Abstract
The majority of deaths due to Streptococcus pneumoniae infections are in developing countries. Although polysaccharide-based pneumococcal vaccines are available, newer types of vaccines are needed to increase vaccine affordability, particularly in developing countries, and to provide broader protection across all pneumococcal serotypes. To attenuate pneumococcal virulence with the aim of engineering candidate live attenuated vaccines (LAVs), we constructed knockouts in S. pneumoniae D39 of one of the capsular biosynthetic genes, cpsE that encodes glycosyltransferase, and the endonuclease gene, endA, that had been implicated in the uptake of DNA from the environment as well as bacterial escape from neutrophil-mediated killing. The cpsE gene knockout significantly lowered peak bacterial density, BALB/c mice nasopharyngeal (NP) colonisation but increased biofilm formation when compared to the wild-type D39 strain as well as the endA gene knockout mutant. All constructed mutant strains were able to induce significantly high serum and mucosal antibody response in BALB/c mice. However, the cpsE-endA double mutant strain, designated SPEC, was able to protect mice from high dose mucosal challenge of the D39 wild-type. Furthermore, SPEC showed 23-fold attenuation of virulence compared to the wild-type. Thus, the cpsE-endA double-mutant strain could be a promising candidate for further development of a LAV for S. pneumoniae.
Keywords: Streptococcus pneumoniae, vaccine, live attenuated vaccine, capsule biosynthesis gene, cpsE, endonuclease A, endA
1. Introduction
Streptococcus pneumoniae (pneumococcus) is responsible for about 1.2 million deaths globally, more than half of which happened in children aged <5 years [1]. Most pneumococcal-implicated deaths happened in developing countries such as India, Nigeria, the Democratic Republic of the Congo, and Pakistan, which constitute 49% of the global pneumococcal fatalities [2]. Mortalities are mainly due to pneumococcal pneumonia (81%) with the rest attributed to meningitis (12%), bacteraemia, and sepsis (7%) [2,3,4]. Even with modern antimicrobial treatment, survivors of invasive pneumococcal diseases usually develop long term complications such as hearing loss and neuropsychological impairments [5]. Still, these alarming figures of pneumococcal morbidity and mortality might underestimate the actual burden of pneumococcal diseases because many developing countries lack sensitive diagnostic tests for detecting the disease aetiology.
Currently, two types of vaccines are used to prevent pneumococcal diseases: pneumococcal conjugate vaccines (PCV10 and PCV13) and pneumococcal polysaccharide vaccines (PPV23). The use of these vaccines has led to a considerable reduction in mortality ascribed to pneumococcal infections [2]. These vaccines are designed to generate antibodies against the capsular polysaccharide of the pneumococcus [6] which is structurally distinct among S. pneumoniae strains [7,8]. Currently used PCVs were initially designed for use in Western industrialized countries. Therefore, the vaccines would thus include the dominant serotypes causing invasive disease in those regions that are not well matched to other geographic areas of the world such as Asia and Africa. For example, in countries of the Asia–Pacific region, the potential serotype coverage of PCV13 ranges from 30% to 96.5% due to dissimilar serotype prevalence in different parts of the region [9]. Inclusion of new serotypes into the conjugate vaccine formula is a complex task and may further increase the price of the vaccine, thereby limiting its affordability for developing countries and would therefore not be a reasonable way of solving the problem. Even now, out of 144 countries which recently added PCV to their national immunisation programs, only 60 countries are categorized as low- or middle-income countries which could not afford PCV immunisation without support from the Gavi, the Vaccine Alliance [10,11].
Although vaccine serotypes have decreased after the introduction of the PCV’s, there has been an increase in non-vaccine serotypes in both carriage and invasive diseases due to serotype replacement [12,13] and clonal expansion [12,14,15,16,17]. Additionally, the polysaccharide nature of the vaccines, as compared to pure protein or peptide vaccines, does not provide efficient immunologic memory as T cell major histocompatibility complex (MHC) II receptors can only bind peptide antigens presented by B cell receptors [18]. Therefore, the immune response to polysaccharide vaccines is traditionally reported as T cell-independent and conjugation of polysaccharides to protein conjugates provides a T cell response [19]. Despite this, PCVs remain less efficient against more common non-invasive pneumococcal disease such as middle-ear infections, sinusitis, and bronchitis in comparison to invasive pneumococcal disease (IPD) [20,21].
The limitations outlined above necessitate the development of a serotype-independent vaccine that would confer protection in a serotype-independent manner against both mucosal and invasive pneumococcal disease, while keeping the cost sufficiently low to make it accessible for developing countries. S. pneumoniae live attenuated vaccines (LAVs) would ideally colonise in the upper respiratory tract of the host and induce effective immune protection by activating all phases of the immune system, provide more durable immunity that require boosters less frequently and have a lower cost. Attenuated vaccines are considered the most effective type of vaccine that induce durable immunity against diseases, with a lessened need for booster doses [22]. Unencapsulation is one of the effective strategies to attenuate the bacteria and to expose pneumococcal surface proteins to the immune system. In previous preclinical studies, mice immunised with unencapsulated strains of S. pneumoniae induced a robust immune response and protected the immunised animals against wild-type challenges [23,24]. We chose to knockout the cpsE gene as mutations in the gene has been associated with capsule loss, and increased pneumococcal colonisation, which is one of the predictors of effective immune response [25].
One of the biggest challenges in constructing LAVs is choosing a strategy that, besides attenuating virulence, would also eliminate the risk of reversion of the vaccine strain to a fully virulent strain because of the natural competence for transformation and the genetic plasticity of the pneumococcal genome. Therefore, besides unencapsulation by targeting one of the capsule synthesis genes (cpsE gene) we used a second knock out of the endonuclease endA gene that focuses on the safety of the candidate vaccine strains. Mutations in the endA gene in pneumococci have been reported to show decreased competence for natural transformation [26,27], thereby minimizing the potential for reversion of the constructed candidate vaccine strain into the wild-type. The goal of the current study was to determine the immune protection and attenuation of the constructed endA and cpsE gene knockout strains as a means of assessing their suitability as a candidate LAV.
2. Materials and Methods
2.1. Bacterial Strain and Growth Conditions
Streptococcus pneumoniae was grown in Todd-Hewitt broth (THB; Oxoid, Basingstoke, Hampshire, UK) supplemented with 0.5% yeast extract (Oxoid, Basingstoke, Hampshire, UK), brain heart infusion (BHI) broth (Oxoid, Basingstoke, Hampshire, UK), or on blood agar (BA) plates (Isolab, Shah Alam, Selangor, Malaysia) at 37 °C and in 5% CO2. For storage, S. pneumoniae were grown in BHI broth at 37 °C to an OD600 of around 0.5, and 800 μL culture was frozen with the addition of 200 μL 80% glycerol (final concentration of glycerol in the stock media was 20%) and kept at −80 °C.
2.2. Construction of cpsE::tetL and endA::aphA-3 Knockout Mutants
Knockout mutants of cpsE and endA genes in S. pneumoniae strain National Collection of Type Cultures (NCTC) 7466 (referred to as strain D39, serotype 2) were constructed by homologous recombination with disrupted gene modules as described previously [28,29,30]. Briefly, the disrupted gene modules were constructed by firstly amplifying the flanking regions (few hundred base-pairs) of the target genes which were then ligated to a selectable marker in the middle. The middle selectable marker used was either the tetL tetracycline resistance gene (to interrupt the cpsE gene) or the aphA3 kanamycin resistance gene (to disrupt the endA gene) which were amplified from plasmid pMV158GFP [31] or from the genomic DNA of S. pneumoniae strain CP2460 (kindly gifted by Professor Don Morrison, University of Illinois, Chicago, IL, USA), respectively. In the second step, all the three PCR products were combined into one molecule by a splicing overlap extension-PCR (SOE-PCR) reaction. The resulting SOE-PCR product was validated by conventional Sanger dideoxy sequencing using appropriate primers prior to transformation into wild-type S. pneumoniae D39. Competence of S. pneumoniae D39 was controlled by the appropriate addition of competence-specific peptide [24] and transformation was performed as described previously [32]. Competence media used contained Todd-Hewitt broth supplemented with 0.2% yeast extract (THY), 0.01% CaCl2, 0.2% bovine serum albumin (BSA) and 100 ng/mL competence-stimulating peptide 1 (CSP-1; EMRLSKFFRDFILQRKK). Briefly, frozen pneumococcal competent cells (20 µL) were thawed on ice, diluted 1:10 in the competence medium (180 µL), and the cells were incubated at 37 °C for 15 min. Subsequently 10 µl of purified recombinant gene disruption modules (either cpsE::tetL or endA::aphA-3 of approximately 30–50 ng) were added and the cells were incubated for 3 h at 37 °C before plating them on THY agar plates containing tetracycline (1.25 µg/mL) for the selection of S. pneumoniae D39 cpsE::tetL knockout mutants or kanamycin (200 μg/mL) to select S. pneumoniae D39 endA::aphA-3 knockout mutants. Transformants obtained were validated by PCR and sequencing of the PCR products. To obtain the double knockout mutant, the D39 (cpsE::tetL) mutant, designated SPC, was made competent and was transformed with the endA::aphA-3 module. The double knockout mutant was then selected on THY supplemented with both tetracycline and kanamycin and validated by PCR-sequencing.
2.3. Transmission Electron Microscopy
Transmission electron microscopy was used to determine the presence of the capsule in the constructed mutant S. pneumoniae strains. Samples were prepared by lysine-acetate-based formaldehyde-glutaraldehyde ruthenium red-osmium fixation procedure (LRR fixation) as described previously [33]. The samples were examined with a transmission electron microscope (FeiTecnai G2 Spirit BIO TWIN, Hillsboro, OR, USA) at an acceleration voltage of 80 kV.
