Abstract
Methylphenidate (MPD) is a widely prescribed psychostimulants used for the treatment of attention deficit hyperactive disorder (ADHD). Unlike the psychostimulants cocaine and amphetamine, MPD does not exhibit direct actions on the serotonin transporter, however there is evidence suggesting that the therapeutic effects of MPD may be mediated in part by alterations in serotonin transmission. This study aimed to investigate the role of the dorsal raphe (DR) nucleus, one of the major sources of serotonergic innervation in the mammalian brain, in the response to MPD exposure. Freely behaving adolescent rats previously implanted bilaterally with permanent electrodes were used. An open field assay and a wireless neuronal recording system were used to concomitantly record behavioral and DR electrophysiological activity following acute and chronic MPD exposure. Four groups were used: one control (saline) and three experimental groups treated with 0.6, 2.5, and 10.0 mg/kg MPD respectively. Animals received daily MPD or saline injections on experimental days 1–6, followed by 3 washout days and MPD rechallenge dose on experimental day (ED)10. The same chronic dose of MPD resulted in either behavioral sensitization or tolerance, and we found that neuronal activity recorded from the DR neuronal units of rats expressing behavioral sensitization to chronic MPD exposure responded significantly differently to MPD rechallenge on ED10 compared to the DR unit activity recorded from animals that expressed behavioral tolerance. This correlation between behavioral response and DR neuronal activity following chronic MPD exposure provides evidence that the DR is involved in the acute effects as well as the chronic effects of MPD in adolescent rats.
Keywords: methylphenidate, dorsal raphe, neuronal activity, behavior, adolescent, rats
Introduction
Attention deficit hyperactive disorder (ADHD) is a neuropsychiatric disorder characterized by increased impulsivity, hyperactivity, and inattentiveness (Newcorn, 2000), although precise neurophysiologic deficits underlying ADHD remains undetermined (Krain and Castellanos, 2006; Dalley and Roiser, 2012). ADHD is commonly diagnosed during adolescence and management with psychostimulants often continues into adulthood (Arnsten, 2006; Homberg et al., 2016; Polanczyk and Rohde, 2007; Wilens, 2008). Methylphenidate (MPD) is the most widely prescribed stimulant in children and is the drug of choice for treatment of ADHD (Accardo and Blondis, 2001; Bolaños et al., 2003; Volkow et al., 1995). Adolescent and adult rats differ in both acute and long term responses to psychostimulants, and it was reported that chronic MPD exposure during development can modify long term behavioral responses to emotional stimuli, resulting in depression-like behavior in adult rats (Andersen et al., 2002; Bolaños et al., 2003; Bolaños et al., 2008). However, despite extensive use of MPD by children when the developing brain is still going through profound changes, the long term effects of chronic MPD administration on brain ontogenesis are poorly understood (Bolaños et al., 2003; Koda et al., 2010).
Research on MPD has focused primarily on its effects on dopamine and norepinephrine transmission (Arnsten and Dudley, 2005; Berridge et al., 2006). However, the role of 5HT in ADHD remains ambiguous (Oades, 2008). Behavioral and neurochemical studies (Kuczenski and Segal, 1997; Volkow et al., 2000) indicate that MPD affects the serotonergic system. Serotonin is a key modulatory neurotransmitters in the CNS and has been implicated in the regulation of behavioral function (Cools et al., 2008). A major source of 5-HT in the brain is the serotonergic neurons of the dorsal raphe nucleus (Tao and Auerbach, 1995; Miyazaki et al., 2012). 5HT exhibits complex interactions with the other catecholamines, including selective regulation of DA release (Porras et al., 2002) and modulation of DR serotonergic neurons has been shown to result in increased action impulsivity and failure to wait for delayed rewards (Miyazaki et al., 2012) which warrant further investigation of its relationship with DA and NE and its role in ADHD (Gatley et al., 1999).
Repetitive exposure to psychostimulants has been shown to elicit dose dependent behavioral tolerance or sensitization (Lee et al., 2014; Frolov et al., 2015) and it is this behavioral outcome that predicts drug abuse liability (Robinson and Berridge, 2001). Behavioral sensitization or tolerance is a phenomenon characterized by a further increased or decreased response to repeated drug exposure compared to the initial (acute) effect respectively (Claussen et al., 2014; Jones et al., 2014). At therapeutic doses, MPD is known to cause robust regulatory changes in dopaminergic output of the sensorimotor striatum; a region heavily involved in learning and habit formation (Yano and Steiner, 2005). Studies suggest that 5HT transmission is involved in the modification of gene regulation by psychostimulants and in the formation of addictive behaviors (Bhat and Baraban, 1993; Andersen et al., 2002; Van Waes et al., 2010). Paradoxically, chronic exposure to identical doses of MPD has been shown to elicit behavioral sensitization in some animals and tolerance in others (Frolov et al., 2015; Jones et al., 2014) however the reason for this dichotomy in adult rats is unclear.
The aim of this study is to further investigate the effects of acute and repetitive (chronic) MPD on both behavior and DR neuronal events in adolescent rats by concomitantly recording the behavioral and neuronal activity of DR neurons before and after acute and chronic exposure to MPD. The hypothesis of the study is 1) the dorsal raphe participates in MPD action, 2) the same MPD dose in some adolescent animals will elicit behavioral sensitization and in others behavioral tolerance and 3) the DR neuronal activity recorded from adolescent animals expressing behavioral sensitization will have a significantly different response to MPD exposure than those DR neurons recorded from animals expressing behavioral tolerance. Therefore, freely behaving animals previously implanted with permanent electrodes within the DR and MPD dose response protocol were used to concomitantly record the behavioral and the DR neuronal activity following acute and repetitive MPD injection.
2. Materials and Methods
2.1 Animals
One hundred and sixty one (161) Male Sprague-Dawley rats at about post-natal day 30 (P- 30) were purchased (Harlan, Indianapolis, IN, USA) and allowed 4–5 days of acclimation in our vivarium on a 12 hour light/dark schedule (lights on 6:00am) prior to electrode implantation, while temperature (21 ± 2°C) and humidity (37–45%) were kept constant. Food and water were given ad libitum. After electrode implantation, the animals were housed individually in clear acrylic standard cages that served as both home cage and test cage for the electrophysiological and behavioral recordings for an additional 5 to 6 days prior to the recording (experimental) days. Recording began on P-40 following saline (control) and MPD exposure. The experimental procedures were approved by the UTHSC Animal Welfare Committee and carried out in accordance with the National Institute of Health Guide for Care and Use of Laboratory Animals.
2.2 Surgery
Animals were anesthetized with an intraperitoneal (i.p.) injection of 30mg /kg pentobarbital prior to surgery. The animal’s head was shaved and a lidocaine hydrochloride gel was applied for local anesthetic. The animal was then placed in a stereotaxic apparatus and an incision was made to expose the skull. Bilateral holes were drilled above the DR at 7.8mm posterior to bregma and .2mm lateral to the midline using the Sherwood and Timiras adolescent rat brain atlas(Sherwood and Timiras, 1970). An additional hole was drilled in the frontal sinus for the reference electrode, and six anchor screws were inserted at vacant spots in the skull. Two Nickel-Chromium Teflon coated (insulated except at tips, resistance approx. 80 mΩ) wires 60μm in diameter were twisted and each was secured to a 1 cm copper connector pin and individually inserted into the DR on each side to be used as recording electrodes (four total recording electrodes). During the electrode placement, the DR unit activity was monitored using a Grass P511 amplifier with its cathode follower connected to an audiomonitor and oscilloscope. The electrodes were inserted at a depth of 6mm and, if satisfactory neuronal activity was observed, they were secured to the anchor screws and to the skull by dental acrylic cement. Electrodes failing to detect satisfactory activity were lowered in steps of 5 to 10 μm increments until a 3:1 ratio spike activity was detected, to a maximum depth of 6.6 mm below the skull, and then secured with dental acrylic cement. Rats were allowed to recover from the surgical procedure for 5 to 6 days. During this recovery period, the animals were placed with their home cage in the experimental room for about two hours each day and connected to the wireless head stage (Triangle BioSystems Inc., TBSI, Durham, NC, USA) to adapt and acclimate to the neuronal and behavioral recording systems.
2.3 Drugs
Methylphenidate hydrochloride (MPD) was obtained from NIDA. Previous dose response MPD experiments (from 0.1 to 40.0 mg/kg i.p.) found that behavioral effects of MPD were observed from 0.6mg/kg doses of MPD and higher (Gaytan et al., 1996; Algahim et al., 2009; Yang et al., 2011) therefore 0.6, 2.5, and 10.0 mg/kg doses were selected as low, moderate, and high experimental doses. MPD was dissolved in a 0.9% isotonic saline solution for injection. Control subjects received injections of 0.8ml isotonic saline solution (0.9% NaCl). All MPD injections were titrated to a volume of 0.8ml with 0.9% saline to equalize injection volumes for all animals. Injections were given i.p.
