Versatility and specificity are usually mutually exclusive terms. However, as we discuss calcium's role in plant nutrition, we are obliged to contrast the plethora of general housekeeping functions of this element against the ability of calcium (Ca2+) to impart signaling specificity during biological responses (Marschner, 1995). Plants have evolved to rely on the unique properties of Ca2+ for a range of structural, enzymatic, and signaling functions. In this brief update, I will attempt to touch upon the various roles of Ca2+ in plant nutrition, growth, and development.
Ca2+ is a relatively large divalent cation, and in contrast to other macronutrients, a high proportion of total Ca2+ is found in the cell walls (apoplast). Some of this Ca2+ is associated to the cell wall, while another portion is exchangeable at the plasma membrane. Additionally, Ca2+ can be found at high concentrations within the vacuole of plant cells. In some tissues of particular plants, Ca2+ can be more than 10% of the dry weight and cause no deleterious effects to plant growth (Marschner, 1995). Juxtaposed against these concentrations of total calcium, the cytosolic-free Ca2+ activity within these cells remains around the 0.1 to 0.2 μm level. In this update, the first portion will detail the general role of Ca2+ in plant growth and development. In the middle section, we will focus on how almost imperceptible fluctuations in Ca2+ within the cytosol may be translated into plant growth and adaptation. This then leads to the final part of this broad review, to a discussion regarding how biologists are attempting to manipulate Ca2+ levels to improve plant productivity and human nutrition.
“EN GARDE”: CALCIUM PROVIDES MEMBRANE STABILITY AND STRESS TOLERANCE
While we need Ca2+ to build strong bones, plants need Ca2+ to strengthen cell walls and provide stress protection. When human diets are low in Ca2+, this leads to fragile bones or osteoporosis. With plants, soils or media deficient in Ca2+ can cause the disintegration of cell walls and the collapse of the affected tissues (Kirby and Pilbeam, 1984). Ca2+ deficiencies have been associated with bacterial diseases, fruit rotting, and other postharvest problems (Marschner, 1995). Thus, Ca2+ is cited for its beneficial effect on plant vigor and stiffness and also on grain and seed formation (Bennett, 1993). Increasing the Ca2+ content of fruits, for example, by spraying several times with Ca2+ salts during fruit development or by postharvest dipping in CaCl2 solution, leads to an increase in firmness of the fruit and can delay fruit ripening (Bramlage, 1994; Bramlage and Weis, 1994).
Ca2+ stabilizes cell membranes by connecting various proteins and lipids at membrane surfaces. Additionally, Ca2+ can be exchanged with other cations (K+, Na+, or H+) during stress responses. To protect the plasma membrane from various stresses, Ca2+ must always be present in the external solution, where it can regulate the selectivity of ion uptake. In fact, when plants are challenged with salinity stress, an increase in the concentration of Ca2+ often can ameliorate the inhibitory effect on growth (Epstein, 1972). In general, Ca2+ is involved in a plethora of plant functions (Marschner, 1995). Ca2+ is involved in cell elongation and cell division, influences the pH of cells, and also acts as a regulatory ion in the source-sink translocation of carbohydrates through its effect on cells and cell walls.
When Ca2+ is deficient in the media/soil, there will be problems with cell wall stability (see above; Marschner, 1995). Ca2+ in the xylem sap is translocated upward in the transpiration system, but once deposited, it is almost immobile. As a result of this immobility, deficiency symptoms are most pronounced in young tissues—where cell division is occurring. However, true Ca2+ deficiencies in soils are rather rare, and problems associated with low soil Ca2+ may be attributed to soil problems rather than a true Ca2+ deficiency. For example, Ca2+ deficiencies are favored by very low soil pH and on soils high in magnesium and potassium. Probably the most recognizable Ca2+ deficiency, especially to the weekend gardener, is blossom-end rot of tomato fruits, which is induced by water stress (Bennett, 1993). At the time of fruit set, cells at the blossom end of fruits are injured when insufficient Ca2+ translocation to the flower results in a dry-rot area on the expanding fruit (Fig. 1).