2.4. In Vitro Studies
Growth of the constructed mutant strains was assessed against wild type S. pneumoniae D39. The strains were grown in 5 mL BHI at 37 °C in 5% CO2 overnight in 15-mL tubes. From the overnight culture, 100 µL was transferred into a 15-mL tube containing 5 mL THY broth and grown to mid-log phase (OD600 = 0.5). Subsequently, 100 µL of the mid-log phase culture was transferred into a fresh sterile tube containing 5 mL THY and the optical density at 600 nm was measured every 30 min for 12 h using a spectrophotometer (Eppendorf BioSpectrometer plus, Hamburg, Germany).
Biofilm formation analysis of the gene knockout strains was compared to the wild-type D39 strain using the Method described by Yadav, et al. [34]. Briefly, the constructed mutant strains and wild-type S. pneumoniae D39 were grown to a mid-logarithmic phase (OD600 = 0.4) and after 1:1000 dilution, 200 µL were transferred to a 96-well polystyrene microtiter plates. The plates were incubated at 37 °C in 5% CO2 and biofilm formation was assessed at 6, 12, 18, 24 and 36 h using the crystal violet assay as described elsewhere [34]. Optical density was measured at 570 nm using an Infinite M200 PRO Microplate Reader (Tecan, Männedorf, Switzerland). The biofilm assays were performed in triplicates and the means ± standard deviations were calculated. Background absorbance was compensated by subtracting the average values of crystal violet with the values obtained in sterile medium.
2.5. In Vivo Studies
Ethical approval (UAPREC/04/008) was obtained from the University Animal and Plant Research Ethics Committee to conduct experiments on animal models. The colonisation potential, immunogenicity, and protection against mucosal challenges of the constructed mutant S. pneumoniae strains were performed in BALB/c mice. For colonisation assay, six 10-12 week-old BALB/c mice in each group was inoculated with 10 μL phosphate-buffered saline (PBS) containing 107 colony forming units (CFU)/mL of the relevant bacterial suspensions intranasally and at the 2nd, 10th, and 18th days post-inoculation, the mice were sacrificed, the nasopharyngeal (NP) fluid was collected, and bacteria were quantified by plating the NP fluid on BHI agar plates supplemented with 5% defibrinated blood and neomycin (20 μg/mL) to suppress the growth of contaminants.
Mice immunisation was carried out by previously published methods (Rosch et al., 2007; Yasser 2013). The antibody titres against S. pneumoniae were determined by whole cell ELISA. Briefly, 5 groups of mice (each group consisting of 6 mice) was immunised with the constructed gene knockout strains, i.e., D39 (cpsE::tetL) (designated SPC), D39 (endA::aphA3) (designated SPE), and D39 (cpsE::tetL; endA::aphA3) (designated SPEC), and wild type D39 strain as a positive control to assess their ability to induce effective antibody responses. Mice were intranasally immunised by administering 10 µL PBS containing 1 × 107 CFU of the bacteria in two doses, two weeks apart. Three weeks after the second dose, samples of blood and NP lavage fluid were collected for the measurement of antibody concentrations by enzyme-linked immunosorbent assay ELISA. Three-week periods were chosen because after 10 to 14 days following antigen exposure, IgG antibodies to protein antigens first appear in the blood and peaks within 4 to 6 weeks after primary immunisation [18]. Briefly, whole cell ELISA was conducted by fixing wild-type S. pneumoniae D39 in 96-well plates as antigens and blocking with 1% BSA. Subsequently, serum or nasal washes was added in 10-fold serial dilutions. Goat anti-Mouse IgG (Fc) and IgA (alpha chain) secondary antibodies were added and antibodies were measured with a 2,2’-azinobis (3-ethylbenzothiazoline-6-sulfonic acid)-diammonium salt (ABNS) substrate solution in citrate-phosphate buffer. Readings were carried out using an Infinite M200 PRO Multimode Microplate Reader at 493 nm and samples were considered negative for the presence of specific antibodies when OD493 < 0.1.
Protection against high dose intranasal challenge with the wild-type D39 strain was assessed after three weeks of immunisation. BALB/c mice were challenged with approximately 5 × 108 CFU of wild-type S. pneumoniae D39 intranasally. Survival time (h) and rate of survival (%) was calculated using the Kaplan–Meier survival test.
The median lethal dose (LD50) in colony-forming units (CFU) was measured to evaluate the effect of the endA and cpsE mutation on the virulence of S. pneumoniae in BALB/c mice. Virulence of the strains was determined by the number of CFU required to kill 50% (LD50) of the exposed mice using the Reed and Muench formula [35].
Statistical analyses of in vitro growth potential, biofilm formation, in vivo colonisation density, and antibody levels between groups were assessed by analysis of variance (ANOVA) and post-hoc test. Comparison of survival between groups of mice was analysed with the Log Rank chi-squared test on the Kaplan–Meier survival data. In all cases, statistically significant differences were defined as p < 0.05.
3. Results
3.1. Disruption of the cpsE Gene Led to Loss of the Capsule in S. Pneumoniae
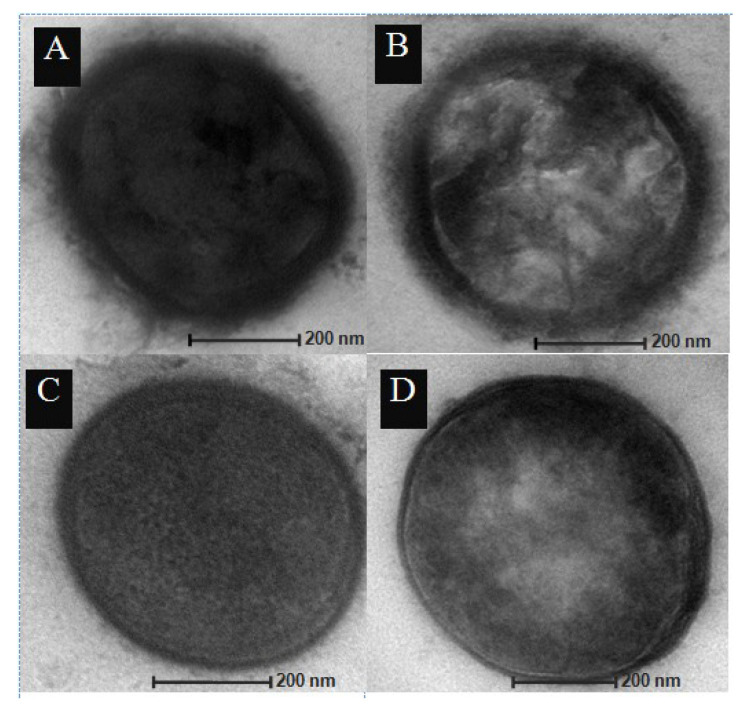
To confirm that disruption of the cpsE gene by the cpsE::tetL knockout module affected the capsule of the recombinant pneumococcal mutant strain designated SPC, transmission electron microscope (TEM) was used to investigate the morphology of the pneumococcal cells (Figure 1). The parental S. pneumoniae D39 wild-type strain as well as the D39 (endA::aphA3) knockout strain, SPE, showed a thick layer of capsule whereas the capsule was absent in the cpsE::tetL knockout strains, SPC and SPEC.
Figure 1.
Detection of capsule by transmission electron microscope (TEM). (A) wild-type parental D39 strain; (B) SPE strain; (C) SPC strain; (D) SPEC strain.
3.2. In Vitro Growth Characteristics
The growth kinetics of the gene-disrupted mutant strains did not differ in the duration of the lag phase (around 2–3 h after inoculation in broth) from the D39 wild-type but the SPC and SPEC knockout strains showed much lower peak bacterial density (OD600 = 1.01 and 0.94, respectively) than the wild-type and SPE strain (OD600 = 1.49 and 1.38, respectively) in the transition from exponential growth to the stationary growth phases (Figure 2). There was a significant (p < 0.05) effect of the cpsE capsular gene knockout on the OD600 values starting from 6 h of growth. A Tukey post hoc test revealed that mutant strains containing the capsule gene knockout, cpsE::tetL, had significantly lowered peak bacterial density [SPC (OD600 = 0.923 ± 0.054; p < 0.01) and SPEC (OD600 = 1.032 ± 0.133; p < 0.01)] compared to the wild-type D39 strain (OD600 = 1.497 ± 0.005). However, no statistically significant difference was observed between the SPE strain and the wild-type strain.
Figure 2.
In vitro growth curve of S. pneumoniae constructed mutant strains as compared to the wild-type D39 strain in brain heart infusion (BHI) media as determined by the OD600 values with time of inoculation. The statistical analysis of growth potential between the groups was performed using analysis of variance (ANOVA) followed by a post hoc Tukey’s test from three independent experiments. Abbreviations used: wild, S. pneumoniae D39 wild-type strain; SPE, S. pneumoniae D39 (endA::aphA3) knockout strain; SPC, S. pneumoniae D39 (cpsE::tetL) knockout strain; SPEC, S. pneumoniae D39 (cpsE::tetL; endA::aphA3) double-knockout strain.
3.3. In Vitro Biofilm Formation
All constructed gene-disrupted mutant strains and wild-type S. pneumoniae D39 were able to attach to the 96-well polystyrene surface; however, the cpsE gene knockout had significant effects on biofilm formation with post hoc comparisons using the Tukey test showing that the mean scores for the SPC and the SPEC strains were significantly different than the wild-type D39 strain at all the studied time intervals (Figure 3). However, for the SPE strain, biofilm formation did not significantly differ from the parental strain at any studied time intervals. The greatest biofilm formation capacity was observed at 18 h for the SPC (2.10 ± 0.11) and the SPEC mutant strains (2.09 ± 0.16), which were almost 20 times greater than the SPE (0.13 ± 0.01) and wild-type S. pneumoniae D39 (0.11 ± 0.01) strains.
