Abstract
Background
Alcohol use disorder (AUD) is one of the most widespread psychiatric disorders leading to detrimental consequences to people with this disorder and others. Worldwide, the prevalence of heavy episodic drinking (30‐day prevalence of at least one occasion of 60 g of pure alcohol intake among current drinkers) is estimated at 20% and the prevalence of AUD at 5% of the adult general population, with highest prevalence in Europe and North America. Therapeutic approaches, including pharmacotherapy, play an important role in treating people with AUD.
This is an update of a Cochrane Review first published in 2018.
Objectives
To evaluate the benefits and harms of baclofen on achieving and maintaining abstinence or reducing alcohol consumption in people with AUD compared to placebo, no treatment or any other pharmacological relapse prevention treatment.
Search methods
We used standard, extensive Cochrane search methods. The latest search was 22 November 2021.
Selection criteria
Randomised controlled trials (RCTs) of at least four weeks' treatment duration and 12 weeks' overall study duration comparing baclofen for AUD treatment with placebo, no treatment or other treatments.
Data collection and analysis
We used standard Cochrane methods. Our primary outcomes were 1. relapse, 2. frequency of use, 3. amount of use, 4. adverse events, 5. dropouts from treatment and 6. dropouts from treatment due to adverse events. Our secondary outcomes were 7. craving, 8. anxiety, 9. depression and 10. frequency of most relevant adverse events.
Main results
We included 17 RCTs (1818 participants) with a diagnosis of alcohol dependence according to the Diagnostic and Statistical Manual of Mental Disorders, 4th edition or International Classification of Diseases 10th edition criteria. Mean age was 46.5 years and 70% were men. Ten studies compared baclofen to placebo or another medication; seven compared two baclofen doses to placebo or another medication. Globally, 15 studies compared baclofen to placebo, two baclofen to acamprosate and two baclofen to naltrexone. In 16 studies, participants received psychosocial treatments.
We judged most studies at low risk of selection, performance, detection (subjective outcome), attrition and reporting bias.
Ten studies detoxified participants before treatment; in seven studies, participants were still drinking at the beginning of treatment. Treatment duration was 12 weeks for 15 RCTs and longer in two studies. Baclofen daily dose was 30 mg to 300 mg: 10 RCTs used low doses (30 mg or less); eight RCTs medium doses (above 30 and 100 mg or less) and four RCTs high doses (above 100 mg).
Compared to placebo, moderate‐certainty evidence found that baclofen probably decreases the risk to relapse (risk ratio (RR) 0.87, 95% confidence interval (CI) 0.77 to 0.99; 12 studies, 1057 participants). This result was confirmed among detoxified participants but not among other subgroups of participants.
High‐certainty evidence found that baclofen increases the percentage of days abstinent (mean difference (MD) 9.07, 95% CI 3.30 to 14.85; 16 studies, 1273 participants). This result was confirmed among all subgroups of participants except non‐detoxified or those who received medium doses.
There was no difference between baclofen and placebo in the other primary outcomes: heavy drinking days (standardised mean difference (SMD) −0.18, 95% CI −0.48 to 0.11; 13 studies, 840 participants; moderate‐certainty evidence); number of drinks per drinking days (MD −0.45, 95% CI −1.20 to 0.30; 9 studies, 392 participants; moderate‐certainty evidence); number of participants with at least one adverse event (RR 1.05, 95% CI 0.99 to 1.11; 10 studies, 738 participants; high‐certainty evidence); dropouts (RR 0.88, 95% CI 0.74 to 1.03; 17 studies, 1563 participants; high‐certainty evidence); dropouts due to adverse events (RR 1.39, 95% CI 0.89 to 2.18; 16 studies, 1499 participants; high‐certainty evidence). These results were confirmed by subgroup analyses except than for the dropouts that resulted lower among participants who received high doses of baclofen and studies longer than 12 weeks.
Compared to placebo, there was no difference in craving (SMD −0.16, 95% CI −0.37 to 0.04; 17 studies, 1275 participants), anxiety (MD −0.01, 95% CI −0.14 to 0.11; 15 studies, 1123 participants) and depression (SMD 0.07, 95% CI −0.12 to 0.27; 11 studies, 1029 participants).
Concerning the specific adverse events, baclofen increases fatigue, dizziness, somnolence/sedation, dry mouth, paraesthesia and muscle spasms/rigidity. There was no difference in the other adverse events.
Compared to acamprosate, one study (60 participants) found no differences in any outcomes but the evidence was very uncertain: relapse (RR 1.25, 95% CI 0.71 to 2.20; very low‐certainty evidence); number of participants with at least one adverse event (RR 0.63, 95% CI 0.23 to 1.69; very low‐certainty evidence); dropouts (RR 0.56, 95% CI 0.21 to 1.46; very low‐certainty evidence); dropouts due to adverse events (RR 0.33, 95% CI 0.01 to 7.87; very low‐certainty evidence) and craving (MD 5.80, 95% CI −11.84 to 23.44); and all the adverse events evaluated.
Compared to naltrexone, baclofen may increase the risk of relapse (RR 2.50, 95% CI 1.12 to 5.56; 1 study, 60 participants; very low‐certainty evidence) and decrease the number of participants with at least one adverse event (RR 0.35, 95% CI 0.15 to 0.80; 2 studies, 80 participants; very low‐certainty evidence) but the evidence is very uncertain. One study (60 participants) found no difference between baclofen and naltrexone in the dropouts at the end of treatment (RR 1.00, 95% CI 0.32 to 3.10; very low‐certainty evidence), craving (MD 2.08, 95% CI −3.71 to 7.87), and all the adverse events evaluated.
Authors' conclusions
Baclofen likely reduces the risk of relapse to any drinking and increases the percentage of abstinent days, mainly among detoxified participants. It does not increase the number of participants with at least one adverse event, those who dropout for any reason or due to adverse events. It probably does not reduce number of heavy drinking days and the number of drinks per drinking days. Current evidence suggests that baclofen may help people with AUD in maintaining abstinence. The results of comparisons of baclofen with acamprosate and naltrexone were mainly based on only one study.
Keywords: Adult, Female, Humans, Male, Middle Aged, Acamprosate, Acamprosate/adverse effects, Acamprosate/therapeutic use, Alcohol Drinking, Alcohol Drinking/drug therapy, Alcoholism, Alcoholism/drug therapy, Baclofen, Baclofen/adverse effects, Baclofen/therapeutic use, Chronic Disease, Naltrexone, Naltrexone/adverse effects, Naltrexone/therapeutic use
Plain language summary
Baclofen for alcohol use disorder
Key messages of the review
We reviewed the evidence about the effectiveness and safety of baclofen for treating people with alcohol use disorder (AUD) in order to achieve and maintain abstinence (stopping drinking) or reduce alcohol consumption.
Current evidence suggests that it may help people with AUD in maintaining abstinence, particularly in people who are already detoxified. The results of comparisons with other medications were mainly based on a single study and do not allow us to draw conclusions.
Review topics and aims
AUD is one of the most widespread psychiatric disorders, leading to specific physical, mood, learning and memory problems, and consequences for overall well‐being and health. The misuse of alcohol is one of the biggest risks to health worldwide, causing 20% to 30% of oesophageal (food pipe) cancer, liver disease, epilepsy (fits), motor vehicle accidents, murders and other intentional injuries.
For many years, the main treatments for AUD have been psychosocial strategies (helping people to recognise that they need help), but using only psychosocial treatments has limited success. A high proportion of people with AUD do not respond to treatment at all, and those who do respond do not stay alcohol‐free in the long‐term. Medications such as baclofen could play an important role in treating people with AUD.
Review methods
We searched for randomised controlled trials (studies where people were allocated at random to one of two or more treatments or control groups) evaluating the effect of baclofen in reducing alcohol consumption or in achieving and maintaining abstinence (or both) when compared to placebo (inactive medication) or other medications. We pooled similar studies and evaluated the effects dividing the studies according to the doses of baclofen, duration of treatment, and alcohol consumption and the beginning of treatment (i.e. into detoxified or non‐detoxified participants on the basis if they were abstinent or were still drinking at the beginning of treatment).
Summary of results
We found 17 studies with 1818 participants with AUD. The duration of the interventions ranged from three months to one year. Five studies were in the USA; two each in Australia, France, India and Italy; and one each in Germany, Israel and the Netherlands.
Doses of baclofen were 30 mg a day to 300 mg a day, and, in some cases, the doses were increased during the treatment. Seventeen studies compared baclofen to placebo (dummy medication), two compared baclofen to acamprosate (medication used to treat AUD), and two compared baclofen to naltrexone (medication used to treat AUD).
Compared with placebo, baclofen probably helps people with AUD at reducing the risk of relapse and increasing the rate of abstinent days. These effects may be more evident among detoxified than non‐detoxified people with AUD. Baclofen probably makes no difference in the rate of heavy drinking days; drinks per drinking days; craving, anxiety and depression severity; number of participants who dropout from treatment for any reason; those who dropped out due to adverse events (side effects); or the number of participants with at least one adverse event. Baclofen may increase adverse events such as fatigue, vertigo (a feeling that everything is spinning), sleepiness, dry mouth, numbness and muscle spasm but we found no differences between baclofen and placebo for other adverse events.
Certainty of evidence The certainty of evidence (how much we can be confident that the evidence is reliable) was high for results about the percentage of days of abstinence, the number of participants with at least one adverse event, people dropping out of the studies for any reason, and people dropping out due to adverse events. The certainty of the evidence was moderate for results about returning to any drinking, heavy drinking days and number of drinks per drinking days.
Search date The evidence is current to November 2021. This is an update of a Cochrane Review first published in 2018.
Summary of findings
Summary of findings 1. Summary of findings table ‐ Baclofen compared to placebo for alcohol use disorder.
| Baclofen compared to placebo for alcohol use disorder | ||||||
| Patient or population: alcohol use disorder Setting: outpatients Intervention: baclofen Comparison: placebo | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | Comments | |
| Risk with placebo | Risk with baclofen | |||||
| Relapse: return to any drinking at end of treatment | 816 per 1000 | 710 per 1000 (628 to 808) | RR 0.87 (0.77 to 0.99) | 1057 (12 RCTs) | ⊕⊕⊕⊝ Moderatea | Compared to placebo, baclofen probably decreases the risk to relapse to any drinking. |
| Frequency of use: % days abstinence at end of treatment | The mean frequency of use: % days abstinence at end of treatment was 53.58 | MD 9.07 higher (3.3 higher to 14.85 higher) | ‐ | 1273 (16 RCTs) | ⊕⊕⊕⊕ High | Compared to placebo, baclofen increases the % of days abstinent. |
| Frequency of use: heavy drinking days at end of treatment | ‐ | SMD 0.18 lower (0.48 lower to 0.11 higher) | ‐ | 840 (13 RCTs) | ⊕⊕⊕⊝ Moderateb | Compared to placebo, baclofen may not reduce heavy drinking days. |
| Amount of use: drink per drinking days at end of treatment | The mean amount of use: drink per drinking days at end of treatment was 4.28 | MD 0.45 lower (1.2 lower to 0.3 higher) | ‐ | 392 (9 RCTs) | ⊕⊕⊕⊝ Moderatec | Compared to placebo, baclofen probably does not reduce the number of drinks per drinking days. |
| Adverse events: number of participants with ≥ 1 adverse event at end of treatment | 636 per 1000 | 668 per 1000 (629 to 706) | RR 1.05 (0.99 to 1.11) | 738 (10 RCTs) | ⊕⊕⊕⊕ High | Compared to placebo, baclofen does not increase the number of participants with ≥ 1 adverse event at the end of treatment. |
| Dropouts at end of treatment | 420 per 1000 | 369 per 1000 (311 to 432) | RR 0.88 (0.74 to 1.03) | 1563 (17 RCTs) | ⊕⊕⊕⊕ High | Compared to placebo, baclofen does not increase the number of participants who dropout at the end of treatment. |
| Dropouts due to adverse events | 44 per 1000 | 61 per 1000 (39 to 95) | RR 1.39 (0.89 to 2.18) | 1499 (16 RCTs) | ⊕⊕⊕⊕ High | Compared to placebo, baclofen does not increase the number of dropouts due to adverse events. |
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; MD: mean difference; RR: risk ratio; SMD: standardised mean difference | ||||||
| GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | ||||||
| See interactive version of this table: https://gdt.gradepro.org/presentations/#/isof/isof_question_revman_web_432179672169955326. | ||||||
a Downgraded one level for inconsistency I² = 73%. b Downgraded one level for inconsistency I² = 71%. c Downgraded one level for risk of bias: one study at high risk for attrition and reporting bias.
Summary of findings 2. Summary of findings table ‐ Baclofen compared to acamprosate for alcohol use disorder.
| Baclofen compared to acamprosate for alcohol use disorder | ||||||
| Patient or population: alcohol use disorder Setting: Intervention: baclofen Comparison: acamprosate | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | Comments | |
| Risk with acamprosate | Risk with baclofen | |||||
| Relapse: return to any drinking at end of treatment | 400 per 1000 | 500 per 1000 (284 to 880) | RR 1.25 (0.71 to 2.20) | 60 (1 RCT) | ⊕⊝⊝⊝ Very lowa,b | It is uncertain whether baclofen and acamprosate differ in the return to any drinking. |
| Adverse events: number of participants with at least one adverse event at end of treatment | 267 per 1000 | 168 per 1000 (61 to 451) | RR 0.63 (0.23 to 1.69) | 60 (1 RCT) | ⊕⊝⊝⊝ Very lowa,b | It is uncertain whether baclofen and acamprosate differ in the number of participants with ≥ 1 adverse event at the end of treatment. |
| Dropouts at end of treatment | 300 per 1000 | 168 per 1000 (63 to 438) | RR 0.56 (0.21 to 1.46) | 60 (1 RCT) | ⊕⊝⊝⊝ Very lowa,b | It is uncertain whether baclofen and acamprosate differ in dropouts at the end of treatment. |
| Dropouts due to adverse events | 33 per 1000 | 11 per 1000 (0 to 262) | RR 0.33 (0.01 to 7.87) | 60 (1 RCT) | ⊕⊝⊝⊝ Very lowa,b | It is uncertain whether baclofen and acamprosate differ in dropouts due to adverse events at the end of treatment. |
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; RR: risk ratio | ||||||
| GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | ||||||
| See interactive version of this table: https://gdt.gradepro.org/presentations/#/isof/isof_question_revman_web_432318545502233572. | ||||||
a Downgraded one level: one study at high risk of performance, detection, and attrition bias and at unclear risk of selection bias. b Downgraded two levels for imprecision: fewer than 100 events.
Summary of findings 3. Summary of findings table ‐ Baclofen compared to naltrexone for alcohol use disorder.
| Baclofen compared to naltrexone for alcohol use disorder | ||||||
| Patient or population: alcohol use disorder Setting: Intervention: baclofen Comparison: naltrexone | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | Comments | |
| Risk with naltrexone | Risk with baclofen | |||||
| Relapse: return to any drinking at end of treatment | 200 per 1000 | 500 per 1000 (224 to 1000) | RR 2.50 (1.12 to 5.56) | 60 (1 RCT) | ⊕⊝⊝⊝ Very lowa,b | It is uncertain whether baclofen and naltrexone differ in the risk to return to any drinking. |
| Adverse events: number of participants with ≥ 1 adverse event at end of treatment | 450 per 1000 | 158 per 1000 (68 to 360) | RR 0.35 (0.15 to 0.80) | 80 (2 RCTs) | ⊕⊝⊝⊝ Very lowa,b | It is uncertain whether baclofen and naltrexone differ in the number of participants with ≥ 1 adverse event at the end of treatment. |
| Dropouts at end of treatment | 167 per 1000 | 167 per 1000 (53 to 517) | RR 1.00 (0.32 to 3.10) | 60 (1 RCT) | ⊕⊝⊝⊝ Very lowa,b | It is uncertain whether baclofen and naltrexone differ in dropouts at the end of treatment. |
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; RR: risk ratio | ||||||
| GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | ||||||
| See interactive version of this table: https://gdt.gradepro.org/presentations/#/isof/isof_question_revman_web_432318841614592889. | ||||||
a Downgraded one level: one study at high risk of performance, detection and attrition bias. b Downgraded two levels due to imprecision: fewer than 100 events.
Background
Description of the condition
Alcohol use disorder (AUD) is a severe mental disorder characterised by the inability to control alcohol consumption and frequent episodes of heavy drinking leading to detrimental consequences for both people with this disorder and others (Carvalho 2019; MacKillop 2022). AUD as a disease category has been introduced in the latest version of the Diagnostic and Statistical Manual of Mental Disorders (DSM), the Diagnostic and Statistical Manual of Mental Disorders, 5th edition (DSM‐5) (APA 2013). While the Diagnostic and Statistical Manual of Mental Disorders, 4th edition (DSM‐IV) (APA 2000) and International Classification of Diseases 11th edition (ICD)‐11 (WHO 2019) subdivided substance use disorders into dependence and a secondary category, called 'abuse' in DSM‐IV and 'harmful use' in ICD‐11 (MacKillop 2022), the DSM‐5 integrates both categories into a single substance use disorder concept that ranges along a continuum from mild to severe (APA 2013).
AUD and alcohol‐related impairments belong to the most widespread psychiatric disorders (Carvalho 2019; Grant 2015). Worldwide, according to the World Health Organization (WHO), one‐year AUD prevalence was estimated at approximately 5% of the general population in 2016, equivalent to 1 in 20 adults (WHO 2018). Statistics vary between regions, with a higher prevalence of AUD in high‐income countries compared to low‐ and middle‐income countries (Glantz 2020), with the highest one‐year prevalence of AUD in European countries and North America (WHO 2018). The latest WHO status report on alcohol and health selected the heavy episodic drinking (HED, defined as 60 g or more of pure alcohol on at least one single occasion at least once per month) as an indicator of the pattern of excessive alcohol consumption (WHO 2018). Worldwide, using survey data from 118 countries, the prevalence of HED was estimated at 20% of the adult general population (aged 15 years and older) (Manthey 2019), with the highest values in European countries (26% of the general population) and America (21% of the general population) (WHO 2018). There is evidence indicating an enormous increase in alcohol use, high‐risk drinking and DSM‐IV AUD in the US population between 2001/2002 and 2012/2013 (Grant 2017). Nevertheless, with the improved industrialisation and centralisation of alcohol production, alcohol consumption is increasingly becoming a problem in many low‐ and middle‐income countries (WHO 2018).
Irrespective of region, AUD is prevalent in more men than women (Glantz 2020), with lifetime prevalence estimated at 14.8% among men and 3.5% among women in the Europe and 11.5% among men and 5.1% among women in the Americas (WHO 2018). However, there is evidence on converging drinking patterns between genders (Slade 2016; White 2015).
The misuse of alcohol is one of the leading global health risk factors causing cancer, liver disease, heart disease, neurological disorders, fetal alcohol spectrum disorder, motor vehicle accidents, murder and other intentional injuries (GBD 2019 Cancer Risk Factors Collaborators 2022; GBD 2020 Alcohol Collaborators 2022). Worldwide, in 2016, alcohol use was the seventh leading risk factor for deaths accounting for 2.2% of female and 6.8% of male deaths (GBD 2018). It has been estimated that alcohol use will remain one of the leading risk factors for the burden of disease for the foreseeable future (Manthey 2019). The risk of all‐cause mortality is dose related and the level of consumption that minimises health loss is zero (GBD 2018). Men with AUD have a three‐fold higher mortality risk compared to men without AUD; in women, the risk is even higher (Roerecke 2013). In 2016, 5.3% of all global deaths and 5.0% of global disability‐adjusted life‐years were attributable to alcohol (Shield 2020). Studies also suggest that AUD increases the risk of COVID‐19 infections and mortality (Fond 2021; Spagnolo 2020). The costs attributable to alcohol consumption are estimated to be on average 2.6% of gross domestic product, the majority of costs due to loss in productivity (Manthey 2021). At the same time, alcohol consumption is one of the major, potentially avoidable risk factors, underscoring the need for effective strategies to reduce excessive drinking and maintain abstinence in people with AUD (Haber 2021).
The treatment of AUD was exclusively dominated by psychosocial strategies for many decades. Even though techniques from different theoretical and therapeutic backgrounds have been developed, treatment effects obtained by an exclusive application of psychosocial treatment are limited. A high proportion of patients do not respond to the interventions and, of those who respond, few succeed in maintaining abstinence in the long term (Moos 2006). With the investigation of the neurobiological mechanism of AUD, several pharmacological agents have been examined for their potential to support people with AUD in achieving abstinence or in cutting down their alcohol consumption (Witkiewitz 2019). Four agents are approved for the pharmacological treatment of AUD: the opioid antagonists naltrexone and nalmefene; the glutamate antagonist acamprosate; and the aversive agent disulfiram (Mutschler 2016; Reus 2018). While the primary mechanism of action of the two opioid antagonists is to block alcohol‐induced reward effects, acamprosate is assumed to work by restoring the balance between inhibitory and excitatory neurotransmitters (Rösner 2010a; Rösner 2010b). In contrast to naltrexone, nalmefene and acamprosate, which are assumed to modulate neurobiological mechanisms of addiction, disulfiram works by producing an aversive reaction when combined with alcohol (Mutschler 2016; Skinner 2014). Various Cochrane Reviews have investigated the effects of pharmacological interventions for the treatment of alcohol withdrawal (Amato 2010; Amato 2011; Gillman 2007; Leone 2010; Liu 2015; Minozzi 2010; Sarai 2013), and for relapse prevention in AUD (Pani 2014; Pedersen 2013; Rösner 2010a; Rösner 2010b; Vaz de Lima 2010). The best practice in the treatment of AUD should comprise both pharmacotherapy and psychotherapy (Haber 2021; Ray 2020). However, in the US, one recent national study that interviewed more than 42,000 adults estimated that only 1.6% of people with AUD received medications approved for this disorder (Han 2021). In Italy, among a sample of 345 people with severe AUD admitted to public services for outpatient treatment of this disorder, less than 30% received the combination of pharmacotherapy and psychotherapy even if they were still drinking during treatment (Agabio 2021a).
Description of the intervention
Baclofen, also known as beta‐4‐chlorophenyl‐gamma‐aminobutyric acid (Steardo 1984), is an agonist of the γ‐aminobutyric acid type B (GABAB) receptor (Kent 2020). The substance was originally approved for use in spasticity associated with neurological conditions and has recently emerged as a treatment of major interest for AUD (Agabio 2013; Agabio 2014; Agabio 2018a; Andrade 2020; Brennan 2013; Burnette 2022; de Beaurepaire 2019; Morley 2021). Baclofen is available in oral formulations and intrathecal solutions, the latter reserved for treatment of spasticity unresponsive to oral administration of baclofen. The recommended oral daily dose for baclofen ranges from 15 mg to 80 mg, starting from 15 mg a day and increasing by 5 mg every three days (Agabio 2018a; de Beaurepaire 2019). Despite high individual variation in pharmacokinetics, baclofen is rapidly absorbed from the gastrointestinal tract following oral administration. Up to 80% of an oral dose is excreted in the urine, with only a limited hepatic metabolism, making it a useful agent in people with impaired hepatic function, while renal function should be carefully assessed prior to baclofen administration (Agabio 2018a; Brennan 2013). In addition, baclofen has a short half‐life of three to four hours and is rapidly cleared from the blood. As a result, baclofen needs to be administered three or four times per day to maintain therapeutic effects (Schwarz Pharma 2003).
Preclinical studies have found that baclofen reduces the acquisition of alcohol‐drinking behaviour and its maintenance and reinstatement in alcohol‐experienced rats (Agabio 2014; Colombo 2018). Effects on reinstatement were assessed by the modification of the so‐called 'alcohol deprivation effect', a rodent model for studying relapse behaviour referring to the temporary increase in voluntary alcohol intake after a period of forced abstinence from alcohol in a free‐choice experiment (Martin‐Fardon 2013). Furthermore, baclofen has been shown to dose‐dependently reduce the number of lever‐responses for alcohol and the amount of self‐administered alcohol in rats under operant self‐administration conditions (e.g. Colombo 2018; Maccioni 2005; Maccioni 2012). In operant self‐administration experiments, a specific amount of 'work' is required to access alcohol, with predictive validity for human alcohol craving, and providing measures of alcohol consumption and the reinforcing properties of alcohol (Agabio 2014; Colombo 2018). Thereby baclofen was more potent and effective in those rats seeking and taking larger amounts of alcohol (Colombo 2018; Maccioni 2012; Walker 2007).
A series of case reports found that the administration of baclofen markedly reduced alcohol intake in people with AUD (Agabio 2007; Ameisen 2005; Bucknam 2006; Flannery 2004; Krupitsky 1993; Pastor 2012). Ameisen 2005 described the personal history of a French physician, who conducted an original dose‐finding curve with baclofen with the aim of treating his own AUD (Agabio 2014; de Beaurepaire 2019). He tested oral baclofen, starting with the 'conventional' dose of 30 mg a day and increased the daily dose up to 270 mg after five weeks, experiencing, "complete medication‐induced suppression of craving" for alcohol with alleviation of comorbid anxiety (Ameisen 2005).
While numerous case reports, case series and open‐label studies have been published indicating the effectiveness of baclofen in people with AUD, randomised controlled trials (RCTs) conducted to date have yielded conflicting results (for an overview see de Beaurepaire 2019). The diversity of findings has stimulated the discussion on potential moderators of effectiveness (Agabio 2018a; de Beaurepaire 2019; Leggio 2010; Morley 2021). Among others, severity of alcohol dependence (Agabio 2018a; de Beaurepaire 2019), baclofen dosing (Agabio 2018a; de Beaurepaire 2019; Thompson 2017), and comorbid anxiety (Agabio 2018b; Agabio 2021b; de Beaurepaire 2019) have been discussed to moderate the effects of baclofen on alcohol consumption.
Although data are mixed regarding baclofen's efficacy in AUD, they are consistent in terms of safety if used as recommended (Agabio 2018a; Brennan 2013). Extensive information on baclofen's safety is available from its widespread use for the treatment of spasticity for decades. Most adverse events are not severe, are dose‐related and are transient (Agabio 2018a; de Beaurepaire 2019). Sedation, somnolence, weakness, vertigo and psychological disturbances are the most common adverse events (Dario 2004). The administration of low doses of baclofen is considered safe in people affected by AUD, while an abrupt interruption and withdrawal from higher doses of baclofen may lead to serious adverse effects (Agabio 2018a; de Beaurepaire 2019). While such events are rare, the lack of noted adverse events may be a result of small sample sizes and low doses of baclofen utilised in available studies (Brennan 2013). In October 2018, the French Medicines Agency (Agence Nationale de Sécurité du Médicament et des Produits de Santé (ANSM)) approved baclofen for the treatment of AUD at the maximum dose of 80 mg a day (Rolland 2020). In France, where this medication was frequently prescribed for the treatment of AUD at high doses, an alarming increase of self‐poisonings with baclofen, including suicide attempts and unintentional overdose, has been reported (e.g. Holla 2015; Logge 2022; Pelissier 2017; Reynoard 2020) leading to the authorisation of a maximum daily dose of baclofen 80 mg for the treatment of AUD (Rolland 2020).
How the intervention might work
The mesolimbic dopamine pathway, including dopamine cells in the ventral tegmental area projecting into the nucleus accumbens, is crucial for drug reward and addiction (Volkow 2011; Wise 2009). Baclofen's primary mechanism of action for alcohol dependence is presumed to be the reduction of the reinforcing properties of alcohol by suppressing alcohol‐stimulated dopamine release in the mesolimbic dopamine system (de Beaurepaire 2018). Agonistic effects of baclofen at GABAB receptors located in several brain areas including the mesolimbic circuit (Pitman 2014), and the ventral tegmental area, inhibit alcohol‐induced firing of dopaminergic neurons and of alcohol‐stimulated dopamine release in the nucleus accumbens, resulting in the reduction of reinforcing properties of alcohol and drugs (Agabio 2014; Maccioni 2009; Young 2014). As baclofen did not affect spontaneous motor activity in rats, baclofen's effects are not due to muscle‐relaxant or sedative properties of the drug, but can rather be considered as a result of its ability to reduce the appetitive strength of alcohol (Colombo 2003; Colombo 2018). In addition, substitutional effects of baclofen to replace alcohol have been discussed (Chick 2012; Rolland 2013).
In addition, GABAB receptors are highly expressed in limbic structures involved in the mediation of anxiety (Cryan 2005), while activation of GABAB receptors in these structures might reduce anxiety (Morley 2014‐LD; Morley 2014‐MD). Evidence from various clinical studies shows that baclofen may reduce anxiety in people with AUD (Addolorato 2002; Flannery 2004; Krupitsky 1993; Morley 2014‐LD; Morley 2014‐MD). Post‐hoc analyses in one of the studies found that treatment with baclofen was most effective in people with comorbid anxiety (Morley 2014‐LD; Morley 2018‐MD). This result has been confirmed by one recent meta‐analysis (Agabio 2021b). Thus, besides dampening the reinforcing properties of alcohol through the suppression of alcohol‐stimulated dopamine release in the mesolimbic dopamine system, baclofen's effects on drinking might also be related to the presence of anxiety symptoms (Agabio 2014; Agabio 2021b).
Why it is important to do this review
While several pharmacological treatments have been approved for the treatment of AUD (see Background), their clinical use is limited by a number of factors (Sinclair 2016). The aversive agent disulfiram supports abstinence (Jorgensen 2011), but due to its safety profile it is a therapeutic option for only selected patient groups with a high self‐efficacy expectation to abstain from drinking after taking disulfiram (Mutschler 2016). Further substances such as the opioid‐antagonists, naltrexone and nalmefene, and the glutamate antagonist, acamprosate, appear to be safe and effective in people with AUD, but the strong variation in treatment response (Rösner 2010a; Rösner 2010b), indicates that patients might benefit from an extended range of treatment options that allow a further individualisation of pharmacological approaches (Witkiewitz 2019). Baclofen as a treatment option for AUD has received much attention in recent years, initiated by pharmacological self‐experimentation of Olivier Ameisen (Ameisen 2005), and suggested for people with AUD who do not respond to other medications for AUD (i.e. acamprosate, naltrexone and disulfiram) or have contraindications for the use of other medications (i.e. sever liver disease) (de Beaurepaire 2019). Even though numerous case reports and case series confirmed the effectiveness of baclofen in people with AUD, data from RCTs yielded conflicting results (de Beaurepaire 2019).
The planned review of baclofen to achieve abstinence or to reduce alcohol consumption in people with AUD will provide a systematic integration of the available evidence for health decision makers, therapists and patients, and aims to offer illustrative measures for estimating the therapeutic benefits and risks of baclofen, while indicating gaps in knowledge and methodological demands for future clinical research. In addition, the planned review will evaluate the effects due to the different doses of baclofen considering that, in clinical practice, the daily dose used to help people with AUD to achieve abstinence or reduce alcohol consumption varies between patients over a 10‐fold range (Agabio 2018a). In addition, the planned review will also evaluate two other factors that may influence the effects of baclofen in the treatment of AUD such as the duration of the treatment and the consumption of alcohol at the beginning of treatment (Agabio 2018a).
Objectives
To evaluate the benefits and harms of baclofen on achieving and maintaining abstinence or reducing alcohol consumption in people with AUD compared to placebo, no treatment or any other pharmacological relapse prevention treatment.
Methods
Criteria for considering studies for this review
Types of studies
RCTs of at least four weeks' treatment duration and 12 weeks' overall study duration, comparing baclofen with placebo, no treatment or other pharmacological treatments for achieving alcohol abstinence or reducing consumption in people with AUD. We planned to include studies employing a cross‐over design, using data from the first active treatment stage only.
Types of participants
Adults (aged 18 years and older), currently with AUD according to Diagnostic and Statistical Manual of Mental Disorders, 3rd edition (DSM‐III; APA 1980), Diagnostic and Statistical Manual of Mental Disorders, 3rd edition, revised (DSM‐ III‐R; APA 1987), Diagnostic and Statistical Manual of Mental Disorders, 4th edition, text revision (DSM‐IV‐TR; APA 2000), DSM‐5 (APA 2013), and ICD‐10 (WHO 1992; WHO 2010). There were no limitations on other participant characteristics such as concomitant substance use disorders or other comorbid psychiatric conditions. We included participants who were still drinking and those in the postdetoxification phase, if the detoxification was completed 28 days or less before starting treatment. We also included studies with participants in methadone maintenance schemes. We excluded studies where baclofen was used to treat alcohol withdrawal.
We excluded people younger than 18 years of age and pregnant women because of the substantially different approach required for clinical management of these individuals.
Types of interventions
Experimental intervention
We included baclofen at any dose and route of administration. Studies could consider baclofen as monotherapy or in combination with further treatments provided that concomitant treatments were provided equally in both the experimental and control groups.
Control intervention
Placebo, no treatment or any other pharmacological relapse prevention treatment, including acamprosate, naltrexone or nalmefene. We did not consider disulfiram as a control intervention due to the psychological mediation of its effects.
Types of outcome measures
Primary outcomes
Relapse: return to any drinking, measured by the number of people who had returned to any drinking at the end of the study.
Frequency of use: measured as mean number or percentage of abstinent days (ratio of the total sum of days with abstinence, related to the entire duration of the study, multiplied by 100; and mean number or percentage of heavy drinking days (HDD)).
Amount of use: number of drinks per drinking day or drinking occasion.
Adverse events: measured by number of people with at least one adverse event, both subjectively and objectively assessed.
Dropouts from treatment: number of participants who did not complete the study protocol.
Dropouts from treatment due to adverse events.
We included drinking outcomes irrespective of the source of information, considering, for example, participant self‐reports, breathalyser tests, laboratory tests and collateral reports of others.
Secondary outcomes
Craving, as measured by validated scales.
Anxiety, as measured by validated scales.
Depression, as measured by validated scales.
Frequency of most relevant adverse events.
Search methods for identification of studies
The Cochrane Drugs and Alcohol Information Specialist conducted systematic searches for RCTs and controlled clinical studies. There were no language, publication year or publication status restrictions. The date of the search was 22 November 2021.
Electronic searches
We searched the following databases:
Cochrane Drugs and Alcohol Group (CDAG) Specialised Register (22 November 2021; Appendix 1);
Cochrane Central Register of Controlled Trials (CENTRAL; 2021, issue 11) via onlinelibrary.wiley.com (Appendix 2);
MEDLINE via Ovid (from January 2018 to 22 November 2021; Appendix 3);
Embase via Ovid (from January 2018 to 30 January 2018; Appendix 4);
PsycINFO (Ovid; from January 2018 to 22 November 2021; Appendix 5);
Web of Science (Thomson Reuters; from January 2018 to 22 November 2021; Appendix 6);
CINAHL (EBSCOhost; from January 2018 to 22 November 2021; Appendix 7).
We searched the databases using MeSH and free‐text terms relating to baclofen and alcohol dependence. We combined the PubMed search with the Cochrane Highly Sensitive Search Strategy for identifying RCTs in MEDLINE: sensitivity‐maximising version (Lefebvre 2011). We revised this strategy appropriately for each database to take account of differences in controlled vocabulary and syntax rules.
We searched the following trials registries on 22 November 2021:
ClinicalTrials.gov (www.clinicaltrials.gov);
WHO International Clinical Trials Registry Platform (ICTRP) (apps.who.int/trialsearch/).
Searching other resources
We contacted key informants and experts to request any further, potentially relevant studies and seek information about unpublished or incomplete studies. We handsearched the reference lists of included studies and current reviews. We included all eligible studies identified by the search, irrespective of language, publication type or status.
Data collection and analysis
Selection of studies
After removing duplicates, two review authors (RA, SM) independently screened the abstracts of all publications obtained by the search strategy. Two review authors (RA, SM) independently assessed the full text of potentially relevant studies for inclusion. We excluded studies that did not meet eligibility criteria and recorded the reasons in the Characteristics of excluded studies table. We used Covidence software for study selection (Covidence). We resolved any disagreement by discussion, involving a third review author (RS) in case of persisting disagreements. We documented the search process in sufficient details to complete a PRISMA study flow diagram.
Data extraction and management
Two review authors (RA, SM) independently extracted data onto a data extraction form. We resolved any doubts by discussion. We extracted the following information: number and characteristics of participants, setting, type of experimental and control intervention, length of follow‐up, types of outcomes, country of origin, funding and conflict of interest. We used Covidence software for data extraction (Covidence).
Assessment of risk of bias in included studies
Two review authors (RA, SM) independently assessed the risk of bias of the included studies using the criteria recommended in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2017). The recommended approach for assessing risk of bias in studies included in Cochrane Reviews is a two‐part tool, addressing the following specific domains: sequence generation and allocation concealment (selection bias), blinding of participants and providers (performance bias), blinding of outcome assessors (detection bias), incomplete outcome data (attrition bias) and selective outcome reporting (reporting bias). The first part of the tool involves describing what was reported to have happened in the study. The second part of the tool involves assigning a judgement relating to the risk of bias for that entry, in terms of low, high or unclear risk. To make these judgements, we used the criteria indicated by the Cochrane Handbook for Systematic Reviews of Interventions, adapted to the addiction field. See Appendix 8 for details.
We addressed the domains of sequence generation and allocation concealment (avoidance of selection bias) by a single entry for each study.
We considered blinding of participants, personnel and outcome assessors (avoidance of performance bias and detection bias), separately for objective outcomes (e.g. relapse measured by urine or breath analysis, duration of abstinence and dropout), and subjective outcomes (e.g. participant self‐reported use of substance, adverse events, craving, psychiatric symptoms).
We considered incomplete outcome data (avoidance of attrition bias), for all outcomes except dropout from the treatment, which is very often the primary outcome measure in studies on addiction.
Measures of treatment effect
We analysed dichotomous outcomes by calculating the risk ratio (RR) for each trial with the uncertainty in each result being expressed with 95% confidence intervals (CI). We analysed continuous outcomes by calculating the mean difference (MD) with 95% CIs when the studies used the same instrument for assessing the outcome. We used the standardised mean difference (SMD) when the studies used different instruments.
Unit of analysis issues
If we included multi‐armed studies in the meta‐analyses and one arm was considered more than once in the same comparisons (e.g. two different doses of baclofen compared to the same control group), we divided the control group into two different groups, each group comprising half the participants of the original group to avoid double counting of participants in the control groups. For cross‐over studies in meta‐analyses, we planned to use data from the first period only (i.e. before cross‐over), to address the risk of carry‐over effects. However, we did not include any cross‐over studies.
Dealing with missing data
We contacted the original investigators to request information on incomplete or missing data from the studies. We included all randomised participants in the statistical analysis, without any imputation of missing data. If standard deviations (SD) were missing, we used the mean of the available SDs of the other included studies (Furukawa 2006).
Assessment of heterogeneity
We analysed heterogeneity using the I² statistic and the Chi² test (Higgins 2003). We regarded heterogeneity as substantial if the I² statistic was greater than 50% or P less than 0.10 for the Chi² test for heterogeneity (Deeks 2017). Following the guidance in the Cochrane Handbook for Systematic Reviews of Interventions (Deeks 2017), we distinguished the following values: 0% to 40% denoted no important, 30% to 60% moderate, 50% to 90% substantial and 75% to 100% considerable heterogeneity. If we found considerable heterogeneity (i.e. 75% or above), we explored possible reasons by visually inspecting the forest plot to identify studies that might be contributing to heterogeneity.
Assessment of reporting biases
We used visual inspection of funnel plots (plots of the effect estimate from each study against the sample size or effect standard error) to indicate possible publication bias if there were at least 10 studies included in the meta‐analysis (Sterne 2017).
Data synthesis
We combined the outcomes from the individual studies through meta‐analysis where possible (comparability of intervention and outcomes between studies), using a random‐effects model, because we expected a certain degree of heterogeneity between studies. If the clinical or statistical heterogeneity between studies was too high (i.e. 75% to 100%), we considered not pooling the data.
Subgroup analysis and investigation of heterogeneity
We performed subgroup analysis to investigate the sources of heterogeneity and assess any difference in treatment efficacy and safety in different subgroups. In detail, we planned to perform subgroup analyses for the following variables:
daily doses of baclofen;
participants with concomitant substance use disorders;
comorbid psychiatric conditions;
consumption of alcohol at the beginning of treatment;
duration of treatment.
According to the daily doses of baclofen, we divided the studies into three subgroups: low doses (up to 30 mg), medium doses (above 30 and 100 mg or less) and high doses (above 100 mg).
According to the consumption of alcohol at the beginning of the treatment, we divided the studies into two subgroups: detoxified participants (when participants were abstinent for at least three days before treatment) and non‐detoxified participants (when participants were still drinking at the beginning of the treatment).
According to the duration of treatment, we divided studies into two subgroups: 12‐week studies and longer than 12‐week studies.
We were able to perform subgroup analyses for different daily doses of baclofen, detoxified versus non‐detoxified participants and duration of treatment.
Subgroup analyses did not explain heterogeneity, which remained very high in the subgroups were there was one outlying study with results that conflicted with the rest of the studies (Leggio 2015). Participants included in this study were both nicotine and alcohol dependent, the study had the aimed to obtain both smoking and drinking abstinence and the study was conducted in a laboratory setting. As these characteristics could explain the heterogeneity due to the different population of participants compared to those recruited by the other studies, we excluded the study from the meta‐analyses.
Sensitivity analysis
To incorporate our assessment of risk of bias in the review process we first plotted the intervention effect estimates stratified by risk of bias for allocation concealment (selection bias). If differences in the results were present among studies at different risks of selection bias, we planned to perform sensitivity analysis by excluding studies at high risk of bias from the analysis. We also planned to perform sensitivity analysis to assess how sensitive the results were to changes in the assumptions about missing data for relapse, assuming dropout or loss to follow‐up as not relapsed. We did not perform any sensitivity analyses because there were no such cases.
Summary of findings and assessment of the certainty of the evidence
We assessed the overall certainty of the evidence for the following primary outcomes using GRADE: relapse, frequency of use, amount of use, adverse events and dropout from treatment. The GRADE Working Group developed a system for grading the certainty of evidence that considers issues related to internal validity and external validity, such as directness of results. (GRADE 2004; Guyatt 2008; Guyatt 2011; Schünemann 2006).
We presented the main findings of the review in three summary of findings tables: baclofen versus placebo, baclofen versus acamprosate and baclofen versus naltrexone. This is a transparent and simple tabular format that provides key information concerning the certainty of evidence, the magnitude of effect of the interventions examined and the sum of available data on the main outcomes.
The GRADE system uses the following criteria for assigning grades of evidence.
High: we are very confident that the true effect lies close to that of the estimate of the effect.
Moderate: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different.
Low: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect.
Very low: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect.
Grading is decreased for the following reasons:
serious (−1) or very serious (−2) study limitations for risk of bias;
serious (−1) or very serious (−2) inconsistency between study results;
some (−1) or major (−2) uncertainty about directness (the correspondence between the population, the intervention, or the outcomes measured in the studies actually found and those under consideration in our systematic review);
serious (−1) or very serious (−2) imprecision of the pooled estimate;
publication bias strongly suspected (−1).
We used GRADEpro GDT software to prepare the summary of findings tables (GRADEpro GDT).
Results
Description of studies
Results of the search
Figure 1 shows the PRISMA flow diagram for screening, selection and assessment of studies.
1.
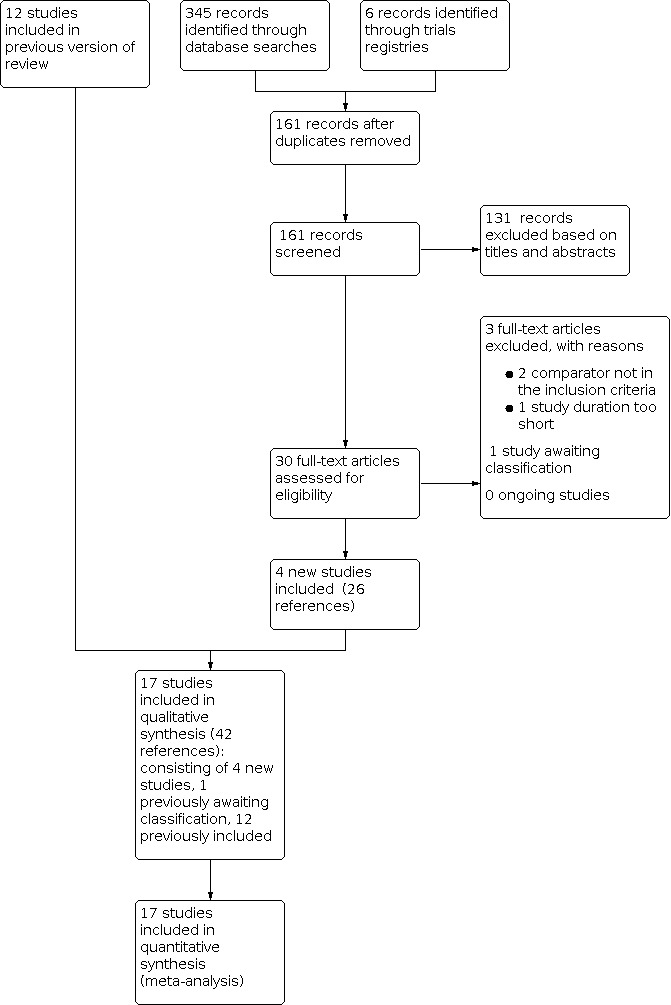
PRISMA study flow diagram.
For this update, we identified 161 studies from all possible sources following the removal of duplicates. We excluded 131 studies based on title and abstract and assessed 30 full‐text articles. We identified four studies meeting our inclusion criteria reported in 22 references. One study compared a single dose of baclofen to placebo (Rigal 2020), two studies compared two doses of baclofen to placebo (each included as two discrete data sets; Garbutt 2021‐LD; Garbutt 2021‐MD; Morley 2018‐LD; Morley 2018‐MD), and one study compared a single dose of baclofen to acamprosate and naltrexone (Kumar 2020a). In addition, the authors of a study previously awaiting classification that compared two doses of baclofen to placebo provided data allowing us to include the study (included as two discrete data sets; Addolorato 2011‐LD; Addolorato 2011‐MD). We excluded three studies (see Characteristics of excluded studies table) and one is awaiting classification (see Characteristics of studies awaiting classification table). Overall, we included five new RCTs.
We previously identified 12 studies, eight that compared a single dose of baclofen to placebo (Addolorato 2007; Garbutt 2010a; Hauser 2017; Krupitskii 2017; Leggio 2015; Muller 2015; Ponizovsky 2015; Reynaud 2017), two that compared two doses of baclofen to placebo (each included as two discrete data sets; Beraha 2016‐HD; Beraha 2016‐LD; Morley 2014‐LD; Morley 2014‐MD), one study that compared a single dose of baclofen to acamprosate (Mishra 2010), and one study that compared baclofen to placebo and naltrexone (included as two discrete data sets; Garbutt 2010b1; Garbutt 2010b2) in our original review (see Characteristics of included studies table).
Included studies
We included 17 RCTs involving 1818 participants (see Characteristics of included studies table). The mean study size was 107 participants, ranging from 30 in Leggio 2015 to 320 in Reynaud 2017. Six studies recruited more than 100 participants (Beraha 2016‐LD; Beraha 2016‐LD; Garbutt 2021‐LD; Garbutt 2021‐MD; Hauser 2017; Morley 2018‐LD; Morley 2018‐MD; Reynaud 2017; Rigal 2020). The mean age of participants was 46.5 years, and 69.6% were men. All studies except one recruited participants with a diagnosis of alcohol dependence according to the DSM‐IV or ICD‐10 criteria. Rigal 2020 did not require the diagnosis of alcohol dependence and recruited high‐risk drinkers according to the WHO definition of at‐risk drinking (alcohol consumption greater than 40 g/day or single occasion, greater than 280 g/week, or both for women; greater than 60 g/day or single occasion, greater than 420 g/week, or both for men; WHO 2000). To be included, these participants also had to voluntarily consult a physician for their alcohol problem and express the desire to achieve abstinence or reduce alcohol consumption. Accordingly, we assumed that these participants met at least three criteria for AUD (alcohol consumption in higher amounts than intended; desire to cut down or control alcohol use; craving; APA 2013), and we included the study.
Five studies took place in the USA (Garbutt 2010a; Garbutt 2010b1; Garbutt 2010b2; Garbutt 2021‐LD; Garbutt 2021‐MD; Hauser 2017; Leggio 2015); two studies in Australia (Morley 2014‐LD; Morley 2014‐MD; Morley 2018‐LD; Morley 2018‐MD), France (Reynaud 2017; Rigal 2020), Italy (Addolorato 2007; Addolorato 2011‐LD; Addolorato 2011‐MD), and India (Kumar 2020a; Mishra 2010); and one in Germany (Muller 2015), Israel (Ponizovsky 2015), Russia (Krupitskii 2017), and the Netherlands (Beraha 2016‐HD; Beraha 2016‐LD).
All studies excluded people with substance use disorders by substances other than alcohol or nicotine. One trial recruited people dependent by both alcohol and nicotine (Leggio 2015). All studies excluded people with comorbid severe mental disorders but five studies recruited people receiving stable doses of antidepressants (Beraha 2016‐HD; Beraha 2016‐LD; Garbutt 2010a; Garbutt 2021‐LD; Garbutt 2021‐MD; Morley 2018‐LD; Morley 2018‐MD; Reynaud 2017). Three studies recruited people with severe liver disease (i.e. cirrhosis including Child‐Pugh, hepatitis B virus‐positive, hepatitis C virus‐positive: Addolorato 2007; chronic HCV: Hauser 2017; alcoholic liver disease: Morley 2018‐LD; Morley 2018‐MD).
Most studies required participants to abstain from alcohol for at least three days before beginning the pharmacological treatment (Addolorato 2007; Addolorato 2011‐LD; Addolorato 2011‐MD; Beraha 2016‐HD; Beraha 2016‐LD; Garbutt 2010a; Krupitskii 2017; Kumar 2020a; Morley 2014‐LD; Morley 2014‐MD; Morley 2018‐LD; Morley 2018‐MD; Muller 2015; Reynaud 2017). In these studies, abstinence ranged from three to 28 days (Beraha 2016‐HD; Beraha 2016‐LD). Seven studies recruited people who were still drinking at the beginning of the pharmacological treatment (Garbutt 2010b1; Garbutt 2010b2; Garbutt 2021‐LD; Garbutt 2021‐MD; Hauser 2017; Leggio 2015; Mishra 2010; Ponizovsky 2015; Rigal 2020).
Most studies were had a duration of 12 weeks (Addolorato 2007; Addolorato 2011‐LD; Addolorato 2011‐MD; Garbutt 2010a; Garbutt 2010b1; Garbutt 2010b2; Hauser 2017; Krupitskii 2017; Leggio 2015; Mishra 2010; Morley 2014‐LD; Morley 2014‐MD; Morley 2018‐LD; Morley 2018‐MD; Muller 2015; Ponizovsky 2015), while five studies were longer than 12 weeks (16 weeks: Beraha 2016‐HD; Beraha 2016‐LD; Garbutt 2021‐LD; Garbutt 2021‐MD; 24 weeks: Kumar 2020a; 26 weeks: Reynaud 2017; 48 weeks: Rigal 2020). The mean duration of the interventions was 16.1 weeks (range 12 to 48 weeks).
Interventions
The studies administered baclofen in daily doses ranging from 30 mg to 300 mg. Ten studies administered low daily doses (Addolorato 2007; Addolorato 2011‐LD; Beraha 2016‐LD; Garbutt 2010a; Garbutt 2010b1; Garbutt 2010b2; Garbutt 2021‐LD; Hauser 2017; Mishra 2010; Morley 2014‐LD; Morley 2018‐LD), eight studies medium daily doses (Addolorato 2011‐MD; Garbutt 2021‐MD; Krupitskii 2017; Kumar 2020a; Leggio 2015; Morley 2014‐MD; Morley 2018‐MD; Ponizovsky 2015), and four studies high daily doses of baclofen (Beraha 2016‐HD; Muller 2015; Reynaud 2017; Rigal 2020).
Most studies administered fixed doses of baclofen, starting with a daily dose of 5 mg, three times a day, and gradually increasing up to 30 mg/day to 80 mg/day. Four studies administered flexible doses of baclofen starting from low daily doses and progressively increasing up to 300 mg, according to the beneficial and unwanted (or both) effects (Beraha 2016‐HD; Muller 2015; Reynaud 2017; Rigal 2020).
In all but one study (Mishra 2010), participants in both the baclofen and placebo groups received psychosocial treatment or counselling of various intensity.
Comparisons
Fifteen RCTs compared baclofen with placebo (Addolorato 2007; Addolorato 2011‐LD; Addolorato 2011‐MD; Beraha 2016‐HD; Beraha 2016‐LD; Garbutt 2010a; Garbutt 2010b1; Garbutt 2021‐LD; Garbutt 2021‐MD; Hauser 2017; Krupitskii 2017; Leggio 2015; Morley 2014‐LD; Morley 2014‐MD; Morley 2018‐LD; Morley 2018‐MD; Muller 2015; Ponizovsky 2015; Reynaud 2017; Rigal 2020); two RCTs compared baclofen to naltrexone (Garbutt 2010b2; Kumar 2020a); and two RCTs compared baclofen to acamprosate (Kumar 2020a; Mishra 2010).
Ten RCTs compared one dose of baclofen to placebo (Addolorato 2007; Garbutt 2010a; Hauser 2017; Krupitskii 2017; Leggio 2015; Mishra 2010; Muller 2015; Ponizovsky 2015; Reynaud 2017; Rigal 2020); five RCTs compared two doses of baclofen to placebo (Addolorato 2011‐LD; Addolorato 2011‐MD; Beraha 2016‐HD; Beraha 2016‐LD; Garbutt 2021‐LD; Garbutt 2021‐MD; Morley 2014‐LD; Morley 2014‐MD; Morley 2018‐LD; Morley 2018‐MD); one RCT compared baclofen to placebo and another medication (Garbutt 2010b1; Garbutt 2010b2); and one RCT compared baclofen to two other medications (Kumar 2020a). We found no studies comparing baclofen to no treatment.
Excluded studies
We excluded three studies: Gupta 2017 because the inactive medication used in the control group (benfothiamine) has an effect on both alcohol consumption and anxiety (Manzardo 2013; Manzardo 2015); Jose 2019 because its duration was less than 12 weeks and Kumar 2020b because both groups received baclofen. The previous version of this review excluded four studies (Addolorato 2002; Flannery 2004; Leggio 2011; Leggio 2013). In total, we excluded seven studies. For more details, see Characteristics of excluded studies table.
Studies awaiting classification
One new study is awaiting classification as it was a conference abstract providing insufficient information regarding the design and length of the study (Karthik 2018). We moved some references awaiting classification in the previous version of this review to the included studies as primary or secondary references (Addolorato 2011‐LD; Addolorato 2011‐MD; Farokhnia 2014; Jaury 2014; Morley 2013). In this review update, two studies are awaiting classification (Karthik 2018; Sharma 2012; see Characteristics of studies awaiting classification table).
Ongoing studies
We identified no new ongoing studies. Among the two studies considered ongoing in the previous version of this review, one (NCT01980706) was moved to the included studies section (Garbutt 2021‐LD; Garbutt 2021‐MD), and one remains in the ongoing studies section (CTRI/2011/11/002154). See Characteristics of ongoing studies table.
Risk of bias in included studies
See Figure 2 and Figure 3. For a detailed description of the reasons supporting our judgements, see the risk of bias table in the Characteristics of included studies table.
2.

