Abstract
In plant cells, the Golgi apparatus is the key organelle for polysaccharide and glycolipid synthesis, protein glycosylation and protein sorting towards various cellular compartments. Protein import from the endoplasmic reticulum (ER) is a highly dynamic process, and new data suggest that transport, at least of soluble proteins, occurs via bulk flow. In this Botanical Briefing, we review the latest data on ER/Golgi inter‐relations and the models for transport between the two organelles. Whether vesicles are involved in this transport event or if direct ER–Golgi connections exist are questions that are open to discussion. Whereas the majority of proteins pass through the Golgi on their way to other cell destinations, either by vesicular shuttles or through maturation of cisternae from the cis‐ to the trans‐face, a number of membrane proteins reside in the different Golgi cisternae. Experimental evidence suggests that the length of the transmembrane domain is of crucial importance for the retention of proteins within the Golgi. In non‐dividing cells, protein transport out of the Golgi is either directed towards the plasma membrane/cell wall (secretion) or to the vacuolar system. The latter comprises the lytic vacuole and protein storage vacuoles. In general, transport to either of these from the Golgi depends on different sorting signals and receptors and is mediated by clathrin‐coated and dense vesicles, respectively. Being at the heart of the secretory pathway, the Golgi (transiently) accommodates regulatory proteins of secretion (e.g. SNAREs and small GTPases), of which many have been cloned in plants over the last decade. In this context, we present a list of regulatory proteins, along with structural and processing proteins, that have been located to the Golgi and the ‘trans‐Golgi network’ by microscopy.
Key words: Review, Golgi, endoplasmic reticulum, prevacuolar compartment, vacuole, plasma membrane, protein transport, protein sorting, vesicles, SNAREs, small GTPases
INTRODUCTION
In plants, the Golgi apparatus is central to the synthesis of complex cell wall polysaccharides and of glycolipids for the plasma membrane, as well as the addition of oligosaccharides to proteins destined to reach the cell wall, plasma membrane or storage vacuoles. The Golgi apparatus is also the key organelle in sorting proteins, sending them to their various destinations within the cell. The majority of these proteins are imported into the Golgi from the endoplasmic reticulum (ER), a major organelle of the endomembrane system involved in the folding, processing, assembly and storage of proteins, as well as in lipid biosynthesis and storage (Vitale and Denecke, 1999). The relative importance of the two major Golgi functions in a plant cell, the assembly and processing of oligo‐ and polysaccharides on the one hand and protein sorting on the other, depends on the cell type and its developmental and physiological state (Juniper et al., 1982). Nevertheless, the two functions cannot be regarded as completely unrelated processes; newly synthesized cell wall polysaccharides have to reach the correct target destination and must therefore be appropriately sorted.
The plant Golgi apparatus shares many features with its animal counterpart, but also has unique characteristics. The most important difference concerns its structure. Whereas in animal cells the Golgi apparatus occupies a rather stationary perinuclear position, in plant cells the Golgi is divided into individual Golgi stacks, which are generally considered to be functionally independent (Staehelin and Moore, 1995) (Fig. 1). The number of Golgi stacks per cell and the number of cisternae per stack vary with the species and cell type, but also reflect the physiological conditions, the developmental stage and the functional requirement of a cell (reviewed by Staehelin and Moore, 1995; Andreeva et al., 1998b). Despite these variations, each individual Golgi stack can be described as a polarized structure with its cisternal morphology and its enzymatic activities changing gradually from the ER‐adjacent cis‐face to the trans‐face (Fitchette et al., 1999). Proteins destined for secretion enter the Golgi at the cis‐face and subsequently move towards the trans‐face where the majority of proteins exit the stack en route to the plasma membrane or vacuolar system (for exceptions see below). In between the cisternae of the Golgi stack, intercisternal elements can be observed, mainly towards the trans‐face (Ritzenthaler et al., 2002) (Fig. 1A). Although no matrix proteins surrounding the plant Golgi stack have yet been identified, the existence of a matrix has been predicted from the appearance on micrographs from ultra‐rapidly frozen root cells of a clear zone, excluding ribosomes, around each Golgi (Staehelin et al., 1990). The Golgi matrix has been suggested to play an important role in the maintenance of stack organization against the shearing forces during cytoplasmic streaming (Staehelin and Moore, 1995).
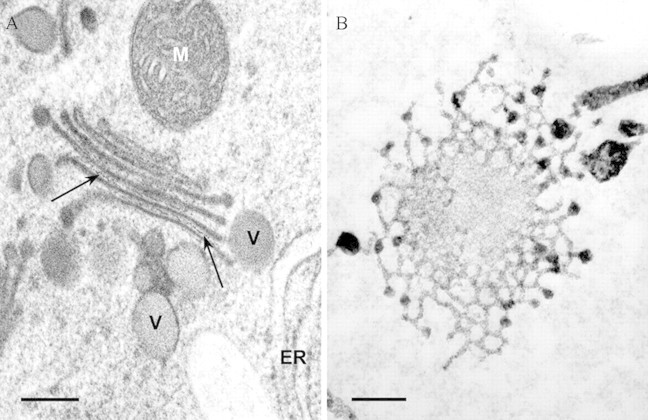
Fig. 1. Transmission electron micrographs of Golgi stacks in tobacco (A) and maize roots (B). A, Cross‐section of a Golgi stack in a tobacco root cap cell. High‐pressure freezing and freeze‐substitution improves the ultrastructural preservation of intercisternal filaments towards the trans‐face of the Golgi stack (arrows). B, Face view of a Golgi cisternum in a maize root meristematic cell. Zinc iodide and osmium tetroxide impregnation selectively stains the ER and the Golgi and clearly shows the fenestrated margins of the Golgi cisternum. ER, Endoplasmic reticulum; M, mitochondrion; V, vesicle. Bars = 200 nm.
Confocal microscopy of Golgi‐targeted proteins or peptides fused to the green fluorescent protein (GFP) has revealed that individual stacks are highly mobile within the plant cell, moving over the ER on an actin‐network (Boevink et al., 1998; Nebenführ et al., 1999; Brandizzi et al., 2002b) (Fig. 2); this has resulted in them being christened ‘stacks on tracks’ (Boevink et al., 1998) or ‘mobile factories’ (Nebenführ and Staehelin, 2001). The fact that the plant Golgi apparatus is divided into highly mobile biosynthetic subunits certainly poses major problems when trying to elucidate mechanisms for controlled protein import into and targeted product export out of the stack.
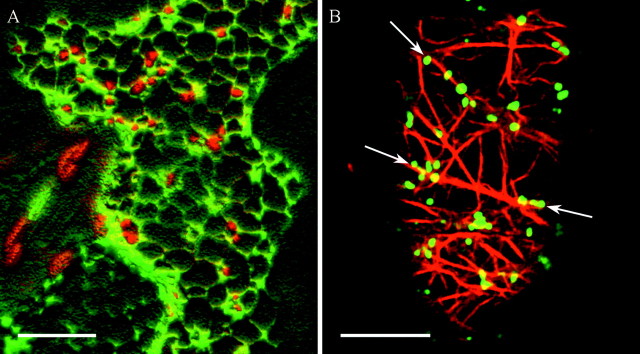
Fig. 2. Confocal laser scanning micrographs showing the spatial relationship between Golgi stacks and ER (A) and between Golgi stacks and actin filaments (B). A, 3D‐reconstruction (Velocity®) of serial optical sections through the cortical cytoplasm of a tobacco leaf epidermal cell transiently transformed with a GFP‐fusion targeted to the ER (GFP‐HDEL in green) and a YFP‐fusion labelling the Golgi (ST‐YFP in red). Golgi stacks are in close association with the ER network. B, Optical section through the cortical cytoplasm of a tobacco BY2 cell stably transformed with ST‐GFP (in green). Affinity labelling of actin by rhodamine‐phalloidine (in red) reveals that Golgi stacks are aligned with actin filaments (arrows). Bars = 10 µm.
In this Botanical Briefing, we summarize recent findings regarding protein transport from the ER to the Golgi apparatus and sorting of proteins and membranes as they exit the Golgi.
