Abstract
While lung cancer has been the leading cause of cancer-related deaths for many years in the United States, incidence and mortality statistics – among other measures – vary widely worldwide. The aim of this study was to review the evidence on lung cancer epidemiology, including data of international scope with comparisons of economically, socially, and biologically different patient groups. In industrialized nations, evolving social and cultural smoking patterns have led to rising or plateauing rates of lung cancer in women, lagging the long-declining smoking and cancer incidence rates in men. In contrast, emerging economies vary widely in smoking practices and cancer incidence but commonly also harbor risks from environmental exposures, particularly widespread air pollution. Recent research has also revealed clinical, radiologic, and pathologic correlates, leading to greater knowledge in molecular profiling and targeted therapeutics, as well as an emphasis on the rising incidence of adenocarcinoma histology. Furthermore, emergent evidence about the benefits of lung cancer screening has led to efforts to identify high-risk smokers and development of prediction tools. This review also includes a discussion on the epidemiologic characteristics of special groups including women and nonsmokers. Varying trends in smoking largely dictate international patterns in lung cancer incidence and mortality. With declining smoking rates in developed countries and knowledge gains made through molecular profiling of tumors, the emergence of new risk factors and disease features will lead to changes in the landscape of lung cancer epidemiology.
Burden of Disease
Internationally, lung cancer continues to be the leading cause of cancer-related deaths in men and women [1]. A breakdown by level of economic development shows no differences in cancer deaths in men but a higher rate of lung cancer deaths in women in industrialized countries as compared with developing nations. Among females in developing countries, lung cancer deaths lag behind those due to breast cancer [2]. Lung cancer incidence and mortality are tightly linked to cigarette smoking patterns. As smoking rates peak – generally first in men, followed by women – lung cancer incidence and mortality rise in subsequent decades before declining following the initiation of comprehensive tobacco control programs [3,4,5]. These trends have occurred earlier in industrialized countries as compared with the developing world. In the United States (US) and the United Kingdom (UK), lung cancer incidence and mortality rates have in fact been falling since the 1990s. In contrast, emerging nations – including Brazil, Russia, India, China, and South Africa (BRICS) – continue to have high rates of cigarette smoking in both men and women. They exhibit a lower incidence of cancer but a higher mortality burden compared with developed countries. Reasons for these patterns include unequal access to healthcare leading to delayed diagnosis and treatment, environmental contamination, and sociocultural barriers [6].
Industrialized Countries
In the US, the incidence of lung cancer in men peaked in the 1980s, followed by a subsequent decline, with similar patterns in women following 20 years later [7]. Thun et al. found that in the 1960s, the relative risk of lung cancer death in smokers versus non-smokers was more than four times higher in men than in women. In the 40 years since, women’s risk has risen markedly, becoming nearly identical to that of men [3,8]. Lung cancer deaths in men are now declining at an average of 2.9% annually with a percent decrease roughly double that of women [7]. With regard to differences between racial and ethnic groups, non-Hispanic whites and blacks have the highest incidence and death rates [9]. In particular, black men have the highest mortality, approximately double that of Asian Americans, the group with the lowest cancer-specific mortality [7,10]. These racial and ethnic disparities are largely due to differences in cigarette smoking prevalence, as well as lower rates of resection and higher probability of advanced stage at diagnosis in minorities [11,12,13].
The UK has similar smoking and lung cancer incidence trends to the US. Male smoking prevalence peaked in the 1940s to 1950s, followed by a peak in lung cancer incidence in the 1970s. Despite declining rates in both sexes, lung cancer remains the second most common malignancy in the UK [14]. Mainland Europe exhibits wide geographic variations in lung cancer incidence (Figure 1). In general, rates are highest in central and eastern Europe, but incidence throughout the continent has been declining in men since the early 1990s. Exceptions include Norway, Finland, Spain, and France, where lung cancer rates have remained stable. In women, rising lung cancer incidence has slowed in the US and UK, but rates continue to increase in central and eastern Europe [15,16,17,18,19,20]. These regional differences reflect earlier stages of the tobacco epidemic in countries such as Belarus, Hungary, Poland, and the Russian Federation [4,5,15,17]. Additionally, socioeconomic and educational inequalities, as well as diagnosis at later stages of disease, contribute to variability in lung cancer incidence and mortality within Europe [21,22]. Finally, similarly to the US, lung cancer survival is lower than that of any other common malignancies in Europe. EUROCARE-5 reported a mean five-year survival of 13% for all lung cancer patients diagnosed in 2000–2007, with a range from 9% in the UK and Ireland to 15% in central Europe [23,24].
Figure 1.
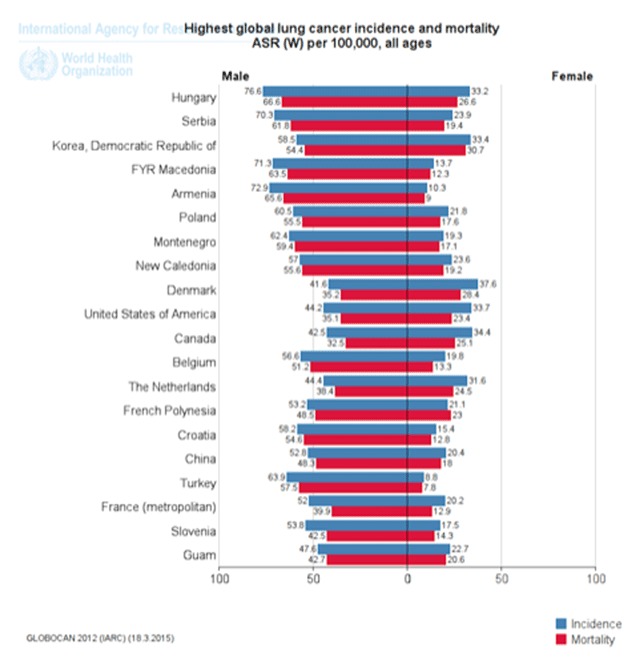
Global lung cancer incidence and mortality. Global age-standardized incidence and mortality rates for lung cancer, 20 countries with the highest rates internationally. Reproduced from GLOBOCAN 2012 data with permission.
In Asia, Japan has high incidence and mortality rates from lung cancer, comparable to those of the US and Europe [25]. Men have had a higher incidence of lung cancer than women since the 1970s and continue to comprise the majority of new lung cancer cases in Japan today, largely due to gender differences in smoking prevalence [26]. Conversely, mortality rates in women are lower in Japan than in other industrialized nations, perhaps due to the higher incidence of adenocarcinomas with mutations responsive to targeted therapies [27].
Emerging Economies
Brazil, Russia, India, China, and South Africa are recognized by their large and fast-growing economies [28]. One of the few South American countries with a cancer registry is Brazil, where tobacco smoking peaked in the 1970s and lung cancer mortality in men peaked in 1993 and continues to rise among women [29,30]. Likewise, in the Russian Federation, all-cause mortality in men is largely attributed to very high rates (60%) of both smoking and alcohol consumption, which are much lower in women [6]. Accordingly, Russia has among the highest lung cancer mortality rate in men of all European countries but among the lowest in women. Mortality is now declining, after peaking in the early 1990s, but tobacco use remains a major barrier to effective cancer control [16]. Additional risk factors in Russia include environmental pollution and workplace exposures in nuclear facilities and asbestos mines [6].
Comparatively, lung cancer incidence and mortality rates in India are among the lowest in the world [4]. The most common cancers in men are head and neck, gastric, and esophageal cancers, attributed to high usage of smokeless tobacco; the most common cancers among women are cervical and breast. One study in northern India noted that squamous cell lung cancer was the most common histology overall and among smokers [31]. While cigarette smoking has a reported prevalence ranging from 28 to 57% among men, bidi smoking (hand-rolled tobacco) is the most commonly used (92%) tobacco product [32].
In 2005, the total number of new lung cancer cases in China was over 500,000. According to GLOBOCAN 2012, lung cancer is the most common malignancy and cause of cancer mortality in China, representing 21% of all cancers and 27% of all cancer-related deaths [6]. Lung cancer incidence and mortality is higher in eastern China and in urban areas, which has been attributed to westernization of lifestyle [33]. However, mortality rates are increasing faster in rural areas due to poor access to care [6]. Additionally, age-adjusted mortality rates are higher in Chinese men – 68% of whom are smokers – but lung cancer incidence rates are rising faster in women [34]. Risk factors among Chinese women include secondhand smoking, air pollution, and domestic use of biomass fuels [35].
Developing Countries
Reporting of cancer epidemiology in Africa is limited by the lack of reliable registries. Among countries on the African continent as whole, both incidence and mortality rates are low – lung cancer was the fifth most common site of cancer in African men and not even in the top 10 for women. This is likely due to the low prevalence of smoking (10% in men and <2% in women) as well as the lower life expectancy of the population. Lung cancer does have a high incidence in certain regions including the northern African countries of Western Sahara, Morocco, Algeria, Tunisia, and Libya, and it is the leading cause of cancer death in men in northern and southern Africa [36].
South America has a wide range of lung cancer incidence across countries and markedly higher rates in men compared with women. The highest incidence and mortality in men can be found in Uruguay and in women of Venezuela and Argentina. Less populated countries such as Ecuador, Bolivia, and Guyana have very low age-standardized rates, just higher than those of central Africa and the Middle East [25].
The remainder of Asia has extremely diverse lung cancer incidences, which are nevertheless consistent within different regions. Asian countries closest to Eastern Europe such as Armenia, Turkey, and Kazakhstan have among the highest rates of lung cancer in the world. Korea and southeast Asia have slightly lower rates, and Middle Eastern countries including Yemen and Saudi Arabia have among the lowest lung cancer incidence rates in the world [25]. These notable regional differences reflect geographic trends in the tobacco epidemic [37].
Histopathology
Lung cancer was traditionally classified into two primary groups, small versus non–small cell type. This grouping was progressively specified with the use of histopathologic features and immunohistochemical markers, and now inroads are being made in distinguishing invasive adenocarcinomas from pre-invasive lesions. Moreover, further knowledge about the molecular characteristics of lung cancers and the availability of targeted therapies has substantially impacted the classification of lung cancers.
