Abstract
We investigated the frequency of brain fog in a large cohort of patients with documented coronavirus disease‐2019 (COVID‐19) who have survived the illness. We also scrutinized the potential risk factors associated with the development of brain fog. Adult patients (18–55 years of age), who were referred to the healthcare facilities anywhere in Fars province from February 19, 2020 to November 20, 2020 were included. All patients had a confirmed COVID‐19 diagnosis. In a phone call, at least 3 months after their discharge from the hospital, we obtained their current information. A questionnaire was specifically designed for data collection. In total, 2696 patients had the inclusion criteria; 1680 (62.3%) people reported long COVID syndrome (LCS). LCS‐associated brain fog was reported by 194 (7.2%) patients. Female sex (odds ratio [OR]: 1.4), respiratory problems at the onset (OR: 1.9), and intensive care unit (ICU) admission (OR: 1.7) were significantly associated with reporting chronic post‐COVID “brain fog” by the patients. In this large population‐based study, we report that chronic post‐COVID “brain fog” has significant associations with sex (female), respiratory symptoms at the onset, and the severity of the illness (ICU admission).
Keywords: brain, coronavirus, COVID‐19, SARS‐CoV‐2
1. INTRODUCTION
It has been more than 18 months since the start of the coronavirus disease‐2019 (COVID‐19) pandemic. While during the early stages of the pandemic people were largely concerned by its risk of death and later on everybody recognized the psychosocial consequences of the pandemic, recently, the postacute phase lingering symptoms of the disease has attracted attention. 1 Many people, who survived the COVID‐19, have reported a variety of persistent signs and symptoms after the acute phase of the disease. 2 The National Institute for Health and Clinical Excellence, the Scottish Intercollegiate Guidelines Network, and the Royal College of General Practitioners have jointly developed the following definition for the post‐COVID syndrome: “signs and symptoms that develop during or after an infection consistent with COVID‐19, continue for more than 12 weeks, and are not explained by an alternative diagnosis.” 3 Some other experts have considered the persistence of symptoms (e.g., fatigue, memory problems, breathlessness, muscle pain that could not be attributed to any other cause) beyond 2 weeks for mild disease, beyond 4 weeks for moderate to severe illness, and beyond 6 weeks for critically ill patients as “long COVID syndrome” (LCS). 4
COVID‐19 may damage the brain in various ways; encephalitis and cerebrovascular accidents have been reported in the literature before. 5 However, other long‐term effects of the COVID‐19 on the brain may be subtler; persistent impairment in sustained attention or cognition are two examples. 6 , 7 , 8 Brain fog is a general term used to describe the feeling of being mentally slow, fuzzy, or spaced out. 9 It affects one's ability to think or concentrate. 10 As physicians, we have been hearing from many survivors of COVID‐19 who are complaining of “brain fog” after recovering from the illness.
In the current study, we investigated the frequency of LCS‐associated brain fog in patients who have survived the COVID‐19. Also, we scrutinized the potential risk factors associated with the development of brain fog in a large cohort of patients with documented COVID‐19 to add to the existing literature.
2. METHODS
2.1. Participants
In this study, all consecutive adult patients (18–55 years of age) who were referred to and admitted at healthcare facilities (55 centers) anywhere in Fars province (located in the south of Iran with a population of 4 851 000 people) from February 19, 2020 (the start date of the pandemic in Iran) 11 to November 20, 2020 were included. All patients had a confirmed COVID‐19 diagnosis by a positive result on real‐time polymerase chain reaction (PCR) testing of nasopharyngeal and oropharyngeal samples.
