Abstract
We used stable isotope–labeled glucose and palmitate tracer infusions, a hyperinsulinemic-euglycemic clamp, positron emission tomography of muscles and adipose tissue after [18F]fluorodeoxyglucose and [15O]water injections, and subcutaneous adipose tissue (SAT) biopsy to test the hypotheses that 1) increased glucose uptake in SAT is responsible for high insulin-stimulated whole-body glucose uptake in people with obesity who are insulin sensitive and 2) putative SAT factors thought to cause insulin resistance are present in people with obesity who are insulin resistant but not in those who are insulin sensitive. We found that high insulin-stimulated whole-body glucose uptake in insulin-sensitive participants with obesity was not due to channeling of glucose into SAT but, rather, was due to high insulin-stimulated muscle glucose uptake. Furthermore, insulin-stimulated muscle glucose uptake was not different between insulin-sensitive obese and lean participants even though adipocytes were larger, SAT perfusion and oxygenation were lower, and markers of SAT inflammation, fatty acid appearance in plasma in relation to fat-free mass, and plasma fatty acid concentration were higher in the insulin-sensitive obese than in lean participants. In addition, we observed only marginal or no differences in adipocyte size, SAT perfusion and oxygenation, and markers of SAT inflammation between insulin-resistant and insulin-sensitive obese participants. Plasma fatty acid concentration was also not different between insulin-sensitive and insulin-resistant obese participants, even though SAT was resistant to the inhibitory effect of insulin on lipolysis in the insulin-resistant obese group. These data suggest that several putative SAT factors commonly implicated in causing insulin resistance are normal consequences of SAT expansion unrelated to insulin resistance.
Introduction
There is considerable heterogeneity in insulin-stimulated whole-body glucose uptake among people with obesity (1). It is commonly believed that differences in adipocyte size and adipose tissue function are responsible for the differences in insulin sensitivity (1–3). Adipocyte hypertrophy, but not adipocyte hyperplasia, presumably initiates a cascade of events that includes impaired adipose tissue perfusion, oxygenation, inflammation, and fibrosis; this in turn causes adipose tissue insulin resistance, leading to increased fatty acid release from adipose tissue, increased plasma fatty acid concentration, and fatty acidmediated insulin resistance in skeletal muscles (i.e., impaired insulin-stimulated muscle glucose uptake) and the liver (impaired insulin-mediated suppression of glucose production) (1–3). However, the importance of fatty acid release from adipose tissue in causing obesity-associated muscle and liver insulin resistance has been challenged because plasma fatty acid concentration is often not different in people who are insulin resistant and those who are insulin sensitive (4,5). Based on results from studies in animals, it has also been proposed that increased insulin-stimulated adipose tissue glucose uptake per se might be responsible for high whole-body glucose uptake in some people with obesity (6). In mice, overexpressing GLUT4 in adipose tissue increases fat mass by causing adipocyte proliferation (not hypertrophy) and improves whole-body glucose uptake, whereas downregulating GLUT4 in adipose tissue decreases whole-body insulin sensitivity (6,7).
The goal of our study was to test the hypothesis that increased adipose tissue glucose uptake is responsible for high insulin-stimulated whole-body glucose uptake in people with obesity who are insulin sensitive. In addition, we hypothesized that putative adipose tissue factors that are thought to cause insulin resistance are present in insulin-resistant, but not in insulin-sensitive, people with obesity. We assessed whole-body, muscle, and adipose tissue glucose uptake; adipose tissue perfusion and oxygenation; and fatty acid appearance rate in plasma by using stable isotope–labeled glucose and palmitate tracer infusions in conjunction with a hyperinsulinemic-euglycemic clamp procedure and positron emission tomography of adipose tissue and skeletal muscles after [18F]fluorodeoxyglucose and [15O]water injections in healthy lean people and people with obesity who were grouped according to their insulin-stimulated whole-body glucose uptake rate. Subcutaneous abdominal adipose tissue biopsies were collected to assess adipocyte size and the expression of genes associated with inflammation and extracellular matrix accumulation. Untargeted gene expression analysis was used to identify potential additional genes associated with insulin resistance.
Research Design and Methods
Study Participants
The data reported here were obtained from 15 healthy lean people (10 women, 5 men) and 37 people with obesity (28 women, 9 men) who participated in two different studies (ClinicalTrials.gov, clinical trial reg. nos. NCT02994459 and NCT03408613) with the same experimental protocol. The studies were approved by the Human Research Protection Office at Washington University in St. Louis, St. Louis, MO. Written informed consent was obtained from all participants before they enrolled in the study. All participants completed detailed body composition analysis and a comprehensive screening evaluation (described in Supplementary Material) for exclusion of those with medical or other conditions that could affect the outcomes. All participants were sedentary (i.e., they engaged in <90 min of exercise per week) and were instructed to abstain from exercise or other vigorous physical activities for 3 days before metabolic testing.
Metabolic Testing
Each participant completed a basal metabolic study for determination of basal glucose and palmitate kinetics with use of intravenous [6,6-2H2]glucose and [U-13C]palmitate infusions (8,9). They also completed a two-stage hyperinsulinemic-euglycemic clamp procedure in conjunction with [6,6-2H2]glucose and [U-13C]palmitate infusions and positron emission tomography scanning after injections of [18F]fluorodeoxyglucose and [15O]water for determination of insulin-stimulated whole-body, subcutaneous adipose tissue (SAT), and muscle glucose uptake; SAT perfusion; and insulin action on adipose tissue lipolysis. Adipose tissue oxygenation was assessed with an oxygen-sensitive fiber-optic probe (OxyLite; Oxford Optronix) (10,11). Biopsies from abdominal SAT were obtained to evaluate adipocyte size and gene expression (12). A detailed description of all procedures is provided in Supplementary Material.
Participant Grouping
Identifying people with obesity who are insulin sensitive and have high insulin-stimulated whole-body glucose uptake is difficult because there are no standardized criteria for insulin resistance. Furthermore, adjusting whole-body glucose uptake rates for differences in body size between lean people and people with obesity—by expressing the data in relation to total body mass or various components of body mass, such as fat-free mass—can affect the interpretation of the data (13). In addition, these adjustments include a priori assumptions regarding the major sites (e.g., fat-free mass) of glucose uptake. Therefore, we initially used total insulin-stimulated whole-body glucose uptake rate (micromoles per minute) to stratify participants with obesity into quartiles (Q), representing the most insulin-sensitive (Q1) and progressively more insulin-resistant (Q2–Q4) groups. We also used glucose uptake in relation to body surface area (to take into account differences in stature among people without making assumptions about the sites of glucose uptake) to identify the most insulin-sensitive and insulin-resistant participants (Supplementary Fig. 1). The two methods captured the same participants in Q1. Therefore, our selection of the most insulin-sensitive participants with obesity was not affected by body size. However, there was some discrepancy in participant categorization into Q2–Q4 with use of glucose uptake in micromoles per minute and glucose uptake in micromoles per minute per meter squared of body surface area. Nevertheless, the same participants were captured in Q3 and Q4 combined with both approaches. Therefore, we defined the most insulin-resistant participants as those in Q3/4. Our primary comparisons focused on the participants with obesity who were most insulin sensitive (Q1) versus the lean participants and versus participants with obesity who were most insulin-resistant (Q3/4). The sample size of each of these groups matched or exceeded the minimum number of participants per group needed to detect the expected differences in outcomes between groups.
