Abstract
Antimicrobial resistance (AMR) is a significant threat to global health. The conventional antibiotic pool has been depleted, forcing the investigation of novel and alternative antimicrobial strategies. Antimicrobial peptides (AMPs) have shown potential as alternative diagnostic and therapeutic agents in biomedical applications. To date, over 3000 AMPs have been identified, but only a fraction of these have been approved for clinical trials. Their clinical applications are limited to topical application due to their systemic toxicity, susceptibility to protease degradation, short half-life, and rapid renal clearance. To circumvent these challenges and improve AMP’s efficacy, different approaches such as peptide chemical modifications and the development of AMP delivery systems have been employed. Nanomaterials have been shown to improve the activity of antimicrobial drugs by providing support and synergistic effect against pathogenic microbes. This paper describes the role of nanotechnology in the targeted delivery of AMPs, and some of the nano-based delivery strategies for AMPs are discussed with a clear focus on metallic nanoparticle (MNP) formulations.
Keywords: antimicrobial peptides, antimicrobial resistance, nanotechnology, nanocarriers, drug resistance, nanoparticles, drug delivery systems
1. Introduction
Antibiotics have been used for decades to cure infectious diseases and enabled most of modern medicine. Without antibiotics, even routine medical procedures can lead to life-threatening infections. The first widely used antibiotic, penicillin, was discovered in 1928. In 1945, Alexander Fleming warned that bacterial resistance had the potential to ruin the miracle of antibiotics [1]. Shortly thereafter, beta-lactam antibiotics were discovered and proved to be effective against penicillin-resistant microbes [2,3]. This was followed by the methicillin-resistant Staphylococcus aureus (MRSA) [4]; since then, resistance has been reported against almost all known antibiotics to date [5]. In the high-priority list of antibiotic-resistant strains that require urgent attention are Enterococcus faecium, Staphylococcus aureus (S. aureus), Klebsiella pneumoniae (K. pneumoniae), Acinetobacter baumannii, Pseudomonas aeruginosa (P. aeruginosa), and Enterobacter species (ESKAPE). The ESKAPE are among the 12 microorganisms that are listed as critical to medium priority pathogens by the World Health Organization (WHO). These bacteria place a significant burden on the healthcare systems and global economic costs [2,3]. Efforts to impede the spread of these pathogens have been hindered by their ability to resist antibacterial drugs.
As it stands, the misuse and overuse of these drugs are the major contributing factors toward antibiotic resistance, which often reduces the efficacy of newly discovered antibiotics and their derivatives [6]. Antibiotic-resistant infections account for about 700,000 mortalities per year, which is estimated to increase to over 10 million deaths by 2050, making AMR a global health crisis [7]. AMR occurs naturally, but the process has been accelerated by the misuse of antibiotics in both humans and animals. As reported by WHO, increased hospitalization, higher medical costs, and elevated mortality have been associated with AMR. AMR threatens the successful treatment of infections caused by drug-resistant microbes [8]. Infectious diseases such as pneumonia, tuberculosis, gonorrhea, malaria, HIV/AIDS, and salmonellosis are becoming increasingly difficult to manage as known potent antimicrobial agents are becoming less effective. In this context, the loss of antimicrobial potency due to resistance in addition to the lack of new and alternative antimicrobial agents underscores the requirement for novel therapeutic agents.
AMPs of natural and artificial origin have gained attention in recent years due to their biological activities as potential alternatives to conventional antimicrobial agents that can combat pathogenic and drug-resistant microorganisms [9]. These biomolecules serve as a natural first-line of defense system against invading pathogens, working either individually or synergistically with the innate immune system to inhibit the growth of pathogenic microorganisms. AMPs are divided into four categories based on their structural conformation and characteristics. This class of peptides (AMPs) are stable over a wide pH range, can work in synergy with immune cells, and possess effective antimicrobial activities against all kinds of pathogens including viruses. The discovery of defensins among other identified AMPs have been a major breakthrough as alternative molecules for use against antibiotic-resistance and in the development of novel antimicrobial agents [10].
Over 14 manually curated AMP databases have been developed to provide information and enable researchers to synthesize AMPs with better therapeutic index. Examples of these databases include the Database of Antimicrobial Activity and Structure of Peptides (DBAASP v3.0) [11], The Antimicrobial Peptide Database (APD3) [12], dbAMP [13], Yet Another Database of Antimicrobial Peptides (YADAMP), etc. To give insight into the growing number of available AMPs, the APD3 for example is composed of 3257 AMPs from six kingdoms (bacteria, archaea, protists, fungi, plants, and animals) with a broad spectrum of antimicrobial activities [14].
AMPs have been used in the treatment of microbial infections emerging from bacteria, fungi, and viruses [15]. The two most studied AMPs of human origin are the cathelicidin LL-37 and defensins. The antiviral effect of defensins has been demonstrated against human viral infections [16,17,18]. The recombinant human β-defensins (mouse β-defensin 3) showed antiviral activity against influenza A virus in both in vitro and in vivo studies [17]. Several studies have also demonstrated the immunomodulatory [19,20], antimicrobial [21], and wound-healing activities of the human LL-37 [22]. Of note, LL-37 interacts with keratinocytes through the P2X7–SFK–Akt–CREB/ATF1 signaling [23,24] and COX-2 [25] pathways as well as with fibroblasts through the P2X7R pathway [26] and the protein kinase/ERK pathway, supporting its healing effect in polymicrobial-infected wounds [27,28].
Despite the health properties offered by AMPs, they have some limitations, which ultimately delay their progress into clinical trials [15]. Chemical modification of the AMPs and the use of delivery systems have been reported to improve their pharmacokinetics [29]. Nanotechnology-based delivery systems are now being considered as effective alternatives to increase the therapeutic efficacy of the AMPs by preventing proteolysis, increase AMP accumulation at the infection sites, and reduce bystander toxicity [30]. The review paper summarizes the nano-delivery systems for AMPs, current progress, and their applications.
2. Antimicrobial Agents and their Activity
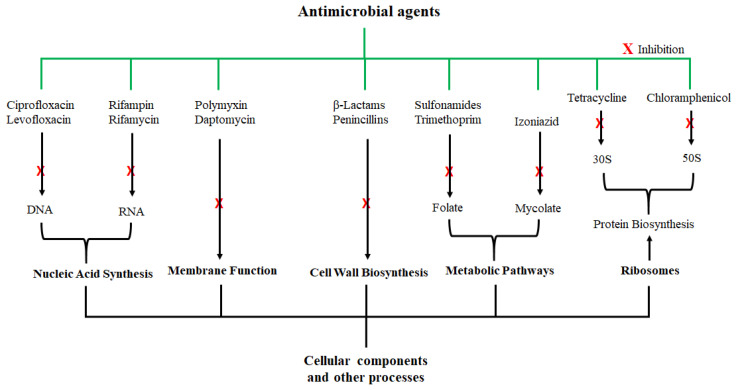
The correlation between microorganisms and infectious diseases is well established. Therefore, molecules that can kill, inhibit, or slow down the growth of these pathogens are vital for treatment of microbial infections. After the discovery of Penicillium notatum by Sir Alexander Fleming in 1928, several other antimicrobial agents were identified, and their mechanisms of antimicrobial activity have also been thoroughly investigated. Some of the antimicrobial agents and their modes of action are shown in Figure 1; they exert their antimicrobial actions by interfering with various cellular and metabolic processes of the microorganisms. Actions of antibacterial agents can either be bacteriostatic if the antimicrobial activity involves growth inhibition or bactericidal if the activity involves membrane disruption leading to death of the bacteria [31]. Sulfonamide and spectinomycin inhibit folate and protein synthesis, respectively; and are classified as bacteriostatic agents. Antibacterial agents such as vancomycin and penicillin are bactericidal agents due to their killing effect on bacteria. Other classifications of antibacterial agents are based on the mechanism of inhibition, origin, composition, and spectrum activity [32]. While these agents were initially considered highly potent against certain microorganisms, the development of microbial resistance has been reported for nearly all these antibiotics. To make matters worse, the rate of discovering new antimicrobial agents has also been declining.
Figure 1.
Examples of antimicrobial agents and their modes of action. These compounds are classified according to their cellular or molecular targets.
The misuse and overuse of antibiotics is the main cause of AMR, and have led to the increasing resistance of human and animal pathogens to these antimicrobial agents. The mode of microbial resistance toward the antimicrobial agents include (a) alteration or modification of the drug, the drug target, and drug binding, (b) inactivation of drugs, (c) blockage or decrease in drug uptake or penetration, (d) increase in drug efflux, and (e) the degradation of the drug. In 2015, 8.7% and 24.3% AMR were reported for MRSA and Streptococcus pneumoniae, respectively [33], and the mechanism of bacterial resistance was through drug inactivation, increased efflux, and ribosomal protection [34].
3. Overview and Properties of AMPs
AMPs, also known as host defense peptides, are short amino acid sequences or biomolecules, ranging from 12 to 100 amino acids in length. AMPs possess antimicrobial activities against various microorganisms and are crucial to both the innate and the acquired immune systems as a defense mechanism [35]. Some of these AMPs have also been shown to have antimicrobial activities against multidrug-resistant (MDR) bacterial strains. They are cationic and amphiphilic in nature [36], and these characteristics play a major role in the mechanism by which they intercalate with the phospholipid bilayer of the microbial cell membrane, leading to membrane depolarization and cell permeabilization [37]. Consequently, this will cause the release of biologically important cellular contents and subsequently result in microbial death [38,39].
AMPs are classified based on their sequence composition, structure, and origin; typical AMP structural conformations are highlighted in Figure 2 and Table 1. The structural and physicochemical properties of AMPs, which include charge, hydrophobicity, and amphipathicity, dictate their specificity against the target microorganisms [40].
Figure 2.
Structural conformations of AMPs. (A) α-helical, (B) β-sheet, (C) αβ-peptides, and (D). Non-αβ peptides or extended structure. Images were deciphered by Schrodinger software v2020-3 after structural retrieval from the Protein Data Bank (PDB) at https://www.rcsb.org/ (accessed on 20 May 2021) using the PDB IDs: 2K6O, 1ZMM, 1FD3, and 1G89 for A to D, respectively. AMPs are colored by properties.
Table 1.
Classifications of some AMPs with specific examples.
| Groups | Characteristics | Examples | Mode of Action | Refs |
|---|---|---|---|---|
| α-helical peptides | Amidated C-terminus, N-terminal signal peptides |
FALL-39 Magainins Cecropins |
Pore formation | [47] |
| [48,49] | ||||
| [50] | ||||
| β-sheet | cationic with disulfide bridges | β-defensins | Membrane disruption | [51,52] |
| plectasin | [53] | |||
| protegrins | [54] | |||
| Extended AMPs or Non-αβ peptides | Contains proline, arginine, tryptophan, glycine or histidine rich amino acids | Indolicidin | Membrane disruption Disruption of intracellular function |
[55] |
| Bactenecins | [56] | |||
| Histatins | [57] | |||
| Loop peptides | Dodecapeptides Tachyplesins Protigrin-1 Bactenecin-1 Ranalexin Brevinin 1E Lactoferricin |
Disruption of bacterial membrane | [58] [59,60] [61] |
The antimicrobial activity of these peptides appears to be more rapid when compared to conventional antimicrobial drugs [41,42], which together with new drugs are faced with the challenge of AMR. In light of this, there is a likelihood of AMPs succeeding where the conventional antimicrobial agents have failed and can possibly overcome microbial resistance due to their unique target interactions. AMPs execute their antimicrobial activities through attacks on the microbial membrane and as such would most likely require restructuring in the microbial membrane to bring about resistance to AMPs [43]. The mechanism of AMP internalization is largely dependent on the molecular properties and membrane composition. Binding and interaction occurs through the electrostatic force of attraction between the negatively charged bacterial membrane and the positively charged amino acids of the AMPs, which is followed by hydrophobic interactions between the amphipathic AMP domains and the phospholipids in the microbial membrane [41]. However, challenges such as proteolytic degradation, tissue toxicity, low stability, and difficulties associated with up-scaling must be met for AMPs to be considered as potential alternative to conventional antimicrobial drugs [44]. Modified AMPs in combination with drug delivery systems can lead to the development of novel antimicrobial agents with improved therapeutic efficacy against MDR microbes [45,46].
3.1. Mechanism of Action of AMPs
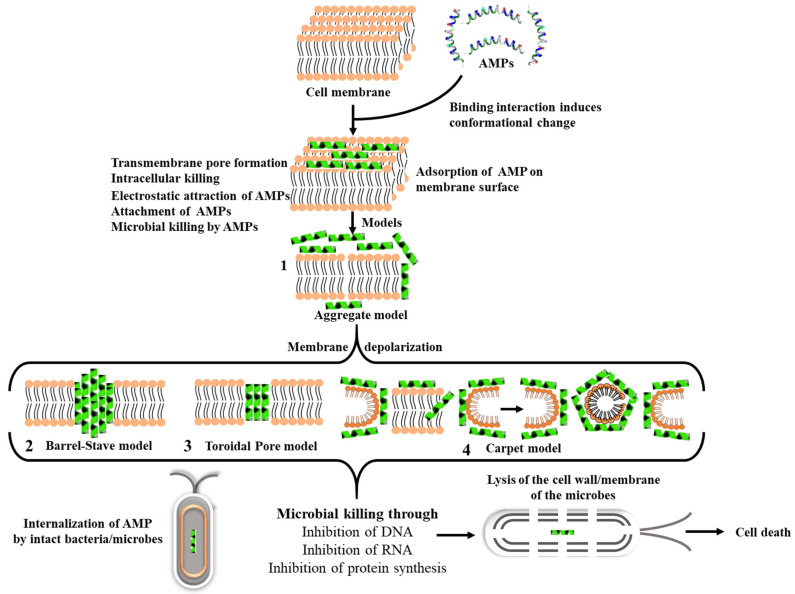
AMPs exert their antimicrobial activity on pathogenic microbes through different mechanism of actions. Their mechanisms are largely associated with the nature, structure, and sequence composition of the AMPs. Amphiphilicity, charge, and secondary structures of AMPs have all been associated with their different modes of action [62], suggesting that the mechanism by which the AMPs interact or disrupt the microbial membranes differ. As such, the way AMPs interact with microbial membranes is key not only to their antimicrobial action but also their application in therapeutics [63]. Other than membrane disruption, other mechanisms of action such as direct killing and immune modulation have been reported for AMPs [64]. The AMP’s ability to target the microbial membrane can occur in various ways, most notably through aggregation, barrel-stave, toroidal pore formation, and carpet model, as shown in Figure 3 [65]. All these mechanisms are extensively reviewed elsewhere [66,67].
Figure 3.
Mode of action used by AMPs to target and disrupt the bacterial membrane, leading to cell lysis and bacterial death.
3.2. Challenges of AMPs and the Role of Carriers for Improved Therapeutic Efficiency
Of all the AMPs identified, a limited number have made it to the clinical trials, and very few have been approved by the US Food and Drug Administration (FDA). Challenges such as systemic toxicity, proteolytic susceptibility, rapid clearance by the kidney, and short half-life have been mitigating against their implementation [68]. Chemical modification and delivery strategies have been suggested to overcome these challenges, and these have significantly improved their therapeutic index [29,69]. The focal point for this paper is to review the use of nano-based systems as drug carriers for AMPs.