2.2. Data collection
For all patients, the following data were collected at the emergency room by the admitting physician: sex, age, presence of fever, respiratory distress, muscle pain, cough, change in mental status, loss of smell, dizziness, headache, nausea, vomiting, abdominal pain, and anorexia. Other collected data included: Real‐time PCR test results and admission to a hospital ward (i.e., COVID‐specific ward, intensive care unit [ICU], or others). Underlying chronic health problems were also collected (self‐declared) (i.e., liver, cardiac, renal, or neurological, and also diabetes mellitus [DM], cancer, hypertension [HTN], pulmonary disorders). No data were available about the hospital course of the patients (e.g., laboratory test results, management, complications). However, the outcome was also recorded in the database, as dead or discharged.
In a phone call to the discharged patients (survivors) (made by the last 12 authors), at least 3 months after their acute illness (from 1 to 14 March 2021), we investigated their current health status and obtained their information if they agreed to participate and answer the questions (consented orally—Supporting Information Appendix S1). We randomly selected every other adult patient in our database (sorted by their phone numbers). If someone did not answer, we selected the previous patient in the list (who was skipped initially) (Figure 1).
Figure 1.
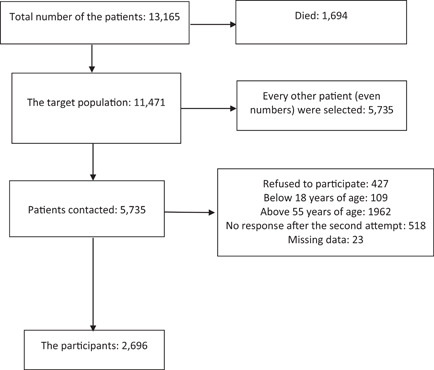
The recruitment process of the study
A questionnaire (Supporting Information Appendix S1) was specifically designed for the purpose of data collection and all the team members were instructed by the first author on how to inquire the data consistently and in the same manner. The first part of the questionnaire (seven questions) collected the demographic data and also some confirmatory data (e.g., date of admission, underlying disorders before COVID‐19) to cross‐check with the primary database. In the second part (20 questions), we asked if the patient has noticed any problems (e.g., self‐declared concentration difficulty) or is suffering from any condition during the past week, compared with their pre‐COVID‐19 condition (any symptoms or complaints or problems that you did not have before your COVID‐19, but have had ever since after your illness and specifically during the past 7 days). For the purpose of this study, we defined LCS as any symptoms or complaints, or problems that the patients did not have before their COVID‐19, but have persistently had for more than 12 weeks (postdischarge) and particularly, during the past 7 days. We specifically asked their complaints during the past 7 days to minimize the risk of recall bias. In the third part of the questionnaire (one question), we asked the patients to compare their “Ability to concentrate and think” with their pre‐COVID‐19 status based on a Likert scale (1. Much worse; 2. Somewhat worse; 3. The same as before; 4; Somewhat better; 5. Much better). For the purpose of this study, we defined “brain fog” as “concentration difficulty” and a worse status in the “ability to concentrate and think” (both items in Supporting Information Appendix S1 should be declared by the patient for securing an increased accuracy of the collected data since it is a subjective feeling).
2.3. Statistical analyses
The frequency of “brain fog” was reported. Kolmogorov–Smirnov normality test was performed. Values were presented as mean ± standard deviation (SD) or median/interquartile range (IQR) (based on their normality) for continuous variables and as number (percent) of subjects for categorical variables. The following variables were selected as the risk factors potentially associated with “brain fog” in LCS: sex (female: male), age, length of hospital stay, respiratory problems at the onset, neurological problems at the onset, gastrointestinal (GI) problems at the onset, pre‐existing chronic medical problems, and ICU admission. Fisher's exact test, t‐test (or Kruskal–Wallis test), and binary logistic regression analysis model were used for statistical analyses (significant variables from univariate analyses were entered into the logistic regression analysis model). Odds ratios and 95% confidence intervals were estimated. A p‐value (two‐sided) less than 0.05 was considered significant.
2.4. Standard protocol approvals
The Shiraz University of Medical Sciences Institutional Review Board approved this study (IR.SUMS.Rec.1399.022).