Figure 1.
A, Blossom-end rot of tomato is caused in part by calcium deficiencies. Scanning electron micrographs of calcium oxalate crystals from Trifolium pratense (B), Vigna unguiculata (C), and Vicia faba (D). Note the different shapes among the various plants. Scale bar = 1 μm (from Nakata, 2003, with permission).
The distribution of Ca2+ at the cell wall and plasma membrane is mainly the result of a plethora of binding sites for Ca2+ in the cell walls as well as the carefully regulated transport of Ca2+ into the cytoplasm (Han et al., 2003; see below). Pectins and pectates bind a large portion of Ca2+. As Ca2+ supplies increase within certain cells, the proportion of Ca2+ oxalate increases. In certain situations, oxalate-bound Ca2+ may represent the dominant binding form of Ca2+ (Fig. 1; Nakata, 2003). Intracellular Ca2+ is also found in the endoplasmic reticulum (ER) and chloroplast, and most of the water-soluble calcium is in the vacuole. By virtue of their size and capacity for calcium accumulation, the vacuole is the most prominent sink for calcium storage.
Much of our knowledge regarding the role of Ca2+ in cell wall stability and expansion has been obtained from classic physiology experiments. Since it is difficult (impossible?) to generate plant Ca2+ auxotrophs, the onus is now on plant biologists to generate a set of molecular tools to unravel the mechanisms behind these actions (Braam, 1999).
“HUSHHHHH”: KEEPING FREE CYTOSOLIC CALCIUM LEVELS LOW
Numerous cytosolic proteins bind Ca2+ to dampen free cytosolic Ca2+ concentrations (Sanders et al., 1999; Luan et al., 2002; Sanders et al., 2002). Some of the most prominent Ca2+-binding proteins are the molecular chaperone binding proteins, calnexin, calsequestrin, and calreticulin (CRT; Pittman and Hirschi, 2003). Of these, CRT is responsible for the main Ca2+-retaining pool in plants. During signal transduction events (see below), proteins like calmodulin and Ca2+-dependent protein kinases (CDPKs) also bind Ca2+. Calmodulin and CDPKs contains four Ca2+-binding EF-hand motifs. Arabidopsis appears to have around 250 putative proteins that contain at least one EF-hand motif (Day et al., 2002). Future work will need to be directed at the function of these various proteins in Ca2+ homeostasis, signaling, and plant architecture.
Another means of reducing cytosolic calcium levels is by transporting the Ca2+ into endomembranes such as the ER, chloroplast, and vacuole (Sze et al., 2000). Homeostasis of Ca2+ must be achieved by moving Ca2+ out of the cytosol across the plasma membrane and various endomembranes. Efflux across the plasma membrane may be the ultimate fate of excess cytosolic Ca2+ because both biochemical buffering and sequestration have finite capacities. At the plasma membrane, the Ca2+ concentration ratio (inside/outside) is typically of the order of 10−4. Efflux of Ca2+ from the cytosol is mediated by pumps energized by either ATP hydrolysis or the proton motive force. Passive entry of Ca2+ into the cytosol is mediated by ion channels (Sanders et al., 2002).
“STOP AND GO”: CHANGES IN CYTOSOLIC CALCIUM LEVELS MEDIATE PLANT RESPONSES
Ca2+ is a fundamental component of eukaryotic signaling. Ca2+-triggered events are critical for both normal cellular activity and for adapted responses (Sanders et al., 2002). At one level, these Ca2+ signaling events appear simple: cells at rest have a low level of cytosolic Ca2+ that rises during a signal transduction event. However, this signaling is quite complex when one contemplates how a ubiquitous nutrient becomes translated into a myriad of unique stimulus dependent responses.