Figure 3.
Biofilm formation capacities of S. pneumoniae D39 wild-type, SPE, SPC, and SPEC gene-disrupted mutantstrains in a 96-well microtiter plate as measured by the OD570 values following crystal violet staining. Biofilm formation between the groups was assessed by analysis of variance (ANOVA) and post-hoc test. Abbreviations used: wild, S. pneumoniae D39 wild-type strain; SPE, S. pneumoniae D39 (endA::aphA3) knockout strain; SPC, S. pneumoniae D39 (cpsE::tetL) knockout strain; SPEC, S. pneumoniae D39 (cpsE::tetL; endA::aphA3) double-knockout strain.
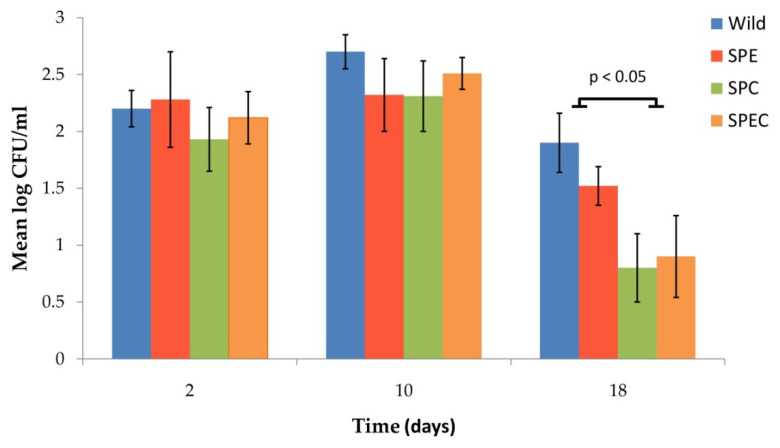
3.4. Density and Duration of Nasopharyngeal (NP) Colonisation in Mice
Since the longer colonisation duration of live attenuated vaccines in the NP mucosa induces better immune protection [36], the effect of the gene knockouts on NP colonisation of the pneumococcal mutants was determined. Following experimental colonisation by intranasal inoculation with 1 × 107 CFU of each of the gene-disrupted mutant strains, S. pneumoniae colonies were recovered from the NP washes of the inoculated BALB/c mice at days 2, 10 and 18 post-inoculation (Figure 4) but had cleared by day 26 for all strains, including the D39 wild-type. Nevertheless, there were differences in the colonisation density and duration of mice for the different strains and at different time points. At 2 days post-inoculation, the highest colonisation density was observed in the D39 wild-type strain followed by the SPE, SPC and SPEC mutant strains. The highest colonisation density continued to be observed for the wild-type strain at days 10 and 18 post-inoculation. However, at day 18 of the colonisation study, half of the mice had cleared the bacteria in groups that were infected with the capsule mutant strains (i.e., SPC and SPEC) and the colonisation density was significantly lower when compared to the wild-type and SPE strains. The result suggests that the capsule gene knockout (i.e., cpsE::tetL) led to a decrease in the duration of NP colonisation in mice. At day 26, no bacteria were recovered from the NP wash fluids in all groups of mice.
Figure 4.
Number of S. pneumoniae cells (presented as log10CFU/mL) recovered from the nasopharyngeal wash fluids of BALB/c mice at various time intervals following intranasal inoculation with 1 × 107 CFU of the respective bacterial strains. Error bars represent standard deviation (SD). The statistical analysis of nasopharyngeal (NP) colonisation between the groups was performed using analysis of variance (ANOVA) followed by a post-hoc Tukey’s test. Abbreviations used: wild, S. pneumoniae D39 wild-type strain; SPE, S. pneumoniae D39 (endA::aphA3) knockout strain; SPC, S. pneumoniae D39 (cpsE::tetL) knockout strain; SPEC, S. pneumoniae D39 (cpsE::tetL; endA::aphA3) double-knockout strain.
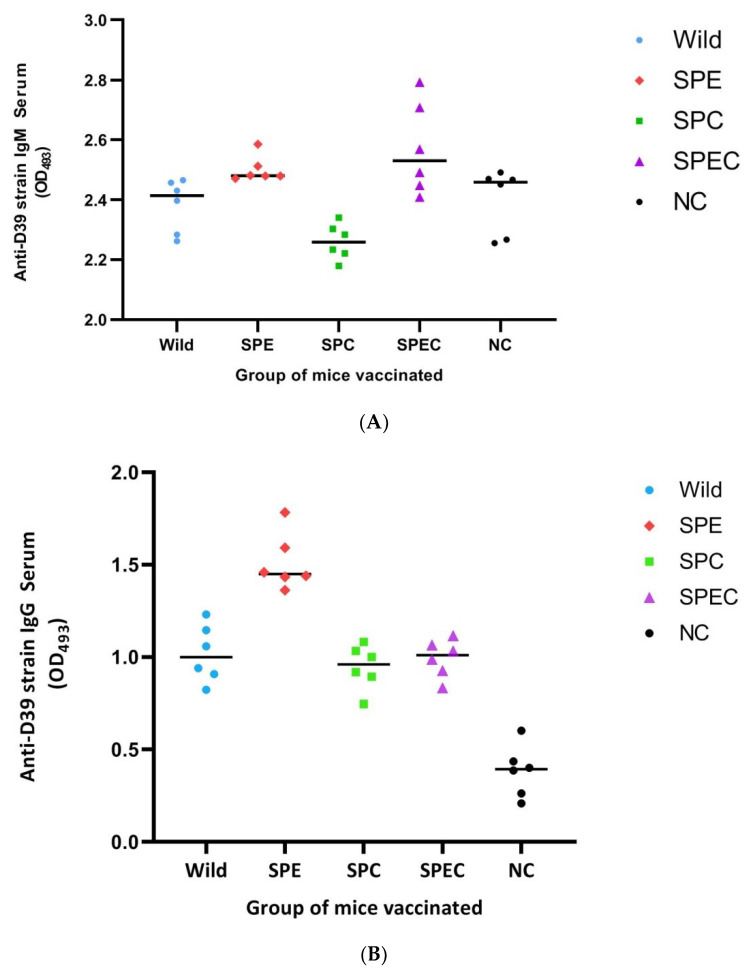
3.5. Effect of Immunisation with the Gene-Disrupted Mutant Strains on Local and Systemic Antibody Titres
To explore the antibody response of BALB/c mice to the constructed gene-disrupted pneumococcal mutant strains, levels of serum IgM, IgG, and IgA from NP wash samples were determined three weeks following two rounds of inoculation of 1 × 107 CFU of the bacteria two weeks apart. Compared to the PBS-inoculated negative control group, levels of pneumococcal-specific IgG and IgA antibodies were high (p < 0.01) in all groups of inoculated mice; however, levels of IgM were significantly higher (p < 0.05) only in the SPEC-immunised group. Similar to serum IgG and NP mucosal IgA antibody levels, unencapsulation of the bacteria decreased serum IgM antibody response in the SPC strain. Compared to mock-immunised mice (2.40 ± 0.10) significantly higher IgM response was only observed with SPEC-immunised mice (2.57 ± 0.51; p < 0.05). Furthermore, the mice immunised with SPEC (2.57 ± 0.51; p < 0.01) and SPE (2.50 ± 0.43; p < 0.01) strains showed significantly higher IgM response compared to mice immunised with the SPC strain (2.26 ± 0.06) (Figure 5A). The mice immunised with the SPE knockout strain showed the highest level of IgG (1.51 ± 0.15; p < 0.01) compared to SPEC (0.99 ± 0.10), SPC (0.95 ± 0.12), and wild-type (1.02 ± 0.15) pneumococci-immunised mice (Figure 5B). However, the level of IgA antibodies was highest in mice that were immunised with wild-type pneumococci (1.17 ± 0.12; p < 0.01) compared to mice that were immunised with SPEC (0.76 ± 0.09), SPC (0.75 ± 0.10), and SPE (0.96 ± 0.08) mutant strains (Figure 5C).
Figure 5.
Comparison of the anti-D39 strain serum IgM (A), IgG (B), and NP lavage IgA (C) antibody levels of mice after vaccination with the indicated strains. The serum utilised was diluted to 1:100 and NP fluid was diluted to 1:10. Bars represent the respective mean values. The statistical analysis of antibody levels between the groups was performed using analysis of variance (ANOVA) followed by a post hoc Tukey’s test. In (A), significant differences were found between groups SPEC and NC (p < 0.05), between groups SPEC and SPC (p < 0.01), and between groups SPE and SPC (p < 0.05). In (B), significant differences (p < 0.01) were observed in all groups except between Wild-, SPC- and SPEC-immunised mice. In (C), significant difference (p < 0.01) were found in all groups except between SPC- and SPEC-immunised mice. Abbreviations used: Wild, S. pneumoniae D39 wild-type strain; SPE, S. pneumoniae D39 (endA::aphA3) knockout strain; SPC, S. pneumoniae D39 (cpsE::tetL) knockout strain; SPEC, S. pneumoniae D39 (cpsE::tetL; endA::aphA3) double-knockout strain; and NC, negative control, i.e., mice vaccinated with phosphate-buffered saline (PBS).
3.6. Survival Analysis
The ability of the constructed gene-disrupted mutant strains to protect against a lethal dose (≈5 × 108 CFU) of intranasal wild-type S. pneumoniae D39 infection was assessed after immunisation of mice with each of the constructed mutant strains. Kaplan-Meier survival analysis showed that mice immunised with the SPEC strain demonstrated the highest survival rate (56%) and significantly longer survival time 176.0 h (95% CI 136.2–228.6) compared to the SPC strain immunised 99.0 h (p < 0.05, 95% CI 52.6–145.4) and mock-immunised mice 96.0 h (p < 0.05, 95% CI 57.5–134.5) (Figure 6). Mice immunised with the wild-type strain showed 50% survival rate and 172.2 h (95% CI 123.7–220.7) survival time was also significantly different compared to survival time of mice immunised with SPC and mock-immunised mice. Moderate survival rate (40%) and time 148.8 h (95% CI 98.7–198.9) were observed in mice immunised with the SPE strain; however, the survival time was not significantly different compared to the other groups. In contrast, mice immunised with the SPC strain showed the lowest survival rate and time, 20% and 99.0 h (±23.7), respectively.