Statistical Analysis
Differences in outcomes among groups were evaluated with ANOVA followed by multiple comparisons–adjusted post hoc analysis. Skewed data sets were transformed to achieve normal distribution before analysis. Regression analysis was used to evaluate the relationship among variables. A P value ≤0.05 was considered statistically significant. For untargeted gene expression analysis, we focused on genes that were at least twofold different between the insulin-resistant or insulin-sensitive obese and both the insulin-sensitive obese and the lean groups. A q value ≤0.05 was considered significant. Values are reported as mean ± SD or median and interquartile range (quartiles) unless otherwise noted.
Sample Size Estimation
Based on published differences in insulin-stimulated SAT glucose uptake between lean and obese people (14), we estimated that nine participants per group would be needed to detect at least a 7.8 µmol/kg SAT/min difference among groups with a power of 0.8 and an α-value of 0.05.
Data and Resource Availability
The data generated from this study are available from the corresponding author upon reasonable request. No resources were generated.
Results
Participants’ Body Composition
Percent body fat was less in the lean than in all obese groups and was not different among the obese groups (Table 1). Android/gynoid fat distribution was also not different among the obese groups, but intra-abdominal adipose tissue volume was greater in all obese groups compared with the lean group and increased progressively from the insulin-sensitive to the most insulin-resistant obese groups (Table 1).
Table 1.
Participants’ sex, age, body composition, plasma lipids, and insulin sensitivity indices
| Obese | ||||
|---|---|---|---|---|
| Lean | Insulin sensitive | Quartile 2 | Insulin resistant | |
| N (n female/n male) | 15 (10/5) | 11 (7/4) | 9 (8/1) | 17 (13/4) |
| Age (years) | 39 ± 12 | 46 ± 10 | 48 ± 11 | 41 ± 11 |
| BMI (kg/m2) | 22.7 ± 1.8 | 36.6 ± 5.4a | 36.8 ± 3.8a | 38.6 ± 5.1a |
| Body mass (kg) | 64 ± 6 | 102 ± 18a | 99 ± 13a | 108 ± 18a |
| FFM (kg) | 45 ± 7 | 57 ± 12a | 50 ± 9a | 59 ± 10a |
| Fat mass (kg) | 19 ± 5 | 46 ± 13a | 49 ± 9a | 50 ± 10a |
| Body fat (%) | 30 (28, 37) | 46 (34, 51)a | 51 (46, 53)a | 45 (43, 48)a |
| Android fat mass (% total body fat mass) | 6 ± 1 | 9 ± 2a | 9 ± 2a | 10 ± 2a |
| Gynoid fat mass (% total body fat mass) | 19 ± 2 | 16 ± 3a | 16 ± 2a | 16 ± 2a |
| Android–to–gynoid fat mass ratio | 0.34 ± 0.07 | 0.57 ± 0.14a | 0.54 ± 0.09a | 0.60 ± 0.13a |
| IAAT volume (cm3) | 975 (771, 1,396) | 2,732 (2,107, 4,011)a | 3,294 (2,743, 3,611)a | 3,697 (2,037, 4,235)a† |
| ASAT volume (cm3) | 2,458 ± 1,012 | 9,164 ± 2,706a | 11,018 ± 3,147a | 11,567 ± 2,608a |
| Insulin-stimulated glucose Rd | ||||
| µmol/min | 3,906 ± 282a | 4,215 ± 239a | 2,745 ± 111b | 1,752 ± 91c |
| (µmol/min)/(pmol insulin per liter plasma) | 6.6 ± 2.3a | 6.0 ± 1.0a | 4.3 ± 1.1b | 2.6 ± 0.5c |
| µmol/kg body wt/min | 65 (52, 74)a | 40 (37, 42)b | 27 (25, 30)c | 16 (13, 19)d |
| µmol/m2 BSA/min | 2,385 (1,741, 2,606)a | 1,938 (1,793, 2,051)a | 1,380 (1,178, 1,458)b | 815 (717, 936)c |
| µmol/kg FFM/min | 88 (73, 104)a | 78 (68, 85)a | 54 (49, 67)b | 31 (26, 36)c |
| Clamp stage 2 GIR in relation to plasma insulin | ||||
| (µmol/min)/(pmol/L) | 5.9 ± 2.0a | 5.9 ± 0.9a | 4.2 ± 1.1b | 2.3 ± 0.6c |
| (µmol/kg body wt/min)/(pmol/L) | 0.10 (0.00, 0.11)a | 0.06 (0.05, 0.06)b | 0.04 (0.04, 0.05)c | 0.02 (0.02, 0.03)d |
| (µmol/m2 BSA/min)/(pmol/L) | 3.2 (2.3, 4.0)a | 2.8 (2.5, 2.9)ab | 2.0 (1.7, 2.4)c | 1.0 (0.9, 1.3)d |
| (µmol/kg FFM/min)/(pmol/L) | 0.12 (0.10, 0.15)a | 0.10 (0.10, 0.12)ab | 0.08 (0.06, 0.11)b | 0.04 (0.03, 0.05)c |
| Hepatic insulin sensitivity index** | 15.5 (14.0, 24.5)a | 14.7 (10.6, 18.7)ab | 9.6 (7.3, 11.4)bc | 5.7 (4.6, 7.6)c† |
| Adipose tissue insulin sensitivity index (lipolysis)* | 74 (54, 102) | 70 (49, 88) | 57 (44, 73) | 26 (20, 38)a† |
| Plasma free fatty acids (µmol/L) | 488 ± 141 | 640 ± 117a | 515 ± 116a | 643 ± 155a |
| Plasma triglycerides (mmol/L) | 0.7 ± 0.3a | 0.8 ± 0.2ab | 1.2 ± 0.3bc | 1.5 ± 0.4c† |
Data are mean ± SD or median (IQR) unless otherwise indicated. ANOVA was used to evaluate differences in outcome variables among groups. Skewed data sets were log transformed to achieve normal distribution before analysis. ASAT, abdominal SAT; BSA, body surface area; FFM, fat-free mass; GIR, glucose infusion rate during stage 2 of the hyperinsulinemic-euglycemic clamp; IAAT, intra-abdominal adipose tissue.
Expressed as 10,000/[(µmol palmitate/kg fat mass/min)×(pmol insulin/L)].
Expressed as 10,000/[(µmol glucose/kg fat-free mass/min)×(pmol insulin/L)].
Linear trend among the obese groups, P < 0.05. Values in each row not sharing letters are significantly different from each other, P < 0.05.
Participants’ Basic Metabolic Characteristics
Total insulin-stimulated whole-body glucose uptake (micromoles per minute), by design, decreased progressively from the insulin-sensitive to the most insulin-resistant obese groups (Table 1). The group differences for whole-body glucose uptake among the obese groups remained even when the data were expressed in relation to body mass, fat-free mass, or body surface area (Table 1). Differences (or similarities) of whole-body glucose uptake between the obese and lean groups, on the other hand, depended on the unit of measurement (Table 1). Adjusting glucose uptake for plasma insulin concentration during the clamp did not affect the results (Table 1) because plasma insulin concentration was not different among the groups (Supplementary Table 1). The glucose infusion rate during the clamp, which provides an assessment of whole-body insulin sensitivity, mirrored whole-body glucose uptake (Table 1).
The hepatic insulin sensitivity index (which assesses the inhibitory effect of insulin on glucose production) and the insulin sensitivity index of adipose tissue lipolysis (which assesses the inhibitory effect of insulin on adipose tissue lipolytic rate) were not different between the insulin-sensitive obese and the lean groups and decreased progressively from the insulin-sensitive to the most insulin-resistant obese groups (Table 1). Plasma triglyceride concentration was also not different between the insulin-sensitive obese and the lean groups and increased progressively from the insulin-sensitive to the most insulin-resistant obese groups (Table 1). Plasma fatty acid concentration was greater in all obese groups compared with the lean group and was not different among the obese groups (Table 1).
