Graphical abstract
Abbreviations: ACE, Angiotensin-converting enzyme; ACEI, ACE inhibitors; AD, Alzheimer's disease; ALI/ARDS, Acute lung injury/Acute respiratory distress syndrome; Ang, Angiotensin; ARBs, Angiotensin receptor blockers; AT1R, Angiotensin II receptor type 1; AT2R, Angiotensin II receptor type 2; Aβ, β-amyloid; BKs, Bradykinins; BR, Bradykinin receptor; COVID-19, Coronavirus disease 2019; CQ/HCQ, Chloroquine/Hydroxychloroquine; ECs, Endothelial cells; ET-1, Endothelin-1; eNOS, endothelial Nitric oxide synthase; fMLP, formyl Methionyl-meucyl-proline; GM-CSF, Granulocyte-macrophage colony-stimulating factor; GPCR, G protein-coupled; GRP, Gastrin-releasing peptide; HO-1, Heme oxygenase-1; IL, Interleukin; MAPK/ERK, Mitogen-activated protein kinases/ Extracellular signal-regulated kinases; MasR, Mas receptor; MCP-1, Monocyte chemoattractant protein-1; MERS, Middle East Respiratory Syndrome; NADPH, Nicotinamide adenine dinucleotide phosphate (reduced form); NEP, Neprilysin; NEPi, Neprilysin inhibitors; NF-kB, Nuclear factor kappa B; NO, Nitric oxide; NPs, Natriuretic peptides; Nrf2, Nuclear factor erythroid 2–related factor 2; NSAIDs, Non steroidal anti-inflammatory drugs; PLCb/IP3, Phospholipase C beta/inositol trisphosphate; PNECs, Pulmonary neuroendocrine cells; PPARs, Peroxisome proliferator activator receptors; RAS, Renin angiotensin system; ROS, Reactive oxygen species; SARS-CoV, Severe acute respiratory syndrome coronavirus; SERMs, Selective estrogen receptor modulators; STZ, Streptozotocin; TGF-β1, Transforming growth factor- β1; TNF, Tumor necrosis factor; VEGF, Vascular endothelial growth factor
Keywords: Coronavirus disease, Neprilysin, fMLP, GRP, Bradykinins, Ang (1–7), Endothelin-1
Abstract
COVID-19 is an ongoing viral pandemic disease that is caused by SARS-CoV2, inducing severe pneumonia in humans. However, several classes of repurposed drugs have been recommended, no specific vaccines or effective therapeutic interventions for COVID-19 are developed till now. Viral dependence on ACE-2, as entry receptors, drove the researchers into RAS impact on COVID-19 pathogenesis. Several evidences have pointed at Neprilysin (NEP) as one of pulmonary RAS components. Considering the protective effect of NEP against pulmonary inflammatory reactions and fibrosis, it is suggested to direct the future efforts towards its potential role in COVID-19 pathophysiology. Thus, the review aimed to shed light on the potential beneficial effects of NEP pathways as a novel target for COVID-19 therapy by summarizing its possible molecular mechanisms. Additional experimental and clinical studies explaining more the relationships between NEP and COVID-19 will greatly benefit in designing the future treatment approaches.
1. Introduction
Coronavirus disease 2019 (COVID-19) is an infectious viral disease that is caused by a newly discovered coronavirus, namely severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) [1]. It is a virus belonging to order Nidovirales of family Coronaviridae, which are single-stranded RNA viruses [2] and broadly distributed in both humans and other mammals [3].
Majority of the people infected with 2019 novel coronavirus (2019-nCoV) may recover without requiring special treatment [4], except the elderly people and those with some medical problems such as hypertension, diabetes [5] and chronic respiratory disease [6]. Such patients are more liable to be infected with COVID-19 and might develop a fatal pneumonia with clinical presentation greatly resembling that caused by the previous beta-coronavirus SARS-COV appeared in 2003; severe acute respiratory syndrome (SARS) [7].
In such cohort, patients firstly complain of fever, dry cough, and at late stage, may suffer from dyspnea that makes them in a desperate need of intensive care unit (ICU) admission and oxygen therapy [8]. However, the time between ICU admission and ARDS development is recorded to be as short as 2 days [9], being the reason for the high mortality rate associated with COVID-19 at this stage [10].
Till present, no COVID-19-specific vaccines or medications are available, although some national medical authorities recommend testing the efficacy of antiviral medication in especially sever clinical trials [11]. In addition, several medical researchers suggest starting the supportive and symptomatic treatment till the new medications are developed [12], [13].
Early evidences have shown that the pathogenicity for COVID-19 may be enhanced with increased the body susceptibility to induce a phenomenon called cytokine storm, in which the immune system gears up by overreacting and producing more inflammatory mediators to fight the infection [14]. As a result, a severe inflammatory reaction is produced accompanied with significant damage, including organ failure [15], [16].
In view of cytokines storm caused by COVID-19 infection, high amounts of proinflammatory cytokines (e.g. IL-1β, IL-6, TNF-α, VEGF, GM-CSF and MCP-1) are detected [10], probably leading to associated pulmonary inflammation that can drive fibrosis in the injured tissues [17].
In addition to the dysregulated lung inflammation, emerging research and clinical studies increasingly indicate an alternative point of view for COVID-19 pathogenesis. They suggest that extreme lung tissue injury in COVID-19 may be attributed to vascular endothelial dysfunction, associated with subsequent activation, accumulation of platelets, and then, thrombus formation at the injured site [18], [19], developing a stroke which is suggested to be the leading cause of death in-between sever COVID-19 cases [20].
Lymphocytopenia, a common striking finding in acute infections, is suggested to be a significant indicator of COVID-19 severity [16]. However, this decrease in lymphocytes count is noticed in both severe and non-severe COVID-19 patients [21], expecting that there may be another critical factor driving the development and deterioration of the disease. The most supportive observation now is the disclosure of higher neutrophil–lymphocyte-ratio (NLR) in COVID-19 severe patients than others [22]. Moreover, COVID-19 patients’ laboratory examinations recently show thrombocytopenia, indicating the association of platelet-to-lymphocyte ratio (PLR) with COVID-19 progression and prognosis [23].
Given that the pulmonary tissue damage and microvascular injury recorded in COVID-19 patients is mainly caused by uncontrolled inflammatory reactions and its associated pro-thrombotic conditions, inhibiting the release of underlying inflammatory cytokines may be of great importance [24]. Hence, many studies targeted the utilization of some immunomodulatory agents including hydroxy chloroquine and corticosteroids as COVID-19 therapies to minimize the disease severity [22]. However, that therapeutic direction may interfere with the body immune system to efficiently act as a defense weapon against any other infections [25], [26].
Simultaneously, many studies emphasized on the exploitation of COVID-19 virus to the angiotensin converting enzyme-2 (ACE-2) found on the host pulmonary system as entry receptors [27]; disrupting, of course, the normal physiological function of ACE-2/Ang (1–7)/MasR axis of the renin–angiotensin system (RAS) pathway [28].
Therefore, researchers suggested that the use of angiotensin converting enzyme inhibitors (ACEIs) and/or angiotensin receptor blockers (ARBs), may show a positive trend towards the severe inflammatory reactions and endothelial dysfunction caused by stimulating the function of ACE/Ang II/AT-1 axis and thereby, towards the bad pulmonary effects associated with the COVID-19 infection [29], [30]. However, no clinical trial, till this time, has been proved its efficacy in preventing or even reducing the COVID-19 severity [31], [32].
Pursuing clinical trials with the absence of whole data describing the structure of COVID-19 virus and its related mechanisms may lead to unsatisfied outcomes. As well, understanding such data will greatly help in determining the most appropriate effective treatment protocols. Consequently, the first step to prevent and slow down the disease progression should involve the deduction of proper pathophysiology of that novel viral disease.
Identifying ACE-2 as a viral entry receptor will emphasize on the important role of classical RAS pathway in COVID-19 pathophysiology, however, neither the link between ACE-2 and other RAS components nor their exact roles in the COVID-19 pathogenesis have been neatly studied till now.
One RAS component, namely neprilysin (NEP), has been established to potentiate the physiological beneficial role ascribed to the ACE-2/ang (1–7)/MasR axis; by an alternative pathway [33]. Nowadays, NEP has emerged as a pharmaceutical target for many drugs; especially those used in treating cardiovascular diseases and Alzheimer's disease (AD) [34], [35].
Considering the respiratory system, NEP was reported to play a vital role in protecting lungs from inflammation and fibrosis [36], [37], [38]. A long time ago, Borson et al.,1989 mentioned that rats infected with common respiratory tract pathogens, such as parainfluenza virus type-1, rat coronavirus, and Mycoplasma pulmonis showed low NEP activity; resulting in an increase in their susceptibility to inflammatory responses [39]. However, the precise NEP's role against life-threatening lung injuries has not been investigated yet.
Consequently, this current review aims to examine the cellular signaling pathways involving NEP in COVID-19 pathogenesis and to summarize the evidences supporting the potential beneficial effects of NEP as a novel target for therapy.
2. COVID-19 virus structure and disease pathophysiology
All previous and recent studies agreed with the fact that the surface of all human pathogenic coronaviruses is covered with crown like projections called spike (S) glycoproteins; giving the viruses their name [40]. In addition to S protein, another three main structural proteins have been recognized in SARS-CoV-2, namely envelope (E), nucleocapsid (N) and membrane (M) proteins [41], Fig. 1 .
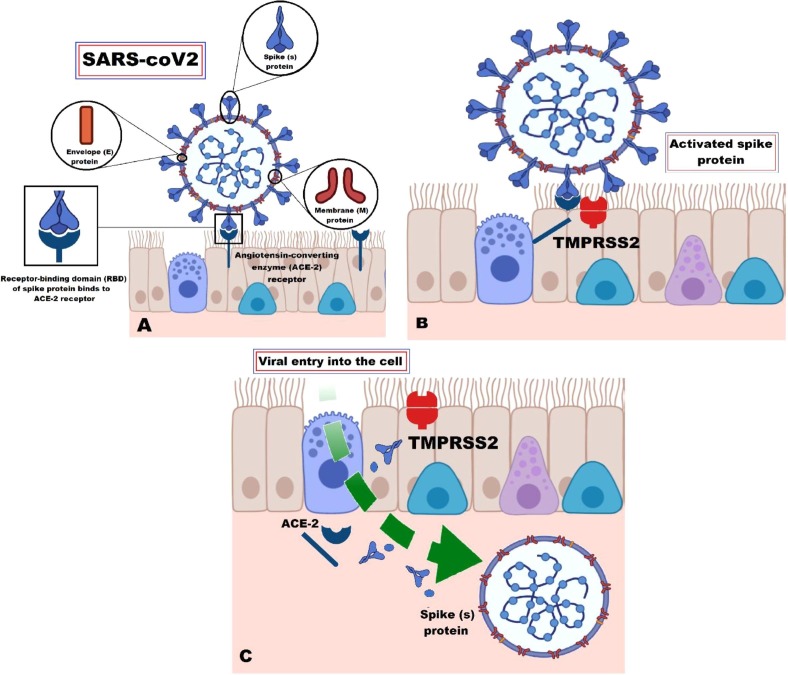
Fig. 1.
A schematic representation of COVID-19 virus structure and disease pathophysiology. SARS-CoV-2 virus is one of coronaviruses that is covered with crown like projections called spike (S) glycoproteins and other three structural proteins; namely envelope (E), nucleocapsid (N) and membrane (M) proteins. SARS-CoV-2 can start infection by (A) binding S proteins functional subunits to the angiotensin converting enzyme-2 receptors on host cell surface; (B) activating transmembrane protease serine 2; followed by (C) cleavage of virus’s S1 subunits from its S2 subunits and so, of the angiotensin converting enzyme-2 receptors; facilitating the virus-cell fusion. ACE-2 = Angiotensin converting enzyme-2; SARS-CoV-2 = Severe acute respiratory syndrome coronavirus-2; TMPRSS2 = Transmembrane protease serine 2.
Specifically, S proteins are very important for SARS-CoV-2 infection to get started [42]. They possess two functional subunits by which the virus can enter into its target host cells involving; S1 subunit, which enables it to bind with receptors on the host cell surface and S2 subunit, that allows the virus to fuse into the cellular membrane [43].
Unlike other coronaviruses, several experimental analyses proved that SARS-CoV-2 does not use the common known viral entry receptors, such as aminopeptidase N (APN) [41] and dipeptidyl peptidase 4 (DPP4) [44], but, instead, can utilize ACE-2 receptor [27].
Additionally, it was revealed that transmembrane protease serine 2 (TMPRSS2) is very critical for the host cell entry of SARS-CoV-2 [45] and its plasma membrane fusion [46] resembling SARS-CoV [47]. Firstly, the virus starts its destructive journey with binding of S1 subunit to ACE-2 enzyme receptor on the cell membrane surface, which in turn activates TMPRSS2. Activation of TMPRSS2 is followed by cleavage of both virus’s S1 subunits from its S2 subunits and so of the ACE-2 receptors. Secondly, activated TMPRSS2 can also act on the S2 subunit, activating it through prompting an irreversible conformational change that will facilitate the virus-cell fusion [48], [49].
Within this view, understanding the precise involvement ACE-2, as a member of pulmonary RAS pathway, in COVID-19 pathophysiology may open new therapeutic possibilities for management.
3. Pulmonary Renin–Angiotensin system (RAS) and COVID-19
3.1. RAS overview
For years, RAS was depicted as a hormonal circulating system involved in fluid and electrolyte balance, systemic vascular resistance and blood pressure regulation [50], [51]. However, a wealth of data showed that lung epithelial cells, fibroblasts and alveolar macrophages could also express the major constituents of the RAS [52], [53] supporting the existence of a “local” pulmonary RAS that can be differentiated from the “systemic” circulating RAS [54].
As regards “local” RAS in the lung, it has been reported to involve multiple cellular mechanisms affecting the vascular permeability, fibroblast activity and alveolar epithelial cells [55], [56]. By the time, it is well recognized that activating pulmonary RAS can influence the pathogenesis of lung injury in different lung diseases such as pulmonary hypertension, acute respiratory distress syndrome (ARDS), asthma and pulmonary fibrosis [54]. As a consequence, pulmonary RAS has been described to participate in disease process or play a protective role against tissue injury [57] and thus, modulating RAS in the lung can successfully play a good role in the treatment of inflammatory lung disease [52], [58].
The functional steps of RAS was firstly initiated by hepatic synthesis of a plasma globulin called renin substrate (or angiotensinogen) which could be enzymatically converted through renin secreted by juxtaglomerular cells of the kidney, into the biologically inactive decapeptide, namely angiotensin (Ang) I [59]. Subsequently, RAS exhibits two main axes based on two distinct enzymes responsible for cleavage of Ang I into angiotensin (Ang) II or Ang 1–7 [51], Fig. 2 .
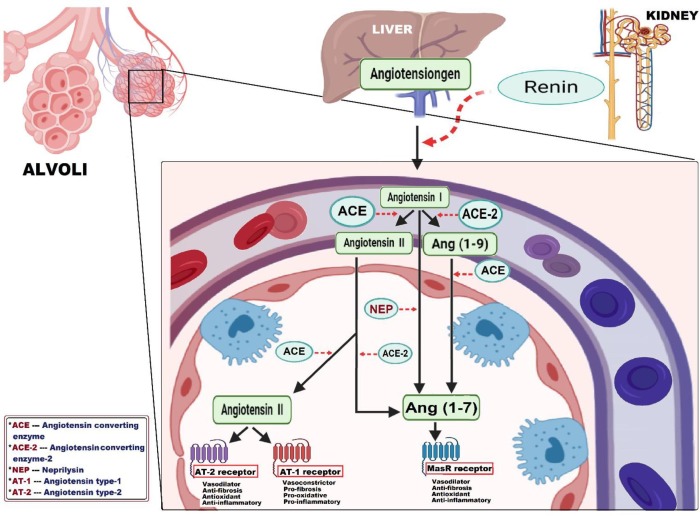
Fig. 2.
A schematic diagram of pulmonary renin-angiotensin system (RAS) cascade. The pulmonary RAS cascade is initiated by converting angiotensinogen (synthesized in liver) through renin (secreted by juxtaglomerular cells of the kidney) into the biologically inactive angiotensin I. Based on angiotensin I degradation, RAS exhibits two main counterregulatory axes: the classical vasopressor arm; named ACE/ Ang II/ AT-1 axis, involves the conversion of angiotensin I by angiotensin converting enzyme into angiotensin II. Angiotensin II acts via angiotensin II type 1 receptor to induce vasoconstriction, fibrotic, oxidative and inflammatory reactions, which can be counteracted via stimulating angiotensin II type 2 receptor. The second depressor arm; named ACE-2 /Ang (1–7)/MasR axis, competes for hydrolyzing angiotensin I into angiotensin (1–9) by angiotensin converting enzyme-2 and then, into angiotensin (1–7) by angiotensin converting enzyme, in addition to neprilysin that directly produce angiotensin (1–7) from angiotensin I. To the same extent, angiotensin converting enzyme-2 can also degrade angiotensin II into angiotensin (1–7), that acts via MasR receptor to produce vasodilatation, anti-fibrotic, anti-oxidative and anti-inflammatory reactions. ACE = Angiotensin converting enzyme; ACE-2 = Angiotensin converting enzyme-2; Ang (1–9) = Angiotensin (1–9); Ang (1–7) = Angiotensin (1–7); AT-1 = Angiotensin II type 1 receptor; AT-2 = Angiotensin II type 2 receptor; NEP = Neprilysin.
First axis involves generation of Ang II as the main effector via the angiotensin converting enzyme (ACE) and named the classical vasopressor axis ACE/ Ang II/ Ang II type 1 receptor (AT1) [60]. For the second axis, angiotensin converting enzyme II (ACE-2) was identified as a depressor for RAS activation through producing Ang 1–7 [61]. Henceforth, this axis ACE-2/ Ang 1–7/ Mas-1 has become an important area of scientific research interest [62].
3.2. ACE/Ang II/AT-1 axis and COVID-19
The ACE/Ang II/AT-1 axis has been well documented to drive a set of deleterious reactions in the lung through generating Ang II, the principal effector of this classical axis, involved in RAS mediated vasoconstriction, sodium retention, fluid overload, inflammation and fibrosis [63], [64]. In addition, ACE has been reported to evoke bradykinin and substance P degradation, two local pro-inflammatory and protussive peptides which can stimulate cough reflex and nitric oxide (NO) release [65], [66].
Specifically, pulmonary vascular inflammation contributes to a phenomenon called ACE “shedding,” in which endothelial surface-bound ACE is released into the interstitium resulting in significant increase in the level of Ang II [67], which explains the detection of higher Ang II level in COVID-19 patients than normal [68].
Several studies declared that Ang II could act through two distinct G protein-coupled receptors (GPCR) subtypes, angiotensin II receptor type 1 (AT1R) and type 2 (AT2R), that were found to be expressed in human lung tissue; proving the local generation of Ang II in lung [65], [69].
The majority of actions evolved by Ang II could be mediated by AT1 receptor via enhancing many complex intracellular signaling pathways including MAPK/ERK, PLCb/IP3/diacylglycerol, tyrosine kinases, and NF-kB [70]. Activating AT1 receptor had been shown to further stimulate monocytes, macrophages and vascular smooth muscle cells to produce the proinflammatory cytokines such as TNF-α and IL-6 [71], [72], [73]. On the contrary, AT2 receptor was documented to have a number of counterregulatory interactions against lung tissue injury via inhibiting inflammation, and fibrosis [74], [75].
In addition to the known Ang II effects on promoting vasoconstriction, and pro-inflammatory cytokine release, there is also an increasing evidence that Ang II could induce vascular endothelial dysfunction [76], [77], promoting the activation and aggregation of platelets and thereby, pro-thrombotic milieu [78].
