Abstract
With the aging process of the global population and the development of medical technology, the cases of postoperative cognitive dysfunction (POCD) are also increasing. Due to the complexity of the pathogenesis, urgent treatment has been sought. Neuroinflammation induced by the accumulation of lipid droplets (LDs) in microglia has been closely watched in recent years and is also considered to be an important cause of nerve damage. Our study found that derived from Withania somnifera, Withaferin A (WA) could reduce the accumulation of LDs in the hippocampus of POCD mice, inhibit the expression of inflammatory factor interleukin-1β (IL-1β), and improve the cognitive ability of mice. Further in vitro experimental studies showed that WA increased the autophagy level of microglia, promoted the degradation of LDs, and reduced the production of inflammatory factors. In this regard, our comprehensive research endeavor holds the potential to furnish novel insights into therapeutic strategies aimed at addressing POCD and its associated neural impairments.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-024-75284-6.
Keywords: Withaferin A, LDs, Neuroinflammation, POCD
Subject terms: Cell biology, Neuroscience
Introduction
Postoperative cognitive dysfunction (POCD) stands as a significant concern within the realm of surgical interventions, encompassing a transient but often noticeable decline in cognitive function following surgery1–3. This phenomenon, characterized by deficits in memory, attention, and executive functions, has garnered attention due to its potential impact on patients’ quality of life and long-term outcomes. Although the exact etiology of POCD remains multifaceted and not entirely elucidated, factors such as anesthesia, inflammatory response, and underlying comorbidity are believed to contribute to its development4,5. A comprehensive understanding of the mechanisms underlying POCD holds the promise of advancing perioperative care strategies and improving patient recovery trajectories.
LDs accumulation, a pivotal aspect of cellular metabolism, have gained increasing recognition for its relevance in the context of neurological health. Neuroinflammation and its intricate interplay with glial cells further underscore the significance of LDs in maintaining neural homeostasis6,7. LDs, once perceived solely as passive reservoirs of energy, have emerged as dynamic organelles intricately linked to cellular function8,9. In neurons, LDs participate in regulating lipid metabolism, trafficking, and membrane composition, thus impacting synaptic plasticity and neuronal signaling10,11. In addition, metabolically active glial cells, including astrocytes and microglia, profoundly influence neuronal health and function. Accumulating evidence suggests that glial cells modulate LDs dynamics in response to metabolic demands and inflammatory cues12–14. Actually, glial cells are particularly sensitive to lipotoxicity. If accumulated LDs are not removed in time, the peroxidation of LDs will be accelerated, causing nerve damage. Excessive LDs content may trigger non-oxidative metabolic pathways and cytotoxic responses12,15. Moreover, the bidirectional interaction between glial cells and LDs extends to neuroinflammation. glial cells, particularly microglia, can undergo phenotypic changes upon exposure to lipid-rich environments, contributing to neuroinflammatory response observed in various neurological disorders12. Neuroinflammation, a hallmark of conditions like neurodegenerative diseases and traumatic brain injuries, can trigger lipid droplet accumulation in glial cells5,16,17. This phenomenon, often accompanied by altered lipid metabolism, serves as a potential mediator of neuroinflammatory processes. The crosstalk between LDs, glial cells, and neuroinflammation highlights their intertwined roles in shaping the neurobiological landscape.
Derived from Withania somnifera, WA is a steroidal lactone that exhibits considerable potential in counteracting neuroinflammation and addressing instances of POCD. Its appealing attributes encompass noteworthy lipid solubility, enabling efficient penetration of the blood-brain barrier, thus positioning it as an attractive candidate for neuroprotection18,19. By virtue of its anti-inflammatory and antioxidant attributes, WA emerges as a prime contender in the battle against neuroinflammation, a pivotal factor in diverse neurological disorders19,20. Moreover, pertinence of WA extends to the domain of postsurgical cognitive impairments, potentially offering a therapeutic pathway for mitigating POCD. The manifold advantages of WA in contexts of neuroinflammation and cognitive dysfunction underscore its significance as a fresh agent carrying extensive implications for safeguarding neurological health and cognition, for example in the study of parkinson disease21. Delving deeper into mechanisms of WA and its potential applications holds the potential to advance interventions targeting conditions linked to neuroinflammation and surgery-induced cognitive deficits.
In our study, we have validated the efficacy of WA in enhancing cognitive levels in POCD mice. This enhancement is coupled with a reduction in neuroinflammation caused by LDs accumulation on microglia, and an elevation in autophagy levels within the hippocampal region of the mice. Furthermore, subsequent ex vivo experiments have provided additional evidence, demonstrating that WA can mitigate LDs aggregation induced by lipopolysaccharide (LPS) and the release of interleukin-1β (IL-1β) inflammatory cytokines. The underlying mechanism for these effects involves WA’s ability to heighten microglia cell autophagy levels, expediting the degradation of LDs molecules and thereby mitigating the inflammatory response associated with LDs accumulation. Consequently, our findings contribute to a deeper understanding of the potential therapeutic implications of WA for ameliorating cognitive dysfunction and neuroinflammation linked to POCD.
Methods and materials
Animals
Male C57BL/6J mice at the age of 12 months were acquired from GemPharmatech (China, Nanjing) and housed at the Animal Resource Center of Nanchang University. The mice were maintained under controlled conditions with ad libitum access to food and water. The ambient temperature was maintained at 22 °C ± 2 °C, and a 12-hour light-dark cycle was upheld, with a humidity level of 60%. All animal procedures strictly adhered to ARVO and ARRIVE guidelines for the Use of Animals in Research, as well as the guidelines established by the Institutional Animal Care and Use Committee of Nanchang University (Approval No.NCULAE-20221031176).
Surgical model and WA adminstration
After one-week acclimatization, mice were subjected to an intramedullary fixation surgery for tibial fracture under isoflurane anesthesia. Briefly, mice were fasted for 12 h before surgery, then anesthetized with 1.8% isoflurane and oxygen at 2 L/min in the small animal anesthesia machine (R500, RWD Life Science). A skin incision was made below the knee to expose the tibia, and a 0.3 mm pin was inserted into its medullary cavity for intramedullary fixation. Next, the bone was fractured at the midpoint with a surgical scissor. Lastly, the wound was sutured after necessary debridement. 2% lidocaine solution was applied locally before the incision, and 1% tetracaine hydrochloride mucilage was applied to the wound twice daily to treat the pain. The administration of WA drugs (Selleck, S8587) is divided into three doses, low (1 mg/kg), medium (2 mg/kg) and high (4 mg/kg), intraperitoneal injection, three times a week, for 2 months (The animal study protocol was approved by Institutional Animal Care and Use Committee of Nanchang University (Approval No.NCULAE-20221031176)).