3. RESULTS
3.1. General characteristics of the patients
From the beginning of the pandemic to November 20, 2020, 13 165 patients with confirmed COVID‐19 were referred to healthcare facilities in the province; 1694 individuals died (case fatality rate: 12.8%) and 11 471 individuals (adults and children) were discharged from the hospitals. In the follow‐up phase of this study, 2696 patients had the inclusion criteria and were studied (Figure 1). The participants included 1474 men (54.7%) and 1222 women (45.3%). Their mean age was 41 years (minimum: 18; maximum: 55; SD: 9 years).
3.2. Clinical characteristics of the patients
Manifestations of COVID‐19 on admission were as follows: respiratory/pulmonary in 2375 (88.1%) (hypoxemia [SpO2 < 93% by pulse oximeter reading] in 50.2%, cough in 55.2%, and chest pain in 4.8%), neurological in 526 (19.5%) (headache in 15.8%, loss of smell in 3.1%, change in mental status in 1.2%), and GI in 432 (16%) patients (nausea in 9.2%, vomiting in 4.9%, and abdominal pain in 3.1%). Some patients had nonspecific manifestations (fever in 42.2% and myalgia in 41.7%). A minority (228 [8.5%]) of the patients needed ICU admission. Duration of the hospital stay was as short as a few hours (in five patients) to as long as 102 days (2 months or more in four patients) (median: 5; IQR: 5). Seven hundred and eight patients (26.3%) had pre‐existing chronic medical conditions (11.9% had HTN, 10.7% had DM, 5.1% had cardiac problems, and 2.5% had asthma).
3.3. Chronic post‐COVID “brain fog”
In total, 1680 (62.3%) people reported chronic symptoms/complaints of LCS. The most common symptoms included: exercise intolerance (619; 23%), fatigue (781; 29%), dyspnea (554; 20.5%), muscle pain (441; 16.4%), sleep difficulty (392; 14.5%), cough (234; 8.7%), brain fog (194; 7.2%), chest pain (130; 4.8%), and loss of smell (129; 4.8%).
3.4. Factors associated with “brain fog”
Table 1 shows factors in association with reporting “brain fog.” Reporting chronic post‐COVID “brain fog” was significantly more frequent among women, those with respiratory problems at the onset of infection, and those who had ICU admission, in univariate analyses. We included these three variables in a regression analysis model. The model that was generated by this test was significant (p = 0·0001) and could predict the occurrence of brain fog in 92.8% of the patients. Table 2 shows these factors in association with reporting chronic post‐COVID “brain fog.” All three factors (i.e., female sex, respiratory problems at the onset of infection, and ICU admission) were significantly associated with reporting chronic post‐COVID “brain fog” by the patients.
Table 1.
Factors in association with reporting “brain fog” in univariate analysis
| Brain fog, N = 194 | No brain fog, N = 2502 | p, df | |
|---|---|---|---|
| Sex (female: male) | 102: 92 (ratio: 1.11) | 1120: 1382 (ratio: 0.81) | 0.036, 1 |
| Age (mean ± SD), years | 42 ± 8 | 41 ± 9 | 0.109 |
| Length of hospital stay (mean ± SD), days | 6·9 ± 4.9 | 6·4 ± 6.1 | 0.221 |
| Respiratory problems at the onset | 180 (93%) | 2195 (88%) | 0.038, 1 |
| Neurological problems at the onset | 40 (21%) | 486 (19%) | 0.707, 1 |
| Gastrointestinal problems at the onset | 27 (14%) | 405 (16%) | 0.417, 1 |
| Pre‐existing chronic medical problems | 56 (29%) | 652 (26%) | 0.398, 1 |
| ICU admissiona | 28 (14%) | 200 (8%) | 0.004, 1 |
Abbreviations: df, degree of freedom; ICU, intensive care unit; SD, standard deviation.
Nine missing values.
Table 2.
