Abstract
Alzheimer’s disease (AD) is a neurodegenerative disorder characterized by macroscopic features such as cortical atrophy, narrowing of the gyri, widening of the sulci, and enlargement of the ventricles. At the cellular level, the pathological characteristics include the extracellular aggregation of β-amyloid (Aβ) forming senile plaques, and the intracellular accumulation of hyperphosphorylated tau proteins forming neurofibrillary tangles. AD leads to the progressive decline of cognitive, behavioral, and social abilities, with no effective treatment available currently. The pathophysiology of AD is complex, involving mechanisms such as immune dysregulation and lipid metabolism alterations. Immune cells, such as microglia, can identify and clear pathological aggregates like Aβ early in the disease. However, prolonged or excessive activation of immune cells may trigger chronic neuroinflammation, thereby accelerating neuronal damage and the progression of AD. Lipid metabolism plays a critical role in maintaining cell membrane structure and function, regulating the production and clearance of Aβ, and supplying energy to the brain. Disruptions in these processes are closely linked to the pathological progression of AD. The interaction between lipid metabolism and the immune system further exacerbates the disease progression of AD. In this review, we discuss the lipid metabolism and immune response in AD, summarize their intricate interactions, and highlight the complexity of the multifactorial pathogenic cascade, offering insights into new interventions targeting the immune-metabolic axis in AD.
Keywords: Alzheimer's disease, Immune response, Lipid metabolism, Immunometabolism, Molecular mechanism
Introduction
Alzheimer’s Disease (AD) is a common neurodegenerative disease characterized by progressive cognitive dysfunction and behavioral impairment in the elderly, and is one of the most prevalent forms of dementia worldwide. The main clinical manifestations of AD are the continuous deterioration of cognitive and memory function, the progressive decline in daily living abilities, and a variety of neurological, psychiatric symptoms and behavioral disorders [1]. Worldwide, at least 50 million individuals suffer from dementia, and this number is projected to reach 152 million by 2050 [2]. Currently, patients with AD often require long-term treatment and care, imposing significant economic and psychological burdens on both families and society [3, 4]. The pathological hallmarks of AD include the abnormal deposition of β-amyloid (Aβ) in the brain, forming amyloid plaques, and the pathological post-translational modifications and aggregation of tau protein within neurons, leading to neurofibrillary tangles [5]. Several hypotheses have been proposed to explain the potential mechanisms underlying AD pathogenesis, including the cholinergic hypothesis, the β-amyloid hypothesis, the tau hypothesis, the genetic hypothesis, the immune-inflammatory hypothesis, and the oxidative stress hypothesis [6]. Each of these theories explores different aspects of AD pathology; however, the exact etiology of the disease remains unclear. In recent years, increasing attention has been given to the roles of immunity and lipid metabolism in AD, highlighting their potential contributions to disease progression.
Immunity refers to the ability of an organism to recognize and defend against pathogens, foreign substances, and harmful agents through a series of physiological and biochemical processes. It includes innate immunity, mediated by macrophages and other immune cells, and adaptive immunity, driven by T cells and B cells. This self-defense mechanism is crucial for maintaining homeostasis. In the context of AD, inflammation reflects an innate immune response primarily mediated by activated microglia and astrocytes, which respond to pathological stimuli such as Aβ accumulation and tau pathology. Multiple studies have reported that nonsteroidal anti-inflammatory drugs (NSAIDs) may have preventive and therapeutic effects against AD [7–9]. Increasing evidence also links changes in immune cell levels in dementia and AD, identifying spatial associations between infiltrating immune cells in the AD brain and neurons, microglia, Aβ, and tau pathology. In a recent study, Van Olst et al. [10] employed cytometry by time-of-flight (CyTOF) to comprehensively map peripheral immune changes in patients with mild cognitive impairment (MCI) or dementia due to AD. Their findings revealed significant adaptive immune signatures in AD patients, demonstrating a strong correlation between peripheral immune alterations and both early and late clinical stages of the disease.
Metabolism encompasses all biochemical reactions within cells, tissues, and organisms that sustain homeostasis. Broadly, cellular metabolism is defined by complex biochemical pathways involved in biomolecule synthesis, maintenance, and degradation [11]. A well-regulated metabolic system is essential for maintaining physiological functions such as growth, reproduction, structural integrity, and environmental responses [12]. The brain is highly enriched in lipids, and lipid metabolism dysregulation has been recognized as a key contributor to AD pathogenesis [13]. As early as 1907, Alois Alzheimer observed lipid granule accumulation in microglial cells, suggesting a potential link between lipid metabolism dysfunction and AD-related pathological changes. Clinically, lipidomics and metabolomics studies consistently indicate altered levels of various lipid species in the early stages of AD. Mechanistically, extensive research has uncovered multifaceted interactions between lipid metabolism and core AD pathophysiological processes, including amyloidogenesis, bioenergetic deficits, oxidative stress, neuroinflammation, and myelin degeneration [14].
Immunometabolism is an emerging research field that investigates the metabolic processes of immune cells and their influence on immune responses, elucidating the intricate interplay between metabolism and immunity [15]. The fundamental mechanism of immunometabolism involves cellular activation and antigen stimulation, which trigger metabolic reprogramming to support downstream signaling pathways. Typically, upon the transmission of stimulatory signals into the cell, downstream signaling events are initiated, leading to changes in cytokine secretion and other functionally relevant processes. On one hand, metabolic alterations influence immune cell activity, thereby modulating immune responses [16]. Different immune cell subtypes, depending on their developmental stage and functional demands, require distinct energy sources, requiring metabolic reprogramming for proper regulation [17]. On the other hand, the immune system can regulate systemic energy metabolism through inflammatory responses. Cytokines, as immune cell-derived signaling molecules, can activate metabolic pathways, promoting energy production and macromolecule synthesis. Investigating the interplay between the immune and metabolic systems could offer novel insights into the prevention and treatment of chronic diseases.
In recent years, accumulating evidence has highlighted the crucial roles of immunity and lipid metabolism in AD onset and progression. These two processes are intricately connected and mutually influence each other, collectively driving AD pathogenesis. Therefore, further exploration of the relationship between immunity, lipid metabolism, and AD is crucial for elucidating disease mechanisms and developing novel therapeutic strategies.
Characteristics of the immune response in Alzheimer’s disease
Overview of the brain’s immune system
The immune system is a complex network composed of various organs, cells, humoral factors, and cytokines, responsible for recognizing and eliminating invading pathogens and abnormal cells to maintain overall health [18]. Its function is broadly categorized into innate immunity and adaptive immunity. Innate immunity serves as the body’s first line of defense, comprising physical barriers (e.g., skin, mucosa), immune cells (e.g., macrophages, neutrophils, natural killer cells), and immune molecules (e.g., complement proteins, cytokines). This system responds rapidly to pathogens but lacks specificity. In contrast, adaptive immunity is highly specific and has immune memory, primarily mediated by T cells and B cells. T cells can be further divided into subtypes, such as helper T cells (Th cells) and cytotoxic T cells (Tc cells), which are activated upon recognizing antigenic peptides presented by antigen-presenting cells (APCs) via major histocompatibility complex (MHC) molecules. These activated T cells exert immunoregulatory or cytotoxic effects. B cells, on the other hand, produce antigen-specific antibodies that facilitate pathogen clearance through antibody-mediated immune responses [19].
The central nervous system (CNS), which consists of the brain and spinal cord, plays a crucial role in processing and transmitting information to the peripheral nervous system while regulating bodily functions. Within the CNS, a unique neuroimmune regulatory system exists [20]. In response to disease or injury, the CNS immune system mobilizes immune cells by releasing cytokines, prompting their differentiation into specialized immune cells based on the severity and location of the injury. These immune cells facilitate the recognition and clearance of inflammatory factors while aiding neuronal repair [21]. However, persistent immune cell activation may lead to further damage to the CNS [22, 23].
Innate immunity in Alzheimer’s disease
Microglia are the primary immune cells of the CNS, originating from myeloid progenitor cells during embryonic development, and are widely distributed throughout the brain and spinal cord [24]. Under physiological conditions, microglia remain in a resting state, continuously extending and retracting their processes to monitor the surrounding environment and maintain neural homeostasis. They secrete neurotrophic factors such as brain-derived neurotrophic factor (BDNF) and nerve growth factor (NGF), which promote neuronal survival, growth, and synaptic plasticity [25], [26]. Additionally, microglia regulate neurovascular units, maintaining cerebral blood supply and molecular exchange [27].
In the early stages of AD, Aβ deposition triggers microglial activation, shifting them from a resting to an activated state [28]. This is characterized by increased cell body size, shortened processes, and enhanced phagocytic capacity, allowing microglia to clear pathogens, apoptotic cells, and neurotoxic substances [29]. However, activated microglia also release pro-inflammatory cytokines such as tumor necrosis factor-alpha (TNF-α), interleukin-1β (IL-1β), and interleukin-6 (IL-6), as well as chemokines that recruit peripheral immune cells into the CNS, thereby initiating neuroinflammatory responses [30, 31]. While neuroinflammation may initially contribute to pathogen clearance and tissue repair, chronic or excessive neuroinflammation exacerbates neuronal damage and promotes neurodegeneration. Moreover, overactivated microglia may undergo phenotypic shifts, transitioning from a neuroprotective M2 phenotype to a neurotoxic M1 phenotype, which exacerbates neuroinflammation and neurodegeneration [32, 33]. The NLRP3 inflammasome, an inflammation-sensing protein complex, is highly expressed in activated microglia and macrophages. Its activation leads to the formation of oligomeric complexes containing apoptosis-associated speck-like protein (ASC) and caspase-1, perpetuating neuroinflammation. Knockdown of NLRP3 enhances Aβ phagocytosis by microglia, reduces their pro-inflammatory response and pyroptosis, and improves spatial learning and memory in 5×FAD mice [34].
Astrocytes, another major type of glial cell in the CNS, play key roles in neuronal metabolic support and ion homeostasis [35]. In AD, Aβ deposition and tau hyperphosphorylation directly activate astrocytes [36]. Furthermore, cytokines secreted by activated microglia, such as IL-1α, TNF-α, and C1q, can transform astrocytes from a resting to an activated state [37]. Activated astrocytes release large amounts of pro-inflammatory cytokines and chemokines, such as CXCL10, CCL2, IL-6, and BAFF, which further recruit immune cells to affected regions, exacerbating inflammation [38]. Additionally, astrocytes can release excitatory neurotransmitters such as glutamate, leading to excessive neuronal excitation, calcium influx, and subsequent neuronal damage [39]. Thus, astrocyte overactivation is a key pathogenic mechanism in AD.
The complement system, a crucial component of innate immunity, is abnormally activated in the AD brain, promoting Aβ deposition and neuroinflammation. The classical and alternative complement pathways identify and tag Aβ, facilitating its phagocytosis by microglia [40]. However, complement system dysregulation may also cause neuronal damage, contributing to AD pathology [41, 42]. C1q, an initiator of the classical complement cascade, plays a critical role in synaptic toxicity induced by soluble Aβ oligomers. C1q deficiency reduces astrocyte-synapse interactions in tauopathy models and decreases microglial phagocytosis of synapses [43–45]. Additionally, in response to soluble Aβ oligomers, adult microglia phagocytose synapses in a CR3-dependent manner [46]. The C5b-9 membrane attack complex (MAC) can directly damage neuronal membranes, leading to neuronal death. Complement inhibition prevents MAC-mediated blood-brain barrier disruption in Tg-SwDI/B mice, demonstrating its potential therapeutic value [47].
Adaptive immunity in Alzheimer’s disease
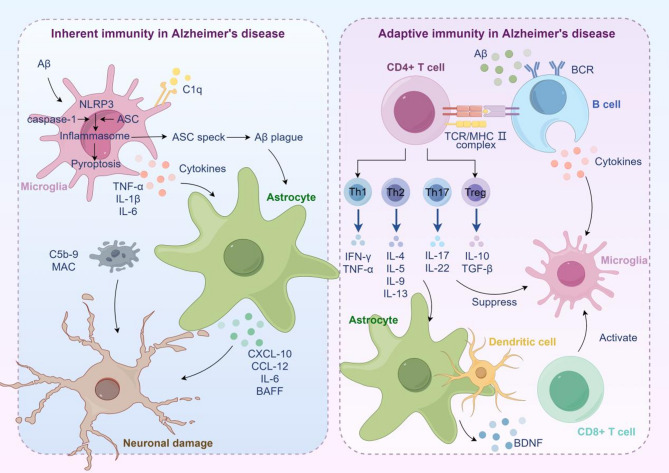
Recent evidence suggests the presence of adaptive immune responses in both the blood and cerebrospinal fluid (CSF) of AD patients [48, 49]. Peripheral immune cells—including neutrophils, T cells, B cells, natural killer (NK) cells, and monocytes—infiltrate the brain vasculature and parenchyma, modulating immune and inflammatory responses and playing critical roles in AD progression [50] (Fig. 1).
Fig. 1.
Immune response in Alzheimer’s Disease. Under the stimulation of Aβ, microglia are activated and secrete multiple inflammatory cytokines and chemokines, which trigger neuroinflammatory response. It also activates astrocytes, further exacerbating the inflammatory response. Activated T cells and B cells secrete pro-inflammatory and anti-inflammatory cytokines, regulate the state transition of microglia, as well as astrocytes and dendritic cells, and participate in the regulation of immunity and inflammation in the progression of AD. Overall, during the progression of AD, the immune response shifts from an activated and elevated state in the early stage, attempting to counteract pathological products such as Aβ, to a complex imbalanced state in the later stage. Excessive inflammatory reactions persist and mutually promote neurodegenerative changes. Meanwhile, although regulatory immune mechanisms are involved, they are difficult to restore immune balance
CD4 + T cells differentiate into subtypes such as Th1, Th2, Th17, and Treg cells. Under normal conditions, Th1 (producing IFN-γ) and Th2 (producing IL-4) cells reside in the meninges, where IFN-γ regulates meningeal dendritic cells and astrocytic BDNF expression, influencing neural circuits underlying social behavior. However, T-cell imbalance can lead to dysregulated immune-inflammatory responses. In AD, abnormal T-cell crosstalk with glial cells results in excessive pro-inflammatory cytokine secretion, promoting neuroinflammation and neurodegeneration [51, 52]. In tauopathy mouse models and AD brains, the number of T cells, especially cytotoxic T cells is significantly increased in tau pathology regions, correlating with neuronal loss [53]。.
The exact role of CD8 + T cells in AD remains elusive—both their depletion and accumulation can accelerate disease progression [54]. Studies have observed increased CD8 + T cell infiltration in the hippocampus of human AD and APP-PS1 mice [55]. Some studies have reported that brain CD8 + T cells limit AD pathology, including β -amyloid deposition and cognitive decline [56]. However, Jorfi et al. [57] observed the infiltration of CD8 + T cells into AD cultures resulted in increased microglial activation, neuroinflammation, and neurodegeneration by constructing a three-dimensional human neuroimmune axis model of stem cell-derived neurons, astrocytes, and microglia, and peripheral immune cells. Conversely, regulatory T cells (Tregs), particularly Aβ antigen-specific Tregs (Aβ + Tregs), suppress pro-inflammatory microglial activity, alleviating cognitive impairment, Aβ accumulation, tau hyperphosphorylation, and neuroinflammation in AD models [58], [59].