3.3. Nano-Delivery Systems for AMPs
Enhancements of the properties of AMPs such as stability, toxicity, half-life, and release profile can be achieved by using delivery vehicles [70]. AMPs can be easily attached or encapsulated into delivery vehicles by covalent and non-covalent methods. This section will briefly review different vehicles or carriers for AMPs, with more emphasis on polymeric and metallic nanocarriers.
Nanotechnology-based systems through the use of nanomaterials at a size range of 1–100 nm emerged as ideal delivery systems for the AMPs. Various nanomaterials, including metallic (gold nanoparticles (AuNPs), silver nanoparticles (AgNPs), gold nanodots (Au-nanodots)) and polymeric (chitosan, poly (lactic-co-glycolic acid) or PLGA) NPs have been widely explored for the delivery of AMPs, which include but are not limited to surfactin [71], cecropin [72], and a pro-apoptotic peptide [73]. The success of nanocarriers was also demonstrated with other antimicrobial agents, which include AgNPs for ampicillin [74], silicon NPs for LL-37 [75], polymeric NPs (PLGA NPs) for colistin [76], plectasin [77], and chitosan/PGA NPs for the delivery of nisin [78], vancomycin, LL-37, cryptdin-2, and temporin B [79]. In addition, microgels, polyelectrolyte complexes, and macroscopic hydrogels have also been extensively studied for AMP delivery. These systems have been used to study the protection of antimicrobial agents against proteolytic degradation in vivo as well as to study controlled and stimuli-responsive drug release. For instance, the controlled release of Ponericin G1 (GWKDWAKKAGGWLKKKGPGMAKAALKAAMQ) was demonstrated using polyelectrolytes [80]. With the use of nanogels, the controlled release of encapsulated bradykinin in poly-2-hydroxyethyl methacrylate NPs was studied using one-pot dispersion polymerization [81]. When compared to free AMP, the encapsulated AMP showed sustained release and improved bactericidal effect against S. aureus. In another study, a hydrogel composed of nanofiber RADA16 (self-assembling peptide with the sequence: Ac-RADARADARADARADA-CONH2) was prepared in the presence of Tet213 (KRWWKWWRRC), and the antimicrobial activity of free RADA16 and RADA16-Tet213 was determined. In addition, the sustained release of Tet213 by RADA16 over the course of 28 days was also accounted for. At the end of the study, RADA16-Tet213 was effective against S. aureus. The hydrogel RADA16-Tet213 showed sustained AMP release and supported cell growth in bone mesenchymal stem cells of Sprague–Dawley rats [82]. Zetterberg et al. also investigated the use of the PEG-stabilized liposome as an AMP carrier. Protease susceptibility and the antimicrobial effects of the melittin liposome were compared to free melittin after repeated exposure to E. coli. Melittin liposomes demonstrated significant bactericidal activity upon second exposure when compared to free melittin, showing time-dependent release of AMP from the liposomes. In addition, melittin encapsulated within liposomes was totally protected against trypsin degradation [83]. Taken together, the results of these studies show that the use of carriers could facilitate the transition of AMPs from research laboratory into a clinical trial followed by widespread clinical use.
4. NPs with Antimicrobial Activity and Their Mode of Action
The search for novel antimicrobial agents has increased geometrically due to an increased incidence of microbial infections and AMR [84]. Research advancement has also led to an increased use of NPs for biomedical applications due to their broad spectrum of activities against microorganisms. In vitro studies have shown the bactericidal activity of various nanomaterials against both Gram-positive and Gram-negative bacteria [85,86,87], and similar findings were made using in vivo studies in mouse models that were infected with bacteria [88].
The antimicrobial activity of NPs has been reported for various MNPs such as zinc oxide NPs, AuNPs, and AgNPs against a number of Gram-positive and Gram-negative bacteria [89], fungi [90,91,92,93], and viruses [94,95,96]. Although most of the known polymeric NPs are commonly used as drug carriers, NPs fabricated using these polymers have been shown to possess endogenous activities. This applies to both passive (PLGA) as well as those with known antimicrobial activity (chitosan). Drugs loaded on these systems had improved biocompatibility and bio-activity. Drug-loaded PLGA-NPs exhibited higher antibacterial activity compared to the drugs alone or the unloaded NPs [97,98]. Chitosan NPs enhanced the delivery and efficacy of HIV-1 P24 protein-derived peptides [99]. Triclabendazole, which is used for the treatment of fascioliasis, is poorly soluble in water. The incorporation of this drug in chitosan NPs increased its bioavailability and stability at both low (pH 1.2) and high (pH 7.4) pH. The cytotoxic effects of the drug were significantly reduced in these nanoformulations. Thus, these nanosystems have the potential to lead to the development of orally ingested formulations for the treatment of fascioliasis [100]. This section will emphasize on MNPs with antimicrobial activity and their mechanism of inhibition.
MNPs, which include AgNPs, AuNPs, and copper oxide NPs (CuO-NPs), have all been investigated and confirmed to possess antimicrobial activity against bacteria, viruses, and fungi [101,102,103]. Although the exact mode of action of these NPs remains unclear, mechanisms such as reactive oxygen species (ROS) generation, metal ion release, and electrostatic interaction between the MNPs and the bacterial cell membrane have all been postulated. When compared to their respective salts, MNPs possess significantly higher antimicrobial activities against MDR microbes [104]. The toxicity or antimicrobial effects of AgNPs [105] and CuO-NPs [106] on microbes was size-dependent, suggesting its role in the mechanism of microbial killing. The CuO-NP size was directly proportional to their antimicrobial activity, and also small-sized monodispersed NPs showed a significant increase in antibacterial activity [106].
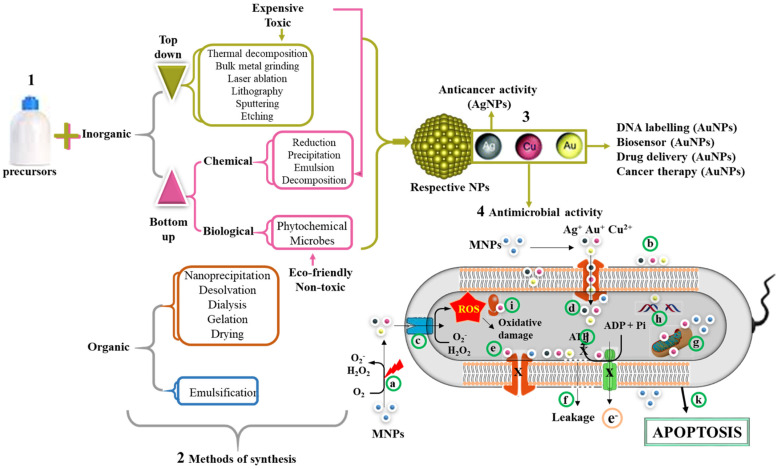
Specifically, the antimicrobial effect of AgNPs has been extensively studied against several microorganisms, and these are arguably some of the most promising MNPs used for the treatment of bacterial infections [107]. Characteristics such as shape, size distribution, stability, and charge make them one of the most widely explored MNPs in science, medicine, and physical science [108,109]. Figure 4 summarizes the mechanisms used in the synthesis of NPs, of which the chemical reduction method under the bottom–up approach is widely preferred over the top–down approaches. In recent years, green synthesis has been adopted to replace the toxic chemical reducing agents by benign natural resources such as microorganisms and plant extracts [110]. This method is safer and eco-friendly when compared to physical and chemical methods of synthesis. The plant-mediated synthesis is economical and make use of readily available and renewable plant materials such leaves, stems, roots, etc. Moreover, synthesis occurs in just one step, since the phytochemicals are able to act as reducing, capping, and stabilizing agents [110,111,112]. Previous studies have confirmed the safety of the biogenic method for MNP synthesis with effective antimicrobial activities against MDR bacteria [112,113].
Figure 4.
Synthesis of organic and inorganic NPs and their antimicrobial mechanism. MNPs can be synthesized using either a bottom–up or top–down approach (1). The methods of NP synthesis are broadly classified into physical, chemical, and biological methods (2). The reduction of metallic salts by these methods leads to the formation of MNPs. Examples of MNPs include AgNPs, AuNPs, and CuONPs (3). Various applications of MNPs include DNA labeling, biosensor, drug delivery, anticancer, and antimicrobial properties (4). The possible mechanism of their antimicrobial activity involves release of metallic ion when excited by laser (a) or in the presence of oxygen. The size of MNPs exhibited electronic effects and thus improved surface attraction (b). These metallic radicals in addition to their penetrative ability can easily pass through membrane channels (c). The MNPs triggers the generation of ROS inside the cells, which in turn leads to cellular damage. In addition, MNPs inhibit the channel transport of solutes/ions (e). The accumulation of these metallic ions leads to membrane depolarization and ultimately membrane leakage of cellular contents (f), mitochondrial dysfunction (g), DNA damage (h), ribosome disassembly (i), and inhibition of the electron transport chain and ATP synthesis (j). Collectively, all these processes can lead to cell death through apoptosis (k).
The antimicrobial property of Acacia rigidula biosynthesized AgNPs was demonstrated against Gram-positive and Gram-negative MDR bacteria. The effect of the AgNPs was evaluated against E. coli, P. aeruginosa, and Bacillus subtilis. Their safety profile was monitored in a murine skin infection model. The outcome of this study suggested that the AgNPs are compatible for use as a therapeutic agent against infectious diseases associated with drug resistant and drug susceptible bacterial strains [98,114]. Due to their larger surface area, the surface of the MNPs can be modified to assign a specific activity by changing their surface composition. Lopez-Abarrategui et al. demonstrated that modification of citrate-coated MnFe2O4-NPs with antifungal peptide (Cm-p5) enhanced their antifungal activity in comparison to the free peptide and unmodified NPs. Cm-p5-MnFe2O4 completely inhibited Candida albicans (C. albicans) growth with a MIC of 100 µg/mL, compared to that of citrate-coated MnFe2O4-NPs at 250 µg/mL. The NPs showed no antibacterial activities against S. aureus and E. coli [115]. Therefore, MNPs could serve as excellent drug carriers due to their low cytotoxicity, ease of preparation, the ability to modify their surface, and good stability.
5. Nanocarriers of AMPs
NP–protein interaction possesses crucial application in biomedicine such as delivery systems and theranostic agents [116]. However, the mechanism of recognition, specificity, and selectivity are poorly understood and remain a challenge. This section discusses the nanomaterials capable of transporting AMPs into the site of action in order to overcome their afore-mentioned limitations and exact the expected therapeutic effect.
Studies have reported specific microbial resistance and their mechanisms against AMPs [117,118,119,120,121,122,123]. Pathogens can rapidly evolve and confer resistance to AMPs in vitro [124]. Resistance evolution by Baydaa et al. was arguably the first study to explore the pharmacodynamic and bacterial AMP resistance. The study showed that AMP resistance in S. aureus and some strains resulted not only in increased MICs but also an altered Hill coefficient (κ), resulting in steeper pharmacodynamic curves [125]. Although AMR is also observed in AMPs, MDR against AMPs is not as prevalent when compared to antibiotics [126]. Several approaches have been studied to improve the therapeutic use of AMPs. These include the combination of AMPs with traditional antibiotics since both have shown synergistic effect in the reduction of microbial resistance. Another method is the use of nanocarriers, which have been shown to reduce other side effects while exacting maximum suicidal activity against microbial populations [127].
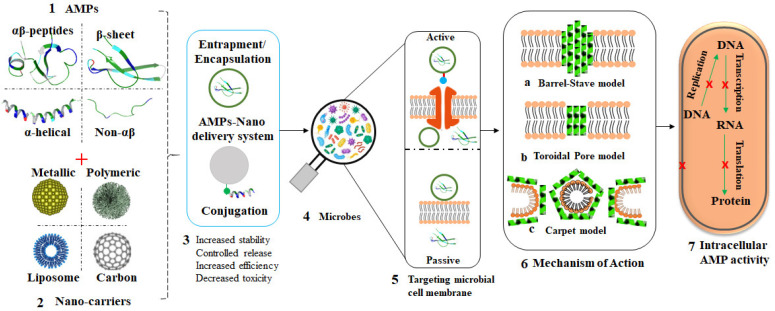
In a bid to prevent AMR or bypass drug resistance, AMPs are now being considered as potential alternative for antibiotics. Nanomaterials, especially polymeric and MNPs, provide one of the promising drug delivery systems, as highlighted in Figure 5 [128,129,130]. Aside from the fact that some nanomaterials possess antimicrobial activities and can inhibit the growth of microbes through several mechanisms, they can also act as carriers for either antibiotics or AMPs to overcome the defense mechanisms of microbes and further enhance antimicrobial effects [131]. These materials can be functionalized with antimicrobial agents to prevent and treat microbial infections as well as to improve the effectiveness of the conventional drugs [132].
Figure 5.
Overview of AMP-loaded nanocarriers and their mode of action. Structurally, AMPs are classified into four groups (1); different nanocarriers have been studied as effective carries of AMPs (2); different AMP nanoformulations can be obtained through various chemistries between peptides and nanocarriers (3); exposure of microbes to these AMP nanoformulations (4); via passive or active transportation (5); leading to bacterial membrane attack by the AMPs through various AMP-dependent mechanisms (6) and ultimately bacterial death (7).
Nanomaterials such as MNPs (AuNPs, AgNPs), polymeric NPs, dendrimers, liposomes, micelles, and carbon nanotubes have all been used as carriers or transporters of drugs [133,134,135,136]. The function of these carriers is to decrease side effects, lower drug dosage, maintain constant drug levels in the blood, maximize therapeutic index, and reduce drug degradation and undesirable side effects [137]. MNPs display striking different size and shape-dependent properties when compared to their bulk material. These properties include a wide surface plasmon resonance (SPR) band, which is directly correlated to particle size, a large surface to volume ratio, biocompatibility, and low toxicity [138,139]. The MNPs’ parameters such as charge, size, and surface composition have all been reported to influence the activity of the NPs [140,141,142]. MNPs have been explored and utilized in several biomedical applications such as therapy, drug and gene delivery, probes, sensors, diagnostics, and photocatalyst.
MNPs have been shown to improve the antimicrobial activity of drugs by providing support and synergistic effects against pathogenic microbes. Specifically, MNPs have incredible physicochemical properties and have shown novel bioactivities, which can be enhanced by attaching bioactive molecules [143]. The functionalization of MNPs is achieved by conjugating different molecules through different mechanisms. MNPs employed in biological applications are usually modified with biocompatible polymers such as polyethylene glycol (PEG), proteins/peptides (e.g., bovine serum albumin), and oligonucleotides. The process of linking molecules to the surface of the MNPs can be achieved by physisorption or by taking advantage of the metal’s affinity for the sulfhydryl group of thiolated molecules. Electrostatic interactions and non-covalent conjugation can also be used to functionalize the MNPs with molecules that contain reactive groups such as hydroxyl, carboxylic group, and amine groups, which can be used to attach other biologically active molecules.
