Abstract
The microbiome plays an important role in maintaining human health. Despite multiple factors being attributed to the shaping of the human microbiome, extrinsic factors such diet and use of medications including antibiotics appear to dominate. Mucosal surfaces, particularly in the gut, are highly adapted to be able to tolerate a large population of microorganisms whilst still being able to produce a rapid and effective immune response against infection. The intestinal microbiome is not functionally independent from the host mucosa and can, through presentation of microbe-associated molecular patterns (MAMPs) and generation of microbe-derived metabolites, fundamentally influence mucosal barrier integrity and modulate host immunity. In a healthy gut there is an abundance of beneficial bacteria that help to preserve intestinal homoeostasis, promote protective immune responses, and limit excessive inflammation. The importance of the microbiome is further highlighted during dysbiosis where a loss of this finely balanced microbial population can lead to mucosal barrier dysfunction, aberrant immune responses, and chronic inflammation that increases the risk of disease development. Improvements in our understanding of the microbiome are providing opportunities to harness members of a healthy microbiota to help reverse dysbiosis, reduce inflammation, and ultimately prevent disease progression.
Keywords: Dysbiosis, Gastrointestinal physiology, Homeostasis, Inflammation, Microbiome, Mucosa
Introduction
The human body is inhabited by a highly diverse population of microorganisms (microbiota) that has co-evolved with their human hosts over many millennia [1]. The human microbiome, a term more precisely used to describe the genomes of these microorganisms [2], is predominantly made up of bacteria [3], however archaea, viruses, and single-cell eukaryotes (e.g. fungi and protists) are also present [4–7]. These microorganisms are at least as abundant as the number of human host cells [3,8] and combined contain far more genes than the entire human genome [9]. Over the past few decades, research related to the microbiome has intensified, facilitated by rapid advances in culture-independent, high-throughput genomic and metabolomic techniques [10–12]. Consequently, a greater understanding of microbiota population composition and host–microbe interactions has been achieved, especially in the context of human health and disease [11,13,14]. Whereas a balanced microbiota has been shown to play an important role in the maintenance of human health, impairment or imbalance in the makeup of the human microbiota (dysbiosis) can disrupt homoeostasis and lead to the onset or exacerbation of human disease [15]. Multiple factors are known to influence the microbiota, however, studies have shown that the microbiome is more strongly influenced by an individual’s environment [16,17]. There are significant similarities in microbiota composition of genetically unrelated individuals who share a household, with approximately 20% of interperson microbiota variability associated with environmental factors such as diet, lifestyle, and medication [16].
The human microbiome can be separated into compartment-specific ecosystems that exist on the skin and along mucosal surfaces such as those of the oral cavity, gastrointestinal tract, lungs, and genitourinary system [1]. The largest concentration and diversity of microbiota can be found within the gut especially in the colon [1]. The mucosa, which consists of a single cell thick epithelium overlaying a layer of connective tissue called the lamina propria, provides the interface between the host and the environment and is equipped with specialised features, particularly along its apical surface, to allow physiological function whilst also being in contact with the microbiota [18]. The microbiota is however not functionally independent from the host mucosa and can fundamentally influence mucosal integrity, modulating host immune responses and mucosal inflammation.
Here we review the relationship between the microbiota and the mucosa, especially in relation to gut homoeostasis and mucosal inflammation. We first discuss factors that shape an individual’s microbiome and the impact the microbiome has on the intestinal mucosa during homoeostasis. We then explore how dysbiosis of the microbiome can lead to mucosal inflammation, resulting in the development of human disease, and highlight current and emerging therapies being used to suppress mucosal inflammation through targeting of the microbiome.
Factors shaping the microbiome
There is increasing evidence to suggest that there is a core microbiome shared between all individuals [19]. However, the composition and diversity of much of the gut microbiome varies greatly from person to person, adapting to both intrinsic and environmental factors [20,21]. Research to date has shown that environmental factors, mainly diet and medication, dominate over intrinsic factors, such as host genetics, in shaping the microbiome [16,22]. Age [23,24], geography [25], and birthing practices [26,27] are also known to be particularly important for determining microbiome composition (Figure 1).
Figure 1. Factors that contribute to the shaping of the human microbiome.
Diet
In the first year of life, the gut microbiome is relatively unstable becoming progressively more stable following weaning, taking on an adult form typically around 3 years of age [28]. Infant feeding practices as well as adult habitual diet play an important role in shaping the gut microbiome [29]. Studies looking into the effect of diet on the make-up of the intestinal microbiota have to date mainly focused on the so-called ‘Western’ diet, which is characterised by high levels of fat, sugar and refined protein [30,31], and diets that are high in fibre and low in red meat, such as the Mediterranean diet [24,32].
Differences in gut microbiome composition prior to weaning have been observed between breastfed and formula-fed infants. Breastfed infants have a microbiome dominated by Lactobacilli and Prevotella, whereas formula-fed infants exhibit a more diverse microbial population, dominated by Enterococci, Enterobacteria, Bacteroides, Clostridia, and Streptococci [33,34]. Breastmilk contains oligosaccharides which promote the growth of beneficial Bifidobacteria [35]. Bifidobacteria play a major role in the fermentation and conversion of oligosaccharides into short-chain fatty acids (SCFAs), such as butyrate and propionate, which promote healthy immune function (reviewed in detail below ‘The microbiome and intestinal homeostasis’) [36]. In addition to providing critical nutrients and bioactive compounds, human breast milk also plays an important role in the seeding of an infant’s gut microbiome, containing a variety of beneficial bacteria, including Lactobacilli and Bifidobacteria [37]. After weaning, the microbiota becomes more diverse and is dominated by Bacteroidetes and Firmicutes [38].
Studies looking at the adult gut microbiome have found that individuals consuming a Western diet experience a decrease in the total number of gut bacteria, particularly Bifidobacteria and Eubacteria, and an increase in pro-inflammatory bacteria-derived compounds [39–41]. A key aspect of the Western diet is a high intake of saturated fatty acids which has been linked to both a decrease in Gram-negative bacteria within the gut, particularly Bacteroidetes, and an increase in Lactococci [42,43]. Whilst there is currently a lack of consensus as to the precise effect of these dietary components on the microbiome, most studies have observed an overall decrease in bacterial diversity, a decrease in SCFA production, and an increase in harmful bacterial strains, such as pathogenic Escherichia coli (E. coli) [44,45]. In contrast with a Western diet, adults who consume a Mediterranean diet exhibit increased levels of Bifidobacteria, Lactobacilli, Eubacteria, and Bacteroides [46,47]. Furthermore, individuals who consume a Mediterranean diet have been shown to have increased levels of SCFA-producing bacteria, such as Provetella [48]. In addition to habitual diet, research has shown that dietary diversity, meal timing as well as short- and long-term dietary modifications can change the composition and activity of the adult gut microbiome [49–52]. Caloric restriction, for example, which is a nutritional intervention of reduced energy intake, has a strong influence on the gut microbiota [53,54]. It has been found that caloric restriction can slow down age-related decline in the microbiome, increase both microbial diversity and Bacteroidetes/Firmicutes ratio, as well as change host microbial co-metabolites leading to a decrease in host lipid biosynthesis and an increase in fatty acid catabolism [55,56].
Antibiotics and drugs
Antibiotics are medicines used in the treatment of bacterial infections. Whilst they have proved to be an effective treatment against many bacterial diseases, their antimicrobial action profoundly affects the composition and function of the gut microbiome, causing dysbiosis by killing both pathological and beneficial bacteria, and allowing the expansion of resistant microbes [57]. The effects of antibiotics on the gut microbiome are potentially long-lasting, and their use in early life has been associated with an increased risk of developing several conditions including inflammatory bowel disease (IBD) and asthma [58,59].
Antibiotics can drastically reduce, or even fully eliminate, beneficial anaerobic bacterial species such as Bifidobacteria, Lactobacilli, Bacteroides, and Clostridia [60]. After only 7 days of antibiotic treatment, microbial diversity has been found to decrease by 25%, with core phylogenetic microbiota reducing from 29 to 12 taxa and antibiotic-resistant Bacteroidetes increasing 2.5-fold [61]. Consequently, antibiotic use can also result in reduced SCFA production [62]. The effects of antibiotics on the microbiome are however dependent on the type of antibiotic used. Clindamycin, which is a broad-spectrum antibiotic, can cause microbial changes that last for up to 2 years with no recovery in Bacteroides diversity [63]. Clarithromycin and Ciprofloxacin, which are used against Helicobacter pylori, are associated with a decrease in Actinobacteria and Ruminococci, respectively [64,65]. Vancomycin, which is used against Clostridium difficile (C. difficile), causes an increase in Proteobacteria species and a decrease in Bacteroidetes, Ruminoccoci and Faecalibacteria levels, which can lead to both recurrent C. difficile infection (rCDI) and the growth of unwanted bacterial species, such as pathogenic E. coli [66,67].
Non-antibiotic drugs are also known to influence the composition and stability of the microbiome. A recent meta-analysis revealed that in addition to antibiotics, proton pump inhibitors (PPIs), metformin, and laxatives exhibit the greatest effects on gut microbiome composition and function [68]. PPIs reduce microbial diversity and cause taxonomical changes in the gut. Metformin significantly increases E. coli abundance and effects the number of SCFA-producing bacteria [68,69].
Birth mode of delivery
Studies have shown that whereas vaginally delivered babies have a microbiome dominated by Lactobacilli and Prevotella, babies born by caesarean section (C-section) carry a microbiome dominated by Streptococci, Corynebacteria, and Propionibacteria [70,71]. Furthermore, babies born by C-section have been shown to have an abundance of potentially pro-inflammatory Klebsiella and Enterococcus bacteria [26]. A recent study reported that the abundance of Klebsiella and Enterococcus species in C-section born children at 1 week of life was associated with an increased number of respiratory infections over the first year [26]. Additionally, babies delivered by C-section have been shown to have lower total gut microbial diversity, delayed Bacteroidetes colonisation, and a subsequent immune system imbalance during the first 2 years of life which may result in the development of allergies [72,73].
Age
Many studies have observed age-related changes to the gut microbiome. In infancy, the developing gut microbiome undergoes three distinct phases of progression: a developmental phase (months 3–14), a transitional phase (months 15–30), and a stable phase (months 31–46) [74]. Children and young adults have a higher abundance of Bifidobacteria and Clostridia, and a lower microbial diversity compared with adults [75]. In general, healthy adults exhibit high levels of Bacteroidetes and Firmicutes, and low levels of Proteobacteria, Actinobacteria, Fusobacteria, and Verrucomicrobia [20,76,77]. Throughout life, intestinal levels of Firmicutes decrease whilst Bacteroidetes levels increase. Elderly people have a gut microbiome enriched with Bacteroidetes and Proteobacteria and depleted levels of Bifidobacteria and Lactobacilli [24,78]. The transition from healthy adult to healthy old age is characterised by a decrease in microbial diversity, as well as an accumulation of potentially pro-inflammatory microbes and decrease in beneficial microbes [79].
Development geography
To date, most studies investigating the link between the microbiome and geography have focused on differences in microbiome composition amongst three contrasting human populations: hunter gatherers, traditional farming or fishing communities, and Western industrialised communities [80–84]. When comparing the microbiomes of hunter gatherers with those of more developed communities, hunter gatherers were found to have a higher microbial diversity, with enrichment of Prevotella, Treponema, and Bacteroidetes [80,81]. In contrast, Western industrialised communities have higher levels of Bacteroides and Firmicutes, with an overall lower microbial diversity. Some studies suggest that the microbiomes of traditional farming and fishing communities exhibit an intermediate state between hunter gatherers and Western industrialised communities [82,85]. Factors thought to influence gut microbiome composition amongst hunter gatherers include a diet consisting of predominately starchy foods, limited access to modern medicine, and exposure to a wide variety of pathogens and parasites [82,83]. Traditional farming or fishing communities are thought to possess microbiomes with a relatively high taxonomic diversity, allowing the host to withstand pathogens and parasites, as well as to be able to respond to dietary fluctuations due to crop seasonality [83]. In Western industrialised societies, the gut microbiome is thought to be largely determined by diets high in refined protein and fat, good sanitation and hygiene practices, and the habitual use of antibiotics and other medications [80,81,84]. Some studies have also proposed that the lower microbiome diversity found in Western industrialised communities can be attributed to an overall loss of biodiversity due to industrialisation, pollution, and use of chemicals [86,87]. Furthermore, differences in sanitised drinking water may also have an effect on the composition of the gut microbiome [88,89].
The microbiome and intestinal homoeostasis
The intestinal mucosa is highly adapted to be able to tolerate a large population of microorganisms and dietary antigens whilst preserving nutrient uptake and raising an effective immune response to pathogenic infection or commensal intrusion into the underlying host tissue [90]. For the most part, the microbiota maintains symbiosis with the gut environment forming a mutually beneficial relationship with the host. The gut provides a nutrient-rich habitat for the microbiota whilst the microbiota stimulates the host’s immune system, aids digestion, and provides otherwise unobtainable metabolites. In a normal healthy gut, the microbiota is diverse with an abundance of beneficial bacteria that help to maintain gut homoeostasis, promoting protective intestinal immune responses at the mucosal surface, and limiting excessive mucosal inflammation [91].
The microbiota can communicate directly with the host through host recognition of highly conserved structural components, termed microbe-associated molecular patterns (MAMPs) [92], such as lipopolysaccharides (LPSs), peptidoglycan (PGN), and flagellin. Recognition of MAMPs are achieved primarily through binding to pattern-recognition receptors (PRRs) expressed by intestinal epithelial cells (IECs) and immune cells. PPRs are a diverse family of transmembrane and cytoplasmic innate immune receptors, that include Toll-like receptors (TLRs) and nucleotide-binding oligomerisation domain (NOD)-like receptors (NLRs) [93]. PPR stimulation triggers intracellular signalling cascades leading to the expression of a range of immunomodulatory molecules that orchestrate early immune responses resulting in mucosal inflammation and further activation of innate and adaptive immune processes [94]. Whereas activation of PRR by pathogens and pathobionts is known to initiate pro-inflammatory signalling cascades that lead to mucosal inflammation, the commensal microbiota can use similar mechanisms to dampen inflammation and promote intestinal homoeostasis [95]. For example, polysaccharide A (PSA) from the ubiquitous gut commensal Bacteroides fragilis (B. fragilis) is recognised by the TLR1/TLR2 heterodimer, in co-operation with the C-type lectin PRR Dectin-1, triggering a signalling cascade through the phosphoinositide 3-kinase (PI3K) pathway to promote 3′,5′-cyclic adenosine monophosphate (cAMP) response element-binding protein (CREB)-dependent transcription of anti-inflammatory genes [96]. NOD2 stimulation by muramyl-dipeptide (MDP), a PGN motif, triggers intestinal leucine-rich repeat-containing G-protein coupled receptor 5 (Lgr5)+ stem cell survival and epithelial regeneration [97]. In addition to microbe specific constituents, there are also numerous microbiota-derived metabolites, such as SCFA, that stimulate a range of signalling pathways to further regulate mucosal immune responses and aid microbial symbiosis/tolerance [98].
Direct microbial maintenance of intestinal barrier integrity
The intestinal mucosa forms physical, biochemical, and immunological barriers which allow for the symbiotic microbiota–host relationship to be maintained, controlling the microbial population, and reducing direct contact with the host [99]. Maintenance of these barriers are essential for preventing microbial invasion, excessive immune responses, and mucosal inflammation. As well as defending against pathogens through competition for nutrients and production of antimicrobial molecules [100,101], the gut microbiota also plays an active role in the maintenance of host mucosal barriers, which further prevents colonisation by opportunistic pathogens, limiting excessive mucosal inflammation, and preserving gut homoeostasis [99,100].
The physical barrier consists of a wall of IECs that are held together by cell junctions, particularly tight junctions (TJs), allowing only selective paracellular transport of water, ions, solutes, and some nutrients, preventing passage of microorganisms [102]. A mucus layer, predominantly formed of highly glycosylated mucins secreted by goblet cells, covers IECs and further contributes to the physical barrier preventing bacteria from interacting directly with host tissue [103]. The mucus layer also provides moisture and lubrication to protect IECs from dehydration and mechanical stress caused by the passage of food and peristaltic forces [104]. The small intestine contains one layer of mucus whereas the colon contains two: a loose outer layer that is permeable to bacteria and a dense inner layer that is impermeable and devoid of bacteria [105]. In the small intestine particularly, secretory molecules such as antimicrobial peptides (AMPs) and immunoglobulin (Ig)A are released and concentrated in the mucus layer, which further aid separation of the microbiota from the host mucosa [101,106]. In addition to targeting microbes directly and sequestering key nutrients to control microbiota biodiversity, these barriers can also modulate the host’s innate and adaptive immune responses [107,108] and drive up-regulation of mucin and TJ protein expression in IECs to maintain intestinal barrier integrity [109,110].