Ang II could be associated with noticeable rise in the plasma level of endothelin-1 (ET-1) [79], which is a peptide secreted mainly from vascular endothelial cells (ECs), and basically works through the endothelin type A receptor (ETA) in vascular smooth muscle cells and the endothelin type B receptor (ETB) in ECs [80]. Consequently, ET-1 is described as the most potent bronchoconstrictor [81] and vasoconstrictor in the airways [82]. Practically, ET-1 acts as a key player of angiotensin II-induced endothelial dysfunction and platelets activation via inducing IL-6 release [83], [84], which was documented to be correlated directly with the extent of endothelial dysfunction [85].
Endothelial dysfunction is generally characterized by an imbalance between endothelium-dependent relaxing and contracting factors, attributed to reduced production/availability of nitric oxide (NO); namely endothelium-derived relaxing factor (EDRF) [86], [87]. NO acts as a potent endogenous vasodilator that could prevent platelet aggregation and leukocyte adhesion to ECs [88], [89]. Since IL-6 would inactivate endothelial nitric oxide synthase (eNOS), it could disrupt NO production [90], decreasing its level and inducing a state of oxidative stress that may lead to Ang II-induced impairment in endothelial responses [91]
Postulating impaired endothelium functions as a principal factor in the pathogenesis of heart failure, hypertension and diabetes, it will be expected to classify the patients of such diseases as high risk groups for COVID-19 development [92], [93], [94].
Furthermore, platelets activation and aggregation may be noticed as a result of inflammatory reactions aggravated by endothelial dysfunction, leading to imbalance between coagulation and fibrinolysis [87]. It is clearly evident that continuing release of IL-6 can enhance hepatic thrombopoietin (TPO) mRNA expression resulting in thrombopoiesis stimulation and increase in circulatory platelets' numbers, known as (inflammatory thrombocytosis). Within this context, ET-1 can also mediate the synthesis of platelet activating factor (PAF), a potent phospholipid mediator of platelet activation and aggregation, that may activate platelets to stick together and aggregate. As a result, a platelet plug is formed as an initiator for blood clotting and intravascular thrombus formation [88], [89], which is considered as a starting point for developing stroke [95].
Even though the high incidence of inflammatory thrombocytosis in COVID-19 patients, the laboratory results of severe cases showed the opposite; suffering from thrombocytopenia [23].The possible explanation is the depletion of both platelets and megakaryocytes as a result of multiple blood clots formed at the injured site, leading to less platelet production with more consumption as the disease severity increases [96].
Interestingly, endothelial dysfunction and platelet activation can successively together worsen the severity of COVID-19 infection. As known, both ET-1 and activated platelets, associated with endothelial dysfunction, [97] could promote leukocytes rolling, adherence to endothelium, activation and migration into the inflammatory sites, sharing in enhancement of leukocytes recruitment [98], [99]. In addition, endothelial dysfunction can drive the fibrotic consequences following SARS-CoV-2 infection, developing pulmonary fibrosis as a result of releasing transforming growth factor- β1 (TGF-β1), the main fibrogenic cytokines implicated in pulmonary fibrosis, which could be induced by ET-1 action [100], [101].
Taken into consideration the numerous harmful effects possibly induced by Ang II during COVID-19 pathogenesis, we found that most novel studies aim to use the anti-hypertensive drugs which act either by inhibiting the ACE activity or by blocking AT1 receptor, suggesting that action may mitigate the disease severity in COVID-19 patients.
On the other side, it has been proposed that enhancing the counter regulatory axis composed by ACE-2/Ang (1–7)/MasR axis may be the most helpful.
3.3. ACE-2/Ang (1–7)/MasR axis and COVID-19
ACE-2/Ang (1–7)/MasR axis, the depressor arm of RAS, was identified to mitigate the deleterious actions mediated by ACE/ Ang II/ AT1 [51]. Within this axis, ACE-2 competes with ACE by hydrolyzing Ang I into the nonapeptide angiotensin (Ang 1–9) which is further cleaved by the action of ACE into heptapeptide angiotensin (Ang 1–7), thus decreasing the amount of Ang I available for Ang II generation by ACE [102], [103]. To the same extent, ACE-2 could also hydrolyze Ang II converting it into Ang (1–7) [104], [105].
Until the year 2000, ACE has emerged as the key enzyme in the pulmonary RAS, but this was challenged by the discovery of its homologue; ACE-2 [106] which was found out to be broadly expressed in almost all types of lung cells, within the vascular endothelial and smooth muscle cells, types I and II alveolar epithelial cells and bronchial epithelial cells [107], [108].
ACE-2 could negatively regulate the RAS in the lung through reducing Ang II/AT1 receptor signaling and activating the counterregulatory Ang (1–7)/ Mas receptor pathway [54]. That finding was compatible with the animal studies showed that the use of RAS inhibitors could effectively relieve the symptoms of acute severe pneumonia and respiratory failure [109]. Consequently, the increased ACE-2 was addressed as a target for protection from predisposition to inflammatory lung diseases such as, acute respiratory distress syndrome (ARDS) [108], [110].
Concerning COVID-19 pathogenesis, binding of SARS-CoV-2 to ACE-2 resulted in exhaustion of ACE-2, breaking the balance of the RAS system within the lung and then, exacerbation of pulmonary inflammatory reactions [109], [111].
Being ACE-2 receptors widely expressed on vascular endothelial cells (EC) within the lungs [112], SARS-coV-2 can exploit them to induce and diffuse inflammatory cascades within endothelial cells disrupting their function; evidenced by the existence of viral elements and inflammatory cells within endothelial cells [113].
Thus, distributing ACE-2, as the main functional receptor for SARS-CoV-2, within human tissues will be the determinator for spreading of viral infection within the lung and other organs [108], [114]. Both alveolar and bronchial membranes were reported to highly express ACE-2, which may explain the higher tendency of SARS-CoV-2 virus to harmfully affect lower airways than the upper ones [110], [115]. However, recent reports indicate that few COVID-19 patients may also develop some signs and symptoms of upper respiratory tract infection (e.g. rhinorrhea, sneezing, or sore throat) [7], [17], [116].
Furthermore, it was reported that ACE-2 is also expressed in many epithelial cells of other organs than lung including kidney, blood vessels, intestine [110] and brain [117]. The fact which may explain the existence of some extra-pulmonary co-morbidities as myocardial dysfunction [30] and acute kidney injury (AKI) [118], gastrointestinal manifestations (diarrhea, vomiting or abdominal pain) [119], neurologic manifestations (altered mental status or seizures) [120], [121].
Therefore, increased ACE-2 may be useless by promoting viral entry into lung cells, potentiating its devastating effect and enhancing mortality [122]. Consistent with these findings, suggesting ACE-2 activators such as, Diminazene aceturate (DIZE) [65], 4-[2-(4-carbamimidoyl phenyl) imino hydrazinyl] benzene carboximidamide and Xanthenone (XNT) [54] for counteracting COVID-19 pathogenesis will be in vain.
As a consequence, in order to diminish SARS-CoV-2 entry and its subsequent lung injury, pulmonary ACE-2 activity should be reduced. However, at the same time, reduced ACE-2 activity may contribute to worsening of the lung inflammation, by unopposed angiotensin II accumulation [31] and forcing the RAS to continuously increase the expression of cytokines [123].
Simultaneously, a previous data indicated that the decrease in the ACE-2 activity in a rat model of ARDS was paralleled by low amounts of Ang-(1–7) [124], which has been reported to play a beneficial role against pulmonary inflammation and fibrosis [125], [126]. In this context, another study stated that treatment with protease-resistant, a cyclic form of Ang (1–7) (cAng 1–7), could restore the ACE-2 activity; attenuated the inflammatory response; decreased lung injury and improved lung function [124], [127]. That confirms the positive relationship between Ang 1–7 and preserving the depressor role of ACE-2/Ang (1–7)/MasR axis in pulmonary RAS. Hence, these data will attract the attention to the pivotal role of Ang (1–7) in COVID-19 pathophysiology and therapy.
3.4. Ang (1–7) and COVID-19
Ang (1–7) is a biologically active metabolite of the RAS that had become a peptide of interest in the last decade, because of its effective role in activating a number of crucial events for the homeostasis of normal physiological functions [128]. It was reported that Ang (1–7) could exert various effects, which are greatly in opposition to those of AT-1 receptor activation such as vasodilator, anti-inflammatory, anti-hypertrophy, anti-proliferative, anti-fibrosis and antioxidant effects [54], [129].
Clinical and epidemiological studies have revealed the existence of a powerful link between the vasodilator effect of Ang 1–7 and its higher plasma levels in females; making them less liability to hypertension than males [130], [131]. Several mechanisms have been attributed to Ang 1–7 in lowering blood pressure which can be explained as follows (i) Triggering eNOS to stimulate the release of NO, which could play a critical role in promoting the relaxation of blood vessels and inhibiting the platelet aggregation [132], [133] (ii) Inducing natriuresis/diuresis [134], and (iii) Activating peroxisome proliferator activator receptors (PPARs), which in turn supports the availability of NO [135].
Interestingly, Ang 1–7 was documented to directly blunt the activation of pro-inflammatory signaling pathways induced by the Ang II-associated phosphorylation of MAPKs and NF-kB signaling [136], [137], suggesting also the anti-hypertrophic effects of Ang 1–7 through normalization of MAPKs activity [130]. Moreover, Ang (1–7), by counteracting the Ang II effects, could also preserve the endothelial function through increasing nitric oxide bioavailability and inhibiting oxidative stress [138].
Taken together, Ang 1–7 could also reduce lung injury by suppressing the expression of fibrogenic molecules such as TGF-β1[139], which acts as a key mediator involved in pulmonary fibrosis [140].
Moreover, several researches have pointed out the antioxidant role of Ang 1–7 [54], [130] that might be established by; (i) limiting the activation of NADPH oxidase, which is a membrane-bound enzyme complex involved in triggering ROS production through generating superoxide radicals [141], and/or (ii) normalizing the expression of antioxidant enzymes such as catalase and heme oxygenase-1 (HO-1) [130], as well as the nuclear factor erythroid 2–related factor 2 (Nrf2), that acts as an emerging regulator of cellular resistance to oxidants [142].
Other observation and experimental evidence suggested that endogenous ACEIs effects of Ang (1–7) can be mediated by its unique membrane bound G protein-coupled receptor known as Mas (MasR) [143], which was revealed to be found in the thin areas of the bronchial epithelium and smooth muscle [144]. MasR could direct the Ang 1–7 biological responses via the MasR/cAMP/protein kinase A (PKA) signaling [145], which was previously validated by administrating AVE 0991 (a nonpeptide mimetic of Ang 1–7 as a specific ligand (agonist) for MasR [130].
Subsequently, attention should be drawn to specifically trigger the Ang (1–7) as a potential therapeutic agent able to mitigate the lung injury in patients with COVID-19 infection, without increasing ACE-2 activity.
There are different formulations of Ang 1–7 are being developed e.g. AVE-0991 [146], HPβCD/Ang 1–7 [147], CGEN-856 [148], NorLeu3-A [149] and cyclic Ang 1–7 [150] and used to demonstrate its therapeutic potential in numerous animal models of human diseases, including hypertension, heart failure, stroke, diabetes mellitus, atherosclerosis, renal disease and pulmonary arterial hypertension [130], [145], [151]. However, therapeutic attempts and clinical trials are still underway because of Ang (1–7) rapid in vivo degradation by ACE [130].
On the other hand, previous studies emphasized that Ang (1–7) could induce vasodilatation in rats of Mas-deficient vessels [152] and in rats pretreated with A779 (MasR blocker) [153]. The former observed data suggested that Ang (1–7) might also interact with an additional specific receptor other than MasR to elicit vasodilatation [154]. Indeed, Ang (1–7) have been shown to stimulate also the bradykinin (BKs) pathway via preventing the BK hydrolysis [155].
BKs are one of the formed kinins that can play significant roles in regulating tissue injury, inflammatory responses and vascular permeability [156]. They have a little direct impact on the activation and recruitment of inflammatory cells, but they could work indirectly through stimulating the airway epithelial cells and lung fibroblasts through producing a wide array of cytokines, including IL-6, IL-1, IL-8, granulocyte colony stimulating factor (G-CSF), GM-CSF and macrophage chemoattractant protein-1 (MCP-1) [157], [158], [159], [160]. BK action was mediated by G-coupled receptors, namely BK receptor (BR) [156] that amazingly found to be interacted with the Mas receptors in order to regulate the vasodilator effect of the Ang (1–7) [161].
At the same time, it was found that the reduction in ACE-2 mRNA expression within the lungs of STZ diabetic rats was linked with an increase in circulating Ang II, but without any significant change in the production of Ang (1–7) [162], [163], ensuring that pulmonary ACE-2 was not the only enzyme responsible for Ang (1–7) production, but there might be another enzyme contributing to its synthesis.
By the time, the classical view of RAS has been further expanded and become well established than previously conceived. As a consequence, it was discovered that there was another alternative pathway by which Ang (1–7) is produced, instead of that based on ACE-2. That way was disclosed to degrade Ang I directly into Ang (1–7) by another RAS member, called NEP.
3.5. NEP and COVID-19
NEP (neutral endopeptidase or neprilysin, previously known as CD10) is a member of transmembrane zink-metalloendopeptidase that particularly highly expressed in both kidney and lung [164], [165], [166]. NEP was also found in a number of other tissues, as epithelia of breast, prostate, stomach and in the central nervous system [167], [168], [169]. NEP had been also shown to be present in a soluble circulating form (cNEP) within several body fluids including urine, cerebrospinal fluid and plasma [170].
Although NEP can discriminately hydrolyze a broad spectrum of physiologically relevant substrates, it was found to possess an obvious substrate specificity. Classically, NEP exhibits a size-related specificity that enables it to hydrolyze only peptides with a small molecular weight generally at or below 3,000 Dalton [171]. As well, NEP has been also described to show a distinction between substrates being cleaved 'in vitro' and that being cleaved 'in vivo'. Initially, NEP was reported to specifically cleave more than 7 peptides 'in vivo' including natriuretic peptides (NPs) (Atrial natriuretic peptide (ANP), C-type natriuretic peptide (CNP), and B-type natriuretic peptide (BNP)), BKs, neuropeptides (substance P, enkephalins), Gastrin, Chemotactic peptide formyl-Met-Leu-Phe (fMLP), versus 26 peptides 'in vitro' such as IL-lβ, Oxytocin, Gastrin-releasing peptide (GRP), ET-1, Ang I and II……etc [172], [173], [174]. By time, more substrates had been proposed with varying levels of 'in vitro' and/or 'in vivo' proof of cleavage. Among that, GRP, ET-1, Ang I which have been proved to be additionally metabolized 'in vivo' by NEP [162], [175], [176], emerging the evidence that NEP could play an important role in many physiological and pathological conditions [177], Table 1 .
Table 1.
Main evidence about the several pharmacological mechanisms of NEP.
| Drug action | Drug name | Disease | Population | Main effects | References | |
|---|---|---|---|---|---|---|
| Neprilysin (neutral endopeptidase; NEP) | Inhibitors | Racecadotril (acetorphan) | Acute Secretory Diarrhea | Rats, children and adult patients | Reduced the water and electrolytes hypersecretion into intestinal lumen by inhibiting the degradation of endogenous enkephalins in the intestinal epithelium | [181], [272], [273] |
| Sacubitril/ Valsartan (Entresto) | Congestive heart failure (CHF) | Rats and CHF patients | Sacubitril elevates the NPs level, increasing sodium and water excretion in urine, dilating blood vessels, and reducing preload and ventricular remodeling | [182], [274], [275], [276] | ||
| Omapatrilat | Hypertension | Rats and hypertensive patients | Inhibiting the degradation of vasoactive peptides, including NPs and BKs, causing blood vessel dilation and reduction in ECF volume | [277], [278], [279], [280] | ||
| Sacubitril | Diabetes | Mice, rats and diabetic patients | Demonstrated to regulate glucose metabolism by improving glycemia and reducing time to initiation of insulin therapy | [185], [281], [282] | ||
| Agonists | Recombinant soluble sNEP | Alzheimer's disease (AD) | Mice and hippocampal cells HT22 | Reduced the amyloid-beta accumulation and improved memory impairment | [190], [283] | |
| Recombinant neutral endopeptidase (EC 3.4.24.11) | Ulcerative colitis | Mice | Inactivated the expression of proinflammatory neuropeptide Substance P and the adhesion molecule ICAM-1 | [284], [285] | ||
| Recombinant neutral endopeptidase (EC 3.4.24.11) | Lung cancer | Lung cancer cell lines and mice | Degrading the biologically active peptides implicated in the stimulation of lung cancer; inhibiting lung cancer cell growth | [286] | ||
| Recombinant human neutral endopeptidase (rhNEP) | Pancreatic elastase-induced lung injury | Mice | Degraded the proinflammatory mediators (chemokines, endothelin, bradykinin) involved in neutrophil sequestration. | [287] | ||
| Recombinant neutral endopeptidase (EC 3.4.24.11) | Tachykinins -induced cough and dyspnea | Guinea pigs | Inhibiting substance P-induced cough is suggested by high affinity of substance P and other tachykinins to neutral endopeptidase | [288], [289] |
BKs = Bradykinins; ECF = Extracellular fluid; ICAM-1 = Intercellular adhesion molecule-1; NPs = Natriuretic peptides.
For CVS patients, NPs were known to be of therapeutic importance in lowering blood volume by inducing natriuresis, in which excess sodium can be excreted in urine with accompanying water by the renal tubules [35]. Previous clinitcal trials proved that administering synthetic NPs might be associated with some limitations especially on the long run [178], [179], that pushed them to depend on using Neprilysin inhibitors (NEPi) such as (thiorphan or candoxatrilat) to prolong and potentiate the beneficial effects of vasoactive/NPs via inhibiting endogenous NPs degradation [180].
NEPi have been used for decades to treat acute diarrhea [181]. However, they are now developed and emerged as a pharmaceutical target for different CVS diseases. NEPi have been used mainly in people with congestive heart failure and a reduced left ventricular ejection fraction (LVEF) [182]. However, using NEPi (e.g. candoxatril) solely for hypertension treatment was ineffective, because NEPi could inhibit Ang II degradation, increasing its associated simultaneous detrimental effects. Therefore, combining NEPi either with ACEi (e.g. omapatrilat) or ARBs (e.g. LCZ696) become a necessity to overcome this limitation [183], [184].
Another critical aspect for the pharmacological role of NEPi was achieved in treating diabetes via inhibiting the breakdown of some substrates known to modulate glucose metabolism, such as incretin glucagon-like peptide-1 (GLP-1), NPs and BKs [185].
Within the brain, NEP had also proved to hold a great beneficial role in the neurological disorders such as Alzheimer's disease (AD) on both in vitro and in vivo studies [186], [187], [188]. Multiple lines of evidence highlighted the presence of many amyloid plaques in the form of β-amyloid (Aβ) peptide in the brains of AD patients with dementia [189]. It was worthy mentioned that NEP was one of the major Aβ peptide-degrading enzymes in the brain, whose expression was documented to be lower in the brain of AD patients [190] supporting the role that NEP can play in the prevention and treatment of AD.
In addition to the above mentioned, NEP seems to play a protective role in the pathogenesis of lung injury since significant decrease in the NEP enzymatic activity in the lung of mice with ALI was associated with inactivation of the tachykinins degradation pathway and consequently, reducing uncontrolled inflammation in ALI/ARDS and in other neurogenic respiratory diseases [37], [191], [192].
Lung inflammation could induce hyperplasia in the pulmonary neuroendocrine cells (PNECs) of both respiratory bronchioles and alveoli [193], [194], resulting in excessive production of GRP [195].
GRP was known to be one of the Bombesin-like peptides that can be expressed and released by PNECs into the surrounding airway parenchyma in response to various stimuli like hypoxia or irritation [196], [197] to regulate the neutrophil chemotaxis and macrophage infiltration within the lung tissue [198], [199]. GRP could act via stimulating Gastrin-releasing peptide receptor (GRPR) at the surface of macrophages, which in turn, would enhance the release of early inflammatory mediators contributing to the recruitment of neutrophils [200], [201].
