Abstract
Salmonella enterica is a gastroenteric Gram-negative bacterium that can infect both humans and animals and causes millions of illnesses per year around the world. Salmonella infections usually occur after the consumption of contaminated food or water. Infections with Salmonella species can cause diseases ranging from enterocolitis to typhoid fever. Salmonella has developed multiple strategies to invade and establish a systemic infection in the host. Different cell types, including epithelial cells, macrophages, dendritic cells, and M cells, are important in the infection process of Salmonella. Dissemination throughout the body and colonization of remote organs are hallmarks of Salmonella infection. There are several routes for the dissemination of Salmonella typhimurium. This review summarizes the current understanding of the infection mechanisms of Salmonella. Additionally, different routes of Salmonella infection will be discussed. In this review, the strategies used by Salmonella enterica to establish persistent infection will be discussed. Understanding both the bacterial and host factors leading to the successful colonization of Salmonella enterica may enable the rational design of effective therapeutic strategies.
1. Introduction
Salmonella is a Gram-negative, intracellular pathogen. There are more than 2,600 serovars of Salmonella characterized to date that are differentiated on the basis of the lipopolysaccharide (LPS) O antigen and the flagellar H antigen [1]. There are just two species of Salmonella: S. enterica and S. bongori [2]. Salmonella typhi and Salmonella typhimurium are well-known members of the S. enterica species. S. typhimurium and S. enteritidis are predominantly associated with gastroenteritis in humans [3]. Each year, there are about 155,000 deaths due to nontyphoidal Salmonella (NTS) infections. Salmonella enterica serovar typhi infections cause a staggering 20 million infections and 200,000 deaths annually [4]. Gastroenteritis induced by Salmonella infections is a major cause of morbidity and mortality in children under 5 years of age [5]. Diarrhea caused by Salmonella species causes a global human health burden that contributes to significant annual morbidity and mortality and requires new therapeutic strategies for effective management. Almost 60% of Salmonella strains have developed resistance to first-line antibiotics [6]. Most patients recover from infections after treatment. However, 3–5% of patients become chronic carriers, with chronic infection in the gall bladder [7]. Chronic carriers can intermittently shed the bacteria through their feces and urine throughout the rest of their lives [8]. The liver is also a reservoir for chronic infections with Salmonella Typhi; from the liver, the bacteria can be intermittently shed into the gallbladder [9].
Salmonella typhi infections can cause fever, hepatomegaly, splenomegaly, and bacteremia. In the disease process, the bacteria disseminate into the gall bladder, liver, and spleen [10]. Approximately 90% of chronic Salmonella carriers have gallstones [10–12], and are at significantly increased risk for gallbladder cancer (GC) [12, 13]. DelGiorno et al. reported that persistent Salmonella infections can cause pancreatitis in a murine model of infection [14]. Some Salmonella carriers are asymptomatic. Roughly 2–5% of Salmonella-infected patients fail to clear the bacteria within one year [12]. Such chronic infections, especially asymptomatic infections, pose a huge socioeconomic burden, especially in South Asian and African countries, by unknowingly spreading infections to others, who may experience symptomatic infections and suffer economic costs as a result. Understanding the cellular routes of Salmonella invasion and dissemination in the host and the mechanisms of Salmonella persistent infection may facilitate the exploration of novel treatment strategies for patients with chronic infections. Ultimately, this may help eliminate the asymptomatic carriage of Salmonella as a concern for public health.
Salmonella infections can result from the ingestion of contaminated foods because they can survive the low pH of the stomach [15]. Although bile in the small intestine poses a challenge for S. typhimurium, the PhoQ/PhoP two-component regulatory system mediates resistance to bile [16]. Salmonella predominantly causes inflammation of the terminal ileum and colon [17]. S. typhimurium can spread systemically in mice, and S. typhimurium infections in mice are used as an animal model for typhoid fever in humans [18]. Pretreatment of mice with streptomycin prior toSalmonella infection disturbs the healthy microbiota and facilitates infection of the intestinal lumen with Salmonella typhimurium [19]. Streptomycin-treated mice are therefore often used as animal models of S. Typhimurium-induced gastroenteritis [19]. Before the oral infection of S. typhimurium and S. enteritidis, approximately 20 mg of streptomycin treatment by intragastric administration in the mice will allow a high colonization level in the cecum and colon of the mice [20]. Acute microbiota depletion will reduce the colonization resistance and facilitate the infection of the bacteria. Microbiota can limit Salmonella colonization, and diet can affect microbiota composition. Low-fiber or high-fat diets will increase S. typhimurium colonization in mice [21]. Fat can promote S. typhimurium infection in mice by eliciting bile salts, which help fat digestion [21]. A high-fat diet will cause microbiota perturbation [21]. E. coli may limit S. typhimurium infections during diet shifts [21].
Mice with a mutation in the natural resistance-associated macrophage protein 1 gene (Nramp+), such as CL57/BL6 or BALB/C mice, are susceptible to Salmonella infection [22]. Nramp1 is a macrophage-specific exporter, and the Nramp1 gene codes for an ion transporter that pumps ions out of Salmonella-containing vacuoles (SCV) [22]. The SCV is the intracellular vacuolar niche in which Salmonella can replicate and achieve dormant infection. wild type 129 × 1/Sv mice, which possess the Nramp+/+ allele, are used as an animal model for chronic S. typhimurium infection [23]. Mice with a mutation in the natural resistance-associated macrophage protein 1 gene (Nramp1), such as CL57/BL6 or BALB/C mice, are susceptible to Salmonella infection [22]. Nramp1 is a macrophage-specific exporter, and the Nramp1 gene codes for an ion transporter that pumps ions out of SCV [22]. The SCV is the intracellular vacuolar niche in which Salmonella can replicate and achieve dormant infection. Wild type 129 × 1/Sv mice, which possess the Nramp1+/+ genotype, are used as an animal model for chronic S. typhimurium [23].
2. M Cells
Enteropathogenic infections start in the intestinal lumen. Dissemination through microfold or membranous (M) cells is one of the best-understood routes of Salmonella dissemination [24]. M cells are specialized follicle-associated epithelial (FAE) enterocytes on the surface of mucosa-associated lymphoid tissues [25, 26]. Salmonella typhimurium initiates infection in mice by infecting and destroying the specialized epithelial M cells and then traveling to the mesenteric lymph nodes [24]. See Figure 1.
Figure 1.
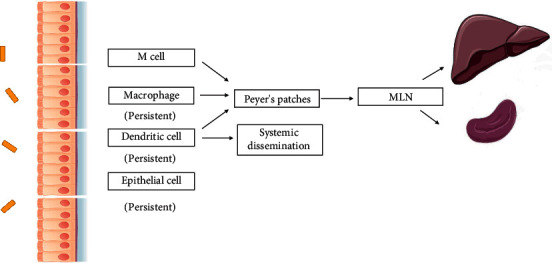
The multiple routes of Salmonella dissemination. Salmonella can be phagocytized by macrophages or dendritic cells and disseminate to the Peyer's patches (PP), mesenteric lymph nodes, and eventually the liver and spleen. Salmonella can also reach the circulation by the transportation of dendritic cells, e.g., CD18-expressing phagocytes. Salmonella can also invade the intestinal epithelial cells directly by way of a zipper or trigger mechanism. Salmonella can achieve persistent infection in epithelial cells and phagocytic cells. The dormant persisters can be released and induce the recurrence of the infection.
Salmonella directly invades M cells but can also transform follicle-associated epithelial cells into M cells to provide additional routes for intestinal invasion [27]. Indeed, Tahoun et al. found that S. Typhimurium can induce an epithelial-mesenchymal transition (EMT) of FAE enterocytes and transition the FAE to M cells [27]. These processes rely on the bacterial type III effector protein SopB [27]. Through the activation of NF-κB and Wnt/b-Catenin signaling pathways, Salmonella induces host cell transdifferentiation through receptor activator of NF-kB ligand (RANKL) [27]. This finding was the first report that S. typhimurium can transform epithelial cells into M cells using a single virulence factor.
Intestinal immunity is the first defense barrier that enteropathogens encounter during infection. Lymphotoxin signaling is important for maintaining intestinal immune balance. LTβR can also be activated by lymphotoxin (LTαβ) [28]. Lymphotoxin signaling promotes the differentiation of M cells from intestinal epithelial cells [29]. This signaling is involved in the regulation of intestinal inflammation, as shown by the DSS-induced colitis model [30]. Mice with knocked-out lymphotoxin signaling molecules (LTα3, LTα2β1, and LTα1β2) have abnormal lymphoid development [31]. Lymphotoxin β-receptor knockout mice lack all lymph nodes and gut-associated lymphatic tissues, including Peyer's patches (PPs) [32]. These lymph node-defective mice are a good model for the systemic dissemination of S. typhimurium. Infection of Salmonella in LTβR−/− mice demonstrates that organized lymph tissues are dispensable for the systemic infection of the host [20]. As shown by a study from Barthel et al., without Peyer's patches (PPs), bacteria can still reach remote organs [20]. This phenomenon indicates the importance of dendritic cell-mediated transportation in the dissemination of S. typhimurium [33]. Salmonella exploited dendritic cells as vesicles for dissemination. Cheminay et al. showed that after infection by Salmonella, dendritic cells could upregulate the CCR7 receptor and migrate via the CCR7 ligands CCL19 and CCL211 [13, 33–35].