The biologically active moieties can include small drug molecules [144] or proteins [145], while targeted delivery/therapy can be achieved by conjugating target specific aptamers [146], antigen/antibodies [147], or peptides [148]. Different conjugation chemistries have been studied for effective drug loading and delivery. Examples of these chemistries include the conjugation of RR-11a endopeptidase to liposomes using the amine/carboxyl chemistry [149], the polyclonal Rabbit IgG conjugated to AuNPs using the amine/carboxylate chemistry [150], S2P peptide (CRTLTVRKC) conjugated to chitosan NPs through the amine/carboxylate, thiol/maleimide chemistry [151], liposome functionalized with adiponectin, globular domain by thiol/maleimide chemistry [152], and the J18 RNA aptamer conjugated to AuNPs by the base-pairing hybridization [153]. Immobilization of the biomolecules/AMPs on the NPs can occur in two ways; i.e., the AMPs are either temporarily (reversible) or permanently (irreversible) tethered on or within the NPs. The reversible chemistry ensures targeted delivery and release of the AMPs in its native form, and their functions are independent of the nanocarriers. The reversible nanosystems are usually responsive to biological stimuli, where a change in pH or presence of an analyte will trigger release of the attached AMPs. In the irreversible conjugation, the AMPs are permanently attached on the nanoparticle and act in synergy with the NPs [154,155,156].
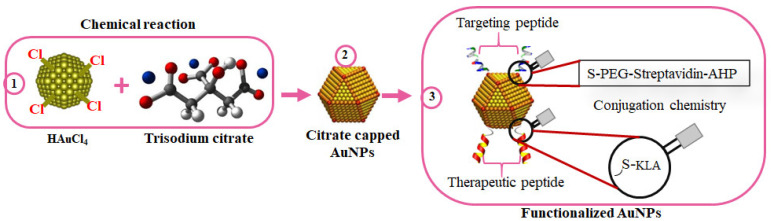
Several nanoconjugates with biologically active molecules have shown promising outcomes for the treatment of some diseases. In a comprehensive study by Sibuyi et al., AuNPs were used in the development of targeted nanotherapy for cancer. The AuNPs were bifunctionalized with adipose homing (targeting) moiety and an AMP (DKLAKKLAK2/KLA) with proapoptotic activity for the selective induction of apoptosis in target cells (Figure 6). The homing peptide was conjugated to the AuNPs through an irreversible chemistry, while the KLA used a reversible chemistry where a caspase-3 cleavage site (DEVD) was used as a linker between the AuNPs and KLA. Upon internalization into the cells, the KLA is detached from the AuNPs by caspase-3 and triggers cell death through apoptosis [157]. Similarly, KLA-loaded liposomes with the adipose homing peptide accumulated in the white adipose tissues (WATs), resulting in body weight loss in obese mice models, as shown by Hossen, et al. [158]. The bifunctionalized NPs showed potential of repurposing AMPs for different applications by selectively targeted the cells that express the receptor for the targeting peptide, i.e., the colon cancer cells [157] and the endothelial cells in the WATs of obese mice [129].
Figure 6.
Surface chemistry of bifunctionalized citrate-capped AuNPs. AuNPs were chemically synthesized using trisodium citrate as the reducing agent (1); to form citrate-capped AuNPs (2); while exploring the AuNP-thiol affinity for the attachment of the targeting and therapeutic peptides (3).
5.1. Antimicrobial Activity of AuNPs
AuNPs, in fact most MNPs, can have dual functions by serving as both drug transporters and antimicrobial agents. Although the bactericidal effects of AuNPs were reported against MDR Gram-negative bacteria [159], AuNPs are preferably used as carriers. The therapeutic effects of AuNPs are attributed to their size, shape, and surface composition. The size and shape can be manipulated during synthesis by varying the ratios between metal precursor and reducing agents; while the surface composition can be modified by attaching biomolecules through electrostatic interaction and covalent conjugation. Targeting moieties such as antibodies are attached for target specificity purposes [160,161,162]. Anti-17β-estradiol immobilization on AuNPs achieved ultrasensitive detection of 17β-estradiol [163]. Listeria monocytogenes polyclonal antibodies were also conjugated to AuNPs to improve the stability of the secondary structure and the efficacy of the antibody [164].
Due to their optical properties, AuNPs can absorb light and convert it to thermal energy, and it is often used in photothermal therapy. Pedrosa et al. reported the therapeutic effect of metallic compound (TS265) functionalized AuNPs in doxorubicin-resistant cancer cells. The combination therapy resulted in 65% cancer cell death after laser treatment, and showed no cytotoxicity toward the normal cells [165]. A AuNP-based photothermal effect was also used against pathogens. Accordingly, gold nanorods (AuNRs) functionalized with either PEG (hydrophilic PEG-AuNRs) or polystyrene (hydrophobic PS-AuNRs) showed minimal antibacterial activities against S. aureus and Propionibacterium acnes (P. acnes) (≤85% reduction). Laser treatment enhanced the antimicrobial property of the AuNRs, resulting in ≥99.9% reduction in bacteria numbers [166]. As a result of the AuNPs’ larger surface area, multiple molecules can be attached on their surface through covalent and non-covalent conjugations. This property, together with its superior biocompatibility and targeting ability, make AuNPs a promising delivery vehicle for various biological applications. AuNPs have been used in combination with chemotherapeutic drugs such as methotrexate [167], doxorubicin [168], and oxaliplatin [169] to improve drug efficacy, delivery, stability, and to ensure a rapid increase in intracellular drug concentration [170].
The use of AuNPs, among other nanomaterials, in the regulation of the immune system by developing prophylactic and therapeutic vaccines are currently considered a promising biomedical application. Based on AuNPs’ parameters, they can be used as immunogens or as a vehicle for adjuvants and antigens [171,172] The size of AuNPs influences their uptake and internalization by cells, 3 nm AuNPs were taken up by cells through pinocytosis and were nontoxic, nonimmunogenic, and repressed ROS generation [173]. The uptake of 60 nm AuNP by cells were through endocytosis. These NPs were also nontoxic and repressed cellular responses induced by interleukin 1 beta (IL-1β) [174].
To investigate the role of drug-conjugated AuNPs, AuNPs were synthesized by the green method using sophorolipid (SL) as a reducing and stabilizing agent [175]. The antimicrobial activity of AuNPs-SL against S. aureus and Vibrio cholerae (V. cholerae) was compared to AuNPs and SL. The results indicated that SL alone exhibited antimicrobial activity against S. aureus but not V. cholerae. The AuNP-SL inhibited the growths of the microorganisms more effectively, while the AuNPs showed no activity [164]. The synergistic effect of AuNPs-SL used in combination with three antibiotics—namely, ampicillin, kanamycin, and polymyxin—were further studied against S. aureus and V. cholerae. The combined treatments had higher efficacy than the individual test agents, and the activity was highest for ampicillin followed by polymyxin. The differences in their activity could be attributed to the mechanism of the drugs, since both antibiotics interact and disrupt cell wall synthesis. Kanamycin, on the other hand, induces cell death by interfering with protein synthesis [164]. Additionally, the activity of the nanoconstructs can be channeled by attaching targeting moieties onto the NPs and achieve target specific delivery.
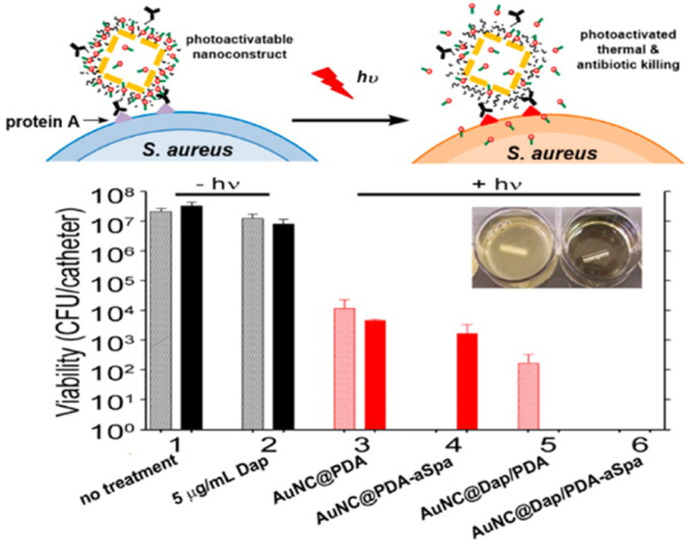
Incorporating antibodies that target staphylococcal protein A (aSpa) into polydopamine (PDA)-coated Au nanocages conjugated with daptomycin (Dap) improved targeting and selectivity. As shown in Figure 7, there was a complete eradication of bacteria treated with AuNP@Dap conjugates post 24 h laser exposure. Interestingly, the effects of AuNP@Dap/PDA-aSpa were immediate and persistent from 0 to 24 h, while the untargeted AuNP@Dap/PDA had a delayed effect, indicating that targeting also plays a crucial role by facilitating NP uptake and accumulation at the target site. The fact that ~50% of bacteria were viable after exposure to AuNP@PDA constructs (with and without aSpa) as a result of photothermal effects of the Au nanocages suggests that the additive effects observed with the AuNP@Dap conjugates are caused by Dap. The Au nanocages served as both carriers and photothermal agents, resulting in synergistic antibacterial effects. The AuNPs enhanced the potency of antibiotics conjugated to the nanocarriers and proved to be effective therapy against intrinsically resistant biofilm infections by MDR pathogens [176]. Therefore, the studies confirmed the effectiveness of AuNPs as nanocarriers for antimicrobial agents and that they have the potential to improve the clinical efficacy of drugs even at lower doses and prevent early drug clearance and degradation. The mechanisms by which AuNPs exert their antimicrobial activity include inhibition of transcription and energy metabolism. Simply put, AuNPs inhibit the ATPase function, which leads to ATP reduction and collapse of the membrane potential. The other mechanism is the inhibition of protein synthesis by binding to the ribosome subunit, preventing tRNA from binding [177].
Figure 7.
Synergistic effect of laser-induced and controlled release of Dap in S. aureus. Reprinted with permission from Copyright © 2016 American Chemical Society: https://pubs.acs.org/doi/full/10.1021/acsinfecdis.5b00117, accessed on 20 May 2021, further permissions related to the material excerpted should be directed to the ACS.
5.2. AgNPs as Potent Antimicrobial Agents
AgNPs have been extensively studied and used commercially as antimicrobial agents. Synergistic effects have been achieved when AgNPs were used together with other antimicrobial agents. The NP conjugates are more potent and have the potential to kill MDR bacteria. The efficacy of Andersonin-Y1 against K. pneumoniae was improved after conjugation to AgNPs. The MIC of the AMP-tagged AgNPs was in the range of 5–15 µM when compared to the AMPs, which had a MIC of 50 µM. Further insights into their antibacterial mechanism by nuclear magnetic resonance spectroscopy and molecular dynamic simulation suggested killing by membrane pore formation through the hydrophobic collapse mechanism, thus proposing nanocarriers as potential antibiotic substitutes [130].
AgNPs have been functionalized with different molecules that have antimicrobial activity in order to achieve synergistic antimicrobial effects [178,179,180,181]. These results were further validated against E coli. Morphological changes, cellular uptake, and the mechanism of antimicrobial activity were determined by scanning electron microscope, transmission electron microscopes, and the lactate dehydrogenase assay (LDH), respectively. When compared to the control, the treated cells exhibited irregular shape and size, cell lysis, and cell membrane disruption, suggesting that the NPs caused the cells to rupture. In addition, the LDH assay indicated reduced cellular activities in both bacterial strains, suggesting the inhibition of the electron transport chain. This result was in agreement with the previously proposed mechanism of antimicrobial activity of nanomaterials [182].
To bypass the limitations of physical and chemical methods of MNPs synthesis, green synthesis has been explored to produce biogenic NPs that are biocompatible. Plant-mediated MNPs are synthesized using extracts from medicinal plants that are used in disease treatments. Generally, AgNPs often have more pronounced antimicrobial properties [183,184] when compared to AuNPs [185]. For example, biogenic AgNPs from Salvia africana-lutea (SAL) and Sutherlandia frutescens (SF) exhibited significant antibacterial activity against Staphylococcus epidermidis (S. epidermidis) and P. aeruginosa. The MIC values for SAL-AgNPs was <0.75 mg/mL. However, the MIC values of the extract was >50 mg/mL for both strains, suggesting that the NPs have enhanced antibacterial activities [183]. The activity of the biogenic MNPs can be further improved by co-treatment or biofunctionalization with other antimicrobial agents [115]. The progression of nano-based drugs into clinical trials provides evidence that nanomaterials can serve as good alternative strategies for the treatment of infectious diseases.
5.3. Nanohybrids for Enhanced Biocompatibility and Efficacy
The combination of two or more nanomaterials (nanohybrids) with different physico-chemical properties is becoming popular, as nanohybrids have been shown to have improved pharmacokinetics and synergistic bioactivities. Several nanohybrid compositions developed from inorganic/inorganic or inorganic/organic nanocomposites have been explored for biomedical applications. Among inorganic NPs, AgNPs are widely explored as broad-spectrum antimicrobial agents and are used in certified consumer products. In an attempt to prolong their activity and enhance their stability, inorganic/inorganic nanocomposites were created from various metal precursors. Silver has been complexed with various other metals such as copper oxide [186], gold [187], platinum [188], etc., to produce AgNP nanohybrids with synergistic or unique properties that are superior to AgNPs alone. The nanohybrids improved the efficacy of antimicrobial agents [189] when used as carriers or in combination therapy. In an interesting study by Kazemzadeh-Narbat et al., titanium oxide nanotube implants consisting of multiple layers of thin films were synthesized and used for the time-dependent release of an AMP (HHC-36/KRWWKWWRR-NH2). This was done by encapsulating the AMP with calcium phosphate and titania nanotube. The study demonstrated the therapeutic efficacy of the nanotubes against S. aureus and P. aeruginosa through the controlled release of AMP [190].
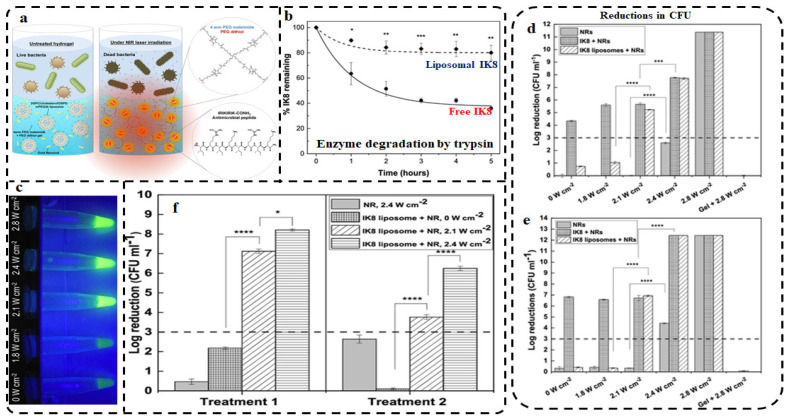
Similarly, the inorganic/organic nanocomposites present a benign strategy that can be used to encapsulate the inorganic nanomaterial within the organic nanomaterial, which may render the nanocomposite more bio-friendly, increasing its bioavailability and safety profiles. Liposomes and chitosan NPs are some of the bioinert and biodegradable nanomaterials used to avoid the proteolytic degradation of drugs. Moorcroft et al. created AuNR/AMP-liposome loaded hydrogels that are responsive to laser irradiation due to the presence of AuNRs. The study demonstrates the controlled release of AMP liposomes in response to laser irradiation (Figure 8a). IK8 (IRIKIRIK-CONH2), which is the AMP used in this study, was shown to be potent against the test microorganisms (P. aeruginosa and S. aureus). IK8 is susceptible to proteolytic enzyme degradation, leading to loss of the antimicrobial activity. However, the entrapment of IK8 inside liposomes protected the AMP from trypsin-induced degradation for up to 5 h (Figure 8b), and further encapsulation with AuNR in hydrogel allowed for the stimuli-responsive release of the AMPs. Thus, the AuNR/AMP liposome-loaded hydrogel hybrids enhanced the AMP stability, protected it from degradation, facilitated the controlled release of AMP, and facilitated the retention of the nanomaterials within the hydrogel. The AuNR/AMP liposome-loaded hydrogel showed dual and synergistic effects, resulting from the antibacterial effects of IK8 and the photothermal activity of the AuNRs. A single dose was used in two cycles of treatment [191].
