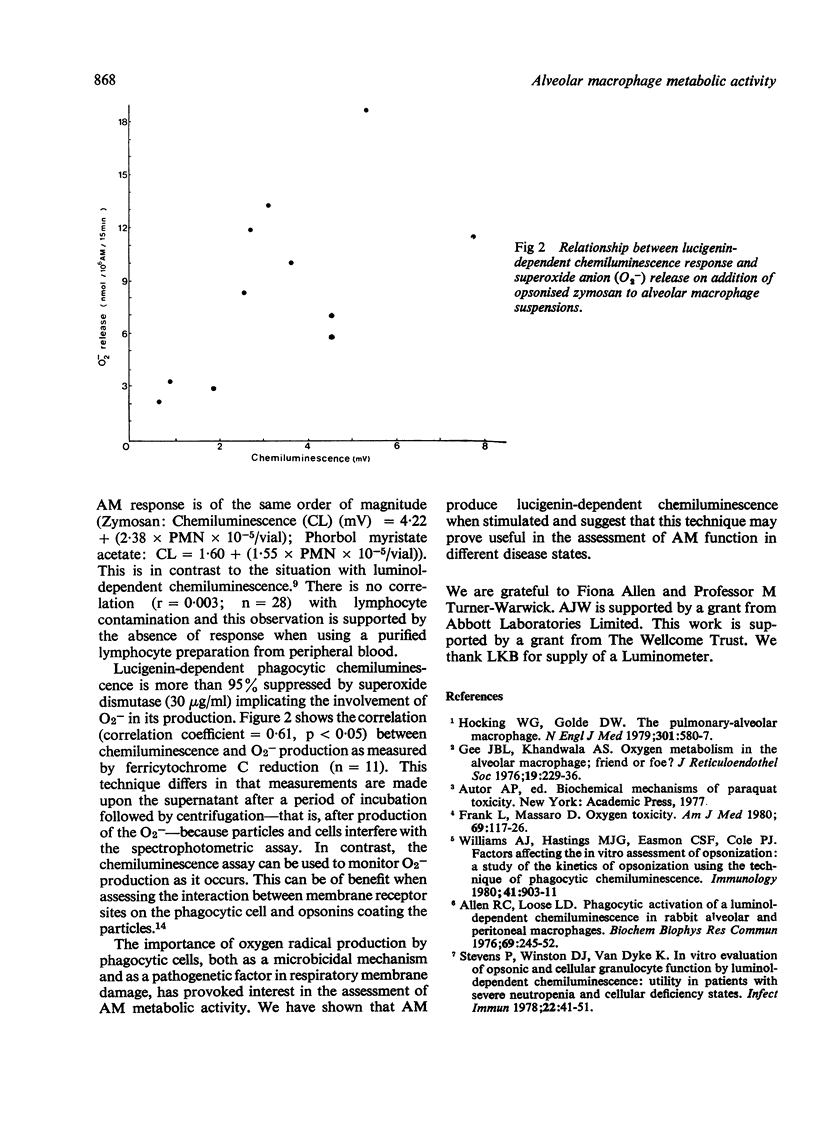
The method using lucigenin-dependent phagocytic chemiluminescence is described for the assessment of alveolar macrophage metabolic activity in response to stimulation by opsonised particles or soluble agents. The requirement for superoxide anion (O2-) in the production of chemiluminescence is suggested by inhibition (95%) using superoxide dismutase. The results obtained are correlated with those obtained using another method of detecting O2- release (r = 0.61; p less than 0.05) and are also related by regression analysis to polymorphonuclear leucocyte contamination of the alveolar macrophage suspension. This shows that alveolar macrophages produce lucigenin-dependent chemiluminescence of the same order of magnitude as do polymorphonuclear leucocytes.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Allen R. C. Evaluation of serum opsonic capacity by quantitating the initial chemiluminescent response from phagocytizing polymorphonuclear leukocytes. Infect Immun. 1977 Mar;15(3):828–833. doi: 10.1128/iai.15.3.828-833.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allen R. C., Loose L. D. Phagocytic activation of a luminol-dependent chemiluminescence in rabbit alveolar and peritoneal macrophages. Biochem Biophys Res Commun. 1976 Mar 8;69(1):245–252. doi: 10.1016/s0006-291x(76)80299-9. [DOI] [PubMed] [Google Scholar]
- Babior B. M., Kipnes R. S., Curnutte J. T. Biological defense mechanisms. The production by leukocytes of superoxide, a potential bactericidal agent. J Clin Invest. 1973 Mar;52(3):741–744. doi: 10.1172/JCI107236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bernard J., Gee J. B., Khandwala A. S. Oxygen metabolism in the alveolar macrophage: friend and foe? J Reticuloendothel Soc. 1976 Apr;19(4):229–236. [PubMed] [Google Scholar]
- Biggar W. D., Sturgess J. M. Peroxidase activity of alveolar macrophages. Lab Invest. 1976 Jan;34(1):31–42. [PubMed] [Google Scholar]
- Frank L., Massaro D. Oxygen toxicity. Am J Med. 1980 Jul;69(1):117–126. doi: 10.1016/0002-9343(80)90509-4. [DOI] [PubMed] [Google Scholar]
- GREENLEE L., FRIDOVICH I., HANDLER P. Chemiluminescence induced by operation of iron-flavoproteins. Biochemistry. 1962 Sep;1:779–783. doi: 10.1021/bi00911a008. [DOI] [PubMed] [Google Scholar]
- Hocking W. G., Golde D. W. The pulmonary-alveolar macrophage (first of two parts). N Engl J Med. 1979 Sep 13;301(11):580–587. doi: 10.1056/NEJM197909133011104. [DOI] [PubMed] [Google Scholar]
- Hunninghake G. W., Gadek J. E., Kawanami O., Ferrans V. J., Crystal R. G. Inflammatory and immune processes in the human lung in health and disease: evaluation by bronchoalveolar lavage. Am J Pathol. 1979 Oct;97(1):149–206. [PMC free article] [PubMed] [Google Scholar]
- Stevens P., Winston D. J., Van Dyke K. In vitro evaluation of opsonic and cellular granulocyte function by luminol-dependent chemiluminescence: utility in patients with severe neutropenia and cellular deficiency states. Infect Immun. 1978 Oct;22(1):41–51. doi: 10.1128/iai.22.1.41-51.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams A. J., Cole P. J. Human bronchoalveolar lavage cells and luminol-dependent chemiluminescence. J Clin Pathol. 1981 Feb;34(2):167–171. doi: 10.1136/jcp.34.2.167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams A. J., Hastings M. J., Easmon C. S., Cole P. J. Factor affecting the in vitro assessment of opsonization: a study of the kinetics of opsonization using the technique of phagocytic chemiluminescence. Immunology. 1980 Dec;41(4):903–911. [PMC free article] [PubMed] [Google Scholar]
- Yam L. T., Li C. Y., Crosby W. H. Cytochemical identification of monocytes and granulocytes. Am J Clin Pathol. 1971 Mar;55(3):283–290. doi: 10.1093/ajcp/55.3.283. [DOI] [PubMed] [Google Scholar]


