Abstract
MicroRNAs (miRNAs) are ~22-nt RNAs that direct posttranscriptional repression of mRNA targets in diverse eukaryotic lineages. In humans and other mammals, these small RNAs help sculpt the expression of most mRNAs. This article reviews advances in our understanding of the defining features of metazoan miRNAs and their biogenesis, genomics, and evolution. It then reviews how metazoan miRNAs are regulated, how they recognize and cause repression of their targets, and the biological functions of this repression, with a compilation of knockout phenotypes that shows that important biological functions have been identified for most of the broadly conserved miRNAs of mammals.
INTRODUCTION
MicroRNAs (miRNAs) are small regulatory RNAs that are processed from stem-loop regions of longer RNA transcripts. Hundreds of different miRNAs have been identified in humans, many of which are conserved in other animals, and these conserved miRNAs have preferentially conserved interactions with most human mRNAs (Friedman et al., 2009). This inferred regulation of most human mRNAs suggests that miRNAs influence essentially all developmental process and diseases. Indeed, loss-of-function studies disrupting miRNA genes in mice have revealed diverse phenotypes, including defects in the development of the skeleton, teeth, brain, eyes, neurons, muscle, heart, lungs, kidneys, vasculature, liver, pancreas, intestine, skin, fat, breast, ovaries, testes, placenta, thymus, and each hematopoietic lineage, as well as cellular, physiological, and behavioral defects. Many of these developmental and physiological defects affect embryonic or postnatal viability, or cause other severe conditions, such as epilepsy, deafness, retinal degeneration, infertility, immune disorders, or cancer. In addition, some miRNA-knockout strains have altered susceptibility to infections, and many have differential responses to mouse models of diseases or injuries.
As with many discoveries of fundamental importance to mammalian development, physiology, and disease, the first known miRNA was not found in humans or other mammals but was instead found in an invertebrate model organism. Molecular geneticists studying the lin-4 and let-7 genes, which are each required for the proper timing of C. elegans development, found that instead of producing mRNAs, these genes produce noncoding RNAs, including short RNAs ~22 nt in length (Lee et al., 1993; Reinhart et al., 2000). The lin-4 and let-7 RNAs both had imperfect complementarity to conserved sites within the 3′ UTRs of genetically identified regulatory targets, which led to a model in which these small RNAs mediate translational repression through antisense interactions (Lee et al., 1993; Wightman et al., 1993; Moss et al., 1997; Olsen and Ambros, 1999; Reinhart et al., 2000). The let-7 RNA was subsequently recognized in humans and other bilaterian animals, with temporal expression resembling that observed in C. elegans (Pasquinelli et al., 2000). This discovery showed that these regulatory RNAs were not mere curiosities of worms and led to the idea that additional “small temporal RNAs” might exist to regulate the timing of other developmental transitions (Pasquinelli et al., 2000). Soon thereafter, molecular searches for endogenous small RNAs refined the identities of the lin-4 and let-7 RNAs and revealed that these RNAs were actually part of a much larger class of small RNAs (Lagos-Quintana et al., 2001; Lau et al., 2001; Lee and Ambros, 2001). Members of this class all resembled lin-4 and let-7 RNAs in their small size and potential to be processed from hairpin precursors, but most were not expressed in a temporal manner. Because they were identified without the help of genetics, their functions were not known—what was known is that they were small, and so they were called “microRNAs” (Lagos-Quintana et al., 2001; Lau et al., 2001; Lee and Ambros, 2001).
At this point, interest in these small regulatory RNAs surged. It was clear that there were hundreds of different miRNAs in humans, flies, nematodes, and presumably other animals, which implied that in each of these species there were hundreds of different mRNAs that were regulated. Moreover, these potential regulatory targets were not just mRNAs involved in the timing of development—any mRNA that a biologist was studying might be regulated by one or more miRNAs. In this review I touch on what has since been learned about the miRNAs of animals, particularly with respect to their biogenesis, genomics, regulation, mechanisms of action, target recognition, and biological functions.
miRNA BIOGENESIS, GENOMICS, AND EVOLUTION
The metazoan miRNA pathway derived from a more basal RNA-silencing pathway known as RNA interference (RNAi), which appears to have been present in the last common ancestor of eukaryotes and continues to defend against viruses and transposons in many extant eukaryotes (Shabalina and Koonin, 2008). The hallmark innovation of the miRNA pathway is the use of short hairpins to produce defined guide RNAs appropriate for directing the silencing machinery to specific cellular mRNAs, whereas the ancestral RNAi pathway starts with longer dsRNA precursors that each produce a large diversity of siRNAs (Bartel, 2004). When considering the intrinsic advantages of short hairpins for generating defined guide RNAs appropriate for enlisting RNA silencing for endogenous gene regulation, miRNAs might have been expected to have arisen more than once in eukaryotic evolution. Indeed, miRNAs or miRNA-like RNAs have emerged independently in other diverse eukaryotic lineages, including land plants (Jones-Rhoades et al., 2006), green algae (Molnar et al., 2007; Zhao et al., 2007), brown algae (Cock et al., 2010), filamentous fungi (Lee et al., 2010), and slime mold (Avesson et al., 2012).
Biogenesis of Canonical miRNAs
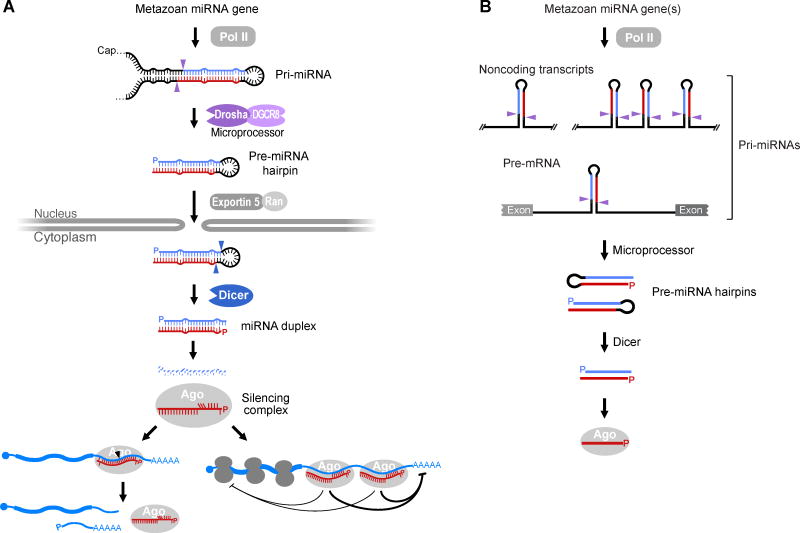
In animals, canonical miRNAs are transcribed by RNA Polymerase II (Pol II) as part of much longer RNAs called “pri-miRNAs” (Lee et al., 2002; Cai et al., 2004; Lee et al., 2004) (Figure 1). Each pri-miRNA has at least one region that folds back on itself to form a hairpin substrate for Microprocessor, a heterotrimeric complex containing one molecule of the Drosha endonuclease and two molecules of its partner protein, DGCR8 (named Pasha in flies and nematodes) (Nguyen et al., 2015). Drosha has two RNase III domains that each cut one strand of the stem of the pri-miRNA hairpin with a 2-bp offset, which liberates a ~60-nt stem-loop called a “pre-miRNA” (Lee et al., 2003) (Figure 1A). Note that although all canonical pri-miRNAs have a 5′ cap, as expected for Pol II transcripts, they do not necessarily have a poly(A) tail because cotranscriptional processing by Microprocessor can sometimes trigger transcription termination in a manner that preempts normal 3′-end maturation (Ballarino et al., 2009).
Figure 1. The Biogenesis and Function of Canonical miRNAs of Animals.
(A) Biogenesis and function of a typical miRNA. Once transcribed by Pol II, the pri-miRNA folds back on itself to form at least one distinctive hairpin structure (further described in Figure 2), which is cleaved by Microprocessor (purple arrowheads, cleavage sites) to release a pre-miRNA (P, 5′ phosphate). The pre-miRNA hairpin is exported to the cytoplasm through the action of Exportin 5 and RAN–GTP. In the cytoplasm, the pre-miRNA is cleaved by Dicer (blue arrowheads, cleavage sites) to produce a ~20-bp miRNA duplex with a 5′ phosphate (P) and a 2-nt 3′ overhang on each end. One strand of the miRNA duplex, the mature miRNA (red), is loaded into the guide-strand channel of an Argonaute protein (Ago) to form a silencing complex, whereas the other strand, the miRNA* (blue), is degraded. Within the free silencing complex, miRNA nucleotides 2–5 (upward red vertical lines) are poised to initially interact with target RNAs (blue; filled circle, cap; AAAAA, poly(A) tail). This pairing usually extends to nucleotide 7 or 8 of the miRNA, and occasionally is more extensive (Figures 5 and 6). If pairing is very extensive, the target can be sliced (left; black arrowhead, cleavage site), whereas if it is not, the target can undergo other types of repression (right; further described in Figure 4).
(B) Typical sources of canonical miRNAs. Most canonical miRNAs derive from the introns or exons of non-coding primary transcripts, some of which harbor hairpins for more than one miRNA. In addition, many canonical miRNAs derive from introns of pre-mRNAs.
After export to the cytoplasm through the action of Exportin 5 and RAN–GTP (Yi et al., 2003; Bohnsack et al., 2004; Lund et al., 2004), the pre-miRNA is further processed by Dicer (Grishok et al., 2001; Hutvagner et al., 2001). Like Drosha, Dicer is an endonuclease with two RNase III domains (Bernstein et al., 2001; Zhang et al., 2004), and like Drosha, it associates with a partner protein, although the Dicer partner protein (named TRBP in mammals and Loquacious in flies) is essential for pre-miRNA processing in flies but not mammals (Ha and Kim, 2014). Dicer cuts both strands near the loop to generate the miRNA duplex, which contains the miRNA paired to its passenger strand (often called the “miRNA*,” pronounced “miRNA star”). This duplex has a ~2-nt 3′ overhang on each end, resulting from the offset cuts made by both Drosha and Dicer (Lee et al., 2003; Zhang et al., 2004) (Figure 1A). Once formed, the miRNA duplex is loaded into an Argonaute protein with assistance from chaperone proteins (HSC70/HSP90), which use ATP to help Argonaute assume a high-energy, open conformation suitable for binding the rigid miRNA duplex (Iwasaki et al., 2010). Following loading of the duplex, relaxation of Argonaute back to its ground-state conformation is thought to promote expulsion of the miRNA* to form the mature silencing complex (Kawamata and Tomari, 2010).
The choice of which strand of the duplex usually becomes the miRNA, i.e., the guide strand of the silencing complex, and which one usually is discarded and degraded as the miRNA* depends on the preferred orientation by which the duplex binds Argonaute, and this orientation of the duplex depends on which strand of the duplex has a 5′ terminus most suitable for loading into the pocket within Argonaute that binds the 5′-nucleoside monophosphate of guide RNAs. This pocket prefers a 5′-terminal pU or pA (Frank et al., 2010; Suzuki et al., 2015) as well as the 5′-nucleoside monophosphate of the strand with the least stable 5′ terminal pairing (Khvorova et al., 2003; Schwarz et al., 2003). Because the preferred loading orientation is independent of the duplex orientation within the pre-miRNA, the strand from either arm of the hairpin can be retained to become the miRNA (Figure 1B). Once loaded into the silencing complex, the miRNA pairs to sites within mRNAs and other transcripts to direct their posttranscriptional repression (Figure 1A).
Genomics and Evolution of Canonical miRNAs
High-throughput sequencing of small RNAs has transformed miRNA gene discovery (Lu et al., 2005; Ruby et al., 2006). Of course, not every small RNA sequenced from the cell is a miRNA. Some are other types of small RNAs, such as piwi-interacting RNAs (piRNAs) and endogenous small interfering RNAs (siRNAs), which derive from related RNA-silencing pathways (Malone and Hannon, 2009), and others are degradation fragments of longer RNAs. To prevent these other types of small RNAs from being misannotated as miRNAs, stringent analysis pipelines require that the annotated miRNA have a consistent 5′ terminus and map to a potential hairpin supported by reads corresponding to both strands of the miRNA duplex, with its diagnostic ~2-nt 3′ overhangs (Ruby et al., 2006). Using these criteria, recent analyses have supported the authenticity of many previously annotated genes and identified some new genes, bringing tallies of confidently identified canonical miRNA genes to 147 in C. elegans (Jan et al., 2011), 164 in Drosophila (Fromm et al., 2015), 475 in mouse (Chiang et al., 2010), and 519 in human (Fromm et al., 2015).
Although lin-4, let-7, and a couple of other metazoan miRNAs were named based on their mutant phenotypes, the other miRNAs, which were identified before a mutant phenotype was known, were named with numbers, in the order of their discovery (Ambros et al., 2003). Some, albeit imperfect, attempt was also made to give orthologs from different species the same name. For example, 11 of the 12 human orthologs of C. elegans let-7 bear the let-7 name (the other one being mir-98). Likewise, an attempt was made to assign similar names to paralogs within a species, with letter suffixes (a, b, c, …) distinguishing genes producing similar mature miRNAs and number suffixes (-1, -2, -3, …) distinguishing those producing identical mature miRNAs.
Of the more than 100 studies that have sought to annotate miRNAs, not all have imposed stringent criteria for gene annotation, leading to many additional gene annotations. For example, miRBase v21 lists 1,193 miRNA gene annotations in mouse and 1,881 in human (Kozomara and Griffiths-Jones, 2014), raising the question of how many of these additional annotations represent authentic miRNAs missed by the stringent annotation criteria (which might happen, for instance, if a miRNA was not expressed at sufficient levels in the sequenced samples), and how many are false positive annotations. Experimental evaluation of a subset of these additional annotations indicates that a large majority are false-positives (Chiang et al., 2010), implying that biologists interested in exploring miRNA functions should focus on only miRNA annotations that satisfy the stringent criteria, based on either existing or newly acquired high-throughput sequencing data.
MicroRNAs are grouped into families based on their targeting properties, which depend primarily on the identity of their extended seed region (miRNA nucleotides 2–8) (Bartel, 2009). For example, mice and humans each have three members of the miR-1/206 seed family (miR-1-1, miR-1-2, and miR-206), which are paralogous miRNAs that arose through duplication of an ancestral gene inherited from a common ancestor of all bilaterian animals. Indeed, members of the same seed family are usually evolutionarily related, and evolutionarily related miRNAs are usually members of the same seed family. However, the use of the term “family” does not strictly denote common ancestry. For example, miR-32, which is not related to other members of the miR-25/32/92/363/367 seed family, is nonetheless an adopted member because it has converged on the same extended seed and thus has the same targeting preferences. Other miRNAs, such as miR-200a and miR-200b, which are clearly related, have a single-nucleotide difference in their seed regions that place them into different families because of their divergent targeting preferences. As with paralogous proteins, members of the same seed families often have at least partially redundant functions, with severe loss-of-function phenotypes apparent only after multiple family members are disrupted (Tables 1 and 2). However, phenotypes are often observed after disruption of a single member, particularly in contexts in which that member is preferentially expressed (Table 1 and 2, e.g., miR-206, miR-7a-2, miR-9-3, etc.).
Table 1. Abnormal phenotypes observed in mice after knocking out one or more members of a very broadly conserved miRNA family.
This table lists results for 20 of the 27 murine miRNA families conserved since the bilaterian ancestor (Table S1). With regard to the other seven families, for four (miR-183, miR-193, 216a, and miR-365) a member has been knocked out but only in the context of a deletion that also removes one or two miRNAs from other families (Table 3), which complicates attribution of the phenotypes, and for the remaining three a mouse knockout has yet to be reported. Some strains have more than one miRNA gene disrupted; multiple miRNAs from a disrupted polycistronic locus are linked with a tilde (~), whereas those from distant loci are separated with a comma.
| miRNA family (# of genes) | miRNA(s) removed | Phenotype |
|---|---|---|
| miR-1/206 (3) | miR-1-1, miR-1-2 | Postnatal lethality before weaning, complete penetrance, due to heart defects (Heidersbach et al., 2013; Wei et al., 2014) |
| miR-206 | Abnormal airway smooth muscle innervation (Radzikinas et al., 2011); reduced regeneration of muscle and neuromuscular junctions after injury; accelerated pathology in neuromuscular disease models (ALS, Duchenne muscular dystrophy) (Williams et al., 2009; Liu et al., 2012) | |
| miR-7 (3) | miR-7a-2 | Male and female infertility with hypogonadism due to pituitary defect (Ahmed et al., 2017); abnormal function of pancreatic beta cells (Latreille et al., 2014) |
| let-7/miR-98 (12) | let7c-2 ~ let7b | Modest intestinal hypertrophy, with increased crypt fission (Madison et al., 2013); increase in megakaryocyte–erythroid progenitors and decrease in granulocyte–monocyte progenitors (Rowe et al., 2016); faster liver regeneration after resection (Wu et al., 2015) |
| miR-9 (3) | miR-9-2, miR-9-3 | Postnatal lethality, complete penetrance; abnormal brain development (Shibata et al., 2011) |
| miR-9-3 | Male and female infertility (Shibata et al., 2011) | |
| miR-10 (2) | miR-10a | Increased intestinal adenomas in sensitized background (Apc) (Stadthagen et al., 2013) |
| miR-10b | Enlarged spleen with expansion of germinal centers; reduced tumorigenesis of breast cancer in sensitized background (MMTV-PyMT) (Kim et al., 2016b) | |
| miR-22 (1) | miR-22 | Reduced response of Th17 cells (Lu et al., 2015b); stress-induced cardiac dilation and contractile dysfunction (Gurha et al., 2012; Huang et al., 2013); reduced pathology in two disease models |
| miR-29 (4) | miR-29b-1 ~ 29a, | Embryonic lethality, partial penetrance, with death of survivors at ~4 weeks |
| miR-29b-2 ~ 29c | (Dooley et al., 2016; Sassi et al., 2017) | |
| miR-29b-1 ~ 29a | Reduced lifespan; decreased body weight; progressive locomotor impairment and ataxia with Purkinje cell defects; reduced numbers of hematopoietic stem and progenitor cells; premature thymic involution; perturbed development of Th1 cells and other lymphoid cells; reduced susceptibility to induced autoimmune disease (Papadopoulou et al., 2011; Smith et al., 2012; Hu et al., 2015; Papadopoulou et al., 2015); reduced pathology in models of obesity and arthritis (Dooley et al., 2016; van Nieuwenhuijze et al., 2017) | |
| miR-29b-2 ~ 29c | Reduced pathology in models of obesity, arthritis, and heart disease (Dooley et al., 2016; Sassi et al., 2017; van Nieuwenhuijze et al., 2017) | |
| miR-31 (1) | miR-31 | Perturbed T cell response and function; improved recovery from lymphocytic choriomeningitis virus infection; reduced pathology in model of autoimmune encephalomyelitis (Zhang et al., 2015; Moffett et al., 2017); increased tumorigenesis in model of colorectal cancer (Liu et al., 2017) |
| miR-33 (1) | miR-33 | Increased cholesterol efflux and serum HDL (Horie et al., 2010); increased ApoE lipidation and Aβ degradation in the brain (Kim et al., 2015); increased obesity and liver steatosis on high-fat diet (Horie et al., 2013); reduced fibrosis in model of heart disease (Nishiga et al., 2017) |
| miR-34/449 (6) | miR-34a, miR-34b ~ 34c, miR-449c ~ 449b ~ 449a | Postnatal lethality, incomplete penetrance; growth retardation; male and female infertility, and respiratory dysfunction due to defective motile cilia (Song et al., 2014) |
| miR-34b ~ 34c, miR-449c ~ 449b ~ 449a | Postnatal lethality, incomplete penetrance; growth retardation; abnormal brain development; male and female infertility and pulmonary pathology due to defective motile cilia (Wu et al., 2014); mitotic spindle orientation defects, neurogenesis defects with increased radial glial cells and decreased cortical neurons; thinner brain cortex (Fededa et al., 2016) | |
| miR-34a, miR-34b ~ 34c | Resilience to stress-induced anxiety (Andolina et al., 2016); increased tumorigenesis in several sensitized backgrounds | |
| miR-34a | Increased bone resorption (Krzeszinski et al., 2014); increased tumorigenesis in several sensitized backgrounds | |
| miR-25/32/92/ 363/367 (7) | miR-92a-1 | Embryonic lethality, partial penetrance; smaller size; minor skeletal defects (Penzkofer et al., 2014; Han et al., 2015); reduced pathology in model of kidney disease (Henique et al., 2017) |
| miR-96 (1) | miR-96 | Progressive hearing loss caused by sensory hair-cell degeneration due to arrested development of hair cells and auditory nerve connections (Lewis et al., 2009; Mencia et al., 2009; Kuhn et al., 2011) |
| miR-124 (3) | miR-124-1 | Postnatal lethality, incomplete penetrance; reduced brain size and abnormal brain development with increased neuronal apoptosis, aberrant axon sprouting in hippocampal dentate granule neurons, and reduced retinal cone photoreceptor cells (Sanuki et al., 2011) |
| miR-125 (3) | miR-125a | Defective differentiation of Treg cells; disrupted immune homeostasis with increased inflammation in immune disease models (Pan et al., 2015); perturbed development of kidneys and seminal vesicles; myeloproliferative disorder (Tatsumi et al., 2016) |
| miR-133 (3) | miR-133a-1, miR-133a-2 | Perinatal lethality, incomplete penetrance, due to heart defects; adult heart failure (Liu et al., 2008); defective skeletal muscle development and function (Liu et al., 2011); increased browning of white adipose tissue (Liu et al., 2013) |
| miR-184 (1) | miR-184 | Increased pancreatic beta cell proliferation and mass; decreased fasting blood glucose levels and increased fasting plasma insulin levels (Tattikota et al., 2014); thicker epidermis due to increased epidermal cell proliferation 29198823 |
| miR-200bc/429 (3) | miR-200b, miR-429 | Female infertility due to pituitary defect (Hasuwa et al., 2013) |
| miR-429 | Liver inflammation and dysfunction (Chen et al., 2018) | |
| miR-210 (1) | miR-210 | Increased spontaneous autoantibodies (Mok et al., 2013); increased differentiation of Th17 cells with increased pathology in immune disease model (colitis) (Wang et al., 2014); decreased pathology in pulmonary hypertension model (White et al., 2015) |
| miR-219 (2) | miR-219-1, miR-219-2 | Mostly penetrant neonatal lethality; knockout in oligodendrocytes causes myelination defects with tremors beginning at week 3, progressing to severe seizures, ataxia and death by ~4 months (Wang et al., 2017) |
| miR-219-2 | Tremors at adulthood (Wang et al., 2017) | |
| miR-375 (1) | miR-375 | Increased pancreatic alpha cells and decreased beta cells; hyperglycemia (Poy et al., 2009); altered membrane potential of beta cells (Salunkhe et al., 2015) |
Table 2. Abnormal phenotypes observed in mice after knocking out one or more members of a broadly conserved miRNA family.
This table lists results for 36 of the 62 families that are conserved among mammals and fish but emerged since the bilaterian ancestor (Table S1). For two of the remaining 26 families, a knockout has been generated and reported to have no observed phenotype; these are the miR-30 family, for which two of the six members (miR-30d ~ miR-30b) have been simultaneously deleted (Park et al., 2012), and the miR-144 family, for which the one-and-only member was deleted (Rasmussen et al., 2010). Some strains have more than one miRNA gene disrupted; multiple miRNAs from a disrupted polycistronic locus are linked with a tilde (~), whereas those from distant loci are separated with a comma.