Figure 8.
Controlled release and thermal enhancement of IK8-conjugated AuNR-loaded hydrogel. Mechanism of IK8 release triggered by laser irradiation of AuNRs (a); proteolytic effect of trypsin on IK8 and liposome IK8 over the course of 5 h, * p ≤ 0.05, ** p ≤ 0.01, *** p ≤ 0.001 (b); irradiation of the AuNR-loaded hydrogels at 850 nm with ranging intensities from 0 to 2.8 W/cm2 (c); Antibacterial effect of laser-irradiated hydrogels against S. aureus, *** p ≤ 0.001, **** p ≤ 0.0001 (d); and P. aeruginosa, **** p ≤ 0.0001 (e); and activity of different formulations on S. aureus over two treatment cycles after 5 (treatment 1) and 10 (treatment 2) min laser irradiation, * p ≤ 0.05, **** p ≤ 0.0001 (f). Reprinted with permission: Copyright © 2020 American Chemical Society: https://pubs.acs.org/doi/10.1021/acsami.9b22587, accessed on 20 May 2021, further permissions related to the material excerpted should be directed to the ACS [191].
In an independent study, a similar strategy was used to develop pH-sensitive AuNP-liposome-loaded hydrogels. Carboxyl-modified AuNPs (AuC) were used to stabilize cationic liposomes (AuC-liposomes), which were then loaded into a polyacrylamide hydrogel, and the AuC-liposomes release profile was studied in S. aureus at pH 7.4 and 4.5. Higher uptake was observed at pH 4.5, suggesting that AUC-liposomes are possibly released from the hydrogels at pH 7.4 and the AuC detaches from the liposomes at acidic pH below the pKa of the carboxylic group. This proved that these systems can be used to deliver antimicrobial agents for the treatment of bacterial infections. In vivo studies showed that the formulation was also biocompatible and showed no toxicity when topically administered daily on a skin of mice for 7 days [192].
6. Nanocarriers in Clinical Trials
Several functional nanomaterials that has been investigated in preclinical and clinical studies led to the development of nanomedicines that are currently on the market, most notably some clinically approved liposome drug formulations and metallic imaging agents [193]. Most of the nanocarriers in preclinical and clinical trials as well in clinical use are for cancer targeting [194]. Lipid and polymeric nanocarriers are the most widely used, and examples of these carriers in clinical trials include LiPlaCis, a lipid-based nano-formulation with cisplatin currently in phase II for refractory solid tumors and NK105 conjugated with paclitaxel currently in phase II for the treatment of gastric cancer [195]. Moreover, the utilization of nanotechnology-based treatments for diseases other than cancer has increased exponentially in recent years. Antimicrobial therapy is another pivotal clinical focus that is being investigated for the advancement of nanomedicine. MNPs can serve as carriers and also as potential antimicrobial agents. Specifically, AgNPs can diffuse through the microbial cell membrane, leading to toxicity through Ag+ release. This nanomaterial has been extensively studied and used in medicine for decades. Due to their activities, AgNPs with antimicrobial properties were approved by the FDA for wound therapy [196,197]. Currently, there are nine ongoing clinical trials investigating AgNPs as an antibacterial therapy in different diseases.
AMPs have been ascertained as outstanding alternative antimicrobial agents to overcome AMR, and a growing body of evidence suggests that AMPs are swiftly gaining more attention for their potential clinical application as they present remarkable benefits over conventional antibiotics. AMPs are uniquely implicated in all life forms and display significant roles in the innate immune system. Some AMPs are evolutionarily conserved, which could be an indication that these molecules may be capable of restricting tendencies for the development of microbial resistance.
Despite the therapeutic benefits of AMPs, only a few AMP-based formulations have successfully progressed into clinical trials. [198]. AMPs in clinical trial are grouped based on their mechanism of action on cell membrane of microbes (Temporin10a [199], Ruminococcin C [200]), immune system (IDR-1002 [201]), and intracellular functions (HB-107 [202], Buforin II [203]. Specific modifications to AMPs can be used to improve their delivery, biological activity, and stability as well as reduce toxicity. Advances in nanotechnology for drug delivery have been extensively reviewed. The successful application of nanotechnology to develop improved drug delivery systems has been demonstrated successfully, and therefore, its application for the delivery of AMPs is also attainable and can lead to the development of novel antimicrobial agents that can fight MDR.
It is further crucial to investigate the fundamental biological effect, biodistribution, and pharmacokinetics of MNPs, most especially silver and other nano-based nanocarrier for clinical applications.
Merits and Limitations of Nanocarriers
As described above, NPs have been involved in different applications in the field of biomedicine and have proven to be effective drug delivery vehicles and a potential alternative antimicrobial agent. Nanomaterials such as MNPs, liposomes, dendrimers, polymeric, and carbon nanotubes have been widely implicated in the design of AMPs with enhanced activity toward MDR microorganisms. However, drawbacks such as cytotoxicity, conjugation protocols, stability profiles, and shelf-life have been reported for the AMPs. Table 2 shows some of the nanocarriers that were used as vehicles for AMPs, together with their limitations. Carbon nanotube synthesis is costly, while these NPs have poor solubility. Liposomes have low loading capacity and could also induce immune response. While liposomes are biodegradable and both hydrophobic and hydrophilic drugs can be loaded, factors such as drug-loading efficiency and immunogenicity remain a challenge. Dendrimers are monodispersed molecules with a high control over the critical molecular design parameter. However, the cost of synthesis together with its non-specificity remain major limiting factors for dendrimers. Studies have reported the biocompatibility and nature-dependent biodegradability of polymeric NPs. This class of carriers is easy to modify and to control drug release. Its disadvantages include low cell affinity and the toxicity of their by-products.
Table 2.
Advantages and limitations of various nanocarriers.
| Nanocarriers | Advantages | Limitations |
|---|---|---|
| MNPs | Multipurpose High surface to volume ratio |
Cytotoxicity Shelf-life Solubility |
| Liposomes | Biodegradable Hydrophobic and hydrophilic molecules can be loaded |
Loading efficiency Immunogenicity |
| Dendrimers | High control over the critical molecular design parameter | High cost of synthesis Non-specific toxicity |
| Carbon nanotubes | Soluble in water Multiple application |
High cost of synthesis Less degradable |
| Polymeric NPs | Easy modification Biocompatibility Nature-dependent biodegradability Time-dependent drug release. |
Low cell affinity toxicity of byproducts. |
7. Conclusions and Future Perspectives
As a challenge, microbes evolve geometrically faster than the discovery and implementation of antibiotics. Moreover, these antibiotics gradually lost their antimicrobial activity and drug-resistant bacteria appeared with antibiotics overuse and even the misuse of several other antibiotics. Although, AMPs have been thought as an alternative to antibiotics, there is still a pool of antibiotics in AMPs undiscovered and poor pharmacokinetics of peptide drugs is a disadvantage to the application of existing AMPs.
The use of AMP delivery systems can facilitate their progress into clinical trials and ultimately be fundamental for their implementation.
Antibiotics exert their action by targeting specific cellular components or metabolic intermediates, resulting in microbial growth inhibition or microbial death. The most common mechanism is through the inhibition of vital enzymes involved in microbial growth and metabolism. This could result in genetic mutations, leading to microbial drug resistance. Conversely, AMPs due to their amphipathic properties bind to the membrane bilayer of the negatively charged bacteria, which allows rapid penetration. This process is not affected by mutations. More so, AMPs carry out their antimicrobial activity on the entire cellular membrane, making it burdensome for microbes to develop resistance against them. AMPs capable of immune regulation make their mode of action comprehensive. Antibiotics undergo detoxification, and their complete renal clearance is not guaranteed. However, the metabolic degradation of AMPs involves monomeric amino acids that can be channeled to essential biosynthetic pathways.
Due to the continuous resistance development against current antibiotics and antimicrobial agents, novel therapeutics are urgently needed. AMPs have emerged as alternatives due to the mechanism by which they cause physical disruption of the phospholipid bilayer of the pathogens, resulting in their death. The metabolic requirement of membrane repair limits the risk of drug resistance against these peptides. Nonetheless, instability and proteolytic degradation among others have been correlated with AMPs and have in turn limited their implementation. Drug delivery systems are proposed as the right approach toward their uptake, release (sustained, controlled, and triggered), and protection against proteases in order to overcome MDR challenges. Nanomaterials are now being used as drug delivery systems to improve therapeutic activity and reduce undesirable side effects. With the broad therapeutic involvement of NPs, it is worth establishing the mechanism by which their conjugation, functionalization, encapsulation, and complexes can influence bacterial population. Nanocarriers such as MNPs, polymeric, and liposomes among others have all been reported as effective drug carriers with good therapeutic indexes.
Of specific benefit, NPs are able to target infection sites. As such, the synergistic activity of the nanocarriers together with AMPs in cell wall penetration, particle aggregation, ROS formation, and inhibition of cellular activities are extremely important in fighting pathogens and MDR infections. Although challenges of AMP-carrier systems include finding an appropriate carrier, entrapment efficiency, and conjugation chemistry; there are numerous ongoing research studies for AMP-nanocarriers optimization. When compared with the number of AMP complexed with nanocarriers in clinical trials, basic studies with in vitro end points are geometrically greater. With ever-expanding AMP discovery, more in vivo studies are required to understand the physiological barriers and immunological responses in order to simplify the challenges in clinical trials. Nanotechnology is capable of revolutionizing the world of medicine, and AMP nanocarriers are worth the investment in order to tackle MDR pathogens.
Acknowledgments
DSI/Mintek NIC, Biolabels Node, University of the Western Cape, South Africa.
Author Contributions
All authors made significant contributions to the submission of the article. A.O.F., A.M.M. and M.M. conceived the concept and the design of the review; A.O.F. and N.R.S.S., prepared the initial draft and also substantially revised the manuscript; A.M.M. and M.M. thoroughly revised the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the DSI/Mintek NIC, Biolabels Node, University of the Western Cape, South Africa.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Wall S. Prevention of antibiotic resistance—An epidemiological scoping review to identify research categories and knowledge gaps. Glob. Health Action. 2019;12:1756191. doi: 10.1080/16549716.2020.1756191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Sengupta S., Chattopadhyay M.K., Grossart H.-P. The multifaceted roles of antibiotics and antibiotic resistance in nature. Front. Microbiol. 2013;4:47. doi: 10.3389/fmicb.2013.00047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Spellberg B., Gilbert D.N. The future of antibiotics and resistance: A tribute to a career of leadership by John Bartlett. Clin. Infect. Dis. 2014;59:S71–S75. doi: 10.1093/cid/ciu392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Centers for Disease Control and Prevention . Antibiotic Resistance Threats in the United States. CDC; Atlanta, GA, USA: 2013. Office of Infectious Disease; pp. 1–114. [Google Scholar]
- 5.Ventola C.L. The antibiotic resistance crisis: Part 1: Causes and threats. Pharm. Ther. 2015;40:277. [PMC free article] [PubMed] [Google Scholar]
- 6.Smith R.A., M’ikanatha N.M., Read A.F. Antibiotic resistance: A primer and call to action. Health Commun. 2015;30:309–314. doi: 10.1080/10410236.2014.943634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Jasovský D., Littmann J., Zorzet A., Cars O. Antimicrobial resistance—A threat to the world’s sustainable development. Upsala J. Med. Sci. 2016;121:159–164. doi: 10.1080/03009734.2016.1195900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Prestinaci F., Pezzotti P., Pantosti A. Antimicrobial resistance: A global multifaceted phenomenon. Pathog. Glob. Health. 2015;109:309–318. doi: 10.1179/2047773215Y.0000000030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Davies J., Davies D. Origins and evolution of antibiotic resistance. Microbiol. Mol. Biol. Rev. 2010;74:417–433. doi: 10.1128/MMBR.00016-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Nuti R., Goud N.S., Saraswati A.P., Alvala R., Alvala M. Antimicrobial peptides: A promising therapeutic strategy in tackling antimicrobial resistance. Curr. Med. Chem. 2017;24:4303–4314. doi: 10.2174/0929867324666170815102441. [DOI] [PubMed] [Google Scholar]
