SUMMARY
Enteric commensal bacteria of food animals may serve as a reservoir of genes encoding antimicrobial resistance (AMR). The genes are often plasmidic. Different aspects of bacterial ecology can be targeted by interventions to control plasmid-mediated AMR. The field efficacy of interventions remains unclear. We developed a deterministic mathematical model of commensal Escherichia coli in its animate and non-animate habitats within a beef feedlot's pen, with some E. coli having plasmid-mediated resistance to the cephalosporin ceftiofur. We evaluated relative potential efficacy of within- or outside-host biological interventions delivered throughout rearing depending on the targeted parameter of bacterial ecology. Most instrumental in reducing the fraction of resistant enteric E. coli at steer slaughter age were interventions acting on the enteric E. coli and capable of either ‘plasmid curing’ E. coli, or lowering maximum E. coli numbers or the rate of plasmid transfer in this habitat. Also efficient was to increase the regular replacement of enteric E. coli. Lowering replication rate of resistant E. coli alone was not an efficient intervention target.
Key words: Antimicrobial resistance, cattle, ceftiofur, cephalosporin, enteric commensal bacteria, Escherichia coli
INTRODUCTION
The enteric commensal bacteria of food animals may act as a reservoir of antimicrobial resistance (AMR) genes, which are often plasmidic [1,2]. Escherichia coli constitutes up to 87% of the culturable Gram-negative faecal bacteria in cattle [3], and it is proposed as an indicator organism for the extent of resistance of enteric bacteria to antimicrobials [4]. Resistance of E. coli in cattle in the USA to the cephalosporin ceftiofur has been most often mediated by the plasmidic gene blaCMY-2 [5]. Containing resistance to ceftiofur is essential for maintaining veterinary treatment options, as it is the only third-generation cephalosporin licensed for therapeutic use in cattle in the USA. From the public health perspective, if zoonotic transmission may occur, bacteria resistant to ceftiofur are also resistant to cephalosporins used in human medicine [6]. This is also a well-studied example of predominantly plasmid-mediated AMR in the cattle enteric commensals [5]. Both ceftiofur-sensitive and -resistant (bearing blaCMY-2 plasmid) enteric E. coli of cattle are genetically diverse rather than clonal [5, 7, 8]. The plasmids are passed vertically during cell divisions [9, 10], and transferred horizontally via conjugation between bacteria [5]. E. coli inhabits not only the enteric but also various non-animate habitats [11]; the bacterial populations bear the same types of blaCMY-2 plasmids [12], and plasmid transfer occurs, albeit at different rates, in the enteric [13] and non-animate habitats [14, 15]. Other plasmidic genes have recently been recognized as mediators of ceftiofur resistance in E. coli of cattle in the USA [16].
Some compounds have been proposed for application in food animals to limit AMR spread in their enteric bacteria. In vitro, bambermycin antibiotics modify the plasmid transfer rate among Enterobacteriaceae with the effect's direction (enhanced or reduced rate) dependent on the plasmid [17], and reduce net growth of plasmid-bearing bacteria [17, 18]. These effects may be sustained in vivo; in-feed bambermycin administration reduces the frequency and degree of AMR in faecal E. coli in cattle [19] and pigs [20], with little or no decrease in total E. coli numbers [19, 20]. The exact molecular mechanism of action of bambermycins is unknown; it has been hypothesized that pili expressed by plasmid-bearing bacteria attempting to conjugate serve as the drug attachment point [21]. Constructed bacteriophages may be another tool for reducing plasmid transfer or killing plasmid-bearing bacteria; that certain phages selectively infect bacteria expressing pili and are thereby able to alter the conjugation rate has been known for some time [22–25], and is being revisited [26, 27]. Expressing these effects in terms of the parameters of bacterial ecology, bambermycins or phages may be altering the plasmid transfer rate, alone or with killing the plasmid-donor cells, or reducing the rate of replication of the resistant bacteria.
Inducing plasmid loss, termed ‘plasmid curing’ [28, 29], has been also proposed as an intervention target (see Trevors [28] for a list of plasmid-curing compounds and procedures). In terms of the bacterial ecology parameters, the effect of plasmid curing is that the resistant cells lose the plasmids and join the pool of antimicrobial-sensitive E. coli at a certain rate.
The size of the realized niche for E. coli among enteric bacteria can be thought of as another intervention target. The niche size may be altered by introducing competitive microflora that are less likely to carry AMR genes (similarly, certain direct-fed microbial strains reduce the number of E. coli O157 in beef cattle faeces [30]), or by changing the animal's diet [31]. In non-animate habitats, E. coli numbers may be altered by water sanitation or heat-treatment of the feed. In terms of the bacterial ecology parameters, these interventions would reduce the maximum possible number of E. coli.
Another control option may be offered through exploiting E. coli movement between its animate and non-animate habitats. Although it is generally perceived that enteric bacterial communities are relatively stable, field studies have shown that at the same time E. coli circulates readily between the habitats. A variable fraction of faecal E. coli in beef cattle is composed of strains ingested the day before [32]. In another experiment, a fed E. coli strain constituted 10–50% of the faecal coliforms for 10 days and a lesser fraction for 4 months post-ingestion, and was recovered in high numbers from the cattle's bedding and drinking water [33]. E. coli strains may differ in their propensity to appear in faeces following ingestion [32, 33]. Field experiments are yet to address how the partial replacement of enteric E. coli with that ingested and its circulation between the animate and non-animate habitats impact the frequency of AMR. In principle, this parameter of the replacement rate of enteric E. coli may be altered by changing the animal's diet.
In summary, there are several potentially alterable parameters of bacterial ecology that can be targeted by biological interventions to control plasmid-mediated AMR in the cattle enteric commensals. Mathematical modelling is a well-suited method to quantitatively compare the potential efficacy of interventions so that existing intervention approaches can be tailored to, or new approaches can be crafted upon the most promising targets. Here we compared the potential intervention efficacy depending on the targeted parameter in reducing the fraction of resistant enteric E. coli, as an exemplar enteric commensal, in beef cattle by the end of feedlot rearing. We considered interventions within the enteric and non-animate habitats that were targeted to modify the rate of AMR plasmid transfer, the replication rate of resistant E. coli or maximum number of E. coli, to kill the plasmid-donor cells, or to induce plasmid-curing of resistant E. coli. Further, we considered interventions that would change the rate of regular partial replacement of enteric E. coli, or lower maximum E. coli numbers or the frequency of resistant E. coli in animal water or feed supply. Cross-habitat interventions acting similarly on E. coli in water or feed and in the enteric habitat were also considered. Each intervention was delivered consistently throughout the feedlot rearing period.
METHODS
To assess the relative efficacy of the interventions, we developed a deterministic mathematical model of commensal E. coli in a beef feedlot's pen throughout rearing of a group of steers from age 6 to 18 months. For this, we first combined a model of dynamics of ceftiofur-sensitive and -resistant E. coli in the beef cattle large intestine [34], with a model of the dynamics of E. coli in the pen's non-animate habitats: water in troughs, feed in bunks and the rest of the environment [35]. We further developed the resulting model to account for (a) change in the volumes of consumption and faecal deposition by the animals as their body mass grows, (b) E. coli numbers and fractions of resistance in animal water and feed supplies, (c) fractions of resistance in the enteric and pen's non-animate habitats at the start of rearing, (d) retention time of steers' ingesta as it passes through the intestinal tract to the large intestine, and (e) plasmid transfer in E. coli in the non-animate habitats. The model is generally applicable to plasmid-mediated resistance to any antimicrobial. Thereafter, although we use parameter estimates for blaCMY-2 plasmids, we simply refer to plasmid-mediated AMR in commensal E. coli of cattle.
Model assumptions
-
(1)
The habitats of commensal E. coli in a beef feedlot's pen are the large intestines of the cattle, water in troughs, feed in bunks, and the rest of the environment (faecal–soil mix on the floor, other non-animate objects and animal hides). E. coli move between the habitats with substances, e.g. water consumption by cattle or their faecal deposition onto the floor. Transmission of E. coli between the intestines occurs via the other habitats.
-
(2)
In each habitat, population growth of E. coli is density-dependent being restricted by the maximum possible number of E. coli.
-
(3)
E. coli resistance to ceftiofur is plasmid-mediated; AMR plasmids are passed vertically during cell division, and transferred horizontally from resistant to sensitive cells via conjugation.
-
(4)
Bearing AMR plasmid imposes a fitness cost that manifests as a reduction in the net rate of replication of resistant cells compared to sensitive ones [9].
-
(5)
There is a continuous fractional replacement of enteric commensal E. coli with equal rates of bacteria out-flow in faeces and in-flow from ingestion [34].
-
(6)
Steers were not administered ceftiofur therapy or any other compound, e.g. in-feed antimicrobials, neither was any other intervention affecting commensal E. coli delivered to the animals or within the pen's non-animate habitats during the rearing period (except for the targeted interventions modelled).
Model description
In the absence of intervention
A schematic diagram of the model of commensal E. coli in its habitats within a feedlot's pen is given in Figure 1, and a detailed model diagram scaled per ml of matter is given in Figure 2. For the matter in each habitat, a weight-to-volume ratio of 1 was assumed. Because the focus was bacterial ecology, the unit of time for model parameterization was 1 hour.