Immunofluorescence staining and BODIPY staining
Following anesthesia with pentobarbital sodium, post-experimental mice were perfused with physiological saline, followed by perfusion with 4% paraformaldehyde. Subsequent to whole brain extraction, the mice brains underwent dehydration in 20% and 30% sucrose solutions and were subsequently frozen for sectioning. Slices of 25 μm thickness were rinsed three times in phosphate-buffered saline (PBS) and then incubated with a 5% solution of blocking agent serum (bovine serum albumin (BSA)-PBS-Triton(PBST)) at room temperature for one hour. Following removal of the blocking agent solution, primary antibodies (GFAP: Abcam, ab7260, 1:500; IL-1β: Santa, sc-515598, 1:300; IBA-1: Wako, 019-19741,1:500 ; MAP1LC3B: Santa, sc-398822, 1:200; SQSTM1: Santa, sc-48402, 1:200) were applied and incubated overnight at 4 °C. The following day, brain slices were washed and exposed to fluorescent secondary antibodies (1:1000, Invitrogen). Hoechst solution (Sigma, B2261) was employed to label cellular nuclei within the brain slices.
BODIPY staining is employed to label LDs in cells based on the lipophilic properties of BODIPY dyes. These dyes are highly fluorescent and have an affinity for neutral lipids, which are abundant in LDs. When BODIPY dye is applied to cells, it selectively incorporates into the LDs due to its hydrophobic nature, making the LDs distinctly fluorescent under fluorescence microscopy. This selective staining allows for the visualization and quantitative analysis of LDs within various cell types. For the labeling of LDs in brain slices using BODIPY (D3861, Invitrogen), brain slices were incubated in a BODIPY solution for 15 min, followed by PBS rinsing. Finally, the brain slices were mounted onto slides and observed using a confocal microscope (Zeiss, Germany).
Oil red O staining of brain slices
Oil Red O staining is a method widely used for detecting lipids, particularly neutral triglycerides and lipoproteins, in tissue sections and cell cultures. The principle behind this staining technique lies in the solubility characteristics of the Oil Red O dye, which is a lysochrome (fat -soluble dye). When applied to a sample, Oil Red O selectively binds to lipid components due to its lipophilic nature. Therefore, it can be used to detect intracellular lipid content. Sliced brain sections of 25 μm thickness were immersed in PBS solution and subjected to three washes. They were then treated with 60% isopropanol for 20–30 s. Subsequently, the brain sections were immersed in an Oil Red staining solution (Sigma, MAK194) and allowed to undergo closed staining for 10–15 min. Following completion of staining, brain sections were placed in 60% isopropanol for a brief wash to remove excess stain. Optionally, differentiation with a 1% hydrochloric acid solution could be performed, followed by thorough rinsing with distilled water. After blotting away excess moisture from the brain sections using filter paper, they were mounted using glycerol gelatin and observed under a microscope (Olympus, Tokyo, Japan).
Immunohistochemical staining
Sliced brain sections with a thickness of 25 μm were rinsed three times in PBS. To quench endogenous peroxidase activity, the brain sections were incubated in a 3% hydrogen peroxide solution at room temperature in the dark for 10 min. After removal of the hydrogen peroxide solution, the brain sections were washed three times with PBS and then blocked with PBST solution containing 5% BSA at room temperature for 1 h. The blocking solution was discarded, and primary antibody (Rabbit anti-IBA-1 antibody, 1:500, 019-19741, Wako) was added overnight at 4 °C. On the following day, the primary antibody was removed, and secondary antibody (1:1000, Invitrogen) was applied, followed by incubation at room temperature in the dark for 1 h. After removal of the secondary antibody, brain sections were washed three times with PBS again. Freshly prepared substrate buffer containing diaminobenzidine (DAB) (Wuhan Boshide) was added for light-avoiding color development. The sections were then mounted and allowed to air-dry in a ventilated area. Subsequently, a dehydration process was carried out (70% ethanol for 30 s; 80% ethanol for 30 s; 95% ethanol for 60 s; 100% ethanol I for 60 s; 100% ethanol II for 60 s; xylene I for 120 s; xylene II for 300 s). Mounting was done using a mounting medium, and the sections were observed and captured using a stereomicroscope (Olympus, Tokyo, Japan).
Behavioral test
Y-maze
The Y-maze consisted of three rectangular arms, each measuring 34 cm in length, 10 cm in width, and 10 cm in height. These arms were designated as the Start Arm, Novel Arm, and Other Arm. Prior to the initiation of experiments, the experimental animals were acclimated to the testing environment for 24 h. At the onset of testing, the Novel Arm was blocked off by a partition, and the mice were placed into the Start Arm. Following a 5-minute period of free exploration in both the Start and Other Arms, the movement trajectories were recorded using the CLEVER-SYM system software. After a one-hour interval, the mice were reintroduced into the Start Arm, and the Novel Arm was opened for a 5-minute exploration period. During this time, the movement trajectories of mice were once again recorded. The time spent exploring the Novel Arm was divided by the total exploration time to assess the short-term memory function of mice.
Novel object recognition test
In an open field arena, objects labeled as A and B (identical in nature) were placed. The experimental mice were introduced into the open field for a duration of 5 min. During this period, their interactions with both objects A and B were observed and recorded, noting both the frequency and duration of contact. Subsequently, object B was replaced with a distinct object labeled as C (different from B). The mice’s interactions with the new object were observed and recorded for another 5-minute interval. A higher number of interactions and longer duration of contact with the new object indicate a stronger ability of the mice to explore novel stimuli. Additionally, the recognition index can be employed as an evaluative metric. This index is calculated as follows: Novel Object Recognition Time / (Novel Object Recognition Time + Familiar Object Recognition Time) × 100%. This index serves to quantify the proportion of time spent recognizing the novel object relative to the total time spent recognizing both the novel and familiar objects.
Culturing and treatment of primary microglia
Newborn mice aged 1–3 days were utilized for the isolation of primary microglia. The meninges were carefully removed using scissors and ophthalmic forceps, following which the whole brain tissue was extracted. After digestion with pancreatic enzyme, the digestion process was terminated using complete culture medium containing 10% fetal bovine serum (FBS). The digested mixture was then passed through a 40 μm cell strainer, and centrifuged at 1000 g for 10 min. After discarding the supernatant, the tissue pellet at the bottom of the centrifuge tube was resuspended and plated in T-75 flasks. The medium was replaced every three days, and after approximately 12 days, microglia that floated above the mixed glial cell layer could be aspirated for experimental use. Prior to treatment, the cells were pretreated with WA for 1 h. Subsequently, microglia were stimulated with LPS (Sigma-Aldrich, L2630 ) to induce an inflammatory response, thus establishing a cellular model of inflammation.