Histology
Adenocarcinoma is the most common histologic subtype of lung cancer in men and women [38]. Prior to the 1990s, squamous cell lung carcinoma was the most common histologic subtype, particularly among men. Since then, the incidence of adenocarcinoma rose to be greater than that of squamous cell carcinomas in the US, Canada, many European countries, and Japan [26,39]. However, this switch has not yet been observed in other countries such as Spain and the Netherlands [39]. The higher rates of adenocarcinoma relative to squamous and small cell lung cancer are greater in women [4]. Consequently, the proportion of adenocarcinomas is rising in many countries in parallel to increased incidence of lung cancer in women. These findings may reflect differences in the types of cigarettes (including filtered and low-tar versions) more frequently used by women as well as genetic predisposition and environmental exposures in female never-smokers [39].
In 2011, the International Association for the Study of Lung Cancer, American Thoracic Society, and European Respiratory Society proposed a new adenocarcinoma categorization based upon histological evidence of invasion. Preinvasive lesions are classified on a continuum from atypical adenomatous hyperplasia (AAH) to adenocarcinoma in situ (AIS), and minimally invasive adenocarcinoma (MIA) includes small (<3 cm) lesions with ≤5 mm of invasion. Invasive adenocarcinomas include a variety of patterns (e.g., lepidic predominant adenocarcinoma [LPA], acinar, papillary, micropapillary, and solid) characterized by tumor disruption of >5 mm of the alveolar basement membrane. This grouping correlates with clinical outcomes, with pre-invasive lesions having an indolent clinical course with almost 100% curability, in contrast to invasive carcinomas, which have a considerably worse prognosis [40]. Recent studies have shown AIS, MIA, and LPA to have a higher incidence in Japan compared with western populations [27].
Squamous cell lung cancer is the second most common subtype, comprising approximately 20% of primary lung neoplasms in the US. These tumors are distinguished histologically by squamous pearl formation, keratin production, and intercellular bridging. Historically, squamous cell lung cancer occurred more commonly as central lesions, but peripheral tumors are rising in incidence [41]. Small cell lung cancer, which has an aggressive clinical course, comprises 14% of lung cancers and typically presents as a perihilar mass with early and extensive lymph node metastases. It has a strong association with smoking history and commonly causes paraneoplastic syndromes. Less frequent histologic subtypes of lung cancer include large cell (3%), adenosquamous (1–2%), and carcinoid tumors (1–2%) [41].
Molecular Markers
The most common genetic alterations in lung adenocarcinoma are epidermal growth factor receptor (EGFR) and KRAS activating mutations (Figure 2). EGFR insertions and deletions are found in roughly 15% of lung adenocarcinomas in the US, with increased frequency in nonsmokers (43% vs. 11% in smokers) [42] and Asians (up to 60% in Asian women) [43]. In advanced stages of disease, this mutation predicts a more favorable prognosis and sensitivity to EGFR tyrosine kinase inhibitors (TKIs) such as erlotinib, gefitinib, and afatinib [44]. Conversely, KRAS mutations occur more commonly in smokers and appear to confer worse prognosis [42]. While no targeted therapeutics are currently available for this mutation, clinical trials are in progress to test drugs that target downstream effectors of activated KRAS [44]. Additional driver mutations in lung adenocarcinoma occur with a frequency of <1–4%, including ALK gene rearrangements, ROS1 translocations, HER2 mutations, BRAF mutations, and RET translocations. ALK rearrangement are clinically important however, as this mutation creates a fusion product, most frequently with EML4, which predicts sensitivity to ALK tyrosine kinase inhibitors such as crizotinib and ceritinib [44]. Additionally, ALK-positive tumors have been associated with acinar or solid histological patterns with signet ring features [45,46].
Figure 2.

Frequencies of common driver mutations in lung adenocarcinoma in the US and Europe. By overall frequency (A) and population group (B). Data are derived from large clinicopathologic cohort studies published since 2008 and are representative of US and European populations.
Risk Factors and Environmental Exposures
The myriad risk factors for lung cancer most commonly include lifestyle, environmental, and occupational exposures. The roles these factors play vary depending on geographic location, sex and race characteristics, genetic predisposition, as well as their synergistic interactions.
Cigarette Smoking, Secondhand Smoking, and E-cigarettes
Cigarette smoking is the most recognized risk factor for developing lung cancer. Since the Surgeon General’s report on smoking and health in 1964, at which time 52% of American men and 35% of American women were active smokers, the prevalence of cigarette smoking in US and consequently lung cancer has markedly decreased [47]. While nicotine itself is not carcinogenic, there be as many as 55 substances in cigarette smoke that have been deemed carcinogenic by the International Agency for Research on Cancer including polycyclic aromatic hydrocarbons and 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK). Their activation leads to the formation of DNA adducts and subsequent gene methylation, DNA sequence changes, DNA segment amplification or deletion, or whole chromosome gains or losses [48]. Relative risk of lung cancer in smokers as compared with smokers varies from 10- to 30-fold, and the degree of risk is dependent on number of cigarettes smoked daily and pack-years of smoking. Cigar and pipe tobacco smoking are also associated with increased odds of developing lung cancer [49].
Secondhand smoke exposure also leads to a dose-dependent risk of lung cancer. Öberg and colleagues studied the effects of environmental tobacco exposure (ETS) in 192 countries on six continents and found that 40% of children and 33%–35% of non-smokers are exposed to secondhand smoke. The highest rates were in Europe, the western Pacific, and parts of Southeast Asia; the lowest rates were found in Africa. Over 600,000 deaths worldwide, most of them in women, were attributable to secondhand smoking in 2004 [50]. Similar to Öberg’s finding of the relative risk for lung cancer in adult non-smokers exposed to secondhand smoke of 1.21 (95% confidence interval [CI]: 1.13–1.3), many regulatory bodies have reported an increase in lung cancer risk by 20 to 30% upon exposure to ETS [51,52,53,54,55]. The largest numbers of estimated deaths in adults attributable to secondhand smoke, however, are not due to lung cancer but rather ischemic heart disease and asthma [50].
Electronic cigarettes have sparked much recent controversy over potential risks from long-term use, as well as their role in smoking initiation and potentially cessation [56]. The 2012 National Youth Tobacco Survey found the prevalence of ever-use of e-cigarettes among middle and high school students in the US to be 6.8%, while prevalence among adults in the simultaneous National Adult Tobacco Survey was 1.9% [57,58]. Although conventional cigarette use is far higher (18% among adults), the incidence of e-cigarette use is rising rapidly and has been associated with higher odds of cigarette smoking and lower odds of abstinence [59,60]. Even more concerning, early research has shown that an e-cigarette vapor-conditioned media induced gene expression patterns in human bronchial epithelial cells concordant with that of cells exposed to a cigarette smoke-conditioned media [61].
Domestic Biomass Fuels
Unprocessed biomass fuels, including wood, crop residues, dung, and coal, are used by approximately half of the world’s population for in-home cooking or heating, primarily in eastern and southern Asia [62]. Indoor emissions in these households contain high concentrations of polycyclic aromatic hydrocarbons, benzene, and other carcinogenic compounds [63]. Several studies have confirmed an increased lung cancer risk associated with biomass fuels, with one pooled analysis showing an odds ratio (OR) of 4.93 (95% CI: 3.73–6.52) among coal users in Asia when compared with nonsolid-fuel users [64,65]. A meta-analysis including subjects from Europe and North America, in addition to Asia, reported similar trends in lung cancer risk with exposure to coal, biomass, and mixed fuels [62]. Additional studies have reported increased risk with bituminous “smoky coal” use compared with anthracite-based “smokeless coal,” as well as higher risk with domestic exposures in smokers compared with nonsmokers [63,66].
COPD and other Pulmonary Conditions
While epidemiological studies report that approximately 20–30% of smokers develop COPD and 10–15% develop lung cancer, COPD is by far the most common comorbidity in patients with lung cancer, with a varying prevalence between 30 and 70% [67]. A cohort of newly diagnosed lung cancer cases was reported to have a prevalence of COPD as high as six-fold greater than matched smokers without cancer [67]. Additional studies have shown factors such as increasing degree of airway obstruction, increasing age, lower body mass index, and a diffusing lung capacity of carbon monoxide < 80% to be associated with a diagnosis of lung cancer [68]. Furthermore, extent of emphysema on CT is an independent risk factor for lung cancer, as well as a predictor of cancer-specific mortality [69,70]. A recent pooled analysis of almost 25,000 cases from the International Lung Cancer Consortium also showed both lung cancer incidence and mortality to be significantly associated with emphysema [71,72]. Proposed mechanisms for the link between COPD and lung cancer include matrix remodeling and lung repair processes which lead to development of epithelial-mesenchymal transition and carcinogenesis. Additionally, several genome-wide association and candidate gene studies have identified associations between emphysema and lung cancer at several chromosomal loci, supporting that susceptibility to lung cancer may include COPD-related gene variants [73]. In a large meta-analysis, never-smokers with a history of chronic bronchitis, tuberculosis, or pneumonia were found to have an increased risk of lung cancer [71,74].
Occupational Exposures
Exposure to asbestos is one of the most well-recognized occupational causes of lung cancer. Workers in asbestos mining and milling, shipbuilding, construction, textiles and insulation, and automobile repair are at the highest risk. Multiple mechanisms exist for carcinogenesis, including induction of oxidative damage and subsequent DNA deletions, somatic gene alterations, and enhanced delivery of tobacco carcinogens to the airway epithelium [75]. Markowitz and colleagues evaluated 2,377 North American insulators and found increased lung cancer risk to be associated with asbestos exposure (rate ratio: 3.6, 95% CI: 1.7–7.6) and asbestosis (rate ratio: 7.4, 95% CI: 4.0–13.7), with synergistic effects in smokers [76]. Diesel exhaust exposure has also been studied in trucking industry workers and coal miners. The SYNERGY project, a pooled analysis of 11 case-control studies conducted in Europe and Canada, which included 13,304 cases, showed that cumulative diesel exposure was associated with an increased lung cancer risk (OR: 1.31, 95% CI: 1.19–1.43) after controlling for other occupational exposures [77]. Similar findings were obtained in studies conducted in the US trucking and non-metal mining industries [78,79,80]. Other occupations with an increased incidence of lung cancer include coal-mining [81], asphalt paving with coal tar exposure [82], chimney sweeping [83], and painting [84] although the risk appears to be lower than that of asbestos and diesel exhaust. Other organic and metal exposures that have been associated with lung cancer include beryllium, cadmium, chromium, silica, formaldehyde, benzo[a]pyrene, nickel, hard metal dust, and vinyl chloride, which often act synergistically with tobacco smoking [84,85,86].