Insulin-Stimulated SAT Perfusion and SAT Oxygenation
Adequate SAT perfusion is an important determinant of SAT insulin and glucose delivery and tissue oxygenation. Abdominal SAT perfusion was less in all obese groups compared with the lean group and was not different among the obese groups (Table 2). Abdominal SAT oxygen tension was also less in all obese groups compared with the lean group and was not different among the obese groups (Table 2). Thigh SAT perfusion was not different between the lean and any of the obese groups (Table 2). In the lean group, abdominal SAT perfusion was markedly greater than thigh SAT perfusion (Table 2), whereas in the obese groups abdominal SAT perfusion tended to be greater than thigh SAT perfusion, but the difference was not statistically significant (Table 2).
Table 2.
SAT perfusion and oxygenation
| Lean | Obese | |||
|---|---|---|---|---|
| Insulin sensitive | Quartile 2 | Insulin resistant | ||
| Abdominal SAT perfusion (mL/kg tissue/min) | 33 (24, 56) | 15 (11, 28)* | 18 (2, 57)* | 14 (4, 25)* |
| Abdominal SAT oxygenation (mmHg) | 58 (52, 67) | 49 (45, 53)* | 50 (47, 58)* | 49 (45, 56)* |
| Thigh SAT perfusion (mL/kg tissue/min) | 9 (7, 18)† | 10 (8, 13) | 12 (10, 15) | 8 (7, 11) |
Data are median (IQR). Perfusion was assessed during the hyperinsulinemic-euglycemic clamp at the same time as adipose tissue glucose uptake was assessed. ANOVA was used to evaluate differences in outcome variables among groups. Data were log transformed to achieve normal distribution before analysis.
Significant main effect of obesity (P < 0.05).
Significantly different from abdomen (P < 0.05).
Insulin-Stimulated Glucose Uptake in SAT
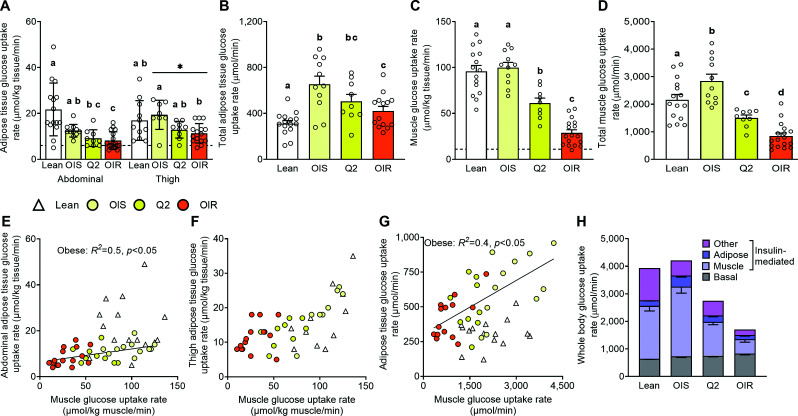
In the obese groups, but not the lean group, glucose uptake rate per kilogram of SAT was less in abdominal than thigh SAT (Fig. 1A). In both regions, glucose uptake rate per kilogram of SAT was not different between the insulin-sensitive obese group and the lean group and was ∼30% less in the insulin-resistant obese than the insulin-sensitive obese group (Fig. 1A). However, because of the large SAT mass in the obese compared with the lean participants, total SAT glucose uptake was greater in all obese groups compared with the lean group (Fig. 1B).
Figure 1.
SAT, skeletal muscle, and total whole-body glucose uptake rates. Insulin-stimulated glucose uptake rate per kilogram of abdominal and thigh SAT (A), total insulin-stimulated adipose tissue glucose uptake rate (B), average insulin-stimulated glucose uptake in skeletal muscles per kilogram of muscle (C), total insulin-stimulated skeletal muscle glucose uptake rate (D), relationship between insulin-stimulated glucose uptake rates in muscle and abdominal SAT (E), relationship between insulin-stimulated glucose uptake rates in muscle and thigh SAT (F), relationship between total insulin-stimulated glucose uptake in SAT and skeletal muscles (G), and biodistribution of insulin-stimulated whole-body glucose uptake rate, which consists of basal (non–insulin stimulated) and insulin-mediated (increase above basal) glucose uptake rate (H), in healthy lean participants and participants with obesity who were grouped according to insulin-simulated whole-body glucose uptake rate. The broken horizontal lines in A and C indicate average basal tissue glucose uptake rates. Values are mean ± SEM (upward error bar only) or median (IQR). ANOVA was used to evaluate differences in outcome variables among groups in A–D. Skewed data sets were log transformed to achieve normal distribution before analysis. Regression analysis was used to evaluate the relationships between outcome variables in panels E–G. Bars not sharing letters are different from each other, P < 0.05. *Main effect of site (thigh vs. abdomen, P < 0.05). OIR, obese insulin-resistant (Q3/4); OIS, obese insulin-sensitive (Q1).
Insulin-Stimulated Glucose Uptake in Skeletal Muscles
Muscle glucose uptake rate per kilogram of muscle was not different between the insulin-sensitive obese group and the lean group (Fig. 1C and Supplementary Fig. 2). However, total muscle glucose uptake (micromoles per minute) was ∼30% greater in the insulin-sensitive obese compared with the lean group because of the greater muscle mass in the obese participants (Fig. 1D). Muscle glucose uptake was ∼75% less in the most insulin-resistant obese than the insulin-sensitive obese group (Fig. 1C and D).
Relationship Between Insulin-Stimulated Glucose Uptake in SAT and Muscle
There was a direct linear relationship between muscle and abdominal SAT glucose uptake rate (expressed per kilogram of tissue) among the obese, but not the lean, participants (Fig. 1E). However, glucose uptake rate per kilogram of tissue was several-fold greater in muscle than SAT. So, the difference in SAT glucose uptake rate between the most insulin-sensitive and the most insulin-resistant obese participants was very small compared with the corresponding difference in muscle glucose uptake rate. There was no relationship between muscle and thigh SAT glucose uptake rate (Figs. 1F). At any total muscle glucose uptake (micromoles per minute) value, total SAT uptake was greater in the obese groups than the lean group because of the greater fat mass in the obese participants (Fig. 1G).
Tissue Distribution of Insulin-Stimulated Whole-Body Glucose Uptake
Insulin-stimulated tissue glucose uptake rates represent the composite of basal glucose uptake rate and the additional glucose uptake induced by insulin infusion (i.e., insulin-mediated glucose uptake). In both the lean and insulin-sensitive obese groups, basal glucose uptake accounted for ∼20% of total insulin-stimulated whole-body glucose uptake, whereas in the most insulin-resistant obese group basal glucose uptake accounted for ∼50% of total insulin-stimulated glucose uptake (Fig. 1H). Insulin-mediated glucose uptake in skeletal muscles accounted for ∼50%, 60%, and 30% in the lean, insulin-sensitive obese, and insulin-resistant obese groups, respectively (Fig. 1H). Insulin-mediated glucose uptake in SAT accounted for <10% and ∼15% and 10%, respectively (Fig. 1H). Approximately 20% of insulin-stimulated whole-body glucose uptake were unaccounted for, presumably representing insulin-mediated glucose uptake in other tissues (15–18).