2.4 Experimental protocol and data acquisition
The rats were allowed to recover from electrode implantation and adapt to the behavioral and electrophysiological recording systems prior to recording for 5 to 6 days. On experimental day 1 (ED1), rats within their home cage were placed again within a Faraday cage to reduce background noise during recording sessions. The animals were allowed an additional 20 to 30 minutes for adaptation to the wireless head stage, during which the neuronal activity was monitored and the software parameters were selected for spike recording and sorting. The DR neuronal activity and the locomotive activity were recorded simultaneously using a wireless neuronal recording system (Triangle BioSystems Inc., TBSI, Durham, NC, USA) and an open field computerized animal activity system (Accuscan, Columbus, OH, USA). The TBSI wireless electrophysiological recording system consists of a head stage (weighing less than 5g) and a remote receiver that was connected to an analog- to-digital converter (Micro1401-3; Cambridge Electronic Design (CED)) that digitized and stored the data on a PC using the CED Spike 2.7 software.
After 20 to 30 minutes of acclimatization to the recording apparatus, the rats were injected with 0.8ml saline and the baseline neuronal and behavioral activity was recorded concomitantly for one hour followed by a second injection of either saline or 0.6, 2.5, or 10.0 mg/kg MPD and an additional one hour of recording following the second injection was obtained (Table 1). On ED2 through ED6 animals received either saline or MPD injections in their home cage without behavioral or neuronal recordings. On ED7 through ED9, no injections were given and the animals underwent a 3 day washout period. On ED10 after 20–30min of acclimation to the experimental system, animals were administered 0.8ml saline and baseline neuronal and behavioral activity were recorded for one hour, followed by a rechallenge administration of saline or a 0.6, 2.5, 10.0 mg/kg dose of MPD and an additional hour of recording as on ED (Claussen et al., 2014; Jones et al., 2014; Tang and Dafny, 2013).
Table 1. Experimental method.
Table 1 summarizes the experimental protocol.
| Group | ED1* | ED 2-6 | ED 7-9 | ED10* |
|---|---|---|---|---|
|
| ||||
| Saline (control) | saline/saline | saline | washout | saline/saline |
| 0.6 mg/kg | saline/0.6 | 0.6 | washout | saline/0.6 |
| 2.5 mg/kg | saline/2.5 | 2.5 | washout | saline/2.5 |
| 10.0 mg/kg | saline/10.0 | 10.0 | washout | saline/10.0 |
denotes days on which animal behavioral activity and dorsal raphe neuronal activity were recorded for 60 minutes following i.p. saline injection and for an additional 60 minutes following i.p. injection of either saline or 0.6, 2.5, or 10.0 mg/kg methylphenidate (MPD).
On experimental days 2–6 (ED 2–6) animals received only the injection of either saline the experimental dose. On ED 7–9 animals underwent washout and received no injections or recordings. On ED10, a rechallenge saline or MPD dose was administered and behavioral and neuronal recordings resumed following the same protocol as on ED1. All injections were titrated to 0.8ml with 0.9% saline and given in the morning.
2.5 Behavioral data
2.5.1 Locomotor activity recording
Locomotor activity was recorded using an open field computerized animal activity system (Accuscan, Columbus, Ohio) consisting of a 40 x 40 x 32cm cage with 16 x 16 infrared beams and sensors located 5cm above the floor of the cage. This cage was used as both home and test cage to eliminate environmental stimulation. Interruption of the infrared beams by animal movement was detected by the monitoring system at 100Hz frequency and the interruptions compiled by the Oasis software (Accuscan, Columbus, OH, USA) and downloaded to a PC every 10 minutes. The interrupted beams were categorized by the software into three different locomotor behaviors including: horizontal activity (HA), total distance (TD) traveled in cm, and number of stereotypic movements (NOS), which is a count of repetitive purposeless movement with at least one second intervals between movements. These data were recorded for 60 minutes following saline or MPD injection at ED1 and ED10 and evaluated to distinguish animals that expressed behavioral sensitization following repetitive (chronic) MPD exposure from those that expressed behavioral tolerance.
2.5.2 Behavioral Data Analysis
The data was used to make 3 comparisons: 1) locomotor activity after MPD administration on experimental day 1 (ED1) was compared to ED1 baseline (post-saline) activity to determine the acute effects of MPD; 2) locomotor activity after saline injection on ED10 (baseline) was compared to activity after saline injection on ED1 to observe whether the locomotor activity following 6 daily MPD doses (ED1-ED6) and three washout days (ED7- ED9) exhibited baseline changes (Table 1); and 3) locomotor activity after MPD administration on ED10 was compared to activity recording following the initial MPD treatment on ED1 to detect whether the animal displayed behavioral sensitization or tolerance to MPD. Animals exhibiting a significant increase in their locomotor activity in response to MPD rechallenge on ED10 compared to the acute effect of MPD on ED1 were considered to express behavioral sensitization, and those animals displaying a decrease in their locomotor activity following MPD rechallenge on ED10 compared to post-MPD activity on ED1 were considered to exhibit behavioral tolerance to chronic MPD exposure. The student t test and the Critical Ratio (CR) test was used to determine the effect on MPD on individual animals.
For the acute effect, E represents activity count after MPD injection on ED1 and C the activity after saline (control) injection. For the chronic effect, E represents activity following MPD injection on ED10 and C is the activity after MPD dose on ED1. Individual rats were categorized as exhibiting either behavioral sensitization or tolerance and sorted based on this classification into 2 groups: the sensitized subgroup or the tolerant subgroup. For each comparison, the subgroups were evaluated as a group using analysis of variance (ANOVA) test with repeated measure and statistical significance set at p < 0.05.
2.6 Neuronal data
2.6.1 Data acquisition
The neuronal recordings used the Triangle Biosystem Inc. (TBSI) Durham NC USA wireless system. The TBSI head stage connected to the rat head cap.
2.6.2. Spike Sorting
CED Spike 2.7 software was used for spike sorting and statistical analysis of the sorted neuronal activity. The data are captured by the program and processed using low and high pass filters (.3–3 kHz). There are two window discriminator levels, one for positive-going spikes and one for negative-going spikes. Only spikes falling within the previously determined window and exhibiting spike durations of 0.8–1.2ms were extracted. One of the spikes was then selected to create a template using 1000 waveform data points that the program can use as high-dimensional reference points for accurate and reliable spike sorting (Figure 1), despite minor influence of background noise, false threshold crossing, and waveform overlap. Incoming spikes are compared with all temporal templates to find the best fitting template that yields the minimum residue variance. When the distance between the spike waveform and the template exceeds threshold (80%), the waveforms are rejected. This means that the spike sorting accuracy in the reconstructed data was approximately 95%. The same parameters and template used to sort and count ED1 neuronal activity were loaded onto the ED10 file of the same electrode to sort and count the ED10 activity, ensuring that the waveform sorting parameters used on ED1 and ED10 were identical.
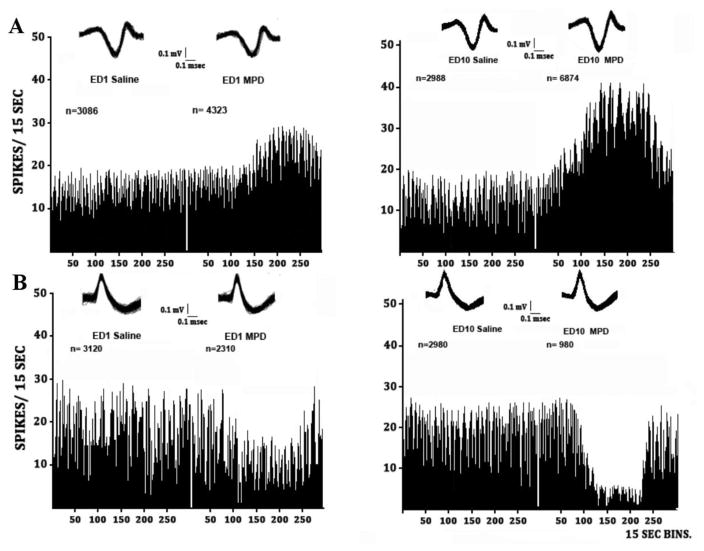
Figure 1. DR neuronal activity histogram.