Ambient Air Pollution and Other Environmental Exposures
European and American studies have evaluated the association of ambient air pollution with lung cancer risk. The ESCAPE study, an analysis of multiple cohorts from nine European countries, found particulate matter (PM) concentration in ambient air to be significantly associated with lung cancer risk (hazard ratio [HR]: 1.22, 95% CI; 1.03–1.45), particularly adenocarcinoma [87]. Studies in Canada [88], the Netherlands [89,90], and the UK [91] also found PM with median aerodynamic diameter less than 2.5 μm (PM2.5), in addition to nitrogen oxides, nitrogen dioxide, and sulfur dioxide to be associated with greater risk of lung cancer. In the US, analysis of the Cancer Prevention Study (CPS) II cohort found an increase in lung cancer mortality with increasing concentrations of PM2.5 in both non-smokers and smokers [92,93].
Arsenic occurring naturally in drinking water and food has been implicated in lung cancer [94,95]. Heck and colleagues evaluated 223 lung cancer cases and found an OR of 2.75 (95% CI; 1.00–7.57) for small cell and squamous cancers in subjects with increased toenail arsenic concentration [96]. Residential radon exposure is another known risk factor for lung cancer. An analysis of the CPS II cohort demonstrated a significant linear relationship between radon concentration and lung cancer mortality [97]. A Spanish case-control study found similar results, and also noted a strong interaction with tobacco (OR: 2.21 95% CI: 1.33–3.69 in non-smokers vs. OR: 73 95% CI: 20–268 in heavy smokers) [98].
Diet and Nutrition
Fruit and vegetable consumption have been associated with decreased lung cancer risk in current smokers; ingestion of cruciferous vegetables, in particular, has been inversely associated with lung cancer risk [99,100]. An analysis of 264 lung cancer tissue samples showed differentially expressed miRNAs among subjects with intake of quercetin-rich versus quercetin-poor fruit and vegetables [101]. Many studies have attempted to evaluate the effects of vitamin levels and intake on lung cancer risk. Dietary and supplemental calcium intake has been shown to be inversely associated with lung cancer risk in female nonsmokers (HR: 0.66, 95% CI: 0.48–0.91), current smokers, particularly for lung adenocarcinoma [102,103]. In addition, total iron intake was inversely associated with lung cancer risk in women, while total magnesium intake increased risk in men and current smokers; no significant association was found between copper, selenium, and zinc with lung cancer risk. Johansson and colleagues found elevated serum vitamin B6 and methionine levels associated with a lower risk for lung cancer in never, former, and current smokers in Europe [104]. Two studies showed a protective effect in never-smoking women with vitamin D intake ≥ 400 IU/day and with supplemental soy intake [105,106]. Conversely, the Alpha-Tocopherol, Beta-Carotene Cancer Prevention Study, revealed a greater incidence of lung cancer and overall mortality in male smokers supplemented with 20mg b-carotene daily [107]. and prompted the US Preventive Services Task Force (USPSTF) to uphold their recommendation against the use of vitamin E for the lung cancer prevention. Moreover, the USPSTF recently concluded that there is insufficient evidence to recommend any vitamins, minerals, and multivitamin supplementation for lung cancer prevention [108].
Genetic Factors
Genetic factors leading to increased susceptibility to lung cancer have been poorly studied. First-degree relatives of patients with lung cancer are at increased risk, even after adjusting for smoking habits [109]. A meta-analysis of 28 case-control studies and 17 observational cohort studies of individuals with positive family histories found a RR of 1.84 (95% CI: 1.64–2.05) for developing lung cancer [110]. Additionally, genome-wide association studies have suggested susceptibility loci on various chromosomes, including 5p15.33 and 3q28, among others, but later analyses have not replicated these results [111,112]. Other studies have identified polymorphisms in various enzymes such as cytochrome p450 enzymes and DNA repair genes [113], as well as germline mutations in the EGFR [114].
Measures of Lung Cancer Risk and Impact on Early Diagnosis
Except for smoking cessation, perhaps the highest reduction in lung cancer mortality rates is related to diagnosis at early stage followed by surgical resection. While chest X-rays and sputum cytology screening have not shown a benefit, early detection via low-dose chest tomography (CT) is now endorsed by the USPSTF [115]. The National Lung Screening Trial (NLST) compared annual screening by low-dose chest tomography (LDCT) with chest X-ray for three years at 33 US medical centers in 53,454 high-risk subjects 55 to 74 years of age with at least 30 pack-years of smoking and found a 20% lung cancer mortality reduction [116]. Ongoing European trials will provide additional critical information regarding the potential benefits of lung cancer screening [117,118]. However, CT screening is associated with high rates of positive findings and may lead to identification of some lung cancers with low aggressiveness [119]. Thus, better risk stratification using prediction models or biomarkers of lung cancer risk, as well as a better understanding of the biologic characteristics of aggressive cancers is required to maximize the benefit of screening.
Even within smokers, lung cancer risk varies considerably based on factors such as age, quantity and duration of smoking, and environmental exposures [3]. There is an ongoing effort to stratify lung cancer risk to identify individuals best suited for lung cancer screening or refine eligibility for prevention trials. Several models based on sociodemographic characteristics, smoking, and other risk factors have been empirically derived using relatively large cohorts [120,121]. Among these, the Bach model was developed using information from 18,172 subjects in the Carotene and Retinol Efficacy Trial, which followed heavy smokers and asbestos-exposed workers from 1989 to 1996 [122]. This model was validated in the Alpha-Tocopherol Beta-Carotene Cancer Prevention Study and was found to underestimate 10-year absolute lung cancer risk while having a discriminatory power comparable to breast cancer risk models [123]. Likewise, the Spitz model was derived and validated in a cohort of 1,851 lung cancer patients and 2,001 matched controls from a single tertiary center. Risk factors among current and former smokers, including exposure to environmental tobacco smoke, dust, fumes, chemicals, history of emphysema, and family history of cancer, predict an increased risk for cancer [124]. Finally, the Liverpool Lung Project model, validated in multiple independent populations, is based on data about smoking duration, history of pneumonia or cancer, family history of lung cancer, and asbestos exposure to predict five-year lung cancer risk [125,126].
Recent research has focused on identifying biomarkers of lung cancer risk, aggressive behavior among early cancers, and prognosis. These biomarkers may be produced by neoplastic cells themselves, the tumor microenvironment, or the host. A variety of methods to identify biosignatures utilizing tissue- and biofluids-based assays have been tested and include genome-wide association studies (GWAS), epigenetics, microRNA, and proteomics [127]. Using GWAS, several single nucleotide polymorphisms (SNPs) have been identified on specific chromosomal loci – such as the 15q25 locus – that are associated with tobacco exposure and lung cancer [128]. It remains to be seen whether these SNPs can be utilized in the clinical setting to assess lung cancer risk. Recent epigenetic studies have shown promise in risk stratification, with one case-control study using methylation of genes in sputum to identify asymptomatic patients with stage I lung cancer [129]. Several studies have assessed circulating microRNA biomarkers. A 34-microRNA signature was reported to identify early-stage NSCLC patients with 80% accuracy [130], while others have been shown to predict recurrent disease [131] in plasma, surgically resected specimens, and in small biopsies [132]. Similarly, a 24-micro RNA signature [133], validated in a correlative study within the MILD CT-screening trial, was found to reduce by five-fold the false positive rate after low-dose CT [134]. Finally, serum proteomic signatures have been integrated with CT imaging features to predict lung cancer diagnosis in subjects with indeterminate lung nodules [135]. A seven-autoantibody signature has been shown to have high specificity for lung cancer-associated antigens for early detection of lung cancer in a high-risk population, and also in distinguishing benign from malignant disease in CT-detected lung nodules; prospective validation is ongoing [136]. Ajona and colleagues demonstrated that C4d, a degradation product of complement activation, was elevated in tumors, bronchoalveolar lavage fluid, and plasma samples from stage I–II lung cancer patients compared to controls. Additionally, C4d levels were associated with worse survival and increased lung cancer risk in screen-detected lung nodules [137]. Despite a substantial progress in biomarker discovery, challenges that remain include selection of appropriate candidate signatures based on tumor-specificity and high-throughput approaches, genetic heterogeneity of tumors, and reproducibility in external validation studies [138,139].
Special Populations
Lung cancer incidence is rising in women and has in fact more than doubled since the mid1970s. This increase has been attributed to increased susceptibility in women compared with men, although studies have found conflicting results. Data from the UK’s Health Improvement Network showed that female heavy smokers (>20 cigarettes daily) had a greater odds of developing lung cancer than men with comparable smoking histories, with an adjusted OR of 19.2 (95% CI: 17.1–21.3) in women versus 13.0 (95% CI: 11.7–14.5) in men [140]. However, a large prospective cohort study in the US disputed this increased susceptibility to lung cancer given equal smoking exposure [141,142]. Rising rates of lung cancer in women have also been attributed to genetic variants, environmental exposures, hormonal factors, and oncogenic viruses [141,143]. The role of reproductive and hormonal factors remains controversial, with mixed results in studies evaluating associations between parity, age at menarche, and menopause. Although multiple case-control studies report increased lung cancer risk with exogenous hormone therapy [144,145,146], prospective cohort studies show equal lung cancer rates after adjusting for smoking rates [144,145,147,148,149,150]. HPV infection has been implicated in the pathogenesis of lung cancer in women in Asia, but in the US, infection rates in tumors have been much lower [141], and one Finnish study of 311 women with lung cancer, found no evidence of increased risk for lung cancer with HPV 16 and 18 type-specific infections among both nonsmokers and smokers [151]. Finally, with regard to diet, a Japanese prospective cohort study of 126 newly diagnosed women with lung cancer found only a trend toward an association with plasma genistein, an isoflavone found in soy and previously shown to act as estrogen agonists and antagonists [152]. Although adenocarcinoma is the most common histologic subtype in both genders, women have an even higher predominance of this cell type and have a higher likelihood of developing adenocarcinoma in situ, a preinvasive lesion [141]. Additionally, several gender differences in lung cancer mutations have been described. EGFR mutations are more prevalent in women, especially in non-smokers [153]. In particular, the L858R mutation has been shown to be associated with genetic polymorphisms related to estrogen biosynthesis and metabolism in never-smoking females with lung adenocarcinomas [153]. In one large study of the molecular epidemiology of lung cancer, the KRAS G12C mutation, the most common G>T transversion mutation in smokers, was more frequent in women, particularly of younger age [42]. Additionally, women with lung cancer have been shown in multiple studies to have better survival rates than men across different age groups, disease stage, and treatment types [3,141,154].