| miRNA family (# of genes) | miRNA(s) removed | Phenotype |
|---|---|---|
| miR-15/16/195/322/497 (7) | miR-15a ~ 16-1 | Increased proliferation of B cells; development of lymphoproliferative disorders (Klein et al., 2010); arrested maturation of natural killer cells (Sullivan et al., 2015); increased phagocytosis of macrophages; reduced mortality in bacterial sepsis models (Moon et al., 2014) |
| miR-15b ~ 16-2 | Development of B cell lymphoproliferative disorders (Lovat et al., 2015) | |
| miR-17/20/93/ 106 (6) | miR-17, miR-20a | Perinatal lethality, incomplete penetrance; vertebral homeotic transformations and other skeletal defects; reduced body weight; reduced pre-B cells (Han et al., 2015) |
| miR-18 (2) | miR-18a | Reduced body weight (Han et al., 2015) |
| miR-19 (3) | miR-19a, miR-19b-1 | Perinatal lethality, incomplete penetrance; reduced body weight; reduced tumorigenesis in sensitized background (Myc) (Han et al., 2015) |
| miR-21 (1) | miR-21 | Reduced growth and increased apoptosis of eosinophil progenitors (Lu et al., 2013b); altered macrophage polarization with more M2 macrophages and fewer M1 macrophages (Wang et al., 2015); increased bone mass with decreased bone resorption (Hu et al., 2017); altered pathology in > 20 disease/injury models |
| miR-23 (2) | miR-23a | Deletion in T cells causes reduced survival of activated T cells and increased susceptibility to bacterial infection (Listeria) (Zhang et al., 2016a) |
| miR-122 (1) | miR-122 | Development of fatty liver, hepatitis, fibrosis, and hepatocellular carcinoma (Hsu et al., 2012; Tsai et al., 2012); smaller hepatocytes with substantially fewer polyploid cells (Hsu et al., 2016); increased susceptibility to acetaminophen toxicity (Chowdhary et al., 2017) |
| miR-126 (1) | miR-126 | Embryonic lethality, incomplete penetrance, with hemorrhaging due to loss of vascular integrity and defects in endothelial cell proliferation, migration, and angiogenesis (Kuhnert et al., 2008; Wang et al., 2008); defects in innate immune response (Agudo et al., 2014); exacerbated atherosclerosis in sensitized background (Apoe) (Schober et al., 2014) |
| miR-128 (2) | miR-128-2 | Development of fatal epilepsy, complete penetrance, preceded by hyperactivity and increased exploration (Tan et al., 2013) |
| miR-130/301 (4) | miR-301a | Reduced pathology in models of colitis and colitis-associated cancer; reduced tumorigenesis of lung, thymus, and skin cancers in sensitized background (Kras) (Ma et al., 2015) |
| miR-132/212 (2) | miR-212 ~ 132 | Impaired memory formation and retention (Hernandez-Rapp et al., 2015; Hansen et al., 2016); reduced depth perception with impaired maturation of binocular cortical cells (Mazziotti et al., 2017); perturbed circadian regulation with reduced dendritic spines in neurons of suprachiasmatic nucleus (Kiessling et al., 2017; Mendoza-Viveros et al., 2017); perturbed cortical synaptic transmission and plasticity (Remenyi et al., 2013); increased endothelial vasodilatory function; increased angiogenic responses (Kumarswamy et al., 2014); altered survival, proliferation, and differentiation of hematopoietic stem cells (Mehta et al., 2015); altered pathology in six disease/injury models |
| miR-137 (1) | miR-137 | Early embryonic lethality, complete penetrance (Crowley et al., 2015) |
| miR-139 (1) | miR-139 | Increased pathology in models of colitis and colitis-associated cancer (Maoa et al., 2015; Zou et al., 2016) |
| miR-140 (1) | miR-140 | Dwarfism and craniofacial deformities, with premature differentiation of chondrocytes; osteoarthritis (Miyaki et al., 2010; Nakamura et al., 2011); increased Leydig cells in developing testes (Rakoczy et al., 2013) |
| miR-142 (1) | miR-142 | Reduced CD4+ dendritic cells (Mildner et al., 2013); decreased platelets due to impaired megakaryocyte maturation (Chapnik et al., 2014); reduced function of mast cells (Yamada et al., 2014); impaired T and B1B cell development and function with immunodeficiency (Kramer et al., 2015; Sun et al., 2015; Mildner et al., 2017); impaired erythropoiesis leading to anemia (Rivkin et al., 2017) |
| miR-143 (1) | miR-143 | Arterial smooth muscle defects with perturbed actin stress fibers; less scarring in response to vascular injury (Xin et al., 2009) |
| miR-145 (1) | miR-145 | Reduced vascular tone with reduced blood pressure; reduced heart mass; arterial smooth muscle defects with perturbed actin stress fibers; increased lethality and less scarring in response to vascular injury (Xin et al., 2009); reduced pathology in pulmonary disease model (fibrosis) (Yang et al., 2013) |
| miR-146 (2) | miR-146a | Hyperresponsive macrophages, exaggerated inflammatory response, and myeloid cell proliferation; eventual autoimmune disorders, exhaustion of hematopoietic stem cells, and hematopoietic neoplasms (Lu et al., 2010; Boldin et al., 2011; Zhao et al., 2013); altered pathology in five disease models |
| miR-146b | Enlarged spleens and increased myeloid cells, spontaneous intestinal inflammation, enhanced M1 macrophage polarization (Peng et al., 2016) | |
| miR-148/152 (3) | miR-148a | Elevated serum and hepatic cholesterol; accelerated carcinogenesis in chemically induced liver cancer (Cheng et al., 2017) |
| miR-150 (1) | miR-150 | Increased B1 cells and enhanced humoral immune response (Xiao et al., 2007); impaired maturation of natural killer T cells (Bezman et al., 2011; Zheng et al., 2012); impaired CD8+ T cell differentiation and function (Smith et al., 2015); reduced bone mass (Choi et al., 2015); altered pathology in eight disease models |
| miR-155 (1) | miR-155 | Impaired development and function of B cells, T cells, and dendritic cells (Rodriguez et al., 2007; Thai et al., 2007; Kohlhaas et al., 2009; Lu et al., 2009; O’Connell et al., 2010; Dunand-Sauthier et al., 2011); perturbed function of macrophages and mast cells (O’Connell et al., 2009; Qayum et al., 2016); altered pathology in > 60 disease/injury models |
| miR-181 (6) | miR-181a-1 ~181b-1, miR-181a-2 ~ 181b-2, miR-181c ~ 181d | Embryonic lethality, complete penetrance (Fragoso et al., 2012; Henao-Mejia et al., 2013) |
| miR-181a-1 ~ 181b-1, miR-181a-2 ~ 181b-2 | Postnatal lethality, incomplete penetrance; reduced body weight; defects in B and T cell development (Henao-Mejia et al., 2013) | |
| miR-181a-1 ~ 181b-1 | Defects in B and T cell development, loss of natural killer T cells (Fragoso et al., 2012; Henao-Mejia et al., 2013; Zietara et al., 2013); reduced leukemia in sensitized background (Notch1) (Fragoso et al., 2012) | |
| miR-182 (1) | miR-182 | Defective antibody response (Li et al., 2016); muscle loss; fast-to-slow fiber-type switching; impaired glucose metabolism (Zhang et al., 2016b); fewer circulating tumor cells and reduced metastasis in model of lung metastasis (Sachdeva et al., 2014) |
| miR-191 (1) | miR-191 | Deletion in T cells causes reduced survival of T cells (Lykken and Li, 2016) |
| miR-192/215 (2) | miR-192 | Reduced kidney pathology in diabetes model (Deshpande et al., 2013) |
| miR-196 (3) | miR-196a, miR-196a-2,miR-196b | Vertebral homeotic transformations, increased vertebral number (Wong et al., 2015) |
| miR-199 (3) | miR-199a-2 | Premature death, starting at 6 weeks; smaller body size; perturbed gait, body trembling, irregular breathing, and hypoactivity; smaller brain size with perturbed hippocampal and cortical neuron shape and density (Tsujimura et al., 2015) |
| miR-203 (1) | miR-203 | Thickened epidermis, enhanced tumorigenesis in chemically induced skin cancer (Riemondy et al., 2015) |
| miR-204/211 (2) | miR-204 | Improved glucose tolerance; reduced pathology in model of diabetes (Jo et al., 2018) |
| miR-211 | Retinal cone degeneration and impaired vision (Barbato et al., 2017) | |
| miR-205 (1) | miR-205 | Postnatal lethality, incomplete penetrance; fragile skin and impaired hair growth due to reduced neonatal expansion of progenitors and stem cells of hair follicles and skin (Farmer et al., 2013; Wang et al., 2013); no tear glands, 50% penetrance, or defective tear-gland development (Farmer et al., 2017); deletion in thymic epithelial cells causes of T cell defects (Hoover et al., 2016) |
| miR-208 (2) | miR-208a | Abnormal atrial cardiac conduction (Callis et al., 2009); reduced cardiac pathology in response to stress and in model of hypothyroidism (van Rooij et al., 2007) |
| miR-214 (1) | miR-214 | Increased pathology in heart disease model (Aurora et al., 2012) |
| miR-218 (2) | miR-218-1, miR-218-2 | Neonatal lethality, complete penetrance, due to lack of respiration; failure of many motor neurons to establish neuromuscular junctions; motor neuron hyperexcitability and degeneration (Amin et al., 2015) |
| miR-223 (1) | miR-223 | Perturbed development of myeloid lineages, with increased proliferation of progenitors and hyperactive neutrophils and macrophages; increased spontaneous and induced lung pathology (after endotoxin challenge); altered susceptibility to infection (Johnnidis et al., 2008; Zhuang et al., 2012; Lu et al., 2013a); increased serum cholesterol and triglycerides (Vickers et al., 2014); altered pathology in > 12 disease models. |
| miR-338 (1) | miR-338 | Heterozygous deletion causes disrupted synaptic transmission at auditory thalamocortical projections (Chun et al., 2017) |
| miR-451 (1) | miR-451 | Mild anemia and splenomegaly due to impaired erythroblast maturation (Patrick et al., 2010; Rasmussen et al., 2010) |
In some cases, the same pri-miRNA harbors multiple miRNA hairpins (Figure 1B). To simplify annotation, each miRNA hairpin is annotated as deriving from an individual miRNA gene, with clustered miRNAs processed from the same transcript thought of as polycistronic (Lagos-Quintana et al., 2001; Lau et al., 2001; Lee et al., 2002). Although many miRNAs are processed from long noncoding RNAs whose only known function is to produce the miRNA, some miRNA genes overlap other genes. For example, more than a quarter of the conserved miRNAs and more than half of the poorly conserved miRNAs of mammals appear to be processed from introns of protein-coding genes (Chiang et al., 2010). Thus, some pre-mRNAs are also pri-miRNAs, and vice versa (Figure 1B).
Of the 500-plus canonical miRNA genes confidently identified in the human genome, 296 fall within 177 seed families conserved among placental mammals (Table S1). Among these, 200 genes fall within 89 seed families conserved since the vertebrate ancestor of humans and bony fish, and among these, 75 genes fall within 27 seed families conserved since the bilaterian ancestor of humans, flies and nematodes (Table S1). One of these seed families (miR-99/100) predates the emergence of bilaterian animals, in that a closely related miRNA is found in sea anemone (Grimson et al., 2008). As a class of regulatory molecules, metazoan miRNAs apparently emerged even earlier—with the advent of multicellular animals, as indicated by the presence of both miRNAs and the metazoan Microprocessor in sponge but not more deeply branching species (Grimson et al., 2008). Assuming both a common ancestry of sponge and bilaterian miRNA pathways and a phylogeny in which sponges are the most deeply branching animal lineage (Simion et al., 2017), miRNAs were lost in both the placozoan and ctenophore lineages (Grimson et al., 2008; Moroz et al., 2014).
Because conserved miRNA genes can be found computationally (Lim et al., 2003) and tend to be expressed at easily detectable levels (Landgraf et al., 2007; Ruby et al., 2007b), the lists of conserved miRNA genes are likely to be nearly complete. Indeed, with one exception, all human miRNA genes conserved at least to fish were found at least 12 years ago, before the era of high-throughput sequencing. The exception is the MIR1306 gene (Friedlander et al., 2008; Morin et al., 2008), which was subsequently shown to be a negative-control element that overlaps the DGCR8 coding sequence (Han et al., 2009). Weak accumulation of mature miR-1306 suggests that its conservation is primarily due to protein-coding constraints and a negative-feedback role for the pri-miR-1306 hairpin in controlling DGCR8 expression, in which excess Drosha–DGCR8 activity crops the hairpin, thereby severing the DGCR8 mRNA (Han et al., 2009).
Although the lists of conserved miRNA genes appear to have approached completion, the number of confidently identified but poorly conserved genes continues to grow as more samples are subjected to high-throughput small-RNA sequencing, allowing very lowly or narrowly expressed miRNAs, which also tend to be poorly conserved, to exceed the thresholds for confident annotation. Thus, the recent tally of 519 is only a lower bound on the number of canonical miRNA genes in the human genome, and the actual number seems likely to exceed 600, with the number of confidently identified but poorly conserved human genes likely to surpass the 296 human miRNA genes belonging to families that were present in the last common ancestor of placental mammals. However, most of the poorly conserved miRNAs that remain unannotated have avoided identification because of either their very low or narrow expression levels, and thus current profiles of miRNAs presumably fail to account for only a tiny fraction of the miRNA molecules in the cell or tissue—an omission that seems all the more tolerable when considering the evidence that detectable miRNA-mediated repression requires high miRNA levels (i.e., hundreds if not thousands of molecules representing the miRNA seed family in each cell) (Bosson et al., 2014; Denzler et al., 2016).
Most poorly conserved miRNAs are classified as such because they are relatively young, deriving either from duplication and divergence of an ancestral gene or from the de novo emergence of a new gene (Ruby et al., 2007b; Fromm et al., 2015). Some of the recently emergent miRNAs are expressed at levels that could mediate consequential repression, and some of these found in humans might have critical primate-specific or even species-specific functions—perhaps helping to determine what makes us human. Indeed, in the Drosophila clade, clusters of poorly conserved miRNAs expressed in the testes are evolving under positive selection (Mohammed et al., 2018). More generally, however, the recent origins of most poorly conserved miRNAs suggests that poorly conserved miRNAs have not only been rapidly gained but also rapidly lost over the course of metazoan evolution, which in turn suggests that most poorly conserved miRNAs in contemporary species have probably not yet acquired a fitness-enhancing targeting interaction and seem destined to be lost before they do acquire an interaction favoring their selective retention.
Defining Features of Canonical miRNA Genes
Although hundreds of different conserved and nonconserved hairpins enter the miRNA biogenesis pathway, many thousands of others do not. The gatekeeper of the canonical pathway, which discriminates between authentic pri-miRNAs and the other hairpin-containing transcripts, is Microprocessor. When choosing the hairpins that will enter the pathway, the mammalian Microprocessor prefers a stem of 35±1 bp (Fang and Bartel, 2015), a terminal unstructured loop (Zeng et al., 2005), and single-stranded regions flanking the base of the hairpin (Zeng and Cullen, 2005; Han et al., 2006) (Figure 2). Although a few mismatches or small bulges are tolerated, pairing is preferred throughout the stem, except near the base of the stem, where a complex motif that centers on a mismatch at position 8 can modestly enhance processing (Fang and Bartel, 2015). In addition, three simple primary-sequence motifs—one at each end of the stem and another in the 3′ flanking region—enhance processing (Auyeung et al., 2013) (Figure 2).
Figure 2. The Menu of Structural and Primary-Sequence Features that Define Canonical miRNA Hairpins.
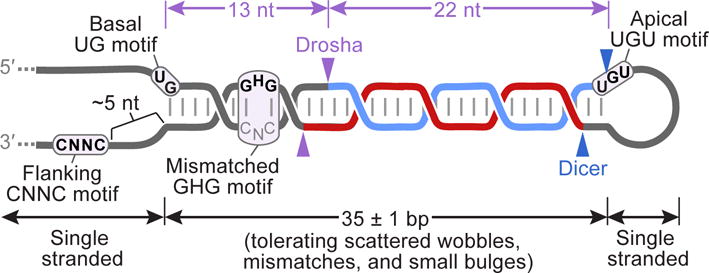
Structural features include a 35-bp stem with an unstructured loop and unstructured flanking regions. At every position within the stem except for one, pairing is preferred; this is typically Watson–Crick pairing, although at many positions one or both of the G–U wobble pairs is as favorable as some of the Watson–Crick pairs. Natural miRNA hairpins typically also have a few mismatches or small bulges scattered at various locations within the stem, and the mismatches are each counted as base pairs when describing the optimal length of 35 ± 1 bp. Additional features can enhance processing and help specify the sites of cleavage (purple arrowheads); these include a basal UG motif, an apical UGU motif, a flanking CNNC motif (in which N is any of the four nucleotides), and a mismatched GHG motif (in which H is A, C, or U), each located at the indicated positions relative to the Drosha cleavage sites (Auyeung et al., 2013; Fang and Bartel, 2015). Drosha recognizes the base of the hairpin, including the basal UG and probably the mismatched GHG motif, whereas the DGCR8 dimer recognizes the apical region, including the apical UGU (Nguyen et al., 2015), and together Drosha and DGCR8 form a molecular caliper that measures the length of the stem. An auxiliary factor, such as SRp20 or p72, recognizes the flanking CNNC motif (Auyeung et al., 2013; Mori et al., 2014). The sites of Dicer cleavage are also shown (blue arrowheads).
Microprocessor measures from both ends of a 35 bp stem, making offset cuts 13 and 11 nt from one end of the stem and 22 and 24 nt from the other end (Han et al., 2006; Ma et al., 2013; Fang and Bartel, 2015) (Figure 2). The motifs interact with either Microprocessor or its auxiliary factors to break the symmetry of this recognition, ensuring that Microprocessor binds in the orientation that cleaves closer to the base of the hairpin rather than the one that cleaves closer to the loop (Fang and Bartel, 2015; Nguyen et al., 2015). Interactions with these motifs also compensate for the structural defects (e.g., sub-optimal stem length, mismatches, and small bulges) observed in most natural pri-miRNAs, enhancing recognition and helping to specify the cleavage site (Fang and Bartel, 2015). Only a subset of the four motifs are required to orient the cleavage and compensate for the structural imperfections of most mammalian miRNA hairpins, and thus very few mammalian pri-miRNAs have all four motifs.
Although Microprocessor takes a lead role in choosing the transcripts that will proceed down the canonical miRNA pathway, the specificity of downstream processes, such as nucleocytoplasmic transport, Dicer-catalyzed cleavage, and Argonaute-loading (Figure 1A), can impose secondary constraints on the hairpins that can ultimately give rise to miRNAs. For example, some artificial hairpins with all four motifs can be cleaved by Microprocessor even if their stems are too short to generate a product suitable for Dicer cleavage (Fang and Bartel, 2015). However, the ideal substrates of Microprocessor give rise to pre-miRNA hairpins that have a 22-bp stem with a 5′ phosphate and a 2-nt 3′ overhang (Fang and Bartel, 2015), which are ideal substrates of the mammalian Dicer (Zhang et al., 2004; Park et al., 2011; Gu et al., 2012). Indeed, artificial genes designed solely on Microprocessor-preferred features generate miRNAs that accumulate to higher levels than do miRNAs deriving from co-expressed natural miRNA hairpins, further illustrating how Microprocessor preferences are in sync with those of downstream processes and largely define the genes that give rise to miRNAs (Fang and Bartel, 2015).
In most other bilaterian animals, the defining features of canonical miRNA genes resemble those identified in mammals. In nematodes, however, these features have diverged to the point that little if any miRNA is produced when C. elegans pri-miRNAs are expressed within mammalian cells (Auyeung et al., 2013). The defining features of C. elegans miRNA genes, as well as those from deeply branching animals (such as sponge or sea anemone) are mostly unknown.
Noncanoncial miRNA Genes
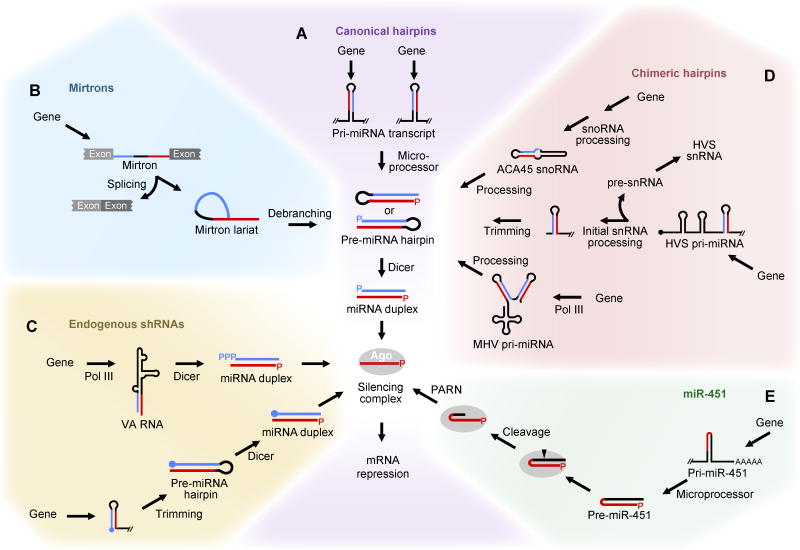
Some miRNA genes produce pri-miRNAs that bypass the requirement for either Drosha- or Dicer-catalyzed cleavage. For example, certain debranched introns enter the miRNA biogenesis pathway as pre-miRNA hairpins, with their ends having been defined by the spliceosome rather than by Drosha (Okamura et al., 2007; Ruby et al., 2007a) (Figure 3B). These RNAs that are both pre-miRNAs and introns are called “mirtrons” (Ruby et al., 2007a). In addition, some miRNA genes produce tailed mirtrons, which resemble classical mirtrons except they have a tail of flanking RNA at either the 5′ or 3′ end of the pre-miRNA hairpin, which is removed by non-Drosha nucleases before the pre-miRNA enters the pathway (Ruby et al., 2007a; Babiarz et al., 2008). Indeed, most mammalian mirtrons have a 5′ tail (Wen et al., 2015).
Figure 3. Comparison of Canonical and Noncanonical Biogenesis Pathways.
(A) The processing of canonical miRNA hairpins.
(B–D) The processing of mirtrons (B), endogenous shRNAs (C), and chimeric hairpins (D), which each feed into the canonical pathway after bypassing Microprocessor.
(E) The biogenesis of miR-451, which bypasses Dicer.
All genes are transcribed by Pol II, except as noted. Drawing conventions are as in Figure 1.
A second class of noncanonical miRNA genes produce endogenous short-hairpin RNAs (shRNAs) (Babiarz et al., 2008) (Figure 3C), which resemble the Drosha-independent type of artificial shRNAs designed for gene-knockout experiments (Tuschl, 2002). For a few endogenous shRNAs, namely those from two adenoviral hairpins transcribed by Pol III, transcriptional initiation and termination define the respective ends of the Dicer substrate (Andersson et al., 2005; Bellutti et al., 2015). However, for the best-characterized cellular endogenous shRNAs, which are Pol II products, the transcription start site defines the 5′ terminus of the Dicer substrate, and maturation of the 3′ terminus, although known to be Drosha-independent, is uncharacterized (Babiarz et al., 2008; Xie et al., 2013). Definition of the 5′ terminus by Pol II has downstream ramifications, in that the 5′ cap structure prevents the 5′ (5p) strand of the miRNA duplex from entering the silencing complex, and thus mature miRNAs accumulate from only the 3p strand (Babiarz et al., 2008; Xie et al., 2013). Many additional noncanonical miRNAs derive from hairpins that map to the 5′ regions of mRNAs or their divergent antisense transcripts; for these endogenous shRNAs, maturation of the 5′ and 3′ termini of the Dicer substrate, although known to be Drosha-independent, has not been characterized (Zamudio et al., 2014).
A third class of noncanonical miRNA genes are transcribed in tandem with or as part of another type of small-RNA gene, often depending on the transcriptional and processing determinants of the other RNA to help generate the hairpin substrate of Dicer (Figure 3D). For example, some miRNAs from murine γ-herpesvirus 68 (MHV68) derive from hairpins transcribed in tandem with tRNA-like molecules (Pfeffer et al., 2005), and miRNAs from a primate γ-herpesvirus derive from hairpins transcribed in tandem with Sm class snRNAs (Cazalla et al., 2011). Other examples of these chimeric hairpin RNAs include miR-1839, a mammalian miRNA that derives from a hairpin that falls within the ACA45 snoRNA (Ender et al., 2008), and miR-1983, a murine miRNA that derives from a hairpin that partially overlaps a tRNA (Babiarz et al., 2008).
The mammalian noncanonical miRNA with the most clearly established function is miR-451. miR-451 is one of the most highly expressed miRNAs in the erythrocytes of vertebrate animals, and mice lacking this miRNA have defects in erythroblast maturation (Patrick et al., 2010; Rasmussen et al., 2010). In contrast to the other known noncanonical miRNAs, which require Dicer but not Drosha for their biogenesis (Figure 3B–D), miR-451 requires Drosha but not Dicer (Cheloufi et al., 2010; Cifuentes et al., 2010; Yang et al., 2010). Following Drosha cleavage, the pre-miR-451 hairpin is too short to be cleaved by Dicer and instead directly enters Argonaute2 (Ago2), which cleaves the 3′ arm of the hairpin to generate a product that is further resected by the PARN exonuclease to produce the mature miRNA (Cheloufi et al., 2010; Cifuentes et al., 2010; Yang et al., 2010; Yoda et al., 2013) (Figure 3E). The question as to why throughout all vertebrate species miR-451, and seemingly only miR-451, is generated through this unusual pathway is one of the curiosities of miRNA evolution.
As more high-throughput sequencing datasets are analyzed, loci encoding mirtrons, tailed mirtrons, and endogenous shRNAs are making up a growing fraction of the reported miRNA genes of animals. Indeed, in species with the deepest small-RNA sequencing coverage, including human, the number of noncanonical miRNA loci appears to exceed the number of confidently annotated canonical miRNA genes (Zamudio et al., 2014; Wen et al., 2015). However, with a few exceptions, which include miR-63, a mirtron-derived miRNA highly expressed in C. elegans (Ruby et al., 2007a), miR-320, an endogenous shRNA highly expressed in mammals (Babiarz et al., 2008), and miR-451, noncanonical miRNAs of animals are both poorly conserved and lowly expressed, which suggests that most lack important regulatory functions. Indeed, in flies a degradation pathway preferentially destroys mirtronic miRNAs, implying that fly mirtrons might generally be detrimental if left unchecked (Bortolamiol-Becet et al., 2015; Reimao-Pinto et al., 2015).
miRNA Isoforms
Alternative choices along the miRNA biogenesis pathway can generate multiple miRNA isoforms (sometimes called “isomiRs”) from the same miRNA gene. For example, inconsistent choice of the strand loaded into Argonaute can generate functional miRNAs from both strands of the miRNA duplex, each with different sets of regulatory targets. Furthermore, imprecise cleavage by Drosha or Dicer can generate miRNAs with heterogeneous 5′ or 3′ ends, with heterogeneity at the 5′ end, which produces miRNAs with different seeds, having a profound effect on target recognition. In addition, different miRNA genes can be transcribed from the two different strands of the same DNA locus.
For most conserved miRNA hairpins, evolutionary selection appears to have enhanced the precision of biogenesis so as to maximize targeting specificity. Despite frequent heterogeneity at miRNA 3′ ends, conserved miRNAs typically have > 98% homogeneity with respect to both strand choice and 5′-end identity, and even higher homogeneity with respect to the strand of the DNA that gives rise to the miRNA (Chiang et al., 2010). There are, however, some interesting exceptions in which alternative isoforms expand the regulatory repertoire of conserved miRNA genes. For example, isoforms from both the 5′ and the 3′ strand of the miR-10 duplex (named miR-10-5p and miR-10-3p, respectively) appear to have regulatory roles in Drosophila (Ruby et al., 2007b; Stark et al., 2007a), and both the major and the minor 5′-end isoform of miR-223 (named miR-223.1 and miR-223.2, respectively) repress their respective targets in mouse neutrophils (Chiang et al., 2010). Overall, among the 90 miRNA families conserved to fish (89 canonical families plus miR-451), 12 produce at least one additional isoform that both has unique targeting potential and is expressed in human, mouse and zebrafish at ≥ 33% the level of the dominant isoform (Table S1)(Agarwal et al., 2015). Although no more than a very low level of miRNA production appears to occur from transcription in the antisense orientation in mammals (Chiang et al., 2010), transcription from both strands does expand the regulatory functions of one Drosophila locus (Bender, 2008; Stark et al., 2008; Tyler et al., 2008).
For a few miRNAs, alternative isoforms are also generated from adenosine-to-inosine (A-to-I) RNA-editing, which primarily occurs in the brain and is more prevalent in the human than in the mouse (Landgraf et al., 2007). Because I forms a Watson–Crick pair with C, not U, these edits can impact targeting, especially when they alter a seed nucleotide in a large fraction of the miRNA molecules (Kawahara et al., 2007b), although this occurs only rarely. Interestingly, the few miRNA edits likely to impact targeting alter mostly miRNAs that derive from an imprinted locus that includes 36 miRNA genes (Kawahara et al., 2008; Chiang et al., 2010).
Additional alternative isoforms are generated from terminal-nucleotide– transferase or 3′-exonuclease activities, which extend or trim miRNA 3′ termini. Although much more prevalent than A-to-I edits (Kim et al., 2016c), these modifications are not expected to substantially influence target recognition because they occur at the 3′ ends of the miRNAs. Instead, these terminal modifications are sometimes associated with pathways that influence miRNA processing and stability (Ha and Kim, 2014).
REGULATION OF miRNA EXPRESSION
Regulation of miRNA Production
MicroRNAs processed from the introns of mRNAs are under the same transcriptional regulation as the mRNA of their host genes (Baskerville and Bartel, 2005). All other canonical and nearly all other noncanonical miRNA loci are also under Pol II transcriptional control, which is indistinguishable from that of protein-coding genes (O’Donnell et al., 2005; Marson et al., 2008; Ozsolak et al., 2008). Exceptions include the two adenovirus shRNAs and the MHV68 chimeric hairpin RNAs, which are Pol III transcripts (Andersson et al., 2005; Bogerd et al., 2010; Diebel et al., 2010) (Figure 3).
Once the loci have been transcribed, diverse regulatory factors (often with additional, previously established cellular functions) influence the stability or processing of individual hairpins (Ha and Kim, 2014; Du et al., 2015; Treiber et al., 2017). For example, in C. elegans, targeting of the let-7 pri-miRNA by the mature let-7 miRNA can enhance processing in a positive-feedback loop (Zisoulis et al., 2012). In addition, multiple regulatory mechanisms influence the accumulation and activity of both Microprocessor and Dicer (Ha and Kim, 2014). Although changing these core activities broadly impacts miRNA production, individual miRNAs are differentially sensitive, which causes expression of some miRNAs to be affected more than others.
Of the posttranscriptional regulatory pathways that influence pri- or pre-miRNA processing or stability, one of the best-characterized involves the Lin28–directed oligo(U) tailing of the let-7 family of pre-miRNAs. Lin28 binds to the terminal loop of let-7 pre-RNAs, which promotes oligo(U)-tailing at the 3′ end of these pre-miRNAs, through the recruitment of terminal uridylyl transferases, TUT4 and TUT7 (Heo et al., 2008; Hagan et al., 2009; Heo et al., 2009; Nam et al., 2011). The oligo(U) blocks Dicer processing and stimulates pre-miRNA decay through the action of the 3′–5′ exonuclease, DIS3L2 (Heo et al., 2008; Chang et al., 2013; Ustianenko et al., 2013). Highlighting the potential biological relevance of this pathway, the let-7 miRNAs promote cellular differentiation (Melton et al., 2010), and Lin28 is one of the factors that can help reverse this differentiation during induction of pluripotent stem cells (Yu et al., 2007).
In a few cases, addition of a single nucleotide to the end of a pre-miRNA acts to enhance processing rather than inhibit it (Heo et al., 2012). Another post-transcriptional modification that influences miRNA production is A-to-I editing of pri-miRNAs, which can inhibit pri-miRNA processing and stimulate pri-miRNA degradation (Yang et al., 2006; Kawahara et al., 2007a).
Regulation of miRNA Decay
Once loaded into the silencing complex, most miRNAs are very stable, with half-lives of days (van Rooij et al., 2007; Bail et al., 2010; Gantier et al., 2011). Indeed, for many cases in which miRNAs are processed from the same primary transcripts as mRNAs, the miRNA accumulates to a much higher level than that of the co-transcribed mRNA, implying that the miRNA is much more stable. However, not all miRNAs are so stable. For example, some neuronal miRNAs have variable stability (Krol et al., 2010), and a few miRNAs are constitutively unstable, enabling a more rapid response to transcriptional changes (Rissland et al., 2011).
Artificial targets with extensive pairing to the miRNA can trigger miRNA degradation accompanied by 3′-terminal tailing and trimming of the mature miRNA (Ameres et al., 2010). In mammals, this phenomenon of target RNA–directed miRNA degradation (TDMD) is most robust in neuronal cells (de la Mata et al., 2015). In contrast to target repression, TDMD is particularly sensitive to the extent of pairing to the 3′ end of the miRNA, suggesting that this pairing might pull the 3′ end of the miRNA from the protective confines of the Argonaute protein, where it can then be acted upon by terminal transferases and exonucleases. The occurrence of TDMD among diverse eukaryotic species would explain why many classes of small silencing RNAs that routinely recognize targets with extensive complementarity (including piRNAs and endogenous siRNAs of animals, and miRNAs and siRNAs of plants) are methylated at their terminal 2′ oxygen, as this methylation prevents tailing (Li et al., 2005; Ameres et al., 2010).