Fig. 1.
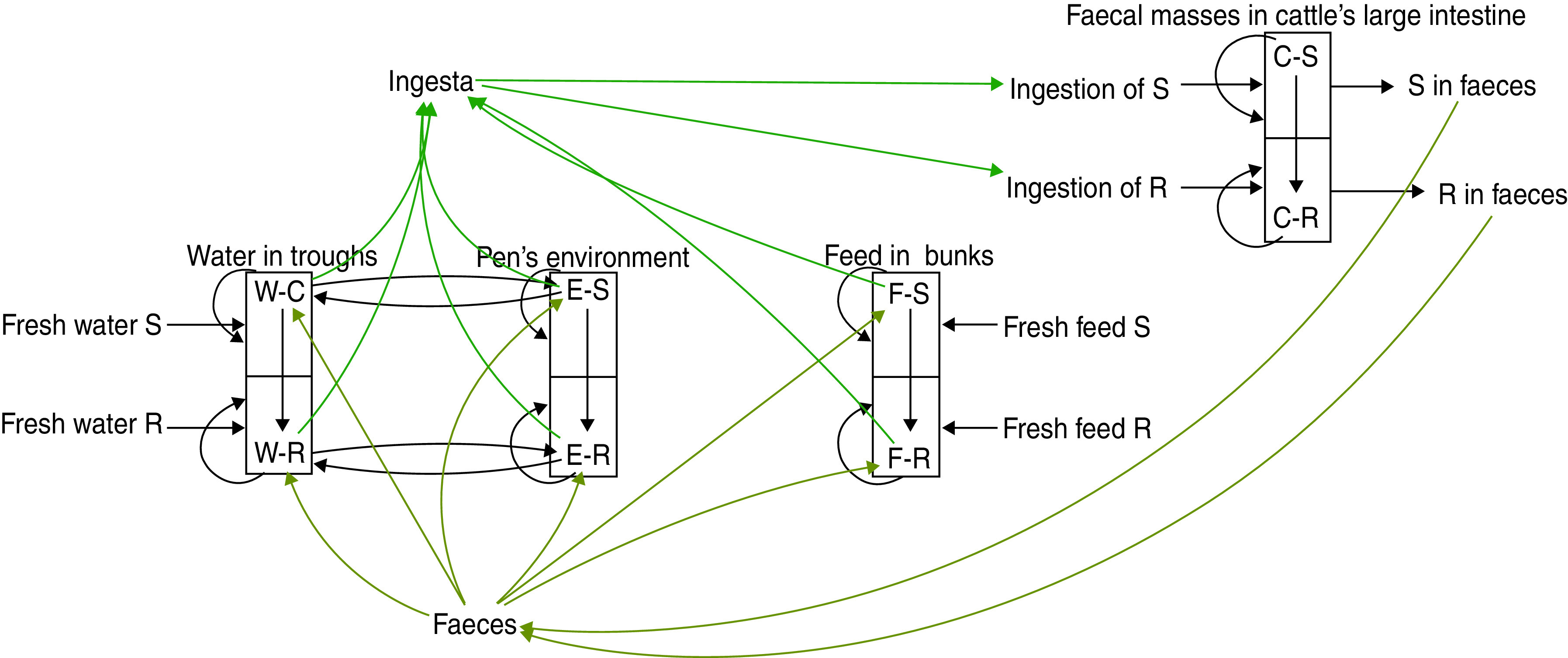
[colour online]. Schematic diagram of the model of commensal E. coli in the within-pen habitats. C, Enteric; W, water in troughs; F, feed in bunks; E, the rest of the environment. S, R, the number of sensitive (S) and resistant (R) E. coli. The arrows show bacterial flows with green for those related to ingesta, yellow for those related to faeces, and black for all others including bacterial population growth and the flow of sensitive cells becoming resistant upon acquisition of plasmids carrying genes of antimicrobial resistance.
Fig. 2.
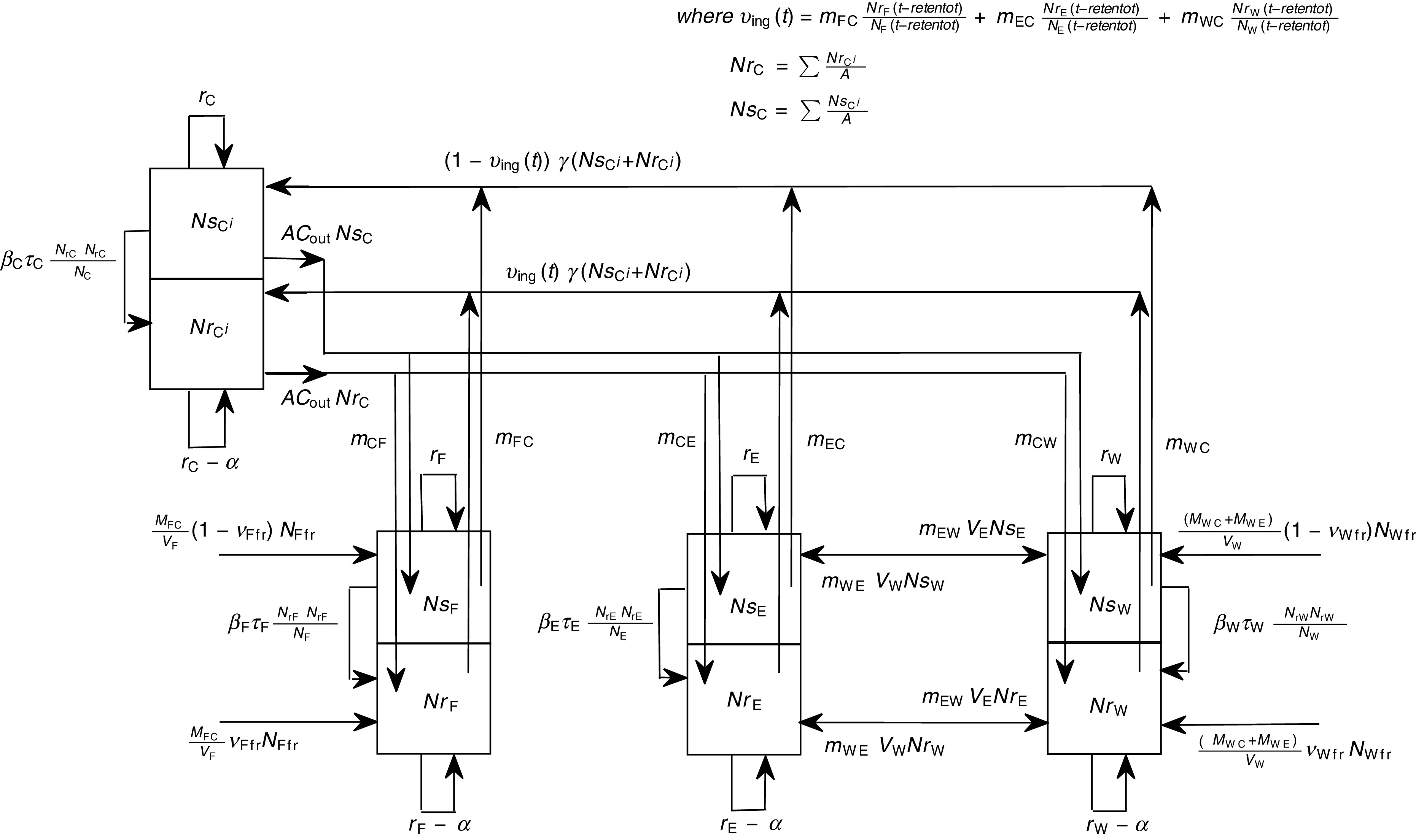
Detailed diagram and parameters of the model of commensal E. coli in within-pen habitats scaled per ml of matter. The parameters are defined in Table 1.
Definitions and values of parameters for the animals and bacterial ecology are given in Table 1. Let A denote the number of steers in the pen. Body weight (bw) of a steer at a given time is bw(t). Volume of faeces deposited by an animal per kg of bw per unit time was faeces_dep, with total volume of faeces deposited Cout=bw(t)*faeces_dep. All animals deposited Cout*A volume of faeces per unit time, of which fraction mCj was deposited into non-animate habitat j.
Table 1.