A study by Wroblewska et al. showed that lymphotoxin signaling is essential for the clearance of Salmonella from the intestinal lumen [36]. A lack of LTβR signaling did not impact the initiation of inflammation induced by Salmonella. However, the resolution of Salmonella infection was impaired [36]. The infectious processes in S. typhimurium in WT and LTβR−/− mice lacking Peyer's patches (PPs) and MLN are highly similar [20].
3. Epithelial Cell
S. typhimurium can invade polarized gallbladder epithelial cells and replicate inside the epithelial cells [37]. Gallbladder epithelial cells are a reservoir for Salmonella colonization [37]. Long-term colonization of Salmonella in the gallbladder cells can drive the premalignant transformations of the cells. Salmonella can invade the polarized gallbladder cells. Salmonella can induce the extrusion of epithelial cells, which is accompanied by caspase-1 activation-related cell death. Epithelial cells can provide a shelter for the bacteria to survive and replicate in the cytosol of the epithelial cells [38, 39]. The type III secretion system is involved in the priming of the bacteria for invasion. Cytosolic bacteria can induce the extrusion of epithelial cells and be released into the intestinal lumen [38, 39].
Unlike M cells, Salmonella's invasion of epithelial cells does not rely on phagocytosis. The type III secretion system (T3SS) is the most important virulence factor for Salmonella species, and one is encoded on Salmonella pathogenicity island 1 (SPI1) and the other is encoded on Salmonella pathogenicity island 2 (SPI2) [40]. The type III secretion system is a molecular syringe that can translocate the effector proteins directly from the bacteria into the cytosol of cells. Effector proteins are injected into the cytoplasm of the host by a T3SS gene cluster. SPI1 is involved in the invasion process of Salmonella [41]. After invading host cells, Salmonella survives in SCVs by using elements encoded on SPI2 [42–45]. Approximately 4–6 h after the cellular invasion, bacterial replication is initiated [46].
Salmonella can induce membrane ruffling in intestinal cells to cause them to engulf the bacteria [47]. Various S. Typhimurium fimbrial operons contribute to bacterial attachment and invasion of epithelial cells [48]. The zipper and trigger mechanisms are two well-studied mechanisms of Salmonella entry into epithelial cells [49, 50]. The trigger mechanism is activated by the type III secretory system [49]. SipB/C in Salmonella type III secretory system assembles a pore in the epithelial cell, bacteria and epithelial cells can contact through the continuum created by the SipB/C [49]. Cytoskeletal reorganizations known as “membrane ruffles” and “internalization” are two key elements of the trigger mechanism [51]. Bacteria are internalized in SCV following a trigger mechanism [49].
In contrast, there are only minor cytoskeletal protein rearrangements involved in the zipper mechanism [50]. Instead, the zipper mechanism is mainly mediated by interactions between bacterial ligands such as Rck and host cell surface receptors [52]. There are many outer membrane proteins that participate in the invasion process of Salmonella typhimurium [53]. Rck is a 17 kDa outer membrane protein (OMP), which are membrane proteins found in the outer membranes of Gram-negative bacteria. Rck is encoded by the rck gene on the large virulence plasmid [54]. They are a family of highly conserved OMPs within the Enterobacteriaceae family. This receptor binding leads to downstream signal activation mediated by the phosphorylation of tyrosine kinase. The zipper mechanism is activated by the binding of host cell receptors by the bacterial ligands. Actin polymerization and membrane extension are initiated by the activated downstream signaling.
PagN is another OMP [54] and is widely conserved in the Salmonella genus [55]. The PagN protein interacts with cell surface heparin sulfate proteoglycans to invade cells [53]. Binding between OmpV and the extracellular matrix components fibronectin and α1β1 integrin leads to the adhesion of Salmonella typhimurium to intestinal epithelial cells and ultimately activates actin modulation [56]. PAMPs of Salmonella can be recognized by the innate immune response receptors through MyD88-dependent TLR signaling [57]. Infection with SPI1 T3SS disrupted Salmonella can still induce colitis in C57BL/6 mice through a mechanism that is dependent on MyD88 signaling [58]. The effectors of type III secretion systems in the invasion and dissemination of Salmonella are summarized in Table 1.
Table 1.
Effectors of Type III secretion systems in the invasion and dissemination of Salmonella.
| Type III secretion System | Function | References |
|---|---|---|
| SPI-1 | Invasion of nonphagocytic cells, including epithelial cells | [40] |
| SPI-2 | Survive in the phagocytic cells | [59] |
| SseI | Inhibition of macrophages and DCs migration | [60] |
| SipA | Promotion of cytoskeletal rearrangements, invasion of epithelial cells | [61, 62] |
| SopA | Invasion of epithelial cells | [63] |
| SopB | Invasion of epithelial cells | [63] |
| SopD | Invasion of epithelial cells | [63] |
| SopE2 | Invasion of epithelial cells | [63] |
| SipB/C | Translocator to deliver the effectors into the cell | [49] |
The binding of pattern recognition receptors (PRRs) with pathogen-associated molecular patterns (PAMPs), including peptidoglycan, lipopolysaccharide, flagellin, can mediate Salmonella invasion [64, 65]. TLR4 and TLR5 play a role in the host response to Salmonella [66]. In human macrophages, Salmonella can activate NAIP/NLRC4 and canonical NLRP3 Inflammasomes by its flagellin [67]. Caspase-1 will be activated after binding with NLRC4 and NLRP3 inflammasomes in response to Salmonella. Salmonella colonization was much higher in caspase 11 deficient mice than in wild-type mice [68]. Casp1−/− and Casp1/11−/− monolayers showed significantly increased intracellular bacteria, accompanied by low intestinal epithelial cells (IECs) shedding and death [68]. Caspase activation is important for limiting the intracellular replication of Salmonella [68].
4. Dendritic Cells
Intestinal dendritic cells are found in Peyer's patches [69], in the lamina propria [70], in the subepithelial dome [71], and under the follicle epithelium [72, 73]. The phagocytosis of Salmonella by dendritic cells and macrophages is mediated by the interactions between specific pathogen-associated molecular patterns (PAMP) and cellular receptors on the phagocyte surface, such as pattern recognition receptors (PRRs), which include Toll-like receptors (TLRs), NOD-like receptors (NLRs), and C-type Lectin receptors [74]. NOD-like receptors (NLRs), nucleotide-binding leucine-rich repeat-containing proteins, are intracellular innate immune receptors that belong to the pattern recognition receptors (PRRs) [75]. NLR is short for nucleotide-binding domain leucine-rich repeat. MyD88-and TRIF-dependent pathways can be regulated by NLRs [75].
Dendritic cells are exploited by Salmonella typhimurium as “Trojan horses” to enable systemic dissemination [76]. This strategy of manipulating host cell migration to facilitate broader dissemination is common among other pathogens such as Mycobacterium tuberculosis, HIV, and a range of other Gram-negative bacteria [77–80]. For example, after phagocytes are infected by HIV, the gp120 protein on the virus binds with the C-type Lectin receptor DC-Sign, initiating phagocytosis by dendritic cells that then migrate to lymph nodes and release viral particles that proceed to infect CD4+ lymphocytes [77–79]. Several Gram-negative bacteria also disseminate through antigen-presenting cells, as demonstrated by Yang et al. [76, 81–88]. After binding to C-type lectin receptors with core LPS, bacteria are transported throughout the host by antigen-presenting cells [76, 81–88].
By exploiting migratory dendritic cells, the Salmonella can thus traffic from the intestinal lumen to systemic organs [34]. During active infection, the dendritic cells' expression of CCR7, a receptor for the chemokines CCL19 and CCL21, is increased [34]. This allows dendritic cells to migrate along chemotactic gradients to remote sites like the lymph nodes and spleen [34]. Salmonella survives inside the dendritic cells, subverts the function of dendritic cells, impairs the activation of adaptive immune responses, prevents fusion and lyso-endosomal degradation, and achieves systemic dissemination [45]. Cheminay et al. published the first example that Salmonella can inhibit antigen presentation by dendritic cells by altering MHC-II-dependent antigen presentation in an SPI2-dependent manner [89]. Through subversion of the antigen presentation of dendritic cells, the bacteria reduce the activation of the active immune response. Lapaque et al. demonstrated that Salmonella can inhibit the surface expression of MHC class II antigens on dendritic cells through ubiquitination [90].
CD103+CD11b+ DCs have been reported to transport Salmonella typhimurium to the mesenteric lymph nodes (MLN) after oral infection [91]. CD103+ dendritic cells (DCs) typically phagocytose bacteria from the small intestine and present antigens to T cells [91]. Another group of dendritic cells that can facilitate the dissemination of Salmonella is intestinal CD11c+ lamina propria cells (LPCs), which do so in a TLR5−dependent manner [92]; the migration of Salmonella typhimurium from the intestinal tract to MLN is impaired in TLR5−/− mice. In TLR5−/−mice, migration of bacteria by CD11c + LPCs is impaired [92, 93].
Distinct populations of dendritic cells participate in the processing and immune sampling of Salmonella. Specialized DC subsets in Peyer's patches (PPs), CCR6 (+) DCs, are recruited to the dome regions of Peyer's patches (PPs) to sample the bacteria and present to CD4+ T cells [94, 95]. CX3CR1-positive lamina propria DCs take up S. typhimurium by transepithelial processes [96]. Indeed, CX3CR1 deficiency leads to reduced bacterial sampling in the intestinal lumen by lamina propria DCs [96]. Further, these CX3CR1-positive DCs lacked CCR6 expression, which is different from the Peyer's patches (PPs) associated-dendritic cells [96].