Adipose Tissue Lipolytic Rate
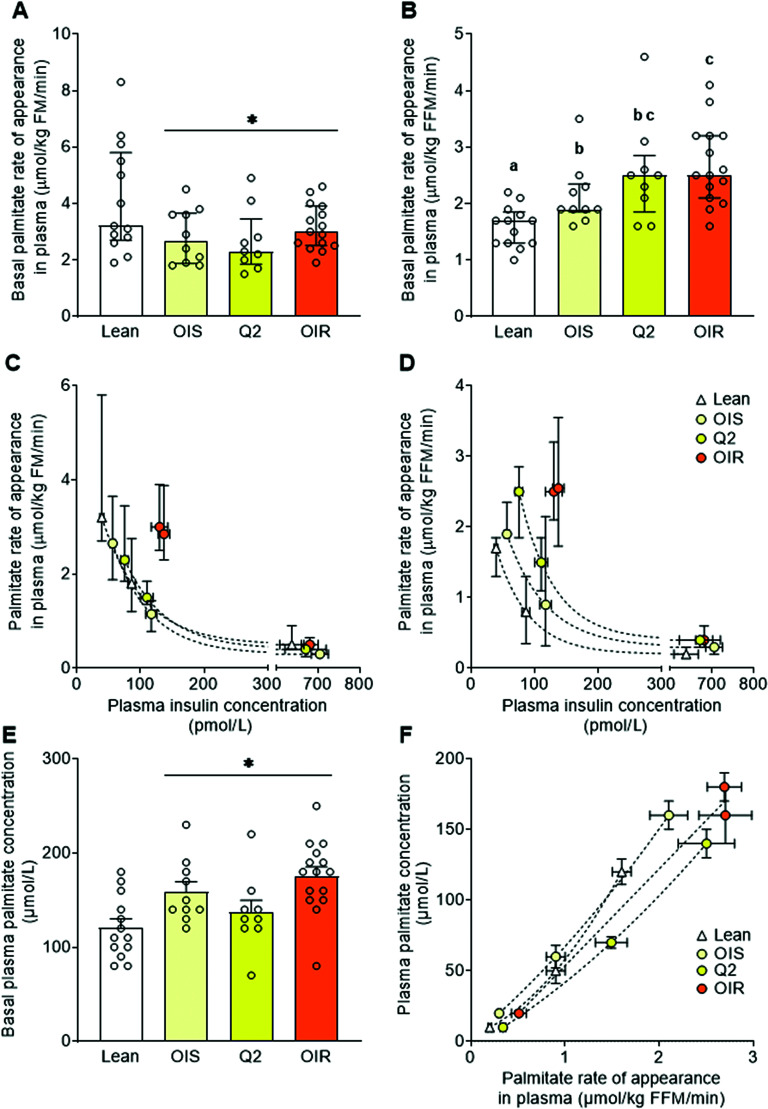
Adipose tissue lipolytic rate was assessed as palmitate Ra in plasma (9). Palmitate Ra was expressed in relation to both fat mass and fat-free mass to provide an assessment of fatty acid release per unit of adipose tissue mass and fatty acid delivery to lean tissues, respectively. Basal palmitate Ra in relation to fat mass was not different among the obese groups and less in all obese groups compared with the lean group (Fig. 2A). Basal palmitate Ra in relation to fat-free mass, on the other hand, was greater in all obese groups compared with the lean group and progressively increased from the insulin-sensitive to the most insulin-resistant obese groups (Fig. 2B). In all groups (lean and obese), there was an inverse relationship between palmitate Ra and plasma insulin concentration assessed during both basal conditions and during the hyperinsulinemic clamp procedure. The relationship between plasma insulin and palmitate Ra in relation to fat mass was not different between the insulin-sensitive obese group and the lean group, but there was marked insulin resistance of adipose tissue lipolysis in the most insulin-resistant obese group (Fig. 2C). Palmitate Ra in relation to fat-free mass was greater in all obese groups compared with the lean group at any given insulin concentration, and the difference was greater in the most insulin-resistant than in the insulin-sensitive obese group (Fig. 2D).
Figure 2.
Palmitate Ra in plasma and plasma palmitate concentration. Basal palmitate Ra in plasma, an index of adipose tissue lipolytic rate, expressed in relation to fat mass (A) and fat-free mass (B); the relationships between plasma insulin concentration during both basal conditions and the hyperinsulinemic-euglycemic clamp procedure and palmitate Ra in plasma expressed in relation to fat mass (C), fat-free mass (D), and basal plasma palmitate concentration (E); and relationship between palmitate Ra in plasma and plasma palmitate concentration (F) in healthy lean participants and participants with obesity who were grouped, by quartiles, according to insulin-simulated whole-body glucose uptake rate. Values are expressed as mean ± SEM (upward error bar only) or median (IQR). ANOVA was used to evaluate differences in outcome variables among groups in A, B, and E. Skewed data sets were log transformed to achieve normal distribution before analysis. Curve fitting was used to evaluate the relationships between outcome variables in panels C, D, and F. Bars not sharing letters are significantly different from each other, P < 0.05. *Significant main effect of obesity, P < 0.05. FFM, fat-free mass; FM, fat mass; OIR, obese insulin-resistant (Q3/4); OIS, obese insulin-sensitive (Q1).
Relationship Between Adipose Tissue Lipolytic Rate and Plasma Fatty Acid Concentration
Basal plasma palmitate concentration (like total free fatty acid concentration) was greater in all obese groups compared with the lean group and was not different among the insulin-sensitive and insulin-resistant obese groups (Figs. 2E and Table 1). At any given palmitate Ra, plasma palmitate concentration was less in the insulin-resistant than the insulin-sensitive obese groups (Fig. 2F) because of increased plasma palmitate clearance in the insulin-resistant obese groups.
Relationships Among Adipose Tissue Insulin Sensitivity, Plasma Fatty Acid Concentrations, and Muscle and Hepatic Insulin Sensitivity
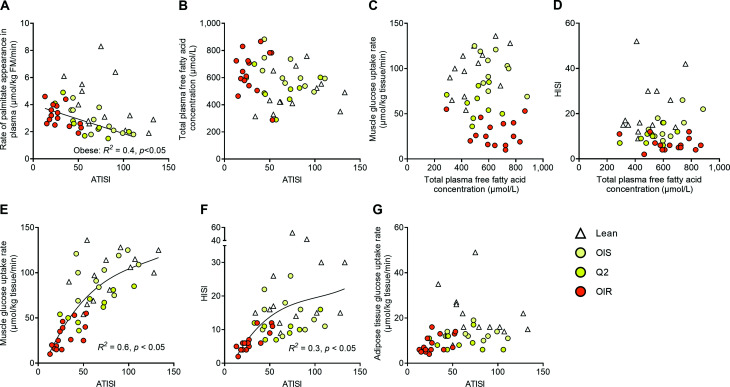
There was no correlation between basal palmitate Ra and insulin sensitivity of adipose tissue lipolysis (i.e., the ability of insulin to suppress lipolysis) in the entire study cohort (all lean and obese participants) (Fig. 3A). However, among the obese participants alone, basal palmitate Ra was inversely related (P < 0.05) to insulin sensitivity of adipose tissue lipolysis. There were no correlations between insulin sensitivity of adipose tissue lipolysis and plasma fatty acid concentration (Fig. 3B), plasma fatty acid concentration and insulin-stimulated muscle glucose uptake (Fig. 3C), and plasma fatty acid concentration and hepatic insulin sensitivity (Fig. 3D) in the entire study cohort or within any of the lean and obese groups. The relationships between insulin sensitivity of adipose tissue lipolysis and insulin-stimulated muscle glucose uptake (Fig. 3E) and insulin sensitivity of adipose tissue lipolysis and hepatic insulin sensitivity (Fig. 3F) were curvilinear. Muscle and hepatic insulin sensitivity correlated (P < 0.05) with insulin sensitivity of adipose tissue lipolysis among the insulin-resistant obese groups but not among the insulin-sensitive obese and the lean groups. Insulin-stimulated SAT glucose uptake was unrelated to insulin sensitivity of adipose tissue lipolysis in the entire study cohort and the subgroups (Fig. 3G).
