Abstract
Antibiotic resistance is a major global health challenge and, worryingly, several key Gram negative pathogens can become resistant to most currently available antibiotics. Polymyxins have been revived as a last-line therapeutic option for the treatment of infections caused by multidrug-resistant Gram negative bacteria, in particular Acinetobacter baumannii, Pseudomonas aeruginosa, and Enterobacterales. Polymyxins were first discovered in the late 1940s but were abandoned soon after their approval in the late 1950s as a result of toxicities (e.g., nephrotoxicity) and the availability of “safer” antibiotics approved at that time. Therefore, knowledge on polymyxins had been scarce until recently, when enormous efforts have been made by several research teams around the world to elucidate the chemical, microbiological, pharmacokinetic/pharmacodynamic, and toxicological properties of polymyxins. One of the major achievements is the development of the first scientifically based dosage regimens for colistin that are crucial to ensure its safe and effective use in patients. Although the guideline has not been developed for polymyxin B, a large clinical trial is currently being conducted to optimize its clinical use. Importantly, several novel, safer polymyxin-like lipopeptides are developed to overcome the nephrotoxicity, poor efficacy against pulmonary infections, and narrow therapeutic windows of the currently used polymyxin B and colistin. This review discusses the latest achievements on polymyxins and highlights the major challenges ahead in optimizing their clinical use and discovering new-generation polymyxins. To save lives from the deadly infections caused by Gram negative “superbugs,” every effort must be made to improve the clinical utility of the last-line polymyxins.
Significance Statement
Antimicrobial resistance poses a significant threat to global health. The increasing prevalence of multidrug-resistant (MDR) bacterial infections has been highlighted by leading global health organizations and authorities. Polymyxins are a last-line defense against difficult-to-treat MDR Gram negative pathogens. Unfortunately, the pharmacological information on polymyxins was very limited until recently. This review provides a comprehensive overview on the major achievements and challenges in polymyxin pharmacology and clinical use and how the recent findings have been employed to improve clinical practice worldwide.
I. Introduction
The global mortality rate has rapidly declined over the last century after antibiotics were introduced into clinical practice (Armstrong et al., 1999). Unfortunately, resistance to the “magic bullet” antibiotics has become a major global health challenge since the 1990s. Furthermore, increasing development cost, regulatory stringency, scientific challenges, and financial hurdles have led to the disengagement of the pharmaceutical industry from antibiotic discovery and development (O’Neil, 2016). The Infectious Diseases Society of America, the Centers for Disease Control and Prevention, the World Health Organization, and many other major health authorities have highlighted the urgent unmet medical need due to the rapid emergence of resistance and diminishing antibiotic arsenal (Infectious Diseases Society of America, 2004; Centers for Disease Control and Prevention, 2013, 2019; World Health Organization, 2017). In the 2017 World Health Organization Priority Pathogen List, carbapenem-resistant Acinetobacter baumannii, Pseudomonas aeruginosa, and Enterobacteriaceae are identified as the “Priority 1: Critical” pathogens, which desperately require new antibiotics (World Health Organization, 2017). Even though several new antibiotics have been approved by the US Food and Drug Administration (FDA) over the last few years, emergence of resistance has already been reported (Abdallah et al., 2015; de Man et al., 2018; Giddins et al., 2018; Karaiskos et al., 2019; Morrissey et al., 2020). Many clinicians worldwide have been forced to use polymyxins as the last-line therapy to treat life-threatening infections caused by the three aforementioned Gram negative “superbugs” because of their resistance to all currently available antibiotics (Wertheim et al., 2013; Andrei et al., 2019; Zhang et al., 2020). Polymyxins (i.e., polymyxin B and colistin) are an “old” class of lipopeptide antibiotics that were approved in the late 1950s (Li et al., 2006b). However, their use rapidly waned in the 1970s because of their potential nephrotoxicity and the availability of other antibiotics (e.g., aminoglycosides and fluoroquinolones). As neither polymyxin was ever evaluated with the contemporary drug development procedures, there was scarce pharmacological knowledge for polymyxins in the literature until recently. This review presents the latest achievements in the pharmacology of polymyxins, which have improved clinical practice globally. As an example, the first scientifically based dosing recommendations for intravenous colistin [as colistin methanesulfonate (CMS)] underpinned by modern pharmacokinetics/pharmacodynamics (PK/PD) research have been adopted by hospitals worldwide and contributed to the optimization of their use in patients (Nation et al., 2017; Tsuji et al., 2019). In addition, challenges in polymyxin pharmacology research and the recent discovery of new-generation polymyxin-like lipopeptides are also reviewed.
II. History and Chemistry of Polymyxins
A. History of Polymyxins
The naturally occurring lipopeptide polymyxins belong to a class of antibiotics isolated from the Gram positive spore-forming bacterium Paenibacillus polymyxa (also known as Bacillus polymyxa). In 1947, Benedict and Langlykke in the United States first reported the antibacterial activity of the crude mixture from P. polymyxa (Benedict and Langlykke, 1947). At the same time, Stansly, Shepherd, and White of the American Cyanamid Company described the isolation and purification of another antibiotic from the same bacterium, P. polymyxa, and named it as “polymyxin” (Stansly et al., 1947). In England, an antibiotic (aerosporin) from Bacillus aerosporus was isolated and identified by Brownlee and coworkers and was active against certain Gram negative bacteria (Ainsworth et al., 1947; Brownlee and Bushby, 1948). Further research disclosed that polymyxin and aerosporin were basic peptides and possessed very similar antimicrobial activities; therefore, they were classified as the same class of antibiotics (Shepherd et al., 1948; Brownlee et al., 1949a,b; Jones, 1949; White et al., 1949). As both antibiotics were isolated from P. polymyxa, this family of antibiotics was designated as polymyxin, and a nomenclature system was developed internationally (Brownlee, 1949; Stansly and Brownlee, 1949). Later on, the names polymyxin A and polymyxin D were determined for aerosporin and polymyxin, respectively, and other groups of polymyxins, such as polymyxin B, C, and E, were developed (Jones, 1948). Although another antibiotic, colistin, was isolated from Bacillus (Aerobacillus) colistinus in Japan and was initially described as a new class, eventually it was identified that both colistin and polymyxin E are the same compounds (Dautrevaux and Biserte, 1961; Wilkinson, 1963; Suzuki et al., 1965). Both colistin and polymyxin B showed very similar antimicrobial properties (Schwartz et al., 1959–1960; Wright and Welch, 1959–1960). In polymyxins (Table 1), the two major components of the polymyxin B, polymyxin B1 and B2, were first discovered in 1954 (Bell et al., 1949; Catch et al., 1949; Jones, 1949; Wilkinson, 1949; Hausmann and Craig, 1954). The chemical structures of different polymyxins, such as polymyxin B1, polymyxin B2, colistin A (polymyxin E1), colistin B (polymyxin E2), and polymyxin D1 and D2, were determined afterward (Hayashi et al., 1966) (Table 1). To date, polymyxin A to F, M, P, S, and T have been discovered from P. polymyxa strains (Wilkinson and Lowe, 1966; Parker et al., 1977; Shoji et al., 1977a,b; Niu et al., 2013) (Table 1).
TABLE 1.
Chemical structures of the naturally occurring polymyxins
Reprinted by permission from the Springer Nature: Springer, Adv Exp Med Biol, History, chemistry and antibacterial spectrum, Velkov T. et al. Copyright © 2019.
| Polymyxin | Fatty Acyl Group | Position 3 | Position 6 | Position 7 | Position 10 |
|---|---|---|---|---|---|
| A1 | (S)-6-Methyloctanoyl | d-Dab | d-Leu | l-Thr | l-Thr |
| A2 | 6-Methylheptanoyl | d-Dab | d-Leu | l-Thr | l-Thr |
| B1 | (S)-6-Methyloctanoyl | l-Dab | d-Phe | l-Leu | l-Thr |
| B2 | 6-Methylheptanoyl | l-Dab | d-Phe | l-Leu | l-Thr |
| B3 | Octanoyl | l-Dab | d-Phe | l-Leu | l-Thr |
| B4 | Heptanoyl | l-Dab | d-Phe | l-Leu | l-Thr |
| B5 | Nonanoyl | l-Dab | d-Phe | l-Leu | l-Thr |
| B6 | 3-Hydroxy-6-methyloctanoyla | l-Dab | d-Phe | l-Leu | l-Thr |
| B1-Ile | (S)-6-Methyloctanoyl | l-Dab | d-Phe | l-Ile | l-Thr |
| B2-Ile | 6-Methylheptanoyl | l-Dab | d-Phe | l-Ile | l-Thr |
| Dab3-B1 | (S)-6-Methyloctanoyl | d-Dab | d-Phe | l-Leu | l-Thr |
| Dab3-B2 | 6-Methylheptanoyl | d-Dab | d-Phe | l-Leu | l-Thr |
| C1b | 6-Methyloctanoyla | l/d-Dab | d-Phe | l-Thr | l-Thr |
| C2b | 6-Methylheptanoyl | l/d-Dab | d-Phe | l-Thr | l-Thr |
| D1 | (S)-6-Methyloctanoyl | d-Ser | d-Leu | l-Thr | l-Thr |
| D2 | 6-Methylheptanoyl | d-Ser | d-Leu | l-Thr | l-Thr |
| E1 (Colistin A) | (S)-6-Methyloctanoyl | l-Dab | d-Leu | l-Leu | l-Thr |
| E2 (Colistin B) | 6-Methylheptanoyl | l-Dab | d-Leu | l-Leu | l-Thr |
| E3 | Octanoyl | l-Dab | d-Leu | l-Leu | l-Thr |
| E4 | Heptanoyl | l-Dab | d-Leu | l-Leu | l-Thr |
| E7 | 7-Methyloctanoyl | l-Dab | d-Leu | l-Leu | l-Thr |
| E1-Ile | (S)-6-Methyloctanoyl | l-Dab | d-Leu | l-Ile | l-Thr |
| E1-Val | (S)-6-Methyloctanoyl | l-Dab | d-Leu | l-Val | l-Thr |
| E1-Nva | (S)-6-Methyloctanoyl | l-Dab | d-Leu | l-Nva | l-Thr |
| E2-Ile | 6-Methylheptanoyl | l-Dab | d-Leu | l-Ile | l-Thr |
| E2-Val | 6-Methylheptanoyl | l-Dab | d-Leu | l-Val | l-Thr |
| E8-Ile | 7-Methylnonanoyl | l-Dab | d-Leu | l-Ile | l-Thr |
| Fb | 6-Methyloctanoylc | l/d-Dab | d-Leu/d-Ile | l-Leu/l-Ile/l-Ser | l-Leu/l-Ile/l-Ser |
| Fb | 6-Methylheptanoyl | l/d-Dab | d-Leu/d-Ile | l-Leu/l-Ile/l-Ser | l-Leu/l-Ile/l-Ser |
| Fb | Octanoyl | l/d-Dab | d-Leu/d-Ile | l-Leu/l-Ile/l-Ser | l-Leu/l-Ile/l-Ser |
| M1 (Mattacin) | (S)-6-Methyloctanoyl | d-Dab | d-Leu | l-Thr | l-Thr |
| M2 (Mattacin) | 6-Methylheptanoyl | d-Dab | d-Leu | l-Thr | l-Thr |
| P1 | (S)-6-Methyloctanoyl | d-Dab | d-Phe | l-Thr | l-Thr |
| P2 | 6-Methylheptanoyl | d-Dab | d-Phe | l-Thr | l-Thr |
| S1 | 6-Methyloctanoylc | d-Ser | d-Phe | l-Thr | l-Thr |
| T1 | 6-Methyloctanoylc | l-Dab | d-Phe | l-Leu | l-Leu |
| T2 | 6-Methylheptanoyl | l-Dab | d-Phe | l-Leu | l-Leu |
Stereochemistry at C3 and C6 not confirmed.
Position of amino acid residues is speculative.
Stereochemistry at C6 not confirmed.
Soon after their discovery, reversible nephrotoxicity was reported for different polymyxins, and polymyxin B and colistin (polymyxin E) were the least nephrotoxic using in vivo models; however, all of them showed very similar antibacterial activity (Brownlee et al., 1952; Nord and Hoeprich, 1964; Storm et al., 1977). Consequently, both colistin and polymyxin B were further developed for clinical uses and evaluated for toxicity (Barnett et al., 1964; Nord and Hoeprich, 1964; Beveridge and Martin, 1967). For the first time, Stansly and Brownlee (1949) reported that the sulfomethyl derivatives of polymyxins are less toxic and irritant at the injection site compared with the parent antibiotic and possess similar in vivo antibacterial activity (Barnett et al., 1964; Beveridge and Martin, 1967). Since the 1950s, colistin has been commercially available for clinical use as a sulfomethylated derivative, CMS [8068-28-8 Chemical Abstracts Service (CAS) registry number], whereas colistin sulfate (1264-72-8) has been available for intravenous administration only in China. In the clinic, polymyxin B is available as polymyxin B sulfate (1405-20-5) for intravenous administration (Kwa et al., 2007). The commercial products of polymyxin B, CMS, and colistin available for therapeutic uses are in the form of mixtures of major components, such as polymyxin B1, polymyxin B2, colistin A, and colistin B (Kimura et al., 1981; Orwa et al., 2001b; Govaerts et al., 2002a,b). As a consequence, batch-to-batch variations in the abundance of individual components can be observed even in the same commercial products (Decolin et al., 1997; Orwa et al., 2001a,b; He et al., 2010). For example, according to the British Pharmacopeia, the proportions of colistin A and B have to be 50%–75% and 5%–20%, respectively, of the colistin product, and the proportion of polymyxin B1, B2, B3, and B1-I components should not be less than 80% of the total content of the polymyxin B product (British Pharmacopoeia Commission, 2018).
B. Chemistry of Polymyxin B and Colistin
To date, at least 10 individual polymyxin B lipopeptide components have been identified in the literature (Table 1) (Orwa et al., 2001b; Govaerts et al., 2002a; Shaheen et al., 2011). Polymyxin B1 to B6 possess branched and nonbranched N-terminal fatty acyl groups. Polymyxin B1-Ile is highly similar to polymyxin B1, with isoleucine (structural isomer of leucine) at position 7 instead of leucine. Polymyxin B1 and B2 are always the major lipopeptide components. Importantly, variations in proportions of the different lipopeptide components in commercially available polymyxin B products have been reported for different brands and batches of the same manufacturer (He et al., 2010).
For colistin, 11 individual components have been reported, and they have branched and nonbranched N-terminal fatty acyl groups (Table 1) (Orwa et al., 2001b; Govaerts et al., 2002b). Colistin A (polymyxin E1) and colistin B (polymyxin E2) are the two major components. In patients, colistin is administered as a sodium salt of an inactive prodrug, CMS, intravenously or by inhalation (Li et al., 2003a, 2004; Bergen et al., 2006; Garonzik et al., 2011; He et al., 2013). CMS is not stable and converts to its active form colistin in vitro and in vivo (Barnett et al., 1964; Beveridge and Martin, 1967). Because of the uncontrollable sulfomethylation of the Dab residues, the commercial products of CMS consist of a complex mixture of fully and partially sulfomethylated derivatives (He et al., 2013). Importantly, international regulatory compendiums recommend no specific limits on these sulfomethylated derivatives in CMS products (United States Pharmacopeial Convention, 2018, British Pharmacopoeia Commission, 2019, European Pharmacopoeia Commission, 2020).
Overall, the recent advancement of analytical tools for chemical identification and structure elucidation will expedite the development and discovery of new polymyxins with favorable pharmacological and pharmaceutical properties. New-generation polymyxins will strengthen our arsenals to combat antibiotic resistance due to problematic Gram negative pathogens.
III. Antibacterial Spectrum, Mechanisms of Activity, and Resistance
A. Antibacterial Spectrum
The two clinically available polymyxins, colistin and polymyxin B, demonstrate comparable spectra of antibacterial activity, mechanism of action, and resistance because of their similar structures (Kwa et al., 2007). Polymyxins are generally active against Gram negative bacilli and coccobacilli, including Acinetobacter spp. (Kuck, 1976; Catchpole et al., 1997; Gales et al., 2001, 2011, 2012; Tan and Ng, 2006b; Walkty et al., 2009; Queenan et al., 2012), P. aeruginosa (Catchpole et al., 1997; Gales et al., 2001, 2011, 2012; Schulin, 2002; Niks et al., 2004; Tan and Ng, 2006b; Cernohorska and Slavikova, 2009; Walkty et al., 2009; Bogiel et al., 2010; Jones et al., 2013; Zhanel et al., 2013), Haemophilus spp. (Kosakai and Oguri, 1976), Legionella spp. (Thornsberry et al., 1978), Bordetella pertussis (Li et al., 2005a), and Enterobacterales, such as Escherichia coli (Catchpole et al., 1997; Gales et al., 2006, 2011, 2012; Tan and Ng, 2006b; Walkty et al., 2009; Nakamura et al., 2014), Klebsiella spp. (Catchpole et al., 1997; Gales et al., 2006, 2011, 2012; Tan and Ng, 2006b; Walkty et al., 2009; Hawser, 2010; Sader et al., 2011; Nakamura et al., 2014), Salmonella spp. (Catchpole et al., 1997; Gales et al., 2006), Enterobacter spp. (Catchpole et al., 1997; Gales et al., 2006; Walkty et al., 2009), Citrobacter spp. (Catchpole et al., 1997; Gales et al., 2006), and Shigella spp. (Catchpole et al., 1997; Gales et al., 2006). Of these Gram negative bacteria, polymyxin resistance has been increasingly reported in Acinetobacter spp. (Ko et al., 2007; Falagas et al., 2008; Al-Sweih et al., 2011), P. aeruginosa (Landman et al., 2005; Falagas et al., 2008; Johansen et al., 2008), Klebsiella spp. (Antoniadou et al., 2007; Falagas et al., 2008; Suh et al., 2010; Mezzatesta et al., 2011), and E. coli (Liu et al., 2016b; Nang et al., 2019). Worryingly, A. baumannii (Li et al., 2006c; Lo-Ten-Foe et al., 2007; Tan et al., 2007; Hawley et al., 2008; Charretier et al., 2018; Srinivas et al., 2018), P. aeruginosa (Bergen et al., 2011a; Lin et al., 2019a), Klebsiella pneumoniae (Meletis et al., 2011; Jayol et al., 2015; Bardet et al., 2017; Cheong et al., 2019), and Enterobacter cloacae (Lo-Ten-Foe et al., 2007) have been shown to display heteroresistance to polymyxins, which is defined as the presence of a polymyxin-resistant subpopulation [minimum inhibitory concentration (MIC) > 2 mg/l] within a susceptible population (MIC ≤ 2 mg/l). Heteroresistance is of particular concern, as the minor resistant subpopulation would not be detected by the current susceptibility testing in clinical microbiology laboratories and can lead to suboptimal dosing in patients, which may select for the resistant population and result in treatment failure. Further investigations are warranted to examine the impact of heteroresistance on the efficacy of polymyxins in clinical settings, and it is crucial to optimize the current recommended dosage regimens to minimize the emergence of resistance.
Polymyxins have not been shown to be active against a number of Gram negative species, such as Providencia spp. (von Graevenitz and Nourbakhsh, 1972; Catchpole et al., 1997), Serratia spp. (Greenfield and Feingold, 1970; von Graevenitz and Nourbakhsh, 1972; Catchpole et al., 1997; Gales et al., 2006), Proteus spp. (von Graevenitz and Nourbakhsh, 1972; Gales et al., 2006), Vibrio spp. (Lesmana et al., 2002; Gales et al., 2006), Morganella morganii (Gales et al., 2006), Neisseria spp. (Doern and Morse, 1980; Gales et al., 2006), Brucella spp. (Gales et al., 2006), Helicobacter pylori (Glupczynski et al., 1988; García-Rodríguez et al., 1989; Gales et al., 2006), Edwardsiella tarda (Muyembe et al., 1973), Burkholderia cepacia (Gales et al., 2006, 2001), Pseudomonas pseudomallei (Dance et al., 1989), and Moraxella catarrhalis (Gales et al., 2006). Variable susceptibility has been reported against Stenotrophomonas maltophilia (Niks et al., 2004; Tan and Ng, 2006b) and Campylobacter spp. (Kiehlbauch et al., 1992; Aydin et al., 2001). Lastly, at clinically achievable concentrations, polymyxins are not active against Gram positive bacteria, anaerobes, fungi, or parasites (Schwartz et al., 1959–1960; Finland et al., 1976; Storm et al., 1977).
B. Mechanisms of Antibacterial Activity
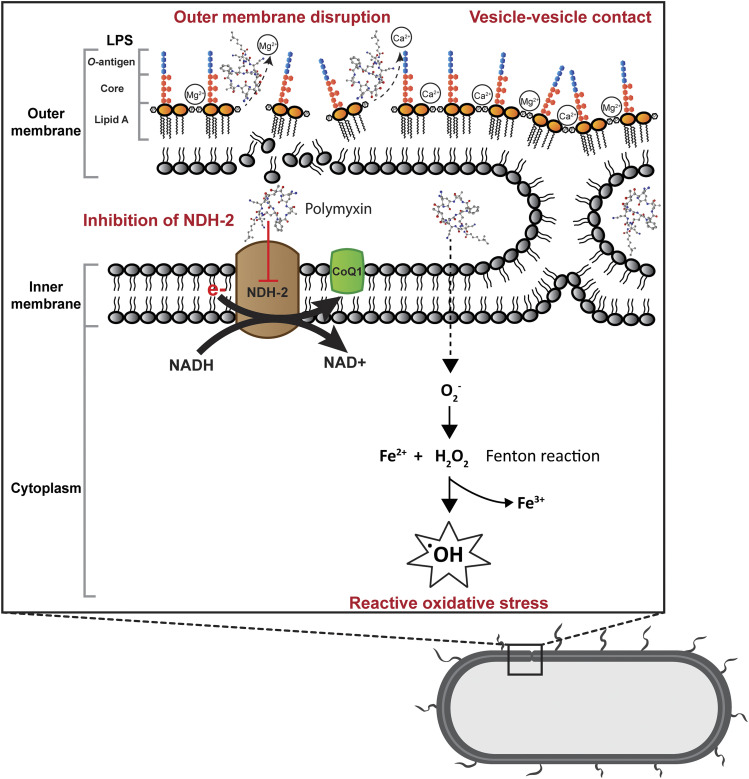
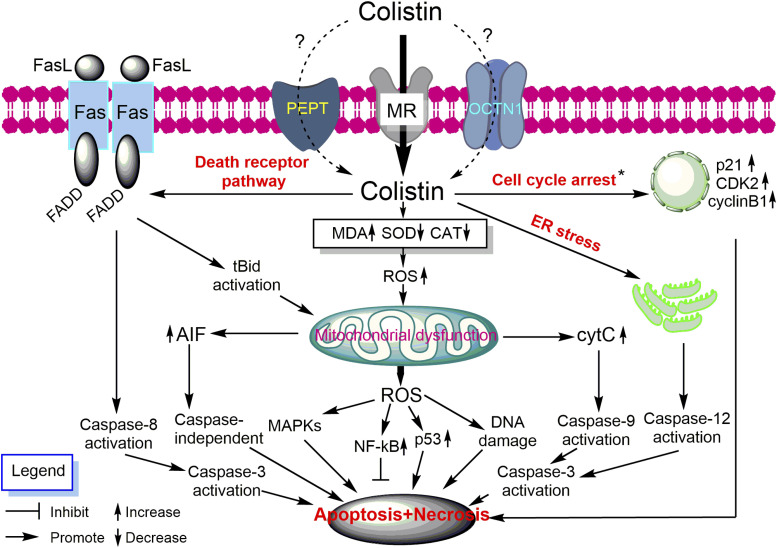
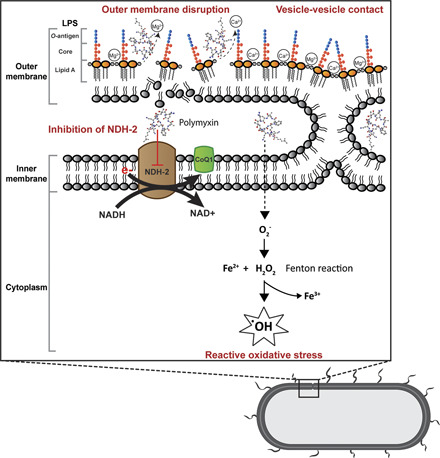
Gram negative bacteria consist of outer and inner membranes (Miura and Mizushima, 1968). The outer membrane is an important barrier against noxious components such as antimicrobial agents (Nikaido, 2003). It is an asymmetric bilayer with an inner leaflet of phospholipids and an outer leaflet that is composed of mainly lipopolysaccharide (LPS) (Zgurskaya et al., 2015). The LPS contains three key domains: O-antigen with an outer repeating polysaccharides unit, an inner core 2-keto-3-deoxyoctonoic acid (Kdo), and lipid A (β-1,6–linked d-glucosamine disaccharide that is phosphorylated at the 1 and 4′ positions with variable number of acyl chains) (Caroff and Karibian, 2003; Kim et al., 2016). The LPS molecules are bridged together by divalent cations (Mg2+ and Ca2+), which bind to the anionic lipid A phosphate groups via electrostatic interaction (Clifton et al., 2015). The primary target of polymyxins on Gram negative cells is the lipid A of the LPS in the outer membrane. Penetration of polymyxins across the outer membrane is highly dependent on the amphipathic property of polymyxins (Hancock, 1997; Clausell et al., 2007; Meredith et al., 2009). The initial binding of polymyxins to the outer membrane of Gram negative bacteria occurs via the initial electrostatic interaction between positively charged L-α,γ-Dab residues of polymyxins and the negatively charged lipid A phosphates. The aforementioned electrostatic interaction causes the displacement of divalent cations (Ca2+ and Mg2+) and destabilizes the outer leaflet of the outer membrane, thus allowing the hydrophobic N-terminal fatty acyl chain and position 6-7 motif (d‐Leu6‐l‐Leu7 for colistin or d‐Phe6‐l‐Leu7 for polymyxin B) to insert into the hydrophobic region of the outer membrane consisting of mainly fatty acyl chains of lipid A (Zhu et al., 2020). This hydrophobic interaction weakens the packing of adjacent fatty acyl chains and leads to expansion of the outer membrane. The insertion of polymyxins into the phospholipid bilayer disrupts the structural integrity of the membrane and promotes other polymyxin molecules to pass through the outer membrane (Velkov et al., 2010; Jiang et al., 2020b). It is postulated that this self-promoted uptake mechanism leads to an increased membrane permeability and leakage of cellular constituents, thus leading to cell death (Velkov et al., 2010; Yu et al., 2015) (Fig. 1). Both polar (e.g., Dab residues) and hydrophobic (N-terminal fatty acyl chain and position 6-7 motif) regions of polymyxins are essential for their interaction with LPS (Jiang et al., 2020b), and a structure-activity relationship model was developed based on the interaction with a single lipid A molecule (Velkov et al., 2010).
Fig. 1.
Mechanisms of antibacterial activity of polymyxins in Gram negative bacteria via disruption of the outer membrane, vesicle-vesicle contact, inhibition of respiratory enzyme NDH-2, and hydroxyl radical formation. CoQ1, coenzyme Q1.
Another proposed mode of action by polymyxins is via vesicle-vesicle contact, whereby polymyxins induce the contacts between outer and inner membranes and cause the exchange of phospholipids between both membranes (Fig. 1). This mechanism is believed to disrupt the phospholipid composition, which results in osmotic imbalance and cell lysis (Cajal et al., 1995, 1996; Clausell et al., 2007; Yu et al., 2015). The disruption of bacterial membrane is supported by transcriptomic and metabolomic analyses, which show the genes and metabolites associated with cell envelope biogenesis and membrane lipids, mainly fatty acids and glycerophospholipids, are significantly perturbated after polymyxin treatment (Henry et al., 2015; Maifiah et al., 2017; Han et al., 2019; Hussein et al., 2019; Lin et al., 2019c).
Accumulation of polymyxins has been observed in the inner membrane and cytoplasm in K. pneumoniae (Deris et al., 2014b). A secondary mechanism of polymyxin antibacterial activity involves the inhibition of NADH oxidases in bacterial respiratory chain in Bacillus subtilis and Mycobacterium smegmatis (Tochikubo et al., 1986; Deris et al., 2014a; Mogi et al., 2009). Notably, polymyxins have been demonstrated to inhibit the inner membrane type II NADH-quinone oxidoreductase (NDH-2) in E. coli, K. pneumoniae, and A. baumannii; and a slightly greater inhibition of NDH-2 by polymyxin B than colistin was reported (Deris et al., 2014a) (Fig. 1). The NDH-2 inhibitory effect was not observed with polymyxin B nonapeptide, colistin nonapeptide, or CMS; therefore, both hydrophobic N-terminal fatty acyl chain and positively charged moieties of polymyxins are important for the NDH-2 inhibitory activity of polymyxins (Deris et al., 2014a). Another secondary mode of action is the induction of endogenous production of reactive oxygen species (ROS) by polymyxins, thus leading to oxidative killing of the bacterial cell via hydroxyl radicals (Sampson et al., 2012; Yu et al., 2015) (Fig. 1). The killing of A. baumannii has been demonstrated via hydroxyl radical production, and inhibiting the formation of hydroxyl radicals attenuates polymyxin killing (Sampson et al., 2012). It is proposed that bactericidal antibiotics usually share a common killing activity via the disruption of metabolism, thus causing the formation of ROS, which eventually leads to bacterial cell death (Kohanski et al., 2007). However, bactericidal activity of polymyxins against P. aeruginosa has been reported to be an independent event of ROS production (Brochmann et al., 2014). The roles of ROS in bacterial killing by polymyxins are therefore inconclusive, and further studies are warranted.
In summary, increasing evidence has been reported on the potential secondary antibacterial mechanisms of polymyxins, including their effects on bacterial metabolism (Han et al., 2019). Elucidation of the exact antibacterial mechanism is crucial for the development of new-generation polymyxins with much-improved killing activity and higher therapeutic indices.
C. Mechanisms of Resistance
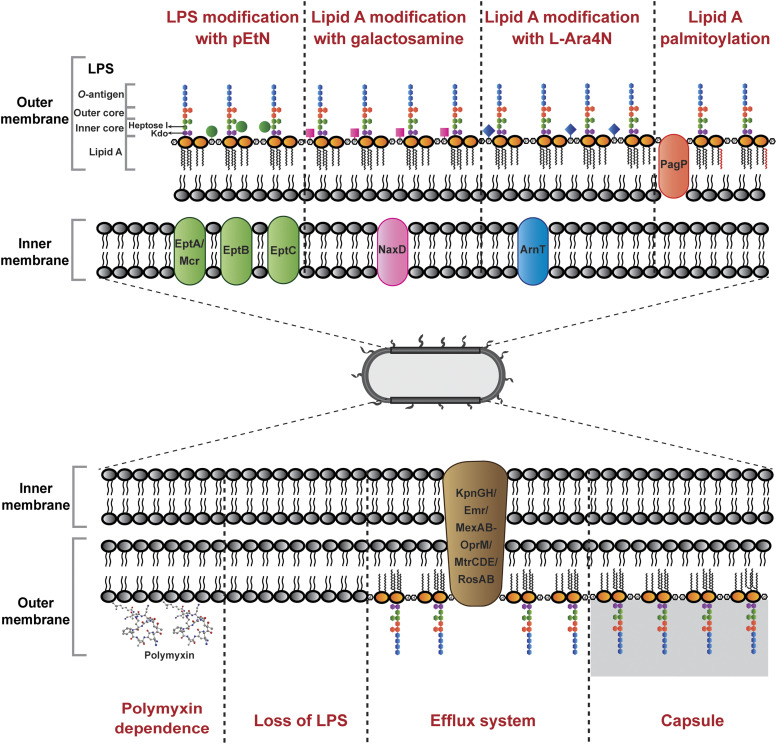
The outer membrane is the initial target of polymyxins, and most resistance mechanisms involve the alteration of the outer membrane to become less permeable to polymyxins. Although the resistance mechanisms vary in different bacterial species, the most common mechanism is LPS modification with the addition of positively charge moieties, such as phosphoethanolamine (pEtN) and 4-amino-l-arabinose (L-Ara4N) (Olaitan et al., 2014; Baron et al., 2016) (Fig. 2). These modifications result in reduced negative charges and impact the initial electrostatic interaction with the positively charged polymyxins (Jiang et al., 2020a).
Fig. 2.
Mechanisms of polymyxin resistance in Gram negative bacteria via lipid A modifications with L-Ara4N, pEtN, galactosamine, and palmitoylation; efflux pump systems, capsule shielding, loss of LPS, and polymyxin dependence.
Modification of the lipid A or Kdo with L-Ara4N has been observed in Gram negative Salmonella enterica (Vaara et al., 1981; Helander et al., 1994; Trent et al., 2001), E. coli (Nummila et al., 1995; Trent et al., 2001), K. pneumoniae (Kidd et al., 2017), P. aeruginosa (Moskowitz et al., 2004), Serratia marcescens (Lin et al., 2014), and B. cepacia (Shimomura et al., 2003). The biosynthesis and addition of L-Ara4N involve a number of enzymes: Ugd (PmrE), ArnB (PmrH), ArnC (PmrF), ArnA (PmrI), ArnD (PmrJ), ArnT (PmrK), ArnE (PmrL), and ArnF (PmrM) (Yan et al., 2007). The initial step for the synthesis of L-Ara4N takes place in the cytoplasm, where UDP-glucose is converted to UDP-glucuronic acid by UgpD, followed by oxidative decarboxylation by ArnA to UDP-4-keto-pyranose (Breazeale et al., 2002; Williams et al., 2005). The UDP-4-keto-pyranose is then converted by the transaminase ArnB to UDP-β-L-Ara4N, which subsequently undergoes formylation to form UDP-β-L-Ara4FN by ArnA (Breazeale et al., 2003; Williams et al., 2005). ArnC then transfers UDP-β-L-Ara4FN to undecaprenyl phosphate carrier located in the inner membrane, where it undergoes deformylation to undecaprenyl phosphate-α-L-Ara4N by ArnD (Breazeale et al., 2005). ArnE and ArnF function to flip the undecaprenyl phosphate-α-L-Ara4N to the outer surface of the inner membrane, where the L-Ara4N is finally transferred to the lipid A portion by the glycosyltransferase ArnT (Trent et al., 2001; Yan et al., 2007). The L-Ara4N moieties can be added to 1- and/or 4′-phosphate group; however, preference to 4′-phosphate has been demonstrated in S. enterica and E. coli (Zhou et al., 2001). The presence of 3′-acyloxyacyl–linked myristate group on the lipid A (conferred by LpxM) is crucial for the addition of L-Ara4N in S. enterica and E. coli (Tran et al., 2005). In contrast, lpxM deletion was reported to have no significant effect on lipid A modification in K. pneumoniae; however, increased susceptibility to polymyxins was observed, and it was postulated that the decreased abundance of acyl chains enhanced the penetration of polymyxins into the membrane (Clements et al., 2007). The same study also reported the occurrence of increased palmitoylation to compensate the loss of myristate (Clements et al., 2007). The outer membrane PagP is related to palmitoylation of lipid A, and its transcription can be stimulated in response to PhoPQ activation (Bishop et al., 2000). Palmitoylation of lipid A is proposed to increase the hydrophobicity of the outer membrane, which prevents the insertion of polymyxins (Fig. 2). The L-Ara4N modification has not been reported in A. baumannii; however, lipid A has been shown to be decorated with structurally similar galactosamine mediated by NaxD (Chin et al., 2015) (Fig. 2). Notably, homologs of UgpD and ArnBCADTEF are absent in A. baumannii.
The modifications of LPS with pEtN have been identified on several locations of LPS, which is dependent on the type of transferase. Addition of pEtN to lipid A has been observed in S. enterica (Zhou et al., 2001; Lee et al., 2004), E. coli (Kim et al., 2006; Liu et al., 2016b), K. pneumoniae (Jayol et al., 2014), and A. baumannii (Arroyo et al., 2011), mediated by the chromosomally encoded eptA (pmrC) and plasmid-mediated mcr (Liu et al., 2016b; AbuOun et al., 2017; Borowiak et al., 2017; Carattoli et al., 2017; Eichhorn et al., 2018; Kieffer et al., 2019; Wang et al., 2018, 2020; Yang et al., 2018); to Kdo in S. enterica (Gibbons et al., 2008) and E. coli (Reynolds et al., 2005) by chromosomally encoded eptB; and to heptose I in E. coli (Salazar et al., 2017) by chromosomally encoded eptC. Similar to the addition of L-Ara4N to lipid A, both 1- and 4′-phosphate groups can be decorated with pEtN moieties; however, in S. enterica, L-Ara4N is preferentially added to the 4′-phosphate and pEtN to the 1-phosphate of lipid A (Zhou et al., 2001). Interestingly, lack of L-Ara4N–modified lipid A has been demonstrated to result in greater decrease in resistance to polymyxins than the loss of pEtN modification in S. enterica, and vice versa in E. coli; this shows that different bacterial species could have different preference for the type of lipid A modifications (Lee et al., 2004; Herrera et al., 2010). Interestingly, modification of lipid A of Vibrio cholerae with glycine and diglycine, mediated by AlmEFG, has been demonstrated to be important for polymyxin resistance (Hankins et al., 2012).
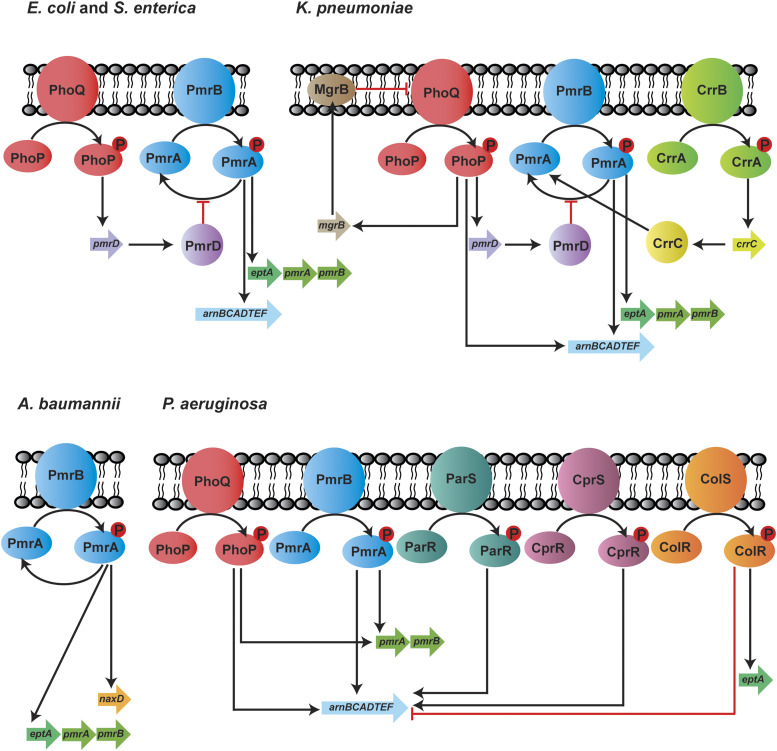
Expression of the chromosomally encoded genes involved in lipid A modifications is commonly regulated by two-component regulatory systems (TCSs), and these can vary in different bacterial species (Fig. 3). In S. enterica, E. coli, and K. pneumoniae, exposure to polymyxins, low pH, and low Mg2+ induce PhoQ and lead to phosphorylation of PhoP and subsequently increase pmrD expression (Gunn et al., 2000; Cheng et al., 2010; Rubin et al., 2015). Inhibition of PmrA dephosphorylation by PmrD results in enhanced PmrA activity, thus leading to continuous transcription of the arn operon and eptA (Gunn et al., 2000; Kox et al., 2000; Cheng et al., 2010; Rubin et al., 2015; Jeannot et al., 2017). Unlike the indirect interaction between PhoPQ and PmrAB via PmrD, the phosphorylated PhoP activates PmrAB directly in P. aeruginosa, leading to the upregulation of arnBCADTEF-ugpD (Macfarlane et al., 1999; Jeannot et al., 2017). Additionally, in K. pneumoniae, the arn operon can be directly activated by PhoPQ (Cheng et al., 2010; Jeannot et al., 2017). Another molecular determinant that plays a key role in polymyxin resistance in K. pneumoniae is MgrB, which is a negative regulator of PhoPQ (Cannatelli et al., 2014). Functional inactivation of MgrB leads to PhoPQ upregulation and subsequently increases the transcription of the downstream genes involved in polymyxin resistance (Poirel et al., 2015). In addition to PhoPQ and PmrAB, CrrAB has been recently identified exclusively in K. pneumoniae as another TCS that participates in polymyxin resistance (Wright et al., 2015; McConville et al., 2020). Mutations in crrB lead to the upregulation of crrC, causing increased expression of arn operon and eptA via the PmrAB system (Cheng et al., 2016). The regulatory systems are far more complex in P. aeruginosa because of the presence of several other TCSs—namely, ParRS, CprRS, and ColRS. ParRS and CprRS induce the arn operon when exposed to antimicrobial peptides (Fernández et al., 2010, 2012). ColRS upregulates eptA but downregulates arnT in the presence of Zn2+; however, Zn2+-induced pEtN modification does not affect polymyxin resistance (Gutu et al., 2013; Nowicki et al., 2015). Thus far, only one TCS (PmrAB) has been identified in A. baumannii to control polymyxin resistance. The activation of PmrAB induces the expression of eptA and naxD, leading to lipid A modification with pEtN and galactosamine, respectively (Chin et al., 2015). The activation of PmrA also results in pmrR expression, which is responsible for the inhibition of lipid A phosphorylase LpxT and reduces the addition of phosphate group to lipid A. This process ultimately promotes resistance to cationic antimicrobial peptides such as polymyxins (Herrera et al., 2010). Additionally, in A. baumannii, polymyxin resistance has been shown to be mediated by increased expression of a homolog of pmrC via the inactivation of a global transcriptional regulator, histone-like nucleoid-structuring protein (Deveson Lucas et al., 2018).
Fig. 3.
Two-component systems regulating polymyxin resistance in E. coli, S. enterica, K. pneumoniae, A. baumannii, and P. aeruginosa.
A very interesting mechanism of polymyxin resistance is the loss of LPS in A. baumannii (Moffatt et al., 2010) (Fig. 2). The loss of LPS is a result of mutations in the lipid A biosynthesis genes lpxA, lpxC, and lpxD (Moffatt et al., 2010, 2011). LPS-deficient A. baumannii isolates lack the initial polymyxin binding target and demonstrate a high level of polymyxin resistance (MIC > 256 mg/l) (Moffatt et al., 2010). Fortunately, LPS-deficient outer membrane appears to be highly permeable and thus increases bacterial susceptibility to other antimicrobial agents, such as rifampicin, cefepime, teicoplanin, and azithromycin (Li et al., 2007; Moffatt et al., 2010). The ability of A. baumannii to survive in the absence of LPS is intriguing, and it has been suggested that this is attributed to the alteration in the production of lipoproteins, phospholipids, and surface polysaccharide poly-β-1,6-N-acetylglucosamine (Henry et al., 2012). Recently, an intriguing polymyxin resistance mechanism was reported in LPS-deficient A. baumannii due to the dependence on polymyxins (Zhu et al., 2020) (Fig. 2). On agar plates, polymyxin-dependent A. baumannii isolates can only be cultured in the presence of polymyxins or polymyxin-like peptides. Importantly, polymyxin-dependent isolates are capable of causing infections in neutropenic mice. This discovery highlights a significant concern in clinic, as polymyxin-dependent resistance phenotype is undetectable with the conventional clinical antibiotic susceptibility testing, which utilizes drug-free agar plates (Zhu et al., 2020).
Polymyxin resistance due to outer membrane proteins and efflux pumps has also been reported (Fig. 2). In K. pneumoniae, inactivation of the KpnGH efflux pump causes an increase in bacterial susceptibility toward several antibiotics, including polymyxin B (Srinivasan et al., 2014). The deletion of emrB in A. baumannii leads to an increased susceptibility to polymyxins, suggesting the role of Emr efflux pumps in polymyxin resistance (Tietgen et al., 2018). The MexAB-OprM efflux system is associated with unspecific adaptive polymyxin resistance in metabolically active subpopulations of P. aeruginosa in biofilm (Pamp et al., 2008). In addition, the efflux pump MtrC-MtrD-MtrE and outer membrane porin PorB are important for the intrinsic polymyxin resistance in Neisseria meningitidis (Tzeng et al., 2005). In Vibrio spp., the outer membrane protein OmpU contributes to polymyxin resistance by regulating sigmaE-associated periplasmic stress response (Mathur and Waldor, 2004; Mathur et al., 2007; Duperthuy et al., 2010). The RosA/RosB efflux pump/potassium antiporter system is identified as a polymyxin resistance contributor in Yersinia enterocolitica, and it is postulated that this system plays a role in the removal of polymyxins from cytoplasm and acidification of the cytoplasm, which leads to the inactivation of polymyxins (Bengoechea and Skurnik, 2000). In addition, the outer membrane remodeling plays a role in bacterial survival in response to polymyxin killing. Upregulation of the genes associated with the maintenance of lipid asymmetry (Mla) system in A. baumannii after colistin treatment suggests the importance of maintaining the asymmetry and integrity of the outer membrane (Malinverni and Silhavy, 2009; Henry et al., 2015).
Another mechanism of polymyxin resistance that is independent of the outer membrane is the involvement of capsule (Fig. 2). Certain capsulated strains of K. pneumoniae are more resistant to polymyxins by reducing the binding of polymyxin molecules to the bacterial outer membrane (Campos et al., 2004). Purified capsular polysaccharides from K. pneumoniae and P. aeruginosa have been demonstrated to bind directly to polymyxins, and the addition of the capsular polysaccharides enhanced polymyxin resistance of an acapsular K. pneumoniae (Llobet et al., 2008). Another mechanism of polymyxin resistance involves a putative serine protease colistinase that degrades colistin that is produced by polymyxin-producing P. polymyxa (Ito-Kagawa and Koyama, 1980). Although inconclusive, in view of the increased production of intracellular ROS in the presence of polymyxins, the colistinase might be necessary for the survival of P. polymyxa during polymyxin synthesis (Ito-Kagawa and Koyama, 1980). Additionally, the antioxidant superoxide dismutase A (SodA) has been reported to play a protective role against polymyxin-induced oxidative stress in P. polymyxa (Yu et al., 2017b). The contribution of Sod to polymyxin resistance has also been illustrated in A. baumannii, in which the knockout of sod2343 led to increased colistin susceptibility (Heindorf et al., 2014).
Overall, mechanisms of polymyxin resistance in Gram negative bacteria are multifaceted. Developing an in-depth understanding of different resistance mechanisms is the key toward minimizing the emergence of polymyxin resistance using combinations with other antibiotics or adjuvant compounds that maximize the killing and target the resistance pathway.