S. typhimurium can be taken up by sub-epithelial DCs and can survive within murine PP dendritic cells [97]. The S. typhimurium strain PhoPc has a point mutation in the phoP/Q locus [98] that attenuates its capacity to survive in macrophages but was able to persist for several weeks in vivo [97]. Salmonella can persist in the dendritic cells in the Peyer's patch. They can also be directly sampled by dendritic cells that express tight junction proteins, such as the interepithelial dendritic cells in the intestinal villi that penetrate gut epithelial monolayers by opening tight junctions and directly sampling bacteria from the mucus [99].
Infection of CD11c–CD18+ dendritic cells can lead to rapid entry into the systemic circulation. It has been reported by Vazquez-Torres et al. that Salmonella can achieve systemic dissemination through CD18-expressing phagocytes [100]. One hour after infection, Salmonella can be detected in the blood. At sites other than M cells and Peyer's patches, Salmonella can also disseminate from the gastrointestinal tract to the spleen. Downregulation of DC cells in the lamina propria can limit the invasion of Salmonella [100].
5. Macrophage
During the intracellular life of Salmonella in the host cells, Salmonella can interfere with the antigen-presenting process of the dendritic cells, for example, by interfering with the antigen presentation of bacteria on dendritic cells and inhibiting the adaptive immunity, Salmonella can affect the polarization of macrophages to the M2 phenotype, which will inhibit the inflammatory process and facilitate the persistent survival of Salmonella in the host. The manipulation of the macrophage is a strategy that Salmonella derived during its evolution. Uchiya et al. demonstrated that Salmonella can interfere with the function of macrophages to escape immune responses. Uchiya et al. reported that Salmonella can inhibit cytokine signaling in macrophages via the Janus kinase/signal transducer and activator of transcription (JAK/STAT) signaling pathway through SPI2 [101].
In addition to dendritic cells and M cells, S. typhimurium can also disseminate via inflammatory monocytes. Monocytes are recruited to the inflammatory sites where they differentiate into macrophages. Macrophages serve as a reservoir in which Salmonella can survive and replicate [102]. Inside the macrophage, Salmonella can induce micropinocytosis [103], and spacious phagosomes (SP) are formed after Salmonella enters the macrophage and persists in the cytoplasm [103]. A T3SS encoded by SPI2 allows survival and avoids the NADPH oxidase-dependent killing of macrophages [104].
The PhoQ/PhoP regulatory system is utilized by S. typhimurium to enable survival in macrophages [105]. The PhoQ/PhoP two-component system is one of the most important regulatory mechanisms for the virulence of Salmonella. Inside the SCV, the low PH and low Mg2+environment activate the two-component PhoQ/PhoP system [106]. The gene regulating the expression of O antigen, rfb, is inhibited inside the SCV [107]. Thus, the length of O antigen is decreased under the regulation of the two-component PhoQ/PhoP system. The protease PgtE in Salmonella typhimurium, a homologue for Pla in Yersinia Pestis and OmpT in E. coli, is then expressed [108]. Expression of PgtE protease dissolves the extracellular matrix and facilitates the cellular dissemination of Salmonella in vivo. S. typhimurium, when released from the macrophage, can then be phagocytosed by other cells, including other macrophages [109].
Salmonella can modify macrophage polarization during chronic infection. Macrophages can differentiate into two groups after bacterial infection; the classically activated macrophages (M1 type) or the alternatively activated macrophages (M2 type). Cytokines are the primary determinant of macrophage polarization. The M1 type is proinflammatory and activates a Th1 immune response [110]. IFNγ- and LPS-induced activation of TLR4 signaling can shift the macrophage to the M1 phenotype. In contrast, the M2 type is antiinflammatory and activates the Th2 immune response [110]. The cytokine IL-4 shifts macrophages to the M2 phenotype. Usually, macrophages will exhibit M1 polarization after sensing the stimuli from bacteria or viruses. Salmonella phagocytized by the macrophage can shift the macrophage polarization state. Saliba et al. reported that macrophages harboring nongrowing Salmonella are prone to proinflammatory M1 polarization, but macrophages harboring growing bacteria shifted to an antiinflammatory M2-like state [111]. S. typhimurium preferentially lives in M2 macrophages during chronic infections [110]. Thus, Salmonella has mechanisms to shift the differentiation of macrophages into the M2 phenotype [110]. Intracellular glucose levels are higher in M2 macrophages, contributing to their permissiveness for the intracellular replication of Salmonella [112].
S. typhimurium persists within splenic granulomas enriched with CD11b + CD11c+Ly6C+ macrophages [4, 113]. Trung et al. previously reported that Salmonella can manipulate granuloma macrophage polarization towards the M2 phenotype [4]. As previously discussed, S. typhimurium preferentially persists in M2-reprogrammed macrophages. The bacterial effector SteE contributes to the establishment of persistent infection by downregulating tumor necrosis factor (TNF) signaling [4]. The bacteria have to develop strategies to overcome the immune response and persist chronically. S. typhimurium can polarize the primary macrophages to M2 polarization through the e SPI2 T3SS effector SteE. Macrophage M2 polarization can contribute to the systemic persistence of the bacteria [113].
Studies have shown that Salmonella can induce host cell death during infection [114]. Monack et al. found that caspase-1 is exploited by Salmonella to colonize the Peyer's patches (PPs) [115]. Systemic dissemination after an oral challenge with Salmonella is impaired in Casp-1−/− mice. This indicates that caspase-1 is important for the systemic dissemination of Salmonella [115]. Caspase-1 (Casp-1), an interleukin [IL]-1β–converting enzymes, can induce apoptosis in mammalian cells. Caspase 1 can cleave the proinflammatory cytokines IL-1β and IL-18. Mice lacking Casp-1 (Casp-1−/−mice) showed a 1,000-fold higher lethal dose (LD50) of S. typhimurium in the mice than wide-type mice [115]. Casp-1-/−mice were colonized by lower intracellular bacteria and did not show systemic dissemination of the bacteria, reduced colonization of bacteria in the Peyer's patches (PP) and spleens [115]. It suggests that Casp-1 is necessary for the establishment of systematic infection by S. typhimurium in mice [38, 67, 68, 115, 116]. Salmonella colonization was much higher in Caspase 11 deficient mice than in wild-type mice [68]. Casp1−/− and Casp1/11−/− monolayers showed significantly increased intracellular bacteria, accompanied by low intestinal epithelial cells (IECs) shedding and death [68]. Caspase activation is important for limiting the intracellular replication of Salmonella.
Inflammasome activation is one important pathway during the infection of Salmonella in the intestinal epithelial cells [38]. The infection of Salmonella typhimurium can also lead to the activation of Caspase 4, and Caspase 4 can limit the replication of S. typhimurium in the cells [117]. Activation of caspase 4 can lead to the noncanonical activation of the inflammasome pathway [117].
Salmonella can activate apoptosis of Salmonella-infected macrophages using effectors encoded in pathogenicity island-1 through both intrinsic and extrinsic pathways [118]. Cell death induced by the infected cells gives the bacteria an opportunity to be released and infect further cells. Salmonella can induce cell death in macrophages through several mechanisms. Immediate cell death can be induced by the type III secretion system (T3SS) of Salmonella. Or, the macrophages harboring Salmonella can be further phagocytosed by neighboring macrophages. Bacteria are released from dead cells and phagocytized by local macrophages, enabling another cycle of intracellular replication and cell-to-cell spread [114]. Ultimately, this cycle helps ensure the intracellular survival and persistent infection of phagocyte populations with Salmonella.
6. Chronic and Systemic Infection of Salmonella Typhimurium
Supershedders are the hosts responsible for the host-to-host transmission and reoccurrence of S. typhimurium since supershedders shed the bacteria in their feces. Foxp3þ Regulatory T cells play a role in the persistent infection of Salmonella [119]. Foxp3+ Treg ablation early after infection will accelerate bacterial eradication [119]. This indicated that immune regulatory T cells function in the early stages of infection to establish a persistent Salmonella infection [119].
Monack et al. demonstrated that Salmonella can persist in the MLNs of mice for up to one year. Macrophages in the MLNs can be the reservoirs of the bacteria. Voedisch et al. suggested that the MLN represents a restrictive site for the growth and dissemination of Salmonella [33]. In mice whose mesenteric lymph nodes have been surgically excised, the colonization of Salmonella in the liver and spleen is increased [33]. In such mice, Salmonella forms nonreplicating “persisters” in macrophages [120]. Persisters are in a state of dormant infection that is tolerant to drug treatment [121]. Indeed, they have resistance to antibiotics and can eventually reactivate and begin to replicate once more [122]. Persister cells are one important reason for relapsed infections. Persisters facilitate the chronic infection with S. typhimurium. Persisters can undermine the host immune response [123]. These persisters can reprogram the macrophages they dominate [123]. After exposure to ciprofloxacin, a fluoroquinolone antibiotic, Salmonella enterica persisters form unstable small colony variants. These phenotypes help the bacteria survive in the face of environmental stress or antibiotic treatments.