ENTERING THE GOLGI:IMPORT FROM THE ER
Towards a more dynamic model of ER‐to‐Golgi protein transport
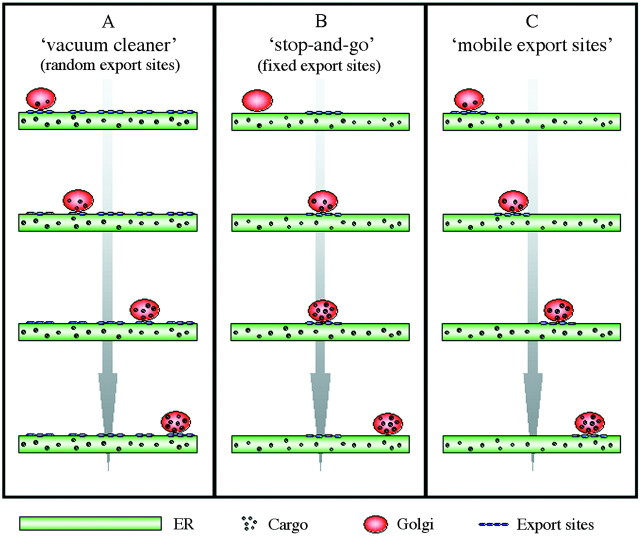
The discovery that Golgi stacks move in close association with the ER and that this movement is actin‐dependent (Boevink et al., 1998) led to the question of whether Golgi movement is necessary for ER‐to‐Golgi transport. The ‘vacuum cleaner model’ (Boevink et al., 1998) suggested that Golgi stacks move over the surface of the ER picking up products, similar to a vacuum cleaner picking up dust whilst moving over a carpet (Fig. 3A). This implies that the whole surface of the ER is capable of protein export, and that continued formation of cargo vectors occurs. In contrast, the hypothesis underlying the ‘stop‐and‐go’ or ‘recruitment model’ (Nebenführ et al., 1999) is that Golgi stacks receive cargo from the ER at defined export sites, which produce a local stop signal that transiently halts stack movement (Fig. 3B). This model is supported by the observation that actin‐based Golgi movement is not necessary for ER‐to‐Golgi membrane protein transport (Brandizzi et al., 2002b). Although more attractive, as protein transport out of the ER would be restricted to confined areas, the recruitment model suggests a rather stationary image of the ER surface. A third model might therefore propose that protein export from the ER is restricted to specific export sites, which could be either highly mobile within the ER membrane or mobile due to the movement of the ER surface (Fig. 3C). Thus, Golgi stacks and ER export sites may move together in an actin‐dependent fashion, forming discrete ‘secretory units’ (Brandizzi et al., 2002b).
Fig. 3. Models of ER‐to‐Golgi protein transport. A, The ‘vacuum cleaner model’ (Boevink et al., 1998) suggests that Golgi stacks move over the ER constantly picking up cargo. According to this model, the whole ER surface is capable of forming export sites, resulting in their random distribution. In contrast, the ‘stop‐and‐go’ model (B) hypothesizes that Golgi stacks stop at fixed ER export sites to take up cargo from the ER, before moving onto the next stop. In the more dynamic ‘mobile export sites’ model (C), Golgi stacks and ER export sites move together as ‘secretory units’ (Brandizzi et al., 2002b) allowing cargo to be transported from the ER towards the Golgi at any time during movement.
Receptor‐mediated protein transport or bulk flow?
Regardless of the physical model of ER‐to‐Golgi transport, export from the ER and import into the Golgi are two intimately related processes. How proteins are sorted and concentrated at sites of ER export is therefore a key question. Are they actively sorted with the help of specific ER export signals linking with differential affinity to a common ER export receptor, or is there bulk flow of product with sorting occurring via retention signals by which proteins are deviated from the default route at different levels of the pathway? Support for the second model, at least for soluble proteins, came from the cloning of an ERD2 homologue from Arabidopsis thaliana (Lee et al., 1993). This protein acts as a transmembrane receptor that binds to a specific sorting tetrapeptide (H/KDEL) at the carboxyl terminus of ER‐resident proteins in yeast and mammals (Lewis and Pelham, 1990; Lewis et al., 1990). The arabidopsis ERD2 is capable of functionally complementing a yeast null mutant (Lee et al., 1993), and its GFP fusion locates to the ER and to Golgi stacks (Boevink et al., 1998). Recent data from quantitative biochemical in vivo assays measuring ER export of well‐characterized cargo molecules in Nicotiana tabacum protoplasts provided good evidence for the bulk flow theory (Phillipson et al., 2001).
In vivo observations of membrane protein transport from ER to Golgi have recently been facilitated by the use of fluorescent protein chimeras. Selective photobleaching experiments using two fluorescent marker proteins locating to the Golgi (ST‐YFP and ERD2‐GFP) have permitted confocal imaging of ER‐to‐Golgi protein transport (Brandizzi et al., 2002b). Fluorescence recovery in individual Golgi stacks after photobleaching of ST‐YFP or ERD2‐GFP reached 80–90 % of the pre‐bleach level after only 5 min in cells treated with latrunculin B alone to disrupt actin filaments or in conjunction with colchicine to affect microtubule integrity. This indicated a rapid exchange with pools of the fusion protein from other parts of the cell, independent from an intact actin or microtubule cytoskeleton. The rapid cycling of both fusion proteins was dependent on energy, as could be shown by loss of fluorescence recovery in cells after ATP depletion.
COPII vesicles vs. direct connections
In animal cells, protein transport between the ER and the Golgi apparatus occurs through intermediate compartments known as vesicular–tubular clusters (VTCs) (or the ER–Golgi intermediate compartment, ERGIC). These compartments represent the first site of segregation of anterograde (forward from the ER to the Golgi) and retrograde (backwards from the Golgi to ER) protein transport (reviewed by Klumperman, 2000). Transport between the ER and VTCs is supposedly mediated by specialized protein‐coated vesicles, the so‐called COPII vesicles. Cargo packaging and vesicle formation require sequential binding to the ER membrane of the GTPase Sar1p and two heterodimeric coat protein complexes, Sec23/24p and Sec13/31p (reviewed by Barlowe, 2002). Transport between the VTCs and the Golgi apparatus seems not to be mediated by distinct vesicles but by the fusion of peripheral VTCs to form the cis‐Golgi cisternae (Klumperman, 2000). The involvement of another set of coated vesicles (COPI vesicles formed under the influence of the small GTPase Arf1p), normally thought to act as retrograde protein transporters, between the Golgi cisternae and from VTCs back to the ER (see below), in anterograde protein transport is still hotly debated (Klumperman, 2000; Spang, 2002). Finally, delivery of cargo to the Golgi complex involves the small GTPase Rab1, which controls tethering and fusion events at the Golgi level (Allan et al., 2000).
In plant cells, ER‐to‐Golgi protein transport might follow a simpler route. To date, no equivalent of the VTCs of animal cells has been identified. On the contrary, recent findings based on GFP expression and transmission electron microscopy (TEM) indicate that direct connections in the form of tubular extensions may exist between the ER and the cis‐face of Golgi stacks in tobacco leaf cells (Brandizzi et al., 2002b), suggesting that vesicles might not mediate protein transport between the two organelles. At first glance, this finding seems to contradict the fact that components of the COPII machinery have been identified in plants by EST database searches (Andreeva et al., 1998a), are associated with the ER (Bar‐Peled and Raikhel, 1997; Movafeghi et al., 1999), and are necessary for transport of proteins to the Golgi (Andreeva et al., 2000; Phillipson et al., 2001). This has been interpreted as evidence that ER–Golgi protein transport shows structural and functional similarities between animal and plant cells. However, it is conceivable that COPII components simply determine the site of formation of ER‐to‐cis‐Golgi‐connections, and that vesicle vectors are not a prerequisite of transport (Hawes et al., 1999). Considering that protein transport in mammalian cells between the VTCs and the cis‐Golgi seems to be by fusion of peripheral VTCs to form the cis‐cisternae, and that VTCs have not been identified in plant cells to date, direct fusion events between the ER and the Golgi are not implausible.
Similar to its mammalian counterpart, the A. thaliana small GTPase AtRab1b, seems to be involved in protein transport at early steps of the secretory pathway. A dominant‐inhibitory mutant of AtRab1b inhibited the secretion of a fluorescent marker protein as well as the Golgi localization of a Golgi‐targeted fluorescent marker, leading to fluorescence accumulating in the ER in both cases (Batoko et al., 2000).