IV. Susceptibility Testing and Breakpoints
A. Susceptibility Testing
The optimal susceptibility testing method had not been developed for polymyxins until recently. It should be noted that colistin sulfate and polymyxin sulfate should be used for measurement of MICs (Clinical and Laboratory Standards Institute, 2015). As CMS is an inactive prodrug of colistin and is not stable in vitro (Bergen et al., 2006), it should not be used for measuring the MIC of colistin. Disk diffusion is among the classic antimicrobial susceptibility testing methods and is still being widely used (Sandle, 2016). However, the size of the inhibitory zone obtained using disk diffusion is often poorly correlated with MICs obtained using the reference broth microdilution method (Matsen et al., 1969). Additionally, many studies have also reported a high false susceptibility rate with disk diffusion and concluded its unreliability (Gales et al., 2001; Tan and Ng, 2006a; Lo-Ten-Foe et al., 2007; Moskowitz et al., 2010; Maalej et al., 2011). Another method for antibiotic susceptibility testing is the gradient diffusion with the use of strips impregnated with predefined gradient of antibiotic concentration (e.g., Etest, bioMérieux and MTS, Liofilchem). A number of studies compared the gradient diffusion method (mostly Etest) to other susceptibility testing methods for polymyxin B and colistin, and variable results have been reported (Arroyo et al., 2005; Lo-Ten-Foe et al., 2007; Tan and Ng, 2007; Behera et al., 2010; Moskowitz et al., 2010; Maalej et al., 2011; Hindler and Humphries, 2013; Landman et al., 2013; Matuschek et al., 2018) with high false susceptibility rates (Arroyo et al., 2005; Hindler and Humphries, 2013; Landman et al., 2013; Matuschek et al., 2018). Overall, disk diffusion and gradient diffusion methods are not recommended, as these methods are associated with relatively high false susceptibility results (Matuschek et al., 2018). Good concordance between agar dilution and broth microdilution was reported in three studies (Lo-Ten-Foe et al., 2007; Behera et al., 2010; Hindler and Humphries, 2013), but conflicting results have also been published (Hogardt et al., 2004; Moskowitz et al., 2010). Notably, a high agreement was demonstrated between broth microdilution and macrodilution methods (Haeili et al., 2019). To date, broth microdilution is the recommended reference method for determining the MICs of polymyxin B and colistin by the Clinical and Laboratory Standards Institute (CLSI) and the European Committee on Antimicrobial Susceptibility Testing (EUCAST) (Clinical and Laboratory Standards Institute, 2020, European Committee on Antimicrobial Susceptibility Testing, 2021).
Automated systems are available commercially for polymyxin MIC measurements in clinical microbiology laboratories. Underestimation of polymyxin resistance has been found with the Vitek 2 system (bioMérieux) and Phoenix automated system (BD Phoenix 100; BD Diagnostic) (Lo-Ten-Foe et al., 2007; Tan and Ng, 2007; Vourli et al., 2017; Jayol et al., 2018). The MicroScan system (Beckman Coulter Diagnostics) demonstrated a categorical agreement of 87% and 88% when compared with agar dilution and broth microdilution, respectively (Lee et al., 2013b; Chew et al., 2017). A high essential agreement with broth microdilution was seen with the use of Sensititre (96%; Thermo Fisher Scientific), MICRONAUT-S (96%; Merlin Diagnostika), and MICRONAUT MIC-Strip (99%; Merlin Diagnostika), whereas a slightly lower essential agreement was reported with SensiTest (88%; Liofilchem) (Matuschek et al., 2018). The same study also reported an essential agreement of 82% with UMIC Colistine kit (Biocentric), whereas a higher essential agreement of 94% was demonstrated in a separate study (Bardet et al., 2019).
Many experimental factors can affect the MIC values for polymyxins; hence, standardization is crucial when performing the susceptibility test for polymyxin B and colistin. The concentrations of Ca2+ and Mg2+ in cation-adjusted Mueller-Hinton broth (CAMHB) are accurately controlled, as these divalent cations influence the activity of polymyxins. The addition of Ca2+ and Mg2+ into broth media can reduce the in vitro activity of polymyxins (Newton, 1954; Davis et al., 1971; Chen and Feingold, 1972; D’Amato R et al., 1975). Very likely, the divalent cations interact with bacterial outer membrane, affecting the interaction between lipid A and polymyxins (Newton, 1954; Chen and Feingold, 1972). The bactericidal effect of polymyxin B against E. coli and P. aeruginosa was inhibited in the presence of 80 mg/l of Ca2+ or 24 mg/l of Mg2+ in Luria-Bertani broth (Chen and Feingold, 1972). Overall, the current standard protocol recommends adjusting CAMHB to final concentrations of 20–25 mg/l of Ca2+ and 10–12.5 mg/l of Mg2+ for the susceptibility testing of polymyxin B and colistin (Clinical and Laboratory Standards Institute, 2018). Another concern is the nonspecific binding of polymyxins to the surface of certain plasticware and glass due to its amphiphilic nature. An overall increase of 5.3-fold in colistin MICs [noncoated microtiter tray (0.54 ± 0.58 mg/l) and tissue culture–coated microtiter tray (2.84 ± 1.93 mg/l)] was observed when broth microdilution was conducted using negatively charged tissue culture microtiter plates coated with polystyrene due to nonspecific binding (Albur et al., 2014). It has also been demonstrated that polymyxin adsorption was higher at lower concentrations and substantial with microtiter plates made of polystyrene, glass, polypropylene, and low-protein-binding polypropylene (Karvanen et al., 2017). Polysorbate 80 (i.e., Tween 80) is a nonionic surfactant and is commonly used to prevent the binding of antimicrobial agents to plastic and other materials during susceptibility testing (Clinical and Laboratory Standards Institute, 2018). The addition of polysorbate 80 to a final concentration of 0.002% resulted in reduced polymyxin MICs; notably, a concentration-dependent binding effect was observed, and the magnitude of MIC reduction was variable in different bacterial strains (Sader et al., 2012; Hindler and Humphries, 2013). Notably, polysorbate 80 may have synergistic antibacterial activity with polymyxins (Brown and Winsley, 1968). The two postulated synergistic mechanisms are as follows: 1) the increased cell permeability by polysorbate 80 enhances the penetration of polymyxins in bacterial cells; 2) the destabilization of bacterial outer membrane by polymyxins facilitates the access of polysorbate 80 into the cell (Brown et al., 1979). However, both studies (Brown and Winsley, 1968; Brown et al., 1979) were conducted prior to the discovery of nonspecific binding of polymyxins to plastic and other materials; hence, the synergistic results reported could be merely an artefact of polymyxin binding to the surface of the experimental vessel used in these early studies (Brown and Winsley, 1968; Brown et al., 1979). Notably, the latest recommended broth microdilution method does not include polysorbate 80 (Clinical and Laboratory Standards Institute, 2020, European Committee on Antimicrobial Susceptibility Testing, 2021).
Overall, the current recommended susceptibility testing of polymyxins should be conducted using the broth microdilution method with CAMHB (final concentrations of 20–25 mg/l of Ca2+ and 10–12.5 mg/l of Mg2+).
B. Polymyxin Breakpoints
Determination of the susceptibility breakpoint for polymyxins has been a challenge. The CLSI (formerly known as National Committee on Clinical Laboratory Standards) published the first polymyxin breakpoints in 1976 based on disk diffusion, which was then abolished in 1980 because of concerns of polymyxin toxicity and the availability of other classes of safer antibiotics (National Committee for Clinical Laboratory Standards, 1976, 1980; Satlin et al., 2020). Revised polymyxin disk diffusion and MIC breakpoints were made available by the CLSI in 2003 (Satlin et al., 2020). Owing to the increasing threat from MDR Gram negative bacteria, colistin breakpoints were established for Enterobacteriaceae, Pseudomonas spp., and Acinetobacter spp. by the EUCAST in 2010 (European Committee on Antimicrobial Susceptibility Testing, 2010). In 2013, colistin breakpoints for P. aeruginosa and Acinetobacter spp. were revised by the CLSI; however, no breakpoint was made for Enterobacteriaceae (Clinical and Laboratory Standards Institute, 2013). Efforts have been made into the interpretation of MIC distribution statistically to separate bacteria into wild-type (susceptible) and non–wild-type (resistant) populations. This leads to the introduction of the epidemiologic cutoff value, referring to MIC values marking the upper end of the wild-type populations. Although PK/PD and clinical data were insufficient to generate a breakpoint for the polymyxins, an epidemiologic cutoff value of colistin (wild-type ≤ 2 mg/l and non–wild type ≥ 4 mg/l) is established by the CLSI for Enterobacteriaceae (E. coli, K. pneumoniae, Klebsiella aerogenes, E. cloacae, and Raoultella ornithinolytica) (Clinical and Laboratory Standards Institute, 2018). In 2020, the CLSI reviewed the polymyxin breakpoints and decided to include the breakpoints for Enterobacterales (previously as Enterobacteriaceae) (Clinical and Laboratory Standards Institute, 2020). However, in this latest CLSI guideline, the “susceptible” interpretive category has been removed, and ≤2 mg/l is set as an “intermediate” breakpoint due to limited clinical effectiveness (e.g., based upon 28-day mortality) of polymyxins, even against Gram negative bacteria with MIC ≤2 mg/l. This change has caused substantial confusions in clinical practice and is not in agreement with the latest polymyxin breakpoints provided by the EUCAST (Satlin et al., 2020). There are minor variations between the guidelines on the breakpoints from the CLSI and EUCAST (Table 2). The MIC breakpoints for colistin and polymyxin B are stated for Enterobacterales, P. aeruginosa, and Acinetobacter spp. by the CLSI and EUCAST (Clinical and Laboratory Standards Institute, 2020, European Committee on Antimicrobial Susceptibility Testing, 2021). The EUCAST has introduced the epidemiologic cutoff value of polymyxin B for Enterobacterales (2 mg/l), P. aeruginosa (4 mg/l), and Acinetobacter spp. (2 mg/l). Notably, polymyxin breakpoints based on the inhibition zone diameter are not established by either CLSI or EUCAST, as the disk diffusion method is not recommended for polymyxin susceptibility testing.
TABLE 2.
MIC breakpoints for colistin and polymyxin B established by CLSI (2020) and EUCAST (2021) guidelines
| MIC breakpoints | ||||||
|---|---|---|---|---|---|---|
| CLSI | EUCAST | |||||
| S | I | R | S | I | R | |
| Colistin | mg/l | |||||
| Enterobacterales | — a | ≤2 | ≥4 | ≤2 | —a | >2 |
| P. aeruginosa (CLSI) Pseudomonas spp. (EUCAST) | —a | ≤2 | ≥4 | ≤2 | —a | >2 |
| Acinetobacter spp. | —a | ≤2 | ≥4 | ≤2 | —a | >2 |
| Polymyxin B | ||||||
| Enterobacterales | —a | ≤2 | ≥4 | —a | —a | —a |
| Pseudomonas spp. | —a | ≤2 | ≥4 | —a | —a | —a |
| Acinetobacter spp. | —a | ≤2 | ≥4 | —a | —a | —a |
I, intermediate; R, resistant; S, susceptible.
Not determined.
In November 2019, the US Committee on Antimicrobial Susceptibility Testing recommended an MIC of ≤2 mg/l as the susceptibility breakpoint for colistin and polymyxin B against P. aeruginosa, A. baumannii, and Enterobacterales (Pogue et al., 2020). Current clinical data are very limited on the efficacy (most with 28-day mortality as the primary outcome) of intravenous CMS and polymyxin B for the treatment of pulmonary infections caused by P. aeruginosa, A. baumannii, and Enterobacterales. Therefore, the US Committee on Antimicrobial Susceptibility Testing susceptibility breakpoint for colistin and polymyxin B is not applicable to lower respiratory tract infections (Pogue et al., 2020). Similarly, considering the very low urinary recovery after intravenous administration, no breakpoint is recommended for polymyxin B for lower urinary tract infections (UTIs) (Sandri et al., 2013a; Pogue et al., 2020). The susceptibility breakpoint for both colistin and polymyxin B after inhalation is not available either. Very limited PK information has shown that concentrations of formed colistin in the epithelial lining fluid (ELF) are within the range of 100–200 mg/l after inhalation of CMS in patients (Gkoufa et al., 2019). Therefore, it is very likely that the susceptibility breakpoint for both colistin and polymyxin B is higher than 2 mg/l for pulmonary infections after inhaled administration due to PK/PD considerations. Clearly, well designed clinical PK/PD studies are required to determine the accurate susceptibility breakpoint for the two polymyxins in different types of infections.
V. Pharmacokinetics and Pharmacodynamics
PK and PD data are essential for optimization of antibiotic use with maximal efficacy and minimal adverse effects. Significant advancements have been made over the last two decades toward the elucidation of PK/PD for polymyxins using in vitro and infected animal models (Li et al., 2006a; Bergen et al., 2012; Cheah et al., 2015; Kaye et al., 2016; Landersdorfer et al., 2018; Nation and Forrest, 2019).
A. Pharmacokinetics
With specific high-performance liquid chromatography and liquid chromatography–mass spectrometry assays, a number of studies investigated the PK of colistin, CMS, and polymyxin B in rats, mice, rabbits, pigs, baboons, and humans (Li et al., 2005a, 2006a,b; Marchand et al., 2019; Nation and Forrest, 2019). The PK of colistin and polymyxin B are comparable in animals, considering that there is only one amino acid difference (Sivanesan et al., 2017b). To date, it is not possible to compare their PK in patients, as an inactive prodrug CMS is used for colistin in the clinic. Colistin sulfate is only available for intravenous administration in China, and future clinical PK results will make the comparison of the two polymyxins possible. In rats, colistin A, colistin B, polymyxin B1, and polymyxin B2 display similar clearance, volume of distribution, elimination half-life, and very low urinary recovery (<1%) (Sivanesan et al., 2017b). Both positively charged Dab residues and the hydrophobic moieties (N-terminus and position 6/7) play key roles in the renal elimination of polymyxins, as polymyxin analogs with fewer Dab residues and polymyxin nonapeptide (i.e., lack of the N-terminus and Dab1) have a higher urinary recovery (Vaara et al., 2010a; Marchand et al., 2019; Vaara, 2019a). The difference in the length of N-terminal fatty acyl group and position 6 (d-Phe for polymyxin B and d-Leu for colistin) causes different plasma binding in rats (colistin A 56.6% ± 9.25%, colistin B 41.7% ± 12.4%, polymyxin B1 82.3% ± 4.30%, and polymyxin B2 68.4% ± 3.50%) (Sivanesan et al., 2017b).
CMS is a mixture of a large number of fully and partially methanesulfonated entities, plus possibly a very small portion of colistin (Li et al., 2019); therefore, determining the PK of CMS is challenging because of the potential conversion of CMS to colistin during the experiment (e.g., blood samples not placed on ice immediately after collection) and measurement of concentrations of CMS and formed colistin (e.g., suboptimal pretreatment procedure). As colistin is polycationic and CMS is polyanionic, their PK are significantly different (renal handling in particular) (Li et al., 2003b, 2004). After filtration by glomeruli, the negatively charged CMS is extensively secreted, whereas the positively charged colistin (and polymyxin B) undergoes extensive reabsorption by renal tubular cells (Li et al., 2004). In animals, CMS is cleared much faster than colistin, and approximately 60% of the CMS dose is eliminated in urine; the high concentration of formed colistin observed in urine is due to the ongoing conversion from CMS in the kidneys and bladder (Li et al., 2004; Marchand et al., 2019; Nation and Forrest, 2019). Limited information is available on the metabolism of polymyxins. A recent mass spectrometry imaging study identified 10 metabolites of polymyxin B1 in rat kidneys, and six of them were also detected in urine samples; similarly, three metabolites of colistin were discovered in urine and kidney homogenates (Nilsson et al., 2015). Metabolism of polymyxin B and colistin in kidneys is mainly via amide hydrolysis, demethylation, and oxidation (Nilsson et al., 2015).
Clinical PK data in healthy volunteers are not available for polymyxin B and only became available recently for CMS in ∼40 health subjects (Couet et al., 2011; Mizuyachi et al., 2011; Zhao et al., 2018). These studies reported higher plasma concentrations of CMS than formed colistin, with CMS achieving its maximum concentration at the end of the infusion, whereas the maximum concentration of formed colistin was achieved hours after the end of the infusion. The renal clearance of colistin was much lower than that of CMS because of the extensive renal tubular reabsorption (Nation and Forrest, 2019). Following normalization of CMS doses, a wide range of achievable plasma colistin concentrations (0.69–3.8 mg/l) were reported in humans across these three studies, and 40%–70% of the CMS dose was recovered in the urine as CMS and formed colistin (Couet et al., 2011; Mizuyachi et al., 2011; Zhao et al., 2018). Overall, the conversion of CMS to colistin in healthy subjects is low. There are at least two clinical PK studies on CMS and formed colistin in a small number of patients with cystic fibrosis using high-performance liquid chromatography assays, and both showed very similar disposition profiles (Li et al., 2003a; Yapa et al., 2014), as well as to those observed in healthy subjects (Couet et al., 2011; Mizuyachi et al., 2011; Zhao et al., 2018). The terminal half-lives of CMS and formed colistin are 2 to 3 hours and 3–8 hours, respectively, indicating that colistin is rate-limited by its own elimination, not the conversion from CMS (Couet et al., 2011; Mizuyachi et al., 2011; Zhao et al., 2018).
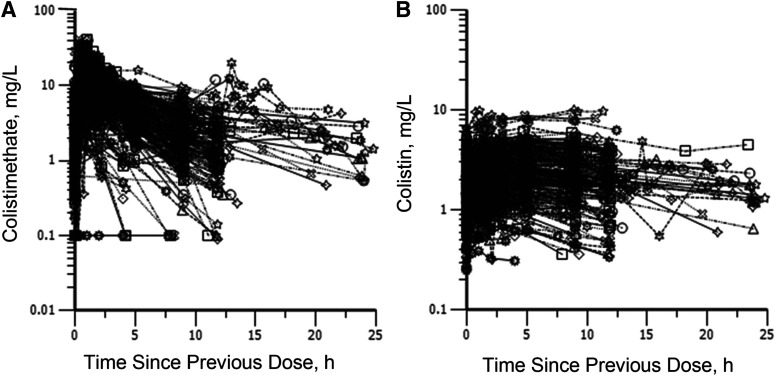
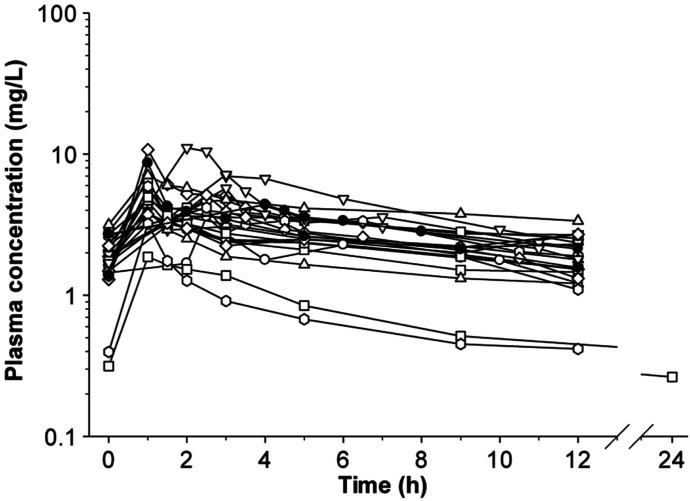
Over the last two decades, there have been a number of clinical PK studies for CMS and formed colistin in ∼250 critically ill patients (Li et al., 2005b; Markou et al., 2008, 2012; Plachouras et al., 2009; Karaiskos et al., 2015; Nation et al., 2017). After the first dose, a slow increase in the concentration of formed colistin was evident in most critically ill patients (Li et al., 2003a, 2005b; Markou et al., 2008, 2012; Plachouras et al., 2009; Yapa et al., 2014; Karaiskos et al., 2015; Nation et al., 2017; Nation and Forrest, 2019). Furthermore, flat plasma concentration versus time profiles of formed colistin were shown at steady state in critically ill patients without renal replacement therapy (Li et al., 2005b; Markou et al., 2008, 2012; Plachouras et al., 2009; Karaiskos et al., 2015; Nation et al., 2017) (Fig. 4). With the current recommended dosage regimens, the average steady-state plasma concentration (Css,avg) of formed colistin is approximately 2 to 3 mg/l in critically ill patients not on renal replacement therapy (Li et al., 2005b; Markou et al., 2008, 2012; Plachouras et al., 2009; Karaiskos et al., 2015; Nation et al., 2017). The mean unbound fraction of colistin in human plasma is 0.49 (Nation et al., 2017). In the largest clinical PK study in critically ill patients to date, significant interpatient variability (up to ∼10-fold) was revealed at a given creatinine clearance, and the apparent clearance of formed colistin was closely correlated to the renal function of the patient (Garonzik et al., 2011; Nation et al., 2017) (Fig. 5). With the current dosage regimens, it is very challenging to achieve Css,avg 2 mg/l of formed colistin in patients with good renal function, as most of the CMS dose is eliminated by the kidneys (Garonzik et al., 2011; Nation et al., 2017). For patients on renal replacement therapy (e.g., intermittent hemodialysis and continuous renal replacement), both CMS and formed colistin are able to be efficiently removed in the extracorporeal unit. As concentrations of CMS are much higher than that of formed colistin in plasma during most of the dosing interval, removal of CMS from blood by hemodialysis significantly decreases the proportion of CMS, which converts to colistin in vivo, thereby impacting the area under the plasma concentration curve (AUC) of formed colistin (Garonzik et al., 2011; Nation et al., 2017). There is very limited information on the PK of CMS and formed colistin in pediatric and burn patients, and prospective clinical studies are warranted to optimize CMS/colistin use in these types of patients (Nation and Forrest, 2019).
Fig. 4.
Steady-state plasma concentrations of colistimethate (A) and formed colistin (B) across a dosage interval in 215 critically ill patients. Patients were receiving colistimethate every 8, 12, or 24 hours. Each symbol represents the data from an individual. Permission obtained from Oxford University Press (Nation et al., 2017).
Fig. 5.
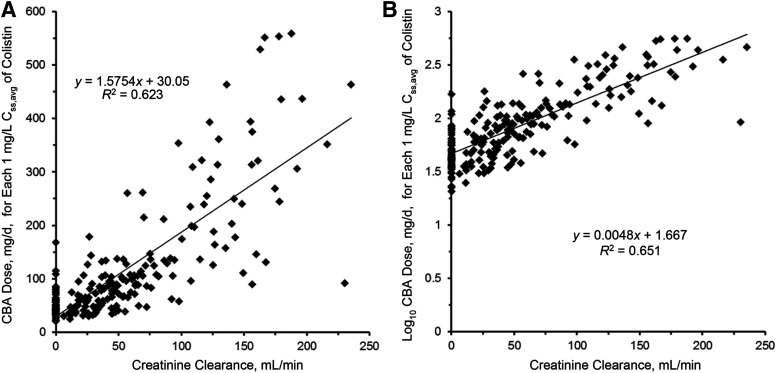
Linear (A) and log-linear (B) plots of the relationship between the daily dose of CBA needed for each 1 mg/l of the average steady-state plasma concentration of colistin (Css,avg) and creatinine clearance. The regression equation in (B) with the intercept adjusted from 1.667 to 1.825 is the renally based dosing algorithm. Permission obtained from Oxford University Press (Nation et al., 2017).
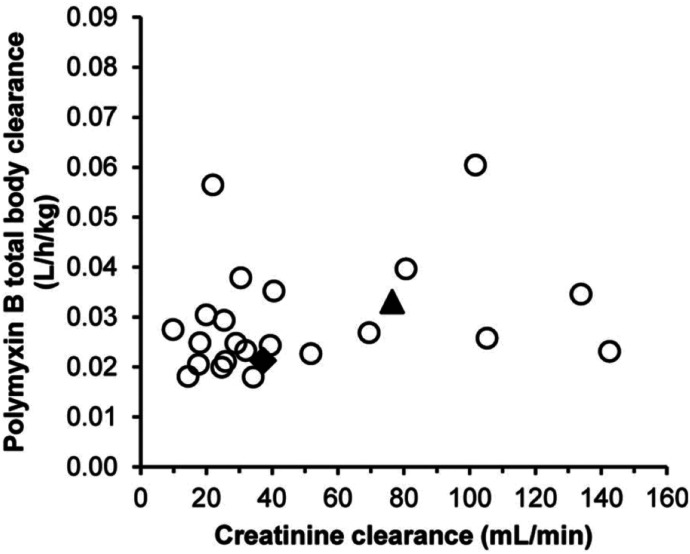
Since 2008 there have been at least seven clinical studies on the PK of polymyxin B in ∼150 patients after intravenous administration (Nation and Forrest, 2019). In a population PK study with 24 critically ill patients, the Css,avg of polymyxin B is ∼2.8 mg/l (range 0.68–4.88 mg/l) with dosage regimens of 0.45–3.38 mg/kg per day (Sandri et al., 2013a). Unlike the slow increase of formed colistin concentrations after intravenous CMS in critically ill patients (Nation et al., 2017) (Fig. 4), polymyxin B reached the peak concentration (∼2.4–14 mg/l) after the infusion (Sandri et al., 2013a) (Fig. 6). Compared with the PK of formed colistin after intravenous CMS, the interpatient variability of polymyxin B clearance (∼3-fold) was much smaller across a wide range of creatinine clearance (Nation and Forrest, 2019). The median unbound fraction of polymyxin B in human plasma is 0.42, and the half-life of polymyxin B in critically ill patients is approximately 12 to 13 hours (Nation and Forrest, 2019). Different from CMS, the urinary recovery of polymyxin B is less than 5%, which is consistent with the results in animals (Nation and Forrest, 2019). One of the most important differences between the PK of polymyxin B and CMS/colistin in critically ill patients is that creatinine clearance is not a covariate of the total body clearance of polymyxin B (Sandri et al., 2013a) (Fig. 7). Current clinical PK data indicate that daily doses of polymyxin B should not be adjusted based on the kidney function of patients due to PK/PD considerations. Therefore, caution is required when clinicians reduce the daily dose of polymyxin B in renally impaired patients based on the inaccurate and outdated product information. There is very limited information on the PK of polymyxin B in patients on renal replacement therapy. Data from two patients showed ∼5%–12% removal of the polymyxin B dose by continuous venovenous hemodialysis during the 12-hour dosing interval (Sandri et al., 2013b). In summary, polymyxin B and CMS have very different PK in patients, and their impacts on the clinical use will be discussed in the PK/PD and toxicities sections below.
Fig. 6.
Plasma concentration-time profiles of polymyxin B in 24 critically ill patients. Permission obtained from Oxford University Press (Sandri et al., 2013a).
Fig. 7.
Individual polymyxin B clearance estimates vs. creatinine clearance in critically ill patients. Polymyxin B clearance was scaled by total body weight (liters per hour per kilogram). Open circles represent patients not on hemodialysis, the filled diamond represents the continuous venovenous hemodialysis patient who weighed 250 kg, and the filled triangle represents the lean continuous venovenous hemodialysis patient. Permission obtained from Oxford University Press (Sandri et al., 2013a).
B. Pharmacodynamics
Concentration-dependent killing against P. aeruginosa by polymyxins (most literature on colistin) has been demonstrated in static time-kill (Li et al., 2001; Tam et al., 2005; Bulitta et al., 2010; Bergen et al., 2011a), in vitro one-compartment dynamic model (IVM) (Bergen et al., 2011b), and hollow-fiber infection model (HFIM) (Tam et al., 2005). As P. aeruginosa is a notorious biofilm producer, it is important to note that concentration-dependent antibiofilm activity of colistin has also been demonstrated using biofilm models (Hengzhuang et al., 2011; Lora-Tamayo et al., 2014). However, eradication of biofilm formed by P. aeruginosa involves a higher colistin concentration and longer exposure as compared with planktonic cells, with mucoid biofilm being harder to eradicate than nonmucoid biofilm (Hengzhuang et al., 2011). Interestingly, in an in vitro model of THP-1 monocyte infection, colistin demonstrated concentration-dependent killing of P. aeruginosa both intracellularly and extracellularly; however, colistin exhibited poorer efficacy against intracellular P. aeruginosa (Buyck et al., 2013). The concentration-dependent killing of polymyxins has also been demonstrated against A. baumannii (Kroeger et al., 2007; Owen et al., 2007; Tan et al., 2007) and K. pneumoniae (Poudyal et al., 2008; Deris et al., 2012; Lin et al., 2019b). Variable postantibiotic effects (PAEs) have been demonstrated across different bacteria, including P. aeruginosa (Li et al., 2001; Bozkurt-Guzel and Gerceker, 2012), A. baumannii (Owen et al., 2007; Plachouras et al., 2007), and K. pneumoniae (Poudyal et al., 2008). It should be noted that at clinically achievable concentrations, the PAE of polymyxin B and colistin is not significant. In addition, the bacterial inoculum also affects the measurement of the killing kinetics and PAE of polymyxins. For example, the extent of killing by polymyxins has been significantly reduced at higher inocula (e.g., 108 cfu/ml) of P. aeruginosa and K. pneumoniae (Tam et al., 2005; Deris et al., 2012). The inoculum effect was also demonstrated in a mechanism-based mathematical model, with 6- and 23-fold slower killing rate at 107 cfu/ml and 109 cfu/ml of P. aeruginosa, respectively, as compared with 106 cfu/ml (Bulitta et al., 2010).
C. Pharmacokinetics/Pharmacodynamics
Mimicking the PK of formed colistin in patients with the recommended maximum daily dose of CMS, different dosing intervals of 8, 12, and 24 hours did not affect the magnitude of colistin antibacterial killing against P. aeruginosa in an IVM study (Bergen et al., 2008). This shows that the ratio of maximum concentration of unbound drug to MIC (fCmax/MIC) is not the most predictive PK/PD index for colistin efficacy (Bergen et al., 2008). An intensive IVM study involving a range of dosing regimens examined the relationship between the three PK/PD indices and efficacy in eradicating P. aeruginosa (Bergen et al., 2010). The results revealed the ratio of area under the plasma concentration curve of unbound drug to MIC (fAUC/MIC) (R2 = 93%) as the PK/PD index that best correlates with the killing activity of colistin, as compared with fCmax/MIC (R2 = 87%) and the duration that the unbound plasma concentration remains above the MIC (fT > MIC) (R2 = 79%). Using the gold-standard murine thigh and lung infection models, fAUC/MIC has also been demonstrated to be the best predictive PK/PD index for colistin and polymyxin B against P. aeruginosa (Hengzhuang et al., 2012; Cheah et al., 2015; Lin et al., 2017a,b), A. baumannii (Cheah et al., 2015; Lin et al., 2018), and K. pneumoniae (Landersdorfer et al., 2018).
The relationship between the magnitude of fAUC/MIC and AUC/MIC with bacterial killing has been evaluated for polymyxins (Table 3). Targeted values of fAUC/MIC for different magnitudes of antibacterial killing by colistin were determined in neutropenic mouse thigh and lung infection models using three strains of P. aeruginosa [American Type Culture Collection (ATCC) 27853, MIC 1 mg/l; PAO1, 1 mg/l; and a clinical MDR mucoid clinical isolate 19056, 0.5 mg/l) and three strains of A. baumannii (ATCC 19606, 1 mg/l, and two MDR clinical isolates: 248-01-C.248, 1 mg/l, and N-16870.213, 0.5 mg/l] (Cheah et al., 2015). Plasma binding of colistin in mouse plasma was determined within the concentration range of approximately 2–50 mg/l using two different methods, and the binding was 92.9% ± 3.3% (mean ± SD%) by ultracentrifugation and 90.4% ± 1.1% by equilibrium dialysis. In the mouse thigh infection model, an fAUC/MIC of 7.4–13.7 for P. aeruginosa and 7.4–17.6 for A. baumannii was required for 2-log10 kill (Cheah et al., 2015). In the mouse lung infection model, colistin showed much weaker efficacy due to poor disposition in the lungs with higher target values of fAUC/MIC (36.8–105) against two of the three P. aeruginosa strains and one strain of A. baumannii; colistin was not even bacteriostatic against the other two A. baumannii strains (Cheah et al., 2015).
TABLE 3.
Studies evaluating the fAUC/MIC targets of polymyxins with different magnitudes of bacterial killing
| Experimental Model | Species | Antibiotic | PK/PD Index | PK/PD Target |
|---|---|---|---|---|
| IVM (Bergen et al., 2010) | P. aeruginosa (n = 3) | Colistin | fAUC/MIC | 1-log10 kill: 5.04–27.1 |
| 2-log10 kill: 6.81–35.7 | ||||
| Neutropenic mouse thigh infection model (Cheah et al., 2015) | P. aeruginosa (n = 3) | Colistin (subcutaneous administration) | fAUC/MIC | Plasma |
| Stasis: 6.0–8.7 | ||||
| 1-log10 kill: 6.6–10.9 | ||||
| 2-log10 kill: 7.4–13.7 | ||||
| Neutropenic mouse lung infection model (Cheah et al., 2015) | P. aeruginosa (n = 3) | Colistin (subcutaneous administration) | fAUC/MIC | Plasma |
| Stasis: 15.2–38.6 | ||||
| 1-log10 kill: 43.3–57.9 | ||||
| 2-log10 kill: 51.8–105 | ||||
| Neutropenic mouse lung infection model (Lin et al., 2017b) | P. aeruginosa (n = 3) | Colistin (pulmonary administration) | AUC/MIC, fAUC/MIC | ELF (AUC/MIC) |
| Stasis: 684–1050 | ||||
| 1-log10 kill: 796–1236 | ||||
| Plasma (fAUC/MIC) | ||||
| Stasis: 2.15–3.29 | ||||
| 1-log10 kill: 2.6–4.68 | ||||
| Neutropenic mouse lung infection model (Lin et al., 2017a) | P. aeruginosa (n = 3) | Polymyxin B (pulmonary administration) | AUC/MIC, fAUC/MIC | ELF (AUC/MIC) |
| Stasis: 1326–1506 | ||||
| 1-log10 kill: 1453–1922 | ||||
| 2-log10 kill: 1693–1778 | ||||
| Plasma (fAUC/MIC) | ||||
| Stasis: 3.14–4.03 | ||||
| 1-log10 kill: 3.73–5.24 | ||||
| 2-log10 kill: 4.49–6.7 | ||||
| Biofilm lung infection model (Hengzhuang et al., 2012) | P. aeruginosa (n = 1) | Colistin (intraperitoneal administration) | AUC/MIC | Serum |
| Stasis: 167 (planktonic), 500 (biofilm) | ||||
| 1-log10 kill: 297 (planktonic), 867 (biofilm) | ||||
| 2-log10 kill: 433 (planktonic), 867 (biofilm) | ||||
| Neutropenic mouse thigh infection model (Cheah et al., 2015) | A. baumannii (n = 3) | Colistin (subcutaneous administration) | fAUC/MIC | Plasma |
| Stasis: 1.4–9.3 | ||||
| 1-log10 kill: 3.5–13.9 | ||||
| 2-log10 kill: 7.4–17.6 | ||||
| Neutropenic mouse lung infection model (Cheah et al., 2015) | A. baumannii (n = 1a) | Colistin (subcutaneous administration) | fAUC/MIC | Plasma |
| Stasis: 11.6 | ||||
| 1-log10 kill: 20.8 | ||||
| 2-log10 kill: 36.8 | ||||
| Neutropenic mouse lung infection model (Lin et al., 2018) | A. baumannii (n = 3) | Colistin (pulmonary administration) | AUC/MIC, fAUC/MIC | ELF (AUC/MIC) |
| Stasis: 326–989 | ||||
| 1-log10 kill: 430–1213 | ||||
| 2-log10 kill: 540–1404 | ||||
| Plasma (fAUC/MIC) | ||||
| Stasis: 0.86–2.18 | ||||
| 1-log10 kill: 1.2–3.29 | ||||
| 2-log10 kill: 1.58–4.45 | ||||
| Neutropenic mouse thigh infection model (Landersdorfer et al., 2018) | K. pneumoniae (n = 3) | Polymyxin B (subcutaneous administration) | fAUC/MIC | Plasma |
| Stasis: 1.22–13.5 | ||||
| 1-log10 kill: 3.72–28 | ||||
| Neutropenic mouse lung infection model (Lin et al., 2018) | K. pneumoniae (n = 3) | Colistin (pulmonary administration) | AUC/MIC, fAUC/MIC | ELF (AUC/MIC) |
| Stasis: 525–2924 | ||||
| 1-log10 kill: 597b | ||||
| 2-log10 kill: 866b | ||||
| Plasma (fAUC/MIC) | ||||
| Stasis: 1.34–6.62 | ||||
| 1-log10 kill: 1.74b | ||||
| 2-log10 kill: 2.44b |
Only one isolate was included, as stasis was not achieved with the highest tolerable dose of colistin against the other two isolates.
Determined with only one isolate.
Similarly, over the range of approximately 0.9–37 mg/l, the binding of polymyxin B in mouse plasma, determined by ultracentrifugation, is 91.4% +/− 1.65% (Landersdorfer et al., 2018). The targeted values of fAUC/MIC for polymyxin B were determined in infected mice using three strains of K. pneumoniae (ATCC BAA-2146 and two clinical isolates, FADDI-KP042 and FADDIKP032; MIC = 0.5 mg/l for all). The fAUC/MIC targets for bacteriostasis and 1-log10 kill of polymyxin B against K. pneumoniae were 1.22–13.5 and 3.72–28.0, respectively, whereas 2-log10 kill was not observed with any strain (Landersdorfer et al., 2018). Polymyxin B and colistin show comparable efficacy against K. pneumoniae with equimolar doses in the mouse thigh infection model (Landersdorfer et al., 2018). In the mouse lung infection model, polymyxin B does not show any killing effect against any of the three K. pneumoniae strains examined, even at the highest subcutaneous dose tolerated by mice (i.e., 120 mg/kg per 24 hours) (Landersdorfer et al., 2018). Collectively, PK/PD results from recent mouse infection studies indicate that parenteral polymyxin B and colistin may not be efficacious against pulmonary infections due to poor PK in the lungs.
Importantly, PK/PD of pulmonary delivery of polymyxins has been recently reported and highlights the superiority of pulmonary delivery over parenteral administration in lung infection model with P. aeruginosa, A. baumannii, and K. pneumoniae (Lin et al., 2017a,b). Additionally, in comparison with colistin, higher fAUC/MIC values were obtained with polymyxin B, which was likely due to different binding affinities of colistin and polymyxin B to plasma and lung surfactant (Lin et al., 2017a,b). These preclinical PK/PD data are essential for the optimization of polymyxin dosing in patients.
Three small clinical studies conducted among 47 critically ill patients demonstrated that, because of the slow and low conversion of CMS to colistin in vivo, a loading dose is important for achieving reasonable concentrations of formed colistin as soon as possible after intravenous CMS (Plachouras et al., 2009; Mohamed et al., 2012; Karaiskos et al., 2015). To date, the largest National Institutes of Health–funded clinical PK study of intravenous CMS in critically ill patients (n = 215) revealed that creatinine clearance was an important covariate in the clearance of both CMS and formed colistin, and adequate plasma concentrations of formed colistin is difficult to achieve, in particular in patients with good renal function (Garonzik et al., 2011; Nation et al., 2017). As an example, for patients not on renal replacement therapy, Css,avg ≥2 mg/l was achieved in >80% of patients with creatinine clearance <80 ml/min, whereas Css,avg ≥2 mg/l was achieved in <40% in patients with creatinine clearance ≥80 ml/min, even with the maximum allowed daily dose [360 mg colistin base activity (CBA)]) (Garonzik et al., 2011; Nation et al., 2017). Considering the difficulty to achieve Css,avg ≥2 mg/l of formed colistin with CMS monotherapy, combination therapy is recommended for intravenous CMS, especially for a patient with a creatinine clearance of ≥80 ml/min and a bacterial pathogen with an MIC of >0.5 mg/l (Garonzik et al., 2011; Nation et al., 2017; Tsuji et al., 2019). Based on recent clinical PK and preclinical PK/PD findings in the literature, the first scientifically based dosing recommendation was proposed for intravenous CMS, and a dosing algorithm was designed for different categories of patients (based on renal function) and the determination of a loading dose based on body weight (Garonzik et al., 2011; Nation et al., 2017). To minimize potential describing errors due to the complex PK and different dose units of CMS around the world, a free clinician-friendly iPhone/iPad app has been developed based on the latest dosing recommendation to help clinicians calculate the dosage regimens of CMS in different types of patients (https://apps.apple.com/us/app/colistindose/id1336806844).
Even though high concentrations of polymyxin B (e.g., 3–14 mg/l) can be achieved after a short infusion of the first dose, Monte Carlo simulations indicated that lack of a loading dose resulted in substantially lower AUC0–24h of polymyxin B on day 1 than at steady state (Sandri et al., 2013a). In contrast to CMS, clinical studies demonstrated that the total body clearance of polymyxin B is not affected by renal function; hence, in terms of PK/PD, polymyxin B is a much better option for patients with good renal function, and its dosage regimens should not be adjusted according to the renal function (Zavascki et al., 2008; Kwa et al., 2011; Sandri et al., 2013a,b; Thamlikitkul et al., 2016; Manchandani et al., 2018; Miglis et al., 2018). The dosing guideline for polymyxin B is currently unavailable; however, a large National Institutes of Health–funded clinical PK, PD, and toxicodynamics (TD) study on polymyxin B is currently being undertaken for the optimization of its dosage regimens in critically ill patients (https://clinicaltrials.gov/ct2/show/NCT02682355). Considering the slow increase in the concentration of formed colistin in patients at the start of therapy (even with a loading dose), polymyxin B has significant PK/PD advantages for the treatment of bloodstream infections; whereas for UTIs, very high concentrations of formed colistin due to the high urinary recovery of CMS make it much more advantageous than polymyxin B. Pneumonia is often the most common infection in the majority of recent polymyxin clinical studies, and the reported poor clinical outcome with intravenous polymyxin B and CMS is very likely due to the suboptimal drug exposure at the infection site (e.g., lungs) and their binding to lung surfactant. As reviewed above, current mouse PK/PD data revealed the very limited efficacy of parenteral CMS and polymyxin B for the treatment of pneumonia and support the clinical results.
There are a number of challenges in conducting clinical PK/PD/TD studies on CMS and polymyxin B. First, most polymyxin-related clinical studies to date employed 28- or 30-day mortality as the primary endpoint. Because of major underlying diseases in critically ill patients, it has been difficult to obtain solid clinical PD data for intravenous polymyxin B and CMS. Microbiological outcome (e.g., bacterial eradication) may serve as a better endpoint for the clinical PK/PD evaluations of intravenous polymyxin B and CMS. Second, many patients were on combination therapy with other antibiotics and nephrotoxic drugs, which has complicated the evaluation of PK/PD and PK/TD of polymyxins. Clearly, future well-designed prospective clinical studies are warranted to optimize the clinical use of this last-line class of antibiotics.
VI. Labeling of Polymyxins
Two different labeling conventions have been used across the world for parenteral CMS products: international unit (IU) and CBA (Li et al., 2006a; Nation et al., 2014). It is important to note that both IU and CBA are formulated according to the antibacterial activity based on in vitro microbiological assays but not the absolute amount of CMS that is present in the product (Li et al., 2006a,b). For the conversion of these two different conventions, ∼80 mg of CMS is equivalent to ∼33.3 mg of CBA and corresponds to 1 MIU. Worryingly, the use of different conventions has been confusing, and an international harmonization has been urgently called upon. Several recommendations have been put in place to minimize confusions. For hospital guidelines and prescription orders, the CMS doses should be specified clearly in IU or CBA (Tsuji et al., 2019). It is recommended that an equivalence should be included for IU and CBA in clinical papers, and an absolute mass of CMS is essential in PK studies (Nation et al., 2014). Fortunately, only one labeling convention (i.e., unit) is made available for polymyxin B. One milligram of polymyxin B base is equivalent to 10,000 U and this corresponds to ∼1.2 mg of polymyxin B sulfate (Lightbown et al., 1973). Similar to CMS, absolute mass of polymyxin B base should be employed for PK studies. Overall, it is essential that clinicians and researchers are familiar with the different terms, labeling conventions and the conversion factors when dealing with polymyxins (Li et al., 2019).
VII. Clinical Utility
Colistin is commercially available in two forms: CMS (for intravenous and inhalation administration) and colistin sulfate (primarily for topical use) (Li et al., 2005a). Colistin sulfate is only available for intravenous administration in China (Li et al., 2019). It is important to note that unfortunately it can be difficult to ascertain the form of colistin used in some studies in the literature, as colistin has been used interchangeably with CMS. In contrast to colistin, polymyxin B is only available in the form of sulfate and can be administered via intravenous, inhalation, and topical routes (Goto and Al-Hasan, 2013; Avedissian et al., 2019; Li et al., 2019).
A. Pulmonary Infections
Lower respiratory infections caused by MDR Gram negative bacteria represent a major global health and economic burden. Polymyxins have been proven to be an important therapeutic option against these difficult-to-treat MDR P. aeruginosa, A. baumannii, and Enterobacterales; however, current clinical knowledge on intravenous administration of polymyxins for the treatment of pulmonary infections is limited, as the intravenous route is less preferable because of low drug exposure in the lungs based on current animal data (Antoniu and Cojocaru, 2012; Landersdorfer et al., 2018; Tsuji et al., 2019). Notwithstanding that, intravenous CMS at 2.5–5 mg/kg per day (with adjustment for patients with renal impairment) was shown to be effective in treating ventilator-associated pneumonia (VAP) caused by A. baumannii and P. aeruginosa that were susceptible to only colistin (Garnacho-Montero et al., 2003; Rios et al., 2007). Monotherapy of intravenous CMS at daily doses of 6–9 MIU (i.e., 480–720 mg) has been shown to be as effective as imipenem and ampicillin/sulbactam in treating VAP caused by MDR and pandrug-resistant A. baumannii and P. aeruginosa (Kallel et al., 2007; Betrosian et al., 2008; Zalts et al., 2016). Importantly, intravenous CMS at 2 MIU (i.e., 160 mg) three times daily was also effective in treating pulmonary exacerbations caused by P. aeruginosa in patients with cystic fibrosis (Conway et al., 1997; Ledson et al., 1998). A study involving 74 patients with nosocomial pneumonia who received intravenous polymyxin B at 1.5–3 mg/kg per day (with dose adjustment according to the renal function) reported a 47% favorable clinical response (Furtado et al., 2007). A high favorable response of 76% was also illustrated in another study with 29 cases of MDR Gram negative respiratory tract infections, in which 21 (72%) cases received intravenous polymyxin B (2.5–3 mg/kg per day with subsequent doses adjusted according to the renal function) with concomitant antibiotic therapy (Sobieszczyk et al., 2004). Both studies suggested that polymyxin B is a potential option for the treatment of MDR Gram negative respiratory infections in which therapeutic options are limited (Sobieszczyk et al., 2004; Furtado et al., 2007). On the contrary, a prospective cohort study was performed with 67 episodes of VAP and ventilator-associated tracheobronchitis treated with either polymyxin B (45 episodes; 67%) and other antimicrobials (22 episodes; 33%) (Rigatto et al., 2013). A higher crude 30-day mortality rate of 53% was observed in the polymyxin B group (median dose of 150 mg/day, ranges from 150 to 200 mg/day) as compared with the comparator group (ceftazidime, meropenem, cefepime, piperacillin-tazobactam, ciprofloxacin, levofloxacin, and imipenem) with 27% of mortality, suggesting that polymyxin B treatment could be inferior to other antibiotics (Rigatto et al., 2013). It is evident that current clinical data on the efficacy of intravenous polymyxin B or CMS against pulmonary infections are not consistent and that future large-scale prospective clinical studies are warranted.
MDR infections in the lower airways are often fatal and difficult to treat, as many parenteral or oral antibiotics cannot reach the bacterial site of infection in the deep lung. Animal and clinical PK/PD studies demonstrate the poor efficacy of parenteral polymyxins for the treatment of Gram negative lung infections (Garonzik et al., 2011), which is mainly due to the insufficient drug penetration from the blood circulation to the infection sites on airway surface with the current dosage regimens. Simply increasing the dose of parenteral polymyxin therapy is not a viable option because of the dose-limiting nephrotoxicity and neurotoxicity (Garonzik et al., 2011; Nation et al., 2017; Velkov et al., 2018a). Considering the PK/PD, inhalation of antibiotics such as polymyxins is very likely more efficacious for the treatment of bacterial lung infections. In clinical practice, inhaled antibiotic therapy is often a complementary treatment in addition to intravenous administration to reduce systemic toxicity (e.g., nephrotoxicity). For chronic lung infections, such as in patients with cystic fibrosis, inhaled therapy is recognized to be more effective than parental administration. A clinical PK study of nebulized CMS (4 MIU) in patients with cystic fibrosis demonstrated high concentrations of colistin in the sputum (Cmax 4–16 mg/l) for a prolonged period (>3 mg/l at 12 hours), with negligible systemic exposure (Cmax <0.5 mg/l in plasma) (Yapa et al., 2014).