Figure 3.
Relationships among adipose tissue insulin sensitivity, plasma fatty acid concentration, and muscle and hepatic insulin sensitivity. Relationships between the insulin sensitivity index of adipose tissue lipolysis and palmitate release from adipose tissue (A), the insulin sensitivity index of adipose tissue lipolysis and plasma fatty acid concentration (B), plasma fatty acid concentration and insulin-stimulated muscle glucose uptake rate (C), plasma fatty acid concentration and the hepatic insulin sensitivity index (D), the insulin sensitivity index of adipose tissue lipolysis and insulin-stimulated muscle glucose uptake rate (E), the insulin sensitivity index of adipose tissue lipolysis and the hepatic insulin sensitivity index (F), and the insulin sensitivity index of adipose tissue lipolysis and insulin-stimulated glucose uptake rate in subcutaneous abdominal adipose tissue (G) in healthy lean participants and participants with obesity who were grouped according to insulin-simulated whole-body glucose uptake rate. ATISI, adipose tissue insulin sensitivity index for lipolysis (which assesses the inhibitory effect of insulin on adipose tissue lipolysis); FM, fat mass; HISI, hepatic insulin sensitivity index (which assesses the inhibitory effect of insulin on glucose production); OIR, obese insulin-resistant (Q3/4); OIS, obese insulin-sensitive (Q1).
Adipocyte Size and Adipose Tissue Gene Expression
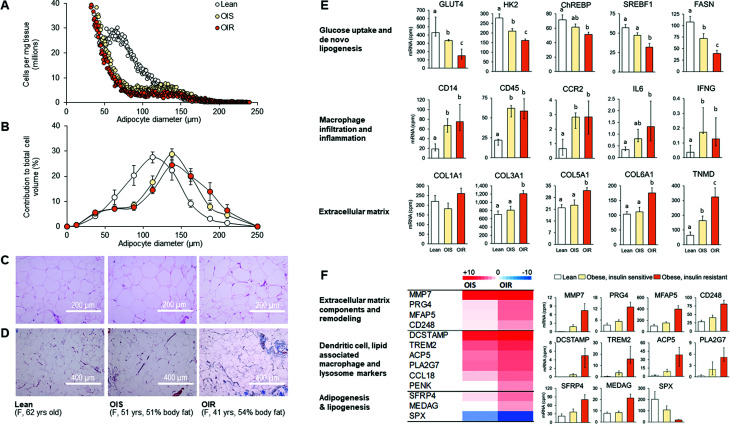
The adipocyte size profile in abdominal SAT was characterized by fewer small (≥50 µm but <100 µm) and more large (≥100 µm but <150 µm) and very large (≥150 µm) cells in the obese groups compared with the lean groups (Fig. 4A–C). Among the obese groups, the total number of large cells (≥100 µm but <150 µm) was greater (361 ± 54 vs. 214 ± 26 cells/ng tissue; P < 0.05) in the insulin-sensitive than in the insulin-resistant group, whereas the total number of very large cells (≥150 µm) was not different (26 ± 8 vs. 31 ± 9 cells/ng tissue; P > 0.05) between the two groups and the total number of small cells (≥50 µm but <100 µm) tended to be greater in the insulin-sensitive compared with the insulin-resistant obese group (642 ± 96 vs. 471 ± 51 cells/ng tissue).
Figure 4.
Adipocyte size, histology, and gene expression in abdominal SAT. Left: adipocyte size distribution profile (A), proportional contribution of differently sized adipocytes to total cell volume (B), and representative hematoxylin-eosin–stained (C) and Masson’s trichrome–stained (D) tissue images in the lean, insulin-sensitive obese, and insulin-resistant obese groups. Values are group averages in A and mean ± SEM in B. Right: mRNA expression of proteins involved in 1) glucose uptake and de novo lipogenesis, 2) markers of macrophage infiltration (CD68, CD206, CCL3 not shown but follow the same pattern as CD14, CD45, and CCR2) and inflammation, and 3) extracellular matrix components in the lean, insulin-sensitive obese, and insulin-resistant obese groups (E). Values are mean ± SEM (upward error bar only) or median (IQR). ANOVA was used to evaluate differences in outcome variables among groups in panels A, B, and E. Skewed data sets were log transformed to achieve normal distribution before analysis. Bars not sharing letters are significantly different from each other, P < 0.05. F: mRNA expression (heat map and representative bar graphs) of key genes that were differentially expressed (false discovery rate <0.05) in the insulin-resistant obese compared with both the insulin-sensitive obese and lean groups. Values are mean ± SEM (upward error bar only) or median (IQR). ACP5, acid phosphatase 5; CCL18, C-C motif chemokine ligand 18; CCR2, C-C motif chemokine receptor 2; CD14, cluster of differentiation 14; CD45, cluster of differentiation 45; CD248, endosialin; ChREBP, carbohydrate response element binding protein; COL1A1, collagen type I α-1; COL3A1, collagen type III α-1; COL5A1, collagen type IV α-1; DCSTAMP, dendritic cell–specific transmembrane protein; F, female; FASN, fatty acid synthase; HK2, hexokinase 2; IFNG, interferon γ; IL6, interleukin 6; MEDAG, mesenteric estrogen-dependent adipogenesis; MFAP5, microfibril-associated protein 5; MMP7, matrix metalloproteinase type 7; OIR, obese insulin-resistant (Q3/4); OIS, obese insulin-sensitive (Q1); PENK, proenkephalin; PLA2G7, phospholipase A2 group VII; PRG4, proteoglycan 4; SFRP4, secreted frizzled-related protein 4; SPX, spexin; SREBF1, sterol regulatory element binding transcription factor 1; TNMD, tenomodulin; TREM2, triggering receptor expressed on myeloid cells 2; yrs, years.
The mRNA expression of genes involved in glucose uptake and de novo fatty acid synthesis was lesser and the mRNA expression of extracellular matrix markers was greater in the insulin-resistant obese than in the insulin-sensitive obese and lean groups (Fig. 4E). This finding was corroborated by increased Masson’s trichrome connective tissue staining in the insulin-resistant obese group (Fig. 4D). In contrast, the mRNA expression of markers of macrophage infiltration and inflammation was greater in the obese than in the lean groups without a difference between the insulin-sensitive and insulin-resistant obese groups (Fig. 4E). In addition, we found that 49 genes were more than twofold different (48 greater, 1 lower) in the insulin-resistant obese compared with both the insulin-sensitive obese and lean groups. Key genes that were upregulated in the insulin-resistant obese group included those that encode extracellular matrix components and proteins involved in extracellular matrix remodeling, and proteins associated with dendritic cells, lipid-associated macrophages, lysosomal activity, and lipogenesis and adipogenesis (Fig. 4F). Because lysosomal lipid hydrolysis has been implicated in regulating adipose tissue fatty acid release and systemic metabolic function in obese mice (19), we subsequently evaluated the expression of additional genes involved in this process and found that several of them were also upregulated in the insulin-resistant obese compared with both the insulin-sensitive obese and lean groups (Supplementary Fig. 3). The mRNA expression of SPX, a protein involved in adipose tissue lipid turnover, was downregulated in the insulin-resistant obese compared with the insulin-sensitive obese and lean groups (Fig. 4F).