More neutrophil infiltration within the lung is usually associated with high tendency for lung tissue damage [202], [203], since they would be involved in breakdown of basement membrane integrity within the bronchiolar/alveolar architecture and thereby, diminution of pulmonary function. Moreover, during the deleterious inflammatory reactions, as in pneumonia, neutrophil lifespan is prolonged to generate more superoxide radicals resulting in damage of the surrounding normal tissue [204].
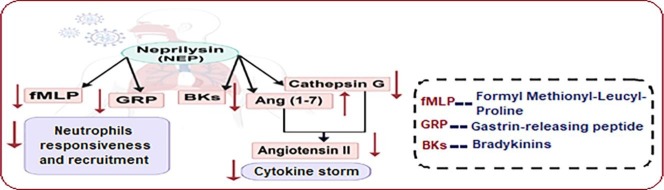
Interestingly, NEP can play a vital role during lung inflammation through its catabolic effect on (GRP) [205] in addition to its presence on the plasma membrane of neutrophils modulating their chemotactic responsiveness via cleaving the chemotactic peptide formyl-Met-Leu-Phe (fMLP) which resembles an effective chemotactic agonist in response to GRP [206], [207]. Thus, breaking both GRB and fMLP peptide by NEP will cut the way for recruiting more neutrophils into site of injury and consequently, grab the reins.
On the other hand, once neutrophils being activated at inflammatory sites, they could secrete high concentration of several serine proteases into the extracellular environment to degrade host pathogens, recruit more cytokines and stimulate further tissue damage [208], [209]. Cathepsin G was reported to be one of the neutrophil-derived serine proteases that is abundantly found in the azurophil granules and known to degrade both Angiotensinogen and Ang I into Ang II [209], [210].
So far, NEP can additionally take a part in decreasing the pro-inflammatory, oxidative and pro-fibrotic effects of Ang II by minimizing the release of cathepsin G and consequently, its action on Ang I [211], [212], [213].
As NEP was reported to have more catalytic activity than ACE-2 in cleaving Ang I into Ang (1–7), it could also effectively enhance the protective activities associated with Ang (1–7) in the lung [214]. As well, NEP could not affect lung Ang (1–7) metabolism because it was involved in the metabolism of Ang (1–7) within tissues other than pulmonary tissues, as renal cortex. On the other hand, ACE was recorded to be the major enzyme responsible for Ang (1–7) metabolism in the pulmonary membranes by hydrolyzing it to Ang (1–5), and then, into Ang (3–5) by the action of aminopeptidases [215]. Besides, NEP shows higher activity than ACE towards BKs degradation, resulting in inhibition of the bradykinin-induced inflammatory cells influx [125], [216].
Despite multiple data confirming the expected role of NEP in relieving the pulmonary inflammatory response, its effect on reducing exacerbation of acute severe pneumonia in COVID 19 patients has not been highlighted yet.
Hence, the question now, may the reviewed actions of NEP pathways be sufficient to impose a new effective strategy for COVID-19 management as compared to the suggested repurposed drugs?
4. Main repurposed drugs for COVID-19
World Health Organization (WHO) and Centers for Disease Control and Prevention (CDC) guidelines only provide supportive care. However, many drugs repurposed based on host response in order to defeat COVID-19, Table 2 [217].
Table 2.
Main repurposed drugs for COVID-19.
| Classes of drug used | Main member (s) | Main molecular mechanisms of action | Use Limitations | References |
|---|---|---|---|---|
| Anti-inflammatory agents | Non-steroidal anti-inflammatory drugs (NSIDs)
|
Inhibit cyclooxygenase enzymes (COX-1/COX-2) that mediate the bioconversion of arachidonic acid into prostaglandins (PGs) which, in turn, control inflammation | Increased risk of stroke and myocardial infarction in acute respiratory tract infections, induced nephrotoxicity in susceptible covid-19 patient groups which is exacerbated by fever and dehydration. | [219], [220], [221] |
Corticosteroids
|
Modulate a variety of the inflammatory cytokines (including IL-1, IL-6, IL-8, IL-12 and TNFα), reduce IFN-γ- (IP-10) and MCP-1, potent mediators of the immune inflammatory response | Inhibit synthesis of anti-inflammatory molecules as lipoxins and resolvins, delaying resolution of inflammation with increase susceptibility for secondary infection, stimulation of hypothalamic-pituitary adrenal axis can also induce lymphocytopenia | [24], [219], [260], [261] | |
Specific anti-cytokines
|
Anakinra is IL-1 receptor antagonist and tocilizumab is IL-6 inhibitor, reducing virally driven hyperinflammation | Anakinra increases risk of infection, mainly pneumonia, induce neutropenia since IL-1 is a neutrophil attractant and growth factor and Tocilizumab reduces CRP levels, masking the clinical symptoms | [10], [22], [262] | |
Anti-inflammatory cytokines
|
Suppress inflammatory activity via IL-1 inhibition that produces other proinflammatory cytokines | Inhibition of gamma-interferon production resulting in more viral burden and early dissemination of viral infections | [17], [263] | |
| Chloroquine (CQ) & Hydroxychloroquine (HCQ) | Anti-inflammatory actions by reducing production of the inflammatory cytokines, might interfere with ACE2 receptor glycosylation, thus preventing SARS-CoV-2 binding to target cells and immunomodulatory benefits by affecting cell signaling in viral infections | Its poisoning has been favored with possible life threatening arrythmias in cardiac patients, retinal toxicity has been described with long term administration of CQ and HCQ, may precipitate liver & renal impairment in susceptible patients | [217], [227], [264], [265] | |
| Estrogens | Estrogens
|
Could suppress TMPRSS2 expression in the lung which is essential for SARS-CoV-2 entry, increase Ang-(1–7) production through ER-α- mediated stimulation of ACE1 and ACE2 expression and activity | Male sexual functions regarding erection, spermatogenesis and libido are adversely affected by estrogen exposure, long-term therapy may precipitate hypertension, cerebrovascular stroke and thromboembolism, increased risk of cervical and breast cancer in high risk patients | [235], [237], [238], [266], [267] |
Selective estrogen receptor modulators (SERMs)
| ||||
| Renin-angiotensin system (RAS) blockers | ACE inhibitors
|
Enhancing the ACE-2/ Ang-(1–7)/ Mas axis to increase the production of ang (1–7) which, in turn, can counteract the activity of ACE/ Ang II/ AT1R axis, ARBs may preserve ACE-2 in competition with SARS-CoV-2 entry into the cell | ACE inhibition suppress kininase-II, which may be followed by accumulation of the protrusive mediators (bradykinin and substance P) in the upper respiratory tract or lungs resulting in dry cough and angioedema in susceptible individuals, ARBs may show upregulation in the membrane bound ACE-2 facilitating the coronavirus entry and worsen then its course | [240], [241], [268], [269] |
Angiotensin II type 1 receptor blockers (ARBs)
| ||||
Direct renin inhibitors (DRI)
|
Competitive inhibition of renin, lower the formation of Ang I, and therefore of Ang II and Ang (1–7) reduction in ACE2 expression | Same adverse effects as other RAS blockers mainly cough, contraindicated in old patients with renal or hepatic impairment | [28], [270], [271] |
ACE = Renin Angiotensin System; ACE-1 = Angiotensin Converting Enzyme 1; ACE-2 = Angiotensin Converting Enzyme 2; Ang-(1–7) = Angiotensin 1–7; COX-1 = Cyclooxygenase-1; COX-2 = Cyclooxygenase-2; CRP = C-reactive protein; ER-α = Estrogen receptors alpha; IFN-γ- (IP-10) = Interferon gamma-Induced Protein 10; IL = Interleukin; MCP-1 = Monocyte chemoattractant protein 1; TMPRSS2 = Transmembrane Protease Serine 2; TNFα = Tumor Necrosis Factor Alpha.
4.1. Anti-inflammatory agents
Recently, there is an empirical use of anti-inflammation therapy in critical patients of COVID-19 presented with severe complications in order to prevent further injury and suppress Cytokine storm manifestations as ARDS and other organs damage till even death. Main anti-inflammatory medications include, non steroidal anti-inflammatory drugs (NSAIDs), corticosteroids, chloroquine and statins [22].
Numerous observational data support a strong association between use of NSAIDs in management of lower respiratory tract infections and higher incidence of complications including fulminating pneumonia, pleural effusions and dissemination of infection [218]. NSAIDs may also induce nephrotoxicity among susceptible covid-19 patient groups and is exacerbated by fever and dehydration [219]. Consequently, some studies recommend avoiding use of NSAIDs e.g. ibuprofen and diclofenac as the first choice for fever control and pain symptoms in COVID-19 infection and using paracetamol instead [220], [221].
Previous study demonstrated that corticosteroids-treated asthmatic patients showed enhanced NEP expression in their airway epithelium as compared to nonsteroid-treated ones. This fits the hypothesis that the anti-inflammatory effect of corticosteroids within the airways may be partially mediated by upregulating NEP [222].
Despite of possible benefits gained from the anti-inflammatory effect of corticosteroids, they will be associated with serious impairment in the immune system of severe COVID-19 cases [22]. Corticosteroids may delay the viral elimination and increase susceptibility for the secondary infection resulting in deterioration of the disease especially with immune system impairment [223]. Other side effects associated with corticosteroid treatment include hyperglycemia, central obesity and hypertension which represent an obstacle against their use in people at higher risk for COVID-19 especially diabetic, cardiac and hypertensive patients [217].
Auyeung et al., 2005 reported that there is no survival benefit for treatment of SARS patients with corticosteroids [224]. Another study on corticosteroid therapy of MERS patients has revealed no mortality difference with delayed clearance of MERS-CoV from lower respiratory tract [225]. Moreover, there is no evidence from any clinical trials supporting its administration for COVID-19 [22].
In order to overcome the immune suppression induced by corticosteroids, a variety of other therapies have been developed to act directly as specific anti-cytokines (e.g. anakinra or tocilizumab) [10] or anti-inflammatory cytokines (e.g. IL-37 or IL-38) [17] without targeting the immune system and have proven to be effective in treating several syndromes that triggered by cytokine storm [123].
According to previous studies, Chloroquine (CQ) and its less toxic metabolite, hydroxychloroquine (HCQ) possess anti-inflammatory and immunomodulatory benefits by affecting cell signaling in viral infections. CQ/HCQ also exhibit a wide variety of antiviral reactions against several viruses including members of the flaviviruses, retroviruses, and coronaviruses [25].
CQ and HCQ can prevent the attachment of viral particles to their cell surface receptor, modulate pH in order to inhibit pH-dependent steps of viral replication or interfere with post-translational modifications (PTMs) of viral proteins [217], [226].
An enough pre-clinical evidence considering CQ effectiveness for treatment of COVID-19 showed reduction in the pneumonia exacerbation, improving lung imaging findings and high rate of virus nucleic acid test negativity. Accordingly, CQ phosphate in Guidelines (version 6) for treatment of COVID-19 has been recommended with oral administration twice daily at a dose of 500 mg (300 mg for chloroquine) for adults and no more than 10 days [22].
However, there is a narrow margin between CQ/HCQ therapeutic and toxic dose. Its poisoning has been favored and life-threatening in patients with cardiac disorders [227]. It is also contraindicated for people with retinopathy, elevated liver enzymes, heart rhythm disorders (as QT prolongation) or allergy to CQ/HCQ [228]. Further, efficiency and safety of CQ/HCQ for COVID-19 is still unclear and needs a confirmation depending on more preclinical and clinical trials [229].
Statins are the widely used cholesterol lowering drugs, that were also reported to improve endothelial functions via lipid-independent mechanisms; mediated by their anti-inflammatory and anti-oxidant properties as well as their ability to restore vascular NO bioavailability [230]. Because of their immunomodulatory effect, they have also proven to be beneficial as adjuvant therapy in patients with different auto-immune inflammatory conditions (e.g. systemic lupus erythematosus, rheumatoid arthritis and multiple sclerosis) [231]. As a consequence, some hospitals included statins in the treatment protocol of COVID-19. Although statin therapy is usually well tolerated, their use in COVID-19 patients may increase the incidence and severity of myopathies and acute kidney injury [232]. Moreover, statin drugs may increase IL-18 levels which can promote severe pneumonia, deteriorating SARS-CoV-2–induced ARDS and mortality, especially in elderly patients who are more likely to use these drugs [233].
4.2. Estrogens and selective estrogen receptor modulators (SERMs)
Sex differences in health outcomes following COVID-19 may be attributed to sex-dependent production of steroid hormones. Estrogen has been reported to attenuate inflammation which might protect women compared to men [234]. Estrogen signaling are also known to downregulate MCP-1 expression and promote adaptive T cell response by stimulating neutrophil recruitment [235].
It has been also reported that SERMs, like Toremifene, exhibits potential effects in blocking various viral infections as SARSCoV and MERSCoV virus [236] through interacting with and destabilizing the virus membrane glycoprotein resulting in inhibition of its replication [237].
Wang et al., 2020 suggests that treatment with estrogens and estrogen-related compounds as estradiol (E2) could suppress the expression of TMPRSS2 in the lung resulting in decreased mortality to SARS-CoV infection [235].
An estrogen receptors/RAS interaction has been demonstrated in several studies. E2 was detected to drive RAS to increase Ang-(1–7) production through estrogen receptor (ER α) mediated stimulation of ACE1 and ACE2 mRNA expression and activity. A sex difference has been revealed in the expression of RAS components [238].
4.3. Renin-angiotensin system (RAS) blockers
There is a controversy about the role of RAS blockers including, ACE inhibitors and/or angiotensin II type 1 receptor blockers (ARBs) in treating COVID-19 and their exact roles still remain unclear [28], [32], [239].
Their use has been suggested to be of a value through increasing ACE-2 that may have a protective effect against virus-induced lung injury by preserving ACE-2 in competition with SARS-CoV-2 entry into the cells [28]. Another mechanism by which ACE-2 can prevent lung damage and attenuate the pulmonary fibrosis, will be via enhancing the ACE-2/Ang-(1–7)/Mas axis to increase the production of ang (1–7), which in turn can counteract the activity of the ACE/AngII/AT1R axis [137].
On the other hand, it was known that ACE inhibitors could block the breakdown of bradykinin increasing its level and then, promoting its associated inflammatory reactions resulting in more deterioration in the health state [240]. Additionally, it has been expected that patients receiving ARBs may show upregulation in the membrane bound ACE-2 facilitating the coronavirus entry and worsen then its course [241].
The suggested explanation may be attributed to the increase in angiotensin II level that probably pushes it to act as an increased substrate loaded on the ACE enzyme, resulting in shifting a part of Ang II to be converted by the action of ACE2 into Ang (1–7), that may be associated with ACE-2 upregulation [32].
Other agents acting on the RAS, such as beta-blockers and direct renin inhibitors (DRI) to lower AngI formation and thereby, AngII and Ang (1–7). However, till now, no one discussed their impact on the severity and prognosis of COVID-19 [28].
On the contrary, lacking Ang (1–7) will be of negative effect on lung health. So, there is an urgent need to suppose a pathway that can ensure the increase of Ang (1–7) level without upregulating ACE-2. That effect may be attained by keeping ACE activity to enhance the pulmonary metabolism of Ang (1–7) and at the same time, shifting the RAS system away from ACE/Ang II/AT-1 axis to avoid its associated inflammatory and oxidative activities. We suggest that NEP may achieve this complicated equation.
5. NEP-dependent strategy for COVID-19 therapy
Based on previous literature that addressed several beneficial and protective effects displayed by NEP during lung injury, we postulate that increasing NEP activity may mitigate COVID-19 pathogenesis. Lung of COVID-19 patients showed pneumocyte hyperplasia with inflammatory cellular infiltration [242], confirming the release of excessive GRP into the surrounding airway parenchyma as a result of PNECs hyperplasia [193], [194], [196], [197].
Considering the documented links between high GRP level and both neutrophil chemotaxis and infiltration as well as reduction in food and water intake [243], it is not surprising to detect high neutrophil count [244] and anorexia in severe COVID-19 patients [245].
Thus, we expect that GRP is the first spark in initiating neutrophils recruitment as well as cytokine storm, which are the main pillars in COVID-19 pathophysiology. Our suggested hypothesis herein depends on two main aspects, Fig. 3 :
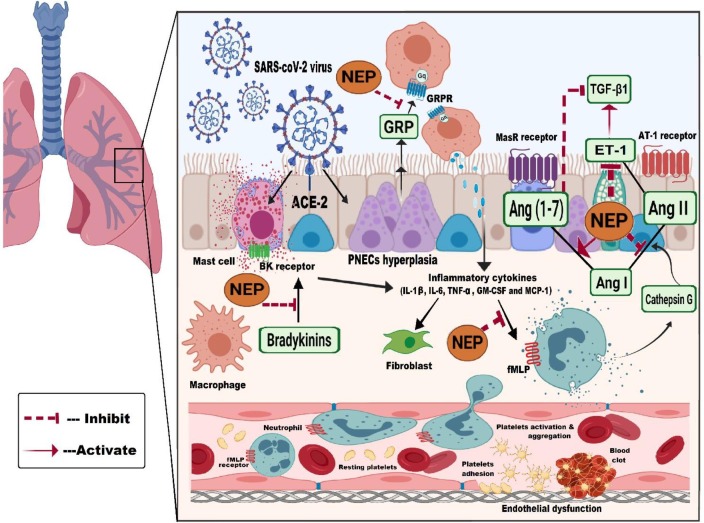
Fig. 3.
A schematic diagram showing the NEP-dependent therapeutic strategy for COVID-19. Following binding of SARS-coV-2 virus to ACE-2 receptor on the cell membrane surface, lung may show pulmonary neuroendocrine cells hyperplasia with infiltration of several inflammatory cells. The hyperplasia may produce excessive Gastrin-releasing peptide into the surrounding airway parenchyma to stimulate Gastrin-releasing peptide receptor on the surface of macrophages, which in turn, will enhance release of inflammatory mediators such as (IL-1β, IL-6, TNF-α, GM-CSF and MCP-1) contributing to neutrophils recruitment. Neprilysin may degrade the produced gastrin-releasing peptide inhibiting subsequent release of inflammatory cytokines. At the same time, neprilysin may also cleave the chemotactic peptide Formyl Methionyl-Leucyl-Proline; by which neutrophils are efficiently migrated; altering their chemotactic responsiveness and recruitment. NEP may withstand the potent cytokine storm, through :(i) minimizing Angiotensin II via preventing the proteolytic cleavage of angiotensinogen and Angiotensin Ⅰ into Angiotensin Ⅱ by neutrophil-derived Cathepsin G and via regenerating the synthesis of endogenous Angiotensin (1–7) that by itself may protect against pulmonary fibrosis through reducing TGF-β1 expression, (ii) breaking bradykinins, blocking its action on its receptors on mast cell, inhibiting release of inflammatory cells and thereby, fibroblasts activation that may participate in the development of lung fibrosis, (iii) degrading endothelin-1 and consequently, inhibiting TGF-β1 release and (iv) stabilizing Ang II-induced endothelial dysfunction as well as suppressing platelet activation and aggregation that initiate blood clot formation. ACE-2 = Angiotensin-converting enzyme 2; Ang I = Angiotensin I; Ang II = Angiotensin II; Ang (1–7) = Angiotensin (1–7); AT-1 = Angiotensin II type 1 receptor; BKs = Bradykinins; ET-1 = Endothelin-1; fMLP = Formyl Methionyl-Leucyl-Proline; GRP = Gastrin-releasing peptide; GRPR = Gastrin-releasing peptide receptor; MasR = Mas receptor; NEP = Neprilysin; PNECs = Pulmonary neuroendocrine cells; SARS-CoV-2 = Severe acute respiratory syndrome coronavirus-2.
NEP may abrogate GRP-induced neutrophil chemotaxis via cleaving GRP and degrading fMLP peptide that can modulate the chemotactic responsiveness of neutrophils. On the other aspect, NEP may withstand the potent cytokine storm, which was prescribed to be one of causes for lung damage progression and death in COVID-19 patients.