Retention and distribution of proteins in the Golgi
The means by which Golgi‐resident proteins are retained in the stack is still largely a matter of debate. Whilst many of the ER‐resident processing proteins are soluble, Golgi‐resident enzymes are transmembrane proteins (Table 1). Information regarding the retention of membrane proteins at various points of the secretory pathway has recently been revealed by a study investigating the default pathway of type I membrane‐bound proteins (Brandizzi et al., 2002c). In this study, GFP was fused to proteins with transmembrane domains of variable length and the fusion proteins were shown to be distributed along the organelles of the secretory pathway in a transmembrane length‐dependent manner. As for animal cells (Munro, 1995), it is expected that the membrane thickness increases in plant cells from the ER to the Golgi and finally to the plasma membrane/tonoplast owing to a sterol gradient in the lipid composition (Moreau et al., 1998). Accumulation in the Golgi occurred when GFP was fused to transmembrane domains of 19 or 20 amino acids (Brandizzi et al., 2002c). Whether this retention mechanism is universally valid for true Golgi residents remains to be elucidated, although the signal anchor sequence of a rat sialyl transferase targets GFP to the plant Golgi (Boevink et al., 1998; Saint‐Jore et al., 2002), and equivalent sequences of plant glycosyltransferases give the same result (Essl et al., 1999; Dirnberger et al., 2002; Pagny et al., 2003). Phe residues, suggested to play a role in Golgi retention in mammalian cells, only seem to play a minor role in plants, as indicated by comparison of the number and position of Phe residues of the transmembrane domain of proteins within the BP80 family (Brandizzi et al., 2002c).
Table 1.
Examples of proteins microscopically located to the Golgi and the ‘trans‐Golgi network’ in cells of higher plants
| Protein (species of origin) | Experimental system | Microscopical technique | Localization | Putative function | Original reference (providing location data) |
| Regulatory proteins | |||||
| ERD2 (Arabidopsis thaliana) | Nicotiana clevelandii leaves transiently transformed with a GFP‐fusion of ERD2 (full length) | GFP imaging, TEM immunogold labelling with anti‐GFP antibodies | ER, all Golgi cisternae | H/KDEL receptor for retrieval of escaped soluble ER‐resident proteins | Boevink et al. (1998) |
| AtRER1B (Arabidopsis thaliana) | Nicotiana tabacum BY2 cells stably transformed with a GFP‐fusion of AtRER1B (full length) | GFP imaging | Golgi | Recycling receptor for membrane‐bound ER proteins | Takeuchi et al. (2000) |
| BP‐80 (also called VSRPS‐1) (Pisum sativum) | Pisum sativum root tips | Immunofluorescence and TEM immunogold labelling using anti‐VSRPS‐1 antibodies | Golgi, PVC | Sorting receptor for the lytic vacuole | Paris et al. (1997)* |
| AtELP (Arabidopsis thaliana) | Arabidopsis thaliana roots | TEM immunogold labelling with anti‐AtELP antibodies | Golgi, ‘trans‐Golgi network’. PVC | Sorting receptor for the lytic vacuole | Sanderfoot et al. (1998)* |
| AtArf1p (Arabidopsis thaliana) | Arabidopsis thaliana and Zea mays roots | TEM immunogold labelling with anti‐AtArf1p antibodies | Rims of Golgi cisternae and COPI vesicles | Small GTPase involved in COPI vesicle formation | Pimpl et al. (2000) |
| Rho‐like protein (N/A) | Nicotiana tabacum BY2 cells | Immunofluorescence using anti‐human Rac1 antibodies | Golgi | Small GTPase involved in regulating the cytoskeleton organization | Couchy et al. (1998)* |
| NtRab2 (Nicotiana tabacum) | Nicotiana tabacum pollen tubes transiently and stably transformed with a GFP‐fusion of NtRab2 (full length) | GFP imaging, TEM immunogold labelling with anti‐GFP antibodies | Golgi stack periphery | Small GTPase involved in ER‐to‐Golgi traffic | Cheung et al. (2002) |
| AtRab2a (Arabidopsis thaliana) | Nicotiana tabacum leaves transiently transformed with a GFP‐fusion of AtRab2a (full length) | GFP imaging, TEM immunogold labelling with anti‐GFP antibodies | Golgi, cytosol and small spherical bodies | Small GTPase involved in post‐Golgi trafficking | U. Neumann, I, Moore, C. Hawes and H. Batoko(unpubl. res.) |
| Pra2, Pra3 (Pisum sativum) | Nicotiana tabacum BY2 cells stably transformed with a GFP‐fusion of Pra2 and Pra3 (full length) | GFP imaging | Pra2: Golgi and ‘endosomes’; Pra3: ‘TGN’ and/or PVC | Rab GTPases (Rab11 homologues) | Inaba et al. (2002)* |
| AtRac7 (Arabidopsis thaliana) | Nicotiana tabacum pollen tubes transformed with a GFP‐fusion of AtRac7 (full length) | GFP imaging | Golgi, plasma membrane (not at the pollen tube tip) | Rac‐like GTPase | Cheung et al. (2003) |
| ADL6 (Arabidopsis thaliana) | Arabidopsis thaliana root tips; A. thaliana protoplasts transformed with a GFP‐fusion of ADL6 (full length) | Immunofluorescence using anti‐ADL6 antibodies (root tips); GFP imaging (protoplasts) | Golgi | Dynamin‐like protein involved in vesicle formation for vacuolar traffic at the ‘TGN’ | Jin et al. (2001)* |
| AtSH3P1 (Arabidopsis thaliana) | Arabidopsis thaliana pollen grains | TEM immunogold labelling with anti‐AtSH3P1 antibodies | PM and adjacent vesicles, vesicles of the ‘TGN’, PCR | Protein involved in the fission and uncoating of clathrin‐coated vesicles | Lam et al. (2001)* |
| α‐actinin‐like protein (N/A) | Lilium davidii, pollen and pollen tubes | Immunofluorescence, TEM immunogold labelling using commercially available anti‐α‐actinin antibodies | Membranes of Golgi‐associated vesicles | Budding and sorting of Golgi‐associated vesicles | Li and Yen (2001)* |
| Structural proteins | |||||
| Clathrin (N/A) | N/A | Electron microscopy (identification of clathrin interlocking triskelions) | Golgi, partially coated reticulum, multivesicular bodies, plasma membrane, clathrin‐coated vesicles | Coat protein of clathrin coated vesicles | for original references see Coleman et al. (1988)* |
| Atγ‐COP (Arabidopsis thaliana) Zmδ‐COP, Zmϵ‐COP (Zea mays) | Arabidopsis thaliana and Zea mays roots | TEM immunogold labelling using antibodies raised against the different COP components | Rims of Golgi cisternae and COPI vesicles | Coatomer subunit of COPI vesicles | Pimpl et al. (2000) |
| SNAREs | |||||
| AtVTI1a (now AtVTI11) (Arabidopsis thaliana) | Arabidopsis thaliana roots from plants stably transformed with T7‐tagged AtVTI1a | TEM immunogold labelling using anti‐T7 antibodies | ‘Trans‐Golgi network’, dense vesicles, PVC | SNARE involved in protein transport from the Golgi to the PVC | Zheng et al. (1999)* |
| AtVPS45, AtTLG2a, ‐b (now AtSYP41, AtSYP42) (Arabidopsis thaliana) | Arabidopsis thaliana roots: wild type or from plants transformed with HA‐tagged AtTLG2a and T7‐tagged AtTLG2b | TEM immunogold labelling using anti‐AtVPS45, anti‐AtTLG2a, anti‐HA and/or anti‐T7 antibodies | ‘Trans‐Golgi network’ | SNAREs | Bassham et al. (2000)* |
| AtSYP51, AtSYP61 (Arabidopsis thaliana) | Arabidopsis thaliana roots: wild type or from plants transformed with HA‐AtSYP41 and T7‐AtSYP42 | TEM immunogold labelling | AtSYP51: ‘TGN’, PVC; AtSYP61: ‘TGN’ | Syntaxins (SNAREs) | Sanderfoot et al. (2001)* |