B cells, key components of adaptive immunity, also play complex roles in AD. They produce Aβ-targeting antibodies, present antigens to T cells, and regulate immune responses [60]. First, B cells can produce antibodies, and the induction of β amyloid plaque formation using 44B cells was used to construct AD cell models [61]. The increase in B cells is associated with increased brain amyloid deposition and with hyperactivation of induced pluripotent stem cell-derived microglia, leading to a loss of β -amyloid clearance function [62]. Loss of B cells also reduced Aβ plaque burden and disease-associated microglia, reversing behavioral and memory deficits and restoring TGF β + microglia, respectively, delaying AD progression in mice [63]. However, Feng et al. [64] reported that B cell-derived IL-35 was able to inhibit Aβ production in the frontal cortex in 5×FAD mice. Xiong et al. [65] detected a significant reduction in B cells in the blood of AD patients and identified 18 genes that were specifically up-or downregulated in B cells strongly associated with AD severity. Therefore, the exact contributions of B cells in the pathogenesis of AD require further investigation.
Immune-related animal models of Alzheimer’s disease
In vivo models of AD aim to replicate the neuropathological features of the human disease in order to create platforms for studying how the disease develops and progresses, as well as for testing potential treatments that could modify its course. Using immune-related animal models, the investigators explore the role of inflammation in AD progression and aim to identify immune-modulating therapeutic targets. The following animal models have been developed over the years (Table 1).
Table 1.
Immune-related animal models of Alzheimer’s disease
| Model Type | Description | Intervention | Reference |
|---|---|---|---|
| Immunoinflammatory Animal Models | Focus on the roles of immunocytes and cytokines in AD | IL−1β overexpression in APPswe/PS−1dE9 mice | [66] |
| IL−3 deficiency in 5xFAD mice | [67] | ||
| IL−6 deficiency in 5xFAD mice | [31] | ||
| IL−12 p40 deficiency in APP/PS1 mice | [68] | ||
| IL−12 p40 deficiency in APP23 mice | [69] | ||
| NLRP3-targeted 5×FAD mice | [34] | ||
| CX3CR1 deficiency in 5xFAD mice | [70] | ||
| CD33 deficiency in 5xFAD mice | [71] | ||
| TREM2 deficiency in 5xFAD mice | [71] | ||
| TREM2 overexpression on microglia in the brain of P301S mice | [72] | ||
| Animal Models with Induced Inflammation | Animals with induced inflammation to mimic neuroinflammation | lipopolysaccharide (LPS)-induced neuroinflammation in WT mice | [73, 74] |
| LPS-induced neuroinflammation in 5xFAD mice | [75] | ||
| LPS-induced neuroinflammation in APP/PS1 mice | [76, 77] | ||
| Animal Models of Immune Modulation Therapy | Immune-modulatory therapies to reduce neuroinflammation in AD models | IL−2 treatment for APP/PS1ΔE9 mice | [78] |
| Low-dose IL−2 treatment for APP/PS1 mice | [79] | ||
| anti-IL−17 treatment for 3xTg mice | [80] | ||
| IL−17Ab treatment for CD−1 mice with Aβ injection | [81] | ||
| IL−33 treatment for APP/PS1 mice | [82, 83] | ||
| IL−35 injected in 5xFAD mice | [64] | ||
| NSAIDs | [9] | ||
| Minocycline (TNF-α Inhibitor) treatment for APP-tg mice | [84] | ||
| Minocycline (TNF-α Inhibitor) treatment for APP/PS1 mice | [85] | ||
| Imipramine (TNF-α Inhibitor) treatment for APP-tg mice | [86] | ||
| Thalidomide (TNF-α Inhibitor) treatment for 3xTg mice | [87, 88] | ||
| Etanercept (TNF-α Inhibitor) treatment for 3xTg mice | [89] | ||
| TfRMAb-TNFR (TNF-α Inhibitor) treatment for APP/PS1 mice | [90] | ||
| Complementary suppressor therapy | [47] | ||
| Aβ-specific Tregs treatment in 3xTg mice | [59] | ||
| TREM2 wild-type microglia transplantation therapy | [91] |
Characteristics of lipid metabolism in Alzheimer’s disease
Overview of brain lipid metabolism
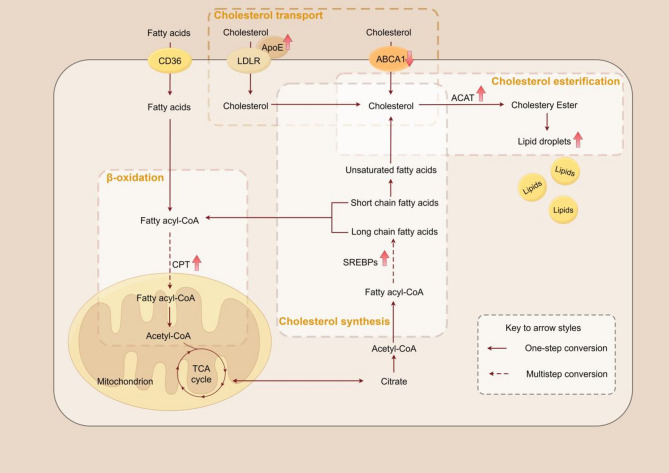
Lipids are a diverse group of organic compounds widely found in living organisms, including cholesterol, fatty acids, phospholipids, and sphingolipids, among others. They play critical roles in cellular structure, energy storage, and signal transduction. The brain, being one of the most lipid-rich organs, relies on the transport and distribution of lipids to maintain homeostasis and neuronal function [92]. Cholesterol is essential for maintaining the fluidity and stability of neuronal cell membranes [93], and it also mediates neuroinflammation [94]. Fatty acids are key components of phospholipids, and the degradation of fatty acids in astrocyte mitochondria leads to neuroinflammation and neurodegeneration [95]. Phospholipids and sphingolipids are major components of biological membranes, crucial for maintaining the structural and functional integrity of cell membranes. These lipids not only provide a physical barrier but also give rise to metabolites involved in various cellular signaling processes [96]. In AD, lipid metabolism processes involve cholesterol synthesis, esterification, transport, and fatty acid oxidation, all of which are tightly regulated by a series of enzymes and transport proteins [97] (Fig. 2).
Fig. 2.
Lipid metabolism in Alzheimer’s Disease. Lipid metabolism in AD involves cholesterol synthesis, esterification and transport, and oxidation of fatty acids. The upward arrows indicate an increase in enzyme expression/activity and associated metabolic pathways. By targeting these enzymes and transporters, such as ACAT, ABCA1, CPT, and SREBPs, which regulate lipid metabolism and affect lipid synthesis, transport, and storage, it is possible to reduce AD pathology. This could potentially slow down or mitigate the progression of Alzheimer’s disease by modulating the abnormal lipid metabolism that is associated with the disease
Cholesterol metabolism in Alzheimer’s disease
Numerous studies have shown that elevated cholesterol levels are associated with an increased risk of dementia [98, 99]. A recent report by Livingston et al. identified elevated low-density lipoprotein cholesterol (LDL-C) levels as a risk factor for mid-life dementia, based on new evidence from large cohort studies involving over a million participants and a Mendelian meta-analysis of 27 studies [100, 101]. Paiva et al. [102] found that the downregulation of cholesterol biosynthesis genes in AD impairs memory function in hippocampal neurons under pathological conditions. Sterol regulatory element-binding proteins (SREBPs), including SREBP1 and SREBP2, are transcription factors involved in cholesterol and fatty acid synthesis. Spell et al. [103] found that polymorphisms in SREBP-1a affect the risk of AD in ApoE4 allele carriers. Overexpression of SREBP-2 in APP/PS1 mice results in Aβ deposition and tau aggregation typical of AD [104], and inhibiting SREBP-2 improves neurodegenerative pathology by significantly reducing Aβ production, abundance, and aggregation, as well as restoring synaptic function and alleviating cognitive and memory deficits [105]. Additionally, SREBP-2 is influenced by tau alterations in AD, causing the translocation of nuclear mSREBP-2, leading to disrupted cholesterol homeostasis in AD [106].
Cholesterol is stored in cells as cholesteryl esters (CE). Studies have shown that CE is an early upstream regulator of tau accumulation in iPSC-derived neurons from AD patients [107]. West et al. [108] similarly observed that inhibiting cholesterol esterification significantly increases Aβ-induced synaptic damage, and that using cholesterol ester hydrolase inhibitors can protect neurons. The biosynthesis of CE is catalyzed by Acyl-CoA acyltransferase (ACAT), which esterifies cholesterol to help cells reduce free cholesterol content and prevent excessive cholesterol accumulation in the membrane, maintaining cellular cholesterol balance. Studies have shown that ACAT inhibitors, such as CP-113,818, significantly reduce amyloid pathology in AD mouse models [109]. Another team used high concentrations of the ACAT1 inhibitor F12511 encapsulated in stealth liposomes to significantly improve amyloid pathology in aged 3xTg AD mice, reducing hyperphosphorylated tau and non-phosphorylated tau, as well as alleviating neuroinflammation [110]. Furthermore, AAV-mediated ACAT1 knockdown has potential therapeutic effects in AD, as delivery of AAV-ACAT1 to the brains of 10-month-old AD mice reduced brain Aβ and human amyloid precursor protein (hAPP) levels at 12 months of age [111].
The cholesterol synthesis capacity differs among various brain cell types. Cholesterol levels in the brain are regulated through the crosstalk between the plasma membrane and the endoplasmic reticulum, not only to meet the distinct cholesterol demands of different cells but also to regulate cellular functions [112, 113]. ApoE4, a key protein for cholesterol transport, is primarily synthesized and secreted by astrocytes in the brain. There are three ApoE isoforms (ApoE2, ApoE3, and ApoE4), with ApoE4 being the strongest genetic risk factor for sporadic Alzheimer’s disease (SAD). Recent studies show that by age 55, nearly all ApoE4 homozygotes exhibit AD pathology and higher levels of disease-related biomarkers. By age 65, over 95% of ApoE4 homozygotes show AD biological features in the brain or abnormal Aβ levels in cerebrospinal fluid, with 75% testing positive for amyloid scans [114]. Early detection of Alzheimer’s disease (AD) relies primarily on biomarkers in cerebrospinal fluid (CSF), such as Aβ42, Aβ42/Aβ40 ratio, p-Tau181, and total Tau (t-Tau). But the new study found that the ApoE4 gene affects the levels of these biomarkers, causing the same test to behave differently in different populations [115]. This means that if the ApoE4 status is not considered, it may result in misdiagnosis or missed diagnosis.ApoE4 variants not only increase the risk of AD but also influence the disease’s progression and severity [116]. ApoE4-containing microglia contribute to tau phosphorylation and neurotoxicity in an ApoE-dependent manner [117]. Shi et al. [118] showed that overexpression of low-density lipoprotein receptors reduces tau-related neurodegeneration associated with ApoE mechanisms.
Cholesterol reverse transport refers to the process of transporting cholesterol from peripheral tissues back to the liver through the bloodstream, where it is metabolized and excreted [119]. ATP-binding cassette transporter A1 (ABCA1) is a key protein in this reverse transport process and plays an essential role in the transport of cholesterol and phospholipids. It transfers excess cellular cholesterol and phospholipids to apolipoproteins, forming high-density lipoprotein (HDL)-like particles that maintain cellular cholesterol balance. In the brain, ABCA1 lipidates ApoE [120], facilitating Aβ clearance [121, 122], regulating peripheral vasculature and blood-brain barrier integrity, as well as anti-inflammatory signaling [123], influencing myelination, synaptogenesis, and neurotransmission [124], playing a crucial role in the onset and progression of AD. Holstege et al. [125] found a significant association between ABCA1 loss-of-function mutations and increased AD risk, based on exome sequencing data from 16,036 AD cases and 16,522 controls. Moulton et al. [126] found that ABCA1 peptide agonists promoted the formation of protective glial lipid droplets in ApoE4 humanized fly models.
Fatty acid metabolism in Alzheimer’s disease
The brain heavily relies on astrocytes for oxidative phosphorylation (OxPhos) to degrade fatty acids (FAs) and maintain lipid homeostasis. Both the brain and blood in AD patients show alterations in fatty acid profiles, with notable differences in the lipidomic fatty acid profiles of CSF and plasma [127]. Specific changes in erythrocyte FA composition even occur prior to the onset of cognitive impairment [128]. Chen et al. [129] utilized metabolomics to identify polyunsaturated fatty acid (PUFA) metabolites in 3xTg mice and wild-type mice, finding that gut microbiota regulates AD pathology and cognitive dysfunction through PUFA-associated neuroinflammation.
Carnitine palmitoyltransferase (CPT), a multi-protein complex, is a rate-limiting enzyme in fatty acid oxidation. CPT1c, a neuronal subtype, is predominantly located in brain regions such as the hypothalamus, amygdala, and hippocampus, and plays a crucial role in neurodegenerative diseases, including AD [130]. Sarnowski et al. [131] identified a distinct association between CPT1A loci and insulin resistance in AD through multi-tissue epigenetic analyses. Upregulation of CPT1a expression in APP/PS1 mice enhances fatty acid oxidation in astrocytes, improving memory deficits [132].
Lipid metabolism-related animal models of Alzheimer’s disease
Animal models have been instrumental in studying the link between lipid metabolism and AD. Through animal models related to lipid metabolism, researchers are able to better understand how lipid abnormalities affect nerve cell function, inflammatory response and proteostasis. These models provide valuable experimental evidence for developing therapeutic strategies against abnormal lipid metabolism and provide new research directions for early diagnosis and prevention of AD. The following in vivo models have been developed over the years (Table 2).
Table 2.