Salmonella persister cells are important components of biofilms [124]. Biofilm formation is an important strategy for persistent bacterial infections [125]. Forming biofilm can confer the bacteria survival advantages. Biofilm formation on gallstones is important for the chronic carriage of Salmonella. Antibiotic therapy efficiency is compromised in patients with a biofilm in the gall bladder. Salmonella infection in the gall bladder can induce the destruction of the epithelial cell integrity.
Biofilms are just one strategy for the bacteria to survive harsh environments. Even without animal reservoirs, biofilms can help Salmonella spp. to survive in the environment until uptake into a new host. However, the Salmonella Typhimurium ST313 strain which can cause blood stream infections and is typically seen in Sub Saharan Africa [126], has poor biofilm-forming ability and cannot survive long outside a host [127].
Except in antigen-presenting cells, Salmonella achieves a persistent infection in epithelial cells [128] by remaining in a dormant state. Luk et al. found that Salmonella can live in a dormant state in the vesicular compartment, different from the Salmonella-containing vacuoles (SCV). Contrary to macrophages, Salmonella in epithelial cells can express Salmonella Pathogenicity Island 2 (SPI-2) virulence factors. This report is the first to describe another persistent infection state and mechanism for S. typhimurium [128].
The Salmonella SPI2 effector SseI (also called SrfH) binds with host factor IQ motifs containing GTPase activating protein 1 (IQGAP1). SseI has been reported to mediate long-term systemic infections [60]. Pseudogenization of SseI leads to rapid systemic dissemination of Salmonella typhimurium through migratory dendritic cells [129]. In the sub-Saharan African Salmonella typhimurium strain ST313 lineage II, sseI is lost by pseudogenization. ST313 can disseminate from the gut to mesenteric lymph nodes (MLNs) via CD11b + migratory dendritic cells (DCs) [129]. However, recovery of the gene function by expressing functional SseI in ST313 isolates reduces the dissemination of the bacteria [129].
The interplay between the host immune system and pathogens is a complex process during chronic infections. Dendritic cells and macrophages are important reservoirs for the bacteria that enable long-term survival. Helicobacter pylori, Mycobacterium tuberculosis, and Salmonella enterica all survive inside antigen-presenting cells (APCs). The gall bladder, bone marrow [130], and mesenteric lymph nodes are sites that can support persistent infection with Salmonella. Persistent infection with Salmonella can cause disease in multiple organs, from gallbladder cancer to pancreatitis. Pancreatitis can be caused by persistent infection of mice with Salmonella [14]. Inflammatory, fibrotic, and epithelial responses can be detected in the pancreases of mice persistently infected with S. typhimurium [14]. Pancreatic acinar cells can be invaded by S. typhimurium.
Salmonella infections are associated with the development of IBD (inflammatory bowel diseases) and colon cancer [131, 132]. One study by Katrin et al. reported that mice with chronic infections with S. typhimurium develop severe and persistent intestinal fibrosis and have upregulation of several matrix metalloproteinases (MMPs) [133]. Transforming growth factor–β1, insulin-like growth factor-I, and type I collagen deposition levels are increased during persistent infection of S. typhimurium [134, 135].
As shown in mouse models, chronic infection with S. typhimurium increases the susceptibility to intestinal inflammation [136]. The dDextran sulfate sodium (DSS)-induced colitis and interleukin (IL)-10−/− spontaneous inflammation mice models were used in this particular study [137]. Because of persistent infection of S. typhimurium in the liver and spleen, these mice are more susceptible to intestinal inflammation. This indicated S. typhimurium persistent infection might be related to the accelerated onset of IBD (inflammatory bowel diseases) of the host [137].
Various studies support the mesenteric lymph nodes as a site that harbors Salmonella to sustain a chronic infection [138]. Salmonella can persist in the hemophagocytic macrophages of MLN. Removal of MLN increases the bacterial burdens in mice, however, indicating that another reservoir of Salmonella exists other than MLN [138]. Bacteria can be cultured from the liver tissue of chronically infected mice [139]. Liver macrophages are shifted to the M2 phenotype during persistent infection. An immune response balance exists during chronic infection with Salmonella, for example, the proinflammatory IFNγ and antiinflammatory signals IL-10. This balance allows the bacteria to survive in the persistent infection sites [139].
The cytokine Interleukin-22 (IL-22) can help the colonization of Salmonella by suppressing other commensal bacteria [140]. IL-22 can function in tissue repair and host defense; it is induced during pathogen infection. Behnsen et al. reported that IL-22 can suppress the intestinal microbiota [140]. IL-22 suppresses commensal Enterobacteriaceae and boosts the colonization of Salmonella. Binding of bacteria with APCs will induce the release of cytokine IL-23; IL-23 induces IL-17 and IL-22 release [141, 142]. In IL-22−/− mice has higher E. coli burden and reduced Salmonella colonization in the intestine than wide type mice. IL-22 can induce the antimicrobial proteins lipocalin-2 and calprotectin release to inhibit the growth of commensal microbiota. This mechanism is exploited by Salmonella to outcompete intestinal microbiota [140].
7. Concluding Remarks
Achieving a better understanding of the pathogenesis of Salmonella will provide further insights into key host-pathogen interactions that affect persistent bacterial infections. Understanding the detailed mechanisms and the specific host cell types involved in Salmonella infections may help guide the future development of therapeutic interventions. Understanding the mechanisms of Salmonella persistent infection will enable researchers to improve upon current treatment strategies, especially for asymptomatically infected patients. Treating chronically infected patients will help reduce the reservoirs for the bacteria and limit the transmission of the disease.
Acknowledgments
This study was funded by a local grant from the Tongji Hospital of the Huazhong University of Science and Technology.
Conflicts of Interest
The author declares that they have no conflicts of interest.
References
- 1.Li P., Liu Q., Luo H., et al. O-serotype conversion in salmonella typhimurium induces protective immune responses against invasive non-typhoidal Salmonella infections. Frontiers in Immunology . 2017;8:p. 1647. doi: 10.3389/fimmu.2017.01647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Issenhuth-Jeanjean S., Roggentin P., Mikoleit M., et al. Supplement 2008–2010 (no. 48) to the white–Kauffmann–le minor scheme. Research in Microbiology . 2014;165(7):526–530. doi: 10.1016/j.resmic.2014.07.004. [DOI] [PubMed] [Google Scholar]
- 3.Ohl M. E. M. S., Miller S. I. Salmonella: a model for bacterial pathogenesis. Annual Review of Medicine . 2001;52(1):259–274. doi: 10.1146/annurev.med.52.1.259. [DOI] [PubMed] [Google Scholar]
- 4.Majowicz S. E., Musto J., Scallan E., et al. The global burden of nontyphoidal Salmonella gastroenteritis. Clinical Infectious Diseases . 2010;50(6):882–889. doi: 10.1086/650733. [DOI] [PubMed] [Google Scholar]
- 5.Kosek M., Bern C., Guerrant R. L. The global burden of diarrhoeal disease, as estimated from studies published between 1992 and 2000. Bulletin of the World Health Organization . 2003;81(3):197–204. [PMC free article] [PubMed] [Google Scholar]
- 6.Threlfall E. J., Bone A., Murdoch D., Banatvala N., Shoismatulloev B. I., Ward L. R. Epidemic ciprofloxacin-resistant Salmonella typhi in Tajikistan. The Lancet . 1998;351(9099):p. 339. doi: 10.1016/s0140-6736(05)78338-0. [DOI] [PubMed] [Google Scholar]
- 7.Levine M. M., Black R. E., Lanata C. Precise estimation of the numbers of chronic carriers of Salmonella typhi in Santiago, chile, an endemic area. The Journal of Infectious Diseases . 1982;146:724–726. doi: 10.1093/infdis/146.6.724. [DOI] [PubMed] [Google Scholar]
- 8.Vogelsang T. M., Bøe J. Temporary and chronic carriers of Salmonella typhi and salmonella paratyphi B. Journal of Hygiene . 1948;46(3):252–261. doi: 10.1017/s0022172400036378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Nath G., Singh Y. K., Maurya P., Gulati A. K., Srivastava R. C., Tripathi S. K. Does Salmonella Typhi primarily reside in the liver of chronic typhoid carriers? Journal of Infection in Developing Countries . 2010;4(4):259–261. doi: 10.3855/jidc.820. [DOI] [PubMed] [Google Scholar]