Figure 6.
Survival rate (%) of vaccinated mice after exposure to a lethal dose (≈5 × 108 CFU) of intranasal wild-type S. pneumoniae D39 infection. Comparison of survival between groups of mice was analysed with the Log Rank chi-squared test on the Kaplan–Meier survival data. * pairwise comparisons show SPEC and wild strain immunised mice had significant differences (p < 0.05) between SPC and mock immunised mice. Abbreviations used: NC, mice immunised with PBS; Wild, mice immunised with wild-type S. pneumoniae D39; SPE, mice immunised with S. pneumoniae D39 (endA::aphA3) knockout mutant; SPC, mice immunised with S. pneumoniae D39 (cpsE::tetL) knockout mutant; SPEC, mice immunised with S. pneumoniae D39 (cpsE::tetL; endA::aphA3) double-knockout mutant.
3.7. Attenuation of Virulence
Virulence of the constructed gene-knockout strains was measured as the lethal dose for BALB/c mice. The median lethal dose (LD50) in CFUs was measured by intranasal infection of four groups of ten mice each with 5-fold serial dilutions. Since the highest protection was observed in mice immunised with the SPEC mutant strain (see “Survival analysis” subsection above), we studied the level of attenuation of this strain as compared to the wild-type S. pneumoniae D39.
Although both parental and SPEC strains were grown to OD600 = 0.8, the SPEC strain showed almost 10 times higher CFU/mL, i.e., 1.46 × 1010 as compared to 1.42 × 1011 for the wild-type D39 strain, at which concentrations both strains showed 100% lethality in mice. The LD50 for the intranasal parental wild-type D39 strain was 1.5 × 109 CFU/mL. However, the SPEC double-mutant strain showed 23 times higher LD50 (at 3.5 × 1010 CFU/mL) compared to the parental wild-type strain. Thus, the SPEC double-knockout mutant strain displayed a 23-fold level of attenuation of virulence when compared to the wild-type D39 strain (Table 1).
Table 1.
Attenuated virulence of live SPEC strain in vivo.
| Strains | LD50 (CFU) | Fold of Attenuation * |
|---|---|---|
| SPEC | 3.5 × 1010 | 23 |
| Wild-type D39 | 1.5 × 109 | 1 |
* Fold attenuation is calculated by dividing LD50 of the SPEC mutant strain to LD50 of the wild-type [37].
4. Discussion
Unencapsulation is one of the strategies to unmask subcapsular protein antigens [38] in order to induce immune response against multi-epitope subcapsular proteins [39]. An anti-protein immune response is crucial for anti-pneumococcal immunity as non-encapsulated strains are emerging following the widespread use of capsule-based vaccines (i.e., PCV and PPV) worldwide [40].
The polysaccharide capsule in S. pneumoniae is synthesised from a single capsule operon (designated cps) which consists of 17 genes in the D39 strain [41]. Deletions or single base-pair mutations in any of the capsule operon genes affect expression of the polysaccharide capsule and pneumococcal virulence. For example, deletion of cpsA causes partial encapsulation of S. pneumoniae, and deletion of cpsB and cpsD or point mutations in cpsE gene led to the deletion of the entire pneumococcal capsule [25,42].
The cpsE gene is responsible for the addition of activated sugars to the lipid carrier in the bacterial membrane [43,44] and, in this study, a cpsE knockout led to the unencapsulation of the resulting recombinant S. pneumoniae mutant strain. The cpsE gene has been reported to be essential for S. pneumoniae encapsulation in serotypes 13, 14, 15B, and 19F [45,46]. A single mutation in the cpsE gene was found to cause loss of capsule expression [25]. Previously, unencapsulated pneumococcal live vaccines were constructed by total capsule operon deletion [24]. We avoided the entire operon deletion because the capsule synthesis genes are also involved in other pneumococcal metabolic processes besides polysaccharide capsule production [47]. Even some unencapsulated pneumococcal human isolates were found to preserve the capsule operon [48]. However, we cannot exclude the possibility that insertion of the antibiotic resistance marker into the cpsE gene could have a polar effect in the expression of downstream genes.
4.1. Capsule Mutations Affected the Growth of S. Pneumoniae
To induce vaccine-mediated protective response, live vaccines should persist within the host in an appropriate location in sufficient quantities and for a substantial duration of time and should contain both antigen and immune-activating endogenous danger signals as adjuvants [49,50].
The in vitro growth behaviour of constructed knockout strains could predict certain characteristics of S. pneumoniae, especially the invasiveness and colonisation potential of the pneumococcal strains. Any alteration in the capsular operon genes (even single point mutations) could cause changes from decreased levels of capsule production to the complete loss of the capsule [51,52] and may alter growth, adherence, invasiveness, colonisation, and competence of the microorganism [25,53].
This study showed that the cpsE gene knockout led to a complete loss of the polysaccharide capsule in the SPC and SPEC mutant strains (Figure 1). The capsule knockout strains did not, however, show any differences in the initial lag phase of the growth compared to the D39 wild-type parental strain. In contrast, an earlier study had showed that deletion of several downstream capsule genes such as cpsK, cpsP, cpsK–O, and cpsM–O led to a significantly prolonged lag phase when compared to wild-type S. pneumoniae. However, deletion of other serotype-specific capsule genes such as cpsF–O and cpsL–O or several other genes which are downstream of cpsK did not significantly changed the duration of the in vitro lag phase [54]. A possible reason for the slowing of the lag phase in these mutants is that the proteins and enzymes encoded by the capsule genes are also involved in other bacterial metabolic pathways besides polysaccharide capsule production. Therefore, during its growth the mutant bacteria may need time to switch to alternative pathways leading to the observed longer lag phase [54]. Schaffner et al. (2014), who worked on clinical S. pneumonia serotype 18C isolate, also demonstrated an increase in the lag phase in naturally-occurring unencapsulated S. pneumoniae strains along with a laboratory-constructed unencapsulated strain by point mutations in the cpsE gene [25].
We found that the cpsE mutant strains had significantly lower maximal OD600nm value (i.e., at stationary phase) compared to that of wild-type and the endA-disrupted mutant strain, SPE. After the exponential (log) phase, the bacterial growth slowed down most likely from a shortage of nutrients, thus entering the stationary phase. However, encapsulated bacteria could utilize their capsular carbohydrates for their nutritional needs. This agrees with the findings of Hamaguchi et al. (2018) who demonstrated clear survival advantage of capsulated strains of S. pneumoniae in aqueous media devoid of nutrients compared to unencapsulated strains [55]. Nonetheless, this finding appeared to be serotype-specific, as Schaffner et al. (2014) who studied point mutation in naturally occurring cpsE (C to G change at gene position 1135) which rendered pneumococci non-encapsulated showed opposite data where mutant S. pneumonia had the maximal OD600nm values compared with encapsulated strain of serotype 18C [25].
4.2. Biofilm Profiles of the Candidate Vaccine Strains
Growth in biofilm enables S. pneumoniae to not only resist the bactericidal effects of antimicrobial agents and to help in evading the host immune system, but to also facilitate microbial persistence and their dissemination [56]. Studies of pneumococcal gene expression in in vitro biofilm models showed altered production of several virulence factors including capsular polysaccharides [57,58,59].
Although previous reports have shown altered biofilm formation when the capsule operon, or specific genes of the operon were deleted, the biofilm formation of a specific cpsE gene-knockout strain has not been investigated. We found that the cpsE mutation significantly increased in vitro biofilm formation by several folds in S. pneumoniae. This is likely due to exposure of sub-capsular adherence factors to the surface of polystyrene plates as a result of impaired polysaccharide capsule in these mutants. This finding broadly supports the work of others who have reported that impaired pneumococcal capsule led to increased biofilm formation. Qin et al. (2013) demonstrated unencapsulation by knocking out the cpsD gene which resulted in an increase in biofilm formation but decreased virulence in a S. pneumoniae TIGR4 strain. In vivo studies also showed that pneumococcal strains that lack polysaccharide capsules form much stronger biofilms than encapsulated ones [60].
We hypothesised that the endA gene might also be involved in pneumococcal biofilm formation since a recent study showed that endA gene knockouts led to the impairment of biofilm formation in Pseudomonas aeruginosa [61]. However, our data showed that in S. pneumoniae, biofilm formation is not affected by endA interruption since there was no difference in biofilm density between the wild-type parental strain and the endA knockout strain. The pneumococcal-encoded endA may thus serve a different function in relation to biofilms as compared to the P. aeruginosa-encoded endA.