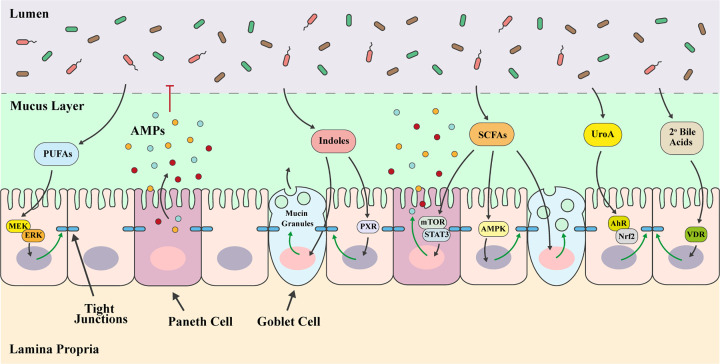
Normal maturation and function of the mucus layer is strongly influenced by the gut microbiota, either through bacterial degradation and turn-over of mucin glycans or by bacteria-mediated processes to regulate host glycosylation of mucins [111]. Additionally, microbe-derived signals and metabolites have been shown to protect the intestinal epithelial barrier, up-regulating and strengthening cell junctions as well as promoting maintenance of the mucus layer and release of antimicrobial molecules (Figure 2). For example, indoles, which are microbiota-derived metabolites produced from the amino acid tryptophan have been shown to increase gene expression linked to TJ formation and mucus production [112,113]. Indoles further protect IECs through attenuation of tumour necrosis factor-α (TNF-α)-mediated activation of nuclear factor κ-light-chain-enhancer of activated B cells (NF-κB), decreased expression of pro-inflammatory cytokine interleukin (IL)-8, reduced attachment of pathogenic E. coli, and increased expression of anti-inflammatory IL-10 [112]. Studies using mice have shown that indole 3-propionic acid (IPA) stimulates the pregnane X receptor (PXR) resulting in up-regulation of TJ proteins in enterocytes and down-regulation of TNF-α [114]. Urolithin A (UroA), a sole microbiota-derived metabolite produced from polyphenolic compounds also enhances intestinal barrier integrity by increasing TJ proteins in IECs through activation of aryl hydrocarbon receptor (AhR)-nuclear factor erythroid 2-related factor 2 (Nrf2)-dependent pathways [115]. SCFAs, in particular butyrate, are the main energy source for colonocytes and are known to promote epithelial barrier integrity [116–119]. SCFAs are taken up by cells either by passive diffusion or facilitated by solute transporters such as monocarboxylate-transporter 1 (MCT-1) and sodium-coupled MCT-1 (SMCT1) where they can then be detected by intracellular receptors such as peroxisome proliferator-activated receptor γ (PPARγ) [120–122]. Alternatively, SCFAs may signal through G-protein coupled receptors (GPRs), such as GPR41, GPR43, and GPR109A, to activate signalling cascades that regulate immune responses [123–125]. SCFAs directly promote mucosal barrier integrity through induction of genes encoding TJ proteins [126], mucins [127], and AMPs [128]. The gut microbial-derived metabolite of polyunsaturated omega-6 fatty acid linoleic acid, 10-hydroxy-cis-12-octadecenoic acid (HYA), is able to ameliorate intestinal barrier damage and changes to cell junction proteins partially via a GPR40-mitogen activated protein kinase kinase (MEK)-extracellular signal-regulated kinase (ERK) pathway [129]. Secondary bile acids, such as lithocholic acid [48], produced by gut microbial conversion of primary bile acids, have also been shown to protect IECs from a TNF-α-induced decrease in TJ proteins through activation of the vitamin D receptor (VDR) [130].
Figure 2. The direct effect of microbiota-derived metabolites on intestinal barrier integrity.
Microbiota-derived metabolites play an important role in maintaining intestinal barrier integrity to prevent epithelial damage and limit mucosal inflammation. Metabolites of polyunsaturated fatty acids (PUFAs) via a GPR40-MEK-ERK pathway have been shown to prevent loss of TJ proteins. Indoles, SCFAs, UroA, and secondary bile acids have also been shown to increase expression of TJ proteins via pathways involving PXR, adenosine monophosphate-activated protein kinase (AMPK), AhR-Nrf2, and VDR, respectively. Indoles and SCFAs promote the production and secretion of mucin, reinforcing the mucus layer. SCFAs activate a mechanistic target of rapamycin (mTOR)-signal transducer and activator of transcription (STAT) 3 pathway in a GPR43-dependent manner to induce production of AMPs.
Mucosal immune regulation by the microbiota
The mucosal immune system is fundamental to intestinal barrier integrity and inflammation. The microbiota plays a vital role, especially in early life, in the maturation and regulation of host immunity to ensure mucosal inflammation is controlled and that the host can differentiate between commensal and pathogenic bacteria [131].
Commensal bacteria have long been associated with the correct development of mucosa-associated lymphoid tissues (MALT), in particular the gut-associated lymphoid tissue (GALT) which includes Peyer’s patches. Early studies using germ-free (GF) mice have shown that the absence of a commensal microbiota correlates with extensive defects in lymphoid tissue architecture and immune responses [132]. A significant reduction in intra-epithelial lymphocytes (IELs), such as αβ and γδ IELs, as well as secretory IgA, is seen in GF mice (compared with their colonised counterparts), which can be reversed following microbial colonisation [133,134]. Gestational maternal colonisation in mice has been shown to increase immune cell subtypes including intestinal group 3 innate lymphoid cells (ILC3s) and F4/80+ CD11c+ mononuclear cells [135]. Pro-inflammatory IL-17+ CD4+ T helper (Th17) cells, which normally exist in large numbers in the lamina propria of the small intestine are absent from GF mice, however, they can be induced upon commensal colonisation [136–138]. This is most notable with segmented filamentous bacteria (SFB), which upon adhesion to IECs, are known to stimulate T-cell responses as well as enhance IgA production [126,139]. PSA from B. fragilis aids cellular and physical maturation of the developing immune system in mice, correcting T-cell deficiencies and imbalances in CD4+ T helper 1 (Th1) and Th2 cell subtypes, directing lymphoid organogenesis [140]. In neonatal mice, B. fragilis is also known to supplement the endogenous lipid antigen milieu with inhibitory sphingolipids, impeding invariant natural killer T (iNKT) cell proliferation in the colonic lamina propria, providing protection against iNKT cell-mediated mucosal inflammation and injury [141]. Microbial colonisation also influences the development of early B-cell lineages in the intestinal mucosa, modulating gut immunoglobulin repertoires [142]. Sufficient intestinal microbiota diversity during early life colonisation has been shown to be essential for the establishment of an immunoregulatory network that protects against elevated induction of IgE at mucosal sites, which is linked to immune hypersensitivity, mucosal inflammation, and allergies [72].
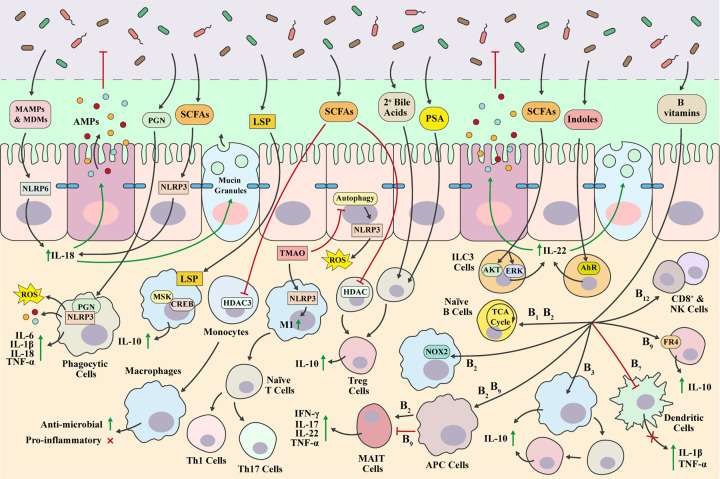
Beyond infancy, the gut microbiota continues to influence the host immune system to maintain host–microbiota symbiosis and intestinal homoeostasis (Figure 3). For example, MAMPs and microbiota-derived metabolites can signal through activation of NLR complexes, called inflammasomes, to shape host immune responses and regulate mucosal barrier function. The microbiota induces NOD-, leucine-rich repeat (LRR)-, and pyrin domain containing 6 (NLRP6) inflammasome signalling to promote steady-state pro-inflammatory IL-18 mucosal secretion, which in turn activates AMP and mucin production in the intestinal mucosa, refining microbiota composition [143]. SCFAs signal through GPR43 and GPR109A to activate NLRP3 leading to IL-18 mucosal secretion [124]. Members of the microbiota, specifically Proteus mirabilis, can induce robust IL-1β production via the NLRP3 inflammasome to promote intestinal mucosal inflammation, mediated by monocytes that are recruited to the intestine in response to epithelial injury [144]. The sensing of PGN fragments and PGN from intact commensal bacteria through multiple PPRs is necessary for the proper development and activation of immune cells. Phagocytes sense internalised PGNs through NLRs and inflammasome complexes (e.g. NLRP3) which induce secretion of pro-inflammatory cytokines (e.g. TNF-α, IL-6, IL-1β, and IL-18) as well as increase antimicrobial responses, such as reactive oxygen species (ROS) and AMP production [145]. Macrophages play a vital role as innate immune effector cells to maintain intestinal homoeostasis, being able to initiate both pro-inflammatory and anti-inflammatory signalling pathways. In mice, intestinal microbial colonisation has been shown to drive continuous replenishment of macrophages in the intestinal mucosa by monocytes that express C–C chemokine receptor type 2 (CCR2) [146]. Helicobacter hepaticus induce an early IL-10 response in intestinal lamina propria-resident macrophages and produce a large soluble polysaccharide (LSP) that activates a specific mitogen and stress-activated protein kinase (MSK)/CREB-dependent anti-inflammatory signalling cascade via TLR2, aiding tolerance and mutualism [147]. Butyrate drives monocyte to macrophage differentiation through histone deacetylase (HDAC) 3 (HDAC3) inhibition to promote an antimicrobial state without inducing pro-inflammatory cytokine production [148]. Trimethylamine N-oxide (TMAO), the oxidated product of gut microbiota-derived trimethylamine, triggers M1 macrophage polarisation via NLRP3 inflammasome activation in mice resulting in Th1 and Th17 differentiation [149]. Furthermore, TMAO has been shown to prime the NLRP3 inflammasome and increase generation of ROS via inhibition of autophagy in colonic epithelial cells contributing to mucosal inflammation [150].
Figure 3. Regulation of mucosal immunity by the intestinal microbiota.
The mucosal immune system is complex with cross-talk between both innate and adaptive components that are primed to counter pathogens and preserve mucosal barrier integrity. MAMPs and microbial-derived metabolites (MDMs) can directly influence this network, aiding the development of host immune responses against pathogens whilst also limiting excessive mucosal inflammation to ensure microbiota tolerance.
ILCs are a heterogeneous innate cell population that specialise in rapid secretion of polarising cytokines and are involved in the initiation of mucosal inflammation to fight infection and inflammatory resolution for mucosal tissue repair [151,152]. Many of the functions of ILCs are mediated by the microbiota [152,153]. For example, proliferation and function of colonic ILC3s is regulated by SCFA activation of GPR43. GPR43 agonism differentially activates protein kinase B (AKT) and ERK signalling, leading to increased colonic ILC3-derived IL-22, ensuring correct mucosal mucin and AMP production from IECs [154,155]. Dichotomous regulation of ILCs has been observed by a pair of Helicobacter species, activating ILCs but negatively regulating proliferation of ILC3s [156].
PSA mediates the conversion of CD4+ cells into anti-inflammatory forkhead box P3 (Foxp3)+ regulatory T (Treg) cells and subsequent production of IL-10, both via TLR2, to suppress mucosal inflammation [157]. SCFAs, such as butyrate and propionate, also induce Treg generation via HDAC inhibition [158]. Microbiota-derived secondary bile acids have recently been shown to regulate colonic retinoic acid receptor-related orphan receptor γ (RORγ)+ Treg induction and homoeostasis [159]. Indoles, such as indole-3-aldehyde, signal through AhR in immune cells to regulate IL-22 production and promote mucosal immune homoeostasis [160]. Bacteria-derived B vitamins have an impact on many aspects of immunological maintenance [161]. Vitamins B1 and B2 act as cofactors for enzymes involved in the TCA cycle and are important for immunometabolism and immune cell differentiation [161,162]. Vitamin B2 is also associated with ROS generation in phagocytic immune cells through priming nicotinamide adenine dinucleotide phosphate (NADPH) oxidase 2 (NOX2) [163]. The vitamin B2 metabolite, 6-hydroxymethyl-8-d-ribityllumazine, bound to major histocompatibility complex (MHC) class I-related protein (MR1) on antigen-presenting cells (APCs), activates mucosal-associated invariant T (MAIT) cells to promote production of pro-inflammatory interferon-γ (IFN-γ) and IL-17 [164]. In contrast, the vitamin B9 metabolite, acetyl-6-formylpterin, inhibits activation of MAIT cells [165]. Vitamin B3 binds to GPR109A on macrophages and dendritic cells leading to an increase in anti-inflammatory cytokines and Treg differentiation [166]. Vitamin B7 (biotin) suppresses the production of pro-inflammatory cytokines [167,168]. Vitamin B9 (folate) binds to the folate receptor 4 (FR4) on differentiated Tregs, promoting cell survival [161]. Vitamin B12 is required for CD8+ T cell differentiation and NK cell activation [169].
As detailed, the intestinal microbiota is not functionally independent from the host mucosa, playing an important role in gut homoeostasis. When there is a perturbation in this finely balanced relationship, loss of mucosal barrier integrity and a rise in abnormal immune responses can occur leading to a risk of sustained pathogenic inflammation and development of disease.
Dysbiosis and disease
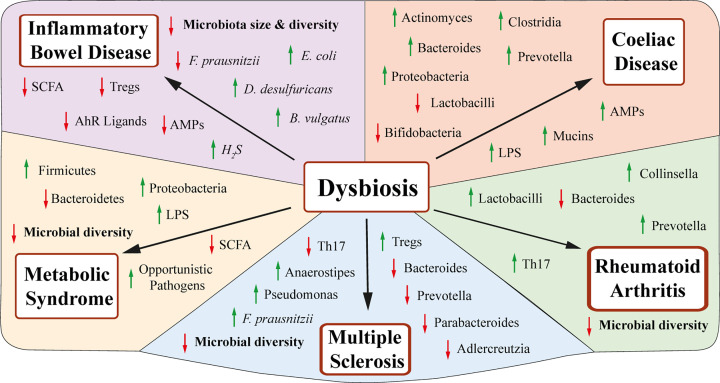
Environmental changes as well as host genetic susceptibility can contribute to dysbiosis [170,171]. In a dysbiotic state, altered relative abundances of certain microbial species and/or microbiota-derived metabolites can lead to the disruption of intestinal barrier integrity and host immune responses. Dysregulated mucosal immune responses are often characterised by an up-regulation of Th1, Th2, and Th17 cells and a down-regulation of Tregs and IgA [172,173]. Dysbiosis is linked to the development of numerous disease states including IBD, rheumatoid arthritis (RA), multiple sclerosis (MS), and metabolic syndrome [172,174] (Figure 4). However, it is worth noting that many of the studies to date, particularly those highlighting immunological pathways, have been solely based on findings from rodent models, which have inherent limitations [175].
Figure 4. Linking dysbiosis and disease.
Several diseases have been linked to dysbiosis. A dysbiotic state is often characterised by a loss of beneficial microbes, increased levels of pathobionts, and a decrease in microbial diversity. Changes in relative bacterial abundance, as well as microbe-derived metabolites, are thought to cause dysregulation in host gut permeability, leading to a compromised immune response and in turn the development of disease.
IBD
IBD is an umbrella term encompassing a group of complex chronic inflammatory disorders of the gastrointestinal tract [176]. Most commonly in the form of Crohn’s disease (CD) and ulcerative colitis (UC), IBD has been associated with changes in gut microbiota. However, it is not clear whether these changes contribute to disease pathogenesis or develop because of disease-related inflammation. IBD patients exhibit a reduction in microbiota size, functional diversity, and stability compared with healthy controls. In general, the microbiome of IBD patients show a decrease in Firmicutes of the Clostridium leptum group, particularly Faecalibacterium prausnitzii (F. prausnitzii), and an increase in Bacteroidetes and Proteobacteria such as Desulfovibrio desulfuricans (D. desulfricans) and E. coli [177–179]. On average, IBD patients harbour 25% less microbial genes than a healthy person [180].
The changes observed in the gut microbiome of IBD patients have been linked to bacteria known to have a role in either suppressing or promoting inflammation. Individuals with CD have a lower abundance of F. prausnitzii, an SCFA-producing bacterium, that promotes good gut health through up-regulation of Tregs and anti-inflammatory cytokines [181,182]. In humans, a reduction in F. prausnitzii is associated with an increased risk of postoperative recurrence of CD [182]. Furthermore, in IBD patients, an increase in the abundance of sulphate-reducing bacteria, such as D. desulfuricans, is attributed to increased production of hydrogen sulphate, which can damage IECs and in turn induce mucosal inflammation [183,184]. Several human studies have also reported a mucosa-associated E. coli richness in CD patients [179,185], leading to increased gut permeability and inflammation [186]. Both human and murine models have found that a reduction in tryptophan levels are also associated with IBD [187,188]. In IBD patients, tryptophan serum levels were found to inversely correlate with IL-22 levels and disease activity [187].