We suggest that NEP can diminish the release of inflammatory cytokines that may increase sensitivity of target cells for further stimulation by SARA-CO-2 virus [246], [247]. Thus, NEP can improve lung histopathology and enhance tissue survival through two mechanisms:
Firstly, interfering with Ang II formation via preventing the proteolytic cleavage of angiotensinogen and Ang I into Ang II by neutrophil-derived Cathepsin G [209], [210], [211] that is expected to be released continuously in response to uncontrolled neutrophil activation associated with COVID-19 patients and via regenerating the synthesis of endogenous Ang (1–7), expected to be minimized because of ACE-2 exhaustion by SARA-CO-2 virus [109]. Ang (1–7) by itself may protect against pulmonary fibrosis through reducing TGF-β1 expression [139].
Secondly, breaking BKs and thereby, inhibiting its role in activation and recruitment of the inflammatory cells.
However, recorded findings suggest that lung damage caused by COVID-19 is induced by an alternative mechanism rather than hyperinfammatory injury. It likely seems that endothelial activation and associated pulmonary intravascular coagulopathy are the contributing factors in COVID-19 pathogenesis [18].
Within this context, NEP may exert a critical role in suppressing ET-1 that mediates angiotensin II-induced endothelial and platelets dysfunction. Yet, NEP may minimize the chance for Ang II formation, which was reported to be a potent stimulator of ET-1 in endothelial cells [248]. Even, NEP can additionally degrades ET-1, preventing its associated inflammatory injury and eventual fibrotic cascade in the lung [81], [175].
Furthermore, we speculate that NEP may be helpful in dealing with individuals at high risk groups for COVID-19 that exhibit many obstacles in their management. NEP may regulate blood pressure in cardiovascular and hypertensive patients indirectly via decreasing both blood and tissue levels of Ang II by: (i) increasing Ang I substrate availability a result of inhibiting cathepsin G-mediated neutrophil release, and (ii) augmenting the rate of Ang I conversion into Ang (1–7), that previously reported to exert a stimulatory effect on ANP secretion via MasR [249].
For diabetic patients, we demonstrate that NEP may regulate pancreatic RAS flux to improve glycemic conditions. In response to NEP-mediated degradation 'in vitro', NEP may evoke the insulin secretory ability of Ang (1–7) via hydrolyzing it into the biologically active Ang (1–2) dipeptide in pancreatic islets [33], [250].
Numerous 'in vivo' and 'in vitro' experiments have been made to increase NEP expression. One study speculated that dexamethasone could enhance NEP expression in airway epithelial cells via promoting its transcription and synthesis [251]. Another prior finding demonstrated that Valproic acid could reduce plaque formation and improve learning deficits via up-regulating NEP in APP/PS1 transgenic mice [252]. Recently, serotonin and its derivatives were explored to ameliorate symptoms of AD induced in mouse via enhancing NEP up-regulation [253].
Several lines of evidence identified that hormones such as androgens [254] and estrogen [255] could also positively regulate NEP expression, suggesting that decline in levels of androgen or estrogen associated with aging would accompany by decrease in NEP synthesis, which may be an important factor for increasing the risk of COVID-19 infection among elderly.
On the other hand, several findings investigated the up-regulating effect of some natural products on NEP expression such as Apigenin, Luteolin, and Curcumin, (-)-Epigallocatechin-3-gallate as well as Resveratrol [256], [257], [258]. In China, Naoerkang (NEK), a traditional Chinese herbal medicine, improved the ability of learning and memory in rats model of AD by increasing NEP expression in their hippocampal tissues [259].
However, some scientists aimed to produce recombinant NEP (r NEP) instead, as Park et al., 2013 who prepared recombinant soluble NEP from insect cells to be intracerebrally injected into AD mice [238].
Therefore, we finally suggest that therapeutic strategies aimed to increase NEP expression and/or activity may be of great benefit for prevention and treatment of COVID-19.
6. Conclusions and areas for future work
Despite the high widespread of COVID-19 contagion worldwide, there is no specific efficient treatment that has been proved till now. Several pharmacological interventions for COVID-19 have been suggested targeting the host's immune response. Following SARS-coV-2 exposure, there may be an overproduction of GRP within lung tissue resulting in increased release of inflammatory cytokines such as IL-1β, IL-6, TNF-α, VEGF, GM-CSF and MCP-1. Such cytokines are widely known to enhance neutrophil infiltration which, in turn, induce lung inflammation and respiratory distress as reported in COVID-19 infected patients. As well, cytokines release is also presumed to develop SARS-coV-2 - associated pulmonary fibrosis. Novel findings define COVID-19 as one of the pulmonary diseases associated with endothelial and platelets dysfunction. Now, it is definitely known that viral invasion can be mediated by one of the RAS signaling system components, namely ACE-2. Hence, COVID-19 occurrence and progression can be attributed to imbalance in the pulmonary RAS signaling resulting from SARS-coV-2-induced ACE-2 drain. Interestingly, the decrease in ACE-2 activity will be accompanied with a decrease in the generation of Ang1-7 which was known to be the light side of RAS. Regarding the data emphasized on the protective role of Ang (1–7) in lung injury, it may be recommended as a COVID-19 therapy, but because of its short half-life, Ang (1–7) exhibits a limitation for its use. Since NEP is a more potent alternative way than ACE-2 for producing Ang1-7, it is suggested to assess the possible beneficial role of NEP against COVID-induced lung injury. Few studies have discussed NEP-mediated protective pathways in experimental models of lung injury and fibrosis, however its actual role as a lung protective therapy has not been yet recognized. NEP has been involved in degradation of many peptides that may be incorporated in COVID-19 pathophysiology. So, we expect that NEP can effectively interfere with the chemotactic responsiveness and recruitment of neutrophils by degrading both fMLP peptide and GPR, respectively. Furthermore, we suggest that NEP can minimize cytokine storm associated with SARS-coV-2 invasion through inhibiting Ang II formation by neutrophil-derived Cathepsin G and directing Ang I for generating Ang (1–7) which can in turn suppress TGF-β1 expression and its fibrogenic action, protecting against fibrosis. Degrading both BKs and ET-1 by NEP may be associated with low IL-6 levels, which will be beneficial for stabilizing endothelium and restoring its function. In addition to its catabolic properties, we postulate that NEP may possess an advantage for COVID-19 high risk patients through modulating blood pressure and glucose homeostasis. Practically, numerous in-vivo and in-vitro experimental manipulations were made to upregulate NEP expression either by using drugs (Dexamethasone and Valproic acid), hormones (Androgens and Estrogen) or natural substances (Apigenin, Luteolin, Curcumin and (-)-Epigallocatechin-3-gallate). However, others directed their efforts towards preparing the recombinant NEP (r NEP). Because most pre-clinical and clinical studies within the medical field are interested in studying NEP inhibitors, there is a little data concerning use of NEP as a therapeutic agent. Consequently, its associated adverse effects have not yet been studied well. Finally, we hope our hypothesis will be somewhat enough to direct a future work towards the therapeutic role of NEP in modulating COVID-19 pandemic and to target the subsequent therapies for enhancing NEP activity in COVID-19 patients.
Conflict of interest
The authors declare no conflict of interest. The authors and their institutions are the only responsible for the financial support and the content of this work in the submitted manuscript. All other authors have no conflict of interests to disclose.
Acknowledgments
The authors would like to thank all staff members of Environmental Studies & Research Institute-University of Sadat City, Egypt; Department of Pharmacology and Toxicology, Faculty of Pharmacy-Tanta University, Egypt; Department of Medical Physiology, Faculty of Medicine-Tanta University, Egypt for providing technical support. The authors are thankful to Dr. Emad Abdelmaksoud Elnaggar, Specialist of Cardiology, Mahala Cardiac Center, Egypt for his helpful comments and suggestions from his clinical point of view.
Contributor Information
Manar Mohammed El Tabaa, Email: manar.eltabaa@esri.usc.edu.eg.
Maram Mohammed El Tabaa, Email: maram.tabaa@med.tanta.edu.eg.
References
- 1.C. Lai, Y.H. Liu, C. Wang, Y. Wang, S. Hsueh, M. Yen, W. Ko, P.R. Hsueh, Asymptomatic carrier state, acute respiratory disease, and pneumonia due to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2): Facts and myths, J. Microbiol. Immunol. Infect. (2020). [DOI] [PMC free article] [PubMed]
- 2.M. Cascella, M. Rajnik, A. Cuomo, S.C. Dulebohn, R. Di Napoli, Features, evaluation and treatment coronavirus (COVID-19), in: StatPearls, 2020. [PubMed]
- 3.Huang C., Wang Y., Li X., Ren L., Zhao J., Hu Y., Zhang L., Fan G., Xu J., Gu X., Cheng Z., Yu T., Xia J., Wei Y., Wu W., Xie X., Yin W., Li H., Liu M., Xiao Y., Gao H., Guo L., Xie J., Wang G., Jiang R., Gao Z., Jin Q., Wang J., Cao B. Clinical features of patients infected with, novel coronavirus in Wuhan, China. Lancet. 2019;395(2020):497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mahase E. Coronavirus: covid-19 has killed more people than SARS and MERS combined, despite lower case fatality rate. BMJ. 2020;368:m641. doi: 10.1136/bmj.m641. [DOI] [PubMed] [Google Scholar]
- 5.Chen A.T.C., Coura-Filho G.B., Rehder M.H.H. Clinical characteristics of Covid-19 in China. N. Engl. J. Med. 2020;382:1–3. doi: 10.1056/NEJMc2005203. [DOI] [PubMed] [Google Scholar]
- 6.Shi H., Han X., Jiang N., Cao Y., Alwalid O., Gu J., Fan Y., Zheng C. Radiological findings from 81 patients with COVID-19 pneumonia in Wuhan, China: a descriptive study. Lancet. Infect. Dis. 2020;20:425–434. doi: 10.1016/S1473-3099(20)30086-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Raoult D., Zumla A., Locatelli F., Ippolito G., Kroemer G. Coronavirus infections: epidemiological, clinical and immunological features and hypotheses. Cell Stress. 2020;4:66–75. doi: 10.15698/cst2020.04.216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.A.F. Rabi, S.M. Al Zoubi, A.G. Kasasbeh, M.D. Salameh, D.A. Al-Nasser, SARS-CoV-2 and Coronavirus Disease 2019: What We Know So Far, Pathogens. 9 (2020). [DOI] [PMC free article] [PubMed]
- 9.Zhou F., Yu T., Du R., Fan G., Liu Y., Liu Z., Xiang J., Wang Y., Song B., Gu X., Guan L., Wei Y., Li H., Wu X., Xu J., Tu S., Zhang Y., Chen H., Cao B. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054–1062. doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Mehta P., McAuley D.F., Brown M., Sanchez E., Tattersall R.S., Manson J.J. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020;395:1033–1034. doi: 10.1016/S0140-6736(20)30628-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mitjà O., Clotet B. Correspondence Use of antiviral drugs to reduce COVID-19. Lancet Glob. Heal. 2020:2019–2020. doi: 10.1016/S2214-109X(20)30114-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.G. Li, E. De Clercq, Therapeutic options for the 2019 novel coronavirus (2019-nCoV), Nat. Rev. Drug Discov. 19 (2020) 149–150. [DOI] [PubMed]
- 13.Lu H. Drug treatment options for the 2019-new coronavirus (2019-nCoV) Biosci. Trends. 2020;14:69–71. doi: 10.5582/bst.2020.01020. [DOI] [PubMed] [Google Scholar]
- 14.X. Li, M. Geng, Y. Peng, L. Meng, S. Lu, Molecular immune pathogenesis and diagnosis of COVID-19, J. Pharm. Anal. (2020). [DOI] [PMC free article] [PubMed]
- 15.K. Tveito, Cytokinstormer ved covid-19?, Tidsskr. Den Nor. Legeforening. (2020). [DOI] [PubMed]
- 16.G. Zhang, C. Hu, L. Luo, F. Fang, Y. Chen, J. Li, Z. Peng, H. Pan, Clinical features and outcomes of 221 patients with COVID-19 in Wuhan, China, MedRxiv. (2020). [DOI] [PMC free article] [PubMed]
- 17.Conti P., Ronconi G., Caraffa A., Gallenga C., Ross R., Frydas I., Kritas S. Induction of pro-inflammatory cytokines (IL-1 and IL-6) and lung inflammation by Coronavirus-19 (COVI-19 or SARS-CoV- 2): anti-inflammatory strategies. J. Biol. Regul. Homeost. Agents. 2020;34 doi: 10.23812/CONTI-E. [DOI] [PubMed] [Google Scholar]
- 18.D.E. Leisman, C.S. Deutschman, M. Legrand, Facing COVID-19 in the ICU: vascular dysfunction, thrombosis, and dysregulated inflammation, Intensive Care Med. (2020) 1–4. [DOI] [PMC free article] [PubMed]
- 19.Millar F.R., Summers C., Griffiths M.J., Toshner M.R., Proudfoot A.G. The pulmonary endothelium in acute respiratory distress syndrome: insights and therapeutic opportunities. Thorax. 2016;71:462–473. doi: 10.1136/thoraxjnl-2015-207461. [DOI] [PubMed] [Google Scholar]
- 20.Avula A., Nalleballe K., Narula N., Sapozhnikov S., Dandu V., Toom S., Glaser A., Elsayegh D. Brain, behavior, and immunity COVID-19 presenting as stroke. Brain Behav. Immun. 2020;1 doi: 10.1016/j.bbi.2020.04.077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zheng Y., Huang Z., Ying G., Zhang X., Ye W., Hu Z., Hu C., Wei H., Zeng Y., Chi Y. Comparative study of the lymphocyte change between COVID-19 and non-COVID-19 pneumonia cases suggesting uncontrolled inflammation might not be the main reason of tissue injury. MedRxiv. 2020 [Google Scholar]
- 22.Zhang W., Zhao Y., Zhang F., Wang Q., Li T., Liu Z. The use of anti-inflammatory drugs in the treatment of people with severe coronavirus disease 2019 (COVID-19): the experience of clinical immunologists from China. Clin. Immunol. 2020;214 doi: 10.1016/j.clim.2020.108393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Rong Qu, Yun Ling, Yi‐huizhi Zhang, Li‐ya Wei, Xiao Chen, Xumian Li, Xuan‐yong Liu, Han‐mian Liu, Zhi Guo, Hua Ren, Qiang Wang, Platelet-to-lymphocyte ratio is associated with prognosis in patients with Corona Virus Disease-19, J. Med. Virol. (2020) 0–3. [DOI] [PMC free article] [PubMed]
- 24.Russell B., Moss C., George G., Santaolalla A., Cope A., Papa S., Van Hemelrijck M. Associations between immune-suppressive and stimulating drugs and novel COVID-19—a systematic review of current evidence. Ecancermedicalscience. 2020;14:1022. doi: 10.3332/ecancer.2020.1022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Savarino A., Boelaert J.R., Cassone A., Majori G., Cauda R. Effects of chloroquine on viral infections: an old drug against today’s diseases. Lancet Infect. Dis. 2003;3:722–727. doi: 10.1016/S1473-3099(03)00806-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.P. Smith, T. BCPS, P. Tony Prosser, COVID-19 drug therapy-potential options, Clin. Drug Information, Clin. Solut. March (2020).
- 27.J. Sun, W.-T. He, L. Wang, A. Lai, X. Ji, X. Zhai, G. Li, M.A. Suchard, J. Tian, J. Zhou, M. Veit, S. Su, COVID-19: epidemiology, evolution, and cross-disciplinary perspectives, Trends Mol. Med. (2020). [DOI] [PMC free article] [PubMed]
- 28.L. Malha, F.B. Mueller, M.S. Pecker, S.J. Mann, P. August, P.U. Feig, COVID-19 and the Renin-Angiotensin System, Kidney Int. Reports. (2020). [DOI] [PMC free article] [PubMed]
- 29.G.M. Kuster, O. Pfister, T. Burkard, Q. Zhou, R. Twerenbold, P. Haaf, A.F. Widmer, S. Osswald, SARS-CoV2: should inhibitors of the renin-angiotensin system be withdrawn in patients with COVID-19?, Eur. Heart J. (2020) 1–3. [DOI] [PMC free article] [PubMed]
- 30.Junyi G., Zheng H., Li L., Jiagao L. Coronavirus disease 2019 (COVID-19) and cardiovascular disease: a viewpoint on the potential influence of angiotensin-converting enzyme inhibitors/angiotensin receptor blockers on onset and severity of severe acute respiratory syndrome Coronavirus 2 infec. J. Am. Heart Assoc. 2020;9 doi: 10.1161/JAHA.120.016219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.M. Vaduganathan, O. Vardeny, T. Michel, J.J. V McMurray, M.A. Pfeffer, S.D. Solomon, Renin–angiotensin–aldosterone system inhibitors in patients with Covid-19, N. Engl. J. Med. (2020). [DOI] [PMC free article] [PubMed]
- 32.Esler M., Esler D. Can angiotensin receptor-blocking drugs perhaps be harmful in the COVID-19 pandemic? J. Hypertens. 2020;38:781–783. doi: 10.1097/HJH.0000000000002450. [DOI] [PubMed] [Google Scholar]
- 33.Brar G.S., Barrow B.M., Watson M., Griesbach R., Choung E., Welch A., Ruzsicska B., Raleigh D.P., Zraika S. Neprilysin is required for angiotensin-(1–7)’s ability to enhance insulin secretion via its proteolytic activity to generate angiotensin-(1–2) Diabetes. 2017;66:2201–2212. doi: 10.2337/db16-1318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Esser N., Zraika S. Neprilysin inhibition: a new therapeutic option for type 2 diabetes? Diabetologia. 2019;62:1113–1122. doi: 10.1007/s00125-019-4889-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Docherty K.F., McMurray J.J.V. PIONEER-HF: a new frontier in the role of neprilysin inhibition in the management of heart failure with reduced ejection fraction. Cardiovasc. Res. 2019;115:e136–e139. doi: 10.1093/cvr/cvz223. [DOI] [PubMed] [Google Scholar]
- 36.Baliga R.S., Scotton C.J., Trinder S.L., Chambers R.C., MacAllister R.J., Hobbs A.J. Intrinsic defence capacity and therapeutic potential of natriuretic peptides in pulmonary hypertension associated with lung fibrosis. Br. J. Pharmacol. 2014;171:3463–3475. doi: 10.1111/bph.12694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hashimoto S., Amaya F., Oh-hashi K., Kiuchi K., Hashimoto S. Expression of neutral endopeptidase activity during clinical and experimental acute lung injury. Respir. Res. 2010;11:164. doi: 10.1186/1465-9921-11-164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wick M.J., Buesing E.J., Wehling C.A., Loomis Z.L., Cool C.D., Zamora M.R., Miller Y.E., Colgan S.P., Hersh L.B., Voelkel N.F., Dempsey E.C. Decreased neprilysin and pulmonary vascular remodeling in chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 2011;183:330–340. doi: 10.1164/rccm.201002-0154OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Borson D.B., Brokaw J.J., Sekizawa K., McDonald D.M., Nadel J.A. Neutral endopeptidase and neurogenic inflammation in rats with respiratory infections. J. Appl. Physiol. 1989;66:2653–2658. doi: 10.1152/jappl.1989.66.6.2653. [DOI] [PubMed] [Google Scholar]
- 40.Li F. Structure, function, and evolution of coronavirus spike proteins. Annu. Rev. Virol. 2016;3:237–261. doi: 10.1146/annurev-virology-110615-042301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ashour H.M., Elkhatib W.F., Rahman M., Elshabrawy H.A. Insights into the recent 2019 novel Coronavirus (SARS-CoV-2) in light of past human coronavirus outbreaks. Pathogens. 2020;9:186. doi: 10.3390/pathogens9030186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kirchdoerfer R.N., Cottrell C.A., Wang N., Pallesen J., Yassine H.M., Turner H.L., Corbett K.S., Graham B.S., McLellan J.S., Ward A.B. Pre-fusion structure of a human coronavirus spike protein. Nature. 2016;531:118–121. doi: 10.1038/nature17200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.C. Yin, Genotyping coronavirus SARS-CoV-2: methods and implications, ArXiv Prepr. ArXiv2003.10965 (2020). [DOI] [PMC free article] [PubMed]
- 44.Lau S.K.P., Zhang L., Luk H.K.H., Xiong L., Peng X., Li K.S.M., He X., Zhao P.S.H., Fan R.Y.Y., Wong A.C.P. Receptor usage of a novel bat lineage c betacoronavirus reveals evolution of Middle East respiratory syndrome-related coronavirus spike proteins for human dipeptidyl peptidase 4 binding. J. Infect. Dis. 2018;218:197–207. doi: 10.1093/infdis/jiy018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Matsuyama S., Nao N., Shirato K., Kawase M., Saito S., Takayama I., Nagata N., Sekizuka T., Katoh H., Kato F., Sakata M., Tahara M., Kutsuna S., Ohmagari N., Kuroda M., Suzuki T., Kageyama T., Takeda M. Enhanced isolation of SARS-CoV-2 by TMPRSS2-expressing cells. Proc. Natl. Acad. Sci. 2020;117:7001–7003. doi: 10.1073/pnas.2002589117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Zhu Y., Yu D., Yan H., Chong H., He Y. Design of potent membrane fusion inhibitors against SARS-CoV-2, an emerging coronavirus with high fusogenic activity. BioRxiv. 2020 doi: 10.1128/JVI.00635-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Matsuyama S., Nagata N., Shirato K., Kawase M., Takeda M., Taguchi F. Efficient activation of the severe acute respiratory syndrome coronavirus spike protein by the transmembrane protease TMPRSS2. J. Virol. 2010;84:12658–12664. doi: 10.1128/JVI.01542-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Hoffmann M., Kleine-Weber H., Schroeder S., Krüger N., Herrler T., Erichsen S., Schiergens T.S., Herrler G., Wu N.-H., Nitsche A., Müller M.A., Drosten C., Pöhlmann S. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell. 2020 doi: 10.1016/j.cell.2020.02.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.X. Ou, Y. Liu, X. Lei, P. Li, D. Mi, L. Ren, L. Guo, R. Guo, T. Chen, J. Hu, Z. Xiang, Z. Mu, X. Chen, J. Chen, K. Hu, Q. Jin, J. Wang, Z. Qian, Characterization of spike glycoprotein of SARS-CoV-2 on virus entry and its immune cross-reactivity with SARS-CoV, Nat. Commun. (2020). [DOI] [PMC free article] [PubMed]
- 50.Ibrahim M.M. RAS inhibition in hypertension. J. Hum. Hypertens. 2006;20:101–108. doi: 10.1038/sj.jhh.1001960. [DOI] [PubMed] [Google Scholar]
- 51.Guang C., Phillips R.D., Jiang B., Milani F. Three key proteases–angiotensin-I-converting enzyme (ACE), ACE2 and renin–within and beyond the renin-angiotensin system. Arch. Cardiovasc. Dis. 2012;105:373–385. doi: 10.1016/j.acvd.2012.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Marshall R.P. The pulmonary renin-angiotensin system. Curr. Pharm. Des. 2003;9:715–722. doi: 10.2174/1381612033455431. [DOI] [PubMed] [Google Scholar]
- 53.Wang L., Wang Y., Yang T., Guo Y., Sun T. Angiotensin-converting enzyme 2 attenuates bleomycin-induced lung fibrosis in mice. Cell. Physiol. Biochem. 2015;36:697–711. doi: 10.1159/000430131. [DOI] [PubMed] [Google Scholar]
- 54.G.S. Magalhães, M.J. Campagnole-Santos, M. da Glória Rodrigues-Machado, Lung BT - Angiotensin-(1-7): A Comprehensive Review, in: R.A.S. Santos (Ed.), Cham, 2019, pp. 131–152.