| AtSed5 (now called AtSYP31) (Arabidopsis thaliana) | Nicotiana tabacum BY2 cells transformed with a GFP‐fusion of AtSed5 (full length) | GFP imaging | Golgi | Syntaxin (SNARE) | Takeuchi et al. (2002) |
| Processing proteins | |||||
| α‐2,6‐sialyl transferase (rat) | Nicotiana clevelandii leaves transiently transformed with a GFP‐fusion of the 52 N‐terminal amino acids of sialyl transferase | GFP imaging, TEM immunogold labelling with anti‐GFP antibodies | Trans‐face of the Golgi | Glycosyltransferase | Boevink et al. (1998) |
| α‐2,6‐sialyl transferase (rat) | Arabisopsis thaliana (roots and callus) stably transformed with Myc‐tagged sialyl transferase (full length) | Immunofluorescence and TEM immunogold labelling using 9E10 and A14 anti‐Myc antibodies | Trans‐face of the Golgi | Glycosyltransferase | Wee et al. (1998)* |
| N‐acetylglucoso‐aminyltransferase I (GnTI) (Nicotiana tabacum) | Nicotiana benthamiana leaves transiently transformed with a GFP‐fusion of the GnTI cytoplasmic transmembrane stem (CTS) domain | GFP imaging | Golgi | Glycosyltransferase | Essl et al. (1999) |
| α‐1,2 mannosidase I soybean (Glycine max) | Nicotiana tabacum BY2 cells stably transformed with a GFP‐fusion of mannosidase I (deletion of C‐terminal 11 amino acids) | GFP imaging, TEM immunogold labelling with anti‐GFP antibodies | Cis‐face of the Golgi | N‐linked oligosaccharide processing enzyme | Nebenführ et al. (1999) |
| GONST1 (Arabidopsis thaliana) | Onion epidermal cells transiently transformed with a YFP‐fusion of GONST1 (full length) | YFP imaging | Golgi | Nucleotide sugar (GDP‐mannose) transporter | Baldwin et al. (2001)* |
| β 1,2‐xylosyltransferase (Arabidopsis thaliana) | Nicotiana benthamiana leaves transiently transformed with GFP‐fusions of different β 1,2‐xylosyltransferase domains (CTS, CT, T, C) | GFP imaging | Golgi | Glycosyltransferase | Dirnberger et al. (2002) |
| Xylan synthase (Phaseolus vulgaris) | Phaseolus vulgaris hypocotyl | TEM immunogold labelling with anti‐bean xylan synthase antibodies | Golgi and post‐Golgi vesicles of developing xylem cells | Synthesis of secondary wall xylan | Gregory et al. (2002)* |
| β 1,3‐glucan (callose) synthase (Phaseolus vulgaris) | Phaseolus vulgaris hypocotyls and root tips | TEM immunogold labelling with anti‐β 1,3‐glucan synthase antibodies | Golgi of root tip meristematic cells during cell plate formation, surface of secondary wall thickenings and PM in pits in developing xylem cells | Callose synthesis | Gregory et al. (2002)* |
| β‐1,4‐galactosyl transferase (human) | Nicotiana tabacum leaves transiently transformed with a GFP‐fusion of galactosyl transferase (60 C‐terminal amino acids) | GFP imaging | ER and Golgi | Glycosyltransferase | Saint‐Jore et al. (2002) |
| MUR4 (Arabidopsis thaliana) | Root protoplasts of Arabidopsis thaliana plants stably transformed with a GFP‐fusion of MUR4 (full length) | GFP imaging | Golgi | UDP‐D‐Xyl 4‐epimerase | Burget et al. (2003)* |
* Literature cited in this table only, not in the main text:
Baldwin TC, Handford MG, Yuseff MI, Orellana A, Dupree P. 2001. Identification and characterization of GONST1, a Golgi‐localized GDP‐mannose transporter in Arabidopsis. The Plant Cell 13: 2283–2295.
Bassham DC, Sanderfoot AA, Kovaleva V, Zheng H, Raikhel NV. 2000. AtVPS45 complex formation at the trans‐Golgi network. Molecular Biology of the Cell 11: 2251–2265.
Burget EG, Verma R, Mølhøj M, Reiter W‐D. 2003. The biosynthesis of l‐arabinose in plants: molecular cloning and characterization of a Golgi‐localized UDP‐D‐xylose 4‐epimerase encoded by the MUR4 gene of Arabidopsis. The Plant Cell 15: 523–531.
Cheung AY, Chen CYH, Tao LZ, Andreyeva T, Twell D, Wu HM. 2003. Regulation of pollen tube growth by Rac‐like GTPases. Journal of Experimental Botany 54: 73–81.
Coleman J, Evans H, Hawes C. 1988. Plant coated vesicles. Plant, Cell and Environment 11: 669–684.
Couchy I, Minic Z, Laporte J, Brown S, Satiat‐Jeunemaitre B. 1998. Immunodetection of Rho‐like plant proteins with Rac1 and Cdc42Hs antibodies. Journal of Experimental Botany 49: 1647–1659.
Gregory ACE, Smith C, Kerry ME, Wheatley ER, Bolwell GP. 2002. Comparative subcellular immunolocation of polypeptides associated with xylan and callose synthases in French bean (Phaseolus vulgaris) during secondary wall formation. Phytochemistry 59: 249–259.
Inaba T, Nagano Y, Nagasaki T, Sasaki Y. 2002. Distinct localization of two closely related Ypt3/Rab11 proteins on the trafficking pathway in higher plants. Journal of Biological Chemistry 277: 9183–9188.
Jin JB, Kim YA, Kim SJ, Lee SH, Kim DH, Cheong GW, Hwang I. 2001. A new dynamin‐like protein, ADL6, is involved in trafficking from the trans‐Golgi network to the central vacuole in Arabidopsis. The Plant Cell 13: 1511–1525.
Lam BCH, Sage TL, Bianchi F, Blumwald E. 2001. Role of SH3‐containing proteins in clathrin‐mediated vesicle trafficking in Arabidopsis. The Plant Cell 13: 2499–2512.
Li Y, Yen LF. 2001. Plant Golgi‐associated vesicles contain a novel alpha‐actinin‐like protein. European Journal of Cell Biology 80: 703–710.
Paris N, Rogers SW, Jiang L, Kirsch T, Beevers L, Phillips TE, Rogers JC. 1997. Molecular cloning and further characterization of a probable plant vacuolar sorting receptor. Plant Physiology 115: 29–39.
Sanderfoot AA, Ahmed SU, Marty‐Mazars D, Rapoport I, Kirchhausen T, Marty F, Raikhel NV. 1998. A putative vacuolar cargo receptor partially colocalizes with AtPEP12p on a prevacuolar compartment in Arabidopsis roots. Proceedings of the National Academy of Sciences of the USA 95: 9920–9925.
Sanderfoot AA, Kovaleva V, Bassham DC, Raikhel NV. 2001. Interactions between syntaxins identify at least five SNARE complexes within the Golgi/prevacuolar system of the Arabidopsis cell. Molecular Biology of the Cell 12: 3733–3743.
Takeuchi M, Ueda T, Sato K, Abe H, Nagata T, Nakano A. 2000. A dominant negative mutant of Sar1p GTPase inhibits protein transport from the endoplasmic reticulum to the Golgi apparatus in tobacco and Arabidopsis cultured cells. The Plant Journal 23: 517–525.
Wee EGT, Sherrier J, Prime TA, Dupree P. 1998. Targeting of active sialyltransferase to the plant Golgi apparatus. The Plant Cell 10: 1759–1768.
Zheng H, Fischer von Mollard G, Kovaleva V, Stevens TH, Raikhel NV. 1999. The plant vesicle‐associated SNARE AtVTI1a likely mediates vesicle transport from the trans‐Golgi network to the prevacuolar compartment. Molecular Biology of the Cell 10: 2251–2264.
As mentioned earlier, enzymatic activities change gradually from the cis‐face to the trans‐face of the Golgi stack, reflecting a sequential processing down the stack. Therefore, a logical question is what controls the distribution of resident enzymes across the different cisternae. This has only been addressed experimentally a couple of times in plant cells (Fitchette et al., 1999), and models were first formulated for mammalian Golgi enzymes. Again, the bilayer/membrane thickness model provides one possible explanation. Alternatively, as in mammalian cells, retrograde transport of processing machinery in vesicle vectors may both reduce loss and control positioning of enzymes whilst cisternae continually mature from the cis‐ to the trans‐face (Opat et al., 2001). More evidence is needed before ruling out one or other model for plant Golgi enzymes.