Definitions and values of parameters for the animals and bacterial ecology. The model was simulated for a 12-month feedlot rearing period.
| Parameter | Definition, units | Base value and distribution | References |
|---|---|---|---|
| Animals, and matter movement | |||
| A | Number of steers | 150 | [35] |
| bw(t) | Body weight (kg) | — | Approximated with a model [38] |
| faeces_dep | Faeces production (ml/kg body weight per h) | 0·992 | Estimated from [46, 47] |
| mCW | Fraction of faeces shed to water | 0·05 | [35] |
| mCF | Fraction of faeces shed to feed | 0·02 | [35] |
| mCE | Fraction of faeces shed to environment | 0·93 | [35] |
| consum_water | Consumption of water (ml/kg bw per h) | 5·11 | Estimated from [48] |
| consum_feed | Consumption of feed (g/kg bw per h) | 1·10 | Estimated from [49] |
| consum_env | Consumption of environment (g/kg bw per h) | 0·08 | Estimated from [35] |
| mWC | Water as fraction of consumption | 0·8124 | Calculated |
| mFC | Feed as fraction of consumption | 0·1749 | Calculated |
| mEC | Environment as fraction of consumption | 0·0127 | Calculated |
| reten_rumen | Ingesta retention time in rumen (h) | 36 | Estimated from [39] |
| reten_rumentolgin | Ingesta retention time from rumen to large intestine (h) | 12 | Estimated from [39] |
| mfrEW | Fractional spillage of environment into water (per h) | 0·0002 | [35] |
| mfrWE | Fractional spillage of water into environment (per h) | 0·0042 | [35] |
| E. coli populations | |||
| υstart | Starting fraction of resistant E. coli in within-pen habitats | 0·02, uniform (0·01, 0·03) | Assumed |
| α | Fitness cost, fractional decrease in net replication rate for resistant E. coli | 0·05, uniform (0·025, 0·075) | Estimated from [9] |
| ζj | Maximum fractional plasmid curing resistant E. coli in habitat j (per h) | 1 | Intervention target |
| τj | Probability of completed conjugation per contact between sensitive cell and plasmid-donor cell in habitat j | 1 | Intervention target |
| E. coli in large intestine | |||
| rC | Net replication rate (per h) | 0·17, uniform (0·085, 0·255) | [34] |
| NmaxC | Maximum E. coli (log c.f.u./ml) | 5·50, log-uniform (4·95, 6·05) | [34] |
| βC | Plasmid transfer term (log/h) | −2·70, log-uniform (−2·97, −2·43) | [34] |
| γ | Fractional in-flow/out-flow (per h) | 0·0100, uniform (0·0050, 0·0150) | [32, 34] |
| E. coli in water | |||
| rW | Net replication rate (per h) | 0·14, uniform (0·07, 0·21) | [35] |
| NmaxW | Maximum E. coli (log c.f.u./ml) | 5·00, log-uniform (4·50, 5·50) | Assumed equal to nmaxE |
| βW | Plasmid transfer term (log/h) | −4·00, log-uniform (−4·40, −3·60) | [37] |
| VW | Volume (l) | 50 | [35] |
| NWfr | Number of E. coli in water supplied (log c.f.u./ml) | 3·00, log-uniform (2·70, 3·30) | [11] |
| υWfr | Fraction of resistant E. coli in water supplied | 0·02, uniform (0·01, 0·03) | Assumed |
| E. coli in feed | |||
| rF | Net replication rate (per h) | 0·02, uniform (0·01, 0·03) | [35] |
| NmaxF | Maximum E. coli (log c.f.u./ml) | 4·00, log-uniform (3·60, 4·40) | [35] |
| βF | Plasmid transfer term (log/h) | −6·00, log-uniform (−6·60, −5·40) | Assumed |
| VF | Volume (l) | 2000 | [35] |
| NFfr | Number of E. coli in feed supplied (log c.f.u./ml) | 3·00, log-uniform (2·70, 3·30) | Assumed equal to NWfr |
| υFfr | Fraction of resistant E. coli in feed supplied | 0·02, uniform (0·01, 0·03) | Assumed |
| E. coli in pen's environment | |||
| rE | Net replication rate (per h) | 0·05, uniform (0·025, 0·075) | [35] |
| NmaxE | Maximum E. coli (log c.f.u./ml) | 5·00, log-uniform (4·5, 5·50) | [35] |
| βE | Plasmid transfer term (log/h) | −4·00, log-uniform (−4·40, −3·60) | Assumed equal to βW |
| VE | Volume (l) | 500 | [35] |
Volume of water consumed by an animal from the troughs per kg of bw per unit time was consum_water, of feed consumed from the bunks was consum_feed, and of the pen's environment was consum_env, with total volume of consumption Cin=bw(t)* consum_water+bw(t)*consum_feed+bw(t)*consum_env. Fraction of Cin coming from non-animate habitat j was denoted mjC (e.g. mwc=consum_water*bw(t)/Cin). Hence, total volumes of matter moving between the enteric and non-animate habitats at a given time, t, were:
Faecal contamination of water: MCW=ACoutmCW.
Faecal contamination of feed: MCF=ACoutmCF.
Faecal contamination of environment: MCE=ACoutmCE.
Water consumption by cattle: MWC=ACinmWC.
Feed consumption by cattle: MFC=ACinmFC.
Ingestion of environment by cattle: MEC=ACinmEC.
Volume of non-animate habitat j was Vj. For matter movement between the non-animate habitats per unit time, mfrjk was fractional spillage of j into k, with Mjk the total volume of matter spilled. Total volumes of matter moving between the non-animate habitats at a given time were:
Contamination of water by environment: MEW=VEmfrEW.
Water spillage into environment: MWE=VWmfrWE.
In habitat j, Nsj was number of antimicrobial sensitive, Nrj the number resistant, and Nj the total number of E. coli/ml. The net rate of E. coli replication was rj; the density-dependent population growth was restricted by the maximum possible number of E. coli/ml, Nmaxj. Fitness cost to E. coli bearing AMR plasmid was a fractional reduction α in rj. The frequency-dependent [36] plasmid transfer rate from resistant to sensitive E. coli was βjτj(NrjNsj/Nj) per unit time, where the plasmid transfer term βj can be thought of as the rate of contact of sensitive cells with resistant cells attempting to conjugate and τj as probability of completed conjugation per contact [in the absence of intervention, τj = 1 and ‘force of transfer’ per sensitive cell per unit time = βj(Nrj/Nj)].
For animal water and feed supplies, NWfr and NFfr denoted total number of E. coli/ml, and υWfr and υFfr fractions of resistant E. coli, respectively.
The ordinary differential equations (1)–(6) describe the change in Nsj and Nrj at a given time in the pen's non-animate habitats in the absence of intervention:
Water in troughs
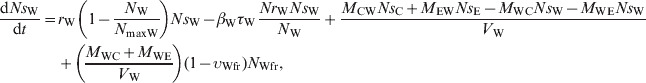 |
(1) |
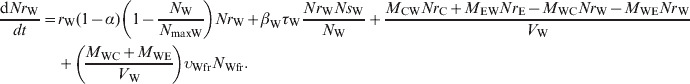 |
(2) |
Feed in bunks
| (3) |
| (4) |
Environment
| (5) |
| (6) |
In the large intestine, E. coli in-flow and out-flow were at equal fractional rates, γ [34]. Of in-flowing E. coli, a fraction υing(t) was resistant, being equal to that in E. coli ingested reten_tot hours earlier. The reten_tot was the sum of durations of ingesta retention in the rumen, reten_rumen, and between the rumen and large intestine, reten_rumentolgin. While t, the steer's time in the feedlot, was 0<t⩽reten_tot, the fraction of resistant enteric E. coli in in-flow, υing(t), was equal to that in ingesta consumed by animals prior to their arrival at the feedlot; the latter was assumed to be constant and equal to υstart. The ordinary differential equations (7) and (8) describe the change in NsCi and NrCi at a given time in faecal masses in the large intestine of steer i in the absence of intervention while 0<t⩽reten_tot:
| (7) |
| (8) |
Equation (9) describes υing(t) when t > reten_tot:
| (9) |
The delay differential equations (10) and (11) describe the change in NrCi and NrCi at a given time in faecal masses in the large intestine of steer i in the absence of intervention when t > reten_tot:
| (10) |
| (11) |
The values of variables NrW, NW, NrF, NF, NrE and NE from the solutions of equations (1)–(6), while 0<t⩽reten_tot were used as the initial values to calculate υing(t) for equations (10) and (11).
As each animal contributed the same amount Cout to the total faecal deposits, the number of antimicrobial-sensitive E. coli/ml of the deposits was 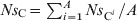 and the number of resistant E. coli was
and the number of resistant E. coli was 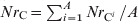 .
.
In the presence of intervention
Definitions and values of parameters reflecting the targeted effects of interventions are given in Table 2. For an intervention in habitat j, Rj denotes fractional reduction in net rate of replication of resistant E. coli. Tj denotes fractional reduction in probability of completed conjugation per contact between sensitive cell and plasmid-donor cell due to death of the latter, and Bj is the fractional reduction in the plasmid transfer rate via any other mechanism. Kj is the fractional reduction in maximum number of E. coli. Pj is the fractional rate of plasmid-curing resistant E. coli (the sensitized cells did not differ from the originally sensitive cells in replication rate or propensity to receive plasmids), with ζj the maximum rate of 1 per hour. The change in rates of fractional in-flow and out-flow of enteric E. coli was modelled by varying the corresponding parameter, γ [34]. VWfr denotes a fractional decrease in υWfr, and VFfr in υFfr.
Table 2.