Discussion
We hypothesized that channeling of glucose uptake into SAT is responsible for high insulin-stimulated whole-body glucose uptake in some people with obesity not only because SAT provides an additional important site for glucose disposal but also because it prevents adipocyte hypertrophy and associated adipose tissue dysfunction that has been implicated in causing insulin resistance. However, we found that high insulin-stimulated whole-body glucose uptake in insulin-sensitive participants with obesity was not due to preferential channeling of glucose into SAT but, rather, was due to high insulin-stimulated muscle glucose uptake. Furthermore, adipocytes were much larger, SAT perfusion and oxygenation were lower, and markers of SAT inflammation were higher in insulin-sensitive obese participants than lean participants. Despite these presumably harmful SAT alterations in insulin-sensitive obese participants, basal adipose tissue lipolytic rate, the inhibitory effect of insulin on adipose tissue lipolysis, and insulin-stimulated muscle glucose uptake rate were not different between insulin-sensitive obese and lean participants. Fatty acid appearance rate in relation to fat-free mass and plasma fatty acid concentration were greater in insulin-sensitive obese than in lean participants because of the greater fat mass in the obese. These data demonstrate that in people with obesity who are as insulin-sensitive as lean people, SAT exhibits many of the features that are thought to cause adipose tissue and muscle insulin resistance. These findings therefore suggest that these SAT features are normal consequences of SAT expansion and are unrelated to insulin resistance.
The fatty acid kinetic data in the current study demonstrate that the sensitivity of adipose tissue lipolysis to the inhibitory effect of insulin per se is not a determinant of fatty acid release from adipose tissue and plasma fatty acid concentration and, presumably, cellular fatty acid availability in lean tissues. First, the high basal plasma insulin concentration in participants with obesity fully compensated for insulin resistance of adipose tissue lipolysis. In addition, plasma fatty acid clearance was enhanced in insulin-resistant compared with insulin-sensitive participants with obesity. These findings corroborate previous observations that suggest SAT mass is a major determinant of fatty acid release into the circulation and that fatty acid release relative to fat mass is greater in lean people than people with obesity (20,21). These findings also highlight the potential importance of increased fatty acid extraction by lean tissues in insulin-resistant people (22–24), which helps explain the dissociation between plasma fatty acid concentration and insulin sensitivity in the present and previous studies (4,5). Accelerated plasma fatty acid clearance by lean tissues (including muscle and liver) presumably increases intracellular fatty acid appearance in these tissues, which can impair insulin signaling (2) and cause impaired insulin-stimulated muscle glucose uptake and increased hepatic glucose production, lipogenesis, and triglyceride secretion (25–27). We found that plasma triglyceride concentration was greater in the insulin-resistant obese than in both the insulin-sensitive obese and lean groups. These data support the common association between plasma triglyceride concentration and insulin resistance (22,28) and suggest that tissue-specific factors that regulate fatty acid uptake and intracellular fatty acid handling (29) are important determinants of lipid-induced muscle insulin resistance. In addition, triglycerides can exacerbate fatty acid–induced muscle insulin resistance (30–32), presumably because triglyceride-derived fatty acids are an important component of total fatty acid uptake in skeletal muscles (22,33–35).
We observed no relationship between insulin sensitivity of adipose tissue lipolysis (i.e., the inhibitory effect of insulin on adipose tissue lipolytic rate) and insulin-stimulated glucose uptake in SAT. The apparent uncoupling of insulin action on adipose tissue lipolysis and glucose uptake is most likely due to the very high insulin sensitivity of glucose uptake in adipocytes, which results in near-maximal glucose uptake rates at basal plasma insulin concentrations (36). In addition, we observed a direct, but curvilinear, relationship between insulin sensitivity of adipose tissue lipolysis and insulin-stimulated muscle glucose uptake and a direct, but curvilinear, relationship between insulin sensitivity of adipose tissue lipolysis and hepatic insulin sensitivity (ability of insulin to suppress glucose production). The relationships among insulin sensitivity in adipose tissue, muscle, and liver were tightest among the most insulin-resistant obese participants and weak or not significant among the insulin-sensitive obese and lean participants. This suggests that severe insulin resistance is a systemic phenomenon but insulin sensitivity differs by tissue/organ in people with moderate insulin resistance and particularly those who are insulin sensitive.
It has been proposed that adipocyte hypertrophy causes insulin resistance because it impairs adipose tissue perfusion and oxygenation, resulting in adipose tissue inflammation and fibrosis (3,37). In addition, it has been proposed that insulin resistance itself impairs tissue perfusion (38,39), resulting in a vicious cycle that exaggerates metabolic dysfunction. However, we found that adipocytes were much larger in both the insulin-sensitive and insulin-resistant obese groups compared with the lean group. In addition, there was only a small shift toward even larger cells in the insulin-resistant compared with the insulin-sensitive obese group, as previously observed (40,41). Abdominal SAT perfusion and oxygenation were also significantly lower in both the insulin-sensitive and insulin-resistant obese groups than the lean group and were not different between the insulin-sensitive and insulin-resistant obese groups. Thigh SAT perfusion was not different between any of the obese and lean groups. These data confirm the results from previous studies of similar regional differences in SAT perfusion and differences in SAT perfusion and oxygenation between insulin-resistant obese and lean people (both qualitatively and quantitatively) (10,11,38,39). However, they contradict the results from studies that included both lean and obese participants where investigators concluded that insulin resistance is associated with decreased SAT oxygenation (10,11), presumably because the independent effect of increased fat mass could have confounded this assessment. The composite of available data suggests that insulin resistance is not responsible for reduced abdominal SAT perfusion in people with obesity and, vice versa, SAT perfusion and oxygenation are not major determinants of adipose tissue, whole-body, and muscle insulin sensitivity.
We also found that SAT markers of macrophage infiltration and inflammation were increased in both the insulin-sensitive and insulin-resistant obese groups, compared with the lean group, and they were not different between the insulin-sensitive and insulin-resistant obese groups. Although these findings contradict the results from many studies on rodents, they are in agreement with the bulk of data obtained in people. Even in studies that demonstrated an increase in some markers of inflammation in adipose tissue of metabolically unhealthy compared with metabolically healthy people with obesity, the differences were small and often only present in visceral adipose tissue—not SAT (1,42). In addition, several studies in both mice and people concluded that increased fat mass itself is associated with an increase in SAT macrophages (19,43,44).
Our gene expression analysis specifically identified increased SAT expression of markers of lipid-associated macrophages, lysosomes, lysosomal lipid hydrolysis, and dendritic cells (19,45) in the insulin-resistant obese compared with the insulin-sensitive obese and lean groups. In mice, these immune cells are involved in regulating adipose tissue lipid homeostasis; in addition, lysosomal acid lipid hydrolysis is an important determinant of fatty acid release from adipose tissue (19,46,47). These features may thus have contributed (in addition to impaired insulin action on neutral lipases) to the greater fatty acid release at any plasma insulin concentration in the insulin-resistant obese compared with the insulin-sensitive obese and lean groups in the current study. Our gene expression analysis also revealed that markers of extracellular matrix deposition are increased in insulin-resistant obese compared with insulin-sensitive obese and lean participants. Although the mechanisms involved in adipose tissue remodeling are unclear, the increase in extracellular matrix could provide an explanation for insulin resistance. In mice, increased extracellular matrix accumulation in SAT is associated with metabolic abnormalities, and genetic disruption of SAT extracellular matrix formation prevents them (37,48).