Factors in association with reporting “brain fog” in the regression analysis model
| Odds ratio | 95% Confidence interval | p | |
|---|---|---|---|
| Sex (female) | 1.417 | 1.056–1.902 | 0.020 |
| Respiratory problems at the onset | 1.952 | 1.273–2.994 | 0.002 |
| ICU admission | 1.793 | 1.026–3.134 | 0.040 |
Abbreviation: ICU, intensive care unit.
4. DISCUSSION
Publications on long‐lasting COVID‐19 symptoms, the so‐called “long COVID syndrome (LCS),” are increasing very fast, but more should be known about its details and the associated risk factors for various symptoms. 12 In the current study, we observed that 62.3% of the participants reported chronic symptoms/complaints of LCS and 7.2% of these young adults (18–55 years of age) patients with COVID‐19 (requiring a hospitalization) had long‐lasting complaints of brain fog (inability to think and concentrate). In one study of 384 patients (mean age: 59.9 years; 62% male) followed for a median of 54 days postdischarge, 53% of the patients reported persistent breathlessness and 69% reported fatigue. 13 In a study of 478 patients, 244 patients (51%) declared at least one symptom that did not exist before COVID‐19: fatigue was reported by 31% and cognitive symptoms by 21%. 14 In a small study of 29 patients, the authors investigated the frequency and severity of cognitive impairments 3–4 months after COVID‐19 hospital discharge with two cognition test batteries (screen for cognitive impairment in psychiatry danish version and the trail making test‐part B). 15 The percentage of patients with cognitive impairment was more than 59%. 15 In another study of 4182 patients with COVID‐19, individuals self‐reported their symptoms prospectively in the COVID‐symptom study application. A total of 558 (13.3%) participants reported symptoms lasting ≥28 days. 12 The difference in the methodology may explain the different rates of LCS reported by these studies; while active inquiry suggests a prevalence of more than 50% (observed in our study and the reports by Mandal et al. 13 Morin et al. 14 ), passive inquiry suggests a much lower prevalence of LCS. It is likely that in the passive inquiry of the symptoms in the study by Sudre et al. 12 people with no LCS (e.g., with no fatigue or brain fog) more frequently used the designed app to participate. Furthermore, the application of cognition test batteries provides higher percentages of cognitive impairments than that by self‐declaration by the patients. It is noteworthy to mention that persistent COVID‐19‐associated neurocognitive symptoms have also been reported by nonhospitalized patients. 16
Cognitive impairment is associated with lower work function and poorer quality of life in the affected people. 15 Therefore, it should be acknowledged more robustly by the scientific community. Screening of all patients, who have recovered from COVID‐19, for early detection of any cognitive impairment should be considered and the cost‐effectiveness of such a strategy should be investigated in future studies.
In the current study, we observed that female sex, respiratory problems at the onset of infection, and ICU admission were significantly associated with reporting chronic post‐COVID “brain fog” by the patients. In a previous study, LCS was more likely in women. 12 In another study of 599 patients, female sex and ICU admission were independent risk factors for the post‐COVID‐19 syndrome. 