Definitions and values of parameters representing targeted effects of interventions. Examples of existing and potential biological intervention approaches that have been shown, based on empirical data for E. coli in vitro or enteric E. coli in vivo, or are hypothesized to produce such effects
| Parameter | Definition | Distribution tested | Example (see Introduction for references) |
|---|---|---|---|
| Tj | Fractional kill of resistant E. coli attempting to conjugate in habitat j (including reduced probability of completed conjugation per contact between sensitive cell and plasmid-donor cell) | 0, Uniform (0, 1) | Bambermycins, bacteriophages |
| Bj | Fractional decrease in plasmid transfer in habitat j (without kill) on log scale | 0, Uniform (0, 1) | Bacteriophages |
| Rj | Fractional decrease in resistant E. coli replication in habitat j | 0, Uniform (0, 1) | Exploiting AMR fitness cost; bambermycins |
| Kj | Fractional decrease in maximum number of E. coli in habitat j on log-scale | 0, Uniform (0, 0·4*) | Direct-fed microbial products, animal diet |
| Pj | Fractional plasmid curing resistant E. coli in habitat j | 0, Uniform (0, 0·2*) | Elevated temperature, dyes acridine orange and ethidium bromide, antiprotozoal quinacrine, coumarin antimicrobial coumermycin, surfactant sodium dodecyl sulfate |
| γ | Fractional in-flow/out-flow of enteric E. coli per hour, as a target | 0·0100, Uniform (0·0042, 0·0375) | Animal diet; exploiting E. coli strain differences |
| KW(F)fr | Fractional decrease in number of E. coli in water (feed) supplied on log-scale | 0, Uniform (0, 1) | — |
| VW(F)fr | Fractional decrease in fraction of resistant E. coli in water (feed) supplied | 0, Uniform (0, 1) | — |
AMR, Antimicrobial resistance.
aFurther increasing the parameter value for an intervention in the enteric habitat did not lead to a further reduction in the number of resistant enteric E. coli.
The ordinary differential equations (12)–(17) describe the change in Nsj and Nrj at a given time in the pen's non-animate habitats in the presence of intervention:
Water in troughs
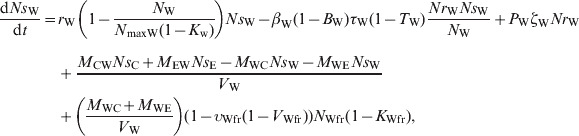 |
(12) |
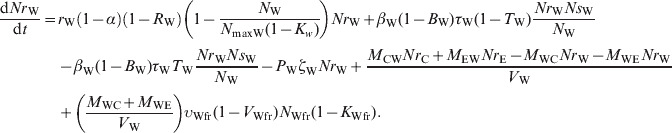 |
(13) |
Feed in bunks
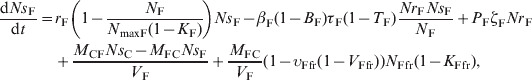 |
(14) |
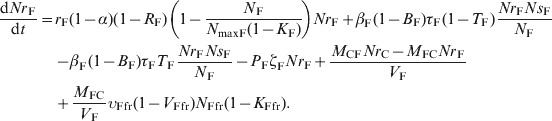 |
(15) |
Environment
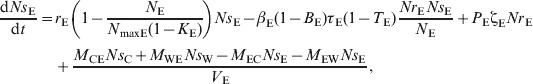 |
(16) |
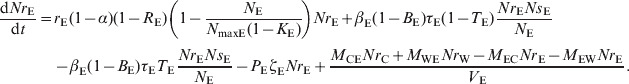 |
(17) |
The ordinary differential equations (18) and (19) describe the change in NrCi and NrCi at a given time in faecal masses in the large intestine of steer i in the presence of intervention while 0<t⩽reten_tot:
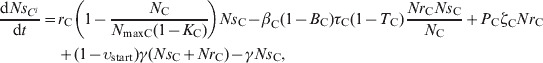 |
(18) |
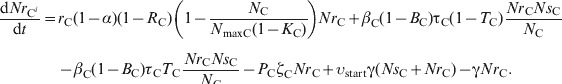 |
(19) |
The delay differential equations (20) and (21) describe the change in NrCi and NrCi at a given time in faecal masses in the large intestine of steer i in the presence of intervention when t > reten_tot[the values of variables NrW, NW, NrF, NF, NrE and NE from the solutions of equations (12)–(17) while 0<t⩽reten_tot were used as the initial values to calculate υing(t) for equations (20) and (21)]
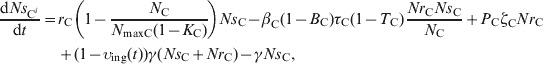 |
(20) |
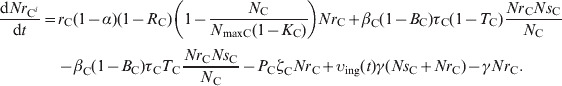 |
(21) |
Model parameterization
The base parameter values were adopted or estimated from the published literature (Table 1) with the following exceptions. The number of E. coli in animal feed supply was taken to be equal to a value reported in the literature for watersheds of grazed cattle pastures and feedlots [11]. The plasmid transfer term (thought of as the rate of contact of sensitive cells with resistant cells attempting to conjugate) in E. coli in the pen's environment was taken to be equal to a reported value for water [37]. A lower term was assumed for transfer in feed, because of a likely larger degree of separation of bacteria in this matrix.
Steer body weight, bw(t), was approximated with a published model of body weight of male beef cattle by day of rearing [38]. The durations of ingesta retention in the rumen, and between the rumen and large intestine, were estimated as the averages of those reported in beef steers for high-concentrate grain-based diets [39].
Low fractions of E. coli with blaCMY-2 plasmids are present on cattle farms even in the absence of ceftiofur use [32, 40]. The plasmidic genes coding for cephalosporin resistance are also present in E. coli in surface and drinking water [41–43]. Reflecting these field data, model simulations were started, at the time of steer placement into the pen, with a base 2·0% fraction of resistant E. coli in all four within-pen habitats, υstart, as well as in fresh water, υWfr, and feed, υFfr, supplies. The model was simulated with 150 steers reared in the pen for 360 days from age 6 to 18 months.
Model solutions and analysis
Solutions of model equations were approximated numerically using the embedded fourth-order Runge–Kutta method implemented in Vensim® PLE Plus software (Ventana Systems Inc., USA). The model analysis included three steps:
Step 1. Sensitivity of the fraction of resistant enteric E. coli and of the fraction of resistant E. coli in steers' ingesta by slaughter age (by the end of 12-month feedlot rearing) to variability in parameter values of bacterial ecology (Table 1, parameters of E. coli populations) in the absence of intervention was evaluated. For this, 200 model simulations were performed with the parameter values obtained by Latin hypercube sampling [44]. Due to a paucity of empirical data on the parameters' distributions, each was assumed to follow a uniform (with boundaries at ±50% of the base value) or a log-uniform (with boundaries at ±10% of the base value on log-scale) distribution (Table 1). With the simulation output, a multiple linear regression model with standardized regression coefficients was built for each outcome as the dependent variable and the parameters as candidate predictors. The regression models were built in SAS® v. 9·1 software for Windows (SAS Institute Inc., USA), and used partial type III sum of squares to test the significance of association between the predictor values and dependent variable given the effects of the other predictors. Backward elimination was the predictor selection routine with the 0·05 significance level to stay in the model. Additionally, sensitivity of the final fractions of resistance to the starting fraction, υstart (in the range 0·00001–0·5), was investigated.
Step 2. Intervention parameters Rj, Kj, Bj, Tj, Pj, VWfr or VWfr were introduced into the mathematical model one at a time, and taken to follow a uniform (0, 1) (except for Kj and Pj; see Table 2). In another simulation, parameter γ was sampled with the boundaries corresponding to a 10% and 90% daily replacement of enteric E. coli (Table 1). Cross-habitat interventions, acting on the same parameter in E. coli in water in troughs or feed in bunks and in the intestines, were also simulated. When simulating each intervention, the rest of the parameters' distributions were set and 100 model simulations performed as in step 1. With the simulation output, for each intervention parameter and each of the two outcome resistance fractions: (a) relationships were explored graphically; (b) correlation, given outcome variability introduced by the other parameters of bacterial ecology, was estimated with Spearman's correlation coefficient (ρ) and considered significant if P ⩽ 0·05; and (c) a multiple linear regression model was built as in step 1 to check that the intervention was significantly associated (P ⩽ 0·05) with the outcome after accounting for the effects of the other parameters of bacterial ecology.
Step 3. For each significant intervention target identified in step 2, uncertainty in the effects on the outcome given variability introduced by the other parameters of bacterial ecology was explored graphically with a box-and-whisker plot. The data for each value of the intervention parameter were obtained with the rest of the parameters' distributions set and 100 model simulations performed as in step 1. Figures were made in SigmaPlot™ (Systat Software Inc., USA).