Figure 1 is a series of sequential frequency histograms of the neuronal activity of two dorsal raphe (DR) units recorded after saline injection (baseline) and after 2.5 mg/kg methylphenidate (MPD) exposure on experimental day 1 (ED1) and ED10. A is a representative DR unit in which the initial (acute) MPD exposure elicited significant (p < 0.05) excitation, and B represents a unit that responded with attenuation when comparing the total neuronal activity post MPD exposure (ED1 MPD) to ED1 baseline (ED1 saline). On the right side are the recordings obtained at ED10 after 6 daily doses of 2.5 mg/kg MPD and three washout days before (ED10 saline) and following 2.5 mg/kg MPD rechallenge (ED10 MPD). In both DR units the ED10 saline (baseline) exhibit similar activity to ED1 saline. On ED10 following MPD challenge unit A exhibits further significant (p < 0.05) acceleration following 2.5 mg/kg MPD compared to the initial effect of MPD at ED1, while unit B exhibited the opposite effect. At ED1 the initial effect of MPD was attenuation compared to ED1 saline (baseline) and MPD at ED10 elicits further significant (p < 0.05) attenuation of this DR firing rate compared to the initial effect of MPD on ED1. The insert at the top of each frequency histograms is 20 superimposed spikes showing that the same spike pattern and amplitude was counted during all the experimental sessions. n= is the total number of spikes recorded in a 60 minute session on ED1 and ED10 following saline and 2.5 mg/kg exposure respectively.
2.6.3 Electrophysiological Data Analysis
The sorted spike activity was counted and used to calculate the neuronal firing rates before and following treatment and to produce sequential firing rate graphs. Several comparisons were made using these data: 1) ED1 baseline firing rates (first 60 minute recording) following initial saline injection were compared to activity after MPD or saline administration (second 60 minute recording) on ED1 to assess the acute effect on drug exposure on DR unit activity; 2) ED10 baseline activity was compared to ED1 baseline activity to evaluate whether of 6 daily doses of MPD followed by three washout days modify the DR baseline activity on ED10; and 3) neuronal activity following MPD injection on ED10 was compared to post-MPD activity on ED1 to assess the effects of chronic MPD exposure on DR activity. Significant change in neuronal firing rate and direction of change (decrease or increase) for each DR unit was determined using the Critical Ratio (CR) test. C.R. value above +1.96 for an individual unit indicated a significant increase in firing activity after MPD exposure while a value below −1.96 indicated a significant decrease in activity.
After collection of behavioral data, the electrophysiological data was sorted into three subgroups based on the animal’s behavioral response to MPD: 1) DR neuronal activity recordings from all animals; 2) DR neuronal activity recorded from those animals exhibiting behavioral sensitization to chronic MPD exposure; 3) DR neuronal activity recorded from animals exhibiting behavioral tolerance to chronic MPD exposure. The significance of differences in the firing rates of DR units between these subgroups was analyzed using the Chi-square test where p < 0.05 was considered as significant.
2.7 Histological verification of electrode placement
An overdose of phenobarbital was administered upon completion of recording on ED10. The animals were then perfused intracardially with 10% formaldehyde solution containing 3% potassium ferrocyanide and a 50μA current was passed through the electrode to produce a small lesion at the recording site. The brain was removed and preserved in 10% formalin for histological processing. The position of the electrodes in the DR was confirmed by the location of the lesion and the Prussian blue spot using the Adolescent Rat Brain Atlas (Sherwood and Timiras, 1970). Only data from electrodes confirmed to be within the DR and exhibiting similar spike amplitude and pattern at the ED1 and ED10 recording sessions was included in data evaluation.
3. Results
3.1 Locomotor Behavior
One hundred and sixty one animals were used: 15, 49, 51, and 46 animals received acute and repetitive (chronic) saline, 0.6, 2.5, and 10.0 mg/kg MPD respectively.
3.1.1 Control
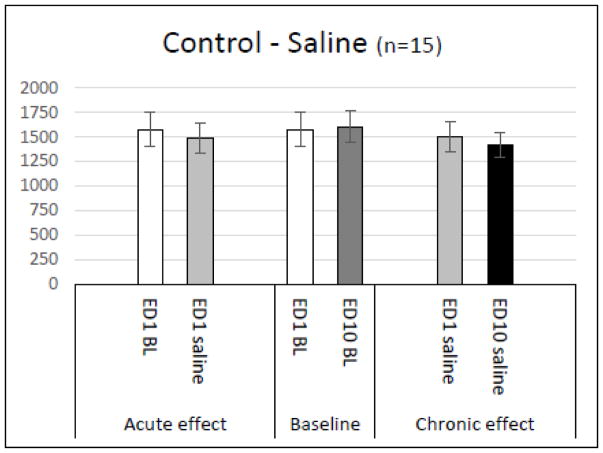
In order to determine the effects of injection volume and handling on locomotor activity, fifteen animals received saline injections on ED1-ED6 and ED10 (Table 1 and Figure 2). Only minor, non-significant fluctuations were noted in TD, HA, and NOS following acute and repetitive (chronic) saline administration, indicating that locomotor behavior was not significantly affected by the injection process, animal handling time, or laboratory environment. Since the activity following the first saline injection was similar to all the other recordings, the activity following the first saline injection can be used as a control for the drug groups. Thus any significant changes in locomotor activity are presumably due to the effects of MPD exposure (Figure 2). Since TD, HA, and NOS exhibit similar activity only TD is shown.
Figure 2. Behavioral recordings from control group.
Summarizes acute and chronic effects of 8ml 0.9% NaCl (control) injection on horizontal locomotor activity (n = 15). No significant changes in activity were observed following the first or second saline injections on ED1 and ED10.
3.1.2 Behavioral response to acute and repetitive 0.6 mg/kg MPD
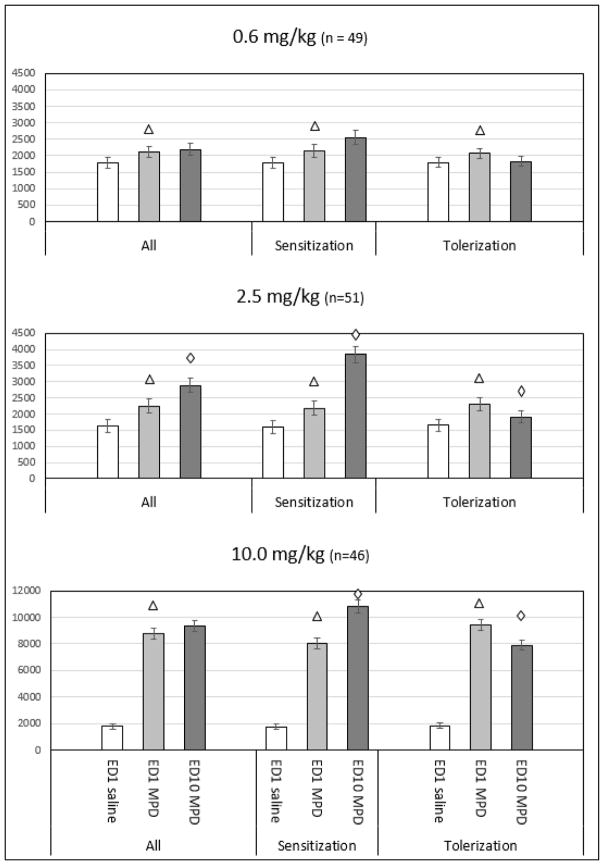
Forty nine animals received acute and chronic injections of 0.6 mg/kg MPD (Figure 3). When analyzed as a whole, this group exhibited a significant (p < 0.001) increase in locomotor activity following acute MPD but did not exhibit significant changes in baseline activity or in activity following MPD on ED10 compared to ED1. However, when the animals were sorted into subgroups based on their individual behavioral response to MPD on ED10 compared to ED1 (i.e. sensitization or tolerance) using the CR test and student t-test, 32 animals displayed both a significant (p < 0.001) increase in locomotor activity following acute MPD administration as well as a significant (p < 0.001) increase in locomotor activity following MPD rechallenge on ED10 compared to activity post ED1 injection, i.e. these animals expressed significant behavioral sensitization. The individuals animals in the subgroup that exhibited behavioral tolerance (n = 17) to repetitive MPD doses displayed a significant (p = 0.004) increase in activity following acute MPD dose and no change or significant decrease (p = 0.008) in locomotor activity after MPD rechallenge compared to activity post ED1 injection determined using the Tukey test. Baseline locomotor activity on ED10 compared to baseline activity on ED1 experienced only minor, non-significant fluctuations in both sensitized and tolerized groups receiving 0.6 mg/kg MPD.
Figure 3. Behavioral recordings following acute and chronic 0.6, 2.5, and 10.0 mg/kg MPD.
Figure 3 summarizes the behavioral activity of the experimental groups in response to acute and chronic 0.6, 2.5, or 10.0 mg/kg MPD. For each dose, the leftmost histogram summarizes the horizontal locomotor activity (HA) of all animals as one group. The middle histogram summarizes the HA of the individual animals that express behavioral sensitization and the rightmost histogram summarizes the individual animals that expressed behavioral tolerance. ED1 saline–the activity at baseline (ED1 BL); ED1 MPD–HA following MPD administration on ED1; ED10 MPD–the activity following rechallenge MPD administration. indicates significant (p < 0.05) change in activity following acute MPD administration on ED1 compared to ED1 baseline (saline) activity. indicates a significant (p < 0.05) difference in the effect of initial MPD exposure on locomotor activity at ED10 compared to activity following acute MPD administration on ED1.