- 10.Crawford R. W., Rosales-Reyes R., Ramírez-Aguilar Mde L., Chapa-Azuela O., Alpuche-Aranda C., Gunn J. S. Gallstones play a significant role in Salmonella spp. gallbladder colonization and carriage. Proceedings of the National Academy of Sciences of the United States of America . 2010;107(9):4353–4358. doi: 10.1073/pnas.1000862107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Schiøler H., Christiansen E. D., Høybye G., Rasmussen S. N., Greibe J. Biliary calculi in chronic salmonella carriers and healthy controls: a controlled study. Scandinavian Journal of Infectious Diseases . 1983;15(1):17–19. doi: 10.3109/inf.1983.15.issue-1.04. [DOI] [PubMed] [Google Scholar]
- 12.Gunn J. S., Marshall J. M., Baker S., Dongol S., Charles R. C., Ryan E. T. Salmonella chronic carriage: epidemiology, diagnosis, and gallbladder persistence. Trends in Microbiology . 2014;22(11):648–655. doi: 10.1016/j.tim.2014.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dutta U., Garg P. K., Kumar R., Tandon R. K. Typhoid carriers among patients with gallstones are at increased risk for carcinoma of the gallbladder. American Journal of Gastroenterology . 2000;95(3):784–787. doi: 10.1111/j.1572-0241.2000.01860.x. [DOI] [PubMed] [Google Scholar]
- 14.DelGiorno K. E., Tam J. W., Hall J. C., Thotakura G., Crawford H. C., van der Velden A. W. M. Persistent salmonellosis causes pancreatitis in a murine model of infection. PLoS One . 2014;9(4) doi: 10.1371/journal.pone.0092807.e92807 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wilmes-Riesenberg M. R., Bearson B., Curtis R., CuRtis R. Role of the acid tolerance response in virulence of Salmonella typhimurium. Infection and Immunity . 1996;64(4):1085–1092. doi: 10.1128/iai.64.4.1085-1092.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Miller S. I. K. A., Kukral A. M., Mekalanos J. J. A two-component regulatory system (phoP phoQ) controls Salmonella typhimurium virulence. Proceedings of the National Academy of Sciences of the United States of America . 1989;86(13):5054–5058. doi: 10.1073/pnas.86.13.5054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bb F. Molecular and cellular mechanisms of Salmonella pathogenesis. Current Topics in Microbiology and Immunology . 1994;192:163–185. doi: 10.1007/978-3-642-78624-2_8. [DOI] [PubMed] [Google Scholar]
- 18.Khan C. M. A. The dynamic interactions between Salmonella and the microbiota, within the challenging niche of the gastrointestinal tract. International Scholarly Research Notices . 2014;2014:23. doi: 10.1155/2014/846049.846049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Que Ju H. D., Hentges D. J. Effect of streptomycin administration on colonization resistance to Salmonella typhimurium in mice. Infection and Immunity . 1985;48(1):169–174. doi: 10.1128/iai.48.1.169-174.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Barthel M., Hapfelmeier S., Quintanilla-Martinez L., et al. Pretreatment of mice with streptomycin provides a Salmonella enterica serovar typhimurium colitis model that allows analysis of both pathogen and host. Infection and Immunity . 2003;71(5):2839–2858. doi: 10.1128/iai.71.5.2839-2858.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sandra Y Wotzka M. K., Kreuzer M., Maier L., et al. Escherichia coli limits salmonella typhimurium infections after diet shifts and fat-mediated microbiota perturbation in mice. Nat Microbiol . 2019;4(12):2164–2174. doi: 10.1038/s41564-019-0568-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jabado N. J. A., Jankowski A., Picard V., Grinstein S., Gros P., Gros P. Natural resistance to intracellular infections: natural resistance-associated macrophage protein 1 (Nramp1) functions as a pH-dependent manganese transporter at the phagosomal membrane. Journal of Experimental Medicine . 2000;192(9):1237–1248. doi: 10.1084/jem.192.9.1237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Stecher B., Paesold G., Barthel M., et al. Chronic Salmonella enterica serovar typhimurium-induced colitis and cholangitis in streptomycin-pretreated nramp1+/+ mice. Infection and Immunity . 2006;74(9):5047–5057. doi: 10.1128/IAI.00072-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jones B. D., Ghori N., Falkow S. Salmonella typhimurium initiates murine infection by penetrating and destroying the specialized epithelial M cells of the peyer’s patches. Journal of Experimental Medicine . 1994;180(1):15–23. doi: 10.1084/jem.180.1.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kraehenbuhl J. P., Neutra M. R. Epithelial M cells: differentiation and function. Annual Review of Cell and Developmental Biology . 2000;16(1):301–332. doi: 10.1146/annurev.cellbio.16.1.301. [DOI] [PubMed] [Google Scholar]
- 26.Corr S. C., Gahan C. C., Hill C. M-cells: origin, morphology and role in mucosal immunity and microbial pathogenesis. FEMS Immunology and Medical Microbiology . 2008;52(1):2–12. doi: 10.1111/j.1574-695X.2007.00359.x. [DOI] [PubMed] [Google Scholar]
- 27.Tahoun A., Mahajan S., Paxton E., et al. Salmonella transforms follicle-associated epithelial cells into M cells to promote intestinal invasion. Cell Host & Microbe . 2012;12(5):645–656. doi: 10.1016/j.chom.2012.10.009. [DOI] [PubMed] [Google Scholar]
- 28.Schneider K. P. K., Potter K. G., Ware C. F. Lymphotoxin and LIGHT signaling pathways and target genes. Immunological Reviews . 2004;202(1):49–66. doi: 10.1111/j.0105-2896.2004.00206.x. [DOI] [PubMed] [Google Scholar]
- 29.Ding S., Song Y., Brulois K. F., et al. Retinoic acid and lymphotoxin signaling promote differentiation of human intestinal M cells. Gastroenterology . 2020;159(1):214–226. doi: 10.1053/j.gastro.2020.03.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Giles D. A., Zahner S., Krause P., et al. The tumor necrosis factor superfamily members TNFSF14 (LIGHT), lymphotoxin beta and lymphotoxin beta receptor interact to regulate intestinal inflammation. Frontiers in Immunology . 2018;9:p. 2585. doi: 10.3389/fimmu.2018.02585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sudhamsu J., Yin J., Chiang E. Y., Starovasnik M. A., Grogan J. L., Hymowitz S. G. Dimerization of LTβR by LTα1β2 is necessary and sufficient for signal transduction. Proceedings of the National Academy of Sciences of the United States of America . 2013;110(49):19896–19901. doi: 10.1073/pnas.1310838110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Fütterer A. M. K., Mink K., Kosco-Vilbois M. H., Pfeffer K., Pfeffer K. The lymphotoxin beta receptor controls organogenesis and affinity maturation in peripheral lymphoid tissues. Immunity . 1998;9(1):59–70. doi: 10.1016/s1074-7613(00)80588-9. [DOI] [PubMed] [Google Scholar]
- 33.Voedisch S., Koenecke C., David S., et al. Mesenteric lymph nodes confine dendritic cell-mediated dissemination of Salmonella enterica serovar typhimurium and limit systemic disease in mice. Infection and Immunity . 2009;77(8):3170–3180. doi: 10.1128/iai.00272-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Cheminay C., Schoen M., Hensel M., et al. Migration of Salmonella typhimurium -harboring bone marrow-derived dendritic cells towards the chemokines CCL19 and CCL21. Microbial Pathogenesis . 2002;32(5):207–218. doi: 10.1006/mpat.2002.0497. [DOI] [PubMed] [Google Scholar]
- 35.Worley M. J., Nieman G. S., Geddes K., Heffron F. Salmonella typhimurium disseminates within its host by manipulating the motility of infected cells. Proceedings of the National Academy of Sciences of the United States of America . 2006;103(47):17915–17920. doi: 10.1073/pnas.0604054103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Wroblewska J. A., Zhang Y., Tang H., Guo X., Nagler C., Fu Y. X. Cutting edge: lymphotoxin signaling is essential for clearance of Salmonella from the gut lumen and generation of anti-Salmonella protective immunity. The Journal of Immunology . 2017;198(1):55–60. doi: 10.4049/jimmunol.1600867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Gonzalez-Escobedo G., Gunn J. S. Gallbladder epithelium as a niche for chronic Salmonella carriage. Infection and Immunity . 2013;81(8):2920–2930. doi: 10.1128/IAI.00258-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Knodler L. A. C. S., Crowley S., Yang H., et al. Noncanonical inflammasome activation of caspase-4/caspase-11 mediates epithelial defenses against enteric bacterial pathogens. Cell Host & Microbe . 2014;16(2):249–256. doi: 10.1016/j.chom.2014.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Knodler L. A., Vallance B. A., Celli J., et al. Dissemination of invasive salmonella via bacterial-induced extrusion of mucosal epithelia. Proceedings of the National Academy of Sciences of the United States of America . 2010;107(41):17733–17738. doi: 10.1073/pnas.1006098107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lou L., Zhang P., Piao R., Wang Y. Salmonella pathogenicity island 1 (SPI-1) and its complex regulatory network. Frontiers in Cellular and Infection Microbiology . 2019;9:p. 270. doi: 10.3389/fcimb.2019.00270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Baumler A. J., Zhang K., Riba A., et al. Minimal SPI1-T3SS effector requirement for salmonella enterocyte invasion and intracellular proliferation in vivo. PLoS Pathogens . 2018;14(3) doi: 10.1371/journal.ppat.1006925.e1006925 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Bakowski M. A., Braun V., Brumell J. H. Salmonella-containing vacuoles: directing traffic and nesting to grow. Traffic . 2008;9(12):2022–2031. doi: 10.1111/j.1600-0854.2008.00827.x. [DOI] [PubMed] [Google Scholar]