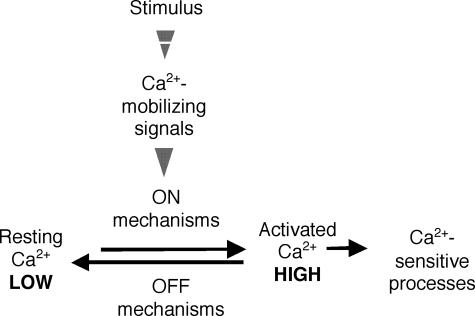
Ca2+ signal transduction can be divided into four components (Fig. 2; Berridge et al., 2000): (1) signaling elicits various Ca2+ mobilizing signals; (2) the mobilizing signals feed Ca2+ into the cytoplasm generating the ON events; (3) Ca2+ functions as a messenger to activate Ca2+ sensitive processes, which are mediated by proteins like calmodulin and CDPKs; and (4) the OFF mechanisms, composed of transporters and binding proteins, remove the Ca2+ from the cytoplasm to restore the resting state.
Figure 2.
The four units of the Ca2+-signaling network (Berridge et al., 2000). Stimuli act by generating Ca2+-mobilizing signals that act on various ON mechanisms to trigger an increase in the intracellular concentration of Ca2+. The response is terminated by OFF mechanisms that restore Ca2+ to resting levels.
These distinctions are somewhat arbitrary given that the mechanisms are tightly coupled and there is often a link between Ca2+ efflux or depletion and Ca2+ entry. A central facet of these four components of signaling is that local spatial and temporal patterns of Ca2+ signals are important in encoding the specificity of cellular responses (Putney, 1998). In other words, Ca2+ signaling, like real-estate prices, depends on location.
These localized alterations in Ca2+ must be judiciously regulated. A marked and prolonged increase in Ca2+ is harmful to cells because it leads to activation of particular Ca2+-dependent enzymes, having a potentially adverse effect. In some mammalian cells, Ca2+ overload can also cause mitochondrial failure; if this becomes irreversible it leads to cell death (Duchen, 2000).
There are several fundamental aspects of plant Ca2+ signaling that should be noted. While animals predominately utilize Na+ as the coupling ion to circulate Ca2+ across biological membranes, plants almost exclusively use protons as the coupling ion (Sze et al., 1999). Also, the design and architecture of the plant cell mediate spatial features to the Ca2+ spike not seen in mammalian systems—in particular, the Ca2+ spikes around the vacuole. Did you know the plant vacuole can occupy up to 99% of a plant cell's volume (Marty, 1999)? Aside from the Ca2+ fluctuations that occur around the ER and plasma membrane, various findings now suggest that localized Ca2+ spikes around the plant vacuole play a pivotal role in determining a plant's growth, development, and adaptation to environmental responses (Sanders et al., 2002). However, Ca2+ spikes in and around the mitochondria and other endomembranes are also likely to be important in particular cellular responses (Logan and Knight, 2003).
The technology to visualize fluctuations in cytosolic Ca2+ levels and combine these approaches with molecular genetics is an exciting new vista in plant biology (Allen et al., 1999; Kiegle et al., 2000). For example, this technology has been used to visualize the perturbations in cytosolic calcium oscillations associated with reduced vacuolar H+-ATPase (V-ATPase) activity (Allen et al., 2000, 2001). These studies have gone on to demonstrate that specific cytosolic Ca2+ oscillations are essential to elicit processes like stomatal closure.
DESIGNING FOR YIELD: ALTERING CALCIUM LEVELS FOR INCREASED PLANT PRODUCTIVITY
For more than 2,000 years, it has been standard agricultural practice to add mineral elements to soils to improve plant growth. As we mentioned previously, increased Ca2+ levels in the soils can improve membrane stability, and Ca2+ is applied to soils to ameliorate salinity effects and decrease pathogen infection. It is also added exogenously to ripe fruits to improve durability. The genetic manipulation of the processes that govern the passage of Ca2+ through the cytoplasm may also have a substantial impact not only on improving growth but also on manipulating particular cellular responses (Pittman and Hirschi, 2003). For example, rice plants have been generated with increased levels of a particular CDPK (Saijo et al., 2000, 2001). The extent of tolerance to cold and salt/drought stresses of these plants correlated well with the level of CDPK proteins. Plants have also been engineered to express higher levels of the Ca2+-binding protein CRT (Persson et al., 2001; Wyatt et al., 2002). These plants are more vigorous than the controls and contain slightly more total Ca2+ than wild-type plants. Therefore, it appears that the manipulation of CDPK, CRT, and other Ca2+-binding proteins may be one way to engineer more robust plant varieties.