- 55.Imai Y., Kuba K., Penninger J.M. The discovery of angiotensin-converting enzyme 2 and its role in acute lung injury in mice. Exp. Physiol. 2008;93:543–548. doi: 10.1113/expphysiol.2007.040048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Kuba K., Imai Y., Penninger J.M. Angiotensin-converting enzyme 2 in lung diseases. Curr. Opin. Pharmacol. 2006;6:271–276. doi: 10.1016/j.coph.2006.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Campbell D.J. Clinical relevance of local renin angiotensin systems. Front. Endocrinol. (Lausanne). 2014;5:113. doi: 10.3389/fendo.2014.00113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Gu H., Xie Z., Li T., Zhang S., Lai C., Zhu P., Wang K., Han L., Duan Y., Zhao Z., Yang X., Xing L., Zhang P., Wang Z., Li R., Yu J.J., Wang X., Yang P. Angiotensin-converting enzyme 2 inhibits lung injury induced by respiratory syncytial virus. Sci. Rep. 2016;6:19840. doi: 10.1038/srep19840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.A.C. Guyton, Textbook Of Medical Physiology, 2016.
- 60.B. Greenberg, An ACE in the hole: alternative pathways of the renin angiotensin system and their potential role in cardiac remodeling, J. Am. Coll. Cardiol. (2008) 755–757. [DOI] [PubMed]
- 61.Fyhrquist F., Saijonmaa O. Renin-angiotensin system revisited. J. Intern. Med. 2008;264:224–236. doi: 10.1111/j.1365-2796.2008.01981.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Santos R.A.S., Sampaio W.O., Alzamora A.C., Motta-Santos D., Alenina N., Bader M., Campagnole-Santos M.J. The ACE2/Angiotensin-(1–7)/MAS axis of the Renin-Angiotensin system: focus on Angiotensin-(1–7) Physiol. Rev. 2017;98:505–553. doi: 10.1152/physrev.00023.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Aguilera G. Role of angiotensin II receptor subtypes on the regulation of aldosterone secretion in the adrenal glomerulosa zone in the rat. Mol. Cell. Endocrinol. 1992;90:53–60. doi: 10.1016/0303-7207(92)90101-b. [DOI] [PubMed] [Google Scholar]
- 64.Benigni A., Cassis P., Remuzzi G. Angiotensin II revisited: new roles in inflammation, immunology and aging. EMBO Mol. Med. 2010;2:247–257. doi: 10.1002/emmm.201000080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Tan W.S.D., Liao W., Zhou S., Mei D., Wong W.S.F. Targeting the renin–angiotensin system as novel therapeutic strategy for pulmonary diseases. Curr. Opin. Pharmacol. 2018;40:9–17. doi: 10.1016/j.coph.2017.12.002. [DOI] [PubMed] [Google Scholar]
- 66.Blaes N., Girolami J.-P. Targeting the ‘Janus face’’ of the B2-bradykinin receptor’. Expert Opin. Ther. Targets. 2013;17:1145–1166. doi: 10.1517/14728222.2013.827664. [DOI] [PubMed] [Google Scholar]
- 67.Nukiwa T., Matsuoka R., Takagi H., Ishii Y., Arai T., Kira S. Responses of serum and lung angiotensin-converting enzyme activities in the early phase of pulmonary damage induced by oleic acid in dogs. Am. Rev. Respir. Dis. 1982;126:1080–1086. doi: 10.1164/arrd.1982.126.6.1080. [DOI] [PubMed] [Google Scholar]
- 68.Liu Y., Yang Y., Zhang C., Huang F., Wang F., Yuan J., Wang Z., Li J., Li J., Feng C., Zhang Z., Wang L., Peng L., Chen L., Qin Y., Zhao D., Tan S., Yin L., Xu J., Zhou C., Jiang C., Liu L. Clinical and biochemical indexes from 2019-nCoV infected patients linked to viral loads and lung injury. Sci. China Life Sci. 2020;63:364–374. doi: 10.1007/s11427-020-1643-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Hampl V., Herget J., Bíbová J., Baňasová A., Husková Z., Vaňourková Z., Jíchová Š., Kujal P., Vernerová Z., Sadowski J. Intrapulmonary activation of the angiotensin-converting enzyme type 2/angiotensin 1–7/G-protein-coupled Mas receptor axis attenuates pulmonary hypertension in Ren-2 transgenic rats exposed to chronic hypoxia. Physiol. Res. 2015;64 doi: 10.33549/physiolres.932861. [DOI] [PubMed] [Google Scholar]
- 70.Balakumar P., Jagadeesh G. A century old renin-angiotensin system still grows with endless possibilities: AT1 receptor signaling cascades in cardiovascular physiopathology. Cell. Signal. 2014;26:2147–2160. doi: 10.1016/j.cellsig.2014.06.011. [DOI] [PubMed] [Google Scholar]
- 71.Hahn A.W.A., Jonas U., Bühler F.R., Resink T.J. Activation of human peripheral monocytes by angiotensin II. FEBS Lett. 1994;347:178–180. doi: 10.1016/0014-5793(94)00531-1. [DOI] [PubMed] [Google Scholar]
- 72.Han Y., Runge M.S., Brasier A.R. Angiotensin II induces interleukin-6 transcription in vascular smooth muscle cells through pleiotropic activation of nuclear factor-kappa B transcription factors. Circ. Res. 1999;84:695–703. doi: 10.1161/01.res.84.6.695. [DOI] [PubMed] [Google Scholar]
- 73.Tsutamoto T., Wada A., Maeda K., Mabuchi N., Hayashi M., Tsutsui T., Ohnishi M., Sawaki M., Fujii M., Matsumoto T., Kinoshita M. Angiotensin II type 1 receptor antagonist decreases plasma levels of tumor necrosis factor alpha, interleukin-6 and soluble adhesion molecules in patients with chronic heart failure. J. Am. Coll. Cardiol. 2000;35:714–721. doi: 10.1016/s0735-1097(99)00594-x. [DOI] [PubMed] [Google Scholar]
- 74.Rathinasabapathy A., Horowitz A., Horton K., Kumar A., Gladson S., Unger T., Martinez D., Bedse G., West J., Raizada M.K., Steckelings U.M., Sumners C., Katovich M.J., Shenoy V. The selective angiotensin II type 2 receptor agonist, compound 21, attenuates the progression of lung fibrosis and pulmonary hypertension in an experimental model of bleomycin-induced lung injury. Front. Physiol. 2018;9 doi: 10.3389/fphys.2018.00180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Namsolleck P., Recarti C., Foulquier S., Steckelings U.M., Unger T. AT2 receptor and tissue injury: therapeutic implications topical collection on mediators, mechanisms, and pathways in tissue injury. Curr. Hypertens. Rep. 2014;16 doi: 10.1007/s11906-013-0416-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Nakashima H., Suzuki H., Ohtsu H., Chao J., Utsunomiya H., Frank G., Eguchi S. Angiotensin II regulates vascular and endothelial dysfunction: recent topics of angiotensin II Type-1 receptor signaling in the vasculature. Curr. Vasc. Pharmacol. 2005;4:67–78. doi: 10.2174/157016106775203126. [DOI] [PubMed] [Google Scholar]
- 77.Shatanawi A., Romero M.J., Iddings J.A., Chandra S., Umapathy N.S., Verin A.D., Caldwell R.B., Caldwell R.W. Angiotensin II-induced vascular endothelial dysfunction through RhoA/Rho kinase/p38 mitogen-activated protein kinase/arginase pathway. Am. J. Physiol. - Cell Physiol. 2011;300:C1181. doi: 10.1152/ajpcell.00328.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Duprez D.A. Angiotensin II, platelets and oxidative stress. J. Hypertens. 2004;22:1085–1086. doi: 10.1097/00004872-200406000-00005. [DOI] [PubMed] [Google Scholar]
- 79.Rajagopalan S., Laursen J.B., Borthayre A., Kurz S., Keiser J., Haleen S., Giaid A., Harrison D.G. Role for endothelin-1 in angiotensin II-mediated hypertension. Hypertension. 1997;30:29–34. doi: 10.1161/01.hyp.30.1.29. [DOI] [PubMed] [Google Scholar]
- 80.Kowalczyk A., Kleniewska P., Kolodziejczyk M., Skibska B., Goraca A. The role of endothelin-1 and endothelin receptor antagonists in inflammatory response and sepsis. Arch. Immunol. Ther. Exp. (Warsz) 2015;63:41–52. doi: 10.1007/s00005-014-0310-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Fagan K.A., McMurtry I.F., Rodman D.M. Role of endothelin-1 in lung disease. Respir. Res. 2001;2:90–101. doi: 10.1186/rr44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Yanagisawa M., Kurihara H., Kimura S., Tomobe Y., Kobayashi M., Mitsui Y., Yazaki Y., Goto K., Masaki T. A novel potent vasoconstrictor peptide produced by vascular endothelial cells. Nature. 1988;332:411–415. doi: 10.1038/332411a0. [DOI] [PubMed] [Google Scholar]
- 83.Browatzki M., Schmidt J., Kübler W., Kranzhöfer R. Endothelin-1 induces interleukin-6 release via activation of the transcription factor NF-κB in human vascular smooth muscle cells. Basic Res. Cardiol. 2000;95:98–105. doi: 10.1007/s003950050170. [DOI] [PubMed] [Google Scholar]
- 84.Touyz R.M., Schiffrin E.L. Effects of angiotensin II and endothelin-1 on platelet aggregation and cytosolic pH and free Ca2+ concentrations in essential hypertension. Hypertension. 1993;22:853–862. doi: 10.1161/01.hyp.22.6.853. [DOI] [PubMed] [Google Scholar]
- 85.Esteve E., Castro A., López-Bermejo A., Vendrell J., Ricart W., Fernández-Real J.M. Serum interleukin-6 correlates with endothelial dysfunction in healthy men independently of insulin sensitivity. Diabetes Care. 2007;30:939–945. doi: 10.2337/dc06-1793. [DOI] [PubMed] [Google Scholar]
- 86.Y. Pirahanchi, K.N. Brown, Physiology, Endothelial Derived Relaxation Factor (EDRF), 2019. [PubMed]
- 87.Vanhoutte P.M., Shimokawa H., Tang E.H.C., Feletou M. Endothelial dysfunction and vascular disease. Acta Physiol. 2009;196:193–222. doi: 10.1111/j.1748-1716.2009.01964.x. [DOI] [PubMed] [Google Scholar]
- 88.Radomski M.W., Palmer R.M.J., Moncada S. Endogenous nitric oxide inhibits human platelet adhesion to vascular endothelium. Lancet. 1987;330:1057–1058. doi: 10.1016/s0140-6736(87)91481-4. [DOI] [PubMed] [Google Scholar]
- 89.Kubes P., Suzuki M., Granger D.N. Nitric oxide: An endogenous modulator of leukocyte adhesion. Proc. Natl. Acad. Sci. U. S. A. 1991;88:4651–4655. doi: 10.1073/pnas.88.11.4651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Hung M.J., Cherng W.J., Hung M.Y., Wu H.T., Pang J.H.S. Interleukin-6 inhibits endothelial nitric oxide synthase activation and increases endothelial nitric oxide synthase binding to stabilized caveolin-1 in human vascular endothelial cells. J. Hypertens. 2010;28:940–951. doi: 10.1097/HJH.0b013e32833992ef. [DOI] [PubMed] [Google Scholar]
- 91.Wassmann S., Stumpf M., Strehlow K., Schmid A., Schieffer B., Böhm M., Nickenig G. Interleukin-6 induces oxidative stress and endothelial dysfunction by overexpression of the angiotensin II Type 1 receptor. Circ. Res. 2004;94:534–541. doi: 10.1161/01.RES.0000115557.25127.8D. [DOI] [PubMed] [Google Scholar]
- 92.Drexler H. Endothelium as a therapeutic target in heart failure. Circulation. 1998;98:2652–2655. doi: 10.1161/01.cir.98.24.2652. [DOI] [PubMed] [Google Scholar]
- 93.Versari D., Daghini E., Virdis A., Ghiadoni L., Taddei S. Endothelium-dependent contractions and endothelial dysfunction in human hypertension. Br. J. Pharmacol. 2009;157:527–536. doi: 10.1111/j.1476-5381.2009.00240.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.De Vriese A.S., Verbeuren T.J., Van De Voorde J., Lameire N.H., Vanhoutte P.M. Endothelial dysfunction in diabetes. Br. J. Pharmacol. 2000;130:963–974. doi: 10.1038/sj.bjp.0703393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Stoll G., Kleinschnitz C., Nieswandt B. Molecular mechanisms of thrombus formation in ischemic stroke: novel insights and targets for treatment. Blood. 2008;112:3555–3562. doi: 10.1182/blood-2008-04-144758. [DOI] [PubMed] [Google Scholar]
- 96.Pilaczyńska-Cemel M., Gołda R., Dąbrowska A., Przybylski G. Analysis of the level of selected parameters of inflammation, circulating immune complexes, and related indicators (neutrophil/ lymphocyte, platelet/lymphocyte, CRP/CIC) in patients with obstructive diseases. Cent. Eur. J. Immunol. 2019;44:292–298. doi: 10.5114/ceji.2019.87498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Thorin E., Webb D.J. Endothelium-derived endothelin-1. Pflugers Arch. Eur. J. Physiol. 2010;459:951–958. doi: 10.1007/s00424-009-0763-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Kaser A., Brandacher G., Steurer W., Kaser S., Offner F.A., Zoller H., Theurl I., Widder W., Molnar C., Ludwiczek O., Atkins M.B., Mier J.W., Tilg H. Interleukin-6 stimulates thrombopoiesis through thrombopoietin: role in inflammatory thrombocytosis. Blood. 2001;98:2720–2725. doi: 10.1182/blood.v98.9.2720. [DOI] [PubMed] [Google Scholar]
- 99.Mustafa S.B., Gandhi C.R., Harvey S.A., Olson M.S. Endothelin stimulates platelet-activating factor synthesis by cultured rat Kupffer cells. Hepatology. 1995;21:545–553. [PubMed] [Google Scholar]
- 100.Wermuth P.J., Li Z., Mendoza F.A., Jimenez S.A. Stimulation of transforming growth factor-β1-induced endothelial-to-mesenchymal transition and tissue fibrosis by endothelin-1 (ET-1): a novel profibrotic effect of ET-1. PLoS ONE. 2016;11 doi: 10.1371/journal.pone.0161988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.George P.M., Wells A.U., Jenkins G. Personal view pulmonary fibrosis and COVID- 19: the potential role for antifibrotic therapy. Lancet Respir. 2020 doi: 10.1016/S2213-2600(20)30225-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Rivière G., Michaud A., Breton C., VanCamp G., Laborie C., Enache M., Lesage J., Deloof S., Corvol P., Vieau D. Angiotensin-converting enzyme 2 (ACE2) and ACE activities display tissue-specific sensitivity to undernutrition-programmed hypertension in the adult rat. Hypertension. 2005;46:1169–1174. doi: 10.1161/01.HYP.0000185148.27901.fe. [DOI] [PubMed] [Google Scholar]
- 103.C. Bavishi, T.M. Maddox, F.H. Messerli, Coronavirus Disease 2019 (COVID-19) Infection and Renin Angiotensin System Blockers, JAMA Cardiol. (2020). [DOI] [PubMed]
- 104.Vickers C., Hales P., Kaushik V., Dick L., Gavin J., Tang J., Godbout K., Parsons T., Baronas E., Hsieh F. Hydrolysis of biological peptides by human angiotensin-converting enzyme-related carboxypeptidase. J. Biol. Chem. 2002;277:14838–14843. doi: 10.1074/jbc.M200581200. [DOI] [PubMed] [Google Scholar]
- 105.A.C. Simões E Silva, K.D. Silveira, A.J. Ferreira, M.M. Teixeira, ACE2, angiotensin-(1-7) and Mas receptor axis in inflammation and fibrosis, Br. J. Pharmacol. 169 (2013) 477–492. [DOI] [PMC free article] [PubMed]
- 106.Chappell M.C. Angiotensin-converting enzyme 2 autoantibodies: further evidence for a role of the renin-angiotensin system in inflammation. Arthritis Res. Ther. 2010;12:128. doi: 10.1186/ar3052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Li Y., Cao Y., Zeng Z., Liang M., Xue Y., Xi C., Zhou M., Jiang W. Angiotensin-converting enzyme 2/angiotensin-(1–7)/Mas axis prevents lipopolysaccharide-induced apoptosis of pulmonary microvascular endothelial cells by inhibiting JNK/NF-κB pathways. Sci. Rep. 2015;5:8209. doi: 10.1038/srep08209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Zhang H., Penninger J.M., Li Y., Zhong N., Slutsky A.S. Angiotensin-converting enzyme 2 (ACE2) as a SARS-CoV-2 receptor: molecular mechanisms and potential therapeutic target. Intensive Care Med. 2020;46:586–590. doi: 10.1007/s00134-020-05985-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Sun M.L., Yang J.M., Sun Y.P., Su G.H. Inhibitors of RAS might be a good choice for the therapy of COVID-19 pneumonia. Chinese J. Tuberc. Respir. Dis. 2020;43:E014. doi: 10.3760/cma.j.issn.1001-0939.2020.0014. [DOI] [PubMed] [Google Scholar]
- 110.Monteil V., Kwon H., Prado P., Hagelkrüys A., Wimmer R.A. Inhibition of SARS-CoV-2 infections in engineered human tissues using clinical-grade soluble human ACE2. Cell. 2020 doi: 10.1016/j.cell.2020.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.K. Dhama, K. Sharun, R. Tiwari, M. Dadar, Y.S. Malik, K.P. Singh, W. Chaicumpa, COVID-19, an emerging coronavirus infection: advances and prospects in designing and developing vaccines, immunotherapeutics, and therapeutics, Hum. Vaccin. Immunother. (2020) 1–7. [DOI] [PMC free article] [PubMed]
- 112.Ferrario C.M., Jessup J., Chappell M.C., Averill D.B., Brosnihan K.B., Tallant E.A., Diz D.I., Gallagher P.E. Effect of angiotensin-converting enzyme inhibition and angiotensin II receptor blockers on cardiac angiotensin-converting enzyme 2. Circulation. 2005;111:2605–2610. doi: 10.1161/CIRCULATIONAHA.104.510461. [DOI] [PubMed] [Google Scholar]
- 113.Varga Z., Flammer A.J., Steiger P., Haberecker M., Andermatt R., Zinkernagel A.S., Mehra M.R., Schuepbach R.A., Ruschitzka F., Moch H. Correspondence Endothelial cell infection and endotheliitis in. Lancet. 2019;395:1417–1418. doi: 10.1016/S0140-6736(20)30937-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Y. Zhao, Z. Zhao, Y. Wang, Y. Zhou, Y. Ma, W. Zuo, Single-cell RNA expression profiling of ACE2, the putative receptor of Wuhan 2019-nCov, BioRxiv. (2020). [DOI] [PMC free article] [PubMed]
- 115.Bertram S., Heurich A., Lavender H., Gierer S., Danisch S., Perin P., Lucas J.M., Nelson P.S., Pöhlmann S., Soilleux E.J. Influenza and SARS-coronavirus activating proteases TMPRSS2 and HAT are expressed at multiple sites in human respiratory and gastrointestinal tracts. PLoS ONE. 2012;7 doi: 10.1371/journal.pone.0035876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Lukassen S., Trefzer T., Kahn N.C., Schneider M.A., Muley T., Winter H., Meister M., Boots A.W., Hennig B.P., Kreuter M., Eils R. SARS-CoV-2 receptor ACE2 and TMPRSS2 are predominantly expressed in a transient secretory cell type in subsegmental bronchial branches. BioRxiv. 2020 doi: 10.15252/embj.20105114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.G. Li, R. Hu, X. Zhang, Antihypertensive treatment with ACEI/ARB of patients with COVID-19 complicated by hypertension, Hypertens. Res. (2020) 1–3. [DOI] [PMC free article] [PubMed]
- 118.L. Guo, D. Li, H. Yang, L. Liu, J. Fan, Potential pathogenesis of multiple organ injury in COVID-19, Preprint. (2020).