Intra‐Golgi transport and retrograde transport (COPI machinery)
The fact that each Golgi stack is divided into a number of cisternae leads to the question of how cargo is transported down the stack. In analogy to mammalian cells, two models have been proposed, the ‘vesicle shuttle’ and the ‘cisternal maturation’ models (reviewed by Hawes and Satiat‐Jeunemaitre, 1996; Nebenführ and Staehelin, 2001). According to the vesicle shuttle model, Golgi cisternae are stable entities with a specific set of processing proteins, and cargo is sequentially transported down the stack in vesicular shuttles. The second model suggests that cisternae progressively move down the stack and mature from the cis‐face to the trans‐face of the Golgi. New cis‐cisternae are formed by fusion of ER‐to‐Golgi transport intermediates and retrograde transport vesicles, which shuttle back the processing enzymes from the trans‐cisternum. Experimental evidence exists for both models and it cannot be ruled out that either mechanism or a mixture of both is active. In addition to the algal scale argument (Becker et al., 1995), recent data on the ultrastructural morphology of Golgi stacks in BY2 cells and the redistribution of a fluorescent cis‐Golgi marker protein after brefeldin A treatment (a fungal toxin widely used to study protein transport along the secretory pathway; Nebenführ et al., 2002) seem to favour the cisternal maturation model (Ritzenthaler et al., 2002). Finally, it cannot be excluded that cisternae of the stack are all joined by interconnecting tubules, necessitating a different model to explain the regulation of cis‐to‐trans‐transport.
In addition to anterograde protein transport from the ER to the Golgi and down the Golgi stack, retrograde protein transport occurs between the Golgi and the ER (e.g. transport of escaped ER residents) as well as inside the Golgi stack, from the trans‐ towards the cis‐face. In mammalian cells, retrograde transport is likely to be mediated by COPI vesicles and regulated by the Arf1p GTPase (Spang, 2002). Components of the COPI machinery have been located to the Golgi by means of immunofluorescence (Ritzenthaler et al., 2002) (Fig. 4A and B). More precisely, a study combining biochemistry with cryo‐section immunogold labelling techniques, revealed that in plant cells, components of the COPI coat as well as Arf1p mainly locate to the cis‐half of Golgi stacks and that COPI coat proteins are present on small vesicles budding off from cis‐cisternae (Pimpl et al., 2000). In addition, in vitro COPI vesicle induction from ER/Golgi membranes of transgenic tobacco plants overproducing the soluble secretory marker α‐amylase fused to HDEL (the C‐terminal sorting peptide of ER residents), showed that COPI vesicles contained the modified secretory marker as well as the ER‐resident calreticulin. This was the first indication in plants that COPI vesicles might be involved in retrograde transport from the Golgi (Pimpl et al., 2000). The involvement of AtArf1 in this particular transport event was also deduced from the effect of two dominant negative mutant forms of AtArf1 on the distribution of three fluorescent Golgi markers (Takeuchi et al., 2002). To date, the demonstration of the role of COPI vesicles in intra‐Golgi retrograde transport remains a challenge, mainly owing to the lack of marker molecules for this specific transport event.
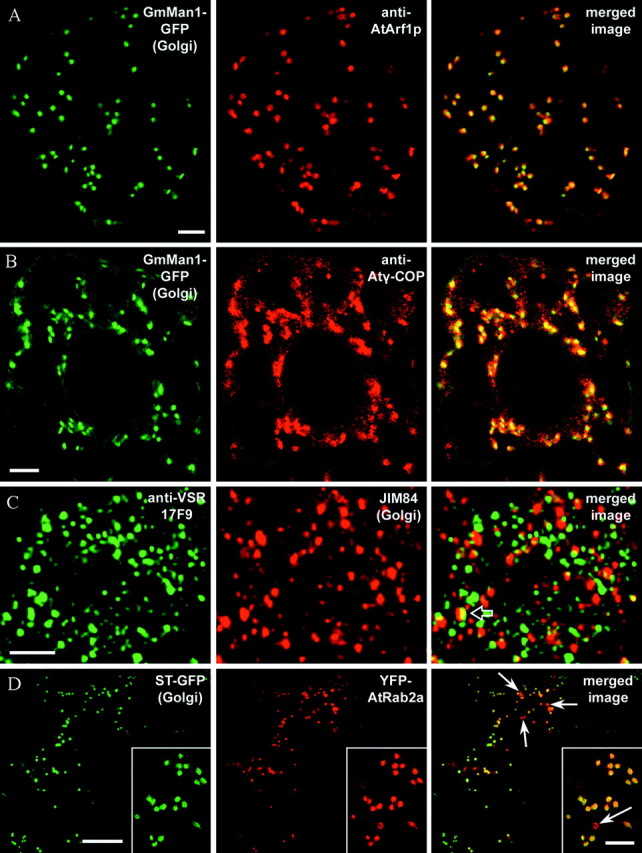
Fig. 4. Confocal laser scanning micrographs showing the location of different regulatory and structural proteins of the secretory pathway in relation to Golgi stacks. A, Optical section through the cortical cytoplasm of a tobacco BY2 cell stably transformed with the Golgi marker GmMan1‐GFP (Glycine max α‐mannosidase 1‐GFP, green channel) after fixation and immunolocalization with anti‐AtArf1p antibodies (red channel). The merged image clearly reveals that anti‐AtArf1p labelling is associated with the Golgi, forming a ring‐shaped pattern confined to the periphery of each stack (Ritzenthaler et al., 2002). B, Optical section through the centre of a transgenic GmMan1‐GFP BY2 cell after fixation (GFP signal in green channel) and immunolocalization with anti‐Atγ‐COP antibodies (red channel). As can be seen in the merged image, the COPI coatomer subunit co‐localizes with Golgi‐associated GFP‐fluorescence. As for Arf1p, anti‐Atγ‐COP fluorescence is restricted to the margins of the Golgi stacks (Ritzenthaler et al., 2002). C, Projection of 15 optical sections (1 µm per section) through a pea root tip cell after fixation and double‐immunolabelling with anti‐VSR antibodies (17F9, green channel) and JIM 84, a trans‐Golgi marker (red channel). As shown in the merged image, more than 90 % of the prevacuolar organelles labelled by 17F9 are separate from Golgi stacks (Li et al., 2002). Occasionally, fluorescence labelling by the two antibodies co‐localizes (merged image, open arrow). D, Optical section through the cortical cytoplasm of a leaf epidermal cell of a transgenic ST‐GFP tobacco plant (GFP signal in green channel) transiently transformed with YFP‐AtRab2a (YFP signal in red channel). As can be seen in the merged image, both fluorescent fusion proteins co‐localize in Golgi stacks. In addition to the Golgi, YFP‐AtRab2a labels small spherical structures (sometimes measuring up to 3 µm in diameter) in which no GFP signal can be detected (merged image, arrows). Bars = 5 µm (A–C, insert D) and 20 µm (D). Micrographs for A and B kindly provided by Christophe Ritzenthaler and that for C by Liwen Jiang.
Recycling through the Golgi: protein import from destinations other than the ER
During secretion, vesicle‐derived membrane is continuously added to the plasma membrane. To balance this increase in membrane surface area, clathrin‐coated endocytic vesicles pinch off from the plasma membrane towards the inside of the cell (reviewed by Holstein, 2002). The different endocytic compartments in plant cells are still not fully characterized. In analogy to mammalian cells, the term ‘endosome’ is often found in the plant literature indicating a compartment containing endocytosed material (Jürgens and Geldner, 2002). Ultrastructurally, the ‘plant endosome’ most likely corresponds to the partially coated reticulum (PCR), a compartment that may originate from the ‘trans‐Golgi network’ (Hillmer et al., 1988). From this compartment, protein might be recycled towards the Golgi or transported towards the lytic vacuole via multi‐vesicular bodies (MVBs) for degradation. This was shown by elegant time‐course studies following the uptake of cationized ferritin (CF) in soybean protoplasts (Fowke et al., 1991), reflecting vesicle‐mediated plasma membrane recycling (and not receptor‐mediated endocytosis). This marker is quickly endocytosed through coated pits into coated vesicles. Within the cytoplasm, CF sequentially labelled the following organelles: tubular elements of the PCR; periphery of Golgi cisternae; MVBs; and finally the central vacuole. Labelling of the Golgi by CF almost certainly reflects membrane recycling from the plasma membrane, presumably through the PCR (Fowke et al., 1991). Whether direct plasma membrane recycling to the Golgi can occur in plant cells still has to be elucidated.
In addition to maintaining the plasma membrane surface, protein import into the Golgi through the ‘plant endosome’ could also be important for regulating, by endocytosis at the plasma membrane, the number of ion channels, proton pumps (Crooks et al., 1999) and carrier proteins, such as PIN1 (auxin efflux carrier; see Geldner et al., 2001). Remodelling the distribution of such proteins at the plasma membrane via vesicle trafficking must certainly be regarded as an important way in which a plant can react to changes in environmental conditions (Levine, 2002).