Approximately 10 to 20% of lung cancers occur in never-smokers with a much higher incidence in women than in men [155]. In fact, in South Asia, it is estimated that 83% of women with lung cancer may be never-smokers [156]. It is unclear if never-smoking Asian women who emigrate to the US and adopt western lifestyles continue to have elevated risks of lung cancer, although prior literature in gastric cancer appears to suggest that risk is environmental in nature [157]. Additional risk factors thought to contribute to lung cancer in never-smokers are environmental and occupational exposures and genetic susceptibility [156]. Never-smoking women present at a more advanced stage, and studies from Asia have reported an earlier age at diagnosis compared with smokers. However, in the US and Europe, never-smokers and ever-smokers are diagnosed with lung cancer at a similar age [156]. With regard to histology, never-smokers have a higher prevalence of adenocarcinoma [156]. Additionally, while genomic mutations occur more frequently in current and former smokers compared with never-smokers, the latter have a higher prevalence of driver mutations including EGFR and ALK-EML4 [158,159]. The EGFR mutation via an exon 19 deletion or exon 21 mutation is found in up to 40–60% of never-smokers [42,43,160]. HER2 mutations, a member of the EGFR family, also occurs predominantly in never-smokers [44]. In contrast, KRAS and BRAF mutations occur mainly in former and current smokers [161]. Moreover, approximately two-thirds of patients with ALK-EML4 rearrangements are never-smokers [45,46,162]. Finally, never-smokers exhibit improved survival compared with smokers, even after adjusting for known prognostic factors [156].
Since the advent of antiretroviral therapy in the 1990s, cancer mortality among human immunodeficiency virus (HIV) patients has shifted from acquired immunodeficiency syndrome (AIDS)-related malignancies to solid-organ cancers. Lung cancer incidence is increased two- to four-fold in the HIV-infected population compared with the general population and occurs with a lower pack-year rate of smoking [163,164,165]. Using the Veterans Affairs Central Cancer Registry, Sigel et al. found HIV to be an independent risk factor for the development of lung cancer, with an incidence rate ratio of 1.7 (95% CI: 1.5–1.9) [166]. Multiple cohorts have shown that there is no difference in age, stage at presentation, or histology between HIV-positive and -negative patients [166,167]. Lung cancer has also been found to be more aggressive in HIV patients, and positive HIV status has been associated with greater risk of lung cancer-specific mortality [168]. However, cancer-related survival has been shown to be better in patients with CD4 counts > 200 cells/mL [168,169,170]. Additionally, in subjects undergoing surgical resection, HIV patients have been found to have worse post-operative pulmonary and infectious complications as well as shorter median time to cancer progression [169].
Conclusions
This review has examined international trends in lung cancer epidemiology. Emerging economies and developing countries face many challenges in initiating tobacco cessation campaigns while also addressing environmental risk factors and cultural barriers. Over the past three to four decades, in contrast, industrialized nations have seen large declines in cigarette smoking and, consequently, lung cancer. However, despite knowledge gains in tumor biology that have led to targeted therapies, mortality from lung cancer remains high for most patients around the world. Future directions must include improvements in early detection and technological advances in genomics and genetics to achieve a more personalized approach to therapy and ultimately improve lung cancer survival.
Funding Statement
National Institutes of Health grant 1 RO1 CA163772.
Funding Information
National Institutes of Health grant 1 RO1 CA163772.
All authors had access to the data and a role in writing the manuscript.
Competing Interests
JAB and CAP have no conflicts of interests to disclose. Dr. Wisnivesky is a member of the research board of EHE International, has received consulting honorarium from Merck, Quintiles and AstraZeneca and research grants from Sanofi and Quorum.
References
- 1.Fitzmaurice C, Dicker D, Pain A, et al. The Global Burden of Cancer 2013. JAMA Oncology. 2015; 1(4): 505–527. DOI: 10.1001/jamaoncol.2015.0735 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J and Jemal A. Global cancer statistics, 2012. CA Cancer J Clin; 2015. DOI: 10.3322/caac.21262 [DOI] [PubMed] [Google Scholar]
- 3.Thun MJ, Carter BD, Feskanich D, et al. 50-year trends in smoking-related mortality in the United States. The New England Journal of Medicine. 2013; 368(4): 351–364. DOI: 10.1056/NEJMsa1211127 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Youlden D, Cramb S and Baade P. The international epidemiology of lung cancer: Geographic distribution and secular trends. Journal of Thoracic Oncology. 2008; 3(8): 819–831. DOI: 10.1097/JTO.0b013e31818020eb [DOI] [PubMed] [Google Scholar]
- 5.Jemal A, Center MM, DeSantis C and Ward EM. Global patterns of cancer incidence and mortality rates and trends. Cancer Epidemiology Biomarkers & Prevention. 2010; 19(8): 1893–1907. DOI: 10.1158/1055-9965.EPI-10-0437 [DOI] [PubMed] [Google Scholar]
- 6.Goss PE, Strasser-Weippl K, Lee-Bychkovsky BL, et al. Challenges to effective cancer control in China, India, and Russia. The Lancet Oncology. 2014; 15(5): 489–538. DOI: 10.1016/S1470-2045(14)70029-4 [DOI] [PubMed] [Google Scholar]
- 7.Siegel R, Ma J, Zou Z and Jemal A. Cancer statistics, 2014. CA: A Cancer Journal for Clinicians. 2014; 64(1): 9–29. DOI: 10.3322/caac.21208 [DOI] [PubMed] [Google Scholar]
- 8.Freedman ND, Abnet CC, Caporaso NE, et al. Impact of changing US cigarette smoking patterns on incident cancer: Risks of 20 smoking-related cancers among the women and men of the NIH-AARP cohort. Int J Epidemiol; 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Meza R, Meernik C, Jeon J and Cote ML. Lung cancer incidence trends by gender, race and histology in the United States, 1973–2010. PLoS One. 2015; 10(3): e0121323 DOI: 10.1371/journal.pone.0121323 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Trinh QD, Nguyen PL, Leow JJ, et al. Cancer-specific mortality of Asian Americans diagnosed with cancer: A nationwide population-based assessment. J Natl Cancer Inst. 2015; 107(6). DOI: 10.1093/jnci/djv054 [DOI] [PubMed] [Google Scholar]
- 11.Bach PB, Cramer LD, Warren JL and Begg CB. Racial differences in the treatment of early-stage lung cancer. New England Journal of Medicine. 1999; 341(16): 1198–1205. DOI: 10.1056/NEJM199910143411606 [DOI] [PubMed] [Google Scholar]
- 12.Wisnivesky JP, McGinn T, Henschke C, Hebert P, Iannuzzi MC and Halm EA. Ethnic disparities in the treatment of stage I non–small cell lung cancer. American Journal of Respiratory and Critical Care Medicine. 2005; 171(10): 1158–1163. DOI: 10.1164/rccm.200411-1475OC [DOI] [PubMed] [Google Scholar]
- 13.Ward E, Jemal A, Cokkinides V, et al. Cancer disparities by race/ethnicity and socioeconomic status. CA Cancer J Clin. 2004; 54(2): 78–93. DOI: 10.3322/canjclin.54.2.78 [DOI] [PubMed] [Google Scholar]
- 14.UK CR. Lung cancer incidence projections to 2024: Future rates and numbers of new cases in Great Britain and the UK. Cancer Stats Cancer Projections Series; 2009. [Google Scholar]
- 15.Lortet-Tieulent J, Renteria E, Sharp L, et al. Convergence of decreasing male and increasing female incidence rates in major tobacco-related cancers in Europe in 1988–2010. European Journal of Cancer. [DOI] [PubMed] [Google Scholar]
- 16.Bray FI and Weiderpass E. Lung cancer mortality trends in 36 European countries: Secular trends and birth cohort patterns by sex and region 1970–2007. International Journal of Cancer. 2010; 126(6): 1454–1466. DOI: 10.1002/ijc.24855 [DOI] [PubMed] [Google Scholar]
- 17.Torre LA, Siegel RL, Ward EM and Jemal A. International variation in lung cancer mortality rates and trends among women. Cancer Epidemiology Biomarkers & Prevention. 2014; 23(6): 1025–1036. DOI: 10.1158/1055-9965.EPI-13-1220 [DOI] [PubMed] [Google Scholar]
- 18.Malvezzi M, Bosetti C, Rosso T, et al. Lung cancer mortality in European men: Trends and predictions. Lung Cancer. 2013; 80(2): 138–145. DOI: 10.1016/j.lungcan.2013.01.020 [DOI] [PubMed] [Google Scholar]
- 19.Malvezzi M, Bertuccio P, Levi F, La Vecchia C and Negri E. European cancer mortality predictions for the year 2014. Annals of Oncology; 2014. DOI: 10.1093/annonc/mdu138 [DOI] [PubMed] [Google Scholar]
- 20.Gilhodes J, Belot A, Bouvier AM, et al. Incidence of major smoking-related cancers: Trends among adults aged 20–44 in France from 1982 to 2012. Cancer Epidemiol. 2015; 39(5): 707–713. DOI: 10.1016/j.canep.2015.07.001 [DOI] [PubMed] [Google Scholar]
- 21.Van der Heyden JHA, Schaap MM, Kunst AE, et al. Socioeconomic inequalities in lung cancer mortality in 16 European populations. Lung Cancer. 2009; 63(3): 322–330. DOI: 10.1016/j.lungcan.2008.06.006 [DOI] [PubMed] [Google Scholar]
- 22.Walters S, Maringe C, Coleman MP, et al. Lung cancer survival and stage at diagnosis in Australia, Canada, Denmark, Norway, Sweden and the UK: A population-based study, 2004–2007. Thorax. 2013; 68(6): 551–564. DOI: 10.1136/thoraxjnl-2012-202297 [DOI] [PubMed] [Google Scholar]
- 23.De Angelis R, Sant M, Coleman MP, et al. Cancer survival in Europe 1999–2007 by country and age: Results of EUROCARE-5 – A population-based study. The Lancet Oncology. 2014; 15(1): 23–34. DOI: 10.1016/S1470-2045(13)70546-1 [DOI] [PubMed] [Google Scholar]
- 24.Francisci S, Minicozzi P, Pierannunzio D, et al. Survival patterns in lung and pleural cancer in Europe 1999–2007: Results from the EUROCARE-5 study. European Journal of Cancer (Oxford, England: 1990); 2015. [DOI] [PubMed] [Google Scholar]
- 25.Ferlay J, Soerjomataram I, Ervik M, et al. GLOBOCAN 2012 v1.0. Cancer Incidence and Mortality Worldwide: IARC CancerBase No. 11 2013. http://globocan.iarc.fr. Accessed July 18, 2014.