- 119.Wong S.H., Lui R.N.S., Sung J.J.Y. Covid-19 and the digestive system. J. Gastroenterol. Hepatol. 2020;35:744–748. doi: 10.1111/jgh.15047. [DOI] [PubMed] [Google Scholar]
- 120.Poyiadji N., Shahin G., Noujaim D., Stone M., Patel S., Griffith B. COVID-19–associated acute hemorrhagic necrotizing encephalopathy: CT and MRI features. Radiology. 2020;201187 doi: 10.1148/radiol.2020201187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Karimi N., Sharifi Razavi A., Rouhani N. Frequent convulsive seizures in an adult patient with COVID-19: a case report. Iran. Red Crescent Med. J. 2020;22 [Google Scholar]
- 122.Patel A.B., Verma A. COVID-19 and angiotensin-converting enzyme inhibitors and angiotensin receptor blockers: what is the evidence? JAMA. 2020 doi: 10.1001/jama.2020.4812. [DOI] [PubMed] [Google Scholar]
- 123.M.L. Chiusano, The modelling of COVID19 pathways sheds light on mechanisms, opportunities and on controversial interpretations of medical treatments. v2, ArXiv Prepr. ArXiv2003.11614. (2020).
- 124.Wösten-Van Asperen R.M., Lutter R., Specht P.A., Moll G.N., Van Woensel J.B., Van Der Loos C.M., Van Goor H., Kamilic J., Florquin S., Bos A.P. Acute respiratory distress syndrome leads to reduced ratio of ACE/ACE2 activities and is prevented by angiotensin-(1–7) or an angiotensin II receptor antagonist. J. Pathol. 2011;225:618–627. doi: 10.1002/path.2987. [DOI] [PubMed] [Google Scholar]
- 125.Shenoy V., Ferreira A.J., Qi Y., Fraga-Silva R.A., Díez-Freire C., Dooies A., Jun J.Y., Sriramula S., Mariappan N., Pourang D., Venugopal C.S., Francis J., Reudelhuber T., Santos R.A., Patel J.M., Raizada M.K., Katovich M.J. The angiotensin-converting enzyme 2/angiogenesis-(1–7)/mas axis confers cardiopulmonary protection against lung fibrosis and pulmonary hypertension. Am. J. Respir. Crit. Care Med. 2010;182:1065–1072. doi: 10.1164/rccm.200912-1840OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Dai H., Gong Y., Xiao Z., Guang X., Yin X. Decreased levels of serum Angiotensin-(1–7) in patients with pulmonary arterial hypertension due to congenital heart disease. Int. J. Cardiol. 2014;176:1399–1401. doi: 10.1016/j.ijcard.2014.08.021. [DOI] [PubMed] [Google Scholar]
- 127.Wang D., Chai X., Magnussen C.G., Zosky G.R., Shu S., Wei X., Hu S. Renin-angiotensin-system, a potential pharmacological candidate, in acute respiratory distress syndrome during mechanical ventilation. Pulm. Pharmacol. Ther. 2019;58 doi: 10.1016/j.pupt.2019.101833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Breitling S., Krauszman A., Parihar R., Walther T., Friedberg M.K., Kuebler W.M. Dose-dependent, therapeutic potential of angiotensin-(1–7) for the treatment of pulmonary arterial hypertension. Pulm. Circ. 2015;5:649–657. doi: 10.1086/683696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Chow B.S.M., Allen T.J. Angiotensin II type 2 receptor (AT2R) in renal and cardiovascular disease. Clin. Sci. 2016;130:1307–1326. doi: 10.1042/CS20160243. [DOI] [PubMed] [Google Scholar]
- 130.Padda R.S., Shi Y., Lo C.-S., Zhang S.-L., Chan J.S.D. Angiotensin-(1–7): a novel peptide to treat hypertension and nephropathy in diabetes? J. Diabetes Metab. 2015;6 doi: 10.4172/2155-6156.1000615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Sullivan J.C., Rodriguez-Miguelez P., Zimmerman M.A., Harris R.A. Differences in angiotensin (1–7) between men and women. Am. J. Physiol. Heart Circ. Physiol. 2015;308:H1171–H1176. doi: 10.1152/ajpheart.00897.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Fraga-Silva R.A., Pinheiro S.V.B., Gonçalves A.C.C., Alenina N., Bader M., Santos R.A.S. The antithrombotic effect of angiotensin-(1–7) involves mas-mediated NO release from platelets. Mol. Med. 2008;14:28–35. doi: 10.2119/2007-00073.Fraga-Silva. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Patel K.P., Schultz H.D. Angiotensin peptides and nitric oxide in cardiovascular disease. Antioxid. Redox Signal. 2013;19:1121–1132. doi: 10.1089/ars.2012.4614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Kuczeriszka M., Kompanowska-Jezierska E., Sadowski J., Prieto M.C., Navar L.G. Modulating role of Ang1-7 in control of blood pressure and renal function in AngII-infused hypertensive rats. Am. J. Hypertens. 2017;31:504–511. doi: 10.1093/ajh/hpy006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Dhaunsi G.S., Yousif M.H.M., Akhtar S., Chappell M.C., Diz D.I., Benter I.F. Angiotensin-(1–7) prevents diabetes-induced attenuation in PPAR-gamma and catalase activities. Eur. J. Pharmacol. 2010;638:108–114. doi: 10.1016/j.ejphar.2010.04.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Magalhaes G.S., Barroso L.C., Reis A.C., Rodrigues-Machado M.G., Gregório J.F., Motta-Santos D., Oliveira A.C., Perez D.A., Barcelos L.S., Teixeira M.M., Santos R.A.S., Pinho V., Campagnole-Santos M.J. Angiotensin-(1–7) promotes resolution of eosinophilic inflammation in an experimental model of asthma. Front. Immunol. 2018;9:58. doi: 10.3389/fimmu.2018.00058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Meng Y., Yu C.H., Li W., Li T., Luo W., Huang S., Wu P.S., Cai S.X., Li X. Angiotensin-converting enzyme 2/angiotensin-(1–7)/mas axis protects against lung fibrosis by inhibiting the MAPK/NF-κB pathway. Am. J. Respir. Cell Mol. Biol. 2014;50:723–736. doi: 10.1165/rcmb.2012-0451OC. [DOI] [PubMed] [Google Scholar]
- 138.Zhang Y., Liu J., Luo J.Y., Tian X.Y., Cheang W.S., Xu J., Lau C.W., Wang L., Wong W.T., Wong C.M., Lan H.Y., Yao X., Raizada M.K., Huang Y. Upregulation of angiotensin (1–7)-mediated signaling preserves endothelial function through reducing oxidative stress in diabetes. Antioxidants Redox Signal. 2015;23:880–892. doi: 10.1089/ars.2014.6070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.M. Shao, Z. Bin Wen, H.H. Yang, C.Y. Zhang, J.B. Xiong, X.X. Guan, W.J. Zhong, H.L. Jiang, C.C. Sun, X.Q. Luo, X. an He, Y. Zhou, C.X. Guan, Exogenous angiotensin (1-7) directly inhibits epithelial-mesenchymal transformation induced by transforming growth factor-β1 in alveolar epithelial cells, Biomed. Pharmacother. 117 (2019) 109193. [DOI] [PubMed]
- 140.Liu Y., Zheng Y. Bach1 siRNA attenuates bleomycin-induced pulmonary fibrosis by modulating oxidative stress in mice. Int. J. Mol. Med. 2017;39:91–100. doi: 10.3892/ijmm.2016.2823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Villalobos L.A., San Hipólito-Luengo Á., Ramos-González M., Cercas E., Vallejo S., Romero A., Romacho T., Carraro R., Sánchez-Ferrer C.F., Peiró C. The Angiotensin-(1–7)/mas axis counteracts Angiotensin II-dependent and -independent pro-inflammatory signaling in human vascular smooth muscle cells. Front. Pharmacol. 2016;7:482. doi: 10.3389/fphar.2016.00482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Fang Y., Gao F., Liu Z. Angiotensin-converting enzyme 2 attenuates inflammatory response and oxidative stress in hyperoxic lung injury by regulating NF-kappaB and Nrf2 pathways. QJM. 2019;112:914–924. doi: 10.1093/qjmed/hcz206. [DOI] [PubMed] [Google Scholar]
- 143.Povlsen A.L., Grimm D., Wehland M., Infanger M., Krüger M. The vasoactive mas receptor in essential hypertension. J. Clin. Med. 2020;9:267. doi: 10.3390/jcm9010267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Magalhaes G.S., Rodrigues-Machado M.G., Motta-Santos D., Silva A.R., Caliari M.V., Prata L.O., Abreu S.C., Rocco P.R.M., Barcelos L.S., Santos R.A.S., Campagnole-Santos M.J. Angiotensin-(1–7) attenuates airway remodelling and hyperresponsiveness in a model of chronic allergic lung inflammation. Br. J. Pharmacol. 2015;172:2330–2342. doi: 10.1111/bph.13057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Jiang F., Yang J., Zhang Y., Dong M., Wang S., Zhang Q., Liu F.F., Zhang K., Zhang C. Angiotensin-converting enzyme 2 and angiotensin 1–7: novel therapeutic targets. Nat. Rev. Cardiol. 2014;11:413–426. doi: 10.1038/nrcardio.2014.59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Wiemer G., Dobrucki L.W., Louka F.R., Malinski T., Heitsch H. AVE 0991, a nonpeptide mimic of the effects of angiotensin-(1–7) on the endothelium. Hypertension. 2002;40:847–852. doi: 10.1161/01.hyp.0000037979.53963.8f. [DOI] [PubMed] [Google Scholar]
- 147.Lula I., Denadai Â.L., Resende J.M., de Sousa F.B., de Lima G.F., Pilo-Veloso D., Heine T., Duarte H.A., Santos R.A.S., Sinisterra R.D. Study of angiotensin-(1–7) vasoactive peptide and its β-cyclodextrin inclusion complexes: complete sequence-specific NMR assignments and structural studies. Peptides. 2007;28:2199–2210. doi: 10.1016/j.peptides.2007.08.011. [DOI] [PubMed] [Google Scholar]
- 148.Savergnini S.Q., Beiman M., Lautner R.Q., de Paula-Carvalho V., Allahdadi K., Pessoa D.C., Costa-Fraga F.P., Fraga-Silva R.A., Cojocaru G., Cohen Y., Bader M., de Almeida A.P., Rotman G., Santos R.A.S. Vascular relaxation, antihypertensive effect, and cardioprotection of a novel peptide agonist of the MAS receptor. Hypertension. 2010;56:112–120. doi: 10.1161/HYPERTENSIONAHA.110.152942. [DOI] [PubMed] [Google Scholar]
- 149.Balingit P.P., Armstrong D.G., Reyzelman A.M., Bolton L., Verco S.J., Rodgers K.E., Nigh K.A., DiZerega G.S. NorLeu3-A(1–7) stimulation of diabetic foot ulcer healing: results of a randomized, parallel-group, double-blind, placebo-controlled phase 2 clinical trial. Wound Repair Regen. Off. Publ. Wound Heal. Soc. Eur. Tissue Repair Soc. 2012;20:482–490. doi: 10.1111/j.1524-475X.2012.00804.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Cassis P., Locatelli M., Corna D., Villa S., Rottoli D., Cerullo D., Abbate M., Remuzzi G., Benigni A., Zoja C. Addition of cyclic angiotensin-(1–7) to angiotensin-converting enzyme inhibitor therapy has a positive add-on effect in experimental diabetic nephropathy. Kidney Int. 2019;96:906–917. doi: 10.1016/j.kint.2019.04.024. [DOI] [PubMed] [Google Scholar]
- 151.Ferreira A.J., Santos R.A.S., Raizada M.K. Angiotensin-(1–7)/Angiotensin-converting enzyme 2/mas receptor axis and related mechanisms. Int. J. Hypertens. 2012;2012 doi: 10.1155/2012/690785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Peiró C., Vallejo S., Gembardt F., Azcutia V., Heringer-Walther S., Rodríguez-Mañas L., Schultheiss H.-P., Sánchez-Ferrer C.F., Walther T. Endothelial dysfunction through genetic deletion or inhibition of the G protein-coupled receptor Mas: a new target to improve endothelial function. J. Hypertens. 2007;25:2421–2425. doi: 10.1097/HJH.0b013e3282f0143c. [DOI] [PubMed] [Google Scholar]
- 153.Dehghani A., Saberi S., Nematbakhsh M. Role of mas receptor Antagonist A799 in renal blood flow response to Ang 1–7 after bradykinin administration in ovariectomized estradiol-treated rats. Adv. Pharmacol. Sci. 2015;2015 doi: 10.1155/2015/801053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Silva D.M.R., Vianna H.R., Cortes S.F., Campagnole-Santos M.J., Santos R.A.S., Lemos V.S. Evidence for a new angiotensin-(1–7) receptor subtype in the aorta of Sprague-Dawley rats. Peptides. 2007;28:702–707. doi: 10.1016/j.peptides.2006.10.007. [DOI] [PubMed] [Google Scholar]
- 155.T. Beril, de V. René, S.P. R., D.A.H. Jan, Bradykinin Potentiation by Angiotensin-(1-7) and ACE Inhibitors Correlates With ACE C- and N-Domain Blockade, Hypertension. 38 (2001) 95–99. [DOI] [PubMed]
- 156.Golias C., Charalabopoulos A., Stagikas D., Charalabopoulos K., Batistatou A. The kinin system–bradykinin: biological effects and clinical implications. Multiple role of the kinin system–bradykinin. Hippokratia. 2007;11:124–128. [PMC free article] [PubMed] [Google Scholar]
- 157.Da Huang C., Tliba O., Panettieri R.A., Amrani Y. Bradykinin induces interleukin-6 production in human airway smooth muscle cells: modulation by Th2 cytokines and dexamethasone. Am. J. Respir. Cell Mol. Biol. 2003;28:330–338. doi: 10.1165/rcmb.2002-0040OC. [DOI] [PubMed] [Google Scholar]
- 158.Paegelow I., Werner H., Vietinghoff G., Wartner U. Release of cytokines from isolated lung strips by bradykinin. Inflamm. Res. 1995;44:306–311. doi: 10.1007/BF02032574. [DOI] [PubMed] [Google Scholar]
- 159.Pan Z.K., Zuraw B.L., Lung C.C., Prossnitz E.R., Browning D.D., Ye R.D. Bradykinin stimulates NF-κB activation and interleukin 1β gene expression in cultured human fibroblasts. J. Clin. Invest. 1996;98:2042–2049. doi: 10.1172/JCI119009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Koyama S., Sato E., Numanami H., Kubo K., Nagai S., Izumi T. Bradykinin stimulates lung fibroblasts to release neutrophil and monocyte chemotactic activity. Am. J. Respir. Cell Mol. Biol. 2000;22:75–84. doi: 10.1165/ajrcmb.22.1.3752. [DOI] [PubMed] [Google Scholar]
- 161.L. de S. Silva, D. de B. Peruchetti, C.T.F.-D. Silva, A.T. Ferreira-DaSilva, J. Perales, C. Caruso-Neves, A.A.S. Pinheiro, Interaction between bradykinin B2 and Ang-(1–7) Mas receptors regulates erythrocyte invasion by Plasmodium falciparum, Biochim. Biophys. Acta - Gen. Subj. 1860 (2016) 2438–2444. [DOI] [PubMed]
- 162.K. Yamamoto, M.C. Chappell, K.B. Brosnihan, C.M. Ferrario, In vivo metabolism of angiotensin I by neutral endopeptidase (EC 3.4. 24.11) in spontaneously hypertensive rats, Hypertension. 19 (1992) 692–696. [DOI] [PubMed]
- 163.Romaní-Pérez M., Outeiriño-Iglesias V., Moya C.M., Santisteban P., González-Matías L.C., Vigo E., Mallo F. Activation of the GLP-1 receptor by liraglutide increases ACE2 expression, reversing right ventricle hypertrophy, and improving the production of SP-A and SP-B in the lungs of type 1 diabetes rats. Endocrinology. 2015;156:3559–3569. doi: 10.1210/en.2014-1685. [DOI] [PubMed] [Google Scholar]
- 164.Baraniuk J.N., Ohkubo K., Kwon O.J., Mak J., Ali M., Davies R., Twort C., Kaliner M., Letarte M., Barnes P.J. Localization of neutral endopeptidase (NEP) mRNA in human bronchi. Eur. Respir. J. 1995;8:1458–1464. [PubMed] [Google Scholar]
- 165.Knecht M., Pagel I., Langenickel T., Philipp S., Scheuermann-Freestone M., Willnow T., Bruemmer D., Graf K., Dietz R., Willenbrock R. Increased expression of renal neutral endopeptidase in severe heart failure. Life Sci. 2002;71:2701–2712. doi: 10.1016/s0024-3205(02)01990-2. [DOI] [PubMed] [Google Scholar]
- 166.Maguer-Satta V., Besançon R., Bachelard-Cascales E. Concise review: neutral endopeptidase (CD10): a multifaceted environment actor in stem cells, physiological mechanisms, and cancer. Stem Cells. 2011;29:389–396. doi: 10.1002/stem.592. [DOI] [PubMed] [Google Scholar]
- 167.Terauchi M., Kajiyama H., Shibata K., Ino K., Mizutani S., Kikkawa F. Anti-progressive effect of neutral endopeptidase 24.11 (NEP/CD10) on cervical carcinoma in vitro and in vivo. Oncology. 2005;69:52–62. doi: 10.1159/000087476. [DOI] [PubMed] [Google Scholar]
- 168.Smollich M., Götte M., Yip G.W., Yong E.-S., Kersting C., Fischgräbe J., Radke I., Kiesel L., Wülfing P. On the role of endothelin-converting enzyme-1 (ECE-1) and neprilysin in human breast cancer. Breast Cancer Res. Treat. 2007;106:361–369. doi: 10.1007/s10549-007-9516-9. [DOI] [PubMed] [Google Scholar]
- 169.Kuniyasu H., Luo Y., Fujii K., Sasahira T., Moriwaka Y., Tatsumoto N., Sasaki T., Yamashita Y., Ohmori H. CD10 enhances metastasis of colorectal cancer by abrogating the anti-tumoural effect of methionine-enkephalin in the liver. Gut. 2010;59:348–356. doi: 10.1136/gut.2009.178376. [DOI] [PubMed] [Google Scholar]
- 170.Pavo N., Arfsten H., Cho A., Goliasch G., Bartko P.E., Wurm R., Freitag C., Gisslinger H., Kornek G., Strunk G., Raderer M., Zielinski C., Hülsmann M. The circulating form of neprilysin is not a general biomarker for overall survival in treatment-naïve cancer patients. Sci. Rep. 2019;9:2554. doi: 10.1038/s41598-019-38867-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Pankow K., Schwiebs A., Becker M., Siems W.E., Krause G., Walther T. Structural substrate conditions required for neutral endopeptidase-mediated natriuretic peptide degradation. J. Mol. Biol. 2009;393:496–503. doi: 10.1016/j.jmb.2009.08.025. [DOI] [PubMed] [Google Scholar]