17 The reproduced observation that female sex is more often associated with LCS, in general, and brain fog, in particular, is intriguing and should be further explored in future studies. Analysis of the pathophysiological drivers underlying the female sex as a risk factor for LCS is a critical next step. Our observation that ICU admission was significantly associated with experiencing chronic post‐COVID “brain fog” and the observations by Peghin et al. 17 that ICU admission was associated with LCS may suggest that a more severe COVID‐19 at presentation is a significant risk factor for experiencing LCS, in general, and brain fog in particular. Speculatively, we can hypothesize that a more severe COVID‐19 is a risk factor for LCS (including brain fog) because of the following possibilities: 1. Severe COVID‐19 is associated with a more severe immune response and cytokine storm, and, consequently, more organ damage (e.g., brain damage) 18 , 19 , 20 ; 2. Severe COVID‐19 is usually more aggressively treated and is more often associated with iatrogenic harm (e.g., due to intubation or nosocomial infections) with long‐lasting sequelae; 3. Alterations in brain functioning, especially in regions associated with cognition (e.g., cingulate cortex), can result from infection with severe acute respiratory syndrome coronavirus 2. 7 , 21 Therefore, it is reasonable to assume that LCS (including brain fog) is a biological phenomenon. It is also plausible to assume that LCS (including brain fog) is the result of psychosocial consequences of COVID‐19, at least to some extent and in some patients. 22 In one study of 382 patients, abnormalities in functional outcomes, activities of daily living, anxiety, depression, and sleep occurred in over 90% of the patients 6 months after hospitalization for COVID‐19. 23 The biological/psychosocial underpinnings of the symptoms of LCS (including brain fog) should be explored in future studies and the scientific community should propose appropriate treatment strategies to mitigate the long‐lasting cognitive consequences of COVID‐19. 21 , 24 , 25
5. LIMITATIONS
A major limitation of this manuscript is that the study is entirely based on phone consultations, with no clinical, psychological, or biological evaluations. In addition, the data on brain fog were not collected prospectively and we cannot provide the information on the temporal relationship (start date) with COVID‐19 and brain fog based on the current study. Also, we did not investigate those with a mild illness and asymptomatic infections. Furthermore, if we had included additional questions and if we had not limited the study to the past 7 days, the incidence of brain fog could have been different. Finally, our findings are limited by the absence of a control group and of pre‐COVID assessments in this cohort.
6. CONCLUSION
In this large study, we report that chronic post‐COVID “brain fog” has significant associations with sex (female), respiratory symptoms at the onset, and the severity of the illness (ICU admission). The scientific community should investigate the pathophysiology of this condition to discover the biological underpinnings of LCS.
CONFLICT OF INTERESTS
Ali A. Asadi‐Pooya received honoraria from Cobel Daruo, RaymandRad, and Tekaje and Royalty from Oxford University Press (book publication). Other authors have no conflict of interests.
AUTHOR CONTRIBUTIONS
Ali A. Asadi‐Pooya: Conceptualization, data curation, formal analysis, investigation, methodology, project administration, resources, software, supervision, validation, visualization, writing – original draft, and writing – review and editing. Hamid Nemati, Mina Shahisavandi, Ali Akbari, Amir Emami, Mehrzad Lotfi, Mahtab Rostamihosseinkhani, Zohreh Barzegar, Maryam Kabiri, Zahra Zeraatpisheh, Mohsen Farjoud‐Kouhanjani, Anahita Jafari, Sarvin Sasannia, Shayan Ashrafi, Masoume Nazeri, and Sara Nasiri: Data curation, investigation, methodology, project administration, validation, visualization, and writing – review and editing. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Supporting information
Supplementary information.
Asadi‐Pooya AA, Akbari A, Emami A, et al. Long COVID syndrome‐associated brain fog. J Med Virol. 2022;94:979‐984. 10.1002/jmv.27404
DATA AVAILABILITY STATEMENT
The data are confidential and will not be shared as per the regulations of Shiraz University of Medical Sciences.