OUT OF THE GOLGI
The major destinations for proteins exiting the Golgi apparatus are the plasma membrane and the vacuolar system. During cytokinesis, another protein trafficking route leads towards the developing cell plate (reviewed by Bednarek and Falbel, 2002). Here we focus on how sorting of proteins to the plasma membrane or the vacuolar system occurs and where this sorting takes place.
As to the place of sorting, the term ‘trans‐Golgi network’ (TGN) from the mammalian ‘endomembrane nomenclature’ is becoming more common in the plant literature. However, in plant cells, no discrete protein‐sorting compartment with a characteristic set of proteins, downstream from the trans‐face of the Golgi has yet been described. In addition, in plants, protein sorting and secretory vesicle production can take place as early as at the cis‐cisternae of the Golgi stack (see below). Therefore, it might be better to suggest that production of clathrin‐coated vesicles destined for the lytic vacuolar system is limited to the trans‐face of the Golgi. Whether this face of the Golgi, which certainly can comprise a network of tubules and vesicles, is homologous to the mammalian TGN is open for debate.
Secretion: transport towards the plasma membrane and the cell wall
The default destination of soluble proteins and complex carbohydrates has been suggested to be the plasma membrane (Denecke et al., 1990). Secretory proteins usually carry an amino terminal signal peptide for insertion into the ER, which is clipped off upon translocation across the ER membrane (Vitale and Denecke, 1999). Soluble secretory proteins are not known to carry any positive targeting information, which would divert them from the default pathway to the plasma membrane, either to the vacuolar system or back to the ER (Hadlington and Denecke, 2000).
In contrast, the situation with membrane proteins is less clear. Until recently, the tonoplast had been regarded as the default destination (Barrieu and Chrispeels, 1999). However, a VSR‐based (vacuolar sorting receptor) GFP‐fusion with a lengthened transmembrane domain accumulated on the plasma membrane, indicating that positive sorting information might be necessary to direct membrane proteins from the Golgi towards the tonoplast (Brandizzi et al., 2002c). Nevertheless, transport of proteins to specific areas of the plasma membrane, nicely illustrated by proteins such as the auxin efflux carrier PIN1, which locates to the distal part of the plasma membrane in root cells of arabidopsis seedlings (Geldner et al., 2001; Jürgens and Geldner, 2002), is difficult to imagine without some form of either positive targeting or retention mechanism.
Transport towards the vacuolar system: different types of vacuoles, vesicles, sorting signals, receptors and sorting sites
As mentioned earlier, sorting signals are necessary for transport to the vacuolar system. In plant cells, two main types of vacuoles [distinguishable by different sets of TIPs (tonoplast intrinsic proteins) and lumenal contents] may co‐exist, and sorting towards either of them depends on different peptide targeting signals and is mediated by different sets of transport vesicles (Paris et al., 1996; Jiang and Rogers, 1998; Hinz et al., 1999).
Transport to the lytic vacuole, characterized by γ‐TIPs, occurs via an intermediate compartment known as the prevacuolar compartment (PVC) and relies on amino‐terminal, sequence‐specific propeptides (NPIR or equivalent), which are recognized by VSRs. The first VSR to be identified was BP80 from Pisum sativum (now called VSRPS‐1; reviewed by Paris and Neuhaus, 2002). VCRs are thought to cycle between the Golgi apparatus and the PVC (Mitsuhashi et al., 2000) where they are preferentially located, as was shown by confocal immunofluorescence for several VSRPS‐1 homologues (Li et al., 2002) (Fig. 4C). It is thought that VSRs mediate the packaging of cargo destined to the lytic vacuole in trans‐Golgi‐located clathrin‐coated vesicles (Hinz et al., 1999).
In contrast, proteins delivered to the protein storage vacuole (α‐TIP vacuoles) reach their destination in vesicles apparently devoid of any specific protein coating and, in legume seeds, have been termed dense vesicles (Hohl et al., 1996). Sorting relies on a carboxyl‐terminal propeptide and an as yet unidentified sorting receptor, the existence of which is deduced from the observation that transport towards the storage vacuole can be saturated (Frigerio et al., 1998). Immunogold labelling indicates that sorting of some storage proteins can occur as early as the cis‐cisternae of the Golgi in pea cotyledons (Hillmer et al., 2001). Other storage vacuole proteins as well as integral membrane proteins are sorted even earlier, at the ER level, where they are packed into so‐called precursor‐accumulating (PAC) vesicles and transported to the storage vacuole via a route bypassing the Golgi (Mitsuhashi et al., 2001). A vacuolar sorting receptor for this pathway has been identified in pumpkin seeds (Shimada et al., 2002).
REGULATORY PROTEINS AT THE HEART OF THE SECRETORY PATHWAY
In recent years, it has become apparent that the different transport events along the secretory/biosynthetic and endocytic pathway are orchestrated by a plethora of proteins. The functional importance of the plant Golgi apparatus in the secretory pathway is reflected by the fact that numerous regulatory proteins are structurally and functionally linked to the Golgi and the ‘trans‐Golgi network’ (Hawes et al., 1999; Sanderfoot and Raikhel, 1999; Nebenführ, 2002; Rutherford and Moore, 2002; see Table 1). For example, small GTPases act as molecular switches involved in the formation, transport and fusion of transport vesicles, while SNAREs are integral membrane proteins involved in determining the specificity of fusion events along the endomembrane system, residing on transport vesicles (R‐SNAREs) and target membranes (Q‐SNAREs). Considerable effort has been put into comparing various genomes to identify plant homologues of proteins that have been shown to regulate various trafficking events along the secretory pathway in yeast and animals (Andreeva et al., 1998a; Sanderfoot et al., 2000; Jürgens and Geldner, 2002). Statements such as ‘Rab functions are conserved across eukaryotes, such that their subcellular localisation can be inferred from known localisations of members of the same subfamily in other species’ (Jürgens and Geldner, 2002) may be correct as to the functional aspect. However, even though homologous Rab proteins may show the same subcellular location, it cannot be necessarily concluded that they exert the same function. For instance, despite the Golgi‐location of two mammalian splice‐variants of Rab6, which differ only in three amino acid residues, Rab6A and Rab6A′ seem to function in different membrane‐trafficking events (Echard et al., 2000). Likewise, two plant Rab2 isoforms have been found to locate to Golgi stacks (Fig. 4D) but seem to regulate quite different transport steps. In tobacco pollen tubes, NtRab2 seems to sustain ER‐to‐Golgi traffic (Cheung et al., 2002), while studies conducted in our group seem to indicate that an arabidopsis Rab2 isoform regulates vesicle traffic between the Golgi and a post‐Golgi compartment (U. Neumann, I. Moore, C. Hawes and H. Batoko, unpubl. res.).
It is outside the scope of this current Briefing to consider the putative roles of the various small GTPases and SNAREs that have been identified in plants. A summary of the major regulatory proteins locating to the plant Golgi identified to date is given in Table 1.
CONCLUSIONS
Immense progress has been made in recent years to elucidate the various transport events along the secretory and endocytic pathways in eukaryotic cells. This is certainly true for the Golgi apparatus, which has seen a renaissance in research popularity since the 100th anniversary of its discovery by Camillo Golgi in 1898. New data regarding the molecular machinery that drives and regulates trafficking events at the Golgi level are published almost weekly. In this context, comparative genomic analyses have helped to identify plant homologues of yeast and animal proteins regulating Golgi‐related transport steps. One of the main challenges of the post‐genomic era is to provide functional and structural evidence for the specific roles of plant proteins putatively playing a role in the secretory/endocytic pathway. As to structural data at the subcellular level, technological progress in light microscopy such as confocal microscopy and deconvolution technology for improving images, combined with developments in immunolabelling and fluorescent protein technology have revolutionized the study of cell biology (Brandizzi et al., 2002a). However, especially with regard to the Golgi apparatus, confocal microscopy combined with GFP technology in order to locate proteins is not without its pitfalls. It becomes more and more common to assume that a punctate distribution of GFP fluorescence in the cytoplasm is sufficient to establish that a specific protein is located to the Golgi without additional confirmatory evidence such as co‐localization with a known Golgi marker at the light microscopical level (e.g. a fluorescent protein marker or by immunocytochemical labelling with ‘anti‐Golgi’ antibodies) or immunogold labelling at the TEM level. Cryotechniques for specimen preparation, such as ultra‐rapid freezing at ambient or high pressure and freeze‐substitution, allow for excellent ultrastructural preservation, but even without these more sophisticated microscopical techniques, immunogold labelling at the TEM level is an excellent way to localize proteins at the subcellular level.