Coeliac disease
Coeliac disease, prevalent in 1–2% of the global population, is an immune-mediated inflammatory disorder that primarily affects the small intestine and is initiated following ingestion of gluten in genetically predisposed individuals [189,190]. Research has suggested that dysbiosis plays a role in triggering coeliac disease with a dysregulated immune response and failure to maintain intestinal barrier integrity, leading to mucosal inflammation [191]. However, like IBD, it remains unclear as to whether the dysbiotic state characteristic of coeliac disease is a cause or consequence of a dysregulated immune response.
As coeliac disease generally presents in childhood and young adulthood, most studies looking at a link between coeliac disease and the microbiome have focused on children [191]. Rod-shaped bacteria, including Clostridia, Provotella, and Actinomyces, are more frequently found in the small bowel of children with active coeliac compared with healthy controls [192]. Whilst no consistent microbial signature has been determined for patients with coeliac disease, most studies report an imbalance between Gram-negative and Gram-positive bacteria, characterised by both an increase in Gram-negative Bacteroides and Proteobacteria, and a decrease in Gram-positive Lactobacilli and Bifidobacteria, which have a protective anti-inflammatory effect [193,194]. Experimental murine models have reported that some Bacteroidetes species are involved in the disruption of intestinal barrier integrity, exhibiting pro-inflammatory effects [46,195,196]. Both mice and human studies have shown that Lactobacilli and Bifidobacteria may play a role in modifying the immunogenic potential of gluten, through breakdown of both gluten and its peptide derivatives [197,198]. For example, Lactobacilli can detoxify gliadin peptides after their partial digestion by human proteases. Both mice and human studies report that Bifidobacterium strains also play a role in reducing the epithelial permeability triggered by gluten, diminishing pro-inflammatory cytokine synthesis and decreasing jejunal barrier damage [199–201].
Whilst the exact mechanisms involved in coeliac disease remain unclear, studies in mice have shown that a dysbiotic microbiota can result in increased levels of LPS in the intestine, which result in a dysregulation of the immune response through the activation of both IELs and IECs, triggering the production of AMPs and mucin [202,203]. Additionally, mouse studies have linked alterations in microbial metabolites to the induction of Treg cells and dendritic cells, which produce IL-10 and retinoic acid and thereby contribute to the activation of various cellular inflammatory processes within the lamina propria [158,204].
Other autoimmune diseases
RA is a systemic autoimmune disorder that results in joint destruction, affecting approximately 0.5–1% of the global population [205]. Patients with RA exhibit decreased gut microbial diversity and microbial gut dysbiosis characterised by an abundance of Prevotella, Lactobacilli, and Collinsella [206–208]. Mouse models show that Prevotella and Collinsella can induce a pro-inflammatory Th17 response and increase gut permeability [206]. Colonisation of K/BxN mice, an established RA model, with SFB was shown to induce Th17 cell proliferation, ultimately leading to the differentiation of B cells and the production of autoantibodies [209]. It is thought that these autoantibodies target joints, leading to the inflammation seen in RA.
MS is a neurodegenerative autoimmune disease that affects the central nervous system (CNS) [210]. Whilst a typical microbiota phenotype for MS has not yet been described, patients with active disease generally exhibit decreased species richness, an abundance of Anaerostipes, Faecalibacteria, and Psuedomonas, and decreased levels of Bacteroides, Prevotella, Parabacteroides and Adlercreutzia [211,212]. An autoimmune encephalomyelitis (EAE) GF mouse model, which is also a model for MS, showed lower levels of IL-17 in both the gut and CNS, and an increase in peripheral Tregs [213]. Furthermore, disease severity in EAE models is also closely related to altered intestinal permeability, reduced submucosa thickness, and altered TJ expression in IECs [214,215].
Metabolic syndrome
Metabolic syndrome describes a group of risk factors, including obesity, hyperglycaemia, hypertension, and dyslipidaemia, which can lead to the development of various conditions including cardiovascular disease. The pathogenesis of metabolic syndrome is linked to a variety of factors such as insulin resistance, chronic low-grade inflammation in metabolic tissue, and oxidative stress [216]. In recent years, gut dysbiosis has been identified as a risk factor for metabolic syndrome [217]. It is believed that environmental factors, such as a high-fat diet, linked to decreased microbial diversity, promotes both general and metabolic tissue inflammation that may lead to the development of metabolic syndrome.
The links between the gut microbiota and both obesity and type 2 diabetes (T2D) have been most extensively studied. Studies in mice have shown that transplantation of gut microbiota from obese to lean GF mice resulted in an obesogenic phenotype [218]. Furthermore, GF mice fed a high-fat, high-sugar diet were found to be resistant to weight gain [219]. Studies in humans suggest that compared with lean individuals, obese individuals have increased levels of Firmicutes and decreased levels of Bacteroidetes [220,221]. In both humans and murine models, Roux-en-Y gastric bypass surgery has been found to rapidly change the gut microbiota, with gut microbiota normalising close to non-obese controls [222,223]. Patients with T2D typically exhibit reduced microbiome diversity, reduced SCFA-producing bacteria, and an increased number of opportunistic pathogens [224]. Rodent studies have found that SCFA play a key role in metabolic disorders, particularly in obesity and T2D. The SCFAs, propionate and acetate, were found to influence gut motility, intestinal transit rate, and caloric energy extraction from the diet through GPR41 activation [225]. Increased insulin sensitivity and increased satiety was also observed in mouse models, thought to be linked to the induction of glucagon-like peptide (GLP)-1 secretion through the activation of GPR43 and GPR41 [226]. Butyrate provides energy to enterocytes by exerting a trophic effect and inducing GLP-2 synthesis that in turn strengthens the gut barrier function [227].
It has been suggested that the gut microbial dysbiosis experienced in metabolic disorders leads to impaired intestinal cell function and increased gut permeability, partly induced by a high-fat diet [228]. Rodent studies have reported an increase in Gram-negative bacteria, including Proteobacteria, leading to a local increase in LPS in the mucosal layer [229,230]. MAMPs and microbiota-derived metabolites, including LPS, can translocate through the epithelial layer and reach the lamina propria where they are internalised by phagocytes. Furthermore, it has been hypothesised that microbial gut dysbiosis impairs communication between phagocytes and other immune cells in animal models, allowing the translocation of bacterial components to metabolic tissue [231,232]. In the metabolic tissue of mice, bacterial components trigger inflammation by promoting the proliferation of preadipocytes and macrophages, increasing ILC3 frequency and increasing the infiltration of B and T lymphocytes. Associated pro-inflammatory cytokines can also contribute to reduced insulin signalling, exacerbating the effects of diabetes.
Microbiota-targeted therapies
As detailed previously, dysbiosis in the gut is implicated in multiple gastrointestinal and non-gastrointestinal diseases. Intervention aiming to ameliorate this pathological environment with the delivery of targeted beneficial or wholesale bacterial populations in the form of probiotics and faecal microbiota transplantation (FMT), respectively, has been in clinical practice for many years [233,234]. The various mechanisms by which probiotics and FMT exert their therapeutic effect has been reviewed in detail elsewhere [235–237], but centre their interaction with the host mucosal immune system via MAMPs [238,239] or extracellular vesicles [240–242], the surrounding microbiota via AMPs [243], microbial cross-feeding [244] or nutrient competition, and their contribution to the broader mucosal metabolic environment [118,148]. Here we review the latest developments and innovations in probiotics and FMT.
Probiotics
The main probiotic genera, including Lactobacilli, Bifidobacteria, Saccharomyces, and Streptococci, as well as combination commercial probiotics, have been researched extensively and touted as potential therapies for many diseases or symptoms [245]. Certain probiotic strains have discrete effects on mucosal immune function, such as that seen with Lactobacillus plantarum TIFN1010 which modulates gene transcription pathways related to cell-cell adhesion and mucosal healing processes [238]. However, robust clinical data to support their use remain limited, with systematic reviews in IBD [246–248], Irritable bowel syndrome (IBS) [249] and C. difficile-associated diarrhoea (CDAD) [250] showing neutral or only qualified evidence for use. Similarly, practice guidelines do not recommend the routine use of probiotics [233], partly due to uncertainty regarding species or strain-dependent effects [251,252].
Recent advances in genomic sequencing and metabolic modelling have offered a way to reduce microbial uncertainty and the chance to optimise probiotic use through genetic engineering tools such as CRISPR-Cas [253–256]. For example, genetic modification of Lactobacillus casei (L. casei) to overexpress the mcra gene, and so enhance bioactive compound production, such as conjugate linoleic acid, can result in elimination of Campylobacter jejuni, an important diarrhoea-associated pathogen [257]. As well as optimising established probiotic species such as L. casei, confirmation of species such as Akkermansia muciniphilia (A. muciniphilia) as probiotic therapeutic candidates has become possible through the use of genome-scale modelling. Using this method, the complete microbial genome sequence can be screened to predict genes that influence particular metabolic pathways [258]. For A. muciniphilia, genes linked to sugar degradation and vitamin biosynthesis, as well as SCFA production, were predicted using this approach and validated by transcriptomic and proteomic analysis in vitro [259]. Antibiotic resistance and metabolic variation can also be assessed by whole-genome assembly undertaken on patient-derived stool samples [260]. Genomic sequencing technology has also been used to identify individuals resistant to probiotic colonisation at the mucosal level [261], allowing therapy to then be tailored, reducing treatment variability currently seen with probiotics [262].
Overall, whilst significant advances in probiotic therapy have been made, there is a need for a greater understanding of probiotic formulation, in addition to a requirement for more robust human clinical trial data to justify its routine use.
Postbiotics
An important additional consideration regarding probiotic preparations is the intrinsic effect of microbial cell surface components and metabolites. Whereas, by definition, probiotics are live microorganisms [263], there is also a role for postbiotics, as inanimate microorganisms and/or their components [264], prepared specifically for their health benefits on the host. These narrow criteria exclude purified microbial metabolites applied in isolation and instead focus on thermal inactivation and quantification of products that possess microbial effector molecules such as bile salt hydrolase [265] and exopolysaccharides [239].
Murine studies have shown the effect of postbiotics on gastrointestinal mucosa in a Citrobacter-induced colitis model [266], whereas the mechanistic impact on mucosal inflammation in humans is more limited to specific metabolites such as butyrate as in the case of diversion colitis [267]. However, clinical studies focussing on subjective outcomes such as symptom scores have shown benefit of postbiotics in IBS [268].
To date, the application of postbiotics in gastrointestinal disease remains limited with the mainstay of evidence [269,270] and regulation [271], centred on secondary prevention of respiratory infections. Further mechanistic and clinical trial data are required to characterise the effect of specific postbiotics on gastrointestinal inflammation.
FMT
FMT, the delivery of donor stool into the gastrointestinal tract of a patient, is an established and guideline-supported intervention for rCDI [234,272,273], independent of route of delivery [274], and a potential option in severe primary CDI [275]. Meta-analysis has indicated a positive association between FMT and the treatment of IBD, particularly with active UC [276,277]. Further trials are currently underway [278] to confirm FMT efficacy before being adopted into routine clinical practice [279]. Similarly, with CD, there is evidence supporting the benefits of FMT [280], however, it has not yet been recommended for clinical use [281]. Evidence remains lacking for routine use of FMT in IBS [282] with evidence for only conditional use in metabolic syndrome [283,284] and hepatic encephalopathy [285]. The use of FMT in non-gastrointestinal diseases is an area of ongoing study with randomised clinical trials in type 1 diabetes showing promise [286]. FMT clinicals trials are also underway to assess effectivity in treating Coeliac disease (NCT 04014413), RA (NCT03944096), Sjogren’s syndrome (NCT03926286) and MS (NCT03183869; NCT03975413; NCT04150549), building upon prior animal and uncontrolled human studies [287]. It is in malignancy, and specifically anticancer immunotherapies, where microbiota and their manipulation have shown great promise, building on evidence that certain genera, for example, Bifidobacteria [288] or Bacteroides [289], can affect the efficacy of malignant melanoma treatments. A recent clinical trial revealed that some patients refractory to anti-programmed cell death protein 1 (PD-1) immunotherapy could overcome this resistance to therapy by undergoing FMT from donors who were responders to the same anti-PD-1 immunotherapy [290]. PD-1 is an immune checkpoint receptor on T cells that prevents overstimulation of immune responses and contributes to the maintenance of immune tolerance to self-antigens. The fact that FMT impacts on anti-PD-1 melanoma therapy demonstrates that the composition of the microbiota influences host systemic immune responses. FMT is now being applied to metastatic hormone-resistant prostate cancer (NCT04116775) and to ameliorate chemotherapy-induced toxicity (NCT04040712).
An important factor in FMT is the role of viruses and mycobiota given that whole stool transplantation involves a transfer of these microorganisms to the new host along with bacteria. Bacteriophages contribute to host immunity by adhering to mucosal mucus creating an additional antimicrobial layer that reduces bacterial attachment and colonisation of the mucosa [291]. Both Caudovirales [292] and Saccharomyces [293] have been shown as important drivers for successful treatment of rCDI by FMT. Faecal filtrate transfer (FFT), a supernatant composed of bacterial debris, AMPs, metabolic products and oligonucleotides, but not live bacteria, was also seen to improve outcomes in rCDI [294].
A limitation of widespread FMT use outside of the trial setting is the conceptual acceptability of single or pooled donor stool being transferred to a patient. Synthetic microbiomes can be cultured from donors and purified, or compiled from metagenomic studies [295]. Purified intestinal bacterial culture have been shown to be as effective in treating rCDI in a proof-of-principle study [296]. A recent randomised-controlled trial reported that a 12-strain bacterial mixture cultured from donor stool was inferior to conventional FMT but equivalent to using vancomycin for the treatment of rCDI [297]. FMT using freeze-dried or lyophilised matter has been shown in observational studies to also be effective in treating rCDI [298], with a propagated, lyophilised and encapsulated formulation currently under investigation in clinical trials for the treatment of rCDI (NCT02865616), UC (NCT03832400), and other diseases. These technologies, if efficacy is confirmed, herald the opportunity of a ‘post-FMT’ treatment model centred on highly selected donors yielding a purified, standardised, and cryopreserved microbiota preparation for systematic clinical use.
Conclusion
The microbiome is a metabolically and immunologically active presence within the gastrointestinal tract that plays a vital role in the maintenance of human health. This population of highly diverse microorganisms is shaped by numerous factors, most notably, diet and the use of medications such as antibiotics. The intestinal mucosa provides an important interface between the microbiota and host, where the microbiota not only aids in development of effective host immune responses against pathogens and injury but also limits excessive mucosal inflammation to promote tolerance and stability of the gut environment. Microbial components and microbe-derived metabolites contribute to both mucosal barrier integrity and the regulation of underlying immune responses to preserve intestinal homoeostasis. When there is a loss of this balanced relationship, as seen in dysbiosis, then there is a risk of sustained pathogenic inflammation and the development of numerous diseases. As our understanding of the microbiome and microbiota–host interactions has improved, so has our ability to harness members of the microbiota to reverse dysbiosis, reduce mucosal inflammation, and prevent disease progression. The outcome of ongoing clinical trials and mechanistic studies will hopefully extend our current knowledge of the microbiome and further our understanding of the role it plays in mucosal inflammation.
Abbreviations
- AhR
aryl hydrocarbon receptor
- AMP
antimicrobial peptide
- CD
Crohn’s disease
- CNS
central nervous system
- CREB
cAMP response element-binding protein
- C-section
caesarean section
- EAE
autoimmune encephalomyelitis
- ERK
extracellular signal-regulated kinase
- FMT
faecal microbiota transplantation
- GF
germ-free
- GLP
glucagon-like peptide
- GPR
G-protein coupled receptor
- HDAC
histone deacetylase
- IBD
inflammatory bowel disease
- IBS
irritable bowel syndrome
- IEC
intestinal epithelial cell
- IEL
intra-epithelial lymphocyte
- IL
interleukin
- ILC
innate lymphoid cell
- ILC3
intestinal group 3 innate lymphoid cell
- LPS
lipopolysaccharide
- MAIT
mucosal-associated invariant T
- MAMP
microbe-associated molecular pattern
- MS
multiple sclerosis
- NLR
nucleotide-binding oligomerisation domain (NOD)-like receptor
- NLRP
NOD-, leucine-rich repeat (LRR)-, and pyrin domain containing
- NOD
nucleotide-binding oligomerisation domain
- PD-1
programmed cell death protein 1
- PGN
peptidoglycan
- PPI
proton pump inhibitor
- PRR
pattern-recognition receptor
- PSA
polysaccharide A
- RA
rheumatoid arthritis
- rCDI
recurrent Clostridium difficile infection
- ROS
reactive oxygen species
- SCFA
short-chain fatty acid
- Th
T helper
- TJ
tight junction
- TLR
Toll-like receptor
- TMAO
trimethylamine N-oxide
- TNF-α
tumour necrosis factor-α
- Treg
regulatory T
- T2D
type 2 diabetes
- UC
ulcerative colitis
Competing Interests
The authors declare that there are no competing interests associated with the manuscript.