4.3. Colonisation Potential of the Pneumococcal Gene-Knockout Strains
Pneumococcal polysaccharide capsule is an important virulence factor required for both colonisation and disease severity [62,63]. In a mouse model, we demonstrated that unencapsulated mutant strains of S. pneumoniae D39 were capable of NP colonisation; however, compared to the wild-type parental strain, unencapsulated strains had a reduced duration and density of colonisation. It can be hypothesised that partially unencapsulated S. pneumoniae strains or strains that were completely devoid of the polysaccharide capsule are more susceptible to phagocytosis by polymorphonuclear leukocytes (PMNs) [64]. Nevertheless, the variability of the colonisation potential among unencapsulated strains of different capsular serotypes inferred that the colonisation potential appeared to be serotype-specific. Previously constructed unencapsulated mutants of serotypes 6A and 4 by capsular operon deletion reported significantly decreased colonisation density and duration when compared to their respective wild-type strains; however, serotype 4 had higher colonisation density than serotype 6A. Furthermore, the study did not find any association of colonisation density with protective immune responses [24]. Findings of our study also revealed that the constructed endA and cpsE mutant strains and the wild-type parental D39 strain sustained longer colonisation duration compared to that of other previous reports [24,65]. Pneumococcal colonisation of the upper NP of mice remained for more than 18 days for all strains but in half of the group of mice that were administered with the unencapsulated SPEC and SPC mutant strains, the bacteria were non-detectable and presumably cleared by the host after 18 days. The unencapsulated strains also had lower cell density compared to the capsulated strains. An earlier study conducted with the S. pneumoniae TIGR4 strain showed colonisation duration of only 14 days for unencapsulated mutants whereas encapsulated strains continued to be recovered from NP washes after that period [65]. Variability in host colonisation duration in different vaccine studies could be explained by the use of different pneumococcal serotypes and host-related factors such as the strain of mice used in these studies [66].
The results of this study also indicated that the colonisation potential of S. pneumoniae was not affected by deletion of the endA gene. We initially postulated that the endA knockout might also affect colonisation duration because of its reported role in mediating resistance against neutrophil-mediated killing [67,68]. However, our data suggested that endA mutation in S. pneumoniae strain D39 did not lead to any loss in its capability to colonise the upper respiratory tract of mice. Perhaps the role of endA in resistance towards neutrophil-mediated killing needs to be reassessed in the D39 strain and could be another serotype-dependent characteristic of S. pneumoniae.
4.4. Antibody Response to Candidate Vaccine Strains
In the serum, the majority of natural antibodies are composed of IgM which are reactive with many conserved pneumococcal epitopes. These antibodies limit the progression of infections during the early stages of inflammation and contribute, through somatic hypermutation and affinity maturation process, to produce high affinity specific IgG and IgA antibodies [69]. In the secondary immune response lower affinity adaptive IgM antibodies are also produced by IgM+ memory B cells which have enhanced complement activation property that protect the organism from pathogenic infection [70]. Our study also demonstrated that high level of pneumococcus specific IgM antibodies correlate with better protection in SPEC immunised mice. Other groups of mice immunised with single knockout strains, including wild type, did not show significantly higher IgM compared to mock immunised mice which is in agreement with Jason et al. (2014) who compared the IgM levels of mice immunised with a ftsY-knockout pneumococcal strain [28]. Furthermore, mice immunised with cpsD-, lgt-, and pabB-knockout D39 pneumococcal strains demonstrated better survival than mock immunised mice although those strains did not show significantly different IgM levels [36].
The levels of pneumococcus-specific IgA in NP lavage and IgG in serum were significantly increased after immunisation with the gene-disrupted strains SPEC, SPE, SPC as well as the wild-type parental D39 strain as compared to mock-immunised mice. We initially postulated that unencapsulation will increase the antibody response against subcapsular proteins since the polysaccharide capsule hides or masks these proteins from the host antibody recognition. However, there was no difference in serum IgG levels between the unencapsulated mutant strains (i.e., SPC and SPEC) and the encapsulated parental wild-type S. pneumoniae D39 strain. Roche et al., (2007) also demonstrated high serum IgG levels after immunisation with an unencapsulated strain and capsule-intact ply/pspA-knockout 6A strains [24]. Similar to our findings, there was no significant difference in the IgG levels between these groups of mice. Furthermore, experiments with µMT mice (a mice strain that lack mature B cells to produce specific antibodies) and MHCII−/− mice that lack CD4+ T cells demonstrated absence of protection following immunisation indicating the importance of antibodies and CD4+ T cells in mucosal and systemic protection [24].
In our study, the serum IgG and NP lavage IgA levels of mice immunised with the SPC, SPE, and SPEC strains and the wild-type parental D39 strain were greater than mock immunised mice. However, the serum IgG levels of mice immunised with the SPE strain were significantly higher than that of mice immunised with the SPC, SPEC, and wild-type D39 strains. The reason for the higher serum IgG response to the SPE strain is possibly due to the increased sensitivity of the strain to neutrophil-mediated phagocytosis and neutrophil activation of the adaptive immune system by engaging with lymphocytes and antigen-presenting cells [71]. Although immune protection against pneumococcal infections after NP colonisation was found to be mostly dependent on antibodies that recognise several S. pneumonia cell wall proteins in mice, in humans the dominant role of anti-protein antibodies over anti-capsule antibodies still remains unclear. In recent years, however, the growing number of evidence showed the role of serum anti-protein antibodies (IgG) to protect from systemic invasive infection and Th17 CD4+ cells for mucosal infections [72,73,74,75]. Furthermore, prospective cohort studies in humans also prove the role of antibodies and T cells in the protection from pneumococcal infections [76,77].
To achieve protection against S. pneumoniae infection, it is necessary to prevent pneumococcal NP carriage since invasion of the microorganism is usually preceded by at least a short duration of carriage. Immunity against pneumococcal carriage is provided by passive mucosal IgA antibody and L-17-dependent T cell immunity [78]. In this study, all the constructed gene-knockout strains (i.e., SPC, SPE, SPEC) and wild-type parental D39 strain induced significantly high IgA antibody levels in mice NP lavage. The IgA levels of mice induced by the wild-type strain were higher than the IgA levels induced by the gene-knockout strains. Similar findings were obtained by Ibrahim et al. with the pneumococcal HtrA mutant strain which is sensitive to high temperatures and oxidative stress [79,80]. However, Cohen et al. (2012), showed that the IgA response to their candidate vaccine strains (a D39Δlgt and a D39Δpab strains) was similar to the wild-type parental strain.
4.5. Immune Protection Conferred by Gene-Knockout Strains
In this study, all the gene-knockout strains conferred better protection against very high doses of wild-type mucosal challenge compared to mock (PBS)-immunised mice. The highest protection was observed in mice immunised with the double mutant strain (SPEC) followed by the wild-type parental strain. Even though mice immunised with the SPE strain demonstrated the highest serum IgG level, protection from mucosal challenge was lower than mice immunised with SPEC and wild-type parental strain. This discrepancy between pneumococcus-specific serum IgG level and the protection against lethal challenges can perhaps be explained by the in vitro nature of antibody measurement used in this study. Although serum IgG from immunised mice bind to pneumococci whole-cells during ELISA, they might not be functional during in vivo protection. Cohen et al. (2012) obtained similar findings with high serum IgG levels but lower protection from heterologous challenge. They postulated that the discrepancy might be due to bacterial lysis that occurred during overnight incubation which enabled intracellular antigens to become accessible to serum antibodies in whole cell ELISA [81].
The lower level of protection observed in this study may also be due to the use of the highly encapsulated pneumococcal strain, D39, in the lethal challenge as it has been shown that capsule thickness affects opsonophagocytosis, which reduces as the thickness of the capsule increases [82]. However, in our research the wild-type strain conferred excellent protection in mice. In our experiments, we used a higher challenge dose (5 × 108 CFU) of the wild-type strain compared to that used in the previous studies (1 × 107 to 5 × 107 CFU) [24,28,36]; this could be one of the reasons why we did not achieve 100% protection. On the other hand, the genetic background of the animal models used should also be considered in studying pneumococcal immunity. Although BALB/c mice, which was used in this work, is preferred for immunity studies, it is fairly resistant to S. pneumonia lethality compared to CD1 and C57BL/6 mice [83,84].
Since intranasal challenge studies showed that SPEC and the wild-type parental strain conferred the highest protection in mice, we further investigated the attenuation of virulence of these strains by measuring the LD50 dose, i.e., the dose which causes the death of 50% of mice. It was found that the double-knockout SPEC strain showed 23-fold of attenuation in the BALB/c mice when compared to the wild-type parental D39 strain. It is likely that the level of attenuation was due to unencapsulation as a result of the cpsE gene knockout as the polysaccharide capsule is considered one of the most important virulence factors of S. pneumoniae [65].
Here, we also did not directly investigate the effect of the endA gene knockout on virulence, but we postulated that the endA knockout would also have a considerable contribution in the attenuation of pneumococcal virulence observed in the SPEC mutant. We were unable to measure the level of attenuation for the single knockout strains as this would have been prohibitive in terms of cost as well as infrastructure due to the large number of mice involved in assessing the LD50 dose. Previous studies have shown that pneumococcal infections caused abundant neutrophil infiltration which, upon activation, releases neutrophil extracellular traps (NETs), which bind and kill pneumococci. However, pneumococci escape these neutrophil NETs by degrading the DNA component of NETs using a cell-surface endonuclease encoded by endA [85,86]. Therefore, it was postulated that exposure of the host to the endA knockout strain and the resultant increased neutrophil killing would also lead to attenuation of the SPEC strain. It was previously reported that vaccination with a pep27 mutant strain of S. pneumoniae had led to increased neutrophil killing of the bacteria, and this, in turn, caused the more than 50-fold attenuation in intranasal administration [37]. Unfortunately, most pneumococcal live vaccine studies did not evaluate the fold of attenuation of the constructed mutant strains [28,29,79].