Lipid metabolism-related animal models of Alzheimer’s disease
| Model Type | Description | Intervention | Reference |
|---|---|---|---|
| High-fat diet-induced AD models | AD mice were given a high-fat diet to mimic the effects of disturbed lipid metabolism | High-fat diet for 5×FAD mouse model | [133] |
| High-fat diet for Tg601 mice | [134] | ||
| High-fat diet for 3xTg mice | [135] | ||
| Animal models of the altered lipid transport process | Targeting lipid transport | Deletion of the cholesterol sensor SCAP in C57BL/6 mice | [136] |
| Transgenic knockin mouse expressing HaloTag-tagged ApoE | [137] | ||
| Deletion of microglial ApoE4 in APP/PS1 mice | [138] | ||
| Deletion of astrocytes ApoE in APP/PS1−21 mice | [139] | ||
| ApoE4 knockin APP/PS1 mice | [140] | ||
| Thy1-ApoE4/C/EBPβ double transgenic mouse model | [141] | ||
| Neuronal specific Thy1-ApoE4/C/EBPβ double transgenic mice | [142] | ||
| LDLR-targeted AD models | [118, 143] | ||
| Pericyte LRP1-deficient in APPSwe/0mice | [144] | ||
| LXR agonist GW3965 treatment for P301S/ApoE4 mice | [145] | ||
| LXR agonist CE9A215 treatment for 3xTg-AD mice | [146] | ||
| LXR agonists T1317 and GW3965 treatment for rats and C57Bl6/SJL respectively | [147] | ||
| Animal models of altered lipid metabolism processes | Targeting key enzymes for lipid metabolism | SREBP2 overexpression in APP/PS1 mice | [104, 148] |
| Lovastatin (HMGCR inhibitor) treatment for rats | [149] | ||
| CP−113,818 (ACAT inhibitor) for hAPP mice | [109] | ||
| Avasimibe (ACAT inhibitor) treatment for Tg mice | [150] | ||
| ACAT-knock out in Tg mice | [150] | ||
| ACAT1 knockdown gene therapy in 3xTg mice | [111] | ||
| Peptide agonist of ABCA1 treatment for humanized ApoE4 fly model | [126] | ||
| NLAI (ABCA1-inducers) treatment for E3/4FAD mice | [120] | ||
| Atorvastatin (Cholesterol inhibitor) treatment for Wistar rats injected Aβ1–42 | [151] | ||
| CPT1a-targeted APP/PS1 mice | [132] |
Immune-lipid metabolic interactions at the tissue level
Immune-Lipid metabolic interactions in the choroid plexus
The choroid plexus (CP), located between the blood-brain barrier and CSF, consists of tightly connected epithelial cells, a vascular-rich stroma, and mesenchymal, glial, neuronal, and immune cells. It is the primary site of CSF production and also produces a variety of neurotrophic factors (NTFs) that circulate throughout different brain regions [152]. The CP serves as a selective and strictly regulated physiological gateway for immune cells entering the CNS, playing a key role in regulating solute exchange between the blood and CSF, mediating CNS immune surveillance and repair, and removing toxic waste, such as Aβ, to protect the brain.
CD4 + T helper (Th) cells in the CP stroma are essential for maintaining the expression of BDNF and leukocyte trafficking determinant factors. The CP’s immune environment controls its function, which, in turn, impacts CNS function and repair [153, 154]. The high secretory nature and strong molecular transport activity at the CP require high metabolic activity, which is linked to inflammation regulation. Lipid metabolic components may play important roles in modulating local immune responses. For instance, liver X receptors (LXRs) are critical for CP function and anatomical integrity, with LXR signaling regulating CSF fluid production, Aβ clearance, and inflammation modulation [155]. Systemic treatment with liver LXR agonists can enhance ApoE- and cholesterol-mediated Aβ transport to the CSF [147]. Endogenous LXR ligands, such as 24 S-hydroxycholesterol, induce ATP-binding cassette transporters ABCA1 and ABCG1 in an ApoE subtype-dependent manner, promoting cholesterol release from CP epithelial cells into the CSF [156]. Additionally, CP epithelial cells express the brain-specific cholesterol 24-hydroxylase CYP46A1, with its expression levels reduced in amyloid degeneration and aging mouse and human brains. Overexpression of CYP46A1 in amyloid-degenerative transgenic mice is associated with better cognitive performance and reduced brain inflammation [157]. Therefore, CP metabolism plays a crucial role in regulating inflammation, and lipid metabolism and immune homeostasis within the CP represent promising avenues for enhancing brain protection [158].
Immune-lipid metabolic interactions in the blood-brain barrier
Vascular dysfunction is a major risk factor for neurological diseases, with early blood-brain barrier (BBB) disruption and/or dysfunction in AD often observed before the onset of dementia, neurodegeneration, or brain atrophy [159, 160]. The BBB consists of a plasma-brain cell barrier formed by the walls of cerebral capillaries and glial cells, as well as a plasma-CSF barrier formed by the choroid plexus, both of which block harmful substances from entering brain tissue. When the BBB is compromised, fibrinogen exudes into the CNS, where it is converted to fibrin after coagulation activation. Fibrin further activates glial cells and innate immune cells via CD11b/CD18 integrins, triggering pathological inflammation and oxidative damage, leading to neurodegenerative lesions.
The lipid nutrient metabolism and immune regulation of the BBB are highly interconnected processes. Excessive saturated fatty acids and monosaccharides in the diet may contribute to AD-related pathology by accelerating inflammation, metabolic inflammation, and systemic inflammation, driving BBB damage and neuroinflammation, which ultimately leads to synaptic dysfunction and memory and cognitive impairment [161]. High-density lipoprotein (HDL) reduces Aβ accumulation in the vasculature and dampens Aβ-induced endothelial inflammation, thus lowering AD risk [162–164]. Furthermore, BBB disruption is mediated by ApoE4. In ApoE4 mice, disrupted signaling in endothelial and pericyte cells earlier reflects progressive BBB failure, preceding postsynaptic damage and the development of behavioral defects 2–5 months later [165]. In terms of treatment, overcoming the BBB is a major challenge for AD therapies, as drugs must pass through the BBB to reach their target sites. Liposomes, with their unique phospholipid bilayer structure (similar to physiological membranes), are more compatible with the lipid layers of the BBB and facilitate drug entry into the brain, showing significant potential in AD therapy [166, 167].
Immune-lipid metabolic interactions in the glymphatic system
The glymphatic system, a unique structure filled with CSF from small vascular perivascular spaces, facilitates the clearance of CNS-derived antigens, metabolites, and molecules (such as Aβ and tau), while regulating immune responses and fluid balance in the brain [168, 169]. In addition to waste removal, the lymphatic system functions as a distribution system for electrolytes, macromolecules, and other large compounds, providing an important route for the distribution of these molecules and improving the effectiveness of intrathecal drug delivery [170, 171].
The glial lymphatic system is thought to be mediated by aquaporin-4 (AQP4) expressed on astrocytes, which facilitates the exchange of CSF and interstitial fluid (ISF) within the perivascular spaces. This system primarily consists of the perivascular space surrounding arteries, AQP4 on astrocytic endfeet, and the perivenous space [172]. Physiological factors, including arterial pulsations, respiration, and CSF pressure gradients, drive the CSF flow from the subarachnoid space into deep brain structures via the perivascular space, and subsequently into the brain parenchymal spaces. The CSF metabolic products and toxic proteins are eventually drained through meningeal lymphatics to the cervical lymph nodes and peripheral lymphatic systems, maintaining CSF-ISF balance and clearing brain metabolites [173, 174].
Recent assessments of lymphatic function in aged versus young mice have shown that lymphatic function declines dramatically (by 80–90%) in older mice compared to younger controls [175]. In healthy young humans, CSF enters the brain parenchyma through the perivascular route, clearing solutes from the interstitial spaces and draining along the venous routes. In AD, compared to age-matched controls, patients frequently show abnormal enlargement of perivascular spaces, suggesting that vascular amyloidosis may reduce the influx of lymphatic CSF, with CSF stasis accelerating Aβ accumulation [176, 177].
Interestingly, reduced CSF-ISF exchange does not correlate with Aβ clearance, potentially due to the direct clearance of some Aβ via low-density lipoprotein receptor-related protein 1 (LRP1)-mediated trans-endothelial transport into the bloodstream. LRP1, a member of the LDL receptor family, plays a significant role in the endocytosis of tau proteins, and LRP1 gene silencing markedly inhibits tau internalization [143]. Additionally, LRP1 promotes mitochondrial transfer from astrocytes to neurons, reducing neuronal sensitivity to acute stress [178]. Neuronal LRP1 deletion leads to neuroinflammation [179]. Thus, LRP1 may serve as a bridge between lipid metabolism and immune regulation in AD progression. Moreover, dietary supplementation with polyunsaturated fatty acids (PUFAs) can improve glial lymphatic function, reduce neuroinflammation and cerebrovascular dysfunction, and ultimately improve cognitive abilities [180, 181].
Immune-metabolic interactions at the cellular level
Membrane-associated signal transduction
Extracellular vesicles (EVs) are membrane-bound vesicles released into the extracellular space, carrying proteins, miRNAs, and metabolites into the circulatory system. Their role in intercellular communication has garnered increasing attention and is believed to be involved in the pathogenesis of neurodegenerative diseases, including AD [182–184]. In AD, brain-derived EVs not only mediate synaptic dysfunction but also propagate tau protein in hippocampal GABAergic interneurons, ultimately leading to neuronal dysfunction [185]. When these EVs are released from the brain into the periphery, they provide AD-specific immune-metabolic biomarkers [186–188]. For example, the inflammatory factor ITGB1 is significantly elevated in astrocyte-specific EVs enriched with brain-derived AD EVs, and this elevation correlates with brain Aβ and tau burden in independent cohorts [189]. Compared to normal controls, brain-derived EVs in AD significantly alter glycerophospholipid and sphingolipid levels, particularly increasing alkenylglycerophosphoethanolamine and decreasing polyunsaturated acyl-lipid content. Changes were also observed in the acyl-chain content of amide-linked sphingolipids and ceramides. The most notable change was a two-fold reduction in lipids containing the anti-inflammatory/pro-resolving eicosapentaenoic acid [190]. Recently, a research team has developed a method for treating AD using mesenchymal stem cell-derived extracellular vesicles (MSC-EVs-SHP2) with high expression of tyrosine phosphatase-2 (SHP2). In AD mouse models, MSC-EVs-SHP2 were shown to significantly induce mitochondrial engulfment in neuronal cells, thereby alleviating mitochondrial damage-mediated apoptosis and NLRP3 inflammasome-induced neuroinflammation, ultimately preventing synaptic loss and cognitive decline [191]. Therefore, EV-mediated immune-metabolic responses provide a promising new platform for AD treatment.
ApoE, the main cholesterol transporter in the brain, mediates the transfer of cholesterol and other lipids between neurons and glial cells. ApoE achieves lipid transport by interacting with members of the LDL receptor family on the surfaces of neurons and glial cells. After binding to these receptors, ApoE releases lipids into the cells, providing the material basis for their physiological functions. Studies have shown that pro-inflammatory stimuli can induce metabolic reprogramming in microglial cells. In induced pluripotent stem cell-derived microglia-like cells (iMGLs), ApoE4 iMGLs showed a significant increase in lipid droplet content and a reduction in genes related to lipid degradation and membrane fatty acid transporters, such as CD36 [192, 193]. ApoE4-induced lipid accumulation increases the inward rectifier potassium (Kir) current in the neuronal cell membrane, hyperpolarizing the resting membrane potential and reducing neuronal excitability [194]. Transcriptional profiling revealed that ApoE4 iMGLs are significantly enriched in HIF-1, JAK-STAT signaling, and cytokine-cytokine receptor interactions, suggesting a strong pro-inflammatory response [195].
Triggering receptor expressed on myeloid cells 2 (TREM2) is a transmembrane immune receptor predominantly expressed on microglial cells in the brain and macrophages in the periphery. Loss-of-function (LOF) variants of TREM2 increase the risk of AD [196, 197]. In mice and humans carrying TREM2 LOF variants, higher levels of apoptotic synapses have been observed [198]. In AD, TREM2 plays a crucial regulatory role in both central and peripheral immune and metabolic responses, making it a potential target for AD immunometabolic therapy [199–202]. In the CNS, TREM2 serves as a surface receptor required for microglial response to neurodegeneration. It supports microglial energy and biosynthesis metabolism, making their activation possible during AD [203, 204], while also reducing Aβ seeding and inhibiting disease-associated microglial activation [205]. However, Jain et al. [206] reported that prolonged use of activated TREM2 antibodies might exacerbate Aβ-induced tau pathology, highlighting the importance of considering the duration and dosage in clinical TREM2 interventions. Additionally, Aβ-induced microglial depolarization, K + inward current induction, cytokine expression and secretion, migration, proliferation, apoptosis, and morphological changes are all dependent on TREM2 [207–209]. TREM2 also protects complement-mediated synapse loss through its interaction with complement C1q during neurodegeneration [210]. TREM2 mediates lipid patterns associated with neurodegeneration [211] and promotes myelin engulfment, regulating cholesterol metabolism in microglia under chronic phagocytic stress. Its LOF leads to pathogenic lipid accumulation in microglia [212, 213]. Replacing TREM2-deficient microglia with TREM2 wild-type cells derived from hematopoietic cells has been shown to correct microglial dysfunction in the 5×FAD mouse model, offering a potential new treatment approach for AD [91]. In the periphery, TREM2 affects lipid metabolism by regulating the onset and progression of obesity and its complications, including hypercholesterolemia, atherosclerosis, and non-alcoholic fatty liver disease. All of these altered lipid metabolic processes can influence AD progression through their effects on inflammation, insulin resistance, and AD pathology [214] (Table 3; Fig. 3).
Table 3.
AD risk genes associated with both the lipid metabolism and the immune system
| Gene symbol | Lipid metabolic role | Immune function | AD-related pathology |
|---|---|---|---|
| ApoE4 | Major apolipoprotein in the brain; transports cholesterol and phospholipids [192] | Modulates inflammation and microglial activation [137, 221] | Strongest genetic risk factor for late-onset AD; affects Aβ clearance and neuroinflammation [114] |
| TREM2 | Senses lipid debris; regulates lipid uptake in microglia [211] | Controls microglial phagocytosis and anti-inflammatory state [91] | TREM2 variants (e.g. R47H) impair Aβ clearance and increase AD risk [197] |
| ABCA1 | Promotes cholesterol efflux and ApoE lipidation [120] | Indirectly regulates microglial response via ApoE lipidation [156] | Impaired ABCA1 function linked to reduced ApoE function and increased Aβ deposition [121] |
| NLRP3 | Activated by cholesterol crystals and fatty acids [191] | Key component of inflammasome activation [34] | Drives chronic neuroinflammation in AD; inflammasome activation worsens Aβ pathology [34] |
| CPT1A | Controls fatty acid β-oxidation, mitochondrial lipid metabolism [130] | Influences immune cell energy balance [132] | Altered energy metabolism may contribute to astrocytes dysfunction in AD [132] |
| LRP1 | Mediates ApoE and Aβ transport; involved in lipoprotein clearance [144] | Regulates cellular signaling and immune neuroinflammation in the brain [179] | Mediates tau internalization [143] |
| TLR4 | Can be activated by oxidized lipids and fatty acid derivatives [225] | Recognizes danger signals, triggers innate immune response [226] | TLR4 activation leads to neuroinflammation and is linked to cognitive impairment [77] |
| PPARs | Regulates mitochondrial biogenesis and fatty acid oxidation [234] | Suppresses inflammation, promotes microglial phagocytosis [133] | PPAR-α agonists reduce amyloid pathology and reverse memory deficits [233] |
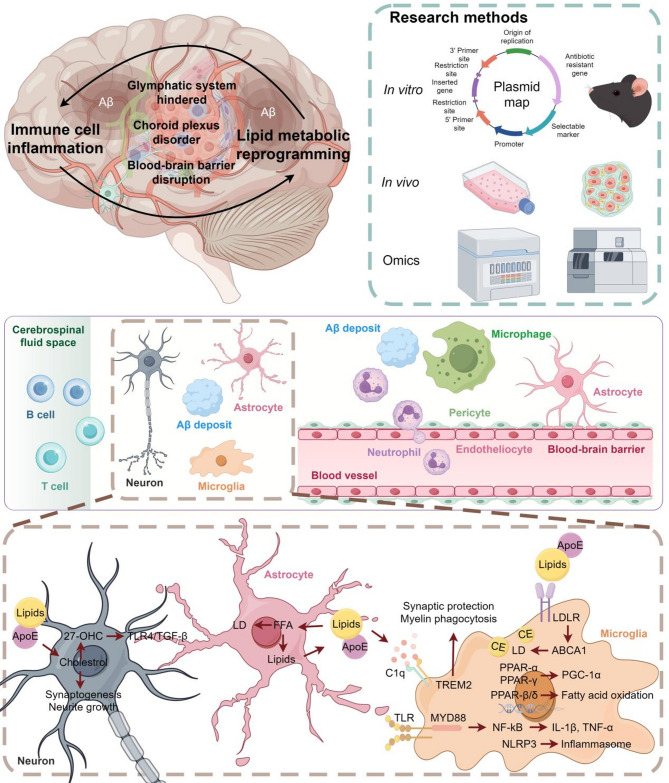
Fig. 3.