3.1.3 Behavioral response to acute and repetitive 2.5 mg/kg MPD
Fifty one rats were treated with acute and chronic 2.5 mg/kg MPD (Figure 3). Overall, MPD exposure at ED1 resulted in a significant (p < 0.001) increase in locomotor activity compared to baseline. On ED10, animals exhibited a significant (p < 0.001) increase in activity following rechallenge MPD compared to the response on ED1. When these 51 animals were separated based on individual behavioral response using the C.R test, 32 rats were observed to exhibit behavioral sensitization and 19 exhibited behavioral tolerance. The sensitized subgroup displayed a significant (p < 0.001) increase in locomotor activity following acute MPD dose on ED1 and a significant (p < 0.001) further increase in activity following rechallenge dose on ED10 compared to the initial increase following treatment on ED1 using the Tukey test. The group exhibiting tolerance had a significant (p < 0.001) increase in locomotor activity following acute MPD dose on ED1, but exhibited a significant (p < 0.001) decrease in activity following rechallenge MPD dose on ED10 compared to ED1. In both groups, baseline locomotor activity on ED10 did not experience a significant change compared to ED1 baseline activity.
3.1.4 Behavioral response to acute and repetitive 10.0 mg/kg MPD
Forty six animals were given chronic and acute injections of 10.0 mg/kg MPD (Figure 3). This group displayed a significant (p < 0.001) increase in locomotor activity following acute MPD dose and a significant (p < 0.001) further increase in activity following rechallenge MPD dose at ED10 compared to activity following acute injection on ED1. Animals exhibiting behavioral sensitization (N = 23) responded to acute MPD injection on ED1 with a significant (p < 0.001) increase in locomotor activity. On ED10, baseline activity for this group was significantly (p < 0.001) increased compared to ED1 baseline activity, and rechallenge MPD administration elicited a significant (p < 0.001) further increase in activity as compared to activity following acute injection on ED1 using the Tukey test. The individual rats displaying behavioral tolerance (N =23) to 10.0 mg/kg showed a significant (p < 0.001) increase in behavioral activity following acute MPD dose and a small but significant (p < 0.001) increase in baseline activity on ED10 compared to ED1 using the Tukey test. However, following rechallenge MPD injection on ED10, this subgroup displayed a significant (p < 0.001) decrease in locomotor activity compared to response elicited by acute injection on ED1, i.e. they expressed behavioral tolerance.
3.2 Dorsal Raphe (DR) neuronal activity
3.2.1 Control
A total of 479 units were histologically confirmed to be recorded from the DR and exhibited similar amplitude and wave form spike at ED1 and ED10. Fifty seven units were recorded from control rats following acute (ED1) and repetitive (ED10) saline injection. Of these 57 units, 3 showed increased firing activity following the 2nd saline injection on ED1 and 2 showed decreased activity. The baseline activity of 1 DR unit on ED10 compared to ED1 showed an increase in its firing rate, and 1 DR unit showed a decrease in baseline activity on ED10 compared to ED1. Following the ED10 rechallenge saline injection, 2 DR units exhibited an increase in activity and 1 exhibited a decrease. These observations indicate that the alterations in DR activity were random and that the saline injections and animal handling did not have a significant effect on the neuronal activity of the DR.
3.2.2 All Animals
Four hundred and twenty two DR units were recorded from 146 rats following 0.6 (49 rats), 2.5 (51 rats), and 10.0 mg/kg (46 rats) MPD injection. Sixty-four percent (268/422) of the total units recorded following acute (ED1) MPD exposure exhibited a significant (p < 0.05) change firing activity compared to ED1 baseline levels, with 57% (153/268) of these responsive units exhibiting an increase in activity following MPD injection (Figure 4) (Table 2). After 6 daily injections and 3 washout days, 69% (292/422) of the recorded DR units exhibited a significant (p < 0.05) change in their baseline activity recorded on ED10 compared to ED1, with 52% (153/292) of these units exhibiting an increase in baseline activity prior to MPD injection on ED10. Upon rechallenge of MPD on ED10, 67% (283/422) units responded with a change in firing rate, with 52% (147/283) of these units exhibiting a decrease in firing activity compared to the acute response to MPD on ED1. (Table 2)
Figure 4. ED1 neuronal recordings following saline and 2.5 mg/kg MPD.
Figure 4 is two tracings of raw analog data recorded from DR units before (upper tracing) and after (lower tracing) 2.5 mg/kg MPD on ED1. Recordings were obtained 17 minutes post saline and MPD injection.
Table 2. summarizes the DR neuronal activity of all animals.
Table 2 summarizes the acute and chronic effect of MPD injection on DR neuronal activity from all experimental groups (0.6, 2.5, and 10.0 mg/kg MPD).
 indicates a significant (p < 0.05) increase in activity following MPD treatment,
indicates a significant (p < 0.05) increase in activity following MPD treatment,
 indicates a significant (p < 0.05) decrease in neuronal activity following treatment, and
indicates a significant (p < 0.05) decrease in neuronal activity following treatment, and
 indicates no change. Acute summarizes the acute effect of MPD; Baseline compares neuronal activity following rechallenge MPD exposure to the initial effect of MPD at ED1.
indicates no change. Acute summarizes the acute effect of MPD; Baseline compares neuronal activity following rechallenge MPD exposure to the initial effect of MPD at ED1.
| Population | Response direction | Acute ED1 | Baseline ED10 to ED1 | Rechallenge ED10 |
|---|---|---|---|---|
| All Animals N = 422 |
|
153 (36.2%) | 153 (36.3%) | 136 (32.2%) |
|
|
115 (27.3%) | 139 (32.9%) | 147 (34.8%) | |
|
|
154 (36.5%) | 130 (30.8%) | 139 (32.9%) |
3.2.3 Effect of 0.6 mg/kg MPD on DR units of all animals (Table 3a)
Table 3. summarizes the DR neuronal activity following 0.6 mg/kg MPD.
Table 3 summarizes the effects of acute and chronic injection of 0.6 mg/kg MPD on DR neuronal activity. Section a. summarizes neuronal activity of DR units recorded from all rats, section b. summarizes activity recorded only from animals exhibiting behavioral sensitization, and section c. summarizes activity recorded only from animals expressing behavioral tolerance respectively.
 indicates a significant (p < 0.05) increase in activity following MPD treatment,
indicates a significant (p < 0.05) increase in activity following MPD treatment,
 indicates a significant (p < 0.05) decrease in activity following treatment, and
indicates a significant (p < 0.05) decrease in activity following treatment, and
 indicates no change.
indicates no change.
| Dose | Population | Response direction | Acute ED1 | Baseline ED10 to ED1 | Rechallenge ED10 |
|---|---|---|---|---|---|
| 0.6 mg/kg | |||||
| a. Total N = 127 |
|
37 (29.1%) | 32 (25.1%) | 23 (18.1%) | |
|
|
24 (18.9%) | 31 (22.4%) | 39 (30.7%) | ||
|
|
66 (52.0%) | 64 (50.4%) | 65 (51.2%) | ||
| b. Behavioral Sensitization N = 61 |
|
28 (45.9%) | 39 (63.9%) | 33 (54.1%) | |
|
|
26 (42.6%) | 19 (31.1%) | 22 (36.1%) | ||
|
|
7 (11.5%) | 3 (4.9%) | 6 (9.8%) | ||
| c. Behavioral Tolerance N = 66 |
|
14 (21.2%) | 13 (19.7%) | 10 (15.2%) | |
|
|
13 (19.7%) | 17 (25.8%) | 27 (40.9%) | ||
|
|
39 (59.1%) | 36 (54.5%) | 29 (43.9%) | ||
Neuronal activity following injection of 0.6 mg/kg MPD was recorded in 127 DR units (Table 3a). Of these units, 48% (61/127) responded to acute injection with a significant (p < 0.05) change in their neuronal activity with a majority (37/61) of these responding units exhibiting a significant (p < 0.05) increase in firing rate. Comparing the baseline neuronal activity on ED10 to that of ED1 showed that 49% (63/127) of the DR units had altered baseline firing activity following 6 daily doses of 0.6 mg/kg MPD and 3 washout days, with a fairly even split between the units that expressed an increase (51%, 32/63) and those that expressed a decrease (49%, 31/63) in activity. Following MPD rechallenge on ED10, 49% (62/127) of the DR units displayed a significant (p < 0.05) change in activity compared to post-MPD on ED1, with a decrease in firing activity observed in the majority (63%, 39/62) of the DR units. (Table 3a)
3.2.4 DR neuronal activity recorded from animals expressing behavioral sensitization to 0.6 mg/kg (Table 3b)
Sixty one units were recorded from animals that expressed behavioral sensitization in response to chronic exposure to 0.6 mg/kg MPD (Table 3b). Of these units, 89% (54/61) responded to acute MPD administration on ED1 with a significant (p < 0.05) change in firing activity, with 52% (28/54) of them experiencing an increase in firing rate. Ninety five percent (58/61) of the units exhibited a significant (p < 0.05) change in baseline activity comparing ED10 baseline to ED1 baseline, with a majority (67%, 39/58) displaying an increase in baseline activity on ED10 compared to ED1. Following ED10 MPD rechallenge, 90% (55/61) of the DR units had a significant response to MPD administration, with an increase in activity reported for the majority (60%, 33/55) of the responsive units. (Table 3b)
3.2.5 DR activity recorded from animals expressing behavioral tolerance to 0.6 mg/kg (Table 3c)
Sixty six DR units were recorded from animals exhibiting behavioral tolerance (Table 3c). Acute injection of MPD resulted in a significant (p < 0.05) change in their neuronal activity in 41% (27/66) of the units, with the majority (52%, 14/27) of them displaying an increase and 48% (13/27) displaying a decrease in firing activity. Significant (p < 0.05) changes in ED10 baseline activity prior to MPD rechallenge administration on ED10 compared to ED1 were noted for 45% (30/66) of the units, and 57% (17/30) of the responsive exhibited a decrease in baseline firing activity. Following ED10 MPD rechallenge, 56% (37/66) of the DR units displayed a significant (p < 0.05) response to MPD exposure by changing their firing rate, with a majority of the units (73%, 27/37) displaying a decrease in their neuronal activity in response to the drug. (Table 3c)
3.2.6 Effect of 2.5 mg/kg MPD on DR units of all animals (Table 4a)
Table 4. summarizes the DR neuronal activity following 2.5 mg/kg MPD.