Inhalation products of CMS [nebulization and dry powder inhalers (DPIs)] have been approved in several European countries and Australia, but no polymyxin products are approved in the United States for inhalation. Several clinical studies reported the efficacy of inhaled CMS therapy for lung infections in patients with bronchiectasis (Haworth et al., 2014), cystic fibrosis (Schuster et al., 2013), and pneumonia (Kwa et al., 2005). Nebulized CMS is increasingly used for MDR Gram negative lung infections in patients with cystic fibrosis as a complementary treatment to intravenous use and the use of nebulized CMS for lung infections in patients without cystic fibrosis is also common. Vardakas et al. (2018) systematically reviewed and conducted a meta-analysis on 12 clinical studies on nebulized CMS monotherapy for respiratory infections in adults with non–cystic fibrosis. Among these 12 studies (Kwa et al., 2005; Motaouakkil et al., 2006; Lin et al., 2010; Rattanaumpawan et al., 2010; Athanassa et al., 2012; Kuo et al., 2012; Lu et al., 2013; Chen et al., 2014; Choi et al., 2014; Maskin et al., 2015; Abdellatif et al., 2016; Hsieh et al., 2016), 10 included patients with pneumonia, and two included patients with ventilator-associated tracheobronchitis. MDR A. baumannii and P. aeruginosa were the major causative pathogens of the lung infections in these studies. It was revealed that nebulized CMS therapy is generally safe, and the clinical and microbiological efficacies in patients with non–cystic fibrosis are encouraging (Vardakas et al., 2018). It was recommended by the study that nebulized CMS therapy should be considered for the treatment of lung infections against polymyxin-susceptible MDR Gram negative bacteria (Vardakas et al., 2018). As compared with nebulization, DPI products become popular for inhaled therapy of high-dose antibiotics (Zhou et al., 2015). Nebulization often requires bulky devices, tedious preparation (frequent cleaning), oversight by professionals, and long administration time (Tiddens et al., 2014). Furthermore, traditional jet nebulization technologies generally have very low drug delivery efficiency (Reychler et al., 2004), with only 9%–15% of drug deposited in the lungs after CMS nebulization (Boisson et al., 2014). These issues can be overcome by DPIs, as generally they are cheaper, more portable, easier to use, less labor intensive, and have better patient compliance (Zhou et al., 2014). A phase 3 centrally randomized open-label study was conducted for a dry powder inhalation product, Colobreathe, in 380 patients with cystic fibrosis with chronic P. aeruginosa lung infections (Schuster et al., 2013). Patients in the Colobreathe group received CMS powder inhalation of 1,662,500 IU twice a day for 24 weeks, whereas the other cohort received 300 mg/5 ml tobramycin nebulized solution twice daily for three 28-day cycles with a 28-day off period after each tobramycin course; Colobreathe treatment has the same efficacy as tobramycin treatment in maintaining lung function (Schuster et al., 2013). The major benefit for such a DPI product is the convenience for use (90.7% for Colobreathe vs. 53.9% for tobramycin, P < 0.001) (Schuster et al., 2013). Although the total number of adverse events was similar in both groups, higher number of coughs, throat irritation, and treatment withdrawals due to adverse events were reported for the Colobreathe group (Schuster et al., 2013). Such respiratory adverse events have often been reported for inhaled polymyxins, likely because of the local toxicity to the airways. As suggested by the clinical studies to date, it appears that inhaled CMS has fewer adverse effects than inhaled polymyxin B and colistin sulfate (Velkov et al., 2015); however, there is a lack of well designed clinical study to substantiate such implications. It should be noted that CMS is an inactive prodrug and needs to be converted to active colistin in vivo (Bergen et al., 2006). The conversion of CMS to colistin after the inhalation is only 1.4% in the lung ELF (Boisson et al., 2014; Yapa et al., 2014). Although inhaled polymyxin therapies have been clinically used for several decades (e.g., in people with cystic fibrosis in Europe), it is worth noting that current inhaled polymyxin therapies have not been subjected to systematic PK/PD/TD optimization, and the dosage regimens are empirical (Velkov et al., 2015). Inhalation of injectable CMS and polymyxin B in the United States is of off-label use for the treatment of chronic MDR lung infections and has not been approved by the FDA (Moskowitz et al., 2008). Because of the current empirical dosing practice, excessive high doses of CMS have been used for nebulization (up to 6 MIU/day) and DPI (3.3 MIU/day) (Conole and Keating, 2014), which are comparable to the intravenous dosage regimens (Yapa et al., 2014). There is a lack of PK/PD targets for inhaled polymyxins even though the inhaled CMS products have been approved for many years in the United Kingdom and several European countries.
Recent PK/PD studies in the mouse lung infection model demonstrated that AUC/MIC in ELF is the most predictive PK/PD index for inhaled polymyxins (Lin et al., 2017a,b). However, because of the challenges in the inhalation dose translation from mice to humans and the lack of solid PK/PD data in patients, the AUC/MICELF target for various antibacterial magnitudes in patients is still unknown, and optimizing clinical dosage regimens for inhaled polymyxin B and CMS is urgently needed. Considering potentially different infection sites in the lungs, it may be prudent to use both intravenous and inhalation polymyxins for the treatment of pneumonia.
B. Bacteremia
The concern with high mortality rates associated with bacteremia has been escalated because of the rise of MDR Gram negative bacteria such as P. aeruginosa, A. baumannii, and K. pneumoniae (Anderson et al., 2006; Tumbarello et al., 2011; Nielsen, 2015; Zhou et al., 2019). A retrospective study investigated the impact of CMS dosage on the clinical and microbiological cure in 127 patients with bacteremia, 82 (65%) of whom received low-dosage regimens of CMS (≤4.4 mg CBA/kg per day), and 45 (35%) received high-dosage regimens (>4.4 mg CBA/kg per day) (Gibson et al., 2015). The high-dose CMS was significantly associated with greater 7-day clinical cure (40%) and microbiological cure (84%) and lower mortality rate (6.7%) as compared with the low-dose CMS (22%, 67%, and 22%, respectively) (Gibson et al., 2015). Similarly, the same group also reported the association between a higher CMS dose (2.9 mg CBA/kg per day (1.70–3.68)] with greater 7-day microbiological cure and lower 7-day mortality rate in a retrospective study with 76 patients with bacteremia at a large, academic, tertiary-care medical center in the United States between 2005 and 2010 (Vicari et al., 2013). Notably, CMS dose was not associated with the 28-day mortality rate in either study, whereas a higher CMS dose was associated with a higher rate of acute kidney injury (3.8 vs. 1.6 mg CBA/kg per day) (Vicari et al., 2013). Therefore, it is important to reconsider whether 28-day (or 30-day) mortality is an appropriate clinical endpoint for the evaluation of polymyxin efficacy in patients.
Another retrospective study from South Korea examined the effect of the desired target of colistin Css,avg on the efficacy and safety of intravenous CMS in 153 critically ill patients with pneumonia (112 patients; 73%) and bacteremia (41 patients; 27%) caused by P. aeruginosa and A. baumannii (Jung et al., 2019). A greater microbiological eradication in patients with bacteremia (28 patients; 58%) was observed as compared with those with pneumonia (20 patients; 42%). It should be noted that all 153 patients received intravenous CMS, and only 19 (12%) were given additional inhaled CMS therapy. Given that only intravenous CMS was administered in most of these pneumonia cases, this might lead to inadequate exposure of formed colistin in the lungs and lower bacterial eradication (Jung et al., 2019). Additionally, successful treatment of K. pneumoniae associated bacteremia was reported in another case study with intravenous CMS at 9 MIU per day in three divided doses (Karabinis et al., 2004). The impact of polymyxin B dosage regimens on in-hospital mortality was analyzed in a retrospective cohort study involving 276 patients receiving intravenous polymyxin B for ≥72 hours (Elias et al., 2010). Among 276 patients, 53 (19.2%) patients were presented with bacteremia caused by P. aeruginosa (32 patients; 60.4%) and A. baumannii (21 patients; 39.6%). Intravenous polymyxin B at dosage regimens of ≥200 mg per day resulted in lower mortality in patients with bacteremia (adjusted odds ratio 0.04; 95% confidence interval 0.006–0.28) but was associated with higher risk of severe renal impairment (Elias et al., 2010). Another retrospective study from the same group compared the efficacy of intravenous polymyxin B with other antibiotics for the treatment of P. aeruginosa bacteremia in 133 patients (Kvitko et al., 2011). In this cohort, 45 (34%) patients received polymyxin B, and 88 (66%) patients received other antibiotics. The mean average daily polymyxin B dose was 141 ± 54 mg in two divided doses, with 11 (24%) patients receiving a dose ≥200 mg per day. The inferiority of polymyxin B for the treatment of P. aeruginosa bacteremia was demonstrated, with a higher mortality associated with polymyxin B (30 of 45; 67%) in comparison with other antibiotics (25 of 88; 28%). Furthermore, a higher incidence of nephrotoxicity was revealed with the use of polymyxin B (Kvitko et al., 2011). A multicenter case study examined the risk factors and outcomes of extensively drug-resistant (XDR) A. baumannii bacteremia (Ng et al., 2014). For patients receiving polymyxin B, survivors at 30 days had been given a higher daily dose (median of 840,000 IU) of polymyxin B as compared with nonsurvivors (median of 700,000 IU) (Ng et al., 2014). Overall, higher concentrations of polymyxins have been associated with better clinical outcomes. However, increased concentrations have often resulted in renal impairment. These reports highlight the importance of using an appropriate dosage of polymyxins.
C. Urinary Tract Infections
UTI is one of the most common nosocomial infections with Enterobacterales, and carbapenems are often the first choice of therapeutic agents (Stamm and Norrby, 2001). Unfortunately, the increasing emergence of carbapenem-resistant Enterobacterales has limited the treatment options to polymyxins in many hospitals (Cassir et al., 2014; Dortet et al., 2014). The achievable concentrations of formed colistin and polymyxin B in urine are different in patients because of the different renal handling of CMS (mainly eliminated by the kidneys) from polymyxin B (extensively reabsorbed by the renal tubular cells) (see section V. Pharmacokinetics and Pharmacodynamics). A total number of 33 patients with symptomatic UTI (24 cystitis and 9 pyelonephritis) caused by P. aeruginosa were given intravenous CMS (5 ± 2.54 MIU per day) for at least 48 hours, 19 of whom (16 cystitis and 3 pyelonephritis) were treated with CMS monotherapy (Sorlí et al., 2019). A high clinical cure rate (17 patients; 90%) was achieved even though only five (30%) patients achieved the optimal plasma PK/PD target (fAUC/MIC ≥ 60), with one (6%) patient attaining the Css target in plasma (2.5 mg/l). Among patients receiving CMS monotherapy with clinical cure, five (30%) patients were presented with acute kidney injury. Based on the PK/PD of formed colistin in urine, lower CMS doses were suggested for the treatment of UTI with a reduced risk of nephrotoxicity (Sorlí et al., 2019). Another study also reported a high clinical cure of 83% with a low dose of intravenous CMS (1.9 ± 0.6 mg/kg; it is unclear whether the dose unit is mg CBA) for the treatment of UTI by MDR Gram negative bacteria; however, it was not stated whether CMS was used as monotherapy or in combination with other antibiotics (Zaidi et al., 2014).
In contrast to CMS, a much lower urinary recovery (<4%) was reported with intravenous polymyxin B in critically ill patients because of the low renal clearance (Zavascki et al., 2008; Sandri et al., 2013a). However, successful use of polymyxin B for the treatment of UTIs has been reported. Efficacious treatment of UTI with polymyxin B (1.5–2.5 mg/kg per day) was demonstrated among 92 solid organ transplant patients with 77.2% clinical cure and 100% microbiological cure (only 24 follow-up urine cultures from patients were obtained) (Mostardeiro et al., 2013). Notably, renal dysfunction was associated with the duration of polymyxin B treatment (Mostardeiro et al., 2013). Another retrospective study was performed with 87 cases of UTI caused by carbapenem-resistant K. pneumoniae, and 25 (29%) of these cases involved the use of polymyxin B (median of 2.25 mg/kg per day, range of 1.1–3.3 mg/kg per day) (Satlin et al., 2011). Polymyxin B demonstrated a superior microbiological clearance rate (64%) as compared with the untreated (36%) and tigecycline (43%) groups; however, it is inferior to aminoglycosides (88%) (Satlin et al., 2011). In another retrospective study, a clinical cure rate of 73% was observed among 23 patients treated with polymyxin B (1.5–2.5 mg/kg per day), including 10 patients with UTI caused by carbapenem-resistant K. pneumoniae (Dubrovskaya et al., 2013).
Bladder instillation is an alternative administration route for polymyxins to treat UTIs. A 7-day continuous intravesical administration of CMS in saline (3.5 mg/kg of CMS in 500 cm3) was effective in eradicating MDR A. baumannii while sparing the renal function in a patient (Volkow-Fernández et al., 2012). Efficacy of bladder instillation with CMS was also reported in three patients with UTI caused by MDR A. baumannii (100,000 IU in 50 ml via a single urinary catheter three times daily; 2 days for one patient and 7 days for two patients) (Giua et al., 2014). In another study with 17 patients with acute cystitis, an overall effectiveness rate (based on the degree of bacteriuria, pyuria, and pain on micturition) of 82% was observed with intravesical instillation of polymyxin B (Suzuki et al., 1989). Nonetheless, conflicting results were also presented in the literature showing that bladder irrigation with a combination of polymyxins and aminoglycosides (kanamycin-colistin/neomycin-polymyxin B) was ineffective (Pearman et al., 1988; Waites et al., 2006). With very limited pharmacological knowledge, intravesical instillation of polymyxins remains a potential alternative for the treatment of UTIs, especially in patients with severe renal impairment. Further investigations are warranted to optimize the administration method and dosing strategy of polymyxins for treating UTIs.
D. Central Nervous System Infections
Very limited therapeutic options are available for central nervous system (CNS) infections caused by MDR Gram negative bacteria, largely because of the blood-brain barrier. As intravenous polymyxins demonstrate poor CNS penetration, intrathecal and intraventricular routes have been employed to achieve high polymyxin concentrations in the CNS (Falagas et al., 2007; Jin et al., 2009, 2011). A case study examined intraventricular CMS for the treatment of ventriculitis caused by carbapenem-resistant A. baumannii in five patients with ventriculitis; two patients who received intraventricular CMS (5 mg twice daily) plus other intravenous antibiotics (sulbactam-tobramycin or tobramycin) survived, whereas the remaining three patients without intraventricular CMS died as a result of inadequate therapy (Fernandez-Viladrich et al., 1999). Similarly, intraventricular CMS at 125,000 IU (i.e., 10 mg) twice daily for 3 weeks successfully treated a patient with meningitis caused by MDR A. baumannii (Bukhary et al., 2005). Another successful case employed a lower daily dose of intrathecal CMS at 40,000 IU (i.e., ∼3.2 mg) for meningitis caused by A. baumannii (Benifla et al., 2004). Several other clinical studies showed that intrathecal and intraventricular CMS administration was effective against A. baumannii meningitis with daily doses of 3.2–20 mg (Vasen et al., 2000; Berlana et al., 2005; Al Shirawi et al., 2006) and P. aeruginosa ventriculitis, meningitis, and ventriculoperitoneal shunt infection with daily doses of 5–10 mg (Quinn et al., 2005; Yagmur and Esen, 2006; Baiocchi et al., 2010). The efficacy of intrathecal and intraventricular CMS with a daily dose of 125,000 IU was reported for the eradication of extended-spectrum β-lactamase (ESBL)-producing K. pneumoniae from the cerebrospinal fluid (Enfanto and Shin, 2007). A case series was conducted for the treatment of CNS infections caused by A. baumannii in 24 patients; 11 received intrathecal/intraventricular CMS [40,000–400,000 IU/day (3.2–32 mg/day)] as monotherapy, and 13 received coadministered systemic antibiotics (Khawcharoenporn et al., 2010). High clinical and microbiological cure rates were demonstrated in both groups of patients receiving intrathecal/intraventricular colistin monotherapy (91% and 91%, respectively) and combinations with systemic antibiotics (77% and 92%, respectively). Among the patients receiving intrathecal/intraventricular CMS, several patients experienced chemical ventriculitis (3 of 24; 13%) and treatment-associated seizures (1 of 24; 4%) (Khawcharoenporn et al., 2010). In another case series, six patients with meningitis and ventriculitis caused by XDR A. baumannii were successfully treated with intraventricular CMS with a loading dose of 500,000 IU (i.e., 40 mg) followed by 125,000–250,000 IU (i.e., 10–20 mg) every 24–48 hours in addition to intravenous CMS (Karaiskos et al., 2013). Notably, a patient (17%) developed chemical meningitis, and another one (17%) experienced chemical ventriculitis (Karaiskos et al., 2013). Several other clinical studies showed the efficacy of intrathecal/intraventricular CMS against difficult-to-treat CNS infections. A recurrent episode of meningitis was resolved only after the administration of the combination consisting of intraventricular amikacin, intraventricular CMS, and intravenous CMS for 6 weeks (Kasiakou et al., 2005b). The combination of intravenous and intrathecal/intraventricular CMS has been used by many clinicians as an effective and safe therapeutic option against CNS infections caused by MDR Gram negative bacteria, including in critically ill patients and children (Schina et al., 2006; Ho et al., 2007; Rodríguez Guardado et al., 2008; Dalgic et al., 2009; Ozdemir et al., 2010; Ziaka et al., 2013). There is very limited PK information on polymyxins after intrathecal/intraventricular administration in patients. In a study with nine patients, after administration of intraventricular CMS at 2.61–5.22 mg per day, cerebrospinal fluid concentrations of formed colistin were above 5.41 mg/l over 72 hours, which cannot be achieved with intravenous administration (Imberti et al., 2012). In addition, colistin did not accumulate in the CSF during this period, and intraventricular administration of CMS at doses of ≥5.22 mg per day seems appropriate in patients considering the PK/PD and safety of colistin. Given that the variable external cerebrospinal fluid efflux may affect the clearance of colistin, this study supports the recommended daily dose of 125,000 IU CMS (i.e., 10 mg), as suggested by the Infectious Diseases Society of America (Imberti et al., 2012).
In a systemic review (64 episodes), a high cure rate of 80% (51 of 64) was reported for treatment of Gram negative bacterial meningitis with intrathecal/intraventricular polymyxins, in which intrathecal polymyxin B was used in almost all episodes prior to 1974 (Falagas et al., 2007). Polymyxin B was intrathecally/intraventricularly administered with a daily dose of 50,000 IU (i.e., 5 mg) for most adults, and a CMS daily dose of ≥62,500 IU (i.e., 5 mg) was commonly used. A wide range of 5000–120,000 IU (in two divided doses) of polymyxins (CMS and polymyxin B) was reported for pediatric patients (Falagas et al., 2007). Additional case reports have also supported the use of intrathecal/intraventricular polymyxin B at a daily dose of 50,000 IU (i.e., 5 mg) in adults for the treatment of meningoencephalitis and ventriculitis caused by A. baumannii and P. aeruginosa (Macedo et al., 2011; Guo et al., 2018), whereas a lower dose of 40,000 IU (i.e., 4 mg) on alternate days was effective in treating neonatal meningoventriculitis caused by MDR A. baumannii (Piparsania et al., 2012). Unfortunately, there is still a lack of information on the cerebrospinal fluid PK of polymyxin B in patients.
Overall, the current literature on the treatment of meningitis and ventriculitis caused by MDR Gram negative bacteria supports a daily dosage of intrathecal/intraventricular CMS at 125,000 IU (i.e., 10 mg or 4.1 mg CBA) or polymyxin B at 50,000 IU (i.e., 5 mg) with concomitant intravenous administration of polymyxins. Additionally, CMS appears in favor over polymyxin B because of more clinical experience. Nonetheless, more clinical data on the safety and PK/PD/TD of intrathecal/intraventricular polymyxins are in need to optimize their dosage recommendations (Tsuji et al., 2019).
E. Selective Decontamination of Mucosal Surface
Selective digestive tract decontamination (SDD) and selective oropharyngeal decontamination (SOD) are preventive measures to reduce the incidence of nosocomial infections and ultimately decrease the mortality rate, especially among intensive care unit (ICU) patients. Topical forms of polymyxins, mostly colistin sulfate, have been commonly used as part of SDD and SOD regimens. The SDD and SOD approaches with polymyxins have been demonstrated to reduce mortality (Kerver et al., 1988; Rocha et al., 1992; Tetteroo et al., 1993; Luiten et al., 1995; Sánchez García et al., 1998; de Jonge et al., 2003; de Smet et al., 2009b; Melsen et al., 2012) and decrease the incidence of infections (Cockerill et al., 1992; Camus et al., 2005), bacteremia (Oostdijk et al., 2011), VAP (Pugin et al., 1991; Bergmans et al., 2001; Pneumatikos et al., 2002; Koeman et al., 2006), pneumonia (Stoutenbeek et al., 1987; Godard et al., 1990; Rodríguez-Roldán et al., 1990; Abele-Horn et al., 1997), and pancreatic infections (Luiten et al., 1995). However, SDD and SOD might also be associated with a high rate of post-ICU incidence of hospital-acquired infections (de Smet et al., 2009a). For pediatric patients, SDD has been reported effective in reducing Gram negative infections (Smith et al., 1993) and urinary tract and respiratory infections (Ruza et al., 1998); however, in severely burned pediatric patients, SDD was found ineffective in decreasing bacterial colonization and incidence of infection (Barret et al., 2001). The regimens that have been reported consist of topical amphotericin B, tobramycin, polymyxins, gentamicin, vancomycin, neomycin, nystatin, and netilmicin, usually in triple combinations (Daneman et al., 2013). Among these topical agents, colistin, tobramycin, and amphotericin B have been frequently used (Daneman et al., 2013). Although previous studies reported insignificant association between the use of SDD and SOD with increased antimicrobial resistance, concerns remain in regions where a high prevalence of MDR bacteria has been detected (Krueger et al., 2002; Heininger et al., 2006; de Smet et al., 2011; Ochoa-Ardila et al., 2011). Worryingly, the emergence of polymyxin-resistant Gram negative bacteria has been reported with the use of colistin for selective decontamination (Halaby et al., 2013; Lübbert et al., 2013). The administration of SDD to ICU patients has also been demonstrated to affect the gut microbiome by reducing the abundance of Enterobacterales, anaerobic Gram positive, butyrate-producing Clostridium clusters IV and XIVa bacteria, while elevating the abundance of Bacteroidetes and enterococci; however, the impact of altered microbiome on the physiologic condition of patients is currently unknown and requires further investigations (Benus et al., 2010; Buelow et al., 2017).
F. Ocular and Otologic Infections
The topical form of polymyxins has long been used for the treatment of ocular infections. An early study in 1969 reported successful treatment of Pseudomonas ocular infections using ophthalmic colistin sulfate (1.2 mg CBA/ml; average dose of one to two drops every one-half to 1 hour) with satisfactory response of 89% and minimal adverse effects (Lund, 1969). In 1991, a study was performed to compare the efficacy of 1% tetracycline ointment, combination of 1% tetracycline ointment and 0.1% dexamethasone eye drop, and Eubetal ointment (tetracycline 0.5%, dexamethasone 0.1%, chloramphenicol 1%, and colistin) for the treatment of chlamydial conjunctivitis (Maichuk, 1991). Similar efficacy was reported for both tetracycline-dexamethasone combination and Eubetal ointment, and they were more effective than tetracycline alone (Maichuk, 1991). In another study, the combination of polymyxin B (10,000 IU/g) and bacitracin (500 U/g) ointment enhanced bacterial eradication during the treatment of bacterial conjunctivitis (Gigliotti et al., 1984). Polymyxin B-trimethoprim has been demonstrated to be effective for conjunctivitis, with comparable efficacy to chloramphenicol (Behrens-Baumann et al., 1988), moxifloxacin (Williams et al., 2013), and polymyxin B-neomycin-gramicidin (Genée et al., 1982). Similar to ophthalmic formulations, polymyxins are often used in combination with other antibiotic(s) and corticosteroid for otologic infection, with the combination of polymyxins, neomycin, and hydrocortisone being the typical formulation. Polymyxin B-neomycin-hydrocortisone has been shown effective for acute otitis externa with comparable efficacy to ciprofloxacin, but it is inferior to ciprofloxacin-dexamethasone (Rahman et al., 2007; Drehobl et al., 2008).
G. Bone and Joint Infections
Because of the poor penetration of colistin into bones after parenteral administration, studies in the 1970s and 1980s reported the use of colistin by incorporation into orthopedic cement (Rosenthal et al., 1976; Murray, 1984). Successful treatment and prevention of deep-wound infection with the use of colistin-erythromycin–containing cement has been demonstrated (Waterman et al., 2012). Although the release of CMS from cement beads was adequate for the inhibition of bacterial growth in vitro, data on the appropriate concentrations for clinical use are lacking (Waterman et al., 2012). The current clinical experience with the use of intravenous polymyxins for bone- and joint-associated infections is limited. A case report of total knee arthroplasty infected by MDR P. aeruginosa was successfully treated with surgical removal of prosthesis, colistin-loaded cement, and intravenous CMS (Papagelopoulos et al., 2007). An initial CMS dose of 3 MIU (i.e., 240 mg) every 8 hours was given for 7 days, and the dose was adjusted to 2 MIU (i.e., 160 mg) every 6 hours for 5 weeks with the intention to prevent renal impairment. During the course of intravenous CMS, the creatinine level decreased to 2.2 mg/dl, which fortunately reverted to the baseline after discontinuation of the treatment (Papagelopoulos et al., 2007). Two cases of fixation device–related orthopedic infections by MDR A. baumannii were successfully treated with intravenous CMS [1 MIU (i.e., 80 mg) bolus followed by 6 MIU (i.e., 480 mg) in a 24-hour continuous infusion for 36 days or 2 MIU (i.e., 160 mg) every 8 hours for 22 days], and no colistin-associated toxicity was observed (Kasiakou et al., 2005a). In a retrospective study involving 19 patients, prolonged intravenous CMS at 50,000 IU/kg per day (i.e., 4 mg/kg per day) was reported to be effective against bone and joint infections caused by difficult-to-treat Gram negative bacteria (i.e., P. aeruginosa and Enterobacter spp.); however, a higher dose of 75,000 IU/kg per day (i.e., 6 mg/kg per day) might be required for implant-associated infections (Valour et al., 2013). Another retrospective study involving 34 patients reported success in treating difficult-to-treat osteoarticular infections with appropriate surgical treatment (implant removal) using the combination of CMS [2.8–6 MIU/day (i.e., 224–480 mg/day)] and β-lactam (Ribera et al., 2015). Overall, limited clinical data support the treatment of joint and bone infections with polymyxins; however, the optimal dosage regimens should be investigated with well designed prospective clinical PK/PD/TD studies.
In summary, preclinical and clinical PK/PD/TD of polymyxins, particularly at the infection sites, require extensive investigations that will provide the pharmacological evidence to develop scientifically based dosage regimens for different types of infections in patients.
VIII. Polymyxin Toxicity
Dose-dependent nephrotoxicity is the most reported adverse event of intravenous polymyxins, with 30%–60% occurrence in patients (Holmes, 1964; Ryan et al., 1969; Brown et al., 1970; Koch-Weser et al., 1970; Price and Graham, 1970; Devlieger et al., 1977; Evans et al., 1999; Falagas and Kasiakou, 2005, 2006; Li et al., 2005a; Pastewski et al., 2008; Elias et al., 2010; Garonzik et al., 2011; Kvitko et al., 2011; Dezoti Fonseca et al., 2012; Kubin et al., 2012; Mingeot-Leclercq et al., 2012). In critically ill patients, acute kidney injury can lead to high mortality. Intravenous polymyxins can also cause neurotoxicity and skin hyperpigmentation in patients, and inhalation can lead to toxicities in the respiratory tract (Grill and Maganti, 2011; Li et al., 2020b).
A. Nephrotoxicity
Acute kidney injury may occur with intravenous polymyxins in patients (e.g., within a week); fortunately, it can revert after the discontinuation of treatment (Pogue and Tam, 2019). Key clinical characteristics of polymyxin-associated nephrotoxicity include renal tubular damage, decreased glomerular filtration rate, and creatinine clearance, as well as increased serum urea and creatinine concentrations (Falagas and Kasiakou, 2005). As reviewed in section V. Pharmacokinetics and Pharmacodynamics, very extensive tubular reabsorption of polymyxin B and colistin occurs in the kidney (Zavascki et al., 2008; Abdelraouf et al., 2012; Manchandani et al., 2016; Sivanesan et al., 2017b), which plays a critical role in their nephrotoxicity. Molecular imaging [e.g., immunostaining, mass spectrometry imaging, fluorescence microscopy, and X-ray fluorescence microscopy (XFM)] revealed significant accumulation of polymyxins in renal tubules (Azad et al., 2015c; Manchandani et al., 2015; Nilsson et al., 2015; Yun et al., 2015a,b; Vattimo et al., 2016; Velkov et al., 2016b). Preferential accumulation of polymyxins in mouse renal cortex was observed by matrix-assisted laser desorption/ionizing mass spectroscopy imaging after subcutaneous administration of polymyxins (Azad et al., 2015b). Similarly, a study with polymyxin-specific monoclonal antibody–based immunostaining revealed substantial accumulation of polymyxins in renal cortex, particularly in the renal proximal tubular cells in mice treated with polymyxin B, and very low accumulation was observed in the cells from distal tubules (Yun et al., 2015a). No major accumulation was detected in the other organs, such as lungs, liver, or heart, in mice (Azad et al., 2015b).
A novel dual-module fluorescent probe, FADDI-96, was developed based on the polymyxin structure-activity relationship model (Velkov et al., 2010). This probe possesses a dansyl group in the N-terminus and an iodine fluorophore at position 6 d-Phe of polymyxin B and exerts comparable antibacterial activity and renal epithelial cell toxicity as polymyxin B. It should be noted that the commercially available fluorescent polymyxin probes BODIPY-polymyxin B and dansyl-polymyxin B are devoid of these pharmacological properties, as the modification of Dab residues with the fluorophores damages the structure-activity relationship of polymyxins (Velkov et al., 2010, 2013; Azad et al., 2014, 2015c). A correlative microscopic study employed synchrotron-based XFM, fluorescence microscopy, and scanning electron microscopy to reveal the remarkable dose- and time-dependent accumulation (∼2000- to 4760-fold) of polymyxins (as FADDI-96) in rat and human kidney proximal tubular NRK-52E and HK-2 cells, respectively (Azad et al., 2015c). The current literature indicates that the uptake of polymyxins in renal tubular cells is transporter-mediated (Yousef et al., 2012; Azad et al., 2015c; Sivanesan et al., 2017a).
Megalin, a key endocytic receptor in renal tubules, plays an important role in the accumulation of polymyxins in renal tubular cells (Suzuki et al., 2013; Eshbach and Weisz, 2017; Hori et al., 2017; Manchandani et al., 2017). Binding of cytochrome C, a megalin substrate, to megalin was competitively inhibited by colistin (Suzuki et al., 2013). A decreased accumulation of colistin in rat kidneys and increased urinary excretion in megalin-shed rats indicate the important role of megalin in tubular reabsorption of colistin (Suzuki et al., 2013). Coadministration of cytochrome C, fragment of albumin, or succinylated bovine gelatin polypeptides with colistin decreased the accumulation of colistin in the kidneys and reduced tubular damage marker N-acetyl-β-d-glucosaminidase in urine, indicating the attenuation of polymyxin-induced tubular damage by decreasing the megalin-mediated uptake (Suzuki et al., 2013; Hori et al., 2017; Sivanesan et al., 2017a). Members of the organic anion transporters (Jacquemin et al., 1994; Abe et al., 1998) and organic cation transporters (Busch et al., 1996; Okuda et al., 1996) have been discovered in kidney tubular cells and effectively mediate the transport of a diverse range of anions and cations. An ex vivo study using isolated perfused rat kidney demonstrated substantial tubular reabsorption of colistin from the perfusate (approximately 90%), suggesting the carrier-mediated renal tubular reabsorption (Ma et al., 2009); furthermore, this reabsorption was significantly inhibited by tetraethylammonium (substrate of organic cation transporter OCTN1), hydrogen ions, and glycine-glycine (substrate of peptide transporters) (Shen et al., 1999; Ma et al., 2009). In a recent study, human peptide transporter 2 (PEPT2) was demonstrated to mediate the uptake of polymyxins in HEK-293 cells (Lu et al., 2016). The human PEPT2-specific substrate [3H]glycylsarcosine was used, and the IC50 values of colistin and polymyxin B were 11.4 ± 3.1 µM and 18.3 ± 4.2 µM, respectively. Similar results were observed with [3H]polymyxin B1 and a fluorescent polymyxin probe MIPS-9541 when determining the PEPT2-mediated uptake of polymyxins in HEK-293 cells (Lu et al., 2016).
Information on the intracellular localization of polymyxins in renal tubular cells is limited. A confocal fluorescence imaging study with regioselectively labeled monodansylated polymyxin B probes (MIPS-9543 and MIPS-9544) revealed partial colocalization of polymyxins within the endoplasmic reticulum and mitochondria in rat NRK-52E cells (Yun et al., 2015b). High-resolution XFM imaging indicated preferential accumulation of polymyxins around the nuclear region compared with the cytoplasmic areas in rat NRK-52E and human HK-2 kidney proximal tubular cells (Azad et al., 2015c). Overall, deciphering the mechanisms of intracellular uptake and disposition of polymyxins in kidney tubular cells is critical for the optimization of their parenteral use, amelioration of nephrotoxicity, and the discovery of new-generation, safer polymyxins.
The present literature highlights the key role of DNA damage, metabolic and inflammatory perturbations, oxidative stress, cell cycle arrest, apoptosis, and autophagy in polymyxin-induced death of renal tubular cells in vitro and in vivo. A recent study showed the formation of micronuclei and multilobed nuclei, chromosome mis-segregation, and genome instability in HK-2 cells and mouse kidneys after polymyxin B treatment (Yun et al., 2018). Increased expression of kidney γH2AX (phosphorylated H2A histone family member X) suggested persistent DNA damage in kidneys by polymyxins. A transient cell cycle arrest followed by continuation with unrepaired DNA damage was speculated as a result of checkpoint perturbations by polymyxin B treatment (Yun et al., 2018). Intraperitoneal administration of colistin (16 mg/kg per day in two divided doses) caused the upregulation of CCNB1 and CDC2 genes in mouse kidneys, suggesting cell cycle arrest at the G2/M phase (Eadon et al., 2013). Furthermore, the translocation of cyclin B1 to the nucleus is consistent with the observed cell cycle arrest at the G2/M phase, and the upregulation of galectin-3 gene also highlighted the G1/S and G2/M as the points of arrest in the cell cycle (Eadon et al., 2013). A 3-day colistin treatment resulted in increased expression of proliferating cell nuclear antigen, which suggested the inhibition of DNA replication at S phase and kidney injury (Eadon et al., 2013). In addition, the activation of p53 and galectin-3 by colistin indicated the progression of the cell to the apoptotic pathways with unrepairable cellular damage (Sutton et al., 2013; Chen and Kuo, 2016).
Several studies revealed that polymyxins induced renal tubular apoptosis both in vitro and in vivo (Ozyilmaz et al., 2011; Yousef et al., 2011, 2012). Colistin treatment (a cumulative dose of 20.5 mg/kg over 5 days) caused an increase of terminal dUTP nick end labeling (TUNEL)-positive nuclei in rat kidneys and fragmentation of DNA, a biochemical signature of apoptosis (Yousef et al., 2012). Similar results were observed in rat and human proximal tubular cells treated with polymyxin B (Azad et al., 2013; Yun et al., 2018). Intravenous administration of colistin in mice (7.5 or 15 mg/kg per day in two doses for 7 days) caused a significant increase of cytochrome C, apoptosis-inducing factor, cleaved caspase-9, and cleaved caspase-3 with a simultaneous decrease of B-cell lymphoma (Bcl-2) (Dai et al., 2014). Furthermore, a significant increase in the expression of Fas, FasL, and Fas-associated death domain (FADD) and the cleavage of caspase-8 was observed. These results indicated the involvement of both death receptor and mitochondrial apoptotic pathways in polymyxin-induced apoptosis, and the increased expression of truncated Bax-inhibiting peptide (tBid) suggested the crosstalk between both cell death pathways. In addition, the endoplasmic reticulum pathway was also involved in colistin-induced apoptosis, as significant upregulation of glucose-regulated protein 78 kDa/Bax-inhibiting peptide (Grp78/Bip), cleaved activating transcription factor 6 (ATF6), DNA damage inducible gene 153/C/EBP-homologous protein (GADD153/CHOP), and caspase-12 was evident in colistin-treated mice (Dai et al., 2014).
In vitro cell culture studies using rat NRK-52E and human HK-2 kidney tubular cells demonstrated both concentration- and time-dependent apoptosis after the treatment with polymyxins (Azad et al., 2013, 2015c). NRK-52E cells showed a higher resistance to polymyxin B–induced cytotoxicity in comparison with HK-2 cells. In NRK-52 cells, polymyxin B treatment caused the concentration-dependent activation of caspase-3, -8, and -9; DNA damage; and translocation of membrane phosphatidylserine (Azad et al., 2013, 2015a). Furthermore, in NRK-52E cells, polymyxin B also caused concentration- and time-dependent mitochondrial damage, including mitochondrial morphology changes from filamentous to fragmented, loss of mitochondrial membrane potential, and generation of ROS (Azad et al., 2015a). A recent metabolomics study with NRK-52E cells treated with polymyxin B showed depletion of cellular metabolites taurine and hypotaurine, suggesting the oxidative stress induced by polymyxins (Azad et al., 2015b). Using transcriptomics, perturbations in both inflammatory and metabolic pathways were observed in polymyxin-induced damage in NRK-52E cells, as evidenced by the key differentially expressed genes Cxcl10, Cxcl1, Ccl2, Ccl20, Tnf, Tpi1, Pfkl, Ldh-A, and Gapdh (Azad et al., 2018).
There is limited information in the literature on the chemical biology of polymyxin-induced nephrotoxicity. The toxic effect of major components of polymyxin B (polymyxin B1 and polymyxin B2) and colistin (colistin A and colistin B) (Table 1) was assessed in vitro and in vivo (Roberts et al., 2015). The mild to moderate histologic damage observed in the mouse kidney treated with all four major components of polymyxins is comparable. However, polymyxin B1 and colistin A induced a >3-fold higher apoptotic effect on HK-2 cells than polymyxin B2 and colistin B. As there is only one methylene (-CH2-) group difference in the N-terminal fatty acyl group of polymyxin B1 and B2 and colistin A and B (Table 1), the hydrophobicity of this N-terminal fatty acyl group is critical in polymyxin-induced apoptosis. Furthermore, >2-fold differences in EC50 of polymyxin B and colistin indicate the importance of hydrophobicity at position 6 for polymyxin toxicity in renal tubular cells, considering that the only difference between polymyxin B and colistin is at position 6 (i.e., d-Phe vs. d-Leu) (Roberts et al., 2015; Azad et al., 2018). A recent molecular dynamic simulation study revealed the structure-interaction relationship of polymyxins with the membrane of human kidney proximal tubular cells at the atomic level and highlighted that the hydrophobicity at positions 6/7 and stereochemistry at position 3 regulated the interactions of polymyxins with the cell membrane (Jiang et al., 2020c).
Overall, recent studies on the mechanism of polymyxin-induced nephrotoxicity indicate a complex interplay of multiple biochemical processes in renal tubular cells (Fig. 8) (Dai et al., 2014), and further studies using systems pharmacology and chemical biology are warranted to decipher the kinetics of this interplay.
Fig. 8.
The proposed mechanisms of polymyxin-induced apoptotic cell death in renal tubular cells. AIF, apoptosis-induced factor; CAT, catalase; CDK2, cyclin-dependent kinase 2; cytC, cytochrome C; MAPK, mitogen-activated protein kinase; MDA, malondialdehyde; MR, endocytic receptor megalin; OCTN1, organic cation transporter 1; SOD, superoxide dismutase; tBid, truncated Bax-inhibiting peptide . Reproduced with permission (Dai et al., 2014). Copyright © American Society for Microbiology.
To alleviate polymyxin-associated nephrotoxicity in patients, the importance of optimization of the polymyxin dose, therapeutic drug monitoring, electrolyte balance, coadministration of nephrotoxic drugs, and early detection of kidney injury is paramount (Hartzell et al., 2009; Zavascki and Nation, 2017). Furthermore, several other approaches have been investigated to attenuate polymyxin-induced nephrotoxicity, including inhibiting the accumulation in renal tubular cells, coadministration of antioxidants to counteract polymyxin-induced oxidative stress, and structural modification of the polymyxins (Ma et al., 2009; Velkov et al., 2010; Ozyilmaz et al., 2011; Yousef et al., 2011, 2012; Ozkan et al., 2013; Azad et al., 2017a; Sivanesan et al., 2017a). A number of animal studies investigated the protective effect of antioxidants such as N-acetylcysteine, ascorbic acid, melatonin, grape seed proanthocyanidin extract, polyaspartic acid, and methionine on polymyxin-induced nephrotoxicity (Ozkan et al., 2013; Ozyilmaz et al., 2011; Yousef et al., 2011, 2012; Azad et al., 2017a,b). Methionine (100 or 400 mg/kg) attenuated polymyxin-induced kidney damage in mice (polymyxin B 35 mg/kg, twice daily over 3.5 days) and also significantly reduced mitochondrial oxidative stress in NRK-52E cells treated with polymyxin B (Azad et al., 2017a). Importantly, methionine did not alter the PK of polymyxin B in rats (Azad et al., 2017a). Similar protective activity by grape seed proanthocyanidin extract was observed in rats (Ozkan et al., 2013). Interestingly, coadministration of succinylated bovine gelatin polypeptides (Gelofusine) did not alter the pharmacokinetics of colistin in rats but decreased the accumulation of polymyxins in renal tissue and nephrotoxicity (Sivanesan et al., 2017a).
Information on the protection of polymyxin-associated nephrotoxicity in patients is scarce. A preliminary randomized controlled study with only 28 patients did not show the nephroprotective effect with ascorbic acid (2 g every 12 hours, intravenously) in patients treated with intravenous CMS (Sirijatuphat et al., 2015). No difference in the PK of formed colistin in plasma was observed in both groups. The low number of patients, administered doses of ascorbic acid, and the lack of resemblance of animal models to patients could account for the absence of protective effect from ascorbic acid against polymyxin-associated nephrotoxicity in patients (Sirijatuphat et al., 2015). In contrast, a prospective, observational cohort study revealed strong independent renal protection by intravenous ascorbic acid [3 (2–4) g every 12 hours] in critically ill patients treated with CMS (Dalfino et al., 2015). In the group without acute kidney injury, 76.9% (30 of 39) patients received concurrent ascorbic acid, whereas in the patients with acute kidney injury, the value was 41.9% (13 of 31); higher risk of acute kidney injury and greater probability of earlier onset of acute kidney injury were identified in the group without ascorbic acid as the adjuvant to CMS therapy (Dalfino et al., 2015). Several potential limitations were demonstrated in this study, including the small sample size, absence of randomization, and no characterization of patients between the groups (Dalfino et al., 2015). Nevertheless, well designed clinical studies are required to develop novel approaches to attenuate polymyxin-induced nephrotoxicity.
Detecting the early onset of acute kidney injury after intravenous administration of polymyxins is critical to alleviate polymyxin-induced nephrotoxicity in patients. Importantly, early biomarkers have been investigated in animal studies to predict polymyxin-induced kidney injury. Recently, a urinary proteomics study with colistin-treated mice identified multiple proteins, such as complements (C3 and CB-4), carbonic anhydrase 3, hexokinase, and kininogen-1, as the potential biomarkers for early detection of polymyxin-induced acute kidney injury (Sivanesan et al., 2017c). Furthermore, a correlation between polymyxin-induced kidney injury and urinary levels of kidney injury molecule-1 (KIM-1), neutrophil gelatinase-associated lipocalin, and α-glutathione S-transferase in rodents was demonstrated (Burt et al., 2014; Keirstead et al., 2014). Normalized urinary KIM-1 (ratio between urinary KIM-1 and creatinine) in patients appeared more sensitive for early prediction of acute kidney injury after polymyxin B treatments (25,000 U/kg loading dose and 12,500–15,000 U/kg per 12 hours) as compared with the level of serum creatinine (Babic et al., 2017). Comprehensive clinical validations are warranted to minimize polymyxin-induced nephrotoxicity in patients.
Overall, it is evident that polymyxin-induced nephrotoxicity involves substantial accumulation in renal tubular cells, oxidative stresses, induction of DNA damage, and apoptosis. Further systems and molecular investigations are required to elucidate the precise mechanisms of polymyxin-induced nephrotoxicity, which will not only facilitate the optimization of their clinical use but also expediate the discovery of next-generation polymyxins with better safety and efficacy.
B. Neurotoxicity
In old literature, neurotoxicity was also often associated with the use of polymyxins in patients (Koch-Weser et al., 1970). The incidence rate of neurotoxicity associated with polymyxins in patients is approximately 7% or less and substantially lower than that of nephrotoxicity (Bosso et al., 1991; Falagas and Kasiakou, 2006). The clinical features of polymyxin-induced neurotoxicity are often minor and include paresthesia, apnea, nausea and vomiting, neuropathy, myopathy, dizziness, psychosis, and seizures (Koch-Weser et al., 1970; Falagas and Kasiakou, 2006). One of the major risk factors associated with neurotoxicity is the extended exposure (duration and concentration) to polymyxins (Wolinsky and Hines, 1962; Falagas and Kasiakou, 2006). Other risk factors include the presence of medical conditions such as myasthenia gravis, renal impairment, and hypoxia; concomitant use of other medications (e.g., sedatives, anesthetics, narcotics, and muscle relaxants); and gender (Koch-Weser et al., 1970; Decker and Fincham, 1971). The onset of neurotoxicity usually occurs shortly after intravenous infusion (e.g., transient paresthesia and apnea) or within the first few days of polymyxin therapy (Koch-Weser et al., 1970; Decker and Fincham, 1971; Falagas and Kasiakou, 2006). Polymyxin-induced neurotoxicity is generally reversible with discontinuation of the therapy (Falagas and Kasiakou, 2006; Dalfino et al., 2012) or via a longer infusion duration. The exact mechanism of polymyxin-induced neurotoxicity is not clear, and the literature suggests that it could involve the inhibition of acetylcholine, acetylcholine release, prolonged depolarization, and calcium depletion (Falagas and Kasiakou, 2006).