Natural triggers of TDMD could in principle explain some of the differential stability observed among endogenous miRNAs (Marzi et al., 2016). Such triggers were first discovered among the noncoding RNAs (ncRNAs) of the herpesvirus family (Cazalla et al., 2010; Libri et al., 2012; Marcinowski et al., 2012; Lee et al., 2013), with pairing between the viral ncRNAs and host miRNAs shown to be critical for virus production, presumably because of the consequent TDMD (Marcinowski et al., 2012; Lee et al., 2013). More recently, transcripts that trigger TDMD have also been identified among cellular RNAs (Bitetti et al., 2018; Kleaveland et al., 2018). For example, in some tissues, the long ncRNA Cyrano reduces miR-7 levels by > 97%, with the efficiency of this TDMD greatly exceeding that of artificial or viral transcripts (Kleaveland et al., 2018). This targeted degradation of miR-7 can derepress mRNA targets of miR-7, but more strikingly, it enables a circular RNA called “Cdr1as” to accumulate in processes and other cytoplasmic regions of mammalian neurons (Kleaveland et al., 2018). Cdr1as is required for proper neuronal activity, acting through an unknown mechanism thought to involve miR-7, as Cdr1as orthologs found throughout placental mammals each have many sites to miR-7 (e.g., 73 and 130 miR-7 sites in human and mouse Cdr1as, respectively) (Hansen et al., 2013; Memczak et al., 2013; Piwecka et al., 2017).
miRNA REGULATORY MECHANISMS
In animals, the recognition and repression of target transcripts can occur through two alternative modes (Figure 1A). If pairing between the miRNA and the target site is sufficiently extensive, and if the miRNA is associated with an Argonaute protein that has retained its ancestral ability to catalyze the endonucleolytic cleavage characteristic of RNA interference (RNAi) (Liu et al., 2004; Meister et al., 2004), then the miRNA directs slicing of the target transcript (Hutvagner and Zamore, 2002; Yekta et al., 2004) (Figure 1A). This slicing mode of repression is common for plant miRNAs (Jones-Rhoades et al., 2006) and might also play a widespread role in sea anemone (Moran et al., 2014). It is also the basis of siRNA-mediated mRNA-knockdown technologies that have transformed biomedical research and show promise in the clinic (Elbashir et al., 2001; Bobbin and Rossi, 2016). In humans and other mammals, however, miRNA-directed slicing has been reported for only 20 cellular transcripts (Yekta et al., 2004; Davis et al., 2005; Shin et al., 2010), one circular RNA (Hansen et al., 2011), and a few viral mRNAs (Sullivan et al., 2005; Barth et al., 2008), and examples of endogenous miRNA-directed slicing have yet to be reported in non-mammalian bilaterian animals. Thus, most bilaterian miRNAs do not direct the slicing of a single endogenous target.
The repression mode that dominates in humans and other bilaterian animals acts without slicing the mRNA and does not require extensive pairing to the miRNA (Figure 1A). Instead, it requires the adaptor protein TNRC6 (present in mammals as three paralogs, TNRC6A/B/C, which have a single ortholog in flies, named GW182, and two in nematodes, named AIN-1/2) (Ding et al., 2005; Rehwinkel et al., 2005; Jonas and Izaurralde, 2015). Recruited by Ago, TNRC6 interacts with the poly(A)-binding protein (PABPC) associated with the mRNA poly(A) tail and also recruits deadenylase complexes, i.e., the PAN2–PAN3 complex and most importantly the CCR4–NOT complex (Jonas and Izaurralde, 2015) (Figure 4). The deadenylases shorten the poly(A) tail, which in most systems causes mRNA destabilization through decapping and 5′-to-3′ exonucleolytic decay (Chen and Shyu, 2011). Recruitment of TNRC6 also causes the mRNA to be less efficiently translated, an effect on translation initiation thought to be mediated, at least in part, by CCR4-NOT and its recruitment of DDX6, a helicase that binds the decapping complex and is reported to inhibit translation (Chu and Rana, 2006; Jonas and Izaurralde, 2015) (Figure 4). Moreover, DDX6 interacts with eIF4E transporter (4E-T), which competes with eIF4G for binding to eIF4E and enhances both the decay and translational repression of miRNA targets (Kamenska et al., 2014; Nishimura et al., 2015; Ozgur et al., 2015; Kamenska et al., 2016).
Figure 4. The Dominant Mechanisms of miRNA-guided Repression in Bilaterian Animals.
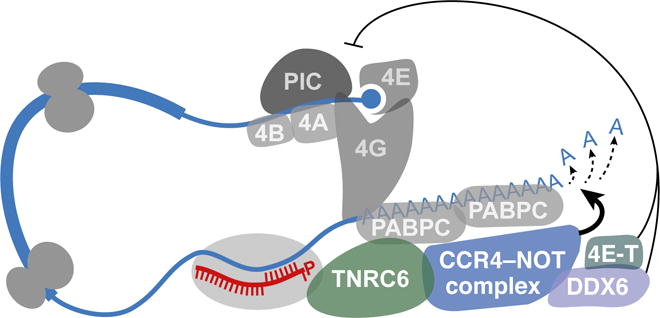
Guided by the miRNA, the silencing complex associates with the mRNA and recruits TNRC6, which interacts with PABPC and recruits either the PAN2–PAN3 deadenylase complex (not shown) or the CCR4–NOT deadenylase complex, either of which shortens the mRNA poly(A) tail. Alternative downstream consequences of poly(A)-tail shortening, which are not depicted in this figure, consummate this major mode of TNRC6-mediated repression; in early embryos tail shortening reduces translation initiation with little effect on mRNA stability, whereas in most other developmental contexts tail shortening hastens decapping and degradation of the mRNA with relatively little effect on translation initiation. Although not through tail shortening, recruitment of TNRC6 can nonetheless repress translation initiation in postembryonic cells through a parallel mechanism that involves CCR4–NOT-mediated recruitment of DDX6 and 4E-T. This translation initiation normally involves the recruitment of the 43S preinitiation complex (PIC) through the action of initiation factors (4A, 4B, 4E, 4G).
The relative importance of mRNA decay and translational repression, the two repressive consequences of this TNRC6-mediated regulatory mode, depends on the developmental context of the cell. In all post-embryonic cells examined, mRNA decay dominates, as revealed by experiments comparing changes in mRNA levels with changes in either protein levels or translational efficiency after either introducing or knocking out a miRNA (Baek et al., 2008; Hendrickson et al., 2009; Guo et al., 2010; Eichhorn et al., 2014). Indeed, regardless of miRNA identity, cell type, growth condition, or translational state, mRNA destabilization explains most (66% to >90%) of the steady-state repression mediated by mammalian miRNAs (Eichhorn et al., 2014). Examination of the dynamics of translational repression and mRNA destabilization as a miRNA is induced shows that although translational repression occurs rapidly (Bethune et al., 2012; Djuranovic et al., 2012), its effect on endogenous mRNAs is relatively weak, such that by the time consequential repression ensues, the effect of mRNA destabilization dominates (Eichhorn et al., 2014). These results are welcome news for those seeking to determine the molecular consequences of a miRNA. In post-embryonic contexts, the miRNA can be perturbed, and the effects on target mRNA levels (which are easier to measure than effects on either protein levels or translational efficiency) provide a nearly quantitative readout of the miRNA-mediated repression.
In the early zebrafish embryo, however, translational repression is the only repressive consequence of the TNRC6-mediated regulation, and thus monitoring mRNA levels completely misses the effects of miRNAs (Bazzini et al., 2012; Subtelny et al., 2014). This difference in the consequences of miRNA repression occurs because the post- transcriptional regulatory context is very different in the early embryo compared to later embryos or post-embryonic cells, which leads to very different consequences of miRNA-mediated poly(A)-tail shortening. In early embryos, shortening the tail of an mRNA does not change its stability but dramatically decreases its translational efficiency, whereas at gastrulation and in post-embryonic cells, shortening the tail of an mRNA reduces its stability but does not change its translational efficiency (Subtelny et al., 2014).
miRNA TARGET RECOGNITION
Canonical Regulatory Sites
For sites that promote TNRC6-mediated repression, target recognition is primarily through Watson–Crick pairing between the miRNA seed (miRNA nucleotides 2–7; distinguished in this review from the “extended seed” or the “seed region,” which include miRNA nucleotides 2–8) and sites within the 3′ UTRs of target mRNAs (Bartel, 2009). Augmenting the 6-nt seed match, most of these regulatory sites also have either an additional match to miRNA nucleotide 8 or an A across from miRNA nucleotide 1, or both, to make 7- or 8-nt sites, respectively (Lewis et al., 2005) (Figure 5A). These 7–8-nt sites mediate the bulk of the repression for each miRNA and are the sites identified by the most effective target-prediction tools (Bartel, 2009; Agarwal et al., 2015). Nonetheless, the 6-nt sites that either match only the seed or are offset by one nucleotide in either the 5′ or 3′ direction (Figure 5A), can sometimes also mediate detectable repression (Friedman et al., 2009; Jan et al., 2011; Kim et al., 2016a), as can 7–8-nt sites in ORFs or 5′ UTRs (Lewis et al., 2005; Stark et al., 2007b; Schnall-Levin et al., 2010; Agarwal et al., 2017). Pairing to the 3′ region of the miRNA, particularly pairing involving miRNA nucleotides 13–16, can supplement pairing to the seed region (Bartel, 2009) (Figure 5B), but this 3′-supplementary pairing has surprisingly little influence on site affinity and efficacy (Grimson et al., 2007; Wee et al., 2012; Salomon et al., 2015), and only about 5% of the seed-matched regulatory sites undergoing purifying selection appear to include this additional pairing (Friedman et al., 2009).
Figure 5. MicroRNA Target Sites.
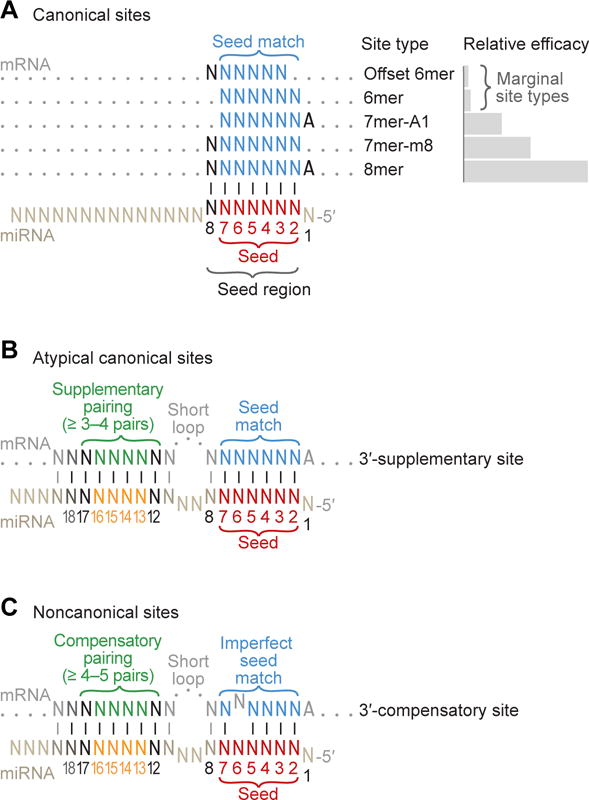
(A) Canonical sites of mammalian miRNAs. These canonical sites each have 6–7 contiguous Watson–Crick pairs (vertical lines) to the seed region of the miRNA (miRNA positions 2–8). Two of these sites also include an A at position 1. Relative site efficacy in mammalian cells is graphed to the right (log scale). The most effective canonical sites are 7–8 nt sites that include a perfect match to the miRNA seed (positions 2–7, red), whereas the 6 nt sites are the least effective. Not shown is the 6mer-A1 site (the same as the 7mer-A1 but lacking a pair at position 7), which is rarely conserved above background in mammalian mRNAs and has negligible efficacy in mammalian cells yet is sometimes included among the canonical sites because of its more robust conservation and efficacy in C. elegans (Jan et al., 2011).
(B) The 3′-supplementary site, an atypical type of canonical site. A small fraction of the canonical sites (< 5%) benefit from pairing to the 3′ region of the miRNA. Productive 3′-supplementary pairing typically centers on nucleotides 13–16.
(C) The 3′-compensatory site, a functional type of noncanonical site. The noncanonical sites do not have six contiguous Watson–Crick pairs to the seed region. Shown is a mismatch at position 6, but the imperfection (either a wobble, mismatch, or single-nucleotide bulge) can instead occur at another seed position. Compensating for the imperfect seed match is extensive pairing to the 3′ region of the miRNA, which typically centers on miRNA nucleotides 13–16.
The preference for an A across from the first nucleotide of the miRNA, which is observed regardless of the miRNA sequence (Figure 5A), implies that the miRNA does not pair with this target nucleotide but rather a protein prefers an A at this position (Lewis et al., 2005). Indeed, structural analyses show that the first nucleotide of the miRNA is buried within Ago, unable to interact with the target (Ma et al., 2005; Parker et al., 2005), and that Argonaute has a pocket that specifically binds an A at this position (Schirle et al., 2015).
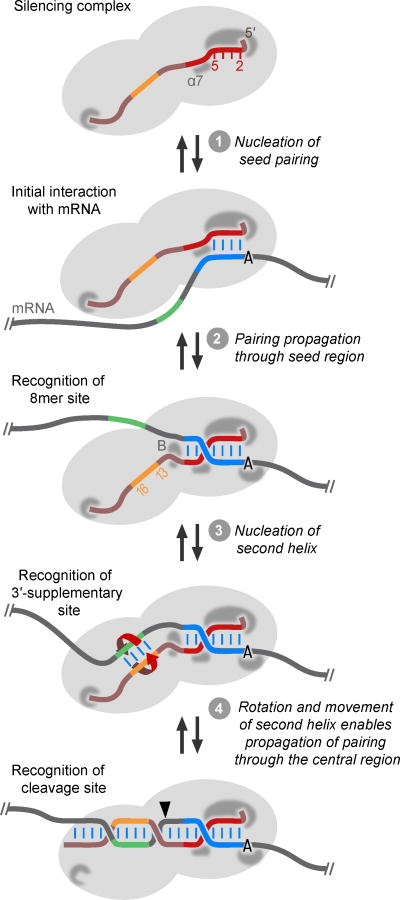
The ‘seed’ was originally thought to be a segment of the miRNA that was fully preorganized and accessible for target recognition (Bartel, 2004). Structural results have revised this idea, revealing that a guide-RNA kink and steric barrier imposed by Ago α–helix 7 leaves only miRNA nucleotides 2–5 suitably preorganized and accessible for the target search (Elkayam et al., 2012; Nakanishi et al., 2012; Schirle and MacRae, 2012) (Figure 6A, step 1), and single-molecule analyses demonstrate that this sub-seed segment is indeed most critical for target association (Chandradoss et al., 2015; Salomon et al., 2015). As pairing propagates to miRNA nucleotides 6–8, α-helix 7 is displaced and assumes a conformation that reinforces perfect pairing to nucleotides 6 and 7 (Schirle et al., 2014; Klum et al., 2018) (Figure 6, step 2). Also in this region of the protein is a series of serine residues that undergo a phosphorylation cycle that promotes proper targeting (Golden et al., 2017; Quevillon Huberdeau et al., 2017).
Figure 6. A Unified Model for miRNA Target Recognition and Pairing Propagation.
The miRNA is tightly bound within the silencing complex. A pocket within the Ago MID domain binds the miRNA 5′ terminus, and one within the PAZ domain binds its 3′ terminus. In addition, interactions with the seed region (red) pre-organize nucleotides 2–5 for initial pairing with the target (step 1). As pairing propagates through the seed region (step 2), α-helix 7 (α7), which initially imposes a kink in the seed and steric block to this propagation, shifts to a location at which it reinforces Watson–Crick paring at positions 6 and 7. Most canonical sites rely on this pairing to the seed region, often supplemented with an additional interaction between Ago and an A at target position 1, but with a steric and conformational block (B) preventing contiguous pairing from propagating beyond position 8. Some sites have a segment (green) that can pair to the miRNA 3′ region, optimally centering on miRNA nucleotides 13–16 (orange) to nucleate a second RNA helix (step 3). Because this second helix forms on the other side of the block, it can form without either steric hindrance or the conformational challenge of wrapping the miRNA around the mRNA. This second helix contributes the 3′-supplementary/compensatory pairing observed for some sites. For the very few sites that also have pairing through the center of the miRNA, pairing can then form between the second helix and the seed helix, propagating from either helix or from both simultaneously (step 4). To accommodate the torsional strain of forming this pairing between the helices, the second helix rotates around its helical axis, while the seed helix remains fixed. As the second helix begins to rotate and as pairing propagates from the second helix towards the miRNA 3′ terminus, the 3′ terminus is pulled from its binding pocket, relieving strain for further rotation of the second helix and full propagation of pairing (step 4). Propagation of pairing through the block and other RNA conformational changes that generate a long contiguous helix are coupled to protein conformational changes that help create the active site for target slicing (arrowhead). Each step prior to slicing is reversible, and thus this model suggests an analogous pathway for the stepwise release of the passenger strand during silencing-complex maturation, in which a miRNA duplex replaces the fully paired miRNA–target complex, and the pathway proceeds in reverse.
The ‘seed’ was given its name with the idea that it nucleated pairing that could then propagate to other parts of the miRNA (Bartel, 2004), and for the extensively paired sites that can be sliced, pairing is still commonly thought to grow from the seed, propagating 5′-to-3′ along the miRNA (Wang et al., 2009). However, this idea faces some serious topological obstacles, as tight binding of the seed region would keep this region stationary during propagation, and thus contiguous propagation would require challenging conformational gymnastics to enable the guide strand to wrap two full turns around the target strand (Bartel, 2009). Here, I propose an alternative model, which is more parsimonious with these topological considerations as well as the preferred region of 3′-supplementary pairing. In this model, instead of propagating directly from the seed helix, pairing skips from the seed region to a second nucleation region, generating an intermediate that resembles the conformation observed with most 3′-supplementary sites (Figure 6, step 3). Pairing then forms on both sides of this second helix. Because the seed region is fixed within its binding channel, it remains stationary while the second helix rotates around its helical axis to enable pairing to propagate between the two helices (from either or both directions) to include miRNA nucleotides 9–12 (Figure 6, step 4). As the second helix begins to rotate and as pairing propagates beyond miRNA nucleotides 17, the miRNA 3′ terminus is pulled from its binding pocket, thereby helping to free the second helix for further rotation (Figure 6, step 4). In such a model, pairing to the miRNA central region and the accompanying conformational change required to position the Argonaute active site for target slicing would occur only for targets with suitable pairing to both the seed and the miRNA 3′ region. Importantly, all four steps are reversible, which also provides a pathway for the stepwise release of the passenger strand after a miRNA duplex has been loaded.
Additional Regulatory Sites and Binding Sites
Noncanonical sites, which lack a contiguous 6-nt match to the seed region, can also mediate repression, although only one type of noncanonical site is known to both mediate readily detectable repression and yield a significant signal above background in global analyses of conservation. This is the 3′-compensatory site, for which extensive pairing to the miRNA 3′ region compensates for imperfect pairing to a core seed nucleotide (Figure 5C) (Bartel, 2009). These 3′-compensatory sites account for less than 1% of the preferentially conserved miRNA sites in mammalian mRNAs, presumably because their large size compared to the size of canonical sites makes them correspondingly more difficult to acquire and retain in mRNA sequences (Friedman et al., 2009). Nonetheless, a potential advantage of these larger sites is that their strong dependence on pairing to miRNA nucleotides falling outside the seed region offers a mechanism to avoid the regulatory redundancy often observed for co-expressed miRNAs from the same seed family. For example, the need to respond with temporal precision explains why the lin-41 mRNA of nematodes has two 3′-compensatory sites that respond specifically to the let-7 miRNA and not to earlier-expressed paralogous miRNAs with the same seed (Brennecke et al., 2005; Lewis et al., 2005; Ecsedi et al., 2015).
Although miRNAs primarily function at canonical sites, their binding appears more promiscuous. High-throughput analyses of mRNA segments that crosslink to Argonaute show that in cells miRNA silencing complexes bind to many sites that lack contiguous 6- nt matches to the seed region, such that in aggregate these noncanonical sites account for approximately half of the intracellular crosslinks (Chi et al., 2009; Hafner et al., 2010; Loeb et al., 2012; Helwak et al., 2013; Grosswendt et al., 2014). However, the noncanonical binding sites identified by crosslinking do not function to mediate detectable repression, in that mRNAs with only these sites are no more responsive to miRNA perturbation than are other mRNAs without canonical sites (Agarwal et al., 2015). Presumably each miRNA typically samples many low-affinity noncanonical sites before encountering a site with sufficient dwell time to enact repression, such that in aggregate it spends at least as much time at the abundant noncanonical sites as at the functional canonical ones (Agarwal et al., 2015; Denzler et al., 2016).
miRNA Target Abundance
By virtue of their small size, seed-matched regulatory sites can readily emerge in 3′ UTRs, enabling each miRNA to have a widespread influence on mRNA regulation and 3′ UTR evolution (Farh et al., 2005; Stark et al., 2005). Indeed, within human 3′ UTRs, each of the 90 broadly conserved miRNA families has an average of >300 7–8-nt sites under selective pressure to maintain their pairing to the miRNA, and this average exceeds 500 when including the 6-nt sites (Friedman et al., 2009). Some 3′ UTRs are targeted more than once by the same miRNA, and 3′ UTRs with a preferentially conserved site to one miRNA family usually have preferentially conserved sites to other families (with each conserved miRNA target preferentially conserving sites to an average of 4–5 different miRNA families). Yet despite this concentration of targeting of some miRNAs compared to others, the average number of conserved targets per family exceeds 400, and 3′ UTRs of 57.8% ± 3.0% (95% confidence interval) of the human mRNAs are conserved targets of miRNAs (Friedman et al., 2009). When also considering other preferentially conserved sites, such as those observed in open reading frames, albeit at a lower density than in 3′ UTRs (Lewis et al., 2005; Stark et al., 2007b; Schnall-Levin et al., 2010), and those observed for more recently emergent miRNA families, the estimate of conserved targets confidently exceeds 60% of human mRNAs (Friedman et al., 2009). For nematodes and flies, the respective estimates are lower, at 27% and 37% of annotated mRNAs, respectively (Jan et al., 2011; Agarwal et al., 2017). Experiments that globally examine the molecular effects of perturbing a miRNA family not only confirm repression of many mRNAs with conserved sites but also reveal repression of even more mRNAs with non-conserved seed-matched sites (Krutzfeldt et al., 2005; Lim et al., 2005; Giraldez et al., 2006; Rodriguez et al., 2007). Thus, it would seem difficult for a developmental or physiological process to escape the influence of miRNAs.
Although low-affinity noncanonical sites contribute little to repression, they do contribute, by virtue of their sheer numbers, to the effective number of sites in the cell that compete for miRNA binding. As a result, the effective target abundance (defined as the number of sites that must be added to achieve half-maximal de-repression of targets) in each cell typically exceeds the miRNA abundance—even for very highly expressed miRNAs such as miR-122 in hepatocytes, which is present at 120,000 molecules per cell, yet has an even greater effective target abundance (Denzler et al., 2014; Denzler et al., 2016).
The high effective target abundances have important ramifications. For example, with so many sites competing for miRNA binding, few if any sites are saturated, and thus even the most repressed targets are sensitive to increased miRNA levels. High effective target abundances also refute the plausibility of the competing endogenous RNA (ceRNA) hypothesis (Denzler et al., 2014; Denzler et al., 2016), which posits that changes in the expression of individual cellular targets commonly and perceptibly influence the repression of other cellular targets by modulating the amount of free miRNA (Salmena et al., 2011). Moreover, the high effective target abundances explains why lowly expressed miRNAs have negligible effects, which in turn seriously challenges the idea that the miRNAs delivered by exosomes might reach consequential levels in recipient cells.
miRNA Target-Prediction Strategies
Numerous algorithms have been developed to predict the regulatory targets of miRNAs, with at least 25 sets of target predictions from at least 16 different groups available for the human miRNAs (Bartel, 2009; Agarwal et al., 2015). To reliably predict targets of a miRNA, the first step is to search for 7–8-nt canonical sites in annotated 3′ UTRs. On its own, this simple text-search step generates predictions that are more responsive to the miRNA than those of some of the popular algorithms (Agarwal et al., 2015). However, not all mRNAs with a 7–8-nt canonical site respond to the miRNA (Baek et al., 2008; Selbach et al., 2008), and thus additional strategies that further differentiate among these predictions can add value. One such strategy is to choose 3′ UTRs with sites that appear to have been selectively conserved in evolution (Bartel, 2009), as mRNAs with conserved sites to a broadly conserved miRNA are about twice as likely to respond to the loss of that miRNA than those with poorly conserved sites (Baek et al., 2008). However, even when imposing a conservation cutoff, the lists of predicted targets typically include hundreds of unique mRNAs for each highly conserved miRNA family. If these lists are too large for experimental follow-up, one option is to apply a more stringent conservation cutoff to prioritize those with the greatest probability of conserved targeting, which tend to be even more responsive to the miRNA than mRNAs with lower probability of conserved targeting (Friedman et al., 2009).
Despite utility of prioritizing conserved targets, some of the most responsive targets are not among those with the most confidently detected site conservation. Moreover, the miRNAs that emerged more recently, including those conserved no further than placental mammals, have too few targets under selective conservation for any of the conserved targets to be confidently predicted above the background (Friedman et al., 2009). Thus, to supplement and complement the analysis of conservation, quantitative models have been developed that use the features of the sites and their contexts within 3′ UTRs to predict targeting efficacy. Features of sites that correlate with efficacy include the type of site (Figure 5A), the number of sites, the potential for 3′-supplementary pairing, and site context (Grimson et al., 2007). With respect to site context, the more effective sites tend to be near the edges of UTRs (but ≥ 15 nt downstream of the stop codon to avoid displacement of the silencing complex by the ribosome) and within regions predicted to be more structurally accessible (Bartel, 2009). Also correlating with efficacy are features of the miRNA, including its predicted seed-pairing stability and the number of its transcriptome sites competing for binding (Garcia et al., 2011), and features of the mRNAs, including UTR length, ORF length, presence of alternative 3′-UTR isoforms, and presence of additional marginal sites in 3′ UTRs and ORFs (Agarwal et al., 2015). Quantitative models that parameterize informative features and then train on high-throughput measurements of miRNA effects are becoming increasingly accurate at predicting the mRNAs most responsive to miRNA perturbation (Agarwal et al., 2015). Indeed, the most accurate of these quantitative models is as informative as the best high-throughput in vivo crosslinking approaches, even after filtering the crosslinking results to include only mRNAs with a canonical site to the miRNA (Agarwal et al., 2015). Despite matching the utility of the crosslinking methods, the computational approach still has room for improvement, with both false positives and false negatives. Nonetheless, because the experimental approach also yields false positives and false negatives, the computational model and experimental approach happen to perform similarly.
miRNA BIOLOGICAL FUNCTIONS
Biological Importance of miRNAs
Systematic generation and analysis of miRNA knockouts in C. elegans created the initial impression that most metazoan miRNAs—even most miRNAs that are broadly conserved—have only subtle or redundant biological functions (Miska et al., 2007; Alvarez-Saavedra and Horvitz, 2010). Excluding genes that fall within the lin-4, let-7, and lsy-6 miRNA families, which already had genetically informed functions (Lee et al., 1993; Reinhart et al., 2000; Johnston and Hobert, 2003), (and also excluding mutations of 13 loci that corresponded to false-positive miRNA annotations), this effort examined 83 miRNA genes representing 48 seed families (including 38 families that are both conserved in other nematodes and for which every known member was simultaneously disrupted). Despite the scale of the effort, abnormal phenotypes were initially observed for mutants in only five of the 48 families (miR-35, miR-51, miR-58, miR-240, and miR-786) (Miska et al., 2007; Alvarez-Saavedra and Horvitz, 2010). With more thorough analyses, abnormal phenotypes have since been observed for mutants in another seven of these families when grown under normal lab conditions (miR-34, miR-71, miR-79, miR-83, miR-238, miR-246, miR-259) (de Lencastre et al., 2010; Boulias and Horvitz, 2012; Pedersen et al., 2013; Burke et al., 2015), and for mutants in another four when examined in the context of stress conditions (miR-63, miR-67, miR-86, and miR-231) (Nehammer et al., 2015; Yang et al., 2016; Ma et al., 2017). In addition, an abnormal phenotype has been observed after disrupting a member of a family not examined in the initial effort (miR-791) (Drexel et al., 2016). However, for most miRNA families disrupted in C. elegans, abnormal phenotypes have either not been detected or have been detected only in the context of sensitized genetic backgrounds (Brenner et al., 2010).