Table 4 summarizes the effects of acute and chronic injection of 2.5 mg/kg MPD on DR neuronal activity. Section a. summarizes neuronal activity of DR units recorded from all rats, section b. summarizes activity recorded only from animals exhibiting behavioral sensitization, and section c. summarizes activity recorded only from tolerant animals.
 indicates a significant (p < 0.05) increase in activity following MPD treatment,
indicates a significant (p < 0.05) increase in activity following MPD treatment,
 indicates a significant (p < 0.05) decrease in activity following treatment, and
indicates a significant (p < 0.05) decrease in activity following treatment, and
 indicates no change.
indicates no change.
| Dose | Population | Response direction | Acute ED1 | Baseline ED10 to ED1 | Rechallenge ED10 |
|---|---|---|---|---|---|
| 2.5 mg/kg | |||||
| a. Total N = 142 |
|
49 (34.5%) | 53 (37.3%) | 48 (33.8%) | |
|
|
36 (25.4%) | 45 (31.7%) | 48 (33.8%) | ||
|
|
57 (40.1%) | 44 (31.0%) | 46 (32.4%) | ||
| b. Behavioral Sensitization N = 62 |
|
44 (71.0%) | 49 (79.0%) | 48 (77.4%) | |
|
|
11 (17.7%) | 9 (14.5%) | 11 (17.7%) | ||
|
|
7 (11.3%) | 4 (6.5%) | 3 (4.8%) | ||
| c. Behavioral Tolerance N = 80 |
|
19 (23.8%) | 14 (17.5%) | 12 (15.0%) | |
|
|
22 (27.5%) | 36 (45.0%) | 37 (46.3%) | ||
|
|
39 (48.8%) | 30 (37.5%) | 31 (38.8%) |
The neuronal activity of 142 DR units was recorded following injection of 2.5 mg/kg MPD (Table 4a). Acute MPD injection resulted in a change in firing rate for 59.9% (85/142) of these units, with 57.6% (49/85) of the responsive units showing an increase in their firing rate activity. Following 6 daily doses of MPD and 3 washout days, 69% (98/142) of the DR units exhibited a significant (p < 0.05) change in baseline activity between ED10 and ED1 and 54% (53/98) of these units exhibited an increase in baseline activity at ED10 compared to ED1 baseline. Following rechallenge MPD injection of 2.5 mg/kg on ED10, 68% of all DR units displayed a change in neuronal firing rate, with an equal number of units displaying a significant (p < 0.05) increase (50%, 48/96) and decrease (50%, 48/96) in their neuronal activity in response to MPD. (Table 4a)
3.2.7 DR activity recorded from animals expressing behavioral sensitization to 2.5 mg/kg (Table 4b)
Sixty two DR units were recorded from animals expressing behavioral sensitization to repetitive doses of 2.5 mg/kg MPD (Table 4b). On ED1 following acute injection, 89% (55/62) of the DR units responded with a significant (p < 0.05) change in firing rate, with 80% (44/55) of the responsive units exhibiting a significant (p < 0.05) increase in their neuronal activity compared to baseline activity. Eighty four percent (58/62) of these DR units displayed a significant (p < 0.05) change in baseline activity between ED10 and ED1, with 84% (49/58) of these unit displaying an increase in firing rate prior to injection on ED10 compared to ED1. In response to rechallenge MPD injection at ED10, 95% (59/62) of DR units experienced a significant (p < 0.05) change in neuronal activity with a majority (81%, 48/59) exhibiting an increase in firing rate. (Table 4b)
3.2.8 DR activity recorded from animals expressing behavioral tolerance to 2.5 mg/kg (Table 4c)
Eighty units were recorded from rats expressing behavioral tolerance to repetitive doses of 2.5 mg/kg MPD (Table 4c). Of these units, 51% (41/80) displayed a significant (p < 0.05) change in firing rate in response to MPD dose on ED1, with 54% (22/41) of the responsive units exhibiting a decrease in activity. A significant (p < 0.05) change in baseline activity on ED10 compared to baseline activity on ED1 was observed for 63% (50/80) of these units, with a majority (72%, 36/50) of these units displaying decreased activity. Upon administration of the MPD rechallenge dose on day 10, 61% (49/80) DR units significantly (p < 0.05) change firing activity, with 76% (37/49) DR units exhibiting decreased neuronal activity. (Table 4c)
3.2.9 Effect of 10.0 mg/kg MPD on DR units of all animals (Table 5a)
Table 5. summarizes the DR neuronal activity following 10.0 mg/kg MPD.
Table 5 summarizes the effects of acute and chronic injection of 10.0 mg/kg MPD on DR neuronal activity. Section a. summarizes neuronal activity of DR units recorded from all rats, section b. summarizes activity recorded only from animals exhibiting behavioral sensitization, and section c. summarizes activity recorded only from tolerant animals.
 indicates a significant (p < 0.05) increase in activity following MPD treatment,
indicates a significant (p < 0.05) increase in activity following MPD treatment,
 indicates a significant (p < 0.05) decrease in activity following treatment, and
indicates a significant (p < 0.05) decrease in activity following treatment, and
 indicates no change.
indicates no change.