RESULTS
Fraction of resistant E. coli in the absence of intervention
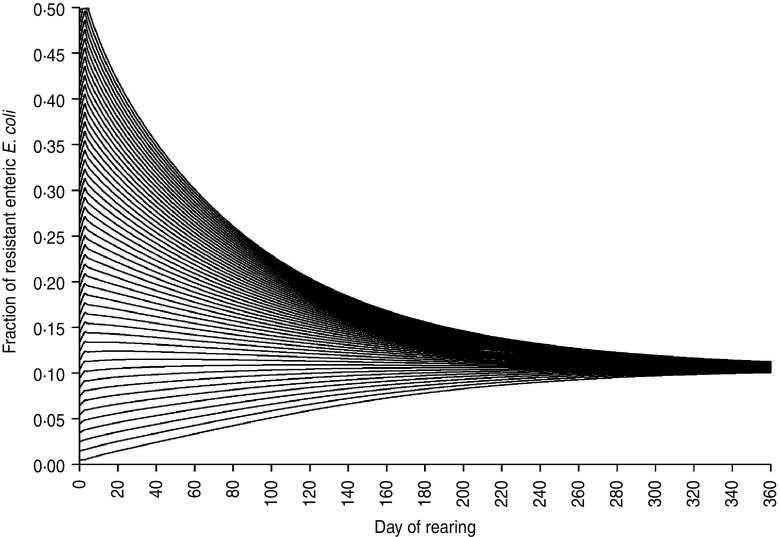
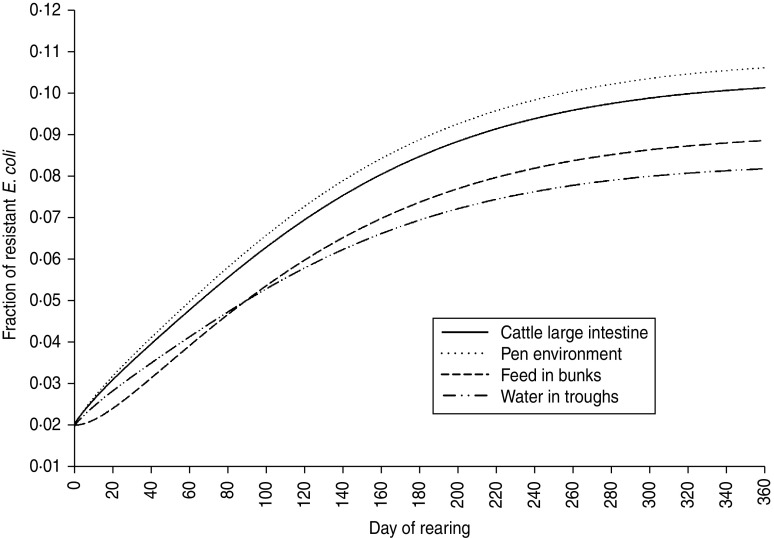
The fraction of resistant enteric E. coli by the end of feedlot rearing did not depend on the starting fraction (Fig. 3), as long as at least some AMR-plasmid-bearing E. coli (e.g. 0·001% of total cells) were present at the start. Equilibrium of the fraction of resistant E. coli was reached in a 12-month rearing period; this was independent of the starting fraction of resistance (Fig. 3). In the base scenario with 2% of resistant E. coli at the start, the fractions of resistance in all the habitats steadily rose throughout the rearing period (Fig. 4). Beyond the 5th day of rearing, the fraction of resistant E. coli in the pen's environment was slightly higher than that in enteric E. coli, where it rose from 2·0% at steer placement to 10·1% at slaughter age (Fig. 4). In terms of uncertainty in the outcome in the absence of intervention, of 200 model simulations, 48% produced the final fraction of resistant enteric E. coli of ⩽0·10, with a wide distribution of the outcome across the other half of simulations (Fig. 5 a). This outcome was positively associated with rate of plasmid transfer and maximum possible E. coli numbers in the enteric habitat, and less so with fraction of resistant E. coli in the water supplied (Fig. 5 c). The outcome was negatively associated with rate of replacement of enteric E. coli and E. coli numbers in fresh water and, to a lesser extent, feed supplies (Fig. 5 c). The final fraction of resistant enteric E. coli was not highly sensitive to the total ingesta retention time prior to the large intestine, varying between 10 and 100 hours (data not shown).
Fig. 3.
Fraction of resistant enteric E. coli throughout the 12-month feedlot rearing period depending on the starting value.
Fig. 4.
Model solutions with base parameter values. Fractions of resistant commensal E. coli in the within-pen habitats throughout the 12-month feedlot rearing period.
Fig. 5.
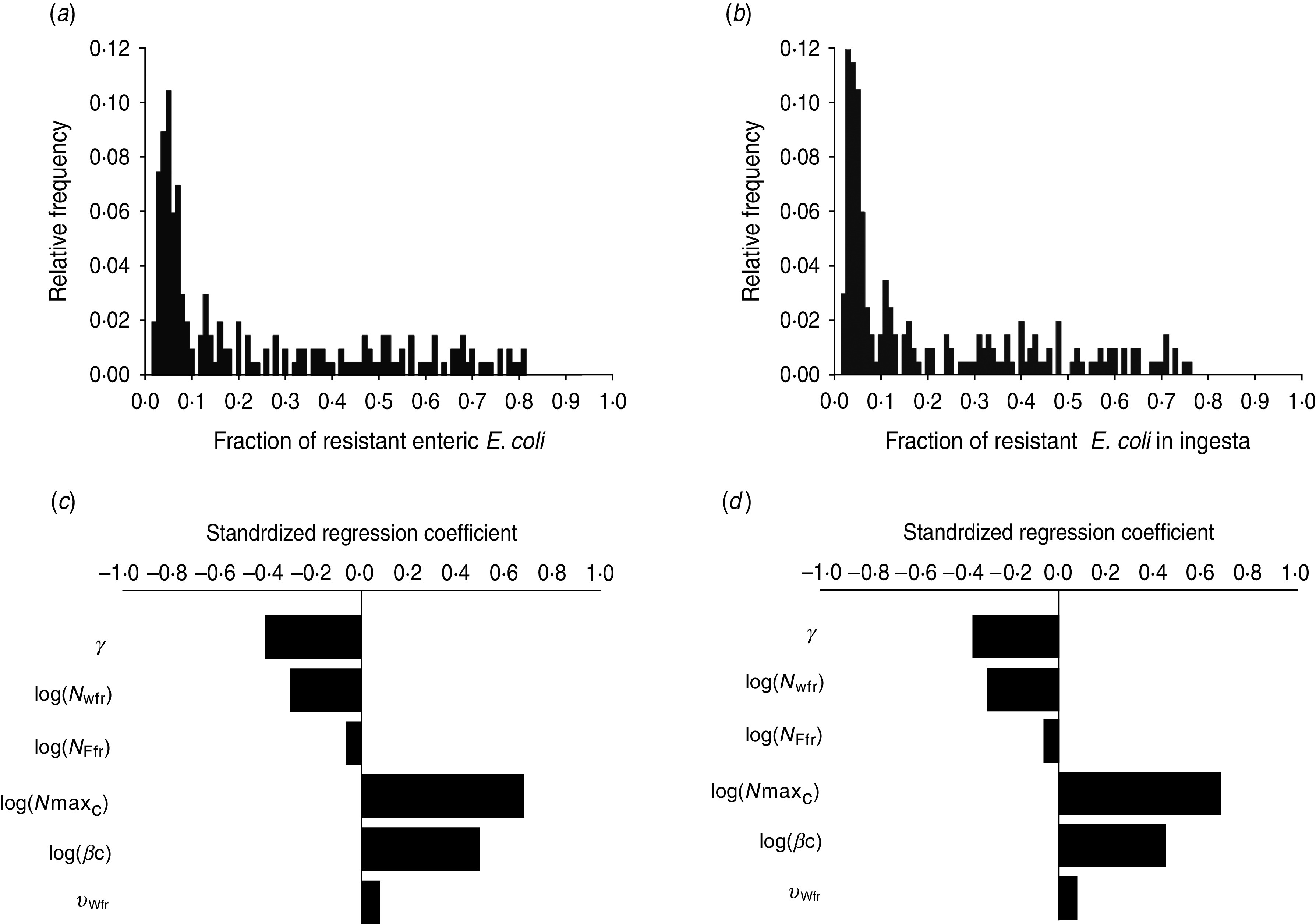
Uncertainty in and sensitivity of the outcomes to variation in the parameters of bacterial ecology in the absence of intervention. Uncertainty: fraction of resistant (a) enteric E. coli or (b) E. coli in ingesta by the end of feedlot rearing (over 200 model simulations). Sensitivity: standardized regression coefficients for parameters (P ⩽ 0·05) in the multiple linear regression model where the dependent variable was the fraction of resistant (c) enteric E. coli or (d) E. coli in ingesta by the end of rearing. Parameters: γ, fractional regular replacement rate of enteric E. coli; log(NWfr), log(NFfr), log c.f.u. E. coli/ml in fresh water and feed supplied, respectively; log(NmaxC), log(βC), maximum log c.f.u. E. coli/ml and the rate of plasmid transfer in the enteric habitat, respectively; υWfr, fraction of resistant E. coli in fresh water supplied.
We then considered if any additional parameters influenced the fraction of resistant E. coli in the steer's ingesta by the end of the feedlot rearing period, given the build-up of resistance in the pen's non-animate habitats (Fig. 4). Uncertainty in this outcome was similar to that in the final fraction of resistant enteric E. coli (Fig. 5 b). The fraction of resistant E. coli in ingesta was also associated with the same parameters of bacterial ecology (Fig. 5 d).
Potential efficacy of interventions
The efficacy of interventions delivered throughout feedlot rearing in reducing the fraction of resistance in enteric E. coli or in E. coli in the steer's ingesta by the end of rearing varied depending on the targeted parameter of bacterial ecology (Fig. 6). Twenty-one interventions targeting a single parameter in a given E. coli habitat, along with four acting on the same parameter in water in troughs and intestines or feed in bunks and intestines, and four acting on E. coli in water or feed supplies were evaluated. Six one-habitat interventions were significantly correlated with a reduction in the final fractions of resistance after accounting for the effects of the other parameters of bacterial ecology. Five of these six acted on enteric E. coli (BC, TC, PC, KC, y); the magnitude of the effects was not sizably enhanced by simultaneous action on E. coli in water or feed (Fig. 6). One potential intervention (KWfr) was significantly correlated with an increase in the final fractions of resistance (Fig. 6), and is discussed below.