Polymyxins can accumulate in neuron cells, generate mitochondrial ROS, and induce oxidative stress (Jin et al., 2009, 2011, 2012, 2013; Dai et al. 2016, 2018a,b, 2019; Wang et al., 2016). Similar to polymyxin-induced nephrotoxicity and pulmonary toxicity (Ahmed et al., 2017), polymyxin-induced neurotoxicity also involves autophagy. Both in vitro and in vivo results demonstrate the induction of substantial autophagy in neuron cells during polymyxin treatment as a protective mechanism against apoptosis (Dai et al., 2016, 2019). Polymyxin-induced autophagy is regulated by a complex signal network among the phosphatidylinositol-3-kinase/Akt, mitogen-activated protein kinase, nuclear factor-κB, p53, mammalian target of rapamycin, and nuclear factor erythroid 2-related factor 2/heme oxygenase-1 pathways (Dai et al., 2013, 2016, 2018a,b; Barcelos et al., 2016). Repurposing of clinically available drugs and food supplements such as minocycline, rapamycin, salidroside, and curcumin has shown to be protective against polymyxin-induced neurotoxicity in vitro and in animals (Dai et al., 2013, 2018a,b; Barcelos et al., 2016; Lu et al., 2017). Clinical evidence is required to develop novel combination therapies of polymyxins with neuroprotective agents to ameliorate the neurotoxicity (Dai et al., 2019). Additionally, several other approaches could potentially minimize polymyxin-induced neurotoxicity in patients, including early detection of toxicity and discontinuation of therapy, use of therapeutic drug monitoring, and employment of specific biomarkers (Jin et al., 2009, 2011; Dalfino et al., 2012; Justo and Bosso, 2015).
C. Skin Hyperpigmentation
Recent case reports highlight skin hyperpigmentation after treatments with intravenous polymyxin B in patients (Mattos et al., 2016, 2017; Zheng et al., 2018; Li et al., 2020b); however, no such reports have been published on CMS/colistin to date. Polymyxin B–induced skin hyperpigmentation has been reported in approximately 8%–15% of the subjects (Mattos et al., 2016, 2017; Zheng et al., 2018; Li et al., 2020b). The onset of skin hyperpigmentation can be noticed as early as 3 to 4 days after the initiation of polymyxin B treatment with the appearance of red rashes that gradually darken as the treatment progresses (Lahiry et al., 2017). For patients suffering from hyperpigmentation, skin color can be darkened to various shades of brown, particularly in the neck, face, and upper part of the body (Mattos et al., 2016); these symptoms have also been reported on the skin of lower body parts and limbs (Zheng et al., 2018). To date, polymyxin B–induced hyperpigmentation has been observed in patients of various age groups, ranging from neonates to adults (Gothwal et al., 2016; Zheng et al., 2018).
The mechanism of polymyxin B–induced skin pigmentation is not clear. Several possible mechanisms have been proposed, including 1) the production and accumulation of melanin within cutaneous cells, 2) accumulation of polymyxin B within the extracellular matrix, 3) synthesis of pigments such as lipofuscin, 4) deposition of iron and red blood cells from damaged dermal vessels, 5) extensive inflammation due to the release of histamine by polymyxin B, 6) increased proliferation of epidermal Langerhans cells and dermal dendritic cells, and 7) low expression of interleukin-6 after polymyxin B treatment (Miori et al., 1990; Voitenko et al., 1990; Fechner et al., 1993; Marone et al., 2003; Choi et al., 2012; Zavascki et al., 2015, 2016; Mattos et al., 2016, 2017). Decreasing the dose or discontinuation of the treatment allows gradual restoration of skin color to normal state (Zavascki et al., 2015; Zavascki et al., 2016). Clearly, position 6 d-Phe plays an important role in polymyxin B–induced skin pigmentation, as this is the only difference in the structure from colistin (i.e., d-Leu at position 6).
D. Pulmonary Toxicity
Inhalation of polymyxins has been widely used in people with cystic fibrosis for several decades and appears to be a safe and effective alternative to intravenous administration for the treatment of lung infections caused by MDR Gram negative pathogens (section V. Pharmacokinetics and Pharmacodynamics) (Min et al., 2018). However, adverse events can occur in approximately 10%–20% patients treated with inhaled polymyxins, and the common symptoms include cough, throat irritation, bronchospasm, bronchoconstriction, and abnormal taste (Marschke and Sarauw, 1971; Le Brun et al., 2002; Alothman et al., 2005; Falagas and Kasiakou, 2006; McCoy, 2007; Pereira et al., 2007; Wunsch et al., 2012; Velkov et al., 2015; Min et al., 2018; Biagi et al., 2019). A recent study reported the occurrence of pulmonary eosinophilia after inhalation of 75 mg CMS in an 83-year-old woman with a history of chronic bronchiectasis (Lépine et al., 2017). Hypersensitivity pneumonitis and eosinophilia were reported in a 69-year-old woman after treatment with a high dose of CMS [2 MIU (i.e., 160 mg) every 8 hours] (Leong et al., 2010). In another case study, a 29-year-old woman with cystic fibrosis and acute airway infection with P. aeruginosa developed acute respiratory distress syndrome after inhalation of 75 mg CMS twice daily (McCoy, 2007). In patients with cystic fibrosis (children and adults), inhaled CMS could induce chest tightness and a transient decrease in the respiratory function (Dodd et al., 1997; Cunningham et al., 2001; Alothman et al., 2005). Bronchoconstriction and decreased respiratory function are more commonly associated with inhalation of colistin sulfate than CMS (Westerman et al., 2004). Because of the potential conversion of CMS to colistin in vitro and sterility consideration, reconstituted CMS solution should be used within 24 hours to reduce any potential pulmonary toxicity, as recommended by the FDA and product information (Biagi et al., 2019). Prior to the use of inhaled polymyxins in patients, clinicians should consider the risk factors associated with pulmonary toxicity, such as the form of polymyxins (CMS or polymyxin B) administered, stability of the product, delivery techniques (nebulization or dry powder inhalation), and the presence of comorbidities.
The mechanism of polymyxin-induced pulmonary toxicity in patients is unknown. It is believed to be associated with local irritation of airways and histamine release (Marschke and Sarauw, 1971). In old literature, in vitro degranulation of mast cells and histamine release were reported in the presence of polymyxin B and colistin (Jasani et al., 1979). Recent studies evaluated the toxicity of polymyxins in human lung epithelial A549 cells and revealed the concentration- and time-dependent mitochondrial damage, such as the loss of mitochondrial membrane potential, mitochondrial fragmentation, generation of mitochondrial ROS, and activation of apoptotic pathways, by polymyxins at concentrations >2,000 mg/l (Ahmed et al., 2017). Colocalization of polymyxins with mitochondria supports its critical role in polymyxin-induced toxicities in lung epithelial cells (Azad et al., 2015c; Ahmed et al., 2019). Recent mouse studies showed that the ELF concentration after aerosolized polymyxins reached 607 mg/l and induced only minor lung inflammation (Lin et al., 2017a,c). In a recent PK study with 10 critically ill patients, the mean ELF concentrations of formed colistin were up to ∼300 mg/l after inhalation of 5 MIU (i.e., 400 mg) CMS, and no major adverse effect was observed (Gkoufa et al., 2019). In addition, a recent transcriptomics study revealed that polymyxin B significantly increased the expression of CD86 gene (encoding a costimulator of T- and B-lymphocyte function) and caused perturbations of metabolism, DNA repair and replication, and cell cycle in A549 cells (Azad et al., 2019).
Overall, the toxicity of polymyxins is a critical factor that needs to be considered for the optimization of their clinical use. Current clinical literature on polymyxin-induced toxicities has several limitations, including the lack of proper control groups, the presence of comorbidities, and coadministration of other toxic drugs. To maintain the clinical utility of this last-line class of antibiotics, several strategies have been proposed to minimize polymyxin-induced toxicities, such as therapeutic drug monitoring, personalized dosing with adaptive feedback control (Lakota et al., 2018), and avoidance of simultaneous administration of toxic medications.
IX. Polymyxin Combination Therapy
Considering potentially suboptimal PK/PD/TD, emergence of resistance, and narrow therapeutic windows, combination therapy is recommended for polymyxins even though there is a lack of strong clinical evidence (Tan et al., 2007; Bergen et al., 2008; Garonzik et al., 2011; Sandri et al., 2013a; Cheah et al., 2016; Nation et al., 2017; Tsuji et al., 2019). PK/PD principles have been largely overlooked in the current clinical literature on antibiotic combinations; in particular, drug exposure at the infection site is seldom considered. Another key factor that should be considered for evaluations of antibiotic combinations is whether synergy (e.g., >2-log10 killing than the more active monotherapy) or additive killing is acceptable given that decreased bacterial load can lead to lower potential of resistance development. Because of the different PK between CMS and colistin/polymyxin B and their poor disposition in the lungs after intravenous administration, PK/PD at the infection site should be considered when optimizing polymyxin combination therapies for pneumonia and other type of infections.
A. Combinations of Polymyxins and Carbapenems
Carbapenems have been widely used in combination therapy with polymyxins against MDR Gram negative bacteria. The combination of colistin or polymyxin B (concentrations at 2 × MIC) with doripenem (6 mg/l) resulted in synergistic killing against 4 polymyxin-susceptible KPC-3–producing K. pneumoniae (Lee and Burgess, 2013). An interesting study was performed to evaluate the efficacy of doripenem-colistin combination against KPC-producing K. pneumoniae based on the molecular resistance mechanisms (Clancy et al., 2013). An association was observed between higher doripenem MICs and reduced efficacy of colistin-doripenem with ompK36 (a gene encoding for the outer membraneporin OmpK36) mutations (Clancy et al., 2013). It was suggested that ompK36 genotyping could be used as an indicator for the efficacy of doripenem or colistin-doripenem treatment and facilitate the selection of therapeutic options (Clancy et al., 2013). Time-kill assays with clinically achievable concentrations of colistin (2 mg/l), doripenem (8 mg/l), and tigecycline (2 mg/l) also supported the use of colistin-doripenem combination against MDR and XDR A. baumannii, with greatest synergistic (53.6%) and bactericidal (75.4%) activities among those antibiotics tested in mono and double combinations (Park et al., 2016). The addition of colistin (0.12–16 mg/l) to doripenem (0.06–32 mg/l) demonstrated superior synergistic activity in comparison with the combination of doripenem with levofloxacin or amikacin against 25 A. baumannii isolates (Pankuch et al., 2010). The synergistic activity of colistin-doripenem combination was illustrated in an IVM study against MDR K. pneumoniae using clinically relevant concentrations of colistin (constant concentrations of 0.5 and 2 mg/l) and doripenem (Cmax of 2.5 and 25 mg/l over 8 hours) (Deris et al., 2012). Using the same combination regimens, increased bacterial killing activity was observed against colistin-heteroresistant and colistin-resistant MDR P. aeruginosa, whereas only the combinations consisting of colistin at 2 mg/l resulted in increased activity against colistin-resistant strains (Bergen et al., 2011b). Importantly, emergence of colistin-resistant subpopulations could be prevented with colistin-doripenem combination (Bergen et al., 2011b; Deris et al., 2012). The combination of colistin (constant concentrations of 2 or 5 mg/l) and doripenem (Cmax of 25 mg/l over 8 hours) has been shown to eliminate colistin-resistant subpopulations in addition to the synergistic killing activity using HFIM (Ly et al., 2015). The synergy of colistin (constant concentration of 3.5 mg/l) and doripenem (Cmax of 25 mg/l over 8 hours) combination was further extended to biofilm-associated infections using a dynamic biofilm model, and enhanced killing and minimized colistin emergence were observed against colistin-susceptible P. aeruginosa strains (Lora-Tamayo et al., 2014).
It is important to note that the combination of colistin (1.2, 2.5, or 4.2 mg/l) and meropenem (6.8 mg/l) was reported to exhibit limited efficacy against high-level carbapenem-resistant NDM-producing K. pneumoniae (meropenem MIC ≥16 mg/l) in vitro (Lagerbäck et al., 2016). In contrast, an IVM study demonstrated that the combination of meropenem (1 g in 3-hour infusion) and colistin (constant concentration of 1 mg/l) resulted in bactericidal activity against carbapenem-resistant A. baumannii (meropenem MIC of 128 mg/l), which was otherwise unachievable with monotherapies (Liu et al., 2016a). In another IVM study, enhanced bacterial killing was also reported with the combination of colistin (initial concentration of 0.5 mg/l; half-life of 4 hours) and meropenem (initial concentration of 40 mg/l; half-life of 1 hour) against P. aeruginosa and A. baumannii (Tängdén et al., 2017). The addition of imipenem (1–2 × MIC; 8–16 mg/l) to colistin (1–2 × MIC; 2–4 mg/l) was found to be synergistic and bactericidal against beta-lactamase–producing K. pneumoniae (Tang et al., 2015). However, the bactericidal effect was inoculum-dependent at the lower concentration of colistin-imipenem (Tang et al., 2015). Against 42 K. pneumoniae isolates, the combination of colistin sulfate (5 mg/l or 4 × MIC) and imipenem (10 mg/l or 4 × MIC) was synergistic against 50% of colistin-susceptible strains but antagonistic against 56% of colistin-resistant strains (Souli et al., 2009). This demonstrated that the colistin susceptibility should be considered and that colistin-imipenem combination may not be synergistic against colistin-resistant VIM-producing K. pneumoniae (Souli et al., 2009). Nonetheless, clinically relevant concentrations of colistin in combination with imipenem showed promising killing activity against MDR P. aeruginosa that were resistant to both colistin and imipenem (Bergen et al., 2011a). In addition to PK/PD considerations, the inconsistent in vitro results in the literature suggest that the effect of different mechanisms of carbapenem resistance on the synergy of polymyxin-carbapenem combinations may need further investigation.
Using a mouse sepsis model, the combination of colistin (1 mg/kg) and imipenem (20 mg/kg) resulted in a lower lethality rate (10%–15%) as compared with each monotherapy (colistin, 30%–40%; imipenem, 30%–80%) (Cirioni et al., 2007). In addition, the combination of intranasal CMS (10 mg/kg per day) and subcutaneous injection of imipenem (60 mg/kg per day) improved the survival rate in a mouse pneumonia model with MDR P. aeruginosa (Aoki et al., 2009). Some clinical data also support the use of polymyxin-carbapenem combination for the treatment of bacteremia caused by KPC-producing K. pneumoniae and carbapenem-resistant A. baumannii, with lower mortality rates observed for patients treated with polymyxin-carbapenem as compared with polymyxins alone (K. pneumoniae, 57.1% vs. 20%; A. baumannii, 47.5% vs. 25.8%) (Qureshi et al., 2012; Park et al., 2019).
A recent open-label, randomized, controlled trial evaluated colistin monotherapy versus colistin-meropenem combination therapy for the treatment of severe infections caused by carbapenem-resistant Gram negative bacteria (Paul et al., 2018). The results showed that, compared with colistin monotherapy, the combination therapy was not superior and did not improve clinical efficacy in severe A. baumannii infections (Paul et al., 2018). It should be noted that 99% and 98% of the pathogens in the combination group and colistin group, respectively, were meropenem-resistant, with MICs >8 mg/l (Paul et al., 2018). It would be valuable to examine the synergy of this combination against the pathogens even in vitro. In the same study, ∼50% of patients in each group had pneumonia for which intravenous polymyxins are not efficacious (section V. Pharmacokinetics and Pharmacodynamics); therefore, in this clinical study the colistin-meropenem combination is virtually equivalent to meropenem monotherapy against meropenem-resistant pathogens at the lung infection site (Paul et al., 2018). Overall, the conclusion can be specifically stated that colistin-meropenem combination therapy was not superior than colistin monotherapy for the treatment of pneumonia caused by meropenem-resistant A. baumannii (Paul et al., 2018). Nevertheless, well designed prospective clinical studies are urgently required to evaluate the clinical benefit of polymyxin combination therapy for the treatment of different types of infections.
B. Combinations of Polymyxins and Rifampicin
One of the most commonly studied antibiotics for polymyxin combination therapy is rifampicin. The in vitro synergistic effect of polymyxins and rifampicin has been reported against NDM-producing, KPC-producing K. pneumoniae, including polymyxin-resistant strains (Elemam et al., 2010; Tascini et al., 2013; Gaibani et al., 2014). In vitro time-kill kinetic studies showed that the combinations of colistin (1.2–4.2 mg/l) and rifampicin (1.7–2 mg/l) at clinically relevant concentrations were bacteriostatic or bactericidal against NDM-producing, KPC-producing K. pneumoniae (Gaibani et al., 2014; Lagerbäck et al., 2016). Against carbapenem-resistant Acinetobacter spp., polymyxin B-rifampicin combination exhibited bactericidal activity against 13 of 31 (polymyxin B 2 mg/l and rifampicin 2 mg/l) and two of three isolates (polymyxin B at concentration of 0.5 × MIC and rifampicin 2 mg/l) in two time-kill kinetic studies (Lim et al., 2009, 2011). In an IVM study, two MDR A. baumannii isolates were treated with three combination regimens of colistin (constant concentrations of 0.5, 2, and 5 mg/l) and rifampicin (Cmax of 5 mg/l) (Lee et al., 2013a). In comparison with each monotherapy, all combination regimens resulted in greater killing of A. baumannii at a low inoculum (∼106 cfu/ml), whereas only regimens containing colistin at 2 and 5 mg/l increased the killing activity against A. baumannii at a high inoculum (∼108 cfu/ml) (Lee et al., 2013a). Importantly, colistin-rifampicin combination was effective in suppressing the emergence of colistin resistance (Lee et al., 2013a). The in vivo efficacy of colistin (3 mg/kg intramuscularly) in addition to rifampicin (5 mg/kg intravenously) was demonstrated in a rat thigh infection caused by MDR A. baumannii (Pantopoulou et al., 2007). Colistin monotherapy and colistin-rifampicin resulted in a better median survival (4 days) in comparison with the untreated control (2 days) and rifampicin monotherapy (2.5 days) (Pantopoulou et al., 2007). Colistin-rifampicin combination was superior, with a lower 6-day mortality rate (70%) as compared with colistin monotherapy (100%) (Pantopoulou et al., 2007). In a mouse pneumonia model with MDR P. aeruginosa, the efficacy of CMS (10 mg/kg subcutaneously or 5 mg/kg per 12 hours intranasally) in combination with imipenem (30 mg/kg per 12 hours subcutaneously) or rifampicin (25 mg/kg per day orally) was examined (Aoki et al., 2009). Overall, an increased survival rate was observed for mice receiving intranasal CMS in combination with both imipenem (62.5%) and rifampicin (75%) (Aoki et al., 2009). It is important to note that a much greater survivability was observed after the combination therapy with intranasal CMS as compared with those with subcutaneous CMS because of higher drug exposure in the lungs via intranasal administration (Aoki et al., 2009). In addition, use of CMS (rather than colistin) and the lower subcutaneous dose (considering animal scaling) might also contribute to the lower survival rates with CMS alone and the combination via subcutaneous administration (Aoki et al., 2009). The efficacy of polymyxin-rifampicin combination is further extended against S. maltophilia, for which bactericidal effect was observed only with the combination of colistin (2 mg/l) and rifampicin (8 mg/l) (Betts et al., 2014b). The same study also demonstrated an increased survival of Galleria mellonella with colistin-rifampicin combination for the treatment of S. maltophilia infection in comparison with each monotherapy (Betts et al., 2014b). More importantly, clinical evidence on the efficacy of polymyxin-rifampicin combination has been reported against difficult-to-treat infections (meningitis, sepsis, pneumonia, and UTI) caused by P. aeruginosa [intravenous CMS 2 MIU/day (i.e., 160 mg/day) and intravenous rifampicin 600 mg/day; with additional aerosolized CMS at 1 MIU (i.e., 80 mg) twice daily for pneumonia], meningitis caused by A. baumannii (intrathecal CMS 10 mg and intravenous rifampicin 600 mg), and bacteremia and UTI caused by K. pneumoniae (intravenous CMS 100 mg/8 h and intravenous rifampicin 600 mg) (Tascini et al., 2004; Nastro et al., 2014). Large clinical studies are required to confirm the clinical benefit of polymyxin-rifampicin combination.
C. Combinations of Polymyxins and Tigecycline
Infections caused by carbapenem-resistant Gram negative bacteria (e.g., A. baumannii and Enterobacterales) often limit the therapeutic options to polymyxins and tigecycline. However, the use of these last-line antibiotics has been a concern due to the emergence of resistance, and studies have been undertaken to examine the combination of polymyxins and tigecycline. Against 15 carbapenem-resistant Enterobacterales (E. coli, K. pneumoniae, and Enterobacter spp.), a synergistic effect was demonstrated against 47% of the strains, and no antagonism was detected (Betts et al., 2014a). In contrast, a study reported that colistin-tigecycline combination did not enhance the bacterial killing against NDM-1–producing Enterobacterales; however, it should be noted that the colistin concentrations (0.1–0.29 mg/l) used in this study were extremely low (Albur et al., 2012). Colistin-tigecycline combination is promising in treating ESBL-producing K. pneumoniae infections, with a high rate of synergy (89%) at sub-MIC concentrations (0.25 × MIC) (Ku et al., 2017), and exhibits synergistic killing against colistin-susceptible (treated with colistin and tigecycline at 1, 2, and 4 × MIC) and colistin-resistant (treated with colistin 2 mg/l and tigecycline 1 mg/l) KPC-producing K. pneumoniae (Pournaras et al., 2011; Betts et al., 2014b). In another study, the combination of colistin (2 mg/l) and tigecycline (1 mg/l) was less effective than the combination of colistin (2 mg/l) and rifampicin (8 mg/l) against polymyxin-resistant S. maltophilia (MICs: 4–32 mg/l for colistin, 4–16 mg/l for rifampicin, and 0.5–8 mg/l for tigecycline) (Betts et al., 2014b). The combination of colistin-tigecycline also displayed synergistic killing against MDR A. baumannii (Moland et al., 2008; Sheng et al., 2011), as well as in an IVM study mimicking clinically relevant doses of colistin (5 mg/kg per day) and tigecycline (50 or 100 mg twice daily) (Cai et al., 2017). Another IVM study demonstrated the importance of tigecycline dosage regimen in determining the efficacy of polymyxin B-tigecycline combination against MDR A. baumannii, as synergy was achieved only when polymyxin B (1 mg/kg per 12 hours) was used in combination with tigecycline at a higher dose (200 mg/12 h) but not at 100 mg/12 h (Hagihara et al., 2014). In a mouse sepsis model, the combination of CMS (2.5 mg/kg per 12 hours) and tigecycline (10 mg/kg per 12 hours) did not result in better efficacy against carbapenem-resistant K. pneumoniae as compared with monotherapies, which is very likely due to the very low doses of both antibiotics considering animal scaling (Demiraslan et al., 2014). Similar PK/PD issues also occurred in a mouse pneumonia model caused by XDR A. baumannii, as the doses of colistin and tigecycline (CMS 1.25 mg/kg per 6 hours and tigecycline 10 mg/kg per 12 hours) were very low (Mutlu Yilmaz et al., 2012). In a small clinical study, treatment of bacteremia caused by KPC-producing K. pneumoniae with colistin and tigecycline as monotherapies resulted in mortality rates of 66.7% (4 of 7) and 40% (2 of 5), respectively, whereas no mortality (0 of 9) was observed with the colistin-tigecycline combination (Zarkotou et al., 2011). Similar results were also demonstrated in a small retrospective cohort study with mortality rates of 57.1% (4 of 7) and 80% (4 of 5) with polymyxin B (or CMS) and tigecycline as monotherapies, respectively, and only the one patient who received polymyxin-tigecycline survived (Qureshi et al., 2012). Despite the small sample size of these clinical studies, colistin-tigecycline combination therapy remains a promising therapeutic option, especially against KPC-producing K. pneumoniae bacteremia, and further clinical studies are warranted.
D. Other Polymyxin Combinations
Combinations of polymyxins with other antibiotics have also been conducted in preclinical and clinical studies. Synergy between colistin and vancomycin/teicoplanin has been demonstrated in vitro against MDR A. baumannii, including colistin-resistant strains (Gordon et al., 2010; Wareham et al., 2011; Percin et al., 2014); however, the limited clinical evidence did not support the use of this combination therapy (Leite et al., 2016). With the current recommended dosage regimens, colistin-vancomycin combination did not lower the mortality rate in patients (5 of 7; 71%) when compared with patients who received antibiotic therapy without vancomycin (8 of 14; 57%); however, definite conclusions could not be made because of the small sample size and study design (Leite et al., 2016). Furthermore, nephrotoxicity in patients is another major concern with this combination.
Colistin-fosfomycin is another promising combination that has shown in vitro synergy and bactericidal effect against P. aeruginosa (Di et al., 2015; Walsh et al., 2016); however, despite the increased killing activity, emergence of fosfomycin resistance was not eliminated (Walsh et al., 2016). An IVM study with clinically achievable concentrations of colistin and fosfomycin demonstrated the efficacy of the combination of colistin (Cmax/Cmin of 3.0/0.75 mg/l) and fosfomycin (Cmax/Cmin of 250/40 mg/l) against NDM-1–producing E. coli, K. pneumoniae, and Klebsiella oxytoca, with increased bacterial killing activity (Albur et al., 2015). Similar results were also demonstrated against KPC2-producing K. pneumoniae in an in vitro study for the combination of colistin (75,000 IU/kg per 12 hours) and fosfomycin (8 g/8 h) (Yu et al., 2017a). Furthermore, the combination of colistin (10 mg/kg) and fosfomycin (150 mg/kg) eradicated ESBL-producing E. coli biofilm in a guinea pig foreign-body infection model, suggesting the potential role of this combination for biofilm-related infections (Corvec et al., 2013). In a clinical study, 11 critically ill patients with carbapenem-resistant K. pneumoniae infection were treated with fosfomycin (2–4 g/6 h) in addition to CMS (six patients), gentamicin (three patients), or piperacillin/tazobactam (one patient) (Michalopoulos et al., 2010). Although a direct comparison between the effectiveness of these combinations could not be made, fosfomycin appeared as a safe adjuvant agent with no adverse events reported from these patients (Michalopoulos et al., 2010).
There are limited preclinical and clinical data on the combinations of colistin with other antibiotics such as doxycycline, daptomycin, ceftazidime, ciprofloxacin, aminoglycoside, and ampicillin/sulbactam. In an IVM study with XDR A. baumannii, daptomycin (10 mg/kg per day) and colistin [mimicking CMS 3 MIU/8 h (i.e., 240 mg/8 h)] combination therapy was found to be the most effective in comparison with colistin-imipenem (1 g/8 h) and imipenem-ertapenem (1 g/8 h and 1 g/day, respectively) (Córdoba et al., 2015). There is a lack of data supporting the in vitro synergistic activity of the combination of polymyxins and ampicillin/sulbactam; however, in a prospective, open-label, randomized study, the combination of colistin (as CMS) with a high dose of ampicillin/sulbactam showed favorable clinical response in 50 patients with VAP due to A. baumannii resistant to carbapenems but susceptible to colistin and ampicillin-sulbactam (Makris et al., 2018). In the same study, the combination of intravenous CMS [3 MIU/8 h (i.e., 240 mg/8 h)] and ampicillin/sulbactam ([6 (4/2)] g/6 h) demonstrated a favorable clinical response than those receiving CMS monotherapy (Makris et al., 2018). Combinations of colistin with ciprofloxacin or ceftazidime have been shown to exhibit good synergy against MDR P. aeruginosa using a checkerboard method (D'Souza et al., 2014). Additionally, the synergistic activity with the combination of colistin (Cmax 6 or 18 mg/l) and ceftazidime (constant Css 50 mg/l) was shown against MDR P. aeruginosa in an IVM study (Gunderson et al., 2003). Despite the positive results in the literature, a recent in vitro report suggested that the addition of colistin (2 mg/l) to ceftazidime-avibactam (concentrations at 0.25, 1, and 4 × MIC) was unable to potentiate the killing activity against most isolates of carbapenem-resistant Enterobacterales (Shields et al., 2018). Colistin combination with the next-generation aminoglycoside plazomicin showed synergy against 60% of 164 isolates of carbapenemase-producing Enterobacterales in a checkerboard study, which was superior over plazomicin-meropenem (20%) and plazomicin-fosfomycin (25%) (Rodríguez-Avial et al., 2015). Against KPC-producing K. pneumoniae, doxycycline-polymyxin B combination can also be a potential option with good synergistic activity (Elemam et al., 2010).
E. Triple Combination Treatment with Polymyxins
To date, no polymyxin double combinations can kill 100% of isolates of MDR Gram negative pathogens, and investigations on polymyxin triple combinations are emerging. In the clinic, triple combinations may be the only option for patients with bacterial infections that are resistant to all current antibiotics, including polymyxins. In a static time-kill study with polymyxin B, doripenem, and rifampicin at 0.25 × MIC, bactericidal activity was achieved with the triple combination against all carbapenem-resistant clinical isolates of P. aeruginosa (5 of 5) and E. coli (5 of 5), 80% of K. pneumoniae (4 of 5), and 60% of A. baumannii (3 of 5) (Urban et al., 2010). Among these isolates, double combinations were ineffective against 3 P. aeruginosa, 3 K. pneumoniae, 1 E. coli, and 1 A. baumannii (Urban et al., 2010). The potential of colistin triple combination with carbapenem and rifampicin was also demonstrated against VIM- and NDM-producing K. pneumoniae in a time-kill study (Tängdén et al., 2014). Although synergy was demonstrated by aztreonam (17 mg/l), fosfomycin (83 mg/l), or rifampicin (1.7 mg/l) in triple combinations with colistin (4 mg/l) and meropenem (6.8 mg/l), the colistin-meropenem-rifampicin combination was the most effective and the only one with synergistic and bactericidal effect against all four VIM- and NDM-producing K. pneumoniae (Tängdén et al., 2014). An IVM study stimulated clinically relevant concentrations of polymyxin B (Css 0.5, 1, or 2 mg/l), meropenem (1 or 2 g/8 h), and rifampicin (600 mg every 8 or 12 hours) in mono, double, or triple combination therapies and showed that only the triple combination was effective against two polymyxin-resistant KPC-producing K. pneumoniae (Diep et al., 2017). In an in vitro study, double combinations polymyxin B-rifampicin (0.25 mg/l and 0.5 mg/l, respectively) and polymyxin B-imipenem (0.25 mg/l and 8 mg/l, respectively) were bactericidal against seven out of eight MDR A. baumannii isolates, and only the triple combination of polymyxin B-imipenem-rifampicin (0.25 mg/l, 8 mg/l, and 0.5 mg/l, respectively) was bactericidal against all eight isolates (Yoon et al., 2004).
An in vitro study examined the triple combination of colistin (2 mg/l), doripenem (8 mg/l), and sulbactam (4 mg/l) against 18 A. baumannii isolates collected from nine patients with respiratory tract infections prior and after treatment with CMS and doripenem (Oleksiuk et al., 2014). An overall greater killing at 24 hours was observed with the triple combination than colistin-doripenem and colistin-sulbactam combinations, with the mean log10 killing of -5.74, -2.88, and -1.51, respectively (Oleksiuk et al., 2014). The effectiveness of polymyxin B, meropenem, and ampicillin/sulbactam triple combination against polymyxin-resistant, carbapenem-resistant A. baumannii was demonstrated in two HFIM studies (Lenhard et al., 2017a,b). With the triple combination of polymyxin B (1.43 mg/kg per 12 hours with a loading dose), meropenem (2 g/8 h), and ampicillin/sulbactam (8 or 4 g/8 h), a rapid eradication of A. baumannii was observed by 96 hours, which was otherwise unachievable with any mono and double combination therapies (Lenhard et al., 2017a,b). Importantly, the use of this triple combination is also supported by a small clinical study involving 17 patients with infections (mainly ventilator-associated pneumonia) caused by colistin-resistant A. baumannii (Qureshi et al., 2015). Among these patients, all seven patients who received CMS-carbapenem-ampicillin/sulbactam survived, whereas 6 out of the 10 patients who received other antibiotic regimens deceased within 30 days of infection (Qureshi et al., 2015). Another triple combination with clinical evidence is the combination of CMS (100 mg CBA/36 h), doripenem (250 mg/12 h), and tobramycin (40 mg/48 h) (Sun et al., 2010). A liver transplant recipient with bacteremia caused by MDR P. aeruginosa was successfully treated with this novel combination after treatment failure with conventional monotherapies of piperacillin/tazobactam, ciprofloxacin, and tobramycin, and even with a triple combination of piperacillin/tazobactam, tobramycin, and rifampicin (Sun et al., 2010). In a retrospective study conducted in three large Italian teaching hospitals, 125 patients with bacteremia caused by KPC-producing K. pneumoniae were treated with a range of antibiotics (Tumbarello et al., 2012). In a total of 52 (41.6%) nonsurvivors, 25 (48.1%), 23 (44.2%), and 4 (7.7%) patients were treated with mono, double, and triple combination therapies, respectively. Among the 125 patients (52 nonsurvivors and 73 survivors), 23 patients were treated with a triple combination; for the nonsurvivors, two were from the 16 (12.5%) patients who received CMS-meropenem-tigecycline, and two were from the seven (28.6%) patients who received gentamicin-meropenem-tigecycline (n = 6) or CMS-meropenem-gentamicin (n = 1). Overall, the multivariate analysis demonstrated that the triple combination of CMS-meropenem-tigecycline was associated with a reduced risk of mortality (Tumbarello et al., 2012).
It is evident that the clinical evidence on polymyxin combination therapy is limited, and the safety of polymyxin combinations has not been well evaluated, in particular the nephrotoxicity. Importantly, there is often a lack of PK/PD considerations, in particular at the infection site. A number of major challenges need to be addressed in clinical evaluations of antibiotic combination therapies (including polymyxins), such as ethics, appropriate endpoints, sample size, the optimal dosage regimen of each antibiotic, appropriate control groups, and PK/PD/TD modeling. Synergy occurs only when different antibiotics achieve optimal exposure at the infection site with the right timing.
F. Mechanistic Insights into Polymyxin Combination Therapy
It is generally believed that the synergy of polymyxin combinations is due to the disorganization of bacterial outer membrane by polymyxins, which facilitates the access of other antibiotics to their intracellular targets. In fact, at the transcriptomic and metabolomic levels the synergy mechanisms of polymyxin combinations are far more complicated. A pan-transcriptomic analysis revealed that polymyxin treatment caused rapid outer membrane remodeling, upregulation of efflux pumps, and downregulation of fatty acid biosynthesis in A. baumannii (Li et al., 2020a). Integrative modeling of correlative transcriptomics and metabolomics data using a strain-specific genome-scale metabolic model discovered the interplay of multiple key metabolic pathways in A. baumannii in response to colistin treatment: 1) downregulation of tricarboxylic acid cycle and biosynthesis of peptidoglycan and lipopolysaccharides; 2) upregulated fluxes through gluconeogenesis, pentose phosphate pathway, and biosynthesis of nucleotides and amino acids; and 3) altered fluxes over respiratory chain (Zhu et al., 2019). Using an IVM to mimic their PK in patients, colistin treatment (continuous infusion at 2 mg/l) within 1 hour caused >400 differentially regulated genes, in particular those for outer membrane biogenesis, phospholipid trafficking, and fatty acid metabolism in A. baumannii (Henry et al., 2015). Interestingly, no differentially regulated genes were shown after doripenem treatment (Cmax 25 mg/l; half-life 1.5 hours) at 15 minutes, but 45 genes were differentially expressed at 1 hour. The combination of colistin and doripenem led to >450 differentially expressed genes in A. baumannii at 1 hour, and >70% of the significant genes were also observed with colistin monotherapy (Henry et al., 2015). A metabolomics study demonstrated that the synergistic killing of A. baumannii by colistin and doripenem occurred via the inhibition of different key metabolic pathways in a time-dependent manner (Maifiah et al., 2017), which was consistent with the transcriptomics results (Henry et al., 2015). An early (15 minutes and 1 hour) disruption of outer membrane and cell wall was induced by colistin, and inhibition of cell wall synthesis by doripenem occurred subsequently (at 4 hours) (Maifiah et al., 2017). The colistin-doripenem combination significantly perturbed several key metabolic pathways in A. baumannii relative to either monotherapy, including the pentose phosphate pathway (induced initially by colistin at 15 minutes and 1 hour and by doripenem at 4 hours) and downregulation of cell wall biosynthesis (via d-sedoheptulose 7-phosphate) and nucleotide metabolism (via d-ribose 5-phosphate) (Maifiah et al., 2017). For colistin-aztreonam combination against A. baumannii, colistin caused an early (1 hour) disruption to the cell envelope, and aztreonam inhibited cell wall synthesis at later time points (4 and 24 hours) (Han et al., 2018). The level of two amino sugars, UDP-N-acetylglucosamine and UDP-N-acetylmuramate was significantly decreased by aztreonam alone and the combination at 4 and 24 hours (Han et al., 2018).
In P. aeruginosa, polymyxins disrupted cell envelope and induced significant perturbations in lipid and fatty acid metabolism pathways at early time points (15 minutes and 1 hour) (Hussein et al., 2019; Lin et al., 2019c). Amikacin monotherapy caused significant perturbations in amino acid metabolism in P. aeruginosa at 1 and 4 hours, and the synergistic killing by the polymyxin-amikacin combination involved the inhibition of cell envelope biogenesis and central carbohydrate metabolism, decreased levels of amino sugars, and a downregulated nucleotide pool (Hussein et al., 2019). The polymyxin B-enrofloxacin combination perturbed lipid metabolites in XDR P. aeruginosa at 1 hour, which was mainly driven by polymyxin B (Lin et al., 2019c). At 4 hours, the synergy was mainly driven by enrofloxacin through the inhibition of DNA replication; and importantly, polymyxin resistance was minimized by the combination via the inhibition of lipid A modification with L-Ara4N (Lin et al., 2019c). In K. pneumoniae, a transcriptomic study revealed that the synergy of the combination of polymyxin B (1 mg/l as continuous infusion) and chloramphenicol (Cmax 8 mg/l; half-life 4 hours) was persistent over 24 hours and mainly driven by chloramphenicol via perturbations of cell envelope synthesis and metabolism of carbohydrates, nucleotides, and amino acids (Abdul Rahim et al., 2020). Notably, chloramphenicol and its combination with polymyxin B significantly inhibited lipid A modification and decreased the resistance to polymyxin via downregulating the expression of the arn operon (Abdul Rahim et al., 2015, 2020). Overall, recent systems pharmacological studies highlight the significance of time-dependent synergistic effect of polymyxin combinations, and synergistic combinations may not be able to directly inhibit polymyxin resistance due to lipid A modifications. These results are crucial for the optimization of the dosage regimens of polymyxin combination therapy in patients.
In summary, the currently available preclinical and clinical data suggest a potential benefit with synergistic polymyxin combinations to treat MDR Gram negative pathogens. Ultimately, personalized combination therapy should be achieved based upon the condition of patient, type of infection, pathogen, rapid diagnosis, mechanisms of killing, resistance and toxicity, therapeutic drug monitoring, and PK/PD/TD using a systems approach.
X. Antiendotoxin Properties of Polymyxins
Endotoxins are the major component of Gram negative bacterial outer membrane that can cause fever, increased heart rate, inflammation, septic shock, and many other pathogenic effects (Peterson, 1996). Sepsis is a major global health problem, and Gram negative bacteremia and endotoxemia are often strong predictive of mortality in patients (Hurley et al., 2012). Thus, antiendotoxin therapy is an important therapeutic strategy in patients with Gram negative bacteremia. Owing to the ability of polymyxins to specifically bind to the lipid A (endotoxin) (Velkov et al., 2010), polymyxin B is a strong candidate as an antiendotoxin therapeutic agent (Morrison and Jacobs, 1976). The antiendotoxin activity of polymyxin B was illustrated in endotoxemia and septicemia animal models (Corrigan and Kiernat, 1979; Ingoldby, 1980; Flynn et al., 1987). Polymyxin B–immobilized fibers, in which polymyxin B is covalently bound and immobilized to polystyrene fiber, were introduced to selectively remove endotoxin in blood. Using an endotoxemia canine model, endotoxin was successfully removed from the blood by extracorporeal hemoperfusion with polymyxin B–immobilized fibers, and improved survivability was observed (Hanasawa et al., 1988). An increased survival rate was also reported in an E. coli sepsis canine model after extracorporeal hemoperfusion with polymyxin B–immobilized fibers (Hanasawa et al., 1989). Even though bactericidal activity was observed with polymyxin B–immobilized fibers against P. aeruginosa in vitro (Shoji et al., 1998), it is unclear whether the antibacterial killing was due to the release of polymyxin B from the immobilized fibers.
Toraymyxin, a blood purification cartridge containing polymyxin B–immobilized fibers, has been developed for the removal of endotoxin from blood in humans (Tani et al., 2019). The first clinical data with Toraymyxin were reported in 1994, showing a decreased level of circulating endotoxin and an improved hyperdynamic state in patients with septic multiple organ failure (n = 16) (Aoki et al., 1994). Since the employment of Toraymyxin for endotoxemia and septic shock treatment in Japan in 1994, the safety profile of Toraymyxin has been well established in more than 100,000 emergency and ICU cases in Japan (Shimizu et al., 2017). Since the Conformitè Europëenne (CE) mark approval in Europe in 1998, two randomized controlled trials were conducted in Europe in 2005 (n = 36) and 2009 (n = 64), and the results showed that Toraymyxin is safe, improves sepsis-associated cardiac and renal dysfunction, and reduces mortality caused by sepsis (Vincent et al., 2005; Cruz et al., 2009). A meta-analysis revealed 33%–80% reduction in plasma endotoxin level and significantly lower mortality risk with Toraymyxin with a pooled sample size of 1,425 patients from 28 studies; however, bias (e.g., preference of the publication of trials with positive results rather than negative and neutral results) and lack of blinding should be taken into consideration (Cruz et al., 2007). Notably, the randomized controlled trials published in 2015 (France; n = 243) and 2018 (North America; n = 450) reported conflicting results with no significant improvement in organ failure and survivability in favor of Toraymyxin over conventional treatments (Payen et al., 2015; Dellinger et al., 2018). Nevertheless, Toraymyxin remains a promising antiendotoxin therapy, especially in patients who are presented with severe septicemia; and further clinical evidence is required.
XI. Current Landscape of Polymyxin Drug Discovery
In addition to optimizing the clinical use of polymyxin B, colistin, and their combinations, the discovery of new-generation polymyxins is also crucial in combating infections caused by MDR Gram negative pathogens. There are a number of major challenges in the discovery of novel polymyxins, including the complex PK/PD, lack of understanding on the mechanisms of toxicities (e.g., acute toxicity, nephrotoxicity, and hyperpigmentation), and narrow chemical space due to the closely related relationships between the structure, activity, PK, and toxicity. Nevertheless, significant progress has been made over the last decade in the discovery of novel, safer polymyxins with several groups working actively in the field (Brown and Dawson, 2017; Vaara, 2019b; Velkov and Roberts, 2019). One of the leading groups at Monash University is the first to employ structure-activity-PK-toxicity relationship based mechanistic models to design novel polymyxins that target polymyxin resistance in Gram negative bacteria with reduced nephrotoxicity in comparison with polymyxin B and colistin (Velkov et al., 2014, 2018b; Velkov and Roberts, 2019). Their first-generation lipopeptide (FADDI-002) incorporated modifications of the core polymyxin scaffold (Table 1) with l-octylglycine at position 7 to increase the hydrophobic interactions with lipid A, thereby circumventing polymyxin resistance as a result of the modifications of the lipid A phosphates. Their novel polymyxins with lipidic groups at positions 6/7 and the N-terminus displayed significantly increased antimicrobial activity against polymyxin-resistant clinical isolates while also maintaining their activity against polymyxin-susceptible strains of P. aeruginosa, A. baumannii, and K. pneumoniae (Velkov et al., 2014). Notably, several lipopeptides showed, in addition to their activities against Gram negative superbugs, unexpected activity against the problematic Gram positive vancomycin-resistant Enterococcus faecium and methicillin- or vancomycin-resistant Staphylococcus aureus (Velkov et al., 2014; Zhao et al., 2016). In a neutropenic mouse lung infection model, FADDI-002 displayed improved in vivo efficacy against a polymyxin-resistant clinical isolate of P. aeruginosa as compared with colistin (Velkov et al., 2014). The kidneys of mice subcutaneously treated with lipopeptides FADDI-003 or FADDI-019 (accumulated dose 105 mg/kg) revealed no significant lesions in the cortex and medulla, and papilla regions essentially resembled the kidneys of mice treated with the saline control; no histologic grade was given (Velkov et al., 2014). One of their second-generation lipopeptides, QPX9003, is 4-fold more potent than colistin against MDR P. aeruginosa (n = 1000, including resistance to β-lactams/β-lactamase inhibitors) and carbapenem-resistant A. baumannii (n = 503) (Castanheira et al., 2019). Importantly, QPX9003 shows efficacy in mouse and rat pneumonia models that is superior to polymyxin B and is not inactivated by lung surfactant. QPX9003 is currently in development at Qpex Biopharma, Inc. (CA) with an investigational new drug submission in the near future (Sabet et al., 2019).
Northern Antibiotics (Helsinki, Finland) is developing several promising polymyxin analogs that possess only three positive charges (compared with the five carried by polymyxin B and colistin) (Vaara et al., 2008, 2010a,b, 2013; Ali et al., 2009; Vaara and Vaara, 2010, 2013; Vingsbo Lundberg et al., 2010; Vaara, 2013). The most promising lead compound reported was NAB739, which shared an identical cyclic core to that of polymyxin B and a modified linear segment consisting of octanoyl-[Thr1-d-Ser2]polymyxin B (Vaara et al., 2008). NAB739 displays good activity against E. coli (n = 66), with MIC90 values (1 to 2 mg/l) comparable to that of polymyxin B (Vaara et al., 2008; Vaara et al., 2013); and it can also sensitize A. baumannii strains to rifampicin, clarithromycin, and vancomycin by facilitating their entry into the cell (Vaara et al., 2008). In human renal proximal tubular HK-2 cells, NAB739 was 26-fold less toxic than polymyxin B and 7.5-fold less toxic than colistin (Vaara and Vaara, 2013).
Researchers at Hokuriku University reported some interesting des-fatty acyl polymyxin B analogs with potent antipseudomonal activity (MICs 0.5–1 mg/l) (Katsuma et al., 2009; Sato et al., 2011). Cubist Pharmaceuticals (MA) had a significant discovery program on novel N-terminal modified polymyxin analogs based on intellectual property developed by BioSource Pharmaceuticals (Leese, 2010). The Cubist polymyxin program centered around a novel semisynthetic methodology that involved enzymatically removing the N-terminal fatty acyl groups of polymyxin B or colistin mixtures to provide a single polymyxin core, of which the N-terminus was derivatized with individual novel haloaryl groups. The idea behind these modifications was to reduce nephrotoxicity by decreasing the hydrophobicity of the N-terminal fatty acyl group. Out of over 200 analogs, the Cubist lead compound was the 2-chlorophenylurea derivative CB-182,804 (Keith et al., 2010). In vivo studies in neutropenic mouse lung and thigh infection model showed that CB-182,804 had comparable or slightly improved in vivo efficacy to polymyxin B (Leese, 2010). In an in vitro cytotoxicity assay utilizing rat kidney proximal tubule cells, CB-182,804 displayed significantly reduced cytotoxicity (EC50 > 1000 µg/ml) compared with polymyxin B (EC50 318 µg/ml). Unfortunately, CB-182,804 failed in a phase 1 study as a result of severe nephrotoxicity (Blaskovich et al., 2018).
Pfizer (NY) also instigated a discovery program trying to alleviate polymyxin nephrotoxicity through modifications to the core and N-terminus of polymyxin B (Velkov et al., 2016a; Brown and Dawson, 2017; Velkov and Roberts, 2019). Through this work, it was demonstrated that lipopeptide 5a with the substitution of Dab3 with a diaminopropionic acid residue displayed a 2-fold improvement in MICs compared with polymyxin B against P. aeruginosa and A. baumannii strains, including several polymyxin-resistant strains. Furthermore, screening of lipopeptide 5a for in vitro nephrotoxicity utilizing human renal proximal epithelial cells showed a 2-fold decrease in cytotoxicity relative to polymyxin B. Based upon the nephrotoxicity results in rats and dogs, the overall therapeutic index of lipopeptide 5a is not better than that of polymyxin B (Brown and Dawson, 2017). Unfortunately, this polymyxin discovery program is discontinued by Pfizer (Velkov et al., 2016a).