Funding
This work was supported by the NIHR Biomedical Research Centre at University College London Hospital NHS Foundation and University College London [grant number BRC727/OHD/AS/110380 (to D.J.S. and A.M.S.)]; and the Crohn’s & Colitis U.K. Fellowship [grant number 2019-4 Smith (Sebepos-Rogers); award number 179344 (to G.S.-R.)].
Open Access
Open access for this article was enabled by the participation of University College London in an all-inclusive Read & Publish pilot with Portland Press and the Biochemical Society under a transformative agreement with JISC.
Author Contribution
D.J.S., S.I., and G.S.-R. wrote the manuscript. D.J.S. produced the figures. A.M.S. and F.Z.R. revised the manuscript. All authors read and approved the final manuscript.
References
- 1.Dethlefsen L., McFall-Ngai M. and Relman D.A. (2007) An ecological and evolutionary perspective on human-microbe mutualism and disease. Nature 449, 811–818 10.1038/nature06245 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Turnbaugh P.J., Ley R.E., Hamady M., Fraser-Liggett C.M., Knight R. and Gordon J.I. (2007) The human microbiome project. Nature 449, 804–810 10.1038/nature06244 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sender R., Fuchs S. and Milo R. (2016) Revised estimates for the number of human and bacteria cells in the body. PLoS Biol. 14, e1002533 10.1371/journal.pbio.1002533 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Koskinen K., Pausan M.R., Perras A.K., Beck M., Bang C., Mora M.et al. (2017) First insights into the diverse human archaeome: specific detection of archaea in the gastrointestinal tract, lung, and nose and on skin. mBio 8, e00824–17 10.1128/mBio.00824-17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kumata R., Ito J., Takahashi K., Suzuki T. and Sato K. (2020) A tissue level atlas of the healthy human virome. BMC Biol. 18, 55 10.1186/s12915-020-00785-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ghannoum M.A., Jurevic R.J., Mukherjee P.K., Cui F., Sikaroodi M., Naqvi A.et al. (2010) Characterization of the oral fungal microbiome (mycobiome) in healthy individuals. PLoS Pathog. 6, e1000713 10.1371/journal.ppat.1000713 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Nieves-Ramirez M.E., Partida-Rodriguez O., Laforest-Lapointe I., Reynolds L.A., Brown E.M., Valdez-Salazar A.et al. (2018) Asymptomatic intestinal colonization with protist blastocystis is strongly associated with distinct microbiome ecological patterns. mSystems 3, e00007–18, 10.1128/mSystems.00007-18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sender R., Fuchs S. and Milo R. (2016) Are we really vastly outnumbered? Revisiting the ratio of bacterial to host cells in humans. Cell 164, 337–340 10.1016/j.cell.2016.01.013 [DOI] [PubMed] [Google Scholar]
- 9.Tierney B.T., Yang Z., Luber J.M., Beaudin M., Wibowo M.C., Baek C.et al. (2019) The landscape of genetic content in the gut and oral human microbiome. Cell Host Microbe 26, 283.e8–295.e8 10.1016/j.chom.2019.07.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bharti R. and Grimm D.G. (2019) Current challenges and best-practice protocols for microbiome analysis. Brief. Bioinform. 10.1093/bib/bbz155 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Integrative HMPRNC (2019) The Integrative Human Microbiome Project. Nature 569, 641–648 10.1038/s41586-019-1238-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zhao L., Ni Y., Su M., Li H., Dong F., Chen W.et al. (2017) High throughput and quantitative measurement of microbial metabolome by gas chromatography/mass spectrometry using automated alkyl chloroformate derivatization. Anal. Chem. 89, 5565–5577 10.1021/acs.analchem.7b00660 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mas-Lloret J., Obon-Santacana M., Ibanez-Sanz G., Guino E., Pato M.L., Rodriguez-Moranta F.et al. (2020) Gut microbiome diversity detected by high-coverage 16S and shotgun sequencing of paired stool and colon sample. Sci. Data 7, 92 10.1038/s41597-020-0427-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tauzin A.S., Pereira M.R., Van Vliet L.D., Colin P.Y., Laville E., Esque J.et al. (2020) Investigating host-microbiome interactions by droplet based microfluidics. Microbiome 8, 141 10.1186/s40168-020-00911-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wilkins L.J., Monga M. and Miller A.W. (2019) Defining dysbiosis for a cluster of chronic diseases. Sci. Rep. 9, 12918 10.1038/s41598-019-49452-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Rothschild D., Weissbrod O., Barkan E., Kurilshikov A., Korem T., Zeevi D.et al. (2018) Environment dominates over host genetics in shaping human gut microbiota. Nature 555, 210–215 10.1038/nature25973 [DOI] [PubMed] [Google Scholar]
- 17.Shaw L., Ribeiro A.L.R., Levine A.P., Pontikos N., Balloux F., Segal A.W.et al. (2017) The human salivary microbiome is shaped by shared environment rather than genetics: evidence from a large family of closely related individuals. mBio 8, e01237–17, 10.1128/mBio.01237-17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Luissint A.C., Parkos C.A. and Nusrat A. (2016) Inflammation and the intestinal barrier: leukocyte-epithelial cell interactions, cell junction remodeling, and mucosal repair. Gastroenterology 151, 616–632 10.1053/j.gastro.2016.07.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Jalanka-Tuovinen J., Salonen A., Nikkilä J., Immonen O., Kekkonen R., Lahti L.et al. (2011) Intestinal microbiota in healthy adults: temporal analysis reveals individual and common core and relation to intestinal symptoms. PLoS ONE 6, e23035 10.1371/journal.pone.0023035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Eckburg P.B., Bik E.M., Bernstein C.N., Purdom E., Dethlefsen L., Sargent M.et al. (2005) Diversity of the human intestinal microbial flora. Science 308, 1635–1638 10.1126/science.1110591 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Qin J., Li R., Raes J., Arumugam M., Burgdorf K.S., Manichanh C.et al. (2010) A human gut microbial gene catalogue established by metagenomic sequencing. Nature 464, 59–65 10.1038/nature08821 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jung Y., Kerfahi D., Quang Pham H., Hyunwoo S., Ibal J., Park M.et al. (2020) Although host-related factors are important for the formation of gut microbiota, environmental factors cannot be ignored, bioRxiv 10.1101/2020.02.03.93331 [DOI] [Google Scholar]
- 23.Kim M. and Benayoun B.A. (2020) The microbiome: an emerging key player in aging and longevity. Transl. Med. Aging 4, 103–116 [PMC free article] [PubMed] [Google Scholar]
- 24.Nagpal R., Mainali R., Ahmadi S., Wang S., Singh R., Kavanagh K.et al. (2018) Gut microbiome and aging: Physiological and mechanistic insights. Nutr. Healthy Aging 4, 267–285 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gupta V.K., Paul S. and Dutta C. (2017) Geography, ethnicity or subsistence-specific variations in human microbiome composition and diversity. Front. Microbiol. 8, 1162, 10.3389/fmicb.2017.01162 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Reyman M., van Houten M.A., van Baarle D., Bosch A.A.T.M., Man W.H., Chu M.L.J.N.et al. (2019) Impact of delivery mode-associated gut microbiota dynamics on health in the first year of life. Nat. Commun. 10, 4997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Rutayisire E., Huang K., Liu Y. and Tao F. (2016) The mode of delivery affects the diversity and colonization pattern of the gut microbiota during the first year of infants’ life: a systematic review. BMC Gastroenterol. 16, 86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Rodríguez J.M., Murphy K., Stanton C., Ross R.P., Kober O.I., Juge N.et al. (2015) The composition of the gut microbiota throughout life, with an emphasis on early life. Microb. Ecol. Health Dis. 26, 26050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Leeming E.R., Johnson A.J., Spector T.D. and Le Roy C.I. (2019) Effect of diet on the gut microbiota: rethinking intervention duration. Nutrients 11, 2862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Zinöcker M.K. and Lindseth I.A. (2018) The Western diet-microbiome-host interaction and its role in metabolic disease. Nutrients 10, 365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hasegawa Y., Chen S.-Y., Sheng L., Jena P.K., Kalanetra K.M., Mills D.A.et al. (2020) Long-term effects of western diet consumption in male and female mice. Sci. Rep. 10, 14686 10.1038/s41598-020-71592-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.De Filippis F., Pellegrini N., Vannini L., Jeffery I.B., La Storia A., Laghi L.et al. (2016) High-level adherence to a Mediterranean diet beneficially impacts the gut microbiota and associated metabolome. Gut 65, 1812 10.1136/gutjnl-2015-309957 [DOI] [PubMed] [Google Scholar]
- 33.Bäckhed F., Roswall J., Peng Y., Feng Q., Jia H., Kovatcheva-Datchary P.et al. (2015) Dynamics and stabilization of the human gut microbiome during the first year of life resource. Cell Host Microbe 17, 690–703, 10.1016/j.chom.2015.04.004 [DOI] [PubMed] [Google Scholar]
- 34.Rautava S. (2016) Early microbial contact, the breast milk microbiome and child health. J. Dev. Origins Health Dis. 7, 5–14 10.1017/S2040174415001233 [DOI] [PubMed] [Google Scholar]
- 35.Lawson M.A.E., O’Neill I.J., Kujawska M., Gowrinadh Javvadi S., Wijeyesekera A., Flegg Z.et al. (2020) Breast milk-derived human milk oligosaccharides promote Bifidobacterium interactions within a single ecosystem. ISME J. 14, 635–648 10.1038/s41396-019-0553-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Marcobal A., Barboza M., Froehlich J.W., Block D.E., German J.B., Lebrilla C.B.et al. (2010) Consumption of human milk oligosaccharides by gut-related microbes. J. Agric. Food Chem. 58, 5334–5340 10.1021/jf9044205 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Lyons K.E., Ryan C.A., Dempsey E.M., Ross R.P. and Stanton C. (2020) Breast milk, a source of beneficial microbes and associated benefits for infant health. Nutrients 12, 1039, 10.3390/nu12041039 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Fallani M., Amarri S., Uusijarvi A., Adam R., Khanna S., Aguilera M.et al. (2011) Determinants of the human infant intestinal microbiota after the introduction of first complementary foods in infant samples from five European centres. Microbiology 157, 1385–1392 10.1099/mic.0.042143-0 [DOI] [PubMed] [Google Scholar]
- 39.Wu G.D., Chen J., Hoffmann C., Bittinger K., Chen Y.Y., Keilbaugh S.A.et al. (2011) Linking long-term dietary patterns with gut microbial enterotypes. Science 334, 105–108 10.1126/science.1208344 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Reddy B.S., Weisburger J.H. and Wynder E.L. (1975) Effects of high risk and low risk diets for colon carcinogenesis on fecal microflora and steroids in man. J. Nutr. 105, 878–884 10.1093/jn/105.7.878 [DOI] [PubMed] [Google Scholar]
- 41.Drasar B.S., Crowther J.S., Goddard P., Hawksworth G., Hill M.J., Peach S.et al. (1973) The relation between diet and the gut microflora in man. Proc. Nutr. Soc. 32, 49–52 10.1079/PNS19730014 [DOI] [PubMed] [Google Scholar]
- 42.Bisanz J.E., Upadhyay V., Turnbaugh J.A., Ly K. and Turnbaugh P.J. (2019) Meta-analysis reveals reproducible gut microbiome alterations in response to a high-fat diet. Cell Host Microbe 26, 265.e4–272.e4 10.1016/j.chom.2019.06.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Moreira A.P., Texeira T.F., Ferreira A.B., Peluzio Mdo C. and Alfenas Rde C. (2012) Influence of a high-fat diet on gut microbiota, intestinal permeability and metabolic endotoxaemia. Br. J. Nutr. 108, 801–809 10.1017/S0007114512001213 [DOI] [PubMed] [Google Scholar]
- 44.Gerasimidis K., Bryden K., Chen X., Papachristou E., Verney A., Roig M.et al. (2020) The impact of food additives, artificial sweeteners and domestic hygiene products on the human gut microbiome and its fibre fermentation capacity. Eur. J. Nutr. 59, 3213–3230 10.1007/s00394-019-02161-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Partridge D., Lloyd K.A., Rhodes J.M., Walker A.W., Johnstone A.M. and Campbell B.J. (2019) Food additives: Assessing the impact of exposure to permitted emulsifiers on bowel and metabolic health - introducing the FADiets study. Nutr. Bull. 44, 329–349 10.1111/nbu.12408 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Sydora B.C., MacFarlane S.M., Walker J.W., Dmytrash A.L., Churchill T.A., Doyle J.et al. (2007) Epithelial barrier disruption allows nondisease-causing bacteria to initiate and sustain IBD in the IL-10 gene-deficient mouse. Inflamm. Bowel Dis. 13, 947–954 10.1002/ibd.20155 [DOI] [PubMed] [Google Scholar]
- 47.Del Chierico F., Vernocchi P., Dallapiccola B. and Putignani L. (2014) Mediterranean diet and health: food effects on gut microbiota and disease control. Int. J. Mol. Sci. 15, 11678–11699 10.3390/ijms150711678 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Garcia-Mantrana I., Selma-Royo M., Alcantara C. and Collado M.C. (2018) Shifts on gut microbiota associated to mediterranean diet adherence and specific dietary intakes on general adult population. Front. Microbiol. 9, 890, 10.3389/fmicb.2018.00890 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Heiman M.L. and Greenway F.L. (2016) A healthy gastrointestinal microbiome is dependent on dietary diversity. Mol. Metab. 5, 317–320 10.1016/j.molmet.2016.02.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zarrinpar A., Chaix A., Yooseph S. and Panda S. (2014) Diet and feeding pattern affect the diurnal dynamics of the gut microbiome. Cell Metab. 20, 1006–1017 10.1016/j.cmet.2014.11.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kaczmarek J.L., Musaad S.M. and Holscher H.D. (2017) Time of day and eating behaviors are associated with the composition and function of the human gastrointestinal microbiota. Am. J. Clin. Nutr. 106, 1220–1231 10.3945/ajcn.117.156380 [DOI] [PubMed] [Google Scholar]
- 52.De Filippis F., Vitaglione P., Cuomo R., Berni Canani R. and Ercolini D. (2018) Dietary interventions to modulate the gut microbiome—how far away are we from precision medicine. Inflamm. Bowel Dis. 24, 2142–2154, 10.1093/ibd/izy080 [DOI] [PubMed] [Google Scholar]
- 53.Zhang C., Li S., Yang L., Huang P., Li W., Wang S.et al. (2013) Structural modulation of gut microbiota in life-long calorie-restricted mice. Nat. Commun. 4, 2163 10.1038/ncomms3163 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Santacruz A., Marcos A., Wärnberg J., Martí A., Martin-Matillas M., Campoy C.et al. (2009) Interplay between weight loss and gut microbiota composition in overweight adolescents. Obesity 17, 1906–1915 10.1038/oby.2009.112 [DOI] [PubMed] [Google Scholar]
- 55.Kurup K., Matyi S., Giles C.B., Wren J.D., Jones K., Ericsson A.et al. (2021) Calorie restriction prevents age-related changes in the intestinal microbiota. Aging (Albany N.Y.) 13, 6298–6329 10.18632/aging.202753 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Zheng X., Wang S. and Jia W. (2018) Calorie restriction and its impact on gut microbial composition and global metabolism. Front. Med. 12, 634–644 10.1007/s11684-018-0670-8 [DOI] [PubMed] [Google Scholar]
- 57.Klingensmith N.J. and Coopersmith C.M. (2016) The gut as the motor of multiple organ dysfunction in critical illness. Crit. Care Clin. 32, 203–212 10.1016/j.ccc.2015.11.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Nguyen L.H., Örtqvist A.K., Cao Y., Simon T.G., Roelstraete B., Song M.et al. (2020) Antibiotic use and the development of inflammatory bowel disease: a national case-control study in Sweden. Lancet Gastroenterol. Hepatol. 5, 986–995 10.1016/S2468-1253(20)30267-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Kim D.H., Han K. and Kim S.W. (2018) Effects of antibiotics on the development of asthma and other allergic diseases in children and adolescents. Allergy Asthma Immunol. Res. 10, 457–465 10.4168/aair.2018.10.5.457 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Bhalodi A.A., van Engelen T.S.R., Virk H.S. and Wiersinga W.J. (2019) Impact of antimicrobial therapy on the gut microbiome. J. Antimicrob. Chemother. 74, i6–i15 10.1093/jac/dky530 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Panda S., El khader I., Casellas F., López Vivancos J., García Cors M., Santiago A.et al. (2014) Short-term effect of antibiotics on human gut microbiota. PLoS ONE 9, e95476 10.1371/journal.pone.0095476 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Scott N.A., Andrusaite A., Andersen P., Lawson M., Alcon-Giner C., Leclaire C.et al. (2018) Antibiotics induce sustained dysregulation of intestinal T cell immunity by perturbing macrophage homeostasis. Sci. Transl. Med. 10, 10.1126/scitranslmed.aao4755 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Jernberg C., Löfmark S., Edlund C. and Jansson J.K. (2007) Long-term ecological impacts of antibiotic administration on the human intestinal microbiota. ISME J. 1, 56–66 10.1038/ismej.2007.3 [DOI] [PubMed] [Google Scholar]