Interestingly, this notion that most individual miRNA families have only subtle or redundant biological functions seems to apply uniquely to nematodes, in that a very different conclusion has emerged for both flies and mammals. In a systematic analysis of miRNA knockout phenotypes in flies, 69 of 88 knockouts examined (78%) had severe mutant phenotypes scored with >99% confidence (Chen et al., 2014), and this fraction increased slightly to 65 of 81 (80%) when considering knockouts of only canonical miRNAs conserved at least throughout flies, and to 23 of 28 (82%) for the subset of mutants that disrupt a miRNA in a family conserved since the ancestor of bilaterian animals.
With respect to mice, knockouts have been reported for members of 20 of the 27 mammalian miRNA families conserved since the bilaterian ancestor, and for each of these 20, abnormal phenotypes have been observed (Table 1). Abnormal knockout phenotypes also have been reported for another 36 miRNA families conserved at least to fish (Table 2). Thus, of the 90 mammalian miRNA families conserved to fish, 56 (62%) have been associated with an abnormal knockout phenotype. Among these 56, four have abnormalities observed only in the context of a disease or injury model (miR-130/301, miR-139, miR-192/215, and miR-214; Table 2). However, for the other 52, a broad diversity of defects are observed for mice examined under normal lab conditions, often with severe consequences. For example, mutants in 15 families have significantly reduced viability manifested either in utero or prior to weaning, and others cause fatal neurological disorders, infertility, blindness, deafness, immune disorders, or cancer (Tables 1 and 2). Analyses of the effects of simultaneously disrupting miRNAs from multiple families (typically expressed from polycistronic loci) attribute functions to as many as 13 additional miRNA families conserved at least to fish (Table 3). Thus, of the 90 mammalian miRNA families conserved to fish, 69 (77%) have been associated with an abnormal knockout phenotype, and many of the remaining 21 have yet to be examined.
Table 3. Abnormal phenotypes observed in mice after knocking out miRNAs representing multiple miRNA families.
For each miRNA family, its conservation is listed in parentheses, together with the number of murine genes that contribute to that family. The conservation categories are bilaterian (bilat., conserved since the bilaterian ancestor), vertebrate (vert., conserved among mammals and fish but emerged since the bilaterian ancestor), eutherian (euth., conserved among placental mammals but not to fish), and poorly conserved (p.c., not conserved among placental mammals). Each strain has more than one miRNA gene disrupted; multiple miRNAs from a disrupted polycistronic locus are linked with a tilde (~), whereas those from distant loci are separated with a comma.
| miRNA families (conservation, # of genes) | miRNA(s) removed | Phenotype |
|---|---|---|
| miR-1/206 (bilat. 3), miR-133 (bilat. 3) | miR-1-1 ~ miR-133a-2, miR-1-2 ~ miR-133a-1 | Early embryonic lethality, complete penetrance, due to severe heart malformations (Wystub et al., 2013) |
| miR-96 (bilat. 1), miR-183 (bilat. 1), miR-182 (vert. 1) | miR-183 ~ 96 ~ 182 | Retinal degeneration following progressive synaptic defects of photoreceptors; impaired balance (Lumayag et al., 2013); decreased corneal nerve density; increased neutrophil activity; decreased disease with Pseudomonas infection of the cornea (Muraleedharan et al., 2016) |
| miR-96 (bilat. 1), miR-183 (bilat. 1) | miR-183 ~ 96 | Defects in retinal cone development, progressive loss of photoreceptor fuction, retinal degeneration; impaired balance; impaired olfactory function (Xiang et al., 2017) |
| miR-193 (bilat. 2), miR-365 (bilat. 2) | miR-193b ~ 365-1 | Increased mammary stem/progenitor cells; accelerated differentiation of mammary epithelium during puberty and pregnancy (Yoo et al., 2014) |
| miR-25/32/92/363/367 (bilat. 7), miR- 17/20/93/106 (vert. 6), miR-18 (vert. 2), miR-19 (vert. 3) | miR-17 ~ 18 ~ 19a ~ 20a ~ 19b-1 ~ 92a-1 | Perinatal lethality, complete penetrance, due to lung and heart defects; vertebral homeotic transformations and other skeletal defects; reduced body weight; reduced pre-B cells (Ventura et al., 2008; Han et al., 2015); conditional deletion perturbs development and/or function in each of 18 different cell types |
| miR-25/32/92/363/367 (bilat. 7), miR-17/20/93/106 (vert. 6), miR-18 (vert. 2), miR-19 (vert. 3) | miR-17 ~ 18 ~ 19a ~ 20a ~ 19b-1 ~ 92a-1, miR-106b ~ 93 ~ 25 | Embryonic lethality, complete penetrance, at midgestation (Ventura et al., 2008) |
| miR-25/32/92/363/367 (bilat. 7), miR-17/20/93/106 (vert. 6) | miR-106b ~ 93 ~ 25 | Enhanced adiposity leading to insulin resistance (Cioffi et al., 2015); cardiac arrhythmia (Chiang et al., 2014) |
| miR-23 (vert. 2), miR-24 (vert. 2), miR-27 (vert. 2) | miR-23a ~ 27a ~ 24-2, miR-23b ~ 27b ~ 24-1 | Hypersensitive T cells; increased T helper cell responses and tissue pathology in an immune disease model (asthma) (Pua et al., 2016); decreased hematopoietic stem cells and progenitors; skewed hematopoiesis, with B cells produced at the expense of myeloid cells (Kurkewich et al., 2016; Kurkewich et al., 2017) |
| miR-200bc/429 (bilat. 3), miR-200a/141 (vert. 2) | miR-200c ~ 141 | Impaired enamel formation (Cao et al., 2013) |
| miR-200bc/429 (bilat. 3), miR-200a/141 (vert. 2) | miR-200b ~ 200a ~ 429 | Adipocyte-specific knockout causes increased obesity with high-fat diet (Tao et al., 2016) |
| miR-143 (vert. 1), miR-145 (vert. 1) | miR-143 ~ 145 | Arterial smooth muscle defects with perturbed actin stress fibers; increased lethality and less scarring in response to vascular injury (Elia et al., 2009; Xin et al., 2009); swelling of the kidney due to impaired ureter function (Medrano et al., 2014); reduced intraocular pressure (Li et al., 2017); altered pathology in several disease/injury models |
| miR-144 (vert. 1), miR-451 (vert. 1) | miR-144 ~ 451 | Increased incidence of spontaneous B lymphoma and acute myeloid leukemia in aged mice (Ding et al., 2017) |
| miR-216a (bilat. 1) miR-216b (vert. 1), miR-217 (vert. 1) | miR-216b ~ 216a ~ 217 | Embryonic lethality at about E9.5, complete penetrance (Azevedo-Pouly et al., 2017) |
| miR-199 (vert. 3), miR-214 (vert. 1) | miR-199a-2 ~ 214 | Neonatal and postnatal lethality, partial penetrance, smaller body size, skeletal abnormalities, partial paralysis (Watanabe et al., 2008) |
| miR-291a/294/302abd (vert. 5), 4 other less broadly conserved families | miR-290 ~ 291a ~ 292 ~ 291b ~ 293 ~ 294 ~ 295 | Embryonic lethality, partially penetrant; female sterility with ovarian failure due to defects in primordial germ cell migration (Medeiros et al., 2011); smaller placenta with defective maternal–fetal transport (Paikari et al., 2017) |
| miR-291a/294/302abd (vert. 5), miR-302c (vert. 1) | miR-302b ~ 302c ~ 302a ~ 302d | Late embryonic lethality, complete penetrance; failure of neural tube closure, with precocious neuronal differentiation and increased proliferation of neural progenitors; abnormal eye development (Parchem et al., 2015) |
| miR-291a/294/302abd (vert. 5), miR-302c (vert. 1), 4 other less broadly conserved families | miR-290 ~ 291a ~ 292 ~ 291b ~ 293 ~ 294 ~ 295, miR-302b ~ 302c ~ 302a ~ 302d | Early embryonic lethality, complete penetrance (Parchem et al., 2015) |
| miR-208 (vert. 2), miR-499 (vert. 1) | miR-208b, miR-499 | Reduced proportion of slow myofibers in skeletal muscle (van Rooij et al., 2009) |
| miR-15/16/195/322/497 (vert. 7), miR-503 (euth. 1) | miR-322 ~ 503 | Defects in mammary gland involution after weaning; increased body weight due to more white fat (Llobet-Navas et al., 2014); increased tumorigensis of breast cancer in sensitized background (Rodriguez-Barrueco et al., 2017); increased angiogenic response to inflammation (Lee et al., 2017) |
| miR-15/16/195/322/497 (vert. 7), miR-503 (euth. 1), miR-351 (p.c. 1) | miR-322 ~ 503 ~ 351 | Deletion in cartilage causes perinatal lethality, partial penetrance, due to tracheal defects (observed specifically in pure C57BL/6N genetic background) (Bluhm et al., 2017) |
| miR-431 (euth. 1), miR-433 (euth. 1), miR-127 (euth. 1), miR-434 (p.c. 1) | miR-431 ~ 433 ~ 127 ~ 434 | Note that the following phenotypes are observed only when inheriting deletion from maternal allele (part of paternally imprinted cluster expressed from only maternal allele). Either neonatal lethality or slower growth after weaning, depending on genetic background; enlarged placenta with defects (Sekita et al., 2008) |
| Cluster of 36 genes from 35 families; all but two of the 35 families have only a single murine gene (exceptions, miR-381/539 and miR-329/362); 27 families conserved among eutheria, the other 8 conserved less broadly | miR-379 ~ 411 ~ 299 ~ 380 ~ 1197 ~ 323 ~ 758 ~ 329 ~ 494 ~ 679 ~ 1193 ~ 666 ~ 543 ~ 495 ~ 667 ~ 376c ~ 376b ~ 376a ~ 300 ~ 381 ~ 487b ~ 539 ~ 544 ~ 382 ~ 134 ~ 668 ~ 485 ~ 154 ~ 496a ~ 377 ~ 541 ~ 409 ~ 412 ~ 369 ~ 410 ~ 3072 | Note that the following phenotypes are observed only when inheriting deletion from maternal allele (paternally imprinted cluster expressed from only maternal allele). Neonatal lethality, partial penetrance; transient postnatal growth retardation; postnatal metabolic defects, including inefficient mobilization of glycogen stores (Labialle et al., 2014); increased anxiety-related behavior in adult mice (Marty et al., 2016) |
Why might broadly conserved miRNAs be less consequential in nematodes than in either flies or mammals? Perhaps much of nematode development is too rapid for optimal miRNA regulation. Mammalian miRNAs often take at least a day to reach the levels needed for consequential regulation, but C. elegans larvae hatch, with many cells already terminally differentiated, only 14 hours after fertilization. Regardless of the reason for this reduced role, the more frequent loss in nematodes of otherwise broadly conserved miRNA families supports the idea that in nematodes other regulatory processes have exapted ancestral miRNA functions. For example, the C. elegans lineage has lost eight of the 31 miRNA families that non-nematode lineages have inherited from the bilaterian ancestor (the miR-7, -33, -96, -153, -184, -210, -219, -278 families), and as many as 15 of the 31 have been lost in the nematode lineage that gave rise to Globodera pallida (Fromm et al., 2015; Sarkies et al., 2015). In contrast, the Drosophila lineage has lost only two of the 31 (miR-153 and miR-10, with retention of alternative miR-10 isoforms), and the mammalian lineage has lost only four (miR-76, -252, -278, and -281). The seemingly reduced role for miRNAs in nematodes makes the original discovery of the lin-4 RNA all the more impressive.
Although abnormal knockout phenotypes have revealed functions for most of the mammalian miRNA families conserved to fish, less is known of the biological functions of more recently emerged mammalian miRNAs. In humans, 96 miRNA genes from 88 families are conserved among placental mammals but not to fish (Table S1). Genes for a third of these families fall within two paternally imprinted miRNA clusters, which when disrupted in mouse yield phenotypes typical of imprinted genes, including defective placental development, viability/growth defects, and anxiety-related behavior (Sekita et al., 2008; Labialle et al., 2014; Marty et al., 2016) (Table 3). Among the others, six have been individually disrupted (each the sole member of its family), and four have abnormal phenotypes (miR-188, miR-378, miR-486, and miR-505) (Carrer et al., 2012; Li et al., 2015; Yang et al., 2017; Jee et al., 2018), whereas two are reported to have no observable phenotype (miR-296 and miR-335) (Park et al., 2012; Hiramuki et al., 2015).
For the hundreds of other human miRNAs not conserved to mouse, confidently inferring function without the possibility of a mouse model is difficult. Indeed, even for the 88 pan-mammalian miRNA families, mouse models might be of more limited utility. These pan-mammalian miRNAs each have an average of only ~11 targets conserved above background, which is far less than the average of > 400 found for each of the more broadly conserved miRNAs (Friedman et al., 2009). Therefore, any abnormal phenotypes observed for knockouts of these miRNAs in mouse are more likely to be attributable to non-conserved targeting and thus less likely to report on their functions in humans.
One difference between flies and mice is that in flies abnormal phenotypes are usually apparent even when only one member of a conserved family is disrupted (Chen et al., 2014), whereas in mice redundancy among family members often requires disruption of multiple family members before phenotypes become apparent. Thus, when considering some of the broadly conserved families for which only relatively subtle abnormalities have been reported, such as let-7 and miR-10 (Table 1), more severe abnormalities are expected when additional family members are simultaneously disrupted. In both flies and mice, animals heterozygous for a disrupted miRNA gene typically appear normal—the most notable exceptions being for mutations at the miR-96 and miR-17~92 loci, which each cause haploinsufficient abnormalities in both mice and humans, with loss of one copy of miR-96 causing deafness (Lewis et al., 2009; Mencia et al., 2009), and loss of one copy of the miR-17~92 miRNA cluster causing skeletal anomalies, growth defects, and learning disabilities (de Pontual et al., 2011; Fiori et al., 2015; Han et al., 2015).
Although the impact or full impact of many conserved miRNA families has yet to be explored, the results to date already implicate mammalian miRNAs in diverse developmental, cellular, and physiological processes. As mentioned in the opening paragraph of this review, many miRNAs are required for proper development, including that of the skeleton, teeth, brain, eyes, neurons, muscle, heart, lungs, kidneys, vasculature, liver, pancreas, intestine, skin, fat, breast, ovaries, testes, placenta, thymus, and each hematopoietic lineage (Tables 1, 2, and 3). In addition, many influence cellular phenomena and functions, including axon sprouting, synapse formation and function, mitotic spindle orientation, polyploidization, ciliogenesis, and diverse functions in various hematopoietic lineages (Tables 1, 2, and 3). Moreover, many miRNAs influence physiological processes, including cardiac conduction, blood pressure, lipid or cholesterol metabolism, insulin production, pituitary function, mobilization of glycogen, Aβ peptide degradation, bone resorption, fibrosis, and the overall growth of embryos or pups (Tables 1, 2, and 3). In addition, many miRNA knockout strains have differential responses to models of neuronal, cardiac, muscular, pulmonary, vascular, endocrinological, hepatic, intestinal, renal, immunological, or metabolic diseases or injuries, and some have altered susceptibility to fungal or bacterial infections, or altered propensity to develop tumors in cancer models (Tables 1, 2, and 3).
Sculptors of the Transcriptome
The reason that metazoan miRNAs are so important for mammals, flies, and presumably most other animals, is that they add a dense layer of posttranscriptional gene control that complements and extends the regulation occurring at other levels of the gene-expression program. The rates of mRNA transcription, processing, and transport determine the identities and amounts of mRNAs that enter the cytoplasm. Once in the cytoplasm, the mRNAs encounter the miRNA milieu of the cell and undergo different shades of repression, depending on the identities and activities of the miRNAs in the cell, the number of sites to these co-expressed miRNAs, and the efficacies of these sites. For each site, the repression is typically quite modest—usually less than 50%, and often less than 20% (Baek et al., 2008; Selbach et al., 2008). However, multiple sites in the same UTR can add up to much more substantial repression, as each additional site tends to act additively, and some sites, particularly those within about 8–40 nt from each other, act cooperatively (Grimson et al., 2007; Saetrom et al., 2007). Because the miRNA milieu differs between cell types and over the course of development, the animal can achieve complex gene-expression patterns with relatively simple promoters for both the mRNAs and the miRNAs.
In this view, the metazoan miRNAs can be thought of as the sculptors of the transcriptome. Transcription and other nuclear events set up a column of gene expression, and then the miRNAs, like a stone sculptor, chip away at this column. Occasionally they chip away enough to either trigger or sharpen a developmental transition, but more generally they produce a much more complex topology of gene expression, with more optimal levels of many proteins in each cell of each tissue.
As participants in many selectively conserved regulatory interactions (e.g., >40,000 in mammals) (Friedman et al., 2009), miRNAs have been extensively integrated within gene-regulatory networks—of potential utility in any regulatory architecture with a repressive component. With this modular, combinatorial regulatory pathway at its disposal, evolution can more easily generate complex, highly connected regulatory networks required to robustly cope with environmental, genetic, or stochastic contingencies (Hornstein and Shomron, 2006). Indeed, as might be expected for transcription factors or any other component of these networks, deletion of miRNAs can reduce tolerance to environmental challenges, genetic perturbations, injury, or disease (Ebert and Sharp, 2012; Mendell and Olson, 2012; Cassidy et al., 2016; Kasper et al., 2017) (Tables 1, 2, and 3).
The regulatory and developmental robustness conferred by miRNAs stems from their integration within larger regulatory networks and not a supernatural ability to preferentially repress mRNAs that are stochastically too abundant in a cell. Nonetheless, as with other post-transcriptional processes that reduce the protein output from each mRNA, miRNA-mediated repression, when coupled with a compensatory increase in transcription, can in principle decrease the cell-to-cell variability (i.e., the noise) of gene expression (Bartel and Chen, 2004). On the other hand, adding a regulatory component, such as a miRNA, to a system also adds some intrinsic noise, due to variable expression of that component (Schmiedel et al., 2015). Thus, depending upon whether the decreased noise imparted by increased transcription can overcome the increased noise imparted by adding the miRNA component, the net effect of adding the miRNA component varies, with lowly expressed genes tending to undergo a net decrease in noise and highly expressed genes tending to undergo a net increase (Schmiedel et al., 2015).
Functions of Individual Interactions
The analyses of miRNA knockouts, which have greatly expanded the known functions of mammalian miRNAs (Tables 1, 2, and 3), often investigate the effects of the miRNAs on predicted targets and sometimes implicate the de-repression of just one or two targets as the primary cause of an abnormal phenotype. The most definitive approach for isolating and confirming the importance of a particular target is to disrupt the miRNA-binding site(s) within the endogenous target gene and examine the extent to which the resulting de-repression of that target phenocopies the miRNA knockout (Dorsett et al., 2008; Teng et al., 2008; Ecsedi et al., 2015; Lu et al., 2015a; Drexel et al., 2016; McJunkin and Ambros, 2017; Mildner et al., 2017). With the development of more convenient genome-editing methods, this approach is gaining popularity and yielding insights that would be difficult to obtain by previous approaches, showing, for instance, that a single miRNA target site can be essential for viability (McJunkin and Ambros, 2017).
As the compendium of phenotypic effects of individual target interactions grows, so will a more robust molecular understanding of miRNA biological functions. However, at least for the foreseeable future, these experiments can query only a small sampling of the biological targets of each miRNA. Thus, the focus will presumably remain on targets considered most likely to help explain the miRNA-knockout phenotype, as judged using site conservation, repression magnitude, and known or suspected functions of the targets.
In addition to its low throughput, the approach of disrupting individual interactions faces other limitations. A major challenge is to design phenotypic analyses with the sensitivity needed to uncover the functional roles of many of these individual interactions. This challenge stems from the modest repression conferred by most interactions, the potential buffering effect of interactions occurring within complex regulatory networks with bifurcating pathways and feedback control that enable robust response despite a defective node in the network, and the permissiveness of standard lab conditions, which have been developed to preserve and propagate mutant lines rather than to simulate life in the wild (Bartel, 2009). In contrast, comparative sequence analysis, the approach that reveals the large scope of biological targets, discerns the results of the big experiment (i.e., biological evolution) that has been operating over many millions of years at the level of populations evolving in the wild. Because a very subtle fitness disadvantage—even one contingent on an intermittent environmental or genetic stress—can prevent alleles with mutated regulatory sites from becoming fixed in a population, purifying selection is an exquisitely sensitive indicator of biological function. Although comparable sensitivity will never be achieved in the lab, the innovative strategies that have revealed additional phenotypes for miRNA knockouts in the contexts of environmental/genetic stress or injury/disease models (Mendell and Olson, 2012; Cassidy et al., 2016; Kasper et al., 2017) (Tables 1, 2, and 3) should also be productive when examining the effects of disrupting individual targeting interactions.
Concluding Remarks
After the seminal discovery of the lin-4 miRNA 25 years ago, the next big surprise was the abundance of this class of regulatory RNAs in animals, a finding that was soon followed by the surprising abundance of their regulatory targets. The more recent surprises have included the molecular consequences of miRNA-mediated repression in post-embryonic cells (i.e., the dominance of mRNA destabilization as compared to translational repression), as well as preponderance of fly and mouse miRNA families with abnormal knockout phenotypes. The next surprise? Time will tell, but based on their past record, miRNAs will not disappoint.