- 11.Pirtskhalava M., Amstrong A.A., Grigolava M., Chubinidze M., Alimbarashvili E., Vishnepolsky B., Gabrielian A., Rosenthal A., Hurt D.E., Tartakovsky M. DBAASP v3: Database of antimicrobial/cytotoxic activity and structure of peptides as a resource for development of new therapeutics. Nucleic Acids Res. 2021;49:D288–D297. doi: 10.1093/nar/gkaa991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wang Z., Wang G. APD: The Antimicrobial Peptide Database. Nucleic Acids Res. 2004;32:D590–D592. doi: 10.1093/nar/gkh025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Jhong J.-H., Chi Y.-H., Li W.-C., Lin T.-H., Huang K.-Y., Lee T.-Y. dbAMP: An integrated resource for exploring antimicrobial peptides with functional activities and physicochemical properties on transcriptome and proteome data. Nucleic Acids Res. 2018;47:D285–D297. doi: 10.1093/nar/gky1030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wang G., Li X., Wang Z. APD3: The antimicrobial peptide database as a tool for research and education. Nucleic Acids Res. 2016;44:D1087–D1093. doi: 10.1093/nar/gkv1278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lei J., Sun L., Huang S., Zhu C., Li P., He J., Mackey V., Coy D.H., He Q. The antimicrobial peptides and their potential clinical applications. Am. J. Transl. Res. 2019;11:3919–3931. [PMC free article] [PubMed] [Google Scholar]
- 16.Mulder K., Lima L.A., Miranda V., Dias S.C., Franco O.L. Current scenario of peptide-based drugs: The key roles of cationic antitumor and antiviral peptides. Front. Microbiol. 2013;4:321. doi: 10.3389/fmicb.2013.00321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Jiang Y., Yang D., Li W., Wang B., Jiang Z., Li M. Antiviral activity of recombinant mouse β-defensin 3 against influenza A virus in vitro and in vivo. Antivir. Chem. Chemother. 2012;22:255–262. doi: 10.3851/IMP2077. [DOI] [PubMed] [Google Scholar]
- 18.Pachón-Ibáñez M.E., Smani Y., Pachón J., Sánchez-Céspedes J. Perspectives for clinical use of engineered human host defense antimicrobial peptides. FEMS Microbiol. Rev. 2017;41:323–342. doi: 10.1093/femsre/fux012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Dürr U.H., Sudheendra U., Ramamoorthy A. LL-37, the only human member of the cathelicidin family of antimicrobial peptides. Biochim. Biophys. Acta (BBA)—Biomembr. 2006;1758:1408–1425. doi: 10.1016/j.bbamem.2006.03.030. [DOI] [PubMed] [Google Scholar]
- 20.Méndez-Samperio P. The human cathelicidin hCAP18/LL-37: A multifunctional peptide involved in mycobacterial infections. Peptides. 2010;31:1791–1798. doi: 10.1016/j.peptides.2010.06.016. [DOI] [PubMed] [Google Scholar]
- 21.Kanthawong S., Bolscher J.G., Veerman E.C., van Marle J., de Soet H.J., Nazmi K., Wongratanacheewin S., Taweechaisupapong S. Antimicrobial and antibiofilm activity of LL-37 and its truncated variants against Burkholderia pseudomallei. Int. J. Antimicrob. Agents. 2012;39:39–44. doi: 10.1016/j.ijantimicag.2011.09.010. [DOI] [PubMed] [Google Scholar]
- 22.Vandamme D., Landuyt B., Luyten W., Schoofs L. A comprehensive summary of LL-37, the factotum human cathelicidin peptide. Cell. Immunol. 2012;280:22–35. doi: 10.1016/j.cellimm.2012.11.009. [DOI] [PubMed] [Google Scholar]
- 23.Sørensen O.E., Cowland J.B., Theilgaard-Mönch K., Liu L., Ganz T., Borregaard N. Wound healing and expression of antimicrobial peptides/polypeptides in human keratinocytes, a consequence of common growth factors. J. Immunol. 2003;170:5583–5589. doi: 10.4049/jimmunol.170.11.5583. [DOI] [PubMed] [Google Scholar]
- 24.Grossman P., Tiefenthaler-Gilmer U., Raysz A., Kesper U. Mindfulness training as an intervention for fibromyalgia: Evidence of postintervention and 3-year follow-up benefits in well-being. Psychother. Psychosom. 2007;76:226–233. doi: 10.1159/000101501. [DOI] [PubMed] [Google Scholar]
- 25.Chamorro C.I., Weber G., Grönberg A., Pivarcsi A., Ståhle M. The human antimicrobial peptide LL-37 suppresses apoptosis in keratinocytes. J. Investig. Dermatol. 2009;129:937–944. doi: 10.1038/jid.2008.321. [DOI] [PubMed] [Google Scholar]
- 26.Tomasinsig L., Pizzirani C., Skerlavaj B., Pellegatti P., Gulinelli S., Tossi A., Di Virgilio F., Zanetti M. The human cathelicidin LL-37 modulates the activities of the P2X7 receptor in a structure-dependent manner. J. Biol. Chem. 2008;283:30471–30481. doi: 10.1074/jbc.M802185200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Girnita A., Zheng H., Grönberg A., Girnita L., Ståhle M. Identification of the cathelicidin peptide LL-37 as agonist for the type I insulin-like growth factor receptor. Oncogene. 2012;31:352–365. doi: 10.1038/onc.2011.239. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 28.Ramos R., Silva J.P., Rodrigues A.C., Costa R., Guardão L., Schmitt F., Soares R., Vilanova M., Domingues L., Gama M. Wound healing activity of the human antimicrobial peptide LL37. Peptides. 2011;32:1469–1476. doi: 10.1016/j.peptides.2011.06.005. [DOI] [PubMed] [Google Scholar]
- 29.Nordström R., Malmsten M. Delivery systems for antimicrobial peptides. Adv. Colloid Interface Sci. 2017;242:17–34. doi: 10.1016/j.cis.2017.01.005. [DOI] [PubMed] [Google Scholar]
- 30.Deng Y., Huang R., Huang S., Xiong M. Nanoparticles Enable Efficient Delivery of Antimicrobial Peptides for the Treatment of Deep Infections. BIO Integr. 2021 doi: 10.15212/bioi-2021-0003. [DOI] [Google Scholar]
- 31.Nemeth J., Oesch G., Kuster S.P. Bacteriostatic versus bactericidal antibiotics for patients with serious bacterial infections: Systematic review and meta-analysis. J. Antimicrob. Chemother. 2014;70:382–395. doi: 10.1093/jac/dku379. [DOI] [PubMed] [Google Scholar]
- 32.Adzitey F. Antibiotic Classes and Antibiotic Susceptibility of Bacterial Isolates from Selected Poultry; A Mini Review. World’s Vet. J. 2015;5:36–41. doi: 10.5455/wvj.20150853. [DOI] [Google Scholar]
- 33.Grossman T.H. Tetracycline Antibiotics and Resistance. Cold Spring Harb. Perspect. Med. 2016;6:a025387. doi: 10.1101/cshperspect.a025387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Mendes R.E., Farrell D.J., Sader H.S., Streit J.M., Jones R.N. Update of the telavancin activity in vitro tested against a worldwide collection of Gram-positive clinical isolates (2013), when applying the revised susceptibility testing method. Diagn. Microbiol. Infect. Dis. 2015;81:275–279. doi: 10.1016/j.diagmicrobio.2014.12.011. [DOI] [PubMed] [Google Scholar]
- 35.Mallapragada S., Wadhwa A., Agrawal P. Antimicrobial peptides: The miraculous biological molecules. J. Indian Soc. Periodontol. 2017;21:434. doi: 10.4103/jisp.jisp_325_16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hancock R.E. Peptide antibiotics. Lancet. 1997;349:418–422. doi: 10.1016/S0140-6736(97)80051-7. [DOI] [PubMed] [Google Scholar]
- 37.Tennessen J. Molecular evolution of animal antimicrobial peptides: Widespread moderate positive selection. J. Evol. Biol. 2005;18:1387–1394. doi: 10.1111/j.1420-9101.2005.00925.x. [DOI] [PubMed] [Google Scholar]
- 38.Li A., Lee P., Ho B., Ding J., Lim C. Atomic force microscopy study of the antimicrobial action of Sushi peptides on Gram negative bacteria. Biochim. Biophys. Acta (BBA)—Biomembr. 2007;1768:411–418. doi: 10.1016/j.bbamem.2006.12.010. [DOI] [PubMed] [Google Scholar]
- 39.Meincken M., Holroyd D., Rautenbach M. Atomic force microscopy study of the effect of antimicrobial peptides on the cell envelope of Escherichia coli. Antimicrob. Agents Chemother. 2005;49:4085–4092. doi: 10.1128/AAC.49.10.4085-4092.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Pushpanathan M., Gunasekaran P., Rajendhran J. Antimicrobial Peptides: Versatile Biological Properties. Int. J. Pept. 2013;2013:675391. doi: 10.1155/2013/675391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Brogden K.A. Antimicrobial peptides: Pore formers or metabolic inhibitors in bacteria? Nat. Rev. Microbiol. 2005;3:238–250. doi: 10.1038/nrmicro1098. [DOI] [PubMed] [Google Scholar]
- 42.Roversi D., Luca V., Aureli S., Park Y., Mangoni M.L., Stella L. How many antimicrobial peptide molecules kill a bacterium? The case of PMAP-23. ACS Chem. Biol. 2014;9:2003–2007. doi: 10.1021/cb500426r. [DOI] [PubMed] [Google Scholar]
- 43.Marr A.K., Gooderham W.J., Hancock R.E. Antibacterial peptides for therapeutic use: Obstacles and realistic outlook. Curr. Opin. Pharmacol. 2006;6:468–472. doi: 10.1016/j.coph.2006.04.006. [DOI] [PubMed] [Google Scholar]
- 44.Yount N.Y., Yeaman M.R. Emerging themes and therapeutic prospects for anti-infective peptides. Annu. Rev. Pharmacol. Toxicol. 2012;52:337–360. doi: 10.1146/annurev-pharmtox-010611-134535. [DOI] [PubMed] [Google Scholar]
- 45.Fox J.L. Antimicrobial peptides stage a comeback: Better understanding of the mechanisms of action, modification and synthesis of antimicrobial peptides is reigniting commercial development. Nat. Biotechnol. 2013;31:379–383. doi: 10.1038/nbt.2572. [DOI] [PubMed] [Google Scholar]
- 46.Riool M., de Breij A., de Boer L., Kwakman P.H., Cordfunke R.A., Cohen O., Malanovic N., Emanuel N., Lohner K., Drijfhout J.W. Controlled Release of LL-37-Derived Synthetic Antimicrobial and Anti-Biofilm Peptides SAAP-145 and SAAP-276 Prevents Experimental Biomaterial-Associated Staphylococcus aureus Infection. Adv. Funct. Mater. 2017;27:1606623. doi: 10.1002/adfm.201606623. [DOI] [Google Scholar]
- 47.Agerberth B., Gunne H., Odeberg J., Kogner P., Boman H.G., Gudmundsson G.H. FALL-39, a putative human peptide antibiotic, is cysteine-free and expressed in bone marrow and testis. Proc. Natl. Acad. Sci. USA. 1995;92:195–199. doi: 10.1073/pnas.92.1.195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Berkowitz B.A., Bevins C.L., Zasloff M.A. Magainins: A new family of membrane-active host defense peptides. Biochem. Pharmacol. 1990;39:625–629. doi: 10.1016/0006-2952(90)90138-B. [DOI] [PubMed] [Google Scholar]
- 49.Zasloff M. Magainins, a class of antimicrobial peptides from Xenopus skin: Isolation, characterization of two active forms, and partial cDNA sequence of a precursor. Proc. Natl. Acad. Sci. USA. 1987;84:5449–5453. doi: 10.1073/pnas.84.15.5449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Andersson M., Boman A., Boman H. Ascaris nematodes from pig and human make three anti-bacterial peptides: Isolation of cecropin P1 and two ASABF peptides. Cell. Mol. Life Sci. 2003;60:599–606. doi: 10.1007/s000180300051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Nguyen L.T., Haney E.F., Vogel H.J. The expanding scope of antimicrobial peptide structures and their modes of action. Trends Biotechnol. 2011;29:464–472. doi: 10.1016/j.tibtech.2011.05.001. [DOI] [PubMed] [Google Scholar]
- 52.Ganz T. Defensins: Antimicrobial peptides of innate immunity. Nat. Rev. Immunol. 2003;3:710–720. doi: 10.1038/nri1180. [DOI] [PubMed] [Google Scholar]
- 53.Mygind P.H., Fischer R.L., Schnorr K.M., Hansen M.T., Sönksen C.P., Ludvigsen S., Raventós D., Buskov S., Christensen B., De Maria L. Plectasin is a peptide antibiotic with therapeutic potential from a saprophytic fungus. Nature. 2005;437:975–980. doi: 10.1038/nature04051. [DOI] [PubMed] [Google Scholar]
- 54.Kokryakov V.N., Harwig S.S., Panyutich E.A., Shevchenko A.A., Aleshina G.M., Shamova O.V., Korneva H.A., Lehrer R.I. Protegrins: Leukocyte antimicrobial peptides that combine features of corticostatic defensins and tachyplesins. FEBS Lett. 1993;327:231–236. doi: 10.1016/0014-5793(93)80175-T. [DOI] [PubMed] [Google Scholar]
- 55.Falla T.J., Karunaratne D.N., Hancock R.E. Mode of action of the antimicrobial peptide indolicidin. J. Biol. Chem. 1996;271:19298–19303. doi: 10.1074/jbc.271.32.19298. [DOI] [PubMed] [Google Scholar]
- 56.Frank R.W., Gennaro R., Schneider K., Przybylski M., Romeo D. Amino acid sequences of two proline-rich bactenecins. Antimicrobial peptides of bovine neutrophils. J. Biol. Chem. 1990;265:18871–18874. doi: 10.1016/S0021-9258(17)30595-1. [DOI] [PubMed] [Google Scholar]
- 57.Takahashi D., Shukla S.K., Prakash O., Zhang G. Structural determinants of host defense peptides for antimicrobial activity and target cell selectivity. Biochimie. 2010;92:1236–1241. doi: 10.1016/j.biochi.2010.02.023. [DOI] [PubMed] [Google Scholar]
- 58.Dings R.P., Haseman J.R., Leslie D.B., Luong M., Dunn D.L., Mayo K.H. Bacterial membrane disrupting dodecapeptide SC4 improves survival of mice challenged with Pseudomonas aeruginosa. Biochim. Et Biophys. Acta (Bba)-Gen. Subj. 2013;1830:3454–3457. doi: 10.1016/j.bbagen.2013.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Nakamura T., Furunaka H., Miyata T., Tokunaga F., Muta T., Iwanaga S., Niwa M., Takao T., Shimonishi Y. Tachyplesin, a class of antimicrobial peptide from the hemocytes of the horseshoe crab (Tachypleus tridentatus). Isolation and chemical structure. J. Biol. Chem. 1988;263:16709–16713. doi: 10.1016/S0021-9258(18)37448-9. [DOI] [PubMed] [Google Scholar]
- 60.Xu F., Meng K., Wang Y.-R., Luo H.-Y., Yang P.-L., Wu N.-F., Fan Y.-L., Yao B. Eukaryotic expression and antimicrobial spectrum determination of the peptide tachyplesin II. Protein Expr. Purif. 2008;58:175–183. doi: 10.1016/j.pep.2007.12.007. [DOI] [PubMed] [Google Scholar]