| Dose | Population | Response direction | Acute ED1 | Baseline ED10 to ED1 | Rechallenge ED10 |
|---|---|---|---|---|---|
| 10.0 mg/kg | |||||
| a. Total N = 153 |
|
67 (43.8%) | 68 (44.4%) | 65 (42.5%) | |
|
|
55 (35.9%) | 63 (41.2%) | 60 (39.2%) | ||
|
|
31 (20.3%) | 22 (14.4%) | 28 (18.3%) | ||
| b. Behavioral Sensitization N = 98 |
|
50 (51.0%) | 63 (64.2%) | 58 (59.1%) | |
|
|
33 (33.7%) | 29 (29.6%) | 35 (35.7%) | ||
|
|
15 (15.3%) | 6 (6.1%) | 5 (5.1%) | ||
| c. Behavioral Tolerance N = 55 |
|
20 (36.4%) | 7 (12.7%) | 6 (10.9%) | |
|
|
17 (30.9%) | 34 (61.8%) | 24 (43.6%) | ||
|
|
18 (32.7%) | 14 (25.4%) | 25 (45.5%) |
One hundred and fifty three DR units were recorded from animals receiving acute and chronic injections of 10.0 mg/kg (Table 5a). Following acute administration of MPD on ED1, 80% (122/153) of the DR units exhibited a significant (p < 0.05) change in firing rate, with the majority (55%, 67/122) of these units responding to the drug by increasing their neuronal activity. Baseline activity on ED10 compared to baseline activity on ED1 significantly (p < 0.05) changed for 86% (131/153) of these DR units, with 52% (68/131) of the units displaying an increase and 48% (63/131) displaying a decrease in baseline activity on ED10 compared to ED1 baseline. On ED10, 82% (125/153) of DR units responded to rechallenge MPD administration with a significant (p < 0.05) change in neuronal activity compared to the effect elicited by acute MPD at ED1, with 52% (65/125) of the responsive units increasing activity after MPD exposure. (Table 5a)
3.2.10 DR activity recorded from animals expressing behavioral sensitization to 10.0 mg/kg (Table 5b)
Ninety eight DR units were recorded from animals exhibiting behavioral sensitization to repetitive (chronic) administration of 10.0 mg/kg MPD (Table 5b). In response to acute MPD administration, 85% (83/98) of the DR units significantly changed firing activity, with the majority (60%, 50/83) of the responsive units exhibiting a significant (p < 0.05) increase in their firing rate. Six daily doses of 10.0 mg/kg MPD followed by 3 washout days resulted in a significant (p < 0.05) change in baseline activity on ED10 compared to ED1 for 94% (92/98) of the units, with 68% (63/92) of the units increasing baseline activity. Ninety five percent (95%, 93/98) of the DR units responded to rechallenge MPD dose with a significant (p < 0.05) change in activity, and the majority (62%, 58/93) displayed a further increase in their firing activity compared to the activity recorded following 10.0 mg/kg MPD on ED1. (Table 5b)
3.2.11 DR activity recorded from animals expressing behavioral tolerance to 10.0 mg/kg (Table 5c)
Fifty five DR units were recorded from animals expressing behavioral tolerance to repetitive doses of 10.0 mg/kg MPD (Table 5c). Following acute MPD administration, 67% (37/55) of the DR units recorded from tolerized animals changed firing rate, with 54% (20/37) of the responding units exhibiting an increase in firing rate in response to MPD exposure. The baseline activity on ED10 compared to the baseline activity on ED1 significantly (p < 0.05) changed for 75% (41/55) units, with a majority (83%, 34/41) of the modulated units displaying a decrease in baseline activity. On ED10, 55% (30/55) of the DR units responded to rechallenge MPD administration with a significant (p < 0.05) change in neuronal activity, with 80% (24/30) of the responsive units exhibiting a decrease in firing rate compared to the initial MPD effect. (Table 5c)
4. Discussion
Methylphenidate is the most widely prescribed medication for ADHD treatment, but the neuronal mechanisms underlying its action on the CNS remain unclear. The objective of this study was to test the hypothesis that the DR participates at least in part in the effects of MPD. The main findings of this study were 1) the same dose (0.6, 2.5, or 10.0mg/kg) of MPD elicits behavioral tolerance in some rats and sensitization in others. 2) MPD elicits a dose dependent effect on DR neuronal activity, with increasing doses causing an increase in the proportion of DR units responding with a significant change in firing rate 3) DR neurons recorded from animals exhibiting behavioral sensitization to chronic MPD treatment are significantly more likely to exhibit an increased firing rate following chronic MPD exposure compared to the effect of acute MPD injection, while DR units recorded from behaviorally tolerant animals tended to respond to chronic MPD with a decrease or no change in firing rate compared to the DR acute MPD response. These observations support our hypotheses and indicate that the DR participates at least in part in the effects of MPD.
4.1 MPD effects on locomotor behavior
Adolescent rats exposed to identical doses of MPD showed contrasting behavioral responses to chronic treatment. Acute injection of 0.6, 2.5, or 10.0 mg/kg on ED1 significantly increased locomotor activity in all test groups in a dose response characteristic. This is consistent with previous studies using the same MPD dosages (Claussen et al., 2014; Frolov et al., 2015). Following 6 daily MPD injections and 3 washout days, the animals received a rechallenge MPD dose on ED10. The group receiving 0.6 mg/kg MPD responded similarly to the initial exposure. The group who received 2.5 and 10.0 mg/kg MPD rechallenge on ED10 responded with an increase in activity that was significantly higher than the increase observed following treatment on ED1. Each animal in this study was also evaluated independently based on their individual behavioral response to chronic MPD exposure compared to their initial response. Some animals were found to have a significantly attenuated response to MPD on ED10 compared to the initial dose on ED1 while other animals exhibited a further significant increased response to MPD on ED10 compared to ED1. These observations support the hypothesis that the same repetitive dose of MPD elicits behavioral sensitization in some rats and behavioral tolerance in others.
4.2 MPD effects on DR neuronal activity
As the acute dose of MPD was increased from 0.6 to 10.0 mg/kg, more DR neurons responded by changing their firing rate. The baseline (post-saline) activity of these units on ED10 compared to ED1 baseline activity also showed dose-dependent increases in the number of units whose baseline was changed. Initial analysis of all recorded DR units as one group showed that for each MPD dose used, about one third of the units responded to MPD with an increase, decrease, or no change in firing rate respectively. However, when the DR neuronal responses were evaluated based on the animals’ behavioral response to chronic MPD and sorted into neuronal recordings obtained from behaviorally sensitized or from behaviorally tolerant subgroups, contrasting trends were observed between the two groups. While a majority of DR units recorded from the behaviorally sensitized subgroup exhibited an increase in neuronal activity following MPD rechallenge on ED10 compared to their response to initial MPD exposure on ED1, most units recorded from animals categorized as behaviorally tolerant exhibited a decrease in firing rate in response to MPD rechallenge at ED10 compared to the acute response to MPD exposure. The dichotomy between the initial observation from all animals which did not exhibit a clear response pattern and the statistically significant trends observed when the DR recordings were analyzed based on behavioral subgroup suggests that it is essential to evaluate the neuronal activity in the context of the animals’ behavioral response to chronic MPD exposure.
The DR, in addition to serotonergic neurons, is also populated by inhibitory GABA interneurons as well as small populations of glutamatergic, dopaminergic, and substance P- containing neurons (Allers and Sharp, 2003). Studies investigating the electrophysiological properties of DR neurons in vitro found that DR serotonergic neurons exhibit a slow, regular firing pattern and have a longer duration action potential compared to non-serotonergic DR neurons, (Vandermaelen and Aghajanian, 1983; Jacobs and Fornal, 1991; Allers and Sharp, 2003). Based on the low firing rate and the spike duration that was used to sort and analyze the recorded spikes, it is likely that most of the units included in this study are serotonergic neurons. The use of freely behaving rats and recordings from the same permanent semi micro (60μm) implanted electrodes on both ED1 and ED10 complicates discrimination of 5HT neurons from non-5HT neurons based on electrophysiology alone. Further studies are needed to identify the specific neuronal subtypes within the DR that participate in MPD actions.
4.3 Correlations between DR neuronal activity and behavioral phenotype
MPD is unique among psychostimulants in that, unlike amphetamine and cocaine, it does not bind to the 5HT transporter with high affinity (Pan et al., 1994; Gatley et al., 1996) and it is because of this that serotonin is rarely investigated as an effector of MPD action, despite evidence supporting 5HT involvement in the neurophysiology of ADHD (Cools et al., 2008; Dalley and Roiser, 2012). In contrast to amphetamine treatment, MPD exposure produces no direct increase in extracellular 5HT levels in the caudate, putamen, or nucleus accumbens (Segal and Kuczenski, 1999; Koda et al., 2010). However, in a high-performance liquid chromatography (HPLC) assay study, MPD exposure has been shown to decrease serotonin levels in the rat prefrontal cortex, dorsal striatum, and hippocampus, suggesting that MPD indirectly affects 5HT content via its actions on NE and DA transmission in the CNS and that this alteration in 5HT has a role in the behavioral effects of MPD (Borycz et al., 2008; Ruocco et al., 2010).
The DR units recorded from rats exhibiting behavioral tolerance had a larger proportion of units that exhibited a decreased response to rechallenge MPD on ED10 compared to the acute response on ED1, while DR units recorded from behaviorally sensitized rats were more likely to respond to MPD on ED10 with a further increase in firing rate compared to the acute response. It has been reported that the firing rate of serotonergic neurons is regulated in part by inhibitory autoreceptors that act in a negative feedback loop to inhibit further 5HT release when extracellular 5HT is elevated (Filip et al., 2005). Although it does not have strong affinity for the 5HT transporter, MPD may affect serotonin transmission by direct interaction with serotonergic autoreceptors. There are seven major classes of 5HT receptor in the CNS and seventeen different subtypes, each with a different target and effect (Filip et al., 2005). Markowitz et al. (2009) reported that MPD selectively binds to and exhibits agonist effects at 5HT1a inhibitory autoreceptors found on serotonergic neurons in the CNS. In vivo, the 5HT1a receptor has numerous neuroregulatory effects, including autoinhibition of serotonergic neurons as well as modulation of dopaminergic activity in the mesolimbic system, and there is evidence suggesting that low dose 5HT1a agonists improve learning and cognitive performance (Barnes and Sharp, 1999; King et al., 2008). 5HT1a agonists injected into adult rat brain slices have been shown to result in a decreased frequency of DR neuron firing and reduced 5HT release (Davidson and Stamford, 1995) and chronic treatment with MPD followed by 5HT1a agonist 8-OH-DPAT resulted in an increase in sensitivity to the agonist (Alam et al., 2016) and a dose dependent decrease in extracellular 5HT in the ventral tegmental area (VTA), a region important in drug reward and reinforcement (Kauer, 2004).
Studies have also reported that MPD induces an increase in DR neuronal activity directly and as measured by the expression of Fos-like immunoreactivity in DR neurons following treatment (Tang and Dafny 2013a; Tang and Dafny 2013b; Trinh et al., 2003). Adolescent mice have been shown to increase Fos expression in DR relative to other brain regions (Karanges et al., 2016). The activation is due, at least in part, to changes in DA transmission as mutant mice that lacked DAT expression did not display an increase expression of Fos-like immunoreactivity in DR neurons (Trinh et al., 2003). This is consistent with the reports that the elevation in extracellular dopamine elicited by MPD is crucial to the induction and expression of behavioral sensitization (Jones et al., 1998).