Supplement
Acknowledgments
I thank current and former members of the lab as wells as many other colleagues for stimulating discussions over the years, and I apologize to many whose work I did not adequately describe or cite due to space constraints. I am an HHMI investigator and our work on miRNAs is supported by NIGMS of the National Institutes of Health.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Agarwal A, Subtelny AO, Thiru P, Ulitsky I, Bartel DP. Predicting microRNA targeting efficacy in Drosophila. bioRxiv. 2017:198689. doi: 10.1186/s13059-018-1504-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Agarwal V, Bell GW, Nam JW, Bartel DP. Predicting effective microRNA target sites in mammalian mRNAs. eLife. 2015;4:e05005. doi: 10.7554/eLife.05005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Agudo J, Ruzo A, Tung N, Salmon H, Leboeuf M, Hashimoto D, Becker C, Garrett-Sinha LA, Baccarini A, Merad M, et al. The miR-126-VEGFR2 axis controls the innate response to pathogen-associated nucleic acids. Nature immunology. 2014;15:54–62. doi: 10.1038/ni.2767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahmed K, LaPierre MP, Gasser E, Denzler R, Yang Y, Rulicke T, Kero J, Latreille M, Stoffel M. Loss of microRNA-7a2 induces hypogonadotropic hypogonadism and infertility. The Journal of clinical investigation. 2017;127:1061–1074. doi: 10.1172/JCI90031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alvarez-Saavedra E, Horvitz HR. Many families of C. elegans microRNAs are not essential for development or viability. Curr Biol. 2010;20:367–373. doi: 10.1016/j.cub.2009.12.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ambros V, Bartel B, Bartel DP, Burge CB, Carrington JC, Chen X, Dreyfuss G, Eddy S, Griffiths-Jones S, Matzke M, et al. A uniform system for microRNA annotation. RNA. 2003;9:277–279. doi: 10.1261/rna.2183803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ameres SL, Horwich MD, Hung JH, Xu J, Ghildiyal M, Weng Z, Zamore PD. Target RNA-directed trimming and tailing of small silencing RNAs. Science. 2010;328:1534–1539. doi: 10.1126/science.1187058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amin ND, Bai G, Klug JR, Bonanomi D, Pankratz MT, Gifford WD, Hinckley CA, Sternfeld MJ, Driscoll SP, Dominguez B, et al. Loss of motoneuron-specific microRNA-218 causes systemic neuromuscular failure. Science. 2015;350:1525–1529. doi: 10.1126/science.aad2509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andersson MG, Haasnoot PC, Xu N, Berenjian S, Berkhout B, Akusjarvi G. Suppression of RNA interference by adenovirus virus-associated RNA. Journal of virology. 2005;79:9556–9565. doi: 10.1128/JVI.79.15.9556-9565.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andolina D, Di Segni M, Bisicchia E, D’Alessandro F, Cestari V, Ventura A, Concepcion C, Puglisi-Allegra S, Ventura R. Effects of lack of microRNA-34 on the neural circuitry underlying the stress response and anxiety. Neuropharmacology. 2016;107:305–316. doi: 10.1016/j.neuropharm.2016.03.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aurora AB, Mahmoud AI, Luo X, Johnson BA, van Rooij E, Matsuzaki S, Humphries KM, Hill JA, Bassel-Duby R, Sadek HA, et al. MicroRNA-214 protects the mouse heart from ischemic injury by controlling Ca(2)(+) overload and cell death. The Journal of clinical investigation. 2012;122:1222–1232. doi: 10.1172/JCI59327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Auyeung VC, Ulitsky I, McGeary SE, Bartel DP. Beyond secondary structure: primary-sequence determinants license pri-miRNA hairpins for processing. Cell. 2013;152:844–858. doi: 10.1016/j.cell.2013.01.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avesson L, Reimegard J, Wagner EG, Soderbom F. MicroRNAs in Amoebozoa: deep sequencing of the small RNA population in the social amoeba Dictyostelium discoideum reveals developmentally regulated microRNAs. RNA. 2012;18:1771–1782. doi: 10.1261/rna.033175.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azevedo-Pouly AC, Sutaria DS, Jiang J, Elgamal OA, Amari F, Allard D, Grippo PJ, Coppola V, Schmittgen TD. miR-216 and miR-217 expression is reduced in transgenic mouse models of pancreatic adenocarcinoma, knockout of miR-216/miR-217 host gene is embryonic lethal. Functional & integrative genomics. 2017;17:203–212. doi: 10.1007/s10142-016-0512-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Babiarz JE, Ruby JG, Wang Y, Bartel DP, Blelloch R. Mouse ES cells express endogenous shRNAs, siRNAs, and other Microprocessor-independent, Dicer-dependent small RNAs. Genes Dev. 2008;22:2773–2785. doi: 10.1101/gad.1705308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baek D, Villen J, Shin C, Camargo FD, Gygi SP, Bartel DP. The impact of microRNAs on protein output. Nature. 2008;455:64–71. doi: 10.1038/nature07242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bail S, Swerdel M, Liu H, Jiao X, Goff LA, Hart RP, Kiledjian M. Differential regulation of microRNA stability. RNA. 2010;16:1032–1039. doi: 10.1261/rna.1851510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ballarino M, Pagano F, Girardi E, Morlando M, Cacchiarelli D, Marchioni M, Proudfoot NJ, Bozzoni I. Coupled RNA processing and transcription of intergenic primary microRNAs. Molecular and cellular biology. 2009;29:5632–5638. doi: 10.1128/MCB.00664-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barbato S, Marrocco E, Intartaglia D, Pizzo M, Asteriti S, Naso F, Falanga D, Bhat RS, Meola N, Carissimo A, et al. MiR-211 is essential for adult cone photoreceptor maintenance and visual function. Scientific reports. 2017;7:17004. doi: 10.1038/s41598-017-17331-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004;116:281–297. doi: 10.1016/s0092-8674(04)00045-5. [DOI] [PubMed] [Google Scholar]
- Bartel DP. MicroRNAs: target recognition and regulatory functions. Cell. 2009;136:215–233. doi: 10.1016/j.cell.2009.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bartel DP, Chen CZ. Micromanagers of gene expression: the potentially widespread influence of metazoan microRNAs. Nat Rev Genet. 2004;5:396–400. doi: 10.1038/nrg1328. [DOI] [PubMed] [Google Scholar]
- Barth S, Pfuhl T, Mamiani A, Ehses C, Roemer K, Kremmer E, Jaker C, Hock J, Meister G, Grasser FA. Epstein-Barr virus-encoded microRNA miR-BART2 down-regulates the viral DNA polymerase BALF5. Nucleic Acids Res. 2008;36:666–675. doi: 10.1093/nar/gkm1080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baskerville S, Bartel DP. Microarray profiling of microRNAs reveals frequent coexpression with neighboring miRNAs and host genes. RNA. 2005;11:241–247. doi: 10.1261/rna.7240905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bazzini AA, Lee MT, Giraldez AJ. Ribosome profiling shows that miR-430 reduces translation before causing mRNA decay in zebrafish. Science. 2012;336:233–237. doi: 10.1126/science.1215704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bellutti F, Kauer M, Kneidinger D, Lion T, Klein R. Identification of RISC-associated adenoviral microRNAs, a subset of their direct targets, and global changes in the targetome upon lytic adenovirus 5 infection. Journal of virology. 2015;89:1608–1627. doi: 10.1128/JVI.02336-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bender W. MicroRNAs in the Drosophila bithorax complex. Genes Dev. 2008;22:14–19. doi: 10.1101/gad.1614208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bernstein E, Caudy AA, Hammond SM, Hannon GJ. Role for a bidentate ribonuclease in the initiation step of RNA interference. Nature. 2001;409:363–366. doi: 10.1038/35053110. [DOI] [PubMed] [Google Scholar]
- Bethune J, Artus-Revel CG, Filipowicz W. Kinetic analysis reveals successive steps leading to miRNA-mediated silencing in mammalian cells. EMBO reports. 2012;13:716–723. doi: 10.1038/embor.2012.82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bezman NA, Chakraborty T, Bender T, Lanier LL. miR-150 regulates the development of NK and iNKT cells. The Journal of experimental medicine. 2011;208:2717–2731. doi: 10.1084/jem.20111386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bitetti A. MicroRNA degradation by a conserved target RNA regulates animal behavior. Nat Struct Mol Biol. 2018;25:244–251. doi: 10.1038/s41594-018-0032-x. [DOI] [PubMed] [Google Scholar]
- Bluhm B, Ehlen HWA, Holzer T, Georgieva VS, Heilig J, Pitzler L, Etich J, Bortecen T, Frie C, Probst K, et al. miR-322 stabilizes MEK1 expression to inhibit RAF/MEK/ERK pathway activation in cartilage. Development. 2017;144:3562–3577. doi: 10.1242/dev.148429. [DOI] [PubMed] [Google Scholar]
- Bobbin ML, Rossi JJ. RNA Interference (RNAi)-Based Therapeutics: Delivering on the Promise? Annual review of pharmacology and toxicology. 2016;56:103–122. doi: 10.1146/annurev-pharmtox-010715-103633. [DOI] [PubMed] [Google Scholar]
- Bogerd HP, Karnowski HW, Cai X, Shin J, Pohlers M, Cullen BR. A mammalian herpesvirus uses noncanonical expression and processing mechanisms to generate viral microRNAs. Mol Cell. 2010;37:135–142. doi: 10.1016/j.molcel.2009.12.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bohnsack MT, Czaplinski K, Gorlich D. Exportin 5 is a RanGTP-dependent dsRNA-binding protein that mediates nuclear export of pre-miRNAs. RNA. 2004;10:185–191. doi: 10.1261/rna.5167604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boldin MP, Taganov KD, Rao DS, Yang L, Zhao JL, Kalwani M, Garcia-Flores Y, Luong M, Devrekanli A, Xu J, et al. miR-146a is a significant brake on autoimmunity, myeloproliferation, and cancer in mice. The Journal of experimental medicine. 2011;208:1189–1201. doi: 10.1084/jem.20101823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bortolamiol-Becet D, Hu F, Jee D, Wen J, Okamura K, Lin CJ, Ameres SL, Lai EC. Selective suppression of the splicing-mediated microRNA pathway by the terminal uridyltransferase tailor. Mol Cell. 2015;59:217–228. doi: 10.1016/j.molcel.2015.05.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bosson AD, Zamudio JR, Sharp PA. Endogenous miRNA and target concentrations determine susceptibility to potential ceRNA competition. Mol Cell. 2014;56:347–359. doi: 10.1016/j.molcel.2014.09.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boulias K, Horvitz HR. The C. elegans microRNA mir-71 acts in neurons to promote germline-mediated longevity through regulation of DAF-16/FOXO. Cell metabolism. 2012;15:439–450. doi: 10.1016/j.cmet.2012.02.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brennecke J, Stark A, Russell RB, Cohen SM. Principles of microRNA-target recognition. PLoS Biol. 2005;3:e85. doi: 10.1371/journal.pbio.0030085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brenner JL, Jasiewicz KL, Fahley AF, Kemp BJ, Abbott AL. Loss of individual microRNAs causes mutant phenotypes in sensitized genetic backgrounds in C. elegans. Curr Biol. 2010;20:1321–1325. doi: 10.1016/j.cub.2010.05.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burke SL, Hammell M, Ambros V. Robust distal tip cell pathfinding in the face of temperature stress is ensured by two conserved microRNAs in Caenorhabditis elegans. Genetics. 2015;200:1201–1218. doi: 10.1534/genetics.115.179184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cai X, Hagedorn CH, Cullen BR. Human microRNAs are processed from capped, polyadenylated transcripts that can also function as mRNAs. RNA. 2004;10:1957–1966. doi: 10.1261/rna.7135204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Callis TE, Pandya K, Seok HY, Tang RH, Tatsuguchi M, Huang ZP, Chen JF, Deng Z, Gunn B, Shumate J, et al. MicroRNA-208a is a regulator of cardiac hypertrophy and conduction in mice. The Journal of clinical investigation. 2009;119:2772–2786. doi: 10.1172/JCI36154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao H, Jheon A, Li X, Sun Z, Wang J, Florez S, Zhang Z, McManus MT, Klein OD, Amendt BA. The Pitx2:miR-200c/141:noggin pathway regulates Bmp signaling and ameloblast differentiation. Development. 2013;140:3348–3359. doi: 10.1242/dev.089193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carrer M, Liu N, Grueter CE, Williams AH, Frisard MI, Hulver MW, Bassel-Duby R, Olson EN. Control of mitochondrial metabolism and systemic energy homeostasis by microRNAs 378 and 378*. Proc Natl Acad Sci U S A. 2012;109:15330–15335. doi: 10.1073/pnas.1207605109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cassidy JJ, Straughan AJ, Carthew RW. Differential masking of natural genetic variation by miR-9a in Drosophila. Genetics. 2016;202:675–687. doi: 10.1534/genetics.115.183822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cazalla D, Xie M, Steitz JA. A primate herpesvirus uses the integrator complex to generate viral microRNAs. Mol Cell. 2011;43:982–992. doi: 10.1016/j.molcel.2011.07.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cazalla D, Yario T, Steitz JA. Down-regulation of a host microRNA by a Herpesvirus saimiri noncoding RNA. Science. 2010;328:1563–1566. doi: 10.1126/science.1187197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chandradoss SD, Schirle NT, Szczepaniak M, MacRae IJ, Joo C. A dynamic search process underlies microRNA targeting. Cell. 2015;162:96–107. doi: 10.1016/j.cell.2015.06.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang HM, Triboulet R, Thornton JE, Gregory RI. A role for the Perlman syndrome exonuclease Dis3l2 in the Lin28-let-7 pathway. Nature. 2013;497:244–248. doi: 10.1038/nature12119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chapnik E, Rivkin N, Mildner A, Beck G, Pasvolsky R, Metzl-Raz E, Birger Y, Amir G, Tirosh I, Porat Z, et al. miR-142 orchestrates a network of actin cytoskeleton regulators during megakaryopoiesis. eLife. 2014;3:e01964. doi: 10.7554/eLife.01964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheloufi S, Dos Santos CO, Chong MM, Hannon GJ. A dicer-independent miRNA biogenesis pathway that requires Ago catalysis. Nature. 2010;465:584–589. doi: 10.1038/nature09092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen CY, Shyu AB. Mechanisms of deadenylation-dependent decay. Wiley interdisciplinary reviews RNA. 2011;2:167–183. doi: 10.1002/wrna.40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen X, Ling Y, Wei Y, Tang J, Ren Y, Zhang B, Jiang F, Li H, Wang R, Wen W, et al. FASEB journal : official publication of the Federation of American Societies for Experimental Biology. 2018. Dual regulation of HMGB1 by combined JNK1/2-ATF2 axis with miR-200 family in nonalcoholic steatohepatitis in mice. [DOI] [PubMed] [Google Scholar]
- Chen YW, Song S, Weng R, Verma P, Kugler JM, Buescher M, Rouam S, Cohen SM. Systematic study of Drosophila microRNA functions using a collection of targeted knockout mutations. Dev Cell. 2014;31:784–800. doi: 10.1016/j.devcel.2014.11.029. [DOI] [PubMed] [Google Scholar]
- Cheng L, Zhu Y, Han H, Zhang Q, Cui K, Shen H, Zhang J, Yan J, Prochownik E, Li Y. MicroRNA-148a deficiency promotes hepatic lipid metabolism and hepatocarcinogenesis in mice. Cell death & disease. 2017;8:e2916. doi: 10.1038/cddis.2017.309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chi SW, Zang JB, Mele A, Darnell RB. Argonaute HITS-CLIP decodes microRNA-mRNA interaction maps. Nature. 2009;460:479–486. doi: 10.1038/nature08170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chiang DY, Kongchan N, Beavers DL, Alsina KM, Voigt N, Neilson JR, Jakob H, Martin JF, Dobrev D, Wehrens XH, et al. Loss of microRNA-106b-25 cluster promotes atrial fibrillation by enhancing ryanodine receptor type-2 expression and calcium release. Circulation Arrhythmia and electrophysiology. 2014;7:1214–1222. doi: 10.1161/CIRCEP.114.001973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chiang HR, Schoenfeld LW, Ruby JG, Auyeung VC, Spies N, Baek D, Johnston WK, Russ C, Luo S, Babiarz JE, et al. Mammalian microRNAs: experimental evaluation of novel and previously annotated genes. Genes Dev. 2010;24:992–1009. doi: 10.1101/gad.1884710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choi SW, Lee SU, Kim EH, Park SJ, Choi I, Kim TD, Kim SH. Osteoporotic bone of miR-150-deficient mice: Possibly due to low serum OPG-mediated osteoclast activation. Bone reports. 2015;3:5–10. doi: 10.1016/j.bonr.2015.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chowdhary V, Teng KY, Thakral S, Zhang B, Lin CH, Wani N, Bruschweiler-Li L, Zhang X, James L, Yang D, et al. miRNA-122 protects mice and human hepatocytes from acetaminophen toxicity by regulating cytochrome P450 family 1 subfamily A member 2 and family 2 subfamily E member 1 expression. The American journal of pathology. 2017;187:2758–2774. doi: 10.1016/j.ajpath.2017.08.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chu CY, Rana TM. Translation repression in human cells by microRNA-induced gene silencing requires RCK/p54. PLoS Biol. 2006;4:e210. doi: 10.1371/journal.pbio.0040210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chun S, Du F, Westmoreland JJ, Han SB, Wang YD, Eddins D, Bayazitov IT, Devaraju P, Yu J, Mellado Lagarde MM, et al. Thalamic miR-338-3p mediates auditory thalamocortical disruption and its late onset in models of 22q11.2 microdeletion. Nature medicine. 2017;23:39–48. doi: 10.1038/nm.4240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cifuentes D, Xue H, Taylor DW, Patnode H, Mishima Y, Cheloufi S, Ma E, Mane S, Hannon GJ, Lawson ND, et al. A novel miRNA processing pathway independent of Dicer requires Argonaute2 catalytic activity. Science. 2010;328:1694–1698. doi: 10.1126/science.1190809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cioffi M, Vallespinos-Serrano M, Trabulo SM, Fernandez-Marcos PJ, Firment AN, Vazquez BN, Vieira CR, Mulero F, Camara JA, Cronin UP, et al. MiR-93 controls adiposity via inhibition of Sirt7 and Tbx3. Cell reports. 2015;12:1594–1605. doi: 10.1016/j.celrep.2015.08.006. [DOI] [PubMed] [Google Scholar]
- Cock JM, Sterck L, Rouze P, Scornet D, Allen AE, Amoutzias G, Anthouard V, Artiguenave F, Aury JM, Badger JH, et al. The Ectocarpus genome and the independent evolution of multicellularity in brown algae. Nature. 2010;465:617–621. doi: 10.1038/nature09016. [DOI] [PubMed] [Google Scholar]
- Crowley JJ, Collins AL, Lee RJ, Nonneman RJ, Farrell MS, Ancalade N, Mugford JW, Agster KL, Nikolova VD, Moy SS, et al. Disruption of the microRNA 137 primary transcript results in early embryonic lethality in mice. Biological psychiatry. 2015;77:e5–7. doi: 10.1016/j.biopsych.2014.05.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis E, Caiment F, Tordoir X, Cavaille J, Ferguson-Smith A, Cockett N, Georges M, Charlier C. RNAi-mediated allelic trans-interaction at the imprinted Rtl1/Peg11 locus. Curr Biol. 2005;15:743–749. doi: 10.1016/j.cub.2005.02.060. [DOI] [PubMed] [Google Scholar]
- de la Mata M, Gaidatzis D, Vitanescu M, Stadler MB, Wentzel C, Scheiffele P, Filipowicz W, Grosshans H. Potent degradation of neuronal miRNAs induced by highly complementary targets. EMBO reports. 2015;16:500–511. doi: 10.15252/embr.201540078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Lencastre A, Pincus Z, Zhou K, Kato M, Lee SS, Slack FJ. MicroRNAs both promote and antagonize longevity in C. elegans. Curr Biol. 2010;20:2159–2168. doi: 10.1016/j.cub.2010.11.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Pontual L, Yao E, Callier P, Faivre L, Drouin V, Cariou S, Van Haeringen A, Genevieve D, Goldenberg A, Oufadem M, et al. Germline deletion of the miR-17 ~ 92 cluster causes skeletal and growth defects in humans. Nat Genet. 2011;43:1026–1030. doi: 10.1038/ng.915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Denzler R, Agarwal V, Stefano J, Bartel DP, Stoffel M. Assessing the ceRNA hypothesis with quantitative measurements of miRNA and target abundance. Mol Cell. 2014;54:766–776. doi: 10.1016/j.molcel.2014.03.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Denzler R, McGeary SE, Title AC, Agarwal V, Bartel DP, Stoffel M. Impact of microRNA levels, target-site complementarity, and cooperativity on competing endogenous RNA-regulated gene expression. Mol Cell. 2016;64:565–579. doi: 10.1016/j.molcel.2016.09.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deshpande SD, Putta S, Wang M, Lai JY, Bitzer M, Nelson RG, Lanting LL, Kato M, Natarajan R. Transforming growth factor-beta-induced cross talk between p53 and a microRNA in the pathogenesis of diabetic nephropathy. Diabetes. 2013;62:3151–3162. doi: 10.2337/db13-0305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diebel KW, Smith AL, van Dyk LF. Mature and functional viral miRNAs transcribed from novel RNA polymerase III promoters. RNA. 2010;16:170–185. doi: 10.1261/rna.1873910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ding L, Spencer A, Morita K, Han M. The developmental timing regulator AIN-1 interacts with miRISCs and may target the argonaute protein ALG-1 to cytoplasmic P bodies in C. elegans. Mol Cell. 2005;19:437–447. doi: 10.1016/j.molcel.2005.07.013. [DOI] [PubMed] [Google Scholar]
- Ding L, Zhang Y, Han L, Fu L, Mei X, Wang J, Itkow J, Elabid AEI, Pang L, Yu D. Activating and sustaining c-Myc by depletion of miR-144/451 gene locus contributes to B-lymphomagenesis. Oncogene. 2017 doi: 10.1038/s41388-017-0055-5. [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Djuranovic S, Nahvi A, Green R. miRNA-mediated gene silencing by translational repression followed by mRNA deadenylation and decay. Science. 2012;336:237–240. doi: 10.1126/science.1215691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dooley J, Garcia-Perez JE, Sreenivasan J, Schlenner SM, Vangoitsenhoven R, Papadopoulou AS, Tian L, Schonefeldt S, Serneels L, Deroose C, et al. The microRNA-29 family dictates the balance between homeostatic and pathological glucose handling in diabetes and obesity. Diabetes. 2016;65:53–61. doi: 10.2337/db15-0770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dorsett Y, McBride KM, Jankovic M, Gazumyan A, Thai TH, Robbiani DF, Di Virgilio M, San-Martin BR, Heidkamp G, Schwickert TA, et al. MicroRNA-155 suppresses activation-induced cytidine deaminase-mediated Myc-Igh translocation. Immunity. 2008;28:630–638. doi: 10.1016/j.immuni.2008.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drexel T, Mahofsky K, Latham R, Zimmer M, Cochella L. Neuron type-specific miRNA represses two broadly expressed genes to modulate an avoidance behavior in C. elegans. Genes Dev. 2016;30:2042–2047. doi: 10.1101/gad.287904.116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Du P, Wang L, Sliz P, Gregory RI. A biogenesis step upstream of Microprocessor controls miR-17~92 expression. Cell. 2015;162:885–899. doi: 10.1016/j.cell.2015.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dunand-Sauthier I, Santiago-Raber ML, Capponi L, Vejnar CE, Schaad O, Irla M, Seguin-Estevez Q, Descombes P, Zdobnov EM, Acha-Orbea H, et al. Silencing of c-Fos expression by microRNA-155 is critical for dendritic cell maturation and function. Blood. 2011;117:4490–4500. doi: 10.1182/blood-2010-09-308064. [DOI] [PubMed] [Google Scholar]
- Ebert MS, Sharp PA. Roles for microRNAs in conferring robustness to biological processes. Cell. 2012;149:515–524. doi: 10.1016/j.cell.2012.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ecsedi M, Rausch M, Grosshans H. The let-7 microRNA directs vulval development through a single target. Dev Cell. 2015;32:335–344. doi: 10.1016/j.devcel.2014.12.018. [DOI] [PubMed] [Google Scholar]
- Eichhorn SW, Guo H, McGeary SE, Rodriguez-Mias RA, Shin C, Baek D, Hsu SH, Ghoshal K, Villen J, Bartel DP. mRNA destabilization is the dominant effect of mammalian microRNAs by the time substantial repression ensues. Mol Cell. 2014;56:104–115. doi: 10.1016/j.molcel.2014.08.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elbashir SM, Harborth J, Lendeckel W, Yalcin A, Weber K, Tuschl T. Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature. 2001;411:494–498. doi: 10.1038/35078107. [DOI] [PubMed] [Google Scholar]
- Elia L, Quintavalle M, Zhang J, Contu R, Cossu L, Latronico MV, Peterson KL, Indolfi C, Catalucci D, Chen J, et al. The knockout of miR-143 and -145 alters smooth muscle cell maintenance and vascular homeostasis in mice: correlates with human disease. Cell death and differentiation. 2009;16:1590–1598. doi: 10.1038/cdd.2009.153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elkayam E, Kuhn CD, Tocilj A, Haase AD, Greene EM, Hannon GJ, Joshua-Tor L. The structure of human argonaute-2 in complex with miR-20a. Cell. 2012;150:100–110. doi: 10.1016/j.cell.2012.05.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ender C, Krek A, Friedlander MR, Beitzinger M, Weinmann L, Chen W, Pfeffer S, Rajewsky N, Meister G. A human snoRNA with microRNA-like functions. Mol Cell. 2008;32:519–528. doi: 10.1016/j.molcel.2008.10.017. [DOI] [PubMed] [Google Scholar]
- Fang W, Bartel DP. The menu of features that define primary microRNAs and enable de novo design of microRNA genes. Mol Cell. 2015;60:131–145. doi: 10.1016/j.molcel.2015.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farh KK, Grimson A, Jan C, Lewis BP, Johnston WK, Lim LP, Burge CB, Bartel DP. The widespread impact of mammalian microRNAs on mRNA repression and evolution. Science. 2005;310:1817–1821. doi: 10.1126/science.1121158. [DOI] [PubMed] [Google Scholar]
- Farmer DT, Finley JK, Chen FY, Tarifeno-Saldivia E, McNamara NA, Knox SM, McManus MT. miR-205 is a critical regulator of lacrimal gland development. Dev Biol. 2017;427:12–20. doi: 10.1016/j.ydbio.2017.05.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farmer DT, Shariat N, Park CY, Liu HJ, Mavropoulos A, McManus MT. Partially penetrant postnatal lethality of an epithelial specific microRNA in a mouse knockout. PLoS One. 2013;8:e76634. doi: 10.1371/journal.pone.0076634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fededa JP, Esk C, Mierzwa B, Stanyte R, Yuan S, Zheng H, Ebnet K, Yan W, Knoblich JA, Gerlich DW. MicroRNA-34/449 controls mitotic spindle orientation during mammalian cortex development. EMBO J. 2016;35:2386–2398. doi: 10.15252/embj.201694056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fiori E, Babicola L, Andolina D, Coassin A, Pascucci T, Patella L, Han YC, Ventura A, Ventura R. Neurobehavioral alterations in a genetic murine model of Feingold syndrome 2. Behavior genetics. 2015;45:547–559. doi: 10.1007/s10519-015-9724-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fragoso R, Mao T, Wang S, Schaffert S, Gong X, Yue S, Luong R, Min H, Yashiro-Ohtani Y, Davis M, et al. Modulating the strength and threshold of NOTCH oncogenic signals by mir-181a-1/b-1. PLoS Genet. 2012;8:e1002855. doi: 10.1371/journal.pgen.1002855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frank F, Sonenberg N, Nagar B. Structural basis for 5′-nucleotide base-specific recognition of guide RNA by human AGO2. Nature. 2010;465:818–822. doi: 10.1038/nature09039. [DOI] [PubMed] [Google Scholar]
- Friedlander MR, Chen W, Adamidi C, Maaskola J, Einspanier R, Knespel S, Rajewsky N. Discovering microRNAs from deep sequencing data using miRDeep. Nat Biotechnol. 2008;26:407–415. doi: 10.1038/nbt1394. [DOI] [PubMed] [Google Scholar]