- 172.R.A. Ervin, G. Skidgel, Neutral endopeptidase 24.11 (enkephalinase) and related regulators of peptide hormones1, FASEB J. 3 (1989) 145–151. [PubMed]
- 173.Mangiafico S., Costello-Boerrigter L.C., Andersen I.A., Cataliotti A., Burnett J.C., Jr Neutral endopeptidase inhibition and the natriuretic peptide system: an evolving strategy in cardiovascular therapeutics. Eur. Heart J. 2012;34:886–893. doi: 10.1093/eurheartj/ehs262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Volpe M., Carnovali M., Mastromarino V. The natriuretic peptides system in the pathophysiology of heart failure: from molecular basis to treatment. Clin. Sci. (Lond) 2016;130:57–77. doi: 10.1042/CS20150469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Abassi Z.A., Tate J.E., Golomb E., Keiser H.R. Role of neutral endopeptidase in the metabolism of endothelin. Hypertension. 1992;20:89–95. doi: 10.1161/01.hyp.20.1.89. [DOI] [PubMed] [Google Scholar]
- 176.Roques B.P., Noble F., Daugé V., Fournié-Zaluski M.C., Beaumont A. Neutral endopeptidase 24.11: structure, inhibition, and experimental and clinical pharmacology. Pharmacol. Rev. 45. 1993 [PubMed] [Google Scholar]
- 177.Parilla J.H., Hull R.L., Zraika S. neprilysin deficiency is associated with expansion of Islet β-cell mass in high fat-fed mice. J. Histochem. Cytochem. 2018;66:523–530. doi: 10.1369/0022155418765164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Sagnella G.A. Atrial natriuretic peptide mimetics and vasopeptidase inhibitors. Cardiovasc. Res. 2001;51:416–428. doi: 10.1016/s0008-6363(01)00289-9. [DOI] [PubMed] [Google Scholar]
- 179.Nishikimi T., Maeda N., Matsuoka H. The role of natriuretic peptides in cardioprotection. Cardiovasc. Res. 2006;69:318–328. doi: 10.1016/j.cardiores.2005.10.001. [DOI] [PubMed] [Google Scholar]
- 180.McMurray J.J.V., Packer M., Desai A.S., Gong J., Lefkowitz M.P., Rizkala A.R., Rouleau J.L., Shi V.C., Solomon S.D., Swedberg K., Zile M.R. Angiotensin-neprilysin inhibition versus enalapril in heart failure. N. Engl. J. Med. 2014;371:993–1004. doi: 10.1056/NEJMoa1409077. [DOI] [PubMed] [Google Scholar]
- 181.Griggs D.W., Prinsen M.J., Oliva J., Campbell M.A., Arnett S.D., Tajfirouz D., Ruminski P.G., Yu Y., Bond B.R., Ji Y., Neckermann G., Choy R.K.M., De Hostos E., Meyers M.J. Pharmacologic comparison of clinical neutral endopeptidase inhibitors in a rat model of acute secretory diarrheas. J. Pharmacol. Exp. Ther. 2016;357:423–431. doi: 10.1124/jpet.115.231167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Y. Khder, V. Shi, J.J.V. McMurray, M.P. Lefkowitz, Sacubitril/valsartan (LCZ696) in heart failure, in: Handb. Exp. Pharmacol., Springer New York LLC, 2017, pp. 133–165. [DOI] [PubMed]
- 183.McDowell G., Nicholls D.P. The therapeutic potential of candoxatril, a neutral endopeptidase inhibitor, in humans. Cardiovasc. Drug Rev. 2000;18:259–270. [Google Scholar]
- 184.Mills J., Vardeny O. The role of neprilysin inhibitors in cardiovascular disease. Curr. Heart Fail. Rep. 2015;12:389–394. doi: 10.1007/s11897-015-0270-8. [DOI] [PubMed] [Google Scholar]
- 185.Parilla J.H., Mongovin S., Barrow B., Esser N., Zraika S. Pharmacological neprilysin inhibition improves glucose homeostasis in a mouse model of type 2 diabetes. Diabetes. 2018;67:1825. [Google Scholar]
- 186.Marr R.A., Hafez D.M. Amyloid-beta and Alzheimer’s disease: the role of neprilysin-2 in amyloid-beta clearance. Front. Aging Neurosci. 2014;6:187. doi: 10.3389/fnagi.2014.00187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Howell S., Nalbantoglu J., Crine P. Neutral endopeptidase can hydrolyze β-amyloid (1–40) but shows no effect on β-amyloid precursor protein metabolism. Peptides. 1995;16:647–652. doi: 10.1016/0196-9781(95)00021-b. [DOI] [PubMed] [Google Scholar]
- 188.N. Iwata, S. Tsubuki, Y. Takaki, K. Shirotani, B. Lu, N.P. Gerard, C. Gerard, E. Hama, H.J. Lee, T.C. Saido, Metabolic regulation of brain Aβ by neprilysin, Science (80-.). 292 (2001) 1550–1552. [DOI] [PubMed]
- 189.Sorrentino P., Iuliano A., Polverino A., Jacini F., Sorrentino G. The dark sides of amyloid in Alzheimer’s disease pathogenesis. FEBS Lett. 2014;588:641–652. doi: 10.1016/j.febslet.2013.12.038. [DOI] [PubMed] [Google Scholar]
- 190.Oh J.H., Choi S., Shin J., Park J.S. Protective effect of recombinant soluble neprilysin against β-amyloid induced neurotoxicity. Biochem. Biophys. Res. Commun. 2016;477:614–619. doi: 10.1016/j.bbrc.2016.06.103. [DOI] [PubMed] [Google Scholar]
- 191.Jacob S., Deyo D.J., Cox R.A., Traber D.L., Herndon D.N., Hawkins H.K. Mechanisms of toxic smoke inhalation and burn injury: role of neutral endopeptidase and vascular leakage in mice. Toxicol. Mech. Methods. 2009;19:191–196. doi: 10.1080/15376510902725649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Sio S.W.S., Moochhala S., Lu J., Bhatia M. Early protection from burn-induced acute lung injury by deletion of preprotachykinin-A gene. Am. J. Respir. Crit. Care Med. 2010;181:36–46. doi: 10.1164/rccm.200907-1073OC. [DOI] [PubMed] [Google Scholar]
- 193.Reyes L.J., Majó J., Perich D., Morell F. Neuroendocrine cell hyperplasia as an unusual form of interstitial lung disease. Respir. Med. 2007;101:1840–1843. doi: 10.1016/j.rmed.2005.10.024. [DOI] [PubMed] [Google Scholar]
- 194.Sanaee M.S., O’Byrne P.M., Nair P. Diffuse idiopathic pulmonary neuroendocrine hyperplasia, chronic eosinophilic pneumonia, and asthma. Eur. Respir. J. 2009;34:1489–1492. doi: 10.1183/09031936.00109409. [DOI] [PubMed] [Google Scholar]
- 195.Sunday M.E. Oxygen, gastrin-releasing Peptide, and pediatric lung disease: life in the balance. Front. Pediatr. 2014;2:72. doi: 10.3389/fped.2014.00072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Aguayo S.M., Kane M.A., Miller Y.E. Increased pulmonary neuroendocrine cells with bombesin-like immunoreactivity in adult patients with eosinophilic granuloma. J. Clin. Invest. 1990;86 doi: 10.1172/JCI114782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Van Lommel A. Pulmonary neuroendocrine cells (PNEC) and neuroepithelial bodies (NEB): chemoreceptors and regulators of lung development. Paediatr. Respir. Rev. 2001;2:171–176. doi: 10.1053/prrv.2000.0126. [DOI] [PubMed] [Google Scholar]
- 198.Song H., Yao E., Lin C., Gacayan R., Chen M.H., Chuang P.T. Functional characterization of pulmonary neuroendocrine cells in lung development, injury, and tumorigenesis. Proc. Natl. Acad. Sci. U. S. A. 2012;109:17531–17536. doi: 10.1073/pnas.1207238109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.K. Branchfield, L. Nantie, J.M. Verheyden, P. Sui, M.D. Wienhold, X. Sun, Pulmonary neuroendocrine cells function as airway sensors to control lung immune response, Science (80-.). 351 (2016) 707–710. [DOI] [PMC free article] [PubMed]
- 200.Ganter M.T., Pittet J.F. Bombesin-like peptides: modulators of inflammation in acute lung injury? Am. J. Respir. Crit. Care Med. 2006;173:1–2. doi: 10.1164/rccm.2510002. [DOI] [PubMed] [Google Scholar]
- 201.Czepielewski R.S., Porto B.N., Rizzo L.B., Roesler R., Abujamra A.L., Pinto L.G., Schwartsmann G., Cunha F.D.Q., Bonorino C. Gastrin-releasing peptide receptor (GRPR) mediates chemotaxis in neutrophils. Proc. Natl. Acad. Sci. U. S. A. 2012;109:547–552. doi: 10.1073/pnas.1110996109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.W. MacNee, Pulmonary neutrophil kinetics., Clin. Phys. Physiol. Meas. 11 Suppl A (1990) 133–139. [DOI] [PubMed]
- 203.Summers C., Singh N.R., White J.F., Mackenzie I.M., Johnston A., Solanki C., Balan K.K., Peters A.M., Chilvers E.R. Pulmonary retention of primed neutrophils: a novel protective host response, which is impaired in the acute respiratory distress syndrome. Thorax. 2014;69:623–629. doi: 10.1136/thoraxjnl-2013-204742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Bordon J., Aliberti S., Fernandez-Botran R., Uriarte S.M., Rane M.J., Duvvuri P., Peyrani P., Morlacchi L.C., Blasi F., Ramirez J.A. Understanding the roles of cytokines and neutrophil activity and neutrophil apoptosis in the protective versus deleterious inflammatory response in pneumonia. Int. J. Infect. Dis. 2013;17:e76–e83. doi: 10.1016/j.ijid.2012.06.006. [DOI] [PubMed] [Google Scholar]
- 205.M.A. Shipp, G.E. Tarr, C.Y. Chen, S.N. Switzer, L.B. Hersh, H. Stein, M.E. Sunday, E.L. Reinherz, CD10/neutral endopeptidase 24.11 hydrolyzes bombesin-like peptides and regulates the growth of small cell carcinomas of the lung, Proc. Natl. Acad. Sci. U. S. A. 88 (1991) 10662–10666. [DOI] [PMC free article] [PubMed]
- 206.J.C. Connelly, R.A. Skidgel, W.W. Schulz, A.R. Johnson, E.G. Erdös, Neutral endopeptidase 24.11 in human neutrophils: Cleavage of chemotactic peptide, Proc. Natl. Acad. Sci. U. S. A. 82 (1985) 8737–8741. [DOI] [PMC free article] [PubMed]
- 207.R.G. Painter, R. Dukes, J. Sullivan, R. Carter, E.G. Erdos, A.R. Johnson, Function of Neutral Endopeptidase on the Cell Membrane of Human Neutrophils, J. Biol. Chem. 263 (1988) 9456–9461. [PubMed]
- 208.Savill J.S., Wyllie A.H., Henson J.E., Walport M.J., Henson P.M., Haslett C. Macrophage phagocytosis of aging neutrophils in inflammation. Programmed cell death in the neutrophil leads to its recognition by macrophages. J. Clin. Invest. 1989;83:865–875. doi: 10.1172/JCI113970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Pham C.T.N. Neutrophil serine proteases: specific regulators of inflammation. Nat. Rev. Immunol. 2006;6:541–550. doi: 10.1038/nri1841. [DOI] [PubMed] [Google Scholar]
- 210.Meyer-Hoffert U. Neutrophil-derived serine proteases modulate innate immune responses. Front. Biosci. 2009:3409. doi: 10.2741/3462. [DOI] [PubMed] [Google Scholar]
- 211.Reilly C.F., Tewksbury D.A., Schechter N.M., Travis J. Rapid conversion of angiotensin I to angiotensin II by neutrophil and mast cell proteinases. J. Biol. Chem. 1982;257:8619–8622. [PubMed] [Google Scholar]
- 212.Ramaha A., Patston P.A. Release and degradation of angiotensin I and angiotensin II from angiotensinogen by neutrophil serine proteinases. Arch. Biochem. Biophys. 2002;397:77–83. doi: 10.1006/abbi.2001.2687. [DOI] [PubMed] [Google Scholar]
- 213.Owen C.A., Campbell E.J. Angiotensin II generation at the cell surface of activated neutrophils: novel cathepsin G-mediated catalytic activity that is resistant to inhibition. J. Immunol. 1998;160:1436–1443. [PubMed] [Google Scholar]
- 214.Rice G.I., Thomas D.A., Grant P.J., Turner A.J., Hooper N.M. Evaluation of angiotensin-converting enzyme (ACE), its homologue ACE2 and neprilysin in angiotensin peptide metabolism. Biochem. J. 2004;383:45–51. doi: 10.1042/BJ20040634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Allred A.J., Diz D.I., Ferrario C.M., Chappell M.C. Pathways for angiotensin-(1–7) metabolism in pulmonary and renal tissues. Am. J. Physiol. Physiol. 2000;279:F841–F850. doi: 10.1152/ajprenal.2000.279.5.F841. [DOI] [PubMed] [Google Scholar]
- 216.Campbell D.J. Long-term neprilysin inhibition — implications for ARNIs. Nat. Rev. Cardiol. 2017;14:171–186. doi: 10.1038/nrcardio.2016.200. [DOI] [PubMed] [Google Scholar]
- 217.Y. Song, M. Zhang, L. Yin, K. Wang, Y. Zhou, M. Zhou, Y. Lu, COVID-19 treatment: close to a cure ? A rapid review of pharmacotherapies for the novel coronavirus, Med. Pharmacol. (2020) 1–25. [DOI] [PMC free article] [PubMed]
- 218.Voiriot G., Philippot Q., Elabbadi A., Elbim C., Chalumeau M., Fartoukh M. Risks related to the use of non-steroidal anti-inflammatory drugs in community-acquired pneumonia in adult and pediatric patients. J. Clin. Med. 2019;8:786. doi: 10.3390/jcm8060786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Little P. Non-steroidal anti-inflammatory drugs and covid-19. BMJ. 2020;368 doi: 10.1136/bmj.m1185. [DOI] [PubMed] [Google Scholar]
- 220.R. Gupta, A. Misra, Contentious issues and evolving concepts in the clinical presentation and management of patients with COVID-19 infection with reference to use of therapeutic and other drugs used in co-morbid diseases (Hypertension, Diabetes etc), Diabetes Metab. Syndr. Clin. Res. Rev. (2020). [DOI] [PMC free article] [PubMed]
- 221.Day M. Covid-19: ibuprofen should not be used for managing symptoms, say doctors and scientists. BMJ. 2020;368:m1086. doi: 10.1136/bmj.m1086. [DOI] [PubMed] [Google Scholar]
- 222.Sont J.K., Van Krieken J.H.J.M., Van Klink H.C.J., Roldaan A.C., Apap C.R., Willems L.N.A., Sterk P.J. Enhanced expression of neutral endopeptidase (NEP) in airway epithelium in biopsies from steroid- versus nonsteroid-treated patients with atopic asthma. Am. J. Respir. Cell Mol. Biol. 1997;16:549–556. doi: 10.1165/ajrcmb.16.5.9160837. [DOI] [PubMed] [Google Scholar]
- 223.Coutinho A.E., Chapman K.E. The anti-inflammatory and immunosuppressive effects of glucocorticoids, recent developments and mechanistic insights. Mol. Cell. Endocrinol. 2011;335:2–13. doi: 10.1016/j.mce.2010.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Auyeung T.W., Lee J.S.W., Lai W.K., Choi C.H., Lee H.K., Lee J.S., Li P.C., Lok K.H., Ng Y.Y., Wong W.M., Yeung Y.M. The use of corticosteroid as treatment in SARS was associated with adverse outcomes: a retrospective cohort study. J. Infect. 2005;51:98–102. doi: 10.1016/j.jinf.2004.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Y.M. Arabi, Y. Mandourah, F. Al-Hameed, A.A. Sindi, G.A. Almekhlafi, M.A. Hussein, J. Jose, R. Pinto, A. Al-Omari, A. Kharaba, A. Almotairi, K. Al Khatib, B. Alraddadi, S. Shalhoub, A. Abdulmomen, I. Qushmaq, A. Mady, O. Solaiman, A.M. Al-Aithan, R. Al-Raddadi, A. Ragab, H.H. Balkhy, A. Al Harthy, A.M. Deeb, H. Al Mutairi, A. Al-Dawood, L. Merson, F.G. Hayden, R.A. Fowler, Corticosteroid therapy for critically ill patients with middle east respiratory syndrome., Am. J. Respir. Crit. Care Med. 197 (2018) 757–767. [DOI] [PubMed]
- 226.Vincent M.J., Bergeron E., Benjannet S., Erickson B.R., Rollin P.E., Ksiazek T.G., Seidah N.G., Nichol S.T. Chloroquine is a potent inhibitor of SARS coronavirus infection and spread. Virol. J. 2005;2:69. doi: 10.1186/1743-422X-2-69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Touret F., de Lamballerie X. Of chloroquine and COVID-19. Antiviral Res. 2020;177 doi: 10.1016/j.antiviral.2020.104762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.P. Gautret, J.-C. Lagier, P. Parola, V.T. Hoang, L. Meddeb, M. Mailhe, B. Doudier, J. Courjon, V. Giordanengo, V.E. Vieira, H.T. Dupont, S. Honoré, P. Colson, E. Chabrière, B. La Scola, J.-M. Rolain, P. Brouqui, D. Raoult, Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial, Int. J. Antimicrob. Agents. (2020). [DOI] [PMC free article] [PubMed]
- 229.Cortegiani A., Ingoglia G., Ippolito M., Giarratano A., Einav S. A systematic review on the efficacy and safety of chloroquine for the treatment of COVID-19. J. Crit. Care. 2020 doi: 10.1016/j.jcrc.2020.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Ii M., Losordo D.W. Statins and the endothelium. Vascul. Pharmacol. 2007;46:1–9. doi: 10.1016/j.vph.2006.06.012. [DOI] [PubMed] [Google Scholar]
- 231.Zeiser R. Immune modulatory effects of statins. Immunology. 2018;154:69–75. doi: 10.1111/imm.12902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Dashti-Khavidaki S., Khalili H. Considerations for statin therapy in patients with COVID-19. Pharmacotherapy. 2020;40:484–486. doi: 10.1002/phar.2397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.M.R. Goldstein, G.A. Poland, C.W. Graeber, Are certain drugs associated with enhanced mortality in COVID-19?, QJM Int. J. Med. (2020). [DOI] [PMC free article] [PubMed]
- 234.J. Li, Y. Zhang, F. Wang, B. Liu, H. Li, G. Tang, Z. Chang, A. Liu, C. Fu, J. Gao, J. Li, Sex differences in clinical findings among patients with coronavirus disease 2019 (COVID-19) and severe condition, MedRxiv. (2020) 2020.02.27.20027524.