- 64.Jakobsson H.E., Jernberg C., Andersson A.F., Sjölund-Karlsson M., Jansson J.K. and Engstrand L. (2010) Short-term antibiotic treatment has differing long-term impacts on the human throat and gut microbiome. PLoS ONE 5, e9836 10.1371/journal.pone.0009836 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Dethlefsen L. and Relman D.A. (2011) Incomplete recovery and individualized responses of the human distal gut microbiota to repeated antibiotic perturbation. Proc. Natl. Acad. Sci. U.S.A. 108, 4554–4561 10.1073/pnas.1000087107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Isaac S., Scher J.U., Djukovic A., Jiménez N., Littman D.R., Abramson S.B.et al. (2017) Short- and long-term effects of oral vancomycin on the human intestinal microbiota. J. Antimicrob. Chemother. 72, 128–136 10.1093/jac/dkw383 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Vrieze A., Out C., Fuentes S., Jonker L., Reuling I., Kootte R.S.et al. (2014) Impact of oral vancomycin on gut microbiota, bile acid metabolism, and insulin sensitivity. J. Hepatol. 60, 824–831 10.1016/j.jhep.2013.11.034 [DOI] [PubMed] [Google Scholar]
- 68.Vich Vila A., Collij V., Sanna S., Sinha T., Imhann F., Bourgonje A.R.et al. (2020) Impact of commonly used drugs on the composition and metabolic function of the gut microbiota. Nat. Commun. 11, 362 10.1038/s41467-019-14177-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Weersma R.K., Zhernakova A. and Fu J. (2020) Interaction between drugs and the gut microbiome. Gut 69, 1510–1519 10.1136/gutjnl-2019-320204 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Dominguez-Bello M.G., Costello E.K., Contreras M., Magris M., Hidalgo G., Fierer N.et al. (2010) Delivery mode shapes the acquisition and structure of the initial microbiota across multiple body habitats in newborns. Proc. Natl. Acad. Sci. U.S.A. 107, 11971–11975 10.1073/pnas.1002601107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Mackie R.I., Sghir A. and Gaskins H.R. (1999) Developmental microbial ecology of the neonatal gastrointestinal tract. Am. J. Clin. Nutr. 69, 1035s–1045s 10.1093/ajcn/69.5.1035s [DOI] [PubMed] [Google Scholar]
- 72.Cahenzli J., Koller Y., Wyss M., Geuking M.B. and McCoy K.D. (2013) Intestinal microbial diversity during early-life colonization shapes long-term IgE levels. Cell Host Microbe 14, 559–570 10.1016/j.chom.2013.10.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Jakobsson H.E., Abrahamsson T.R., Jenmalm M.C., Harris K., Quince C., Jernberg C.et al. (2014) Decreased gut microbiota diversity, delayed Bacteroidetes colonisation and reduced Th1 responses in infants delivered by caesarean section. Gut 63, 559–566 10.1136/gutjnl-2012-303249 [DOI] [PubMed] [Google Scholar]
- 74.Stewart C.J., Ajami N.J., O’Brien J.L., Hutchinson D.S., Smith D.P., Wong M.C.et al. (2018) Temporal development of the gut microbiome in early childhood from the TEDDY study. Nature 562, 583–588 10.1038/s41586-018-0617-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Agans R., Rigsbee L., Kenche H., Michail S., Khamis H.J. and Paliy O. (2011) Distal gut microbiota of adolescent children is different from that of adults. FEMS Microbiol. Ecol. 77, 404–412 10.1111/j.1574-6941.2011.01120.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Huttenhower C., Gevers D., Knight R., Abubucker S., Badger J.H., Chinwalla A.T.et al. (2012) Structure, function and diversity of the healthy human microbiome. Nature 486, 207 10.1038/nature11234 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Quigley E.M. (2013) Gut bacteria in health and disease. Gastroenterol. Hepatol. (N.Y.) 9, 560. [PMC free article] [PubMed] [Google Scholar]
- 78.Odamaki T., Kato K., Sugahara H., Hashikura N., Takahashi S., Xiao J.-Z.et al. (2016) Age-related changes in gut microbiota composition from newborn to centenarian: a cross-sectional study. BMC Microbiol. 16, 1–12 10.1186/s12866-016-0708-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Ragonnaud E. and Biragyn A. (2021) Gut microbiota as the key controllers of “healthy” aging of elderly people. Immun. Ageing 18, 2 10.1186/s12979-020-00213-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.De Filippo C., Cavalieri D., Di Paola M., Ramazzotti M., Poullet J.B., Massart S.et al. (2010) Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc. Natl. Acad. Sci. U.S.A. 107, 14691–14696 10.1073/pnas.1005963107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Grześkowiak Ł., Collado M.C., Mangani C., Maleta K., Laitinen K., Ashorn P.et al. (2012) Distinct gut microbiota in southeastern African and northern European infants. J. Pediatr. Gastroenterol. Nutr. 54, 812–816 10.1097/MPG.0b013e318249039c [DOI] [PubMed] [Google Scholar]
- 82.Gomez A., Petrzelkova Klara J., Burns Michael B., Yeoman Carl J., Amato Katherine R., Vlckova K.et al. (2016) Gut microbiome of coexisting BaAka pygmies and Bantu reflects gradients of traditional subsistence patterns. Cell Rep. 14, 2142–2153 10.1016/j.celrep.2016.02.013 [DOI] [PubMed] [Google Scholar]
- 83.Morton E.R., Lynch J., Froment A., Lafosse S., Heyer E., Przeworski M.et al. (2015) Variation in rural African gut microbiota is strongly correlated with colonization by Entamoeba and subsistence. PLoS Genet. 11, e1005658 10.1371/journal.pgen.1005658 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Greenhill A.R., Tsuji H., Ogata K., Natsuhara K., Morita A., Soli K.et al. (2015) Characterization of the gut microbiota of Papua New Guineans using reverse transcription quantitative PCR. PLoS ONE 10, e0117427 10.1371/journal.pone.0117427 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Obregon-Tito A.J., Tito R.Y., Metcalf J., Sankaranarayanan K., Clemente J.C., Ursell L.K.et al. (2015) Subsistence strategies in traditional societies distinguish gut microbiomes. Nat. Commun. 6, 6505 10.1038/ncomms7505 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Schnorr S.L., Candela M., Rampelli S., Centanni M., Consolandi C., Basaglia G.et al. (2014) Gut microbiome of the Hadza hunter-gatherers. Nat. Commun. 5, 3654 10.1038/ncomms4654 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Shreiner A., Huffnagle G.B. and Noverr M.C. (2008) The “Microflora Hypothesis” of allergic disease. Adv. Exp. Med. Biol. 635, 113–134 10.1007/978-0-387-09550-9_10 [DOI] [PubMed] [Google Scholar]
- 88.Jha A.R., Davenport E.R., Gautam Y., Bhandari D., Tandukar S., Ng K.M.et al. (2018) Gut microbiome transition across a lifestyle gradient in Himalaya. PLoS Biol. 16, e2005396 10.1371/journal.pbio.2005396 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Martino D. (2019) The effects of chlorinated drinking water on the assembly of the intestinal microbiome. Challenges 10, 10 10.3390/challe10010010 [DOI] [Google Scholar]
- 90.Mowat A.M. (2018) To respond or not to respond - a personal perspective of intestinal tolerance. Nat. Rev. Immunol. 18, 405–415 10.1038/s41577-018-0002-x [DOI] [PubMed] [Google Scholar]
- 91.Ohland C.L. and Jobin C. (2015) Microbial activities and intestinal homeostasis: a delicate balance between health and disease. Cell Mol. Gastroenterol. Hepatol. 1, 28–40 10.1016/j.jcmgh.2014.11.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Chu H. and Mazmanian S.K. (2013) Innate immune recognition of the microbiota promotes host-microbial symbiosis. Nat. Immunol. 14, 668–675 10.1038/ni.2635 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Takeuchi O. and Akira S. (2010) Pattern recognition receptors and inflammation. Cell 140, 805–820 10.1016/j.cell.2010.01.022 [DOI] [PubMed] [Google Scholar]
- 94.Fukata M. and Arditi M. (2013) The role of pattern recognition receptors in intestinal inflammation. Mucosal Immunol. 6, 451–463 10.1038/mi.2013.13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Rakoff-Nahoum S., Paglino J., Eslami-Varzaneh F., Edberg S. and Medzhitov R. (2004) Recognition of commensal microflora by toll-like receptors is required for intestinal homeostasis. Cell 118, 229–241 10.1016/j.cell.2004.07.002 [DOI] [PubMed] [Google Scholar]
- 96.Erturk-Hasdemir D., Oh S.F., Okan N.A., Stefanetti G., Gazzaniga F.S., Seeberger P.H.et al. (2019) Symbionts exploit complex signaling to educate the immune system. Proc. Natl. Acad. Sci. U.S.A., 116, 26157–26166 10.1073/pnas.1915978116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Nigro G., Rossi R., Commere P.H., Jay P. and Sansonetti P.J. (2014) The cytosolic bacterial peptidoglycan sensor Nod2 affords stem cell protection and links microbes to gut epithelial regeneration. Cell Host Microbe 15, 792–798 10.1016/j.chom.2014.05.003 [DOI] [PubMed] [Google Scholar]
- 98.Krautkramer K.A., Fan J. and Backhed F. (2021) Gut microbial metabolites as multi-kingdom intermediates. Nat. Rev. Microbiol. 19, 77–94 10.1038/s41579-020-0438-4 [DOI] [PubMed] [Google Scholar]
- 99.Okumura R. and Takeda K. (2018) Maintenance of intestinal homeostasis by mucosal barriers. Inflamm. Regen. 38, 5 10.1186/s41232-018-0063-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Pickard J.M., Zeng M.Y., Caruso R. and Nunez G. (2017) Gut microbiota: Role in pathogen colonization, immune responses, and inflammatory disease. Immunol. Rev. 279, 70–89 10.1111/imr.12567 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Zong X., Fu J., Xu B., Wang Y. and Jin M. (2020) Interplay between gut microbiota and antimicrobial peptides. Anim. Nutr. 6, 389–396 10.1016/j.aninu.2020.09.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Lee B., Moon K.M. and Kim C.Y. (2018) Tight junction in the intestinal epithelium: its association with diseases and regulation by phytochemicals. J. Immunol. Res. 2018, 2645465 10.1155/2018/2645465 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Bansil R. and Turner B.S. (2018) The biology of mucus: composition, synthesis and organization. Adv. Drug Deliv. Rev. 124, 3–15 10.1016/j.addr.2017.09.023 [DOI] [PubMed] [Google Scholar]
- 104.Paone P. and Cani P.D. (2020) Mucus barrier, mucins and gut microbiota: the expected slimy partners? Gut 69, 2232–2243 10.1136/gutjnl-2020-322260 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Johansson M.E., Larsson J.M. and Hansson G.C. (2011) The two mucus layers of colon are organized by the MUC2 mucin, whereas the outer layer is a legislator of host-microbial interactions. Proc. Natl. Acad. Sci. U.S.A. 108, 4659–4665 10.1073/pnas.1006451107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Lueschow S.R. and McElroy S.J. (2020) The Paneth cell: the curator and defender of the immature small intestine. Front. Immunol. 11, 587 10.3389/fimmu.2020.00587 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Hilchie A.L., Wuerth K. and Hancock R.E. (2013) Immune modulation by multifaceted cationic host defense (antimicrobial) peptides. Nat. Chem. Biol. 9, 761–768 10.1038/nchembio.1393 [DOI] [PubMed] [Google Scholar]
- 108.Mansour S.C., Pena O.M. and Hancock R.E. (2014) Host defense peptides: front-line immunomodulators. Trends Immunol. 35, 443–450 10.1016/j.it.2014.07.004 [DOI] [PubMed] [Google Scholar]
- 109.Tai E.K., Wong H.P., Lam E.K., Wu W.K., Yu L., Koo M.W.et al. (2008) Cathelicidin stimulates colonic mucus synthesis by up-regulating MUC1 and MUC2 expression through a mitogen-activated protein kinase pathway. J. Cell. Biochem. 104, 251–258 10.1002/jcb.21615 [DOI] [PubMed] [Google Scholar]
- 110.Robinson K., Deng Z., Hou Y. and Zhang G. (2015) Regulation of the intestinal barrier function by host defense peptides. Front. Vet. Sci. 2, 57 10.3389/fvets.2015.00057 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Schroeder B.O. (2019) Fight them or feed them: how the intestinal mucus layer manages the gut microbiota. Gastroenterol. Rep. 7, 3–12 10.1093/gastro/goy052 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Bansal T., Alaniz R.C., Wood T.K. and Jayaraman A. (2010) The bacterial signal indole increases epithelial-cell tight-junction resistance and attenuates indicators of inflammation. Proc. Natl. Acad. Sci. U.S.A. 107, 228–233 10.1073/pnas.0906112107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Shimada Y., Kinoshita M., Harada K., Mizutani M., Masahata K., Kayama H.et al. (2013) Commensal bacteria-dependent indole production enhances epithelial barrier function in the colon. PLoS ONE 8, e80604 10.1371/journal.pone.0080604 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Venkatesh M., Mukherjee S., Wang H., Li H., Sun K., Benechet A.P.et al. (2014) Symbiotic bacterial metabolites regulate gastrointestinal barrier function via the xenobiotic sensor PXR and Toll-like receptor 4. Immunity 41, 296–310 10.1016/j.immuni.2014.06.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Singh R., Chandrashekharappa S., Bodduluri S.R., Baby B.V., Hegde B., Kotla N.G.et al. (2019) Enhancement of the gut barrier integrity by a microbial metabolite through the Nrf2 pathway. Nat. Commun. 10, 89 10.1038/s41467-018-07859-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Roediger W.E. (1980) Role of anaerobic bacteria in the metabolic welfare of the colonic mucosa in man. Gut 21, 793–798 10.1136/gut.21.9.793 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Parada Venegas D., De la Fuente M.K., Landskron G., Gonzalez M.J., Quera R., Dijkstra G.et al. (2019) Short chain fatty acids (SCFAs)-mediated gut epithelial and immune regulation and its relevance for inflammatory bowel diseases. Front. Immunol. 10, 277 10.3389/fimmu.2019.00277 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Kelly C.J., Zheng L., Campbell E.L., Saeedi B., Scholz C.C., Bayless A.J.et al. (2015) Crosstalk between microbiota-derived short-chain fatty acids and intestinal epithelial HIF augments tissue barrier function. Cell Host Microbe 17, 662–671 10.1016/j.chom.2015.03.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Nepelska M., de Wouters T., Jacouton E., Beguet-Crespel F., Lapaque N., Dore J.et al. (2017) Commensal gut bacteria modulate phosphorylation-dependent PPARgamma transcriptional activity in human intestinal epithelial cells. Sci. Rep. 7, 43199 10.1038/srep43199 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Ritzhaupt A., Wood I.S., Ellis A., Hosie K.B. and Shirazi-Beechey S.P. (1998) Identification and characterization of a monocarboxylate transporter (MCT1) in pig and human colon: its potential to transport L-lactate as well as butyrate. J. Physiol. 513, 719–732 10.1111/j.1469-7793.1998.719ba.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Sivaprakasam S., Bhutia Y.D., Yang S. and Ganapathy V. (2017) Short-chain fatty acid transporters: role in colonic homeostasis. Compr. Physiol. 8, 299–314 10.1002/cphy.c170014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Alex S., Lange K., Amolo T., Grinstead J.S., Haakonsson A.K., Szalowska E.et al. (2013) Short-chain fatty acids stimulate angiopoietin-like 4 synthesis in human colon adenocarcinoma cells by activating peroxisome proliferator-activated receptor gamma. Mol. Cell. Biol. 33, 1303–1316 10.1128/MCB.00858-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Sivaprakasam S., Prasad P.D. and Singh N. (2016) Benefits of short-chain fatty acids and their receptors in inflammation and carcinogenesis. Pharmacol. Ther. 164, 144–151 10.1016/j.pharmthera.2016.04.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Macia L., Tan J., Vieira A.T., Leach K., Stanley D., Luong S.et al. (2015) Metabolite-sensing receptors GPR43 and GPR109A facilitate dietary fibre-induced gut homeostasis through regulation of the inflammasome. Nat. Commun. 6, 6734 10.1038/ncomms7734 [DOI] [PubMed] [Google Scholar]