- Friedman RC, Farh KK, Burge CB, Bartel DP. Most mammalian mRNAs are conserved targets of microRNAs. Genome Res. 2009;19:92–105. doi: 10.1101/gr.082701.108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fromm B, Billipp T, Peck LE, Johansen M, Tarver JE, King BL, Newcomb JM, Sempere LF, Flatmark K, Hovig E, et al. A uniform system for the annotation of vertebrate microRNA genes and the evolution of the human microRNAome. Annual review of genetics. 2015;49:213–242. doi: 10.1146/annurev-genet-120213-092023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gantier MP, McCoy CE, Rusinova I, Saulep D, Wang D, Xu D, Irving AT, Behlke MA, Hertzog PJ, Mackay F, et al. Analysis of microRNA turnover in mammalian cells following Dicer1 ablation. Nucleic Acids Res. 2011;39:5692–5703. doi: 10.1093/nar/gkr148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia DM, Baek D, Shin C, Bell GW, Grimson A, Bartel DP. Weak seed-pairing stability and high target-site abundance decrease the proficiency of lsy-6 and other microRNAs. Nat Struct Mol Biol. 2011;18:1139–1146. doi: 10.1038/nsmb.2115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giraldez AJ, Mishima Y, Rihel J, Grocock RJ, Van Dongen S, Inoue K, Enright AJ, Schier AF. Zebrafish miR-430 promotes deadenylation and clearance of maternal mRNAs. Science. 2006;312:75–79. doi: 10.1126/science.1122689. [DOI] [PubMed] [Google Scholar]
- Golden RJ, Chen B, Li T, Braun J, Manjunath H, Chen X, Wu J, Schmid V, Chang TC, Kopp F, et al. An Argonaute phosphorylation cycle promotes microRNA-mediated silencing. Nature. 2017;542:197–202. doi: 10.1038/nature21025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grimson A, Farh KK, Johnston WK, Garrett-Engele P, Lim LP, Bartel DP. MicroRNA targeting specificity in mammals: determinants beyond seed pairing. Mol Cell. 2007;27:91–105. doi: 10.1016/j.molcel.2007.06.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grimson A, Srivastava M, Fahey B, Woodcroft BJ, Chiang HR, King N, Degnan BM, Rokhsar DS, Bartel DP. Early origins and evolution of microRNAs and Piwi-interacting RNAs in animals. Nature. 2008;455:1193–1197. doi: 10.1038/nature07415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grishok A, Pasquinelli AE, Conte D, Li N, Parrish S, Ha I, Baillie DL, Fire A, Ruvkun G, Mello CC. Genes and mechanisms related to RNA interference regulate expression of the small temporal RNAs that control C. elegans developmental timing. Cell. 2001;106:23–34. doi: 10.1016/s0092-8674(01)00431-7. [DOI] [PubMed] [Google Scholar]
- Grosswendt S, Filipchyk A, Manzano M, Klironomos F, Schilling M, Herzog M, Gottwein E, Rajewsky N. Unambiguous identification of miRNA:target site interactions by different types of ligation reactions. Mol Cell. 2014;54:1042–1054. doi: 10.1016/j.molcel.2014.03.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gu S, Jin L, Zhang Y, Huang Y, Zhang F, Valdmanis PN, Kay MA. The loop position of shRNAs and pre-miRNAs is critical for the accuracy of dicer processing in vivo. Cell. 2012;151:900–911. doi: 10.1016/j.cell.2012.09.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo H, Ingolia NT, Weissman JS, Bartel DP. Mammalian microRNAs predominantly act to decrease target mRNA levels. Nature. 2010;466:835–840. doi: 10.1038/nature09267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gurha P, Abreu-Goodger C, Wang T, Ramirez MO, Drumond AL, van Dongen S, Chen Y, Bartonicek N, Enright AJ, Lee B, et al. Targeted deletion of microRNA-22 promotes stress-induced cardiac dilation and contractile dysfunction. Circulation. 2012;125:2751–2761. doi: 10.1161/CIRCULATIONAHA.111.044354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ha M, Kim VN. Regulation of microRNA biogenesis. Nature reviews Molecular cell biology. 2014;15:509–524. doi: 10.1038/nrm3838. [DOI] [PubMed] [Google Scholar]
- Hafner M, Landthaler M, Burger L, Khorshid M, Hausser J, Berninger P, Rothballer A, Ascano M, Jungkamp AC, Munschauer M, et al. Transcriptome-wide identification of RNA-binding protein and microRNA target sites by PAR-CLIP. Cell. 2010;141:129–141. doi: 10.1016/j.cell.2010.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hagan JP, Piskounova E, Gregory RI. Lin28 recruits the TUTase Zcchc11 to inhibit let-7 maturation in mouse embryonic stem cells. Nat Struct Mol Biol. 2009;16:1021–1025. doi: 10.1038/nsmb.1676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han J, Lee Y, Yeom KH, Nam JW, Heo I, Rhee JK, Sohn SY, Cho Y, Zhang BT, Kim VN. Molecular basis for the recognition of primary microRNAs by the Drosha-DGCR8 complex. Cell. 2006;125:887–901. doi: 10.1016/j.cell.2006.03.043. [DOI] [PubMed] [Google Scholar]
- Han J, Pedersen JS, Kwon SC, Belair CD, Kim YK, Yeom KH, Yang WY, Haussler D, Blelloch R, Kim VN. Posttranscriptional crossregulation between Drosha and DGCR8. Cell. 2009;136:75–84. doi: 10.1016/j.cell.2008.10.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han YC, Vidigal JA, Mu P, Yao E, Singh I, Gonzalez AJ, Concepcion CP, Bonetti C, Ogrodowski P, Carver B, et al. An allelic series of miR-17 ~ 92-mutant mice uncovers functional specialization and cooperation among members of a microRNA polycistron. Nat Genet. 2015;47:766–775. doi: 10.1038/ng.3321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hansen KF, Sakamoto K, Aten S, Snider KH, Loeser J, Hesse AM, Page CE, Pelz C, Arthur JS, Impey S, et al. Targeted deletion of miR-132/-212 impairs memory and alters the hippocampal transcriptome. Learning & memory. 2016;23:61–71. doi: 10.1101/lm.039578.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hansen TB, Jensen TI, Clausen BH, Bramsen JB, Finsen B, Damgaard CK, Kjems J. Natural RNA circles function as efficient microRNA sponges. Nature. 2013;495:384–388. doi: 10.1038/nature11993. [DOI] [PubMed] [Google Scholar]
- Hansen TB, Wiklund ED, Bramsen JB, Villadsen SB, Statham AL, Clark SJ, Kjems J. miRNA-dependent gene silencing involving Ago2-mediated cleavage of a circular antisense RNA. EMBO J. 2011;30:4414–4422. doi: 10.1038/emboj.2011.359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hasuwa H, Ueda J, Ikawa M, Okabe M. miR-200b and miR-429 function in mouse ovulation and are essential for female fertility. Science. 2013;341:71–73. doi: 10.1126/science.1237999. [DOI] [PubMed] [Google Scholar]
- Heidersbach A, Saxby C, Carver-Moore K, Huang Y, Ang YS, de Jong PJ, Ivey KN, Srivastava D. MicroRNA-1 regulates sarcomere formation and suppresses smooth muscle gene expression in the mammalian heart. eLife. 2013;2:e01323. doi: 10.7554/eLife.01323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Helwak A, Kudla G, Dudnakova T, Tollervey D. Mapping the human miRNA interactome by CLASH reveals frequent noncanonical binding. Cell. 2013;153:654–665. doi: 10.1016/j.cell.2013.03.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henao-Mejia J, Williams A, Goff LA, Staron M, Licona-Limon P, Kaech SM, Nakayama M, Rinn JL, Flavell RA. The microRNA miR-181 is a critical cellular metabolic rheostat essential for NKT cell ontogenesis and lymphocyte development and homeostasis. Immunity. 2013;38:984–997. doi: 10.1016/j.immuni.2013.02.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hendrickson DG, Hogan DJ, McCullough HL, Myers JW, Herschlag D, Ferrell JE, Brown PO. Concordant regulation of translation and mRNA abundance for hundreds of targets of a human microRNA. PLoS Biol. 2009;7:e1000238. doi: 10.1371/journal.pbio.1000238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henique C, Bollee G, Loyer X, Grahammer F, Dhaun N, Camus M, Vernerey J, Guyonnet L, Gaillard F, Lazareth H, et al. Genetic and pharmacological inhibition of microRNA-92a maintains podocyte cell cycle quiescence and limits crescentic glomerulonephritis. Nature communications. 2017;8:1829. doi: 10.1038/s41467-017-01885-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heo I, Ha M, Lim J, Yoon MJ, Park JE, Kwon SC, Chang H, Kim VN. Mono-uridylation of pre-microRNA as a key step in the biogenesis of group II let-7 microRNAs. Cell. 2012;151:521–532. doi: 10.1016/j.cell.2012.09.022. [DOI] [PubMed] [Google Scholar]
- Heo I, Joo C, Cho J, Ha M, Han J, Kim VN. Lin28 mediates the terminal uridylation of let-7 precursor microRNA. Mol Cell. 2008;32:276–284. doi: 10.1016/j.molcel.2008.09.014. [DOI] [PubMed] [Google Scholar]
- Heo I, Joo C, Kim YK, Ha M, Yoon MJ, Cho J, Yeom KH, Han J, Kim VN. TUT4 in concert with Lin28 suppresses microRNA biogenesis through pre-microRNA uridylation. Cell. 2009;138:696–708. doi: 10.1016/j.cell.2009.08.002. [DOI] [PubMed] [Google Scholar]
- Hernandez-Rapp J, Smith PY, Filali M, Goupil C, Planel E, Magill ST, Goodman RH, Hebert SS. Memory formation and retention are affected in adult miR-132/212 knockout mice. Behavioural brain research. 2015;287:15–26. doi: 10.1016/j.bbr.2015.03.032. [DOI] [PubMed] [Google Scholar]
- Hiramuki Y, Sato T, Furuta Y, Surani MA, Sehara-Fujisawa A. Mest but not miR-335 affects skeletal muscle growth and regeneration. PLoS One. 2015;10:e0130436. doi: 10.1371/journal.pone.0130436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoover AR, Dozmorov I, MacLeod J, Du Q, de la Morena MT, Forbess J, Guleserian K, Cleaver OB, van Oers NS. MicroRNA-205 maintains T cell development following stress by regulating Forkhead Box N1 and selected chemokines. J Biol Chem. 2016;291:23237–23247. doi: 10.1074/jbc.M116.744508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horie T, Nishino T, Baba O, Kuwabara Y, Nakao T, Nishiga M, Usami S, Izuhara M, Sowa N, Yahagi N, et al. MicroRNA-33 regulates sterol regulatory element-binding protein 1 expression in mice. Nature communications. 2013;4:2883. doi: 10.1038/ncomms3883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horie T, Ono K, Horiguchi M, Nishi H, Nakamura T, Nagao K, Kinoshita M, Kuwabara Y, Marusawa H, Iwanaga Y, et al. MicroRNA-33 encoded by an intron of sterol regulatory element-binding protein 2 (Srebp2) regulates HDL in vivo. Proc Natl Acad Sci U S A. 2010;107:17321–17326. doi: 10.1073/pnas.1008499107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hornstein E, Shomron N. Canalization of development by microRNAs. Nat Genet. 2006;38(Suppl):S20–24. doi: 10.1038/ng1803. [DOI] [PubMed] [Google Scholar]
- Hsu SH, Delgado ER, Otero PA, Teng KY, Kutay H, Meehan KM, Moroney JB, Monga JK, Hand NJ, Friedman JR, et al. MicroRNA-122 regulates polyploidization in the murine liver. Hepatology. 2016;64:599–615. doi: 10.1002/hep.28573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsu SH, Wang B, Kota J, Yu J, Costinean S, Kutay H, Yu L, Bai S, La Perle K, Chivukula RR, et al. Essential metabolic, anti-inflammatory, and anti-tumorigenic functions of miR-122 in liver. The Journal of clinical investigation. 2012;122:2871–2883. doi: 10.1172/JCI63539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu CH, Sui BD, Du FY, Shuai Y, Zheng CX, Zhao P, Yu XR, Jin Y. miR-21 deficiency inhibits osteoclast function and prevents bone loss in mice. Scientific reports. 2017;7:43191. doi: 10.1038/srep43191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu W, Dooley J, Chung SS, Chandramohan D, Cimmino L, Mukherjee S, Mason CE, de Strooper B, Liston A, Park CY. miR-29a maintains mouse hematopoietic stem cell self-renewal by regulating Dnmt3a. Blood. 2015;125:2206–2216. doi: 10.1182/blood-2014-06-585273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang ZP, Chen J, Seok HY, Zhang Z, Kataoka M, Hu X, Wang DZ. MicroRNA-22 regulates cardiac hypertrophy and remodeling in response to stress. Circulation research. 2013;112:1234–1243. doi: 10.1161/CIRCRESAHA.112.300682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hutvagner G, McLachlan J, Pasquinelli AE, Balint E, Tuschl T, Zamore PD. A cellular function for the RNA-interference enzyme Dicer in the maturation of the let-7 small temporal RNA. Science. 2001;293:834–838. doi: 10.1126/science.1062961. [DOI] [PubMed] [Google Scholar]
- Hutvagner G, Zamore PD. A microRNA in a multiple-turnover RNAi enzyme complex. Science. 2002;297:2056–2060. doi: 10.1126/science.1073827. [DOI] [PubMed] [Google Scholar]
- Iwasaki S, Kobayashi M, Yoda M, Sakaguchi Y, Katsuma S, Suzuki T, Tomari Y. Hsc70/Hsp90 chaperone machinery mediates ATP-dependent RISC loading of small RNA duplexes. Mol Cell. 2010;39:292–299. doi: 10.1016/j.molcel.2010.05.015. [DOI] [PubMed] [Google Scholar]
- Jan CH, Friedman RC, Ruby JG, Bartel DP. Formation, regulation and evolution of Caenorhabditis elegans 3′UTRs. Nature. 2011;469:97–101. doi: 10.1038/nature09616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jee D, Yang JS, Park SM, Farmer DT, Wen J, Chou T, Chow A, McManus MT, Kharas MG, Lai EC. Dual strategies for Argonaute2-mediated biogenesis of erythroid miRNAs underlie conserved requirements for slicing in mammals. Mol Cell. 2018;69:265–278 e266. doi: 10.1016/j.molcel.2017.12.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jo S, Chen J, Xu G, Grayson TB, Thielen LA, Shalev A. miR-204 controls Glucagon-like Peptide 1 Receptor expression and agonist function. Diabetes. 2018;67:256–264. doi: 10.2337/db17-0506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnnidis JB, Harris MH, Wheeler RT, Stehling-Sun S, Lam MH, Kirak O, Brummelkamp TR, Fleming MD, Camargo FD. Regulation of progenitor cell proliferation and granulocyte function by microRNA-223. Nature. 2008;451:1125–1129. doi: 10.1038/nature06607. [DOI] [PubMed] [Google Scholar]
- Johnston RJ, Hobert O. A microRNA controlling left/right neuronal asymmetry in Caenorhabditis elegans. Nature. 2003;426:845–849. doi: 10.1038/nature02255. [DOI] [PubMed] [Google Scholar]
- Jonas S, Izaurralde E. Towards a molecular understanding of microRNA-mediated gene silencing. Nat Rev Genet. 2015;16:421–433. doi: 10.1038/nrg3965. [DOI] [PubMed] [Google Scholar]
- Jones-Rhoades MW, Bartel DP, Bartel B. MicroRNAs and their regulatory roles in plants. Annu Rev Plant Biol. 2006;57:19–53. doi: 10.1146/annurev.arplant.57.032905.105218. [DOI] [PubMed] [Google Scholar]
- Kamenska A, Lu WT, Kubacka D, Broomhead H, Minshall N, Bushell M, Standart N. Human 4E-T represses translation of bound mRNAs and enhances microRNA-mediated silencing. Nucleic Acids Res. 2014;42:3298–3313. doi: 10.1093/nar/gkt1265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamenska A, Simpson C, Vindry C, Broomhead H, Benard M, Ernoult-Lange M, Lee BP, Harries LW, Weil D, Standart N. The DDX6-4E-T interaction mediates translational repression and P-body assembly. Nucleic Acids Res. 2016;44:6318–6334. doi: 10.1093/nar/gkw565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kasper DM, Moro A, Ristori E, Narayanan A, Hill-Teran G, Fleming E, Moreno-Mateos M, Vejnar CE, Zhang J, Lee D, et al. MicroRNAs establish uniform traits during the architecture of vertebrate embryos. Dev Cell. 2017;40:552–565 e555. doi: 10.1016/j.devcel.2017.02.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawahara Y, Megraw M, Kreider E, Iizasa H, Valente L, Hatzigeorgiou AG, Nishikura K. Frequency and fate of microRNA editing in human brain. Nucleic Acids Res. 2008;36:5270–5280. doi: 10.1093/nar/gkn479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawahara Y, Zinshteyn B, Chendrimada TP, Shiekhattar R, Nishikura K. RNA editing of the microRNA-151 precursor blocks cleavage by the Dicer-TRBP complex. EMBO reports. 2007a;8:763–769. doi: 10.1038/sj.embor.7401011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawahara Y, Zinshteyn B, Sethupathy P, Iizasa H, Hatzigeorgiou AG, Nishikura K. Redirection of silencing targets by adenosine-to-inosine editing of miRNAs. Science. 2007b;315:1137–1140. doi: 10.1126/science.1138050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawamata T, Tomari Y. Making RISC. Trends in biochemical sciences. 2010;35:368–376. doi: 10.1016/j.tibs.2010.03.009. [DOI] [PubMed] [Google Scholar]
- Khvorova A, Reynolds A, Jayasena SD. Functional siRNAs and miRNAs exhibit strand bias. Cell. 2003;115:209–216. doi: 10.1016/s0092-8674(03)00801-8. [DOI] [PubMed] [Google Scholar]
- Kiessling S, Ucar A, Chowdhury K, Oster H, Eichele G. Genetic background-dependent effects of murine micro RNAs on circadian clock function. PLoS One. 2017;12:e0176547. doi: 10.1371/journal.pone.0176547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim D, Sung YM, Park J, Kim S, Kim J, Park J, Ha H, Bae JY, Kim S, Baek D. General rules for functional microRNA targeting. Nat Genet. 2016a;48:1517–1526. doi: 10.1038/ng.3694. [DOI] [PubMed] [Google Scholar]
- Kim J, Siverly AN, Chen D, Wang M, Yuan Y, Wang Y, Lee H, Zhang J, Muller WJ, Liang H, et al. Ablation of miR-10b suppresses oncogene-induced mammary tumorigenesis and metastasis and reactivates tumor-suppressive pathways. Cancer research. 2016b;76:6424–6435. doi: 10.1158/0008-5472.CAN-16-1571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim J, Yoon H, Horie T, Burchett JM, Restivo JL, Rotllan N, Ramirez CM, Verghese PB, Ihara M, Hoe HS, et al. microRNA-33 regulates ApoE lipidation and Amyloid-beta metabolism in the brain. The Journal of neuroscience: the official journal of the Society for Neuroscience. 2015;35:14717–14726. doi: 10.1523/JNEUROSCI.2053-15.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim YK, Kim B, Kim VN. Re-evaluation of the roles of DROSHA, Exportin 5, and DICER in microRNA biogenesis. Proc Natl Acad Sci U S A. 2016c;113:E1881–1889. doi: 10.1073/pnas.1602532113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kleaveland B, Shi CY, Bartel DP. A network of noncoding regulatory RNAs acts in the mammalian brain. bioRxiv. 2018:279687. doi: 10.1016/j.cell.2018.05.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klein U, Lia M, Crespo M, Siegel R, Shen Q, Mo T, Ambesi-Impiombato A, Califano A, Migliazza A, Bhagat G, et al. The DLEU2/miR-15a/16-1 cluster controls B cell proliferation and its deletion leads to chronic lymphocytic leukemia. Cancer cell. 2010;17:28–40. doi: 10.1016/j.ccr.2009.11.019. [DOI] [PubMed] [Google Scholar]
- Klum SM, Chandradoss SD, Schirle NT, Joo C, MacRae IJ. Helix-7 in Argonaute2 shapes the microRNA seed region for rapid target recognition. EMBO J. 2018;37:75–88. doi: 10.15252/embj.201796474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kohlhaas S, Garden OA, Scudamore C, Turner M, Okkenhaug K, Vigorito E. Cutting edge: the Foxp3 target miR-155 contributes to the development of regulatory T cells. Journal of immunology. 2009;182:2578–2582. doi: 10.4049/jimmunol.0803162. [DOI] [PubMed] [Google Scholar]
- Kozomara A, Griffiths-Jones S. miRBase: annotating high confidence microRNAs using deep sequencing data. Nucleic Acids Res. 2014;42:D68–73. doi: 10.1093/nar/gkt1181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kramer NJ, Wang WL, Reyes EY, Kumar B, Chen CC, Ramakrishna C, Cantin EM, Vonderfecht SL, Taganov KD, Chau N, et al. Altered lymphopoiesis and immunodeficiency in miR-142 null mice. Blood. 2015;125:3720–3730. doi: 10.1182/blood-2014-10-603951. [DOI] [PubMed] [Google Scholar]
- Krol J, Busskamp V, Markiewicz I, Stadler MB, Ribi S, Richter J, Duebel J, Bicker S, Fehling HJ, Schubeler D, et al. Characterizing light-regulated retinal microRNAs reveals rapid turnover as a common property of neuronal microRNAs. Cell. 2010;141:618–631. doi: 10.1016/j.cell.2010.03.039. [DOI] [PubMed] [Google Scholar]
- Krutzfeldt J, Rajewsky N, Braich R, Rajeev KG, Tuschl T, Manoharan M, Stoffel M. Silencing of microRNAs in vivo with ‘antagomirs’. Nature. 2005;438:685–689. doi: 10.1038/nature04303. [DOI] [PubMed] [Google Scholar]
- Krzeszinski JY, Wei W, Huynh H, Jin Z, Wang X, Chang TC, Xie XJ, He L, Mangala LS, Lopez-Berestein G, et al. miR-34a blocks osteoporosis and bone metastasis by inhibiting osteoclastogenesis and Tgif2. Nature. 2014;512:431–435. doi: 10.1038/nature13375. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Kuhn S, Johnson SL, Furness DN, Chen J, Ingham N, Hilton JM, Steffes G, Lewis MA, Zampini V, Hackney CM, et al. miR-96 regulates the progression of differentiation in mammalian cochlear inner and outer hair cells. Proc Natl Acad Sci U S A. 2011;108:2355–2360. doi: 10.1073/pnas.1016646108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuhnert F, Mancuso MR, Hampton J, Stankunas K, Asano T, Chen CZ, Kuo CJ. Attribution of vascular phenotypes of the murine Egfl7 locus to the microRNA miR-126. Development. 2008;135:3989–3993. doi: 10.1242/dev.029736. [DOI] [PubMed] [Google Scholar]
- Kumarswamy R, Volkmann I, Beermann J, Napp LC, Jabs O, Bhayadia R, Melk A, Ucar A, Chowdhury K, Lorenzen JM, et al. Vascular importance of the miR-212/132 cluster. European heart journal. 2014;35:3224–3231. doi: 10.1093/eurheartj/ehu344. [DOI] [PubMed] [Google Scholar]
- Kurkewich JL, Bikorimana E, Nguyen T, Klopfenstein N, Zhang H, Hallas WM, Stayback G, McDowell MA, Dahl R. The mirn23a microRNA cluster antagonizes B cell development. Journal of leukocyte biology. 2016;100:665–677. doi: 10.1189/jlb.1HI0915-398RR. [DOI] [PubMed] [Google Scholar]
- Kurkewich JL, Boucher A, Klopfenstein N, Baskar R, Kapur R, Dahl R. The mirn23a and mirn23b microrna clusters are necessary for proper hematopoietic progenitor cell production and differentiation. Experimental hematology. 2017 doi: 10.1016/j.exphem.2017.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Labialle S, Marty V, Bortolin-Cavaille ML, Hoareau-Osman M, Pradere JP, Valet P, Martin PG, Cavaille J. The miR-379/miR-410 cluster at the imprinted Dlk1-Dio3 domain controls neonatal metabolic adaptation. EMBO J. 2014;33:2216–2230. doi: 10.15252/embj.201387038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lagos-Quintana M, Rauhut R, Lendeckel W, Tuschl T. Identification of novel genes coding for small expressed RNAs. Science. 2001;294:853–858. doi: 10.1126/science.1064921. [DOI] [PubMed] [Google Scholar]
- Landgraf P, Rusu M, Sheridan R, Sewer A, Iovino N, Aravin A, Pfeffer S, Rice A, Kamphorst AO, Landthaler M, et al. A mammalian microRNA expression atlas based on small RNA library sequencing. Cell. 2007;129:1401–1414. doi: 10.1016/j.cell.2007.04.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Latreille M, Hausser J, Stutzer I, Zhang Q, Hastoy B, Gargani S, Kerr-Conte J, Pattou F, Zavolan M, Esguerra JL, et al. MicroRNA-7a regulates pancreatic beta cell function. The Journal of clinical investigation. 2014;124:2722–2735. doi: 10.1172/JCI73066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lau NC, Lim LP, Weinstein EG, Bartel DP. An abundant class of tiny RNAs with probable regulatory roles in Caenorhabditis elegans. Science. 2001;294:858–862. doi: 10.1126/science.1065062. [DOI] [PubMed] [Google Scholar]
- Lee A, Papangeli I, Park Y, Jeong HN, Choi J, Kang H, Jo HN, Kim J, Chun HJ. A PPARgamma-dependent miR-424/503-CD40 axis regulates inflammation mediated angiogenesis. Scientific reports. 2017;7:2528. doi: 10.1038/s41598-017-02852-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee HC, Li L, Gu W, Xue Z, Crosthwaite SK, Pertsemlidis A, Lewis ZA, Freitag M, Selker EU, Mello CC, et al. Diverse pathways generate microRNA-like RNAs and Dicer-independent small interfering RNAs in fungi. Mol Cell. 2010;38:803–814. doi: 10.1016/j.molcel.2010.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee RC, Ambros V. An extensive class of small RNAs in Caenorhabditis elegans. Science. 2001;294:862–864. doi: 10.1126/science.1065329. [DOI] [PubMed] [Google Scholar]
- Lee RC, Feinbaum RL, Ambros V. The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell. 1993;75:843–854. doi: 10.1016/0092-8674(93)90529-y. [DOI] [PubMed] [Google Scholar]
- Lee S, Song J, Kim S, Kim J, Hong Y, Kim Y, Kim D, Baek D, Ahn K. Selective degradation of host MicroRNAs by an intergenic HCMV noncoding RNA accelerates virus production. Cell host & microbe. 2013;13:678–690. doi: 10.1016/j.chom.2013.05.007. [DOI] [PubMed] [Google Scholar]
- Lee Y, Ahn C, Han J, Choi H, Kim J, Yim J, Lee J, Provost P, Radmark O, Kim S, et al. The nuclear RNase III Drosha initiates microRNA processing. Nature. 2003;425:415–419. doi: 10.1038/nature01957. [DOI] [PubMed] [Google Scholar]
- Lee Y, Jeon K, Lee JT, Kim S, Kim VN. MicroRNA maturation: stepwise processing and subcellular localization. EMBO J. 2002;21:4663–4670. doi: 10.1093/emboj/cdf476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee Y, Kim M, Han J, Yeom KH, Lee S, Baek SH, Kim VN. MicroRNA genes are transcribed by RNA polymerase II. EMBO J. 2004;23:4051–4060. doi: 10.1038/sj.emboj.7600385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lewis BP, Burge CB, Bartel DP. Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell. 2005;120:15–20. doi: 10.1016/j.cell.2004.12.035. [DOI] [PubMed] [Google Scholar]
- Lewis MA, Quint E, Glazier AM, Fuchs H, De Angelis MH, Langford C, van Dongen S, Abreu-Goodger C, Piipari M, Redshaw N, et al. An ENU-induced mutation of miR-96 associated with progressive hearing loss in mice. Nat Genet. 2009;41:614–618. doi: 10.1038/ng.369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li CJ, Cheng P, Liang MK, Chen YS, Lu Q, Wang JY, Xia ZY, Zhou HD, Cao X, Xie H, et al. MicroRNA-188 regulates age-related switch between osteoblast and adipocyte differentiation. The Journal of clinical investigation. 2015;125:1509–1522. doi: 10.1172/JCI77716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li J, Yang Z, Yu B, Liu J, Chen X. Methylation protects miRNAs and siRNAs from a 3′-end uridylation activity in Arabidopsis. Curr Biol. 2005;15:1501–1507. doi: 10.1016/j.cub.2005.07.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li X, Zhao F, Xin M, Li G, Luna C, Li G, Zhou Q, He Y, Yu B, Olson E, et al. Regulation of intraocular pressure by microRNA cluster miR-143/145. Scientific reports. 2017;7:915. doi: 10.1038/s41598-017-01003-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li YF, Ou X, Xu S, Jin ZB, Iwai N, Lam KP. Loss of miR-182 affects B-cell extrafollicular antibody response. Immunology. 2016;148:140–149. doi: 10.1111/imm.12592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Libri V, Helwak A, Miesen P, Santhakumar D, Borger JG, Kudla G, Grey F, Tollervey D, Buck AH. Murine cytomegalovirus encodes a miR-27 inhibitor disguised as a target. Proc Natl Acad Sci U S A. 2012;109:279–284. doi: 10.1073/pnas.1114204109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lim LP, Lau NC, Garrett-Engele P, Grimson A, Schelter JM, Castle J, Bartel DP, Linsley PS, Johnson JM. Microarray analysis shows that some microRNAs downregulate large numbers of target mRNAs. Nature. 2005;433:769–773. doi: 10.1038/nature03315. [DOI] [PubMed] [Google Scholar]
- Lim LP, Lau NC, Weinstein EG, Abdelhakim A, Yekta S, Rhoades MW, Burge CB, Bartel DP. The microRNAs of Caenorhabditis elegans. Genes Dev. 2003;17:991–1008. doi: 10.1101/gad.1074403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu J, Carmell MA, Rivas FV, Marsden CG, Thomson JM, Song JJ, Hammond SM, Joshua-Tor L, Hannon GJ. Argonaute2 is the catalytic engine of mammalian RNAi. Science. 2004;305:1437–1441. doi: 10.1126/science.1102513. [DOI] [PubMed] [Google Scholar]
- Liu N, Bezprozvannaya S, Shelton JM, Frisard MI, Hulver MW, McMillan RP, Wu Y, Voelker KA, Grange RW, Richardson JA, et al. Mice lacking microRNA 133a develop dynamin 2-dependent centronuclear myopathy. The Journal of clinical investigation. 2011;121:3258–3268. doi: 10.1172/JCI46267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu N, Bezprozvannaya S, Williams AH, Qi X, Richardson JA, Bassel-Duby R, Olson EN. microRNA-133a regulates cardiomyocyte proliferation and suppresses smooth muscle gene expression in the heart. Genes Dev. 2008;22:3242–3254. doi: 10.1101/gad.1738708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu N, Williams AH, Maxeiner JM, Bezprozvannaya S, Shelton JM, Richardson JA, Bassel-Duby R, Olson EN. microRNA-206 promotes skeletal muscle regeneration and delays progression of Duchenne muscular dystrophy in mice. The Journal of clinical investigation. 2012;122:2054–2065. doi: 10.1172/JCI62656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu W, Bi P, Shan T, Yang X, Yin H, Wang YX, Liu N, Rudnicki MA, Kuang S. miR-133a regulates adipocyte browning in vivo. PLoS Genet. 2013;9:e1003626. doi: 10.1371/journal.pgen.1003626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Z, Bai J, Zhang L, Lou F, Ke F, Cai W, Wang H. Conditional knockout of microRNA-31 promotes the development of colitis associated cancer. Biochemical and biophysical research communications. 2017;490:62–68. doi: 10.1016/j.bbrc.2017.06.012. [DOI] [PubMed] [Google Scholar]