- 43.Jantsch J. C. D., Chikkaballi D., Hensel M. Cellular aspects of immunity to intracellular Salmonella enterica. Immunological Reviews . 2011;240(1):185–195. doi: 10.1111/j.1600-065x.2010.00981.x. [DOI] [PubMed] [Google Scholar]
- 44.Malik-Kale P., Jolly C. E., Lathrop S., Winfree S., Luterbach C., Steele-Mortimer O. Salmonella—at home in the host cell. Frontiers in Microbiology . 2011;2:p. 125. doi: 10.3389/fmicb.2011.00125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Swart A. L., Hensel M. Interactions of Salmonella enterica with dendritic cells. Virulence . 2012;3:660–667. doi: 10.4161/viru.22761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Guignot J., Caron E., Beuzon C., et al. Microtubule motors control membrane dynamics of salmonella-containing vacuoles. Journal of Cell Science . 2004;117(7):1033–1045. doi: 10.1242/jcs.00949. [DOI] [PubMed] [Google Scholar]
- 47.Takeuchi A. Electron microscope studies of experimental salmonella infection. I. penetration into the intestinal epithelium by Salmonella typhimurium. American Journal of Pathology . 1967;50(1):109–136. [PMC free article] [PubMed] [Google Scholar]
- 48.Bäumler A. J. T. R., Tsolis R. M., Heffron F. Contribution of fimbrial operons to attachment to and invasion of epithelial cell lines by Salmonella typhimurium. Infection and Immunity . 1996;64:1862–1865. doi: 10.1128/iai.64.5.1862-1865.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Cossart P., Sansonetti P. J. Bacterial invasion: the paradigms of enteroinvasive pathogens. Science . 2004;304(5668):242–248. doi: 10.1126/science.1090124. [DOI] [PubMed] [Google Scholar]
- 50.Hanisch J., Stradal T. E., Rottner K. A novel contractility pathway operating in salmonella invasion. Virulence . 2012;3(1):81–86. doi: 10.4161/viru.3.1.18454. [DOI] [PubMed] [Google Scholar]
- 51.Patel J. C., Galan J. E. Manipulation of the host actin cytoskeleton by Salmonella-all in the name of entry. Current Opinion in Microbiology . 2005;8(1):10–15. doi: 10.1016/j.mib.2004.09.001. [DOI] [PubMed] [Google Scholar]
- 52.Rosselin M., Virlogeux-Payant I., Roy C., et al. Rck of Salmonella enterica, subspecies enterica serovar Enteritidis, mediates Zipper-like internalization. Cell Research . 2010;20(6):647–664. doi: 10.1038/cr.2010.45. [DOI] [PubMed] [Google Scholar]
- 53.Lambert M. A., Smith S. G. J. The PagN protein mediates invasion via interaction with proteoglycan. FEMS Microbiology Letters . 2009;297(2):209–216. doi: 10.1111/j.1574-6968.2009.01666.x. [DOI] [PubMed] [Google Scholar]
- 54.Heffernan E. J. W. L., Wu L., Okamoto S., Fierer J., Guiney D. G., Guiney D. G. Specificity of the complement resistance and cell association phenotypes encoded by the outer membrane protein genes rck from Salmonella typhimurium and ail from Yersinia enterocolitica. Infection and Immunity . 1994;62(11):5183–5186. doi: 10.1128/iai.62.11.5183-5186.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Lambert M. A., Smith S. G. The PagN protein of Salmonella enterica serovar typhimurium is an adhesin and invasin. BMC Microbiology . 2008;8(1):p. 142. doi: 10.1186/1471-2180-8-142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Kaur D., Mukhopadhaya A. Outer membrane protein OmpV mediates salmonella entericaserovar typhimurium adhesion to intestinal epithelial cells via fibronectin and α1β1 integrin. Cellular Microbiology . 2020;22(5) doi: 10.1111/cmi.13172.e13172 [DOI] [PubMed] [Google Scholar]
- 57.Takeda K., Kaisho T., Akira S. Toll-like receptors. Annual Review of Immunology . 2003;21(1):335–376. doi: 10.1146/annurev.immunol.21.120601.141126. [DOI] [PubMed] [Google Scholar]
- 58.Hapfelmeier S., Stecher B., Barthel M., et al. The Salmonella pathogenicity island (SPI)-2 and SPI-1 type III secretion systems allow Salmonella serovar typhimurium to trigger colitis via MyD88-dependent and MyD88-independent mechanisms. The Journal of Immunology . 2005;174(3):1675–1685. doi: 10.4049/jimmunol.174.3.1675. [DOI] [PubMed] [Google Scholar]
- 59.Jennings E., Thurston T. L. M., Holden D. W. Salmonella SPI-2 type III secretion system effectors: molecular mechanisms and physiological consequences. Cell Host & Microbe . 2017;22(2):217–231. doi: 10.1016/j.chom.2017.07.009. [DOI] [PubMed] [Google Scholar]
- 60.Hensel M., McLaughlin L. M., Govoni G. R., et al. The Salmonella SPI2 effector SseI mediates long-term systemic infection by modulating host cell migration. PLoS Pathogens . 2009;5(11) doi: 10.1371/journal.ppat.1000671.e1000671 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Jepson M. A., Kenny B., Leard A. D. Role of sipA in the early stages of Salmonella typhimurium entry into epithelial cells. Cellular Microbiology . 2001;3(6):417–426. doi: 10.1046/j.1462-5822.2001.00124.x. [DOI] [PubMed] [Google Scholar]
- 62.Zhou D., Mooseker M. S., Galán J. E. Role of the S. typhimurium actin-binding protein SipA in bacterial internalization. Science . 1999;283(5410):2092–2095. doi: 10.1126/science.283.5410.2092.101126 [DOI] [PubMed] [Google Scholar]
- 63.Raffatellu M., Wilson R. P., Chessa D., et al. SipA, SopA, SopB, SopD, and SopE2 contribute to Salmonella enterica serotype typhimurium invasion of epithelial cells. Infection and Immunity . 2005;73(1):146–154. doi: 10.1128/IAI.73.1.146-154.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Fattinger S. A., Sellin M. E., Hardt W. D. Epithelial inflammasomes in the defense against Salmonella gut infection. Current Opinion in Microbiology . 2021;59:86–94. doi: 10.1016/j.mib.2020.09.014. [DOI] [PubMed] [Google Scholar]
- 65.Galan J. E. Salmonella typhimurium and inflammation: a pathogen-centric affair. Nature Reviews Microbiology . 2021;19(11):716–725. doi: 10.1038/s41579-021-00561-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Feuillet V., Medjane S., Mondor I., et al. Involvement of Toll-like receptor 5 in the recognition of flagellated bacteria. Proceedings of the National Academy of Sciences of the United States of America . 2006;103:12487–12492. doi: 10.1073/pnas.0605200103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Gram A. M., Wright J. A., Pickering R. J., et al. Salmonella flagellin activates NAIP/NLRC4 and canonical NLRP3 inflammasomes in human macrophages. The Journal of Immunology . 2021;206(3):631–640. doi: 10.4049/jimmunol.2000382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Crowley S. M., Han X., Allaire J. M., et al. Intestinal restriction of Salmonella Typhimurium requires caspase-1 and caspase-11 epithelial intrinsic inflammasomes. PLoS Pathogens . 2020;16(4) doi: 10.1371/journal.ppat.1008498.e1008498 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Iwasaki A. K. B., Kelsall B. L. Unique functions of CD11b+, CD8α+, and double-negative Peyer’s patch dendritic cells. The Journal of Immunology . 2001;166(8):4884–4890. doi: 10.4049/jimmunol.166.8.4884. [DOI] [PubMed] [Google Scholar]
- 70.Maric I. H. P., Holt P. G., Bienenstock J., Bienenstock J. Class II MHC antigen (Ia)-bearing dendritic cells in the epithelium of the rat intestine. The Journal of Immunology . 1996;156(4):1408–1414. [PubMed] [Google Scholar]
- 71.Ruedl C. H. S., Hubele S. Maturation of Peyer’s patch dendritic cells in vitro upon stimulation via cytokines or CD40 triggering. European Journal of Immunology . 1997;27(6):1325–1330. doi: 10.1002/eji.1830270605. [DOI] [PubMed] [Google Scholar]
- 72.Kelsall B. L. S. W., Strober W. Distinct populations of dendritic cells are present in the subepithelial dome and T cell regions of the murine Peyer’s patch. Journal of Experimental Medicine . 1996;183(1):237–247. doi: 10.1084/jem.183.1.237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Iwasaki A. K. B., Kelsall B. L. Localization of distinct Peyer’s patch dendritic cell subsets and their recruitment by chemokines macrophage inflammatory protein (Mip)-3α, mip-3β, and secondary lymphoid organ chemokine. Journal of Experimental Medicine . 2000;191(8):1381–1394. doi: 10.1084/jem.191.8.1381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Cutler C. W., Jotwani R., Pulendran B. Dendritic cells: immune saviors or Achilles’ heel? Infection and Immunity . 2001;69:4703–4708. doi: 10.1128/IAI.69.8.4703-4708.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Tuladhar S., Kanneganti T. D. NLRP12 in innate immunity and inflammation. Molecular Aspects of Medicine . 2020;76 doi: 10.1016/j.mam.2020.100887.100887 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Ye C.-L., Qiao L., Li X., et al. Salmonella typhimurium interacts CD209 receptors to promote host dissemination and infection. Infection and Immunity . 2019;87 doi: 10.1128/IAI.00100-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Geijtenbeek T. B., Kwon D. S., Torensma R., et al. DC-SIGN, a dendritic cell–specific HIV-1-Binding protein that enhances trans-infection of T cells. Cell . 2000;100(5):587–597. doi: 10.1016/s0092-8674(00)80694-7. [DOI] [PubMed] [Google Scholar]