- 125.Brown A.J., Goldsworthy S.M., Barnes A.A., Eilert M.M., Tcheang L., Daniels D.et al. (2003) The Orphan G protein-coupled receptors GPR41 and GPR43 are activated by propionate and other short chain carboxylic acids. J. Biol. Chem. 278, 11312–11319 10.1074/jbc.M211609200 [DOI] [PubMed] [Google Scholar]
- 126.Chen B., Chen H., Shu X., Yin Y., Li J., Qin J.et al. (2018) Presence of segmented filamentous bacteria in human children and its potential role in the modulation of human gut immunity. Front. Microbiol. 9, 1403 10.3389/fmicb.2018.01403 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Burger-van Paassen N., Vincent A., Puiman P.J., van der Sluis M., Bouma J., Boehm G.et al. (2009) The regulation of intestinal mucin MUC2 expression by short-chain fatty acids: implications for epithelial protection. Biochem. J. 420, 211–219 10.1042/BJ20082222 [DOI] [PubMed] [Google Scholar]
- 128.Zhao Y., Chen F., Wu W., Sun M., Bilotta A.J., Yao S.et al. (2018) GPR43 mediates microbiota metabolite SCFA regulation of antimicrobial peptide expression in intestinal epithelial cells via activation of mTOR and STAT3. Mucosal Immunol. 11, 752–762 10.1038/mi.2017.118 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Miyamoto J., Mizukure T., Park S.B., Kishino S., Kimura I., Hirano K.et al. (2015) A gut microbial metabolite of linoleic acid, 10-hydroxy-cis-12-octadecenoic acid, ameliorates intestinal epithelial barrier impairment partially via GPR40-MEK-ERK pathway. J. Biol. Chem. 290, 2902–2918 10.1074/jbc.M114.610733 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Yao B., He J., Yin X., Shi Y., Wan J. and Tian Z. (2019) The protective effect of lithocholic acid on the intestinal epithelial barrier is mediated by the vitamin D receptor via a SIRT1/Nrf2 and NF-kappaB dependent mechanism in Caco-2 cells. Toxicol. Lett. 316, 109–118 10.1016/j.toxlet.2019.08.024 [DOI] [PubMed] [Google Scholar]
- 131.Gensollen T., Iyer S.S., Kasper D.L. and Blumberg R.S. (2016) How colonization by microbiota in early life shapes the immune system. Science 352, 539–544 10.1126/science.aad9378 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Bauer H., Horowitz R.E., Levenson S.M. and Popper H. (1963) The response of the lymphatic tissue to the microbial flora. Studies on germfree mice. Am. J. Pathol. 42, 471–483 [PMC free article] [PubMed] [Google Scholar]
- 133.Umesaki Y., Setoyama H., Matsumoto S. and Okada Y. (1993) Expansion of alpha beta T-cell receptor-bearing intestinal intraepithelial lymphocytes after microbial colonization in germ-free mice and its independence from thymus. Immunology 79, 32–37 [PMC free article] [PubMed] [Google Scholar]
- 134.Hapfelmeier S., Lawson M.A., Slack E., Kirundi J.K., Stoel M., Heikenwalder M.et al. (2010) Reversible microbial colonization of germ-free mice reveals the dynamics of IgA immune responses. Science 328, 1705–1709 10.1126/science.1188454 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Gomez de Aguero M., Ganal-Vonarburg S.C., Fuhrer T., Rupp S., Uchimura Y., Li H.et al. (2016) The maternal microbiota drives early postnatal innate immune development. Science 351, 1296–1302 10.1126/science.aad2571 [DOI] [PubMed] [Google Scholar]
- 136.Ivanov I.I., Frutos Rde L., Manel N., Yoshinaga K., Rifkin D.B., Sartor R.B.et al. (2008) Specific microbiota direct the differentiation of IL-17-producing T-helper cells in the mucosa of the small intestine. Cell Host Microbe 4, 337–349 10.1016/j.chom.2008.09.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Ivanov I.I., Atarashi K., Manel N., Brodie E.L., Shima T., Karaoz U.et al. (2009) Induction of intestinal Th17 cells by segmented filamentous bacteria. Cell 139, 485–498 10.1016/j.cell.2009.09.033 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Tan T.G., Sefik E., Geva-Zatorsky N., Kua L., Naskar D., Teng F.et al. (2016) Identifying species of symbiont bacteria from the human gut that, alone, can induce intestinal Th17 cells in mice. Proc. Natl. Acad. Sci. U.S.A. 113, E8141–E8150 10.1073/pnas.1617460113 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Atarashi K., Tanoue T., Ando M., Kamada N., Nagano Y., Narushima S.et al. (2015) Th17 cell induction by adhesion of microbes to intestinal epithelial cells. Cell 163, 367–380 10.1016/j.cell.2015.08.058 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Mazmanian S.K., Liu C.H., Tzianabos A.O. and Kasper D.L. (2005) An immunomodulatory molecule of symbiotic bacteria directs maturation of the host immune system. Cell 122, 107–118 10.1016/j.cell.2005.05.007 [DOI] [PubMed] [Google Scholar]
- 141.An D., Oh S.F., Olszak T., Neves J.F., Avci F.Y., Erturk-Hasdemir D.et al. (2014) Sphingolipids from a symbiotic microbe regulate homeostasis of host intestinal natural killer T cells. Cell 156, 123–133 10.1016/j.cell.2013.11.042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Wesemann D.R., Portuguese A.J., Meyers R.M., Gallagher M.P., Cluff-Jones K., Magee J.M.et al. (2013) Microbial colonization influences early B-lineage development in the gut lamina propria. Nature 501, 112–115 10.1038/nature12496 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Levy M., Thaiss C.A., Zeevi D., Dohnalova L., Zilberman-Schapira G., Mahdi J.A.et al. (2015) Microbiota-modulated metabolites shape the intestinal microenvironment by regulating NLRP6 inflammasome signaling. Cell 163, 1428–1443 10.1016/j.cell.2015.10.048 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Seo S.U., Kamada N., Munoz-Planillo R., Kim Y.G., Kim D., Koizumi Y.et al. (2015) Distinct commensals induce interleukin-1beta via NLRP3 inflammasome in inflammatory monocytes to promote intestinal inflammation in response to injury. Immunity 42, 744–755 10.1016/j.immuni.2015.03.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Wolf A.J. and Underhill D.M. (2018) Peptidoglycan recognition by the innate immune system. Nat. Rev. Immunol. 18, 243–254 10.1038/nri.2017.136 [DOI] [PubMed] [Google Scholar]
- 146.Bain C.C., Bravo-Blas A., Scott C.L., Perdiguero E.G., Geissmann F., Henri S.et al. (2014) Constant replenishment from circulating monocytes maintains the macrophage pool in the intestine of adult mice. Nat. Immunol. 15, 929–937 10.1038/ni.2967 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Danne C., Ryzhakov G., Martinez-Lopez M., Ilott N.E., Franchini F., Cuskin F.et al. (2017) A large polysaccharide produced by Helicobacter hepaticus induces an anti-inflammatory gene signature in macrophages. Cell Host Microbe 22, 733.e5–745.e5 10.1016/j.chom.2017.11.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Schulthess J., Pandey S., Capitani M., Rue-Albrecht K.C., Arnold I., Franchini F.et al. (2019) The short chain fatty acid butyrate imprints an antimicrobial program in macrophages. Immunity 50, 432.e7–445.e7 10.1016/j.immuni.2018.12.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Wu K., Yuan Y., Yu H., Dai X., Wang S., Sun Z.et al. (2020) The gut microbial metabolite trimethylamine N-oxide aggravates GVHD by inducing M1 macrophage polarization in mice. Blood 136, 501–515 10.1182/blood.2019003990 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Yue C., Yang X., Li J., Chen X., Zhao X., Chen Y.et al. (2017) Trimethylamine N-oxide prime NLRP3 inflammasome via inhibiting ATG16L1-induced autophagy in colonic epithelial cells. Biochem. Biophys. Res. Commun. 490, 541–551 10.1016/j.bbrc.2017.06.075 [DOI] [PubMed] [Google Scholar]
- 151.Sonnenberg G.F. and Artis D. (2015) Innate lymphoid cells in the initiation, regulation and resolution of inflammation. Nat. Med. 21, 698–708 10.1038/nm.3892 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Ganal-Vonarburg S.C. and Duerr C.U. (2020) The interaction of intestinal microbiota and innate lymphoid cells in health and disease throughout life. Immunology 159, 39–51 10.1111/imm.13138 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Gury-BenAri M., Thaiss C.A., Serafini N., Winter D.R., Giladi A., Lara-Astiaso D.et al. (2016) The spectrum and regulatory landscape of intestinal innate lymphoid cells are shaped by the microbiome. Cell 166, 1231.e13–1246.e13 10.1016/j.cell.2016.07.043 [DOI] [PubMed] [Google Scholar]
- 154.Chun E., Lavoie S., Fonseca-Pereira D., Bae S., Michaud M., Hoveyda H.R.et al. (2019) Metabolite-sensing receptor Ffar2 regulates colonic group 3 innate lymphoid cells and gut immunity. Immunity 51, 871.e6–884.e6 10.1016/j.immuni.2019.09.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Keir M., Yi Y., Lu T. and Ghilardi N. (2020) The role of IL-22 in intestinal health and disease. J. Exp. Med. 217, e20192195 10.1084/jem.20192195 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Bostick J.W., Wang Y., Shen Z., Ge Y., Brown J., Chen Z.E.et al. (2019) Dichotomous regulation of group 3 innate lymphoid cells by nongastric Helicobacter species. Proc. Natl. Acad. Sci. U.S.A. 116, 24760–24769 10.1073/pnas.1908128116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Round J.L. and Mazmanian S.K. (2010) Inducible Foxp3+ regulatory T-cell development by a commensal bacterium of the intestinal microbiota. Proc. Natl. Acad. Sci. U.S.A. 107, 12204–12209 10.1073/pnas.0909122107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Arpaia N., Campbell C., Fan X., Dikiy S., van der Veeken J., Deroos P.et al. (2013) Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation. Nature 504, 451–455 10.1038/nature12726 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Song X., Sun X., Oh S.F., Wu M., Zhang Y., Zheng W.et al. (2020) Microbial bile acid metabolites modulate gut RORgamma(+) regulatory T cell homeostasis. Nature 577, 410–415 10.1038/s41586-019-1865-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Zelante T., Iannitti R.G., Cunha C., De Luca A., Giovannini G., Pieraccini G.et al. (2013) Tryptophan catabolites from microbiota engage aryl hydrocarbon receptor and balance mucosal reactivity via interleukin-22. Immunity 39, 372–385 10.1016/j.immuni.2013.08.003 [DOI] [PubMed] [Google Scholar]
- 161.Yoshii K., Hosomi K., Sawane K. and Kunisawa J. (2019) Metabolism of dietary and microbial vitamin B family in the regulation of host immunity. Front. Nutr. 6, 48 10.3389/fnut.2019.00048 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Kunisawa J., Sugiura Y., Wake T., Nagatake T., Suzuki H., Nagasawa R.et al. (2015) Mode of Bioenergetic metabolism during B cell differentiation in the intestine determines the distinct requirement for vitamin B1. Cell Rep. 13, 122–131 10.1016/j.celrep.2015.08.063 [DOI] [PubMed] [Google Scholar]
- 163.Schramm M., Wiegmann K., Schramm S., Gluschko A., Herb M., Utermohlen O.et al. (2014) Riboflavin (vitamin B2) deficiency impairs NADPH oxidase 2 (Nox2) priming and defense against Listeria monocytogenes. Eur. J. Immunol. 44, 728–741 10.1002/eji.201343940 [DOI] [PubMed] [Google Scholar]
- 164.Kjer-Nielsen L., Patel O., Corbett A.J., Le Nours J., Meehan B., Liu L.et al. (2012) MR1 presents microbial vitamin B metabolites to MAIT cells. Nature 491, 717–723 10.1038/nature11605 [DOI] [PubMed] [Google Scholar]
- 165.Eckle S.B., Birkinshaw R.W., Kostenko L., Corbett A.J., McWilliam H.E., Reantragoon R.et al. (2014) A molecular basis underpinning the T cell receptor heterogeneity of mucosal-associated invariant T cells. J. Exp. Med. 211, 1585–1600 10.1084/jem.20140484 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Singh N., Gurav A., Sivaprakasam S., Brady E., Padia R., Shi H.et al. (2014) Activation of Gpr109a, receptor for niacin and the commensal metabolite butyrate, suppresses colonic inflammation and carcinogenesis. Immunity 40, 128–139 10.1016/j.immuni.2013.12.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Agrawal S., Agrawal A. and Said H.M. (2016) Biotin deficiency enhances the inflammatory response of human dendritic cells. Am. J. Physiol. Cell Physiol. 311, C386–C391 10.1152/ajpcell.00141.2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Skupsky J., Sabui S., Hwang M., Nakasaki M., Cahalan M.D. and Said H.M. (2020) Biotin supplementation ameliorates murine colitis by preventing NF-kappaB activation. Cell Mol. Gastroenterol. Hepatol. 9, 557–567 10.1016/j.jcmgh.2019.11.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Tamura J., Kubota K., Murakami H., Sawamura M., Matsushima T., Tamura T.et al. (1999) Immunomodulation by vitamin B12: augmentation of CD8+ T lymphocytes and natural killer (NK) cell activity in vitamin B12-deficient patients by methyl-B12 treatment. Clin. Exp. Immunol. 116, 28–32 10.1046/j.1365-2249.1999.00870.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Lee Y.K. and Mazmanian S.K. (2010) Has the microbiota played a critical role in the evolution of the adaptive immune system? Science 330, 1768–1773 10.1126/science.1195568 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Harmsen H.J. and de Goffau M.C. (2016) The human gut microbiota. Microbiota Human Body 902, 95–108 10.1007/978-3-319-31248-4_7 [DOI] [PubMed] [Google Scholar]
- 172.DeGruttola A.K., Low D., Mizoguchi A. and Mizoguchi E. (2016) Current understanding of dysbiosis in disease in human and animal models. Inflamm. Bowel Dis. 22, 1137–1150 10.1097/MIB.0000000000000750 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Belkaid Y. and Hand T.W. (2014) Role of the microbiota in immunity and inflammation. Cell 157, 121–141 10.1016/j.cell.2014.03.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Das B. and Nair G.B. (2019) Homeostasis and dysbiosis of the gut microbiome in health and disease. J. Biosci. 44, 1–8 10.1007/s12038-019-9926-y [DOI] [PubMed] [Google Scholar]
- 175.Nguyen T.L.A., Vieira-Silva S., Liston A. and Raes J. (2015) How informative is the mouse for human gut microbiota research? Dis. Model Mech. 8, 1–16 10.1242/dmm.017400 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Mulder D.J., Noble A.J., Justinich C.J. and Duffin J.M. (2014) A tale of two diseases: the history of inflammatory bowel disease. J. Crohns Colitis 8, 341–348 10.1016/j.crohns.2013.09.009 [DOI] [PubMed] [Google Scholar]
- 177.Pascal V., Pozuelo M., Borruel N., Casellas F., Campos D., Santiago A.et al. (2017) A microbial signature for Crohn's disease. Gut 66, 813–822 10.1136/gutjnl-2016-313235 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Gevers D., Kugathasan S., Denson Lee A., Vázquez-Baeza Y., Van Treuren W., Ren B.et al. (2014) The treatment-naive microbiome in new-onset Crohn’s disease. Cell Host Microbe 15, 382–392 10.1016/j.chom.2014.02.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Martinez-Medina M., Aldeguer X., Lopez-Siles M., González-Huix F., López-Oliu C., Dahbi G.et al. (2009) Molecular diversity of Escherichia coli in the human gut: new ecological evidence supporting the role of adherent-invasive E. coli (AIEC) in Crohn’s disease. Inflamm. Bowel Dis. 15, 872–882 10.1002/ibd.20860 [DOI] [PubMed] [Google Scholar]
- 180.Qin J., Li R., Raes J., Arumugam M., Burgdorf K.S., Manichanh C.et al. (2010) A human gut microbial gene catalogue established by metagenomic sequencing. Nature 464, 59–65 10.1038/nature08821 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Willing B., Halfvarson J., Dicksved J., Rosenquist M., Järnerot G., Engstrand L.et al. (2009) Twin studies reveal specific imbalances in the mucosaassociated microbiota of patients with ileal Crohn’s disease. Inflamm. Bowel Dis. 15, 653–660 10.1002/ibd.20783 [DOI] [PubMed] [Google Scholar]
- 182.Sokol H., Pigneur B., Watterlot L., Lakhdari O., Bermúdez-Humarán L.G., Gratadoux J.-J.et al. (2008) Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of Crohn disease patients. Proc. Natl. Acad. Sci. U.S.A. 105, 16731–16736 10.1073/pnas.0804812105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Loubinoux J., Bronowicki J.-P., Pereira I.A.C., Mougenel J.-L. and Le Faou A.E. (2002) Sulfate-reducing bacteria in human feces and their association with inflammatory bowel diseases. FEMS Microbiol. Ecol. 40, 107–112 10.1111/j.1574-6941.2002.tb00942.x [DOI] [PubMed] [Google Scholar]
- 184.Jia W., Whitehead R.N., Griffiths L., Dawson C., Bai H., Waring R.H.et al. (2012) Diversity and distribution of sulphate-reducing bacteria in human faeces from healthy subjects and patients with inflammatory bowel disease. FEMS Immunol. Med. Microbiol. 65, 55–68 10.1111/j.1574-695X.2012.00935.x [DOI] [PubMed] [Google Scholar]