Cell BODIPY staining
Primary microglia were cultured on 14 mm cell culture dishes. BODIPY staining was performed as follows: a staining solution of BODIPY (2.0 mM, Invitrogen, D3861) was added to the prepared cells and incubated in a cell culture incubator at 37 °C for 15 min. Subsequently, the cells were removed from the incubator. The cells were treated with 4% paraformaldehyde for 20 min. After rinsing three times with PBS to remove residual paraformaldehyde, Hoechst stain was applied to the cells for nuclear labeling of primary astrocytes and incubated for 15 min. Following this, the cells were washed three times with PBS to remove excess Hoechst stain while avoiding exposure to light. Finally, the cell culture dishes were placed under a confocal microscope (Zeiss, Germany) for image acquisition. This comprehensive staining procedure allowed for the visualization of primary microglia, specifically highlighting their nuclei through Hoechst staining, in conjunction with BODIPY staining for cellular visualization.
Cell oil red O staining
The Oil Red O staining procedure for cells cultured in a 24-well plate involved initial steps such as removing the cell culture medium and washing the cells with PBS. Subsequent fixation was achieved using Oil Red fixative solution for a duration of 20–30 min, followed by discarding the fixative and rinsing with distilled water. Permeabilization was facilitated through a brief exposure to isopropanol, and subsequent staining was performed using a freshly prepared Oil Red staining solution, allowing for an incubation period of 10–20 min. The staining solution was removed, and excess stain was eliminated by rinsing with 60% isopropanol. To counterstain the nuclei, Hematoxylin stain was applied for 1–2 min, and comprehensive water washing was conducted to achieve desired contrast. The cells were then covered with distilled water and examined under a microscope, providing insights into the lipid content within the cells.
Cell immunofluorescence staining
The cell culture medium within the 24-well plate was discarded, and subsequent to two washes with PBS, the cells were fixed using 4% paraformaldehyde for 15 min. Following the removal of paraformaldehyde, the cells were subjected to two additional PBS washes. Subsequently, the cells were incubated for one hour with a solution consisting of 5% BSA-PBST, effectively blocking non-specific binding sites. After this incubation period, the blocking solution was removed. The primary antibody (IL-1β: Santa, sc-515598, 1:300) was introduced and allowed to incubate overnight at 4 °C. On the subsequent day, a fluorescent secondary antibody (1:1000, Invitrogen) was applied and incubated at room temperature for one hour under light-avoidant conditions. Finally, the nuclei were labeled using Hoechst solution, and the cell culture dishes were positioned on glass slides for observation under a confocal microscope (Zeiss, Germany).
Transfection of GFP-RFP autophagy dual-fluorescence virus
Primary microglia were seeded into a 24-well plate with cell coverslips, and autophagy dual-fluorescence virus (HanBio, HB-AP2100001) was diluted in a culture medium containing 1% FBS-F12/DMEM. The virus was added to the wells of the 24-well plate. After an 8-hour incubation period, the viral solution was aspirated, and the cells were replenished with culture medium containing 10% FBS-F12/DMEM for an additional 48-hour incubation. Subsequent to the incubation period, a pre-treatment with WA was administered for 1 h, followed by a 12-hour stimulation with LPS. Ultimately, the cells were stained using Hoechst to label the cell nuclei, enabling subsequent microscopic observation of the cellular state.
Western blot analysis
The cells were treated with RIPA lysis buffer containing a phosphatase inhibitor (Halt Protease Inhibitor Single-Use Cocktail, Thermo Fisher Scientific). Cell proteins were extracted using cell scrapers, and the resulting lysates were centrifuged to isolate cellular supernatants. Protein concentration was determined using the BCA assay on 1 µL of extracted protein. Subsequently, 1/4 of the determined protein volume was mixed with 5X loading buffer and boiled at 95 °C for 5 min. The resultant protein samples were stored at -20 °C. Approximately 40 µg of the protein samples were subjected to SDS-polyacrylamide gel electrophoresis, employing the following parameters: 80 V for 40 min and 120 V for 60 min. The separated SDS-polyacrylamide gel was subsequently wet-transferred at a constant current of 200 mA onto a PVDF membrane (Millipore, USA). Post transfer, the entire PVDF membrane was immersed in a solution of TBST (pH 7.4, containing 10 mM Tris-HCl, 150 mM NaCl, 0.1% Tween-20) containing 5% skim milk for 1 h. After gently removing the membrane from the milk solution, it was rinsed with TBST to eliminate milk residues. For primary antibody incubation, the membrane was subjected to an overnight incubation at 4 °C (MAP1LC3B: Cell Signaling Technology, 2775, 1:1000; SQSTM1: Santa, sc-48402, 1:500; GAPDH antibody: Cell Signaling Technology, 5174, 1:3000). On the subsequent day, the primary antibody was discarded, and the PVDF membrane was washed with TBST three times for 10 min each at room temperature. The membrane was then incubated with the secondary antibody for 1 h at room temperature. Following the secondary antibody incubation, the membrane was washed again with TBST. Finally, the membrane was subjected to ECL chemiluminescent substrate for visualization, and the protein blot images were captured using a developer solution. Image analysis and processing were performed using Image J software. The grayscale intensity of the target protein bands was semi-quantitatively analyzed by comparing them to the reference GAPDH grayscale values. This Western blot methodology enables the assessment of protein expression levels in the prepared primary microglia samples.
Results
Microglia-mediated neuroinflammation and LDs accumulation in the hippocampus of mice with postoperative cognitive impairment
This study investigated the inflammatory response and LDs accumulation in hippocampal glial cells as a consequence of surgical trauma. Numerous studies have demonstrated the pivotal role of glial cells in executing inflammatory responses within the brain22,23. Dysregulation of glial cell homeostasis has been associated with exacerbation of pathological inflammation and disease progression24,25. POCD shares pathogenic mechanisms with most neurodegenerative diseases, involving neuroinflammatory responses mediated by glial cells. Additionally, research indicated a significant elevation in serum lipid and inflammation factor levels in elderly patients after surgery, suggesting that abnormal lipid metabolism may contribute to postoperative inflammation. Given that astrocytes and microglia are key players in postoperative cognitive impairment, we conducted staining of IL-1β expression on astrocytes and microglia in the dentate gyrus (DG) region of the hippocampus in mice subjected to surgery and anesthesia. Our findings suggested that the inflammatory response primarily originated from microglia rather than astrocytes (Fig. 1A-D), implying that microglia may initiate a cascade of inflammatory events, resulting in the release of inflammatory factors damaging the surrounding neurons. Furthermore, analysis of LDs on microglia revealed that surgical and anesthetic stress induces lipid deposition in the hippocampal region, manifested by increased fluorescence intensity in BODIPY staining and an augmented number of LDs (Fig. 1E-G). This phenomenon could potentially exacerbate inflammation and contribute to cognitive impairment. In summary, our experimental evidence confirmed that anesthesia and surgery induce an inflammatory response in microglia cells along with LDs accumulation, emphasizing their roles in postoperative neuroinflammation.