5. Conclusions
Our findings clearly demonstrated that the cpsE capsule gene knockout decreased peak bacterial growth, colonisation density and duration and furthermore increased biofilm formation in the constructed S. pneumoniae SPE and SPEC gene knockout strains. However, no differences were observed in these experiments between the wild-type and SPE strains which indicated that the endA gene likely had no observable role in biofilm formation and growth of S. pneumoniae. Our findings provided additional evidence that unencapsulation decreased serum IgG antibody levels. Although mice immunised with the SPE strain showed highest anti-pneumoccal IgG levels, we did not find better survival advantage in mice immunised with the SPE strain compared to mice immunised with other candidate vaccine strains. Mice immunised with the SPEC strain showed the highest survival rate and longest survival time after a lethal dose challenge when compared to the single gene knockout strains. Furthermore, the virulence of the SPEC double-mutant was attenuated by 23-fold compared to the parental wild-type strain. Nevertheless, the process of attenuation might also decrease the immunogenicity of candidate LAVs, because attenuation is mostly done by knocking out the pathogenic mechanisms of the microorganism. Those pathogenic mechanisms are usually responsible for inducing robust immune responses as well. In this instance, no drastic decrease in the immune response was observed in mice that were immunised with the SPEC double-mutant strain. Thus, the SPEC double-mutant strain could be considered a promising candidate for a LAV strain. Further studies to compare its immune response with heat-inactivated S. pneumoniae and currently available vaccines will be conducted in the future.
Author Contributions
M.A. conceptualised, performed the experiments, analysed the data and wrote the original draft of the manuscript; N.S., C.C.Y. and S.C.C. supervised the project, reviewed and edited the manuscript; W.M.R.W.H., N.S., S.I., N.I.A.R. and C.C.Y. obtained funding for the project. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by a UniSZA/LABMAT/2018/03 grant from Universiti Sultan Zainal Abdin, Kuala Terengganu, Malaysia.
Conflicts of Interest
The authors declare no conflict of interest. The funding sponsors had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, and in the decision to publish the results.
References
- 1.Troeger C., Blacker B., Khalil I., Rao P., Cao J., Zimsen S., Albertson S., Deshpande A., Farag T., Abebe Z. Estimates of the global, regional, and national morbidity, mortality, and aetiologies of lower respiratory infections in 195 countries, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Infect. Dis. 2018;18:1191–1210. doi: 10.1016/S1473-3099(18)30310-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Wahl B., O’Brien K.L., Greenbaum A., Majumder A., Liu L., Chu Y., Luksic I., Nair H., McAllister D.A., Campbell H., et al. Burden of Streptococcus pneumoniae and Haemophilus influenzae type b disease in children in the era of conjugate vaccines: Global, regional, and national estimates for 2000–15. Lancet Glob. Health. 2018;6:e744–e757. doi: 10.1016/S2214-109X(18)30247-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.O’Brien K.L., Wolfson L.J., Watt J.P., Henkle E., Deloria-Knoll M., McCall N., Lee E., Mulholland K., Levine O.S., Cherian T., et al. Burden of disease caused by Streptococcus pneumoniae in children younger than 5 years: Global estimates. Lancet. 2009;374:893–902. doi: 10.1016/S0140-6736(09)61204-6. [DOI] [PubMed] [Google Scholar]
- 4.Bryce J., Boschi-Pinto C., Shibuya K., Black R.E., WHO Child Health Epidemiology Reference Group WHO estimates of the causes of death in children. Lancet. 2005;365:1147–1152. doi: 10.1016/S0140-6736(05)71877-8. [DOI] [PubMed] [Google Scholar]
- 5.Mook-Kanamori B.B., Geldhoff M., van der Poll T., van de Beek D. Pathogenesis and pathophysiology of pneumococcal meningitis. Clin. Microbiol. Rev. 2011;24:557–591. doi: 10.1128/CMR.00008-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Malley R. Antibody and cell-mediated immunity to Streptococcus pneumoniae: Implications for vaccine development. J. Mol. Med (Berl.) 2010;88:135–142. doi: 10.1007/s00109-009-0579-4. [DOI] [PubMed] [Google Scholar]
- 7.Geno K.A., Gilbert G.L., Song J.Y., Skovsted I.C., Klugman K.P., Jones C., Konradsen H.B., Nahm M.H. Pneumococcal Capsules and Their Types: Past, Present, and Future. Clin. Microbiol. Rev. 2015;28:871–899. doi: 10.1128/CMR.00024-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sugimoto N., Yamagishi Y., Hirai J., Sakanashi D., Suematsu H., Nishiyama N., Koizumi Y., Mikamo H. Invasive pneumococcal disease caused by mucoid serotype 3 Streptococcus pneumoniae: A case report and literature review. BMC Res. Notes. 2017;10:21. doi: 10.1186/s13104-016-2353-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Jauneikaite E., Jefferies J.M., Hibberd M.L., Clarke S.C. Prevalence of Streptococcus pneumoniae serotypes causing invasive and non-invasive disease in South East Asia: A review. Vaccine. 2012;30:3503–3514. doi: 10.1016/j.vaccine.2012.03.066. [DOI] [PubMed] [Google Scholar]
- 10.VIEW-hub Global Vaccine Introduction Report, December 2018. [(accessed on 14 June 2019)]; Available online: www.jhsph.edu/ivac/view-hub.
- 11.Tricarico S., McNeil H.C., Cleary D.W., Head M.G., Lim V., Yap I.K.S., Wie C.C., Tan C.S., Norazmi M.N., Aziah I., et al. Pneumococcal conjugate vaccine implementation in middle-income countries. Pneumonia (Nathan) 2017;9:6. doi: 10.1186/s41479-017-0030-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hicks L.A., Harrison L.H., Flannery B., Hadler J.L., Schaffner W., Craig A.S., Jackson D., Thomas A., Beall B., Lynfield R., et al. Incidence of pneumococcal disease due to non-pneumococcal conjugate vaccine (PCV7) serotypes in the United States during the era of widespread PCV7 vaccination, 1998-2004. J. Infect. Dis. 2007;196:1346–1354. doi: 10.1086/521626. [DOI] [PubMed] [Google Scholar]
- 13.Miller E., Andrews N.J., Waight P.A., Slack M.P., George R.C. Herd immunity and serotype replacement 4 years after seven-valent pneumococcal conjugate vaccination in England and Wales: An observational cohort study. Lancet Infect. Dis. 2011;11:760–768. doi: 10.1016/S1473-3099(11)70090-1. [DOI] [PubMed] [Google Scholar]
- 14.Henriques-Normark B., Tuomanen E.I. The pneumococcus: Epidemiology, microbiology, and pathogenesis. Cold Spring Harb. Perspect. Med. 2013;3 doi: 10.1101/cshperspect.a010215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gladstone R.A., Jefferies J.M., Tocheva A.S., Beard K.R., Garley D., Chong W.W., Bentley S.D., Faust S.N., Clarke S.C. Five winters of pneumococcal serotype replacement in UK carriage following PCV introduction. Vaccine. 2015;33:2015–2021. doi: 10.1016/j.vaccine.2015.03.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Janapatla R.P., Su L.H., Chen H.H., Chang H.J., Tsai T.C., Chen P.Y., Chen C.L., Chiu C.H. Epidemiology of culture-confirmed infections of Streptococcus pneumoniae (2012–2015) and nasopharyngeal carriage in children and households in Taiwan (2014–2015) J. Med. Microbiol. 2017;66:729–736. doi: 10.1099/jmm.0.000488. [DOI] [PubMed] [Google Scholar]
- 17.Vestrheim D.F., Steinbakk M., Aaberge I.S., Caugant D.A. Postvaccination increase in serotype 19A pneumococcal disease in Norway is driven by expansion of penicillin-susceptible strains of the ST199 complex. Clin. Vaccine. Immunol. 2012;19:443–445. doi: 10.1128/CVI.05563-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Plotkin S.A., Orenstein W.A., Offit P.A., Edwards K.M. Plotkin’s Vaccines. Elsevier; Philadelphia, PA, USA: 2018. pp. 18–26. [Google Scholar]
- 19.Lockhart S. Conjugate vaccines. Expert. Rev. Vaccine. 2003;2:633–648. doi: 10.1586/14760584.2.5.633. [DOI] [PubMed] [Google Scholar]
- 20.Guevara M., Barricarte A., Torroba L., Herranz M., Gil-Setas A., Gil F., Bernaola E., Ezpeleta C., Castilla J., Working Group for Surveillance of the Pneumococcal Invasive Disease in Navarra Direct, indirect and total effects of 13-valent pneumococcal conjugate vaccination on invasive pneumococcal disease in children in Navarra, Spain, 2001 to 2014: Cohort and case-control study. Euro. Surveill. 2016;21 doi: 10.2807/1560-7917.ES.2016.21.14.30186. [DOI] [PubMed] [Google Scholar]
- 21.Esposito S., Principi N., Group E.V.S. Direct and indirect effects of the 13-valent pneumococcal conjugate vaccine administered to infants and young children. Future Microbiol. 2015;10:1599–1607. doi: 10.2217/fmb.15.81. [DOI] [PubMed] [Google Scholar]
- 22.Vetter V., Denizer G., Friedland L.R., Krishnan J., Shapiro M. Understanding modern-day vaccines: What you need to know. An. Med. 2018;50:110–120. doi: 10.1080/07853890.2017.1407035. [DOI] [PubMed] [Google Scholar]