- 78.Geijtenbeek T. B. H., van Kooyk Y. Pathogens target DC-SIGN to influence their fate DC-SIGN functions as a pathogen receptor with broad specificity. Acta Pathologica, Microbiologica et Immunologica Scandinavica . 2003;111(7-8):698–714. doi: 10.1034/j.1600-0463.2003.11107803.x. [DOI] [PubMed] [Google Scholar]
- 79.McDonald D., Wu L., Bohks S. M., KewalRamani V. N., Unutmaz D., Hope T. J. Recruitment of HIV and its receptors to dendritic cell-T cell junctions. Science . 2003;300(5623):1295–1297. doi: 10.1126/science.1084238. [DOI] [PubMed] [Google Scholar]
- 80.Tailleux L., Pham-Thi N., Bergeron-Lafaurie A., et al. DC-SIGN induction in alveolar macrophages defines privileged target host cells for mycobacteria in patients with tuberculosis. PLoS Medicine . 2005;2(12) doi: 10.1371/journal.pmed.0020381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.He Y. X., Ye C. L., Zhang P., et al. Yersinia pseudotuberculosis exploits CD209 receptors for promoting host dissemination and infection. Infection and Immunity . 2019;87(1) doi: 10.1128/IAI.00654-18.e00654-18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Klena J., Zhang P., Schwartz O., Hull S., Chen T. The core lipopolysaccharide of Escherichia coli is a ligand for the dendritic-cell-specific intercellular adhesion molecule nonintegrin CD209 receptor. Journal of Bacteriology . 2005;187:1710–1715. doi: 10.1128/JB.187.5.1710-1715.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Yang K., He Y., Park C. G., et al. Yersinia pestis interacts with SIGNR1 (CD209b) for promoting host dissemination and infection. Frontiers in Immunology . 2019;10:p. 96. doi: 10.3389/fimmu.2019.00096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Yang K., Park C. G., Cheong C., et al. Host langerin (CD207) is a receptor for Yersinia pestis phagocytosis and promotes dissemination. Immunology & Cell Biology . 2015;93(9):815–824. doi: 10.1038/icb.2015.46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Zhang P., Schwartz O., Pantelic M., et al. Dc-SIGN (DC-SIGN (CD209) recognition of Neisseria gonorrhoeae is circumvented by lipooligosaccharide variation. Journal of Leukocyte Biology . 2006;79(4):731–738. doi: 10.1189/jlb.0405184. [DOI] [PubMed] [Google Scholar]
- 86.Zhang P., Skurnik M., Zhang S. S., et al. Human dendritic cell-specific intercellular adhesion molecule-grabbing nonintegrin (CD209) is a receptor for Yersinia pestis that promotes phagocytosis by dendritic cells. Infection and Immunity . 2008;76(5):2070–2079. doi: 10.1128/IAI.01246-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Zhang P., Snyder S., Feng P., et al. Role of N-acetylglucosamine within core lipopolysaccharide of several species of gram-negative bacteria in targeting the DC-SIGN (CD209) The Journal of Immunology . 2006;177(6):4002–4011. doi: 10.4049/jimmunol.177.6.4002. [DOI] [PubMed] [Google Scholar]
- 88.Zhang S. S., Park C. G., Zhang P., et al. Plasminogen activator Pla of Yersinia pestis utilizes murine DEC-205 (CD205) as a receptor to promote dissemination. Journal of Biological Chemistry . 2008;283(46):31511–31521. doi: 10.1074/jbc.M804646200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Cheminay C., Möhlenbrink A., Hensel M. Intracellular Salmonella Inhibit antigen presentation by dendritic cells. The Journal of Immunology . 2005;174(5):2892–2899. doi: 10.4049/jimmunol.174.5.2892. [DOI] [PubMed] [Google Scholar]
- 90.Lapaque N., Hutchinson J. L., Jones D. C., et al. Salmonella regulates polyubiquitination and surface expression of MHC class II antigens. Proceedings of the National Academy of Sciences of the United States of America . 2009;106(33):14052–14057. doi: 10.1073/pnas.0906735106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Farache J., Koren I., Milo I., et al. Luminal bacteria recruit CD103+ dendritic cells into the intestinal epithelium to sample bacterial antigens for presentation. Immunity . 2013;38(3):581–595. doi: 10.1016/j.immuni.2013.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Uematsu S., Jang M. H., Chevrier N., et al. Detection of pathogenic intestinal bacteria by Toll-like receptor 5 on intestinal CD11c+ lamina propria cells. Nature Immunology . 2006;7(8):868–874. doi: 10.1038/ni1362. [DOI] [PubMed] [Google Scholar]
- 93.Uematsu S., Akira S. Immune responses of TLR5 (+) lamina propria dendritic cells in enterobacterial infection. Journal of Gastroenterology . 2009;44(8):803–811. doi: 10.1007/s00535-009-0094-y. [DOI] [PubMed] [Google Scholar]
- 94.Rescigno M. CCR6 (+) dendritic cells: the gut tactical-response unit. Immunity . 2006;24(5):508–510. doi: 10.1016/j.immuni.2006.05.005. [DOI] [PubMed] [Google Scholar]
- 95.Salazar-Gonzalez R. M., Niess J. H., Zammit D. J., et al. CCR6-mediated dendritic cell activation of pathogen-specific T cells in Peyer’s patches. Immunity . 2006;24(5):623–632. doi: 10.1016/j.immuni.2006.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Niess J. H. B. S., Brand S., Landsman L., et al. CX3CR1-mediated dendritic cell access to the intestinal lumen and bacterial clearance. Science . 2005;307(5707):254–258. doi: 10.1126/science.1102901. [DOI] [PubMed] [Google Scholar]
- 97.Hopkins S. A. N. F., Corthesy-Theulaz I. E., Kraehenbuhl J. P. A recombinant Salmonella typhimurium vaccine strain is taken up and survives within murine Peyer’s patch dendritic cells. Cellular Microbiology . 2000;2(1):59–68. doi: 10.1046/j.1462-5822.2000.00035.x. [DOI] [PubMed] [Google Scholar]
- 98.Gunn J. S., Miller S. I., Miller S. PhoP-PhoQ activates transcription of pmrAB, encoding a two-component regulatory system involved in Salmonella typhimurium antimicrobial peptide resistance. Journal of Bacteriology . 1996;178(23):6857–6864. doi: 10.1128/jb.178.23.6857-6864.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Rescigno M., Urbano M., Valzasina B., et al. Dendritic cells express tight junction proteins and penetrate gut epithelial monolayers to sample bacteria. Nature Immunology . 2001;2(4):361–367. doi: 10.1038/86373. [DOI] [PubMed] [Google Scholar]
- 100.Vazquez-Torres A. J.-C. J., Jones-Carson J., Bäumler A. J., et al. Extraintestinal dissemination of Salmonella by CD18-expressing phagocytes. Nature . 1999;401(6755):804–808. doi: 10.1038/44593. [DOI] [PubMed] [Google Scholar]
- 101.Uchiya K. I., Nikai T. Salmonella pathogenicity island 2-dependent expression of suppressor of cytokine signaling 3 in macrophages. Infection and Immunity . 2005;73(9):5587–5594. doi: 10.1128/IAI.73.9.5587-5594.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Wijburg O. L. C., Simmons C. P., Strugnell R. A., Strugnell R. A. Dual role for macrophages in vivo in pathogenesis and control of murine Salmonella enterica var. typhimurium infections. European Journal of Immunology . 2000;30(3):944–953. doi: 10.1002/1521-4141(200003)30:3<944::aid-immu944>3.0.co;2-1. [DOI] [PubMed] [Google Scholar]
- 103.Alpuche-Aranda C. M. R. E., Racoosin E. L., Miller S. I., Miller S. I. Salmonella stimulate macrophage macropinocytosis and persist within spacious phagosomes. Journal of Experimental Medicine . 1994;179(2):601–608. doi: 10.1084/jem.179.2.601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Vazquez-Torres A., Xu Y., Jones-Carson J., et al. Salmonella pathogenicity island 2-dependent evasion of the phagocyte NADPH oxidase. Science . 2000;287(5458):1655–1658. doi: 10.1126/science.287.5458.1655. [DOI] [PubMed] [Google Scholar]
- 105.Gogoi M., Shreenivas M. M., Chakravortty D. Hoodwinking the big-eater to prosper: the salmonella-macrophage paradigm. Journal of Innate Immunity . 2019;11(3):289–299. doi: 10.1159/000490953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Alpuche Aranda C. M. S. J., Swanson J. A., Miller S. I., Miller S. I. Salmonella typhimurium activates virulence gene transcription within acidified macrophage phagosomes. Proceedings of the National Academy of Sciences of the United States of America . 1992;89(21):10079–10083. doi: 10.1073/pnas.89.21.10079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Eriksson S. L. S., Lucchini S., Rhen M., Hinton J. C., Hinton J. C. D. Unravelling the biology of macrophage infection by gene expression profiling of intracellular Salmonella enterica. Molecular Microbiology . 2003;47(1):103–118. doi: 10.1046/j.1365-2958.2003.03313.x. [DOI] [PubMed] [Google Scholar]
- 108.Lähteenmäki K., Kyllönen P., Partanen L., Korhonen T. K. Antiprotease inactivation by Salmonella enterica released from infected macrophages. Cellular Microbiology . 2004;7(4):529–538. doi: 10.1111/j.1462-5822.2004.00483.x. [DOI] [PubMed] [Google Scholar]