Risk of bias graph: review authors' judgements about each risk of bias item presented as percentages across all included studies
3.

Risk of bias summary: review authors' judgements about each risk of bias item for each included study
Allocation
Random sequence generation
We judged 14 studies at low risk of bias. The study authors did not report any information about methods of sequence random generation for the other three studies, so we judged them at unclear risk of bias (Addolorato 2007; Garbutt 2010b1; Garbutt 2010b2).
Allocation concealment
We judged 12 studies at low risk of bias. The other five studies did not report methods of allocation concealment, so we judged them at unclear risk of bias (Garbutt 2010a; Garbutt 2010b1; Garbutt 2010b2; Kumar 2020a; Leggio 2015; Mishra 2010).
Blinding
Performance bias
We judged 13 studies at low risk of bias for objective outcomes and two at high risk (Kumar 2020a; Mishra 2010). Two studies did not provide information about blinding, so we judged them at unclear risk of bias (Garbutt 2010a; Garbutt 2010b1; Garbutt 2010b2).
We judged 13 studies at low risk of bias for subjective outcomes and two at high risk (Kumar 2020a; Mishra 2010). Two studies did not report methods for blinding, so we judged them at unclear risk of bias (Garbutt 2010a; Garbutt 2010b1; Garbutt 2010b2).
Detection bias
For objective outcomes, we considered 16 studies at low risk of bias and one study at unclear risk (Garbutt 2010b1; Garbutt 2010b2).
For subjective outcomes, we considered 12 studies at low risk of bias and three studies at high risk of bias (Garbutt 2010a; Kumar 2020a; Mishra 2010). Two studies did not report sufficient information, so we judged them at unclear risk of bias (Garbutt 2010b1; Garbutt 2010b2; Hauser 2017).
Incomplete outcome data
We judged 11 studies at low risk of bias and two studies at high risk of bias (Addolorato 2011‐LD; Addolorato 2011‐MD; Kumar 2020a). Four studies did not report numbers and reasons of dropouts or missing data for each group, so we judged them at unclear risk of bias (Krupitskii 2017; Leggio 2015; Mishra 2010; Morley 2014‐LD; Morley 2014‐MD).
Selective reporting
We judged four studies at low risk of bias and two studies at high risk of bias (Addolorato 2011‐LD; Addolorato 2011‐MD; Mishra 2010). Eleven studies did not provide enough information, so we judged them at unclear risk of bias (Garbutt 2010a; Garbutt 2010b1; Garbutt 2010b2; Hauser 2017; Krupitskii 2017; Kumar 2020a; Morley 2014‐LD; Morley 2014‐MD; Morley 2018‐LD; Morley 2018‐MD; Muller 2015; Ponizovsky 2015; Reynaud 2017; Rigal 2020).
Other potential sources of bias
We identified no other sources of bias.
Effects of interventions
See: Table 1; Table 2; Table 3
For the comparison baclofen versus placebo, we first conducted the analyses including from all studies and found considerable heterogeneity in the rates of abstinent days (17 studies, 1303 participants; I² = 94%) and HDDs at the end of treatment (14 studies, 870 participants; I² = 98%). Conducting the subgroup analyses, we still found considerable heterogeneity in all subgroups where one outlying study was included (Leggio 2015). Accordingly, we excluded this study from all analyses and, for each primary outcome, provided its results separately.
We found sufficient data for conducting subgroup analyses according to the daily doses of baclofen, consumption of alcohol at the beginning of treatment and duration of treatment. We conducted subgroup analyses for the primary outcomes (relapse, frequency of use (rate of abstinent days), amount of use (rate of HDDs, drink per drinking days), adverse events (number of participants with at least one adverse event), dropouts from treatment, and dropouts from treatment due to adverse events).
We did not conduct subgroup analyses according to the presence or not of comorbid substance use disorders or other mental disorders (or both) because all studies excluded people with substance use disorders of substances other than alcohol or nicotine and people with other comorbid severe mental disorders.
Comparison 1: baclofen versus placebo
1.1 Relapse: return to any drinking at end of treatment
Twelve studies involving 1057 participants reported relapse. Baclofen reduced the risk of relapse compared to placebo (RR 0.87, 95% CI 0.77 to 0.99; moderate‐certainty evidence; Analysis 1.1; Figure 4; Table 1). There was substantial heterogeneity (Chi² = 40.60, degrees of freedom (df) = 11 (P < 0.001); I² = 73%). The study excluded from this meta‐analysis found no difference in relapse between baclofen and placebo (RR −0.53, 95% CI −3.11 to 2.05; 30 participants; Leggio 2015).
1.1. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 1: Relapse: return to any drinking at end of treatment
4.

1.1.1 Subgroup analyses: daily doses of baclofen
We found no differences between baclofen and placebo among studies using low, medium or high doses (low: RR 0.82, 95% CI 0.64 to 1.04; considerable heterogeneity (Chi² = 32.98, df = 5 (P < 0.001); I² = 85%); 6 studies, 463 participants; medium: RR 0.73, 95% CI 0.37 to 1.45; considerable heterogeneity (Chi² = 11.20, df = 2 (P = 0.004); I² = 82%); 3 studies, 129 participants; high: RR 0.90, 95% CI 0.71 to 1.15; moderate heterogeneity (Chi² = 4.78, df = 2 (P = 0.09); I² = 58%); 3 studies, 465 participants; Analysis 2.1).
2.1. Analysis.

Comparison 2: Baclofen versus placebo (divided into low, medium and high doses of baclofen), Outcome 1: Relapse: return to any drinking at end of treatment
1.1.2 Subgroup analyses: duration of treatment
We found no differences between baclofen and placebo among 12‐week studies and longer than 12‐week studies (12 weeks: RR 0.63, 95% CI 0.40 to 1.00; considerable heterogeneity (Chi² = 54.47, df = 6 (P < 0.001); I² = 89%); 7 studies, 466 participants; longer than 12 weeks: RR 0.98, 95% CI 0.93 to 1.03; I² = 0%; 5 studies, 591 participants; Analysis 3.1).
3.1. Analysis.

Comparison 3: Baclofen versus placebo (divided into 12‐week and longer than 12‐week studies), Outcome 1: Relapse: return to any drinking at end of treatment
1.1.3 Subgroup analyses: consumption of alcohol at beginning of treatment
Baclofen reduced the risk of relapse compared to placebo among studies with detoxified participants (RR 0.73, 95% CI 0.55 to 0.95; considerable heterogeneity (Chi² = 33.42, df = 8 (P < 0.001); I² = 76%); 9 studies, 757 participants; Analysis 4.1). There were no differences between baclofen and placebo among studies with non‐detoxified participants (RR 1.00, 95% CI 0.94 to 1.06; I² = 0%; 3 studies, 300 participants; Analysis 4.1).
4.1. Analysis.

Comparison 4: Baclofen versus placebo (divided into detoxified and non‐detoxified participants), Outcome 1: Relapse: return to any drinking at end of treatment
1.2 Frequency of use: percentage of days abstinent at end of treatment
We identified 16 studies involving 1273 participants reporting percentage of days abstinent at the end of treatment. Baclofen increased the rate of abstinent days compared to placebo (MD 9.07%, 95% CI 3.30 to 14.85; high‐certainty evidence; Analysis 1.2; Figure 5; Table 1). There was substantial heterogeneity (Chi² = 43.69, df = 15 (P = 0.001); I² = 66%). The study excluded from this meta‐analysis found that placebo increased the rate of abstinent days compared to baclofen (MD −19.00 days, 95% CI −21.18 to −16.82; 30 participants; Leggio 2015).
1.2. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 2: Frequency of use: % days abstinence at end of treatment
5.

1.2.1 Subgroup analyses: daily doses of baclofen
Baclofen increased the rate of abstinent days compared to placebo among studies using low and high doses (low: MD 10.59%, 95% CI 0.77 to 20.41; substantial heterogeneity (Chi² = 19.68, df = 7 (P = 0.006); I² = 64%); 8 studies, 583 participants; high: MD 11.09%, 95% CI 4.39 to 17.80; I² = 6%; 3 studies, 465 participants; Analysis 2.2). We found no differences between baclofen and placebo among studies using medium doses (MD 7.14%, 95% CI −3.10 to 17.38; moderate heterogeneity (Chi² = 8.39, df = 4 (P = 0.08); I² = 52%); 5 studies, 225 participants; Analysis 2.2).
2.2. Analysis.

Comparison 2: Baclofen versus placebo (divided into low, medium and high doses of baclofen), Outcome 2: Frequency of use: % days abstinence at end of treatment
1.2.2 Subgroup analyses: duration of treatment
Baclofen increased the rate of abstinent days compared to placebo among 12‐week studies and longer than 12‐week studies (12 weeks: MD 10.90, 95% CI 3.17 to 18.62; substantial heterogeneity (Chi² = 33.61, df = 10 (P = 0.001); I² = 70%); 11 studies, 682 participants; longer than 12 weeks: MD 8.05, 95% CI 1.09 to 15.01; I² = 18%; 5 studies, 591 participants; Analysis 3.2).
3.2. Analysis.

Comparison 3: Baclofen versus placebo (divided into 12‐week and longer than 12‐week studies), Outcome 2: Frequency of use: % days abstinence at end of treatment
1.2.3 Subgroup analyses: consumption of alcohol at beginning of treatment
Baclofen increased the rate of abstinent days compared to placebo among studies with detoxified participants (MD 11.76, 95% CI 3.22 to 20.29; moderate heterogeneity (Chi² = 19.42, df = 9 (P = 0.02); I² = 54%); 10 studies, 549 participants; Analysis 4.2). There was no difference between baclofen and placebo among studies with non‐detoxified participants (MD 6.03, 95% CI −1.59 to 13.64; substantial heterogeneity (Chi² = 16.88, df = 5 (P = 0.005); I² = 70%); 6 studies, 724 participants; Analysis 4.2).
4.2. Analysis.

Comparison 4: Baclofen versus placebo (divided into detoxified and non‐detoxified participants), Outcome 2: Frequency of use: % days abstinence at end of treatment
1.3 Frequency of use: heavy drinking days at end of treatment
We identified 13 studies involving 840 participants reporting HDDs at the end of treatment. There was no difference between baclofen and placebo (SMD −0.18, 95% CI −0.48 to 0.11; moderate‐certainty evidence; Analysis 1.3; Figure 6; Table 1). There was substantial heterogeneity (Chi² = 41.40.55, df = 12 (P < 0.001); I² = 71%). The study excluded from this meta‐analysis found no difference between baclofen and placebo (MD −2.00, 95% CI −13.22 to 9.22; 30 participants; Leggio 2015).
1.3. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 3: Frequency of use: heavy drinking days at end of treatment
6.

1.3.1 Subgroup analyses: daily doses of baclofen
We found no difference between baclofen and placebo among the studies low, medium and high doses (low: SMD 0.10, 95% CI −0.15 to 0.34; I² = 0%; 6 studies, 278 participants; medium: SMD −0.47, 95% CI −1.15 to 0.20; considerable heterogeneity (Chi² = 28.68, df = 5 (P < 0.001); I² = 83%); 6 studies, 242 participants; high: SMD −0.21, 95% CI −0.43 to 0.01; 1 study, 320 participants; Analysis 2.3).
2.3. Analysis.

Comparison 2: Baclofen versus placebo (divided into low, medium and high doses of baclofen), Outcome 3: Frequency of use: % of heavy drinking days at end of treatment
1.3.2 Subgroup analyses: duration of treatment
We found no difference between baclofen and placebo among 12‐week and longer than 12‐week studies (12 weeks: SMD −0.07, 95% CI −0.27 to 0.13; I² = 0%; 10 studies, 400 participants; longer than 12 weeks: SMD −0.54, 95% CI −1.68 to 0.60; considerable heterogeneity (Chi² = 36.40, df = 2 (P < 0.001); I² = 95%); 3 studies, 440 participants; Analysis 3.3).
3.3. Analysis.

Comparison 3: Baclofen versus placebo (divided into 12‐week and longer than 12‐week studies), Outcome 3: Frequency of use: % of heavy drinking days at end of treatment
1.3.3 Subgroup analyses: consumption of alcohol at beginning of treatment
We found no difference between baclofen and placebo among the studies with detoxified and non‐detoxified participants (toxified: SMD −0.08, 95% CI −0.32 to 0.16; I² = 0%; 8 studies, 296 participants; non‐detoxified: SMD −0.34, 95% CI −0.98 to 0.30; considerable heterogeneity (Chi² = 37.90, df = 4 (P < 0.001); I² = 89%); 5 studies, 544 participants; Analysis 4.3).
4.3. Analysis.

Comparison 4: Baclofen versus placebo (divided into detoxified and non‐detoxified participants), Outcome 3: Frequency of use: % of heavy drinking days at end of treatment
1.4 Amount of use: drinks per drinking days at end of treatment
We identified nine studies involving 392 participants reporting drinks per drinking days at the end of treatment. There were no differences between baclofen and placebo (MD −0.45, 95% CI −1.20 to 0.30; moderate‐certainty evidence; Analysis 1.4; Table 1). There was no important heterogeneity (Chi² = 11.65, df = 8 (P = 0.17); I² = 31%). The study excluded from this meta‐analysis found no difference between baclofen and placebo (MD −0.53, 95% CI −3.11 to 2.05; 30 participants; Leggio 2015).
1.4. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 4: Amount of use: drinks per drinking days at end of treatment
1.4.1 Subgroup analyses: daily doses of baclofen
We found no differences between baclofen and placebo among studies using low and medium doses (low: MD −0.06, 95% CI −1.33 to 1.22; moderate heterogeneity (Chi² = 7.39, df = 4 (P = 0.12); I² = 46%); 5 studies, 242 participants; medium: MD −0.64, 95% CI −1.95 to 0.68; I² = 27%; 4 studies, 150 participants; Analysis 2.4). No study using high doses provided data on this outcome.
2.4. Analysis.

Comparison 2: Baclofen versus placebo (divided into low, medium and high doses of baclofen), Outcome 4: Amount of use: drinks per drinking days at end of treatment
1.4.2 Subgroup analyses: duration of treatment
We found no difference between baclofen and placebo among 12‐week and longer than 12‐week studies (12 weeks: MD −0.36, 95% CI −1.29 to 0.57; moderate heterogeneity (Chi² = 11.07, df = 6 (P = 0.09); I² = 46%); 7 studies, 272 participants; longer than 12 weeks: MD −0.49, 95% CI −2.31 to 1.32; I² = 0%; 2 studies, 120 participants; Analysis 3.4).
3.4. Analysis.

Comparison 3: Baclofen versus placebo (divided into 12‐week and longer than 12‐week studies), Outcome 4: Amount of use: drink per drinking days at end of treatment
1.4.3 Subgroup analyses: consumption of alcohol at beginning of treatment
We found no differences between baclofen and placebo among studies with detoxified and non‐detoxified participants (detoxified: MD −0.36, 95% CI −1.29 to 0.57; moderate heterogeneity (Chi² = 11.07, df = 6 (P = 0.09); I² = 46%); 7 studies, 272 participants; non‐detoxified: MD −0.49, 95% CI −2.31 to 1.32; I² = 0%; 2 studies, 120 participants; Analysis 4.4).
4.4. Analysis.

Comparison 4: Baclofen versus placebo (divided into detoxified and non‐detoxified participants), Outcome 4: Amount of use: drink per drinking days at end of treatment
1.5 Adverse events: number of participants with at least one adverse event at end of treatment
We identified 10 studies involving 738 participants reporting the number of participants with at least one adverse event at the end of treatment. There was no difference between baclofen and placebo (RR 1.05, 95% CI 0.99 to 1.11; high‐certainty evidence; Analysis 1.5; Table 1). There was no important heterogeneity (Chi² = 7.06, df = 9 (P = 0.63); I² = 0%). The study excluded from this meta‐analysis found no difference between baclofen and placebo (RR 1.15, 95% CI 0.91 to 1.44; 30 participants; Leggio 2015).
1.5. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 5: Adverse events: number of participants with at least one adverse event at end of treatment
1.5.1 Subgroup analyses: daily doses of baclofen
We identified no differences between baclofen and placebo among studies using low, medium and high doses (low: RR 1.23, 95% CI 0.92 to 1.64; I² = 0%; 5 studies, 260 participants; medium: RR 0.90, 95% CI 0.63 to 1.28; I² = 0%; 4 studies, 162 participants; high: RR 1.05, 95% CI 0.99 to 1.11; 1 study, 316 participants; Analysis 2.5).
2.5. Analysis.

Comparison 2: Baclofen versus placebo (divided into low, medium and high doses of baclofen), Outcome 5: Adverse events: number of participants with at least one adverse event at end of treatment
1.5.2 Subgroup analyses: duration of treatment
We found no differences between baclofen and placebo among 12‐week and longer than 12‐week studies (12 weeks: RR 0.99, 95% CI 0.70 to 1.39; I² = 0%; 7 studies, 302 participants; longer than 12 weeks: RR 1.05, 95% CI 1.00 to 1.11; I² = 0%; 3 studies, 436 participants; Analysis 3.5).
3.5. Analysis.

Comparison 3: Baclofen versus placebo (divided into 12‐week and longer than 12‐week studies), Outcome 5: Adverse events: number of participants with at least one adverse event at end of treatment
1.5.3 Subgroup analyses: consumption of alcohol at beginning of treatment
We found no differences between baclofen and placebo among studies with detoxified and non‐detoxified participants (detoxified: RR 1.05, 95% CI 0.99 to 1.11; I² = 0%; 7 studies, 578 participants; non‐detoxified: RR 1.11, 95% CI 0.84 to 1.45; I² = 0%; 3 studies, 160 participants; Analysis 4.5).
4.5. Analysis.