- Llobet-Navas D, Rodriguez-Barrueco R, Castro V, Ugalde AP, Sumazin P, Jacob-Sendler D, Demircan B, Castillo-Martin M, Putcha P, Marshall N, et al. The miR-424(322)/503 cluster orchestrates remodeling of the epithelium in the involuting mammary gland. Genes Dev. 2014;28:765–782. doi: 10.1101/gad.237404.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loeb GB, Khan AA, Canner D, Hiatt JB, Shendure J, Darnell RB, Leslie CS, Rudensky AY. Transcriptome-wide miR-155 binding map reveals widespread noncanonical microRNA targeting. Molecular Cell. 2012;48:760–770. doi: 10.1016/j.molcel.2012.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lovat F, Fassan M, Gasparini P, Rizzotto L, Cascione L, Pizzi M, Vicentini C, Balatti V, Palmieri D, Costinean S, et al. miR-15b/16-2 deletion promotes B-cell malignancies. Proc Natl Acad Sci U S A. 2015;112:11636–11641. doi: 10.1073/pnas.1514954112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu C, Tej SS, Luo S, Haudenschild CD, Meyers BC, Green PJ. Elucidation of the small RNA component of the transcriptome. Science. 2005;309:1567–1569. doi: 10.1126/science.1114112. [DOI] [PubMed] [Google Scholar]
- Lu LF, Boldin MP, Chaudhry A, Lin LL, Taganov KD, Hanada T, Yoshimura A, Baltimore D, Rudensky AY. Function of miR-146a in controlling Treg cell-mediated regulation of Th1 responses. Cell. 2010;142:914–929. doi: 10.1016/j.cell.2010.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu LF, Gasteiger G, Yu IS, Chaudhry A, Hsin JP, Lu Y, Bos PD, Lin LL, Zawislak CL, Cho S, et al. A single miRNA-mRNA interaction affects the immune response in a context- and cell-type-specific manner. Immunity. 2015a;43:52–64. doi: 10.1016/j.immuni.2015.04.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu LF, Thai TH, Calado DP, Chaudhry A, Kubo M, Tanaka K, Loeb GB, Lee H, Yoshimura A, Rajewsky K, et al. Foxp3-dependent microRNA155 confers competitive fitness to regulatory T cells by targeting SOCS1 protein. Immunity. 2009;30:80–91. doi: 10.1016/j.immuni.2008.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu TX, Lim EJ, Besse JA, Itskovich S, Plassard AJ, Fulkerson PC, Aronow BJ, Rothenberg ME. MiR-223 deficiency increases eosinophil progenitor proliferation. Journal of immunology. 2013a;190:1576–1582. doi: 10.4049/jimmunol.1202897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu TX, Lim EJ, Itskovich S, Besse JA, Plassard AJ, Mingler MK, Rothenberg JA, Fulkerson PC, Aronow BJ, Rothenberg ME. Targeted ablation of miR-21 decreases murine eosinophil progenitor cell growth. PLoS One. 2013b;8:e59397. doi: 10.1371/journal.pone.0059397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu W, You R, Yuan X, Yang T, Samuel EL, Marcano DC, Sikkema WK, Tour JM, Rodriguez A, Kheradmand F, et al. The microRNA miR-22 inhibits the histone deacetylase HDAC4 to promote T(H)17 cell-dependent emphysema. Nature immunology. 2015b;16:1185–1194. doi: 10.1038/ni.3292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lumayag S, Haldin CE, Corbett NJ, Wahlin KJ, Cowan C, Turturro S, Larsen PE, Kovacs B, Witmer PD, Valle D, et al. Inactivation of the microRNA-183/96/182 cluster results in syndromic retinal degeneration. Proc Natl Acad Sci U S A. 2013;110:E507–516. doi: 10.1073/pnas.1212655110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lund E, Guttinger S, Calado A, Dahlberg JE, Kutay U. Nuclear export of microRNA precursors. Science. 2004;303:95–98. doi: 10.1126/science.1090599. [DOI] [PubMed] [Google Scholar]
- Lykken EA, Li QJ. The microRNA miR-191 supports T cell survival following common gamma chain signaling. J Biol Chem. 2016;291:23532–23544. doi: 10.1074/jbc.M116.741264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma H, Wu Y, Choi JG, Wu H. Lower and upper stem-single-stranded RNA junctions together determine the Drosha cleavage site. Proc Natl Acad Sci U S A. 2013;110:20687–20692. doi: 10.1073/pnas.1311639110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma JB, Yuan YR, Meister G, Pei Y, Tuschl T, Patel DJ. Structural basis for 5′-end-specific recognition of guide RNA by the A. fulgidus Piwi protein. Nature. 2005;434:666–670. doi: 10.1038/nature03514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma X, Yan F, Deng Q, Li F, Lu Z, Liu M, Wang L, Conklin DJ, McCracken J, Srivastava S, et al. Modulation of tumorigenesis by the pro-inflammatory microRNA miR-301a in mouse models of lung cancer and colorectal cancer. Cell discovery. 2015;1:15005. doi: 10.1038/celldisc.2015.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma YC, Zhang L, Dai LL, Khan RU, Zou CG. mir-67 regulates P. aeruginosa avoidance behavior in C. elegans. Biochemical and biophysical research communications. 2017;494:120–125. doi: 10.1016/j.bbrc.2017.10.069. [DOI] [PubMed] [Google Scholar]
- Madison BB, Liu Q, Zhong X, Hahn CM, Lin N, Emmett MJ, Stanger BZ, Lee JS, Rustgi AK. LIN28B promotes growth and tumorigenesis of the intestinal epithelium via Let-7. Genes Dev. 2013;27:2233–2245. doi: 10.1101/gad.224659.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malone CD, Hannon GJ. Small RNAs as guardians of the genome. Cell. 2009;136:656–668. doi: 10.1016/j.cell.2009.01.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maoa R, Zou F, Yang L, Lin S, Li Y, Ma M, Yin P, Liang X, Liu J. The loss of miR-139-5p promotes colitis-associated tumorigenesis by mediating PI3K/AKT/Wnt signaling. The international journal of biochemistry & cell biology. 2015;69:153–161. doi: 10.1016/j.biocel.2015.10.008. [DOI] [PubMed] [Google Scholar]
- Marcinowski L, Tanguy M, Krmpotic A, Radle B, Lisnic VJ, Tuddenham L, Chane-Woon-Ming B, Ruzsics Z, Erhard F, Benkartek C, et al. Degradation of cellular mir-27 by a novel, highly abundant viral transcript is important for efficient virus replication in vivo. PLoS pathogens. 2012;8:e1002510. doi: 10.1371/journal.ppat.1002510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marson A, Levine SS, Cole MF, Frampton GM, Brambrink T, Johnstone S, Guenther MG, Johnston WK, Wernig M, Newman J, et al. Connecting microRNA genes to the core transcriptional regulatory circuitry of embryonic stem cells. Cell. 2008;134:521–533. doi: 10.1016/j.cell.2008.07.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marty V, Labialle S, Bortolin-Cavaille ML, Ferreira De Medeiros G, Moisan MP, Florian C, Cavaille J. Deletion of the miR-379/miR-410 gene cluster at the imprinted Dlk1-Dio3 locus enhances anxiety-related behaviour. Human molecular genetics. 2016;25:728–739. doi: 10.1093/hmg/ddv510. [DOI] [PubMed] [Google Scholar]
- Marzi MJ, Ghini F, Cerruti B, de Pretis S, Bonetti P, Giacomelli C, Gorski MM, Kress T, Pelizzola M, Muller H, et al. Degradation dynamics of microRNAs revealed by a novel pulse-chase approach. Genome Res. 2016;26:554–565. doi: 10.1101/gr.198788.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mazziotti R, Baroncelli L, Ceglia N, Chelini G, Sala GD, Magnan C, Napoli D, Putignano E, Silingardi D, Tola J, et al. Mir-132/212 is required for maturation of binocular matching of orientation preference and depth perception. Nature communications. 2017;8:15488. doi: 10.1038/ncomms15488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McJunkin K, Ambros V. A microRNA family exerts maternal control on sex determination in C. elegans. Genes Dev. 2017;31:422–437. doi: 10.1101/gad.290155.116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Medeiros LA, Dennis LM, Gill ME, Houbaviy H, Markoulaki S, Fu D, White AC, Kirak O, Sharp PA, Page DC, et al. Mir-290-295 deficiency in mice results in partially penetrant embryonic lethality and germ cell defects. Proc Natl Acad Sci U S A. 2011;108:14163–14168. doi: 10.1073/pnas.1111241108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Medrano S, Sequeira-Lopez ML, Gomez RA. Deletion of the miR-143/145 cluster leads to hydronephrosis in mice. The American journal of pathology. 2014;184:3226–3238. doi: 10.1016/j.ajpath.2014.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mehta A, Zhao JL, Sinha N, Marinov GK, Mann M, Kowalczyk MS, Galimidi RP, Du X, Erikci E, Regev A, et al. The microRNA-132 and microRNA-212 cluster regulates hematopoietic stem cell maintenance and survival with age by buffering FOXO3 expression. Immunity. 2015;42:1021–1032. doi: 10.1016/j.immuni.2015.05.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meister G, Landthaler M, Patkaniowska A, Dorsett Y, Teng G, Tuschl T. Human Argonaute2 mediates RNA cleavage targeted by miRNAs and siRNAs. Mol Cell. 2004;15:185–197. doi: 10.1016/j.molcel.2004.07.007. [DOI] [PubMed] [Google Scholar]
- Melton C, Judson RL, Blelloch R. Opposing microRNA families regulate self-renewal in mouse embryonic stem cells. Nature. 2010;463:621–626. doi: 10.1038/nature08725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Memczak S, Jens M, Elefsinioti A, Torti F, Krueger J, Rybak A, Maier L, Mackowiak SD, Gregersen LH, Munschauer M, et al. Circular RNAs are a large class of animal RNAs with regulatory potency. Nature. 2013;495:333–338. doi: 10.1038/nature11928. [DOI] [PubMed] [Google Scholar]
- Mencia A, Modamio-Hoybjor S, Redshaw N, Morin M, Mayo-Merino F, Olavarrieta L, Aguirre LA, del Castillo I, Steel KP, Dalmay T, et al. Mutations in the seed region of human miR-96 are responsible for nonsyndromic progressive hearing loss. Nat Genet. 2009;41:609–613. doi: 10.1038/ng.355. [DOI] [PubMed] [Google Scholar]
- Mendell JT, Olson EN. MicroRNAs in stress signaling and human disease. Cell. 2012;148:1172–1187. doi: 10.1016/j.cell.2012.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mendoza-Viveros L, Chiang CK, Ong JLK, Hegazi S, Cheng AH, Bouchard-Cannon P, Fana M, Lowden C, Zhang P, Bothorel B, et al. miR-132/212 modulates seasonal adaptation and dendritic morphology of the central circadian clock. Cell reports. 2017;19:505–520. doi: 10.1016/j.celrep.2017.03.057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mildner A, Chapnik E, Manor O, Yona S, Kim KW, Aychek T, Varol D, Beck G, Itzhaki ZB, Feldmesser E, et al. Mononuclear phagocyte miRNome analysis identifies miR-142 as critical regulator of murine dendritic cell homeostasis. Blood. 2013;121:1016–1027. doi: 10.1182/blood-2012-07-445999. [DOI] [PubMed] [Google Scholar]
- Mildner A, Chapnik E, Varol D, Aychek T, Lampl N, Rivkin N, Bringmann A, Paul F, Boura-Halfon S, Hayoun YS, et al. MicroRNA-142 controls thymocyte proliferation. European journal of immunology. 2017;47:1142–1152. doi: 10.1002/eji.201746987. [DOI] [PubMed] [Google Scholar]
- Miska EA, Alvarez-Saavedra E, Abbott AL, Lau NC, Hellman AB, McGonagle SM, Bartel DP, Ambros VR, Horvitz HR. Most Caenorhabditis elegans microRNAs are individually not essential for development or viability. PLoS Genet. 2007;3:e215. doi: 10.1371/journal.pgen.0030215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyaki S, Sato T, Inoue A, Otsuki S, Ito Y, Yokoyama S, Kato Y, Takemoto F, Nakasa T, Yamashita S, et al. MicroRNA-140 plays dual roles in both cartilage development and homeostasis. Genes Dev. 2010;24:1173–1185. doi: 10.1101/gad.1915510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moffett HF, Cartwright ANR, Kim HJ, Godec J, Pyrdol J, Aijo T, Martinez GJ, Rao A, Lu J, Golub TR, et al. The microRNA miR-31 inhibits CD8(+) T cell function in chronic viral infection. Nature immunology. 2017;18:791–799. doi: 10.1038/ni.3755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mohammed J, Flynt AS, Panzarino AM, Mondal MMH, DeCruz M, Siepel A, Lai EC. Deep experimental profiling of microRNA diversity, deployment, and evolution across the Drosophila genus. Genome Res. 2018;28:52–65. doi: 10.1101/gr.226068.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mok Y, Schwierzeck V, Thomas DC, Vigorito E, Rayner TF, Jarvis LB, Prosser HM, Bradley A, Withers DR, Martensson IL, et al. MiR-210 is induced by Oct-2, regulates B cells, and inhibits autoantibody production. Journal of immunology. 2013;191:3037–3048. doi: 10.4049/jimmunol.1301289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Molnar A, Schwach F, Studholme DJ, Thuenemann EC, Baulcombe DC. miRNAs control gene expression in the single-cell alga Chlamydomonas reinhardtii. Nature. 2007;447:1126–1129. doi: 10.1038/nature05903. [DOI] [PubMed] [Google Scholar]
- Moon HG, Yang J, Zheng Y, Jin Y. miR-15a/16 regulates macrophage phagocytosis after bacterial infection. Journal of immunology. 2014;193:4558–4567. doi: 10.4049/jimmunol.1401372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moran Y, Fredman D, Praher D, Li XZ, Wee LM, Rentzsch F, Zamore PD, Technau U, Seitz H. Cnidarian microRNAs frequently regulate targets by cleavage. Genome Res. 2014;24:651–663. doi: 10.1101/gr.162503.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mori M, Triboulet R, Mohseni M, Schlegelmilch K, Shrestha K, Camargo FD, Gregory RI. Hippo signaling regulates microprocessor and links cell-density-dependent miRNA biogenesis to cancer. Cell. 2014;156:893–906. doi: 10.1016/j.cell.2013.12.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morin RD, O’Connor MD, Griffith M, Kuchenbauer F, Delaney A, Prabhu AL, Zhao Y, McDonald H, Zeng T, Hirst M, et al. Application of massively parallel sequencing to microRNA profiling and discovery in human embryonic stem cells. Genome Res. 2008;18:610–621. doi: 10.1101/gr.7179508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moroz LL, Kocot KM, Citarella MR, Dosung S, Norekian TP, Povolotskaya IS, Grigorenko AP, Dailey C, Berezikov E, Buckley KM, et al. The ctenophore genome and the evolutionary origins of neural systems. Nature. 2014;510:109–114. doi: 10.1038/nature13400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moss EG, Lee RC, Ambros V. The cold shock domain protein LIN-28 controls developmental timing in C. elegans and is regulated by the lin-4 RNA. Cell. 1997;88:637–646. doi: 10.1016/s0092-8674(00)81906-6. [DOI] [PubMed] [Google Scholar]
- Muraleedharan CK, McClellan SA, Barrett RP, Li C, Montenegro D, Carion T, Berger E, Hazlett LD, Xu S. Inactivation of the miR-183/96/182 cluster decreases the severity of Pseudomonas aeruginosa-induced keratitis. Investigative ophthalmology & visual science. 2016;57:1506–1517. doi: 10.1167/iovs.16-19134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakamura Y, Inloes JB, Katagiri T, Kobayashi T. Chondrocyte-specific microRNA-140 regulates endochondral bone development and targets Dnpep to modulate Bone Morphogenetic Protein signaling. Molecular and cellular biology. 2011;31:3019–3028. doi: 10.1128/MCB.05178-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakanishi K, Weinberg DE, Bartel DP, Patel DJ. Structure of yeast Argonaute with guide RNA. Nature. 2012;486:368–374. doi: 10.1038/nature11211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nam Y, Chen C, Gregory RI, Chou JJ, Sliz P. Molecular basis for interaction of let-7 microRNAs with Lin28. Cell. 2011;147:1080–1091. doi: 10.1016/j.cell.2011.10.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nehammer C, Podolska A, Mackowiak SD, Kagias K, Pocock R. Specific microRNAs regulate heat stress responses in Caenorhabditis elegans. Scientific reports. 2015;5:8866. doi: 10.1038/srep08866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nguyen TA, Jo MH, Choi YG, Park J, Kwon SC, Hohng S, Kim VN, Woo JS. Functional anatomy of the human Microprocessor. Cell. 2015;161:1374–1387. doi: 10.1016/j.cell.2015.05.010. [DOI] [PubMed] [Google Scholar]
- Nishiga M, Horie T, Kuwabara Y, Nagao K, Baba O, Nakao T, Nishino T, Hakuno D, Nakashima Y, Nishi H, et al. MicroRNA-33 controls adaptive fibrotic response in the remodeling heart by preserving lipid raft cholesterol. Circulation research. 2017;120:835–847. doi: 10.1161/CIRCRESAHA.116.309528. [DOI] [PubMed] [Google Scholar]
- Nishimura T, Padamsi Z, Fakim H, Milette S, Dunham WH, Gingras AC, Fabian MR. The eIF4E-Binding Protein 4E-T Is a Component of the mRNA Decay Machinery that Bridges the 5′ and 3′ Termini of Target mRNAs. Cell reports. 2015;11:1425–1436. doi: 10.1016/j.celrep.2015.04.065. [DOI] [PubMed] [Google Scholar]
- O’Connell RM, Chaudhuri AA, Rao DS, Baltimore D. Inositol phosphatase SHIP1 is a primary target of miR-155. Proc Natl Acad Sci U S A. 2009;106:7113–7118. doi: 10.1073/pnas.0902636106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Connell RM, Kahn D, Gibson WS, Round JL, Scholz RL, Chaudhuri AA, Kahn ME, Rao DS, Baltimore D. MicroRNA-155 promotes autoimmune inflammation by enhancing inflammatory T cell development. Immunity. 2010;33:607–619. doi: 10.1016/j.immuni.2010.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Donnell KA, Wentzel EA, Zeller KI, Dang CV, Mendell JT. c-Myc-regulated microRNAs modulate E2F1 expression. Nature. 2005;435:839–843. doi: 10.1038/nature03677. [DOI] [PubMed] [Google Scholar]
- Okamura K, Hagen JW, Duan H, Tyler DM, Lai EC. The mirtron pathway generates microRNA-class regulatory RNAs in Drosophila. Cell. 2007;130:89–100. doi: 10.1016/j.cell.2007.06.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olsen PH, Ambros V. The lin-4 regulatory RNA controls developmental timing in Caenorhabditis elegans by blocking LIN-14 protein synthesis after the initiation of translation. Dev Biol. 1999;216:671–680. doi: 10.1006/dbio.1999.9523. [DOI] [PubMed] [Google Scholar]
- Ozgur S, Basquin J, Kamenska A, Filipowicz W, Standart N, Conti E. Structure of a human 4E-T/DDX6/CNOT1 complex reveals the different interplay of DDX6-binding proteins with the CCR4-NOT complex. Cell reports. 2015;13:703–711. doi: 10.1016/j.celrep.2015.09.033. [DOI] [PubMed] [Google Scholar]
- Ozsolak F, Poling LL, Wang Z, Liu H, Liu XS, Roeder RG, Zhang X, Song JS, Fisher DE. Chromatin structure analyses identify miRNA promoters. Genes Dev. 2008;22:3172–3183. doi: 10.1101/gad.1706508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paikari A, C DB, Saw D, Blelloch R. The eutheria-specific miR-290 cluster modulates placental growth and maternal-fetal transport. Development. 2017;144:3731–3743. doi: 10.1242/dev.151654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pan W, Zhu S, Dai D, Liu Z, Li D, Li B, Gagliani N, Zheng Y, Tang Y, Weirauch MT, et al. miR-125a targets effector programs to stabilize Treg-mediated immune homeostasis. Nature communications. 2015;6:7096. doi: 10.1038/ncomms8096. [DOI] [PubMed] [Google Scholar]
- Papadopoulou AS, Dooley J, Linterman MA, Pierson W, Ucar O, Kyewski B, Zuklys S, Hollander GA, Matthys P, Gray DH, et al. The thymic epithelial microRNA network elevates the threshold for infection-associated thymic involution via miR-29a mediated suppression of the IFN-alpha receptor. Nature immunology. 2011;13:181–187. doi: 10.1038/ni.2193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Papadopoulou AS, Serneels L, Achsel T, Mandemakers W, Callaerts-Vegh Z, Dooley J, Lau P, Ayoubi T, Radaelli E, Spinazzi M, et al. Deficiency of the miR-29a/b-1 cluster leads to ataxic features and cerebellar alterations in mice. Neurobiology of disease. 2015;73:275–288. doi: 10.1016/j.nbd.2014.10.006. [DOI] [PubMed] [Google Scholar]
- Parchem RJ, Moore N, Fish JL, Parchem JG, Braga TT, Shenoy A, Oldham MC, Rubenstein JL, Schneider RA, Blelloch R. miR-302 Is required for timing of neural differentiation, neural tube closure, and embryonic viability. Cell reports. 2015;12:760–773. doi: 10.1016/j.celrep.2015.06.074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park CY, Jeker LT, Carver-Moore K, Oh A, Liu HJ, Cameron R, Richards H, Li Z, Adler D, Yoshinaga Y, et al. A resource for the conditional ablation of microRNAs in the mouse. Cell reports. 2012;1:385–391. doi: 10.1016/j.celrep.2012.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park JE, Heo I, Tian Y, Simanshu DK, Chang H, Jee D, Patel DJ, Kim VN. Dicer recognizes the 5′ end of RNA for efficient and accurate processing. Nature. 2011;475:201–205. doi: 10.1038/nature10198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parker JS, Roe SM, Barford D. Structural insights into mRNA recognition from a PIWI domain-siRNA guide complex. Nature. 2005;434:663–666. doi: 10.1038/nature03462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pasquinelli AE, Reinhart BJ, Slack F, Martindale MQ, Kuroda MI, Maller B, Hayward DC, Ball EE, Degnan B, Muller P, et al. Conservation of the sequence and temporal expression of let-7 heterochronic regulatory RNA. Nature. 2000;408:86–89. doi: 10.1038/35040556. [DOI] [PubMed] [Google Scholar]
- Patrick DM, Zhang CC, Tao Y, Yao H, Qi X, Schwartz RJ, Jun-Shen Huang L, Olson EN. Defective erythroid differentiation in miR-451 mutant mice mediated by 14-3-3zeta. Genes Dev. 2010;24:1614–1619. doi: 10.1101/gad.1942810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pedersen ME, Snieckute G, Kagias K, Nehammer C, Multhaupt HA, Couchman JR, Pocock R. An epidermal microRNA regulates neuronal migration through control of the cellular glycosylation state. Science. 2013;341:1404–1408. doi: 10.1126/science.1242528. [DOI] [PubMed] [Google Scholar]
- Peng L, Zhang H, Hao Y, Xu F, Yang J, Zhang R, Lu G, Zheng Z, Cui M, Qi CF, et al. Reprogramming macrophage orientation by microRNA 146b targeting transcription factor IRF5. EBioMedicine. 2016;14:83–96. doi: 10.1016/j.ebiom.2016.10.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Penzkofer D, Bonauer A, Fischer A, Tups A, Brandes RP, Zeiher AM, Dimmeler S. Phenotypic characterization of miR-92a-/- mice reveals an important function of miR-92a in skeletal development. PLoS One. 2014;9:e101153. doi: 10.1371/journal.pone.0101153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pfeffer S, Sewer A, Lagos-Quintana M, Sheridan R, Sander C, Grasser FA, van Dyk LF, Ho CK, Shuman S, Chien M, et al. Identification of microRNAs of the herpesvirus family. Nat Methods. 2005;2:269–276. doi: 10.1038/nmeth746. [DOI] [PubMed] [Google Scholar]
- Piwecka M, Glazar P, Hernandez-Miranda LR, Memczak S, Wolf SA, Rybak-Wolf A, Filipchyk A, Klironomos F, Cerda Jara CA, Fenske P, et al. Loss of a mammalian circular RNA locus causes miRNA deregulation and affects brain function. Science. 2017;357 doi: 10.1126/science.aam8526. [DOI] [PubMed] [Google Scholar]
- Poy MN, Hausser J, Trajkovski M, Braun M, Collins S, Rorsman P, Zavolan M, Stoffel M. miR-375 maintains normal pancreatic alpha- and beta-cell mass. Proc Natl Acad Sci U S A. 2009;106:5813–5818. doi: 10.1073/pnas.0810550106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pua HH, Steiner DF, Patel S, Gonzalez JR, Ortiz-Carpena JF, Kageyama R, Chiou NT, Gallman A, de Kouchkovsky D, Jeker LT, et al. MicroRNAs 24 and 27 suppress allergic inflammation and target a network of regulators of T helper 2 cell-associated cytokine production. Immunity. 2016;44:821–832. doi: 10.1016/j.immuni.2016.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qayum AA, Paranjape A, Abebayehu D, Kolawole EM, Haque TT, McLeod JJ, Spence AJ, Caslin HL, Taruselli MT, Chumanevich AP, et al. IL-10-induced miR-155 targets SOCS1 To enhance IgE-mediated mast cell function. Journal of immunology. 2016;196:4457–4467. doi: 10.4049/jimmunol.1502240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quevillon Huberdeau M, Zeitler DM, Hauptmann J, Bruckmann A, Fressigne L, Danner J, Piquet S, Strieder N, Engelmann JC, Jannot G, et al. Phosphorylation of Argonaute proteins affects mRNA binding and is essential for microRNA-guided gene silencing in vivo. EMBO J. 2017;36:2088–2106. doi: 10.15252/embj.201696386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Radzikinas K, Aven L, Jiang Z, Tran T, Paez-Cortez J, Boppidi K, Lu J, Fine A, Ai X. A Shh/miR-206/BDNF cascade coordinates innervation and formation of airway smooth muscle. The Journal of neuroscience : the official journal of the Society for Neuroscience. 2011;31:15407–15415. doi: 10.1523/JNEUROSCI.2745-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rakoczy J, Fernandez-Valverde SL, Glazov EA, Wainwright EN, Sato T, Takada S, Combes AN, Korbie DJ, Miller D, Grimmond SM, et al. MicroRNAs-140-5p/140-3p modulate Leydig cell numbers in the developing mouse testis. Biology of reproduction. 2013;88:143. doi: 10.1095/biolreprod.113.107607. [DOI] [PubMed] [Google Scholar]
- Rasmussen KD, Simmini S, Abreu-Goodger C, Bartonicek N, Di Giacomo M, Bilbao-Cortes D, Horos R, Von Lindern M, Enright AJ, O’Carroll D. The miR-144/451 locus is required for erythroid homeostasis. The Journal of experimental medicine. 2010;207:1351–1358. doi: 10.1084/jem.20100458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rehwinkel J, Behm-Ansmant I, Gatfield D, Izaurralde E. A crucial role for GW182 and the DCP1:DCP2 decapping complex in miRNA-mediated gene silencing. RNA. 2005;11:1640–1647. doi: 10.1261/rna.2191905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reimao-Pinto MM, Ignatova V, Burkard TR, Hung JH, Manzenreither RA, Sowemimo I, Herzog VA, Reichholf B, Farina-Lopez S, Ameres SL. Uridylation of RNA hairpins by Tailor confines the emergence of microRNAs in Drosophila. Mol Cell. 2015;59:203–216. doi: 10.1016/j.molcel.2015.05.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reinhart BJ, Slack FJ, Basson M, Pasquinelli AE, Bettinger JC, Rougvie AE, Horvitz HR, Ruvkun G. The 21-nucleotide let-7 RNA regulates developmental timing in Caenorhabditis elegans. Nature. 2000;403:901–906. doi: 10.1038/35002607. [DOI] [PubMed] [Google Scholar]
- Remenyi J, van den Bosch MW, Palygin O, Mistry RB, McKenzie C, Macdonald A, Hutvagner G, Arthur JS, Frenguelli BG, Pankratov Y. miR-132/212 knockout mice reveal roles for these miRNAs in regulating cortical synaptic transmission and plasticity. PLoS One. 2013;8:e62509. doi: 10.1371/journal.pone.0062509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riemondy K, Wang XJ, Torchia EC, Roop DR, Yi R. MicroRNA-203 represses selection and expansion of oncogenic Hras transformed tumor initiating cells. eLife. 2015;4 doi: 10.7554/eLife.07004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rissland OS, Hong SJ, Bartel DP. MicroRNA destabilization enables dynamic regulation of the miR-16 family in response to cell-cycle changes. Mol Cell. 2011;43:993–1004. doi: 10.1016/j.molcel.2011.08.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rivkin N, Chapnik E, Mildner A, Barshtein G, Porat Z, Kartvelishvily E, Dadosh T, Birger Y, Amir G, Yedgar S, et al. Erythrocyte survival is controlled by microRNA-142. Haematologica. 2017;102:676–685. doi: 10.3324/haematol.2016.156109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodriguez A, Vigorito E, Clare S, Warren MV, Couttet P, Soond DR, van Dongen S, Grocock RJ, Das PP, Miska EA, et al. Requirement of bic/microRNA-155 for normal immune function. Science. 2007;316:608–611. doi: 10.1126/science.1139253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodriguez-Barrueco R, Nekritz EA, Bertucci F, Yu J, Sanchez-Garcia F, Zeleke TZ, Gorbatenko A, Birnbaum D, Ezhkova E, Cordon-Cardo C, et al. miR-424(322)/503 is a breast cancer tumor suppressor whose loss promotes resistance to chemotherapy. Genes Dev. 2017;31:553–566. doi: 10.1101/gad.292318.116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rowe RG, Wang LD, Coma S, Han A, Mathieu R, Pearson DS, Ross S, Sousa P, Nguyen PT, Rodriguez A, et al. Developmental regulation of myeloerythroid progenitor function by the Lin28b-let-7-Hmga2 axis. The Journal of experimental medicine. 2016;213:1497–1512. doi: 10.1084/jem.20151912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruby JG, Jan C, Player C, Axtell MJ, Lee W, Nusbaum C, Ge H, Bartel DP. Large-scale sequencing reveals 21U-RNAs and additional microRNAs and endogenous siRNAs in C. elegans. Cell. 2006;127:1193–1207. doi: 10.1016/j.cell.2006.10.040. [DOI] [PubMed] [Google Scholar]