- 109.Monack D. M., Mueller A., Falkow S. Persistent bacterial infections: the interface of the pathogen and the host immune system. Nature Reviews Microbiology . 2004;2(9):747–765. doi: 10.1038/nrmicro955. [DOI] [PubMed] [Google Scholar]
- 110.Eisele N. A., Ruby T., Jacobson A., et al. Salmonella require the fatty acid regulator PPARδ for the establishment of a metabolic environment essential for long-term persistence. Cell Host & Microbe . 2013;14(2):171–182. doi: 10.1016/j.chom.2013.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Saliba A. E., Li L., Westermann A. J., et al. Single-cell RNA-seq ties macrophage polarization to growth rate of intracellular Salmonella. Nature Microbiology . 2016;2 doi: 10.1038/nmicrobiol.2016.206.16206 [DOI] [PubMed] [Google Scholar]
- 112.Roop R. M., Caswell C. C. Bacterial persistence: finding the “sweet spot”. Cell Host & Microbe . 2013;14(2):119–120. doi: 10.1016/j.chom.2013.07.016. [DOI] [PubMed] [Google Scholar]
- 113.Pham T. H. M., Brewer S. M., Thurston T., et al. Salmonella-driven polarization of granuloma macrophages antagonizes TNF-mediated pathogen restriction during persistent infection. Cell Host & Microbe . 2020;27(1):54–67. doi: 10.1016/j.chom.2019.11.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Guiney D. G. The role of host cell death in Salmonella infections. Current Topics in Microbiology and Immunology . 2005;289 doi: 10.1007/3-540-27320-4_6. [DOI] [PubMed] [Google Scholar]
- 115.Monack D. M. H. D., Hersh D., Bouley D., Zychlinsky A., Falkow S., Falkow S. Salmonella exploits caspase-1 to colonize Peyer’s patches in a murine typhoid model. Journal of Experimental Medicine . 2000;192(2):249–258. doi: 10.1084/jem.192.2.249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Miao E. A., Rajan J. V. Salmonella and caspase-1: a complex interplay of detection and evasion. Frontiers in Microbiology . 2011;2:p. 85. doi: 10.3389/fmicb.2011.00085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Holly M. K., Han X., Zhao E. J., et al. Salmonella enterica infection of murine and human enteroid-derived monolayers elicits differential activation of epithelium-intrinsic inflammasomes. Infection and Immunity . 2020;88(7) doi: 10.1128/IAI.00017-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Lin H. H., Chen H. L., Weng C. C., Janapatla R. P., Chen C. L., Chiu C. H. Activation of apoptosis by salmonella pathogenicity island-1 effectors through both intrinsic and extrinsic pathways in Salmonella-infected macrophages. Journal of Microbiology, Immunology, and Infection . 2021;54(4):616–626. doi: 10.1016/j.jmii.2020.02.008. [DOI] [PubMed] [Google Scholar]
- 119.Johanns T. M., Ertelt J. M., Rowe J. H., Way S. S. Regulatory T cell suppressive potency dictates the balance between bacterial proliferation and clearance during persistent Salmonella infection. PLoS Pathogens . 2010;6(8) doi: 10.1371/journal.ppat.1001043.e1001043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Helaine S., Cheverton A. M., Watson K. G., Faure L. M., Matthews S. A., Holden D. W. Internalization of Salmonella by macrophages induces formation of nonreplicating persisters. Science . 2014;343(6167):204–208. doi: 10.1126/science.1244705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Lewis K. Persister cells. Annual Review of Microbiology . 2010;64(1):357–372. doi: 10.1146/annurev.micro.112408.134306. [DOI] [PubMed] [Google Scholar]
- 122.Fisher R. A., Gollan B., Helaine S. Persistent bacterial infections and persister cells. Nature Reviews Microbiology . 2017;15(8):453–464. doi: 10.1038/nrmicro.2017.42. [DOI] [PubMed] [Google Scholar]
- 123.Stapels D. A. C., Hill P. W. S., Westermann A. J., et al. Salmonella persisters undermine host immune defenses during antibiotic treatment. Science . 2018;362(6419):1156–1160. doi: 10.1126/science.aat7148. [DOI] [PubMed] [Google Scholar]
- 124.Abdallah M., Benoliel C., Drider D., Dhulster P., Chihib N. E. Biofilm formation and persistence on abiotic surfaces in the context of food and medical environments. Archives of Microbiology . 2014;196(7):453–472. doi: 10.1007/s00203-014-0983-1. [DOI] [PubMed] [Google Scholar]
- 125.Romling U., Balsalobre C. Biofilm infections, their resilience to therapy and innovative treatment strategies. Journal of Internal Medicine . 2012;272(6):541–561. doi: 10.1111/joim.12004. [DOI] [PubMed] [Google Scholar]
- 126.Feasey N. A., Dougan G., Kingsley R. A., Heyderman R. S., Gordon M. A. Invasive non-typhoidal salmonella disease: an emerging and neglected tropical disease in Africa. The Lancet . 2012;379(9835):2489–2499. doi: 10.1016/s0140-6736(11)61752-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Ramachandran G., Aheto K., Shirtliff M. E., Tennant S. M., Kaper J. Poor biofilm-forming ability and long-term survival of invasiveSalmonellaTyphimurium ST313. Pathogens and Disease . 2016;74(5) doi: 10.1093/femspd/ftw049.ftw049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Luk C. H., Valenzuela C., Gil M., et al. Salmonella enters a dormant state within human epithelial cells for persistent infection. PLoS Pathogens . 2021;17(4) doi: 10.1371/journal.ppat.1009550.e1009550 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Carden S. E., Walker G. T., Honeycutt J., et al. Pseudogenization of the secreted effector gene sseI confers rapid systemic dissemination of S. Typhimurium ST313 within migratory dendritic cells. Cell Host & Microbe . 2017;21(2):182–194. doi: 10.1016/j.chom.2017.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Niedergang F. S. J., Sirard J. C., Kraehenbuhl J. P., Kraehenbuhl J. P. Entry and survival of Salmonella typhimurium in dendritic cells and presentation of recombinant antigens do not require macrophage-specific virulence factors. Proceedings of the National Academy of Sciences of the United States of America . 2000;97(26):14650–14655. doi: 10.1073/pnas.97.26.14650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Mughini-Gras L., Schaapveld M., Kramers J., et al. Increased colon cancer risk after severe salmonella infection. PLoS One . 2018;13(1) doi: 10.1371/journal.pone.0189721.e0189721 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Lu R. B. M., Bosland M., Zhang Y. G., Kato I., Sun J., Sun J. Presence of Salmonella AvrA in colorectal tumor and its precursor lesions in mouse intestine and human specimens. Oncotarget . 2017;8(33):55104–55115. doi: 10.18632/oncotarget.19052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Ehrhardt K., Steck N., Kappelhoff R., et al. Persistent Salmonella enterica serovar typhimurium infection induces protease expression during intestinal fibrosis. Inflammatory Bowel Diseases . 2019;25(10):1629–1643. doi: 10.1093/ibd/izz070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Grassl G. A., Valdez Y., Bergstrom K. S., Vallance B. A., Finlay B. B. Chronic enteric salmonella infection in mice leads to severe and persistent intestinal fibrosis. Gastroenterology . 2008;134(3):768–780. doi: 10.1053/j.gastro.2007.12.043. [DOI] [PubMed] [Google Scholar]
- 135.Månsson L. E., Montero M., Zarepour M., et al. MyD88 signaling promotes both mucosal homeostatic and fibrotic responses during Salmonella-induced colitis. American Journal of Physiology—Gastrointestinal and Liver Physiology . 2012;303(3):G311–G323. doi: 10.1152/ajpgi.00038.2012. [DOI] [PubMed] [Google Scholar]
- 136.Schultz B. M., Paduro C. A., Salazar G. A., et al. A potential role of salmonella infection in the onset of inflammatory bowel diseases. Frontiers in Immunology . 2017;8:p. 191. doi: 10.3389/fimmu.2017.00191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Schultz B. M., Salazar G. A., Paduro C. A., et al. Persistent Salmonella enterica serovar typhimurium infection increases the susceptibility of mice to develop intestinal inflammation. Frontiers in Immunology . 2018;9:p. 1166. doi: 10.3389/fimmu.2018.01166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Griffin A. J., Li L. X., Voedisch S., Pabst O., McSorley S. J. Dissemination of persistent intestinal bacteria via the mesenteric lymph nodes causes typhoid relapse. Infection and Immunity . 2011;79(4):1479–1488. doi: 10.1128/IAI.01033-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Kurtz J. R., Nieves W., Bauer D., et al. Salmonella persistence and host immunity is dictated by the anatomical microenvironment. Infect Immun . 2020;88 doi: 10.1128/IAI.00026-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Behnsen J., Jellbauer S., Wong C. P., et al. The cytokine IL-22 promotes pathogen colonization by suppressing related commensal bacteria. Immunity . 2014;40(2):262–273. doi: 10.1016/j.immuni.2014.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Blaschitz C., Raffatellu M. Th17 cytokines and the gut mucosal barrier. Journal of Clinical Immunology . 2010;30(2):196–203. doi: 10.1007/s10875-010-9368-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Khader S. A., Gopal R. IL-17 in protective immunity to intracellular pathogens. Virulence . 2010;1(5):423–427. doi: 10.4161/viru.1.5.12862. [DOI] [PMC free article] [PubMed] [Google Scholar]