- 61.Di Giulio A., Zhao H. Antimicrobial peptides: Basic mechanisms of action and emerging pharmacological interest. Asian J. Biochem. 2006;1:28–40. [Google Scholar]
- 62.Mojsoska B., Zuckermann R.N., Jenssen H. Structure-activity relationship study of novel peptoids that mimic the structure of antimicrobial peptides. Antimicrob. Agents Chemother. 2015;59:4112–4120. doi: 10.1128/AAC.00237-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Bayer A., Lammel J., Tohidnezhad M., Lippross S., Behrendt P., Klüter T., Pufe T., Cremer J., Jahr H., Rademacher F. The antimicrobial peptide human beta-defensin-3 is induced by platelet-released growth factors in primary keratinocytes. Mediat. Inflamm. 2017;2017:6157491. doi: 10.1155/2017/6157491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Ulm H., Wilmes M., Shai Y., Sahl H.-G. Antimicrobial host defensins–specific antibiotic activities and innate defense modulation. Front. Immunol. 2012;3:249. doi: 10.3389/fimmu.2012.00249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Kumar P., Kizhakkedathu J.N., Straus S.K. Antimicrobial peptides: Diversity, mechanism of action and strategies to improve the activity and biocompatibility in vivo. Biomolecules. 2018;8:4. doi: 10.3390/biom8010004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Seyfi R., Kahaki F.A., Ebrahimi T., Montazersaheb S., Eyvazi S., Babaeipour V., Tarhriz V. Antimicrobial peptides (AMPs): Roles, functions and mechanism of action. Int. J. Peptide Res. Ther. 2020;26:1451–1463. doi: 10.1007/s10989-019-09946-9. [DOI] [Google Scholar]
- 67.Raheem N., Straus S.K. Mechanisms of action for antimicrobial peptides with antibacterial and antibiofilm functions. Front. Microbiol. 2019;10:2866. doi: 10.3389/fmicb.2019.02866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Haney E.F., Hancock R.E. Peptide design for antimicrobial and immunomodulatory applications. Pept. Sci. 2013;100:572–583. doi: 10.1002/bip.22250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Gentilucci L., De Marco R., Cerisoli L. Chemical modifications designed to improve peptide stability: Incorporation of non-natural amino acids, pseudo-peptide bonds, and cyclization. Curr. Pharm. Des. 2010;16:3185–3203. doi: 10.2174/138161210793292555. [DOI] [PubMed] [Google Scholar]
- 70.Wang C., Wang Y., Zhang L., Miron R.J., Liang J., Shi M., Mo W., Zheng S., Zhao Y., Zhang Y. Pretreated Macrophage-Membrane-Coated Gold Nanocages for Precise Drug Delivery for Treatment of Bacterial Infections. Adv. Mater. 2018;30:e1804023. doi: 10.1002/adma.201804023. [DOI] [PubMed] [Google Scholar]
- 71.Chen W.Y., Chang H.Y., Lu J.K., Huang Y.C., Harroun S.G., Tseng Y.T., Li Y.J., Huang C.C., Chang H.T. Self-assembly of antimicrobial peptides on gold nanodots: Against multidrug-resistant bacteria and wound-healing application. Adv. Funct. Mater. 2015;25:7189–7199. doi: 10.1002/adfm.201503248. [DOI] [Google Scholar]
- 72.Rai A., Pinto S., Velho T.R., Ferreira A.F., Moita C., Trivedi U., Evangelista M., Comune M., Rumbaugh K.P., Simões P.N. One-step synthesis of high-density peptide-conjugated gold nanoparticles with antimicrobial efficacy in a systemic infection model. Biomaterials. 2016;85:99–110. doi: 10.1016/j.biomaterials.2016.01.051. [DOI] [PubMed] [Google Scholar]
- 73.Akrami M., Balalaie S., Hosseinkhani S., Alipour M., Salehi F., Bahador A., Haririan I. Tuning the anticancer activity of a novel pro-apoptotic peptide using gold nanoparticle platforms. Sci. Rep. 2016;6:1–12. doi: 10.1038/srep31030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Geilich B.M., van de Ven A.L., Singleton G.L., Sepúlveda L.J., Sridhar S., Webster T.J. Silver nanoparticle-embedded polymersome nanocarriers for the treatment of antibiotic-resistant infections. Nanoscale. 2015;7:3511–3519. doi: 10.1039/C4NR05823B. [DOI] [PubMed] [Google Scholar]
- 75.Braun K., Pochert A., Lindén M., Davoudi M., Schmidtchen A., Nordström R., Malmsten M. Membrane interactions of mesoporous silica nanoparticles as carriers of antimicrobial peptides. J. Colloid Interface Sci. 2016;475:161–170. doi: 10.1016/j.jcis.2016.05.002. [DOI] [PubMed] [Google Scholar]
- 76.d’Angelo I., Casciaro B., Miro A., Quaglia F., Mangoni M.L., Ungaro F. Overcoming barriers in Pseudomonas aeruginosa lung infections: Engineered nanoparticles for local delivery of a cationic antimicrobial peptide. Colloids Surf. B Biointerfaces. 2015;135:717–725. doi: 10.1016/j.colsurfb.2015.08.027. [DOI] [PubMed] [Google Scholar]
- 77.Water J.J., Smart S., Franzyk H., Foged C., Nielsen H.M. Nanoparticle-mediated delivery of the antimicrobial peptide plectasin. Eur. J. Pharm. Biopharm. 2015;92:65–73. doi: 10.1016/j.ejpb.2015.02.009. [DOI] [PubMed] [Google Scholar]
- 78.Wu C., Wu T., Fang Z., Zheng J., Xu S., Chen S., Hu Y., Ye X. Formation, characterization and release kinetics of chitosan/γ-PGA encapsulated nisin nanoparticles. RSC Adv. 2016;6:46686–46695. doi: 10.1039/C6RA06003J. [DOI] [Google Scholar]
- 79.Teixeira M.C., Carbone C., Sousa M.C., Espina M., Garcia M.L., Sanchez-Lopez E., Souto E.B. Nanomedicines for the delivery of antimicrobial peptides (Amps) Nanomaterials. 2020;10:560. doi: 10.3390/nano10030560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Shukla A., Fleming K.E., Chuang H.F., Chau T.M., Loose C.R., Stephanopoulos G.N., Hammond P.T. Controlling the release of peptide antimicrobial agents from surfaces. Biomaterials. 2010;31:2348–2357. doi: 10.1016/j.biomaterials.2009.11.082. [DOI] [PubMed] [Google Scholar]
- 81.Parilti R., Caprasse J., Riva R., Alexandre M., Vandegaart H., Bebrone C., Dupont-Gillain C., Howdle S.M., Jérôme C. Antimicrobial peptide encapsulation and sustained release from polymer network particles prepared in supercritical carbon dioxide. J. Colloid Interface Sci. 2018;532:112–117. doi: 10.1016/j.jcis.2018.07.125. [DOI] [PubMed] [Google Scholar]
- 82.Yang G., Huang T., Wang Y., Wang H., Li Y., Yu K., Dong L. Sustained Release of Antimicrobial Peptide from Self-Assembling Hydrogel Enhanced Osteogenesis. J. Biomater. Sci. Polym. Ed. 2018;29:1812–1824. doi: 10.1080/09205063.2018.1504191. [DOI] [PubMed] [Google Scholar]
- 83.Zetterberg M.M., Reijmar K., Pränting M., Engström Å., Andersson D.I., Edwards K. PEG-stabilized lipid disks as carriers for amphiphilic antimicrobial peptides. J. Control. Release. 2011;156:323–328. doi: 10.1016/j.jconrel.2011.08.029. [DOI] [PubMed] [Google Scholar]
- 84.Correa M.G., Martínez F.B., Vidal C.P., Streitt C., Escrig J., de Dicastillo C.L. Antimicrobial metal-based nanoparticles: A review on their synthesis, types and antimicrobial action. Beilstein J. Nanotechnol. 2020;11:1450–1469. doi: 10.3762/bjnano.11.129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Alves M.M., Bouchami O., Tavares A., Córdoba L., Santos C.F., Miragaia M., de Fátima Montemor M. New insights into antibiofilm effect of a nanosized ZnO coating against the pathogenic methicillin resistant Staphylococcus aureus. ACS Appl. Mater. Interfaces. 2017;9:28157–28167. doi: 10.1021/acsami.7b02320. [DOI] [PubMed] [Google Scholar]
- 86.Oun A.A., Rhim J.-W. Carrageenan-based hydrogels and films: Effect of ZnO and CuO nanoparticles on the physical, mechanical, and antimicrobial properties. Food Hydrocoll. 2017;67:45–53. doi: 10.1016/j.foodhyd.2016.12.040. [DOI] [Google Scholar]
- 87.Shankar S., Rhim J.-W. Facile approach for large-scale production of metal and metal oxide nanoparticles and preparation of antibacterial cotton pads. Carbohydr. Polym. 2017;163:137–145. doi: 10.1016/j.carbpol.2017.01.059. [DOI] [PubMed] [Google Scholar]
- 88.Li J., Cha R., Zhao X., Guo H., Luo H., Wang M., Zhou F., Jiang X.J.A.N. Gold nanoparticles cure bacterial infection with benefit to intestinal microflora. ACS Nano. 2019;13:5002–5014. doi: 10.1021/acsnano.9b01002. [DOI] [PubMed] [Google Scholar]
- 89.Akbar A., Sadiq M.B., Ali I., Muhammad N., Rehman Z., Khan M.N., Muhammad J., Khan S.A., Rehman F.U., Anal A.K. Synthesis and antimicrobial activity of zinc oxide nanoparticles against foodborne pathogens Salmonella typhimurium and Staphylococcus aureus. Biocatal. Agric. Biotechnol. 2019;17:36–42. doi: 10.1016/j.bcab.2018.11.005. [DOI] [Google Scholar]
- 90.Chen J., Wu L., Lu M., Lu S., Li Z., Ding W. Comparative Study on the Fungicidal Activity of Metallic MgO Nanoparticles and Macroscale MgO Against Soilborne Fungal Phytopathogens. Front. Microbiol. 2020;11:365. doi: 10.3389/fmicb.2020.00365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Guilger-Casagrande M., Lima R.D. Synthesis of Silver Nanoparticles Mediated by Fungi: A Review. Front. Bioeng. Biotechnol. 2019;7:287. doi: 10.3389/fbioe.2019.00287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Swain P., Nayak S.K., Sasmal A., Behera T., Barik S.K., Swain S.K., Mishra S.S., Sen A.K., Das J.K., Jayasankar P. Antimicrobial activity of metal based nanoparticles against microbes associated with diseases in aquaculture. World J. Microbiol. Biotechnol. 2014;30:2491–2502. doi: 10.1007/s11274-014-1674-4. [DOI] [PubMed] [Google Scholar]
- 93.Mallmann EJ J., Cunha F.A., Castro B.N., Maciel A.M., Menezes E.A., Fechine P.B.A. Antifungal activity of silver nanoparticles obtained by green synthesis. Rev. Inst. Med. Trop. São Paulo. 2015;57:165–167. doi: 10.1590/S0036-46652015000200011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Galdiero S., Falanga A., Vitiello M., Cantisani M., Marra V., Galdiero M. Silver nanoparticles as potential antiviral agents. Molecules. 2011;16:8894–8918. doi: 10.3390/molecules16108894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Sharmin S., Rahaman M.M., Sarkar C., Atolani O., Islam M.T., Adeyemi O.S. Nanoparticles as antimicrobial and antiviral agents: A literature-based perspective study. Heliyon. 2021;7:e06456. doi: 10.1016/j.heliyon.2021.e06456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Erkoc P., Ulucan-Karnak F.J.P. Nanotechnology-Based Antimicrobial and Antiviral Surface Coating Strategies. Prosthesis. 2021;3:25–52. doi: 10.3390/prosthesis3010005. [DOI] [Google Scholar]
- 97.Deepika M.S., Thangam R., Sundarraj S., Sheena T.S., Sivasubramanian S., Kulandaivel J., Thirumurugan R. Co-delivery of diverse therapeutic compounds using PEG–PLGA nanoparticle cargo against drug-resistant bacteria: An improved anti-biofilm strategy. ACS Appl. Bio Mater. 2019;3:385–399. doi: 10.1021/acsabm.9b00850. [DOI] [PubMed] [Google Scholar]
- 98.Durak S., Arasoglu T., Ates S.C., Derman S. Enhanced antibacterial and antiparasitic activity of multifunctional polymeric nanoparticles. Nanotechnology. 2020;31:175705. doi: 10.1088/1361-6528/ab6ab9. [DOI] [PubMed] [Google Scholar]
- 99.Alamdaran M., Movahedi B., Mohabatkar H., Behbahani M. In-vitro study of the novel nanocarrier of chitosan-based nanoparticles conjugated HIV-1 P24 protein-derived peptides. J. Mol. Liq. 2018;265:243–250. doi: 10.1016/j.molliq.2018.05.137. [DOI] [Google Scholar]
- 100.Real D., Hoffmann S., Leonardi D., Salomon C., Goycoolea F.M. Chitosan-based nanodelivery systems applied to the development of novel triclabendazole formulations. PLoS ONE. 2018;13:e0207625. doi: 10.1371/journal.pone.0207625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Zafar N., Shamaila S., Nazir J., Sharif R., Rafique M.S., Ul-Hasan J., Ammara S., Khalid H. Antibacterial action of chemically synthesized and laser generated silver nanoparticles against human pathogenic bacteria. J. Mater. Sci. Technol. 2016;32:721–728. doi: 10.1016/j.jmst.2016.05.009. [DOI] [Google Scholar]
- 102.Khalid H., Shamaila S., Zafar N., Shahzadi S. Synthesis of copper nanoparticles by chemical reduction method. Sci. Int. 2015;27:3085–3088. [Google Scholar]
- 103.Adewale O.B., Anadozie S.O., Potts-Johnson S.S., Onwuelu J.O., Obafemi T.O., Osukoya O.A., Fadaka A.O., Davids H., Roux S. Investigation of bioactive compounds in Crassocephalum rubens leaf and in vitro anticancer activity of its biosynthesized gold nanoparticles. Biotechnol. Rep. 2020;28:e00560. doi: 10.1016/j.btre.2020.e00560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Pelgrift R.Y., Friedman A.J. Nanotechnology as a therapeutic tool to combat microbial resistance. Adv. Drug Deliv. Rev. 2013;65:1803–1815. doi: 10.1016/j.addr.2013.07.011. [DOI] [PubMed] [Google Scholar]
- 105.Ivask A., Kurvet I., Kasemets K., Blinova I., Aruoja V., Suppi S., Vija H., Käkinen A., Titma T., Heinlaan M. Size-dependent toxicity of silver nanoparticles to bacteria, yeast, algae, crustaceans and mammalian cells in vitro. PLoS ONE. 2014;9:e102108. doi: 10.1371/journal.pone.0102108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Azam A., Ahmed A.S., Oves M., Khan M., Memic A. Size-dependent antimicrobial properties of CuO nanoparticles against Gram-positive and-negative bacterial strains. Int. J. Nanomed. 2012;7:3527. doi: 10.2147/IJN.S29020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Natan M., Banin E. From Nano to Micro: Using nanotechnology to combat microorganisms and their multidrug resistance. Fems Microbiol. Rev. 2017;41:302–322. doi: 10.1093/femsre/fux003. [DOI] [PubMed] [Google Scholar]
- 108.Popa M., Pradell T., Crespo D., Calderón-Moreno J.M. Stable silver colloidal dispersions using short chain polyethylene glycol. Colloids Surf. A Physicochem. Eng. Asp. 2007;303:184–190. doi: 10.1016/j.colsurfa.2007.03.050. [DOI] [Google Scholar]