Fig. 1.
Microglia-mediated neuroinflammation and LDs accumulation in the hippocampus of mice with postoperative cognitive impairment. (A) Co-labeling of astrocytic glial fibrillary acidic protein (GFAP) and inflammatory cytokine IL-1β in the hippocampal region of mice (scale bar = 40–20 μm); quantification of co-labeled cell count in (B) (n = 3). (C Co-labeling of microglia marker IBA-1 and inflammatory cytokine IL-1β in the hippocampal region of mice (scale bar = 40–20 μm); quantification of co-labeled cell count in (D) (n = 3). (E Immunofluorescence staining of hippocampal microglia cells, with BODIPY probe marking LDs in the hippocampal area (scale bar = 10 μm). Fluorescence intensity of BODIPY in (F) and quantification of labeled LDs in (G) (n = 3). *P < 0.05, **P < 0.01, ***P < 0.001 compared with the corresponding group, as determined by t test.
WA could reduce the accumulation of LDs in the hippocampus of mice with postoperative cognitive impairment
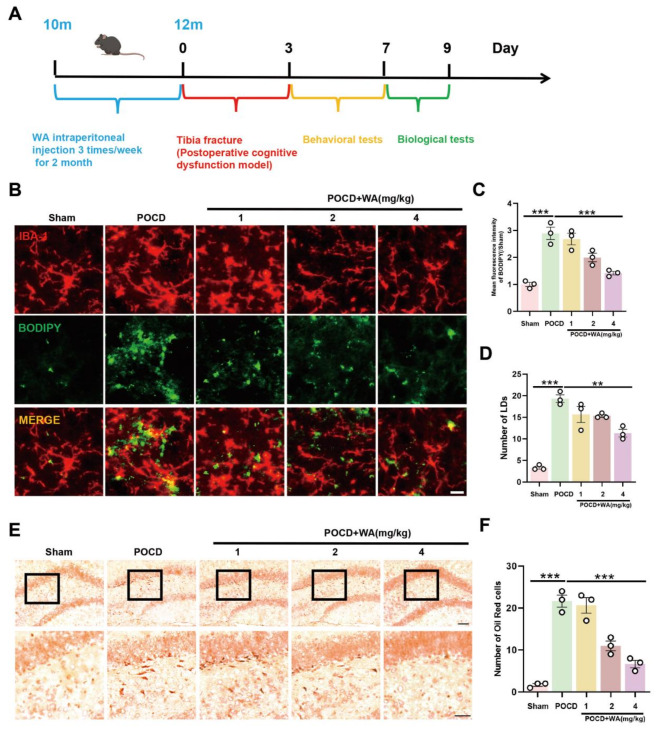
WA, a steroidal lactone with high lipid solubility, isolated from Withania somnifera, possesses the ability to cross the blood-brain barrier. Characterized by its robust neuroprotective properties, including antioxidant and anti-inflammatory effects, WA presents an intriguing candidate for assessing its potential as a therapeutic agent for POCD. In this study, we administered WA to 10-month-old mice at three different concentrations (1 /2 /4 mg/kg) (Fig. 2A) according to previous studies21. Initially, we examined the deposition of LDs in the hippocampal region of the mice. By utilizing BODIPY labeling to visualize LDs, we observed that WA administration led to reduced LDs accumulation in microglia cells, evidenced by diminished BODIPY fluorescence intensity and decreased droplet count (Fig. 2B-D). Subsequently, for a comprehensive evaluation of LDs accumulation in the hippocampal region, we employed oil red staining, a lipophilic dye widely used to identify triglycerides and lipoproteins in tissues and cells (Fig. 2E-F). Our findings revealed an increased deposition of LDs in the hippocampal region following surgical anesthesia, while treatment with 4 mg/kg of WA significantly suppressed this accumulation. Consequently, these results further validated that WA could mitigate LDs deposition, thereby exerting neuroprotective effects. The observed reduction in LDs accumulation substantiates the potential of WA as a therapeutic approach to alleviate postoperative cognitive impairment.
Fig. 2.
WA could reduce the accumulation of LDs in the hippocampus of mice with postoperative cognitive impairment. (A) Experimental schematic. Ten-month-old mice were administered WA at doses of 1, 2, and 4 mg/kg, three times per week, for a duration of two months. Subsequently, a tibial fracture model was established to induce postoperative cognitive impairment in mice. Behavioral and biological assessments were conducted thereafter. (B) Fluorescence staining of hippocampal microglia cells and detection of LDs accumulation using BODIPY probe (scale bar = 10 μm, n = 3). Average fluorescence intensity of BODIPY-labeled LDs and droplet count depicted in (C) and (D), respectively. Hippocampal area Oil Red staining (scale bar = 100–50 μm, n = 3), with statistical representation shown in (E, F). *P < 0.05, **P < 0.01, ***P < 0.001 compared with the corresponding group, as determined by the one-way ANOVA .