- 185.Darfeuille-Michaud A., Boudeau J., Bulois P., Neut C., Glasser A.L., Barnich N.et al. (2004) High prevalence of adherent-invasive Escherichia coli associated with ileal mucosa in Crohn’s disease. Gastroenterology 127, 412–421 10.1053/j.gastro.2004.04.061 [DOI] [PubMed] [Google Scholar]
- 186.Palmela C., Chevarin C., Xu Z., Torres J., Sevrin G., Hirten R.et al. (2018) Adherent-invasive Escherichia coli in inflammatory bowel disease. Gut 67, 574–587 10.1136/gutjnl-2017-314903 [DOI] [PubMed] [Google Scholar]
- 187.Nikolaus S., Schulte B., Al-Massad N., Thieme F., Schulte D.M., Bethge J.et al. (2017) Increased tryptophan metabolism is associated with activity of inflammatory bowel diseases. Gastroenterology 153, 1504.e2–1516.e2 10.1053/j.gastro.2017.08.028 [DOI] [PubMed] [Google Scholar]
- 188.Lamas B., Richard M.L., Leducq V., Pham H.-P., Michel M.-L., Da Costa G.et al. (2016) CARD9 impacts colitis by altering gut microbiota metabolism of tryptophan into aryl hydrocarbon receptor ligands. Nat. Med. 22, 598–605 10.1038/nm.4102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Singh P., Arora A., Strand T.A., Leffler D.A., Catassi C., Green P.H.et al. (2018) Global prevalence of celiac disease: systematic review and meta-analysis. Clin. Gastroenterol. Hepatol. 16, 823.e2–836.e2 10.1016/j.cgh.2017.06.037 [DOI] [PubMed] [Google Scholar]
- 190.De Re V., Magris R. and Cannizzaro R. (2017) New insights into the pathogenesis of Celiac disease. Front. Med. 4, 137, 10.3389/fmed.2017.00137 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Akobeng A.K., Singh P., Kumar M. and Al Khodor S. (2020) Role of the gut microbiota in the pathogenesis of coeliac disease and potential therapeutic implications. Eur. J. Nutr. 59, 3369–3390 10.1007/s00394-020-02324-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Collado M.C., Calabuig M. and Sanz Y. (2007) Differences between the fecal microbiota of coeliac infants and healthy controls. Curr. Issues Intest. Microbiol. 8, 9–14 [PubMed] [Google Scholar]
- 193.De Palma G., Nadal I., Medina M., Donat E., Ribes-Koninckx C., Calabuig M.et al. (2010) Intestinal dysbiosis and reduced immunoglobulin-coated bacteria associated with coeliac disease in children. BMC Microbiol. 10, 63 10.1186/1471-2180-10-63 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Sanz Y., Sánchez E., Marzotto M., Calabuig M., Torriani S. and Dellaglio F. (2007) Differences in faecal bacterial communities in coeliac and healthy children as detected by PCR and denaturing gradient gel electrophoresis. FEMS Immunol. Med. Microbiol. 51, 562–568 10.1111/j.1574-695X.2007.00337.x [DOI] [PubMed] [Google Scholar]
- 195.Sánchez E., Laparra J.M. and Sanz Y. (2012) Discerning the role of Bacteroides fragilis in celiac disease pathogenesis. Appl. Environ. Microbiol. 78, 6507–6515 10.1128/AEM.00563-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Shiba T., Aiba Y., Ishikawa H., Ushiyama A., Takagi A., Mine T.et al. (2003) The suppressive effect of bifidobacteria on Bacteroides vulgatus, a putative pathogenic microbe in inflammatory bowel disease. Microbiol. Immunol. 47, 371–378 10.1111/j.1348-0421.2003.tb03368.x [DOI] [PubMed] [Google Scholar]
- 197.Caminero A., Herrán A.R., Nistal E., Pérez-Andrés J., Vaquero L., Vivas S.et al. (2014) Diversity of the cultivable human gut microbiome involved in gluten metabolism: isolation of microorganisms with potential interest for coeliac disease. FEMS Microbiol. Ecol. 88, 309–319 10.1111/1574-6941.12295 [DOI] [PubMed] [Google Scholar]
- 198.Olivares M., Laparra M. and Sanz Y. (2011) Influence of Bifidobacterium longum CECT 7347 and gliadin peptides on intestinal epithelial cell proteome. J. Agric. Food Chem. 59, 7666–7671 10.1021/jf201212m [DOI] [PubMed] [Google Scholar]
- 199.Lindfors K., Blomqvist T., Juuti-Uusitalo K., Stenman S., Venäläinen J., Mäki M.et al. (2008) Live probiotic Bifidobacterium lactis bacteria inhibit the toxic effects induced by wheat gliadin in epithelial cell culture. Clin. Exp. Immunol. 152, 552–558 10.1111/j.1365-2249.2008.03635.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Medina M., De Palma G., Ribes-Koninckx C., Calabuig M. and Sanz Y. (2008) Bifidobacterium strains suppress in vitro the pro-inflammatory milieu triggered by the large intestinal microbiota of coeliac patients. J. Inflamm. 5, 19 10.1186/1476-9255-5-19 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Laparra J.M., Olivares M., Gallina O. and Sanz Y. (2012) Bifidobacterium longum CECT 7347 modulates immune responses in a gliadin-induced enteropathy animal model. PLoS ONE 7, e30744 10.1371/journal.pone.0030744 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Chen V.L. and Kasper D.L. (2014) Interactions between the intestinal microbiota and innate lymphoid cells. Gut Microbes 5, 129–140 10.4161/gmic.27289 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Moro K. and Koyasu S. (2015) Innate lymphoid cells, possible interaction with microbiota. Semin. Immunopathol. 37, 27–37 10.1007/s00281-014-0470-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Hrncir T., Stepankova R., Kozakova H., Hudcovic T. and Tlaskalova-Hogenova H. (2008) Gut microbiota and lipopolysaccharide content of the diet influence development of regulatory T cells: studies in germ-free mice. BMC Immunol. 9, 65 10.1186/1471-2172-9-65 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Silman A.J. and Pearson J.E. (2002) Epidemiology and genetics of rheumatoid arthritis. Arthritis Res. 4, S265–S272 10.1186/ar578 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Chen J., Wright K., Davis J.M., Jeraldo P., Marietta E.V., Murray J.et al. (2016) An expansion of rare lineage intestinal microbes characterizes rheumatoid arthritis. Genome Med. 8, 43. 10.1186/s13073-016-0299-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Liu X., Zou Q., Zeng B., Fang Y. and Wei H. (2013) Analysis of fecal Lactobacillus community structure in patients with early rheumatoid arthritis. Curr. Microbiol. 67, 170–176 10.1007/s00284-013-0338-1 [DOI] [PubMed] [Google Scholar]
- 208.Maeda Y., Kurakawa T., Umemoto E., Motooka D., Ito Y., Gotoh K.et al. (2016) Dysbiosis contributes to arthritis development via activation of autoreactive T cells in the intestine. Arthritis Rheumatol. 68, 2646–2661 10.1002/art.39783 [DOI] [PubMed] [Google Scholar]
- 209.Wu H.-J., Ivanov I.I., Darce J., Hattori K., Shima T., Umesaki Y.et al. (2010) Gut-residing segmented filamentous bacteria drive autoimmune arthritis via T helper 17 cells. Immunity 32, 815–827 10.1016/j.immuni.2010.06.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Wallin M.T., Culpepper W.J., Nichols E., Bhutta Z.A., Gebrehiwot T.T., Hay S.I.et al. (2019) Global, regional, and national burden of multiple sclerosis 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 18, 269–285 10.1016/S1474-4422(18)30443-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Chen J., Chia N., Kalari K.R., Yao J.Z., Novotna M., Paz Soldan M.M.et al. (2016) Multiple sclerosis patients have a distinct gut microbiota compared to healthy controls. Sci. Rep. 6, 28484 10.1038/srep28484 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Cantarel B.L., Waubant E., Chehoud C., Kuczynski J., DeSantis T.Z., Warrington J.et al. (2015) Gut microbiota in multiple sclerosis: possible influence of immunomodulators. J. Investig. Med. 63, 729–734 10.1097/JIM.0000000000000192 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Lee Y.K., Menezes J.S., Umesaki Y. and Mazmanian S.K. (2011) Proinflammatory T-cell responses to gut microbiota promote experimental autoimmune encephalomyelitis. Proc. Natl. Acad. Sci. U.S.A. 108, 4615–4622 10.1073/pnas.1000082107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Nouri M., Bredberg A., Weström B. and Lavasani S. (2014) Intestinal barrier dysfunction develops at the onset of experimental autoimmune encephalomyelitis, and can be induced by adoptive transfer of auto-reactive T cells. PLoS ONE 9, e106335 10.1371/journal.pone.0106335 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Secher T., Kassem S., Benamar M., Bernard I., Boury M., Barreau F.et al. (2017) Oral administration of the probiotic strain Escherichia coli Nissle 1917 reduces susceptibility to neuroinflammation and repairs experimental autoimmune encephalomyelitis-induced intestinal barrier dysfunction. Front. Immunol. 8, 1096 10.3389/fimmu.2017.01096 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Huang P.L. (2009) A comprehensive definition for metabolic syndrome. Dis. Model Mech. 2, 231–237 10.1242/dmm.001180 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Wang P.-X., Deng X.-R., Zhang C.-H. and Yuan H.-J. (2020) Gut microbiota and metabolic syndrome. Chin. Med. J. (Engl.) 133, 808–816 10.1097/CM9.0000000000000696 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Ridaura V.K., Faith J.J., Rey F.E., Cheng J., Duncan A.E., Kau A.L.et al. (2013) Gut microbiota from twins discordant for obesity modulate metabolism in mice. Science 341, 1241214 10.1126/science.1241214 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Bäckhed F., Manchester J.K., Semenkovich C.F. and Gordon J.I. (2007) Mechanisms underlying the resistance to diet-induced obesity in germ-free mice. Proc. Natl. Acad. Sci. U.S.A. 104, 979–984 10.1073/pnas.0605374104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Armougom F., Henry M., Vialettes B., Raccah D. and Raoult D. (2009) Monitoring bacterial community of human gut microbiota reveals an increase in Lactobacillus in obese patients and Methanogens in anorexic patients. PLoS ONE 4, e7125 10.1371/journal.pone.0007125 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Koliada A., Syzenko G., Moseiko V., Budovska L., Puchkov K., Perederiy V.et al. (2017) Association between body mass index and Firmicutes/Bacteroidetes ratio in an adult Ukrainian population. BMC Microbiol. 17, 120. 10.1186/s12866-017-1027-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Kong L.C., Tap J., Aron-Wisnewsky J., Pelloux V., Basdevant A., Bouillot J.L.et al. (2013) Gut microbiota after gastric bypass in human obesity: increased richness and associations of bacterial genera with adipose tissue genes. Am. J. Clin. Nutr. 98, 16–24 10.3945/ajcn.113.058743 [DOI] [PubMed] [Google Scholar]
- 223.Li J.V., Ashrafian H., Bueter M., Kinross J., Sands C., le Roux C.W.et al. (2011) Metabolic surgery profoundly influences gut microbial-host metabolic cross-talk. Gut 60, 1214–1223 10.1136/gut.2010.234708 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Qin J., Li Y., Cai Z., Li S., Zhu J., Zhang F.et al. (2012) A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature 490, 55–60 10.1038/nature11450 [DOI] [PubMed] [Google Scholar]
- 225.Samuel B.S., Shaito A., Motoike T., Rey F.E., Backhed F., Manchester J.K.et al. (2008) Effects of the gut microbiota on host adiposity are modulated by the short-chain fatty-acid binding G protein-coupled receptor, Gpr41. Proc. Natl. Acad. Sci. U.S.A. 105, 16767–16772 10.1073/pnas.0808567105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Lin H.V., Frassetto A., Kowalik E.J. Jr, Nawrocki A.R., Lu M.M., Kosinski J.R.et al. (2012) Butyrate and propionate protect against diet-induced obesity and regulate gut hormones via free fatty acid receptor 3-independent mechanisms. PLoS ONE 7, e35240 10.1371/journal.pone.0035240 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Cani P.D., Possemiers S., Van de Wiele T., Guiot Y., Everard A., Rottier O.et al. (2009) Changes in gut microbiota control inflammation in obese mice through a mechanism involving GLP-2-driven improvement of gut permeability. Gut 58, 1091–1103 10.1136/gut.2008.165886 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Chakaroun R.M., Massier L. and Kovacs P. (2020) Gut microbiome, intestinal permeability, and tissue bacteria in metabolic disease: perpetrators or bystanders? Nutrients 12, 1082, 10.3390/nu12041082 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Raso G.M., Simeoli R., Iacono A., Santoro A., Amero P., Paciello O.et al. (2014) Effects of a Lactobacillus paracasei B21060 based synbiotic on steatosis, insulin signaling and toll-like receptor expression in rats fed a high-fat diet. J. Nutr. Biochem. 25, 81–90 10.1016/j.jnutbio.2013.09.006 [DOI] [PubMed] [Google Scholar]
- 230.Martinez-Medina M., Denizot J., Dreux N., Robin F., Billard E., Bonnet R.et al. (2014) Western diet induces dysbiosis with increased E coli in CEABAC10 mice, alters host barrier function favouring AIEC colonisation. Gut 63, 116–124 10.1136/gutjnl-2012-304119 [DOI] [PubMed] [Google Scholar]
- 231.Wang X., Ota N., Manzanillo P., Kates L., Zavala-Solorio J., Eidenschenk C.et al. (2014) Interleukin-22 alleviates metabolic disorders and restores mucosal immunity in diabetes. Nature 514, 237–241 10.1038/nature13564 [DOI] [PubMed] [Google Scholar]
- 232.Burcelin R. (2016) Gut microbiota and immune crosstalk in metabolic disease. Mol. Metab. 5, 771–781 10.1016/j.molmet.2016.05.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Su G.L., Ko C.W., Bercik P., Falck-Ytter Y., Sultan S., Weizman A.V.et al. (2020) AGA Clinical Practice Guidelines on the role of probiotics in the management of gastrointestinal disorders. Gastroenterology 159, 697–705 10.1053/j.gastro.2020.05.059 [DOI] [PubMed] [Google Scholar]
- 234.Mullish B.H., Quraishi M.N., Segal J.P., McCune V.L., Baxter M., Marsden G.L.et al. (2018) The use of faecal microbiota transplant as treatment for recurrent or refractory Clostridium difficile infection and other potential indications: joint British Society of Gastroenterology (BSG) and Healthcare Infection Society (HIS) guidelines. Gut 67, 1920–1941 10.1136/gutjnl-2018-316818 [DOI] [PubMed] [Google Scholar]
- 235.Sanders M.E., Merenstein D.J., Reid G., Gibson G.R. and Rastall R.A. (2019) Probiotics and prebiotics in intestinal health and disease: from biology to the clinic. Nat. Rev. Gastroenterol. Hepatol. 16, 605–616 10.1038/s41575-019-0173-3 [DOI] [PubMed] [Google Scholar]
- 236.Segal J.P., Mullish B.H., Quraishi M.N., Iqbal T., Marchesi J.R. and Sokol H. (2020) Mechanisms underpinning the efficacy of faecal microbiota transplantation in treating gastrointestinal disease. Ther. Adv. Gastroenterol. 13, 1756284820946904 10.1177/1756284820946904 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Yan F. and Polk D.B. (2020) Probiotics and probiotic-derived functional factors-mechanistic insights into applications for intestinal homeostasis. Front. Immunol. 11, 1428. 10.3389/fimmu.2020.01428 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Mujagic Z., de Vos P., Boekschoten M.V., Govers C., Pieters H.H., de Wit N.J.et al. (2017) The effects of Lactobacillus plantarum on small intestinal barrier function and mucosal gene transcription; a randomized double-blind placebo controlled trial. Sci. Rep. 7, 40128 10.1038/srep40128 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Schiavi E., Gleinser M., Molloy E., Groeger D., Frei R., Ferstl R.et al. (2016) The surface-associated exopolysaccharide of Bifidobacterium longum 35624 plays an essential role in dampening host proinflammatory responses and repressing local TH17 responses. Appl. Environ. Microbiol. 82, 7185–7196 10.1128/AEM.02238-16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Alessandri G., Ossiprandi M.C., MacSharry J., van Sinderen D. and Ventura M. (2019) Bifidobacterial dialogue with its human host and consequent modulation of the immune system. Front. Immunol. 10, 2348, 10.3389/fimmu.2019.02348 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Chelakkot C., Choi Y., Kim D.K., Park H.T., Ghim J., Kwon Y.et al. (2018) Akkermansia muciniphila-derived extracellular vesicles influence gut permeability through the regulation of tight junctions. Exp. Mol. Med. 50, e450 10.1038/emm.2017.282 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 242.Bäuerl C., Coll-Marqués J.M., Tarazona-González C. and Pérez-Martínez G. (2020) Lactobacillus casei extracellular vesicles stimulate EGFR pathway likely due to the presence of proteins P40 and P75 bound to their surface. Sci. Rep. 10, 19237 10.1038/s41598-020-75930-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Hols P., Ledesma-García L., Gabant P. and Mignolet J. (2019) Mobilization of microbiota commensals and their bacteriocins for therapeutics. Trends Microbiol. 27, 690–702 10.1016/j.tim.2019.03.007 [DOI] [PubMed] [Google Scholar]