Fig. 6.

Ranking intervention targets. Value of Spearman's correlation coefficient (P ⩽ 0·05) between parameter values and fraction of resistant (a) enteric E. coli or (b) E. coli in ingesta by the end of feedlot rearing, given outcome variability introduced by the other parameters of bacterial ecology. For each intervention parameter the data are from 100 model simulations. Parameters: BC, BW, BF, fractional reduction on log scale in plasmid transfer in E. coli in the enteric habitat, water in troughs or feed in bunks, respectively; TC, TW, TF, fractional death of the plasmid-donor cells in E. coli in the enteric habitat, water in troughs or feed in bunks, respectively; γ, fractional regular replacement rate of enteric E. coli; KC, KW, KF, fractional reduction (between 0 and 0·4) in maximum E. coli log c.f.u./ml enteric habitat, water in troughs or feed in bunks, respectively; KWfr, fractional reduction in E. coli log c.f.u./ml fresh water supplied; PC, PW, PF, PE, fractional plasmid curing (between 0 and 0·2) resistant E. coli in the enteric habitat, water in troughs, feed in bunks or the pen's environment, respectively.
The degree of uncertainty in the effects due to variability in the other parameters of bacterial ecology varied between the interventions (Fig. 7). Highly effective were interventions capable of plasmid-curing enteric E. coli or reducing their maximum number (Fig. 7 a, b). The effects of reducing the rate of plasmid transfer or increasing the rate of replacement of enteric E. coli, although also significant, were more gradual (Fig. 7 c, d).
Fig. 7.
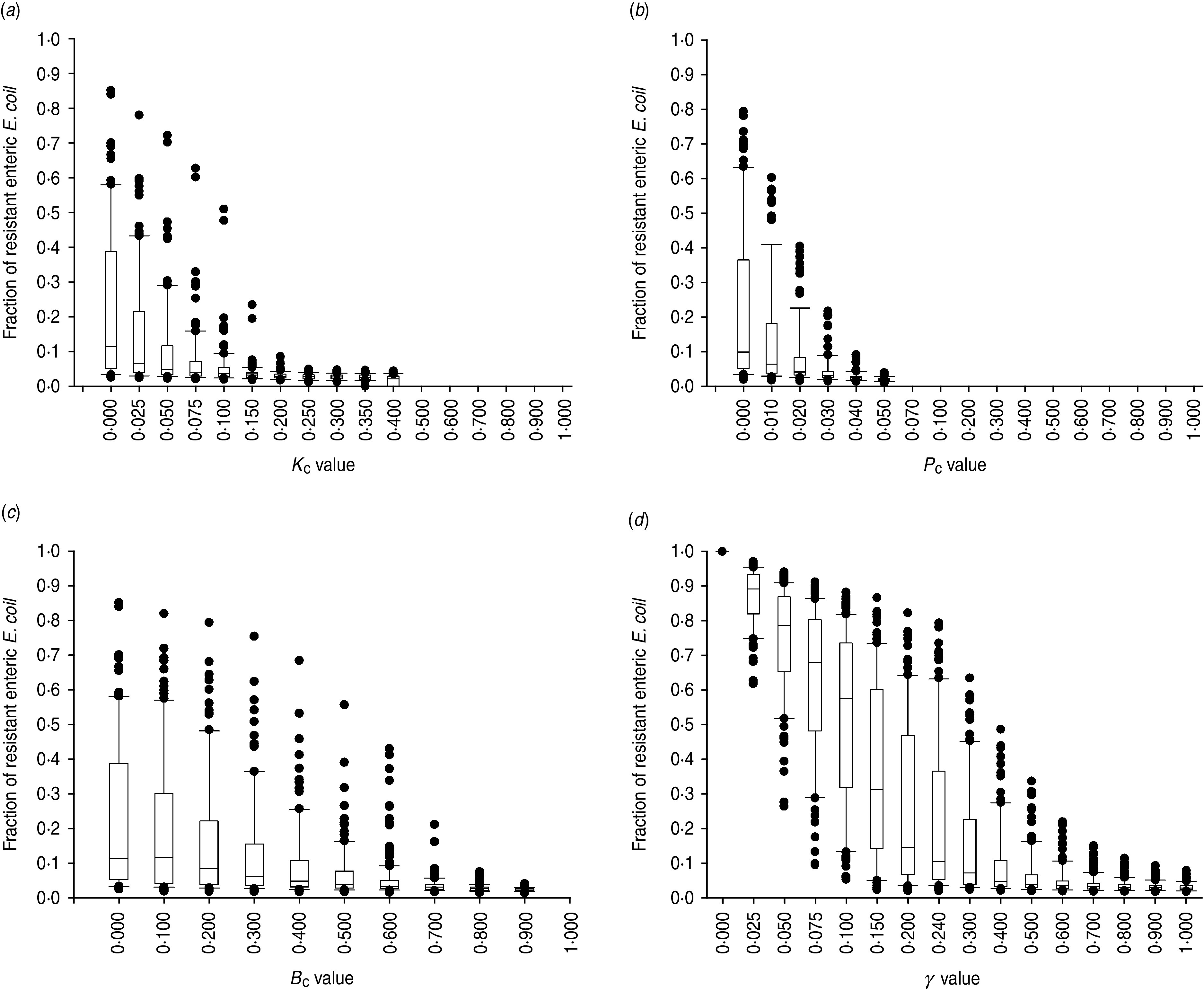
Uncertainty in interventions lowering the fraction of resistant enteric E. coli by the end of feedlot rearing due to outcome variability introduced by the other parameters of bacterial ecology. For each value of intervention parameter the data are from 100 model simulations. The box-and-whisker plot is included for each value until the median outcome of 100 simulations ⩽0·02 (starting fraction of resistance). The effects of an intervention that in the enteric habitat (a) reduces maximum E. coli log c.f.u./ml by fraction KC, (b) induces plasmid curing of PC fraction of resistant E. coli, (c) reduces plasmid transfer rate on log scale by fraction BC; or (d) alters fractional daily replacement rate of enteric E. coli, γ.
DISCUSSION
Within-pen ecology of commensal E. coli during the rearing period determined what fraction of steers' enteric E. coli bear AMR plasmids by the end of rearing, as long as AMR plasmids were present at the start. The final fraction of resistance was insensitive to that at steer placement and, in terms of supplies, lightly sensitive to the frequency of resistance in E. coli in fresh water supplied. Values of individual parameters of the within-pen ecology are likely to vary between the feedlots; this was addressed by considering uncertainty in the outcome in the absence (Fig. 5 a, b) and presence (Fig. 7) of intervention. Similarity between the parameters associated with fractions of resistance in enteric E. coli and in E. coli in steers' ingesta (Fig. 5 c, d) showed that circulation of commensal E. coli between the enteric and non-animate habitats within the pen is an important component in determining the final frequencies of resistance across the habitats. In further discussion we simply refer to the final fraction of resistant enteric (faecal) E. coli as a representative within-pen outcome, and because of the significance of faeces as a vehicle of transmission of microbial AMR genes beyond the farm systems.
In the absence of intervention, a higher fraction of resistant enteric E. coli was associated with higher plasmid transfer rate and maximum E. coli number in this habitat, and, to a much lesser extent, with a higher frequency of resistance in the water supplied (Fig. 5 c). The former two were significant because the rate of plasmid transfer in E. coli in faecal masses in the large intestines was the highest of all the habitats. Consequently, an intervention targeted at lowering the number of enteric E. coli throughout rearing was highly efficient (Figs 6, 7 a). Reducing maximum E. coli numbers in the large intestines' faecal masses by 20% on log-scale (from 316 000 to 25 000/ml) led to a marked decrease in the final fraction of resistance; the effect was relatively insensitive to other variations in bacterial ecology (Fig. 7 a). This was because lowering enteric E. coli led to fewer resistant E. coli shed in faeces (KC, varying from 0 to 0·4, and the number of resistant E. coli/ml of the steers' faecal deposits by the end of rearing: ρ = −0·91, P < 0·001). This reduced the plasmid transfer in the non-animate habitats (e.g. KC and the number of sensitive E. coli in water in troughs receiving plasmids per hour: ρ = −0·59, P < 0·001), and so ultimately reduced ingestion of resistant E. coli (KC and the fraction of resistant E. coli in ingesta: ρ = −0·50, P < 0·001). The magnitude of the effect was not sizably enhanced if maximum E. coli numbers were simultaneously reduced in water in troughs or in feed in bunks (Fig. 6).
Plasmid-curing resistant enteric E. coli (without a reduction in their propensity to receive new AMR plasmids) was also highly efficient (Figs 6, 7 b). There was a very minor synergistic effect if the intervention was simultaneously applied to E. coli in the feed bunks (Fig. 6 a). The sensitizing significantly decreased faecal shedding of resistant E. coli (PC, varying from 0 to 0·2, and the number of resistant E. coli/ml of the steers' faecal deposits by the end of rearing: ρ = −0·78, P < 0·001); a consistently low fraction of resistant E. coli in ingesta with a PC intervention did not contain enough variability to estimate the degree of correlation with this outcome (data not shown). Plasmid-curing E. coli in the feed bunks by itself was associated with lower E. coli fractions in ingesta and the intestines; however, the degree of effect was modest compared with that when targeting enteric E. coli (Fig. 6). A similar effect was not observed for plasmid-curing E. coli in the water in troughs, because of the high degree of turnover of water due to consumption and re-fill.