WA mitigated microglia inflammation and improved cognitive ability in mice
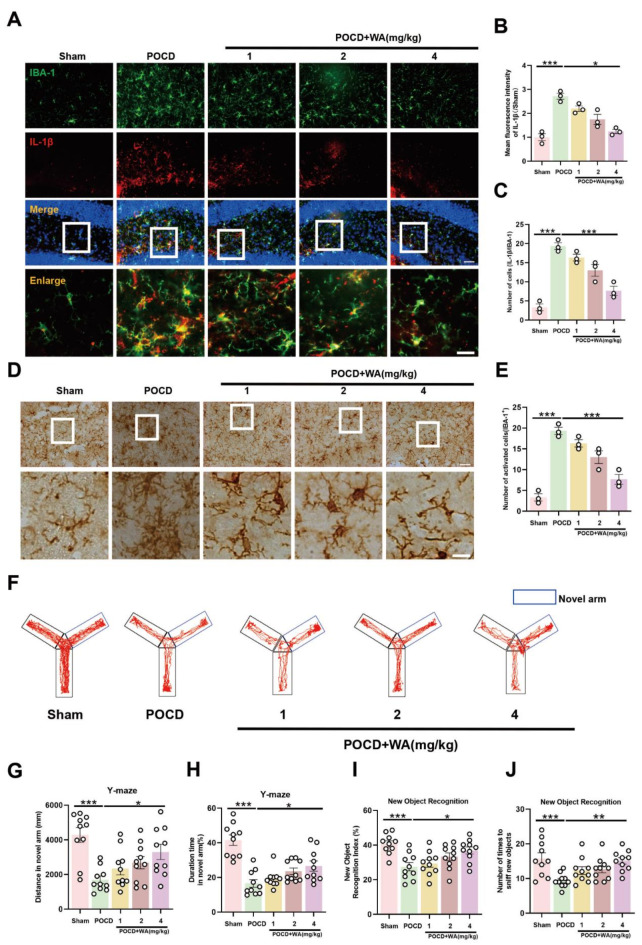
Subsequently, to ascertain the anti-inflammatory effects of WA, we examined the expression of the pro-inflammatory cytokine IL-1β on microglia cells in the hippocampal region of mice. Postoperative anesthesia led to an elevated expression of IL-1β on microglia cells, while WA exhibited a dose-dependent reduction in IL-1β expression (Fig. 3A-C). Further immunohistochemical experiments revealed that microglia in the hippocampal region of POCD mice exhibited an activated state. Pre-administration of WA significantly attenuated this activation, leading to a decrease in the number of activated microglia (Fig. 3D-E). Moving forward, we investigated the impact of WA on mice cognitive memory using the Y-maze and novel object recognition tests to assess short-term memory. In the Y-maze experiment, 4 mg/kg of WA notably improved the cognitive function of POCD mice, evidenced by increased time and frequency of exploration in the novel arm (Fig. 3F-H). Similarly, in the novel object recognition test, mice pre-treated with 4 mg/kg WA exhibited an increased number of interactions with the novel object and prolonged recognition time, indicating an enhanced recognition index (Fig. 3I-J). Consequently, the aforementioned experiments substantiate that WA could alleviate postoperative inflammation mediated by microglia cells and enhance cognitive abilities of mice.
Fig. 3.
WA mitigated microglia inflammation and improved cognitive ability in mice. (A) Co-labeling of microglia marker IBA-1 and inflammatory cytokine IL-1β in the hippocampal region (scale bar = 40–20 μm). Relative fluorescence intensity of IL-1β and co-labeled cell count shown in (B) and (C), respectively (n = 3). (D Immunohistochemical staining of hippocampal IBA-1, indicating activated microglia cell count shown in (E) (scale bar = 40–20 μm, n = 3). (F Movement trajectory of mice in the Y-maze experiment, with blue representing the Novel arm. (G-H) Mouse movement paths within the Novel arm of the Y-maze and number of explorations of the Novel arm (n = 10). (I) Recognition index of mice in the novel object recognition test (n = 10). (J Number of instances of mice recognizing the novel object in the test. *P < 0.05, **P < 0.01, ***P < 0.001 compared with the corresponding group, as determined by the one-way ANOVA .
WA decreased LDs accumulation and inflammatory response induced by LPS
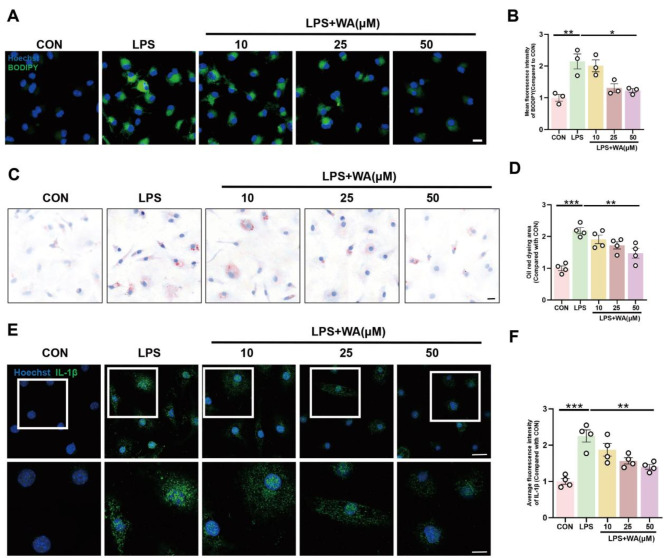
In order to comprehensively investigated the precise mechanisms underlying actions of WA, we conducted ex vivo primary microglia cultures. Building upon prior research and reports, we established an in vitro inflammation model using LPS. Employing BODIPY as a fluorescent probe, we discovered that LPS induced intracellular LDs accumulation in microglia. Pre-treatment with WA effectively mitigated the LPS-induced LDs aggregation in primary microglia, evidenced by reduced BODIPY fluorescence intensity (Fig. 4A-B). Additionally, in cellular Oil Red staining experiments, WA demonstrated the capability to diminish the stained area, thereby inhibiting LPS-induced LDs accumulation (Fig. 4C-D). Lastly, aiming to comprehend and validate the impact of LPS-induced LDs accumulation on intracellular inflammation, we assessed the expression of the inflammatory cytokine IL-1β in primary microglia (Fig. 4E-F). Aligning with the aforementioned in vivo results, WA exhibited a dose-dependent reduction in IL-1β production, further reinforcing its anti-inflammatory effects.
Fig. 4.
WA decreased LDs accumulation and inflammatory response induced by LPS. Microglia were pretreated with WA (10, 25, 50 µM) for 1 h. Subsequently, microglia were stimulated with LPS for 12 h to induce an inflammatory response. (A) Primary microglia were labeled with BODIPY fuorescence probe to visualize LDs molecules (scale bar = 5 μm). (B) Average fluorescence intensity of labeled LDs was quantified (Three independent replicate experiments were performed). (C) Primary microglia were subjected to Oil Red staining to assess LDs accumulation (scale bar = 5 μm). (D) Average area covered by labeled LDs on cells was determined (Four independent replicate experiments were performed). (E) Immunofluorescence staining of IL-1β was performed on primary microglia (scale bar = 10–5 μm), and the average fluorescence intensity of IL-1β is shown in (F) (Four independent replicate experiments were performed). *P < 0.05, **P < 0.01, ***P < 0.001 compared with the corresponding group, as determined by the one-way ANOVA .