Cantab Anti-Infectives (Hertfordshire, UK) has focused on the development of novel polymyxin nonapeptide derivatives to address the toxicity issues of the polymyxins (Saadi et al., 2013). Similar to the work of Sato and coworkers discussed earlier, Cantab compounds used the polymyxin nonapeptide core with the introduction of novel branched amine-containing acyl moieties with specific regio- and stereochemistry at the N-terminus. The lead lipopeptide from the Cantab discovery program was Cantab Ant-infectives (CA) CA1206, now SPR206, which is being developed by Spero Therapeutics (Brown et al., 2019; Vaara, 2019b). SPR206 contains a polymyxin B nonapeptide and a β-branched aminobutyrate N-terminus with an aryl substituent that displays low cytotoxicity, kidney exposure, and nephrotoxicity and an increased therapeutic window in mice compared with polymyxin B (Brown et al., 2019). A phase 1 clinical trial of SPR206 was conducted in 96 healthy volunteers (SPR206 or placebo) with seven multiple ascending dose cohorts (single intravenous doses from 10 to 400 mg) and 75–450 mg per day for 7 days plus 300 mg per day for 14 days across five multiple ascending dose cohorts. Preliminary data indicate that SPR206 was well tolerated in healthy subjects at doses up to 100 mg every 8 h for 14 days; and the PK was linear in the tested range, with mean plasma exposure of SPR206 consistent with preclinical models (https://investors.sperotherapeutics.com/news-releases/news-release-details/spero-reports-preliminary-findings-phase-1-clinical-trial-spr206).
MicuRx pharmaceuticals also has an active polymyxin discovery program that adopts a novel MRX-8 prodrug approach wherein the fatty acyl tail of the polymyxin core structure is linked to a cycle via an ester bond (Gordeev, 2018; Vaara, 2019b). This ester bond facilitates the breakdown of the parent compound in plasma into a des-fatty acyl less toxic nonapeptide form (Gordeev et al., 2016; Gordeev, 2018). Compared with polymyxin B, MRX-8 has higher MICs but better efficacy against lung infections caused by Gram negative pathogens in mice (Gordeev, 2018). Currently, an investigational new drug application of MRX-8 has been submitted to the FDA in the United States (http://micurxchina.com/project-progress).
In summary, in the wake of our increasing understanding of polymyxin PK/PD/TD, mode of action, and toxicity mechanisms, recent medicinal chemistry efforts have yielded several promising novel polymyxin lipopeptides with improved activity and/or toxicity compared with polymyxin B and colistin. Hopefully, new-generation, safer polymyxins will be approved for the treatment of Gram negative superbugs in patients in the near future.
XII. Conclusions
In stark contrast to most other antibiotic classes, the polymyxins did not follow the conventional antibiotic development pathway, with little preclinical development before being implemented for human use more than 60 years ago. Not surprisingly, the preclinical and clinical pharmacological knowledge base for polymyxins has largely been generated by academic research groups as opposed to pharmaceutical companies. Over the last two decades, polymyxins have virtually undergone a redevelopment phase, funded mainly by government agencies. The latest pharmacological research from academia led to the first scientifically based dosing guidelines for intravenous CMS. Clinicians are now in a much better position to optimize its use, and clinical practice continues to improve worldwide. Notwithstanding, there are still several major knowledge gaps that are crucial for the development of PK/PD/TD–informed dosing guidelines, in particular in the case of polymyxin B. This cannot be understated, as no novel classes of antibiotics will be available for Gram negative superbugs in the next few years. It is crucial to optimize the use of polymyxins in the clinic by maximizing their efficacy while minimizing the emergence of resistance and toxicity. Underpinned by the recent significant pharmacological achievements, several novel, safer polymyxin candidates are undergoing preclinical and clinical evaluations. It is without question that polymyxins will continue playing a major role in the battle against life-threatening infections caused by MDR Gram negative pathogens.
Abbreviations
- ATCC
American Type Culture Collection
- AUC
area under the plasma concentration curve
- CAMHB
cation-adjusted Mueller-Hinton broth
- CAS
Chemical Abstracts Service
- CBA
colistin base activity
- cfu
colony forming unit
- CLSI
Clinical and Laboratory Standards Institute
- CMS
colistin methanesulfonate
- CNS
central nervous system
- Css
steady-state concentration
- Css,avg
average steady-state concentration
- Dab
2,4-diaminobutyric acid
- DPI
dry powder inhaler
- ELF
epithelial lining fluid
- ESBL
extended-spectrum β-lactamase
- EUCAST
European Committee on Antimicrobial Susceptibility Testing
- FADD
Fas-associated death domain
- fAUC
area under the curve of unbound drug
- fCmax
maximum concentration of unbound drug
- FDA
Food and Drug Administration
- HFIM
hollow-fiber infection model
- ICU
intensive care unit
- IU
international unit
- IVM
in vitro one-compartment dynamic model
- Kdo
2-keto-3-deoxyoctonoic acid
- KIM-1
kidney injury molecule-1
- KPC
Klebsiella pneumoniae carbapenemase
- L-Ara4N
4-amino-l-arabinose
- LPS
lipopolysaccharide
- MDR
multidrug-resistant
- MIC
minimum inhibitory concentration
- MIU
million international unit
- NDH-2
type II NADH-quinone oxidoreductase
- NDM
New Delhi metallo-β-lactamase
- PAE
postantibiotic effect
- PD
pharmacodynamics
- PEPT2
peptide transporter 2
- pEtN
phosphoethanolamine
- PK
pharmacokinetics
- ROS
reactive oxygen species
- SDD
selective digestive tract decontamination
- SOD
selective oropharyngeal decontamination
- TCS
two-component regulatory system
- TD
toxicodynamics
- UTI
urinary tract infection
- VAP
ventilator-associated pneumonia
- XDR
extensively drug-resistant
- XFM
X-ray fluorescence microscopy
Authorship Contributions
Wrote or contributed to the writing of the manuscript: Nang, Azad, Velkov, Zhou, Li.
Footnotes
T.V., Q.T.Z., and J.L. are supported by the National Institutes of Health/National Institute of Allergy and Infectious Diseases under the award number [R01AI146160] and [R01AI132681]; and T.V. and J.L. under [R01AI132154].
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Allergy and Infectious Diseases or the National Institutes of Health. J.L. is an Australia National Health and Medical Research Council (NHMRC) Principal Research Fellow. The novel polymyxin candidates developed by J.L. and T.V.’s group were licensed to Qpex Biopharma. J.L. received seminar honoraria on the pharmacology of colistin and polymyxin B from Genentech, Inc.; DocMode; Healcare Pharmaceuticals; and a number of international conferences and research grants from Northern Antibiotics. No author has other actual or perceived conflicts of interest with the other contents of this article.
References
- Abdallah M, Olafisoye O, Cortes C, Urban C, Landman D, Quale J (2015) Activity of eravacycline against Enterobacteriaceae and Acinetobacter baumannii, including multidrug-resistant isolates, from New York City. Antimicrob Agents Chemother 59:1802–1805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Abdellatif S, Trifi A, Daly F, Mahjoub K, Nasri R, Ben Lakhal S (2016) Efficacy and toxicity of aerosolised colistin in ventilator-associated pneumonia: a prospective, randomised trial. Ann Intensive Care 6:26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Abdelraouf K, He J, Ledesma KR, Hu M, Tam VH (2012) Pharmacokinetics and renal disposition of polymyxin B in an animal model. Antimicrob Agents Chemother 56:5724–5727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Abdul Rahim N, Cheah SE, Johnson MD, Yu H, Sidjabat HE, Boyce J, Butler MS, Cooper MA, Fu J, Paterson DL, et al. (2015) Synergistic killing of NDM-producing MDR Klebsiella pneumoniae by two ‘old’ antibiotics-polymyxin B and chloramphenicol. J Antimicrob Chemother 70:2589–2597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Abdul Rahim N, Cheah SE, Johnson MD, Zhu Y, Yu HH, Sidjabat HE, Butler MS, Cooper MA, Fu J, Paterson DL, et al. (2020) Transcriptomic responses of a New Delhi metallo-β-lactamase-producing Klebsiella pneumoniae isolate to the combination of polymyxin B and chloramphenicol. Int J Antimicrob Agents 56:106061. [DOI] [PubMed] [Google Scholar]
- Abe T, Kakyo M, Sakagami H, Tokui T, Nishio T, Tanemoto M, Nomura H, Hebert SC, Matsuno S, Kondo H, et al. (1998) Molecular characterization and tissue distribution of a new organic anion transporter subtype (oatp3) that transports thyroid hormones and taurocholate and comparison with oatp2. J Biol Chem 273:22395–22401. [DOI] [PubMed] [Google Scholar]
- Abele-Horn M, Dauber A, Bauernfeind A, Russwurm W, Seyfarth-Metzger I, Gleich P, Ruckdeschel G (1997) Decrease in nosocomial pneumonia in ventilated patients by selective oropharyngeal decontamination (SOD). Intensive Care Med 23:187–195. [DOI] [PubMed] [Google Scholar]
- AbuOun M, Stubberfield EJ, Duggett NA, Kirchner M, Dormer L, Nunez-Garcia J, Randall LP, Lemma F, Crook DW, Teale C, et al. (2017) mcr-1 and mcr-2 variant genes identified in Moraxella species isolated from pigs in Great Britain from 2014 to 2015. J Antimicrob Chemother 72:2745–2749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahmed MU, Velkov T, Lin YW, Yun B, Nowell CJ, Zhou F, Zhou QT, Chan K, Azad MAK, Li J (2017) Potential toxicity of polymyxins in human lung epithelial cells. Antimicrob Agents Chemother 61:e02690-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahmed MU, Velkov T, Zhou QT, Fulcher AJ, Callaghan J, Zhou F, Chan K, Azad MAK, Li J (2019) Intracellular localization of polymyxins in human alveolar epithelial cells. J Antimicrob Chemother 74:48–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ainsworth GC, Brown AM, Brownlee G (1947) Aerosporin, an antibiotic produced by Bacillus aerosporus Greer. Nature 159:263. [DOI] [PubMed] [Google Scholar]
- Albur M, Noel A, Bowker K, MacGowan A (2012) Bactericidal activity of multiple combinations of tigecycline and colistin against NDM-1-producing Enterobacteriaceae. Antimicrob Agents Chemother 56:3441–3443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Albur M, Noel A, Bowker K, Macgowan A (2014) Colistin susceptibility testing: time for a review. J Antimicrob Chemother 69:1432–1434. [DOI] [PubMed] [Google Scholar]
- Albur MS, Noel A, Bowker K, MacGowan A (2015) The combination of colistin and fosfomycin is synergistic against NDM-1-producing Enterobacteriaceae in in vitro pharmacokinetic/pharmacodynamic model experiments. Int J Antimicrob Agents 46:560–567. [DOI] [PubMed] [Google Scholar]
- Ali FE, Cao G, Poudyal A, Vaara T, Nation RL, Vaara M, Li J (2009) Pharmacokinetics of novel antimicrobial cationic peptides NAB 7061 and NAB 739 in rats following intravenous administration. J Antimicrob Chemother 64:1067–1070. [DOI] [PubMed] [Google Scholar]
- Alothman GA, Ho B, Alsaadi MM, Ho SL, O’Drowsky L, Louca E, Coates AL (2005) Bronchial constriction and inhaled colistin in cystic fibrosis. Chest 127:522–529. [DOI] [PubMed] [Google Scholar]
- Al-Sweih NA, Al-Hubail MA, Rotimi VO (2011) Emergence of tigecycline and colistin resistance in Acinetobacter species isolated from patients in Kuwait hospitals. J Chemother 23:13–16. [DOI] [PubMed] [Google Scholar]
- Al Shirawi N, Memish ZA, Cherfan A, Al Shimemeri A (2006) Post-neurosurgical meningitis due to multidrug-resistant Acinetobacter baumanii treated with intrathecal colistin: case report and review of the literature. J Chemother 18:554–558. [DOI] [PubMed] [Google Scholar]
- Anderson DJ, Engemann JJ, Harrell LJ, Carmeli Y, Reller LB, Kaye KS (2006) Predictors of mortality in patients with bloodstream infection due to ceftazidime-resistant Klebsiella pneumoniae. Antimicrob Agents Chemother 50:1715–1720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andrei S, Droc G, Stefan G (2019) FDA approved antibacterial drugs: 2018-2019. Discoveries (Craiova) 7:e102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Antoniadou A, Kontopidou F, Poulakou G, Koratzanis E, Galani I, Papadomichelakis E, Kopterides P, Souli M, Armaganidis A, Giamarellou H (2007) Colistin-resistant isolates of Klebsiella pneumoniae emerging in intensive care unit patients: first report of a multiclonal cluster. J Antimicrob Chemother 59:786–790. [DOI] [PubMed] [Google Scholar]
- Antoniu SA, Cojocaru I (2012) Inhaled colistin for lower respiratory tract infections. Expert Opin Drug Deliv 9:333–342. [DOI] [PubMed] [Google Scholar]
- Aoki H, Kodama M, Tani T, Hanasawa K (1994) Treatment of sepsis by extracorporeal elimination of endotoxin using polymyxin B-immobilized fiber. Am J Surg 167:412–417. [DOI] [PubMed] [Google Scholar]
- Aoki N, Tateda K, Kikuchi Y, Kimura S, Miyazaki C, Ishii Y, Tanabe Y, Gejyo F, Yamaguchi K (2009) Efficacy of colistin combination therapy in a mouse model of pneumonia caused by multidrug-resistant Pseudomonas aeruginosa. J Antimicrob Chemother 63:534–542. [DOI] [PubMed] [Google Scholar]
- Armstrong GL, Conn LA, Pinner RW (1999) Trends in infectious disease mortality in the United States during the 20th century. JAMA 281:61–66. [DOI] [PubMed] [Google Scholar]
- Arroyo LA, García-Curiel A, Pachón-Ibañez ME, Llanos AC, Ruiz M, Pachón J, Aznar J (2005) Reliability of the E-test method for detection of colistin resistance in clinical isolates of Acinetobacter baumannii. J Clin Microbiol 43:903–905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arroyo LA, Herrera CM, Fernandez L, Hankins JV, Trent MS, Hancock REW (2011) The pmrCAB operon mediates polymyxin resistance in Acinetobacter baumannii ATCC 17978 and clinical isolates through phosphoethanolamine modification of lipid A. Antimicrob Agents Chemother 55:3743–3751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Athanassa ZE, Markantonis SL, Fousteri M-ZF, Myrianthefs PM, Boutzouka EG, Tsakris A, Baltopoulos GJ (2012) Pharmacokinetics of inhaled colistimethate sodium (CMS) in mechanically ventilated critically ill patients. Intensive Care Med 38:1779–1786. [DOI] [PubMed] [Google Scholar]
- Avedissian SN, Liu J, Rhodes NJ, Lee A, Pais GM, Hauser AR, Scheetz MH (2019) A review of the clinical pharmacokinetics of polymyxin B. Antibiotics (Basel) 8:31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aydin F, Atabay HI, Akan M (2001) The isolation and characterization of Campylobacter jejuni subsp. jejuni from domestic geese (Anser anser). J Appl Microbiol 90:637–642. [DOI] [PubMed] [Google Scholar]
- Azad MAK, Akter J, Rogers KL, Nation RL, Velkov T, Li J (2015a) Major pathways of polymyxin-induced apoptosis in rat kidney proximal tubular cells. Antimicrob Agents Chemother 59:2136–2143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azad MAK, Cheah SE, Yun B, Maifiah MHM, Johnson MD, Wang J, Boughton BA, Chapman R, Gould J, Hertzog P, et al. (2015b) P-A942: Understanding polymyxin-induced nephrotoxicity: combination of transcriptomics, metabolomics and mass spectrometry imaging, in Interscience Conference of Antimicrobial Agents and Chemotherapy/International Congress of Chemotherapy, Sandiego, CA.
- Azad MAK, Finnin BA, Poudyal A, Davis K, Li J, Hill PA, Nation RL, Velkov T, Li J (2013) Polymyxin B induces apoptosis in kidney proximal tubular cells. Antimicrob Agents Chemother 57:4329–4335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azad MAK, Li M, Zhu Y, Chapman R, Gould J, Hertzog P, Thompson PE, Roberts KD, Velkov T, Li J (2018) Understanding the structure-nephrotoxicity relationship of polymyxins: a transcriptomic approach, in Gordon Research Conference on New Antibacterial Discovery and Development, CA.
- Azad MAK, Roberts KD, Yu HH, Liu B, Schofield AV, James SA, Howard DL, Nation RL, Rogers K, de Jonge MD, et al. (2015c) Significant accumulation of polymyxin in single renal tubular cells: a medicinal chemistry and triple correlative microscopy approach. Anal Chem 87:1590–1595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azad MAK, Sivanesan S, Wang J, Chen K, Nation RL, Thompson PE, Roberts KD, Velkov T, Li J (2017a) Methionine ameliorates polymyxin-induced nephrotoxicity by attenuating cellular oxidative stress. Antimicrob Agents Chemother 62:e01254-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azad MAK, Yun B, Roberts KD, Nation RL, Thompson PE, Velkov T, Li J (2014) Measuring polymyxin uptake by renal tubular cells: is BODIPY-polymyxin B an appropriate probe? Antimicrob Agents Chemother 58:6337–6338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azad MAK, Zhu Y, Han ML, Wang J, Creek DJ, Velkov T, Li J (2017b) P-217: Effect of poly-L-aspartic acid on polymyxin-induced nephrotoxicity: a system pharmacology approach, in ASM Microbe, New Orleans. [Google Scholar]
- Azad MAK, Zhu Y, Yu HH, Velkov T, Li J (2019) P-8: Transcriptomic analysis reveals perturbations of cellular signalling networks in human lung epithelial cells due to polymyxin treatment, in VIIN Young Investigator Symposium, Melbourne, Australia.
- Babic JT, Manchandani P, Ledesma KR, Tam VH (2017) Evaluation of urinary KIM-1 for prediction of polymyxin B-induced nephrotoxicity. Antimicrob Agents Chemother 61:e01735-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baiocchi M, Catena V, Zago S, Badolati L, Baccarin M (2010) Intrathecal colistin for treatment of multidrug resistant (MDR) Pseudomonas aeruginosa after neurosurgical ventriculitis. Infez Med 18:182–186. [PubMed] [Google Scholar]
- Barcelos RP, Bresciani G, Rodriguez-Miguelez P, Cuevas MJ, Soares FA, Barbosa NV, González-Gallego J (2016) Diclofenac pretreatment effects on the toll-like receptor 4/nuclear factor kappa B-mediated inflammatory response to eccentric exercise in rat liver. Life Sci 148:247–253. [DOI] [PubMed] [Google Scholar]
- Bardet L, Baron S, Leangapichart T, Okdah L, Diene SM, Rolain JM (2017) Deciphering heteroresistance to colistin in a Klebsiella pneumoniae isolate from Marseille, France. Antimicrob Agents Chemother 61:e00356-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bardet L, Okdah L, Le Page S, Baron SA, Rolain JM (2019) Comparative evaluation of the UMIC Colistine kit to assess MIC of colistin of Gram-negative rods. BMC Microbiol 19:60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barnett M, Bushby SR, Wilkinson S (1964) Sodium sulphomethyl derivatives of polymyxins. Br J Pharmacol Chemother 23:552–574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baron S, Hadjadj L, Rolain JM, Olaitan AO (2016) Molecular mechanisms of polymyxin resistance: knowns and unknowns. Int J Antimicrob Agents 48:583–591. [DOI] [PubMed] [Google Scholar]
- Barret JP, Jeschke MG, Herndon DN (2001) Selective decontamination of the digestive tract in severely burned pediatric patients. Burns 27:439–445. [DOI] [PubMed] [Google Scholar]
- Behera B, Mathur P, Das A, Kapil A, Gupta B, Bhoi S, Farooque K, Sharma V, Misra MC (2010) Evaluation of susceptibility testing methods for polymyxin. Int J Infect Dis 14:e596–e601. [DOI] [PubMed] [Google Scholar]
- Behrens-Baumann W, Quentin CD, Gibson JR, Calthrop JG, Harvey SG, Booth K (1988) Trimethoprim-polymyxin B sulphate ophthalmic ointment in the treatment of bacterial conjunctivitis: a double-blind study versus chloramphenicol ophthalmic ointment. Curr Med Res Opin 11:227–231. [DOI] [PubMed] [Google Scholar]
- Bell PH, Bone JF, English JP, Fellows CE, Howard KS, Rogers MM, Shepherd RG, Winterbottom R, Dornbush AC, Kushner S, et al. (1949) Chemical studies on polymyxin; comparison with aerosporin. Ann N Y Acad Sci 51:897–908. [DOI] [PubMed] [Google Scholar]
- Benedict RG, Langlykke AF (1947) Antibiotic activity of Bacillus polymyxa. J Bacteriol 54:24–25. [PubMed] [Google Scholar]
- Bengoechea JA, Skurnik M (2000) Temperature-regulated efflux pump/potassium antiporter system mediates resistance to cationic antimicrobial peptides in Yersinia. Mol Microbiol 37:67–80. [DOI] [PubMed] [Google Scholar]
- Benifla M, Zucker G, Cohen A, Alkan M (2004) Successful treatment of Acinetobacter meningitis with intrathecal polymyxin E. J Antimicrob Chemother 54:290–292. [DOI] [PubMed] [Google Scholar]
- Benus RF, Harmsen HJ, Welling GW, Spanjersberg R, Zijlstra JG, Degener JE, van der Werf TS (2010) Impact of digestive and oropharyngeal decontamination on the intestinal microbiota in ICU patients. Intensive Care Med 36:1394–1402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bergen PJ, Bulitta JB, Forrest A, Tsuji BT, Li J, Nation RL (2010) Pharmacokinetic/pharmacodynamic investigation of colistin against Pseudomonas aeruginosa using an in vitro model. Antimicrob Agents Chemother 54:3783–3789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bergen PJ, Forrest A, Bulitta JB, Tsuji BT, Sidjabat HE, Paterson DL, Li J, Nation RL (2011a) Clinically relevant plasma concentrations of colistin in combination with imipenem enhance pharmacodynamic activity against multidrug-resistant Pseudomonas aeruginosa at multiple inocula. Antimicrob Agents Chemother 55:5134–5142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bergen PJ, Landersdorfer CB, Zhang J, Zhao M, Lee HJ, Nation RL, Li J (2012) Pharmacokinetics and pharmacodynamics of ‘old’ polymyxins: what is new? Diagn Microbiol Infect Dis 74:213–223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bergen PJ, Li J, Nation RL, Turnidge JD, Coulthard K, Milne RW (2008) Comparison of once-, twice- and thrice-daily dosing of colistin on antibacterial effect and emergence of resistance: studies with Pseudomonas aeruginosa in an in vitro pharmacodynamic model. J Antimicrob Chemother 61:636–642. [DOI] [PubMed] [Google Scholar]
- Bergen PJ, Li J, Rayner CR, Nation RL (2006) Colistin methanesulfonate is an inactive prodrug of colistin against Pseudomonas aeruginosa. Antimicrob Agents Chemother 50:1953–1958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bergen PJ, Tsuji BT, Bulitta JB, Forrest A, Jacob J, Sidjabat HE, Paterson DL, Nation RL, Li J (2011b) Synergistic killing of multidrug-resistant Pseudomonas aeruginosa at multiple inocula by colistin combined with doripenem in an in vitro pharmacokinetic/pharmacodynamic model. Antimicrob Agents Chemother 55:5685–5695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bergmans DC, Bonten MJ, Gaillard CA, Paling JC, van der Geest S, van Tiel FH, Beysens AJ, de Leeuw PW, Stobberingh EE (2001) Prevention of ventilator-associated pneumonia by oral decontamination: a prospective, randomized, double-blind, placebo-controlled study. Am J Respir Crit Care Med 164:382–388. [DOI] [PubMed] [Google Scholar]
- Berlana D, Llop JM, Fort E, Badia MB, Jódar R (2005) Use of colistin in the treatment of multiple-drug-resistant Gram-negative infections. Am J Health Syst Pharm 62:39–47. [DOI] [PubMed] [Google Scholar]
- Betrosian AP, Frantzeskaki F, Xanthaki A, Douzinas EE (2008) Efficacy and safety of high-dose ampicillin/sulbactam vs. colistin as monotherapy for the treatment of multidrug resistant Acinetobacter baumannii ventilator-associated pneumonia. J Infect 56:432–436. [DOI] [PubMed] [Google Scholar]
- Betts JW, Phee LM, Hornsey M, Woodford N, Wareham DW (2014a) In vitro and in vivo activities of tigecycline-colistin combination therapies against carbapenem-resistant Enterobacteriaceae. Antimicrob Agents Chemother 58:3541–3546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Betts JW, Phee LM, Woodford N, Wareham DW (2014b) Activity of colistin in combination with tigecycline or rifampicin against multidrug-resistant Stenotrophomonas maltophilia. Eur J Clin Microbiol Infect Dis 33:1565–1572. [DOI] [PubMed] [Google Scholar]
- Beveridge EG, Martin AJ (1967) Sodium sulphomethyl derivatives of polymyxins. Br J Pharmacol Chemother 29:125–135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Biagi M, Butler D, Tan X, Qasmieh S, Wenzler E (2019) A breath of fresh air in the fog of antimicrobial resistance: inhaled polymyxins for Gram-negative pneumonia. Antibiotics (Basel) 8:27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bishop RE, Gibbons HS, Guina T, Trent MS, Miller SI, Raetz CR (2000) Transfer of palmitate from phospholipids to lipid A in outer membranes of Gram-negative bacteria. EMBO J 19:5071–5080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blaskovich MAT, Pitt ME, Elliott AG, Cooper MA (2018) Can octapeptin antibiotics combat extensively drug-resistant (XDR) bacteria? Expert Rev Anti Infect Ther 16:485–499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bogiel T, Mikucka A, Skalski T, Gospodarek E (2010) [Occurrence and susceptibility to antibiotics of carbapenem-resistant Pseudomonas aeruginosa strains between 1998 and 2009]. Med Dosw Mikrobiol 62:221–229. [PubMed] [Google Scholar]
- Boisson M, Jacobs M, Grégoire N, Gobin P, Marchand S, Couet W, Mimoz O (2014) Comparison of intrapulmonary and systemic pharmacokinetics of colistin methanesulfonate (CMS) and colistin after aerosol delivery and intravenous administration of CMS in critically ill patients. Antimicrob Agents Chemother 58:7331–7339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borowiak M, Fischer J, Hammerl JA, Hendriksen RS, Szabo I, Malorny B (2017) Identification of a novel transposon-associated phosphoethanolamine transferase gene, mcr-5, conferring colistin resistance in d-tartrate fermenting Salmonella enterica subsp. enterica serovar Paratyphi B. J Antimicrob Chemother 72:3317–3324. [DOI] [PubMed] [Google Scholar]
- Bosso JA, Liptak CA, Seilheimer DK, Harrison GM (1991) Toxicity of colistin in cystic fibrosis patients. DICP 25:1168–1170. [DOI] [PubMed] [Google Scholar]
- Bozkurt-Guzel C, Gerceker AA (2012) In vitro pharmacodynamic properties of colistin methanesulfonate and amikacin against Pseudomonas aeruginosa. Indian J Med Microbiol 30:34–38. [DOI] [PubMed] [Google Scholar]
- Breazeale SD, Ribeiro AA, McClerren AL, Raetz CR (2005) A formyltransferase required for polymyxin resistance in Escherichia coli and the modification of lipid A with 4-Amino-4-deoxy-L-arabinose. Identification and function oF UDP-4-deoxy-4-formamido-L-arabinose. J Biol Chem 280:14154–14167. [DOI] [PubMed] [Google Scholar]
- Breazeale SD, Ribeiro AA, Raetz CR (2002) Oxidative decarboxylation of UDP-glucuronic acid in extracts of polymyxin-resistant Escherichia coli. Origin of lipid a species modified with 4-amino-4-deoxy-L-arabinose. J Biol Chem 277:2886–2896. [DOI] [PubMed] [Google Scholar]
- Breazeale SD, Ribeiro AA, Raetz CR (2003) Origin of lipid A species modified with 4-amino-4-deoxy-L-arabinose in polymyxin-resistant mutants of Escherichia coli. an aminotransferase (ArnB) that generates UDP-4-deoxyl-L-arabinose. J Biol Chem 278:24731–24739. [DOI] [PubMed] [Google Scholar]
- British Pharmacopoeia Commission (2018) British Pharmacopoeia, Stationary Office, London. [Google Scholar]
- British Pharmacopoeia Commission (2019) British Pharmacopoeia, Stationary Office, London. [Google Scholar]
- Brochmann RP, Toft A, Ciofu O, Briales A, Kolpen M, Hempel C, Bjarnsholt T, Høiby N, Jensen PO (2014) Bactericidal effect of colistin on planktonic Pseudomonas aeruginosa is independent of hydroxyl radical formation. Int J Antimicrob Agents 43:140–147. [DOI] [PubMed] [Google Scholar]
- Brown JM, Dorman DC, Roy LP (1970) Acute renal failure due to overdosage of colistin. Med J Aust 2:923–924. [DOI] [PubMed] [Google Scholar]
- Brown MR, Geaton EM, Gilbert P (1979) Additivity of action between polysorbate 80 and polymyxin B towards spheroplasts of Pseudomonas aeruginosa NCTC 6750. J Pharm Pharmacol 31:168–170. [DOI] [PubMed] [Google Scholar]
- Brown MR, Winsley BE (1968) Synergistic action of polysorbate 80 and polymyxin B sulphate on Pseudomonas aeruginosa. J Gen Microbiol 50:Suppl:ix. [PubMed] [Google Scholar]
- Brown P, Abbott E, Abdulle O, Boakes S, Coleman S, Divall N, Duperchy E, Moss S, Rivers D, Simonovic M, et al. (2019) Design of next generation polymyxins with lower toxicity: the discovery of SPR206. ACS Infect Dis 5:1645–1656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown P, Dawson MJ (2017) Development of new polymyxin derivatives for multi-drug resistant Gram-negative infections. J Antibiot (Tokyo) 70:386–394. [DOI] [PubMed] [Google Scholar]
- Brownlee G (1949) Antibiotics derived from bacillus polymyxa. Ann N Y Acad Sci 51:875–878. [DOI] [PubMed] [Google Scholar]
- Brownlee G, Bushby SR (1948) Chemotherapy and pharmacology of aerosporin; a selective Gram-negative antibiotic. Lancet 1:127–132. [DOI] [PubMed] [Google Scholar]
- Brownlee G, Bushby SR, Short EI (1949a) Comparative biological studies of polymyxin A and polymyxin D. Ann N Y Acad Sci 51:891–896. [DOI] [PubMed] [Google Scholar]
- Brownlee G, Bushby SR, Short EI (1949b) The pharmacology of polymyxin A, B, and D. Ann N Y Acad Sci 51:952–967. [DOI] [PubMed] [Google Scholar]
- Brownlee G, Bushby SR, Short EI (1952) The chemotherapy and pharmacology of the polymyxins. Br J Pharmacol Chemother 7:170–188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buelow E, Bello González TDJ, Fuentes S, de Steenhuijsen Piters WAA, Lahti L, Bayjanov JR, Majoor EAM, Braat JC, van Mourik MSM, Oostdijk EAN, et al. (2017) Comparative gut microbiota and resistome profiling of intensive care patients receiving selective digestive tract decontamination and healthy subjects. Microbiome 5:88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bukhary Z, Mahmood W, Al-Khani A, Al-Abdely HM (2005) Treatment of nosocomial meningitis due to a multidrug resistant Acinetobacter baumannii with intraventricular colistin. Saudi Med J 26:656–658. [PubMed] [Google Scholar]
- Bulitta JB, Yang JC, Yohonn L, Ly NS, Brown SV, D’Hondt RE, Jusko WJ, Forrest A, Tsuji BT (2010) Attenuation of colistin bactericidal activity by high inoculum of Pseudomonas aeruginosa characterized by a new mechanism-based population pharmacodynamic model. Antimicrob Agents Chemother 54:2051–2062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burt D, Crowell SJ, Ackley DC, Magee TV, Aubrecht J (2014) Application of emerging biomarkers of acute kidney injury in development of kidney-sparing polypeptide-based antibiotics. Drug Chem Toxicol 37:204–212. [DOI] [PubMed] [Google Scholar]
- Busch AE, Quester S, Ulzheimer JC, Waldegger S, Gorboulev V, Arndt P, Lang F, Koepsell H (1996) Electrogenic properties and substrate specificity of the polyspecific rat cation transporter rOCT1. J Biol Chem 271:32599–32604. [DOI] [PubMed] [Google Scholar]
- Buyck JM, Tulkens PM, Van Bambeke F (2013) Pharmacodynamic evaluation of the intracellular activity of antibiotics towards Pseudomonas aeruginosa PAO1 in a model of THP-1 human monocytes. Antimicrob Agents Chemother 57:2310–2318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cai X, Yang Z, Dai J, Chen K, Zhang L, Ni W, Wei C, Cui J (2017) Pharmacodynamics of tigecycline alone and in combination with colistin against clinical isolates of multidrug-resistant Acinetobacter baumannii in an in vitro pharmacodynamic model. Int J Antimicrob Agents 49:609–616. [DOI] [PubMed] [Google Scholar]
- Cajal Y, Berg OG, Jain MK (1995) Direct vesicle-vesicle exchange of phospholipids mediated by polymyxin B. Biochem Biophys Res Commun 210:746–752. [DOI] [PubMed] [Google Scholar]
- Cajal Y, Rogers J, Berg OG, Jain MK (1996) Intermembrane molecular contacts by polymyxin B mediate exchange of phospholipids. Biochemistry 35:299–308. [DOI] [PubMed] [Google Scholar]
- Campos MA, Vargas MA, Regueiro V, Llompart CM, Albertí S, Bengoechea JA (2004) Capsule polysaccharide mediates bacterial resistance to antimicrobial peptides. Infect Immun 72:7107–7114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Camus C, Bellissant E, Sebille V, Perrotin D, Garo B, Legras A, Renault A, Le Corre P, Donnio PY, Gacouin A, et al. (2005) Prevention of acquired infections in intubated patients with the combination of two decontamination regimens. Crit Care Med 33:307–314. [DOI] [PubMed] [Google Scholar]
- Cannatelli A, Giani T, D’Andrea MM, Di Pilato V, Arena F, Conte V, Tryfinopoulou K, Vatopoulos A, Rossolini GM, COLGRIT Study Group (2014) MgrB inactivation is a common mechanism of colistin resistance in KPC-producing Klebsiella pneumoniae of clinical origin. Antimicrob Agents Chemother 58:5696–5703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carattoli A, Villa L, Feudi C, Curcio L, Orsini S, Luppi A, Pezzotti G, Magistrali CF (2017) Novel plasmid-mediated colistin resistance mcr-4 gene in Salmonella and Escherichia coli, Italy 2013, Spain and Belgium, 2015 to 2016. Euro Surveill 22:30589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caroff M, Karibian D (2003) Structure of bacterial lipopolysaccharides. Carbohydr Res 338:2431–2447. [DOI] [PubMed] [Google Scholar]
- Cassir N, Rolain JM, Brouqui P (2014) A new strategy to fight antimicrobial resistance: the revival of old antibiotics. Front Microbiol 5:551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Castanheira M, Lindley J, Huynh H, Mendes RE, Lomovskaya O (2019) 690. Activity of a novel polymyxin analog, QPX9003, tested against resistant Gram-negative pathogens, including carbapenem-resistant Acinetobacter, Enterobacterales, and Pseudomonas. Open Forum Infect Dis 6:S313. [Google Scholar]
- Catch JR, Jones TS, Wilkinson S (1949) The chemistry of polymyxin A. Ann N Y Acad Sci 51:917–923. [DOI] [PubMed] [Google Scholar]
- Catchpole CR, Andrews JM, Brenwald N, Wise R (1997) A reassessment of the in-vitro activity of colistin sulphomethate sodium. J Antimicrob Chemother 39:255–260. [DOI] [PubMed] [Google Scholar]
- Centers for Disease Control and Prevention (2013) Antibiotic Resistance Threats in the United States, Centers for Disease Control and Prevention, Atlanta, GA. [Google Scholar]
- Centers for Disease Control and Prevention (2019) Antibiotic Resistance Threats in the United States, Centers for Disease Control and Prevention, Atlanta, GA. [Google Scholar]
- Cernohorská L, Sláviková P (2009) [Antibiotic resistance and biofilm formation in Pseudomonas aeruginosa strains isolated from patients with urinary tract infections]. Epidemiol Mikrobiol Imunol 58:154–157. [PubMed] [Google Scholar]
- Charretier Y, Diene SM, Baud D, Chatellier S, Santiago-Allexant E, van Belkum A, Guigon G, Schrenzel J (2018) Colistin heteroresistance and involvement of the PmrAB regulatory system in Acinetobacter baumannii. Antimicrob Agents Chemother 62:e00788-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheah SE, Li J, Tsuji BT, Forrest A, Bulitta JB, Nation RL (2016) Colistin and polymyxin B dosage regimens against Acinetobacter baumannii: differences in activity and the emergence of resistance. Antimicrob Agents Chemother 60:3921–3933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheah SE, Wang J, Nguyen VT, Turnidge JD, Li J, Nation RL (2015) New pharmacokinetic/pharmacodynamic studies of systemically administered colistin against Pseudomonas aeruginosa and Acinetobacter baumannii in mouse thigh and lung infection models: smaller response in lung infection. J Antimicrob Chemother 70:3291–3297. [DOI] [PubMed] [Google Scholar]
- Chen CC, Feingold DS (1972) Locus of divalent cation inhibition of the bactericidal action of polymyxin B. Antimicrob Agents Chemother 2:331–335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen SC, Kuo PL (2016) The role of galectin-3 in the kidneys. Int J Mol Sci 17:565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen YM, Fang WF, Kao HC, Chen HC, Tsai YC, Shen LS, Li CL, Chang HC, Huang KT, Lin MC, et al. (2014) Influencing factors of successful eradication of multidrug-resistant Acinetobacter baumannii in the respiratory tract with aerosolized colistin. Biomed J 37:314–320. [DOI] [PubMed] [Google Scholar]
- Cheng HY, Chen YF, Peng HL (2010) Molecular characterization of the PhoPQ-PmrD-PmrAB mediated pathway regulating polymyxin B resistance in Klebsiella pneumoniae CG43. J Biomed Sci 17:60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng YH, Lin TL, Lin YT, Wang JT (2016) Amino acid substitutions of CrrB responsible for resistance to colistin through CrrC in Klebsiella pneumoniae. Antimicrob Agents Chemother 60:3709–3716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheong HS, Kim SY, Wi YM, Peck KR, Ko KS (2019) Colistin heteroresistance in Klebsiella pneumoniae isolates and diverse mutations of PmrAB and PhoPQ in resistant subpopulations. J Clin Med 8:1444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chew KL, La MV, Lin RTP, Teo JWP (2017) Colistin and polymyxin B susceptibility testing for carbapenem-resistant and mcr-positive Enterobacteriaceae: comparison of Sensititre, Microscan, Vitek 2, and Etest with broth microdilution. J Clin Microbiol 55:2609–2616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chin CY, Gregg KA, Napier BA, Ernst RK, Weiss DS (2015) A PmrB-regulated deacetylase required for lipid A modification and polymyxin resistance in Acinetobacter baumannii. Antimicrob Agents Chemother 59:7911–7914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choi H, Kim K, Han J, Choi H, Jin SH, Lee EK, Shin DW, Lee TR, Lee AY, Noh M (2012) Kojic acid-induced IL-6 production in human keratinocytes plays a role in its anti-melanogenic activity in skin. J Dermatol Sci 66:207–215. [DOI] [PubMed] [Google Scholar]
- Choi HK, Kim YK, Kim HY, Uh Y (2014) Inhaled colistin for treatment of pneumonia due to colistin-only-susceptible Acinetobacter baumannii. Yonsei Med J 55:118–125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cirioni O, Ghiselli R, Silvestri C, Kamysz W, Orlando F, Mocchegiani F, Di Matteo F, Riva A, Lukasiak J, Scalise G, et al. (2007) Efficacy of tachyplesin III, colistin, and imipenem against a multiresistant Pseudomonas aeruginosa strain. Antimicrob Agents Chemother 51:2005–2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clancy CJ, Chen L, Hong JH, Cheng S, Hao B, Shields RK, Farrell AN, Doi Y, Zhao Y, Perlin DS, et al. (2013) Mutations of the ompK36 porin gene and promoter impact responses of sequence type 258, KPC-2-producing Klebsiella pneumoniae strains to doripenem and doripenem-colistin. Antimicrob Agents Chemother 57:5258–5265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clausell A, Garcia-Subirats M, Pujol M, Busquets MA, Rabanal F, Cajal Y (2007) Gram-negative outer and inner membrane models: insertion of cyclic cationic lipopeptides. J Phys Chem B 111:551–563. [DOI] [PubMed] [Google Scholar]
- Clements A, Tull D, Jenney AW, Farn JL, Kim SH, Bishop RE, McPhee JB, Hancock RE, Hartland EL, Pearse MJ, et al. (2007) Secondary acylation of Klebsiella pneumoniae lipopolysaccharide contributes to sensitivity to antibacterial peptides. J Biol Chem 282:15569–15577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clifton LA, Skoda MWA, Le Brun AP, Ciesielski F, Kuzmenko I, Holt SA, Lakey JH (2015) Effect of divalent cation removal on the structure of Gram-negative bacterial outer membrane models. Langmuir 31:404–412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clinical and Laboratory Standards Institute (2013) Performance Standards for Antimicrobial Susceptibility Testing, 23rd information supplement, Clinical and Laboratory Standards Institute, Wayne, PA. [Google Scholar]
- Clinical and Laboratory Standards Institute (2015) Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically, 10th ed, Clinical and Laboratory Standards Institute, Wayne, PA. [Google Scholar]
- Clinical and Laboratory Standards Institute (2018) Performance Standards for Antimicrobial Susceptibility Testing, 28th ed, Clinical and Laboratory Standards Institute, Wayne, PA. [Google Scholar]
- Clinical and Laboratory Standards Institute (2020) Performance Standards for Antimicrobial Susceptibility Testing, 30th ed, Clinical and Laboratory Standards Institute, Wayne, PA. [Google Scholar]
- Cockerill FR III, Muller SR, Anhalt JP, Marsh HM, Farnell MB, Mucha P, Gillespie DJ, Ilstrup DM, Larson-Keller JJ, Thompson RL (1992) Prevention of infection in critically ill patients by selective decontamination of the digestive tract. Ann Intern Med 117:545–553. [DOI] [PubMed] [Google Scholar]
- Conole D, Keating GM (2014) Colistimethate sodium dry powder for inhalation: a review of its use in the treatment of chronic Pseudomonas aeruginosa infection in patients with cystic fibrosis. Drugs 74:377–387. [DOI] [PubMed] [Google Scholar]
- Conway SP, Pond MN, Watson A, Etherington C, Robey HL, Goldman MH (1997) Intravenous colistin sulphomethate in acute respiratory exacerbations in adult patients with cystic fibrosis. Thorax 52:987–993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Córdoba J, Coronado-Álvarez NM, Parra D, Parra-Ruiz J (2015) In vitro activities of novel antimicrobial combinations against extensively drug-resistant Acinetobacter baumannii. Antimicrob Agents Chemother 59:7316–7319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corrigan JJ Jr., Kiernat JF (1979) Effect of polymyxin B sulfate on endotoxin activity in a Gram-negative septicemia model. Pediatr Res 13:48–51. [DOI] [PubMed] [Google Scholar]
- Corvec S, Furustrand Tafin U, Betrisey B, Borens O, Trampuz A (2013) Activities of fosfomycin, tigecycline, colistin, and gentamicin against extended-spectrum-β-lactamase-producing Escherichia coli in a foreign-body infection model. Antimicrob Agents Chemother 57:1421–1427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Couet W, Grégoire N, Gobin P, Saulnier PJ, Frasca D, Marchand S, Mimoz O (2011) Pharmacokinetics of colistin and colistimethate sodium after a single 80-mg intravenous dose of CMS in young healthy volunteers. Clin Pharmacol Ther 89:875–879. [DOI] [PubMed] [Google Scholar]
- Cruz DN, Antonelli M, Fumagalli R, Foltran F, Brienza N, Donati A, Malcangi V, Petrini F, Volta G, Bobbio Pallavicini FM, et al. (2009) Early use of polymyxin B hemoperfusion in abdominal septic shock: the EUPHAS randomized controlled trial. JAMA 301:2445–2452. [DOI] [PubMed] [Google Scholar]
- Cruz DN, Perazella MA, Bellomo R, de Cal M, Polanco N, Corradi V, Lentini P, Nalesso F, Ueno T, Ranieri VM, et al. (2007) Effectiveness of polymyxin B-immobilized fiber column in sepsis: a systematic review. Crit Care 11:R47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cunningham S, Prasad A, Collyer L, Carr S, Lynn IB, Wallis C (2001) Bronchoconstriction following nebulised colistin in cystic fibrosis. Arch Dis Child 84:432–433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dai C, Ciccotosto GD, Cappai R, Tang S, Li D, Xie S, Xiao X, Velkov T (2018a) Curcumin attenuates colistin-induced neurotoxicity in N2a cells via anti-inflammatory activity, suppression of oxidative stress, and apoptosis. Mol Neurobiol 55:421–434. [DOI] [PubMed] [Google Scholar]
- Dai C, Ciccotosto GD, Cappai R, Wang Y, Tang S, Hoyer D, Schneider EK, Velkov T, Xiao X (2018b) Rapamycin confers neuroprotection against colistin-induced oxidative stress, mitochondria dysfunction, and apoptosis through the activation of autophagy and mTOR/Akt/CREB signaling pathways. ACS Chem Neurosci 9:824–837. [DOI] [PubMed] [Google Scholar]
- Dai C, Li J, Tang S, Li J, Xiao X (2014) Colistin-induced nephrotoxicity in mice involves the mitochondrial, death receptor, and endoplasmic reticulum pathways. Antimicrob Agents Chemother 58:4075–4085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dai C, Tang S, Velkov T, Xiao X (2016) Colistin-induced apoptosis of neuroblastoma-2a cells involves the generation of reactive oxygen species, mitochondrial dysfunction, and autophagy. Mol Neurobiol 53:4685–4700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dai C, Xiao X, Li J, Ciccotosto GD, Cappai R, Tang S, Schneider-Futschik EK, Hoyer D, Velkov T, Shen J (2019) Molecular mechanisms of neurotoxicity induced by polymyxins and chemoprevention. ACS Chem Neurosci 10:120–131. [DOI] [PubMed] [Google Scholar]
- Dai C, Zhang D, Li J, Li J (2013) Effect of colistin exposure on calcium homeostasis and mitochondria functions in chick cortex neurons. Toxicol Mech Methods 23:281–288. [DOI] [PubMed] [Google Scholar]
- Dalfino L, Puntillo F, Mosca A, Monno R, Spada ML, Coppolecchia S, Miragliotta G, Bruno F, Brienza N (2012) High-dose, extended-interval colistin administration in critically ill patients: is this the right dosing strategy? A preliminary study. Clin Infect Dis 54:1720–1726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dalfino L, Puntillo F, Ondok MJM, Mosca A, Monno R, Coppolecchia S, Spada ML, Bruno F, Brienza N (2015) Colistin-associated acute kidney injury in severely ill patients: a step toward a better renal care? A prospective cohort study. Clin Infect Dis 61:1771–1777. [DOI] [PubMed] [Google Scholar]
- Dalgic N, Ceylan Y, Sancar M, Telhan L, Kafadar I, Cavusoglu H, Ceylan O, Hasim O (2009) Successful treatment of multidrug-resistant Acinetobacter baumannii ventriculitis with intravenous and intraventricular colistin. Ann Trop Paediatr 29:141–147. [DOI] [PubMed] [Google Scholar]
- D’amato RF, Thornsberry C, Baker CN, Kirven LA (1975) Effect of calcium and magnesium ions on the susceptibility of Pseudomonas species to tetracycline, gentamicin polymyxin B, and carbenicillin. Antimicrob Agents Chemother 7:596–600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dance DA, Wuthiekanun V, Naigowit P, White NJ (1989) Identification of Pseudomonas pseudomallei in clinical practice: use of simple screening tests and API 20NE. J Clin Pathol 42:645–648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daneman N, Sarwar S, Fowler RA, Cuthbertson BH, SuDDICU Canadian Study Group (2013) Effect of selective decontamination on antimicrobial resistance in intensive care units: a systematic review and meta-analysis. Lancet Infect Dis 13:328–341. [DOI] [PubMed] [Google Scholar]
- Dautrevaux M, Biserte G (1961) [Structure of colimycin]. Bull Soc Chim Biol (Paris) 43:495–504. [PubMed] [Google Scholar]
- Davis SD, Iannetta A, Wedgwood RJ (1971) Activity of colistin against Pseudomonas aeruginosa: inhibition by calcium. J Infect Dis 124:610–612. [DOI] [PubMed] [Google Scholar]
- Decker DA, Fincham RW (1971) Respiratory arrest in myasthenia gravis with colistimethate therapy. Arch Neurol 25:141–144. [DOI] [PubMed] [Google Scholar]
- Decolin D, Leroy P, Nicolas A, Archimbault P (1997) Hyphenated liquid chromatographic method for the determination of colistin residues in bovine tissues. J Chromatogr Sci 35:557–564. [DOI] [PubMed] [Google Scholar]