- Ruby JG, Jan CH, Bartel DP. Intronic microRNA precursors that bypass Drosha processing. Nature. 2007a;448:83–86. doi: 10.1038/nature05983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruby JG, Stark A, Johnston WK, Kellis M, Bartel DP, Lai EC. Evolution, biogenesis, expression, and target predictions of a substantially expanded set of Drosophila microRNAs. Genome Res. 2007b;17:1850–1864. doi: 10.1101/gr.6597907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sachdeva M, Mito JK, Lee CL, Zhang M, Li Z, Dodd RD, Cason D, Luo L, Ma Y, Van Mater D, et al. MicroRNA-182 drives metastasis of primary sarcomas by targeting multiple genes. The Journal of clinical investigation. 2014;124:4305–4319. doi: 10.1172/JCI77116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saetrom P, Heale BS, Snove O, Jr, Aagaard L, Alluin J, Rossi JJ. Distance constraints between microRNA target sites dictate efficacy and cooperativity. Nucleic Acids Res. 2007;35:2333–2342. doi: 10.1093/nar/gkm133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salmena L, Poliseno L, Tay Y, Kats L, Pandolfi PP. A ceRNA hypothesis: the Rosetta Stone of a hidden RNA language? Cell. 2011;146:353–358. doi: 10.1016/j.cell.2011.07.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salomon WE, Jolly SM, Moore MJ, Zamore PD, Serebrov V. Single-molecule imaging reveals that Argonaute reshapes the binding properties of its nucleic acid guides. Cell. 2015;162:84–95. doi: 10.1016/j.cell.2015.06.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salunkhe VA, Esguerra JL, Ofori JK, Mollet IG, Braun M, Stoffel M, Wendt A, Eliasson L. Modulation of microRNA-375 expression alters voltage-gated Na(+) channel properties and exocytosis in insulin-secreting cells. Acta physiologica. 2015;213:882–892. doi: 10.1111/apha.12460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanuki R, Onishi A, Koike C, Muramatsu R, Watanabe S, Muranishi Y, Irie S, Uneo S, Koyasu T, Matsui R, et al. miR-124a is required for hippocampal axogenesis and retinal cone survival through Lhx2 suppression. Nature neuroscience. 2011;14:1125–1134. doi: 10.1038/nn.2897. [DOI] [PubMed] [Google Scholar]
- Sarkies P, Selkirk ME, Jones JT, Blok V, Boothby T, Goldstein B, Hanelt B, Ardila-Garcia A, Fast NM, Schiffer PM, et al. Ancient and novel small RNA pathways compensate for the loss of piRNAs in multiple independent nematode lineages. PLoS Biol. 2015;13:e1002061. doi: 10.1371/journal.pbio.1002061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sassi Y, Avramopoulos P, Ramanujam D, Gruter L, Werfel S, Giosele S, Brunner AD, Esfandyari D, Papadopoulou AS, De Strooper B, et al. Cardiac myocyte miR-29 promotes pathological remodeling of the heart by activating Wnt signaling. Nature communications. 2017;8:1614. doi: 10.1038/s41467-017-01737-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schirle NT, MacRae IJ. The crystal structure of human Argonaute2. Science. 2012;336:1037–1040. doi: 10.1126/science.1221551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schirle NT, Sheu-Gruttadauria J, Chandradoss SD, Joo C, MacRae IJ. Water-mediated recognition of t1-adenosine anchors Argonaute2 to microRNA targets. eLife. 2015;4 doi: 10.7554/eLife.07646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schirle NT, Sheu-Gruttadauria J, MacRae IJ. Structural basis for microRNA targeting. Science. 2014;346:608–613. doi: 10.1126/science.1258040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmiedel JM, Klemm SL, Zheng Y, Sahay A, Bluthgen N, Marks DS, van Oudenaarden A. MicroRNA control of protein expression noise. Science. 2015;348:128–132. doi: 10.1126/science.aaa1738. [DOI] [PubMed] [Google Scholar]
- Schnall-Levin M, Zhao Y, Perrimon N, Berger B. Conserved microRNA targeting in Drosophila is as widespread in coding regions as in 3′UTRs. Proc Natl Acad Sci U S A. 2010;107:15751–15756. doi: 10.1073/pnas.1006172107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schober A, Nazari-Jahantigh M, Wei Y, Bidzhekov K, Gremse F, Grommes J, Megens RT, Heyll K, Noels H, Hristov M, et al. MicroRNA-126-5p promotes endothelial proliferation and limits atherosclerosis by suppressing Dlk1. Nature medicine. 2014;20:368–376. doi: 10.1038/nm.3487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwarz DS, Hutvagner G, Du T, Xu Z, Aronin N, Zamore PD. Asymmetry in the assembly of the RNAi enzyme complex. Cell. 2003;115:199–208. doi: 10.1016/s0092-8674(03)00759-1. [DOI] [PubMed] [Google Scholar]
- Sekita Y, Wagatsuma H, Nakamura K, Ono R, Kagami M, Wakisaka N, Hino T, Suzuki-Migishima R, Kohda T, Ogura A, et al. Role of retrotransposon-derived imprinted gene, Rtl1, in the feto-maternal interface of mouse placenta. Nat Genet. 2008;40:243–248. doi: 10.1038/ng.2007.51. [DOI] [PubMed] [Google Scholar]
- Selbach M, Schwanhausser B, Thierfelder N, Fang Z, Khanin R, Rajewsky N. Widespread changes in protein synthesis induced by microRNAs. Nature. 2008;455:58–63. doi: 10.1038/nature07228. [DOI] [PubMed] [Google Scholar]
- Shabalina SA, Koonin EV. Origins and evolution of eukaryotic RNA interference. Trends in ecology & evolution. 2008;23:578–587. doi: 10.1016/j.tree.2008.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shibata M, Nakao H, Kiyonari H, Abe T, Aizawa S. MicroRNA-9 regulates neurogenesis in mouse telencephalon by targeting multiple transcription factors. The Journal of neuroscience : the official journal of the Society for Neuroscience. 2011;31:3407–3422. doi: 10.1523/JNEUROSCI.5085-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shin C, Nam JW, Farh KK, Chiang HR, Shkumatava A, Bartel DP. Expanding the microRNA targeting code: functional sites with centered pairing. Mol Cell. 2010;38:789–802. doi: 10.1016/j.molcel.2010.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simion P, Philippe H, Baurain D, Jager M, Richter DJ, Di Franco A, Roure B, Satoh N, Queinnec E, Ereskovsky A, et al. A large and consistent phylogenomic dataset supports sponges as the sister group to all other animals. Curr Biol. 2017;27:958–967. doi: 10.1016/j.cub.2017.02.031. [DOI] [PubMed] [Google Scholar]
- Smith KM, Guerau-de-Arellano M, Costinean S, Williams JL, Bottoni A, Mavrikis Cox G, Satoskar AR, Croce CM, Racke MK, Lovett-Racke AE, et al. miR-29ab1 deficiency identifies a negative feedback loop controlling Th1 bias that is dysregulated in multiple sclerosis. Journal of immunology. 2012;189:1567–1576. doi: 10.4049/jimmunol.1103171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith NL, Wissink EM, Grimson A, Rudd BD. miR-150 regulates differentiation and cytolytic effector function in CD8+ T cells. Scientific reports. 2015;5:16399. doi: 10.1038/srep16399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song R, Walentek P, Sponer N, Klimke A, Lee JS, Dixon G, Harland R, Wan Y, Lishko P, Lize M, et al. miR-34/449 miRNAs are required for motile ciliogenesis by repressing cp110. Nature. 2014;510:115–120. doi: 10.1038/nature13413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stadthagen G, Tehler D, Hoyland-Kroghsbo NM, Wen J, Krogh A, Jensen KT, Santoni-Rugiu E, Engelholm LH, Lund AH. Loss of miR-10a activates lpo and collaborates with activated Wnt signaling in inducing intestinal neoplasia in female mice. PLoS Genet. 2013;9:e1003913. doi: 10.1371/journal.pgen.1003913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stark A, Brennecke J, Bushati N, Russell RB, Cohen SM. Animal microRNAs confer robustness to gene expression and have a significant impact on 3′UTR evolution. Cell. 2005;123:1133–1146. doi: 10.1016/j.cell.2005.11.023. [DOI] [PubMed] [Google Scholar]
- Stark A, Bushati N, Jan CH, Kheradpour P, Hodges E, Brennecke J, Bartel DP, Cohen SM, Kellis M. A single Hox locus in Drosophila produces functional microRNAs from opposite DNA strands. Genes Dev. 2008;22:8–13. doi: 10.1101/gad.1613108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stark A, Kheradpour P, Parts L, Brennecke J, Hodges E, Hannon GJ, Kellis M. Systematic discovery and characterization of fly microRNAs using 12 Drosophila genomes. Genome Res. 2007a;17:1865–1879. doi: 10.1101/gr.6593807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stark A, Lin MF, Kheradpour P, Pedersen JS, Parts L, Carlson JW, Crosby MA, Rasmussen MD, Roy S, Deoras AN, et al. Discovery of functional elements in 12 Drosophila genomes using evolutionary signatures. Nature. 2007b;450:219–232. doi: 10.1038/nature06340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Subtelny AO, Eichhorn SW, Chen GR, Sive H, Bartel DP. Poly(A)-tail profiling reveals an embryonic switch in translational control. Nature. 2014;508:66–71. doi: 10.1038/nature13007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sullivan CS, Grundhoff AT, Tevethia S, Pipas JM, Ganem D. SV40-encoded microRNAs regulate viral gene expression and reduce susceptibility to cytotoxic T cells. Nature. 2005;435:682–686. doi: 10.1038/nature03576. [DOI] [PubMed] [Google Scholar]
- Sullivan RP, Leong JW, Schneider SE, Ireland AR, Berrien-Elliott MM, Singh A, Schappe T, Jewell BA, Sexl V, Fehniger TA. MicroRNA-15/16 antagonizes Myb to control NK cell maturation. Journal of immunology. 2015;195:2806–2817. doi: 10.4049/jimmunol.1500949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun Y, Oravecz-Wilson K, Mathewson N, Wang Y, McEachin R, Liu C, Toubai T, Wu J, Rossi C, Braun T, et al. Mature T cell responses are controlled by microRNA-142. The Journal of clinical investigation. 2015;125:2825–2840. doi: 10.1172/JCI78753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki HI, Katsura A, Yasuda T, Ueno T, Mano H, Sugimoto K, Miyazono K. Small-RNA asymmetry is directly driven by mammalian Argonautes. Nat Struct Mol Biol. 2015;22:512–521. doi: 10.1038/nsmb.3050. [DOI] [PubMed] [Google Scholar]
- Tan CL, Plotkin JL, Veno MT, von Schimmelmann M, Feinberg P, Mann S, Handler A, Kjems J, Surmeier DJ, O’Carroll D, et al. MicroRNA-128 governs neuronal excitability and motor behavior in mice. Science. 2013;342:1254–1258. doi: 10.1126/science.1244193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tao C, Ren H, Xu P, Cheng J, Huang S, Zhou R, Mu Y, Yang S, Qi D, Wang Y, et al. Adipocyte miR-200b/a/429 ablation in mice leads to high-fat-diet-induced obesity. Oncotarget. 2016;7:67796–67807. doi: 10.18632/oncotarget.12080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tatsumi N, Hojo N, Yamada O, Ogawa M, Katsura Y, Kawata S, Morii E, Sakamoto H, Inaba R, Tsuda A, et al. Deficiency in WT1-targeting microRNA-125a leads to myeloid malignancies and urogenital abnormalities. Oncogene. 2016;35:1003–1014. doi: 10.1038/onc.2015.154. [DOI] [PubMed] [Google Scholar]
- Tattikota SG, Rathjen T, McAnulty SJ, Wessels HH, Akerman I, van de Bunt M, Hausser J, Esguerra JL, Musahl A, Pandey AK, et al. Argonaute2 mediates compensatory expansion of the pancreatic beta cell. Cell metabolism. 2014;19:122–134. doi: 10.1016/j.cmet.2013.11.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Teng G, Hakimpour P, Landgraf P, Rice A, Tuschl T, Casellas R, Papavasiliou FN. MicroRNA-155 is a negative regulator of activation-induced cytidine deaminase. Immunity. 2008;28:621–629. doi: 10.1016/j.immuni.2008.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thai TH, Calado DP, Casola S, Ansel KM, Xiao C, Xue Y, Murphy A, Frendewey D, Valenzuela D, Kutok JL, et al. Regulation of the germinal center response by microRNA-155. Science. 2007;316:604–608. doi: 10.1126/science.1141229. [DOI] [PubMed] [Google Scholar]
- Treiber T, Treiber N, Plessmann U, Harlander S, Daiss JL, Eichner N, Lehmann G, Schall K, Urlaub H, Meister G. A compendium of RNA-binding proteins that regulate microRNA biogenesis. Mol Cell. 2017;66:270–284 e213. doi: 10.1016/j.molcel.2017.03.014. [DOI] [PubMed] [Google Scholar]
- Tsai WC, Hsu SD, Hsu CS, Lai TC, Chen SJ, Shen R, Huang Y, Chen HC, Lee CH, Tsai TF, et al. MicroRNA-122 plays a critical role in liver homeostasis and hepatocarcinogenesis. The Journal of clinical investigation. 2012;122:2884–2897. doi: 10.1172/JCI63455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsujimura K, Irie K, Nakashima H, Egashira Y, Fukao Y, Fujiwara M, Itoh M, Uesaka M, Imamura T, Nakahata Y, et al. miR-199a links MeCP2 with mTOR signaling and its dysregulation leads to Rett syndrome phenotypes. Cell reports. 2015;12:1887–1901. doi: 10.1016/j.celrep.2015.08.028. [DOI] [PubMed] [Google Scholar]
- Tuschl T. Expanding small RNA interference. Nat Biotechnol. 2002;20:446–448. doi: 10.1038/nbt0502-446. [DOI] [PubMed] [Google Scholar]
- Tyler DM, Okamura K, Chung WJ, Hagen JW, Berezikov E, Hannon GJ, Lai EC. Functionally distinct regulatory RNAs generated by bidirectional transcription and processing of microRNA loci. Genes Dev. 2008;22:26–36. doi: 10.1101/gad.1615208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ustianenko D, Hrossova D, Potesil D, Chalupnikova K, Hrazdilova K, Pachernik J, Cetkovska K, Uldrijan S, Zdrahal Z, Vanacova S. Mammalian DIS3L2 exoribonuclease targets the uridylated precursors of let-7 miRNAs. RNA. 2013;19:1632–1638. doi: 10.1261/rna.040055.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Nieuwenhuijze A, Dooley J, Humblet-Baron S, Sreenivasan J, Koenders M, Schlenner SM, Linterman M, Liston A. Defective germinal center B-cell response and reduced arthritic pathology in microRNA-29a-deficient mice. Cellular and molecular life sciences: CMLS. 2017;74:2095–2106. doi: 10.1007/s00018-017-2456-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Rooij E, Quiat D, Johnson BA, Sutherland LB, Qi X, Richardson JA, Kelm RJ, Jr, Olson EN. A family of microRNAs encoded by myosin genes governs myosin expression and muscle performance. Dev Cell. 2009;17:662–673. doi: 10.1016/j.devcel.2009.10.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Rooij E, Sutherland LB, Qi X, Richardson JA, Hill J, Olson EN. Control of stress-dependent cardiac growth and gene expression by a microRNA. Science. 2007;316:575–579. doi: 10.1126/science.1139089. [DOI] [PubMed] [Google Scholar]
- Ventura A, Young AG, Winslow MM, Lintault L, Meissner A, Erkeland SJ, Newman J, Bronson RT, Crowley D, Stone JR, et al. Targeted deletion reveals essential and overlapping functions of the miR-17~92 family of miRNA clusters. Cell. 2008;132:875–886. doi: 10.1016/j.cell.2008.02.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vickers KC, Landstreet SR, Levin MG, Shoucri BM, Toth CL, Taylor RC, Palmisano BT, Tabet F, Cui HL, Rye KA, et al. MicroRNA-223 coordinates cholesterol homeostasis. Proc Natl Acad Sci U S A. 2014;111:14518–14523. doi: 10.1073/pnas.1215767111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang D, Zhang Z, O’Loughlin E, Wang L, Fan X, Lai EC, Yi R. MicroRNA-205 controls neonatal expansion of skin stem cells by modulating the PI(3)K pathway. Nat Cell Biol. 2013;15:1153–1163. doi: 10.1038/ncb2827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang H, Flach H, Onizawa M, Wei L, McManus MT, Weiss A. Negative regulation of Hif1a expression and TH17 differentiation by the hypoxia-regulated microRNA miR-210. Nature immunology. 2014;15:393–401. doi: 10.1038/ni.2846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang H, Moyano AL, Ma Z, Deng Y, Lin Y, Zhao C, Zhang L, Jiang M, He X, Ma Z, et al. miR-219 cooperates with miR-338 in myelination and promotes myelin repair in the CNS. Dev Cell. 2017;40:566–582 e565. doi: 10.1016/j.devcel.2017.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang S, Aurora AB, Johnson BA, Qi X, McAnally J, Hill JA, Richardson JA, Bassel-Duby R, Olson EN. The endothelial-specific microRNA miR-126 governs vascular integrity and angiogenesis. Dev Cell. 2008;15:261–271. doi: 10.1016/j.devcel.2008.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y, Juranek S, Li H, Sheng G, Wardle GS, Tuschl T, Patel DJ. Nucleation, propagation and cleavage of target RNAs in Ago silencing complexes. Nature. 2009;461:754–761. doi: 10.1038/nature08434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Z, Brandt S, Medeiros A, Wang S, Wu H, Dent A, Serezani CH. MicroRNA 21 is a homeostatic regulator of macrophage polarization and prevents prostaglandin E2-mediated M2 generation. PLoS One. 2015;10:e0115855. doi: 10.1371/journal.pone.0115855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watanabe T, Sato T, Amano T, Kawamura Y, Kawamura N, Kawaguchi H, Yamashita N, Kurihara H, Nakaoka T. Dnm3os, a non-coding RNA, is required for normal growth and skeletal development in mice. Developmental dynamics: an official publication of the American Association of Anatomists. 2008;237:3738–3748. doi: 10.1002/dvdy.21787. [DOI] [PubMed] [Google Scholar]
- Wee LM, Flores-Jasso CF, Salomon WE, Zamore PD. Argonaute divides its RNA guide into domains with distinct functions and RNA-binding properties. Cell. 2012;151:1055–1067. doi: 10.1016/j.cell.2012.10.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei Y, Peng S, Wu M, Sachidanandam R, Tu Z, Zhang S, Falce C, Sobie EA, Lebeche D, Zhao Y. Multifaceted roles of miR-1s in repressing the fetal gene program in the heart. Cell research. 2014;24:278–292. doi: 10.1038/cr.2014.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wen J, Ladewig E, Shenker S, Mohammed J, Lai EC. Analysis of nearly one thousand mammalian mirtrons reveals novel features of Dicer substrates. PLoS computational biology. 2015;11:e1004441. doi: 10.1371/journal.pcbi.1004441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- White K, Lu Y, Annis S, Hale AE, Chau BN, Dahlman JE, Hemann C, Opotowsky AR, Vargas SO, Rosas I, et al. Genetic and hypoxic alterations of the microRNA-210-ISCU1/2 axis promote iron-sulfur deficiency and pulmonary hypertension. EMBO molecular medicine. 2015;7:695–713. doi: 10.15252/emmm.201404511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wightman B, Ha I, Ruvkun G. Posttranscriptional regulation of the heterochronic gene lin-14 by lin-4 mediates temporal pattern formation in C. elegans. Cell. 1993;75:855–862. doi: 10.1016/0092-8674(93)90530-4. [DOI] [PubMed] [Google Scholar]
- Williams AH, Valdez G, Moresi V, Qi X, McAnally J, Elliott JL, Bassel-Duby R, Sanes JR, Olson EN. MicroRNA-206 delays ALS progression and promotes regeneration of neuromuscular synapses in mice. Science. 2009;326:1549–1554. doi: 10.1126/science.1181046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong SF, Agarwal V, Mansfield JH, Denans N, Schwartz MG, Prosser HM, Pourquie O, Bartel DP, Tabin CJ, McGlinn E. Independent regulation of vertebral number and vertebral identity by microRNA-196 paralogs. Proc Natl Acad Sci USA. 2015;112:E4884–4893. doi: 10.1073/pnas.1512655112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu J, Bao J, Kim M, Yuan S, Tang C, Zheng H, Mastick GS, Xu C, Yan W. Two miRNA clusters, miR-34b/c and miR-449, are essential for normal brain development, motile ciliogenesis, and spermatogenesis. Proc Natl Acad Sci U S A. 2014;111:E2851–2857. doi: 10.1073/pnas.1407777111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu L, Nguyen LH, Zhou K, de Soysa TY, Li L, Miller JB, Tian J, Locker J, Zhang S, Shinoda G, et al. Precise let-7 expression levels balance organ regeneration against tumor suppression. eLife. 2015;4:e09431. doi: 10.7554/eLife.09431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wystub K, Besser J, Bachmann A, Boettger T, Braun T. miR-1/133a clusters cooperatively specify the cardiomyogenic lineage by adjustment of myocardin levels during embryonic heart development. PLoS Genet. 2013;9:e1003793. doi: 10.1371/journal.pgen.1003793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiang L, Chen XJ, Wu KC, Zhang CJ, Zhou GH, Lv JN, Sun LF, Cheng FF, Cai XB, Jin ZB. miR-183/96 plays a pivotal regulatory role in mouse photoreceptor maturation and maintenance. Proc Natl Acad Sci U S A. 2017;114:6376–6381. doi: 10.1073/pnas.1618757114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiao C, Calado DP, Galler G, Thai TH, Patterson HC, Wang J, Rajewsky N, Bender TP, Rajewsky K. MiR-150 controls B cell differentiation by targeting the transcription factor c-Myb. Cell. 2007;131:146–159. doi: 10.1016/j.cell.2007.07.021. [DOI] [PubMed] [Google Scholar]
- Xie M, Li M, Vilborg A, Lee N, Shu MD, Yartseva V, Sestan N, Steitz JA. Mammalian 5′-capped microRNA precursors that generate a single microRNA. Cell. 2013;155:1568–1580. doi: 10.1016/j.cell.2013.11.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xin M, Small EM, Sutherland LB, Qi X, McAnally J, Plato CF, Richardson JA, Bassel-Duby R, Olson EN. MicroRNAs miR-143 and miR-145 modulate cytoskeletal dynamics and responsiveness of smooth muscle cells to injury. Genes Dev. 2009;23:2166–2178. doi: 10.1101/gad.1842409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamada Y, Kosaka K, Miyazawa T, Kurata-Miura K, Yoshida T. miR-142-3p enhances FcepsilonRI-mediated degranulation in mast cells. Biochemical and biophysical research communications. 2014;443:980–986. doi: 10.1016/j.bbrc.2013.12.078. [DOI] [PubMed] [Google Scholar]
- Yang JS, Maurin T, Robine N, Rasmussen KD, Jeffrey KL, Chandwani R, Papapetrou EP, Sadelain M, O’Carroll D, Lai EC. Conserved vertebrate mir-451 provides a platform for Dicer-independent, Ago2-mediated microRNA biogenesis. Proc Natl Acad Sci U S A. 2010;107:15163–15168. doi: 10.1073/pnas.1006432107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang K, Yu B, Cheng C, Cheng T, Yuan B, Li K, Xiao J, Qiu Z, Zhou Y. Mir505-3p regulates axonal development via inhibiting the autophagy pathway by targeting Atg12. Autophagy. 2017;13:1679–1696. doi: 10.1080/15548627.2017.1353841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang R, Ren M, Rui Q, Wang D. A mir-231-regulated protection mechanism against the toxicity of graphene oxide in nematode Caenorhabditis elegans. Scientific reports. 2016;6:32214. doi: 10.1038/srep32214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang S, Cui H, Xie N, Icyuz M, Banerjee S, Antony VB, Abraham E, Thannickal VJ, Liu G. miR-145 regulates myofibroblast differentiation and lung fibrosis. FASEB journal: official publication of the Federation of American Societies for Experimental Biology. 2013;27:2382–2391. doi: 10.1096/fj.12-219493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang W, Chendrimada TP, Wang Q, Higuchi M, Seeburg PH, Shiekhattar R, Nishikura K. Modulation of microRNA processing and expression through RNA editing by ADAR deaminases. Nat Struct Mol Biol. 2006;13:13–21. doi: 10.1038/nsmb1041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yekta S, Shih IH, Bartel DP. MicroRNA-directed cleavage of HOXB8 mRNA. Science. 2004;304:594–596. doi: 10.1126/science.1097434. [DOI] [PubMed] [Google Scholar]
- Yi R, Qin Y, Macara IG, Cullen BR. Exportin-5 mediates the nuclear export of pre-microRNAs and short hairpin RNAs. Genes Dev. 2003;17:3011–3016. doi: 10.1101/gad.1158803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoda M, Cifuentes D, Izumi N, Sakaguchi Y, Suzuki T, Giraldez AJ, Tomari Y. Poly(A)-specific ribonuclease mediates 3′-end trimming of Argonaute2-cleaved precursor microRNAs. Cell reports. 2013;5:715–726. doi: 10.1016/j.celrep.2013.09.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoo KH, Kang K, Feuermann Y, Jang SJ, Robinson GW, Hennighausen L. The STAT5-regulated miR-193b locus restrains mammary stem and progenitor cell activity and alveolar differentiation. Dev Biol. 2014;395:245–254. doi: 10.1016/j.ydbio.2014.09.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu J, Vodyanik MA, Smuga-Otto K, Antosiewicz-Bourget J, Frane JL, Tian S, Nie J, Jonsdottir GA, Ruotti V, Stewart R, et al. Induced pluripotent stem cell lines derived from human somatic cells. Science. 2007;318:1917–1920. doi: 10.1126/science.1151526. [DOI] [PubMed] [Google Scholar]
- Zamudio JR, Kelly TJ, Sharp PA. Argonaute-bound small RNAs from promoter-proximal RNA polymerase II. Cell. 2014;156:920–934. doi: 10.1016/j.cell.2014.01.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zeng Y, Cullen BR. Efficient processing of primary microRNA hairpins by Drosha requires flanking nonstructured RNA sequences. J Biol Chem. 2005;280:27595–27603. doi: 10.1074/jbc.M504714200. [DOI] [PubMed] [Google Scholar]
- Zeng Y, Yi R, Cullen BR. Recognition and cleavage of primary microRNA precursors by the nuclear processing enzyme Drosha. EMBO J. 2005;24:138–148. doi: 10.1038/sj.emboj.7600491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang B, Liu SQ, Li C, Lykken E, Jiang S, Wong E, Gong Z, Tao Z, Zhu B, Wan Y, et al. MicroRNA-23a curbs necrosis during early T cell activation by enforcing intracellular reactive oxygen species equilibrium. Immunity. 2016a;44:568–581. doi: 10.1016/j.immuni.2016.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang D, Li Y, Yao X, Wang H, Zhao L, Jiang H, Yao X, Zhang S, Ye C, Liu W, et al. miR-182 regulates metabolic homeostasis by modulating glucose utilization in muscle. Cell reports. 2016b;16:757–768. doi: 10.1016/j.celrep.2016.06.040. [DOI] [PubMed] [Google Scholar]
- Zhang H, Kolb FA, Jaskiewicz L, Westhof E, Filipowicz W. Single processing center models for human Dicer and bacterial RNase III. Cell. 2004;118:57–68. doi: 10.1016/j.cell.2004.06.017. [DOI] [PubMed] [Google Scholar]
- Zhang L, Ke F, Liu Z, Bai J, Liu J, Yan S, Xu Z, Lou F, Wang H, Zhu H, et al. MicroRNA-31 negatively regulates peripherally derived regulatory T-cell generation by repressing Retinoic Acid-Inducible Protein 3. Nature communications. 2015;6:7639. doi: 10.1038/ncomms8639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao JL, Rao DS, O’Connell RM, Garcia-Flores Y, Baltimore D. MicroRNA-146a acts as a guardian of the quality and longevity of hematopoietic stem cells in mice. eLife. 2013;2:e00537. doi: 10.7554/eLife.00537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao T, Li G, Mi S, Li S, Hannon GJ, Wang XJ, Qi Y. A complex system of small RNAs in the unicellular green alga Chlamydomonas reinhardtii. Genes Dev. 2007;21:1190–1203. doi: 10.1101/gad.1543507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng Q, Zhou L, Mi QS. MicroRNA miR-150 is involved in Valpha14 invariant NKT cell development and function. Journal of immunology. 2012;188:2118–2126. doi: 10.4049/jimmunol.1103342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhuang G, Meng C, Guo X, Cheruku PS, Shi L, Xu H, Li H, Wang G, Evans AR, Safe S, et al. A novel regulator of macrophage activation: miR-223 in obesity-associated adipose tissue inflammation. Circulation. 2012;125:2892–2903. doi: 10.1161/CIRCULATIONAHA.111.087817. [DOI] [PubMed] [Google Scholar]
- Zietara N, Lyszkiewicz M, Witzlau K, Naumann R, Hurwitz R, Langemeier J, Bohne J, Sandrock I, Ballmaier M, Weiss S, et al. Critical role for miR-181a/b-1 in agonist selection of invariant natural killer T cells. Proc Natl Acad Sci U S A. 2013;110:7407–7412. doi: 10.1073/pnas.1221984110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zisoulis DG, Kai ZS, Chang RK, Pasquinelli AE. Autoregulation of microRNA biogenesis by let-7 and Argonaute. Nature. 2012;486:541–544. doi: 10.1038/nature11134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zou F, Mao R, Yang L, Lin S, Lei K, Zheng Y, Ding Y, Zhang P, Cai G, Liang X, et al. Targeted deletion of miR-139-5p activates MAPK, NF-kappaB and STAT3 signaling and promotes intestinal inflammation and colorectal cancer. The FEBS journal. 2016;283:1438–1452. doi: 10.1111/febs.13678. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.