WA promoted microglia autophagy and accelerated the degradation of LDs
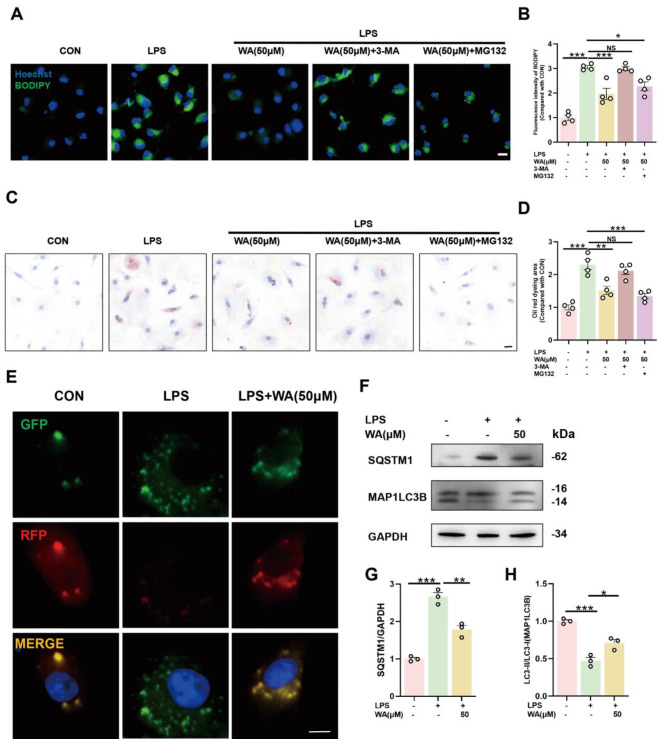
Noteworthy literature has emphasized the significance of accelerating LDs metabolism and degradation as a pivotal strategy to attenuate inflammatory processes14. Given neuroprotective properties of WA encompassing anti-inflammatory and antioxidant effects, we conjectured whether WA could influence the expedited breakdown of LDs to mitigate accumulation. Considering the two major intracellular degradation systems, the ubiquitin-proteasome system and the autophagy-lysosome pathway, we employed inhibitors (Consistent with the doses used in our previous studies) to obstruct these degradation processes within cells17,26. Surprisingly, inhibition of the ubiquitin-proteasome system with MG132 did not impede facilitation of lipid degradation; WA continued to weaken BODIPY fluorescence intensity and reduce Oil Red staining area (Fig. 5A-D). However, upon utilizing 3-methyladenine (3-MA) to suppress cellular autophagy, the action of WA was indeed obstructed (Fig. 5A-D). This led us to hypothesize that WA potentially promoted LDs degradation through the autophagy pathway. To validate this hypothesis, we employed a GFP-RFP dual-fluorescence autophagy virus to observe intracellular autophagic flux in microglia. In the advanced stages of autophagy, lysosomes merge with autophagosomes to form autophagolysosomes, resulting in a decrease in intracellular pH. Given that GFP exhibits diminished fluorescence in acidic conditions while RFP remains relatively stable, the progression of autophagic flux can be assessed quantitatively through intensity of GFP fluorescence. LPS-induced autophagic blockade intensified intracellular green fluorescence intensity, which was subsequently diminished by WA, indicating a facilitated intracellular autophagic flux (Fig. 5E). Subsequent Western blot analyses confirmed these findings by illustrating that WA promoted microtubule-associated protein 1 light chain 3 beta (MAP1LC3B) generation while reducing SQSTM1 deposition (Fig. 5F-H). Hence, the aforementioned experimental investigations collectively indicated that the key mechanism of WA for promoting LDs metabolism and mitigating inflammation release lied in its ability to stimulate cellular autophagy, thereby alleviating neuroinflammation.
Fig. 5.
WA promoted microglia autophagy and accelerated the degradation of LDs. Microglia were pre-treated with MG132 (10 µM) or 3-MA (5 mM) for one hour, followed by WA protection for one hour, and subsequently stimulated with LPS for 12 h. (A) BODIPY fluorescence probe was used to label LDs molecules in primary microglia (scale bar = 5 μm). (B) Average fluorescence intensity of labeled LDs was measured. (Four independent replicate experiments were conducted). (C) LDs accumulation was assessed in primary microglia through Oil Red staining (scale bar = 5 μm). (D Average area covered by labeled LDs on cells was determined (Four independent replicate experiments were conducted). (E) Primary microglia were labeled using a dual-fluorescence virus (GFP-RFP-MAP1LC3B) to visualize autophagosomes (scale bar = 5 μm). (F) Immunoblotting was performed to detect SQSTM1 and MAP1LC3B expression levels. Band intensities of SQSTM1 and MAP1LC3B in Western blots were quantified and presented in (G) and (H), respectively (Three independent replicate experiments were performed). *P < 0.05, **P < 0.01, ***P < 0.001 compared with the corresponding group, as determined by the one-way ANOVA .
WA improved the autophagy level in mice with POCD
The previous cellular-level investigations have already demonstrated that WA promoted the autophagy process. Next, we aimed to examine whether WA could similarly enhance autophagy levels in an in vivo animal model. Mirroring the preceding in vivo experiments, we focused on the hippocampal dentate gyrus (DG) region, a brain area implicated in mouse cognition. Our findings revealed that WA exhibited a dose-dependent increase in the expression of MAP1LC3B, indicative of heightened autophagy levels. This was manifested by an elevated total protein quantity of MAP1LC3B in the hippocampal DG region and an increased expression of MAP1LC3B on microglia (Fig. 6A-B). Moreover, results from fluorescence co-labeling of SQSTM1 further indicated that WA reduced SQSTM1 deposition on microglia cells, underscoring the augmented cellular autophagy activity (Fig. 6C-E). Thus, the aforementioned outcomes confirmed that WA had the capacity to enhance autophagy in microglia cells both in vitro and in vivo, leading to a reduction in LDs accumulation and attenuation of inflammatory responses.
Fig. 6.
WA improved the autophagy level in mice with POCD. (A) Co-labeling of microglia in the hippocampal region of mice with MAP1LC3B was performed (scale bar = 10–5 μm), analyzed by Imaris software and the quantified data were presented in (B) (N = 3). (C) Co-labeling of microglia in the hippocampal region of mice with SQSTM1 was carried out (scale bar = 40–20 μm)). The relative fluorescence intensity of SQSTM1 was shown in (D), and the number of co-labeled cells was displayed in (E) (N = 3).*P < 0.05, **P < 0.01, ***P < 0.001 compared with the corresponding group, as determined by the one-way ANOVA .