DESIGNING FOR FEED: INCREASING BIOAVAILABLE CALCIUM LEVELS IN FOODS
Some portion of Ca2+ in foods is bioavailable, meaning it is digested, absorbed, and metabolized. This bioavailable Ca2+ affects various developmental processes, including bone formation and calcification. An estimated $13.8 billion in health-care costs each year is used on osteoporosis-related care (Bachrach, 1999, 2001). Unfortunately, the majority of people do not consume enough Ca2+. The low Ca2+ content of the most widely consumed vegetables make them a minor contributor to the Ca2+ intake for most Americans (Weaver et al., 1999). Many plant foods are enriched in Ca2+, but the Ca2+ is often found sequestered as an oxalate salt (Fig. 1). Oxalate is an antinutrient that sequesters Ca2+ in a state that renders it unavailable for nutritional absorption (Weaver et al., 1987). In general, Ca2+ absorption is inversely proportional to the oxalic acid content of the food. The ability to genetically alter the Ca2+ content of agriculturally important crops is just emerging.
Recently, scientists have manipulated Ca2+ oxalate crystal formation in Medicago truncatula (a forage legume; Nakata, 2003). Medicago is not consumed by humans; however, the plant contains Ca2+ oxalate crystals in the leaf tissue that are very similar to those found in other plant foods such as spinach (Fig. 1). Several allelic mutants (cod 5) devoid of crystal formation have been isolated. In the greenhouse, plant phenotype and growth studies indicated little difference between the cod 5 and unmutagenized control plants. Oxalate measurements show that cod 5 has oxalate levels at the limit of detection. Ca2+ levels, on the other hand, are comparable to unmutagenized control plants. Overall, the isolation of cod 5 shows the feasibility of manipulating Ca2+ oxalate formation in plants, including many crop plants (e.g. leafy green vegetables), via a mutation at a single loci. These findings suggest that in the future, it may be possible to alter the function of a single gene in spinach to remove all Ca2+ oxalate crystals.
Another approach to alter the content of bioavailable Ca2+ content in plants is to engineer high expression of Ca2+ transporters in the edible portion of the plant. Simplistically, this strategy can be thought of as nutrient mining, where the nutrient is transported from the soil into the edible portions of the plant. Specifically, one potential model for increasing the Ca2+ content in edible foods would be to manipulate plant endomembrane transporters to transport more Ca2+. In plants, we have characterized a vacuolar Ca2+ antiporter termed cation exchanger 1 (CAX1). In both tobacco and carrots, high-level expression of CAX1 displays dramatic increases in calcium content when compared to vector control plants (Hirschi, 1999; Park et al., 2004). In a similar fashion, ectopic expression in tobacco of a wheat cation transporter LCT1 increases shoot Ca2+ levels (Antosiewicz and Hennig, 2004). In the future, using mouse and human feeding studies, biologists will test if these genetically modified plants have altered calcium bioavailability.
CONUNDRUM REDOX
In this general overview, I have attempted to illustrate that while Ca2+ is required for basic plant nutrition, it is also the most common signal transduction element in all eukaryotic cells. To paraphrase the paradox, Ca2+ levels can climb to a huge percentage of the plant dry mass; however, minute fluctuations in cytosolic Ca2+ levels determine how plants respond to developmental and external cues. While Ca2+ is required for life, prolonged high intracellular Ca2+ levels lead to cell death. Ca2+ cannot be metabolized like other second messenger molecules, so cells tightly regulate cytosolic levels through numerous binding proteins and transporters.
Remember, next time you bite into an apple or squeeze a tomato, you are, in part, assessing the Ca2+ status of the fruit. Using the tools of modern molecular genetics and in vivo Ca2+ imaging, plant scientists are trying to assess the cytosolic Ca2+ fluctuations in plants. The outcome of these studies should aid in conceptualizing and harnessing this useful signal/nutrient.