Despite the progress made in recent years regarding the functioning of the secretory/endocytic pathway in general and the plant Golgi apparatus in particular, many aspects still remain to be discovered. The necessary advances will be made only if we are able to link molecular with structural and functional data.
ACKNOWLEDGEMENTS
We thank David Evans for critical reading of the manuscript, Barry Martin for skilful assistance with high‐pressure freezing and Liwen Jiang and Christophe Ritzenthaler for kindly providing micrographs. We acknowledge the Biotechnology and Biological Sciences Research Council, UK, for supporting the work undertaken in our laboratory.
Supplementary Material
Received: 3 March 2003; ; Returned for revision: 8 April 2003. Accepted: 6 May 2003
References
- AllanBB, Moyer BD, Balch WE.2000. Rab1 recruitment of p115 into a cis‐SNARE complex: programming budding COPII vesicles for fusion. Science 289: 444–448. [DOI] [PubMed] [Google Scholar]
- AndreevaAV, Kutuzov MA, Evans DE, Hawes CR.1998a. Proteins involved in membrane transport between the ER and the Golgi apparatus: 21 putative plant homologues revealed by dbEST searching. Cell Biology International 22: 145–160. [DOI] [PubMed] [Google Scholar]
- AndreevaAV, Kutuzov MA, Evans DE, Hawes CR.1998b. The structure and function of the Golgi apparatus: a hundred years of questions. Journal of Experimental Botany 49: 1281–1291. [Google Scholar]
- AndreevaAV, Zheng H, Saint‐Jore CM, Kutuzov MA, Evans DE, Hawes CR.2000. Organization of transport from endoplasmic reticulum to Golgi in higher plants. Biochemical Society Transactions 28: 505–512. [PubMed] [Google Scholar]
- BarloweC.2002. COPII‐dependent transport from the endoplasmic reticulum. Current Opinion in Cell Biology 14: 417–422. [DOI] [PubMed] [Google Scholar]
- Bar‐PeledM, Raikhel N.1997. Characterization of AtSEC12 and AtSAR1. Proteins likely involved in endoplasmic reticulum and Golgi transport. Plant Physiology 114: 315–324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- BarrieuF, Chrispeels MJ.1999. Delivery of a secreted soluble protein to the vacuole via a membrane anchor. Plant Physiology 120: 961–968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- BatokoH, Zheng HQ, Hawes C, Moore I.2000. A Rab1 GTPase is required for transport between the endoplasmic reticulum and Golgi apparatus and for normal Golgi movement in plants. The Plant Cell 12: 2201–2217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- BeckerB, Bölinger B, Melkonian M.1995. Anterograde transport of algal scales through the Golgi complex is not mediated by vesicles. Trends in Cell Biology 5: 305–307. [DOI] [PubMed] [Google Scholar]
- BednarekSY, Falbel TG.2002. Membrane trafficking during plant cytokinesis. Traffic 3: 621–629. [DOI] [PubMed] [Google Scholar]
- BoevinkP, Oparka K, Santa‐Cruz S, Martin B, Betteridge A, Hawes C.1998. Stacks on tracks: the plant Golgi apparatus traffics on an actin/ER network. The Plant Journal 15: 441–447. [DOI] [PubMed] [Google Scholar]
- BrandizziF, Fricker M, Hawes C.2002a. A greener world: the revolution in plant bioimaging. Nature Reviews Molecular Cell Biology 3: 520–530. [DOI] [PubMed] [Google Scholar]
- BrandizziF, Snapp EL, Roberts AG, Lippincott‐Schwartz J, Hawes C.2002b. Membrane protein transport between the endoplasmic reticulum and the Golgi in tobacco leaves is energy dependent but cytoskeleton independent: evidence from selective photobleaching. The Plant Cell 14: 1293–1309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- BrandizziF, Frangne N, Marc‐Martin S, Hawes C, Neuhaus JM, Paris N.2002c. The destination for single‐pass membrane proteins is influenced markedly by the length of the hydrophobic domain. The Plant Cell 14: 1077–1092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CheungAY, Chen CY‐H, Glaven RH, de Graaff BHJ, Vidali L, Hepler PK, Wu H‐M.2002. Rab2 GTPase regulates trafficking between the endoplasmic reticulum and the Golgi bodies and is important for pollen tube growth. The Plant Cell 14: 945–962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CrooksK, Coleman J, Hawes C.1999. The turnover of cell surface proteins of carrot protoplasts. Planta 208: 46–58. [Google Scholar]
- DeneckeJ, Botterman J, Deblaere R.1990. Protein secretion in plant cells can occur via a default pathway. The Plant Cell 2: 51–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DirnbergerD, Bencur P, Mach L, Steinkellner H.2002. The Golgi localization of Arabidopsis thaliana beta 1,2‐xylosyltransferase in plant cells is dependent on its cytoplasmic and transmembrane sequences. Plant Molecular Biology 50: 273–281. [DOI] [PubMed] [Google Scholar]
- EchardA, Opdam FJ, de Leeuw HJ, Jollivet F, Savelkoul P, Hendriks W, Voorberg J, Goud B, Fransen JA.2000. Alternative splicing of the human Rab6A gene generates two close but functionally different isoforms. Molecular Biology of the Cell 11: 3819–3833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- EsslD, Dirnberger D, Gomord V, Strasser R, Faye L, Glössl J, Steinkellner H.1999. The N‐terminal 77 amino acids from tobacco N‐acetylglucosoaminyltransferase I are sufficient to retain a reporter protein in the Golgi apparatus of Nicotiana benthamiana cells. FEBS Letters 453: 169–173. [DOI] [PubMed] [Google Scholar]
- FitchetteAC, Cabanes‐Macheteau M, Marvin L, Martin B, Satiat‐Jeunemaitre B, Gomord V, Crooks K, Lerouge P, Faye L, Hawes C.1999. Biosynthesis and immunolocalization of Lewis a‐containing N‐glycans in the plant cell. Plant Physiology 121: 333–344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- FowkeLC, Tanchak MA, Galway ME.1991. Ultrastructural cytology of the endocytotic pathway in plants. In: Hawes CR, Coleman JOD, Evans DE, eds. Endocytosis, exocytosis and vesicle traffic in plants. Society for Experimental Biology, Seminar Series 45 Cambridge: Cambridge University Press, 15–40. [Google Scholar]
- FrigerioL, de Virgilio M, Prada A, Faoro F, Vitale A.1998. Sorting of phaseolin to the vacuole is saturable and requires a short C‐terminal peptide. The Plant Cell 10: 1031–1042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GeldnerN, Friml J, Stierhof YD, Jürgens G, Palme K.2001. Auxin transport inhibitors block PIN1 cycling and vesicle trafficking. Nature 413: 425–428. [DOI] [PubMed] [Google Scholar]
- HadlingtonJL, Denecke J.2000. Sorting of soluble proteins in the secretory pathway of plants. Current Opinion in Plant Biology 3: 461–468. [DOI] [PubMed] [Google Scholar]
- HawesC, Satiat‐Jenuemaitre B.1996. Stacks of questions: how does the plant Golgi work? Trends in Plant Science 1: 395–401. [Google Scholar]
- HawesCR, Brandizzi F, Andreeva AV.1999. Endomembranes and vesicle trafficking. Current Opinion in Plant Biology 2: 454–461. [DOI] [PubMed] [Google Scholar]
- HillmerS, Freundt H, Robinson DG.1988. The partially coated reticulum and its relationship to the Golgi apparatus in higher plants. European Journal of Cell Biology 47: 206–212. [Google Scholar]
- HillmerS, Movafeghi A, Robinson DG, Hinz G.2001. Vacuolar storage proteins are sorted in the cis‐cisternae of the pea cotyledon Golgi apparatus. Journal of Cell Biology 152: 41–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HinzG, Hillmer S, Baumer M, Hohl I.1999. Vacuolar storage proteins and the putative vacuolar sorting receptor BP‐80 exit the Golgi apparatus of developing pea cotyledons in different transport vesicles. The Plant Cell 11: 1509–1524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HohlI, Robinson DG, Chrispeels M, Hinz G.1996. Transport of storage proteins to the vacuole is mediated by vesicles without a clathrin coat. Journal of Cell Science 109: 2539–2550. [DOI] [PubMed] [Google Scholar]