Comparison 4: Baclofen versus placebo (divided into detoxified and non‐detoxified participants), Outcome 5: Adverse events: number of participants with at least one adverse event at end of treatment
1.6 Dropouts at end of treatment
We identified 17 studies involving 1563 participants reporting dropouts at the end of treatment. There was no difference between baclofen and placebo (RR 0.88, 95% CI 0.74 to 1.03; high‐certainty evidence; Analysis 1.6; Figure 7; Table 1). There was no important heterogeneity (Chi² = 19.55, df = 16 (P = 0.24); I² = 18%). The study excluded from this meta‐analysis found no difference between baclofen and placebo (RR 0.50, 95% CI 0.11 to 2.33; 30 participants; Leggio 2015).
1.6. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 6: Dropouts at end of treatment
7.

1.6.1 Subgroup analyses: daily doses of baclofen
We found no differences between baclofen and placebo among studies using low and medium doses (low: RR 0.96, 95% CI 0.70 to 1.32; I² = 16%; 8 studies, 564 participants; medium: RR 1.03, 95% CI 0.65 to 1.61; I² = 19%; 5 studies, 214 participants). Baclofen decreased the number of dropouts at the end of treatment compared with placebo among the studies using high doses (RR 0.76, 95% CI 0.67 to 0.87; I² = 0%; 4 studies, 785 participants; Analysis 2.6).
2.6. Analysis.

Comparison 2: Baclofen versus placebo (divided into low, medium and high doses of baclofen), Outcome 6: Dropouts at end of treatment
1.6.2 Subgroup analyses: duration of treatment
We found no differences between baclofen and placebo among 12‐week studies (RR 0.98, 95% CI 0.73 to 1.31; I² = 13%; 11 studies, 652 participants; Analysis 3.6). Baclofen decreased the number of dropouts at the end of treatment compared with placebo in longer than 12‐week studies (RR 0.78, 95% CI 0.69 to 0.88; I² = 0%; 6 studies, 911 participants; Analysis 3.6).
3.6. Analysis.

Comparison 3: Baclofen versus placebo (divided into 12‐week and longer than 12‐week studies), Outcome 6: Dropouts at end of treatment
1.6.3 Subgroup analyses: consumption of alcohol at beginning of treatment
There were no differences between baclofen and placebo among studies with detoxified and non‐detoxified participants (detoxified: RR 0.87, 95% CI 0.71 to 1.07; I² = 0%; 12 studies, 879 participants; non‐detoxified: RR 0.93, 95% CI 0.69 to 1.28; moderate heterogeneity (Chi² = 8.28, df = 4 (P = 0.08); I² = 52%); 5 studies, 684 participants; Analysis 4.6).
4.6. Analysis.

Comparison 4: Baclofen versus placebo (divided into detoxified and non‐detoxified participants), Outcome 6: Dropouts at end of treatment
1.7 Dropouts from treatment due to adverse events
We identified 16 studies involving 1499 participants reporting dropouts from treatment due to adverse events. There was no difference between baclofen and placebo (RR 1.39, 95% CI 0.89 to 2.18; high‐certainty evidence; Analysis 1.7 and Figure 8; Table 1). There was no important heterogeneity (Chi² = 7.55, df = 10 (P = 0.67); I² = 0%). The study excluded from this meta‐analysis found zero events in both groups (RR not estimable; Leggio 2015).
1.7. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 7: Dropouts due to adverse events
8.

1.7.1 Subgroup analyses: daily doses of baclofen
We found no differences between baclofen and placebo among studies using low, medium and high doses (low: RR 1.81, 95% CI 0.61 to 5.32; I² = 0%; 8 studies, 564 participants; medium: RR 6.11, 95% CI 0.82 to 42.25; I² = 0%; 4 studies, 150 participants; high: RR 1.21, 95% CI 0.68 to 2.13; I² = 13%; 4 studies, 785 participants; Analysis 2.7).
2.7. Analysis.

Comparison 2: Baclofen versus placebo (divided into low, medium and high doses of baclofen), Outcome 7: Dropouts due to adverse events
1.7.2 Subgroup analyses: duration of treatment
We found no differences between baclofen and placebo among 12‐week and longer than 12‐week studies (12 weeks: RR 3.00, 95% CI 0.93 to 9.66; I² = 0%; 10 studies, 588 participants; longer than 12‐weeks: RR 1.22, 95% CI 0.76 to 1.98; I² = 0%; 6 studies, 911 participants; Analysis 3.7).
3.7. Analysis.

Comparison 3: Baclofen versus placebo (divided into 12‐week and longer than 12‐week studies), Outcome 7: Dropout due to adverse events
1.7.3 Subgroup analyses: consumption of alcohol at beginning of treatment
We found no differences between baclofen and placebo among studies with detoxified and non‐detoxified participants (toxified: RR 1.08, 95% CI 0.59 to 1.98; I² = 0%; 12 studies, 879 participants; non‐detoxified: RR 1.87, 95% CI 0.97 to 3.61; I² = 0%; 4 studies, 620 participants; Analysis 4.7).
4.7. Analysis.

Comparison 4: Baclofen versus placebo (divided into detoxified and non‐detoxified participants), Outcome 7: Dropouts due to adverse events
1.8 Craving
We identified 17 studies with 1275 people reporting cravings. There was no difference between baclofen and placebo (SMD −0.16, 95% CI −0.37 to 0.04). Heterogeneity was substantial (Chi² = 39.36, df = 15 (P < 0.001); I² = 62%; Analysis 1.8).
1.8. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 8: Craving
1.9 Anxiety
We identified 15 studies with 1123 participants reporting anxiety. There was no difference between baclofen and placebo (SMD −0.01, 95% CI −0.14 to 0.11; Analysis 1.9).
1.9. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 9: Anxiety
1.10 Depression
We identified 11 studies with 1029 participants reporting depression. There was no difference between baclofen and placebo (SMD 0.07, 95% CI −0.12 to 0.27; Analysis 1.10).
1.10. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 10: Depression
1.11 Adverse events: fatigue, tiredness
We identified 13 studies involving 1311 participants. Baclofen increased the frequency of fatigue or tiredness (or both) compared with placebo (RR 1.31, 95% CI 1.09 to 1.59; Analysis 1.11).
1.11. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 11: Adverse events: fatigue, tiredness
1.12 Adverse events: insomnia
We identified 11 studies with 1100 participants reporting insomnia. There was no difference between baclofen and placebo (RR 0.92, 95% CI 0.62 to 1.35; Analysis 1.12).
1.12. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 12: Adverse events: insomnia
1.13 Adverse events: pain (diverse)
We identified three studies with 98 participants reporting pain. There was no difference between baclofen and placebo (RR 0.51, 95% CI 0.19 to 1.33; Analysis 1.13).
1.13. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 13: Adverse events: pain (diverse)
1.14 Adverse events: vertigo, dizziness
We identified 15 studies with 1415 participants. Baclofen increased the frequency of vertigo compared with placebo (RR 1.70, 95% CI 1.35 to 2.13; Analysis 1.14).
1.14. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 14: Adverse events: vertigo, dizziness
1.15 Adverse events: constipation
We identified 11 studies with 1124 participants reporting constipation. There was no difference between baclofen and placebo (RR 0.95, 95% CI 0.61 to 1.47; Analysis 1.15).
1.15. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 15: Adverse events: constipation
1.16 Adverse events: somnolence, sleepiness, drowsiness or sedation
We identified 17 studies with 1543 participants. Baclofen increased the frequency of somnolence compared with placebo (RR 1.36, 95% CI 1.19 to 1.56; Analysis 1.16).
1.16. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 16: Adverse events: somnolence, sleepiness, drowsiness or sedation
1.17 Adverse events: muscle pain
We identified five studies with 594 participants reporting muscle pain. There was no difference between baclofen and placebo (RR 0.93, 95% CI 0.38 to 2.25; Analysis 1.17).
1.17. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 17: Adverse events: muscle pain
1.18 Adverse events: dry mouth
We identified eight studies with 933 participants. Baclofen increased the frequency of dry mouth compared with placebo (RR 1.79, 95% CI 1.01 to 3.18; Analysis 1.18).
1.18. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 18: Adverse events: dry mouth
1.19 Adverse events: nausea
We identified 10 studies with 1060 participants reporting nausea. There was no difference between baclofen and placebo (RR 1.17, 95% CI 0.88 to 1.54; Analysis 1.19).
1.19. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 19: Adverse events: nausea
1.20 Adverse events: skin rash
We identified seven studies with 446 participants reporting skin rash. There was no difference between baclofen and placebo (RR 0.80, 95% CI 0.33 to 1.93; Analysis 1.20).
1.20. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 20: Adverse events: skin rash
1.21 Adverse events: headaches
We identified 13 studies with 1304 participants. Baclofen increased the frequency of headaches compared with placebo (RR 1.31, 95% CI 1.04 to 1.64; Analysis 1.21).
1.21. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 21: Adverse events: headaches
1.22 Adverse events: paraesthesia/numbness
We identified six studies with 914 participants. Baclofen increased the frequency of paraesthesia (RR 2.87, 95% CI 1.17 to 7.06; Analysis 1.22).
1.22. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 22: Adverse events: paraesthesia/numbness
1.23 Adverse events: diarrhoea
We identified three studies with 816 participants. There was no difference between baclofen and placebo (RR 0.64, 95% CI 0.40 to 1.02; Analysis 1.23).
1.23. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 23: Adverse events: diarrhoea
1.24 Adverse events: tinnitus
We identified two studies with 496 participants reporting tinnitus. There was no difference between baclofen and placebo (RR 1.77, 95% CI 0.16 to 20.16). Heterogeneity was considerable (Chi² = 8.44, df = 1 (P = 0.004); I² = 88%; Analysis 1.24).
1.24. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 24: Adverse events: tinnitus
1.25 Adverse events: muscle spasm/rigidity
We identified three studies with 552 participants. Baclofen increased the frequency of muscle spasm (RR 1.94, 95% CI 1.07 to 3.52; Analysis 1.25).
1.25. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 25: Adverse events: muscle spasm/rigidity
1.26 Adverse events: hyperhidrosis
We identified three studies with 816 participants reporting hyperhidrosis. There was no difference between baclofen and placebo (RR 1.50, 95% CI 0.89 to 2.51; Analysis 1.26).
1.26. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 26: Adverse events: hyperhidrosis
1.27 Adverse events: nasopharyngitis
We identified four studies with 872 participants reporting nasopharyngitis. There was no difference between baclofen and placebo (RR 0.89, 95% CI 0.40 to 1.95). Heterogeneity was considerable (Chi² = 7.53, df = 3 (P = 0.06); I² = 60%; Analysis 1.27).
1.27. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 27: Adverse events: nasopharyngitis
1.28 Adverse events: decreased appetite/anorexia
We identified three studies with 816 participants reporting decreased appetite/anorexia. There was no difference baclofen and placebo (RR 0.74, 95% CI 0.31 to 1.75). Heterogeneity was substantial (Chi² = 5.16, df = 2 (P = 0.08); I² = 61%; Analysis 1.28).
1.28. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 28: Adverse events: decrease appetite/anorexia
1.29 Adverse events: dysgeusia/ageusia
We identified three studies with 816 participants reporting dysgeusia/ageusia. There was no difference between baclofen and placebo (RR 1.73, 95% CI 0.57 to 5.24). Heterogeneity was moderate (Chi² = 3.65, df = 2 (P = 0.16); I² = 45%; Analysis 1.29).
1.29. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 29: Adverse events: dysgeusia/ageusia
1.30 Adverse events: tremor
We identified three studies with 816 participants reporting tremor. There was no difference between baclofen and placebo (RR 0.69, 95% CI 0.42 to 1.15; Analysis 1.30).
1.30. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 30: Adverse events: tremor
1.31 Adverse events: weakness
We identified three studies with 552 participants reporting weakness. There was no difference between baclofen and placebo (RR 1.19, 95% CI 0.70 to 2.01; Analysis 1.31).
1.31. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 31: Adverse events: weakness
1.32 Adverse events: vomiting
We identified three studies with 816 participants reporting vomiting. There was no difference between baclofen and placebo (RR 1.07, 95% CI 0.66 to 1.71; Analysis 1.32).
1.32. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 32: Adverse events: vomiting
1.33 Adverse events: urinary frequency
We identified four studies with 340 participants reporting urinary frequency. There was no difference between baclofen and placebo (RR 1.10, 95% CI 0.45 to 2.68; Analysis 1.33).
1.33. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 33: Adverse events: urinary frequency
1.34 Adverse events: shortness of breath
We identified two studies with 104 participants reporting shortness of breath. There was no difference between baclofen and placebo (RR 0.89, 95% CI 0.04 to 22.31; Analysis 1.34).
1.34. Analysis.

Comparison 1: Baclofen versus placebo (all studies), Outcome 34: Adverse events: shortness of breath
Comparison 2: baclofen versus acamprosate
Two RCTs compared baclofen to acamprosate (Kumar 2020a; Mishra 2010).
2.1 Relapse: return to any drinking at end of treatment
We identified one study with 60 participants reporting return to any drinking at the end of treatment. There was no difference between baclofen and acamprosate (RR 1.25, 95% CI 0.71 to 2.20; very low‐certainty evidence; Analysis 5.1; Table 2).
5.1. Analysis.

Comparison 5: Baclofen versus acamprosate, Outcome 1: Relapse: return to any drinking at end of treatment
2.2 Adverse events: number of participants with at least one adverse event at end of treatment
We identified one study with 60 participants reporting number of participants with at least one adverse event at the end of treatment. There was no difference between baclofen and acamprosate (RR 0.63, 95% CI 0.23 to 1.69; very low‐certainty evidence; Analysis 5.2; Table 2).
5.2. Analysis.

Comparison 5: Baclofen versus acamprosate, Outcome 2: Adverse events: number of participants with at least one adverse event at end of treatment
2.3 Dropouts at end of treatment
We identified one study with 60 participants reporting dropouts at the end of treatment. There was no difference between baclofen and acamprosate (RR 0.56, 95% CI 0.21 to 1.46; very low‐certainty evidence; Analysis 5.3; Table 2).
5.3. Analysis.

Comparison 5: Baclofen versus acamprosate, Outcome 3: Dropouts at end of treatment
2.4 Dropouts due to adverse events
We identified one study with 60 participants reporting dropouts due to adverse events. There was no difference between baclofen and acamprosate (RR 0.33, 95% CI 0.01 to 7.87; very low‐certainty evidence; Analysis 5.4; Table 2).
5.4. Analysis.

Comparison 5: Baclofen versus acamprosate, Outcome 4: Dropouts due to adverse events
2.5 Craving
We identified two studies with 109 participants. There was no difference between baclofen and acamprosate (MD 5.80, 95% CI −11.84 to 23.44). Heterogeneity was considerable (Chi² = 38.87, df = 1 (P < 0.001); I² = 97%; Analysis 5.5).
5.5. Analysis.

Comparison 5: Baclofen versus acamprosate, Outcome 5: Craving
2.6 Adverse events: fatigue, tiredness
We identified one study with 60 participants reporting fatigue or tiredness (or both). There was no difference between baclofen and acamprosate (RR 3.00, 95% CI 0.13 to 70.83; Analysis 5.6).
5.6. Analysis.

Comparison 5: Baclofen versus acamprosate, Outcome 6: Adverse events: fatigue, tiredness
2.7 Adverse events: vertigo, dizziness
We identified one study with 60 participants reporting vertigo or dizziness (or both). There was no difference between baclofen and acamprosate (RR 2.00, 95% CI 0.19 to 20.90; Analysis 5.7).
5.7. Analysis.

Comparison 5: Baclofen versus acamprosate, Outcome 7: Adverse events: vertigo, dizziness
2.8 Adverse events: nausea
We identified one study with 60 participants reporting nausea. There was no difference between baclofen and acamprosate (RR 0.33, 95% CI 0.01 to 7.87; Analysis 5.8).
5.8. Analysis.

Comparison 5: Baclofen versus acamprosate, Outcome 8: Adverse events: nausea
2.9 Adverse events: skin rash
We identified one study with 60 participants reporting skin rash. There was no difference between baclofen and acamprosate (RR 0.20, 95% CI 0.01 to 4.00; Analysis 5.9).
5.9. Analysis.

Comparison 5: Baclofen versus acamprosate, Outcome 9: Adverse events: skin rash
2.10 Adverse events: decreased appetite/anorexia
We identified one study with 60 participants reporting decreased appetite/anorexia. There was no difference between baclofen and acamprosate (RR 1.00, 95% CI 0.15 to 6.64; Analysis 5.10).
5.10. Analysis.

Comparison 5: Baclofen versus acamprosate, Outcome 10: Adverse events: decrease appetite/anorexia
2.11 Adverse events: acidity
We identified one study with 60 participants reporting acidity. There was no difference between baclofen and acamprosate (RR 0.33, 95% CI 0.01 to 7.87; Analysis 5.11).
5.11. Analysis.

Comparison 5: Baclofen versus acamprosate, Outcome 11: Adverse events: acidity
2.12 Adverse events: palpitations
We identified one study with 60 participants reporting palpitations. There was no difference between baclofen and acamprosate (RR 0.33, 95% CI 0.01 to 7.87; Analysis 5.12).
5.12. Analysis.

Comparison 5: Baclofen versus acamprosate, Outcome 12: Adverse events: palpitations
2.13 Adverse events: tremor
We identified one study with 60 participants reporting tremor (Kumar 2020a). There were no events in either group.
2.14 Adverse events: erectile dysfunction
We identified one study with 60 participants reporting erectile dysfunction (Kumar 2020a). There were no events in either group.
Comparison 3: baclofen versus naltrexone
Two RCTs compared baclofen to naltrexone (Garbutt 2010b2; Kumar 2020a).
3.1 Relapse: return to any drinking at end of treatment
We identified one study with 60 participants reporting return to any drinking at the end of treatment. Naltrexone reduced the risk of relapse compared to baclofen (RR 2.50, 95% CI 1.12 to 5.56; very low‐certainty evidence; Analysis 6.1; Table 3).
6.1. Analysis.

Comparison 6: Baclofen versus naltrexone, Outcome 1: Relapse: return to any drinking at end of treatment
3.2 Frequency of use: percentage of days abstinent at end of treatment
We identified one study with 20 participants (Garbutt 2010b1). The percentage of days abstinent at the of treatment was 55.4% with baclofen and 33.4% with placebo. Standard deviations were not reported.
3.3 Frequency of use: percentage of heavy drinking days at end of treatment
We identified one study with 20 participants (Garbutt 2010b1). The percentage of HDDs at the end of treatment was 17.9% with baclofen and 10.7% with placebo. Standard deviations were not reported.
3.4 Adverse events: number of participants with at least one adverse event at end of treatment
We identified two studies with 80 participants. Naltrexone may increase the risk of adverse events slightly compared to baclofen (RR 0.35, 95% CI 0.15 to 0.80; very low‐certainty evidence; Table 3). There was no heterogeneity (Chi² = 0.73, df = 1 (P = 0.39); I² = 0%; Analysis 6.2).
6.2. Analysis.

Comparison 6: Baclofen versus naltrexone, Outcome 2: Adverse events: number of participants with at least one adverse event at end of treatment
3.5 Dropouts at end of treatment
We identified one study with 60 participants. There was no difference between baclofen and naltrexone (RR 1.00, 95% CI 0.32 to 3.10; very low‐certainty evidence; see Table 3). There was no heterogeneity (Chi² = 0.73, df = 1 (P = 0.39); I² = 0%; Analysis 6.3).
6.3. Analysis.

Comparison 6: Baclofen versus naltrexone, Outcome 3: Dropouts at end of treatment
3.6 Dropouts due to adverse events
We identified one study with 60 participants (Kumar 2020a). There were no dropouts in either group.
3.7 Craving
We identified one study with 60 participants reporting cravings. There was no difference between baclofen and naltrexone (MD 2.08, 95% CI −3.71 to 7.87; Analysis 6.4).
6.4. Analysis.

Comparison 6: Baclofen versus naltrexone, Outcome 4: Craving
3.8 Adverse events: fatigue, tiredness
We identified one study with 60 participants reporting fatigue or tiredness (or both). There was no difference between baclofen and naltrexone (RR 3.00, 95% CI 0.13 to 70.83; Analysis 6.5).
6.5. Analysis.

Comparison 6: Baclofen versus naltrexone, Outcome 5: Adverse events: fatigue, tiredness
3.9 Adverse events: insomnia
We identified one study with 20 participants reporting insomnia. There was no difference between baclofen and naltrexone (RR 0.14, 95% CI 0.01 to 2.45; Analysis 6.6).
6.6. Analysis.

Comparison 6: Baclofen versus naltrexone, Outcome 6: Adverse events: insomnia
3.10 Adverse events: vertigo, dizziness
We identified one study with 60 participants reporting vertigo or dizziness or both. There was no difference between baclofen and naltrexone (RR 0.40, 95% CI 0.08 to 1.90; Analysis 6.7).
6.7. Analysis.

Comparison 6: Baclofen versus naltrexone, Outcome 7: Adverse events: vertigo, dizziness
3.11 Adverse events: somnolence, sleepiness, drowsiness or sedation
We identified one study with 20 participants reporting somnolence, sleepiness, drowsiness or sedation. There was no difference between baclofen and naltrexone (RR 0.33, 95% CI 0.04 to 2.69; Analysis 6.8).
6.8. Analysis.

Comparison 6: Baclofen versus naltrexone, Outcome 8: Adverse events: somnolence, sleepiness, drowsiness or sedation
3.12 Adverse events: nausea
We identified two studies with 80 participants reporting nausea. There was no difference between baclofen and naltrexone (RR 0.26, 95% CI 0.03 to 2.20; Analysis 6.9).
6.9. Analysis.

Comparison 6: Baclofen versus naltrexone, Outcome 9: Adverse events: nausea
3.13 Adverse events: skin rash
We identified one study with 60 participants reporting skin rash (Kumar 2020a). There were no events in either group.
3.14 Adverse events: decreased appetite/anorexia
We identified one study with 60 participants. There was no difference between baclofen and naltrexone (RR 2.00, 95% CI 0.19 to 20.90; Analysis 6.10).
6.10. Analysis.

Comparison 6: Baclofen versus naltrexone, Outcome 10: Adverse events: decrease appetite/anorexia
3.15 Adverse events: tremor
We identified one study with 60 participants reporting tremor. There was no difference between baclofen and naltrexone (RR 0.33, 95% CI 0.01 to 7.87; Analysis 6.11).
6.11. Analysis.

Comparison 6: Baclofen versus naltrexone, Outcome 11: Adverse events: tremor
3.16 Adverse events: acidity
We identified one study with 60 participants reporting acidity. There was no difference between baclofen and naltrexone (RR 0.20, 95% CI 0.01 to 4.00; Analysis 6.12).
6.12. Analysis.

Comparison 6: Baclofen versus naltrexone, Outcome 12: Adverse events: acidity
3.17 Adverse events: palpitations
We identified one study with 60 participants reporting palpitations (Kumar 2020a). There were no events in either group.
3.18 Adverse events: erectile dysfunction
We identified one study with 60 participants reporting erectile dysfunction. There was no difference between baclofen and naltrexone (RR 0.33, 95% CI 0.01 to 7.87; Analysis 6.13).
6.13. Analysis.