Immunometabolism in Alzheimer’s disease. The diagram illustrates the interaction between the immune response and lipid metabolism in AD, covering complex mechanisms from the tissue level to the cellular level. The figure presents the immune system activation, inflammatory response, and how lipid metabolism affects the healthy state of nerve cells, revealing the pathological process and potential therapeutic targets in AD. Current methodologies used to study the interaction between immunity and lipid metabolism primarily include in vivo and in vitro experiments, along with bioinformatics approaches such as omics analyses. In vivo studies commonly employ transgenic or gene knockout mouse models, LPS-induced neuroinflammation models, and high-fat diet-induced models. In vitro experiments typically utilize central nervous system immune cells to investigate the crosstalk between lipid metabolism and immune responses. Omics approaches such as RNA sequencing, lipidomics, and single-cell sequencing, facilitate the identification of key regulatory networks involving inflammatory factors and lipid metabolism-related genes. Future research on the interaction between immunity and lipid metabolism in AD should focus on identifying critical molecules and signaling pathways, ultimately facilitating the construction of a more comprehensive immune–lipid metabolism interaction network in AD
Intracellular signaling pathways
ApoE4 not only facilitates lipid transport at the cell membrane but also plays a differential role in amyloid and tau pathology, lipid metabolic changes, glial cell reactivity, neurodegeneration, demyelination, and BBB dysfunction in a time-space interactive manner. ApoE4 expressed in various types of CNS cells (including astrocytes, neurons, microglia, oligodendrocytes, and endothelial cells) has distinct roles in AD pathogenesis [215]. Lee et al. [216] employed RNA-seq, metabolomics, spatial transcriptomics, and mass spectrometry imaging to report that ApoE4 exacerbates amyloid plaque-induced microglial activation and lipid metabolic changes, emphasizing the central role of ApoE4 in regulating microglial immune metabolism. In AD, ApoE4 is linked to lipid droplet damage in microglia [217], and lipid-laden microglia carrying APOE4 increase tau phosphorylation and neurotoxicity [218]. ApoE4 microglia derived from AD patient-induced pluripotent stem cells and ApoE4-associated tauopathy mouse models exhibit significantly increased cholesterol biosynthesis and accumulation, which correlates with persistent microglial activation and elevated antigen presentation via the major histocompatibility complex II, followed by T-cell infiltration [219]. ApoE4 expression downregulates complement and lysosomal pathways and promotes stress-related responses [220]. Moreover, APOE4 microglia show altered morphology, slower movement toward Aβ, and reduced surveillance and phagocytosis of Aβ [221, 222]. In astrocytes, APOE4 induces lysosomal cholesterol chelation, reducing free cytoplasmic cholesterol, thereby leading to lipid dysregulation by increasing cholesterol biosynthesis and reducing efflux [192]. Selective removal of ApoE4 from astrocytes confers strong protection against tau-mediated neurodegeneration and reduces microglial synaptic phagocytosis [223]. In oligodendrocytes, studies by Manolis Kellis and Li-Huei Tsai have demonstrated that ApoE4 alters cholesterol biosynthesis, transport, and localization in both human and mouse oligodendrocytes, correlating with reduced myelination and endoplasmic reticulum stress. Pharmacological enhancement of cholesterol transport can increase myelination in aged ApoE4 mice and improve their learning and memory abilities [224].
Additionally, lipid peroxidation products can serve as endogenous danger signals, binding to toll-like receptors (TLRs) on microglial and peripheral immune cells, activating the TLR4/NF-κB pathway, and inducing early-stage pro-inflammatory M1 microglial-mediated Aβ phagocytosis defects, promoting the release of pro-inflammatory factors that damage neurons and accelerate AD progression [225]. 25-hydroxycholesterol (25-HC) promotes the production of brain cytokine IL-1β and leukocyte infiltration in LPS-induced neuroinflammation models [73]. 27-hydroxycholesterol (27-OHC) causes inflammatory neuronal damage by activating the TGF-β/NF-κB signaling pathway and induces inflammatory damage in astrocytes via the TLR4/TGF-β signaling pathway, leading to the release of inflammatory cytokines [226]. Therefore, cholesterol depletion can also serve as a biomarker for chronic neuroinflammation.
Regulation at the nuclear transcription level
The peroxisome proliferator-activated receptor (PPAR) family is a key nuclear receptor involved in lipid metabolism, consisting of three subtypes: PPAR-α, PPAR-β/δ, and PPAR-γ, all of which have a negative feedback relationship with inflammation. PPARs regulate cholesterol transport to the mitochondria and lipid metabolism in neurons. Recently, due to their additional anti-inflammatory and neuroprotective effects, they have garnered attention as potential therapeutic targets for neurodegenerative diseases [227, 228]. PPAR-α and PPAR-β/δ primarily stimulate oxidative lipid metabolism, whereas PPAR-γ is involved in lipid cell assimilation through anabolic pathways [229]. In AD brain tissue, gene expression of PPAR-α and PPAR-γ coactivator-1α (PGC-1α) is significantly reduced [230]. PPAR-γ agonists enhance Aβ phagocytosis and reduce the inflammatory cytokine IL-1β [231]. Targeting PPAR-γ can suppress the pro-inflammatory M1 phenotype in microglia, promote microglial phagocytosis, inhibit neuroinflammation, and improve cognitive deficits caused by a high-fat diet in the 5×FAD mouse model [133]. In APP-PSEN1 ΔE9 mice, PPAR-α agonists reduce amyloid pathology and reverse memory deficits, anxiety symptoms, and cognitive decline [232]. The exposure to inflammatory factor IL-15 promotes PPAR-β/δ pathway activation, suppresses aerobic glycolysis, and enhances oxidative metabolism and fatty acid oxidation, thus counteracting metabolic stress-induced CD8 + T-cell apoptosis [233]. Furthermore, PPARs can regulate lipid deposition in vascular wall cells, reducing the risk of atherosclerosis [234], which is significant for AD prevention, as cerebrovascular pathology is closely linked to AD development.
Summary and prospect
Immune cells such as microglia can recognize and clear abnormal substances such as Aβ in the early stage of AD, while excessive immune responses can cause chronic neuroinflammation and accelerate neuronal damage and AD disease process. Lipid metabolism is able to affect the structure and function of the cell membrane, provide energy to the brain, and participate in cell signaling. Abnormalities in these processes are closely related to the disease progression of AD. The intersection between lipid metabolism and immune response, such as ApoE4, TREM2, and PPAR pathway, further amplified the disease progression of AD. However, many key questions remain unresolved, such as which other lipid metabolic enzymes, receptors or lipid metabolite supplements can regulate immune cell function and can be used to prevent, alleviate, or treat AD; whether immune cells can be divided into different metabolic subgroups according to their different lipid metabolic states and have specific functions of anti-neuroinflammation; how spatial and temporal changes of immune factors and metabolic factors in the progression of AD, how they form a positive feedback pathway in the progress of AD, and how to target lipid-immune interactions to intervene in the process of AD still need further study. In order to answer these questions, in-depth mechanistic research can be conducted through gene-editing and genetic breeding, which will help to more completely reveal the complex role network of lipid metabolism and immune response in AD, and provide new ideas for revealing the pathogenesis of AD.
Acknowledgements
Figures were created using FigDraw (https://www.figdraw.com/).
Author contributions
This review was conceived and designed by all authors. The original manuscript was drafted by Zitong Wang, with all authors contributing to its revision. Ling Zhang was responsible for project administration. Chuan Qin contributed to supervision and funding acquisition. The final version was reviewed and approved by all authors.
Funding
This work was supported by Chinese Academy of Medical Sciences Innovation Fund for Medical Sciences(2021-I2M-1-034), Non-profit Central Research Institute Fund of Chinese Academy of Medical Sciences (2023-PT180-01).
Data availability
No datasets were generated or analysed during the current study.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
All authors read and approved the final manuscript.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Jack CJ, Andrews JS, Beach TG, Buracchio T, Dunn B, Graf A, Hansson O, Ho C, Jagust W, Mcdade E et al. Revised criteria for diagnosis and staging of alzheimer’s disease: alzheimer’s association workgroup. Alzheimers Dement. 2024;20(8):5143–5169. [DOI] [PMC free article] [PubMed]
- 2.Scheltens P, De Strooper B, Kivipelto M, Holstege H, Chételat G, Teunissen CE, Cummings J, van der Flier WM. Alzheimer’s disease. Lancet. 2021;397(10284):1577–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.2024 alzheimer’s disease facts and figures. Alzheimers Dement. 2024;20(5):3708–821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chen S, Cao Z, Nandi A, Counts N, Jiao L, Prettner K, Kuhn M, Seligman B, Tortorice D, Vigo D, et al. The global macroeconomic burden of alzheimer’s disease and other dementias: estimates and projections for 152 countries or territories. Lancet Glob Health. 2024;12(9):e1534–43. [DOI] [PubMed] [Google Scholar]
- 5.Long JM, Holtzman DM. Alzheimer disease: an update on pathobiology and treatment strategies. Cell. 2019;179(2):312–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Liu PP, Xie Y, Meng XY, Kang JS. History and progress of hypotheses and clinical trials for alzheimer’s disease. Signal Transduct Target Ther. 2019;4:29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Breitner JC. Nsaids and alzheimer’s disease: how Far to generalise from trials? Lancet Neurol. 2003;2(9):527. [DOI] [PubMed] [Google Scholar]
- 8.Weggen S, Rogers M, Eriksen J. Nsaids: small molecules for prevention of alzheimer’s disease or precursors for future drug development? Trends Pharmacol Sci. 2007;28(10):536–43. [DOI] [PubMed] [Google Scholar]
- 9.Malkki H. Alzheimer disease: Nsaids protect neurons and preserve memory in a mouse model of ad. Nat Rev Neurol. 2016;12(7):370–1. [DOI] [PubMed] [Google Scholar]
- 10.van Olst L, Kamermans A, Halters S, van der Pol S, Rodriguez E, Verberk I, Verberk S, Wessels D, Rodriguez-Mogeda C, Verhoeff J, et al. Adaptive immune changes associate with clinical progression of alzheimer’s disease. Mol Neurodegener. 2024;19(1):38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Stincone A, Prigione A, Cramer T, Wamelink MM, Campbell K, Cheung E, Olin-Sandoval V, Grüning NM, Krüger A, Tauqeer AM, et al. The return of metabolism: biochemistry and physiology of the Pentose phosphate pathway. Biol Rev Camb Philos Soc. 2015;90(3):927–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lepiarz-Raba I, Gbadamosi I, Florea R, Paolicelli RC, Jawaid A. Metabolic regulation of microglial phagocytosis: implications for alzheimer’s disease therapeutics. Transl Neurodegener. 2023;12(1):48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zhang X, Chen C, Liu Y. Navigating the metabolic maze: anomalies in fatty acid and cholesterol processes in alzheimer’s astrocytes. Alzheimers Res Ther. 2024;16(1):63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yin F. Lipid metabolism and alzheimer’s disease: clinical evidence, mechanistic link and therapeutic promise. Febs J. 2023;290(6):1420–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hu T, Liu CH, Lei M, Zeng Q, Li L, Tang H, Zhang N. Metabolic regulation of the immune system in health and diseases: mechanisms and interventions. Signal Transduct Target Ther. 2024;9(1):268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ganeshan K, Chawla A. Metabolic regulation of immune responses. Annu Rev Immunol. 2014;32:609–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Boothby M, Rickert RC. Metabolic regulation of the immune humoral response. Immunity. 2017;46(5):743–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Parkin J, Cohen B. An overview of the immune system. Lancet. 2001;357(9270):1777–89. [DOI] [PubMed] [Google Scholar]
- 19.Alam R, Gorska M. 3. Lymphocytes. J Allergy Clin Immunol. 2003;111(2 Suppl):S476–85. [DOI] [PubMed] [Google Scholar]
- 20.van Olst L, Coenen L, Nieuwland JM, Rodriguez-Mogeda C, de Wit NM, Kamermans A, Middeldorp J, de Vries HE. Crossing borders in alzheimer’s disease: a t cell’s perspective. Adv Drug Deliv Rev. 2022;188:114398. [DOI] [PubMed] [Google Scholar]
- 21.Rustenhoven J, Kipnis J. Brain borders at the central stage of neuroimmunology. Nature. 2022;612(7940):417–29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Xu J, Ma C, Hua M, Li J, Xiang Z, Wu J. Cns and Cns diseases in relation to their immune system. Front Immunol. 2022;13:1063928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Heneka MT, van der Flier WM, Jessen F, Hoozemanns J, Thal DR, Boche D, Brosseron F, Teunissen C, Zetterberg H, Jacobs AH et al. Neuroinflammation in alzheimer disease. Nat Rev Immunol. 2024;25(5):321–352. [DOI] [PubMed]
- 24.Dalmau GA, Glavan M, Samawar S, Kapupara K, Kelk J, Rubio M, Fumagalli S, Sorokin L, Vivien D, Prinz M. The niche matters: origin, function and fate of cns-associated macrophages during health and disease. Acta Neuropathol. 2024;147(1):37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zipp F, Bittner S, Schafer DP. Cytokines as emerging regulators of central nervous system synapses. Immunity. 2023;56(5):914–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Colonna M, Butovsky O. Microglia function in the central nervous system during health and neurodegeneration. Annu Rev Immunol. 2017;35:441–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Marinelli S, Basilico B, Marrone MC, Ragozzino D. Microglia-neuron crosstalk: signaling mechanism and control of synaptic transmission. Semin Cell Dev Biol. 2019;94:138–51. [DOI] [PubMed] [Google Scholar]