- 109.Tolaymat T.M., El Badawy A.M., Genaidy A., Scheckel K.G., Luxton T.P., Suidan M. An evidence-based environmental perspective of manufactured silver nanoparticle in syntheses and applications: A systematic review and critical appraisal of peer-reviewed scientific papers. Sci. Total Environ. 2010;408:999–1006. doi: 10.1016/j.scitotenv.2009.11.003. [DOI] [PubMed] [Google Scholar]
- 110.Aboyewa J.A., Sibuyi N.R., Meyer M., Oguntibeju O.O. Green Synthesis of Metallic Nanoparticles Using Some Selected Medicinal Plants from Southern Africa and Their Biological Applications. Plants. 2021;10:1929. doi: 10.3390/plants10091929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Sibuyi N.R.S., Thipe V.C., Panjtan-Amiri K., Meyer M., Katti K.V. Green synthesis of gold nanoparticles using Acai berry and Elderberry extracts and investigation of their effect on prostate and pancreatic cancer cells. Nanobiomedicine. 2021;8:1849543521995310. doi: 10.1177/1849543521995310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Adewale O.B., Egbeyemi K.A., Onwuelu J.O., Potts-Johnson S.S., Anadozie S.O., Fadaka A.O., Osukoya O.A., Aluko B.T., Johnson J., Obafemi T.O. Biological synthesis of gold and silver nanoparticles using leaf extracts of Crassocephalum rubens and their comparative in vitro antioxidant activities. Heliyon. 2020;6:e05501. doi: 10.1016/j.heliyon.2020.e05501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Monowar T., Rahman M.S., Bhore S.J., Raju G., Sathasivam K.V. Silver Nanoparticles Synthesized by Using the Endophytic Bacterium Pantoea ananatis are Promising Antimicrobial Agents against Multidrug Resistant Bacteria. Molecules. 2018;23:3220. doi: 10.3390/molecules23123220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Escárcega-González C.E., Garza-Cervantes J.A., Vazquez-Rodríguez A., Montelongo-Peralta L.Z., Treviño-Gonzalez M.T., Castro E.D.B., Saucedo-Salazar E.M., Morales R.C., Soto D.R., González F.T. In vivo antimicrobial activity of silver nanoparticles produced via a green chemistry synthesis using Acacia rigidula as a reducing and capping agent. Int. J. Nanomed. 2018;13:2349. doi: 10.2147/IJN.S160605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Lopez-Abarrategui C., Figueroa-Espi V., Lugo-Alvarez M.B., Pereira C.D., Garay H., Barbosa J.A., Falcão R., Jiménez-Hernández L., Estévez-Hernández O., Reguera E., et al. The intrinsic antimicrobial activity of citric acid-coated manganese ferrite nanoparticles is enhanced after conjugation with the antifungal peptide Cm-p5. Int. J. Nanomed. 2016;11:3849–3857. doi: 10.2147/IJN.S107561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Brancolini G., Kokh D.B., Calzolai L., Wade R.C., Corni S. Docking of ubiquitin to gold nanoparticles. ACS Nano. 2012;6:9863–9878. doi: 10.1021/nn303444b. [DOI] [PubMed] [Google Scholar]
- 117.Kamar R., Réjasse A., Jéhanno I., Attieh Z., Courtin P., Chapot-Chartier M.-P., Nielsen-Leroux C., Lereclus D., El Chamy L., Kallassy M. DltX of Bacillus thuringiensis is essential for D-alanylation of teichoic acids and resistance to antimicrobial response in insects. Front. Microbiol. 2017;8:1437. doi: 10.3389/fmicb.2017.01437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Meireles D., Pombinho R., Carvalho F., Sousa S., Cabanes D. Listeria monocytogenes wall teichoic acid glycosylation promotes surface anchoring of virulence factors, resistance to antimicrobial peptides, and decreased susceptibility to antibiotics. Pathogens. 2020;9:290. doi: 10.3390/pathogens9040290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Joo H.-S., Otto M. Mechanisms of resistance to antimicrobial peptides in staphylococci. Biochim. Biophys. Acta (BBA)—Biomembr. 2015;1848:3055–3061. doi: 10.1016/j.bbamem.2015.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Joo H.-S., Fu C.-I., Otto M. Bacterial strategies of resistance to antimicrobial peptides. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2016;371:20150292. doi: 10.1098/rstb.2015.0292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Spohn R., Daruka L., Lázár V., Martins A., Vidovics F., Grézal G., Méhi O., Kintses B., Számel M., Jangir P.K., et al. Integrated evolutionary analysis reveals antimicrobial peptides with limited resistance. Nat. Commun. 2019;10:4538. doi: 10.1038/s41467-019-12364-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Rodríguez-Rojas A., Baeder D.Y., Johnston P., Regoes R.R., Rolff J. Bacteria primed by antimicrobial peptides develop tolerance and persist. PLoS Pathog. 2021;17:e1009443. doi: 10.1371/journal.ppat.1009443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Baindara P., Ghosh A.K., Mandal S.M. Coevolution of resistance against antimicrobial peptides. Microb. Drug Resist. 2020;26:880–899. doi: 10.1089/mdr.2019.0291. [DOI] [PubMed] [Google Scholar]
- 124.Andersson D.I., Hughes D., Kubicek-Sutherland J.Z. Mechanisms and consequences of bacterial resistance to antimicrobial peptides. Drug Resist. Updates. 2016;26:43–57. doi: 10.1016/j.drup.2016.04.002. [DOI] [PubMed] [Google Scholar]
- 125.El Shazely B., Yu G., Johnston P.R., Rolff J. Resistance Evolution Against Antimicrobial Peptides in Staphylococcus aureus Alters Pharmacodynamics Beyond the MIC. Front. Microbiol. 2020;11 doi: 10.3389/fmicb.2020.00103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Mahlapuu M., Håkansson J., Ringstad L., Björn C. Antimicrobial peptides: An emerging category of therapeutic agents. Front. Cell. Infect. Microbiol. 2016;6:194. doi: 10.3389/fcimb.2016.00194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Moravej H., Moravej Z., Yazdanparast M., Heiat M., Mirhosseini A., Moosazadeh Moghaddam M., Mirnejad R. Antimicrobial peptides: Features, action, and their resistance mechanisms in bacteria. Microb. Drug Resist. 2018;24:747–767. doi: 10.1089/mdr.2017.0392. [DOI] [PubMed] [Google Scholar]
- 128.Abo-zeid Y., Williams G.R. The potential anti-infective applications of metal oxide nanoparticles: A systematic review. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2020;12:e1592. doi: 10.1002/wnan.1592. [DOI] [PubMed] [Google Scholar]
- 129.Beyth N., Houri-Haddad Y., Domb A., Khan W., Hazan R. Alternative antimicrobial approach: Nano-antimicrobial materials. Evid.-Based Complement. Altern. Med. 2015;2015:246012. doi: 10.1155/2015/246012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Caster J.M., Patel A.N., Zhang T., Wang A. Investigational nanomedicines in 2016: A review of nanotherapeutics currently undergoing clinical trials. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2017;9:e1416. doi: 10.1002/wnan.1416. [DOI] [PubMed] [Google Scholar]
- 131.Smerkova K., Dolezelikova K., Bozdechova L., Heger Z., Zurek L., Adam V. Nanomaterials with active targeting as advanced antimicrobials. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2020;12:e1636. doi: 10.1002/wnan.1636. [DOI] [PubMed] [Google Scholar]
- 132.Jelinkova P., Mazumdar A., Sur V.P., Kociova S., Dolezelikova K., Jimenez A.M.J., Koudelkova Z., Mishra P.K., Smerkova K., Heger Z. Nanoparticle-drug conjugates treating bacterial infections. J. Control. Release. 2019;307:166–185. doi: 10.1016/j.jconrel.2019.06.013. [DOI] [PubMed] [Google Scholar]
- 133.Baier G., Cavallaro A., Vasilev K., Mailänder V., Musyanovych A., Landfester K. Enzyme responsive hyaluronic acid nanocapsules containing polyhexanide and their exposure to bacteria to prevent infection. Biomacromolecules. 2013;14:1103–1112. doi: 10.1021/bm302003m. [DOI] [PubMed] [Google Scholar]
- 134.Boas U., Heegaard P.M. Dendrimers in drug research. Chem. Soc. Rev. 2004;33:43–63. doi: 10.1039/b309043b. [DOI] [PubMed] [Google Scholar]
- 135.Esmaeili F., Hosseini-Nasr M., Rad-Malekshahi M., Samadi N., Atyabi F., Dinarvand R. Preparation and antibacterial activity evaluation of rifampicin-loaded poly lactide-co-glycolide nanoparticles. Nanomed. Nanotechnol. Biol. Med. 2007;3:161–167. doi: 10.1016/j.nano.2007.03.003. [DOI] [PubMed] [Google Scholar]
- 136.Schumacher I., Margalit R. Liposome-encapsulated ampicillin: Physicochemical and antibacterial properties. J. Pharm. Sci. 1997;86:635–641. doi: 10.1021/js9503690. [DOI] [PubMed] [Google Scholar]
- 137.Torchilin V.P. Micellar nanocarriers: Pharmaceutical perspectives. Pharm. Res. 2007;24:1–16. doi: 10.1007/s11095-006-9132-0. [DOI] [PubMed] [Google Scholar]
- 138.Amina S.J., Guo B. A Review on the Synthesis and Functionalization of Gold Nanoparticles as a Drug Delivery Vehicle. Int. J. Nanomed. 2020;15:9823–9857. doi: 10.2147/IJN.S279094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Murphy C.J., Gole A.M., Stone J.W., Sisco P.N., Alkilany A.M., Goldsmith E.C., Baxter S.C. Gold nanoparticles in biology: Beyond toxicity to cellular imaging. Acc. Chem. Res. 2008;41:1721–1730. doi: 10.1021/ar800035u. [DOI] [PubMed] [Google Scholar]
- 140.Lee K.-S., El-Sayed M.A. Gold and silver nanoparticles in sensing and imaging: Sensitivity of plasmon response to size, shape, and metal composition. J. Phys. Chem. B. 2006;110:19220–19225. doi: 10.1021/jp062536y. [DOI] [PubMed] [Google Scholar]
- 141.Toderas F., Baia M., Maniu D., Astilean S. Tuning the plasmon resonances of gold nanoparticles by controlling their size and shape. J. Optoelectron. Adv. Mater. 2008;10:2282–2284. [Google Scholar]
- 142.Boisselier E., Astruc D. Gold nanoparticles in nanomedicine: Preparations, imaging, diagnostics, therapies and toxicity. Chem. Soc. Rev. 2009;38:1759–1782. doi: 10.1039/b806051g. [DOI] [PubMed] [Google Scholar]
- 143.Tran Q.H., Le A.-T. Silver nanoparticles: Synthesis, properties, toxicology, applications and perspectives. Adv. Nat. Sci. Nanosci. Nanotechnol. 2013;4:033001. doi: 10.1088/2043-6262/4/3/033001. [DOI] [Google Scholar]
- 144.Shen J., Yu M., Meng Q., Li J., Lv Y., Lu W. Fatty acid-based strategy for efficient brain targeted gene delivery. Pharm. Res. 2013;30:2573–2583. doi: 10.1007/s11095-013-1056-x. [DOI] [PubMed] [Google Scholar]
- 145.Nikanjam M., Gibbs A.R., Hunt C.A., Budinger T.F., Forte T.M. Synthetic nano-LDL with paclitaxel oleate as a targeted drug delivery vehicle for glioblastoma multiforme. J. Control. Release. 2007;124:163–171. doi: 10.1016/j.jconrel.2007.09.007. [DOI] [PubMed] [Google Scholar]
- 146.Dhar S., Gu F.X., Langer R., Farokhzad O.C., Lippard S.J. Targeted delivery of cisplatin to prostate cancer cells by aptamer functionalized Pt(IV) prodrug-PLGA-PEG nanoparticles. Proc. Natl. Acad. Sci. USA. 2008;105:17356–17361. doi: 10.1073/pnas.0809154105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Górski A., Wasik M., Nowaczyk M., Korczak-Kowalska G. Immunomodulating activity of heparin. FASEB J. 1991;5:2287–2291. doi: 10.1096/fasebj.5.9.1860620. [DOI] [PubMed] [Google Scholar]
- 148.Zhang H., Luo J., Li Y., Henderson P.T., Wang Y., Wachsmann-Hogiu S., Zhao W., Lam K.S., Pan C.X. Characterization of high-affinity peptides and their feasibility for use in nanotherapeutics targeting leukemia stem cells. Nanomedicine. 2012;8:1116–1124. doi: 10.1016/j.nano.2011.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Liao D., Liu Z., Wrasidlo W., Chen T., Luo Y., Xiang R., Reisfeld R.A. Synthetic enzyme inhibitor: A novel targeting ligand for nanotherapeutic drug delivery inhibiting tumor growth without systemic toxicity. Nanomed. Nanotechnol. Biol. Med. 2011;7:665–673. doi: 10.1016/j.nano.2011.03.001. [DOI] [PubMed] [Google Scholar]
- 150.Chikkaveeraiah B.V., Soldà A., Choudhary D., Maran F., Rusling J.F. Ultrasensitive nanostructured immunosensor for stem and carcinoma cell pluripotency gatekeeper protein NANOG. Nanomedicine. 2012;7:957–965. doi: 10.2217/nnm.11.178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Lee G.Y., Kim J.H., Oh G.T., Lee B.H., Kwon I.C., Kim I.S. Molecular targeting of atherosclerotic plaques by a stabilin-2-specific peptide ligand. J. Control. Release. 2011;155:211–217. doi: 10.1016/j.jconrel.2011.07.010. [DOI] [PubMed] [Google Scholar]
- 152.Almer G., Wernig K., Saba-Lepek M., Haj-Yahya S., Rattenberger J., Wagner J., Gradauer K., Frascione D., Pabst G., Leitinger G., et al. Adiponectin-coated nanoparticles for enhanced imaging of atherosclerotic plaques. Int. J. Nanomed. 2011;6:1279–1290. doi: 10.2147/ijn.S18739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Li N., Larson T., Nguyen H.H., Sokolov K.V., Ellington A.D. Directed evolution of gold nanoparticle delivery to cells. Chem. Commun. 2010;46:392–394. doi: 10.1039/B920865H. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Iqbal M.Z., Ali I., Khan W.S., Kong X., Dempsey E. Reversible self-assembly of gold nanoparticles in response to external stimuli. Mater. Des. 2021;205:109694. doi: 10.1016/j.matdes.2021.109694. [DOI] [Google Scholar]
- 155.Park S., Lee W.J., Park S., Choi D., Kim S., Park N. Reversibly pH-responsive gold nanoparticles and their applications for photothermal cancer therapy. Sci. Rep. 2019;9:1–9. doi: 10.1038/s41598-019-56754-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Bernardim B., Matos M.J., Ferhati X., Compañón I., Guerreiro A., Akkapeddi P., Burtoloso A.C., Jiménez-Osés G., Corzana F., Bernardes G.J. Efficient and irreversible antibody–cysteine bioconjugation using carbonylacrylic reagents. Nat. Protoc. 2019;14:86–99. doi: 10.1038/s41596-018-0083-9. [DOI] [PubMed] [Google Scholar]
- 157.S Sibuyi N.R., Thovhogi N., Gabuza K.B., Meyer M.D., Drah M., Onani M.O., Skepu A., Madiehe A.M., Meyer M. Peptide-functionalized nanoparticles for the selective induction of apoptosis in target cells. Nanomedicine. 2017;12:1631–1645. doi: 10.2217/nnm-2017-0085. [DOI] [PubMed] [Google Scholar]