- de Jonge E, Schultz MJ, Spanjaard L, Bossuyt PMM, Vroom MB, Dankert J, Kesecioglu J (2003) Effects of selective decontamination of digestive tract on mortality and acquisition of resistant bacteria in intensive care: a randomised controlled trial. Lancet 362:1011–1016. [DOI] [PubMed] [Google Scholar]
- Dellinger RP, Bagshaw SM, Antonelli M, Foster DM, Klein DJ, Marshall JC, Palevsky PM, Weisberg LS, Schorr CA, Trzeciak S, et al. EUPHRATES Trial Investigators (2018) Effect of targeted polymyxin B hemoperfusion on 28-day mortality in patients with septic shock and elevated endotoxin level: the EUPHRATES randomized clinical trial. JAMA 320:1455–1463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Man TJB, Lutgring JD, Lonsway DR, Anderson KF, Kiehlbauch JA, Chen L, Walters MS, Sjölund-Karlsson M, Rasheed JK, Kallen A, et al. (2018) Genomic analysis of a pan-resistant isolate of Klebsiella pneumoniae, United States 2016. MBio 9:e00440-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Demiraslan H, Dinc G, Ahmed SS, Elmali F, Metan G, Alp E, Doganay M (2014) Carbapenem-resistant Klebsiella pneumoniae sepsis in corticosteroid receipt mice: tigecycline or colistin monotherapy versus tigecycline/colistin combination. J Chemother 26:276–281. [DOI] [PubMed] [Google Scholar]
- Deris ZZ, Akter J, Sivanesan S, Roberts KD, Thompson PE, Nation RL, Li J, Velkov T (2014a) A secondary mode of action of polymyxins against Gram-negative bacteria involves the inhibition of NADH-quinone oxidoreductase activity. J Antibiot (Tokyo) 67:147–151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deris ZZ, Swarbrick JD, Roberts KD, Azad MA, Akter J, Horne AS, Nation RL, Rogers KL, Thompson PE, Velkov T, et al. (2014b) Probing the penetration of antimicrobial polymyxin lipopeptides into Gram-negative bacteria. Bioconjug Chem 25:750–760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deris ZZ, Yu HH, Davis K, Soon RL, Jacob J, Ku CK, Poudyal A, Bergen PJ, Tsuji BT, Bulitta JB, et al. (2012) The combination of colistin and doripenem is synergistic against Klebsiella pneumoniae at multiple inocula and suppresses colistin resistance in an in vitro pharmacokinetic/pharmacodynamic model. Antimicrob Agents Chemother 56:5103–5112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Smet AMGA, Hopmans TEM, Minderhoud ALC, Blok HEM, Gossink-Franssen A, Bernards AT, Bonten MJM (2009a) Decontamination of the digestive tract and oropharynx: hospital acquired infections after discharge from the intensive care unit. Intensive Care Med 35:1609–1613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Smet AMGA, Kluytmans JAJW, Blok HEM, Mascini EM, Benus RFJ, Bernards AT, Kuijper EJ, Leverstein-van Hall MA, Jansz AR, de Jongh BM, et al. (2011) Selective digestive tract decontamination and selective oropharyngeal decontamination and antibiotic resistance in patients in intensive-care units: an open-label, clustered group-randomised, crossover study. Lancet Infect Dis 11:372–380. [DOI] [PubMed] [Google Scholar]
- de Smet AMGA, Kluytmans JAJW, Cooper BS, Mascini EM, Benus RFJ, van der Werf TS, van der Hoeven JG, Pickkers P, Bogaers-Hofman D, van der Meer NJM, et al. (2009b) Decontamination of the digestive tract and oropharynx in ICU patients. N Engl J Med 360:20–31. [DOI] [PubMed] [Google Scholar]
- Deveson Lucas D, Crane B, Wright A, Han ML, Moffatt J, Bulach D, Gladman SL, Powell D, Aranda J, Seemann T, et al. (2018) Emergence of high-level colistin resistance in an Acinetobacter baumannii clinical isolate mediated by inactivation of the global regulator H-NS. Antimicrob Agents Chemother 62:e02442-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Devlieger H, Casteels-Van Daele M, ki Muaka B, Proesmans W (1977) Acute renal failure due to colistin intoxication. Acta Paediatr Belg 30:179–181. [PubMed] [Google Scholar]
- Dezoti Fonseca C, Watanabe M, Vattimo MdeF (2012) Role of heme oxygenase-1 in polymyxin B-induced nephrotoxicity in rats. Antimicrob Agents Chemother 56:5082–5087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Di X, Wang R, Liu B, Zhang X, Ni W, Wang J, Liang B, Cai Y, Liu Y (2015) In vitro activity of fosfomycin in combination with colistin against clinical isolates of carbapenem-resistant Pseudomas aeruginosa. J Antibiot (Tokyo) 68:551–555. [DOI] [PubMed] [Google Scholar]
- Diep JK, Jacobs DM, Sharma R, Covelli J, Bowers DR, Russo TA, Rao GG (2017) Polymyxin B in combination with rifampin and meropenem against polymyxin B-resistant KPC-producing Klebsiella pneumoniae. Antimicrob Agents Chemother 61:e02121-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dodd ME, Abbott J, Maddison J, Moorcroft AJ, Webb AK (1997) Effect of tonicity of nebulised colistin on chest tightness and pulmonary function in adults with cystic fibrosis. Thorax 52:656–658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doern GV, Morse SA (1980) Branhamella (Neisseria) catarrhalis: criteria for laboratory identification. J Clin Microbiol 11:193–195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dortet L, Poirel L, Nordmann P (2014) Worldwide dissemination of the NDM-type carbapenemases in Gram-negative bacteria. BioMed Res Int 2014:249856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drehobl M, Guerrero JL, Lacarte PR, Goldstein G, Mata FS, Luber S (2008) Comparison of efficacy and safety of ciprofloxacin otic solution 0.2% versus polymyxin B-neomycin-hydrocortisone in the treatment of acute diffuse otitis externa. Curr Med Res Opin 24:3531–3542. [DOI] [PubMed] [Google Scholar]
- D’Souza BB, Padmaraj SR, Rekha PD, Tellis RC, Prabhu S, Pothen P (2014) In vitro synergistic activity of colistin and ceftazidime or ciprofloxacin against multidrug-resistant clinical strains of Pseudomonas aeruginosa. Microb Drug Resist 20:550–554. [DOI] [PubMed] [Google Scholar]
- Dubrovskaya Y, Chen TY, Scipione MR, Esaian D, Phillips MS, Papadopoulos J, Mehta SA (2013) Risk factors for treatment failure of polymyxin B monotherapy for carbapenem-resistant Klebsiella pneumoniae infections. Antimicrob Agents Chemother 57:5394–5397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duperthuy M, Binesse J, Le Roux F, Romestand B, Caro A, Got P, Givaudan A, Mazel D, Bachère E, Destoumieux-Garzón D (2010) The major outer membrane protein OmpU of Vibrio splendidus contributes to host antimicrobial peptide resistance and is required for virulence in the oyster Crassostrea gigas. Environ Microbiol 12:951–963. [DOI] [PubMed] [Google Scholar]
- Eadon MT, Hack BK, Alexander JJ, Xu C, Dolan ME, Cunningham PN (2013) Cell cycle arrest in a model of colistin nephrotoxicity. Physiol Genomics 45:877–888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eichhorn I, Feudi C, Wang Y, Kaspar H, Feßler AT, Lübke-Becker A, Michael GB, Shen J, Schwarz S (2018) Identification of novel variants of the colistin resistance gene mcr-3 in Aeromonas spp. from the national resistance monitoring programme GERM-Vet and from diagnostic submissions. J Antimicrob Chemother 73:1217–1221. [DOI] [PubMed] [Google Scholar]
- Elemam A, Rahimian J, Doymaz M (2010) In vitro evaluation of antibiotic synergy for polymyxin B-resistant carbapenemase-producing Klebsiella pneumoniae. J Clin Microbiol 48:3558–3562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elias LS, Konzen D, Krebs JM, Zavascki AP (2010) The impact of polymyxin B dosage on in-hospital mortality of patients treated with this antibiotic. J Antimicrob Chemother 65:2231–2237. [DOI] [PubMed] [Google Scholar]
- Enfanto C, Shin S (2007) Treatment with intrathecal colistin of a resistant extended-spectrum β-lactamase-producing Klebsiella pneumoniae in the cerebrospinal fluid. Infect Dis Clin Pract 15:274–277. [Google Scholar]
- Eshbach ML, Weisz OA (2017) Receptor-mediated endocytosis in the proximal tubule. Annu Rev Physiol 79:425–448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- European Committee on Antimicrobial Susceptibility Testing (2010) Breakpoint tables for interpretation of MICs and zone diameters. Version 1.0, European Society of Clinical Microbiology and Infectious Diseases, Basel, Switzerland. [Google Scholar]
- European Committee on Antimicrobial Susceptibility Testing (2021) Breakpoint tables for interpretation of MICs and zone diameters. Version 11.0, European Society of Clinical Microbiology and Infectious Diseases, Basel, Switzerland. [Google Scholar]
- European Pharmacopoeia Commission (2020) European Pharmacopoeia, Council of Europe, Strasbourg, France. [Google Scholar]
- Evans ME, Feola DJ, Rapp RP (1999) Polymyxin B sulfate and colistin: old antibiotics for emerging multiresistant Gram-negative bacteria. Ann Pharmacother 33:960–967. [DOI] [PubMed] [Google Scholar]
- Falagas ME, Bliziotis IA, Tam VH (2007) Intraventricular or intrathecal use of polymyxins in patients with Gram-negative meningitis: a systematic review of the available evidence. Int J Antimicrob Agents 29:9–25. [DOI] [PubMed] [Google Scholar]
- Falagas ME, Kasiakou SK (2005) Colistin: the revival of polymyxins for the management of multidrug-resistant Gram-negative bacterial infections [published correction appears in Clin Infect Dis (2006) 42:1819]. Clin Infect Dis 40:1333–1341. [DOI] [PubMed] [Google Scholar]
- Falagas ME, Kasiakou SK (2006) Toxicity of polymyxins: a systematic review of the evidence from old and recent studies. Crit Care 10:R27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Falagas ME, Rafailidis PI, Matthaiou DK, Virtzili S, Nikita D, Michalopoulos A (2008) Pandrug-resistant Klebsiella pneumoniae, Pseudomonas aeruginosa and Acinetobacter baumannii infections: characteristics and outcome in a series of 28 patients. Int J Antimicrob Agents 32:450–454. [DOI] [PubMed] [Google Scholar]
- Fechner GA, Jacobs JJ, Parsons PG (1993) Inhibition of melanogenesis in human melanoma cells by novel analogues of the partial histamine (H2) agonist nordimaprit. Biochem Pharmacol 46:47–54. [DOI] [PubMed] [Google Scholar]
- Fernández L, Gooderham WJ, Bains M, McPhee JB, Wiegand I, Hancock REW (2010) Adaptive resistance to the “last hope” antibiotics polymyxin B and colistin in Pseudomonas aeruginosa is mediated by the novel two-component regulatory system ParR-ParS. Antimicrob Agents Chemother 54:3372–3382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fernández L, Jenssen H, Bains M, Wiegand I, Gooderham WJ, Hancock RE (2012) The two-component system CprRS senses cationic peptides and triggers adaptive resistance in Pseudomonas aeruginosa independently of ParRS. Antimicrob Agents Chemother 56:6212–6222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fernandez-Viladrich P, Corbella X, Corral L, Tubau F, Mateu A (1999) Successful treatment of ventriculitis due to carbapenem-resistant Acinetobacter baumannii with intraventricular colistin sulfomethate sodium. Clin Infect Dis 28:916–917. [DOI] [PubMed] [Google Scholar]
- Finland M, Garner C, Wilcox C, Sabath LD (1976) Susceptibility of beta-hemolytic streptococci to 65 antibacterial agents. Antimicrob Agents Chemother 9:11–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flynn PM, Shenep JL, Stokes DC, Fairclough D, Hildner WK (1987) Polymyxin B moderates acidosis and hypotension in established, experimental Gram-negative septicemia. J Infect Dis 156:706–712. [DOI] [PubMed] [Google Scholar]
- Furtado GHC, d’Azevedo PA, Santos AF, Gales AC, Pignatari ACC, Medeiros EAS (2007) Intravenous polymyxin B for the treatment of nosocomial pneumonia caused by multidrug-resistant Pseudomonas aeruginosa. Int J Antimicrob Agents 30:315–319. [DOI] [PubMed] [Google Scholar]
- Gaibani P, Lombardo D, Lewis RE, Mercuri M, Bonora S, Landini MP, Ambretti S (2014) In vitro activity and post-antibiotic effects of colistin in combination with other antimicrobials against colistin-resistant KPC-producing Klebsiella pneumoniae bloodstream isolates. J Antimicrob Chemother 69:1856–1865. [DOI] [PubMed] [Google Scholar]
- Gales AC, Castanheira M, Jones RN, Sader HS (2012) Antimicrobial resistance among Gram-negative bacilli isolated from Latin America: results from SENTRY Antimicrobial Surveillance Program (Latin America, 2008-2010). Diagn Microbiol Infect Dis 73:354–360. [DOI] [PubMed] [Google Scholar]
- Gales AC, Jones RN, Sader HS (2006) Global assessment of the antimicrobial activity of polymyxin B against 54 731 clinical isolates of Gram-negative bacilli: report from the SENTRY antimicrobial surveillance programme (2001-2004). Clin Microbiol Infect 12:315–321. [DOI] [PubMed] [Google Scholar]
- Gales AC, Jones RN, Sader HS (2011) Contemporary activity of colistin and polymyxin B against a worldwide collection of Gram-negative pathogens: results from the SENTRY Antimicrobial Surveillance Program (2006-09). J Antimicrob Chemother 66:2070–2074. [DOI] [PubMed] [Google Scholar]
- Gales AC, Reis AO, Jones RN (2001) Contemporary assessment of antimicrobial susceptibility testing methods for polymyxin B and colistin: review of available interpretative criteria and quality control guidelines. J Clin Microbiol 39:183–190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- García-Rodríguez JA, García-García MI, García-Sánchez E, García-Sánchez JE, Muñoz Bellido JL (1989) [In vitro activity of 16 antimicrobial agents against Helicobacter (Campylobacter) pylori]. Enferm Infecc Microbiol Clin 7:544–546. [PubMed] [Google Scholar]
- Garnacho-Montero J, Ortiz-Leyba C, Jiménez-Jiménez FJ, Barrero-Almodóvar AE, García-Garmendia JL, Bernabeu-WittelI M, Gallego-Lara SL, Madrazo-Osuna J (2003) Treatment of multidrug-resistant Acinetobacter baumannii ventilator-associated pneumonia (VAP) with intravenous colistin: a comparison with imipenem-susceptible VAP. Clin Infect Dis 36:1111–1118. [DOI] [PubMed] [Google Scholar]
- Garonzik SM, Li J, Thamlikitkul V, Paterson DL, Shoham S, Jacob J, Silveira FP, Forrest A, Nation RL (2011) Population pharmacokinetics of colistin methanesulfonate and formed colistin in critically ill patients from a multicenter study provide dosing suggestions for various categories of patients. Antimicrob Agents Chemother 55:3284–3294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Genée E, Schlechtweg C, Bauerreiss P, Gibson JR (1982) Trimethoprim-polymyxin eye drops versus neomycin-polymyxin-gramicidin eye drops in the treatment of presumptive bacterial conjunctivitis - a double-blind study. Ophthalmologica 184:92–96. [DOI] [PubMed] [Google Scholar]
- Gibbons HS, Reynolds CM, Guan Z, Raetz CRH (2008) An inner membrane dioxygenase that generates the 2-hydroxymyristate moiety of Salmonella lipid A. Biochemistry 47:2814–2825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibson GA, Bauer SR, Neuner EA, Bass SN, Lam SW (2015) Influence of colistin dose on global cure in patients with bacteremia due to carbapenem-resistant Gram-negative bacilli. Antimicrob Agents Chemother 60:431–436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giddins MJ, Macesic N, Annavajhala MK, Stump S, Khan S, McConville TH, Mehta M, Gomez-Simmonds A, Uhlemann AC (2018) Successive emergence of ceftazidime-avibactam resistance through distinct genomic adaptations in bla(KPC-2)-harboring Klebsiella pneumoniae sequence type 307 isolates. Antimicrob Agents Chemother 62:e02101-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gigliotti F, Hendley JO, Morgan J, Michaels R, Dickens M, Lohr J (1984) Efficacy of topical antibiotic therapy in acute conjunctivitis in children. J Pediatr 104:623–626. [DOI] [PubMed] [Google Scholar]
- Giua R, Pedone C, Cortese L, Antonelli Incalzi R (2014) Colistin bladder instillation, an alternative way of treating multi-resistant Acinetobacter urinary tract infection: a case series and review of literature. Infection 42:199–202. [DOI] [PubMed] [Google Scholar]
- Gkoufa A, Karaiskos I, Routsi C, Friberg L, Sou T, Lin YW, Li J, Giamarellou H (2019) P-3296: Pulmonary and systemic pharmacokinetics of colistin methanesulfonate (CMS) and formed colistin following nebulization of CMS after a single inhalation dose of 3 MIU and 5 MIU in ICU patients with ventilator-associated pneumonia, in European Congress of Clinical Microbiology and Infectious Diseases, Viena, Austria. [Google Scholar]
- Glupczynski Y, Delmee M, Bruck C, Labbe M, Avesani V, Burette A (1988) Susceptibility of clinical isolates of Campylobacter pylori to 24 antimicrobial and anti-ulcer agents. Eur J Epidemiol 4:154–157. [DOI] [PubMed] [Google Scholar]
- Godard J, Guillaume C, Reverdy ME, Bachmann P, Bui-Xuan B, Nageotte A, Motin J (1990) Intestinal decontamination in a polyvalent ICU. A double-blind study. Intensive Care Med 16:307–311. [DOI] [PubMed] [Google Scholar]
- Gordeev MF (2018) Polymyxin soft drug MRX-8 with potential to address the class nephrotoxicity, in 3rd International Conference on Polymyxins, Madrid, Spain.
- Gordeev MF, Liu J, Wang Z, Yuan Z (2016) inventors, Micurx Pharmaceuticals Inc, assignee. Antimicrobial polymyxins for treatment of bacterial infections. Patent WO2016100578. 2016 Jun 23.
- Gordon NC, Png K, Wareham DW (2010) Potent synergy and sustained bactericidal activity of a vancomycin-colistin combination versus multidrug-resistant strains of Acinetobacter baumannii. Antimicrob Agents Chemother 54:5316–5322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gothwal S, Meena K, Sharma SD (2016) Polymyxin B induced generalized hyperpigmentation in neonates. Indian J Pediatr 83:179–180. [DOI] [PubMed] [Google Scholar]
- Goto M, Al-Hasan MN (2013) Overall burden of bloodstream infection and nosocomial bloodstream infection in North America and Europe. Clin Microbiol Infect 19:501–509. [DOI] [PubMed] [Google Scholar]
- Govaerts C, Orwa J, Van Schepdael A, Roets E, Hoogmartens J (2002a) Characterization of polypeptide antibiotics of the polymyxin series by liquid chromatography electrospray ionization ion trap tandem mass spectrometry. J Pept Sci 8:45–55. [DOI] [PubMed] [Google Scholar]
- Govaerts C, Orwa J, Van Schepdael A, Roets E, Hoogmartens J (2002b) Liquid chromatography-ion trap tandem mass spectrometry for the characterization of polypeptide antibiotics of the colistin series in commercial samples. J Chromatogr A 976:65–78. [DOI] [PubMed] [Google Scholar]
- Greenfield S, Feingold DS (1970) The synergistic action of the sulfonamides and the polymyxins against Serratia marcescens. J Infect Dis 121:555–558. [DOI] [PubMed] [Google Scholar]
- Grill MF, Maganti RK (2011) Neurotoxic effects associated with antibiotic use: management considerations. Br J Clin Pharmacol 72:381–393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gunderson BW, Ibrahim KH, Hovde LB, Fromm TL, Reed MD, Rotschafer JC (2003) Synergistic activity of colistin and ceftazidime against multiantibiotic-resistant Pseudomonas aeruginosa in an in vitro pharmacodynamic model. Antimicrob Agents Chemother 47:905–909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gunn JS, Ryan SS, Van Velkinburgh JC, Ernst RK, Miller SI (2000) Genetic and functional analysis of a PmrA-PmrB-regulated locus necessary for lipopolysaccharide modification, antimicrobial peptide resistance, and oral virulence of Salmonella enterica serovar typhimurium. Infect Immun 68:6139–6146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo W, Guo SC, Li M, Li LH, Qu Y (2018) Successful treatment of extensively drug-resistant Acinetobacter baumannii ventriculitis with polymyxin B and tigecycline- a case report. Antimicrob Resist Infect Control 7:22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gutu AD, Sgambati N, Strasbourger P, Brannon MK, Jacobs MA, Haugen E, Kaul RK, Johansen HK, Høiby N, Moskowitz SM (2013) Polymyxin resistance of Pseudomonas aeruginosa phoQ mutants is dependent on additional two-component regulatory systems. Antimicrob Agents Chemother 57:2204–2215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haeili M, Kafshdouz M, Pishnian Z, Feizabadi MM, Martinez-Martinez L (2019) Comparison of susceptibility testing methods for determining the activity of colistin against Gram-negative bacilli of clinical origin. J Med Microbiol 68:60–66. [DOI] [PubMed] [Google Scholar]
- Hagihara M, Housman ST, Nicolau DP, Kuti JL (2014) In vitro pharmacodynamics of polymyxin B and tigecycline alone and in combination against carbapenem-resistant Acinetobacter baumannii. Antimicrob Agents Chemother 58:874–879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halaby T, Al Naiemi N, Kluytmans J, van der Palen J, Vandenbroucke-Grauls CMJE (2013) Emergence of colistin resistance in Enterobacteriaceae after the introduction of selective digestive tract decontamination in an intensive care unit. Antimicrob Agents Chemother 57:3224–3229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han ML, Liu X, Velkov T, Lin YW, Zhu Y, Li M, Yu HH, Zhou Z, Creek DJ, Zhang J, et al. (2018) Metabolic analyses revealed time-dependent synergistic killing by colistin and aztreonam combination against multidrug-resistant Acinetobacter baumannii. Front Microbiol 9:2776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han ML, Zhu Y, Creek DJ, Lin YW, Gutu AD, Hertzog P, Purcell T, Shen HH, Moskowitz SM, Velkov T, et al. (2019) Comparative metabolomics and transcriptomics reveal multiple pathways associated with polymyxin killing in Pseudomonas aeruginosa. mSystems 4:e00149-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanasawa K, Tani T, Kodama M (1989) New approach to endotoxic and septic shock by means of polymyxin B immobilized fiber. Surg Gynecol Obstet 168:323–331. [PubMed] [Google Scholar]
- Hanasawa K, Tani T, Oka T, Yoshioka T, Aoki H, Endo Y, Kodama M (1988) Selective removal of endotoxin from the blood by extracorporeal hemoperfusion with polymyxin B immobilized fiber. Prog Clin Biol Res 264:337–341. [PubMed] [Google Scholar]
- Hancock RE (1997) Antibacterial peptides and the outer membranes of Gram-negative bacilli. J Med Microbiol 46:1–3. [DOI] [PubMed] [Google Scholar]
- Hankins JV, Madsen JA, Giles DK, Brodbelt JS, Trent MS (2012) Amino acid addition to Vibrio cholerae LPS establishes a link between surface remodeling in Gram-positive and Gram-negative bacteria. Proc Natl Acad Sci USA 109:8722–8727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hartzell JD, Neff R, Ake J, Howard R, Olson S, Paolino K, Vishnepolsky M, Weintrob A, Wortmann G (2009) Nephrotoxicity associated with intravenous colistin (colistimethate sodium) treatment at a tertiary care medical center. Clin Infect Dis 48:1724–1728. [DOI] [PubMed] [Google Scholar]
- Hausmann W, Craig LC (1954) Polymyxin-B1 - fractionation, molecular weight determination, amino acid and fatty acid composition. J Am Chem Soc 76:4892–4896. [Google Scholar]
- Hawley JS, Murray CK, Jorgensen JH (2008) Colistin heteroresistance in Acinetobacter and its association with previous colistin therapy. Antimicrob Agents Chemother 52:351–352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haworth CS, Foweraker JE, Wilkinson P, Kenyon RF, Bilton D (2014) Inhaled colistin in patients with bronchiectasis and chronic Pseudomonas aeruginosa infection. Am J Respir Crit Care Med 189:975–982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hawser SP (2010) Susceptibility of Klebsiella pneumoniae clinical isolates from 2007 to 2009 to colistin and comparator antibiotics. Int J Antimicrob Agents 36:383–384. [DOI] [PubMed] [Google Scholar]
- Hayashi K, Suketa Y, Tsukamoto K, Suzuki T (1966) Chemical structure of polymyxin D1. Experientia 22:354–355. [DOI] [PubMed] [Google Scholar]
- He H, Li JC, Nation RL, Jacob J, Chen G, Lee HJ, Tsuji BT, Thompson PE, Roberts K, Velkov T, et al. (2013) Pharmacokinetics of four different brands of colistimethate and formed colistin in rats. J Antimicrob Chemother 68:2311–2317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- He J, Ledesma KR, Lam WY, Figueroa DA, Lim TP, Chow DS, Tam VH (2010) Variability of polymyxin B major components in commercial formulations. Int J Antimicrob Agents 35:308–310. [DOI] [PubMed] [Google Scholar]
- Heindorf M, Kadari M, Heider C, Skiebe E, Wilharm G (2014) Impact of Acinetobacter baumannii superoxide dismutase on motility, virulence, oxidative stress resistance and susceptibility to antibiotics. PLoS One 9:e101033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heininger A, Meyer E, Schwab F, Marschal M, Unertl K, Krueger WA (2006) Effects of long-term routine use of selective digestive decontamination on antimicrobial resistance. Intensive Care Med 32:1569–1576. [DOI] [PubMed] [Google Scholar]
- Helander IM, Kilpeläinen I, Vaara M (1994) Increased substitution of phosphate groups in lipopolysaccharides and lipid A of the polymyxin-resistant pmrA mutants of Salmonella typhimurium: a 31P-NMR study. Mol Microbiol 11:481–487. [DOI] [PubMed] [Google Scholar]
- Hengzhuang W, Wu H, Ciofu O, Song Z, Høiby N (2011) Pharmacokinetics/pharmacodynamics of colistin and imipenem on mucoid and nonmucoid Pseudomonas aeruginosa biofilms. Antimicrob Agents Chemother 55:4469–4474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hengzhuang W, Wu H, Ciofu O, Song Z, Høiby N (2012) In vivo pharmacokinetics/pharmacodynamics of colistin and imipenem in Pseudomonas aeruginosa biofilm infection. Antimicrob Agents Chemother 56:2683–2690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henry R, Crane B, Powell D, Deveson Lucas D, Li Z, Aranda J, Harrison P, Nation RL, Adler B, Harper M, et al. (2015) The transcriptomic response of Acinetobacter baumannii to colistin and doripenem alone and in combination in an in vitro pharmacokinetics/pharmacodynamics model. J Antimicrob Chemother 70:1303–1313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henry R, Vithanage N, Harrison P, Seemann T, Coutts S, Moffatt JH, Nation RL, Li J, Harper M, Adler B, et al. (2012) Colistin-resistant, lipopolysaccharide-deficient Acinetobacter baumannii responds to lipopolysaccharide loss through increased expression of genes involved in the synthesis and transport of lipoproteins, phospholipids, and poly-β-1,6-N-acetylglucosamine. Antimicrob Agents Chemother 56:59–69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herrera CM, Hankins JV, Trent MS (2010) Activation of PmrA inhibits LpxT-dependent phosphorylation of lipid A promoting resistance to antimicrobial peptides. Mol Microbiol 76:1444–1460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hindler JA, Humphries RM (2013) Colistin MIC variability by method for contemporary clinical isolates of multidrug-resistant Gram-negative bacilli. J Clin Microbiol 51:1678–1684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ho YH, Wang LS, Chao HJ, Chang KC, Su CF (2007) Successful treatment of meningitis caused by multidrug-resistant Acinetobacter baumannii with intravenous and intrathecal colistin. J Microbiol Immunol Infect 40:537–540. [PubMed] [Google Scholar]
- Hogardt M, Schmoldt S, Götzfried M, Adler K, Heesemann J (2004) Pitfalls of polymyxin antimicrobial susceptibility testing of Pseudomonas aeruginosa isolated from cystic fibrosis patients. J Antimicrob Chemother 54:1057–1061. [DOI] [PubMed] [Google Scholar]
- Holmes KK (1964) Toxicity of colistin and polymixin B. N Engl J Med 271:633–634. [DOI] [PubMed] [Google Scholar]
- Hori Y, Aoki N, Kuwahara S, Hosojima M, Kaseda R, Goto S, Iida T, De S, Kabasawa H, Kaneko R, et al. (2017) Megalin blockade with cilastatin suppresses drug-induced nephrotoxicity. J Am Soc Nephrol 28:1783–1791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsieh TC, Chen FL, Ou TY, Jean SS, Lee WS (2016) Role of aerosolized colistin methanesulfonate therapy for extensively-drug-resistant Acinetobacter baumannii complex pneumonia and airway colonization. J Microbiol Immunol Infect 49:523–530. [DOI] [PubMed] [Google Scholar]
- Hurley JC, Guidet B, Offenstadt G, Maury E (2012) Endotoxemia and mortality prediction in ICU and other settings: underlying risk and co-detection of gram negative bacteremia are confounders. Crit Care 16:R148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hussein M, Han ML, Zhu Y, Zhou Q, Lin YW, Hancock REW, Hoyer D, Creek DJ, Li J, Velkov T (2019) Metabolomics study of the synergistic killing of polymyxin B in combination with amikacin against polymyxin-susceptible and -resistant Pseudomonas aeruginosa. Antimicrob Agents Chemother 64:e01587-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Imberti R, Cusato M, Accetta G, Marinò V, Procaccio F, Del Gaudio A, Iotti GA, Regazzi M (2012) Pharmacokinetics of colistin in cerebrospinal fluid after intraventricular administration of colistin methanesulfonate. Antimicrob Agents Chemother 56:4416–4421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Infectious Diseases Society of America (2004) Bad Bugs, No Drugs, Infectious Diseases Society of America,Arlington, VA. [Google Scholar]
- Ingoldby CJ (1980) The value of polymixin B in endotoxaemia due to experimental obstructive jaundice and mesenteric ischaemia. Br J Surg 67:565–567. [DOI] [PubMed] [Google Scholar]
- Ito-Kagawa M, Koyama Y (1980) Selective cleavage of a peptide antibiotic, colistin by colistinase. J Antibiot (Tokyo) 33:1551–1555. [DOI] [PubMed] [Google Scholar]
- Jacquemin E, Hagenbuch B, Stieger B, Wolkoff AW, Meier PJ (1994) Expression cloning of a rat liver Na(+)-independent organic anion transporter. Proc Natl Acad Sci USA 91:133–137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jasani B, Kreil G, Mackler BF, Stanworth DR (1979) Further studies on the structural requirements for polypeptide-mediated histamine release from rat mast cells. Biochem J 181:623–632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jayol A, Nordmann P, Brink A, Poirel L (2015) Heteroresistance to colistin in Klebsiella pneumoniae associated with alterations in the PhoPQ regulatory system. Antimicrob Agents Chemother 59:2780–2784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jayol A, Nordmann P, Lehours P, Poirel L, Dubois V (2018) Comparison of methods for detection of plasmid-mediated and chromosomally encoded colistin resistance in Enterobacteriaceae. Clin Microbiol Infect 24:175–179. [DOI] [PubMed] [Google Scholar]
- Jayol A, Poirel L, Brink A, Villegas MV, Yilmaz M, Nordmann P (2014) Resistance to colistin associated with a single amino acid change in protein PmrB among Klebsiella pneumoniae isolates of worldwide origin. Antimicrob Agents Chemother 58:4762–4766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jeannot K, Bolard A, Plésiat P (2017) Resistance to polymyxins in Gram-negative organisms. Int J Antimicrob Agents 49:526–535. [DOI] [PubMed] [Google Scholar]
- Jiang X, Yang K, Han ML, Yuan B, Li J, Gong B, Velkov T, Schreiber F, Wang L, Li J (2020a) Outer membranes of polymyxin-resistant Acinetobacter baumannii with phosphoethanolamine-modified lipid A and lipopolysaccharide loss display different atomic-scale interactions with polymyxins. ACS Infect Dis 6:2698–2708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang X, Yang K, Yuan B, Han M, Zhu Y, Roberts KD, Patil NA, Li J, Gong B, Hancock REW, et al. (2020b) Molecular dynamics simulations informed by membrane lipidomics reveal the structure-interaction relationship of polymyxins with the lipid A-based outer membrane of Acinetobacter baumannii. J Antimicrob Chemother 75:3534–3543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang X, Zhang S, Azad MAK, Roberts KD, Wan L, Gong B, Yang K, Yuan B, Uddin H, Li J, et al. (2020c) Structure-interaction relationship of polymyxins with the membrane of human kidney proximal tubular cells. ACS Infect Dis 6:2110–2119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin L, Li J, Nation RL, Nicolazzo JA (2009) Brain penetration of colistin in mice assessed by a novel high-performance liquid chromatographic technique. Antimicrob Agents Chemother 53:4247–4251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin L, Li J, Nation RL, Nicolazzo JA (2011) Impact of p-glycoprotein inhibition and lipopolysaccharide administration on blood-brain barrier transport of colistin in mice. Antimicrob Agents Chemother 55:502–507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin L, Li J, Nation RL, Nicolazzo JA (2012) Effect of systemic infection induced by Pseudomonas aeruginosa on the brain uptake of colistin in mice. Antimicrob Agents Chemother 56:5240–5246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin L, Nation RL, Li J, Nicolazzo JA (2013) Species-dependent blood-brain barrier disruption of lipopolysaccharide: amelioration by colistin in vitro and in vivo. Antimicrob Agents Chemother 57:4336–4342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johansen HK, Moskowitz SM, Ciofu O, Pressler T, Høiby N (2008) Spread of colistin resistant non-mucoid Pseudomonas aeruginosa among chronically infected Danish cystic fibrosis patients. J Cyst Fibros 7:391–397. [DOI] [PubMed] [Google Scholar]
- Jones RN, Guzman-Blanco M, Gales AC, Gallegos B, Castro AL, Martino MD, Vega S, Zurita J, Cepparulo M, Castanheira M (2013) Susceptibility rates in Latin American nations: report from a regional resistance surveillance program (2011). Braz J Infect Dis 17:672–681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones TS (1948) The chemical basis for the classification of the polymyxins. Biochem J 43:xxvi. [PubMed] [Google Scholar]
- Jones TS (1949) Chemical evidence for the multiplicity of the antibiotics produced by Bacillus polymyxa. Ann N Y Acad Sci 51:909–916. [DOI] [PubMed] [Google Scholar]
- Jung S, Chung EK, Jun MS, Son ES, Rhie SJ (2019) Differences in colistin administration and bacterial and treatment outcomes in critically ill patients. Sci Rep 9:8781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Justo JA, Bosso JA (2015) Adverse reactions associated with systemic polymyxin therapy. Pharmacotherapy 35:28–33. [DOI] [PubMed] [Google Scholar]
- Kallel H, Hergafi L, Bahloul M, Hakim A, Dammak H, Chelly H, Hamida CB, Chaari A, Rekik N, Bouaziz M (2007) Safety and efficacy of colistin compared with imipenem in the treatment of ventilator-associated pneumonia: a matched case-control study. Intensive Care Med 33:1162–1167. [DOI] [PubMed] [Google Scholar]
- Karabinis A, Paramythiotou E, Mylona-Petropoulou D, Kalogeromitros A, Katsarelis N, Kontopidou F, Poularas I, Malamou-Lada H (2004) Colistin for Klebsiella pneumoniae-associated sepsis. Clin Infect Dis 38:e7–e9. [DOI] [PubMed] [Google Scholar]
- Karaiskos I, Friberg LE, Pontikis K, Ioannidis K, Tsagkari V, Galani L, Kostakou E, Baziaka F, Paskalis C, Koutsoukou A, et al. (2015) Colistin population pharmacokinetics after application of a loading dose of 9 MU colistin methanesulfonate in critically ill patients. Antimicrob Agents Chemother 59:7240–7248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karaiskos I, Galani L, Baziaka F, Katsouda E, Ioannidis I, Andreou A, Paskalis H, Giamarellou H (2013) Successful treatment of extensively drug-resistant Acinetobacter baumannii ventriculitis and meningitis with intraventricular colistin after application of a loading dose: a case series. Int J Antimicrob Agents 41:480–483. [DOI] [PubMed] [Google Scholar]
- Karaiskos I, Lagou S, Pontikis K, Rapti V, Poulakou G (2019) The “old” and the “new” antibiotics for MDR Gram-negative pathogens: from whom, when, and how. Front Public Health 7:151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karvanen M, Malmberg C, Lagerbäck P, Friberg LE, Cars O (2017) Colistin is extensively lost during standard in vitro experimental conditions. Antimicrob Agents Chemother 61:e00857-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kasiakou SK, Fragoulis K, Tzagarakis G, Mistidis P, Kapaskelis A, Falagas ME (2005a) Cure of multidrug-resistant Acinetobacter baumannii fixation device-related orthopedic infections in two patients with intravenous colistin. Microb Drug Resist 11:287–289. [DOI] [PubMed] [Google Scholar]
- Kasiakou SK, Rafailidis PI, Liaropoulos K, Falagas ME (2005b) Cure of post-traumatic recurrent multiresistant Gram-negative rod meningitis with intraventricular colistin. J Infect 50:348–352. [DOI] [PubMed] [Google Scholar]
- Katsuma N, Sato Y, Ohki K, Okimura K, Ohnishi K, Sakura N (2009) Development of des-fatty acyl-polymyxin B decapeptide analogs with Pseudomonas aeruginosa-specific antimicrobial activity. Chem Pharm Bull (Tokyo) 57:332–336. [DOI] [PubMed] [Google Scholar]
- Kaye KS, Pogue JM, Tran TB, Nation RL, Li J (2016) Agents of last resort: polymyxin resistance. Infect Dis Clin North Am 30:391–414. [DOI] [PubMed] [Google Scholar]
- Keirstead ND, Wagoner MP, Bentley P, Blais M, Brown C, Cheatham L, Ciaccio P, Dragan Y, Ferguson D, Fikes J, et al. (2014) Early prediction of polymyxin-induced nephrotoxicity with next-generation urinary kidney injury biomarkers. Toxicol Sci 137:278–291. [DOI] [PubMed] [Google Scholar]
- Keith DD, Borders D, Curran W, Leese R, Mahamoon A, Jarolmen H, Francis N, Zhang Y, Arya A, Cotroneo N, et al. (2010) F1-1619: Synthesis and activity of CB-182,804, a novel polymyxin analog active against clinically relevant Gram-negative bacteria, in 50th Interscience Conference on Antimicrobial Agents and Chemotherapy; 12–15 September 2010; Boston, MA.
- Kerver AJ, Rommes JH, Mevissen-Verhage EA, Hulstaert PF, Vos A, Verhoef J, Wittebol P (1988) Prevention of colonization and infection in critically ill patients: a prospective randomized study. Crit Care Med 16:1087–1093. [DOI] [PubMed] [Google Scholar]
- Khawcharoenporn T, Apisarnthanarak A, Mundy LM (2010) Intrathecal colistin for drug-resistant Acinetobacter baumannii central nervous system infection: a case series and systematic review. Clin Microbiol Infect 16:888–894. [DOI] [PubMed] [Google Scholar]
- Kidd TJ, Mills G, Sá-Pessoa J, Dumigan A, Frank CG, Insua JL, Ingram R, Hobley L, Bengoechea JA (2017) A Klebsiella pneumoniae antibiotic resistance mechanism that subdues host defences and promotes virulence. EMBO Mol Med 9:430–447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kieffer N, Royer G, Decousser JW, Bourrel AS, Palmieri M, Ortiz De La Rosa JM, Jacquier H, Denamur E, Nordmann P, Poirel L (2019) mcr-9, an inducible gene encoding an acquired phosphoethanolamine transferase in Escherichia coli, and its origin. Antimicrob Agents Chemother 63:e00965-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kiehlbauch JA, Baker CN, Wachsmuth IK (1992) In vitro susceptibilities of aerotolerant Campylobacter isolates to 22 antimicrobial agents. Antimicrob Agents Chemother 36:717–722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim S, Patel DS, Park S, Slusky J, Klauda JB, Widmalm G, Im W (2016) Bilayer properties of lipid A from various Gram-negative bacteria. Biophys J 111:1750–1760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim SH, Jia W, Parreira VR, Bishop RE, Gyles CL (2006) Phosphoethanolamine substitution in the lipid A of Escherichia coli O157: H7 and its association with PmrC. Microbiology (Reading) 152:657–666. [DOI] [PubMed] [Google Scholar]
- Kimura Y, Kitamura H, Araki T, Noguchi K, Baba M, Hori M (1981) Analytical and preparative methods for polymyxin antibiotics using high-performance liquid-chromatography with a porous styrene-divinylbenzene co-polymer packing. J Chromatogr A 206:563–572. [Google Scholar]
- Ko KS, Suh JY, Kwon KT, Jung SI, Park KH, Kang CI, Chung DR, Peck KR, Song JH (2007) High rates of resistance to colistin and polymyxin B in subgroups of Acinetobacter baumannii isolates from Korea. J Antimicrob Chemother 60:1163–1167. [DOI] [PubMed] [Google Scholar]
- Koch-Weser J, Sidel VW, Federman EB, Kanarek P, Finer DC, Eaton AE (1970) Adverse effects of sodium colistimethate. Manifestations and specific reaction rates during 317 courses of therapy. Ann Intern Med 72:857–868. [DOI] [PubMed] [Google Scholar]
- Koeman M, van der Ven AJAM, Hak E, Joore HCA, Kaasjager K, de Smet AGA, Ramsay G, Dormans TPJ, Aarts LPHJ, de Bel EE, et al. (2006) Oral decontamination with chlorhexidine reduces the incidence of ventilator-associated pneumonia. Am J Respir Crit Care Med 173:1348–1355. [DOI] [PubMed] [Google Scholar]
- Kohanski MA, Dwyer DJ, Hayete B, Lawrence CA, Collins JJ (2007) A common mechanism of cellular death induced by bactericidal antibiotics. Cell 130:797–810. [DOI] [PubMed] [Google Scholar]
- Kosakai N, Oguri T (1976) [Distribution and changes of antibiotic susceptibility of genus Haemophilus (author’s transl)]. Jpn J Antibiot 29:159–166. [PubMed] [Google Scholar]
- Kox LF, Wösten MM, Groisman EA (2000) A small protein that mediates the activation of a two-component system by another two-component system. EMBO J 19:1861–1872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kroeger LA, Hovde LB, Mitropoulos IF, Schafer J, Rotschafer JC (2007) Colistin methanesulfonate against multidrug-resistant Acinetobacter baumannii in an in vitro pharmacodynamic model. Antimicrob Agents Chemother 51:3431–3433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krueger WA, Lenhart FP, Neeser G, Ruckdeschel G, Schreckhase H, Eissner HJ, Forst H, Eckart J, Peter K, Unertl KE (2002) Influence of combined intravenous and topical antibiotic prophylaxis on the incidence of infections, organ dysfunctions, and mortality in critically ill surgical patients: a prospective, stratified, randomized, double-blind, placebo-controlled clinical trial. Am J Respir Crit Care Med 166:1029–1037. [DOI] [PubMed] [Google Scholar]
- Ku YH, Chen CC, Lee MF, Chuang YC, Tang HJ, Yu WL (2017) Comparison of synergism between colistin, fosfomycin and tigecycline against extended-spectrum β-lactamase-producing Klebsiella pneumoniae isolates or with carbapenem resistance. J Microbiol Immunol Infect 50:931–939. [DOI] [PubMed] [Google Scholar]
- Kubin CJ, Ellman TM, Phadke V, Haynes LJ, Calfee DP, Yin MT (2012) Incidence and predictors of acute kidney injury associated with intravenous polymyxin B therapy. J Infect 65:80–87. [DOI] [PubMed] [Google Scholar]
- Kuck NA (1976) In vitro and in vivo activities of minocycline and other antibiotics against Acinetobacter (Herellea-Mima). Antimicrob Agents Chemother 9:493–497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuo SC, Lee YT, Yang SP, Chen CP, Chen TL, Hsieh SL, Siu LK, Fung CP (2012) Eradication of multidrug-resistant Acinetobacter baumannii from the respiratory tract with inhaled colistin methanesulfonate: a matched case-control study. Clin Microbiol Infect 18:870–876. [DOI] [PubMed] [Google Scholar]
- Kvitko CH, Rigatto MH, Moro AL, Zavascki AP (2011) Polymyxin B versus other antimicrobials for the treatment of pseudomonas aeruginosa bacteraemia. J Antimicrob Chemother 66:175–179. [DOI] [PubMed] [Google Scholar]
- Kwa A, Kasiakou SK, Tam VH, Falagas ME (2007) Polymyxin B: similarities to and differences from colistin (polymyxin E). Expert Rev Anti Infect Ther 5:811–821. [DOI] [PubMed] [Google Scholar]
- Kwa AL, Abdelraouf K, Low JG, Tam VH (2011) Pharmacokinetics of polymyxin B in a patient with renal insufficiency: a case report. Clin Infect Dis 52:1280–1281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwa AL, Loh C, Low JG, Kurup A, Tam VH (2005) Nebulized colistin in the treatment of pneumonia due to multidrug-resistant Acinetobacter baumannii and Pseudomonas aeruginosa. Clin Infect Dis 41:754–757. [DOI] [PubMed] [Google Scholar]
- Lagerbäck P, Khine WWT, Giske CG, Tängdén T (2016) Evaluation of antibacterial activities of colistin, rifampicin and meropenem combinations against NDM-1-producing Klebsiella pneumoniae in 24 h in vitro time-kill experiments. J Antimicrob Chemother 71:2321–2325. [DOI] [PubMed] [Google Scholar]
- Lahiry S, Choudhury S, Mukherjee A, Bhunya PK, Bala M (2017) Polymyxin B-induced diffuse cutaneous hyperpigmentation. J Clin Diagn Res 11:FD01–FD02. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lakota EA, Landersdorfer CB, Nation RL, Li J, Kaye KS, Rao GG, Forrest A (2018) Personalizing polymyxin B dosing using an adaptive feedback control algorithm. Antimicrob Agents Chemother 62:e00483-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Landersdorfer CB, Wang J, Wirth V, Chen K, Kaye KS, Tsuji BT, Li J, Nation RL (2018) Pharmacokinetics/pharmacodynamics of systemically administered polymyxin B against Klebsiella pneumoniae in mouse thigh and lung infection models. J Antimicrob Chemother 73:462–468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Landman D, Bratu S, Alam M, Quale J (2005) Citywide emergence of Pseudomonas aeruginosa strains with reduced susceptibility to polymyxin B. J Antimicrob Chemother 55:954–957. [DOI] [PubMed] [Google Scholar]
- Landman D, Salamera J, Quale J (2013) Irreproducible and uninterpretable polymyxin B MICs for Enterobacter cloacae and Enterobacter aerogenes. J Clin Microbiol 51:4106–4111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Le Brun PP, de Boer AH, Mannes GP, de Fraîture DM, Brimicombe RW, Touw DJ, Vinks AA, Frijlink HW, Heijerman HG (2002) Dry powder inhalation of antibiotics in cystic fibrosis therapy: part 2. Inhalation of a novel colistin dry powder formulation: a feasibility study in healthy volunteers and patients. Eur J Pharm Biopharm 54:25–32. [DOI] [PubMed] [Google Scholar]
- Ledson MJ, Gallagher MJ, Cowperthwaite C, Convery RP, Walshaw MJ (1998) Four years’ experience of intravenous colomycin in an adult cystic fibrosis unit. Eur Respir J 12:592–594. [DOI] [PubMed] [Google Scholar]
- Lee GC, Burgess DS (2013) Polymyxins and doripenem combination against KPC-producing Klebsiella pneumoniae. J Clin Med Res 5:97–100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee H, Hsu FF, Turk J, Groisman EA (2004) The PmrA-regulated pmrC gene mediates phosphoethanolamine modification of lipid A and polymyxin resistance in Salmonella enterica. J Bacteriol 186:4124–4133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee HJ, Bergen PJ, Bulitta JB, Tsuji B, Forrest A, Nation RL, Li J (2013a) Synergistic activity of colistin and rifampin combination against multidrug-resistant Acinetobacter baumannii in an in vitro pharmacokinetic/pharmacodynamic model. Antimicrob Agents Chemother 57:3738–3745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee SY, Shin JH, Lee K, Joo MY, Park KH, Shin MG, Suh SP, Ryang DW, Kim SH (2013b) Comparison of the Vitek 2, MicroScan, and Etest methods with the agar dilution method in assessing colistin susceptibility of bloodstream isolates of Acinetobacter species from a Korean university hospital. J Clin Microbiol 51:1924–1926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leese RA (2010) inventors, BioSource Pharm Inc, assignee. Antibiotic composition for the treatment of Gram-negative infections. U.S. patent 2010/0160215 A1. 2010 Jun 24.