Comparison 6: Baclofen versus naltrexone, Outcome 13: Adverse events: erectile dysfunction
Discussion
Summary of main results
We included 17 RCTs with 1818 participants. Ten RCTs had a single arm and the other 7 RCTs had two arms each. Fifteen RCTs compared baclofen with placebo, two RCTs compared baclofen with acamprosate, and two RCTs compared baclofen with naltrexone.
Comparison 1: baclofen versus placebo
In comparison to placebo, meta‐analyses found moderate‐certainty evidence suggesting that baclofen probably reduces the risk to return to any drinking and high‐certainty evidence that it increases the rate of abstinent days at the end of treatment. Subgroup analyses found evidence that baclofen may reduce the risk to return to any drinking among detoxified participants but not among other subgroups of participants. We found evidence that baclofen may increase the rate of abstinent days in all the subgroups of participants except among those who received moderate doses of baclofen.
In addition, meta‐analyses found moderate‐certainty evidence suggesting that baclofen probably has no effect in the rate of HDDs and in the number of drinks per drinking days, and high‐certainty evidence that it does not increase the number of participants with at least one adverse event, the number of participants who dropped out for any reason, and those who dropped out due to adverse events at the end of treatment. These results were confirmed in all the subgroup analyses except for dropout that was lower among participants who received high doses of baclofen compared to placebo and among participants recruited to studies longer than 12 weeks.
Meta‐analyses of all the studies found no differences between baclofen and placebo in craving, anxiety, and depression.
Finally, compared to placebo, baclofen increased the frequency of fatigue, dizziness, somnolence, dry mouth, paraesthesia and muscle spasms while, for all the other adverse events, there were no differences between baclofen and placebo.
Comparison 2: baclofen versus acamprosate
Compared to acamprosate, meta‐analyses found no differences in any primary outcome evaluated: relapse, number of participants with at least one adverse event, dropouts, dropouts due to adverse events (very low‐certainty evidence). We found no differences between baclofen and acamprosate in craving and nine different adverse events. However, most of these meta‐analyses were based on a single study with 60 participants, so should be interpreted with caution.
Comparison 3: baclofen versus naltrexone
Compared to naltrexone, meta‐analyses found very low‐certainty evidence that baclofen may increase the risk of relapse and decrease slightly the number of participants with at least one adverse event. We found no differences between the treatments in dropouts at the end of treatment (very low‐certainty evidence), craving, and 11 different adverse events. However, for most of these meta‐analyses, results were based on a single study of 60 participants, so should be interpreted with caution.
Overall completeness and applicability of evidence
Overall completeness
In this review, the completeness rates of the analyses of primary outcomes range from 36% to 86%. In detail, considering that our database comprised 14 RCTs (after the exclusion of one study because of heterogeneity), the completeness rates of primary outcome analyses were the following: relapse (return to any drinking): 8 RCTs (57.1%); percentage of days abstinent: 11 RCTs (78.6%); HDDs: 9 RCTs (64.3%); number of drinks per drinking day: 5 RCTs (35.7%); number of people with at least one adverse event: 6 RCTs (42.9%); dropouts from treatment: 12 RCTs (85.7%); and dropouts due to adverse events: 11 RCTs (78.6%). The completeness rates of the analyses of secondary outcomes ranged from 7.1% (shortness of breath; 1 RCT) to 85.7% (sedation; 12 RCTs). The completeness rates of primary outcomes overlap those of other Cochrane Reviews on other medications for the treatment of AUD reflecting the large variety of outcomes used to evaluate the effectiveness of treatments in the field of AUD research (Rösner 2010a; Rösner 2010b). Despite this large variety, the present review achieves a good overall completeness. The larger range of completeness of secondary outcomes was mainly due to the inclusion of rare specific adverse event (e.g. shortness of breath) into the list of secondary outcomes.
Applicability of evidence
The RCTs included in this review largely vary in the daily dose of baclofen, titration schedules, duration of treatment, comorbidity, and baseline alcohol use. Despite these differences among RCTs and their participants, meta‐analyses including all the selected RCTs found moderate‐certainty evidence that baclofen reduces the risk of relapse and subgroup analyses confirmed this evidence among certain participants (i.e. detoxified participants) but not others (non‐detoxified participants). Similarly, including all the RCTs, this review found high‐certainty evidence that baclofen increases the rate of abstinent days and subgroup analyses confirmed this evidence among almost all the subgroup analyses. Finally, this review found moderate‐ to high‐certainty evidence that baclofen probably has no effect in the other primary outcomes (HDDs, drinks per drinking days, participants with at least one adverse event, participants who dropout for any reason, participants who dropped out due to adverse events) and this evidence was confirmed by almost all the subgroup analyses. Globally, subgroup analyses sustained the generalisability of the evidence of the demonstrated results.
In addition, RCTs selected by this review took place in different countries including the USA (29.4%); Australia, France, Italy and India (11.8% each); and Germany, Israel, the Netherlands and Russia (5.9% each). This is an important issue in terms of generalisability of the evidence, because different social contexts can influence AUD severity and the availability to receive medical treatment for this disorder.
However, generalisability might be restricted from the exclusion of participants with severe psychiatric disorders by most of the selected RCTs. Furthermore, this review found that baclofen increased the risk of adverse events such as fatigue, dizziness and somnolence but did not increase the number of participants with at least one adverse event. Subgroup analyses indicated that no daily dose of baclofen increased the number of participants with at least one adverse event. However, the subgroup of high doses included only one large RCT (Reynaud 2017). These findings underline the paucity of literature for some important outcomes. Nevertheless, our findings do not allow conclusions on the safety of baclofen in combination with other sedative drugs (e.g. alcohol and benzodiazepines) or in specific patient subgroups, such as people with suicidal ideation (e.g. Agabio 2018a; de Beaurepaire 2019; Holla 2015; Pelissier 2017).
Finally, the results of two recent studies suggest that women may respond better to baclofen for the treatment of AUD than men (Garbutt 2021‐LD; Garbutt 2021‐MD; Morley 2022). In detail, one study observed that, compared to men, women required lower doses of baclofen to increase the rate of abstinent days (Garbutt 2021‐LD; Garbutt 2021‐MD). Another study found that baclofen delayed the time to the first drink for women with AUD but not men (Morley 2022). These results provide some support for the hypothesis that gender may be a potential moderator of baclofen response in the treatment of AUD. Although medications for the treatment of AUD have been studied almost exclusively in men (Agabio 2016), these findings are in line with other studies showing numerous gender differences in response to the other pharmacological treatments (Mauvais‐Jarvis 2021). Unfortunately, the generalisability might be restricted as most RCTs did not provide separate data for men and women.
Quality of the evidence
Applying the criteria recommended in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011), we judged most of the RCTs to have a low or unclear risk of selection, performance, detection (subjective outcome), attrition and reporting bias.
One potential threat of bias that concerns all RCTs applying inert placebo is the unmasking of blinding, which is a methodological limitation that is associated with an overestimation of effects for antidepressant substances (Moncrieff 2004). Even though baclofen and placebo were of identical appearance in most RCTs, perceptible differences between treatment groups such as adverse events can reveal participants' affiliation to treatment conditions. Thus, from a theoretical perspective, we could not definitely exclude the possibility that proof of efficacy in some studies is attributable to an overestimation of effects caused by an unmasking of treatment allocation.
Overall, we rated the certainty of evidence as high for the outcomes of percentage of days abstinent, participants with at least one adverse event, dropouts due to any reason and dropouts due to adverse events; moderate for relapse to any drinking, HDDs, number of drinks per drinking days and percent days abstinent. Reasons for downgrading were heterogeneity among study for the outcomes relapse and HDDs and inconsistency for the outcome drinks per drinking days.
Potential biases in the review process
To lower the risk of bias in the review process, two review authors independently screened abstracts, assessed full texts, rated risk of bias for included studies and extracted data from primary studies. None of the review authors had any personal, scientific or financial conflicts of interest or expected advantages from significant results in terms of reviewer decisions or raising funds, lowering the risk for confirmation bias (Nickerson 1998), and significance bias (Kagereki 2016). Visual inspection of funnel plots for the analyses with more than 10 studies showed no asymmetry suggesting the risk of publication bias. Furthermore, we wrote to the authors of published studies and searched on ClincialTrials.gov and ICTRP to search for unpublished studies to minimise the risk of publication bias.
Agreements and disagreements with other studies or reviews
Compared to the previous Cochrane (Minozzi 2018) and non‐Cochrane meta‐analyses (Agabio 2021b; Bschor 2018; Lesouef 2014; Pierce 2018; Rose 2018), the present review included four recent RCTs (Garbutt 2021‐LD; Garbutt 2021‐MD; Kumar 2020a; Morley 2018‐LD; Morley 2018‐MD; Rigal 2020), and one previously unavailable RCT (Addolorato 2011‐LD; Addolorato 2011‐MD).
The results of the present review suggest that baclofen may reduce the risk to return to any drinking. Other non‐Cochrane systematic reviews found that baclofen significantly increases the number of abstinent participants (Lesouef 2014; Pierce 2018; Rose 2018). As return to any drinking and abstinent participants are complementary outcomes, these results of the present review are in line with those of other systematic reviews. In addition, the present review provides the results of subgroup analyses conducted dividing participants according to the daily dose of baclofen, the duration of the studies and the consumption of alcohol at the beginning of the treatment. These subgroup analyses showed that baclofen probably reduces the risk to return to any drinking (or increases the number of abstinent participants) only among participants already detoxified at the beginning of treatment but not among other subgroups of participants (e.g. participants who were still drinking at the beginning of treatment).
The present review found that baclofen increases the rate of abstinent days at the end of treatment while the previous version of this Cochrane Review (Minozzi 2018), and many of the non‐Cochrane Reviews (Bschor 2018; Lesouef 2014; Pierce 2018; Rose 2018), found no differences between baclofen and placebo in this outcome. However, another systematic review found that baclofen may increase the rate of abstinent days (Agabio 2021b). As this review was aimed at investigating the potential role of anxiety in influencing the effects of baclofen, it included only RCTs that provided anxiety levels of participants both at baseline and at the end of treatment. This review found that baclofen increases the rate of abstinent days only among participants with high anxiety levels and not among participants with low anxiety levels at baseline (Agabio 2021b). These findings suggest that baclofen may be useful only for certain people with AUD and that treatment should be personalised.
The present review found moderate‐certainty evidence of no difference between baclofen and placebo in the number of drinks per drinking days. In the previous version of this review, we found that baclofen increased the number of drinks per drinking days compared to placebo (Minozzi 2018). However, this finding was mainly based on one study that we excluded in the present update from the meta‐analyses because of the elevated heterogeneity due to its inclusion and the substantial difference of the participants and setting of this study when compared to the other included studies (Leggio 2015).
Finally, the present review found no difference between baclofen and placebo in anxiety, depression and anxiety. These findings are in line with those of previous meta‐analyses (Agabio 2021b; Lesouef 2014; Minozzi 2018; Rose 2018).
Authors' conclusions
Implications for practice.
Based on evidence from 17 randomised controlled trials with 1818 participants with alcohol use disorder (AUD), the review demonstrates the effectiveness of baclofen for treatment of AUD in reducing the risk of relapse and increasing the percentage of abstinent days. Compared to placebo, baclofen reduces the risk to return to any drinking at the end of treatment by approximately 13%. That is, 13 additional participants out of 100 avoid any drinking, for the entire duration of treatment when they receive baclofen compared to participants who receive placebo. This finding is particularly relevant considering that the duration of treatment of studies selected by this review ranged from three months to one year. Among detoxified participants, this effect was higher with an additional 27 participants out of 100 maintaining abstinence when treated with baclofen compared to placebo.
Regarding the percentage of abstinent days, baclofen increases this percentage by 9% compared to placebo. This finding means that participants treated with baclofen spend almost three additional abstinent days, per month, compared to participants who receive placebo. This effect is equal to almost four additional abstinent days, per month, among the subgroup of detoxified participants, compared to participants who receive placebo.
This review also demonstrates that baclofen does not increase the number of participants with at least one adverse event at the end of treatment, compared to placebo. That means that participants who receive baclofen do not experience a higher number of adverse events compared to those who receive placebo. In addition, participants who receive baclofen do not differ from those who receive placebo in the numbers who prematurely interrupt the trial because of any reason or adverse events. Similarly, baclofen does seem to reduce HDDs, the number of drinks per drinking days, or the severity of craving, anxiety, and depression. These findings mean that baclofen does not have an effect in other important measures of alcohol consumption compared to placebo.
Finally, this review demonstrates that, compared to placebo, baclofen increases specific adverse events such as fatigue, dizziness and somnolence/sedation but not others.
Conversely, this review provides no evidence on possible differences between baclofen and acamprosate or naltrexone because of the limited number of the available studies for these comparisons.
Regarding its safety, our findings do not allow conclusions on the possible risk related to the combination of baclofen with sedative drugs (e.g. alcohol or benzodiazepines, or both) or in specific patient subgroups, such as people with suicidal ideation.
Globally, current evidence suggests that baclofen may help people with AUD aimed at maintaining abstinence.
Implications for research.
Further studies should investigate baclofen's profile of efficacy and safety among certain categories of people with AUD such as people with other mental disorders (bipolar disorder, anxiety disorders) or physical disorders (e.g. severe liver disease) and data should always be reported separately for men and women to investigate possible gender differences.
One potential threat to bias that needs to be further addressed by research is the unmasking of blinding in the course of treatment. Even though quality of blinding is ensured by various measures of bias control, such as allocation concealment and blinding of participants and personnel, adverse events can potentially reveal a participant's affiliation to treatment conditions in the course of treatment. The use of active placebo mimicking adverse events of baclofen could prevent an overestimation of effects through response bias, placebo effects or differential attrition (Hróbjartsson 2014). If inert placebo is used, testing of blinding integrity by querying participants about their assumed group allocation would be a simple method that allows at least a retrospective assessment of blinding integrity (Wilsey 2016).
The exploration of baclofen's profile of efficacy and safety and the identification of moderators and mediators of baclofen's effects remains a challenge mainly due to the risk of specific adverse events such as sedation. Preclinical data suggest that γ‐aminobutyric acid type B (GABAB)‐positive allosteric modulators may constitute the possible solution to overcome these risks (Maccioni 2019). GABAB‐positive allosteric modulators indeed reproduce baclofen‐induced effects on alcohol‐related behaviours displaying a more favourable safety profile. At present, the first clinical trial to evaluate the efficacy and safety of a GABAB‐positive allosteric modulator for the treatment of AUD is ongoing (NCT05096117).
As most people with AUD still receive no treatment (Han 2021), the development of new, effective, safe and manageable medications is expected to considerably increase the number of people who seek and receive medical treatment.
What's new
| Date | Event | Description |
|---|---|---|
| 5 May 2022 | New search has been performed | The latest search was conducted on 22 November 2021 and identified five new studies. These studies were added to the previously identified 12 studies. Meta‐analyses of these 17 studies were updated and subgroups analyses were conducted. |
| 5 May 2022 | New citation required and conclusions have changed | Baclofen likely reduces the risk of relapse to any drinking and increases the percentage of abstinent days, mainly among detoxified participants. It does not increase the number of participants with at least one adverse event, those who dropout for any reason, and those who dropout due to adverse events. It probably does not reduce heavy drinking days and the number of drinks per drinking days. Current evidence suggests that baclofen may help people with alcohol use disorder in maintaining abstinence. The results of comparisons of baclofen with naltrexone and acamprosate were mainly based on a single study and do not allow conclusions. |
History
Protocol first published: Issue 2, 2017 Review first published: Issue 11, 2018
Acknowledgements
We thank Zuzana Mitrova for the editorial support.
Appendices
Appendix 1. Cochrane Drug and Alcohol Group Specialised Register search strategy
CDAG Specialised Register (via CRSLive)
From 2018 to 22 November 2021 (53 hits)
baclofen (all fields)
Appendix 2. CENTRAL search
Cochrane Central Register of Controlled Trials (CENTRAL) (via onlinelibrary.wiley.com)
2021, issue 11 (46 hits)
#1 MeSH descriptor: [Alcohol‐Related Disorders] explode all trees #2 MeSH descriptor: [Alcohol Drinking] explode all trees #3 (alcohol and (abuse* or addict* or dependen* or disorder* or drink* or consumption or treatment)):ti,ab,kw #4 #1 or #2 or #3 #5 MeSH descriptor: [Baclofen] explode all trees #6 "baclofen" (Word variations have been searched) #7 Lioresal:ti,ab,kw (Word variations have been searched) #8 #5 or #6 or #7 #9 #4 and #8 with Publication Year from 2018 to present, in Trials
Appendix 3. MEDLINE (Ovid) search strategy
From January 2018 to 22 November (41 hits)
exp Alcohol‐Related Disorders/
((alcohol$ or drink$) adj5 (abstinen$ or abstain$ or abus$ or addict$ or crav$ or dependen$ or detox$ or disease$ or disorder$ or excessiv $ or heavy or intoxicat$ or misus$ or overdos$ or problem$ or rehab$ or relaps$ or treatment$ or withdraw$)).mp.
1 or 2
BACLOFEN.mp. or exp Baclofen/
Lioresal.mp.
4 or 5
3 and 6
randomized controlled trial.pt.
controlled clinical trial.pt.
random*.ab.
placebo.ab.
clinical trials as topic.sh.
random allocation.sh.
trial.ti.
8 or 9 or 10 or 11 or 12 or 13 or 14
exp animals/ not humans.sh.
15 not 16
7 and 17
limit 18 to yr="2018 ‐Current"
Appendix 4. Embase (Ovid) search strategy
From January 2018 to 22 November 2021 (78 hits)
exp alcoholism/
exp drinking behavior/
alcohol.mp.
(abuse* or addict* or dependen* or disorder* or drink* or consumption or treatment).ti,ab.
3 and 4
1 or 2 or 5
exp baclofen/ or baclofen.mp.
6 and 7
exp randomized controlled trial/
exp randomization/
exp double blind procedure/
exp single blind procedure/
random$.tw.
9 or 10 or 11 or 12 or 13
(animal or animal experiment).sh.
human.sh.
15 and 16
15 not 17
14 not 18
exp clinical trial/
(clin$ adj3 trial$).tw.
exp crossover procedure/
exp double blind procedure/
exp controlled clinical trial/
(placebo or assign* or allocat* or volunteer* or random* or factorial* or crossover).ti,ab.
((singl$ or doubl$ or trebl$ or tripl$) adj3 (blind$ or mask$)).tw.
20 or 21 or 22 or 23 or 24 or 25
27 not 18
19 or 28
8 and 29
Appendix 5. PsycINFO (Ovid)
from January 2018 to 22 November (82 hits)
exp Alcoholism/
(alcohol$ or drink$) adj5 (abstinen$ or abstain$ or abus$ or addict$ or crav$ or dependen$ or detox$ or disease$ or disorder$ or excessiv $ or heavy or intoxicat$ or misus$ or overdos$ or problem$ or rehab$ or relaps$ or treatment$ or withdraw$)).mp.
1 or 2
exp Baclofen/ or baclofen.mp.
3 and 4
limit 5 to yr="2018 ‐Current"
Appendix 6. Web of Science search strategy
From January 2018 to 22 November 2021 (62 hits)
TOPIC: (((alcohol$ or drink$) NEAR/5 (abstinen$ or abstain$ or abus$ or addict$ or crav$ or dependen$ or detox$ or disease$ or disorder$ or excessiv$ or heavy or intoxicat$ or misus$ or overdos$ or problem$ or rehab$ or relaps$ or treatment$ or withdraw$)))
TOPIC: (baclofen)
TOPIC: (randomi* OR randomly OR trial*)
#3 AND #2 AND #1
Appendix 7. CINAHL (EBSCOhost) search strategy
from January 2018 to 22 November 2021 (25 hits)
MH "Alcoholism"
TX (alcohol N3 (drink* or abus* or misus* or risk* or consum* or withdraw* or intoxicat* or detox* or treat* or therap* or excess* or reduc* or cessation or intervention))
TX(overdos* or intoxicat* or abstinen* or withdraw* or relaps*)
TX (drink* N3 (heavy or heavily or hazard* or binge or harmful))
MH "Clinical Trials+"
PT Clinical trial
TI clinic* N1 trial* or AB clinic* N1 trial*
TI ( singl* or doubl* or trebl* or tripl* ) and TI ( blind* or mask* )
AB ( singl* or doubl* or trebl* or tripl* ) and AB ( blind* or mask* )
TI randomi?ed control* trial* or AB randomi?ed control* trial*
MH "Random Assignment"
TI random* allocat* or AB random* allocat*
MH "Placebos"
TI placebo* or AB placebo*
MH "Quantitative Studies"
S5 OR S6 OR S7 OR S8 OR S9 OR S10 OR S11 OR S12 OR S13 OR S14 OR S15
baclofen
S1 OR S2 OR S3
S16 AND S17 AND S18
Appendix 8. Criteria for risk of bias assessment
| Item | Judgement | Description |
| 1. Random sequence generation (selection bias) | Low risk | The investigators described a random component in the sequence generation process such as: random number table; computer random number generator; coin tossing; shuffling cards or envelopes; throwing dice; drawing of lots; minimisation. |
| High risk | The investigators described a non‐random component in the sequence generation process such as: odd or even date of birth; date (or day) of admission; hospital or clinic record number; alternation; judgement of the clinician; results of a laboratory test or a series of tests; availability of the intervention. | |
| Unclear risk | Insufficient information about the sequence generation process to permit judgement of low or high risk. | |
| 2. Allocation concealment (selection bias) | Low risk | Investigators enrolling participants could not foresee assignment because 1 of the following, or an equivalent method, was used to conceal allocation: central allocation (including telephone, web‐based and pharmacy‐controlled randomisation); sequentially numbered drug containers of identical appearance; sequentially numbered, opaque, sealed envelopes. |
| High risk | Investigators enrolling participants could possibly foresee assignments because 1 of the following methods was used: open random allocation schedule (e.g. a list of random numbers); assignment envelopes without appropriate safeguards (e.g. if envelopes were unsealed or non‐opaque or not sequentially numbered); alternation or rotation; date of birth; case record number; any other explicitly unconcealed procedure. | |
| Unclear risk | Insufficient information to permit judgement of low or high risk. This is usually the case if the method of concealment was not described or not described in sufficient detail to allow a definite judgement. | |
| 3. Blinding of participants and providers (performance bias) Objective outcomes |
Low risk | No blinding or incomplete blinding, but the review authors judged that the outcome was unlikely to be influenced by lack of blinding. Blinding of participants and key study personnel ensured, and unlikely that the blinding could have been broken. |
| High risk | No blinding or incomplete blinding, and the outcome was likely to be influenced by lack of blinding. Blinding of key study participants and personnel attempted, but likely that the blinding could have been broken, and the outcome was likely to be influenced by lack of blinding. |
|
| Unclear risk | Insufficient information to permit judgement of low or high risk. | |
| 4. Blinding of participants and providers (performance bias) Subjective outcomes |
Low risk | Blinding of participants and providers ensured and unlikely that the blinding could have been broken. |
| High risk | No blinding or incomplete blinding, and the outcome was likely to be influenced by lack of blinding. Blinding of key study participants and personnel attempted, but likely that the blinding could have been broken, and the outcome was likely to be influenced by lack of blinding. |
|
| Unclear risk | Insufficient information to permit judgement of low or high risk. | |
| 5. Blinding of outcome assessor (detection bias) Objective outcomes |
Low risk | No blinding of outcome assessment, but the review authors judged that the outcome measurement was unlikely to be influenced by lack of blinding. Blinding of outcome assessment ensured, and unlikely that the blinding could have been broken. |
| High risk | No blinding of outcome assessment, and the outcome measurement was likely to be influenced by lack of blinding. Blinding of outcome assessment, but likely that the blinding could have been broken, and the outcome measurement was likely to be influenced by lack of blinding. |
|
| Unclear risk | Insufficient information to permit judgement of low or high risk. | |
| 6. Blinding of outcome assessor (detection bias) Subjective outcomes |
Low risk | Blinding of outcome assessment ensured, and unlikely that the blinding could have been broken. |
| High risk | No blinding of outcome assessment, and the outcome measurement was likely to be influenced by lack of blinding. Blinding of outcome assessment, but likely that the blinding could have been broken, and the outcome measurement was likely to be influenced by lack of blinding. |
|
| Unclear risk | Insufficient information to permit judgement of low or high risk. | |
| 7. Incomplete outcome data (attrition bias) For all outcomes except retention in treatment or dropout |
Low risk | No missing outcome data. Reasons for missing outcome data unlikely to be related to true outcome (for survival data, censoring unlikely to be introducing bias). Missing outcome data balanced in numbers across intervention groups, with similar reasons for missing data across groups. For dichotomous outcome data, the proportion of missing outcomes compared with observed event risk not enough to have a clinically relevant impact on the intervention effect estimate. For continuous outcome data, plausible effect size (mean differences or standardised mean differences) among missing outcomes not enough to have a clinically relevant impact on observed effect size. Missing data had been imputed using appropriate methods. All randomised participants were reported/analysed in the group they were allocated to by randomisation irrespective of non‐compliance and co‐interventions (intention to treat). |
| High risk | Reason for missing outcome data likely to be related to true outcome, with either imbalance in numbers or reasons for missing data across intervention groups. For dichotomous outcome data, the proportion of missing outcomes compared with observed event risk enough to induce clinically relevant bias in intervention effect estimate. For continuous outcome data, plausible effect size (difference in means or standardised difference in means) among missing outcomes enough to induce clinically relevant bias in observed effect size. 'As‐treated' analysis done with substantial departure of the intervention received from that assigned at randomisation. |
|
| Unclear risk | Insufficient information to permit judgement of low or high risk (e.g. number randomised not stated, no reasons for missing data provided; number of dropouts not reported for each group). | |
| 8. Selective reporting (reporting bias) | Low risk | The study protocol was available and all the study's prespecified (primary and secondary) outcomes that were of interest in the review were reported in the prespecified way. The study protocol was unavailable but it was clear that the published reports included all expected outcomes, including those that were prespecified (convincing text of this nature may be uncommon). |
| High risk | Not all the study's prespecified primary outcomes were reported. ≥ 1 primary outcomes was reported using measurements, analysis methods or subsets of the data (e.g. subscales) that were not prespecified. ≥ 1 reported primary outcomes were not prespecified (unless clear justification for their reporting was provided, such as an unexpected adverse effect). ≥ 1 outcomes of interest in the review were reported incompletely so that they could not be entered in a meta‐analysis. The study report failed to include results for a key outcome that would be expected to have been reported for such a study. |
|
| Unclear risk | Insufficient information to permit judgement of low or high risk. |
Appendix 9. Trials registers
ClinicalTrials.gov
22 November 2021
3 new records from January 2018 for: baclofen | Interventional Studies | alcohol (www.clinicaltrials.gov)
World Health Organization (WHO) International Clinical Trials Registry Platform (ICTRP)
22 November 2021
3 new records from January 2018 for: baclofen AND alcohol (apps.who.int/trialsearch/)
Data and analyses
Comparison 1. Baclofen versus placebo (all studies).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1.1 Relapse: return to any drinking at end of treatment | 12 | 1057 | Risk Ratio (M‐H, Random, 95% CI) | 0.87 [0.77, 0.99] |
| 1.2 Frequency of use: % days abstinence at end of treatment | 16 | 1273 | Mean Difference (IV, Random, 95% CI) | 9.07 [3.30, 14.85] |
| 1.3 Frequency of use: heavy drinking days at end of treatment | 13 | 840 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.18 [‐0.48, 0.11] |
| 1.4 Amount of use: drinks per drinking days at end of treatment | 9 | 392 | Mean Difference (IV, Random, 95% CI) | ‐0.45 [‐1.20, 0.30] |
| 1.5 Adverse events: number of participants with at least one adverse event at end of treatment | 10 | 738 | Risk Ratio (M‐H, Random, 95% CI) | 1.05 [0.99, 1.11] |
| 1.6 Dropouts at end of treatment | 17 | 1563 | Risk Ratio (M‐H, Random, 95% CI) | 0.88 [0.74, 1.03] |
| 1.7 Dropouts due to adverse events | 16 | 1499 | Risk Ratio (M‐H, Random, 95% CI) | 1.39 [0.89, 2.18] |
| 1.8 Craving | 17 | 1275 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.16 [‐0.37, 0.04] |
| 1.9 Anxiety | 15 | 1127 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.01 [‐0.14, 0.11] |
| 1.10 Depression | 12 | 1029 | Std. Mean Difference (IV, Random, 95% CI) | 0.07 [‐0.12, 0.27] |
| 1.11 Adverse events: fatigue, tiredness | 13 | 1311 | Risk Ratio (M‐H, Random, 95% CI) | 1.31 [1.09, 1.59] |
| 1.12 Adverse events: insomnia | 11 | 1100 | Risk Ratio (M‐H, Random, 95% CI) | 0.92 [0.62, 1.35] |
| 1.13 Adverse events: pain (diverse) | 3 | 98 | Risk Ratio (M‐H, Random, 95% CI) | 0.51 [0.19, 1.33] |
| 1.14 Adverse events: vertigo, dizziness | 15 | 1415 | Risk Ratio (M‐H, Random, 95% CI) | 1.70 [1.35, 2.13] |
| 1.15 Adverse events: constipation | 11 | 1124 | Risk Ratio (M‐H, Random, 95% CI) | 0.95 [0.61, 1.47] |
| 1.16 Adverse events: somnolence, sleepiness, drowsiness or sedation | 17 | 1543 | Risk Ratio (M‐H, Random, 95% CI) | 1.36 [1.19, 1.56] |
| 1.17 Adverse events: muscle pain | 5 | 594 | Risk Ratio (M‐H, Random, 95% CI) | 0.93 [0.38, 2.25] |
| 1.18 Adverse events: dry mouth | 8 | 933 | Risk Ratio (M‐H, Random, 95% CI) | 1.79 [1.01, 3.18] |
| 1.19 Adverse events: nausea | 10 | 1060 | Risk Ratio (M‐H, Random, 95% CI) | 1.17 [0.88, 1.54] |
| 1.20 Adverse events: skin rash | 7 | 446 | Risk Ratio (M‐H, Random, 95% CI) | 0.80 [0.33, 1.93] |
| 1.21 Adverse events: headaches | 13 | 1304 | Risk Ratio (M‐H, Random, 95% CI) | 1.31 [1.04, 1.64] |
| 1.22 Adverse events: paraesthesia/numbness | 6 | 914 | Risk Ratio (M‐H, Random, 95% CI) | 2.87 [1.17, 7.06] |
| 1.23 Adverse events: diarrhoea | 3 | 816 | Risk Ratio (M‐H, Random, 95% CI) | 0.64 [0.40, 1.02] |
| 1.24 Adverse events: tinnitus | 2 | 496 | Risk Ratio (M‐H, Random, 95% CI) | 1.77 [0.16, 20.16] |
| 1.25 Adverse events: muscle spasm/rigidity | 3 | 552 | Risk Ratio (M‐H, Random, 95% CI) | 1.94 [1.07, 3.52] |
| 1.26 Adverse events: hyperhidrosis | 3 | 816 | Risk Ratio (M‐H, Random, 95% CI) | 1.50 [0.89, 2.51] |
| 1.27 Adverse events: nasopharyngitis | 4 | 872 | Risk Ratio (M‐H, Random, 95% CI) | 0.89 [0.40, 1.95] |
| 1.28 Adverse events: decrease appetite/anorexia | 3 | 816 | Risk Ratio (M‐H, Random, 95% CI) | 0.74 [0.31, 1.75] |
| 1.29 Adverse events: dysgeusia/ageusia | 3 | 816 | Risk Ratio (M‐H, Random, 95% CI) | 1.73 [0.57, 5.24] |
| 1.30 Adverse events: tremor | 3 | 816 | Risk Ratio (M‐H, Random, 95% CI) | 0.69 [0.42, 1.15] |
| 1.31 Adverse events: weakness | 3 | 552 | Risk Ratio (M‐H, Random, 95% CI) | 1.19 [0.70, 2.01] |
| 1.32 Adverse events: vomiting | 3 | 816 | Risk Ratio (M‐H, Random, 95% CI) | 1.07 [0.66, 1.71] |
| 1.33 Adverse events: urinary frequency | 4 | 340 | Risk Ratio (M‐H, Random, 95% CI) | 1.10 [0.45, 2.68] |
| 1.34 Adverse events: shortness of breath | 2 | 104 | Risk Ratio (M‐H, Random, 95% CI) | 0.89 [0.04, 22.31] |
Comparison 2. Baclofen versus placebo (divided into low, medium and high doses of baclofen).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 2.1 Relapse: return to any drinking at end of treatment | 12 | 1057 | Risk Ratio (M‐H, Random, 95% CI) | 0.87 [0.77, 0.99] |
| 2.1.1 Low doses | 6 | 463 | Risk Ratio (M‐H, Random, 95% CI) | 0.82 [0.64, 1.04] |
| 2.1.2 Medium doses | 3 | 129 | Risk Ratio (M‐H, Random, 95% CI) | 0.73 [0.37, 1.45] |
| 2.1.3 High doses | 3 | 465 | Risk Ratio (M‐H, Random, 95% CI) | 0.90 [0.71, 1.15] |
| 2.2 Frequency of use: % days abstinence at end of treatment | 16 | 1273 | Mean Difference (IV, Random, 95% CI) | 9.07 [3.30, 14.85] |
| 2.2.1 Low doses | 8 | 583 | Mean Difference (IV, Random, 95% CI) | 10.59 [0.77, 20.41] |
| 2.2.2 Medium doses | 5 | 225 | Mean Difference (IV, Random, 95% CI) | 7.14 [‐3.10, 17.38] |
| 2.2.3 High doses | 3 | 465 | Mean Difference (IV, Random, 95% CI) | 11.09 [4.39, 17.80] |
| 2.3 Frequency of use: % of heavy drinking days at end of treatment | 13 | 840 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.18 [‐0.48, 0.11] |
| 2.3.1 Low doses | 6 | 278 | Std. Mean Difference (IV, Random, 95% CI) | 0.10 [‐0.15, 0.34] |
| 2.3.2 Medium doses | 6 | 242 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.47 [‐1.15, 0.20] |
| 2.3.3 High doses | 1 | 320 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.21 [‐0.43, 0.01] |
| 2.4 Amount of use: drinks per drinking days at end of treatment | 9 | 392 | Mean Difference (IV, Random, 95% CI) | ‐0.45 [‐1.20, 0.30] |
| 2.4.1 Low doses | 5 | 242 | Mean Difference (IV, Random, 95% CI) | ‐0.06 [‐1.33, 1.22] |
| 2.4.2 Medium doses | 4 | 150 | Mean Difference (IV, Random, 95% CI) | ‐0.64 [‐1.95, 0.68] |
| 2.5 Adverse events: number of participants with at least one adverse event at end of treatment | 10 | 738 | Risk Ratio (M‐H, Random, 95% CI) | 1.05 [0.99, 1.11] |
| 2.5.1 Low doses | 5 | 260 | Risk Ratio (M‐H, Random, 95% CI) | 1.23 [0.92, 1.64] |
| 2.5.2 Medium doses | 4 | 162 | Risk Ratio (M‐H, Random, 95% CI) | 0.90 [0.63, 1.28] |
| 2.5.3 High doses | 1 | 316 | Risk Ratio (M‐H, Random, 95% CI) | 1.05 [0.99, 1.11] |
| 2.6 Dropouts at end of treatment | 17 | 1563 | Risk Ratio (M‐H, Random, 95% CI) | 0.88 [0.74, 1.03] |
| 2.6.1 Low doses | 8 | 564 | Risk Ratio (M‐H, Random, 95% CI) | 0.96 [0.70, 1.32] |
| 2.6.2 Medium doses | 5 | 214 | Risk Ratio (M‐H, Random, 95% CI) | 1.03 [0.65, 1.61] |
| 2.6.3 High doses | 4 | 785 | Risk Ratio (M‐H, Random, 95% CI) | 0.76 [0.67, 0.87] |
| 2.7 Dropouts due to adverse events | 16 | 1499 | Risk Ratio (M‐H, Random, 95% CI) | 1.39 [0.89, 2.18] |
| 2.7.1 Low doses | 8 | 564 | Risk Ratio (M‐H, Random, 95% CI) | 1.81 [0.61, 5.32] |
| 2.7.2 Medium doses | 4 | 150 | Risk Ratio (M‐H, Random, 95% CI) | 6.11 [0.82, 45.25] |
| 2.7.3 High doses | 4 | 785 | Risk Ratio (M‐H, Random, 95% CI) | 1.21 [0.68, 2.13] |
Comparison 3. Baclofen versus placebo (divided into 12‐week and longer than 12‐week studies).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 3.1 Relapse: return to any drinking at end of treatment | 12 | 1057 | Risk Ratio (M‐H, Random, 95% CI) | 0.87 [0.77, 0.99] |
| 3.1.1 12‐week studies | 7 | 466 | Risk Ratio (M‐H, Random, 95% CI) | 0.63 [0.40, 1.00] |
| 3.1.2 Longer than 12‐week studies | 5 | 591 | Risk Ratio (M‐H, Random, 95% CI) | 0.98 [0.93, 1.03] |
| 3.2 Frequency of use: % days abstinence at end of treatment | 16 | 1273 | Mean Difference (IV, Random, 95% CI) | 9.07 [3.30, 14.85] |
| 3.2.1 12‐week studies | 11 | 682 | Mean Difference (IV, Random, 95% CI) | 10.90 [3.17, 18.62] |
| 3.2.2 Longer than 12‐week studies | 5 | 591 | Mean Difference (IV, Random, 95% CI) | 8.05 [1.09, 15.01] |
| 3.3 Frequency of use: % of heavy drinking days at end of treatment | 13 | 840 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.18 [‐0.48, 0.11] |
| 3.3.1 12‐week studies | 10 | 400 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.07 [‐0.27, 0.13] |
| 3.3.2 Longer than 12‐week studies | 3 | 440 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.54 [‐1.68, 0.60] |
| 3.4 Amount of use: drink per drinking days at end of treatment | 9 | 392 | Mean Difference (IV, Random, 95% CI) | ‐0.45 [‐1.20, 0.30] |
| 3.4.1 12‐week studies | 7 | 272 | Mean Difference (IV, Random, 95% CI) | ‐0.36 [‐1.29, 0.57] |
| 3.4.2 Longer than 12‐week studies | 2 | 120 | Mean Difference (IV, Random, 95% CI) | ‐0.49 [‐2.31, 1.32] |
| 3.5 Adverse events: number of participants with at least one adverse event at end of treatment | 10 | 738 | Risk Ratio (M‐H, Random, 95% CI) | 1.05 [0.99, 1.11] |
| 3.5.1 12‐week studies | 7 | 302 | Risk Ratio (M‐H, Random, 95% CI) | 0.99 [0.70, 1.39] |
| 3.5.2 Longer than 12‐week studies | 3 | 436 | Risk Ratio (M‐H, Random, 95% CI) | 1.05 [1.00, 1.11] |
| 3.6 Dropouts at end of treatment | 17 | 1563 | Risk Ratio (M‐H, Random, 95% CI) | 0.88 [0.74, 1.03] |
| 3.6.1 12‐week studies | 11 | 652 | Risk Ratio (M‐H, Random, 95% CI) | 0.98 [0.73, 1.31] |
| 3.6.2 Longer than 12‐week studies | 6 | 911 | Risk Ratio (M‐H, Random, 95% CI) | 0.78 [0.69, 0.88] |
| 3.7 Dropout due to adverse events | 16 | 1499 | Risk Ratio (M‐H, Random, 95% CI) | 1.39 [0.89, 2.18] |
| 3.7.1 12‐week studies | 10 | 588 | Risk Ratio (M‐H, Random, 95% CI) | 3.00 [0.93, 9.66] |
| 3.7.2 Longer than 12‐week studies | 6 | 911 | Risk Ratio (M‐H, Random, 95% CI) | 1.22 [0.76, 1.98] |