In summary, the present study showed that the same doses of MPD exposure for 6 consecutive days followed by 3 washout days and rechallenge with the same MPD dose on ED10 elicited behavioral tolerance in some adolescent animals and behavioral sensitization in others and that the correlation between the behavioral phenotypes induced by chronic MPD and the DR neuronal activity suggests underlying neurochemical differences between the animals exhibiting behavioral sensitization and those exhibiting tolerance. It is possible that in behaviorally sensitized rats the excitatory effects of MPD override its inhibitory effects as a 5HT1a agonist to a greater extent than in behaviorally tolerant rats. The behavioral and neurophysiological observations obtained in this study show that the DR participates at least in part in the acute effects as well as the chronic effects (sensitization and tolerance) of MPD in adolescent rats.
Highlights.
The same MPD dose elicits either behavioral tolerance or sensitization.
MPD elicits a dose dependent effect on DR neuronal activity.
DR neuronal activity further increased in sensitized rats (chronic MPD).
DR neuronal activity decreased or remained unchanged in tolerant rats (chronic MPD).
Correlation between DR neuronal activity and behavioral response.
Acknowledgments
We would like to thank Alice Z. Chuang for statistical assistance. This study was supported by NIH ROI DA027222 grant.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Accardo P, Blondis TA. What's all the fuss about Ritalin? The Journal of pediatrics. 2001;138:6–9. doi: 10.1067/mpd.2001.111505. [DOI] [PubMed] [Google Scholar]
- Alam N, Najam R, Naeem S. Attenuation of methylphenidate-induced sensitization by co-administration of buspirone. Pak J Pharm Sci. 2016;29(2):585–90. [PubMed] [Google Scholar]
- Algahim MF, Yang PB, Wilcox VT, Burau KD, Swann AC, Dafny N. Prolonged methylphenidate treatment alters the behavioral diurnal activity pattern of adult male Sprague-Dawley rats. Pharmacology, biochemistry, and behavior. 2009;92:93–99. doi: 10.1016/j.pbb.2008.10.021. [DOI] [PubMed] [Google Scholar]
- Allers KA, Sharp T. Neurochemical and anatomical identification of fast- and slow-firing neurones in the rat dorsal raphe nucleus using juxtacellular labelling methods in vivo. Neuroscience. 2003;122:193–204. doi: 10.1016/s0306-4522(03)00518-9. [DOI] [PubMed] [Google Scholar]
- Andersen SL, Arvanitogiannis A, Pliakas AM, LeBlanc C, Carlezon WA. Altered responsiveness to cocaine in rats exposed to methylphenidate during development. Nature neuroscience. 2002;5:13–14. doi: 10.1038/nn777. [DOI] [PubMed] [Google Scholar]
- Arnsten AF, Dudley AG. Methylphenidate improves prefrontal cortical cognitive function through alpha2 adrenoceptor and dopamine D1 receptor actions: Relevance to therapeutic effects in Attention Deficit Hyperactivity Disorder. Behavioral and brain functions. 2005;1:2. doi: 10.1186/1744-9081-1-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arnsten AFT. Stimulants: Therapeutic actions in ADHD. Neuropsychopharmacology. 2006;31:2376–2383. doi: 10.1038/sj.npp.1301164. [DOI] [PubMed] [Google Scholar]
- Barnes NM, Sharp T. A review of central 5-HT receptors and their function. Neuropharmacology. 1999;38:1083–1152. doi: 10.1016/s0028-3908(99)00010-6. [DOI] [PubMed] [Google Scholar]
- Berridge CW, Devilbiss DM, Andrzejewski ME, Arnsten AFT, Kelley AE, Schmeichel B, Hamilton C, Spencer RC. Methylphenidate preferentially increases catecholamine neurotransmission within the prefrontal cortex at low doses that enhance cognitive function. Biological psychiatry. 2006;60:1111–1120. doi: 10.1016/j.biopsych.2006.04.022. [DOI] [PubMed] [Google Scholar]
- Bhat RV, Baraban JM. Activation of transcription factor genes in striatum by cocaine: role of both serotonin and dopamine systems. The Journal of pharmacology and experimental therapeutics. 1993;267:496–505. [PubMed] [Google Scholar]
- Bolaños CA, Barrot M, Berton O, Wallace-Black D, Nestler EJ. Methylphenidate treatment during pre- and periadolescence alters behavioral responses to emotional stimuli at adulthood. Biological psychiatry. 2003;54:1317–1329. doi: 10.1016/s0006-3223(03)00570-5. [DOI] [PubMed] [Google Scholar]
- Bolaños CA, Willey MD, Maffeo ML, Powers KD, Kinka DW, Grausam KB, Henderson RP. Antidepressant treatment can normalize adult behavioral deficits induced by early-life exposure to methylphenidate. Biological psychiatry. 2008;63:309–316. doi: 10.1016/j.biopsych.2007.06.024. [DOI] [PubMed] [Google Scholar]
- Borycz J, Zapata A, Quiroz C, Volkow ND, Ferré S. 5-HT 1B receptor-mediated serotoninergic modulation of methylphenidate-induced locomotor activation in rats. Neuropsychopharmacology. 2008;33:619–626. doi: 10.1038/sj.npp.1301445. [DOI] [PubMed] [Google Scholar]
- Claussen CM, Chong SL, Dafny N. Nucleus accumbens neuronal activity correlates to the animal's behavioral response to acute and chronic methylphenidate. Physiology & behavior. 2014;129:85–94. doi: 10.1016/j.physbeh.2014.02.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cools R, Roberts AC, Robbins TW. Serotoninergic regulation of emotional and behavioural control processes. Trends in cognitive sciences. 2008;12:31–40. doi: 10.1016/j.tics.2007.10.011. [DOI] [PubMed] [Google Scholar]
- Dalley JW, Roiser JP. Dopamine, serotonin and impulsivity. Neuroscience. 2012;215:42–58. doi: 10.1016/j.neuroscience.2012.03.065. [DOI] [PubMed] [Google Scholar]
- Davidson C, Stamford JA. Evidence that 5-hydroxytryptamine release in rat dorsal raphé nucleus is controlled by 5-HT1A, 5-HT1B and 5-HT1D autoreceptors. British journal of pharmacology. 1995;114:1107–1109. doi: 10.1111/j.1476-5381.1995.tb13321.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Filip M, Frankowska M, Zaniewska M, Gołda A, Przegaliński E. The serotonergic system and its role in cocaine addiction. Pharmacological reports. 2005;57:685–700. [PubMed] [Google Scholar]
- Frolov A, Reyes-Vasquez C, Dafny N. Behavioral and neuronal recording of the nucleus accumbens in adolescent rats following acute and repetitive exposure to methylphenidate. Journal of neurophysiology. 2015;113:369–379. doi: 10.1152/jn.00633.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gatley SJ, Pan D, Chen R, Chaturvedi G, Ding YS. Affinities of methylphenidate derivatives for dopamine, norepinephrine and serotonin transporters. Life sciences. 1996;58:231–239. doi: 10.1016/0024-3205(96)00052-5. [DOI] [PubMed] [Google Scholar]
- Gatley SJ, Volkow ND, Gifford AN, Fowler JS, Dewey SL, Ding YS, Logan J. Dopamine-transporter occupancy after intravenous doses of cocaine and methylphenidate in mice and humans. Psychopharmacology. 1999;146:93–100. doi: 10.1007/s002130051093. [DOI] [PubMed] [Google Scholar]
- Gaytan O, Ghelani D, Martin S, Swann A, Dafny N. Dose response characteristics of methylphenidate on different indices of rats' locomotor activity at the beginning of the dark cycle. Brain research. 1996;727:13–21. doi: 10.1016/0006-8993(96)00296-x. [DOI] [PubMed] [Google Scholar]
- Homberg JR, Kyzar EJ, Scattoni ML, Norton WH, Pittman J, Gaikwad S, Nguyen M, Poudel MK, Ullmann JF, Diamond DM, Kaluyeva AA, Parker MO, Brown RE, Song C, Gainetdinov RR, Gottesman II, Kalueff AV. Genetic and environmental modulation of neurodevelopmental disorders: Translational insights from labs to beds. Brain Res Bull. 2016;125:79–91. doi: 10.1016/j.brainresbull.2016.04.015. [DOI] [PubMed] [Google Scholar]
- Jacobs BL, Fornal CA. Activity of brain serotonergic neurons in the behaving animal. Pharmacological reviews. 1991;43:563–578. [PubMed] [Google Scholar]