- Leite GC, Oliveira MS, Perdigão-Neto LV, Rocha CKD, Guimarães T, Rizek C, Levin AS, Costa SF (2016) Antimicrobial combinations against pan-resistant Acinetobacter baumannii isolates with different resistance mechanisms. PLoS One 11:e0151270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lenhard JR, Smith NM, Bulman ZP, Tao X, Thamlikitkul V, Shin BS, Nation RL, Li J, Bulitta JB, Tsuji BT (2017a) High-dose ampicillin-sulbactam combinations combat polymyxin-resistant Acinetobacter baumannii in a hollow-fiber infection model. Antimicrob Agents Chemother 61:e01268-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lenhard JR, Thamlikitkul V, Silveira FP, Garonzik SM, Tao X, Forrest A, Soo Shin B, Kaye KS, Bulitta JB, Nation RL, et al. (2017b) Polymyxin-resistant, carbapenem-resistant Acinetobacter baumannii is eradicated by a triple combination of agents that lack individual activity. J Antimicrob Chemother 72:1415–1420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leong KW, Ong S, Chee HL, Lee W, Kwa AL (2010) Hypersensitivity pneumonitis due to high-dose colistin aerosol therapy. Int J Infect Dis 14:e1018–e1019. [DOI] [PubMed] [Google Scholar]
- Lépine PA, Dumas A, Boulet LP (2017) Pulmonary eosinophilia from inhaled colistin. Chest 151:e1–e3. [DOI] [PubMed] [Google Scholar]
- Lesmana M, Subekti DS, Tjaniadi P, Simanjuntak CH, Punjabi NH, Campbell JR, Oyofo BA (2002) Spectrum of Vibrio species associated with acute diarrhea in North Jakarta, Indonesia. Diagn Microbiol Infect Dis 43:91–97. [DOI] [PubMed] [Google Scholar]
- Li J, Coulthard K, Milne R, Nation RL, Conway S, Peckham D, Etherington C, Turnidge J (2003a) Steady-state pharmacokinetics of intravenous colistin methanesulphonate in patients with cystic fibrosis. J Antimicrob Chemother 52:987–992. [DOI] [PubMed] [Google Scholar]
- Li J, Coulthard K, Nation RL (2019) Labelling conventions and product package insert of parenteral polymyxins: factors causing potential medication errors and impeding optimal clinical use. Adv Exp Med Biol 1145:133–141. [DOI] [PubMed] [Google Scholar]
- Li J, Milne RW, Nation RL, Turnidge JD, Smeaton TC, Coulthard K (2003b) Use of high-performance liquid chromatography to study the pharmacokinetics of colistin sulfate in rats following intravenous administration. Antimicrob Agents Chemother 47:1766–1770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li J, Milne RW, Nation RL, Turnidge JD, Smeaton TC, Coulthard K (2004) Pharmacokinetics of colistin methanesulphonate and colistin in rats following an intravenous dose of colistin methanesulphonate. J Antimicrob Chemother 53:837–840. [DOI] [PubMed] [Google Scholar]
- Li J, Nation RL, Milne RW, Turnidge JD, Coulthard K (2005a) Evaluation of colistin as an agent against multi-resistant Gram-negative bacteria. Int J Antimicrob Agents 25:11–25. [DOI] [PubMed] [Google Scholar]
- Li J, Nation RL, Owen RJ, Wong S, Spelman D, Franklin C (2007) Antibiograms of multidrug-resistant clinical Acinetobacter baumannii: promising therapeutic options for treatment of infection with colistin-resistant strains. Clin Infect Dis 45:594–598. [DOI] [PubMed] [Google Scholar]
- Li J, Nation RL, Turnidge JD (2006a) Defining the dosage units for colistin methanesulfonate: urgent need for international harmonization. Antimicrob Agents Chemother 50:4231–4232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li J, Nation RL, Turnidge JD, Milne RW, Coulthard K, Rayner CR, Paterson DL (2006b) Colistin: the re-emerging antibiotic for multidrug-resistant Gram-negative bacterial infections. Lancet Infect Dis 6:589–601. [DOI] [PubMed] [Google Scholar]
- Li J, Rayner CR, Nation RL, Deans R, Boots R, Widdecombe N, Douglas A, Lipman J (2005b) Pharmacokinetics of colistin methanesulfonate and colistin in a critically ill patient receiving continuous venovenous hemodiafiltration. Antimicrob Agents Chemother 49:4814–4815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li J, Rayner CR, Nation RL, Owen RJ, Spelman D, Tan KE, Liolios L (2006c) Heteroresistance to colistin in multidrug-resistant Acinetobacter baumannii. Antimicrob Agents Chemother 50:2946–2950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li J, Turnidge J, Milne R, Nation RL, Coulthard K (2001) In vitro pharmacodynamic properties of colistin and colistin methanesulfonate against Pseudomonas aeruginosa isolates from patients with cystic fibrosis. Antimicrob Agents Chemother 45:781–785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li M, Aye SM, Ahmed MU, Han ML, Li C, Song J, Boyce JD, Powell DR, Azad MAK, Velkov T, et al. (2020a) Pan-transcriptomic analysis identified common differentially expressed genes of Acinetobacter baumannii in response to polymyxin treatments. Mol Omics 16:327–338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li YM, Milikowski C, Selvaggi G, Abbo LM, Skiada D, Galimberti F (2020b) Polymyxin B-induced skin hyperpigmentation. Transpl Infect Dis 22:e13312. [DOI] [PubMed] [Google Scholar]
- Lightbown JW, Thomas AH, Grab B, Outschoorn AS (1973) The second international standard for polymyxin B. Bull World Health Organ 48:85–90. [PMC free article] [PubMed] [Google Scholar]
- Lim TP, Tan TY, Lee W, Sasikala S, Tan TT, Hsu LY, Kwa AL (2009) In vitro activity of various combinations of antimicrobials against carbapenem-resistant Acinetobacter species in Singapore. J Antibiot (Tokyo) 62:675–679. [DOI] [PubMed] [Google Scholar]
- Lim TP, Tan TY, Lee W, Sasikala S, Tan TT, Hsu LY, Kwa AL (2011) In-vitro activity of polymyxin B, rifampicin, tigecycline alone and in combination against carbapenem-resistant Acinetobacter baumannii in Singapore. PLoS One 6:e18485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin CC, Liu TC, Kuo CF, Liu CP, Lee CM (2010) Aerosolized colistin for the treatment of multidrug-resistant Acinetobacter baumannii pneumonia: experience in a tertiary care hospital in northern Taiwan. J Microbiol Immunol Infect 43:323–331. [DOI] [PubMed] [Google Scholar]
- Lin J, Xu C, Fang R, Cao J, Zhang X, Zhao Y, Dong G, Sun Y, Zhou T (2019a) Resistance and heteroresistance to colistin in Pseudomonas aeruginosa isolates from Wenzhou, China. Antimicrob Agents Chemother 63:e00556-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin QY, Tsai YL, Liu MC, Lin WC, Hsueh PR, Liaw SJ (2014) Serratia marcescens arn, a PhoP-regulated locus necessary for polymyxin B resistance. Antimicrob Agents Chemother 58:5181–5190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin YW, Abdul Rahim N, Zhao J, Han ML, Yu HH, Wickremasinghe H, Chen K, Wang J, Paterson DL, Zhu Y, et al. (2019b) Novel polymyxin combination with the antiretroviral zidovudine exerts synergistic killing against NDM-producing multidrug-resistant Klebsiella pneumoniae. Antimicrob Agents Chemother 63:e02176-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin YW, Han ML, Zhao J, Zhu Y, Rao G, Forrest A, Song J, Kaye KS, Hertzog P, Purcell A, et al. (2019c) Synergistic combination of polymyxin B and enrofloxacin induced metabolic perturbations in extensive drug-resistant Pseudomonas aeruginosa. Front Pharmacol 10:1146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin YW, Zhou Q, Onufrak NJ, Wirth V, Chen K, Wang J, Forrest A, Chan HK, Li J (2017a) Aerosolized polymyxin B for treatment of respiratory tract infections: determination of pharmacokinetic-pharmacodynamic indices for aerosolized polymyxin B against Pseudomonas aeruginosa in a mouse lung infection model. Antimicrob Agents Chemother 61:e00211–e00217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin YW, Zhou QT, Cheah SE, Zhao J, Chen K, Wang J, Chan HK, Li J (2017b) Pharmacokinetics/Pharmacodynamics of pulmonary delivery of colistin against Pseudomonas aeruginosa in a mouse lung infection model. Antimicrob Agents Chemother 61:e02025-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin YW, Zhou QT, Han ML, Chen K, Onufrak NJ, Wang J, Turnidge JD, Howden BP, Forrest A, Chan HK, et al. (2018) Elucidating the pharmacokinetics/pharmacodynamics of aerosolized colistin against multidrug-resistant Acinetobacter baumannii and Klebsiella pneumoniae in a mouse lung infection model. Antimicrob Agents Chemother 62:e01790-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin YW, Zhou QT, Hu Y, Onufrak NJ, Sun S, Wang J, Forrest A, Chan HK, Li J (2017c) Pulmonary pharmacokinetics of colistin following administration of dry powder aerosols in rats. Antimicrob Agents Chemother 61:e00973-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu X, Zhao M, Chen Y, Bian X, Li Y, Shi J, Zhang J (2016a) Synergistic killing by meropenem and colistin combination of carbapenem-resistant Acinetobacter baumannii isolates from Chinese patients in an in vitro pharmacokinetic/pharmacodynamic model. Int J Antimicrob Agents 48:559–563. [DOI] [PubMed] [Google Scholar]
- Liu YY, Wang Y, Walsh TR, Yi LX, Zhang R, Spencer J, Doi Y, Tian G, Dong B, Huang X, et al. (2016b) Emergence of plasmid-mediated colistin resistance mechanism MCR-1 in animals and human beings in China: a microbiological and molecular biological study. Lancet Infect Dis 16:161–168. [DOI] [PubMed] [Google Scholar]
- Llobet E, Tomás JM, Bengoechea JA (2008) Capsule polysaccharide is a bacterial decoy for antimicrobial peptides. Microbiology (Reading) 154:3877–3886. [DOI] [PubMed] [Google Scholar]
- Lora-Tamayo J, Murillo O, Bergen PJ, Nation RL, Poudyal A, Luo X, Yu HY, Ariza J, Li J (2014) Activity of colistin combined with doripenem at clinically relevant concentrations against multidrug-resistant Pseudomonas aeruginosa in an in vitro dynamic biofilm model. J Antimicrob Chemother 69:2434–2442. [DOI] [PubMed] [Google Scholar]
- Lo-Ten-Foe JR, de Smet AMGA, Diederen BMW, Kluytmans JAJW, van Keulen PHJ (2007) Comparative evaluation of the VITEK 2, disk diffusion, etest, broth microdilution, and agar dilution susceptibility testing methods for colistin in clinical isolates, including heteroresistant Enterobacter cloacae and Acinetobacter baumannii strains. Antimicrob Agents Chemother 51:3726–3730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu Q, Luo R, Bodin L, Yang J, Zahr N, Aubry A, Golmard JL, Rouby JJ, Nebulized Antibiotics Study Group (2013) Efficacy of high-dose nebulized colistin in ventilator-associated pneumonia caused by multidrug-resistant Pseudomonas aeruginosa and Acinetobacter baumannii. Anesthesiology 57:222–223. [DOI] [PubMed] [Google Scholar]
- Lu X, Chan T, Xu C, Zhu L, Zhou QT, Roberts KD, Chan HK, Li J, Zhou F (2016) Human oligopeptide transporter 2 (PEPT2) mediates cellular uptake of polymyxins. J Antimicrob Chemother 71:403–412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu Z, Jiang G, Chen Y, Wang J, Muhammad I, Zhang L, Wang R, Liu F, Li R, Qian F, et al. (2017) Salidroside attenuates colistin-induced neurotoxicity in RSC96 Schwann cells through PI3K/Akt pathway. Chem Biol Interact 271:67–78. [DOI] [PubMed] [Google Scholar]
- Lübbert C, Faucheux S, Becker-Rux D, Laudi S, Dürrbeck A, Busch T, Gastmeier P, Eckmanns T, Rodloff AC, Kaisers UX (2013) Rapid emergence of secondary resistance to gentamicin and colistin following selective digestive decontamination in patients with KPC-2-producing Klebsiella pneumoniae: a single-centre experience. Int J Antimicrob Agents 42:565–570. [DOI] [PubMed] [Google Scholar]
- Luiten EJ, Hop WC, Lange JF, Bruining HA (1995) Controlled clinical trial of selective decontamination for the treatment of severe acute pancreatitis. Ann Surg 222:57–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lund MH (1969) Colistin sulfate ophthalmic in the treatment of ocular infections. Arch Ophthalmol 81:4–10. [DOI] [PubMed] [Google Scholar]
- Ly NS, Bulitta JB, Rao GG, Landersdorfer CB, Holden PN, Forrest A, Bergen PJ, Nation RL, Li J, Tsuji BT (2015) Colistin and doripenem combinations against Pseudomonas aeruginosa: profiling the time course of synergistic killing and prevention of resistance. J Antimicrob Chemother 70:1434–1442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma Z, Wang J, Nation RL, Li J, Turnidge JD, Coulthard K, Milne RW (2009) Renal disposition of colistin in the isolated perfused rat kidney. Antimicrob Agents Chemother 53:2857–2864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maalej SM, Meziou MR, Rhimi FM, Hammami A (2011) Comparison of disc diffusion, Etest and agar dilution for susceptibility testing of colistin against Enterobacteriaceae. Lett Appl Microbiol 53:546–551. [DOI] [PubMed] [Google Scholar]
- Macedo SK, Gonçalves IP, Bispo G, Almeida L, Brito L (2011) Intrathecal (intraventricular) polymyxin B in the treatment of patients with meningoencephalitis by Acinetobacter baumannii and Pseudomonas aeruginosa. Crit Care 15:235.22216838 [Google Scholar]
- Macfarlane EL, Kwasnicka A, Ochs MM, Hancock RE (1999) PhoP-PhoQ homologues in Pseudomonas aeruginosa regulate expression of the outer-membrane protein OprH and polymyxin B resistance. Mol Microbiol 34:305–316. [DOI] [PubMed] [Google Scholar]
- Maichuk YF (1991) Dexamethasone in the complex treatment of Chlamydial conjunctivitis. Rev Int Trach Pathol Ocul Trop Subtrop Sante Publique 68:83–93. [PubMed] [Google Scholar]
- Maifiah MHM, Creek DJ, Nation RL, Forrest A, Tsuji BT, Velkov T, Li J (2017) Untargeted metabolomics analysis reveals key pathways responsible for the synergistic killing of colistin and doripenem combination against Acinetobacter baumannii. Sci Rep 7:45527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Makris D, Petinaki E, Tsolaki V, Manoulakas E, Mantzarlis K, Apostolopoulou O, Sfyras D, Zakynthinos E (2018) Colistin versus colistin combined with ampicillin-sulbactam for multiresistant Acinetobacter baumannii ventilator-associated pneumonia treatment: an open-label prospective study. Indian J Crit Care Med 22:67–77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malinverni JC, Silhavy TJ (2009) An ABC transport system that maintains lipid asymmetry in the Gram-negative outer membrane. Proc Natl Acad Sci USA 106:8009–8014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manchandani P, Dubrovskaya Y, Gao S, Tam VH (2016) Comparative pharmacokinetic profiling of different polymyxin B components. Antimicrob Agents Chemother 60:6980–6982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manchandani P, Thamlikitkul V, Dubrovskaya Y, Babic JT, Lye DC, Lee LS, Tam VH (2018) Population pharmacokinetics of polymyxin B. Clin Pharmacol Ther 104:534–538. [DOI] [PubMed] [Google Scholar]
- Manchandani P, Zhou J, Babic JT, Ledesma KR, Truong LD, Tam VH (2017) Role of renal drug exposure in polymyxin B-induced nephrotoxicity. Antimicrob Agents Chemother 61:e02391-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manchandani P, Zhou J, Ledesma KR, Truong LD, Chow DS, Eriksen JL, Tam VH (2015) Characterization of polymyxin B biodistribution and disposition in an animal model. Antimicrob Agents Chemother 60:1029–1034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marchand S, Grégoire N, Couet W (2019) Pharmacokinetics of polymyxins in animals. Adv Exp Med Biol 1145:89–103. [DOI] [PubMed] [Google Scholar]
- Markou N, Fousteri M, Markantonis SL, Zidianakis B, Hroni D, Boutzouka E, Baltopoulos G (2012) Colistin pharmacokinetics in intensive care unit patients on continuous venovenous haemodiafiltration: an observational study. J Antimicrob Chemother 67:2459–2462. [DOI] [PubMed] [Google Scholar]
- Markou N, Markantonis SL, Dimitrakis E, Panidis D, Boutzouka E, Karatzas S, Rafailidis P, Apostolakos H, Baltopoulos G (2008) Colistin serum concentrations after intravenous administration in critically ill patients with serious multidrug-resistant, Gram-negative bacilli infections: a prospective, open-label, uncontrolled study. Clin Ther 30:143–151. [DOI] [PubMed] [Google Scholar]
- Marone G, Granata F, Spadaro G, Genovese A, Triggiani M (2003) The histamine-cytokine network in allergic inflammation. J Allergy Clin Immunol 112(4 Suppl):S83–S88. [DOI] [PubMed] [Google Scholar]
- Marschke G, Sarauw A (1971) Polymyxin inhalation therapeutic hazard. Ann Intern Med 74:144–145. [DOI] [PubMed] [Google Scholar]
- Maskin LP, Setten M, Rodríguez PO, Bonelli I, Attie S, Stryjewski ME, Valentini R (2015) Inhaled colistimethate sodium in ventilator-associated tracheobronchitis due to multidrug-resistant Gram-negative bacteria. Int J Antimicrob Agents 45:199–200. [DOI] [PubMed] [Google Scholar]
- Mathur J, Davis BM, Waldor MK (2007) Antimicrobial peptides activate the Vibrio cholerae sigmaE regulon through an OmpU-dependent signalling pathway. Mol Microbiol 63:848–858. [DOI] [PubMed] [Google Scholar]
- Mathur J, Waldor MK (2004) The Vibrio cholerae ToxR-regulated porin OmpU confers resistance to antimicrobial peptides. Infect Immun 72:3577–3583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsen JM, Koepcke MJ, Quie PG (1969) Evaluation of the Bauer-Kirby-Sherris-Turck single-disc diffusion method of antibiotic susceptibility testing. Antimicrob Agents Chemother 9:445–453. [PubMed] [Google Scholar]
- Mattos KP, Lloret GR, Cintra ML, Gouvêa IR, Betoni TR, Mazzola PG, Moriel P (2016) Acquired skin hyperpigmentation following intravenous polymyxin B treatment: a cohort study. Pigment Cell Melanoma Res 29:388–390. [DOI] [PubMed] [Google Scholar]
- Mattos KPH, Cintra ML, Gouvêa IR, Ferreira LÁ, Velho PENF, Moriel P (2017) Skin hyperpigmentation following intravenous polymyxin B treatment associated with melanocyte activation and inflammatory process. J Clin Pharm Ther 42:573–578. [DOI] [PubMed] [Google Scholar]
- Matuschek E, Åhman J, Webster C, Kahlmeter G (2018) Antimicrobial susceptibility testing of colistin - evaluation of seven commercial MIC products against standard broth microdilution for Escherichia coli, Klebsiella pneumoniae, Pseudomonas aeruginosa, and Acinetobacter spp. Clin Microbiol Infect 24:865–870. [DOI] [PubMed] [Google Scholar]
- McConville TH, Annavajhala MK, Giddins MJ, Macesic N, Herrera CM, Rozenberg FD, Bhushan GL, Ahn D, Mancia F, Trent MS, et al. (2020) CrrB positively regulates high-level polymyxin resistance and virulence in Klebsiella pneumoniae. Cell Rep 33:108313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCoy KS (2007) Compounded colistimethate as possible cause of fatal acute respiratory distress syndrome. N Engl J Med 357:2310–2311. [DOI] [PubMed] [Google Scholar]
- Meletis G, Tzampaz E, Sianou E, Tzavaras I, Sofianou D (2011) Colistin heteroresistance in carbapenemase-producing Klebsiella pneumoniae. J Antimicrob Chemother 66:946–947. [DOI] [PubMed] [Google Scholar]
- Melsen WG, de Smet AMGA, Kluytmans JAJW, Bonten MJM, Dutch SOD-SDD Trialists’ Group (2012) Selective decontamination of the oral and digestive tract in surgical versus non-surgical patients in intensive care in a cluster-randomized trial. Br J Surg 99:232–237. [DOI] [PubMed] [Google Scholar]
- Meredith JJ, Dufour A, Bruch MD (2009) Comparison of the structure and dynamics of the antibiotic peptide polymyxin B and the inactive nonapeptide in aqueous trifluoroethanol by NMR spectroscopy. J Phys Chem B 113:544–551. [DOI] [PubMed] [Google Scholar]
- Mezzatesta ML, Gona F, Caio C, Petrolito V, Sciortino D, Sciacca A, Santangelo C, Stefani S (2011) Outbreak of KPC-3-producing, and colistin-resistant, Klebsiella pneumoniae infections in two Sicilian hospitals. Clin Microbiol Infect 17:1444–1447. [DOI] [PubMed] [Google Scholar]
- Michalopoulos A, Virtzili S, Rafailidis P, Chalevelakis G, Damala M, Falagas ME (2010) Intravenous fosfomycin for the treatment of nosocomial infections caused by carbapenem-resistant Klebsiella pneumoniae in critically ill patients: a prospective evaluation. Clin Microbiol Infect 16:184–186. [DOI] [PubMed] [Google Scholar]
- Miglis C, Rhodes NJ, Avedissian SN, Kubin CJ, Yin MT, Nelson BC, Pai MP, Scheetz MH (2018) Population pharmacokinetics of polymyxin B in acutely ill adult patients. Antimicrob Agents Chemother 62:e01475-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Min KL, Son ES, Kim JS, Kim SH, Jung SM, Chang MJ (2018) Risk factors of colistin safety according to administration routes: intravenous and aerosolized colistin. PLoS One 13:e0207588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mingeot-Leclercq MP, Tulkens PM, Denamur S, Vaara T, Vaara M (2012) Novel polymyxin derivatives are less cytotoxic than polymyxin B to renal proximal tubular cells. Peptides 35:248–252. [DOI] [PubMed] [Google Scholar]
- Miori L, Vignini M, Rabbiosi G (1990) Flagellate dermatitis after bleomycin. A histological and immunohistochemical study. Am J Dermatopathol 12:598–602. [DOI] [PubMed] [Google Scholar]
- Miura T, Mizushima S (1968) Separation by density gradient centrifugation of two types of membranes from spheroplast membrane of Escherichia coli K12. Biochim Biophys Acta 150:159–161. [DOI] [PubMed] [Google Scholar]
- Mizuyachi K, Hara K, Wakamatsu A, Nohda S, Hirama T (2011) Safety and pharmacokinetic evaluation of intravenous colistin methanesulfonate sodium in Japanese healthy male subjects. Curr Med Res Opin 27:2261–2270. [DOI] [PubMed] [Google Scholar]
- Moffatt JH, Harper M, Adler B, Nation RL, Li J, Boyce JD (2011) Insertion sequence ISAba11 is involved in colistin resistance and loss of lipopolysaccharide in Acinetobacter baumannii. Antimicrob Agents Chemother 55:3022–3024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moffatt JH, Harper M, Harrison P, Hale JDF, Vinogradov E, Seemann T, Henry R, Crane B, St Michael F, Cox AD, et al. (2010) Colistin resistance in Acinetobacter baumannii is mediated by complete loss of lipopolysaccharide production. Antimicrob Agents Chemother 54:4971–4977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mogi T, Murase Y, Mori M, Shiomi K, Omura S, Paranagama MP, Kita K (2009) Polymyxin B identified as an inhibitor of alternative NADH dehydrogenase and malate: quinone oxidoreductase from the Gram-positive bacterium Mycobacterium smegmatis. J Biochem 146:491–499. [DOI] [PubMed] [Google Scholar]
- Mohamed AF, Karaiskos I, Plachouras D, Karvanen M, Pontikis K, Jansson B, Papadomichelakis E, Antoniadou A, Giamarellou H, Armaganidis A, et al. (2012) Application of a loading dose of colistin methanesulfonate in critically ill patients: population pharmacokinetics, protein binding, and prediction of bacterial kill. Antimicrob Agents Chemother 56:4241–4249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moland ES, Craft DW, Hong SG, Kim SY, Hachmeister L, Sayed SD, Thomson KS (2008) In vitro activity of tigecycline against multidrug-resistant Acinetobacter baumannii and selection of tigecycline-amikacin synergy. Antimicrob Agents Chemother 52:2940–2942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morrissey I, Olesky M, Hawser S, Lob SH, Karlowsky JA, Corey GR, Bassetti M, Fyfe C (2020) In vitro activity of eravacycline against Gram-negative bacilli isolated in clinical laboratories worldwide from 2013 to 2017. Antimicrob Agents Chemother 64:e01699-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morrison DC, Jacobs DM (1976) Binding of polymyxin B to the lipid A portion of bacterial lipopolysaccharides. Immunochemistry 13:813–818. [DOI] [PubMed] [Google Scholar]
- Moskowitz SM, Ernst RK, Miller SI (2004) PmrAB, a two-component regulatory system of Pseudomonas aeruginosa that modulates resistance to cationic antimicrobial peptides and addition of aminoarabinose to lipid A. J Bacteriol 186:575–579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moskowitz SM, Garber E, Chen Y, Clock SA, Tabibi S, Miller AK, Doctor M, Saiman L (2010) Colistin susceptibility testing: evaluation of reliability for cystic fibrosis isolates of Pseudomonas aeruginosa and Stenotrophomonas maltophilia. J Antimicrob Chemother 65:1416–1423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moskowitz SM, Silva SJ, Mayer-Hamblett N, Pasta DJ, Mink DR, Mabie JA, Konstan MW, Wagener JS, Investigators and Coordinators of the Epidemiologic Study of Cystic Fibrosis (ESCF) (2008) Shifting patterns of inhaled antibiotic use in cystic fibrosis. Pediatr Pulmonol 43:874–881. [DOI] [PubMed] [Google Scholar]
- Mostardeiro MM, Pereira CAP, Marra AR, Pestana JOM, Camargo LFA (2013) Nephrotoxicity and efficacy assessment of polymyxin use in 92 transplant patients. Antimicrob Agents Chemother 57:1442–1446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Motaouakkil S, Charra B, Hachimi A, Nejmi H, Benslama A, Elmdaghri N, Belabbes H, Benbachir M (2006) Colistin and rifampicin in the treatment of nosocomial infections from multiresistant Acinetobacter baumannii. J Infect 53:274–278. [DOI] [PubMed] [Google Scholar]
- Murray WR (1984) Use of antibiotic-containing bone cement. Clin Orthop Relat Res:89–95. [PubMed] [Google Scholar]
- Mutlu Yilmaz E, Sunbul M, Aksoy A, Yilmaz H, Guney AK, Guvenc T (2012) Efficacy of tigecycline/colistin combination in a pneumonia model caused by extensively drug-resistant Acinetobacter baumannii. Int J Antimicrob Agents 40:332–336. [DOI] [PubMed] [Google Scholar]
- Muyembe T, Vandepitte J, Desmyter J (1973) Natural colistin resistance in Edwardsiella tarda. Antimicrob Agents Chemother 4:521–524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakamura T, Komatsu M, Yamasaki K, Fukuda S, Higuchi T, Ono T, Nishio H, Sueyoshi N, Kida K, Satoh K, et al. (2014) Susceptibility of various oral antibacterial agents against extended spectrum β-lactamase producing Escherichia coli and Klebsiella pneumoniae. J Infect Chemother 20:48–51. [DOI] [PubMed] [Google Scholar]
- Nang SC, Li J, Velkov T (2019) The rise and spread of mcr plasmid-mediated polymyxin resistance. Crit Rev Microbiol 45:131–161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nastro M, Rodríguez CH, Monge R, Zintgraff J, Neira L, Rebollo M, Vay C, Famiglietti A (2014) Activity of the colistin-rifampicin combination against colistin-resistant, carbapenemase-producing Gram-negative bacteria. J Chemother 26:211–216. [DOI] [PubMed] [Google Scholar]
- Nation RL, Forrest A (2019) Clinical pharmacokinetics, pharmacodynamics and toxicodynamics of polymyxins: implications for therapeutic use. Adv Exp Med Biol 1145:219–249. [DOI] [PubMed] [Google Scholar]
- Nation RL, Garonzik SM, Thamlikitkul V, Giamarellos-Bourboulis EJ, Forrest A, Paterson DL, Li J, Silveira FP (2017) Dosing guidance for intravenous colistin in critically-ill patients. Clin Infect Dis 64:565–571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nation RL, Li J, Cars O, Couet W, Dudley MN, Kaye KS, Mouton JW, Paterson DL, Tam VH, Theuretzbacher U, et al. (2014) Consistent global approach on reporting of colistin doses to promote safe and effective use. Clin Infect Dis 58:139–141. [DOI] [PubMed] [Google Scholar]
- National Committee for Clinical Laboratory Standards (1976) Performance Standards for Antimicrobial Disk Susceptibility Tests, 1st ed, National Committee for Clinical Laboratory Standards, Wayne, PA. [Google Scholar]
- National Committee for Clinical Laboratory Standards (1980) Performance Standards for Antimicrobial Susceptibility Testing, 1st ed, National Committee for Clinical Laboratory Standards, Wayne, PA. [Google Scholar]
- Newton BA (1954) Site of action of polymyxin on Pseudomonas aeruginosa: antagonism by cations. J Gen Microbiol 10:491–499. [DOI] [PubMed] [Google Scholar]
- Ng TM, Teng CB, Lye DC, Apisarnthanarak A (2014) A multicenter case-case control study for risk factors and outcomes of extensively drug-resistant Acinetobacter baumannii bacteremia. Infect Control Hosp Epidemiol 35:49–55. [DOI] [PubMed] [Google Scholar]
- Nielsen SL (2015) The incidence and prognosis of patients with bacteremia. Dan Med J 62:B5128. [PubMed] [Google Scholar]
- Nikaido H (2003) Molecular basis of bacterial outer membrane permeability revisited. Microbiol Mol Biol Rev 67:593–656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niks M, Hanzen J, Ohlasová D, Rovná D, Purgelová A, Szövényiová Z, Vaculíková A (2004) [Multiresistant nosocomial bacterial strains and their “in vitro” susceptibility to chloramphenicol and colistin]. Klin Mikrobiol Infekc Lek 10:124–129. [PubMed] [Google Scholar]
- Nilsson A, Goodwin RJA, Swales JG, Gallagher R, Shankaran H, Sathe A, Pradeepan S, Xue A, Keirstead N, Sasaki JC, et al. (2015) Investigating nephrotoxicity of polymyxin derivatives by mapping renal distribution using mass spectrometry imaging. Chem Res Toxicol 28:1823–1830. [DOI] [PubMed] [Google Scholar]
- Niu B, Vater J, Rueckert C, Blom J, Lehmann M, Ru JJ, Chen XH, Wang Q, Borriss R (2013) Polymyxin P is the active principle in suppressing phytopathogenic Erwinia spp. by the biocontrol rhizobacterium Paenibacillus polymyxa M-1. BMC Microbiol 13:137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nord NM, Hoeprich PD (1964) Polymyxin B and colistin. A critical comparison. N Engl J Med 270:1030–1035. [DOI] [PubMed] [Google Scholar]
- Nowicki EM, O’Brien JP, Brodbelt JS, Trent MS (2015) Extracellular zinc induces phosphoethanolamine addition to Pseudomonas aeruginosa lipid A via the ColRS two-component system. Mol Microbiol 97:166–178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nummila K, Kilpeläinen I, Zähringer U, Vaara M, Helander IM (1995) Lipopolysaccharides of polymyxin B-resistant mutants of Escherichia coli are extensively substituted by 2-aminoethyl pyrophosphate and contain aminoarabinose in lipid A. Mol Microbiol 16:271–278. [DOI] [PubMed] [Google Scholar]
- O’Neil J (2016) Tackling drug-resistant infections globally: final report and recommendations, Review on Antimicrobial Resistance, London.
- Ochoa-Ardila ME, García-Cañas A, Gómez-Mediavilla K, González-Torralba A, Alía I, García-Hierro P, Taylor N, van Saene HKF, de la Cal MA (2011) Long-term use of selective decontamination of the digestive tract does not increase antibiotic resistance: a 5-year prospective cohort study. Intensive Care Med 37:1458–1465. [DOI] [PubMed] [Google Scholar]
- Okuda M, Saito H, Urakami Y, Takano M, Inui K (1996) cDNA cloning and functional expression of a novel rat kidney organic cation transporter, OCT2. Biochem Biophys Res Commun 224:500–507. [DOI] [PubMed] [Google Scholar]
- Olaitan AO, Morand S, Rolain JM (2014) Mechanisms of polymyxin resistance: acquired and intrinsic resistance in bacteria. Front Microbiol 5:643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oleksiuk LM, Nguyen MH, Press EG, Updike CL, O’Hara JA, Doi Y, Clancy CJ, Shields RK (2014) In vitro responses of Acinetobacter baumannii to two- and three-drug combinations following exposure to colistin and doripenem. Antimicrob Agents Chemother 58:1195–1199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oostdijk EAN, de Smet AMGA, Kesecioglu J, Bonten MJM, Dutch SOD-SDD Trialists Group (2011) The role of intestinal colonization with Gram-negative bacteria as a source for intensive care unit-acquired bacteremia. Crit Care Med 39:961–966. [DOI] [PubMed] [Google Scholar]
- Orwa JA, Govaerts C, Busson R, Roets E, Van Schepdael A, Hoogmartens J (2001a) Isolation and structural characterization of colistin components. J Antibiot (Tokyo) 54:595–599. [DOI] [PubMed] [Google Scholar]
- Orwa JA, Govaerts C, Busson R, Roets E, Van Schepdael A, Hoogmartens J (2001b) Isolation and structural characterization of polymyxin B components. J Chromatogr A 912:369–373. [DOI] [PubMed] [Google Scholar]
- Owen RJ, Li J, Nation RL, Spelman D (2007) In vitro pharmacodynamics of colistin against Acinetobacter baumannii clinical isolates. J Antimicrob Chemother 59:473–477. [DOI] [PubMed] [Google Scholar]
- Ozdemir H, Tapisiz A, Ciftçi E, Ince E, Mokhtari H, Güriz H, Aysev AD, Doğru U (2010) Successful treatment of three children with post-neurosurgical multidrug-resistant Acinetobacter baumannii meningitis. Infection 38:241–244. [DOI] [PubMed] [Google Scholar]
- Ozkan G, Ulusoy S, Orem A, Alkanat M, Mungan S, Yulug E, Yucesan FB (2013) How does colistin-induced nephropathy develop and can it be treated? Antimicrob Agents Chemother 57:3463–3469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ozyilmaz E, Ebinc FA, Derici U, Gulbahar O, Goktas G, Elmas C, Oguzulgen IK, Sindel S (2011) Could nephrotoxicity due to colistin be ameliorated with the use of N-acetylcysteine? Intensive Care Med 37:141–146. [DOI] [PubMed] [Google Scholar]
- Pamp SJ, Gjermansen M, Johansen HK, Tolker-Nielsen T (2008) Tolerance to the antimicrobial peptide colistin in Pseudomonas aeruginosa biofilms is linked to metabolically active cells, and depends on the pmr and mexAB-oprM genes. Mol Microbiol 68:223–240. [DOI] [PubMed] [Google Scholar]
- Pankuch GA, Seifert H, Appelbaum PC (2010) Activity of doripenem with and without levofloxacin, amikacin, and colistin against Pseudomonas aeruginosa and Acinetobacter baumannii. Diagn Microbiol Infect Dis 67:191–197. [DOI] [PubMed] [Google Scholar]
- Pantopoulou A, Giamarellos-Bourboulis EJ, Raftogannis M, Tsaganos T, Dontas I, Koutoukas P, Baziaka F, Giamarellou H, Perrea D (2007) Colistin offers prolonged survival in experimental infection by multidrug-resistant Acinetobacter baumannii: the significance of co-administration of rifampicin. Int J Antimicrob Agents 29:51–55. [DOI] [PubMed] [Google Scholar]
- Papagelopoulos PJ, Mavrogenis AF, Giannitsioti E, Kikilas A, Kanellakopoulou K, Soucacos PN (2007) Management of a multidrug-resistant Pseudomonas aeruginosa infected total knee arthroplasty using colistin. A case report and review of the literature. J Arthroplasty 22:457–463. [DOI] [PubMed] [Google Scholar]
- Park GC, Choi JA, Jang SJ, Jeong SH, Kim CM, Choi IS, Kang SH, Park G, Moon DS (2016) In vitro interactions of antibiotic combinations of colistin, tigecycline, and doripenem against extensively drug-resistant and multidrug-resistant Acinetobacter baumannii. Ann Lab Med 36:124–130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park SY, Si HJ, Eom JS, Lee JS (2019) Survival of carbapenem-resistant Acinetobacter baumannii bacteremia: colistin monotherapy versus colistin plus meropenem. J Int Med Res 47:5977–5985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parker WL, Rathnum ML, Dean LD, Nimeck MW, Brown WE, Meyers E (1977) Polymyxin E, a new peptide antibiotic. J Antibiot (Tokyo) 30:767–769. [DOI] [PubMed] [Google Scholar]
- Pastewski AA, Caruso P, Parris AR, Dizon R, Kopec R, Sharma S, Mayer S, Ghitan M, Chapnick EK (2008) Parenteral polymyxin B use in patients with multidrug-resistant Gram-negative bacteremia and urinary tract infections: a retrospective case series. Ann Pharmacother 42:1177–1187. [DOI] [PubMed] [Google Scholar]
- Paul M, Daikos GL, Durante-Mangoni E, Yahav D, Carmeli Y, Benattar YD, Skiada A, Andini R, Eliakim-Raz N, Nutman A, et al. (2018) Colistin alone versus colistin plus meropenem for treatment of severe infections caused by carbapenem-resistant Gram-negative bacteria: an open-label, randomised controlled trial. Lancet Infect Dis 18:391–400. [DOI] [PubMed] [Google Scholar]
- Payen DM, Guilhot J, Launey Y, Lukaszewicz AC, Kaaki M, Veber B, Pottecher J, Joannes-Boyau O, Martin-Lefevre L, Jabaudon M, et al. ABDOMIX Group (2015) Early use of polymyxin B hemoperfusion in patients with septic shock due to peritonitis: a multicenter randomized control trial. Intensive Care Med 41:975–984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pearman JW, Bailey M, Harper WE (1988) Comparison of the efficacy of “Trisdine” and kanamycin-colistin bladder instillations in reducing bacteriuria during intermittent catheterisation of patients with acute spinal cord trauma. Br J Urol 62:140–144. [DOI] [PubMed] [Google Scholar]
- Percin D, Akyol S, Kalin G (2014) In vitro synergism of combinations of colistin with selected antibiotics against colistin-resistant Acinetobacter baumannii. GMS Hyg Infect Control 9:Doc14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pereira GH, Muller PR, Levin AS (2007) Salvage treatment of pneumonia and initial treatment of tracheobronchitis caused by multidrug-resistant Gram-negative bacilli with inhaled polymyxin B. Diagn Microbiol Infect Dis 58:235–240. [DOI] [PubMed] [Google Scholar]
- Peterson JW (1996) Bacterial pathogenesis, in Medical Microbiology (Baron S ed) 4th ed, University of Texas Medical Branch, Galveston, TX. [PubMed] [Google Scholar]
- Piparsania S, Rajput N, Bhatambare G (2012) Intraventricular polymyxin B for the treatment of neonatal meningo-ventriculitis caused by multi-resistant Acinetobacter baumannii--case report and review of literature. Turk J Pediatr 54:548–554. [PubMed] [Google Scholar]
- Plachouras D, Giamarellos-Bourboulis EJ, Kentepozidis N, Baziaka F, Karagianni V, Giamarellou H (2007) In vitro postantibiotic effect of colistin on multidrug-resistant Acinetobacter baumannii. Diagn Microbiol Infect Dis 57:419–422. [DOI] [PubMed] [Google Scholar]
- Plachouras D, Karvanen M, Friberg LE, Papadomichelakis E, Antoniadou A, Tsangaris I, Karaiskos I, Poulakou G, Kontopidou F, Armaganidis A, et al. (2009) Population pharmacokinetic analysis of colistin methanesulfonate and colistin after intravenous administration in critically ill patients with infections caused by Gram-negative bacteria. Antimicrob Agents Chemother 53:3430–3436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pneumatikos I, Koulouras V, Nathanail C, Goe D, Nakos G (2002) Selective decontamination of subglottic area in mechanically ventilated patients with multiple trauma. Intensive Care Med 28:432–437. [DOI] [PubMed] [Google Scholar]
- Pogue JM, Jones RN, Bradley JS, Andes DR, Bhavnani SM, Drusano GL, Dudley MN, Flamm RK, Rodvold KA, Ambrose PG (2020) Polymyxin susceptibility testing and interpretive breakpoints: recommendations from the United States Committee on Antimicrobial Susceptibility Testing (USCAST). Antimicrob Agents Chemother 64:e01495-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pogue JM, Tam VH (2019) Toxicity in patients. Adv Exp Med Biol 1145:289–304. [DOI] [PubMed] [Google Scholar]
- Poirel L, Jayol A, Bontron S, Villegas MV, Ozdamar M, Türkoglu S, Nordmann P (2015) The mgrB gene as a key target for acquired resistance to colistin in Klebsiella pneumoniae. J Antimicrob Chemother 70:75–80. [DOI] [PubMed] [Google Scholar]
- Poudyal A, Howden BP, Bell JM, Gao W, Owen RJ, Turnidge JD, Nation RL, Li J (2008) In vitro pharmacodynamics of colistin against multidrug-resistant Klebsiella pneumoniae. J Antimicrob Chemother 62:1311–1318. [DOI] [PubMed] [Google Scholar]
- Pournaras S, Vrioni G, Neou E, Dendrinos J, Dimitroulia E, Poulou A, Tsakris A (2011) Activity of tigecycline alone and in combination with colistin and meropenem against Klebsiella pneumoniae carbapenemase (KPC)-producing Enterobacteriaceae strains by time-kill assay. Int J Antimicrob Agents 37:244–247. [DOI] [PubMed] [Google Scholar]
- Price DJ, Graham DI (1970) Effects of large doses of colistin sulphomethate sodium on renal function. BMJ 4:525–527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pugin J, Auckenthaler R, Lew DP, Suter PM (1991) Oropharyngeal decontamination decreases incidence of ventilator-associated pneumonia. A randomized, placebo-controlled, double-blind clinical trial. JAMA 265:2704–2710. [PubMed] [Google Scholar]
- Queenan AM, Pillar CM, Deane J, Sahm DF, Lynch AS, Flamm RK, Peterson J, Davies TA (2012) Multidrug resistance among Acinetobacter spp. in the USA and activity profile of key agents: results from CAPITAL Surveillance 2010. Diagn Microbiol Infect Dis 73:267–270. [DOI] [PubMed] [Google Scholar]
- Quinn AL, Parada JP, Belmares J, O’Keefe JP (2005) Intrathecal colistin and sterilization of resistant Pseudomonas aeruginosa shunt infection. Ann Pharmacother 39:949–952. [DOI] [PubMed] [Google Scholar]
- Qureshi ZA, Hittle LE, O’Hara JA, Rivera JI, Syed A, Shields RK, Pasculle AW, Ernst RK, Doi Y (2015) Colistin-resistant Acinetobacter baumannii: beyond carbapenem resistance. Clin Infect Dis 60:1295–1303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qureshi ZA, Paterson DL, Potoski BA, Kilayko MC, Sandovsky G, Sordillo E, Polsky B, Adams-Haduch JM, Doi Y (2012) Treatment outcome of bacteremia due to KPC-producing Klebsiella pneumoniae: superiority of combination antimicrobial regimens. Antimicrob Agents Chemother 56:2108–2113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rahman A, Rizwan S, Waycaster C, Wall GM (2007) Pooled analysis of two clinical trials comparing the clinical outcomes of topical ciprofloxacin/dexamethasone otic suspension and polymyxin B/neomycin/hydrocortisone otic suspension for the treatment of acute otitis externa in adults and children. Clin Ther 29:1950–1956. [DOI] [PubMed] [Google Scholar]
- Rattanaumpawan P, Lorsutthitham J, Ungprasert P, Angkasekwinai N, Thamlikitkul V (2010) Randomized controlled trial of nebulized colistimethate sodium as adjunctive therapy of ventilator-associated pneumonia caused by Gram-negative bacteria. J Antimicrob Chemother 65:2645–2649. [DOI] [PubMed] [Google Scholar]
- Reychler G, Keyeux A, Cremers C, Veriter C, Rodenstein DO, Liistro G (2004) Comparison of lung deposition in two types of nebulization: intrapulmonary percussive ventilation vs jet nebulization. Chest 125:502–508. [DOI] [PubMed] [Google Scholar]
- Reynolds CM, Kalb SR, Cotter RJ, Raetz CR (2005) A phosphoethanolamine transferase specific for the outer 3-deoxy-D-manno-octulosonic acid residue of Escherichia coli lipopolysaccharide. Identification of the eptB gene and Ca2+ hypersensitivity of an eptB deletion mutant. J Biol Chem 280:21202–21211. [DOI] [PubMed] [Google Scholar]
- Ribera A, Benavent E, Lora-Tamayo J, Tubau F, Pedrero S, Cabo X, Ariza J, Murillo O (2015) Osteoarticular infection caused by MDR Pseudomonas aeruginosa: the benefits of combination therapy with colistin plus β-lactams. J Antimicrob Chemother 70:3357–3365. [DOI] [PubMed] [Google Scholar]
- Rigatto MH, Ribeiro VB, Konzen D, Zavascki AP (2013) Comparison of polymyxin B with other antimicrobials in the treatment of ventilator-associated pneumonia and tracheobronchitis caused by Pseudomonas aeruginosa or Acinetobacter baumannii. Infection 41:321–328. [DOI] [PubMed] [Google Scholar]
- Rios FG, Luna CM, Maskin B, Saenz Valiente A, Lloria M, Gando S, Sosa C, Baquero S, Llerena C, Petrati C, et al. (2007) Ventilator-associated pneumonia due to colistin susceptible-only microorganisms. Eur Respir J 30:307–313. [DOI] [PubMed] [Google Scholar]
- Roberts KD, Azad MAK, Wang J, Horne AS, Thompson PE, Nation RL, Velkov T, Li J (2015) Antimicrobial activity and toxicity of the major lipopeptide components of polymyxin B and colistin: last-line antibiotics against multidrug-resistant Gram-negative bacteria. ACS Infect Dis 1:568–575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rocha LA, Martín MJ, Pita S, Paz J, Seco C, Margusino L, Villanueva R, Durán MT (1992) Prevention of nosocomial infection in critically ill patients by selective decontamination of the digestive tract. A randomized, double blind, placebo-controlled study. Intensive Care Med 18:398–404. [DOI] [PubMed] [Google Scholar]
- Rodríguez-Avial I, Pena I, Picazo JJ, Rodríguez-Avial C, Culebras E (2015) In vitro activity of the next-generation aminoglycoside plazomicin alone and in combination with colistin, meropenem, fosfomycin or tigecycline against carbapenemase-producing Enterobacteriaceae strains. Int J Antimicrob Agents 46:616–621. [DOI] [PubMed] [Google Scholar]
- Rodríguez-Roldán JM, Altuna-Cuesta A, López A, Carrillo A, Garcia J, León J, Martínez-Pellús AJ (1990) Prevention of nosocomial lung infection in ventilated patients: use of an antimicrobial pharyngeal nonabsorbable paste. Crit Care Med 18:1239–1242. [DOI] [PubMed] [Google Scholar]
- Rodríguez Guardado A, Blanco A, Asensi V, Pérez F, Rial JC, Pintado V, Bustillo E, Lantero M, Tenza E, Alvarez M, et al. (2008) Multidrug-resistant Acinetobacter meningitis in neurosurgical patients with intraventricular catheters: assessment of different treatments. J Antimicrob Chemother 61:908–913. [DOI] [PubMed] [Google Scholar]
- Rosenthal AL, Rovell JM, Girard AE (1976) Polyacrylic bone cement containing erythromycin and colistin. I. In vitro bacteriological activity and diffusion properties of erythromycin, colistin and erythromycin/colistin comibination. J Int Med Res 4:296–304. [DOI] [PubMed] [Google Scholar]
- Rubin EJ, Herrera CM, Crofts AA, Trent MS (2015) PmrD is required for modifications to escherichia coli endotoxin that promote antimicrobial resistance. Antimicrob Agents Chemother 59:2051–2061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruza F, Alvarado F, Herruzo R, Delgado MA, García S, Dorao P, Goded F (1998) Prevention of nosocomial infection in a pediatric intensive care unit (PICU) through the use of selective digestive decontamination. Eur J Epidemiol 14:719–727. [DOI] [PubMed] [Google Scholar]
- Ryan KJ, Schainuck LI, Hickman RO, Striker GE (1969) Colistimethate toxicity. Report of a fatal case in a previously healthy child. JAMA 207:2099–2101. [DOI] [PubMed] [Google Scholar]
- Saadi M, Duperchy E, Brown P, Dawson MJ, Wadman SN (2013) inventors, Cantab Anti-Infectives, assignee. Polymyxin derivatives. WO2013072695. 2013 May 23.