- 235.X. Wang, R. Dhindsa, G. Povysil, A. Zoghbi, J. Motelow, J. Hostyk, D. Goldstein, Transcriptional Inhibition of Host Viral Entry Proteins as a Therapeutic Strategy for SARS-CoV-2, Preprints. (2020).
- 236.Dyall J., Coleman C.M., Hart B.J., Venkataraman T., Holbrook M.R., Kindrachuk J., Johnson R.F., Olinger G.G.J., Jahrling P.B., Laidlaw M., Johansen L.M., Lear-Rooney C.M., Glass P.J., Hensley L.E., Frieman M.B. Repurposing of clinically developed drugs for treatment of Middle East respiratory syndrome coronavirus infection. Antimicrob. Agents Chemother. 2014;58:4885–4893. doi: 10.1128/AAC.03036-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Zhou Y., Hou Y., Shen J., Huang Y., Martin W., Cheng F. Network-based drug repurposing for novel coronavirus 2019-nCoV/SARS-CoV-2. Cell Discov. 2020;6 doi: 10.1038/s41421-020-0153-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Mompeón A., Lázaro-franco M., Bueno-betí C., Pérez- D., Vidal-gómez X., Monsalve E., Gironacci M.M., Novella S. Estradiol, acting through ERα, induces endothelial non-classic renin-angiotensin system increasing angiotensin 1–7 production. Mol. Cell. Endocrinol. 2015;422:1–8. doi: 10.1016/j.mce.2015.11.004. [DOI] [PubMed] [Google Scholar]
- 239.A.H.J. Danser, M. Epstein, D. Batlle, Renin-angiotensin system blockers and the COVID-19 pandemic: at present there is no evidence to abandon renin-angiotensin system blockers, Hypertension. (2020) HYPERTENSIONAHA-120. [DOI] [PMC free article] [PubMed]
- 240.Hammerschmidt S., Kuhn H., Gessner C., Seyfarth H.-J., Wirtz H. Stretch-induced alveolar type II cell apoptosis: role of endogenous bradykinin and PI3K-Akt signaling. Am. J. Respir. Cell Mol. Biol. 2007;37:699–705. doi: 10.1165/rcmb.2006-0429OC. [DOI] [PubMed] [Google Scholar]
- 241.D.A.H. Jan, E. Murray, B. Daniel, Renin-angiotensin system blockers and the COVID-19 pandemic, Hypertension. (2020) HYPERTENSIONAHA-120.
- 242.S. Tian, W. Hu, L. Niu, H. Liu, H. Xu, S.Y. Xiao, Pulmonary pathology of early-phase 2019 novel coronavirus (COVID-19) pneumonia in two patients with lung cancer, J. Thorac. Oncol. (2020). [DOI] [PMC free article] [PubMed]
- 243.Bohler M., Dougherty C., Tachibana T., Gilbert E.R., Cline M.A. Gastrin releasing peptide-induced satiety is associated with hypothalamic and brainstem changes in chicks. Neurosci. Lett. 2019;713 doi: 10.1016/j.neulet.2019.134529. [DOI] [PubMed] [Google Scholar]
- 244.J. Liu, Y. Liu, P. Xiang, L. Pu, H. Xiong, C. Li, M. Zhang, J. Tan, Y. Xu, R. Song, M. Song, L. Wang, W. Zhang, B. Han, L. Yang, X. Wang, G. Zhou, T. Zhang, B. Li, Y. Wang, Z. Chen, X. Wang, Neutrophil-to-Lymphocyte Ratio Predicts Severe Illness Patients with 2019 Novel Coronavirus in the Early Stage, MedRxiv. 807 (2020) 2020.02.10.20021584. doi:10.1101/2020.02.10.20021584.
- 245.Pan L., Mu M., Yang P., Sun Y., Wang R., Yan J., Li P., Hu B., Wang J., Hu C., Jin Y., Niu X., Ping R., Du Y., Li T., Xu G., Hu Q., Tu L. Clinical characteristics of COVID-19 patients with digestive symptoms in Hubei, China: a descriptive, cross-sectional, multicenter study. Am J Gastroenterol. 2020;20 doi: 10.14309/ajg.0000000000000620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Courtney J.M., Ennis M., Elborn J.S. Cytokines and inflammatory mediators in cystic fibrosis. J. Cyst. Fibros. 2004;3:223–231. doi: 10.1016/j.jcf.2004.06.006. [DOI] [PubMed] [Google Scholar]
- 247.Yu J. Inflammatory mechanisms in the lung. J. Inflamm. Res. 2008;2:1. [PMC free article] [PubMed] [Google Scholar]
- 248.D’Uscio L.V., Shaw S., Barton M., Lüscher T.F. Losartan but not verapamil inhibits angiotensin II-induced tissue endothelin-1 increase role of blood pressure and endothelial function. Hypertension. 1998;31:1305–1310. doi: 10.1161/01.hyp.31.6.1305. [DOI] [PubMed] [Google Scholar]
- 249.Shah A., Gul R., Yuan K., Gao S., Bin Oh Y., Kim U.H., Kim S.H. Angiotensin-(1–7) stimulates high atrial pacing-induced ANP secretion via Mas/PI3-kinase/Akt axis and Na+/H+ exchanger. Am. J. Physiol. - Hear. Circ. Physiol. 298. 2010 doi: 10.1152/ajpheart.00608.2009. [DOI] [PubMed] [Google Scholar]
- 250.Bindom S.M., Lazartigues E. The sweeter side of ACE2: Physiological evidence for a role in diabetes. Mol. Cell. Endocrinol. 2009;302:193–202. doi: 10.1016/j.mce.2008.09.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Z. Lang, C.G. Murlas, Dexamethasone Increases Airway Epithelial Cell Neutral Endopeptidase by Enhancing Transcription and New Protein Synthesis, 1993. [DOI] [PubMed]
- 252.Wang Z., Zhang X.J., Li T., Li J., Tang Y., Le W. Valproic acid reduces neuritic plaque formation and improves learning deficits in APPSwe/PS1A246E transgenic mice via preventing the prenatal hypoxia-induced down-regulation of neprilysin. CNS Neurosci. Ther. 2014;20:209–217. doi: 10.1111/cns.12186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Klein C., Roussel G., Brun S., Rusu C., Patte-Mensah C., Maitre M., Mensah-Nyagan A.G. 5-HIAA induces neprilysin to ameliorate pathophysiology and symptoms in a mouse model for Alzheimer’s disease. Acta Neuropathol. Commun. 2018;6:136. doi: 10.1186/s40478-018-0640-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254.Yao M., Nguyen T.V.V., Rosario E.R., Ramsden M., Pike C.J. Androgens regulate neprilysin expression: Role in reducing β-amyloid levels. J. Neurochem. 2008;105:2477–2488. doi: 10.1111/j.1471-4159.2008.05341.x. [DOI] [PubMed] [Google Scholar]
- 255.Xiao Z.M., Sun L., Liu Y.M., Zhang J.J., Huang J. Estrogen regulation of the neprilysin gene through a hormone-responsive element. J. Mol. Neurosci. 2009;39:22–26. doi: 10.1007/s12031-008-9168-1. [DOI] [PubMed] [Google Scholar]
- 256.Ayoub S., Melzig M.F. Influence of selected natural products on neutral endopeptidase activity and β-Amyloid production in SK-N-SH cells. Pharm. Biol. 2008;46:425–432. [Google Scholar]
- 257.El-Sayed N.S., Bayan Y. Possible role of Resveratrol targeting Estradiol and Neprilysin pathways in Lipopolysaccharide model of Alzheimer disease. Adv. Exp. Med. Biol. 2015;822:107–118. doi: 10.1007/978-3-319-08927-0_12. [DOI] [PubMed] [Google Scholar]
- 258.Chang X., Rong C., Chen Y., Yang C., Hu Q., Mo Y., Zhang C., Gu X., Zhang L., He W., Cheng S., Hou X., Su R., Liu S., Dun W., Wang Q., Fang S. (-)-Epigallocatechin-3-gallate attenuates cognitive deterioration in Alzheimer’s disease model mice by upregulating neprilysin expression. Exp. Cell Res. 2015;334:136–145. doi: 10.1016/j.yexcr.2015.04.004. [DOI] [PubMed] [Google Scholar]
- 259.Yuan H.F., Li X., Quan Q.K., Wang N.N., Li Y., Li M. Effects of Naoerkang on expressions of β-amyloid peptide 1–42 and neprilysin in hippocampus in a rat model of Alzheimer’s disease. J. Chinese Integr. Med. 2010;8:152–157. doi: 10.3736/jcim20100210. [DOI] [PubMed] [Google Scholar]
- 260.Mahmud-Al-Rafat A., Majumder A., Taufiqur Rahman K.M., Mahedi Hasan A.M., Didarul Islam K.M., Taylor-Robinson A.W., Billah M.M. Decoding the enigma of antiviral crisis: does one target molecule regulate all? Cytokine. 2019;115:13–23. doi: 10.1016/j.cyto.2018.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 261.Lam C.W.K., Chan M.H.M., Wong C.K. Severe acute respiratory syndrome: clinical and laboratory manifestations. Clin. Biochem. Rev. 2004;25:121–132. [PMC free article] [PubMed] [Google Scholar]
- 262.Rider P., Carmi Y., Cohen I. Biologics for targeting inflammatory cytokines, clinical uses, and limitations. Int. J. Cell Biol. 2016;2016:9259646. doi: 10.1155/2016/9259646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 263.van der Meer J.W.M., Popa C., Netea M.G. Side effects of anticytokine strategies. Neth. J. Med. 2005;63:78–80. [PubMed] [Google Scholar]
- 264.K. Frie, K. Gbinigie, Chloroquine and hydroxychloroquine: current evidence for their effectiveness in treating COVID-19, Cent. Evidence-Based Med. Nuff. Dep. Prim. Care Heal. Sci. Univ. Oxford--25 Maart. 2020 (2020).
- 265.Devaux C.A., Rolain J.-M., Colson P., Raoult D. New insights on the antiviral effects of chloroquine against coronavirus: what to expect for COVID-19? Int. J. Antimicrob. Agents. 2020;2020 doi: 10.1016/j.ijantimicag.2020.105938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 266.Maximov P.Y., Lee T.M., Jordan V.C. The discovery and development of selective estrogen receptor modulators (SERMs) for clinical practice. Curr. Clin. Pharmacol. 2013;8:135–155. doi: 10.2174/1574884711308020006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 267.B.J. Delgado, W. Lopez-Ojeda, Estrogen, in: StatPearls [Internet], 2019.
- 268.Nadeem S., Batisky D.L. Aliskiren, the first direct renin inhibitor: assessing a role in pediatric hypertension and kidney diseases. Pediatr. Nephrol. 2014;29:2105–2111. doi: 10.1007/s00467-013-2716-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 269.Meng Y., Yu C.-H., Li W., Li T., Luo W., Huang S., Wu P.-S., Cai S.-X., Li X. Angiotensin-converting enzyme 2/angiotensin-(1–7)/Mas axis protects against lung fibrosis by inhibiting the MAPK/NF-kappaB pathway. Am. J. Respir. Cell Mol. Biol. 2014;50:723–736. doi: 10.1165/rcmb.2012-0451OC. [DOI] [PubMed] [Google Scholar]
- 270.Zhu J.-R., Sun N.-L., Yang K., Hu J., Xu G., Hong H., Wang R., Tu Y.-M., Ritter S., Keefe D. Efficacy and safety of aliskiren, a direct renin inhibitor, compared with ramipril in Asian patients with mild to moderate hypertension. Hypertens. Res. 2012;35:28–33. doi: 10.1038/hr.2011.150. [DOI] [PubMed] [Google Scholar]
- 271.Mourad J.-J., Levy B.I. Interaction between RAAS inhibitors and ACE2 in the context of COVID-19. Nat. Rev. Cardiol. 2020;1 doi: 10.1038/s41569-020-0368-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 272.Wang H.H., Shieh M.J., Liao K.F. A blind, randomized comparison of racecadotril and loperamide for stopping acute diarrhea in adults. World J. Gastroenterol. 2005;11:1540–1543. doi: 10.3748/wjg.v11.i10.1540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 273.Lehert P., Chéron G., Calatayud G.A., Cézard J.P., Castrellón P.G., Garcia J.M.M., Santos M., Savitha M.R. Racecadotril for childhood gastroenteritis: an individual patient data meta-analysis. Dig. Liver Dis. 2011;43:707–713. doi: 10.1016/j.dld.2011.03.001. [DOI] [PubMed] [Google Scholar]
- 274.Adum J.P.S., Arora R. Role of neprilysin inhibitors in heart failure. Am. J. Ther. 2015;24:e737–e743. doi: 10.1097/MJT.0000000000000354. [DOI] [PubMed] [Google Scholar]
- 275.Chang P.C., Lin S.F., Chu Y., Wo H.T., Lee H.L., Huang Y.C., Wen M.S., Chou C.C. LCZ696 therapy reduces ventricular tachyarrhythmia inducibility in a myocardial infarction-induced heart failure rat model. Cardiovasc. Ther. 2019;2019 doi: 10.1155/2019/6032631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 276.Pfau D., Thorn S.L., Zhang J., Mikush N., Renaud J.M., Klein R., DeKemp R.A., Wu X., Hu X., Sinusas A.J., Young L.H., Tirziu D. Angiotensin receptor neprilysin inhibitor attenuates myocardial remodeling and improves infarct perfusion in experimental heart failure. Sci. Rep. 2019;9:1–13. doi: 10.1038/s41598-019-42113-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 277.L.M. Burrell, J. Droogh, O. Man In’t Veld, M.D. Rockell, N.K. Farina, C.I. Johnston, Antihypertensive and antihypertrophic effects of omapatrilat in SHR, Am. J. Hypertens. 13 (2000) 1110–1116. [DOI] [PubMed]
- 278.Campese V.M., Lasseter K.C., Ferrario C.M., Smith W.B., Ruddy M.C., Grim C.E., Smith R.D., Vargas R., Habashy M.F., Vesterqvist O., Delaney C.L., Liao W.-C. Omapatrilat versus lisinopril. Hypertension. 2001;38:1342–1348. doi: 10.1161/hy1201.096569. [DOI] [PubMed] [Google Scholar]
- 279.I. Rodriguez-Gomez, R. Wangensteen, N.M. Atucha, F. O’valle, R.G. Del Moral, J. Garcia-Estañ, F. Vargas, A. Osuna, Effects of Omapatrilat on Blood Pressure and Renal Injury in L-NAME and L-NAME Plus DOCA-Treated Rats, Am. J. Hypertens. 16 (2003) 33–38. [DOI] [PubMed]
- 280.Kostis J.B., Packer M., Black H.R., Schmieder R., Henry D., Levy E. Omapatrilat and enalapril in patients with hypertension: the Omapatrilat Cardiovascular Treatment vs. Enalapril (OCTAVE) trial. Am. J. Hypertens. 2004;17:103–111. doi: 10.1016/j.amjhyper.2003.09.014. [DOI] [PubMed] [Google Scholar]
- 281.Seferovic J., Seidelmann S., Clagett B., Packer M., Zile M., Rouleau J., Swedberg K., Lefkowitz M., Shi V., Desai A. Influence of Sacubtril/Valsartan on glycemic control in patients with type 2 diabetes and heart failure with reduced ejection fraction. J. Am. Coll. Cardiol. 2017;69:682. [Google Scholar]
- 282.Uijl E., Hart D.C., Roksnoer L.C.W., Clahsen-van Groningen M.C., van Veghel R., Garrelds I.M., de Vries R., van der Vlag J., Zietse R., Nijenhuis T., Joles J.A., Hoorn E.J., Danser A.H.J. Angiotensin–neprilysin inhibition confers renoprotection in rats with diabetes and hypertension by limiting podocyte injury. J. Hypertens. 2019;38:1. doi: 10.1097/HJH.0000000000002326. [DOI] [PubMed] [Google Scholar]
- 283.Park M.H., Lee J.K., Choi S., Ahn J., Jin H.K., Park J.-S., Bae J. Recombinant soluble neprilysin reduces amyloid-beta accumulation and improves memory impairment in Alzheimer’s disease mice. Brain Res. 2013;1529:113–124. doi: 10.1016/j.brainres.2013.05.045. [DOI] [PubMed] [Google Scholar]
- 284.S. Sturiale, G. Barbara, B. Qiu, M. Figini, P. Geppetti, N. Gerard, C. Gerard, E.F. Grady, N.W. Bunnett, S.M. Collins, Neutral endopeptidase (EC 3.4.24.11) terminates colitis by degrading substance P, Proc. Natl. Acad. Sci. U. S. A. 96 (1999) 11653–11658. [DOI] [PMC free article] [PubMed]
- 285.Barbara G., De Giorgio R. Neutral endopeptidase (EC 3.4.24.11) downregulates the onset of intestinal inflammation in the nematode infected mouse. Inflamm. Inflamm. Bowel Dis. 2003;52:1457–1464. doi: 10.1136/gut.52.10.1457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 286.Bunn P.A., Helfrich B.A., Brenner D.G., Chan D.C., Dykes D.J., Cohen A.J., Miller Y.E. Effects of recombinant neutral endopeptidase (EC 3.4.24.11) on the growth of lung cancer cell lines in vitro and in vivo. Clin. Cancer Res. 1998;4:2849–2858. [PubMed] [Google Scholar]
- 287.Lightner A.M., Jordan T.H., Bunnett N.W., Grady E.F., Kirkwood K.S. Recombinant human neutral endopeptidase ameliorates pancreatic elastase-induced lung injury. Surgery. 2002;132:193–199. doi: 10.1067/msy.2002.125309. [DOI] [PubMed] [Google Scholar]
- 288.Kohrogi H., Nadel J.A., Malfroy B., Gorman C., Bridenbaugh R., Patton J.S., Borson D.B. Recombinant human enkephalinase (neutral endopeptidase) prevents cough induced by tachykinins in awake guinea pigs. J. Clin. Invest. 1989;84:781–786. doi: 10.1172/JCI114236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 289.Kusner E.J., Buckner C.K., DeHaas C.J., Lengel D.J., Marks R.L., Krell R.D. Tachykinin-induced dyspnea in conscious guinea pigs. Eur. J. Pharmacol. 1992;210:299–306. doi: 10.1016/0014-2999(92)90419-5. [DOI] [PubMed] [Google Scholar]