Discussion
The results presented in this study offer significant insights into the potential of WA as a promising therapeutic agent for mitigating POCD by targeting LDs metabolism, inflammation, and autophagy. The multifaceted approach employed in this investigation provided a comprehensive understanding of mechanisms of WA, both at the cellular and in vivo levels. These findings have implications for the development of novel strategies to combat neuroinflammatory conditions and improve cognitive outcomes.
The cellular-level investigations provided a foundation for understanding impact of WA on LDs accumulation and inflammation. The reduction in LDs accumulation observed in microglia following WA administration aligns with emerging research suggesting that LDs played a role in exacerbating inflammatory responses. The unique contribution of this study is the identification of the ability of WA to promote autophagy as a mechanism for diminishing LDs accumulation. This autophagy-mediated reduction in LDs could be pivotal in curbing neuroinflammatory cascades triggered by excessive LDs, potentially extending the scope of therapeutic application of WA beyond POCD.
The transition to in vivo experiments reinforced the cellular findings and extended the scope of investigation to a more complex biological environment. The observation of dose-dependent increases in MAP1LC3B expression, indicative of heightened autophagy, in the hippocampal DG region following WA administration strengthens the argument for autophagy-inducing effects of WA. The alteration of autophagic flux as evidenced by reduced SQSTM1 deposition provided further confirmation of this effect. These results highlighted the potential of WA to modulate autophagy not only in isolated cells but also within the intricate network of the brain, further substantiating its neuroprotective potential.
The link between autophagy and neuroinflammation unveiled in this study underscores the importance of exploring autophagy modulation as a therapeutic strategy. Autophagy has emerged as a fundamental cellular process for maintaining homeostasis and managing cellular stress27,28. By enhancing autophagy, WA could potentially alleviate not only LDs accumulation but also broader inflammatory responses, offering a multifaceted approach to combatting neuroinflammation. This aligns with recent studies that have demonstrated the potential of autophagy modulation as a therapeutic strategy for neurodegenerative disorders29,32. Finally, in the Y-maze experiment, individual variability among mice led to a wide range of behaviors, with some high-dose group mice spending more time in the novel arm than Sham mice. This trend, while notable, does not negate the overall observation that the average time spent in the novel arm by the high-dose group remained lower. The Y-maze is a widely used behavioral test for assessing spatial memory and cognitive flexibility in rodents, capitalizing on their natural inclination to explore new environments. In the context of cognitive impairment, such as POCD, the Y-maze can reveal deficits in exploratory behavior and memory, indicating potential underlying neurological changes. These findings suggest that while WA may influence exploratory behavior, further investigation into the underlying mechanisms and statistical analyses is necessary to fully understand its effects on cognitive function in POCD.
However, as with any complex study, several limitations should be acknowledged. While the results demonstrated a consistent trend of autophagy stimulation and inflammation reduction with WA administration, the precise molecular mechanisms underlying these effects require further elucidation. Additionally, the current study primarily focused on microglia cells, which are key players in neuroinflammation, but the potential impact of WA on other cell types within the brain should not be overlooked. Moreover, the long-term effects of sustained WA administration, its potential interactions with other pathways, and its safety profile in vivo need to be thoroughly investigated.
In conclusion, the findings presented in this study highlight the multifaceted neuroprotective properties of WA in the context of neuroinflammation and cognitive dysfunction. By promoting autophagy and thereby reducing LDs accumulation and inflammation, WA presented itself as a compelling candidate for therapeutic interventions in neuroinflammatory conditions. The results provide a foundation for further research to uncover the detailed mechanisms of WA, assess its long-term effects, and evaluate its potential clinical translation. Ultimately, the discoveries made in this study contribute to the broader understanding of autophagy modulation as a strategy to counteract neuroinflammatory processes and enhance cognitive function.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
Thanks to all the researchers in the team for their hard work.
Author contributions
Lelin Huang and Lieliang Zhang took charge of designing the entire experiment and providing guidance for its practical execution. Haijun Hu and Bingbing Cao were mainly responsible for in vivo experimental research and manuscript writing. Dan Huang and Yue Lin chiefly oversaw research concerning primary dissociated microglia in vitro. Bin Zhou and Jun Ying undertook the tasks of gathering information and consulting literature.
Funding
This work was supported by National Natural Science Foundation of China (82360227); Natural Science Foundation of Jiangxi Province (20232BAB206060, 20212BAB216048); Health Commission of Jangxi Province (202310513).
Data availability
Data is provided within the manuscript or supplementary information files.
Declarations
Competing interests
The authors declare no competing interests.
Consent for publication
All authors have read and agreed with the final paper.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: Haijun Hu and Bingbing Cao.
Contributor Information
Lelin Huang, Email: h13507029069@163.com.
Lieliang Zhang, Email: zllndefy@163.com.