An intervention lowering the rate of plasmid transfer in E. coli in the enteric habitat was also efficient (Fig. 7 c). Killing the plasmid-donor cells did not sizably enhance this effect (Fig. 6). Because of a relatively low number of resistant E. coli attempting to conjugate, the main impact of killing these cells was a reduction in plasmid transfer. The effects of lowering the transfer rate, whether by killing the donor cells or via another mechanism, by less than 40% were sensitive to variation in the other parameters of bacterial ecology and therefore uncertain. The effects were stable if the transfer rate was reduced further (Fig. 7 c).
Another target for an efficient intervention may be the rate of replacement of enteric E. coli with that ingested (given a lower fraction of resistant E. coli in ingesta). With a 10% daily replacement throughout rearing, the median final fraction of resistant enteric E. coli over 100 model simulations was 57·4%, with the base value used in this study of 24% daily replacement it was 10·4%, and with a 50% daily replacement it was 3·9% (Fig. 7 d). In our previous research [34], with the enteric plasmid transfer rates adopted from in vivo experiments [32], the relatively low fractions of ceftiofur-resistant faecal E. coli in beef cattle reported by field studies could only be reproduced by allowing for regular partial replacement of enteric E. coli with that ingested (see also Fig. 7 d). Notably, then and now, we modelled enteric E. coli under ‘normal’ conditions, without disturbance by exogenous factors such as in-feed or parental antimicrobials. However, in the current model that accounted for ecology of resistance in E. coli in the pen's non-animate habitats, the final fraction of resistant enteric E. coli (median over 100 model simulations) was slightly higher, 2·7%, than that at steer placement, 2·0%, even if all enteric commensal E. coli were replaced daily (Fig. 7 d). These findings support an earlier hypothesis that controlling plasmid-mediated AMR in cattle faecal E. coli requires a more comprehensive understanding of the epidemiology of commensal E. coli, in particular its distribution and movement across hosts and habitats [32].
Targeting interventions at reducing the replication rate of resistant E. coli (analogous to increasing the fitness cost to resistant E. coli) was not efficient. This may be because of a relatively slow growth of E. coli, a facultative anaerobe, in the enteric habitat, which appeared to be a crucial habitat for propagating plasmid-mediated AMR in commensal E. coli within the pen.
Lowering the total number of E. coli in the fresh water supplied to cattle (increasing intervention parameter KWfr) was associated with a higher fraction of resistant enteric E. coli by the end of rearing (Fig. 6). Because the fractions of resistant E. coli within the pen steadily rose throughout rearing (Fig. 4) but the fraction of resistance in water supplied remained relatively low, E. coli from fresh water acted to ‘dilute’ the frequency of resistance in E. coli in cattle ingesta. On the other hand, further lowering the fraction of resistant E. coli in water or feed supply (within the ranges considered) did not significantly alter the final fraction of resistant enteric E. coli (υWfr and υFfr were not significant intervention targets).
It must be noted that given the biology of plasmid-mediated AMR (i.e. horizontal and vertical plasmid transfers) and expected variability in bacterial ecology in the field realm, one cannot state achieving which frequency of resistance in enteric E. coli would constitute a ‘success’. Hence, comparison of relative rather than absolute efficacy of interventions was the only feasible goal for this modelling study, and the model analysis was designed correspondingly (the non-parametric Spearman's correlation coefficient and the standardized regression coefficients). To better visualize the relative efficacies, we opted to present in Figure 7 box-and-whisker plots for all values of an intervention parameter until such value that the median fraction of resistant enteric E. coli at the end of rearing over 100 model simulations was equal to or below that at steer placement. In other words, the intervention mitigated the propagating impact of feedlot rearing on AMR frequency in enteric E. coli. The methods also ensured that the relative comparison of intervention effects did not depend on the actual frequency of resistance. For example, with qualitative interference from Figure 7, one would expect an intervention inducing plasmid-curing to outperform the one reducing plasmid transfer rate, if both achieved the same fractional change in the targeted parameter of bacterial ecology, irrespective of the starting AMR frequency in that E. coli population.
A change in the target parameters may be induced through different biological mechanisms. Table 2 provides examples of existing and potential biological intervention approaches that have been shown or are hypothesized to affect bacterial ecology in similar ways to those modelled. The feasibility of achieving a particular reduction in a target parameter (to compare with Fig. 7) by the existing approaches in the field realm is generally unknown.
In terms of potential intervention approaches that were not considered, a recent proof of concept study shows that, at least with some AMR mechanisms, the resistant bacteria can be rendered to become sensitive by genetic material delivered by constructed phages [45]. Presumably, this could sensitize bacteria without plasmid loss, with the phages essentially acting as adjuvants for antimicrobials [27]. Such an intervention was not included because the related research is at too early a stage to support a mathematical formulation of the resulting plasmid ecology. Introducing new plasmid types for ‘incompatibility curing’ resistant bacteria from AMR plasmids has been researched in the past [28]. However, given current knowledge on plasmid ecology, it would probably not be acceptable from a safety standpoint [45], and therefore was not modelled.
Another intervention that was not considered was killing E. coli without altering the size of the niche available for their population growth (the latter was represented in the model by maximum E. coli numbers in the habitat). First, it is difficult to distinguish which of the two effects a given intervention produces. For example, water sanitization may be thought of as ‘killing’ E. coli; however, essentially, it controls the density of bacteria in this habitat. Second, altering the niche by introducing competitive microflora would be a more self-sustainable intervention compared to the continuously enforced kill.
Insights from this modelling study could help understand the field observations for some of the compounds tried for application in food animals to limit AMR spread in their enteric bacteria. For example, it is unlikely that the observed effects of in-feed bambermycins [19, 20] correspond to a mere reduction in replication of plasmid-bearing Enterobacteriaceae. More likely, the in vivo action either reduces the plasmid transfer (perhaps via selective kill of plasmid-donor cells), or both does this and reduces the replication. Given that the effect of bambermycins on the transfer rate is plasmid-dependent, variability in the efficacy observed in vivo may be due to variable plasmid profiles of field E. coli.
Results of this modelling study also suggest that plasmid-mediated AMR in commensal E. coli can persist and propagate in feedlot operations even in the absence of use of therapeutic or growth-promoting antimicrobials. This is because the concentration of animals in the closed pen supports close circulation of E. coli between the enteric and non-animate habitats throughout the rearing period; this, coupled with high rates of AMR plasmid transfer in the intestines, in the current model led to a build-up of plasmid-mediated AMR in E. coli in all habitats. Presence of fitness cost for bearing AMR plasmids (even if high in magnitude, i.e. RC parameter) was insufficient to impede the propagation of resistance. Given the ‘natural’ presence of AMR plasmids in E. coli in the enteric and non-animate habitats, this means that discontinuing the use of antimicrobials in feedlot cattle alone may not be sufficient to prevent presence of resistant enteric E. coli in animal faeces (a vector of zoonotic transmission) at slaughter age. To prevent such a presence, the no-use policies may need to be coupled with either biological interventions into bacterial ecology producing the effects found significant in this study or disrupting E. coli circulation within the pens. However, the conclusions of this study are based on deterministic modelling, albeit with robust sensitivity and uncertainty analyses. Clarifying the likelihood of resistance extinction under the scenario of no-antimicrobial use with or without further interventions would require accounting for stochasticity in bacterial propagation and movement, and this will be the subject of future research.
ACKNOWLEDGEMENTS
We thank Julia Hertl of Cornell University for help with the statistical analysis. This work was funded by USDA NIFA grant no. 2010-51110-21083.
DECLARATION OF INTEREST
None.