- 28.Deng Q, Wu C, Parker E, Liu TC, Duan R, Yang L. Microglia and astrocytes in alzheimer’s disease: significance and summary of recent advances. Aging Dis. 2024;15(4):1537–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cserép C, Pósfai B, Dénes Á. Shaping neuronal fate: functional heterogeneity of direct microglia-neuron interactions. Neuron. 2021;109(2):222–40. [DOI] [PubMed] [Google Scholar]
- 30.Prinz M, Masuda T, Wheeler MA, Quintana FJ. Microglia and central nervous system-associated macrophages-from origin to disease modulation. Annu Rev Immunol. 2021;39:251–77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Liu M, Pan J, Li X, Zhang X, Tian F, Li M, Wu X, Zhang L, Qin C. Interleukin-6 deficiency reduces neuroinflammation by inhibiting the stat3-cgas-sting pathway in alzheimer’s disease mice. J Neuroinflammation. 2024;21(1):282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kwon HS, Koh SH. Neuroinflammation in neurodegenerative disorders: the roles of microglia and astrocytes. Transl Neurodegener. 2020;9(1):42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sun N, Victor MB, Park YP, Xiong X, Scannail AN, Leary N, Prosper S, Viswanathan S, Luna X, Boix CA, et al. Human microglial state dynamics in alzheimer’s disease progression. Cell. 2023;186(20):4386–e440329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zhang D, Zhang Y, Pan J, Cao J, Sun X, Li X, Zhang L, Qin C. Degradation of nlrp3 by p62-dependent-autophagy improves cognitive function in alzheimer’s disease by maintaining the phagocytic function of microglia. Cns Neurosci Ther. 2023;29(10):2826–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Endo F, Kasai A, Soto JS, Yu X, Qu Z, Hashimoto H, Gradinaru V, Kawaguchi R, Khakh BS. Molecular basis of astrocyte diversity and morphology across the Cns in health and disease. Science. 2022;378(6619):eadc9020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Jiwaji Z, Tiwari SS, Avilés-Reyes RX, Hooley M, Hampton D, Torvell M, Johnson DA, Mcqueen J, Baxter P, Sabari-Sankar K, et al. Reactive astrocytes acquire neuroprotective as well as deleterious signatures in response to Tau and Aß pathology. Nat Commun. 2022;13(1):135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Singh D. Astrocytic and microglial cells as the modulators of neuroinflammation in alzheimer’s disease. J Neuroinflammation. 2022;19(1):206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Farina C, Aloisi F, Meinl E. Astrocytes are active players in cerebral innate immunity. Trends Immunol. 2007;28(3):138–45. [DOI] [PubMed] [Google Scholar]
- 39.Shah D, Gsell W, Wahis J, Luckett ES, Jamoulle T, Vermaercke B, Preman P, Moechars D, Hendrickx V, Jaspers T, et al. Astrocyte calcium dysfunction causes early network hyperactivity in alzheimer’s disease. Cell Rep. 2022;40(8):111280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Dalakas MC, Alexopoulos H, Spaeth PJ. Complement in neurological disorders and emerging complement-targeted therapeutics. Nat Rev Neurol. 2020;16(11):601–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Veteleanu A, Stevenson-Hoare J, Keat S, Daskoulidou N, Zetterberg H, Heslegrave A, Escott-Price V, Williams J, Sims R, Zelek WM, et al. Alzheimer’s disease-associated complement gene variants influence plasma complement protein levels. J Neuroinflammation. 2023;20(1):169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Veteleanu A, Pape S, Davies K, Kodosaki E, Hye A, Zelek WM, Strydom A, Morgan BP. Complement dysregulation and alzheimer’s disease in down syndrome. Alzheimers Dement. 2023;19(4):1383–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Dejanovic B, Wu T, Tsai MC, Graykowski D, Gandham VD, Rose CM, Bakalarski CE, Ngu H, Wang Y, Pandey S, et al. Complement c1q-dependent excitatory and inhibitory synapse elimination by astrocytes and microglia in alzheimer’s disease mouse models. Nat Aging. 2022;2(9):837–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zhou J, Wade SD, Graykowski D, Xiao MF, Zhao B, Giannini L, Hanson JE, van Swieten JC, Sheng M, Worley PF, et al. The neuronal pentraxin nptx2 regulates complement activity and restrains microglia-mediated synapse loss in neurodegeneration. Sci Transl Med. 2023;15(689):eadf0141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Lv Z, Chen L, Chen P, Peng H, Rong Y, Hong W, Zhou Q, Li N, Li B, Paolicelli RC, et al. Clearance of β-amyloid and synapses by the optogenetic depolarization of microglia is complement selective. Neuron. 2024;112(5):740–e7547. [DOI] [PubMed] [Google Scholar]
- 46.Hong S, Beja-Glasser VF, Nfonoyim BM, Frouin A, Li S, Ramakrishnan S, Merry KM, Shi Q, Rosenthal A, Barres BA, et al. Complement and microglia mediate early synapse loss in alzheimer mouse models. Science. 2016;352(6286):712–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hu M, Li T, Ma X, Liu S, Li C, Huang Z, Lin Y, Wu R, Wang S, Lu D, et al. Macrophage lineage cells-derived migrasomes activate complement-dependent blood-brain barrier damage in cerebral amyloid angiopathy mouse model. Nat Commun. 2023;14(1):3945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Gate D, Saligrama N, Leventhal O, Yang AC, Unger MS, Middeldorp J, Chen K, Lehallier B, Channappa D, De Los SM, et al. Clonally expanded cd8 t cells patrol the cerebrospinal fluid in alzheimer’s disease. Nature. 2020;577(7790):399–404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Xu H, Jia J. Single-cell Rna sequencing of peripheral blood reveals immune cell signatures in alzheimer’s disease. Front Immunol. 2021;12:645666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zhang Q, Yang G, Luo Y, Jiang L, Chi H, Tian G. Neuroinflammation in alzheimer’s disease: insights from peripheral immune cells. Immun Ageing. 2024;21(1):38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Dai L, Shen Y. Insights into t-cell dysfunction in alzheimer’s disease. Aging Cell. 2021;20(12):e13511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Machhi J, Yeapuri P, Lu Y, Foster E, Chikhale R, Herskovitz J, Namminga KL, Olson KE, Abdelmoaty MM, Gao J, et al. Cd4 + effector t cells accelerate alzheimer’s disease in mice. J Neuroinflammation. 2021;18(1):272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Chen X, Firulyova M, Manis M, Herz J, Smirnov I, Aladyeva E, Wang C, Bao X, Finn MB, Hu H, et al. Microglia-mediated t cell infiltration drives neurodegeneration in tauopathy. Nature. 2023;615(7953):668–77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Feng Y, Fan J, Cheng Y, Dai Q, Ma S. Stress regulates alzheimer’s disease progression via selective enrichment of cd8(+) t cells. Cell Rep. 2023;42(10):113313. [DOI] [PubMed] [Google Scholar]
- 55.Unger MS, Li E, Scharnagl L, Poupardin R, Altendorfer B, Mrowetz H, Hutter-Paier B, Weiger TM, Heneka MT, Attems J, et al. Cd8(+) t-cells infiltrate alzheimer’s disease brains and regulate neuronal- and synapse-related gene expression in app-ps1 Transgenic mice. Brain Behav Immun. 2020;89:67–86. [DOI] [PubMed] [Google Scholar]
- 56.Su W, Saravia J, Risch I, Rankin S, Guy C, Chapman NM, Shi H, Sun Y, Kc A, Li W, et al. Cxcr6 orchestrates brain cd8(+) t cell residency and limits mouse alzheimer’s disease pathology. Nat Immunol. 2023;24(10):1735–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Jorfi M, Park J, Hall CK, Lin CJ, Chen M, von Maydell D, Kruskop JM, Kang B, Choi Y, Prokopenko D, et al. Infiltrating cd8(+) t cells exacerbate alzheimer’s disease pathology in a 3d human neuroimmune axis model. Nat Neurosci. 2023;26(9):1489–504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Yeapuri P, Machhi J, Lu Y, Abdelmoaty MM, Kadry R, Patel M, Bhattarai S, Lu E, Namminga KL, Olson KE, et al. Amyloid-β specific regulatory t cells attenuate alzheimer’s disease pathobiology in app/ps1 mice. Mol Neurodegener. 2023;18(1):97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Yang H, Park SY, Baek H, Lee C, Chung G, Liu X, Lee JH, Kim B, Kwon M, Choi H, et al. Adoptive therapy with amyloid-β specific regulatory t cells alleviates alzheimer’s disease. Theranostics. 2022;12(18):7668–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Sabatino JJ, Pröbstel AK, Zamvil SS. B cells in autoimmune and neurodegenerative central nervous system diseases. Nat Rev Neurosci. 2019;20(12):728–45. [DOI] [PubMed] [Google Scholar]
- 61.Dezfulian M. A new alzheimer’s disease cell model using b cells to induce beta amyloid plaque formation and increase Tnf alpha expression. Int Immunopharmacol. 2018;59:106–12. [DOI] [PubMed] [Google Scholar]
- 62.Park JC, Noh J, Jang S, Kim KH, Choi H, Lee D, Kim J, Chung J, Lee DY, Lee Y, et al. Association of b cell profile and receptor repertoire with the progression of alzheimer’s disease. Cell Rep. 2022;40(12):111391. [DOI] [PubMed] [Google Scholar]
- 63.Kim K, Wang X, Ragonnaud E, Bodogai M, Illouz T, Deluca M, Mcdevitt RA, Gusev F, Okun E, Rogaev E, et al. Therapeutic b-cell depletion reverses progression of alzheimer’s disease. Nat Commun. 2021;12(1):2185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Feng W, Zhang Y, Ding S, Chen S, Wang T, Wang Z, Zou Y, Sheng C, Chen Y, Pang Y, et al. B lymphocytes ameliorate alzheimer’s disease-like neuropathology via interleukin-35. Brain Behav Immun. 2023;108:16–31. [DOI] [PubMed] [Google Scholar]
- 65.Xiong LL, Xue LL, Du RL, Niu RZ, Chen L, Chen J, Hu Q, Tan YX, Shang HF, Liu J, et al. Single-cell Rna sequencing reveals b cell-related molecular biomarkers for alzheimer’s disease. Exp Mol Med. 2021;53(12):1888–901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Matousek SB, Ghosh S, Shaftel SS, Kyrkanides S, Olschowka JA, O’Banion MK. Chronic il-1β-mediated neuroinflammation mitigates amyloid pathology in a mouse model of alzheimer’s disease without inducing overt neurodegeneration. J Neuroimmune Pharmacol. 2012;7(1):156–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Mcalpine CS, Park J, Griciuc A, Kim E, Choi SH, Iwamoto Y, Kiss MG, Christie KA, Vinegoni C, Poller WC, et al. Astrocytic interleukin-3 programs microglia and limits alzheimer’s disease. Nature. 2021;595(7869):701–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Vom BJ, Prokop S, Miller KR, Obst J, Kälin RE, Lopategui-Cabezas I, Wegner A, Mair F, Schipke CG, Peters O, et al. Inhibition of il-12/il-23 signaling reduces alzheimer’s disease-like pathology and cognitive decline. Nat Med. 2012;18(12):1812–9. [DOI] [PubMed] [Google Scholar]
- 69.Eede P, Obst J, Benke E, Yvon-Durocher G, Richard BC, Gimber N, Schmoranzer J, Böddrich A, Wanker EE, Prokop S, et al. Interleukin-12/23 deficiency differentially affects pathology in male and female alzheimer’s disease-like mice. Embo Rep. 2020;21(3):e48530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Puntambekar SS, Moutinho M, Lin PB, Jadhav V, Tumbleson-Brink D, Balaji A, Benito MA, Xu G, Oblak A, Lasagna-Reeves CA, et al. Cx3cr1 deficiency aggravates amyloid driven neuronal pathology and cognitive decline in alzheimer’s disease. Mol Neurodegener. 2022;17(1):47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Griciuc A, Patel S, Federico AN, Choi SH, Innes BJ, Oram MK, Cereghetti G, Mcginty D, Anselmo A, Sadreyev RI, et al. Trem2 acts downstream of cd33 in modulating microglial pathology in alzheimer’s disease. Neuron. 2019;103(5):820–e8357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Jiang T, Zhang YD, Chen Q, Gao Q, Zhu XC, Zhou JS, Shi JQ, Lu H, Tan L, Yu JT. Trem2 modifies microglial phenotype and provides neuroprotection in p301s Tau Transgenic mice. Neuropharmacology. 2016;105:196–206. [DOI] [PubMed] [Google Scholar]
- 73.Romero J, Toral-Rios D, Yu J, Paul SM, Cashikar AG. 25-hydroxycholesterol promotes brain cytokine production and leukocyte infiltration in a mouse model of lipopolysaccharide-induced neuroinflammation. J Neuroinflammation. 2024;21(1):251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Belarbi K, Jopson T, Tweedie D, Arellano C, Luo W, Greig NH, Rosi S. Tnf-α protein synthesis inhibitor restores neuronal function and reverses cognitive deficits induced by chronic neuroinflammation. J Neuroinflammation. 2012;9:23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Yang Y, García-Cruzado M, Zeng H, Camprubí-Ferrer L, Bahatyrevich-Kharitonik B, Bachiller S, Deierborg T. Lps priming before plaque deposition impedes microglial activation and restrains aβ pathology in the 5xfad mouse model of alzheimer’s disease. Brain Behav Immun. 2023;113:228–47. [DOI] [PubMed] [Google Scholar]
- 76.Lopez-Rodriguez AB, Hennessy E, Murray CL, Nazmi A, Delaney HJ, Healy D, Fagan SG, Rooney M, Stewart E, Lewis A, et al. Acute systemic inflammation exacerbates neuroinflammation in alzheimer’s disease: il-1β drives amplified responses in primed astrocytes and neuronal network dysfunction. Alzheimers Dement. 2021;17(10):1735–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Zhou J, Yu W, Zhang M, Tian X, Li Y, Lü Y. Imbalance of microglial tlr4/trem2 in lps-treated app/ps1 Transgenic mice: a potential link between alzheimer’s disease and systemic inflammation. Neurochem Res. 2019;44(5):1138–51. [DOI] [PubMed] [Google Scholar]
- 78.Alves S, Churlaud G, Audrain M, Michaelsen-Preusse K, Fol R, Souchet B, Braudeau J, Korte M, Klatzmann D, Cartier N. Interleukin-2 improves amyloid pathology, synaptic failure and memory in alzheimer’s disease mice. Brain. 2017;140(3):826–42. [DOI] [PubMed] [Google Scholar]
- 79.Yuan L, Xie L, Zhang H, Zhang Y, Wei Y, Feng J, Cui L, Tian R, Feng J, Yu D, et al. Low-dose il-2 treatment rescues cognitive deficits by repairing the imbalance between Treg and th17 cells at the middle alzheimer’s disease stage. J Neuroimmune Pharmacol. 2023;18(4):674–89. [DOI] [PubMed] [Google Scholar]