References
- 1.Needham, M. J., Webb, C. E. & Bryden, D. C. Postoperative cognitive dysfunction and dementia: What we need to know and do. Br. J. Anaesth.119, i115–i125 (2017). [DOI] [PubMed] [Google Scholar]
- 2.Travica, N., Lotfaliany, M., Marriott, A., Safavynia, S. A., Lane, M. M., Gray, L., Veronese, N., Berk, M., Skvarc, D., Aslam, H., Gamage, E., Formica, M., Bishop, K. & Marx, W. Peri-operative risk factors associated with post-operative cognitive dysfunction (POCD): An umbrella review of meta-analyses of observational studies. J. Clin. Med.12 (2023). [DOI] [PMC free article] [PubMed]
- 3.Migirov, A., Chahar, P. & Maheshwari, K. Postoperative delirium and neurocognitive disorders. Curr. Opin. Crit. Care27, 686–693 (2021). [DOI] [PubMed] [Google Scholar]
- 4.Li, D., Chen, M., Meng, T. & Fei, J. Hippocampal microglial activation triggers a neurotoxic-specific astrocyte response and mediates etomidate-induced long-term synaptic inhibition. J. Neuroinflamm.17, 109 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Li, Z., Zhu, Y., Kang, Y., Qin, S. & Chai, J. Neuroinflammation as the underlying mechanism of postoperative cognitive dysfunction and therapeutic strategies. Front. Cell. Neurosci.16, 843069 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Farmer, B. C., Kluemper, J,& Johnson, L.A. Apolipoprotein E4 alters astrocyte fatty acid metabolism and lipid Droplet formation. Cells8 (2019). [DOI] [PMC free article] [PubMed]
- 7.Nguyen, T. B. et al. DGAT1-dependent lipid droplet biogenesis protects mitochondrial function during starvation-induced autophagy. Dev. Cell42, 9-21.e5 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Walther, T. C., Chung, J. & Farese, R. V. Jr. Lipid Droplet biogenesis. Annu. Rev. Cell Dev. Biol.33, 491–510 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Olzmann, J. A. & Carvalho, P. Dynamics and functions of lipid droplets. Nat. Rev. Mol. Cell Biol.20, 137–155 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ioannou, M. S. et al. Neuron-astrocyte metabolic coupling protects against activity-induced fatty acid toxicity. Cell177, 1522-1535.e14 (2019). [DOI] [PubMed] [Google Scholar]
- 11. Moulton, M. J., Barish, S., Ralhan, I., Chang, J., Goodman, L. D., Harland, J. G., Marcogliese, P. C., Johansson, J. O., Ioannou, M. S. & Bellen, H. J. Neuronal ROS-induced glial lipid droplet formation is altered by loss of Alzheimer’s disease-associated genes. Proc. Natl. Acad. Sci. USA118 (2021). [DOI] [PMC free article] [PubMed]
- 12.Marschallinger, J. et al. Lipid-droplet-accumulating microglia represent a dysfunctional and proinflammatory state in the aging brain. Nat. Neurosci.23, 194–208 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Victor, M. B. et al. Lipid accumulation induced by APOE4 impairs microglial surveillance of neuronal-network activity. Cell Stem Cell29, 1197-1212.e8 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zhang, L. et al. CB2R activation regulates TFEB-mediated autophagy and affects lipid metabolism and inflammation of astrocytes in POCD. Front. Immunol.13, 836494 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Liu, L., MacKenzie, K. R., Putluri, N., Maletić-Savatić, M. & Bellen, H. J. The glia-neuron lactate shuttle and elevated ros promote lipid synthesis in neurons and lipid droplet accumulation in glia via APOE/D. Cell Metab.26, 719-737.e6 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Yang, Y. et al. Neuroinflammation-mediated mitochondrial dysregulation involved in postoperative cognitive dysfunction. Free Radic. Bbiol. Med.178, 134–146 (2022). [DOI] [PubMed] [Google Scholar]
- 17.Zhang, L. et al. Dexmedetomidine mitigated NLRP3-mediated neuroinflammation via the ubiquitin-autophagy pathway to improve perioperative neurocognitive disorder in mice. Front. Pharmacol.12, 646265 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Das, R., Rauf, A., Akhter, S., Islam, M. N., Emran, T. B., Mitra, S., Khan, I. N. & Mubarak, M. S. Role of Withaferin A and its derivatives in the management of Alzheimer’s disease: Recent trends and future perspectives. Molecules26 (2021). [DOI] [PMC free article] [PubMed]
- 19.Zhou, Z. X. et al. Withaferin A inhibits ferroptosis and protects against intracerebral hemorrhage. Neural Regener. Res.18, 1308–1315 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhou, Z. et al. Withaferin A alleviates traumatic brain injury induced secondary brain injury via suppressing apoptosis in endothelia cells and modulating activation in the microglia. Eur. J. Pharmacol.874, 172988 (2020). [DOI] [PubMed] [Google Scholar]
- 21.Zhao, M. et al. The DJ1-Nrf2-STING axis mediates the neuroprotective effects of Withaferin A in Parkinson’s disease. Cell Death Differ.28, 2517–2535 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kwon, H. S. & Koh, S. H. Neuroinflammation in neurodegenerative disorders: The roles of microglia and astrocytes. Transl. Neurodegener.9, 42 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Woodburn, S. C., Bollinger, J. L. & Wohleb, E. S. The semantics of microglia activation: Neuroinflammation, homeostasis, and stress. J. Neuroinflamm.18, 258 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hickman, S., Izzy, S., Sen, P., Morsett, L. & El Khoury, J. Microglia in neurodegeneration. Nat. Neurosci.21, 1359–1369 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Colonna, M. & Butovsky, O. Microglia function in the central nervous system during health and neurodegeneration. Annu. Revi. Immunol.35, 441–468 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zhu, H. et al. Targeting CB2R in astrocytes for Parkinson’s disease therapy: Unraveling the Foxg1-mediated neuroprotective mechanism through autophagy-mediated NLRP3 degradation. J. Neuroinflamm.20, 304 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Fleming, A. et al. The different autophagy degradation pathways and neurodegeneration. Neuron110, 935–966 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Vargas, J. N. S., Hamasaki, M., Kawabata, T., Youle, R. J. & Yoshimori, T. The mechanisms and roles of selective autophagy in mammals. Nat. Revi. Mol. Cell Biol.24, 167–185 (2023). [DOI] [PubMed] [Google Scholar]
- 29.Klionsky, D. J., Petroni, G., Amaravadi, R. K., Baehrecke, E. H., Ballabio, A., Boya, P., Bravo-San Pedro, J. M., Cadwell, K., Cecconi, F., Choi, A. M. K., Choi, M. E., Chu, C. T., Codogno, P., Colombo, M. I., Cuervo, A. M., Deretic, V., Dikic, I., Elazar, Z., Eskelinen, E. L., Fimia, G. M., Gewirtz, D. A., Green, D. R., Hansen, M., Jäättelä, M., Johansen, T., Juhász, G., Karantza, V., Kraft, C., Kroemer, G., Ktistakis, N. T., Kumar, S., Lopez-Otin, C., Macleod, K. F., Madeo, F., Martinez, J., Meléndez, A., Mizushima, N., Münz, C., Penninger, J. M., Perera, R. M., Piacentini, M., Reggiori, F., Rubinsztein, D. C., Ryan, K. M., Sadoshima, J., Santambrogio, L., Scorrano, L., Simon, H. U., Simon, A. K., Simonsen, A., Stolz, A., Tavernarakis, N., Tooze, S. A., Yoshimori, T., Yuan, J., Yue, Z., Zhong, Q., Galluzzi, L. & Pietrocola, F. Autophagy in major human diseases. EMBO J.40, e108863 (2021). [DOI] [PMC free article] [PubMed]
- 30.Ajoolabady, A. et al. Targeting autophagy in ischemic stroke: From molecular mechanisms to clinical therapeutics. Pharmacol. Therap.225, 107848 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Choi, I. et al. Autophagy enables microglia to engage amyloid plaques and prevents microglial senescence. Nat. Cell Biol.25, 963–974 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Han, X. et al. Small molecule-driven NLRP3 inflammation inhibition via interplay between ubiquitination and autophagy: Implications for Parkinson disease. Autophagy15, 1860–1881 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data is provided within the manuscript or supplementary information files.