Acknowledgments
I thank the members of my lab and Jon K. Pittman for critical reading of this manuscript.
This work was supported by the U.S. Department of Agriculture/Agricultural Research Service (cooperative agreement no. 58–6250–6001), by the National Science Foundation (grant no. IBN–0209777), and by the National Institute of Health (grant no. 1R01 DK 062366).
References
- Allen GJ, Kwak JM, Chu SP, Llopis J, Tsien RY, Harper JF, Schroeder JI (1999) Cameleon calcium indicator reports cytoplasmic calcium dynamics in Arabidopsis guard cells. Plant J 19: 735–747 [DOI] [PubMed] [Google Scholar]
- Allen GJ, Chu SP, Harrington CL, Schumacher K, Hoffman T, Tang YY, Grill E, Schroeder JI (2001) A defined range of guard cell calcium oscillation parameters encodes stomatal movements. Nature 411: 1053–1057 [DOI] [PubMed] [Google Scholar]
- Allen GJ, Chu SP, Schumacher K, Shimazaki CT, Vafeados D, Kemper A, Hawke SK, Tallman G, Tsien RY, Harper JF, et al (2000) Alteration of stimulus-specific guard cell calcium oscillations and stomatal closing in Arabidopsis det3 mutant. Science 289: 2338–2342 [DOI] [PubMed] [Google Scholar]
- Antosiewicz DM, Hennig J (2004) Overexpression of LCT1 in tobacco enhances the protective action of calcium against cadmium toxicity. Environ Pollut 129: 237–245 [DOI] [PubMed] [Google Scholar]
- Bachrach LK (1999) Malnutrition, endocrinopathies, and deficits in bone mass acquisition. In J-P Bonjour, RC Tsang, eds, Nutrition and Bone Development. Vevey/Lippincott-Raven, Philadelphia, pp 261–277
- Bachrach LK (2001) Acquisition of optimal bone mass in childhood and adolescence. Trends Endocrinol Metab 12: 22–28 [DOI] [PubMed] [Google Scholar]
- Bennett WF (1993) Nutrient Deficiencies and Toxicities in Crop Plants. APS Press, St. Paul
- Berridge MJ, Lipp P, Bootman MD (2000) The versatility and universality of calcium signaling. Nat Rev Mol Cell Biol 1: 11–21 [DOI] [PubMed] [Google Scholar]
- Braam J (1999) If walls could talk. Curr Opin Plant Biol 2: 521–524 [DOI] [PubMed] [Google Scholar]
- Bramlage WJ (1994) Physiological role of calcium in fruit. In AB Petersen, RG Stevens, eds, Tree Fruit Nutrition. Good Fruit Grower, Yakima, WA, pp 101–107
- Bramlage WJ, Weis SA (1994) Postharvest use of calcium. In AB Petersen, RG Stevens, eds, Tree Fruit Nutrition. Good Fruit Grower, Yakima, WA, pp 125–134
- Day IS, Reddy VS, Ali GS, Reddy ASN (2002) Analysis of EF-hand-containing proteins in Arabidopsis. Genome Biol 3: 1–24 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duchen MR (2000) Mitochondria and Ca2+ in cell physiology and pathophysiology. Cell Calcium 28: 339–348 [DOI] [PubMed] [Google Scholar]
- Epstein E (1972) Mineral Nutrition of Plants: Principles and Perspectives. Wiley, New York
- Han S, Tang R, Anderson LK, Woerner TE, Pei Z-M (2003) A cell surface receptor mediates extracellular Ca2+ sensing in guard cells. Nature 425: 196–200 [DOI] [PubMed] [Google Scholar]
- Hirschi KD (1999) Expression of Arabidopsis CAX1 in tobacco: altered calcium homeostasis and increased stress sensitivity. Plant Cell 11: 2113–2122 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kiegle E, Moore CA, Haseloff J, Tester MA, Knight MR (2000) Cell-type-specific calcium responses to drought, salt and cold in the Arabidopsis root. Plant J 23: 267–278 [DOI] [PubMed] [Google Scholar]