- 26.Toyoda Y, Nakayama T, Ioka A and Tsukuma H. Trends in lung cancer incidence by histological type in Osaka, Japan. Japanese Journal of Clinical Oncology. 2008; 38(8): 534–539. DOI: 10.1093/jjco/hyn072 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Woo T, Okudela K, Mitsui H, et al. Prognostic value of the IASLC/ATS/ERS classification of lung adenocarcinoma in stage I disease of Japanese cases. Pathology International. 2012; 62(12): 785–791. DOI: 10.1111/pin.12016 [DOI] [PubMed] [Google Scholar]
- 28.O’Neill J. Building Better Global Economic BRICs; 2001(Global Economics Paper No: 66). [Google Scholar]
- 29.Chatenoud L, Bertuccio P, Bosetti C, et al. Trends in cancer mortality in Brazil, 1980–2004. European Journal of Cancer Prevention. 2010; 19(2): 79–86. DOI: 10.1097/CEJ.0b013e32833233be [DOI] [PubMed] [Google Scholar]
- 30.Souza MC, Vasconcelos AG and Cruz OG. Trends in lung cancer mortality in Brazil from the 1980s into the early 21st century: Age-period-cohort analysis. Cad Saude Publica; 2012. (1678-4464 [Electronic]). [DOI] [PubMed] [Google Scholar]
- 31.Singh N, Aggarwal AN, Gupta D, Behera D and Jindal SK. Unchanging clinico-epidemiological profile of lung cancer in North India over three decades. Cancer Epidemiology. 2010; 34(1): 101–104. DOI: 10.1016/j.canep.2009.12.015 [DOI] [PubMed] [Google Scholar]
- 32.Sharma PK and Bansal R. Profile of lung cancer in predominantly Bidi smoking rural population of northern Himachal Pradesh. Indian J Chest Dis Allied Sci. 2013; 55(2): 75–78. [PubMed] [Google Scholar]
- 33.Zhang Y, Tao S, Shen H and Ma J. Inhalation exposure to ambient polycyclic aromatic hydrocarbons and lung cancer risk of Chinese population. Proc Natl Acad Sci USA. (1091-6490 [Electronic]). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Chen ZM, Peto R, Iona A, et al. Emerging tobacco-related cancer risks in China: A nationwide, prospective study of 0.5 million adults. Cancer. 2015; 121(Suppl 17): 3097–3106. DOI: 10.1002/cncr.29560 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.She J, Yang P, Hong Q and Bai C. Lung cancer in China: Challenges and interventions. Chest. 2013; 143(4): 1117–1126. DOI: 10.1378/chest.11-2948 [DOI] [PubMed] [Google Scholar]
- 36.Jemal A, Bray F, Forman D, et al. Cancer burden in Africa and opportunities for prevention. Cancer. 2012; 118(18): 4372–4384. DOI: 10.1002/cncr.27410 [DOI] [PubMed] [Google Scholar]
- 37.Ferlay J, Soerjomataram I, Ervik M, et al. GLOBOCAN 2012 v1.0. Cancer Incidence and Mortality Worldwide: IARC CancerBase No. 11 2013. http://globocan.iarc.fr. Accessed October 29, 2014.
- 38.Travis W, Brambilla E, Noguchi M, et al. International Association for the Study of Lung Cancer/American Thoracic Society/European Respiratory Society international multidisciplinary classification of lung adenocarcinoma. J Thorac Oncol. 2011; 6(2): 244–285. DOI: 10.1097/JTO.0b013e318206a221 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lortet-Tieulent J, Soerjomataram I, Ferlay J, Rutherford M, Weiderpass E and Bray F. International trends in lung cancer incidence by histological subtype: Adenocarcinoma stabilizing in men but still increasing in women. Lung Cancer. 2014; 84(1): 13–22. DOI: 10.1016/j.lungcan.2014.01.009 [DOI] [PubMed] [Google Scholar]
- 40.Yoshizawa A, Motoi N, Riely GJ, et al. Impact of proposed IASLC/ATS/ERS classification of lung adenocarcinoma: Prognostic subgroups and implications for further revision of staging based on analysis of 514 stage I cases. Modern Pathology; 2011. (1530-0285 [Electronic]). [DOI] [PubMed] [Google Scholar]
- 41.Travis WD. Pathology of lung cancer. Clinics in Chest Medicine. 2011; 32(4): 669–692. DOI: 10.1016/j.ccm.2011.08.005 [DOI] [PubMed] [Google Scholar]
- 42.Dogan S, Shen R, Ang DC, et al. Molecular epidemiology of EGFR and KRAS mutations in 3,026 lung adenocarcinomas: Higher susceptibility of women to smoking-related KRAS-mutant cancers. Clinical Cancer Research. 2012; 18(22): 6169–6177. DOI: 10.1158/1078-0432.CCR-11-3265 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Shi Y, Au JS-K, Thongprasert S, et al. A prospective, molecular epidemiology study of EGFR mutations in Asian patients with advanced non–small cell lung cancer of adenocarcinoma histology (PIONEER). Journal of Thoracic Oncology. 2014; 9: 154–162. DOI: 10.1097/JTO.0000000000000033 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Ou SH. Lung cancer in never-smokers. Does smoking history matter in the era of molecular diagnostics and targeted therapy? Journal of Clinical Pathology. 2013; 66(10): 839–846. DOI: 10.1136/jclinpath-2012-201296 [DOI] [PubMed] [Google Scholar]
- 45.Koivunen JP, Mermel C, Zejnullahu K, et al. EML4-ALK fusion gene and efficacy of an ALK kinase inhibitor in lung cancer. Clinical Cancer Research. 2008; 14(13): 4275–4283. DOI: 10.1158/1078-0432.CCR-08-0168 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Shaw AT, Yeap BY, Mino-Kenudson M, et al. Clinical features and outcome of patients with non–small cell lung cancer who harbor EML4-ALK. Journal of Clinical Oncology. 2009; 27(26): 4247–4253. DOI: 10.1200/JCO.2009.22.6993 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Brawley OW, Glynn TJ, Khuri FR, Wender RC and Seffrin JR. The first Surgeon General’s report on smoking and health: The 50th anniversary. CA Cancer J Clin. 2014; 64(1): 5–8. DOI: 10.3322/caac.21210 [DOI] [PubMed] [Google Scholar]
- 48.Hecht SS. Tobacco smoke carcinogens and lung cancer. Journal of the National Cancer Institute. 1999; 91(14): 1194–1210. DOI: 10.1093/jnci/91.14.1194 [DOI] [PubMed] [Google Scholar]
- 49.Boffetta P, Pershagen G, Jöckel K-H, et al. Cigar and pipe smoking and lung cancer risk: A multicenter study from Europe. Journal of the National Cancer Institute. 1999; 91(8): 697–701. DOI: 10.1093/jnci/91.8.697 [DOI] [PubMed] [Google Scholar]
- 50.Oberg M, Jaakkola MS, Woodward A, Peruga A and Pruss-Ustun A. Worldwide burden of disease from exposure to second-hand smoke: A retrospective analysis of data from 192 countries. Lancet. 2011; 377(9760): 139–146. DOI: 10.1016/S0140-6736(10)61388-8 [DOI] [PubMed] [Google Scholar]
- 51.IARC. Tobacco Smoke and Involuntary Smoking IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. 2002; 83 Lyon, France. [PMC free article] [PubMed] [Google Scholar]
- 52.US Department of Health and Human Services. The Health Consequences of Involuntary Exposure to Tobacco Smoke: A Report of the Surgeon General In: U.S. Department of Health and Human Services, Coordinating Center for Health Promotion, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health. Rockville, MD; 2006. [Google Scholar]
- 53.US Department of Health and Human Services. How Tobacco Smoke Causes Disease: The Biology and Behavioral Basis for Smoking-Attributable Disease In: Department of Health and Human Services, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health. 2010; Atlanta, GA. [Google Scholar]
- 54.Agency USEP. Respiratory Health Effects of Passive Smoking (Also Known as Exposure to Secondhand Smoke or Environmental Tobacco Smoke—ETS). In: Agency USEP; 1992. [Google Scholar]
- 55.Kim CH, Lee YA, Hung RJ, et al. Secondhand tobacco smoke exposure and lung adenocarcinoma in situ/minimally invasive adenocarcinoma (AIS/MIA). In: Cancer Epidemiology, Biomarkers & Prevention: A Publication of the American Association for Cancer Research, Cosponsored by the American Society of Preventive Oncology; 2015. DOI: 10.1158/1055-9965.EPI-15-0436 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Abrams DB. Promise and peril of e-cigarettes: Can disruptive technology make cigarettes obsolete? JAMA. 2014; 311(2): 135–136. DOI: 10.1001/jama.2013.285347 [DOI] [PubMed] [Google Scholar]
- 57.Wang B, King BA, Corey CG, Arrazola RA and Johnson SE. Awareness and use of non-conventional tobacco products among U.S. students, 2012. American Journal of Preventive Medicine. 2014; 47(2, Supplement 1): S36–S52. DOI: 10.1016/j.amepre.2014.05.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Agaku IT, King BA, Husten CG, et al. Tobacco product use among adults – United States, 2012–2013. Morbidity and Mortality Weekly Report. 2014; 63(25): 542–547. [PMC free article] [PubMed] [Google Scholar]
- 59.Dutra LM and Glantz SA. Electronic cigarettes and conventional cigarette use among US adolescents: A cross-sectional study. JAMA Pediatrics. 2014; 168(7): 610–617. DOI: 10.1001/jamapediatrics.2013.5488 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Kmietowicz Z. Use of e-cigarettes in UK has tripled in two years, finds survey. 2014; 348. [DOI] [PubMed] [Google Scholar]
- 61.Park SJ, Walser TC, Perdomo C, et al. Abstract B16: The effect of e-cigarette exposure on airway epithelial cell gene expression and transformation. Clinical Cancer Research. 2014; 20(2 Supplement): B16 DOI: 10.1158/1078-0432.14AACRIASLC-B16 [DOI] [Google Scholar]
- 62.Kurmi OP, Arya PH, Lam K-BH, Sorahan T and Ayres JG. Lung cancer risk and solid fuel smoke exposure: A systematic review and meta-analysis. European Respiratory Journal. 2012; 40(5): 1228–1237. DOI: 10.1183/09031936.00099511 [DOI] [PubMed] [Google Scholar]