- 80.Brigas HC, Ribeiro M, Coelho JE, Gomes R, Gomez-Murcia V, Carvalho K, Faivre E, Costa-Pereira S, Darrigues J, de Almeida AA, et al. Il-17 triggers the onset of cognitive and synaptic deficits in early stages of alzheimer’s disease. Cell Rep. 2021;36(9):109574. [DOI] [PubMed] [Google Scholar]
- 81.Vellecco V, Saviano A, Raucci F, Casillo GM, Mansour AA, Panza E, Mitidieri E, Femminella GD, Ferrara N, Cirino G, et al. Interleukin-17 (il-17) triggers systemic inflammation, peripheral vascular dysfunction, and related prothrombotic state in a mouse model of alzheimer’s disease. Pharmacol Res. 2023;187:106595. [DOI] [PubMed] [Google Scholar]
- 82.Fu AK, Hung KW, Yuen MY, Zhou X, Mak DS, Chan IC, Cheung TH, Zhang B, Fu WY, Liew FY, et al. Il-33 ameliorates alzheimer’s disease-like pathology and cognitive decline. Proc Natl Acad Sci U S A. 2016;113(19):E2705–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Lau SF, Chen C, Fu WY, Qu JY, Cheung TH, Fu A, Ip NY. Il-33-pu.1 transcriptome reprogramming drives functional state transition and clearance activity of microglia in alzheimer’s disease. Cell Rep. 2020;31(3):107530. [DOI] [PubMed] [Google Scholar]
- 84.Seabrook TJ, Jiang L, Maier M, Lemere CA. Minocycline affects microglia activation, Abeta deposition, and behavior in app-tg mice. Glia. 2006;53(7):776–82. [DOI] [PubMed] [Google Scholar]
- 85.Biscaro B, Lindvall O, Tesco G, Ekdahl CT, Nitsch RM. Inhibition of microglial activation protects hippocampal neurogenesis and improves cognitive deficits in a Transgenic mouse model for alzheimer’s disease. Neurodegener Dis. 2012;9(4):187–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Chavant F, Deguil J, Pain S, Ingrand I, Milin S, Fauconneau B, Pérault-Pochat MC, Lafay-Chebassier C. Imipramine, in part through tumor necrosis factor alpha inhibition, prevents cognitive decline and beta-amyloid accumulation in a mouse model of alzheimer’s disease. J Pharmacol Exp Ther. 2010;332(2):505–14. [DOI] [PubMed] [Google Scholar]
- 87.Gabbita SP, Srivastava MK, Eslami P, Johnson MF, Kobritz NK, Tweedie D, Greig NH, Zemlan FP, Sharma SP, Harris-White ME. Early intervention with a small molecule inhibitor for tumor necrosis factor-α prevents cognitive deficits in a triple Transgenic mouse model of alzheimer’s disease. J Neuroinflammation. 2012;9:99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Tweedie D, Ferguson RA, Fishman K, Frankola KA, Van Praag H, Holloway HW, Luo W, Li Y, Caracciolo L, Russo I, et al. Tumor necrosis factor-α synthesis inhibitor 3,6’-dithiothalidomide attenuates markers of inflammation, alzheimer pathology and behavioral deficits in animal models of neuroinflammation and alzheimer’s disease. J Neuroinflammation. 2012;9:106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Li Y, Fan H, Ni M, Zhang W, Fang F, Sun J, Lyu P, Ma P. Etanercept reduces neuron injury and neuroinflammation via inactivating c-jun n-terminal kinase and nuclear factor-κb pathways in alzheimer’s disease: an in vitro and in vivo investigation. Neuroscience. 2022;484:140–50. [DOI] [PubMed] [Google Scholar]
- 90.Ou W, Ohno Y, Yang J, Chandrashekar DV, Abdullah T, Sun J, Murphy R, Roules C, Jagadeesan N, Cribbs DH et al. Efficacy and safety of a brain-penetrant biologic tnf-α inhibitor in aged app/ps1 mice. Pharmaceutics. 2022;14(10). [DOI] [PMC free article] [PubMed]
- 91.Yoo Y, Neumayer G, Shibuya Y, Mader MM, Wernig M. A cell therapy approach to restore microglial trem2 function in a mouse model of alzheimer’s disease. Cell Stem Cell. 2023;30(8):1043–e10536. [DOI] [PubMed] [Google Scholar]
- 92.Raulin AC, Martens YA, Bu G. Lipoproteins in the central nervous system: from biology to pathobiology. Annu Rev Biochem. 2022;91:731–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Luo J, Yang H, Song BL. Mechanisms and regulation of cholesterol homeostasis. Nat Rev Mol Cell Biol. 2020;21(4):225–45. [DOI] [PubMed] [Google Scholar]
- 94.Hansen SB, Wang H. The shared role of cholesterol in neuronal and peripheral inflammation. Pharmacol Ther. 2023;249:108486. [DOI] [PubMed] [Google Scholar]
- 95.Mi Y, Qi G, Vitali F, Shang Y, Raikes AC, Wang T, Jin Y, Brinton RD, Gu H, Yin F. Loss of fatty acid degradation by astrocytic mitochondria triggers neuroinflammation and neurodegeneration. Nat Metab. 2023;5(3):445–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Farooqui AA, Farooqui T. Phospholipids, sphingolipids, and cholesterol-derived lipid mediators and their role in neurological disorders. Int J Mol Sci. 2024;25(19). [DOI] [PMC free article] [PubMed]
- 97.Zeng Q, Gong Y, Zhu N, Shi Y, Zhang C, Qin L. Lipids and lipid metabolism in cellular senescence: emerging targets for age-related diseases. Ageing Res Rev. 2024;97:102294. [DOI] [PubMed] [Google Scholar]
- 98.Ferguson EL, Zimmerman SC, Jiang C, Choi M, Swinnerton K, Choudhary V, Meyers TJ, Hoffmann TJ, Gilsanz P, Oni-Orisan A, et al. Low- and high-density lipoprotein cholesterol and dementia risk over 17 years of follow-up among members of a large health care plan. Neurology. 2023;101(21):e2172–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Mistry H, Richardson CD, Higginbottom A, Ashford B, Ahamed SU, Moore Z, Matthews FE, Brayne C, Simpson JE, Wharton SB. Relationships of brain cholesterol and cholesterol biosynthetic enzymes to alzheimer’s pathology and dementia in the Cfas population-derived neuropathology cohort. Neurosci Res. 2024;204:22–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Livingston G, Huntley J, Liu KY, Costafreda SG, Selbæk G, Alladi S, Ames D, Banerjee S, Burns A, Brayne C, et al. Dementia prevention, intervention, and care: 2024 report of the lancet standing commission. Lancet. 2024;404(10452):572–628. [DOI] [PubMed] [Google Scholar]
- 101.Kivipelto M, Mangialasche F, Anstey KJ. Pivotal points in the science of dementia risk reduction. Lancet. 2024;404(10452):501–3. [DOI] [PubMed] [Google Scholar]
- 102.Paiva I, Seguin J, Grgurina I, Singh AK, Cosquer B, Plassard D, Tzeplaeff L, Le Gras S, Cotellessa L, Decraene C, et al. Dysregulated expression of cholesterol biosynthetic genes in alzheimer’s disease alters epigenomic signatures of hippocampal neurons. Neurobiol Dis. 2024;198:106538. [DOI] [PubMed] [Google Scholar]
- 103.Spell C, Kölsch H, Lütjohann D, Kerksiek A, Hentschel F, Damian M, von Bergmann K, Rao ML, Maier W, Heun R. Srebp-1a polymorphism influences the risk of alzheimer’s disease in carriers of the apoe4 allele. Dement Geriatr Cogn Disord. 2004;18(3–4):245–9. [DOI] [PubMed] [Google Scholar]
- 104.Barbero-Camps E, Fernández A, Martínez L, Fernández-Checa JC, Colell A. App/ps1 mice overexpressing srebp-2 exhibit combined aβ accumulation and Tau pathology underlying alzheimer’s disease. Hum Mol Genet. 2013;22(17):3460–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Shah SA, Yoon GH, Chung SS, Abid MN, Kim TH, Lee HY, Kim MO. Novel osmotin inhibits srebp2 via the adipor1/ampk/sirt1 pathway to improve alzheimer’s disease neuropathological deficits. Mol Psychiatry. 2017;22(3):407–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Wang C, Zhao F, Shen K, Wang W, Siedlak SL, Lee HG, Phelix CF, Perry G, Shen L, Tang B, et al. The sterol regulatory element-binding protein 2 is dysregulated by Tau alterations in alzheimer disease. Brain Pathol. 2019;29(4):530–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.van der Kant R, Langness VF, Herrera CM, Williams DA, Fong LK, Leestemaker Y, Steenvoorden E, Rynearson KD, Brouwers JF, Helms JB, et al. Cholesterol metabolism is a druggable axis that independently regulates Tau and amyloid-β in ipsc-derived alzheimer’s disease neurons. Cell Stem Cell. 2019;24(3):363–e3759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.West E, Osborne C, Bate C. The cholesterol ester cycle regulates signalling complexes and synapse damage caused by amyloid-β. J Cell Sci. 2017;130(18):3050–9. [DOI] [PubMed] [Google Scholar]
- 109.Hutter-Paier B, Huttunen HJ, Puglielli L, Eckman CB, Kim DY, Hofmeister A, Moir RD, Domnitz SB, Frosch MP, Windisch M, et al. The Acat inhibitor cp-113,818 markedly reduces amyloid pathology in a mouse model of alzheimer’s disease. Neuron. 2004;44(2):227–38. [DOI] [PubMed] [Google Scholar]
- 110.De La Torre AL, Huynh TN, Chang C, Pooler DB, Ness DB, Lewis LD, Pannem S, Feng Y, Samkoe KS, Hickey WF, et al. Stealth liposomes encapsulating a potent acat1/soat1 inhibitor f12511: pharmacokinetic, biodistribution, and toxicity studies in wild-type mice and efficacy studies in triple Transgenic alzheimer’s disease mice. Int J Mol Sci. 2023;24:13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Murphy SR, Chang CC, Dogbevia G, Bryleva EY, Bowen Z, Hasan MT, Chang TY. Acat1 knockdown gene therapy decreases amyloid-β in a mouse model of alzheimer’s disease. Mol Ther. 2013;21(8):1497–506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Montesinos J, Pera M, Larrea D, Guardia-Laguarta C, Agrawal RR, Velasco KR, Yun TD, Stavrovskaya IG, Xu Y, Koo SY, et al. The alzheimer’s disease-associated c99 fragment of app regulates cellular cholesterol trafficking. Embo J. 2020;39(20):e103791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Li D, Zhang J, Liu Q. Brain cell type-specific cholesterol metabolism and implications for learning and memory. Trends Neurosci. 2022;45(5):401–14. [DOI] [PubMed] [Google Scholar]
- 114.Fortea J, Pegueroles J, Alcolea D, Belbin O, Dols-Icardo O, Vaqué-Alcázar L, Videla L, Gispert JD, Suárez-Calvet M, Johnson SC, et al. Apoe4 homozygozity represents a distinct genetic form of alzheimer’s disease. Nat Med. 2024;30(5):1284–91. [DOI] [PubMed] [Google Scholar]
- 115.Wang Y, Li F, Qin Q, Li T, Wang Q, Li Y, Li Y, Jia J. Influence of apoe ε4 on performance of csf biomarkers in differentiating clinical alzheimer’s disease. J Prev Alzheimers Dis. 2025;12(4):100065. [DOI] [PubMed]
- 116.Qi G, Mi Y, Shi X, Gu H, Brinton RD, Yin F. Apoe4 impairs neuron-astrocyte coupling of fatty acid metabolism. Cell Rep. 2021;34(1):108572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Yang LG, March ZM, Stephenson RA, Narayan PS. Apolipoprotein e in lipid metabolism and neurodegenerative disease. Trends Endocrinol Metab. 2023;34(8):430–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Shi Y, Andhey PS, Ising C, Wang K, Snipes LL, Boyer K, Lawson S, Yamada K, Qin W, Manis M, et al. Overexpressing low-density lipoprotein receptor reduces tau-associated neurodegeneration in relation to apoe-linked mechanisms. Neuron. 2021;109(15):2413–e24267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Chen L, Zhao ZW, Zeng PH, Zhou YJ, Yin WJ. Molecular mechanisms for abca1-mediated cholesterol efflux. Cell Cycle. 2022;21(11):1121–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Velma GR, Laham MS, Lewandowski C, Valencia-Olvera AC, Balu D, Moore A, Ackerman-Berrier M, Rychetsky P, Penton C, Musku SR, et al. Nonlipogenic abca1 inducers (nlai) for alzheimer’s disease validated in a mouse model expressing human apoe3/apoe4. J Med Chem. 2024;67(17):15061–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Nordestgaard LT, Tybjærg-Hansen A, Nordestgaard BG, Frikke-Schmidt R. Loss-of-function mutation in abca1 and risk of alzheimer’s disease and cerebrovascular disease. Alzheimers Dement. 2015;11(12):1430–8. [DOI] [PubMed] [Google Scholar]
- 122.Bossaerts L, Cacace R, Van Broeckhoven C. The role of atp-binding cassette subfamily a in the etiology of alzheimer’s disease. Mol Neurodegener. 2022;17(1):31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Lewandowski CT, Laham MS, Thatcher G. Remembering your a, b, c’s: alzheimer’s disease and abca1. Acta Pharm Sin B. 2022;12(3):995–1018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Paseban T, Alavi MS, Etemad L, Roohbakhsh A. The role of the atp-binding cassette a1 (abca1) in neurological disorders: a mechanistic review. Expert Opin Ther Targets. 2023;27(7):531–52. [DOI] [PubMed] [Google Scholar]
- 125.Holstege H, Hulsman M, Charbonnier C, Grenier-Boley B, Quenez O, Grozeva D, van Rooij J, Sims R, Ahmad S, Amin N, et al. Exome sequencing identifies rare damaging variants in atp8b4 and abca1 as risk factors for alzheimer’s disease. Nat Genet. 2022;54(12):1786–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Moulton MJ, Barish S, Ralhan I, Chang J, Goodman LD, Harland JG, Marcogliese PC, Johansson JO, Ioannou MS, Bellen HJ. Neuronal ros-induced glial lipid droplet formation is altered by loss of alzheimer’s disease-associated genes. Proc Natl Acad Sci U S a. 2021;118(52). [DOI] [PMC free article] [PubMed]
- 127.Dakterzada F, Jové M, Cantero JL, Mota-Martorell N, Pamplona R, Piñoll-Ripoll G. The shift in the fatty acid composition of the Circulating lipidome in alzheimer’s disease. Alzheimers Dement. 2024;20(5):3322–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Goozee K, Chatterjee P, James I, Shen K, Sohrabi HR, Asih PR, Dave P, Ball B, Manyan C, Taddei K, et al. Alterations in erythrocyte fatty acid composition in preclinical alzheimer’s disease. Sci Rep. 2017;7(1):676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Chen C, Liao J, Xia Y, Liu X, Jones R, Haran J, Mccormick B, Sampson TR, Alam A, Ye K. Gut microbiota regulate alzheimer’s disease pathologies and cognitive disorders via pufa-associated neuroinflammation. Gut. 2022;71(11):2233–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Virmani A, Pinto L, Bauermann O, Zerelli S, Diedenhofen A, Binienda ZK, Ali SF, van der Leij FR. The carnitine palmitoyl transferase (cpt) system and possible relevance for neuropsychiatric and neurological conditions. Mol Neurobiol. 2015;52(2):826–36. [DOI] [PubMed] [Google Scholar]