We found significant differences in insulin-stimulated SAT glucose uptake rates between the insulin-sensitive and the insulin-resistant obese groups and also regional differences in SAT glucose uptake rates (i.e., greater in thigh than abdomen) among the obese groups. The clinical significance of these findings is unclear because SAT glucose uptake, compared with muscle glucose uptake, is low and the relative difference between the insulin-resistant and insulin-sensitive obese groups in both SAT depots was similar. Furthermore, the marked difference in insulin-stimulated whole-body glucose uptake between the insulin-sensitive and insulin-resistant obese groups was due to insulin-resistant muscle, not SAT, glucose uptake. Therefore, it is unlikely that the beneficial effect of lower, compared with upper, body fat accumulation (1) is due to increased glucose uptake in lower-body SAT.
We measured tissue glucose uptake rates but not intracellular glucose handling. Therefore, we cannot rule out that the lower glucose uptake per kilogram of SAT in insulin-resistant compared with insulin-sensitive people with obesity has potentially important consequences. Studies conducted in mice demonstrated that reducing the conversion of glucose to fatty acids in SAT can cause SAT dysfunction by increasing macrophage recruitment and inflammation (6,49). Nevertheless, we consider this unlikely because de novo fatty acid synthesis in abdominal SAT is minimal (<2%) and not different between insulin-sensitive and insulin-resistant people with obesity (27). Furthermore, thigh SAT glucose uptake was not different in any of the obese groups compared with the lean group. Moreover, markers of macrophages and inflammation in SAT were greater in the obese groups than in the lean group and not different in the insulin-sensitive and insulin-resistant obese groups.
In summary, the findings from our study suggest that many SAT factors that are thought to cause insulin resistance, including enlarged adipocytes, reduced SAT perfusion and oxygenation, SAT inflammation, and increased fatty acid release in relation to fat-free mass and plasma fatty acid concentration, were also found in participants with obesity who were as insulin sensitive as lean participants; furthermore, they were only marginally or not different between participants with obesity who were insulin sensitive or insulin resistant. Therefore, these presumably harmful alterations in SAT function appear to be normal consequences of SAT expansion and are unrelated to insulin resistance.
Article Information
Acknowledgments. The authors thank the staff of the Center for Human Nutrition, the Clinical Translational Research Unit, the Clinical Translational Imaging Unit, and the Division of Radiological Sciences for help with participant recruitment, scheduling, and testing and technical assistance with sample processing and data analysis, and the study participants for their time and commitment.
Funding. The work presented in this manuscript was supported by National Institutes of Health grants R01 DK115400, P30 DK56341 (Nutrition Obesity Research Center), P30 DK020579 (Diabetes Research Center), P30 DK052574 (Digestive Disease Research Core Center), and UL1TR000448 (Clinical Translational Science Award) and grants from the American Diabetes Association (1-18-ICTS-119) and the Longer Life Foundation.
Duality of Interest. No potential conflicts of interest relevant to this article were reported.
Author Contributions. R.J.G. and B.M. designed the study. H.-C.E.K., S.v.V., T.A.P., G.A.M., B.R., R.L., R.J.G., and B.M. contributed to data acquisition, data analysis, and data interpretation. H.-C.E.K. and B.M. wrote the first draft of the manuscript. All authors contributed to the revision of the manuscript for crucial intellectual content and approved the final manuscript for publication. B.M. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Footnotes
This article contains supplementary material online at https://doi.org/10.2337/figshare.14939088.
References
- 1. Smith GI, Mittendorfer B, Klein S. Metabolically healthy obesity: facts and fantasies. J Clin Invest 2019;129:3978–3989 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Petersen MC, Shulman GI. Mechanisms of insulin action and insulin resistance. Physiol Rev 2018;98:2133–2223 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Ghaben AL, Scherer PE. Adipogenesis and metabolic health. Nat Rev Mol Cell Biol 2019;20:242–258 [DOI] [PubMed] [Google Scholar]
- 4. Karpe F, Dickmann JR, Frayn KN. Fatty acids, obesity, and insulin resistance: time for a reevaluation. Diabetes 2011;60:2441–2449 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Arner P, Rydén M. Fatty acids, obesity and insulin resistance. Obes Facts 2015;8:147–155 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Kahn BB. Adipose tissue, inter-organ communication, and the path to type 2 diabetes: the 2016 Banting Medal for Scientific Achievement Lecture. Diabetes 2019;68:3–14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Shepherd PR, Gnudi L, Tozzo E, Yang H, Leach F, Kahn BB. Adipose cell hyperplasia and enhanced glucose disposal in transgenic mice overexpressing GLUT4 selectively in adipose tissue. J Biol Chem 1993;268:22243–22246 [PubMed] [Google Scholar]
- 8. Smith GI, Yoshino J, Kelly SC, et al. High-protein intake during weight loss therapy eliminates the weight-loss-induced improvement in insulin action in obese postmenopausal women. Cell Rep 2016;17:849–861 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Mittendorfer B, Liem O, Patterson BW, Miles JM, Klein S. What does the measurement of whole-body fatty acid rate of appearance in plasma by using a fatty acid tracer really mean? Diabetes 2003;52:1641–1648 [DOI] [PubMed] [Google Scholar]
- 10. Cifarelli V, Beeman SC, Smith GI, et al. Decreased adipose tissue oxygenation associates with insulin resistance in individuals with obesity. J Clin Invest 2020;130:6688–6699 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Lempesis IG, van Meijel RLJ, Manolopoulos KN, Goossens GH. Oxygenation of adipose tissue: a human perspective. Acta Physiol (Oxf) 2020;228:e13298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Laforest S, Michaud A, Paris G, et al. Comparative analysis of three human adipocyte size measurement methods and their relevance for cardiometabolic risk. Obesity (Silver Spring) 2017;25:122–131 [DOI] [PubMed] [Google Scholar]
- 13. Ter Horst KW, Serlie MJ. Normalization of metabolic flux data during clamp studies in humans. Metabolism 2020;104:154168. [DOI] [PubMed] [Google Scholar]
- 14. Dadson P, Landini L, Helmiö M, et al. Effect of bariatric surgery on adipose tissue glucose metabolism in different depots in patients with or without type 2 diabetes. Diabetes Care 2016;39:292–299 [DOI] [PubMed] [Google Scholar]
- 15. Honka H, Mäkinen J, Hannukainen JC, et al. Validation of [18F]fluorodeoxyglucose and positron emission tomography (PET) for the measurement of intestinal metabolism in pigs, and evidence of intestinal insulin resistance in patients with morbid obesity. Diabetologia 2013;56:893–900 [DOI] [PubMed] [Google Scholar]
- 16. Koffert JP, Mikkola K, Virtanen KA, et al. Metformin treatment significantly enhances intestinal glucose uptake in patients with type 2 diabetes: results from a randomized clinical trial. Diabetes Res Clin Pract 2017;131:208–216 [DOI] [PubMed] [Google Scholar]