Comparison 4. Baclofen versus placebo (divided into detoxified and non‐detoxified participants).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 4.1 Relapse: return to any drinking at end of treatment | 12 | 1057 | Risk Ratio (M‐H, Random, 95% CI) | 0.87 [0.77, 0.99] |
| 4.1.1 Detoxified participants | 9 | 757 | Risk Ratio (M‐H, Random, 95% CI) | 0.73 [0.55, 0.95] |
| 4.1.2 Non‐detoxified participants | 3 | 300 | Risk Ratio (M‐H, Random, 95% CI) | 1.00 [0.94, 1.06] |
| 4.2 Frequency of use: % days abstinence at end of treatment | 16 | 1273 | Mean Difference (IV, Random, 95% CI) | 9.07 [3.30, 14.85] |
| 4.2.1 Detoxified participants | 10 | 549 | Mean Difference (IV, Random, 95% CI) | 11.76 [3.22, 20.29] |
| 4.2.2 Non‐detoxified participants | 6 | 724 | Mean Difference (IV, Random, 95% CI) | 6.03 [‐1.59, 13.64] |
| 4.3 Frequency of use: % of heavy drinking days at end of treatment | 13 | 840 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.18 [‐0.48, 0.11] |
| 4.3.1 Detoxified participants | 8 | 296 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.08 [‐0.32, 0.16] |
| 4.3.2 Non‐detoxified participants | 5 | 544 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.34 [‐0.98, 0.30] |
| 4.4 Amount of use: drink per drinking days at end of treatment | 9 | 392 | Mean Difference (IV, Random, 95% CI) | ‐0.45 [‐1.20, 0.30] |
| 4.4.1 Detoxified participants | 7 | 272 | Mean Difference (IV, Random, 95% CI) | ‐0.36 [‐1.29, 0.57] |
| 4.4.2 Non‐detoxified participants | 2 | 120 | Mean Difference (IV, Random, 95% CI) | ‐0.49 [‐2.31, 1.32] |
| 4.5 Adverse events: number of participants with at least one adverse event at end of treatment | 10 | 738 | Risk Ratio (M‐H, Random, 95% CI) | 1.05 [0.99, 1.11] |
| 4.5.1 Detoxified participants | 7 | 578 | Risk Ratio (M‐H, Random, 95% CI) | 1.05 [0.99, 1.11] |
| 4.5.2 Non‐detoxified participants | 3 | 160 | Risk Ratio (M‐H, Random, 95% CI) | 1.11 [0.84, 1.45] |
| 4.6 Dropouts at end of treatment | 17 | 1563 | Risk Ratio (M‐H, Random, 95% CI) | 0.88 [0.74, 1.03] |
| 4.6.1 Detoxified participants | 12 | 879 | Risk Ratio (M‐H, Random, 95% CI) | 0.87 [0.71, 1.07] |
| 4.6.2 No‐detoxified participants | 5 | 684 | Risk Ratio (M‐H, Random, 95% CI) | 0.93 [0.69, 1.28] |
| 4.7 Dropouts due to adverse events | 16 | 1499 | Risk Ratio (M‐H, Random, 95% CI) | 1.39 [0.89, 2.18] |
| 4.7.1 Detoxified participants | 12 | 879 | Risk Ratio (M‐H, Random, 95% CI) | 1.08 [0.59, 1.98] |
| 4.7.2 No‐detoxified participants | 4 | 620 | Risk Ratio (M‐H, Random, 95% CI) | 1.87 [0.97, 3.61] |
Comparison 5. Baclofen versus acamprosate.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 5.1 Relapse: return to any drinking at end of treatment | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 1.25 [0.71, 2.20] |
| 5.2 Adverse events: number of participants with at least one adverse event at end of treatment | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.62 [0.23, 1.69] |
| 5.3 Dropouts at end of treatment | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.56 [0.21, 1.46] |
| 5.4 Dropouts due to adverse events | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.33 [0.01, 7.87] |
| 5.5 Craving | 2 | 109 | Mean Difference (IV, Random, 95% CI) | 5.80 [‐11.84, 23.44] |
| 5.6 Adverse events: fatigue, tiredness | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 3.00 [0.13, 70.83] |
| 5.7 Adverse events: vertigo, dizziness | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 2.00 [0.19, 20.90] |
| 5.8 Adverse events: nausea | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.33 [0.01, 7.87] |
| 5.9 Adverse events: skin rash | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.20 [0.01, 4.00] |
| 5.10 Adverse events: decrease appetite/anorexia | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 1.00 [0.15, 6.64] |
| 5.11 Adverse events: acidity | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.33 [0.01, 7.87] |
| 5.12 Adverse events: palpitations | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.33 [0.01, 7.87] |
Comparison 6. Baclofen versus naltrexone.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 6.1 Relapse: return to any drinking at end of treatment | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 2.50 [1.12, 5.56] |
| 6.2 Adverse events: number of participants with at least one adverse event at end of treatment | 2 | 80 | Risk Ratio (M‐H, Random, 95% CI) | 0.35 [0.15, 0.80] |
| 6.3 Dropouts at end of treatment | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 1.00 [0.32, 3.10] |
| 6.4 Craving | 1 | 60 | Mean Difference (IV, Random, 95% CI) | 2.08 [‐3.71, 7.87] |
| 6.5 Adverse events: fatigue, tiredness | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 3.00 [0.13, 70.83] |
| 6.6 Adverse events: insomnia | 1 | 20 | Risk Ratio (M‐H, Random, 95% CI) | 0.14 [0.01, 2.45] |
| 6.7 Adverse events: vertigo, dizziness | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.40 [0.08, 1.90] |
| 6.8 Adverse events: somnolence, sleepiness, drowsiness or sedation | 1 | 20 | Risk Ratio (M‐H, Random, 95% CI) | 0.33 [0.04, 2.69] |
| 6.9 Adverse events: nausea | 2 | 80 | Risk Ratio (M‐H, Random, 95% CI) | 0.26 [0.03, 2.20] |
| 6.10 Adverse events: decrease appetite/anorexia | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 2.00 [0.19, 20.90] |
| 6.11 Adverse events: tremor | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.33 [0.01, 7.87] |
| 6.12 Adverse events: acidity | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.20 [0.01, 4.00] |
| 6.13 Adverse events: erectile dysfunction | 1 | 60 | Risk Ratio (M‐H, Random, 95% CI) | 0.33 [0.01, 7.87] |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Addolorato 2007.
| Study characteristics | ||
| Methods |
Study design: RCT Study grouping: parallel group Country: Italy Setting: Institute of Internal Medicine of the Catholic University in Rome, Italy (which has both a liver unit and an alcohol addiction unit) |
|
| Participants |
Baseline characteristics Baclofen
Placebo
Inclusion criteria: aged 18–75 years; diagnosis of alcohol dependence according to DSM‐IV criteria; diagnosis of liver cirrhosis; an alcohol intake of ≥ 2 HDD/week on average (men ≥ 5 drinks/day; women ≥ 4 drinks/day) and an average overall consumption of ≥ 21 drinks/week for men and ≥ 14 drinks/week for women during the 4 weeks before enrolment (1 standard drink = 12 g absolute alcohol); and presence of a referred family member able to assist with drug administration and monitoring. Exclusion criteria: severe heart or lung disease; abnormal renal function, hepatorenal syndrome, or both; malignant disease; metabolic diseases; hepatic encephalopathy; treatment with interferon or corticosteroids within the past 60 days; psychopathological illness treated with psychoactive drugs; epilepsy; and addiction to drugs other than nicotine. Detoxification: participants had an alcohol intake of ≥ 2 HDD/week on average (men ≥ 5 drinks/day; women ≥ 4 drinks/day) and an average overall consumption of 21 drinks/week or more for men and 14 drinks/week or more for women during the 4 weeks before enrolment (1 standard drink = 12 g absolute alcohol). |
|
| Interventions | Baclofen + counselling (42 participants)
Placebo + counselling (42 participants)
|
|
| Outcomes |
Primary outcomes
Secondary outcomes
|
|
| Identification |
Sponsorship source: Italian Ministry for University, Scientific and Technological Research (MURST), and by the European Research Advisory Board (ERAB) Author's name: Dr Giovanni Addolorato Institution: Institute of Internal Medicine, Catholic University of Rome Email: giovanni.addolorato@.unicatt.it Address: Largo A Gemelli 8, I‐00168 Rome, Italy |
|
| Notes | During the updating, the authors allowed us to use the original database to collect the lacking data. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Quote: "Randomisation was balanced with blocks." Quote: "eligible patients who provided informed consent were randomly allocated either oral baclofen or placebo." |
| Allocation concealment (selection bias) | Low risk | Quote: "were randomly allocated either oral baclofen or placebo. The randomisation sequence was generated by the pharmacist who prepared drug and placebo. Randomisation was balanced with blocks. The pharmacist did not have any further role in the study." |
| Blinding of participants and personnel (performance bias) Objective outcomes | Low risk | Quote: "Participants and investigators were unaware of treatment assignment. To maintain masking, the randomisation code was concealed in a safe box in the pharmacy. For the duration of the study (including also the 4 weeks of follow‐up), the pharmacist or another employee of the same pharmacy could be contacted at any time to open the safe box in the case of a specific emergency." Quote: "Placebo tablets were identical in size, colour, shape, and taste to baclofen." |
| Blinding of participants and personnel (performance bias) Subjective outcomes | Low risk | Quote: "Participants and investigators were unaware of treatment assignment. To maintain masking, the randomisation code was concealed in a safe box in the pharmacy. For the duration of the study (including also the 4 weeks of follow‐up), the pharmacist or another employee of the same pharmacy could be contacted at any time to open the safe box in the case of a specific emergency." Quote: "Placebo tablets were identical in size, colour, shape, and taste to baclofen." |
| Blinding of outcome assessment (detection bias) Objective outcomes | Low risk | Quote: "Participants and investigators were unaware of treatment assignment. To maintain masking, the randomisation code was concealed in a safe box in the pharmacy. For the duration of the study (including also the 4 weeks of follow‐up), the pharmacist or another employee of the same pharmacy could be contacted at any time to open the safe box in the case of a specific emergency." |
| Blinding of outcome assessment (detection bias) Subjective outcomes | Low risk | Quote: "Participants and investigators were unaware of treatment assignment. To maintain masking, the randomisation code was concealed in a safe box in the pharmacy. For the duration of the study (including also the 4 weeks of follow‐up), the pharmacist or another employee of the same pharmacy could be contacted at any time to open the safe box in the case of a specific emergency." |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Quote: "We assumed for this analysis that all patients who terminated treatment before the end of the study had relapsed." Quote: "Our analysis was done by intention to treat, insomuch that we assumed every patient took the drug allocated to them and counted them in that group." |
| Selective reporting (reporting bias) | Low risk | Study protocol available; results reported for all primary and secondary outcomes described in protocol. |
Addolorato 2011‐LD.
| Study characteristics | ||
| Methods |
Study design: 12‐week, 3‐arm parallel, double‐blind, RCT Country: Italy |
|
| Participants |
Baseline characteristics Baclofen: low dose (14 participants)
Placebo (7 participants)
Inclusion criteria: aged 18–60 years (inclusive); diagnosis of alcohol dependence according to DSM IV‐TR; alcohol intake ≥ 2 HDD (≥ 5 drinks/day for men and ≥ 4 drinks/day for women) per week on average and a mean overall consumption ≥ 21 drinks/week for men and ≥ 14 drinks/week for women in the 4 weeks before enrolment. Exclusion criteria: clinically significant medical and psychiatric disease that might interfere with the evaluation of the study medication or that might represent a safety concern; concurrent use of psychotropic medication, including antidepressant, mood stabilisers, antipsychotics, anxiolytics or hypnotics; concurrent use of anticonvulsants, insulin or oral hypoglycaemics. Detoxification: participants had to refrain from drinking for ≥ 3 days prior to randomisation day; participants abstinent from alcohol for > 10 days prior to randomisation were excluded. |
|
| Interventions | Baclofen: low dose + medical management
Placebo (7 participants) + medical management
|
|
| Outcomes |
Primary outcomes
Secondary outcomes
|
|
| Identification | EudraCT Number: 2006‐000713‐37 | |
| Notes | In this review update, we divided the study into the 2 following arms:
|
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐generated randomisation list produced by the pharmacist who prepared the drug and the placebo. |
| Allocation concealment (selection bias) | Low risk | Quote: "To maintain masking, an independent colleague, who did not have any further role in the study, concealed the randomization codes in a safe box." |
| Blinding of participants and personnel (performance bias) Objective outcomes | Low risk | Quote: "Participants and investigators were unaware of treatment assignment. Placebo and baclofen were prepared by a local compounding pharmacy. Placebo, baclofen 10 mg and baclofen 20 mg tablets were identical in size, colour, shape and taste." |
| Blinding of participants and personnel (performance bias) Subjective outcomes | Low risk | Quote: "Participants and investigators were unaware of treatment assignment. Placebo and baclofen were prepared by a local compounding pharmacy. Placebo, baclofen 10 mg and baclofen 20 mg tablets were identical in size, colour, shape and taste." |
| Blinding of outcome assessment (detection bias) Objective outcomes | Low risk | No information. However, objective outcomes are unlikely to be biased by lack of blinding. |
| Blinding of outcome assessment (detection bias) Subjective outcomes | Low risk | Quote: "Participants were blinded to treatment assignment." |
| Incomplete outcome data (attrition bias) All outcomes | High risk | 24% lost at follow‐up. Unbalanced between groups. |
| Selective reporting (reporting bias) | High risk | Study protocol unavailable. The authors declared that (quote) "they reported the results of a secondary analysis, which was also conducted only on the Italian sub sample of the original multicentre study. The planned outcome was based on the reduction of heavy drinking, this post hoc analysis used the number of drinks per day." |
Addolorato 2011‐MD.
| Study characteristics | ||
| Methods |
Study design: 12‐week, 3‐arm parallel, double‐blind, RCT Country: Italy |
|
| Participants |
Baseline characteristics Baclofen: medium dose (14 participants)
Placebo (7 participants)
Inclusion criteria: aged 18–60 years (inclusive); diagnosis of alcohol dependence according to DSM IV‐TR; alcohol intake ≥ 2 HDD (≥ 5 drinks/day for men and ≥ 4 drinks/day for women) per week on average and a mean overall consumption ≥ 21 drinks/week for men and ≥ 14 drinks/week for women in the 4 weeks before enrolment. Exclusion criteria: clinically significant medical and psychiatric disease that might interfere with the evaluation of the study medication or that might represent a safety concern; concurrent use of psychotropic medication, including antidepressant, mood stabilisers, antipsychotics, anxiolytics or hypnotics; concurrent use of anticonvulsants, insulin or oral hypoglycaemics. Detoxification: participants had to refrain from drinking for ≥ 3 days prior to randomisation day; participants abstinent from alcohol for > 10 days prior to randomisation were excluded. |
|
| Interventions | Baclofen medium dose (14 participants) + medical management
Placebo (14 participants) + medical management
|
|
| Outcomes |
Primary outcomes
Secondary outcomes
|
|
| Identification | EudraCT Number: 2006‐000713‐37 | |
| Notes | In this review update, we divided the study into the 2 following arms:
|
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐generated randomisation list produced by the pharmacist who prepared the drug and the placebo. |
| Allocation concealment (selection bias) | Low risk | Quote: "To maintain masking, an independent colleague, who did not have any further role in the study, concealed the randomization codes in a safe box." |
| Blinding of participants and personnel (performance bias) Objective outcomes | Low risk | Quote: "Participants and investigators were unaware of treatment assignment. Placebo and baclofen were prepared by a local compounding pharmacy. Placebo, baclofen 10 mg and baclofen 20 mg tablets were identical in size, colour, shape and taste." |
| Blinding of participants and personnel (performance bias) Subjective outcomes | Low risk | Quote: "Participants and investigators were unaware of treatment assignment. Placebo and baclofen were prepared by a local compounding pharmacy. Placebo, baclofen 10 mg and baclofen 20 mg tablets were identical in size, colour, shape and taste." |
| Blinding of outcome assessment (detection bias) Objective outcomes | Low risk | No information. However, objective outcomes are unlikely to be biased by lack of blinding. |
| Blinding of outcome assessment (detection bias) Subjective outcomes | Low risk | Quote: "Participants were blinded to treatment assignment." |
| Incomplete outcome data (attrition bias) All outcomes | High risk | 24% lost at follow‐up. Unbalanced between groups. |
| Selective reporting (reporting bias) | High risk | Study protocol unavailable. The authors declared that (quote) "they reported the results of a secondary analysis, which was also conducted only on the Italian sub sample of the original multicentre study. The planned outcome was based on the reduction of heavy drinking, this post hoc analysis used the number of drinks per day." |
Beraha 2016‐HD.
| Study characteristics | ||
| Methods |
Study design: RCT Study grouping: parallel group Country: the Netherlands Setting: SolutionS Center, Voorthuizen, the Netherlands; U‐Center, Epen, The Netherlands; The Home Clinic, Weesp, the Netherlands; Roder Sana Addiction Treatment, Oirschot, the Netherlands |
|
| Participants |
Baseline characteristics Baclofen: high dose (58 participants)
Placebo (31 participants)
Inclusion criteria: aged 18−70 years; DSM‐IV AD‐diagnosis; breath alcohol concentration < 0.5% at the screening visit (informed consent); mean alcohol consumption ≥ 14 units for women and ≥ 21 units for men/week over consecutive 30 days in the 90‐day period before start of study and ≥ 2 HDDs (women ≥ 5 units; men ≥ 6 units) in past 90 days; minimum of 96 hours and maximum of 21 days of abstinence prior to start of study medication; sufficient Dutch language skills; provision of a contact person in the event of loss of contact. Exclusion criteria: current severe axis I disorder (other than depression, anxiety and bipolar disorder); any primary diagnosis of substance dependence other than alcohol dependence (nicotine dependence was allowed); severe physical illnesses (e.g. Parkinson's disease, gastric ulcer, duodenal ulcer, cerebrovascular disease, respiratory insufficiency, hepatic or renal insufficiency, epilepsy); antihypertensive medication; risk of suicide; cognitive impairment interfering with the understanding of the study; current or recent (3 months before start of study), pharmacological treatment for alcohol dependence (i.e. acamprosate, naltrexone, disulfiram, ortopiramate); pregnancy or breastfeeding; > 7 days inpatient treatment for SUD in the 30 days before the start of the study; and use of baclofen in the past 30 days. Detoxification: participants followed 4–6 weeks' inpatient detoxification. |
|
| Interventions | Baclofen: high dose (58 participants) + CBT
Placebo (31 participants) + CBT
|
|
| Outcomes |
Primary outcomes
Secondary outcomes
|
|
| Identification |
Sponsorship source: funding by private donation through the University of Amsterdam Fund (AUF7344) Author's name: Dr Esther M Beraha Institution: Addiction Development and Psychopathology (ADAPT) Laboratory, Department of Psychology, University of Amsterdam, Amsterdam, the Netherlands Email: R.W.H.J.Wiers@uva.nl (RW Wiers) Address: Addiction Development and Psychopathology Lab, Department of Psychology, University of Amsterdam, Nieuwe Achtergracht 129 B, 1018 WS Amsterdam, the Netherlands |
|
| Notes | In this review update, we divided the study into the 2 following arms:
|
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Randomization (blocks of 6; pre‐stratification by gender and centre) was conducted by the Clinical Research Unit of the AMC and patients were assigned to one of the three groups via an electronic database after baseline assessment." |
| Allocation concealment (selection bias) | Low risk | Quote: "Only the study pharmacist had access to the randomization list and had no further role in the trial." |
| Blinding of participants and personnel (performance bias) Objective outcomes | Low risk | Stated as double‐blind; quote: "The study medication was manufactured, packaged, and labelled by Tiofarma and stored and provided by the pharmacy of the AMC. Baclofen and placebo were provided in identical 10 mg tablets and supplied in containers with 24, 42, 63, 147, or 168 tablets." |
| Blinding of participants and personnel (performance bias) Subjective outcomes | Low risk | Stated as double‐blind; quote: "The study medication was manufactured, packaged, and labelled by Tiofarma and stored and provided by the pharmacy of the AMC. Baclofen and placebo were provided in identical 10 mg tablets and supplied in containers with 24, 42, 63, 147, or 168 tablets." |
| Blinding of outcome assessment (detection bias) Objective outcomes | Low risk | Quote: "The investigators and responsible physicians remained blind with regard to the study medication during the whole study period." |
| Blinding of outcome assessment (detection bias) Subjective outcomes | Low risk | Quote: "The investigators and responsible physicians remained blind with regard to the study medication during the whole study period." |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Quote: "Analyses were done by intention to treat, counting pills every visit and assuming that patients took the drugs they were given." Comment: only 3 participants were lost at follow‐up in the baclofen groups (3%) (Beraha 2016‐HD: 1 participant lost; Beraha 2016‐LD: 2 participants lost). |
| Selective reporting (reporting bias) | Low risk | Study protocol available; results reported for all primary and secondary outcomes reported in protocol. |
Beraha 2016‐LD.
| Study characteristics | ||
| Methods |
Study design: RCT Study grouping: parallel group Country: the Netherlands Setting: SolutionS Center, Voorthuizen, the Netherlands; U‐Center, Epen, The Netherlands; The Home Clinic, Weesp, the Netherlands; Roder Sana Addiction Treatment, Oirschot, the Netherlands |
|
| Participants |
Baseline characteristics Baclofen: low dose (31 participants)
Placebo (31 participants)
Inclusion criteria: aged 18−70 years; DSM‐IV AD‐diagnosis; breath alcohol concentration < 0.5% at screening visit (informed consent); mean alcohol consumption ≥ 14 units for women and ≥ 21 units for men/week over consecutive 30 days in 90‐day period before start of study and ≥ 2 HDDs (women ≥ 5 units; men ≥ 6 units) in past 90 days; minimum of 96 hours and maximum of 21 days of abstinence prior to start of study medication; sufficient Dutch language skills; provision of a contact person in the event of loss of contact. Exclusion criteria: current severe axis I disorder (other than depression, anxiety and bipolar disorder); any primary diagnosis of substance dependence other than alcohol dependence (nicotine dependence was allowed); severe physical illnesses (e.g. Parkinson's disease, gastric ulcer, duodenal ulcer, cerebrovascular disease, respiratory insufficiency, hepatic or renal insufficiency, epilepsy); antihypertensive medication; risk of suicide; cognitive impairment interfering with the understanding of the study; current or recent (3 months before start of the study), pharmacological treatment for alcohol dependence (i.e. acamprosate, naltrexone, disulfiram, ortopiramate); pregnancy or breastfeeding; > 7 days inpatient treatment for SUD in 30 days before start of study; and use of baclofen in past 30 days. Detoxification: participants followed 4–6 weeks' inpatient detoxification. |
|
| Interventions | Baclofen: low dose (31 participants) + CBT
Placebo (31 participants) + CBT
|
|
| Outcomes |
Primary outcomes
Secondary outcomes
|
|
| Identification |
Sponsorship source: funding provided by a private donation through the University of Amsterdam Fund (AUF7344) Author's name: Dr Esther M Beraha Institution: Addiction Development and Psychopathology (ADAPT) Laboratory, Department of Psychology, University of Amsterdam, Amsterdam, the Netherlands Email: R.W.H.J.Wiers@uva.nl (RW Wiers) Address: Addiction Development and Psychopathology Lab, Department of Psychology, University of Amsterdam, Nieuwe Achtergracht 129 B, 1018 WS Amsterdam, the Netherlands |
|
| Notes | In this review update, we divided the study into the 2 following arms:
|
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Randomization (blocks of 6; pre‐stratification by gender and centre) was conducted by the Clinical Research Unit of the AMC and patients were assigned to one of the three groups via an electronic database after baseline assessment." |
| Allocation concealment (selection bias) | Low risk | Quote: "Only the study pharmacist had access to the randomization list and had no further role in the trial." |
| Blinding of participants and personnel (performance bias) Objective outcomes | Low risk | Stated as double‐blind; quote: "The study medication was manufactured, packaged, and labelled by Tiofarma and stored and provided by the pharmacy of the AMC. Baclofen and placebo were provided in identical 10 mg tablets and supplied in containers with 24, 42, 63, 147, or 168 tablets." |
| Blinding of participants and personnel (performance bias) Subjective outcomes | Low risk | Stated as double‐blind; quote: "The study medication was manufactured, packaged, and labelled by Tiofarma and stored and provided by the pharmacy of the AMC. Baclofen and placebo were provided in identical 10 mg tablets and supplied in containers with 24, 42, 63, 147, or 168 tablets." |
| Blinding of outcome assessment (detection bias) Objective outcomes | Low risk | Quote: "The investigators and responsible physicians remained blind with regard to the study medication during the whole study period." |
| Blinding of outcome assessment (detection bias) Subjective outcomes | Low risk | Quote: "The investigators and responsible physicians remained blind with regard to the study medication during the whole study period." |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Quote: "Analyses were done by intention to treat, counting pills every visit and assuming that patients took the drugs they were given." Comment: only 3 participants were lost at follow‐up in the baclofen groups (3%) (Beraha 2016‐HD: 1 participant lost; Beraha 2016‐LD: 2 participants lost). |
| Selective reporting (reporting bias) | Low risk | Study protocol available; results reported for all primary and secondary outcomes reported in the protocol. |
Garbutt 2010a.
| Study characteristics | ||
| Methods |
Study design: RCT Study grouping: parallel group Country: USA Setting: Department of Psychiatry and the Bowles Center for Alcohol Studies (JCG, ABKP, LKJ, BAF), University of North Carolina (outpatients) |
|
| Participants |
Baseline characteristics Baclofen
Placebo
Inclusion criteria: meeting DSM‐IV criteria for current alcohol dependence; ≥ 2 HDDs/week (men ≥ 5 standard drinks/day, women ≥ 4 standard drinks/day) on average during the 4 weeks prior to screening; ability to understand and sign written informed consent; ability to refrain from alcohol for 3 days prior to the randomisation visit. Exclusion criteria: clinically significant medical disease that might interfere with the evaluation of the study medication or presence of a safety concern (e.g. cirrhosis, kidney impairment, unstable hypertension, diabetes mellitus, seizure disorder); clinically significant psychiatric illness including any psychotic disorder, bipolar disorder or severe depression; suicidal ideation; concurrent use of any psychotropic medication including antidepressants, mood stabilisers, antipsychotics, anxiolytics, stimulants or hypnotics with the exception that stable doses of antidepressants for 2 months prior to screening was permitted (6 men and 17 women were receiving antidepressants); concurrent use of anticonvulsants, insulin or oral hypoglycaemics; AST, ALT, or GGT level > 3 times the ULN, bilirubin > ULN, or serum creatinine > ULN; positive urine toxicology screen except cannabis (individuals with positive cannabis screens were excluded only if they had a history of cannabis dependence); pregnant women and women of childbearing potential who did not practice a medically acceptable form of contraception (oral or depot contraceptive, or barrier methods such as diaphragm or condom with spermicidal); people requiring inpatient treatment or more intense outpatient treatment for their alcohol dependency. Detoxification: participants consumed ≥ 2 HDDs/week (men ≥ 5 standard drinks/day, women ≥ 4 standard drinks/day) on average during the 4 weeks prior to screening. |
|
| Interventions | Baclofen (40 participants)
Placebo (40 participants)
|
|
| Outcomes |
Primary outcomes
Secondary outcomes
|
|
| Identification |
Sponsorship source: National Institute of Alcohol Abuse and Alcoholism Grant R21 AA015392 and by GCRC Grant RR00046 and CTSA grant UL1RR025747 from the NIH Author's name: James C Garbutt Institution: Department of Psychiatry and the Bowles Center for Alcohol Studies Email: jc_garbutt@med.unc.edu Address: Chapel Hill, CB#7160, Chapel Hill, North Carolina, 27599‐7160, USA |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Randomization was implemented stratifying on gender, based on a computerized random number generator where assignment to baclofen or placebo was randomly ordered within gender." |
| Allocation concealment (selection bias) | Unclear risk | Information not reported. |
| Blinding of participants and personnel (performance bias) Objective outcomes | Unclear risk | Only stated that study was double‐blind. No further description provided. |
| Blinding of participants and personnel (performance bias) Subjective outcomes | Unclear risk | Only stated that study was double‐blind: no further description provided. |
| Blinding of outcome assessment (detection bias) Objective outcomes | Low risk | Quote: "Medical monitoring was conducted by study physicians and consisted of taking vital signs, recording use of concomitant medication(s), and in‐depth side effect monitoring using an Adverse Events Form that included common baclofen side effects (e.g., drowsiness, sedation, fatigue)." Comment: not stated whether the outcome assessor was blinded, but objective outcomes unlikely to be biased by lack or incomplete blinding. |
| Blinding of outcome assessment (detection bias) Subjective outcomes | High risk | Quote: "Medical monitoring was conducted by study physicians and consisted of taking vital signs, recording use of concomitant medication(s), and in‐depth side effect monitoring using an Adverse Events Form that included common baclofen side effects (e.g., drowsiness, sedation, fatigue)." Comment: not stated whether the outcome assessor was blinded; common adverse events of baclofen easy recognisable. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 20% of participants from placebo group and 27.5% from baclofen group lost at follow‐up. But reasons balanced between groups and most unlikely to be related to the intervention or the outcome. |
| Selective reporting (reporting bias) | Unclear risk | Study protocol unavailable. |
Garbutt 2010b1.
| Study characteristics | ||
| Methods |
Study design: randomised phase 1 study Study grouping: parallel group Blinding: quadruple (participant, care provider, investigator, outcomes assessor) Country: USA |
|
| Participants |
Baseline characteristics 40 men and women aged 25–60 years meeting DSM‐IV criteria for current alcohol dependence Inclusion criteria: admitted to University of North Carolina Hospitals for a medical detoxification from alcohol; receiving benzodiazepines for detoxification or recruited from the general population; mean ≥ 2 HDDs per/week (≥ 5 drinks/day for men, ≥ 4 drinks/day for women); mean overall consumption ≥ 21 drinks/week (men) and ≥ 14 drinks/week (women) during 4 weeks prior to admission or screening; ≤ 3 months of abstinence in previous year; able to understand and sign written informed consent; willingness to engage in treatment and motivation to achieve abstinence or to greatly reduce alcohol consumption; stable residence and able to identify an individual who could locate them if needed; must have the logistical ability to come to each weekly study visit for 12 weeks in Chapel Hill, North Carolina, USA. Detoxification: individuals were not required to be abstinent from alcohol prior to randomisation. |
|
| Interventions |
Intervention characteristics Baclofen + placebo naltrexone + behaviour therapy (10 participants)
Baclofen + naltrexone + behaviour therapy (10 participants)
Placebo characteristics Placebo baclofen + naltrexone + behaviour therapy (10 participants)
Placebo baclofen + placebo naltrexone + behaviour therapy (10 participants)
|
|
| Outcomes | This is an exploratory study to gain experience with the combination of baclofen and naltrexone Primary outcomes
Secondary outcomes
|
|
| Identification | ||
| Notes | The pilot study is a protocol published on ClinicalTrials.gov, where it is reported that the study has been completed without results being posted. Preliminary results have been reported in a conference proceeding. In this review update, we divided the study into the 2 following arms:
|
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | No information provided. |
| Allocation concealment (selection bias) | Unclear risk | No information provided. |
| Blinding of participants and personnel (performance bias) Objective outcomes | Unclear risk | Stated as double‐blind, double‐dummy without further details. |
| Blinding of participants and personnel (performance bias) Subjective outcomes | Unclear risk | Stated as double‐blind, double‐dummy without further details. |
| Blinding of outcome assessment (detection bias) Objective outcomes | Unclear risk | Stated as double‐blind, double‐dummy without further details. |
| Blinding of outcome assessment (detection bias) Subjective outcomes | Unclear risk | Stated as double‐blind, double‐dummy without further details. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No withdrawals from the study. |
| Selective reporting (reporting bias) | Unclear risk | Study protocol unavailable; data available only from conference proceedings. |
Garbutt 2010b2.
| Study characteristics | ||
| Methods |
Study design: randomised phase 1 study Study grouping: parallel group Blinding: quadruple (participant, care provider, investigator, outcomes assessors) Country: USA |
|
| Participants |
Baseline characteristics 20 men and women aged 25–60 years meeting DSM‐IV criteria for current alcohol dependence Inclusion criteria: admitted to University of North Carolina Hospitals for a medical detoxification from alcohol; receiving benzodiazepines for detoxification or recruited from the general population; mean ≥ 2 HDDs per/week (≥ 5 drinks/day for men, ≥ 4 drinks/day for women); mean overall consumption ≥ 21 drinks/week (men) and ≥ 14 drinks/week (women) during 4 weeks prior to admission or screening; ≤ 3 months of abstinence in previous year; able to understand and sign written informed consent; willingness to engage in treatment and motivation to achieve abstinence or to greatly reduce alcohol consumption; stable residence and able to identify an individual who could locate them if needed; must have the logistical ability to come to each weekly study visit for 12 weeks in Chapel Hill, North Carolina, USA. Detoxification: individuals were not required to be abstinent from alcohol prior to randomisation. |
|
| Interventions |
Intervention characteristics Baclofen + placebo naltrexone + behaviour therapy (10 participants)
Placebo characteristics Placebo baclofen + naltrexone + behaviour therapy (10 participants)
|
|
| Outcomes | This is an exploratory study to gain experience with the combination of baclofen + naltrexone Primary outcomes
Secondary outcomes
|
|
| Identification | ||
| Notes | The pilot study is a protocol published on ClinicalTrials.gov, where it is reported that the study has been completed without results being posted. Preliminary results have been reported in a conference proceeding. In this review update, we divided the study into the 2 following arms:
|
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | No information provided. |
| Allocation concealment (selection bias) | Unclear risk | No information provided. |
| Blinding of participants and personnel (performance bias) Objective outcomes | Unclear risk | Stated as double‐blind, double‐dummy without further details. |
| Blinding of participants and personnel (performance bias) Subjective outcomes | Unclear risk | Stated as double‐blind, double‐dummy without further details. |
| Blinding of outcome assessment (detection bias) Objective outcomes | Unclear risk | Stated as double‐blind, double‐dummy without further details. |
| Blinding of outcome assessment (detection bias) Subjective outcomes | Unclear risk | Stated as double‐blind, double‐dummy without further details. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No withdrawals from the study. |
| Selective reporting (reporting bias) | Unclear risk | Study protocol unavailable, data available only from conference proceedings. |
Garbutt 2021‐LD.
| Study characteristics | ||
| Methods |
Study design: RCT Study grouping: parallel group Country: USA Setting: Department of Psychiatry and the Bowles Center for Alcohol Studies (JCG, ABKP, CP, MS, RJ, LW), University of North Carolina (outpatients) |
|
| Participants |
Baseline characteristics Baclofen: low dose (43 participants)
Placebo (20 participants)
Inclusion criteria: aged 21–65 years, diagnosis of DSM‐IV alcohol dependence, to have a reported drinking pattern > 14 standard drinks (women) or 21 drinks (men)/week including ≥ 2 HDDs/week on average (men ≥ 5 standard drinks/day; women ≥ 4 standard drinks/day) during a consecutive 30‐day period within the 90 days prior to screening based on Time‐Line Follow Back interview and to express an interest in significantly reducing or stopping alcohol use. Exclusion criteria: significant medical illness, e.g. diabetes mellitus; significant psychiatric illness, e.g. bipolar disorder or were receiving psychotropic medication except for stable doses of antidepressants for ≥ 1 month; evidence of another SUD except for nicotine dependence or cannabis abuse. Detoxification: sobriety was not a requirement for randomisation. |
|
| Interventions | Baclofen low dose (43 participants) + medical management
Placebo (20 participants) + medical management
|
|
| Outcomes |
Primary outcomes
Secondary outcomes
|
|
| Identification |
Sponsorship source: National Institute of Alcohol Abuse and Alcoholism Grant R21 AA020824 Author's name: James C Garbutt Institution: Department of Psychiatry and the Bowles Center for Alcohol Studies Email: jc_garbutt@med.unc.edu Address: Chapel Hill, CB#7160, Chapel Hill, North Carolina 27599‐7160, USA |
|
| Notes | In the previous version of this review (Minozzi 2018), this was included among the ongoing studies (NCT01980706). In this review update, we divided the study into the 2 following arms:
|
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Participants who met criteria were randomized to either placebo, 30 mg/day baclofen or 90 mg/day baclofen using randomization blocks provided by Dr. Gallop [SAS Institute Inc.] (2015). "A minimization random assignment procedure were used to match subjects." |
| Allocation concealment (selection bias) | Low risk | Quote: "Randomization blocks were prepared by the statistician and provided to the Investigational Drug Services. Randomization was linked to sequential study number which subjects received as they were enrolled in the study (see Protocol, page 10)." |
| Blinding of participants and personnel (performance bias) Objective outcomes | Low risk | Quote: "Blinded baclofen/placebo was provided in blister packs to the research assistant. Investigators had no access to this information. If a medical or other emergency necessitated breaking the blind, IDS personnel had to provide the identity of the medication to the medical study team member" (see protocol, page 10; NCT01980706). |