- 63.Barone-Adesi F, Chapman RS, Silverman DT, et al. Risk of lung cancer associated with domestic use of coal in Xuanwei, China: Retrospective cohort study. BMJ (Clinical Research Ed.). 2012; 345: e5414 DOI: 10.1136/bmj.e5414 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Hosgood HD, 3rd, Boffetta P, Greenland S, et al. In-home coal and wood use and lung cancer risk: A pooled analysis of the International Lung Cancer Consortium. Environmental Health Perspectives. 2010; 118(12): 1743–1747. DOI: 10.1289/ehp.1002217 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Hosgood HD, Wei H, Sapkota A, et al. Household coal use and lung cancer: Systematic review and meta-analysis of case-control studies, with an emphasis on geographic variation. International Journal of Epidemiology. 2011; 40(3): 719–728. DOI: 10.1093/ije/dyq259 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Tang L, Lim WY, Eng P, et al. Lung cancer in Chinese women: Evidence for an interaction between tobacco smoking and exposure to inhalants in the indoor environment. Environmental Health Perspectives. 2010; 118(9): 1257–1260. DOI: 10.1289/ehp.0901587 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Young RP, Hopkins RJ, Christmas T, Black PN, Metcalf P and Gamble GD. COPD prevalence is increased in lung cancer, independent of age, sex and smoking history. European Respiratory Journal. 2009; 34(2): 380–386. DOI: 10.1183/09031936.00144208 [DOI] [PubMed] [Google Scholar]
- 68.de Torres JP, Marin JM, Casanova C, et al. Lung cancer in patients with chronic obstructive pulmonary disease – Incidence and predicting factors. Am J Respir Crit Care Med. 2011; 184(8): 913–919. DOI: 10.1164/rccm.201103-0430OC [DOI] [PubMed] [Google Scholar]
- 69.Wilson DO, Weissfeld JL, Balkan A, et al. Association of radiographic emphysema and airflow obstruction with lung cancer. American Journal of Respiratory and Critical Care Medicine. 2008; 178(7): 738–744. DOI: 10.1164/rccm.200803-435OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Zulueta JJ, Wisnivesky JP, Henschke CI, et al. Emphysema scores predict death from COPD and lung cancer. Chest. 2012; 141(5): 1216–1223. DOI: 10.1378/chest.11-0101 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Brenner DR, Boffetta P, Duell EJ, et al. Previous lung diseases and lung cancer risk: A pooled analysis from the International Lung Cancer Consortium. American Journal of Epidemiology. 2012; 176(7): 573–585. DOI: 10.1093/aje/kws151 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Turner MC, Chen Y, Krewski D, Calle EE and Thun MJ. Chronic obstructive pulmonary disease is associated with lung cancer mortality in a prospective study of never-smokers. American Journal of Respiratory and Critical Care Medicine. 2007; 176(3): 285–290. DOI: 10.1164/rccm.200612-1792OC [DOI] [PubMed] [Google Scholar]
- 73.Young RP, Whittington CF, Hopkins RJ, et al. Chromosome 4q31 locus in COPD is also associated with lung cancer. European Respiratory Journal. 2010; 36(6): 1375–1382. DOI: 10.1183/09031936.00033310 [DOI] [PubMed] [Google Scholar]
- 74.Brenner DR, McLaughlin JR and Hung RJ. Previous lung diseases and lung cancer risk: A systematic review and meta-analysis. PLoS ONE. 2011; 6(3): e17479 DOI: 10.1371/journal.pone.0017479 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Nelson HK and Kelsey KT. The molecular epidemiology of asbestos and tobacco in lung cancer. Oncogene. 2002; 21: 7284–7288. DOI: 10.1038/sj.onc.1205804 [DOI] [PubMed] [Google Scholar]
- 76.Markowitz SB, Levin SM, Miller A and Morabia A. Asbestos, asbestosis, smoking, and lung cancer. New findings from the North American insulator cohort. Am J Respir Crit Care Med. 2013; 188(1): 90–96. DOI: 10.1164/rccm.201302-0257OC [DOI] [PubMed] [Google Scholar]
- 77.Olsson AC, Gustavsson P, Kromhout H, et al. Exposure to diesel motor exhaust and lung cancer risk in a pooled analysis from case-control studies in Europe and Canada. Am J Respir Crit Care Med. 2011; 183(7): 941–948. DOI: 10.1164/rccm.201006-0940OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Garshick E, Laden F, Hart JE, Davis ME, Eisen EA and Smith TJ. Lung cancer and elemental carbon exposure in trucking industry workers. Environmental Health Perspectives. 2012; 120(9): 1301–1306. DOI: 10.1289/ehp.1204989 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Silverman DT, Samanic CM, Lubin JH, et al. The diesel exhaust in miners study: A nested case-control study of lung cancer and diesel exhaust. Journal of the National Cancer Institute. 2012; 104(11): 855–868. DOI: 10.1093/jnci/djs034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Vermeulen R, Silverman DT, Garshick E, Vlaanderen J, Portengen L and Steenland K. Exposure-response estimates for diesel engine exhaust and lung cancer mortality based on data from three occupational cohorts. Environmental Health Perspectives. 2014; 122(2): 172–177. DOI: 10.1289/ehp.1306880 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Christian WJ, Huang B, Rinehart J and Hopenhayn C. Exploring geographic variation in lung cancer incidence in Kentucky using a spatial scan statistic: Elevated risk in the Appalachian coal-mining region. Public Health Reports (Washington, D.C.: 1974). 2011; 126(6): 789–796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Olsson A, Kromhout H, Agostini M, et al. A case-control study of lung cancer nested in a cohort of European asphalt workers. Environmental Health Perspectives. 2010; 118(10): 1418–1424. DOI: 10.1289/ehp.0901800 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Hogstedt C, Jansson C, Hugosson M, Tinnerberg H and Gustavsson P. Cancer incidence in a cohort of Swedish chimney sweeps, 1958–2006. American Journal of Public Health. 2013; 103(9): 1708–1714. DOI: 10.2105/AJPH.2012.300860 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Guha N, Merletti F, Steenland NK, Altieri A, Cogliano V and Straif K. Lung cancer risk in painters: A meta-analysis. Environmental Health Perspectives. 2010; 118(3): 303–312. DOI: 10.1289/ehp.0901402 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.De Matteis S, Consonni D, Lubin JH, et al. Impact of occupational carcinogens on lung cancer risk in a general population. International Journal of Epidemiology. 2012; 41(3): 711–721. DOI: 10.1093/ije/dys042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Vida S, Pintos J, Parent M-É, Lavoué J and Siemiatycki J. Occupational exposure to silica and lung cancer: Pooled analysis of two case-control studies in Montreal, Canada. Cancer Epidemiology Biomarkers & Prevention. 2010; 19(6): 1602–1611. DOI: 10.1158/1055-9965.EPI-10-0015 [DOI] [PubMed] [Google Scholar]
- 87.Raaschou-Nielsen O, Andersen ZJ, Beelen R, et al. Air pollution and lung cancer incidence in 17 European cohorts: Prospective analyses from the European Study of Cohorts for Air Pollution Effects (ESCAPE). The Lancet Oncology. 2013; 14(9): 813–822. DOI: 10.1016/S1470-2045(13)70279-1 [DOI] [PubMed] [Google Scholar]
- 88.Hystad P, Demers PA, Johnson KC, Carpiano RM and Brauer M. Long-term residential exposure to air pollution and lung cancer risk. Epidemiology. 2013; 24(5): 762–772. [DOI] [PubMed] [Google Scholar]
- 89.Raaschou-Nielsen O, Andersen ZJ, Hvidberg M, et al. Lung cancer incidence and long-term exposure to air pollution from traffic. Environmental Health Perspectives. 2011; 119(6): 860–865. DOI: 10.1289/ehp.1002353 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Raaschou-Nielsen O, Bak H, Sørensen M, et al. Air pollution from traffic and risk for lung cancer in three Danish cohorts. Cancer Epidemiology Biomarkers & Prevention. 2010; 19(5): 1284–1291. DOI: 10.1158/1055-9965.EPI-10-0036 [DOI] [PubMed] [Google Scholar]
- 91.Carey IM, Atkinson RW, Kent AJ, van Staa T, Cook DG and Anderson HR. Mortality associations with long-term exposure to outdoor air pollution in a national English cohort. Am J Respir Crit Care Med. 2013; 187(11): 1226–1233. DOI: 10.1164/rccm.201210-1758OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Turner MC, Krewski D, Pope CA, 3rd, Chen Y, Gapstur SM and Thun MJ. Long-term ambient fine particulate matter air pollution and lung cancer in a large cohort of never-smokers. Am J Respir Crit Care Med. 2011; 184(12): 1374–1381. DOI: 10.1164/rccm.201106-1011OC [DOI] [PubMed] [Google Scholar]
- 93.Pope CA, 3rd, Burnett RT, Turner MC, et al. Lung cancer and cardiovascular disease mortality associated with ambient air pollution and cigarette smoke: Shape of the exposure-response relationships. Environmental Health Perspectives. 2011; 119(11): 1616–1621. DOI: 10.1289/ehp.1103639 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Oberoi S, Barchowsky A and Wu F. The global burden of disease for skin, lung and bladder cancer caused by arsenic in food. Cancer Epidemiology Biomarkers & Prevention; 2014. DOI: 10.1158/1055-9965.EPI-13-1317 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Ferreccio C, Yuan Y, Calle J, et al. Arsenic, tobacco smoke, and occupation: associations of multiple agents with lung and bladder cancer. Epidemiology. 2013; 24(6): 898–905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Heck JE, Andrew AS, Onega T, et al. Lung cancer in a U.S. population with low to moderate arsenic exposure. Environmental Health Perspectives. 2009; 117(11): 1718–1723. DOI: 10.1289/ehp.0900566 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Turner MC, Krewski D, Chen Y, Pope CA, Gapstur S and Thun MJ. Radon and lung cancer in the American Cancer Society cohort. Cancer Epidemiology Biomarkers & Prevention. 2011; 20(3): 438–448. DOI: 10.1158/1055-9965.EPI-10-1153 [DOI] [PubMed] [Google Scholar]
- 98.Barros-Dios JM, Ruano-Ravina A, Pérez-Ríos M, Castro-Bernárdez M, Abal-Arca J and Tojo-Castro M. Residential radon exposure, histologic types, and lung cancer risk. A case-control study in Galicia, Spain. Cancer Epidemiology Biomarkers & Prevention. 2012; 21(6): 951–958. DOI: 10.1158/1055-9965.EPI-12-0146-T [DOI] [PubMed] [Google Scholar]