- Sabet M, Tarazi Z, Parkinson J, Roberts K, Thompson P, Nation R, Velkov T, Hecker S, Lomovskaya O, Dudley M, et al. (2019) 707. QPX9003: Pharmacology of a novel polymyxin in mice and rats. Open Forum Infect Dis 6:S319. [Google Scholar]
- Sader HS, Farrell DJ, Jones RN (2011) Susceptibility of Klebsiella spp. to colistin and polymyxin B: results from the SENTRY Antimicrobial Surveillance Program (2006-2009). Int J Antimicrob Agents 37:174–175. [DOI] [PubMed] [Google Scholar]
- Sader HS, Rhomberg PR, Flamm RK, Jones RN (2012) Use of a surfactant (polysorbate 80) to improve MIC susceptibility testing results for polymyxin B and colistin. Diagn Microbiol Infect Dis 74:412–414. [DOI] [PubMed] [Google Scholar]
- Salazar J, Alarcón M, Huerta J, Navarro B, Aguayo D (2017) Phosphoethanolamine addition to the heptose I of the lipopolysaccharide modifies the inner core structure and has an impact on the binding of polymyxin B to the Escherichia coli outer membrane. Arch Biochem Biophys 620:28–34. [DOI] [PubMed] [Google Scholar]
- Sampson TR, Liu X, Schroeder MR, Kraft CS, Burd EM, Weiss DS (2012) Rapid killing of Acinetobacter baumannii by polymyxins is mediated by a hydroxyl radical death pathway. Antimicrob Agents Chemother 56:5642–5649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sánchez García M, Cambronero Galache JA, López Diaz J, Cerdá Cerdá E, Rubio Blasco J, Gómez Aguinaga MA, Núnez Reiz A, Rogero Marín S, Onoro Canaveral JJ, Sacristán del Castillo JA (1998) Effectiveness and cost of selective decontamination of the digestive tract in critically ill intubated patients. A randomized, double-blind, placebo-controlled, multicenter trial. Am J Respir Crit Care Med 158:908–916. [DOI] [PubMed] [Google Scholar]
- Sandle T (2016) Antibiotics and preservatives, in Pharmaceutical Microbiology, pp 171–183, Woodhead Publishing, Oxford. [Google Scholar]
- Sandri AM, Landersdorfer CB, Jacob J, Boniatti MM, Dalarosa MG, Falci DR, Behle TF, Bordinhão RC, Wang J, Forrest A, et al. (2013a) Population pharmacokinetics of intravenous polymyxin B in critically ill patients: implications for selection of dosage regimens. Clin Infect Dis 57:524–531. [DOI] [PubMed] [Google Scholar]
- Sandri AM, Landersdorfer CB, Jacob J, Boniatti MM, Dalarosa MG, Falci DR, Behle TF, Saitovitch D, Wang J, Forrest A, et al. (2013b) Pharmacokinetics of polymyxin B in patients on continuous venovenous haemodialysis. J Antimicrob Chemother 68:674–677. [DOI] [PubMed] [Google Scholar]
- Satlin MJ, Kubin CJ, Blumenthal JS, Cohen AB, Furuya EY, Wilson SJ, Jenkins SG, Calfee DP (2011) Comparative effectiveness of aminoglycosides, polymyxin B, and tigecycline for clearance of carbapenem-resistant Klebsiella pneumoniae from urine. Antimicrob Agents Chemother 55:5893–5899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Satlin MJ, Lewis JS II, Weinstein MP, Patel J, Humphries RM, Kahlmeter G, Giske CG, Turnidge J (2020) Clinical and Laboratory Standards Institute and European Committee on Antimicrobial Susceptibility Testing position statements on polymyxin B and colistin clinical breakpoints. Clin Infect Dis 71:e523–e529. [DOI] [PubMed] [Google Scholar]
- Sato Y, Shindo M, Sakura N, Uchida Y, Kato I (2011) Novel des-fatty acyl-polymyxin B derivatives with Pseudomonas aeruginosa-specific antimicrobial activity. Chem Pharm Bull (Tokyo) 59:597–602. [DOI] [PubMed] [Google Scholar]
- Schina M, Spyridi E, Daoudakis M, Mertzanos E, Korfias S (2006) Successful treatment of multidrug-resistant Pseudomonas aeruginosa meningitis with intravenous and intrathecal colistin. Int J Infect Dis 10:178–179. [DOI] [PubMed] [Google Scholar]
- Schülin T (2002) In vitro activity of the aerosolized agents colistin and tobramycin and five intravenous agents against Pseudomonas aeruginosa isolated from cystic fibrosis patients in southwestern Germany. J Antimicrob Chemother 49:403–406. [DOI] [PubMed] [Google Scholar]
- Schuster A, Haliburn C, Döring G, Goldman MH, Freedom Study Group (2013) Safety, efficacy and convenience of colistimethate sodium dry powder for inhalation (Colobreathe DPI) in patients with cystic fibrosis: a randomised study. Thorax 68:344–350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwartz BS, Warren MR, Barkley FA, Landis L (1959–1960) Microbiological and pharmacological studies of colistin sulfate and sodium colistinmethanesulfonate. Antibiot Annu 7:41–60. [PubMed] [Google Scholar]
- Shaheen M, Li J, Ross AC, Vederas JC, Jensen SE (2011) Paenibacillus polymyxa PKB1 produces variants of polymyxin B-type antibiotics. Chem Biol 18:1640–1648. [DOI] [PubMed] [Google Scholar]
- Shen H, Smith DE, Yang T, Huang YG, Schnermann JB, Brosius FC III (1999) Localization of PEPT1 and PEPT2 proton-coupled oligopeptide transporter mRNA and protein in rat kidney. Am J Physiol 276:F658–F665. [DOI] [PubMed] [Google Scholar]
- Sheng WH, Wang JT, Li SY, Lin YC, Cheng A, Chen YC, Chang SC (2011) Comparative in vitro antimicrobial susceptibilities and synergistic activities of antimicrobial combinations against carbapenem-resistant Acinetobacter species: Acinetobacter baumannii versus Acinetobacter genospecies 3 and 13TU. Diagn Microbiol Infect Dis 70:380–386. [DOI] [PubMed] [Google Scholar]
- Shepherd RG, Stansly PG, Winterbottom R, English JP, Fellows CE, Ananenko NH, Guillet GL (1948) Chemical studies on polymyxin; isolation and preliminary purification. J Am Chem Soc 70:3771–3774. [DOI] [PubMed] [Google Scholar]
- Shields RK, Nguyen MH, Hao B, Kline EG, Clancy CJ (2018) Colistin does not potentiate ceftazidime-avibactam killing of carbapenem-resistant Enterobacteriaceae in vitro or suppress emergence of ceftazidime-avibactam resistance. Antimicrob Agents Chemother 62:e01018-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimizu T, Miyake T, Tani M (2017) History and current status of polymyxin B-immobilized fiber column for treatment of severe sepsis and septic shock. Ann Gastroenterol Surg 1:105–113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimomura H, Matsuura M, Saito S, Hirai Y, Isshiki Y, Kawahara K (2003) Unusual interaction of a lipopolysaccharide isolated from Burkholderia cepacia with polymyxin B. Infect Immun 71:5225–5230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shoji H, Tani T, Hanasawa K, Kodama M (1998) Extracorporeal endotoxin removal by polymyxin B immobilized fiber cartridge: designing and antiendotoxin efficacy in the clinical application. Ther Apher 2:3–12. [DOI] [PubMed] [Google Scholar]
- Shoji J, Kato T, Hinoo H (1977a) The structure of polymyxin T1. (Studies on antibiotics from the genus Bacillus. XXII). J Antibiot (Tokyo) 30:1042–1048. [DOI] [PubMed] [Google Scholar]
- Shoji J, Kato T, Hinoo H (1977b) The structures of two new polymyxin group antibiotics. J Antibiot (Tokyo) 30:427–429. [DOI] [PubMed] [Google Scholar]
- Sirijatuphat R, Limmahakhun S, Sirivatanauksorn V, Nation RL, Li J, Thamlikitkul V (2015) Preliminary clinical study of the effect of ascorbic acid on colistin-associated nephrotoxicity. Antimicrob Agents Chemother 59:3224–3232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sivanesan SS, Azad MAK, Schneider EK, Ahmed MU, Huang J, Wang J, Li J, Nation RL, Velkov T (2017a) Gelofusine ameliorates colistin-induced nephrotoxicity. Antimicrob Agents Chemother 61:e00985-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sivanesan S, Roberts K, Wang J, Chea SE, Thompson PE, Li J, Nation RL, Velkov T (2017b) Pharmacokinetics of the individual major components of polymyxin B and colistin in rats. J Nat Prod 80:225–229. [DOI] [PubMed] [Google Scholar]
- Sivanesan S, Wang J, Cheah SE, Schittenhelm R, Purcell AW, Velkov T, Li J (2017c) EP0951: Identification of potential early urinary biomarkers for polymyxin-induced nephrotoxicity using proteomics, in 27th European Congress of Clinical Microbiology and Infectious Diseases, Vienna, Austria.
- Smith SD, Jackson RJ, Hannakan CJ, Wadowsky RM, Tzakis AG, Rowe MI (1993) Selective decontamination in pediatric liver transplants. A randomized prospective study. Transplantation 55:1306–1309. [DOI] [PubMed] [Google Scholar]
- Sobieszczyk ME, Furuya EY, Hay CM, Pancholi P, Della-Latta P, Hammer SM, Kubin CJ (2004) Combination therapy with polymyxin B for the treatment of multidrug-resistant Gram-negative respiratory tract infections. J Antimicrob Chemother 54:566–569. [DOI] [PubMed] [Google Scholar]
- Sorlí L, Luque S, Li J, Campillo N, Danés M, Montero M, Segura C, Grau S, Horcajada JP (2019) Colistin for the treatment of urinary tract infections caused by extremely drug-resistant Pseudomonas aeruginosa: dose is critical. J Infect 79:253–261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Souli M, Rekatsina PD, Chryssouli Z, Galani I, Giamarellou H, Kanellakopoulou K (2009) Does the activity of the combination of imipenem and colistin in vitro exceed the problem of resistance in metallo-beta-lactamase-producing Klebsiella pneumoniae isolates? Antimicrob Agents Chemother 53:2133–2135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Srinivas P, Hunt LN, Pouch SM, Thomas K, Goff DA, Pancholi P, Balada-Llasat JM, Bauer KA (2018) Detection of colistin heteroresistance in Acinetobacter baumannii from blood and respiratory isolates. Diagn Microbiol Infect Dis 91:194–198. [DOI] [PubMed] [Google Scholar]
- Srinivasan VB, Singh BB, Priyadarshi N, Chauhan NK, Rajamohan G (2014) Role of novel multidrug efflux pump involved in drug resistance in Klebsiella pneumoniae. PLoS One 9:e96288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stamm WE, Norrby SR (2001) Urinary tract infections: disease panorama and challenges. J Infect Dis 183 (Suppl 1):S1–S4. [DOI] [PubMed] [Google Scholar]
- Stansly PG, Brownlee G (1949) Nomenclature of polymyxin antibiotics. Nature 163:611. [DOI] [PubMed] [Google Scholar]
- Stansly PG, Shepherd RG, White HJ (1947) Polymyxin: a new chemotherapeutic agent. Bull Johns Hopkins Hosp 81:43–54. [PubMed] [Google Scholar]
- Storm DR, Rosenthal KS, Swanson PE (1977) Polymyxin and related peptide antibiotics. Annu Rev Biochem 46:723–763. [DOI] [PubMed] [Google Scholar]
- Stoutenbeek CP, van Saene HK, Miranda DR, Zandstra DF, Langrehr D (1987) The effect of oropharyngeal decontamination using topical nonabsorbable antibiotics on the incidence of nosocomial respiratory tract infections in multiple trauma patients. J Trauma 27:357–364. [DOI] [PubMed] [Google Scholar]
- Suh JY, Son JS, Chung DR, Peck KR, Ko KS, Song JH (2010) Nonclonal emergence of colistin-resistant Klebsiella pneumoniae isolates from blood samples in South Korea. Antimicrob Agents Chemother 54:560–562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun HY, Shields RK, Cacciarelli TV, Muder RR, Singh N (2010) A novel combination regimen for the treatment of refractory bacteremia due to multidrug-resistant Pseudomonas aeruginosa in a liver transplant recipient. Transpl Infect Dis 12:555–560. [DOI] [PubMed] [Google Scholar]
- Sutton TA, Hato T, Mai E, Yoshimoto M, Kuehl S, Anderson M, Mang H, Plotkin Z, Chan RJ, Dagher PC (2013) p53 is renoprotective after ischemic kidney injury by reducing inflammation. J Am Soc Nephrol 24:113–124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki T, Hayashi K, Fujikawa K, Tsukamoto K (1965) The chemical structure of polymyxin E: the identities of polymyxin E1 with colistin A and of polymyxin E2 with colistin B. J Biochem 57:226–227. [DOI] [PubMed] [Google Scholar]
- Suzuki T, Yamaguchi H, Ogura J, Kobayashi M, Yamada T, Iseki K (2013) Megalin contributes to kidney accumulation and nephrotoxicity of colistin. Antimicrob Agents Chemother 57:6319–6324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki Y, Toyota S, Katoh S, Fukushi Y, Orikasa S, Majima K (1989) [Clinical effects of intravesical instillation of polymyxin B in patients with acute cystitis]. Hinyokika Kiyo 35:593–596. [PubMed] [Google Scholar]
- Tam VH, Schilling AN, Vo G, Kabbara S, Kwa AL, Wiederhold NP, Lewis RE (2005) Pharmacodynamics of polymyxin B against Pseudomonas aeruginosa. Antimicrob Agents Chemother 49:3624–3630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tan CH, Li J, Nation RL (2007) Activity of colistin against heteroresistant Acinetobacter baumannii and emergence of resistance in an in vitro pharmacokinetic/pharmacodynamic model. Antimicrob Agents Chemother 51:3413–3415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tan TY, Ng LSY (2006a) Comparison of three standardized disc susceptibility testing methods for colistin. J Antimicrob Chemother 58:864–867. [DOI] [PubMed] [Google Scholar]
- Tan TY, Ng SY (2006b) The in-vitro activity of colistin in Gram-negative bacteria. Singapore Med J 47:621–624. [PubMed] [Google Scholar]
- Tan TY, Ng SY (2007) Comparison of Etest, Vitek and agar dilution for susceptibility testing of colistin. Clin Microbiol Infect 13:541–544. [DOI] [PubMed] [Google Scholar]
- Tang HJ, Ku YH, Lee MF, Chuang YC, Yu WL (2015) In vitro activity of imipenem and colistin against a carbapenem-resistant Klebsiella pneumoniae isolate coproducing SHV-31, CMY-2, and DHA-1. BioMed Res Int 2015:568079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tängdén T, Hickman RA, Forsberg P, Lagerbäck P, Giske CG, Cars O (2014) Evaluation of double- and triple-antibiotic combinations for VIM- and NDM-producing Klebsiella pneumoniae by in vitro time-kill experiments. Antimicrob Agents Chemother 58:1757–1762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tängdén T, Karvanen M, Friberg LE, Odenholt I, Cars O (2017) Assessment of early combination effects of colistin and meropenem against Pseudomonas aeruginosa and Acinetobacter baumannii in dynamic time-kill experiments. Infect Dis (Lond) 49:521–527. [DOI] [PubMed] [Google Scholar]
- Tani T, Shimizu T, Tani M, Shoji H, Endo Y (2019) Anti-endotoxin properties of polymyxin B-immobilized fibers, in Polymyxin Antibiotics: From Laboratory Bench to Bedside (Li J, Nation RL, Kaye K eds) pp 321–341, Springer International Publishing, New York. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tascini C, Gemignani G, Ferranti S, Tagliaferri E, Leonildi A, Lucarini A, Menichetti F (2004) Microbiological activity and clinical efficacy of a colistin and rifampin combination in multidrug-resistant Pseudomonas aeruginosa infections. J Chemother 16:282–287. [DOI] [PubMed] [Google Scholar]
- Tascini C, Tagliaferri E, Giani T, Leonildi A, Flammini S, Casini B, Lewis R, Ferranti S, Rossolini GM, Menichetti F (2013) Synergistic activity of colistin plus rifampin against colistin-resistant KPC-producing Klebsiella pneumoniae. Antimicrob Agents Chemother 57:3990–3993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tetteroo GW, Wagenvoort JH, Mulder PG, Ince C, Bruining HA (1993) Decreased mortality rate and length of hospital stay in surgical intensive care unit patients with successful selective decontamination of the gut. Crit Care Med 21:1692–1698. [DOI] [PubMed] [Google Scholar]
- Thamlikitkul V, Dubrovskaya Y, Manchandani P, Ngamprasertchai T, Boonyasiri A, Babic JT, Tam VH (2016) Dosing and pharmacokinetics of polymyxin B in patients with renal insufficiency. Antimicrob Agents Chemother 61:e01337-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thornsberry C, Baker CN, Kirven LA (1978) In vitro activity of antimicrobial agents on Legionnaires disease bacterium. Antimicrob Agents Chemother 13:78–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tiddens HA, Bos AC, Mouton JW, Devadason S, Janssens HM (2014) Inhaled antibiotics: dry or wet? Eur Respir J 44:1308–1318. [DOI] [PubMed] [Google Scholar]
- Tietgen M, Semmler T, Riedel-Christ S, Kempf VAJ, Molinaro A, Ewers C, Göttig S (2018) Impact of the colistin resistance gene mcr-1 on bacterial fitness. Int J Antimicrob Agents 51:554–561. [DOI] [PubMed] [Google Scholar]
- Tochikubo K, Yasuda Y, Kozuka S (1986) Decreased particulate NADH oxidase activity in Bacillus subtilis spores after polymyxin B treatment. J Gen Microbiol 132:277–287. [DOI] [PubMed] [Google Scholar]
- Tran AX, Lester ME, Stead CM, Raetz CRH, Maskell DJ, McGrath SC, Cotter RJ, Trent MS (2005) Resistance to the antimicrobial peptide polymyxin requires myristoylation of Escherichia coli and Salmonella typhimurium lipid A. J Biol Chem 280:28186–28194. [DOI] [PubMed] [Google Scholar]
- Trent MS, Ribeiro AA, Lin S, Cotter RJ, Raetz CR (2001) An inner membrane enzyme in Salmonella and Escherichia coli that transfers 4-amino-4-deoxy-L-arabinose to lipid A: induction on polymyxin-resistant mutants and role of a novel lipid-linked donor. J Biol Chem 276:43122–43131. [DOI] [PubMed] [Google Scholar]
- Tsuji BT, Pogue JM, Zavascki AP, Paul M, Daikos GL, Forrest A, Giacobbe DR, Viscoli C, Giamarellou H, Karaiskos I, et al. (2019) International Consensus Guidelines for the Optimal Use of the Polymyxins: Endorsed by the American College of Clinical Pharmacy (ACCP), European Society of Clinical Microbiology and Infectious Diseases (ESCMID), Infectious Diseases Society of America (IDSA), International Society for Anti-infective Pharmacology (ISAP), Society of Critical Care Medicine (SCCM), and Society of Infectious Diseases Pharmacists (SIDP). Pharmacotherapy 39:10–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tumbarello M, Repetto E, Trecarichi EM, Bernardini C, De Pascale G, Parisini A, Rossi M, Molinari MP, Spanu T, Viscoli C, et al. (2011) Multidrug-resistant Pseudomonas aeruginosa bloodstream infections: risk factors and mortality. Epidemiol Infect 139:1740–1749. [DOI] [PubMed] [Google Scholar]
- Tumbarello M, Viale P, Viscoli C, Trecarichi EM, Tumietto F, Marchese A, Spanu T, Ambretti S, Ginocchio F, Cristini F, et al. (2012) Predictors of mortality in bloodstream infections caused by Klebsiella pneumoniae carbapenemase-producing K. pneumoniae: importance of combination therapy. Clin Infect Dis 55:943–950. [DOI] [PubMed] [Google Scholar]
- Tzeng YL, Ambrose KD, Zughaier S, Zhou X, Miller YK, Shafer WM, Stephens DS (2005) Cationic antimicrobial peptide resistance in Neisseria meningitidis. J Bacteriol 187:5387–5396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- United States Pharmacopeial Convention (2018) United States pharmacopeia 40: National Formulary 31, United States Pharmacopeial Convention, Rockville, MD.
- Urban C, Mariano N, Rahal JJ (2010) In vitro double and triple bactericidal activities of doripenem, polymyxin B, and rifampin against multidrug-resistant Acinetobacter baumannii, Pseudomonas aeruginosa, Klebsiella pneumoniae, and Escherichia coli. Antimicrob Agents Chemother 54:2732–2734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaara M (2013) Novel derivatives of polymyxins. J Antimicrob Chemother 68:1213–1219. [DOI] [PubMed] [Google Scholar]
- Vaara M (2019a) Polymyxin derivatives that sensitize Gram-negative bacteria to other antibiotics. Molecules 24:249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaara M (2019b) Polymyxins and their potential next generation as therapeutic antibiotics. Front Microbiol 10:1689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaara M, Fox J, Loidl G, Siikanen O, Apajalahti J, Hansen F, Frimodt-Møller N, Nagai J, Takano M, Vaara T (2008) Novel polymyxin derivatives carrying only three positive charges are effective antibacterial agents. Antimicrob Agents Chemother 52:3229–3236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaara M, Sader HS, Rhomberg PR, Jones RN, Vaara T (2013) Antimicrobial activity of the novel polymyxin derivative NAB739 tested against Gram-negative pathogens. J Antimicrob Chemother 68:636–639. [DOI] [PubMed] [Google Scholar]
- Vaara M, Siikanen O, Apajalahti J, Fox J, Frimodt-Møller N, He H, Poudyal A, Li J, Nation RL, Vaara T (2010a) A novel polymyxin derivative that lacks the fatty acid tail and carries only three positive charges has strong synergism with agents excluded by the intact outer membrane. Antimicrob Agents Chemother 54:3341–3346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaara M, Siikanen O, Apajalahti J, Frimodt-Møller N, Vaara T (2010b) Susceptibility of carbapenemase-producing strains of Klebsiella pneumoniae and Escherichia coli to the direct antibacterial activity of NAB739 and to the synergistic activity of NAB7061 with rifampicin and clarithromycin. J Antimicrob Chemother 65:942–945. [DOI] [PubMed] [Google Scholar]
- Vaara M, Vaara T (2010) Structure-activity studies on novel polymyxin derivatives that carry only three positive charges. Peptides 31:2318–2321. [DOI] [PubMed] [Google Scholar]
- Vaara M, Vaara T (2013) The novel polymyxin derivative NAB739 is remarkably less cytotoxic than polymyxin B and colistin to human kidney proximal tubular cells. Int J Antimicrob Agents 41:292–293. [DOI] [PubMed] [Google Scholar]
- Vaara M, Vaara T, Jensen M, Helander I, Nurminen M, Rietschel ET, Mäkelä PH (1981) Characterization of the lipopolysaccharide from the polymyxin-resistant pmrA mutants of Salmonella typhimurium. FEBS Lett 129:145–149. [DOI] [PubMed] [Google Scholar]
- Valour F, Dutronc H, Dinh A, Cazorla C, Pavèse P, Lesens O, Uçkay I, Chidiac C, Ferry T, Colistin BJIs Study Group (2013) Difficult-to-treat Gram-negative bone and joint infections: efficacy and safety of prolonged intravenous colistin. Int J Antimicrob Agents 41:197–199. [DOI] [PubMed] [Google Scholar]
- Vardakas KZ, Voulgaris GL, Samonis G, Falagas ME (2018) Inhaled colistin monotherapy for respiratory tract infections in adults without cystic fibrosis: a systematic review and meta-analysis. Int J Antimicrob Agents 51:1–9. [DOI] [PubMed] [Google Scholar]
- Vasen W, Desmery P, Ilutovich S, Di Martino A (2000) Intrathecal use of colistin. J Clin Microbiol 38:3523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vattimo MdeF, Watanabe M, da Fonseca CD, Neiva LB, Pessoa EA, Borges FT (2016) Polymyxin B nephrotoxicity: from organ to cell damage. PLoS One 11:e0161057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Velkov T, Abdul Rahim N, Zhou QT, Chan HK, Li J (2015) Inhaled anti-infective chemotherapy for respiratory tract infections: successes, challenges and the road ahead. Adv Drug Deliv Rev 85:65–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Velkov T, Dai C, Ciccotosto GD, Cappai R, Hoyer D, Li J (2018a) Polymyxins for CNS infections: pharmacology and neurotoxicity. Pharmacol Ther 181:85–90. [DOI] [PubMed] [Google Scholar]
- Velkov T, Gallardo-Godoy A, Swarbrick JD, Blaskovich MAT, Elliott AG, Han M, Thompson PE, Roberts KD, Huang JX, Becker B, et al. (2018b) Structure, function, and biosynthetic origin of octapeptin antibiotics active against extensively drug-resistant Gram-negative bacteria. Cell Chem Biol 25:380–391.e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Velkov T, Roberts KD (2019) Discovery of novel polymyxin-like antibiotics, in Polymyxin Antibiotics: From Laboratory Bench to Bedside (Li J, Nation RL, Kaye K eds) pp 343–362, Springer International Publishing, New York. [DOI] [PubMed] [Google Scholar]
- Velkov T, Roberts KD, Nation RL, Thompson PE, Li J (2013) Pharmacology of polymyxins: new insights into an ‘old’ class of antibiotics. Future Microbiol 8:711–724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Velkov T, Roberts KD, Nation RL, Wang J, Thompson PE, Li J (2014) Teaching ‘old’ polymyxins new tricks: new-generation lipopeptides targeting Gram-negative ‘superbugs’. ACS Chem Biol 9:1172–1177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Velkov T, Roberts KD, Thompson PE, Li J (2016a) Polymyxins: a new hope in combating Gram-negative superbugs? Future Med Chem 8:1017–1025. [DOI] [PubMed] [Google Scholar]
- Velkov T, Thompson PE, Nation RL, Li J (2010) Structure--activity relationships of polymyxin antibiotics. J Med Chem 53:1898–1916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Velkov T, Yun B, Schneider EK, Azad MA, Dolezal O, Morris FC, Nation RL, Wang J, Chen K, Yu HH, et al. (2016b) A novel chemical biology approach for mapping of polymyxin lipopeptide antibody binding epitopes. ACS Infect Dis 2:341–351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vicari G, Bauer SR, Neuner EA, Lam SW (2013) Association between colistin dose and microbiologic outcomes in patients with multidrug-resistant Gram-negative bacteremia. Clin Infect Dis 56:398–404. [DOI] [PubMed] [Google Scholar]
- Vincent JL, Laterre PF, Cohen J, Burchardi H, Bruining H, Lerma FA, Wittebole X, De Backer D, Brett S, Marzo D, et al. (2005) A pilot-controlled study of a polymyxin B-immobilized hemoperfusion cartridge in patients with severe sepsis secondary to intra-abdominal infection. Shock 23:400–405. [DOI] [PubMed] [Google Scholar]
- Vingsbo Lundberg C, Vaara T, Frimodt-Møller N, Vaara M (2010) Novel polymyxin derivatives are effective in treating experimental Escherichia coli peritoneal infection in mice. J Antimicrob Chemother 65:981–985. [DOI] [PubMed] [Google Scholar]
- Voitenko VG, Bayramashvili DI, Zebrev AI, Zinchenko AA (1990) Relationship between structure and histamine releasing action of polymyxin B and its analogues. Agents Actions 30:153–156. [DOI] [PubMed] [Google Scholar]
- Volkow-Fernández P, Rodríguez CF, Cornejo-Juárez P (2012) Intravesical colistin irrigation to treat multidrug-resistant Acinetobacter baumannii urinary tract infection: a case report. J Med Case Reports 6:426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- von Graevenitz A, Nourbakhsh M (1972) Antimicrobial resistance of the genera Proteus, Providencia and Serratia with special reference to multiple resistance patterns. Med Microbiol Immunol (Berl) 157:142–148. [DOI] [PubMed] [Google Scholar]
- Vourli S, Dafopoulou K, Vrioni G, Tsakris A, Pournaras S (2017) Evaluation of two automated systems for colistin susceptibility testing of carbapenem-resistant Acinetobacter baumannii clinical isolates. J Antimicrob Chemother 72:2528–2530. [DOI] [PubMed] [Google Scholar]
- Waites KB, Canupp KC, Roper JF, Camp SM, Chen Y (2006) Evaluation of 3 methods of bladder irrigation to treat bacteriuria in persons with neurogenic bladder. J Spinal Cord Med 29:217–226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walkty A, DeCorby M, Nichol K, Karlowsky JA, Hoban DJ, Zhanel GG (2009) In vitro activity of colistin (polymyxin E) against 3,480 isolates of Gram-negative bacilli obtained from patients in Canadian hospitals in the CANWARD study, 2007-2008. Antimicrob Agents Chemother 53:4924–4926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walsh CC, Landersdorfer CB, McIntosh MP, Peleg AY, Hirsch EB, Kirkpatrick CM, Bergen PJ (2016) Clinically relevant concentrations of fosfomycin combined with polymyxin B, tobramycin or ciprofloxacin enhance bacterial killing of Pseudomonas aeruginosa, but do not suppress the emergence of fosfomycin resistance. J Antimicrob Chemother 71:2218–2229. [DOI] [PubMed] [Google Scholar]
- Wang C, Feng Y, Liu L, Wei L, Kang M, Zong Z (2020) Identification of novel mobile colistin resistance gene mcr-10. Emerg Microbes Infect 9:508–516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J, Yi M, Chen X, Muhammad I, Liu F, Li R, Li J, Li J (2016) Effects of colistin on amino acid neurotransmitters and blood-brain barrier in the mouse brain. Neurotoxicol Teratol 55:32–37. [DOI] [PubMed] [Google Scholar]
- Wang X, Wang Y, Zhou Y, Li J, Yin W, Wang S, Zhang S, Shen J, Shen Z, Wang Y (2018) Emergence of a novel mobile colistin resistance gene, mcr-8, in NDM-producing Klebsiella pneumoniae. Emerg Microbes Infect 7:122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wareham DW, Gordon NC, Hornsey M (2011) In vitro activity of teicoplanin combined with colistin versus multidrug-resistant strains of Acinetobacter baumannii. J Antimicrob Chemother 66:1047–1051. [DOI] [PubMed] [Google Scholar]
- Waterman P, Barber M, Weintrob AC, VanBrakle R, Howard R, Kozar MP, Andersen R, Wortmann G (2012) The elution of colistimethate sodium from polymethylmethacrylate and calcium phosphate cement beads. Am J Orthop 41:256–259. [PubMed] [Google Scholar]
- Wertheim H, Van Nguyen K, Hara GL, Gelband H, Laxminarayan R, Mouton J, Cars O (2013) Global survey of polymyxin use: a call for international guidelines. J Glob Antimicrob Resist 1:131–134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Westerman EM, Le Brun PP, Touw DJ, Frijlink HW, Heijerman HG (2004) Effect of nebulized colistin sulphate and colistin sulphomethate on lung function in patients with cystic fibrosis: a pilot study. J Cyst Fibros 3:23–28. [DOI] [PubMed] [Google Scholar]
- White HJ, Alverson CM, Baker MJ, Jackson ER (1949) Comparative biological studies of polymyxin and aerosporin. Ann N Y Acad Sci 51:879–890. [DOI] [PubMed] [Google Scholar]
- Wilkinson S (1949) Crystalline derivatives of the polymyxins and the identification of the fatty acid component. Nature 164:622. [DOI] [PubMed] [Google Scholar]
- Wilkinson S (1963) Identity of colistin and polymyxin E. Lancet 1:922–923. [DOI] [PubMed] [Google Scholar]
- Wilkinson S, Lowe LA (1966) Structures of the polymyxins A and the question of identity with the polymyxins M. Nature 212:311. [DOI] [PubMed] [Google Scholar]
- Williams GJ, Breazeale SD, Raetz CR, Naismith JH (2005) Structure and function of both domains of ArnA, a dual function decarboxylase and a formyltransferase, involved in 4-amino-4-deoxy-L-arabinose biosynthesis. J Biol Chem 280:23000–23008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams L, Malhotra Y, Murante B, Laverty S, Cook S, Topa D, Hardy D, Wang H, Gigliotti F (2013) A single-blinded randomized clinical trial comparing polymyxin B-trimethoprim and moxifloxacin for treatment of acute conjunctivitis in children. J Pediatr 162:857–861. [DOI] [PubMed] [Google Scholar]
- Wolinsky E, Hines JD (1962) Neurotoxic and nephrotoxic effects of colistin in patients with renal disease. N Engl J Med 266:759–762. [DOI] [PubMed] [Google Scholar]
- World Health Organization (2017) Global Priority List of Antibiotic-Resistant Bacteria Guide to Guide Research, Discovery, and Development of New Antibiotics, World Health Organization, Geneva, Switzerland. [Google Scholar]
- Wright MS, Suzuki Y, Jones MB, Marshall SH, Rudin SD, van Duin D, Kaye K, Jacobs MR, Bonomo RA, Adams MD (2015) Genomic and transcriptomic analyses of colistin-resistant clinical isolates of Klebsiella pneumoniae reveal multiple pathways of resistance. Antimicrob Agents Chemother 59:536–543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wright WW, Welch H (1959–1960) Chemical, biological and clinical observations on colistin. Antibiot Annu 7:61–74. [PubMed] [Google Scholar]
- Wunsch H, Moitra VK, Patel M, Dzierba AL (2012) Polymyxin use associated with respiratory arrest. Chest 141:515–517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yagmur R, Esen F (2006) Intrathecal colistin for treatment of Pseudomonas aeruginosa ventriculitis: report of a case with successful outcome. Crit Care 10:428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yan A, Guan Z, Raetz CR (2007) An undecaprenyl phosphate-aminoarabinose flippase required for polymyxin resistance in Escherichia coli. J Biol Chem 282:36077–36089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang YQ, Li YX, Lei CW, Zhang AY, Wang HN (2018) Novel plasmid-mediated colistin resistance gene mcr-7.1 in Klebsiella pneumoniae. J Antimicrob Chemother 73:1791–1795. [DOI] [PubMed] [Google Scholar]
- Yapa SWS, Li J, Patel K, Wilson JW, Dooley MJ, George J, Clark D, Poole S, Williams E, Porter CJ, et al. (2014) Pulmonary and systemic pharmacokinetics of inhaled and intravenous colistin methanesulfonate in cystic fibrosis patients: targeting advantage of inhalational administration. Antimicrob Agents Chemother 58:2570–2579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoon J, Urban C, Terzian C, Mariano N, Rahal JJ (2004) In vitro double and triple synergistic activities of polymyxin B, imipenem, and rifampin against multidrug-resistant Acinetobacter baumannii. Antimicrob Agents Chemother 48:753–757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yousef JM, Chen G, Hill PA, Nation RL, Li J (2011) Melatonin attenuates colistin-induced nephrotoxicity in rats. Antimicrob Agents Chemother 55:4044–4049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yousef JM, Chen G, Hill PA, Nation RL, Li J (2012) Ascorbic acid protects against the nephrotoxicity and apoptosis caused by colistin and affects its pharmacokinetics. J Antimicrob Chemother 67:452–459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu W, Zhou K, Guo L, Ji J, Niu T, Xiao T, Shen P, Xiao Y (2017a) In vitro pharmacokinetics/pharmacodynamics evaluation of fosfomycin combined with amikacin or colistin against KPC2-producing Klebsiella pneumoniae. Front Cell Infect Microbiol 7:246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu Z, Qin W, Lin J, Fang S, Qiu J (2015) Antibacterial mechanisms of polymyxin and bacterial resistance. BioMed Res Int 2015:679109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu Z, Zhu Y, Qin W, Yin J, Qiu J (2017b) Oxidative stress induced by polymyxin E is involved in rapid killing of Paenibacillus polymyxa. BioMed Res Int 2017:5437139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yun B, Azad MA, Wang J, Nation RL, Thompson PE, Roberts KD, Velkov T, Li J (2015a) Imaging the distribution of polymyxins in the kidney. J Antimicrob Chemother 70:827–829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yun B, Azad MAK, Nowell CJ, Nation RL, Thompson PE, Roberts KD, Velkov T, Li J (2015b) Cellular uptake and localization of polymyxins in renal tubular cells using rationally designed fluorescent probes. Antimicrob Agents Chemother 59:7489–7496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yun B, Zhang T, Azad MAK, Wang J, Nowell CJ, Kalitsis P, Velkov T, Hudson DF, Li J (2018) Polymyxin B causes DNA damage in HK-2 cells and mice. Arch Toxicol 92:2259–2271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zaidi STR, Al Omran S, Al Aithan ASM, Al Sultan M (2014) Efficacy and safety of low-dose colistin in the treatment for infections caused by multidrug-resistant Gram-negative bacteria. J Clin Pharm Ther 39:272–276. [DOI] [PubMed] [Google Scholar]
- Zalts R, Neuberger A, Hussein K, Raz-Pasteur A, Geffen Y, Mashiach T, Finkelstein R (2016) Treatment of carbapenem-resistant Acinetobacter baumannii ventilator-associated pneumonia: retrospective comparison between intravenous colistin and intravenous ampicillin-sulbactam. Am J Ther 23:e78–e85. [DOI] [PubMed] [Google Scholar]
- Zarkotou O, Pournaras S, Tselioti P, Dragoumanos V, Pitiriga V, Ranellou K, Prekates A, Themeli-Digalaki K, Tsakris A (2011) Predictors of mortality in patients with bloodstream infections caused by KPC-producing Klebsiella pneumoniae and impact of appropriate antimicrobial treatment. Clin Microbiol Infect 17:1798–1803. [DOI] [PubMed] [Google Scholar]
- Zavascki AP, Goldani LZ, Cao G, Superti SV, Lutz L, Barth AL, Ramos F, Boniatti MM, Nation RL, Li J (2008) Pharmacokinetics of intravenous polymyxin B in critically ill patients. Clin Infect Dis 47:1298–1304. [DOI] [PubMed] [Google Scholar]
- Zavascki AP, Manfro RC, Maciel RA, Falci DR (2015) Head and neck hyperpigmentation probably associated with polymyxin B therapy. Ann Pharmacother 49:1171–1172. [DOI] [PubMed] [Google Scholar]
- Zavascki AP, Nation RL (2017) Nephrotoxicity of polymyxins: is there any difference between colistimethate and polymyxin B? Antimicrob Agents Chemother 61:e02319-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zavascki AP, Schuster LF, Duquia RP (2016) Histopathological findings of pigmented lesion and recovery of natural skin colour in a patient with polymyxin B-associated diffuse hyperpigmentation. Int J Antimicrob Agents 48:579–580. [DOI] [PubMed] [Google Scholar]
- Zgurskaya HI, Löpez CA, Gnanakaran S (2015) Permeability barrier of Gram-negative cell envelopes and approaches to bypass it. ACS Infect Dis 1:512–522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhanel GG, Adam HJ, Baxter MR, Fuller J, Nichol KA, Denisuik AJ, Lagacé-Wiens PR, Walkty A, Karlowsky JA, Schweizer F, et al. Canadian Antimicrobial Resistance Alliance (2013) Antimicrobial susceptibility of 22746 pathogens from Canadian hospitals: results of the CANWARD 2007-11 study. J Antimicrob Chemother 68 (Suppl 1):i7–i22. [DOI] [PubMed] [Google Scholar]
- Zhang R, Shen Y, Walsh TR, Wang Y, Hu F (2020) Use of polymyxins in Chinese hospitals. Lancet Infect Dis 20:1125–1126. [DOI] [PubMed] [Google Scholar]
- Zhao J, Cheah SE, Roberts KD, Nation RL, Thompson PE, Velkov T, Du Z, Johnson MD, Li J (2016) Transcriptomic analysis of the activity of a novel polymyxin against Staphylococcus aureus. mSphere 1:e00119-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao M, Wu XJ, Fan YX, Zhang YY, Guo BN, Yu JC, Cao GY, Chen YC, Wu JF, Shi YG, et al. (2018) Pharmacokinetics of colistin methanesulfonate (CMS) in healthy Chinese subjects after single and multiple intravenous doses. Int J Antimicrob Agents 51:714–720. [DOI] [PubMed] [Google Scholar]
- Zheng G, Cao L, Che Z, Mao E, Chen E, He J (2018) Polymyxin B-induced skin hyperpigmentation: a rare case report and literature review. BMC Pharmacol Toxicol 19:41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou H, Yao Y, Zhu B, Ren D, Yang Q, Fu Y, Yu Y, Zhou J (2019) Risk factors for acquisition and mortality of multidrug-resistant Acinetobacter baumannii bacteremia: a retrospective study from a Chinese hospital. Medicine (Baltimore) 98:e14937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou QT, Leung SSY, Tang P, Parumasivam T, Loh ZH, Chan HK (2015) Inhaled formulations and pulmonary drug delivery systems for respiratory infections. Adv Drug Deliv Rev 85:83–99. [DOI] [PubMed] [Google Scholar]
- Zhou QT, Tang P, Leung SSY, Chan JGY, Chan HK (2014) Emerging inhalation aerosol devices and strategies: where are we headed? Adv Drug Deliv Rev 75:3–17. [DOI] [PubMed] [Google Scholar]
- Zhou Z, Ribeiro AA, Lin S, Cotter RJ, Miller SI, Raetz CR (2001) Lipid A modifications in polymyxin-resistant Salmonella typhimurium: PmrA-dependent 4-amino-4-deoxy-L-arabinose, and phosphoethanolamine incorporation. J Biol Chem 276:43111–43121. [DOI] [PubMed] [Google Scholar]
- Zhu Y, Lu J, Han ML, Jiang X, Azad MAK, Patil NA, Lin YW, Zhao J, Hu Y, Yu HH, et al. (2020) Polymyxins bind to the cell surface of unculturable Acinetobacter baumannii and cause unique dependent resistance. Adv Sci (Weinh) 7:2000704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu Y, Zhao J, Maifiah MHM, Velkov T, Schreiber F, Li J (2019) Metabolic responses to polymyxin treatment in Acinetobacter baumannii ATCC 19606: integrating transcriptomics and metabolomics with genome-scale metabolic modeling. mSystems 4:e00157-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ziaka M, Markantonis SL, Fousteri M, Zygoulis P, Panidis D, Karvouniaris M, Makris D, Zakynthinos E (2013) Combined intravenous and intraventricular administration of colistin methanesulfonate in critically ill patients with central nervous system infection. Antimicrob Agents Chemother 57:1938–1940. [DOI] [PMC free article] [PubMed] [Google Scholar]