- 17. Boersma GJ, Johansson E, Pereira MJ, et al. Altered glucose uptake in muscle, visceral adipose tissue, and brain predict whole-body insulin resistance and may contribute to the development of type 2 diabetes: a combined PET/MR study. Horm Metab Res 2018;50:627–639 [DOI] [PubMed] [Google Scholar]
- 18. Ferrannini E, Iozzo P, Virtanen KA, Honka MJ, Bucci M, Nuutila P. Adipose tissue and skeletal muscle insulin-mediated glucose uptake in insulin resistance: role of blood flow and diabetes. Am J Clin Nutr 2018;108:749–758 [DOI] [PubMed] [Google Scholar]
- 19. Xu X, Grijalva A, Skowronski A, van Eijk M, Serlie MJ, Ferrante AW Jr. Obesity activates a program of lysosomal-dependent lipid metabolism in adipose tissue macrophages independently of classic activation. Cell Metab 2013;18:816–830 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Mittendorfer B, Magkos F, Fabbrini E, Mohammed BS, Klein S. Relationship between body fat mass and free fatty acid kinetics in men and women. Obesity (Silver Spring) 2009;17:1872–1877 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Horowitz JF, Coppack SW, Paramore D, Cryer PE, Zhao G, Klein S. Effect of short-term fasting on lipid kinetics in lean and obese women. Am J Physiol 1999;276:E278–E284 [DOI] [PubMed] [Google Scholar]
- 22. Bickerton AS, Roberts R, Fielding BA, et al. Adipose tissue fatty acid metabolism in insulin-resistant men. Diabetologia 2008;51:1466–1474 [DOI] [PubMed] [Google Scholar]
- 23. Van Pelt DW, Newsom SA, Schenk S, Horowitz JF. Relatively low endogenous fatty acid mobilization and uptake helps preserve insulin sensitivity in obese women. Int J Obes 2015;39:149–155 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Viljanen AP, Lautamäki R, Järvisalo M, et al. Effects of weight loss on visceral and abdominal subcutaneous adipose tissue blood-flow and insulin-mediated glucose uptake in healthy obese subjects. Ann Med 2009;41:152–160 [DOI] [PubMed] [Google Scholar]
- 25. Sparks JD, Sparks CE, Adeli K. Selective hepatic insulin resistance, VLDL overproduction, and hypertriglyceridemia. Arterioscler Thromb Vasc Biol 2012;32:2104–2112 [DOI] [PubMed] [Google Scholar]
- 26. Mittendorfer B, Yoshino M, Patterson BW, Klein S. VLDL triglyceride kinetics in lean, overweight, and obese men and women. J Clin Endocrinol Metab 2016;101:4151–4160 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Smith GI, Shankaran M, Yoshino M, et al. Insulin resistance drives hepatic de novo lipogenesis in nonalcoholic fatty liver disease. J Clin Invest 2020;130:1453–1460 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Reaven GM. Pathophysiology of insulin resistance in human disease. Physiol Rev 1995;75:473–486 [DOI] [PubMed] [Google Scholar]
- 29. Fabbrini E, Magkos F, Mohammed BS, et al. Intrahepatic fat, not visceral fat, is linked with metabolic complications of obesity. Proc Natl Acad Sci USA 2009;106:15430–15435 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Piatti PM, Monti LD, Baruffaldi L, et al. Effects of an acute increase in plasma triglyceride levels on glucose metabolism in man. Metabolism 1995;44:883–889 [DOI] [PubMed] [Google Scholar]
- 31. Storgaard H, Jensen CB, Björnholm M, et al. Dissociation between fat-induced in vivo insulin resistance and proximal insulin signaling in skeletal muscle in men at risk for type 2 diabetes. J Clin Endocrinol Metab 2004;89:1301–1311 [DOI] [PubMed] [Google Scholar]
- 32. Sakata S, Katsumi S, Mera Y, et al. Mechanism of action of hypoglycemic effects of an intestine-specific inhibitor of microsomal triglyceride transfer protein (MTP) in obese rats. J Pharmacol Sci 2015;127:103–108 [DOI] [PubMed] [Google Scholar]
- 33. Andersen IR, Søndergaard E, Sørensen LP, et al. Increased VLDL-TG fatty acid storage in skeletal muscle in men with type 2 diabetes. J Clin Endocrinol Metab 2017;102:831–839 [DOI] [PubMed] [Google Scholar]
- 34. Hames KC, Vella A, Kemp BJ, Jensen MD. Free fatty acid uptake in humans with CD36 deficiency. Diabetes 2014;63:3606–3614 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Jans A, Konings E, Goossens GH, et al. PUFAs acutely affect triacylglycerol-derived skeletal muscle fatty acid uptake and increase postprandial insulin sensitivity. Am J Clin Nutr 2012;95:825–836 [DOI] [PubMed] [Google Scholar]
- 36. Ciaraldi TP, Kolterman OG, Olefsky JM. Mechanism of the postreceptor defect in insulin action in human obesity. Decrease in glucose transport system activity. J Clin Invest 1981;68:875–880 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Williams AS, Kang L, Wasserman DH. The extracellular matrix and insulin resistance. Trends Endocrinol Metab 2015;26:357–366 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Frayn KN, Karpe F. Regulation of human subcutaneous adipose tissue blood flow. Int J Obes 2014;38:1019–1026 [DOI] [PubMed] [Google Scholar]
- 39. Hodson L, Humphreys SM, Karpe F, Frayn KN. Metabolic signatures of human adipose tissue hypoxia in obesity. Diabetes 2013;62:1417–1425 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. McLaughlin T, Lamendola C, Coghlan N, et al. Subcutaneous adipose cell size and distribution: relationship to insulin resistance and body fat. Obesity (Silver Spring) 2014;22:673–680 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. McLaughlin T, Sherman A, Tsao P, et al. Enhanced proportion of small adipose cells in insulin-resistant vs insulin-sensitive obese individuals implicates impaired adipogenesis. Diabetologia 2007;50:1707–1715 [DOI] [PubMed] [Google Scholar]
- 42. Fryk E, Olausson J, Mossberg K, et al. Hyperinsulinemia and insulin resistance in the obese may develop as part of a homeostatic response to elevated free fatty acids: a mechanistic case-control and a population-based cohort study. EBioMedicine 2021;65:103264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Jia Q, Morgan-Bathke ME, Jensen MD. Adipose tissue macrophage burden, systemic inflammation, and insulin resistance. Am J Physiol Endocrinol Metab 2020;319:E254–E264 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Ortega Martinez de Victoria E, Xu X, Koska J, et al. Macrophage content in subcutaneous adipose tissue: associations with adiposity, age, inflammatory markers, and whole-body insulin action in healthy Pima Indians. Diabetes 2009;58:385–393 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Jaitin DA, Adlung L, Thaiss CA, et al. Lipid-associated macrophages control metabolic homeostasis in a Trem2-dependent manner. Cell 2019;178:686–698.e14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Zechner R, Madeo F, Kratky D. Cytosolic lipolysis and lipophagy: two sides of the same coin. Nat Rev Mol Cell Biol 2017;18:671–684 [DOI] [PubMed] [Google Scholar]
- 47. Russo L, Lumeng CN. Properties and functions of adipose tissue macrophages in obesity. Immunology 2018;155:407–417 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Khan T, Muise ES, Iyengar P, et al. Metabolic dysregulation and adipose tissue fibrosis: role of collagen VI. Mol Cell Biol 2009;29:1575–1591 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Vijayakumar A, Aryal P, Wen J, et al. Absence of carbohydrate response element binding protein in adipocytes causes systemic insulin resistance and impairs glucose transport. Cell Rep 2017;21:1021–1035 [DOI] [PMC free article] [PubMed] [Google Scholar]