REFERENCES
- 1. Walitt B, Bartrum E. A clinical primer for the expected and potential post‐COVID‐19 syndromes. Pain Rep. 2021;6:e887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Arnold DT, Hamilton FW, Milne A, et al. Patient outcomes after hospitalisation with COVID‐19 and implications for follow‐up: results from a prospective UK cohort. Thorax. 2021;76:399‐401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Accessed June 18, 2021. https://www.nice.org.uk/guidance/ng188/
- 4. Raveendran AV. Long COVID‐19: Challenges in the diagnosis and proposed diagnostic criteria. Diabetes Metab Syndr. 2021;15:145‐146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Asadi‐Pooya AA, Simani L. Central nervous system manifestations of COVID‐19: a systematic review. J Neurol Sci. 2020;413:116832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Zhou H, Lu S, Chen J, et al. The landscape of cognitive function in recovered COVID‐19 patients. J Psychiatr Res. 2020;129:98‐102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Hugon J, Msika EF, Queneau M, Farid K, Paquet C. Long COVID: cognitive complaints (brain fog) and dysfunction of the cingulate cortex. J Neurol. Published online 2021 June, 18:1‐3. 10.1007/s00415-021-10655-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Orrù G, Bertelloni D, Diolaiuti F, et al. Long‐COVID syndrome? A study on the persistence of neurological, psychological and physiological symptoms. Healthcare (Basel). 2021;9:575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Accessed July 13, 2021. https://www.healthline.com/health/brain-fog/
- 10.Accessed July 13, 2021. https://www.merriam-webster.com/dictionary/brain%20fog/
- 11.Accessed March 14, 2021. https://www.worldometers.info/coronavirus/country/iran/
- 12. Sudre CH, Murray B, Varsavsky T, et al. Attributes and predictors of long COVID. Nat Med. 2021;27:626‐631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Mandal S, Barnett J, Brill SE, et al. "Long‐COVID": a cross‐sectional study of persisting symptoms, biomarker and imaging abnormalities following hospitalisation for COVID‐19. Thorax. 2021;76:396‐398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Morin L, Savale L, Pham T, et al. Four‐month clinical status of a cohort of patients after hospitalization for COVID‐19. JAMA. 2021;325:1525‐1534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Miskowiak KW, Johnsen S, Sattler SM, et al. Cognitive impairments four months after COVID‐19 hospital discharge: pattern, severity and association with illness variables. Eur Neuropsychopharmacol. 2021;46:39‐48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Hellmuth J, Barnett TA, Asken BM, et al. Persistent COVID‐19‐associated neurocognitive symptoms in non‐hospitalized patients. J Neurovirol. 2021;27:191‐195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Peghin M, Palese A, Venturini M, et al. Post‐COVID‐19 symptoms 6 months after acute infection among hospitalized and non‐hospitalized patients. Clin Microbiol Infect. 2021;27:1507‐1513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Sykes DL, Holdsworth L, Jawad N, Gunasekera P, Morice AH, Crooks MG. Post‐COVID‐19 symptom burden: what is long‐COVID and how should we manage it? Lung. 2021;199:113‐119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Guedj E, Campion JY, Dudouet P, et al. 18F‐FDG brain PET hypometabolism in patients with long COVID. Eur J Nucl Med Mol Imaging. 2021;48:2823‐2833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Batabyal R, Freishtat N, Hill E, Rehman M, Freishtat R, Koutroulis I. Metabolic dysfunction and immunometabolism in COVID‐19 pathophysiology and therapeutics. Int J Obes (Lond). 2021;45:1163‐1169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Stefano GB, Ptacek R, Ptackova H, Martin A, Kream RM. Selective neuronal mitochondrial targeting in SARS‐CoV‐2 infection affects cognitive processes to induce “brain fog” and results in behavioral changes that favor viral survival. Med Sci Monit. 2021;27:e930886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Humphreys H, Kilby L, Kudiersky N, Copeland R. Long COVID and the role of physical activity: a qualitative study. BMJ Open. 2021;11:e047632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Frontera JA, Yang D, Lewis A, et al. A prospective study of long‐term outcomes among hospitalized COVID‐19 patients with and without neurological complications. J Neurol Sci. 2021;426:117486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Theoharides TC, Cholevas C, Polyzoidis K, Politis A. Long‐COVID syndrome‐associated brain fog and chemofog: luteolin to the rescue. Biofactors. 2021;47:232‐241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Theoharides TC, Conti P. Be aware of SARS‐CoV‐2 spike protein: there is more than meets the eye. J Biol Regul Homeost Agents. 2021;35:833‐838. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary information.
Data Availability Statement
The data are confidential and will not be shared as per the regulations of Shiraz University of Medical Sciences.