- 131.Sarnowski C, Huan T, Ma Y, Joehanes R, Beiser A, Decarli CS, Heard-Costa NL, Levy D, Lin H, Liu CT, et al. Multi-tissue epigenetic analysis identifies distinct associations underlying insulin resistance and alzheimer’s disease at cpt1a locus. Clin Epigenetics. 2023;15(1):173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Konttinen H, Gureviciene I, Oksanen M, Grubman A, Loppi S, Huuskonen MT, Korhonen P, Lampinen R, Keuters M, Belaya I, et al. Pparβ/δ-agonist gw0742 ameliorates dysfunction in fatty acid oxidation in psen1δe9 astrocytes. Glia. 2019;67(1):146–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Medrano-Jiménez E, Jiménez-Ferrer CI, Pedraza-Escalona M, Ramírez-Serrano CE, álvarez-Arellano L, Cortés-Mendoza J, Herrera-Ruiz M, Jiménez-Ferrer E, Zamilpa A, Tortoriello J, et al. Malva parviflora extract ameliorates the deleterious effects of a high fat diet on the cognitive deficit in a mouse model of alzheimer’s disease by restoring microglial function via a ppar-γ-dependent mechanism. J Neuroinflammation. 2019;16(1):143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Elahi M, Motoi Y, Shimonaka S, Ishida Y, Hioki H, Takanashi M, Ishiguro K, Imai Y, Hattori N. High-fat diet-induced activation of sgk1 promotes alzheimer’s disease-associated Tau pathology. Hum Mol Genet. 2021;30(18):1693–710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Gannon OJ, Robison LS, Salinero AE, Abi-Ghanem C, Mansour FM, Kelly RD, Tyagi A, Brawley RR, Ogg JD, Zuloaga KL. High-fat diet exacerbates cognitive decline in mouse models of alzheimer’s disease and mixed dementia in a sex-dependent manner. J Neuroinflammation. 2022;19(1):110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Suzuki R, Ferris HA, Chee MJ, Maratos-Flier E, Kahn CR. Reduction of the cholesterol sensor Scap in the brains of mice causes impaired synaptic transmission and altered cognitive function. Plos Biol. 2013;11(4):e1001532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Kaji S, Berghoff SA, Spieth L, Schlaphoff L, Sasmita AO, Vitale S, Büschgens L, Kedia S, Zirngibl M, Nazarenko T, et al. Apolipoprotein e aggregation in microglia initiates alzheimer’s disease pathology by seeding β-amyloidosis. Immunity. 2024;57(11):2651–e266812. [DOI] [PubMed] [Google Scholar]
- 138.Yin Z, Rosenzweig N, Kleemann KL, Zhang X, Brandão W, Margeta MA, Schroeder C, Sivanathan KN, Silveira S, Gauthier C, et al. Apoe4 impairs the microglial response in alzheimer’s disease by inducing tgfβ-mediated checkpoints. Nat Immunol. 2023;24(11):1839–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Mahan TE, Wang C, Bao X, Choudhury A, Ulrich JD, Holtzman DM. Selective reduction of astrocyte apoe3 and apoe4 strongly reduces aβ accumulation and plaque-related pathology in a mouse model of amyloidosis. Mol Neurodegener. 2022;17(1):13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.He K, Nie L, Zhou Q, Rahman SU, Liu J, Yang X, Li S. Proteomic profiles of the early mitochondrial changes in app/ps1 and apoe4 Transgenic mice models of alzheimer’s disease. J Proteome Res. 2019;18(6):2632–42. [DOI] [PubMed] [Google Scholar]
- 141.Qian Z, Wang Z, Li B, Meng X, Kuang Z, Li Y, Yang Y, Ye K. Thy1-apoe4/c/ebpβ double Transgenic mice act as a sporadic model with alzheimer’s disease. Mol Psychiatry. 2024;29(10):3040–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Wang ZH, Xia Y, Wu Z, Kang SS, Zhang JC, Liu P, Liu X, Song W, Huin V, Dhaenens CM, et al. Neuronal apoe4 stimulates c/ebpβ activation, promoting alzheimer’s disease pathology in a mouse model. Prog Neurobiol. 2022;209:102212. [DOI] [PubMed] [Google Scholar]
- 143.Rauch JN, Luna G, Guzman E, Audouard M, Challis C, Sibih YE, Leshuk C, Hernandez I, Wegmann S, Hyman BT, et al. Lrp1 is a master regulator of Tau uptake and spread. Nature. 2020;580(7803):381–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Ma Q, Zhao Z, Sagare AP, Wu Y, Wang M, Owens NC, Verghese PB, Herz J, Holtzman DM, Zlokovic BV. Blood-brain barrier-associated pericytes internalize and clear aggregated amyloid-β42 by lrp1-dependent Apolipoprotein e isoform-specific mechanism. Mol Neurodegener. 2018;13(1):57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Litvinchuk A, Suh JH, Guo JL, Lin K, Davis SS, Bien-Ly N, Tycksen E, Tabor GT, Remolina SJ, Manis M, et al. Amelioration of Tau and apoe4-linked glial lipid accumulation and neurodegeneration with an Lxr agonist. Neuron. 2024;112(3):384–e4038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Ban SY, Nam Y, Do TT, Kim BH, Shin SJ, Thi NM, Kim J, Moon M, Park JT. Liver-x receptor β-selective agonist ce9a215 regulates alzheimer’s disease-associated pathology in a 3xtg-ad mouse model. Biomed Pharmacother. 2025;184:117895. [DOI] [PubMed] [Google Scholar]
- 147.Suon S, Zhao J, Villarreal SA, Anumula N, Liu M, Carangia LM, Renger JJ, Zerbinatti CV. Systemic treatment with liver x receptor agonists raises Apolipoprotein e, cholesterol, and amyloid-β peptides in the cerebral spinal fluid of rats. Mol Neurodegener. 2010;5:44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Fernández A, Llacuna L, Fernández-Checa JC, Colell A. Mitochondrial cholesterol loading exacerbates amyloid beta peptide-induced inflammation and neurotoxicity. J Neurosci. 2009;29(20):6394–405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Mendoza-Oliva A, Ferrera P, Fragoso-Medina J, Arias C. Lovastatin differentially affects neuronal cholesterol and amyloid-β production in vivo and in vitro. Cns Neurosci Ther. 2015;21(8):631–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Valencia-Olvera AC, Balu D, Faulk N, Amiridis A, Wang Y, Pham C, Avila-Munoz E, York JM, Thatcher G, Ladu MJ. Inhibition of Acat as a therapeutic target for alzheimer’s disease is independent of apoe4 lipidation. Neurotherapeutics. 2023;20(4):1120–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Zhang YY, Fan YC, Wang M, Wang D, Li XH. Atorvastatin attenuates the production of il-1β, il-6, and tnf-α in the hippocampus of an amyloid β1-42-induced rat model of alzheimer’s disease. Clin Interv Aging. 2013;8:103–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Sadanandan J, Sathyanesan M, Newton SS. Aging alters the expression of trophic factors and tight junction proteins in the mouse choroid plexus. Fluids Barriers Cns. 2024;21(1):77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Baruch K, Ron-Harel N, Gal H, Deczkowska A, Shifrut E, Ndifon W, Mirlas-Neisberg N, Cardon M, Vaknin I, Cahalon L, et al. Cns-specific immunity at the choroid plexus shifts toward destructive th2 inflammation in brain aging. Proc Natl Acad Sci U S A. 2013;110(6):2264–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Kunis G, Baruch K, Rosenzweig N, Kertser A, Miller O, Berkutzki T, Schwartz M. Ifn-γ-dependent activation of the brain’s choroid plexus for Cns immune surveillance and repair. Brain. 2013;136(Pt 11):3427–40. [DOI] [PubMed] [Google Scholar]
- 155.Dai YB, Wu WF, Huang B, Miao YF, Nadarshina S, Warner M, Gustafsson JÅ. Liver x receptors regulate cerebrospinal fluid production. Mol Psychiatry. 2016;21(6):844–56. [DOI] [PubMed] [Google Scholar]
- 156.Fujiyoshi M, Ohtsuki S, Hori S, Tachikawa M, Terasaki T. 24s-hydroxycholesterol induces cholesterol release from choroid plexus epithelial cells in an apical- and Apoe isoform-dependent manner concomitantly with the induction of abca1 and abcg1 expression. J Neurochem. 2007;100(4):968–78. [DOI] [PubMed] [Google Scholar]
- 157.Tsitsou-Kampeli A, Suzzi S, Kenigsbuch M, Satomi A, Strobelt R, Singer O, Feldmesser E, Purnapatre M, Colaiuta SP, David E, et al. Cholesterol 24-hydroxylase at the choroid plexus contributes to brain immune homeostasis. Cell Rep Med. 2023;4(11):101278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Tsitsou-Kampeli A, Suzzi S, Schwartz M. The immune and metabolic milieu of the choroid plexus as a potential target in brain protection. Trends Neurosci. 2024;47(8):573–82. [DOI] [PubMed] [Google Scholar]
- 159.Montagne A, Zhao Z, Zlokovic BV. Alzheimer’s disease: a matter of blood-brain barrier dysfunction? J Exp Med. 2017;214(11):3151–69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Zlokovic BV. Neurovascular pathways to neurodegeneration in alzheimer’s disease and other disorders. Nat Rev Neurosci. 2011;12(12):723–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Więckowska-Gacek A, Mietelska-Porowska A, Wydrych M, Wojda U. Western diet as a trigger of alzheimer’s disease: from metabolic syndrome and systemic inflammation to neuroinflammation and neurodegeneration. Ageing Res Rev. 2021;70:101397. [DOI] [PubMed] [Google Scholar]
- 162.Robert J, Button EB, Stukas S, Boyce GK, Gibbs E, Cowan CM, Gilmour M, Cheng WH, Soo SK, Yuen B, et al. High-density lipoproteins suppress aβ-induced Pbmc adhesion to human endothelial cells in bioengineered vessels and in monoculture. Mol Neurodegener. 2017;12(1):60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Button EB, Gilmour M, Cheema HK, Martin EM, Agbay A, Robert J, Wellington CL. Vasoprotective functions of high-density lipoproteins relevant to alzheimer’s disease are partially conserved in apolipoprotein b-depleted plasma. Int J Mol Sci. 2019;20(3). [DOI] [PMC free article] [PubMed]
- 164.Robert J, Button EB, Martin EM, Mcalary L, Gidden Z, Gilmour M, Boyce G, Caffrey TM, Agbay A, Clark A, et al. Cerebrovascular amyloid angiopathy in bioengineered vessels is reduced by high-density lipoprotein particles enriched in Apolipoprotein e. Mol Neurodegener. 2020;15(1):23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Barisano G, Kisler K, Wilkinson B, Nikolakopoulou AM, Sagare AP, Wang Y, Gilliam W, Huuskonen MT, Hung ST, Ichida JK et al. A multi-omics analysis of blood-brain barrier and synaptic dysfunction in apoe4 mice. J Exp Med. 2022;219(11). [DOI] [PMC free article] [PubMed]
- 166.Agrawal M, Ajazuddin, Tripathi DK, Saraf S, Saraf S, Antimisiaris SG, Mourtas S, Hammarlund-Udenaes M, Alexander A. Recent advancements in liposomes targeting strategies to cross blood-brain barrier (bbb) for the treatment of alzheimer’s disease. J Control Release. 2017;260:61–77. [DOI] [PubMed] [Google Scholar]
- 167.Gao C, Liu Y, Zhang TL, Luo Y, Gao J, Chu JJ, Gong BF, Chen XH, Yin T, Zhang J, et al. Biomembrane-derived nanoparticles in alzheimer’s disease therapy: a comprehensive review of synthetic lipid nanoparticles and natural cell-derived vesicles. Int J Nanomed. 2023;18:7441–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Iliff JJ, Wang M, Liao Y, Plogg BA, Peng W, Gundersen GA, Benveniste H, Vates GE, Deane R, Goldman SA, et al. A paravascular pathway facilitates Csf flow through the brain parenchyma and the clearance of interstitial solutes, including amyloid β. Sci Transl Med. 2012;4(147):147ra111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Harrison IF, Ismail O, Machhada A, Colgan N, Ohene Y, Nahavandi P, Ahmed Z, Fisher A, Meftah S, Murray TK, et al. Impaired glymphatic function and clearance of Tau in an alzheimer’s disease model. Brain. 2020;143(8):2576–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Rangroo TV, Thrane AS, Plog BA, Thiyagarajan M, Iliff JJ, Deane R, Nagelhus EA, Nedergaard M. Paravascular microcirculation facilitates rapid lipid transport and astrocyte signaling in the brain. Sci Rep. 2013;3:2582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Lohela TJ, Lilius TO, Nedergaard M. The glymphatic system: implications for drugs for central nervous system diseases. Nat Rev Drug Discov. 2022;21(10):763–79. [DOI] [PubMed] [Google Scholar]
- 172.Rasmussen MK, Mestre H, Nedergaard M. Fluid transport in the brain. Physiol Rev. 2022;102(2):1025–151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Rasmussen MK, Mestre H, Nedergaard M. The glymphatic pathway in neurological disorders. Lancet Neurol. 2018;17(11):1016–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Mestre H, Mori Y, Nedergaard M. The brain’s glymphatic system: current controversies. Trends Neurosci. 2020;43(7):458–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Da MS, Louveau A, Vaccari A, Smirnov I, Cornelison RC, Kingsmore KM, Contarino C, Onengut-Gumuscu S, Farber E, Raper D, et al. Functional aspects of meningeal lymphatics in ageing and alzheimer’s disease. Nature. 2018;560(7717):185–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Ma Y, Han Y. Targeting the brain’s glymphatic pathway: a novel therapeutic approach for cerebral small vessel disease. Neural Regen Res. 2026;21(2):433–442. [DOI] [PMC free article] [PubMed]
- 177.Nedergaard M, Goldman SA. Glymphatic failure as a final common pathway to dementia. Science. 2020;370(6512):50–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Zhou J, Zhang L, Peng J, Zhang X, Zhang F, Wu Y, Huang A, Du F, Liao Y, He Y, et al. Astrocytic lrp1 enables mitochondria transfer to neurons and mitigates brain ischemic stroke by suppressing arf1 lactylation. Cell Metab. 2024;36(9):2054–e206814. [DOI] [PubMed] [Google Scholar]
- 179.Liu Q, Trotter J, Zhang J, Peters MM, Cheng H, Bao J, Han X, Weeber EJ, Bu G. Neuronal lrp1 knockout in adult mice leads to impaired brain lipid metabolism and progressive, age-dependent synapse loss and neurodegeneration. J Neurosci. 2010;30(50):17068–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Liu X, Hao J, Yao E, Cao J, Zheng X, Yao D, Zhang C, Li J, Pan D, Luo X, et al. Polyunsaturated fatty acid supplement alleviates depression-incident cognitive dysfunction by protecting the cerebrovascular and glymphatic systems. Brain Behav Immun. 2020;89:357–70. [DOI] [PubMed] [Google Scholar]
- 181.Wen J, Satyanarayanan SK, Li A, Yan L, Zhao Z, Yuan Q, Su KP, Su H. Unraveling the impact of omega-3 polyunsaturated fatty acids on blood-brain barrier (bbb) integrity and glymphatic function. Brain Behav Immun. 2024;115:335–55. [DOI] [PubMed] [Google Scholar]