- 23.Moffitt K.L., Yadav P., Weinberger D.M., Anderson P.W., Malley R. Broad antibody and T cell reactivity induced by a pneumococcal whole-cell vaccine. Vaccine. 2012;30:4316–4322. doi: 10.1016/j.vaccine.2012.01.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Roche A.M., King S.J., Weiser J.N. Live attenuated Streptococcus pneumoniae strains induce serotype-independent mucosal and systemic protection in mice. Infect. Immun. 2007;75:2469–2475. doi: 10.1128/IAI.01972-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Schaffner T.O., Hinds J., Gould K.A., Wuthrich D., Bruggmann R., Kuffer M., Muhlemann K., Hilty M., Hathaway L.J. A point mutation in cpsE renders Streptococcus pneumoniae nonencapsulated and enhances its growth, adherence and competence. BMC Microbiol. 2014;14:210. doi: 10.1186/s12866-014-0210-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Beiter K., Wartha F., Albiger B., Normark S., Zychlinsky A., Henriques-Normark B. An endonuclease allows Streptococcus pneumoniae to escape from neutrophil extracellular traps. Curr. Biol. 2006;16:401–407. doi: 10.1016/j.cub.2006.01.056. [DOI] [PubMed] [Google Scholar]
- 27.Peterson E.J., Kireev D., Moon A.F., Midon M., Janzen W.P., Pingoud A., Pedersen L.C., Singleton S.F. Inhibitors of Streptococcus pneumoniae surface endonuclease EndA discovered by high-throughput screening using a PicoGreen fluorescence assay. J. Biomol. Screen. 2013;18:247–257. doi: 10.1177/1087057112461153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Rosch J.W., Iverson A.R., Humann J., Mann B., Gao G., Vogel P., Mina M., Murrah K.A., Perez A.C., Edward Swords W., et al. A live-attenuated pneumococcal vaccine elicits CD4+ T-cell dependent class switching and provides serotype independent protection against acute otitis media. EMBO Mol. Med. 2014;6:141–154. doi: 10.1002/emmm.201202150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Xu X., Wang H., Liu Y., Wang Y., Zeng L., Wu K., Wang J., Ma F., Xu W., Yin Y., et al. Mucosal immunization with the live attenuated vaccine SPY1 induces humoral and Th2-Th17-regulatory T cell cellular immunity and protects against pneumococcal infection. Infect. Immun. 2015;83:90–100. doi: 10.1128/IAI.02334-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Zhang X., Cui J., Wu Y., Wang H., Wang J., Qiu Y., Mo Y., He Y., Zhang X., Yin Y., et al. Streptococcus pneumoniae Attenuated Strain SPY1 with an Artificial Mineral Shell Induces Humoral and Th17 Cellular Immunity and Protects Mice against Pneumococcal Infection. Front. Immunol. 2017;8:1983. doi: 10.3389/fimmu.2017.01983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Nieto C., Espinosa M. Construction of the mobilizable plasmid pMV158GFP, a derivative of pMV158 that carries the gene encoding the green fluorescent protein. Plasmid. 2003;49:281–285. doi: 10.1016/S0147-619X(03)00020-9. [DOI] [PubMed] [Google Scholar]
- 32.Thanassi J.A., Hartman-Neumann S.L., Dougherty T.J., Dougherty B.A., Pucci M.J. Identification of 113 conserved essential genes using a high-throughput gene disruption system in Streptococcus pneumoniae. Nucleic. Acids. Res. 2002;30:3152–3162. doi: 10.1093/nar/gkf418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hammerschmidt S., Wolff S., Hocke A., Rosseau S., Muller E., Rohde M. Illustration of pneumococcal polysaccharide capsule during adherence and invasion of epithelial cells. Infect. Immun. 2005;73:4653–4667. doi: 10.1128/IAI.73.8.4653-4667.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Yadav M.K., Chae S.W., Song J.J. In VitroStreptococcus pneumoniae Biofilm Formation and In Vivo Middle Ear Mucosal Biofilm in a Rat Model of Acute Otitis Induced by S. pneumoniae. Clin. Exp. Otorhinolaryngol. 2012;5:139–144. doi: 10.3342/ceo.2012.5.3.139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Reed L.J., Muench H. A simple method of estimating fifty per cent endpoints. Am. J. Epidemiol. 1938;27:493–497. doi: 10.1093/oxfordjournals.aje.a118408. [DOI] [Google Scholar]
- 36.Cohen J.M., Chimalapati S., de Vogel C., van Belkum A., Baxendale H.E., Brown J.S. Contributions of capsule, lipoproteins and duration of colonisation towards the protective immunity of prior Streptococcus pneumoniae nasopharyngeal colonisation. Vaccine. 2012;30:4453–4459. doi: 10.1016/j.vaccine.2012.04.080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Choi S.Y., Tran T.D., Briles D.E., Rhee D.K. Inactivated pep27 mutant as an effective mucosal vaccine against a secondary lethal pneumococcal challenge in mice. Clin. Exp. Vaccine. Res. 2013;2:58–65. doi: 10.7774/cevr.2013.2.1.58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Avci F.Y., Kasper D.L. How bacterial carbohydrates influence the adaptive immune system. Annu. Rev. Immunol. 2010;28:107–130. doi: 10.1146/annurev-immunol-030409-101159. [DOI] [PubMed] [Google Scholar]
- 39.Brodsky I.E., Medzhitov R. Targeting of immune signalling networks by bacterial pathogens. Nat. Cell Biol. 2009;11:521–526. doi: 10.1038/ncb0509-521. [DOI] [PubMed] [Google Scholar]
- 40.Keller L.E., Robinson D.A., McDaniel L.S. Nonencapsulated Streptococcus pneumoniae: Emergence and Pathogenesis. MBio. 2016;7:e01792. doi: 10.1128/mBio.01792-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Iannelli F., Pearce B.J., Pozzi G. The type 2 capsule locus of Streptococcus pneumoniae. J. Bacteriol. 1999;181:2652–2654. doi: 10.1128/JB.181.8.2652-2654.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Morona J.K., Miller D.C., Morona R., Paton J.C. The effect that mutations in the conserved capsular polysaccharide biosynthesis genes cpsA, cpsB, and cpsD have on virulence of Streptococcus pneumoniae. J. Infect. Dis. 2004;189:1905–1913. doi: 10.1086/383352. [DOI] [PubMed] [Google Scholar]
- 43.Cartee R.T., Forsee W.T., Bender M.H., Ambrose K.D., Yother J. CpsE from type 2 Streptococcus pneumoniae catalyzes the reversible addition of glucose-1-phosphate to a polyprenyl phosphate acceptor, initiating type 2 capsule repeat unit formation. J. Bacteriol. 2005;187:7425–7433. doi: 10.1128/JB.187.21.7425-7433.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Pelosi L., Boumedienne M., Saksouk N., Geiselmann J., Geremia R.A. The glucosyl-1-phosphate transferase WchA (Cap8E) primes the capsular polysaccharide repeat unit biosynthesis of Streptococcus pneumoniae serotype 8. Biochem. Biophys. Res. Commun. 2005;327:857–865. doi: 10.1016/j.bbrc.2004.12.082. [DOI] [PubMed] [Google Scholar]
- 45.Guidolin A., Morona J.K., Morona R., Hansman D., Paton J.C. Nucleotide sequence analysis of genes essential for capsular polysaccharide biosynthesis in Streptococcus pneumoniae type 19F. Infect. Immun. 1994;62:5384–5396. doi: 10.1128/IAI.62.12.5384-5396.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kolkman M.A., Morrison D.A., Van Der Zeijst B.A., Nuijten P.J. The capsule polysaccharide synthesis locus of streptococcus pneumoniae serotype 14: Identification of the glycosyl transferase gene cps14E. J. Bacteriol. 1996;178:3736–3741. doi: 10.1128/JB.178.13.3736-3741.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hardy G.G., Caimano M.J., Yother J. Capsule biosynthesis and basic metabolism in Streptococcus pneumoniae are linked through the cellular phosphoglucomutase. J. Bacteriol. 2000;182:1854–1863. doi: 10.1128/JB.182.7.1854-1863.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kurola P., Erkkila L., Kaijalainen T., Palmu A.A., Hausdorff W.P., Poolman J., Jokinen J., Kilpi T.M., Leinonen M., Saukkoriipi A. Presence of capsular locus genes in immunochemically identified encapsulated and unencapsulated Streptococcus pneumoniae sputum isolates obtained from elderly patients with acute lower respiratory tract infection. J. Med. Microbiol. 2010;59:1140–1145. doi: 10.1099/jmm.0.016956-0. [DOI] [PubMed] [Google Scholar]
- 49.Fox C.B., Kramer R.M., Barnes V.L., Dowling Q.M., Vedvick T.S. Working together: Interactions between vaccine antigens and adjuvants. Ther. Adv. Vaccine. 2013;1:7–20. doi: 10.1177/2051013613480144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.McKee A.S., Munks M.W., Marrack P. How do adjuvants work? Important considerations for new generation adjuvants. Immunity. 2007;27:687–690. doi: 10.1016/j.immuni.2007.11.003. [DOI] [PubMed] [Google Scholar]
- 51.Arrecubieta C., Lopez R., Garcia E. Molecular characterization of cap3A, a gene from the operon required for the synthesis of the capsule of Streptococcus pneumoniae type 3: Sequencing of mutations responsible for the unencapsulated phenotype and localization of the capsular cluster on the pneumococcal chromosome. J. Bacteriol. 1994;176:6375–6383. doi: 10.1128/jb.176.20.6375-6383.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Hathaway L.J., Stutzmann Meier P., Battig P., Aebi S., Muhlemann K. A homologue of aliB is found in the capsule region of nonencapsulated Streptococcus pneumoniae. J. Bacteriol. 2004;186:3721–3729. doi: 10.1128/JB.186.12.3721-3729.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Battig P., Muhlemann K. Capsule genes of Streptococcus pneumoniae influence growth in vitro. FEMS Immunol. Med. Microbiol. 2007;50:324–329. doi: 10.1111/j.1574-695X.2007.00246.x. [DOI] [PubMed] [Google Scholar]