- 99.Lam TK, Ruczinski I, Helzlsouer KJ, Shugart YY, Caulfield LE and Alberg AJ. Cruciferous vegetable intake and lung cancer risk: A nested case-control study matched on cigarette smoking. Cancer Epidemiology Biomarkers & Prevention. 2010; 19(10): 2534–2540. DOI: 10.1158/1055-9965.EPI-10-0475 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Büchner FL, Bueno-de-Mesquita HB, Ros MM, et al. Variety in fruit and vegetable consumption and the risk of lung cancer in the European prospective investigation into cancer and nutrition. Cancer Epidemiology Biomarkers & Prevention. 2010; 19(9): 2278–2286. DOI: 10.1158/1055-9965.EPI-10-0489 [DOI] [PubMed] [Google Scholar]
- 101.Lam TK, Shao S, Zhao Y, et al. Influence of quercetin-rich food intake on microRNA expression in lung cancer tissues. Cancer Epidemiology Biomarkers & Prevention. 2012; 21(12): 2176–2184. DOI: 10.1158/1055-9965.EPI-12-0745 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Mahabir S, Forman MR, Dong YQ, Park Y, Hollenbeck A and Schatzkin A. Mineral intake and lung cancer risk in the NIH-American Association of Retired Persons diet and health study. Cancer Epidemiology Biomarkers & Prevention. 2010; 19(8): 1976–1983. DOI: 10.1158/1055-9965.EPI-10-0067 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Takata Y, Shu X-O, Yang G, et al. Calcium intake and lung cancer risk among female nonsmokers: A report from the Shanghai women’s health study. Cancer Epidemiology Biomarkers & Prevention. 2013; 22(1): 50–57. DOI: 10.1158/1055-9965.EPI-12-0915-T [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Johansson M, Relton C, Ueland PM, et al. Serum B vitamin levels and risk of lung cancer. JAMA. 2010; 303(23): 2377–2385. DOI: 10.1001/jama.2010.808 [DOI] [PubMed] [Google Scholar]
- 105.Cheng TY, Lacroix AZ, Beresford SA, et al. Vitamin D intake and lung cancer risk in the Women’s Health Initiative. The American Journal of Clinical Nutrition. 2013; 98(4): 1002–1011. DOI: 10.3945/ajcn.112.055905 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Yang WS, Va P, Wong MY, Zhang HL and Xiang YB. Soy intake is associated with lower lung cancer risk: results from a meta-analysis of epidemiologic studies. The American Journal of Clinical Nutrition. 2011; 94(6): 1575–1583. DOI: 10.3945/ajcn.111.020966 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.The Alpha-Tocopherol BCCPSG. The effect of vitamin E and beta carotene on the incidence of lung cancer and other cancers in male smokers. New England Journal of Medicine. 1994; 330(15): 1029–1035. DOI: 10.1056/NEJM199404143301501 [DOI] [PubMed] [Google Scholar]
- 108.Fortmann SP, Burda BU, Senger CA, Lin JS and Whitlock EP. Vitamin and mineral supplements in the primary prevention of cardiovascular disease and cancer: An updated systematic evidence review for the U.S. Preventive Services Task Force. Annals of Internal Medicine. 2013; 159(12): 824–834. DOI: 10.7326/0003-4819-159-12-201312170-00729 [DOI] [PubMed] [Google Scholar]
- 109.Coté ML, Liu M, Bonassi S, et al. Increased risk of lung cancer in individuals with a family history of the disease: A pooled analysis from the International Lung Cancer Consortium. European Journal of Cancer. 2012; 48(13): 1957–1968. DOI: 10.1016/j.ejca.2012.01.038 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Matakidou A, Eisen T and Houlston RS. Systematic review of the relationship between family history and lung cancer risk. The British Journal of Cancer. 2005; 93(7): 825–833. DOI: 10.1038/sj.bjc.6602769 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Lan Q, Hsiung CA, Matsuo K, et al. Genome-wide association analysis identifies new lung cancer susceptibility loci in never-smoking women in Asia. Nature Genetics. 2012; 44(12): 1330–1335. DOI: 10.1038/ng.2456 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Wu X, Wang L, Ye Y, et al. Genome-wide association study of genetic predictors of overall survival for non–small cell lung cancer in never-smokers. Cancer Research. 2013; 73(13): 4028–4038. DOI: 10.1158/0008-5472.CAN-12-4033 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Schwartz AG, Prysak GM, Bock CH and Cote ML. The molecular epidemiology of lung cancer. Carcinogenesis. 2006; 28(3): 507–518. DOI: 10.1093/carcin/bgl253 [DOI] [PubMed] [Google Scholar]
- 114.Ohtsuka K, Ohnishi H, Kurai D, et al. Familial lung adenocarcinoma caused by the EGFR V843I germ-line mutation. Journal of Clinical Oncology. 2011; 29(8): e191–e192. DOI: 10.1200/JCO.2010.31.4492 [DOI] [PubMed] [Google Scholar]
- 115.Bach PB, Mirkin JN, Oliver TK, et al. Benefits and harms of CT screening for lung cancer: A systematic review. JAMA. 2012; 307(22): 2418–2429. DOI: 10.1001/jama.2012.5521 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Aberle DR, Adams AM, Berg CD, et al. Reduced lung cancer mortality with low-dose computed tomographic screening. The New England Journal of Medicine. 2011; 365(5): 395–409. DOI: 10.1056/NEJMoa1102873 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.van Iersel CA, de Koning HJ, Draisma G, et al. Risk-based selection from the general population in a screening trial: Selection criteria, recruitment and power for the Dutch-Belgian randomised lung cancer multi-slice CT screening trial (NELSON). International Journal of Cancer. 2007; 120(4): 868–874. DOI: 10.1002/ijc.22134 [DOI] [PubMed] [Google Scholar]
- 118.Infante M, Cavuto S, Lutman FR, et al. Long-term follow-up results of the DANTE trial, a randomized study of lung cancer screening with spiral computed tomography. Am J Respir Crit Care Med. 2015; 191(10): 1166–1175. DOI: 10.1164/rccm.201408-1475OC [DOI] [PubMed] [Google Scholar]
- 119.Reich JM. A critical appraisal of overdiagnosis: Estimates of its magnitude and implications for lung cancer screening. Thorax. 2008; 63(4): 377–383. DOI: 10.1136/thx.2007.079673 [DOI] [PubMed] [Google Scholar]
- 120.Tammemagi CM, Pinsky PF, Caporaso NE, et al. Lung cancer risk prediction: Prostate, lung, colorectal and ovarian cancer screening trial models and validation. Journal of the National Cancer Institute. 2011; 103(13): 1058–1068. DOI: 10.1093/jnci/djr173 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Etzel CJ and Bach PB. Estimating individual risk for lung cancer. Semin Respir Crit Care Med. 2011; 32(01): 003–009. DOI: 10.1055/s-0031-1272864 [DOI] [PubMed] [Google Scholar]
- 122.Bach PB, Kattan MW, Thornquist MD, et al. Variations in lung cancer risk among smokers. Journal of the National Cancer Institute. 2003; 95(6): 470–478. DOI: 10.1093/jnci/95.6.470 [DOI] [PubMed] [Google Scholar]
- 123.Cronin KA, Gail MH, Zou Z, Bach PB, Virtamo J and Albanes D. Validation of a model of lung cancer risk prediction among smokers. Journal of the National Cancer Institute. 2006; 98(9): 637–640. DOI: 10.1093/jnci/djj163 [DOI] [PubMed] [Google Scholar]
- 124.Spitz MR, Hong WK, Amos CI, et al. A risk model for prediction of lung cancer. Journal of the National Cancer Institute. 2007; 99(9): 715–726. DOI: 10.1093/jnci/djk153 [DOI] [PubMed] [Google Scholar]
- 125.Cassidy A, Duffy SW, Myles JP, Liloglou T and Field JK. Lung cancer risk prediction: A tool for early detection. International Journal of Cancer. 2007; 120(1): 1–6. DOI: 10.1002/ijc.22331 [DOI] [PubMed] [Google Scholar]
- 126.Raji OY, Duffy SW, Agbaje OF, et al. Predictive accuracy of the Liverpool lung project risk model for stratifying patients for computed tomography screening for lung cancer: A case-control and cohort validation study. Annals of Internal Medicine. 2012; 157(4): 242–250. DOI: 10.7326/0003-4819-157-4-201208210-00004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Powell CA, Halmos B and Nana-Sinkam SP. Update in lung cancer and mesothelioma 2012. American Journal of Respiratory and Critical Care Medicine. 2013; 188(2): 157–166. DOI: 10.1164/rccm.201304-0716UP [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Munafò MR, Timofeeva MN, Morris RW, et al. Association between genetic variants on chromosome 15q25 locus and objective measures of tobacco exposure. Journal of the National Cancer Institute. 2012; 104(10): 740–748. DOI: 10.1093/jnci/djs191 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Leng S, Do K, Yingling CM, et al. Defining a gene promoter methylation signature in sputum for lung cancer risk assessment. Clinical Cancer Research. 2012; 18(12): 3387–3395. DOI: 10.1158/1078-0432.CCR-11-3049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Bianchi F, Nicassio F, Marzi M, et al. A serum circulating miRNA diagnostic test to identify asymptomatic high-risk individuals with early stage lung cancer. Journal Article. 2011; 3(8): 495–503. DOI: 10.1002/emmm.201100154 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Lu Y, Govindan R, Wang L, et al. MicroRNA profiling and prediction of recurrence/relapse-free survival in stage I lung cancer. Carcinogenesis. 2012; 33(5): 1046–1054. DOI: 10.1093/carcin/bgs100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Huang W, Hu J, Yang D-W, et al. Two microRNA panels to discriminate three subtypes of lung carcinoma in bronchial brushing specimens. American Journal of Respiratory and Critical Care Medicine. 2012; 186(11): 1160–1167. DOI: 10.1164/rccm.201203-0534OC [DOI] [PubMed] [Google Scholar]