- Kirby EA, Pilbeam DJ (1984) Calcium as a plant nutrient. Plant Cell Environ 7: 397–405 [Google Scholar]
- Logan DC, Knight MR (2003) Mitochondrial and cytosolic calcium dynamics are differentially regulated in plants. Plant Physiol 133: 21–24 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luan S, Kudla J, Rodriguez-Concepcion M, Yalovsky S, Gruissem W (2002) Calmodulins and calcineurin B-like proteins: calcium sensors for specific signal response coupling in plants. Plant Cell 14 (Suppl): S389–S400 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marschner H (1995) Mineral Nutrition of Plants, Ed 2. Academic Press, Boston
- Marty F (1999) Plant vacuoles. Plant Cell 11: 587–599 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakata PA (2003) Advances in our understanding of calcium oxylate crystals formed in plants. Plant Sci 164: 901–909 [Google Scholar]
- Park SH, Kim C-K, Pike LM, Smith RH, Hirschi KD (2004) Increased calcium in carrots by expression of an Arabidopsis H+/Ca2+ transporter. Mol Breed (in press)
- Persson S, Wyatt SE, Love J, Thompson WF, Robertson D, Boss WF (2001) The Ca2+ status of the endoplasmic reticulum is altered by induction of calreticulin expression in transgenic plants. Plant Physiol 126: 1092–1104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pittman JK, Hirschi K (2003) Don't shoot the (second) messenger: endomembrane transporters and binding proteins modulate cytosolic Ca2+ levels. Curr Opin Plant Biol 6: 257–262 [DOI] [PubMed] [Google Scholar]
- Putney JW (1998) Calcium signaling: up, down, up, down…what's the point? Science 279: 191–192 [DOI] [PubMed] [Google Scholar]
- Saijo Y, Hata S, Kyozuka J, Shimamoto K, Izui K (2000) Over-expression of a single Ca2+-dependent protein kinase confers both cold and salt/drought tolerance on rice plants. Plant J 23: 319–327 [DOI] [PubMed] [Google Scholar]
- Saijo Y, Kinoshita N, Ishiyama K, Hata S, Kyozuka J, Hayakawa T, Nakamura T, Shimamoto K, Yamaya T, Izui K (2001) A Ca2+-dependent protein kinase that endows rice plants with cold- and salt-stress tolerance functions in vascular bundles. Plant Cell Physiol 42: 1228–1233 [DOI] [PubMed] [Google Scholar]
- Sanders D, Brownlee C, Harper JF (1999) Communicating with calcium. Plant Cell 11: 691–706 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanders D, Pelloux J, Brownlee C, Harper JF (2002) Calcium at the crossroads of signaling. Plant Cell 14 (Suppl): S401–S417 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sze H, Li X, Palmgren MG (1999) Energization of plant cell membranes by H+-pumping ATPases: regulation and biosynthesis. Plant Cell 11: 677–689 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sze H, Liang F, Hwang I, Harper J (2000) Diversity and regulation of Ca2+ pumps: insights from expression in yeast. Annu Rev Plant Physiol Plant Mol Biol 51: 433–462 [DOI] [PubMed] [Google Scholar]
- Weaver CM, Martin BR, Ebner JS, Krueger CA (1987) Oxalic acid decreases calcium absorption in rats. J Nutr 117: 1903–1906 [DOI] [PubMed] [Google Scholar]
- Weaver CM, Proulx WR, Heaney R (1999) Choices for achieving adequate dietary calcium with a vegetarian diet. Am J Clin Nutr 70: 543S–548S [DOI] [PubMed] [Google Scholar]
- Wyatt SE, Tsou P-L, Robertson D (2002) Expression of the high capacity calcium binding domain of calreticulin increase bioavailable calcium stores in plants. Transgenic Res 11: 1–10 [DOI] [PubMed] [Google Scholar]