- Jones SR, Gainetdinov RR, Jaber M, Giros B, Wightman RM, Caron MG. Profound neuronal plasticity in response to inactivation of the dopamine transporter. Proceedings of the National Academy of Sciences of the United States of America. 1998;95:4029–4034. doi: 10.1073/pnas.95.7.4029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones Z, Reyes-Vazquez C, Dafny N. Ventral Tegmental Area Neuronal Activity Correlates to Animals’ Behavioral Response to Chronic Methylphenidate Recorded from Adolescent SD Male Rats. Journal of Behavioral and Brain Science Volume. 2014;4:168–189. [Google Scholar]
- Karanges EA, Ramos L, Dampney B, Suraev AS, Li KM, McGregor IS, Hunt GE. Contrasting regional Fos expression in adolescent and young adult rats following acute administration of the antidepressant paroxetine. Brain Res Bull. 2016;121:246–54. doi: 10.1016/j.brainresbull.2016.02.008. [DOI] [PubMed] [Google Scholar]
- Kauer JA. Learning mechanisms in addiction: synaptic plasticity in the ventral tegmental area as a result of exposure to drugs of abuse. Annual review of physiology. 2004;66:447–475. doi: 10.1146/annurev.physiol.66.032102.112534. [DOI] [PubMed] [Google Scholar]
- King MV, Marsden CA, Fone KCF. A role for the 5-HT(1A), 5-HT4 and 5-HT6 receptors in learning and memory. Trends in pharmacological sciences. 2008;29:482–492. doi: 10.1016/j.tips.2008.07.001. [DOI] [PubMed] [Google Scholar]
- Koda K, Ago Y, Cong Y, Kita Y, Takuma K, Matsuda T. Effects of acute and chronic administration of atomoxetine and methylphenidate on extracellular levels of noradrenaline, dopamine and serotonin in the prefrontal cortex and striatum of mice. Journal of neurochemistry. 2010;114:259–270. doi: 10.1111/j.1471-4159.2010.06750.x. [DOI] [PubMed] [Google Scholar]
- Krain AL, Castellanos FX. Brain development and ADHD. Clinical psychology review. 2006;26:433–444. doi: 10.1016/j.cpr.2006.01.005. [DOI] [PubMed] [Google Scholar]
- Kuczenski R, Segal DS. Effects of methylphenidate on extracellular dopamine, serotonin, and norepinephrine: comparison with amphetamine. Journal of neurochemistry. 1997;68:2032–2037. doi: 10.1046/j.1471-4159.1997.68052032.x. [DOI] [PubMed] [Google Scholar]
- Lee M, Dafny N, DS Cocaine Alters the Daily Activity Patterns of Adult Rats. Journal of Behavioral and Brain Science. 2014;4(11):523. [Google Scholar]
- Miyazaki KW, Miyazaki K, Doya K. Activation of dorsal raphe serotonin neurons is necessary for waiting for delayed rewards. The Journal of neuroscience : the official journal of the Society for Neuroscience. 2012;32:10451–10457. doi: 10.1523/JNEUROSCI.0915-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newcorn JH. A glimpse into key issues in ADHD. CNS spectrums. 2000;5:25. doi: 10.1017/s1092852900007033. [DOI] [PubMed] [Google Scholar]
- Oades RD. Dopamine-serotonin interactions in attention-deficit hyperactivity disorder (ADHD) Progress in brain research. 2008;172:543–565. doi: 10.1016/S0079-6123(08)00926-6. [DOI] [PubMed] [Google Scholar]
- Pan D, Gatley SJ, Dewey SL, Chen R, Alexoff DA, Ding YS, Fowler JS. Binding of bromine-substituted analogs of methylphenidate to monoamine transporters. European journal of pharmacology. 1994;264:177–182. doi: 10.1016/0014-2999(94)00460-9. [DOI] [PubMed] [Google Scholar]
- Polanczyk G, Rohde LA. Epidemiology of attention-deficit/hyperactivity disorder across the lifespan. Current opinion in psychiatry. 2007;20:386–392. doi: 10.1097/YCO.0b013e3281568d7a. [DOI] [PubMed] [Google Scholar]
- Porras G, Di Matteo V, Fracasso C, Lucas G, De Deurwaerdère P, Caccia S, Esposito E, Spampinato U. 5-HT2A and 5-HT2C/2B receptor subtypes modulate dopamine release induced in vivo by amphetamine and morphine in both the rat nucleus accumbens and striatum. Neuropsychopharmacology. 2002;26:311–324. doi: 10.1016/S0893-133X(01)00333-5. [DOI] [PubMed] [Google Scholar]
- Robinson TE, Berridge KC. Incentive-sensitization and addiction. Addiction (Abingdon, England) 2001;96:103–114. doi: 10.1046/j.1360-0443.2001.9611038.x. [DOI] [PubMed] [Google Scholar]
- Ruocco LA, Carnevale UAG, Treno C, Sadile AG, Melisi D, Arra C, Ibba M, Schirru C, Carboni E. Prepuberal subchronic methylphenidate and atomoxetine induce different long-term effects on adult behaviour and forebrain dopamine, norepinephrine and serotonin in Naples high- excitability rats. Behavioural brain research. 2010;210:99–106. doi: 10.1016/j.bbr.2010.02.020. [DOI] [PubMed] [Google Scholar]
- Segal DS, Kuczenski R. Escalating dose-binge treatment with methylphenidate: role of serotonin in the emergent behavioral profile. The Journal of pharmacology and experimental therapeutics. 1999;291:19–30. [PubMed] [Google Scholar]
- Sherwood NM, Timiras PS. A Stereotaxic Atlas of the Developing Rat Brain. Univ of California Press; 1970. [Google Scholar]
- Tang B, Dafny N. Behavioral and dorsal raphe neuronal activity following acute and chronic methylphenidate in freely behaving rats. Brain Res Bull. 2013a;98:53–63. doi: 10.1016/j.brainresbull.2013.06.004. [DOI] [PubMed] [Google Scholar]
- Tang B, Dafny N. Dorsal raphe neuronal activities are modulated by methylphenidate. J Neural Trans. 2013b;120:721–731. doi: 10.1007/s00702-012-0917-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tao R, Auerbach SB. Involvement of the dorsal raphe but not median raphe nucleus in morphine- induced increases in serotonin release in the rat forebrain. Neuroscience. 1995;68:553–561. doi: 10.1016/0306-4522(95)00154-b. [DOI] [PubMed] [Google Scholar]
- Trinh JV, Nehrenberg DL, Jacobsen JPR, Caron MG, Wetsel WC. Differential psychostimulant- induced activation of neural circuits in dopamine transporter knockout and wild type mice. Neuroscience. 2003;118:297–310. doi: 10.1016/s0306-4522(03)00165-9. [DOI] [PubMed] [Google Scholar]
- Van Waes V, Beverley J, Marinelli M, Steiner H. Selective serotonin reuptake inhibitor antidepressants potentiate methylphenidate (Ritalin)-induced gene regulation in the adolescent striatum. The European journal of neuroscience. 2010;32:435–447. doi: 10.1111/j.1460-9568.2010.07294.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vandermaelen CP, Aghajanian GK. Electrophysiological and pharmacological characterization of serotonergic dorsal raphe neurons recorded extracellularly and intracellularly in rat brain slices. Brain research. 1983;289:109–119. doi: 10.1016/0006-8993(83)90011-2. [DOI] [PubMed] [Google Scholar]
- Volkow ND1, Ding YS, Fowler JS, Wang GJ, Logan J, Gatley JS, Dewey S, Ashby C, Liebermann J, Hitzemann R. Is methylphenidate like cocaine? Studies on their pharmacokinetics and distribution in the human brain. Arch Gen Psychiatry. 1995;52(6):456–63. doi: 10.1001/archpsyc.1995.03950180042006. [DOI] [PubMed] [Google Scholar]
- Volkow ND, Gatley SJ, Fowler JS, Wang GJ, Swanson J. Serotonin and the therapeutic effects of ritalin. Science. 2000;288:11. doi: 10.1126/science.288.5463.11a. [DOI] [PubMed] [Google Scholar]
- Wilens TE. Effects of methylphenidate on the catecholaminergic system in attention- deficit/hyperactivity disorder. Journal of clinical psychopharmacology. 2008;28:S46–53. doi: 10.1097/JCP.0b013e318173312f. [DOI] [PubMed] [Google Scholar]
- Yang PB, Atkins KD, Dafny N. Behavioral sensitization and cross-sensitization between methylphenidate amphetamine, and 3,4-methylenedioxymethamphetamine (MDMA) in female SD rats. European journal of pharmacology. 2011;661:72–85. doi: 10.1016/j.ejphar.2011.04.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yano M, Steiner H. Topography of methylphenidate (ritalin)-induced gene regulation in the striatum: differential effects on c-fos, substance P and opioid peptides. Neuropsychopharmacology : official publication of the American College of Neuropsychopharmacology. 2005;30:901–915. doi: 10.1038/sj.npp.1300613. [DOI] [PubMed] [Google Scholar]