- 182.Yuyama K, Sun H, Fujii R, Hemmi I, Ueda K, Igeta Y. Extracellular vesicle proteome unveils cathepsin b connection to alzheimer’s disease pathogenesis. Brain. 2024;147(2):627–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Deleo AM, Ikezu T. Extracellular vesicle biology in alzheimer’s disease and related tauopathy. J Neuroimmune Pharmacol. 2018;13(3):292–308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Visconte C, Fenoglio C, Serpente M, Muti P, Sacconi A, Rigoni M, Arighi A, Borracci V, Arcaro M, Arosio B, et al. Altered extracellular vesicle Mirna profile in prodromal alzheimer’s disease. Int J Mol Sci. 2023;24:19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Ruan Z, Pathak D, Venkatesan KS, Yoshii-Kitahara A, Muraoka S, Bhatt N, Takamatsu-Yukawa K, Hu J, Wang Y, Hersh S, et al. Alzheimer’s disease brain-derived extracellular vesicles spread Tau pathology in interneurons. Brain. 2021;144(1):288–309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Pei J, Palanisamy CP, Jayaraman S, Natarajan PM, Umapathy VR, Roy JR, Thalamati D, Ahalliya RM, Kanniappan GV, Mironescu M. Proteomics profiling of extracellular vesicle for identification of potential biomarkers in alzheimer’s disease: a comprehensive review. Ageing Res Rev. 2024;99:102359. [DOI] [PubMed] [Google Scholar]
- 187.Cai H, Pang Y, Wang Q, Qin W, Wei C, Li Y, Li T, Li F, Wang Q, Li Y, et al. Proteomic profiling of Circulating plasma exosomes reveals novel biomarkers of alzheimer’s disease. Alzheimers Res Ther. 2022;14(1):181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Kumar A, Su Y, Sharma M, Singh S, Kim S, Peavey JJ, Suerken CK, Lockhart SN, Whitlow CT, Craft S, et al. Microrna expression in extracellular vesicles as a novel blood-based biomarker for alzheimer’s disease. Alzheimers Dement. 2023;19(11):4952–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.You Y, Muraoka S, Jedrychowski MP, Hu J, Mcquade AK, Young-Pearse T, Aslebagh R, Shaffer SA, Gygi SP, Blurton-Jones M, et al. Human neural cell type-specific extracellular vesicle proteome defines disease-related molecules associated with activated astrocytes in alzheimer’s disease brain. J Extracell Vesicles. 2022;11(1):e12183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Su H, Rustam YH, Masters CL, Makalic E, Mclean CA, Hill AF, Barnham KJ, Reid GE, Vella LJ. Characterization of brain-derived extracellular vesicle lipids in alzheimer’s disease. J Extracell Vesicles. 2021;10(7):e12089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Xu F, Wu Y, Yang Q, Cheng Y, Xu J, Zhang Y, Dai H, Wang B, Ma Q, Chen Y, et al. Engineered extracellular vesicles with shp2 high expression promote mitophagy for alzheimer’s disease treatment. Adv Mater. 2022;34(49):e2207107. [DOI] [PubMed] [Google Scholar]
- 192.Tcw J, Qian L, Pipalia NH, Chao MJ, Liang SA, Shi Y, Jain BR, Bertelsen SE, Kapoor M, Marcora E, et al. Cholesterol and matrisome pathways dysregulated in astrocytes and microglia. Cell. 2022;185(13):2213–e223325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Sienski G, Narayan P, Bonner JM, Kory N, Boland S, Arczewska AA, Ralvenius WT, Akay L, Lockshin E, He L, et al. Apoe4 disrupts intracellular lipid homeostasis in human ipsc-derived glia. Sci Transl Med. 2021;13:583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Victor MB, Leary N, Luna X, Meharena HS, Scannail AN, Bozzelli PL, Samaan G, Murdock MH, von Maydell D, Effenberger AH, et al. Lipid accumulation induced by apoe4 impairs microglial surveillance of neuronal-network activity. Cell Stem Cell. 2022;29(8):1197–e12128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Mathys H, Davila-Velderrain J, Peng Z, Gao F, Mohammadi S, Young JZ, Menon M, He L, Abdurrob F, Jiang X, et al. Single-cell transcriptomic analysis of alzheimer’s disease. Nature. 2019;570(7761):332–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Yeh FL, Hansen DV, Sheng M. Trem2, microglia, and neurodegenerative diseases. Trends Mol Med. 2017;23(6):512–33. [DOI] [PubMed] [Google Scholar]
- 197.Jonsson T, Stefansson H, Steinberg S, Jonsdottir I, Jonsson PV, Snaedal J, Bjornsson S, Huttenlocher J, Levey AI, Lah JJ, et al. Variant of trem2 associated with the risk of alzheimer’s disease. N Engl J Med. 2013;368(2):107–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Rueda-Carrasco J, Sokolova D, Lee SE, Childs T, Jurčáková N, Crowley G, De Schepper S, Ge JZ, Lachica JI, Toomey CE, et al. Microglia-synapse engulfment via ptdser-trem2 ameliorates neuronal hyperactivity in alzheimer’s disease models. Embo J. 2023;42(19):e113246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Wang S, Mustafa M, Yuede CM, Salazar SV, Kong P, Long H, Ward M, Siddiqui O, Paul R, Gilfillan S et al. Anti-human trem2 induces microglia proliferation and reduces pathology in an alzheimer’s disease model. J Exp Med. 2020;217(9). [DOI] [PMC free article] [PubMed]
- 200.Wang S, Sudan R, Peng V, Zhou Y, Du S, Yuede CM, Lei T, Hou J, Cai Z, Cella M, et al. Trem2 drives microglia response to amyloid-β via syk-dependent and -independent pathways. Cell. 2022;185(22):4153–e416919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Parhizkar S, Gent G, Chen Y, Rensing N, Gratuze M, Strout G, Sviben S, Tycksen E, Zhang Q, Gilmore PE, et al. Sleep deprivation exacerbates microglial reactivity and aβ deposition in a trem2-dependent manner in mice. Sci Transl Med. 2023;15(693):eade6285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.van Lengerich B, Zhan L, Xia D, Chan D, Joy D, Park JI, Tatarakis D, Calvert M, Hummel S, Lianoglou S, et al. A trem2-activating antibody with a blood-brain barrier transport vehicle enhances microglial metabolism in alzheimer’s disease models. Nat Neurosci. 2023;26(3):416–29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Ulland TK, Song WM, Huang SC, Ulrich JD, Sergushichev A, Beatty WL, Loboda AA, Zhou Y, Cairns NJ, Kambal A, et al. Trem2 maintains microglial metabolic fitness in alzheimer’s disease. Cell. 2017;170(4):649–e66313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Zhou Y, Song WM, Andhey PS, Swain A, Levy T, Miller KR, Poliani PL, Cominelli M, Grover S, Gilfillan S, et al. Human and mouse single-nucleus transcriptomics reveal trem2-dependent and trem2-independent cellular responses in alzheimer’s disease. Nat Med. 2020;26(1):131–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Zhao N, Qiao W, Li F, Ren Y, Zheng J, Martens YA, Wang X, Li L, Liu CC, Chen K et al. Elevating microglia trem2 reduces amyloid seeding and suppresses disease-associated microglia. J Exp Med. 2022;219(12). [DOI] [PMC free article] [PubMed]
- 206.Jain N, Lewis CA, Ulrich JD, Holtzman DM. Chronic trem2 activation exacerbates aβ-associated Tau seeding and spreading. J Exp Med. 2023;220(1). [DOI] [PMC free article] [PubMed]
- 207.Zhao Y, Wu X, Li X, Jiang LL, Gui X, Liu Y, Sun Y, Zhu B, Piña-Crespo JC, Zhang M, et al. Trem2 is a receptor for β-amyloid that mediates microglial function. Neuron. 2018;97(5):1023–e10317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Keren-Shaul H, Spinrad A, Weiner A, Matcovitch-Natan O, Dvir-Szternfeld R, Ulland TK, David E, Baruch K, Lara-Astaiso D, Toth B, et al. A unique microglia type associated with restricting development of alzheimer’s disease. Cell. 2017;169(7):1276–e129017. [DOI] [PubMed] [Google Scholar]
- 209.Zhu B, Liu Y, Hwang S, Archuleta K, Huang H, Campos A, Murad R, Piña-Crespo J, Xu H, Huang TY. Trem2 deletion enhances Tau dispersion and pathology through microglia exosomes. Mol Neurodegener. 2022;17(1):58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Zhong L, Sheng X, Wang W, Li Y, Zhuo R, Wang K, Zhang L, Hu DD, Hong Y, Chen L, et al. Trem2 receptor protects against complement-mediated synaptic loss by binding to complement c1q during neurodegeneration. Immunity. 2023;56(8):1794–e18088. [DOI] [PubMed] [Google Scholar]
- 211.Wang Y, Cella M, Mallinson K, Ulrich JD, Young KL, Robinette ML, Gilfillan S, Krishnan GM, Sudhakar S, Zinselmeyer BH, et al. Trem2 lipid sensing sustains the microglial response in an alzheimer’s disease model. Cell. 2015;160(6):1061–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Nugent AA, Lin K, van Lengerich B, Lianoglou S, Przybyla L, Davis SS, Llapashtica C, Wang J, Kim DJ, Xia D, et al. Trem2 regulates microglial cholesterol metabolism upon chronic phagocytic challenge. Neuron. 2020;105(5):837–e8549. [DOI] [PubMed] [Google Scholar]
- 213.Filipello F, You SF, Mirfakhar FS, Mahali S, Bollman B, Acquarone M, Korvatska O, Marsh JA, Sivaraman A, Martinez R, et al. Defects in lysosomal function and lipid metabolism in human microglia harboring a trem2 loss of function mutation. Acta Neuropathol. 2023;145(6):749–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Li RY, Qin Q, Yang HC, Wang YY, Mi YX, Yin YS, Wang M, Yu CJ, Tang Y. Trem2 in the pathogenesis of ad: a lipid metabolism regulator and potential metabolic therapeutic target. Mol Neurodegener. 2022;17(1):40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Blumenfeld J, Yip O, Kim MJ, Huang Y. Cell type-specific roles of apoe4 in alzheimer disease. Nat Rev Neurosci. 2024;25(2):91–110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Lee S, Devanney NA, Golden LR, Smith CT, Schwartz JL, Walsh AE, Clarke HA, Goulding DS, Allenger EJ, Morillo-Segovia G, et al. Apoe modulates microglial immunometabolism in response to age, amyloid pathology, and inflammatory challenge. Cell Rep. 2023;42(3):112196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Wang N, Wang M, Jeevaratnam S, Rosenberg C, Ikezu TC, Shue F, Doss SV, Alnobani A, Martens YA, Wren M, et al. Opposing effects of apoe2 and apoe4 on microglial activation and lipid metabolism in response to demyelination. Mol Neurodegener. 2022;17(1):75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Haney MS, Pálovics R, Munson CN, Long C, Johansson PK, Yip O, Dong W, Rawat E, West E, Schlachetzki J, et al. Apoe4/4 is linked to damaging lipid droplets in alzheimer’s disease microglia. Nature. 2024;628(8006):154–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Lu J, Wu K, Sha X, Lin J, Chen H, Yu Z. Trpv1 alleviates apoe4-dependent microglial antigen presentation and t cell infiltration in alzheimer’s disease. Transl Neurodegener. 2024;13(1):52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Liu CC, Wang N, Chen Y, Inoue Y, Shue F, Ren Y, Wang M, Qiao W, Ikezu TC, Li Z, et al. Cell-autonomous effects of apoe4 in restricting microglial response in brain homeostasis and alzheimer’s disease. Nat Immunol. 2023;24(11):1854–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Sepulveda J, Kim JY, Binder J, Vicini S, Rebeck GW. Apoe4 genotype and aging impair injury-induced microglial behavior in brain slices, including toward aβ, through p2ry12. Mol Neurodegener. 2024;19(1):24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Lin YT, Seo J, Gao F, Feldman HM, Wen HL, Penney J, Cam HP, Gjoneska E, Raja WK, Cheng J, et al. Apoe4 causes widespread molecular and cellular alterations associated with alzheimer’s disease phenotypes in human ipsc-derived brain cell types. Neuron. 2018;98(6):1141–e11547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Wang C, Xiong M, Gratuze M, Bao X, Shi Y, Andhey PS, Manis M, Schroeder C, Yin Z, Madore C, et al. Selective removal of astrocytic apoe4 strongly protects against tau-mediated neurodegeneration and decreases synaptic phagocytosis by microglia. Neuron. 2021;109(10):1657–e16747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Blanchard JW, Akay LA, Davila-Velderrain J, von Maydell D, Mathys H, Davidson SM, Effenberger A, Chen CY, Maner-Smith K, Hajjar I, et al. Apoe4 impairs myelination via cholesterol dysregulation in oligodendrocytes. Nature. 2022;611(7937):769–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Grubman A, Choo XY, Chew G, Ouyang JF, Sun G, Croft NP, Rossello FJ, Simmons R, Buckberry S, Landin DV, et al. Transcriptional signature in microglia associated with aβ plaque phagocytosis. Nat Commun. 2021;12(1):3015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Ma WW, Li CQ, Zhao L, Wang YS, Xiao R. Nf-κb-mediated inflammatory damage is differentially affected in sh-sy5y and c6 cells treated with 27-hydroxycholesterol. Food Sci Nutr. 2019;7(5):1685–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Titus C, Hoque MT, Bendayan R. Ppar agonists for the treatment of neuroinflammatory diseases. Trends Pharmacol Sci. 2024;45(1):9–23. [DOI] [PubMed] [Google Scholar]
- 228.Jamwal S, Blackburn JK, Elsworth JD. Pparγ/pgc1α signaling as a potential therapeutic target for mitochondrial biogenesis in neurodegenerative disorders. Pharmacol Ther. 2021;219:107705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Semple RK, Chatterjee VK, O’Rahilly S. Ppar gamma and human metabolic disease. J Clin Invest. 2006;116(3):581–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Wójtowicz S, Strosznajder AK, Jeżyna M, Strosznajder JB. The novel role of Ppar alpha in the brain: promising target in therapy of alzheimer’s disease and other neurodegenerative disorders. Neurochem Res. 2020;45(5):972–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231.Thakur S, Dhapola R, Sarma P, Medhi B, Reddy DH. Neuroinflammation in alzheimer’s disease: current progress in molecular signaling and therapeutics. Inflammation. 2023;46(1):1–17. [DOI] [PubMed] [Google Scholar]
- 232.Luo R, Su LY, Li G, Yang J, Liu Q, Yang LX, Zhang DF, Zhou H, Xu M, Fan Y, et al. Activation of ppara-mediated autophagy reduces alzheimer disease-like pathology and cognitive decline in a murine model. Autophagy. 2020;16(1):52–69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Bevilacqua A, Franco F, Lu YT, Rahiman N, Kao KC, Chuang YM, Zhu Y, Held W, Xie X, Gunsalus KC, et al. Pparβ/δ-orchestrated metabolic reprogramming supports the formation and maintenance of memory cd8(+) t cells. Sci Immunol. 2024;9(98):eadn2717. [DOI] [PubMed] [Google Scholar]
- 234.Cao D, Khan Z, Li X, Saito S, Bernstein EA, Victor AR, Ahmed F, Hoshi AO, Veiras LC, Shibata T, et al. Macrophage angiotensin-converting enzyme reduces atherosclerosis by increasing peroxisome proliferator-activated receptor α and fundamentally changing lipid metabolism. Cardiovasc Res. 2023;119(9):1825–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No datasets were generated or analysed during the current study.