| Blinding of participants and personnel (performance bias) Subjective outcomes | Low risk | Quote: "Blinded baclofen/placebo was provided in blister packs to the research assistant. Investigators had no access to this information. If a medical or other emergency necessitated breaking the blind, IDS personnel had to provide the identity of the medication to the medical study team member" (see protocol, page 10; NCT01980706). |
| Blinding of outcome assessment (detection bias) Objective outcomes | Low risk | Information not reported; however, objective outcomes are unlikely to be biased by lack of blinding. |
| Blinding of outcome assessment (detection bias) Subjective outcomes | Low risk | Participants were blinded to treatment assignment. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 39% dropout. However, proportions and reasons were balanced across groups. |
| Selective reporting (reporting bias) | Low risk | Study protocol was available and all study's prespecified outcomes were provided. |
Garbutt 2021‐MD.
| Study characteristics | ||
| Methods |
Study design: RCT Study grouping: parallel group Country: USA Setting: Department of Psychiatry and the Bowles Center for Alcohol Studies (JCG, ABKP, CP, MS, RJ, LW), University of North Carolina (outpatients) |
|
| Participants |
Baseline characteristics Baclofen: medium dose (37 participants)
Placebo (20 participants)
Inclusion criteria: aged 21–65 years, diagnosis of DSM‐IV alcohol dependence, to have a reported drinking pattern > 14 standard drinks (women) or 21 drinks (men)/week including ≥ 2 HDDs/week on average (men ≥ 5 standard drinks/day; women ≥ 4 standard drinks/day) during a consecutive 30‐day period within the 90 days prior to screening based on Time‐Line Follow Back interview and to express an interest in significantly reducing or stopping alcohol use. Exclusion criteria: significant medical illness, e.g. diabetes mellitus; significant psychiatric illness, e.g. bipolar disorder or were receiving psychotropic medication except for stable doses of antidepressants for ≥ 1 month, or had evidence of another SUD except for nicotine dependence or cannabis abuse. Detoxification: sobriety was not a requirement for randomisation. |
|
| Interventions | Baclofen medium dose (37 participants) + medical management
Placebo (40 participants) + medical management
|
|
| Outcomes |
Primary outcomes
Secondary outcomes
|
|
| Identification |
Sponsorship source: National Institute of Alcohol Abuse and Alcoholism Grant R21 AA020824 Author's name: James C Garbutt Institution: Department of Psychiatry and the Bowles Center for Alcohol Studies Email: jc_garbutt@med.unc.edu Address: Chapel Hill, CB#7160, Chapel Hill, North Carolina 27599‐7160, USA |
|
| Notes | In the previous version of this review (Minozzi 2018), this was included among the ongoing studies (NCT01980706). In this review update, we divided the study into the 2 following arms:
|
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Participants who met criteria were randomized to either placebo, 30 mg/day baclofen or 90 mg/day baclofen using randomization blocks provided by Dr. Gallop [SAS Institute Inc.] (2015). "A minimization random assignment procedure were used to match subjects." |
| Allocation concealment (selection bias) | Low risk | Quote: "Randomization blocks were prepared by the statistician and provided to the Investigational Drug Services. Randomization was linked to sequential study number which subjects received as they were enrolled in the study (see protocol, page 10; NCT01980706)." |
| Blinding of participants and personnel (performance bias) Objective outcomes | Low risk | Quote: "Blinded baclofen/placebo was provided in blister packs to the research assistant. Investigators had no access to this information. If a medical or other emergency necessitated breaking the blind, IDS personnel had to provide the identity of the medication to the medical study team member" (see protocol, page 10; NCT01980706). |
| Blinding of participants and personnel (performance bias) Subjective outcomes | Low risk | Quote: "Blinded baclofen/placebo was provided in blister packs to the research assistant. Investigators had no access to this information. If a medical or other emergency necessitated breaking the blind, IDS personnel had to provide the identity of the medication to the medical study team member" (see protocol, page 10; NCT01980706). |
| Blinding of outcome assessment (detection bias) Objective outcomes | Low risk | Information not reported; however, objective outcomes are unlikely to be biased by lack of blinding. |
| Blinding of outcome assessment (detection bias) Subjective outcomes | Low risk | Participants were blinded to treatment assignment. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 39% dropout. However, proportions and reasons were balanced across groups. |
| Selective reporting (reporting bias) | Low risk | Study protocol available and all study's prespecified outcomes were provided. |
Hauser 2017.
| Study characteristics | ||
| Methods |
Study design: RCT Study grouping: parallel group Country: USA Setting: outpatients setting in 4 VA medical centres with established hepatology clinics (VA Long Beach, VA Minneapolis, VA Portland and VA San Diego) |
|
| Participants |
Baseline characteristics Baclofen
Placebo
Inclusion criteria: men or women, veterans aged ≥ 18 years; chronic HCV diagnosis with confirmed viraemia by polymerase chain reaction; an AUD (abuse or dependence) according to the DSM‐IV or DSM‐IV‐TR, using the Structured Clinical Interview for the DSM‐IV‐TR; and ongoing alcohol use defined by > 7 standard drinks/week for each preceding 2 weeks or 1 HDD/week for each preceding 2 weeks (HDD: > 4 drinks in 1 day for men and > 3 drinks in 1 day for women). No requirements for any period of abstinence before entry into study. Exclusion criteria: presence of DSM‐IV‐TR (Structured Clinical Interview for DSM‐IV) cocaine, methamphetamine or opioid dependence within the past 6 months; concurrent use of ondansetron, disulfiram, topiramate, naltrexone, acamprosate, buprenorphine or methadone; any metabolic kidney condition affecting renal function, including haemodialysis, renal replacement or peritoneal dialysis; and any known pre‐existing medical conditions that could interfere with participation in the protocol, such as known cognitive impairment, dementia, encephalopathy from liver disease, acute psychiatric instability (significant psychosis, mania, or elevated risk for suicide). Detoxification: participants were required an ongoing alcohol use as defined by > 7 standard drinks/week for each preceding 2 weeks or 1 HDD/week for each preceding 2 weeks (HDD: > 4 drinks in 1 day for men and > 3 drinks in 1 day for women). No requirements for any period of abstinence before entry into study. |
|
| Interventions |
Intervention characteristics Baclofen (88 participants) + BBCET
Placebo (92 participants) + BBCET
Concurrent with dispensing study medication, study personnel provided BBCET, which is a standardised 15‐min psychosocial treatment intervention that emphasises medication adherence as a crucial element to change alcohol use behaviour. |
|
| Outcomes |
Primary outcomes
Secondary outcomes
|
|
| Identification |
Sponsorship source: US Department of Veteran Affairs Clinical Sciences Research and Development Service – VA Merit Author's name: Peter Hauser Institution: Division of Mental Health, Long Beach VA Medical Center, Long Beach, California, USA Email: peter.hauser2@va.gov Address: Long Beach VA Medical Center, 300 Oceangate, Suite 700, Long Beach, California 90802, USA |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Consented patients were randomized to oral baclofen 30 mg (10 mg three times per day) or placebo at each site 1: 1 using random assignment software administered by the central site." |
| Allocation concealment (selection bias) | Low risk | Quote: "The blocked randomization sequence code was concealed from research staff. The research pharmacy at each site kept the randomization code in sequential sealed envelopes and provided study medication to research staff who then distributed study medication to participants." |
| Blinding of participants and personnel (performance bias) Objective outcomes | Low risk | Stated as double‐blind; quote: "Study medication and placebo were placed in identical capsules." |
| Blinding of participants and personnel (performance bias) Subjective outcomes | Low risk | Stated as double‐blind; quote: "Study medication and placebo were placed in identical capsules." |
| Blinding of outcome assessment (detection bias) Objective outcomes | Low risk | Information not reported but objective outcomes unlikely to be biased by lack of blinding. |
| Blinding of outcome assessment (detection bias) Subjective outcomes | Unclear risk | Information not reported. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 24% dropout from baclofen group and 21% dropout from placebo group. Reason reported and balanced between groups. |
| Selective reporting (reporting bias) | Unclear risk | Study protocol unavailable. |
Krupitskii 2017.
| Study characteristics | ||
| Methods |
Study design: RCT Study grouping: parallel group Country: Russia Setting: Department of Narcology, Bekhterev St Petersburg Science Research Psychoneurological Institute |
|
| Participants |
Baseline characteristics Baclofen
Placebo
Inclusion criteria: men and women aged 18–65 years with negative tests for alcohol in exhaled air, ICD‐10 diagnoses of alcohol dependence, and abstention from alcohol consumption for ≥ 7 days (recovered alcohol withdrawal syndrome); pregnancy; women agreeing to use adequate contraception during study; provided telephone number for qualitative follow‐up assessment (compliance with weekly clinic visits). Exclusion criteria: marked organic brain disease, marked somatic pathology (liver, kidney, cardiovascular, nervous system pathology), psychotic states or history of severe mental illness (schizophrenia, epilepsy, manic‐depressive psychosis, etc.); any additional chemical dependency other than on alcohol and tobacco; use of any other medication or psychotherapy for alcohol dependence (including so‐called placebo therapy ('chemical protection'), 'coding', etc). Detoxification: individuals were required to be abstinent from alcohol prior to randomisation for ≥ 7 days (recovered alcohol withdrawal syndrome). |
|
| Interventions |
Intervention characteristics Baclofen (16 participants) + CBT
Placebo (16 participants) + CBT
|
|
| Outcomes |
Primary outcomes
Secondary outcomes
|
|
| Identification |
Sponsorship source: Pharmaceutical Company Akrikhin Author's name: EM Krupitskii Institution: Bekhterev St Petersburg Research Psychoneurological Institute Email: kruenator@gmail.com Address: Bekhterev St, Petersburg Research Psychoneurological Institute, St Petersburg, Russia |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Patients were randomized using a random number generator running in Excel." |
| Allocation concealment (selection bias) | Low risk | Quote: "Agents (baclofen, placebo) were placed in numbered containers and issued to patients in double‐blind conditions at each weekly visit for three months such that they always had a three‐week supply of agent (in case of missing 1–2 visits). Randomization codes were kept at the institute and could be opened at any moment in case of emergency." |
| Blinding of participants and personnel (performance bias) Objective outcomes | Low risk | Stated as double‐blind, quote: "The institute pharmacists, working with the pharmaceuticals company Bios (St. Petersburg), ground baclofen tablets and prepared baclofen capsules (25 mg) and externally identical placebo capsules (starch). Both baclofen capsules and placebo were supplemented with 50 mg of riboflavin as fluorescent marker of drug ingestion." |
| Blinding of participants and personnel (performance bias) Subjective outcomes | Low risk | Stated as double‐blind, quote: "The institute pharmacists, working with the pharmaceuticals company Bios (St. Petersburg), ground baclofen tablets and prepared baclofen capsules (25 mg) and externally identical placebo capsules (starch). Both baclofen capsules and placebo were supplemented with 50 mg of riboflavin as fluorescent marker of drug ingestion." |
| Blinding of outcome assessment (detection bias) Objective outcomes | Low risk | Quote: "Investigators, physicians, and other staff taking part in the study, like the patients, were unaware of which treatment group they were in." |
| Blinding of outcome assessment (detection bias) Subjective outcomes | Low risk | Quote: "Investigators, physicians, and other staff taking part in the study, like the patients, were unaware of which treatment group they were in." |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | No clear data reported about dropout from study. |
| Selective reporting (reporting bias) | Unclear risk | Study protocol unavailable. |
Kumar 2020a.
| Study characteristics | ||
| Methods |
Study design: comparative open RCT Country: India |
|
| Participants |
Baseline characteristics Baclofen
Acamprosate
Naltrexone
Inclusion criteria: diagnosis of current alcohol dependence according to ICD‐10 criteria. Exclusion criteria: individuals with comorbid medical or psychiatric illness; participants were detoxified before treatment; use of other psychoactive substances. Detoxification: participants started study after detoxification. Duration of detoxification not reported. |
|
| Interventions | Baclofen (30 participants)
Acamprosate (30 participants)
Naltrexone (30 participants)
|
|
| Outcomes |
|
|
| Identification | ||
| Notes | Participants had to purchase the baclofen and acamprosate treatment. Naltrexone was free of cost. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Randomisation of the participants was done using a computerised random table. |
| Allocation concealment (selection bias) | Unclear risk | No information provided. |
| Blinding of participants and personnel (performance bias) Objective outcomes | High risk | Open label study. |
| Blinding of participants and personnel (performance bias) Subjective outcomes | High risk | Open label study. |
| Blinding of outcome assessment (detection bias) Objective outcomes | Low risk | No blinding of outcome assessment; however, the objective outcomes were unlikely to be influenced by lack of blinding. |
| Blinding of outcome assessment (detection bias) Subjective outcomes | High risk | No blinding of outcome assessment, and the outcome measurement was likely to be influenced by lack of blinding. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | 15% dropout from baclofen and acamprosate groups; 30% dropout from naltrexone group; unbalanced. |
| Selective reporting (reporting bias) | Unclear risk | Study protocol was unavailable. |
Leggio 2015.
| Study characteristics | ||
| Methods |
Study design: RCT Study grouping: parallel group Country: USA Setting: Brown University Center for Alcohol and Addiction Studies (CAAS) and Roger Williams Medical Center (RWMC), Providence, Rhode Island, USA |
|
| Participants |
Baseline characteristics Baclofen
Placebo
Inclusion criteria: participants were alcohol‐dependent heavy‐drinking and heavy‐smoking individuals: aged 18−75 years; DSM‐IV diagnoses of both alcohol and nicotine codependency, with heavy use of alcohol (men ≥ 5 SDUs/day, women ≥ 4 SDUs/day on average) and cigarettes (≥ 10 cigarettes/day on average) during the last 90 days before screening; interested in receiving treatment for both drinking and smoking (either reducing or stopping both substances; or reducing 1 substance and stopping the other). Exclusion criteria: current (i.e. past year) DSM‐IV diagnosis of dependence on any psychoactive substance other than alcohol and nicotine; lifetime DSM‐IV diagnosis of schizophrenia, bipolar disorder or other psychosis; past year diagnosis of major depression, anxiety disorders and eating disorders; risk of suicide (e.g. active plan or recent attempt in last year); positive urine drug screen at baseline for any illegal substance other than marijuana; significant alcohol withdrawal symptoms, as assessed by a Clinical Institute Withdrawal Assessment for Alcohol Revised score > 10; history of hospitalisation for alcohol intoxication delirium, alcohol withdrawal delirium or seizure; participation in any research study for alcoholism or smoking treatment (or both) within 3 months prior to signing the consent document; pharmacological treatment with naltrexone, acamprosate, topiramate, disulfiram, nicotine replacement, bupropion and varenicline within 1 month prior to randomisation; current use of psychotropic medications or medications that interfere with the metabolism of baclofen, history of allergy to baclofen or medical contraindications to take baclofen; severe medical diseases, such as cancer, cirrhosis, chronic kidney failure and chronic neurological disorders; and women who were of childbearing potential and not using effective contraception. Detoxification: participants were heavy drinkers (men ≥ 5 SDUs/day, women ≥ 4 SDUs/day on average) and smokers (≥ 10 cigarettes/day on average) during the last 90 days before screening. They were interested in receiving treatment for both drinking and smoking (either reducing or stopping both substances; or reducing 1 substance and stopping the other). |
|
| Interventions | Baclofen (15 participants) + medical management (personalised education regarding alcohol and smoking, helped participants to develop and implement a plan to reduce/stop alcohol and smoking, motivated participants for medication adherence)
Placebo (15 participants) + medical management (personalised education regarding alcohol and smoking, helped participants to develop and implement a plan to reduce/stop alcohol and smoking, motivated participants for medication adherence)
|
|
| Outcomes |
Primary outcomes/secondary outcomes (no distinction)
|
|
| Identification |
Sponsorship source: grant from the ABMRF/The Foundation for Alcohol Research (Principal investigator: Leggio). The human laboratory cue‐reactivity substudy was supported by an NIH grant jointly funded by the National Institute on Alcohol Abuse and Alcoholism (NIAAA) and the National Institute on Drug Abuse (NIDA) (R03AA020169; Principal Investigator: Leggio). Both grants were awarded to Dr Leggio while he was at Brown University. Dr Leggio's current work is supported by the NIAAA Division of Intramural Clinical and Biological Research and the NIDA Intramural Research Program Author's name: Lorenzo Leggio Institution: Section on Clinical Psychoneuroendocrinology and Neuropsychopharmacology, Laboratory of Clinical and Translational Studies, National Institute on Alcohol Abuse and Alcoholism, NIH Email: lorenzo.leggio@nih.gov Address: 10 Center Drive (10CRC/15330) MSC1108; Room 1‐5429, Bethesda, Maryland 20892‐1108, USA |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "At week 01 visit, eligible participants were randomized to BACL [baclofen] or PLA [placebo] using a 3‐urn variable procedure." |
| Allocation concealment (selection bias) | Unclear risk | Information not reported. |
| Blinding of participants and personnel (performance bias) Objective outcomes | Low risk | Quote: "BACL [baclofen] at 80 mg/day (20 mg, q.i.d. [4 times/day]), or PLA [placebo] was placed into blister packs as opaque capsules containing drug and 25 mg riboflavine." |
| Blinding of participants and personnel (performance bias) Subjective outcomes | Low risk | Quote: "BACL [baclofen] at 80 mg/day (20 mg, q.i.d. [4 times/day]), or PLA [placebo] was placed into blister packs as opaque capsules containing drug and 25 mg riboflavine." |
| Blinding of outcome assessment (detection bias) Objective outcomes | Low risk | Study described as double‐blind. Outcome unlikely to be biased by lack or inadequate blinding. |
| Blinding of outcome assessment (detection bias) Subjective outcomes | Low risk | Participants blinded to treatments assignment. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Quote: "while 30 individuals were eligible and randomized; 24 completed the study (Supplemental Fig. 1) [of the publication]." Comment: 20% dropout. Number of participants who dropped out from each group not reported. Figure 1 (of the publication) reporting participants lost to follow‐up and those who completed the study is unreadable in the supplementary file so that it is impossible to give a judgement on this. |
| Selective reporting (reporting bias) | Low risk | Study protocol available. Results reported for all the primary and secondary outcomes declared in the protocol |
Mishra 2010.
| Study characteristics | ||
| Methods |
Study design: RCT Study grouping: parallel group Country: India Setting: outpatients |
|
| Participants |
Baseline characteristics Baclofen
Acamprosate
Inclusion criteria: aged 18−70 years; diagnosis of current alcohol dependence according to ICD‐10 criteria; last alcohol intake reported to have taken place in the 24 hours preceding observation; presence of a key relative. Exclusion criteria: presence of severe liver, kidney, heart or lung diseases requiring urgent medical attention; currently under treatment for any mental disorder with psychotic drugs; any dependence to drug other than nicotine; age > 70 years. Detoxification: last alcohol intake reported to have taken place in the 24 hours preceding observation was an inclusion criterion. |
|
| Interventions | Baclofen (25 participants)
Acamprosate (24 participants)
|
|
| Outcomes |
Primary outcome/secondary outcome (no distinction)
|
|
| Identification |
Sponsorship source: NR Author's name: Suvendu N Mishra Institution: IMS & SUM Hospital, Bhubaneswar. Mental Health Institute, Dept of Psychiatry SCB Medical College, Cuttack Email: dr.sar07@gmail.com Address: Mental Health Institute, SCB Medical college, Cuttack |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Information not provided. |
| Allocation concealment (selection bias) | Unclear risk | Information not provided. |
| Blinding of participants and personnel (performance bias) Objective outcomes | High risk | Quote: "Participants given written informed consent, subsequently randomised into 2 groups to receive either open‐label baclofen or acamprosate." |
| Blinding of participants and personnel (performance bias) Subjective outcomes | High risk | Quote: "Participants given written informed consent, subsequently randomised into 2 groups to receive either open‐label baclofen or acamprosate." |
| Blinding of outcome assessment (detection bias) Objective outcomes | Low risk | No information provided. However, the objective outcomes are unlikely to be biased by lack of blinding. |
| Blinding of outcome assessment (detection bias) Subjective outcomes | High risk | Quote: "Participants given written informed consent, subsequently randomised into 2 groups to receive either open‐label baclofen or acamprosate." |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Information not reported. |
| Selective reporting (reporting bias) | High risk | Study protocol unavailable. Primary and secondary outcomes not reported in the method section. Some results on specific outcomes were partially reported (e.g. for abstinence: quote: "A significantly higher number of patients who achieved and maintained abstinence throughout the experiment were found in the group of patients treated with baclofen compared with acamprosate."). |
Morley 2014‐LD.
| Study characteristics | ||
| Methods |
Study design: RCT Study grouping: parallel group Country: Australia Setting: outpatients |
|
| Participants |
Baseline characteristics Baclofen
Placebo
Inclusion criteria: men and women aged 18–60 years; DSM‐IV criteria for current alcohol dependence; ability to understand and provide written informed consent; abstinence from alcohol for ≥ 3 days prior to randomisation and resolution of any withdrawal symptoms; desire to achieve abstinence or to reduce alcohol consumption; evidence of a stable residence; proof of an individual who could locate participant if needed. Exclusion criteria: clinically significant medical diseases that might interfere with the evaluation of the study medication or present a safety concern (e.g. kidney impairment, unstable hypertension, hypotension, diabetes mellitus, seizure disorder); clinically significant psychiatric illness (e.g. psychotic disorder, bipolar disorder, severe depression); suicidal ideation or concurrent SUD other than nicotine or cannabis, concurrent use of any psychotropic medication (participants who had been on a stable dose of selective serotonin reuptake inhibitors for 2 months were still considered eligible); concurrent use of anticonvulsants, insulin or oral hypoglycaemic; pregnant or breastfeeding; participation in any clinical trial within last 60 days; use of alcohol pharmacotherapy within last 60 days; court‐mandated participation in alcohol treatment or pending incarceration. Detoxification: participants had to be abstinent from alcohol for ≥ 3 days prior to randomisation and resolution of any withdrawal symptoms. |
|
| Interventions | Baclofen (14 participants) + up to 9 sessions of BRENDA, a low‐intensity psychosocial intervention
Placebo (7 participants) + up to 9 sessions of BRENDA, a low‐intensity psychosocial intervention
|
|
| Outcomes |
Primary outcomes
Secondary outcomes
|
|
| Identification |
Sponsorship source: New South Wales Health Drug and Alcohol grant (PH, AB, KM). Conflict of interest statement. None declared Author's name: KC Morley Institution: NHMRC Centre of Research Excellence in Mental Health and Substance Use, Discipline of Addiction Medicine, University of Sydney, Sydney, New South Wales, Australia Email: kirsten.morley@sydney.edu.au Address: University of Sydney, Sydney, New South Wales 2006, Australia |
|
| Notes | In this review update, we divided the study into the 2 following arms:
|
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Participants were allocated 1:1:1 as per a computer‐generated randomization sequence conducted by the hospital clinical trials pharmacist." |
| Allocation concealment (selection bias) | Low risk | Quote: "Participants were allocated 1:1:1 as per a computer‐generated randomization sequence conducted by the hospital clinical trials pharmacist." |
| Blinding of participants and personnel (performance bias) Objective outcomes | Low risk | Quote: "participants in the (a) baclofen 30 mg/day group took a dose of 5 mg, three times a day, for the first 3 days, a dose of 10 mg, three times a day, on Days 4–81, and finally a dose of 5 mg, three times a day, for the last 3 days; (b) baclofen 60 mg/day group took a dose of 5 mg, three times a day, for the first 3 days, a dose of 10 mg, three times a day on Days 4–7, a dose of 20 mg, three times a day, on Days 8–77, a dose of 10 mg three times a day on Days 78–81, and finally a dose of 5 mg, three times a day, for the last 3 days. The placebo pills, which were identical in appearance, were also titrated upward and downward to maintain the double blind." |
| Blinding of participants and personnel (performance bias) Subjective outcomes | Low risk | Quote: "participants in the (a) baclofen 30 mg/day group took a dose of 5 mg, three times a day, for the first 3 days, a dose of 10 mg, three times a day, on Days 4–81, and finally a dose of 5 mg, three times a day, for the last 3 days; (b) baclofen 60 mg/day group took a dose of 5 mg, three times a day, for the first 3 days, a dose of 10 mg, three times a day on Days 4–7, a dose of 20 mg, three times a day, on Days 8–77, a dose of 10 mg three times a day on Days 78–81, and finally a dose of 5 mg, three times a day, for the last 3 days. The placebo pills, which were identical in appearance, were also titrated upward and downward to maintain the double blind." |
| Blinding of outcome assessment (detection bias) Objective outcomes | Low risk | Information not reported; however, objective outcomes unlikely to be biased by lack or incomplete blinding of outcome assessor. |
| Blinding of outcome assessment (detection bias) Subjective outcomes | Low risk | Participants were blinded to treatment assignment. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Quote: "Twenty‐eight (67%) patients out of the enrolled 42 completed the follow‐up interviews (up to Week 12–16)." Quote: "There were no significant differences between groups on study retention (χ [Chi]2 = 1.5, P = 0.47) or medication compliance rates (χ2 = 0.77, P = 0.68), the number of BRENDA sessions attended (F = 1.93, P = 0.826) or days on medication (F = 1.04, P = 0.36). Time‐survival analysis revealed that the treatment retention rates did not significantly differ between randomized groups over the follow‐up assessments (χ2 = 1.21, P = 0.55)." Comment: reason for discontinuation not reported for each group |
| Selective reporting (reporting bias) | Unclear risk | Study protocol available. 2 primary outcomes not listed in the protocol were reported in the full publication (time to relapse, time to lapse); however, authors found no difference for either of these outcomes. Comment: selective reporting bias judged unlikely. |
Morley 2014‐MD.
| Study characteristics | ||
| Methods |
Study design: RCT Study grouping: parallel group Country: Australia Setting: outpatients |
|
| Participants |
Baseline characteristics Baclofen
Placebo
Inclusion criteria: men and women aged 18–60 years; DSM‐IV criteria for current alcohol dependence; ability to understand and provide written informed consent; abstinence from alcohol for ≥ 3 days prior to randomisation and resolution of any withdrawal symptoms; desire to achieve abstinence or to reduce alcohol consumption; evidence of a stable residence; proof of an individual who could locate participant if needed. Exclusion criteria: clinically significant medical diseases that might interfere with the evaluation of the study medication or present a safety concern (e.g. kidney impairment, unstable hypertension, hypotension, diabetes mellitus, seizure disorder); clinically significant psychiatric illness (e.g. psychotic disorder, bipolar disorder, severe depression); suicidal ideation or concurrent SUD other than nicotine or cannabis, concurrent use of any psychotropic medication (participants who had been on a stable dose of selective serotonin reuptake inhibitors (SSRIs) for 2 months were still considered eligible); concurrent use of anticonvulsants, insulin or oral hypoglycaemic; pregnant or breastfeeding; participation in any clinical trial within last 60 days; use of alcohol pharmacotherapy within the last 60 days; court‐mandated participation in alcohol treatment or pending incarceration. Detoxification: participants had to be abstinent from alcohol for ≥ 3 days prior to randomisation and resolution of any withdrawal symptoms. |
|
| Interventions | Baclofen (14 participants) + up to 9 sessions of BRENDA, a low‐intensity psychosocial intervention
Placebo (7 participants) + up to 9 sessions of BRENDA, a low‐intensity psychosocial intervention
|
|
| Outcomes |
Primary outcomes
Secondary outcomes
|
|
| Identification |
Sponsorship source: New South Wales Health Drug and Alcohol grant (PH, AB, KM). Conflict of interest statement. None declared Author's name: KC Morley Institution: NHMRC Centre of Research Excellence in Mental Health and Substance Use, Discipline of Addiction Medicine, University of Sydney, Sydney, New South Wales, Australia Email: kirsten.morley@sydney.edu.au Address: University of Sydney, Sydney, New South Wales 2006, Australia |
|
| Notes | In this review update, we divided the study into the 2 following arms:
|
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Participants were allocated 1:1:1 as per a computer‐generated randomization sequence conducted by the hospital clinical trials pharmacist." |
| Allocation concealment (selection bias) | Low risk | Quote: "Participants were allocated 1:1:1 as per a computer‐generated randomization sequence conducted by the hospital clinical trials pharmacist." |
| Blinding of participants and personnel (performance bias) Objective outcomes | Low risk | Quote: "participants in the (a) baclofen 30 mg/day group took a dose of 5 mg, three times a day, for the first 3 days, a dose of 10 mg, three times a day, on Days 4–81, and finally a dose of 5 mg, three times a day, for the last 3 days; (b) baclofen 60 mg/day group took a dose of 5 mg, three times a day, for the first 3 days, a dose of 10 mg, three times a day on Days 4–7, a dose of 20 mg, three times a day, on Days 8–77, a dose of 10 mg three times a day on Days 78–81, and finally a dose of 5 mg, three times a day, for the last 3 days. The placebo pills, which were identical in appearance, were also titrated upward and downward to maintain the double blind." |
| Blinding of participants and personnel (performance bias) Subjective outcomes | Low risk | Quote: "participants in the (a) baclofen 30 mg/day group took a dose of 5 mg, three times a day, for the first 3 days, a dose of 10 mg, three times a day, on Days 4–81, and finally a dose of 5 mg, three times a day, for the last 3 days; (b) baclofen 60 mg/day group took a dose of 5 mg, three times a day, for the first 3 days, a dose of 10 mg, three times a day on Days 4–7, a dose of 20 mg, three times a day, on Days 8–77, a dose of 10 mg three times a day on Days 78–81, and finally a dose of 5 mg, three times a day, for the last 3 days. The placebo pills, which were identical in appearance, were also titrated upward and downward to maintain the double blind." |
| Blinding of outcome assessment (detection bias) Objective outcomes | Low risk | Information not reported; however, objective outcomes unlikely to be biased by lack or incomplete blinding of outcome assessor. |
| Blinding of outcome assessment (detection bias) Subjective outcomes | Low risk | Participants were blinded to treatment assignment. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Quote: "Twenty‐eight (67%) patients out of the enrolled 42 completed the follow‐up interviews (up to Week 12–16)." Quote: "There were no significant differences between groups on study retention (χ [Chi]2 = 1.5, P = 0.47) or medication compliance rates (χ2 = 0.77, P = 0.68), the number of BRENDA sessions attended (F = 1.93, P = 0.826) or days on medication (F = 1.04, P = 0.36). Time‐survival analysis revealed that the treatment retention rates did not significantly differ between randomized groups over the follow‐up assessments (χ2 = 1.21, P = 0.55)." Comment: reason for discontinuation not reported for each group. |
| Selective reporting (reporting bias) | Unclear risk | Study protocol available. 2 primary outcomes not listed in the protocol were reported in the full publication (time to relapse, time to lapse); however. authors found no difference for either of these outcomes. Comment: selective reporting bias judged unlikely. |
Morley 2018‐LD.
| Study characteristics | ||
| Methods |
Study design: RCT Study grouping: parallel group Country: Australia Setting: outpatients |
|
| Participants |
Baseline characteristics Baclofen: low dose (36 participants)
Placebo (17 participants)
Inclusion criteria: alcohol dependence according to ICD‐10 criteria; aged 18–75 years; adequate cognition and English language skills to give valid consent and complete research interviews; willingness to give written informed consent; abstinence from alcohol for 3–21 days before enrolment; resolution of any clinically evident alcohol withdrawal (on the Clinical Institute Withdrawal Assessment for alcohol); < 48 hours after ceasing any diazepam required for withdrawal management. Exclusion criteria: active major mental disorder associated with psychosis or significant suicide risk; pregnant or breastfeeding; concurrent use of any psychotropic medication other than antidepressants (provided these are taken at stable doses for ≥ 2 months); unstable substance use; clinical evidence of persisting hepatic encephalopathy (drowsiness, sleep inversion or asterixis); pending incarceration; lack of stable housing; peptic ulcer; unstable diabetes mellitus. Detoxification: participants had to be abstinent from alcohol for 3–21 days before enrolment. |
|
| Interventions | Baclofen low dose (36 participants) + medical care + brief adherence therapy
Placebo (17 participants) + medical care + brief adherence therapy
|
|
| Outcomes |
Primary outcomes
Secondary outcomes
|
|
| Identification |
Sponsorship source: grant from the National Health and Medical Research Council of Australia (PSH, AB, KCM). Conflict of interest statement: none declared Author's name: KC Morley Institution: NHMRC Centre of Research Excellence in Mental Health and Substance Use, Discipline of Addiction Medicine, University of Sydney, Sydney, New South Wales, Australia Email: kirsten.morley@sydney.edu.au Address: University of Sydney, Sydney, New South Wales 2006, Australia Trial registration: ClinicalTrials.gov: NCT01711125 |
|
| Notes | In this review update, we divided the study into the 2 following arms:
|
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Participants randomly were allocated 1:1:1. Allocation was made by a computer‐generated block randomisation sequence that was developed by an independent team at a neighbouring institution (National Drug and Alcohol Research Centre, University of New South Wales), and then sent to the hospital clinical trials pharmacists." |
| Allocation concealment (selection bias) | Low risk | Quote: "Allocation was made by a computer‐generated block randomisation sequence that was developed by an independent team at a neighbouring institution (National Drug and Alcohol Research Centre, University of New South Wales), and then sent to the hospital clinical trials pharmacists." |
| Blinding of participants and personnel (performance bias) Objective outcomes | Low risk | Quote: "Participants, clinicians and research team members were masked. The integrity of the double‐blind procedure was assessed by obtaining a prediction from each client as to their allocated treatment and, in addition, a prediction from the therapist and researcher (active or placebo)." |
| Blinding of participants and personnel (performance bias) Subjective outcomes | Low risk | Quote: "Participants, clinicians and research team members were masked. The integrity of the double‐blind procedure was assessed by obtaining a prediction from each client as to their allocated treatment and, in addition, a prediction from the therapist and researcher (active or placebo)." |
| Blinding of outcome assessment (detection bias) Objective outcomes | Low risk | Research team members were blinded to treatment assignment. |
| Blinding of outcome assessment (detection bias) Subjective outcomes | Low risk | Participants were blinded to treatment assignment. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 27% dropped out of baclofen group and 31% from placebo group, reasons for withdrawal provided. Quote: "in the case of primary outcomes (survival and multivariate), we employed sensitivity analyses to compare the results from raw data with those from multiple imputation (with ten iterations). Raw data are presented given that analyses revealed no change between the results for raw data v. using multiple imputation. Mixed models were also used where possible." |
| Selective reporting (reporting bias) | Unclear risk | Study protocol was unavailable. |
Morley 2018‐MD.
| Study characteristics | ||
| Methods |
Study design: RCT Study grouping: parallel group Country: Australia Setting: outpatients |
|
| Participants |
Baseline characteristics Baclofen: medium dose (35 participants)
Placebo (16 participants)
Inclusion criteria: alcohol dependence according to ICD‐10 criteria; aged 18–75 years; adequate cognition and English language skills to give valid consent and complete research interviews; willingness to give written informed consent; abstinence from alcohol for 3–21 days before enrolment; resolution of any clinically evident alcohol withdrawal (on the Clinical Institute Withdrawal Assessment for alcohol; < 48 hours after ceasing any diazepam required for withdrawal management. Exclusion criteria: active major mental disorder associated with psychosis or significant suicide risk; pregnant or breastfeeding; concurrent use of any psychotropic medication other than antidepressants (provided these are taken at stable doses for ≥ 2 months); unstable substance use; clinical evidence of persisting hepatic encephalopathy (drowsiness, sleep inversion or asterixis); pending incarceration; lack of stable housing; peptic ulcer; unstable diabetes mellitus. Detoxification: participants had to be abstinent from alcohol for 3–21 days before enrolment. |
|
| Interventions | Baclofen medium dose (35 participants) + medical care + brief adherence therapy
Placebo (16 participants) + medical care + brief adherence therapy
|
|
| Outcomes |
Primary outcomes
Secondary outcomes
|
|