- HolsteinS.2002. Clathrin and endocytosis. Traffic 3: 614–620. [DOI] [PubMed] [Google Scholar]
- JiangLW, Rogers JC.1998. Integral membrane protein sorting to vacuoles in plant cells: Evidence for two pathways. Journal of Cell Biology 143: 1183–1199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- JuniperB, Hawes CR, Horne JC.1982. The relationship between dictyosomes and the forms of endoplasmic reticulum in plant cells with different export programs. Botanical Gazette 143: 135–145. [Google Scholar]
- JürgensG, Geldner N.2002. Protein secretion in plants: from the trans‐Golgi network to the outer space. Traffic 3: 605–613. [DOI] [PubMed] [Google Scholar]
- KlumpermanJ.2000. Transport between ER and Golgi. Current Opinion in Cell Biology 12:445–449. [DOI] [PubMed] [Google Scholar]
- LeeHI, Gal S, Newman TC, Raikhel NV.1993. The Arabidopsis endoplasmic reticulum retention receptor functions in yeast. Proceedings of the National Academy of Sciences of the USA 90: 11433–11437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LevineA.2002. Regulation of stress responses by intracellular vesicle trafficking? Plant Physiology and Biochemistry 40: 531–535. [Google Scholar]
- LewisMJ, Pelham HR.1990. A human homologue of the yeast HDEL receptor. Nature 348: 163–163. [DOI] [PubMed] [Google Scholar]
- LewisMJ, Sweet DJ, Pelham HRB.1990. The ERD2 gene determines the specificity of the luminal ER protein retention system. Cell 61: 1359–1363. [DOI] [PubMed] [Google Scholar]
- LiYB, Rogers SW, Tse YC, Lo SW, Sun SSM, Jauh GY, Jiang LW.2002. BP‐80 homologs are concentrated on post‐Golgi, probably lytic prevacuolar compartments. Plant and Cell Physiology 43: 726–742. [DOI] [PubMed] [Google Scholar]
- MitsuhashiN, Shimada T, Mano S, Nishimura M, Hara‐Nishimura I.2000. Characterization of organelles in the vacuolar‐sorting pathway by visualization with GFP in tobacco BY‐2 cells. Plant and Cell Physiology 41: 993–1001. [DOI] [PubMed] [Google Scholar]
- MitsuhashiN, Hayashi Y, Koumoto Y, Shimada T, Fukasawa‐Akada T, Nishimura M, Hara‐Nishimura I.2001. A novel membrane protein that is transported to protein storage vacuoles via precursor‐accumulating vesicles. The Plant Cell 13: 2361–2372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MoreauP, Hartmann MA, Perret AM, Sturbois‐Balcerzak B, Cassagne C.1998. Transport of sterols to the plasma membrane of leek seedlings. Plant Physiology 117: 931–937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MovafeghiA, Happel N, Pimpl P, Tai GH, Robinson DG.1999.Arabidopsis Sec21p and Sec23p homologs. Probable coat proteins of plant COP‐coated vesicles. Plant Physiology 119: 1437–1445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MunroS.1995. A comparison of the transmembrane domains of Golgi and plasma membrane proteins. Biochemical Society Transactions 23: 527–530. [DOI] [PubMed] [Google Scholar]
- NebenführA.2002. Vesicle traffic in the endomembrane system: a tale of COPs, Rabs and SNAREs. Current Opinion in Plant Biology 5: 507–512. [DOI] [PubMed] [Google Scholar]
- NebenführA, Staehelin, LA.2001. Mobile factories: Golgi dynamics in plant cells. Trends in Plant Science 6: 160–167. [DOI] [PubMed] [Google Scholar]
- NebenführA, Ritzenthaler C, Robinson DG.2002. Brefeldin A: deciphering an enigmatic inhibitor of secretion. Plant Physiology 130: 1102–1108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- NebenführA, Gallagher LA, Dunahay TG, Frohlick JA, Mazurkiewicz AM, Meehl JB, Staehelin LA.1999. Stop‐and‐go movements of plant Golgi stacks are mediated by the acto‐myosin system. Plant Physiology 121: 1127–1141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- OpatAS, Houghton F, Gleeson PA.2001. Steady‐state localization of a medial‐Golgi glycosyltransferase involves transit through the trans‐Golgi network. Biochemical Journal 358: 33–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- PagnyS, Bouissonnie F, Sarakar M, Follet‐Gueye ML, Driouich A, Schachter H, Faye L, Gomord V.2003. Structural requirements for Arabidopsis β 1,2‐xylosyltransferase activity and targeting to the Golgi. The Plant Journal 33: 189–203. [DOI] [PubMed] [Google Scholar]
- ParisN, Neuhaus JM.2002. BP‐80 as a vacuolar sorting receptor. Plant Molecular Biology 50: 903–914. [DOI] [PubMed] [Google Scholar]
- ParisN, Stanley CM, Jones RL, Rogers JC.1996. Plant cells contain two functionally distinct vacuolar compartments. Cell 85: 563–572. [DOI] [PubMed] [Google Scholar]
- PhillipsonBA, Pimpl P, Lamberti Pinto daSilva L, Crofts AJ, Tayler JP, Movafeghi A, Robinson DG, Denecke J.2001. Secretory bulk flow of soluble proteins is efficient and COPII dependent. The Plant Cell 13: 2005–2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- PimplP, Movafeghi A, Coughlan S, Denecke J, Hillmer S, Robinson DG.2000.In situ localization and in vitro induction of plant COPI‐coated vesicles. The Plant Cell 12: 2219–2235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- RitzenthalerC, Nebenführ A, Movafeghi A, Stussi‐Garaud C, Behnia L, Pimpl P, Staehelin LA, Robinson DG.2002. Reevaluation of the effects of Brefeldin A on plant cells using tobacco Bright Yellow 2 cells expressing Golgi‐targeted green fluorescent protein and COPI antisera. The Plant Cell 14: 237–261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- RutherfordS, Moore I.2002. The Arabidopsis Rab GTPase family: another enigma variation. Current Opinion in Plant Biology 5: 518–528. [DOI] [PubMed] [Google Scholar]
- Saint‐JoreCM, Evins J, Batoko H, Brandizzi F, Moore I, Hawes C.2002. Redistribution of membrane proteins between the Golgi apparatus and endoplasmic reticulum in plants is reversible and not dependent on cytoskeletal networks. The Plant Journal 29: 661–678. [DOI] [PubMed] [Google Scholar]
- SanderfootAA, Raikhel NV.1999. The specificity of vesicle trafficking: coat proteins and SNAREs. The Plant Cell 11: 629–641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SanderfootAA, Assaad FF, Raikhel NV.2000. The Arabidopsis genome. An abundance of soluble N‐ethylmaleimide‐sensitive factor adaptor protein receptors. Plant Physiology 124: 1558–1569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ShimadaT, Watanabe E, Tamura K, Hayashi Y, Nishimura M, Hara‐Nishimura I.2002. A vacuolar sorting receptor PV72 on the membrane of vesicles that accumulate precursors of seed storage proteins (PAC vesicles). Plant and Cell Physiology 43: 1086–1095. [DOI] [PubMed] [Google Scholar]
- SpangA.2002. ARF1 regulatory factors and COPI vesicle formation. Current Opinion in Cell Biology 14: 423–427. [DOI] [PubMed] [Google Scholar]
- StaehelinLA, Moore I.1995. The plant Golgi apparatus: structure, functional organization and trafficking mechanisms. Annual Review of Plant Physiology and Plant Molecular Biology 46: 261–288. [Google Scholar]
- StaehelinLA, Giddings TH Jr, Kiss JZ, Sack FD.1990. Macromolecular differentiation of Golgi stacks in root tips of Arabidopsis and Nicotiana seedlings as visualized in high pressure frozen and freeze‐substituted samples. Protoplasma 157: 75–91. [DOI] [PubMed] [Google Scholar]
- TakeuchiM, Ueda T, Yahara N, Nakano A.2002. Arf1 GTPase plays roles in the protein traffic between the endoplasmic reticulum and the Golgi apparatus in tobacco and Arabidopsis cultured cells. The Plant Journal 31: 499–515. [DOI] [PubMed] [Google Scholar]
- VitaleA, Denecke J.1999. The endoplasmic reticulum – gateway of the secretory pathway. The Plant Cell 11: 615–628. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.