- 244.Rivière A., Selak M., Lantin D., Leroy F. and De Vuyst L. (2016) Bifidobacteria and butyrate-producing colon bacteria: importance and strategies for their stimulation in the human gut. Front. Microbiol. 7, 979 10.3389/fmicb.2016.00979 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Parker E.A., Roy T., D’Adamo C.R. and Wieland L.S. (2018) Probiotics and gastrointestinal conditions: an overview of evidence from the Cochrane Collaboration. Nutrition 45, 125.e11–134.e11 10.1016/j.nut.2017.06.024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Limketkai B.N., Akobeng A.K., Gordon M. and Adepoju A.A. (2020) Probiotics for induction of remission in Crohn's disease. Cochrane Database Syst. Rev. 7, CD006634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247.Kaur L., Gordon M., Baines P.A., Iheozor-Ejiofor Z., Sinopoulou V. and Akobeng A.K. (2020) Probiotics for induction of remission in ulcerative colitis. Cochrane Database Syst. Rev. 3, CD005573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 248.Iheozor-Ejiofor Z., Kaur L., Gordon M., Baines P.A., Sinopoulou V. and Akobeng A.K. (2020) Probiotics for maintenance of remission in ulcerative colitis. Cochrane Database Syst. Rev. 3, CD007443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 249.Ford A.C., Harris L.A., Lacy B.E., Quigley E.M.M. and Moayyedi P. (2018) Systematic review with meta-analysis: the efficacy of prebiotics, probiotics, synbiotics and antibiotics in irritable bowel syndrome. Aliment. Pharmacol. Ther. 48, 1044–1060 10.1111/apt.15001 [DOI] [PubMed] [Google Scholar]
- 250.Goldenberg J.Z., Yap C., Lytvyn L., Lo C.K., Beardsley J., Mertz D.et al. (2017) Probiotics for the prevention of Clostridium difficile-associated diarrhea in adults and children. Cochrane Database Syst. Rev. 12, CD006095 10.1002/14651858.CD006095.pub4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Sniffen J.C., McFarland L.V., Evans C.T. and Goldstein E.J.C. (2018) Choosing an appropriate probiotic product for your patient: an evidence-based practical guide. PLoS ONE 13, e0209205 10.1371/journal.pone.0209205 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 252.Ansari J.M., Colasacco C., Emmanouil E., Kohlhepp S. and Harriott O. (2019) Strain-level diversity of commercial probiotic isolates of Bacillus, Lactobacillus, and Saccharomyces species illustrated by molecular identification and phenotypic profiling. PLoS ONE 14, e0213841 10.1371/journal.pone.0213841 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Magnúsdóttir S., Heinken A., Kutt L., Ravcheev D.A., Bauer E., Noronha A.et al. (2017) Generation of genome-scale metabolic reconstructions for 773 members of the human gut microbiota. Nat. Biotechnol. 35, 81–89 10.1038/nbt.3703 [DOI] [PubMed] [Google Scholar]
- 254.Poyet M., Groussin M., Gibbons S.M., Avila-Pacheco J., Jiang X., Kearney S.M.et al. (2019) A library of human gut bacterial isolates paired with longitudinal multiomics data enables mechanistic microbiome research. Nat. Med. 25, 1442–1452 10.1038/s41591-019-0559-3 [DOI] [PubMed] [Google Scholar]
- 255.Devika N.T. and Raman K. (2019) Deciphering the metabolic capabilities of Bifidobacteria using genome-scale metabolic models. Sci. Rep. 9, 18222 10.1038/s41598-019-54696-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 256.Yadav M. and Shukla P. (2019) Recent systems biology approaches for probiotics use in health aspects: a review. 3 Biotech 9, 448 10.1007/s13205-019-1980-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 257.Tabashsum Z., Peng M., Salaheen S., Comis C. and Debabrata B. (2017) Competitive elimination and virulence property alteration of Campylobacter jejuni by genetically engineered Lactobacillus casei. Food Control 85, 283–291, 10.1016/j.foodcont.2017.10.010, 29332999 [DOI] [Google Scholar]
- 258.Schellenberger J.D., Que R., Fleming R.M.T., Thiele I., Orth J.D., Feist A.M.et al. (2011) Quantitative prediction of cellular metabolism with constraint-based models: the COBRA Toolbox v2.0. Nat. Protoc. 6, 1290–1307 10.1038/nprot.2011.308 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259.Ottman N., Davids M., Suarez-Diez M., Boeren S., Schaap P.J., Martins Dos Santos V.A.P.et al. (2017) Genome-scale model and omics analysis of metabolic capacities of Akkermansia muciniphila reveal a preferential mucin-degrading lifestyle. Appl. Environ. Microbiol. 83, e01014–17, 10.1128/AEM.01014-17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 260.Caputo A., Dubourg G., Croce O., Gupta S., Robert C., Papazian L.et al. (2015) Whole-genome assembly of Akkermansia muciniphila sequenced directly from human stool. Biol. Direct 10, 5 10.1186/s13062-015-0041-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 261.Zmora N., Zilberman-Schapira G., Suez J., Mor U., Dori-Bachash M., Bashiardes S.et al. (2018) Personalized gut mucosal colonization resistance to empiric probiotics is associated with unique host and microbiome features. Cell 174, 1388.e21–1405.e21 10.1016/j.cell.2018.08.041 [DOI] [PubMed] [Google Scholar]
- 262.Kristensen N.B., Bryrup T., Allin K.H., Nielsen T., Hansen T.H. and Pedersen O. (2016) Alterations in fecal microbiota composition by probiotic supplementation in healthy adults: a systematic review of randomized controlled trials. Genome Med. 8, 52 10.1186/s13073-016-0300-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 263.Hill C., Guarner F., Reid G., Gibson G.R., Merenstein D.J., Pot B.et al. (2014) The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 11, 506–514 10.1038/nrgastro.2014.66 [DOI] [PubMed] [Google Scholar]
- 264.Salminen S., Collado M.C., Endo A., Hill C., Lebeer S., Quigley E.M.M.et al. (2021) The International Scientific Association of Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of postbiotics. Nat. Rev. Gastroenterol. Hepatol. 10.1038/s41575-021-00440-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 265.Travers M.-A., Sow C., Zirah S., Deregnaucourt C., Chaouch S., Queiroz R.M.L.et al. (2016) Deconjugated bile salts produced by extracellular bile-salt hydrolase-like activities from the probiotic Lactobacillus johnsonii La1 inhibit giardia duodenalis in vitro growth. Front. Microbiol. 7, 1453, 10.3389/fmicb.2016.01453 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 266.Warda A.K., de Almeida Bettio P.H., Hueston C.M., Di Benedetto G., Clooney A.G. and Hill C. (2020) Oral administration of heat-treated lactobacilli modifies the murine microbiome and reduces citrobacter induced colitis. Front. Microbiol. 11, 69, 10.3389/fmicb.2020.00069 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 267.Luceri C., Femia A.P., Fazi M., Di Martino C., Zolfanelli F., Dolara P.et al. (2016) Effect of butyrate enemas on gene expression profiles and endoscopic/histopathological scores of diverted colorectal mucosa: a randomized trial. Dig. Liver Dis. 48, 27–33 10.1016/j.dld.2015.09.005 [DOI] [PubMed] [Google Scholar]
- 268.Andresen V., Gschossmann J. and Layer P. (2020) Heat-inactivated Bifidobacterium bifidum MIMBb75 (SYN-HI-001) in the treatment of irritable bowel syndrome: a multicentre, randomised, double-blind, placebo-controlled clinical trial. Lancet Gastroenterol. Hepatol. 5, 658–666 10.1016/S2468-1253(20)30056-X [DOI] [PubMed] [Google Scholar]
- 269.Cardinale F., Lombardi E., Rossi O., Bagnasco D., Bellocchi A. and Menzella F. (2020) Epithelial dysfunction, respiratory infections and asthma: the importance of immunomodulation. A focus on OM-85. Expert Rev. Respir. Med. 14, 1019–1026 10.1080/17476348.2020.1793673 [DOI] [PubMed] [Google Scholar]
- 270.Yin J., Xu B., Zeng X. and Shen K. (2018) Broncho-Vaxom in pediatric recurrent respiratory tract infections: a systematic review and meta-analysis. Int. Immunopharmacol. 54, 198–209 10.1016/j.intimp.2017.10.032 [DOI] [PubMed] [Google Scholar]
- 271.(EMA) EMA (2019) Bacterial lysates-containing medicinal products for respiratory conditions. Assessment Rep. https://ec.europa.eu/health/sites/default/files/files/eudralex/vol-1/dir_2001_83_consol_2012/dir_2001_83_cons_2012_en.pdf [Google Scholar]
- 272.Surawicz C.M., Brandt L.J., Binion D.G., Ananthakrishnan A.N., Curry S.R., Gilligan P.H.et al. (2013) Guidelines for diagnosis, treatment, and prevention of Clostridium difficile infections. Am. J. Gastroenterol. 108, 478–498, quiz 499 10.1038/ajg.2013.4 [DOI] [PubMed] [Google Scholar]
- 273.Cammarota G., Ianiro G., Tilg H., Rajilić-Stojanović M., Kump P., Satokari R.et al. (2017) European consensus conference on faecal microbiota transplantation in clinical practice. Gut 66, 569–580 10.1136/gutjnl-2016-313017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 274.Quraishi M.N., Widlak M., Bhala N., Moore D., Price M., Sharma N.et al. (2017) Systematic review with meta-analysis: the efficacy of faecal microbiota transplantation for the treatment of recurrent and refractory Clostridium difficile infection. Aliment. Pharmacol. Ther. 46, 479–493 10.1111/apt.14201 [DOI] [PubMed] [Google Scholar]
- 275.Fischer M., Sipe B., Cheng Y.W., Phelps E., Rogers N., Sagi S.et al. (2017) Fecal microbiota transplant in severe and severe-complicated Clostridium difficile: a promising treatment approach. Gut Microbes 8, 289–302 10.1080/19490976.2016.1273998 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 276.Dang X., Xu M., Liu D., Zhou D. and Yang W. (2020) Assessing the efficacy and safety of fecal microbiota transplantation and probiotic VSL#3 for active ulcerative colitis: a systematic review and meta-analysis. PLoS ONE 15, e0228846 10.1371/journal.pone.0228846 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 277.Zhao H.L., Chen S.Z., Xu H.M., Zhou Y.L., He J., Huang H.L.et al. (2020) Efficacy and safety of fecal microbiota transplantation for treating patients with ulcerative colitis: a systematic review and meta-analysis. J. Digest. Dis. 21, 534–548 10.1111/1751-2980.12933 [DOI] [PubMed] [Google Scholar]
- 278.Quraishi M.N., Yalchin M., Blackwell C., Segal J., Sharma N., Hawkey P.et al. (2019) STOP-Colitis pilot trial protocol: a prospective, open-label, randomised pilot study to assess two possible routes of faecal microbiota transplant delivery in patients with ulcerative colitis. BMJ Open 9, e030659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 279.Harbord M., Eliakim R., Bettenworth D., Karmiris K., Katsanos K., Kopylov U.et al. (2017) Third European evidence-based consensus on diagnosis and management of ulcerative colitis. Part 2: current management. J. Crohns Colitis 11, 769–784 10.1093/ecco-jcc/jjx009 [DOI] [PubMed] [Google Scholar]
- 280.Caldeira L.F., Borba H.H., Tonin F.S., Wiens A., Fernandez-Llimos F. and Pontarolo R. (2020) Fecal microbiota transplantation in inflammatory bowel disease patients: a systematic review and meta-analysis. PLoS ONE 15, e0238910 10.1371/journal.pone.0238910 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 281.Gomollón F., Dignass A., Annese V., Tilg H., Van Assche G., Lindsay J.O.et al. (2017) 3rd European Evidence-based Consensus on the Diagnosis and Management of Crohn’s Disease 2016: Part 1: diagnosis and medical management. J. Crohns Colitis 11, 3–25 10.1093/ecco-jcc/jjw168 [DOI] [PubMed] [Google Scholar]
- 282.Ianiro G., Eusebi L.H., Black C.J., Gasbarrini A., Cammarota G. and Ford A.C. (2019) Systematic review with meta-analysis: efficacy of faecal microbiota transplantation for the treatment of irritable bowel syndrome. Aliment. Pharmacol. Ther. 50, 240–248 10.1111/apt.15330 [DOI] [PubMed] [Google Scholar]
- 283.Allegretti J.R., Kassam Z., Mullish B.H., Chiang A., Carrellas M., Hurtado J.et al. (2020) Effects of fecal microbiota transplantation with oral capsules in obese patients. Clin. Gastroenterol. Hepatol. 18, 855.e2–863.e2 10.1016/j.cgh.2019.07.006 [DOI] [PubMed] [Google Scholar]
- 284.Kootte R.S., Levin E., Salojärvi J., Smits L.P., Hartstra A.V., Udayappan S.D.et al. (2017) Improvement of insulin sensitivity after lean donor feces in metabolic syndrome is driven by baseline intestinal microbiota composition. Cell Metab. 26, 611.e6–619.e6 10.1016/j.cmet.2017.09.008 [DOI] [PubMed] [Google Scholar]
- 285.Bajaj J.S., Salzman N.H., Acharya C., Sterling R.K., White M.B., Gavis E.A.et al. (2019) Fecal microbial transplant capsules are safe in hepatic encephalopathy: a phase 1, randomized, placebo-controlled trial. Hepatology 70, 1690–1703 10.1002/hep.30690 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 286.de Groot P., Nikolic T., Pellegrini S., Sordi V., Imangaliyev S., Rampanelli E.et al. (2021) Faecal microbiota transplantation halts progression of human new-onset type 1 diabetes in a randomised controlled trial. Gut 70, 92–105 10.1136/gutjnl-2020-322630 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 287.Vendrik K.E.W., Ooijevaar R.E., de Jong P.R.C., Laman J.D., van Oosten B.W., van Hilten J.J.et al. (2020) Fecal microbiota transplantation in neurological disorders. Front. Cell Infect. Microbiol. 10, 98 10.3389/fcimb.2020.00098 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 288.Sivan A., Corrales L., Hubert N., Williams J.B., Aquino-Michaels K., Earley Z.M.et al. (2015) Commensal Bifidobacterium promotes antitumor immunity and facilitates anti-PD-L1 efficacy. Science 350, 1084–1089 10.1126/science.aac4255 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 289.Vétizou M., Pitt J.M., Daillère R., Lepage P., Waldschmitt N., Flament C.et al. (2015) Anticancer immunotherapy by CTLA-4 blockade relies on the gut microbiota. Science 350, 1079–1084 10.1126/science.aad1329 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 290.Davar D., Dzutsev A.K., McCulloch J.A., Rodrigues R.R., Chauvin J.M., Morrison R.M.et al. (2021) Fecal microbiota transplant overcomes resistance to anti-PD-1 therapy in melanoma patients. Science 371, 595–602 10.1126/science.abf3363 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 291.Barr J.J., Auro R., Furlan M., Whiteson K.L., Erb M.L., Pogliano J.et al. (2013) Bacteriophage adhering to mucus provide a non–host-derived immunity. Proc. Natl. Acad. Sci. U.S.A. 110, 10771–10776 10.1073/pnas.1305923110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 292.Zuo T., Wong S.H., Lam K., Lui R., Cheung K., Tang W.et al. (2018) Bacteriophage transfer during faecal microbiota transplantation in Clostridium difficile infection is associated with treatment outcome. Gut 67, 634–643 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 293.Zuo T., Wong S.H., Cheung C.P., Lam K., Lui R., Cheung K.et al. (2018) Gut fungal dysbiosis correlates with reduced efficacy of fecal microbiota transplantation in Clostridium difficile infection. Nat. Commun. 9, 3663 10.1038/s41467-018-06103-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 294.Ott S.J., Waetzig G.H., Rehman A., Moltzau-Anderson J., Bharti R., Grasis J.A.et al. (2017) Efficacy of sterile fecal filtrate transfer for treating patients with Clostridium difficile infection. Gastroenterology 152, 799.e7–811.e7 10.1053/j.gastro.2016.11.010 [DOI] [PubMed] [Google Scholar]
- 295.Mabwi H.A., Kim E., Song D.-G., Yoon H.S., Pan C.-H., Komba E.V.G.et al. (2021) Synthetic gut microbiome: advances and challenges. Comput. Struct. Biotechnol. J. 19, 363–371 10.1016/j.csbj.2020.12.029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 296.Petrof E.O., Gloor G.B., Vanner S.J., Weese S.J., Carter D., Daigneault M.C.et al. (2013) Stool substitute transplant therapy for the eradication of Clostridium difficile infection: ‘RePOOPulating’ the gut. Microbiome 1, 3 10.1186/2049-2618-1-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 297.Rode A.A., Chehri M., Krogsgaard L.R., Heno K.K., Svendsen A.T., Ribberholt I.et al., (2021) Randomised clinical trial: a 12-strain bacterial mixture versus faecal microbiota transplantation versus vancomycin for recurrent Clostridioides difficile infections. Aliment. Pharmacol. Ther. 53, 999–1009, 10.1111/apt.16309 [DOI] [PubMed] [Google Scholar]
- 298.Staley C., Hamilton M.J., Vaughn B.P., Graiziger C.T., Newman K.M., Kabage A.J.et al. (2017) Successful resolution of recurrent Clostridium difficile infection using freeze-dried, encapsulated fecal microbiota; pragmatic cohort study. Am. J. Gastroenterol. 112, 940–947 10.1038/ajg.2017.6 [DOI] [PMC free article] [PubMed] [Google Scholar]