| Identification |
Sponsorship source: the study was supported by a grant from the National Health and Medical Research Council of Australia (P.S.H., A.B., K.C.M.). Conflict of interest statement. None declared Author's name: KC Morley Institution: NHMRC Centre of Research Excellence in Mental Health and Substance Use, Discipline of Addiction Medicine, University of Sydney, Sydney, New South Wales, Australia Email: kirsten.morley@sydney.edu.au Address: University of Sydney, Sydney, New South Wales 2006, Australia Trial registration: ClinicalTrials.gov: NCT01711125 |
|
| Notes | In this review update, we divided the study into the 2 following arms:
|
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Participants randomly were allocated 1:1:1. Allocation was made by a computer‐generated block randomisation sequence that was developed by an independent team at a neighbouring institution (National Drug and Alcohol Research Centre, University of New South Wales), and then sent to the hospital clinical trials pharmacists." |
| Allocation concealment (selection bias) | Low risk | Quote: "Allocation was made by a computer‐generated block randomisation sequence that was developed by an independent team at a neighbouring institution (National Drug and Alcohol Research Centre, University of New South Wales), and then sent to the hospital clinical trials pharmacists." |
| Blinding of participants and personnel (performance bias) Objective outcomes | Low risk | Quote: "Participants, clinicians and research team members were masked. The integrity of the double‐blind procedure was assessed by obtaining a prediction from each client as to their allocated treatment and, in addition, a prediction from the therapist and researcher (active or placebo)." |
| Blinding of participants and personnel (performance bias) Subjective outcomes | Low risk | Quote: "Participants, clinicians and research team members were masked. The integrity of the double‐blind procedure was assessed by obtaining a prediction from each client as to their allocated treatment and, in addition, a prediction from the therapist and researcher (active or placebo)." |
| Blinding of outcome assessment (detection bias) Objective outcomes | Low risk | Research team members were blinded to treatment assignment. |
| Blinding of outcome assessment (detection bias) Subjective outcomes | Low risk | Participants were blinded to treatment assignment. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 31% and 27% dropped out from placebo and baclofen group respectively, reason for withdrawal provided. Quote: "in the case of primary outcomes (survival and multivariate), we employed sensitivity analyses to compare the results from raw data with those from multiple imputation (with ten iterations). Raw data are presented given that analyses revealed no change between the results for raw data v. using multiple imputation. Mixed models were also used where possible." |
| Selective reporting (reporting bias) | Unclear risk | Study protocol unavailable. |
Muller 2015.
| Study characteristics | ||
| Methods |
Study design: RCT Study grouping: parallel group Country: Germany Setting: outpatients |
|
| Participants |
Baseline characteristics Baclofen
Placebo
Inclusion criteria: aged ≥ 18 to < 65 years; diagnosis of alcohol dependence according to ICD‐10 and DSM‐IV‐TR; alcohol consumption of ≥ 2 HDDs/week on average (men ≥ 5 drinks/day; women < 4 drinks/day; 1 standard drink = 12 g absolute alcohol) and a mean overall alcohol intake of ≥ 21 drinks/week (men) and ≥ 14 drinks/week (women) during the 4 weeks before detoxification; completed inpatient or outpatient detoxification before randomisation; last alcohol consumption within 7–21 days before randomisation; sufficient German language skills. Exclusion criteria: neurological conditions; current treatment with psychotropic drugs that could affect study outcome (i.e. sedatives, alcohol relapse prevention such as acamprosate, disulfiram, naltrexone, antidepressants, antipsychotics, anticonvulsants); epilepsy or epileptiform convulsions; pregnancy or currently breastfeeding (or both); intolerance to baclofen; terminal renal failure; ALT or AST values 5 times the ULN, bilirubin 41.9 mg/dL, international normalised ratio 41.6; gastrointestinal ulcers; treatment mandated by a legal authority. Detoxification: a complete inpatient or outpatient detoxification before randomisation was an inclusion criterion. However, participants had their last alcohol consumption within 7–21 days before randomisation. |
|
| Interventions | Baclofen (28 participants) + up to 9 medical management sessions that focused on psychoeducation and enhancement of motivation and adherence
Placebo (28 participants) + up to 9 Medical Management sessions that focus on psychoeducation and enhancement of motivation and adherence
|
|
| Outcomes |
Primary outcomes
Secondary outcomes
|
|
| Identification |
Trial Registration Identifier: BACLAD study at ClinicalTrials.gov: NCT01266655. Sponsorship source: supported by the German Research Foundation (DFG; Cluster of Excellence EXC 257) Author's name: Christian A Müllera Institution: Department of Psychiatry, Campus Charité Mitte, Charité, Universitätsmedizin Berlin, Germany Email: ch.mueller@charite.de Address: Universitätsmedizin Berlin, Charitéplatz 1, 10117 Berlin, Germany |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "patients were randomly assigned at the baseline visit to double‐blind treatment with baclofen or placebo in a 1:1 ratio according to a computer‐generated randomization list (in blocks of 4; stratification with regard to sex)." |
| Allocation concealment (selection bias) | Low risk | Quote: "The randomization list was kept by the biometrician and the study pharmacist who prepared the study medication packages. The study pharmacist did not have any further role in the trial. Sealed envelopes containing study medication details were kept at the outpatient unit to be opened by a staff member in case of a study drug‐related emergency. During the whole study, no unblinding was necessary." |
| Blinding of participants and personnel (performance bias) Objective outcomes | Low risk | Stated as double‐blind. Quote: "Participants received baclofen or placebo in identical capsules in a dose of 5 mg 3 times/d. Sealed envelopes containing study medication details were kept at the outpatient unit to be opened by a staff member in case of a study drug‐related emergency. During the whole study, no unblinding was necessary." |
| Blinding of participants and personnel (performance bias) Subjective outcomes | Low risk | Stated as double‐blind. Quote: "Participants received baclofen or placebo in identical capsules in a dose of 5 mg 3 times/d. Sealed envelopes containing study medication details were kept at the outpatient unit to be opened by a staff member in case of a study drug‐related emergency. During the whole study, no unblinding was necessary." |
| Blinding of outcome assessment (detection bias) Objective outcomes | Low risk | Quote: "Sealed envelopes containing study medication details were kept at the outpatient unit to be opened by a staff member in case of a study drug‐related emergency. During the whole study, no unblinding was necessary." |
| Blinding of outcome assessment (detection bias) Subjective outcomes | Low risk | Participants were blinded to treatment assignment. Quote: "Sealed envelopes containing study medication details were kept at the outpatient unit to be opened by a staff member in case of a study drug‐related emergency. During the whole study, no unblinding was necessary." |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 7% lost at follow‐up; balanced between groups |
| Selective reporting (reporting bias) | Unclear risk | Study protocol unavailable. |
Ponizovsky 2015.
| Study characteristics | ||
| Methods |
Study design: RCT Study grouping: parallel group Country: Israel Setting: outpatients |
|
| Participants |
Baseline characteristics Baclofen
Placebo
Inclusion criteria: aged 18–60 years; ICD‐10 diagnosis of AD; sought treatment to stop alcohol consumption; alcohol intake ≥ 2 HDDs/week (men ≥ 5 drinks/day; women ≥ 4 drinks/day) and mean overall consumption of ≥ 21 drinks/week (men) and ≥ 14 drinks/week (women) during the month preceding recruitment (1 standard drink = 12 g absolute alcohol); < 6 total abstinent days per month on average; reliable family member able to help with drug administration and monitoring. Study population defined as mild AUD according to DSM‐5 criteria. Exclusion criteria: detoxification treatment for acute alcohol withdrawal syndrome (requiring hospitalisation) during the month before randomisation; chronic use of psychotropic medication before randomisation; dependence on psychoactive substances other than nicotine; liver cirrhosis; acute alcohol psychosis; severe depression; organic brain syndromes; pregnancy and lactation. Detoxification: a detoxification treatment for acute alcohol withdrawal syndrome (requiring hospitalisation) during the month before randomisation was an exclusion criterion. |
|
| Interventions | Baclofen (32 participants) + standard psychosocial intervention (motivational interviewing, education and therapy)
Placebo (32 participants) + standard psychosocial intervention (motivational interviewing, education and therapy)
|
|
| Outcomes |
Primary outcomes
Secondary outcomes
|
|
| Identification |
Sponsorship source: Israeli Anti‐Drug Authority Grant #535‐28. AM Ponizovsky was supported in part by the Ministry of Immigrant Absorption Author's name: AM Ponizovsky Institution: Research Unit, Mental Health Services, Ministry of Health, Jerusalem, Israel Email: alexpon8@gmail.com Address: Ministry of Health 39 Yirmiyahu St, PO Box 1176, Jerusalem 9446724, Israel |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Randomization was performed by the pharmacist who prepared drug and placebo by using a random number generator, with the only restriction that the groups should be of equal size." |
| Allocation concealment (selection bias) | Low risk | Quote: "Randomization was performed by the pharmacist who prepared drug and placebo by using a random number generator, with the only restriction that the groups should be of equal size." |
| Blinding of participants and personnel (performance bias) Objective outcomes | Low risk | Stated as double‐blind. Quote: "Placebo tablets were identical in all organoleptic characteristics to baclofen." |
| Blinding of participants and personnel (performance bias) Subjective outcomes | Low risk | Stated as double‐blind. Quote: "Placebo tablets were identical in all organoleptic characteristics to baclofen." |
| Blinding of outcome assessment (detection bias) Objective outcomes | Low risk | Not reported whether the outcome assessor was blinded. However, objective outcomes are unlikely to be biased by lack of blinding. |
| Blinding of outcome assessment (detection bias) Subjective outcomes | Low risk | Participants were blinded to treatment assignment. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Quote: "All outcome variables for patients who discontinued treatment before the end of the medication period were computed with the registers of the visit preceding discontinuation (last observation carried forward), which were compared between the treatment groups by Fisher's exact test. Based on the assumption that every patient took the allocated drug, intention to treat analysis was made All randomized patients were analyzed (intent‐to‐treat)." |
| Selective reporting (reporting bias) | Unclear risk | Study protocol unavailable. |
Reynaud 2017.
| Study characteristics | ||
| Methods |
Study design: RCT Study grouping: parallel group Country: France Setting: outpatients |
|
| Participants |
Baseline characteristics Baclofen
Placebo
Inclusion criteria: adult men or non‐pregnant, non‐breastfeeding women, with a diagnosis of alcohol dependence according to DSM‐IV, who had experienced ≥ 1 previous abstinence attempt, and had been fully abstinent for 3–14 days before randomisation; this range of 3–14 days was established in order to allow participants needing inpatient detoxification to participate in the study. Comorbid psychiatric diseases were assessed according to the investigator's judgement. Exclusion criteria: need for a prolonged residential treatment after detoxification; need for an intensive psychosocial intervention during follow‐up; history of baclofen intake by prescription or by self‐medication; epilepsy or history of epilepsy; concomitant treatment with ≥ 1 drugs for the maintenance of abstinence; concomitant treatment with psychotropic medications, except antidepressants at stable dose for ≥ 2 months, diazepam and oxazepam; severe renal, cardiac or pulmonary disorders; severe psychiatric conditions (schizophrenia and bipolar disorder); clinically significant cognitive disorders; hepatic encephalopathy; suicidal risk or history of suicide; other current dependence except nicotine. Detoxification: participants had been fully abstinent for 3–14 days before randomisation; this range of 3–14 days was established to allow participants needing inpatient detoxification to participate in the study. |
|
| Interventions | Baclofen (158 participants) + BRENDA (a low‐intensity psychosocial intervention) sessions were provided during each visit to support participants in changing their behaviour and to enhance adherence to treatment.
Placebo (162 participants) + BRENDA (a low‐intensity psychosocial intervention) sessions were provided during each visit to support participants in changing their behaviour and to enhance adherence to treatment.
|
|
| Outcomes |
Primary outcomes
Secondary outcomes
|
|
| Identification |
Sponsorship source: study was supported by Ethypharm SAS Author's name: Michel Reynaud Institution: Actions Addictions‐Saint Joseph Hospital, 185 Rue Raymond Losserand, 75014 Paris, France Email: michel.reynaud@actions‐addictions.org Address: Saint Joseph Hospital, 185 rue Raymond Losserand, 75014 Paris, France |
|
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Patients were randomly assigned to baclofen or placebo in a 1:1 ratio, according to a computer generated randomization list (blocks size of 4)." |
| Allocation concealment (selection bias) | Low risk | Quote: "The allocation sequence was centralized via an Interactive Web Response System." |
| Blinding of participants and personnel (performance bias) Objective outcomes | Low risk | Quote: "The identical aspect of verum (20 mg coated scored baclofen tablet) and placebo tablets allowed a double‐blind design. Sealed code envelopes were sent to the investigator centres with the corresponding study treatments. Access to the randomization codes and unblinding could only be performed in case of emergency." |
| Blinding of participants and personnel (performance bias) Subjective outcomes | Low risk | Quote: "The identical aspect of verum (20 mg coated scored baclofen tablet) and placebo tablets allowed a double‐blind design. Sealed code envelopes were sent to the investigator centres with the corresponding study treatments. Access to the randomization codes and unblinding could only be performed in case of emergency." |
| Blinding of outcome assessment (detection bias) Objective outcomes | Low risk | Quote: "The identical aspect of verum (20 mg coated scored baclofen tablet) and placebo tablets allowed a double‐blind design. Sealed code envelopes were sent to the investigator centres with the corresponding study treatments. Access to the randomization codes and unblinding could only be performed in case of emergency." |
| Blinding of outcome assessment (detection bias) Subjective outcomes | Low risk | Quote: "The identical aspect of verum (20 mg coated scored baclofen tablet) and placebo tablets allowed a double‐blind design. Sealed code envelopes were sent to the investigator centres with the corresponding study treatments. Access to the randomization codes and unblinding could only be performed in case of emergency." |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Quote: "The safety population, defined as patients having received at least one dose of study treatment, and considered for safety purpose; The full analysis set population, defined as randomized patients, having received at least one dose of study treatment and having reported at least one data regarding alcohol consumption in their diary, which is the main population for efficacy assessments." Quote: "Three methods of imputation were used for the management of missing data related to alcohol consumption: Multiple imputation with a placebo pattern mixture model (assuming that the alcohol consumption of dropped out patients was the same as those in the placebo group) was the main imputation method; it is one of the recommended method for handling missing data in alcohol clinical trials. Most plausible outcome (for abstinence endpoint only): for patients who did not report any alcohol consumption during the 20 consecutive weeks and had at least one missing data during the period, their profiles were reviewed by 2 blinded medical experts who filled in the missing data; Worst case: missing data were imputed to alcohol intake." |
| Selective reporting (reporting bias) | Unclear risk | Study protocol unavailable. |
Rigal 2020.
| Study characteristics | ||
| Methods |
Study design: double‐blind RCT Country: France |
|
| Participants |
Baseline characteristics Baclofen
Placebo
Inclusion criteria: aged 18–65 years; at high‐risk drinking during last 3 months (at least twice per month); for women, > 40 g/day or > 280 g/week or > 40 g in 1 session; for men, > 60 g/day or > 420 g/week, or > 60 g at in 1 session. Exclusion criteria: pregnant, nursing, or of childbearing age and not using effective contraception; serious psychiatric illness (psychosis, notably schizophrenia and bipolar disorders); sufficiently serious organic pathology interfering with his/her inclusion in the study in the investigator's opinion. Detoxification: sobriety was not required. |
|
| Interventions | Baclofen (162 participants)
Placebo (158 participants)
|
|
| Outcomes |
Primary outcomes
Secondary outcomes
|
|
| Identification | ||
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "An independent statistician provided a computer generated randomization list, with permuted blocks of size 4 stratified by primary care center." |
| Allocation concealment (selection bias) | Low risk | Quote: "Allocation was concealed by use of a Web‐based inclusion system." |
| Blinding of participants and personnel (performance bias) Objective outcomes | Low risk | Quote: "Patients, treating physicians, data collectors and statisticians were blinded to treatment allocation." Quote: "The appearance, size, weight and taste of the placebo tablets were identical to those of the 10‐mg baclofen tablets and the scored tablets were provided in identical containers." |
| Blinding of participants and personnel (performance bias) Subjective outcomes | Low risk | Quote: "Patients, treating physicians, data collectors and statisticians were blinded to treatment allocation." Quote: "The appearance, size, weight and taste of the placebo tablets were identical to those of the 10‐mg baclofen tablets and the scored tablets were provided in identical containers." |
| Blinding of outcome assessment (detection bias) Objective outcomes | Low risk | Quote: "Patients, treating physicians, data collectors and statisticians were blinded to treatment allocation." |
| Blinding of outcome assessment (detection bias) Subjective outcomes | Low risk | Quote: "Patients, treating physicians, data collectors and statisticians were blinded to treatment allocation." |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Quote: "Efficacy analyses were performed in the intent‐to‐treat (ITT) population (i.e. all randomized patients were analyzed in their randomization arm whatever the treatment actually received or any other deviation to the protocol). Safety analyses were performed in the ITT population and in the safety population, in which patients received baclofen whatever their randomization arm in the ‘baclofen’ group. We used multiple imputation of missing alcohol consumption during follow‐up." |
| Selective reporting (reporting bias) | Unclear risk | Study protocol unavailable. |
ADS: Alcohol Dependence Scale; ALT: alanine aminotransferase; AST: aspartate aminotransferase; AUD: alcohol use disorder; AUDIT‐C: Alcohol Use Disorders Identification Test; BBCET: brief behavioral compliance enhancement treatment; BDI: Beck's Depression Inventory; BSI: Brief Symptom Inventory; CBT: cognitive behavioural therapy; CDT: carbohydrate‐deficient transferrin; CGI‐I: Clinical Global Impression – Improvement; CGI‐S: Clinical Global Impression – Severity; DSM‐IV: Diagnostic and Statistical Manual of Mental Disorders, 4th edition; DSM‐IV‐TR: Diagnostic and Statistical Manual of Mental Disorders, 4th revision (text revision); DSM‐5: Diagnostic and Statistical Manual of Mental Disorders, 5th edition; GGT: gamma‐glutamyl transferase; HCV: hepatitis C virus; HDD: heavy drinking day; ICD: International Classification of Diseases; IQR: interquartile range; N/A: not applicable; NIH: National Institutes of Health; NR: not reported; OCDS: Obsessive Compulsive Drinking Scale; RCT: randomised controlled trial; SD: standard deviation; SDU: standard drink unit; STAI: State‐Trait Anxiety Inventory; SUD: substance use disorder; TAC: total alcohol consumption; ULN: upper limit of normal.
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Addolorato 2002 | Duration of study < 12 weeks. |
| Flannery 2004 | Study design not in the inclusion criteria: uncontrolled preliminary study on 14 participants. |
| Gupta 2017 | Benfothiamine used as inactive medication for the control group has been found to have an effect on both alcohol consumption and anxiety (Manzardo 2013; Manzardo 2015). |
| Jose 2019 | Study duration < 12 weeks. |
| Kumar 2020b | Both groups received baclofen. |
| Leggio 2011 | Study objective not in the inclusion criteria: human laboratory study to investigate putative behavioural mechanisms by which baclofen reduces drinking. |
| Leggio 2013 | Study objective not in the inclusion criteria: human laboratory study to investigate putative behavioural mechanisms by which baclofen reduces drinking. |
Characteristics of studies awaiting classification [ordered by study ID]
Karthik 2018.
| Methods | |
| Participants | 42 participants with severe alcohol dependence, after detoxification |
| Interventions | Baclofen 60–80 mg/day (16 participants) vs naltrexone 50–100 mg/day (16 participants) |
| Outcomes | Carving, abstinence |
| Notes | Conference abstract; insufficient information provided about study design and length of follow‐up. |
Sharma 2012.
| Methods | Randomised, double‐blind, controlled trial |
| Participants | 54 participants |
| Interventions | Baclofen 10 mg/day vs 12 mg/day for 12 weeks (18 participants) vs placebo (18 participants) |
| Outcomes | Daily number of drinks |
| Notes | Conference abstract without enough usable data |
Characteristics of ongoing studies [ordered by study ID]
CTRI/2011/11/002154.
| Study name | CTRI/2011/11/002154 |
| Methods | Randomised, phase III, parallel‐group, placebo‐controlled trial |
| Participants | 180 men or women aged 21−75 years, meeting DSM‐IV‐TR criteria for current AD |
| Interventions | Baclofen vs placebo |
| Outcomes |
Safety variables
|
| Starting date | 24 December 2011 |
| Contact information | shravanti.bhowmik@sparcmail.com |
| Notes |
AD: alcohol dependence; DSM‐IV‐TR: Diagnostic and Statistical Manual of Mental Disorders, 4th revision (text revision); HDD: heavy drinking days.
Differences between protocol and review
The updated review differs from the previous version (Minozzi 2018) and the protocol (Minozzi 2017) for the following reasons.
We did not investigate 'Cumulative abstinence duration' as planned as a secondary outcome in the protocol in either the previous version or updated review because we evaluated the rate of abstinent days as a primary outcome considered that the cumulative abstinence duration would add no further information.
We added one study that was previously awaiting classification to the included studies section (Addolorato 2011‐LD; Addolorato 2011‐MD). This was a post‐hoc analysis and data were unavailable despite us requesting them from the study authors for the previous version of this review. However, for this update, the authors allowed us to use the original database (see Included studies and Characteristics of included studies table for further details).
In the previous version of this review, none of the subgroup analyses planned in the protocol were conducted because of the limited number of included studies; in this update, the larger number of included studies allowed us to perform one of the planned subgroup analyses (i.e. the daily doses of baclofen).
In this update, we did not perform the other two subgroup analyses planned in the protocol (i.e. according to the presence of concomitant substance use disorders and other comorbid psychiatric disorders) because all studies excluded people with substance use disorders other than alcohol or nicotine except for one study that recruited participants dependent on both alcohol and nicotine (Leggio 2015). In addition, all studies excluded people with comorbid severe mental disorders.
In this update, we performed subgroup analyses according to other variables (i.e. the duration of the studies and the consumption of alcohol at the beginning of treatment) (see Why it is important to do this review; Subgroup analysis and investigation of heterogeneity for further details).
In the previous version of this review, we combined data of multiple‐armed studies (e.g. two different doses of baclofen) into a single group and compared it to the placebo group. In this update, we did not combine the data of multiple‐armed studies preferring to compare them each with placebo in the meta‐analyses. To do so we divided the control group into two different subgroups to avoid double counting of participants in the control group (see Included studies; Subgroup analysis and investigation of heterogeneity for further details).
Contributions of authors
RA and SR wrote the background section.
SM, RS and RA wrote the methods section.
RA, SM and RS conducted screening, extracted data, completed the risk of bias assessment and undertook data analysis.
All review authors contributed to writing the discussion and conclusion sections and revised the final report.
Sources of support
Internal sources
-
Department of Epidemiology, Lazio Regional Health Service, Rome, Italy
Sources of support
External sources
No sources of support provided
Declarations of interest
RA: none.
RS: none.
SR: none.
SM: none.
New search for studies and content updated (conclusions changed)
References
References to studies included in this review
Addolorato 2007 {published and unpublished data}
- Addolorato G, Leggio L, Ferrulli A, Cardone S, Vonghia L, Mirijello A, et al. Effectiveness and safety of baclofen for maintenance of alcohol abstinence in alcohol-dependent patients with liver cirrhosis: randomised, double-blind controlled study. Lancet 2007;370(9603):1915-22. [DOI: 10.1016/S0140-6736(07)61814-5] [DOI] [PubMed] [Google Scholar]
Addolorato 2011‐LD {published and unpublished data}
- Addolorato G, Leggio L, Ferrulli A, Cardone S, Bedogni G, Caputo F, et al. Dose–response effect of baclofen in reducing daily alcohol intake in alcohol dependence: secondary analysis of a randomized, double-blind, placebo-controlled trial. Alcohol and Alcoholism 2011;46(3):312-7. [PMID: ] [DOI] [PubMed] [Google Scholar]
Addolorato 2011‐MD {published and unpublished data}
- Addolorato G, Leggio L, Ferrulli A, Cardone S, Bedogni G, Caputo F, et al. Dose–response effect of baclofen in reducing daily alcohol intake in alcohol dependence: secondary analysis of a randomized, double-blind, placebo-controlled trial. Alcohol and Alcoholism 2011;46(3):312-7. [PMID: ] [DOI] [PubMed] [Google Scholar]
Beraha 2016‐HD {published data only}
- Beraha EM, Salemink E, Goudriaan AE, Bakker A, De Jong D, Smits N, et al. Efficacy and safety of high-dose baclofen for the treatment of alcohol dependence: a multicentre, randomised, double-blind controlled trial. European Neuropsychopharmacology 2017;26(12):1950-9. [DOI] [PubMed] [Google Scholar]
- Beraha EM, Salemink E, Krediet E, Wiers RW. Can baclofen change alcohol-related cognitive biases and what is the role of anxiety herein? Journal of Psychopharmacology 2018;32(8):867-75. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Beraha 2016‐LD {published data only}
- Beraha EM, Salemink E, Goudriaan AE, Bakker A, De Jong D, Smits N, et al. Efficacy and safety of high-dose baclofen for the treatment of alcohol dependence: a multicentre, randomised, double-blind controlled trial. European Neuropsychopharmacology 2017;26(12):1950-9. [DOI] [PubMed] [Google Scholar]
- Beraha EM, Salemink E, Krediet E, Wiers RW. Can baclofen change alcohol-related cognitive biases and what is the role of anxiety herein? Journal of Psychopharmacology 2018;32(8):867-75. [DOI] [PMC free article] [PubMed] [Google Scholar]
Garbutt 2010a {published data only}
- Garbutt JC, Kampov-Polevoy AB, Gallop R, Kalka-Juhl L, Flannery BA. Efficacy and safety of baclofen for alcohol dependence: a randomized, double-blind, placebo-controlled trial. Alcoholism, Clinical and Experimental Research 2010;34(11):1849-57. [DOI: 10.1111/j.1530-0277.2010.01273.x] [DOI] [PMC free article] [PubMed] [Google Scholar]
Garbutt 2010b1 {published data only}
- Garbutt JC, Kalka-Juhl L, Kampov-Polevoy AB, Wells S, Nicholas L, Gallop R, et al. Feasibility and tolerability of a combination of naltrexone and baclofen for alcohol dependence: a pilot study. Alcoholism: Clinical and Experimental Research 2010;34(6):178A. [DOI] [PMC free article] [PubMed] [Google Scholar]
Garbutt 2010b2 {published data only}
- Garbutt JC, Kalka-Juhl L, Kampov-Polevoy AB, Wells S, Nicholas L, Gallop R, et al. Feasibility and tolerability of a combination of naltrexone and baclofen for alcohol dependence: a pilot study. Alcoholism: Clinical and Experimental Research 2010;34(6):178A. [DOI] [PMC free article] [PubMed] [Google Scholar]
Garbutt 2021‐LD {published and unpublished data}
- Garbutt JC, Kampov-Polevoy AB, Pedersen C, Stansbury M, Jordan R, Willing L, et al. Efficacy and tolerability of baclofen in a U.S. community population with alcohol use disorder: a dose–response, randomized, controlled trial. Neuropsychopharmacology 2021;46(13):2250-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- NCT01980706. Efficacy and safety of high-dose baclofen for alcohol dependence. www.clinicaltrials.gov/ct2/show/NCT01980706 (first received 11 November 2013).
- Pedersen CA, Kampov-Polevoi WA, Jordan R, Casey R, Tatrau J, Gallop R, et al. Efficacy and tolerability of high-dose baclofen in a U.S. community population: a randomized, placebo-controlled trial. Alcoholism: Clinical and Experimental Research 2018;42(Suppl 1):65A. [Google Scholar]
Garbutt 2021‐MD {published and unpublished data}
- Garbutt JC, Kampov-Polevoy AB, Pedersen C, Stansbury M, Jordan R, Willing L, et al. Efficacy and tolerability of baclofen in a U.S. community population with alcohol use disorder: a dose–response, randomized, controlled trial. Neuropsychopharmacology 2021;46(13):2250-6. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
- NCT01980706. Efficacy and safety of high-dose baclofen for alcohol dependence. www.clinicaltrials.gov/ct2/show/NCT01980706 (first received 11 November 2013).
- Pedersen CA, Kampov-Polevoi WA, Jordan R, Casey R, Tatrau J, Gallop R, et al. Efficacy and tolerability of high-dose baclofen in a U.S. community population: a randomized, placebo-controlled trial. Alcoholism: Clinical and Experimental Research 2018;42(Suppl 1):65A. [Google Scholar]
Hauser 2017 {published data only}
- Hauser P, Fuller B Ho SB, Thuras P, Kern S, Dieperink E. The safety and efficacy of baclofen to reduce alcohol use in veterans with chronic hepatitis C: a randomized controlled trial. Addiction 2017;112(7):1173-83. [DOI: 10.1111/add.13787.] [DOI] [PubMed] [Google Scholar]
Krupitskii 2017 {published data only}
- Krupitskii EM, Rybakova KV, Kiselev AS, Alekseeva YV, Berntsev VA, Chekhlatyi EI, et al. Efficacy and safety of the use of baclofen in the treatment of alcohol dependent (a double-blind, randomized, placebo-controlled pilot study). Neuroscience and Behavioral Physiology 2017;47(2):153-62. [DOI: 10.1007/s11055-016-0379-6] [DOI] [Google Scholar]
- Krupitsky EM, Rybakova KV, Kiselev AS, Alexeeva YV, Berntsev VA, Chekhlaty EI, et al. Double blind placebo controlled randomized pilot clinical trial of baclofen (Baclosan®) for alcohol dependence. Zhurnal Nevrologii i Psikhiatrii Imeni S.S. Korsakova 2015;115(6):53-62. [DOI: 10.17116/jnevro20151156153-62] [DOI] [PubMed] [Google Scholar]
Kumar 2020a {published data only}
- Kumar A, Sharma A, Bansal PD, Bahetra M, Gill HK, Kumar R. A comparative study on the safety and efficacy of naltrexone versus baclofen versus acamprosate in the management of alcohol dependence. Indian Journal of Psychiatry 2020;62(6):650-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Leggio 2015 {published and unpublished data}
- Bollinger J, Edwards SM, Zywiak WK, Tidey JW, Swift RW, Kenna GA, et al. A double-blind, placebo-controlled randomized pilot study of the effects of baclofen on alcohol-tobacco co-use and alcohol cue-reactivity in alcoholic smokers. Clinical and Translational Science 2014;7(3):265. [Google Scholar]
- Farokhnia M, Edwards SM, Bollinger J, Amodio J, Zywiak WH, Tidey JW, et al. Baclofen as a pharmacotherapy for the treatment of concurrent alcohol and nicotine dependence: a double-blind, placebo-controlled, randomized trial. Erratum for: Neuropsychopharmacology. 2014 Dec;39 Suppl 1:S1-647. Neuropsychopharmacology 2015;40(6):1560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farokhnia M, Edwards SM, Bollinger J, Amodio J, Zywiak WH, Tidey JW. Baclofen as a pharmacotherapy for the treatment of concurrent alcohol and nicotine dependence: a double-blind, placebo-controlled, randomized trial. Neuropsychopharmacology 2014;39:S340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leggio L, Zywiak WH, Edwards SM, Tidey JW, Swift RM, Kenna GA. A preliminary double-blind, placebo-controlled randomized study of baclofen effects in alcoholic smokers. Psychopharmacology 2015;232(1):233-43. [DOI: 10.1007/s00213-014-3652-9] [DOI] [PMC free article] [PubMed] [Google Scholar]
Mishra 2010 {published data only}
- Mishra SN, Swain SP, Shukla RK, Sarkar P. A study of comparative efficacy of baclofen vs acamprosate in reducing alcohol craving and abuse. Orissa Journal of Psychiatry 2010;-:48-53. [Google Scholar]
Morley 2014‐LD {published data only}
- Morley KC, Baillie A, Leung S, Addolorato G, Leggio L, Haber PS. Baclofen for the treatment of alcohol dependence and possible role of comorbid anxiety. Alcohol and Alcoholism 2014;49(6):654-60. [DOI: 10.1093/alcalc/agu062] [DOI] [PMC free article] [PubMed] [Google Scholar]
Morley 2014‐MD {published data only}
- Morley KC, Baillie A, Leung S, Addolorato G, Leggio L, Haber PS. Baclofen for the treatment of alcohol dependence and possible role of comorbid anxiety. Alcohol and Alcoholism 2014;49(6):654-60. [DOI: 10.1093/alcalc/agu062] [DOI] [PMC free article] [PubMed] [Google Scholar]
Morley 2018‐LD {published and unpublished data}
- Heng S, Jamshidi N, Baillie A, Louie E, Dore G, Phung N, et al. Baclofen response in alcohol dependent patients concurrently receiving antidepressants: secondary analysis from the BacALD study. Frontiers in Psychiatry 2018;9:576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Logge W, Baillie AJ, Morris RW, Haber PS, Morley KC. Baclofen reduces alcohol cue-induced fMRI brain activation in treatment seeking alcohol dependent individuals. Alcoholism: Clinical and Experimental Research 2018;42(Suppl 1):65A. [Google Scholar]
- Logge WB, Baillie AJ, Haber PS, Morley KC. Baclofen modulates cardiovascular responses to appetitive cues in treatment-seeking alcohol use disorder individuals. Human Psychopharmacology 2020;35(2):e2722. [DOI] [PubMed] [Google Scholar]
- Logge WB, Haber PS, Baillie AJ, Kosinski P, Morris RW, Fraser I, et al. Dose-specific baclofen effects on brain structure, function and treatment responses: neuroimaging findings from the BacALD trial. Alcoholism: Clinical and Experimental Research 2021;45(Suppl 1):20A-21A. [Google Scholar]
- Logge WB, Morley KC, Haber PS, Baillie AJ. Executive functioning moderates responses to appetitive cues: a study in severe alcohol use disorder and alcoholic liver disease. Alcohol and Alcoholism 2019;54(1):38-46. [DOI] [PubMed] [Google Scholar]
- Logge WB, Morris RW, Baillie AJ, Haber PS, Morley KC. Baclofen attenuates fMRI alcohol cue reactivity in treatment-seeking alcohol dependent individuals. Psychopharmacology (Berlin) 2021;238(5):1291-302. [DOI] [PubMed] [Google Scholar]
- Morley K, Luquin N, Lagopoulos J, Logge W, Baillie A, Haber PS. Baclofen for treatment of alcohol use disorder: secondary analyses and implications of the Australian BacALD trial. Drug and Alcohol Review 2019;38:S75. [Google Scholar]
- Morley K, Luquin N, Lagopoulos J, Logge W, Baillie A, Haber PS. Exploration of the modulating role of the GABAb receptor polymorphism rs29220 on response to baclofen in alcohol dependent patients. Alcoholism: Clinical and Experimental Research 2021;45(Suppl 1):41A. [Google Scholar]
- Morley KC, Baillie A, Fraser I, Furneaux-Bate A, Dore G, Roberts M, et al. Baclofen in the treatment of alcohol dependence with or without liver disease: multisite, randomised, double-blind, placebo-controlled trial. British Journal of Psychiatry 2018;212(6):362-9. [DOI] [PubMed] [Google Scholar]
- Morley KC, Lagopoulos J, Logge W, Baillie A, Adams C, Haber PS. Brain GABA levels are reduced in alcoholic liver disease: a proton magnetic resonance spectroscopy study. Addiction Biology 2020;25(1):e12702. [DOI] [PubMed] [Google Scholar]
- Morley KC, Leung S, Baillie A, Haber PS. The efficacy and biobehavioural basis of baclofen in the treatment of alcoholic liver disease (BacALD): study protocol for a randomised controlled trial. Contemporary Clinical Trials 2013;36(2):348-55. [DOI] [PubMed] [Google Scholar]
- Morley KC, Logge WB, Fraser I, Morris RW, Baillie AJ, Haber PS. High-dose baclofen attenuates insula activation during anticipatory anxiety in treatment-seeking alcohol dependant individuals: preliminary findings from a pharmaco-fMRI study. European Neuropsychopharmacology 2021;46:28-36. [DOI] [PubMed] [Google Scholar]
- Morley KC, Luquin N, Baillie A, Fraser I, Trent RJ, Dore G, et al. Moderation of baclofen response by a GABAB receptor polymorphism: results from the BacALD randomized controlled trial. Addiction 2018;113(12):2205-13. [DOI] [PubMed] [Google Scholar]
- Morley KC, Morris R, Logge W, Baillie A, Fraser I, Haber PS. Baclofen administration attenuates insula response during anticipatory anxiety in alcohol dependent patients. Alcoholism: Clinical and Experimental Research 2018;42(Suppl 1):65A. [Google Scholar]
- Morley KC, Riordan B, Logge W, Baillie A, Haber PS. Moderation of baclofen on the daily relationship between craving and alcohol use: results from the Australian BacALD trial. Alcoholism: Clinical and Experimental Research 2021;45(Suppl 1):53A. [Google Scholar]
- Morley LC, Luquin N, Baillie A, Fraser I, Trent R, Dore G, et al. Moderation of baclofen response by a GABAb receptor polymorphism. Alcoholism: Clinical and Experimental Research 2018;42(Suppl 1):65A. [DOI] [PubMed] [Google Scholar]
- Rombouts SA, Baillie A, Haber PS, Morley KC. Clinical predictors of response to baclofen in the treatment of alcohol use disorder: results from the BacALD trial. Alcohol and Alcoholism 2019;54(3):272-8. [DOI] [PubMed] [Google Scholar]
Morley 2018‐MD {published and unpublished data}
- Heng S, Jamshidi N, Baillie A, Louie E, Dore G, Phung N, et al. Baclofen response in alcohol dependent patients concurrently receiving antidepressants: secondary analysis from the BacALD study. Frontiers in Psychiatry 2018;9:576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Logge W, Baillie AJ, Morris RW, Haber PS, Morley KC. Baclofen reduces alcohol cue-induced fMRI brain activation in treatment seeking alcohol dependent individuals. Alcoholism: Clinical and Experimental Research 2018;42(Suppl 1):65A. [Google Scholar]
- Logge WB, Baillie AJ, Haber PS, Morley KC. Baclofen modulates cardiovascular responses to appetitive cues in treatment-seeking alcohol use disorder individuals. Human Psychopharmacology 2020;35(2):e2722. [DOI] [PubMed] [Google Scholar]