- 133.Boeri M, Verri C, Conte D, et al. MicroRNA signatures in tissues and plasma predict development and prognosis of computed tomography detected lung cancer. Proceedings of the National Academy of Sciences. 2011; 108(9): 3713–3718. DOI: 10.1073/pnas.1100048108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Sozzi G, Boeri M, Rossi M, et al. Clinical utility of a plasma-based miRNA signature classifier within computed tomography lung cancer screening: A correlative MILD trial study. Journal of Clinical Oncology. 2014; 32(8): 768–773. DOI: 10.1200/JCO.2013.50.4357 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Pecot CV, Li M, Zhang XJ, et al. Added value of a serum proteomic signature in the diagnostic evaluation of lung nodules. Cancer Epidemiology Biomarkers & Prevention. 2012; 21(5): 786–792. DOI: 10.1158/1055-9965.EPI-11-0932 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Jett JR, Peek LJ, Fredericks L, Jewell W, Pingleton WW and Robertson JFR. Audit of the autoantibody test, EarlyCDT®-Lung, in 1600 patients: An evaluation of its performance in routine clinical practice. Lung Cancer. 2014; 83(1): 51–55. DOI: 10.1016/j.lungcan.2013.10.008 [DOI] [PubMed] [Google Scholar]
- 137.Ajona D, Pajares MJ, Corrales L, et al. Investigation of complement activation product C4d as a diagnostic and prognostic biomarker for lung cancer. Journal of the National Cancer Institute. 2013; 105(18): 1385–1393. DOI: 10.1093/jnci/djt205 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Use of Biomarkers for Lung Cancer Screening. American Thoracic Society International Conference; 2014. [Google Scholar]
- 139.Hassanein M, Callison JC, Callaway-Lane C, Aldrich MC, Grogan EL and Massion PP. The state of molecular biomarkers for the early detection of lung cancer. Cancer Prevention Research. 2012; 5(8): 992–1006. DOI: 10.1158/1940-6207.CAPR-11-0441 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Powell HA, Iyen-Omofoman B, Hubbard RB, Baldwin DR and Tata LJ. The association between smoking quantity and lung cancer in men and women. Chest. 2013; 143(1): 123–129. DOI: 10.1378/chest.12-1068 [DOI] [PubMed] [Google Scholar]
- 141.Kligerman S and White C. Epidemiology of lung cancer in women: Risk factors, survival, and screening. AJR. American Journal of Roentgenology. 2011; 196(2): 287–295. DOI: 10.2214/AJR.10.5412 [DOI] [PubMed] [Google Scholar]
- 142.Bain C, Feskanich D, Speizer FE, et al. Lung cancer rates in men and women with comparable histories of smoking. Journal of the National Cancer Institute. 2004; 96(11): 826–834. DOI: 10.1093/jnci/djh143 [DOI] [PubMed] [Google Scholar]
- 143.Patel JD, Bach PB and Kris MG. Lung cancer in US women: A contemporary epidemic. JAMA. 2004; 291(14): 1763–1768. DOI: 10.1001/jama.291.14.1763 [DOI] [PubMed] [Google Scholar]
- 144.Brinton LA, Gierach GL, Andaya A, et al. Reproductive and hormonal factors and lung cancer risk in the NIH-AARP diet and health study cohort. Cancer Epidemiology Biomarkers & Prevention. 2011; 20(5): 900–911. DOI: 10.1158/1055-9965.EPI-10-1325 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Baik CS, Strauss GM, Speizer FE and Feskanich D. Reproductive factors, hormone use, and risk for lung cancer in postmenopausal women, the nurses’ health study. Cancer Epidemiology Biomarkers & Prevention. 2010; 19(10): 2525–2533. DOI: 10.1158/1055-9965.EPI-10-0450 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Paulus JK, Asomaning K, Kraft P, Johnson BE, Lin X and Christiani DC. Parity and risk of lung cancer in women. American Journal of Epidemiology. 2010; 171(5): 557–563. DOI: 10.1093/aje/kwp441 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Clague J, Reynolds P, Sullivan-Halley J, et al. Menopausal hormone therapy does not influence lung cancer risk: Results from the California teachers study. Cancer Epidemiology Biomarkers & Prevention. 2011; 20(3): 560–564. DOI: 10.1158/1055-9965.EPI-10-1182 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Kabat GC, Miller AB and Rohan TE. Reproductive and hormonal factors and risk of lung cancer in women: A prospective cohort study. International Journal of Cancer. 2007; 120(10): 2214–2220. DOI: 10.1002/ijc.22543 [DOI] [PubMed] [Google Scholar]
- 149.Chlebowski RT, Anderson GL, Manson JE, et al. Lung cancer among postmenopausal women treated with estrogen alone in the women’s health initiative randomized trial. Journal of the National Cancer Institute. 2010; 102(18): 1413–1421. DOI: 10.1093/jnci/djq285 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Chlebowski RT, Schwartz AG, Wakelee H, et al. Oestrogen plus progestin and lung cancer in postmenopausal women (Women’s Health Initiative trial): A post-hoc analysis of a randomised controlled trial. The Lancet. 374(9697): 1243–1251. DOI: 10.1016/S0140-6736(09)61526-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Simen-Kapeu A, Surcel H-M, Koskela P, Pukkala E and Lehtinen M. Lack of association between human papillomavirus type 16 and 18 infections and female lung cancer. Cancer Epidemiology Biomarkers & Prevention. 2010; 19(7): 1879–1881. DOI: 10.1158/1055-9965.EPI-10-0356 [DOI] [PubMed] [Google Scholar]
- 152.Shimazu T, Inoue M, Sasazuki S, et al. Plasma isoflavones and the risk of lung cancer in women: A nested case-control study in Japan. Cancer Epidemiology Biomarkers & Prevention. 2011; 20(3): 419–427. DOI: 10.1158/1055-9965.EPI-10-1025 [DOI] [PubMed] [Google Scholar]
- 153.Yang S-Y, Yang T-Y, Chen K-C, et al. EGFR L858R mutation and polymorphisms of genes related to estrogen biosynthesis and metabolism in never-smoking female lung adenocarcinoma patients. Clinical Cancer Research. 2011; 17(8): 2149–2158. DOI: 10.1158/1078-0432.CCR-10-2045 [DOI] [PubMed] [Google Scholar]
- 154.Nakamura H, Ando K, Shinmyo T, et al. Female gender is an independent prognostic factor in non–small cell lung cancer: A meta-analysis. Annals of Thoracic and Cardiovascular Surgery. 2011; 17(5): 469–480. DOI: 10.5761/atcs.oa.10.01637 [DOI] [PubMed] [Google Scholar]
- 155.Wakelee HA, Chang ET, Gomez SL, et al. Lung cancer incidence in never-smokers. Journal of Clinical Oncology. 2007; 25(5): 472–478. DOI: 10.1200/JCO.2006.07.2983 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Sun S, Schiller JH and Gazdar AF. Lung cancer in never smokers – A different disease. Nature Reviews. Cancer. 2007; 7(10): 778–790. DOI: 10.1038/nrc2190 [DOI] [PubMed] [Google Scholar]
- 157.Buell P and Dunn JE. Cancer mortality among Japanese ISSEI and NISEI of California. Cancer. 1965; 18(5): 656–664. DOI: [DOI] [PubMed] [Google Scholar]
- 158.Govindan R, Ding L, Griffith M, et al. Genomic landscape of non–small cell lung cancer in smokers and never-smokers. Cell. 2012; 150(6): 1121–1134. DOI: 10.1016/j.cell.2012.08.024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Paik PK, Johnson ML, D’Angelo SP, et al. Driver mutations determine survival in smokers and never-smokers with stage IIIB/IV lung adenocarcinomas. Cancer. 2012; 118(23): 5840–5847. DOI: 10.1002/cncr.27637 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Warth A, Penzel R, Lindenmaier H, et al. EGFR, KRAS, BRAF and ALK gene alterations in lung adenocarcinomas: Patient outcome, interplay with morphology and immunophenotype. European Respiratory Journal. 2014; 43(3): 872–883. DOI: 10.1183/09031936.00018013 [DOI] [PubMed] [Google Scholar]
- 161.Sequist LV, Heist RS, Shaw AT, et al. Implementing multiplexed genotyping of non–small cell lung cancers into routine clinical practice. Annals of Oncology. 2011; 22(12): 2616–2624. DOI: 10.1093/annonc/mdr489 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Solomon B, Varella-Garcia M and DR C. ALK Gene rearrangements – A new therapeutic target in a molecularly defined subset of non–small cell lung cancer. Journal of Thoracic Oncology. 2009; 4: 1450–1454. DOI: 10.1097/JTO.0b013e3181c4dedb [DOI] [PubMed] [Google Scholar]
- 163.Winstone TA, Man SF, Hull M, Montaner JS and Sin DD. Epidemic of lung cancer in patients with HIV infection. Chest. 2013; 143(2): 305–314. DOI: 10.1378/chest.12-1699 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Patel P, Hanson DL, Sullivan PS, et al. Incidence of types of cancer among HIV-infected persons compared with the general population in the United States, 1992–2003. Annals of Internal Medicine. 2008; 148(10): 728–736. DOI: 10.7326/0003-4819-148-10-200805200-00005 [DOI] [PubMed] [Google Scholar]
- 165.Kirk GD, Merlo C, O’Driscoll P, et al. HIV infection is associated with an increased risk for lung cancer, independent of smoking. Clinical Infectious Diseases. 2007; 45(1): 103–110. DOI: 10.1086/518606 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Sigel K, Wisnivesky J, Gordon K, et al. HIV as an independent risk factor for incident lung cancer. AIDS (London, England). 2012; 26(8): 1017–1025. DOI: 10.1097/QAD.0b013e328352d1ad [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Sigel K, Crothers K, Dubrow R, et al. Prognosis in HIV-infected patients with non–small cell lung cancer. British Journal of Cancer. 2013; 109(7): 1974–1980. DOI: 10.1038/bjc.2013.545 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Pakkala S, Chen Z, Rimland D, et al. Human immunodeficiency virus-associated lung cancer in the era of highly active antiretroviral therapy. Cancer. 2012; 118(1): 164–172. DOI: 10.1002/cncr.26242 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Hooker CM, Meguid RA, Hulbert A, et al. Human immunodeficiency virus infection as a prognostic factor in surgical patients with non–small cell lung cancer. The Annals of Thoracic Surgery. 2012; 93(2): 405–412. DOI: 10.1016/j.athoracsur.2011.11.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Hessol NA, Martinez-Maza O, Levine AM, et al. Lung cancer incidence and survival among HIV-infected and uninfected women and men. AIDS (London, England). 2015; 29(10): 1183–1193. DOI: 10.1097/QAD.0000000000000690 [DOI] [PMC free article] [PubMed] [Google Scholar]