REFERENCES
- 1.Karczmarczyk M, et al. Molecular characterization of multidrug-resistant Escherichia Coli isolates from Irish cattle farms. Applied and Environmental Microbiology 2011; 77: 7121–7127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Salyers AA, Gupta A, Wang Y. Human intestinal bacteria as reservoirs for antibiotic resistance genes. Trends Microbiology 2004; 12: 412–416. [DOI] [PubMed] [Google Scholar]
- 3.Sawant AA, et al. Antimicrobial-resistant enteric bacteria from dairy cattle. Applied and Environmental Microbiology 2007; 73: 156–163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sharma R, et al. Diversity and distribution of commensal fecal Escherichia coli bacteria in beef cattle administered selected subtherapeutic antimicrobials in a feedlot setting. Applied and Environmental Microbiology 2008; 74: 6178–6186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Daniels JB, Call DR, Besser TE. Molecular epidemiology of Blacmy-2 plasmids carried by Salmonella enterica and Escherichia coli isolates from cattle in the Pacific Northwest. Applied and Environmental Microbiology 2007; 73: 8005–8011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.McDermott PF, et al. The food safety perspective of antibiotic resistance. Animal Biotechnology 2002; 13: 71–784. [DOI] [PubMed] [Google Scholar]
- 7.Aslam M, et al. Origin of contamination and genetic diversity of Escherichia coli in beef cattle. Applied and Environmental Microbiology 2003; 69: 2794–2799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Aslam M, et al. Genetic diversity of Escherichia coli recovered from the oral cavity of beef cattle and their relatedness to faecal E. coli. Letters in Applied Microbiology 2004; 39: 523–527. [DOI] [PubMed] [Google Scholar]
- 9.Subbiah M, et al. Selection pressure required for long-term persistence of Blacmy-2-positive Inca/C plasmids. Applied and Environmental Microbiology 2011; 77: 4486–4493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Poole TL, et al. Persistence of resistance plasmids carried by beta-hemolytic Escherichia coli when maintained in a continuous-flow fermentation system without antimicrobial selection pressure. Foodborne Pathogens and Disease 2011; 8: 535–540. [DOI] [PubMed] [Google Scholar]
- 11.Savageau MA. Escherichia coli habitats, cell types, and molecular mechanisms of gene control. American Naturalist 1983; 122: 732–744. [Google Scholar]
- 12.Mataseje LF, et al. Comparison of Cmy-2 plasmids isolated from human, animal, and environmental Escherichia coli and Salmonella spp. from Canada. Diagnostic Microbiology and Infectious Disease 2010; 67: 387–391. [DOI] [PubMed] [Google Scholar]
- 13.Licht TR, Wilcks A. Conjugative gene transfer in the gastrointestinal environment. Advances in Applied Microbiology 2006; 58: 77–95. [PubMed] [Google Scholar]
- 14.Kruse H, Sorum H. Transfer of multiple drug resistance plasmids between bacteria of diverse origins in natural microenvironments. Applied and Environmental Microbiology 1994; 60: 4015–4021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Haack BJ, Andrews RE Jr.. Isolation of Tn916-like conjugal elements from swine lot effluent. Canadian Journal of Microbiology 2000; 46: 542–549. [DOI] [PubMed] [Google Scholar]
- 16.Wittum TE, et al. Ctx-M-type extended-spectrum beta-lactamases present in Escherichia coli from the feces of cattle in Ohio, United States. Foodborne Pathogens and Disease 2010; 7: 1575–1579. [DOI] [PubMed] [Google Scholar]
- 17.George BA, Fagerberg DJ. Effect of bambermycins, in Vitro, on plasmid-mediated antimicrobial resistance. American Journal of Veterinary Research 1984; 45: 2336–2241. [PubMed] [Google Scholar]
- 18.Watanabe T, Ogata Y, Sugawara K. Increase of flavomycin sensitivity of bacteria by R-factors. In: Proceedings of the First International Symposium on Infectious (Transferable) Antibiotic Resistance in Smolenice (CSSR), 1971, 30: 8–29. [Google Scholar]
- 19.Dealy J, Moeller MW. Effect of bambermycins on Escherichia coli and antibiotic resistance in calves. Journal of Animal Science 1977; 45: 1239–1242. [DOI] [PubMed] [Google Scholar]
- 20.van den Bogaard AE, et al. Effects of flavophospholipol on resistance in fecal Escherichia coli and enterococci of fattening pigs. Antimicrobial Agents and Chemotherapy 2002; 46: 110–118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pfaller MA. Flavophospholipol use in animals: positive implications for antimicrobial resistance based on its microbiologic properties. Diagnostic Microbiology and Infectious Disease 2006; 56: 115–1121. [DOI] [PubMed] [Google Scholar]
- 22.Meynell GG, Lawn AM. Filamentous phages specific for 1 sex factor. Nature 1968; 217: 1184–1186. [DOI] [PubMed] [Google Scholar]
- 23.Burman LG. R-plasmid transfer and its response to nalidixic acid. Journal of Bacteriology 1977; 131: 76–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Anderson ES. The ecology of transferable drug resistance in the Enterobacteria. Annual Reviews of Microbiology 1968; 22: 131–180. [DOI] [PubMed] [Google Scholar]
- 25.Novotny C, Knight WS, Brinton CC. Inhibition of bacterial conjugation by ribonucleic acid and deoxyribonucleic acid male-specific bacteriophages. Journal of Bacteriology 1968; 95: 314–326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lin A, et al. Inhibition of bacterial conjugation by phage M13 and its protein G3p: quantitative analysis and model. PLoS One 2011; 6: e19991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lu TK, Collins JJ. Engineered bacteriophage targeting gene networks as adjuvants for antibiotic therapy. Proceedings of the National Academy of Sciences USA 2009; 106: 4629–4634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Trevors JT. Plasmid curing in bacteria. FEMS Microbiology Reviews 1986; 32: 149–157. [Google Scholar]
- 29.Spengler G, et al. The mechanism of plasmid curing in bacteria. Current Drug Targets 2006; 7: 823–841. [DOI] [PubMed] [Google Scholar]
- 30.Loneragan GH, Brashears MM. Pre-harvest interventions to reduce carriage of E. coli O157 by harvest-ready feedlot cattle. Meat Science 2005; 71: 72–78. [DOI] [PubMed] [Google Scholar]
- 31.Callaway TR, et al. Forage feeding to reduce preharvest Escherichia coli populations in cattle: a review. Journal of Dairy Science 2003; 86: 852–860. [DOI] [PubMed] [Google Scholar]
- 32.Daniels JB, et al. Role of ceftiofur in selection and dissemination of Blacmy-2-mediated cephalosporin resistance in Salmonella enterica and commensal Escherichia coli isolates from cattle. Applied and Environmental Microbiology 2009; 75: 3648–3655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Marshall B, Petrowski D, Levy SB. Interspecies and intraspecies spread of Escherichia coli in a farm environment in the absence of antibiotic usage. Proceedings of the National Academy of Sciences USA 1990; 87: 6609–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Volkova VV, Lanzas C, Lu Z, Grohn YT. Mathematical model of plasmid-mediated resistance to ceftiofur in commensal enteric Escherichia coli of cattle. PLoS One 2012; 7: e36738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ayscue P, et al. Modeling on-farm Escherichia coli O157:H7 population dynamics. Foodborne Pathogens and Disease 2009; 6: 461–470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Begon M, et al. A clarification of transmission terms in host-microparasite models: numbers, densities and areas. Epidemiology and Infection 2002; 129: 147–153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Cernat R, et al. Distribution and diversity of conjugative plasmids among some multiple antibiotic resistant E. coli strains isolated from river waters. Bacteriology Virology Parasitology Epidemiology 2002; 47: 147–53. [PubMed] [Google Scholar]
- 38.Hassen A, et al. Use of linear and non-linear growth curves to describe body weight changes in young Angus bulls and heifers. Iowa State University Animal Industry Report, 2004 (AS leaflet R1869), 2004.
- 39.Martin C, Philippeau C, Michalet-Doreau B. Effect of wheat and corn variety on fiber digestion in beef steers fed high-grain diets. Journal of Animal Science 1999; 77: 2269–2278. [DOI] [PubMed] [Google Scholar]
- 40.Morley PS, et al. Effects of restricted antimicrobial exposure on antimicrobial resistance in fecal Escherichia coli from feedlot cattle. Foodborne Pathogens and Disease 2011; 8: 87–98. [DOI] [PubMed] [Google Scholar]
- 41.Mataseje LF, et al. Characterization of cefoxitin-resistant Escherichia coli isolates from recreational beaches and private drinking water in Canada between 2004 and 2006. Antimicrobial Agents and Chemotherapy 2009; 53: 3126–3130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Hamelin K, et al. A virulence and antimicrobial resistance DNA Microarray detects a high frequency of virulence genes in Escherichia coli isolates from Great Lakes recreational waters. Applied and Environmental Microbiology 2006; 72: 4200–4206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hamelin K, et al. Occurrence of virulence and antimicrobial resistance genes in Escherichia coli isolates from different aquatic ecosystems within the St. Clair River and Detroit River areas. Applied and Environmental Microbiology 2007; 73: 477–484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.McKay MD, Beckman RJ, Conover WJ. A Comparison of three methods for selecting values of input variables in the analysis of output from a computer code. Technometrics 1979; 21: 239–45. [Google Scholar]
- 45.Edgar R, et al. Reversing bacterial resistance to antibiotics by phage-mediated delivery of dominant sensitive genes. Applied and Environmental Microbiology 2012; 78: 744–751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Orr RJ, et al. Defaecation and urination behaviour in beef cattle grazing semi-natural grassland. Applied Animal Behaviour Science 2012; 139: 18–25. [Google Scholar]
- 47.The Ohio State University Extension. Ohio Livestock Manure Management Guide, Bulletin 604, 2006.
- 48.Gadberry S.Water for beef cattle (Fsa 3021): University of Arkansas, Division of Agriculture; (http://www.uaex.edu/Other_Areas/publications/PDF/FSA-3021.pdf). Accessed on 2 June 2012. [Google Scholar]
- 49.Robinson B, Okine E. Nutrition and management: feed intake in feedlot cattle. In: Alberta Feedlot Management Guide, 2nd edn. 2000. (revised 2009). Feeder Associations of Alberta Ltd, Barrhead, Alberta, Canada: (http://Www1.Agric.Gov.Ab.Ca/$Department/Deptdocs.Nsf/All/Beef11677). Accessed 2 June 2012. [Google Scholar]