- Logge WB, Haber PS, Baillie AJ, Kosinski P, Morris RW, Fraser I, et al. Dose-specific baclofen effects on brain structure, function and treatment responses: neuroimaging findings from the BacALD trial. Alcoholism: Clinical and Experimental Research 2021;45(Suppl 1):20A-21A. [Google Scholar]
- Logge WB, Morley KC, Haber PS, Baillie AJ. Executive functioning moderates responses to appetitive cues: a study in severe alcohol use disorder and alcoholic liver disease. Alcohol and Alcoholism 2019;54(1):38-46. [DOI] [PubMed] [Google Scholar]
- Logge WB, Morris RW, Baillie AJ, Haber PS, Morley KC. Baclofen attenuates fMRI alcohol cue reactivity in treatment-seeking alcohol dependent individuals. Psychopharmacology (Berlin) 2021;238(5):1291-302. [DOI] [PubMed] [Google Scholar]
- Morley K, Luquin N, Lagopoulos J, Logge W, Baillie A, Haber PS. Baclofen for treatment of alcohol use disorder: secondary analyses and implications of the Australian BacALD trial. Drug and Alcohol Review 2019;38:S75. [Google Scholar]
- Morley K, Luquin N, Lagopoulos J, Logge W, Baillie A, Haber PS. Exploration of the modulating role of the GABAb receptor polymorphism rs29220 on response to baclofen in alcohol dependent patients. Alcoholism: Clinical and Experimental Research 2021;45(Suppl 1):41A. [Google Scholar]
- Morley KC, Baillie A, Fraser I, Furneaux-Bate A, Dore G, Roberts M, et al. Baclofen in the treatment of alcohol dependence with or without liver disease: multisite, randomised, double-blind, placebo-controlled trial. British Journal of Psychiatry 2018;212(6):362-9. [DOI] [PubMed] [Google Scholar]
- Morley KC, Lagopoulos J, Logge W, Baillie A, Adams C, Haber PS. Brain GABA levels are reduced in alcoholic liver disease: a proton magnetic resonance spectroscopy study. Addiction Biology 2020;25(1):e12702. [DOI] [PubMed] [Google Scholar]
- Morley KC, Leung S, Baillie A, Haber PS. The efficacy and biobehavioural basis of baclofen in the treatment of alcoholic liver disease (BacALD): study protocol for a randomised controlled trial. Contemporary Clinical Trials 2013;36(2):348-55. [DOI] [PubMed] [Google Scholar]
- Morley KC, Logge WB, Fraser I, Morris RW, Baillie AJ, Haber PS. High-dose baclofen attenuates insula activation during anticipatory anxiety in treatment-seeking alcohol dependant individuals: preliminary findings from a pharmaco-fMRI study. European Neuropsychopharmacology 2021;46:28-36. [DOI] [PubMed] [Google Scholar]
- Morley KC, Luquin N, Baillie A, Fraser I, Trent RJ, Dore G, et al. Moderation of baclofen response by a GABAB receptor polymorphism: results from the BacALD randomized controlled trial. Addiction 2018;113(12):2205-13. [DOI] [PubMed] [Google Scholar]
- Morley KC, Morris R, Logge W, Baillie A, Fraser I, Haber PS. Baclofen administration attenuates insula response during anticipatory anxiety in alcohol dependent patients. Alcoholism: Clinical and Experimental Research 2018;42(Suppl 1):65A. [Google Scholar]
- Morley KC, Riordan B, Logge W, Baillie A, Haber PS. Moderation of baclofen on the daily relationship between craving and alcohol use: results from the Australian BacALD trial. Alcoholism: Clinical and Experimental Research 2021;45(Suppl 1):53A. [Google Scholar]
- Rombouts SA, Baillie A, Haber PS, Morley KC. Clinical predictors of response to baclofen in the treatment of alcohol use disorder: results from the BacALD Trial. Alcohol and Alcoholism 2019;54(3):272-8. [DOI] [PubMed] [Google Scholar]
Muller 2015 {published data only}
- Beck A, Pelz P, Lorenz RC, Charlet K, Geisel O, Heinz A, et al. Effects of high-dose baclofen on cue reactivity in alcohol dependence: a randomized, placebo-controlled pharmaco-fMRI study. European Neuropsychopharmacology 2018;28(11):1206-16. [DOI] [PubMed] [Google Scholar]
- Geisel O, Hellweg R, Wernecke KD, Wiedemann K, Müller CA. Total and acylated ghrelin plasma levels as potential long-term response markers in alcohol-dependent patients receiving high-dose of the GABA-B receptor agonist baclofen. Psychiatry Research 2019;272:431-7. [DOI] [PubMed] [Google Scholar]
- Geisel O, Schlemm L, Hellweg R, Wiedemann K, Müller CA. Hypothalamic-pituitary-adrenocortical axis activity in alcohol-dependent patients during treatment with high-dose baclofen. Pharmacopsychiatry 2019;52(1):32-7. [DOI] [PubMed] [Google Scholar]
- Muller CA, Geisel O, Pelz P, Higl V, Kruger J, Stickel A, et al. High-dose baclofen for the treatment of alcohol dependence (BACLAD study): a randomized, placebo-controlled trial. European Neuropsychopharmacology 2015;25(8):1167-77. [DOI: 10.1016/j.euroneuro.2015.04.002] [DOI] [PubMed] [Google Scholar]
- Muller CA, Geisel O, Pelz P, Higl V, Kruger J, Stickel A, et al. Individually titrated high-dose baclofen for the treatment of alcohol dependence (BACLAD study) – a randomized controlled trial. European Psychiatry 2015;30(S1):1611. [DOI: 10.1016/S0924-9338(15)31243-8] [DOI] [PubMed] [Google Scholar]
- Pelz P, Genauck A, Gleich T, Lorenz RC, Heinz A, Muller CA, et al. Baclofen desensitizes the fronto-striatal reward network in alcohol dependent patients: a randomized, placebo-controlled pharmaco FMRI trial. Alcoholism: Clinical and Experimental Research 2018;42:126A. [Google Scholar]
- Pelz P, Genauck A, Lorenz RC, Wustenberg C, Wackerhagen K, Charlet T, et al. Baclofen desensitizes insular gain anticipation in alcohol-dependent patients – a randomized, placebo-controlled, patient-tailored pharmaco fMRI trial. Alcoholism: Clinical and Experimental Research 2019;43(Suppl 1):246A. [Google Scholar]
Ponizovsky 2015 {published data only}
- Ponizovsky AM, Rosca P, Aronovich E, Weizman A, Grinshpoon A. Baclofen as add-on to standard psychosocial treatment for alcohol dependence: a randomized, double-blind, placebo-controlled trial with 1 year follow-up. Journal of Substance Abuse Treatment 2015;52:24-30. [DOI: 10.1016/j.jsat.2014.11.007] [DOI] [PubMed] [Google Scholar]
Reynaud 2017 {published data only}
- Pouchain D. A randomized, placebo-controlled study of high-dose baclofen in alcohol-dependent patients. The Alpadir study. Exercer-La Revue Francophone De Medecine Generale 2018;142:163-5. [Google Scholar]
- Reynaud M, Aubin HJ, Trinquet F, Zakine B, Dano C, Dematteis M, et al. A randomized, placebo-controlled study of high-dose baclofen in alcohol-dependent patients – the ALPADIR Study. Alcohol and Alcoholism 2017;52(4):439-46. [DOI] [PubMed] [Google Scholar]
Rigal 2020 {published and unpublished data}
- Jaury PH. Baclofen for the treatment of alcohol drinkers (Bacloville). Alcoholism: Clinical and Experimental Research 2014;38:215a. [Google Scholar]
- Rigal L, Sidorkiewicz S, Tréluyer JM, Perrodeau E, Le Jeunne C, Porcher R, et al. Titrated baclofen for high-risk alcohol consumption: a randomized placebo-controlled trial in out-patients with 1-year follow-up. Addiction 2020;115(7):1265-76. [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
Addolorato 2002 {published data only}
- Addolorato G, Caputo F, Capristo E, Domenicali M, Bernardi M, Janiri M, et al. Baclofen efficacy in reducing alcohol craving and intake: a preliminary double-blind randomised controlled study. Alcohol & Alcoholism 2002;37:504-8. [DOI] [PubMed] [Google Scholar]
Flannery 2004 {published data only}
- Flannery BA, Garbutt JC, Cody MW, Renn W, Grace K, Osborne M, et al. Baclofen for alcohol dependence: a preliminary open-label study. Alcoholism, Clinical and Experimental Research 2004;28(10):1517-23. [DOI] [PubMed] [Google Scholar]
Gupta 2017 {published data only}
- Gupta M, Verma P, Rastogi R, Arora S, Elwadhi D. Randomized open-label trial of baclofen for relapse prevention in alcohol dependence. American Journal of Drug and Alcohol Abuse 2017;43(3):324-31. [PMID: ] [DOI] [PubMed] [Google Scholar]
Jose 2019 {published data only}
- CTRI/2016/01/006525. Comparison of baclofen and topiramate in the treatment of alcohol dependence. ctri.nic.in/Clinicaltrials/pdf_generate.php?trialid=13646 (first received 27 March 2012).
- Jose NA, Yadav P, Kapoor A, Mahla VP. Comparison between baclofen and topiramate in alcohol dependence: a prospective study. Indian Journal of Psychiatry 2019;28(1):44-50. [DOI] [PMC free article] [PubMed] [Google Scholar]
Kumar 2020b {published data only}
- Kumar R, Kumar KJ, Benegal V, Roopesh BN, Ravi GS. Integrated intervention program for alcoholism improves impulsiveness and disadvantageous reward processing/risk-taking. Indian Journal of Psychiatry 2020;62(4):384-91. [DOI] [PMC free article] [PubMed] [Google Scholar]
Leggio 2011 {published data only}
- Leggio L, Kenna G, Zywiak W, Edwards S, Fricchione S, Taveres T. Baclofen as a novel pharmacotherapy for alcohol dependence: preliminary findings from a human laboratory double-blind placebo-controlled randomized study. Neuropsychopharmacology 2011;36:187. [Google Scholar]
Leggio 2013 {published data only}
- Leggio L, Zywiak WH, McGeary JE, Edwards S, Fricchione SR, Shoaff JR, et al. A human laboratory pilot study with baclofen in alcoholic individuals. Pharmacology, Biochemistry, and Behavior 2013;103(4):784-91. [DOI: 10.1016/j.pbb.2012.11.013] [DOI] [PMC free article] [PubMed] [Google Scholar]
References to studies awaiting assessment
Karthik 2018 {published data only}
- Karthik S, Holla B, Bharath RD, Venkatasubramanian G, Benegal V. Baclofen is more efficacious than oral naltrexone in individuals with severe alcohol dependence for maintaining abstinence – a preliminary report. European Psychiatry 2018;48(Suppl 1):S335. [Google Scholar]
Sharma 2012 {published data only}
- Sharma V, Sharma R, Chaturvedi M, Bharadwaj B. A dose–response effect of baclofen in reducing daily alcohol intake in alcohol-dependent subjects. Journal of Gastroenterology and Hepatology 2012;27:263. [DOI: 10.1111/jgh.12006] [DOI] [Google Scholar]
References to ongoing studies
CTRI/2011/11/002154 {published data only}
- CTRI/2011/11/002154. Efficacy and safety of baclofen GRS for treatment of alcohol dependence, a 12 week study. ctri.nic.in/Clinicaltrials/pmaindet2.php?trialid=3752 (first received 21 November 2011).
Additional references
Agabio 2007
- Agabio R, Marras P, Addolorato G, Carpiniello B, Gessa GL. Baclofen suppresses alcohol intake and craving for alcohol in a schizophrenic alcohol-dependent patient: a case report. Journal of Clinical Psychopharmacology 2007;27(3):319-20. [DOI] [PubMed] [Google Scholar]
Agabio 2013
- Agabio R, Preti A, Gessa GL. Efficacy and tolerability of baclofen in substance use disorders: a systematic review. European Addiction Research 2013;19(6):325-45. [DOI] [PubMed] [Google Scholar]
Agabio 2014
- Agabio R, Colombo G. GABAB receptor ligands for the treatment of alcohol use disorder: preclinical and clinical evidence. Frontiers in Neuroscience 2014;8:140. [DOI: 10.3389/fnins.2014.00140] [DOI] [PMC free article] [PubMed] [Google Scholar]
Agabio 2016
- Agabio R, Pani PP, Preti A, Gessa GL, Franconi F. Efficacy of medications approved for the treatment of alcohol dependence and alcohol withdrawal syndrome in female patients: a descriptive review. European Addiction Research 2016;22(1):1-16. [PMID: ] [DOI] [PubMed] [Google Scholar]
Agabio 2018a
- Agabio R, Sinclair JM, Addolorato G, Aubin HJ, Beraha EM, Caputo F, et al. Baclofen for the treatment of alcohol use disorder: the Cagliari Statement. Lancet Psychiatry 2018;5(12):957-60. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Agabio 2018b
- Agabio R, Leggio L. Baclofen in the treatment of patients with alcohol use disorder and other mental health disorders. Frontiers in Psychiatry 2018;9:464. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Agabio 2021a
- Agabio R, Balia S, Gessa GL, Pani PP. Use of medications for the treatment of alcohol dependence: a retrospective study conducted in 2011–2012. Current Drug Research Reviews 2021;13(2):154-64. [PMID: ] [DOI] [PubMed] [Google Scholar]
Agabio 2021b
- Agabio R, Baldwin DS, Amaro H, Leggio L, Sinclair JM. The influence of anxiety symptoms on clinical outcomes during baclofen treatment of alcohol use disorder: a systematic review and meta-analysis. Neuroscience & Biobehavioral Reviews 2021;125:296-313. [PMID: ] [DOI] [PubMed] [Google Scholar]
Amato 2010
- Amato L, Minozzi S, Vecchi S, Davoli M. Benzodiazepines for alcohol withdrawal. Cochrane Database of Systematic Reviews 2010, Issue 3. Art. No: CD005063. [DOI: 10.1002/14651858.CD005063.pub3] [DOI] [PMC free article] [PubMed] [Google Scholar]
Amato 2011
- Amato L, Minozzi S, Davoli M. Efficacy and safety of pharmacological interventions for the treatment of the alcohol withdrawal syndrome. Cochrane Database of Systematic Reviews 2011, Issue 6. Art. No: CD008537. [DOI: 10.1002/14651858.CD008537.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Ameisen 2005
- Ameisen O. Complete and prolonged suppression of symptoms and consequences of alcohol-dependence using high-dose baclofen: a self-case report of a physician. Alcohol and Alcoholism 2005;40(2):147-50. [DOI] [PubMed] [Google Scholar]
Andrade 2020
- Andrade C. Individualized, high-dose baclofen for reduction in alcohol intake in persons with high levels of consumption. Journal of Clinical Psychiatry 2020;81(4):20f13606. [PMID: ] [DOI] [PubMed] [Google Scholar]
APA 1980
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 3rd edition. Washington (DC): American Psychiatric Association, 1980. [Google Scholar]
APA 1987
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 3rd Revised edition. Washington (DC): American Psychiatric Association, 1987. [Google Scholar]
APA 2000
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 4th edition. Washington (DC): American Psychiatric Association, 2000. [Google Scholar]
APA 2013
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th edition. Washington (DC): American Psychiatric Association, 2013. [Google Scholar]
Brennan 2013
- Brennan JL, Leung JG, Gagliardi JP, Rivelli SK, Muzyk AJ. Clinical effectiveness of baclofen for the treatment of alcohol dependence: a review. Clinical Pharmacology 2013;5:99-107. [DOI: 10.2147/CPAA.S32434] [DOI] [PMC free article] [PubMed] [Google Scholar]
Bschor 2018
- Bschor T, Henssler J, Müller M, Baethge C. Baclofen for alcohol use disorder – a systematic meta-analysis. Acta Psychiatrica Scandinavica 2018;138(3):232-42. [PMID: ] [DOI] [PubMed] [Google Scholar]
Bucknam 2006
- Bucknam W. Suppression of symptoms of alcohol dependence and craving using high-dose baclofen. Alcohol and Alcoholism 2006;2(42):158-60. [DOI] [PubMed] [Google Scholar]
Burnette 2022
- Burnette EM, Nieto SJ, Grodin EN, Meredith LR, Hurley B, Miotto K, et al. Novel agents for the pharmacological treatment of alcohol use disorder. Drugs 2022;82(3):251-74. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Carvalho 2019
- Carvalho AF, Heilig M, Perez A, Probst C, Rehm J. Alcohol use disorders. Lancet 2019;394(10200):781-92. [PMID: ] [DOI] [PubMed] [Google Scholar]
Chick 2012
- Chick J, Nutt DJ. Substitution therapy for alcoholism: time for a reappraisal? Journal of Psychopharmacology 2012;26(2):205-12. [DOI] [PubMed] [Google Scholar]
Colombo 2003
- Colombo G, Vacca G, Serra S, Brunetti G, Carai MA, Gessa GL. Baclofen suppresses motivation to consume alcohol in rats. Psychopharmacology 2003;167(3):221-4. [DOI] [PubMed] [Google Scholar]
Colombo 2018
- Colombo G, Gessa GL. Suppressing effect of baclofen on multiple alcohol-related behaviors in laboratory animals. Frontiers in Psychiatry 2018;9:475. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Covidence [Computer program]
- Covidence. Melbourne, Australia: Veritas Health Innovation, accessed 30 January 2018. Available at www.covidence.org.
Cryan 2005
- Cryan JF, Kaupmann K. Don't worry 'B' happy!: a role for GABA(B) receptors in anxiety and depression. Trends in Pharmacological Sciences 2005;26:36-43. [DOI] [PubMed] [Google Scholar]
Dario 2004
- Dario A, Tomei G. A benefit–risk assessment of baclofen in severe spinal spasticity. Drug Safety 2004;27(11):799-818. [DOI] [PubMed] [Google Scholar]
de Beaurepaire 2018
- Beaurepaire R. A review of the potential mechanisms of action of baclofen in alcohol use disorder. Frontiers in Psychiatry 2018;9:506. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
de Beaurepaire 2019
- Beaurepaire R, Sinclair JM, Heydtmann M, Addolorato G, Aubin HJ, Beraha EM, et al. The use of baclofen as a treatment for alcohol use disorder: a clinical practice perspective. Frontiers in Psychiatry 2019;9:708. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Deeks 2017
- Deeks JJ, Higgins JP, Altman DG, editor(s) on behalf of the Cochrane Statistical Methods Group. Chapter 9: Analysing data and undertaking meta-analyses. In: Higgins JP, Churchill R, Chandler J, Cumpston MS, editor(s), Cochrane Handbook for Systematic Reviews of Interventions Version 5.2.0 (updated June 2017), Cochrane, 2017. Available from training.cochrane.org/handbook/archive/v5.2.
Farokhnia 2014
- Farokhnia M, Edwards SM, Bollinger J, Amodio J, Zywiak WH, Tidey JW. Baclofen as a pharmacotherapy for the treatment of concurrent alcohol and nicotine dependence: a double-blind, placebo-controlled, randomized trial. Neuropsychopharmacology 2014;39:S340. [DOI] [PMC free article] [PubMed] [Google Scholar]
Fond 2021
- Fond G, Nemani K, Etchecopar-Etchart D, Loundou A, Goff DC, Lee SW, et al. Association between mental health disorders and mortality among patients with COVID-19 in 7 countries: a systematic review and meta-analysis. JAMA Psychiatry 2021;78(11):1208-17. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Furukawa 2006
- Furukawa TA, Barbui C, Cipriani A, Brambilla P, Watanabe N. Imputing missing standard deviations in meta-analyses can provide accurate results. Journal of Clinical Epidemiology 2006;59:7-10. [DOI] [PubMed] [Google Scholar]
GBD 2018
- GBD 2016 Alcohol Collaborators. Alcohol use and burden for 195 countries and territories, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet 2018;392(10152):1015-35. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
GBD 2019 Cancer Risk Factors Collaborators 2022
- GBD 2019 Cancer Risk Factors Collaborators. The global burden of cancer attributable to risk factors, 2010-19: a systematic analysis for the Global Burden of Disease Study 2019. Lancet 2022;400(10352):563-91. [DOI] [PMC free article] [PubMed] [Google Scholar]
GBD 2020 Alcohol Collaborators 2022
- GBD 2020 Alcohol Collaborators. Population-level risks of alcohol consumption by amount, geography, age, sex, and year: a systematic analysis for the Global Burden of Disease Study 2020. Lancet 2022;400(10347):185-235. [DOI: 10.1016/S0140-6736(22)00847-9] [DOI] [PMC free article] [PubMed] [Google Scholar]
Gillman 2007
- Gillman MA, Lichtigfeld F, Young T. Psychotropic analgesic nitrous oxide for alcoholic withdrawal states. Cochrane Database of Systematic Reviews 2007, Issue 2. Art. No: CD005190. [DOI: 10.1002/14651858.CD005190.pub2] [DOI] [PubMed] [Google Scholar]
Glantz 2020
- Glantz MD, Bharat C, Degenhardt L, Sampson NA, Scott KM, Lim CC, et al, WHO World Mental Health Survey Collaborators. The epidemiology of alcohol use disorders cross-nationally: findings from the World Mental Health Surveys. Addictive Behaviors 2020;102:106128. [DOI: 10.1016/j.addbeh.2019.106128] [DOI] [PMC free article] [PubMed] [Google Scholar]
GRADE 2004
- GRADE Working Group. Grading quality of evidence and strength of recommendations. BMJ 2004;328(19):1490-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
GRADEpro GDT [Computer program]
- GRADEpro GDT. Version accessed 30 January 2018. Hamilton (ON): McMaster University (developed by Evidence Prime), 2015. Available at gradepro.org.
Grant 2015
- Grant BF, Goldstein RB, Saha TD, Chou SP, Jung J, Zhang H, et al. Epidemiology of DSM-5 alcohol use disorder: results From the National Epidemiologic Survey on Alcohol and Related Conditions III. JAMA Psychiatry 2015;72(8):757-66. [DOI] [PMC free article] [PubMed] [Google Scholar]
Grant 2017
- Grant BF, Chou SP, Saha TD, Pickering RP, Kerridge BT, Ruan WJ, et al. Prevalence of 12-month alcohol use, high-risk drinking, and DSM-IV alcohol use disorder in the United States, 2001–2002 to 2012–2013: results from the National Epidemiologic Survey on Alcohol and Related Conditions. JAMA Psychiatry 2017;74(9):911-23. [DOI] [PMC free article] [PubMed] [Google Scholar]
Gupta 2017
- Gupta M, Verma P, Rastogi R, Arora S, Elwadhi D. Randomized open-label trial of baclofen for relapse prevention in alcohol dependence. American Journal of Drug and Alcohol Abuse 2017;43(3):324-31. [PMID: ] [DOI] [PubMed] [Google Scholar]
Guyatt 2008
- Guyatt GH, Oxman AD, Vist GE, Kunz R, Falck-Ytter Y, Alonso-Coello P, et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ 2008;336(7560):924-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
Guyatt 2011
- Guyatt G, Oxman AD, Akl EA, Kunz R, Vist G, Brozek J, et al. GRADE guidelines 1. Introduction – GRADE evidence profiles and summary of findings tables. Journal of Clinical Epidemiology 2011;64:383-94. [DOI] [PubMed] [Google Scholar]
Haber 2021
- Haber PS, Riordan BC, Winter DT, Barrett L, Saunders J, Hides L, et al. New Australian guidelines for the treatment of alcohol problems: an overview of recommendations. Medical Journal of Australia 2021;215(Suppl 7):S3-32. [PMID: ] [DOI] [PubMed] [Google Scholar]
Han 2021
- Han B, Jones CM, Einstein EB, Powell PA, Compton WM. Use of medications for alcohol use disorder in the US: results from the 2019 national survey on drug use and health. JAMA Psychiatry 2021;78(8):922-4. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2003
- Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ 2003;327:557-60. [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2011
- Higgins JP, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from training.cochrane.org/handbook/archive/v5.1/.
Higgins 2017
- Higgins JP, Altman DG, Sterne JA. Chapter 8: Assessing risk of bias in included studies. In: Higgins JP, Churchill R, Chandler J, Cumpston MS, editor(s), Cochrane Handbook for Systematic Reviews of Interventions Version 5.2.0 (updated June 2017), Cochrane, 2017. Available from training.cochrane.org/handbook/archive/v5.2.
Holla 2015
- Holla B, Gowda GS, Prabhu L, Baby S, Viswanath B, Chand P, et al. High doses of baclofen as suicide attempt in patients with alcohol use disorders – a serious concern. Asian Journal of Psychiatry 2015;17:99-100. [DOI] [PubMed] [Google Scholar]
Hróbjartsson 2014
- Hróbjartsson A, Emanuelsson F, Skou Thomsen AS, Hilden J, Brorson S. Bias due to lack of patient blinding in clinical trials. A systematic review of trials randomizing patients to blind and nonblind sub-studies. International Journal of Epidemiology 2014;43(4):1272-83. [DOI] [PMC free article] [PubMed] [Google Scholar]
Jaury 2014
- Jaury PH. Baclofen for the treatment of alcohol drinkers (Bacloville). Alcoholism: Clinical and Experimental Research 2014;38:215a. [Google Scholar]
Jorgensen 2011
- Jorgensen CH, Pedersen B, Tonnesen H. The efficacy of disulfiram for the treatment of alcohol use disorder. Alcoholism, Clinical and Experimental Research 2011;35:1749-58. [DOI] [PubMed] [Google Scholar]
Jose 2019
- Jose NA, Yadav P, Kapoor A, Mahla VP. Comparison between baclofen and topiramate in alcohol dependence: a prospective study. Indian Journal of Psychiatry 2019;28(1):44-50. [CTRI: 2016/01/006525] [DOI] [PMC free article] [PubMed] [Google Scholar]
Kagereki 2016
- Kagereki E, Gakonyo J, Simila H. Significance bias: an empirical evaluation of the oral health literature. BMC Oral Health 2016;16(1):53. [DOI] [PMC free article] [PubMed] [Google Scholar]
Karthik 2018
- Karthik S, Holla B, Bharath RD, Venkatasubramanian G, Benegal V. Baclofen is more efficacious than oral naltrexone in individuals with severe alcohol dependence for maintaining abstinence – a preliminary report. European Psychiatry 2018;48(Suppl 1):S335. [Google Scholar]
Kent 2020
- Kent CN, Park C, Lindsley CW. Classics in chemical neuroscience: baclofen. Chemical Neuroscience 2020;11(12):1740-55. [PMID: ] [DOI] [PubMed] [Google Scholar]
Krupitsky 1993
- Krupitsky EM, Burakov AM, Ivanov VB, Krandashova GF, Lapin IP, Grinenko AJ, et al. Baclofen administration for the treatment of affective disorders in alcoholic patients. Drug and Alcohol Dependence 1993;33(2):157-63. [DOI: 10.1016/0376-8716(93)90057-w] [DOI] [PubMed] [Google Scholar]
Kumar 2020b
- Kumar R, Kumar KJ, Benegal V, Roopesh BN, Ravi GS. Integrated intervention program for alcoholism improves impulsiveness and disadvantageous reward processing/risk-taking. Indian Journal of Psychiatry 2020;62(4):384-91. [DOI] [PMC free article] [PubMed] [Google Scholar]
Lefebvre 2011
- Lefebvre C, Manheimer E, Glanville J. Chapter 6: Searching for studies. In: Higgins JP, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from training.cochrane.org/handbook/archive/v5.1/.
Leggio 2010
- Leggio L, Garbutt JC, Addolorato G. Effectiveness and safety of baclofen in the treatment of alcohol dependent patients. CNS & Neurological Disorders Drug Targets 2010;9:33-44. [DOI] [PubMed] [Google Scholar]
Leone 2010
- Leone MA, Vigna-Taglianti F, Avanzi G, Brambilla R, Faggiano F. Gamma-hydroxybutyrate (GHB) for treatment of alcohol withdrawal and prevention of relapses. Cochrane Database of Systematic Reviews 2010, Issue 2. Art. No: CD006266. [DOI: 10.1002/14651858.CD006266.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Lesouef 2014
- Lesouef N, Bellet F, Mounier G, Beyens MN. Efficacy of baclofen on abstinence and craving in alcohol-dependent patients: a meta-analysis of randomized controlled trials. Therapie 2014;69(5):427-35. [DOI] [PubMed] [Google Scholar]
Liu 2015
- Liu J, Wang LN. Baclofen for alcohol withdrawal. Cochrane Database of Systematic Reviews 2015, Issue 4. Art. No: CD008502. [DOI: 10.1002/14651858.CD008502.pub4] [DOI] [PubMed] [Google Scholar]
Logge 2022
- Logge WB, Morley KC, Haber PS. GABAB receptors and alcohol use disorders: clinical studies. Current Topics in Behavioral Neurosciences 2022;52:195-212. [PMID: ] [DOI] [PubMed] [Google Scholar]
Maccioni 2005
- Maccioni P, Serra S, Vacca G, Orrù A, Pes D, Agabio R, et al. Baclofen-induced reduction of alcohol reinforcement in alcohol-preferring rats. Alcohol 2005;36(3):161-8. [DOI] [PubMed] [Google Scholar]
Maccioni 2009
- Maccioni P, Colombo G. Role of the GABA(B) receptor in alcohol-seeking and drinking behavior. Alcohol 2009;43(7):555-8. [DOI] [PubMed] [Google Scholar]
Maccioni 2012
- Maccioni P, Zaru A, Loi B, Lobina C, Carai MA, Gessa GL, et al. Comparison of the effect of the GABAΒ receptor agonist, baclofen, and the positive allosteric modulator of the GABAB receptor, GS39783, on alcohol self-administration in 3 different lines of alcohol-preferring rats. Alcoholism, Clinical and Experimental Research 2012;36(10):1748-66. [DOI] [PMC free article] [PubMed] [Google Scholar]
Maccioni 2019
- Maccioni P, Colombo G. Potential of GABAB receptor positive allosteric modulators in the treatment of alcohol use disorder. CNS Drugs 2019;33(2):107-23. [PMID: ] [DOI] [PubMed] [Google Scholar]
MacKillop 2022
- MacKillop J, Agabio R, Feldstein Ewing S, Heilig M, Kelly J, Leggio L, et al. Hazardous drinking and alcohol use disorders. Nature Reviews Disease Primers 2022;8:80. [DOI: Hazardous drinking and alcohol use disorders] [DOI] [PMC free article] [PubMed] [Google Scholar]
Manthey 2019
- Manthey J, Shield KD, Rylett M, Hasan OS, Probst C, Rehm J. Global alcohol exposure between 1990 and 2017 and forecasts until 2030: a modelling study. Lancet 2019;393(10190):2493-502. [PMID: ] [DOI] [PubMed] [Google Scholar]
Manthey 2021
- Manthey J, Hassan SA, Carr S, Kilian C, Kuitunen-Paul S, Rehm J. What are the economic costs to society attributable to alcohol use? A systematic review and modelling study. Pharmacoeconomics 2021;39(7):809-22. [DOI: 10.1007/s40273-021-01031-8] [DOI] [PMC free article] [PubMed] [Google Scholar]
Manzardo 2013
- Manzardo AM, He J, Poje A, Penick EC, Campbell J, Butler MG. Double-blind, randomized placebo-controlled clinical trial of benfotiamine for severe alcohol dependence. Drug and Alcohol Dependence 2013;133(2):562-70. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Manzardo 2015
- Manzardo AM, Pendleton T, Poje A, Penick EC, Butler MG. Change in psychiatric symptomatology after benfotiamine treatment in males is related to lifetime alcoholism severity. Drug and Alcohol Dependence 2015;152:257-63. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Martin‐Fardon 2013
- Martin-Fardon R, Weiss F. Modeling relapse in animals. Current Topics in Behavioral Neurosciences 2013;13:403-32. [DOI] [PMC free article] [PubMed] [Google Scholar]
Mauvais‐Jarvis 2021
- Mauvais-Jarvis F, Berthold HK, Campesi I, Carrero JJ, Dakal S, Franconi F, et al. Sex- and gender-based pharmacological response to drugs. Pharmacological Reviews 2021;73(2):730-62. [DOI: 10.1124/pharmrev.120.000206] [DOI] [PMC free article] [PubMed] [Google Scholar]
Minozzi 2010
- Minozzi S, Amato L, Vecchi S, Davoli M. Anticonvulsants for alcohol withdrawal. Cochrane Database of Systematic Reviews 2010, Issue 3. Art. No: CD006754. [DOI: 10.1002/14651858.CD006754.pub4] [DOI] [PMC free article] [PubMed] [Google Scholar]
Moncrieff 2004
- Moncrieff J, Wessely S, Hardy R. Active placebos versus antidepressants for depression. Cochrane Database of Systematic Reviews 2004, Issue 1. Art. No: CD003012. [DOI: 10.1002/14651858.CD003012.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Moos 2006
- Moos RH, Moos BS. Rates and predictors of relapse after natural and treated remission from alcohol use disorders. Addiction 2006;101(2):212-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
Morley 2013
Morley 2021
- Morley KC, Perry CJ, Watt J, Hurzeler T, Leggio L, Lawrence AJ, et al. New approved and emerging pharmacological approaches to alcohol use disorder: a review of clinical studies. Expert Opinion on Pharmacotherapy 2021;22(10):1291-303. [PMID: ] [DOI] [PubMed] [Google Scholar]
Morley 2022
- Morley KC, Louie E, Hurzeler T, Baillie A, Dore G, Phung N, et al. Sex as a potential moderator for baclofen response in the treatment of alcohol dependence. Frontiers in Global Women's Health 2022;3:807269. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Mutschler 2016
- Mutschler J, Grosshans M, Soyka M, Rösner S. Current findings and mechanisms of action of disulfiram in the treatment of alcohol dependence. Pharmacopsychiatry 2016;49(4):137-41. [DOI] [PubMed] [Google Scholar]
NCT01980706
- NCT01980706. Efficacy and safety of high-dose baclofen for alcohol dependence. www.clinicaltrials.gov/ct2/show/NCT01980706 (first received 11 November 2013).
Nickerson 1998
- Nickerson RS. Confirmation bias: a ubiquitous phenomenon in many guises. Review of General Psychology 1998;2(2):175-220. [Google Scholar]
Pani 2014
- Pani PP, Trogu E, Pacini M, Maremmani I. Anticonvulsants for alcohol dependence. Cochrane Database of Systematic Reviews 2014, Issue 2. Art. No: CD008544. [DOI: 10.1002/14651858.CD008544.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Pastor 2012
- Pastor A, Jones D, Currie J. High-dose baclofen for treatment-resistant alcohol dependence. Journal of Clinical Psychopharmacology 2012;32:266-8. [DOI] [PubMed] [Google Scholar]
Pedersen 2013
- Pedersen B, Askgaard G, Oppedal K. Disulfiram for alcohol use disorder. Cochrane Database of Systematic Reviews 2013, Issue 4. Art. No: CD010487. [DOI: 10.1002/14651858.CD010487] [DOI] [Google Scholar]
Pelissier 2017
- Pelissier F, Haro L, Cardona F, Picot C, Puskarczyk E, Sapori JM, et al. Self-poisoning with baclofen in alcohol-dependent patients: national reports to French Poison Control Centers, 2008–2013. Clinical Toxicology 2017;55(4):275-84. [DOI] [PubMed] [Google Scholar]
Pierce 2018
- Pierce M, Sutterland A, Beraha EM, Morley K, den Brink W. Efficacy, tolerability, and safety of low-dose and high-dose baclofen in the treatment of alcohol dependence: a systematic review and meta-analysis. European Neuropsychopharmacology 2018;28(7):795-806. [DOI: 10.1016/j.euroneuro.2018.03.017] [DOI] [PubMed] [Google Scholar]
Pitman 2014
- Pitman KA, Puil E, Borgland SL. GABA(B) modulation of dopamine release in the nucleus accumbens core. European Journal of Neuroscience 2014;40(10):3472-80. [DOI] [PubMed] [Google Scholar]
Ray 2020
- Ray LA, Meredith LR, Kiluk BD, Walthers J, Carroll KM, Magill M. Combined pharmacotherapy and cognitive behavioral therapy for adults with alcohol or substance use disorders: a systematic review and meta-analysis. JAMA Network Open 2020;3(6):e208279. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Reus 2018
- Reus VI, Fochtmann LJ, Bukstein O, Eyler AE, Hilty DM, Horvitz-Lennon M, et al. The American Psychiatric Association practice guideline for the pharmacological treatment of patients with alcohol use disorder. American Journal of Psychiatry 2018;175(1):86-90. [PMID: ] [DOI] [PubMed] [Google Scholar]
Reynoard 2020
- Reynoard J, Schmitt C, Torrents R, Simon N. Toxicological considerations in the prescription of baclofen for the treatment of substance use disorders. Expert Opinion on Drug Metabolism & Toxicology 2020;16(4):309-17. [PMID: ] [DOI] [PubMed] [Google Scholar]
Roerecke 2013
- Roerecke M, Rehm J. Alcohol use disorders and mortality: a systematic review and meta-analysis. Addiction 2013;108(9):1562-78. [DOI] [PubMed] [Google Scholar]
Rolland 2013
- Rolland B, Bordet R, Deheul S, Cottencin O. Baclofen for alcohol-dependence: anticraving or partial substitution? Journal of Clinical Psychopharmacology 2013;33(2):280-1. [DOI] [PubMed] [Google Scholar]
Rolland 2020
- Rolland B, Simon N, Franchitto N, Aubin HJ. France grants an approval to baclofen for alcohol dependence. Alcohol and Alcoholism 2020;55(1):44-5. [PMID: ] [DOI] [PubMed] [Google Scholar]
Rose 2018
- Rose AK, Jones A. Baclofen: its effectiveness in reducing harmful drinking, craving, and negative mood. A meta-analysis. Addiction 2018;113(8):1396-406. [DOI] [PubMed] [Google Scholar]
Rösner 2010a
- Rösner S, Hackl-Herrwerth A, Leucht S, Lehert P, Vecchi S, Soyka M. Acamprosate for alcohol dependence. Cochrane Database of Systematic Reviews 2010, Issue 9. Art. No: CD004332. [DOI: 10.1002/14651858.CD004332.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Rösner 2010b
- Rösner S, Hackl-Herrwerth A, Leucht S, Vecchi S, Srisurapanont M, Soyka M. Opioid antagonists for alcohol dependence. Cochrane Database of Systematic Reviews 2010, Issue 12. Art. No: CD001867. [DOI: 10.1002/14651858.CD001867.pub3] [DOI] [PubMed] [Google Scholar]
Sarai 2013
- Sarai M, Tejani AM, Chan AH, Kuo IF, Li J. Magnesium for alcohol withdrawal. Cochrane Database of Systematic Reviews 2013, Issue 6. Art. No: CD008358. [DOI: 10.1002/14651858.CD008358.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Schünemann 2006
- Schünemann HJ, Jaeschke R, Cook D, Bria W, El-Solh A, Ernst A. An official ATS statement: grading the quality of evidence and strength of recommendations in ATS guidelines and recommendations. American Journal of Respiratory and Critical Care Medicine 2006;174(5):605-14. [DOI] [PubMed] [Google Scholar]
Schwarz Pharma 2003
- Schwarz Pharma. Kemstro® (oral baclofen tablets) package insert. Monheim (Germany) 2003.
Sharma 2012
- Sharma V, Sharma R, Chaturvedi M, Bharadwaj B. A dose–response effect of baclofen in reducing daily alcohol intake in alcohol-dependent subjects. Journal of Gastroenterology and Hepatology 2012;27:263. [DOI: 10.1111/jgh.12006] [DOI] [Google Scholar]
Shield 2020
- Shield K, Manthey J, Rylett M, Probst C, Wettlaufer A, Parry CD, et al. National, regional, and global burdens of disease from 2000 to 2016 attributable to alcohol use: a comparative risk assessment study. Lancet Public Health 2020;5(1):e51-61. [DOI: 10.1016/S2468-2667(19)30231-2] [DOI] [PubMed] [Google Scholar]
Sinclair 2016
- Sinclair JM, Chambers SE, Shiles CJ, Baldwin DS. Safety and tolerability of pharmacological treatment of alcohol dependence: comprehensive review of evidence. Drug Safety 2016;39(7):627-45. [PMID: ] [DOI] [PubMed] [Google Scholar]
Skinner 2014
- Skinner MD, Lahmek P, Pham H, Aubin HJ. Disulfiram efficacy in the treatment of alcohol dependence: a meta-analysis. PLOS One 2014;9(2):e87366. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Slade 2016
- Slade T, Chapman C, Swift W, Keyes K, Tonks Z, Teesson M. Birth cohort trends in the global epidemiology of alcohol use and alcohol-related harms in men and women: systematic review and metaregression. BMJ Open 2016;6(10):e011827. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Spagnolo 2020
- Spagnolo PA, Montemitro C, Leggio L. New challenges in addiction medicine: COVID-19 infection in patients with alcohol and substance use disorders – the perfect storm. American Journal of Psychiatry 2020;177(9):805-7. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Steardo 1984
- Steardo L, Leo A, Marano E. Efficacy of baclofen in trigeminal neuralgia and some other painful conditions. A clinical trial. European Neurology 1984;23(1):51-5. [DOI] [PubMed] [Google Scholar]
Sterne 2017
- Sterne JA, Egger M, Moher D, Boutron I, editor(s). Chapter 10: Addressing reporting biases. In: Higgins JP, Churchill R, Chandler J, Cumpston MS, editor(s), Cochrane Handbook for Systematic Reviews of Interventions Version 5.2.0 (updated June 2017), Cochrane, 2017. Available from training.cochrane.org/handbook/archive/v5.2.
Thompson 2017
- Thompson A, Owens L, Richardson P, Pirmohamed M. Systematic review: baclofen dosing protocols for alcohol use disorders used in observational studies. European Neuropsychopharmacology 2017;27(11):1077-89. [DOI] [PubMed] [Google Scholar]
Vaz de Lima 2010
- Vaz de Lima FB, Andriolo RB, da Silveira DX. Dopaminergic antagonists for alcohol dependence. Cochrane Database of Systematic Reviews 2010, Issue 4. Art. No: CD003352. [DOI: 10.1002/14651858.CD003352.pub4] [DOI] [Google Scholar]
Volkow 2011
- Volkow ND, Wang GJ, Fowler JS, Tomasi D, Telang F. Addiction: beyond dopamine reward circuitry. Proceedings of the National Academy of Sciences USA 2011;108(37):15037-42. [DOI] [PMC free article] [PubMed] [Google Scholar]
Walker 2007
- Walker BM, Koob GF. The gamma-aminobutyric acid-B receptor agonist baclofen attenuates responding for ethanol in ethanol-dependent rats. Alcoholism, Clinical and Experimental Research 2007;31(1):11-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
White 2015
- White A, Castle IJ, Chen CM, Shirley M, Roach D, Hingson R. Converging patterns of alcohol use and related outcomes among females and males in the United States, 2002 to 2012. Alcoholism, Clinical and Experimental Research 2015;39(9):1712-26. [DOI] [PubMed] [Google Scholar]
WHO 1992
- World Health Organization. International Statistical Classification of Diseases and Related Health Problems, 10th Revision (ICD-10). Geneva (Switzerland): World Health Organization, 1992. [Google Scholar]
WHO 2000
- World Health Organization. International guide for monitoring alcohol consumption and related harm. apps.who.int/iris/handle/10665/66529 (accessed 8 December 2022). [www.who.int/iris/handle/10665/66529]
WHO 2018
- World Health Organization. Global status report on alcohol and health 2018. www.who.int/publications/i/item/9789241565639 (accessed 8 December 2022).
WHO 2019
- World Health Organization. International Classification of Diseases 11th revision (ICD-11). World Health Organization 2019.
Wilsey 2016
- Wilsey B, Deutsch R, Marcotte TD. Maintenance of blinding in clinical trials and the implications for studying analgesia using cannabinoids. Cannabis and Cannabinoid Research 2016;1(1):139-48. [DOI] [PMC free article] [PubMed] [Google Scholar]
Wise 2009
- Wise RA. Roles for nigrostriatal – not just mesocorticolimbic–dopamine in reward and addiction. Trends in Neurosciences 2009;32(10):517-24. [DOI] [PMC free article] [PubMed] [Google Scholar]
Witkiewitz 2019
- Witkiewitz K, Litten RZ, Leggio L. Advances in the science and treatment of alcohol use disorder. Science Advances 2019;5(9):eaax4043. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Young 2014
- Young KA, Franklin TR, Roberts DC, Jagannathan K, Suh JJ, Wetherill RR, et al. Nipping cue reactivity in the bud: baclofen prevents limbic activation elicited by subliminal drug cues. Journal of Neuroscience 2014;34(14):5038-43. [DOI] [PMC free article] [PubMed] [Google Scholar]
References to other published versions of this review
Minozzi 2017
- Minozzi S, Saulle R, Rösner S. Baclofen for alcohol use disorder. Cochrane Database of Systematic Reviews 2017, Issue 2. Art. No: CD012557. [DOI: 10.1002/14651858.CD012557] [DOI] [PMC free article] [PubMed] [Google Scholar]
Minozzi 2018
- Minozzi S, Saulle R, Rösner S. Baclofen for alcohol use disorder. Cochrane Database of Systematic Reviews 2018, Issue 11. Art. No: CD012557. [DOI: 10.1002/14651858.CD012557.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]