- 54.Iyer R., Baliga N.S., Camilli A. Catabolite control protein A (CcpA) contributes to virulence and regulation of sugar metabolism in Streptococcus pneumoniae. J. Bacteriol. 2005;187:8340–8349. doi: 10.1128/JB.187.24.8340-8349.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Hamaguchi S., Zafar M.A., Cammer M., Weiser J.N. Capsule prolongs survival of Streptococcus pneumoniae during starvation. Infect. Immun. 2018 doi: 10.1128/IAI.00802-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Sanchez C.J., Kumar N., Lizcano A., Shivshankar P., Dunning Hotopp J.C., Jorgensen J.H., Tettelin H., Orihuela C.J. Streptococcus pneumoniae in biofilms are unable to cause invasive disease due to altered virulence determinant production. PLoS ONE. 2011;6:e28738. doi: 10.1371/journal.pone.0028738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Rubins J.B., Charboneau D., Paton J.C., Mitchell T.J., Andrew P.W., Janoff E.N. Dual function of pneumolysin in the early pathogenesis of murine pneumococcal pneumonia. J. Clin. Invest. 1995;95:142–150. doi: 10.1172/JCI117631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Orihuela C.J., Gao G., Francis K.P., Yu J., Tuomanen E.I. Tissue-specific contributions of pneumococcal virulence factors to pathogenesis. J. Infect. Dis. 2004;190:1661–1669. doi: 10.1086/424596. [DOI] [PubMed] [Google Scholar]
- 59.Briles D.E., Crain M.J., Gray B.M., Forman C., Yother J. Strong association between capsular type and virulence for mice among human isolates of Streptococcus pneumoniae. Infect. Immun. 1992;60:111–116. doi: 10.1128/IAI.60.1.111-116.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Marks L.R., Parameswaran G.I., Hakansson A.P. Pneumococcal interactions with epithelial cells are crucial for optimal biofilm formation and colonization in vitro and in vivo. Infect. Immun. 2012;80:2744–2760. doi: 10.1128/IAI.00488-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Cherny K.E., Sauer K. Pseudomonas aeruginosa requires the DNA-specific endonuclease EndA to degrade eDNA to disperse from the biofilm. J. Bacteriol. 2019 doi: 10.1128/JB.00059-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Magee A.D., Yother J. Requirement for capsule in colonization by Streptococcus pneumoniae. Infect. Immun. 2001;69:3755–3761. doi: 10.1128/IAI.69.6.3755-3761.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Hathaway L.J., Grandgirard D., Valente L.G., Tauber M.G., Leib S.L. Streptococcus pneumoniae capsule determines disease severity in experimental pneumococcal meningitis. Open. Biol. 2016;6 doi: 10.1098/rsob.150269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Hyams C., Camberlein E., Cohen J.M., Bax K., Brown J.S. The Streptococcus pneumoniae capsule inhibits complement activity and neutrophil phagocytosis by multiple mechanisms. Infect. Immun. 2010;78:704–715. doi: 10.1128/IAI.00881-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Nelson A.L., Roche A.M., Gould J.M., Chim K., Ratner A.J., Weiser J.N. Capsule enhances pneumococcal colonization by limiting mucus-mediated clearance. Infect. Immun. 2007;75:83–90. doi: 10.1128/IAI.01475-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Krone C.L., Trzcinski K., Zborowski T., Sanders E.A., Bogaert D. Impaired innate mucosal immunity in aged mice permits prolonged Streptococcus pneumoniae colonization. Infect. Immun. 2013;81:4615–4625. doi: 10.1128/IAI.00618-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Brinkmann V., Reichard U., Goosmann C., Fauler B., Uhlemann Y., Weiss D.S., Weinrauch Y., Zychlinsky A. Neutrophil extracellular traps kill bacteria. Science. 2004;303:1532–1535. doi: 10.1126/science.1092385. [DOI] [PubMed] [Google Scholar]
- 68.von Kockritz-Blickwede M., Goldmann O., Thulin P., Heinemann K., Norrby-Teglund A., Rohde M., Medina E. Phagocytosis-independent antimicrobial activity of mast cells by means of extracellular trap formation. Blood. 2008;111:3070–3080. doi: 10.1182/blood-2007-07-104018. [DOI] [PubMed] [Google Scholar]
- 69.Ochsenbein A.F., Fehr T., Lutz C., Suter M., Brombacher F., Hengartner H., Zinkernagel R.M. Control of early viral and bacterial distribution and disease by natural antibodies. Science. 1999;286:2156–2159. doi: 10.1126/science.286.5447.2156. [DOI] [PubMed] [Google Scholar]
- 70.Murakami A., Moriyama H., Osako-Kabasawa M., Endo K., Nishimura M., Udaka K., Muramatsu M., Honjo T., Azuma T., Shimizu T. Low-affinity IgM antibodies lacking somatic hypermutations are produced in the secondary response of C57BL/6 mice to (4-hydroxy-3-nitrophenyl)acetyl hapten. Int. Immunol. 2014;26:195–208. doi: 10.1093/intimm/dxt057. [DOI] [PubMed] [Google Scholar]
- 71.Leliefeld P.H., Koenderman L., Pillay J. How Neutrophils Shape Adaptive Immune Responses. Front. Immunol. 2015;6:471. doi: 10.3389/fimmu.2015.00471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Ramos-Sevillano E., Ercoli G., Brown J.S. Mechanisms of Naturally Acquired Immunity to Streptococcus pneumoniae. Front. Immunol. 2019;10:358. doi: 10.3389/fimmu.2019.00358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Prevaes S.M., van Wamel W.J., de Vogel C.P., Veenhoven R.H., van Gils E.J., van Belkum A., Sanders E.A., Bogaert D. Nasopharyngeal colonization elicits antibody responses to staphylococcal and pneumococcal proteins that are not associated with a reduced risk of subsequent carriage. Infect. Immun. 2012;80:2186–2193. doi: 10.1128/IAI.00037-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Wilson R., Cohen J.M., Reglinski M., Jose R.J., Chan W.Y., Marshall H., de Vogel C., Gordon S., Goldblatt D., Petersen F.C., et al. Naturally Acquired Human Immunity to Pneumococcus Is Dependent on Antibody to Protein Antigens. PLoS Pathog. 2017;13:e1006137. doi: 10.1371/journal.ppat.1006137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Turner P., Turner C., Green N., Ashton L., Lwe E., Jankhot A., Day N.P., White N.J., Nosten F., Goldblatt D. Serum antibody responses to pneumococcal colonization in the first 2 years of life: Results from an SE Asian longitudinal cohort study. Clin. Microbiol. Infect. 2013;19:E551–E558. doi: 10.1111/1469-0691.12286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Martinot M., Oswald L., Parisi E., Etienne E., Argy N., Grawey I., De Briel D., Zadeh M.M., Federici L., Blaison G., et al. Immunoglobulin deficiency in patients with Streptococcus pneumoniae or Haemophilus influenzae invasive infections. Int. J. Infect. Dis. 2014;19:79–84. doi: 10.1016/j.ijid.2013.10.020. [DOI] [PubMed] [Google Scholar]
- 77.Klein C., Lisowska-Grospierre B., LeDeist F., Fischer A., Griscelli C. Major histocompatibility complex class II deficiency: Clinical manifestations, immunologic features, and outcome. J. Pediatr. 1993;123:921–928. doi: 10.1016/S0022-3476(05)80388-9. [DOI] [PubMed] [Google Scholar]
- 78.Briles D.E., Miyaji E., Fukuyama Y., Ferreira D.M., Fujihashi K. Elicitation of mucosal immunity by proteins of Streptococcus pneumoniae. Adv. Otorhinolaryngol. 2011;72:25–27. doi: 10.1159/000324589. [DOI] [PubMed] [Google Scholar]
- 79.Ibrahim Y.M. Attenuated HtrA-mutant of Streptococcus pneumoniae induces protection in murine models of pneumococcal pneumonia and bacteraemia. Afr. J. Microbiol. Res. 2013;7:237–244. [Google Scholar]
- 80.Ibrahim Y.M., Kerr A.R., McCluskey J., Mitchell T.J. Role of HtrA in the virulence and competence of Streptococcus pneumoniae. Infect. Immun. 2004;72:3584–3591. doi: 10.1128/IAI.72.6.3584-3591.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Cohen J.M., Wilson R., Shah P., Baxendale H.E., Brown J.S. Lack of cross-protection against invasive pneumonia caused by heterologous strains following murine Streptococcus pneumoniae nasopharyngeal colonisation despite whole cell ELISAs showing significant cross-reactive IgG. Vaccine. 2013;31:2328–2332. doi: 10.1016/j.vaccine.2013.03.013. [DOI] [PubMed] [Google Scholar]
- 82.Kim J.O., Romero-Steiner S., Sørensen U.B.S., Blom J., Carvalho M., Barnard S., Carlone G., Weiser J.N. Relationship between Cell Surface Carbohydrates and Intrastrain Variation on Opsonophagocytosis ofStreptococcus pneumoniae. Infect. Immun. 1999;67:2327–2333. doi: 10.1128/IAI.67.5.2327-2333.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Jeong D.G., Jeong E.S., Seo J.H., Heo S.H., Choi Y.K. Difference in Resistance to Streptococcus pneumoniae Infection in Mice. Lab. Anim. Res. 2011;27:91–98. doi: 10.5625/lar.2011.27.2.91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Gingles N.A., Alexander J.E., Kadioglu A., Andrew P.W., Kerr A., Mitchell T.J., Hopes E., Denny P., Brown S., Jones H.B., et al. Role of genetic resistance in invasive pneumococcal infection: Identification and study of susceptibility and resistance in inbred mouse strains. Infect. Immun. 2001;69:426–434. doi: 10.1128/IAI.69.1.426-434.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Kadioglu A., Andrew P.W. The innate immune response to pneumococcal lung infection: The untold story. Trends Immunol. 2004;25:143–149. doi: 10.1016/j.it.2003.12.006. [DOI] [PubMed] [Google Scholar]
- 86.Storisteanu D.M., Pocock J.M., Cowburn A.S., Juss J.K., Nadesalingam A., Nizet V., Chilvers E.R. Evasion of Neutrophil Extracellular Traps by Respiratory Pathogens. Am. J. Respir. Cell Mol. Biol. 2017;56:423–431. doi: 10.1165/rcmb.2016-0193PS. [DOI] [PMC free article] [PubMed] [Google Scholar]