- 158.Hossen M.N., Kajimoto K., Akita H., Hyodo M., Ishitsuka T., Harashima H. Ligand-based targeted delivery of a peptide modified nanocarrier to endothelial cells in adipose tissue. J. Control. Release. 2010;147:261–268. doi: 10.1016/j.jconrel.2010.07.100. [DOI] [PubMed] [Google Scholar]
- 159.Zhou Y., Kong Y., Kundu S., Cirillo J.D., Liang H. Antibacterial activities of gold and silver nanoparticles against Escherichia coli and bacillus Calmette-Guérin. J. Nanobiotechnol. 2012;10:1–9. doi: 10.1186/1477-3155-10-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Zhang K., Lv S., Lin Z., Li M., Tang D. Bio-bar-code-based photoelectrochemical immunoassay for sensitive detection of prostate-specific antigen using rolling circle amplification and enzymatic biocatalytic precipitation. Biosens. Bioelectron. 2018;101:159–166. doi: 10.1016/j.bios.2017.10.031. [DOI] [PubMed] [Google Scholar]
- 161.Ren R., Cai G., Yu Z., Zeng Y., Tang D. Metal-polydopamine framework: An innovative signal-generation tag for colorimetric immunoassay. Anal. Chem. 2018;90:11099–11105. doi: 10.1021/acs.analchem.8b03538. [DOI] [PubMed] [Google Scholar]
- 162.Lin Y., Zhou Q., Tang D., Niessner R., Knopp D. Signal-on photoelectrochemical immunoassay for aflatoxin B1 based on enzymatic product-etching MnO2 nanosheets for dissociation of carbon dots. Anal. Chem. 2017;89:5637–5645. doi: 10.1021/acs.analchem.7b00942. [DOI] [PubMed] [Google Scholar]
- 163.Oliveira J.P., Prado A.R., Keijok W.J., Antunes P.W.P., Yapuchura E.R., Guimarães M.C.C. Impact of conjugation strategies for targeting of antibodies in gold nanoparticles for ultrasensitive detection of 17β-estradiol. Sci. Rep. 2019;9:1–8. doi: 10.1038/s41598-019-50424-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Busch R.T., Karim F., Weis J., Sun Y., Zhao C., Vasquez E.S. Optimization and Structural Stability of Gold Nanoparticle–Antibody Bioconjugates. ACS Omega. 2019;4:15269–15279. doi: 10.1021/acsomega.9b02276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Pedrosa P., Mendes R., Cabral R., Martins L.M., Baptista P.V., Fernandes A.R. Combination of chemotherapy and Au-nanoparticle photothermy in the visible light to tackle doxorubicin resistance in cancer cells. Sci. Rep. 2018;8:1–8. doi: 10.1038/s41598-018-29870-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Mahmoud N.N., Alkilany A.M., Khalil E.A., Al-Bakri A.G. Nano-Photothermal ablation effect of Hydrophilic and Hydrophobic Functionalized Gold Nanorods on Staphylococcus aureus and Propionibacterium acnes. Sci. Rep. 2018;8:6881. doi: 10.1038/s41598-018-24837-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Chen Y.H., Tsai C.Y., Huang P.Y., Chang M.Y., Cheng P.C., Chou C.H., Chen D.H., Wang C.R., Shiau A.L., Wu C.L. Methotrexate conjugated to gold nanoparticles inhibits tumor growth in a syngeneic lung tumor model. Mol. Pharm. 2007;4:713–722. doi: 10.1021/mp060132k. [DOI] [PubMed] [Google Scholar]
- 168.Wang F., Wang Y.C., Dou S., Xiong M.H., Sun T.M., Wang J. Doxorubicin-tethered responsive gold nanoparticles facilitate intracellular drug delivery for overcoming multidrug resistance in cancer cells. ACS Nano. 2011;5:3679–3692. doi: 10.1021/nn200007z. [DOI] [PubMed] [Google Scholar]
- 169.Brown S.D., Nativo P., Smith J.A., Stirling D., Edwards P.R., Venugopal B., Flint D.J., Plumb J.A., Graham D., Wheate N.J. Gold nanoparticles for the improved anticancer drug delivery of the active component of oxaliplatin. J. Am. Chem. Soc. 2010;132:4678–4684. doi: 10.1021/ja908117a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Kong F.-Y., Zhang J.-W., Li R.-F., Wang Z.-X., Wang W.-J., Wang W. Unique roles of gold nanoparticles in drug delivery, targeting and imaging applications. Molecules. 2017;22:1445. doi: 10.3390/molecules22091445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Tapia D., Sanchez-Villamil J.I., Torres A.G. Multicomponent gold nano-glycoconjugate as a highly immunogenic and protective platform against Burkholderia mallei. NPJ Vaccines. 2020;5:1–11. doi: 10.1038/s41541-020-00229-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Mateu Ferrando R., Lay L., Polito L. Gold nanoparticle-based platforms for vaccine development. Drug Discov. Today Technol. 2021 doi: 10.1016/j.ddtec.2021.02.001. [DOI] [PubMed] [Google Scholar]
- 173.Shukla R., Bansal V., Chaudhary M., Basu A., Bhonde R.R., Sastry M. Biocompatibility of Gold Nanoparticles and Their Endocytotic Fate Inside the Cellular Compartment: A Microscopic Overview. Langmuir. 2005;21:10644–10654. doi: 10.1021/la0513712. [DOI] [PubMed] [Google Scholar]
- 174.Zhang Q., Hitchins V.M., Schrand A.M., Hussain S.M., Goering P.L. Uptake of gold nanoparticles in murine macrophage cells without cytotoxicity or production of pro-inflammatory mediators. Nanotoxicology. 2011;5:284–295. doi: 10.3109/17435390.2010.512401. [DOI] [PubMed] [Google Scholar]
- 175.Shikha S., Chaudhuri S.R., Bhattacharyya M.S.J.S.r. Facile one pot greener synthesis of sophorolipid capped gold nanoparticles and its antimicrobial activity having special efficacy against gram negative Vibrio cholerae. Sci. Rep. 2020;10:1–13. doi: 10.1038/s41598-019-57399-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Meeker D.G., Jenkins S.V., Miller E.K., Beenken K.E., Loughran A.J., Powless A., Muldoon T.J., Galanzha E.I., Zharov V.P., Smeltzer M.S. Synergistic photothermal and antibiotic killing of biofilm-associated Staphylococcus aureus using targeted antibiotic-loaded gold nanoconstructs. ACS Infect. Dis. 2016;2:241–250. doi: 10.1021/acsinfecdis.5b00117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Cui Y., Zhao Y., Tian Y., Zhang W., Lü X., Jiang X. The molecular mechanism of action of bactericidal gold nanoparticles on Escherichia coli. Biomaterials. 2012;33:2327–2333. doi: 10.1016/j.biomaterials.2011.11.057. [DOI] [PubMed] [Google Scholar]
- 178.Li P., Li J., Wu C., Wu Q., Li J. Synergistic antibacterial effects of β-lactam antibiotic combined with silver nanoparticles. Nanotechnology. 2005;16:1912. doi: 10.1088/0957-4484/16/9/082. [DOI] [Google Scholar]
- 179.Kvítek L., Panáček A., Soukupova J., Kolář M., Večeřová R., Prucek R., Holecová M., Zbořil R. Effect of surfactants and polymers on stability and antibacterial activity of silver nanoparticles (NPs) J. Phys. Chem. C. 2008;112:5825–5834. doi: 10.1021/jp711616v. [DOI] [Google Scholar]
- 180.Liu L., Yang J., Xie J., Luo Z., Jiang J., Yang Y.Y., Liu S. The potent antimicrobial properties of cell penetrating peptide-conjugated silver nanoparticles with excellent selectivity for Gram-positive bacteria over erythrocytes. Nanoscale. 2013;5:3834–3840. doi: 10.1039/c3nr34254a. [DOI] [PubMed] [Google Scholar]
- 181.Lee H.J., Lee S.G., Oh E.J., Chung H.Y., Han S.I., Kim E.J., Seo S.Y., Do Ghim H., Yeum J.H., Choi J.H. Antimicrobial polyethyleneimine-silver nanoparticles in a stable colloidal dispersion. Colloids Surf. B Biointerfaces. 2011;88:505–511. doi: 10.1016/j.colsurfb.2011.07.041. [DOI] [PubMed] [Google Scholar]
- 182.Li W.-R., Xie X.-B., Shi Q.-S., Zeng H.-Y., You-Sheng O.-Y., Chen Y.-B. Antibacterial activity and mechanism of silver nanoparticles on Escherichia coli. Appl. Microbiol. Biotechnol. 2010;85:1115–1122. doi: 10.1007/s00253-009-2159-5. [DOI] [PubMed] [Google Scholar]
- 183.Dube P., Meyer S., Madiehe A., Meyer M. Antibacterial activity of biogenic silver and gold nanoparticles synthesized from Salvia africana-lutea and Sutherlandia frutescens. Nanotechnology. 2020;31:505607. doi: 10.1088/1361-6528/abb6a8. [DOI] [PubMed] [Google Scholar]
- 184.Simon S., Sibuyi N.R.S., Fadaka A.O., Meyer M., Madiehe A.M., du Preez M.G. The antimicrobial activity of biogenic silver nanoparticles synthesized from extracts of Red and Green European pear cultivars. Artif. Cells Nanomed. Biotechnol. 2021;49:614–625. doi: 10.1080/21691401.2021.1980884. [DOI] [PubMed] [Google Scholar]
- 185.Elbagory A.M., Meyer M., Cupido C.N., Hussein A.A.J.N. Inhibition of bacteria associated with wound infection by biocompatible green synthesized gold nanoparticles from South African plant extracts. Nanomaterials. 2017;7:417. doi: 10.3390/nano7120417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Baker S., Olga P., Tatiana R., Nadezhda P., Tatyana G., Tatyana R., Saveleva E., Olga K., Elizaveta G., Karina G., et al. Phyto-nano-hybrids of Ag-CuO particles for antibacterial activity against drug-resistant pathogens. J. Genet. Eng. Biotechnol. 2020;18:1–8. doi: 10.1186/s43141-020-00068-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Ghosh S., Jagtap S., More P., Shete U.J., Maheshwari N.O., Rao S.J., Kitture R., Kale S., Bellare J., Patil S., et al. Dioscorea bulbifera mediated synthesis of novel AucoreAgshell nanoparticles with potent antibiofilm and antileishmanial activity. J. Nanomater. 2015;2015:161. doi: 10.1155/2015/562938. [DOI] [Google Scholar]
- 188.Unuofin J.O., Oladipo A.O., Msagati T.A., Lebelo S.L., Meddows-Taylor S., More G.K. Novel silver-platinum bimetallic nanoalloy synthesized from Vernonia mespilifolia extract: Antioxidant, antimicrobial, and cytotoxic activities. Arab. J. Chem. 2020;13:6639–6648. doi: 10.1016/j.arabjc.2020.06.019. [DOI] [Google Scholar]
- 189.Ranpariya B., Salunke G., Karmakar S., Babiya K., Sutar S., Kadoo N., Kumbhakar P., Ghosh S. Antimicrobial Synergy of Silver-Platinum Nanohybrids With Antibiotics. Front. Microbiol. 2020;11:610968. doi: 10.3389/fmicb.2020.610968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Kazemzadeh-Narbat M., Lai B.F.L., Ding C., Kizhakkedathu J.N., Hancock R.E.W., Wang R. Multilayered coating on titanium for controlled release of antimicrobial peptides for the prevention of implant-associated infections. Biomaterials. 2013;34:5969–5977. doi: 10.1016/j.biomaterials.2013.04.036. [DOI] [PubMed] [Google Scholar]
- 191.Moorcroft S.C., Roach L., Jayne D.G., Ong Z.Y., Evans S.D. Nanoparticle-loaded hydrogel for the light-activated release and photothermal enhancement of antimicrobial peptides. ACS Appl. Mater. Interfaces. 2020;12:24544–24554. doi: 10.1021/acsami.9b22587. [DOI] [PubMed] [Google Scholar]
- 192.Gao W., Vecchio D., Li J., Zhu J., Zhang Q., Fu V., Li J., Thamphiwatana S., Lu D., Zhang L. Hydrogel Containing Nanoparticle-Stabilized Liposomes for Topical Antimicrobial Delivery. ACS Nano. 2014;8:2900–2907. doi: 10.1021/nn500110a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Svenson S. Clinical translation of nanomedicines. Curr. Opin. Solid State Mater. Sci. 2012;16:287–294. doi: 10.1016/j.cossms.2012.10.001. [DOI] [Google Scholar]
- 194.Hare J.I., Lammers T., Ashford M.B., Puri S., Storm G., Barry S.T. Challenges and strategies in anti-cancer nanomedicine development: An industry perspective. Adv. Drug Deliv. Rev. 2017;108:25–38. doi: 10.1016/j.addr.2016.04.025. [DOI] [PubMed] [Google Scholar]
- 195.Hua S., de Matos M.B.C., Metselaar J.M., Storm G. Current Trends and Challenges in the Clinical Translation of Nanoparticulate Nanomedicines: Pathways for Translational Development and Commercialization. Front. Pharmacol. 2018;9:790. doi: 10.3389/fphar.2018.00790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Chopra I. The increasing use of silver-based products as antimicrobial agents: A useful development or a cause for concern? J. Antimicrob. Chemother. 2007;59:587–590. doi: 10.1093/jac/dkm006. [DOI] [PubMed] [Google Scholar]
- 197.Nqakala Z.B., Sibuyi N.R.S., Fadaka A.O., Meyer M., Onani M.O., Madiehe A.M. Advances in Nanotechnology towards Development of Silver Nanoparticle-Based Wound-Healing Agents. Int. J. Mol. Sci. 2021;22:11272. doi: 10.3390/ijms222011272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Browne K., Chakraborty S., Chen R., Willcox M.D., Black D.S., Walsh W.R., Kumar N. A New Era of Antibiotics: The Clinical Potential of Antimicrobial Peptides. Int. J. Mol. Sci. 2020;21:7047. doi: 10.3390/ijms21197047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Kim J., Conlon J.M., Iwamuro S., Knoop F. Antimicrobial peptides from the skin of the Japanese mountain brown frog, Rana ornativentris. J. Pept. Res. 2001;58:349–356. doi: 10.1034/j.1399-3011.2001.00947.x. [DOI] [PubMed] [Google Scholar]
- 200.Crost E., Ajandouz E., Villard C., Geraert P., Puigserver A., Fons M. Ruminococcin C, a new anti-Clostridium perfringens bacteriocin produced in the gut by the commensal bacterium Ruminococcus gnavus E1. Biochimie. 2011;93:1487–1494. doi: 10.1016/j.biochi.2011.05.001. [DOI] [PubMed] [Google Scholar]
- 201.Nijnik A., Madera L., Ma S., Waldbrook M., Elliott M.R., Easton D.M., Mayer M.L., Mullaly S.C., Kindrachuk J., Jenssen H. Synthetic cationic peptide IDR-1002 provides protection against bacterial infections through chemokine induction and enhanced leukocyte recruitment. J. Immunol. 2010;184:2539–2550. doi: 10.4049/jimmunol.0901813. [DOI] [PubMed] [Google Scholar]
- 202.Steinstraesser L., Hirsch T., Schulte M., Kueckelhaus M., Jacobsen F., Mersch E.A., Stricker I., Afacan N., Jenssen H., Hancock R.E. Innate defense regulator peptide 1018 in wound healing and wound infection. PLoS ONE. 2012;7:e39373. doi: 10.1371/journal.pone.0039373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Giacometti A., Cirioni O., Del Prete M.S., Paggi A.M., D’Errico M.M., Scalise G. Combination studies between polycationic peptides and clinically used antibiotics against Gram-positive and Gram-negative bacteria. Peptides. 2000;21:1155–1160. doi: 10.1016/S0196-9781(00)00254-0. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.